*** START OF THE PROJECT GUTENBERG EBOOK 47766 ***
Transcriber’s notes:
In this transcription a black dotted underline indicates a hyperlink
to a page, illustration or footnote; hyperlinks are also highlighted in
colour when the mouse pointer hovers over them. A red dashed underline
marks a hidden comment that can be revealed by hovering the mouse
pointer over the underlined text. Page numbers are shown in the right
margin. Footnotes are located at the end of the book.
The text contains uncommon characters that will not necessarily display
correctly with all viewing devices. If some of the characters look
abnormal, first ensure that the device’s character encoding is set to
Unicode (UTF-8). The default font might also need to be changed to
a Unicode font such as Arial Unicode MS, DejaVu, Segoe UI Symbol or
FreeSerif.
Small-caps font is used extensively, and specifically in the
index to differentiate topics discussed within the text from those
discussed elsewhere. Readers using handheld devices should be aware
that small-caps font is sometimes lost during conversion of the text to
the e-reader format.
Chemical formulae similarly might not render correctly on
handheld devices
The book contains quotations, comments and reports relating to
debates between medical authorities and pharmaceutical suppliers, and
the text is formatted in such a way (in both the original and in this
transcription) as to help readers differentiate the parties to the
debates. Thus, text originating from pharmaceutical sources is
displayed with a smaller font and greater indentation, while that
from medical authorities has closer line spacing and only slight
indentation; however, this pattern is not entirely consistent.
Inconsistent use of opening and closing quotation marks is as in the
original.
There are numerous hyphenation and spelling inconsistencies, the
most noticeable perhaps being the inclusion or omission of a final
‘e’ from chemical terms such as oxid/oxide. These remain as in the
original. A representative list of
the inconsistencies is appended at the end of the transcription
together with a list of the spelling errors that have been corrected
silently. Inconsistent formatting of fractions, e.g. 1-4 cf. 1/4 is
as in the original.
The text contains various words, phrases and comments enclosed by
square brackets. These were inserted by the authors. A few omissions
noted by the transcriber have been inserted within curly brackets to
help differentiate them – hence {of} {a} {be} {“}. A missing full stop
and a missing parenthesis have each been inserted silently; redundant
duplicated punctuation (. ,) has been deleted silently; and on page 299
a duplicated phrase has been deleted silently, viz. this dread disease
is no longer a matter of doubt."
Several large landscape-format tables have been modified in this
transcription to fit narrower viewing screens and ‘key’s added to
identify the column headings. A mathematical error in Table 2 on
page 390 is as in the original – Rat 4 average weight gain/loss
should be 2.1 not 2.7. Table 11 on page 444 contains a footnote
cross-reference to Tables 8 and 10 but these do not exist.
An linked alphabetic table has been inserted at the head of the very
long index to help readers search its contents.

THE PROPAGANDA
FOR REFORM
—IN—
Proprietary Medicines
Part I.
Council Reports
Part II.
Laboratory Contributions
Part III.
Contributions from the Journal: Nostrums
Part IV.
Contributions from the Journal: Miscellany
[Ninth Edition]
REPRINTED FROM THE JOURNAL OF THE
AMERICAN MEDICAL ASSOCIATION
PREFACE
From time to time The Journal of the American
Medical Association has published the reports of the
Council on Pharmacy and Chemistry and the Chemical
Laboratory, as well as other matter on proprietary
medicines. Repeated requests for some of the matter
have led to the compilation of “The Propaganda for
Reform in Proprietary Medicines,” which, in the present
volume, attains its ninth edition.
The seventh, eighth and ninth editions have been
compiled on slightly different principles from their
predecessors. The therapeutic reform work of The Journal and of the Association’s Chemical Laboratory
was at first confined almost entirely to the criticism
and analysis of the so-called ethical proprietaries.
This was right; the medical profession owed it to the
public to combat the nostrum evil within its own ranks.
As the more flagrant evils of the “ethical proprietary”
question were mitigated, the Association has
turned the light on the more widespread and dangerous
“patent medicine” evil. The articles devoted to
“patent medicines” or quackery being naturally of
greater interest to the general public than to the medical
profession, the number of inquiries from laymen
regarding various quacks and nostrums has steadily
increased. It has been thought best, therefore, to
publish separately1 all of the matter from The Journal
relative to quackery and to those nostrums
exploited only or chiefly to the public, and to include
in the Propaganda for Reform practically none of the
matter that is of direct interest primarily to laymen.
In one or two instances in which the subjects were of
equal interest to the profession and to the public matter
that has already appeared in “Nostrums and Quackery”
is also given here; but as a general rule the contents
of the ninth edition of “The Propaganda for
Reform” are of strictly professional interest. Those
physicians who are desirous of obtaining in convenient
form the matter dealing with “patent medicines”
should order the book “Nostrums and Quackery” or
the various pamphlets on the same subjects that have
been issued since “Nostrums and Quackery” came
from the press.
The ninth edition of “Propaganda for Reform”
contains a number of new articles, greatly increasing
the size of the book. It also contains one novel feature
which greatly enhances its value. The index
includes references not only to articles in the book,
but also to matter on proprietaries not accepted by the
Council on Pharmacy and Chemistry which appeared
in The Journal of the American Medical Association
and elsewhere. This index makes of this edition
of “Propaganda for Reform” a very full work of
reference on proprietaries which are undeserving of
recognition. It should be understood, however, that
not all articles indexed are condemned; some are
merely discussed and compared.
TABLE OF CONTENTS
| PART I: COUNCIL REPORTS |
| PAGE |
Acetanilid Mixtures | 9 |
Agar-Lac | 10 |
Anasarcin and Anedemin | 11 |
Maignen Antiseptic Powder | 19 |
Tyree’s Antiseptic Powder | 21 |
Apergels | 26 |
Aseptikons | 26 |
Betul-Ol | 27 |
Peacock’s Bromides and Chionia | 28 |
Bromidia | 31 |
Cactus Grandiflorus | 36 |
Calcreose | 40 |
Campho-Phenique | 40 |
Celerina, Aletris Cordial and Kennedy’s Pinus Canadensis, Light and Dark | 43 |
Cineraria Maritima | 49 |
Hagee’s Cordial of the Extract of Cod Liver Oil Compound | 51 |
Wampole’s Perfected and Tasteless Preparation of an Extract of Cod Liver | 52 |
Waterbury’s Metabolized Cod-Liver Oil Compound | 54 |
Waterbury’s Compound | 57 |
Colchi-Sal | 58 |
Cypridol Capsules | 59 |
Cystogen, Cystogen Aperient and Cystogen-Lithia | 60 |
Cysto-Sedative | 61 |
Taka-Diastase and Liquid Taka-Diastase | 62 |
Digalen Omitted from N. N. R. | 68 |
Dioradin Refused Recognition | 73 |
Echinacea | 79 |
Echtisia, Ecthol and Echitone | 81 |
Ergoapiol | 82 |
Erpiol (Dr. Schrader) | 83 |
False Unicorn (Helonias) | 84 |
Formurol | 85 |
Gastrogen Tablets | 87 |
Glyco-Heroin, Smith | 88 |
Glyco-Thymoline | 92 |
Glycozone | 95 |
Gardner’s Syrup of Hydriodic Acid | 97 |
Hyperol | 100 |
Ingluvin | 101 |
Intestinal Antiseptic W-A | 103 |
Bannerman’s Intravenous Solution | 105 |
Iodalia | 106 |
Iodex | 107 |
Iodia | 108 |
Burnham’s Soluble Iodine | 110 |
Iodotone | 113 |
Iosaline | 113 |
Nourry Wine | 115 |
Labordine | 115 |
Lactobacilline Omitted from N. N. R. | 120 |
Reexamination of Lactopeptine | 121 |
Meat and Beef Juices | 123 |
Valentine’s Meat Juice | 129 |
Medicinal Foods | 131 |
Migrainin | 135 |
Neurilla | 136 |
Neurosine, Dioviburnia, Germiletum and Palpebrine | 139 |
Oxychlorine | 147 |
Pam-Ala, Another Worthless Quinin Substitute | 149 |
Papayans Bell | 151 |
Passiflora and Daniel’s Concentrated Tincture of Passiflora | 156 |
Liquid Combinations Containing Pepsin and Pancreatin | 157 |
Pepto-Mangan (Gude) | 159 |
Liquid Petrolatum or “Russian Mineral Oil” | 161 |
Clinical Experience with Liquid Paraffin (Liquid Petrolatum) | 167 |
Angier’s Emulsion | 169 |
Phecolates, Phecolax, Phecozymes and Phecotones | 174 |
Phenol Sodique | 175 |
Phytin and Fortossan | 178 |
Prunoids | 178 |
Sal Hepatica | 179 |
Sanmetto | 182 |
Secretogen | 185 |
Sinkina | 188 |
Somnos | 193 |
Succus Alterans | 195 |
Sulpho-Lythin | 196 |
Taurocol | 198 |
Tri-Iodides, Three Chlorides and Maizo-Lithium | 198 |
Thialion | 205 |
Unguentum Selenio Vanadic (V. Roemer) | 207 |
Unicorn Root, Wild Yam and Wild Indigo | 208 |
Proprietary Vanadium Preparations | 209 |
Venarsen | 212 |
Venodine | 214 |
Veracolate | 216 |
Hayden’s Viburnum Compound | 218 |
Vin Mariani | 221 |
Virol | 225 |
| PART II: CONTRIBUTIONS FROM THE CHEMICAL LABORATORY |
Anusol Hemorrhoidal Suppositories | 227 |
Aromatic Digestive Tablets | 229 |
Burnham’s Soluble Iodin | 233 |
“Hydrocyanate of Iron-Tilden” | 235 |
Hymosa | 238 |
Micajah’s Medicated Uterine Wafers | 240 |
Noitol and Anadol | 245 |
Pix Cresol | 247 |
Saliodin | 249 |
Theobromin Sodium Salicylate Versus “Diuretin”; The Economical Aspect | 251 |
Unguentine | 254 |
Uricedin | 256 |
Uriseptin | 256 |
Zemacol | 259 |
Zyme-Oid | 261 |
PART III: CONTRIBUTIONS FROM THE JOURNAL:
NOSTRUMS |
Alleotone | 264 |
Baume Analgésique Bengué | 267 |
Antidiabeticum-Bauer | 267 |
Antikamnia | 268 |
Anusol Suppositories | 280 |
Aspiro-Lithine | 281 |
Bell-Ans (Pa-Pay-Ans, Bell) | 282 |
Biosol | 284 |
Bromin-Iodin Compound | 285 |
Calmine | 286 |
Camphenol | 287 |
Chologen | 288 |
Hagee’s Cordial of Cod-Liver Oil | 289 |
Waterbury’s Compound Once More | 291 |
Collyrium-Wyeth | 292 |
Diatussin | 293 |
Enteronol | 294 |
Expurgo (Sanol) Anti-Diabetes | 299 |
Formamint | 303 |
Gomenol | 304 |
Headache Cures | 305 |
Hectine | 308 |
Hydronaphthol | 308 |
Hydrozone | 309 |
Hypoquinidol | 310 |
Iodonucleoid | 310 |
Iridium | 312 |
Iron Tropon | 313 |
Kutnow’s Powder | 314 |
Lymph Compound R-H and Orchitic Fluid Tablets | 317 |
Lysol—The Evolution of a Proprietary | 318 |
Thompson’s Malted Food Company | 319 |
Manola | 323 |
Mercol | 326 |
Midol and Nurito | 327 |
Mu-col | 329 |
Narkine | 329 |
Papine | 330 |
Pasadyne | 332 |
Pas-Avena | 333 |
Pertussin | 334 |
Phenalgin—A Typical Example | 335 |
Pheno-Bromate | 343 |
Phenolphthalein | 343 |
Mixed Vaccine and Phylacogens | 346 |
The Danger in Protonuclein, a Preparation Containing Thyroid | 348 |
Purgen | 349 |
Pyo-Atoxin | 350 |
Resinol | 352 |
Resor-Bisnol | 353 |
Robinol and Sevetol | 353 |
Salacetin | 356 |
Sal-Codeia-Bell | 357 |
Sanatogen | 358 |
| Sanatogen: a Scientific Investigation of Its Alleged Action | |
on the Recuperating Powers of the Blood | 378 |
| The Feeding Value of Sanatogen Compared with Commercial Casein | |
with Respect to Maintenance and Growth | 385 |
Poehl’s Spermin in Arteriosclerosis | 395 |
Syrup of Cocillana Compound | 396 |
Aubergier’s Syrup of Lactucarium | 399 |
Tartarlithine | 401 |
Thoxos | 402 |
Trypsogen | 403 |
Tyree’s Antiseptic Powder | 404 |
Vapo-Cresolene | 408 |
Vasogen and Iodovasogen | 408 |
Viburnum Compound—and other Nostrums | 409 |
Wheeler’s Nerve Vitalizer | 411 |
Zymotoid | 412 |
PART IV: CONTRIBUTIONS FROM THE JOURNAL:
MISCELLANEOUS MATTER |
Acetphenetidin and Phenacetin—Their Relative Purity | 414 |
Clean Advertising | 418 |
Lippincott’s Magazine | 419 |
Medical Journal Advertising | 422 |
Medical Journals and the Great American Fraud | 426 |
The Army and Navy Medical Record | 432 |
The Medical Times Advertisements | 438 |
Cause for Optimism | 440 |
The Comparative Nutrient Value of Cod Liver Oil and Cod Liver Oil Cordials | 442 |
Diabetic Foods Offered for Sale in the United States | 446 |
The Jireh Diabetic Food Company | 451 |
The Name “Epinephrin” Versus the Name “Adrenalin” | 454 |
The Hord Sanitarium | 456 |
The German Propaganda for Reform | 458 |
The German Council on Pharmacy and Chemistry | 459 |
Grand Prix and Gold Medals for Sale | 462 |
The Hypophosphite Fallacy | 464 |
Buffalo Lithia Water | 467 |
Meat Extracts and Meat Juices | 470 |
Pharmaceutical Manufacturers and the Great American Fraud | 474 |
Dowd’s Phosphatometer | 476 |
Amorphous Phosphorus | 478 |
THE PROPAGANDA FOR REFORM
IN PROPRIETARY MEDICINES
PART I
COUNCIL REPORTS
ACETANILID MIXTURES[A]
Report of the Council on Pharmacy and Chemistry
To the Council on Pharmacy and Chemistry:
In response to the request of your chairman we have
investigated the below-mentioned preparations and report
as follows:
Specimens of the articles were bought in different cities
in the open market, and in original sealed packages, and
were analyzed by some of us or under our direction. Each
article was examined by at least two chemists, and some
were subjected to several analyses. While certain of the
preparations are represented as being chemical compounds,
the specimens examined were all found to be mixtures, the
principal ingredient being acetanilid. The percentage proportions
of acetanilid given below are the minimum obtained
by any of the analysts.
Soda and ammonia, combined with carbonic acid, are calculated
and reported as sodium bicarbonate and as
ammonium carbonate (U. S. P.) respectively. Salicylic acid
is calculated and reported as sodium salicylate. Diluents
and other constituents than those reported were not determined.
AMMONOL
According to the analyses of the contents of the original
sealed packages as purchased, this was found to be a mixture,
and to contain the following ingredients approximately
in the proportions given:
| Acetanilid. | Sodium Bicarb. | Ammonium Carb. |
| 50. | 25. | 20. |
ANTIKAMNIA[B]
According to the analyses of the contents of the original
sealed packages as purchased, this was found to be a mixture,
and to contain the following ingredients approximately
in the proportions given:
| Acetanilid | Caffein | Citric Acid | Sodium Bicarb. |
| 68. | 5. | 5. | 20. |
KOEHLER’S HEADACHE POWDERS
According to the analyses of the contents of the original
sealed packages as purchased, this was found to be a mixture,
and to contain the following ingredients approximately
in the proportions given:
ORANGEINE
According to the analyses of the contents of the original
sealed packages as purchased, this was found to be a mixture,
and to contain the following ingredients approximately
in the proportions given:
| Acetanilid | Sodium Bicarb. | Caffein |
| 43. | 18. | 10. |
Other constituents said to be present were not determined.
PHENALGIN[C]
According to the analyses of the contents of the original
sealed packages as purchased, this was found to be a mixture,
and to contain the following ingredients approximately
in the proportions given:
| Acetanilid | Sodium Bicarb. | Ammonium Carb. |
| 57. | 29. | 10. |
Certain packages of phenalgin were purchased which on
analysis did not show ammonium carbonate.
SALACETIN[D]
According to the analyses of the contents of the original
sealed packages as purchased, this was found to be a mixture,
and to contain the following ingredients approximately
in the proportions given:
| Acetanilid | Sodium Bicarb. | Sodium Salicylate |
| 43. | 21. | 20. |
We recommend that this report be printed in The Journal
of the American Medical Association.
| Respectfully submitted, |
| J. H. Long, M.S., Sc.D., |  | Committee on Chemistry,
Council on Pharmacy and
Chemistry of the A. M. A. |
| W. A. Puckner, Ph.G., |
| S. P. Sadtler, Ph.D., |
| J. Stieglitz, Ph.D., |
| H. W. Wiley, M.D., Ph.D., |
(From The Journal A. M. A., June 3, 1905).
AGAR-LAC
Report of the Council on Pharmacy and Chemistry
Agar-lac, said to be the product of “Agar-lac, Inc.,” is sold
by E. Fougera and Company, New York. The following
“formula” for Agar-lac is published:
“Agar-Agar with Lactic Ferments | Grs. | 41⁄2 |
Phenolphthalein | Grs. | 1⁄2” |
Regarding the “lactic ferment,” the identity of which is
not declared by the manufacturer and for the viability of
which no precautions appear to be taken, the Council’s expert
on lactic acid ferments reported that Bacillus bulgaricus was
present in small numbers only and that there were at least
two other bacteria present, one of which is a gas-former of
the Bacillus coli type.
The Council found that the amount of agar-agar in Agar-lac
and the identity of the “lactic ferment” are not declared;
that the name “Agar-lac” is blown in the glass and that the
method of its exploitation will lead laymen to use it to their
detriment; that the claims that it “facilitates assimilation
of proteids” and that it is of value as an aid to “gastro-intestinal
digestion” give a false value to the mixture and
that the claims emphasize the action of agar-agar when
from the composition it is evident that the phenolphthalein
action will predominate; that the name does not indicate its
predominating constituent, phenolphthalein, and that the use
of a ready-made combination of cathartic drugs, such as agar-agar
and phenolphthalein with lactic acid ferments, is
unscientific. The Council therefore refused recognition to
Agar-lac.—(From The Journal A. M. A., Nov. 14, 1914.)
ANASARCIN AND ANEDEMIN
Reports of the Council on Pharmacy and Chemistry and
Comments Thereon
The following reports were submitted to the Council by the
subcommittee to which these articles were assigned:
ANASARCIN
To the Council on Pharmacy and Chemistry:—Your subcommittee
to whom Anasarcin (Anasarcin Chemical Co., Winchester,
Tenn.) was assigned, herewith submits its report:
This remedy is offered in two forms: “Anasarcin Tablets,”
a pretended combination of the active principles of oxydendron
arboreum, sambucus canadensis, and urginea scilla;
and “Anasarcin Elixir,” said to contain the active principles
of oxydendron, sambucus, hepatica and potassium nitrate.
The advertisements of these articles conflict with the rules
of the Council as follows:
With Rules 1 and 2: The composition of these articles is
kept secret, in that the proportion of the ingredients is not
furnished. The statement that it contains the “active principles”
is misleading, since these are for the most part
unknown.
With Rule 6: The description of the pharmacologic action
of Anasarcin agrees practically with that of squill. No
material part of its effects can be attributed to the other
ingredients. Nevertheless, the advertisement studiously cultivates
the impression that Anasarcin has no relation whatever
to the digitalis group in which scilla is commonly placed.
The claims are therefore misleading. The claim of its infinite
superiority to digitalis, the claims that it cures neurasthenia,
eliminates uric acid in rheumatism, and is useful
in obesity, cystitis, lumbago and eclampsia, dyspepsia and
asthma, and that it works wonders in exophthalmic goiter,
appear exaggerated or false.
The recommendation of its indiscriminate use in nephritis,
for lowering the blood-pressure and the statement (contradicted
in the firm’s own literature) that it is not depressing,
are actually dangerous.
It is recommended that the articles be refused recognition,
and that the report, with explanations, be published.
ANEDEMIN
To the Council:—Your subcommittee to whom Anedemin
(Anedemin Chemical Co., Winchester, Tenn.) was assigned
herewith submits its report:
Anedemin is an evident imitation of Anasarcin. It is marketed
as tablets, said to contain the isolated active principles
of strophanthus, apocynum, squill and sambucus, chemically
combined. The quantities are not stated. The therapeutic
claims are copied almost literally from the Anasarcin circulars
and are equally false. Anedemin, therefore, conflicts
with Rules 1, 5, 6 and 7.
It is recommended that this report be published, with comments.
The reports were adopted by the Council and are herewith
published.
W. A. Puckner, Secretary.
Anasarcin
This wonderful remedy, Anasarcin, has already been
exposed in these columns (The Journal A. M. A., Jan. 27,
1906), but it deserves additional mention, as it teaches several
important lessons of general application. It is a typical
example of the revival, under a new name and a thin
disguise, of an old, time-worn article, squill, presumably
because experience has demonstrated its general inferiority
to other drugs. Anasarcin further illustrates the dangers
involved in the use of semi-secret nostrums. It also shows
how a short experience with a widely advertised but little
understood drug is apt to lead to conclusions which more
extensive experience demonstrates to be entirely fallacious.
The first lesson is, that formulas are not always what
they seem. A hasty glance at the formula of Anasarcin
tablets, the basis of the Anasarcin dropsy cure, creates the
impression that it is a non-secret remedy; for it is said to
represent a combination of the active principles of oxydendron,
sambucus and scilla. As a matter of fact, it is
a secret nostrum of the insidious kind. A formula which
omits the quantities of its potent ingredients means very little.
Further than this, we do not hesitate to charge that
the claimed composition is a deliberate deception. The circulars
emphasize the claim that Anasarcin consists of the
isolated principles, and not of the crude drugs. Now, the
isolated active principles of sambucus and oxydendron are
not on the market, for the good and sufficient reason that
no active principles have ever been isolated. Are we to
believe that the Anasarcin Company has surpassed the
accredited chemists and has discovered such principles and is
isolating them? We shall have more to say on this subject
presently; but any one in the least familiar with the difficulties
attending the isolation of organic principles knows such
an idea to be preposterous. Indeed, it is absolutely incompatible
with the exhibition of ignorance of the elementary
facts of pharmaceutical chemistry which is given by these
people when they call the active principles of digitalis and
squill “alkaloids.”
It is an axiom that the effects of a mixture can only be
understood if the action of its components are known. So
far as we know, the physiologic effects of oxydendron and
sambucus have never been scientifically investigated, for the
simple reason that they are too slight and indefinite to
promise results. Both are credited with some slight, obscure
diuretic action. Oxydendron, the sour wood or sorrel tree,
is a small tree of the heath family, the acid leaves of which
are said to be chewed by hunters for their pleasant taste and
for the relief of thirst. Sambucus is the common elder. It
is most unlikely that these two innocuous substances should
play any part in the claimed powerful effect of Anasarcin;
they are evidently put in the formula, we do not say in the
preparation, to obscure the fact that Anasarcin is composed
principally of squill. That this is so can be gathered unmistakably
from a study of the pharmacologic action of Anasarcin
as described by its promoters:
Acting primarily on the heart and arterial systems through the nerve
ganglia, a natural physiologic balance is established between the arterial
and venous systems, whereby effusions ... are eliminated.... Coincident
with this action there is a noteworthy slowing of the pulse.... If
the remedy is pushed, can be brought down to 20 or 30
beats per minute.... Its physiological action is to stimulate the
cardiac motor-ganglia through the cardiac plexus of the sympathetic
system and at the same time exert an inhibitory influence upon the
cardiac fibers of the pneumogastric, thereby dilating the arterioles,
slowing the heart’s action, and increasing the force of the systole.... The
prolonged diastole allows the ventricle time to completely
fill, and the more forcible contraction causes the mitral valve to
close more thoroughly and at the same time increases pressure in the
coronary arteries, serving thereby the double purpose of relieving pulmonary
engorgement and increasing heart nutrition.
Anasarcin will nauseate some persons.
To appreciate fully the meaning of this description of the
actions of Anasarcin, it should be compared with the effects
of the digitalis group, to which squill belongs. The following
account is quoted literally from a recent text-book of
pharmacology (Sollmann):
The phenomena of the therapeutic stage of digitalis action
are said to be:
1. Slowing of the heart, with systole and diastole both
lengthened.
2. Increased strength of beat, leading to greater efficiency
of the individual contractions, and to an increase in the
total efficiency.
3. A tendency to the systolic phase.
4. A rise of blood-pressure, due mainly to the increased
action of the heart, but partly also to a vasoconstriction.
The therapeutic action may be explained, in part, as
follows:
A larger amount of blood will be thrown into the aorta
and coronary circulation. The first effect will be an improved
nutrition of the heart.... The tonic action ... narrows
the ring of the valves, brings them together, narrows
the orifice.... The venous congestion will tend to be
relieved. This relief ... will fall in the first place on
the lungs.... The lowering of the venous pressure will
tend to cause absorption of the effusions.
The nauseant action of squill, which is alluded to in connection
with Anasarcin, is too well known to require more
than a mention.
In brief, then, it appears from the statements of the Anasarcin
Company that the action of the remedy is that of
squill and that the other ingredients are a mere blind. It is,
of course, well known that squill can be used as a substitute
for digitalis in cardiac dropsy, although it is generally
considered very inferior to the latter drug. Rose Bradford,
for instance, states: “Squill is not used to any extent in the
treatment of cardiac disease and cardiac dropsy, digitalis
being a far more efficient and less toxic substance.” However,
it has been frequently observed that digitalis occasionally
fails, and it may then be replaced successfully by
another member of the group. At all events, it is very likely
that squill is a fairly efficient substitute for digitalis, especially
when it is supplemented by a very free course of
Epsom salts and by potassium nitrate (the active ingredient
of Anasarcin Elixir), both of which are stated to be essential
adjuvants to the Anasarcin (or squill) tablets. There
can be no objection to the use of squill when it is indicated;
but any one who wishes to use it should do so with his
eyes open, knowing what substance he is using and how
much (which he does not in Anasarcin); knowing also that
it has the same indications and limitations as digitalis. He
should not be misled by such statements as the following:
“Does what dropsy medicaments have hitherto failed to accomplish.”
“Superior to digitalis, strophanthus, scoparius, squills, acetate of
potash and the hydragogue cathartics all put together.”
“The only known relief [how modest!] and permanent cure of
dropsies.”
“Unrivaled heart tonic.” “The most powerful agent known.”
Any one wishing to use squill should take the trouble to
acquaint himself with the results obtained by competent and
independent observers, and not rely on it in eclampsia, septicemia,
“vices of civilization,” all forms of neurasthenia, as
“an active eliminator of uric acid in rheumatism,” in hepatic
cirrhosis, dyspepsia, asthma, obesity, cystitis (!), lumbago,
exophthalmic goiter, etc.
He should also learn the contra-indications to the use of
squill, deducible from the fact that it causes vasoconstriction
and raises the blood-pressure (prohibiting its use in Bright’s
disease and arteriosclerosis), and that it produces marked
gastric irritation, consequently nausea and depression, that it
is a very toxic agent, and that the dangers of cumulative
action must be borne in mind. In respect to these the advertisements
of the Anasarcin people are little short of criminal,
for these state:
“Safe in administration.” “Non-toxic as ordinarily administered.”
“Will nauseate some persons,” but “the reaction from the temporary
depression is prompt.” “In Bright’s disease, both the interstitial and
parenchymatous forms of nephritis, acute or chronic, no remedy ... to
equal it in efficacy.” “Without increasing the debility of the patient
or interfering with nutrition by producing loss of appetite....”
“This treatment is to be continued without cessation until all symptoms
of dropsy have disappeared.”
Physicians who are inclined to disregard this warning, and
who follow the advice of the Anasarcin people, should
remember that their patients—or their friends—will put the
blame for the results, which are bound to follow sooner or
later, on the prescribers, and not on the deceptive advertisements
of the Anasarcin Chemical Company.
There is another little matter which throws an illuminating
side-light on the Anasarcin Company. They take every occasion
to say that Anasarcin is “not offered to the laity,”
“never sold to the laity,” etc.; but witness the following,
which was found in the Retail Druggist of May, 1906, p. 179.
The italics are ours.
CURE FOR DROPSY.
“As every druggist knows, dropsy has been one of the incurable
diseases when caused either from heart, liver or kidney trouble. A
pharmacist in Winchester, Tenn., has worked out a remedy called
Anasarcin, which he is exploiting to the physicians, and his remedy is
showing itself as possessing great merit. Several hopeless cases have
been treated as a last resort by Anasarcin and in a very short time the
patient has shown marked improvement and has effected permanent cures.
“The result of the cases as handled by the physician with the aid of
Anasarcin has been so easily and quickly cured that physicians of
Tennessee and the southern states are high in their praises of the
remedy. The company which now manufactures and sells it is known
as the Anasarcin Chemical Co., of Winchester, Tenn. Any druggist
who knows of a case of dropsy would be conferring a favor on the
patient and mankind in general by telling the party or his physician of
the southern pharmacist, and we have no doubt but what a prompt
relief and permanent cure would be affected.” [Probably means
effected.—Ed.]
Anedemin
If we are disposed to doubt the vaunted scientific ability
of the Anasarcin Company, we are forced to admire their
business methods, at least, if there is any truth in the saying
that imitation is the seal of success. Anasarcin has had
this rather undesirable compliment paid to it, for its native
town of Winchester has given birth to another remedy,
Anedemin, which looks like a fair-haired twin brother. The
Anedemin Company has adopted Anasarcin almost bodily.
The name—“opposed to edema”—is about as close as the
copyright laws permit. The pharmacologic and therapeutic
claims agree almost literally with those of Anasarcin and
contain the same exaggerations and dangerous misstatements.
There is the same emphasis on free purgation with
Epsom salts. The dose is the same. Both are marketed at
$2.00 for a box of 100—only the Anedemin people have
adopted the prize package device of throwing in 20 or 30
tablets extra, for good measure, and give a discount of
75 cents or so.

Laboratory and Warehouse of the Anasarcin Chemical Company,
Winchester, Tenn.
In short, the Anedemin Company has appropriated all of
Anasarcin which they considered of any value. It is, therefore,
rather suggestive that they drew the line at the formula.
Anasarcin is said to contain squill, sambucus and
oxydendron; Anedemin discards the oxydendron and reinforces
the squill with strophanthus and apocynum. Notwithstanding
this material change in composition, the actions
are described as identical; this is again rather suggestive.
The Anedemin Company, like the Anasarcin Company,
scorns crude drugs and claims to use only the isolated principles.
It was saved the trouble of discovering active principles
for strophanthus and apocynum, for these are known;
but it managed to find some scope for its inventive genius,
“both drugs being so chemically treated and disposed as
to absolutely eliminate all objectionable and disagreeable
properties and effects” so as to convert a vasoconstrictor
action into a dilator action; so as to render them non-toxic
and non-cumulative; so as to deprive apocynum of its characteristic
nauseant effect. Who can say that the days of
miracles are past? Even this is not the limit of Anedemin
alchemy; if we are to believe their claims, they have succeeded
in forcing strophanthin, apocynum, scillain, etc., to
combine with each other: “It is a definite chemical compound
of the active principles” of these drugs! This makes the
achievements of Emil Fischer in synthesizing sugars and
proteids appear as mere child’s play.
Since the formulas were completed, however, clinical
reports have been numerous enough—almost too numerous,
if we are to believe them. Anedemin has been on the market
for less than three years; the circulars emphasize that testimonials
and endorsements are not solicited. Nevertheless,
we are told that it is “endorsed by over fifty thousand clinicians
throughout the United States.” Since the total number
of physicians in the United States and Canada is only
about 128,000, this means that nearly every second physician
has endorsed Anedemin. The Anasarcin Company solicits
endorsements and they seem to do the larger business.
Hence the majority of physicians of the United States must
have written an endorsement of either Anedemin or Anasarcin,
or both. Or is this statement another “invention”?
It is a little peculiar that nearly all the endorsements come
from small towns in sparsely settled districts; practically
none from the centers of population. Does this mean that
dropsy is more common in the rural communities than in
the cities?
THE INVENTORS OF ANASARCIN AND ANEDEMIN
Even the newspapers, when they tax our credulity with
pretended scientific “discoveries,” feel the moral obligation
of justifying themselves by telling us something of the
personality and experience of the discoverers. We may
ask, therefore, who are these expert pharmaceutic and synthetic
chemists, these manufacturers of active principles,
these skilled clinicians of wide experience, who have “intelligently
built up the formula by wide application”? What
are we told of these men who ask us to believe, on their
mere assurance, in miracles and feats of magic; who tell
us that they have converted neutral principles into alkaloids,
that they have effected definite chemical compounds between
these neutral principles, that they have discovered principles
that do not exist, that they have changed the actions
of these principles to suit their wishes, that, in short, they
have reversed the laws of Nature?
These companies are located in Winchester, Tenn., a town
of about 1,500 inhabitants, situated in an agricultural country.
The town boasts of neither scientific schools, colleges,
universities nor laboratories. The Anasarcin Company was
organized in 1902, the incorporators and directors being Dr.
John W. Grisard and his sons, Dr. John P. Grisard, B. A.
Grisard, and A. F. Grisard, and Will E. Walker, all of
Winchester. Dr. John W. Grisard seems to be the originator
and promoter of Anasarcin. W. E. Walker is an insurance
solicitor of Winchester and is not actively identified
with the business. We are informed that he owns but a
single share of stock having a face value of $100, and that
he was added to the company in order to comply with the
laws of Tennessee, which require five directors for any corporation.
Dr. John W. Grisard, the father, has practically
retired, but still has a general supervising interest in the
business. There is no regularly licensed pharmacist or
chemist connected with the company. The office is in the
rear of a jewelry store in the business part of Winchester
and on the second floor above. According to our reporter,
an office force of about ten stenographers and clerks handles
the correspondence and labels and sends out the preparation
which is made in a crude frame building located on
a side street and without laboratory equipment. According
to our reporter, the work is done by the Grisards and a
colored man.
The Anedemin Chemical Company was organized in 1905
with a capital of $20,000, the incorporators and directors
being Dr. T. B. Anderton, Floyd Estill, J. J. Lynch, J. M.
Littleton and I. G. Phillips, all residents of Winchester,
and all lawyers with the exception of Dr. T. B. Anderton.
A Mr. Gordon, a clerical employee of the company, is
reported to have active charge of the business, to prepare
the medicine and conduct the correspondence. The office
headquarters, laboratory and complete outfit of the Anedemin
Company comprises two rooms over the law office of Estill
& Littleton. No one connected with the company is a
regularly licensed pharmacist or graduate chemist.
Of the six physicians located in Winchester, three (50 per
cent.) are engaged in the dropsical cure business. Poor
Winchester! Aside from their connection with these two
nostrums, these physicians may be estimable and worthy
citizens, but where, pray, did they find the extensive clinical
facilities and pharmaceutical knowledge necessary for their
wonderful and epoch-making discovery? Were they aided
in their scientific work by the four lawyers connected with
the Anedemin Company or by the insurance solicitor who
is a director of the Anasarcin Company? Did the 1,500
inhabitants of the town furnish the vast clinical material
necessary for discovering and working out the formulas of
these two preparations? If so, we fear that dropsical affections
are much more prevalent in Winchester than in any
other known spot on the globe. This matter should be
investigated. Without doubt the vital statistics of Franklin
County would be most interesting and we commend them
to the special attention of the medical profession in
Tennessee.—(From The Journal A. M. A., May 4 and 11, 1907.)
MAIGNEN ANTISEPTIC POWDER
Report of the Council on Pharmacy and Chemistry
The report which appears below was submitted by a referee
and after adoption by the Council was sent to the manufacturer
for comment, in accordance with the Council’s regular
procedure in such cases. The manufacturer’s comments
were transmitted to a second referee, who reported that after
a careful consideration of the manufacturer’s reply he saw
no valid reason for a modification of the report. The referee
also reported that a visit to the Maignen Institute further
served to convince him of the viciousness of the treatment
as given and that the records made by the persons in the
employ of the institute were too inadequate to serve as clinical
evidence. On the referee’s recommendation, the report as
originally adopted was reendorsed by the Council and authorized
for publication.
W. A. Puckner, Secretary.
Maignen Antiseptic Powder is marketed by the “Maignen
Institute for the Study of Bacterial Diseases,” Philadelphia.
It is claimed to be a mixture of calcium hydroxid, sodium
carbonate, aluminum sulphate and boric acid, but no statement
as to the amount of the several constituents is furnished.
Its action depends on the sodium hydroxid which is formed
when the powder is treated with water, 1 Gm. of the powder
as now submitted to the Council yielding 0.32 Gm. of sodium
hydroxid (NaOH) and a specimen obtained a year ago
yielding 0.28 Gm. Its promiscuous use is recommended both
to physicians and to the public with claims which are
extravagant, preposterous and even dangerous.
A pamphlet, clearly intended for the laity, entitled “What
Is Catarrh?” gives direction for the “sterilization” of the
nose, throat, stomach, lungs, eyes, gums, mouth and the
genito-urinary tract. The following, taken from this pamphlet,
illustrates the absurdity of the claims made for
Maignen Antiseptic Powder:
“STERILIZATION OF THE STOMACH
“Take of the Maignen Antiseptic Powder half the quantity raised
on a dime, scant.
“ADD to a tumbler of water, preferably warm, and stir.
“Drink slowly.
“This is what may happen:
“1). Belching may be the first indication of the sterilization of the
stomach.
“2). The excess of acidity is corrected.
“3). The fermentation is stopped.
“4). The sterilization extends to the Intestinal Tract.
“5). The bowels are regulated without purgation.
“6). The whole metabolic process is improved.
“When and how often to drink the antiseptic solution.
“a). For Indigestion, whenever distressed, before or after meals.
“b). For Constipation, half an hour before breakfast or last thing at
night.
“c). For Gastro-Intestinal troubles, such as Typhoid Fever, Dysentery
and Cholera, which are the most serious forms of catarrhal inflammation,
take half a tumbler or a whole tumbler of hot water with half
the quantity of Powder raised on a dime every hour, and between
times a glass of generous [sic] wine.
“Remarks
“The sterilization recommended here is a plain disinfecting process
which does not interfere with medical treatment. It is, on the contrary,
of great assistance to it.
“It has been found very effective in breaking up the cigarette habit.
It does away with the craving by removing the morbid irritation of the
mucous membrane.”
Eighty-eight disorders are listed in a pamphlet entitled
“Antiseptic Therapeutics” all of which are reported as having
been treated with success. The dangerous character of the
Maignen “sterilization” propaganda is illustrated by a pamphlet
“First Aid to Baby-Sick” and by the recommendation
on the trade package:
“To prevent Blood Poisoning, Lockjaw, Hydrophobia and Infectious
Diseases.”
The legend on the trade package and the advertising
matter contained in it are likely to lead the public to place
dependence on a weak sodium hydroxid solution as a means
of preventing blood-poison, lockjaw, hydrophobia and infectious
diseases. The pamphlet “First Aid to Baby-Sick”
recommends its use in sore eyes, teething and sore mouth,
sore throat, running ears, running nose, sore chest, summer
complaint, skin troubles and infection after vaccination; if
any trust is put in these claims, they are bound to lead to
the sacrifice of many infants through neglect of proper
treatment.
Patent No. 1,086,339 has been granted on this powder to
P. J. A. Maignen of Philadelphia by the U. S. patent office
on the following specification of claim made in the application:
“1. A process for destroying microorganisms on living tissue, without
injuring the latter ... whereby the growth of such organisms is
inhibited and their substance dissolved without deleterious effect upon
contiguous healthy tissue.”
With brazen assurance this grant has been twisted by the
unscrupulous promoters into a government endorsement of
the preparation. It, of course, means nothing of the sort, as,
no doubt, in accordance with legal routine the patent was
granted without any investigation by the patent office to
determine the effectiveness of the powder for the purpose
claimed.
In view of the dangerous, unwarranted and absurd claims
made for Maignen Antiseptic Powder the referee recommends
that it be refused recognition, and that the Council declare
its agreement with views expressed in the article “Maignen
Pulv.” published in The Journal, Feb. 15, 1913, p. 537, particularly
the following:
“The germicidal powers of strong alkalies have long been known,
but the inconvenience of their application to tissues and mucous membranes
has prevented their use. That they will be of service when
sufficiently diluted not to irritate the tissues is improbable, for the antiseptic
power of such solution is slight and the disinfectant value practically
nil.”
Because the Maignen Institute has twisted the granting
of U. S. patent No. 1,086,339 into a quasi-endorsement of the
claims made for Maignen Antiseptic Powder it is recommended
that a copy of this report be sent to the Commissioner
of Patents as a protest against the present law, which
authorizes the granting of patents on unproved and improbable
medical claims.—(From The Journal A. M. A., Nov. 14,
1914.)
TYREE’S ANTISEPTIC POWDER[E]
Report of the Council on Pharmacy and Chemistry with Comments
Tyree’s antiseptic powder was assigned for examination to
a subcommittee of the Council, which made the following
report:
To the Council on Pharmacy and Chemistry:—Your subcommittee,
to whom was assigned Tyree’s Pulv. Antiseptic
Comp., marketed by J. S. Tyree, Washington, D. C., reports
as follows: The label on the package states: “This
preparation is a scientific combination of borate of sodium,
alumen, carbolic acid, glycerin and the crystallized principles
of thyme, eucalyptus, gaultheria and mentha, in the
form of a powder,” etc.
The statement that the powder contains the crystalline
principles of thyme, eucalyptus, gaultheria and mentha is
vague and misleading, since the chief medical constituents
of eucalyptus and gaultheria are liquids, but it tends to
convey the impression that the powder contains the essential
constituents of these drugs, namely, thymol, oil of eucalyptus
or eucalyptol, oil of wintergreen, or methyl salicylate, and
menthol.
The literature supplied to physicians claims its composition
to be: “Parts, sod. bor., 50; alumen, 50; ac. carbol., 5;
glycerin, 5; the cryst. principles of thyme, 5; eucalyptus, 5;
gaultheria, 5, and mentha, 5.”
The composition, therefore, might be expressed as follows:
Sodium borate (borax) | 50 parts, or | 38.46 | per cent. |
Alum | 50 parts, or | 38.46 | per cent. |
Phenol (carbolic acid) | 5 parts, or | 3.85 | per cent. |
Glycerin | 5 parts, or | 3.85 | per cent. |
Thymol | 5 parts, or | 3.85 | per cent. |
Oil of eucalyptus or eucalyptol | 5 parts, or | 3.85 | per cent. |
Oil of gaultheria (or methyl salicylate) | 5 parts, or | 3.85 | per cent. |
Menthol | 5 parts, or | 3.85 | per cent. |
Analysis of specimens purchased from different sources in
the open market were made under our direction. The reports
of the chemists show that Tyree’s antiseptic powder contains
no borax, or mere traces only, and that it contains no
alum, or mere traces only. Instead, the analyses show that
boric acid and zinc sulphate are the essential constituents.
The amounts of carbolic acid, thymol, menthol, etc., contained
in the powder, if present, were far below the quantities
indicated by the formula. The presence of glycerin could
not be demonstrated, and, if present, the amount must be
very small.
One chemist reports: The result of analysis shows that
different samples differ slightly in composition, but that the
following indicates the average composition of the product:
| Per Cent. |
Zinc sulphate, anhydrous | 15.56 |
Boric acid | 81.26 |
Volatile matter at 100° C. for four hours | 0.45 |
The undetermined portion consists of salicylic acid, carbolic
acid, menthol and eucalyptol; possibly other antiseptic
agents may be present in very minute quantities.
From the above findings we conclude that Tyree’s antiseptic
powder is a mixture of boric acid and dried zinc
sulphate and antiseptic bodies, such as menthol, salicylic
acid and carbolic acid, eucalyptol, etc. From this it can be
readily seen that the label, which is supposed to set forth
the composition of Tyree’s antiseptic powder, is not in accord
with the facts. The powder does not contain either borate
of sodium or alum, and the presence of glycerin could not
be established. The antiseptic agents, exclusive of the boric
acid, are present only in small amounts.
The report of another analysis concludes as follows:
It evidently contains less than the amount stated of the
principles of thyme, eucalyptus, wintergreen and mint. It
also contains a very small amount indeed of carbolic acid,
much less than that stated. We have been unable to identify
certainly the presence of glycerin, and it is doubtful if it
be present.
From the result of the analysis we feel confident that the
preparation is to all intents and purposes a mixture of boric
acid and sulphate of zinc.
The carbolic acid, thyme, eucalyptus, wintergreen, etc., if
present, are present only in sufficient amount to give the
compound a satisfactory odor.
In view of the fact that J. S. Tyree has given wide publicity
to a formula which the preceding report has shown
to be a deliberate misrepresentation of facts, it is recommended
that the article be refused recognition by the Council
on Pharmacy and Chemistry, and that this report be
published in The Journal of the American Medical Association.
The recommendation of the subcommittee was adopted by
the Council in accordance with which the report is published.
W. A. Puckner, Secretary.
Mr. Tyree, in a letter to Dr. Simmons (which he states he
writes at the request of Dr. Kebler of the Drug Laboratory
of the Department of Agriculture, though he is under no
moral or financial obligation to do so), says that it has been
his intention to inform the medical profession of his reasons
for changing the formula of Tyree’s Antiseptic Powder from
an alum and borax base to a boracic acid and zinc base.
He states that this change was made at the suggestion of
prominent physicians connected with hospital clinics on nose
and throat, venereal and other conditions and that he has
had in contemplation the omission from the label of the
various conditions to which the preparation is applicable.
Mr. Tyree, it will be seen, assumes the right to sell to
physicians a preparation with a descriptive formula which
he acknowledges is false, and he presumes to use his own
pleasure as to the time when he will inform them of its true
composition.
Mr. Tyree does not state when he changed the formula.
We do not know whether it was a year ago, five years ago
or ten years ago, but we do know that the package which
was used in making the first analysis was purchased as
early as last February, and the first chemist’s report was
submitted to the Council March 5, 1906. On April 4 Mr.
Tyree was notified by the Council that the composition of
Tyree’s Antiseptic Powder did not correspond to the formula
published by him.
Whether or not Mr. Tyree is justified in offering our profession
a preparation as composed chiefly of borax and
alum when in reality it is chiefly composed of boric acid
and zinc sulphate, we leave physicians to judge.
Discrepancies Between Facts and Claims—Unfortunate
Attempts of Mr. Tyree at Explanation
A report from the Council on Pharmacy and Chemistry on
Tyree’s Antiseptic Powder appeared in The Journal, Oct.
20, 1906. This showed that the preparation, advertised as
a “scientific combination of borate of sodium, alumen, carbolic
acid, glycerin and the crystallized principles of thyme,
eucalyptus, gaultheria and mentha, in the form of a powder.”
was essentially a mixture of boric acid and sulphate of zinc—approximately
four-fifths of the former to one-fifth of the
latter. “The carbolic acid, thyme, eucalyptus, wintergreen,
etc., if present, are present only in sufficient amount to give
the compound a satisfactory odor.” As will be remembered,
in the correspondence published at that time, Mr. Tyree
attempted to explain the discrepancies between his statements
and the proved facts by intimating that he had recently
changed the formula, and that it was his intention “on or
about the first of February to state to the medical profession
his reasons for changing the formula,” and that the
change had been made “a short time ago, at the suggestion
of several prominent gentlemen.” Since that time, through
circulars and other advertisements, Mr. Tyree has attempted
to explain the matter in various ways. In his latest circular
letter he seems to make a deliberate attempt to mislead
our profession and to misrepresent facts to a degree that
makes it almost impossible to believe that the circular came
from a man who claims to be honorable.
First, however, we shall take this opportunity to publish
some matter which we have had in reserve since the first
exposé was made last October. When it was realized that
Mr. Tyree intended to defend himself by claiming that a
change had recently been made in the powder, we took
occasion to try to secure some of the preparation that had
been on the market for a long time. In this we succeeded
very well. From a Chicago druggist one package was
bought which had been in the store at least since July, 1902—how
much longer is not known. The druggist from whom
the powder was obtained bought the drug store in July,
1902, and this powder was on hand at that time, none having
been bought since. This particular powder was analyzed
by a chemist, who found the composition practically the
same as that given in the Council’s report, this chemist estimating
that it contained approximately 81 per cent. boric
acid and 14 per cent. anhydrous zinc sulphate. Bearing in
mind that for at least four years and ten months Tyree’s
Powder has been essentially the same as it is today, this
letter is very interesting: (The comments in brackets are,
of course, ours.)
“J. S. TYREE,
“Chemist,
“WASHINGTON, D. C.
“April 16, 1907.
“Dr. ————,
“——,
“My Dear Sir:—Doctors and medical publications of
extreme and prejudicial minds often hold and express opinions
in honorable faith, but like all critics, they are not
always familiar with the conditions composing their opinions,
and are often given to expressing them without knowledge
of the true motives and facts in the case.
“If you will read an article that appeared in one of the
medical weeklies some time ago [The Journal of the American
Medical Association, of course] and which has been
copied by several of its offsprings [not many we regret to
say] relating to Tyree’s Antiseptic Powder, you will see that
I had previously informed the editor as well as his council
of investigators, that at the suggestion of prominent physicians,
extensive clinical experimenting [sic] were being made
with some slight [! ! !] changes in my powder, the object
being to develop and extend its usefulness in new lines
[It had already been recommended for about everything2]
and at the same time make it more acceptable to the general
run of the profession. I also notified this editor that these
investigations would not be completed until the first of the
present year, after which time these slight [! ! !] changes
in the formula of Tyree’s Powder would be announced.
[It is now the middle of May; when and where were the
changes announced?3]
“There is nothing new, startling or dangerous in such
changes in formulas. The Pharmacopeias and national
books of authority are continuously improving their formulas.
It is the same with every preparation on the market.
[Mr. Tyree, as a nostrum maker, is in a position to know.
His plea evidently is: “I am no worse than others.”] The
apparent difficulty in my case is caused by my exceptional
frankness [“exceptional frankness” is good under the circumstances]
with the profession in telling them [when and
where?] about this improvement before I was ready to
announce full details and particulars, or place my improved
[sic] powder on the market.
“Yours very truly,
“J. S. Tyree.”
For years Mr. Tyree has been misleading physicians by
making false statements regarding the composition of his
powder and regarding its value as a therapeutic agent. When
exposed he tries to defend himself and his business by statements
and excuses that are worthy of a schoolboy trying
to get out of a bad scrape. We would respectfully suggest
to him that he either take his wonderful powder off the
market, or—which would probably amount to the same
thing—tell the truth, and the whole truth, about it.—(From
The Journal A. M. A., May 18, 1907.)
APERGOLS[F]
Abstract of Report of the Council on Pharmacy
and Chemistry
Apergols, put out by H. K. Wampole Co., Inc., is alleged
to be a “Uterine Stimulant.” Apergols is apparently an
inversion of the name Ergoapiol and the preparation appears
to have essentially the same formula, namely:
Apiol | 5 | min. |
Oil Savine | 1⁄2 | min. |
Ergotin | 1 | gr. |
Aloin | 1⁄8 | gr. |
Aromatics | q. s. |
As in Ergoapiol, the constituent referred to in the formula
as “Apiol” appears to be oleoresin of parsley-seed instead of
the definite substance apiol described in New and Nonofficial
Remedies. In general the claims made for Apergols are the
same as those made for Ergoapiol (see p. 82). The Council
refused admission to Apergols because they are advertised
indirectly to the public, because of unwarranted therapeutic
claims, because of the non-descriptive name and
because the product is unscientific.—(From The Journal
A. M. A., Dec. 12, 1914.)
ASEPTIKONS
Report of the Council on Pharmacy and Chemistry
Aseptikons are vaginal suppositories sold by the Chinosol
Co. of New York. Each suppository is said to contain:
Ac. Salicylici | 2 | gr. |
Ac. Borici | 10 | gr. |
Quin. purae (Alkal.) | 1 | gr. |
Chinosol | 2 | gr. |
But. Cacao | 60 | gr. |
The following claims appear in advertisements:
“These suppositories are indicated in cervicitis, leucorrhea, specific
and non-specific vulvo-vaginitis and in all cases where complete vaginal
antisepsis is desired.”
“Non Toxic, Non Irritating; No Damage to Membranes. Yet a More
Powerful Antiseptic than Bichloride.”
The Council decided that the foregoing claims in the
absence of evidence must be held exaggerated and likely to
mislead, and also that the claim “Stronger than Bichloride”
which appears on the box is misleading.
The position of the Council is that “In the case of pharmaceutical
preparations or mixtures the trade name must be
so framed as to indicate the most potent ingredients.” The
name Aseptikons does not give any indication of the ingredients
of the product.
The Council holds that “The combination of two or more
remedies in a mixture must be considered contrary to scientific
medicine unless a distinct reason exists for such combination.”
No evidence has been submitted to establish the
value of the combination in Aseptikons.
On the basis of the evidence submitted the Council voted
that Aseptikons be refused recognition because unwarranted
and misleading therapeutic claims are made, because the
name does not indicate its potent constituents, and because
the combination of two or more remedies in a mixture is considered
contrary to scientific medicine unless a distinct reason
exists for such combination.—(From The Journal A. M. A.,
Nov. 14, 1914.)
BETUL-OL[G]
Abstract of Report of the Council on Pharmacy
and Chemistry
Betul-ol (E. Fougera and Co., New York) is a methyl
salicylate preparation advertised to physicians (and indirectly
to the public) as an external analgesic and anti-rheumatic.
The statements regarding its composition are
vague, misleading and, as shown by examination in the
Chemical Laboratory of the American Medical Association,
untrue. The therapeutic claims are based on discarded
theories. Although the alleged superiority of natural over
synthetic salicylates has been disproved, physicians are
urged to use Betul-ol because it contains, or is alleged to
contain, a natural salicylate. Another discarded theory is
pressed into service in the claim that the chloral in the
mixture will be absorbed and converted into chloroform in
the blood. The recommendations for the use of Betul-ol in
rheumatism are likely to lead the public to the self-treatment
of rheumatism. In view of the serious complications and
sequelae of rheumatic fever this recommendation is utterly
unjustifiable and a danger to public health—even if the
external application of this mixture in uncertain doses were
as effective as a proper internal use of salicylates—a theory
contrary to experience and unsupported by adequate evidence.
The Council therefore refused recognition to Betul-ol.—(From
The Journal A. M. A., Dec. 12, 1914.)
PEACOCK’S BROMIDES AND CHIONIA
Reports of the Council on Pharmacy and Chemistry
The Council has authorized publication of the following
reports on Peacock’s Bromides and Chionia, sold by the
Peacock Chemical Company, St. Louis.
W. A. Puckner, Secretary.
PEACOCK’S BROMIDES
This is another nostrum of the ordinary mixture type. Of
the various statements concerning composition furnished by
the company, the following gives as much information as
any:
“In Peacock’s Bromides it is designed to unite fifteen grains of the
purest bromides of Potassium, Sodium, Ammonium, Calcium and
Lithium, in such proportion as to insure the bromine equivalent of
potassium bromide. Each fluid drachm about equals, in medicinal
strength, fifteen grains of potassium bromide.”
The label on the trade package indicates the presence of
10 per cent. of alcohol. It will be observed that the proportions
of the different bromids are not stated. Hence, the
assertion of the Peacock Chemical Company that “there is
nothing secret in this compound” cannot be true. A physician
prescribing it cannot know how much of each ingredient
he is giving; it may be 141⁄2 grains of potassium bromid
and 1⁄8 grain each of sodium, ammonium, calcium and lithium
bromid, or any other of an enormous number of possible
permutations of the proportions.
While the theoretical basis of bromid medication is not
yet fully settled, the weight of the best pharmacologic
authority and clinical experience is decidedly against the
dogmatic claim of the Peacock Chemical Company that “the
best result is obtained by prescribing a combination of
bromides.” And if there were any advantage in prescribing
such a combination, the physician ought to regulate the
proportions.
The following quotations are from the advertising matter:
“Being uniform in purity and therapeutic power, it can be relied upon
to produce clinical results which it is believed cannot be obtained from
the use of commercial bromide substitutes.”
“The purity, quality and constant uniformity of this high grade
product have long made it a standard bromide preparation.”
These claims are unfounded. The analyses published in
the concern’s own advertising “literature” show a variation
of 8 per cent., in the bromid content, which certainly indicates
a sufficient lack of uniformity.
Again quoting:
“In order to insure the best results the bromides must be pure, i. e.,
free from alkalies and almost free from chlorides. The U. S. P. allows
three per cent. of chlorides. Peacock’s Bromides contains the least
possible amount of this impurity. Bromism is therefore less frequent
in those cases in which this preparation is employed.”
In view of the claim of low chlorid content, it is interesting
to note that the analyses above referred to show that the
chlorid content is actually higher than that of some other
bromid preparations on the market.
The claim of merit on the ground of freedom from chlorids
is, of course, absurd, and must be regarded as an attempt to
play upon the credulity of the doctor. As a matter of fact,
the average individual takes with his food many times the
amount of chlorid he could possibly take in contaminated
bromid. The 10 per cent. of alcohol would undoubtedly have
a greater disturbing influence on the bromid action than the
amount of chlorid that might be present in any bromid on
the market.
Then we have the statement that, owing to this freedom
from chlorids:
“Bromism is therefore less in those cases in which the preparation
is employed.”
Sodium chlorid, even as an impurity, would retard rather
than favor the development of bromism; sodium chlorid is
even used as an antidote in bromid poisoning.
The therapeutic claims lay stress on the value of the
bromids in sleeplessness, epilepsy, sexual excitement, tetanus,
infantile convulsions, chorea, delirium tremens, the climacteric,
migraine, headache due to pelvic conditions, ovarian
neuralgia, etc. These and other claims, while too vague to
be branded as falsehoods, are misleading and not in accordance
with modern teaching or practice; the latter recognize
the limitations of bromid therapy as well as its scope and
advantages. For instance, in epilepsy the company asserts
that:
“Large doses must be given if we expect to control the convulsions.
We are to be guided by the frequency and the severity of the seizures,
the saturation of the system by bromides and by the age of the patient.
The rule is ‘large doses for long periods but with occasional periodic
monthly or quarterly omissions.’ When we have succeeded in controlling
the convulsions in so far as greatly diminishing the frequency and
severity of the attacks we may then attempt to decrease the dose, but
the results must be carefully watched. Increase in frequency of convulsive
seizures is a sign that the bromides must again be pushed as
before.”
The best modern clinical teaching concerning the treatment
of epilepsy is that bromids should be avoided except as a last
resort. Bromids do not cure, and the amount necessary to
control the convulsions may produce a degree of mental
hebetude that is a greater evil than the disease itself.
It is recommended that the preparation be held ineligible
for admission to N. N. R., because of its conflict with Rules
1, 4, 6 and 10 of the Council, and that this report be published.
CHIONIA
Chionia, according to the statement of the Peacock Chemical
Company, which exploits the product, contains 19 per
cent. alcohol and is “A Preparation of Chionanthus
Virginica.”4
This preparation is advertised particularly as “a potent
hepatic stimulant” and special claims are made for it in
various disturbances of the liver:
“Chionia is very well adapted in the treatment of hepatic congestion
owing to its specific action in depleting the portal circulation.”
In passive congestion of the liver, the manufacturers
would have us believe
“... we have a drug in Chionia that will stimulate the circulation
of the blood and lymphatics of the liver as well as stimulate its physiological
activities and instead of the patient vomiting the blood an internal
depletion of the liver occurs.”
“... in cases of simple jaundice due to circulatory (congestive)
changes in the liver, Chionia is the drug ‘par excellence’ that will
rapidly cause a disappearance of this symptom.”
As a prophylactic against eclampsia, if a history of torpidity
of the liver is obtained:
“CHIONIA should be used during the major portion of child-bearing
period because it acts directly on the liver stimulating its functional
activity.”
Chionanthus virginica has never been shown to have the
slightest pharmacologic activity and no evidence is presented
that its offspring, Chionia, has any therapeutic value whatever
in any disturbance of the liver. The promoters themselves
indicate a lack of faith in their own preparation, for
they advise the use of old and efficient forms of treatment
along with Chionia—heart tonics and laxatives in passive
congestion of the liver, mercurial purge or podophyllin and
sodium phosphate in “biliousness,” and quinin in malaria.
Finally, with delightful English and elaborate insouciance,
they advise in the treatment of eclampsia:
“In all cases the uterus should be emptied as quick as possible. (Version
of Cæsarian Section.)”
The physician who prescribes Chionia promotes a fraud.
The Council held Chionia ineligible for admission to
N. N. R.
[Editorial Comment: In Peacock’s Bromides and Chionia
the Peacock Chemical Company has, for a third of a
century, been foisting on the medical profession nostrums
composed of drugs that are easily combined in any proportion
that the physician may want to prescribe. The company
has been inflicting on the unthinking physician pseudo-scientific
rubbish in the form of advertising literature that
should long ago have been regarded as an insult to the
intelligence of the medical profession. The following medical
journals are carrying advertisements of Peacock’s Bromides
and Chionia:
| Alienist and Neurologist | Medical Fortnightly |
| American Journal of Surgery | Medical Herald |
| American Medicine | Medical Record |
| Archives of Pediatrics | Medical Review of Reviews |
| Atlanta Journal-Record of Medicine | Medical Sentinel |
| Buffalo Medical Journal | Medical Standard |
| Charlotte Medical Journal | Medical Summary |
| Chicago Medical Recorder | Medical Times |
Denver Medical Times and Utah Medical
Journal | Medical World |
| Nashville Journal of Medicine and Surgery |
| Eclectic Medical Journal | New Orleans Medical and Surgical Journal |
| Ellingwood’s Therapeutist | New York Medical Journal |
| Indianapolis Medical Journal | Pacific Medical Journal |
| International Journal of Surgery | Southern Practitioner |
| Lancet-Clinic | Texas Medical Journal |
Louisville Monthly Journal of Medicine
and Surgery | Texas Medical News |
| Therapeutic Gazette |
| Maryland Medical Journal | Wisconsin Medical Recorder |
| Medical Brief | Woman’s Medical Journal] |
| —(From The Journal A. M. A., April 3, 1915.) |
BROMIDIA
Report of the Council on Pharmacy and Chemistry
The following report was submitted to the Council by a
member of its Committee on Therapeutics, with the recommendation
that publication be authorized. This recommendation
was adopted.
W. A. Puckner, Secretary.
Bromidia (Battle & Co., St. Louis) at once suggests
bromids; yet Bromidia is essentially a chloral rather than a
bromid preparation. This nostrum illustrates the need of
the provision in the Council’s Rule 8 under which recognition
is refused pharmaceutical mixtures whose names do not
indicate their most potent ingredients. While the chloral
content of Bromidia has been given considerable publicity,
yet the preparation is used both by physicians and by the
public without due consideration of its potent ingredient.
This fact is attested not only by the fatal results which have
followed its use but also by the many reports of habit formation.
As long ago as in 1887 a fatal case of poisoning was
reported5 to the medical society of the District of Columbia
due to an overdose taken by a Bromidia addict. The physician
who reported this case also gave his experience with
another patient who had the Bromidia habit. In the discussion
of the paper a number of cases were reported by others
present in which Bromidia had been taken without a physician’s
advice and with more or less grave symptoms of
poisoning.
In the report of a death of one who had been a slave
to Bromidia it was said:6 “When the body was found, there
were eleven one-ounce Bromidia bottles about the room or
on his person. Nine were entirely empty and the other two
were about half full. None of these bottles indicated that
they had been purchased on a physician’s prescription, only
the druggist’s label marked ‘Bromidia’ being on them.”
Dr. Horatio C. Wood, Jr., gave7 a striking illustration of
how preparations like Bromidia come to be used even by
physicians without consideration of their constituents:
“Within an hour after his father, a Brooklyn physician, had given
him a dose of bromid, H.G.P., a prodigal son, died yesterday at his
father’s home in Brooklyn. Two years ago, when he appeared to have
sown his wild oats, the father made him superintendent of his country
place, near Grants Mills, Delaware County. A week ago the son left
his place, and at 1 o’clock yesterday morning appeared at his father’s
Brooklyn home. He was nervous, and at 9 a. m. begged for a sedative.
“‘I prescribed the usual quantity of bromidia,’ the young man’s
father told a reporter. ‘He was weak and had suffered from weak heart
and kidney trouble for some time.’
“An hour later the father found the son dying and administered
restoratives, but to no avail.”
A circular, “The Advantages of Bromidia,” makes it plain
how physicians come to use a preparation like Bromidia
without consideration of its potent constituent. In this circular
the presence of chloral is at first frankly admitted,
then it is suggested that in the combination the evil effects
of chloral are completely eliminated and in the end the
impression is left that Bromidia is practically innocuous.
Thus at the beginning while arguing that Bromidia is better
than extemporaneous preparations the chloral content is
plainly acknowledged:
“In the untoward effects so frequently attending the use of extemporaneously
prepared mixtures of chloral and the bromides, may be
found the reason for BROMIDIA’S preference when the need for a hypnotic
agent arises. Were it not for the well known disadvantages of these
drugs which become still more marked with their continued use, there
could be no special need for such a preparation as BROMIDIA (Battle),
for the therapeutic powers of chloral and the bromides are among the
most positive facts in medicine.”
Again:
“It was to meet the growing professional demand for a combination
of chloral and the bromides with their evil effects eliminated, that led
to the manufacture of BROMIDIA (Battle).”
Then, suggesting the indiscriminate use of Bromidia—as
an entity as Dr. Wood suggests—the claim is made that:
“... its constituents have been chosen with a view of enabling
Bromidia to meet every requirement for an agent of its class.”
“Owing to the exceptional purity of its component parts and its freedom
from untoward effects when continued over long periods, this
product will be found of the highest utility in epilepsy.”
“... its action is that of chloral and the bromides minus their
evil effects.”
Finally Bromidia becomes a simple bromid preparation.
Thus an advertisement reads:
“Bromidia’s (Battle) Marked Sedative and Antispasmodic Qualities
eminently fit it for the treatment of Maniacal Excitement, Epilepsy,
Spasmodic Asthma, Convulsive Seizures of Reflex Origin, Sexual Neuroses,
and other disorders attendant upon nervous irritability.
“Through its exhibition, the fullest therapeutic power of the bromides
may be secured with a minimum of their evil effects; a feature of the
greatest service when the necessity for continued treatment becomes
necessary.”
In addition to the general invitation to use Bromidia in
epilepsy and various nervous disorders, a circular also
recommends its use in typhoid, a recommendation, which, if
followed, may turn the scale in favor of a fatal result. The
circular states:
“As a soothing agent in the extreme restlessness and irritability of
typhoid fever and other infectious diseases, BROMIDIA (Battle) is a
therapeutic weapon of definite service. Relief of the headache of
typhoid may also be secured through the use of BROMIDIA (Battle). By
means of its administration for the above purposes, the patient’s
strength is conserved and as a result he is much better prepared to
stand the force of the infection.”
Particularly vicious is the recommendation that it be
given to children. Thus, in a pamphlet entitled “Effective
Drugs Effectively Combined”:
“Another point of advantage to be found in bromidia is the ease
with which it is borne by children. Owing to this tolerance, it is of
distinct service in a considerable list of disorders of childhood. Thus,
of course, employed with care and an understanding of its potency,
bromidia has a field of usefulness in chorea, laryngismus stridulus, and
whooping-cough. In other nervous disorders of childhood—those
attending acute infections, for instance—bromidia is a definitely indicated
therapeutic aid, owing to the soothing influence exerted by even
a moderate dose and the absence of untoward effects. More specifically,
the correcting influence of bromidia in the night-terrors of children
may be mentioned.”
Formerly advertisements asserted that each fluidram of
Bromidia contained:
“Chloral hydrate | 15 | grains |
“Potassium bromid | 15 | grains |
“Extract of Cannabis indica | 1⁄8 | grain |
“Extract of henbane | 1⁄8 | grain” |
This formula also appears on the label of a sample package
sent through the mails during 1914. A recent circular
contains a somewhat different formula. Instead of “1⁄8 gr.
each of gen. Imp. Ext. Cannabis Ind. and Hyoscyam.” as
was formerly claimed, each fluidram of Bromidia is now
said, not to “contain” but to “represent,” not the extracts
but the far less potent drugs “Cannabis indica 1⁄8 grain,
Hyoscyamus 1⁄8 grain,” thus:
“Chloral hydrate | 15 | grains |
“Pot. brom. | 15 | grains |
“Cannabis indica | 1⁄8 | grain |
“Hyoscyamus | 1⁄8 | grain” |
Furnishing still greater variety, the labels on a recently
purchased bottle of Bromidia, where under the Food and
Drugs Act the presence of narcotic drugs must be declared,
read:
“Alcohol 10 per cent., Chloral Hydrate, 91 grs. per ounce. Cannabis
indica indeterminate in finished product.”
“In the manufacture of BROMIDIA to each drachm of fluid used are
added 15 grains of pure chloral hydrate and purified brom. pot., and
1⁄8 grain each of gen. imp. ext. cannabis ind. and hyosciam.”
These various statements as to the composition of Bromidia
leave one very much “in the air.” As chloral and potassium
bromid are easily determined and since lying on the labels
of widely exploited proprietaries has become somewhat
risky recently, it is probable that the statements on the trade
package are to be depended on and that each fluidram of
Bromidia contains something like 12 grains each of chloral
and potassium bromid and not 15 grains as the medical profession
has been and is being told.
Pharmacists who have attempted to put up a nonproprietary
preparation similar to or, more correctly, having
the alleged composition of Bromidia have found it practically
impossible to do so. The reason is that extract of
cannabis indica is almost insoluble in a menstruum such as
that found in Bromidia. The National Formulary, first edition,
listed Mistura Chlorali et Potassii Bromidi Composita
of which it was said: “Each fluidram contains 15 grains
each of Chloral and of Bromid of Potassium, and 1⁄8 grain
each of Extract of Indian Cannabis and Extract of
Hyoscyamus.” In this the pharmacists attempted to incorporate
the cannabis indica by using the tincture of the drug and
suspending it by the addition of tincture of soap bark. In
the present edition of the National Formulary, the preparation
is made by triturating the extract of cannabis indica
with pumice stone and then filtering the finished product.
This gives an “elegant” preparation—but one from which
the cannabis indica is filtered out! A sad commentary on
the National Formulary. It should not be supposed, however,
that the manufacturers of Bromidia have solved the
problem that has baffled the pharmacists; not at all. Bromidia
probably contains no more cannabis indica than does
its National Formulary prototype. The statement on the
present trade packages, that the amount of cannabis indica
in Bromidia is “indeterminate,” is but a tardy acknowledgment
of the fact that the stuff has not, and never had, the
amount of cannabis indica claimed for it for so many years.
The “indications” named on the Bromidia labels are, in
common with nostrums of this type, but suggestions for self-drugging.
They will appeal to the layman who has purchased,
either by prescription or otherwise, an “original
package” of Bromidia and who may imagine he suffers from
“nervousness,” “sleeplessness,” “headache” or “neuralgia.”
But while the manufacturers in their advertising matter
have on the whole not disguised the presence of chloral so
much as they have attempted to make it appear that the
chloral has been robbed of its dangers—for all hypnotics if
used thoughtlessly are dangerous—after all the name has
created the false impression that Bromidia is a bromid
preparation. It is because of this false impression carried
by its name, that Bromidia came to be used indiscriminately
by the profession and in the course of time still more
indiscriminately and recklessly by the public. Bromidia is
a vicious chloral preparation masquerading under a misleading
name. That physicians have been impressed by the
claims of its harmlessness and by the mystery connected with
the formula is not a credit to the intelligence of our profession.
There is no doubt but that physicians are responsible
for the use and abuse of this chloral preparation by the
public.
There is no scientific or rational excuse for a ready-made
preparation of this sort. When chloral or a bromid is indicated
the proper dose of each of these, if they are to be combined,
should be determined for each patient. Potassium
bromid and chloral hydrate both are readily soluble in water,
syrup or elixirs and it is a simple matter to prescribe the
required dose of chloral and of bromid dissolved in some
aromatic water like cinnamon-water (Aqua Cinnamomi), in
some syrup like syrup of orange (Syrupus Aurantii) or in
an elixir like the aromatic elixir (Elixir Aromaticum)
or adjuvant elixir (Elixir Adjuvans). If this mixture is
prescribed thus the physician is alive, alike to the dangers
and the limitations of the drugs; if it is prescribed under a
misleading proprietary name, the physician endangers his
patient, stultifies his profession and tends to perpetuate the
great American fraud.
[Editor’s Note.—A list of some of the medical journals that
advertise Bromidia:
| Texas Medical News | Southern Practitioner |
| Nashville Journal of Medicine & Surgery | New Orleans Medical & Surgical Journal |
| Medical Brief | Therapeutic Gazette |
| Annals of Surgery | Medical Herald |
| Charlotte Medical Journal | Medical Times |
| Medical Sentinel | Texas Medical Journal |
| Memphis Medical Monthly | Wisconsin Medical Recorder |
| Laryngoscope | International Journal of Surgery |
| Medical World | Vermont Medical Monthly |
| Medical Review of Reviews | Atlanta Journal-Record of Medicine |
| Louisville Monthly Journal | St. Paul Medical Journal |
| Indianapolis Medical Journal | Hospital Bulletin of the University of Maryland |
| Monthly Cyclopedia & Medical Bulletin | Denver Medical Times |
| Journal of Nervous & Mental Diseases | Buffalo Medical Journal |
| Maryland Medical Journal | Medical Review |
| Merck’s Archives | Ellingwood’s Therapeutist |
| Iowa Medical Journal | Eclectic Medical Journal |
| Medical Standard | Massachusetts Medical Journal] |
| —(From The Journal A. M. A., May 16, 1914.) |
CACTUS GRANDIFLORUS
Report of the Council on Pharmacy and Chemistry [H]
The Council voted that cactus grandiflorus should not be
accepted for New and Nonofficial Remedies, and that a statement
be prepared for The Journal giving the reasons for
this action. Accordingly the following report has been
adopted by the Council and its publication authorized.
W. A. Puckner, Secretary.
Cactus Grandiflorus
The therapeutic value of this plant has been variously
estimated by different observers. Experimental evidence as
to its action is scanty and no complete chemical examination
has ever been made.
Reputable men have testified that some of the plants of
the cactus family contain very active principles, but so
far experiments seem to prove that cactus grandiflorus has
neither the action of digitalis nor that of strychnin. The
principal contributions, clinical and experimental, for and
against the drug, are set out below.
EXPERIMENTAL EVIDENCE
O. H. Myers8 worked with a product which he calls cactina
and which he regards as the active principle of the drug.
(As no such substance as cactina is described in any materia
medica, it is impossible to state what Myers really used.)
He found that it had a strychnin-like action and raised
the blood-pressure.
Hatcher comes to the conclusion: “Either Myers’ work
was a pure fabrication or he was dealing not with cactin
but with a substance similar to the pellotin of Heffter, the
action of which resembles that of strychnin to a certain
extent.”
E. Boinet and J. Boy-Teissier9 experimented with an
aqueous extract, an alcoholic extract, and with an alkaloid
which they call “cactine.” They concluded from three sets
of experiments on frogs that extract of cactus produces, in
ten minutes, a temporary increase in the heart’s action which
frequently repeated doses are required to maintain; and
that large doses slow the heart and produce arrhythmia.
L. E. Sayre10 experimented with a preparation of cactus,
made from the stem of the plant, by injecting it into the
dorsal lymph space of the frog. There was seemingly an
increase in the amplitude of the heart’s action and an
indication of a strengthened beat or increased force.
R. A. Hatcher11 states that it is possible that cactus grandiflorus,
under certain conditions, may contain a principle with
a strychnin-like action. But Hatcher made ten experiments
on frogs, four on cats, six on dogs, two on rabbits, and one
on a guinea-pig, with Cactina pillets of the Sultan Drug
Company and the Cactin of the Abbott Alkaloidal Company.
From 1 to 15 pillets in frogs and up to 25 in dogs were
used at each dose. In no single instance was there any
evidence of a digitalis-like or strychnin-like action, or, in
fact, of any decided action of any kind whatever.
Gordon Sharp12 was unable to obtain either alkaloid or
glucosid from the plant, but found a series of resins that
caused contraction of the blood-vessels of a frog. This was
not a digitalis-like contraction, but depended, he believed,
on simple acidity. On the heart of the frog the resins have
little or no effect, comparisons being made with digitalis
in the same animals. There is no proof that cactus grandiflorus
itself shortens diastole, or in fact, that it has any
special action on the heart muscle at all. Sharp experimented
on himself with large doses of an extract made
with alcohol 1 to 5, but got no noticeable results. He thinks
that the plant may have some slight diuretic action.
Sayre submitted the preparation which he used in his
experiments for more careful testing to E. M. Houghton,
who reported that it had practically no action on the heart.
In commenting on Houghton’s results, Reid Hunt said
that they were confirmed by his own experiments. He did
not deny, however, that the drug might have some therapeutic
effect and that, in very large doses, it did affect the kidneys.
S. A. Matthews13
found one preparation of cactus (cactin—Abbott)
absolutely inert so far as any effect on the heart
is concerned. He found that cactina (Sultan Drug Co.) in
very large doses depressed both the circulation and respiration.
In this regard it differs from strychnin, and it has
no resemblance to the action of digitalis, strophanthus or
any of the heart stimulants. A dose of from 10 to 12 pillets
administered intravenously to a 10 to 12 kg. dog exerted
little or no influence on the heart or circulation; the larger
dose may cause a slight fall in blood-pressure. When 70 or
more pillets were administered within two and a half hours
the animal generally died.
The work of Boinet and Boy-Teissier also has been criticized
by Hatcher on the ground that their most positive
results were obtained with an alkaloid which no one at
this day is able to prepare. The results quoted in this
report, however, were obtained by the use of extracts of
cactus so that it does not seem that they should be entirely
rejected, whatever their value may be.
CLINICAL EVIDENCE
Clinical observations have been more abundant than exact,
and a favorable action of the drug in some organic diseases
of the heart has been reported; other observers would limit
its use to functional arrhythmia, insisting that it is not a
substitute for digitalis or aconite, but that it occupies a place
distinct from either of those remedies.
P. W. Williams14 recommends cactus for functional heart
disease, but, as a rule, found it useless in organic disease.
He thinks it one of a class of remedies which act on the
accelerator nerves and sympathetic ganglia, shortening the
diastole and stimulating the spinal vasomotor nerve centers.
Williams apparently relied on Myers for his knowledge of
the pharmacologic action, and his paper is a fair example
of the clinical studies of cactus.
Ellingwood15 claims that cactus is a cardiac tonic, acting on
the accelerator nerves and heart ganglia, increasing muscular
force and arterial tension. He recommends it in both organic
and functional diseases.
Boinet and Boy-Teissier found that therapeutic doses of
40 drops of tincture of cactus were without effect on the
normal heart. In patients with noisy asystole (asystolie
bruyante) the same dose produced no appreciable effect. In
the period of latent non-compensation of true cardiac
patients, from 80 to 100 drops a day increased the force
of the failing heart. In patients with secondary heart disease
with arrhythmia of nervous origin, daily doses of 80,
100 and 120 drops of the tincture were well tolerated for
weeks; they seemed to increase the fulness of the pulse
and regulated its rhythm. In spite of such large doses,
these observers never noticed any symptoms that could be
attributed to a cumulative action. It must be remembered
that the precise preparation of cactus which they used is
not known.
Aulde16
recommends it as a cardiac tonic free from cumulative
effects.
Gordon Sharp says: “The therapeutics of the subject,
I think, are clear enough. Cactus grandiflorus cannot be
included in our list of cardiac drugs. It is not even a
simple stomachic tonic and at most all one can say is that
it has small diuretic action.”
Hatcher says: “Clinical testimony is so conflicting that
between the extreme views of Gordon Sharp and those of
Ellingwood there is room for an honest difference of opinion
concerning cactus grandiflorus.”
Matthews himself took 100 granules of cactin (1⁄67 gr.—1 mg.
each), 25 every four hours, without experiencing the
least effect.
CONCLUSIONS
Reliable conclusions regarding the therapeutic use of cactus
grandiflorus are rendered difficult on account of several
factors.
1. It is uncertain what part of the plant contains the
active principle if one exists; and its nature is unknown.
The National Standard Dispensatory states that its “activity
must be confined to the flower in some special stage of its
development or to a certain part of it or to some parts
gathered with it.” This uncertainty may explain the negative
results obtained by some observers, but it makes the
drug one that cannot be generally relied on and gives an
excellent opportunity for the exploitation of proprietary
preparations.
2. Some of the experimental work and much of the clinical
evidence has been obtained and published under proprietary
auspices. For this reason many of the therapeutic claims
made for the drug must be viewed as merely the reflection
of the exaggerated statements made by the advertisers of
proprietary preparations.
3. The value of clinical evidence when unsupported by an
animal experimentation is much diminished by the tendency
of enthusiastic and untrained observers to attribute to the
drug given the effect really due to general remedial measures,
psychic suggestion and so forth. While it must be admitted
that valuable remedies may exist whose therapeutic properties
cannot be revealed by animal experimentation, yet
in the absence of such experimental evidence conclusions
should be drawn with extreme caution.
Bearing these conditions in mind, the following statements
seem to be justified: (a) The botanical, chemical and pharmaceutical
properties of cactus are not sufficiently determined
to make any available preparation a reliable remedy.
(b) There is some evidence that cactus may be capable of
affecting the animal heart and nervous system, but its action
is not that ordinarily attributed to it. It does not increase
the force of the heart-beat. (c) While there is some clinical
testimony as to its usefulness in functional diseases of the
heart, the indications for its administration are at present
too uncertain to afford a safe basis for recommending it.
4. While the drug may be deserving of further experimental
and clinical investigation, this should be carried on
in reliable pharmacologic laboratories and in clinics provided
with facilities for exact observation.—(From The
Journal A. M. A., March 12, 1910.)
CALCREOSE
Report of the Council on Pharmacy and Chemistry
In response to inquiries and in view of the extensive
advertising propaganda, the Council, on Dec. 19, 1913, took
up for consideration Calcreose (Maltbie Chemical Company,
Newark, N. J.). Examination showed that the preparation
contained, in loose combination, approximately equal weights
of creosote and lime. The claims made in the advertising
“literature” were extravagant and uncritical, and the Council
therefore held Calcreose ineligible for New and Nonofficial
Remedies.
In June, 1914, at the request of the Maltbie Chemical Company,
the Council undertook a reconsideration of the preparation.
The advertising claims were now found more conservative.
Before the existing claims could be judged, however,
the Council deemed it necessary to require from the company
satisfactory proof (1) that the large doses of Calcreose
recommended and administered actually furnish large
amounts of creosote to the blood, and (2) that patients taking
these large doses do not suffer from digestive disturbances,
loss of nutrition, albumin in the urine or phenol urine, as
claimed. The Council accordingly advised the company of
this requirement, at the same time stipulating that nothing
in the report should be interpreted as indicating a belief on
the part of the Council that enormous doses of creosote are
necessary for, or will promote a cure of tuberculosis.
The Maltbie Chemical Company has not up to the present
date furnished this proof, but has evinced a disposition to
make the Council’s holding Calcreose under advisement
appear in the guise of a quasi-approval. It is therefore
recommended that Calcreose be refused recognition for conflict
with Rule 6.—(From the Journal A. M. A., June 26, 1915.)
CAMPHO-PHENIQUE
Report of the Council on Pharmacy and Chemistry and
Some Comments Thereon
The following report was submitted to the Council on
Pharmacy and Chemistry by the subcommittee to which
Campho-Phenique had been assigned:
To the Council on Pharmacy and Chemistry:—Campho-Phenique,
sold by the Campho-Phenique Co., St. Louis, Mo.,
is claimed to be composed of phenol 49 per cent., and camphor
51 per cent.
Examination of specimens, purchased in the open market,
made under our direction, demonstrates that the statements
made in regard to the composition are not true. Instead of
containing 49 per cent. of phenol (carbolic acid), the analysis
showed that it contains not more than 20 per cent. Instead
of containing 51 per cent. of camphor, the analysis demonstrates
that the amount of camphor is not more than 38 per
cent. Besides phenol and camphor, a third substance was
found which proved to be liquid petrolatum and to be present
to the extent of 38 per cent. or more.
Since the statements made in regard to the composition
of Campho-Phenique are deliberate misrepresentations of
the facts, it is recommended that the article be not approved.
Besides Campho-Phenique, the above-mentioned firm also
sells a preparation labeled Campho-Phenique Powder. While
no statement in regard to the composition of this product is
made on the label or in the literature, such expressions as
“Campho-Phenique in a powdered form” and “Powdered
Campho-Phenique” lead to the inference that it has essentially
the same composition as that stated for the liquid
preparation. An examination of a specimen of Campho-Phenique
Powder purchased in the open market showed that
92 per cent. of it was a talcum-like inorganic substance.
The remaining 8 per cent. consisted chiefly of camphor with
a small amount of phenol.
In view of the fact that Campho-Phenique Powder contains
very little phenol, but instead consists chiefly of an
inorganic talcum-like substance, its name is misleading and
deceptive. It having been shown that Campho-Phenique
Powder corresponds to a camphorated talcum powder, the
claims that it “has no equal as a dry dressing,” that it is
“absolutely superior to iodoform,” and that it has “all the
excellent properties of aristol and iodoform,” are unwarranted.
It is recommended that the article be not approved,
and that this report be published.
The recommendations of the subcommittee were adopted
by the Council, and in accordance therewith the above report
is published.
W. A. Puckner, Secretary.
Campho-Phenique
The above report on a much advertised “ethical” proprietary
medicine is worthy of the thoughtful consideration
of the members of the medical profession, as it illustrates
admirably some of the conditions connected with this proprietary
medicine business.
THE FORMULA A FAKE
First, it illustrates the fact that the published formulas
of the “ethical” proprietaries are not always reliable. The
Campho-Phenique Company has been very willing to give
out a formula, purporting their product to be 51 per cent.
camphor and 49 per cent. phenol (carbolic acid). Now,
these two drugs will make a liquid mixture, and any druggist
can make it, and the mixture will have about the same
consistency and appearance as Campho-Phenique. But its
effect differs decidedly from that of Campho-Phenique. Some
months ago a very intelligent physician, in discussing the
proprietary medicine business, said that in some cases physicians
could not get druggists to make preparations which
were as satisfactory as those which could be bought ready-made.
He cited Campho-Phenique as an illustration. He
said that he had used this preparation for burns, etc., but
as he did not like to use preparations put up by companies
about which he knew nothing, he asked his druggist to
make the mixture in accordance with the published formula.
The druggist’s preparation was not satisfactory; it had a
decidedly different effect from Campho-Phenique, and so
he tried another druggist. This druggist also followed the
published formula, but his results, too, differed materially
from the proprietary article.
The various analyses that have been made show why the
preparations put up by the druggists did not resemble that
made by the company; since, according to the analyses,
Campho-Phenique consists of 40 per cent. liquid petrolatum,
which is an inert but soothing diluent, while instead of
49 per cent. of carbolic acid, as claimed, it really contains
less than 20 per cent. This is an entirely different proposition.
Now, if the physician referred to above will have
his druggist make a mixture of 20 per cent. of carbolic
acid, 40 per cent. of camphor and 40 per cent. of liquid
petrolatum, and will then compare this resulting compound
with Campho-Phenique, he will find that there is not much
difference. Furthermore, he will realize that there is nothing
either new or wonderful about the preparation. Camphorated
oil and carbolized oil are both in common use.
Campho-Phenique is apparently simply a mixture of the two.
THE POWDER STILL WORSE
So much for the liquid. The powder seems to be something
entirely different, for, according to the chemist’s report,
over 90 per cent. of it is inert, absorbent, talcum-like
material. There is enough camphor and carbolic acid to
give the powder an odor and thus mislead physicians,
especially those who are in the habit of taking for granted
that whatever statements nostrum manufacturers make are
true. Perhaps it is a fairly good dressing for wounds—at
least it will do no harm—but its name is misleading and
deceptive. For all practical purposes it is essentially a
camphorated talcum powder.
COMPANY A “PATENT-MEDICINE” CONCERN
The second interesting phase of this “ethical” proprietary
is that it illustrates another point, i. e., that many of these
articles are supplied to our profession by those who are not
legitimate manufacturing pharmacists. The Campho-Phenique
Company of St. Louis, according to all reports, is owned
and controlled by a gentleman named Ballard. This “company”
supplies the medical profession with the preparations
under consideration and also with Chloro-Phenique and
Scrofonol. We are informed that this same Mr. Ballard
is the principal owner, if not the sole owner, of quite a
number of “patent-medicine” companies, such as Ballard-Snow
Liniment Co., Brown’s Iron Bitters Co., Mayfield
Medicine Mfg. Co., Smith Bile Beans Co., Swain’s Laboratory,
and several others. We learn from the wholesale drug
trade lists that these various “companies” make and sell,
besides the Campho-Phenique preparations, Ballard-Snow
Liniment, Ballard’s Herbine, Brown’s Iron Bitters, Dr.
Herrick’s Pills, Richardson’s Life-Preserving Bitters, Smith’s
Bile Beans, Swain’s All Healing Ointment, and several
other “patent medicines.”
It is hardly necessary to make any further comments. The
whole business is nauseating to those who know the actual
conditions of this nostrum business and how our profession
is being deluded. The Campho-Phenique matter is not
an exception; it is simply another illustration of these
conditions.
The majority of “ethical” proprietaries are foisted on our
profession, either without any formula accompanying them,
or with a “formula” that is a fake. The majority of the
“ethical” proprietaries are manufactured and supplied to
physicians, with instructions regarding their use, by men
who bear the same relation to legitimate pharmacy that
the veriest quack that ever swindled a credulous public
bears to scientific medicine.—(From The Journal A. M. A.,
April 20, 1907.)
CELERINA, ALETRIS CORDIAL AND KENNEDY’S
PINUS CANADENSIS, LIGHT AND DARK
Report of the Council on Pharmacy and Chemistry
The following reports on products of the Rio Chemical
Company have been submitted by a referee. The Council
recommends that they be published, as the preparations
discussed are glaring instances of nostrums exploited through
physicians on unscientific claims and false representations.
W. A. Puckner, Secretary.
Celerina
Celerina belongs to what Samuel Hopkins Adams calls
the “bracer” type of nostrum. According to the label it contains
42 per cent. alcohol (whisky contains about 50 per cent.).
The other ingredients of Celerina are declared to be as
follows:
“Each fluidounce represents Forty grains each Kola, Viburnum,
Forty-eight grains Celery, Twenty grains Cypripedium, Sixteen grains
Xanthoxylum and Aromatics.
“Dose—1 or 2 teaspoonfuls 3 times a day.”
Kola contains a very small percentage each of caffein and
theobromin. It is impossible for the infinitesimal amounts
of these alkaloids in an ordinary dose of Celerina to produce
any physiologic effect.
Viburnum has been called a “uterine sedative,” whatever
that may be. Its only real activity is the psychic one due to
its taste and odor.
Celery at one time was credited with being both an antispasmodic
and a nerve stimulant—a remarkable combination
of opposing qualities! Scientific investigation has failed to
show that celery has any physiologic or therapeutic activities.
If it had the slightest medicinal value, the rational course
would be to prescribe it in its fresh and natural state. The
small dose contained in a teaspoonful of Celerina is inappreciable
and not even equivalent to that contained in a stalk of
celery.
Ladyslipper, more imposing under the Latin name of
“cypripedium,” is a flowering plant with a legendary reputation
as an “antispasmodic and nerve stimulant.” It has
been in the therapeutic scrap-heap for years. It contains a
little tannic acid, gallic acid and a volatile oil. Even a
tannic acid action cannot be expected from a teaspoonful of
a preparation containing 20 grains of ladyslipper to the
ounce.
Prickly ash (xanthoxylum) has never been shown to have
any activity other than that of a local irritant, especially to
mucous membranes. The slight “bite” from this drug would
be entirely covered up by the alcohol in Celerina. Any stimulating
effect which this drug may have on the stomach is
greatly inferior to that produced by a very small glass of
ordinary ginger ale.
In short, there is no ingredient in Celerina, except the
alcohol, that has any recognizable activity; and the alcohol
content is nearly as great as that of ordinary whisky. Some
of the claims and recommendations for this nostrum are:
“Celerina (Nerve Tonic), for Nervousness, Hysteria, Insomnia,
Nervous Indigestion, Languid and Debilitated Conditions, Recovery
from Alcoholic Excess.”
Think of prescribing an alcoholic nostrum four times a day
to promote recovery from alcoholic excess!
“NEURASTHENIA: The bane of the general practitioner; the puzzle
of the neurologist; the juicy fruit of the quack and faddist; the opportunity
of the intelligent therapist.... For the medical treatment
CELERINA is the preparation of wide utility.”
The sang froid with which the exploiters of this nostrum
refer to other “quacks and faddists” as reaping “juicy fruit”
from neurasthenics would command admiration were it not
so pitiful.
“Celerina has substantial endorsement in nervous disorders characterized
by Aphonia (nervous).”
Of course, the disappearance of nervous aphonia might
follow the application of any treatment whatever, be it Eddyism,
Chiropractic, Peruna or Celerina.
In
“CLIMACTERIC (the Menopause) flattering results have been
reported from a combination of equal parts Celerina and Aletris Cordial
Rio.”
“Teaspoonful doses after meals and upon retiring have proven efficacious
[in “dyspepsia”] when other remedies have failed.”
Here is a good example of proprietary-house therapeutics:
Such widely different conditions as digestive trouble and the
climacteric are to be treated with a combination of alcohol,
simple bitters and aromatics! Why not order a cocktail
under its own name? It would be equally efficacious, less
mysterious and its dangers might be better realized!
“A teaspoonful or two in three tablespoonfuls of boiling hot water
[for insomnia] upon retiring.”
Any other hot toddy at bedtime (and it need not cost
a dollar a bottle) might give relief; but the intelligent physician
to-day recognizes the danger of prescribing alcohol in
such conditions.
“In the case of brain workers who suffer from nervous excitability
and mental fatigue, the administration of Celerina in teaspoonful doses,
three times a day and at bedtime, rapidly controls the condition and
increases mental capacity.”
And the same effect follows its use:
“In cases involving worry, anxiety, overwork, and excesses of various
kinds....”
Moreover:
“Celerina is the most prompt and efficient of remedies for devitalized
or broken-down constitutions—doses four times a day.”
The statement made by its manufacturers that this preparation
is free from narcotics or habit-forming drugs is not
true. Alcohol is both a narcotic and a habit-forming drug.
As in the case of other nostrums containing no potent drugs
but alcohol, Celerina is recommended for various diseased
conditions in combination with a familiar form of treatment
by drugs of more or less value. The physician who thoughtlessly
prescribes one of these combinations will without
doubt unthinkingly attribute any subsequent improvement to
the Celerina. Thus, for malaria, a prescription of quinin and
Celerina is advised; for chorea in children, arsenic with
Celerina; in “Convalescence from La Grippe,” strychnin
sulphate, Fowler’s solution, and Celerina; for impotence, nux
vomica, dilute phosphoric acid and Celerina. In none of
these conditions would Celerina affect favorably anything
except the pockets of the exploiters; in some, as in the chorea
of children, the alcohol would be positively detrimental. Of
course, the value of such prescriptions (so far as they have
any apart from the fictitious value lent by the alcohol) resides
altogether in the standard drugs prescribed with Celerina.
There is no possible excuse for writing a prescription for
Celerina, either in original package or mixed with well-known
or valuable drugs. The sooner it is realized that this preparation
has no place in medicine, should never be prescribed
by physicians and is essentially nothing but alcohol and bitters
exploited under a fancy name, the better for the public
health and the science of medicine. The continued sale and
use of Celerina is a disgrace to the medical profession.
Aletris Cordial
Aletris Cordial is a nostrum containing therapeutically
worthless drugs in alcohol (28 per cent.).
The “formula” on the label reads:
“Each fluidounce represents ten grains Aletris, thirty grains Helonias
and thirty grains Scrophularia.”
At one time these drugs had some vogue, chiefly as
domestic remedies. They have been discarded as valueless
by modern scientific medicine.
Aletris, or unicorn root (Aletris farinosa), contains a bitter
principle and starch. The remarkable uterine tonic properties
formerly ascribed to it have not been confirmed by
reliable observers. It is practically worthless.17
Helonias, or false unicorn (Chamaelirium luteum), is
asserted to be a hemostatic and uterine tonic. No trustworthy
evidence has ever been offered in support of the
claims made for this drug; reliable medical literature contains
no reference to it; it has no valid claim on the attention
of physicians.18
Scrophularia, or figwort (Scrophularia marilandica), contains
a principle which has a digitalis-like action on the
heart. Its activity is so slight in comparison with that of
digitalis, however, that there was nothing to be gained by
studying it. The drug is consequently little known and is
not mentioned in critical works on pharmacology. If the
drug were therapeutically active in the quantities used,
another danger would be added to that of the alcohol content
of Aletris Cordial. Since the recommended dose (a teaspoonful)
contains, if the formula be correct, only about 4
grains of figwort, this drug too may be regarded as practically
inert in this preparation.
Not one of these drugs has been deemed worthy of mention
in the Pharmacopeia. The Council has previously discussed
them and declared them valueless (Reports Council
Pharm. and Chem., 1909, p. 146; 1910, p. 10; 1912, p. 42).
In Aletris Cordial, then, there is no ingredient capable of
producing any other effect than the alcohol stimulation and
such psychic effect as may be due to the bitter taste. Yet
physicians are asked to believe that
“Probably no remedy is so uniformly successful in the prevention
of threatened miscarriage as ALETRIS CORDIAL Rio.”
“HABITUAL MISCARRIAGE can be effectually overcome by the
systematic use of Aletris Cordial Rio.”
“... regulates the local circulation and imparts normal tone and
strength to the uterine muscle.”
“The use of Aletris Cordial Rio throughout pregnancy goes far to
assure normal, uncomplicated labor.”
Such claims as these, when made for a mixture containing
no therapeutically active constituent except alcohol, are
absolutely preposterous. It should be noted that the declared
alcohol content of Aletris Cordial is much higher than that
of the strongest wines, and, in the light of medical experience,
quite high enough to promote the formation of the
alcohol habit in a steady user. The following recommendation,
taken from the company’s “Budding into Womanhood”
circular, therefore, is outrageous:
“Many medical practitioners recommend to mothers the use of
Aletris Cordial Rio for their growing daughters, ranging in age from
twelve to eighteen years....”
It is to be hoped that no medical practitioner is so heedless
of consequences as to prescribe for adolescent girls a
worthless nostrum capable of creating a craving for alcohol.
The temperance societies might with profit take steps to
inform laymen, especially women, concerning the worthlessness
of this nostrum, the risk involved in taking it, and the
outrageous character of the recommendations made for it by
the manufacturers.
Kennedy’s Pinus Canadensis, Light and Dark
(Abican and Darpin)
Kennedy’s Pinus Canadensis, Light (recently renamed
“Abican”) and Dark (renamed “Darpin”) are also exploited
by the Rio Chemical Company. Although they have been on
the market some thirty or forty years they appear to have
achieved no marked degree of commercial success. Yet
they have been imitated by most of the pharmaceutic houses.
They are of interest chiefly through the barefaced fraud
involved in their exploitation.
COMPOSITION CLAIMED
Apparently the dark preparation (“Darpin”) was first put
on the market; then the light one (“Abican”) was offered, to
be used only “as an injection and externally.” The reason
for the existence of the light preparation evidently was the
objectionable property of the dark, which stained linen. The
two preparations are both said to be extracts of Pinus Canadensis
or hemlock bark. A circular issued some years ago
contained the following statement:
“Pinus Can. (Ken.)—Dark—A non-alcoholic extract of Pinus Canadensis,
to each fluidounce of which is added 0.48 grains Thymol.
“Pinus Can. (Ken.)—Light—A non-alcoholic extract of Pinus Canadensis,
to each fluidounce of which is added 24 grains each of pure
Alum Potash and Sulphate of Zinc and 0.48 grains of Thymol.”
The labels on the packages of the light and dark preparations
sent out to-day bear, respectively, only the following
references to composition, the first on the dark and the second
on the light:
“Each fluidounce also contains 0.48 grains Thymol.”
“A non-alcoholic preparation of Pinus Canadensis, to which is added
twenty-four grains each pure alum potash and sulphate of zinc and 0.48
grains thymol to the fluidounce.”
ACTUAL COMPOSITION
“Darpin” or Kennedy’s Pinus Canadensis, Dark, does contain
tannin, but, as the simplest of chemical tests demonstrate,
Pinus Canadensis, Light, does not contain tannin. It
might as truthfully be called maple syrup or beef tea.
It is almost a work of supererogation to discuss the therapeutic
claims made for preparations sold under false pretenses
as to composition. It is enough to mention that Kennedy’s
Pinus Canadensis, Dark or Light, is recommended in
| Albuminuria | Hemorrhage from the Nose |
| Diarrhea-Dysentery | Uterine Hemorrhage |
| Fetid Perspiration | Leucorrhea |
| Endometritis | Nasal and Pharyngeal Catarrh |
| Fissures | Piles |
| Fistula | Sore Throat |
| Gonorrhea | Ulceration of the Cervix |
The intelligent physician of to-day knows that his forefathers
in the days of the stage-coach employed tannic acid
in its crude form and treated intestinal disease in a very
unsatisfactory manner; he knows, further, that advances in
our knowledge of pathology have rendered the use of tannic
acid in gastro-intestinal therapeutics largely unnecessary and
that when it is used it should be in some form that will pass
the stomach unchanged. So far as its use as local application
is concerned, he knows, without need of instruction from
the Rio Chemical Company, when tannin is indicated, and
the Pharmacopeia furnishes a suitable preparation for the
physician so that he need not resort to an unscientific nostrum
like Darpin.
The physician who is competent to treat a case of gonorrhea
does not need to be told that alum and zinc sulphate
may be useful in such conditions, and he does not want them
palmed off on him for something else under the name of
Pinus Canadensis, Light, Abican or what not. Also, he prefers
to use them, when they are needed, singly and in
strength suited to the conditions of the individual case.
[Editorial Comment.—Celerina, Aletris Cordial and Kennedy’s
Pinus Canadensis, Light and Dark, appear to be the
entire output of the Rio Chemical Company, which was one
of the earliest of the various companies organized by James J.
Lawrence of Medical Brief fame. The business was moved
from St. Louis to New York City in 1901. According to
what we believe to be reliable information, the Rio Chemical
Company is now composed of James P. Dawson, president;
William W. Conley, vice-president and treasurer; and E. D.
Pinkerton, secretary. These also constitute the directors.
It appears that James P. Dawson is a member of the law
firm of Dawson and Garven, St. Louis; E. D. Pinkerton is
said to be Miss Effie D. Pinkerton, stenographer for Dawson
and Garven. We know little concerning William W. Conley
except that he appears to be in charge of the establishment
in New York. We find no evidence that he is either a
chemist or a pharmacist; his name does not appear in the
membership list of the American Chemical Society or of
the American Pharmaceutical Association, nor can we discover
that he has published anything in the way of chemistry
or pharmacy. As a matter of fact, the Rio Chemical Company
is another of the pseudo-chemical companies created to
exploit one or more proprietaries—in this instance Celerina,
Aletris Cordial and Pinus Canadensis. The following medical
journals carry advertisements of the Rio products (or
did late in 1914): American Journal of Surgery, American
Medicine, Denver Medical Times and Utah Medical Journal,
Eclectic Medical Journal, International Journal of Surgery,
Interstate Medical Journal, Massachusetts Medical Journal,
Medical Brief, Medical Century, Medical Council, Medical
Review of Reviews, Medical Sentinel, Medical Standard,
Texas Medical Journal and Woman’s Medical Journal.].—(From
The Journal A. M. A., Feb. 13, 1915.)
CINERARIA MARITIMA
Report of the Council on Pharmacy and Chemistry
Occasional inquiries in regard to the therapeutic value of
Cineraria maritima caused the Council to consider the drug
with reference to its fitness for inclusion in N. N. R. among
non-official, non-proprietary remedies. The following report,
having been submitted to the Council by a subcommittee,
was adopted and its publication authorized.
W. A. Puckner, Secretary.
To the Council:—The juice of a plant referred to as
Cineraria maritima was at one time supposed to be of value
in the treatment of cataract and certain other affections of
the eye. No scientific evidence is available to show that
the drug is therapeutically active, and its value is no doubt
correctly estimated by Dr. Casey Wood, who (“Ophthalmic
Therapeutics,” p. 446; Cleveland Press, Chicago, 1909) says:
“Still, a few respectable names have been associated with its
[Cineraria maritima] employment in that capacity and it only remains
to be said that the instillation into the conjunctival sac of a preparation
of this or any other member of the Senecio family has about as
much effect on the resolution or dispersal of opacities due to organic
changes in the lens as pouring the same down the back of the patient’s
neck!”
The plant from which Cineraria maritima juice is claimed
to be prepared is commonly referred to in literature as
Cineraria maritima, but is more correctly described as
Senecio cineraria, D. C.
It may be considered a matter of indifference whether a
remedy like this be advertised for the treatment of such
diseases as cataract, providing its application could do no
harm, but it must be remembered that it is recommended
also for other diseases of the eye in which its use, by
postponing efficient treatment, would be the means of serious
damage or even loss of vision.
Since there is no evidence to show that this drug is of
any therapeutic value, it is recommended that it be not
admitted to the list of non-official, non-proprietary remedies
in N. N. R., and that the Council formally expresses its
opinion that the drug, as judged by the evidence which is
available, is without value in the treatment of cataract or
similar diseases of the eye.
[Editorial Comment.—Cineraria maritima would long since
have been relegated to the limbo of discarded and discredited
drugs had it not been given a semiproprietary character by a
St. Louis nostrum house—the Walker Pharmacal Company—which,
like the Manola Chemical Company, is, we understand,
practically a subsidiary concern of the Luyties Homeopathic
Pharmacy Company. The Walker concern exploits
this drug under the name Succus Cineraria Maritima
(Walker). Its method of exploitation consists in publishing
testimonials, which it dignifies with the name “clinical
reports,” from men whom it designates as “representative
physicians.” As indicative of what constitutes representative
physicians, we find that of the seven testimonials given in
their pamphlet the names of three of the signers are not to
be found in any medical directory.
The exploitation of Succus Cineraria Maritima (Walker)
is the oft-repeated story of the resurrection of discarded
and worthless drugs for the purpose of creating proprietorship
in a nostrum. Cineraria maritima is worthless; its
therapeutic value is nil. By the prodigal use of printers’
ink, the medical profession—and through it the public—has
been humbugged into believing that it possesses curative
value.]—(From The Journal A. M. A., Nov. 11, 1911.)
HAGEE’S CORDIAL OF THE EXTRACT OF COD LIVER OIL COMPOUND [I]
Report of the Council on Pharmacy and Chemistry
This is one of the “oilless” cod liver cordials. Like other
manufacturers of such extracts, the Katharmon Chemical
Company, St. Louis, which owns Hagee’s Cordial, attempts
to trade on the reputation long enjoyed by cod liver oil as
a promoter of growth and nutrition. The following is the
statement of composition furnished by the company:
“Each fluid ounce of Hagee’s Cordial of the extract of Cod Liver Oil
Compound represents the extract obtainable from 1⁄8 fluid ounce of
Cod Liver Oil (the fatty portion being eliminated), 6 grs. Calcium
Hypophosphite, 3 grs. Sodium Hypophosphite, 1⁄2 gr. Salicylic Acid
(made from Oil Wintergreen), with Glycerin and Aromatics.”
And here are some of the therapeutic claims:
“Tonic, Stimulant, Alterative, Reconstructive, Nutritive and Digestive.”
“Useful in phthisis pulmonalis, scrofula and all chronic pectoral complaints,
coughs, colds, brain exhaustion, nervous debility, palsy, chronic
cutaneous eruptions and impaired digestion.”
Of course, these absurd claims hark back to the time of
the prevalence of the now discarded theory that the valuable
properties of cod liver oil reside, not in the fat, but in certain
nitrogenous, alkaloid-like constituents present in
infinitesimal amounts. Further “playing up” this theory:
“The prescriber may know that in our preparation he is getting, in
easily assimilable and palatable form, the very properties that make
cod liver oil the best of reconstructives.
“When you prescribe cod liver oil you are after the active principles—why
not give the active principles themselves.”
Proprietary manufacturers usually ignore scientific investigations
which establish facts adverse to proprietary claims;
but the same proprietary manufacturers are quick to seize
on any theory that can be twisted into support of their interests.
Thus, recent investigations having shown that cod
liver oil, like butter and egg yolk, possesses certain growth-promoting
properties not found in some other fats, the promoters
of Hagee’s Cordial claim these properties of cod liver
oil for their extract. They assert:
“Recent Chemical Investigations of Cod Liver Oil show that the active
principles contain the nutritive qualities attributed to the whole oil.”
The Council has previously expressed the opinion19 that
the preponderance of evidence indicates that whatever therapeutic
value cod liver oil may have depends chiefly, if not
entirely, on its fat (oil). There never was any evidence or
scientific authority for the theory that the therapeutic value
of cod liver oil was independent of its fat content. The fact
that the fat is the growth-promoting element has already
been shown, and J. P. Street, chemist for the Connecticut
Agricultural Experiment Station (The Journal A. M. A.,
Feb. 20, 1915, p. 638), in a series of experiments on a number
of the so-called extracts of cod liver or cod liver oil (including
Hagee’s Cordial) has conclusively demonstrated that the
growth-promoting properties of the oil are not to be found in
the extracts. Street placed rats on a ration not sufficient to
maintain normal nutrition and growth for an extended period.
After the rats had been on this ration for some time and a
failure to maintain weight was indicated, an amount of
dealcoholized Hagee’s Cordial was substituted for a portion
of the lard contained in the ration. Later Hagee’s Cordial
was replaced by cod liver oil.
Street says:
“None of the four rats did well on Hagee’s Cordial; in fact, they
lost 1.2 to 15.4 gm. during feeding periods of from seven to fourteen
days.”
“The rats failed so quickly when put on Hagee’s Cordial that in two
cases the animals did not recover even when put on the full cod liver
oil ration.”
“... the four rats during the Hagee period, instead of gaining
the normal 24 gm., actually lost 36.2 gm., while during the cod liver oil
period instead of gaining 114 gm., they gained 156.4 gm.”
“The inferiority of Hagee’s Cordial as a reconstructive and a nutrient
compared with ordinary cod liver oil is apparent.”
Hagee’s Cordial of the Extract of Cod Liver Oil Compound
has neither the nutritive qualities nor the reconstructive
efficacy of cod liver oil. This mixture is worthless for
the conditions for which it is advertised, and is marketed
under misleading and unwarranted claims. It is recommended
that Hagee’s Cordial be held ineligible for New and
Nonofficial Remedies.—(From The Journal A. M. A., April
10, 1915.)
WAMPOLE’S PERFECTED AND TASTELESS PREPARATION OF AN EXTRACT OF COD LIVER [J]
Report of the Council on Pharmacy and Chemistry
Wampole’s Preparation is another of the oil-free “extracts”
of cod liver. The following formula (which, be it observed,
is non-quantitative and therefore practically worthless) is
published by the owners, Henry K. Wampole & Co., Inc.:
“Contains a solution of an extractive obtainable from fresh cod livers,
the oily or fatty portion being afterward eliminated. This extractive
is combined with Liquid Extract of Malt, Fluid Extract of Wild Cherry
and Compound Syrup of Hypophosphites (containing Calcium, Sodium,
Potassium, Iron, Manganese, Quinin and Strychnin).”
An alcohol content of 17 per cent. is declared on the label.
The following claims are typical of those made for the
preparation:
“This grease, or oil, is not present in Wampole’s Preparation of the
Extract, which is palatable and, at the same time, very efficient as a
stimulant to the centers of nutrition and assimilation. It is unsurpassed
as a reconstructive tonic ...”
“[Cases] with a marked tendency to pulmonary troubles,... if
a timely impulse be given them will easily shake off the impending evil.
Wampole’s Preparation gives that timely impulse ...”
In the Council’s opinion, as previously expressed,20 such
therapeutic value as there may be in cod liver oil is chiefly,
if not altogether, due to the fat (oil). Lately, the investigations
of J. P. Street of the Connecticut Agricultural Experiment
Station have definitely disproved the claims made for
the Wampole’s and similar preparations. In Street’s experiments,
rats were placed on a ration insufficient for normal
nutrition and growth. After the rats had been on the ration
for a time long enough for inability to maintain weight to
become evident, dealcoholized Wampole preparation was
substituted for a portion of the lard contained in the ration.
Later the Wampole preparation was replaced by cod liver
oil. From these experiments it appears that, although the
Wampole preparation is said to contain malt extract and
sugar, it does not show the advantage over ordinary cod liver
oil as a source of nutriment which is claimed for it by the
manufacturers. Street emphasizes that the Wampole preparation
does not possess to any marked degree the reconstructive
properties of cod liver oil, butter fat and egg yolk,
on which foods rats gain weight rapidly and steadily after
having been on a deficient diet. Street calls attention to the
fact that the amount of alcohol consumed daily by the user
of the Wampole preparation (the equivalent of 0.7 fluidounces
of whiskey) explains to a considerable extent the
asserted tonic virtues of the preparation.
Though offered as an efficient substitute for cod liver oil,
Wampole’s “Perfected and Tasteless Preparation of an
Extract of Cod Liver” lacks both the nutritive and the reconstructive
properties and is marketed under an indefinite
name and unwarranted and untrue claims. It is recommended
that Wampole’s Preparation be held ineligible for
New and Nonofficial Remedies.—(From The Journal A. M. A.,
April 10, 1915.)
WATERBURY’S METABOLIZED COD-LIVER OIL
COMPOUND [K]
Report of the Council on Pharmacy and Chemistry and
Laboratory Contribution on Which It Is Based
The following report has been adopted by the Council and
its publication directed
W. A. Puckner, Secretary.
To the Council:—Your committee on pharmacology has
read with interest the contribution from the Association’s
laboratory on Waterbury’s Metabolized Cod-Liver Oil Compound.
The report shows that misleading and false statements
are made in regard to the composition of the product
and also that exaggerated and unwarranted claims are made
for its therapeutic value. In view of the attempt of the
Waterbury Chemical Co. to create a false impression in
regard to the therapeutic value of the composition of its
product, it is recommended that the following report be
adopted and published:
The Council believes that there is a preponderance of evidence
to indicate that whatever therapeutic value cod-liver
oil has, that value depends chiefly, if not entirely, on its
fat (oil). In the opinion of the Council, the word cod-liver
oil should not be used in connection with any preparation
unless it consists to a large extent (25 per cent. or more)
of cod-liver oil. Since Waterbury’s Metabolized Cod-Liver
Oil Compound contains no appreciable quantity of cod-liver
oil, the name is incorrect and misleading, and as a cod-liver
oil preparation it is believed to be wholly valueless. The
Council has previously voted that Waterbury’s Cod-Liver
Oil Compound be refused recognition because of conflict with
Rules 1 and 6.—(From The Journal A. M. A., Oct. 9, 1909.)
[Contribution from the Chemical Laboratory of the
American Medical Association]
Waterbury’s Metabolized Cod-Liver Oil Compound
W. A. Puckner and L. E. Warren
A full page advertisement of Waterbury’s Metabolized
Cod-Liver Oil Compound appeared in the Iowa Medical
Journal, March 15, 1909, in the form of a letter purporting
to give the results of an analysis of the product made for
the firm by a Chicago chemist. In this letter-advertisement
the chemist states at the outset that the results of his
examination “are somewhat at variance with the statements
made in The Journal.” These statements he quotes as
follows:
1. It is a clear liquid and no globules of oil are seen under the
microscope. It is therefore not an emulsion.
2. It is of acid reaction when mixed with water and remains clear
when strongly acidified. Hence it does not contain a soap, and is not
a saponification of fat.
3. It mixes with water without precipitation, hence, it can not
contain more than traces of a fatty acid.
The chemist admits in his letter to the firm that his
analyses verify statements 1 and 3, but regarding statement
2 he says: “I find that your preparation is acid in reaction,
but when strongly acidified gives a distinct turbidity within
10 minutes and a voluminous precipitate within 1 hour. This
precipitate is shown to consist of fatty acids of cod-liver
oil, which are thrown down by the splitting of the soaps, on
acidifying either with sulphuric or hydrochloric acid.” From
these results he states that to him it seems that the “preparation
does not deserve the statement that it contains no
soap, as there is no question whatever of the presence of
cod-liver oil.”
While in the letter published in this advertisement the
chemist claims to have demonstrated the presence in the
product of “saponified cod-liver oil,” he omits to mention
the quantities of the soap present. In the article that originally
appeared in The Journal (Oct. 13, 1906), in addition
to the three paragraphs quoted by the chemist, the following
statements were made:
“By these simple tests a physician is easily able to demonstrate
that the preparation does not contain cod-liver oil. It
is therefore valueless for the purpose of nutrition for which
we give the oil. More careful analysis confirms the results
of these tests and shows that it contains no fat or fatty
acids (except the merest traces)....”
At the time these statements were published in The
Journal, the St. Paul Medical Journal, October, 1906, contained
an advertisement for Waterbury’s Metabolized Cod-Liver
Oil Compound, which contained this statement:
“The only tasteless preparation on the market which contains Cod-Liver
Oil in its entirety. The metabolized product is obtained by the
action of digestive ferments on pure Cod-Liver Oil.”
In the Ohio Medical Journal of Feb. 15, 1907, there
appeared in the form of an advertisement what purported
to be an analysis of Waterbury’s Metabolized Cod-Liver
Oil Compound by Prof. C. N. Kinney of Drake University.
While Professor Kinney made a quantitative analysis of the
preparation, the quantities were omitted from the analysis as
published. A footnote added by the Waterbury Chemical
Company called attention to this fact and closed as follows:
“Any physician who is not satisfied with the analysis we will be only
too glad to furnish the complete analysis by our representatives.”
If this weirdly constructed sentence meant anything, it
meant that the complete analysis would be furnished on
request. Such requests to the company, however, from
various sources failed to elicit the information required nor
was the “complete analysis” forthcoming. The inference to
be drawn is fairly plain.
In a circular accompanying the product as sold at present,
this statement occurs:
WATERBURY’S
METABOLIZED COD LIVER OIL COMPOUND
With Creosote and Guaiacol or Plain
DOES CONTAIN COD LIVER OIL
DOES ALLAY FERMENTATION
DOES AID DIGESTION
DOES ASSIST ASSIMILATION
BUT DOES NOT DISTURB THE STOMACH
As previous examination disclosed only the merest traces
of cod-liver oil in the product though claims were made
that it “represents cod-liver oil in its entirety,” and in view
of the fact, too, that present advertisements emphatically
declare that cod-liver oil is present in the preparation as
now sold, it was thought best to examine some of the
preparation with especial reference to the quantities of fatty
acids from cod-liver oil.

