
The Project Gutenberg EBook of Scientific American Supplement, No. 401, September 8, 1883, by Various This eBook is for the use of anyone anywhere at no cost and with almost no restrictions whatsoever. You may copy it, give it away or re-use it under the terms of the Project Gutenberg License included with this eBook or online at www.gutenberg.org Title: Scientific American Supplement, No. 401, September 8, 1883 Author: Various Posting Date: October 10, 2012 [EBook #8718] Release Date: August, 2005 First Posted: August 3, 2003 Language: English Character set encoding: ISO-8859-1 *** START OF THIS PROJECT GUTENBERG EBOOK SCIENTIFIC AMERICAN SUPPL., NO. 401 *** Produced by Olaf Voss, Don Kretz, Juliet Sutherland, Charles Franks and the Distributed Proofreaders Team
The cultivated and patriotic city of Barcelona is about to erect a magnificent monument in honor of Columbus, the personage most distinguished in the historic annals of all nations and all epochs. The City of Earls does not forget that here the discoverer of America disembarked on the 3d of April, 1493, to present to the Catholic monarchs the evidences of the happy termination of his enterprise. In honoring Columbus they honor and exalt the sons of Catalonia, who also took part in the discovery and civilization of the New World, among whom may be named the Treasurer Santangel, Captain Margarit, Friar Benardo Boyl, first patriarch of the Indies, and the twelve missionaries of Monserrat, who accompanied the illustrious admiral on his second voyage.
In September, 1881, a national competition was opened by the central executive committee for the monument, and by the unanimous voice of the committee the premium plans of the architect, Don Cayetano Buigas Monraba, were adopted. From these plans, which we find in La Ilustracion Española, we give an engraving. Richness, grandeur, and expression, worthily combined, are the characteristics of these plans. The landing structure is divided into three parts, a central and two laterals, each of which extends forward, after the manner of a cutwater, in the form of the bow of a vessel of the fifteenth century, bringing to mind the two caravels, the Pinta and Niña; two great lights occupy the advance points on each side; a rich balustrade and four statues of celebrated persons complete the magnificent frontage. A noble monument, surmounted by a statue of the discoverer, is seen on the esplanade.
MONUMENTAL LANDING AND STATUE TO COLUMBUS, AT BARCELONA, SPAIN.
The commission appointed in France to consider the phylloxera has not awarded to anybody the prize of three hundred thousand francs that was offered to the discoverer of a trustworthy remedy or preventive for the fatal grape disease. There were not less than 182 competitors for the prize; but none had made a discovery that filled the bill. It is said, however, that a Strasbourg physician has found in naphthaline an absolutely trustworthy remedy. This liquid is poured upon the ground about the root of the vine, and it is said that it kills the parasites without hurting the grape.
Mr. R.W. Raymond gives the following interesting account of the remarkable scenery on this recently opened route from Denver to Salt Lake:
Having just made the trip from Salt Lake City to this place on the Denver & Rio Grande line, I cannot write you on any other subject at present. There is not in the world a railroad journey of thirty hours so filled with grand and beautiful views. I should perhaps qualify this statement by deducting the hours of darkness; yet this is really a fortunate enhancement of the traveler's enjoyment; it seems providential that there is one part of the way just long enough and uninteresting enough to permit one to go to sleep without the fear of missing anything sublime. Leaving Salt Lake City at noon, we sped through the fertile and populous Jordan Valley, past the fresh and lovely Utah Lake, and up the Valley of Spanish Fork. All the way the superb granite walls and summits of the Wahsatch accompanied us on the east, while westward, across the wide valley, were the blue outlines of the Oquirrh range. One after another of the magnificent cañons of the Wahsatch we passed, their mouths seeming mere gashes in the massive rock, but promising wild and rugged variety to him who enters--a promise which I have abundantly tested in other days. Parley's Cañon, the Big and Little Cottonwood, and most wonderful of all, the cañon of the American Fork, form a series not inferior to those of Boulder, Clear Creek, the Platte, and the Arkansas, in the front range of the Rockies.
Following Spanish Fork eastward so far as it served our purpose, we crossed the divide to the head waters of the South Fork of Price River, a tributary of Green River. It was a regret to me, in choosing this route, that I should miss the familiar and beloved scenery of Weber and Echo cañons--the only part of the Union Pacific road which tempts one to look out of a car window, unless one may be tempted by the boundless monotony of the plains or the chance of a prairie dog. Great was my satisfaction, therefore, to find that this part of the new road, parallel with the Union Pacific, but a hundred miles farther south, traverses the same belt of rocks, and exhibits them in forms not less picturesque. Castle Cañon, on the South Fork of the Price, is the equivalent of Echo Cañon, and is equal or superior in everything except color. The brilliant red of the Echo cliffs is wanting. The towers and walls of Castle Cañon are yellowish-gray. But their forms are incomparably various and grotesque--in some instances sublime. The valley of Green River at this point is a cheerless sage-brush desert, as it is further north. To be sure, this uninviting stream, a couple of hundred miles further south, having united with the Grande, and formed the Rio Colorado, does indeed, by dint of burrowing deeper and deeper into the sunless chasms, become at last sublime. But here it gives no hint of its future somber glory. I remained awake till we had crossed Green River, to make sure that no striking scenery should be missed by sleep. But I got nothing for my pains except the moonlight on the muddy water; and next time I shall go to bed comfortably, proving to the conductor that I am a veteran and not a tender-foot.
In the morning, we breakfasted at Cimarron, having in the interval passed the foot-hills of the Roan Mountains, crossed the Grande, and ascended for some distance the Gunnison, a tributary of the Grande, the Uncompahgre, a tributary of the Gunnison, and finally a branch, flowing westward, of the Uncompahgre. A high divide at the head of the latter was laboriously surmounted; and then, one of our two engines shooting ahead and piloting us, we slid speedily down to Cimarron. It is in such descents that the unaccustomed traveler usually feels alarmed. But the experience of the Rio Grande Railroad people is, that derailment is likely to occur on up-grades, and almost never in going down.
From this point, comparison with the Union Pacific line in the matter of scenery ceases. As everybody knows, that road crosses the Rocky Mountains proper in a pass so wide and of such gradual ascent that the high summits are quite out of sight. If it were not for the monument to the Ameses, there would be nothing to mark the highest point. For all the wonderful scenery on the Rio Grande road, between Cimarron and Pueblo, the Union Pacific in the same longitudes has nothing to show. From an artistic stand-point, one road has crossed the ranges at the most tame and uninteresting point that could be found, and the other at the most picturesque.
At Cimarron, the road again strikes the Gunnison, and plunges into the famous Black Cañon. In length, variety, and certain elements of beauty, such as forest-ravines and waterfalls, this cañon surpasses the Royal Gorge of the Arkansas. There is, however, one spot in the latter (I mean, of course, the point where the turbulent river fills the whole space between walls 2,800 ft. high, and the railroad is hung over it) which is superior in desolate, overwhelming grandeur to anything on the Gunnison. Take them all in all, it is difficult to say which is the finer. I have usually found the opinion of travelers to favor the Gunnison Cañon. But why need the question be solved at all? This one matchless journey comprises them both; and he who was overwhelmed in the morning by the one, holds his breath in the afternoon before the mighty precipices of the other. To excuse myself from even hinting such folly as a comparison of scenery, I will merely remark that these two cañons are more capable of a comparison than different scenes usually are; for they belong to the same type--deep cuts in crystalline rocks.
Between them come the Marshall Pass (nearly 11,000 ft. above sea-level), over the continental divide, and the Poncha Pass, over the Sangre di Cristo range. This range contains Harvard, Yale, Princeton, Elbert, Massive (the peak opposite Leadville), and other summits exceeding the altitude of 14,000 ft. To the east of it is the valley of the Arkansas, into which and down which we pass, and so through the Royal Gorge to Cañon City and Pueblo, where we arrived before dark on the day after leaving Salt Lake.
Salt Lake, the Jordan Valley, Utah Lake, the Wahsatch, Castle Cañon, the Black Cañon of the Gunnison, Marshall Pass, Poncha Pass, the Arkansas Valley, the Royal Gorge--what a catalogue for so brief a journey! No wonder everybody who has made it is "wild about it!" If enthusiastic urgency of recommendation from every passenger has any influence (and I know it has a great deal), this road will continue to be, as it is at present, crowded with tourists. It furnishes a delightful route for those who wish on the overland journey to see Denver (as who does not?) and to visit Colorado Springs and Manitou. All this can be done en route, without retracing the steps.
In the natural course of things it must necessarily have occurred to practical men to utilize photography in the case of terra-cotta, as it has already been employed in connection with so many other wares; but I have not to this day known of its successful application to terra-cotta. Now this is strange, if one considers how fashionable plaque and plate painting have become of late, and the good photographic results that are easily obtained on these as on sundry articles of this same "burnt earth." Portraits, animals, landscapes, seascapes, and reproductions are one and all easily transferred, whether for painting upon or to be left purely photographic. As a matter of business, too, one fails to see that it would not be remunerative, but rather the contrary. It was with something of this feeling that I was led to try and see what could be done to attain the end in view, and as I knew of no data to go by, I had to use my own experience, or rather experiment on my own account.
Since emulsion was constantly at hand in my establishment, in the commercial production of my gelatine dry plates, it was but natural I should first have turned to this as a mode of obtaining the desired results; but, alas! all attempts in that direction signally failed--the ware most persistently refused to have anything to do with emulsion. The bugbear was the fixing agent or hypo., which not only left indelible marks, but, despite any amount of washing, the image on a finished plate vanished to nothing at the end of an hour's exposure in the show window. There was nothing left but to seek other means for the attainment of my object. I would not have troubled the reader as to this unsuccessful line of experiment but that I wished to put him on his guard and save him useless researches in the same direction. To cut matters short, the method I found best and most direct was the now old but still excellent wet collodion transfer. I will now proceed to detail my system of working to facilitate the matter to the inexperienced in collodion transfer.
The first and indispensable operation, in the preparation of the surface to receive the transfer, is the "sizing of the surface." It simply consists of a solution of gelatine chrome-alumed, as follows:
Gelatine. 10 grains. Water. 1 ounce. A trace of chrome alum.
Coat with a soft camel's hair brush and let dry. It is needless to say that numbers of plaques, plates, vases, etc., may be coated right off, and will then be ready for use at any time.
Having settled on the subject and carefully dusted the negative, as well as placed it in situ for reproduction, the next thing required is a suitable collodion, and the following will be found all that can be desired:
TRANSFER COLLODION.
Cotton. 3 drachms. Iodide of cadmium. 65 grains. Ammonium iodide. 25 " Bromide of cadmium. 19 " Ammonium bromide. 11 " Alcohol. 15 ounces. Ether. 15 "
The plate thoroughly cleaned and coated with the collodion is now transferred to a bath, as follows:
Nitrate of silver (common) 25 grains to the ounce.
Made slightly acid with nitric acid.
After sensitizing, the plate is exposed in the usual way and taken to the room where pictures are ordinarily developed, and quantum suff. of the following poured into the developing cup to bring out the image:
DEVELOPING SOLUTION.
A Winchester of water, i.e. 80 ounces. Protosulphate of iron. 240 grains. Citric acid. 240 "
Or the following may be used:
Pyro 3 grains\ Citric acid 2 " } per ounce of water. Glacial acetic acid 30 drops /
After perfect development the picture is well washed and then fixed in a saturated solution of hypo.; after which it is thoroughly washed.
It will now be found that the picture is not altogether satisfactory; it lacks both vigor and color. To improve matters recourse is now had to
Gold. 1 grain. Water. 5 ounces.
With this a very fine depth is soon attained, and a nice picture the result. Leave out the toning, and only a poor, sunken-looking picture will be the outcome; but directly the toning bath is employed richness at once comes to the fore. I have, however, known of instances where the picture needed no toning.
This is still a secret with some in the profession. A limited number of workers have succeeded in bringing out good opals, and their modus operandi is kept from the many. Now this is a pity, when one considers the great charm attached to a good picture on opal, with pure whites and rich blacks, and in many localities the demand that might be created for them. Apart from their beauty, another charm attaches to opals--their absolute permanence; and this, it must be allowed, is no trifle. What, in fact, can be more painful to the worker who values his work, and sets store by it, than to feel it must ere long fade and pass into oblivion! A properly executed opal will no more fade than the glass pictures so common at one time, and which, wherever taken care of, are as perfect now as they were when first taken.
Now, excellent pictures are to be made on opals by means of emulsion; but I propose first taking the transfer method (mainly applicable to ground opal and canvas) as given above for pottery, since in practice it is found very ready, easy of manipulation, and safe. The details are much the same as above, and necessitate double transfer.
After the picture had been obtained on the plate (ordinary glass plate), and after thoroughly fixing, washing, and toning, the picture (and this, remember, is the case likewise with terra-cotta) then has to be loosened from its support, and this is done with a solution of sulphuric acid--one drachm to fifteen ounces of water--which is made to flow between the image and the glass, after which perfectly wash and mount. When the image is loosened a piece of tracing paper is put on the image, evened out, raised (assisted by some one else to hold the two opposite corners during the operation), and with the aid of the helper the picture is carefully centered, gently pressed out or down, and the transfer is so far effected. But what will happen, and does happen, in the case of vignettes, is impurity of the whites, when the picture becomes positively objectionable. Now the way to remedy this lies simply in the application, to the dirty-looking parts, of a solution of iodine dissolved in iodide of potassium to sherry color; after which, well wash and apply a weak solution of cyanide of potassium, and wash well again. This, by the way, is equally applicable to paper transfers; and it is to be remembered that the toning comes last of all. It is a rather difficult matter to clean a ground opal which has been used two or three times, and acid must then be had recourse to (nitric acid is as good as any); but by transferring from the support on the ground surface, all stains are at once avoided.
On the flushed glass, or on the pot metal (unground), after well cleaning the surface it should be covered with a substratum of egg. Then the picture is taken direct, not transferred; that is, the plate is exposed direct in the camera, regularly proceeded with, and, when dried, varnished with a pale negative varnish, or with dead varnish if intended for chalk or water-color. This, when a good negative is used, gives a remarkably fine picture, not requiring a vestige of retouching, and having likewise the invaluable advantage of being perfectly durable if varnished with the negative varnish. Moreover, on that, effective pictures may be made in oil with simply tinting.
A gentleman, who has a right to be considered a good judge in all art matters, on looking at one of these pictures transferred on flushed glass, said it was one of the finest productions of photography. He urged that negatives ad rem should be taken most carefully, and that, like the picture I showed him, they should be full of half-tone and detail, and yet have plenty of vigor. They should, he said, be robust in the high lights, have perfectly clear glass in the few points of deep shadows, and thus have powerful relief. Moreover, the negatives should be retouched only by a competent hand, and care taken that the likeness shall be in no way altered, which is so frequently the case now.
If done as thus suggested there is no doubt that remarkably fine pictures are to be produced on opal, whether ground or not. Most artistic results are to be obtained, and, with proper care, absolute permanency. In this age of keen competition, all have to think of what may be really recommended to one's clientèle, and likely to meet with approbation from strangers and friends when the picture has once been delivered; and I candidly think that the opal, of all, is the picture most likely to meet with this general approbation.
I hope I have left it clearly to be understood that the class of opal picture to which I have chiefly alluded is one that remains untouched after the transfer--that is, absolutely unpainted upon. It is pure photography in every sense of the word, and the resultant picture one hardly to be surpassed in any way. I have rather laid a stress on this point, well knowing how pictures are at times irretrievably ruined by the barbarous hand of would-be artists, who by far exceed the true artists in number; and the hint on retouching should not be lost sight of, either, at a period when the tendency is to stereotype every one in marble-like texture, or rather lack of texture, as if the face were devoid of all fleshiness and as hard and rigid as cast-iron. It might be wise to weigh this point carefully, and act upon it, before the enlightened public have raised a cry against the pernicious practice and made photographers smart for their want of applying timely remedial measures to a decided evil.
On reading the above again, fearing lest any misconception should arise in the mind of the reader, I deem it expedient, to clearly state that for terra-cotta recourse is had to double transfer; that is, the picture first taken is lifted from the support on tracing paper, put in the right position on terra-cotta, and pressed down while wet with blotting-paper, left to dry, and is then so far ready.
Respecting the production of pictures by means of emulsion, ground opal being the best, the system I employ is as follows: After well cleaning the glass, coat it with emulsion (which had better not be too thick). When dry it is exposed and developed with the usual oxalate developer, to which a little bromide of potassium has been added. The remainder of the operations is as usual. Those varnished with dead varnish can be tinted and worked up with colored crayons or black lead pencil and make very pleasing pictures. It is needless to add that they are also to be finished in water-colors if thought preferable.--G. W. Martyn, in Br. Jour. Photo.
The process of A.C.A. Thiebaut is as follows: the paper has the following advantages:
First. The sensitive coating is regular, and its thickness is uniform throughout the entire surface of each sheet.
Second. It can be exposed for a luminous impression in any kind of slide as usually constructed.
Third. It can be developed and fixed as easily as a negative on glass.
Fourth. The negative obtained dries quite flat on blotting paper.
Fifth. The film which constitutes the negative can be detached or peeled from its support or backing easily and readily by the hand, without the assistance of any dissolving or other agent. Thus this invention does away with all sensitive preparations on glass, which latter is both a brittle and relatively heavy material, thus diminishing the bulk and weight of amateur and scientific photographers' luggage when traveling; it produces photographic negatives as fine and as transparent as those on glass, in so much that the film does not contain any grain; and, lastly, it admits of printing from either face of the film, as regards the production of positives on paper or other material, as well as plates for phototypy and photo-engraving, which latter processes require a negative to be reversed.
For the manufacture of my sensitized film paper:
First. A gelatinized sheet of paper is properly damped with cold water, and when evenly saturated it is placed on a glass, to which it is attached by means of bands of paper pasted partially on the glass, and partially on the edges of the said sheet; in this state it is allowed to dry, whereby it is stretched quite flat.
Secondly. I coat the dry sheet with a solution of ordinary collodion, containing from one to two per cent. cubic measure of azotic cotton (1½ per cent. gives very good results) and from 1½ to 2½ per cent. of castor oil (2 per cent. gives very good results); this coating is allowed to dry; and,
Thirdly. The glass, with the prepared paper upward, is leveled, and then it is coated, in a room from which all rays but red rays of light are excluded, with a tepid emulsion of bromide of silver to the extent of about one millimeter thick, and after leaving it in this position until the gelatine has set (say) about five minutes, with the film paper still attached, it is placed upright in a drying-room, where it should remain about twelve hours exposed to a temperature of from 62 to 66 degrees Fahrenheit; and,
Fourthly. The film paper is detached from the glass ready for exposure, development, and fixing in the usual manner. For the purpose of developing, oxalate of iron or pyrogallic acid answers equally well; for the purpose of fixing, I have found that a mixture by weight, water, 1,000, hyposulphite of soda 150, and powdered alum 60, produces excellent results, after being allowed to dry.
Fifthly. The film is peeled off the paper by hand, and can be immediately used for producing negatives recto or verso as above mentioned.
I claim as my invention:
First. The preparation or formation of gelatino-bromide film paper for photographic negatives, in the manner and for the purposes above described; and,
Secondly. The use for this purpose of castor oil, or any other analogous oil, more especially with the view of peeling off the film from the paper backing as above described.
A substance very much used by photographers of late years--in fact, so much used that no well-appointed laboratory could be considered complete without it--is the substance known is common alum, or potash alum, being a double sulphate of alumina and potash; but it is interesting to note that much of the commercial alum met with at the present time is ammonia alum, or the double sulphate of alum and ammonia. It is quite a matter of indifference to the photographer whether he uses potash alum or ammonia alum.
Besides its great value to the autotype, Woodburytype, and mechanical printers as an agent for hardening the gelatine films, it has been recommended for all sorts of ailments photographic. The silver printer adds a small portion to his sensitizing bath to keep it in working order, and to prevent blistering of the albumen; then, again, silver prints are soaked in a dilute solution of alum, having for its object the thorough elimination of the last traces of the fixing salt. A very good proportion to use for this latter purpose is four fluid ounces of a saturated solution, diluted with one gallon of water, the prints being well agitated during an immersion of ten minutes.
Of all the uses to which alum is put, perhaps not in any single instance can so much satisfaction be derived as when it is used to arrest frilling of gelatine plates. This it has the power to do instantaneously, and many of the most careful workers, both amateur and professional, or at least those who do net care to run any unnecessary risks with negatives which have cost them a good deal of anxiety and trouble to secure, but prefer to make assurance doubly sure--such individuals may be numbered by the hundred--make it a point in every-day practice to immerse all their plates in a solution of alum, either before fixing, or immediately afterward. In fact, some operators have two alum baths in use, one a normal bath, as above mentioned, for immersing the plates in when of the ordinary printing intensity; and the other a saturated solution strongly acidified by means of a vegetable acid (such as citric) or a mineral acid (such as sulphuric), for use when there is too much printing density, since it has been found in practice that an acid solution of alum in contact with sodium thio-sulphate on the gelatine image (after fixing, but before washing) not only removes the color or stain caused by the alkaline or pyrogallol, but perceptibly reduces the strength of the image. Moreover, the color does not again reappear after washing, as it does sometimes when the fixing salt has been partially washed away. In cases where there is great tendency to frill--such, for instance, as when a soft sample of gelatine has been employed, or old decomposed emulsion worked in with the fresh emulsion--it will in such cases be safer to put the plates in the normal-bath for a few minutes previous to immersing them in the acid bath.
Potash alum is obtained tolerably pure in commerce in colorless transparent crystalline masses, having an acid, sweetish, astringent taste. It is soluble in 18 parts of water at 60° F., and in its own weight of water at 212° F.; but the excess crystallizes out upon cooling. The solution reddens litmus paper, and, when impure, usually contains traces of oxide of iron. Upon the addition of either caustic soda or potash, a white gelatinous precipitate is formed (hydrate of alumina), which is soluble in excess of the reagent employed. The precipitate thus obtained has much of the character of the opalescent film sometimes observed on gelatine plates, when dry, which have been soaked in alum, and not well washed afterward.
Alkaline carbonates--such as washing soda, for instance--precipitate hydrate of alumina, which does not dissolve in an excess of the reagents, and carbon dioxide is evolved.
Ammonia hydrate produces a precipitate in a much finer state of divison, which does not dissolve in excess when examined in a test-tube, it somewhat resembles thin starch paste.
The presence of traces of iron may be known by adding a few drops of hydrochloric acid to a small quantity of a saturated solution of alum in a test-tube, to which add strong liquid ammonia; should any iron be present, the mixture will have a reddish-brown tinge when examined over a sheet of white paper. Other alums exist, such as the double sulphate of alumina and sodium, and sodium or aluminum and ammonium; but hitherto their uses have been confined to the experimental portion of the community rather than the practical.--Photo. News.
As is well known, in the process of bleaching and dyeing, cotton cloths become considerably contracted in the width, in consequence of carrying on the operations when the cloth is in the form of a rope. The effect is that, together with the tension, although slight, and the drying, the weft partly shrinks and partly curls up, the latter, however, being scarcely observable to the naked eye. It may almost be said that as regards the width the shrinkage is due to a number of minute crumples because the cloth is easily streatched again by the fingers almost to its gray width. The main use of a stretching machine, therefore, is not so much to make the cloth more than it is as to bring it again to its normal or woven width after operations that tend to shrinkage have been performed upon it. The stretching operation, therefore, is especially useful to calico printers, as it enables them to obtain when desired a white margin of even width, the irregularities due to bleaching being corrected before printing.
IMPROVED CLOTH STRETCHING MACHINE.