OLD LABEL |
|

NEW LABEL |
| It is interesting in this connection to note that this product is
no longer being sold under the name “Metabolized Cod Liver Oil Compound.”
See the illustrations of the old and new labels. |
The results of the examination are briefly as follows:
The total quantity of acids isolated amounted to about
0.3 per cent., and of this amount about two-thirds was
salicylic acid. Thus it appears from the examination of the
specimens bought on the open market that the preparation
contains at most but 0.1 per cent. of the fatty acids from
cod-liver oil, a totally insignificant quantity.
Notwithstanding the protestations by the manufacturers,
in the form of published analyses and circulars, it is seen
that the statements published in The Journal, Oct. 13, 1906,
p. 1207, are essentially substantiated; it is further evident
that the product does not deserve to be designated as a
cod-liver oil preparation. To obtain a medicinal dose of
cod-liver oil the patient would be compelled to swallow the
contents of a bottle of this mixture, and as the product
contains 11 per cent. alcohol the patient who did so would
probably experience a degree of exhilaration not referable
to cod-liver oil.—(From The Journal A. M. A., Oct. 9, 1909.)
Declared Misbranded
This product of the Waterbury Chemical Company, of
Des Moines, Iowa, was exposed in The Journal of the
American Medical Association, October 9, 1909. In May,
1910, the United States Government issued a notice of judgment
in which it was declared that Waterbury’s Metabolized
Cod Liver Oil Compound was misbranded. The court rendered
its decree of condemnation and forfeiture.—[Notice
of Judgment, No. 303.]
WATERBURY’S COMPOUND
Report of the Council on Pharmacy and Chemistry
The Waterbury Chemical Company having requested that
the Council reconsider its action of four years ago (see preceding
report) on the product then known as Waterbury’s
Cod-Liver Oil Compound, now called Waterbury’s Compound,
the matter was submitted to a referee. The referee reported
that the statement now made as to the composition of this
product is as follows:
“Made from Cod Liver Oil, Digestive Ferments, Malt Extract Unfermented,
Hypophosphites Comp. Special, Ext. Cherry, Eucalyptus, Aromatics,
etc.”
He held that the Waterbury Chemical Company has not
submitted satisfactory evidence to indicate that the objections
of the Council’s former unfavorable report have been met; that
there is no evidence that the product is a substitute for cod-liver
oil in any way; and that under the present methods of
exploitation it constitutes what is at least an inferential
fraud; and recommended that no further consideration be
given to Waterbury’s Compound. The report was adopted
by the Council.—(From The Journal A. M. A., March 20,
1915.)
COLCHI-SAL
Report of the Council on Pharmacy and Chemistry
Colchi-Sal, said to be made by the Anglo-American Pharmaceutical
Co., Ltd., New York, is advertised, sold and
“guaranteed” (sic) by E. Fougera and Co., Inc., New York.
According to the label of a recently purchased specimen:
“Each Capsule contains Cannabis Indica (Active Principle of) 1-500th
Grain (1⁄8 Milligram); Colchicine (Crystallized) 1-250th Grain (1⁄4
Milligram); Methyl Salicylate 20 Centigrams.”
The advertising circular around the bottle adds that the
mixture also contains “appropriate aromatic adjuvants.”
It is recommended in “Gouty and Chronic Rheumatic
Manifestations,” “acute cases of Gout,” “intestinal auto-intoxication
or dyspepsia,” “bilious headaches,” etc. Salicylates
are generally recognized as valuable in acute manifestations
of acute articular rheumatism; colchicum is useless
in these conditions. Both salicylates and colchicum are
practically useless in chronic rheumatic and in chronic gouty
affections. For dyspepsia, bilious headache, etc., salicylates
are distinctly contra-indicated and the drastic purgation
produced by colchicum would not be thought desirable.
Though methyl salicylate administered internally is not generally
considered so efficient as sodium salicylate, it is
asserted that the former
“... is found far more effective than salicylate of soda or other
salicylic derivatives when given in conjunction with colchicine as Colchi-Sal.”
Further, the highly improbable and unsubstantiated claim
is made that “the active principle of Cannabis indica” (whatever
that may be) “corrects any tendency of the colchicine to
irritate the gastro-intestinal tract” and that the “appropriate
aromatic adjuvants” “prevent intolerance of the methyl
salicylate.”
Colchi-Sal is put up in a way to appeal to the public; the
bottle has the name “Colchi-Sal” blown in the glass; the
label gives full instruction for the use of Colchi-Sal, and also
the price, suggesting that the preparation may be freely purchased.
Wrapped around the bottle is a circular advising
its use in various affections.
The physician who acts on the advice that it is well to
“insist on the pharmacist dispensing original bottles ...”
of the “little green capsules” actually suggests to his patient
the use of this preparation of methyl salicylate and colchicum
in conditions in which these drugs may do much harm and in
which proper treatment is imperative.
Colchi-Sal is typical of unscientific ready-to-take proprietaries.
It was held ineligible for New and Nonofficial
Remedies because of its secret composition, viz., the unknown
nature of the “active principle of Cannabis indica” (Rule 1);
because the circular in the package and the name blown in
the bottle constitute advertisement to the laity (Rule 4);
because the claim that cannabis indica removes the gastro-intestinal
irritation, and the claim of the superiority of
methyl salicylate are unwarranted therapeutic claims (Rule
6); because the name does not indicate the presence of the
habit-forming cannabis indica, and because of its unscientific
composition (Rule 10).—(From The Journal A. M. A.,
March 20, 1915.)
CYPRIDOL CAPSULES
Report of the Council on Pharmacy and Chemistry
Having voted that Cypridol Capsules be refused recognition,
the Council directed that for the information of physicians
publication of the following report be authorized.
W. A. Puckner, Secretary.
Cypridol Capsules, sold by E. Fougera & Co., New York,
are stated to be “Bottled in the New York Laboratories of
Vial, late Rigaud and Chapoteaut, Paris,” and to contain, in
each capsule, 2 mg. (1⁄32 grain) mercuric iodid (biniodid of
mercury) dissolved in a fatty oil. They are claimed to permit
the administration of mercury without danger of salivation—an
obvious misrepresentation.21 Cypridol Capsules are marketed
in a way to appeal to the public. If they are once prescribed,
the directions on the bottle and the full instructions
for the treatment of syphilis by means of Cypridol and by
other proprietaries sold by Fougera & Co. is likely to lead
the patient to attempt the treatment of this malady on his
own accord, and thus probably to forfeit his chances of cure.
Cypridol is a vicious example of the “ready-to-take” proprietaries.
Cypridol Capsules are in conflict with the rules of the
Council as follows:
Rule 4: The dosage, price, etc., on the label, and the name
“Cypridol” blown in the bottle, all tend to a direct self-prescribing
by the public. In addition to the objectionable statements
on the bottle itself, the preparation is put up in patent
medicine style and is accompanied by a circular giving full
directions for the use of this and of other proprietaries for
the treatment of syphilis in all of its stages. The circular
states that “a 1 per cent. solution of bin-iodide of mercury
in an aseptic oil” is “An Improved Specific in the Treatment
of Syphilis,” and after lauding the virtues of Cypridol, gives
full directions for the treatment of syphilis in its various
stages by means of Capsules of Cypridol augmented, during
periodical cessation of treatment, by “small doses of iodide
of strontium (Paraf-Javal’s standard solution, thirty grains
to the ounce).” Further, the circular expounds the need of
“a toning up of the general system” and by means of obsolete
theories and obviously untrue assertions recommends “Chapoteaut’s
Wine [another of their proprietary preparations],
each ounce of which contains 10 grains of phospho-glycerate
of lime. This is a delicious, nutritive tonic. A pint bottle
costs $1.00.”
Rule 6: Whereas it is evident that Cypridol, depending for
its effects on mercuric iodid, the ordinary well-known
hydrargyri iodidum rubrum of the U. S. Pharmacopeia, must
naturally have the properties of a mercuric compound, unwarranted
claims such as the following are made:
“CYPRIDOL does not render patients anemic. Ptyalism never follows
the administration of the capsules or injections. On the contrary,
patients rapidly put on flesh and keep well. There are no diarrhoeas or
other symptoms of intolerance even when the dose is pushed.”
Rule 8: The non-informing name “Cypridol” for a mercuric
iodid preparation is bound to lead to its use without
consideration of the fact that a potent mercury preparation is
being used, requiring a careful adjustment of dosage, a consideration
of the needs of the individual case, a correct diagnosis,
etc. While the advertising propaganda argues that
“physicians recognize the advantage of prescribing this solution
of mercuric iodide in an aseptic oil under the name of
‘Cypridol,’ because it does not betray to the laity the fact
that mercury is being used,” not only the physician but also
the patient has a right to know, and ought to know, the
potent character of the remedy which is being administered.
It is recommended that Cypridol be refused recognition
and that publication of this report be authorized.—(From
The Journal A. M. A., Dec. 19, 1914.)
CYSTOGEN, CYSTOGEN APERIENT AND
CYSTOGEN-LITHIA [L]
Abstract of Report of the Council on Pharmacy and
Chemistry
Cystogen is the therapeutically suggestive name applied to
hexamethylenamin by the Cystogen Chemical Company.
While investigation has shown that hexamethylenamin yields
formaldehyd only in the presence of an acid and consequently
can produce an antiseptic effect only in the gastric juice
and in the urine, it is claimed that Cystogen is an “intestinal
antiseptic” and that it “bears its disinfectant and antitoxic
qualities into well-nigh every important bodily cavity.”
As the sale of a simple drug even with the aid of the most
extravagant claims probably did not offer sufficient opportunity
for an extensive proprietary propaganda, the Cystogen
Company has put out two other preparations, Cystogen
Aperient and Cystogen-Lithia, and finds it an easy matter
by means of extravagant claims, unwarranted assertions and
pseudo-scientific arguments to recommend the use of one or
another, or often all three, in a well-nigh endless number
of diseases.
As the continued patronage of the medical profession cannot
be relied on for proprietaries of this sort, the Cystogen
Chemical Company takes good care that every Cystogen prescription
is likely to spread the Cystogen gospel among the
people. The Council has directed publication of its report on
the Cystogen products to call attention to the way in which a
simple drug of established value may be made the basis of an
extensive proprietary propaganda. A conservative discussion
of the action of hexamethylenamin appears in the Council’s
publication, “Useful Drugs.” The Council therefore refused
recognition to Cystogen, Cystogen Aperient and Cystogen-Lithia.—(From
The Journal A. M. A., Dec. 12, 1914.)
CYSTO-SEDATIVE
Report of the Council on Pharmacy and Chemistry
Cysto-Sedative is sold by Strong, Cobb and Company,
Cleveland, Ohio, with the claim:
“Each fluid ounce represents:
Thuja Occidentalis, 31⁄2 grains.
Pichi, 18 grs.
Saw Palmetto berries, 36 grs.
Triticum Repens, 36 grs.
Hyoscyamus 8 grs.
All inert extractive matter being eliminated.”
The therapeutically active constituents of arbor vitæ, pichi,
saw palmetto and couch grass have never been isolated—indeed,
it has not been proved that all of these drugs contain
any therapeutically active constituents. Yet the absurd
claims are made that all inert matter has been eliminated
and each lot of drug used in the preparation of Cysto-Sedative
is “tested in reference to its medicinal activity.”
Equally preposterous is the claim:
“In formulating Cysto-Sedative each drug entering into its composition
was subjected to careful study clinically to determine the exact
proportion required when combined to increase their efficiency as a
whole. Cysto-Sedative is scientifically prepared, the proportion of each
individual drug being so finely adjusted as to increase their therapeutic
action in the conditions for which they are intended, forming a preparation
always reliable and of the very highest medicinal activity.”
Some other extravagant claims made for this complex
unscientific mixture are:
“It gives relief in almost every form of cystitis and prostatitis....”
“The best results are obtained in the worst chronic cases of cystitis
and prostatitis....”
“In Cystitis, Urethritis, Prostatitis, Inflammation of the Vesicle Neck,
complicated with Gonorrhoea, Enuresis, Painful Micturition, the action
of Cysto-Sedative is prompt.”
The Council voted that Cysto-Sedative be refused recognition.—(From
The Journal A. M. A., Dec. 12, 1914.)
TAKA-DIASTASE AND LIQUID TAKA-DIASTASE
Report of the Council on Pharmacy and Chemistry
Some time ago it was decided that a reexamination should
be made of Taka-Diastase and Liquid Taka-Diastase, both
of which had previously been rejected, to ascertain whether
or not the preparations were in accord with the claims made
for them by the manufacturers. Accordingly, the matter was
referred to a committee of the Council, and an examination
of specimens of these two preparations bought in the market
was made. The referee’s report, which appears below, according
to the usual procedure, and before final confirmation
by the Council, was first submitted to the manufacturers of
Taka-Diastase for comment. The report recommends that
the rejection of Taka-Diastase and Liquid Taka-Diastase
be allowed to stand, and that the report be published. Parke,
Davis & Co., in their reply, which is given in full below,
claim that the report is unjust concerning Liquid Taka-Diastase,
because the period of activity of the preparation
has been greatly prolonged by reducing the amount of alcohol
from 18 per cent. to 10 per cent. and by adding glycerin.
They reiterate their claims for the digestive power of Taka-Diastase,
but admit that it will not reduce the stated amount
of starch to the colorless end-point in ten minutes (the
standard method for the valuation of diastase). They further
state that they would change the word “digest” on the
label to “liquefy.”
The conclusion of the report having been questioned, the
entire matter was referred to a member of the Council’s
staff of clinical consultants. His report, which, also, is given
in full below, states that the material before him was sufficient
to decide the matter, and no further tests were necessary.
He concludes that the claims of the manufacturers regarding
the strength and properties of the material are erroneous
and exaggerated; that the literature still sent out by Parke,
Davis & Co. is misleading; and that if substitution of the
word “liquefy” for “digest” were endorsed by the Council
confusion would result which would give an exaggerated and
false value to Taka-Diastase. He therefore recommends
that the report of the reinvestigation of Taka-Diastase be
accepted by the Council and published.
This report of the second referee was referred to Parke,
Davis & Co. with the request that they state more definitely
the actual amylolytic strength of their preparations. To this
they replied that they had no desire to discuss the subject
further, or to make any additional statements.
In accordance with the second referee’s recommendations,
the Council confirmed its provisional action and voted that
the rejection of Taka-Diastase and Liquid Taka-Diastase be
allowed to stand, and that the report which appears below
be authorized for publication.
W. A. Puckner, Secretary.
REFEREE’S REPORT ON TAKA-DIASTASE AND LIQUID TAKA-DIASTASE
Following is the report of the committee to which was
referred the reexamination of Taka-Diastase and Liquid
Taka-Diastase:
Some time ago a comparison was made of the various
methods proposed for the valuation of preparations claimed
to have amylolytic power. This work was reported in The
Journal,22 and the method proposed for the testing of diastase
preparations now appears in New and Nonofficial
Remedies.23 In view of the incorrect and exaggerated claims
made for Taka-Diastase, the Council in 1908 was obliged to
rescind its acceptance and to direct its omission from New
and Nonofficial Remedies. The report contained the following
reference to Taka-Diastase (Parke, Davis & Company),
a product that had been accepted for inclusion with New and
Nonofficial Remedies:
“The widest discrepancy between the values as claimed
by the manufacturer and those found by actual tests seems
to be shown in the case of Taka-Diastase. The liquid
preparation has been tested a number of times in different
samples and has always been found weak. Some samples,
in fact, were quite inert. This ferment appears to lose
strength very rapidly in solution, as the manufacturers
now concede. The stability of the solid product is also
far from satisfactory, and appears to be less than that of
the ferment as marketed some years ago. The two samples
examined recently were weak.”
More than three years have now elapsed since the publication
of the Council’s findings regarding Taka-Diastase—sufficient
time, it is believed, for the manufacturers to modify
either their claims or the product itself, and thus again make
it eligible for inclusion with New and Nonofficial Remedies.
With this idea in mind new specimens of Taka-Diastase and
Liquid Taka-Diastase were purchased from a Chicago drug
house and the preparations reinvestigated. The following is
the report of this reinvestigation.
REPORT OF THE REEXAMINATION
In our report on the diastase preparations three years ago,
it was recommended that Taka-Diastase be removed from
New and Nonofficial Remedies, because the examinations
showed that it did not have the digestive strength claimed
for it. This was true both for Taka-Diastase itself and for
Liquid Taka-Diastase. So far as the latter was concerned,
the starch-converting power was practically nil in those
preparations which had been in the drug stores for some
months.
During the last few weeks new tests have been carried out
with several samples of the Taka-Diastase preparations and
the results obtained are essentially the same as those obtained
in the former examinations. The liquid preparation is still
extremely weak in starch-converting power, while we found
that Taka-Diastase itself would convert only 16.6 parts of
pure anhydrous starch to the colorless end-point in ten
minutes, as explained below.
In our method of experimentation we determine the weight
of the diastase in question which will convert a given weight
of starch in uniform paste to the so-called colorless end-point
in ten minutes, that is to the point where it will no longer
give any color reaction with a standard iodin solution. The
standard starch weight in 50 c.c. always is 1 gm. or 1,000 mg.
and to a series of flasks containing this amount of starch,
maintained at a constant temperature of 40 C., the diastase
dilutions are added. These diastase dilutions are made by
dissolving small, accurately weighed amounts of the sample
in some small, constant volume of water, usually 5 or 10 c.c.
and they are then poured into the starch flasks at the right
temperature, and agitated regularly.
Tests are made by taking a few drops from each flask and
mixing with the iodin solution. The end-point is reached
when a dilution is found which, at ten minutes from the
mixing time, gives no color with the iodin reagent. The first
set of tests is taken as a general guide, and quite accurate
results may be obtained in a second set of dilutions.
We first used a sample of Taka-Diastase bought in the
open market. It was found that 140 mg. were required to
convert the gram of starch as explained. This is equivalent
to a conversion of 7.14 parts of starch by 1 part of the
Taka-Diastase.
A new, and possibly fresher, sample was then obtained and
the test repeated. With this new sample it was found that
60 mg. were necessary to convert the gram of starch to the
colorless end-point in ten minutes, from which it follows that
1 part of the ferment will convert 16.6 parts of starch to the
colorless end-point in the same time. With a new sample of
Liquid Taka-Diastase obtained simultaneously it was found
that 3.5 c.c. were necessary to convert 1 gram of starch to
the colorless end-point in ten minutes. As a fluidounce of
this liquid is said to contain 20 grains of the solid it will be
seen that the results approximately agree with those of the
first sample of the solid, and that they are both very low.
In the earlier tests 16 parts of starch converted by 1 part
of the ferment was the value found. These results are in
close agreement with values reported by Sherman (Jour. Am.
Chem. Soc., xxxii, 1073) for a sample of recent purchase. He
found a conversion of 51 parts of starch to the colorless end-point
in thirty minutes for one sample, while for another he
found 66 parts, in the same time. It will be noted that our
time limit is ten minutes. It is worthy of note that for a
perfectly fresh and specially prepared sample furnished by
Dr. Takamine, a conversion of 278 parts in thirty minutes
was found by Sherman. Taking the time into consideration,
it will be seen that the results are about the same for the
market samples as those found by us and much lower than
claimed, as well as much lower than for other makes of similar
products. The difference in the behavior of fresh specially
prepared Taka-Diastase and the market sample is very
clearly shown. No one questions the fact that fresh laboratory
samples of Taka-Diastase may show a moderate converting
power on starch. But we have to deal with the
activity of market samples only, and Sherman’s work and our
own show the low digesting power of the product as physicians
may secure it on the market.
The marked difference in activity between perfectly fresh
and ordinary market samples of Taka-Diastase is very
clearly shown also in a recent paper published by Wohlgemuth.24
In the digestion of starch paste to the “dextrin”
stage Wohlgemuth found in the commercial sample a
strength approximately a hundred times less than that
observed in a fresh sample sent him by Dr. Takamine.
Wohlgemuth’s results were obtained by a method not essentially
different from ours, with this difference, however, that
he digested through 24 hours in the cases reported, and carried
the reaction to the “dextrin” stage only, in place of to
a colorless end-point. Making the proper reductions, it is
evident that the actual values found by him for the market
samples bought in Germany are not greater than those
reported by us.
The reference to the work of Sherman is made because, in
a following paper in the same journal, he recommends the
use of salt as an activator in finding the strength of certain
diastase preparations. It is well known that dialyzed diastase
preparations and starch of highest purity have but
slight action on each other; a little salt increases the activity
greatly, and also increases the activity of commercial
diastase preparations. These facts Sherman utilizes in working
out a method for valuation of commercial diastases.
The facts were well known to us at the time of our former
report, but it was not thought best to depart from the general
method which had been in use by all analysts following
the general scheme of Roberts. Quite recently, I. Bang has
published a paper on the investigation of diastase (Biochem.
Ztschr., xxxii, 417) in which he studies the behavior of
sodium chlorid and other salts on the rapidity of starch
conversion, and finds that a much smaller amount of salt
than Sherman recommends brings the maximum increase.
The method employed in our former tests is a good comparative
method, and this is all that may be claimed at
present for any method. By adding salt to our starch solution
the activity of Panase and other ferments is likewise
greatly increased. For Panase, a preparation possessing
rather high starch-converting power, we have recently found
an increase of about 30 per cent. in the converting power,
with salt present. Working to loss of blue color, merely,
it is possible in this way to get a higher value than that
claimed by the manufacturer. There is no practical gain
in using the salt for our purpose as the methods are at
best arbitrary, and the results only comparative.
Taking all the facts into consideration, it is recommended
that the rejection of Taka-Diastase and Liquid Taka-Diastase
be allowed to stand and that, in view of their extensive
exploitation, this report be authorized for publication
so that physicians may know the facts.
This report was referred to Parke, Davis & Co., and they
made the following reply:
“The report submitted in your letter of the 23d is, we
contend, erroneous and unjust: first, to our Liquid Taka-Diastase,
because over three years ago we changed our
formula, reducing the alcohol from 18 per cent. to 10 per
cent., increasing the glycerin and thus prolonging greatly
the period of activity.
“As for our regular Taka-Diastase, our claim is and has
been for years simply that Taka-Diastase will digest or
hydrolyze 150 times its weight of starch in ten minutes,
under proper conditions. We do not claim, we do not permit
our representatives to claim, that Taka-Diastase will
completely transform starch, to the colorless end-point, into
sugars. Taka-Diastase is used to supplement a deficiency
of ptyalin and converts the starch into soluble material with
great rapidity, thus giving the gastric fluid immediate access
to the proteids.
“If in the enclosed labels the word ‘digest’ were replaced
with the word ‘liquefy,’ the claim could not be assailed by
the most carping critic. To save any possible question, we
shall therefore make this change in our label, having it read:
‘Taka-Diastase will liquefy 150 times its weight of starch in
ten minutes, under proper conditions.’ Is there the slightest
question in your mind that this statement as just quoted is
entirely correct and entirely supported by clinical experience?
“It is our conviction that Taka-Diastase has a very remarkable
power to hydrolyze starch either in the test-tube or in
the stomach, and that this property is of great utility in
clinical work. We do not claim that its conversion of the
starch into sugars is complete, to the colorless end-point of
the Johnson test; and on this point we have been perfectly
frank with the Council, as well as with every physician who
has taken sufficient interest to inquire.”
In view of the above protest, the matter was submitted to
a second referee, who reported as follows:
“Your referee on the matter of Taka-Diastase has made a
careful investigation of the reports and correspondence submitted,
and begs to make the following report:
“The question at issue, viz., whether Taka-Diastase should
be included in New and Nonofficial Remedies, I believe, can
be determined by the material before me, and further tests
of the material are not necessary.
“The letter of the makers of Taka-Diastase admits that the
early claims regarding the strength and properties of the
material were erroneous and exaggerated. Since the product
was once admitted to New and Nonofficial Remedies, it may
be claimed that as the Council on Pharmacy and Chemistry
must have been in error then, it may be now. Your referee
does not consider this supposition worth discussing. The
conclusion he draws is that the Council was too hasty in
accepting the preparation, and that the incident shows how
much better it would be in all cases to accept no remedy
until sufficient time has been given for conclusive tests.
“The literature still sent out by Parke, Davis & Co.
regarding Taka-Diastase is misleading and of a kind more
appropriate for a nostrum than a standard chemical substance.
What would we think if morphin, quinin or even
heroin were advertised in the same way? I cite the statement,
‘Taka-Diastase digests starchy food with vigor and
directness.’ It seems to the referee that the proposition to
modify the label to indicate the amount of starch which is
liquefied rather than the amount which is saccharified, in
accordance with the Council’s standard, is bound to lead
to confusion and to give an exaggerated and false value to
Taka-Diastase.
“Your referee recommends that the report of the reinvestigation
of Taka-Diastase which has been submitted to me,
be made available to the medical profession, and that the
rejection of Taka-Diastase and Liquid Taka-Diastase be
allowed to stand.”
This report of the second referee was submitted to Parke,
Davis & Co., with the request that they state more explicitly
their claims regarding the activity of Taka-Diastase and
Liquid Taka-Diastase, in order that, if they decided to revise
their claims for the preparations, such revision of claims
might be published along with the reports of the Council.
They replied:
“Answering your note of the 15th instant: We have no
desire to discuss further the subject of your letter of February
24, or to make any statement beyond that set forth in
our letter to you of Dec. 27, 1911.”—(From The Journal A. M. A.,
July 6, 1912.)
DIGALEN OMITTED FROM N. N. R.
Report of the Council on Pharmacy and Chemistry
Digalen is a proprietary said to contain a soluble form
(digitoxinum solubile Cloetta) of digitoxin, the chief active
principle of digitalis. This preparation was accepted25 by the
Council in 1909 for inclusion in New and Nonofficial Remedies.
The Council had not at that time determined whether
Digalen contained “soluble amorphous digitoxin,” as claimed,
or not. The product was accepted merely as a standardized
soluble and fairly stable digitalis preparation.
After the acceptance of Digalen, the therapeutic claims
made for it by the manufacturers increased in extravagance.
Meanwhile, evidence was brought forward by various independent
investigators which tended not only to show that
these therapeutic claims were unfounded, but also to discredit
the claim that Digalen contained a principle chemically
identical with digitoxin. In view of the obscurity of
the whole subject of the chemistry of the digitalis principles,
the latter claim (that Digalen was a solution of
“amorphous digitoxin”) had been an academic issue at the
time of the acceptance of the product. When, however,
the manufacturers of Digalen sought to mislead physicians
by increased and unwarranted therapeutic claims, the Council
felt that investigation of the whole matter was imperatively
demanded to decide whether or not Digalen should
be retained in N. N. R.
The questions at issue were: (1) the presence in Digalen
of “amorphous digitoxin”; (2) the constancy of composition
and reliability of action of Digalen, and (3) the claim
that it causes less gastric disturbance than digitoxin. No
satisfactory proof has yet been offered that Digalen contains
“amorphous digitoxin.” The mass of evidence tends to
show that Digalen is not constant in composition or reliable
in action, and that, when given in doses corresponding
in therapeutic activity, Digalen causes quite as much gastric
disturbance as the official galenical preparations of digitalis.
The outcome of protracted negotiations between the Council
and the Hoffmann-La Roche Chemical Works may be
summed up as follows: 1. The manufacturers promise to
hold in abeyance the claim regarding the presence of
“amorphous digitoxin.” 2. They refuse to concede the variable
composition of Digalen. 3. They reassert the claim that
Digalen is superior to other digitalis products with respect
to liability to cause gastric irritation and consequent
vomiting.
In view of the unsatisfactory character of the reply on
the second and third points, the Council voted that Digalen
be omitted from N. N. R. and that publication of the report
on Digalen which appears below be authorized, as well as
of the two reports26 (A and B) referred to therein.
W. A. Puckner, Secretary.
Referee’s Report on Digalen
Because of persistent conflict with Rule 6 (unwarranted
therapeutic claims) and Rule 1 (composition) it is recommended
that Digalen be omitted from New and Nonofficial
Remedies; also that a copy of the report be sent to the
manufacturers, and that publication of this report and the
two previous reports submitted to the Council be authorized.
The nature of the problems involved necessitates a somewhat
extended discussion of the subject.
Digalein (liquid) is said to contain 1 part of soluble
amorphous digitoxin Cloetta in 1,000 parts of glycerin and
1,600 parts of water with 7.5 per cent. of alcohol. One c.c.
is said to contain 0.0003 gm. of the amorphous digitoxin.
Digalen was accepted by the Council27 and the following
footnote was appended to the description in New and Nonofficial
Remedies:
“The Council has not determined whether digalen contains ‘soluble
amorphous digitoxin’ or not, but accepts it simply as a soluble digitalis
preparation.”
Tablets of Digalen were accepted by the Council as a
dosage form of Digalen. Each tablet is said to represent
0.5 c.c. (eight minims) of Digalen (liquid).
One of the principal considerations which led the Council
to accept Digalen was that it was regarded as affording a
fairly constant and stable preparation of digitalis suitable
for intravenous administration. If Digalen is not fairly stable
and of fairly constant composition it has no obvious advantage
over an active soluble digitalis preparation, such as
digitalein.
The evidence now at hand seems to show: 1. Digalen is
not of constant composition or activity. 2. The manufacturers,
or their agents, continue to make misleading statements.
3. It is merely a solution of certain digitalis principles,
probably of digitalen mainly, in impure form.
COMPOSITION
Cloetta28 prepared a soluble amorphous substance which
he called “Digitoxinum solubile Cloetta,” but no information
concerning the method of preparation has been published.
Cloetta reported the result of an elementary analysis of
his product which he compared to the analyses of digitoxin
(crystalline) made by Schmiedeberg and by Kiliani, and
stated that there could be no doubt concerning the chemical
identity of the two substances.
Kiliani29 characterized as preposterous Cloetta’s claim that
the active constituent of Digalen is chemically identical with
digitoxin and stated that Digalen was merely an impure
digitalein. Kiliani has recently reiterated the statement that
the so-called “amorphous digitoxin” is not identical with
digitoxin.30
Cloetta’s failure to publish his method of preparing
Digalen places an additional burden of proof on him (or the
manufacturers of Digalen), concerning the identity of the
product, and in the face of Kiliani’s denial of the correctness
of Cloetta’s contention we must have strong corroborative
evidence of Cloetta’s claim before we can accept it as being
established.
The difficulties of dealing with the chemistry of digitalis
are so well known that they hardly require further mention
here, but under the circumstances Cloetta cannot be considered
as being wholly unprejudiced, and, while the same
might perhaps be said of Kiliani, such evidence as can be
deduced tends strongly to support Kiliani’s view, and to
disprove the contention of Cloetta.
There is much confusion regarding the names which have
been applied to the various principles obtained from digitalis,
and while it is undesirable that an established name
should be given to a newly discovered principle, one might
overlook this if no effort were made to associate the therapeutic
actions of the two substances to an extent which
the truth did not justify.
While the Council at that time did not challenge the existence
of “amorphous digitoxin” and made no attempt to
determine the identity with digitoxin of the substance forming
the basis of Digalen, the manufacturers of Digalen
have sought to show that Digalen and digitoxin were identical
so far as their therapeutic actions were concerned, but
that Digalen lacked the disadvantages of digitoxin. What
was a purely academic question when the acceptance of
Digalen was under discussion by the Council becomes a
matter of very great practical importance when the manufacturers
of Digalen seek to mislead the physician by these
claims.
The evidence which lends support to the view that Digalen
and digitoxin are wholly dissimilar may be summarized as
follows: Digalen differs greatly in its physical properties
from digitoxin and in certain of its physiologic actions, as
the manufacturers themselves state, Digalen being amorphous,
and soluble in water, while digitoxin is crystalline
and insoluble in water. The manufacturers state that
Digalen differs from digitoxin in certain of its physiologic
actions, but the two substances do indeed differ far more
than they admit.
Cloetta and the manufacturers of Digalen lay especial
stress on the claim that Digalen is cumulative to a far
less extent than digitoxin, and that it has far less tendency
to cause gastric disturbance than the latter. The first of
these claims is true; the second is the very opposite of the
truth, as we shall show.
We know nothing of the structure of any of the digitalis
principles, and even though one were to admit (purely for
the sake of argument) that Digalen and digitoxin were
chemical isomers, that fact could not be taken to lend any
support to the contention that the two substances were
identical, in the face of the established fact that they differ
physically and physiologically in nearly every particular,
and agree only in that they both cause standstill of the
heart in the same way—an action possessed also by so dissimilar
a substance as barium.
The manufacturers of Digalen support the claim of the
identity of their product with digitoxin by stating that
“Digalen is a solution of the most active glucoside of
digitalis.”31 Of course, it is very generally admitted that
digitoxin is the most active principle of digitalis, though
there is some question concerning its glucosidal nature.
Digalen is in fact far less active than digitoxin, as has
been shown by a number of independent observers[M] (Worth
Hale, 1910; Hatcher and Brody, 1910; Neave, 1907; Miller,
1908; the referee; Weis, 1912).
The essential fact which appears from the investigation
of Weis is that Digalen did not behave like digitoxin in
any case.
Hale also found that Digalen gave atypical actions in
which the effects on the central nervous system became
prominent. The referee can corroborate these observations of
Hale’s on frogs, but the convulsive symptoms were prominent
with some specimens of Digalen on mammals, though not
with others, the more recent specimens of the preparation
showing the action prominently.
The results of all these biologic tests, as well as of the
physical tests made by Weis, certainly lend no support to
the contention of Cloetta that the potent constituent of
Digalen is identical with digitoxin, but, on the contrary,
they show conclusively that the two substances differ widely
in many essentials, and the continued claim of the manufacturers
that the “amorphous digitoxin” said to be contained
in Digalen is the same as digitoxin, or that it is the
most active glucosid of digitalis, can be considered only as
misleading, and therefore in conflict with the rules of the
Council.
CONSTANCY OF COMPOSITION AND ACIDITY
The manufacturers of Digalen continue to claim that it
is of constant and uniform activity,32 and they imply this
even when they do not state it in those words; for example,
a substance cannot be considered reliable if it is variable in
activity. “Digalen is Absolutely Reliable. It is Standardized
and consequently always uniform. It does not produce
gastric disturbances.”
That the foregoing is absolutely untrue can be shown
abundantly. Hale33 found digalen not to be uniformly
stable; Weis found very different degrees of activity for
Digalen in the liquid and tablet forms, the tablets being
but one-third as active as the liquid, and the referee found
very great variations in the activity of different specimens of
Digalen, one specimen being almost inert. The results
obtained by Miller show that Digalen is sometimes very
slightly active, or not at all so.
The foregoing citations show conclusively that Digalen is
not of uniform activity. When reliability is claimed for
Digalen in contrast to the known variability of digitalis, it
must be considered as tantamount to the claim that Digalen
is not subject to such variability and it must be held that the
manufacturers make misleading statements when they assert
that Digalen is absolutely reliable.
The manufacturers claim that Digalen does not produce
gastric disturbances (see advertisement cited).
It is quite true that when small doses of Digalen are used
therapeutically it fails to produce gastric disturbances
because it is of such slight activity, as previously stated,
but when it is used in amounts which correspond in activity
to such doses of the ordinary galenical preparations of digitalis
as commonly cause nausea and vomiting it does cause
gastric disturbances quite as readily as the latter.
Among the clinicians who have found that Digalen causes
gastric disturbances may be cited: Veiel,34 Mueller,35 Eichhorst36
and Teichmann.37
Eggleston and Hatcher38 compared the emetic and cardiac
activity of Digalen and numerous other digitalis bodies and
preparations and found that the emetic activity of Digalen
was decidedly greater in proportion to its cardiac (or therapeutic)
action than was that of digitalis or digitoxin.
In the absence of any evidence to controvert this clinical
and experimental evidence, the continued claim that Digalen
does not disturb the stomach must be looked on as deliberate
misrepresentation.
MISLEADING THERAPEUTIC CLAIMS
The recommendation that Digalen be dismissed from
N. N. R. is made with the full appreciation of the fact that
the manufacturers of Digalen and their agents have
repeatedly stated that they desired to comply with the rules
of the Council, and that they have withdrawn several statements
to which the Council has taken exception, but the
fact remains that despite these reiterations the advertisements
of Digalen continue to embody statements which the
Council can only consider misleading.
The Council believes that the following advertisements
constitute gross therapeutic exaggerations:
“Digalen a sheet anchor in pneumonia; a strong support to the heart
in this deadliest of infectious diseases among adults. The prompt
action of Digalen, by intravenous or intramuscular injection makes it
possible to save lives which might be otherwise hopelessly lost. The
best digitalis preparation which we have at the present time.”39
“The digitalis for children. Because its dosage can be controlled.
Endorsed by pediatrists everywhere.”40
“The myocarditis of Tuberculosis so frequently encountered, especially
in the advanced stage of the disease, may be controlled with the aid of
Digalen. The standard digitalis preparation.”41
“Digalen is Absolutely Reliable. It is standardized and consequently
always uniform. It does not produce gastric disturbances.”42
Digalen is not a sheet anchor in pneumonia, for there is
no drug deserving such a title. Digalen has no action which
other digitalis preparations lack, and cannot save lives
otherwise hopelessly lost. The dosage of Digalen cannot
be controlled any better than that of other digitalis preparations,
since its activity is variable. We cannot control the
myocarditis of advanced tuberculosis by this or any other
means.
CLAIMED SUPERIORITY
Various digitalis principles, including digitoxin, digitalin
(true) and digitalein, have been known for many years.
Therapeutically they have been found wanting and there
appears to be no basis for the continued claim that Digalen
has any superiority over these several digitalis principles.
On the contrary, the evidence is accumulating that Digalen
has no advantage in any particular over a solution of digitalein,
and misleading claims of the manufacturers and their
agents certainly interfere with the formation of that calm
and unbiased opinion on the part of the general practitioner,
which, when applied to the non-proprietary digitalis principles
has caused them to fall into disuse.—(From The Journal
A. M. A., Sept. 5, 1914.)
DIORADIN REFUSED RECOGNITION
Report of the Council on Pharmacy and Chemistry
A preparation called Dioradin was placed on the market
as a cure for consumption three years ago in Europe and
somewhat later in this country. It was first submitted to the
Council in July, 1911. Because of the manifestly unwarranted
claims made for its use in the treatment of tuberculosis,
the Council voted that the product be refused recognition
for conflict with Rule 8, without at that time taking
under consideration the question whether or not it was in
conflict with other rules of the Council.
In June, 1912, further consideration of Dioradin was
requested. The American agent having promised a reform
in the methods of advertising, the Council considered the
available evidence regarding the identity and value of the
preparation. Examination of evidence regarding the composition
of Dioradin—claimed to consist of radium chlorid,
iodoform and menthol in an ether-oil solution—showed serious
discrepancies as to the amount of radium as well as
to the identity and amounts of other constituents. It was
further found that the experimental evidence was insufficient
and biased. Then, too, in view of the difficulty of judging
the effects of medicines in tuberculosis, the clinical data
were unconvincing. There was nothing to prove that the
reported improvements, even if they actually occurred, were
to be ascribed to the mixture as a whole rather than to any
one of its constituents.
As a result of these findings, the Council voted that
Dioradin be refused recognition and that the publication of
these facts be authorized. In accordance with its regular
procedure, it also submitted the report to the agent. In
reply the agent submitted evidence which showed that he
was not responsible for the misstatements about Dioradin
but offered no facts that affected the Council’s findings.
The entire matter having been referred to a second referee,
minor modifications of the first draft of the report were
authorized. Since then the Dioradin Company has submitted
two reports of examinations of Dioradin made for the company
in Germany showing a higher radium content than that
previously found. These reports do not alter the facts
brought out in the report of the Council that the composition
of Dioradin has been variable, which past variability
arouses a feeling of uncertainty or lack of confidence. In
view of this the amended report was ordered published and
appears below.
W. A. Puckner, Secretary.
FIRST SUBMISSION OF DIORADIN
Dioradin, a preparation for the treatment of consumption
originated by Dr. R. de Szendeffy, Budapest, Hungary, was
submitted to the Council by Louis Gero, Ltd., New York,
with the following statement of composition:
“A radio-active preparation of Menthol, Iodin and Radium Barium
Chlorid 1⁄10 of a drop; in ether solution.”
A circular which accompanied the submission stated:
“Preparation No. 3 of Dioradin contains not only terpins but also
iodin salts.... In view of the fact that emanations of the
radium as well as the combinations of the evasive iodin terpins enter
into the organism through the lung....”
Later these indefinite statements of composition were supplemented
by the following:
| “In 100 c.c. there are: |
| 1 | gr. Iodoform. |
| 5 | " Menthol. |
| 10 | drops Radium chlorid solution (1 milligr. in 100 c.c. of water). |
| 5 | gr. ether. |
| 90 | " Oil (ol. amygd. frig. press).” |
In a circular contained in the package these claims were
made:
“The preparations of the Dioradin are based on the miraculous
effects which scientific researches have shown in regard to the
different sicknesses treated with radium.
“It is generally known that radium, even if externally employed,
has proved itself to be a bactericidal remedy. Its effect is multiplied
if one employs it internally even in infinitesimal doses, in consequence
of its permanent action of emanation on the organism.
“The preparations of the Dioradin contain the radium itself. For
this reason their antiseptic and bactericidal effect is much more
intensive than with medicaments which contain only its emanation,
which disappears in a short time.”
In view of the general extravagance of the claims made for
its therapeutic action the preparation was rejected without
considering other possible conflicts with the rules of the
Council.
SECOND SUBMISSION OF DIORADIN
Having been advised of the rejection by the Council of
Dioradin the American agency, which in the meantime had
become the Dioradin Co., requested further consideration.
The Council therefore took up the subject again. After certain
typographical errors had been corrected the following
was now given as the composition:
| “1 | gram Iodoform. |
| 5 | grams Menthol. |
| 10 | drops Radium Chlorid Solution (containing 1 milligram of radium chlorid in 100 cubic centimeters of water). |
| 5 | grams Ether. |
| 89 | grams expressed oil of almond. |
| This liquid is put up in ampules containing one cubic centimeter of liquid.” |
In support of the therapeutic claims for Dioradin the
American agent submitted literature consisting chiefly of
articles by Dr. Bernheim of Paris. Before reporting on the
requested reconsideration of Dioradin the referee directed
the secretary of the Council to point out to the American
agent that in the formula given, the amount of non-volatile
matter should be about 90 per cent., whereas the report of
the Lederle Laboratories which accompanied the request for
reconsideration states that but 72.08 per cent. was found in
the analysis. In reply the agent stated that he had called
the attention of Dr. Szendeffy (the originator of Dioradin)
to the discrepancies concerning non-volatile matter and that
he felt sure the discrepancy was wholly accidental (sic).
In a later communication the agent submitted a statement
of analysis from the Lederle Laboratories of a new specimen
of Dioradin according to which the amount of non-volatile
matter agreed essentially with the amount claimed by the
agent.
The referee, having examined the evidence, is of the
opinion that the statement of composition is misleading and
that the therapeutic claims are unwarranted, thus:
DISCREPANCIES IN RADIUM CONTENT
The chief claims for its therapeutic value are based on the
radium content, yet the discrepancies and contradictions,
regarding this are serious.
In connection with the reconsideration of this product the
agent presented a certificate of chemical examination by the
Lederle Laboratories in which the following statement was
made as to the radio-activity:
“Examination shows the preparation to possess slight radioactivity,
corresponding in activity to less than 1-10,000 of 1 milligram of
radium bromid per ampule. According to the sworn statement of Dr.
A. de Szendeffy, the originator of Dioradin, the preparation contains
10 drops of radium chlorid solution (1 milligram in 100 cubic centimeters
of water) in 100 cubic centimeters of the preparation. This
would correspond to 5-1,000 milligram of radium chlorid in 100 cubic
centimeters, or about 1-20,000 of 1 milligram per ampule.”
A cursory reading of this paragraph gives the impression
that Dioradin possesses fully the amount of radio-activity
claimed by its originator, Dr. A. de Szendeffy. This impression
is greatly strengthened by the concluding paragraph
of the Lederle report, which says:
“In conclusion, our examination shows that the preparation submitted
to us as Dioradin possesses radio-activity, and contains a fixed
oil (apparently expressed oil of almond), iodoform, menthol and
ether, thus confirming the sworn statement of Dr. A. de Szendeffy in
regard to the composition of this product.”
On inquiry as to the method used by the Lederle Laboratories,
in determining radio-activity the agent submitted
a further statement of the Lederle Laboratories which
describes the gamma ray test by which the determination
was made and a radium value equivalent to 0.000041 mg. of
radium bromid per capsule was obtained. The report then
says:
“The variations of the single measurements from the mean in
the case of the natural leak and the leak with the Dioradin near
were so large that we did not feel justified in assigning much accuracy
to the figure, 0.000041, but stated that the amount of radium per
capsule could not be greater than 0.0001 mg., with the possibility of
there being a much smaller amount present.”
It is evident that the wording of the reports of the Lederle
Laboratories is liable to give the impression that their examination
confirms the claims made for Dioradin.
It is further evident from these reports that the amount of
radio-active matter has not been definitely ascertained but
that it is at the best very small. The unreliability of the
claims for radium content of Dioradin was recently shown
by Buechner,43 who found a specimen obtained from an
apothecary to contain but 1-1,000 of the amount claimed.
VOLATILE AND NON-VOLATILE MATTER
The varying claims regarding the content of volatile and
non-volatile matter throw doubt on the entire composition of
Dioradin, for if the statement as to these is wrong the rest
of the statement regarding composition cannot be given
credence.
In the first submission of Dioradin about 89 per cent. of
non-volatile matter was claimed but in the report of the
analysis by the Lederle Laboratories, which accompanied the
resubmission, only about 72 per cent. was found. Later the
Lederle Laboratories reported that an examination of a new
specimen of Dioradin had shown about 90 per cent. of non-volatile
matter. The discrepancies between the composition
claimed for Dioradin and that found for the product in the
first Lederle report has shown that the agent was quite
ignorant of the composition of the product which he was
selling.
INDEFINITENESS OF THE IODIN CONTENT
The label on the trade package of Dioradin first submitted
to the Council stated that the product contained iodoform;
a similar statement was made in the submission of the
product; the circular accompanying the first submission
stated that “iodin salts” were contained in the product while
the iodin content was referred to further on in this circular
as “combinations of evasive iodin terpins.” In Bernheim’s
papers, which have been used to advertise Dioradin, and
which are referred to in the same circular, the iodin compound
is called “iode peptonisé,” which, according to information
stated by the American agent to have come from
Budapest, is to be translated “iodized peptone.” What is
the meaning of this confusion? One would naturally suppose
that the preparation to be sold in this country contains
iodoform in an ether-oil solution while the one used by
Bernheim and Dieupart44 was stated to contain an ethereal
solution of “iodized peptone.” This is another mystification,
for an ethereal solution of any kind of peptone would be a
novelty. The matter is of some importance, for Bernheim
and Dieupart lay great stress on the difference between
“peptonized iodin” and other iodin (loc. cit., p. 333) and of
the superiority of ethereal over oily solutions (loc. cit.,
p. 334). The American agents, however, in the second submission,
state that this is all a mistake; that the Dioradin
used by Bernheim is the same Dioradin which was submitted
to the Council; and that this does not contain, and never
did contain, the ethereal solution of “iode peptonisé” to
which Bernheim attached so great importance. Bernheim
(report to Medical Congress of Lyons) himself has come
to the same conclusion; for five months after his first paper
he believes that the “special salt of radium” (sic) is the
principal agent; so that the “peptonized iodin” must be unimportant,
and in a cablegram of July 4, 1912, he now informs
the Dioradin Company that the formula was incorrectly
given in his first papers “owing to my ignorance of actual
composition,” and that all the Dioradin used by him was of
the composition stated in the submission to the Council.
While this vindicates the good faith of the American
Dioradin Company, it does not clear up the mystery. The
question occurs at once: What led Dr. Bernheim to make
such positive statements? Was he drawing purely on his
imagination? If so, why did his imagination take this
peculiar special direction? Or if he did have some reason
to imagine the “iode peptonisé,” who supplied this reason?
And if, at that time, he was given to understand by
Szendeffy, who must have supplied him with the material,
that it contained the iodized peptone, how can he be positive
at this time, that it did not contain it? Has he actually
analyzed the old material?
There is also a further question which needs to be
answered. Why has Dr. Szendeffy waited until Dioradin
was rejected by the Council before correcting Bernheim’s
serious misapprehension, in the meantime permitting the
circulation of Bernheim’s paper?
Until these questions have been satisfactorily answered, the
element of mystery about the composition of Dioradin cannot
be cleared away.
EXPERIMENTAL EVIDENCE
The available experimental evidence regarding “Dioradin”
is restricted to some quotations from its inventor Szendeffy,
in the paper of Bernheim and Dieupart (p. 334). These, if
confirmed, would show that radium alone has practically
no effect on cultures of tubercle or colon bacilli; that 0.1 gm.
of “iode-menthol” (concentration not stated) checks the
growth of the acid-fast organisms; and that this antiseptic
efficiency can be nearly doubled by the addition of a little
radium. No quantitative data are given, so that it is difficult
to judge the accuracy of the observation. Granting that
it is correct, it would have little bearing on the therapeutic
actions of Dioradin, for there is nothing to show that the
effective test-tube concentration is reached in the pulmonary
tissues.
It is also claimed that the injection of Dioradin prevents
tubercle infection. The referee believes that the Council and
the medical profession should hesitate to accept this conclusion
without further details; and these would require
confirmation by unprejudiced observers.
CLINICAL EVIDENCE
The Dioradin Company submits considerable clinical data
in favor of Dioradin. It must be remembered that most
favorable opinions have been published, from time to time,
about scores of “consumption cures,” which have mysteriously
lost their efficiency when their novelty wore away.
There is no more reason to doubt the good faith of those
who are enthusiastic about Dioradin than of those who have
been enthusiastic about other “cures.” There appear to be
features in the course of tuberculosis which make the judgment
of therapeutic measures peculiarly difficult. It is possible
that impartial clinical trials of Dioradin by tuberculosis
experts appointed by the Council might facilitate judgment
as to the actual efficiency of Dioradin. The referee
doubts, however, whether this would advance the Council
very much toward the acceptance of the substance. Such an
investigation would be so lengthy that it should not be
undertaken until the Dioradin Company itself has offered
at least presumptive evidence in this direction, especially in
view of the adverse report recently made by Cecil Wall.45
Ten tuberculous patients were treated by Wall in strict
accordance with the method outlined to him by Bernheim,
yet Wall concludes that none of the cases, though treated
accurately in accordance with the instructions, can be quoted
to justify any of the claims for the therapeutic efficiency of
Dioradin. The Council cannot undertake lengthy investigations
of this character until it is put in possession of
data which would show to its satisfaction that such investigations
would probably be fruitful.
CONCLUSIONS
From investigations made, it appears that the claims in
regard to the composition of Dioradin have contained vague
statements and contradictions which arouse a feeling of
uncertainty and lack of confidence. Until this uncertainty is
cleared away, Dioradin cannot be considered as complying
with Rule 1. The experimental data are insufficient and
unconvincing. Some favorable clinical reports have been
submitted, but the accuracy of the observations is to be
questioned and they are more than offset by the negative
results observed by Cecil Wall. As might be expected, other
negative results, if observed, have not been submitted and
there is nothing in the manufacturer’s claim to show whether
the improvement reported is really due to the peculiar mixture
called Dioradin or to any one of its ingredients.
It is therefore recommended that Dioradin be not accepted
for New and Nonofficial Remedies. In view of the extensive
advertising of this preparation and because of the admittedly
incorrect statements in the earlier papers it is recommended
that publication of this report be authorized.—(From
The Journal A. M. A., Oct. 26, 1912.)
ECHINACEA
Report of the Council on Pharmacy and Chemistry
The Council has voted to reject several non-proprietary
articles and has recommended that the reasons for their
rejection be given in The Journal; among these is echinacea.
The following paper has been submitted by a subcommittee
with the recommendation that it be published. This recommendation
was adopted.
W. A. Puckner, Secretary.
Echinacea
When this drug was first introduced, it was a typical nostrum,
with exaggerations regarding its therapeutic value that
were somewhat more gross than usual. It was later adopted
by the eclectic school without being freed from the stigmata
of its origin. It was also pressed into use as the main ingredient
of such proprietary preparations as Echafolta, Ecthol
Eusoma, etc. Efforts have been made to get the regular profession
to use it in these various forms.
According to J. U. Lloyd (Pharm. Review, vol. xxii, p. 9–14),
the introduction of echinacea into eclectic medicine is due to
the efforts of Dr. H. F. C. Meyer to increase the sale of
Meyer’s Blood Purifier, a secret remedy containing it. The
following is a literal copy of the label on this nostrum:
After Lloyd had identified the plant, Meyer put the preparation
out under another form with the following label:
These absurd claims of an evidently ignorant man have
passed into the more recent proprietary advertising matters
and into much of the eclectic writings. Indeed, the seemingly
impossible had been attained by even surpassing Meyer’s
all-but-all-embracing claims. Not content with endorsing
echinacea as a positive and speedy “specific” for rattlesnake
bite, syphilis, typhoid fever, malaria, diphtheria and hydrophobia,
later enthusiasts have credited it with equally certain
curative effects in tuberculosis, tetanus and exophthalmic
goiter, and with the power of retarding the development of
cancer.
It is worth noticing—although it is not surprising—that
these far-reaching claims have been made on no better basis
than that of clinical trials by unknown men who have not
otherwise achieved any general reputation as acute, discriminating
and reliable observers. No attempt seems to have
been made to verify these claims by accurate scientific
methods, clinical or otherwise, although this could very easily
have been done.
Not one of the eulogistic reporters and exploiters seems
to have considered it worth while to determine by the
simplest control experiments whether the drug possesses any
bactericidal or antiseptic powers whatever. It is therefore
not very strange that discriminating physicians have failed
to show much enthusiasm. One of the warmest endorsers
of echinacea, C. S. Chamberlain (who later became the president
of the Eusoma Pharmaceutical Company), complains
that he has been unable to interest regular physicians in
the remedy. He reviews the statements of previous authors
and reports eight cases of infection, only two being acute or
extensive, in which he used it with asserted success.
In view of the lack of any scientific scrutiny of the claims
made for it, echinacea is deemed unworthy of further consideration
until more reliable evidence is presented in its
favor.
REFERENCES
Meyer, H. F. C.: Eclectic Med. Jour., 1887; Goss: Chicago Med.
Times, 1888; Hages: Eclectic Med. Jour., 1888; Shelly: Medical Gleaner,
1894; Lloyd, C. G.: Eclectic Med. Jour., 1897; Lloyd, J. U.: Eclectic
Med. Jour., 1897; Lloyd, J. U.: Pharm. Review, xxii, 9–14; Schnitz,
Elsie M.: Wis. Med. Recorder, 1898, ii, 202; White, J. N.: Texas Med.
News, 1898, viii, 110–113; Stinson, J. C.: Therap. Gazette, 1900; Hale,
E.: Lancet-Clinic, March, 1901; Thielen, B. F.: Echafolta, Its Uses in
Dental Surgery, Dental Reg., 1903, vii, 462–465; Gorse, C. A.: New
Albany Med. Herald, 1903–4, xxii, 384; Chamberlain, C. S.: Louisville
Monthly Jour. Med. & Surg., 1904–5, xi, 219–223; Lancet-Clinic, 1905,
M. S., liv, 279–283; Ellingwood, F.: Therap. Gazette, 1905, 3, S., xxi,
298–300; French, J. M.: Med. Brief, 1905, xxxiii, 537; Mathews, A. B.:
Georgia Pract., 1905, i, 137–140.—(From The Journal A. M. A., Nov.
27, 1909.)
ECHTISIA, ECTHOL AND ECHITONE
Report of the Council on Pharmacy and Chemistry
Echtisia (Wm. S. Merrell Chemical Co.), Ecthol (Battle
and Co.) and Echitone (Strong, Cobb and Co.) are proprietary
preparations each of which is alleged to contain
echinacea as its chief constituent. In 1909 the Council
examined into the claims made for echinacea. This drug
has been claimed to be a “specific” for rattlesnake bite,
syphilis, typhoid fever, malaria, diphtheria and hydrophobia.
Enthusiasts have credited it with equally certain curative
effects in tuberculosis, tetanus, and exophthalmic goiter and
with power of retarding the development of cancer. Of
course there is no reliable or trustworthy evidence to substantiate
these claims. Echinacea is not often prescribed
under its own name, but is employed as an ingredient in
proprietary preparations mixed with other little-used or
obsolete substances. Thus Echtisia is said to contain
echinacea, wild indigo, arbor vitae and poke root; Ecthol,
to have echinacea and arbor vitae; Echitone, to consist of
echinacea, pansy and blue flag. Naturally the manufacturers
of such proprietaries make use of all available optimistic
reports in promoting their sale, while each manufacturer
ascribes special and peculiar virtues to the combination
represented in his particular preparation. The
Merrell Chemical Company claims that baptisia (wild
indigo) is a “destroyer” of devitalizing elements in the
blood “a vitalizer of the blood as well”; that thuja (arbor
vitae) is a “perfect antiseptic and a generator of vital force
in disorganized tissues,” and that a long list of diseases,
including diphtheria, syphilitic sciatica and gonorrheal
rheumatism, “are all more or less amenable to full doses”
of phytolacca (poke root). Strong, Cobb and Co. maintain
that Iris versicolor (blue flag) is “one of the most powerful
excitants of the biliary, salivary and pancreatic secretions,”
and that the “principal sphere of action of Viola tricolor
[pansy] is in the gastro-intestinal canal and the skin.”
There is no satisfactory evidence that the claims for any
of these substances are any more reliable than those for
echinacea. Notwithstanding, Strong, Cobb and Co. claim
for Echitone “not only the virtues of its constituent parts,
but a wider field and a particular therapeutic value of its
own”; Battle and Co., maintain that Ecthol is the “‘Ideal
Corrector’ of depraved conditions of the fluids and tissues,”
while the Merrell Company urges the virtues of Echtisia
in a list of diseases ranging from acne to appendicitis and
from gangrene to rattlesnake bite. The Council refused
recognition to these three products and directed publication
of reports to call attention to the exaggerated, unwarranted
and often utterly absurd claims made for these and similar
preparations.—(From The Journal A. M. A., Jan. 2, 1915.)
ERGOAPIOL[N]
Abstract of Report of the Council on Pharmacy and Chemistry
Ergoapiol (Martin H. Smith Co., New York) is a mixture
put up in capsules, each of which is said to contain
Apiol (Special M. H. S.) | 5 | grains |
Ergotin | 1 | grain |
Oil Savin | 1⁄2 | grain |
Aloin | 1⁄8 | grain |
Examination discloses the fact that, contrary to the claim
made, each capsule, instead of containing 5 grains of apiol,
really contains some liquid preparation of the type of
oleoresin of parsley seed. Ergoapiol is recommended (on
the label) for such diseases as “Amenorrhea, Dysmenorrhea
and other Menstrual Disorders,” while a circular enclosed
in the package contains many suggestions that may be
counted on to lead to its indiscriminate and uncritical use.
Ergoapiol is an unscientific, shot-gun mixture of drugs
having widely different therapeutic effects. Where the action
of parsley is desired, the effects of ergot would ordinarily,
be contra-indicated; furthermore, neither aloin or savin
would be called for in conditions that demanded the effects
of apiol. It would be impossible to predict the action of a
mixture of this kind in the varying conditions for which
its use is advised by the manufacturers. To combine four
drugs of dissimilar action in fixed proportions for the routine
treatment of conditions which have little in common except
that they involve the female generative organs, is unscientific
and absurd. The Council refused admission to Ergoapiol.—(From
The Journal A. M. A., Dec. 12, 1914.)
ERPIOL (DR. SCHRADER)
Report of the Council on Pharmacy and Chemistry
The original rules of the Council governing the acceptance
of articles have recently been modified, particularly by adoption
of Rule 10, which reads:
“Unscientific and Useless Articles.—No article will be admitted which,
because of its unscientific composition, is useless or inimical to the best
interests of the public or of the medical profession.”
In view of these modifications, the Council is reconsidering
the articles already accepted with the view of determining
their compliance with the rules as amended. In line
with this the Council reconsidered Erpiol (Dr. Schrader),
manufactured by the William S. Merrell Chemical Company,
and from the evidence given below concluded that one of
the constituents, gossypin, is inert and its use unscientific.
The Council therefore voted that Erpiol (Dr. Schrader)
be omitted from New and Nonofficial Remedies and authorized
publication of the following report.
W. A. Puckner, Secretary.
Erpiol
In consequence of the more thorough scrutiny now given
by the Council to the therapeutic value of the remedies
admitted to New and Nonofficial Remedies, the Council has
reconsidered Erpiol (Dr. Schrader), previously accepted for
New and Nonofficial Remedies. Erpiol (Dr. Schrader) is
the name applied to capsules containing apiol, ergotin and
gossypin, which are sold as an emmenagogue. The first two
ingredients have a recognized value in the treatment of
diseases of the female generative organs. The third, gossypin,
is a preparation from cotton-root bark, belonging
to the somewhat indefinite class of pharmaceutical preparations
known as resinoids.
Cotton-root bark (Gossypii radicis cortex, U. S. P.) has
been credited by some with pharmacologic and therapeutic
properties, similar to ergot, especially in its action on the
uterus; experiments on pregnant animals do not confirm
this view. Most authorities on gynecology either make no
reference whatever to the drug or ascribe little or no value
to it. The preparations from the dried bark are inert.
From reports made to him, Professor J. U. Lloyd concluded
(Eclectic Med. Jour., 1876, xxxvi, 545) that a prime
fluidextract of fresh cotton-root bark is an active therapeutic
agent and deserving the attention of the medical profession,
while that of the dry bark is inert and worthless. The
gossypin on the market is made from the dried bark.
Professor Lloyd, who is considered an authority on eclectic
medicine, says: “Were it left to me to admit or exclude it,
by reason of its therapeutical position, I should exclude it,
because, in my opinion, it has never been demonstrated, in
clinical practice, to be worthy of any therapeutic recognition
whatever.”
As the available evidence indicates that gossypin is an
inert preparation, Erpiol (Dr. Schrader) was considered in
conflict with Rule 10 and the Council has therefore voted
that it be deleted from New and Nonofficial Remedies.—(From
the Journal A. M. A., June 3, 1911.)
FALSE UNICORN (HELONIAS)
Report of the Council on Pharmacy and Chemistry
The Council voted to refuse to recognize false unicorn as
a non-proprietary article and the following statements, submitted
by a subcommittee, were ordered published.
W. A. Puckner, Secretary.
False Unicorn-Helonias
Helonias dioica, or more properly Chamælirium luteum,
is a plant, preparations of which enter into various proprietary
mixtures for diseases of the female pelvic organs.
In the advertisements of these preparations it is usually
credited with hemostatic powers and is asserted to be a
uterine tonic.
There is practically no reference to this drug in reliable
medical literature, and as there is no evidence worthy of
credence to support the claims made for it, the drug was not
considered deserving of a place in the Pharmacopeia. Hence,
it may be regarded as a drug not worthy of attention of
physicians.—(From The Journal A. M. A., Nov. 27, 1909.)
FORMUROL
Report of the Council on Pharmacy and Chemistry
Formurol, Citrocoll and Aspirophen were submitted to
the Council by the Cellarius Company of San Francisco.
The manufacturers having failed to substantiate the claims
they make for these products, the Council has voted that
the preparations be refused recognition. The Council also
authorized the publication of the following report, which
deals particularly with one of the preparations—Formurol.
W. A. Puckner, Secretary.
Formurol is the product of the Chemische Fabrik Falkenberg,
Falkenberg-Gruenau, near Berlin, Germany. The
Cellarius Company, San Francisco, acting as selling agents
for the United States, submitted Formurol (along with
Aspirophen and Citrocoll, also made by the same firm) to
the Council, with the statement that it is “hexamethylentetraminsodium-citrate,”
and that it has the following composition: “C6H7O7Na.C6H12N4.”
Zernik,46 who examined these products, reported that
Aspirophen, Citrocoll and Formurol do not have the composition
that is claimed for them by the Fabrik Falkenberg.
Formurol, he states, is not a definite chemical compound, but
a mixture of hexamethylenamin and sodium citrate. The
agents were advised of this fact by the Council and were
asked to submit evidence to substantiate their claims. No
such evidence was submitted.
Since a compound having the composition that is claimed
for Formurol is theoretically possible, the Council requested
that the product be examined in the Association Laboratory
to determine whether it still was the simple mixture reported
by Zernik, or whether, perhaps, it now possessed the formula
claimed for it. The following report was made by the
Association chemists:
Formurol, as submitted to the Council, was in the form
of tablets weighing about 1 gm. each and appeared to be
composed of a fine white substance interspersed with
some transparent particles. The tablets were readily
soluble in water, were odorless and possessed a slightly
acid taste. The aqueous solution responded to tests for
hexamethylenamin, citrate and sodium. To determine
whether hexamethylenamin was present in the free or the
combined state, the method of Zernik was employed. This
consists in the extraction of Formurol with chloroform,
which dissolves out hexamethylenamin, leaving insoluble
sodium citrate. As the use of the solvent, chloroform,
would seem to preclude decomposition of such a hypothetical
compound as “hexamethylenamin-sodium-citrate,”
the extraction of hexamethylenamin from Formurol may
be taken to demonstrate its presence in the free state.
That Formurol is not a compound of hexamethylenamin,
but a mixture of hexamethylenamin and sodium citrate,
was further indicated by the appearance of the crushed
tablets described above. Further, on the low-power
microscope the powder was found to be composed of transparent
crystals and white opaque particles which appeared
to be masses of minute crystals. When treated with
chloroform the transparent crystals dissolved, leaving the
white masses intact, demonstrating the presence of two
distinct substances, one soluble and the other insoluble in
chloroform. It having been demonstrated that the residue
obtained by evaporation of chloroform could not be
weighed as hexamethylenamin, due to enclosed chloroform,
the amount of this substance in the residue was
determined.
The method used has been described in the Report of
the Chemical Laboratory of the American Medical Association,
Vol. I, p. 55, and depends on the decomposition of
hexamethylenamin by means of sulphuric acid to form
ammonium sulphate and formaldehyd. From this solution
the ammonia is liberated, distilled and determined by
titration and from the ammonia found the amount of
hexamethylenamin is calculated. By this method Formurol
was found to contain (a) 35.42 per cent. and (b) 35.32
per cent., or an average of 35.37 per cent. hexamethylenamin.
The residue insoluble in chloroform was shown
to consist essentially of disodium hydrogen citrate by
determining the amount of sodium (Na) contained in
Formurol. The percentage of sodium calculated from the
amount of sodium sulphate found was (a) 11.38 per cent.
and (b) 11.20 per cent., or an average of 11.29 per cent.,
equivalent to 62.50 per cent. disodium hydrogen citrate.
As a check on this determination, the amount of
material contained in Formurol which is insoluble in
chloroform was determined. It was found to be (a) 63.23
per cent. and (b) 63.49 per cent., making an average of
63.36 per cent., and thus agreeing fairly well with the
results obtained when the sodium content was assumed to
be disodium hydrogen citrate. From this analysis it
appears that Formurol is not a definite compound of
hexamethylenamin and sodium citrate, but instead is a
mixture of these substances consisting approximately of
hexamethylenamin 35.37 per cent. and sodium acid citrate
(disodium hydrogen citrate) 63.36 per cent., practically a
mixture of 1 part hexamethylenamin and 2 parts sodium
acid citrate. These results agree with those reported by
Zernik47 and show that the product now, as then, is not
true to claims.
In view of the findings of the laboratory, it is recommended
that Formurol be refused recognition. As the exploitation
of well-known remedies under false and misleading names is
detrimental to the progress of medicine, it is recommended
that publication of this report be authorized.
[Editorial Note: This report illustrates once more the
value of the Council on Pharmacy and Chemistry and the
Chemical Laboratory to the medical profession. Before the
Council was organized there was no agency to protect the
physician’s interests in the matter of pharmaceuticals. Under
the old régime Formurol would have been heralded as a
new “synthetic” of the most approved made-in-Germany
type—and the claims would have gone unchallenged. To-day
its status is made clear and the profession is informed.
Only those who have closely studied the question can
realize what a wonderful power for commercial probity the
Council has proved. Under the laissez faire system of the
past, many large pharmaceutical firms gave little attention
to the accuracy of the claims made for their products. If
the advertising gave good “pulling” results, that was all
that was asked or expected. Within the past five years a
wonderful change has taken place in this regard, and firms
of the better class have so modified their advertising as
to make it not only conservative in tone, but to approximate
scientific accuracy.]—(From The Journal A. M. A., Jan. 21,
1911.)
GASTROGEN TABLETS
Report of the Council on Pharmacy and Chemistry
The Bristol-Myers Co., Brooklyn, N. Y., sells Gastrogen
Tablets which are described as “A Neutralizing Digestive”
to be “used in connection with Sal Hepatica.” Sal Hepatica,
it will be remembered, is another product of the Bristol-Myers
Company and has been the subject of previous
unfavorable comment. The label on a recently purchased
package of Gastrogen Tablets contains the following:
“For gastric distress, weak stomach and dyspepsia, one to two tablets
after eating; repeat in half an hour if needed.
“Also indicated in nausea, flatulence, sour stomach and heartburn.”
While these recommendations sound as if they were
addressed to the public, Gastrogen Tablets are advertised
in medical publications and hence come within the scope of
the Council. Gastrogen Tablets are said to be composed of
pepsin, calcium carbonate, calcium phosphate and “aromatics.”
As each tablet, according to the label, contains 7 grains of
calcium carbonate (chalk), the recommended dosage would
in most cases be sufficient to neutralize the gastric fluids in
the stomach and would thus tend to prevent the pepsin from
exerting its digestive effects. The means adopted to relieve
one symptom of dyspepsia, in other words, defeats the action
of the means for relieving the indigestion. The fact is that
patients who need an antacid do not need pepsin, while those
who need pepsin will be harmed by the administration of an
antacid. Gastrologists hold that, except in rare cases, the
evidence tends to show that wherever there is a sufficiency
of hydrochloric acid there is a sufficiency of pepsin. When
pepsin is lacking it should be administered along with hydrochloric
acid to make it effective. The Council voted that
Gastrogen Tablets be refused recognition.—(From The
Journal A. M. A., Dec. 12, 1914.)
GLYCO-HEROIN, SMITH
Report of the Council on Pharmacy and Chemistry
The following report was submitted to the Council by a
referee and publication authorized.
W. A. Puckner, Secretary.
Glyco-Heroin, Smith (Martin H. Smith Co., New York)
is marketed in a showy “patent-medicine” type of package,
the label on which announces the presence of 1⁄2 grain of
heroin to the fluidounce and admits the presence of 3.5 per
cent. alcohol, an active ingredient that is not discussed in
any way in the literature sent out by the manufacturer.
The composition of Glyco-Heroin, Smith, is given as follows:
“Each teaspoonful represents: Heroin 1⁄16 grain,
White Pine Bark 31⁄2 grains, Ammonium Hypophosphite 3
grains, Balsam Tolu 1⁄4 grain, Hyoscyamus 1 grain, Glycerin
Q. S.” The alcohol is not mentioned in the formula.
The advertising matter says of the merits of the formula:
“Despite the fact that heroin, which is universally recognized as an
invaluable respiratory sedative, is a conspicuous element of Glyco-Heroin,
Smith, the other constituents, henbane, ammonia hypophosphite, balsam
tolu and white pine bark are factors of no less importance; indeed, it
is through the concerted action of its several ingredients that the preparation
proves so notably beneficial in the class of affections in which
it is indicated. The constantly increasing popularity of the preparation
in the treatment of respiratory affections is the best adducible evidence
of its value in such disorders.”
The absurdity of this assertion will be appreciated on comparing
the nature, quantities and activities of the several
ingredients. Thus, while heroin, a potent habit-forming drug,
is present in unusually large proportions, tolu, an innocuous
or comparatively harmless product, is said to be represented
by 1⁄4 grain, a relatively small quantity, hardly sufficient to
impart even a distinctive taste or flavor. Ammonium hypophosphite,
in the amount said to be present, may be considered
to be practically useless, while the dose of hyoscyamus, an
additional narcotic, is fairly large. The white pine bark
present is probably as active as would be a corresponding
amount of white pine shavings or of turpentine sufficient to
give the preparation a slight odor. The vehicle, glycerin, is
claimed to be “notably advantageous,” but not a word occurs
in the discussion by the manufacturer in regard to the presence
of alcohol, which is certainly quite as active medicinally
as the balsam of tolu and contributes fully as much to the
flavor or taste of the preparation as does the white pine bark.
In prominent type on the outer label of the trade package
we are told that the preparation is intended for the treatment
of “COUGH, ASTHMA, PHTHISIS, PNEUMONIA, BRONCHITIS,
LARYNGITIS, WHOOPING-COUGH AND KINDRED INFECTIONS.” In
much smaller type: “Glyco-Heroin (Smith) is distinctly a
product designed expressly for the use of physicians.” The
circular included with the trade package, however, bears
statements which would tend to encourage self-drugging by
the layman, and in view of the manner in which the preparation
is exploited are undoubtedly intended to do so. For
instance:
“Bronchitis.—In the acute form of bronchitis, Glyco-Heroin (Smith)
acts most happily. It tends to diminish the congestion and inflammation
of the lining of the air passages, relieves the pain and institutes
repair....
“Phthisis.—In the treatment of the cough of phthisis, Glyco-Heroin
(Smith) is used with the most gratifying results. It checks the night
sweats, acts favorably upon the reflexes, increases expectoration and
induces refreshing sleep.
“Asthma.—The preparation diminishes the intensity of the paroxysms
and lengthens the intervals between their recurrence. By the administration
of the preparation, asthmatic attacks can frequently be aborted.
“Pneumonia.—In the initial stage of pneumonia, the preparation
exercises a calming, antipyretic and sedative effect. In the latter stages
of the disease, the analgesic and expectorant properties of the product
are well displayed.
“Whooping-Cough.—Administered in doses of from five to ten drops
this preparation affords surprisingly satisfactory results. The cough
rapidly loses its spasmodic character and the frequency of the paroxysms
is considerably diminished.”
How cruelly misleading the literature put out by the manufacturer
of this nostrum is, will be apparent from a comparison
of the rather large dose of heroin in a teaspoonful
of the nostrum and the directions on the package that:
“The adult dose of Glyco-Heroin (Smith) is one teaspoonful repeated
every two hours or at longer intervals, as the case may require.
“Children of 10 or more years, from a quarter to a half-teaspoonful.
“Children of 3 years or more, 5 to 10 drops.”
A WICKED FALSEHOOD
Included in much of the advertising matter that has been
put out is the bare-faced untruth that the preparation does
not produce narcotism or habituation. Here is a quotation
from an undated circular:
“Glyco-Heroin (Smith) is decidedly preferable to preparations containing
codeine or morphine, by reason of the fact that it does not produce
narcotism, constipation, gastric disturbance nor habituation, even
though its administration be protracted.”
That this assertion is not in keeping with facts is evidenced
by the recent report of a study on the sale and use
of heroin made by the U. S. Department of Agriculture.
From the information gathered it appears that the sales of
heroin and heroin-containing preparations have increased
greatly, particularly in those states which have rigid laws
preventing the indiscriminate sale of morphin and cocain.
Investigation of the subject establishes the fact that many
drug victims who formerly used morphin and cocain, and who
under the new laws find it difficult to obtain these substances,
have begun using heroin, the sale of which is not as yet carefully
restricted under state laws. The drug is said to be
fully as dangerous as morphin, and by many is held to be
much worse, for the reason that it occasionally kills the
victim outright, and its habitual use is far harder to overcome
than that of other drugs.
Phillips,48 in discussing the prevalence of the heroin habit,
reports, among others, the case of a physician aged 60 who
began to take heroin because he suffered from a chronic
cough and thought there was no danger of habit from the use
of this drug because he believed the statements of various
manufacturing firms who claimed that there was no danger
of habit.
In a pamphlet now being distributed to the medical profession,
entitled, “Glyco-Heroin (Smith), an exposition of its
components together with references to its value in the treatment
of Bronchitis, Cough, Cough of Phthisis, Laryngitis,
Pneumonia and allied disorders of the Respiratory Tract,”
the several alleged uses of the nostrum in the treatment
of cough, “Regardless of the nature of its underlying cause,... whether
of recent origin or of long duration,” are discussed
at length, and eminent practitioners with degrees
extending the width of the printed page are quoted in support
of the statements made. While it may be permissible for a
theoretically trained medical tyro who lays claim to the right
of appending the abbreviations M.A., M.D., D.C.L., L.R.C.P.
to his name to laud a heterogeneous habit-forming cough-syrup
like Glyco-Heroin, Smith, similar testimonials from a
man entitled to append Ph.G., M.S., M.D. to his name makes
one doubt the value of the training, either scientific, pharmaceutical
or medical, that has been given the poor unfortunate
who, according to his own statements, indiscriminately doses
a female patient of 7 and a male patient of 40 with huge
doses of heroin every two, four or six hours.
The danger of contributing to the spread of the heroin
habit by the use of preparations of this type is indicated by
an editorial in The Journal of the American Medical Association,49
which points out that although heroin and its hydrochlorid
have been in use but a few years they have already
established themselves among the habit-forming drugs and
have become sufficiently conspicuous in this respect to awaken
the thinking public to the deplorable results for which they
may become responsible. Phillips,49 in the article mentioned
above, quotes Petty, who reports that in the last 150 cases
of drug habit coming under his care he saw eight cases of
heroin addiction. Three of these were initial cases; in one
the patient had been cured of the opium habit, but following
an operation heroin was prescribed, and the habit followed.
The remaining four patients purposely substituted heroin for
morphin, to which they had been addicted.
THE GROWTH OF HEROIN ADDICTION
The imminent danger of substituting heroin for either
morphin or cocain is shown by the fact, reported by the U. S.
Department of Agriculture, that during the early months of
1913 the coroner’s office in Philadelphia County, Pa., held
inquests on five sudden deaths from heroin poisoning. In
each case the victim was a heroin fiend and took an overdose.
Drug fiends are apparently able to consume relatively large
quantities of morphin or cocain, but any sudden and material
increase in the amount of heroin taken is liable to prove
fatal. As indicating the wide sale of this substance, it is
known that one druggist in Pennsylvania whose store is
located in an undesirable section of his city has been buying
heroin tablets in 25,000 lots.
GLYCO-HEROIN, SMITH, A “PATENT MEDICINE”
The popularity of Glyco-Heroin, Smith, as a household
nostrum is suggested by the fact that one of the larger
department-store type of drug-stores in the city of Philadelphia
lists this preparation in its “patent-medicine” catalogue
at $1.75 per bottle and sells it freely to all who care
to buy. This is due to the fact that Pennsylvania, like many
other states, does not include heroin in the prohibited list of
habit-forming drugs that can be supplied only on physicians’
prescriptions.
To what extent Glyco-Heroin, Smith, is responsible for
developing the rapidly growing heroin habit is of course
problematic. It is reasonable, however, to suppose that a
preparation, each teaspoonful of which contains so large a
dose of heroin as does this nostrum, when taken as repeatedly
and as indiscriminately as is directed by the manufacturer,
would offer possibilities for harm sufficient in number to
induce the thinking medical practitioner to avoid its use
altogether and at least to suggest to even the most commercial
dabbler in the healing art the desirability of carefully considering
its potency for harm before endorsing its use in the
treatment of “cough and kindred affections.”—(From the
Journal A. M. A., June 6, 1914.)
GLYCO-THYMOLINE
Report of the Council on Pharmacy and Chemistry
The Council, having voted that Glyco-Thymoline be
refused recognition, authorized publication of the following
report.
W. A. Puckner, Secretary
Glyco-Thymoline (Kress and Owen Company, New York)
is a typical example of a “patent medicine” advertised to the
public through the doctors. Bottles of the mixture with the
name blown in the glass are issued to physicians for distribution
to patients, and the circular which comes around
the bottle more or less directly recommends it for use in
almost every form of infectious disease.
COMPOSITION AND VARYING FORMULAS
Different formulas for Glyco-Thymoline have appeared.
At one time it was said to contain:
“Sodium 24, Boric Acid 4, Benzoin 4, Acid Salicylic 0.33, Eucalyptol
0.33, Thymoline 0.17, Betula Lenta 0.08, Menthol 0.08, Pini Pumilionis
0.17, Glycerin and solvents, q.s.”
Another formula, which appeared about the same time,
was:
“Benzo-Salicyl. Sod. 33.33, Eucalyptol 0.33, Thymol 0.17, Salicylate
of Methyl from Betula Lenta 0.16, Pini Pumilionis 0.17, Glycerin and
solvents q.s.”
A later formula was like the second except that it included
“Menthol, 0.08.”
Analysis in the chemical laboratory of the American Medical
Association showed that Glyco-Thymoline contained
borax, but no boric acid; sodium salicylate, but no salicylic
acid; sodium benzoate, but no benzoin; the compound benzo-salicyl.
sod. could not be determined, but a mixture of sodium
benzoate and sodium salicylate was demonstrable.50 Later
Puckner pointed out51 that while such a combination as benzo-salicylate
of sodium is known, it could not possibly be present
in Glyco-Thymoline because the alkalinity of this mixture
would decompose the compound. As the manufacturers
evidently recognize that false formulas can no longer be
made plausible, only vague statements as to the composition
are now offered.
Two points should be noted in this connection:
1. Glyco-Thymoline conflicts with Rule 1 of the Council
on Pharmacy and Chemistry, which declares that no article
shall be accepted for inclusion with New and Nonofficial
Remedies unless its composition be furnished.
2. No matter which published formula be accepted as correct,
it is at best a weak antiseptic. The antiseptic ingredients
present cannot act as germicides in the strength in
which they are used, or in the alkaline solution on the
unique virtues of which the circular lays so much stress
(“the one antiseptic solution based on the alkalinity and
saline strength of normal blood”). As shown by Verhoeff
and Ellis,52 undiluted Glyco-Thymoline does not kill Staphylococcus
aureus in four hours. It evidently, they say, “could
have but little if any greater therapeutic value than sterile
salt solution.”
DANGEROUS RECOMMENDATIONS
In Diphtheria: “Case-reports” in the advertising pamphlet
describe the treatment of diphtheria with Glyco-Thymoline.
It is surely unnecessary to point out that, whatever the possible
merits of Glyco-Thymoline or its ingredients, they are utterly
irrelevant here. But let a “case-report” be quoted:
“............., M.D., states: ‘I have many an interesting
story of Glyco-Thymoline. I just finished up a family in which I
was treating five cases of diphtheria—two of which presented diphtheritic
membrane in nasal cavity. I decided not to use antitoxin in
these cases. I used only the regular constitutional treatment and
Glyco-Thymoline as a local antiseptic. I believe the Glyco-Thymoline
worked wonders. My cases are all now in good health, with no after
troubles. I think it an ideal antiseptic for every trouble in nose and
throat.’”
Words of denunciation fall flat before the complacent self-revelation
of the physician who “decided not to use antitoxin.”
Surely if any other physicians have been misguided
by this example, there must be many another “interesting
story of Glyco-Thymoline” to tell—not to speak of other
families that have been “finished up.”
In Ophthalmia Neonatorum: We gain from the same advertising
pamphlet the following information on prophylaxis:
“The treatment in the past has consisted of instillation of silver
nitrate, boric acid, salts of mercury, nucleinated salts of silver and
mercury, etc. ..., but these agents have proved to be failures as an
absolute specific.... During the past few months experiments have
demonstrated the efficacy of a new mode of treatment that is both
rapid and thorough, and devoid of danger in its use. This method consists
of thorough irrigation of the eyes in fully developed cases of the
disease with a solution of Glyco-Thymoline.”
At the very best, Glyco-Thymoline is a weak, a very weak
antiseptic—not a germicide. To assert, or even to imply,
that it is superior to the well-tried and efficacious Credé
method of treatment for ophthalmia in the new-born is cruelly
wicked.
In Consumption: This from the same pamphlet:
“The indifference of phthisical patients toward the maintenance of
sanitary conditions is proverbial.
“That the environment of all such patients should be absolutely
aseptic both for the good of the patient and for the welfare of those
who are brought into contact with them is a well-established fact.”
It is, instead, an ill-established fiction. To talk about
maintaining an “absolutely aseptic” environment under any
practical conditions of daily life is to talk nonsense; the
thing is impossible, even were it desirable. But, not to be
distracted from the main issue by subsidiary falsehoods:
“In Glyco-Thymoline we have an antiseptic which, while mild and
soothing ... is still a powerful agent for promoting asepsis, and a
potent factor in the maintenance of sanitary environment in the sick
room.
“Inhaled from a vaporizer or a fine spray atomizer, it will loosen the
mucus in a marvelous manner and in a wonderfully short time, shorten
the paroxysms of coughing to a marked degree, at the same time
reducing the danger of contagion to a minimum.”
And this is the preparation, it will be remembered—this
“powerful agent for promoting asepsis”—which, when applied
in undiluted strength, was unable to kill Staphylococcus
aureus in four hours!
It would be a waste of space to cite further evidence to
show that the advertising of Glyco-Thymoline is in conflict
with Rule 6 of the Council, which provides that no article
shall be accepted “concerning which the manufacturer or his
agents make unwarranted, exaggerated or misleading statements
as to the therapeutic value.” It is further in conflict
with Rule 4, against indirect advertising by means of the
label, package or circular accompanying the package.
CLAIMS TO ORIGINALITY
Hatcher and Wilbert have pointed out that from a therapeutic
point of view the composition of Glyco-Thymoline is
based on the formula of the widely known “compound solution
of sodium borate,” or Dobell’s solution. For the phenol
in the original, a mixture of antiseptic acids and volatile
oils has been substituted.
SUMMARY
Glyco-Thymoline is in conflict with Rules 1 and 4 of the
Council on Pharmacy and Chemistry, because of its indefinite
composition and the method of advertising it to the
public. It is in conflict with Rules 10, 6 and 8, in that it
is an unscientific, shot-gun mixture sold under unwarranted
therapeutic claims and under a misleading name. Altogether
it must be considered an unscientific heterogeneous mixture,
in which a few valuable ingredients are hidden by the useless
shrubbery which surrounds them.—(From The Journal
A. M. A., Oct. 10, 1914.)
GLYCOZONE
Report of the Council on Pharmacy and Chemistry, with
Comments
A number of specimens of Glycozone purchased in the open
market were examined by a subcommittee. The product
was found to be a mixture of approximately 90 per cent.
glycerin, 5 per cent. glyceric acid, a small amount of water
and traces of undetermined matter. The absence of hydrogen
peroxid or other peroxids was demonstrated.
In its report the subcommittee held that: (1) The name
of the product is objectionable and misleading; (2) the statements
made in regard to its composition also are misleading;
(3) the claims for its therapeutic value are exaggerated
and untrue. Since the objectionable statements have been
given wide publicity among physicians as well as among
the laity, the subcommittee recommended that attention
should be called to the matter in The Journal.
The report of the subcommittee was adopted by the
Council.
W. A. Puckner, Secretary.
Comment:—While the name gives the impression that
ozone or some similar substance is an essential constituent
of Glycozone, or else that the preparation is a compound
or derivative of ozone, and while the earlier advertisements
stated that Glycozone was “glycerine combined with ozone,”
the examination made by the Council shows that there is
no basis of fact for such inferences.
In the advertisements the “chemical formula” C3H6O4 +
C3H8O3 appears under the word Glycozone. From the
Council’s report it is apparent that C3H6O4 stands for
glyceric acid and the C3H8O3 for glycerin, and that these,
therefore, indicate the chief constituents of Glycozone. Few,
doubtless, would recognize the first formula as being that
of a glyceric acid, a product practically unknown in medicine,
nor would many associate glycerin with the second.
The evident intent is that physicians should accept the
formula as a badge of respectability.
According to the label on a trade package, Glycozone is
“prepared only by Charles Marchand, chemist,” and is {“}an
absolute cure for dyspepsia, catarrh of the stomach, ulcer of
the stomach, heart-burn,” etc. The label further reads:
“This remedy is positively harmless. By destroying the
microbian element in the stomach it prevents the fermentation
of food and stimulates digestion.” An examination of
medical literature fails to reveal any basis for these claims.
While glycerin possesses some antiseptic properties, it is
evident that the glycerin which constitutes 90 per cent. of
this remedy is not the agent that gives the glycozone such
phenomenal virtues. General literature contains nothing that
would indicate that glyceric acid in any quantity, with or
without glycerin, possesses these miraculous properties. If
by “microbian element” is meant microbic organisms, the
statement is without foundation. There is nothing in this
product which possesses these bactericidal powers.
The circular which accompanied a trade package envelops
the preparation in an air of mystery. Derivation from, or
close relation to, ozone and hydrogen peroxid is vaguely
hinted at, without definite assertion. Thus, the chief therapeutic
properties of glycozone and hydrozone are compared
as follows:
“Hydrozone instantly destroys the microbian element, leaving the
tissues beneath in a healthy condition.”
“Glycozone acts more slowly, but not less certain as a stimulant to
healthy granulations.”

Much-reduced photographic reproduction of one of the older Glycozone
advertisements. Attention is directed to the false claim that this
nostrum is “glycerin combined with ozone.”
There is no similarity between the action of hydrozone,
which is a hydrogen peroxid preparation, and glycozone,
which consists of a mixture of glycerin and glyceric acid.
The representation is false and misleading. The following
statement, also, is an unwarranted exaggeration of the facts:
“As an internal medication in fermentation of food, catarrhal and
inflammatory conditions of the stomach, and intestinal disorders, its
action is prompt and effective, giving immediate relief to the patient.”
The following is another illustration of the vague statements
made: After asserting that Glycozone is hygroscopic
and that it will deteriorate by absorption of water unless
securely corked, it is stated that “Its healing properties
increase with age.” Whatever mysterious ingredient there
may be present in this mixture to justify the statement that
the healing properties increase with age can only be conjectured.
To humbug the patient further, the circular advises
him to use only a “silver, glass or hard rubber spoon.”—(From
the Journal A. M. A., June 5, 1909.)
GARDNER’S SYRUP OF HYDRIODIC ACID
Report to the Council on Pharmacy and Chemistry
The following report on Gardner’s Syrup of Hydriodic
Acid was submitted to the Council by a subcommittee:
This product was first taken under consideration in
February, 1906. Reference to several committees was necessary,
on account of the peculiar claims for the pharmaceutical,
and especially the therapeutic, superiority of this
preparation. At this time, as the Council did not have the
necessary facilities for investigating therapeutic claims, the
product was approved by the Council.
Since this time, however, the manufacturers have laid
especial stress in their advertisements on some highly
improbable claims, stating, for instance, that this Syrup of
Hydriodic Acid possesses “all the advantages, with none of
the objectionable symptoms caused by potassium iodid, or
other forms of iodin medication.” To one with even an
elementary knowledge of chemistry, the absurdity of this
statement should be evident. The alkaline reaction of the
tissues makes it impossible that hydriodic acid should persist
as such in the body. In fact, the iodin must circulate
in precisely the same form, whether administered originally
as potassium iodid or as hydrogen iodid. The qualitative
identity of the therapeutic actions is further proof of this
fact, were such needed.
Since the most important objectionable symptoms of iodid
medication arise after the absorption of the drugs, and since
hydrogen iodid is conceded to be readily absorbed, it is evident
that these symptoms must be equally liable to occur
with hydrogen iodid as with potassium iodid, provided that
equivalent doses of iodin are administered. An apparent
difference in clinical results would arise if one drug were
habitually given in smaller doses than the other. Since,
however, the iodin is present in the body in precisely the
same form, whether it is administered as a hydrogen iodid
or potassium iodid, it is evident that a given degree of
therapeutic effect would correspond to an identical tendency
to iodism, whichever drug was used. If, as appears to be
the case, the use of hydriodic acid is commonly restricted
to those cases in which only minimal doses of iodin are
required, the relative infrequency, or even absence of symptoms
with such doses would not prove that the drug itself
is less apt to cause them than is the potassium salt.
These facts are in reality self-evident; but since the
Council now has proper facilities for obtaining the views
and experiences of clinicians, it voted to submit the statement
in question to its staff of clinical consultants, and
to be guided by their advice.
OPINIONS OF THE CLINICAL STAFF
The following is an epitome of the replies of the eleven
members of this staff who had used the article or who
expressed an opinion to the questions sent out by the Council:
1. Query: “Do you think it possible that such a
preparation could be devoid of the usual effects of iodin
preparations?”
Eight reply that they consider this, a priori, impossible;
three stamp the statement as highly improbable, but do not
care to say that it would be impossible. One of the correspondents
remarks: “While distinctly taking the position
that under many conditions we must accept clinical results
which we find not explainable by our theoretical knowledge,
where the conditions are so simple as in this case and where
we know that the iodin, whether administered as hydrogen
iodid or potassium iodid, must behave in the same way,
after absorption, I believe that no properly educated and
correct thinking physician can or will, after due consideration,
fail to reject the claims of superiority made by the
proprietors of this preparation.”
2. Query: “Would you consider it necessary to make
clinical experiments to settle this question?”
Seven of the correspondents consider this superfluous; four
of these have had some experience with the article. Four,
who have not used this product, consider a clinical test
advisable. Under Query 3 we discuss the results of such
tests.
3. Query: “When using Gardner’s Syrup of Hydriodic
Acid, have you ever noticed from it any of the objectionable
effects of iodin preparations?”
Six of the correspondents have not used it, or are uncertain
whether or not they used the product made by Gardner.
One correspondent remarks: “Never used it. Repelled by
claims of superiority which exaggerate disadvantages of
potassium iodid and overlook the small amount of iodin
used in the preparation advertised.” The five clinicians
who have prescribed the preparation report as follows:
1. Objectionable iodin effects in two cases, both patients
being intolerant of all iodin preparations. 2. Has only prescribed
it once or twice, but thinks he has seen iodism
in one case, some years ago; does not recall clearly. 3. No;
but has used this make very little, and then always in very
small but continued doses. 4. No, always used it in small
doses. 5. Yes, several cases in children; typical coryza,
etc., with doses of three drams three times a day.
Conclusions: It appears that typical iodism occurred
in several cases, after doses corresponding to 10 grains or
less of potassium iodid per day, and this is a rather limited
clinical material. Objectionable iodin effects are, therefore,
not uncommon. Several correspondents remark that
the relative infrequency of iodism is easily explainable
by the fact that syrup is rarely employed in conditions which
demand an active iodin medication and that it is, therefore,
always taken in small doses. In fact, the main if not the
only point of superiority of the syrup appears to be in its
flavor.
These clinical opinions and experiences, therefore, are in
complete agreement with the judgment of the committee,
namely, that the therapeutic claims made by the manufacturers
for this article are exaggerated and misleading.
OTHER MISSTATEMENTS
The above is by no means the only misstatement in the
printed matter issued by this manufacturer. In the publication,
“The Applications of Iodin,” issued in 1907, there
occur the following misleading statements which, since they
refer to plainly chemical facts, did not require submission
to the clinical staff:
That the administration of potassium iodid after meals
greatly impairs its physiologic action “by its chemical union
with the various food products” (page 19). So far as the
committee knows, potassium iodid does not combine with
the food products in the stomach.
“Iodid of potassium, having an alkaline reaction, neutralizes
the hydrochloric acid in the gastric secretions, causing
indigestion, loss of appetite and depression” (page 19).
The United States Pharmacopeia states, under Potassii
Iodidum: “Its aqueous solution is neutral or has a slightly
alkaline reaction on litmus paper.” The slight occasional
alkalinity would be physiologically insignificant, and it is
absurd to claim that this alkalinity causes “indigestion, loss
of appetite and depression.”
“The dose of iodid of iron is so small that the amount of
iodin contained therein is of little advantage” (page 19).
As a matter of fact, the pharmacopeial average dose (1 c.c.)
of the Syrup of Iodid of Iron contains as much iodin (0.85
grains) as a teaspoonful of Gardner’s Syrup of Hydriodic
Acid (0.83 grains).
“In hydriodic Acid the iodin is in combination with hydrogen,
one of the elements of the natural secretions of the
body, and is, therefore, in physiologic harmony” (page 21).
No comment is needed.
It is implied elsewhere (page 29) that potassium iodid
decomposes more readily, with the liberation of iodin, than
does hydrogen iodid. This is contrary to the prevailing
opinion, and would require definite evidence before it could
be accepted. It is also stated the large doses of potassium
iodid in syphilis are necessary, because the gastric decomposition
prevents complete absorption. This is certainly
untrue, for potassium iodid is absorbed almost quantitatively.
These, and numerous other misstatements, constitute violations
of Rule 6; and it is, therefore, recommended that
Gardner’s Syrup of Hydriodic Acid be removed from the
list of remedies approved by the Council; it is further
recommended that this report be published.
The Council postponed final action on the report, pending
its submission to R. W. Gardner. This having been done,
and the reply of Mr. Gardner submitted to the Council, the
above report was adopted and ordered published.
W. A. Puckner, Secretary.
(From The Journal A. M. A., Nov. 14, 1908.)
HYPEROL
Report of the Council on Pharmacy and Chemistry
The Purdue Frederick Company, exploiters of Gray’s
Glycerine Tonic, have recently been advertising to the medical
profession a nostrum called Hyperol. The following report
to the Council, by the referee, was adopted and its publication
authorized.
W. A. Puckner, Secretary.
According to the label, Hyperol is “A Utero-Ovarian Corrective
and Tonic.” The circular accompanying the trade
package states that it is:
“Indicated in all functional diseases of women such as: Amenorrhea,
Dysmenorrhea, Menorrhagia, Metrorrhagia, Subinvolution, and in all
conditions requiring a utero-ovarian corrective and tonic.”
From another circular we learn that:
“Hyperol is a combination of Hydrastine, Aloin, Iron, Apiol and
Ergotin. Its components to a certain extent will indicate its action,
but the therapeutic effects of each ingredient seem to be augmented
to an unusual degree by use in this particular combination. The
proportions of each have been determined by extensive clinical experimentation,
and the formula seems to be exactly balanced to produce
the best therapeutic effects in all derangements of the utero-ovarian
functions.”
This “formula” is not very enlightening and a physician
who wrote for further details was told that Hyperol contained:
Hydrastin | 1⁄40 | gr. |
Aloin | 1⁄12 | gr. |
Iron salts | 3 | gr. |
Apiol (Special) | 3 | ♏ |
Ergotin | 1 | gr. |
| And excipients. |
If this is correct, then, so far as its active ingredients are
concerned, Hyperol is but a mixture of well-known drugs,
having contradictory properties. According to the claims
in the circular quoted above, it is useful both in amenorrhea
and in menorrhagia. The mixture is as unscientific as it is
unnecessary. It cannot be adapted to any individual case;
when ergot is indicated, apiol would naturally be contra-indicated;
if aloes is appropriate, hydrastis may defeat the
object sought. It is unnecessary because no intelligent physician
would prescribe such a combination of drugs in any
given case. The claims are exaggerated, improbable and
foolish. Hyperol conflicts with the following rules of the
Council:
Rule 4, in that statements on the label and in the circular
enclosed with the trade package advertise it to the public
in the treatment of diseases.
Rule 6, in that exaggerated and unwarranted claims are
made for its therapeutic qualities.
Rule 8, in that the name of this pharmaceutical mixture
fails to disclose the potent constituents.
Rule 10, in that it is unscientific.
It is recommended that publication of this report be authorized
to call attention to the unscientific character of such
complex mixtures.
[Editor’s Note: Hyperol is advertised in American Medicine
and the St. Paul Medical Journal.]—(From The Journal
A. M. A., April 18, 1914.)
INGLUVIN
Report of the Council on Pharmacy and Chemistry
A subcommittee of the Council reported that unwarranted
claims and misrepresentation were made for Ingluvin by
its manufacturers, William R. Warner & Co., recommended
that the preparation be refused recognition and that the
report be submitted to Warner & Co. for action.
The report was submitted to the firm, and after waiting
one month and no acknowledgement or reply having been
received, the Council directed its publication. It is as
follows:
REPORT ON INGLUVIN
Ingluvin is manufactured by W. R. Warner & Co.,
chemists, Philadelphia, Pa. The printed matter contains
numerous claims and representations of which the following
are specimens:
“A positive specific for indigestion, dyspepsia and the most effective
remedy in obstinate cases of vomiting of gestation.... A specific
for vomiting in pregnancy in doses of from 10 to 20 grains, and a
potent and reliable remedy for the cure of marasmus, cholera infantum,
indigestion, dyspepsia, and sick stomach caused from debility of that
organ. It is superior to the pepsin preparations since it acts with
more certainty, and effects cures where they fail.... The natural
glycocholic acid in Ingluvin is the active principle and the most
efficient agent in the treatment of all stomachic and enteric disorders.”
Two samples were purchased at different times in the open
market and on examination found to consist essentially of
powdered meat fiber mixed with what appeared to {be} a membranous
tissue resembling the lining of a gizzard. Both
samples on being tested by the method prescribed by the
U. S. Pharmacopeia for estimating the strength of pepsin
were found to possess little, if any, proteolytic activity. In
order to determine whether or not the lining of a fowl’s
gizzard possesses proteolytic action, a fresh gizzard was
secured, the lining washed slightly with water, then removed
and on using one-half of same in place of pepsin as prescribed
by the Pharmacopeial method, it was found to
digest 10 grams of albumin within the time limit. Pepsin,
when properly kept, does not lose its strength to any
material extent.
A careful examination was made for the presence of
glycocholic acid, claimed to be the active principle of
Ingluvin, but its presence could not be established. Furthermore,
the anatomic relations of the fowl are such as to
preclude its presence.
The above shows that Ingluvin does not possess nearly as
much proteolytic activity as ordinary saccharated pepsin
recognized by the 1880 Pharmacopeia, which was prepared
on the basis of digesting 300 times its weight of egg
albumin. Inasmuch as no glycocholic acid is present in
Ingluvin, it would seem that saccharated pepsin would be
far more efficacious in treating the abnormal conditions for
which Ingluvin is recommended in the advertising circulars.
Furthermore, the claims made for the preparation
are grossly extravagant.
A communication from Warner & Co. has been received
since the above report was adopted, in which it is stated:
“The reason that previous letter was not replied to was
because we were desirous of securing all the information
possible on the subject. Since that time we have made considerable
research and also made laboratory investigation,
and are enclosing the accumulated data with diagram of a
part of the alimentary canal showing the esophagus, crop
and gizzard.”
Much of the other matter submitted is immaterial. The
following, so far as it means anything, seems to confirm the
correctness of the report of the Council’s referee that Ingluvin
is practically devoid of proteolytic activity: “... the
therapeutic activity must be due to the bitter property,
rather than any proteolytic activity, and it probably increases,
thereby, the functional activity of the stomach, by which
the normal digestive process is increased. Ingluvin in a
0.4 per cent. hydrochloric acid solution at 37 to 40 C. or
if mixed with an aqueous solution of pepsin under the same
conditions possesses an acrid, bitter taste and increases the
secretion of the saliva and this is practically the same condition
as when in the stomach, it no doubt stimulates the
depressed mucosa peptic glands and increases gastric solution.”
W. A. Puckner, Secretary.
COMMENTS
The fallacies attending the use of digestive ferments in
most stomach diseases have been previously noted in The
Journal.53 In most digestive disorders a deficiency of the
digestive ferment has not been proved. In cases in which
pepsin is lacking, its administration is valueless unless it
is combined with large doses of hydrochloric acid, and it
is doubtful whether this combination is either necessary
or conspicuously useful. There is, however, something so
alluring about medication by digestive ferments which are
assumed to supply a physiologic need, that since their
discovery they have formed a fertile field for the activity
of the manufacturer of proprietaries. As by scientific laboratory
tests, it is possible to determine whether a given
preparation has digestive power, the manufacturers of Ingluvin
avoid this point by claiming that the remedy acts, not
on the food, but on the stomach itself. That remedies may
exist which act as stimulants to the digestive secretions
can not be denied, although at the present time this power
has not been satisfactorily demonstrated. The proprietors
of Ingluvin, finding that proteolytic activity is not to be
attributed to this preparation of chickens’ gizzards, announce
a new therapeutic fact in the claim that “the natural glycocholic
acid in Ingluvin is the active principle and the most
efficient agent in the treatment of all stomachic and enteric
disorders. According to the report made to the Council
there is no glycocholic acid in this preparation, nor is it
possible, from the anatomic arrangements of the fowl’s
digestive apparatus, for it to get there. By all the tests
which can be applied to determine its value this preparation
is of much less value in digestive disorders than saccharated
pepsin, which was discontinued in the Pharmacopeia because
of its inferiority to the other forms of the ferment.
The repudiation, by the manufacturers, of the more
absurd claims made for Ingluvin, shows the need of maintaining
an attitude of healthy skepticism toward the advertised
therapeutic virtues of proprietary preparations. If a
physician is disposed to use digestive ferments, he should
give preference to the official preparations, and ferments
from other sources should be required to stand the exact
tests which demonstrate the worthlessness of so many preparations
on the market.—(From The Journal A. M. A., July 11,
1908.)
INTESTINAL ANTISEPTIC W-A
Report of the Council on Pharmacy and Chemistry
The Council voted that Intestinal Antiseptic W-A be refused
recognition, and that the publication of the following report
be authorized.
W. A. Puckner, Secretary.
The Abbott Alkaloidal Company advertises “Intestinal
Antiseptic W-A” as
“... A scientifically blended and physiologically adjusted mixture,
of the pure sulphocarbolates of calcium, sodium and zinc, grs. 5, with
bismuth subsalicylate, gr. 1-4 and aromatics.”
This formula is in conflict with Rule 1 in that it does not
state in what proportion the sulphocarbolates are present.
The name “Intestinal Antiseptic W-A” is in conflict with
Rules 4 and 8, since it is therapeutically suggestive.
The preparation is in conflict with Rules 6 and 10, in that
exaggerated claims are made for it, no evidence being submitted
to prove the superior value of the mixture.
The most serious of these conflicts consists in the exaggerated
and misleading therapeutic claims. The advertisements
say:
“This combination has no equal as an antiseptic and inhibitive agent
in typhoid fever, diarrhea, dysentery,” etc.
“Numerous cleverly devised and scientifically constructed intestinal
antiseptics have been introduced to the profession, but not one of them
has ever rivaled for one moment these salts in popularity.”
“... we are convinced that no small share of the credit for the
reduction of the death rate in infantile diarrheas is due to the widespread
application of this general method of treatment, associated of
course with the calomel clean-out and the regulation of diet, now
known to be essential.”
“But the use of the sulphocarbolates is not restricted to diseases of
the alimentary canal, although in the summer diarrheas, gastric fermentation,
intestinal indigestion, typhoid fever, dysentery—indeed in all
alimentary disturbances—it is the one essential remedy. It is also indicated
in practically all infectious diseases.”
“Typhoid Fever (in this disease the W-A Intestinal Antiseptic is of
great value; used early, with the proper synergistic cleanout, it will
often cut short the disease).”
These extreme claims exceed the limits of permissible
optimism, unless they are supported by strong dependable
evidence. They contrast sharply with the low esteem in
which the phenolsulphonates (sulphocarbolates) are generally
held. To accept these claims, and to justify encouraging
physicians to rely on them, it would be necessary to establish:
First, that feasible concentrations have a distinct antiseptic
action on cultures of intestinal bacteria. This experiment
could be easily made, but the claims do not seem to be
based on evidence of this kind.
Second, that the preparation actually checks putrefaction
in the intestines. There are several methods by which this
proof may be attempted; but the claims do not appear to be
based on evidence of this kind.
Third, that the preparation actually has a favorable influence
on the progress of diseases. This sort of evidence is
exposed to so many fallacies that it would have to be gathered
very carefully and critically, duly discounting the effect
of other treatment; for instance, by comparison with similar
cases which do not receive this preparation. This is especially
important; and yet we find directions to use this preparation
in conjunction with active cathartic treatment, which
in itself has considerable influence on the conditions for
which this preparation is recommended. No evidence of this
kind is presented.
The testimonials contained in the advertisements cannot be
considered as serious evidence. None present any indication
of accurate record or proper control of conditions, or of the
performance of control observations. They are superficial
impressions, to which little or no weight can be attached.
It is recommended that Intestinal Antiseptic W-A be considered
ineligible for New and Nonofficial Remedies.—(From
The Journal A. M. A., Dec. 19, 1914.)
BANNERMAN’S INTRAVENOUS SOLUTION
Report of the Council on Pharmacy and Chemistry
Bannerman’s Intravenous Solution (Wm. Bannerman and
Co., Chicago) was refused recognition because vague, indefinite
and misleading statements were made regarding its
composition, because it was recommended for anemia, tuberculosis
and syphilis under grossly exaggerated and unwarranted
claims and because the intravenous injection of
complex and indefinite mixtures is unscientific and dangerous.
Notice of the action of the Council having been sent
to the Bannerman Company, the firm submitted a revised
statement of composition and also a revised advertising
circular.
The claim is made that Bannerman’s Intravenous Solution
“is a compound of only the purest and proven efficient
U. S. P. drugs.” According to the latest statement:
| Each 10 c.c. of Bannerman’s Solution contains: |
Hydrargyri Albuminas Mercury Content | 1 | 1-9 | Gr. or | 0.075 Gm. |
Ferri Albuminas Iron Content | 4 | 1-4 | Grs. or | 0.286 Gm. |
Sodii Chloridum | 6 | 1-5 | Grs. or | 0.412 Gm. |
Calcii Salicylicum | 4 | | Grs. or | 0.26 Gm. |
Guaiacol | 4 | | Grs. or | 0.26 Gm. |
Creosote (Beechwood) | 5 | | Grs. or | 0.32 Gm. |
The solvent is said to be distilled water.
The formula is unsatisfactory in several particulars. The
stated amounts of some of the ingredients are in excess of
their solubility in water; the nature and amount of
albumin contained in the “Hydrargyri Albuminas” and “Ferri
Albuminas” are not given; the claim that the solution contains
only U. S. P. drugs is not true. But the main objection
to the preparation is its unscientific character and the unwarranted
therapeutic claims made for it.
Even though a patient had all three diseases, syphilis,
tuberculosis and anemia, it would be most irrational to use
a shotgun prescription, containing, in fixed unvarying proportions,
mercury for the syphilis, iron for the anemia and
germicides for the tuberculosis. In syphilis the mercury-content
of Bannerman’s Solution is inadequate; in anemia
the intravenous administration of iron is unwarranted, and
in tuberculosis there is no evidence that the injection of
bactericides is efficient.
Exception must be taken, moreover, to the statement that
“its use is absolutely safe.” The danger of anaphylaxis
from repeated injections of albuminates cannot be disregarded,
and as J. F. Anderson, director of the Hygienic
Laboratory, has pointed out54 we know little of the secondary
or remote effects of the intravenous injection of toxic substances;
some of them probably do permanent harm.
Such claims as the following require no comment:
“It builds up and increases the hemoglobin in the blood.
“It increases the number of red blood corpuscles.
“It regulates the white cells.
“It stimulates cell growth; therefore, it is reconstructive.
“It is a powerful antiseptic.
“It is useful in any septic condition.”
In view of the facts given, the Council again refused
recognition to Bannerman’s Intravenous Solution.—(From
The Journal A. M. A., Jan. 2, 1915.)
IODALIA[O]
Abstract of Report of the Council on Pharmacy
and Chemistry
Iodalia is sold by Geo. J. Wallau, Inc., with the claim
that because of the peculiar combination in which it contains
iodin it is a valuable and efficient substitute for
iodids. The preparation was examined in the Chemical
Laboratory of the American Medical Association, which
reported to the Council that, contrary to claim, iodin in
the form present in Iodalia would, when administered, act
like an ordinary iodid. Further the proportion of iodin
present was so small that to administer the equivalent of
20 grains of potassium iodid it would be necessary to give
the contents of a one-dollar bottle of Iodalia. In view of
this report it is evident that the claim that Iodalia is
“always well tolerated” and that it cannot produce “symptoms
of iodism” is true only because of the small percentage
of iodin it contains. The claims made in the advertising
matter, that Iodalia is an efficient iodin medication in the
treatment of syphilis, that it is a suitable substitute for
cod-liver oil and that it may be used in anemia, dysmenorrhea,
dyspepsia, malaria and diseases of the heart, are
entirely unwarranted.
Iodalia is exploited in a way to suggest its use to the
public for a host of diseases. Particularly reprehensible
are the recommendations contained in a circular which
accompanies the trade package that Iodalia:
“... offers the same protection against grippe, bronchitis, pneumonia,
tuberculosis, pleurisy and other infectious diseases that vaccine
does against small pox ...”
“It is also the best preventive against the slight infections and ailments
to which debilitated and delicate children are subject.”
The Council voted that Iodalia be refused recognition.—(From
The Journal A. M. A., Dec. 12, 1914.)
IODEX
Report of the Council on Pharmacy and Chemistry
Iodex is manufactured by Menley and James, Ltd., New
York. It is advertised as
“... an embodiment of vaporized iodin in an organic base,
reduced and standardized at 5 per cent. by incorporation with a refined
petroleum product.”
The advertising conveys the impression that the effects of
free iodin are to be obtained from the preparation; it is said
to contain “5 per cent. Therapeutically Free Iodine,” and to do
“... everything the doctor expects of FREE iodin employed by
inunction, without one physical or therapeutic drawback.”
The statements are also made that the preparation “neither
stains, irritates, blisters or cracks the skin,” and that “thirty
minutes after inunction iodin can be found in the urine.”
The following report of an examination made by the Chemical
Laboratory of the American Medical Association has
been submitted to the Council:
“Iodex is dark green, practically black. The green color
is apparent when the ointment is rubbed on the skin, but
disappears on continued rubbing. This nonstaining property
is explained by the results of a test for free iodin, made on
five specimens, four of which yielded only minute traces of
free iodin, while the fifth yielded none. Of course, the statements
that Iodex is an ‘Effective Free Iodin Application
Without Drawbacks’ and also a means of ‘Really Efficient
External Iodine Therapy Without Stain or Irritation’ contradict
each other. Free iodin cannot be present in a sufficient
quantity to be therapeutically efficient in any application
which does not stain or irritate the skin.
“The total iodin content of the five specimens was found to
be 2.63 per cent.—a little over one-half of the content claimed.
“Absorption and excretion experiments were performed to
test the claim that ‘thirty minutes after inunction iodin can
be found in the urine.’ In several subjects, from 1 to 2 gm.
of Iodex was rubbed on the skin of the forearms, and the
urine, for periods varying from seven to seventy-two hours,
was collected and tested for iodin. In all of the tests the
results were negative.”
Iodex is advertised as beneficial in muscular soreness,
sprains, sciatica, neuritis, chronic rheumatism, enlarged
glands, orchitis, epididymitis, gout, burns and dermatomycoses.
It is also said to be “Indicated in Glandular Enlargements,
Inflammatory Conditions, Various Joint Diseases, Rheumatism,
Skin Diseases, Chilblains, etc., etc.”
To sum up:
1. As shown in the foregoing laboratory report, the composition
is incorrectly stated, for the actual iodin content is
only about half of that claimed.
2. It is not true that the action of Iodex is essentially that
of free iodin, which is the impression conveyed by the
advertising.
3. The assertion made in the advertising, that iodin may be
found in the urine shortly after Iodex has been rubbed on the
skin, has been experimentally disproved.
In view of these findings, the Council voted that Iodex be
refused recognition for conflict with Rules 1, 4 and 6.—(From
The Journal A. M. A., June 19, 1915.)
IODIA
Report of the Council on Pharmacy and Chemistry
The following report on Iodia was adopted by the Council
and its publication authorized.
W. A. Puckner, Secretary.
Iodia is put on the market by Battle and Company, under
the claim that it contains potassium iodid in combination
with iron phosphate and vegetable “principles.” It is extravagantly
recommended for use in many and varied conditions.
For instance, it is “an unexcelled altero-reconstructive,”
“almost a specific” in eczema and rheumatism and “a
highly efficient form of iodin,” which will not produce iodism!
The therapeutic effects of iodids result from a chemical
transformation by which molecular iodin is set free in the
tissues, thus producing a mild degree of iodism. It follows,
then, that a preparation which cannot give rise to the symptoms
of iodism cannot be expected to produce the therapeutic
effects of the iodids. The claim that Iodia is therapeutically
efficient without producing iodism therefore justifies suspicion,
to put it mildly.
In view of the exaggerated tone of the advertising, together
with the fact that a report from the Chemical Laboratory
of the American Medical Association showed marked discrepancies
between the formula and the composition of Iodia,
it seemed desirable to investigate this product. The report
of the laboratory, which is given below, shows conflict with
Rule 1 (secrecy of composition) and with Rule 2 (false
claims of standardization). A discussion of the claims made
for Iodia follows the report.
LABORATORY REPORT
The composition of Iodia is given thus:
“Formula.—Iodia is a combination of active principles obtained from
the green roots of Stillingia, Helonias, Saxifraga, Menispermum and
aromatics. Each fluid drachm also contains two and one-half grains
Iod.-Potas. and one and one-half grains Phos.-Iron.”
We are told that:
“Its several ingredients are selected with scrupulous care, and the
most exacting methods are constantly employed to insure absolute uniformity
and maximum therapeutic potency.”
This “formula” is an absurdity: First, the amounts of the
“active principles” of the plants named are not given; second,
these “principles,” with the possible exception of menispermum
alkaloids, have not been isolated; and, third, ferric
phosphate and potassium iodid are incompatible! Incidentally,
there are no methods whereby it is possible to secure “absolute
uniformity” of a mixture such as Iodia is claimed to be.
Qualitative tests demonstrated the absence of iron and the
absence of all but traces of phosphorus compounds (0.015 gm.
phosphorus per 100 c.c.). Minute traces of alkaloids, possibly
from menispermum, were found (that amount being about
0.004 gm. per hundred c.c. of the preparation). Therapeutically
this quantity is entirely negligible. Determinations of
iodid demonstrated the presence of only about 60 per cent.
of the amount of potassium iodid claimed. The “formula”
for Iodia is false and misleading.55
DANGEROUS RECOMMENDATIONS
One of the Iodia labels reads:
“Indications.—Syphilitic, scrofulous and cutaneous diseases, dysmenorrhea,
menorrhagia, leucorrhea, amenorrhea, impaired vitality,
habitual abortion and general uterine debility.”
Such recommendations are likely to lead to self-drugging
in conditions that are not only dangerous to the individual
but also a menace to the community. The preparation thus
conflicts with Rule 4 of the Council. After admitting the need
of efficient iodid medication in certain stages of syphilis and
after exaggerating the frequence and severity of symptoms of
iodism, an advertising circular entitled “Practical Therapeutics”
asserts:
“Iodia then is the preparation of iodid of potassium to be preferred
whenever it requires to be administered in large doses or for prolonged
periods of time ...
“Not only does the association of the iodid of potassium with the
vegetable alteratives offer a measure of protection against iodism but the
latter exert depurative effects on their own account ...”
It is generally accepted that in certain stages of syphilis
the only hope of success lies in efficient iodin medication.
The exploiters of Iodia state that a dose of the nostrum
contains 21⁄2 grains of potassium iodid; actually it contains
only 11⁄2 grains. To urge physicians and the public to depend
on this product for efficient iodid medication constitutes an
unwarranted therapeutic exaggeration (Rule 6) which
approaches criminality. The reason Iodia does not produce
iodism is that, in the doses recommended, the iodin action is
extremely feeble.
Likening the human body to a factory and discussing the
“break downs” which are likely to occur, a circular entitled
“Always Trustworthy” says:
“When administered in proper dosage, Iodia stimulates organic functions,
promotes the elimination of waste products, and re-establishes
metabolic activity. It increases the solvent properties of the blood, and
arrests abnormal tissue metamorphosis. In other words, it lends material
assistance to weakened cells and curbs those unduly active. Iodia,
obviously, has a wide range of indications. It has been most generally
and successfully employed, however, in Syphilitic, Scrofulous and Cutaneous
Diseases, Rheumatic and Gouty Ailments, Dysmenorrhea, Menorrhagia,
Leucorrhea, Amenorrhea, Impaired Vitality, Habitual Abortion
and General Uterine Debility, and wherever a reliable altero-reconstructive
is required.”
These recommendations show that in addition to the
objections already given, this nostrum is an unscientific shot-gun
mixture. This brings it in conflict with Rule 10 (unscientific
articles inimical to the medical profession and the
public).
It is recommended that Iodia be refused recognition.—(From
The Journal A. M. A., Nov. 21, 1914.)
BURNHAM’S SOLUBLE IODINE[P]
Report of the Council on Pharmacy and Chemistry
The Council has authorized publication of the following
report on Burnham’s Soluble Iodine.
W. A. Puckner, Secretary.
Burnham’s Soluble Iodine is offered to the medical profession
by the Burnham Soluble Iodine Company, Auburndale,
Mass., under the claim that by
“... a new process hitherto unknown to chemistry,... Iodine
is converted into a soluble article—soluble in water and soluble in gastric
secretions and in the tissues.”
Beyond this no statement as to the qualitative or quantitative
chemical identity of Burnham’s Soluble Iodine is
furnished; this secrecy, of course, has given the preparation
a certain mysterious prestige among unthinking physicians.
Burnham’s Soluble Iodine was examined in the Chemical
Laboratory of the American Medical Association some six
years ago and was found to be an alcoholic solution of free
iodin (approximately 3 gm. per hundred c.c.) and combined
iodin in the form of iodid (equivalent to about 2 gm. of potassium
iodid per hundred c.c.). Thus the total iodin content was
somewhat less than half of that of the official Tincture of
Iodin (Tr. Iodi), which contains 7 gm. of free iodin and 5 gm.
of potassium iodid to each 100 c.c. The official tincture,
diluted one-half, therefore, would be essentially equivalent to
the Burnham preparation, both being miscible with water.
The Burnham Soluble Iodine Company objected to the conclusions
drawn, from this analysis, but admitted the correctness
of the analysis itself.
Any one who gathered his first knowledge of the subject
from the Burnham advertising might readily infer that no
soluble iodin had been known prior to Burnham’s Soluble
Iodine. This, of course, is not the case; the method of producing
a solution of iodin by the use of an iodid has long
been known.
The following statement is not only obviously untrue but
also nonsensical:
“In all the history of iodin medication, covering a period of laboratory
research of many years duration, every effort to produce a free
iodin, prior to the evolution of Burnham’s Soluble Iodine, was attended
by failure.”
The company lays stress on the assumed superiority over
the iodids of a preparation containing free iodin. This
assumption is based on a fallacy. Those who regard free
iodin as superior to combined iodin forget that free iodin
taken by the mouth is converted in the intestines, by the
action of the alkaline intestinal secretions, into an iodid
with a small amount of iodate, while administered intravenously
(a procedure that, while advocated by the Burnham
concern, is therapeutically indefensible), it enters into combination
with the alkaline salts and proteins of the blood.
The free iodin in Burnham’s Soluble Iodine must act in the
system as an iodid, and the whole iodin content, to furnish
a correct estimate of the value of the preparation, should
be reckoned as an iodid.
Bearing this in mind, then, it is evident that the doses of
Burnham’s Soluble Iodine recommended by the manufacturers
are extremely small. They range from 20 minims
(equivalent to 1 grain of potassium iodid) to 1⁄2 minim (equivalent
to 1⁄40 grain of potassium iodid). From 5 to 20 minims
(equivalent to about 1⁄4 to 1 grain of potassium iodid) is the
dosage recommended for syphilis; from “1 to 3 minims
[equivalent to from 1⁄20 to 3⁄20 grain of potassium iodid] three
to six times daily” for typhoid and other intestinal diseases.
No wonder the exploiters can say that this nostrum does not
irritate the intestines, that it is “non-irritating to the weakest
stomach” and that there is an “entire absence of toxic action
from maximum doses”! Its alleged freedom from the irritating
and untoward effects of ordinary iodids is due, not to
any inherent superiority of the preparation, but to the insignificant
amount of iodid present.
The preparation is advertised for use in an extremely wide
range of diseases, in some of which iodid therapy is recognized
as of value, while in others it is generally regarded
as either worthless or harmful. Given orally or intravenously
(the recklessness of the latter method should again
be emphasized) Burnham’s Soluble Iodine is claimed to
be of:
“... great utility as an internal antiseptic in tubercular affections ...”
Since, as previously explained, free iodin, when introduced
into the body, enters into chemical combination before it has
a chance to permeate the tissues, and since the alkali iodids
possess very slight (in fact, for this purpose, negligible)
antiseptic powers, it is evident that this claim is unfounded.
So, for the same reason, is the claim that “as an intestinal
antiseptic,” Burnham’s Soluble Iodine is:
“... efficient in Typhoid Fever, Enteritis and other intestinal
diseases.”
It is recommended in exophthalmic goiter, notwithstanding
that this condition is generally recognized as contraindicating
the administration of iodids, which excite the action of
the thyroid gland, and which therefore must be used with
great circumspection. An especially indefensible recommendation
is that 1⁄2 minim of Burnham’s Soluble Iodine
(equivalent to 1⁄40 grain of potassium iodid) be administered
every five minutes in “membranous croup”—diphtheria—until
relief from dyspnea is obtained. But, of all the extravagant
claims made for this preparation, perhaps the following
is the most reprehensible:
“In the treatment of Phthisis, in its various forms, clinical evidence
clearly indicates that the use of SOLUBLE IODINE affords the most
potent method of treatment available. Dose—2 minims, increasing to
5 minims in four ounces water before meals.”
Remove the mystery and tell physicians that a dose of
1⁄10 or 1⁄4 of a grain of potassium iodid is “the most potent
method of treatment available” in tuberculosis and the
absurdity becomes self-evident. Nor is this the worst feature
of the advice here offered. Iodin, by combining with the
fatty acids of tuberculous tissues, promotes their autolysis
and consequently their softening and breaking down. The
products of this autolysis are carried by the lymphatics
to healthy tissues and thus may spread the infection. Therefore
the use of iodids in tuberculosis, even in small dosage,
should not be undertaken lightly.
It is recommended that Burnham’s Soluble Iodine, a semi-secret
preparation, exploited by means of extravagant and
dangerous therapeutic claims, be held ineligible for admission
to New and Nonofficial Remedies, and that this report be
published.—(From The Journal A. M. A., May 15, 1915.)
IODOTONE[Q]
Abstract of Report of the Council on Pharmacy
and Chemistry
Eimer and Amend, New York, who market Iodotone,
state that it is a solution of hydrogen iodid (hydriodic
acid) in glycerin, containing 1 grain of iodin to each fluid
dram. The unwarranted assertion is made that Iodotone
“... will produce the constitutional effect of iodine in a shorter
time than other preparations ...”
This cannot be true, for it is certain that, because of the
alkaline reactions of the tissues, iodids, whether administered
as hydrogen iodid or as alkali iodids, must circulate
in precisely the same form and therefore exert the same
therapeutic effects in precisely the same way.
Eimer and Amend further assert that the ordinary iodids
may to advantage be replaced by Iodotone. The absurdity
of this claim is apparent when it is considered that it will
be necessary to administer nearly one fluidounce of glycerin
to obtain an amount of Iodotone equivalent to a 10-grain
dose of potassium iodid. The additional claim that Iodotone
will not disturb the stomach or produce the usual disagreeable
symptoms of iodism is evidently unwarranted, for
it is generally conceded that symptoms of iodism can be
avoided only at the risk of insufficient iodin medication.
Because of these unwarranted and misleading claims and
because the name Iodotone would tend toward the uncritical
use of the preparation as a general tonic, the Council
voted that Iodotone be refused recognition.—(From The
Journal A. M. A., Dec. 12, 1914.)
IOSALINE
Report of the Council on Pharmacy and Chemistry
Iosaline is a rheumatism remedy for external application.
In view of the misleading and unwarranted claims which
are made for it, the Council voted that Iosaline be refused
recognition and recommended publication of the Committee’s
report which appears below.
W. A. Puckner, Secretary.
COMMITTEE’S REPORT
The following sweeping but rather indefinite claims are
made for Iosaline.
“Iosaline is a penetrator and overcomes the objectionable escharotic
properties of Iodine; it is readily absorbed and may be used without
discomfort or discoloration.”
“The strong analgesic properties of Iosaline make it especially useful
in controlling pain in cases of Neuralgia, Rheumatism, Gout, and
Arthritis Deformans.”
As there are few, if any, known iodin compounds which
are “readily absorbed” through the skin and which will not
at the same time produce discoloration or discomfort, it
was thought worth while to take up the examination of
Iosaline. The results of this examination are reported by
the Chemical Laboratory of the Association as follows:
Laboratory Report:—Iosaline is advertised by the Iosaline
Company of New York, as a remedy for the treatment by
external application, of rheumatism, gout, neuralgia, pneumonia
and numerous other diseases. Concerning its composition
the following statements are made:
“A transparent, non-staining gelatinoid of combined iodine with
menthol and methyl salicylate.
“Alcohol 070. per cent.
“Chemical tests demonstrate the preparation to contain 5 per cent. of
iodine.”
The placing of a cipher before the percentage figure for
alcohol, though perhaps accidental and not meant to mislead,
might cause a hasty or careless reader to understand
7 per cent. or .07 per cent., instead of 70 per cent., as the
proportion of alcohol present.
The preparation examined was a very pale yellowish,
translucent solid having a strong odor of methyl salicylate
and a fainter odor of menthol. A package sold for 2 ounces
contained 51.7 gm. Qualitative tests indicated the presence
of alcohol, an iodid, menthol, methyl salicylate, potassium,
sodium, combined fatty acids and a trace of glycerin. Thyroid
extract was not found. Quantitative examination indicated
the following approximate composition for Iosaline:
Alcohol (by weight) | 48.05 | per cent. |
Menthol | 2.07 | per cent. |
Methyl salicylate | 10.25 | per cent. |
Potassium iodid (4.25 per cent. iodin) | 5.55 | per cent. |
Soap | 12.68 | per cent. |
Glycerin | a trace |
| Water and undetermined matter to make 100 per cent. |
Iosaline, therefore, appears to be a solidified, watery-alcoholic
solution of soap containing potassium iodid, menthol
and methyl salicylate. Physiologic tests carried out by
rubbing the preparation on the skin and afterward testing
the saliva and the urine for an iodid indicated that none of
the potassium iodid is absorbed. Since Iosaline is claimed
to contain 70 per cent. of alcohol and 5 per cent. of iodin,
the alcohol content is but 68.7 per cent. and the iodin content
but 85 per cent. of the amounts claimed. The phrase
“combined iodin” is evidently meant to mislead, and adds
the element of mystery on which preparations of this class
rely so largely.—(From The Journal A. M. A., March 15,
1913.)
Abstract of Report of the Council on Pharmacy
and Chemistry
Nourry Wine (E. Fougera and Co., New York) is a
proprietary iodin preparation said to contain 12 per cent. of
alcohol and 11⁄2 grains of iodin in combination with tannin
to the fluidounce. Experiments made in the A. M. A. Chemical
Laboratory demonstrate that the iodin contained in
Nourry Wine is present either in the form of iodid ions or in
a form very readily yielding iodid ions and that therefore its
action will be that of ordinary iodid. Yet a circular asserts:
“The Nourry Wine is the one preparation ... able to introduce
into the organism the active metalloid liberated little by little from the
organic combination....”
While Nourry Wine contains but an insignificant proportion
of iodin, the circular claims that “Nourry Wine presents
a high dose of iodin.” Further, the label on Nourry Wine
and the circular which is wrapped with it suggests its use
in a number of diseases in which iodin medication is considered
of minor importance. These recommendations, bolstered
up by testimonials from twelve to twenty-five years old,
are likely to lead the public if not the medical profession to
use this weak iodid wine where efficient treatment is called
for. The attempt is made to give a further false value to
Nourry Wine in the minds of those who prize everything
that is foreign by the suggestion that it comes from France
when in reality it is made in New York. In conclusion the
Council held that, though the alcohol of the wine is the most
potent constituent, the constant use in the advertising matter
of the term “Nourry Wine,” unqualified by the adjective
“Iodinated,” was mischievous as likely to lead to the thoughtless
use of the preparation in cases unsuitable for iodin
medication. The Council refused recognition to Nourry
Wine.—(From The Journal A. M. A., Dec. 12, 1914.)
LABORDINE
A Report by the Council and Some Pertinent Comments
Added Thereto
The following report was submitted to the Council on
Pharmacy and Chemistry by the subcommittee which
examined Labordine:
To the Council on Pharmacy and Chemistry:—Your subcommittee
presents the following report on Labordine, sold
by the Labordine Pharmacal Co., St. Louis.
Labordine is advertised to physicians as having the following
composition:
Apium Graveolens (true active principle) “Process-Laborde” | 353⁄8 |
Gaultheria Fragrantissima (true active principle) “Process-Laborde” | 251⁄8 |
Acete Amide-Phenyle | 151⁄8 |
Quinina | 11⁄8 |
Benzoyl-Sulphyonic-Imide | 231⁄4 |
It is stated to be a “vegetable antipyretic”; that {“}it
reduces temperature without heart depression,” and physicians
are warned to “avoid acetanilid poisoning and danger
from other coal-tar antipyretics.”
While the “formula” and the statement just quoted are
sufficient evidence of the fraudulent character of the product,
yet an abstract of the reports of the chemists who
analyzed it is given further to demonstrate its character.
Taking the average of the reports of analyses, labordine
contains:
Acetanilid | 37.9 |
Free salicylic acid | 6.9 |
Quinin | present |
Corn starch | present |
Milk sugar | 34.7 |
This report of analysis only makes apparent that Labordine
is not what it is claimed to be. While it is claimed
to contain 231⁄4 per cent. saccharin, this substance was
not present, or mere traces only. While, in a disguised
way, it is stated to contain 151⁄8 per cent. acetanilid, it
contained nearly 40 per cent.
It is recommended that Labordine be not approved and
that this report be published.
The recommendation of the subcommittee was adopted by
the Council, and in accordance therewith the above report is
published.
W. A. Puckner, Secretary.
COMMENTS
A concrete illustration of some general principles previously
laid down is furnished by a nostrum too unimportant
to be of any value, save to “point a moral and adorn a tale.”
About thirteen years ago Labordine was advertised under
the name of Analgine-Labordine, “A purely vegetable
product,” “a combination of the active principles of Camellia
Thea, Apium Graveolens, saccharin and carbohydrates,”
“Superior to Antipyrine, Phenacetine, Antifebrine, Acetanilid”—note
the use of two names for the same thing—“or
any of their imitations,” and “unexcelled by any coal-tar
product or their compounds.” In 1894 the name was changed
to Labordine, in order, as its owner stated, to prevent its
being mistaken for a coal-tar product of similar name.
What its composition was at this time we do not know,
since there is no guarantee of the permanence nor stability
of nostrum formulas except “the honor and reputation of the
manufacturers,” which, as investigation has shown, is not
always unimpeachable. There has been nothing to prevent
alteration of the formula, if the proprietors desired, with
every change in the moon. But the name and the general
tone of the advertising has been the same. The claim of
superiority over coal-tar products has been constantly made.
As to the present conditions, a circular enclosed with a
sample of Labordine, recently sent from the St. Louis office,
contains the formula given above in the report of the Council.
In the same circular are also found these illuminating
statements: “The medical profession has long appreciated
the dangers involved in the administration of various mineral
remedies now so commonly employed, and the value of
a safe, effective and reliable vegetable antipyretic is universally
recognized. Such a remedy is Labordine. It is
purely vegetable in its composition and produces none of
the evil after-effects of the coal-tar derivatives.... Labordine
... is a purely vegetable cardiac stimulant.... There
is nothing mysterious about Labordine or its
constituents.... The ‘Process-Laborde’ gives the true
active principles of the Celery and Indian Wintergreen,
something heretofore difficult to obtain. To this is added
the fact that absolutely chemically pure Acet-Amide-Phenyle
is used. The latter is the most valuable and, in fact, the
only vegetable antipyretic known.”
The above report of the Council shows the following facts:
1. Apium Graveolens (true active principle), “Process-Laborde”
is probably powdered celery seed. One chemist
says: “The powder has the characteristic odor of celery,
while a microscopic examination shows the presence of a
substance having the characteristic structure of seeds in
general.” If celery seed has any “active principle” it has
never been isolated. As to its therapeutic value, nothing
whatever is known. It is, we understand, highly beneficial
in the case of singing canaries, but authorities in scientific
therapeutics have never discovered that it possessed any
remarkable medicinal qualities.
2. Gaultheria Fragrantissima (true active principle), “Process-Laborde,”
is probably ordinary everyday salicylic acid.
One analysis showed salicylic acid to be present to the
amount of about 7 per cent. The question of whether or not
salicylic acid could in any way be considered the “true
active principle” of Gaultheria Fragrantissima, was submitted
to Prof. John Uri Lloyd of Cincinnati, the eminent
authority on the chemistry of the proximate principles of
plants, who replies:
“The advertisement is evidently so worded that, although
the name of the Indian plant Gaultheria Fragrantissima is
employed, its true and active principle being wintergreen
oil, the concoctor can mystify his patrons and at the same
time use the well-known wintergreen oil, made in America,
which in my opinion, so far as any chemical test might be
concerned, could not be distinguished from the methyl
salicylic acid (wintergreen oil) derived from the Indian
plant. Concerning whether salicylic acid is a proximate
constituent of Gaultheria Fragrantissima, in my opinion,
it would be a misnomer to make such an announcement.
Salicylic acid, per se, does not exist, in my opinion, in the
plants mentioned, being made by chemistry.”
3. The third and most important ingredient in this “purely
vegetable antipyretic” is brazenly announced as “Acet-Amide-Phenyle,”
but it is only necessary to say that this
imposing designation is an attempt to “Frenchify” a scientific
name for acetanilid.
Analysis shows that this coal-tar product is present to the
amount of 37.9 per cent., or 1.89 grains in a 5-grain tablet.56
In other words, this imposing Labordine, made by a mysterious
and elsewhere unheard of “Process-Laborde,” is
simply one more of the many acetanilid powders that have
been foisted on our profession and that have filled our journals
for years past. The only thing in it that is of practical
therapeutic value is 2 grains of acetanilid to a 5-grain
tablet. The statement that Labordine is a purely vegetable
preparation is probably intended by the proprietors as a
good joke on the medical profession. Acetanilid is not usually
regarded as a vegetable product, at least it is not
ordinarily found in market gardens. The only vegetable
source from which acetanilid can be obtained is the beautiful
flowering coal-tar bush, from which so many other
nostrum vendors obtain their “perfectly harmless, purely
vegetable antipyretics,” all composed of acetanilid and something
to hide it. If the statements made by one of the company’s
employees and quoted below are true, Labordine is
not “manufactured and made chemically pure in the laboratories
of the Labordine Pharmacal Company,” for this company
has no laboratory, and its product is manufactured
for it.
4. Our readers will be interested to know that the important
ingredient entered under the imposing name of Benzoyl-Sulphyonic-Imide
is simply a highly scientific name
for saccharin. Even on this point, however, the formula is
misleading, since it claims 231⁄4 per cent. of this substance,
whereas the analysis shows that the presence of saccharin
could not be proved. If it is present at all it is in quantities
much less than stated, and so small as to be difficult of
recognition. Instead it appears that the product contains
common starch and about 35 per cent. of milk sugar.
THE COMPANY ITSELF
One of the humiliating phases of the proprietary medicine
business is that, in many instances, these preparations are
foisted on our profession by men who know nothing of medicine,
pharmacy or chemistry, yet who not only presume to
concoct our medicines for us, but also assume to instruct us
how to use them.
Gould’s Commercial Register for 1907 gives the officers of
the Labordine Pharmacal Company as H. M. Coudrey, president;
M. Crawley, vice-president, and D. E. Gamble, Jr.,
secretary and treasurer. The place of business is given as
420 Market street, St. Louis. We are informed that Harry
M. Coudrey is an insurance agent and the present member
of Congress from the Twelfth Missouri District; that Mark
Crawley is a clerk in the insurance office of H. M. Coudrey;
and that Mr. Gamble is cashier in the same office. A recent
visit of a representative of The Journal to 420 Market
street, St. Louis, showed that the office of the Labordine
Pharmacal Company is in Room 12 on the third floor of an
old dilapidated building. There was no sign on the door
of the office, but on the wall next to an old elevator was a
very small sign which read “Labordine Chemical Company,
Room 12.” The office at the time of the visit was apparently
in charge of a young woman about 20 years old.
Careful scrutiny of the furniture and fixtures showed that
the room contained an old oak roll-top desk in one corner
and a kitchen table, on which were piled about half a dozen
packages of Labordine. The floor of the room was bare and
very dirty. In an adjoining room, the door of which was
open, was piled a lot of broken furniture. No laboratories
nor chemical apparatus were visible. The young woman in
charge stated that Labordine was made by the Mallinckrodt
Chemical Works, at No. 3600 North Second Street, St. Louis.
This is a fair sample of nostrums and of the methods of
exploiting them. The bitterly humiliating fact about the
whole business is that a preparation, advertised under such
palpably misleading claims, could actually be advertised in
medical journals, even in journals of a supposedly high
scientific standard, and could be bought and prescribed for
years by supposedly intelligent and conscientious physicians.
It is not supposed that every physician should be enough of
a chemist to detect the ridiculous discrepancies between the
published formula and the therapeutic claims made for such
a mixture. But that members of a supposedly learned profession
should fail to have enough interest in the preparations
they prescribe for their confiding patients to find out
that acetanilid is being masked under an obsolete and little
used name, that under an imposing polysyllabic designation
is hidden saccharin, that the so-called “active principle
Process-Laborde” (whatever that may be), is equivalent
only to one-third grain of salicylic acid in a 5-grain tablet,
and that the advertising matter sent out for years by this
company contained absolute falsehoods regarding the composition
and therapeutic benefits of its preparation, is certainly
just cause for shame and humiliation. If a physician,
knowing the composition of Labordine, wishes to prescribe
it and prescribes it intelligently, he has a perfect right to
do so. If he wishes his patient to have 2 grains of acetanilid,
1⁄20 of a grain of quinin, and 1⁄3 of a grain of salicylic
acid, and considers a mixture of ground celery seed, starch
and milk sugar as a proper vehicle for this medication, no
one will question his right to administer it. No physician,
however, has any right, either moral or professional, to prescribe
a preparation, concerning the ingredients of which
he knows absolutely nothing.
Is it possible that such carelessness may be one of the
causes of waning public confidence in our profession? We
leave it to our readers to determine whether such a moral
can be drawn from this typical nostrum story.—(From The
Journal A. M. A., March 30, 1907.)
LACTOBACILLINE OMITTED FROM N. N. R.
Report of the Council on Pharmacy and Chemistry
The Franco-American Ferment Company has advised the
Council on Pharmacy and Chemistry that, in advertising its
products, it will no longer conform to the rules of the
Council. This is evident. The Franco-American Ferment
Company has distributed circulars in which the public is
informed that auto-intoxication is the cause of innumerable
ills ranging all the way from arteriosclerosis, rheumatism
and gout to chronic headache, odorous perspiration, nervous
disorders and melancholia; that the Bulgarian bacillus “is
a wonderful corrective or remedy” for all these conditions,
and that the Lactobacilline products are the only preparations
of Bulgarian bacillus “to be had in America which
bear his [Professor Metchnikoff’s] personal endorsement”—by
inference, the only reliable products. In view of the
action of the Franco-American Ferment Company, and of
the tendency of their advertising to cause the public to
exaggerate slight ailments into alarming conditions, the
Council has voted that the several Lactobacilline products
of this concern be deleted from New and Nonofficial Remedies.—(From
The Journal A. M. A., April 17, 1915.)
REEXAMINATION OF LACTOPEPTINE[T]
Report to the Council on Pharmacy and Chemistry
In 1907 the Council on Pharmacy and Chemistry published
a report on Lactopeptine. At that time it was shown that
Lactopeptine did not have the composition claimed for it.
The same claims as to composition are still being made for
the product. In view of this fact, a second examination of
Lactopeptine has been made and the result reported to the
committee on chemistry. The report confirms the Council’s
findings of six years ago. After adoption by the committee,
it was adopted by the Council and its publication authorized.
W. A. Puckner, Secretary.
SECOND EXAMINATION OF LACTOPEPTINE
Two specimens of Lactopeptine in original unbroken packages
were recently examined. One of these was an American
preparation said to be produced by the New York Pharmaceutical
Association at Yonkers and the other an English
preparation from John Morgan Richards and Sons, London.
When Lactopeptine was first examined by the Council
about six years ago, it was found to be little more than
weak saccharated pepsin, and did not contain the other ferments
which were claimed by the manufacturers to be present.
A statement concerning this was published in the Council
Reports for 1905–1908, p. 43. Because of claims recently
made by the exploiters that this preparation contains not
only pepsin but also pancreatin, diastase, lactic acid and
hydrochloric acid, and that the failure to recognize these
must be due to the lack of ability of the chemists making
the examination, it seemed worth while to undertake a new
series of tests on samples from two sources mentioned, the
products on the British and American markets. The label
on the British sample gives the following as the composition:
Sugar of Milk | 40 | ounces |
Pepsin | 8 | ounces |
Pancreatine | 6 | ounces |
Ptyalin or Diastase | 4 | drachms |
Lactic Acid | 5 | fl. drachms |
Hydrochloric Acid | 5 | fl. drachms |
The label on the American sample gives no quantities but
states that it “represents a combination of the principal
digestive and enzymogenic agents, Pepsin, Pancreatin, Diastase,
Lactic and Hydrochloric Acids, in the proper proportion
to insure best results.”
We have examined both preparations for starch-digesting
power according to the methods employed in our previous
examinations of such ferments and already reported. Diastase
and the amylopsin of pancreatin seem to be completely
absent, or, if present at all, in such minute traces that
digestion of starch is not shown after one hour when quantities
running from 60 mg. up to 150 mg. were allowed to
act on 500 mg. of starch made up into paste. These tests
were repeated, always with the same results, and were controlled
by digestions of the same starch with other diastase
preparations of known value.
Tryptic activity appears likewise to be absent, as in weak
alkaline solution after fifteen hours’ digestion no effect on
coagulated egg albumin or fibrin was observed when 100 mg.
of each preparation was used with 1 gm. of the protein
material.
As was found in the previous investigation the two
products have some peptic activity, but this activity is comparatively
weak, as about 200 mg. of each preparation are
required to digest 10 gm. of coagulated egg albumin with
0.2 per cent. hydrochloric acid in three hours at 40 C.
(104 F.), and 100 mg. portions were unable to completely
digest 10 gm. portions of egg albumin with acid of the same
strength in four hours at 50 C. (122 F.).
Hydrochloric acid is absent, as might be expected from
the character of the preparation, and the amount of combined
chlorid is small; but qualitative tests were obtained
for organic acid resembling in behavior lactic acid, which
is probably present in combined form.
It must be reaffirmed then that in digestive activity both
the Lactopeptine purchased in the United States and that
bought in England are essentially weak saccharated pepsins.
[Editorial Note.—The report of 1907 demonstrated that
Lactopeptine was at that time a weak saccharated pepsin.
The present report shows that Lactopeptine, as it is sold
both in the United States and Great Britain, is still the same
weak pepsin preparation. By the false statements which
appear on the Lactopeptine labels the exploiters lay themselves
liable to prosecution under the Food and Drugs Act—just
as they have laid themselves liable for the past six
years. The continued exploitation of this preparation warrants
a restatement of facts that have been given many
times before:
1. A preparation having the composition claimed for
Lactopeptine—a powder containing pepsin, pancreatin, diastase,
lactic acid and hydrochloric acid—cannot be produced
commercially.
2. Even if such a combination were available, the acidity
of the mixture itself and of the gastric juice would in all
probability destroy the pancreatin before it could reach the
intestinal tract.
3. Even if every constituent could exert its proper function
at the right time, the administration of such a shotgun
mixture would be unscientific and uncalled for.]—(From The
Journal A. M. A., Aug. 2, 1913.)
MEAT AND BEEF JUICES[U]
Report of the Council on Pharmacy and Chemistry
The following was submitted to the Council by a subcommittee:
To the Council:—While meat extracts contain only traces
of coagulable proteids and have little food value, meat
juices are prepared by a process which ensures the presence
in the finished product of considerable quantities of
coagulable proteids and they therefore have considerable
value as foods. Many preparations which are sold as beef
juices or meat juices have no right to these designations.
Since the public and physicians are likely to be misled by
the names given to these products and by the false claims
which are made for them as foods and depend on them in
the nourishment of the sick, it is important that their composition
and their value as foods should be known.
In the following report is presented the results of an
examination of some of the commercial products found on
the American market. The report shows that Wyeth’s Beef
Juice (John Wyeth & Bro., Philadelphia), Bovinine (The
Bovinine Co., New York), Carnine (Carnine Co., Fougera
& Co., New York), and Valentine’s Meat Juice (M. J. Valentine,
Richmond, Va.) are sold under names which are incorrect,
that their composition is not correctly stated by the
manufacturers and that false and misleading statements are
made in regard to their value as food.
It is recommended that the products named be refused
recognition for conflict with Rules 1, 6 and 8. Since these
preparations are typical of many others on the market, and
as their use is a menace to the public health it is recommended
that the report be published.
This report was adopted by the Council.
W. A. Puckner, Secretary.
Beef or meat juices are clearly to be distinguished from
beef or meat extracts. The word “juice” applies solely to
the fluid portion remaining in fresh meat after proper cooling
and storing and may be obtained by pressure or diffusion
with or without a low degree of heat. Under heavy
pressure freshly chopped meat will yield from 25 per cent.
to 40 per cent. of a thick reddish juice and if the meat is
previously frozen or heated to 60 C., as much as 50 per
cent. may be obtained. This gives some idea as to the
probable cost of preparing beef juice at home. The chief
characteristics of meat juice are the presence of a considerable
proportion of coagulable protein and a low content of
meat bases. That above represents the nature of these commodities
as usually understood by the medical profession,
is clearly shown by this quotation:57
“One or two teaspoonfuls of this (meat juice) are added
to a teacupful of cold or warm water, which, however, must
not be boiling, or otherwise the albumin would be coagulated,
but it may, however, be sufficiently warm to drink
comfortably.”
Beef juice is considered by some physicians of much
dietetic service and believed to represent liquid food in
concentrated form. W. O. Atwater,58 relative to this product
says:
“Beef juice obtained from the best steak which has been
merely warmed through over the coals and then entirely
deprived of soluble substance by a screw press, is undoubtedly
the most concentrated of the liquid foods.”
The latter authority gives a number of analyses of beef
juices prepared under known conditions.
DEFINITION OF MEAT JUICE
Meat juice is defined by the standards committee of the
Association of Official Agricultural Chemists as the fluid
portion of muscle fiber obtained by pressure or otherwise,
and may be concentrated by evaporation at a temperature
below the coagulating point of the soluble protein. The
solids contain not more than 15 per cent. of ash, not more
than 2.5 per cent. of sodium chlorid (calculated from the
total chlorin present), not more than 4 nor less than 2 per
cent. of phosphoric acid (P2O5), and not less than 12 per
cent. of nitrogen. The nitrogenous bodies contain not less
than 35 per cent. of coagulable proteins and not more than
40 per cent. of meat bases.
Meat juices of commerce are supposed to be made by subjecting
properly prepared meat to heavy pressure with subsequent
concentration of the juice in vacuo at a low temperature.
The latter is necessary because if the temperature
is raised to any material extent the valuable coagulable,
soluble proteins referred to above are precipitated and lost.
In order to establish a basis of comparison relative to the
composition of natural raw beef juice a number of samples
were prepared under known conditions and submitted to
analysis. The results contained in the subjoined table clearly
show that meat juices made under known conditions vary
according to the mode of preparation, but it is evident that
practically one-half of the nitrogen is present as coagulable
protein.
FOOD VALUES
In order to arrive at the food value of any commodity it
is necessary to consider its chemical composition, available
potential energy, absorbability, etc. On referring to the
analytical table it will be found that the amount of inorganic
material in meat juices Nos. 7 and 10 is unduly high. It
appears that sodium chlorid, per se, has been added to both
Bovinine and Wyeth’s Beef Juice probably as a preservative
in the latter and for condimental purposes in the former.
The relative and absolute proportions of phosphatic material
in both products is excessive. The other constituents
present in the ash are those usually found in meat products.
The amount of sugar and glycerin in Carnine is interesting.
These agents may be added for preserving purposes,
but the resulting product, on account of its syrupy appearance,
leads to the belief and is so represented, that it is a
concentrated food. Glycerin is also present in Bovinine
and Valentine’s meat juice. Bovinine in addition contains
about 8 per cent. alcohol.
The total nitrogen content of the trade products excepting
Carnine, is greater than the amount of nitrogen present in
meat juices proper, but the relative amount of nitrogen present
as coagulable protein—the valuable part of meat juice—is
much greater in the latter. In fact, the amount of
coagulable protein present in Valentine’s Meat Juice may be
considered nil, which indicates that an unduly high temperature
is used in its preparation. In this connection it
should also be noted that even a moderate elevation of temperature
influences the chemical composition of meat juices.
For example, the coagulable matter present in Nos. 3, 4 and
5, is approximately one-half that present in Nos. 1 and 2,
which appears to indicate that the best product can be made
without the use of any heat whatever. Several of the trade
products, namely Nos. 7, 8 and 9, contain about as much
coagulable material as meat juice made by heating beef to
60 C. According to the formula appearing in a circular of
the Bovinine Company, a part of the coagulable matter is
present in the form of egg albumin, but the company claims
egg albumin is not used at present. In the case of Carnine,
the coagulable matter appears to be introduced by the use
of blood itself. The exact nature of the coagulable protein
matter in Wyeth’s Beef Juice has not been ascertained. It
is well known to manufacturers and physiologic chemists
that it is practically impossible to manufacture a genuine
meat juice possessing a reasonable amount of coagulable
proteins, which is stable without a preservative.
Meat juices, in addition to the coagulable protein material,
contain other protein bodies such as albumoses and peptones.
These bodies are largely formed from the original protein
bodies present in the meat juice during the process of manufacture.
They are highly nutritious and largely and readily
absorbed from the alimentary canal, but the amount of these
bodies present in the trade products is relatively small
excepting in Bovinine, which is not a meat juice, particularly
when the high prices are considered.
A considerable proportion of the nitrogenous matter contained
in Valentine’s and Wyeth’s products is present in the
form of amino bodies frequently included in the general
term, “extractives.” These bodies may be oxidized in the
body and thus supply heat in a manner similar to alcohol,
but it should be remembered that there still appears to be
a wide difference of opinion among various observers on
this point. Some appear to be of the opinion that the amino
bodies are devoid of food value in that these bodies appear
in the urine practically unchanged. It would, therefore,
appear that the value of the amino bodies is largely of a
stimulant character.
The food value of meat juices, therefore, resides largely,
if not solely, in the coagulable and other protein material
present. Comparing the calorific value or potential energy
available in meat juices proper on this basis with that present
in the commercial products, excluding Bovinine, it will
be seen that on the average the genuine meat juices—that is,
those made by pressure, direct from the meat itself as
wanted—are much superior to the commercial products, notwithstanding
the marked concentration in some cases. The
calories given in the accompanying table do not include
sugar, alcohol or any other added material of this character.
WYETH’S BEEF JUICE
“Wyeth’s Beef Juice” is not a true beef juice, but resembles
rather a diluted meat extract. It contains much added
inorganic matter, is low in coagulable proteins, and considering
the degree of concentration, relatively deficient in
nutritive value. Some of the claims contained in the circular
accompanying this preparation, in view of its composition
set forth above, may be of interest:
“Wyeth’s Beef Juice ..., containing two fluid ounces and
representing three pounds of prime lean beef,...”
“... beef extracts made by the Liebig process are utterly
devoid of the valuable and nutritious albuminous constituents of
meat,...”
[Wyeth’s Beef Juice] “should not be compared with ordinary beef
extract,...”
COMPOSITION OF MEAT JUICES
Column Headings:
2 = Per cent. volatile matter 100 C.
3 = Per cent. inorganic matter
4 = Per cent. sodium chlorid
5 = Per cent. phosphoric pentoxid (P2O5)
6 = Per cent. ether extract, glycerol and undetermined matter
7 = Per cent. total nitrogen
8 = Per cent. coagulable proteins (N × 6.25)
9 = Per cent. other proteins (N × 6.25)
10 = Amino bodies (N × 3.12)
11 = Calories per 500 gm. obtained from protein factor 4.8
12 = Calories per 500 gm. obtained from amino bodies factor 0.56
| Name of Preparation | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
| Trade Products: | | | | | | | | | | | | | | | | | | | | | |
| Chuck beef, cold pressed | 86 | .85 | 1 | .86 | | .20 | | .31 | 1 | .32 | 1 | .74 | 6 | .13 | 2 | .94 | | .90 | 217 | .68 | 2 | .52 |
| Round beef, cold pressed | 85 | .76 | 1 | .53 | | .12 | | .37 | | .75 | 2 | .08 | 8 | .56 | 2 | .37 | 1 | .03 | 262 | .32 | 2 | .88 |
| Chuck beef pressed at 60 C. | 91 | .90 | 1 | .29 | | .19 | | .29 | | .81 | 1 | .09 | 2 | .56 | 2 | .50 | | .84 | 121 | .44 | 2 | .35 |
| Chuck beef pressed at 60 C. | 89 | .56 | 1 | .27 | | .16 | | .37 | 2 | .98 | 1 | .09 | 3 | .00 | 2 | .63 | | .56 | 135 | .12 | 1 | .57 |
| Round beef pressed at 60 C. | 90 | .65 | 1 | .36 | | .16 | | .36 | 2 | .09 | 1 | .16 | 4 | .25 | | .31 | 1 | .34 | 109 | .44 | 3 | .75 |
| Chuck beef heated six hours before pressing 60–100 C. | 98 | .11 | | .39 | | .05 | | .12 | | .25 | | .24 | .... | 1 | .00 | | .25 | 24 | .00 | | .70 |
| Made in Laboratory: | | | | | | | | | | | | | | | | | | | | | |
Beef Juice, John Wyeth & Bro.,
Philadelphia, Pa. | 58 | .84 | 16 | .21 | 6 | .71 | 3 | .27 | 12 | .51 | 3 | .15* | 2 | .88 | 3 | .56 | 6 | .00 | 154 | .56 | 16 | .80 |
Bovinine, The Bovinine Co.,
75 W. Houston St., New York City | 80 | .40‡ | 1 | .55 | 1 | .05 | | .09 | 3 | .64‖ | 2 | .36 | 3 | .38 | 10 | .75 | | .28 | 339 | .12 | | .78 |
| Carmine Co., Lefranco, Paris, France; Imported by Fougera & Co., Agents, New York City | 24 | .80§ | | .86 | | .09 | | .33 | 68 | .94¶ | | .96 | 2 | .25 | 2 | .56 | | .59 | 115 | .44 | 1 | .65 |
| Meat Juice, M. J. Valentine, Richmond, Va. | 57 | .64 | 10 | .26 | 1 | .77 | 3 | .41 | 20 | .41# | 3 | .06† | | .19 | 5 | .44 | 6 | .06 | 135 | .12 | 16 | .97 |
*: Including 0.20 per cent. as NH3;
‡: 8.17 per cent. alcohol found;
‖: 3.1 per cent. glycerol found;
§: vacuum 70 C.;
¶: 47.50 per cent. cane sugar—14.2 per cent. glycerol found;
#: 8 per cent. of glycerol found.
†: including 0.22 per cent. NH3;
The several samples of beef juice were prepared from practically fat free, finely comminuted, chuck and round beef, first by pressure at
the ordinary temperature; second, by heating the prepared meat for several hours at 60 C., then submitting to pressure. Sample No. 6 was made
from chuck beef, prepared as above, by heating six hours at from 60 to 100 C., and expressing after cooling. It is not a beef juice proper
but was prepared, analyzed and added to the list for information. Its composition resembles several commercial articles closely. A number
of products represented and sold as meat juice in the United States were analyzed and the results recorded in the accompanying table.
BOVININE
Bovinine, advertised as a “condensed beef juice prepared
by a cold process” is a mixture of alcohol, glycerin, added
sodium chlorid, and apparently some form of defibrinated
blood. According to the manufacturer’s literature egg
albumin was used formerly but this ingredient is said to be
no longer employed. It is not a meat juice in any sense of
the word. Numerous misrepresentations will be found on
the label and in the literature of Bovinine, of which the following
are typical:
“The blood of selected steers prepared by a cold process, furnishing
a perfect food, free from insoluble elements.”
“The rapidity with which Bovinine is absorbed and assimilated in
the stomach ...”
“It supplies complete nutrition to the patient.”
“Bovinine contains all the elements of the animal, vegetable and
mineral kingdoms for the production of new blood with great rapidity.
Its principal constituents have been selected with a view to furnish
the largest amount of nutriment in the most condensed form and all
the resources of modern chemical analysis have been brought to bear
on this important problem.”
A series of experiments carried out with dogs under anesthesia,
by injecting Bovinine into the stomach, the pyloric
end of which was ligated, shows that Bovinine is not readily
absorbed and assimilated by the stomach as claimed. The
amount of protein material found in the stomach at the end
of one-half hour to one hour and a quarter was practically
equal to the amount introduced by the Bovinine.
It is also represented that Bovinine is of great service in
case of an irritable stomach. This is not borne out by
experiment. Bovinine fed to dogs by the mouth, either alone
or mixed with food, induced vomiting, which was less marked
when Bovinine was given with the regular diet. An examination
of the urine of these animals showed a marked
diminution of the amount of indican, while the ethereal
sulphates were enormously increased, both absolutely and
relatively, when Bovinine was given. Experiments on rabbits
have shown that Bovinine injected into the peritoneal
cavity was invariably followed by large quantities of albumin
in the urine, which persisted for from twenty-four to forty-eight
hours. Thirty to 50 c.c. per kilo given by mouth daily
caused emaciation and weakness; in some cases, irritation
of the gastro-intestinal canal, with death of the animal in
from seven to twelve days.
CARNINE
Carnine is a French preparation imported into the United
States by Fougera & Co., of New York City. In physical
appearance it looks like highly concentrated food, but analysis
shows that it consists of a small proportion of defibrinated
blood dissolved in a mixture of syrup and glycerol, the
whole agreeably flavored. It is represented as a “juice of
rare meat, prepared by cold process. Each tablespoonful
represents 100 gm. of raw meat, or 31⁄2 ounces.” It is clear
that Carnine is not a meat juice in any sense of the word.
VALENTINE’S MEAT JUICE
Valentine’s Meat Juice resembles in physical appearance
taste, odor and by chemical analysis a diluted meat extract.
The nutritive value of meat extracts is virtually nil, as is
well known by the medical profession. Notwithstanding the
composition of Valentine’s Meat Juice and the fact that beef
extract represents little nutritive value, the manufacturer
makes the following misleading representations:
“The two-ounce oval bottle, adopted for the Meat Juice contains
the concentrated juice of four pounds of the best beef, exclusive of
fat; or the condensed essence of one and a half pints of pure liquid
juice which is obtained from the flesh of beef.”
“The use of hot water with the Meat Juice changes its character and
impairs its value.” [Italics in original.—Ed.]
The company must certainly be aware of the fact that its
product contains little, if any, coagulable proteids.
CONCLUSIONS
In conclusion: Neither Bovinine nor Carnine is a meat
juice, the former is anything but palatable and the latter
soon cloys. “Valentine’s Meat Juice” and “Wyeth’s Beef
Juice” are virtually diluted meat extracts, which are known
to possess little food value. A physician depending on any
of the foregoing products to supply material nourishment, in
case of serious illness, is deceiving himself, starving his
patients, and may be lessening their chances for recovery.
If a patient recovers while using these commodities, it is
certainly not due to the food value contained in them—(From
The Journal A. M. A., Nov. 20, 1909.)
VALENTINE’S MEAT JUICE[V]
Report of the Council on Pharmacy and Chemistry
Some time ago the Council authorized publication of a
report[V] dealing with the composition and claims made for a
number of the more generally advertised meat and beef
juices. Among these was Valentine’s Meat Juice. This it
was shown was sold under an incorrect name, the claims for
its composition were not truthfully stated and its exploiters
made false and misleading claims in regard to its food value.
As Valentine’s Meat Juice is still widely advertised the
referee in charge of this class of products deemed a reexamination
of the product advisable. This was made and on
it was based the following report which has been submitted
to the Council, adopted, and its publication authorized.
W. A. Puckner, Secretary.
Your referee has had examined recently purchased specimens
of Valentine’s Meat Juice (Valentine’s Meat Juice
Company, Richmond, Va.). The examination shows that
it has virtually the same composition as that given in the
report of the Council “Meat and Beef Juices” published in
The Journal, Nov. 20, 1909. It contains practically no
coagulable protein material, one of the products characteristic
of a meat juice. It is essentially a diluted meat extract.
The following statement found in former circulars now
seems to have been eliminated:
“The two-ounce oval bottle, adopted for the Meat Juice contains the
concentrated juice of four pounds of the best beef, exclusive of fat;
or the condensed essence of one and a half pints of pure liquid juice
which is obtained from the flesh of beef.”
An endeavor is still made, however, to convey the idea
that the product contains coagulable protein, as shown by
the following:
“Boiling water changes the character of the preparation.”
“The use of boiling water with the Meat-Juice changes the character
of the Preparation.”
The proprietors undoubtedly know that the product does
not contain any coagulable material and that the statements
just quoted are plain misrepresentations.
The advertising circular contains a large number of “Testimonials
of the Medical Profession.” As all are undated,
one cannot tell how old these testimonials are. One
physician recommends it highly for hypodermic use; another
says, “I have kept cases on it and it alone for days, without
attempting to give any other food, and the results have been
entirely satisfactory.” According to another, it is “most
invaluable in typhoid fever and also in diphtheria.”
Valentine’s Meat Juice conflicts with the following rules
of the Council:
Rule 1, in that its composition is not correctly given;
Rule 6, in that unwarranted therapeutic claims are made,
the profession being led to believe that the product is highly
nutritious and is valuable in the treatment of pneumonia,
diphtheria and typhoid fever;
Rule 8, in that the name is objectionable, for while sold as
a meat juice, in reality it has the character of a meat extract.
Valentine’s Meat Juice is a fraud on the public, and in
view of its continued exploitation under false claims, the
referee recommends that the Council reiterate its former
condemnation and authorize the publication of this report.
[Editor’s Note.—The difference between meat extracts and
meat juices was fully discussed in the previous report of the
Council, Meat “juices” are made by the cold expression of
meat with subsequent evaporation, in such a way that the
nutritious coagulable proteins remain in solution. In making
meat “extracts,” heat is used which almost completely
removes the coagulable proteins and thus renders it practically
devoid of nutrient qualities.
A list of some of the medical journals that carry advertisements
of Valentine’s Meat Juice, follows:
| Pediatrics | Virginia Medical Semi-Monthly |
Old Dominion Journal of Medicine
& Surgery | Medical Times |
| American Medicine.] |
| Medical World | —(From The Journal A. M. A., May 2, 1914.) |
MEDICINAL FOODS
A report, of which the following is an abstract, was submitted
to the Council on Pharmacy and Chemistry by the
subcommittee which examined the medicinal foods:
In order to determine the food value of any food product
it is necessary to consider the following points: Chemical
composition; available potential energy; absorbability and
cost. No attempt is made in this article to discuss each of
these features separately, but they are utilized as required.
The ingredients on which the food value of any article of
food depends are the proteid substances, carbohydrates, fats,
certain inorganic bodies and—under certain conditions—alcohol.
The amount of each of these present in a preparation
must be established by chemical analysis. From the
results thus obtained it is possible to calculate the potential
energy represented by a given food product. In this
report the potential or food value is expressed in the large
or kilocalorie, that is, the amount of heat required to raise
the temperature of one kilogram of water one degree centigrade.
The factors employed in this report for expressing in
calories the actual amount of energy utilized by the system
are 4.8 for proteid substances, 4.1 for carbohydrates, and
9.2 for fats.
The accompanying table embodies the results obtained by
submitting all the well-known so-called “predigested foods”
to chemical examination. The table as published in The
Journal included columns on: Price of bottle, number of
cubic centimeters in a bottle, cost per 500 cubic centimeters,
reaction, specific gravity, percentage of non-volatile residue,
ash, percentage of nitrogen, calories as proteids in 500
grams, carbohydrates before inversion, alcohol by volume,
average recommended adult dose per diem in cubic centimeters,
cost per diem to supply 1,430 calories. These columns
were eliminated from this abstract, as they were unessential,
so far as the practical value of the article is concerned. In
most cases two samples of the same brand were purchased
at an interval of about six months. All the analyses were
made before Jan. 1, 1907. Some of the preparations contain
much glycerin which does not, so far as known at
present, possess any recognized food value, although there
are a number of experiments on record to indicate that it
influences metabolism.
The percentage of nitrogen accredited to each of these
products represents the total amount of nitrogen, irrespective
of the nature of the nitrogenous substances, although some
of this nitrogen has no nutritive value.
By multiplying the percentage of nitrogen found by the
factor 6.25 we obtain the percentage of nitrogenous matter
(proteids) contained in the various preparations. By multiplying
the number of grams of nitrogenous matter present
in 500 grams of material by the factor 4.8 it is found that
the potential energy available by the nitrogenous matter
varies from 10.3 calories to 153.1 calories. Five hundred
grams of the material is made the basis of calculation,
because it approximates a pint, the amount usually believed
to be present in the various trade packages, and because it
affords a ready basis of calculation.
The carbohydrates are represented by cane sugar, maltose,
dextrin and invert sugar. Lactose is probably also present
in some, but it is impossible to establish this. By multiplying
the number of grams of carbohydrates present in 500
grams of the foods by the factor 4.1 we obtain the potential
energy represented by the carbohydrate, which varies from
11.3 to 319.2 calories. The total calorific value of both proteids
and carbohydrates ranges from 54.7 to 397.5 calories.
The total food value of an equal quantity of milk, including
fat, approximates 360 calories.
The value of alcohol as a food product pure and simple
in disease is, however, an open question. There is no doubt
whatever but that it acts to a certain degree as a food even
here, not as a tissue builder, but as a saver of fat and carbohydrate
material, and in order to give the preparations
in question full value as food products, the calories represented
by the alcohol, are credited to each preparation, as
are the proteids and carbohydrates. The factor usually
recognized for expressing the calorific value of alcohol is 7.
By multiplying the number of grams of alcohol present in
500 grams of material by 7, the number of calories varies
from 420 to 658.
On looking over the literature and printed matter distributed
by some manufacturers, the physician is frequently
left under the impression that these preparations contain
all the essential constituents necessary for maintaining normal
nutrition of the body, as is clearly shown by the following
quotation: “Contains sufficient nutritive material to
maintain normal nutrition of the body, a valuable food in
typhoid fever, pneumonia, tuberculosis,... and all the
conditions of the system associated with enfeebled digestion
and malnutrition.”
In order to show the insidiousness of such representations
it is only necessary to give the actual food value of the
average daily dose (the average amount to be taken for
twenty-four hours) recommended by the various manufacturers
for their products. The average adult daily dose
recommended varies from 50 to 150 c.c. The total available
calories per daily dose based on the proteid and carbohydrate
bodies varies from 9.8 to 110.5. Adding to these figures the
amount of energy represented by the alcohol, in each case,
the total available calories varies from 55.0 to 299.5. The
number of calories per diem in sickness should not fall much
below 1,500 during twenty-four hours.
TABULATED RESULTS OF EXAMINATIONS OF MEDICINAL FOODS
Column Headings:
2 = Glycerin and undetermined matter
3 = Per cent nitrogenous matter (6.26)
4 = Calories as proteids in 500 grams
5 = Carbohydrates after inversion
6 = Calories as carbohydrates in 500 grams
7 = Calories as proteids and carbohydrates in 500 grams
8 = Alcohol, by weight
9 = Calories as alcohol in 500 grams
10 = Calories as proteids and carbohydrates per diem dose
11 = Total calories in per diem dose*
12 = Number c.c. required per diem to supply 1,430 calories
| Name of Preparation and Manufacturer | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
| Carpanutrine—John Wyeth & Brother | 28 | .45 | 4 | .28 | 102 | .7 | 5 | .34 | 109 | .5 | 212 | .2 | 12 | .5 | 437 | .5 | 25 | .5 | 78 | .0 | 1,100 | .7 |
| Carpanutrine—John Wyeth & Brother | 21 | .29 | 6 | .24 | 149 | .8 | 5 | .78 | 118 | .5 | 268 | .3 | 14 | .0 | 490 | .0 | 32 | .2 | 91 | .0 | 942 | .9 |
| Liquid Peptones—Eli Lilly & Company. | 3 | .63 | 4 | .50 | 108 | .0 | 6 | .05 | 124 | .0 | 232 | .0 | 18 | .0 | 630 | .0 | 69 | .6 | 258 | .6 | 829 | .4 |
| Liquid Peptones, with Creosote—Eli Lilly & Company | 4 | .34 | 3 | .84 | 92 | .2 | 13 | .47 | 276 | .1 | 368 | .3 | 18 | .0 | 630 | .0 | 110 | .5 | 299 | .5 | 716 | .2 |
| Nutrient Wine of Beef Peptone—Armour & Company | 14 | .97 | 0 | .64 | 15 | .4 | 15 | .43 | 316 | .3 | 331 | .7 | 17 | .5 | 612 | .5 | 66 | .3 | 188 | .8 | 757 | .4 |
| Nutrient Wine of Beef Peptone—Armour & Company | 13 | .70 | 0 | .43 | 10 | .3 | 15 | .57 | 319 | .2 | 329 | .5 | 17 | .0 | 595 | .0 | 65 | .9 | 184 | .9 | 773 | .3 |
| Nutritive Liquid Peptone—Parke, Davis & Company | 1 | .02 | 1 | .86 | 44 | .6 | 12 | .89 | 264 | .2 | 308 | .8 | 18 | .8 | 658 | .0 | 74 | .2 | 232 | .1 | 739 | .5 |
| Nutritive Liquid Peptone—Parke, Davis & Company | 1 | .95 | 1 | .16 | 27 | .8 | 13 | .19 | 270 | .4 | 298 | .2 | 17 | .7 | 619 | .5 | 71 | .5 | 220 | .2 | 779 | .2 |
| Peptonic Elixir—Wm. Merrell Chemical Company | 3 | .21 | 2 | .54 | 61 | .0 | 11 | .46 | 234 | .9 | 295 | .9 | 16 | .5 | 577 | .5 | 53 | .3 | 157 | .2 | 818 | .6 |
| Tonic Beef S. & D.—Sharp & Dohme | 12 | .91 | 3 | .40 | 81 | .6 | 2 | .36 | 48 | .4 | 130 | .0 | 12 | .0 | 420 | .0 | 13 | .0 | 55 | .0 | 1,300 | .0 |
| Tonic Beef S. & D.—Sharp & Dohme | 12 | .63 | 3 | .28 | 78 | .7 | 2 | .22 | 45 | .5 | 124 | .2 | 13 | .0 | 455 | .0 | 12 | .4 | 57 | .9 | 1,234 | .4 |
| Liquid Peptone—Stevenson & Jester Company | 0 | .44 | 1 | .81 | 43 | .4 | 0 | .55 | 11 | .3 | 54 | .7 | 12 | .0 | 420 | .0 | 9 | .8 | 85 | .4 | 1,506 | .8 |
| Cow’s Milk (3.8 per cent. fat) | .... | 3 | .50 | 84 | .0 | 4 | .80 | 98 | .4 | 182 | .4 | .... | .... | 7 | .3 | 1,429 | .6 | 2,000 | .0 |
*: Total calories per diem dose includes the calories of alcohol in the liquid medicinal foods and the calories of the fat in milk.
In order to get a fair conception of the actual food value
of these various preparations, it is desirable to make some
comparison which can be readily comprehended by every
physician. The amount of good milk necessary each twenty-four
hours to sustain the vitality of a patient during a
serious illness is not less than 64 ounces, or approximately
2,000 c.c. The food value in calories represented by this
amount of good milk may be placed at 1,430. This includes
not only the proteid and carbohydrate matter, but the fat
as well. By comparing this available potential energy with
the total energy available in the predigested foods under
consideration, it can be readily seen that if a physician
depends on the representations made by some of the manufacturers,
and feeds his patient accordingly, he is resorting
to a starvation diet. The largest number of available
calories, including alcohol, present in any of the recommended
daily doses, is less than one-fifth of the number of
calories represented by 2,000 c.c. of milk; and the calories
represented by the daily dose of the preparation poorest in
food products is only one-twenty-fifth of the amount present
in 2,000 c.c. of milk. These figures tell their own story.
Making 2,000 c.c. of milk the basis of calculation, and
estimating the amount of the various preparations required
to yield this number of calories, it is found that the quantity
to be administered daily to supply 1,430 calories, including
alcohol, varies from 716.2 to 1,506.2 c.c. In many cases
the amount of alcohol exhibited by these quantities would
keep the patient in an alcoholic stupor continually. The
cost necessary to supply this energy varies from $1.48
to $3.39. Compare these prices with the cost of two quarts
of milk. Is further comment necessary?
It is urged in justification of the use of preparations of
this class that they contain constituents not found in our
ordinary foods and in a more perfectly assimilable condition.
As pointed out above, these so-called predigested foods
contain no fats; the carbohydrates in them are the ordinary
sugars present in our common foods, while the proteins
belong to the peptone or albumose class. It is for these
latter that the greatest claims are made, but even here no
value can be pointed out not found in whey, peptonized full
milk or peptonized skimmed milk.
There is likewise another point of considerable importance
to consider in this connection. The terms peptone
and albumose include bodies of very uncertain composition,
and their suitableness as food substances depends largely on
how they are prepared. Animal experiments have shown
that nitrogen equilibrium may be maintained, for a time at
least, by use of enzymic hydrolytic products of the proteins,
even where the hydrolysis has been carried far beyond the
so-called peptone stage, but it appears to be likewise true
that the mixtures secured by acid or high temperature steam
hydrolysis have no such value. Some of these, indeed, may
exhibit a toxic behavior. This is true in particular of some
of the commercial varieties of peptone, and until more is
known of the source of the bodies of protein character
employed in the makeup of these “predigested” mixtures it
is unwise to assume anything concerning the food value
of the nitrogen compounds found in them by analysis or
even to dignify them by the name of foods.—(Abstracted
from The Journal A. M. A., May 11, 1907.)
MIGRAININ
Report of the Council on Pharmacy and Chemistry
The Council, having voted to rescind the acceptance of
Migrainin and to omit it from New and Nonofficial Remedies
(Appendix), directed publication of the report given
below.
W. A. Puckner, Secretary.
SUPPLEMENTAL REPORT ON MIGRAININ
To the Council:—Koechl & Co., American agents for
Migrainin (Meister Lucius & Bruning) asserted that this
preparation was a mixture of antipyrin 85 parts, caffein 9
parts and citric acid 6 parts. The experiments of F. Zernik
(Apoth.-Ztg., 1906, p. 686), however, showed that Migrainin
consisted of antipyrin 90.88 parts, caffein 8.4 parts and citric
acid 0.45 parts. When the attention of Koechl & Co. was
called to this they informed the Council, on June 20, 1907,
that the formula they gave was given them direct by the
manufacturers abroad and that they, Koechl & Co., did not
question its accuracy. They, however, offered to “write
abroad and have the manufacturers confirm the formula
as given.” On July 23, 1907, Koechl & Co. wrote the secretary
of the Council that the manufacturers had informed them
that Migrainin contains 90 per cent. antipyrin and 9.1 per
cent. caffein citrate. This being an acknowledgment that
the former statement submitted was incorrect, the Council
voted that the approval of Migrainin should be reconsidered.
Examination of the product, therefore, was taken up in the
Association’s laboratory and an original specimen, purchased
in Chicago, was found to contain moisture 0.7 per cent.,
antipyrin 90.93 per cent., and instead of caffein citrate 9.1
per cent., citric acid 0.51 per cent., caffein 8.53 per cent.
This analysis agreed essentially with the composition of
Migrainin as found by Zernik.
While the discrepancies between the statement of the firm
and the facts are perhaps not great, nevertheless they show
that even the formula last given is incorrect, and that the
statements of Koechl & Co., while no doubt made in good
faith, were in this instance unreliable.
In recent advertising matter issued by Koechl & Co.,
“phenozon-caffein citrate” is given as a synonym for
Migrainin, one circular stating that “Migrainin is phenozon-caffein
citrate,” etc. In the same circular the following also
appears: “In the treatment of migraine with phenacetin or
antipyrin, the attack is delayed, while with Migrainin it is
usually permanently stayed.” This will, no doubt, lead physicians
to infer that Migrainin is not a mixture of antipyrin
and caffein citrate, but that it is some new compound.
While the firm disclaims any intention to mislead, it does
not offer to withdraw or modify this circular. It is recommended,
therefore, that the approval of Migrainin be
rescinded and that it be omitted from New and Nonofficial
Remedies.—(From The Journal A. M. A., June 5, 1909.)
NEURILLA
Report of the Council on Pharmacy and Chemistry
The following report was adopted by the Council. Its publication
was authorized to show how a practically worthless
mixture may be exploited by means of ill-considered testimonials.
W. A. Puckner, Secretary.
Neurilla, which appears to be the sole product of the
Dad Chemical Company, New York, is advertised as
“The Ideal Nerve Calmant.”
“... a nerve tonic ... indicated in cases where the nerve
centers are poorly nourished and over-sensitive ...”
“... a stimulant to the nervous system.”
“A Valuable Aid in the Treatment of Fevers, Colds, La Grippe, etc.”
The following non-quantitative and indefinite formula is
given on the label of a recently purchased bottle of Neurilla:
“Prepared from Scutellaria Lateriflora, Passiflora Incarnata and Aromatics.”
“Proportion of Alcohol 20.3%.
“Made by Dad Chemical Co., New York, U. S. A.
“Dose, One Teaspoonful Four Times a Day.”
According to the formula, then, this mixture contains,
aside from alcohol and aromatics, two vegetable drugs,
scutellaria and passiflora, on which the alleged virtues of
the preparation must be presumed to depend.
Scutellaria lateriflora, or skullcap, is a bitter drug, one of
the many “herbs” to which, on wholly unreliable “clinical
evidence,” therapeutic properties were at one time ascribed.
Most pharmacologists do not mention the drug, and those
who do generally state that it has very feeble therapeutic
properties. It was admitted to the Pharmacopeia, but in
1909 its deletion was recommended by a committee of the
Section on Practice of Medicine of the American Medical
Association (The Journal A. M. A., Sept. 4, 1909, p. 792).
We understand that the next edition of the Pharmacopeia
will omit mention of skullcap.
Passiflora incarnata, or passion-flower, is another “herb,”
which, although known for about seventy years, has never
gained the confidence of the medical profession and has not
even been admitted to the Pharmacopeia. According to a
Council Report:
“None of the evidence is sufficient to show that passiflora
has therapeutic value; hence it is deemed inadvisable to
include the drug in the list of nonofficial remedies” (The
Journal A. M. A., March 19, 1910, p. 983).
On these two obsolescent “herbs,” then, rest the remarkable
claims made for Neurilla. A certain degree of appetizing
effect may be expected from the bitter taste and a very
slight degree of physical stimulus from the alcohol. Except
for these effects—and they are largely delusive and temporary—the
preparation is therapeutically inert and worthless.
The evidence on which the manufacturers of Neurilla base
their therapeutic claims appears to consist of testimonials
from physicians. As a matter of fact, this is true of practically
all of the large group of nostrums of which Neurilla
is typical. An analysis of these Neurilla testimonials brings
out clearly what such “evidence” is worth.
ILL-CONSIDERED TESTIMONIALS
The testimonials for Neurilla have been given with reference
to indefinite conditions of nervousness that border on
the psychic and include hysteria, neurasthenia, neuralgia
and the like. Nervousness and indigestion are two diseases
in which suggestion, especially when aided by bitters and
alcohol, produces temporarily a feeling of improvement. As
an illustration, take the following testimonial:
“But more striking was the following case: One evening between
5 and 6 o’clock I was sent for, family lives near me, and I was
informed that the young lady had promised to be bridesmaid, a function
she had never performed. Her mother said the daughter would certainly
drop in her tracks as she walked up to the altar with the procession,
and they had about concluded to send a note saying to the parents of
the bride that she could not come, although that would be very disagreeable
(and no less offensive, said I). They agreed with me. I
ordered Neurilla for two hours. She went to church, and, I was
informed the next morning, passed through the dreaded ordeal simply
fatigued, and was now fast asleep on account of the nice effect of
Neurilla.”
It might provoke a smile to think that a manufacturer
would publish so silly a testimonial were it not that the very
fact of its publication indicates that there are medical men
thoughtless enough to read and accept such stuff as reliable
evidence as to the value of any product.
TESTIMONIALS GIVEN LONG AGO—THE REMEDY ABANDONED
A number of physicians who had given testimonials were
asked in writing whether the testimonials were genuine and
whether they still entertained the high opinion of Neurilla
expressed at a former date. Several replied that, if they had
ever given such testimonials, they had forgotten the circumstance.
From the replies received we select the following:
The testimonial which bears Dr. A’s name reads:
“I am using Neurilla with most satisfactory results.”
Dr. A now says:
“As to its positive value as a therapeutic agent I have not used it
enough to know.... If the language you quote ... appears
as given in or as a ‘testimonial’ it must in some way be garbled and
appears wholly without my knowledge or consent.”
Dr. B is quoted as having written:
“I do not often lend my influence to furthering the fame of a proprietary
remedy, but I have achieved such excellent results from the use
of Neurilla as a calmative in hysteria and other nervous disorders, that
I feel its manufacturers are entitled to an acknowledgment of gratitude
from me.”
Dr. B writes:
“I have not prescribed a dose of the nostrum in years. The use of
my name in connection with Neurilla is unauthorized.”
Dr. C once wrote:
“I have used Neurilla with good results.”
Dr. C now writes:
“In re ‘Neurilla’ I think I used the preparation once or twice and it
seemed to do good work, but if due to the preparation or other
influences, I am not able to verify. I have not used it since nor will,
as I am opposed to using these preparations except in certain cases
where the ℞ contains remedies whose value I have verified under the
most rigid tests.
“P. S. This testimonial must have been given many years ago.”
Dr. D’s testimonial is admitted to be based on a single
case:
“I am using Neurilla in a bad case of neuralgic tic with very good
results on an aged lady. She has taken several bottles, and is still taking
it with very good results.”
Dr. D sums up his later experience by saying:
“I have long since abandoned the use of Neurilla in practice.”
The following bears Dr. E’s name:
“I endorse Neurilla without hesitation. It meets all indications for
which it is intended.”
This is what Dr. E writes now:
“As to the enclosed testimonial in regard to Neurilla said to be
written by me I have no recollection. I am not prescribing Neurilla.”
Dr. F’s experience is similar. The testimonial credited to
him reads:
“I have prescribed Neurilla in nervous disorders with good results.”
Dr. F now writes:
“I don’t remember of ever having prescribed ‘Neurilla’ or of having
given a testimonial for it or any other patent medicine if I knew it to
be so.”
SUMMARY
In the booklet from which the foregoing are taken, there
are forty testimonials. Those which we quote are merely
samples. To sum up the results of this analysis: Of the
testimonials some are said to be unauthorized; a number
were written with so little thought that the writers had since
forgotten their very existence; the conclusions expressed in
most are not in fact justified by the writers’ mature judgment
and experience. A number of writers admit that their
experience is insufficient to determine whether the supposed
good results were due to the medicine used or to other influences.
Of course such evidence is unworthy of credit and
happily, very little is now being furnished by doctors; even
our courts refuse to admit it.
In short, the published formula shows that Neurilla is
nothing more than a preparation of discredited drugs; it is
exploited largely by means of carelessly formed and thoughtlessly
expressed opinions of physicians. It is recommended
that this report be published as an illustration of such
methods and as a protest against them.
[Editorial Comment.—Neurilla is advertised in the following
publications:
| Archives of Pediatrics, | Medical Sentinel, |
| Atlanta Journal Record of Medicine, | Medical Standard, |
| Charlotte Medical Journal, | Pacific Medical Journal, |
| Indianapolis Medical Journal, | Southern Practitioner, |
| International Journal of Surgery, | Texas Medical Journal, |
| Journal of Nervous and Mental Diseases, | Woman’s Medical Journal, |
| Medical Herald, | Eclectic Medical Journal, |
| New York Medical Record, | Ellingwood’s Therapeutist, |
| Medical Review of Reviews, | Journal of the American Institute of Homeopathy.] |
| —(From The Journal A. M. A., March 27, 1914.) |
NEUROSINE, DIOVIBURNIA, GERMILETUM
AND PALPEBRINE
Report of the Council on Pharmacy and Chemistry
Neurosine, Dioviburnia, Germiletum and Palpebrine are
“shotgun” proprietaries typical of the polypharmacy of the
past three or four decades. They are marketed by the Dios
Chemical Company, St. Louis, Mo. On the recommendation
of the referee, the Council has authorized the publication of
this report.
W. A. Puckner, Secretary.
Neurosine
According to the manufacturers, each fluidounce of Neurosine
represents:
“Bromid of potassium, C. P. | 40 | grains |
“Bromid of sodium, C. P. | 40 | grains |
“Bromid of ammonium, C. P. | 40 | grains |
“Bromid of zinc | 1 | grain |
“Extract Lupulin | 32 | grains |
“Cascara sagrada, fl. ex. | 40 | minims |
“Extract Henbane | .075 | grain |
“Extract Belladonna | .075 | grain |
“Extract Cannabis indica | .60 | grain |
“Oil Bitter Almonds | .060 | grain |
| “Aromatic Elixirs.” |
No physician would think for a moment of prescribing all
of the drugs contained in Neurosine for any one condition.
Yet physicians are urged to use this nostrum in insomnia,
hysteria, neurasthenia, migraine, neuralgia, delirium tremens
and in a host of other conditions.
It is recommended in the treatment of epilepsy on this
ground:
“Neurosine is presented in a very palatable and agreeable form and
can be administered for an indefinite time without untoward by-effects
as so often attends the use of the commercial bromides. In order to
secure lasting benefits the treatment should be extended over a long
period of time.”
The evident implication here is that the recognized drawbacks
of bromid medication are due to impurities present
in the commercial bromids and that the teachings of modern
medicine with regard to caution in the use of bromids do
not apply in the case of Neurosine.
The assurance is offered:
“Neurosine contains no chloral, morphin or other objectionable drug—a
fact of the utmost importance when administering medicines to
neurotic women.”
“As a nerve-calmative and sleep-producer nothing can excel Neurosine ... It
should be borne in mind that this preparation contains no
opium, morphine, chloral or habit-forming drugs ... Neurosine
being a harmless remedy is especially indicated for neurotic individuals.”
Apart from cannabis indica, Neurosine contains no efficient
hypnotic. Cannabis indica is a dangerous drug, whose
administration to “neurotic individuals” is by no means free
from danger—especially when it is given under a proprietary
name that carries no warning of its presence.
Here is another recommendation—this time for chorea:
“All authorities recommend the bromides, hyoscyamus and cannabis
indica in this disease. These remedies are all combined in Neurosine,
the ideal calmative for both children and adults.”
On the contrary, practically “all [medical] authorities” will
admit that it is undesirable to keep a child under the influence
of bromids if this can be avoided. Such treatment is mentioned
only for use as a last resort in extreme cases. Hyoscyamus
and cannabis indica are mentioned in connection
with chorea by few authorities, and then merely as probably
valueless.
Not content with recommending the promiscuous use of
this already too complex mixture, the Dios Chemical Company
advises physicians to combine it with iron, when chalybeate
and tonic effects are to be combined with “a nervine,” with
acetanilid for “irritable cough,” headache and neuralgia,
with antipyrin in asthma and with Dioviburnia—another Dios
nostrum—in all female ailments.
Dioviburnia[W]
Dioviburnia, another Dios nostrum, according to the label,
has the following composition:
“Every fl. oz. contains 3-4 dr. each of the fl. extracts, Viburnum
Prunifolium, Viburnum Opulus, Dioscorea Villosa, Aletris Farinosa,
Helonias Dioica, Mitchellae [sic] Repens, Caulophyllum Thalictroides,
Scutellaria Laterifolia.” [Lateriflora?—Ed.]
Further, according to the label, Dioviburnia contains 18
per cent. alcohol. If this statement is correct then the
“formula” is false, for the fluid extracts named contain from
25 to 73 per cent. of alcohol. As—according to the “formula”—these
fluid extracts constitute three-fourths of
Dioviburnia, the average alcohol-content in the whole mixture
must of necessity be much above 18 per cent.59 According
to the makers, Dioviburnia is “unexcelled” for:
“Amenorrhea, Dysmenorrhea, Leucorrhea, Puerperal Convulsions,
Prolapsus Uteri, Menorrhagia, Threatened Abortion, Parturition, Subinvolution,
Miscarriage, and a general relaxed condition of the uterus
and appendages, together with the various aches and pains peculiar to
women.”
Around the sample bottle of Dioviburnia is wrapped a
booklet entitled “A Treatise on Uterine Diseases and Obstetrical
Hints,” said to be “For the Profession Only.” The
booklet has been prepared, physicians are told, to present
“some very valuable suggestions” regarding the treatment of
female disorders and the attention of the medical profession
is “earnestly” called to the “very remarkable medicine,”
Dioviburnia. Further, it is said that Dioviburnia was devised
by the Dios Chemical Company because there was an “absolute
necessity” for some really efficient internal treatment in
female diseases. The company backs up its statements by
such claims as:
“The most valuable preparation, therapeutically, ever offered.”
“[Contains] every essential for toning up the female organs of generation,
and relieving pain.”
“A general and special tonic, antispasmodic and invigorating cordial.”
While the company directs the attention of physicians to
the “well-known, therapeutical effects of each individual constituent”
of Dioviburnia, there is in reality little positive evidence
regarding the action of any of the drugs contained in
the nostrum. Most writers on materia medica do not even
mention these drugs and the few who do discuss them,
either question or deny their medicinal value. But if every
drug claimed to be present in Dioviburnia had some actual
demonstrated therapeutic properties, it still would be impossible
to predict the action of such a combination in the many
and varied conditions for which it is exploited. Certainly
there is no warrant for such statements as:
“In Painful Dysmenorrhea [sic] Dioviburnia is especially indicated,
and its continued use will invariably give relief.”
“In cases of Leucorrhea of long standing, Dioviburnia, together
with local treatment, invariably gives relief.”
“Dioviburnia is efficient in cases of subinvolution; it cures by its
tonic effects ...”
The effects of the drugs alleged to be present in Dioviburnia
are not such as to justify the hope of either “cure” or
“invariable relief.” In a way the Dios Chemical Company
seems to recognize the inefficiency of Dioviburnia since it
frequently suggests that it be used in combination with drugs
of known value; but it ascribes all favorable results to its
own product:
“In chronic constipation fluid extract of cascara sagrada aromatic
may be combined with Dioviburnia.”
“In cases of habitual abortion, depending on syphilitic taint, a
prescription containing the following should be used during the entire
pregnancy:
℞ “Hydrarg. Chlor. Corros | gr. | 1 |
“Potass. Iodid | dram | 1 |
“Dioviburnia | ounces | 16” |
“An Anemic or Chlorotic patient, suffering with absence of the
menstrual flow, should take DIOVIBURNIA combined with Iron.”
“In leucorrhea depending on endocervicitis, hot astringent douches
once daily should be given. Local applications of iodin are useful in
chronic conditions. Internally, Dioviburnia promotes healing.”
“Rest in bed, hot douches of a one-half per cent. solution of compound
cresol solution and Dioviburnia internally, a teaspoonful
every 3 hours will rarely fail to cure endometritis in a few days.”
“In specific vaginitis, a solution of potassium permanganate (1 to
1000) should be used as a douche twice daily. Internally give the following:
“Sodii benzoate | 1⁄2 | ounce |
“Dioviburnia | 16 | ounces |
| “M. sig., Teaspoonful every three hours.” |
“Prolapsus Uteri is benefited, and often cured, by DIOVIBURNIA
combined with local treatment in the shape of tampons, pessaries, electricity,
etc.”
If Dioviburnia will cure specific vaginitis, anemia, etc., so
will a cobblestone make excellent soup. All that is necessary
in the former case is to add certain potent drugs that might
be indicated in the pathologic conditions mentioned and, in
the latter case, to combine suitable amounts of beef, chicken,
green turtle or vegetables, with herbs and other seasoning.
Germiletum
Germiletum is a member of the large class of alkaline antiseptic
solutions with excessively complex formulas. In this
case not only is the formula complex but also the Dios Chemical
Company finds it impossible even to assign a constant
composition to it—at least the “formulas” which appear on
the different styles of Germiletum labels and advertising circulars
vary greatly.
The company says:
“We appeal only to the Doctor’s judgment of his estimation of the
formula.”
“Doctor you will readily determine from the formula the class of cases
in which you have a right to expect satisfactory results.”
Yet the “formulas” given present so great a variety and
such confusion that it is not clear even to a chemist just
what the Dios Company wants the medical profession to
regard as the composition of Germiletum.
The following statements of “composition” have appeared
at various times:
1. In an advertising circular sent out some time ago:
“[HBF4 + BOH (OC6H4COOH)2
+ BOH (OC6H3COOH)2 + C3H5BO3
+ CH2O + C10H14O
+ C10H20O + C24H28N3Cl.]”
2. In advertising circulars which have been received of late,
being wrapped with a sample package and with the “large
size” trade package:
“[C7H6O2 + H3BO3 + C3H5BO3
+ (CH2O)3 + C10H20O + C10H14O + C2H6O]”
3. In another advertising circular:
“Germiletum is a slightly alkaline chemical solution of Borohydrofluoric
Acid, Borosalylbenzoic Acid, Boroglycerine, Formaldehyde with
Menthol, Thymol and Antiseptic Aromatics.”
4. On the label of a sample package sent through the mails
during 1914, and on the label of a “small size” trade package
purchased in 1914:
“FORMULA.—Borohydrofluoric Acid, Borosalylbenzoic Acid, Boroglycerine,
Formaldehyde with Menthol, Thymol, Amyl Acetate and other
Antiseptic Aromatics.”
5. In the circular which was wrapped around the sample
package referred to above, and around the “large size” trade
package purchased at the same time that the “small size”
package was bought:
“Germiletum is a slightly alkaline chemical solution of Borobenzoic
Acid, Boroglycerine Formaldehyde, with Menthol, Thymol and other
Antiseptec [sic] Aromatics.”
6. On the sample package, on the “small size” trade package
and on the wrappers of the “large size” trade package:
“Alcohol 18 per cent.; Formalin 3⁄4 M. per oz.; Amyl Acetate 1⁄3
M. per oz.” (also written “Acetate Amyl.”)
The label on the large trade package states that Germiletum
contains “Formalin 1⁄2 M. per oz.”
One and all of these various formulas spell mystery. The
existence of some of the constituents is problematic; even if
the theoretical possibility of such combinations be conceded,
some of them could not exist in Germiletum, for they would
be broken up by the alkaline fluid. As illustrating the contradictions
which the formulas present: While the wrapper
of the “large size” trade package claims that Germiletum
contains 3⁄4 minims Formalin (Formalin is a proprietary
name for a 40 per cent. formaldehyd solution) the label on
the bottle claims only 1⁄2 minim. Again, while the composition
expressed in chemical symbols asserts that “H3BO3”
(boric acid) is a constituent of Germiletum, the “formula”
which follows it states that Germiletum has an alkaline reaction;
hence it cannot contain much boric acid. Finally, the
“small size” bottle of Germiletum purchased at the same time
as the “large size” bottle and also the label of a sample
package sent through the mails to a physician in 1914, give as
a constituent “Borohydrofluoric Acid,” which is mentioned
neither on the label of the “large size” trade package nor in
the pamphlet wrapped around it. The only information
which these contradictory “formulas” can convey to a physician
is that Germiletum is an unscientific, varying mixture of
many drugs.
A trade package, having the name “Germiletum” blown in
the glass, bears on the label recommendations for its use in
the treatment of “catarrh,” “Gastritis, Stomatitis, Gastric and
Intestinal Catarrh,” “Leucorrhea and Uterine Diseases,”
“Hemorrhoids,” “Whooping Cough,” “Tonsilitis and other
forms of sore throat” and “Eczema.”
The following statement on the label is designed to induce
physicians to place false confidence in Germiletum to the
danger of their patients:
“The lying-in-room should be thoroughly sprayed with Germiletum.
Can be relied upon to destroy the living particles which so generally
constitute contagion.”
This claim, as well as the assertion which appears on the
label of a sample package and of the “small size” trade
package that it is “PAR-EXCELLENT IN OBSTETRICAL PRACTICE”
is almost criminal, as Fussell60 has said, since to depend on
any preparation of this sort is to court disaster.
The booklet around the trade package makes the claim that
Germiletum “is the best antiseptic”—evidently largely because
it is claimed to be “the blandest of all”—and that it is
“thoroughly germicidal” and even that it is “the best disinfectant
obtainable.” It also contains such unwarranted and
misleading claims and suggestions as:
“... preparatory to all operative work—Germiletum should be
used freely in spraying the atmosphere ...”
“Operative wounds, whether large or small, can be rendered thoroughly
antiseptic by freely spraying them with Germiletum....”
“... it may be given internally in many dyspepsias and in all
zymotic diseases.... In such conditions Germiletum is the ideal
internal antiseptic and disinfectant.”
In the present advertising, no evidence whatever is offered
for the value of Germiletum, the Dios Company contenting
itself with unsupported claims and cant phrases such as
“... the truth is only reached through a final appeal to intelligent
practical experience.”
In the old circulars only crude, uncritical and meaningless
tests to establish the antiseptic value of Germiletum are
reported and none whatever as to its germicidal action. In
the advertising matter sent out some time ago, for instance,
were given “Microscopical, Bacteriological and Chemical
Tests, Comparing Germiletum with Carbolic Acid.” These
tests have no value whatever, unless it be to show the worthlessness
of the preparation. This is particularly true as
regards a series of experiments on “Germiletum as a Preventive
of Lactic Fermentation,” in which one part of Germiletum
in thirty parts of milk did not prevent fermentation. Such
effect as indicated is probably due to the formaldehyd present.
The tests show the absurdity of using the preparation
for internal and external purposes. The referee challenges
the therapeutic claims on the basis that they are extravagant
and unsubstantiated. (The Chemical Laboratory of the
American Medical Association reports that the alkalinity of
Germiletum corresponds approximately to a 1 per cent. borax
solution.)
Palpebrine
According to the Dios Chemical Company, Palpebrine is “A
Reliable External Ocular Antiseptic” having, it is said, the
following composition:
“... each fluid ounce contains 1⁄116 grain Sulphate of Morphia,
1⁄7 grain Sulphate of Zinc, 1⁄11 grain Bi-Chloride of Mercury, 53⁄8
grains Boric Acid, 3⁄4 grain Salicylic Acid.”
The essential virtues ascribed to Palpebrine, according to
its makers, are its harmlessness and its therapeutic efficiency
due, presumably, to its complex composition:
“Attention is called to the constituents of this formula, each one of
which is used by ophthalmologists. Their combination in Palpebrine is
such as to blend their action in a very happy manner. Palpebrine acts
as an antiseptic, an irritant, an astringent, and a nerve tonic to the
mucous membrane of the eye.”
“Palpebrine is superior in its action to the remedies now in use.
It contains all the constituents of Aqua Conradi ... But to these
are added a number of other agents which will prove it to be of much
greater value and give it a broader field for action.”
In all external afflictions of the eye the free use of Palpebrine
is suggested in such statements as:
“They [general practitioners] will therefore gladly receive from our
hands an efficacious preparation which may be used with perfect safety.”
“The name of our preparation—Palpebrine, is derived from the Latin
palpebra, the eyelid, and is well fitted, as it designates at a glance the
sphere of action of Palpebrine.”
“With the assistance of Palpebrine the general practitioner can successfully
treat all cases of external eye disease ordinarily encountered
in his practice.”
One of the members of the Council staff of clinical consultants
calls attention to the fact that much vitally valuable
time might be lost in a case of iritis, for example, which
being unrecognized, should be treated with Palpebrine on the
strength of the Dios Chemical Company’s advertisements.
Even more dangerous is the recommendation of Palpebrine
for the prevention of ophthalmia in the newborn, especially
as this recommendation is coupled with an attempt to discredit
the established treatment with silver nitrate solution:
“The use of severe remedies for this purpose has been discarded by
most physicians....”
While it is doubtless true that ophthalmia neonatorum may
be averted by other drugs than silver salts, it is utterly
unjustifiable to suggest that the established method of treatment
by means of silver salt irrigations has been generally
discarded.
[Editorial Note.—The four nostrums mentioned above
have been grouped together for publication to call attention
to one phase of the proprietary business. A fact not mentioned
in the Council’s report is that these nostrums are
manufactured and promoted by a concern that belongs to a
type we have often designated “pseudo-chemical” companies.
By this is meant companies that are not in the legitimate
business of pharmacy or chemistry, but organized to exploit
one, two or in some instances half a dozen proprietaries.
“Patent medicines” are exploited by this class of “companies.”
The Dios Chemical Company is not a chemical company,
except in name. J. H. Chambers, the founder so far as we
can learn, never claimed any special knowledge of chemistry,
pharmacy or medicine. The officers at the present time are:
J. H. Chambers, president; M. E. Chambers, vice-president;
Leslie T. Chambers, treasurer, and Arthur Chambers, secretary.
M. E. is the wife of J. H., and Leslie T. and Arthur
are sons.
This is simply one illustration of the fact noted above.
Some physicians have been and are prescribing nostrums
originated, manufactured and advertised by laymen who are
not in the legitimate pharmacy business. In addition, such
physicians have been accepting the statements of laymen, not
only as to the composition of the nostrums, but as to their
use. In every state the practice of pharmacy is regulated by
law: before assuming the responsibility of compounding
medicines a druggist must have studied and passed an examination
in pharmacy. Public safety demands and the law
requires it. There are some doctors, however, who will
allow laymen who are not chemists, pharmacists or physicians
to formulate and compound a prescription and tell
them what it is good for and how to use it.
The Dios Chemical Company is not an isolated instance:
we have already referred to some; we shall take occasion to
refer to others in the future. That such concerns flourish is
a reflection not so much on the shrewd laymen who exploit
the medical profession—and through it the public—as it is
on the physicians who cast their scientific training to the
winds and permit themselves to be thus exploited.]—(From
The Journal A. M. A., Jan. 9, 1915.)
OXYCHLORINE
Report of the Council on Pharmacy and Chemistry
The following report on Oxychlorine has been submitted
to the Council by the subcommittee to which it was assigned:
To the Council on Pharmacy and Chemistry:—Your subcommittee
submits the following report: The Oxychlorine
Chemical Company, 1326 Wabash Avenue, Chicago, states in
its advertising literature that:
“Chemically, Oxychlorine is the tetraborate of sodium and potassium
combined with oxychlorid of boron, thus: 6 (NaKB4O7) BOCl3.”
Analysis of Oxychlorine showed:
Potassium | 12.26 | per cent. |
Sodium | 8.20 | per cent. |
Chloric acid—CLO3 | 25.32 | per cent. |
Nitric acid—NO3 | 21.70 | per cent. |
Boric acid anhydrid—B2O3 | 18.63 | per cent. |
Water, calculated | 13.29 | per cent. |
Thus, Oxychlorine is not a definite chemical substance of
the composition claimed, but instead is a mixture of alkali
chlorate and nitrate with boric acid. Assuming that the
chlorate is present as potassium chlorate and the nitrate as
sodium nitrate, the analysis above quoted corresponds to a
mixture approximately as follows:
Potassium chlorat | 37.19 |
Sodium nitrate | 29.76 |
Sodium and potassium tetraborate | 2.18 |
Boric acid | 30.52 |
Undetermined | 0.35 |
| ———— |
| 100.00 |
Your committee recommends that Oxychlorine be not
approved and that this report be published.
The report of the subcommittee was adopted by the Council,
and in accordance with the recommendation is published
herewith.
W. A. Puckner, Secretary.
In commenting on the above report it is hardly necessary
to call attention to the palpable untruthfulness of the furnished
formula or its lack of correspondence to the real
composition of the preparation, to the imposing claims made
by its pseudo-scientific exploiters or the absurdities, from a
chemical standpoint, of the statements made in their literature.
These features are more or less common to all nostrums.
The physician who prescribes or uses Oxychlorine
under the impression that he is getting a definite and unique
chemical compound described as tetraborate of sodium and
potassium combined with oxychlorid of boron is, according
to our chemists, getting simply a mixture of potassium
chlorate, sodium nitrate (or, perhaps, sodium chlorate and
potassium nitrate), and boric acid in about equal amounts.
More than one-third of this mixture is potassium (or sodium)
chlorate, drugs by no means harmless.
In order that there may be no suspicion of unfairness to
the promoters of the preparation, we quote from one of the
advertising circulars sent out by the Oxychlorine Company:
“Oxychlorine owes its recognition as a therapeutic agent
to its six principal qualities:
“1. It will oxygenate the blood at the seat of application,
maintain nutrition and heal an uninfected solution of
continuity of first intention without scar formation.
“2. It will disorganize all pus and ferment-producing micro-organisms,
their toxins, ferments and ptomains.
“3. It will restore an inflamed mucous membrane to its
normal condition, except where the membrane is sclerosed or
atrophied.
“4. It will destroy pathogenic micro-organisms and their
toxins in the blood current.
“5. It will stimulate the blood to absorb more oxygen in
the lungs than it at the time carries. [We do not know what
this means; perhaps the Oxychlorine Company does.]
“6. It is absolutely harmless to the tissues and will not
destroy a living cell.”
Surely these people must have access to physiologic and
chemical authorities not found in modern medical libraries,
or else their esoteric researches into the mysteries of life
must have carried them far beyond the ken of our most
advanced workers along these lines. The scientific world
would receive with great interest information as to how a
mixture of potassium chlorate, sodium nitrate and boric acid
oxygenates blood, maintains nutrition and causes healing
without scar formation. A mixture which will destroy
micro-organisms and yet will not destroy a living cell certainly
shows a fine sense of selection and discrimination
not heretofore expected of a combination of chemicals or of
a chemical compound. How like the wonderful elixir of
medieval times, which was to the Christian a tonic and to
the heathen a poison!
Here is another claim made for this nostrum:
“Two or three rectal injections of a one or two per cent.
solution of Oxychlorine and ten grain doses given six to eight
times per day is the best and most reliable treatment for
typhoid fever.”
If 80 grains of Oxychlorine contain 30 grains of potassium
chlorate, three rectal injections each consisting of 1
pint of 2 per cent. solution, would contain approximately
160 grains of potassium chlorate. Such an injection might
prove decidedly dangerous, especially when used by one
ignorant of its true composition. However, the physician,
not the promoters, bears the responsibility.
Oxychlorine sells at $3.50 a pound; the ingredients can be
obtained for about 44 cents a pound. Perhaps the margin of
profit is intended as a reward due the promoters for the
profound physiologic discoveries announced in their reading
matter.—(From The Journal A. M. A., July 6, 1909.)
PAM-ALA, ANOTHER WORTHLESS QUININ
SUBSTITUTE
Report of the Council on Pharmacy and Chemistry
The following report of a referee on Pam-ala, an asserted
malaria specific, was adopted by the Council and its publication
authorized.
W. A. Puckner, Secretary.
Soon after publication of the Council’s report on Sinkina,
an alleged malaria specific proved worthless, the referee’s
attention was called to Pam-ala, which is sold under very
similar claims.
According to the advertisements which have been appearing
in Southern medical journals, Pam-ala is “A new and effective
remedy for MALARIA.”
The label describes Pam-ala as “An Effective Vegetable
Remedy For MALARIA. Guaranteed free of any Quinine, or
other harmful [sic] drugs.” It is said to be indicated in
“Malarial Intermittent and Remittent Fevers, especially curative
in Chronic Malaria and Malarial Cachexia and all conditions
even where Quinine fails.” One tablespoonful three
times a day is said to be the “Curative Dose,” while one
tablespoonful three times a week is stated to be a “Prophylactic
Dose.” The label further claims that Pam-ala
“Surpasses Quinine in its action and has none of its Disadvantages.”
Assertions that Pam-ala is superior to quinin are
followed by the usual “guarantee” claim: “Guaranteed by
the Pam-Ala Co. under the Drugs Act, June, 1906, Ser. No.
2909 A.” Finally, the label says that it is “Endorsed by
Medical Authorities Throughout the world.”
As regards the composition, a circular says that “PAM-ALA
is a purely vegetable remedy for the cure, without Quinine,
of all forms of Malaria.” “‘Pam-ala’ is derived from a
plant of the genus Umbelliferae, a native of the mountainous
regions of Mexico and northern parts of South America. Its
medicinal properties have not been known to anyone but the
native Indians, who for years past have used it as a specific
in all forms of fever and malarial diseases so prevalent in
tropical countries. The seeds are more active as a therapeutic
agent than the dried-up plant; hence their collection
for medicinal purposes requires special skill in the selection
of the same so as to be able to extract all the possible
medicinal properties from them, viz., its active principle. An
oil may be abstracted from the seeds which is of a yellow
color with an intense characteristic odor.”
At the close of the circular the following unenlightening
formula appears:
| Each fluid ounce contains: |
Ext. Fid. Pam-ala | 10 per cent. |
Alcohol | 15 per cent. |
Ol. Aurant Syr. Sacchari aqua ad. q. s. | 100 per cent. |
In addition to being a cure for malaria, Pam-ala is claimed
to have a “favorable influence upon the broncho-pneumonia
of measles ...,” “will avert an attack of acute catarrh,”
and “abort acute tonsilitis.”
The testimonials are of the usual character. Most of them
seem to have been given some four years ago by physicians
in Italy, Cuba, Porto Rico, Guatemala, etc., and therefore
cannot readily be looked into. Two are of more recent
date and come from physicians in this country. They furnish
good illustrations of the manner in which proprietary concerns
make use of opinions hastily formed and thoughtlessly
put in writing. One testimonial was given in July, 1912:
“I take pleasure in testifying to the seemingly marvelous and gratifying
effect of Pam-ala in 2 cases of malaria....”
On Jan. 2, 1914, its writer, in reply to an inquiry whether
in the light of continued experience, his first estimate of
Pam-ala had been confirmed, wrote:
“... Since then I tried Pam-ala on a number of cases
without any results whatever; in fact my patients seemed to
get worse until I resorted to the usual treatment of malaria,
mercurial laxative followed with quinin. I was too hasty
in stating that Pam-ala cured malaria. I now know and have
known since August, 1912, that Pam-ala will not cure
malaria....”
The writer of the second testimonial is reported to have
written that he tried Pam-ala “on a most pronounced case of
malarial spleen with the most excellent results” and that he
“also tried Pam-ala on a case of Malarial Cystitis and Hematuria,
with entire satisfaction.” In reply to inquiry this physician
admits that he was “very favorably impressed with the
preparation at the time.” He states that at that time he
was also trying out Sinkina and that after six months he
“discontinued the use of both as the results did not warrant
further investigation.” He concludes:
“With due allowance for the fact that certain cases will
for a time improve on any kind of treatment, new or old,
I see no reason for supplanting or even augmenting, the
recognized treatment for malarial conditions, with either
Pam-ala or Sinkina.”
Incidentally it should be mentioned that this physician also
noted the general similarity of Sinkina and Pam-ala. He
observes:
“The physical appearance and properties of the two preparations
seem to be identical, the advertising matter and literature
are surprisingly alike and the only marked difference
seems to be that one remedy is purported to be prepared
from a ‘new’ South American plant and the other from an
equally fresh discovered addition to Asiatic flora.”
WHAT IS PAM-ALA?
From a comparison of the statements regarding the composition
which are made for Sinkina and for Pam-ala, as well
as from the physical characteristics of the preparation, particularly
the odor and taste, it seems evident that the
essential constituent is oil of cumin. Although definite proof
that oil of cumin forms the essential constituent of Pam-ala
would have shown the worthlessness of this nostrum for the
reason that the clinical investigation of Sinkina proved the
worthlessness of oil of cumin, it did not seem worth while
to the referee that this be demonstrated by chemical analysis.
It seemed to him that in such cases as these, the secrecy
with which the identity of the preparation is surrounded, as
well as the extravagant and highly improbable claims, should
be sufficient to condemn it.—(From The Journal A. M. A.,
Feb. 28, 1914.)
PAPAYANS BELL[X]
Report of the Council on Pharmacy and Chemistry
The following report of a subcommittee was submitted to,
and adopted by, the Council and its publication directed.
W. A. Puckner, Secretary.
Papayans (Bell) made by Bell & Co., Orangeburg, N. Y.,
is said to consist of the “digestive principle obtained by our
own exclusive process from the fruit of Carica papaya, combined
with willow charcoal, chemically pure sodium bicarbonate
and aromatics.” The following statement appears on
the package: “For the treatment of dyspepsia, flatulence,
nausea, vertigo, hyperacidity, palpitation and other symptoms
of indigestion and the vomiting of pregnancy. Peritonitis,
cholera morbus, alcoholism and seasickness.” “Digests
every variety of food, removes every symptom of indigestion,
restores the entire digestive tract to a normal condition.”
The dosage is recommended as follows: “From one to three
tablets before meals, or two hours after eating. In severe
cases, three tablets dissolved in hot water and repeated as
necessary.”
A circular which accompanies the package details the
therapeutic virtues of the preparation and contains what
purports to be extracts from medical journals, in which
Papayans is recommended.
Examination of specimens purchased in the open market
showed them to contain the following ingredients: Charcoal,
sodium bicarbonate, ginger, saccharin and oil of gaultheria.
As the product is said to contain papain, the presence of
enzymes was tested for, with the result that it was found to
possess neither proteolytic nor amylolytic properties. The
results of our examination are in accord with the results
obtained by a member of the Council, who examined the
product independently, and who writes:
“We have made some extended tests with Papayans Bell,
and find that the tablets consist essentially of sodium bicarbonate
and charcoal, with a little flavoring matter. We find
no digesting power for starch or egg albumin. At any rate,
no appreciable change follows in the albumin in three hours,
and no conversion to sugar in the same time, or change of
starch to a point where the iodin reaction is weakened. The
product seems to be practically inert.”
It is recommended that Papayans Bell be refused recognition,
and that publication of this report be authorized.
Comment: It will be remembered that two other products
of Messrs. Bell & Company have been discussed in this
department: Salacetin (Bell)61 and Sal-Codeia (Bell).62
Salacetin was examined with several “synthetics” which all
turned out to be mere acetanilid mixtures. Salacetin, advertised
as “a combination, with heat, of Salicylic and Glacial
Acetic Acids and Phenylamine” when examined “was found
to be a mixture and to contain the following ingredients
approximately in the proportion given: Acetanilid 43;
sodium bicarbonate, 21; and ammonium carbonate, 20.” Sal-Codeia
(Salacetin-Codein) therefore, would be the same
with codein added.
Papayans (Bell) seems to be consistently fulfilling the
life-history of the average nostrum. Made of well-known
drugs and invested by its manufacturers—or exploiters—with
virtues absurdly disproportionate to the known properties
of the alleged constituents of the nostrum, the preparation
was introduced to the world via the medical profession.
With the help of thoughtless physicians, aided by a skilful
and aggressive advertising campaign and augmented by the
“free sample” device, the business grew and prospered. The
bottles with the name and address of the company blown in
the glass and with the varied therapeutic indications for the
nostrum printed both on the label and on the circular in
which the bottle is wrapped, have carried the manufacturer’s
message to the drug-taking public.
Apropos of this point, the recent “literature” contains what
purports to be endorsements of the nostrum by medical journals.
Thus there is quoted from the New York Medical Journal,
Jan. 2, 1909, in part, the following recommendation:
“... we venture to suggest to our readers who have not
tried this remedy that they prescribe one original sealed package
of Papayans (Bell) and that they carefully note the
results from its use.” [Italics ours.—Ed.] Having seen an
“original sealed package” we believe that we can predict
the “results from its use.” On any patient not mentally
unbalanced, the result would be that the next dose of
Papayans (Bell) he thought that he needed would be purchased
from the druggist direct.
That such results are not hypothetical is evidenced by the
statements of the exploiters of Papayans (Bell) that “the
annual sale now exceeds four hundred million tablets.”
Assuming that statement to be true, it would be necessary
for every physician in the United States to prescribe over
three thousand of these tablets every year—if they reached
patients only through the physician! The company’s own
figures indicate that the time is about ripe to take care of
this vast army of self-drugging laymen and recent circular
letters seem to recognize it. The physician is notified that
druggists are now furnished with Papayans (Bell) “in sealed
packages of thirty and one hundred tablets.” The medical
man is told that the firm has “not forgotten the days when
physicians’ orders made our success possible” and it says
it is “sincerely grateful to the doctors who gave us orders
in the days when we were struggling for recognition.” This
tacit admission of the value of the physician as an unpaid
agent for nostrum houses should be given thought by those
physicians who prescribe such preparations.
While, so far as we know, Bell & Co. have not yet advertised
in the daily press, they are not averse to furnishing the
laity with samples when requested. An Ohio physician sent
us the following letter received by a young woman who had
written asking for samples:
Miss X—— Y——,
Z——.
Dear Madam: As requested, we are mailing you sample of our
Papayans (Bell) for Indigestion.
If a sufferer from Indigestion, we want you to give it a
thorough trial as directed and note remarkable results that
we believe you will get from its use.
Kindly write us if you are unable to obtain it from your
local druggist, as it is stocked by nearly every good
drugstore in the United States.
Yours truly,
Bell & Co.
Evidently Bell & Co., while admitting that their financial
success is largely due to the kindly, though misguided,
efforts of physicians, are not going to let a little thing like
loyalty to the medical profession interfere with a possible
sale of their tablets.
THE L. D. JOHNS COMPANY
A discussion of the methods of Bell & Company would not
be complete without reference to a concern which seems to
be closely connected with it: the L. D. Johns Company,
whose “only product” is a sugar-coated laxative tablet.
Regarding the “sugar coated” tablet, a visitor at the place
of business of Bell & Company and the L. D. Johns Company
wrote: “These companies apparently are not in possession
of any tablet coating machines and in questioning on
this point stated that some of their tablets were sent out
to be coated.” There is a sameness regarding the claims
for the laxative tablets of the two companies that might lead
one to suspect that the same individual prepared both circulars.
For instance:
| Cascarans (Bell) | | Dr. Johns’ Tablets |
“Taken as directed, it permanently
removes the great majority
of cases of habitual constipation.”
“... a harmless vegetable
preparation.”
“... for the removal of
pimples, yellowness and greasiness
of the skin ...”
“... one tablet at night,
one night and morning, or, in
severe cases, one three time a day,
gradually decreasing the frequency
of the dose as improvement permits.” |
|
“Taken as directed ... permanently
remove the great majority
of cases of habitual constipation,
torpid liver and sick headache.”
“A harmless vegetable remedy.”
“... removes pimples,
blotches, sallowness and greasiness
of the skin ...”
“One at night, one night and
morning, or, in severe cases, one
three times a day. Gradually decrease
the frequency of the dose
as improvement permits.” |
According to a leaflet sent out with samples by the L. D.
Johns Company, the company is capitalized for $500,000,
divided into 50,000 shares at $10 each; these shares are sold
to those physicians who will agree “to prescribe the tablets
at every suitable opportunity, to introduce them to other
physicians” and “to promote their sale in every ethical way!”
If the list of physicians’ names and addresses which the
company sends out as comprising the eastern stockholders
is to be relied on, it would seem that many medical men are
promoting their sale. In prescribing it is, of course, “necessary
to specify ‘Dr. Johns’ Tablets No XXX (Original
bottle).’” As the name is on the bottle, it is not unbelievable
that, as the company says in its prospectus, because
of “our method of advertising, a large and very profitable
business is being created.” That the L. D. Johns Company
expects to profit by the self-drugging which this method
of prescribing fosters is evident:
“Physicians not stockholders in this company suffer from the continual
refilling of their prescriptions and from the recommendation of the
preparation prescribed by patients to others. [Italics ours.—Ed.] Our
stockholders benefit by the refilling of their prescriptions and by these
recommendations.”
Put baldly the case amounts to this: Physicians who prescribe
“Dr. Johns’ Tablets” not only are likely to foster self-drugging,
but they will reap dividends therefrom. Truly a
nice business to be in!
While Bell & Company and the L. D. Johns Company are
said to be entirely distinct, they are to be found at the same
address at Orangeburg, New York, and as will be seen, the
officers of the two companies are more or less related.
| BELL & CO. | | L. D. JOHNS CO. |
| President | John L. Dodge | President |
| Secretary | Geo. C. Tennant | Vice-President |
| Vice-President | Chas. B. Smith | Secretary and Treasurer |
EXPLOITING THE PROFESSION
Nostrum promoters have two simple ways of “working”
the medical profession. The first—and the more profitable—is,
by lavish distribution of free samples, to get physicians
to prescribe the blown-in-the-glass “original package” with
the inevitable result of large sales direct to the laity. By
the second method, which is merely a modification of the
first, the physician furnishes the capital for floating the
nostrum and then takes his share of the resulting profits.
There may not be quite as much money in the second method
for the promoter, but then the risks are correspondingly less.
If the firm fails, the stockholders are the losers; the promoter
is not necessarily “out” anything. From a commercial
standpoint, a combination of the two methods is, of course,
ideal—(From The Journal A. M. A., Aug. 14, 1909.)
PASSIFLORA AND DANIEL’S CONCENTRATED
TINCTURE OF PASSIFLORA[Y]
Report of the Council on Pharmacy and Chemistry
The Council has voted that the drug passiflora (passion
flower) be not accepted for New and Nonofficial Remedies,
and has recommended that the following article be published
in The Journal. It is considered important to call attention,
not only to the lack of reliable evidence of the therapeutic
value of passiflora, but also to the absurdity of the
claims which are made for Daniel’s Concentrated Tincture
of Passiflora, a preparation which has been already refused
recognition.
W. A. Puckner, Secretary.
Passiflora
Although passiflora was introduced into medicine nearly
seventy years ago, the literature concerning it is not very
extensive; it is not mentioned in the standard works on
pharmacology and its chemistry seems never to have been
worked out. There appears, also, to be no record of experimental
investigations of the drug with reference to its pharmacologic
action, except an article by I. Ott,63 who used
“Daniel’s Concentrated Tincture.” Ott claimed that it lessened
the reflex irritability of the cord and paralyzed motion
by acting on the motor centers in the cord, and that it
increased the rate of respiration. He also stated that because
of its action on the vasomotor centers it reduced the frequency
of the heart-beat and lowered arterial tension, but
that these effects were only temporary.
On the clinical side the reports are not numerous and such
as have been made do not appear to be based on very extensive
trials nor on conditions of observation that would
entitle them to more than slight consideration. S. D.
Bullington64 reports good results, but no cure, in one case
of epilepsy, and improvement in a case of insomnia. W. J.
Stapleton65 recommends it in the form of a concentrated tincture
(not the one advertised so extensively), and states that
he has used it with great success in insomnia, hysteria,
neurasthenia, neuralgia, nervous and physical prostration,
and in alcoholism. In his opinion its action is most apparent
in cases of nervousness due to causes other than pain.
S. Harnsberger66 reports two cases in which partial blindness
followed the taking of potassium bromid and passion flower.
Extravagant and inconsistent claims are made for Daniel’s
concentrated tincture of passiflora in the advertising literature,
where it is recommended for such a wide range of diseases
as asthma, typhoid fever, convulsions and paralysis.
None of the evidence is sufficient to show that passiflora
has therapeutic value; hence it is deemed inadvisable to
include this drug in the list of nonofficial remedies.—(From
The Journal A. M. A., March 19, 1910.)
LIQUID COMBINATIONS CONTAINING PEPSIN
AND PANCREATIN [Z]
Report of the Council on Pharmacy and Chemistry of the
American Medical Association
The following report was submitted to the Council by a
subcommittee:
To the Council on Pharmacy and Chemistry:—The U. S.
Pharmacopeia, 8th revision, pages 334–5, states: “Pepsin
and pancreatin in solution are incompatible with one
another. If the solution be neutral or alkaline the pancreatin
gradually destroys the pepsin, and if acid the pepsin destroys
the pancreatin.” The correctness of this statement has been
amply demonstrated by the reports which have been submitted
to the Council from time to time on liquid preparations
claimed to contain these two ferments.
Thus an elixir was investigated which was by the manufacturers
claimed to contain “the five active agents of digestion,
pepsin, veg. ptyalin, pancreatin, lactic and hydrochloric
acids,” and to be “superior to all other remedies in dyspepsia
and diseases arising from imperfect digestion,” and the
committee which investigated the article in question reported
that “it was impossible to establish the presence of either
the proteolytic or the amylolytic ferment.”
Similarly, on another liquid preparation, which was said
to contain “pancreatin, pepsin, lactic and muriatic acids,
etc.” ... “the combined principles of digestion to aid
in digesting animal and vegetable cooked food, fatty and
amylaceous substances,” the committee reported “this product
possessed only very slight proteolytic action and failed
to digest 2 per cent. of its own weight of starch.”
Again, the report on still another preparation stated:
“But while it was said to contain pancreatin, the U. S. P.
test for the valuation of pancreatin failed to indicate this
ferment.”
The report on yet another elixir, claimed to be “the only
true digestant, because it contains the enzymes of all the
glands which are necessary for digestion,” showed that this
article did not contain “any appreciable enzyme activity,
either amylolytic or proteolytic.”
The correctness of these findings of the committee of the
Council was generally acknowledged by the manufacturers
when their attention was called to the matter. Thus, one
manufacturer of digestive ferments writes: “We will ask
you to hold this matter up until you hear from us further
on the subject. The reason for this request is that we have
been going over our liquid preparations very carefully in
order to be sure that after aging they would contain the
ferments that we put into them. The pancreatic ferments
in alcoholic liquids seem to lose their strength.”
The chemist for a large manufacturing house writes:
“There are now on the market a number of preparations in
which pepsin and pancreatin are combined in liquid form,
and the result is that we have had numberless requisitions
from our representatives that we also market such a preparation.
As the result of this we have carried out a series
of experiments no less than four or five times in order to
determine whether pepsin, diastase, and pancreatin would
retain their activity in the form of a syrup, wine or elixir.
We have proved incontrovertibly that this cannot be done.
While any two of these substances, or even all three of
them, can be dispensed in the form of a liquid by the retail
druggist and will retain their normal activity for as long a
period as three to six weeks, yet if allowed to stand sufficiently
long, they mutually destroy each other; so that in
a combination of pancreatin and pepsin the pancreatic
enzyme is lost and the pepsin greatly injured, and where
diastase is present, both diastase and pepsin (or diastase
and pancreatin) mutually destroy each other.”
Since it has been demonstrated that pepsin and pancreatin
cannot exist in one and the same solution for any reasonable
length of time, it becomes apparent that liquid preparations
said to contain these two ferments are sold under
impossible claims. It is therefore recommended:
1. That the Council on Pharmacy and Chemistry
refuse to approve liquid preparations that are claimed
to contain both pepsin and pancreatin.
2. That the medical profession through the Journal
of the American Medical Association be advised of the
fallacy of employing such combinations.
3. That the attention of manufacturers be called to
the worthlessness of such incompatible liquid preparations
of pepsin and pancreatin, and that they be urged
to cease offering such products to the profession.
4. That, since the National Formulary has recognized
a preparation of this kind under the title “Elixir
Digestivum Compositum,” the American Pharmaceutical
Association be requested to instruct its committee on
the National Formulary to omit this preparation from
the next edition.
The recommendations of the subcommittee were adopted
by the Council and publication of the report directed.—(From
The Journal A. M. A., Feb. 2, 1907.)
W. A. Puckner, Secretary.
PEPTO-MANGAN (GUDE)
Report of the Council on Pharmacy and Chemistry
The following report was adopted by the Council and its
publication authorized.
W. A. Puckner, Secretary.
About ten years ago the M. J. Breitenbach Company circulated
what pretended to be an abstract of the report
of a government commission for the investigation of the
anemia then prevalent in Porto Rico. The company asserted
that “this report alone would suffice to establish Pepto-Mangan
at once as the foremost hematinic known.” Examination
of the official report of the commission67 revealed
the fact that the administration of iron in hookworm anemia
was considered of secondary importance, and that of the
various preparations of iron, Blaud’s pill was found to be
more efficient than Pepto-Mangan (Gude). A protest68 was
made at this time by the commission against the unwarranted
use of its report by the Breitenbach Company.
Later the Breitenbach Company sent out a report pretending
to prove that at the Infants Hospital, Randall’s Island,
New York City, Pepto-Mangan (Gude) had been found a
most superior preparation in the treatment of infantile
anemia. Inspection of the hospital records and daily charts
of the cases disclosed69 a remarkable disparity between the
claims of the Pepto-Mangan pamphlet and the real results
of treatment. And so here, also, as well as in the Porto Rico
commission’s report the trials, selected by the Breitenbach
Company prove the limitations and non-superiority of Pepto-Mangan.
The preceding false reports, though no longer circulated,
have never been definitely withdrawn and while it is now
generally conceded that the good results in anemia are
obtained by the administration of the various simple inorganic
iron preparations the Breitenbach Company still attempts to
convey the impression that Pepto-Mangan (Gude) is of most
superior efficacy. Thus the present Pepto-Mangan circular
attempts to discredit by obsolete and absurd or untrue statements
the various preparations of iron which are in general
use and to carry the impression that only iron and manganese,
in the particular form and proportion in which they
are contained in Pepto-Mangan—namely, 3 parts Fe to 1 part
Mn—are useful for the treatment of anemia, chlorosis, etc.
Thus contrary to general conceptions, the impression is given
that the now generally accepted course of chlorosis is due to
the three varieties of insufficiency of certain blood elements:
(1) insufficiency of manganese, (2) insufficiency of iron, (3)
insufficiency of iron and manganese; and that the administration
of iron often fails because manganese is not supplied
to the system at the same time and in sufficient amounts.
The following statement is made:
“Doctor:
“If you have a case of ANAEMIA, CHLOROSIS, or AMENORRHOEA,
that shows no visible sign of improvement, and you have
exhausted the entire list of Nauseating Iron preparations with little or
no effect, it is because the blood is deficient in that essential oxidizing
constituent, MANGANESE, in a soluble, readily assimilable form, the
best being in combination with iron.”
Another extravagant claim:
“Usually after taking it for a week its restorative influence on the
functions of the stomach is felt; appetite reappears, and the general
health is improved by the increase in bodily warmth, an effect directly
due to manganese.”
The following statement implies that Pepto-Mangan is
absorbed unchanged, for which there is no justification:
“As the ferruginous and manganic ingredients of Pepto-Mangan
(Gude) exist in the form of organic peptonates, they have already
undergone the changes necessary to insure prompt absorption and appropriation
by the circulating fluid.”
The following declaration implies that it repairs the individual
defective blood-cells which is, of course, also
ridiculous:
“That Pepto-Mangan (Gude) quickly and efficiently builds up defective
red blood cells, and generates, or at least potently encourages the formation
of new ones, and materially increases their richness in hemoglobin,
has been abundantly demonstrated....”
The M. J. Breitenbach Company is still trying to mislead
physicians; it also aims to make use of them in its direct
appeal to the physicians’ patients. For instance, the
name “Pepto-Mangan (Gude)” blown in the bottle, the
advertising circular suggesting Pepto-Mangan as the treatment
for anemia, etc., and the recommendation to physicians
that it be prescribed in “original bottles” all tend to encourage
the use of Pepto-Mangan by the public with the likelihood
that it will be depended on where good food and fresh
air are of prime importance. The attempt to exploit it
directly to the public is further attested by the advertisements
of department-store drug departments.
It is evident from the foregoing that Pepto-Mangan (Gude)
is in conflict with Rules 4 and 6 and therefore not eligible for
admission to New and Nonofficial Remedies.—(From Reports
Council Pharm. and Chem., 1914, p. 121.)
LIQUID PETROLATUM OR “RUSSIAN
MINERAL OIL” [AA]
Report of the Council on Pharmacy and Chemistry
The following report was submitted to the Council by a
referee and its publication authorized by the Council.
W. A. Puckner, Secretary.
Petroleum has been in use as a medicine from time
immemorial. It was known to Herodotus 400 years before
Christ and is mentioned by Plutarch, Dioscorides, Pliny and
other early writers. It was extensively used by the Arabians
and evidently played an important part in the practice of
medicine in India, being known to the Bengalese as Muthe
Katel. The raw product was the substance used in earlier
times and differed much in character and composition, as
obtained from different sources.
As an internal remedy it was early employed in chronic
pulmonary affections, in obstinate skin diseases, in rheumatism,
and for the expelling of tapeworms. It was extensively
used for these several purposes in France under the name
of “Oleum Gabianum” and in North America as “Seneka
oil.”
The internal use of the refined product may be traced to a
patent granted to Robert A. Chesebrough of New York, in
June, 1872, for the manufacture of a “new and useful product
from petroleum, named vaseline.” This name was originally
applied only to a semisolid preparation, but later a liquid
product known as liquid vaseline was marketed and for a
time exploited as a cure for coughs, colds, consumption and
a number of other diseases and conditions.
The liquid petrolatum has since become known under a
variety of names, proprietary and otherwise, in addition to
being used as a substitute or an adulterant for other, more
costly, fats and oils. Some of the names applied to the
product are:
| Adepsine oil | Neutralol |
| Amilee | Olo |
| Atoleine | Paraffin Oil |
| Atolin | Paroline |
| Blandine | Petralol |
| Crysmalin | Petro |
| Deeline | Petrolax |
| Glyco | Petrolia |
| Glycoline | Petronol |
| Glymol | Petrosio |
| Heavy petroleum oil | Rock Oil |
| Liquid Albolene | Russian liquid petrolatum |
| Liquid Cosmoline | Russian mineral oil |
| Liquid Fossiline | Russian paraffin oil |
| Liquid Geoline | Russol |
| Liquid Paraffin | Saxol |
| Liquid Petrolatum | Terraline |
| Liquid Saxoline | Terralbolia |
| Liquid Vaseline | Usoline |
| Mineral Glycerin | Water-white mineral oil |
| Mineral Oil | White paraffin oil. |
A preparation similar to that official in the Pharmacopeia
of the United States as liquid petrolatum has been included
in many, if not all, of the foreign pharmacopeias, the official
titles under which this preparation is recognized being as
follows:
Petrolatum Liquidum, U. S. Pharmacopeia; Paraffinum
Liquidum, pharmacopeias of Great Britain, Germany, the
Netherlands, Japan, Belgium, Austria, Denmark, Switzerland,
Sweden, Servia, Italy, Hungary and Russia; Oleum Paraffinae,
Spanish Pharmacopeia; Vaselinum Liquidum, French
Pharmacopeia, and Oleum Vaselini (as a synonym) pharmacopeias
of Denmark and Russia.
The requirements of the several pharmacopeias differ
somewhat and the specific gravity as given is as follows:
U. S. P. VIII, 1905 | 0. | 870 | to 0.940 | at 25° |
Ph. Brit. IV, 1895 | 0. | 885 | to 0.890 | at 15.5° |
B. P. C. II, 1911, usually | 0. | 875 | or lower | at 15° |
Ph. Germ. V, 1910, at least | 0. | 885 | | at 15° |
Ph. Ross. VI, 1910 | 0. | 880 | to 0.885 | at 15° |
Ph. Hung. III, 1909 | 0. | 88 | to 0.89 | at 15° |
Ph. Ital. III, 1909 | 0. | 875 | to 0.890 | at 15° |
Ph. Fr. V, 1908, about | 0. | 875 | | at 15° |
Ph. Serb. II, 1908, about | 0. | 880 | | at 15° |
Ph. Svec. IX, 1908 | 0. | 88 | to 0.90 | at 15° |
Ph. Helv. IV, 1907 | 0. | 880 | to 0.885 | at 15° |
Ph. Dan. VII, 1907, at least | 0. | 880 | | at 15° |
Ph. Austr. VIII, 1906, at least | 0. | 880 | | at 15° |
Ph. Belg. III, 1906, not below | 0. | 880 | | at 15° |
Ph. Japon. III, 1906 | 0. | 875 | to 0.945 | at 15° |
Ph. Ndl. IV, 1905, not below | 0. | 860 | | at 15° |
Ph. Hisp. VII, 1905 | 0. | 840 | | at 15° |
For pharmaceutical purposes, liquid petrolatum may be
divided into two grades, the lighter or more limpid oil,
used extensively as a vehicle for oil sprays, and the heavier,
more viscid oil generally recognized in European pharmacopeias
and used as an ingredient of ointments and more
recently as a remedy in the treatment of intestinal stasis.
Under petrolatum liquidum the U. S. P. recognizes a
mixture of hydrocarbons, chiefly of the methane series, which
occurs as a colorless or very slightly yellowish, oily, transparent
liquid without odor or taste and having a specific
gravity of about 0.870 to 0.940 at 25 C. For the U. S. P.
IX, it is proposed to change this requirement somewhat so
as to have it apply to a transparent liquid free from fluorescence,
without odor or taste and having a specific gravity
of from 0.845 to 0.940 at 25 C.
Such a requirement would include all of the available
paraffin oils irrespective of origin. The now commonly available
commercial liquid petrolatum, used for pharmaceutical
purposes, is practically colorless and all of the better grades
are free from odor or taste. The specific gravity varies
from 0.855 to 0.895. The lighter oils, having a specific
gravity of from 0.860 to 0.870, are usually preferred in the
making of oil sprays or solutions of substances to be used
as local applications. The product having a specific gravity
above 0.875 evidently contains a considerable amount of
dissolved solid paraffin which separates out at temperatures
at or below 0 C., but readily dissolves again at temperatures
above 10 C.
There is considerable difference in the chemical composition
of the paraffin oils obtained from various sources. The
American oil consists largely of hydrocarbons of the
methane series, while the Russian oil contains naphthenes
or hydrocarbons of the benzene series, having the empirical
composition of ethylene, (CnH2n) which may be considered
as hydrogenated aromatic hydrocarbons, though they behave
with reagents very much in the same way as do the hydrocarbons
of the methane series.
Mineral oils with a naphthene base are best suited for
making white petrolatum, and at the present time the production
of the colorless water-white liquid petrolatum
appears to be confined largely or almost exclusively to the
crude product of the Baku district of Russia, though it is
asserted that it is now also made from the Hanover (Germany)
crude oil and that some is being produced by “cracking”
the white solid paraffin.
It is also said that the American oil can be made water
white but that it is not being so produced at present for
economic reasons; the yellowish oil, free from fluorescence,
having a very wide sale, both as a lubricant and as a substitute
for lard oil and other of the more costly lubricating
oils.
From a pharmaceutical point of view it would appear
important to note the physical characteristics of the oil and
to insist on absence of color, absence of odor and taste,
absence of acid and of alkali and a specific gravity in harmony
with the purposes for which the oil is to be used.
During the past year or two liquid petrolatum has
attracted considerable attention as a remedy in the treatment
of intestinal stasis or chronic constipation, the practice
of using it having been developed largely through its
recommendation by Sir W. Arbuthnot Lane and his associates.
This use of liquid petrolatum and of petrolatum products
generally is by no means novel. N. A. Randolph70 of Philadelphia
was among the first to suggest its use for this purpose
in an article published in 1885. Randolph also appears
to have been the first to experiment with petrolatum and to
determine its non-absorbability from the intestinal tract.
In an article71 in 1884 he concludes that “pure petrolatum
while entirely unirritating to the digestive tract is valueless
as a foodstuff.”
The experiments recorded by Randolph were evidently
prompted by the fact that vaseline and a number of imitation
products then on the market were being sold as substitutes
for lard and butter, and opinions regarding the food
value of petroleum products appear to have differed very
materially. Following the experiments of Randolph, Robert
Hutchison in 1899 made a series of experiments to demonstrate
that petroleum, petrolatum, paraffin and related products
were absolutely unassailable by any of the digestive
fluids, despite the “large vogue that had of late years been
given to various petroleum emulsions, chiefly by ingenious
and unterrified advertising.” He came to practically the
same conclusions arrived at by Randolph fifteen years earlier
and pointed out that “liquid paraffin in one sense may be
regarded as an artificial intestinal mucus and might in that
way have some value on certain forms of constipation.”
William Duffield Robinson72 reports on the use of a perfectly
refined colorless and odorless petrolatum, supposedly
of American origin. He was able to show that all of the
product passed unchanged through the intestinal tract and
could be regained from the feces. In his conclusions he
expressed the belief that the effect of the administration of
these petroleum products is far more than as a simple intestinal
lubricant. In over fifty selected cases in which nutrition,
digestion and body-weight were impaired, and the
purest oil administered in 1- or 2-dram doses each day for
a period of from four to six months, there was in every
instance an improvement of weight, health and feeling of
well-being. The administration of refined paraffin oil gave
no discomfort in any instance, even in cases in which nearly
a pint was given in a few hours.
William Ewart73 suggests liquid paraffin as a safe agent
for the local treatment of the lesions in typhoid fever. He
says in part: “Mineral oil, such as petrolatum or paraffin,
is neither absorbed nor dissolved; therefore, after all absorbable
ingestions are taken up by the lacteals, it will still
remain in the bowel. In this way pure liquid paraffin is
valuable, precisely because it is inert; moreover, it might
some day, perhaps, be made the vehicle for effective topical
remedies.”
A. D. Schmidt74 quotes Stubenrath as having given liquid
paraffin in the treatment of chronic constipation, and he himself
gave as much as 20 gm. of liquid paraffin to adults without
observing any injurious effect whatever. He says, “As a
result of the administration of liquid paraffin, the feces are
softened considerably and are found under the microscope
to contain numerous minute globules of paraffin.” He was,
however, unable to recover from the feces the entire quantity
of paraffin administered and believes that a certain portion
of it, probably the fractions with a low boiling-point, are
absorbed or possibly oxidized in the organism.
Maurice Vejux Tyrode75 also refers to the use of liquid
petroleum in the treatment of constipation.
Sir W. Arbuthnot Lane in his recommendations of liquid
petrolatum calls it an ideal remedy for stasis, but cautions
against the use of the lighter oil as extensively prescribed
in this country as a vehicle for sprays in nose and throat
work.
Paraffin oil is not absorbed from the alimentary tract and
so far as known exerts no deleterious influence. It is usually
given in quantities of from 10 to 20 c.c. half an hour
or an hour before meals or in larger doses, from 30 to 50 c.c.,
at one time on retiring. From available evidence it appears
that comparatively huge doses may be administered without
the production of any untoward results. According to many
observers, liquid paraffin should not be given with or after
meals because of the inhibiting influence that it may have on
the digestion of food. It is not soluble in water or the ordinary
solvents and therefore cannot be diluted. The denser
oils are preferably slightly warmed or drunk with warm
water so as to obviate the disagreeable slimy sensation that
persists when taken cold.
Volatile oils may be used in moderate amounts to give
a distinctive taste to the otherwise rather insipidly tasteless
paraffin oil. Among the more desirable oils to be used
for this purpose would be oil of peppermint, oil of cinnamon,
oil of betula or methyl salicylate and oil of cloves. From
2 to 10 drops of any of these oils can be added to a pint
of the oil. When larger doses of the oil are to be given at
one time, it would, of course, be advisable to use a comparatively
smaller quantity of the volatile oil as a flavor.76
From the foregoing it would appear that apart from the
Pharmacopeia of the United States, practically all other
known pharmacopeias describe a water-white mineral oil
under the title “Paraffinum Liquidum” or “Liquid Paraffin”
as a colorless, odorless, tasteless, non-fluorescent, oily liquid,
free from acids, alkalies and organic impurities. As
explained before, the specific gravity of the preparation as
recognized in other countries and as offered on the American
market at the present time varies considerably, and there
appears to be some difference of opinion as to the exact
nature of the product that is preferable for use for different
purposes. This matter requires further investigation.
Since the definition of liquid petrolatum in the U. S.
Pharmacopeia permits the use of fluorescent products of
widely varying specific gravities, it is recommended that
physicians who desire the water-white non-fluorescent
(Russian) mineral oil should use the term “Petrolatum
Liquidum, Grave,” or “Paraffinum Liquidum, B. P.,” if the
heavy product recommended by Lane is desired, and “Petrolatum
Liquidum, Leve” if the light varieties are required.
It is further recommended that under the foregoing names,
manufacturers and pharmacists be requested to dispense the
products, in accordance with the following descriptions:
Petrolatum Liquidum, Grave.—Heavy (Russian) Liquid
Petrolatum.—Paraffinum Liquidum, B. P., liquid paraffin.—A
transparent, colorless, tasteless, non-fluorescent, oily liquid,
odorless when cold but giving off a faint petroleum odor on
heating. This preparation should correspond to the requirements
of the British Pharmacopeia for liquid paraffin and
have a specific gravity of about 0.885 to 0.890 at 15 C. It is
insoluble in water or alcohol but soluble in boiling absolute
alcohol and readily soluble in ether, chloroform, carbon
disulphid, petroleum benzin, benzene and fixed and volatile
oils. It serves as a solvent for volatile oils and related substances
like camphor, menthol and thymol.
This is the type of preparation used by Sir W. Arbuthnot
Lane, and his associates for internal administration. It is
also used as a basis for ointments and salves and as a
local application to wounds, ulcers and in certain forms of
skin diseases in which a simple protective is desired.
Petrolatum Liquidum, Leve.—Light (Russian) Liquid
Petrolatum.—A transparent, colorless, tasteless, non-fluorescent,
oily liquid, odorless when cold, but giving off a faint
petroleum odor on heating. In other respects this preparation
should correspond to the pharmacopeial tests for liquid
petrolatum and have a specific gravity of about 0.860 to 0.875
at 15 C. Like the heavy variety of liquid petrolatum, it is
insoluble in water and alcohol, but soluble in boiling absolute
alcohol and rapidly soluble in ether, chloroform, carbon
disulphid, petroleum benzin, benzene and fixed and volatile
oils. It serves as a solvent for volatile oils and related substances
like camphor, menthol and thymol.
This is a type of preparation extensively used as a
vehicle for the oily sprays in nose and throat work. It is
also being used as one of the constituents in the now
popular paraffin oil cold cream and has been used to some
extent for internal administration in the treatment of chronic
stasis. Being more limpid than the preparation preferred
by Lane, it is more readily taken, though greater care must
be exercised in securing a sample devoid of the lighter
fractions of petroleum distillates.—(From The Journal
A. M. A., May 30, 1914.)
CLINICAL EXPERIENCE WITH LIQUID PARAFFIN
(LIQUID PETROLATUM) [AB]
A Comparative Investigation Made Under the Auspices of
the Council on Pharmacy and Chemistry [AC]
W. A. Bastedo, M.D., New York
During the past three or four years, “mineral oil” has
come into extensive use in the treatment of constipation.
Preparations of the Russian and the American oil, both
heavy and light, have appeared on the market, but there
have been no satisfactory data on which to base a selection
of oil for use. Therefore, in order to obtain reliable clinical
information concerning the relative efficiency of the different
oils, the Therapeutic Research Committee of the Council
on Pharmacy and Chemistry of the American Medical Association
submitted samples of the oils to various clinicians
for testing. The following is a synopsis of the investigation,
which I have prepared at the request of the committee.
The collaborators were advised that specimens of the best
obtainable light Russian liquid petrolatum, heavy Russian
liquid petrolatum and an American brand of liquid petrolatum
would be sent out, but that, to avoid bias, these
specimens would be distinguished only by numbers or letters.
The oils sent out were (1) a light “Russian” liquid
petrolatum having a specific gravity of 0.860 at 20 C., (2)
a heavy “Russian” liquid petrolatum having a specific
gravity of 0.885 at 20 C., and (3) an “American” liquid
petrolatum having a specific gravity of 0.857 at 20 C. and
being markedly fluorescent. The collaborators were advised
that the reports should furnish information as to size and
frequency of dose, the agreeableness to the taste, the effect
on the stomach, the number and character of the stools, the
degree of admixture of the oil with the other ingredients
of the stool, the degree of leakage of oil about the anus,
and the need of other cathartic measures.
Reports have been received from Drs. L. F. Barker, W. A.
Bastedo, J. B. Champion, Henry A. Christian with C. K.
Drinker and F. A. Hatch, Alfred Stengel and R. L. Wilbur.
CONCLUSIONS
The conclusions to be drawn from the clinical reports are:
Dosage.—Half an ounce to 3 ounces a day. In the same
patient, the same amount of each of the oils was required.
Frequency of Dose.—The same amount daily seemed as
efficient when given in one dose as when given in divided
doses two or three times a day.
Agreeableness to the Taste.—There is a difference of opinion
in this regard. Two reports favored the heavy Russian oil.
One report favored the light Russian petrolatum. But the
taste of any of the samples was so slight as to be a
negligible quantity after the patient had taken the remedy
for two or three days.
Stomach Effects.—In about 20 per cent. of the patients, the
oil produced a slight degree of nausea or tended to repeat.
This is most likely in patients who have gastric stagnation
with retarded emptying of the stomach. All the oils acted
the same in this regard. Vomiting was reported in two
cases.
Number of Stools.—To produce one or two copious stools
a day the dose required varied considerably, but there was
no difference noted on account of difference in the specific
gravity or character of the oils.
Character of Stools.—The stools were soft, usually formed,
sometimes mushy, obviously greasy. They had a peculiar
odor described as sour. Their consistency varied with the
dose, but was the same for the different kinds of oil.
Admixture of Oil with Other Ingredients of Stool.—Generally
well mixed, but from time to time a patient would have
a stool of free oil. This occurred with all varieties of oil.
(It necessitated reduction of the dose, and if then the bowels
were not active enough, the administration in addition of
cascara, aloin, etc.)
Leakage About the Anus.—A disagreeable feature complained
of by many is that when they take enough of the
oil to move the bowels, there is sufficient leakage from the
anus to keep the neighboring skin continually in a greasy
condition, and sometimes to stain the clothes. That there
is any difference in this regard between the oils has not
been determined.
In the reports, one clinician noted no differences that were
not negligible. Another was slightly in favor of No. 2
(heavy Russian) as regards taste. A third reporter did
not make comparative tests. A fourth is slightly in favor
of “B” (heavy Russian) as regards taste and general suitability.
All of the findings of this investigation are based
on hospital cases. A fifth reporter favored No. 1 (light
Russian petrolatum). He considered it the most prompt
in its effect, the most uniform in results, and the most prone
to give a satisfactory admixture of the oil with the other
materials. The difference, however, from the other oils was
not marked. Another reporter noted no special differences.
SUMMARY
The results of this clinical investigation appear to warrant
the conclusion that so far as therapeutic results are
concerned the differences in the action of the three varieties
of liquid petrolatum, namely, light Russian liquid petrolatum,
heavy Russian liquid petrolatum and American liquid
petrolatum, are too slight to be of importance. Hence the
choice between the lighter and the heavier oils, and between
the Russian and the American is an open one, to be determined
not by therapeutic differences, but by palatability,
dependent on the degree to which the refinement of the oil
is carried out. The U. S. Pharmacopeia, the revision of
which is now nearing completion, no doubt will furnish
standards which will insure a suitable product. From the
findings of the foregoing report it would appear that a
satisfactory standard might permit the use of either Russian
or American oil, if suitably refined so as to be as nearly
as possible devoid of odor and taste.—(From The Journal
A. M. A., March 6, 1915.)
ANGIER’S EMULSION[AD]
Report of the Council on Pharmacy and Chemistry
Angier’s Emulsion is essentially a petroleum product.
When it was first put on the market commercial interests
had been fostering the idea that petroleum products had
food-value, and the manufacturers of Angier’s Emulsion,
making use of the idea, advertised it as a “food-medicine”
and an “ideal substitute for cod-liver oil.” The impression
thus created has been kept alive through persistent advertising
in spite of scientific proof to the contrary. To-day many
who know that petroleum products have no food-value are
still likely unconsciously to class Angier’s Emulsion among
nutrients. Although the manufacturers now advertise this
product as “purely mechanical in its action,” they yet show
a disposition to profit by the old misapprehension, since, so
far from expressly disavowing the old claims as erroneous,
they mingle with the new ones vague claims of “tonic and
reconstructive merits” apparently designed to sustain, in
those who do not take time to consider the evidence carefully,
the old faith in the claimed nutritive qualities of the
preparation.
While the Council judges a preparation by the claims
made for it at present, and not by any past misstatements
when these have been thoroughly corrected, the past advertising
of Angier’s Emulsion so instructively illuminates
the scientific worthlessness of proprietary therapeutic claims
in general, and the whole course of its history is so typical
that the referee has thought it well to review the subject
briefly. The Council has authorized the publication of the
following report.
W. A. Puckner, Secretary.
Angier’s Petroleum Emulsion was brought out in 1881—that
is to say, before the food-value of petroleum products
had been experimentally disproved. Its advertising history
well illustrates the weed-like vitality of a financially profitable
therapeutic fallacy. The shifting claims made for this
preparation are such good examples of the generally unreliable
therapeutic pretensions of proprietary medicines—whether
of the “patent medicine” or of the “ethical proprietary”
type—that it has been deemed advisable to present a
brief review of the conflicting claims made for it at various
times.
A PETROLEUM PRODUCT
Angier’s Emulsion is described by the manufacturers as
containing, in addition to “our specially purified Petroleum,”
“the combined hypophosphites of lime and soda, chemically
pure glycerine, and the necessary emulsifying agents.” So
far as the hypophosphites are concerned, it is probably unnecessary
to remark that the latest researches bring to light no
evidence that they influence metabolism in the slightest
degree. The Angier Chemical Company apparently accepts
this view, for in its advertising stress is laid exclusively
on the merits of the emulsion as a petroleum product. It is
therefore proper to consider it from this point of view.
The history of the internal use of liquid petrolatum was
sketched in a recent Council report.77 As mentioned at that
time, a number of petroleum products were put on the market
some thirty years ago as substitutes for lard and butter.
Contemporary opinions regarding the food value of such
products differed widely.
There never was any scientific evidence to support
the view that petroleum and its derivatives are assimilable
by the animal organism. In fact, so far as we can learn,
there was no scientific investigation of the problem until
Randolph’s experiments in 1884. These were probably the
first to demonstrate the non-absorbability of petroleum and
its valuelessness as a foodstuff.
In 1899 Robert Hutchison conclusively demonstrated by
experiment that petrolatum, paraffin and related products
were absolutely unassailable by any of the digestive fluids,
and therefore could not possibly have any food value.
Various investigators later confirmed these findings.
FIRST ADVERTISED AS A “FOOD-MEDICINE”
Let us now take up the advertising history of this nostrum.
In 1895 it was sold under these claims:
“... a ‘Food-Medicine’ that is far more than a substitute for
cod-liver oil”;
“... a Food-Medicine that is readily assimilated and helps to
digest other foods.”
In 1897 it was an:
“Ideal Substitute for Cod Liver Oil.”
In 1899 it:
“... conserves heat and energy by furnishing more material for
oxidation.”
In 1902 it:
“... supplants tissue waste by tissue reconstruction.”
The promoters of Angier’s Emulsion thus for some time
ignored the status definitely assigned to petroleum products
by the experiments of Randolph, Hutchison and others.
This was only natural. If petrolatum was absolutely inert
in the alimentary canal (and this was now proved beyond
controversy) then an emulsion prepared from it most certainly
was not a “food-medicine,” could not “supplant tissue
waste,” or “conserve heat and energy.” All the credit which
previous “unterrified and ingenious advertising” (to quote
Hutchison) had accumulated for Angier’s Emulsion was
bound up with the view that petroleum products were foodstuffs.
LATER ADVERTISED AS NON-ABSORBABLE
The non-absorbability of liquid petrolatum, however, suggested
to Robinson, Schmidt, Lane and others, a new therapeutic
use for it in the treatment of chronic constipation.
This method has rapidly gained popularity and it is not
surprising, therefore, that the promoters of Angier’s Emulsion
changed their claims accordingly, and now began to base
their advertising chiefly on the proved properties of petrolatum.
In 1910 the emulsion was advertised for the treatment
of chronic diarrhea on these grounds:
“... given by the mouth, it passes to the lowermost portions of
the intestines without changing its identity; hence it exerts antiseptic,
soothing and demulcent properties upon every inch of the intestinal
tract, from the duodenum to the rectum.”
The old claims, however, were not discarded altogether,
for in 1911 the preparation was recommended for children’s
diseases as:
“... an aid to appetite and digestion and a splendid tonic and
builder.”
Before long the attempt was made to weave together the
claims based on opposed and mutually incompatible properties.
In 1912 we find Angier’s Emulsion recommended
because it:
“... corrects digestive disturbance and promotes normal action
of the bowels. At the same time it has a most invigorating tonic
influence upon the general health.”
In 1914 medical men are advised through the advertising
pages of the British Medical Journal of the:
“... tonic and reconstructive merits of Angier’s Emulsion.”
A pamphlet on “Constipation,” which is “Presented to
Physicians with Compliments of the Angier Chemical Company”
(copyright, 1913; still distributed in 1914) informs
physicians that Angier’s Emulsion is:
“... purely mechanical in its action.”
Notwithstanding this, we are told later on in the same
pamphlet that it:
“... facilitates, hastens and assists the processes of digestion and
assimilation.” ... “is a most efficacious remedy in Pulmonary
Tuberculosis because it not only maintains normal nutrition, but also
exerts a well-defined specific palliative influence upon the cough and
other symptoms of the disease.”
Evidently the advertisement is written in the hope that in
one paragraph a claim based on the proved properties of
petroleum products may be substantiated, while in another
a totally different and inconsistent claim may be glibly insinuated
in vague phrases designed to lull thought and thus
perform the remarkable feat of securing credence for two
contradictory statements.
UNWARRANTED AND MISLEADING CLAIMS
Further evidence that Angier’s Emulsion is at present
exploited both to the medical profession and to the public
under claims that are unwarranted and misleading, if not
as palpably untrue as the claims made in the past, is found
on the wrapper of a trade package purchased in 1914 and in
the circular accompanying it. Note the following:
“Indicated in Diseases of the Throat and Lungs and of the Digestive
Apparatus. Useful in General Debility and Wasting Diseases, Especially
when due to Faulty Nutrition. The antiseptic properties of the Emulsion
particularly adapt it to the treatment of diseases of septic or
bacterial origin.”
“Angier’s Petroleum Emulsion is indicated in affections of the throat,
lungs and intestinal tract—both subacute and chronic. In diseases
of the digestive apparatus due to catarrhal, ulcerative or tuberculous
conditions, its peculiar soothing, healing and aseptic properties make its
use especially beneficial. Wasting diseases, particularly when due to
faulty nutrition, are greatly benefited by its use, one of the most
noticeable effects being a prompt and decided increase in weight.”
It is, of course, unnecessary to point out that, since petroleum
is non-absorbable, Angier’s Emulsion contains no ingredient
capable of affecting the respiratory mucous membrane
except by local application, for which, indeed, this preparation
is evidently not intended.
COMPOSITION AND FORMULAS
According to a circular which was contained in a trade
package recently purchased
“Each fluidounce of Angier’s Petroleum Emulsion with hypophosphites
contains: 331⁄3 per cent. of our specially purified Petroleum; 9
grains of the combined hypophosphites of lime and soda, chemically pure
glycerin and the necessary emulsifying agents.”
As regards the nature of the product referred to under the
indefinite term “petroleum” the circular states that Angier’s
Emulsion is:
“... prepared with refined petroleum specially purified for the
purpose. By a process peculiarly our own the crude petroleum, obtained
from special wells is so purified that all taste and odor and all objectionable
and irritating properties are removed, while the full medicinal
value of the oil is retained....”
The composition assigned to Angier’s Emulsion in an
advertising pamphlet “The Petroleum Idea,” issued in 1907
differs in that it is said to contain “specially purified crude
petroleum” and that each fluidounce is said to contain 2.84
grains of benzoate of sodium. While these quotations convey
the impression that certain medicinal constituents of the
“specially purified” product obtained from “special wells”
are “retained,” a pamphlet recommending the use of Angier’s
Emulsion for the treatment of constipation assures us that
it produces the “mechanical effects of the purest petroleum”
and that it is “purely mechanical in its action.”78
LABORATORY REPORT
The statements regarding the identity of the “petroleum”
are so unsatisfactory and contradictory (in one place “refined
petroleum specially purified for the purpose,” in another
“specially purified crude petroleum”—in one place “medicinal”
and in another “purely mechanical in its action”) that the
help of the Chemical Laboratory of the Association was
invoked to establish the character of the petroleum product
and to determine the presence or absence of sodium benzoate,
at one time declared by the manufacturers to be present
but later omitted from the formula. The Association’s
chemists reported:
“From a specimen of Angier’s Emulsion recently purchased
there was separated by the customary methods of
analysis, a yellow fluorescent, unsaponifiable, semi-solid
residue which has all the properties of ordinary yellow
petrolatum of a consistence somewhat softer than the product
described in the Pharmacopeia. It was much more
dense than the colorless, non-fluorescent liquid petrolatum
now in vogue as a laxative. The preparation contained
benzoate, both in the form of free benzoic acid and also
in the form of a water-soluble salt probably sodium
benzoate.”
The petroleum product contained in the emulsion was thus
shown to be intermediate between the ordinary (solid) and
the liquid petrolatum. It also appears that a benzoate is
still present, though no longer mentioned in the formula.—(From
The Journal A. M. A., Sept. 12, 1914.)
PHECOLATES, PHECOLAX, PHECOZYMES AND
PHECOTONES
Report of the Council on Pharmacy and Chemistry
Phecolates, Phecolax, Phecozymes and Phecotones were
submitted by F. Waldo Whitney, New York, with “literature”
indicating that they are designed to form parts of a system
of treatment founded on the theory of autotoxemia, which
they are supposed to prevent by their action on the functions
of the intestinal canal. The different preparations consist in
the main of mixtures of well-known remedies. The basic
preparation is Phecolates, which contains bile salts in combination
with phenyl salicylates and benzo-naphthol in about
one-eighth the regular doses and hence not likely to be of
any real service. Since the proportions of these ingredients
ought to be regulated by the physician according to the needs
of the individual patient, they should not be combined in
fixed proportions. The name is not so framed as to indicate
the principal ingredients.
Phecolax contains, in addition to the ingredients of Phecolates,
phenolphthalein and cascarin, of each one-half grain.
Phecozyme is made more complex than Phecolax by the
introduction of additional phenyl salicylate and of pancreatin.
Phecotone contains ten ingredients.
Extravagant claims such as the following are made:
“Our Health is governed by our bowels; Our bowels are governed
by our nerves; Our nerves are governed by our digestion; our digestion
is governed by Phecolates.”
The Council voted to refuse recognition to Phecolates,
Phecolax, Phecozymes and Phecotones as unscientific articles
with objectionable names.—(From The Journal A. M. A.,
Nov. 21, 1914.)
PHENOL SODIQUE
Report of Examination by Council on Pharmacy and
Chemistry and Comments
An examination of this article by a subcommittee of the
Council on Pharmacy and Chemistry revealed unscrupulous
claims which are a positive menace to public health. In
view of this the Council has directed the publication of
the following comments.
W. A. Puckner, Secretary.
COMMENTS
Phenol Sodique was not submitted to the Council by the
manufacturers, but was taken up because it is advertised to
both physicians and the public. Some advertisements state:
“Phenol Sodique was the standard antiseptic thirty years
ago. It’s the same today.” If this were true, it would be
high time to call a halt; for the unscrupulous claims made
for this nostrum, and the effrontery with which they are
pushed, are only rivaled by those of the most shameless
“patent medicines.”
The firm of Hance Bros. & White poses as a reputable
pharmaceutical manufacturing house, but how it can reconcile
this position with their method of exploiting this product
passes all understanding. In the original package of Phenol
Sodique (the latest was purchased on June 20, 1907), there
are little booklets and a folder describing the marvelous
properties of the nostrum. The booklets do not refer to
Phenol Sodique, but they are very instructive. They are
entitled: “Dyspepsia,” “Worm News,” and “Catarrh,” advertising
“Dyspepsia Stop”—some form of dyspepsia tablets,
a remedy for round worms, and “Catarrh Stop,” apparently
some mild antiseptic tablets. These booklets are addressed
frankly to the laity, although recourse to a physician is,
generously, advised if the patient does not respond to treatment!
The folly of prescribing “original packages” which
contain popular literature has been so often emphasized that
further comment seems superfluous. The following from
“Catarrh,” however, throws an interesting sidelight on the
scientific status of Hance Bros. & White:
“Catarrh is due to a minute insect in the inner lining
membrane of the nose. This insect multiplies rapidly,
and, unless checked, and destroyed, will produce the
worst results.”
To return, however, to Phenol Sodique: The folder is
also evidently intended for the lay public rather than for
physicians; at least, if we are to credit Hance Bros. &
White with any intelligence whatsoever. It is headed:
“Montyon Prize of Encouragement, Awarded by the Institute
of France, 1861.” This is rather ancient, but what follows
indicates that a little restraint would have been better
than encouragement. The circular is a compact treatise on
self-medication—apparently all that is necessary to retain
or regain health is the use of Phenol Sodique, externally
and internally. The following conditions are among those
specifically named as amenable to this remedy. Smallpox,
measles, scarlatina, erysipelas, puerperal fever, typhoid fever,
cholera, diarrhea, cramps, burns and scalds, bites, cuts and
wounds, excoriations, chilblains, chaps, sore throat, scratches,
catarrh, tetter, sunburn, swollen veins, ulcers, hemorrhages,
bruises, piles, gangrene, carbuncle, itching, insect stings,
ivy poison, cold in the head, bunions, inflamed eyes, eczema,
ringworm, rheumatism, pains, toothache, seat worms, etc.—besides
numerous diseases of animals.
No antiseptic, whatever its composition, could by any
possibility accomplish anything like what is claimed for
Phenol Sodique, so that the composition of the article is
really of little importance. This is evidently appreciated
by the manufacturers, for they have kept the composition a
profound secret, except in so far as it is implied in the name.
An inquiry addressed to Hance Bros. & White, under date
of April 27, 1907, six months ago, has remained unanswered.
The Council, therefore, directed an analysis of Phenol
Sodique. This was carried out at the chemical laboratory
of the American Medical Association, and a check analysis
was made by an independent firm of chemists.
This shows that Phenol-Sodique contains something like
0.5 or 0.66 per cent. of phenols, dissolved in about 0.75 per
cent. of sodium hydroxid. In other words, it appears to
be essentially a very dilute alkaline solution of some impure
coal-tar product, presumably a crude carbolic acid. The
analysis could not profitably be carried further, because
the amount of the antiseptic agent is so very small.
The consideration of this analysis, in connection with the
claims made for Phenol-Sodique, leaves little doubt as to
one reason for the secrecy concerning its composition;
although no educated physician could be deceived into
believing for a moment that Phenol-Sodique could fulfil the
promises of its promoters, even if it were “the best antiseptic,
hemostatic and disinfectant on the market,” as the
manufacturers say in their advertisements.
From its composition, it can only have the very moderate
and ordinary antiseptic qualities of a dilute phenol or cresol
solution, modified only to a very slight extent by the free
alkali. According to the manufacturers, however, “Phenol-Sodique
is a wonderful preparation.” Just how wonderful
appears from these extracts from the dissertations in the
pamphlet which is enclosed in the package.
“Catarrh, Old Colds, etc.—Drink every morning and evening a glass
of water containing ten to thirty drops of Phenol-Sodique ...”
“Small-Pox.—To prevent attack take internally three or four times
a day, fifteen or twenty drops of Phenol-Sodique in one tablespoonful
of sugar and water....
“Measles, Scarlatina and Erysipelas.—Same treatment as for Small-pox.”
“Typhoid Fever.—To prevent attack take internally three or four
times a day, fifteen or twenty drops of Phenol-Sodique.”
“Cholera.—To prevent, spread sawdust or sand, wet with Phenol-Sodique,
in apartments.
“The very best precaution is to drink, morning and evening, a
glass of water containing from fifteen to thirty drops of Phenol-Sodique....
“... Premonitory Diarrhea.—... Drink a teaspoonful of
Phenol-Sodique diluted in an ounce of water....”
This is the kind of therapeutics and prophylaxis taught
to the medical profession by their self-appointed instructors,
the proprietors!
But this matter has a serious as well as a ludicrous side:
What is the proper epithet to apply to those who, knowingly
and intentionally, impress on the ignorant lay public that
one can with impunity expose himself to smallpox, cholera,
typhoid or scarlet fever, or measles, by taking a few drops
of very dilute carbolic acid, or by sprinkling a little on
sawdust? What must be the consequences to those who
trust in these assurances? And what should be the lawful
penalty for those whose blunted moral instincts permit them
wilfully to endanger the lives of others for a little financial
gain? It would be interesting to know the real opinion of
the responsible members of the firm of Hance Bros. & White
on these questions.
The Montyon Prize was awarded by the French Institute
in 1861—forty-six years ago—how many victims a year?—(From
The Journal A. M. A., Nov. 9, 1907.)
PHYTIN AND FORTOSSAN
Report of the Council on Pharmacy and Chemistry
Phytin, manufactured by the Society of Chemical Industry,
Basel, Switzerland, and sold by A. Klipstein and Co., is an
organic phosphorus compound said to be the “Acid Calcium-Magnesium
Salt of Phytinic Acid (Inosit Phosphoric Acid
or Anhydro-Oxymethylene-Diphosphoric Acid)” obtained
from cereals and legumes.
The trade package of Phytin constitutes an indirect advertisement
to the public.
The Council rejected Phytin because unwarranted and
exaggerated therapeutic claims are made for this product
based on the entirely undemonstrated assumptions: (1) that
phosphorus is assimilated only from organic combinations
(it is even implied that this must be in the form of Phytin,
and that milk is incapable of supplying the phosphorus needs
of infants); (2) that a long list of diseases, ranging from
rickets to hysteria, are due to deranged phosphorus metabolism;
(3) that all these diseases are cured or markedly
benefited by Phytin.
In brief, the claims rehearse every point of the more or
less discredited phosphorus propaganda, in exactly the same
way as it was rehearsed successively by the exploiters of
hypophosphites, lecithin, glycerophosphates, and amorphous
phosphorus. It is conceded by the writers of the advertising
pamphlets for Phytin that the preceding claims were erroneous;
but no evidence is given to warrant the belief that
the Phytin claims are less erroneous.
The misleading statements are most extreme. By the use
of bold type particular stress is laid on the preposterous and
vicious claim that Phytin
“radically and permanently removes sexual debility.”
Fortossan is a preparation of Phytin and sugar of milk,
also manufactured by the Society of Chemical Industry,
Basel, Switzerland, and sold by A. Klipstein and Co. Since
Fortossan is a simple preparation of Phytin the Council voted
that the rejection of Phytin should also apply to Fortossan.—(From
The Journal A. M. A., Jan. 30, 1915.)
PRUNOIDS
Report of the Council on Pharmacy and Chemistry
Prunoids are tablets put out by the Sultan Drug Company,
St. Louis. They are said to be:
“Made of Phenolphthalein (one and one-half grains in each), Cascara
Sagrada, De-emetinized Ipecac and Prunes.”
The following report on the composition of Prunoids is
submitted by the Association’s Laboratory:
“From an examination of Prunoids it is concluded that the
amount of cascara or extract of cascara in the preparation
is very small. Also the quantity of “de-emetinized
ipecac” is insignificant. The claim is made:
“The levulose of prunes, a constituent of Prunoids, is hygroscopic
and thus when brought into contact with the saliva of the mouth or
contents of the stomach, disintegrates and prompt medication is
insured.”
“Actually the amount of prunes which may be present in
Prunoids is negligible. For all practical purposes, therefore,
Prunoids are phenolphthalein.”
According to the information included on and in the box
Prunoids are
“An Ideal Laxative, Purgative, and Intestinal Tonic” ... “particularly
adapted to the treatment of constipation ...”
They are said to act as an “intestinal tonic”—a claim
which in the light of the examination is obviously unwarranted—and
because of this, it is said that they:
“Will permanently remove constipation without causing after constipation.”
The trade package assures the purchaser that Prunoids are:
“Recommended by Physicians Generally.”
A circular sent to physicians makes the unwarranted claim
that Prunoids are “especially serviceable” in “... Neurasthenia,
Jaundice, Chlorosis, Rheumatism, Gout ...”
and that
“... their success in gouty diathesis and vague rheumatic symptoms
tends to confirm the opinion expressed by some physicians that
they have a solvent action on uric acid.”
In the following the haphazard and ill-considered use of
purgatives is suggested:
“For the expectant mother, or in the treatment of female diseases,
for bowel elimination, no happier or safer selection can be made.”
The Council refused recognition to Prunoids because the
statement of composition is incomplete and therefore meaningless;
because unwarranted therapeutic claims are made
for them; because the name “Prunoids” gives the false impression
that they depend on prunes for their effect; and because
it is irrational and a detriment to medicine to disguise a
well-known drug by means of a misleading name and to
attempt to create the impression of special virtues by combining
it with superfluous drugs.—(From The Journal
A. M. A., Jan. 2, 1915.)
SAL HEPATICA
Report of the Council on Pharmacy and Chemistry
Sal Hepatica, marketed by the Bristol-Myers Co. of New
York, has been refused recognition by the Council, because
its composition is secret; because it is advertised indirectly
to the public for the treatment of diseases; because exaggerated
and unwarranted claims are made for its therapeutic
qualities; and because the name fails to indicate its chief
constituents but does suggest its use in liver disorders.
The Council has authorized the publication of the report of
its referee, because it is an important illustration of the ways
in which physicians are being made parties to the introduction
to the public of a patent medicine, whose indiscriminate
use must often have resulted in harm, direct or indirect.
W. A. Puckner, Secretary.
The report of the referee follows:
Sal Hepatica is a saline laxative sold by the Bristol-Myers
Company of New York. No information seems to be given
regarding its composition except such as is contained in the
following vague and uninforming phrases:
“Effervescent saline combination, hepatic stimulant, laxative and
an eliminant of irritating toxins.”
“Sal Hepatica is a saline combination containing the alterative
and laxative properties similar to the natural ‘Bitter Waters’ of Europe
with the addition of sodium phosphate.”
“... more palatable and efficient than sodium phosphate alone or
other salines.”
A circular around the bottle contains the following:
“We invite the physicians’ careful consideration of the merits of
Sal Hepatica in the treatment of Rheumatism and Gout, in Constipation
and Auto-intoxication, and to its highly important property of cleansing
the entire alimentary tract, thereby eliminating and preventing the
absorption of irritating toxins and relieving the conditions arising from
indiscretion in eating and drinking.”
In the same circular, its promiscuous use is invited in
these terms:
“Owing to its palatability, Sal Hepatica is particularly well adapted
to the requirements of childhood or the feeble and delicate.”
Further suggesting its use in the treatment of that popular,
if somewhat vague ailment, “biliousness,” we read:
“It is especially valuable where there is intestinal sluggishness arising
from functional derangements of the liver or portal circulation....”
As further suggestive of its all-around “goodness,” are the
claims:
“It increases the appetite and promotes digestion by stimulating the
flow of gastric juice.”
“In rheumatism and gout Sal Hepatica furnishes the physician with
an ideal eliminant, usually affording prompt relief.”
The label on the Sal Hepatica bottle suggests—both to
physicians and to the public—its use in the following diseases
and conditions:
“Derangements of the stomach and liver.”
“Affections of the kidneys.”
“Bilious attacks.”
“‘Summer complaints,’ colic and alcoholic excesses.”
“Headache, dizziness, heartburn and seasickness.”
“Acute indigestion.”
“Gastric, hepatic and renal disorders.”
“Especially beneficial in rheumatism and gout.”
From these quotations it is evident that Sal Hepatica is
in conflict with:
Rule 1, in that its composition is not disclosed, although
statements are made which are likely to give a false impression
as to what it is;
Rule 4, in that the statements on the label and in the circular
around the bottle advertise it to the public and thus
make the physician who recommends it an advance agent
for the nostrum;
Rule 6, in that exaggerated and unwarranted claims are
made for its therapeutic qualities, and,
Rule 8, in that its name fails to indicate its chief constituents,
but does suggest its use in liver disorders.
The absurd claims made for this preparation are such as
to put it in the “patent medicine” class. Even the most
credulous members of the medical profession certainly can
take no stock in the claim that a preparation can be an
“eliminant” of uric acid, a hepatic stimulant, a remedy for
gout, rheumatism, liver disease, indigestion, etc. Why then
should such a preparation be tolerated?
In its conflict with Rule 4 Sal Hepatica belongs to that
class of nostrums which have been so successfully exploited
by manufacturers through the unwitting efforts of thoughtless
and careless physicians. The Bristol-Myers Company
has been most liberal in distributing free samples, evidently
with the assurance that physicians would do the rest. Thus,
at the present time, the profession is being supplied with a
package containing one regular 25-cent bottle and five single-dose
vials bearing the name Sal Hepatica. If only a small
percentage of the physicians who receive these samples distribute
them, the increase in Sal Hepatica consumers may
be imagined. How successful this scheme of the Bristol-Myers
Company has been is only too evident. Sal Hepatica
is one of the best-selling laxatives in department stores and
drug stores to-day.
While the evils of indiscriminate purgation are now generally
recognized, the referee wishes to quote and to indorse
the pertinent comments on this subject by The Journal:79
“The abuse of saline cathartics by the public is an evil
deserving of serious attention. Rightly or wrongly, the laity
fear constipation and naturally take what they are taught
to believe is the cheapest and simplest course for its relief,
self-drugging by means of saline cathartics or the extensively
advertised purgative mineral waters. This habit is responsible
for much of the distressing spastic constipation that exists,
and its accompanying neurasthenia. The advertisement and
sale to the laity of such a nostrum as “Sal Hepatica” can only
increase these evil results and the physician who aids and
abets the evil by using the preparation should reflect whether
he is thereby not only encouraging a fraud on the public but
also, what is even worse, helping to impair the public health.”
It is recommended that this report be authorized for publication
in order that physicians may know the extent to
which they have been made to act as advance agents for
“patent medicines.” It is hoped its publication may suggest
to those who in thoughtlessness have recommended Sal
Hepatica, that they go to their materia medica and renew
acquaintance with the host of simple and efficient laxative
salts which are available—magnesium sulphate, sodium sulphate,
sodium phosphate and the palatable effervescing preparations
of these which the Pharmacopeia provides—effervescent
magnesium sulphate (Magnesii Sulphas Effervescens,
U. S. P.), effervescent sodium phosphate (Sodii Phosphas
Effervescens, U. S. P.).—(From The Journal A. M. A.,
Feb. 7, 1914.)
SANMETTO
Report of the Council on Pharmacy and Chemistry
The following report on Sanmetto (Od Chemical Company,
New York) has been adopted by the Council on
Pharmacy and Chemistry, which authorized its publication.
W. A. Puckner, Secretary.
Sanmetto is one of the oldest proprietaries on the market.
Its advertisements have been familiar to the readers of
medical journals for several decades past. It is a typical
nostrum. It is secret although the promoters have published
various “near-formulas.” The following are some of the
statements regarding composition:
“A Scientific Blending of True Santal and Saw Palmetto with Soothing
Demulcents in a Pleasant Aromatic Vehicle.”
As this did not disclose the identity of the demulcents or
the quantity of the alleged active constituents, the “formula”
was, of course, meaningless.
Again it is:
“A Scientific blending of true Santal and Saw Palmetto in a pleasant
aromatic vehicle.”
Here the reference to “soothing demulcents” is omitted.
The information furnished physicians at the present time is:
“It is a blend of harmonizing drugs.”
A letter from a physician requesting information as to the
exact composition of Sanmetto recently elicited the following
reply:
“... Sanmetto is a blending of true santal and saw palmetto with
soothing demulcents in a pleasant aromatic vehicle. The demulcents
are introduced not only for the purpose of modifying the irritant properties
of the santal, but to add distinctively to the soothing properties of
the finished product upon the mucous membrane of the urinary tract,
and are not mentioned in our published formula for the simple fact
that if we gave them, then we would do the advertising and the substitute
manufacturer would engage in the ‘unfair competition’ of putting
on the market his concoction, claiming to be made exactly after our
formula, without spending a cent for advertising, relying upon our
propaganda work to sell his substitute, although not the same article
as nor equivalent to Sanmetto, from the fact that he would be working
in the dark as to the processes in the manufacture of our product.
There is no mineral substance in Sanmetto, nor any other ingredient
that is detrimental in any way whatsoever....
“OD CHEM. CO.,
“M. Haman, Pres’t.”
THE VALUE OF SANTAL AND SAW PALMETTO
The foregoing warrants the assumption that the active
ingredients of the mixture are sandalwood oil and saw
palmetto.
There was a period when the internal treatment of gonorrhea
had a marked vogue. Balsamic remedies received the
approbation of the medical profession as the most specific
of internal remedies for this disease. As a representative of
this class, sandalwood oil was very highly esteemed and had
great popularity. As in other similar instances, this popularity
was commercialized and the drug became the basis of
many secret or semisecret mixtures, including “specialties”
of pharmaceutical houses.
Sabal or saw palmetto is an official drug which at one
time was used in genito-urinary affections, but now is seldom
used, presumably because it has been found practically worthless.
It is not mentioned by most pharmacologists, and those
who do mention it regard it as of doubtful value. It is
included among the preparations recommended for deletion
as given in the report of the Committee on the Pharmacopeia
of the American Medical Association (The Journal, Sept. 4,
1909, p. 792).
Even granting that sandalwood oil and saw palmetto do
have therapeutic value, no one would think of regarding
either or both of these preparations as of use except in
inflammatory conditions of the genito-urinary tract, especially
gonorrhea.
If one is to believe the advertisements, however, the combination
of these drugs in Sanmetto is a wonderful medicine.
One might even conclude that there are few conditions in
which it cannot be given with profit. For instance:
“In Nervous Diseases, especially Neurasthenic cases with origin in
some sexual or genito-urinary disorder, for its action as a vitalizing
tonic and reconstructive, restoring nutrition to germ plasm, relieving
pathological conditions and for soothing and sustaining the nerves controlling
the parts.”
Bear in mind in reading the foregoing statement and the
following that we are concerned with two drugs whose effects
are exerted on mucous membranes especially of the genito-urinary
tract.
“In Gestation Cases, showing tendency to albumin and convulsions,
for toning the pelvic organs, clearing up the urine and cleansing the
urinary bladder and outlet. In the Lying-in-Room for relieving the
affections of urethra and bladder, painful strangury of the urethra and
painful micturition due to the pressure of fœtal head upon the neck
of the bladder and upon the urethra during labor, and infection, either
septic or gonorrheal.”
“In Weakness of the Kidneys, causing loss in tone and general
health and Impairment of Eyesight—for strengthening the kidneys and
bladder and toning the nervous system; and also for aiding in the constitutional
treatment of Gonorrheal Infection of the Eyes.
“In the treatment of the Prostate, Testes, Mammæ, Ovaries, and
Urethra, Kidneys and Bladder, for its soothing, slightly antiseptic,
aphrodisiac, toning and restoring action to the mucous membrane and
glands. By its use the parts affected in many cases returning to their
normal condition.”
While the reference to its aphrodisiac action and to the
restoration of parts to the normal may have little interest
to physicians, it may be counted on to appeal to the sexual
neurasthenic. In premature senility:
“Sanmetto ... is unexcelled as a vitalizing tonic to the withered
glands of the reproductive system, promoting their normal secretory
activity.”
These claims are not only absurd but also harmful; they
tend to perpetuate a hypochondriacal state of mind in the
class of patients appealed to—the sexual neurasthenic. There
is, however, a more serious side; the tendency of certain
other claims made for the preparation are vicious and dangerous
as well as misleading. The advertising claims are
likely to induce some physicians—those who accept advertising
“literature” as dependable—to belittle the importance of
serious diseases of the sexual organs and to be content with
Sanmetto, which, even if it gave as good results as other
balsamic remedies, would be, at best, only a halfway measure.
This in an advertising pamphlet physicians are given
this advice as to the treatment of gonorrhea.
“To provide the needed rest the patient should be instructed to
simply keep the parts clean with warm water for the first week and let
the discharge continue until you can control it by internal medication.
I wish to emphasize the fact that there is no way that any acutely
inflamed portion of the genito-urinary tract can get the rest required
so completely as by administration of Sanmetto.... After the
acute gonorrhea has begun to subside the Sanmetto should be aided by
mild astringent injections.”
If there is any well-established fact in medicine, it is that
gonorrhea is a serious disease—serious alike to the sufferer
and to the community—and one which needs careful attention
from the very first. To claim, either directly or by
implication, that it can be cured by such a mixture designed
to act on the kidneys, bladder and nervous system is false
and dangerous doctrine.
The physician who prescribes Sanmetto prescribes a secret
medicine for conditions which he is presumably competent
to treat with simple remedies of which he knows the origin
and action and which he can vary to suit the needs of the
individual.
Sanmetto is a secret nostrum the exploitation of which is
an invitation to haphazard, uncritical therapy and a menace
to public health.—(From The Journal A. M. A., March 13,
1915.)
SECRETOGEN
Report of the Council on Pharmacy and Chemistry
The Council has authorized publication of the following
report dealing with two internal secretion specialties—Secretogen
Elixir and Secretogen Tablets—to call attention to the
unfounded and extravagant claims made for this class of
products.
W. A. Puckner, Secretary.
Test tube experiments show that pepsin hydrolyzes proteins
in acid solutions; that pancreatin digests protein in
alkaline liquids, and that diastase converts starch into sugar.
Based on these facts, it was assumed that these ferments
would aid digestion. This assumption was correct if limited
to certain cases of dyspepsia in which it can be shown that
certain ferments are absent or deficient. But this limitation
was not realized or remembered; on the contrary, the indiscriminate
use of digesting ferments in all kinds of cases of
indigestion became widespread and still continues, although
to a less extent. Herein lies the great disappointment that
has followed the use of these ferments.
More recently hormones were discovered, and while their
importance has not been fully worked out, it has been
assumed that they are responsible for the secretion of digestive
ferments, and that in their absence this secretion fails.
Without waiting for proof of this assumption, that is, that
digestive failure is due to lack of hormones, proprietary
medicine promoters are already placing on the market
various secretion specialties.
As an example of this new class of specialties and of
the unfounded claims made for them, your referee presents
the following report on Secretogen Elixir and Secretogen
Tablets offered to physicians by the G. W. Carnrick
Company.
Secretogen Elixir is said to contain pancreatic secretin
obtained from the duodenum with 1⁄10 of 1 per cent. of
hydrochloric acid. Secretogen Tablets are said to be prepared
from pure secretin and succus entericus obtained from
the epithelial cells of the duodenum. The claims for Secretogen
are based on the physiologic action of secretin as
described by various observers. To determine whether these
claims are justified it becomes necessary to review the evidence
advanced to prove that secretin stimulates the digestive
glands.
Secretin is a hormone, a chemical substance produced by
the action of hydrochloric acid on a previously formed substance,
“prosecretin,” contained in the cells of the intestinal
mucous membrane, especially of the duodenum. Secretin is
absorbed by the blood and carried to the pancreas, liver and
intestinal mucosa, which are thereby stimulated to produce
their characteristic secretions, namely, bile, pancreatic juice
and succus entericus. When secretin is injected into the
blood, it causes an increase in the flow of these secretions.
Some observers have claimed that secretin is absent in cases
of diabetes in which the pancreas is still found normal.
Wentworth80 reported several cases of marasmus in which he
found no evidence of prosecretin. This deficiency, he believes,
is the cause of this disease.
The Carnrick Company, adopting the foregoing views,
namely, that secretin is necessary to secure the normal
action of pancreas, liver and intestine, as proved, placed on
the market their specialty “Secretogen,” to take the place of
the missing secretin.
The foregoing conclusion cannot, however, be sustained.
There are numerous cases in which no hydrochloric acid is
produced in the stomach and hence—as it is produced by
the action of hydrochloric acid—no secretin can be produced
in the intestine. Yet in these cases the pancreatic juice and
bile are secreted in normal amounts and digestion goes on
normally after the food leaves the stomach. In such cases
the pancreas and liver must be stimulated to secretion by
some other mechanism than secretin.
The proof that the absence of secretin is characteristic of
diabetes or of marasmus is not yet available. Sweet and
Pemberton81 found that many circumstances interfered with the
extraction of secretin, so that the mere failure to obtain it in
a given case is not proof of its absence, unless the various
inhibiting influences are given due consideration. The conclusions
reached by these authors are that “the evidence so
far adduced that secretin is absent in some varieties (of
diabetes) does not seem conclusive,” and that “the specific
absence or deficiency of secretin in marasmus seems to
remain as yet unproven.”
The favorable reports of Moore82 in regard to the use of
secretin in diabetes are not confirmed by the experience of
Foster83 in five cases, or by the case reported by Dakin and
Ransom.84
In regard to the use of secretin in intestinal disorders, the
G. W. Carnrick Company refers to an article by J. W.
Beveridge.85 An examination of this article shows it to be
unscientific and uncritical. The author presents four cases
to “demonstrate the peculiar potency exercised by secretin.”
Of the first he says:
“Stomach was dilated, food delay, seventy-two hours; hyperacidity,
vomiting daily, five to twelve times, urine high specific gravity, over
3 per cent. urea, trace albumen.”
The patient improved somewhat after gastro-enterostomy
with removal of the gallbladder; the vomiting ceased, but the
stools continued clay-colored and the high urea output still
kept up. Secretin was given, and after this the report
continues:
“The stools became normal in color at the end of the second month,
weight gradually increased until 1223⁄4 pounds was reached, and the
urea is now normal, averaging about 1 per cent.”
This case is offered to prove the absence of secretin and
its effect when given by the mouth. As evidence of hepatic
insufficiency the author apparently relies on the color of the
stools, and for pancreatic insufficiency he cites the high urea
output. He claims that when the pancreas does not furnish
an efficient secretion, the proteins of the food fail to be
converted into amino-acids, and instead, raise the percentage
of urea. Consequently, he concludes that a high percentage
of urea indicates the absence of secretin. It is usually held
that a high percentage of urea depends on two factors, ingestion
of a large amount of protein and concentration of the
urine. The author gives no data as to the amount of albuminous
food, the amount of urine, or whether the percentage
of urea was learned by examining a single specimen or the
total quantity for twenty-four hours. The mildest judgment
that can be passed on such clinical data is that they are
totally inadequate. Without doubt the percentage of urea
could have been reduced to “normal” by causing the patient
to drink water freely. The remaining cases show similar
hasty conclusions from insufficient data, rendering them
worthless as evidence.
The G. W. Carnrick Company introduces a number of
testimonials as to the value of Secretogen. These testimonials
are similar to all testimonials. They include no
evidence of careful diagnosis, and present an uncritical estimate
of the results. They show that the writers have given
Secretogen Elixir or Tablets indiscriminately in almost the
whole range of digestive disorders, in nephritis, neuralgia,
liver disease and gallstones, exophthalmic goiter, neurasthenia,
epilepsy, etc. As dependable evidence, these testimonials
are not worthy of consideration.
A rational basis for the therapeutic value of Secretogen is
lacking for the following reasons:
1. No evidence has been presented that the absence of
secretin is a cause of gastro-intestinal diseases. It is usually
present, and if not present, as in achylia gastrica, there is
evidently some compensating arrangement by which the pancreas
is stimulated to perform its regular functions.
2. There is no evidence that secretin in any form is physiologically
active when administered by the mouth.
REFERENCES
Fleig, M. C.: Action de la sécrétine, Arch. gén. de méd., lxxx, 24.
Charles, J. R.: Treatment of Diabetes with Secretin, Bristol Med.-Chir.
Jour., September, 1906; Med. Press and Circular, Nov. 21, 1906.
Meltzer, S. J.: Animal Experimentation in Relation to our Knowledge
of Secretions, Especially in Internal Secretions, The Journal
A. M. A., May 7, 1910, p. 1506.
Wentworth, A. H.: The Cause of Infantile Atrophy, Deduced from
A Study of Secretin in Normal and Atrophic Infants, The Journal
A. M. A., July 20, 1907, p. 204.
Bambridge and Beddard: Guy’s Hospital Reports, 1907, lxi, 161.
Enriquez and Hallion: Nuevas nociones sobre la digestion. Secretin,
Importancia fisiologica y patologica, Transactions of 14th Int. Med.
Congress, Madrid, 1904.
Enriquez: La Sécrétine Médication acide duodénale Stimulation de
function sécrétiniques chez l’homm., Rev. de. thérap. méd.-chir., Paris,
1904, lxxi, 187.
—(From The Journal A. M. A., May 1, 1915.)
SINKINA
Report of the Council on Pharmacy and Chemistry
Sinkina is a malaria “cure” put on the market by the
Metropolitan Pharmacal Company, New York. The product
was presented to the Council on Pharmacy and Chemistry
for admission to New and Nonofficial Remedies and was
rejected because insufficient evidence was submitted to substantiate
the improbable claims made for it. The manufacturers
were sent a copy of the report stating that their
product was refused recognition. In view of the advertising
that was persisted in after its rejection, the Council’s
referee for Sinkina submitted the preparation to clinical
tests. Both the original report and the results of the clinical
tests are given in the following report, which was submitted
to the Council and recommended for publication. The complete
report having been sent to the manufacturers and their
reply considered, the Council authorizes its publication.
W. A. Puckner, Secretary.
THE COUNCIL’S FIRST REPORT
The Council, after investigating the claims made for
Sinkina, declared the product unworthy of recognition and
adopted the following report, which was sent to the manufacturers:
No experimental evidence regarding the therapeutic value
has been submitted. The clinical evidence is scant and not
of such character as to deserve much consideration, no
sufficient precautions having been adopted to avoid wrong
conclusions. Judging from the evidence at hand the preparation
is simply a dilute sugar-alcohol-water solution containing
a little oil of cumin—Roman caraway. It is highly
improbable that such a liquid would have the therapeutic
effects claimed for it by the Metropolitan Pharmacal Company.
In view of the improbable claims made for Sinkina,
and the failure to substantiate them by suitable evidence,
it is recommended that the preparation be refused recognition
without at this time considering the claims made in
regard to the identity and amount of the drug claimed to
be the essential constituent.
In spite of its rejection Sinkina was persistently advertised.
It was thought advisable, therefore, to submit the
preparation to clinical tests. This was done and the results
are given in the following report:
THE CLINICAL REPORT
The following quotations indicate the claims made for this
preparation:
“In malarial conditions there is nothing that acts so promptly and
efficaciously as Sinkina. Sinkina destroys radically every trace of the
parasite in the blood from the time of its first appearance, builds up
the damaged corpuscles, revitalizes the system, and completely eliminates
every trace of the disease. Sinkina is deservedly termed the Specific
for Malaria.”
These claims were supported by testimonials which usually
gave no indication of a demonstration of the presence
or absence of malarial plasmodia in the blood. The following
is an example showing the character of most of the
evidence presented by the manufacturers:
“Three weeks ago I prescribed Sinkina for a negro man 40 years of
age suffering from a double tertian malarial infection having a chill
every afternoon for four consecutive days. He came to my office about
8 a. m. and was due to have a chill about 6 p. m. I gave him the
sample of Sinkina and directed him to take a tablespoonful at once,
also at noon and again at 4 p. m., and to continue taking it in same
size dose three times a day till he had taken it all. He reported to
me in a week from that date and told me he was feeling fine and that
he hadn’t had any more chills. The patient up to this time is apparently
cured.”
As the claims were supported by a few testimonials purporting
to be based on exact investigations, the Council
submitted the preparation to careful laboratory and clinical
tests. For this investigation the Council was fortunate in
securing the help of physicians actively engaged in the
study of malaria.
Experiments were made in vitro with the preparation; 1
ounce of Sinkina was used, and its action was compared
with that of 10 grains of quinin sulphate. When these were
added to cultures of malarial plasmodia in proportion corresponding
to 1 ounce of Sinkina or 10 grains of quinin
sulphate for a 150-pound man, the quinin was found to be
unfailingly antagonistic to the malarial organism, the drug
prevented the segmentation of the organism, and finally
killed it in about thirty-six hours. The Sinkina did not kill
the parasite after seventy-two hours of continued action,
and the parasites segmented in the presence of it just as
actively as they did in the control.
The investigator was furnished with two sets of preparations
in plain prescription bottles so as to avoid all influence
of the personal equation. One set consisted of Sinkina,
the other of a mixture of alcohol, sugar and water with
some oil of cumin. The investigator reported that, so
far as the tests on the cultures of malarial plasmodia were
concerned, he could not determine any difference in the
results obtained with the oil of cumin preparation, made
in the laboratory of the Association, and those obtained
with the Sinkina of the Metropolitan Pharmacal Company.
Clinical trials were made by three independent investigators.
Two of them received the two sets of preparations described.
FIRST INVESTIGATION
The first investigator treated two cases with Sinkina:
one was of the ordinary estivo-autumnal type and the other
an ordinary tertian.
Cases 1 and 2.—A good many schizonts were present in the
blood of each patient forty-eight hours after the administration
of Sinkina. In the instance of the case of tertian the
patient had his chill forty-eight hours after the medicine had
been started. As the Sinkina failed to produce any effect
the patients were then put on quinin to stop the disease.
Case 3.—The patient had taken 10 grains of quinin on the
day on which the experiment was begun. He had the tertian
form of the disease, and plasmodia were quite numerous
at the beginning. The quinin was discontinued and Sinkina
was given in doses of 1 ounce three times a day. The day
following the administration of 10 grains of quinin and 1
ounce of Sinkina, no parasites could be found in the blood.
The Sinkina was continued in the doses mentioned. On
the seventh day the patient had another chill, and a great
many parasites were found in his blood. The Sinkina was
discontinued and the patient was at once relieved by quinin.
This investigator gives it as his opinion, based on these
observations, that the preparation (Sinkina) is absolutely
worthless in the treatment of malaria, and he does not
think it necessary to make any further experiments with it.
SECOND INVESTIGATION
The second investigator treated two cases of tertian
malarial fever with these preparations until it was satisfactorily
proved that the drug was having no effect on the
presence of the parasites in the blood, when he began the
administration of quinin.
Case 4.—After the use of the remedies for one week the
investigator still found young rings half-grown and gametes
present in the blood. Apparently there was a relative
increase in the number of parasites. He then began the
administration of quinin. Blood-smears taken the next day
after 40 grains of quinin had been taken showed one parasite
after eighteen minutes’ search of one slide, and two
after thirty minutes’ search of a second slide. At the end
of a week’s treatment the patient was discharged recovered.
The blood examination of two slides was negative.
Case 5.—This was a case of tertian malaria. After treatment
for five days with Sinkina the blood still showed
tertian parasites with increase in the size of the spleen, and
the preparation was without effect on the clinical course
of the disease. Quinin was then begun, and the blood examination
became negative at the end of three days.
The investigator concludes that the preparations furnished
him were absolutely worthless in the treatment of
two cases of the tertian form of malarial fever, and that
these solutions had no effect on the presence of the parasites
in the peripheral circulation. In a case of quartan
malaria, both of the preparations (cumin oil mixture and
Sinkina), sent by the Association Laboratory, were without
effect on the plasmodia in the blood. This investigator
employed the solution made by the Association Laboratory
(cumin oil mixture) as well as Sinkina, and was unable to
note any differences between them.
THIRD INVESTIGATION
The third investigator began the trial of Sinkina at the
instance of the manufacturers, and used it in three cases,
two of them being benign tertian malaria and one case of
mixed infection (benign tertian and estivo-autumnal).
Case 6.—This was one of the cases of benign tertian
malaria. The patient gave a clinical history of malaria with
chills occurring on alternate days for a little over a week.
There was an immediate cessation of all clinical symptoms,
and three days after the patient had been on 1⁄2 ounce of
Sinkina three times daily there was no evidence of any
plasmodia in his blood; his additional treatment consisted
of 5 grains of calomel the evening of the first day with a
saline the next morning. Before the patient was put on
treatment, numerous parasites of both the asexual and sexual
forms were observed. The patient remained in bed for a
few days, and then returned to work. A week later he was
again taken ill with a return of all of his previous clinical
symptoms.
Case 7.—This case was one of mixed infection (benign
tertian and estivo-autumnal). The patient had a clinical
history of malaria dating back two weeks, with a maximum
temperature of 104 on admission. Tertian rings, estivo-autumnal
rings and crescents were found in the blood. The
patient was placed in bed, given thorough eliminating treatment,
and 1⁄2 ounce of Sinkina was administered four times
daily. His clinical symptoms ran on for two days with no
change, and there was no difficulty in finding the plasmodia
in blood-smears, which were taken twice daily. The dose
was then doubled and at the end of four days more there
was no change in either his clinical symptoms or the blood-findings.
The patient was then placed on 10 grains of
quinin sulphate with 15 drops of diluted hydrochloric acid
three times daily, to which he responded in less than forty-eight
hours and made an uneventful recovery.
Case 8.—This was the other case of benign tertian
malaria. The patient had chills every other day while
on the treatment, and laboratory diagnosis confirmed the
clinical findings. Experimental treatment was carried on
for four days, with a negative result.
The investigator calls attention to the fact that the first
case in which improvement resulted does not show any
necessary connection with the Sinkina administered, for
many cases of benign tertian malaria will clear up in just
as short a time under any line of treatment, while practically
all will eventually do so. This investigator later reported
another case and transmitted a clinical chart.
Case 9.—This patient was admitted to the hospital, Dec.
30, 1912, with a history of having had malaria for some
weeks. The diagnosis was confirmed by a blood examination.
He was then carried for four days without treatment
other than rest in bed and a liquid diet. His symptoms
subsided by the third day. On the fourth day a count of the
parasites was made which showed that there were 1,160
asexual parasites and 260 sexual forms to every thousand
leukocytes. The following day he was placed on Sinkina,
1 ounce three times daily. There was exacerbation of symptoms
on the following day, which gradually increased until
the fourth day, remaining about stationary for a day or so.
The fifth day after the patient had been placed on Sinkina,
another count of the parasites showed 5,600 asexual parasites
and 300 sexual forms to the thousand leukocytes, this
being an increase of 4,440 asexual forms and forty sexual
forms to every thousand leukocytes. With the second count
of parasites the dose of Sinkina was increased to 2 ounces
every four hours, the patient being kept on this until
January 14, without result. He was then placed on quinin,
with a complete reduction of the temperature to normal and
the disappearance of the parasites from the blood.
The investigator also reported a case of benign tertian
malaria.
Case 10.—This was in a child of 8 years which was
treated by the investigator’s confrère and gave similar
negative results. Blood examination showed numerous parasites.
The child was placed on 1 ounce of Sinkina three
times a day and kept on it for two weeks. The clinical
picture remained unaltered, and parasites could be detected
in numbers whenever examinations were conducted. A gradually
increasing enlargement of the spleen was also noted.
At the end of two weeks quinin was substituted, and the
child went on to a rapid and uneventful recovery.
This investigator also concludes that the claim put forth
by the Metropolitan Pharmacal Company that Sinkina is a
specific in the treatment of the malarial fevers is entirely
without foundation, and that the firm will be unable to
demonstrate to the contrary.
These investigations demonstrate that Sinkina is not a
specific against malaria, and that it has no more effect than
a mixture of oil of cumin, sugar, alcohol and water. They
further show the fallacy, first, of concluding from a temporary
cessation of the symptoms in malaria that the disease
has been cured and, second, of ascribing such temporary
improvement to the influence of a remedy which has no
known effect on the malarial organism.—(From the Journal
A. M. A., Sept. 27, 1913.)
SOMNOS
Report of the Council on Pharmacy and Chemistry
To the Council on Pharmacy and Chemistry of the American
Medical Association:—Your subcommittee, to whom was
assigned Somnos, H. K. Mulford Company, submits the
following report of experiments, undertaken to compare
the effects of Somnos with those of chloral hydrate. These
experiments demonstrate that the statements made in regard
to the action of Somnos are in conflict with Rule 6 of the
Council, which requires: “No article will be admitted or
retained concerning which the manufacturer or his agents
make unwarranted, exaggerated or misleading statements
as to the therapeutic value.” It is, therefore, recommended
that Somnos be not approved for inclusion in the book until
the claims made for it are corrected. It is also recommended
that the report be published:
The Pharmacology of Somnos
When these experiments were begun, April, 1906, there
was nothing in the advertising literature on Somnos to
indicate whether this article is a solution or a pure substance.
On the label on the bottle, in the circular accompanying
the bottle, and in the booklet “Somnos,” the word
Somnos seemed to be used as a synonym of “Chorethanal
alcoholate,” C9H11O5Cl9, and physicians were prescribing and
pharmacists dispensing it in the belief that it was a pure
substance. “The pure substance; some kind of an alcohol;
nothing to do with choral,” was the way the druggist from
whom the samples were purchased put it. Thus information
absolutely indispensable for any rational comparison of
Somnos with other hypnotics was withheld from the physician.86
Hence, before beginning the physiologic experiments it
was necessary to determine the strength of the preparation;
for this purpose three chlorin determinations (by the Carius
method) were made. On the assumption that all the
chlorin present was in combination as chloral glycerate,
C3H5[CCl3.C(OH)2]3 = C9H11O6Cl9, and calculating the percentage
of this in Somnos, the following results were
obtained: (1) 5.11 per cent.; (2) 5.15 per cent.; (3) 5.10
per cent.
Somnos, therefore, was found to contain approximately
5 per cent. of chloral glycerate and its physiologic action
was compared with that of a 5 per cent. solution of hydrated
chloral. In some experiments the hydrated chloral was
dissolved in water; in others, in 10 per cent. alcohol
(Somnos was found to contain at this time about this percentage
of alcohol); in other experiments glycerin was
added, as Somnos was found to contain this substance.
No very marked differences were found in the physiologic
action of the three solutions.
FATAL DOSE OF SOMNOS FOR THE LOWER ANIMALS
The booklet on Somnos states that “Somnos has no
toxicology”; that while chloral hydrate causes “acute
poisoning,” “deep coma,” etc., Somnos is “harmless in twenty
times the dose prescribed,” “coma unknown, etc.” The
physician would scarcely suspect from such statements that
Somnos is as poisonous a substance as solutions containing
hydrated chloral in corresponding amount; that such is the
case is shown by the following experiments. These experiments
were necessarily made on the lower animals. While
such results do not enable us to draw very definite conclusions
as to the absolute toxicity of poison for man,
the results on animals are conclusive as regards the relative
toxicity for man of such closely related drugs as hydrated
chloral and Somnos.87
*****
CONCLUSIONS
To sum up our results on the physiologic action of Somnos:
We have been completely unable to verify the claims of the
manufacturers that Somnos is less toxic than hydrated chloral,
or that it has a less depressing effect on temperature, respiration
or circulation. On the contrary, the physiologic effects
are indistinguishable from those of hydrated chloral, doubtless
because the action of Somnos is simply the action of
hydrated chloral. We can see nothing in the animal experiments
or in the chemical composition which would suggest
that Somnos would possess therapeutic advantages over an
elixir of hydrated chloral of corresponding strength.88
It is to be hoped that physicians who have been blindly
using Somnos without even knowing the strength of the
preparation, much less what it is, will compare its effects
with those of a 5 per cent. elixir of hydrated chloral.88—(Abbreviated
from The Journal A. M. A., Sept. 15, 1906.)
SUCCUS ALTERANS
Report of the Council on Pharmacy and Chemistry
The following report was adopted by the Council:
It is, believed that unwarranted and exaggerated therapeutic
claims are made for Succus Alterans by its manufacturers,
Eli Lilly & Co., Indianapolis. In view of the
disastrous results which may follow, if, from the statements
made, physicians should be led to rely on the product as
a treatment for syphilis, it is recommended that Succus
Alterans be refused recognition and that this fact be published
with comments.
W. A. Puckner, Secretary.
Comment: Succus Alterans is a preparation which has
been put on the market for some years by Eli Lilly & Co.,
as a remedy for syphilis. The serious character of this
disease and especially the deplorable results that ensue
from its improper or insufficient treatment, should make a
firm hesitate to advise any treatment for it which experience
has not demonstrated to be at least as efficacious as that
which is generally accepted and well proved. Succus
Alterans is the result of a combination of circumstances; no
one person is responsible for it. It was probably the natural
desire for a remedy free from the occasional injurious
results of mercury that led Dr. J. Marion Sims to advocate
the use of a collection of indigenous American plant drugs,
sarsaparilla, stillingia, xanthoxylum, etc., which had a local
reputation for the cure of syphilis. These drugs are supposed
to be inert when the dried plants were used, and this
gave an opportunity for the development of a nostrum. The
ingredients are well known, but as their virtues are supposed
to be lost in drying, the physician can not have his druggist
compound them, but must, perforce, prescribe the proprietary
combination.
Those who consented to experiment with the new remedy
soon found that the claims to curative properties were
unfounded, but the strong commercial interests backing it
have prolonged its life to the present time. Authorities on
syphilis either say nothing about the preparation or mention
it merely to condemn; but the proprietors of the nostrum
continue to assert that it is not only practically a specific
in syphilis, but now recommend it for various derangements
of the blood and all sorts of skin diseases.
This being the case, what shall the wise physician do?
Shall he blindly follow an authority of a past generation or
shall he recognize that the claims of an interested manufacturer
ought not to weigh against the consensus of his
present-day confrères who have given the treatment of
syphilis their special attention? The exploitation of such a
preparation is deserving of strong censure. By such methods
the firm places itself on the same plane as those nostrum
venders, who advertise certain antiseptic sprays and gargles
as cures for epidemic meningitis and diphtheria and thereby
deprive credulous victims of the curative antitoxin treatment.
Succus Alterans is not a new remedy on trial for
its possibilities of improvement in therapeutics; it is an
old mixture which has been tried and found wanting.—(From
the Journal A. M. A., June 26, 1909.)
SULPHO-LYTHIN
Report of the Council on Pharmacy and Chemistry
Sulpho-Lythin is sold by the Laine Chemical Company,
New York. In the literature sent to physicians it is said:
“This product, the sulpho-phosphite of sodium and lithium
(non-effervescent), is entirely new and is unique in its
action.”
Chemical analysis of a specimen of Sulpho-Lythin purchased
in the open market indicated its composition to be:
Sodium sulphate, anhydrous | 10.51 |
Disodium hydrogen phosphate, anhydrous | 56.67 |
Sodium thiosulphate, anhydrous | 20.78 |
Sodium chlorid | 5.98 |
Lithium, as citrate | 3.12 |
Sulphur, free | 0.16 |
Moisture | 1.53 |
Loss | 1.25 |
The examination, therefore, shows that Sulpho-Lythin is
a mixture consisting mainly of sodium sulphate and sodium
phosphate and sodium thiosulphate. The statement that it
is a “sulpho-phosphite of sodium and lithium,” therefore,
is not correct, and a statement that “it is entirely new and
unique in its action” appears unwarranted and misleading.
It is, therefore, recommended that the preparation be refused
recognition. It is also recommended that an article be prepared
for publication calling attention to the exaggerated
claims made for Sulpho-Lythin.
The recommendations of the subcommittee were adopted
by the Council and in accordance therewith the report is
published with comments, substantially as follows: The
formula means that it is a solution of well-known salts,
some of them under partially disguised names. Every one
knows what Glauber’s salts are good for. Disodium hydrogen
phosphate is ordinary common sodium phosphate. Sodium
thiosulphate is familiar as sodium hyposulphite, the “hypo”
of the photographers. Every one knows, of course, that
sodium chlorid is common salt. Examination and analysis
of various specimens of this product demonstrated that its
composition is not always the same. As an indication of the
ignorance of the promoters of this nostrum it is interesting to
note that the label on one of the bottles purchased states that
it is a “sulphophosphate” instead of a sulphophosphite. Extravagant
claims are made for this simple mixture of laxative
salts, and these with the methods of using it are printed on
the labels, and while it is claimed to be only advertised to
the profession, the physician is repeatedly advised in the
advertisements to “order always an original (six ounce)
bottle to prevent substitution.” The natural result of this
would be, of course, to put the patient in the way of prescribing
it for himself and to spread the advertisement of
the drug among the public. Difficulty has been experienced
in finding out who the promoters of this nostrum are and
the correspondence in regard to it is published. They seem
to prefer to be known by their corporate title of Laine
Chemical Company only. It is a sample of many other
so-called ethical proprietary drugs, most of which are simple
mixtures of well-known drugs which physicians are using
every day and which require no skill in their compounding.
Their proprietors not only presume to sell and advertise
medicines but also to tell the physicians how to treat their
patients.—(Abstracted from The Journal A. M. A., Dec.
8, 1906.)
TAUROCOL
Report of the Council on Pharmacy and Chemistry
The Paul Plessner Company, Detroit, places on the market
Taurocol Tablets and Taurocol Compound Tablets. The
company makes a pretense of giving the formula—minus any
quantities—thus:
“Taurocol is a combination of bile salts, extracts of cascara sagrada,
phenolphthalein and aromatics.”
The “formula” given for Taurocol Compound Tablets is:
“Taurocol (Bile Salts) | Gramme | .1296 |
Pepsin 1-3000 | " | .0324 |
Pancreatic Ext | " | .0324 |
Extract Nux Vomica (1⁄8 gr.) | " | .0081 |
Aromatics | Q. S.” |
A comparison of these two “formulas” with those furnished
for Veracolate and Veracolate with Pancreatin and
Pepsin shows that they are nearly the same.
The claims made for the Taurocol preparations are essentially
those made for Veracolate preparations, as instance
the following, which appears on a physician’s sample of
Taurocol:
“For Hepatic Insufficiency, Intestinal Putrefaction, Habitual Constipation.”
Likewise the following, found on a Taurocol circular,
duplicates claims made for Veracolate:
“... Directly stimulates the liver cells, producing an abundant
flow of bile rich in cholates, solvent of cholesterin and a biliary antiseptic.”
Taurocol is objectionable for the reasons that apply to
Veracolate, and Taurocol Compound Tablets are subject to
the objections that apply to Veracolate with Pepsin and
Pancreatin. (See p. 216.) The Council therefore refused
recognition to Taurocol and its preparations.—(From The
Journal A. M. A., April 24, 1915.)
TRI-IODIDES, THREE CHLORIDES AND MAIZO-LITHIUM
Report of the Council on Pharmacy and Chemistry
As an illustration of unreliability of claims and the unscientific
character of proprietary mixtures, the Council has
authorized publication of the following reports on Tri-Iodides,
Three Chlorides and Maizo-Lithium, products of the Henry
Pharmacal Co. (J. F. Ballard, proprietor).
W. A. Puckner, Secretary.
Tri-Iodides (Henry)
Tri-Iodides (Henry Pharmacal Co., St. Louis) is a nostrum
whose ingredients apparently were selected at random.
Since the effects of such a mixture cannot be predicted, no
thoughtful physician would think of prescribing in any one
condition all the drugs named in the formula of Tri-Iodides—if
he had to write out the prescription. Yet because the
misleading name of the preparation gives it the semblance of
a therapeutic entity—and because it is advertised in medical
journals—a certain number of physicians thoughtlessly prescribe
this shotgun mixture.
LABORATORY REPORT
Regarding the composition of “Tri-Iodides” the Association’s
Chemical Laboratory makes the following report:
A trade package of Henry’s Tri-Iodides purchased in
1910 bore the following formula on the label:
“Colchicin, 1-20 grain,
“Phytolaccin, 1-10 grain,
“Solanin, 1-3 grain,
“Sodium Salicylate, C. P., 10 grains,
“Iodic Acid (equal to 7⁄32 gr. of Iodine)
in two fluid drachms
of Aromatic Cordial.”
In the circular which was wrapped with the bottle
the wording of the formula differs somewhat from the
foregoing, “iodic acid” of the label being replaced by
“hydro-iodic acid.” While the label on the bottle named
“phytolaccin” as one of the constituents the label on the
carton which contained the bottle gave “decandrin.” The
following formula appears on a trade package purchased
June, 1914:
“Colchicine, 1-200 Grain,
“Phytolacca, 1 1-5 Grain,
“Mydriatic Alkaloids, 1-500 Grain.
“Sodium Salicylate, 3 1-2 Grain.
“Iodic Acid (equal to 7-125 Grain of Iodine)
in two
fluid drachms.”
The differences between the formulas are striking.
Colchicin has been reduced from 1⁄20 grain to 1⁄200 grain;
sodium salicylate from 10 grains to 31⁄2 grains; iodin
(claimed to be present as iodic acid) from 7⁄32 grain
to 7⁄125 grain. “Phytolaccin” (“Decandrin”) has been
replaced by “Phytolacca” and “Solanin” by “Mydriatic
Alkaloids.” While the formula for the preparation has
been changed, the circular accompanying the package still
refers to “solanin” (in some parts of the circular wrongly
spelled “salonin”) and “phytolaccin.” As no principle
having the characteristic effects of poke-root is known
to have been isolated the terms “decandrin” and “phytolaccin”
are meaningless.
The circular states that solanin is an alkaloid obtained
from the sprouts of Solanum tuberosum, but wrongly calls
this plant “bittersweet” instead of potato. At the market
price the amount of solanin claimed, according to the
old formula, to be present in a bottle of Tri-Iodides,
would cost $1.60, although a bottle of the preparation sold
at wholesale for 67 cents.
Tri-Iodides is a dark brown, mobile liquid having a
faint clove-like odor and a mawkish, sweet taste. Salicylate
was found in considerable amounts. Traces of
alkaloids were found, a portion of which appeared to be
colchicin. Iodic acid and its salts were absent, although
claimed by the formula to be present. Potassium iodid
was present. Determinations of the iodin by distillation
with ferric ammonium sulphate solution and sulphuric
acid indicated the presence of about 1.68 gm. of iodin
(equivalent to 2.18 gm. of potassium iodid) in each 100 c.c.
of the preparation. This is equivalent to about 7.65 grains
of iodin per fluidounce, or more than thirty-four times
the amount claimed by the formula on the bottle. An
approximate determination of the salicylic acid by extraction
of the acidified preparation with ether and evaporation
of the solvent indicated about 2.67 gm. in 100 c.c., equivalent
to 3.09 gm. of sodium salicylate, or about 14.11 grains
per fluidounce. Since the amount of sodium salicylate
claimed is 3.5 grains in 2 fluidrams or 14 grains in each
fluidounce, the amount found agrees essentially with the
claims.
ABSURD CLAIMS
It should be unnecessary, after pointing out the conflict
between the name and the published formula, between the
formula and the actual composition, and between the composition
and all established therapy, to discuss this heterogeneous
and unscientific mixture further. A few specimen
absurdities, however, may be quoted from the advertising
“literature”:
“... Free of the Disagreeable Effects of the Alkaline Iodides.”
[Tri-Iodides, according to the laboratory report, depends
for its iodin action on potassium iodid.]
“... we have an assimilable form of vegetable hydriodates.
“The hydriodates of these valuable vegetable alkaloids afford the
specific alterative action of iodine without such disagreeable results
as the iodism produced by the ordinary iodides.”
[“The hydriodates” is an obsolete term formerly applied
to iodids of vegetable alkaloids. Iodids of vegetable alkaloids,
if present at all in Tri-Iodides, are present in negligible
amounts.]
“Containing Iodine in an available form, it is obvious that the formula
must be beneficial in the majority of syphilitic skin lesions.”
The falsity of the first two of these claims and the mischievousness
of the last are self-evident.
It would be possible, but is unnecessary, to produce an
almost unlimited amount of evidence to show the transparent
character of the deception by which this preparation is
exploited.
The referee feels that the nostrum will have been sufficiently
characterized when he has mentioned further that the
name “Henry’s Tri-Iodides” is blown in the glass of the
bottle, that the label contains the recommendation “For Gout,
Rheumatism and other Diathetic Diseases,” and that the circular
accompanying the bottle recommends the use not only
of Tri-Iodides, but also of Three Chlorides, Maizo-Lithium,
Campho-Phenique and Satyria in the treatment of many
diseases.
Three Chlorides (Henry)
Three Chlorides (Henry) is advertised as:
“An oxygen-carrying ferruginous preparation, suitable for prolonged
treatment of children, adults and the aged. Indicated in anemia and
convalescence from acute diseases and surgical operations.”
The following report on the composition of Three Chlorides
is submitted by the Association’s Chemical Laboratory:
LABORATORY REPORT
It is claimed that each fluidram of Henry’s Three
Chlorides contains:
“Mercuric Bichlorid | 1-72 Gr. |
“Arsenic Chloride | 1-40 Gr. |
“Proto-Chloride Iron | 2-25 Gr. |
| “... in a cordial of Calisaya Alkaloids.” |
The preparation is a pale yellow, clear solution having
an odor of alcohol. The addition of potassium ferricyanid
solution does not produce any blue coloration, thus demonstrating
the absence of ferrous chlorid (iron protochlorid).
Instead potassium ferrocyanid solution produces
at once an intense blue precipitate and potassium
sulphocyanate solution an intense red coloration, thus
proving the presence of iron in the ferric condition. It
is obvious that the claimed superiority of Three Chlorides
over preparations containing ferric iron is absurd. Since
it contains iron in the ferric condition, Three Chlorides
decomposes soluble iodids with the liberation of free iodin.
The assertion that it is a suitable “vehicle” for the administration
of iodids is likely to lead the physician unwittingly
to administer free iodin.
As the laboratory report shows, the “formula” of Three
Chlorides (Henry) is incorrect, for protochlorid of iron
(ferrous chlorid) was absent from the preparation. There
is, however, a more serious objection to the formula than the
misstatement of fact. When the physician is dealing with
conditions that call for mercury, arsenic or iron, it is irrational
and unscientific to prescribe a preparation containing
these three drugs in fixed proportions.
OBJECTIONABLE ADVERTISING
Three Chlorides is marketed in bottles having the name
“Three Chlorides” blown in the glass, in a carton containing
a circular extolling the curative powers of this and
other proprietaries of the same concern. Thus a physician
who prescribes Three Chlorides is likely to place in the
hands of his patient the advice that
“Three Chlorides ... is suitable for the prolonged treatment of
children ...”
“In tertiary syphilis, with or without potassium iodide, it holds first
rank among remedies directed against the specific taint ...”
Further, that “Maizo-Lithium” is:
“A Genito-Urinary Sedative” and a “remarkable uric-acid solvent.”
Also that “Satyria” is:
“An Ideal Genito Tonic and Nerve Reconstituent.”
“Indicated in Prostatic trouble, Cystitis, Urethritis, Gonorrhea, Gleet,
Leucorrhea, Sexual Debility and Impotence.”
We are told that
“As a hematinic, the protochloride of iron justifies the confidence of
the medical profession.”
“The protochloride, more than any other salt of iron, stimulates the
paptic [sic] and hydrochloric glandular system of the stomach, increasing
the flow of acid gastric juice.”
It is unnecessary to discuss the truth or falsity of these
assertions, since Three Chlorides does not contain the protochlorid
of iron. For the same reason, it is obvious that the
small amount of iron which it contains is the only possible
justification for the claim that the preparation is
“... Non-Productive of ... Constipation or Teeth Discoloration.”
It is hardly necessary to point out that it is a therapeutic
exaggeration to claim that Three Chlorides is of particular
value in the treatment of tertiary syphilis, that in eczema it
is “the most effective remedy,” that in any form of constipation
it is “the remedy par excellence,” or that
“After arresting malarial attacks with quinine, the combination of iron,
arsenic and mercury with calisaya is an essential requisite.”
“Whenever gastric troubles and digestive disturbances furnish a
contra-indication to iron, this contra-indication disappears when the iron
is combined with arsenic.”
“The simultaneous exhibition of small doses of arsenic and bichloride
of mercury, besides augmenting the effect of iron upon the red blood-cells,
completely obviates the tendency to vascular congestion and hemorrhage.”
Finally, the suggestion that by the use of Three Chlorides
iodids may be prevented from causing iodism is absurd.
In short, whatever may be the advisability of prescribing
iron, arsenic or mercury in any given case, it is irrational
to prescribe them in fixed proportions. A physician who is
induced by the exaggerated advertising claims to prescribe
these drugs in a proprietary mixture, under a non-informative
name, does grave injustice to his patients.
Maizo-Lithium
Maizo-Lithium (Henry Pharmacal Co., St. Louis) is one
of the many proprietary lithium preparations based on the
disproved theory that lithium dissolves uric acid deposits
in the body. The label on a trade package states that:
“Maizo-Lithium promptly facilitates the elimination of the uric and
phosphatic deposits from the system.”
As might be expected, the promoter of Maizo-Lithium
ascribes a long list of ills to “uric and phosphatic deposits,”
and argues that, therefore, Maizo-Lithium is the proper treatment:
“In lithemia, hematuria, incipient diabetes, cystitis, urethritis, pyelitis
and ALL inflamed conditions requiring a non-irritating diuretic.”
“Inflamed conditions,” naturally, include almost all of the
real or imaginary ills of kidney, bladder, etc.
Maizo-Lithium is distinguished from its congeners chiefly
by the claim that it contains a mythical or problematical
compound, maizenate of lithium.
LABORATORY REPORT
The following report on the composition of Maizo-Lithium
has been submitted by the Chemical Laboratory of the American
Medical Association:
The promoter of Maizo-Lithium makes the following
statement on the label concerning the composition of the
preparation:
“Each fluid drachm contains two grains maizenate of lithium.”
The following is also found in a circular which is
enclosed with the trade package of Maizo-Lithium:
“Maizo-Lithium, the remarkable uric acid solvent, is a nascent
chemic union of maizenic acid, obtained from green corn silk,
with the alkaline base lithium forming maizenate lithium, of
which the mother liquid carries two grains to each drachm.”
Standard works on organic chemistry and pharmacology,
such as Beilstein’s Organische Chemie and Cushny’s
Pharmacology and Therapeutics, do not mention maizenic
acid. Neither is it mentioned in comprehensive bibliographies
of phyto-chemical investigations, such as Huseman-Hilger’s
Die Pflanzenstoffe or Wehmer’s Die Pflanzenstoffe.
The first to use the term appears to have been a
Dr. Vautier (Arch. méd. belg.), but his publication is not
available to the laboratory. Rademacher and Fischer
(Amer. Jour. Pharm., 1886, lviii, 369) claim to have isolated
the substance from green corn-silk, but the record of their
work is unsatisfactory and indefinite and therefore their
results could not be verified; it seems unlikely, however,
that they isolated a pure proximate principle.
Examination of Maizo-Lithium demonstrated the absence
of bromids, chlorids, phosphates, sulphates, acetates, benzoates,
salicylates and tartrates—combinations in which
lithium might be expected to be present. The presence of
a citrate, however, was shown by the usual tests. Lithium
and sodium were present. Free acid was absent. Determination
of lithium citrate and of sodium citrate indicated
the presence of a total of about 3.7 gm. of these two salts
in each 100 c.c. of the preparation, or about 2.1 grains in
each fluidram. About 25 per cent. of the total salts
appeared to be lithium citrate. The examination, therefore,
does not demonstrate the presence of “maizenate of
lithium,” but does show that Maizo-Lithium contains a
mixture of lithium citrate and sodium citrate. Tests for
citric acid and citrates were made on a commercial specimen
of fluidextract of corn-silk. The results were negative,
although the preparation had an acid reaction to
litmus. The presence of maizenate of lithium in Maizo-Lithium—in
fact, its actual existence—thus failed of demonstration.
In view of this fact, it was felt that the
burden of proof rested on the promoter of Maizo-Lithium
to supply some satisfactory evidence with regard to this
substance. The following letter was therefore, sent to
James F. Ballard:
“According to the label on a recently purchased bottle of Maizo-Lithium,
each fluidram of this preparation contains 2 grains of
‘maizenate of lithium.’ From an examination made in this laboratory
we are inclined to conclude that this statement is not in accordance
with the facts. A search of chemical and pharmaceutical publications
does not reveal that such a compound as ‘maizenate of lithium’ has
ever been isolated and described, and we are very much inclined to
question its existence. We should be pleased to receive from you
any evidence which you may care to send in substantiation of your
claim in regard to the content of ‘maizenate of lithium’ in Maizo-Lithium—particularly
a specimen of ‘maizenate of lithium’ or the
method by which it is produced.”
While this letter was sent Oct. 13, 1914, no evidence has
been submitted up to date (January, 1915) to substantiate
the asserted presence of maizenate of lithium in Maizo-Lithium.
The report just given shows that the manufacturer has
found it expedient to surround his worthless nostrum with a
cloak of mystery. A discussion of the jumble of uncritical
claims, baseless assertions and evident falsehoods presented
in favor of Maizo-Lithium would seem a waste of time when
the secrecy of this nostrum is all-sufficient for its condemnation.
[Editorial Note.—When the Council on Pharmacy and
Chemistry was started we announced that we did not see any
clear line of demarcation between “patent medicines” and
many so-called “ethical proprietaries.” Time has not caused
us to change our opinion. As we have already shown, and
as we shall have occasion to show in the future, not a few
of the “ethical proprietaries” offered to physicians are
being advertised by those who are pushing the rankest of
“patent medicines.” The three preparations mentioned
above are sold—and presumably manufactured—by Mr.
Ballard, of St. Louis. Mr. Ballard is the promoter of Ballard’s
Snow Liniment, Brown’s Iron Bitters, Herbine, Dr.
Herrick’s Vegetable Liver Pills, Swaim’s Panacea, Renne’s
Pain Killing Oil, etc. He is also the promoter of Campho-Phenique,
exposed in The Journal some eight years ago.89
The spectacle is not an edifying one. A manufacturer with
one hand offers the public a profusion of cure-alls, while
with the other he endeavors to foist on the medical profession
preparations which are just as fraudulent. Some day
our profession will awake to the disgrace of it all. It will
also awake to the fact, which should have been evident ere
this, that the nostrum business would cease if physicians
would refuse to accept into their offices, even as a gift, the
nostrum-promoting medical journals that live off this trade.
Fraudulent “patent medicines” will continue to thrive just so
long as newspapers will publish “patent medicine” advertisements;
fraudulent “ethical proprietaries” will continue to
exist just so long as medical journals will advertise such
proprietaries. As the better class of newspapers are rejecting
“patent medicine” advertising on their own volition, so are
the better class of medical journals rejecting advertisements
of fraudulent proprietaries. Some newspapers will continue
to carry nostrum advertising until their subscribers raise a
protest that will cause the business department to take notice;
so, too, some medical journals will continue to share the
profits with the nostrum exploiters until an outraged medical
profession repudiates such publications.]—(From The Journal
A. M. A., Feb. 6, 1915.)
THIALION
Report of the Council on Pharmacy and Chemistry
The following report was submitted to the Council by a
subcommittee which examined Thialion (Vass Chemical
Company):
To the Council on Pharmacy and Chemistry:—We beg leave
to report on Thialion as follows:
Thialion is sold by the Vass Chemical Co., Danbury,
Conn. In the literature supplied to physicians and in the
advertisements in medical journals, Thialion is stated to be
“a laxative salt of lithia” with the chemical formula
“3Li2O.NaO.SO3.7HO.” “Sodio-trilithic anhydrosulphate” is
given as a synonym. An elaborate graphic or structural
formula is also given.
According to analyses, this preparation is a mixture consisting
chiefly of sodium sulphate and sodium citrate with
very small amounts of lithium, the average of several estimations
indicating the following composition:
Sodium citrate | 58.6 |
Sodium sulphate, anhydrous | 26.6 |
Sodium chlorid | 3.3 |
Lithium citrate, anhydrous | 1.8 |
Water | 9.7 |
Thus, the advertising literature is a deliberate misrepresentation
of the facts. It is, therefore, recommended that
the preparation be refused recognition, and that this report
be published.
The recommendations of the subcommittee were adopted
by the Council and in accordance therewith the above report
is published.
W. A. Puckner, Secretary.
In publishing the above report, the Council is presenting
to the medical profession another object lesson, and one that
illustrates how easily our profession is being humbugged.
There are several things that we may learn from the report
on this nostrum, but at this time we will take up only one
phase of the lesson. Many of the scientific chemical compounds
and derivatives given us by the German chemists
have been distinct advancements and have proved to be
valuable additions to our therapeutic agents; further, they
were received with so much favor by physicians that they
have been profitable for those who made them. It is not
strange, therefore, that imitators should appear. One of the
first was our old friend, Antikamnia (which was introduced
as a “new synthetical” compound). This was followed by
Ammonol, Phenalgin, Salacetin, and a host of others having
acetanilid as their principal ingredient.
This picturesque “graphic formula” for Thialion appears with many
of the advertisements. To most of us it looks formidable, wonderfully
and deeply scientific and non-understandable; to a chemist it looks
absurd.
But there are hundreds of other so-called “new chemical”
compounds among the “ethical” proprietaries on the market
aside from the acetanilid mixtures. These wonderful compounds,
by the mysterious union of their ingredients, possess
therapeutic properties different from, or more powerful for
good than the drugs from which they are made. At least,
this is what we are told, and this is what many believe or
they would not sell so well.
There is another factor worth noticing connected with this
subject: When to the claim that the mixture is a “chemical
compound” is added a complex chemical formula, it prevents
the impertinent question, “What is it?” For isn’t the
“formula” there, and is not the information given without
the asking? Most of us have been so overcome by the
display of the chemical knowledge of the nostrum maker
that we have been afraid to expose our ignorance by asking
for information or explanation. And thus the promoter
avoids perplexing questions, which, if answered truthfully,
would spell bankruptcy.
To a chemist the formula of Thialion furnished by the
Vass Chemical Company signifies nothing. To a physician
who possesses but little knowledge of chemistry, it will
seem impressive, and he may absorb the idea that it stands
for a preparation that is the result of exhaustive scientific
research. To the chemist, this formula will appear as a
jumble of symbols and numbers that mean nothing.
It is not worth while to call attention to the simplicity of
this simple mixture of ordinary salts, for it is too self-evident.
As to the remarkable therapeutic qualities of
Thialion, the reader is referred to that ably edited “scientific”
periodical, the Uric Acid Monthly, and to the mass of “literature”
relating to this wonderful remedy.
While there is a ridiculous side to this business, there is
also a serious one. Those who have been making money
out of us undoubtedly laugh in their sleeves at our gullibility,
but to us as members of a presumably learned and
intelligent profession, it is not a laughing matter. The
whole nostrum business is a shame and a disgrace.—(Modified
from The Journal A. M. A., Nov. 3, 1906.)
UNGUENTUM SELENIO VANADIC (V. ROEMER)
Report of the Council on Pharmacy and Chemistry
Unguentum Selenio Vanadic (v. Roemer) is an ointment
manufactured by A. von Roemer, Brooklyn, N. Y., and put
on the market by Schering and Glatz, New York. It is claimed
to contain 1 per cent. of selenium oxycyanid and 1 per cent.
of vanadium chlorid “so prepared and incorporated into a
modified lanolin base as to insure complete absorption.” The
preparation is recommended in the later stages of inoperable
carcinoma, sarcoma, epithelioma and other malignant tumors,
as a substitute for morphin and other narcotics to control
pain, as a modifying (ante-operative) treatment in the middle
stage of malignant cases presenting the characteristics of
being inoperable, and as a prophylactic treatment of recurrences
and metastases following excision of malignant tumors.
It is also recommended for use in slow-healing surgical
wounds, abscesses, tuberculous and mixed septic and gangrenous
processes, etc., in lupus, acne, eczema, psoriasis,
scabies, erythemata, adenomata, angiomata, papillomata, etc.
The use of the ointment is further recommended by systemic
inunction in septicemia, pneumonia, erysipelas, cerebrospinal
meningitis, septic rheumatism, septic neuritis, etc. The
Council voted that the preparation be not accepted for inclusion
with New and Nonofficial Remedies because no evidence
has been submitted that the vanadium and selenium are
absorbed or that they produce any of the effects claimed.
When the preceding report was sent to Schering and
Glatz, the firm expressed surprise that evidence of the absorption
of selenium and vanadium should be requested. On
June 8 the firm wrote that within a few days one or more
tests would be sent by which the presence of selenium and
vanadium in the urine could be demonstrated. These tests
were not received. So far (November, 1914) no evidence of the
value of the preparation either in carcinoma or in any of the
very long list of other diseases in which it is recommended
has been submitted, and, the pharmacologic evidence that
such a preparation would be of value in such conditions being
practically nil, the Council authorized publication of this
report.—(From The Journal A. M. A., Nov. 21, 1914.)
UNICORN ROOT, WILD YAM AND WILD INDIGO
Report of the Council on Pharmacy and Chemistry
The Council has voted that recognition be refused to the
following: Unicorn Root (Aletris farinosa), Wild Yam
(Dioscorea villosa), and Wild Indigo (Baptisia tinctoria)
and has authorized the publication of the following statements.
W. A. Puckner, Secretary.
Unicorn Root—Aletris Farinosa
Unicorn Root (Aletris farinosa) contains a bitter principle
and starch. Remarkable powers as a uterine tonic
have been ascribed to it but have not been realized by
reliable observers, the drug being practically valueless in
these conditions. It enters into the composition of a number
of nostrums. As a bitter it is superfluous and it should
not be included among non-official drugs.
Wild Yam—Dioscorea Villosa
Wild Yam (Dioscorea villosa) has been little used in
medicine. It contains a saponin and an acrid resin, and is
said to possess expectorant, diaphoretic and—in large doses—emetic
properties. It has been recommended as a remedy
in biliary colic and in muscular rheumatism. Its value in
such conditions has not been verified to an extent entitling
it to consideration as a useful remedy.
Wild Indigo—Baptisia Tinctoria
Wild Indigo (Baptisia tinctoria) has been in use—chiefly
by the eclectics—for about three-quarters of a century, but
there is no satisfactory evidence that it has any therapeutic
value. The following text-books on pharmacology do not
even mention wild indigo: Cushny, Brunton, Dixon, Binz,
Sollmann. It is not official in the United States or other
leading pharmacopeias.
A preparation of wild indigo is advertised with extravagant
claims for its therapeutic action, but these claims are
not supported by any substantial evidence. Other virtues
ascribed to wild indigo are its properties as a cardiac and
hepatic stimulant and its value in sepsis, particularly in
typhoid fever. It actually has emetic and cathartic properties,
but even these are inferior to those possessed by
many other drugs.
It is very evident that a drug possessing the extraordinary
merits that have been claimed for wild indigo would
not have remained unnoticed by the leading authorities on
pharmacology and therapeutics, especially after its prolonged
use in medicine. Owing, therefore, to the lack of
substantial evidence of its usefulness, baptisia is not considered
as of sufficient importance to warrant its inclusion
in the list of non-official drugs. It is probably entirely
superfluous.—(From The Journal A. M. A., Jan. 22, 1910.)
PROPRIETARY VANADIUM PREPARATIONS
Report of the Council on Pharmacy and Chemistry on
Products of Vanadium Chemical Co.: Vanadiol,
Vanadioseptol, Phospho-Vanadiol,
Vanadoforme, etc.
Vanadiol and preparations thereof, the products of the
Vanadium Chemical Company, were submitted to the Council.
After thorough investigation it was concluded that the
company has not, and never has had, any reliable evidence
for the therapeutic claims it has presented to the medical
profession regarding these products. Accordingly the Council
voted that the several products under consideration be
not accepted for inclusion with New and Nonofficial Remedies.
The findings of the Council having been submitted
to the Vanadium Chemical Company and its reply considered,
the Council authorized publication of the report which
appears below.
W. A. Puckner, Secretary.
The Vanadium Chemical Company, Pittsburgh, Pa., submitted
to the Council on Pharmacy and Chemistry for
inclusion in New and Nonofficial Remedies the following
products: Vanadiol, Vanadioseptol, Phospho-Vanadiol,
Vanadium Solution for Intravenous and Hypodermic Use
and Vanadoforme. At the same time, the company submitted
statements and “literature” regarding the composition
and therapeutic value of these products. The committee
to which the matter was referred, after carefully considering
both the matter presented and certain modifications
in the advertising matter to which the company consented,
reported that the evidence, especially that relating to the
therapeutic value of the preparations, was insufficient to
warrant the acceptance of the articles. Since the validity
of therapeutic claims can be determined to a certain extent
by experimental investigation, the Council decided to postpone
final action until sufficient dependable evidence as to
the therapeutic value had been submitted.
Accordingly, a series of questions was sent to the Vanadium
Chemical Company for the purpose of learning on
what pharmacologic evidence the therapeutic claims were
based. After waiting several months, the information
requested not being furnished, the Council took final action
on the products. This action was based both on the evidence
originally submitted and on the advertising matter
being sent out by the company at the time.
Briefly, Vanadiol is said to contain a compound of vanadium
with oxygen and chlorin, which gives up its oxygen
to readily oxidizable substances, such as the blood. In
addition to this compound it contains an oxidizing agent
(sodium chlorate) which is said to serve as a source of
oxygen, so that, according to the theory of the promoters,
Vanadiol acts in the animal system as an oxygen-carrier.
The following is quoted from an advertising circular:
“Most thorough and conclusive physiological tests were made on
guinea-pigs and other animals, which established undoubted evidence
as to the truth of this theory.
“INFLUENCE
“Under the influence of Vanadiol and the other derivatives, the
appetite is increased, there is greater ability to peptonize ingested
proteid material, and, through the improvement in the assimilative
powers and the checking of abnormal fermentations, leads to an
increase in weight. A greater excretion of urea follows their use.
Phagocytic action is promoted by an increase in the leucocytes. All
phases of the elimination of waste materials are favored by the positive
increase in the number of red blood corpuscles and the percentage of
hemoglobin, hematogenesis being thereby rendered more perfect. The
beneficent effect of nascent (active) oxygen, upon the red corpuscles
and upon tissue cells of low vitality are matters of common knowledge.
The results obtained from the vanadium derivatives are not drug effects,
but are due to improved metabolism, which in turn is due to the
removal of microbian toxins, and the general stimulation of cell activity.
“In a tubercular organism, the action of Vanadiol is two-fold. First,
it acts as an antiseptic and antitoxin, combating the Koch bacilli and
neutralizing their poison. Second, as a reconstituent of the economy,
to which it furnishes nascent oxygen, fortifying the defenseless cells
by the very element that is necessary to make them healthy and
resistant.”
“In Anemia and Chlorosis, the blood cells lack oxygen, and in
Neurasthenia the nerve cells are deficient. Vanadiol brings both blood
and nerve cells from a condition of weakness and decay into vital
energy, by furnishing them with active oxygen in a manner that had
not been possible by any other medicine.”
“Vanadiol accelerates the work of digestion by producing HCl in
small doses; it does not hinder the peptonization of albuminoids as
do beta-naphthol, salicylic acid, boric acid, etc., when used as a
stomachal antiseptic, but on the contrary it favors, by hydrochloric
acid, the transformation of albuminoids into peptone without the
assistance of pepsin. Thus, Vanadiol, when given to consumptives,
favors the digestion of large amounts of proteid materials and causes
oxidation of toxins of the stomach. The stomachic action is reflected
in other parts of the organism by the stimulation of the chief functions;
the pulse becomes stronger and muscular strength increases; and, last,
but of greatest importance, is the tremendous increase which will be
noted in the hemoglobin and the red cell count.”
“Phospho-Vanadiol, a combination of Vanadiol with an easily assimilable
organic phosphorus, is an active accelerator of general nutrition
with a special action on the nervous system.”
Such remarkable statements as these are past credence,
certainly, unless they are supported by scientific evidence.
And evidence, either in support or in contradiction of the
claims made, could be obtained; for many of these actions,
at least, are capable of proof by animal experimentation.
The Vanadium Chemical Company was asked to furnish
such proof but failed to do so. The inference is plain! The
committee has concluded that the company has not, and
never has had, any reliable evidence on which to base the
therapeutic claims it has presented to the medical profession.
Here another fact should be noted. It is the connection
shown in The Journal, June 22, 1912, of the general manager
of the Vanadium Chemical Company, F. M. Turner,
with a fraudulent obesity cure concern, the Dr. Turner
Company of Syracuse, N. Y.
It seems, moreover, by all the evidence available, that
F. M. Turner is not authorized to use the title M.D.; yet,
under this title his name appeared on cards representing the
Vanadium Chemical Company and under this title, also, he
published an article in a medical journal recommending to
the medical profession the use of Vanadiol. Later this
article was distributed as an advertising circular by the
Vanadium Chemical Company. Turner’s connection with
the Dr. Turner Company is known and acknowledged by
the Vanadium Chemical Company, yet it still retains him as
general manager!
While there is not necessarily any direct relation between
the personnel of a proprietary manufacturing company and
the value of that company’s product, it is natural that the
medical profession should view with distrust any concern
managed by one who has previously been connected with
such a fraud as the Turner obesity cure.
The committee therefore recommends that the preparations
of the Vanadium Chemical Company be refused recognition,
and that this report be authorized for publication.—(From
The Journal A. M. A., Jan. 18, 1913.)
VENARSEN
Report of the Council on Pharmacy and Chemistry
The report which appears below was sent to the Intravenous
Products Company for consideration. Having considered
the firm’s reply, the Council has authorized publication of its
report along with the explanation sent by the Intravenous
Products Company in reference to the variable composition
reported for Venarsen, namely, that “only the first few
experimental ampules, sent to the doctors for clinical tests,
were made without the Mercuric Iodide.”
W. A. Puckner, Secretary.
This product is prepared by the Intravenous Products
Company, Denver. The advertising circulars contain inconsistent
statements as to its composition. According to one
circular Venarsen is
“... a comparatively non-toxic organic arsenic compound, 0.6 Gm.
representing 247 Mg. (33⁄4 grains) of metallic arsenic in chemical combination....”
According to another circular Venarsen is
“... a comparatively non-toxic organic arsenic compound, 0.6
Gm., representing 247 Mg. (33⁄4 grains) of metallic arsenic and .78 Mg.
(3⁄250 grain) metallic mercury in chemical combination.”
Neither one of these statements gives any information as
to the actual composition of the product. Inquiry addressed
to the manufacturers elicited the reply that:
“Venarsen contains in each 5 c.c. 0.6 Gm. Sodium Dimethyl Arsenate,
.0016 grams of Mercuric Iodide, .0048 grams of Sodium Iodide in
solution in a suitable vehicle for intravenous administration.”
The following report of the examination of Venarsen is
submitted by the Association’s Chemical Laboratory:
LABORATORY REPORT
Three ampules of Venarsen were examined. The first
ampule was labeled
“A comparatively non-toxic organic arsenic compound, representing
247 Mg. (33⁄4 grs.) of metallic arsenic in chemical combination.
5 c.c.—0.6 Gm.”
Practically the same statement appeared in an advertising
circular wrapped around the ampule. The second and
third ampules bore labels identical with the first. The
circulars differed from that accompanying the first ampule
in that the presence of mercury is also announced, thus:
“Venarsen is a comparatively non-toxic organic arsenic compound,
0.6 Gm., representing 247 Mg. (33⁄4 grains) of metallic arsenic and .78
Mg. (3⁄250 grain) metallic mercury in chemical combination and is so
prepared and enhanced as to present the ingredients to the blood in
their most acceptable form.”
Thus, although the potent elements said to be contained
in Venarsen are named, its chemical character (the combination
in which the elements occur) is not disclosed.
The ampules contained a transparent, odorless solution,
possessing the yellow color of salvarsan solution (an
aqueous solution of sodium cacodylate, mercuric iodid and
sodium iodid in the amounts said to be present in Venarsen
is colorless). Qualitative tests demonstrated the presence
in each of the three ampules of sodium cacodylate (sodium
dimethyl arsenate), and the absence of arsenites, arsenates,
phosphates, arsanilates (atoxyl, soamin) and arsenphenolamins
(salvarsan, neosalvarsan). Titrated with normal
hydrochloric acid, using methyl orange as indicator (as outlined
in New and Nonofficial Remedies, 1915, p. 40), the
three ampules were found to contain the equivalent of
respectively, 0.219, 0.253 and 0.216 Gm., or an average of
0.244 Gm. arsenic. (According to statements of the firm
each 5 c.c. of Venarsen contains 0.6 Gm. sodium dimethyl
arsenate [sodium cacodylate], equivalent to 0.247 Gm.
arsenic or 41.66 per cent. Sodium dimethyl arsenate, as
described in New and Nonofficial Remedies, contains 3
molecules of water and 35 per cent. arsenic. This indicates
that the sodium dimethyl arsenate used in Venarsen contains
less water of crystallization than the N. N. R.
product).
Neither mercury nor iodid could be found in the first
ampule. (The company has since explained that mercury
was absent only from the first experimental samples.)
The second and third ampules contained iodid and mercury
in small amount. The exact quantity was not determined
because, on the basis of the mercury content declared, a
single accurate mercury estimation would have required
the purchase of something like 25 to 100 ampules. As each
ampule sells for two dollars, the cost of the material was
considered prohibitive.
From the foregoing we conclude that the first ampule
examined consisted essentially of a solution containing
0.625 Gm. of sodium cacodylate, N. N. R., while the second
and third ampules contained 0.722 Gm. and 0.617 Gm.
sodium cacodylate, respectively, and in addition, a mercury
compound, probably mercuric iodid, dissolved by sodium
iodid.
In other terms, Venarsen as now marketed is a simple
solution containing approximately 9 grains of sodium cacodylate,
1⁄40 grain of mercury “biniodide” and 3⁄4 grain of
sodium iodid to each full dose.
In the past the preparation has been in conflict—especially
serious because of the potent character of the drug—with
Rule 1 (secrecy of composition). The manufacturers have
removed this conflict by furnishing a statement of composition;
and it is to be expected that they will likewise take
steps to remove the manifestly erroneous impression now
likely to be gathered from the circulars, namely, that the
preparation is rather analogous to salvarsan. These conflicts,
however, call for comment, since physicians have doubtless
used the material under misapprehensions.
As to therapeutic claims, the preparation is said to be
effective and safe in syphilis; “lower toxicity and greater
spirochaetacidal power than other known arsenic compounds”
are among the claims. No real evidence for either of these
claims is presented. Sodium cacodylate has been tried as an
antisyphilitic, but with indifferent success; certainly the
results have not been comparable to those of salvarsan. The
mercury could conceivably enhance its effect, but the dosage
appears too small and the course too short for this influence
to be pronounced. Moreover, a careful physician would
not give arsenic and mercury in fixed proportions.
The claim of comparative non-toxicity is probable enough
from what is known about the cacodylate. No physician
should feel “safe,” however, when injecting intravenously 0.6 gm.
of sodium cacodylate every four to six days. Aside from
the grave dangers of intravenous injection in general, the
possibility of idiosyncrasies to arsenicals should always be
borne in mind.
Finally, Venarsen is claimed to be “indicated” in pellagra,
tuberculosis, anemia, etc. No evidence is presented on which
to base an opinion as to its efficiency in pellagra. Those who
have studied that disease would not be likely to resort to
this treatment. In tuberculosis and anemia, there is no sufficient
advantage in giving the cacodylate intravenously.
To summarize, Venarsen treatment consists essentially in
the intravenous injection of large doses of sodium cacodylate.
The other ingredients, as well as the name, merely constitute
so much mystification. While the cacodylate probably has
some effect on the conditions for which it is advised, there
is no evidence that its value even approaches that of salvarsan
in syphilis, or that the intravenous use is preferable to
the ordinary methods. The dangers are manifest, although
they may not be so great as with salvarsan. No justification
has been established for its use in tuberculosis and pellagra.
Physicians who wish to try intravenous cacodylate administration
should have a full realization of the dangers of such
treatment, and in order to avoid further risks, will do well
to refrain from combining other drugs with the cacodylate
in fixed proportions.
It is recommended that Venarsen be held in conflict with
Rule 6 (unwarranted therapeutic claims), Rule 7 (poisonous
ingredients not stated on label), Rule 8 (name does not
express the chemical composition) and Rule 10 (unscientific
combination) and that this report be published.—(From The
Journal A. M. A., May 22, 1915.)
VENODINE
Report of the Council on Pharmacy and Chemistry
Venodine (The Intravenous Products Co., Denver), according
to information sent to the Council, is “an Intravenous
Iodine Compound” put up in ampules each of which contains
“28 grains of Sodium Iodide, 1⁄8 grain each of Beechwood
Creosote and Guaiacol in a suitable vehicle, and excipients
to enhance its compatibility with the circulating blood.”
The “Therapeutic Indications” include “infectious diseases,
such as syphilis, tuberculosis, bronchitis, bacteraemias associated
with chronic and acute nephritis (Bright’s disease),
and other infections.” The Council held as unwarranted and
grossly exaggerated the following therapeutic claims: (1)
that the full therapeutic value of iodin medication cannot be
readily obtained except by intravenous injection; (2) that
Venodine is “of exceptional value in tuberculosis”; (3) that
in pneumonia Venodine “combines the anaesthetic properties
of creosote and guaiacol with the germicidal value of iodine”;
(4) that “Venodine (or its iodine component) has long
enjoyed an exceptional reputation” as of great value in many
infectious diseases including bacteremias. The facts on these
points are the following:
1. Since iodids are easily absorbed from the mucous membrane
of the gastro-intestinal tract and are usually well
tolerated by the stomach, there is no reason for resorting to
intravenous injection in their administration.
2. The indiscriminate administration of iodids for pulmonary
tuberculosis is strongly to be condemned. The cases
in which they can be given to tuberculous patients without
doing harm must be very carefully selected.
3. There is no evidence either that creosote is excreted by
the lungs in sufficient quantity to exert an anesthetic influence
or that iodin is present in the circulation of the lungs or in
the bronchial secretions in a form which is capable of exerting
any germicidal action whatever.
4. It is generally held that the systemic administration of
iodin compounds in bacteremias is useless.
The Council also held the name “Venodine” objectionable
in that it fails to indicate the chief ingredient (sodium iodid)
of this simple pharmaceutical mixture. The statement in a
circular that “Venodine is a sterile solution representing 1.54 Gm.
(24 grains) of iodine in chemical combination together
with creosote and guaiacol” is likely to lead physicians to
use the preparation without considering that its chief constituent
is the well-known substance sodium iodid, particularly
so since no reference to sodium iodid is made in the
circular.
Furthermore, the Council held that the combination of two
such similar substances as creosote and guaiacol (the second
a constituent of the first) as given in the published formula,
stamps Venodine as unscientific; it adds mystery to the
preparation, but does not increase its efficiency, and is therefore
against the best interests of the public.
The Council voted that Venodine be held ineligible for
conflict with Rules 6, 8 and 10.
This report having been submitted to the manufacturers,
in accordance with the Council’s custom, and the reply
affording no reason for modifying the findings, its publication
has now been authorized.—(From The Journal A. M. A.,
June 26, 1915.)
VERACOLATE[AE]
Report of the Council on Pharmacy and Chemistry
“Veracolate (plain),” “Veracolate with Pancreatin and
Pepsin” and “Veracolate with Iron, Quinine and Strychnine”
are proprietary tablets marketed by the Marcy Company,
Boston.
“Veracolate (plain).”—For this the following non-quantitative
formula is given:
“A compound containing the bile acids, sodium glycocholate, sodium
taurocholate with cascara sagrada and phenolphthalein.”
The dose is three tablets. Examination in the Chemical
Laboratory of the American Medical Association of a specimen
of “Veracolate (plain)” indicated that there was about
20 mg. (1⁄3 grain) of phenolphthalein to each tablet. One
dose, therefore (three tablets), would contain 1 grain of
phenolphthalein—an average dose.
“Veracolate with Pepsin and Pancreatin.”—The following
“formula” is given for this mixture:
“Veracolate | 11⁄4 | grain |
“Pure Pancreatin | 1 | grain |
“Pepsin aseptic (1:3,000) | 1⁄2 | grain |
“Oil peppermint | 1⁄10 | min.” |
(Note the presence of two mutually incompatible digestive
ferments.)
“Veracolate with Iron, Quinine and Strychnine.”—This is
stated to have the following “formula”:
“Veracolate | 11⁄8 | grain |
“Reduced Iron | 1 | grain |
“Quinine Sulphate | 3⁄8 | grain |
“Strychnine Sulphate | 1⁄100 | grain” |
It will be noticed that these mixtures increase in complexity
until a combination of seven diverse ingredients, a
veritable shotgun mixture, is evolved. In none of the “formulas”
are the proportions of the purgative drugs in Veracolate
stated. In the second “formula,” the digestants might
as well be omitted, for the pancreatin is destroyed by peptic
digestion and hence cannot pass the stomach while the
pepsin is useless without hydrochloric acid, and, at any rate,
of no value in the intestine. If one is indicated, the other is
not. Yet this unscientific and complex combination of purgatives,
mutually incompatible digestive ferments, and oil of
peppermint is called:
“A scientific Blending of Digestive Ferments, Cholagogues and Carminatives.”
“... for all forms of indigestion and dyspepsia.”
And the third, an equally irrational and complex combination,
is termed “The Ideal Cholagogic Tonic”!
Extravagant and Misleading Claims.—True to type, the
claims are magnified in accordance with the number of
ingredients. For instance, of “Veracolate (plain),” we are
told:
“Veracolate is a true cholagogue and biliary disinfectant as it directly
stimulates the liver cells producing an increased flow of limpid bile.
Although not a purgative, it moves the bowels and is definite and
dependable in its action.”
“The action of Veracolate is to bring about a profuse flow of healthy
bile which prevents bile stasis. As the flow of bile is stimulated so antiseptic
action ensues, calculi softened and the concretion and mucous
eliminated. Mucosal swelling is diminished and the infection which is
usually present is antagonized. Relief is in plain evidence. As a result
of the treatment the skin, eyes and urine become normal in appearance
in a short time, the appetite and digestion improve and soreness in the
region of the gall-bladder is entirely relieved.”
Similarly, it is said of “Veracolate with Pancreatin and
Pepsin” that:
“It causes a natural flow of bile which checks fermentation, prevents
the absorption of toxines and causes the food elements to be emulsified
and thus rendered easy of assimilation. All this conduces to a natural
movement of the bowels. Digestion is at once improved and the epigastric
pain, nervous symptoms and headache disappear.”
“Veracolate with Iron, Quinine and Strychnine” is said
to be indicated in:
“Hepatic Torpor accompanied by Anemia, Chlorosis, Debility, Neurasthenia
and Neuroses.”
And the physician is asked to believe that it will:
“... give gratifying results in all nervous, anemic, and ‘run down’
conditions in which the liver function is usually subnormal.”
The objections to “Veracolate (plain)” are that it is semi-secret
in composition, unscientific in combination and
exploited under unwarranted claims. The same criticisms
hold with reference to “Veracolate with Pancreatin and
Pepsin” and “Veracolate with Iron, Quinine and Strychnine.”
These products are discreditable to the medical and pharmaceutical
profession alike and their use is against the public
good. The Council therefore refused recognition to Veracolate
and its preparations.—(From The Journal A. M. A.,
April 24, 1915.)
HAYDEN’S VIBURNUM COMPOUND[AF]
Report of the Council on Pharmacy and Chemistry
The following report on Hayden’s Viburnum Compound
was prepared by a member of the Council’s Committee on
Therapeutics. The Council held the preparation in conflict
with its rules and authorized publication of the referee’s
report.
W. A. Puckner, Secretary.
Hayden’s Viburnum Compound, according to the advertising
circulars, was first compounded in 1860 by W. R.
Hayden. The medical profession is told that W. R. Hayden
“... found by his experiments that a combination of the active
principles of Viburnum Opulus, Dioscorea Villosa, combined with
aromatics, proved a valuable remedy for Spasmodic Dysmenorrhea.”
As in 1860 W. R. Hayden was not a physician (he received
a diploma from the Eclectic Medical College, New York, in
1867), and, so far as we can learn, he was not a pharmacist
or chemist, one wonders what kind of “experiments” he made.
Hayden’s Viburnum Compound is put on the market by the
New York Pharmaceutical Company of Bedford Springs,
Mass. The name of this concern may sound imposing, until
it is realized that it is merely a trade name adopted by
Hayden in exploiting his nostrum.
The advertising matter formerly claimed that Scutellaria
(skull-cap) was one of the ingredients of Hayden’s Viburnum
Compound. As this is no longer mentioned, it is fair
to assume that even the manufacturer does not consider the
composition to be of vital importance. Stress is laid on the
superior efficacy of viburnum opulus in the conditions for
which the preparation is recommended; it is emphasized
that it is viburnum opulus, and not viburnum prunifolium,
that is the important ingredient of Hayden’s Viburnum Compound.
The label, in accordance with the requirements of the
Food and Drugs Act, declares that the preparation contains
50 per cent. alcohol. The claim is made:
“It is free from all narcotics and leaves no unpleasant after-effects.”
The medical profession is told that Hayden’s Viburnum
Compound is a remedy in:
“Hysteria, Bilious Colic, Cramps of Cholera Morbus, Muscular
Cramps,... Nervous Diseases of Pregnancy, Threatened Abortion,
Post-Partum Pains, Puerperal Convulsions, Rigid Os, Dysmenorrhea,
Menorrhagia.”
DISCUSSION OF ALLEGED INGREDIENTS
Viburnum Opulus (Cramp Bark).—Botanists and pharmaceutical
chemists declare that this drug has not been on the
American market for many years, if ever, and that the drug
used and even described as viburnum opulus is really the
bark of another plant. Viburnum opulus and its preparations
are therefore to be dropped from the next United States
Pharmacopeia. The principal constituents of viburnum
opulus are stated to be a glucosid, viburnin, a bitter resin,
and a little tannin, with small amounts of earthy carbonates
and phosphates and organic acids (Culbreth, Ed. 4, 1906,
p. 591). The glucosid and resin being bitter, the drug might
have a slight stomachic action (if, indeed, any such effect
is actually produced by “bitters”); the small amount of
tannin might make it slightly astringent; its fruit acids
(citric and malic) might make it slightly diuretic. Even if
viburnum opulus were present in Hayden’s Viburnum Compound
there is no clinical or laboratory proof that it, if
given alone—without alcohol or other drug—has any antispasmodic
or nervous sedative action.
Dioscorea Villosa (Wild Yam).—This drug contains a
saponin and an acrid, irritant resin. It has never been
proved clinically or experimentally that this drug has any
action whatever except that its irritant resin might, if taken
in sufficient quantity, cause irritation of the stomach and
vomiting.
Aromatics.—The irritation produced by concentrated
aromatics causes increased peristalsis and consequently may,
if there is no obstruction, relieve intestinal stasis and intestinal
colics. Therefore, a preparation containing large
amounts of aromatics, especially if given in hot water, would
have practically the same effects in the “cramps of a cholera
morbus” or other forms of acute diarrhea as a home-brewed
cup of spiced tea—and no more.
Alcohol.—This drug is a muscle relaxant, and sufficient
doses might, by relieving spasm, relax a muscularly contracted
os uteri and relieve post-partum pains. Alcohol
dilates the blood-vessels both in the abdomen and on the
surface of the body. It may thus either relieve uterine bleeding
by lowering the blood-pressure and causing more blood to go
to other parts, or increase uterine bleeding by relieving arterial
and muscle spasm. Alcohol is also a habit-forming narcotic
(Hayden’s Viburnum Compound is advertised as “free from
all narcotics”!) and when habitually used by either males or
females tends to impair the capacity to produce normal
offspring.
Even if the manufacturer’s “formula” be accepted, Hayden’s
Viburnum Compound contains no therapeutically active
ingredient except alcohol and aromatics. The recommended
dose of this preparation is “... two teaspoonfuls in six
of boiling hot water or milk and one teaspoonful of sugar,
every fifteen or twenty minutes until relief is obtained.”
“Frequently after taking the Viburnum Compound the patient will
sleep soundly for several hours from the sudden cessation of pain;
in such cases she should never be awakened through any fear of
oversleeping, as Hayden’s Viburnum Compound does not contain any
narcotic whatever, nor does it leave any disagreeable after-effects, and
it may be given to a child when necessary without any special caution.”
Read the foregoing and then remember that it means that
one teaspoonful of alcohol (the equivalent of two teaspoonfuls
of whisky) is to be given in hot water, every fifteen or
twenty minutes until relief is obtained and the patient is
asleep. Why not use plain language and say “until she is
drunk”?
The thoughtful physician would consider it decidedly
unwise to give alcohol to a young girl, to a prospective
mother, or to a young mother, except under extraordinary
circumstances. He would know that the menstrual pains for
which this preparation is recommended are likely to be
recurrent, and that the repeated taking of alcohol for recurrent
pain is fraught with danger—the danger of initiating the
alcohol habit. If, however, a physician does elect to give
alcohol as a drug, he must let the conditions that govern
each individual case determine whether it is not better to
give it as whisky, or to disguise it in a prescription of his
own. Above all the physician should be conscious that he
is giving the drug, alcohol; and this is not the case when he
prescribes a ready-made nostrum. If he writes a prescription
containing whisky or other alcoholic he will take measures
to avert the dangers inseparable from the use of this
drug.
CONCLUSIONS
1. Even if the manufacturer’s formula for Hayden’s
Viburnum Compound be accepted, it is apparent that any
therapeutic activity the preparation may have is due essentially
to the alcohol and aromatics.
2. Alcohol is a narcotic drug, and a habit-forming drug.
Physicians ordering this preparation may, by so doing, be
initiating the alcohol habit.
3. Whatever result is obtained by the use of Hayden’s
Viburnum Compound in the treatment of uterine or pelvic
disturbances, is due to the alcohol it contains. The fact that
menstrual pains are likely to recur might, when this preparation
is relied on, become a factor in the formation of the
alcohol habit.
4. Whatever result is obtained by the administration of
Hayden’s Viburnum Compound in the treatment of gastro-intestinal
disturbances is due to the alcohol and the aromatics
it contains.
5. Whisky has the same alcoholic content (50 per cent.)
as Hayden’s Viburnum Compound; the dangers in the use
of whisky are well known and its value as a therapeutic
agent is being questioned more and more every year.
Holding the exploitation of this proprietary a danger to
the public and a detriment to scientific medicine, the referee
recommends publication of this report as a protest against
such irrational therapeutics. The profession should recognize
that most, if not all, of the preparations recommended for
painful menstruation and for all kinds of pelvic pain contain
large percentages of alcohol, and that whatever physiologic
effect is produced is, for all practical purposes, due to the
alcohol.—(From The Journal A. M. A., Jan. 23, 1915.)
VIN MARIANI
Report by Council on Pharmacy and Chemistry—With Comments
Thereon
This preparation was assigned to a subcommittee of the
Council and the following is an abstract of the report of
the committee:
Samples of Vin Mariani and of the literature distributed
by the manufacturers were examined.
It appears that the beverage or medicine known as
“Vin Mariani” is a preparation of red wine, apparently
imported from Bordeaux, and fortified, in this country,
by an alcoholic preparation of coca leaves or other parts
of the coca plant.
The committee considered first, the character of the
red wine as imported. A sample received from the port
of New York, March 10, 1905, from Henry Clausel &
Co., Bordeaux, and consigned to Mariani & Co., on
analysis was found to have the following composition:
Specific gravity | 0 | .9959 |
Alcohol by volume | per cent. | 10 | .99 |
Extract | per cent. | 2 | .279 |
Volatile acids | per cent. | 0 | .0914 |
Ash | per cent. | 0 | .2801 |
Reducing sugar | trace. |
Pol. direct | degrees | —0 | .8 |
Pol. invert | degrees | —0 | .7 |
K.So | M. per liter | 0 | .092 |
A sample of Vin Mariani, as bought in the open
market in an original package, has also been analyzed
and found to have the following composition:
Specific gravity | 1 | .0125 |
Alcohol by volume | per cent. | 16 | .15 |
Extract | per cent. | 8 | .602 |
Ash | per cent. | 0 | .277 |
Glycerin | per cent. | 0 | .444 |
Volatile acid | per cent. | 0 | .0747 |
Tartaric acid | per cent. | 0 | .2400 |
Alkaloids (coca bases) | per cent. | 0 | .0250 |
Cane sugar | per cent. | 2 | .35 |
Reducing sugar | per cent. | 3 | .38 |
The increased alcoholic strength of Vin Mariani over
the Bordeaux wine from which it is made, as shown by
this analysis, doubtless comes from the alcohol extract
containing the coca bases, as already stated. Approximately
6 per cent. of sugar is also added to the wine.
Judging from the analysis, therefore, Vin Mariani corresponds
to a mixture of an alcoholic preparation of
coca leaves and ordinary Bordeaux red wine, with the
addition of about 6 per cent. of sugar.
Vin Mariani conflicts with Rule 5, which requires
that “No article will be admitted or retained, concerning
which the manufacturer or his agents make misleading
statements as to geographical source, raw material
from which made, or method of collection, or
preparation,” by stating in the advertising literature
that: “The United States government, under the Pure
Food Law of March 3, 1903, further emphasizes all
previous analyses of Vin Mariani by admitting Mariani’s
wine as absolutely pure and unadulterated.”
Whatever may have been the intent of the above statement,
its effect is to deceive. The conjunction of the
terms “Vin Mariani” and “Mariani’s wine” can only be
construed as meaning the same thing. Inasmuch as it
does not appear that Vin Mariani is imported into this
country, it would not have been possible for the United
States government to inspect it, and as to the wine
obtained from Henry Clausel & Co., from Bordeaux, it
is not in any sense Mariani’s wine except that of ownership.
It is the opinion of the committee that this phrase
can only result in deception and the construction of the
language strongly favors the supposition that it is
intentionally meant to deceive.
This false claim is practically repeated in the other
pamphlets published by the Vin Mariani Company,
although not always in the same words.
This preparation also conflicts with Rule 6, which
states that “No article will be admitted or retained of
which the manufacturer or his agents make unwarranted,
exaggerated or misleading statements as to therapeutic
value,” in that the firm’s letter-heads have printed on
them the following:
“Vin Mariani purifies the blood stream, strengthens the circulation,
stimulates muscular fiber and nerve tissue, is a respiratory
stimulant, strengthens the heart muscles, and is an emergency food
in the absence of all other nutriment. Successfully employed as
an adjuvant in anemia, debility, diseases of the chest, nervous
troubles, muscular or mental overstrain, neurasthenia, and allied
conditions, and in certain cases of protracted convalescence.”
The committee believes that Vin Mariani is intended as
a beverage rather than as a medicine.
The report concludes:
“The committee recommends, therefore, that Vin Mariani
be refused recognition and that this report be published in
full or in part.”
In accordance with this recommendation the above extract
of the report is herewith published.
W. A. Puckner, Secretary.
VIN MARIANI MADE IN THIS COUNTRY
According to the above report, Vin Mariani as imported
is simply an ordinary cheap French wine, the preparation
sold in this country as Vin Mariani being compounded in
this country. Yet the advertising literature, the label on
the bottle, etc., state directly or indirectly that it is a French
preparation. Until recently—presumably until the vendors
realized that the truth regarding this point would come out—the
advertisements in medical journals contained an analysis
made by a chemist in Paris. The shape of the bottle,
the character of the printed matter accompanying the bottle,
etc., are evidently intended to convey the impression that
it is imported. So far, then, as this point is concerned,
Vin Mariani is sold under gross misrepresentations and is
a fraud.