The machine now illustrated is one we have recently seen in operation in a Salford finishing works. It is an improved form of another stretching machine which had been turned out in considerable numbers by Mr. Archibald Edmeston, engineer, of Salford, who makes a specialty of calico printers' and finishers' machinery. The improvements consist mainly of a simplification of the working parts and thoroughly substantial construction of the machine. The principle adopted is a well-known one. The selvages of the cloth, or more strictly the two edges of the cloth, of a width of about two inches, are caused to pass over and at the same time are held by the rims of two diverging pulleys. The rims are further apart where the cloth leaves them than where they seize it, hence the stretching is gradually, certainly, and uniformly performed. The cloth is gripped by the pressure of an endless belt acting against the lower half of each pulley, the edges being held between them. In the engraving these stretching pulleys are indicated by the letters AA; the endless leather band passes over the pulleys, CC, of which there are a set of four provided for each stretching pulley. The lower pair of pulleys in each case may be tightened up by a screw for the purpose of imparting the requisite tension to the bands. The stretching pulleys are mounted upon and driven by the same shaft, an ingenious but simple swiveling joint in their bosses enabling them to be set at any angle to the shaft and yet to revolve and be driven by it without throwing any undue strain upon the working parts. The piece, wound upon the ordinary batch shell, is placed upon the running-off center, D; it is led off over the rails, EE, and then downward to the nip of the bands and pulleys, AA. As explained, the selvages are here gripped between the bands and stretching pulleys, the rims of which are wider apart at the back than the front, and thus, in being conveyed underneath, the piece is suitably stretched. Leaving the grip at the back it passes over leading-off rollers, FF, and the scrimp or opening rail, G, and thence downward to the winding-on center, which cannot be seen. The winding-on center is driven by friction. As the batch fills it and tends to wind faster than the machine delivers the cloth, the driving slips. In addition to a capability of being set at an angle to the shaft, the stretching pulleys, AA, may be slided upon, so as to separate or bring them closer together, to allow for the treatment of different widths of cloths. This adjustment is provided for by mounting the stretching pulleys, AA, and the band pulleys, CC, etc., on frames, BB, the ends of which rest, as shown, upon rails, at the back and front of the machine. The adjustment either for width of piece or for the angularity (extent of stretching) is easily made by the hand-wheel, L. By the bevel wheels shown, two cross screws having nuts connected to the ends of frames, BB, are actuated in such a way that as desired the space between the back and front of the pulleys may be closed in or opened out, or the two wheels, maintaining the same angularity, may be separated or closed in, either adjustment being expeditiously made. The wheels, HHH, are called center stretching wheels, the use of which is sometimes advantageous. They act in conjunction with a set of stretching pulleys, of which one, K, may be seen in illustration. By a proper adjustment at the latter the piece is bent into a wavy form, where it passes between the whole of them, the effect of the corrugation being to loosen the center threads and to allow the piece to be more equally stretched with those near the selvages and more easily. This part of the machine may be used or not as required. The production, we observe, was about 120 yards per minute. The machine is solidly built and well fitted together, as was obvious to us from an inspection of some in course of construction at the maker's works. It is also claimed to be of considerable advantage to bleachers and finishers of white goods, on account of the uniformity of the stretching causing but small disturbance to the stiffening.--Textile Manufacturer.
All known methods for chemically purifying woolen stuffs from vegetable fibers depend on the action of acids or substances of acid reaction. The excessive temperature, hitherto unavoidable in the operation, acts injuriously on the woolen fibers, especially during the formation of hydrochloric acid, with which process especially the development of an injuriously high temperature has been hitherto unavoidable. The best method of absorbing the heat developed is in the evaporation of the moisture naturally present in the wool. The patentees find agitation of the fabric and the use of an exhauster during the process of material assistance. The operation maybe successfully performed in two ways--either by acting on the fabric at the ordinary pressure with constant agitation, or by saturation without agitation in a vacuum. For the first method the patentees employ a wooden cylinder with an aperture at one end for inserting and removing the cloth, and having apertures all round to allow free access of air. This cylinder rests on a hollow axle, closed at one end and perforated with holes, through which the acid gas is passed. By the rotation of the cylinder the gas is drawn through the material and the latter exposed to the atmosphere, whereby it gives up a quantity of aqueous vapor. An average temperature of 30° Cent. is best suited to the operation, and it can be regulated according to the supply of gas by opening or shutting a three-way cock between the gas generator and the revolving cylinder. This process is assisted by the use of an exhauster of the usual construction. When fully saturated, the fabric is allowed to remain until the vegetable fibers are sufficiently friable. The treatment in vacuo is as follows:
The hydrochloric acid gas passes into a vessel of suitable material provided with a perforated false bottom. From under this false bottom a pipe connects with a second similar vessel connected itself with a vacuum pump having a let-off pipe. As soon as the maximum vacuum is attained, the gas is turned on through a three-way cock at a pressure of 40 mm. mercury. The gas fills the first vessel and saturates the cloth. The warmth set free (about 500 calories per kilo, gas) is taken up by the combined water in the wool, as, owing to the low pressure, a quantity of vapor is formed sufficient to take up the heat. This vapor streams through the second vessel at a temperature of 35° Cent., penetrates the material, and passes out through the pump. After saturating the contents of the first vessel the gas passes into the second. AS soon as this is one-quarter or one-third saturated the first vessel is taken out and replaced by a third, which receives the overplus from No. 2 in like manner, and so on. This plan of working prevents gas passing through and damaging the pump. Instead of working under reduced pressure, the desired low temperature can be maintained by passing alternately with the gas currents of air which absorb heat in evaporating the moisture of the material. The cloth, after saturation by these processes, is left from six to twelve hours in the vessels, after which it is freely exposed to the air until the vegetable particles are friable. As soon as this occurs, the fabrics are washed. It is advantageous to add to the wash water powdered carbonate of baryta, strontia, magnesia, or preferably lime, and subsequently to rinse in pure water. Phosphate of lime containing carbonate may also be employed for neutralizing the acid, and the residue recovered and separated from the organic residues mixed with it.--"H. J.," Journal of the Society of Chemical Industry.
It is a recognized fact that chemical bodies in a nascent state are characterized by peculiarly energetic affinities, and the results of numerous experiments permit us to affirm that animal and vegetable fibers are rapidly bleached when they are placed in contact with oxides and chlorides which, when submitted to electrolysis, permit oxygen and chlorine to disengage themselves in the nascent state.
The coloring matter that impregnates the majority of vegetable textile substances, such as cotton, flax, and hemp, to cite only those most generally known, is in fact completely destroyed only by the combined action of oxygen and chlorine, which always act in the same manner, whether the fibers be in a raw or woven state.
In the application of electrolysis to the bleaching of textile materials, it is only necessary to have the electrodes of any sufficiently powerful generator of electricity end in a vessel containing in aqueous solution such decolorizing agents as the hypochlorites in general, and chlorides, bromides, and iodides that are capable of disengaging chlorine, and iodine or an iodide in a nascent state. These gases perform the role of oxidizing or decolorizing agents.
The fibers that are immersed in the solution during the passage of the electric current must necessarily remain therein for a greater or less length of time, according to the nature of the material to be bleached, and must, after this first operation, be washed, rinsed, and dried.
The use of an electric current for decomposing the metallic chlorides and disengaging their elements is not new, and there have been specially utilized for this purpose, up to the present time, the alkaline hypochlorites that are obtained by well known processes.
In the latter case the metal is brought to the state of oxide in presence of the water that is necessary for the reaction. But the results obtained in practicing this method are deceiving, as far as bleaching is concerned, and it is evidently more rational and economical to endeavor to compound the hypochlorite directly by borrowing all its elements from the metallic chloride itself, and from the water by means of which such transformation is to be effected. This is a reversal of the problem, and, à propos thereof, we would call the attention of the reader to an apparatus invented by Messrs. Naudin & Schneider for effecting such synthesis in a simple and practical manner.
If a solution of chloride of sodium or kitchen salt, NaCl, be submitted to electrolysis in a hermetically closed vessel containing the material to be bleached, a formation of hypochlorite of soda is produced in the following way:
2NaCl + 2 H2O = NaCl + NaO, ClO + 4H.
In operating in this manner we shall have the advantage that results from the nascent body through the electrical double decomposition of the chloride of sodium and water, which puts the chlorine, the metal, the hydrogen, and the oxygen simultaneously in presence. The chlorine and oxygen will combine their action to decolorize the textile material.
While starting from this idea, it will nevertheless be preferable to adopt Naudin & Schneider's arrangement.
The apparatus consists of a hermetically closed electrolyzer, A, into the lower part of which enters the electrodes, E and F, of any electrical machine whatever. The receptacle, A, is provided with a safety-tube, T, that issues from its upper part and communicates with a reservoir, B. A second tube, D, forms a communication between the electrolyzer and the vessel, C. The liquid contained in this latter is sucked up by a pump, P, and forced to the lower part of the vessel, A, by means of the tubes, G and H.
The apparatus operates as follows:
The closed vessel, C, in which the material to be bleached is put, is filled, as is also the electrolyzer, with a solution of chloride of sodium. This solution is then submitted to the action of an electric current, when, as a consequence of the chemical decomposition of the chloride and the water, the elements in a nascent state form hypochlorite of soda. When the partial or total conversion of the liquid has been effected (this being ascertained by chlorometric tests), the pump, P, is set rapidly in operation, and, as a consequence, draws up the chloride of sodium from the bottom of the vessel, C, to the lower part of the electrolyzer, A. The hypochlorite that has formed passes through the tube, D (as a natural consequence of the elevation of the level of the liquid in A brought about by the entrance of a new supply of chloride), and distributes itself throughout the vessel, C, where it acts upon the textile material.
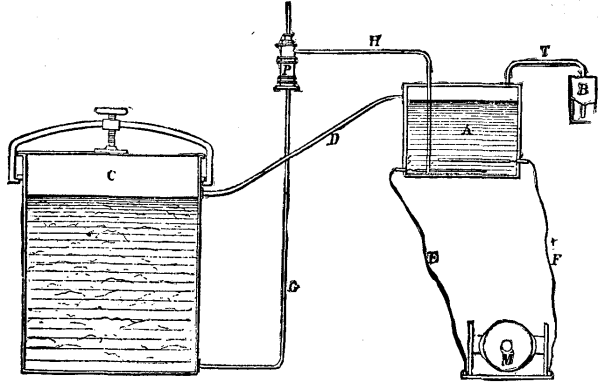
APPARATUS FOR BLEACHING TEXTILE FIBERS
BY ELECTRICITY.
The safety-tube, T, which is attached to the electrolyzer, permits of the escape of the hydrogen which is produced during the chemical reaction, and fixes, through an alkaline solution contained in the reservoir, B, the chloride whose escape might discommode the operator.
As may be conceived, the slow transfer of the saline solution from the receptacle, C, to the electrolyzer, and its rapid conversion into decolorizing chloride, as well as its prompt application upon the materials to be bleached, presents important advantages.
While, in the present state of the industries that make use of bleaching chlorides, the chloride of sodium is converted into hydrochloric acid, which, in order to disengage chlorine, must in its turn react upon binoxide of manganese, we shall be able, with this new method, to utilize the chloride of sodium, which is derived from ordinary salt works, and extract from it the constituent elements of the hypochlorite by a simple displacement of molecules produced under the influence of an electric current.
Another and very serious advantage of electric bleaching is that of having constantly at hand a fresh solution of hypochlorite possessing a uniform decolorizing power, which may be regulated by the always known intensity of the current.
We must remark that the hypochlorites require a certain length of time to permit the chlorine to become disengaged, and that, besides, all chlorides, bromides, and iodides that are isomorphous are capable of undergoing an analogous chemical transformation and of being employed for the same purpose. This is especially the case with the chlorides of potassium or barium, the bromides of strontium or calcium, and the iodides of aluminum or magnesium. On another hand, as sea water contains different chlorides, it results that it might serve directly as a raw material for bleaching textile fibers. Then, when the solution of chloride of sodium has been deprived of its chlorine by electrolysis, there remains a solution of caustic soda which may be utilized for scouring fibers.--H. Danzer, in Le Génie Civil.
Messrs. J. & H. McLaren, of the Midland Engine Works, Hunslet, Leeds, England, for several years past have devoted considerable attention to the question of mounting traction engines on springs. The outcome of this is the engine in question, the front end of which is carried by a pair of Timmis spiral springs, resting on the center pin of the front axle, which is on Messrs. McLaren's principle, which enables it to accommodate itself to the inequalities of the road without throwing any undue strain on the front carriage. The chief difficulty hitherto has been to mount the hind end on springs without interfering with the spur gearing, which must be kept perfectly rigid to prevent breakage of the cogs. This is entirely provided for by the new arrangement, whereby all the spring is allowed for in the spokes of the wheel itself, which will be clearly seen on reference to the illustrations, in which Fig. 1 is a perspective view of the engine, while Fig. 2 shows a detail view of the wheel. The rim of the wheel is built up in the ordinary way of strong T-iron rings, with steel crossplates riveted on. The nave of the wheel has wrought-iron ribs to which the spokes are bolted. These spokes are made of the best spring steel, specially manufactured and rolled for the purpose, 9 inches wide and ½ inch thick. They are bent in a pear shape, with the narrow ends fastened to the nave, and the crown resting upon the rim of the wheel, where they are divided, and held in their places by means of clip fastened with bolts. When the weight of the engine comes on these spokes, those nearest the ground are compressed and those, at the top are elongated a little. In order to avoid any of the driving strain passing through the springs, a strong arm is fixed on the differential wheel and attached to the rim as shown in Fig. 2, so that the springs have really no work to do beyond carrying the weight of the engine. Messrs. McLaren naturally felt a certain amount of diffidence in placing their invention before the public until they had thoroughly tested it in practical work. This, we are informed, they have done, with the most satisfactory results, during the last five or six months; and they have a set of springs which ran during that time between 2,000 and 3,000 miles, besides which there are several of these spring engines in daily use.--Iron.
FIG 1. IMPROVED SPRING TRACTION ENGINE.

FIG. 2
DIAMETER | AREA
|
B.W.G Inch. Milli- | Circu- Square Square
No. metres | lar inches. Milli-
| Mils. metres.
|
0000 .454 11.5313 | 206116 .161883 10.4435
000 .425 10.795 | 180625 .141862 9.152
00 .38 9.6518 | 144400 .113411 7.3165
0 .34 8.6358 | 115600 .0907922 5.8573
1 .3 7.620 | 90000 .070686 4.5602
2 .284 7.2134 | 80656 .0633472 4.0867
3 .259 6.5784 | 67081 .0526854 3.3989
4 .238 6.0451 | 56644 .0444881 2.8701
5 .22 5.5879 | 48400 .0380133 2.4523
6 .203 5.1561 | 41209 .0323655 2.088
7 .18 4.5719 | 32400 .0254469 1.6417
8 .165 4.1909 | 27225 .0213825 1.3794
9 .148 3.7591 | 21904 .0172034 1.1098
10 .134 3.4035 | 17956 .0141026 .9096
11 .12 3.0479 | 14400 .0113097 .7296
12 .109 2.7701 | 11881 .00933133 .60199
13 .095 2.4129 | 9025 .0070882 .4573
14 .083 2.1082 | 6889 .00541062 .34906
15 .072 1.8288 | 5184 .00407151 .2486
16 .065 1.6510 | 4225 .00331831 .21407
17 .058 1.4732 | 3364 .0026421 .17045
18 .049 1.2446 | 2401 .00188574 .12165
19 .042 1.0668 | 1764 .00138544 .0894
20 .035 0.8890 | 1225 .000962115 .06207
21 .032 0.8128 | 1024 .00080425 .05188
22 .028 0.7112 | 784 .000615753 .03972
23 .025 0.635 | 625 .00049087 .03167
24 .022 0.5588 | 484 .000380133 .02452
25 .02 0.508 | 400 .00031416 .02027
26 .018 0.4571 | 324 .000254469 .01642
27 .016 0.4064 | 256 .000201062 .01297
28 .014 0.3556 | 196 .000153938 .00993
29 .013 0.3302 | 169 .000132732 .00856
30 .012 0.3048 | 144 .000113097 .007296
LENGTH AND WEIGHT
B.W.G Pounds Pounds Pounds Pounds Feet Yards 1.000 feet Miles
No. per per per 1.000 per per lb. per lb. per lb. per lb.
foot. Yard ft. mile.
0000 .623924 1.871772 623.924 3294.32 1.60276 .534253 .00160276 .00303553
000 .54676 1.64028 546.76 2886.89 1.82895 .60965 .00182895 .0034639
00 .437105 1.311315 437.105 2307.92 2.28777 .76259 .00228777 .004333
0 .349928 1.049784 349.928 1847.62 2.85773 .9525766 .00285773 .0054124
1 .272435 .817305 272.435 1438.43 3.6706 1.22353 .0036706 .0069519
2 .244151 .732453 244.151 1289.11 4.0958 1.365266 .0040958 .0077573
3 .203058 .609174 203.058 1072.15 4.9247 1.641566 .0049247 .009327
4 .171463 .514395 171.465 905.333 5.8321 1.944033 .0058321 .0110457
5 .14651 .43953 146.510 773.56 6.8255 2.275166 .0068255 .012927
6 .124742 .374226 124.742 658.638 8.0165 2.672166 .0080165 .015183
7 .098076 .294228 98.076 517.844 10.1962 3.39873 .0101962 .019311
8 .082411 .247233 82.411 435.135 12.1345 4.04483 .0121345 .022981
9 .066305 .198915 66.305 350.089 15.0818 5.027266 .0150818 .028564
10 .054354 .163062 54.354 286.99 18.398 6.13266 .018398 .034845
11 .04359 .13077 43.590 230.152 22.9413 7.6471 .0229413 .04345
12 .035964 .107892 35.964 189.893 27.805 9.2683 .027805 .05266
13 .027319 .081957 27.319 144.245 36.6046 12.20153 .0366046 .069326
14 .020853 .062559 20.853 110.1088 47.954 15.98466 .047954 .09082
15 .015692 .047076 15.692 82.855 63.7267 21.24223 .0637261 .12069
16 .012789 .038367 12.789 67.5276 78.1902 26.0634 .0781902 .14809
17 .0101828 .0305484 10.1828 53.7665 98.202 32.734 .098203 .18589
18 .00726795 .02180388 7.26796 38.3748 137.590 45.8633 .137590 .260587
19 .00533972 .01601916 5.33972 28.1937 187.276 62.4253 .187276 .35469
20 .00370815 .01112445 3.70815 19.579 269.676 89.892 .2696676 .51075
21 .00309972 .00929910 3.09972 16.3665 322.610 107.5366 .322610 .61100
22 .00237312 .00711936 2.37312 12.5301 421.384 140.4613 .421334 .798078
23 .0018910 .0056757 1.8919 9.9892 528.570 176.190 .528570 .100108
24 .0014650 .0043950 1.4650 7.7357 682.55 227.5166 .68255 .129271
25 .00121082 .00363246 1.21082 6.39315 825.880 275.2943 .825883 .156417
26 .00098077 .00294231 .98077 5.17844 1019.61 339.870 1.01961 .193108
27 .00077492 .00232476 .77492 4.0916 1290.44 430.1466 1.29044 .24440
28 .0005933 .0017799 .5933 3.13264 1685.48 561.8266 1.68548 .31922
29 .000511571 .001534713 .511571 2.7011 1954.76 651.5866 1.95476 .370220
30 .0004359 .0013077 .4359 2.30152 2294.13 764.710 2.29413 .434496
LENGTH AND RESISTANCE
B.W.G Feet Yards 1.000 feet Miles Ohms Ohms Ohms Ohms
No. per Ohm. per Ohm. per Ohm. per Ohm. per foot. per yard. per 1.000 per mile.
foot.
0000 19966.5 6655.5 19.9665 3.7815 .000050684 .00156252 .050084 .264443
000 17497.15 5832.3833 17.49715 3.31385 .0000571522 .0001714566 .0571522 .301763
00 13988.64 4662.68 13.98804 2.64925 .000071489 .000214467 .071489 .377465
0 11198.17 3732.7333 11.19817 2.12086 .0000893002 .0002679006 .0893002 .471505
1 8718.30 2906.10 8.71830 1.6512 .00011470 .0003441 .114701 .60562
2 7813.50 2604.50 7.81350 1.47973 .00012799 .00038397 .12799 .67580
3 6498.14 2166.0466 6.49814 1.23071 .00015389 .00046167 .15389 .81254
4 5487.107 1829.0357 5.487107 1.03923 .000182245 .000546735 .182245 .962256
5 4688.51 1562.8366 4.68851 .887975 .000213287 .000639861 .213287 1.12616
6 3991.91 1330.6366 3.99191 .756045 .000250506 .000751518 .250506 1.32267
7 3138.59 1046.1966 3.13859 .59443 .000318614 .000955842 .318614 1.68228
8 2637.29 879.0966 2.63729 .499486 .000379177 .001137531 .379177 2.00206
9 2121.84 707.280 2.12184 .401864 .000471289 .001413867 .471289 2.488405
10 1739.40 579.80 1.73940 .329432 .000574911 .001724733 .574911 3.03553
11 1394.93 464.9766 1.39493 .264191 .000716882 .002150646 .716882 3.78514
12 1150.91 383.6366 1.15091 .217976 .000868875 .002606625 .868875 4.58766
13 874.252 291.4173 .874252 .165578 .00114383 .00343149 1.14383 6.03945
14 667.338 222.446 .667338 .12639 .00149849 .00449547 1.49849 7.91203
15 502.175 167.39166 .502175 .095109 .00199134 .00597402 1.99134 10.5142
16 409.276 136.42533 .409276 .077514 .00244334 .00733002 2.44334 12.9008
17 325.871 108.62366 .325871 .061718 .0030687 .0092061 3.0687 16.20274
18 232.585 77.52833 .232585 .04405 .0042995 .0128985 4.2995 22.7014
19 170.879 56.95966 .170879 .032363 .0058521 .0175563 5.8521 30.8991
20 149.3915 49.797166 .1493915 .022475 .00842703 .02528109 8.42703 44.4947
21 99.195 33.065 .099195 .018787 .01008110 .03024348 10.08116 53.2285
22 75.9461 25.315366 .0759461 .014384 .0131672 .0395016 13.1672 69.5230
23 60.54377 20.181256 .06054377 .011467 .0165170 .0495510 16.5170 87.2096
24 46.8851 15.628356 .0468851 .0088798 .02132874 .06398622 21.32874 112.616
25 38.748 12.916 .038748 .0073386 .025808 .077424 25.808 136.265
26 31.3859 10.461966 .0313859 .0059443 .03186144 .09558432 31.86144 168.229
27 24.79873 8.266243 .02479873 .0046967 .0403246 .1209738 40.3246 212.914
28 18.98653 6.328843 .01898653 .0035959 .05266892 .15800676 52.66892 278.092
29 16.3710 5.4570 .0163710 .0031006 .0610834 .1832502 61.0834 322.521
30 13.9493 4.649766 .0139493 .0026419 .07168825 .21506475 71.68825 378.514
RESISTANCE & WEIGHT
B.W.G Ohms Lbs. No. per lb. per Ohm.
0000 .000080272 12457.5 000 .000104529 9566.7 00 .000163553 6114.24 0 .000255196 3918.58 1 .00042102 2375.18 2 .00052422 1907.59 3 .00075786 1319.50 4 .0010629 940.844 5 .0014558 686.911 6 .0020082 497.96 7 .00324863 307.822 8 .00460101 217.343 9 .00710791 140.689 10 .0105772 94.543 11 .0164462 60.842 12 .0241593 41.392 13 .0418692 23.8839 14 .0718583 13.9163 15 .126788 7.8872 16 .191045 5.2344 17 .301355 3.31835 18 .59157 1.6904 19 1.09596 .912445 20 2.27254 .44003 21 3.25229 .30748 22 5.54843 .18023 23 8.73035 .11454 24 14.5579 .068691 25 21.3142 .046917 26 32.4863 .030782 27 52.0367 .019217 28 88.7724 .011265 29 119.404 .008375 30 164.4762 .0060804
PURE COPPER weighs 555 lbs. per cubic foot. The Resistance of 1 mil. foot at 60° Fahr. is, according to Dr. Matthiessen, 10.32311 ohms. Upon these data the above Table has been calculated.