Advertisements of Vin Mariani before and after national Food and
Drugs Act went into effect.
ADVERTISED TO THE PUBLIC
Vin Mariani was at one time advertised to the public in
this country, but, so far as we know, it is not at the present
time; at least, not directly. Yet it is most effectively
advertised to the public indirectly, and this with little
expense to the promoters, the cost of the circular around
the bottle being the only expense—doctors who prescribe
it do the rest. If those who are in the habit of prescribing
Vin Mariani will examine the advertising that goes into
the hands of their patients they will realize how true it is
that our profession is responsible for much of the “patent-medicine”
taking. Few laymen could withstand the temptation
to buy the stuff for any ailment that comes along when
they read in the circular that this “medicine,” which their
doctor evidently thinks is a good thing, is so highly recommended,
for all the ills that befall us mortals, by the Pope
of Rome, the Czar and the Czarina of Russia, the Queen
of England, the Shah of Persia, the King of Norway and
Sweden, the Queen of Portugal, the Queen of Saxony, the
Crown Prince of Cambodia, Ferdinand of Bulgaria, and by
a whole list of ambassadors, generals, politicians, musicians,
actresses, etc. The testimonials of these great men
and women are enough to convince the most skeptical that
this remarkable medicine will do everything but raise the
dead—and under favorable circumstances accomplish even
this. And still more—it will win battles! Witness this
from the governor-general of Madagascar: “We were
refreshed by Vin Mariani, and before morning carried the
stronghold.” Alexander Dumas and Emile Zola are credited
with calling it “the elixir of life.” One very strange thing
about the testimonials in the circular used in this country
is that all are written by foreigners. But Americans
(President McKinley—think of it—among others) are honored
by having their testimonials quoted in the circulars
used on the other side of the Atlantic. Why? Is it possible
that the testimonials are fakes?
AN ETHICAL CURE-ALL
Here are a few of the conditions that the circular says
Vin Mariani is good for: “Anemia, winter cough, debility,
vocal weakness, la grippe, continued fevers, bronchitis, nervous
troubles, muscular weakness, diseases of the aged,
malaria, melancholia, overwork, neurasthenia, impotence,
malnutrition, depression, heart troubles, wasting diseases,
mental overstrain, and in certain cases of protracted convalescence.”
The following quotations are taken from blotters—circulated
in this country—which are evidently intended for the
laity, as well as for physicians:
“Vin Mariani creates and sustains vigor and energy. Guards
against wasting diseases. When everything else has failed try it to
prove merits.”
“Lung, Throat and Stomach Troubles benefited by Vin Mariani;
this Ideal French Tonic strengthens entire system of Body, Brain
and Nerves.”
“Most Efficacious, Most Agreeable, Unequaled by anything in
Fortifying, Strengthening, Refreshing.”
WHY BLAME THE LAYMAN FOR USING NOSTRUMS?
Can we blame the layman for using Peruna, Wine of
Cardui, etc., simply because they are advertised, when there
are physicians who, for the same reason, prescribe concoctions
that are just as quackish and just as useless?
And can editors of medical journals consistently find fault
with newspapers for carrying advertisements of fraudulent
“patent medicines” when they themselves admit to their
pages advertisements of nostrums that are no less fraudulent
and of no more value?
MEMBER OF PROPRIETARY ASSOCIATION
One word more: There is an organization known as the
Proprietary Association of America, but it is usually referred
to in common parlance as the “patent-medicine” men’s association.
It will be remembered that last year we printed a
list of the members of this body, among which was the Vin
Mariani Company. It will be remembered also that in the
list were the names of certain firms who were supplying
medicines to physicians, but practically all these resigned
from membership and their resignations were published by
us. We have not had the pleasure of publishing the resignation
of the Vin Mariani Company. On the contrary, we
note that at the last annual meeting of the “patent-medicine”
men’s association this firm was still an active member,
Mr. A. L. Jaros, who stands for the Mariani Company
in this country, being one of those registered at the meeting.—(From
The Journal A. M. A., Nov. 26, 1906.)
VIROL
Report of the Council on Pharmacy and Chemistry
Virol, sold in the United States by the Etna Chemical
Company, is put out by Virol, Ltd., London, England. It is
said to be
“A preparation of bone marrow, red bone marrow of medullary
structure of ox rib and calf bones, eggs, malt extract, and lemon syrup
made from fresh fruit.”
“Marrow fat is emulsified with extract of malt, lemon syrup and
eggs, it is further enriched with soluble phosphates of lime, iron and
soda, and glycerine solution of red bone marrow.”
Many of the therapeutic claims made for Virol the Council
considered grossly exaggerated; among them are the following:
“... a complete food for children.”
“... a complete nutrient.”
“The value of the Lime-Salts (representing the Egg Shells) contained
in Virol is fully illustrated in its influence upon Rickets; whilst Struma,
Chronic Bronchitis, Anaemia and Influenza are all combated by its use
in a degree, which, we venture to say, has never been approached.”
“The fat, as represented by the Marrow-bone and Egg Yolk, is so
minutely divided that it admits of even far more rapid absorption by
the villi of the intestine than milk.”
“It is an ideal form of food, readily digested and assimilated in the
weakest of conditions.”
“Virol has a marked effect on the metabolism of the body, increasing
the production of opsonins and stimulating phagocytosis. As an adjuvant
to the natural defensive processes of the patient in all diseases of bacterial
origin its value can scarcely be over-estimated.”
The objections of the Council were transmitted to Virol,
Ltd. The firm’s reply was reported to the Council by the
referee of the Committee on Therapeutics who held that no
adequate or satisfactory answer had been made to the objections
raised against the advertising claims for Virol.
The claims made for Virol being unsubstantiated and
unwarranted, the Council voted that the preparation be
refused recognition.
In accordance with the practice of the Council, the
exploiters of Virol were afforded an opportunity to comment
on the foregoing statement before its publication. On their
objecting to the findings, the entire matter was turned over
to a second referee. This referee, in making his report,
reviewed the claims made for Virol and commented on the
lack of evidence to substantiate these claims. He stated that
a chemical analysis of the preparation had yielded the following
result:
“Sugar, as maltose | 60.0 per cent. |
“Fat | 13.2 per cent. |
“Proteins | 3.2 per cent. |
“Ash | 1.6 per cent. |
“Water and volatile matter | 21.6 per cent. |
“There is no appreciable diastatic action. A little glycerol
is present in the volatile matter. The preparation is therefore
an extract of malt with fat and a small amount of
protein.”
He then continues:
“That Virol has food value cannot be denied, but that it
has sufficient value to warrant the claims of the manufacturers
is not evident. Virol cannot be considered a complete
food, as the advertising literature reiterates, or an
ideal food for infants. The amount of protein is far too low
in comparison with the carbohydrates to warrant this view.
The dosage recommended is not large. Thirty gm. a day
would furnish only about 1 gm. of protein and 4 gm. of fat.
The 3 teaspoonfuls a day recommended for children would
furnish protein and fat even below this.
“If employed alone it cannot be a complete or sufficient
food, and if employed along with other articles of diet—milk
and bread, for example—it is not easy to see wherein lies
the efficacy of the small weights of fat and protein added in
the form of Virol. Here the demand made on our credulity
is too great, as the protein in the preparation is the familiar
protein of eggs, meat and malt, and the fat largely that from
the egg-yolk and marrow, according to the claims. It is not
known that any specific virtues reside in these bodies, or in
the egg-shells, also claimed as present.
“In the opinion of the present referee there is nothing in
the composition of Virol to justify the claims made for it.
The judgment and recommendations of the first referee
follow from the facts and must be accepted by the Council.”
The Council directed that the previously prepared report
be allowed to stand and that it be published along with a
suitable reference to the report of the second referee.—(From
The Journal A. M. A., Feb. 20, 1915.)
PART II
CONTRIBUTIONS FROM THE CHEMICAL
LABORATORY
ANUSOL HEMORRHOIDAL SUPPOSITORIES [AG]
W. A. Puckner and L. E. Warren
An abstract of an article concerning “anusol suppositories”
was published in The Journal, Jan. 23, 1909. This gave
the results of an analysis by a foreign chemist, J. F. Suyver,
which were to the effect that “anusol suppositories” contained
no “anusol.” Schering & Glatz, the American agents
for “anusol” suppositories, took exceptions to the abstract,
asked that The Journal retract, and submitted the findings
of a chemist in support of their claim that the suppositories
do contain “anusol.” To determine the composition of
“anusol hemorrhoidal suppositories” as they are found on
the American market, trade packages were purchased (April
6, 1909) and submitted to examination90 in the Association’s
laboratory.
According to the claims of the manufacturers, 12 suppositories
contain:
“Anusoli | 7.5 | grams. |
“Zinc oxid | 6.0 | grams |
“Balsam Peruv | 1.5 | grams |
“Ol. theobrom | 19.0 | grams |
“Ungt. cerat | 2.5 | grams” |
Calculated to percentages the formula reads:
Anusoli | 20.54 | per cent. |
Zinc oxid | 16.44 | per cent. |
Balsam Peruv | 4.11 | per cent. |
Ol. theobrom | 52.06 | per cent. |
Ungt. cerat | 6.85 | per cent. |
When this product was submitted to the Council some time
ago, Schering & Glatz stated that, according to the manufacturer,
“anusol” is the “iodo resorcin sulphonate of bismuth,
having the following rational formula: [C6H2ISO2.O(OH)2]3Bi.
In the meta-dioxybenzol C6H4(OH)2, the resorcin,
one H has been replaced by one I, and for another H
the sulfonic-acid group SO2-OH has been substituted, so that
meta-dioxybenzol is transformed into C6H2ISO2-OH(OH)2.
In the sulfonic acid the H of OH is replaced by Bi and,
as Bi is trivalent the above rational formula results.”
According to this formula “anusol” should contain:
Iodin | 32.99 | per cent. |
Sulphur | 8.34 | per cent. |
Bismuth | 18.07 | per cent. |
And the “anusol” suppositories should contain:
Iodin | 6.77 | per cent. |
Sulphur | 1.71 | per cent. |
Bismuth | 3.71 | per cent. |
Examination showed that the suppositories contain about
0.08 per cent. iodin, or 1.2 per cent. of the amount claimed;
0.28 per cent. sulphur, or 16.3 per cent. of what is claimed;
0.71 per cent. bismuth, or 19 per cent. of what is claimed;
and zinc equivalent to 16.5 per cent. zinc oxid, or about 100
per cent. of claim.
From the standpoint of the iodin content alone, assuming
that all of the iodin found is present in the form of “anusol,”
the results of the examination of the product (as found on
the American market) verifies, for all practical purposes,
Suyver’s statement that “anusol suppositories contain no
anusol,” for the quantity of iodin present is so minute (about
1⁄82 of that required by the formula) as to be unworthy
of serious consideration. The presence of sulphid in appreciable
amounts was demonstrated showing that the sulphur
is present, at least in part, in the form of sulphid and not
as sulphonate as is claimed. In a measure, too, this is in
accord with the findings of Suyver, who concluded that,
in the product which he examined, the bismuth was present
in the form of sulphid. The proportions of sulphur and of
bismuth (respectively about 1⁄6 and 1⁄5 of the required
amounts) indicate still further that the product is not all
that it is claimed to be.
A specimen submitted by Schering & Glatz to the Council
two years ago contained 0.99 per cent. iodin, or 1.3 per cent.
of the amount claimed; 0.23 per cent. sulphur, or 13.4 per
cent. of the claimed amount; and 0.52 per cent. bismuth, or
14 per cent. of what is claimed by the formula. Since the
above determinations were made another specimen of Anusol
Hemorrhoidal Suppositories was received from Schering &
Glatz, July 16, 1909. This sample was found to contain
about: 0.075 per cent. iodin, or 1.1 per cent. of the amount
required by the formula; 0.265 per cent. of sulphur, or 15.5
per cent. of the requirement and 0.88 per cent. bismuth, or
23.7 per cent. of the required amount. It will thus be seen
that the composition of the oldest specimen and also that of
the specimen recently sent, corresponds in a general way to
that of the one first examined.
Whether judgment be based on the determination of the
bismuth, the sulphur or the iodin, the results just given
clearly show that the claims made concerning the composition
of “Anusol Hemorrhoidal Suppositories” are not substantiated
by the facts.—(From The Journal A. M. A., Oct. 2,
1909.)
AROMATIC DIGESTIVE TABLETS
W. A. Puckner and L. E. Warren
It has been amply demonstrated91 that pepsin and pancreatin,
when in solution, mutually destroy each other; if the
solution be acid, the pepsin destroys the pancreatin; if
alkaline, the pancreatin destroys the pepsin. By using the
characteristic effect of pepsin on proteids in acid medium
and that of pancreatin on proteids and starches in an
alkaline solution it can readily be demonstrated that commercial
liquid preparations labeled as containing both of
these ferments actually contain only one ferment. They
are misbranded.
Besides the liquid a goodly number of solid preparations,
chiefly tablets, containing pepsin and pancreatin are offered
to the profession. Among these are tablets consisting simply
of pepsin and pancreatin. Since pepsin and pancreatin interact
only when in solution, it is quite possible to prepare tablets
which contain these ferments. The use of such tablets
is, however, unscientific, since one or the other of the
ferments is destroyed when it comes in contact with the
fluids of the digestive tract. In addition to simple tablets
containing pepsin and pancreatin only there is at present a
host of “digestive tablets” on the market. Among these
are some which must be classed with the “digestive impossibilities”
(Reports of the Council on Pharmacy and Chemistry,
1910, Vol. 1, p. 41). The preparations referred to are
tablets claimed to contain pepsin, pancreatin, diastase,
hydrochloric acid and lactic acid. When it is considered
that the United States Pharmacopeia defines hydrochloric
acid as “a liquid composed of 31.9 per cent. by weight of
absolute hydrochloric acid (HCl = 36.16) and 68.1 per cent.
of water,” i. e., a solution of hydrogen chlorid, a gas, in
water, it would at first appear that the incorporation of any
appreciable quantity of hydrochloric acid in tablets would
be impracticable. Hydrochloric acid, however, possesses to
a limited extent the property of combining loosely with
protein substances so that it becomes possible to bring
about its combination with pepsin and similar substances to
form compounds which are relatively stable at ordinary
temperatures. Because of the volatility of the free acid and
its limited combining power with protein substances (100 gm.
boiled beef combine with 2 gm. absolute hydrochloric
acid92), the quantity of acid in any tablet can never be large,
much less than sufficient to be of any therapeutic value.
A number of firms offer “digestive tablets” for sale having
formulas of which the following may be considered typical:
Sacch. Pepsin | 4 | grains |
Pure Pancreatin | 1 | grain |
Diastase | 1⁄4 | grain |
Aromatic Powder | 1⁄4 | grain |
Lactic Acid | 1⁄8 | grain |
Hydrochloric acid | 1⁄8 | grain |
Some manufacturers use United States Pharmacopeia
pepsin in place of the saccharated article; others do not
give the exact quantities of hydrochloric acid which their
product is supposed to contain, but make use of the indefinite
expression “q. s.;” still others state merely that hydrochloric
acid is present, but make no claim whatever concerning
the quantity.
From purely theoretical considerations it is possible that
the tablets referred to might contain appreciable amounts of
hydrochloric acid. Since the formulas for some of the
tablets furnish no information concerning the content of
hydrochloric acid, it seemed worth while to determine the
quantity, if any, actually present in some of the tablets on
the market. Accordingly a trade package of “digestive
aromatic tablets,” as put up under the label of each of six
American manufacturers, was purchased and submitted to
examination in the Association laboratory.
Qualitative tests made on specimens from each brand of
tablets demonstrated the absence of uncombined hydrochloric
acid in each. Further tests93 showed that hydrochloric
acid in protein combination was present essentially
in the amounts claimed in three of the specimens. In two
of the others hydrochloric acid was entirely absent; in the
remaining one only the merest traces of hydrochloric acid
could be found.
| H. K. Mulford Company |
| “DIGESTIVE AROMATIC” |
“Pepsin, Sacch. | 4 | grains |
“Pancreatin | 1⁄2 | grain |
“Diastase | 1⁄16 | grain |
“Acid Lactic | 1⁄8 | grain |
“Acid Hydrochloric | 1⁄8 | grain |
“Aromatic Powder | 1⁄4 | grain |
| “Dose: 1 or 2 tablets.” |
In the above formula each tablet is said to contain 1⁄8
grain hydrochloric acid. This amount is equivalent to
0.002584 gm. (1⁄25 grain) absolute hydrochloric acid. Analysis
demonstrated that each tablet contains about 0.00267 gm.
hydrochloric acid (absolute HCl) or essentially the
amount claimed. The average dose of diluted hydrochloric
acid United States Pharmacopeia is 1 c.c., equivalent
to 0.1049 gm. absolute hydrochloric acid. To obtain this
quantity from the above preparation the patient would be
required to swallow more than three dozen of the tablets.
| Wm. S. Merrell Chemical Company |
| “DIGESTIVE AROMATIC, 5 GRAINS” |
“Pepsin | 80 | parts |
“Pancreatin | 10 | parts |
“Diastase | 1 | part |
“Acid Lactic | 1 | part |
“Acid Hydrochloric | 3 | parts |
“Aromatic Powder | 5 | parts” |
Calculation shows that each tablet should contain about
0.0031 gm. of absolute hydrochloric acid. The analysis
indicated that each tablet contains about 0.0030 gm. hydrochloric
acid (absolute HCl) in protein combination, or
practically the amount claimed. One pharmacopeial dose
of hydrochloric acid is contained in 35 of the tablets.
| Parke, Davis & Company |
| “DIGESTIVE AROMATIC” |
“Saccharated Pepsin | 4 | grains |
“Pure Pancreatin | 1 | grain |
“Diastase | 1⁄4 | grain |
“Aromatic Powder | 1⁄4 | grain |
| “Lactic Acid. |
| “Hydrochloric Acid. |
| “Dose: 1 to 3 tablets.” |
Chlorid is present in small amounts, but quantitative
examination indicated that hydrochloric acid, either free
or in protein combination is absent. An ammonium salt
is present in small quantities.
| Sharp & Dohme |
| “DIGESTIVE AROMATIC” |
“Pepsin, Sacch., U. S. P. | 4 | grains |
“Pancreatin, pure | 1 | grain |
“Diastase | 1⁄4 | grain |
“Aromatic Powder | 1⁄4 | grain |
“Lactic Acid | q. s.” |
“Hydrochloric Acid | q. s.” |
Small quantities of chlorid are present. Quantitative
examination indicated that hydrochloric acid in protein
combination is present only in very small amounts, each
tablet containing but about 0.00034 gm. of absolute acid, or
about 0.34 per cent. of the pharmacopeial dose. Ammonia
is absent. Inasmuch as more than 300 of these tablets
would be required to furnish a pharmacopeial dose of
hydrochloric acid, this firm’s interpretation of the expression
“q. s.” would prove interesting.
| Truax, Greene & Company |
| “SYNERGIA” |
“Synergia” is claimed to be composed of “pepsin, pancreatin,
veg. diastase, lactic acid, hydrochloric acid and aromatics;
dose, 1 to 3 tablets.” The specimen contained no
hydrochloric acid, either free or in protein combination.
A trace of ammonia and small quantities of chlorid were
found.
| The Fraske Tablet Company |
“Pepsin Sacch. | 80 | parts |
“Pancreatin, Pure | 10 | parts |
“Diastase | 1 | part |
“Lactic Acid | 1 | part |
“Hydrochloric Acid | 3 | parts |
“Aromatic Powder | 6 | parts |
| “Dose: 1 to 2 tablets. |
| “Each tablet represents (5) grains of the above mixture.” |
According to the formula hydrochloric acid (31.9 per cent.
absolute HCl) represents 3 parts in 101 of the preparation
from which the tablets are made. Each tablet (containing
5 grains of the mixture) should have 0.00307 gm. absolute
hydrochloric acid. Analysis showed that each tablet contains
hydrochloric acid in protein combination equivalent
to an average of 0.003066 gm. absolute hydrochloric acid,
or essentially the amount claimed. It would be necessary
to give 34 tablets to administer a pharmacopeial dose of
hydrochloric acid.
[Editorial Note: The above indicates that the use of such
tablets is irrational, unscientific and that it should be condemned.
The only constituent of these tablets, other than
the aromatics, which might possibly be of benefit in stomach
troubles, is the pepsin. But even if it be assumed that the
diastase and pancreatin could exert their characteristic
effects, their aid to digestion (metabolism) would be but
slight, because their amounts in the tablets are too small
to be of any value.
It is claimed that the tablets contain diastase in amounts
varying from 1⁄20 to 1⁄4 grain. Assuming the diastase used
to be of first-class quality, i. e., capable of converting 200
times its own weight of starch into soluble products, the
quantity of one tablet would be capable at the most of
digesting but from 10 to 50 grains of starch, an amount
equal at the most to but a small spoonful of oatmeal or a
very dainty bite of bread. In the same way the quantity of
pancreatin is insufficient to be of any material aid in
digestion should it in some way escape destruction in the
stomach and still retain its full activity when it reaches the
alkaline juices of the intestines. One grain of pancreatin
of full United States Pharmacopeia strength will digest
only 25 grains of starch or the proteids in about 100 c.c.
of milk.
Saccharated pepsin, which was formerly official, was
required to digest 300 times its own weight of moist egg
albumin, while the pepsin that is now official is required to
digest ten times that amount, or 3,000 times its own weight.
It is evident, therefore, that the tablets should contain
sufficient pepsin to digest appreciable amounts of protein.
No intelligent physician would prescribe these tablets simply
for the pepsin they contain or are supposed to contain;
if he wanted to give pepsin he would prescribe the drug
in the simple form.
Clinical experience has shown that in the majority of
cases of so-called dyspepsia the stomach contents contain too
much rather than too little hydrochloric acid, and wherever
there is a sufficiency of acid there is usually no decrease
in the secretion of pepsin. In many of such cases, too,
digestion is normal, or even more active than normal, but
even when it is imperfect there is seldom any lack of pepsin.
Insufficient digestive power is most often due to a deficiency
of hydrochloric acid and not to lack of pepsin in the
stomach contents. In the tablets under consideration, however,
hydrochloric acid is present—if at all—in the most
ridiculously minute quantities; quantities that are so small
as to preclude any therapeutic effect except that due to
the psychic element.
These tablets, with their six or more ingredients, are
typical “shotgun prescriptions.” Such prescriptions catch
the unthinking doctor as well as the self-drugging public,
for, while clinical experience and physiologic experiments
have demonstrated that the old ideas regarding the value of
these digestives and ferments were erroneous, the public
and many members of the medical profession still seem to
be influenced by the old theories.
In conclusion we must not lay all the blame on the
manufacturing firms for supplying these absurd combinations;
the physician who prescribes them should assume a
large share of it. If the doctors did not use them the manufacturing
concerns would soon stop putting them on the
market. We hope, however, that those manufacturing concerns
that like to be classed as reputable will cease to disgrace
their catalogues with what they know to be therapeutic
absurdities.]—(From The Journal A. M. A., Aug. 20, 1910.)
BURNHAM’S SOLUBLE IODIN [AH]
W. A. Puckner and A. H. Clark
Burnham’s Soluble Iodin, according to the manufacturers,
is one of the most noteworthy “discoveries” of the age.
The advertisements aim to create an impression that while
the product contains iodin, pure and simple, yet by some
secret process this element has been so changed as no longer
to possess its usual properties. The Burnham Soluble Iodin
Company makes such extravagant claims for its product
and gives such wide publicity to these claims that it seemed
advisable, in the interests of the profession, to determine
the nature of the preparation. Its examination was accordingly
taken up in the laboratory of the American Medical
Association.
From the analysis, we conclude that Burnham’s Soluble
Iodin is a solution of iodin in alcohol made miscible with
water by the presence of some iodid. Wilbert94 and other
investigators have arrived at practically the same conclusion.
Whatever the secret process, hinted at in the advertisements,
by which this preparation is evolved, the fact remains
that when one prescribes Burnham’s Soluble Iodin, one is
prescribing iodin, together with an iodid, the nature of
which is hard to determine. The iodid is not present as
potassium iodid, nor, entirely, at least, as hydrogen iodid
(hydriodic acid), but this is of slight importance compared
with the fact that it is a solution in alcohol of free iodin
and an iodid, and therefore is essentially the same as
Lugol’s solution.
The amount of iodin found corresponds approximately
to 3 gm. of free iodin and 2 gm. of combined iodin in
100 c.c. of the solution. Lugol’s solution contains 5 gm.
free iodin, and 10 gm. potassium iodid in 100 c.c.
BURNHAM’S SOLUBLE IODIN TABLETS
Burnham’s Soluble Iodin Tablets are light brown compressed
tablets, stamped with the letters B. S. I. in monogram.
Each tablet is said to contain 3 minims Burnham’s
Soluble Iodin.
The average weight of each tablet was found to be
0.3526 gm.; since Burnham’s Soluble Iodin was found to
have a specific gravity of .8527 and to contain 4.5 per cent.
total iodin, the tablets should contain approximately 2.3
per cent. total iodin, about one-half to two-thirds of which,
depending on the condition of the “Soluble Iodin” from
which they are made, should be free iodin. Instead of this,
only 0.317 per cent. free iodin and 1.57 per cent. total iodin
was found. Analysis shows that Burnham’s Soluble Iodin
tablets contain approximately one-fourth the amount of free
iodin and approximately two-thirds the amount of total iodin
which should be contained therein if, in accordance with
the label, each tablet contains 3 minims of Burnham’s
Soluble Iodin.
COMMENT
The literature put out by the Burnham Soluble Iodin
Company is in itself enough to condemn the products it
advertises. The much emphasized statement of the company
that
“Something had to be done; and Burnham’s Soluble
Iodin is that which has been done”
fulfils, in its blatant assertiveness, all the requirements of
nostrum advertising. The results of the analyses are not,
therefore, a surprise.
Secrecy is just as essential today to the successful
exploitation of this class of proprietaries as it was before
the demand for formulas became so universal. The requirement
of publicity is evaded, therefore, in one of two ways:
Either a formula is given which is false, or at least meaningless,
or else the claim is made that the method of preparing
the product is a unique and remarkable secret that is
possessed only by the manufacturers. The Burnham Soluble
Iodin Company uses the latter device.
Meanwhile, physicians will be perfectly justified in viewing
with suspicion all claims based on such conspicuously
unscientific premises, more especially so when these claims
fail to find substantiation on careful and painstaking
analyses. In brief, whenever the physician wishes to administer
free iodin, Lugol’s solution (Liquor Iodi Compositus,
U. S. P., Physician’s Manual, page 84) is an inexpensive
and perfectly available preparation.—(From the Journal
A. M. A., March 28, 1908.)
“HYDROCYANATE OF IRON—TILDEN”
W. A. Puckner and W. S. Hilpert
Among the many inquiries received regarding the composition
of secret remedies was one in reference to “Hydrocyanate
of Iron” manufactured by the Tilden Company,
New Lebanon, N. Y. This preparation is advertised as
being “unexcelled as a remedy for epilepsy, hysteria, chorea,
neurasthenia, locomotor ataxia, neuralgia, migraine, anemic
headaches, and all convulsive or reflex neuroses dependent
on impairment of the brain or spinal cord.” It is also said to
be “valuable in uterine reflex neuroses due to congestion;
in amenorrhea due to anemia and chlorosis and suppressed
menstruation.”
The term “hydrocyanate of iron” is an unfamiliar one and
is not found in any available reference work on chemistry.
Thinking that the term might have been loosely applied to
ferrocyanid of iron, or Prussian blue (a compound once
suggested for epilepsy, but long ago considered useless),
the correspondent wrote to the manufacturers asking if such
were the case. The Tilden Company answered:
“... our preparation Hydrocyanate of Iron is not Prussian blue
in any sense of the word. Prussian blue has no curative properties as
applied to all forms of epilepsy. Prussian blue is Ferrocyanid of Iron
while our preparation is Hydrocyanate of Iron.”
The only statements in the Tilden Company’s advertising
matter regarding the composition of hydrocyanate of
iron are the following:
“Hydrocyanate of Iron (Tilden’s) is a correct and scientific combination
of well known principles.”
“Hydrocyanate of Iron (Tilden’s) combines well known properties of
ferruginous salts with the sedative action of Hydrocyanic acid.”
The last statement would lead one to expect the presence
of available iron and cyanogen ions. In fact, the inference
to be drawn from all the company’s “literature” is that
“hydrocyanate of iron” is a definite chemical compound in
the same sense as is ferrocyanid of iron, and that inference is
still further borne out in the letter to our correspondent.
This being the case, the Tilden Company was again written
to and asked for the chemical formula of “hydrocyanate of
iron,” with the following result:
“Replying to your inquiry regarding the formula of Hydrocyanate
of Iron we beg to state the composition of this preparation is a trade
secret and we therefore do not care to furnish the desired information.”
This reply verified the opinion already formed that “hydrocyanate
of iron” is a secret preparation. Its analysis was
then taken up in the Association’s laboratory.
EXAMINATION OF THE TABLETS
The product appears on the market in cartons said
to contain one ounce of one-grain tablets. On the cartons,
in addition to the name of the preparation and the
name and address of the manufacturers, are the names of
diseases for which it is recommended. The tablets, in the
specimens analyzed, were dark blue, rather hard and
slightly bitter in taste and had an average weight of
0.1382 gm., or about 2 grains. They were found to be
practically insoluble in water and dilute mineral acids;
aqueous oxalic acid solution partially dissolved them, yielding
a blue solution. Boiling with alkali hydroxid solution
decomposed the tablets, yielding iron in an insoluble
form and a solution of alkali ferrocyanid, as demonstrated
by the appearance of a deep blue precipitate on the addition
of ferric chlorid solution. The portion insoluble
in alkali when boiled with hydrochloric acid yielded a
solution containing iron, approximately equivalent to 50
per cent. Prussian blue. These properties are all characteristic
of Prussian blue, and, taken together, identify Prussian
blue as a constituent of “hydrocyanate of iron
(Tilden).” The insoluble residue from the iron determination
possessed the properties and constituents of talc
and constituted practically one-half of the tablets. Extraction
of the tablets with chloroform or ether in the presence
of ammonium hydroxid yielded a small amount of organic
material which contained bodies having the properties
of, and responding to tests for, quinin or cinchona alkaloids
and caffein. The presence of a salicylate was also indicated.95
From the analysis it is concluded that “hydrocyanate
of iron (Tilden)” is essentially a mixture of approximately
equal parts of talc and Prussian blue, containing traces
of organic matter having the general properties of
alkaloids.
Comment: When a firm exploits an abandoned remedy
for so hopeless a disease as epilepsy under a name not
known to chemistry and with a false representation of its
pharmacologic qualities, such action may rightly be assumed
to show ignorance or worse. “Hydrocyanate of iron,” if
it means anything, means the cyanid of iron, but the preparation
put out under that name is, according to our chemists,
not cyanid of iron, but the ferrocyanid of iron commonly
known as Prussian blue. This substance has been tried for
epilepsy and abandoned. Yet the firm recommends it as
a “peerless remedy” for this disease:
“The Tilden Company holds the key to the situation in the treatment
of epilepsy. We have the remedy that does the work.”
Not that epilepsy is the only disease for which this
hypothetical chemical compound may be prescribed. Torticollis
has been “successfully treated with hydrocyanate of
iron.” In chorea, we are told “a richer and better blood
supply” should be furnished the nervous and vascular system
and “the irritation of the motor centers” must be
allayed.
“Hydrocyanate of iron serves admirably to accomplish both of
these purposes. It carries the hemoglobin to the blood in its most
easily assimilable form and its hydrocyanic acid possesses remarkable
sedative powers....”
It is not possible for it to have any value in anemia
because of its insolubility, yet we are told:
“In conditions marked by poverty of the blood producing anemia
or chlorosis, reacting on the nervous system and calling for a chalybeate,
hydrocyanate of iron (Tilden’s) takes a front rank among
the remedies of this class, combining as it does the blood enriching
qualities of ferrum with the sedative action of hydrocyanic acid.”
As Prussian blue yields no appreciable quantity of hydrocyanic
acid under the conditions existing in the animal
organism, “the sedative action of hydrocyanic acid” must
be as hypothetical as the chalybeate properties attributed to it.
It is strange that a manufacturer, in introducing a new
chemical compound, should have to assure his customers
that it “contains no opium or alkaloid of that drug, cocain,
chloral hydrate, conium or any of the bromids.” Imagine
a firm putting, let us say, potassium iodid—a definite
chemical compound—on the market and solemnly guaranteeing
that it contained no cocain or chloral hydrate!
Would the Tilden Company of twenty-five years ago have
served such mental pabulum in its advertising matter?
One would think that the dictates of common humanity
would protect the unfortunate epileptic from the machinations
of the nostrum maker, especially from the exploitation
of a remedy that has been tried and found wanting. A
nostrum, however, merely has to measure up to one standard:
Will it pay? Meeting this requirement nothing else matters.—(From
the Journal A. M. A., June 19, 1909.)
HYMOSA
W. A. Puckner and W. S. Hilpert
Frequent requests for information regarding the composition
of hymosa, manufactured by the Walker Pharmacal
Co., St. Louis, and a perusal of the extensive and nostrum-like
advertising the product is receiving, made a chemical
examination of this preparation seem desirable. If the label
is to be believed, hymosa has been of use in “acute and
chronic muscular and articular rheumatism, gout, sciatica,
lumbago, pleurodynia and neuralgia, whether due to uric
acid diathesis or not ...”
The composition of hymosa as given by the proprietors
is set forth in the following statement:
“... Hymosa, in which the remedies Frangula, Actea Spicata,
Stellaria Media, Franciscea Uniflora, Rhus Toxicodendron, Passiflora
Incarnata, Phytolacca Decandra and Echinacea Angustifolia are combined
in the proportions which experience has shown will obtain the
quickest and best results without any of the stomach and heart complications
so often following the administration of salicylic acid.”
“Contains no Salicylic Acid.”
Thus the explicit statement is made that hymosa contains
certain vegetable drugs (most of them obsolete and valueless)
and that it does not contain salicylic acid. By inference
the claim repeatedly is made that the nostrum does not
contain any salicylates.
“... Hymosa has achieved most remarkable results in overcoming
rheumatism in cases where salicylates have been tried in vain ...”
“Salicylic acid was not successful in this case of rheumatism of
the stomach.”
“Negative results from salicylates—Hymosa cures.”
“... the salicylates didn’t help? Then we must try Hymosa.”
Still harping on the undesirability of salicylates and the
value of hymosa the advertising pamphlets state:
“Salicylic Acid Replaced. The Use of This Dangerous Agent in
Rheumatism Obviated.”
“It seems that the use of the dangerous and ineffective salicylic
acid will soon be given up and hymosa take its place.”
“Former methods of treating rheumatism ... have been very
unsatisfactory ... because of the heart and stomach difficulties
brought on by salicylates of which most rheumatism remedies are
composed.”
“Could not tolerate the salicylates.”
Finally in a letter issued to physicians we are told:
“... you will find hymosa to possess prompt and positive curative
action with the additional advantage of avoiding the heart and stomach
complications, which the salicylates too often cause.”
It is evident from the above quotations, in which the
salicylates are denounced specifically or by implication, and
from the label which states that no salicylic acid is present,
that the exploiters of the nostrum deliberately intended to
give the impression that hymosa is free from salicylates
or salicylic acid and contains only the vegetable or plant
drugs enumerated. The very fact that the proprietors make
such repeated efforts to give the impression that hymosa
is free from salicylates is in itself sufficient to arouse
suspicion and hence in the examination particular attention
was given to the detection of salicylic acid or salicylates
with the following results:

Reproduction (reduced) of an advertisement of Hymosa. This indicates
the attempt made to convey, by implication, the idea that the
salicylates are absent from Hymosa.
Examination.—Hymosa as purchased on the market is
a dark brown liquid with an odor of sassafras and a
rather sweetish taste, reacting acid to litmus. Qualitative
tests having indicated the presence of salicylate, iodid,
sodium, potassium, alcohol and some organic matter, presumably
sugars and some plant extractives, these were
determined quantitatively.96 It was found that a part
of the salicylate was present as free salicylic acid and
part in a combined form. The sodium determinations
indicated that all the salicylate, excepting that in the form
of free salicylate acid, was present as sodium salicylate.
From the results of the potassium estimations, it was
evident that the iodin was present in the form of potassium
iodid.
From the results of the analysis it is believed that the
preparation has approximately the following composition:
Salicylic Acid | 0.32 | gm. |
Sodium Salicylate | 1.15 | gm. |
Potassium Iodid | 0.32 | gm. |
Sugars and extractives | 4.60 | gm. |
Alcohol, U. S. P. | 16.86 | c.c. |
Water to make | 100.00 | c.c. |
These results indicate that hymosa is essentially a solution
containing salicylic acid, sodium salicylate, potassium iodid,
alcohol, sugars and plant extractives in the proportions
given above, and show that the various statements referred
to, regarding the absence of salicylic acid and salicylates
are misleading and untrue. It further illustrates the repeatedly
demonstrated fact that nostrums exploited as wonderful
and new discoveries are new in name only—and whatever
therapeutic value they possess depends on old and tried
medicinal agents.
[Editorial Note: In describing the methods employed by
the manufacturers of Manola in exploiting their product,
attention was called to the fact that the Manola Company
was reported as being a subsidiary affair of the Luyties
Homeopathic Pharmacy Company of St. Louis. It is reported
that this same company also operates the Walker Pharmacal
Company, which exploits Hymosa and Psa-avena.]—(From
the Journal A. M. A., June 11, 1910.)
MICAJAH’S MEDICATED UTERINE WAFERS
W. A. Puckner and W. S. Hilpert
Evidently touched by the generosity of the manufacturer
in sending him a sample and literature, but not too favorably
impressed by the claims made for the preparation
referred to, a correspondent writes:
I enclose a valuable sample and literature just received. Such a
palpable humbug as Micajah’s Uterine Wafers would hardly seem to
need notice were it not probably true that many practitioners habituated
to the use of samples are still influenced by the glowing accounts
of cures wrought; especially when attested by such a name and title
as “Elmore Palmer, M.D., Ex-President Western New York Medical
Society.” This secret gynecologic medicament is recommended for anything
from “Pruritis Vulvæ,” “Enlargement of the Womb,” “Displacements,”
“Cystocele and Rectocele,” to the “Menopause.”
Following the definition that by her “stomach” a woman means
anything from her chin to her knees, the ex-president with truly noble
impartiality has with the wonderful Micajah wafers wrought lightning
cures all the way from “stone-bruise” of the heel to nasal polyp and
influenza, and some of them are male patients too.
With the foregoing as an impetus to investigate the nature
of this much advertised nostrum, the wafers were submitted
to analysis by the Association laboratory. The report
follows:
LABORATORY FINDINGS
Trade packages of the wafers purchased on the open
market bear the name of the preparation and that of the
manufacturers, Micajah & Co., Warren, Pa. The label
states that the nostrum is a:
“Disinfectant, astringent and local alterative of the greatest virtue.
A remedy for the local treatment of the diseases of women. Inflammation,
engorgement and prolapse of the womb, vaginitis, leucorrhea,
menstrual derangements and the disturbances incidental to the
menopause. Also highly recommended for affections of the mucous
membranes in general, particularly those of the nose, the throat,
the rectum, and for gonorrhea, cystitis, etc.”
“This box contains wafers for three months’ treatment.”
“Price per box $1.00.”
The box contained 25 tablets, and a circular entitled,
“Hints on the treatment of diseases of women,” in which
directions for the treatment of many diseases are given.
It ends with a paragraph which contains the following
statement:
“There is no doubt that the field of usefulness of Micajah’s Wafers
can be indefinitely enlarged by the ingenuity and therapeutic skill
of the physician.”
Much of the advertising “literature” is in the form of
leaflets, brochures and small pamphlets full of testimonials
by physicians.
Micajah’s uterine wafers as found on the market are
white, hexagonal tablets, odorless and possessing an
astringent taste. The wafers are soluble in water with
extreme difficulty. Hot hydrochloric acid and alkali
hydroxids dissolve the powdered tablets readily, leaving
a slight residue which under the microscope and by
physical tests was identified as lycopodium.
The acid solution of the wafers responded to qualitative
tests which indicated the presence of potassium, sodium,
aluminum sulphate, borate and a mere trace of a fatty
material. Quantitative estimation of boric acid, aluminum
sulphate, sodium and potassium were made, which indicated
that Micajah’s Uterine Wafers consist of alum more
or less anhydrous or “burnt,” boric acid and borax in
approximately the following proportions:97
Alum, dried | 59.86 | per cent. |
Borax, dried | 15.62 | per cent. |
Boric acid | 5.67 | per cent. |
Water of hydration | 18.85 | per cent. |
The average weight of the tablets is 0.7791 gm (11.8
grains) and allowing for the fact that the quantity of
water present in commercial exsiccated alum varies, each
tablet would contain approximately 0.4986 gm. (7.8 grains)
burnt alum; 0.2337 gm. (3.6 grains) crystallized borax,
and 0.0467 gm. (0.7 grain) boric acid.
COMMENT
Judging from the “literature” that goes with the packages
of this nostrum, one might imagine that it was put up
absolutely for the layman, but this is not the case. It is
advertised only in medical journals and not directly to the
public. But direct advertising to the public is not necessary;
for every physician who prescribes these wafers at
the same time places in the hands of his patient advertising
matter intended to influence that patient—and it usually
does. As a result this preparation is being bought by the
public direct. To what extent we do not know, but physicians
are responsible for it. Probably if physicians realized
that the same interests that control Piso’s Consumption Cure
also control Micajah’s Medicated Uterine Wafers they would
not be so ready to act as the unpaid agents for the concern.
That such simple astringents and feeble antiseptics as
alum, borax and boric acid could have such remarkable
curative effects on uterine diseases is absurd. The serious
aspect of the matter is, that, by the encouragement given
them in the advertising literature to treat themselves, women
may neglect proper surgical or medical attention in the early
stages of serious diseases such as cancer or dangerous
pelvic infections, until they get beyond the hope of proper
management. But when nostrum promoters urge the use
of such inefficient remedies in the treatment of gonorrhea,
it is time to look at the matter seriously. Considering the
vital social significance of the venereal diseases, the employment
of useless remedies can only favor the spread of these
infections, which cause such a large proportion of the
diseases which afflict women particularly.
The medical profession for the most part has become
mentally calloused to the exaggerated claims of the nostrum
makers and does not make sufficient effort to condemn them.
There may be some physicians, however, who use such
preparations as these wafers in their practice, as is indicated
by the circulars wherein the manufacturers suggest
that their “usefulness can be indefinitely enlarged by the
ingenuity and therapeutic skill of the physician.” It is only
occasionally that a physician voices his indignation as to
these humbugs, as in the case of the physician whose letter
is quoted above.—(From the Journal A. M. A., March 26,
1910.)
The Firm Replies
To the Editor:—We have read with interest the report of
your committee on pharmacology recently published in The
Journal, on the subject of Micajah’s Medicated Uterine
Wafers, and your comments thereon.
We are of the opinion that, in your laudable efforts to
reform the practice of pharmacology, it is not your desire
or intention to act other than justly and fairly, and therefore,
with this belief, we submit the following statements for
your consideration, with the hope that you will see fit to
publish them.
1. We do not seek by word or deed the patronage of the
laity, and what few sales are made to the public are not of
our contriving, nor should we be held responsible for them,
any more than is the manufacturer of quinin to be blamed
for the universal use of that drug.
2. Our literature should not be considered extravagant, for
it is for the most part made up of clinical reports received
from physicians and based on the unsolicited testimonials
in our possession from hundreds of practitioners, many of
whom have used Micajah’s Wafers in practice from five
to twenty years and they are therefore as well grounded
as are the clinical reports concerning any preparation.
3. In the past year we have endeavored to place our
preparation on a higher ethical basis by stating in our advertisements
what our wafers contain, and by eliminating whatever
seems to us open to criticism.
4. That the ingredients of the preparation are “simple” is
no reason for considering them valueless. H. A. Kelly, in
his work on medical gynecology, page 266, recommends
these ingredients in a variety of conditions. Bandler also
made important recommendations bearing on this subject in
his “Medical Gynecology,” 1909 edition, page 472. We
feel we have the right to recommend this preparation for
these and similar conditions, especially when our statements
are backed up by the clinical experience of numerous general
practitioners.
5. That the owner of Micajah’s Wafers holds stock in a
corporate firm which manufactures proprietary medicines
and toilet articles, advertised to the laity, should not militate
for or against our right to market a meritorious preparation
on strictly ethical lines to the medical profession, inasmuch
as many of the largest drug houses cater to both the doctor
and the proprietary interests, and several are actively
engaged in exploiting so-called nostrums.
6. We enclose a recent advertisement which has been
accepted after investigation of our methods by careful medical
journals, and we now believe we are conducting our
business in entire conformity with the best interests of the
medical profession and we feel certain of the true merits
of our article.
Micajah & Company, Warren, Pa.
[Comment: This letter brings out still more strongly the
points raised in the article which appeared in The Journal,
March 26, 1910. Being unable to analyze motives we must
perforce, accept Micajah & Co.’s statement that they “do not
seek by word or deed the patronage of the laity.” In
the comments on the laboratory’s report it was very explicitly
stated that this nostrum was advertised only in medical
journals and not directly to the public. Inasmuch, however,
as the container in which this product comes has printed
on it the various diseases in which the “wafers” are indicated,
as, moreover, within the container there is a leaflet
which describes in detail the use of the preparation in
a list of pathologic states varying from “enlargement of the
womb” to “gonorrhea in the male,” and, finally, as the
name “uterine wafers” would seem in itself to be a plain
bid to the public, we still maintain that “one might imagine
that is was put up absolutely for the layman.”
The proposition that advertising matter should not be
considered extravagant because it is largely “made up of
clinical reports received from physicians” is an argument
that is as old as the nostrum business itself—and as fallacious
as it is old. Unfortunately, as our files show, the
most extravagant statements made for proprietary products
frequently emanate from men who legally are entitled to
write M.D. after their name. The fact that it is not the
manufacturer but a Buffalo physician who tells of the
marvelous results he obtained from the use of Micajah’s
Medicated Uterine Wafers in forty-three cases comprising
no fewer than thirty-six pathologic conditions from “otitis
media” to “injured toe,” and from “bunion” to “ophthalmia
neonatorum” does not exempt the firm that prints such stuff
from the charge that its “literature” is not merely extravagant,
but ridiculously so.
As Micajah & Co. say, because the ingredients of their
preparation are simple is no reason for considering them
valueless. On the contrary, if the “wafers” were truthfully
exploited for what they are and what they will do, their
very simplicity would be a virtue. But such has not been
done. And therein lies the viciousness of nostrums. Simple
mixtures of well-known drugs are foisted on the medical
profession with no hint as to their composition and with
claims made that are not only false, but would immediately
be recognized as absurd, if their actual composition were
known.
That a mixture of borax and alum may be of value
in some of the simple ailments of the female genital tract
can easily be granted. That relief might follow the use of
suppositories made of these ingredients—especially when
supplemented by an increased attention to simple cleanliness—can
also be admitted. To say, however, that such
medicaments will quickly and permanently cure gonorrhea,
urethritis, endometritis, etc., is foolish, false and vicious.]—(From
the Journal A. M. A., April 16, 1910.)
NOITOL AND ANADOL
W. A. Puckner and L. E. Warren
Noitol
The Journal received an inquiry concerning the composition
of Noitol, a preparation which is being advertised
to the medical profession as a “specific” for the cure of
eczema and certain other cutaneous diseases. The preparation
is manufactured by the Wheeler Chemical Works,
Chicago. Trade packages of Noitol were purchased and
examined in the Association laboratory. On the label of
the package, Noitol—an inversion of the word “lotion”—is
described as follows:
NOITOL
(Dr. Bradbury’s Eczema Lotion.)
For External Application Only!
Our Most Popular Specialty.
A specific for the cure of Eczema, Scrofulous and Syphilitic
Eruptions, Lupus, Salt Rheum, Tetter, Itch. This remedy is
composed of valuable Oils, combined with Vegetable and
Mineral Acids in such proportions as cause a rapid and
permanent cure of the above complaints.
Noitol is a clear, nearly colorless, acid solution, the
greater portion of which is water. Its specific gravity is
1.0097 at 25 C.
Qualitative tests demonstrated the presence of a chlorid,
a nitrate, a mercuric salt, free acid and glycerin. No “oils”
or “vegetable acids” could be found.
Analysis98 of the preparation indicated that its composition
is essentially as follows:
Mercuric Chlorid | 0.0463 gm. | in 100 c.c. |
Mercuric Nitrate | 0.0450 gm. | in 100 c.c. |
Glycerin | 1.3021 gm. | in 100 c..c. |
Nitric Acid | 1.0265 gm. | in 100 c.c. |
Water (by difference) | 98.5545 gm. | in 100 c.c. |
From the above it appears that Noitol is simply a weak,
acid solution of mercury salts—the total being approximately
equivalent to a 1 to 1,000 bichlorid of mercury solution—exploited
under a meaningless name. It is but one more
example of the old, old story of a well-known remedy
being sold at a high price under a name which is in no
way indicative of its composition, and under claims which
are absurdly false.
The price of the mixture is $2.00 a pint; the estimated
cost, exclusive of the container, is about 6 cents a gallon,
or, put another way: the price of a pint bottle, it is estimated,
would make a barrel (31 gallons) of the nostrum.
The incorrect statement concerning its components, the
unwarranted therapeutic claims made for it, and the exorbitant
price easily place Noitol in the front rank among
the “patent medicine” frauds. Yet it is advertised to physicians
as an ethical proprietary and is evidently being prescribed
by them.
Anadol
In the circular matter accompanying the trade package
of the preparation, “Noitol,” described above, a preparation
called “Anadol” is described. Anadol is claimed to
be an analgesic and antipyretic. In the descriptive circular
there is no information concerning the composition of the
preparation, but from the general therapeutic description
the physician might easily be led to believe that “Anadol”
is a distinct chemical substance.
To reduce temperature the physician is advised to push
the administration of Anadol in 10 grain doses until the
febrile condition is under control or until a maximum of
70 grains of the preparation has been ingested. The
circular further states:
“... in this lies the special value of ANADOL; there are
no annoying by-effects; the stomach bears the remedy well and neither
circulation, respiration, nor the nerve centers show the least disturbance.”
As no evidence could be obtained concerning the composition
of Anadol and, as the preparation is being brought to
the attention of physicians by means of circulars in connection
with the distribution of Noitol, it seemed worth while
to take up its examination in the Association laboratory.
Accordingly a trade package of the material which had
passed into interstate commerce was purchased.
Qualitative tests demonstrated the presence of sodium,
a carbonate, caffein and acetanilid, the latter in considerable
quantities. Analysis99 indicated that the composition of
the specimen examined is essentially as follows:
Acetanilid | 79 | per cent. |
Caffein | 1 | per cent. |
Sodium bicarbonate | 20 | per cent. |
Since, according to the circular, it is permissible to
prescribe 70 grains of this preparation within 21⁄2 hours,
a patient thus treated would receive no less than 55 grains
of acetanilid! In view of the numerous cases of poisoning
due to the misuse of acetanilid (“The Harmful Effects of
Acetanilid, Antipyrin and Phenacetin,” U. S. Dept. Agric.,
Bur. Chem., Bull. No. 126) the physician should be apprised
of the composition of Anadol.
[Editorial Note: The chemical investigations reported
above emphasize once more the need of such an institution
as the Association’s laboratory and again demonstrates the
value of its work. At first sight it seems disheartening to
find that physicians are so easily humbugged. Yet when
it is remembered that it is impracticable for physicians
either to analyze such products themselves or to go to the
expense of having chemists do it for them, it is evident
that the fault lies not so much with the physicians as with
the conditions that make the exploitation of such frauds
possible. It is on the public that the burden ultimately falls,
for it is the layman who has to pay two dollars for a few
cents’ worth of medicine. But—and this is far more serious—that
the physician should be urged to dose his patient
with an insidiously dangerous drug to a point far beyond
the limits of safety, is little less than criminal. Yet so long
as unknown medicinal products are prescribed just so long
will this danger be a very real one.]—(From the Journal
A. M. A., May 21, 1910.)
Anadol Declared Misbranded
Anadol was analyzed at the Bureau of Chemistry and the
chemists reported that it contained over 82 per cent. of
Acetanilid. As the labels did not bear any statement as
to the quantity of acetanilid contained in the nostrum, the
stuff was declared misbranded and the defendant, on pleading
guilty, was fined.—[Notice of Judgment No. 795.]
PIX CRESOL
W. A. Puckner and W. S. Hilpert
In a paper on “The Abuse of Chemical Formulas”100 several
examples were given of the various methods employed
by “patent-medicine” concerns to give standing to their
products by assigning to them a chemical formula. In some
cases the formulas given are impossible, in other cases they
may represent the chemical composition of only one constituent
or it may be an attempt at both. To a chemist
such formulas are absurd and on seeing a formula which
he knows to be wrong he naturally thinks “Fake,” “Ignorance,”
or both. Just such a formula (C5H6N.SO) applied
to a product called Pix Cresol, manufactured by the Pix
Cresol Chemical Co., Kansas City, Mo., attracted our attention.
No mention of such a formula can be found in such
works as Richter’s most complete Index of Carbon Compounds,
nor the three supplemental volumes published,
1901–1905, by the German Chemical Society and Beilstein’s
Organic Chemistry (3d Ed.). This fact, supplemented by
inquiries from correspondents as to the composition of the
substance made it seem worth while to make a chemical
examination of it.
The examination was made and showed that the essential
constituent was oxyquinolin sulphate. As potassium
sulphate was also found it was concluded that Pix Cresol
was a preparation containing a mixture of oxyquinolin sulphate
and potassium sulphate, which has also been known
in the past under the proprietary name, “Chinosol.” At
this time a letter was referred to the laboratory containing
the report of an analysis of Pix Cresol, which showed
the presence of oxyquinolin sulphate but no potassium
sulphate. As this indicated that Pix Cresol contained as
its essential constituent the substance now sold as Chinosol,
the laboratory purchased a new specimen of Pix Cresol
from the Chicago representative of the Pix Cresol Co. The
examination101 of this specimen showed that it consisted of
approximately 21 per cent. oxyquinolin sulphate, about 8.3
per cent. potassium sulphate and the remainder almost
entirely milk sugar.
It is evident, then, that both the specimen of Pix Cresol
obtained directly from the manufacturers and also the one
purchased more recently from the Chicago agent, contain
as an essential constituent Chinosol of the composition sold
formerly. The substance now sold under the name Chinosol
and described in New and Nonofficial Remedies is pure
oxyquinolin sulphate, and as the exploiters of Pix Cresol
probably obtain their supply of oxyquinolin sulphate from
the Chinosol Company, the sole American agents for
Chinosol, it is to be expected that Pix Cresol should change
in composition. It is probable that the analysis referred to
the laboratory deals with a more recent specimen than the
two examined in the Association laboratory.
Editorial Note: In view of the Council on Pharmacy and
Chemistry’s findings, viz., that chinosol is a powerful antiseptic
but a feeble germicide and considering that Pix Cresol
contains but 21 per cent. oxyquinolin sulphate, the absurdity
of the following claims made for Pix Cresol require no
further comment:
“Pix Cresol is an Absolutely Sure and Yet Perfectly Safe, Never
Failing Destroyer of Pus (Staph. Pyogenes Aureus.)”
“It is germicidal, bactericidal, bacillicidal. It is certain as a micro-organism
destroyer. It destroys absolutely.”
“Ridding the blood of germs, it aids in rendering it replete with
oxygen——.”
“It kills the germs.”
“No organism that is causative of morbid processes can withstand it.”
“It destroys micro-organisms of all kinds. It destroys them absolutely.”
“The germ’s tenacity of life does not avail against its action as
germicide.”
“It destroys the spores and toxins utterly.”
A further estimate of the pseudo-chemical company, bearing
the name of this “strongest, safest, least expensive medical
antiseptic, disinfectant and deodorizer known” may be
gained by a cursory glance at some of its “specialties”:
“Maizinin compound, Positive Chill and Malaria Specific”
the firm says, “prepares the parasites for execution by the
leukocytes.” It is said to contain arsenic, while the name
implies the presence of some plant drug.
“Psora, the Syphilis Specific,” is a shot-gun mixture said
to be “the scientific combination of the soluble Triple Iodids
with the active principles of Echinacea, Cascara amagra,
Berberis aquif., and Phytolacca rad.,” and is claimed to
make “the syphilitic lesions disappear and fail to return.”
“Rectoids—Cones for the treatment of all rectal trouble,”
are said to be “a combination of Rectin (Pix) compounded
from Buckeye, Collinsonia, Hamamelis, Belladonna, Pix
Cresol.”
“Tablets for the Female—Pix Cresol Uterettes,” it is said,
“for sanitary purposes may be continued forever ...”
When one realizes that this sort of pseudo-scientific twaddle
is accepted by many physicians at its face value, the
outlook for therapeutics seems dark, indeed. So long as the
existence of such concerns is tolerated by the medical profession,
so long will there be a crying need for a “Propaganda
for Reform in Proprietary Medicines.”—(From The
Journal A. M. A., June 10, 1911.)
SALIODIN
W. A. Puckner and A. H. Clark
[The Council on Pharmacy and Chemistry refused recognition
to Saliodin because it conflicts with Rules 1 and 6,
and directed publication of the following.
W. A. Puckner, Secretary.]
Saliodin is sold by the Saliodin Chemical Co., Scranton,
Pa. In the literature and on the trade package the following
“formula” is given:
This formula being indefinite and vague, the examination
of saliodin was taken up in the Association laboratory.
From the analysis we calculate the composition of
saliodin to be approximately equivalent to a mixture of:
Sodium salicylate | 57.54 |
Potassium iodid | 1.18 |
Potassium acetate | 30.00 |
| Matter volatile at 130° (oil of anise, oil of |
gaultheria, moisture, etc.) | 8.10 |
Undetermined (extractive?) | 3.18 |
| ———— |
| 100.00 |
The analysis shows that the formula is not only
indefinite and vague, but incorrect and false.
To emphasize the incorrectness of the published formula
the following comment on its first two items is
offered:
In the “formula” it is stated that 20 grains of saliodin
contain approximately “salicylic acid (aceto-salicylate)
Grs. XV.” The statement is not clear, but conveys
the impression that 20 grains of saliodin contain an
amount of aceto-salicylate, a salt of acetyl-salicylic acid
(aspirin), equivalent to 15 grains of salicylic acid. But
the chemical examination shows that it contains neither
acetyl-salicylic acid, nor salt of acetyl-salicylic acid, nor
even salicylic acid itself. In the place of these, the
analysis shows that over half of saliodin is the common,
every-day sodium salicylate.
According to the “formula,” each 20 grains of saliodin
contains “iodin (iodate), equivalent to iodid potass. Grs.
XV.” This statement, too, is vague, but conveys the
impression that 20 grains of saliodin contain an amount
of iodin, in combination as an iodate, which corresponds
in iodin content to 15 grains of potassium iodid. But the
analysis shows that the product does not contain any
iodate whatever, and that the amount of iodin contained in
it is sufficient to account for only 1⁄4 grain of potassium
iodid in each 20 grains of saliodin.

Reproduction (much reduced) of a paragraph in the advertising
pamphlet on Saliodin. Note the twenty-one indications for Saliodin.
Lest some condition might be overlooked, we are advised to use it
“internally and externally.” Isn’t this scientific therapy?
COMMENTS
The above report is published simply as another example
of the “ethical proprietaries” that physicians are asked to
prescribe. It is not unique. It is neither better nor worse
than hundreds of others.
To show what absurdities appear in the “literature” (?)
that is sent to physicians, we reproduce a paragraph from
an advertising pamphlet. The promoters’ statement as to
the composition of the product is absurd, but not more so
than are the claims made for it as a therapeutic agent.
There is not a “patent medicine” on the market for which
any more blatant, extravagant and ridiculous claims are
made.
The manner of exploiting saliodin is another illustration
of the tendency on the part of nostrum-makers to advertise
their wares through pseudo-scientific articles published in
a certain class of medical journals. In the pamphlet sent
out by the Saliodin Company appears a reprint of an article
from the Philadelphia Medical Summary of February, 1905.
It is entitled “A Similarity in the Etiologic Factors of
Rheumatism and Malaria,” and was written by J. C. Denston,
M.D. In it occurs this statement: “The manufacturers
(of saliodin) publish their formula and, I think, distribute
samples and literature on request.” The charming ingenuousness
of this statement is fully realized when it is understood
that J. C. Denston is the president of the Saliodin
company. This is also another illustration of what is now
a common occurrence, viz.: men who are engaged in manufacturing
proprietary products and who have an M.D. degree
use that degree as a commercial asset, and by this means
the average reader is led to think that articles written by
them in praise of their own products are spontaneous
tributes from practicing physicians.—(From The Journal
A. M. A., Oct. 26, 1907.)
THEOBROMIN SODIUM SALICYLATE VERSUS
“DIURETIN”: THE ECONOMICAL ASPECT
W. A. Puckner and Paul N. Leech
The following inquiry is from Dr. Reid Hunt, recently
appointed professor of pharmacology at Harvard Medical
School:
“Have you ever made an examination of the theobromin
sodium salicylates on the market to determine if they are
identical with ‘Diuretin?’ The description of theobromin
sodium salicylate in New and Nonofficial Remedies agrees
with the statements as to the composition of Diuretin, but I
wondered if, at times at least, the theobromin sodium salicylate
on the market might be a simple mixture of theobromin
and sodium salicylate just as the Caffeinae Sodio-Salicylas,
N. F., seems to be a simple mixture. Diuretin is
quoted in current price-lists at $1.75 an ounce, whereas the
price of theobromin sodium salicylate is only 35 cents an
ounce. Many hospitals use diuretin, and both physicians
and students often have only hazy ideas as to what it is.
If the preparations of theobromin sodium salicylate now on
the market are identical with Diuretin they should certainly
be used, not only because they are less expensive, but
because the descriptive name will continually remind the
physicians of what they are using.”
Theobromin for some time has been regarded as a valuable
therapeutic agent. The obstacle to its use has been its
insolubility and the consequent uncertainty of the degree of
its absorption. For this reason a soluble salt of theobromin,
theobromin sodium salicylate, first introduced and advertised
under the proprietary name “Diuretin,” has come to be
used to a considerable extent.
Theobromin sodium salicylate—also called theobromin and
sodium salicylate—is prepared by interaction, in molecular
proportions, of theobromin, sodium hydroxid and sodium
salicylate, the theobromin first being treated with sodium
hydroxid in the presence of a suitable solvent, then the
sodium salicylate added and the whole brought to dryness.
The soluble compound which is formed is generally considered
to be a double salt of theobromin sodium and
sodium salicylate.
Theobromin sodium salicylate is described in New and
Nonofficial Remedies and in several foreign pharmacopeias.
It is also to be described in the forthcoming United States
Pharmacopeia.
Although the product is not controlled by patents of any
kind, and although it is offered for sale under its chemical
name by the leading chemical manufacturers at from 35 to
45 cents per ounce, the proprietary product, Diuretin, sells
for $1.75 an ounce. This is probably because the manufacturers
think that those who have been using it under its
chemically non-descriptive but therapeutically suggestive
title, Diuretin, will remain ignorant of the fact that the
same product is on the market under its chemical name.
In view of these conditions, emphasized by Dr. Hunt’s letter,
it was deemed important to examine the market-supply of
theobromin sodium salicylate and to compare the several
specimens with the proprietary brand Diuretin.
The following specimens, purchased in 1-ounce original
packages, were examined:
Diuretin, “Knoll,” Knoll & Co.
Theobromine and Sodium Salicylate, Mallinckrodt Chemical
Works.
Theobromine and Sodium Salicylate, Merck & Co.
Theobromine and Sodium Salicylate Powder, Powers-Weightman-Rosengarten
Co.
Theobromine and Sodium Salicylate, “Roche,” Hoffmann-La
Roche Chemical Works.
Theobromine and Sodium Salicylate, Squibb, E. R. Squibb
& Sons.
The theobromin content prescribed for theobromin sodium
salicylate by the various standards ranges from 45 to 50
per cent., that of New and Nonofficial Remedies being the
highest. A requirement of not less than 46.5 per cent. theobromin
in the dried powder has been proposed for the new
U. S. Pharmacopeia.
The methods of quantitative estimation laid down by the
various authorities are all very similar and consist, in the
main, of a determination of water, of the sodium hydroxid,
free and in combination with theobromin, and of the theobromin
itself. For the theobromin estimation the following
method was employed:
A weighed sample (about 2 gm) which had previously
been dried, under slightly reduced pressure, over sulphuric
acid, to constant weight, was dissolved in five times its
weight of warm water. Two drops of phenolphthalein were
added, and the solution titrated with normal hydrochloric
acid. To the neutral solution, 1 drop of 10 per cent. ammonium
hydroxid solution was added, and the mixture allowed
to stand, with occasional stirring, for three and one-half
hours at the temperature of 15 C. The precipitate was
filtered on a weighed Gooch crucible, washed with just ten
times the weight (of the original sample taken) of water
(temperature 15 C.) and the precipitate dried at from 100
to 104. To the weight obtained, a correction factor (proved
satisfactory by quantitative extraction experiments on the
filtrate) of 0.13 gm. was added, for every 2 grams of the
original sample taken.
The full details of the examination will be published in the
1914 Reports of the A. M. A. Chemical Laboratory. The
results of the examination have been abstracted and are
compiled in the accompanying table:
SUMMARY OF ANALYSIS
Column Headings:
2 = Physical Appearance*
3 = Gm. in 1-Ounce Bottle
4 = Price per Ounce
5 = Moisture, Per Cent.
6 = Alkalin. as NaOH on Dry Powder† Per Cent.
7 = Theobromin in Dry Powder† Per Cent.
8 = Theobromin in Orig. Specimen† Per Cent.
| 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| Diuretin | 3 Pure White | 28 | .5 | $1 | .75 | 0 | .01 | 10 | .44 | 48 | .61 | 48 | .61 |
| Theo. Sod. Sal. M. C. W | 3 Pure White | 27 | .5 | 0 | .35 | 1 | .89 | 9 | .95 | 46 | .11 | 45 | .24 |
| Theo. Sod. Sal. Merck | 1 Pure White | 29 | .0 | 0 | .35 | 0 | .48 | 10 | .38 | 47 | .87 | 47 | .58 |
| Theo. Sod. Sal. P. W. R. Co. | 2 Pure White | 29 | .1 | 0 | .35 | 2 | .46 | 10 | .30 | 47 | .57 | 46 | .39 |
| Theo. Sod. Sal. Roche | 3 Pink | 28 | .6 | 0 | .35 | 2 | .27 | 9 | .92 | 49 | .05 | 47 | .92 |
| Theo. Sod. Sal. Squibb | 1 Pure White | 26 | .8 | 0 | .45 | 0 | .39 | 9 | .97 | 46 | .82 | 46 | .63 |
* In this column, 1, 2 and 3 denote the following:
1. Quite crystalline, under microscope.
2. Fairly crystalline, under microscope.
3. Not crystalline, under microscope.
† Average of determinations.
While the results show some variation in the moisture content
and also in the actual theobromin content of the dried
specimens, the variation is unimportant. The products in
their original state (undried), as compared in relation to the
theobromin content (the highest percentage of theobromin
being 48.61, the lowest 45.24), reveal a variation of only
about 3 per cent.—a variation which is negligible in the
case of drugs such as theobromin.
From the preceding investigation, it is concluded that (1)
practically there is no difference between the non-proprietary
brands of “theobromin sodium salicylate” and “Diuretin;”
(2) the several specimens examined were not simple mixtures
of “theobromin” and “sodium salicylate”; (3) essentially
all the brands complied with the standards laid down
and can be rated as satisfactory; (4) “Diuretin,” though
sold at an exorbitant price, is not superior to the product
supplied under the descriptive term “theobromin sodium
salicylate,” and (5) “Diuretin” sells wholesale for $1.75 an
ounce, against 35 cents for the “theobromin sodium salicylate,”
and therefore its employment cannot be interpreted
otherwise than as a useless and unnecessary expense.—(From
The Journal A. M. A., April 4, 1914.)
UNGUENTINE
W. A. Puckner and A. H. Clark
Attention has been called at various times to the fact that
the value of a published “formula” to a proprietary remedy
is in direct ratio to the reliability of the manufacturer publishing
it. When medical journals first insisted on their
advertisers letting physicians know the contents of the
remedies they wished to sell them, medical literature reeked
with formulas—some of them of weird and wonderful design.
Since the advent of the Food and Drugs Act, which requires
that labels shall approximate truthfulness, and particularly
since the Council on Pharmacy and Chemistry has investigated
a number of proprietary remedies, the publication of
“formulas” is not so common.
Unguentine, manufactured by the Norwich Pharmacal Co.,
is one of those remedies whose advertisement for years
always included “a formula”; more recently, however, this
is not in evidence. In an advertisement which appeared
about ten years ago, the “formula” given is:
“Carbolic acid | 2 | per cent. |
“Ichthyol | 5 | per cent. |
“Alum | 15 to | 16 | per cent.” |
It was claimed that, by a special process of their own, the
manufacturers had eliminated most of the astringent properties
of the alum, rendering it non-irritant. It was also
stated that “the base of Unguentine is pure petrolatum.”
Later the manufacturers seem to have changed the composition
of their product, or at least the “formula” given in the
advertisements was changed. Thus it appeared:
“Alum | 15 | per cent. |
“Zinc oxid | 5 | per cent. |
“Carbolic acid | 2 | per cent. |
“Ichthyol | 5 | per cent. |
“Aromatics and antiseptic oils with specially prepared
petrolatum and animal fat base.” |
The introduction of zinc oxid, aromatic and antiseptic oils
and animal fat was a new feature. Somewhat later, and
particularly since the passage of the national Food and
Drugs Act, no formula or other statement regarding the
composition seems to have appeared in the advertisements
in the medical press. In the 1906 price-list (p. 170) the
following formula appears:
| “Unguentine represents: |
“Alum (non-irritating) | 15 | per cent. |
“Phenol | 2 | per cent. |
“Ichthyol | 5 | per cent. |
“Zinc oxid | 5 | per cent. |
“Aromatic and antiseptic oils, with especially prepared
petrolatum and purified animal fat.” |
In the price-list issued for 1908—after the Food and Drugs
Act went into effect—the following appears:
“Unguentine represents:
“Alum compound (non-irritating)
“Phenol,
“Ichthyol,
“Zinc oxid,
“Aromatic and antiseptic oils, with especially prepared
petrolatum and purified animal fat.”
Thus the proportions are omitted, and alum becomes “alum
compound,” whatever that may mean.
In view of the conflicting statements made by the Norwich
Pharmacal Company, in regard to their leading specialty,
Unguentine, and especially because much stress was laid on
the filing of their “guarantee” under the Food and Drugs
Act, it was decided to ascertain of what Unguentine really
consists.
From our analysis we conclude that Unguentine contains
not alum but aluminum acetate (small amounts of alum may
be present as impurities in the aluminum acetate), zinc oxid,
or more probably impure zinc carbonate, and that the entire
quantity of both does not exceed 5 per cent. It contains no
ichthyol, or if any but the merest traces, and less than 1
per cent. of phenol. The aromatic oils amount to not more
than approximately 1 per cent. in all. The ointment-base
is, in the main, petrolatum.
In Unguentine we have, therefore, another proprietary
“specialty,” regarding the composition of which indefinite,
false or misleading statements have been made—this irrespective
of protestation of honesty by the firm.—(From The
Journal A. M. A., March 27, 1909.)
URICEDIN
W. A. Puckner and A. H. Clark
In view of the results of investigations by Zernik of
Uricedin as sold in Germany, and because it is being advertised
to physicians in this country, an examination of this
product was made in the laboratory of the American Medical
Association. Zernik’s report shows how this remedy has
varied in its composition as put on the market in Germany.
From their analysis the authors find that Uricedin is not a
definite chemical compound as is claimed, but is a simple
mixture whose composition is approximately:
Sodium sulphate (anhydrous) | 61.52 | per cent. |
Sodium citrate (anhydrous) | 29.62 | per cent. |
Sodium chlorid | 2.13 | per cent. |
Citric acid (anhydrous) | 3.25 | per cent. |
Moisture | 2.53 | per cent. |
Undetermined | 0.95 | per cent. |
| ——— |
| 100.00 |
Uricedin, therefore, is not a definite chemical compound as
claimed, but a simple mixture which consists essentially of
sodium sulphate (dried Glauber salt) 2⁄3, and sodium
citrate 1⁄3. It is, therefore, a typical nostrum, and, as it
appears, one the composition of which is changed from time
to time to suit the whim of the manufacturer. The therapeutic
claims made for it are of the usual extravagant character.
According to a recent advertisement it is “used successfully
for Gouty Diathesis, urinary Calculi, Rheumatoid
Arthritis,” “useful in Migraine, Occipital Headache, Epilepsy,
Hay Fever, Asthma,” etc. If such a simple mixture
will do all that this one is claimed to do, let us use it,
but prescribe its ingredients under their proper names.
Such a mixture would cost only a few cents a pound, but this
nostrum is listed at $1.25 a bottle of five ounces, or probably
$1.75 at retail, and this for the benefit of its foreign
manufacturers and their agents.—(Abstracted from The
Journal A. M. A., Nov. 23, 1907.)
URISEPTIN
W. A. Puckner and W. S. Hilpert
“Uriseptin,” manufactured by the Gardner-Barada Chemical
Co. of Chicago and claimed to be a “urinary antiseptic,
uric acid solvent and diuretic,” was examined in the laboratory
of the American Medical Association to determine
to what extent the claims made for it are justified.
The preparation as purchased in the open market bears a
label which presents the claims of the manufacturers, emphasized
by the chemical analysis duly signed by an analyst and
attested by a notary. Accompanying is a reproduction of
part of the label.

Reduced photographic reproduction of part of the Uriseptin label.
Before the examination had extended very far it was found
that discrepancies existed between facts and claims, and by
the time the analysis was complete Uriseptin was found to
be in the same class as many other proprietary remedies
that have been discussed in these columns.
Our examination shows that the most misleading statement
is that concerning the “lithium-formaldehyd” compound
the presence of which is claimed, more or less directly,
by both the manufacturers and the analyst employed by the
manufacturers. Although the chemical properties of lithium
and formaldehyd indicate in themselves that the existence
of such a compound would be most improbable, yet considerable
time was spent in searching the chemical literature
for such a compound. Thorough search, however, demonstrated
that no such compound, nor any that even approximated
it, has been described.
The question then arose as to the form in which the
lithium and the formaldehyd are present. The statements
regarding its properties as a urinary antiseptic and the
fact that the preparation is said to liberate formaldehyd
slowly in the bladder point strongly to the presence of
hexamethylenamin.
Tests102 were applied to demonstrate whether the formaldehyd
was present as a lithium compound, and if not, whether
it existed in the form of hexamethylenamin. By these the
presence of hexamethylenamin was proved and the absence
of formaldehyd in other combinations demonstrated. This
fact alone shows that the preparation is deliberately marketed
under a false claim, and it shows further that the
analysis on the label is worthless. The quantitative method
of analysis demonstrated the presence of 5.51 gm. hexamethylenamin
per 100 c.c. (25.15 grains per fluidounce).
Besides the hexamethylenamin, Uriseptin contains lithium
and a benzoate. Concerning the latter nothing is said in the
analysis, whose worthlessness is again demonstrated. By
quantitative methods Uriseptin was found to contain lithium
and a benzoate in such proportions as would indicate that
the lithium and the benzoate radicle exist as lithium benzoate.
This fact is further indicated by the claims made for
the preparation regarding its properties as a uric acid solvent,
for which purpose lithium benzoate is often used.
Again, the demonstration that the formaldehyd present is
in combination as hexamethylenamin precluded any possible
chemical combination between lithium and formaldehyd and
adds another strong point in support of the conclusion that
the lithium and benzoic acid are in combination as lithium
benzoate.
CONCLUSION
By chemical analysis the active ingredients of Uriseptin are
shown to be hexamethylenamin, approximately 5.5 gm. per
100 c.c. (about 25 gr. to each fluidounce), and lithium
benzoate, approximately 0.70 gm. per 100 c.c. (about 11
grains to each fluidounce), neither of which compounds is
mentioned in the advertising matter on the label or in the
so-called “analysis” on the label. The statements concerning
the composition of Uriseptin are false and appear
to be a deliberate attempt to mislead physicians.
Comment.—Investigation of the various “patent” and
so-called “ethical proprietaries” advertised to the public and
to the medical profession shows that those that have any
value as therapeutic agents depend for that value on some
well known drug or drugs. Hence, while many proprietaries
have some virtue, the ingredients which are of any value
are so concealed by the coined and “near-scientific” names
applied to them that these drugs are usually unrecognizable.
The many and various acetanilid mixtures furnish examples
of this class of proprietaries. And now we find another
example in that much advertised nostrum, Uriseptin.
According to our chemists, the chief ingredients of
Uriseptin are hexamethylenamin and lithium benzoate.
Hexamethylenamin is a valuable so-called urinary antiseptic—probably
one of the best we have. It is a pity that
more physicians do not know the value of this drug in
and of itself; it is a common ingredient of many proprietaries,
and yet too seldom prescribed under its true name.
There is no reason for its being given in the form of a
nostrum; it requires no skill in compounding, for it is best
given in its powdered form, either in capsules or otherwise.
So that, like acetanilid, the old argument of the nostrum
men that the preparation needs skill in compounding will
not hold. If a physician wants to prescribe hexamethylenamin
let him prescribe it in its simplest and best form,
and thus know exactly what he is giving.
Lithium benzoate also has its rightful place in the materia
medica, but not hidden in a proprietary mixture to be prescribed
unknowingly. It is hard to conceive of any one
thing that operates more disastrously against scientific
therapeutics than the vicious practice of marketing under
proprietary names standard and valuable drugs, with their
identity purposely concealed. Yet how frequently it is done.
Well-known drugs of unquestioned worth are combined with
those that are little known and of doubtful value, or more
likely absolutely worthless, the mixture is put on the market
under a high-sounding name and it is exploited through
physicians as a panacea for all kinds of diseases.
In this, as in so many other instances, an “analysis” made
to order is given to lend an air of apparent respectability
and scientific standing to the preparation or to its exploiters,
with the object, of course, of misleading physicians into
thinking they are reading unbiased testimony. In addition,
the “literature” accompanying the preparation is usually a
jargon of pseudo-scientific verbiage put in to serve the same
purpose as the analysis—that of catching the careless
physician.
This state of affairs will continue just so long as the
medical profession will tolerate it—and no longer. So long
as members of our profession will prescribe proprietaries
on the statements of their owners—as to both their composition
and their therapeutic value—just so long will pseudo-chemical
and pseudopharmaceutical companies fatten at the
expense of the medical profession and to the detriment of
the public health.—(From The Journal A. M. A., Aug.
29, 1908.)
ZEMACOL
W. A. Puckner and W. S. Hilpert
Attention has been called to the vague and mysterious
statements regarding a preparation called Zemacol, manufactured
by the Norwich Pharmacal Co., Norwich, N. Y.
Because of the unsatisfactory statements regarding the composition
of the preparation, it was considered of sufficient
interest to make an analysis and determine its chemical
constituents. Accordingly specimens of the preparation
were obtained and examined.
The preparation Zemacol (Norwich Pharmacal Co.), as
found on the market, is a thick, pink, mucilaginous liquid,
highly perfumed and having besides a suggestion of a phenolic
odor. The bottle bears a label on which appear the
following statements:
“A colloidal emollient containing extract of the rete mucosum of
the healthy yearling lamb, combined with glycerin, salicylic acid and
other antiseptic and aromatic oils. Useful in eczema and diseases of
the integument where cell destruction is a prominent factor.”
In the advertising matter the following claims are made:
“An advance in animal therapy....”
“... increases the nutritive activity of the cell tissue of the
skin through the absorbable extract of the rete mucosum.”
“... clinical tests show its efficacy in both the so-called moist
and dry eczematous conditions of all parts of the cutaneous surfaces.”
“... rich in animal cells.”
Since nothing could be found in the literature regarding
the therapeutic action of an extract of the rete mucosum of
the sheep, it was thought possible that the statements on the
label were given simply as a vague and mysterious means of
indicating the presence of wool-fat (lanolin), and tests were
made to determine the presence or absence of the latter
substance. A substance was isolated from Zemacol which
had the physical properties of, and responded to some of
the chemical tests for, wool-fat; but it was found in such
small quantities as to indicate that it was not present as
an active constituent. Since there are no definite tests for
the detection of serums or animal extracts the presence or
absence of these could not be demonstrated. Further examination
indicated the presence of salicylic acid, a gummy
material, having the properties of tragacanth and glycerin.
It is practically free from inorganic matter. By distillation
a small quantity of oil was isolated, which possesses the
characteristic odor of the preparation.
Quantitative estimations103 indicated the presence of the
above-mentioned constituents in approximately the following
quantities:
| Per Cent. |
Gummy matter having the properties of tragacanth | 2.02 |
Salicylic acid | 0.67 |
Matter having the general properties of wool-fat (lanolin) | 0.20 |
Glycerin | 5.50 |
Volatile matter (water and alcohol) | 91.00 |
Aromatic oils and phenol-like bodies | Trace |
The results of the above analysis, together with advertising
matter regarding Zemacol, were submitted to Dr. William
Allen Pusey, professor of dermatology and clinical
dermatology, College of Physicians and Surgeons, Chicago,
and past chairman of the Section on Dermatology of the
American Medical Association, with the inquiry whether
or not there was any record of investigations regarding the
therapeutic value of an extract of the rete mucosum of the
sheep and whether in his opinion the claims made for
Zematol would be warranted. The following reply was
received:
“So far as I know, nobody ever thought of or proposed
the use of an extract of rete mucosum as a therapeutic
agent and if a serious suggestion of that sort had ever
been made I believe I would know it. I can conceive of no
service which such an extract could render and I think the
suggestion of it is a highly fantastic idea. From the
analysis which you furnish I should say that the mixture
described is substantially the ordinary 2 per cent. solution
of tragacanth in glycerin and water with a little antiseptic
added to keep it from decomposing. That is a commonly
known lotion, modifications of which are used in practically
every hospital as a hand lotion, and has no magical virtues
whatever. Incidentally, I should think it cost, aside from
the labor, about twenty cents a gallon to make it.”—(From
The Journal A. M. A., May 14, 1910.)
ZYME-OID
W. A. Puckner and W. S. Hilpert
Zyme-oid, manufactured by the Oxychlorine Chemical
Company of Chicago, is advertised as “a powerful gastro-intestinal
antiferment” which will “arrest and prevent bacterial
fermentation in any portion of the intestinal tract,
whether the media be acid or alkaline.” These extravagant
statements, like many others made regarding the properties
of zyme-oid, are very similar in character to those made
in the circulars accompanying the preparation oxychlorine,
manufactured by the same firm and exposed in The Journal,
July 6, 1907, page 54. (See page 147 of this book.)
As examples, several parallel statements help to show this
similarity. The formula (?) of oxychlorine, as expounded
on the label, is given in full, while in the case of zyme-oid
only a hint is given as to its composition, but still sufficient
to point to a similarity between the two:
| OXYCHLORINE | | ZYME-OID |
| “Oxychlorine is a tetraborate
of sodium and potassium
combined with oxychlorid of
boron, thus: (6NaKB4O7)
BOCl3.” |
|
“Zyme-Oid is a double borate
salt.” |
In the matter of claims for chemical stability the two
seem to be very closely allied:
| Oxychlorine is “a stable salt
under all conditions until
brought in contact with sub-oxygenated
organic matter.” |
|
Zyme-oid is “a product
which is stable enough for
keeping purposes, but which
readily yields nascent oxygen
in the presence of bacterial
products.” |
The therapeutic properties attributed to these sister
products are even more similar, for we find that:
| “Oxychlorine is adapted to
all morbid and abnormal fermentative
alimentary states.” |
|
“Zyme-oid is a powerful
gastrointestinal antiferment.” |
Many more statements and claims could be quoted to
show a similarity between, amounting almost to an identity
of, oxychlorine and zyme-oid.
With these facts in mind, the analysis of zyme-oid was
undertaken in order to compare it with the previously
examined oxychlorine and to determine to what extent the
claims made for zyme-oid are upheld by its composition.
The analysis indicated, as was expected, that zyme-oid
is essentially the same as oxychlorine as is shown in the
following, quoted from the report of the analysis of each:
| ANALYSIS OF OXYCHLORINE | | ANALYSIS OF ZYME-OID |
Potassium (K) | 12.26 | | Potassium (K) | 13.50 |
Sodium (Na) | 8.20 | | Sodium (Na) | 9.84 |
Chlorate (ClO3) | 25.32 | | Chlorate (ClO3) | 27.50 |
Nitrate (NO3) | 21.70 | | Nitrate (NO3) | 24.22 |
Boric acid anhydrid (B2O3) | 18.63 | | Boric acid anhydrid (B2O3) | 13.42 |
Water, calculated | 13.29 | | Water, calculated | 10.42 |
Assuming that the chlorate in zyme-oid is present as
potassium chlorate and the nitrate is present as sodium
nitrate, the figures obtained by analysis correspond to a
mixture approximately as follows:
Potassium chlorate (KClO3) | 40.43 |
Sodium Nitrate (NaNO3) | 33.22 |
Potassium tetraborate (K2B4O7) | 1.60 |
Sodium tetraborate (Na2B4O7) | 3.31 |
Boric acid | 21.14 |
From the results of the analysis and from the physical
properties of zyme-oid we conclude, just as was done in the
case of oxychlorine, that the preparation is not a definite
chemical compound, but is essentially a mixture of alkali
chlorate and nitrate with boric acid, probably produced by
fusing together the constituents.
COMMENT
An examination of the claims made for the firm’s two
products, while, as already proved, disclosing many points
of similarity, will also show one remarkable difference.
We refer to the skilful indefiniteness that pervades the
claims made for zyme-oid and which defies scientific refutation.
This verbal obscurity is becoming daily more common
in the “literature” of firms marketing nostrums. Since
the Council has analyzed many of the much-advertised
articles and proved the unreliability of the pseudo-scientific
claims made for them, the more cautious of the nostrum-mongers
have modified the matter descriptive of their
products. They have called to their aid the principle that
words were given to man to conceal thought rather than
to express it, and they have reduced equivocation to a
fine art. Wherever it was possible to put forward claims
by implication rather than by expression this has been done.
To substantiate further the claims made by the manufacturers
of zyme-oid for their product, a laboratory report
is brought in evidence. This report, which is written more
in the style of a peruna testimonial than that of a conservative
scientific statement, fails to verify the claim that
zyme-oid is a “double borate salt,” but confines itself to a
statement of its harmlessness and its anti-fermentative
properties. In passing, it seems regrettable that scientific
laboratories should, for a pecuniary consideration, be willing
to jeopardize their reputations by lending their names to the
furtherance of nostrum exploitation. The results of the
examination of zyme-oid demonstrate that the product is
no more worthy of the physician’s consideration than its
close, and equally worthless, relative, oxychlorine.—(From
The Journal A. M. A., May 23, 1908.)
PART III
CONTRIBUTIONS FROM THE JOURNAL:
NOSTRUMS
ALLEOTONE
The formula of this preparation, given in the literature,
reads as follows:
Alcoholici (Monatomic) | gr. | 1⁄1000 |
Quininæ Sulphatis | gr. | 1⁄384 |
Ac. Sulph. Dil. (10 per cent.) | gtt. | 21⁄2 |
Ac. Nitrici Dil. (10 per cent.) | gtt. | 1⁄77 |
Ac. Butanol-Dioic | gr. | 1⁄3 |
Tr. Ferri Chloridi | gtt. | 1⁄26 |
Aquæ | gtt. | xx |
The formula is worthless. It can only mislead and
mystify and the greater part of the literature is a mere
jumble of inaccurate and mystifying statements. The various
constituents of the preparation are taken up as follows.
The advertising literature states:
“Monatomic Alcohol is one of the constituents of all nerve tissue:
It is a product of the replacement of one atom of hydrogen of the
hydrocarbons by their hydroxyl group H.O.”
This information does not inform, since there is a vast
number of monatomic alcohols and of every description.
The assertion that the preparation “contains a salt” would be
perfectly analogous and just as enlightening. Of “Ferri
Chlo” the literature says:
“Ferri Chlo is found with all proteids and nucleins and herein acts
as magnetic iron, aiding the play of the electrical travel.”
The first assertion is untrue, for iron does not exist as
chlorid in the cells of the body, but as some organic iron
compound; neither is it found in all proteids, but principally
in nucleo-albumins; and not all proteids contain nucleo-albumins.
The assertion that the iron chlorid “acts as
magnetic iron aiding the play of the electric travel” is
nonsensical and on a par with the electric belt method
of exploitation, and suggests forcibly the class to which
Alleotone belongs. The literature further states:
“Sulphuric and nitric acids act in removing hydrogen atoms and
substitute atoms of the radical NO2; that is, as hydrogen tranquilizes
the speed of burning or oxidation, its action is substituted by the
atom nitrogen which is energy itself, nitrogen being the base of all
explosives.”
Sulphuric acid is certainly an oxidizing agent and in
virtue thereof removes hydrogen; but not in a solution
whose concentration with respect to sulphuric acid is
approximately only 0.82 per cent. The statement that
nitrogen is the “base of all explosives” is another example
of the methods of the promoters. As it is a well-known
fact, however, that nitrogen itself is one of the least
reactive of gaseous elements, little confidence can be placed
in such remarks as “Nitrogen which is energy itself.”
Another mystifying term used in the formula is “Ac.
Butanol-Dioic,” which is a true chemical name, certainly,
but it is one by which few physicians will recognize simple
malic acid, an ordinary vegetable acid widely distributed
in ripe fruits, such as apples and pears, and possessing
the properties simply of a relatively weak organic acid.
To describe it as exercising any potent influence “in the
oxidation of the phosphorus as lecithin in the cell”—especially
in the extremely low concentration in which it is
stated to exist in Alleotone—is simply an absurd juggling
with words. It is not much to be wondered at that the
public should be taken in by pseudoscientific “literature”;
but it is not only strange, it is discreditable to our profession,
that among its members should be found any to
accept such rubbish as the above quoted “literature” as information
worth acting on—yet such there are, judging from the
testimonials.—(Abstracted from The Journal A. M. A.,
Feb. 1, 1908.)
The Commercial Value of Adverse Criticism
For skilful attempts to convert a “knock” into a “boost,”
commend us to the discredited nostrum exploiter. The
federal Food and Drugs Act did much to bring out this
amiable quality—possibly developed it. While somewhat
ancient history, it is well to call to mind what happened when
the excise authorities insisted either that the “patent medicine”
booze, Peruna, have some medicine put in it, or else
that its manufacturers should go into the saloon business.
Hartman at once got out a new label stating that “for
a number of years a multitude of grateful friends” had
urged “that Peruna be given a slight laxative quality.”
Thenceforth the innocents and near-innocents could get their
perunaese jag only at the risk of a “bad quarter of an hour.”
One of the latest attempts to wriggle out of an uncomfortable
position, and at the same time make capital out of
the wriggling, is seen in the advertising of Alleotone, a
nostrum of the pseudoscientific type, which was shown up
in The Journal of Feb. 1, 1908. The “formula” furnished is
for the most part a jargon of misleading and mystifying
nonsense and fulfils the same purpose as the voluble “patter”
of the gentleman who is manipulating three shells and a
pea at the county fair.
Every constituent of the “formula” was discussed in
The Journal and the absurdities and impossibilities of
each dwelt on. Did the manufacturers of Alleotone feel
downcast over the exposure of their humbug? Not to
judge by their advertising, for they write to physicians that
“since the A. M. A. analyzed Alleotone it has made great
strides”—direction not specified. But the choicest piece of
impudence, and one that but for its dishonesty would be
laughable, is found in this portion of their advertising
pamphlet:
In the original, the words “With amendments suggested
in the Journal of the American Medical Association, Feb.
1, 1908,” and also “(Cholesterin.)” and “(Malic Acid.),”
which we have underscored in the illustration, are printed
in red and have been added to the original “formula.”
Such are the uses of adversity.
What claim, if any, the exploiter of this nostrum—B. F.
Copeland—has to medical or pharmaceutical knowledge,
we do not know. In fact, to be consistent with the
“ethics” of the nostrum business he need have none. Such
knowledge, indeed, tends to hamper that free play of the
imagination so necessary in this work. We understand that
he has at different times been in charge of a stave factory
and connected with a brokerage firm, which may exert some
subtle influence in developing the ability to relieve suffering
humanity, though the connection is not quite clear. One
would imagine, however, that the keen business instinct,
untrammeled by any considerations of conscience, which is
exhibited in the exploitation of Alleotone, would in purely
commercial pursuits have long since assured a competence.—(From
The Journal A. M. A., Oct. 17, 1908.)
BAUME ANALGESIQUE BENGUE
A physician writes asking for the formula of Baume
Analgésique Bengué. This product is another of the “patent-medicine”–“ethical-proprietary”
type of nostrums. In Great
Britain, it is advertised to the public as “A Wonderful
Remedy for Rheumatism, Gout, Neuralgia.” In this country,
the exploiters find that space in cheap medical journals,
reinforced by the aid of undiscriminating physicians, is a
cheaper method of getting the stuff to the public. According
to the statements of the manufacturers, Bengué’s Analgesic
Balm contains “menthol, salicylate of methyl and
lanolin.” When analyzed by the chemists of the British
Medical Association, it was reported to have the following
composition:
Menthol | 18 | per cent. |
Methyl salicylate | 20 | per cent. |
Lanolin, anhydrous | 54 | per cent. |
A fat, apparently lard | 8 | per cent. |
The estimated cost of the ingredients of a 50-cent tube of
Bengué’s Analgesic Balm, according to the British chemists,
is 21⁄2 cents. Evidently this imposingly named product is
practically a lanolin ointment containing oil of wintergreen
and menthol. Similar products are catalogued by various
pharmaceutical houses under various names and with varying
degrees of frankness concerning their composition. Two
firms give the medical profession full details regarding the
composition of their products: The H. K. Mulford Company,
who sell it under the name “Methyl Salicylate Ointment,”
and the Pitman-Myers Co., who name their product “Anodyne
Balm, P-M Co.” Some other firms are not so frank. Parke,
Davis & Co., for instance, sell “a combination of methyl
salicylate and menthol with a lanolin base” under the name
“Analgesic Balm,” but do not give the quantities of the
ingredients; Frederick Stearns & Co. sell “Analgesic Cream,
Stearns” without giving the quantities; Nelson Baker & Co.
sell “Anti-Neuralgic Ointment,” and no quantities are given.—(From
The Journal A. M. A., Dec. 14, 1912.)
ANTIDIABETICUM—BAUER
In Germany the makers of nostrums, their methods and
their products are systematically exposed by the Society for
the Suppression of Quackery (Deutsche Gesellschaft zur
Bekämpfung des Kurpfuschertums) through its publication,
the Geseundheitslehrer, under the aggressive editorship of Dr.
Kantor.
Ludwig Bauer,104 the manufacturer of “Antidiabeticum,”
inserted advertisements in daily papers asserting that for
his “humanitarian efforts” the society “Opera Educativa
pacifica” in Rome had granted him a diploma and placed his
publications in the celebrated “Bibliotheca Marciazzi.” Dr.
Kantor, editor of the Gesundheitslehrer, declared that,
according to information received from the German Consulate
in Rome, no such society existed there, and the library
referred to probably was the Bibliotheca Marciana in
Florence, which, like other public libraries, accepts all
donations without critical examination. To offset these
exposures, the promoter of Antidiabeticum published advertisements
libeling Dr. Kantor and attacking the Society for
Suppression of Quackery. This resulted in suits and counter-suits
for libel between Dr. Kantor and the directors of the
antiquackery society on the one side and the promoter of
Antidiabeticum on the other. As a result of the recent combined
trial, the court declared that Dr. Kantor’s charges
had been substantiated and the manufacturer of Antidiabeticum
was fined 600 marks or forty days’ imprisonment,
while apparently on purely technical grounds Dr. Kantor was
fined 50 marks or five days’ imprisonment. The costs were
divided between Bauer and Dr. Kantor in the proportion
of 11 to 1. As Bauer in the course of the trial made
further libelous charges, Dr. Kantor has lately started new
proceedings against Bauer. The incessant persecution of
Dr. Kantor was described in an editorial in The Journal,
May 20, 1911, p. 1486.
The persecution of Dr. Kantor previously described shows
no signs of abatement nor has Dr. Kantor given evidence
of loss of courage. Some of the German medical societies
have subscribed for the Gesundheitslehrer for each of their
members. It is written in popular style for the masses and
is a sharp and effective weapon for the campaign against
quackery.—(From The Journal A. M. A., April 27, 1912.)
ANTIKAMNIA
The Nostrum and Its Method of Exploitation
Our readers will be interested to learn some of the
remarkable properties which, according to the statements
of the manufacturers, this Antikamnia possesses. We quote
from the advertising literature:
The well-known nerve specialist (?), Dr. Harley, in an interview
published in the London Daily Express, says: “I have treated more than
one American for nervousness and ‘brain fag’ directly due to their
incessant energy. I had a young man in here this morning who complained
of headache ‘in the back of the neck.’ He was threatened with
congestion of the brain, and seemed somewhat aggrieved when I told
him he had been trying to do too much. I also treated a young
American woman who, since her arrival in London, had apparently
been living on Antikamnia tablets by the advice of her physician. It
was the only thing, she said, which kept her ‘braced up’ for the strain
of sight-seeing.”
(Why did the young woman consult this Dr. Harley—for
the drug habit?)
Note the following:
For the severe pains of rheumatism, dysmenorrhea, neuralgia, gout,
sciatica and lumbago, as well as for the lightning pains of locomotor
ataxia, there can be no quicker and more lasting relief obtained than
by the administration of Antikamnia and codeine tablets.
Imagine an intelligent physician trying to treat the diseases
mentioned below with the various impotent means of the
pharmacopeia and physiological therapy when he might
depend on Antikamnia! We quote again:
As a Pain Reliever.—In headache, cephalalgia, hemicrania, migraine
[some other words might have been thrown in so as still more to
emphasize the headache business], myalgia, coryza, la grippe and its
sequelae, the lightning pains of locomotor ataxia and all pains due to
irregular menstruation.
As an Anodyne or Sedative.—In alcoholic delirium, indigestion, cardialgia,
gastralgia, dyspepsia, hysteria, insomnia, inebriety, car-sickness,
sea-sickness, worry and sight-seer’s fatigue.
As an Antipyretic.—In typhoid, intermittent, puerperal and malarial
fevers, bronchitis, pneumonia, pleurisy, and tuberculosis.
As an Anti-Neuralgic.—In acute or chronic neuralgia, facial neuralgia,
earache, pain about the teeth, angina pectoris, neurasthenia, palpitation,
pains of locomotor ataxia and sciatica.
As an Anti-Rheumatic.—In acute or chronic rheumatism and gout,
fever and pleurodynia.
There is no remedy so useful and attended with such satisfactory
results as Antikamnia tablets in the treatment of melancholia with vasomotor
disturbances, anemic headaches, emotional distress, and active
delusions of apprehension and distrust. They increase arterial tension
and promote digestion, as well as being particularly serviceable in
relieving the persistent headache which accompanies nervousness.
In neurasthenia, in mild hysteroid affections, and in the various
neuralgias, particularly ovarian, and in the nervous tremor so often
seen in confirmed drunkards, they are of peculiar service. In angina
pectoris this drug has a beneficial action; it relieves the pain and
distress in many cases, even when amyl nitrite and nitroglycerin have
failed entirely. In pseudo-angina, frequently observed in hysterical
women, its action is all that can be desired.
Patients who suffer from irritable, weak, or palpitating heart, needing
at times a pain reliever, can take Antikamnia tablets, without untoward
after-effects, knowing that the heart is being fortified. In delirium
tremens, they relieve when there are great restlessness, insomnia, the
general lowering of the nerve power.
Only the vivid picture of a crisis in locomotor ataxia or the agony
of a true migraine, can impress the observer with the full value of
this pain reliever.
The following testimonials are from physicians:
Dr. Caleb Lyon, an old Bellevue practitioner, in referring
to antikamnia and codein tablets, says:
In my practice they accompany the maid from her virgin couch to
her lying-in chamber, assuaging the perplexities of maidenhood and
easing the trials of maternity with most gratifying results. I earnestly
hope that the proprietors of this valuable remedial agent will keep it
up to its present standard of purity and excellence.
Dr. Walter M. Fleming, A.M., M.D., New York City,
writes:
... With all the experience of more than a quarter of a century,
in the treatment of winter cough, and all its complications of laryngeal,
bronchial and pulmonary irritability, dyspnea, asthmatic spasms, and
finally whooping cough—usually the most persistent and tenacious of
all these membranous maladies—I find no one remedy more strongly
indicated, or which yields more prompt and satisfactory results than
Antikamnia and heroin tablets, composed of Antikamnia 5 grains and
heroin hydrochloride 1⁄12 grain.... Result: a prompt and efficient
expectorant, at once relaxing the harsh and rasping cough, releasing the
tenacious, sticky and gelatinous mucus which is soon readily expectorated,
while the soothing influence of the Antikamnia is at once
manifested, greatly to the comfort and contentment of the patient.
... Independent of the fact of the direct applicability of this
remedy to the various membranous maladies of the lungs, bronchi,
fauces and nose, it proves also, an invariable remedy in all febrile cases
where anodyne is required. This, together with its analgesic and antipyretic
merits, eminently qualify this combination for a responsive agent
in the treatment of nearly all the numerous febrile attacks characterized
by pain, nervousness, insomnia and their accompanying symptoms.
“Antikamnia and Quinin”
If there is any virtue in the particular combination known
as “Antikamnia,” a physician prescribing the tablets supposed
to contain combinations of “Antikamnia” and some other
drugs should have some guarantee that they contain those
remedies. Take, for example, the tablets advertised and
sold as “Antikamnia and quinin.” It might reasonably be
supposed that the tablets contained the combination known
as “Antikamnia”; this, however, seems not to be the case.
Previous analyses, as published105 by us, have shown that antikamnia
contains approximately 20 per cent. of sodium
bicarbonate, yet two chemists, working separately, have been
unable to find this ingredient in the tablets advertised and
sold as “Antikamnia and quinin.” Are we to understand,
therefore, that the manufacturers do not consider the bicarbonate
of sodium of importance in their preparation, Antikamnia;
or are they guilty of misrepresentation and of misleading
physicians in omitting this constituent from their
product Antikamnia when that is combined with the bisulphate
of quinin? The above statement regarding the
omission of bicarbonate of sodium from the quinin combination
may be verified by any physician who desires to
make a few simple chemical tests—carbonic acid is not given
off when the tablets are treated with dilute acids, as would
be the case if sodium bicarbonate were present. Further,
while the ordinary Antikamnia contains no constituent not
soluble either in water or in chloroform, and while quinin
bisulphate is readily soluble in water, the tablets said to contain
Antikamnia and quinin bisulphate, when treated successively
with water and with chloroform, leave a residue of
more than 18 per cent.
One of the chemists who analyzed the preparation for us,
in commenting on this in a letter, says: “The matter which
is insoluble in water, alcohol or in chloroform, i. e., the
substance which is neither ‘Antikamnia’ nor quinin bisulphate,
amounts to more than 18 per cent. in ‘Antikamnia
and quinin bisulphate tablets.’ The tablets weigh close to
five grains and are said to contain 2.5 grains each of
Antikamnia and quinin bisulphate. How is this possible
when each tablet contains almost one grain of foreign substance
(chiefly starch)?”
Further comment is superfluous. We have presented facts
to our readers and leave, them to draw their own conclusions.—(From
The Journal A. M. A., July 1, 1905.)
Adding Insult to Injury
When the Council on Pharmacy and Chemistry began its
work of independent and scientific investigation of proprietary
preparations, some of the questions asked were:
“What guarantee has the medical profession that the
formulas of these proprietary medicines are not changed
at the will of the manufacturers? How can the physician who
confidingly prescribes them for his patients know that the
preparation which he orders to-day is the same as that
which was furnished him last year, or which may be given
him next year, under the same name?”
At once a wail, as of injured innocence, went up from
countless venders of proprietary medicines, who replied
with one voice:
“The honor and reputation of the proprietors and manufacturers
is sufficient guarantee of the stability and permanence
of these preparations.”
So vehement were their protestations and so well simulated
were their declarations of Pecksniffian virtue that many
physicians were deceived thereby. Many medical journals
(whose views were, perhaps, slightly biased by the consideration
of fat advertising contracts) also were apparently
convinced. But the fact was overlooked that guarantees
based on honor are of value only in proportion to the amount
and quality of honor possessed by the guarantors.
The enactment of the national Food and Drugs Act is
bringing many things to light. Some of them are interesting,
some would be amusing were they not so utterly despicable.
Among other things, it has furnished a demonstration
of the value of the “honorable assurances” of nostrum
venders.
The nostrum Antikamnia has pointed out many a moral
in the campaign in the last two years. It was hardly to
be hoped that it would deliberately furnish a demonstration
of the utter lack of honesty on the part of a certain class
of proprietary manufacturers. Yet, relying apparently on
the ignorance of the public and the long-continued lethargy
of the medical profession, its promoters have, in the last
few weeks, unwittingly convicted and stultified themselves.
When the pure food law went into effect, the proprietors
of this mixture found themselves in a sad dilemma; if they
labeled their mixture in accordance with the provisions of
the law they would have to admit that it contained acetanilid
and that the charges against them were true. Failing to
comply with the law, they must go out of business. The
latter alternative was not to be thought of. The profits
gained by selling, with the aid of careless or ignorant physicians,
a five- or ten-cent mixture for $1 were too great
to be surrendered without a struggle. The same brilliant
intellect, perhaps, that first saw the commercial possibilities
in the business said: “Change the formula. Phenacetin is
about as cheap as acetanilid; the patent has just expired
and consequently we can get it at a low price. Let us
substitute phenacetin for acetanilid.”
As a result the profession is treated to an edifying
exhibition of virtue triumphant, a wolf so completely covered
by the harmless coat of a sheep that he flatters himself
that his wolfish nature is completely concealed. No longer
are skulls and skeletons sent out in calendar form as
grinning advance agents to be displayed in every doctor’s
office, but instead a beautiful domestic scene, showing a convalescent
child nestling in the arms of its mother. The
familiar “AK,” however, as usual, is in the lower right-hand
corner. And what a change in labels! No longer is
Antikamnia a chemical entity, but the label now openly
but ingenuously declares that “Antikamnia tablets in this
original package contain 350 grains of acetphenetidin, U. S.
P., per ounce. Guaranteed under the Food and Drugs Act,
June 30, 1906. Serial No. 10.” While, below, as an entirely
unnecessary display of conformity to the Pure Food Act,
appears this statement:
The Antikamnia tablets in this original ounce package contain no
acetanilid, antifebrin, antipyrin, alcohol, morphin, opium, codein, heroin,
cocain, alpha- or beta-eucain, arsenic, strychnin, chloroform, cannabis
indica or chloral hydrate.
Truly, Satan is appearing as an angel of light. What a
gratification it is to the long exploited profession to know
that Antikamnia contains no alcohol, no chloroform, no
cannabis indica, no chloral hydrate. How unfortunate that
this spontaneous display of confidence is not carried far
enough to inform the profession of the ingredients, aside
from phenacetin, contained in the mixture!
The label is an admission that the nostrum does not contain
what it was never supposed to contain, with the exception
of acetanilid, and is directly an attempt to conceal the real
contents. The proprietors know that the dear public, whose
“pains, headaches, neuralgias, women’s aches and ills, grippal
neuroses, nervousness, insomnia, rheumatism, lightning pains
of locomotor ataxia, sciatica, etc.,” they are longing to
assuage, will not know that acetphenetidin is the official designation
for what is popularly known as phenacetin, and that
this dangerous product is found in the new mixture in the
proportion of approximately 4 grains to a 5-grain tablet.
Evidently they also presume considerably on the ignorance
of our profession, or why should they make the brazen
statement that four grains of phenacetin is the “most reliable
remedy” for the long list of diseases enumerated on their
advertising calendar?

A reduced reproduction of a full-page Antikamnia advertisement
appearing in the New York World Almanac, 1911.
When the formula for which such wonderful virtues were
claimed was suddenly thrown overboard, was the medical
profession, which by its short-sighted patronage had built
up this business, notified in any way of the change? Search
the new advertising matter of this nostrum from beginning
to end and you will not find one word to show that
“The Antikamnia tablets in this original ounce package”
differ in the slightest particular from those sold to the
profession and the public for years past. This being true
(and the statements of the promoters themselves are our
authority for it), what remains of the pratings of “honor”
and the “guarantee of the manufacturers”? Has a physician
no right to know when a change is made in the formula of
a preparation which he has been prescribing for years?
What assurance has the profession that, at any moment,
a cheaper or more dangerous drug may not be substituted
for “acetphenetidin” if thereby the law can be evaded or
the profits of the delectable business enhanced?
How can any conscientious physician prescribe, for those
who confide their lives to his care, a preparation the stability
of the formula of which must depend absolutely on its
owner’s whim?
How can a physician with the slightest sense of responsibility
to his patients allow his office to be used as a free
advertising bureau for a preparation manifestly founded
and developed on deceit and misrepresentation?
How can any medical journal, except those avowedly and
unblushingly seeking to aid the nostrum maker to exploit
the profession, whose interests they claim to serve, continue
to carry the deceptive and misleading advertisement of a
twice exposed fraud?
How can any physician with a particle of self-respect or
manhood continue to support, by subscription or contribution,
any medical journal which, by accepting such advertising,
allies itself with the army of deceit and chicanery?—(Abstracted
from The Journal A. M. A., Jan. 26, 1907.)
Still Further Duplicity
When the Food and Drugs Act went into effect the
manufacturers of this preparation, instead of continuing
to put out the same mixture as they had been doing radically
changed the composition by substituting acetphenetidin
(phenacetin) for acetanilid. By doing this the company
avoided the disagreeable necessity for acknowledging on the
label that the nostrum contained acetanilid, as was shown
by the analysis published in The Journal, June 3, 1905.
In addition to stating that the package of Antikamnia
contained acetphenetidin, the company also stated that it
contained no “acetanilid, antifebrin, antipyrin, alcohol,
morphin, opium, codein, heroin, cocain, strychnin, chloroform,
cannabis indica, or chloral hydrate.” Knowing that the
nostrum was being advertised in Great Britain and Canada
as well as in the United States, The Journal obtained some
Antikamnia from London, and it was analyzed in the Association’s
laboratory. As was suspected, the analysis showed
that Antikamnia as sold abroad has the same composition
now as it had in the United States before the Food and
Drugs Act went into force, viz.: Acetanilid, 67.75 per cent.;
caffein, 4.88 per cent., and citric acid and sodium bicarbonate,
by difference, 25.36 per cent. This corresponds with the
analysis previously made and published in The Journal,
June 3, 1905. The Antikamnia on the market in this country
was also analyzed and it was found to contain: acetphenetidin
(phenacetin), 72.05 per cent.; caffein, 13.95 per cent.; citric
acid and sodium bicarbonate, 14 per cent. The preparation
sold as “Antikamnia and Quinin” was also analyzed, and
it was found that starch had been substituted for the bicarbonate
of sodium which is found in the Antikamnia itself.
The details of the analyses are given with the following
comments: “The above are brief statements of bald facts.
Two of these should be emphasized: (1) When the Food
and Drugs Act went into force, January, 1907, the manufacturers
of Antikamnia, rather than acknowledge the truth
of the past—we can imagine no other reason—materially and
radically changed the composition of their preparation, and
did this without notifying the medical profession or intimating
in any way, so far as we can learn, that such
a change had been made. We have no doubt they believed
they had a right to do as they pleased with their own;
that it was nobody’s business but theirs what they did
with their own preparation, or how they changed it. As
they never had told physicians what it contained, there was
no reason why they should do so now. This is logical and
we cannot blame the manufacturers so long as the medical
profession is willing to be humbugged. (2) For the same
reason, we presume, they claim that they have a right
to continue to use acetanilid in the product for the foreign
market. The Food and Drugs Act applies only to the
United States, of course, and acetanilid being cheaper, why
not use it? What is the difference if one is more dangerous
than the other? The fact that the Antikamnia sold abroad
differs from that sold in this country some may say is
of no special interest to us. Still this fact is worth noting:
The dose of acetphenetidin—phenacetin—(71⁄2 grains) is
nearly double that of acetanilid (4 grains): one becoming
accustomed to a certain dosage of the nostrum as sold in
this country might, while abroad, unwittingly be led to take
a double dose of acetanilid.—(Abstracted from The Journal
A. M. A., Feb. 8, 1908.)
Samples, Form Letters and “Prescriptions” Sent to the Laity
To the Editor:—The enclosed “literature” is being sent
broadcast to the laity by the Antikamnia people and still
a great many of the physicians throughout the country are
prescribing the preparation thus advertised. Will the time
ever come when the medical fraternity will awaken to the
fact that it has been humbugged by a great many manufacturing
concerns? I certainly hope so.
J. W. DuVal, M.D., Wichita Falls, Texas.
Comment:—The “literature” referred to by our correspondent
consists of a form letter and a small pamphlet.
The letter was similar to the one reproduced herewith.
The pamphlet accompanying the letter is entitled “Practical
Prescriptions,” and contains a list of diseases and morbid
states arranged alphabetically from “Alcoholism,” “Asthma”
and “Backache,” to “Wind,” “Women’s Pains” and “Worry.”
For the one hundred and twenty-two conditions listed,
“Antikamnia,” “Antikamnia and Codein” or “Laxative Antikamnia
and Quinin” are prescribed, demonstrating that
the “prescriptions” are more “practical” than scientific.
In many respects the methods of the proprietors of
“headache powders” and “anti-pain pills” are less offensive
to one’s sense of professional decency than the course
pursued by the Antikamnia people. The former have at
least never recommended their products as “ethical proprietaries”;
they have not used medical men as their unpaid
agents; the claims made for their products have been no
more exaggerated; and they have not found it necessary,
from the requirements of the Food and Drugs Act, to
substitute acetphenetidin for acetanilid to avoid giving the
lie to their former claims.
As to the query propounded by our correspondent: We
are optimistic enough to believe that the time he longs
for is already here. The fact that the proprietors of nostrums
of the Antikamnia type are finding it necessary to advertise
to the laity is, in itself, evidence of the diminishing demand
for such products on the part of the medical profession.—(From
The Journal A. M. A., April 18, 1908.)
Antikamnia in America and Great Britain
The following letter from the Antikamnia Chemical Company
to The Journal was received about August 1, 1912:
“You have at various times represented in your Journal that the
Antikamnia sold in foreign countries, particularly in Great Britain,
has a different formula from the Antikamnia sold in the United States,
and you have also published alleged formulas of each to show wherein
they are supposed to differ.
“We hereby respectfully notify you that the Antikamnia formula is
the same for all countries, and the publication of any statements to
the effect that the formula of Antikamnia is different in Great Britain,
or any other foreign country, from that sold in the United States is
a libel, and will be prosecuted as such.”
On the receipt of this a letter was written to a correspondent
in London requesting him to purchase in the open
market a package of Antikamnia. This was done and the
original sealed package reached the Association’s laboratory
a few days ago. Careful analysis of this specimen shows
it to contain acetanilid but no acetphenetidin, while the
Antikamnia sold in the United States contains acetphenetidin
but no acetanilid. The company’s protest to the contrary
notwithstanding, the formula of some Antikamnia, at least,
is still different in Great Britain from that sold in the
United States. It is possible, of course, that some time in the
future the composition of every package of this nostrum
on sale in the United Kingdom will be similar to that of
every package sold in the United States. It is even possible
that “Antikamnia & Quinin” tablets will—or do—actually
consist of quinin and the mixture called Antikamnia—although,
as The Journal has shown, this has not been the
case in the past. Since the patent expired on acetphenetidin,
this drug has become so cheap—it can be bought at wholesale
for less than 6 cents an ounce—that, commercially it
must make very little difference whether acetanilid or
acetphenetidin is used in the manufacture of Antikamnia.
But the question arises: Have our British confrères been
notified of the change in formula? A careful study of
the Antikamnia advertisements in English medical journals
shows that the British medical profession has been given
no more consideration by this concern than was the
American medical profession when the change in composition
was made on this side. But then why should it be?
Physicians, British or American, who are addicted to the
prescribing of secret proprietaries such as Antikamnia have
little need of formulas—“Theirs not to reason why!” The
medical profession on both sides of the Atlantic has never
known the exact composition of Antikamnia and does not
know it now. Physicians who call for preparations of the
Antikamnia type are prescribing names, not drugs.—(From
The Journal A. M. A., Oct. 26, 1912.)
Again, Antikamnia

Reproductions of portions of pages in the booklets sent out by the
Antikamnia Chemical Company to physicians (on the right), and laymen
(on the left), respectively. Those who do not realize the character of
the Antikamnia concern naturally imagine the quotation here given
from The Journal is a comparatively recent one. Notice that no dates
are given. As a matter of fact, it is twenty-two years old. Dr.
McIntyre, who wrote it, has been dead eleven years.
In season and out of season The Journal has exposed
the Antikamnia fraud until it would seem that its readers
would become weary of the very name. There is nothing
new to say about this dangerous stuff, and yet the number
of inquiries indicates that thousands of The Journal’s
readers do not know of the previous exposures. More than
fifteen years ago The Journal ceased carrying the Antikamnia
advertisement; more than ten years ago it notified its
readers that the nostrum was being advertised to the public
by means of circular letters; more than six years ago it
proved that, when the Food and Drugs Act went into effect,
acetphenetidin had been substituted for acetanilid in Antikamnia
evidently in order that the presence of the older
drug, of whose dangers the public had been made aware
might not have to be admitted; more than five years ago
The Journal showed that the Antikamnia sold in the British
Isles still contained acetanilid, and as late as last October
it verified this statement although threatened with prosecution
for libel by the Antikamnia Chemical Company.
Yet, in spite of all these exposures, not a week passes
that we do not receive one or more letters calling attention
to the Antikamnia fraud. Most of these letters deal
with one, or more, of three points: first, the fact that the
stuff is being advertised to the public by means of circular
letters and that sample “vest-pocket boxes” of this dangerous
drug are being sent through the mail to laymen; second,
that Antikamnia is being advertised in newspapers, and,
third, that in the booklets sent out by the Antikamnia
Chemical Company both to the medical profession and to the
public, a paragraph is quoted from an article by Dr. John
H. McIntyre that appeared in The Journal.

Photographic reproductions of two typical Antikamnia advertisements
now appearing in newspapers all over the country. These tablets are
advertised in various newspapers as being “safe” and neither “depressant”
nor “habit-forming”—three separate and distinct falsehoods.
The first two points have already been discussed so
frequently that it seems hardly worth while to take them
up again in detail, though it might be said that the medical
profession has at last become so familiar with this widespread
humbug that the Antikamnia Chemical Company
has finally gone over body and soul to the newspapers. So
far as we can learn only three publications professing to be
medical journals still carry the Antikamnia advertisement.
These three are:
| Southern Practitioner | Pacific Medical Journal |
| Therapeutic Record |
As is usual in such cases, the British medical journals
are not so particular, and we still find Antikamnia advertised
in:
| Medical Press and Circular | Lancet |
| Glasgow Medical Journal | Canada Lancet |
| Journal Tropical Medicine and Hygiene | Practitioner |
| Dublin Journal Medical Science |
The reproduction of the McIntyre quotation is evidently
adopted by the Antikamnia concern as a means of “playing
even” with The Journal for the unpleasant things it has
said about it. In quoting Dr. McIntyre, the Antikamnia
Chemical Company carefully avoids giving the date on which
the article appeared. As a matter of fact, the article was
printed in The Journal over twenty years ago (July 4, 1891),
and Dr. McIntyre himself has been dead for eleven years.
Presumably, however, the Antikamnia Chemical Company
will continue to mislead, either directly or by inference,
until the end of the chapter.—(From The Journal A. M. A.,
April 12, 1913.)
ANUSOL SUPPOSITORIES
“In Hemorrhoids and all Inflammatory Rectal Diseases,
let your first thought Continue to be Anusol Hemorrhoidal
Suppositories; they have Earned your lasting Confidence.”
Thus speaks an attractive folder recently sent to physicians.
With a prodigal use of superlatives, the medical profession
is told that these suppositories have for years “maintained
their World-Wide Reputation” as the “Most Effective, the
Safest ... the Most Economical and ... the Most Credit-Bringing
of all Topical Rectal Remedies.” The short
memory of the public is notorious; from the point of view
of the proprietary exploiter, the short memory of the medical
profession must be equally well known. How, otherwise,
would a firm try to make physicians believe that a product
had “earned” their “lasting confidence” when the result
of an examination by the Association’s chemists, published
in The Journal,106 had shown that Anusol Hemorrhoidal
Suppositories contained practically no “anusol.” Moreover,
as the Association’s findings were a practical verification
of the findings of a foreign chemist who also had failed to
find any “anusol” in Anusol Suppositories, it is not quite
clear what is meant by the term “world-wide reputation.”
Incidentally, the observant physician will notice that the list
of the ingredients given on the Anusol Suppositories labels
of 1913 differ from those of the vintage of four years ago.
The label of the old boxes gave the ingredients thus:
Bismuth. iodo-resorcinsulfon (Anusol), Zinc oxydat. pur., Balsam
Peruv., Ol. cacao, Unguent cereum.
On the latest label, however, we find these ingredients
given:
“Bismuth oxyiodid and resorcinsulphonate with Zinc oxid and Balsam
Peru, incorporated in suitable base.”
What will the formula be four years hence?—(From The
Journal A. M. A., Oct. 11, 1913.)
ANUSOL SUPPOSITORIES
To the Editor:—In the “Propaganda for Reform” department
of the October 11 issue of The Journal, you published
a short notice on Anusol Suppositories. We desire to
correct the impression which your readers may have
received, viz.: that there is any actual difference between
Anusol Suppositories of the present and Anusol Suppositories
of the past. We wish, therefore, to state that the composition
of Anusol Suppositories has not been changed;
the only modification that we have made is a revision of the
label to the effect that the active medicinal ingredient of
the preparation is a mixture of bismuth oxyiodid and
bismuth resorcinsulphonate in place of bismuth iodoresorcin-sulphonate.
The latter was originally claimed by the manufacturers,
discovered to be doubtful by an investigation
in the laboratory of the American Medical Association, as
well as by one on the part of a foreign chemist, and
finally disproved to our satisfaction by an independent
investigation on our part. We feel that the remark “What
will the formula be four years hence?” will carry the
impression to your readers that the composition has frequently
been changed and is likely to be changed again,
and it is for this reason that we request the above correction
and an assurance to the contrary.
The statement in the note that “Anusol Suppositories
have been proved to contain no Anusol” is also likely to
create an entirely erroneous impression. We dropped the
use of the word “anusol,” as designating a definite chemical
substance more than two years ago, and changed all our
propaganda matter accordingly. Schering & Glatz.—(From
The Journal A. M. A., Jan. 31, 1914.)
ASPIRO-LITHINE
Aspiro-lithine is another comparatively new example of
the custom of proprietary manufacturers in putting forward
old drugs under a new name and with them bidding for
the favor of physicians. An inquiry has been received
concerning this mixture. It is prepared by McKesson &
Robbins and is said to contain in each tablet 5 grains of
acetysalicylic acid (aspirin) and 21⁄2 grains of acid citro-tartrate
of lithium. It is recommended for all the purposes
for which acetysalicylic acid is commonly used, and on
account of the lithium added is claimed to have much
greater virtues than either of these drugs alone or of both
combined.
We had hoped that the time had passed for reputable houses
to employ such time-worn methods, but probably they will
not stop so long as physicians encourage them by continuing
to use such preparations. Acetysalicylic acid is a
good drug, whose value is pretty well known. It is further
known that lithium salts do not possess any great medicinal
virtue. Just what acid citro-tartrate of lithium may be is
hard to tell, for chemistries do not recognize such a substance.
The name presumably is intended to hide the real
nature of the preparation.
But if there be any advantage in combining lithium salts
with acetysalicylic acid in a prescription, it is a simple
proposition and requires no great skill, either on the part
of the physician who writes the prescription or on the part
of the druggist who puts it up, and such mixtures as
aspiro-lithine, with the exaggerated claims made for them,
should be avoided in the physician’s prescribing.—(From
The Journal A. M. A., May 28, 1910.)
BELL-ANS (PA-PAY-ANS, BELL)[AI]
Another “Patent Medicine” Foisted on the Public Through
the Medical Profession
With such nostrums as Antikamnia and Resinol fresh
in mind as flagrant examples of “patent medicines” introduced
to the public through the medical profession, what
follows regarding Bell-ans, or, as it is better remembered
by physicians, Pa-pay-ans (Bell) will take on an added
interest. Briefly, Bell-ans is the new name of a tablet
that, according to chemists’ reports, is essentially charcoal,
baking soda and ginger, flavored with oil of wintergreen.
Its selling point, in the past at least, has been the alleged
presence of papain. This drug, Bell & Co. allege, is present
in their tablets and they claim is “the digestive principle
obtained by our own exclusive process from the fruit of
Carica papaya.” As long ago as 1909, the Council on
Pharmacy and Chemistry attempted to find papain present
in what was then called Pa-pay-ans (Bell) and to determine
the digestive power of the tablets but with negative results.
The efforts of other chemists were equally unavailing.
In January, 1914, Bell and Company changed the name
of the product “Pa-pay-ans (Bell)” to “Bell-ans.” As The
Journal remarked soon after, it seemed probable that, as the
name of a nostrum of this kind is the manufacturer’s most
valuable asset, the name was hardly changed, as was alleged,
for purely euphonious reasons. It seemed more likely that
as analyses had indicated there was not, and probably never
had been, any appreciable amount of papain in the product,
the change of name might be due to the fear that some day
the misleading name might bring the preparation in conflict
with the federal Food and Drugs Act.
For years physicians have realized that the methods of
exploitation of Pa-pay-ans (Bell) have been such as to make
the medical profession a vast free advertising agency for the
nostrum. So far as we know Pa-pay-ans (Bell) has never
been advertised in lay journals—newspapers, etc. Certain
medical journals, however, have, for a long time, carried
the advertisements of Pa-pay-ans (Bell)—and later of Bell-ans—while
Bell & Co. has been lavish in its distribution of
free samples, blotters and other advertising paraphernalia
direct to the medical profession.