The Resistance of Copper varies with the temperature about 0.38 per cent. per degree Centigrade, or 0.21 per cent. per degree Fahrenheit.
STRANDED WIRES.--With a conductor of a definite lenght, made of Stranded Wires, the total weight is greater, and the Resistance less than is a similar length of Conductor with Wires not Stranded.
To convert--Inches to Millimetres multiply by 25.3994
Feet to Metres " .3048
Yards to Metres " .9144
Miles to Kilometres " .6214
Pounds to Kilogrammes " .45359
PEPARED BY WALTER T. GLOVER & CO., ELECTRICAL WIRE AND CABLE MAKERS, 25, BOOTH STREET MANCHESTER.
The gang mill is regarded as possessing material advantages in the rapid and economical manufacture of lumber. Among the recent improvements tending to perfect such mills, those which are shown in the iron frame stock gang, manufactured by Wickes Bros., East Saginaw, Mich., are eminently valuable. Our large engraving represents one of these mills, constructed to be driven by belt, friction, or direct engine, as may be desired. The important requisite in this class of mills is such design and proportion of parts as will insure durability and continued movement at the highest speed, safely increasing the quantity and improving the quality of work done at a lesser feed, and admitting the use of thinner saws than is practical in the slower moving sash. These are among the advantages gained in the iron frame machine, overcoming the necessity of an expensive mill frame, saving time and expense in setting up, and avoiding the liability of decay or change of position.
IMPROVED IRON FRAME GANG SAW MILL.
Many improvements have been made in the mechanism of oscillation, and from these the builders of this mill have adopted what is known as the Wilkin movement, which oscillates the top and bottom slides. The top slides are pivoted at the top end, and the bottom ones from the bottom end, both being operated by one rock shaft from the center. This movement when properly adjusted gives an easy clearance and the easiest cut yet obtained. It adds no extra weight to the sash, and avoids the cumbrous rock shaft and its attendant joints, usually weighing from three hundred to five hundred pounds, which have been found so objectionable in many other movements. The feed is continuous, and is made variable from ¼ to 1¼ inch to each stroke, controllable by the sawyer. Power is applied to the press rolls in the double screw form with pivot point, also operated by the same hand. A special feature of this machine is the spreading of the lower frame so that its base rests upon an independent portion of the foundation from the main pillow block or crank shaft. The solidity of the whole structure is thus increased, both by the increased width at the base and the prevention of connecting vibrations, which necessarily communicate when resting upon the same part, as in other forms of such machines heretofore in use.
The mill shown in the perspective view is one of twenty-six saws 4½ feet long, sash 38 inches wide in the clear, and stroke 20 inches, capable of making 230 strokes per minute. The crank shaft is nine inches in diameter, of the best forged iron. The main pillow block has a base 6½ feet long by 21 inches bearing, weighing 2,800 pounds. The cap is secured by two forged bolts 3½ inches in diameter, and by this arrangement no unequal strain upon the cap is possible. A disk crank is used with suitable counterbalance, expressly adapted to the weight and speed of sash; a hammered steel wrist pin five inches in diameter, and a forged pitman of the most approved pattern, with best composition boxes. The iron drive pulley is 4 to 4½ feet in diameter and 24 inches face; the fly-wheel six feet in diameter, and weighing 4,700 pounds, turned off at rim. When a wider and heavier sash is required, a proportionate increase is made in all these parts.
In the construction of the sash the stiles are made of steel; the lower girt and upper heads are made in one solid piece, without rivets, giving the greatest strength possible, with the least weight. The outfit also includes eight iron rollers for the floor, 8½ inches in diameter, with iron stands, and geared as live rolls when desired, a full set of Lippencott's steel saw hangings, and gauges for one-inch lumber. The weight of the machine here shown is 18½ tons. They are, however, built in larger or smaller sizes, adapted to any locality, quality or quantity of work desired.
It is said that the St. Gothard Tunnel is diverting the bulk of the Italian trade into the hands of the Belgians, Germans, and Hollanders with startling rapidity. Without breaking bulk, early fruits are taken from all parts of Italy to Ostend, Antwerp, and Rotterdam, whence they are carried by fast steamers to London and other English ports. But, on the other hand, Germany is sending into Italy large quantities of coal, iron, machinery, copper, and other articles of which the latter received nothing before. In two months alone, the Italians imported 1,446 tons of paper.
The system of heat regeneration in the firing of gas retorts, in accordance with the principle which Dr. C.W. Siemens has worked out in such a variety of ways in the industrial arts, has lately been applied with very marked success at the Dalmarnock Station of the Glasgow Corporation Gas Works. Notwithstanding the fact that a period of about twenty years has elapsed since Dr. Siemens successfully adapted his system to the firing of retorts at the Paris Gas Works, it seems to have made but little progress up to the present time; for what reasons it is perhaps difficult to explain. It is certain, however, that so-called regenerator furnaces of various forms have, from time to time, been brought into use at gas works for the purpose in question both on the Continent and in this country; and in recent years the subject has received much attention from gas engineers, the general opinion eventually being that the adoption of such a system of working would be certain to result in so great an amount of economy as to put gas as an illuminating agent on a more secure footing to compete successfully with its modern and somewhat aggressive rival, the electric light. Of course, it is now admitted that the mode of adapting the heat regenerative principle at the Paris Gas Works was attended with a degree of complexity in the structural arrangements that was so great and so expensive as to place it practically beyond the reach of gas companies and gas corporations generally, when the expense as well as the scientific beauty and practical efficiency of the new mode of applying and utilizing heat had to be considered. Fortunately, however, Dr. Siemens was enabled two or three years ago to demonstrate that there was no such thing as "finality" in that department of invention which he had made almost exclusively his own. About the time mentioned he placed his most advanced views on gas producers and on the regeneration and utilization of heat before the world, and within that period a most decided step in advance has been made, the structural arrangements now required for gas producers and regenerator furnaces having been immensely simplified and cheapened, while their practical utility has in no way been interfered with.
Scarcely had Dr. Siemens announced his new form of gas producer and regenerator than communication was opened with him by Mr. W. Foulis, the general manager to the Glasgow Corporation Gas Trust, with the view of entering into arrangements for its adoption on an experimental scale at one of the stations under his charge. Encouraged by the hearty co-operation of the gas committee, two or three of whose members were well known engineers, Mr. Foulis very soon came to an understanding with Dr. Siemens to have the regenerative system put to a thorough test at the Dalmarnock Gas Works, situated in the extreme east end of the city, and the largest establishment of the kind in Scotland, the total number of retorts erected being about 750. The system in its most recent shape was applied to four ovens, each of which had seven retorts, but which number has since been increased to eight, owing to the space occupied by the furnace in the ordinary settings being rendered available for an additional retort in the new or "Siemens" setting. For each oven or chamber of eight retorts there was erected a separate gas-producer, so that even one set of eight retorts might alone be used if thought necessary.
GAS RETORTS WITH REGENERATIVE FURNACES .--GLASGOW CORPORATION GAS WORKS.
In Figs. 1 and 2 of our illustrations, the general arrangement and the relationship of the gas producer, the regenerators, and the retorts to each other are clearly shown. It was a sort of sine qua non of the new method of firing the retorts that the producer should be in as close proximity as possible to the place where the gaseous fuel was to be used, and it was concluded that the most convenient situation would be immediately in front of its own set of eight retorts, and with its top on a level with the working floor of the retort house. To place it in such a position meant a good deal of excavation, which was also required, however, for the regenerator flues. The excavation was carried down to a depth of 10 ft. below the level of the retort house floor, and as a matter of course the operation of underpinning had to be resorted to for the purpose of carrying down the foundations of the division walls, which, together with the main arches and the hydraulic main, were in no way otherwise disturbed. As in most new inventions, a good deal of difficulty was experienced at first in connection with these gas producers and heat regenerator furnaces; but by dint of application and by the adoption of modifications made here and there in the arrangements from time to time, as also by a determination not to be beaten, although often disheartened, Mr. Foulis was ultimately rewarded with complete success. The new system of firing being made so simple that there was scarcely any possibility of failure likely to arise in ordinary practice if it was superintended with but a moderate amount of care.
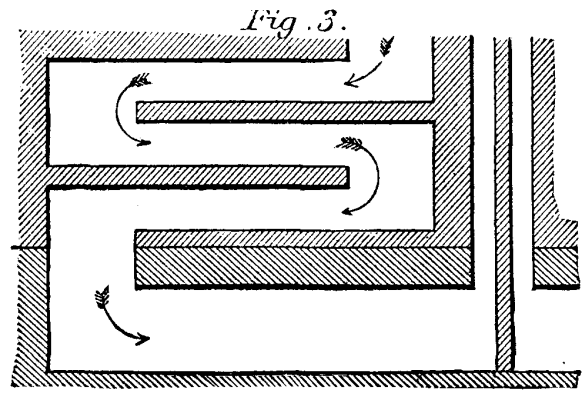
Fig. 3.
The results which were obtained in course of time with four ovens, or a total of 32 retorts, were so exceedingly promising that it was forthwith resolved to extend the new mode of firing to the whole of a double bench of twelve ovens, now containing 96 retorts; and all the improvements which had suggested themselves during the working experiments with the four ovens were adopted from the first in the reconstruction of the remaining eight ovens in the bench. More recently the regenerator system has been applied to other 22 ovens, or 176 additional retorts, being the whole of one of the main divisions of the retort house; and during the very depth of the present winter, when the demand for gas was at its greatest height, all the retorts of the converted or "Siemens" settings, amounting to 272, were in full working activity, in which condition they still remain. It is intended to make another very considerable extension of the heat regenerative system of firing during the ensuing spring and summer. The reconstruction of the present year will extend to the ovens of seven retorts each, giving in this case eighty gas fired retorts; and to twenty ovens of five retorts each, which will become sixteen ovens, each having eight retorts, making 128 retorts in this division, and the total being 208 retorts in place of 170 in the same amount of space. It is confidently anticipated, therefore, that by the month of August of the present year, 480 full sized retorts will be available for working out the new method at the Dalmarnock Gas Works. Furthermore, the confidence which has been inspired in the minds of the members of the Glasgow Corporation Gas Committee and their engineer regarding the actualities and possibilities of the Siemens system of firing gas retorts, in its most improved state, is such that arrangements are being made for starting shortly to apply it throughout at the Dawsholm Station, which is situated in the suburban burgh of Maryhill, and some four or five miles distant from the Dalmarnock Works in a northwestern direction. The station just named, which is also a very large one, will probably require two years for its conversion.
We shall now give some account of the structural arrangements adopted for producing cheap gaseous fuel, and for turning that fuel to the greatest advantage in firing the retorts for the purpose of carbonizing the cannel coal used as the source of the gas.
The gas producer, which is represented in vertical section in Fig. 2, is a cylinder of brickwork inclosed in a casing of malleable iron. It is 7 ft. 6 in. deep, and 3 ft. in diameter, which becomes reduced to 20 in. above, where it is closed by means of a cast-iron lid, which is continuous with the floor of the retort house. There are no firebars at the bottom, so that the fuel rests on a floor of firebrick. At the bottom of the walls of the producer there are several holes about 1 ft. in length by 6 in. in height. By means of these openings any clinker that may form and the ashes of the spent fuel can readily be withdrawn. They also allow of the admission of air to maintain the combustion in the lower portion of the mass of fuel; and at each opening there is a malleable iron tube for delivering a jet of steam direct from a steam boiler. We shall subsequently explain the functions performed by the steam.
The fuel employed is the coke or char resulting from cannel coal when it has yielded up its hydrocarbons and other gases during the process of carbonization in the gas retorts. Being entirely made from Scotch cannel the coke is very poor in quality, as it contains a large percentage of mineral matter or ash relatively to its fixed carbon. The retorts are worked with three-hour charges, but the producer is only charged once in every six hours For each set of eight retorts the charge of raw cannel is about 18 cwt., and it is found in practice that the coke drawn from five of the retorts is quite sufficient to fill up the producer to the top. Formerly a set of seven retorts fired in the ordinary way from a furnace underneath, required from 60 to 75 per cent. of the coke made, but now, with eight retorts in each oven, the quantity has been reduced to about 30 per cent., or less than one-half of what it formerly was. Before the retorts are drawn the lid is removed from the top of the producer, and any fuel still remaining unconsumed is touched up a bit by way of leveling it on the surface, and as soon as it has been filled up to the constricted portion a shovelful of soft luting is spread over the top of the coke, and the lid is laid upon it and driven home, thereby making a perfectly air-tight joint. The contents of the other three retorts, as also the contents of the whole of the retorts at each alternate drawing, are taken to the coke heap in the yard. We have already spoken of a charge of cannel as being about 18 cwt. for each set of eight retorts, but in connection with that matter we should mention that it was formerly about 13 cwt. per oven containing seven retorts, and that there is every prospect of it being increased without increasing the length of time occupied in carbonizing the cannel of each charge.
It may be worth while now to notice briefly what takes place among the mass of coke in the gas producer. The atmospheric air admitted at the several openings previously spoken of ascends through the lower layers of the incandescent coke, the carbon of which burns to carbonic acid gas at the expense of the oxygen of the air. Among the middle and upper layers of the incandescent coke the carbonic acid gas takes up a further quantity of the fixed carbon, and becomes transformed into carbonic oxide gas (CO2+C=2CO), which is an inflammable body, and possesses considerable calorific power. Unless the carbonic acid gas is very completely "baffled" in its ascent through the coke in the producer, a quantity of it passes into the furnace along with the carbonic oxide, the efficiency of which is diminished in proportion as the former increases in quantity. Of course, also, the nitrogen associated with the oxygen in the air admitted to the gas generator passes on with the carbonic oxide gas, this nitrogen acting as a dilutant and being of course absolutely useless as a generator of heat. The steam which we previously spoke of serves two good purposes. In contact with incandescent coke it suffers decomposition, its oxygen uniting with some of the fixed carbon to form carbonic oxide, while the hydrogen which is set free passes onward, and mixes with the other gases to be subsequently consumed with them. The admission of the steam thus causes the absorption of heat in the gas generator where the decomposition takes place, this heat being again evolved on the subsequent combustion of the hydrogen. Then, again, as the steam is delivered in among the coke in a jet, or a series of jets, it has the effect of almost entirely preventing any clinkering or slagging of the earthy and silicious materials, which form such a large portion of the substance of the coke obtained from Scotch cannels, sometimes as much as from 15 to 20 per cent. It is scarcely necessary for the stokers to go down below to the bottom of the producers to remove the ash above once in every six hours. Referring to the composition of the gaseous fuel obtained from cannel coke in one of these gas producers, we give the following typical analysis on the authority of Dr. William Wallace, F.R.S.E., gas examiner, and one of the public analysts for the city of Glasgow:
Per cent.
Hydrogen 8.7
Carbonic oxide 28.1
Carbonic acid 3.5
Oxygen 0.4
Nitrogen 59.3
-----
100.0
By again referring to Fig. 2, it will be observed that an opening is provided for the passage of the gaseous matter as it is formed into the mass of brickwork, the upper half of which is occupied by the retorts of the setting and the lower by the regenerators.
Before following the gas we may first direct attention to the
arrangements for dealing with it, and with the air that has to be
admitted for the combustion of so much of it as is of a combustible
nature. It will be seen by reference to Fig. 1 that the oven proper
is occupied by eight  shaped retorts. These are 9 ft. long (set back to back) by 18 in.
by 13 in., and they are placed on arches which are 8 ft. 6 in.
wide. Underneath the level of the retort oven there are two
regenerators or regenerator chambers, which differ very materially
in form from the regenerators formerly applied by Dr. Siemens to
gas retort ovens, and which are still employed for high temperature
furnaces like those used for steel and glass melting. In the case
of these latter the regenerators are on the alternating
system--that is to say, a mass of brickwork is heated by the waste
heat of the effluent gases, and when that is made sufficiently hot,
the current of waste gases is turned into a second mass of
brickwork, while air is admitted to pass through the brickwork
already heated. The system thus briefly described entails a certain
amount of attention on the part of the workmen in the altering of
the valves or dampers to reverse the currents. The regenerator now
adopted consists of an arrangement of six zigzag flues, three on
each side of the setting. These flues run the whole length of the
setting. As indicated by the arrows pointing downward in Fig. 3,
the waste gases on their way to the chimney stack pass to and fro
through the side flues, thus giving up a large portion of their
contained heat by the process of conduction or contact to the
central flue through which the incoming air passes. The air
necessary for combustion is first admitted into a large chamber in
the center, and then it is divided into two currents, which pass
right and left into the central passages of the two regenerators.
As the air flue is at a very bright heat for a considerable
distance before the air leaves it, the temperature of the air must
be equally great, or nearly so. In its most improved form one of
these heat regenerative furnaces provides an amount of heating
surface extending to 234 square ft., which is exposed to the air on
its way to the combustion chamber.
shaped retorts. These are 9 ft. long (set back to back) by 18 in.
by 13 in., and they are placed on arches which are 8 ft. 6 in.
wide. Underneath the level of the retort oven there are two
regenerators or regenerator chambers, which differ very materially
in form from the regenerators formerly applied by Dr. Siemens to
gas retort ovens, and which are still employed for high temperature
furnaces like those used for steel and glass melting. In the case
of these latter the regenerators are on the alternating
system--that is to say, a mass of brickwork is heated by the waste
heat of the effluent gases, and when that is made sufficiently hot,
the current of waste gases is turned into a second mass of
brickwork, while air is admitted to pass through the brickwork
already heated. The system thus briefly described entails a certain
amount of attention on the part of the workmen in the altering of
the valves or dampers to reverse the currents. The regenerator now
adopted consists of an arrangement of six zigzag flues, three on
each side of the setting. These flues run the whole length of the
setting. As indicated by the arrows pointing downward in Fig. 3,
the waste gases on their way to the chimney stack pass to and fro
through the side flues, thus giving up a large portion of their
contained heat by the process of conduction or contact to the
central flue through which the incoming air passes. The air
necessary for combustion is first admitted into a large chamber in
the center, and then it is divided into two currents, which pass
right and left into the central passages of the two regenerators.
As the air flue is at a very bright heat for a considerable
distance before the air leaves it, the temperature of the air must
be equally great, or nearly so. In its most improved form one of
these heat regenerative furnaces provides an amount of heating
surface extending to 234 square ft., which is exposed to the air on
its way to the combustion chamber.
Passing from the producer through the flue provided for it, the gas enters the retort setting underneath the side retorts, where it meets the air coming from the regenerator. It enters the setting, not by a number of small openings, but by one large opening on each side, and meets the air entering also by a large opening, the effect of which is to avoid the localization of intense heat, as all the retorts of the setting become enveloped in an intensely heating flame, due to the combustion of the carbonic oxide and hydrogen gases.
There are various advantages attending this system of firing gas retorts. First of all, there is already a saving of fuel to the extent of one-half, and not unlikely there will soon be a further very decided increase in the saving of fuel to record, inasmuch as it has been experimentally determined within the past two or three weeks that, by increasing its diameter to 3 ft. 4 in., one producer can be made to provide a sufficient amount of gaseous fuel to fire two sets of eight retorts. By the arrangement just hinted at the relative amount of fuel used will be still further reduced. Then, again, an additional retort can well be placed in each oven, as it occupies the position of the fire in ordinary settings. In the third place, by the greater heat which is obtained, the charges can be more rapidly distilled; or heavier charges can be carbonized in a given space of time. When all the gains are put together, the amount of coal carbonized is increased by about 40 per cent. over any specified time. Of course, in the new or regenerator settings there is much greater regularity of heat; and as the gaseous fuel is perfectly free from all solid matter, and burns without any trace of smoke, there is a total absence of deposit on the outside of the retorts. From these two circumstances combined it is but natural to expect that there should be greater durability of the retorts--which is really the case. Another advantage is that, as the fuel used in the furnaces is wholly gaseous, choking of the flues cannot by any possibility arise. It is the confident opinion of Mr. Foulis that the system in question can be applied with advantage to all sizes of gas works, and that it is certainly well adapted for all works where the summer consumption of gas is sufficiently large to give employment to eight retorts.
As this is the first instance of the new form of gas producer and regenerator having been adopted in any gas works, a very great amount of scientific and practical interest attaches to it. Many persons have visited the Dalmarnock Gas Works during their reconstruction, in order to see the system in operation, and doubtless many more will go and do likewise when they learn of the numerous advantages which it possesses, and which are likely to increase rather than diminish.--Engineering.
During the past few weeks, a highly interesting experiment--and one, moreover, destined to materially influence the development of the uses of gas in a fresh field--has been in progress, under the guidance of Mr. Booer, at a baker's shop in the Blackfriars Road, London. The experiment in question is nothing less than the application of gas for heating bakers' ovens, in a manner not hitherto attempted, and such as to bring the system within the means of the poorest tradesman in all but the smallest towns. It will be remembered that the success of the gas-heated muffles for burning tiles and glass led to the attempted construction of a model baker's oven, heated by the same fuel, which was shown in action at the Smoke Abatement Exhibition at South Kensington in the winter of 1881-82. This model attained considerable success; but its design demanded either a new structure in every case, or considerable alteration of any existing oven. In the proposed system, moreover, the oven was heated wholly from without--a condition supposed to be necessary to meet the objections of the bakers. It is evident, however, that there must be considerable waste of gas in heating a mass of tiles and brickwork, such as go to the construction of a common baker's oven, from the outside; and the objection to handicapping such a costly fuel as gas in this manner becomes more apparent when it is remembered that in the usual way the oven is always heated by an internal coal fire. When it is further considered that the coal commonly used by bakers is of the most ordinary quality, full of dirt that would condemn it in the estimation of a gas manager, the sentimental objection to allowing a purified gas flame to burn in a place which this rubbish is permitted to fill with foul smoke becomes supremely ridiculous. Consequently, when Mr. Booer, whose work in connection with the gas muffle is well known in England and America, seriously addressed himself to construct, upon altogether new lines, a cheap and practical baker's oven, he wisely put the gas inside.
There are many other conditions which Mr. Booer, after consultation with practical bakers and others, set himself to fulfill, the observance of which lends to the present Blackfriars experiment much of its interesting character. Thus it was observed that, while it is not difficult to build an oven in a given spot, and bake bread in it, this cannot truly be called a baker's oven. By this term must be understood in particular an oven in an ordinary bakehouse, set in the usual style and worked by a man with his living to get by it. Before the problem of extending gas to bakers' ovens could be considered solved, it had to be attacked from this aspect. Mr. Booer, to do him full credit, seems to have early appreciated this fact in all its bearings. He not only saw that it was necessary to save gas, as much as possible, by putting it inside the oven; but he was told that, in order to meet with any general success, the cost of converting an oven to the gas system must be rigidly kept down to about ten or twelve guineas. The latter seems a particularly hard condition, when it is remembered that the only improved baker's oven in practical use at the present day is the steam oven invented by Mr. Perkins, which costs two or three hundred pounds to erect. Mr. Booer also had in mind the necessity that everything possible for a coal oven must likewise be performed by a gas oven; and in this respect he set himself to surpass the costly Perkins oven, which will not bake the common "batch" or household bread, generally the principal article of sale, more especially in populous and poor neighborhoods. The peculiar efficacy of the common coal fire in this respect proceeds from the essential principle of action of a brick oven, which is found simply in the fact that the work is done entirely by heat previously imparted to the tile bottom, roof, and sides of the oven, and thence radiated to the bread. No other kind of heat will bake batch-bread--i.e., loaves packed in contact with one another--which requires to be thoroughly soaked by a radiant heat in a close atmosphere of its own steam. Now, as a coal fire is eminently qualified to impart, by radiation and otherwise, this necessary store of heat to the brickwork, it is plainly a difficulty to effect the same purpose with a fuel which, of itself, can scarcely radiate heat at all. The system of the gas cooking-oven--the utilization of the heat of the combustion products as formed--is clearly inapplicable here; for a different kind of heat is needed, under conditions that would not sustain continuous combustion. Therefore, there is nothing for it but to heat the bottom and sides of the brick oven by the direct contact of powerful gas-flames; thus supplanting the coal fire, but leaving the actual work of baking to be done afterward by stored-up heat in the regular way.
Having settled the general principles of a system of this kind, there still remain a number of scarcely less important details, in the dealing with which lies the difference between practical success and failure. Thus it is not merely sufficient to heat an oven for bread baking; it is also necessary to heat it within the times and according to the habits of work to which the baker has been accustomed. Work in town bakeries begins at about midnight, or shortly after, and the condition of the oven must conform to the requirements of the dough, which vary from day to day and from season to season. In order to master all these niceties, as far as a knowledge of them is necessary to his purpose, Mr. Booer has spent many nights in the bakehouse in the Blackfriars Road; and has thereby obtained a command over the technicalities of the work which has served him in good stead, not merely for adjusting his gas heat, but in answering the innumerable objections always raised when a revolution in an immemorial trade is threatened. It is with considerable satisfaction that we are enabled to declare, after duly weighing all the conditions as to first cost and otherwise imposed by himself and others, that Mr. Booer has succeeded, upon these terms, in vindicating the claims of gas to be a cheap, efficient, and cleanly fuel for heating ovens under the control and according to the methods of working of the baker himself.
The oven with which this success has been achieved is one of two in the bakehouse of Mr. Loeber, of 161 Blackfriars Road. It measures 7 feet by 6 feet internally; being what is technically termed a 6 bushel oven. The alterations made by Mr. Booer consist in the first place in the removal of the flooring tiles, and the laying down of a new bottom, under which run a number of flues radiating from the side furnace. The throat of the furnace, where it enters the angle of the oven, is bricked up, and eight pieces of ¾-inch gun-barrel tubing project above this dwarf wall, and radiate fan-shaped under the dome of the roof. These are the gas-burners, which are supplied from a 1½-inch pipe led into the old furnace. The same pipe supplies the similar burners which are inserted in the flues under the oven bottom. This is really all the plant required. It should be remarked that these bottom flues are carried to different points of the side walls, and the products of combustion are allowed to rise upward into the oven through gaps left for the purpose. A supplementary supply of heated air is provided to help the combustion of the gas in these flues, which would otherwise be languid. When the gas is turned on from the main cock in the furnace either to the top or the bottom set of burners, a long match is used to light them from the same point. This is effected without risk of firing back, by the adoption of a specially constructed atmospheric nipple and shield, the pattern of which is registered. The flame from the top burners unites in a sheet of fire, which spreads out all over the crown of the oven, at the same time that the burners below are doing their work, and the products of combustion flow together through the oven to the chimney, which is the same that was used for coal. At first, as might be expected, there was considerable difficulty in finding the most suitable position of the chimney damper, aggravated in this case by the fact that the other oven worked with a coal fire into the same shaft. Finally, however, the two flues were disconnected with the happiest results. During the past fortnight the oven has been in regular use, and the bread has been sold over the counter in the ordinary course of trade. Two and three batches of bread have been baked in one day in this oven; the economy of its use, of course, increasing with the number of loaves turned out. As a rule the gas is lighted for about an hour before the oven is wanted, and about 250 cubic feet are used. Then the cocks are shut and the oven is allowed to stand closed up for ten minutes, in which time it ventilates itself, and the heat spreads over it. Then the batch is set, and the baking occupies from an hour to an hour and a half, according to the different classes of loaves. Two batches are baked with a consumption of about 620 cubic feet of gas; costing, at 2s. 10d. per 1000 cubic feet, just 11d. each batch for fuel. This cannot be considered costly. But the system possesses many other advantages. In the first place, it is much more cleanly than coal; for the oven never requires wiping out, which is usually done with a bundle of old rope called a "scuffle" and the operation is attended with a most unpleasant odor. Then there is no smoke--a great advantage from the point of view of the Smoke Abatement Institution. More to the purpose of the journeyman baker, however, is the fact that there is no stoking to be done, and he can therefore take his repose at night without having to attend to the furnace. Besides this the master has the satisfaction of knowing that the oven will always be hot enough if he simply attends to the time of lighting the gas--a consideration of no small moment. It is no mean testimony to the reality of Mr. Booer's success that Mr. Loeber, having seen his difficulties and troubles from the beginning, and marked how they have been overcome, is content to acknowledge that even this first example is capable of turning out bread in a condition to be sold over the counter. There is a good opening in this direction, for there are 6,000 bakeries in London alone, to every one of which Mr. Booer's system might be applied with advantage to the tradesman and his customers. And what may be done with gas at about 3s. per 1,000 cubic feet may certainly be done to still greater advantage in many towns where the price is lower. Mr. Booer has entered upon his work in a proper spirit. He has begun at the beginning, with the necessities of the baker; and has gone plodding on quietly, until he has achieved a noteworthy success. It may be hoped he will receive the reward which his perseverance merits.--Jour. of Gas Lighting.
Who was drowned on July 24 in attempting to swim through the whirlpool and rapids at the foot of the Falls of Niagara, was born at Irongate, near Dawley, in Shropshire, January 18, 1848. He was 5 feet 8 inches in height, measured 43 inches round the chest, and weighed about 14½ stone. He learnt to swim when about seven years old, and was trained as a sailor on board the Conway training-ship in the Mersey, where he saved the life of a fellow seaman. In 1870 he dived under his ship in the Suez Canal and cleared a foul hawser; and, on April 23, 1873, when serving on board the Cunard steamer Russia, he jumped overboard to save the life of a hand who had fallen from aloft, but failed, and it was an hour before he was picked up almost exhausted. For this he received a gold and other medals. He became captain of a merchant ship, but soon after he relinquished the sea and devoted himself to the sport of swimming.
At long distance swimming in salt water he was facile princeps, but he did not show to such advantage in fresh water. In June, 1874, he swam from Dover to the North-East Varne Buoy, a distance of 11 statute miles. On July 3, 1875, he swam from Blackwall Pier to Gravesend Town Pier, nearly 18 statute miles, in 4 hours 52 minutes. On the 19th of the same month he swam from Dover to Ramsgate, 19¼ statute miles, in 8 hours 45 minutes. On August 12, 1875, he tried to cross from England to France, and although he failed, owing to the heavy sea, he compassed the distance from Dover to the South Sand Head, 15½ statute miles, in 6 hours 48 minutes. On the 24th of the same month he made another attempt, which rendered his name famous all over the English-speaking world. Starting from Dover, he reached the French coast at Calais, after being immersed in the water for 21 hours 44 minutes. He had swum over 39 miles, or, according to another calculation, 45½ miles, without having touched a boat or artificial support of any kind. Subsequently he swam at the Lambeth Baths, and the Westminster Aquarium, and last year, at Boston, U.S., he remained in a tank nearly 128½ hours. Latterly he had suffered from congestion of the lungs, and his health had become much impaired.
CAPT. MATTHEW WEBB.
The story of his final and fatal effort needs here but a brief description. At two minutes past four, on July 24, Webb dived from the boat opposite the Maid of the Mist landing, and, amid the shouts and applause of the crowd, struck the water. He swam leisurely down the river, but made good progress. He passed along the rapids at a great pace, and six minutes after making the first plunge passed under the Suspension Bridge. Immediately below the bridge the river becomes exceedingly violent, and as the water was clear every movement of Webb could be seen. At one moment he was lifted high on the crest of a wave, and the next he sank into the awful hollow created. As the river became narrower, and still more impetuous, Webb would sometimes be struck by a wave, and for a few moments would sink out of sight. He, however, rose to the surface without apparent effort. But his speed momentarily increased, and he was hurried along at a frightful pace. At length he was swept into the neck of the whirlpool. Rising on the crest of the highest wave, he lifted his hands once, and then was precipitated into the yawning gulf. For one moment his head appeared above the angry waters, but he was motionless, and evidently at the mercy of the waves. He was again drawn under the water, and was seen no more alive. Some days later his body was found four miles below the fatal Rapids. It bore tokens of the fearful violence of the struggle which he had undergone. His bathing drawers were torn to fragments, and there was a deep wound in his head. An inquest was held, and the jury returned a verdict of "Found drowned."
Captain Webb was married about three years ago, and leaves a widow and two children. It is understood that he risked his life in this last fatal attempt to obtain money for the support of his family.--London Graphic.
These houses are situated in a pleasant part of Headingley, which is the favorite residential suburb in the locality of Leeds. As regards accommodation, the ground-floor of each house comprises good-sized drawing and dining rooms, each with bay windows; well-lighted entrance halls, opening upon wooden verandas; kitchen, pantry, and scullery; on first floor are three good bedrooms, a bathroom, and other necessary accommodation; on second floor are two additional bedrooms. The basement contains coal-place and larder.
In these houses an attempt has been made to produce conveniently-planned and well-arranged habitations, combined with a pleasing and picturesque exterior, without involving a large outlay of money. The materials used are brick of a deep red color for facings, red terra-cotta from Messrs. Wilcock & Co., of Burmantofts, for moulded strings, sills, etc., and a very sparing use of stone from the Harehills Quarries. The front gables are constructed of timber in solid scantlings, well framed, and pinned together with oak pegs, filled in and well backed behind with brickwork; the panels faced with cement, which, together with the cored cornice, are finished in vellum color. The whole of the woodwork of exterior is painted a neutral shade of peacock blue, forming an admirable contrast with the deep red of the bricks, the sashes and casements only being finished in cream color. The whole of the chimneypieces in the interior are carried out from the architect's special design; those in the drawing-rooms being of mahogany, finished in rosewood color, and those in dining-rooms of oak, stained with ammonia and dull wax polished.
SUGGESTIONS IN ARCHITECTURE.--SEMI-DETACHED
VILLAS,
BROMFIELD CRESCENT, HEADINGLEY, LEEDS.
The houses, with outbuildings and boundary walls, which have been erected for Mr. John Hall Thorp, of Bromfield, Headingley, have cost £1,450, or thereabouts, this amount not including the price of land. They have been carried out from the designs and under the superintendence of Mr. William H. Thorp, A.R.I.B.A., architect, of St. Andrew's Chambers, Park Row, Leeds.--The Architect.
In view of the possible approach of cholera, and the sanitary precautions that even the most neglectful of authorities are constrained to take, it is of some interest to us, says the Building News, to know how the poor are housed in the city of Paris, which contains, more than any city in the world, the opposite poles of luxurious magnificence and of sordid, bestial poverty. The statistics of the Parisian working classes in the way of lodgings are not of an encouraging nature, and reflect great discredit on the powers that be, who can be stern enough in the case of any political question, but are blind to the spectacle of fellow creatures living the life of beasts under their very eyes. In 1880, the Prefect of Police gave licenses to 21,219 arrivals in the city of French origin, and to 7,344 foreigners. In the succeeding year, the former had increased to 22,061, while the latter had somewhat diminished, being only 5,493. There was a census taken in 1881, from which it appeared that Paris contained 677,253 operatives and 255,604 employes and clerks, while out of every 1,000 inhabitants, 322 only were born in the city, and 565 came from the departments or the French colonies. The foreign element in the working classes has increased very rapidly, numbering 119,349 in 1876, to which by 1881 there was an addition of 44,689. To every 1,000 inhabitants, Paris now numbers 75 foreigners, though in 1876 the proportion was only 60. It may not be amiss to state that the annual increase of the Paris population is at the rate of 56,043 persons, and that in the five years 1876-81, the city received 280,217 additional mouths. The total population of the capital is 2,239,928, of whom 1,113,326 are males.
Returning to the poorer classes, we find that in 1872 they were estimated at 100,000; but that in 1873 they had risen to 113,733, and in 1880 to 123,735. It is unfortunate to be obliged to say that the majority of these people are housed worse in Paris than in almost any other great city in the world. There are two classes of lodgings for the poor--the one where the workman rents one or more rooms for his family, and, perhaps, owns a little furniture; the other, a single room tenanted for the night only by the unmarried man who pays for his bed in the morning and gets his meals anywhere that he can. Readers will remember how, under the auspices of M. Haussmann, western Paris was almost pulled down and transformed into a series of palatial boulevards and avenues. While the work lasted the Paris workman was well pleased; but he did not like it quite so much when the demon of restoration and renovation invaded his own quarters, such as the Butte des Moulins, and all that densely populated district through which the splendid Avenue de l'Opera now runs. The effect of all this was to drive the workman into the already crowded quarters at the barriers, such as La Gare, St. Lambert, Javel, and Charonne, where, according to the last statistics of the Annuaire, the increase was at the rate of 415 per 1,000. Of course the ill health that always pervaded these quarters increased also; and, from the reports of Dr. Brouardel and M. Muller, the number of deaths from typhoid and diphtheria were doubled in ten years. Dr. Du Mesnil, in making his returns for 1881 of convalescents from typhoid, remarked that the most unsanitary arrondissements were the 4th, 11th, 15th, 18th, and 19th--precisely those to which the principal migrations of laborers had taken place. The 18th arrondissement, which in 1876 had only 601 lodging houses with 8,933 lodgers, had, in 1882, over 850, with 20,816 inmates. In the 19th arrondissement there were 517 houses in 1876, with 9,074 lodgers, and 752 in 1882, with 17,662 inhabitants.
It is not only the crowded condition of the poor quarters that is such a standing menace to the health of the city, but also the shocking state of the rooms, which the unhappy lodgers are obliged to put up with. The owners of the property are, as happens in other places besides Paris, unscrupulous and grasping to the last degree, and have not only divided and subdivided the accommodation wherever possible, but have even raised the rental in nearly all cases. Whole families are crowded into a small apartment, icy cold in winter, an oven in summer, the only air and daylight which reaches the interior coming from a window which looks on to a dirty staircase or a still fouler court reeking with sewage. There are at the present time in Paris 3,000 lodgings which have neither stove nor chimney; over 5,000 lighted only by a skylight; while in 4,282 rooms there are four children in each below 14 years of age; 7,199 with three children; and 1,049 with four beds in each. The Parisian population has augmented only 15 per cent. in seven years; but the district of poor lodging houses has increased by twenty per cent., and the number of lodgings by about 80 per cent. It is true that a law was passed in 1850 to provide for the sanitary supervision of this class of property; but in Paris the law is a dead letter, and, although it is now active in the provinces and in places like Marseilles, Lyons, Bordeaux, and Nantes, it is applied, even there, in a jerky and intermittent manner.
Perhaps the worst of the abominable dogkennels called houses was the group known as the Cité des Kroumirs, in the 13th arrondissement, which, by a strange irony, was built on land belonging to the Department of Public Assistance, which was let out by that body to a rich tenant, who sublet it to these lodging-house owners. This veritable den of infection and misery has now been demolished; but there are plenty of others quite as bad. Notably, there is the Cite Jeanne d'Arc (a poor compliment to have named it after that sturdy heroine), an enormous barrack of five stories, which contains 1,200 lodgings and 2,486 lodgers. No wonder that it was decimated in 1879 by smallpox, which committed terrible ravages here. The Cité Dore is grimly known by the poor-law doctors as the "Cemetery Gateway." The Cite Gard, in the Rue de Meaux, is inhabited by 1,700 lodgers, although it is almost in ruins. The Cite Philippe is tenanted by 70 chiffonniers, and anybody who knows what are the contents of the chiffonnier's basket, or hotte, may easily guess at the effluvia of that particular group of houses. A large lodging-house in the Rue des Boulangers is tenanted by 210 Italians, who get their living as models or itinerant musicians. Both house and tenants are declared to be unapproachable from the vermin.
It is some satisfaction to know that these houses have lately awakened the apathy of some of the public bodies, and that more than one scheme is being put forward with a view of erecting proper industrial dwellings. The Municipal Council is negotiating with the Credit Foncier for the erection of a certain number of cheap houses, which, for the space of twenty years, will be exempt from all taxes, such as octroi, highway, door and window tax, etc. There are also one or two semi-private companies, which are occupying themselves with the question, and it is to be hoped that the rumors of the pestilence in Egypt may hasten the much-needed reform.
There can be no doubt, says the Engineer, that the inventor who could supply in a really portable form a machine or apparatus that could give out two or three horse power for a day would reap an enormous fortune. Up to the present time, however, nothing of the kind has been placed in the market. Gas is laid on to most houses now, and gas engines are plenty enough, yet they do not meet the want which a storage battery may be made yet perhaps to supply.
To prove the incorrectness of Helmholtz's statement that beats do not colesce into musical sounds, but that the ear will distinguish them as a rumbling noise, even when their number rises as high as 132 vibrations per second, Rudolph Koenig has constructed a series of tuning forks, recently presented by President Morton to the Stevens Institute of Technology. The following table exhibits the number of vibrations per second of these forks, the ratios of their vibrations when two are sounded together, the number of beats produced, and the resultant sound:
| Vibrations per second. | Ratio. | Beats. | Sounds. | |||
|---|---|---|---|---|---|---|
| 3840 | :4096 | 15 | : | 16 | 128 | Ut2 |
| 3904 | : " | 61 | : | 64 | 96 | Sol1 |
| 3936 | : " | 123 | : | 128 | 80 | Mi1 |
| 3968 | : " | 31 | : | 32 | 64 | Ut1 |
| 3976 | : " | 497 | : | 512 | 60 | Si-1 |
| 3989.3 | : " | 187 | : | 192 | 53.3 | La-1 |
| 4000 | : " | 125 | : | 128 | 48 | Sol1 |
| 4010.7 | : " | 47 | : | 48 | 42.7 | Fa-1 |
| 4016 | : " | 251 | : | 256 | 40 | Mi-1 |
| 4024 | : " | 503 | : | 512 | 36 | Re-1 |
| 7936 | :8192 | 31 | : | 32 | 128 | Ut2 |
| 8064 | : " | 63 | : | 64 | 64 | Ut1 |
| 8096 | : " | 253 | : | 256 | 48 | Sol-1 |
| 8106.7 | : " | 95 | : | 96 | 42.7 | Fa-1 |
| 8112 | : " | 507 | : | 512 | 40 | Mi-1 |
| 8120 | : " | 1015 | : | 1024 | 36 | Re-4 |
| 8128 | : " | 127 | : | 128 | 32 | Ut-4 |
On sounding two forks nearly in unison, the sound heard corresponds to a number of vibrations equal to the difference of the numbers of vibrations of the forks.
On sounding two forks, one of which is nearly the octave of the other, the ear perceives a sound, which is that given by vibrations whose number equals the difference in the number of vibrations of the higher fork and the upper octave of the lower fork.
Koenig has also found out the laws of the resultant sounds produced by other intervals than the octave, and has extended his researces to intervals differing by any number of vibrations, as may be seen from the above table.
His conclusion is that beats and resultant sounds are one and the same phenomenon.
Thus, for example, the lowest number of vibrations capable of producing a musical sound is 32 per second; in like manner, a clear musical sound is produced by two simple notes of sufficient intensity which produce 32 beats per second.
Koenig also made a very ingenious modification of the siren for the purpose of enabling Seebeck to sound simultaneously notes whose vibrations had any given ratio. It is furnished for this purpose with eight disks, each of which contains a given number of circles of holes arranged at different angular distances. A description of this instrument, which is also the property of the Stevens Institute, and of Seebeck's experiments is thus given in a letter by Koenig himself.
Effects produced when the isochronism of the shocks is not perfect.
A.
In order to produce a note, the succession of shocks must not deviate much from isochronism.
If the isochronism is but little impaired, we obtain a note corresponding to the mean interval of the shocks.
If the intervals between the shocks are alternately t and t', and if the difference between t and t' is slight, we obtain the two notes t+t' and (t+t')/2. If the intervals between the shocks are alternately t, t', and t'', we obtain the two notes t+t'+t'' and (t+t'+t")/3.
Disk No. 1 has--
On circle No. 1 12 holes, angular distances t=30° " " 2 24 " " " 15° " " 3 36 " " " 10° " " 4 36 " at irregular distances. " " 5 36 " distances t= 10½°, t'=l0°,t''=9½° " " 6 36 " " 11° 10° 9° " " 7 36 " " 16° 14° " " 8 36 " " 16½° 13½°
Circle No. 8 produces the two notes of circles 1 and 2; circle No. 7 the same, but the low note is stronger than in 8.
Circle 6 produces the notes of circles 1 and 3, and so does circle 5, but in the latter the low note is stronger than in 6.
Circle 4 produces a noise approximating only to the note of circle 3.
By pulling out one of the buttons of the wind chest, we admit the air through eleven holes at a time, having an angular distance of 30° and directing it against the corresponding circle of holes on the turning disk. If the arrangement of holes is not repeated identically twelve times on the same circle, we cannot, of course, make use of the above arrangements of holes of the wind tube, and we must then employ one of the movable brass tubes, which communicate with the interior of the wind chest by means of rubber tubes and stopcocks. The experiment with disk 1, circle 4, for example, requires the use of one of these two tubes, while the perforated wind tube of the wind chest may be used with all the other circles of the same disk.
B.
If t is much less than t', while t' is a multiple of t, the note (t+t')/2 disappears, and the notes t+t' and t are heard.
Disk No. 2 has--
On circle No. 1 12 holes, distances 30° " " 2 36 " " 10° " " 3 48 " " 7½° " " 4 60 " " 6° " " 5 24 " " t= 5°, t'=25° " " 6 24 " 6° 24° " " 7 24 " 7½° 22½° " " 8 24 " 10° 20°
Circle 8 produces the notes of circles 1 and 2; circle 7, those of 1 and 3; circle 6, those of 1 and 4; and circle 5, the note of circle 1 and of its sixth harmonic.
C.
If the same circular arc is divided into m and n equal parts; that is to say, if mt=nt', we obtain the notes m and n.
Disk No. 3 has--
Distances.
On circle No. 1 24 holes, distances 15°
" " 2 24 " " 15° & 27 holes, 13-1/3°
" " 3 24 " " 15° " 30 " 12°
" " 4 24 " " 15° " 32 " 11-1/4°
" " 5 24 " " 15° " 36 " 10°
" " 6 24 " " 15° " 40 " 9°
" " 7 24 " " 15° " 45 " 8°
" " 8 24 " " 15° " 30, 36, & 48 holes
Circle 1 produces a single note, circle 2 a second, circle 3 a third, circle 4 a fourth, 5 a fifth, 6 a sixth, 7 a seventh, and 8 a perfect chord.
Experiments to prove that the shocks may proceed from two or several different places to conspire in the formation of a note, provided that the isochronism of the shocks is sufficiently exact, and that the shocks are produced in the same direction.
Disk No. 4 has--
On circle 1 24 holes.
" " 2 36 "
" " 3 23 "
" " 4 12 at an angular distance of 10° from the holes
of circle 3.
" " 5 12 holes at an ang. dist. of 20° from those of circle 3
" " 6 12 " " " 0° "
" " 7 12 " " " 15° "
" " 8 12 " " " 15° "
1. If from the same side two currents of air at an angular distance of 15° are directed against circle No. 8 of 12 holes, we obtain the octave of the note produced by the same circle if only one current is used.
The wind-chest is provided with a special arrangement for this experiment. By pulling out button 8, we give vent to 12 currents of air spaced like the twelve holes of the disk; on pulling out button 9 we also produce 12 currents, but they are situated just between the first. Each of these two buttons pulled out alone will produce the same note corresponding to 12 holes, but drawn together they produce the octave, or the note of circle 1.