Miniature facsimile of a letter received by a layman. It was accompanied
by a small box of Bell-ans. Two points are worth noting:
“... one hundred thousand physicians are now prescribing it”;
“any druggist will tell you” that it is perfectly harmless!
Now it seems Bell and Company are going direct to the
public by means of vest-pocket samples and letters. The
letter, a miniature facsimile of which we reproduce, is one
addressed to laymen and accompanies a vest-pocket box of
the nostrum.
Here are some of the things that Bell and Company claim
Bell-ans will do:
“It removes flatulence, vertigo, weakness and other symptoms of
indigestion quickly and pleasantly.”
“It relieves vomiting in pregnancy, alcoholism, seasickness and
cholera morbus....”
“To promote appetite, digestion and the elimination of toxic and
waste material prescribe the Bell-ans....”
“... prevent eruptions, nausea, vertigo, pain, etc. ...”
“... remove distention, pain, weakness, depression, etc. ...”
“There is no derangement of the digestive organs upon which the
proper dose of Bell-ans (Pa-pay-ans, Bell) will not act quickly, pleasantly
and favorably....”
There is no physician living who really believes such
claims as these! Yet on the medical profession rests the
responsibility for the exploitation of this nostrum. Those
medical journals which accept advertisements for things
of this kind are not so much to blame as those physicians
who unprotestingly tolerate the journals that do so.
Privately owned medical journals are published, usually,
as a commercial proposition; they reflect, to a greater or
less extent, the attitude of their readers, subscribers and
contributors. There are at least three medical journals
which carry the advertisements of Bell-ans. They are the
New York Medical Journal, the International Journal of
Surgery and the Massachusetts Medical Journal.
Bell-ans (Pa-pay-ans, Bell) possesses the virtues—and
they are few—and the limitations—and these are many—inherent
to a mixture of bicarbonate of soda, ginger
and charcoal. Any druggist could put up just as good a
remedy, and any physician could write a prescription for a
better one in those cases in which he might think it indicated.
The whole secret of the commercial success of
Bell-ans lies in the mystery of its composition and the
fraudulence of the claims that have been made for it.
The same tablets put out under a non-proprietary name, as
an open formula and with claims that were reasonable and
true, would have had practically no sale. The commercial
success of Bell-ans is a monument whose foundation rests
equally on the unscrupulousness of the manufacturer and on
the gullibility of a learned profession.—(From The Journal
A. M. A., Jan. 16, 1915.)
BIOSOL
Dr. A. N. Lakin, State Line, Ind., writes:
“Kindly advise me concerning Biosol. I am sending you
herewith a pamphlet describing this product. On the last
page note clinical report from Dr. Buchman of the Indiana
Medical Association and president of the Department of
Public Health, Fort Wayne, Ind.”
H. Hille, once of Heidelberg, now of Oak Park, Ill., having
reached the conclusion that mineral starvation is the
cause of all diseases, devoted his talents to finding a
remedy. He claims to have found it and calls it “Biosol.”
He published his discovery in a pamphlet entitled “Facts of
Modern Science,” and recently published an article in the
Medical Record giving his ideas on this mineral point of
view. Biosol is an indescribable mixture of alcohol, carbohydrates,
and many and various mineral bodies—ranging
all the way from sodium, potassium, calcium and magnesium
to silicon, copper, uranium and thorium—the amount
of each being in most cases extremely minute. It is said
to be a valuable food as well as medicine. A dose of this
food might keep a rabbit alive for several hours, and a
man who could stand the expense and escape death from
delirium tremens might live on three quarts of the mixture
per day. Human beings have little occasion to fear mineral
starvation, and may obviate whatever danger there may
be with a drink of milk. Like other living creatures, we
may be thankful that we are furnished in our own bodies
with a living bioplasm which can use the minerals of the
waters and the rocks and which has its own laboratory in
which to prepare organic compounds to suit its needs.—(From
The Journal A. M. A., March 8, 1913.)
BROMIN-IODIN COMPOUND
The Life-History of a Nostrum
A correspondent writes for information concerning a
remedy known as Bromin-Iodin Comp., which he says is
manufactured by the Bromin-Iodin Chemical Company, formerly
of Binghamton, N. Y., but now located in San
Diego, Calif. In The Journal for Feb. 5, 1898, appeared
an article by Dr. C. W. Ingraham, Binghamton, N. Y.,
entitled “Five Years’ Successful Experience with a Special
Mode of Treating Pulmonary Tuberculosis.” This “special
mode” of treatment consisted in using what Dr. Ingraham
called “bromin-iodin compound,” which he said had the
following formula:
Iodin | gr. | 1⁄2 |
Bromin | gr. | 1⁄14 |
Phosphorus | gr. | 1⁄100 |
Thymol | gr. | 2⁄3 |
Sterilized oil | fl. dr. | 1 |
This “hypodermic treatment of phthisis” was widely
advertised in the late nineties by the Bromin-Iodin Chemical
Co, Binghamton, N. Y., and was but one of the innumerable
“treatments” for pulmonary tuberculosis that have
risen, had their day and, more or less gracefully, retired.
It was first sold “to physicians only” for hypodermic administration.
In 1906, however, physicians were told by the
company that “if we find it impossible to secure your
cooperation ... we will be compelled to do business
with the druggists in your locality ...” Apparently
they found such cooperation impossible, because a leaflet
was issued to the laity and the statement was made that
they intended to advertise “all over North America in
publications of national and international circulation, as
well as in local newspapers ...” Naturally the laity
couldn’t be expected to administer this treatment by the
hypodermic method and it is not surprising to read that
“experiment has proved that the same solution can be taken
internally.” In addition to the advertising leaflet, the public
also was provided with a “pocket calendar good for
200 years” which contained numerous testimonials from physicians
laudatory of the “bromin-iodin” treatment. The
layman who received one of the leaflets was told that if
he was suffering from “asthma, bronchitis, colds, consumption,
coughs, eczema, goiter, hay fever, neuralgia, rheumatism ... also
constipation and kidney troubles,” and
his recovery was “not as rapid as it should be,” should,
moreover, his physician refuse to use the bromin-iodin
compound “it might not be a bad idea to discharge him”
and get a physician who would!
At the time this “treatment” was first tried by its
“inventor,” the results given in fifty cases were: First
stage, 90 per cent. cures; second stage, 50 per cent. cures;
third stage, no cures, but improvement in several cases;
this was in 1895. It now appears that this “treatment” has
after a period of “patent-medicine” exploitation come back
into the “ethical proprietary” field. Presumably a mixture
such as that represented by the “formula” did not lend
itself to administration by mouth; there was nothing to
do, therefore, but enlist the aid of “easy” physicians in
furthering its sale.—(From The Journal A. M. A., June 4,
1910.)
CALMINE
New Names for Old Drugs
“Calmine, the new Hypnotic,” is another example of the
ingenuity of the exploiters of proprietary preparations in
coining new names for old drugs and the recklessness with
which exploiters herald forth renamed remedies to the profession
and the public as new and wonderful discoveries.
This is what the promoters, sustained by a calm confidence
in the credulity of the profession, have to say:
In the medical circles throughout the country a good deal of interest
and even enthusiasm over this new hypnotic is noticeable. Very few
drug products have attracted so much attention as this one.
A really satisfactory hypnotic and sleep-inducer, which Calmine certainly
seems to be, has been awaited expectantly for many years. Of
course, we have always had agents of this sort—a new one has come
out at frequent intervals—but none of them have “filled the bill”;
they have been prescribed only because there was nothing better to
be had.
Now this new and wonderful discovery is nothing but Veronal-sodium
(sodium diethyl-barbiturate) under another
name. It is the sodium salt of the more or less favorably
known hypnotic, Veronal (diethyl-barbituric acid). It is
also sold as Medinal, and differs from Veronal only in that
the combination with sodium has made it more readibly
soluble, and thus, it is claimed, its absorption is more prompt.
Veronal is protected abroad by a trade-mark and in this
country by a patent, and this, undoubtedly, is responsible
for the introduction of this sodium salt under these fanciful
names, because Veronal could not be sold without infringing
on the patent. This in turn induced the manufacturers
of Veronal, in self-protection, also to put the sodium salt on
the market, and now we have it under the name of Calmine.
This probably is only the beginning; soon we may look for
it under a host of other names and the usual result will follow:
thoughtless physicians who have had poor results with
it under one name will try it under others. Or worse still,
physicians will thoughtlessly combine Veronal with Calmine
or with Medinal in the same prescription, thus giving a
dangerous dose.—(From The Journal A. M. A., Jan. 14,
1911.)
CAMPHENOL
Camphenol is made by Johnson & Johnson, New Brunswick,
N. J. Under the name of the article on the carton
appears the following formula: C10H16O─C6H4(CH3)OH═C6H5OH.
This formula consists of the chemical formulas
for camphor, cresol and phenol, written one after another,
and from this one would conclude that Camphenol is a compound
of camphor, phenol and cresol in molecular proportions.
Examination shows, however, that Camphenol is but
a modification of the well-known camphorated phenol (the
liquid produced when solid camphor and phenol are triturated
together). In Camphenol a part of the phenol, in the
camphorated phenol, has been replaced by cresol, and this
liquid has been diluted and emulsified with gelatin or some
similar substance and perfumed. In other words, this preparation
is an emulsion containing relatively small quantities
of cresol, phenol and camphor and is another illustration of
the attempts of would-be pharmaceutical houses to produce
new synthetics in the simplest manner possible—that of
writing the chemical formulas of the constituents of a
remedy in a way to indicate a chemical combination.—(From
The Journal A. M. A., Nov. 5, 1910.)
CHOLOGEN
The proprietary Chologen is interesting some of our readers
and several have sent us samples and literature. Dr.
Philip Marvel, Atlantic City, N. J., for example, writes:
“By the way, I am to-day sending you by mail a package
which the Council on Pharmacy and Chemistry may care to
tackle, or it may not. I shall not be insulted any way, but
since these chologen preparations are being used a good deal
by various globe trotters, who sometimes hook up for a short
stay here, I feel it might be of some interest to know ‘what
fools these mortals be’ and how much the profession is being
fooled with them.”
Chologen as a medical treatment for gall-stones has been
before the German public for a number of years, and it is
somewhat singular that so simple a method, which could
be easily prescribed by the physician if it had merit, should
exhibit such remarkable vitality in proprietary form in spite
of evidence going to show that it rests on erroneous principles.
The Council rejected it as an unscientific mixture.
The treatment is somewhat liberal, consisting of the use, in
varying successions, of three kinds of tablets: No. 1,
calomel and podophyllin; No. 2, calomel, and No. 3, calomel,
podophyllin, camphor and menthol. The proprietors tell us
that the treatment should be proceeded with in spite of disturbances,
such as diarrhea and pain in the abdomen, and
that it should be repeated regularly in intervals for some
years, so long as any trouble exists or recurrence is threatened.
“A course” of Chologen tablets should be taken two
or three times a year, No. 1 being given for ten days, then
Nos. 1 and 2 for forty days and No. 3 for ten days.
It is worthy of note that experimental work seems to have
been performed in the attempt to show that bile produced
by this remedy will cause the disintegration and solution of
gall-stones. Normal bile has a certain solvent action on
gall-stones, but calomel and podophyllin have no demonstrable
effect in increasing the amount of bile. We had imagined
that these facts were generally known.
It is somewhat discouraging to reflect that some physicians
entertain so low an estimate of their ability to prescribe such
well-known remedies as calomel and podophyllin that they
must use them in the fixed combinations provided by Dr.
Glaser. If the self-respecting physician does not consider
himself insulted by a proprietary manufacturer who presumes
to tell him how to use such well-known remedies, this is a
good sign that he needs to take a postgraduate course in
materia medica and elementary prescription-writing. We
feel that medical writers must be short of subjects when
they devote papers to the exploitation of proprietaries consisting
of these simple ingredients.—(From The Journal A. M. A.,
Feb. 1, 1913.)
HAGEE’S CORDIAL OF COD-LIVER OIL [AJ]
Fraud and Deception Connected with So-Called Cod-Liver
Oil Preparations
The introduction of cod-liver oil as a supposedly easily
assimilable nutrient and reconstructive was followed by its
extensive use in wasting diseases, especially in phthisis,
in the treatment of which it came to be considered almost
essential, as it was supposed to possess some mysterious
power different from that of other oils. Its unpalatable character
led to various devices to render it tasteless and make
it more acceptable to the stomach. Emulsions containing
the oil in mixture with other substances were put on the
market and served a useful purpose. But the oily nature,
imperfectly concealed, was disagreeable to many, and gradually
other preparations appeared which attempted to retain
the supposed therapeutic virtues of cod-liver oil while dispensing
with its disagreeable character. This attempt has
been carried to the extreme that in many of the cod-liver
oil preparations now on the market the oil has been entirely
eliminated and all that is left of the oil is the name. This
is a species of fraud which has been tolerated too long,
but which will be kept up so long as physicians are willing to
be duped. Some of these articles are said to “represent”
the oil and to possess all its virtues. Others are said to contain
oil, while still others are stated to contain “all the valuable
constituents.” What is the standard by which we may
determine the true value of these preparations and by which
we may determine whether or not we, and through us our
patients, are being humbugged?
A FOOD OR MEDICINE—WHICH?
Is cod-liver oil to be considered a food or a medicine? A
food, certainly. As a food its value will consist in the fats
it contains. These fats are more easily oxidizable and are
considered more digestible than other fats because of the
presence of compounds derived from the liver which favor
its emulsification and enable it to penetrate the mucous
membrane more easily than other fats. Aside from their
nutrient properties we have no evidence that the fats of
cod-liver oil possess any therapeutic value; if the oil possesses
therapeutic qualities they must reside in its non-fatty
constituents, and the activity of these non-fatty constituents
is not acknowledged by those who have investigated
them scientifically. Most pharmacologists believe that whatever
virtue there is in cod-liver oil depends on its qualities
as an easily assimilable fat.
On the whole, we must conclude with Cushny that “cod-liver
oil has not been shown to have any action apart from
that of an easily digested food, and its superiority to some
other fats and oils has not been satisfactorily established.”
If, then, the value of cod-liver oil depends on the presence
of fat as its nutritive constituent, the amount of fat a
preparation contains will determine the worth or worthlessness
of such a preparation; at all events, a preparation
claiming to represent cod-liver oil which does not contain
fat in some form is fraudulent.
HOW TO PROVE OR DISPROVE THE PRESENCE OF COD-LIVER OIL
Fats may be changed to fatty acids or to soaps, as occurs
under the influence of pancreatic juice in digestion, and still
retain their nutritive value, but it is not possible to manipulate
them in any way so that they are still valuable as food,
and yet do not respond to easily applied chemical tests which
demonstrate their fatty nature.
Any preparation of cod-liver oil in which fat or fatty acid
is not recognizable by proper tests is valueless as food,
since its food value depends on the amount of fat or fatty
acid present. An elementary knowledge of chemistry and the
application of a few simple tests will enable any physician
to learn for himself whether or not a preparation contains
fat or fatty acids.
The preparations claiming to “represent” cod-liver oil are
in liquid form, and if they contain oil it must be one of the
following forms:
1. An emulsion of the oil which may be miscible with
water, but from which the fat tends to separate and rise to
the top. In this form the fat can be seen as globules under
the microscope.
2. A solution, resulting from the saponification of the oil,
containing a soap which usually will be alkaline in reaction,
especially when mixed with water, and from which fatty
acids are separated as a precipitate when the solution
is acidified.
3. A solution of fatty acids. This will be acid in reaction
and will be precipitated by the addition of water, in which
the fatty acids are not soluble.
Hagee’s Cordial of Cod-Liver Oil
Hagee’s Cordial of Cod-Liver Oil Compound is said to
“represent 33 per cent. of pure Norwegian cod-liver oil,”
with other ingredients, in perfect solution. It is also claimed,
according to the advertising pamphlet, that “in this preparation
we have every beneficial constituent of the best and
purest Norwegian cod-liver oil.” Put to the above three
tests, however, Hagee’s cordial of cod-liver oil is not, 1,
an emulsion of cod-liver oil; 2, is not a saponification of cod-liver
oil; and, 3, does not contain fatty acids. It therefore
contains no cod-liver oil. The only nutrients in the mixture,
revealed by analysis, are sugar, alcohol and glycerin, none
of which is contained in cod-liver oil.
In this case the manufacturer misleads by the use of the
word “represents”; he is careful not to say “contains,”
although the average reader would not be apt to notice the
nice distinction. The manufacturer unwittingly admits that
it contains no oil when he says that it “contains everything
of value except the grease.” What else there is of value in
cod-liver oil besides the “grease” we do not know. Certainly,
if we estimate the value of the remedy by its nutrient properties,
it must be set down as practically worthless, if not
fraudulent, for although a mixture of sugar, alcohol and
glycerin does possess certain nutrient value, the materials
can be purchased for it far more cheaply in the open market.
It is evident that claims are made for this preparation which
cannot be substantiated.
Again, some of the so-called cod-liver oil preparations are
termed extracts of cod-liver oil, but are not in fact made
from the oil, but from the cod-livers instead. They are
preparations which, if honestly made, might be worthy of
trial, but they are improperly called “extracts” of cod-liver
oil, since they do not contain the fat, which is the active
constituent of the oil, but the extractives from the liver
which may or may not possess therapeutic virtues. So far as
we know, however, no satisfactory evidence is forthcoming
to indicate that such extractives have any therapeutic value.
The attempt to modify cod-liver oil for therapeutic purposes
may be pronounced a failure and the large variety
and extensive sale of these preparations appear to be owing
to the fact that physicians do not recall the ordinary facts
of chemistry and fail to apply simple tests with little
technical skill, but too readily accept as facts the statements
of the manufacturers.—(Modified from The Journal A. M. A.,
Oct. 13, 1906.)
WATERBURY’S COMPOUND ONCE MORE [AK]
Most of our readers will remember what The Journal has
published about a product that used to be sold as “Waterbury’s
Metabolized Cod-Liver Oil Compound.” Briefly, it
was shown by a report of the Council on Pharmacy and
Chemistry and a contribution from the Association’s laboratory,
that this “Cod-Liver Oil Compound” contained practically
no cod-liver oil! Later the federal government
declared the stuff misbranded.
The product is now sold under the name “Waterbury’s
Compound.” It was recently stated in this department that
“Waterbury’s Compound” was one of the proprietary preparations
advertised both in “display” form and also in the
form of an “original article,” in the Army and Navy Medical
Record—a fraudulent publication that offered its editorial
pages for sale. Physicians are now receiving from the Waterbury
Chemical Company a reprint of what purports to be an
editorial from the Army and Navy Medical Record entitled,
“One of America’s Most Valuable Preparations.” The preparation,
of course, is “Waterbury’s Compound.” The company
in sending out this reprint also reproduces on the
reverse side the title-heading of the Army and Navy Medical
Record. All of which goes to show that some concerns not
only do not mind being found in bad company, but seem
proud of it. By the way, we wonder whether those physicians
who are still prescribing this nostrum think they are
prescribing a preparation containing cod-liver oil!—(From
The Journal A. M. A., Nov. 15, 1913.)
COLLYRIUM-WYETH
“I should be glad of any information about Wyeth’s Collyrium
and would also like to know if the position taken by
this concern measures up to the requirements of the Council
on Pharmacy and Chemistry.”
This inquiry was received from a Boston physician, who
enclosed with his note the letter quoted below that he had
received from John Wyeth & Brother, Philadelphia, the
makers of the preparation in question:
“We have your letter of the 22d inst., in which you request us to
send to you formula for ‘Collyrium,’ and in reply thereto, beg to
advise, being a corporation you will, we think appreciate why we are
not at liberty to disclose the various formulas under which our preparations
are made. Such is the competition in the trade that secrecy in
this respect is a valuable asset.
“You will not for a moment think that we take this position through
any distrust of your discretion or good faith, but because we feel that
our duty to the stockholders of the company prohibits us from disclosing
our formulas.
“Let us assure you however, that the eyewash contains only the
simplest and most harmless remedies well known to the medical
profession.”
John Wyeth & Brother seek the patronage of the medical
profession and desire physicians to use their preparations,
but “being a corporation” they “are not at liberty to disclose
the various formulas” of these preparations. In other words,
they expect physicians of the country to prescribe “patent
medicines” of whose composition they must be ignorant and
to rely wholly on the word of John Wyeth & Brother as to
the innocuousness of these products.
The letter is an insult to the physician to whom it was
written. If physicians were not so apathetic in cases of this
kind, the corporation of John Wyeth & Brother would long
since have been forced “off the fence”—it would have become
either a “patent medicine” concern or would have confined
its activities to the manufacture of pharmaceutical products
and ethically exploited proprietaries. Now what about this
Collyrium-Wyeth? It was analyzed in the Chemical Laboratory
of the American Medical Association and the chemists
report:
“The specimen of Collyrium-Wyeth examined was a clear,
colorless liquid having a faint odor like benzaldehyd. Qualitative
tests demonstrated the presence of antipyrin, free
boric acid and sodium borate. Acetanilid, ammonium salts,
glycerin, nitrates, phosphoric acid and pyramidon were
absent. Such potent alkaloids as atropin, cocain, homatropin
and pilocarpin, which are often used in ocular surgery,
were not found. Preparations of goldenseal were not present.
Quantitive examination indicated that the composition
of the preparation examined is essentially as follows:
“Antipyrin | 0.41 | gm. |
“Sodium borate | 0.55 | gm. |
“Boric acid | 2.14 | gm. |
“Water (by difference) to make | 100.00 | c.c.” |
The secret of such a formula must indeed be a “valuable
asset!” We venture the assertion that if the medical profession
did its duty, the corporation of John Wyeth & Brother
would find that its “duty to the stockholders of the company”
constrained it to abandon secret “patent medicines”
and to confine its activities to a legitimate line of pharmaceutical
products. An examination of the firm’s pricelist
reveals but a very few secret-formula preparations of the
type represented by Collyrium, hence it would probably not
seriously damage the business of the firm either to eliminate
all such formulas from its pricelist or to enable the physician
to use them intelligently, if they deserve it.—(From The
Journal A. M. A., May 17, 1913.)
DIATUSSIN
Dr. I. Fleiss, New York, writes:
“Please state the value of Diatussin, of Bischoff & Co,
in pertussis. Since pertussis is such an intractable disease,
anything which promises improvement is apt to attract the
physician’s attention.”
According to an advertising circular, issued by E. Bischoff
& Co, purporting to be a “reprint from the Munich Medical
Weekly,” Diatussin is “... a dialysate of Herbæ Thymi
and Pinguiculae.” The latter is said to be known in the
Alps as “blue fatweed.” The only further information as to
the composition of this preparation is the statement that
“the dialysate of this blue fatweed is said by the manufacturer
to contain a proteolytic ferment.” The writer of the
article recounts how, after trying a host of remedies, he
finally had such success in the treatment of whooping-cough
that “... a whole procession of mothers with children
affected by whooping-cough came to me from a neighboring
village, only because several children from this place
had been quickly cured by the dialysate.” Nevertheless,
while the “procession of mothers” appears to have been
impressed by the virtues of Diatussin, the writer of the
article, rather modestly for contributors of this sort, admits
that “I am, of course, well aware, that the small number
of cases under my observation allows of no decisive conclusion;
it is only the object of these lines to interest a
wider circle in tests.”—(From The Journal A. M. A., May
17, 1913.)
ENTERONOL
The “Greatest Germicide Known to Science!”
This preparation is put on the market by the Enteronol
Company, Oswego, N. Y., which declares that Enteronol is
“the greatest antiseptic and germicide known to science,”
and that it “destroys the germs of typhoid fever, acute and
chronic diarrhea, dysentery, cholera infantum, cholera morbus,
summer complaint, Asiatic cholera, etc., within two
hours.” The formula furnished by the company reads as
follows: “Ipecac, sub. nit. bismuth, latalia rad., camphor,
lupulin, caffein and rheum.” The attention of the Council
on Pharmacy and Chemistry of the American Medical Association
was directed to this preparation by a correspondent
who had received a circular from the Enteronol Company.
He sent a dollar to the company asking for a sample
of “latalia rad.” that he might study the drug botanically,
as he was unfamiliar with it. He expected to receive by
return mail a sample of root or bark, but instead, he
received three boxes of Enteronol and the information that
as “latalia rad.” costs from $25 to $45 a pound the company
could not afford to send samples. In a circular letter sent
out by this company “latalia rad.” is said to grow on the
sides of the Himalaya Mountains in India, and that the
company is unable to obtain enough for its own use. This
statement is probably correct, and no one else could secure
the drug either. A sample of Enteronol was submitted
to Professor Day, of the University of Illinois, and to
Professor Kraemer of the Philadelphia College of Pharmacy.
Professor Day reports that he was “unable to find any mention
of the drug of ‘latalia rad.’ which is stated as one of
the ingredients of this preparation. I have searched the
usual works of reference on pharmacognosy without being
able to find any reference to a drug of this name. A microscopic
examination of the tablets shows the presence of
rhubarb and of ginger, but no lupulin, at least not in substance;
nor could I locate definitely any ipecac, also stated
to be one of the ingredients. Since ginger is not stated
to be one of the ingredients of the compound, it, perhaps,
may be the mysterious stranger ‘latalia rad.’ I was unable
to locate any of the ordinary astringent drugs, such as kino,
krameria, or nutgall.” The results of Professor Kraemer’s
examination were practically identical with those obtained
by Professor Day. A report from the chemical laboratory
of the American Medical Association states that as Professors
Kraemer and Day suggested the presence of alum,
tests were made for this substance. The analysis, details
of which are given, leads to the conclusion that alum is
the chief constituent of Enteronol. The report adds strongly
to the impression that “latalia rad.” is simply a ruse to catch
the unwary and trusting physician who lacks the time to
look into the botany of every new plant discovered, and who
is willing to trust the honesty of every manufacturer. Attention
is also directed to the fact that while bismuth and
caffein are mentioned as ingredients tests made in the
laboratory failed to discover either of these substances.
Since there is no lupulin, no ipecac, no caffein, no bismuth,
and possibly no “latalia rad.” one is forced to the conclusion
that the “formula” is meaningless and worthless, and that
it is simply used to satisfy the demand for formulas for
proprietary remedies. This is one more beautiful illustration
of the absurdity of accepting a preparation because
the “formula is on every package.”—(Abstracted from The
Journal A. M. A., March 21, 1908.)
An Invitation to The Journal to Humbug the Profession
The Journal has received a circular letter from the
Enteronol Company, in which the following liberal offer
is made:
“We are willing to take one-fourth or one-half page ‘ad’ in your
Journal for a year at the regular rate, on condition that you accept
payment therefor in our GUARANTEED 7 per cent., preferred stock
at par; or if you desire, in ENTERONOL at the net wholesale price
to physicians.”
Not that this offer is made exclusively to The Journal:
“A large number of medical journals have accepted the foregoing
proposition; many carrying this advertising for several years already.”
“Our company is cooperative; we paying no cash for advertising.
The company is owned principally by physicians, medical journals, and
druggists.”
The journals of which we have record that carry the
enteronol advertisement are: Kansas City Medical Record,
Milwaukee Medical Journal, Toledo Medical and Surgical
Reporter, Proctologist, Pediatrics, and the Atlanta Journal-Record
of Medicine. If the statements made by the Enteronol
Co. are true, we might infer that these journals are being
paid for advertising space either with “preferred stock”
or with the nostrum itself. As we have previously shown,
however, the veracity of the enteronol advertising matter
is by no means unimpeachable.
Enteronol, it will be remembered, was exposed in The
Journal, March 21, 1908. It is advertised as the “greatest
antiseptic and germicide known to science,” and possesses
(?) such remarkable power that it “destroys the germs of
typhoid fever, acute and chronic diarrhea, dysentery, cholera
infantum, cholera morbus, summer complaint, Asiatic cholera,
etc., within two hours.” “The original product is found only
high up on the sides of the loftiest mountains in the world—the
Himalayas of India.”
THE “LITERATURE” FORMULA
Of course, it has a “formula”:
| Ipecac | | Lupulin |
| Sub. nit bismuth | Latalia rad. | Caffein |
| Camphor | | Rheum |
This seems very open and above board, except as to
quantities, until one tries to find out what “latalia rad.”
is; then it is discovered that it is the “mysterious stranger”
of pharmacognosy. Experts to whom this “remedy” was
submitted were unable even to find mention of such a
drug or plant as “latalia rad.” Nor was this the only fake
found concerning the stuff; carefully conducted experiments
repeatedly carried out in the Association’s laboratory failed
to disclose even a trace of bismuth subnitrate or caffein.
These experiments did show, however, that the tablets contained
an amount of aluminum corresponding to over 25
per cent. of crystallized alum. This led to the conclusion
that alum, whose presence is not even hinted at in the
“formula,” is the chief constituent of enteronol and as a
corollary that the formula is meaningless and worthless.
THE LABEL FORMULA
There is a curious lack of coordination between the
“formula” as printed on the label and that given in the
“literature.” The Food and Drugs Act, it will be remembered,
makes lying on the label illegal, and therefore dangerous;
statements in advertising matter that does not accompany
the product, however, are not controlled by that law.
The “formula” in the “literature” we have already given;
the “formula” on the label gives the following ingredients:
| Ipecac | | Lupulin |
| Sub. nit. bismuth | Opium, 1⁄4 gr. | Caffein |
| Camphor | | Rheum |
Two things about this are worth noting: One is that the
name of the ingredient on which the manufacturer lays so
much stress—latalia rad., the mysterious Himalayan plant—is
absent from the label. This would seem to indicate that
what has already been intimated by The Journal—namely,
that latalia rad. is a figment of the imagination—is a fact.
The second noticeable thing about the label “formula,” as
distinct from the “formula” in the advertising matter, is
that on the label we find there is opium in the preparation.
Why is no mention made of the presence of this potent
drug in the advertising matter?
To determine how nearly the present statements made
by the Enteronol Company approximate truthfulness, our
chemists were asked to examine the nostrum as it is now
sold. Their report follows:
LABORATORY FINDINGS
An original package of enteronol tablets was purchased
on the open market and submitted to the Association
laboratory for examination. In general appearance, odor
and taste the new tablets are similar to those previously
examined. The formula for the old tablets was given as
“Ipecac, Sub. nit. bismuth, Latalia rad., Camphor, Lupulin,
Caffein, Rheum,” and is still used in the circular. But
the label on the trade package no longer mentions “latalia
rad.” Since the presence of “latalia rad.,” in the old tablets,
was questioned, and as new labels have ceased to display
the name, it was thought possible that caffein and bismuth
might now be constituents of enteronol, as the drugs are
still mentioned in the new formula on the label. Accordingly,
enteronol was examined chemically to verify the
statements on the label regarding the presence of caffein
and bismuth in the tablets.
The specimen submitted to the laboratory some time
ago was found to contain neither bismuth nor caffein.
By employing the same methods as were used before (the
usual tests for detecting caffein and bismuth), neither
caffein nor bismuth could be demonstrated. It is thus
evident that this new specimen of enteronol, the statement
on the label to the contrary notwithstanding, contains
neither bismuth nor caffein—at least, in appreciable
quantities.
One would think that the discrepancy between “formulas”
and facts would prove of interest to the stockholders of the
Enteronol Company, especially as we are told that the
policy of the company is to have “practical men as stockholders.”
We are informed:
“Therefore, we have physicians, advertising experts, printers, publishers,
engravers, boxmakers, lithographers, druggists, lawyers, traveling
salesmen, officers and men holding executive positions in various manufacturing
and commercial corporations, editors of medical publications,
bishops, clergymen and missionaries—men from all the fields particularly
valuable commercially for our great enterprise.”
Yet if the physician-stockholders do not care to concern
themselves about the composition of the nostrum from the
sale of which they derive dividends, it can hardly be expected
that the boxmakers or traveling salesmen will be interested.
STOCK FOR SALE
Medical journals are not alone in being invited to participate
in the exploitation of this nostrum, vide a circular
letter from the Enteronol Company addressed “To Investors”:
“We offer at par of $10 each, 1,000 shares of our Guaranteed 7 per
cent. Preferred Stock, cumulative dividends, payable quarterly ... Profits
on business done last year were 54 cents for every dollar
expended ... We guarantee absolute security for your investment.
Safer than a bank.” [Italics ours.—Ed.]
We are told that at present the Enteronol Company
manufactures two products: a castor-oil preparation, known
as fig-ol, and enteronol. Very shortly, however, the company
expects to “add seven equally efficient products.”
“The average cost to manufacture, ready to ship, a dollar’s worth of
these goods is less than ten cents.”
“In enteronol alone, the company has fortunes and the only thing
needed to bring tremendous results and dividends of 100 per cent. is
the proper amount of judicious advertising.”
Here are some samples of the judicious (?) advertising:
“One Christian missionary, the Rev. Paul Singh of Jubbulpore, India,
testifies that he cured thirteen severe cases of Asiatic Cholera with a
box containing less than thirty tablets” [of enteronol].
“Wm. F. Oldham, bishop of Southern Asia, writes us that enteronol
cured nine cases out of ten of Asiatic Cholera. Now just think of
India and China with their 800,000,000 people who are dying by the
thousands of a disease which we have the power to cure so easily.”
How like a discourse by that delightful character of
Mark Twain’s—the visionary Colonel Sellers—this reads.
As he said about his “Infallible, Imperial, Oriental Optic
Liniment”:
“Why in the Oriental countries ... every square mile of ground
upholds its thousands on thousands of struggling human creatures—and
every separate and individual devil of them’s got the ophthalmia.”
The prospective stockholder is told that an ordinary
business concern reaches the limit of financial possibilities
in a few years, but:
“Not so with the Enteronol Company—it is a mail-order business and
the world is its territory.”
Even so with Colonel Sellers’ “Optic Liniment”:
“... it’s a patent medicine whose field of operations is the
solid earth.”
And we are told elsewhere that “about four-fifths of the
outstanding stock is held by the medical profession alone”!
And this stuff is advertised in medical journals!!
We are sometimes in danger of being too optimistic
regarding the results of the propaganda for reform in
proprietary medicine. Cases like this act as a corrective.—(From
The Journal A. M. A., Nov. 20, 1909.)
EXPURGO (SANOL) ANTI-DIABETES
One More Fraudulent Nostrum for Diabetes
Expurgo Anti-Diabetes is sold and advertised in the
United States by the Expurgo Manufacturing Company,
Chicago. The concern is the United States branch of a
Canadian company, the Sanol Manufacturing Company, Ltd.,
Winnipeg, which sells its product in Canada under the name
of “Sanol Anti-Diabetes.” The parent company is said to
have been incorporated under the Manitoba laws in 1912
and to have for its officers and directors the following men:
Charles Beyer, President.
Frank Beyer, Vice-President.
Charles Bauer, Secretary-Treasurer and Manager.
The manager of the United States branch in Chicago
is said to be one E. M. von Amerongen.
The stuff is such an evident fraud that one would imagine
that even intelligent laymen could not be deceived by it.
Nevertheless medical journals both in the United States
and Canada have accepted advertisements for this preparation
and physicians—of a certain type—have been found to
give testimonials for it. The medical profession is circularized
widely by the concern and “write-ups” have
appeared in pseudomedical journals. Some of the claims
made for Expurgo Anti-Diabetes are:
“The only positive cure for Diabetes.”
“It never fails to effect a Cure in every case of this disease, in whatever
form it may present itself provided the patient has not reached
the last stages of the malady.”
“Expurgo Anti-Diabetes is the New Cure for this deadly affliction.”
“Diabetes is certainly curable by our new discovery—Expurgo Anti-Diabetes,
provided that the course of the disease has not progressed to
the extent that the vital organs are irreparably damaged.”
“... thanks to the discovery of Expurgo Anti-Diabetes, the cure
of this dread disease is no longer a matter of doubt.”
“With the exception of very advanced cases of Diabetes ... all
diabetes can be cured by Expurgo Anti-Diabetes.”
Such claims one would imagine would be more than
sufficient to make plain, even to the most uncritical of
physicians, the evident fraudulence of Expurgo Anti-Diabetes.
Nevertheless, the advertisements of this fraud have
appeared during 1913 in the following medical journals:
| Medical Times | Medical Review |
| Medical Brief | Therapeutic Record |
| Medical Summary | Medical Fortnightly |
| Buffalo Medical Journal | Indianapolis Medical Journal |
| Louisville Monthly Journal | Southwest Journal of Medicine and Surgery |
| Iowa Medical Journal | Western Canada Medical Journal |
| Canada Lancet | Dominion Medical Monthly |
| Detroit Medical Journal | Canadian Medical Association Journal |
| Medical Herald | Canadian Practitioner and Review |
| Medical Review of Reviews | Massachusetts Medical Journal |
| Medical Standard |
Physicians will recognize that, with but few exceptions,
most of these journals are utterly unrepresentative of
scientific medicine.
The Army and Navy Medical Record, shown in The
Journal recently as a journalistic fraud, contained an
editorial puff of Expurgo Anti-Diabetes. The fact that the
Expurgo Company reprints the “editorial” from the Army and
Navy Medical Record as a “voluntary and unsolicited reference”
and distributes it among physicians, indicates how rotten
are the props on which the superstructure of this fraud
rest.
Another alleged “voluntary and unsolicited reference”
used by the Expurgo Company is taken from the Therapeutic
Record of Louisville, Kentucky. The advertising pages of
the Therapeutic Record reek with frauds and it has more
than once given editorial endorsement to some of the frauds
that it advertises. The following enlightening letter is
alleged to have been written by the editor of the Therapeutic
Record to the Expurgo Company in February, 1913:
Gentlemen:—Your favor of February 14th came duly to hand. Let
me advise you to pay no earthly attention to the proceedings of the
Medical Society where your product was criticized. These people exert
no influence with the practical up-to-date element of the profession
and are doing you as they do others. Never fear—you will succeed—your
remedy is all right. No man can talk down a meritorious product.
I stand ready to help you in any way at any time.
With sincere regards, I am,
Robert C. Kenner, M.D.,
Editor, the Therapeutic Record.
This, it will be noticed, was written in February. Soon
thereafter the Therapeutic Record was carrying the Expurgo
advertisement, and the June, 1913, issue contained a puff on
Expurgo, entitled “A Contribution to the Medical Treatment
of Diabetes.” The quid pro quo is fairly evident.
THE ALLEGED FORMULA
The formula for this nostrum is never published, although
in some of the advertising matter it is claimed that it is
“at the disposal of physicians.” A physician wrote to the
Expurgo Manufacturing Company, asking for the formula.
He was told that the preparation was “exclusively derived
from the vegetable kingdom,” from which one may recognize
a family likeness to the “dope” put out by the immortal
Lydia Pinkham. Further, to copy the letter exactly:
“The ingredients of which Antidiabetes is composed are chiefly:
“fructus syzigii jambulani
“cortex syzigii jambulani
“flores Rosmarini
“fructus Anisi stellati
“Extr. fl. Colæ
“Extr. fl. Condurango
“Extr. fl. Chinæ spir. spiss.
“Extr. fl. Calami
“Extr. fl. Gentianæ.”
The recipient of this noncommittal and uninforming “formula”
again wrote the Expurgo Manufacturing Company,
asking for quantities. Evidently this nostrum concern considered
such a request a piece of impertinent inquisitiveness,
for it replied to the physician in these terms, given verbatim
et literatim:
“Dear Sir:—Yours of the 16th duly to hand. We note that you
state ‘... I do not like to be working in the dark, and you can
readily see that this is the case unless I know how much of each
ingredient I am giving....’
“In your letter of the 6th you asked for the composition, which
you promptly received. We would like to state that we are dealing
with about 600 Doctors. Some of them asked for the formula, which
they received. They are all very conscientious gentlemen and none
of them ever pretended ‘to work in the dark.’ You know furthermore
that none of these ingredients is harmful in any way and yet ‘work
in the dark.’ You know that if there were any harmful ingredients in
our preparations, we would expose ourselves to imprisonment. If you
are so anxious to know all about it, why do you not analyse our
medicine? This would enlighten you in your ‘perfect darkness.’ If you
want to deprive your patients and yourselves of the indisputable good
of our preparations, simply do not prescribe them. Why finally do
you not write to the Doctors whose names we gave, who know enough
to be able to enlighten those who need it.
Truly yours
The Expurgo Mfg. Co.,
C. M. v. Amerongen, Manager.
More than a year ago, a Wisconsin physician, himself a
sufferer from diabetes, wrote The Journal that for three
months he had been using Expurgo Anti-Diabetes which the
Expurgo people had sent him. He declared that the nostrum
had greatly reduced the percentage of sugar in his urine.
In its reply, The Journal asked him whether, in testing
his urine he had used portions of twenty-four hour specimens
or merely individual specimens. His attention was called
to the fact that most of the nostrums for diabetes are
diuretics which, by increasing the amount of urine passed,
give an apparent decrease in the amount of sugar excreted.
A few days later, the physician wrote again, stating that
he had committed the very error The Journal had suspected,
and reporting that an examination of a twenty-four-hour
specimen showed that the glucose-excretion, instead of being
diminished, actually increased. This matter was referred to
editorially in The Journal, Nov. 9, 1912, under the title,
“A Possible Fallacy in Testing Diabetic Urine.”
Specimens of Expurgo Anti-Diabetes were examined in
the Association’s laboratory and the chemist’s report follows:
LABORATORY REPORT

The letter head of the Expurgo Manufacturing Co. Note the claim
that Expurgo Anti-Diabetes is “the only positive cure for diabetes.”
And this stuff is foisted on the profession through the medical press!
“The specimen of Expurgo Anti-Diabetes (Sanol Anti-Diabetes)
examined, was a light-brown, opaque liquid,
having a faintly aromatic odor and bitter taste. The
specimen contained considerable amounts of brown, insoluble
residue resembling the deposits often found in fluid
extracts. The absence of ammonium salts, iodids, glycerin,
hexamethylenamin, of antipyrin, pyramidon and similar
substances and of such purgatives as aloes, frangula,
rhubarb, etc., was indicated. Potent alkaloids such as
aconitin, cocain, morphin and strychnin were not found.
Qualitative tests indicated the presence of traces of phosphates,
sulphates, reducing sugars, caffein and cinchona
alkaloids. Alcohol was present only in traces. Small
quantities of chlorids, sodium and a salicylate were found.
The residue on drying amounted to 2.9 gm. in each 100 c.c.
A determination of the salicylic acid indicated approximately
0.17 gm. in each 100 c.c., which is equivalent to
less than 0.2 gm. of sodium salicylate per 100 c.c. (about
1 grain to the ounce). Evidently the preparation contains
plant extractives in aqueous solution and small amounts
of sodium salicylate and sodium chlorid.”
Summed up, the chemist’s report shows that Expurgo
Anti-Diabetes is essentially a watery solution of plant
extractives with small quantities of sodium salicylate and
salt. The exploiters claim their stuff contains the fruit
and bark of jambul, rosemary, star anise and fluid extract
of calamus, cinchona, cola, condurango, and gentian. Since
fluidextracts in general are strongly alcoholic and since the
laboratory’s analysis shows that the preparation contains
only traces of alcohol, the fluidextracts of the various drugs,
if present at all, must be in an infinitesimal amount.
Jambul was in vogue as a remedy for diabetes about twenty
years ago. It was tried and found wanting, and has long
since been relegated to the therapeutic scrap heap. Sanol,
therefore, is but one more proprietary humbug, foisted on
the profession under fraudulent claims, and having for its
essential constituent a drug that has long been discarded by
scientific men and resurrected for the purposes of quackery.
Expurgo will probably be used by uncritical and unthinking
physicians and its existence will be artificially prolonged
through the venality of pseudo-medical journals. That the
medical profession should tolerate such an evident fraud
is not to its credit. There is no excuse, either moral or
otherwise, for a physician giving his patients nostrums of
whose composition he is ignorant, and that is what is done
whenever Expurgo Anti-Diabetes is prescribed.—(From The
Journal A. M. A., Jan. 24, 1914.)
FORMAMINT
The Profession to Be Worked Again
Formamint Tablets are widely advertised and extravagantly
exploited to the laity in Great Britain. Large and expensive
advertisements appear in the English magazines and newspapers
and the tablets are pushed under the most preposterous
claims. The preparation is put out, we understand, by the
same concern that exploits Sanatogen. The medical profession
of this country is now being circularized and advertisements
are appearing in medical journals. They already
appear in the Medical Record, New York Medical Journal
and American Journal of Clinical Medicine.
It seems then that this is another product which, for the
time being at least, is to be a “patent medicine” on the other
side of the Atlantic and an “ethical proprietary” on this.
Doubtless the distinction will be a temporary one and as
soon as American physicians have furnished the requisite
number of testimonials and have recommended it to a
sufficient number of their patients the advertisements will
be quietly dropped from the American medical journals
and the advertising pages of newspapers and magazines will
be called into service.—(From The Journal A. M. A., Jan.
27, 1912.)
The So-Called Germ-Killing Throat Tablet
Formamint tablets have recently been put on the American
market by the same concern that exploits Sanatogen, the
“food tonic” or “tonic food”—according to whether one reads
European or American newspapers. Formamint tablets are
being introduced to the American public by that cheapest of
all methods of advertising “patent medicines,” through the
medical profession. It is not advertised in American newspapers
or lay magazines—at present. For some years this
product has been advertised in newspapers and other periodicals
in Europe under such claims as the following:
“Formamint shields humanity against infectious disease.”
“Cures and prevents sore throat.”
“The dangers of infection from diseases like diphtheria, scarlet fever,
measles, tonsillitis, sore throat, mumps, etc., have now been reduced
to an absolute minimum. This is due to the discovery of Wulfing’s
Formamint—the ‘germ-killing throat tablet.’”
“Cleanses the mouth and throat from disease germs as easily and
rapidly as dirt is removed from the skin.”
“Formamint will certainly prevent diphtheria.”
“Quickly render the whole mouth and throat thoroughly antiseptic.”
“Formamint destroys these [diphtheria] germs so rapidly that when
a physician mixed a little Formamint with water and added it to the
germs taken from the throat of a patient dangerously ill with diphtheria
they were all killed within ten minutes.”
Such are some of the claims by which Formamint goes to
the European public. Doubtless it will be only a matter of
time when the required number of testimonials from American
physicians are forthcoming when we may expect to find
the newspapers of this country heralding through their advertising
pages the fact that Formamint is “recommended by
thousands of American physicians.” The medical journals
that are lending their pages to this preliminary advertising
campaign are the following:
| New York Medical Journal | American Journal of Clinical Medicine |
| Medical Record | Medical Review of Reviews |
| American Medicine |
How much longer will the medical profession permit itself
to be used as an unwitting agency for the exploitation of
“patent medicines”? The game has been worked so often
that it has become transparently thin. It is evidently not
worn out, however, or shrewd nostrum promoters would not
waste their time or money on it. That it should still be
considered workable is complimentary neither to the standard
of advertising ethics of medical journals that accept the
Formamint advertisements nor to the intelligence of the
members of the medical profession who will “fall for it.”—(The
Journal A. M. A., Feb. 24, 1912.)
GOMENOL
A correspondent sends some advertising matter on Gomenol
and calls attention to the number of diseases for which the
preparation is recommended:
Gomenol is apparently a volatile oil. It is a proprietary
said to come from France, and to be prepared from a
species of cajuput (Melaleuca viridiflora, Gaertn.). This
plant is closely related to the cajuput tree or swamp tea-tree
(Melaleuca leucodendron, Linné) from which the official oil
of cajuput is obtained. The oils from these two plants are
very similar in composition and presumably in therapeutic
properties. The oil of the first-named plant appears not to
be marketed except in the form of the proprietary, Gomenol.
It probably has no advantage over the official oil of cajuput,
while in the form of Gomenol it costs about four times as
much. The following are some of the claims made for
Gomenol in the advertising circulars. They need no comment.
“A real specific for suppurations and catarrh.... It immunizes
tissues, excites their vitality and favors the formation of new
cells....
“The least trace of Gomenol prevents the growth in vitro of the
streptococcus, the tuberculous bacillus and the gonococcus.”—(From
The Journal A. M. A., April 4, 1914.)
HEADACHE CURES[AL]
Harmful Effects of Acetanilid, Antipyrin and Acetphenetidin
The United States Department of Agriculture Bulletin107
No. 126, issued July 3, 1909, sets forth the results of an
investigation conducted by the Bureau of Chemistry with
regard to the harmful effects of acetanilid, antipyrin and
acetphenetidin. During recent years the use of these remedies
and preparations containing them by the people at
large, without the supervision of the physician, has increased
rapidly and investigation has shown that coincidently there
has been a marked increase in the number of cases of poisoning
reported, in the number of fatalities, and in the number
of instances of habitual use.
Since the passage of the Food and Drugs Act, June 30,
1906, the attention of the Department of Agriculture has
been directed to this subject, particularly in connection with
the branding of drug products containing one or more of
these agents, and an attempt has been made to obtain full
and reliable data with regard to their poisonous qualities
with the object of furnishing information to the public which
would enable them to understand that these remedies should
be employed with caution in the absence of reliable medical
advice.
The information obtained with regard to the number of
an inquiry addressed to medical practitioners in the United
States with regard to their personal experience with these
drugs; and, second, the study of the cases of poisoning
recorded in medical literature. Nearly a thousand letters,
each containing eighteen questions, were addressed by the
department to physicians throughout the country, the object
being to secure information which would represent as closely
as possible the conditions existing among the people at large
so far as the harmful effects of the drugs in question are
concerned. Four hundred replies were received.
The information obtained with regard to the number of
instances quoted in medical literature in which poisoning,
death, or habitual use has been known to result from the
administration of acetanilid, antipyrin, and acetphenetidin
is set forth in Section A of the accompanying table. The
information summarized in Section B is based on the data
submitted by physicians. Granting that the 525 physicians
who did not reply had no cases to report, the question may
profitably be asked, if 925 physicians have observed 814 cases
of poisoning by these drugs, 28 deaths which are attributed
to their use, and 136 instances of habitual use, how many
such cases have in all probability been observed by the
125,000 physicians scattered throughout the United States?
The summary, C, includes both the number of cases recorded
in medical literature and those reported by physicians.
POISONING BY ACETANILID, ANTIPYRIN AND PHENACETIN
A.—Cases Recorded in Medical Literature
| POISONING. | DEATH. | HABITUAL
USE. |
| Acetanilid | 297 | 13 | 32 |
| Antipyrin | 488 | 10 | .. |
| Acetphenetidin | 70 | 3 | 1 |
| —— | — | — |
| Total | 855 | 26 | 33 |
B.—Data Submitted by Physicians
| POISONING. | DEATH. | HABITUAL
USE. |
| Acetanilid | 614 | 16 | 112 |
| Antipyrin | 105 | 5 | 7 |
| Acetphenetidin | 95 | 7 | 17 |
| —— | — | —— |
| Total | 814 | 28 | 136 |
C.—Total Number of Cases
| POISONING. | DEATH. | HABITUAL
USE. |
| Acetanilid | 911 | 29 | 144 |
| Antipyrin | 593 | 15 | 7 |
| Acetphenetidin | 165 | 10 | 18 |
| ——— | — | —— |
| Total | 1,669 | 54 | 169 |
The bulletin contains information with regard to dosage,
the extent to which these drugs are employed by physicians,
poisoning and habitual use, the nature of the ill effects
produced, etc. It also contains references to the recorded
cases of poisoning, together with a brief abstract of each
case.—(From The Journal A. M. A., July 31, 1909.)
Sanatoriums and the Acetanilid Habit
To the Editor:—I enclose herewith a “form” letter and
question blank which I received recently from St. Louis.
I may be entirely too wary but I am suspicious that this is
a collection of “statistics” to combat the work of the medical
profession in educating the physician and the laity in the
harmfulness of acetanilid and similar preparations.
G. H. Benton, M.D., Chester, W. Va.
Sterling-Worth Sanitarium.
Comment: The letter which Dr. Benton encloses is in
facsimile form and purports to come from Uriel S. Boone,
M.D., of St. Louis, who states that he is “preparing an
exhaustive article for publication in a leading medical
journal” on the question, “Is acetanilid a habit-forming
drug?” To obtain the necessary data Dr. Boone is “writing
to every hospital and sanitarium in the United States.”
Examination of the question blank which accompanies the
form letter discloses the fact that information is wanted
regarding not acetanilid alone, but also antipyrin and
acetphenetidin (phenacetin). The last question asked runs
as follows:
“If your records [of cases of habitual use of these drugs] are incomplete,
would you allow a reputable physician to investigate the above-mentioned
cases so that he could write with positiveness about them,
and, if necessary, make oath to the truth of his report?” [Italics ours.—Ed.]
Dr. Boone opines that the recipients of his queries “may
hesitate to answer” the question just quoted, but he trusts
that its importance will be evident when he explains that
“it is currently reported that the manufacturers of acetanilid,
phenacetin, etc., have decided to prosecute all libelers of these
drugs” [Italics again ours.—Ed.] and he wishes to make no
statement that he “can not substantiate under oath.” Surely
the life {of} the collector of medical statistics is unusually
hazardous.
For the purpose of aiding Dr. Boone in his arduous search
for truth on the “much mooted question, ‘Is acetanilid a
habit-forming drug?’” we direct his attention to a work that
should prove of invaluable assistance. We refer to Bulletin
126 of the Bureau of Chemistry, entitled “The Harmful
Effects of Acetanilid, Antipyrin and Phenacetin.” This interesting
study to which we have previously called attention
records 112 cases of the acetanilid habit. Of this number at
least 50, or 44.6 per cent. of the cases were those of patients
who took proprietary preparations of the drug.
From this we would not wish to give any bias to Dr.
Boone’s statistics. We hardly expect, however, that such
will be the case. Dr. Boone’s name appears as the author
of an article entitled “A Therapeutic Study of Antikamnia
and Heroin Tablets”—an article that has been very extensively
“quoted” and has been sent out in its entirety by the
Antikamnia Chemical Company. Under these circumstances
we may be forgiven if we venture the opinion that Dr. Boone
is not likely to be unduly prejudiced against “headache
tablets” in general and fake “synthetic” coal-tar mixtures in
particular. We await with breathless interest the appearance
of Dr. Boone’s “exhaustive article” and we must confess
to some degree of curiosity regarding the name of the “leading
medical journal” in which these valuable data will appear.—(Modified
from The Journal A. M. A., Aug. 14, 1909.)
HECTINE
Hectine is a French proprietary and is stated to be,
chemically, sodium benzosulphoamino-phenyl-arsenate. If the
asserted composition is correct, Hectine is similar to atoxyl,
which is described in New and Nonofficial Remedies, 1914,
page 38. Hectine has not been examined by the Council
on Pharmacy and Chemistry.—(From The Journal A. M. A.,
Aug. 8, 1914.)
HYDRONAPHTHOL
A correspondent having requested information regarding
the composition of “Hydronaphthol,” the product was investigated
in the Association laboratory, which reports as follows:
Hydronaphthol is sold by Seabury & Johnson. The label
on a trade package of Hydronaphthol gives no clew as to
the nature of the product. The statements on the labels do,
however, make the claim that Hydronaphthol is an antiseptic
of great power, also that it is non-toxic and therefore
may be used with impunity; thus the following statements
are made:
“A harmless, practically odorless, non-poisonous, non-corrosive antiseptic....”
“... it is non-poisonous and can be employed with perfect immunity
as a preservative....”
The substance has the characteristic appearance, odor
and taste of naphthol. It responded to all the tests of the
United States Pharmacopeia for betanaphthol, with the
exception of the melting point, which was found to be 119 C.
instead of 122 C., an indication of impurity. It is evident,
therefore, that Hydronaphthol is merely a trade-name for
betanaphthol. While resublimed betanaphthol is listed at
10 cents an ounce, Hydronaphthol is listed at 75 cents an
ounce.
Hydronaphthol thus furnishes one more illustration of the
fact that most proprietary medicines for which the most
extravagant claims are made are but old and well-known
remedies sold under a fancy name at a price far in advance
of that charged for the constituent or constituents. The
exploiters are extremely positive in their statements regarding
the non-toxic character of the preparation. Yet, as a
matter of fact, betanaphthol is by no means harmless; it
has been absorbed by the diseased skin with injury to the
kidney and with fatal results. In some cases injury to the
eye has also occurred. These toxic actions should be known
to the practitioner. From 3 to 4 gm. (1 dram) applied to the
skin has produced death (Stern: Therap. Monatshefte, 1900,
p. 165). When a manufacturer advertises a preparation
which possesses potentialities for harm, and especially when
he puts it out under a name which conceals its identity,
it is incumbent on him to warn the customer of possible
injurious or inconvenient actions instead of proclaiming that
the preparation is harmless.—(From The Journal A. M. A.,
Sept. 3, 1910.)
HYDROZONE
The moral principle governing the action of secret proprietary
and patent medicine men is an unknown quantity; sometimes
it would seem to be a negative one. Just how much
lower in the scale of humanity a man can go than to prey on
the fears of a people in the time of a terrible epidemic for
the sake of a few dollars we do not know. There may be
something more despicable, but what is it? Two weeks ago
we referred to the cold-blooded methods of the Peruna
people; this week we reproduce an advertisement from the
New Orleans States that tells another story of man’s inhumanity
to man.

This brings up the problem that we are trying to solve,
viz.: “What is the difference between a ‘secret proprietary
medicine’ advertised in medical journals to physicians and
a ‘patent medicine’ advertised in newspapers to the public?”
Hydrozone is being advertised in nearly all medical journals,
and at the same time in newspapers. Where shall we place
it? And if hydrozone, with the methods recently adopted to
exploit it, is tolerated in the medical press, why not peruna?
HYPOQUINIDOL
An examination of the advertising matter fails to show
that R. W. Gardner makes any definite statement in regard to
the nature or the composition of his Hypo-Quinidol. Certain
statements made in the literature sound much as if the
article might be some sort of a quinin hypophosphite preparation.
But if this is true, its action would be the same as
other salts of quinin and the extravagant claims made could
not be substantiated. It is said to be a “non-toxic quinin.”
It is safe to say that a quinin which will not produce the
toxic symptoms—cinchonism—either is not absorbed in sufficient
quantity or has been so changed that it no longer has
the therapeutic properties of quinin. Altogether Hypo-Quinidol
must be put down as a preparation the composition
of which is kept secret and regarding which extravagant
and highly improbable statements are made.—(From The
Journal A. M. A., Jan. 10, 1914.)
IODONUCLEOID
An Iodin Product Under a Misleading Name
Information has been frequently asked concerning Iodonucleoid,
a product not included in New and Nonofficial
Remedies. The Association Laboratory after investigating
this preparation reported as follows:
This preparation was at one time considered for inclusion
with New and Nonofficial Remedies, and at that time
was examined in this laboratory. The examination showed
that iodonucleoid contains:
Phosphorus | 0.79 | per cent. |
Calcium | 0.43 | per cent. |
| (Equal to 0.6 per cent. CaO). |
Iodin | 24.2 | per cent. |
When 2 gm. was dissolved in tenth-normal potassium
hydroxid volumetric solution and acetic acid added until
faintly acid, an abundant, white, flocculent precipitate
formed. This precipitate was collected, washed with water,
transferred to a beaker, phenolphthalein added and tenth-normal
potassium hydroxid volumetric solution run in
until a pink color was produced. This required 15 c.c.
of tenth-normal alkali. Subtracting from the 2 gm. of
iodonucleoid the 24 per cent. iodin, leaves 1.52 gm.; this
divided by the c.c. of alkali used indicates an equivalent
weight of 1013.
Authorities differ widely regarding the amount of phosphorus
contained in nuclein from different sources, the
figures ranging from 2.9 per cent. to as high as 10 per
cent. If the nuclein from which iodonucleoid purports to
be made contained but 2.9 per cent. phosphorus, the
preparation, after allowing for 24 per cent. iodin, should
still contain 2.2 per cent. phosphorus instead of the 0.79
per cent. found by analysis. A true nuclein should contain
no calcium. If iodonucleoid is a casein compound of iodin
we might expect to find, if the casein had been freed from
milk by acidulation without further purification, both calcium
and phosphorus. The equivalent weight of casein
is given by Long (Jour. Am. Chem. Soc., 1906, xxviii, 372)
as 1124. This figure was obtained on a casein of high
purity, and the figure of 1013 given above agrees fairly
well with Long’s figure for casein. The evidence, therefore,
indicates that iodonucleoid is a compound of iodin
and casein, and not a nuclein compound.
The findings of the laboratory were at that time submitted
to Prof. John H. Long of Northwestern University, who said:
“We have also made a number of examinations of iodonucleoid.
We determined in it the iodin and found the
amount 24.2 per cent. by weight, which is a little more than
that claimed by the manufacturer. We have also tested the
solubility of this substance and find it to behave about as
your laboratory did. As you know, we have been making a
number of preparations from casein, and recently we have
determined the combining power of casein with various
acids, including hydriodic acid. This acid when evaporated
in moderately strong solution with casein yields finally a
hard, dry mass, which may be ground up to a powder
resembling very closely the preparation under discussion.
Various amounts of iodin may be combined here, depending
on the strength of the iodin solution used, and we have
secured some containing over 35 per cent. of iodin. Several
of these preparations resemble closely iodonucleoid, so far
as solubility, appearance and reaction with alkalies on titration
are concerned. I am unable, therefore, to distinguish
this preparation from the casein compounds which we are
making.”
From this it would appear that iodonucleoid is not a
compound of nuclein, as indicated by the name, but instead
is a casein compound of iodin.
Iodonucleoid, then, seems to be another one of the many
iodin “substitutes” which have been put on the market. Other
iodin substitutes are Iodalbin, manufactured by Parke, Davis
& Co.; Iodipin manufactured by E. Merck & Co., and
Sajodin, manufactured by the Farbenfabriken of Elberfeld
Co. As these products have been examined by the Council
and found eligible for inclusion with New and Nonofficial
Remedies, physicians who wish to use substitutes for potassium
iodid would do well to use them instead of a product
presented under a misleading name. Physicians should
understand, however, that these organic iodin compounds
are non-irritating because the iodin is held in such combination
that it is much less active. It seems probable that they
are therapeutically active only to the extent that the iodin
content is dissociated from the organic compound and concerted
into ionic iodin.
A discussion of a number of iodin substitutes is found in an
article by von Notthafft (Monatsh. f. prakt. Dermat., Oct. 15,
1910, p. 343), which was abstracted and commented on in
The Journal, March 4, 1911, p. 685. Von Notthafft believes
that the lower degree of toxicity which these remedies
exhibit has its basis in a feebler activity; either the substitutes
evolve too little iodin or they split it off with greater
difficulty. Physicians should, therefore, view with some distrust
the claims of manufacturers that their products are not
only non-irritating but at the same time possess unusual
therapeutic efficiency. This will apply with especial force
if there is any tendency to conceal the nature or origin of
the combination.—(The Journal A. M. A., July 22, 1911.)
IRIDIUM
Dr. C. A. Dexter, Columbus, Ga., asks for information
concerning the use of iridium in the treatment of acute and
chronic rheumatism. Iridium is a well-known element
although we have not found that it has been used as a
medicine; however, we presume our correspondent refers
to “Iridium (Medicinal),” sold by the Platinum Company
of America. We are not able to locate this company, but
in their advertising circular “Iridium (Medicinal)” is said
to be “an agent for the blood, a laxative, an alterative,
indicated in all disorders of the stomach, in Jacksonian
epilepsy,” and “a specific in rheumatism.” As to its origin,
it is said in the circular, “the platinum sands are associated
with and composed of iridium” and some other elements,
so that as far as the circular gives information the nostrum
is alleged to contain the element iridium.
A few statements quoted from the circular will show that
the person who wrote it knows nothing about medicine
and cannot correctly use the English language: “The qualifications
of Medicinal Iridium are its simplicity, purity,
harmless under prolonged use, easily borne by the stomach.”
“It has been observed that when Medicinal Iridium acts
as a laxative, it will regulate the same.” “Called the family
group, Iridium and Osmium are destined to become the
world’s benefactors in medicinal properties, thereby creating
a new chapter in medical science.” The circular quotes
some supposed “excerpts from hundreds of letters on file,
written by physicians, in the hope that they may attract
your attention,” which bear marks of having been written
by the same person who wrote the circular. Note the quality
of the following statements: “Iridium has a power, purity
and simplicity that pleases me; now I can make progress.”
“I say to you frankly, Iridium is my standard. I can get
results and make progress. I am confident it aids the fibrin
in the blood.” “Dr. X is pushing Iridium on five or six
cases.” It is not explained who Dr. X is, but it has this to
say about him: “Dr. X is an eminent practitioner. He has
made a remarkable record with Iridium and has so far
never failed on cases of Jacksonian epilepsy; experimental
tests have shown that Iridium increases blood-corpuscles.”
The man who signs himself president of the Platinum
Company of America is said to be a lawyer, but is not
working at it, and was formerly a promoter, fiscal agent,
etc. It should not be difficult for the physician to fix the
status of iridium under this sort of exploitation.—(From
The Journal A. M. A., April 23, 1910.)
IRON TROPON
The composition of Iron Tropon seems to have varied
from time to time. The manufacturers formerly stated
that it contained fat, sugar, pepsin and iron in organic
combination with albumin, and its use was advocated both
as a food and as a medicine. It was not claimed to contain
over 1 per cent. of pepsin, but tests failed to show that
it contained any pepsin, or if any, such a small amount
that there was not sufficient to digest the albumin in Iron
Tropon itself. It was also claimed that the iron, being in
organic combination with the albumin, possessed advantages
over the widely used aromatic fluid preparations of iron.
Tests, however, showed that the iron was not in organic
combination, though even had it been, late investigations
fail to demonstrate the superiority of the organic over inorganic
iron compounds.
The manufacturers state in their later “literature” that
Iron Tropon is a tonic and a food; that it is a compound of
the food albumin tropon, 2.5 per cent. of iron in its most
assimilable form, and enough chocolate to flavor it agreeably.
It will be noted that they now make no claim for
pepsin, nor do they state that it contains iron in organic
form. In the dose recommended, a teaspoonful three times
a day for an adult, the patient gets something over a grain
of iron, and he might as well take an equivalent quantity
of Blaud’s mass, the value of which has been proved.
As a food, Iron Tropon, weight for weight, is about equal
to beans and a little better than flour, although it contains
a larger percentage of protein than either. In the dose
stated, an invalid would get about 50 calories, or about 1⁄40
the necessary nourishment for a day. Tests have also shown
that the albumin is difficult of digestion. In spite of this
fact, the advertisement of Iron Tropon states: “A patient
who takes Iron Tropon receives not only the benefit of iron
medication, but at the same time his economy is supplied
with perfectly assimilable albumin in sufficient quantity.”
It will thus be seen that the claim for pepsin in this preparation
has been abandoned, that the statement as to the iron
being in organic form has been modified, and that the food
value of the albumin is exaggerated; but perhaps the manufacturers
do not expect the physician to apply his arithmetic
to such problems.—(From The Journal A. M. A., April 23,
1910.)
KUTNOW’S POWDER
Which Is It, a “Proprietary” or a “Patent Medicine”?
The term “patent medicine” has been applied, rather
loosely, to those nostrums sold and exploited directly to the
public, while the name “proprietary” has been given such
preparations as are advertised only to the medical profession.
As has been many times exemplified by reports in
The Journal, the distinction is often a very fine one and
the dividing line frequently reaches the vanishing point.
It is not unusual, for instance, for “proprietary” preparations
to be foisted on the medical profession until a certain
number of testimonials (of doubtful value, it is true, but
still testimonials) have been ingeniously wheedled out of
physicians and the product rather generously prescribed.
When this objective point has been reached the manufacturer
comes into the open and advertises the nostrum to the public
direct and the testimonials previously given for the “proprietary”
are used as advertising assets for the “patent medicine.”
Then again there are certain preparations which are
“proprietaries” or “patent medicines” according to the location.
On one side of the Atlantic the product is advertised
to physicians only, while on the other side it runs indiscriminately
on the billboards and in the newspapers. One
of the best examples of this last class is Kutnow’s Powder.
In England, where it originated, this preparation, which
“dissolves and eliminates uric acid,” is consistently lined
up with Beecham’s Pills and Pink Pills for Pale People.
Full-page newspaper advertisements announce the fact that
free samples will be
“SENT TO ALL APPLICANTS”
In the United States, however, Kutnow’s have learned
from their wide advertising experience that a cheaper and
surer way of introducing a nostrum to the public is to advertise
it to the medical profession only. By means of advertisements
in medical journals (whose space is much less
expensive than that of the daily papers) and the liberal
distribution of samples which are
“SENT FREE TO PHYSICIANS ONLY”
the medical profession becomes the unpaid “barker” for the
nostrum manufacturer. At present, therefore, Kutnow’s
Powder is—in the United States—an ethical (!) “proprietary.”

There exists in this country, as most of our readers
know, an organization of “patent medicine” manufacturers
whose “reason for being” is to get full value received for
the $40,000,000 paid annually in advertising nostrums in the
newspapers of the country. This organization is known as
the Proprietary Association of America. The now familiar
“red clause” in the advertising contracts by which the
newspaper forfeits its contract if state laws are enacted
that are inimical to the “patent medicine” interests, is a
creation of this organization and has been most effective in
making the newspapers the unpaid lobbyists of the nostrum
interests. The “silence clause” is another “joker” in the contracts
by which the agreement is canceled if matter detrimental
to the nostrum “is permitted to appear in the reading
columns” of the paper. It is little wonder that with such
weapons the “patent medicine” manufacturer has assumed
an arrogance that is as disgusting as it is serious.
Great Britain, too, has its “patent medicine” men’s organization,
which is known as the Proprietary Articles Trades
Association. Of both these honorable bodies Mr. S. Kutnow
of Kutnow Brothers, Ltd., is, or was, a conspicuous member.
At a recent meeting of the British organization, Mr.
Kutnow worked himself into a fine frenzy of indignation
because of some articles that had appeared in the Pharmaceutical
Journal of London on the subject of “Secret Remedies
and Proprietaries.” As these articles did not specifically mention
Kutnow’s Powder, and as evidence was directed against
only those preparations as were most disreputable, it is evident
that Mr. Kutnow now appraises his own product at its face
value. He gave his opinion of the Pharmaceutical Journal
and told the meeting that when the advertising man for that
journal solicited advertising he refused to have any more dealings
with him owing to the articles that had appeared in the
Pharmaceutical Journal. He declared that he was quite
independent of any newspaper or journal, and was able to take
care of himself.
Therein Mr. Kutnow is mistaken; he is not independent of
newspapers and journals. On the contrary, he and others
of his ilk are most subserviently dependent on them. Let
reputable papers and medical journals refuse, for but one
year, to carry the high-flown advertisements of his Anglo-American
Patent-Proprietary, and his firm would perforce
seek some worthier, if less profitable, line of business.
The editor of the Pharmaceutical Journal resents Mr.
Kutnow’s “implied assumption that by inserting paid announcements
in the advertising columns of a newspaper, he or any
one else, can dictate the policy of that organ.”
The Pharmaceutical Journal, it should be said, is the
official organ of the Pharmaceutical Society of Great Britain,
and is the most influential organ of the drug trade in the
British Isles. It is refreshing to note, in these days of
“canned” editorials and paid “write-ups” masquerading as
original articles, that there is still to be found a journal that
can not be bought.
One wonders whether a large experience in the advertising
world, and especially his membership in the Proprietary
Association of America, has unconsciously led Mr. Kutnow to
assume that muzzling the press is one of the perquisites
of the large purchasers of advertising space.—(From The
Journal A. M. A., Aug. 31, 1907.)
LYMPH COMPOUND R-H AND ORCHITIC
FLUID TABLETS
A physician wrote The Journal:
“I have under my care a patient with chronic parenchymatous
nephritis. Microscopic examination shows occasional
epithelial casts, with hyaline and granular casts. The
patient feels well and appears to enjoy the best of health.
Please give me your opinion on the use, in such a case, of
the lymph injections put out by the Animal Therapy Company
of Chicago, from whom I have just received a supply
of literature.”
The Journal replied as follows:
“The Council on Pharmacy and Chemistry took up for
consideration the ‘Lymph Compound R-H,’ and ‘Orchitic
Fluid Tablets,’ sold by the Animal Therapy Company and
refused them recognition.
“The treatment of a case of chronic parenchymatous nephritis
is a task requiring the best judgment and the greatest
knowledge that the physician can command. From the first
his aim should be to do no harm, and with this in view he
will recognize that since we do not know the cause of this
disease, and since we are unable to influence the essential
process in the kidney, the administration of a remedy capable
of doing harm should be undertaken only under the clearest
indications. It is probable that the remedy proposed, containing,
it is asserted, a mixture of foreign proteins, might
injure healthy kidneys, to say nothing of sick ones. It is
well known that foreign proteins, such as the white of egg,
if they enter the circulation unchanged, are excreted by the
kidney and are liable to produce serious irritation, which in
the case of parenchymatous nephritis might easily aggravate
the existing condition and frustrate all other efforts at a cure.
More especially is it imperative to do no harm when, as in
the case reported by our correspondent, the patient appears
to be in good health. Two questions should be raised in that
case. First, is there any evidence that the occasional excretion
of a few casts, whatever may be their variety, is actually
doing the patient any harm? And second, since there are no
symptoms, what possible improvement could be expected from
treatment? It is admitted that we have no remedy which
can affect an essential change in the condition of the diseased
kidney. What must be done in such a case is to spare
the kidney—to require of it the minimum of functional
activity. In such a case, the physician who introduces an
animal protein, foreign to the human system, would be taking
a serious responsibility. The chances are that it would do
harm; how great, no one can tell. If the physician can really
make up his mind to experiment at the risk of the life of his
patient, this case appears to be one unusually favorable to
the manufacturer of the serum. There are chances that the
diagnosis may be incorrect and that such a condition of the
urine does not indicate a serious condition of the kidney.
It is frequently the case after an acute infection, or some
similar irritation, that the kidney continues for some time
to excrete albumin and casts, but the condition eventually
clears up. In such a case, if the serum did no harm it would
be given the credit of curing the patient, who recovers in
spite of it. It is a little unfortunate, however, for the purpose
of such demonstration that the patient is said to feel well.
If only he could be persuaded that he has a serious disease
so that he might be somewhat depressed mentally, the effect
of the cure would be more remarkable!
“If the physician to whom such a patient comes for advice,
instead of taking the wise course of seeking reliable information,
were to take at their face value the statements of interested
commercial manufacturers—if he were to administer
this unknown and dangerous remedy, the effects of which
he cannot predict—he would commit a breach of trust more
culpable than the most vicious attempts of the nostrum-maker
to mislead the physician and the public.”—(From the
Journal A. M. A., Dec. 14, 1912.)
LYSOL-THE EVOLUTION OF A PROPRIETARY
Regarding certain proprietary preparations and their equivalents
found in the pharmacopeias or other standard works
of reference, it is often questioned whether the proprietary is
the original and the official preparation the imitation, or vice
versa. As a general proposition, medicinal compounds and
preparations are not born but evolved, as in the case of
epinephrin, in which the credit of discovery belongs to no one
person, but to several.108 So it is often the case that the proprietary
and official preparations may be based one on the
other, while both are usually based on some preparation
which antedates them. This is well illustrated by the proprietary
preparation Lysol, the practical equivalent of which—liquor
cresolis compositus—is official in the United States
Pharmacopeia. After the discovery of phenol (carbolic acid)
and the recognition of its germicidal value, it was gradually
learned that other phenolic compounds occurring in the crude
distillates from tar and wood were more efficient and less
poisonous than phenol (carbolic acid). When this was discovered,
attempts of course were made to utilize these higher
and more efficient phenols, which meant that their insolubility
in water had to be overcome. In these attempts there
were efforts to form new compounds as well as a search for
simple solvents. While the first failed, because these compounds
were less efficient than the phenol from which they
were made, a simple solvent was found in soap. The first
attempt to utilize the solvent power of soap gave creolin, a
mixture of the so-called crude carbolic acid (really containing
but little phenol and consisting largely of higher phenols
along with inert hydrocarbons) with soap. This was followed
in 1884 by Schenkel’s discovery that a portion of this
“crude carbolic acid” could be made soluble in water by
treatment with soap. Schenkel was refused a patent on the
ground that any soap manufacturer should be permitted to
add phenol to his soap, but in 1889 a patent for a cresol-soap
solution was granted to Damann, who used cresol, a constituent
of “crude carbolic acid.” The preparation was put
on the market and has since been widely advertised under
the proprietary name “Lysol.”109 It is thus seen that Lysol is
a good example of the way in which manufacturers appropriate
the discoveries of others, develop them and turn them
to proprietary use.
The ill-deserved patent protection for Lysol happily expired
long ago and the product can now be made by anyone. In
view of the non-descriptive character of the name “Lysol”
and the danger in using such names in connection with
potent and poisonous remedies, this cresol-soap solution has
been admitted to pharmacopeias, not under the original name
“Lysol,” but under descriptive names such as that in the
United States Pharmacopeia—“liquor cresolis compositus.”—(From
The Journal A. M. A., Dec. 14, 1912.)
THOMPSON’S MALTED FOOD COMPANY
And Its Blood, Nerve and Tissue Builder, “Hemo”
During the past eighteen months The Journal has received
inquiries from physicians in various parts of the country
asking for information regarding “Thompson’s Malted Food
Company” of Waukesha, Wis., which for some time has been
selling and trying to sell its stock to physicians and others.
In reply The Journal called attention to the fallacy of any
concern trying to induce people to purchase its stock on
the ground that the Bromo-Seltzer company, the Postum
Cereal company and others had been successful.

A miniature reproduction of a full-page advertisement of “Hemo”
appearing in a Milwaukee paper. The original covered 341 square inches.
The earlier name for the Thompson concern was “American
Malted Food Company” and originally it had its office
and factory in Milwaukee. Its products are “Malted Milk,”
“Malted Beef-Peptone” and “Hemo.” As long ago as 1911
the company was sending out glowing descriptions of the
money that might be expected to be made by investing in the
concern, which was then known as the American Malted
Food Company. According to the company’s booklet it was
estimated that in the United States and Canada alone
$20,000,000 worth of dry malt food products were consumed
annually. Further:
“On the most conserving estimate the American Malted Food Company
will supply 5 per cent. of the demand or $1,000,000 of the consumption
from the very outset....”
At the time this was written (1911) the company claimed
that its stock had “been advanced over 100 per cent. in one
year’s time” and that even then it had “found it advisable
to curtail the sale of stock so as not to exceed the prescribed
number of shares to any one person.” In 1914, however, the
company still had its representatives in the field offering
stock for sale.
In November, 1913, one physician wrote that three years
previously he had purchased stock in the company and at
that time (1910) was given to understand that dividends
would be paid in at least a year! He has received no dividends
to date. Other physicians have written that the company’s
agents in attempting to sell stock have given the
impression that the concern either is paying dividends or is
about to pay dividends. Still other physicians say that the
agents made no claim to them that the concern was paying
dividends. To quote from a few of many letters together
with the date of the communications:
“Alluring promises of big profits in the near future have been held
out by the canvassers....” (September, 1913).
“No actual promise was made but it was ‘estimated’ that dividends
would be declared ‘about the first of the year’” (November, 1913).
“After I said I wouldn’t buy any stock was told the company was
already paying dividends and I was turning down a good thing.”
(March, 1914).
“The agent gave me to understand that the profits of the company
were enormous and that large dividends would be paid in the near
future.” (March, 1914).
“Mr. —— [the agent] told me the company expected to pay dividends
as soon as all the stock had been sold.” (May, 1914.)
“The agent gave me to understand that they were about to pay very
generous dividends and that it was a chance to get in on the ground
floor on a good thing.” (May, 1914).
The price asked for stock during the past year or more
has been $1.50 a share, the par value of the stock being held
at $1.00. At various times, however, the stock seems to have
been purchasable from other sources at a much lower figure.
A physician writing September, 1913, stated that he had
just been solicited to purchase stock in the Thompson Malted
Food Company at $1.50 a share, and that immediately he
wrote to two firms, one in Chicago and one in Milwaukee,
that sell unlisted securities, asking the price of Thompson’s
Malted Food Company stock. Both brokers expressed the
opinion, according to the physician, that at 90 cents a share
the stock would be a “good buy,” and both offered to undertake
to secure stock at that price. One of the concerns sent
a circular to the physician offering the stock at 80 cents.
The physician thereon canceled his order for stock which
he had made at $1.50 and declared that if he bought at all
he would buy from other sources.
HEMO
At the present time the product that Thompson’s Malted
Food Company seems to be “pushing” is a product it calls
“Hemo.” According to advertisements, Hemo is “the food
that builds up weak stomachs.” Hemo, we are told, contains
“the iron of spinach, the juices of prime beef, the tonic properties
of selected malt in powdered form and the richest
sweet milk.” Furthermore, “Hemo contains the active principle
of selected barley malt ...”—whatever the “active
principle” of barley malt may be.
According to the Thompson Malted Food Company “80
per cent. of the American citizens” are “troubled with
anemia” and it is for them that “Hemo has been especially
prepared.” In a sentence:
“It is a well-known fact that organic iron can be obtained from
animal life as well as from vegetable life, and as the digestive organs
of a majority of the people are not equal to the task of supplying their
bodies with a sufficient amount of organic iron to maintain a supply of
a good quality blood, the lack of which results in numerous nervous
ailments—insomnia, diabetes, rheumatism, anemia, tuberculosis, etc.,
it has been found necessary to secure for mankind organic iron in a
form that will be concentrated, palatable and most easily assimilated.”
This is a sample of the farrago of pseudo-scientific nonsense
sent out by this concern in its attempt to sell “Hemo.”
To continue the quotation:
“With this object in view, the laboratories of Thompson’s Malted
Food Company have successfully produced and successfully tried out
Hemo on the most desperate cases.”
In a letter addressed to a physician-stockholder, the statement
is made: “Our Hemo-Malted Milk has never had and
never will have an equal as a builder of blood, nerve and
tissue ... it will build tissue, nerve and blood in less
time than any other food heretofore known.”
Disregarding the question whether or not this is a stock
jobbing scheme or whether the purchase of the stock is a
good investment, there is another side to the matter. It
must be evident that the public is not getting a square deal
when physicians are financially interested in the products
they prescribe for, or recommend to, the sick. Whatever
the value of the Thompson products, the method of exploitation
and the attempts on the part of the company to get
physicians financially interested in its ventures, are to be
deprecated. If laymen of a speculative turn of mind wish
to invest in the stock of companies putting out “bottled
energy,” “blood builders” and “nerve repairers” that is their
business, but it is certainly neither conducive to the scientific
practice of medicine nor to the interest of the public for
physicians to be financially interested in products of this sort.—(From
The Journal A. M. A., Oct. 24, 1914.)
MANOLA
Physicians as Unpaid Pedlers of Nostrums
One of the most disheartening features of the fight against
the proprietary evil within the profession is the slowness with
which physicians awake to their responsibilities in the matter.
It is a notorious fact, familiar to physicians against advertising
men alike, that the simplest and cheapest way to introduce
a nostrum to the public is through the instrumentality of the
medical profession. Ever since the birth of the proprietary
evil in this country, shrewd manufacturers have persuaded
doctors to act as unpaid pedlers for their wretched nostrums
and to become particeps criminis in the exploitation of such
wares.
Manola is an alcoholic nostrum with just enough more or
less inert medicinal products added to exempt it from the
internal revenue tax, but not enough to prevent its being used
as a tipple by those who object to taking their “toddy” in a
simpler form. It is prepared by the Luyties Pharmacy Company
of St. Louis, a homeopathic concern whose hahnemannian
leanings are not so strong but that it is willing to cater
to the various sectarian schools of medicine as well as to the
regular profession. Since the promoters realize, doubtless,
that to put this stuff out under a homeopathic label might not
be conducive to stimulating physicians’ confidence, Manola
is labeled: “Prepared only by the Manola Company, St.
Louis.” In other words, it is the old dodge of forming subsidiary
companies for the purpose of hiding the identity of
the real owners. In this connection, it is worth reminding
our readers, incidentally, that the Walker Pharmacal Company,
St. Louis, is another subsidiary concern of the Luyties
Pharmacy Company, created for the purpose of pushing
another nostrum—Hymosa.
Manola is seldom advertised in medical journals. Instead
the Luyties Pharmacy Company has discovered a more effective
method of “putting one over” on physicians and druggists.
The method which has been pursued for years and
which, under the same title and subtitle that head this article,
was exposed in The Journal as long ago as May 6, 1905,
consists in sending to physicians a letter containing three
postcards—unstamped, of course. With the postcard there is
a slip that reads:
INSTRUCTION FOR OBTAINING
3 BOTTLES OF MANOLA FREE
Dear Doctor: Fill out the attached cards Nos. 1 and 2. Mail No. 1
to us and hand Nos. 2 and 3 to your druggist. Impress upon him the
necessity of mailing postal card No. 3 direct to us, and not to his
jobber.
Yours truly,
THE MANOLA COMPANY.
The postcards are numbered, respectively, 1, 2 and 3. Here
is No. 1:
Dr Ezymark writes the name of his druggist on Card 1,
puts a stamp on it and mails it to the Manola Company,
alias Luyties Pharmacy Co.
Card 2, addressed to his druggist, also is filled out by Dr.
Ezymark. Here it is:
Then the doctor, acting the part of errand-boy, delivers
Card 2 and also Card 3 to his druggist. Here is Card 3:
This, Mr. Goat, the druggist, has to fill out, affix a stamp
and send to the Manola Company. In return for all this, Mr.
Goat has his shelves loaded up with a dozen bottles of
Manola and, for that privilege pays $8 out of his own pocket.
Dr. Ezymark gets three free bottles. Incidentally, he also
gets the contempt of his druggist—and of such patients as
learn of it.
The only one who profits by all this is the Luyties Pharmacy
Co., alias the Manola Co., alias the Walker Pharmacal
Company.
Evidently this method of exploitation pays; that it does
pay is a disgrace to the medical profession. To those physicians
who have in the past acted as pedlers for Manola we
would say: If your patients really need sherry wine let them
purchase it under its own name and at the ordinary market
price. You will then know what they are getting and you
will be able to retain not only your own self-respect but
also the respect of your druggist and the public.
The Composition of Manola
Examination of Manola in the Association laboratory indicates
that its composition is consistent with its origin, for
its medicinal ingredients are present in truly homeopathic
quantities. The laboratory report follows:
An examination of an original bottle of Manola gave the
following results:
Specific gravity at 25 C. | 1 | .0329 |
Alcohol | 18 | .00 | per cent. by vol. |
Non-volatile matter (residue on evaporation) | 15 | .93 | gm. in 100 c.c. |
Ash | | .96 | gm. in 100 c.c. |
Phosphoric pentoxid (P2O5) | | .0668 | gm. in 100 c.c. |
Total alkaloids | | .0047 | gm. in 100 c.c. |
Calcium | Traces. | |
Magnesium | Traces. | |
Iron | Traces. | |
Sodium | Traces. | |
Arsenic | Traces. | |
Manola is a light amber colored liquid having the odor
and taste of sherry wine. The above analysis indicates that
it is nothing more than wine, fortified with alcohol and a
slight amount of medicinal substances added. The non-volatile
matter appears to be nearly all sugar, glycerin, or some
similar substance and the presence of less than one gram of
ash to 100 c.c. excludes the presence of more than a small
amount of organic salts. From the amount of phosphorus
found there appears to be about one dose of phosphoric acid
to a twenty-ounce bottle. Arsenic is present in such small
amounts that the ordinary hydrogen sulphid test failed to
show its presence and the delicate Gutzeit’s test had to be
used to detect it.—(Modified from The Journal A. M. A.,
April 2, 1910.)
MERCOL
R. Hunt and A. Seidell, Washington, D. C., report the result
of an examination of a preparation called Howell’s Mercol,
manufactured by H. B. Howell & Co., Ltd., New Orleans, and
claimed to be a 1 per cent. solution of mercuric iodid in a
non-irritating neutral menstruum, and recommended for hypodermic
use in the treatment of syphilis. Their examination
indicates, as they say, “that although the manufacturers of
Mercol may have used a mercuric iodid in its preparation,
they have not succeeded in obtaining a 1 per cent. solution
of this compound in their ‘non-irritating neutral menstruum.’
It is furthermore evident that the sample examined as above
outlined contains none, or at most only traces, of biniodid
of mercury.” It is stating it mildly to say that a manufacturer
is careless who claims to make an efficient preparation
of what is almost a specific for one of the most serious
of diseases but which contains practically none of the essential
active ingredient.—(Abstracted from The Journal
A. M. A., Jan. 16, 1909.)
The Component Parts and the Finished Product
After the appearance of the first article, a physician wrote
stating he had seen Mercol manufactured, following the
process in detail and had himself weighed out a sufficient
quantity of mercuric iodid to produce a 1 per cent. solution.
He protested that the firm “had no desire to foist on the
medical profession or the public a fraud.” With his letter he
sent a sample of the particular batch of Mercol which he had
seen manufactured. This sample was analyzed with the same
care and thoroughness that the previous sample had been,
and the practical absence of mercuric iodid was again demonstrated.
While The Journal does not question the honesty
and good faith of either the manufacturers or the physician,
it maintains that claims for remedial agent should be based
on the finished product rather than on the component parts
used in its manufacture. Without attempting to explain
what has become of the mercuric iodid, it insists that the
important fact, and the one that vitally concerns both patient
and physician, is that the finished product fails to contain it.
If the manufacturer has made an honest mistake in supposing
he could produce a 1 per cent. solution of mercuric iodid in
liquid petrolatum, he will doubtless see that the mistake is
corrected. If, on the other hand, he is governed by commercial
considerations only, the misrepresentation will probably
be perpetuated.—(From The Journal A. M. A., May 15, 1909.)
MIDOL AND NURITO
Pyramidon Entering the Patent-Medicine Field
Repeated warnings to the public of the dangers of acetanilid,
antipyrin and acetphenetidin and the requirement in the
Food and Drugs Act which makes it obligatory to declare
the presence of acetanilid and acetphenetidin on the labels of
“patent medicines,” have been responsible for the growing
unpopularity of nostrums containing these drugs.
MIDOL
During the past few months advertisements have appeared
in the newspapers of a new “headache cure,” the advertising
slogan of which is that it “contains no acetanilid or
phenacetin.”
The name of this preparation is Midol and it is sold under
the following claims:
“Instantly relieves headache, neuralgia, toothache.”
“Has no depressing effect.”
“More effective than antipyrin, acetanilid, phenacetin or similar pain-relieving
products.”
“Midol is the one safe-to-take aid of sufferers of headache.”
“Quickly relieves pain of whatever nature.”
“There is no cumulative action.”
“No bad effect upon the heart or other organs.”
An original package of Midol was purchased and examined
in the Association laboratory. The chemists’ report follows:
“Midol is sold in the form of white tablets each weighing,
on an average, 0.425 gm. or about six and one-half
grains. The tablets are soluble in water, chloroform or
benzene to the extent of about 80 per cent. The soluble
portion appeared to be largely composed of starch, with
about 4.5 per cent. of some inorganic matter, probably
talc. The chloroform soluble portion was found to consist
chiefly of pyramidon, chemically known as dimethyl-dimethylamino-pyrazolon.
Besides pyramidon, the chloroform
soluble matter contained a small quantity of caffein
and may have contained small amounts of other substances.
“From examination it is concluded that Midol depends
essentially on pyramidon for its therapeutic effect.”
Pyramidon is a proprietary preparation derived from, and
having the antipyretic and anodyne properties of, antipyrin.
While some observers have asserted that it is more likely
to cause collapse than are either antipyrin or acetphenetidin
there is no positive evidence of this assertion. That the use
of pyramidon has been until recently practically restricted to
physicians may account for the fact that its toxic effects are
not as well known as are those of antipyrin, acetphenetidin,
acetanilid, etc., which for some years have been indiscriminately
used by the public. As the use of pyramidon as a
“patent medicine” now bids fair to become as general as the
better known antipyretics, it is probable that its toxicology
will become better known.
It is interesting to note that pyramidon in the form of
Midol is put on the American market by the General Drug
Company, which also acts as a distributor of salvarsan
(“606”). The General Drug Company is said to have for its
president, W. M. Hoge, who was formerly employed in the
comptroller’s office during the administration of Herman A.
Metz, and the latter being employed by the Consolidated
Color and Chemical Works and being president of Victor
Koechl & Co. The General Drug Company, in its price list
to physicians, lists the “ethical proprietary” pyramidon, but
contains no mention of its “patent medicine” Midol.
NURITO
Midol is not the only “patent medicine” in which pyramidon
is the essential drug. Nurito, which is advertised as “not a
patent medicine but a proprietary preparation,” is a nostrum
put on the market by the Magistral Chemical Co., New York.
Here are some of the claims:
“Only U. S. P. ingredients are used in Nurito.”
“Guaranteed to relieve or your money refunded, Rheumatism, Sciatica,
Neuritis.”
“There is no compound known in medicine that so rationally, scientifically
and effectively removes waste and poisons from the human
system as Nurito.”
The Association’s laboratory recently analyzed a specimen
of Nurito. The report follows:
A dollar-size package of Nurito was purchased and found
to contain seven powders. The powders ranged in weight
from 9 to 12 grains, the average weight being nearly 11
grains. The presence of pyramidon, phenolphthalein and
milk sugar was demonstrated. Alkaloids, acetanilid, acetphenetidin,
chlorids, bromids, iodids, heavy metals, starch
and sulphates were absent. Quantitative examination indicated
that the composition of Nurito is essentially as follows:
Milk sugar | 34 | per cent. |
Phenolphthalein | 6 | per cent. |
Pyramidon | 60 | per cent. |
Each powder, therefore, contains about 22⁄3 grains of milk
sugar, 2⁄3 of a grain of phenolphthalein and 62⁄3 grains of
pyramidon.
What was said of pyramidon in the preceding article
applies equally well here. The claim that Nurito is composed
of “U. S. P. ingredients” is evidently a falsehood. The chief
therapeutic ingredients are pyramidon and phenolphthalein,
neither of which is described in the United States Pharmacopeia.—(From
the Journal A. M. A., Aug. 10, 1912.)
MU-COL
Salt and Borax as Wonder-Workers
“Mu-col, for Cleansing Mucous Membranes” is a nostrum
put on the market by the Mu-col Company (Inc.), Buffalo,
New York. As a specimen of the claims made for the preparation,
the following is typical:
“Mu-col obtains most gratifying results in catarrhal inflammations
of the mucous membranes. Leucorrhea, Tonsillitis, Sore Throat,
Cystitis, Internal Hemorrhoids, Nasal Catarrh and Pus Cases respond
at once to irrigation with Mu-col solution. Strong solutions of Mu-col
have proven of sterling value in treating Hives, Prickly Heat, Ivy Poison,
Sunburn, Eczema, Typhoid and Scarlet Fever.”
This, and much more Mu-col will do—according to its
manufacturers! No wonder physicians want to know the
composition of Mu-col. As the manufacturers do not give
this information the aid of the Association’s Laboratory
was invoked. Let the chemists speak:
LABORATORY REPORT
“The specimen examined was a white powder, and from
the odor, thymol, eucalyptol, camphor and oil of wintergreen
could be recognized. Qualitatively sodium, chlorid
and borate were found. Zinc, benzoate, phenolsulphonate
and sulphate could not be found. The solution was
alkaline to litmus. Gravimetric determination of chlorid
as silver chlorid and titration of borax by Thompson’s
method indicated sodium chlorid (NaCl) 47.2 per cent.,
sodium borate (Na2B4O7+10H2O) 50.1 per cent.
“It thus appears that Mu-col is a mixture of ordinary salt
and borax in equal parts with the addition of a small
amount of aromatic substances.”
Mu-col will do just what a solution of salt and borax will
do—no more, no less. And yet, it is claimed:
“Mu-col has been successfully used since the year 1900 by more
than 50,000 physicians, which has proven it to be the most Efficient,
Economical and acceptable, preparation in its field.”—(From The Journal
A. M. A., Feb. 7, 1914.)
NARKINE
The Intangible Product of the Tilden Laboratory
A little book, published by the Druggists Circular, and
called “Modern Materia Medica,” gives in dictionary form
the information regarding new remedies which that journal
publishes in its monthly issues. Such information is not
always acceptable to the manufacturers of various preparations
of doubtful value. A case in point is brought to notice
with reference to a remedy called Narkine, put out by the
Tilden Company of St. Louis. In this little book the following
appears:
“Narkine is described as ‘an opium preparation from
which all deleterious qualities have been eliminated’; an
unsupportable claim, as all opiates and other hypnotics
are essentially deleterious.”
The Tilden Company wrote to the Druggists Circular, stating
that they guaranteed Narkine “to be absolutely free from
coal-tar or opium derivatives,” yet the “literature” of the
company describes it as
“a specially prepared product of opium devoid of the nauseating
and disagreeable properties of this drug, yet possessing the
anodyne and soporific principles of same in the highest degree.”
To remove from opium all its derivatives and yet retain the
anodyne and soporific principles attached to nothing in particular,
indicates a degree of pharmaceutical skill seldom
attained. One is irresistibly reminded of the Cheshire cat in
“Alice in Wonderland,” whose smile remained long after the
cat had vanished.
The absurdity of the thing, however, has apparently not
occurred to many physicians, for these disembodied spirits
of the pharmacologic world are evidently being prescribed.
The Druggists Circular is to be congratulated on exposing
this latest pharmaceutical freak. It does so in a rather
striking manner by means of photographic reproductions of
the claims of the Tilden Company.—(From The Journal
A. M. A., Oct. 24, 1908.)
PAPINE
A Disguised Morphin Solution
To the thinking physician it should be evident that a preparation
containing morphin must possess not only all of the
valuable properties of this drug, but also all of the objectionable
ones. There are still some physicians, apparently,
who give credence to the assertions of the manufacturers concerning
the morphin preparation from which, it is claimed,
all of the undesirable morphin effects have been removed.
The following query from a correspondent illustrates this
fact:
“Will you inform me as to the contents of ‘Papine’? I
have a case of chronic interstitial nephritis, and my consultant
insists on giving this preparation. I asked him if he
knew what drugs it contained and his answer was ‘one-eighth
of a grain of morphin with the objectionable parts of
the drug removed.’”
The query was referred to the Association Laboratory,
which submitted the following report:
For many years Papine has been advertised by its
makers, Battle & Company, St. Louis, as an anodyne. In
the circulars Papine is described in part as follows:
“Papine represents in pharmaceutical form the purely anodyne principles
of opium freed from the narcotic and tetanising constituents.”
“Papine is the anodyne or pain-relieving principle of opium, the
narcotic and convulsive elements being eliminated. One fluid drachm
is equal in anodyne power to one-eighth grain of morphin.”
“Through special methods of preparation, the anodyne and analgesic
principles of Papaver somniferum are so extracted as to free them of
the narcotic and convulsive elements that ever have been, and must
ever continue to be serious objections to the use of opium and its
common derivatives.... No demand is more regularly made
on the physician than that for the relief of pain, and to be able to
afford it promptly and completely, without the slightest deleterious
action, is an advantage that cannot be overestimated.”
“Unlike most derivatives and preparations of opium, Papine neither
nauseates nor constipates; nor does it inhibit the secretory functions
of the body.”
“In conditions of extreme nervousness, especially in women, recourse
to morphin is attended by the very real danger of the formation of a
habit. Lastly, opium and its alkaloids must not be administered to
persons whose kidneys are not in good working order on account of
the risk of toxic accumulation.”
“No such restriction exists in respect of Papine, its action being
exerted exclusively on the element pain; in other words, it is purely
anodyne.”
“Papine does not nauseate, constipate nor create a habit.”

The Papine label before (on left) and after (on right) the passage
of the Food and Drugs Act. And the exploiters of this morphin solution
have the effrontery to claim that it does not create a habit!
From these statements the incautious physician might
be led to infer that Papine is a preparation analogous or
similar to the official tincture of deodorized opium. Formerly
in the manufacture of the latter preparation, in addition
to removal of the odorous substances, narcotin, then
thought to be the principal convulsive alkaloid,110 was also
removed. By the process for the manufacture of this tincture,
which is now official in the United States Pharmacopeia,
most of the narcotine is found in the finished preparation.
While it is a comparatively simple matter to
remove the narcotin from opium and its preparations, thus
eliminating most of the commonly reputed “convulsive elements,”111
to remove the “narcotic elements” from opium
would result in destroying the integrity of the product.
The reasons for this are that morphin is the most powerful
narcotic substance found in opium, and it is present in the
largest proportion of any of the alkaloidal constituents.
Its removal from an opium preparation would, therefore,
render that preparation practically valueless.
From Papine, however, the morphin has not been
removed, for since the passage of the Food and Drugs Act
the label has to admit that Papine contains 1 grain of
morphin in each ounce!
A specimen of Papine was examined and found to be
nothing more than a simple aqueous-alcoholic solution of
morphin, containing glycerin. The preparation is flavored
to imitate cherry and colored with cochineal. With the
exception of morphin, neither narcotin, codein nor other
opianic alkaloids were found, while meconic acid, a characteristic
constituent of opium, was absent. Since Papine is
claimed not to cause constipation, and as is well known, this
condition is frequently produced by morphin, it seemed possible
that Papine might contain laxative substances. On
examination, however, no cascara, rhubarb, phenolphthalein
or laxative salts were found.
While Battle & Co. have persistently exploited Papine as
being an opium preparation having none of the objectionable
qualities of opium, the analysis shows that the paradoxical
claims made for it cannot be substantiated. In prescribing
morphin there is an abundance of official preparations to
choose from, and there certainly is no necessity or excuse for
resorting to the much more expensive and in no way superior
Papine.—(From The Journal A. M. A., April 29, 1911.)
A physician asks: “Can you tell me the formula of a preparation
on the market called Pasadyne, put up by John B.
Daniel, Atlanta, Georgia?”
According to the manufacturer Pasadyne is a tincture of
passion-flower. Formerly this nostrum was sold under the
title “Daniel’s Concentrated Tincture of Passiflora Incarnata.”
While the manufacturer claims marvelous virtues for
this preparation, made from “the fruit, roots and vines of the
passion-flower or May-pop,” passiflora (passion flower) is
now generally recognized as being of little if any value.
A circular makes the following absurd statement:
“Chloral and the bromids, before the recognition and advent into
medicine of Pasadyne (Daniel’s Concentrated Tincture of Passiflora
Incarnata), were widely employed in all turbulent states of the psyche
and, notwithstanding their many untoward, even sometimes dangerous
effects, were held in high favor by physicians. For that matter, they
still retain some of their old-time popularity, but since the superior
value of Pasadyne (Daniel’s Concentrated Tincture of Passiflora Incarnata)
has been demonstrated to the profession’s satisfaction, the erstwhile
high esteem in which chloral and the bromids were held, is fast
waning and ere long Pasadyne will have crowded them out.”
The reasons why the drug passiflora was not deemed of
sufficient value and hence, along with the Daniel preparation,
was refused recognition, are given in a report of the Council
on Pharmacy and Chemistry.—(Abstracted from The Journal
A. M. A., March 8, 1913.)
PAS-AVENA
How Its Formula Evades the Food and Drugs Act
Pas-Avena is a widely advertised “nerve sedative and hypnotic.”
The preparation is put on the market by the Pas-Avena
Company of New York City. As a headliner the
advertisements of the remedy state that the formula has
always been on every bottle, and this, The Journal states,
has a twofold object: It aims to give the impression that the
preparation is non-secret, and it is calculated to inspire confidence
in the—apparently—scientific nature of the product.
As a matter of fact, it should do neither. The preparation
is essentially secret in its composition because of the presence
in the formula of an unknown quantity and the liability
to change of formula at the whim of the manufacturer. On
the bottles some time ago the following formula was given:
| Each tablespoonful contains: |
Passiflora | 20 | minims. |
Avena sativa | 10 | minims. |
Somnalgesine (C30H28N5O6) | 2 | grains. |
The first two ingredients are plants in whose therapeutic
value but little confidence is placed. Somnalgesine, the third
constituent, is a secret preparation, the chemical formula of
which the manufacturers were kind enough to add. To a
chemist, however, the formula is absurd and impossible, and
is included either because of the manufacturer’s ignorance or
because of an intent to deceive the profession. Since the
Food and Drugs Act became law, the label of Pas-Avena has
been changed to read:
Alcohol | 8.37 | per cent. by volume. |
Anilipyrine | 16.00 | grains per fluid ounce. |
| Guaranteed under the Food and Drugs Act of June 30, 1906. |
Substitution of anilpyrine for somnalgesine gives little more
information. Chemists may recognize this as a name applied
to a mixture said to be formed by the fusion of two molecules
of antipyrin and one molecule of acetanilid. To physicians,
however, the name carries with it the same mystery as
did somnalgesine. Attention is directed to the fact that by
publishing the guarantee under the pure food laws the company
presumes to disperse all doubt and criticism, assuming
that the majority of physicians will be satisfied with the
guarantee as it stands. Inasmuch as the preparation contains
acetanilid and antipyrin, however, the manufacturers
are disregarding that part of the Food and Drugs Act which
requires that the name of the parent substance—in this case
acetanilid and antipyrin—be put in parenthesis. The laws
are so well defined that physicians appear to be content to do
nothing, firmly believing that they are safe from the defrauding
methods of unscrupulous manufacturers.—(Abstracted
from The Journal A. M. A., March 7, 1908.)
Proprietary House Insolvent—and Physicians Lose?
The Pas-Avena Chemical Company, whose product, Pas-Avena,
was exposed in The Journal a few months ago, has
recently failed, according to our pharmaceutical exchanges.
In recording the fact, one journal says:
“It is reported that considerable stock of this company
had been sold to physicians.”
At this time, when physicians are importuned daily to invest
money in various wildcat pharmaceutical concerns, this sentence
might well be used “to point a moral or adorn a tale.”
PERTUSSIN
Dr. L. A. Roberts, Dorchester, Mass., writes: “Please tell
me what the composition of Pertussin is.”
Pertussin is a proprietary whooping-cough remedy manufactured
by the Kommandantan Apotheke, Berlin. A “physician’s
sample” bottle of this preparation sent out by Lehn &
Fink bears a label on which appears the following:
| “100 parts Pertussin contains: |
| 1⁄2 | Ol. Thymi, et Thymol |
| 211⁄2 | Ext. Thymi ‘Taeschner’ |
| 50 | Saccharum |
| 2 | Glycerinum |
| 61⁄4 | Alcohol |
| 193⁄4 | Aqua Destillata” |
While it never has had much vogue in this country it has
been and still is used in Germany. It belongs to that class
of vegetable preparations which, since they contain no distinctive
principle, are difficult to analyze—particularly as
concerns the “joker” in the formula, in this case “Ol. Thymi,
et Thymol” and “Ext. Thymi ‘Taeschner’”—hence there has
been much dispute in Germany as to the composition of
this nostrum. In general, it appears that whatever virtues it
has are due to some preparation of common thyme in a menstruum
containing water, sugar and alcohol. At one time the
preparation was found to contain potassium bromid; but
tests recently made in the A. M. A. Chemical Laboratory indicated
the absence of either bromids or iodids.—(From The
Journal A. M. A., March 8, 1913.)
PHENALGIN—A TYPICAL EXAMPLE[AN]
Last June112 we devoted considerable space to the extravagant
therapeutic claims made for “Phenalgin” by its venders.
At this time we propose to refer to the misinformation—to
use a conservative term—that the Etna Chemical Company
has promulgated regarding the composition of their preparation.
In June, 1905, the Council on Pharmacy and Chemistry
officially published to the medical profession of the United
States the information that repeated examinations showed
that “Phenalgin” is a simple mixture of acetanilid and sodium
bicarbonate or ammonium carbonate. So far as we know, no
direct denial of the truth of this has been made. There has
appeared what we presume is meant as an answer; it is
couched in this sentence,
Phenalgin is just what we have always said it to be.
From this expression—which has been repeated in bold,
black letters in practically all the advertisements since last
June—we presume that we are to understand that in the
past they have stated what it is.
It would have been just as easy and more satisfactory if
the Phenalgin people, instead of saying: “Phenalgin is just
what we have always said it to be,” had said what it is,
since the average physician has neither the time nor the
inclination to look up their literature.
For the benefit of those who desire to know what the venders
of Phenalgin “have said it to be,” we have gone over their
advertising literature of the past, with the following results,
which are in the form of quotations from their advertisements:
An American Coal-Tar Product—Phenalgin—the only synthetic stimulant,
non-toxic, antipyretic, analgesic and hypnotic.
Phenalgin is the ONLY ammoniated Synthetic Coal-Tar Product made
from Chemically Pure Materials [What have the Ammonol people to
say to this?—Ed.]
A synthetic Coal-Tar Product of the Amido-Benzine series, containing
Nascent Ammonia.
These two chemicals [“stimulant ammonia of coal-tar origin” and
“chemically pure phenylacetamid”] combine under certain conditions so
as to obtain a produce which he [Dr. Cyrus Edson] named Phenalgin
or Ammoniated Phenylacetamide.
Phenalgin is a compound of peculiar character which can not be
extemporaneously made into tablets from the powdered drug, without
seriously changing and impairing its medicinal qualities.
We believe these quotations are sufficient to show what the
Etna Chemical Company has “always said it to be.” In going
over the literature for several years past we find the above
stated in the same, or similar, words in nearly all of it.
From the above four statements may be deduced: 1. They
have stated that Phenalgin is a synthetic113 preparation; 2,
they have conveyed the impression that Phenalgin is a chemical
compound; 3, they have announced repeatedly that it is
the “only” preparation of the kind, and 4, they have claimed
that Phenalgin is non-toxic.
We believe that these four statements represent in plain
English what the above quotations mean. They are all absolutely
false. Phenalgin is not synthetic; it is not a chemical
compound; it is not the only ammoniated phenylacetamide,
or the only acetanilid mixture containing carbonate of
ammonium—and it is most positively toxic.
In one place it is stated that Dr. Cyrus Edson
Employed his great facilities for chemical research and opportunities
for chemical experiment for the purpose of producing a formula
for a combination of stimulant ammonia of coal-tar origin (sic) and
chemically pure phenylacetamide, also a coal-tar product ... which
he named phenalgin, or ammoniated phenylacetamide.
In another place we read that Phenalgin is made
Under the immediate personal supervision of the original inventor of
ammoniated coal-tar products.
By comparing this last quotation—which is from a current—1905—advertisement—with
the preceding one it will be
noticed that we are asked to believe that Phenalgin is made
“under the immediate supervision of” Dr. Cyrus Edson—and
yet Dr. Cyrus Edson died Dec. 2, 1903. This is equal
to Lydia Pinkham’s prescribing for the suffering women of
America when the dear old soul had been dead for over
twenty years.
We have before us a full-page advertisement taken from
a recent number of a weekly medical journal, which possibly
is meant as an answer to the announcement of the Council
on Pharmacy and Chemistry that Phenalgin is a simple
acetanilid mixture. The advertisement is divided into two
parts; the first part is as follows:
Facts About Acetanilidum (Ancient History)
It has long been recognized that Acetanilidum and most
other coal-tar products are apt to exert a depressing
influence upon the heart, but there has never been any
doubt about its great value as a pain reliever and
temperature reducer. Its therapeutic value has, however,
been practically nullified by the danger of cyanosis and
other evils caused by its well-known depressant action
and the difficulty of obtaining it in a pure state. It
being known that certain deleterious substances are often
to be found in Commercial Acetanilidum and that much of
the injurious effect attributed to this drug is entirely
traceable to these impurities.114
The above are also falsehoods. The therapeutic value
of acetanilid is not “practically nullified ... by the difficulty
of obtaining it in a pure state.” Neither is it true
that “much of the injurious effect attributed to this drug
is entirely traceable to these impurities.” While deleterious
substances may be found in commercial acetanilid, they are
not found in the substance offered as medicinally pure
acetanilid by reputable firms. Pure medicinal acetanilid is
a cheap article, costing less than 30 cents a pound, for it is
a substance that is easily and cheaply purified. It is a
fact that the injurious effects are in the acetanilid itself and
not in the impurities it may occasionally contain.
The second half of the advertisement in part is as follows:
Facts About Phenalgin (Modern Science)
More than a decade ago the late Dr. Cyrus Edson, then
Health Commissioner for New York City and New York State,
recognizing the value of chemically pure Acetanilidum as
a therapeutic agent, if it could be deprived of its
depressant quality, employed his great facilities for
chemical research and opportunities for chemical
experiment for the purpose of producing a formula for a
combination of Stimulant Ammonia of coal-tar origin and
chemically pure Phenylacetamide, also a coal-tar product.
These two chemicals combine under certain conditions so
as to obtain a product which he named Phenalgin or
Ammoniated Phenylacetamide.
There is more of the same character. In the first place,
we call attention to the fact that “Phenylacetamide” is substituted
for “Acetanilidum” when it is to go into Phenalgin.
To mystify is one of the “tricks of the trade.” Few physicians
keep up with chemical terms and, therefore, are not
supposed to know that Phenylacetamide is one of the chemical
names for Acetanilid.
The reference here to Dr. Cyrus Edson brings up another
fact, and that is that the Etna Chemical Company tries to
convey the idea that Dr. Edson was the originator of
Phenalgin. We have always understood that Dr. Cyrus
Edson had something to do with pushing Ammonol and,
if we remember rightly, got into some trouble thereby. We
do not know the exact facts, but the following letter shows
that he had a leaning toward another “ammoniated phenylacetamid.”
The letter is dated “New York, Oct. 6, 1894,”
and is addressed to the “Ammonol Chemical Company.”
“During the past six or eight months I have used Ammonol extensively
in my private practice. I have found it excellent in the treatment of
neuralgias and for rheumatism. I have also verified your statement
in two cases that were suffering from alcoholism. My experience justifies
me in saying that it is the safest and best of the analgesic coal-tar
derivatives.
“Very truly yours,
Cyrus Edson, M.D.”
It may be of interest to know that the principal member
of the firm of the Etna Chemical Company was at one time
a member of the Ammonol Company, and it is usually
understood, we believe, that Phenalgin is practically the same
as Ammonol—in fact, the analyses published regarding the
two preparations show this to be a fact.
We must make one more quotation:
It makes little difference to a physician whether Phenalgin is a
mixture or a compound or a synthetic, with a name that would destroy
the orthographic balance of the universe, provided it is just what he
has always found it to be.
Very complimentary to the intelligence and common sense
of physicians, is it not?
Suppose some fellow should get up a scheme to exploit a
mixture of quinin and some cheap, harmless substance, say,
starch—equal parts of each. Suppose he gives it a fanciful
name, puts it on the market at a high price, say $1.25 an
ounce, and announces it as a new synthetic with wonderful
therapeutic qualities. Suppose that the schemer then adopts
the nostrum vender’s methods of fooling physicians into
using his product by getting some to give testimonials, others
to furnish write-ups, and then subsidizes medical journals
through liberal advertising to print both the testimonials
and the write-ups. The preparation would, of course, prove
to be a good thing if it were used in liberal quantities where
quinin would ordinarily be used, and some patients using it
would get well even if quinin were not indicated. Then
with the psychologic effect of the testimonials, the write-ups,
and good, strong claims rightly pushed, unthinking physicians
would do the rest. And then, after a while, when the
schemer had gotten to the point where, each year, he was
making a fortune out of his preparation, suppose some “self-appointed
chemists” should examine into the preparation and
discover that it was nothing but quinin and starch, and so
announce to the doctors of the country; what would the
doctors say? That it makes little difference “provided
it is just what he has always found it to be!”
This analogy is not far-fetched, for it is practically what
has been done with Phenalgin. One difference is that since
quinin costs as much per ounce as acetanilid does per pound,
the profits on the acetanilid mixture would be sixteen times
greater than that of our imaginary preparation. Another
difference is that acetanilid is really a dangerous drug, unless
used with care, both in its immediate and in its remote
effects; quinin is far less so.
“Little difference” indeed, whether we are being buncoed
or not! Evidently!
In conclusion, we charge the Etna Chemical Company with
intentionally misleading and deceiving the members of the
medical profession, in that the said company has in its literature
and its advertisements conveyed the impression (whether
directly stated or not): First, that its preparation, Phenalgin,
is a synthetic compound; second, that Phenalgin requires
special skill in its preparation; third, that Phenalgin has
therapeutic values which it does not possess; and, fourth,
that Phenalgin is non-toxic.
We also charge that on account of these and other misrepresentations,
this company has inveigled physicians into
prescribing and using a simple mechanical mixture of common
well-known cheap drugs—for which an extravagantly
high price is charged—under the supposition that this combination
of cheap drugs is a chemical compound of special
and peculiar merit as a therapeutic agent, and, therefore,
worthy of their confidence.
Our object in again giving space to this preparation—and
practically all we have said applies to the other acetanilid
mixtures that are exploited under fictitious names or as
chemical compounds (such as ammonol, antikamnia and salacetin
or sal-codeia—Bell)—is to impress on physicians, by
a typical example, the shamefulness of the deceptions practiced
on them by nostrum manufacturers to the great injury
of the public and of the medical profession.
A Pharmaceutical Secret Which Should Not Be Lost
Dr. Gregory Costigan, New York City, writes under date
of January 21, as follows:
“I have been carefully reading and enthusiastically approving
your articles on the nostrum evil, and have been impressed
more than usual on the existence of quack advertising in
medical journals as set forth in last paragraph and quotation
on page 206, bottom of first column, of your issue of
Jan. 20, 1906.
“In Merck’s Archives, page 11, we are told in an advertisement
on ‘Phenalgin’ that it ‘is a compound of peculiar character
which cannot be extemporaneously made from powdered
drug’ and ‘our process of manufacturing tablets is
coincident with the manufacture of Phenalgin and is the
result of a long series of careful experiments by which we
are able to produce tablets of Phenalgin in a friable condition
without losing any of its volatile constituents or undergoing
chemical changes from heat or moisture’! Inasmuch
as Phenalgin tablets are not covered with a waterproof
coating I think this is a remarkable statement to make, and
the manufacturing of a drug coincident with the manufacture
of a tablet must be a very remarkable performance, especially
because it ‘retains the full therapeutic value of the drug
unimpaired’ while the advertisement asserts that no other
manufacturer is cognizant of this wonderful method. This
ad. is for the perusal of physicians only. The Etna Chemical
Company owes it to the medical and pharmaceutical world
not to let this secret die with the company’s dissolution. It
owes it as a duty to the coming generations of science immediately
to jot down the full data of this wonderful performance,
to put it away in an age-proof safe and not allow
it to be lost to humanity as were a great many other arts
that were well known to the ancients. Let them keep it
secret now and profit by it, but do not let it be lost to
posterity.”—(From The Journal A. M. A., Jan. 13, 1906, and
Jan. 29, 1906.)
An Ethical (?) Proprietary Exploited Under Fraudulent
and Lying Claims
“PHENALGIN IS JUST WHAT WE HAVE ALWAYS
SAID IT TO BE.”
—Etna Chemical Co. in 1905.
| | | |
“Phenalgin is a synthetic
coal-tar product.”
—Etna Chem. Co. in 1898. |
“Unlike the coal-tar synthetic, phenalgin is a stimulant rather than a depressant.”
—Etna Chem. Co. in 1910. |
| TEMPUS OMNIA REVELAT! |
“Phenalgin is a synthetic coal-tar product”—thus ran the
advertisements some years ago, when the medical profession
was willing to take—or was compelled to take—the word of
the manufacturer of proprietary remedies at its face value.
Then the Council on Pharmacy and Chemistry was brought
into existence. One of the first pieces of work done by the
Council was the publication of the results of a number of
analyses of headache powders. Phenalgin was among them.
Analysis showed that Phenalgin was not a synthetic but a
simple mixture of the following ingredients in the proportions
given:
Acetanilid | 57 parts |
Sodium bicarbonate | 29 parts |
Ammonium carbonate | 10 parts |
The Etna Chemical Company, which puts out this product,
was considerably disturbed by the Council’s exposure. It
“came back” at the American Medical Association with the
slogan “Phenalgin is just what we have always said it to be.”
What, up to that time, the Etna Chemical Company had
“always said” Phenalgin to be, was:
1.—Phenalgin is a synthetic.
2.—Phenalgin is the only preparation of the kind.
3.—Phenalgin is non-toxic.
These, in brief, were the three things that Phenalgin had
been asserted to be. Each statement has been proved to be
a definite and unequivocal falsehood. Phenalgin is not and
never was a “synthetic.” Phenalgin is not and never was the
only acetanilid mixture containing carbonate of ammonium.
Phenalgin is not and never was in any sense of the word
non-toxic. Phenalgin, in short, possesses the properties—both
good and bad—that are common to acetanilid. It is a
mixture that the merest tyro in pharmacy could dispense
and for which any sophomore medical student could write
a prescription without stopping to think. Acetanilid sells at
8 cents an ounce wholesale; Phenalgin at $1.00 an ounce,
wholesale.
All these facts and many more were given to the profession
by the Council on Pharmacy and Chemistry in The
Journal more than six years ago—before even the Food and
Drugs Act came into effect. After that law became operative,
the Etna Chemical Company was compelled to say
something on the label that it had never said before, namely,
that Phenalgin contained 50 per cent. acetanilid. But the
law not only required them to add a fact to their label, but
it also compelled them to remove a falsehood. When the
pure food law went into effect, Phenalgin was labeled a
“malaria germicide.” It is not a malaria germicide and
never was a malaria germicide, and the Etna Chemical Company
dared not risk taking the question into court so it
removed the statement.
Unfortunately the Food and Drugs Act exercises no control
over the lying statements that may be made for drugs
elsewhere than on the label. So it is that physicians within
the last two or three weeks have received a booklet on
Phenalgin containing the following assertions for this
acetanilid mixture:
“Without the slightest harm, injury or depressing effect.”
“Is never followed by depression.”
“Its prolonged administration does not give rise to destructive blood
metamorphosis.”
“Is of great value in the treatment of neuralgia (especially in the
anemic.)”
“Freedom from the deleterious action or habit-forming tendencies
of the opiates.”
“It aids in destroying the malarial parasite.”
“Safest and most dependable of analgesics.”
It will be seen by this that while the Food and Drugs Act
has forced a certain degree of truthfulness on the Phenalgin
labels, the advertising matter is as fraudulent and as untruthful
as ever it was. It is true that the assertion that it is a
synthetic is no longer made, possibly because the medical
profession has been so thoroughly enlightened on the much-overworked
“synthetic” fraud that the falsehood is no longer
profitable. In other respects, the assertions are just as false
as ever. It is said to have no depressing effect—and yet it
is acetanilid. It is said to produce no habit—and yet it is
acetanilid. It is said to have no injurious effect on the blood—and
yet it is acetanilid. It is said to be the safest analgesic—and
yet it is acetanilid. How long will the medical profession
continue to be hoodwinked by means of such transparent
falsehoods?
The Phenalgin concern takes much credit to itself because
on the cartons in which the bottles of Phenalgin come, it is
stated that the product is “for dispensing purposes only.”
Yet, as a matter of fact, practically any layman can go to
any drug store and obtain this product, for the druggist
appraises this spectacular piece of Pecksniffian virtue at its
face value—a joke. Why, if intended only for physicians,
would it be necessary to include with every bottle a circular
naming the diseases, for which this acetanilid mixture is
supposed to be good—“headache,” “colds,” “lumbago,”
“scanty menstruation,” “pain in any part of the body”—and
why is the name of the product and of the firm making it
blown into the bottle?
To sum up then, Phenalgin is as big a humbug as Peruna
ever was. It is sold to-day under claims that are just as
false as those used six years ago. The Etna Chemical Company
is perpetrating a stupendous fraud on the medical profession
to-day and it is doing it not only through the agency
of the United States mail, but with the aid and support of
the following medical journals—and others—in which the
Phenalgin advertisement appears:
| Medical Record | American Journal of Obstetrics |
| New York Medical Journal | Medical Century |
| Pediatrics | Pacific Medical Journal |
| Lancet-Clinic | Dietetic and Hygienic Gazette |
| American Journal of Surgery | Medical Standard |
| International Journal of Surgery | Eclectic Medical Journal |
| American Medicine | Am. Jour. of Clinical Medicine |
It is conceivable that in some cases it is not easy for those
editors and publishers of medical journals who insist on
relying on their own judgment to satisfy themselves that
certain preparations are not worthy of being advertised. No
such difficulty occurs in the case of Phenalgin. Here the
issues are clear cut. The product is exploited under claims
that are both false and vicious and their falsity and viciousness
are perfectly evident to any freshman medical student.
The only charitable explanation of the appearance of the
Phenalgin advertisements in the medical journals listed is
that the editors and publishers have not given the subject the
attention it deserves and to which their readers are entitled.
Perhaps it would help if their attention were called to the
matter by their subscribers.—(From The Journal A. M. A.,
Jan. 27, 1912.)
PHENO-BROMATE
An analysis of this preparation made at the instance of the
New Haven Medical Association, by its chemist, and sent by
Dr. Charles J. Foote of New Haven to The Journal is in
part as follows:
The package was marked “Sample package, Pheno-Bromate.
The Pheno-Bromate Company, New York,
U. S. A.” The box contained a number of tablets and a
package of powders in papers marked, “Physicians’ 10 grain
powders, Pheno-Bromate.” The substance in the papers was
a white crystalline powder not homogeneous. It was completely
soluble in hot water. The hot water solution on
cooling yielded a mass of thin crystalline plates. This
material was found to melt at 113.5 C. It gave no color
with ferric chlorid and a positive isonitril test. The portion
insoluble in ether amounted to 49.8 per cent. of the powder
and consisted of potassium bromid. Quantitative determinations
of potassium and bromin in the original solution
confirmed this result. In my opinion, the powder consists
of approximately equal quantities of acetanilid and potassium
bromid. Qualitative tests of the tablets indicated that
they had the same composition except for a small quantity
of some excipient not entirely soluble in water. Yours
truly,
Herbert E. Smith,
Chemist New Haven Medical Association.
Before the Food and Drugs Act Pheno-Bromate was advertised
as “a synthetic combination of the phenetidin and bromid
groups, and not, as is the case with many analgesics and
antipyretics, a mixture of various coal-tar derivatives” and
as “the safest and best of all sedatives.” The dose recommended
in most cases is 20 grains—equal to 10 grains each
of acetanilid and potassium bromid. Since the Food and
Drugs Act has gone into effect its label states that it is “a
perfect combination of a phenol and bromin derivative containing
282 grains of acetphenetidin, U. S. P., per ounce.”
What a boon it was to mendacious manufacturers that the
patent rights on phenacetin expired before the Food and
Drugs Act went into effect.—(Abstracted from The Journal
A. M. A., July 14, 1906, and April 18, 1908.)
PHENOLPHTHALEIN
Phenolphthalein has long been used as an indicator in
chemical reactions, but its use as a therapeutic agent115 is
comparatively new. When its laxative properties were first
discovered it was exploited as a proprietary in Germany, and
it was not long before the enterprising manufacturers in
this country saw in it a potential gold mine and now nearly
every proprietary drug manufacturer in this country has
coined a proprietary name for it and is exploiting it, either
alone or in combination with one or more other laxatives, and
with more or less unwarranted claims.
Phenolphthalein itself has certain pretty well defined properties,
but when a little of some other drug has been added
wonderful therapeutic possibilities are claimed for the combination.
The drug also has a definite market value and the
pure substance in the form of powder, tablets or pills could
not be sold at a price greatly in excess of the market value.
Thus manufacturers, from business policy, add to it other
drugs. There are now on the market numerous more or less
secret and “fancy” preparations of phenolphthalein for which
a price is charged out of all proportion to the value of the
preparation. Among these are:
Phenolphthalein Laxative (El Zernac Co.).
Exurgine (Bischoff & Co.).
Probilin (Schering & Glatz).
Prunoids (Sultan Drug Co.).
Laxine (Columbus Pharmacal Co.).
Phenolax Wafers (Upjohn Co.).
Laxaphen (Parke, Davis & Co.).
Phenalein (Pax Chemical Co.).
Thalosen (Abbott Alkaloidal Co.).
Laxothalen Tablets (Pitman-Myers Co.).
Veracolate (Marcy Co.).
And additional preparations are still coming out! Some of
the preparations contain only the phenolphthalein with a
coined non-descriptive proprietary name attached, but most of
them contain in addition one or more of such drugs as cascara,
sulphur, prune, senna, salicylic acid, ipecac and aromatics.
The exploitation of phenolphthalein in this way gives
opportunity to the manufacturers to make all sorts of strong
claims, some of them directly contradictory, for their preparations.
For instance, Phenolax, which is said to contain
phenolphthalein and cane sugar, is claimed to be “a great
success for all forms of constipation, intestinal atony and
hepatic torpor.” Of Laxothalen, which is said to contain
phenolphthalein, aromatics and sugar, it is stated that “its
action is confined to the bowel and it has practically no
hepatic action.” Of Prunoids, which is said to contain
phenolphthalein, cascara, de-emetized ipecac and prunes,
we have the old familiar statement that “the harmonious
blending of the several ingredients will give results that
cannot be obtained through their use separately, nor will
their use be followed by after-constipation.”
At the time phenolphthalein was beginning to be exploited
in this country The Journal116 suggested that physicians who
wished to try the remedy should prescribe it under its own
name and not under fancy, coined names. Since phenolphthalein
occurs in the form of an insoluble and tasteless
powder there is no reason why special pharmaceutical preparations
of it should be placed on the market. It can be
prescribed in powder form, in pills, capsules or tablets. Thus
given, the true therapeutic action of the drug would be
apparent and its actual value arrived at.
The vice of this unscientific habit of prescribing names
instead of drugs is stated in a forcible way in a letter received
from Dr. V. E. Simpson, a teacher of materia medica and
therapeutics in the medical department of the University of
Louisville. He says:
“Recently P. D. & Co.’s representative left on my desk a
sample labeled ‘Laxaphen.’ The formula given is: phenolphthalein,
gr. viii; salicylic acid, gr. 3⁄5, in each fluidounce,
‘incorporated in a palatable chocolate base.’ Now, in the first
place, this name is one that the public will easily learn and
will soon call for; in the second place, it is not a name that
carries with it even a suggestion of its contents; and, finally,
the physician acquires the habit of mechanically prescribing
names instead of drugs, and in the burdening of his memory
with the myriad of fantastic labelings he finds it impossible
to remember even the drugs any one contains, much less the
exact proportions of those drugs. Then suppose that a consultation
is had; the consultant asks what is being given and
the attendant answers that he is giving ‘laxaphen.’ The consultant,
perhaps, has not been sampled and inquires about it;
the attendant must answer. ‘Oh, it contains some phenolphthalein
and a salicylate, but I have forgotten the exact proportions.
I have the literature on my desk.’ Had he used
U. S. P. and N. F. remedies, which the consultant and every
other doctor in the land has access to and should have some
knowledge of, this embarrassment would not occur.”
All of the above should remind the physician that he should
write simple prescriptions, for drugs whose action he knows,
adapted to the particular case and not for money-making
combinations under fanciful, non-descriptive names exploited
by the proprietary manufacturers. In this way he will not
only save money for himself and his patients, but he will be
giving them exact and effective treatment, he will know
exactly what he is giving and learn for himself its effect, and
he will be following the only method which entitles him to be
called a scientific physician.—(From The Journal A. M. A.,
April 30, 1910.)
MIXED VACCINE AND PHYLACOGENS[AO]
The noted advance in therapeutics shown in the development
of vaccine therapy has brought with it grave dangers as
well as advantages. We have, on a number of occasions,
discussed in special articles and in editorials the dangers
which threatened from the rapid commercialization of this
new method. The unscientific character of mixed vaccines
and of the mixed filtered products of a number of vaccines
marketed as “Phylacogens” has been especially emphasized
and the danger from their indiscriminate use pointed out.
A little over a year ago we published a series of articles dealing
with the whole subject in which the nature of mixed vaccines
was described117 as follows:
“The mixed stock vaccine of commerce is a makeshift. It
is offered as a substitute for correct diagnosis. Like all
such makeshifts in science, it is doomed to failure.... A
burden is being forced on the profession which will
speedily assume the proportions attained by proprietary drug
combinations. The menace cannot be counteracted unless
physicians will accept the guidance of unselfish, non-commercial
interests and refuse to purchase and use mixed
commercial vaccines.”
This admonition to seek the guidance of unbiased scientific
observers is deserving of special emphasis at the present
time. Five weeks ago we published the address of the chairman
of the Section on Pharmacology and Therapeutics, Dr.
John F. Anderson,118 one of our foremost workers in this
branch of biologic science, in which attention was very
forcibly drawn to the dangers involved in the use of biologic
products of non-specific character. He says:
“Bacterial therapy undoubtedly in some cases is a most
valuable method of treatment; but when the claim is made
that a combination of the dead bodies or the filtered products
of a number of different bacteria are useful for the treatment
of certain diseases with a different specific cause, it
would seem that the suggestion closely approaches quackery.”
Further he says:
“Aside from the doubtful practice of the indiscriminate use
of unproved methods of treatment, it has seemed to me that
a great injustice is done the patient by their use, since some
of the preparations that have been widely exploited have
been shown to be harmful in certain instances and even to
have caused death. So the first step in attempting to remedy
conditions is to awaken the physician to the importance of
ignoring the claims of those who are pushing these new
methods until their usefulness and harmlessness has been
clearly demonstrated by those best in a position to do so.”
As a result of scientific methods in teaching therapeutics,
physicians have gradually given up almost entirely the use
of “shotgun” prescriptions and now prescribe a drug or a
combination of a few drugs, each given for the purpose
of exerting a definite action. On the other hand, the purveyors
of bacterial vaccines have gradually increased the
number of different bacteria in their mixed vaccines until
some of those now advertised for sale contain as many as
seven different kinds of bacteria, and some of the “Phylacogens”
contain the filtered products of at least eleven bacterial
species!
Under the present federal laws there does not seem to be
any way in which the federal government can do more than
is being done at present. It is a case in which the physician
becomes the sole guardian of the patient committed to his
care. He is the one and the only responsible individual. He
cannot throw the blame for bad results back to the manufacturer.
When he subjects his patient to the possibility of harm
by the use of these unscientific and dangerous preparations,
the physician assumes the responsibility, whether he wants
to or not.
If physicians would report their failures when these
vaccines are used, and especially report the fatalities consequent
on their use, with the name of the manufacturer of the
particular product used, we are quite sure there would result
lessening in the enthusiasm of the purveyors of these products.
When tempted by the optimistic statements of the interested
manufacturer of these mixtures to give them a trial the
physician should remember that the warnings of disinterested
scientists are of far more value than uncritical clinical
reports put out under commercial auspices.
This we quote from a recent book by Victor C. Vaughan,119
President of the American Medical Association:
“Every time an unbroken protein is introduced into the
body it carries with it, and as a part of it, a poison. From
the very careless, rash, and unwarranted way in which
‘vaccines’ of most diverse origin and composition are now
used in the treatment of disease, this matter certainly cannot
be understood or its danger appreciated by those who subject
their patients to such risk. It should be clearly understood
that all proteins contain a poisonous group—a substance
which in a dose of 0.5 mg. injected intravenously kills a
guinea-pig. This poison is present in all the so-called ‘vaccines’
now so largely used, and it is not strange that death
occasionally follows the use of ‘Phylacogen’ or similar
preparations. Not only do these proteins contain a poison,
but when introduced parenterally the poison is set free, not
in the stomach, from which it may be removed, but in the
blood and tissues. It is possible that vaccine therapy may
become of great service in the treatment of disease. Even
now there are occasional brilliant results which are reported
while the failures and disasters are not so widely advertised.”
Such a warning as this quotation contains, from a man so
eminent as Dr. Vaughan, merits and should receive the careful
attention of medical men; at least it should have as much
weight as the “clinical evidence” spread broadcast among
our profession by commercial houses.—(From The Journal
A. M. A., Aug. 29, 1914.)
THE DANGER IN PROTONUCLEIN, A PREPARATION
CONTAINING THYROID
Protonuclein was the subject of a little article in our
Queries and Minor Notes Department, Nov. 16, 1912, page
1812. Dr. Reid Hunt, Washington, D. C., writes:
“To the Editor:—I have been requested by a physician to call
your attention to certain statements which might well have
been added to your reply to J. A. C. in regard to Protonuclein.
Dr. Seidell and I examined several samples of Protonuclein
some time ago120 and by chemical and physiologic tests found
that they contained the equivalent of 10 per cent. thyroid
of 0.1 per cent. iodin strength (the actual amount of thyroid
may have been greater or less for we did not know the percentage
of iodin in the thyroid used). The dose recommended
on the bottle was 6 to 12 grains every three or four
hours; this represents from 0.6 to 1.2 grains of some of the
commercial thyroid powders, and is sufficient to cause pronounced
thyroid effects in many conditions. Protonuclein was
advertised as a ‘perfectly harmless antitoxin, tissue-builder,’
etc., although the dose of thyroid did not differ materially
from that in ‘Rengo’ and ‘Marmola,’ two anti-fat nostrums
which we examined at the same time. We called attention to
the danger of using thyroid, the most powerful tissue-destroying
drug known, in cases of typhoid, phthisis, etc., for which
protonuclein was recommended, though these are conditions
in which the physician is supposed to be exerting every effort
to build up the tissues.
“You also speak of the ‘high’ nuclein content (0.28 per
cent. phosphorus): the largest recommended dose would contain
only about 1⁄3 grain of nucleic acid—an amount which
probably has not the slightest effect, especially when given
by the mouth.
“A sample of ‘Protonuclein Special’ was found to have twice
as much thyroid as the ordinary Protonuclein; this also was
stated to be ‘perfectly harmless.’”—(From The Journal
A. M. A., Feb. 1, 1913.)
PURGEN
The physicians of the United States are receiving a neat
package containing samples of a German proprietary—Purgen.
The container is an ingenious one and, besides the tablets,
includes a circular in English, although mailed in
Europe, describing the remarkable virtues of this “new synthetic
aperient.” It has been considered strange that this proprietary,
which has been advertised so thoroughly in Europe,
Australia, etc., should not have made its appearance in this
country. Now it is here, and it is well that physicians
should know what Purgen is and not be mystified and misled
by the literature that they may receive regarding the preparation.
The following appeared in The Journal, Jan. 5, 1907, page
64, and is reprinted now as being especially timely:
The report of a case of poisoning by Purgen (phenolphthalein)
is the occasion for some pertinent observations
by Dr. G. Brasch as to the proper introduction of such
remedies to the medical profession (Ztschrift für Medizinalbeamte,
Abst. in Apotheker-Zeitung, No. 59, 1906). He
agrees with Best that all such remedies should first
receive a thorough trial in an institution subject to state
supervision, before they are advertised to the medical
profession, so that their harmlessness in appropriate doses
may be ascertained by a method free from liability to
error. The manner in which the manufacturers introduced
Purgen to the profession and the laity is to be
condemned, and probably led to the symptoms of poisoning
exhibited in the case of Dr. Best and tends to discredit
a remedy which is harmless and efficient if used in
proper doses. The manufacturer of such a preparation is
inclined, for obvious reasons, to put the dose of his preparation
much too high. The most important point, however,
is the objectionable character of the names given to
such articles. The organic compound phenolphthalein has
been known for a long time and has been widely used as
an indicator. Accidentally it was discovered that phenolphthalein
possessed laxative properties and thereon
it was proposed (1901) as a medicine under the name
“Purgen.” It is sold in tablets containing 0.05, 0.1 and
0.5 grain phenolphthalein mixed with sugar and flavored
with vanilla. The author says: “But it is very desirable—and
I regard this as the most important part of my
communication—that phenolphthalein should be received
into the materia medica under its own name. The addition
of vanilla and sugar is to the highest degree superfluous
and the arbitrary dosage in three strengths with
the ridiculous designations, ‘baby,’ ‘for adults,’ ‘for patients
confined to bed,’ are merely calculated to prejudice the
physician who is accustomed to individualize in his prescriptions,
against a remedy which is in itself an excellent
one.”
As explanatory to the last sentence, it should be stated
that in Europe Purgen is put up in three dosage forms,
“infant Purgen for children,” containing 3⁄4 of a grain;
“adult Purgen for chronic constipation,” containing 11⁄2
grains, and “strong Purgen for invalids,” containing 71⁄2
grains. The form in which it is being sampled in this country
is in the medium dose, 11⁄2 grains.
Physicians should remember that the promoters of Purgen
are simply introducing a chemical well known to laboratory
workers for the last twenty years, which has been recognized
as an aperient for at least seven years, and which can
be purchased for 40 cents an ounce, whereas an ounce of
phenolphthalein in the form of Purgen will cost $3.20 wholesale.
The enthusiastic praise of the remedy, found in the
advertising circulars, should be subjected to critical judgment
on account of its source and motives.
It is undoubtedly true, however, as we have previously
stated, that phenolphthalein is worthy of a trial. In the British
Medical Journal, Oct. 18, 1902, F. W. Tunnicliffe speaks
of the virtues of phenolphthalein, and the conclusions reached
by him were that it is a useful aperient, without irritating
action on the kidneys, and is especially valuable in jaundice,
its depressing action on the circulation being less than sulphate
of magnesia.
Phenolphthalein is not in the Pharmacopeia, but has been
included in “New and Nonofficial Remedies” by the Council
on Pharmacy and Chemistry. From this we quote:
Actions and Uses.—Phenolphthalein acts as a purgative,
but appears to possess no further physiologic action. A
case of poisoning from taking 1 gm. (15 grains) is reported.
Dosage.—For adults the average dose is 0.1 to 0.2 gm.
(1.5 to 3 grains) given as powder, in cachets, capsules or
pills. It may be given with safety in doses of 0.5 gm.
(8 grains), and these doses seem to be necessary to secure
its effects in bedridden patients or in obstinate cases.
We have gone into this matter again so that our readers
may have some knowledge of this remedy, and we hope that
if they conclude to try it they will use the chemical itself and
under its own name.—(From The Journal A. M. A., Sept. 14,
1907.)
PYO-ATOXIN
“To the Editor:—I am sending you a sample of a proprietary
preparation that for the past two or three years has been
largely retailed in the South and Southwest as a new combination
that liberates larger amounts of formaldehyd, etc.,
in the genito-urinary tract than any known agent, that it is
a methylene-formate, entirely new, etc.
“I asked the representative why he had not submitted a
specimen to the Council, and his reply was that like Wyeth
and others they did not get a fair report, or something to
this effect. My reasons for trying to find the truth for their
claims is that quite a number of general practitioners have
asked me regarding this Pyo-Atoxin.
“W. P. Dey, M.D., Jacksonville, Fla.”
Dr. Dey sent with the foregoing letter a box of Pyo-Atoxin
which bore this label:
Pyo-Atoxin
Reg. in U. S. Pat. Office
(Capsules)
(Pheno-Methylene-Formate)
“Hurley”
An Antitoxic Agent Indicated in
Gonorrhoea, Cystitis, Pyelitis and
Bacteriuric Conditions.
DOSE: One capsule four to six times daily,
Followed by large glass of water.
Guaranteed by
H. O. Hurley,
Manufacturing Pharmacist,
Louisville, Ky.
Under the Food and Drugs Act, June 30, 1906
Serial No. 1710.
The pseudoscientific synonym “pheno-methylene-formate”
carries the idea that Pyo-Atoxin is a definite chemical substance.
It is unnecessary to say that the term “pheno-methylene-formate”
is a meaningless one and its use reminds
one of those preparations exploited seven or eight years ago
before the Council began to expose these mixtures masquerading
as definite chemical compounds.
The chemical laboratory was asked to investigate this
preparation and the following is a report of the chemists:
“The box contained thirty gelatin capsules coated with
some black substance giving them the appearance of some
of the popular gonorrhea nostrums. When the capsules
were opened they were found to contain a powder—about
0.35 gm. or 5 grains per capsule—composed of large white
or colorless crystals mixed with a smaller amount of a
fine dark powder. The crystals when separated out and
dissolved yielded the characteristic tests for hexamethylenamin.
A solution of the entire capsule content was deep
blue and responded to the U. S. P. tests for methylene blue.
“As a result of these and other tests it was concluded that
Pyo-Atoxin consisted essentially of two pharmacopeial
drugs—hexamethylenamin and methylene blue. A quantitative
determination of the constituents was considered
unnecessary. From its general appearance and properties,
however, the hexamethylenamin probably constitutes
approximately from 60 to 80 per cent. of the preparation.”
It thus appears that the capsules contain a mixture consisting
essentially of two well-known official substances, the
value and particularly the limitations of which should be
known by physicians by this time. This nostrum is simply
another example of how physicians are being humbugged.—(From
The Journal A. M. A., Feb. 14, 1914.)
RESINOL
The Philadelphia branch of the American Pharmaceutical
Association issued a pamphlet some two years ago in which
the following appeared relative to Resinol and similar products:
“Within recent years there have been introduced a number
of compound ointments that in their supposed range of
therapeutic usefulness are scarcely equaled and certainly
not excelled by the magic unguents of the quacks and charlatans
of continental Europe, who, several centuries ago,
essayed to cure all manner of disease by inunction or the
simple application of compound ointments of secret composition.
“As typical of this modern class of panaceas we may mention
Resinol. This preparation is being widely advertised
at the present time in the daily papers as a valuable adjunct
to Resinol Soap in the treatment of all kinds and varieties
of diseases of the skin. The makers of this particular mixture,
in the form of an ointment, modestly assert that it will
cure all skin diseases, and is also ‘Specific for Pruritus
Ani, Itching Piles, and Pruritus Vulvæ.’”—(From The Journal
A. M. A., Nov. 6, 1909.)
RESOR-BISNOL
Resor-Bisnol was considered by the Council and refused
recognition. The following formula for Resor-Bisnol was
at one time given in advertisements in a number of medical
journals:
“A scientific combination, in nicely balanced proportions of Bismuth
Salts of antiseptic acids of the aromatic series, and Resorcin.
“Each 100 parts contains 20 parts Resorcin, and 52 parts Bismuth
Oxid, combined with antiseptic acids.”
Besides this formula other “formulas” equally indefinite,
vague and misleading have been given in lieu of an actual
statement of composition, thus:
“—— is a mixture of resorcin and bismuth salts of phenic acids
such as salicylates, etc., and an aromatic alcoholate. Its composition is
as follows.
Bismuth salts of phenic acids | 60 | per cent. |
Aromatic alcoholate of bismuth | 20 | per cent. |
Resorcin | 20 | per cent.” |
The product was recently analyzed in our chemical laboratory.
The chemists report as follows:
“A specimen of Resor-Bisnol examined by us consisted
of a light brown powder possessing a characteristic odor
and a taste at first sweetish and then bitter. It was found
to be only partially soluble in water. The examination
indicates that Resor-Bisnol is probably a mixture consisting
essentially of a basic bismuth salicylate (bismuth
subsalicylate), a basic gallate of bismuth (bismuth subgallate),
a basic compound of beta-naphthol (bismuth
betanaphtholate) and resorcinol (resorcin).”
It thus appears that Resor-Bisnol is probably a simple
mixture of well-known substances. In other words, the
Resor-Bisnol advertising and literature are typical of that
issued by various nostrum houses: it conceals the truth in
a mass of semi-scientific verbiage, and while not frankly
false, it deceives by what is left unsaid rather than by
what is said.—(From The Journal A. M. A., June 1, 1912.)
ROBINOL AND SEVETOL
Revamping Discarded Theories for Commercial Purposes
It is astonishing how rapidly medical hypotheses become
theories and theories are accepted for established facts, when
such alleged facts are favorable to commercial enterprise.
Yet, as a matter of fact, the manufacturers of proprietary
preparations are under a moral obligation, at least, to tell
the truth with reference to the scientific basis for their
claims. To draw from exploded theories reasons for the
use of proprietary preparations is reprehensible, not only
because it may lead physicians to use preparations which
are worthless, but also because it tends to confirm in the
physician’s mind opinions which science has discarded.
Robinol
John Wyeth and Brother put up a mixture of glycerophosphates
which they call Robinol. In their description of
the properties of this mixture they say:
“Phosphorus exists in the brain, nervous system, and vital organs as
lecithin, of which glycerophosphoric acid is the most important constituent
and is essential to the vital processes for the reproduction of life
and maintenance of metabolism in old age, impotence, etc.”
The first impression on reading this sentence is that it
suggests that glycerophosphates are essential to the vital
processes, although the statement strictly applies to phosphorus.
The next sentence confirms this impression and the
mind glissades from the accepted fact of the existence of
phosphorus in nervous tissue to the unfounded hypothesis
that the glycerophosphates are necessary to supply the essential
element. In the next sentence the circular continues:
“In nervous and general debility the glycerophosphates as exhibited in
Robinol, are preferable to the mineral phosphates as they contribute
the essential constituent of nerve tissue and are absorbed by the cells
more readily than any phosphate of vegetable or inorganic origin.”
This statement is utterly unfounded. It is in direct opposition
to the conclusions of pharmacologists. The glycerophosphoric
acid radical is, to be sure, found in the lecithin of nervous
tissues, but its source is not known. There is no evidence
either that it must be present in the food or that it must
be taken as medicine in order that the brain and nervous
tissues shall be nourished. When the glycerophosphates are
taken there is no evidence that they enter into the composition
of the brain or nervous tissue. They are excreted in
the urine and feces as phosphates. It has never been shown
that glycerophosphates are absorbed any more readily than
other phosphates.
But the advertising circular has still more information
to impart to physicians:
“In that group of maladies characterized by faulty nutrition, due
to the excessive elimination of phosphorus from the body, as is evidenced
by the fatigue and weakness following acute attacks and present
in many chronic affections, during the course of fevers and in the later
stages of phthisis and all diseases of the nervous system, physicians
will find the tonic chalybeate properties of the glycerophosphates of great
value.”
Physicians know, if the nostrum makers do not, how difficult
it is to determine whether there is an excessive elimination
of phosphorus from the body. The bulk of the phosphates
found in the urine are derived from the food and
so little comes from the metabolism of the nervous system
that it is not easy to prove that any disease is due to excessive
elimination of phosphorus from the body. That fatigue
and weakness are due to such a loss of phosphorus is mere
assumption, a convenient theory for the exploiters of glycerophosphates.
But admitting that nervous waste or faulty
nutrition is characterized by the loss of phosphorus, it is
easier, cheaper and more rational to supply such loss by the
use of phosphorus-containing foods, such as milk and eggs,
and there is not the slightest evidence that the loss of
phosphorus will be influenced in any way by giving a supply
in the form of glycerophosphates. Thus, in order to bolster
up the sale of a simple solution of glycerophosphates, vague
theories and improbable hypotheses are dressed in all the
dignity of scientific facts.
Sevetol
There was a time, perhaps a generation ago, when physiologists
taught that fats were absorbed into the blood in the
form of a fine emulsion. This theory has been definitely
disproved and it is now known that fats enter the blood only
after a chemical splitting into glycerol and fatty acids, the
latter being, to a large extent, combined with alkalies in
the form of soaps.
“Sevetol,” another Wyeth preparation, is presented to the
profession under the claim that it is a very fine emulsion
of fats, every portion of which “is readily absorbed through
the intestinal wall.” To quote:
“The administration of Sevetol, therefore, does not tax the digestive
power of the patient, for it is absorbed with very little effort on the
part of the digestive apparatus; and even if the organs of digestion be
involved, neither the weakness of the patient nor the severity of the
symptoms necessarily contra-indicates its use. The amount ingested
is limited only by the power of assimilation exhibited in the tissues,
and it may be given in large doses for a continued period of time, or
until symptoms of overfeeding are produced, such as coated tongue,
anorexia, constipation, headache and lassitude. When these symptoms
appear, the administration of Sevetol should be temporarily discontinued
and a mild but effective laxative given for several days, after which
its use may be resumed.”
While the language just quoted is a little more exalted
and dignified than that found in typical “patent medicine”
advertisements, the thought it expresses qualifies it for a
place in the “Lydia Pinkham” or “Peruna” class. Every
sophomore medical student knows, if he gives the matter
any thought, that Sevetol must undergo the same process
of digestion as any other fat. It must be broken up into
a fatty acid and glycerol, and saponified before it can be
absorbed. It is plainly evident that the amount of Sevetol
which can be taken is limited not only by the power of
assimilation, but also by the power of digestion. The symptoms
mentioned in the advertisement and ascribed to overfeeding,
are the symptoms, not of a system saturated with
absorbed fat, but of digestive organs rebelling against an
unusual diet.
The exploitation of Sevetol is but one more case of turning
to commercial account an exploded theory. Isn’t it
about time that our profession demanded that the purveyors
of medicinal products tell the truth? And isn’t it time, too,
that we cease taking our pharmacology and therapeutics
from proprietary manufacturers?—(From The Journal
A. M. A., July 4, 1914.)
SALACETIN
Some time ago we wrote to Messrs. Bell & Co., calling their
attention to the fact that we had made an examination121 of
their product, salacetin, and that as a result of such examination
it was found to be a mixture, which did not coincide
exactly with their description of it. They replied: “Our
description of salacetin is correct and we have nothing more
to impart except that anyone publishing any different formula
from that given in our circulars will be held responsible
by us.”
The description they give is as follows:
Prepared by the interaction, with heat, of salicylic acid, glacial
acetic acid, and purified phenylamine.
This sounds very scientific, but when we remember that
acetanilid is a result of the action of glacial acetic acid on
phenylamine (anilin) their description is cute, to say the
least. Of course, there is “interaction with heat” when salicylic
acid is combining with bicarbonate of sodium to form
salicylate of sodium. Further, there is, no doubt, some
“interaction with heat” when the substances are rubbed
together in mixing them and when they are going through
the mill to form tablets, not to mention the heated imagination
of the promoters of this “synthetic.”
The following taken from the advertising literature furnished
by the manufacturers and distributed by them, is
quoted to show the claims made for this preparation:
Salacetin is free from Toluodine and produces no harmful cyanosis.
In the treatment of Acute Bronchitis, Grippe, Influenza, Tonsillitis,
Lithemic Headaches, Rheumatism and Neuralgias, it relieves pain,
reduces inflammation and abnormal temperature, and eliminates uric
acid more quickly and thoroughly than the salicylates, and without
causing depression or stomachic or renal irritation.
Have personally interviewed thousands of physicians, including every
prominent one in the East, and can honestly state that we have never
known of anything at once so efficient and so unobjectionable in the
removal of rheumatic and neuralgic pain and other symptoms of the
uric-acid accumulation.... In La Grippe and Acute Bronchitis it
relieves pain and coughing, reduces inflammation and temperature, makes
the patient comfortable, and checks the progress of the disease. In
Tonsillitis its action is specific.... In Acid Cystitis, it neutralizes
acidity, reduces inflammation and removes irritation.... In
Dysmenorrhea it relieves pain and congestion with no hallucinations,
constipation or danger of a drug habit.
In Dysmenorrhea and Ovarian Neuralgias try Sal-Codeia—Bell. It
will relieve the pain as well as morphia. It will not check any secretions,
induce any habit, cause any depression or inconvenience of any
kind.
Of course, it is well understood that acetanilid is a
valuable remedy in many instances, if used with caution and
when indicated. It certainly has some therapeutic value.
There is no doubt that it relieves pain of various kinds.
It is to be presumed that combining salicylate of sodium
with it will have certain beneficial effects in certain
rheumatic conditions, on the supposition that salicylate of
sodium and acetanilid are both used with more or less success
in certain of these conditions. Also, the combining of bicarbonate
of sodium, carbonate of ammonium, caffein, citric acid,
one or several of these, may result in a fairly good combination,
but these combinations can be found in the list of
preparations of all our large manufacturing pharmaceutical
houses, which supply them at one-tenth of the cost of these
secret remedies. The physician in using these preparations
put out by reputable recognized manufacturing pharmaceutical
houses not only is prescribing preparations that are
non-secret, but is using remedies that cost one-tenth as much
as the secret preparations, which are exploited under fanciful
names and pushed by ridiculous claims.—(From The Journal
A. M. A., July 1, 1905.)
SAL-CODEIA—BELL
According to the advertisements “Salacetin”
“... is a combination with heat of salicylic and glacial acetic
acids with phenylamine, the irritating, depressing and blood-corpuscle
destroying elements removed.”
According to the Committee on Chemistry of the Council
on Pharmacy and Chemistry of the American Medical
Association, whose report was published in The Journal of
the American Medical Association June 3, 1905, p. 1791,
“Salacetin” is a mixture of acetanilid, salicylate of sodium
and bicarbonate of sodium. Sal-Codeia (Salacetin-Codein)
therefore would be the same as above with codein added.
Of course, acetanilid and codein will relieve pain (it could
not do otherwise) and consequently make a very good combination
in certain conditions, if not used too often and if
used with care. Although the continued use of codein is not
likely to produce a drug habit, it, as well as acetanilid, does
so sometimes, and it must be remembered that codein is a
motor paralysant, and is not the best combination to be used
with acetanilid. For those who wish to give a combination
of acetanilid, salicylate of sodium and codein, the following
prescription is suggested:
℞ Acetanilid | Ʒ i 4| | |
Sodii bicarbonatis | Ʒ ss 2| | |
Sodii salicylatis | Ʒ ss 2| | |
Codein sulph. | gr. vi 0| | 4 |
| M. et div. chart No. xxiv. |
This will make five-grain powders which may be put in
papers, capsules, cachets or tablets. Each will contain 21⁄2
grains (0.15 gm.) of acetanilid and 11⁄4 grains (0.075 gm.)
each of sodium salicylate and sodium bicarbonate, with
1⁄4 grain (0.015 gm.) of codein.
The doses of acetanilid and of codein approximate the
average adult doses, but the sodium salicylate, to have any
appreciable effect, must be increased, for 11⁄4 grains of salicylate
of sodium in a dose is insignificantly small. Sodium
salicylate with acetanilid makes a fairly good combination
in certain rheumatic troubles, but it is not indicated by any
means as a cure-all, as one would judge from the literature
sent out by the Sal-Codeia-Bell people.—(From The Journal
A. M. A., Nov. 4, 1905.)
SANATOGEN
Cottage Cheese—The New Elixir of Life[AP]
The psychology of advertising is nowhere better exemplified
than in the “patent medicine” and proprietary fields. The
reason is evident. Knowing that the general tendency of the
human organism is toward health rather than toward disease
and that the “healing power of nature”—vis medicatrix naturæ—will
account for a large proportion of recoveries from
sickness, it is not to be wondered at that thousands of preparations
sold for medicinal purposes receive credit that is
entirely undeserved. The awarding of such undeserved credit
is largely due to the universal tendency of those who are not
trained in science to apply the post hoc, ergo propter hoc
argument in all matters relating to health and disease.
John Smith suffers from a passing indisposition. When he
recovers he credits his recovery to whatever he may have
done just preceding that recovery. If he has received
medical attention, the physician gets the credit; if he has
taken “absent treatment,” Christian Science is responsible;
if he has taken sugar pills, “Prof.” Munyon gets the praise—while,
as a matter of fact, if he had taken none of these he
would have recovered since he was only temporarily indisposed.
Nor are laymen the only ones that fall into such errors.
Many physicians who prescribe new, widely-advertised preparations
are likely to give those products credit for whatever
favorable change may take place in their patients’ condition.
This failing is not a modern one. In 1842 Dr. Benjamin
Brodie wrote: “We have no doubt that many well-instructed
medical practitioners have not sufficiently considered
what course a given disease would take if it were left
to itself; and as to others, it is not possible that they should
have any real knowledge on the subject. With the majority
of persons a recovery will generally pass for a cure.”
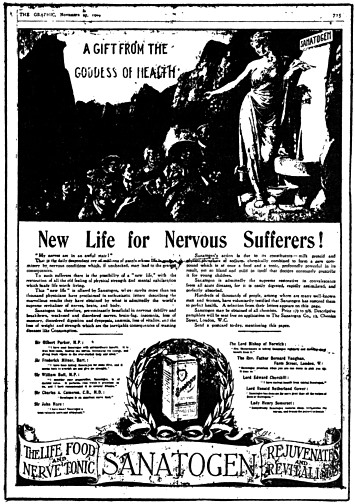
Greatly reduced photographic reproduction of a full-page Sanatogen
advertisement appearing in the London Graphic. The Graphic was
one of the London magazines that refused to accept an advertisement
of the book issued by the British Medical Association, exposing
“patent-medicine” frauds.
THE POWER OF ADVERTISING
While every physician is perfectly familiar with the facts
just stated, it seems worth while to give them as a probable
explanation of what is to follow. Within the last few years
the medical profession and the public of this country have
been asked to believe that a combination of cottage cheese—or
its equivalent—with a small amount of glycerophosphates is
capable, when sold under a proprietary name and with the
right kind of advertising, of producing physiologic effects that
are little short of marvelous.
The name of this elixir of life is Sanatogen, and it is
doubtful if the history of modern advertising furnishes any
more notable example of the commercial potentialities of
publicity than that exhibited in the exploitation of this product.
The Sanatogen advertising campaign is probably the
most skilful piece of work of its kind ever done. On both
sides of the Atlantic, every effort has been made to endow
the advertisements with a dignity which, to those who know
the very ordinary nature of the product advertised, is grotesquely
out of keeping. Only the highest-class magazines
and newspapers have been patronized; the “copy” has been
so written as to appeal not to the ignorant but to the intelligent.
Testimonials from men whose names are well known,
even though by training and education they are incompetent
to pass judgment on a product of this kind, and fulsomely
laudatory letters from men whose education and training
should have taught them better—both have been used with
all the skill of the trained publicity man. In short, Sanatogen
stands as a monument to the power of printers’ ink.
The claims for this product have already been referred to
in The Journal, but it will do no harm to bring them again
before our readers. Here are some taken from advertisements:
“The Re-Creator of Lost Health.”
“Sanatogen is ... a rebuilding food.”
“... revitalizes the overworked nervous system.”
“Specific nerve tonic action.”
“Most reliable and scientific of all nutrients.”
“... in certain diseases it exerts a specific action which renders
it a valuable adjunct to other curative measures.”
“It stimulates metabolic activity of tissue cells and secures more
complete oxidation of energy-yielding elements.”
“Sanatogen nourishes the system in a persistent, gradual, cumulative
way, so that its best effects unfold themselves in a systematic, substantial
progression to health and strength. It follows that a regular
and prolonged administration of Sanatogen is necessary for the attainment
of lasting results.”
“Sanatogen is a scientific compound, every particle of which represents
the finest concentrated, tissue-constructing nutriment, endowed
with unique revitalizing and rejuvenating powers.”
“Sanatogen contains over 700 per cent. more tissue-building, life
sustaining nourishment than wheat flour.”
Truly a wonderful preparation—if these statements are
true! But they are false—most of them at least. And in
that many who can ill afford it may be led to pay a ruinously
high price for a very ordinary food, the statements are
viciously and cruelly false.
In view of the properties with which Sanatogen is credited,
its composition is naturally a matter of more than ordinary
interest. What is this life-giving product? A package of
Sanatogen was purchased and subjected to examination and
analysis in the Association’s laboratory. Our chemists
report:
LABORATORY REPORT
Sanatogen is a fine, nearly white powder having a faint
yellowish tinge. A circular which is enclosed in the
package states:
“Sanatogen is a definite organic combination of 95 per cent. of
pure, specially prepared casein and 5 per cent. of sodium glycerophosphate,...”
Qualitative tests indicated the presence in Sanatogen of
casein, sodium, a phosphorous compound and glycerin or a
glycerin compound. Starch and sugars were absent.
Quantitative analysis showed that the composition of
the specimen was essentially as follows:
Water (loss at 130 C.) | 8.60 |
Ash | 6.23 |
Casein and other proteins (N × 6.38) | 83.10 |
Casein (N in precipitated casein × 6.38) | 80.57 |
Proteins other than casein (by difference) | 2.53 |
| Sodium glycerophosphate (NaC3H7O6P) |
(P in filtrate from casein precipitation × 6.79) | 5.59 |
Insoluble matter | 0.84 |
Undetermined | 1.87 |
While these results show that the claims concerning the
composition of Sanatogen are not entirely correct, they
indicate that the essential element in Sanatogen is casein.
The slight variation between the composition claimed for
Sanatogen and the composition as determined by chemical
analysis is of minor importance. Whether there is 83 per
cent. of casein as found by the Association’s chemists or 95
per cent. as asserted by the manufacturers matters little. The
important fact is that casein makes up about nine-tenths of
the preparation and, as must be perfectly evident, Sanatogen
derives whatever food value it may have from that casein.
Casein is known in its commonest form as the curd in milk,
or as “cottage cheese.” After the cream has been separated,
the milk which remains contains nearly all the casein and
milk sugar originally present but practically none of the fat.
WHY NOT COTTAGE CHEESE?
Whence comes the stimulation of metabolic activity, the
wonderful nourishment of the system, the marvelous revitalizing
and rejuvenating power claimed for Sanatogen? Not
from the sodium glycerophosphate, for the consensus of
opinion among leading physiologists indicates that phosphorus
in the form of glycerophosphates has little influence
on metabolism. Not from the glycerin, surely, for even
granting that glycerin has food value the amount present is
so small as to be negligible. The real source of energy in
Sanatogen, then, lies in the casein which comprises about
nine-tenths of its ingredients.
| Kind of Food Material | Price per
Pound | Cost of
1,000 Calories
Energy | Calories.
Energy for
One Dollar |
| Sanatogen | $4 | .54 | $3.01 | 332 |
| Celery | | .05 | .77 | 1,300 |
| Eggs ($0.36 per doz.) | | .24 | .16 | 6,300 |
| Milk ($0.07 per qt.) | | .035 | .11 | 8,850 |
| Pork, loin roast | | .12 | .10 | 10,350 |
| Butter | | .30 | .09 | 11,250 |
| Mackerel, salt dressed | | .10 | .08 | 11,850 |
| Beef, stew meat | | .05 | .07 | 15,300 |
| Wheat bread | | .06 | .05 | 20,000 |
| Rice | | .08 | .05 | 20,250 |
| Sugar | | .06 | .03 | 29,200 |
| Pork, fat salt | | .12 | .03 | 29,500 |
| Potatoes | | .01 | .03 | 29,500 |
| Beans, white | | .05 | .03 | 30,400 |
| Oatmeal | | .04 | .02 | 45,000 |
| Cornmeal | | .025 | .02 | 65,400 |
| Wheat flour | | .025 | .02 | 65,400 |
Of course Sanatogen, being composed largely of casein, has
some food value. What that food value is may be seen by
the accompanying table which compares the yield of energy
for Sanatogen with that of a number of staple food products,
the figures for the latter having been adapted from Professor
Atwater’s calculations. This table shows that, from
the standpoint of economy in the purchase of energy, no
other food in the list is so poor as Sanatogen. While the
manufacturers claim that “Sanatogen contains over 700 per
cent. more tissue-building, life-sustaining nourishment than
wheat flour,” the table shows that one dollar’s worth of
wheat flour contains as much energy as one hundred and
ninety-seven dollars’ worth of Sanatogen!
AN INQUIRY
Like all “patent medicines,” Sanatogen is exploited by the
testimonial route. Actors, authors, politicians and not a few
physicians—the latter, to the credit of the American profession,
be it said, being chiefly Europeans—have testified to the
wonderful properties of this product. Believing that it would
be of interest to learn what scientific men thought of Sanatogen
a letter of inquiry was written to several men whose
training particularly fits them to express an impartial opinion
on a question of this kind. The following inquiry, expressed
in practically the same words, was propounded:
Is it possible for a product, even if it has the composition
claimed for Sanatogen, to have properties as a food and
medicine which are claimed for this preparation?
The replies to this inquiry are interesting and instructive,
although they are what might have been expected from men
whose judgment has not been warped by the glittering claims
of the Sanatogen publicity agents.

Some of the reasons for the sale of Sanatogen! A few specimen
advertisements of Sanatogen’s enormously expensive advertising campaign.
THE REPLIES
Dr. Lewellys F. Barker, professor of medicine, Johns Hopkins
University, medical department, says in part:
“If Sanatogen consists simply of casein and sodium glycerophosphate,
it is pretty obvious that all of its good effects
(except perhaps the psychic influence of taking an expensive
and, to the layman, mysterious remedy) can be gotten by
including milk and eggs in the food....
“The objection to Sanatogen lies, it seems to me, not in the
assertion of its proprietors that it is a ‘food and a tonic,’ but
in the misleading of the public and physicians into the belief
that it possesses extraordinary powers which make it worth
while to pay the price charged for it in order to get it. Very
extravagant claims are being made for it in advertisements
in the lay press. If just as much, and more, good in the
form of ‘food and tonic’ can be obtained from a dollar’s
worth of milk and eggs as from a dollar and ninety cents’
worth of Sanatogen, it is surely the duty of the medical profession
to inform the public of that fact.”
Dr. Frank Billings, professor of medicine and head of the
Department of Medicine, University of Chicago, expresses his
opinion thus:
“Of course, the thing is a fraud both as a food and as a
tonic. Even if it met all the requirements of the statements
made of it by the makers, it would not be any more of a food
than as much casein taken in milk and probably not as
good; or any more than some other albumin taken in some
other form. I do not know just what pharmacologists say of
the glycerophosphate of soda, but so far as my own clinical
observations go I never saw any result from its use that
could be called specific, that is, due to the drug.”
Dr. Richard C. Cabot, assistant professor of clinical medicine,
Harvard Medical School, says:
“In reply to your letter respecting the properties of Sanatogen,
I would say that in my opinion it is vastly improbable
that it has the properties claimed for it in the advertisements
which you enclosed to me. I have no doubt that it is a
fairly good food. I see no reason to believe that the phosphorus
that it contains has any special action.”
Otto Folin, professor of biological chemistry, Harvard
Medical School, expresses himself thus:
“For myself, or for any one who would take my advice, I
would prefer a glass of milk to Sanatogen when hungry and
plain glycerophosphate to Sanatogen when in need of a tonic.
“Medicated feed used to be sold for horses. To me the
‘food tonic’ combination represents one of the most unscrupulous
fake ideas used by manufacturers of patented articles
to fool the public.”
Ludvig Hektoen, professor of pathology, University of
Chicago, says in part:
“In my opinion, no attention whatsoever should be paid to
the claims advanced in favor of ‘Sanatogen’ as food and as
medicine, because the statements made in the advertisements
of this product are extravagant, misleading and quackish.”
J. H. Long, professor of chemistry and director of chemical
laboratories, Northwestern University Medical School,
expresses the following opinion:
“With every reading of the advertising literature of the
Sanatogen Company I am more and more impressed by the
gross exaggeration of the claims made for this mixture of
casein and sodium glycerophosphate. Cow’s milk contains
31⁄2 to 4 per cent. of casein, associated with soluble phosphates.
It is absurd to think that this casein after precipitation
from the milk has a greater nutritive value than it has
in its native condition. Casein, at best, is probably less
valuable as a food than are certain other proteins, because of
its lack of some of the amino groups essential in tissue
building, and the addition of a glycerophosphate cannot supply
this deficiency.
“This is not the first attempt to exploit casein preparations.
The earlier efforts failed in practice because they were based
on a wrong conception concerning the physiologic value and
importance of this protein. The assumption that in the case
of Sanatogen a ‘definite organic combination’ with the glycerophosphate
is formed cannot be taken seriously by chemists.
We have witnessed many such efforts to palm off mixtures as
definite organic compounds, and in this way to claim for
them a value in excess of that which they actually possess.”
What one dollar will buy in food energy! A comparison of
the caloric values of sanatogen, cow’s milk, sugar and wheat
flour. Based on the table accompanying this article.
Graham Lusk, professor of physiology, Cornell University
Medical College, after calling attention to the falsity of the
claim that Sanatogen is “a life-sustaining agent in disease,”
says:
“If one considers the casein content alone, the dose of
Sanatogen recommended in the circular would furnish, at
best, about what is contained in a pint of milk, or one-fourth
of the total of the protein necessities of the body—using
a low protein requirement. That sodium glycerophosphate
has any distinctly beneficial physiologic action has
never, to my knowledge, been shown.
“It is a great pity that the public does not realize the
splendid and economical value of milk, bread and the ordinary
vegetables, cereals and meats, as true ‘tonic food stuffs,’ in
contradistinction to prepared nostrums whose sale depends on
a psychic stimulus applied to a susceptible populace.”
H. Gideon Wells, associate professor of pathology, University
of Chicago, says:
“There is nothing in my knowledge of physiologic chemistry
which would lead me to believe that a mixture of chemically
isolated casein and sodium glycerophosphate would possess
any effect more favorable than that of a corresponding
amount of milk. I can easily believe that it would be less
valuable than milk. The successful practice of many commercial
houses, of isolating one of the constituents of our
food, and ascribing to it marvelous nutritive or therapeutic
properties, is one of the most telling bits of evidence of the
inadequacy of the education of the medical profession in
physiology and physiologic chemistry that can be conceived.”
The consensus of opinion thus expressed is only what
might have been expected from men who could discuss the
problem in a purely judicial spirit and with a freedom from
that bias which seems to be inseparable from the consideration
of the simplest of mixtures that have been glorified by
a proprietary name.
THE TYRANNY OF WORDS
Herr Teufelsdröckh was right when he panegyrized clothes.
And the worship of clothes is carried to the extreme nowhere
so much as in the case of word-clothes. The most plebeian
things when bedecked in sufficiently imposing word-finery are
endowed with the attributes of royalty before which the average
intellect bows down. Neither cottage-cheese nor glycerophosphates,
when exposed naked to the world, commands any
overweening respect; combined and dressed in the magic
word “Sanatogen,” they receive the homage of those whose
judgment is blinded by the glittering trappings of word-finery.
Some day, possibly, there will be a democracy of intellect
which will refuse to prostrate itself before mere word-raiment
and will insist on appraising things at their naked
worth. When that day comes, proprietary humbugs like
Sanatogen will have become as extinct as the dodo and the
great auk.—(From The Journal A. M. A., April 20, 1912.)
The Bauer Chemical Company’s “Reply”
To the Editor:—Our attention has been called to a most
unfair and unwarranted attack on Sanatogen which appears
in your esteemed publication [April 20, p. 1216]. The article
is such a perversion of the actual facts, and so completely—if
not intentionally—misleading that we request, as a matter of
common justice, that you give this reply equal publicity to
your attack. The admiration and respect we have felt for
your journal and our appreciation of the place it holds in the
field of medical journalism, made your attack on a product
like Sanatogen, representing so definitely the most painstaking
and scientific research, the last thing expected. Indeed,
it seems inconceivable that a journal apparently so alive to
its responsibilities could publish broadcast an article so calculated
to do harm, without first giving those whose interests
are most at stake an opportunity to substantiate their claims.
There never has been a time that we have not been ready
to meet any request from The Journal, or the respected
gentlemen composing the Council on Pharmacy and Chemistry,
for all information and data concerning Sanatogen.
Had we had the slightest inkling that our product—or the
claims made for it—were open to question or criticism, we
would gladly have submitted all of the evidence, clinical,
experimental, and theoretical, on which every statement,
however simple, has been based.
To make a response is difficult, because your article is not
written in a fair, unprejudiced spirit. In fact, although one
would expect a sober, serious consideration of a matter so
fraught with importance (if your contention is right) your
whole attitude is one of ridicule and jocularity. Is it right
to present scientific material in such a way and show so little
respect for those who have offered you no affront or done
you no injury? A little investigation would have shown you
that the statements we have made about Sanatogen are based
on the experiences and opinions of such men as von Noorden,
C. A. Ewald, Duhrssen, Eulenburg, Neisser, Binswanger, von
Leyden, Krafft-Ebing, Tillmanns, Tunnicliffe, and thousands
of other earnest, reputable physicians. Any one might differ
with their conclusions, but is it courteous or decent to hold
them up to ridicule and contumely?
Can a discussion thus conducted hope to solve a scientific
problem or accomplish any real good?
It would hardly seem so, and with all due respect we cannot
help but feel that the situation has its analogy in the legal
doctrine, “when you have no evidence, ridicule and abuse your
opponent and his client.”
Sanatogen is a definite organic combination of (in round
numbers) 95 per cent. casein and 5 per cent. glycerophosphate
of sodium. The analysis as published in The Journal fails
to show that this statement is untrue. The slight deviation
as to the amount of casein present is explained by the fact
that The Journal’s figures include the moisture, while ours
are on the dry substance. Inasmuch as nearly all the moisture
is absorbed after the product leaves the laboratories and
is therefore added weight, the figures should be on the dry
substance. It is hinted in the article that Sanatogen is a
mere mixture of ingredients, in fact one of the gentlemen
you quote openly intimates so. To this we say most emphatically
that anyone asserting Sanatogen to be a mere mechanical
mixture of ingredients and not a definite chemical compound either
wilfully misstates the facts or does not know.
Sanatogen represents a new idea or discovery in the domain
of invalid dietetics and as such its process of manufacture
as well as the product are protected by U. S. Letters Patent.
Assuredly it is the definite chemical combination found in
Sanatogen on which the special value of this product as a
medicinal food and tonic depends. A mere mixture of ingredients
would represent only the sum-total of their individual
virtues, but a definite combination of such ingredients means
the formation of a new compound with properties of its own
which far transcend those of any simple mixture of the original
ingredients.
To compare Sanatogen to cottage cheese is the height of
absurdity—as it was probably intended to be. The casein
of Sanatogen is perhaps the most carefully purified milk
protein available, and this fact is of essential importance
when considering the value of Sanatogen as a medicinal food.
To compare the casein of Sanatogen with crude commercial
casein or with cottage cheese is as ridiculous as to compare a
crude drug with the refined element. The same applies to the
matter of cost. We suggest that an attempt be made to prepare
purified casein according to Hammarsten’s method, if
one wishes to determine what labor and expense is involved
in the operation. Possibly it will be found cheaper to buy
Hammarsten’s casein in the open market where the price is
$3.50 per pound wholesale! And it is not a proprietary
product, either.
Further, to compare the economic value of Sanatogen on
the basis of calories is as unscientific as it is deliberately
misleading. If the caloric standard only counted, a pound of
oleomargarine would be as valuable as fifty eggs, a pound of
laundry soap as valuable as a pound of choice beef. Sanatogen
is not intended or recommended to replace ordinary
foodstuffs. It is not recommended as a caloric or heat producer,
but as a food-tonic supplying the essential elements of
tissue construction and cell-repair in easily and perfectly
assimilable form.
Digestibility, ease and completeness of assimilation count
a great deal, and are the sole determining factors in cases
of illness. Again, starch and fat are not essential substances
to life. Without protein we cannot live. Exclude everything
else from a patient’s dietary, and he will live. Exclude protein
and it is only a question of time before he dies. It
is evident, therefore, that to measure the value of a given
food in calories only is misleading and dangerous, and an
editorial in your valuable publication of November 4 last
distinctly points this out.

Specimens of Sanatogen advertising—(1913).
According to the most careful and extensive experiments,
covering a large number of scientifically studied cases, Sanatogen
is not approached in the matter of rapidity of digestion and absorption by any other known foodstuff. That such
a product does exert a definite stimulating or activating effect
on the digestive and assimilative functions, thus promoting
the digestion and appropriation of nutritive material has been
demonstrated over and over again. That the organic phosphorus
of Sanatogen is almost completely retained and assimilated
has been proved beyond doubt by carefully conducted
metabolism experiments. That from this, and from the
stimulating action on phosphorus and nitrogen metabolism,
a favorable effect on the nervous system could result, is
conceivable. That such an effect does actually take place
has been demonstrated clinically in literally thousands of
cases.
As to our advertisements and literature: Every claim made
emanates from the freely recorded statements of competent
observers, checked and rechecked by men who have been
absolutely free from all bias or prejudice. And these opinions,
moreover, are not the superficial, passing views of a few
physicians. Instead our claims are based on the voluntary,
unbiased written reports of clinical experiences by over 15,000
practicing physicians—among whom a goodly proportion are
members of your esteemed Association—and on over 150
published articles in the leading medical journals of the
world, some of which your journal has considered of sufficient
importance to present to its readers in abstract form,
suppressing, it is true, all mention of Sanatogen, although
thereby the original was sadly emasculated, if not actually
falsified.
Among the physicians who have carefully tested Sanatogen
and determined its dietetic and therapeutic properties are
many men of truly international reputation, men who are
as far above suspicion as was Caesar’s wife. At least one of
these men was the honorary guest of your Association a few
years ago.
It is such men that your article holds up to contempt and
dishonor when you allow the false inference to go forth that
Sanatogen is a mixture of casein and glycerophosphates. It
is such men’s careful researches and experience that you
attempt to offset by the snap judgment of men whom we
claim, without the slightest intent of disparaging them, to be
in the present instance unfitted to give an opinion on Sanatogen
inasmuch as they—with perhaps one honorable exception—have
never tested or used the product. Their lack of
definite knowledge of Sanatogen is shown by their persistent
references to casein and the glycerophosphates, as though
these two ingredients were separate and not chemically combined.
To consider Sanatogen a mixture is to lose the vital
detail of its specific value.
Now after all, is this a fair, judicial spirit, is this true
scientific enquiry? Are we to accept offhand judgments in
preference to the opinions of those who speak from years of
observation of the effects of Sanatogen? In the name of
justice and fair play, is it right for the great Journal of the
A. M. A. to ignore and suppress the accumulated evidence in
favor of Sanatogen and cite instead the cursory opinions of
men who have never seen Sanatogen, tested or observed its
effect, who by the very nature of your enquiry must have
been influenced subconsciously in favor of your side of the
matter.

The above is a reduced reproduction of a full-page advertisement
in a magazine devoted to pseudo-scientific fads. The advertising pages
of this magazine reek with frauds.
During the twelve years Sanatogen has been used, prescribed
and recommended by thousands of competent physicians,
it has been free from all secrecy. The truth has been
told at all times. From the first we have cooperated with the
profession. Never have we failed to safeguard the doctor’s
interests. Never have we suggested by word or inference
that any person should employ Sanatogen to the exclusion of
medical treatment. Not a day passes but we refer people who
inquire about this or that bodily ill, to their physicians for
advice.
We regret the length of this letter but feel that the scientific
character of Sanatogen, its well-defined chemistry and
the respect we owe to the men who have not hesitated to give
their honest opinions concerning its food and tonic effects,
make it imperative that we refute at once errors and misleading
statements, and correct to the best of our ability the
wrong impression you have allowed to go forth. The clinical
reports and statements and the scientific evidence on which
we have based our claims are constantly available and may
be examined by any responsible person for verification or any
other legitimate purpose.
We have tried to make this article temperate, fair and free
from ill temper and ill feeling. We only ask for justice and
feel that you will be willing—possibly anxious—to correct,
so far as you can, the great wrong you have done us.
The Bauer Chemical Co.
By F. W. Hehmeyer, Resident Manager.
[Comment: We devote considerable space to the above
free advertisement of Sanatogen, as The Journal does not
want to be accused of being unfair, even to patent medicine
venders. As our readers will recognize, the above is simply
a reiteration of the statements that have been published in
the advertisements of Sanatogen. The song that runs through
all the advertising matter is that Sanatogen is a chemical
compound, and since it is a chemical compound it therefore
possesses properties not to be found in the ordinary mixture.
It is the old, old story; the “synthetic” argument is as hoary
as the nostrum business itself but fortunately the medical
profession is no longer easily fooled by it.
As a matter of fact, even assuming for the sake of argument
that the casein and glycerophosphate in Sanatogen are
in chemical combination, it would be a union of the loosest
kind, which on entering the digestive tract must be broken up
into its more stable components, casein and glycerophosphate.
To claim that Sanatogen possesses any properties not possessed
by its essential constituents is a silly piece of pseudo-scientific
claptrap.
Of the testimonials on Sanatogen we shall at this time have
nothing to say; The Journal has in the past repeatedly
shown the worthlessness of this kind of evidence.
We have nothing to retract, rather we would emphasize
and, had we space, enlarge on what we have already published,
for we believe that a large and unfortunate portion of
the public, that can ill afford it, is paying a ruinously high
price for a substance having a very mediocre food value.
That indigent consumptives, for instance, should be led by
glittering falsehoods to squander on Sanatogen money that
should go for “food tonics” of infinitely greater value, such
as eggs, milk, vegetables and meats, is not only economic
waste but inhuman cruelty.—Ed.]—(From The Journal
A. M. A., May 18, 1912.)
The Sanatogen “Grand Prix”
A number of letters have been received recently expressing
surprise that Sanatogen had been granted a “grand prix” at
the Exhibition of Medical and Surgical Material held in London
at the same time that the Seventeenth International
Congress of Medicine was in session. The correspondents
have asked what such an “honor” meant. The company
which exploits Sanatogen in the United States has not been
slow to apprise the American public of the award. It has
gone further and has written the advertising managers of
magazines—including those that had refused Sanatogen
advertisements—directing their attention to the fact that
Sanatogen was awarded a “grand prize” and opining that
“this unusual distinction” should make plain “the desirability
of the presence of Sanatogen in the advertising columns of
your esteemed publication.”
Those familiar with the methods of awarding prizes,
medals and certificates to commercial firms and their products
at expositions and exhibitions attach little weight to the
“honors” thus conferred. It is a fact that most purchasers of
large—and expensive—exhibit space at such exhibitions
receive some kind of award which, it is tacitly understood,
will be a useful advertising asset. Every one can call to mind
many food products of mediocre quality that have flaunted on
their labels the gold medals received at various expositions.
Nevertheless, it seemed worth while to find out just what
the connection was between the commercial exhibition at
which Sanatogen received the grand prize and the Seventeenth
International Congress of Medicine. The following facts were
developed: The commercial exhibition was entirely distinct
and separate from the scientific exhibit of the Congress. It
was managed and conducted by a British drug journal which
had been giving annual “exhibitions” of its own for some
years past, and this took the place of its regular exhibition.
Immediately after the awards were made public the advertising
pages of this drug journal were filled with full-page
advertisements of the various products that received prizes.
It may interest our readers to know that while the cottage-cheese-glycerophosphate
product Sanatogen received a “grand
prize” two other proprietary cottage-cheese-glycerophosphate
products received “gold medals” at the same time. In the
pharmaceutical department of the exhibit a widely—and fraudulently—advertised
“patent medicine” received a silver
medal! From the facts given it should not be difficult to
appraise at its right value the “honor” conferred on Sanatogen.
The fact that the exploiters of this preparation are
trying to make capital out of this “award” is significant.

Nor is even the Asiatic neglected in the Sanatogen advertising
campaign. Here is reproduced an advertisement appearing in an
Asian sporting newspaper published in India. The original advertisement
was 10 inches by 15 inches.
Among the members of the Award Jury whose names were
given by this drug journal were three men of prominence in
Great Britain, to whom we have written. A reply has been
received from one, Dr. Stephen Paget, who says: “I was not
on the jury, nor do I know anything about the matter.... I
had nothing whatever to do with the awarding of
prizes.”—(From The Journal A. M. A., Oct. 11, 1913.)
A Restatement of the Case
The case against Sanatogen has been pretty plainly given
at different times in The Journal, but the sale of the stuff
goes on—thanks to the power of advertising. One criticism
that has been made of this patent medicine is the exorbitant
price charged for it. This objection, although but an incidental
one, is the one that apparently appeals to the layman more
strongly than the much more serious criticism, fraud in
exploitation. You arrest the attention of the average man
when you appeal to his purse; he resents paying an exorbitant
price for anything. This probably accounts for the fact that
this particular criticism has apparently hurt the sale of Sanatogen
to a greater degree than the more serious objections
made to the preparation. This also accounts, doubtless, for
the fact that the attempts to answer The Journal criticisms,
by those who are selling Sanatogen, have been largely
devoted to the one point—its outrageously high price.
The fundamental objection to Sanatogen is not its high
price, but the attempt to ascribe to a mixture of casein and
glycerophosphates powers not possessed by these ingredients—in
other words, the misleading and fraudulent claims made
for it. Even if it were sold at cost price, the stuff, as at
present advertised, would still be a fraud. The nub of the
whole matter is: The claims made for Sanatogen are unwarranted,
misleading and fraudulent.
SOME FRAUDULENT CLAIMS
The constituents of Sanatogen are casein and sodium glycerophosphate.
These two very ordinary substances possess,
so the Sanatogen people would have us believe, peculiar properties
when they are brought together in chemical combination.
Sanatogen, they claim, is a chemical combination of
these constituents. The claim may be a good “selling-point,”
but it cannot be, and is not, seriously taken by chemists.
But even supposing, for the sake of argument, that sodium
glycerophosphate and casein could be combined, there is not a
scintilla of evidence to show that such a combination could
survive the destructive influence of digestion and be absorbed.
Whether Sanatogen is a chemical combination of casein and
sodium glycerophosphate or a mere mechanical mixture of
these two substances is really immaterial. In either case, it
would be separated into its constituent parts by the digestive
juices and would have the properties of sodium glycerophosphate
and casein, and nothing more.
Remembering this, let us examine once more some of the
claims made for this patent medicine:
“Sanatogen is a nerve and tissue food for which the brain, spinal
cord and the nerves have a special predilection.”
“... practically identical with the main ingredient of nerve and
muscle cells....”
“Sanatogen stands pre-eminent in its power to feed the nerve centers,
to promote healthy digestion, to give strength and endurance to
the entire system.”
“... food for tired nerves....”
“... a rational, scientific nerve-food.”
To the physiologist, the term “nerve-food” is an absurdity.
The processes of digestion reduce the albuminous substances
(proteins, such as casein) of the food to simpler forms. This
is true no matter what may be their source. Whether the
proteins are derived from the gluten of wheat, the casein of
milk or the albumin of egg, one will “feed the nerves” just
as well as the other. And Sanatogen “feeds the nerves” no
more than, in fact not as much as, do bread and meat and
eggs. Of course, the casein in Sanatogen has food-value, but
so has ordinary casein—cottage cheese, “pot cheese,” or the
German Schmierkäse, for instance—and it is both false and
fraudulent to claim for the casein in Sanatogen any greater
nutritive value than that possessed by the casein in ordinary
milk. To pretend that there are wonderful properties in the
protein of Sanatogen when just as good protein can be purchased
(for much less money) from the milkman, is to perpetrate
a fraud on the purchaser. Here are some more
claims:
“... marvelous revitalizer of nerve health.”
“... Sanatogen has positive reconstructive force in neurasthenia.”
“If You need New Strength and Vitality You should at once get
acquainted with Sanatogen.”
Strangely like the “lost manhood” advertisements, this last.
And this, also:
“... has brought new strength, new vitality and new relish
of life to thousands upon thousands who suffered from starved
nerves....”
“Countless people ... have regained fresh health and vigor
through the vitalizing and invigorating effects of Sanatogen.”
Of course Sanatogen is not sold as a “consumption cure.”
No such crude claims as these emanate from the skilled
advertising agents employed by the Sanatogen people. If
they did they could not get space in high-grade magazines!
As a preventive of consumption, however, we find:
“Sanatogen ... creates new tissue and nerve capital ... This
nerve capital will ... save the individual from attacks of
acute disease. Against tuberculosis it is an excellent investment.”
Also, it is a pick-me-up! Thus:
“Sanatogen promises to pick you up when run down—it does so.”
Most people are under the necessity of working for a living.
If we are to believe the Sanatogen advertisements, it seems
remarkable that the human race has managed to jog along for
so many centuries without this product, for we read:
“It is practically indispensable to all who are unable to take prolonged
rest....”
Naturally we do not expect to find the coarse, “free-to-you-my-sister”
type of claims in Sanatogen advertisements.
Nevertheless:
“Women ... find in Sanatogen a genuine sustaining agent.”

Sanatogen is advertised heavily in the British Isles. Here are some
greatly reduced advertisements appearing in British magazines. The
three largest advertisements shown here measured in the original
10 inches by 14 inches, each.
Finally, we would respectfully direct the attention of those
gentlemen of the medical profession who have so far forgotten
the dignity of their calling as to give fulsome puffs for
this casein-glycerophosphate product to the following claims
and ask whether they really subscribe to them:
“... it revivifies the nerves, promoting sleep and helping
digestion....”
“... it builds up the blood, creating new strength and the
power to do and accomplish.”
“... Sanatogen is a natural, healthful food and tonic....”
“... a health and strength giving food and tonic composed of
those very elements which make cell and tissue grow.”
“Blood and tissues alike hunger for Sanatogen as their concentrated
nourishment.”
“Sanatogen is the one food tonic that commands your absolute
confidence.”
How many intelligent physicians really believe that there
is the slightest basis of fact for the claims we have quoted?
Yet it is by means of these claims that Sanatogen is being
foisted on a public that looks to the medical profession for
enlightenment and truth. And every quotation in this article
is taken from advertising matter issued during the current
year, 1913!
In closing, let us reiterate: The objections to Sanatogen are
primarily the objection to any fraud. It is being sold under
unscientific, misleading and fraudulent claims; moreover,
although this is of less importance, the purchaser pays an
extraordinary price for a most ordinary product. We believe
the time will come when even the artificial stimulus of vast
advertising appropriations will be insufficient to overcome the
inertia inherent in a product of small merit. When that time
comes, Sanatogen will die a natural death. In the meantime,
its exploiters are reaping a golden harvest, of which no small
part is being divided among publishers, medical and otherwise.
And the credulous among the sick and suffering pay the bills!—(From
The Journal A. M. A., Dec. 6, 1913.)
SANATOGEN: A SCIENTIFIC INVESTIGATION OF
ITS ALLEGED ACTION ON THE RECUPERATING
POWERS OF THE BLOOD
Sanatogen, the new elixir of life compounded of casein and
glycerophosphates, has been noticed in The Journal from
time to time. It will be remembered that, while it has been
claimed that “Sanatogen contains over 700 per cent. more
tissue-building, life sustaining nourishment than wheat flour,”
the facts are that one dollar’s worth of wheat flour contains
as much energy as one hundred and ninety-seven dollars’
worth of Sanatogen! To this the manufacturers rejoin, in
effect, that the casein and glycerophosphates in Sanatogen,
being in chemical combination, possess a mystical and esoteric
virtue not measurable in terms of the food-value of the several
ingredients. The fact is, of course, that even assuming,
for the sake of argument, that the ingredients of Sanatogen
are in chemical combination, the compound cannot have any
effect on the organism different from that of the uncombined
casein and glycerophosphate, for the union must be of the
loosest kind and must be broken up as soon as the preparation
enters the digestive tract.
Testimonials are published in the Sanatogen “literature”
which show results in a variety of conditions; cerebral concussion,
alcoholic gastritis, anemia, etc. The patient is given
a chance to recover with rest, proper diet—and Sanatogen.
And the recovery which ensues is attributed to Sanatogen!
Not all of these testimonials are as naive as that of the
Right Reverend the Bishop of Bath and Wells, who contributes
the following bit of second-hand testimony:
“You may like to hear that I am informed by my private secretary
that a member of his family has derived very remarkable benefit
from using Sanatogen.”

Above is a photographic reproduction of one of the exhibits made
at Atlantic City at the last annual meeting of the American Medical
Association.
Jar 1 contains 31⁄3 ounces of the casein-glycerophosphate combination
made abroad and sold under the name “Sanatogen;” it costs
One Dollar.
Jar 2 contains 2 pounds 131⁄2 ounces of a casein-glycerophosphate
combination made by an American casein company; it costs One
Dollar. For the same money, then, one gets nearly 15 times as
much of this casein-glycerophosphate combination made in America
as when the casein-glycerophosphate combination is sold under the
name “Sanatogen” and manufactured abroad.
Jar 3 contains one dollar’s worth of pure casein. The casein
entirely fills the jar, although the weight of the material is the same
as the weight of the casein-glycerophosphate combination in Jar 2.
Pure casein is more bulky than the casein-glycerophosphate combination.
Again we would emphasize that the chief objection to Sanatogen is
not its high price, but the fraudulent claims under which it is sold.
The following selections perhaps fairly represent the value
to science of the clinical evidence offered. Describing a case
of vomiting from cerebral concussion:
“I ordered an ice-bag to the head, a mustard leaf to the epigastrium,
absolute recumbency in bed and small feeds of Sanatogen
with water. This diet was continued for three days, but the vomiting
ceased the second day.”
And here a case of “hungry tired nerves”:
“I have just had a recovery in a remarkable case which scores
a victory for Sanatogen. The patient, a man 63, had been treated
for some years past for heart trouble. When he came to me, however,
I diagnosed his trouble as ‘hungry tired nerves.’ I put him on
Sanatogen and eupeptics. In a month he was much improved.”
In a serious case of the “American disease”:
“I tried Sanatogen on a woman suffering from extreme neurasthenia
and debility. For the past six weeks I have had, and still
have, her under rest-cure treatment, during which time I have
given her Sanatogen. I have been very much elated with the
treatment.”
Anemia in a girl of 23 working in a bookbindery:
“I promptly decided to use Sanatogen. In addition, I was able
to secure the girl’s absence from work so that she had the advantage
of outdoor life and sunshine. Improvement was rapid.... Both
a priori and from results obtained it seems almost justifiable to speak
of Sanatogen as a specific in ordinary uncomplicated anemia.”
Note that in all these cases two or more remedial factors
were introduced, yet any favorable result is promptly ascribed
to only one factor, Sanatogen! And the factor of spontaneous
improvement irrespective of all remedial measures is also
ignored.
A “SCIENTIFIC” TESTIMONIAL
Every physician knows that the kind of evidence just quoted
has the same scientific value as that of the average “patent
medicine” testimonial—none whatever. The exploiters of
Sanatogen put forward some testimony, however, that purports
to have a certain authority. This is a statement to the
effect that “Sanatogen acts as a strong stimulus as far as
the recuperative powers of the blood are concerned.”
This claim is based on biologic experiments carried out by
two physicians, Drs. G. Mann and J. G. Gage, the record of
whose work was published in the Lancet, Oct. 19, 1912. The
article of Mann and Gage was gone over with some care and
the experiments there described did not seem to justify the
conclusions reached by the authors. As it had been published
in the Lancet, a medical journal of standing, whose
publishers apparently thought it of sufficient importance to
warrant the expense of a colored plate insert, it seemed worth
while to have the work of Mann and Gage reviewed and its
conclusions checked by parallel experiments. A. J. Carlson,
professor of physiology at the University of Chicago, was
asked to do this work and kindly undertook it. His report
follows:
Report of Professor Carlson
I am asked to review the work done by Drs. G. Mann and
J. G. Gage, from which they draw the following conclusions:
“Sanatogen [sodium caseinogenate glycerophosphates] further stimulates
blood cells to undergo nuclear division, which during the early
period is mostly amitotic.... Therefore, it is evident that Sanatogen
acts as a strong stimulus as far as the recuperative powers
of the blood are concerned.”
Three series of experiments were made and reported by
Drs. Mann and Gage, comparing the histology of the blood
in starvation and during the height of digestion. The experiments
of the first series were made on six students, the
starvation period being thirty-six hours, ordinary food being
taken after the thirty-six hours’ fast, so far as can be gathered
from Dr. Mann’s report. It was found that during the height
of digestion the nuclei of the lymphocytes and leukocytes stain
more deeply, and that there is a slight decrease in the cytoplasm
and consequent diminution in the size and number of
the granules of the neutrophil and the eosinophil cells, in comparison
with the blood during fasting.
The experiments of the second series were made on 100
frogs that had starved for months, blood-films being taken
at varying periods after feeding 1 gm. Sanatogen. The
changes noted in the white blood-cells of the frogs were practically
identical with those described in the case of the six
students. Increased cell division is stated to occur. The
nuclei of the erythrocytes stain more deeply and the cells are
increased in size, and exhibit increased cell division twenty-four
hours after the feeding.
The third series consisted of one experiment on Dr. Mann
himself. He fasted twenty hours and then took 15 gm. of
Sanatogen in a cup of water. Practically the only change
noted in the blood after the feeding was a deeper staining
of the nuclei of the white cells. To quote:
“The changes in the granules of the polymorphonuclear leukocytes
and in the eosinophils were much less marked than in the specimens
supplied by the students, and the diminution in the size of the white
cells was so insignificant as to be hardly noticeable.”
Assuming that the blood-changes reported are correct, is
the conclusion warranted that Sanatogen is a powerful recuperative
stimulant to the blood?
In the first place, it remains to be proved, for there is as
yet positively no evidence, that the deeper staining of the
nuclei during digestion and absorption of a protein meal represents
“recuperative power of the blood” or processes of
“feeding” on the part of the white cells. The actual significance
of these changes requires further investigation. The
increased division of the blood-cells described by Dr. Mann
in the frogs was not observed by him in man. This phenomenon
in the frogs is, in all probability, associated with the
extraordinary length of the starvation period (months) and
the well-known seasonal variations in the physiology of this
species.
In the second place, waiving for the moment the question
of the significance of the blood-changes observed, the evidence,
so far as it goes, points to the conclusion that the
greater affinity of the nuclei of the white cells for the stain
is brought about by the feeding of any protein food. The
experiments do not demonstrate any different or more marked
effect from Sanatogen than from other protein foods.
The test on the six students with ordinary food can be considered
as a control on the single Sanatogen test on Dr. Mann
himself. The blood-changes in the students were more
marked than in Dr. Mann.
Although Mann and Gage say that six frogs were used as
controls, they do not say how the controls were treated, or
draw any comparisons between the controls and the frogs fed
with Sanatogen. So far as the report goes, therefore, the only
basis for comparison is afforded by the work of one of Dr.
Mann’s pupils, H. G. Butterfield. This worker, as quoted by
Mann, found that on feeding newts after two weeks’ starvation
with a worm the size of a wooden match, the nuclei of
all tissues (excepting nerve cells) take a much deeper stain
than in the control animals. To pronounce Sanatogen, on the
basis of the facts reported, a “powerful stimulus” to blood-formation
is a piece of special pleading, if not of downright
disingenuousness. Considered on its own merits, the work
would not have appeared to me worthy of being repeated
for the purpose of checking up such obviously unwarranted
conclusions. In order to comply with the request made of
me, however, I have repeated Dr. Mann’s experiments:
EXPERIMENTS BY PROFESSOR CARLSON
I. TESTS ON MAN (A. J. C.)
1. Thirty-six hours’ fast followed by a meal of 25 gm. Sanatogen in
water.
2. Thirty-six hours’ fast followed by a meal of 25 gm. casein in
water.
3. Thirty-six hours’ fast followed by a meal of 200 c.c. of milk.
II. TESTS ON RATS
1. Four animals, seventy-two hours’ fast followed by a meal of
Sanatogen in water.
2. Two animals, seventy-two hours’ fast followed by a meal of crackers
and milk.
3. Two animals, seventy-two hours’ fast followed by a meal of casein
in water.
4. Two animals, seventy-two hours’ fast followed by a meal of casein
and sodium glycerophosphate in water.
Blood-films were taken at intervals during the fasting
period and for twenty-four hours after feeding was resumed.
The technic of fixing and staining given by Drs. Mann and
Gage was followed in every detail in order to make my
results comparable with their findings.
RESULTS
My results may be summarized as follows:
1. The only change in the blood-cells that could be detected
with certainty in rats or in man, after feeding with Sanatogen
or after feeding with other foods, was a deeper staining of
the nuclei of the white cells in the films taken after feeding
as compared with the films taken during fasting. This difference
is generally distinct, and unmistakable, although individual
cells can always be found in the fasting and the
feeding preparations that show no difference in affinity for
the dyes. To this extent my results confirm those of Mann
and Gage. I was not able, however, to make out any constant
difference in the size of the cells, quantity of cytoplasm,
or size and number of cytoplasmic granules similar to those
reported by Mann and Gage.
2. There was no difference in the affinity for stains on the
part of the white blood-cells in films taken after feeding
Sanatogen and those taken after feeding milk, crackers,
casein, and casein and sodium glycerophosphate. This is
true for the tests both on man and on rats.
3. The above-mentioned difference in the staining of the
cell nuclei was somewhat more marked in the tests on rats
than in the tests on man, probably owing to the longer starvation
period in the case of rats.
It has already been stated that the significance of this
increased affinity for dyes in the nuclei of the white blood-cells
must be determined by further investigation. It may
be related to the change in the titration alkalinity of the
blood rather than an evidence of “recuperative power” on
the part of the blood, as it is well known that starvation
induces acidosis, while during digestion the alkalinity of the
blood is distinctly increased. If we assume that increased
staining reaction during digestion indicates “increased recuperative
power of the blood” it follows that such common
and inexpensive foods as milk, crackers and casein are just
as “powerful stimuli” to this recuperation as Sanatogen.
The extensive researches of Mendel and Osborne have
shown that casein is in a certain sense a perfect food in that
it is, in normal animals, capable of promoting growth and
maintaining nitrogenous equilibrium, at least for long periods
of time. The burden of proof, however, rests with the promoters
of Sanatogen to show that the casein in Sanatogen
is superior to the natural product of the cow.
Conclusion
From the findings in Professor Carlson’s report on this
disguised puff of a mendaciously exploited proprietary, about
all that remains to be said is that it is humiliating to find
such pseudo-science, not only built up by members of a profession
trained in science, but also given currency and
authority by a medical journal of high standing.—(From the
Journal A. M. A., Sept. 26, 1914.)
As to Sanatogen
For several years it has been known by physiologists that
all proteins taken into the stomach are disintegrated into
their fundamental components, the amino-acids, and that they
are not absorbed at all as proteins, except in most minute
amounts under exceptional conditions. Therefore, no matter
what protein is taken as food, the material obtained from
this protein by the body is a group of amino-acids, always
pretty much the same except in relative proportions, whatever
the food. A few proteins lack certain essential amino-acids,
but any ordinary diet supplies enough, and more than
enough, of each and every amino-acid. Furthermore, since
the proteins are completely disintegrated before absorption, it
follows that any adventitious chemical substance that is
bound to a protein taken in the food does not enter the blood
and circulate in the body in the same protein compound.
All the facts stated above are elementary, and should be
known by any and every physician who pretends to keep even
approximately abreast of the science of medicine. If there
are any who do not know these fundamentals, and from certain
unwelcome evidence we fear there are, they must be
resigned to being told just where they stand. Certainly they
have no good excuse for their lack of information, for the
physiologists and biochemists have informed them of modern
advances in innumerable ways. For ten years and more these
facts have been discussed and demonstrated in societies and
in medical publications. And yet—there is Sanatogen, prescribed
by Geheimrats, Hofrats and also by doctors, and testimonialed
as abundantly by men with medical degrees as
Duffy’s whisky is by centenarians. One can merely throw up
his hands. If, as the exploiters of Sanatogen declare, the
product “has been endorsed by von Noorden, Ewald, Duhrssen,
Eulenburg, Neiser, Binswanger, Leyden, Krafft-Ebing,
Tillmanns, Tunnicliffe and thousands of other earnest, reputable
physicians,” we can only say that their earnestness has
not been in the direction of grasping fundamental advances
in medical sciences, however great the reputation they have
gained may be.
The article by John Phillips Street that follows is another
report of exact experimental observation which shows, as was
obvious beforehand, that Sanatogen has the properties of its
constituents, namely, casein and glycerophosphates. Nothing
more nor nothing less could be the case. Bottling dried
cottage cheese plus some glycerophosphates, and raising the
price many times, may increase its psychic effect, but it will
not alter its physiologic action. These facts we have presented
often enough, but the amount of paid advertising the proprietors
of this compound find it profitable to carry in the
United States makes us feel obliged to give them this bit free.
That laymen may be persuaded to purchase Sanatogen in the
belief that it possesses some occult powers not to be found in
its constituents is not surprising. By blatant and persistent
advertising, the public can be fooled into buying any product—however
valueless—for which medicinal claims are made.
But that physicians should prove equally gullible is a sorry
commentary on the scientific attainments of the followers of
a learned profession.—(Modified from Editorial in The
Journal A. M. A., Nov. 21, 1914.)
THE FEEDING VALUE OF SANATOGEN COMPARED
WITH COMMERCIAL CASEIN WITH
RESPECT TO MAINTENANCE AND GROWTH
John Phillips Street, M.S.
Chemist Connecticut Agricultural Experiment Station
NEW HAVEN, CONN.
The proprietary preparation “Sanatogen” is claimed to consist
of about 95 per cent. casein and 5 per cent. sodium glycerophosphate.
The analysis of C. B. Morison122 is as follows:
Water | 9.97 |
Total nitrogen | 12.81 |
Nitrogen, ppt. by acetic acid | 12.54 |
Casein | 80.01 |
Ether extract | 0.11 |
Ash | 5.59 |
Sulphur, total | 0.73 |
Sulphur in casein | 0.64 |
Phosphorus, total | 1.49 |
Phosphorus in casein | 0.69 |
Phosphorus in inorganic form | 0.11 |
In connection with the above analysis it was demonstrated
that sodium glycerophosphate was present. Sanatogen, therefore,
consists essentially, on the water-free basis, of about 90
per cent. casein and 5 per cent. sodium glycerophosphate, with
a small amount of an unidentified nitrogenous compound containing
both sulphur and phosphorus, and a small amount of
phosphorus in organic combination. The manufacturers
claim that the two essential ingredients exist in Sanatogen as
a definite chemical compound. Certain authorities, on the
other hand, have insisted that the casein and glycerophosphate
have not been chemically combined and that Sanatogen is
simply a mechanical mixture of the two. Which claim is correct
we will not consider here, for indeed it is of little importance,
for whether or not chemically combined the action of
the digestive fluids of the body would speedily break down
the alleged compound into its constituents, and the body
would have casein and the glycerophosphate offered for its
use, just the same as though they had been offered as a
mere mechanical mixture.
It being apparent that Sanatogen consists almost entirely
of casein and sodium glycerophosphate, the former well-known
ingredient making up nine-tenths of its weight, the
question naturally arises how a mixture of these two common
substances can acquire, simply by that admixture, unusually
valuable properties not possessed by the two components.
Leading physiologists quite generally agree that phosphorus
in the form of glycerophosphates influences metabolism very
little. Furthermore, it is obvious that the food value of the
small amount of glycerin present must be slight. It is apparent
therefore, that whatever nutriment or energy Sanatogen
supplies must be dependent on its main constituent casein.
Sanatogen is commonly sold at retail in 100 gm. or 200 gm.
packages for $1 and $1.90, respectively. It is possible that
in larger quantities these prices might be shaded somewhat,
but the fact remains that the ordinary retail purchaser of
Sanatogen pays for it about 1 cent per gram, or about $4.50
per pound. If the value of Sanatogen depends on its casein,
one might ask, in all fairness, why should the patient pay
$4.50 per pound for Sanatogen when he can secure ordinary
commercial casein for 8.5 to 10 cents per pound! (The Casein
Mfg. Co. of New York quoted to me their No. 60 casein at
10 cents per pound in 5-pound lots, 8.5 cents per pound
f. o. b. Bainbridge, N. Y., in 100-pound lots, and 8.5 cents
per pound, freight paid, in 500-pound lots.) I have purchased
Sanatogen from a wholesale druggist at the rate of
$2.75 for 400 gm., or $3.12 per pound, so that under the most
favorable conditions the cost of Sanatogen is more than
thirty times as great as the commercial casein in question.
Is the consumer justified in paying this exceedingly high
price for purified casein? The following feeding experiments
were carried out to answer this query.
FEEDING EXPERIMENTS
White rats were chosen for the experimental animals
because of their adaptability for tests of this kind, as shown
by the extended successful experience of Osborne and Mendel,
and also because, by using white rats, I could take advantage
of the equipment and the experience of my colleagues,
Dr. T. B. Osborne, Prof. L. B. Mendel and Miss Edna L.
Ferry.
The value of Sanatogen as compared with commercial casein
was studied from two points of view:
1. Its value in maintaining the weight of mature rats.
2. Its value in promoting the growth of young rats.
I. THE MAINTENANCE OF WEIGHT OF MATURE RATS
The Sanatogen used contained 12.88 per cent. of nitrogen, and
the casein, 12.82 per cent., so that from the point of view of nitrogen
content they were practically equivalent, gram for gram.
Six healthy male animals were selected, Rats 1, 2 and 3 being fed
the casein ration and Rats 4, 5 and 6 the Sanatogen ration. At
the beginning of the experiment the casein rats were 257, 360 and
376 days old; the Sanatogen rats, 249, 321 and 275 days old, respectively.
These weight conditions, if anything, slightly favored the
Sanatogen rats, as they were slightly less mature, and a greater growth
might naturally be expected.
The rations fed had the following percentage composition:
| Ration 1 | | Ration 2 |
Casein | 20 | | Sanatogen | 20 |
Protein-free milk* | 28 | | Protein-free milk* | 28 |
Lard | 8 | | Lard | 14 |
Unsalted butter | 18 | | Unsalted butter | 18 |
Corn starch | 26 | | Corn starch | 20 |
* Osborne and Mendel: Feeding Experiments with Isolated Food Substances,
Carnegie Inst. of Washington, Publ. 156, 1911, Part 2, p. 80. |
The rats were weighed twice a week for nine weeks, a record of the food consumed also being kept.
Table 1 shows the weekly weights of the casein rats, the weekly gain or loss, and the weekly
consumption of food. Table 2 gives similar data for the Sanatogen rats.
TABLE 1.—WEIGHTS OF CASEIN RATS, ON RATION NO. 1, OVER A PERIOD OF NINE WEEKS*
| Date | Rat 1 | Rat 2 | Rat 3 |
Weight,
gm. | Gain or
Loss, gm. | Food
Eaten, gm. | Weight,
gm. | Gain or
Loss, gm. | Food
Eaten, gm. | Weight,
gm. | Gain or
Loss, gm. | Food
Eaten, gm. |
| 1/2 | 240.0 | ..... | ..... | 264.6 | ..... | ..... | 314.1 | ..... | ..... |
| 1/9 | 250.8 | +10 | .8 | 73 | .7 | 270.9 | + 6 | .3 | 85 | .2 | 314.6 | + 0 | .5 | 95 | .7 |
| 1/16 | 264.3 | +13 | .5 | 89 | .3 | 284.9 | +14 | .0 | 99 | .2 | 308.7 | - 5 | .9 | 117 | .9 |
| 1/23 | 274.9 | +10 | .6 | 90 | .7 | 293.5 | + 8 | .6 | 107 | .7 | 313.8 | + 5 | .1 | 116 | .5 |
| 1/30 | 278.6 | + 3 | .7 | 80 | .2 | 304.5 | +11 | .0 | 100 | .5 | 322.2 | + 8 | .4 | 116 | .7 |
| 2/6 | 278.3 | - 0 | .3 | 82 | .4 | 294.2 | -10 | .3 | 97 | .0 | 311.2 | -11 | .0 | 119 | .9 |
| 2/13 | 288.7 | +10 | .4 | 85 | .5 | 281.5 | -12 | .7 | 94 | .2 | 308.2 | - 3 | .0 | 125 | .6 |
| 2/20 | 286.4 | - 2 | .3 | 86 | .1 | 293.3 | +11 | .8 | 86 | .8 | 309.0 | + 0 | .8 | 126 | .8 |
| 2/27 | 287.6 | + 1 | .2 | 81 | .1 | 297.6 | + 4 | .3 | 100 | .7 | 303.4 | - 5 | .6 | 129 | .3 |
| 3/6 | 292.6 | + 5 | .0 | 80 | .1 | 307.4 | + 9 | .8 | 103 | .1 | 292.4 | -11 | .0 | 101 | .0 |
| Total | ..... | +52 | .6 | 759 | .1 | ..... | +42 | .8 | 874 | .4 | ..... | -21 | .7 | 1,049 | .4 |
Average
per week | ..... | + 5 | .8 | 84 | .3 | ..... | +4 | .8 | 97 | .2 | ..... | -2 | .4 | 116 | .6 |
| * The growth curves of Rats 1, 2 and 3 are given in Chart 1. |
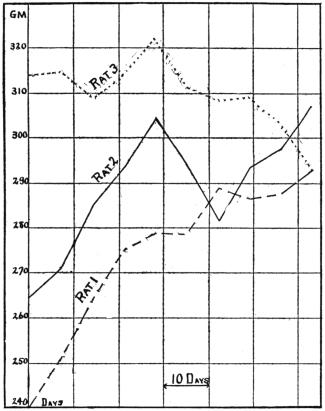
Chart 1.—Growth curves of Rats 1, 2 and 3, on Ration 1: Casein.
TABLE 2.—WEIGHTS OF SANATOGEN RATS, RATION NO. 2, OVER A PERIOD OF NINE WEEKS*
| Date | Rat 4 | Rat 5 | Rat 3 |
Weight,
gm. | Gain or
Loss, gm. | Food
Eaten, gm. | Weight,
gm. | Gain or
Loss, gm. | Food
Eaten, gm. | Weight,
gm. | Gain or
Loss, gm. | Food
Eaten, gm. |
| 1/2 | 251.8 | ..... | ..... | 290.6 | ..... | ..... | 294.0 | ..... | ..... |
| 1/9 | 257.1 | + 5 | .3 | 73 | .4 | 297.2 | + 6 | .6 | 86 | .3 | 289.1 | - 4 | .9 | 58 | .4 |
| 1/16 | 262.4 | + 5 | .3 | 89 | .8 | 292.4 | - 4 | .8 | 88 | .9 | 286.1 | - 3 | .0 | 76 | .8 |
| 1/23 | 274.4 | +12 | .0 | 97 | .3 | 295.7 | + 3 | .3 | 92 | .8 | 287.6 | + 1 | .5 | 85 | .8 |
| 1/30 | 282.7 | + 8 | .3 | 95 | .5 | 299.5 | + 3 | .8 | 92 | .3 | 298.6 | +11 | .0 | 90 | .9 |
| 2/6 | 279.8 | - 2 | .9 | 97 | .7 | 284.2 | -15 | .3 | 87 | .7 | 292.9 | - 5 | .7 | 89 | .8 |
| 2/13 | 271.2 | - 8 | .6 | 96 | .1 | 280.0 | - 4 | .2 | 94 | .9 | 295.1 | + 2 | .2 | 99 | .7 |
| 2/20 | 257.5 | -13 | .7 | 91 | .6 | 274.9 | - 5 | .1 | 96 | .4 | 296.0 | + 0 | .9 | 93 | .9 |
| 2/27 | 258.0 | + 0 | .5 | 93 | .5 | 273.0 | - 1 | .9 | 103 | .3 | 295.0 | - 1 | .0 | 95 | .1 |
| 3/6 | 270.9 | +12 | .9 | 94 | .8 | 278.1 | + 5 | .1 | 94 | .9 | 306.3 | +11 | .3 | 89 | .4 |
| Total | ..... | +19 | .1 | 829 | .7 | ..... | -12 | .5 | 837 | .5 | ..... | +12 | .3 | 779 | .8 |
Average
per week | ..... | + 2 | .7 | 92 | .2 | ..... | - 1 | .4 | 93 | .1 | ..... | + 1 | .4 | 86 | .6 |
| * The growth curves of Rats 4, 5 and 6 are given in Chart 2. |
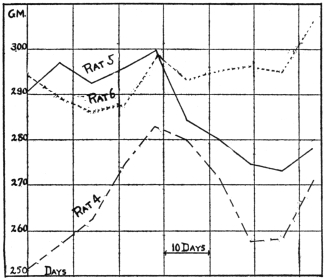
Chart 2.—Growth curves of Rats 4, 5 and 6, on Ration 2: Sanatogen.
Certainly the above data show no superiority of Sanatogen
over commercial casein. In fact the results might be taken
to suggest a slight advantage for the cheaper article, if one
were warranted in drawing fine distinctions from a limited
number of animals.
To conclude, a comparative feeding of six male white rats
during nine weeks showed no nutritive superiority of Sanatogen,
costing $3.12 per pound, over commercial casein costing
10 cents per pound.
II. THE PROMOTION OF GROWTH IN YOUNG RATS
In the second series of experiments ten male rats were used with
four different rations. In these, casein and Sanatogen were compared,
using both Osborne and Mendel’s “protein-free milk” and
their “artificial protein-free milk IV.”123 Butter fat was also substituted
for the unsalted butter used in Rations 1 and 2.
The percentage composition of the rations fed was as follows:
| Ration 3 | | Ration 5 |
Casein | 20 | | Casein | 20 |
Protein-free milk | 28 | | Artificial p.-f. milk IV | 29 |
Lard | 8 | | Lard | 8 |
Butter-fat | 18 | | Butter-fat | 18 |
Corn starch | 26 | | Corn starch | 25 |
| Ration 4 | | Ration 6 |
Sanatogen | 20 | | Sanatogen | 20 |
Protein-free milk | 28 | | Artificial p.-f. milk IV | 29 |
Lard | 14 | | Lard | 14 |
Butter-fat | 18 | | Butter-fat | 18 |
Corn starch | 20 | | Corn starch | 19 |
Two male rats were used with each of Rations 3 and 4, and three
with each of Rations 5 and 6. They were all healthy young rats
ranging from 57 to 71 days old. As in the first series of experiments
the rats were weighed twice a week and a record kept of the
food consumed. Rations 3 and 4 were fed for seventy-seven days,
Rations 5 and 6 for thirty-five days.
Table 3 shows the weekly weights and gains or losses of the rats
fed Rations 3 and 4.
Rat 8 suffered more or less from diarrhea during a considerable
part of the experiment, covering a period from March 16 to April 9,
and from April 13 to May 11. The food intake correspondingly
decreased during those periods.
TABLE 3.—COMPARATIVE WEIGHTS OF RATS FED RATIONS 3 AND 4
| Date | Weights of Rats,
Ration 3, Casein, Protein-Free Milk | Weights of Rats,
Ration 4, Sanatogen, Protein-Free Milk |
| Rat 7 | Rat 8 | Rat 9 | Rat 10 |
Weight,
gm. | Gain or
Loss, gm. | Weight,
gm. | Gain or
Loss, gm. | Weight,
gm. | Gain or
Loss, gm. | Weight,
gm. | Gain or
Loss, gm. |
| 3/9 | 142.2 | ..... | 108.0 | ..... | 127.8 | ..... | 107.0 | ..... |
| 3/16 | 152.8 | +10 | .6 | 120.3 | +12 | .3 | 129.1 | +1 | .3 | 116.0 | +9 | .0 |
| 3/23 | 165.7 | +12 | .9 | 135.1 | +14 | .8 | 144.6 | +15 | .5 | 134.5 | +18 | .5 |
| 3/30 | 178.6 | +12 | .9 | 140.4 | +5 | .3 | 160.0 | +15 | .4 | 149.8 | +15 | .3 |
| 4/6 | 187.8 | +9 | .2 | 148.8 | +8 | .4 | 173.1 | +13 | .1 | 143.8 | -6 | .0 |
| 4/13 | 198.5 | +10 | .7 | 157.6 | +8 | .8 | 185.6 | +12 | .5 | 166.1 | +22 | .3 |
| 4/20 | 204.6 | +6 | .1 | 164.6 | +7 | .0 | 194.8 | +9 | .2 | 179.4 | +13 | .3 |
| 4/27 | 210.3 | +5 | .7 | 167.2 | +2 | .6 | 204.6 | +9 | .8 | 191.0 | +11 | .6 |
| 5/4 | 223.0 | +12 | .7 | 179.6 | +12 | .4 | 222.9 | +18 | .3 | 204.7 | +13 | .7 |
| 5/11 | 223.6 | +0 | .6 | 191.6 | +12 | .0 | 236.4 | +13 | .5 | 210.8 | +6 | .1 |
| 5/18 | 231.5 | +7 | .9 | 203.6 | +12 | .0 | 240.0 | +3 | .6 | 218.6 | +7 | .8 |
| 5/25 | 237.4 | +5 | .9 | 213.9 | +10 | .3 | 247.3 | +7 | .3 | 226.7 | +8 | .1 |
| Total | ..... | +95 | .2 | ..... | +105 | .9 | ..... | +119 | .5 | ..... | +119 | .7 |
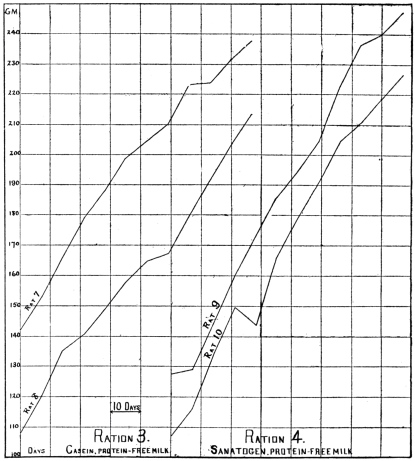
Chart 3.—Growth curves of Rats 7, 8, 9 and 10 on Rations 3,
Casein, protein-free milk, and 4, Sanatogen, protein-free milk.
The difference in the gains in weight shown by the four
rats are not great and certainly do not warrant the conclusion
that Sanatogen possesses any marked superiority over
the commercial casein in promoting growth. The slightly
increased gains shown by Rats 9 and 10 are entirely incommensurate
with the cost of the Sanatogen. All of the rats
showed a vigorous growth and exceeded the average weight
expected for rats of their respective ages, based on the long
series of observations of Osborne and Mendel, as shown in
Table 4.
TABLE 4.—WEIGHT OF RATS 7 TO 10 COMPARED WITH
AVERAGE WEIGHT OF RAT OF SAME AGE
| Rat | Age in Days | Final weight,
gm. | Average Weight
for rat of
Same Age, gm. |
| 7 | 148 | 237 | 204 |
| 8 | 138 | 214 | 201 |
| 9 | 145 | 247 | 204 |
| 10 | 134 | 227 | 197 |
The results secured by Rations 5 and 6 compared with
Rations 3 and 4, the only difference being that natural protein-free
milk was used in the latter and artificial protein-free
milk in the former, are most striking. Such results were
anticipated from the experience of Osborne and Mendel in
feeding similar materials. All of my rats, except Rat 13, a
casein rat, showed a decided loss in weight after five weeks’
feeding on rations containing artificial protein-free milk.
These losses amounted to 19.7, 9.0, 23.1, 6.7 and 27.6 gm.,
whereas rats fed on the same rations, except for the form of
protein-free milk, gained, during the same period of five
weeks, 56.3, 49.6, 57.8 and 59.1 gm. The fact that one casein
rat fed on the artificial protein-free milk showed a substantial
gain is without significance, as such exceptions to the general
rule occur occasionally, and, furthermore, this rat also during
the last week of the experiment likewise lost weight. The
extent of these losses compared with what the average normal
male rat weighs at a corresponding age is shown in Table 7.
TABLE 5.—WEIGHTS OF RATS FED RATION 5, CASEIN,
ARTIFICIAL PROTEIN-FREE MILK*
| Date | Rat 11 | Rat 12 | Rat 13 |
Weight,
gm. | Gain or
Loss, gm. | Weight,
gm. | Gain or
Loss, gm. | Weight,
gm. | Gain or
Loss, gm. |
| 3/9 | 132.1 | ..... | 110.0 | ..... | 99 | .0 | ..... |
| 3/16 | 130.4 | -1 | .7 | 116.8 | +6 | .8 | 110 | .3 | +11 | .3 |
| 3/23 | 136.8 | +6 | .4 | 130.8 | +14 | .0 | 108 | .6 | -1 | .7 |
| 3/30 | 137.1 | +0 | .3 | 138.0 | +7 | .2 | 100 | .2 | -8 | .4 |
| 4/6 | 127.1 | -10 | .0 | 143.9 | +5 | .9 | 95 | .5 | -4 | .7 |
| 4/13 | 112.4 | -14 | .7 | 143.6 | -0 | .3 | 90 | .0 | -5 | .5 |
| Total | ..... | -19 | .7 | ..... | +33 | .6 | ..... | -9 | .0 |
| 4/20 | 146.7 | +33 | .7 | 170.7 | +27 | .1 | 104 | .0 | +14 | .0 |
| * Date 4/20 covers use of mixed food. |
| Rat 13 suffered more or less from diarrhea throughout the whole experiment, and Rat 16 suffered similarly during the first two weeks. |
TABLE 6.—WEIGHTS OF RATS FED RATION 6.—SANATOGEN,
ARTIFICIAL PROTEIN-FREE MILK*
| Date | Rat 14 | Rat 15 | Rat 16 |
Weight,
gm. | Gain or
Loss, gm. | Weight,
gm. | Gain or
Loss, gm. | Weight,
gm. | Gain or
Loss, gm. |
| 3/9 | 118.4 | ..... | 111.3 | ..... | 101.3 | ..... |
| 3/16 | 126.2 | +7 | .8 | 116.2 | +4 | .9 | 100.0 | -1 | .3 |
| 3/23 | 126.6 | +0 | .4 | 123.4 | +7 | .2 | 98.0 | -2 | .0 |
| 3/30 | 124.7 | -1 | .9 | 123.3 | -0 | .1 | 98.7 | +0 | .7 |
| 4/6 | 110.2 | -14 | .5 | 112.3 | -11 | .0 | 88.7 | -10 | .0 |
| 4/13 | 95.3 | -14 | .9 | 104.6 | -7 | .7 | 73.7 | -15 | .0 |
| Total | ..... | -23 | .1 | ..... | -6 | .7 | ..... | -27 | .6 |
| 4/20 | 130.3 | +35 | .0 | 135.3 | +30 | .7 | 106.4 | +32 | .7 |
| * Date 4/20 covers use of mixed food. |
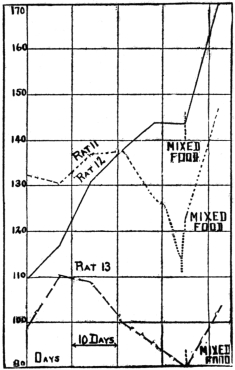
Chart 4.—Growth curves of Rats 11, 12 and 13, on Ration 5:
Casein, artificial protein-free milk.
|
|
Chart 5.—Growth curves of Rats 14, 15 and 16, on Ration 6:
Sanatogen, artificial protein-free milk.
|
TABLE 7.—WEIGHT OF RATS 11 TO 16 COMPARED WITH
AVERAGE WEIGHT OF RAT OF SAME AGE
| Rat | Age, Days | Final Weight,
gm. | Average Weight
of Rat of
Same Age, gm. |
| 11 | 106 | 112 | 167 |
| 12 | 106 | 144 | 167 |
| 13 | 99 | 90 | 162 |
| 14 | 106 | 95 | 167 |
| 15 | 92 | 105 | 153 |
| 16 | 96 | 74 | 158 |
That these losses in weight were not due to any inherent
weakness in the rats is shown by the fact that by feeding
Rats 11 to 16 for one week after the termination of the
experiment with Osborne and Mendel’s “mixed food” (a
mixture of dog bread, sunflower seeds, vegetables and meat)
very large gains were secured in every instance, ranging from
14 to 35 gm.
The growth curves for Rats 7 to 16 are shown in Charts
3, 4 and 5.
To conclude, a comparative feeding of four male white
rats during eleven weeks, showed, if anything, a slightly
greater, but insignificant, increase in weight for Sanatogen
over commercial casein. In a ration in which artificial
had been substituted for natural protein-free milk,
Sanatogen showed no advantage over commercial casein
in checking the failure in weight of the rats.—(From
The Journal A. M. A., Nov. 21, 1914.)
POEHL’S SPERMIN IN ARTERIOSCLEROSIS
A physician addressed the following inquiry to The
Journal:
“In a recent number of the Gazette méd. belge, I read a
detailed report of five cases of arteriosclerosis in which the
patients were all markedly improved and some of them
apparently cured by a course of ‘Sperminum Poehl,’ an
advertised remedy which I have always distrusted and never
prescribed. I am now suffering myself from a somewhat
advanced case of arteriosclerosis and would like to try this
remedy if the Council has learned anything in favor of it
and if there is no reason to fear bad effects from it.”
X. Y. Z.
The Journal replied:
Apparently the exploitation of Poehl’s spermin passed
several years ago from the domain of experimental medicine
to that of nostrums advertised to the laity. So far as we
know, it has received no recent discussion from reliable
clinicians or experimenters. In some medical journals, as
also in lay journals, it is true, one still reads that Poehl’s
spermin is successfully applied in “neurasthenia, senile
marasmus, anemia, rachitis, gout, chronic rheumatism,
syphilis, hysteria, tuberculosis, typhus, diseases of the heart,
nephritis, tabes dorsalis, paralysis, neurasthenic impotence,
overwork, acute diseases, and for convalescents.” Few quack
advertisements would differ much from this puff of Poehl’s
spermin. So far as we know, there is no reason to fear
injurious results from the use of the remedy; neither might
any good be expected from its use in arteriosclerosis.—(From
The Journal A. M. A., April 15, 1911.)
SYRUP OF COCILLANA COMPOUND
A physician in a small town in Nebraska writes: “In
looking over a prescription file not long ago I found a
prescription which I copied and am sending to you. It is a
good example of shotgun prescribing. I do not give the
name of the prescriber, and you will please not mention
from whence this comes. The doctor who wrote this has
had about ten years’ experience.”
Here is the prescription given exactly as transmitted by
our correspondent:
Sp. sticta | Gtt xv |
Sp. ipecac | Gtt x |
Sp. bryonia | Gtt x |
Sp. macrotys | Ʒi |
Bromoform Bronchial Anodyne | ℥ii |
Syrup Cocillana Comp. q. s. ad | ℥vi |
| Teaspoonful every two or three hours. |
It is evident that the prescriber is an eclectic. As a
matter of fact, in a second letter from the physician who
forwarded the prescription, we are informed that the prescriber
is a graduate of an eclectic institution not a thousand
miles from where he practices. The “Sp.” in the prescription
does not mean “Spiritus,” but specific tincture. The prescriber
is an advocate of specific remedies, one of which
should fit the condition, but he is broad-minded enough to
call help from the outside, and so adds fifteen other remedies
to the specific selected, including alcohol. The inability of
one mind to remember all the ingredients of so complex a
mixture will explain the fact that ipecac is duplicated,
occurring both as a specific tincture and as an ingredient
of Bromoform Bronchial Anodyne. The latter, the manufacturers
tell us, contains in one fluidounce:
Alcohol | 5 | per cent. |
Bromoform | 8 | drops |
Ipecac | 1⁄2 | gr. |
Ammonium bromid | 24 | grs. |
Benzoin | 1 | gr. |
Syrup Cocillana Comp., one of the “elegant specialties” of
Parke Davis & Co., of which they certainly ought to be
very proud, contains, we are told, in one fluidounce:
Alcohol | 5 | per cent. |
Heroin hydrochlorid | 8⁄24 | gr. |
Tinct. of euphorbia pilulifera | 120 | min. |
Syrup of wild lettuce | 120 | min. |
Tinct. of cocillana | 40 | min. |
Syrup of squill comp. | 24 | min. |
Cascarin, P. D. & Co. | 8 | grs. |
Menthol | 8⁄100 | gr. |
This “elegant specialty” of Parke, Davis & Co. is not only
a shotgun prescription, but has as one of its ingredients
a mixture itself containing three ingredients, namely: Syrup
Squill Comp. (Coxe’s Hive Syrup), making ten in all—a
beautiful example of scientific pharmacy.
We wonder if our eclectic brother really appreciated that
his prescription, written out, would be as follows:
Sp. sticta | Gtt | xv |
Sp. ipecac | Gtt | x |
Sp. bryonia | Gtt | x |
Sp. macrotys | Ʒi |
Alcohol | 5 | per cent. |
Bromoform | 8 | drops |
Ipecac | 1⁄2 | gr. |
Ammonium bromid | 24 | grs. |
Benzoin | 1 | gr. |
Alcohol | 5 | per cent. |
Heroin hydrochlorid | 8⁄24 | gr. |
Tinct. of euphorbia pilulifera | 120 | min. |
Syrup of wild lettuce | 120 | min. |
Tinct. of cocillana | 40 | min. |
Fluidextract of squill | 60 | min. |
Fluidextract of senega | 60 | min. |
Antimony and potassium tartate | 1 | gr. |
Cascarin, P. D. & Co. | 8 | grs. |
Menthol | 8⁄100 | gr. |
To use a slang expression, this is certainly going some!—(From
The Journal A. M. A., March 18, 1911.)
“A Cough Syrup with a History”
The following letter was received from Dr. Geo. P. Tolman,
Watsonville, Cal.:
To the Editor:—The enclosed advertisement was underscored
and mailed to me by my druggist. The properties
of cocillana are similar to ipecac. The dose of the fluidextract
is from 10 to 20 minims. Each fluidounce of the
extraordinary (!) dark-colored cough marvel of P. D. & Co.
contains 40 minims of the tincture. If the tincture of cocillana
is 10 per cent. (the average tincture strength) you can
see that to get a minimal dose of the drug you would have
to take 21⁄2 fluidounces of the syrup.
“Query: Can we still hang on to the old-fashioned cough
mixtures freshly compounded by our druggists or shall we
put our shoulders to the wheel and help P. D. & Co. save
the nation and make a few dollars for the druggist?”
“The secret of its prompt recognition lay in its unusual
composition.” Nay; its prompt recognition lay in liberal and
persistent advertising. “It quickly made a ‘hit’ with physicians”—because
too many physicians, like other human
beings, are susceptible to the psychology of advertising. Here
is the “unusual composition,” as given by the manufacturers:
“Tinct. Euphorbia pilulifera, 120 mins.; Syrup Wild Lettuce, 120 mins.;
Tinct. Cocillana, 40 mins.; Syrup Squill Compound, 24 mins.;
Cascarin (P. D. & Co.), 8 grs.; Heroin hydrochloride, 8-24 gr.;
Menthol, 8-100 gr.”
The following is a reproduction of the advertisement
referred to:
As we have said above, Parke, Davis & Co. should be
proud of this “elegant specialty.” It would be hard to find
a better specimen of a shotgun prescription; not only does
the prescription contain eight ingredients, but one of these
ingredients (compound syrup of squill) itself contains three.
As our correspondent correctly states, the drug from which
the name (not the action) of the preparation is derived
comes from Bolivia and has properties similar—but evidently
inferior—to ipecac. That it possesses but little therapeutic
value is perhaps best evidenced by the fact that, in spite of
the propaganda made for it by Parke, Davis & Co., neither
the drug nor any preparation of it is listed, so far as we
know, by any other large pharmaceutical house, with one
exception. Besides cocillana the preparation contains two
other obsolete drugs, wild lettuce and euphorbia pilulifera.
The activity of the “cough syrup,” it is needless to say,
depends in the main on the drug which is more or less buried
in the published formula: heroin hydrochlorid. At one time
Parke, Davis & Co. admitted that the preparation owed its
chief value to heroin. In a letter to the Council on Pharmacy
and Chemistry the firm said:
“The physiologic action of this syrup is that which would be suggested
by the constituents. Because of its activity the most prominent
action would be that characteristic of heroin hydrochlorid.”
Without doubt the important ingredient, from the point of
view of therapeutic potency, is the heroin; and it is this drug
doubtless, that makes the mixture a good “repeater.” Syrup
Cocillana Compound is a nostrum sailing under false colors.
Whether its continued use is due to its mysterious, meaningless,
misleading name or merely to insistent and persistent
advertising methods of Parke, Davis & Co. is a question.
Neither explanation is any credit to the medical profession
which tolerates it, or to the physician who prescribes it.—(From
The Journal A. M. A., Feb. 15, 1913.)
AUBERGIER’S SYRUP OF LACTUCARIUM
That clause in the Federal Food and Drugs Act which
requires certain potent drugs to be declared on the label of
the proprietary mixtures containing them has been responsible
for clearing up many mysteries. Physicians have frequently
wondered why they were unable to obtain from the
syrup of lactucarium, U. S. P., the therapeutic results which
they were able to obtain from a proprietary product known
as Aubergier’s Syrup of Lactucarium, sold by Fougera & Co.
at an exorbitant price and put up in “patent-medicine” style.
The milk-juice of lettuce once bore the reputation of being
a soporific—a reputation that has been artificially maintained
largely through the effects of the Aubergier preparation.
With the advent of the Food and Drugs Act the secret of
the soporific effect of the Aubergier product was explained—it
contains morphin.124
The practical difficulties of making a satisfactory syrup
of lactucarium are not realized by most physicians. To such
the following note, presented at a meeting of the Pennsylvania
Pharmaceutical Association by Mr. Louis Emanuel,
president of the Pennsylvania Pharmacy Board, will prove
enlightening:
“Did you ever make a syrup of lactucarium direct from
the crude drug? If you did, shake hands, and let me hail
you as a brother, a brother pharmacist in fact worthy of
the title. If you did not, I am sorry for you; you have
missed something worth knowing.
“The American Journal of Pharmacy tells us that in 1851
‘Aubergier cultivated lactuca and poppy on a larger scale,
in order to obtain lactucarium and opium. Please note the
latter for further reference. In lactucarium he found lactucin,
mannite, resin, cerin, asparmid, brown coloring-substance
and oxalic acid.’ In 1860, in the same publication, Proctor
says: ‘The attention of the medical practitioners has of
late been turned to the syrup of lactucarium, and the preparation
sold usually by apothecaries in this city is that known
as Aubergier’s, a French preparation, made by dissolving
30 parts of alcoholic extract of lactucarium in 500 parts of
boiling water, straining the liquor and adding 15,000 parts
of boiling simple syrup, which is kept boiling, and albuminous
water added from time to time until it is clarified.’ In ’66,
’77, ’78, ’82 and ’84 various writers produced elaborate dissertations
on the supposed improved methods of making this
syrup, but not one has had the temerity of inquiring into the
therapeutic value of this preparation, or to examine the
French preparation to ascertain whence comes its vaunted
superiority.
“The French, it is said, are an impressionable people, but
they appear to have a limit; they do not take any chances
on plain syrup of lactucarium. Theirs contains the added
product, extract of opium. This implies a lack of faith in
soporific properties of lactucarium, and displays a recklessness
in regard to cost and labor.
“The National Dispensatory, fifth edition, says:
“The utility of retaining lactucarium as an official medicine is
very doubtful. It may possibly be desirable as a hypnotic for very
impressionable persons, with whom faith in a remedy supplies its want
of intrinsic efficiency.”
“The official modus operandi for making this syrup looks
laborious, but the innocent-looking task of reducing the drug
to a coarse powder is a revelation to the uninitiated.
“It was {a} hot day in July and it took my 175-pound clerk
and me all that day to reduce 50 gm. of lactucarium to a
satisfactory condition. The stuff looked like old pieces of
discarded rubber shoes, and it really appeared to act like
rubber. After perspiring all day with the Pharmacopeia and
iron mortar, imagine our disgust, if you can, on reading in
the National Dispensatory the following:
“This alcoholic preparation of lactucarium is quite as valueless and
more objectionable than the syrup of the same drug.”
“Moral: Why pay $6.50 a pound for material that has no
medicinal value, and is so hard to manipulate as lactucarium
when decrepit rubber shoes are so cheap? You can have
just as much fun on a hot summer day in reducing the
latter to a coarse powder with clean sand in an iron mortar
as you can with the more expensive material.”
One of the advantages claimed for ready-made prescriptions
over the made-to-order variety, or even over pharmacopeial
preparations, is that they are more elegant in
appearance and less offensive to the nostrils and palate.
This is the common experience of physicians who, having
prescribed some ready-made mixture, wish to change the dose
of one of its constituents and write a prescription or ask
their pharmacists to prepare a similar preparation. The
inability of the pharmacist to prepare a preparation even
approaching the original in appearance, color or taste usually
leads to increased confidence in the skill of the manufacturer
of the proprietary and a correspondingly decreased belief in
the pharmacist’s professional attainments. But these conclusions,
although natural, are based on false premises. As the
proprietary did not have the composition declared on the label,
a mixture based on the formula differed more or less widely
from the proprietary it was expected to resemble.—(From
The Journal A. M. A., Nov. 9, 1912.)
A Protest and a Reply
Three months after publishing the foregoing we received
a nine-page communication from Comar & Co. of Paris, the
promotors of Aubergier’s Syrup of Lactucarium, in which
they took issue with some of the statements in our article.
The company claimed that a possible reason for the difficulty
experienced by Mr. Louis Emmanuel in trying to make the
Syrup of Lactucarium from the crude drug is that he did not
use the same variety of Lactucarium that it employs. Furthermore,
it said that the presence of morphin in the product
was acknowledged before the passage of the Food and Drugs
Act. On more careful investigation, we find that this is true—that
the presence of “a certain proportion of extract of
opium” in the preparation was mentioned even before the
federal Food and Drugs Act compelled the morphin content
to be published on the label. Technically, then, The Journal
was incorrect in making the implication that the medical
profession was not apprised of the fact that Aubergier’s
Syrup of Lactucarium contained morphin; practically it was
right. The information that Comar & Co. gave to physicians
was buried in its advertising “literature” so that it is fair
to assume that not one physician in ten thousand knew—previous
to the Food and Drugs Act—that Aubergier’s Syrup
of Lactucarium contained morphin.—(From The Journal
A. M. A., Nov. 9, 1912.)
TARTARLITHINE
Tartarlithine was examined by two chemists whose reports
indicate that it is an effervescing preparation composed
approximately of 20 per cent. of carbonate of lithium and
about 80 per cent. of tartaric acid. Thus it is simply another
of the hundreds of lithia preparations on the market offered
for the cure of rheumatism. This in spite of the fact that
scientific investigation and clinical experience have demonstrated
that lithia is of very little use in the treatment of that
disease. While the advertisement carries the idea that
Tartarlithine is a product of the Tartarlithine Company,
and that McKesson and Robbins are simply selling agents,
we are informed that the business is owned by McKesson and
Robbins, who under this style manufacture a remedy for
rheumatism.—(Abstracted from The Journal A. M. A., April
23, 1907.)
THOXOS
Thoxos is a “specialty” of John Wyeth and Brother. From
an advertising circular we learn that it “offers to the
physician a rational treatment for Rheumatism, both the
Subacute and Chronic forms, Lithemia, Rheumatic Arthritis,
Gout, Sciatica and the various manifestations of uric diathesis,”
and that “it is a palatable solution of Strontium and
Lithium soluble salts, thirty-two grains, combined with
twenty-four minims Wine of Colchicum Seed and a vegetable
alterative, in each fluidounce, flavored with aromatics.”
This “formula” does not indicate the acid with which the
metals strontium and lithium are combined, or what the
“vegetable alterative” is; it is essentially a secret preparation.
To learn what the missing and presumably active
ingredients are an analysis was made by our chemists.
LABORATORY REPORT
One original bottle of Thoxos, John Wyeth and Brother,
Philadelphia, was purchased and submitted to analysis.
The bottle contained a brown liquid having an aromatic
odor and a sweet taste. The specific gravity of the liquid
was 1.118 at 15 C. (60 F.) The solution was acid to
litmus. Qualitatively the following constituents were
detected: strontium, potassium, sodium, lithium, ammonium,
salicylate, iodid, glycerin, alkaloid, alcohol and water.
By the smell and taste, oil of wintergreen, or methyl salicylate,
and oil of sassafras were recognized. Positive tests
for a saponin-like body indicated the probable presence
of sarsaparilla.
Quantitatively the following results were obtained:
Ammonia (NH3) | 0.006 | per cent. |
Lithium (Li) | 0.13 | per cent. |
Sodium (Na) | 0.03 | per cent. |
Strontium (Sr.) | 1.03 | per cent. |
Iodid (I) | 0.46 | per cent. |
Salicylate (C6H4.OH.COO) | 4.19 | per cent. |
Glycerin | 19.2 | per cent. |
From the analytic results it would appear that the
preparation contains approximately potassium iodid, 0.67 gm.
per hundred c.c., or 3 grains per fluidounce; lithium
salicylate [Li(C7H5O3)], 0.9 gm. per hundred c.c., or 4
grains per fluidounce; strontium salicylate [Sr(C7H5O3)2-2H2O],
5.75 gm. per hundred c.c., or 26 grains per fluidounce,
and some salicylic acid combined with sodium and
also in the free state. The total salicylate found is equal
to 5.47 gm. of sodium salicylate per hundred c.c., or 25
grains per fluidounce.
As strontium salicylate and lithium salicylate are now
generally considered to differ but slightly, if at all, in their
action from that of sodium salicylate, each dose of Thoxos,
1 teaspoonful or 4 c.c., may be considered the equivalent of
0.2 gm. or 3 grains of sodium salicylate with a fractional
dose of colchicum. Hence this nostrum—for this is the
correct definition—is a mixture of no more value than a
prescription of sodium salicylate with a fractional dose of
potassium iodid and colchicum, one that any doctor could
write and any druggist dispense. Yet it is doubtless prescribed
by physicians under the belief that it possesses some
occult power not to be found in ordinary drugs and their combinations.
To prescribe Thoxos is to prescribe a name, and
the patient who takes it would be as well off if he went
to the nearest drug store and purchased a bottle of any of the
thousand and one rheumatism cures with which the country
is flooded.—(From The Journal A. M. A., March 21, 1914.)
TRYPSOGEN
Besides exploiting a clay poultice—“Antithermoline”—the
G. W. Carnrick Company appears to be chiefly concerned in
the promotion of “internal secretion” specialties; a class of
preparations the therapeutic value of which is problematical.
Thus it markets the diabetes remedy, “Trypsogen” tablets,
said to contain “the enzyme of the islands of Langerhans
with the tryptic and amylolytic ferments of the pancreas”
along with gold bromid and arsenic bromid; Secretogen
Elixir, said to be “prepared from gastric secretin obtained
from the pyloric antrum and pancreatic secretin from the
duodenum, combined with the enzymes of the peptic glands,
and one-twentieth of one per cent. HCl”; Secretogen Tablets,
said to be “prepared from prosecretin and succus entericus
obtained from the epithelial cells of the duodenum, combined
with pancreatic extract”; Kinazyme, “a preparation of extract
of spleen, reinforced with trypsin, amylopsin and calcium
lactate.”
While great claims have been made for Trypsogen and
while it has been most widely advertised, it is the consensus
of opinion of the most eminent students of the question
that pancreas is not really efficacious in diabetes. Were it
of any value in this disease, it would have won world-wide
recognition for itself ere now, in view of the great enthusiasm
with which the discovery of the relation of the pancreas to
diabetes was received and of the enormous amount of
clinical, as well as animal, experimentation that followed.
As the conditions of experiment in this question are
extremely complex, it is not surprising that occasionally
apparently positive results should have been obtained. Were
it really useful, it should have yielded positive results much
more uniformly.
Furthermore, if pancreas were really efficacious in the
treatment of diabetes mellitus, the addition of arsenic, of
gold, of bromid would be entirely unnecessary.
Even were it granted that pancreas extracts are valuable
in the treatment of diabetes, and that gold and arsenic also
have beneficial effects, it is our opinion that Trypsogen
should be considered an unscientific shotgun mixture, because
fixed combinations of remedies of different potencies, such as
arsenic, gold, bromid and pancreas, are therapeutically
erroneous, as they do not permit of that accurate adjustment
of dosage of each ingredient that is indispensable to obtain
maximum benefit with minimum danger of poisoning.
Antithermoline and Trypsogen were at one time described
in New and Nonofficial Remedies. These preparations were
omitted when the Council’s rules were revised some years
ago.
When the Council was first organized it undertook only
the correction of the most serious abuses that had become
a part of the proprietary medicine business, and paid less
attention to the therapeutic worth of a remedy; thus at that
time it admitted both Antithermoline and Trypsogen to
New and Nonofficial Remedies. Since then the Council has
modified its rules to exclude unscientific mixtures marketed
under names that are misleading or therapeutically suggestive.
Accordingly it rescinded the acceptance of Antithermoline,
which was essentially the official clay poultice,
Cataplasma Kaolini, U. S. P. For similar reasons and
because the therapeutic claims were held unwarranted
Trypsogen has been omitted from New and Nonofficial
Remedies.
It is to be regretted that the progress of research should
be hindered and the value of genuine products of internal
secretion be depreciated by confusion with such shotgun
mixtures and asserted remedies, whose claims have received
no scientific confirmation.—(From The Journal A. M. A.,
Nov. 1, 1913.)
TYREE’S ANTISEPTIC POWDER[AQ]
Now Advertised Direct to the Public as the “Best
Preventative Known”
When the history of the “patent medicine” business comes
to be written impartially and fairly, it will be realized that
we, the medical profession, have been in no small degree
responsible for its growth. Not a few widely advertised
nostrums owe their commercial success solely to the ill
considered use accorded them by physicians, to whom they
were first exploited. As a well-known and brilliant advertising
man once said:
“The patent medicine of the future is one that will be advertised
only to doctors. Some of the most profitable remedies of the present
time are of this class. They are called proprietary remedies. The
general public never hears of them through the daily press. All their
publicity is secured through the medical press, by means of the manufacturer’s
literature, sometimes gotten out in the shape of a medical
journal, and through samples to doctors.... The medical papers
will reap the harvest and the physician himself, always so loud in the
denunciation of ‘patent medicines,’ will be the most important medium
of advertising at the command of the proprietary manufacturer. In fact,
he is that to-day.”

Advertisement from a newspaper—Tyree’s Powder as a “Patent Medicine”
of the “Preventive” Type.
Of the conditions here described probably no better example
can be found than Tyree’s Antiseptic Powder. For years
this preparation was advertised to the medical profession
under claims that were fraudulent as to both composition
and therapeutic effect. Analysis published in The Journal125
proved that the formula given out by Tyree was absolutely
false and that the preparation was, essentially, nothing but
a simple mixture of sulphate of zinc and boric acid.
From the first it would seem that the manufacturers of
this mixture had for their objective point that period when,
thanks to the use of the nostrum by physicians, it would be
widely purchased by the public. Lavish advertising was done
in medical journals and Tyree’s Antiseptic Powder gained
admission to the pages of even those journals which required
the publication of a “formula”—for a formula was forthcoming.
The Journal itself, until seven years ago, carried
the advertisements with a “formula” until chemical examination
proved the falsity of the formula, and of the therapeutic
claims made for the product. The medical profession in its
turn prescribed the nostrum and the “original package”
scheme did the rest.