2. If two currents of air are directed against two similar circles whose holes are situated on the same radii, we obtain the same result.
In this experiment, circles 7 and 8 are sounded by pulling out buttons 7 and 9.
3. When two currents of air are directed on the same radius against two circles of similar holes arranged alternately, these circles sounded simultaneously will produce the octave of the note which one of them would give alone.
This experiment is performed by sounding circles 6 and 7 and pulling out buttons 6 and 7.
4. If we direct three currents of air on the same radius against three similar circles having holes alternating by a third of the distance between two holes of the same circle, the three circles together produce the fifth of the octave (Note 3) of a single circle.
Circles 3, 4, and 5 sounded together emit the note of circle 2.
(By sounding only two circles, 3 and 4, or 4 and 5, we make the same experiment with two circles as disk No. 2 enabled us to make with circle 8 alone; also, by sounding circle 3 alone, we obtain the note corresponding to 12 holes; then pulling out button 4, the notes corresponding to 12 and 36 holes are heard suddenly and very strongly; but as soon as circle 5 is sounded also, the note of 12 disappears completely, and we have left only that corresponding to 36 holes.)
Effects of interference produced by shocks in opposite directions.
1. If we direct against a circle of holes two currents of air in opposite directions, the note obtained with a single current is very much weakened, if the two currents reach the holes simultaneously. If the impulses are not isochronous, the intensity of the note is increased.
2. If the two currents are directed against two circles of the same number of holes, the effect is the same as for the two preceding cases.
3. If two currents of air are directed against two circles, one of which has twice as many holes as the other, we obtain only the low note if every shock of one is isochronous with every shock of the other.
We obtain the notes of both circles, one of which is the octave of the other, if there is no isochronism between the shocks.
Disk No. 5 has three circles of 36, 36, and 72 holes. The air currents are directed against the circles of holes through the movable tubes, made so that they can be detached at pleasure. All these experiments require great precision in the arrangement of these wind tubes. To make sure that the tubes are simultaneously before two holes of the disk, it is well to put little rods through the holes, reaching into the wind tubes, and to remove them only when the tubes are firmly attached. The experimenter should be careful also to place the two tubes exactly at the same distance from the turning disk. It is clear that notwithstanding all these precautions we never obtain perfect interference, but only the weakening of notes that ought to disappear entirely if all the arrangements were made with mathematical exactness, and also if the ear could have absolutely the same position with regard to impulses produced in opposite directions.
Beats.
Disk No. 6 has--
8 circles of holes to the number of 1, 2, 23, 24, 25, 47, 48, 49.
Circles 3 and 4, 4 and 5, 6 and 7, and 7 and 8 ought to produce as many beats as circle 1 produces simple shocks; and circles 3 and 5, 6 and 8, as many beats as circle 2 produces simple shocks; but we must content ourselves in these experiments with a much less perfect result, for the following reasons: The disk never being rigorously plane, alternately approaches the single wind pipe and recedes from it. No matter how slight this deviation is, every sound given by a single circle is heard with periodical intensities which complicate the phenomenon. This inconvenience could be avoided by placing several wind-pipes around the circle; but while we can extend the period of the holes in two circles (whose difference is 1) around the whole circle by blowing through a single wind tube, we would be compelled to limit it to the distance between two wind tubes, and it would become too short; for, when the disk rotates with a velocity sufficient to produce notes high enough and intense enough, the beats become too numerous to be easily perceived.
Besides these provisions, which sufficiently illustrate the points to which we desire to call especial attention, Koenig also furnishes two more disks.
The seventh contains 8 circles having 48, 54, 60, 64, 72, 80, 90, and 96 holes respectively. The 1st, 3d, 5th, and 8th will produce a perfect chord when the air is admitted through the 11 holes in the wind chest; with one wind tube the entire gamut may be obtained.
Finally the eighth disk contains 8 circles of holes, whose numbers are in the ratio of 1:2:3:4, etc., and which may be used to illustrate harmonics. C. F. K.
[Footnote: Continued from SUPPLEMENT No. 391, page 6240.]
To have these movements occur in a constant and invariable manner upon the surface of water, and especially upon mercury, it is necessary to take precautions in regard to cleanliness, this being something that we have purposely neglected to mention to our readers. For we wished, through this voluntary omission, to stimulate their sagacity by bringing them face to face with difficulties that they will perhaps have succeeded in overcoming, with causes of error that they will have perceived, and the principal one of which is the want of absolute cleanliness in the water, vessels, and instruments that they may have used for the experiments.
Thus, very probably, they will have more than once seen the camphor remain immovable when placed in vessels in which they had hoped to be able to see it undergo its gyratory and other motions. Their astonishment will have been no less than our own was when we noticed the sudden cessation of the camphor's motions under the influence of vitreous or metallic objects, such as glass rods or tubes, pieces of gold, silver, or copper coin, table knives, etc., dipped into the liquid in which such motions were taking place before the immersion of the objects under consideration.
The instantaneously sedative power of the human fingers, or of a hair, will have, perhaps, reminded them of some sort of sorcery, or of some diabolic art worthy of the great Albert.
APPARATUS FOR THE STUDY OF THE MOTIONS
OF CAMPHOR.
As for ourself, we confess that, after repeating the curious experiments of Mr. Dutrochet day after day, and scrupulously following his directions, we have, in the presence of our results, that were exactly identical with his, almost been tempted to believe ourself to be the victim of some occult power, or at least of some optical illusion, the true cause of which remained a mystery to us. Finally, after many fruitless attempts to find a key to the enigma that engaged our attention, the light finally dawned upon us, and then shone straight in our eyes.
In comparing the last results of our experiments with those that we had obtained previously, we saw, for example, that the camphor moved in the test glasses at a level that was notably higher than that at which its gyration took place the day before, or the day before that. And yet we had always used the same vessels, the same water, and particles detached from the same lump of camphor.
To what, then, could be due the difference observed between the two levels at which we had, in the first and last place, seen the camphor execute its movements? In the absence of any answer that was satisfactory, we finally suspected that the difference that we had noticed was ascribable to the fact that, after the numerous washings that the apparatus had been submitted to in having water poured into them to repeat the experiments, they had gradually been freed from impurities of whatever nature they might have been, and which, unbeknown to us, might have soiled their sides.
Starting with this idea, which was as yet a hyphothetical one, we began to wash our hands, glasses, etc., at first with very dilute sulphuric acid, and then with ammonia. Afterward we rinsed them with quantities of water and dried them carefully with white linen rags that had been used for no other purpose; and finally we plunged them again into very clean water. We thus cut the Gordian knot, and were on the right track.
In fact, on again repeating Mr. Dutrochet's experiments, with that minute care as to cleanliness that we had observed to be absolutely necessary, we saw crumble away, one after another, all the pieces of the scaffolding that this master had with so much trouble built up. The camphor moved in all our vessels, of glass or metal, and of every form, at all heights. The immersed bodies, such as glass tubes, table knives, pieces of money, etc., had lost their pretended "sedative effect" on a pretended "activity of the water," and on the vessels that contained it. The so-called phenomenon of habit "transported from physiology into physics," no longer existed.
The likening of the apparatus employed to obtain motions of camphor upon water, with the entirely physiological apparatus by means of which nature effects a circulation of the liquid contained in the internodes of Chara vulgaris, had proved a grave error that was to be erased from the science into which it had been introduced by its author with entire good faith. The true cause of life had not then been unveiled, and the new agent designated as diluo-electricity vanished before the very simple and authentic fact that camphor moves rapidly upon the surface of very pure mercury, in which no one would assuredly suppose that that volatile substance could dissolve.
Mr. Dutrochet attaches great importance to the manner in which the water is poured (with or without agitation) into the vessel with which the experiment is performed. The matter is in fact of little or no importance, and to prove this, it is only necessary to employ a test glass (see figure) provided with a lateral tube, A, that terminates in a lower tubulure, B, above which there is a contraction, C. Upon pouring water into the lateral tube until the level reaches D, and placing a particle of camphor on its surface, the camphor will be seen to continually move about, even when the liquid has reached the upper edge of the vessel. To reduce the level to various heights, it is only necessary to revolve the tube in the cork through which it is fitted to the tubulure. In proceeding thus, agitation or collision of the water is avoided; and yet if the test glass is very clean, the camphor will continue to move at every level of the water.
But, some one will doubtless say, how do you explain the stoppage in the motions of the camphor on the surface of water contained in vessels that are not perfectly clean? Before answering this question, let us say in the first place that the cause of the motions under consideration is due to nothing else but the evaporation of this concrete oil--to effluvia that escape from all parts and that exert upon the body whence they emanate a recoiling action exactly like that which manifests itself in an ælopile mounted upon a brasier, or, better yet, in the explosion of a sky-rocket. A portion of these camphory vapors, as well as a small portion of the camphor itself, dissolves in the water and forms upon its surface an oily layer which is at first very slight, but the thickness of which may increase in time until it becomes (especially if the vessel is narrow) a mechanical obstacle to the gyration of the small fragments of camphor that it imprisons, and whose evaporation it prevents. Now, as this layer of volatile oil may and does evaporate, in fact, after a certain length of time, the camphor then resumes its gyratory motions; but there is not the least reason in the world for saying on that account that it "has habituated itself to the cause which had at first influenced it, and that, too, in modifying itself in such a way as to render null the influence of a cause that has not ceased to be present" (Dutrochet, l.c.., p. 50).
We have been enabled to convince ourself of the existence of this oily layer of camphor when it was of a certain thickness by introducing under the water on which it, had formed, a few drops of sulphuric ether whose sudden evaporation produced sufficient cold to instantaneously congeal the layer in question and thus render it perfectly visible to the eye. The slight layer of greasy matter that habitually lines the sides of vessels from whence no effort has been made to remove it, produces effects exactly like those of the oil of camphor, that is to say, that in measure as it becomes thicker it likewise arrests the motions of the concrete volatile essence.
This is precisely what happens in a test-glass in which we see the camphor in motion become immovable if the level of the water be raised a few centimeters, and, more especially, if it be raised to the upper edge of the apparatus. In its slow ascent the liquid licks up, so to speak, the oily layer that lines the inner surface of the vessel, and this material spreads over the surface of the water and forms thereupon a layer which, in spreading over the bit of camphor itself, prevents its evaporation, and, consequently, its motions. The existence of the layer under consideration cannot be doubted, since it is made to disappear by causing the water to-overflow from the edges of the vessel, and, more easily still, by spreading a piece of filtering paper over the liquid in which the camphor is in a state of rest. As soon as the paper is removed (without the water being touched by the fingers, it should be understood), the camphor resumes its motions and afterward continues them at all levels.
The fingers themselves, provided they are very clean, have no power to stop the gyration. The following experiment, which is easy to repeat, is an unquestionable proof of this.
Wash carefully the middle finger with aqua ammonia, and afterward with plenty of water, and then dip it into a drinking glass in which a fragment of camphor is rapidly moving, and the gyration will not be stopped. But it will be made to stop instantly if the finger in its natural state (that is, covered with the fatty substances that ordinarily soil the fingers, especially in summer) be dipped into this same glass.
Movements of Camphor upon Mercury.--In order to study the motions of camphor, mercury possesses, as compared with water, a great advantage, and that is that we can easily assure ourselves of the degree of cleanliness of this metal by means of the condensed breath. The vapory-deposits thereon in a uniform manner if the mercury is perfectly clean, but forms variously shaded and more persistent spots if it is soiled by foreign bodies But it is extremely difficult to clean mercury completely. To do so Mr. Boisgiraud and I take distilled mercury and leave it for a long time in contact with concentrated sulphuric acid, taking care to often shake the mixture. Then, after removing the greater part of the acid, we throw the metal into a vessel containing quick lime in powder, and finally pass it through a filter containing a few holes in its lower part.
Purified by this process, mercury not only permits of the motions of camphor on its surface, but renders visible the traces of the vapors that escape from it, and which resemble small tadpoles with a long tail that are endowed with very great agility. Nothing is more curious than to see the particle of camphor successively ascend and descend the strongly pronounced curves presented by the mercury near the sides of the vessel that contains it. On raising the temperature of the metal slightly, the motions of the camphor on its surface are accelerated, and the same effects occur with water that has been slightly heated.
The experiments that we have just called attention to show what importance slight impurities may have upon certain results. "They prove," says our learned colleague Mr. Daquin, "that there exists upon polished substances an imperceptible coating of those fatty matters which serve to-day to explain Moser's images." We find therein also a manifest proof and a rational explanation of those grave errors into which the presence of these fatty matters, that have hitherto been scarcely suspected, led so clever and so distinguished a scientist as the illustrious discoverer of endosmosis.--N. Joly, in La Nature.
We present a diagram, on exposition at the last Brewers' Convention in Detroit, of the racking device, devised by J. E. Siebel in 1872, and used at that time in the brewery of Messrs. Bartholomae & Roesing, in Chicago. The object of the apparatus is to retain as much carbonic acid in the beer as possible while racking the same off into smaller packages from the storage vats. The importance of this measure is apparent to every one who knows what pains are taken to preserve the presence of this constituent in all the former stages of the brewing process. In the method of racking off which is in present use in most breweries, the beer is forced through a rubber hose from the cask in the store vault to the barrels, kegs, and smaller packages in the fill room. Owing to the excess of pressure in the beer as it enters the keg, it is evident that a large amount of the carbonic acid gas must escape. The escape of carbonic acid during the process of racking off is indeed so large that even a small difference in the pressure of the atmosphere causes a remarkable difference in this respect. It is, therefore, evident that if a larger pressure can be maintained while racking off, a larger amount of carbonic acid gas will remain in the beer. It is true that the racking off will take a little longer time if done under pressure, but this inconvenience is certainly insignificantly small, when compared with the other labors and troubles daily undergone in a brewery, for the sole purpose to preserve in the beer the carbonic acid in that form in which it has been formed during the fermentation, and in which form it has far more refreshing and other valuable properties than in any other form in which it may be subsequently introduced into the beer by artificial means. The apparatus designed in the accompanying cut is calculated to artificially produce a higher pressure of the atmosphere, at least within the keg which is to be filled with beer. For this purpose, the beer from the store cask running through the pipe, B, enters the keg through a hollow copper bung, fitting light into the bung hole by means of a rubber washer. The air contained in the keg, being replaced by the beer, is forced out by means of the hollow copper bung, taking its course through the pipe, inscribed "Glass Gauge," until it is allowed to escape in the standpipe, C, containing a column of water, the height of which designates the pressure within the keg, and a consequently increased retention of carbonic acid gas. If the keg or barrel is filled with beer, the same becomes apparent from the beer showing itself in the glass gauge; then the faucet, B, is closed, the copper bung is lifted out of the bung hole, and the beer contained in the pipe is just sufficient to completely fill the keg, which is then bunged up, while the apparatus is transferred to the next keg. Should the attendant carelessly neglect to close the faucet in proper time, the surplus beer will not necessarily be wasted, but will be collected in the vessel, D, whence it can be drawn off through e.--Chemical Review.
Hermann W. Vogel has made a comparative study of the properties of silver bromide, obtained by precipitation in an aqueous solution of gelatin, and those of the same compound prepared by precipitation in an alcoholic solution of collodion. In 1874 Stas called attention to six modifications of silver bromide. One of these, granular bromide of silver, obtained by boiling the flocculent precipitate for several days with water, he stated, was the most sensitive to light of all substances known; exposure for two or three seconds to the pale blue flame of a Bunsen burner being sufficient to blacken it. Important as this fact was for photographers it was not applied for years, and it was only in 1878, when, it having been found that silver bromide precipitated in a gelatine solution and boiled for several hours becomes much more sensitive to light, that the remarks of Stas was recalled. Today these observations have become of the greatest importance to practical photography. They have led to the preparation of the silver bromide gelatin emulsion and the silver bromide gelatin plates, which are twenty times more sensitive than the silver iodide collodion plates, and have become indispensable when impressions are to be taken in a dim light.
The extraordinary sensitiveness of silver bromide in gelatin seemed the more remarkable since it was known that silver bromide in collodion is only moderately sensitive. The explanation was sought for in various directions, but as the result of numerous investigations it appears that the chief cause of the difference is the presence of different modifications of silver bromide. From a consideration of the work already done on the subject, Vogel suspected that silver bromide precipitated in an aqueous colloidal liquid would have notably different properties from silver bromide precipitated in an alcoholic colloidal solution. Silver bromide was prepared in many different ways. Emulsions were made in bromide solutions containing gelatin or collodion (the former aqueous, the latter alcoholic), some with the aid of heat, others without. Part of the emulsion was then poured upon plates kept at a moderate temperature and dried. The remainder was boiled or treated with ammonia before being applied to the plates. He also precipitated silver bromide in dilute gelatin or collodion solutions, allowed it to settle completely, washed the precipitate, and mixed it with a new portion of gelatin or collodion before applying it to the plates. Finally he precipitated pure silver bromide, in the absence of all colloids, by means of pure aqueous or alcoholic solutions of bromides and attempted to bring this upon plates, using gelatin or collodion as a cement. The result of all these experiments is that there are essentially two modifications of silver bromide, the one being obtained by precipitation in aqueous, the other in alcoholic solutions. The first, on account of the position of the maximum of sensitiveness for the solar spectrum, he calls blue sensitive, the other, for the same reason, indigo sensitive.
It is of no consequence whether the aqueous or alcoholic solution in which the silver bromide is formed contains gelatin or collodion, or whether the precipitation is effected with excess of bromide or of silver nitrate. It makes no difference whether the solution is hot or cold, or whether the silver bromide is treated with ammonia or whether it is boiled or not. The only necessary condition is that in precipitating indigo sensitive silver bromide the solutions must contain at least 96 per cent of alcohol. From aqueous alcoholic solutions blue sensitive silver bromide is precipitated.
Besides the difference of sensitiveness toward the solar spectrum, these modifications of silver bromide exhibit other characteristic differences in properties which indicate beyond a doubt that they are two essentially different modifications of the same substance. Among these are, 1st. Their unequal divisibility in gelatin or collodion solutions. The indigo sensitive silver bromide cannot be distributed through a gelatin solution, while the blue sensitive modification does so very readily. 2d. Their unequal reducibility; the blue sensitive silver bromide being reduced with much greater difficulty than the indigo sensitive variety. 3d. Their different action toward chemical and physical sensitizers. 4th. Their different action toward photographic developers. 5th. Their different action under the influence of heat. The blue sensitive variety if heated under water has its sensitiveness perceptibly increased, while the other is not changed by such treatment.
A direct transformation of one modification into the other has not yet been accomplished. The effect of the light upon these substances is incipient reduction, and we might hence suppose that the more reducible indigo sensitive variety would be the more sensitive to light. But this is not the case, because it is not chemical reducibility, but the absorption power for light that is of the greatest importance. Now the blue sensitive silver bromide has a greater absorption power than the indigo sensitive variety, and hence its greater sensitiveness. Silver chloride prepared by methods similar to those used in making the two forms of bromides was also found to exist in two modifications. One is designated as ultra violet sensitive, the other as violet sensitive silver chloride.--Amer. Chem. Jour.
[Footnote: Read before the Society of Public Analysts on the 28th June, 1883.]
Some discussion having recently taken place as to the value of New Zealand coal as a fuel, the following results of a somewhat full analysis may be worthy of being placed on record.
The sample to which the results refer consisted of large brownish black lumps, many of which showed woody structure; the fractures were conchyloid, the surface shiny and highly reflecting. It was interspersed with a considerable amount of an amber colored resin. When powdered it appeared chocolate brown. It burned readily, the flame being bright and very smoky. Its ash was light and reddish brown.
It consisted of--
Water (loss at 212° F.) 20.09
Organic and volatile matter 75.19
Ash 4.72
------
100.00
The organic and volatile constituents had the following percentage composition--
Carbon 71.26
Hydrogen 5.62
Oxygen 21.58
Nitrogen 1.06
Sulphur 0.48
------
100.00
The ash was composed of--
Silica 27.26
Alumina 26.48
Oxide of iron 12.98
Lime 20.19
Magnesia 3.42
Sulphuric acid 9.47
Alkalies and loss 0.20
------
100.00
From these figures the composition of the coal itself calculates as under--
Water 20.09
Carbon 53.58
Hydrogen 4.23
Oxygen 16.23
Nitrogen 0.80
Sulphur 0.36
Silica 1.29
Alumina 1.25
Oxide of iron 0.61
Lime 0.95
Magnesia 0.16
Sulphuric acid 0.44
Alkalies 0.01
------
100.00
One ton furnished 8,458 cubic feet of gas and 8 cwt. of coke.
The very high proportion of water contained in the sample is very remarkable. It was so loosely combined, that even at ordinary temperature it gradually escaped, the coal crumbling to small pieces. The large amount as well as the high percentage of oxygen characterize the so called coal as a lignite, with which conclusion the physical characters of the sample are in perfect harmony.
The resin to which I have referred has not been further analyzed. It was found to be insoluble in all ordinary menstrua, such as alcohol, ether, carbon disulphide, benzene, or chloroform, and neither attacked by boiling alcoholic potash nor by fusing alkali. On heating it swells up considerably and undergoes decomposition, but does not fuse.
The coal may be valuable as a gas coal and for local consumption, but the large proportions of water and of oxygen militate against its use as a steam producer, only 58 per cent. of it being really combustible.
The method in question is recommended as easy, expeditious, and accurate. It consists in precipitating all the manganese in the state of peroxide, dissolving it in a ferrous solution so as to bring back the manganese to the manganous slate, and determining volumetrically, by means of potassium permanganate, the quantity of ferrous salt which has been converted into ferric. The method of rapidly precipitating manganese peroxide is peculiar. If we act upon cast-iron or steel with nitric acid and potassium chlorate in certain proportions, and boil the mixture, the manganese is completely precipitated in the state of peroxide insoluble in nitric acid, but retaining a small quantity of ferric oxide. Suppose that we have a sample of steel or manganiferous cast-iron containing less than 7 per cent of manganese. Three grammes are treated in a small flask with 40 c. c. of nitric acid, of sp. gr. 1.20, added little by little. The liquid is stirred, and ultimately heated to complete solution. It is withdrawn from the fire, and 15 grammes potassium chlorate are added, and then 20 c. c. of nitric acid at sp. gr. 1.40. It is boiled for about fifteen minutes, until the escape of chlorine ceases; all the manganese is found thrown down as peroxide; hot water is added, the mixture is filtered, and the precipitate washed with boiling water. To dissolve the manganese peroxide thus obtained we measure exactly 50 c. c. of an acid solution of ferrous sulphate, made up with 40 grammes ferrous sulphate to 750 c. c. water and 230 c. c. sulphuric acid (full strength). The 50 c. c. are poured into the flask in which the sample has been dissolved, and to which a little peroxide adheres, and it is then poured upon the precipitate and the filter in a Berlin-ware capsule. The manganese peroxide dissolves very readily, transforming its equivalent of ferrous sulphate into ferric sulphate. The liquid is then diluted to 100 or 150 c. c. for the next operation. We then take a solution of permanganate formed by the same proportions as are used in determining iron by the process of Margueritte (5.65 grammes of the crystalline salt per liter of water), and determine its standard exactly. By means of this liquid we determine volumetrically the quantity of ferrous sulphate remaining in the solution of manganese. We take then 50 c. c. of the original solution of ferrous sulphate diluted as above, and determine the total ferrous salt.
The difference between the two determinations corresponds to the ferrous salt which has been peroxidized by the manganese peroxide. The quantity of iron thus peroxidized multiplied by 0.491 gives the quantity of manganese contained in the portion operated upon. In the case of a steel or cast iron containing but little manganese it is convenient to dissolve the peroxide in 25 c. c. only of the ferrous solution. Small Gay-Lussac burettes may then be used in the titration of only 0.010 meter internal diameter, and graduated into one-twentieth c. c., which allows of great exactitude in the determination. For a spiegeleisen not more than 1 gramme of the sample should be taken, and for a ferro-manganese 0.3 gramme.
Manganese is one of the heavy metals of which iron may he taken as the representative. It is of a grayish white color, presents a metallic brilliancy, and is capable of a high degree of polish, is so hard as to scratch glass and steel, is non-magnetic, and is only fused at a white heat. As it oxidizes rapidly on exposure to the atmosphere, it should be preserved under naphtha.
It occurs in small quantity in association with iron in meteoric stones; with this exception it is not found native. The metal may be obtained by the reduction of its sesquioxide by carbon at an extreme heat.
Manganese forms no less than six different oxides--viz., protoxide, sesquioxide the red oxide, the binoxide or peroxide, manganic acid, and permanganic acid. The protoxide occurs as olive-green powder, and is obtained by igniting carbonate of manganese in a current of hydrogen. Its salts are colorless, or of a pale rose color, and have a strong tendency to form double salts with the salts of ammonia. The carbonate forms the mineral known as manganese spar. The sulphate is obtained by heating the peroxide with sulphuric acid till there is faint ignition, dissolving the residue in water and crystallizing. It is employed largely in calico printing. The silicate occurs in various minerals.
The sesquioxide is found crystallized in an anhydrous form in braunite, and hydrated in manganite. It is obtained artificially as a black powder by exposing the peroxide to a prolonged heat. When ignited it loses oxygen, and is converted into red oxide. Its salts are isomorphous with those of alumina and sesquioxide of iron. It imparts a violet color to glass, and gives the amethyst its characteristic tint. Its sulphate is a powerful oxidizing agent.
The red oxide corresponds to the black oxide of iron. It occurs native in hausmannite, and may be obtained artificially by igniting the sesquioxide or peroxide in the open air. It is a compound of the two preceding oxides.
The binoxide, or peroxide, is the black manganese of commerce, and the pyrolusite of mineralogists, and is by far the most abundant of the manganese ores. It occurs in a hydrated form in varvicite and wad. Its commercial value depends upon the proportion of chlorine which a given weight of it will liberate when it is heated with hydrochloric acid, the quantity of chlorine being proportional to the excess of oxygen which this oxide contains over that contained in the same weight of protoxide. When mixed with chloride of sodium and sulphuric acid it causes an evolution of chlorine, the other resulting products being sulphate of soda and sulphate of protoxide of manganese. When mixed with acids, it is a valuable oxidizing agent. It is much used for the preparation of oxygen, either by simply heating it, when it yields 12 per cent. of gas, or by heating it with sulphuric acid, when it yields 18 per cent. Besides its many uses in the laboratory, it is employed in the manufacture of glass, porcelain, and kindred wares.
Manganic acid is not known in a free state. Manganate of potash is formed by fusing together hydrated potash and binoxide of manganese. The black mass which results from this operation is soluble in water, to which it communicates a green color, due to the presence of the manganate. From this water the salt is obtained in vacuo in beautiful green crystals. On allowing the solution to stand exposed to the air, it rapidly becomes blue, violet, purple, and finally red, by the gradual conversion of the manganate into the permanganate of potash; and on account of these changes of color the black mass has received the name of mineral chameleon.
Permanganic acid is only known in solution or in a state of combination. Its solution is of a splendid red color, but appears of a dark violet tint when seen by transmitted light. It is obtained by treating a solution of permanganate of baryta with sulphuric acid, when sulphate of baryta falls, and the permanganic acid remains dissolved in the water. Permanganate of potash, which crystallizes in reddish purple prisms, is the most important of its salts. It is largely employed in analytical chemistry, and is the basis of Condy's Disinfectant Fluid.
Manganese is a constituent of many mineral waters, and is found in small quantities in the ash of most vegetables and animal substances. It is always associated with iron.
Various preparations of manganese have been employed in medicine. The sulphate of the protoxide in doses of one or two drachms produces purgative effects, and is supposed to increase the excretion of bile; and in small doses, both this salt and the carbonate have been given with the intention of improving the condition of the blood in cases of anæmia. Manganic acid and permanganate of potash are of great use when applied in lotions (as in Condy's Fluid diluted) to foul and fetid ulcers. In connection with the medicinal applications of manganese it may be mentioned that manganic acid is the agent employed in Dr. Angus Smith's celebrated test for the impurity of the air.
It is the glass maker's soap of glass manufacture, and is used to correct the green color of glass, which is owing to the presence of protoxide of iron. This it converts into the comparatively colorless peroxide.
It is also used in the Bessemer and similar processes, to decompose the oxide of iron. Spiegeleisen, an iron which contains a natural alloy of from 10 to 12 per cent. of manganese, is used for this purpose when conveniently attainable.--Glassware Reporter.
[Footnote: Abstract from a paper read before the New York Academy of Sciences.]
There exists a large mining and manufacturing industry in Austria, that of ozokerite, or earth-wax, which has nothing like it in any other part of the known world, an industry that supplies Europe with a part of its beeswax, without the aid of the bees. It may not be generally known that the mining of petroleum was a profitable industry in Austria long before it was in this country. In 1852, a druggist near Tarnow distilled the oil and had an exhibit of it in the first World's Fair in London. In America, the first borings were made in 1859. Indeed, the use of petroleum as an illuminator was common at a very early age in the world's history. In Persia at Baku, in India on the Irawada, also in the Crimea, and on the river Kuban in Russia, petroleum has been used in lamps for thousands of years. At Baku the fire worshipers have a never-ceasing flame, which has burned from time immemorial. The mines of ozokerite are located in Austrian Poland, now known as Galicia. Near the city of Drohabich, on the railway line running from Cracow to Lemberg, is a town of six thousand inhabitants, called Borislau, which is entirely supported by the ozokerite industry. It lies at the foot of the Carpathian Mountains. About the year 1862, a shaft was sunk for petroleum at that place. After descending about one hundred and eighty feet, the miners found all the cracks in the clay or rock filled with a brown substance, resembling beeswax. At first, the layers were not thicker than writing paper; but they grew thicker gradually below, until at a depth of three hundred feet they attained a thickness of three or four inches. Upon examination, it was found that a yellow wax could be made of a portion of this substance, and at once a substitute for wax was manufactured.
The discovery caused an excitement like the oil fever of 1865 in America. A large number of leases were made. When I saw the wells of Pennsylvania, in 1879, there were more than two thousand. The owner of the land received one-fourth of the product, and the miners three-fourths. In the petroleum region, the leases at first were whole farms, then they were reduced to 20, then 10, then 5, and at last to 1 acre, which is a square of 209 feet.
But in the ozokerite region of Poland, where everything is done on a small scale, when compared with like enterprises in this country, the leases were on tracts thirty-two feet square. These were so small that the surface was not large enough to contain the earth that had to be raised to sink the shaft; consequently the earth had to be transported to a distance, and, when I saw it, there was a mound sixty or seventy feet high. Its weight had become so great that it caused a sinking of the earth, and endangered the shafts to such an extent that the government ordered its removal to a distance and its deposit on ground that was not undermined. The shafts are four feet square, and the sides are supported by timbers six inches through, which leaves a shaft three feet square. The miner digs the well or shaft just as we dig our water wells, and the dirt and rock are hoisted up in a bucket by a rope and windlass. But one man can work in the shaft at a time. For many years no water was found; but, as there is a deposit of petroleum under the ozokerite, at a depth of six hundred feet from the surface, the miners were troubled with gas. This is got rid of by blowing a current of fresh air from a rotary fan through a pipe extending down the shaft as fast as the curbing of timber is put in place. The ozokerite is embedded in a very stiff blue clay for a depth of several hundred feet; below, it is interlaid with rock. [Specimens of crude and manufactured ozokerite were on exhibition, through the kindness of Dr. J. S. Newberry.]
That part of the earth's surface has more miners' shafts to the acre than any other part of the globe. As wages are very low in Poland, averaging not more than forty cents a day for men and ten cents for children, a very small quantity of ozokerite pays for the working. If thirty or forty pounds a day is obtained, it remunerates the two men and one or two children required to work each lease. When the bucket, containing the earth, rock, and wax, is dumped in the little shed covering the shaft, it is picked over by the children, who detach the wax from the clay or rock with knives. The miners use galvanized wire ropes and wooden buckets. When preparing to descend, they invariably cross themselves and utter a short prayer. The business is not free from danger, carelessness on the part of the boy supplying the fresh air, or the caving in of the unsupported roof, causing a large number of deaths. One of the government inspectors of the mines informed me that in one week there had been eight deaths from accidents.
The ozokerite is taken to a crude furnace, and put into a common cast iron kettle, and melted. This allows the dirt to sink to the bottom, and the ozokerite, freed from all other solids, is skimmed off with a ladle, poured into conical moulds, and allowed to cool, in which form it is sold to the refiners, for about six cents per pound. The quantity produced is uncertain, as the miners take care to understate it, for the reason that the government lays a tax upon all incomes, and the landowner demands his one-fourth of the quantity mined. The best authority is Leo Strippelman, who states the quantity produced in fifteen years at from 375,000,000 to 400,000,000 pounds, worth twenty-four millions of dollars. As the owners of the land get one-fourth of the sum, they received six millions. This is at the rate of four hundred thousand a year, a rather valuable crop from some two hundred acres of land.
The miners do not support the earth by timber or pillars, as they should; the result is that the whole plot of about two hundred acres is gradually sinking, and this will eventually ruin the industry in that part of the deposit. In another part of the same field, a French company has purchased forty acres, and it is mining the whole tract and hoisting through one shaft by steam power. In that shaft they have sunk to a depth of six hundred feet, and are troubled with water and petroleum. These they pump out very much the same way as in coal and other mines, worked in a scientific manner. The thickest layer of ozokerite found is about eighteen inches, and this layer or pocket was a great curiosity. When first removed at the bottom of the shaft, it was found to be so soft that it was shoveled out like putty. During the night it oozed into the space that had been emptied the day before; this continued for weeks, or until the pressure of the gas had become too weak to force it out.
I have been occupied in the petroleum region of Pennsylvania since 1860, have seen all the wonderful development of the oil wells, and was very much interested in contrasting the Austrian ozokerite and petroleum industry with the American. It is a good illustration of the difference between the lower class of Poles and Jews and the Yankee. Borislau, after twenty years' work, was unimproved, dirty, squalid, and brutal. It contained one school house, but no church nor printing office. None of its streets were paved, and, in the main road through the town, the mud came up to the hubs of the wagon wheels for over a mile of its length. In places, plank had to be set up on edge to keep the mud out of the houses, which were lower than the road. It contained numerous shops, where potato whisky was sold to men, women, and children. It depends on a dirty, muddy creek for its supply of water. Its houses were generally one-story, built of logs and mud.
On the other hand, Oil City, a town of the same age and size, contained eight school houses (one a high school building), twelve churches, and two printing offices. It has paved streets, which, in 1863, were as deep with mud as those in Borislau in 1879. It has no whisky shops where women and children can drink. Many of its houses are of brick, two, three, four, and five stories high. Its water works cost one hundred and fifty thousand dollars. All this has been done since 1860, when it did not contain forty houses.
I saw in the market place of Borislau women standing ankle deep in the mud, selling vegetables. One woman really had to build a platform of straw, on which to place a bushel of potatoes; if the straw foundation had not been there, the potatoes would have sunk out of sight. Borislau is three miles from Drohobich, a city of thirty thousand inhabitants; between the two places, in wet weather, the road was impassable. For a third of the way, it was in the bed of the creek; and I had to wait a day for the water to fall so as to navigate it in a wagon. On inquiring why they did not improve the road, I found the same difficulty as the Arkansas settler encountered with his leaky roof; when it rained he could not repair it, and when it was dry it did not need repair: so with the road to Borislau.
Ozokerite (from the Greek words, "Ozein," to smell, and "Keros," wax) is found in Turkistan, east of the Caspian Sea; in the Caucasian Mountains, in Russia; in the Carpathian Mountains, in Austria; in the Apennines, in Italy; in Texas, California, and in the Wahsatch Mountains, in the United States. Commercially, it is not worked anywhere but in Austria; although, I believe, we have in Utah a larger deposit than in any other place. I made two journeys to examine the deposits in the Wahsatch Mountains. For a distance of forty miles, it crops out in many places, and on the Minnie Maud, a stream emptying into the Colorado, I found a stratum of sand rock, from ten to twelve feet thick, filled with ozokerite.
No systematic effort has been made to ascertain the quantity of ozokerite in Utah. I saw a drift of some fourteen feet at one place, and a shaft twenty-three feet deep at another. In this shaft, the vein was about ten inches wide; and it could be traced along the slope of the hill, for several hundred feet. The largest vein of pure ozokerite is seen on Soldiers' Fork of Spanish Cañon, which enters Salt Lake Valley near the town of Provo. This vein is very much like the ozokerite of Austria, and contains between thirty and forty per cent. of white ceresin (which resembles bleached beeswax), about thirty per cent. of yellow ceresin (which resembles yellow wax), and twenty per cent. of black petroleum; the residue is dirt. Dr. J. S. Newberry, of Columbia College, and Prof. S. B. Newberry, of Cornell University, made examinations of the ozokerite found in Utah; those who are interested in the subject will find the papers published in the Engineering and Mining Journal for the year 1879.
A deposit of white ozokerite occurs on the top of the Apennine Mountains, in Italy, of which a specimen is here exhibited. An interesting story is told of its discovery. A church at Modena was robbed; among other articles taken was a quantity of wax candles. A short time afterward, a woman brought to a druggist a quantity of wax and offered it for sale. The druggist bought it and afterward suspected it consisted of the stolen candles melted down. Soon after ward she brought another lot. He had her arrested. When questioned by the magistrate, she said she found the wax in the clay on her farm, about twenty miles from the city. This story confirmed him in the belief that she had stolen the candles, or was the receiver of the stolen goods; for such a thing as a deposit of wax in the soil was unheard of. She was therefore remanded to jail. On three several days, she was brought before the court, and, when questioned, told the same story. She was a member of the church, and requested the priest to be sent for. He came, and, after an interview between them, he said it was easy to disprove her story, if it was a lie, by sending her home, in company with an officer, to investigate. The court sent the priest, who was the only one who believed her. On coming to her house, she took her pick and shovel, and going to the place at the top of the hill, she dug out of the clay a quantity of while ozokerite, proved her case, and was at once set at liberty. She performed the same service for me, and I saw her dig the specimen and heard her tell the story as I have told it to you. The hill was composed of loose clay and stones. It appeared as if it had been forced up by gas or some power from below the surface. The quantity that could be gathered, by one person, laboring constantly for a week, was only twenty-five or thirty pounds. An attempt had been made to sink a shaft; but, at a depth of fourteen feet, the pressure of the clay was sufficient to break the boards that held up the sides. The earth caved in, and the shaft was abandoned.
It is not necessary here to describe the various processes of manufacture; it will be sufficient to enumerate some of the forms of ozokerite, and the uses to which it is put. At Borislau, there are several refineries, where candles, tapers, and lubricating oils are made. In Vienna, there are five factories; in one of these, they make white wax, wax candles, matches, yellow beeswax, black heel-ball, colored tapers, and crayon pencils. In Europe, large quantities of the yellow wax are used to wax the floors of the houses, many of the finer ones being waxed every day. It is a curious fact that the Catholic Church does not allow the use of paraffine, sperm, or stearine candles; at the same time nearly all the candles used in the churches in Europe are made from ozokerite, which is a natural paraffine, made from petroleum in nature's laboratory. In the United States, the only uses made of ozokerite, so far as I know, are chewing gum and the adulteration of beeswax. In this the Yankee gives another illustration of the ruling passion strong in money making, which gives us wooden nutmegs, wooden hams, shoddy cloth, glucose candy, chiccory coffee, oleomargarine butter, mineral sperm oil made from petroleum, and beeswax made without bees.
After this paper was written, the following translation from a pamphlet, published by the First Hungarian Galician Railway Company, in 1879, came to my notice. The writer's name is not published:
"Mineral wax, in the condition in which it is taken from the shafts, is not well adapted for exportation, since it occurs with much earthy matter; and, at any rate, an expensive packing in sacks would be necessary. It is therefore first freed from all foreign substances by melting, and cooled in conical cakes of about 25 kilos. weight, and these cakes are exported. There are now, in Borislau, 25 melting works, which, in 1877, with 1 steam and 60 fire kettles, produced 95,000 metric centners (9,500,000 lb.).
"The melted earth wax is sent from Borislau to almost all European countries, to be further refined. Outside of Austro-Hungary, we may specially mention Germany, England, Italy, France, Belgium, and Russia as large purchasers of this article of commerce.
"The products of mineral wax, are:
"(a.) Ceresine, also called ozocerotine or refined ozokerite, a product which possesses a striking resemblance to ordinarily refined beeswax. It replaces this in almost all its uses, and, by its cheapness, is employed for many purposes for which beeswax is too dear. It is much used for wax candles, for waxing floors, and for dressing linen and colored papers. Wax crayons must be mentioned among these products. The house of Offenheim & Ziffer, in Elbeteinitz, makes them of many colors. These crayons are especially adapted to marking wood, stone, and iron; also, for marking linen and paper, as well as for writing and drawing. The writings and drawings made with these crayons can be effaced neither by water, by acids, nor by rubbing.
"Concerning the technical process for the production of ceresine, it should be said that, when the industry was new (the production of ceresine has been known only about eight years, since 1874), it was controlled by patents, which are kept secret. This much is known, that the color and odor are removed by fuming sulphuric acid.
"From mineral wax of good quality about 70 per cent. of white ceresine is obtained. The yellow ceresine is tinted by the addition of coloring matter (annatto).
"(b.) Paraffine, a firm, white, translucent substance, without odor. It is used, chiefly, in the manufacture of candles, and also as a protection against the action of acids, and to make casks and other wooden vessels water-tight, for coating corks, etc., for air-tight wrappings, and, finally, for the preparation of tracing paper. There are several methods of obtaining paraffine from ozokerite (see the Encyclopedic Handbook of Chemistry, by Benno Karl and F. Strohmann, vol. iv., Brunswick, 1877).
"The details of the technical process consists, in every case, in the distillation of the crude material, pressure of the distillate by hydraulic presses, melting, and treating by sulphuric acid.
"In the manufacture of paraffine from ozokerite, there are produced from 2 to 8 per cent. of benzine, from 15 to 20 per cent. of naphtha, 36 to 50 per cent. of paraffine, 15 to 20 per cent. of heavy oil for lubricating, and 10 to 20 per cent. of coke, as a residue.
"(c.) Mineral oils, which are obtained at the same time with paraffine, and are the same as those produced from crude petroleum, described above. The process consists, as in the natural rock oils, besides the distillation, in the treatment of the incidental products with acids and alkalies.
"Of the products of ozokerite, manufactured in Galicia, the greater part goes to Russia, Roumania, Turkey, Italy, and Upper Hungary. The common paraffine candles made in Galicia--which are of various sizes, from 28 to 160 per kilo--are used by the Jews in all Galicia, Bukowuina, Roumania, Upper Hungary, and Southern Russia, and form an important article of commerce. Ceresine is exported to all the ports of the world. Of late a considerable quantity is said to have been sent to the East Indies, where it is used in the printing of cotton."
The President, Dr. J. S. Newberry, stated that ozokerite was undoubtedly a product of petroleum. Little was known by the public concerning its use and value. He exhibited specimens of natural brown ozokerite, of yellow ozokerite, sold as beeswax, and of a white purified form, which had been treated by sulphuric acid. Specimens from Utah had already been shown before the Academy. There was no mystery as to its genesis in either region, as it had been shown to be the result of inspissation of a thick and viscid variety of petroleum. The term "petroleum" includes a great variety of substances, from a limpid liquid, too light to burn, to one that is thick and tarry. These differ widely also in chemical composition: some yielding much asphalt by distillation, resembling a solution of asphalt in turpentine; some containing so much paraffine that a considerable quantity can be strained out in cold weather. The asphalt in its natural form is a solid rock, to which the term "gum beds" has been applied in Canada. These differences in constitution have originated in the differences in the bituminous shales from which the petroleum, ozokerite, etc., have been derived. In Canada, as excavations are sunk through the asphalt, this becomes softer and softer, and finally passes into petroleum. This is also the case in Utah.
[Concluded from SUPPLEMENT No. 400, page 6390.]
[KANSAS CITY REVIEW.]
Professor C. S. Hastings, of the Johns Hopkins University, also includes many interesting details in his account of the trip:
The voyage from New York to Panama was pleasant with the exception of a few hot days near Aspinwall. Somewhat further south the wind changed, obliging them to call their overcoats from the bottom of their trunks to keep out the cold when crossing the equator. During a short stop in Lima the party had an opportunity of studying South American life. The products of this country are fruits and photographs of the young women. The party enjoyed both eating the former and bringing the latter home for the admiration of their friends. The expedition really began at Callao, where the party embarked on the United States man-of-war Hartford. Few circumstances contributed more to the enjoyment of the trip than the lucky chance which threw this vessel in their way. The Hartford was fitted out last August as flag ship of the South Pacific squadron. The admiral had not yet removed his flag to the vessel, but the extra accommodations provided for him and his train condoned the dignity lost by his absence. On March 22 they weighed anchor for a sail of more than four thousand miles over the blue ocean which stretches between Callao and their destination, Caroline Island. The southeast trade winds favored them, and from the first day there was actually no necessity for altering the position of a sail....
The inhabitants--five men, one woman and two children, according to the eclipse census--are natives of Tahiti. The houses are one story structures with clapboard sides, probably cut out in California and brought out in ships, to be erected on this island. The island on which they are built is about three-fourths of a mile in diameter and nearly circular in outline. The edge, which rises from five to twenty inches from the water, according to the tide's phase, goes down under the water to an even table of coral running out many feet into the sea; and is impossible to step on it with bare feet. At the end of this table the reef goes down perpendicularly, a sheer precipice, into the unfathomable sea. No vessel can anchor here, and to make a landing was an exciting matter. The island was approached in small boats on the side sheltered from the wind, and here, with the luck which characterized the trip, was found the only opening in this barrier of coral. A long cleft, perhaps eight feet wide, at the outer edge of the reef, ran in, narrowing to a mere crack near the shore. Watching a favorable chance, the boats were guided through the surf into a cleft as far as shoal water, when the men jumped on to the reef and carried baggage and instruments ashore as quickly as possible. The boats, which were new when they entered the surf, came out much the worse for wear, and the boat in which Dr. Hastings landed was stove in. Once on shore, life became a succession of wonders, rivaling the tales of Gulliver, and needing the conscientious descriptions of exact scientists to make them credible.
The members of the observing party took up their abode in the larger of the three houses, sleeping in swinging cots slung from the verandas, which afforded shade on three sides of the building. The second house was occupied by the sailors, while the third was left to the natives. These latter were sufficiently conversant with English to serve as excellent guides. Each day the party bathed in a lagoon in the center of the island. This lagoon was bordered by a beach of dazzling white coral sand, and all through its water extended reefs of living coral of the more delicate and elaborate kinds. These corals gave the lake a wonderful variety of colors, forming a picture impossible to paint or describe, and with the least ripple from a passing breeze the whole scene changed to new groups of color. The water was very clear, and in some places deep; in others so filled with coral that a boat could barely skim over the surface without scraping the keel. After crossing a long reef, one day, they entered on a sheet of water so deep that their longest line would not reach the bottom, plainly visible beneath. Fish swarmed here, and it was characteristic of them that every species, if not brilliantly colored, was marked in the most peculiar manner. One variety which frequented the shallow water, where it was heated to the degree uncomfortable to the touch, was a pure milky white, with black eyes, fins, and tail.
The French party arrived two days after the Americans. They had steamed directly from Panama with the hope of anticipating the Americans.
It rained on the morning of the eclipse, but cleared off in good time, and the definition was particularly good. Photographs occupied the time of the English and French observers. Professor Holden and Dr. Dickson searched for intra-mercurial planets; Mr. Preston took the times of contact; Dr. Hastings and Mr. Rockwell devoted their attention to spectroscopic observations of the corona. Dr. Hastings' observations have led to the production of a new theory of the corona. Briefly stated, the theory is that the light seen around the sun during a total eclipse is not due to a material substance enveloping the sun, but is a phenomenon of diffraction.
From his observation during the eclipse of 1878, made at Central City, Dr. Hastings conceived the first idea of this explanation of the solar corona. Further study served to convince him of the truth of this theory, but he had no means of proving it. Before the present eclipse, however, he devised a crucial test of his theory. This test is based on the following already known phenomena: When the moon covers the face of the sun, an envelope of light is seen all round it; the envelope is not visible when the sun is shining, on account of the sun's greater brightness; this light is called the corona; it is extremely irregular in outline. According to the drawing of Mr. J. E. Keeler at the eclipse of 1878, it enveloped the sun as a hazy glow, extending for a distance of several minutes of arc from the sun's limb and at two nearly opposite points is extended out in two long streamers feathering off into space. The opinion has been that this light was due to an atmosphere extending millions of miles from the sun. According to Dr. Hastings' view, it must be light from the sun which has undergone refraction, i.e., which has been bent from its regular course by the interposition of an opaque body like the moon.
In order to make this perfectly plain, suppose the front of a surface of waves of any sort to be striking an object which resists them. If an organ of sense is placed in the resisting object, it will judge the direction of the waves or the direction of the object producing them by a line at right angles with the wave front. Now suppose a body is placed between the body producing the waves and the sensitive organ. The waves must go around this body and will produce an eddy behind it, so that the wave front will have a different direction, and the organ of sense will conceive the origin of the waves to lie in a direction different from that before the body was interposed. Now consider the waves to be waves of light, and their origin the sun. The organ of sense is the retina of the eye. The moon is the opaque body interposed in the course of the waves, and they, being bent, make the impression on the eye that the light comes from beyond the edge of the sun. The moon covers the sun during the eclipse and a little more, so that it can move for about five minutes and still cover the sun entirely. This movement is very slight, and if the corona consists of light from a solar atmosphere, it should not change at all during this movement of the moon. But if diffraction is the cause of the light, then the slightest change in the relative positions of the sun and the moon should change the configuration of the corona, i.e., the corona should not remain exactly the same during a total eclipse. The character of the light as shown by a spectrum analysis should change.
To determine this point Dr. Hastings invented the following instrument: Two lozenge-shaped prisms of glass were fastened in the form of a letter V, and so arranged that all the light falling within the aperture of the V was lost, and that falling on the ends of the glass prisms was transmitted by a series of reflections to the apex of the V, where the prisms touched; here was placed a refracting prism, so that the light could be analyzed. This instrument was attached to the eye piece of the telescope, and the image of the eclipse reduced to such a size that the moon just fitted into the aperture of the V, while opposite sides of the corona were reflected through the prisms to the place where they came together. In this way both sides of the corona were seen through the eye-piece at the same time. On looking at the eclipse this is what Dr. Hastings saw: The light of the corona was divided into its constituents. Prominent among them was a bright green line, which is designated by the number 1,474; to this line attention was directed. Its presence in the spectrum has been an argument in favor of the view that the corona is a solar atmosphere. If this is the case, the line should remain fixed during the eclipse; but if the corona is due to diffraction, this line should change. It should grow shorter in the light from one side of the corona, and longer on the other. The observation was now reduced to watching for a change in the relative length of two green lines.
At the beginning of totality the line from the west side was much the longer, but as the eclipse progressed it shortened notably, while the line from the east side, shorter by about one-third at the beginning of the eclipse, grew longer. When the eclipse ended, the proportions of the lines were exactly reversed. There had been a change equal to two-thirds the length of the lines, while the sun and moon had only changed their relative positions by an extremely small amount. The only way in which this phenomenon can be accounted for is on the diffraction theory. The material view of the corona will not answer for it. But there are other discrepancies in the older view which have been known for some time. The principal ones are: 1. It is known from study of the sun that the gaseous pressure at the surface must be less than an inch of mercury, and is probably less than one-tenth of an inch, but an atmosphere extending to the supposed limits would cause an enormous pressure at the sun's surface, especially since the force of gravity on the sun is very much greater than on the earth. 2. The laws of gravitation would require a solar atmosphere to be distributed symmetrically around the sun, while the corona is enormously irregular in form. The sun is irregular in outline, which would make its diffracted phenomena show the observed irregularity, but it is symmetrical as regards density. 3. The most interesting discrepancy of the theory of the solar atmosphere is the fact that while it is supposed to extend for millions of miles from the sun, the recent comet passed within two hundred thousand miles of the sun, and yet its orbit was not affected in the least, as it would have been if it had plowed its way through a material substance. In taking photographs of the corona it is seen to be larger as the time of exposure is longer. This shows that the corona extends indefinitely, and it decreases in brilliancy in exact accordance with the mathematical laws of diffraction. These laws involve very complicated mathematics, but by them alone Dr. Hastings has proved that there must be diffraction where the corona is, and that it must follow the same laws as those observed. There is a small envelope around the sun, but in the opinion of Dr. Hastings it does not extend beyond what is known as the chromosphere.
The question seems to be settled, with considerable certainty, that nothing exists inside of Mercury large enough to be dignified by the name of planet. There may be, and there probably are, for the perturbations of Mercury indicate it, multitudes of small masses circulating around the sun like the planets, being fragments of comets or condensations of primitive matter, whose combined luster is seen in the zodiacal light.
The other results of the work of the Commission, so far as now known, are connected with the structure of the corona, the solar appendage which extends out for millions of miles from the sun's disk. In the photographs of the Egyptian eclipse of last summer these streamers can be traced back of each other where they cross; no better proof of their extreme tenuity could be given.
The duration of an eclipse of the sun depends on three things, the distance of the sun from the earth, the distance of the moon from the earth, and the distance of the station from the equator. All of these were favorable to a long eclipse in the case of the recent one, and the six minutes of totality gave opportunities for deliberate work not often enjoyed.
The excavations at Tell-el-Maskhutah, of which illustrations are given, have resulted in some of the most interesting and important discoveries that have ever rewarded the labors of archæologists. The idea of founding an English society for the purpose of exploring the buried cities of the Delta originated with Miss A. B. Edwards, the well-known authoress of "One Thousand Miles up the Nile," and was carried into effect mainly by her own efforts and the energy and zeal of Mr. Reginald Stuart Poole, of the British Museum, aided by the substantial support of Sir Erasmus Wilson, without whose munificent donations the work could never have been accomplished. The "Egypt Exploration Fund," thus founded and maintained, was fortunate in securing the co-operation of M. Naville, the distinguished Swiss Egyptologist, who set out for Egypt in January of this year with the object of conducting the explorations contemplated by the society. After a consultation with M. Maspero, the Director of Archæology in Egypt, who has throughout acted a friendly part toward the society's enterprise, M. Naville decided to begin his campaign by attacking the mounds at Tell-el-Maskhutah, on the Freshwater Canal, a few miles from Ismailia. The mounds of earth here were known to cover some ancient city, for some sphinxes and statues had already been found; but what city it could be, archæologists were at a loss to determine; though some, with Professor Lepsius at their head, believed it to be none other than the Rameses or "Raamses," which the Children of Israel built for Pharaoh, and whence they started on their final Exodus. Any identification, however, of the sites of the Biblical cities in Egypt was so far merely speculative. Practically nothing definite was known as to the geography of the Israelite sojourn, except that the Land of Goshen was undoubtedly in the eastern part of the Delta, and that Zoan was Tanis, whose immense mounds are to form the next subject of the society's operations. The route of the Exodus was as uncertain as everything else connected with Israel's sojourn in Egypt. What sea they crossed, and where, and by what direction they journeyed to it, remained vexed questions, although Dr. Brugsch had set up a plausible theory, in which the "Serbonian Bog" played an important part.
THE EXCAVATIONS PITHOM-SUCCOTH
Six weeks of steady digging at Tell-el-Maskhutah, under M. Naville's skillful direction, placed all these speculations in quite a new light. The city under the mounds proved to be none other than Pithom, the "store" or "treasure city" which the Children of Israel "built for Pharaoh" (Exod. i. 11). Its character as a store place or granary is seen in its construction; for the greater part of the area is covered with strongly built chambers, without doors, suitable for the storing of grain, which would be introduced through trap doors in the floor above, of which the ends of the beams are still visible. These curious chambers, unique in their appearance, are constructed of large, well made bricks, sometimes mixed with straw, sometimes without it, dried in the sun, and laid with mortar, with great regularity and precision. The walls are 10 ft. thick, and the thickness of the inclosing wall which runs round the whole city is more than 20 ft. In one corner was the temple, dedicated to the god Tum, and hence called Pe-tum or Pithom, the "Abode of Tum." Only a few statues, groups, and tablets (some of which have been presented to the British Museum) remained to testify to its name and purpose; the temple itself was finally destroyed when the Romans turned Pithom into a camp, as is shown by the position of the limestone fragments and of the Roman bricks. The statues, however, and especially a large stele, are extremely valuable, since they tell the history of the city during eighteen centuries. From a study of these monuments, M. Naville has learned that Pithom was its sacred, and Thukut (Succoth) its civil, name; that it was founded by Rameses II., restored by Shishak and others of the twenty-second dynasty; was an important place under the Ptolemies, who set up a great stele to commemorate the founding of the city of Arsinoë in the neighborhood; was called Hero or Heroöpolis by the Greeks (a name derived from the hieroglyphic ara, meaning a "store house"), and Ero Castra by the Romans, who occupied it at all events as late as A.D. 306. Indications are also found of the position of Pihahiroth, where the Israelites encamped before the passage of the "Reedy Sea," and of Clysma. All these data are directly contradictory to preconceived theories: Pithom, Succoth, Heroöpolis, Pihahiroth, and Clysma had all been hypothetically placed in totally different positions. The identification of Pithom with Succoth gives us the first absolutely certain point as yet established in the route of the Exodus, and completely overthrows Dr. Brugsch's theory. It is now certain that the Israelites passed along the valley of the Freshwater Canal and not near the Mediterranean and Lake Serbonis. The first definite geographical fact in connection with the sojourn in the Land of Egypt has been established by the excavations at Pithom. The historical identification of Rameses II. with Pharaoh the oppressor also results from the monumental evidence. One short exploration has upset a hundred theories and furnished a wonderful illustration of the historical character of the Book of Exodus. The finding of Pithom (Succoth) is, however, only the beginning, we hope, of a series of important discoveries. When enough money has been collected for the proposed exploration of Zoan (Tanis), results of the highest interest to students alike of the Bible and of Egyptian antiquities may, with certainty, be predicted.
The uppermost view shows a portion of the diggings; a workman is bringing up a barrow-load of soil from one of the deep store chambers which the Children of Israel built more than three thousand years ago. In the foreground lie the fragments of a fallen granite statue, the head and face of which are intact. The other illustration is taken from the temple end of the excavations. The sculptured group of Rameses the Great seated between divinities is one of a pair that adorned the entrance; its companion and the sphinxes that guarded the pylon are at Ismailia. Beyond this group, and a little to the left, is seen the great Stele of Pithom, set up by Ptolemy Philadelphus and Arsinoë, and containing a mass of important information in its long hieroglyphic inscriptions. Behind this, and on either side, the massive brick walls of the store chambers and the inclosing wall of the temple can be traced; while on the right hand, in the middle distance, is a heap of limestone blocks, already collected by Rameses II. for the completion or enlargement of the temple. The excavations were photographed for M. Naville, by Herr Emil Brugsch, of the Boulak Museum, and our illustrations are taken from these photographs, supplemented by sketches.--S.L.P., in Illustrated London News.
The surprises of archæology are magnificent and apparently inexhaustible. It is continually bringing forth things new and old, and often it happens that the newest are the oldest of all. Whether this or the exact converse is the case in regard to the latest discovery of Biblical archæology is a question not to be determined offhand; but the interest and importance of the question can hardly be overrated. There are now deposited in the British Museum fifteen leather slips, on the forty folds of which are written portions of the Book of Deuteronomy in a recension entirely different from that of the received text. The character employed in the manuscript is similar to that of the famous Moabite stone and of the Siloam inscription, and, therefore, the mere palæographical indication should give the probable date of the slips as the ninth century B. C., or sixteen centuries earlier than any other clearly authenticated manuscript of any portion of the Old Testament. The sheepskin slips are literally black with age, and are impregnated with a faint odor as of funeral spices; the folds are from 6 to 7 inches long and about 3½ inches wide, containing each about ten lines, written only on one side.
So far as they have yet been deciphered, they exhibit two distinct handwritings, though the same archaic character is used throughout. In some cases the same passages of Deuteronomy occur in duplicate on distinct slips, as though the fragments belonged to two contemporary transcriptions made by different scribes from the same original text. At first sight no writing whatever is perceptible; the surface seems to be covered with an oily or glutinous substance, which so completely obscures the writing beneath that a photograph of some of the slips--which we have had an opportunity of examining side by side with the slips themselves--exhibits no trace of the text. But when the leather is moistened with spirits of wine the letters become momentarily visible beneath the glossy surface.
These extraordinary fragments were brought to England by Mr. Shapira, of Jerusalem, a well known bookseller and dealer in antiquities. Mr. Shapira's name will be remembered in connection with certain archæological problems which have been solved by some scholars in a manner not altogether creditable to his sagacity.
The Moabite pottery which reached Europe through Mr. Shapira's agency and is deposited in the Museum at Berlin is now commonly regarded as a modern forgery; but of this forgery, if it be one, it is asserted that Mr. Shapira was the dupe and not the accomplice. The leathern fragments now produced by Mr. Shapira were, as he alleges, obtained by him from certain Arabs near Dibon, the neighborhood where the Moabite stone was discovered. The agent employed by him in their purchase was an Arab "who would steal his mother-in-law for a few piastres," and who would probably be even less scrupulous about a few blackened slips of ancient or modern sheepskin. The value placed by Mr. Shapira on the fragments is, however, a cool million sterling, and at this price they are offered to the British Museum, where they have been temporarily deposited for examination.
Dr. Ginsburg, the well-known Semitic scholar--whose receipt of a grant of £500 from the Prime Minister toward the production of his important work on the "Massorah" we announced with much satisfaction yesterday--is now busily engaged in deciphering the contents of the fragments and examining their genuineness. On this latter question we refrain from pronouncing an opinion. When Dr. Ginsburg's report appears, we shall be able to judge whether these extraordinary fragments are really 2,500 years old, or have been compiled within the last few years.
No complete account of the contents of the fragments can yet be given. To decipher them is a work of time and of infinite patience and skill, as will readily be inferred from the account we have given above of the appearance and condition of the slips. But enough has been deciphered to show that the text employed in them exhibits discrepancies of the most remarkable and important character as compared with that of the received version of the Mosaic books.
In the first verse of the ninth chapter of Deuteronomy, where the received version reads, "Thou art to pass over Jordan this day, to go in to possess nations greater and mightier than thyself," the corresponding passage of the fragments substitutes the plural for the singular, "Ye" for '"Thou," while for "g'dôlîm," the word translated "greater," it reads "rabbîm." But a far more complete idea of the variations of text and signification may be obtained from a comparison of the text of the Decalogue as it appears in the received version in the sixth chapter of Deuteronomy with that contained in the fragments so far as they have yet been deciphered. The version of the fragments, literally rendered, runs as follows:
"I am God, thy God, which liberated thee from the land of Egypt, from the house of bondage. Ye shall have no other gods. Ye shall not make to yourselves any graven image, nor any likeness that is in heaven above or that is in the earth beneath, or that is in the waters under the earth. Ye shall not bow down to them nor serve them. I am God, your God. Sanctify ... in six days I have made the heaven and the earth, and all that is therein, and rested on the seventh day, therefore rest thou also, thou and thy cattle and all that thou hast: I am God, thy God. Honor thy father and thy mother ...: I am God, thy God. Thou shall not kill the person of thy brother: I am God, thy God. Thou shalt not commit adultery with the wife of thy neighbor: I am God, thy God. Thou shalt not steal the property of thy brother: I am God, thy God. Thou shalt not swear by my name falsely, for I visit the iniquity of the fathers upon the children unto the third and fourth generation of those who take my name in vain: I am God, thy God. Thou shalt not bear false witness against thy brother: I am God, thy God. Thou shalt not covet the wife ... or his manservant, or his maidservant, or anything that is his: I am God, thy God. Thou shalt not hate thy brother in thy heart: I am God, thy God. These ten words (or commandments) God spake."
Several points may be noted in this version. The singular refrain "I am God, thy God"--which does not appear at all in the received version--occurs ten times, being, as it were, a solemn ratification of the Divine sanction given at the end of each separate precept. If this be so, the first two commandments, as they are commonly reckoned, are here fused into one, and the tenth place is taken by a commandment which does not appear in the received version of the Decalogue.
It will further be observed that the distinctive Jewish name for the Almighty, "Jehovah," or "the Lord," does not appear at all, the familiar phrase of the received version, "the Lord thy God," being replaced throughout by "God, thy God."
On the many variations in arrangement and detail we need not dwell; they speak for themselves. But we have quoted enough to show that these fragments present problems of the utmost importance and interest both to criticism and exegesis, unless, indeed, they are to be regarded as the ingenious fabrications of some Oriental Ireland, who, knowing the interest felt by scholars in variations of the Sacred Text, has set himself, with infinite pains and skill, to forestall a growing demand. Until this preliminary question is resolved to the satisfaction of all competent scholars, no further questions need be raised. In any case the primá facie presumption must be held to be enormously against the genuineness of the fragments. Such a presumption rests on the improbability of finding manuscripts older by at least sixteen centuries than any extant manuscripts of the same text, on the comparative ease with which such fragments can be forged, and on the powerful motives to such forgery attested by the price placed by Mr. Shapira on his property.
All that we know of the provenance of the fragments is that Mr. Shapira obtained them from an Arab of doubtful character; and that Arabs of doubtful character have driven a splendid trade in Moabite antiquities ever since the discovery of the Moabite stone. On the other hand, the forger, if forgery there be, is assuredly no clumsy and ignorant bungler, as the makers of the Moabite pottery were confidently alleged to be by those who disputed its genuineness. It is, of course, part of his craft, and not, perhaps, much more than the 'prentice part, to give to the sheepskins on which the text is inscribed an appearance of immemorial antiquity. But a good deal more than the skill required to make a new sheepskin look like an old one has gone to the production of Mr. Shapira's fragments. If they are forged, the fabricator must have known what scholars would be likely to expect in genuine fragments, and have set himself to fulfill their expectations. In these days of scientific palæography and minute textual scholarship no forger of ancient manuscripts could hope to take in scholars unless he were a scholar himself. Variations of text would be looked for as a matter of course; palæographical accuracy would be exacted to the minutest turn of a letter. Now, to vary a text so as to furnish a different recension without betraying ignorance or solecism requires scholarship of no mean order, while it is very far from an easy thing to write currently in an archaic and unfamiliar character in such a manner as to deceive experts in palæography. But the fabricator of these fragments, if fabricated they are, has attempted and accomplished a good deal more than this. He has in some cases produced two identical texts written in different hands, both preserving unimpaired the archaic character of the letters. This implies either the employment of two scribes or else an almost incredible skill in the single scribe employed, and in either case it doubles the probability of detection. If, moreover, the supposed fabricator is also himself the scribe, it is evident that he is not only a very ingenious artist, but also a very accomplished scholar, and one can only regret that he has engaged in an industry which has placed him at the mercy of an Arab who would steal his mother-in-law for a few piastres, and is likely, therefore, to enrich no one but Mr. Shapira. We should expect to find, however, that his extraordinary ingenuity has at some point or another overreached itself. Familiar as he must be with the labors of modern Biblical critics--for otherwise he would hardly have ventured to impose upon them--it would be strange if he were not betrayed into some more or less suspicious coincidences with them. In any case, the problem presented by the fragments is one of profound interest, and the whole world of letters will resound with the controversy they are certain to excite.--London Times.
SPECIMENS OF OLD KNOCKING DEVICES FOR DOORS.--From the Building News.
Since the failure last August of the Cape Commercial Bank there has been much depression in South Africa. Ostrich farming, in common with other enterprises, has suffered. Before the crisis a pair of breeding ostriches have been sold for 350 l., now they would not realize 50 l.
The resolution of the Government of South Australia to encourage ostrich breeding came in very opportunely for the Cape dealers, and one or two cargoes of birds have been shipped for Adelaide. The climate of the two colonies is very similar, and the locality selected for the imported birds (the Musgrave Ranges) resembles in dryness and temperature their native habitat.
The first sketch opposite represents the ostriches bidding farewell to their South African home. "The dear old farm where we were reared, good-by!"
One of the boxes, while being slung from the cart to the hold, got into a slanting position. This frightened one of the two inmates, a fine cock. He kicked so hard that he burst open the door of his cage, which was, of course, instantly lowered on deck. Fortunately there was there a gentleman who understood how to handle ostriches. He instantly seized him before he could do himself or the bystanders any injury, and after a brief struggle prevailed on him to re-enter his box. When released in the hold he became quite quiet, and ate his first meal on board ship with a relish.
After being taken out of their boxes the birds are allowed to take a little exercise just to make themselves at home, and are then arranged in wooden kraals, of which there are two hundred on board the vessel. The ostriches are induced to move from one place to another by catching hold of their bodies, and using a little gentle force.
The last sketch represents their first meal on board after a fast of thirty hours. Apple melons were chopped up for them by their "steward," who was to accompany them to Australia. It was curious to see a bird swallow a great lump and then to watch the lump working slowly down the animal's long neck. On the voyage they would be fed with maize or mealies, onions, apple melons, and barley. They require very little water; however, there were five large iron tanks on board in case they would feel thirsty. Our engravings are from sketches by Mr. Dennis Edwards, of Hoff Street, Capetown,
SHIPPING OSTRICHES FROM CAPE TOWN TO AUSTRALIA.
1. Ostriches on the South African Farm Where They Were
Reared.--2.
Attempted Escape and Recapture of an Ostrich on Board Ship.--3.
Lowering
the Birds Into the Hold.--4.A Queer Dinner Party--Ostriches Eating
Apple Melons.
An ordinary weathercock provided with datum points may, in the majority of cases, suffice for the observation of the wind during the day; but recourse has to be had to different means to obtain an automatic transmission of the indications of the vane to the inside of a building. The different systems employed for such a purpose consist of gearings, or are accompanied by a friction that notably diminishes the sensitiveness of the apparatus, especially when the rod has to traverse several stories. Mr. Emile Richard, inspector of the Versailles waterworks, has just devised an ingenious system which, while considerably reducing the weight of the movable part, allows the weathercock to preserve all its sensitiveness. This apparatus consists of two principal parts--one fixed and the other movable. The stationary part is designated in the accompanying figure by the letters A and B and by cross-hatchings. This forms the rod or support. An iron tube, T, with clamps, P, at its lower extremity forms the base of the apparatus, and is hidden, after the mounting of the apparatus, by the ornamental zinc covering, Z. The upper part of the tube carries a shoulder-piece, upon which rests a bronze platform, E, and which is slightly inclined outwardly to prevent the accumulation of water on it. Over the platform there move three crystal balls, which are held and guided by a horizontal disk movable around the stationary tube.
The movable portion, designed to receive the action of the wind and to indicate its direction, is designated by the letters C D and coarse lines. It consists of (1) a zinc tube, K, provided at intervals with copper rings, and entering the rod, A B, which serves as a guide for it; (2) of a bronze disk covered by an external ornament, O, fixed to the tube and resting on the balls; (3) of the vane, G, properly so called; and (4) of the cap, C, provided with bayonet catch, crowning the tube and covering the point of attachment of the wire of transmission. This latter consists of a simple brass or galvanized iron wire, f f, perfectly taut, and made fast in the top of the tube. After traversing as many stories as necessary this wire terminates, in the interior of the room where the observations are made, in a copper rod to which is fastened a horizontal arrow, F. The wire traverses the floorings through small zinc tubes; and, in the rooms through which it passes, it is protected by iron tubes. To the ceiling of the observing room there is affixed a wind-rose, R, on which the arrow reproduces all the motions of the vane.
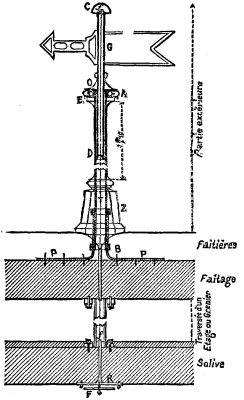
RICHARD'S WEATHERCOCK.
This apparatus is now in operation in the different stations that the Versailles waterworks has established near the reservoirs of the plateau of Trappes, and it is also installed in several primary normal schools, where it is giving very good results.--La Nature.
A correspondent of the Ohio Farmer reports an experiment in curing clover, showing how he just missed breeding fire in his barn, and illustrating the importance of ventilating hay mows:
In 1861 I used a horse fork for the first time. The haying season was not a bright one, and our clover was drawn a little greener than usual, and went into the mow in large and compact forkfuls. The result was intense heating, and consequently very rapid evaporation and sweating of the mow. On a bay holding ordinarily twenty tons we put at least thirty tons, as every load at the top seemed to make room for another. The barn was rather open, which allowed quite free evaporation on all sides as well as at the top. The result was that I had very bright and excellent hay at the bottom, top, and sides of that mow, but severals tons in the center were as completely charred as though burned in a coal pit. What prevented combustion has always been a mystery to me. Since that escape from a conflagration, I have not deemed it prudent to put clover in so green as to cause intense heating, or to fill a mow too rapidly. If we haul six loads per day to one mow, weighing thirty hundred each, which will shrink during the sweating process to one ton each, we have three tons of water to be thrown off by evaporation.
If we continue to put on six loads per day until the mow is full, the principal part of that moisture must rise through the entire mass. To relieve the hay of moisture, I deem it best to have several places of storage, and change daily or semi-daily from one to the other, thus giving time for a share of the moisture to pass off. To facilitate this evaporation and prevent the hay from reabsorbing it and becoming musty, the best of ventilation is necessary. Ventilation above a clover mow is as necessary as it is above a sugar or fruit evaporator. If there is not open space and draught sufficient to carry away the moisture, it is returned to the mow, and mould is the inevitable result. No ordinary amount of drying will prevent hay from becoming musty if ventilation is shut off during the sweating process. If a hole is cut through the floor at the bottom of the mow near the center and under a ventilator in the roof and a barrel placed over it and drawn up as the hay is mowed in, thus leaving a hole from bottom to top, evaporation will be facilitated and the quality of the hay improved. Salt thrown on, as the clover is put in, to the amount of two or three quarts to the ton, will make it a relish with stock.
This beautiful Agave is now in blossom in the garden here, and I am happy to be able to send you photographs of it. This is the first time it has ever blossomed in cultivation, and it has never been seen in flower in a wild state. It is a mature native-grown specimen, dense in habit, and perfectly semi-spherical in form, and the leaves are arranged in spiral fashion with as much regularity as those of a screw pine. The circumference of the plant is 5 ft. 1 in., and it has 268 leaves. Its flower-stem appeared about the middle of June, grew rather fast till it was 7 ft. high, then rather slowly till it reached its full development. The scape is now 10 ft. 4 in. high above the plant, 6½ in. in circumference at the base, or 5¼ in. at a foot above the base; from there it tapers very gradually till near the apex. The flower-spike is exceedingly dense, and 5 ft. 8 in. long; the lower or naked portion, 4 ft. 8 in. long, is prominently marked by abortive flower buds, with, near the base, some bristle-like scales 3½ in. to 4 in. long. The flowers are regularly arranged in parcels of three, all the three being equal in size and opening together; they are greenish white in color, 1½ in. long, or, including the stamens, some 2¾ in. to 3 in. long.

AGAVE VICTORIÆ-REGINÆ.
The first flowers opened on August 3, and they have continued to open in succession, a belt about 3 in. wide opening each day. They remain in good condition for two days; on the third day the stamens wilt and drop down, but the pistil remains erect till the fourth day. On the first day of opening the pistil is not so long as the stamens by ¾ in.; on the second it has grown to be as long as the stamens, but it is not in condition to receive the pollen till after noon of the second day. Although the flowers on some eighteen inches of the spike have already blossomed, none of the ovaries have been fertilized; they are dropping off, but I am rather sanguine regarding those about the middle of the spike. So great is the superfluity of nectar contained in the flowers, that on the afternoon of the second day it often drops from the cups, and the least shake to the scape brings it down in a shower. The main beauty of the inflorescence consists in the dense bottle-brush-like mass of bright yellow anthers. This plant, together with several smaller ones, was contributed to this garden by Dr. Edward Palmer, who collected them in their native wilds--the mountains of Northern Mexico--some three years ago. He found them growing in a limited and rather inaccessible locality in gravelly and rocky soil some miles from Monterey. In addition to those he sent here he also sent a quantity to the garden of the Agricultural Department at Washington, and some to Dr. Engelmann, the eminent botanist at St. Louis. To Dr. Engelmann he also sent a piece of an old flower stem and some dried capsules which he found upon an old plant, and it was from these specimens in 1880 that the doctor was enabled to describe for the first time the inflorescence of this Agave.--The Garden.
In the course of an investigation in which we are at present engaged we have arrived at some results which appear to us to be very interesting. We find that the generally received view that the fats are ethers of glycerin is partially correct, and that instances of a different kind of structure occur among the natural oils and fats.
Ethers of iso-glycerin, or of homologues of iso-glycerin, appear to occur. Iso-glycerin has this structure:
C(OH)2
CH
CH3
It exists in its ethers, but cannot be isolated, and should be resolved into:
COOH + H2O
CH2
CH3
Ethers of iso-glycerin, or ethers of homologues of iso-glycerin, yield no glycerin when saponified.--Chemical News.
A catalogue, containing brief notices of many important scientific papers heretofore published in the SUPPLEMENT, may be had gratis at this office.
Terms of Subscription, $5 a Year.
Sent by mail, postage prepaid, to subscribers in any part of the United States or Canada. Six dollars a year, sent, prepaid, to any foreign country.
All the back numbers of THE SUPPLEMENT, from the commencement, January 1, 1876, can be had. Price, 10 cents each.
All the back volumes of THE SUPPLEMENT can likewise be supplied. Two volumes are issued yearly. Price of each volume, $2.50, stitched in paper, or $3.50, bound in stiff covers.
COMBINED RATES--One copy of SCIENTIFIC AMERICAN and one copy of SCIENTIFIC AMERICAN SUPPLEMENT, one year, postpaid, $7.00.
A liberal discount to booksellers, news agents, and canvassers.
MUNN & CO., Publishers,
261 Broadway, New York, N. Y.
In connection with the Scientific American, Messrs. MUNN & Co. are Solicitors of American and Foreign Patents, have had 38 years' experience, and now have the largest establishment in the world. Patents are obtained on the best terms.
A special notice is made in the Scientific American of all Inventions patented through this Agency, with the name and residence of the Patentee. By the immense circulation thus given, public attention is directed to the merits of the new patent, and sales or introduction often easily effected.
Any person who has made a new discovery or invention can ascertain, free of charge, whether a patent can probably be obtained, by writing to MUNN & Co.
We also send free our Hand Book about the Patent Laws, Patents, Caveats. Trade Marks, their costs, and how procured, with hints for procuring advances on inventions. Address
MUNN & CO., 261 Broadway, New York.
Branch Office, cor. F and 7th Sts., Washington, D. C.
End of the Project Gutenberg EBook of Scientific American Supplement, No.
401, September 8, 1883, by Various
*** END OF THIS PROJECT GUTENBERG EBOOK SCIENTIFIC AMERICAN SUPPL., NO. 401 ***
***** This file should be named 8718-h.htm or 8718-h.zip *****
This and all associated files of various formats will be found in:
http://www.gutenberg.org/8/7/1/8718/
Produced by Olaf Voss, Don Kretz, Juliet Sutherland, Charles
Franks and the Distributed Proofreaders Team
Updated editions will replace the previous one--the old editions
will be renamed.
Creating the works from public domain print editions means that no
one owns a United States copyright in these works, so the Foundation
(and you!) can copy and distribute it in the United States without
permission and without paying copyright royalties. Special rules,
set forth in the General Terms of Use part of this license, apply to
copying and distributing Project Gutenberg-tm electronic works to
protect the PROJECT GUTENBERG-tm concept and trademark. Project
Gutenberg is a registered trademark, and may not be used if you
charge for the eBooks, unless you receive specific permission. If you
do not charge anything for copies of this eBook, complying with the
rules is very easy. You may use this eBook for nearly any purpose
such as creation of derivative works, reports, performances and
research. They may be modified and printed and given away--you may do
practically ANYTHING with public domain eBooks. Redistribution is
subject to the trademark license, especially commercial
redistribution.
*** START: FULL LICENSE ***
THE FULL PROJECT GUTENBERG LICENSE
PLEASE READ THIS BEFORE YOU DISTRIBUTE OR USE THIS WORK
To protect the Project Gutenberg-tm mission of promoting the free
distribution of electronic works, by using or distributing this work
(or any other work associated in any way with the phrase "Project
Gutenberg"), you agree to comply with all the terms of the Full Project
Gutenberg-tm License available with this file or online at
www.gutenberg.org/license.
Section 1. General Terms of Use and Redistributing Project Gutenberg-tm
electronic works
1.A. By reading or using any part of this Project Gutenberg-tm
electronic work, you indicate that you have read, understand, agree to
and accept all the terms of this license and intellectual property
(trademark/copyright) agreement. If you do not agree to abide by all
the terms of this agreement, you must cease using and return or destroy
all copies of Project Gutenberg-tm electronic works in your possession.
If you paid a fee for obtaining a copy of or access to a Project
Gutenberg-tm electronic work and you do not agree to be bound by the
terms of this agreement, you may obtain a refund from the person or
entity to whom you paid the fee as set forth in paragraph 1.E.8.
1.B. "Project Gutenberg" is a registered trademark. It may only be
used on or associated in any way with an electronic work by people who
agree to be bound by the terms of this agreement. There are a few
things that you can do with most Project Gutenberg-tm electronic works
even without complying with the full terms of this agreement. See
paragraph 1.C below. There are a lot of things you can do with Project
Gutenberg-tm electronic works if you follow the terms of this agreement
and help preserve free future access to Project Gutenberg-tm electronic
works. See paragraph 1.E below.
1.C. The Project Gutenberg Literary Archive Foundation ("the Foundation"
or PGLAF), owns a compilation copyright in the collection of Project
Gutenberg-tm electronic works. Nearly all the individual works in the
collection are in the public domain in the United States. If an
individual work is in the public domain in the United States and you are
located in the United States, we do not claim a right to prevent you from
copying, distributing, performing, displaying or creating derivative
works based on the work as long as all references to Project Gutenberg
are removed. Of course, we hope that you will support the Project
Gutenberg-tm mission of promoting free access to electronic works by
freely sharing Project Gutenberg-tm works in compliance with the terms of
this agreement for keeping the Project Gutenberg-tm name associated with
the work. You can easily comply with the terms of this agreement by
keeping this work in the same format with its attached full Project
Gutenberg-tm License when you share it without charge with others.
1.D. The copyright laws of the place where you are located also govern
what you can do with this work. Copyright laws in most countries are in
a constant state of change. If you are outside the United States, check
the laws of your country in addition to the terms of this agreement
before downloading, copying, displaying, performing, distributing or
creating derivative works based on this work or any other Project
Gutenberg-tm work. The Foundation makes no representations concerning
the copyright status of any work in any country outside the United
States.
1.E. Unless you have removed all references to Project Gutenberg:
1.E.1. The following sentence, with active links to, or other immediate
access to, the full Project Gutenberg-tm License must appear prominently
whenever any copy of a Project Gutenberg-tm work (any work on which the
phrase "Project Gutenberg" appears, or with which the phrase "Project
Gutenberg" is associated) is accessed, displayed, performed, viewed,
copied or distributed:
This eBook is for the use of anyone anywhere at no cost and with
almost no restrictions whatsoever. You may copy it, give it away or
re-use it under the terms of the Project Gutenberg License included
with this eBook or online at www.gutenberg.org
1.E.2. If an individual Project Gutenberg-tm electronic work is derived
from the public domain (does not contain a notice indicating that it is
posted with permission of the copyright holder), the work can be copied
and distributed to anyone in the United States without paying any fees
or charges. If you are redistributing or providing access to a work
with the phrase "Project Gutenberg" associated with or appearing on the
work, you must comply either with the requirements of paragraphs 1.E.1
through 1.E.7 or obtain permission for the use of the work and the
Project Gutenberg-tm trademark as set forth in paragraphs 1.E.8 or
1.E.9.
1.E.3. If an individual Project Gutenberg-tm electronic work is posted
with the permission of the copyright holder, your use and distribution
must comply with both paragraphs 1.E.1 through 1.E.7 and any additional
terms imposed by the copyright holder. Additional terms will be linked
to the Project Gutenberg-tm License for all works posted with the
permission of the copyright holder found at the beginning of this work.
1.E.4. Do not unlink or detach or remove the full Project Gutenberg-tm
License terms from this work, or any files containing a part of this
work or any other work associated with Project Gutenberg-tm.
1.E.5. Do not copy, display, perform, distribute or redistribute this
electronic work, or any part of this electronic work, without
prominently displaying the sentence set forth in paragraph 1.E.1 with
active links or immediate access to the full terms of the Project
Gutenberg-tm License.
1.E.6. You may convert to and distribute this work in any binary,
compressed, marked up, nonproprietary or proprietary form, including any
word processing or hypertext form. However, if you provide access to or
distribute copies of a Project Gutenberg-tm work in a format other than
"Plain Vanilla ASCII" or other format used in the official version
posted on the official Project Gutenberg-tm web site (www.gutenberg.org),
you must, at no additional cost, fee or expense to the user, provide a
copy, a means of exporting a copy, or a means of obtaining a copy upon
request, of the work in its original "Plain Vanilla ASCII" or other
form. Any alternate format must include the full Project Gutenberg-tm
License as specified in paragraph 1.E.1.
1.E.7. Do not charge a fee for access to, viewing, displaying,
performing, copying or distributing any Project Gutenberg-tm works
unless you comply with paragraph 1.E.8 or 1.E.9.
1.E.8. You may charge a reasonable fee for copies of or providing
access to or distributing Project Gutenberg-tm electronic works provided
that
- You pay a royalty fee of 20% of the gross profits you derive from
the use of Project Gutenberg-tm works calculated using the method
you already use to calculate your applicable taxes. The fee is
owed to the owner of the Project Gutenberg-tm trademark, but he
has agreed to donate royalties under this paragraph to the
Project Gutenberg Literary Archive Foundation. Royalty payments
must be paid within 60 days following each date on which you
prepare (or are legally required to prepare) your periodic tax
returns. Royalty payments should be clearly marked as such and
sent to the Project Gutenberg Literary Archive Foundation at the
address specified in Section 4, "Information about donations to
the Project Gutenberg Literary Archive Foundation."
- You provide a full refund of any money paid by a user who notifies
you in writing (or by e-mail) within 30 days of receipt that s/he
does not agree to the terms of the full Project Gutenberg-tm
License. You must require such a user to return or
destroy all copies of the works possessed in a physical medium
and discontinue all use of and all access to other copies of
Project Gutenberg-tm works.
- You provide, in accordance with paragraph 1.F.3, a full refund of any
money paid for a work or a replacement copy, if a defect in the
electronic work is discovered and reported to you within 90 days
of receipt of the work.
- You comply with all other terms of this agreement for free
distribution of Project Gutenberg-tm works.
1.E.9. If you wish to charge a fee or distribute a Project Gutenberg-tm
electronic work or group of works on different terms than are set
forth in this agreement, you must obtain permission in writing from
both the Project Gutenberg Literary Archive Foundation and Michael
Hart, the owner of the Project Gutenberg-tm trademark. Contact the
Foundation as set forth in Section 3 below.
1.F.
1.F.1. Project Gutenberg volunteers and employees expend considerable
effort to identify, do copyright research on, transcribe and proofread
public domain works in creating the Project Gutenberg-tm
collection. Despite these efforts, Project Gutenberg-tm electronic
works, and the medium on which they may be stored, may contain
"Defects," such as, but not limited to, incomplete, inaccurate or
corrupt data, transcription errors, a copyright or other intellectual
property infringement, a defective or damaged disk or other medium, a
computer virus, or computer codes that damage or cannot be read by
your equipment.
1.F.2. LIMITED WARRANTY, DISCLAIMER OF DAMAGES - Except for the "Right
of Replacement or Refund" described in paragraph 1.F.3, the Project
Gutenberg Literary Archive Foundation, the owner of the Project
Gutenberg-tm trademark, and any other party distributing a Project
Gutenberg-tm electronic work under this agreement, disclaim all
liability to you for damages, costs and expenses, including legal
fees. YOU AGREE THAT YOU HAVE NO REMEDIES FOR NEGLIGENCE, STRICT
LIABILITY, BREACH OF WARRANTY OR BREACH OF CONTRACT EXCEPT THOSE
PROVIDED IN PARAGRAPH 1.F.3. YOU AGREE THAT THE FOUNDATION, THE
TRADEMARK OWNER, AND ANY DISTRIBUTOR UNDER THIS AGREEMENT WILL NOT BE
LIABLE TO YOU FOR ACTUAL, DIRECT, INDIRECT, CONSEQUENTIAL, PUNITIVE OR
INCIDENTAL DAMAGES EVEN IF YOU GIVE NOTICE OF THE POSSIBILITY OF SUCH
DAMAGE.
1.F.3. LIMITED RIGHT OF REPLACEMENT OR REFUND - If you discover a
defect in this electronic work within 90 days of receiving it, you can
receive a refund of the money (if any) you paid for it by sending a
written explanation to the person you received the work from. If you
received the work on a physical medium, you must return the medium with
your written explanation. The person or entity that provided you with
the defective work may elect to provide a replacement copy in lieu of a
refund. If you received the work electronically, the person or entity
providing it to you may choose to give you a second opportunity to
receive the work electronically in lieu of a refund. If the second copy
is also defective, you may demand a refund in writing without further
opportunities to fix the problem.
1.F.4. Except for the limited right of replacement or refund set forth
in paragraph 1.F.3, this work is provided to you 'AS-IS', WITH NO OTHER
WARRANTIES OF ANY KIND, EXPRESS OR IMPLIED, INCLUDING BUT NOT LIMITED TO
WARRANTIES OF MERCHANTABILITY OR FITNESS FOR ANY PURPOSE.
1.F.5. Some states do not allow disclaimers of certain implied
warranties or the exclusion or limitation of certain types of damages.
If any disclaimer or limitation set forth in this agreement violates the
law of the state applicable to this agreement, the agreement shall be
interpreted to make the maximum disclaimer or limitation permitted by
the applicable state law. The invalidity or unenforceability of any
provision of this agreement shall not void the remaining provisions.
1.F.6. INDEMNITY - You agree to indemnify and hold the Foundation, the
trademark owner, any agent or employee of the Foundation, anyone
providing copies of Project Gutenberg-tm electronic works in accordance
with this agreement, and any volunteers associated with the production,
promotion and distribution of Project Gutenberg-tm electronic works,
harmless from all liability, costs and expenses, including legal fees,
that arise directly or indirectly from any of the following which you do
or cause to occur: (a) distribution of this or any Project Gutenberg-tm
work, (b) alteration, modification, or additions or deletions to any
Project Gutenberg-tm work, and (c) any Defect you cause.
Section 2. Information about the Mission of Project Gutenberg-tm
Project Gutenberg-tm is synonymous with the free distribution of
electronic works in formats readable by the widest variety of computers
including obsolete, old, middle-aged and new computers. It exists
because of the efforts of hundreds of volunteers and donations from
people in all walks of life.
Volunteers and financial support to provide volunteers with the
assistance they need are critical to reaching Project Gutenberg-tm's
goals and ensuring that the Project Gutenberg-tm collection will
remain freely available for generations to come. In 2001, the Project
Gutenberg Literary Archive Foundation was created to provide a secure
and permanent future for Project Gutenberg-tm and future generations.
To learn more about the Project Gutenberg Literary Archive Foundation
and how your efforts and donations can help, see Sections 3 and 4
and the Foundation information page at www.gutenberg.org
Section 3. Information about the Project Gutenberg Literary Archive
Foundation
The Project Gutenberg Literary Archive Foundation is a non profit
501(c)(3) educational corporation organized under the laws of the
state of Mississippi and granted tax exempt status by the Internal
Revenue Service. The Foundation's EIN or federal tax identification
number is 64-6221541. Contributions to the Project Gutenberg
Literary Archive Foundation are tax deductible to the full extent
permitted by U.S. federal laws and your state's laws.
The Foundation's principal office is located at 4557 Melan Dr. S.
Fairbanks, AK, 99712., but its volunteers and employees are scattered
throughout numerous locations. Its business office is located at 809
North 1500 West, Salt Lake City, UT 84116, (801) 596-1887. Email
contact links and up to date contact information can be found at the
Foundation's web site and official page at www.gutenberg.org/contact
For additional contact information:
Dr. Gregory B. Newby
Chief Executive and Director
gbnewby@pglaf.org
Section 4. Information about Donations to the Project Gutenberg
Literary Archive Foundation
Project Gutenberg-tm depends upon and cannot survive without wide
spread public support and donations to carry out its mission of
increasing the number of public domain and licensed works that can be
freely distributed in machine readable form accessible by the widest
array of equipment including outdated equipment. Many small donations
($1 to $5,000) are particularly important to maintaining tax exempt
status with the IRS.
The Foundation is committed to complying with the laws regulating
charities and charitable donations in all 50 states of the United
States. Compliance requirements are not uniform and it takes a
considerable effort, much paperwork and many fees to meet and keep up
with these requirements. We do not solicit donations in locations
where we have not received written confirmation of compliance. To
SEND DONATIONS or determine the status of compliance for any
particular state visit www.gutenberg.org/donate
While we cannot and do not solicit contributions from states where we
have not met the solicitation requirements, we know of no prohibition
against accepting unsolicited donations from donors in such states who
approach us with offers to donate.
International donations are gratefully accepted, but we cannot make
any statements concerning tax treatment of donations received from
outside the United States. U.S. laws alone swamp our small staff.
Please check the Project Gutenberg Web pages for current donation
methods and addresses. Donations are accepted in a number of other
ways including checks, online payments and credit card donations.
To donate, please visit: www.gutenberg.org/donate
Section 5. General Information About Project Gutenberg-tm electronic
works.
Professor Michael S. Hart was the originator of the Project Gutenberg-tm
concept of a library of electronic works that could be freely shared
with anyone. For forty years, he produced and distributed Project
Gutenberg-tm eBooks with only a loose network of volunteer support.
Project Gutenberg-tm eBooks are often created from several printed
editions, all of which are confirmed as Public Domain in the U.S.
unless a copyright notice is included. Thus, we do not necessarily
keep eBooks in compliance with any particular paper edition.
Most people start at our Web site which has the main PG search facility:
www.gutenberg.org
This Web site includes information about Project Gutenberg-tm,
including how to make donations to the Project Gutenberg Literary
Archive Foundation, how to help produce our new eBooks, and how to
subscribe to our email newsletter to hear about new eBooks.