Advertisement from Medical Journal—Tyree’s Powder as a Highly
Respectable “Ethical Proprietary.”
Now, it seems, Tyree considers his preparation so well
known that he can be independent either of the assistance of
the physician or of his good-will. For Tyree’s powder now
goes to the public direct and newspaper readers find it
advertised as:
“Ideal for douche.”
“Unequalled as a douche.”
“Best preventative known.”
“Unequalled as a preventative.”
“Has no equal as a preventative.”
And the following, whose very truth must bring the blush
of shame to all physicians who have the interest of scientific
medicine at heart:
“Prescribed by physicians all over the world for twenty-one years.”
“Ask your doctor or send for booklet.”
“Used by doctors for the last twenty-one years.”
“One of the highest tributes paid Tyree’s Antiseptic Powder is the
fact that the most successful physicians have been using it for the
last twenty-one years.”
Not that Tyree has entirely forsaken the medical journals,
although he seems to be dropping them one by one. At the
beginning of this year at least fifteen medical journals were
carrying the Tyree advertisement; by March the number had
fallen to seven, while in June the only journals carrying it
were:
| Medical Record | Chicago Medical Recorder |
| American Journal of Obstetrics | Pacific Medical Journal |
Those who answer the newspaper advertisements receive
a free sample of the powder and several leaflets and circulars
giving the various uses (?) of the nostrum. Incidentally
these leaflets advertise, in addition, Tyree’s “Elixir
Buchu and Hyoscyamus Comp.,” which is recommended, in
various combinations, for such conditions as acute nephritis,
epilepsy, neurasthenia, gonorrhea and delirium tremens.
Bearing in mind the claim that is made in the newspaper
advertisements that Tyree’s Antiseptic Powder is the “best
preventative” known, it is interesting to see what Tyree has
to say to those druggists whom he offers to supply with
circulars for free distribution:
“As these circulars deal with the care of rubber goods, for both
medicinal and toilet purposes, they are of great value to the customer
and will be retained for further reference. They are boosters for your
rubber goods sales, too.”
That a nostrum of this sort should go to the public is
not surprising, but that it should have reached the public
through the instrumentality of the medical profession is a
serious reflection on the judgment of physicians. But the
incident has a bright side. That the exploiters of this
nostrum no longer find it profitable to use medical journals
as a means of getting their stuff to the public but must needs
use the more expensive newspaper advertising, is cause for
optimism. It means that physicians are no longer prescribing,
indiscriminately, proprietary products and that they
are refusing to be, what they have been in the past, the
unpaid distributing agents for nostrum venders.—(From
The Journal A. M. A., Aug. 24, 1912.)
VAPO-CRESOLENE
Vapo-Cresolene has been examined in the American
Medical Association’s laboratory and the chemists’ report
follows:
According to the statements on the trade package, Vapo-Cresolene
“is a product of coal-tar possessing far greater
power than carbolic acid in destroying germs of disease.”
It is recommended as a remedy for a number of diseases,
including croup, catarrh and diphtheria. According to the
manufacturers, it should be used only in “the Cresolene
vaporizer,” which makes it “unequaled for the disinfection
of sick rooms” and the “safest and simplest method of
destroying infection and purifying the air.” From the
examination we conclude that Vapo-Cresolene is essentially
cresol and corresponds in every respect to cresol U. S. P.
(Physician’s Manual, page 36).
This report indicates that Vapo-Cresolene is a member of
that class of proprietaries in which an ordinary product
is endowed, by the manufacturer, with extraordinary virtues.
The type is so common and has been referred to so frequently
that but for the dangers attendant on the inhalation of any
of the phenols, this particular product need not have been
mentioned.—(From The Journal A. M. A., April 4, 1908.)
VASOGEN AND IODOVASOGEN
Another Case in Which Independent Analyses and Manufacturers’
Labels Disagree
Vasogen, a product of Pearson & Company, Hamburg,
Germany, has been put on the market under the various
designations, “oxygenated vaseline,” “water-soluble hydrocarbon”
and “oxygenated carbon.” The manufacturers, and
also their American agents, Lehn & Fink, claim that by a
special process the apparent impossibility of saponifying
petrolatum has been overcome with Vasogen as the result.
Disinterested chemists who have analyzed Vasogen find that
the product consists essentially of an ammonium soap and
petrolatum—practically an ammonia liniment mixed with
petrolatum.
Just as petrolatum under its various trade names was at
one time recommended as a universal ointment base, so
vasogen is recommended promiscuously as a vehicle for
remedies applied externally and even for internal medication—needless
to say in many cases in which it is directly
contra-indicated.
Iodovasogen, recommended for external application as a
substitute for tincture of iodin, was examined by Zernik
in 1905, who found that the iodin existed not as a free
iodin, but chiefly as ammonium iodid. The therapeutic
character of the preparation is thus entirely different from
that to be inferred from the labels and elsewhere, since
the counter-irritant effects of free iodin are of course absent
in ammonium iodid. Pearson & Co. now claim that when
Zernik’s findings were published they immediately modified
their statements on the label in accordance with the truth.
This is denied by Dr. Lungwitz, the editor of the Therapeutische
Rundschau (Apotheker Zeitung, 1908, p. 900), who
vigorously criticizes the misrepresentation made by Pearson
& Co. in regard to Iodovasogen. He calls attention to the fact
that, while Zernick’s results were published over three years
ago, the labels which are in use today still bear the statement
that Iodovasogen consists of Vasogen 90 parts and resublimed
iodin 10 parts, and Vasogen 94 parts and resublimed iodin
6 parts, respectively.
As Iodovasogen and Vasogen in various combinations are
being advertised to the physicians in the United States, the
above information from our German exchanges is worthy of
consideration.—(From The Journal A. M. A., Feb. 13, 1909.)
VIBURNUM COMPOUND—AND OTHER NOSTRUMS
A number of drugs have some reputation for therapeutic
value without there being any particular evidence to substantiate
the claims. Viburnum, concerning which we recently
received the following letter, is one of these drugs:
To the Editor:—Have you made an analysis of Viburnum
Compound? Extravagant claims are being made for it and
I cannot put my hand on any data. A patient has asked me
concerning it and I wish to advise her honestly. I do not
know but that there may be several “viburnum compounds.”
I rarely use any of these “put-up” preparations, and hence
know but little about them.
A. J. Hesser, M.D., Pittsburgh, Pa.
No analysis of Hayden’s Viburnum Compound, to which
our correspondent refers, has been made in the Association
laboratory. According to advertising circulars, the preparation
contains American skullcap (Scutellaria lateriflora),
cramp-bark (Viburnum opulus) and wild yam (Dioscorea
villosa). Since these drugs contain no well-defined therapeutically
active ingredients, an analysis of the preparation
would necessarily be unsatisfactory.
A number of drugs have in some way obtained a reputation
as being valuable in the treatment of diseases of women,
without their therapeutic claims ever having been proved.
It is said that some were used by the aborigines for such
affections and we find a considerable number of them combined
in various nostrums (sometimes with therapeutically
active drugs) and exploited for the cure of female disorders,
under most extravagant and usually absurd claims. Thus
“Pierce’s Favorite Prescription” is advertised as containing
black cohosh, blue cohosh, goldenseal, lady’s-slipper and
false unicorn-root; “Dioviburnia” (Dios Chemical Co.) as
containing American skullcap, cramp-bark, wild yam, blue
cohosh, black haw, star-grass, trailing arbutus and false
unicorn-root; “Viburnumal” (Louisville Pharmacal Works)
as containing American skullcap, cramp-bark, wild yam, star-grass
and motherwort.
Most pharmaceutical houses, following the lead of nostrum-makers,
put similar mixtures on the market; for example:
“Elixir of Viburnum Compound” (Nelson, Baker & Co.) is
said to contain cramp-bark, American skullcap and wild yam;
“Elixir of Hydrastis and Viburnum Compound” (Smith,
Kline & French Co.), cramp-bark, goldenseal, Jamaica dogwood
and pulsatilla; “Elixir of Hydrastis and Cramp Bark
Compound” (Parke, Davis & Co.), cramp-bark, hydrastis,
Jamaica dogwood and pulsatilla; “Fluid Extract of Cramp
Bark Compound” (H. K. Mulford Co.), American skullcap,
cramp-bark and wild yam; “Mother’s Cordial” (Eli Lilly &
Co.), cramp-bark, blue cohosh, false unicorn and squaw vine;
“Uterine Sedative Elixir” (Eli Lilly & Co.), cramp-bark,
goldenseal, Jamaica dogwood and pulsatilla; “Vibutero”
(Fred. Stearns & Co.), cramp-bark, wild yam, black haw,
squaw vine, Jamaica dogwood, saw palmetto and pulsatilla.
Practically all of these drugs except goldenseal are
ignored in the standard works on pharmacology. Further, the
results of careful examination by the Council on Pharmacy
and Chemistry of the therapeutic claims made for most of
them shows that these claims are not sustained by reliable
clinical experience.
The fact is that the popularity of preparations of this kind
is purely an artificially created one. A nostrum containing,
let us say, extractives of some little-used or worthless drugs
is put on the market and heavily advertised. Should it be
advertised in a manner to make it sell, a host of imitations
appear and the large pharmaceutical houses put out substitutes
for it. The uncritical physician does the rest. He prescribes
it indiscriminately in the class of cases for which it is
advertised. Naturally, a certain proportion of the patients
who take it recover, and the recoveries are credited to the
nostrum. A vicious circle is thus established and the demand
for the stuff increases. Its sale, together with the sale of
similar products, continues until the overwhelming experience
of those who have prescribed it proves its uselessness. In the
meantime the manufacturers have reaped a harvest, at the
expense both of the public and of the medical profession. And
the manufacturers’ excuse for putting such absurd “specialties”
on the market is that physicians prescribe them!—(From
The Journal A. M. A., Aug. 31, 1912.)
WHEELER’S NERVE VITALIZER
Names of nostrums often mislead by the use of fake
nomenclature giving erroneous ideas regarding the composition
of the preparation or misrepresenting the true action of
the nostrum. As an example of the latter class Wheeler’s
Nerve Vitalizer was examined in the Association laboratory
and found to be not a vitalizer, as the name implies, but
rather a nerve sedative. The results of the examination
follow:
Wheeler’s Nerve Vitalizer was packed in a carton bearing
the name of the preparation, its manufacturers, “The
J. W. Brant Co., Ltd., Albion, Mich.,” and an exhaustive list
of the diseases for which the product is intended, besides the
general statement that it is a cure for “all nervous diseases.”
The “Vitalizer” is a brown, syrupy liquid having a peculiar
salty taste partially masked by licorice. Qualitative tests
showed the presence of sodium, potassium and bromin, and no
other acid radicals except small quantities of chlorin. It was
decided therefore that the preparation contained a mixture
of sodium and potassium bromids. In order to separate the
chlorin and bromin the preparation was evaporated, charred,
extracted with water and acetic acid and potassium permanganate
added and the mixture distilled with steam until all
the bromin had been distilled over, thus leaving the chlorin
in the distilling flask. The solution in the distilling flask
was then treated with silver nitrate and the chlorin estimated
in the usual way. The quantity thus obtained was
subtracted from the total silver bromid and chlorin obtained
by precipitating a solution of the preparation with silver
nitrate and the remainder calculated to bromin.
By this method several samples of 5 c.c. each of the
preparation yielded an average of 0.0059 cm. silver chlorid
or 0.0012 gm. per c.c. The total silver haloids obtained
by direct precipitation of the diluted preparation was found
to be 0.3158 gm. per c.c., thus leaving 0.3146 gm. silver bromid
to be calculated to bromin.
The total sodium and potassium was obtained in the
usual way and the potassium determined as the chlor-platinate
and the sodium calculated from the difference. By
this method the quantity of sodium found calculated to
sodium bromid gave the following results: (a) 0.0629 gm.
and (b) 0.0632 gm., or an average of 0.063 gm. per c.c.
From the potassium estimations the following were calculated:
(a) 0.1264 gm. potassium bromid and (b) 0.1259 gm.
potassium bromid per c.c., an average of 0.1261 gm.
potassium bromid per c.c.
The bromids calculated from the sodium and potassium
determinations were found to be 0.0630 gm. sodium bromid
and 0.1261 gm. potassium bromid per c.c., the equivalent of
0.3139 gm. silver bromid. The total silver bromid obtained
was 0.3146 gm., showing practical agreement with the total
bromids calculated from the sodium and potassium determinations.
The preparation contained then 6.30 gm. sodium bromid
and 12.61 gm. potassium bromid per 100 c.c., or 9.73 grains
of potassium bromid and 4.86 grains sodium bromid, a
bromid content equal to 15.35 grains potassium bromid per
fluid dram.
ZYMOTOID
A Fraud of the Liquozone-Oxytonic-Septicide Type
Dr. Arnold’s Zymotoid, a nostrum manufactured by
Arnold’s Zymotoid Company, Rockford, Ill., is claimed to be
an “antiseptic, germicide and antiphlogistic” which “has
absolutely no peer in medicine.” According to the statements
of the manufacturer, Zymotoid is “successfully employed not
only as an external dressing on all wounded and diseased
surfaces, but in all zymotic conditions wherein a reliable
antiseptic and germicide is needed internally.” And in telling
physicians of the great value of Zymotoid the company
says:
“We assured them that if they would simply place Zymotoid ‘next’
to any wounded surface—and nothing else—they would have no
inflammation, no suppuration, no infection or blood poison. Its prompt
use in all cases where such trouble arises gives immediate and certain
relief.”
This is a large contract to be undertaken by Zymotoid—or
any other preparation—which, as will be shown, consists
principally of boric acid and water. The company also
appends to its announcement concerning Zymotoid a number
of the usual testimonials and a lot of alleged “case reports.”
Zymotoid seems to be exploited principally by circulars
addressed to physicians and by agents who attempt to sell
it to physicians. They also try to work factories and other
large employers of labor. In their circular to physicians
they claim that “Zymotoid is strictly ethical.” And “we
publish its composition.” The composition given is: “sulphur,
niter, cinnamon and boric acid in gaseous solution.” It is
also claimed to be “a chemical compound—not a mixture—which
is wholly non-toxic and can be used as freely as
desired internally absolutely without harm to the smallest
child.” On the label of the Zymotoid package is the following:
“Zymotoid is a concentrated chemical compound consisting of the
solids and gases of sulphur, potassium nitrate, cinnamon and carbon
held in a solution of boric acid.”
A specimen of Zymotoid was examined by our chemists and
their report follows. As will be seen, it is simply another
fraud of the Liquozone-Oxytonic-Septicide type.
LABORATORY REPORT ON ZYMOTOID
Zymotoid is a pale yellow liquid having a strong odor
like sulphur dioxid. No odor suggestive of cinnamon was
observed even after the sulphur dioxid had been fixed by
the addition of an alkali. Qualitative tests indicated the
presence of boric acid, sulphuric acid, sulphur dioxid and
traces each, of a nitrate, potassium and some unidentified
organic matter. Alkaloids, cinnamic acid, glycerin and
soaps were absent. From the results of the quantitative
examination it is concluded that the composition of
Zymotoid is essentially as follows:126
Boric acid (H3BO3) | 0.637 | gm. |
Sulphur dioxid (SO2) | 0.129 | gm. |
Sulphuric acid (H2SO4) | 0.048 | gm. |
Potassium nitrate | trace | |
Unidentified organic matter | trace | |
Water (by difference) to make | 100 | c.c. |
The analysis shows that but for the presence of boric acid
the composition of Zymotoid is similar to other fraudulent
“microbe killers” which have been exploited in recent years
and of which some have been declared misbranded by the
federal government. For example, “Radam’s Microbe
Killer”127 was found by the federal chemists to be composed
of water, containing small quantities of sulphur dioxid and
sulphuric acid. “Liquozone,” another nostrum which was
widely exploited a few years ago, is said to have a similar
composition.128 According to an analysis made at the North
Dakota Agricultural Experiment Station,129 “Oxytonic” has a
similar composition. The nostrum “Septicide,” was found
by the federal chemists to be composed of water with small
quantities of sulphur dioxid, sulphuric acid and a trace of a
nitrate.—(From The Journal A. M. A., April 6, 1912.)
PART IV
CONTRIBUTIONS FROM THE JOURNAL:
MISCELLANEOUS MATTER
ACETPHENETIDIN AND PHENACETIN—THEIR
RELATIVE PURITY
Until six years ago the chemical product known as phenacetin
was patented both as to process and to product. As
the patent ran out at that time, anyone, of course, could
manufacture it. It was placed in the Pharmacopeia under
the name “acetphenetidin.” It is on the market now under
both names, “phenacetin” and “acetphenetidin.” The price
of the former is five times130 that of the latter, hence it is
rather important to know whether or not one is, in any way,
better or purer than the other. The original patentees or
manufacturers, the Farbenfabriken of Elberfeld Company,
market the product under the name “phenacetin” and also
under the official name “acetphenetidin,” the former at about
33 cents an ounce and the latter at about 6 or 7 cents an
ounce. Evidently these people believe that acetphenetidin is
all right since their price-list says: “Our product is of the
highest standard of purity,” and in another place: “On
account of the low price of acetphenetidinum, U. S. P., it is
especially suitable for the manufacture of medicinal specialties,
such as headache powders, etc.” Remember that it is
the manufacturers of phenacetin who say this.
The question arose whether or not phenacetin differs from
acetphenetidin. If it does, then physicians should know it.
An inquiry was addressed to Farbenfabriken of Elberfeld
Company and also to Lehn & Fink, two firms which market
the product in this country under both names, asking in
what respect the two products differ. No answer was received
from either firm. With the object of answering the question
our chemists have investigated the preparations on the
market, both those sold under the name “phenacetin” and
those under the official title “acetphenetidin.” The following
is a summary of their report:131
THE CHEMISTS’ REPORT
Physical Appearance.—All the specimens were found to
be fine white crystalline powders, differing somewhat in
appearance as follows: Four specimens—Acetphenetidin
(Farbenfabriken), Phenacetin (Specimen 1132—Farbenfabriken),
Phenacetin (Specimen 2132—Farbenfabriken) and
Acetphenetidin (Squibb)—appeared very much alike, each
being a very fine crystalline powder, differing only slightly
as to fineness. Five other specimens—Phenacetin (Lehn
& Fink), Acetphenetidin, U. S. P. (Lehn & Fink),
Acetphenetidin (Merck), and two specimens of Acetphenetidin
(Powers-Weightman-Rosengarten), had the same
general appearance, each consisting of a fine crystalline
powder containing a considerable proportion of large rectangular
plates. Three specimens—Acetphenetidin (Mallinckrodt)
and two specimens of Acetphenetidin (Powers-Weightman-Rosengarten)—had
the same general appearance,
being a moderately fine and homogeneous crystalline
powder. When examined microscopically with a low-power
lens the Mallinckrodt product appeared to consist principally
of rectangular prisms and the Powers-Weightman-Rosengarten
product to be made up largely of plates.
Identity.—All of the specimens when tested side by side
responded to and complied with the identity tests of the
United States, British, German, Swiss, Dutch, Swedish,
Spanish, and Danish pharmacopeias. The reactions given
by the several specimens were all the same, showing no
difference in any case.
Melting-Points.—As a further proof of identity and similarity
the melting-points of the different specimens were
taken and found to be: Acetphenetidin (Farbenfabriken),
134.2 C.; Phenacetin (Specimen 1—Farbenfabriken) 133.7
C.; Phenacetin (Lehn & Fink), 134.7 C.; Acetphenetidin
(Lehn & Fink) 134.9 C.; Acetphenetidin (Powers-Weightman-Rosengarten),
(1) 134.3 C., (2) 133.6 C., (3) 134.7
C., (4) 134.7 C.; Acetphenetidin (Squibb) 134.2 C.; Acetphenetidin
(Merck), 134.8 C., and Acetphenetidin (Mallinckrodt),
134.2 C. The melting-point is given as 135 C.
in the British, French and Spanish pharmacopeias, and as
134 to 135 C. in the United States, German, Swiss, Danish,
Swedish and Dutch pharmacopeias. Thus all comply with
the standard given in our pharmacopeia and most foreign
pharmacopeias with two exceptions and those respectively
only 0.3 C. and 0.4 C. low.
Absence of Acetanilid.—The absence of acetanilid in all
the specimens was indicated by the bromin test of the
United States, British, German, Swiss, Dutch, Swedish and
Danish pharmacopeias.
Absence of Carbonizable Matter.—The absence of carbonizable
matter was shown in all specimens by the sulphuric
acid test of the United States, British, German, French,
Swiss, Dutch, Swedish and Spanish pharmacopeias.
Water-Soluble Matter.—All specimens when tested for
excess of water-soluble matter came well within the limit
(0.50 per cent.) set by the French pharmacopeia, the greatest
amount being 0.20 per cent.
Ash.—When heated, all the specimens were found to
yield practically no ash, the residues from 1 gm. samples
weighing in no case more than 0.0004 gm.
Absence of Paraphenetidin.—When tested by the methods
of the United States, British, German and French pharmacopeias,
the absence of an impurity of paraphenetidin was
shown in all specimens, with the exception of one specimen
obtained from Powers-Weightman-Rosengarten Co., which
gave a positive, though not strong, reaction and two other
specimens of the same firm which reacted still more faintly.
TABLE SHOWING RESULTS OF ANALYSES OF VARIOUS
SPECIMENS OF ACETPHENETIDIN AND PHENACETIN*
| Name | Physical
Appearance | Melting
Point
(Corr.) C. | Water-Soluble
Matter in,
per Cent. | Ash,
per Cent. | Para-
phenetidin,
U. S. P.
Test† | Para-
phenetidin,
Swiss
Test† |
Acetphenetidin
(Farbenfabriken) (1) | Very fine homogeneous crystalline powder | 134.2 | 0.17 | 0.02 | – | + |
Phenacetin
(Farbenfabriken) | Very fine homogeneous crystalline powder | 133.7 | 0.06 | 0.00 | – | + |
Phenacetin
(Lehn & Fink) | Fine crystalline powder,
not uniform | 134.7 | 0.11 | 0.02 | – | + |
Acetphenetidin
(Lehn & Fink) | Fine crystalline powder,
not uniform | 134.8 | 0.13 | 0.00 | – | + |
Acetphenetidin
(P. W. R.) (1) | Homogeneous crystalline powder | 134.3 | 0.19 | 0.03 | + | + |
Acetphenetidin
(P. W. R.) (2) | Homogeneous crystalline powder | 134.7 | 0.16 | 0.02 | + | + |
Acetphenetidin
(P. W. R.) (3) | Homogeneous crystalline powder | 134.7 | 0.14 | 0.02 | + | + |
Acetphenetidin
(P. W. R.) (4) | Fine crystalline powder | 133.6 | 0.20 | 0.01 | – | – |
Acetphenetidin
(Squibb) | Fine crystalline powder | 134.3 | 0.19 | 0.00 | – | + |
Acetphenetidin
(Merck) | Fine crystalline powder | 134.8 | 0.15 | 0.03 | – | – |
Acetphenetidin
(Mallinckrodt) | Fine crystalline powder | 134.2 | 0.11 | 0.01 | – | – |
| * In all cases identity was confirmed; acetanilid was absent; carbonizable matter was absent. |
| † In this column plus indicates presence; minus, absence. |
While this firm’s product alone gave any reaction whatever
when the U. S. P. test for paraphenetidin was applied with
the test of the Swiss pharmacopeia, all but Acetphenetidin
(Mallinckrodt), Acetphenetidin (Merck) and one specimen
of Powers-Weightman-Rosengarten Co. gave positive,
though very faint, reactions, indicating that the majority of
specimens, including those of the original manufacturer,
contain a minute trace of this impurity.
Our findings regarding the product of Powers-Weightman-Rosengarten
Co. having been communicated to this
firm, their correctness was acknowledged. At the same
time the firm wrote: “All that we have on hand now
gives negative tests for paraphenetidin, and we believe our
present records are correct when we state that all lots
which we are supplying now, and have been supplying for
some time past, answer all U. S. P. requirements.”
This examination appears to demonstrate that the chemical
substance, para-acetphenetidin, whether sold as acetphenetidin,
U. S. P., or as phenacetin, is practically identical.
The impurity of the product of some of the specimens coming
from Powers-Weightman-Rosengarten Co. is too slight
to be considered dangerous. Furthermore, a comparison of
the “lot numbers” indicates that this firm has been improving
its product steadily so that in the future its assurances
of an unimpeachable product may be relied on. Inasmuch,
therefore, as acetphenetidin complies with all the pharmacopeial
requirements as to identity and purity, in just the
same way as phenacetin, which sells for as high as five times
the price of acetphenetidin, physicians need not hesitate in
using the title of the U. S. P. “acetphenetidin” when prescribing
this produce.—(From The Journal A. M. A., March
16, 1912.)
Acetphenetidin and Phenacetin
A physician-pharmacist writes: “If a prescription calls
for ‘phenacetin,’ should the pharmacist dispense ‘phenacetin-Bayer’—that
is, the phenacetin manufactured by the original
patentee—or would he be justified in dispensing the official
acetphenetidin, manufactured by any reliable chemical or
pharmaceutical house?”
Unless the pharmacist happens to know that the physician
in writing the prescription desired the Bayer brand, he
would be justified in dispensing acetphenetidin, U. S. P. As
a general thing, physicians use the word “phenacetin” without
intending to prescribe any particular brand or make,
simply because they are familiar with this word and are not
familiar with the official term “acetphenetidin.” They will
doubtless continue to use the term “phenacetin” and we know
of no sufficient reason for doing otherwise. During the life
of the patent the word “phenacetin” became a familiar one,
and the product became generally known by this term. But a
coined name for a patented article loses its proprietary character
and becomes the common name of the article when
the patent expires. In other words, when the patent expires,
not only the product but also the name itself becomes common
property. This principle has been recognized by the
courts. Those who formerly controlled the product and the
name “phenacetin” evidently recognized this principle, for
they have taken no steps to prosecute a firm in this country
which sells the product openly under the name “phenacetin.”
It might be added that the preparation is official in most
foreign pharmacopeias under the name “phenacetin.” In
agreement also with this principle the Council on Pharmacy
and Chemistry (The Journal, April 27, p. 1298) lists in
New and Nonofficial Remedies such products as “lanolin,”
“phenacetin,” “sulphonal” and “trional” as non-proprietary
names applied to Adeps lanæ hydrosus, U. S. P., Acetphenetidinum,
U. S. P., Sulphonmethanum, U. S. P., and Sulphonethylmethanum,
U. S. P., respectively.
In view of these facts—and also bearing in mind the findings
of the Association’s Chemical Laboratory (The Journal,
March 16, p. 801) that the preparations on the market
under the title “acetphenetidin” are of equal quality with the
preparations sold under the name “phenacetin”—the pharmacist
should recognize that acetphenetidin is identical with
phenacetin, is prescribed, provided, of course, that no special
brand of phenacetin is ordered.
It is the physician’s privilege, of course, to specify the
goods of a particular manufacturer, but in view of the fact
brought out above that all brands of this chemical have tested
up to the U. S. P. standard, it is placing an unnecessary
burden on the pharmacist to require him to have on hand
many different brands of one substance. The physician
should save this privilege for use when prescribing some
product that differs materially in its various forms on the
market, as for example in the case of certain fluidextracts.
Physicians will doubtless find that the above comments
will interest their local pharmacists. It is of mutual value
for physicians to talk these matters over with their pharmacists.—(From
The Journal A. M. A., Oct. 5, 1912).
CLEAN ADVERTISING
It is individual effort that counts for most in every movement
for better things—socially, economically or politically.
Realizing this, The Journal repeatedly urges physicians who
write regarding various fraudulent advertisements to enter
their individual, personal protest against the continuation of
such advertisements.
Within the past few months The Journal has had brought
to its attention a good example of what may be accomplished
by personal effort in cleaning up the advertising pages of
a fraternal publication. The Royal Neighbor, official organ
of a fraternal organization, until comparatively recently,
carried numerous fraudulent medical advertisements. Fake
liquor cures, rheumatism cures, tapeworm expellers, tobacco-habit
cures, asthma and hay-fever cures, epilepsy cures, etc.,
disgraced its advertising pages. These called forth protests
from Dr. E. A. Hall, Henry, Ill., who addressed letters to
the official physicians of the fraternal order that the Royal
Neighbor represents, objecting to such advertisements. These
letters in turn reached the advertising manager, and it was
not long before the board of managers took the matter up
for consideration and decided to eliminate this class of advertising
from their official organ. By December, 1913, the Royal
Neighbor came to its readers clean. There is no doubt that
the same results can be duplicated in similar cases. Whether
they are will depend on the amount of active work done by
individuals interested in the question of clean advertising.—(From
The Journal A. M. A., Feb. 14, 1914.)
LIPPINCOTT’S MAGAZINE
Its Advertising—a Protest and an Excuse
A few days ago a physician wrote to The Journal enclosing
two advertisements taken from the current issue of
Lippincott’s magazine. One of these was a half-page advertisement
of that outrageous fraud, the “Oxydonor;” the other
was a full-page advertisement of J. B. Lippincott Company,
Philadelphia, calling for salesmen “to present standard medical
books to physicians only.”
The physician sending in this material asked us to send to
Lippincott’s a pamphlet showing the fraudulence of the
Oxydonor. Instead of doing so, we sent the pamphlet to the
physician and suggested that he write a personal letter in
the belief that individual missionary work is the most effective
way of fighting fraud. Accordingly the doctor wrote to
Lippincott’s and received in reply this letter from the advertising
department of that publication:
“Your letter of the 23d received, and in reply beg to say
that we do not approve of fraudulent advertising, and we
have never before been advised that the advertisement to
which you call our attention was objectionable. In fact, we
know nothing whatever about it. [Italics ours.—Ed.] It
came to us, as do most of the others, through an advertising
agency, and while we do not willingly publish anything that
is fraudulent or objectionable, it is not our custom to verify
the claims of advertisers [Italics, again, ours.—Ed.] especially
when the same copy is being run in almost every other
high-grade publication.
“We have a very high regard for the American Medical
Association, and they undoubtedly are doing a splendid work
in ridding the country of fake medical preparations, but the
mere fact that they condemn some of our advertisers is
hardly sufficient proof for us to refuse the advertising, because,
if the advertiser desires to do so, he can make us prove in
the courts that he is a faker, or claim damages from us for
refusing to publish his advertisement. [Our italics.—Ed.]
If the American Medical Association will guarantee to protect
the publishers against loss from damage suits brought
by advertisers whose business they refuse to accept, then,
we believe that the publishers would gladly reject them, but
not many of the publishers are in a position to investigate
the merits of all the advertising that is offered, especially
when the claims are backed up by affidavits of reputable
people who believe themselves to have been cured by the
preparation.
“We certainly do not wish to jeopardize our medical publications
by advertising fake schemes or propositions of any
kind, and we thank you for writing us concerning the matter
and will now look into this particular case.”

Photographic reproduction (much reduced) of two advertisements
from January, 1914, Lippincott’s. One of a fraudulent quasi-medical
device, the “Oxydonor”; the other, calling for salesmen to introduce
“standard medical books to physicians.”
This letter discloses the workings of the brain of an
advertising man of the old school. The principles enunciated
therein are those that dominated the advertising field until
quite recently. They represent the laissez faire doctrine as
applied to advertising. At that time the only unacceptable
advertising copy was that which would debar the publication
using it from the United States mails. This was the yardstick
by which all advertising was measured at that time.
The economic conscience has since awakened. There are
few reputable magazines today, we venture to believe, that
would be willing to go on record to the effect that it is not
their “custom to verify the claims of advertisers.” The modern,
progressive advertising man recognizes not only the
responsibilities his profession imposes, but also realizes that,
from the narrower view of enlightened self-interest, the
greatest menace to the future of modern advertising is the
fraudulent advertisement.
The claims on the part of the advertising department of
Lippincott’s that it dares not refuse to accept fraudulent
advertisements because the advertiser might bring suit against
it for refusing to accept his advertisement is a statement
whose falsity is exceeded only by its silliness. Equally preposterous
is the statement that Lippincott’s will willingly
refuse to accept fraudulent advertisements provided the
American Medical Association will guarantee to protect the
Lippincott Company against loss from damage suits that may
be brought by the exploiters of the frauds whose advertisements
are refused.
It may be news to Lippincott’s to learn that there are a
score and more of newspapers and magazines that are accepting
the findings of the American Medical Association on medical
frauds and rejecting advertisements of such frauds.
There are many newspapers that send us the medical advertising
“copy” submitted to them and ask for an opinion on
it. When that opinion is unfavorable, these papers refuse
such advertisements. This is being done daily. We have
yet to hear of any “patent medicine” faker or quack even
threatening to bring suit because his advertisements have
been rejected.
The advertising department of Lippincott’s may therefore
take heart. When an advertisement of an outrageous fake
like the Oxydonor is submitted to it, instead of accepting
the money for it, meantime muttering an inaudible protest
at the unfortunate position in which it has been placed, it
may look the fraud in the eye and say Boo! The faker
will not bite.
Before leaving the subject, we are constrained to refer to
Lippincott’s medical publication, the Annals of Surgery. We
begin to realize now why that journal offers a welcome haven
to such products as Sal Hepatica, Bromidia, Papine, Gray’s
Glycerin Tonic, Fellows’ Syrup of Hypophosphites, et al.
Presumably the same “custom” obtains in the acceptance of
advertising for the Annals of Surgery as for Lippincott’s,
namely that the Lippincott Company does “not verify the
claims of advertisers.” Possibly the Annals of Surgery is
afraid that, should it reject the Sal Hepatica advertisement,
for instance, it might he haled into court! Let us trust, for
their peace of mind, that the publishers of the Annals of
Surgery do not receive an advertisement from Old Doc Hartman
for a full page display of Peruna. The mental anguish
they would undergo in reluctantly accepting this advertisement—under
the fear that Hartman would “claim damages”
if it were rejected—is painful to contemplate.—(From The
Journal A. M. A., Feb. 7, 1914.)
MEDICAL JOURNAL ADVERTISING
And Methods of Obtaining Paid-Up Subscribers
Time was when the postal authorities were lenient with
publishers. The names of individuals who had ever subscribed
for publications of a certain class were carried on
the books indefinitely, whether they paid their subscriptions
or not. This permitted a padding of the circulation figures.
Of late years, however, the postoffice department requires
publishers to have bona-fide paid-up subscriptions if they
wish their publications to be carried at the low second-class
rate. Certain medical journals have been hard put to it to
get a circulation that would be at all attractive to the advertisers,
on whose money they depend for continued existence.
Many and various have been the schemes devised whereby
the dwindled circulation might be “boosted.” Subscriptions
could not be given away because the postal laws forbade it.
One ingenious method of obviating this difficulty is worked
in this fashion: Dr. John Doe writes an article that appears
in a reputable medical journal. A few days after its appearance,
Dr. Doe receives a letter from the editor and publisher
of a medical journal that is in need of a subscription list.
He is told that the editor has read his article with much
interest and would appreciate receiving from Dr. Doe a brief
abstract of it. He does not expect the doctor to go to the
trouble of making this abstract for nothing. He will, therefore,
on receipt of the abstract credit Dr. Doe with three
years’ subscription for himself or for one year for himself
and one year for each of any other two doctors he may name.
For every doctor that bites on this scheme the publisher
increases his circulation by three copies and the federal officials
are assured that they are paid-up subscriptions—not
paid for in cash, it is true, but in “abstracts.”
All of this preliminary to a letter recently received:
To the Editor:—Enclosed find letter which speaks for itself.
Now what I should like to know from you is the following:
Is the Charlotte Medical Journal all it should be? Should a
doctor contribute to a journal—thereby adding to its
prestige and circulation—that carries questionable matter in
the advertising pages? If the above journal is off color, does
that act as a bar for good men to contribute?
Very truly yours,
L. J. Genella, M.D., New Orleans, La.
The letter which our correspondent encloses is on the stationery
of the Charlotte Medical Journal and signed by the
editor of that journal. Here it is:
“My Dear Doctor Genella:—I have just looked over an article
of yours published in the New Orleans Medical and Surgical Journal
entitled ‘Clinical Studies in Pituitary Irritation, with Report of Case.’
I would be very glad indeed to have you send me a manuscript or article
for the Charlotte Medical Journal. Your style of writing is very
attractive.
“If you will send me an article for the journal, I will be glad to
publish same and will place your name on my complimentary mailing
list. Under separate cover I am sending you a copy of the journal.
“Of course I will expect the article to be typewritten.”
Whether or not this is a modification of the “abstract”
scheme or an attempt to boost the circulation of the Charlotte
Medical Journal are questions we shall not attempt to
answer. As to the questions propounded by our correspondent,
they have been answered many times in these pages.
We turn to one of the recent copies of the Charlotte Medical
Journal and examine its advertising pages. On one of the
first we find Anasarcin, a product whose fraudulent character
was described at some length in The Journal, May 4
and 11, 1907. On another page we find Tongaline, which
has also come in for a fair share of attention (see The
Journal, Sept. 23, 1906, and May 10, 1913). A little farther
over we find a half-page advertisement of Bannerman’s
Intravenous Solution, a nostrum first exploited as a “consumption
cure” and now as a cure-all (see The Journal,
May 31, 1913). Cactina Pillets (see The Journal, March 12,
1910), Hagee’s Cordial of the Extract of Cod-Liver Oil (see
The Journal, Oct. 13, 1906), Burnham’s Soluble Iodin (see
The Journal, March 28, 1908), Ecthol (see The Journal,
March 13, 1909), Bromidia (see The Journal, April 21,
1906), Papine (see The Journal, April 29, 1911), Phenalgine—two
advertisements (see The Journal, Jan. 13,
and 27, 1906, and Jan. 27, 1912) and Sal Hepatica (see The
Journal, March 26, 1910) are some more products which
have attained unenviable notoriety but found a safe haven in
the advertising pages of the Charlotte Medical Journal.
Neither must we fail to refer to the advertisement of Duffy’s
Malt Whiskey (see The Journal, Nov. 23, 1912), which
looks thoroughly at home.
Does our correspondent—in fact, does any conscientious
physician having the interest of scientific medicine at heart—want
to do anything that will tend to perpetuate therapeutic
fraud? Subscribing for or contributing to medical
journals whose income is largely derived from nostrums that
are as vicious as many of the “patent medicines” advertised
in the daily press hampers the medical profession in its fight
for honesty in therapeutics and renders largely abortive its
fight against fraudulent “patent medicines.” So long as the
accredited organs of the medical profession tolerate fraudulent
“ethical proprietaries” in their advertising pages, just
so long will the protests of physicians against the swindling
advertisements of “patent medicines” in the daily press fall
largely on deaf ears—and justly so.—(From The Journal
A. M. A., Oct. 11, 1913.)
A Physician Places the Responsibility for Fraudulent
Advertising Where It Belongs
“To the Editor:—The Journal has had much to say in
recent years regarding the ethics, or lack of same, in advertising
matter exploited by its contemporaries. It has been
criticized by many for the stringency of its attack; it has
been criticized by very few because it did not go far enough.
Is it not about time to get to the root of the matter?
“In the last number [see p. 426, this book] dissatisfaction
is expressed with the advertising policy of the Medical Times.
Nothing finer! Go to it! But is the method of attack right?
I have before me a sample copy of the American Journal of
Surgery. Among other articles is one on diseases of joints
and the bone marrow by a man very favorably known in
Denver. He was ‘ethical’ enough to be accorded a place on
the program in the Section on Medicine at Minneapolis.
Another contributor from Baltimore remarks that he took a
patient to the University Hospital. Can it be possible that
Johns Hopkins is admitting men to its wards and clinics that
are below par in professional morals? Another article
appears from a well-known orthopedic man of Washington,
D. C. Personally, I see very little to commend in the advertising
columns of the American Journal of Surgery.
“I, who confess to a state bordering on youth, may be
very wrong; but I believe that the trouble will be solved only
when men who claim to have any professional distinction
refuse to contribute to journals whose pages are not clean
from cover to cover. Pardon the presumption, Mr. Editor,
but were you ever tempted to print anything like this:
“‘Last week’s issue of the New York Medical Squall contains
an article on “Duodenal Ulcer” by John Doe, the well-known
Chicago surgeon. Dr. Doe doubtless knows as well as
any one the disreputable character of the Squall’s advertising
matter, but like most of our great men, is unable to restrain
his appetite for journalistic publicity.’
“Physicians read medical journals because they contain
literature that is worth while. Jump on your erring editorial
brethren, Mr. Editor, but please remember that the problem
of eliminating bogus advertisements will be solved when the
so-called leaders of our profession show enough manhood to
refuse literary support to publications whose columns are in
disrepute. While castigating the little sinner, please don’t
let the big sinner go scot free.
“Clinton E. Harris, M.D., Grinnell, Iowa.”
Dr. Harris sums up the situation correctly. No small
degree of responsibility rests on the prominent members of
the medical profession who lend their support either as subscribers
for or contributors to those medical journals whose
advertising pages are a stench in the nostrils of thinking
physicians. Dr. Harris asks why The Journal does not condemn
the advertising columns of the American Journal of
Surgery. The Journal has done so more than once and in no
uncertain terms, both in the Propaganda department and editorially.
At one time it said:
“In circular letters and in an editorial announcement in its
December issue, the American Journal of Surgery ‘features’—to
use a newspaper term—some of the contributors to its
January issue. The list comprises men who hold, or have
held, high offices in the American Medical Association. Presidents,
vice-presidents, chairmen, secretaries and members of
sections of the Association—these are some of the men whose
names appear as contributors to this nostrum-promoting publication.
Is it any wonder that the proprietors of the American
Journal of Surgery assume an attitude of indifference to
the class of proprietary preparations which they admit to the
pages of their publication?”
What was the result of The Journal thus directing the
attention of its readers to the American Journal of Surgery?
In the next issue of the American Journal of Surgery
appeared a seven-column editorial tirade, entitled “An Unwarranted
Attack on the President and Other Eminent Members
of the American Medical Association and on the Leading
Medical Journals of the Country.”
On many and various occasions has The Journal called
attention to the very evils that Dr. Harris deplores, and for
the benefit of those who care to look up the matter these
references to some of the articles are appended:
“The Mote and the Beam,” editorial, Nov. 18, 1911.
“Activity or Passivity—Sympathy or Sacrifice,” editorial, Dec. 9, 1911.
“Cui Bono,” editorial, Dec. 16, 1911.
“Medical Journals and the Great American Fraud,” Propaganda
Department, Dec. 16, 1911.
“The Profession Must Apply the Penalty,” editorial, Jan. 13, 1912.
“Fraudulent Advertising in High-Class Medical Journals,” editorial,
Jan. 4, 1913.
“Demand Clean Advertising,” editorial, Jan. 4, 1913.
“Medical Journals and the Great American Fraud,” editorial, Jan.
18, 1913.
“A Good Principle to Apply,” editorial, May 13, 1913.
“Medical Journal Advertising,” Propaganda Department, Oct. 11,
1913.
“Medical Journals and the Great American Fraud,” Propaganda
Department, Oct. 18, 1913.
“Medical Journals and the Great American Fraud,” Propaganda
Department, Nov. 1, 1913.
“The Medical Times’ Advertisements,” Propaganda Department, Nov.
8, 1913.
In another letter on the same subject its writer says: “I
think the time has arrived when we have a right to expect
real leadership from the ‘big men’ of the profession.”—(From
The Journal A. M. A., Nov. 22, 1913.)
MEDICAL JOURNALS AND THE GREAT AMERICAN
FRAUD
How the Medical Times Aids and Abets Quackery, with the
Moral Support of Members of the Medical Profession
Two letters have been received, both from physicians. One
comes from New York City and the other from Alexandria,
Va. Each letter contained an advertisement of the Kellam
Hospital, Richmond, Va., cut from the Medical Times. Here
is the New York letter:
“To the Editor:—I am enclosing an advertisement clipped from
the Medical Times. It seemed to me an especially flagrant example
of what may happen in the absence of proper supervision of the
advertising pages of a medical magazine. The condition would seem
all the worse in this instance as among the ‘Board of Contributing
Editors’ are listed men like Howard Lilienthal of New York and
Almuth C. Vandiver, who is Counsel for the Medical Society of the
County of New York. The Medical Times is sent to two of the
physicians who live at this address without charge and without
solicitation. Many advertisements of proprietary preparations are
inserted in type indistinguishable from that of the body of the magazine
and it is of course possible that its financial backing comes entirely
from the manufacturers of these drugs.”

Fig. 1.—Photographic reproductions from the Medical Times. Do
the gentlemen whose names appear in the list of the “Board of Contributing
Editors” realize that they are lending an air of respectability
to an otherwise disreputable business?
And this is from Virginia:
“To the Editor:—The statements made in the advertisement of
the Kellam Hospital in the October number of the Medical Times are
so out of the ordinary that I ask you to tell us something if you can
of their institution and its methods of cure. Can such things as are
stated in this advertisement be true? ‘Physicians Treated Free?’
‘Endorsed by the Senate and Legislature of Virginia?’ What can all
this mean to the sufferer from cancer? If true, let the whole world
of sufferers know the glorious news.”
Collier’s paid its respects to the Kellam concern some time
ago and we cannot do better than quote from its pages.
Thus:

Fig. 2.—One way of drumming up trade in the “cancer cure” business!
The Kellam Hospital sends letters like this to the postmasters of
small towns asking these government officials to furnish it with what,
in the parlance of quackery, is known as a “sucker list.” A delightful
business, isn’t it? And this, gentlemen of the “Board of Contributing
Editors,” is the sort of thing to which you are lending your influence
and good names!
“Grief is the portion of the Kellam Cancer Hospital, of
Richmond, Virginia, because in these editorials it has been
grouped with other exemplars of the Great American Fraud.
It offers the invariable and hollow mockery of testimonials
and endorsements, which, as has been repeatedly shown, can
be wheedled, browbeaten or bribed out of the victims of any
form of quackery. It, of course, courts the fullest investigation,
and desires that we send a representative to investigate
whether its claims are not well founded. Unsuspected by the
Messrs. Kellam, our representative has already investigated
their claims, notably their statement that they are endorsed
by the Legislature of the State of Virginia. Upon request
for a copy of the endorsement they forwarded a weak subterfuge,
and finally, on pressure, admitted that they could not
produce the proof they had boasted. For their further consideration
we present a brief parallel:
| FROM THE KELLAM CIRCULAR |
|
FROM A KELLAM LETTER |
|
The Cancer is removed without the use of the knife or X-Ray ... No
roots or fibers left; hence it can not return. |
|
We do not claim to “cure them all.” We go further, and on
our part, we agree to treat, free of charge, any patient who
suffers a recurrence after having been treated by our method. |
“The italics are our own, but we cheerfully present them
for elucidation to the Kellam Hospital. A little careful
thought devoted to reconciling the irreconcilable may help
them to forget their woe. Meanwhile, they make themselves
out worse than they really are by pretending to withhold from
the bitter need of humanity a true, non-surgical cure for
cancer. If this were true; if, indeed, they had solved the
problem which has baffled the greatest minds of modern
science; if, having a genuine cure for the dreadful ailment
which claims its increasing thousands of tortured victims
yearly, they secrete their discovery for the sake of a few
paltry dollars, then they are as cold-hearted as the sailors
who pass within fair hail of the naked island on which some
shipwrecked crew is starving, and keep their stony eyes on
the compass. They have not even the excuse of the fanatical
among the Christian Scientists who, denying the existence of
pain, refuse to take measures to ease the cancer victim’s
suffering even at the last. Human nature is seldom so callous.”
As for the Medical Times: This publication for years contained
comparatively little advertising. Then it came into
the hands of Romaine Pierson, who also owns the Practical
Druggist. Mr. Pierson is not a physician and to him the medical
profession is but a commercial problem. He is publishing
a medical journal for the money there is in it, and for this he
is not to be censured. Questions of advertising policy, in such
circumstances, are determined on a commercial basis. When
an advertising contract is submitted, for a product that physicians
would know to be fraudulent, the question that arises is,
“Can it be put over?” Manifestly, a medical journal published
purely as a business venture would not dare long to fly
in the face of the opinions of those from whom it received its
support—its subscribers and contributors. If our correspondents
will go through the advertising columns of the Medical
Times they will find many, many other frauds, less cruel perhaps
than the Kellam advertisement, but no less disreputable
or discreditable to the medical profession.
After all is said and done, it is enlightened public opinion
that is causing publishers of lay magazines and newspapers
to eliminate fraudulent “patent medicine” and quack advertisements.
Until the medical profession takes an equally
enlightened stand, physicians may expect to be afflicted with
such commercial medical journals as the Medical Times, the
International Journal of Surgery, the American Journal of
Surgery, American Medicine, and several other papers that
are published primarily in the interest of the advertiser.
When such journals as these find they cannot get a circulation
among physicians so long as they carry advertisements
similar to many now appearing in their pages, these advertisements
will be eliminated, but not before. Many physicians
are receiving such journals at a nominal price or, as one
of our correspondents notes, free. The physician who permits
such journals to come to his office must share with the
paid subscribers the responsibility for the low standard of
medical journalism.—(From the Journal A. M. A., Oct. 18,
1913.)
Two Physicians Express Themselves on the Ethics of
Medical Journalism
After the preceding article was in type, we received, in
the same mail, two letters that are so apropos that we reproduce
them. The first was from a town in Illinois, and was
dated October 11. Here it is:
To the Editor:—About two weeks ago, a representative of
the Surgery Publishing Company, New York, N. Y., came to
—— Ill. soliciting subscriptions for the American Journal
of Surgery. Together with numerous others I subscribed—chiefly
on the strength of the contributors whose articles
appeared in the sample numbers shown by the agent.
Since receiving the first number (October) one look at the
advertising pages has shown me why the subscription price
for a year and a quarter is one dollar. Anasarcin, Tongaline,
Cactina Pillets, Hagee’s Cordial of Ext. Cod Liver, Burnham’s
Soluble Iodin, Papine, Phenalgine, Anusol, etc., etc.
I have written to the Surgery Publishing Company, telling
them in no uncertain language that there is no room on my
reading desk for such. Have you ever exposed this journal,
and the attitude of our big, brilliant, eminent men in permitting
their articles—presumably original—to fill space in
such a journal? [Yes! The Journal, Dec. 16, 1911, pp. 2,000
and 2,013.] This letter is not for publication—at least not
with name of city. Keep up the good work....
The other, dated October 10, follows:
To the Editor:—That little story about medical journal
advertising and methods of obtaining paid-up subscribers,
in this week’s Journal makes me blush (p. 422, this book).
I am guilty. Unlike Dr. Genella, I swallowed the bait—but the
bait was even more tempting in my case; the flattering “editor”
offered me twenty-five subscriptions to distribute among my
friends, all for an “abstract.” Thank goodness, I only accepted
five subscriptions, but worse luck, I sent them to young men
by preference. So I am a deep-dyed offender indeed. Extenuating
circumstances affected my susceptibility somewhat, however.
I have noticed that prominent men like Beverly Robinson,
A. Rose, Tom A. Williams, Wayne Babcock, and Morris—the latter,
at least, a really able man and a brilliant writer—contribute
to these peanut journals occasionally. If they
do, why not I? There’s nothing like being in big company,
you know.
So far as I know, my “abstract” has not yet been published.
On looking over the sample copy of this monthly I found an
advertisement printed right in the list of contents—in fact,
it was the second “original article” in the issue, as brave and
respectable as you please! Then, with characteristic Hibernian
impetuosity I got out my machine and pounded that
editor a strong protest with a dire command not to use my
“abstract” in his miserable organ. But I have never received
the manuscript, nor any reply to my stern rebuke. I wish
I had been cautious like Dr. Genella.
Wm. Brady, M.D., Elmira, N. Y.
—(From The Journal A. M. A., Oct. 18, 1913.)
The Responsibility of Physicians
The responsibility of medical journals for the continued
existence of at least a part of the “great American fraud,”
has been referred to in these pages many times. Within the
past few weeks The Journal has called attention to the
inconsistency of reputable physicians of high ideals lending
their moral, and often financial, support to those medical
journals whose advertising pages are a disgrace to the profession.
Specifically, the Medical Times—originally a homeopathic
medical journal—has been referred to, among others,
as an example of this type of journalism. It must, however,
be regarded simply as a type, for it is no better and no
worse than many other medical journals. Several letters
have been received on the subject, some of which we reproduce.
The first one is from Dr. George G. Ross of
Philadelphia:
“I was very much jarred on receiving the last issue of The
Journal to find under the Propaganda for Reform an article
concerning the Medical Times, among the list of whose contributing
editors my name appears. I enclose you herewith
a copy of my letter of resignation to the Medical Times. I
have a very dim recollection of what occurred at the time
that I was asked to give my name as a contributing editor.
As I recollect it, however, at that time the journal was a
respectable and ethical publication. I had been asked by a
friend of mine to write an article giving my opinion of the
effects of college athletics on undergraduates. This was at
the time that Dr. Stokes had issued his order about athletics
at Annapolis. I want personally to thank you and the committee
for the exposure of this journal and for having drawn
my attention to the fact that I was unwittingly aiding and
abetting such a journal. I trust that if you have space in
some future number of The Journal, you will do me the
justice to publish all or part of this letter.”
Because he feels that he has “unwittingly been put in an
unfavorable light,” Dr. James A. Babbitt, also of Philadelphia,
sends The Journal a copy of a letter written by him to the
editor of the Medical Times. Here it is:
“For reasons of which you are probably cognizant, I deem
it advisable to resign from the board of contributing editors
of the Medical Times, and desire that this resignation be
accepted at once and my name not appear in further issues.”
What shall be done, asks Dr. Sidney Thompson of Humboldt,
Tennessee, in such cases as the following? Says Dr.
Thompson:
“In the Propaganda for Reform, in The Journal, October
18, 1913, in closing your article on ‘Medical Journals and the
Great American Fraud,’ you say: ‘The physician who permits
such journals to come to his office must share with the
paid subscribers the responsibility for the low standard of
medical journalism.’ Now I agree with you in everything
you have said about the Medical Times, but what I want to
know is how to keep such journals from coming into your
office. The Medical Times has been coming to me for a number
of years with repeated duns for the subscription price. I
have written to them several times that I did not want the
journal and never expected to pay for it, but still it comes.
I have a vague recollection that I bit at an offer to send it
three or four months free, not knowing what it was, but I
never authorized them to enter my name as a regular subscriber.”
The simplest course in such a case as that described by Dr.
Thompson, is to write on the unwelcome publication the word
“refused” and either drop it in the nearest mail-box or hand
it back to the postman. The courts have held that a person
who continues to accept publications is legally liable for the
payment of such publications. The postoffice department,
however, has ruled that a magazine—either monthly or weekly—may
not be sent at second-class rates for more than one
year after the expiration of a bona fide subscription. At the
expiration of that time, stamps must be affixed and the
publications sent at third-class postal rates.—(From The
Journal A. M. A., Nov. 1, 1913.)
Medical Journals and Sanatogen[AR]
We have frequently referred to the inquiries that are
received by this office from newspaper and magazine editors
asking for information about products whose advertisements
they have been offered. One of the greatest difficulties in the
way of accomplishing the good that such inquiries otherwise
might lead to is the lack of uniform action on the part of the
medical press of the country. A specific instance may be
given. A layman wrote to a high-glass{sic} weekly magazine published
in New York City protesting against an advertisement
of Sanatogen which the magazine was carrying, and sending
a reprint of The Journal’s article on this product. The
advertising manager of the magazine in question wrote back
that he had seen The Journal’s article, but had sought
further information regarding the preparation from the editor
of a medical journal in his city. The medical editor recommended
that the magazine accept the Sanatogen advertisement,
so the advertising manager said, and in view of this,
the manager suggested that possibly the article published by
the American Medical Association in its journal was inspired
by some “personal prejudice.” Giving weight to the probability
that the advertising manager went for his information
to a source that he knew would be favorable to the acceptance
of the advertisement, the fact remains that it is a disgraceful
state of affairs when editors of medical journals will
give vicious advice in matters on which they are supposedly
competent to pass. The probability is, of course, that the
medical journal whose editor was questioned contained the
self-same advertisement that the lay magazine was carrying.
And the advertising manager of the magazine was willing to
accept—because such information coincided with his wishes—information
that on its face must be biased, and rejected
advice—that did not meet his approval—because of a purely
supposititious “personal prejudice.” It is probably asking too
much to expect advertising managers not to go to sources
that are likely to be favorable for information about products
whose advertisements are offered to them. But we have a
right to expect that physicians, editors of medical journals,
should no longer be participes criminis in the furtherance of
the great American fraud. If our strictures on Sanatogen are
unfair, if the Council on Pharmacy and Chemistry rejected the
product in mere pique, if the opinions of such men as Billings,
Cabot, Hektoen and Lusk are to be brushed aside as “personal
prejudice,” if this mixture of cottage cheese and glycerophosphates
really is the marvelous product which its exploiters
claim—then indeed not only have the editors of medical
journals a right to praise it, but it is also their duty to
proclaim these wonders in their editorial pages. If, on the
other hand, this much-vaunted preparation is a very ordinary
mixture sold at an extraordinary price, if indigent consumptives
and others are being inveigled into spending dollars for
a preparation whose food value could be duplicated for a
few cents—then in the name of humanity and common
decency let the editors of medical journals proclaim these
facts, and not let their scientific judgment be blinded by the
glitter of advertising contracts.—(Modified from The Journal
A. M. A., Jan. 18, 1913.)
THE ARMY AND NAVY MEDICAL RECORD
A Fraudulent Publication Whose Editorial Opinions
Are for Sale
Whenever a business assumes certain proportions, subsidiary
businesses spring up to cater to the needs of the larger
enterprise. For some years the nostrum business has grown
so large that it has furnished a more or less precarious life
for many individuals who have catered to it. There are, for
instance, men whose trade it is to obtain testimonials; others,
claiming a long string of imposing degrees, will furnish fake
reports and bogus analyses; still others issue at irregular
intervals publications with high-sounding names which sell
editorial indorsement to the products of concerns such as are
willing to pay the price asked. “Journals” of this type have
been called to the attention of our readers at different times;
the New York Health Journal and the United States Health
Reports come to mind at this moment. Both of these had
their day and died a natural death, as all such publications
must when once the public is cognizant of their true character.
TWO LETTERS
More recently the attention of The Journal has been called
to a publication calling itself the Army and Navy Medical
Record. A physician in the South sends a letter he has
received from the Army and Navy Medical Record reading as
follows:
“We have had many favorable reports reach us relative to your
most excellent institution, and, as you are doubtless aware, we come
in direct contact with a large number of Army and Navy and other
government attachés who have sons that they desire to provide with
a medical education combined with the higher course included in your
up-to-date laboratory methods and the sciences incidental to clinical
medical practice.
“If you will regard the proposition as confidential, we will agree
to carry a one-fourth page advertisement of your university at the
nominal rate of $38 per year, provided this amount is forwarded in
advance at the time copy is furnished; and we will further promise
to editorially indorse and recommend your school and its methods
without qualification or exception. [Our italics.—Ed.] This article
you should be able to use (and are authorized to do so) after publication
for advertising purposes.
“We will also be able, and are willing, to furnish you with a desirable
list of probable candidates from time to time.
“Kindly let us hear from you at once, if interested, and oblige,
“Yours with best wishes,
“The Army and Navy Medical Record,
“Arthur G. Lewis, Managing Editor.”
The physician to whom this was addressed made a notation
on the letter to the effect that “this looks crooked.” A few
weeks later, Dr. V. C. Vaughan, dean of the University of
Michigan, Department of Medicine and Surgery, sent in a
letter from the Army and Navy Medical Record which he
had received in his official capacity at the university. Here
is the letter; again the italics are ours:
“We are gratified to advise you that in our efforts to select a
strictly ethical and high-grade institution of medicine that this magazine
could consistently indorse and recommend, we have decided on
the University of Michigan, Department of Medicine and Surgery, as
the institution in your territory to whom our special publicity concession
will be made this year.
“You are doubtless aware that we come in direct contact with a
very large number of Army and Navy and other government attachés,
also physicians in private practice who have sons that they desire to
provide with a medical education, combined with the higher courses
included in your up-to-date methods.
“For personal reasons we are particularly anxious to favor your
institution, and frankly believe that we can prove of material service
to you. The special proposition, to be regarded by you as strictly confidential,
is that we will publish a full one-half page announcement of
your institution for the term of one year, you to merely pay a nominal
expense charge of $38 for the year’s service. As our regular rate is
$125 per annum for this service, the necessity of regarding the matter
between ourselves is apparent. [Transparently so.—Ed.] We further
propose, without expense to you, to editorially indorse and recommend
your institution and its methods without qualification or exception. An
electrotype illustration may be used, without charge.
“It is important, however, that we hear from you promptly. Awaiting
your immediate reply, we are, with best wishes,
“Yours faithfully,
“The Army and Navy Medical Record,
“Arthur G. Lewis, Managing Editor.”
Dr. Vaughan, in forwarding the matter to The Journal,
wrote that on receipt of the offer just given, he “was uncertain
whether its writer was a knave or a fool.” After inquiring
into the matter somewhat thoroughly, he concluded that
“the managing editor of the Army and Navy Medical Record
is both a knave and a fool.”
THE ARMY AND NAVY MAGAZINE
The Journal had the Army and Navy Medical Record
under investigation before these two letters were received
and, as a result, the following facts seem to be pretty well
substantiated. Herbert C. Lewis, with his brother, Arthur
G., conducted from Washington, D. C., a publication called
the Army and Navy Magazine. In The Journal’s nostrum
file there is a booklet put out by the Renova Distributing
Company describing the wonderful virtues of its product,
“Anti-Jag,” which, as its name might intimate, is a “liquor
cure” of the fake variety. One page of this booklet is given
over to what purports to be “A Letter from a Great Magazine
Editor.” The letter is dated June 19, 1900, from Washington,
D. C., and says that “the editor of the Army and
Navy Magazine takes pleasure in stating that from his own
personal knowledge he has found ‘Anti-Jag’ to be one of the
most reliable medicines ever introduced for the permanent
cure of drunkenness.” And more to the same effect. The
letter is signed “Herbert C. Lewis, editor.”
The publishing offices of the Army and Navy Magazine
are at 606 F Street, N. W., Washington, D. C. The building
at this address is known as the Baltic Building. Herbert
C. Lewis is said to be a printer by trade.
The Army and Navy Medical Record seems to have been
started within the last few months by Arthur G. Lewis. It
does business from two addresses, the Baltic Building, Washington,
D. C., and the Maple Villa Sanitarium, Hammonton,
N. J. Lewis is said to have purchased the Maple Villa Sanitarium
recently, but apparently his chief source of income
is the Army and Navy Medical Record. He is alleged to
have claimed that some medical officials of the government
are interested with him in this publication but that these
officials do not wish their names known. We do not blame
them.

Photographic facsimile of a letter sent by the Army and Navy
Medical Record to the dean of University of Michigan, Department of
Medicine and Surgery, offering one hundred and twenty-five dollars’
worth of advertising space for a “nominal” thirty-eight dollars—with
editorial indorsements and recommendations thrown in for good measure!
ADVERTISEMENTS AS EDITORIALS
A glance through two issues of the Army and Navy Medical
Record makes perfectly plain the character of the publication.
The January-February, 1913, number leads off with
articles by well-known medical officers in the Army, the
Navy and the Public Health Service. These have been copied
from other publications. Then comes an editorial entitled
“A Much Needed Dietary Reform,” devoted to the laudation
of “Postum,” the widely advertised coffee substitute. Following
this is an editorial on “The Philosophy of Hypnotics”
in which aconitine, saline laxative and digitalin are each
given a “boost.” Then comes an “original article” (save the
mark!) entitled “The Physiological Pathology of Consumption.”
This is by “Alfred S. Gubb, M.D., L.R.C.P., London,
M.R.C.S., Eng., D.P.H., etc. etc., Aix-les-Bains, Savoie,
France.” Two pages are devoted to this. The “joker”
appears in the third paragraphs from the end—Fellows’ Syrup
of Hypophosphites. Dioxogen receives more than three pages
of editorial mention under the caption “The Sterilization of
Milk with Dioxogen.” Under “Another New Electrical Wonder—Magnified
Sound,” the “Acousticon” is given a two-and-a-quarter
page write-up. “What Wise Men Wear” is the
title of a four-page article—unsigned—devoted to the laudation
of suspensories in general and the “O-P-C Suspensory”
in particular. Dr. H. F. Boatman, Los Angeles, contributes
a short article on “A Case of Advanced Pulmonic Tuberculosis
Treated with Injections of Dioradin,” while our good
old friend Willard H. Morse, M.D., “F.S.Sc. (Lond.),” the
champion fake-testimonial-giver of the country, writes more
or less entertainingly on “Putting on a Mustard Plaster.”
The article has nothing to do with mustard plasters but has
a good deal to do with “Zumota,” a nostrum recommended as
a substitute for the mustard plaster. These are but a few
of the nostrums to which the editorial and reading pages of
the Army and Navy Medical Record are devoted.
In the June-July issue, Arthur G. Lewis becomes bolder.
The leading article is entitled “First Aid in the Navy,” by
C. F. Stokes, Surgeon-General, United States Navy. There is
nothing to indicate that this article was not contributed to
the Army and Navy Medical Record by its author. As a
matter of fact, it originally appeared in an official publication,
the United States Naval Medical Bulletin for January, 1913,
and was reprinted by Lewis without credit and without permission.
Following the article by Dr. Stokes is another,
unsigned, entitled “The Passing of ‘The Pie Habit.’” This
describes the surprise of the students of Harvard University
at being served breakfast cereals instead of pie at their noonday
meal and suggests that “Shredded Wheat Biscuits” make
a “delicious dessert.” A two-and-a-half page article on the
“Danger of Corrosive Sublimate in Vaginal Douche” is
reprinted from the Lancet-Clinic of September, 1903. The
reason for resurrecting this ten-year-old article becomes
apparent before one gets half through it. It deals not so
much with the danger of corrosive sublimate as with the
marvelous—alleged—properties of Tyree’s Antiseptic Powder.
Dr. Claude C. Keeler, Denver, has a three-page article on the
“Medical Treatment of Pulmonary Tuberculosis.” The “medical
treatment” referred to is Waterbury’s Compound. An
editorial entitled “One Notch Ahead of Morphin” is devoted
to that vicious morphin solution sold under the proprietary
name “Papine.” Another on “The Treatment of Catarrh by
Palliatives and Curatives” deals with a widely advertised
“patent medicine,” “Kondon’s Catarrhal Jelly.” What appears
to be a contributed article by Charles Wardell Stiles of the
United States Public Health Service on “Country Schools
and Rural Sanitation” has really been “lifted” from an official
publication without credit and, needless to say, without Dr.
Stiles’ permission.
But medicinal preparations are not the only things to which
the Army and Navy Medical Record gives editorial indorsement.
All advertising matter, apparently, is grist to its mill.
Sandwiched in between articles on “Public Health Administrations”
and “Important Army Medical Lectures” is a dissertation
on “The Millennium of Shirt Construction,” in
which are sung the virtues of the tailless shirt! A little farther
along the Hawaiian pineapple is extolled, while the last
pages of the issue are devoted to various banking concerns.
In addition to the advertisements appearing throughout the
reading and editorial pages of these two issues of the Army
and Navy Medical Record, there are a number of display
advertisements. There is no reason to suppose, at least in
the majority of cases, that the advertisers had the slightest
reason to suspect the nature of the Army and Navy Medical
Record. Several pages are devoted to financial advertisements,
there being more than forty banks that have “fallen
for” the wiles of Arthur G. Lewis. In view of the letters
received by the deans of medical colleges and other educational
institutions, the display advertisements of schools and
colleges have a special interest to physicians. Schools for
girls, polytechnics, colleges of music, veterinary, dental and
medical schools—all are to be found in this cosmopolitan
publication.
Among the therapeutic products advertised—in the advertising
pages—are:
Fellows’ Syrup of Hypophosphites | 1 | cover page |
Kondon’s Catarrhal Jelly | 1⁄2 | page |
Expurgo Anti-Diabetes | 1⁄2 | page |
Laxol | 1⁄2 | page |
Campho-Phénique | 1⁄2 | page |
Palpebrine | 1⁄2 | page |
Zumota | 1⁄2 | page |
Sanmetto | 1⁄4 | page |
While in the reading pages the following products are
puffed:
| Tyree’s Antiseptic Powder. | Dioxogen. |
| Waterbury’s Compound. | Palpebrine. |
| Papine. | Bannerman’s Intravenous Solution. |
| Kondon’s Catarrhal Jelly. | Daniel’s Concentrated Tincture of Passiflora. |
| Ranier Natural Soap. | Peacock’s Bromides. |
| Iodia (Battle). | Aletris Cordial Rio. |
| Creo-Derma. | Gonosan. |
| Fellows’ Syrup of Hypophosphites. | Digipuratum. |
| Tannalbin. | Dioradin. |
| Expurgo Anti-Diabetes. | Pepto-Fer. |
| Zumota. | Lactol. |
| Sulfothen. | Campho-Phénique. |
Summed up: The Army and Navy Medical Record is but
another of the parasites of quackery. It is not entered as
second-class matter and it has probably no bona-fide circulation.
While it is claimed to be “Devoted to the Interest of
the Medical and Surgical Corps of the Army and Navy, the
Public Health Marine Hospital Service and the Red Cross
Society” it is actually devoted to none of these. It is devoted
to the exploitation of the advertising public for the special
financial benefit of the man who calls himself its editor—Arthur
G. Lewis. Advertising contracts are obtained under
false and fraudulent pretenses. In brief, Arthur G. Lewis is
using the good name of the various medical services of the
United States government to further his swindling operations.
He has written letters to honorable physicians making
dishonest and insulting propositions to deceive and defraud
the public. Editorial indorsements of the Army and Navy
Medical Record mean nothing except that money has been
paid for them. In short, the Army and Navy Medical Record
is a fraud, and its “editor,” Arthur G. Lewis, a faker.—(From
The Journal A. M. A., Oct. 25, 1913.)
THE MEDICAL TIMES ADVERTISEMENTS
In Which Are Discussed Some “Oversights” and the Ethics
of Journalism
Two or three weeks ago we published letters from two
physicians calling attention to an advertisement of a “cancer
cure” hospital appearing in the Medical Times. As a result
of The Journal’s comments, some of the physicians who
were listed as “contributing editors” of the Medical Times
wrote that they had requested that their names be withdrawn
from this list. In reply to at least some of these letters, the
editor of the Medical Times wrote asking them to reconsider
their decision and offering as an excuse the statement that
the appearance of the “cancer cure” hospital advertisement
was an oversight. In this connection the following letter
from the Medical Times, addressed to The Journal of the
American Medical Association, is pertinent:
“Gentlemen:—We note in your issue of October 18 an article calling
attention to an advertisement which appears in the columns of
this journal, and to which your editor rightly objects.
“The advertisement in the Times was the result of an oversight,
and it will not reappear.
“While we are indebted to you for thus bringing the matter to
our attention, we cannot but feel that a letter written to us would have
been more in keeping with the ethics of journalism.
“Very truly yours,The Medical Times.”
It will be noticed that this letter, like the letters sent to
other physicians, ignores altogether the most important point
made by The Journal in its criticism of that publication’s
advertising policy. The Journal said in this connection:
“If our correspondents will go through the advertising columns
of the Medical Times they will find many, many other
frauds, less cruel perhaps than the Kellam advertisement, but
no less disreputable or discreditable to the medical profession.”
The Medical Times apologizes for the advertisement of the
Kellam Cancer Cure Hospital but ignores altogether the fact
that the hospital advertisement was but one of many equally
discreditable. We turn to recent issues of the Medical Times
and we find it fairly reeking with advertisements of proprietary
preparations that are a disgrace to the medical profession,
many of them having been repeatedly exposed in
The Journal. We find, for instance, a quarter-page advertisement
of the Expurgo Manufacturing Company. “Expurgo
Anti-Diabetes,” we are solemnly told in the pages of the
Medical Times is:
“The only reliable and thoroughly tested remedy for the cure of
Diabetes Mellitus and Insipidus.”
This wretched fraud, which also is advertised in true
“patent medicine” style direct to the public, is presented to
a presumably intelligent profession as a “cure” for a disease
which so far has baffled the best brains in the scientific world.
“Expurgo Lapis” we are told, also via the Medical Times is:
“The only known cure for gall-stones, kidney and bladder stones,
gravel and all kidney trouble arising from uric-acid origin.”
Did Kilmer’s Swamp-Root ever claim more? “Diabetes is
no longer an incurable disease” runs the advertisement of the
Jireh Diabetic Food Company, yet the editor of the Medical
Times must know that in The Journal133 and in the reports
of state chemists the Jireh diabetic foods have been shown
time and again to be among the most dangerous and fraudulently
exploited products sold to the unfortunate diabetic.
Phenalgin,134 twin brother to the Antikamnia fraud, shouts
its inferential falsehoods in a half-page display. Micajah’s
Wafers,135 the alum-borax mixture long advertised as a cure
for gonorrhea, endometritis, etc., may also be found, as well
as many other preparations exposed at various times by The
Journal. For example: Anasarcin,136 Campho-Phenique,137
Papine,138 Bromidia,139 Cactina Pillets,140 Pluto Water,141 Prunoids,142
Sanatogen143 and Sal Hepatica.144
What excuse can the Medical Times offer for the presence
of these frauds in its pages? Are these, too, “the result of
an oversight”? Presumably for thus bringing to its attention
these various other disreputable advertisements, The Journal
will be accused again of violating “the ethics of journalism.”
If calling attention to fraudulent advertisements is
out of keeping with the ethics of journalism, what, pray, must
be said of publications that are willing to share in the profits
of such fraudulent exploitation? But, and we cannot repeat
it too often, the Medical Times is but one of a class, neither
worse nor better than many other medical journals whose
financial support comes from the proprietary interests rather
than from the medical profession. The responsibility for
the existence of these journals really rests not on the business
men who conduct them on a commercial basis, but on
the physicians who tolerate or encourage them in any way.—(From
The Journal A. M. A., Nov. 8, 1913.)
CAUSE FOR OPTIMISM
A Clean Medical Journal—the South Texas Medical Record
Fortunately, there are forces at work in the medical profession
that make for optimism. An editorial in the last issue—April,
1915—of the South Texas Medical Record, the
official organ of the South Texas District Medical Association,
is especially significant. While not a large journal,
the South Texas Medical Record could well stand as
an example to medical publications of a much more pretentious
character. Its advertising pages are above reproach
and the journal is a credit alike to its editors and to those
members of the profession whose support makes its existence
possible. In the editorial referred to, entitled, “Honest
Advertising—Let Us Cleanse Our Own Linen First,” the
editor-in-chief, Dr. W. Burton Thorning, says:
“A recent editorial, entitled ‘Honest Advertising,’ in a daily
newspaper, furnished the occasion for an editorial comment
in the January number of the Southwestern Hospital
Reporter. The latter, while taking the ground that the newspaper
was inconsistent in uttering high editorial sentiments
and in adjoining columns printing patent medicine advertisements,
implied that newspaper men should be allowed
some latitude in the matter of accepting advertising, on the
plea of being laymen and therefore not expected to possess
the same amount of information concerning patent ‘dope’
that medical men have.
“It would appear to be a fair assumption that a layman,
even though a highly educated and able editor of a great
newspaper, does not know, and cannot be expected to know,
the depth of depravity to which the consumption cure faker
and the cancer quack can descend.
“Granting that the newspaper man accepts the advertisements
through ignorance of the facts concerning their possibilities
for evil, what can be offered in defense of the medical
editor who accepts advertising matter equally pernicious in
its influence?
“Indeed, it is not so many years since many of the so-called
ethical medical journals carried the ads of some of the most
notorious quacks this country has ever known.
“Doubtless there are few, if any, who do so at the present
time, but, on the other hand, there are only a few who do
not advertise unethical institutions, and questionable proprietary
medicines. As a matter of fact some of the most widely
advertised patent medicines of today were formerly advertised
as ethical proprietaries in medical periodicals, the great
majority of which are still serving as a sort of preparatory
school for advertisements that will presently appear in the
lay press.
“What shall be offered in defense of the medical publication
which continues to publish the advertising matter of
hundreds of proprietaries which the Council on Pharmacy
and Chemistry of the American Medical Association has
shown to be either generally worthless or an out and out
fake?
“Can the medical editor plead ignorance? Hardly. To do
so would be to admit utter incapacity. There is only one
inference to be drawn; the publication needs the money and
is not overparticular regarding its source.
“There is a remedy, however, a remedy absolutely certain
in its results. If every physician in the United States for a
period of three months would positively refuse to receive
at his desk a medical journal containing questionable advertising,
this blotch on medical journalism could be erased.
“It is true that many of them would sink, never to rise
again, but the profession would be better off without those
whose existence depends upon ‘phoney’ advertising. There
are, unfortunately, several American journals whose reading
pages are well and carefully edited and a credit to medical
literature, whose advertising pages carry such undesirable
matter that the educated physician can only feel a sense of
disgust.
“Such journals could very well succeed on the quality of
their reading matter and undoubtedly would increase their
circulation enough to more than offset the loss in advertising.”—(From
The Journal A. M. A., May 29, 1915.)
THE COMPARATIVE NUTRIENT VALUE OF COD
LIVER OIL AND COD LIVER OIL CORDIALS [AS]
John Phillips Street, M.S.
Chemist, Connecticut Agricultural Experiment Station
NEW HAVEN, CONN.
For a long time cod liver oil has been recognized as an
easily assimilable nutrient and reconstructive and of special
value in wasting diseases. The unpalatability of the oil, however,
has led to various devices to make it tasteless or to
render it more acceptable to the stomach. Emulsions containing
the oil in mixture with other substances were
exploited, and doubtless served a useful purpose. The oil,
however, but imperfectly concealed, was still disagreeable to
many, and other preparations began to appear on the market,
which claimed to retain the therapeutic virtues of cod liver
oil without its disagreeable characteristics. This practice has
been carried so far that now we find for sale cod liver oil
preparations from which the oil has been removed in its
entirety, and only the name remains. Certain of these products
claim to “represent” the oil and to retain all its virtues;
others are said to contain oil, while still others claim “all
the valuable constituents” of the oil without the oil itself.
In the past, cod liver oil has been considered a food
rather than a medicine, and its value attributed to the easily
digestible and metabolizable oil it contains. This position,
however, has been disputed. By some its therapeutic value
has been attributed to the small amount of iodin present in
the oil, but in recent years the suggestion has been made
that its special potency depends on its peculiar fatty acids.
In this connection the U. S. Dispensatory145 says:
“Other oleaginous substances, certainly not less nutritious,
have not been equally efficient, though taken in much larger
quantities. If this be the true explanation, persons living
chiefly on milk, which abounds in oil, or on fat pork, ought
to show a special exemption from scrofulous complaints.
The probability appears to us to be that, in consequence of
some peculiar principle or principles it contains, it exercises
a stimulant and alterative influence on the processes of
assimilation and nutrition, thereby aiding in the production
of healthy tissue.”
Indeed, Osborne and Mendel146 have shown in their experiments
on albino rats that by substituting cod liver oil for a
portion of the lard in their standard diets, growth was
resumed after failure on foods containing commercial lard
alone as the source of fat. Similar results were secured with
butter-fat and egg yolk fat.
In the light of the theories advanced for the therapeutic
value of cod liver oil, and the results secured by Osborne and
Mendel with the oil itself; it seemed a profitable study to
examine some of the prominent “oilless” preparations on the
market to determine whether or not the claims made for
them as nutrients were justified. Certain of the so-called
cod liver oil preparations are termed “extracts” of cod liver
oil, and are not made from the oil, but from the cod livers
instead. As has been well said,147 “They are preparations,
which, if honestly made, might be worthy of trial, but they
are improperly called ‘extracts’ of cod liver oil, since they
do not contain the fat, which is the active constituent of the
oil, but the extractives from the liver, which may or may
not possess therapeutic virtues. So far as we know, however,
no satisfactory evidence is forthcoming to indicate that
such extractives have any therapeutic value.”
It was with preparations of this class that our experiments
were made. Four of the more extensively advertised brands
were selected, as they represent rather distinct types of this
class of products, as the following claims of their label will
show:
Hagee’s Cordial of the Extract of Cod Liver Oil, Compound.—“Tonic,
stimulant, alterative, reconstructive, nutritive
and digestive.” “Each fluid ounce represents the extract
obtainable from 1⁄3 fluid ounce of cod liver oil (the fatty portion
being eliminated), 6 grs. calcium hypophosphite, 3 grs.
sodium hypophosphite, 1⁄2 gr. salicylic acid (made from oil
wintergreen), with glycerin and aromatics.”
Vinol.—“The modern tonic reconstructor containing the
medicinal extractives of fresh cod livers with peptonate of
iron.” “When the blood is poor, when more fresh blood is
needed, when the weak need strength, when the throat and
lungs are affected, TAKE VINOL.”
Wampole’s Perfected and Tasteless Preparation of an
Extract of Cod Liver.—“Contains a solution of an extractive
obtainable from fresh cod livers, the oily or fatty portion
being afterward eliminated. This extractive is combined
with liquid extract of malt, fluid extract of wild cherry
and compound syrup of hypophosphites (containing calcium,
sodium, potassium, iron, manganese, quinine and strychnine).”
Waterbury’s Compound, Plain.—“Made from cod liver oil,
digestive ferments, malt extract unfermented, hypophosphites
comp. special, ext. cherry, eucalyptus, aromatics, etc.”
Thus we have represented in our experiments an “extract”
with hypophosphites, one with peptonate of iron, one with
malt extract and hypophosphites and the alkaloids quinin
and strychnin, and one with malt extract and hypophosphites
without alkaloids.
In order to prepare a dry ration of suitable keeping properties,
it was necessary to remove the alcohol and water from
the various preparations. This was done by evaporation under
reduced pressure at from 40 to 55 C. (104 to 131 F.). In
the case of Hagee’s Cordial it was claimed that one fluidounce
of the preparation represented 1⁄3 fluidounce of cod
liver oil, and in the subsequent substitution for cod liver oil
in our rations, this ratio was used for all four products.... These
are very dissimilar preparations, the alcohol
ranging from 7.50 to 18.69 per cent., the extract from 8.72 to
39.53 per cent. (10.81 of the 13.18 gm. of extract in Hagee’s
Cordial being glycerin), the ash from 0.305 to 1.967 per cent.,
the reducing sugars from 1.35 to 17.10 per cent., and the
glycerin from a trace to 10.81 per cent. Wampole’s contained
quinin and strychnin, the others no alkaloids; salicylates
were present in all but Wampole’s; saccharin in Hagee’s.
The Pettenkoffer test for biliary acids gave a negative result
in Hagee’s and Wampole’s; in Vinol and Waterbury’s, small
amounts of fatty acids were obtained, amounting to 0.016
and 0.032 gm. per hundred c.c., respectively, quite insignificant
amounts.
The feeding experiments were made on albino rats of both
sexes, which were placed, when about 6 weeks old, on a
standard ration, No. 7, and after several months, when a
failure to maintain weight was indicated,148 an amount of
dealcoholized cordial extract equivalent to 18 per cent. of
cod liver oil was substituted for a portion of the lard, the
cordial extract later being replaced by an equivalent amount
of cod liver oil.
*******
SUMMARY
Table 11 gives a summary of the actual gains of the fifteen
rats on the four rations, compared with the gains shown by
cod liver oil and those shown by normal rats at the same
period of their life history.
TABLE 11.—SUMMARY OF RESULTS*
| Rations | Total Normal
Gain, Gm. | Total Actual
Gain, Gm. | Average Normal
Gain, Gm. | Average Actual
Gain, Gm. |
| Hagee ration | 24 | -36 | .2 | 6 | | -9 | .1 |
| Cod liver oil ration | 114 | 156 | .4 | 28 | .5 | 39 | .1 |
| Vinol ration | 42 | -1 | .5 | 10 | .5 | -0 | .4 |
| Cod liver oil ration | 42 | 87 | .5 | 10 | .5 | 21 | .9 |
| Wampole ration | 83 | 51 | .4 | 20 | .8 | 12 | .9 |
| Cod liver oil ration | 62 | 81 | .5 | 15 | .5 | 20 | .4 |
| Waterbury ration | 32 | 0 | .3 | 10 | .7 | 0 | .1 |
| Cod liver oil ration | 42 | 87 | .4 | 14 | | 29 | .1 |
* In this table are given the totals and averages of the figures already
presented
in Tables 4, 6, 8 and 10. |
In considering the effect of these preparations as general
medicines, their alcohol content must not be overlooked.
Hagee’s Cordial contains 7.50 per cent. of alcohol by volume,
Vinol 18.60 per cent., Wampole’s Preparation 16.59 per cent.,
and Waterbury’s Compound 11.25 per cent. Full strength
whisky contains 50 per cent. of alcohol by volume. By following
the doses prescribed by the manufacturers of these
preparations, the user would consume daily the following
equivalents of full strength whisky:
In Hagee’s Cordial | 0.24 | fluidounce |
In Vinol | 0.8 | fluidounce |
In Wampole’s Preparation | 0.7 | fluidounce |
In Waterbury’s Compound | 0.6 | fluidounce |
These amounts of alcohol are by no means negligible and
doubtless explain to a considerable extent the source of the
alleged tonic virtues of these preparations.
The results of the experiments may be summarized as
follows:
Hagee’s Cordial failed to sustain rats during periods of
seven and fourteen days, the rats showing a loss in weight
of 36.2 gm., instead of the normal gain of 24 gm.
Vinol in two cases sustained and in two cases failed to
sustain growth during periods of from eleven to thirty-five
days, the net loss in weight of the four rats being 1.5 gm.,
instead of the normal gain of 42 gm.
Wampole’s Preparation in three Cases sustained and in one
case promoted growth in rats during periods of eighteen
and thirty-nine days, showing, however, only 51.4 gm. gain
in weight instead of the normal 83 gm.
Waterbury’s Compound in two cases sustained and in one
case failed to sustain rats during periods of fourteen and
thirty days, the net gain in weight, however, being but
0.3 gm. instead of the normal 32 gm.
Cod liver oil showed a gain of 42.4 gm. over the normal,
while with the same rats Hagee’s Cordial showed a loss of
60.2 gm. Cod liver oil showed a gain of 45.5 gm. over the
normal, while with the same rats Vinol showed a net loss
of 43.5 gm. Cod liver oil showed a gain of 19.5 gm. over
the normal, while with the same rats Wampole’s Preparation
showed a loss of 31.6 gm. Cod liver oil showed a gain of
45.4 gm. over the normal, while with the same rats Waterbury’s
Compound showed a net loss of 31.7 gm.
Not only did cod liver oil show a marked superiority as
a source of nutriment over Hagee’s Cordial, Vinol, Wampole’s
Preparation and Waterbury’s Compound, but it also
showed a remarkable reconstructive and recuperative power
in its ability to enable rats to gain weight rapidly and steadily
after having suffered from a deficiency in nutriment when
fed the four preparations named above.—(Abbreviated from
The Journal A. M. A., Feb. 20, 1915.)
DIABETIC FOODS OFFERED FOR SALE IN THE UNITED STATES
A Preliminary Report
John Phillips Street, M.S.
Chemist, Connecticut Agricultural Experiment Station
NEW HAVEN, CONN.
Recent references in The Journal to gluten flours and
certain other foods offered for the use of diabetics suggest
that a preliminary report of an investigation just about completed
in my laboratory by Prof. L. B. Mendel and myself
might be useful to many diabetics and to physicians who
are called on to arrange their dietaries.
In 1906 this laboratory, then under the direction of Dr.
A. L. Winton, who is now with the Department of Agriculture,
made its first examination of commercial diabetic
foods. In nearly every year since it has analyzed various
other brands as they appeared on the market. The demand
for the reports on these foods and the many inquiries directed
to us have led us to make a more extensive review of the
situation, and to collect as far as possible all information as
to the quality of the so-called “diabetic” foods offered to
the American public. That the present state of the market
is unsatisfactory is well known, and the inferiority (from the
diabetic’s point of view) of many of the products at present
offered is unfortunately familiar to all careful dietitians.
The most dangerous feature of the present situation is that
the unsuspecting patient is led to purchase foods, generally
at an exorbitant price, which are not only misrepresented
but which may be positively harmful to him. In this day
of self-medication this condition is all the greater menace
to the diabetic.
Without any attempt to suggest methods of treatment for
diabetes, which is the province of the physician, I may say
that it is well recognized that diabetes is primarily a disturbance
of nutrition, in which the normal ability of the
body to make use of carbohydrates is more or less completely
impaired. All recent authorities agree in placing the
chief emphasis on the role of diet in the management of this
disease. Janeway, Benedict, Joslin, Futcher, Falta, Strauss,
von Noorden and other writers on diabetes could be quoted
at length in support of this view. The importance of a
restriction of the carbohydrates in certain cases and certain
aspects of diabetes is admitted by practically all competent
authorities. In order to prescribe a starch-free and sugar-free
dietary, which at times is necessary, and to know
accurately the actual amount of these carbohydrates contained
in the various available foods, the physician must
rely on the cooperation of the chemist to furnish this
requisite information. This is our excuse, if any be needed,
for our present investigation.
There seems to be some uncertainty as to what sort of
preparation is entitled to be sold as a “diabetic” food. Granting
the desirability of feeding the patient all the carbohydrate
he can tolerate, and recognizing the possible value
of the oatmeal, potato, rice and other treatments, in which
a relative abundance of carbohydrate is fed for a limited
period, it would seem that a low percentage of carbohydrates
should be a requisite for a “diabetic” food. Certainly
no special food containing nearly as much carbohydrate as
a normal food of the same class should be entitled to this
appellation. Flours, breads, biscuits, chocolates, breakfast-foods,
macaroni, etc., containing only a slightly lowered percentage
of carbohydrates, are no more entitled to be called
“diabetic” foods than the normal foods themselves. It is true
that, when a patient’s carbohydrate tolerance is well established,
the use of foods containing 20, 25 and even 35 per
cent. of carbohydrates might be permissible, when used under
the direction of a competent physician; but when a strict
diet is necessary, such as is required to determine this tolerance,
even these relatively low percentages are objectionable,
if not dangerous.
It has been our purpose to include in this investigation,
as nearly as possible, all available data on the composition
of all diabetic foods sold in America. Our report, therefore,
will be in part compiled, but in greater part will consist of
our original analyses. It will show 539 analyses of about
400 brands, 200 of which are our own new analyses and 110
those made in this laboratory in previous years.
While the purpose of this preliminary note is to call attention
to the better preparations rather than to emphasize those
which are obviously objectionable and fraudulent, it may not
be out of place to summarize briefly our findings in general.
The full details of the investigation are now being prepared
for publication and will shortly be issued as a report from
the Connecticut Agricultural Experiment Station.
One hundred and eight samples of sixty-eight brands of
flours and meals are included in the report. Sixty-seven
of these were sold as “gluten” flours, twenty of which did
not even satisfy the low government standard of 35 per
cent. protein. Twelve samples contained less than 13 per
cent. carbohydrates, while the remaining gluten flours ranged
from 28 to 76 per cent.
The soy bean flours contained from 23 to 26 per cent. of
carbohydrates, the almond meals 17 per cent., and a cotton-seed
flour 21 per cent. Other “diabetic” flours, not specifically
sold as “gluten” flours, contained from 67 to 80 per
cent.
The purchaser of gluten flours at the present time may
obtain preparations containing from 87 to 11 per cent. of
protein and from 4 to 76 per cent. of carbohydrates, at a
cost of from 9 cents to $1.56 per pound.
In view of the government’s low standard for gluten flour,
and because of the wide variations in composition found in
the brands at present on the market, proper protection of
the diabetic demands that the manufacturers of these flours
should be required to state on the label the guaranteed percentages
of both protein and carbohydrates.
Three samples of American soft gluten breads contained
from 35 to 37 per cent. of carbohydrates; two other brands
contained 49 and 54 per cent., little, if any, lower than found
in ordinary wheat bread.
One hundred and forty-eight analyses of 112 brands of
hard breads, biscuits, rusks, cakes and other bakery products
are included. Eight brands of Luftbrot, or aerated
bread, are reported; two of these contained from 9 to 12
per cent. of carbohydrates, one 20 per cent., two from 31
to 33, and the other three from 44 to 54 per cent.
A number of the brands of rolls, biscuits, breads, etc.,
showed satisfactorily low percentages of carbohydrates,
thirty-five samples containing from 1 to 25 per cent., forty-four
samples containing from 35 to 55 per cent., and forty-one
over 55 per cent., seven of the latter exceeding 72
per cent.
The cost of the Luft breads ranged from 71 cents to $2.33
per pound. Biscuits, containing 11 per cent. or less of carbohydrates,
cost from 72 cents to $3 per pound. A number
of brands, containing from 43 to 77 per cent., cost from
$3 to $3.60 per pound. Even the cheaper preparations, containing
from 50 to 77 per cent., no better, and in some cases
even worse, for the diabetic’s use than ordinary bread, cost
from 30 to 41 cents per pound.
Fourteen samples of breakfast-foods were analyzed, five
of which contained from 44 to 54 per cent. of carbohydrates,
somewhat lower percentages than normal. Seven of the ten
brands of recommended macaroni, noodles, etc., contained
over 70 per cent. of carbohydrates, the other three from
42 to 51 per cent.
The analyses are given of fourteen samples of peanut
butter, five of almond paste and butter, two of pine-nuts, one
of almonds and ten of miscellaneous nut foods. As was to
be expected, most of these preparations proved to be suitable
diabetic foods. The peanut butters contained from 12
to 20 per cent. of carbohydrates, with an average of 15 per
cent. The three almond pastes contained from 30 to 40
per cent., one showing an addition of 11 per cent. cornstarch.
The two almond butters contained only 7 and 8 per cent.,
the pine-nuts from 3 to 8 per cent., and the almonds 16 per
cent. The other nut preparations contained from 6 to 44
per cent. carbohydrates.
Seven brands of diabetic chocolates contained from 10 to
50 per cent. carbohydrates, while four cocoas contained from
21 to 51 per cent. The chocolates cost from $1.63 to $2.06
per pound, and the cocoas were similarly expensive.
Two sugar-free milks were examined which were true to
name, containing only the merest traces of carbohydrates.
One “diabetic” baking-powder examined contained no starch,
another brand from 14 to 16 per cent. Various jams, preserves
and other fruit products were examined which contained
from 1.24 to 7 per cent. of invert sugar, percentages
far below the normal. A currant-juice contained only 0.85
per cent. of invert sugar. Four of the fruit preparations
were artificially colored with a coal-tar dye—a permitted
color to be sure, but seemingly quite out of place in foods
intended primarily for the use of invalids.
As already stated, the main purpose of this investigation
was not so much to detect fraud as to secure information
which would be of benefit to the diabetic and to the physician
who seeks foods suitable for a low carbohydrate diet.
In the accompanying tabulations a summary is given of the
brands, sold as diabetic foods, which showed less than 35
per cent. of carbohydrates, arranged in the order of their
carbohydrate149 content. A date in parentheses following a
brand name signifies that the brand named showed variations
in different years; in other cases, in which the agreement
was close, the results have been averaged.
BRANDS SHOWING UNDER 5 PER CENT. OF CARBOHYDRATES
| Per Cent. |
Casoid Baking Powder | .0 |
Dr. Bouma Sugar-Free Fat-Milk | .0 |
Whiting’s Sugar-Free Milk | .0 |
Rademann’s Currant Juice “ohne Zucker” | 0.9 |
Kalaria Batons (1909) | 0.9 |
Glidine | 1.0 |
Casoid Sugarless Marmalade | 1.2 |
Casoid Sugarless Jam | 1.5 |
Kalari Biscuit | 1.7 |
Casoid Dinner Rolls | 2.1 |
Casoid Flour | 2.2 |
Jireh Diatetic Pine Nuts | 3.4 |
Rademann’s Preserved Fruits, “entzuckert” | 3.5 |
Kellogg’s Protose | 3.6 |
Barker’s Gluten Food “A” | 4.1 |
Kellogg’s Pine Nuts | 4.2 |
Kellogg’s 80 Per Cent. Gluten Biscuit | 4.4 |
Bischof’s Gluten Flour | 5.0 |
BRANDS SHOWING FROM 5 TO 10 PER CENT. OF CARBOHYDRATES
| Per Cent. |
Casoid Biscuits No. 2 | 5.6 |
Rademann’s Preserved Fruits “in eigenen Saft” | 5.7 |
Barker’s Gluten Food “B” | 5.9 |
Kellogg’s Nuttolene | 6.3 |
Nashville Sanitarium Nutcysa | 6.3 |
Huntley and Palmer’s Akoll Biscuit | 6.5 |
Nashville Sanitarium Nutfoda | 6.8 |
Rademann’s Preserved Fruits “ohne Zucker” | 7.0 |
Muller’s Tomatoes für Diabetiker | 7.3 |
Barker’s Gluten Food “C” | 7.7 |
Kalari Batons (1913) | 7.7 |
Casoid Biscuits No. 3 | 7.8 |
Kellogg’s 80 Per Cent. Gluten (1912) | 7.9 |
Casoid Biscuits No. 1 | 8.0 |
Kellogg’s Almond Butter | 8.2 |
Fromm’s Uni Bread | 9.0 |
Metcalf’s Vegetable Gluten (1913) | 9.8 |
BRANDS SHOWING FROM 10 TO 15 PER CENT. OF CARBOHYDRATES
| Per Cent. |
Kellogg’s Pure Gluten Biscuit (1906) | 10.2 |
Health Food Pure Washed Gluten Flour (1913) | 11.1 |
Health Food Alpha Diabetic Wafers | 11.3 |
Loeb’s Imported Gluten Flour | 11.8 |
Health Food No. 1 Proto Puffs | 11.9 |
Kellogg’s Potato Gluten Biscuit (1906, 1909) | 11.9 |
Kellogg’s Nut Meal | 12.1 |
Kellogg’s 80 Per Cent. Gluten (1909) | 12.5 |
Nashville Sanitarium Nut Butter | 13.0 |
Kellogg’s Nut Butter | 13.9 |
Bischof’s Diabetic Gluten Bread | 14.3 |
Jireh Diabetic Baking Powder | 15.0 |
Peanut Butter (range from 12 to 20) | 15.0 |
BRANDS SHOWING FROM 15 TO 20 PER CENT. OF CARBOHYDRATES
| Per Cent. |
Casoid Chocolate Almonds | 16.1 |
California Paper Shell Almonds | 16.3 |
Callard’s Cocoanut Biscuit | 16.4 |
Rademann’s Diabetiker-Chokolade | 16.9 |
Health Food Almond Meal | 16.9 |
Callard’s Ginger Biscuit | 18.1 |
Callard’s Prolactic Biscuit | 19.3 |
BRANDS SHOWING FROM 20 TO 25 PER CENT. OF CARBOHYDRATES
| Per Cent. |
Callard’s Almond Shortbreads | 20.7 |
Callard’s Casoid Rusks | 20.8 |
Rademann’s Diabetiker-Makronen | 20.8 |
Health Food Protosoy Diabetic Wafers | 21.2 |
Jireh Patent Cotton-Seed Flour | 21.3 |
Casoid Lunch Biscuit | 21.6 |
Rademann’s Diabetiker-Chokolade Biscuit | 21.9 |
Cereo Soy Bean Gruel Flour | 23.7 |
Health Food Salvia Sticks | 24.0 |
Health Food Protosoy Soy Flour | 24.5 |
Metcalf’s Soja Bean Meal | 25.0 |
BRANDS SHOWING FROM 25 TO 35 PER CENT. OF CARBOHYDRATES
| Per Cent. |
Jireh Soja Bean Meal | 25.8 |
Brusson Chocolat with Added Gluten | 26.4 |
Rademann’s Diabetiker-Stangen | 27.0 |
Rademann’s Diabetiker-Dessert-Gebäck | 27.5 |
Nashville Sanitarium Malted Nut Food | 27.5 |
Metcalf’s Vegetable Gluten (1906) | 28.1 |
Health Food Pure Washed Gluten Flour (1906) | 29.5 |
Fromm’s Luft Bread | 30.7 |
Spencer’s Almond Paste | 31.6 |
Fromm’s Conglutin-Diabetiker-Schokolade | 32.7 |
Health Food No. 2 Proto Puffs | 33.3 |
Ferbuson Gluten Bread | 33.6 |
Gum Gluten Breakfast Food | 34.2 |
—(From The Journal A. M. A., June 28, 1913.)
THE JIREH DIABETIC FOOD COMPANY
The Company Rises to Explain in a Brief Note of One
Thousand Words
Exploiters of fraudulent and dangerous pharmaceutical
products have no love for The Journal. When such products
are exposed in these pages, their manufacturers seldom reply
to the criticisms except through indirect channels. Then the
replies are frequently replete with billingsgate and denunciation
of The Journal and its editor, the Association and
the medical profession generally.
We have, at different times, had to call the attention of
the public and the medical profession to the fraudulence and
dangers of some of the products of the Jireh Diabetic Food
Company. We have shown that the Jireh company lied
boldly and directly so long as it could do so without getting
into the courts, and that it lies inferentially still; we have
shown that Jireh flour had practically as much carbohydrate
as ordinary flour; we have shown that, probably to escape
prosecution under the Food and Drugs Act, the Jireh concern
has coined the word “diatetic” and substituted it for
the word “diabetic,” which used to appear in its advertisements;
we have shown that the claim made for the Jireh
products that they are “starch-changed” was a false one, and
that the company has modified this to “starch-treated,” presumably
to avoid being haled into the courts under the “pure
food law;” we have shown, in short, the unreliability both
of the Jireh concern and of its products.
Two or three weeks ago a New York physician wrote to
The Journal for information regarding the Jireh products.
We sent him such matter as had been published on the subject,
and he showed this material to a patient who was using
the Jireh products. The patient, in turn, expressed her
opinion of the product to the retailer from whom she had
been purchasing it. A day or two later she received a letter
from the Jireh Diabetic Food Company, which, in spite of
its length and discursiveness, we publish in full, so that
physicians may know just what this company thinks of them.
The letter, which is dated Nov. 20, 1913, really belongs in
the “Knocks and Boosts” department, but its length prevents
its use there. Here it is:
“We learned through Mess. Cushman Co. that you are a
constant user of Jireh Foods for some time past, and that
recently a certain derogatory statement was brought to your
attention relative to our product. We feel an explanation is
due you for two reasons.
“First, because we want you to continue using Jireh Foods
and thus receive the benefits of the same; and second, that
the remarks called to your attention are not only libelous,
but are in no way pertinent enough to detract from the value
of our product. In the first place, we want to state that
the particular journal in question has been endeavoring to
injure our business for some time past and that we are not the
only descent [sic] manufacturers of foods that are suffering
in this way at the hands of the editors of this particular
magazine. Since you are interested in the Jireh Foods, you
may be more interested to learn why this particular magazine
is so anxious to injure our reputation. The reason is
very obvious if you will take into account the fact that this
magazine is the official journal of the medical association of
this country which is known as the backer of the medical
trust. It is very clear to you, no doubt, that there are some
physicians, particularly those that are associated with the
magazine, who are anxious to stamp out of existance [sic]
such concerns that offer a bona fide product, a meritorious
product which actually produces the required results, without
the aid of medicine.
“For the particular maladies for which we offer our foods,
we have been very successful, consequently the antagonism
on the part of this particular journal is the logical thing.
In addition to this, however, and perhaps more important to
us, is the fact that the editors of this magazine have made
it known to us that they will not approve of our product
until such time that we care to be dominated by the moving
influence of the magazine in question. They want us to supply
them with the private formula which we use for manufacturing
our foods and to enlighten them and show them
the various processes applied to our products to produce the
required results. As a bona fide and ethical business house,
we absolutely and unqualifiedly refuse to comply with this
wish, and will always refuse to do so, no matter how often
they may attack us. We stand strictly on principle in this
matter and propose to run our business in our own way,
and will not, under any circumstances, allow a magazine or
anybody else dictate to us under what conditions we are
allowed to do our business.
“The remarks which they make would perhaps hurt us some
if they emanated from a source that was qualified to judge
the merits of our food. The absurd side of the issue, and
perhaps the comical side of it, is the fact that the honorable
gentlemen who assume to condemn a product, know less
about the disease for which the product is offered, and much
less about diet and foods than the average layman. Consequently,
we consider it simply absurd to allow them to step
into our business and dictate policy to us. This is the jist
[sic] of the discontent that prevails between the magazine
editors and ourselves, and as long as we refuse to comply
with their wish, we certainly cannot expect them to speak
well of us. It has become a notorious fact in the medical
profession that their criticisms are almost valueless, inasmuch
as they stop at nothing in order to create sensationalism,
and have attacked not only ourselves, but every bona
fide manufacturer of foods and drugs in this country who
has refused to fall in line with them.
“This explanation, we trust, will appeal to your good
judgment and will convince you of what is said about us is
untrue. Furthermore, our business has grown to colossal
proportions, notwithstanding their endeavors to crush us.
We call your attention to a most peculiar fact: that is, that
they make no comment whatsoever as to the product and
its therapeutic value in the treatment of diabetes. You notice
they make an awful play on our literature, which was changed
merely to suit conditions in business. We also wish to call
your attention to another peculiar fact, and that is that
a great majority of the physicians who are in with this
magazine readily recommended our foods, and we also believe
that your physician, after reading this letter, will feel the
same way as most physicians do in relation to our foods. We
believe that your physician is perfectly willing to be convinced
that our foods are as represented to be, and the very
best clinical evidence as this is the effect our foods have
had upon you. Finally, if the foods are palatable and wholesome
and have alleviated the annoying symptoms of Diabetes,
then why should you be guided by the opinions of
demagogues and yellow journalists?
“You may be pleased to learn that our foods have received
the attention of Dr. Wiley, the well-known chemist and food
scientist of this country, and we have now in our possession
his reports showing the very high standard of our foods.
This, in our opinion, is of more consequence than all the
harangue which those venerable gentlemen of the magazine
may indulge in.
“We want to convince you without an atom of doubt that
we are honest and bona fide in everything we say, and we
extend to you a hearty invitation to call at the first opportunity
and shall be glad to tell you anything you may wish
to know. Thus awaiting the pleasure of this visit, we beg
to remain
“Yours very truly,
“Jireh Diabetic Food Co.”
We learn from this letter that The Journal’s “remarks”
on the Jireh product are “libelous.” We have made them
many times and for several years past; if libelous, the manufacturer
has excellent grounds for damages. We learn, too,
that our remarks “are in no way pertinent enough to detract
from the value” of the Jireh products. Why, then, take any
notice of them? We learn, moreover, for the hundred-and-first
time, that The Journal is the official organ of the
“medical trust,” and we are told that “there are some physicians”
that are opposed to the Jireh Foods because these
products cure diabetes without the use of medicine! The
Journal, so the company says, wants the Jireh Diabetic Food
Company to supply its “private formula” and to show the
“various processes” by which the Jireh products are made.
Such statements indicate that the Jireh Diabetic Food Company
does not confine its mendacity to the mere advertising
of its product, where the necessity for lying is naturally great.
The ambiguous remarks regarding Dr. Wiley are evidently
intended to convey the idea that the doctor approves of the
Jireh products. Dr. Wiley was sent a copy of the Jireh
letter and his attention directed to the statements appearing
therein regarding himself. He replied:
“In regard to the Jireh products and their claims that our
reports show the very high standing of their foods, I would
say that I consider such a claim entirely false.... We
did examine five or six products in our own laboratory, however,
and found them to be of very fair composition per se,
but not of a composition that afforded any legitimate basis
for their claims. We entirely disapproved ... three of
the products making special claim as to their fitness for
diabetics. [Italics ours.—Ed.] These were the Wheat Nuts,
the Jireh Flour and the Patent Barley. Two of the other
products were passed with a non-committal rating, which
means that they are not actually disapproved, but the star
marking is not accorded. These products were the dietetic
Rusks and the Macaroni. For the latter especially no specific
claims seem to be made. We called attention, however,
to the generally objectionable juggling of terms indulged
in by this company....”
The Jireh concern says that in spite of the efforts of The
Journal to “crush” it, “our business has grown to colossal
proportions.” Of this the New York physician who sends
us the letter says: “Their ‘colossal proportions’ must have
received a slight jar or they would not have taken time
to write such a letter.”—(From The Journal A. M. A., Dec.
20, 1913.)
THE NAME “EPINEPHRIN” VERSUS THE
NAME “ADRENALIN” [AT]
There are thirty or more different brands of the blood-pressure-raising
principle of the suprarenal gland on the
market, five being in this country alone. These products
are identical so far as their chief constituent is concerned;
they differ, however as to the solvent and preservative used.
The processes of manufacture of some of them are patented;
all of them are sold under trade names.
Until two years ago there was no common name applicable
to this active principle; whenever reference was made to
it a trade name had to be used. At that time the Council
on Pharmacy and Chemistry, realizing the need of a generic
term, adopted “epinephrin” as such a term. This name was
selected in part because Abel had adopted it in 1899; in part
because, so far as could be discovered, it was the name
under which, through Abel’s publications, the substance first
appeared in medical literature; and in part because it seemed
to be the only suitable one not already appropriated by some
commercial firm.
After the publication of the Council’s report, The Journal
began gradually to use the term in those cases in which it
seemed clear that the proprietary term was used in a generic
sense. The substitution of the name “epinephrin” for “adrenalin”
in the abstracts of certain foreign articles caused
Parke, Davis & Co. to write a letter of protest which called
forth the discussion appearing in the Propaganda Department
of this issue.
The amount of space devoted to this matter may be criticized
and considered unwarranted by those who do not
realize the importance of the subject. The criticism is to
a certain degree, just. The somewhat inordinate length of
the article is due in part to the unfortunate fact that, in
availing themselves of the courtesy extended by The Journal,
Parke, Davis & Co., in their reply, have injected into the discussion
side-issues, such as the priority of discovery, the
superiority of their product, etc., whereas the question under
discussion is simply that which relates to the name. It is,
however, not altogether a matter for regret that the discussion
has been thus broadened, for it brings before our readers
many facts regarding the discovery of an important medicinal
agent that are not generally known, at least by physicians.
Whether or not “adrenalin” is superior to “adrin,” “suprarenalin,”
“suprarenin,” “adnephrin,” or to any other of the
preparations is entirely immaterial in this connection. The
point is that the active principle of the suprarenal gland is
on the market under various trade names, and that a name
common to all has been selected to be used when no particular
brand is referred to. The fact that “adrenalin” is
regarded by many, both here and abroad, as a common, generic
name does not alter the fact that it is claimed as a trade
name by a commercial house and, therefore, presumably at
least, cannot be used except as such.
Among the facts brought out in this discussion, one stands
out clearly: that Abel deserves as much credit for the discovery
as any other man, if not more. Credit belongs to
Takamine for making use of reactions which were already
well known. His work was a step in the progress of knowledge
of the substance, but it was a step which he could not
have taken but for what others, Abel especially, had accomplished
and published. Abel’s magnificent work, covering
several years, deserves as much credit, to say the least, as
that of Takamine. And it should be kept in mind that the
former worked in the interest of science, and published his
results for the benefit of all. He had no hope of pecuniary
reward, asked for none, and received none.
Let us repeat, however, that these are side issues; the question
is simply that of name. It cannot be too strongly emphasized
that “epinephrin” is a true scientific name for the
active principle of the suprarenal gland, and that it should
be used on all occasions when the active principle and not
some particular firm’s make is referred to.—(From The
Journal A. M. A., March 25, 1911.)
THE HORD SANITARIUM
“Propaganda for Reform Department:—One often hears it
declared that the present time is the worst ever known for
a young man to make a fortune or get a start to one.
“All a mistake, as the enclosed letter from the Hord Skinatarium
will certify. At $25 this equals $2,500 for 100 cases, $25,000 for
1,000 cases, and all any young doctor needs is a little push to be
as rich as J. D. in a few months. If you know of any cases send ’em
in and get your $25.
“K. T. Crossen, M.D., Carbondale, Ohio.”
With his letter Dr. Crossen encloses a circular letter from
the Hord Sanitarium, “For Liquor and Drug Habits, A Cure
Positively Guaranteed,” and an unsigned check on the Farmers
National Bank, Shelbyville, Indiana, for $25 payable to
himself. Printed on the check in large red letters is the
statement:
“This check will be countersigned upon you bringing
or sending us a patient.”

Fig. 1.—Photographic reproduction of the unsigned check that the
Hord Sanitarium sends to physicians.

Fig. 2.—Part of the letter that accompanies the check for twenty-five
dollars.
The Journal has received these circular letters and unsigned
checks by the hundreds from physicians who have expressed
very frankly their contempt of the kind of business the Hord
Sanitarium is engaged in. These correspondents seem to
have overlooked the fact that The Journal has already commented
editorially on this particular insult to the medical
profession. For this reason we reprint the editorial note,
“Ethics!” from The Journal, Sept. 27, 1913:
“We will pay you $25 for each patient that you bring or
send us.” Thus, to physicians, writes the Hord Sanitarium
of Shelbyville, Indiana, and continues: “We have a perfect
and an absolute cure for all liquor and drug addictions.”
Fearing doubtless that those to whom these offers are made
may be disgusted with the first proposition and will realize
the evident falsity of the second, the concern encloses a list
of references “showing the high moral and professional
standing of our sanitarium.” The Hord Sanitarium emphasizes
further that it does a strictly “no cure no pay” business.
Suspiciously similar is the offer made by the Mizer Sanatorium
of Coshocton, Ohio, Blake V. Mizer, manager. Not
many months ago Mr. Mizer was running the Hord Sanitorium
(the concern’s own spelling), which at that time
advertised “the only guaranteed cure.” Now, Mr. Mizer
hurls invectives at those concerns that make “unreasonable
guarantees” and adds virtuously that “we resort to no such
unethical and pretended guarantee in order to do business.”
Nevertheless, in small type in the northwest corner of his stationery,
Mr. Mizer admits that his “proposition” is no cure
no pay. The fees of the Mizer Sanatorium “are $125 to $250,
depending on the room.” The physician’s rake-off is “20 per
cent. of the above.” “This,” explains Mr. Mizer blandly,
“is simply a matter between ourselves and does not concern
the patient in any way.” Of course not. All the patient
has to do is to pay the bills. And the Mizer Sanatorium is
“conducted along ethical ... lines”—Mr. Mizer says
so. The Mizer Sanatorium has odd ideas of what constitutes
ethics, medical or otherwise, for not long ago it advertised,
in such medical journals as would accept its “copy,”
that “medical ethics prevents the statement here of the
whole truth about the Mizer treatment.” Of course medical
ethics never prevented truthful statements of any kind. A
dirty business; no other words express it. When the Hord
Sanitarium and the Mizer Sanatorium claim to cure all
cases of drug or liquor addiction, they make claims that
are false—cruelly false. When these concerns try to drum up
trade by offering secret commissions to physicians they insult
an honorable profession. The fact that they send out this
sort of advertising matter is presumptive proof that there
are some physicians who will patronize them. Such as do
so are unfair to their patients and untrue to the ideals of
medicine.—(From The Journal A. M. A., Jan. 31, 1914.)
THE GERMAN PROPAGANDA FOR REFORM
Appreciation by a German Lay Publication
Of all those interested in the reform of the proprietary
drug business, the patient has the most at stake—and the
public is beginning to understand this fact. If physicians
are slow in recognizing the necessity for improvement, laymen
will eventually demand reform in their own interests.
The movement, therefore, will not be halted by the indifference
of the unprogressive element of the medical profession.
New evidence of this fact is furnished by a recent editorial
comment by the German lay periodical, Wohlfahrt und
Wirtschaft (Public Welfare and Economics), on the Arzneimittel-Kommission,
a German organization resembling in
purpose if not in scope the Council on Pharmacy and Chemistry
of the American Medical Association.
“One would suppose,” says this lay journal, “that medicinal
preparations which did not win the approval of scientific
medicine would not be used by any physician, but the contrary
is the case. In fact, those new medicinal preparations
or old ones with new names that flood the market far surpass
the actual demand according to the judgment of all
authorities. The impartial advisers in this field, practitioners
and members of medical faculties, demand, as a matter of
public welfare that this overproduction should be regulated
in the interests of the sick, the consumers; but, unfortunately,
a medical man, like any one else, is impressed by the suggestion
from advertising done on a large scale.”
The movement for reform, Wohlfahrt und Wirtschaft goes
on to explain, is not exclusively a medical one. It is a part
of the reaction of “economic common sense” against a too
individualistic commercial system which leads to overproduction.
In other words, it is a reaction against the system
of making things because they can be sold rather than because
they are needed. The interests of producers need to be harmonized
with those of consumers, not merely in the drug
trade alone, but throughout the commercial world. Wohlfahrt
und Wirtschaft quotes with unqualified approval the ArzneimittelKommission’s
statement of its position: An industry
which serves the science of healing must be guided by that
science. (Eine Industrie die der Heilwissenschaft dient, hat
sich nach der Heilwissenschaft zu richten.)
The movement for reform in Germany has apparently
gathered sufficient impetus among the laity to go on of its
own momentum, even though, with one exception, German
medical journals, reluctant to lose the advertising of drug
houses by publishing criticisms of their wares, have become
lukewarm, if not antagonistic, to the efforts of the ArzneimittelKommission.
The one exception is the Therapeutische
Monatshefte, which, in its May issue, quotes in full the
editorial just referred to and makes the following comment:
“These lines reveal such intimate knowledge and correct
judgment of existing conditions that the suggestions advanced
in regard to possible reforms deserve serious consideration.
For us physicians the editorial is important in that it recognizes
that the efforts of the profession to accomplish the
reforms aimed at are rational and beneficial from the standpoint
of general economics and the public welfare.”—(From
The Journal A. M. A., June 13, 1914.)
THE GERMAN COUNCIL ON PHARMACY
AND CHEMISTRY
At the meeting of the German Congress for Internal Medicine
in 1911, a German council on pharmacy and chemistry,
Die Arzneimittelkommission des Kongresses für innere Medizin,
was organized, with purposes similar to those for which
the Council on Pharmacy and Chemistry of the American
Medical Association was created. As practically nothing has
been done to restrict the advertising of proprietaries in Germany,
the task of the commission was tremendous. Its work
has been noted in The Journal from time to time.150 A review
of what has been done up to the present is given by Heubner,151
and indicates some differences between conditions in Germany
and this country. The members of the commission
found confronting them the same evils that met the early
efforts of the American council, namely, dominant proprietary
interests, a subservient and financially interested medical press
and an indifferent profession. Moreover, the pecuniary interest
of the editors of German medical journals in the profits
of advertising seems to be more direct and more important
than in America. The German commission, in Heubner’s
opinion, was placed at a disadvantage compared with the
American council from the first. Funds for investigation were
lacking, and the commission had no journal in which its
objects could be presented to the medical profession. At the
beginning of its work the commission established rules very
similar to those of the American Council on Pharmacy and
Chemistry. It listed the articles advertised in German medical
journals in three groups: (1) those which conformed to
the rules of the commission in the method of advertising;
(2) those which violated the rules, and (3) those whose
classification could not be determined. This amounted to an
attack on advertising in medical journals and was undoubtedly
premature. It aroused at once the antagonism not only
of the proprietary interests but also of the medical press.
“The establishment of the lists of medicines encountered
opposition or hindrance from three sources,” says Heubner,
“first, from the pharmaceutical and chemical manufacturing
interests; second, from the medical press, and third, from the
medical profession itself. The ‘trade’ naturally was irritated
at any attempt to interfere with ‘business,’ and brought forward
a number of reasons why the procedure adopted by the
commission was especially calculated to injure the ‘general
welfare.’ This opposition was to be expected and might be
disregarded. The extent to which the medical press was
dependent on the drug trade, however, had not been foreseen.
The same journals in which for many years all sorts
of articles on the evils in the trade in medicines had appeared
showed themselves decidedly cool or emphatically critical
toward the accomplished fact of the ‘lists of remedies.’ In
hastily written articles a whole series of mistakes in general
and in particular were published.... One thing,
however, was not explicitly stated—namely, that in any event
the lists of remedies must be rejected, and for this the
cogent reason was anxiety in regard to advertisements. The
editors had been sufficiently warned. The Therapeutische
Monatshefte, which had not submitted to the wish of a great
industrial firm in another matter, was punished for this
offense by the withdrawal of all its advertisements. None
of the other publishers wanted to risk such a reduction in
income, and none of the editors was willing to undertake the
risk to the extent of a conflict with his publisher. Curiously,
the idea does not seem to have arisen that if the threatened
publishers had made common cause they might have freed
their editors from the distressing burden of improper advertisements
with scarcely any risk at all.”
Heubner believes that another motive influencing the editors
was the fact that their efforts in behalf of reform, sporadic
and ineffective at the best, had been replaced by the
propaganda of the commission. It seems clear that the opposition
from the press was due not to principle chiefly but to
financial pressure. The editors, however unworthy their
motives, nevertheless exerted, as in other cases, a powerful
influence on public opinion. Among the medical public, opposition
was encountered because many physicians were interested—sometimes
financially—in one or more of the discredited
remedies. The mass of the profession either were
not interested or misunderstood the position of the council.
Despite the obstacles encountered and the difficulties
involved, the council and the Congress of Internal Medicine
have not wavered. Heubner, however, sums up the work of
the council in a rather pessimistic tone, as follows:
“What are the results of the great amount of labor, self-sacrifice,
hopeful courage and wasted money? Two journals
pretend to be doing wonders in that they are eliminating
some of the worst misstatements, distortions, obscurations
and concealments of truth in the advertisements. Physicians
at certain intervals receive lists of preparations, the manufacturers
of which as a rule do not need to pay any attention
to the council because their dealings are directly with the
public, because their advertisements are usually made to physicians
by word of mouth, or their preparations have already
a sufficient reputation—no matter for what reason....
“There is little doubt that the results have not paid for
the efforts expended. There is no doubt that the whole enterprise
will amount to nothing more than a splash in the water
if the work is not extended, just as a preliminary skirmish
must remain without effect unless followed up by the main
army. The main army in this case is the German medical
profession. However gratifying the progressive attitude of
some individuals and, in fact, of some associations, such as
that of Wurtemburg, may be, the fact remains that the profession
[in Germany] is not advancing but rather tends to
retrograde. The support which the executive committee of
the Aerztevereinsbund at first accorded to the efforts of the
council was later limited. All further progress depends on
the developments of the near future. Will sufficient power
be given to the German medical profession after settlement
with the insurance societies to permit them to follow the
example of their American colleagues?
“It should be made perfectly clear,” Heubner insists, “that
we are concerned with questions of importance for the standing
and influence of the medical profession among the people,
and, consequently, for the conditions of its future existence.
But even now the consequences of the prevailing indifference
to the traffic in nostrums are making themselves felt. The
prevalence of self-medication, which was lately recognized by
a Berlin court as the normal for ‘slight’ affections and which
has already been made an argument against the extension of
the compulsory prescription law, is merely a result of the
great evil based on the loss of control by the medical profession
of the remedies it employs. Only centralized and
energetic measures on the part of the organized profession
can secure a reformation of the intolerable conditions that
prevail in the field of modern industry in medicine and foodstuffs.
The American Medical Association and the German
Arzneimittelkommission have shown that a little sacrifice
and energy can secure a condition in which the medical profession
becomes a powerful factor, able to dictate in the field
of the trade in medicine instead of letting itself be dictated
to.”—(From The Journal A. M. A., April 18, 1914.)
GRAND PRIX AND GOLD MEDALS FOR SALE
Max Kaiser Offers to Procure “Awards of Merit” at Various
International Exhibitions—Price, Four
Hundred Dollars
There was a time when the manufacturer who could point
to the “Grand Prix” or the “Gold Medal” his product had
been awarded at some exhibition was considered to have a
valuable advertising asset. Possibly there was a time when
medals and prizes were awarded with an eye single to the
excellencies of the goods and bore no relation to the amount
of money paid by exhibitors to the organizers of the exhibition.
Possibly there are, even today, occasional awards
made on a basis of pure merit, but they are probably few
and far between. The matter which follows throws an interesting
light on this subject. Within the past two months
manufacturers have received a letter on the stationery of the
“International Exhibition, Paris, 1914.” The letter came from
the “Commissioner-General” of the exhibition, one Max
Kaiser, 24 Holborn, London, E. C. Here it is:
“Dear Sirs:—I beg to draw your attention to the great INTERNATIONAL
EXHIBITION of Alimentation, Pure Food, Hygiene, Beverages, Drugs
and allied trades, which will be held at Paris in March, 1914, inviting
you to partake with your manufacture at this INTERNATIONAL EXHIBIT. I
beg to point out that the aim of this Exhibition is to introduce Foreign
Manufactured Goods, Proprietary Articles, Patents, etc., to the French
and Foreign Markets, and to open up or extend new channels for
such goods.
“A Commercial Office at the Exhibition Building, under the Commissioner
General’s own supervision, with a well-trained staff, will do
everything required in the interest of Exhibitors, such as effect sales
by circularizing, or inviting prominent buyers to call at your particular
stall to judge for themselves the merits of your Exhibit, and
in this way bring the American Manufacturers in direct touch with
the Foreign Markets and the Buying Public.
“This Commercial Office will also negotiate with the Representatives
on your behalf: at the same time undertaking to arrange your Exhibit,
supply all necessary fittings, decoration, the display, maintenance, repacking
and returning of the Exhibit, and also to represent you before the
Public and Jury in such a manner as to make certain that your Exhibit
shall be awarded first honors (GRAND PRIZE OR GOLD MEDAL).
“You will understand that such an award obtained at this INTERNATIONAL
EXHIBITION means an everlasting advertisement as an official
acknowledgment and convincing proof to the Superior Quality of your
goods, and will certainly put you in front of your competitors on the
home market, and naturally increase your sales considerably.
“I might mention here that many a big business has been built up
and small concerns been prominently brought to the notice of the
Public by Exhibiting. In many cases I have been able to interest
Authorities and Reigning Houses in Exhibits under my care, and I
have opened up or extended markets for firms Exhibiting under my
direction.
“I enclose herewith a list containing some of the most prominent
American and English Firms whom I have represented at European
Exhibitions and for whom I achieved splendid results.
“I could arrange your Exhibit for the amount of $400, to be paid
one-half on allotment and the balance on receipt of an award (Grand
Prize or Gold Medal).
“Trusting that this will be of interest to you, I shall be pleased to
receive your reply by return mail, and give you any further particulars
you may desire. Yours faithfully,
The Commissioner General.
(Signed) Max Kaiser.
“I can also accept Exhibits on exactly the same terms for the INTERNATIONAL
EXHIBITION, Rome, 1914.”

Photographic reproduction (reduced) of the letter-head of the stationery
on which Max Kaiser offers to “make certain” that those who
exhibit their products under his direction—price, $400—shall receive
a Grand Prix or Gold Medal.
The list Mr. Kaiser enclosed with his letter was a printed
sheet, giving the names of a number of American and British
manufacturers whom Kaiser claims to have “represented” at
various “International Exhibitions.” The majority of the concerns
named are breweries, but there is a good sprinkling of
“patent medicine” companies and a few miscellaneous manufacturers.
In the American list there are two nostrum concerns
named that will be more or less familiar to our readers.
They are:
Alonzo O. Bliss Company, Washington, D. C. This company
sells “the Great Blood Purifier, Kidney and Liver Regulator”
known as “Bliss’ Native Herbs.” According to Max
Kaiser, the Alonzo O. Bliss Company obtained one Grand
Prize and one Gold Medal.
Waterbury Chemical Company, Des Moines, Iowa. This
company exploits what used to be known as “Waterbury’s
Cod-Liver Oil Compound,” which, from its lack of cod-liver
oil,152 was impelled to change its name to “Waterbury’s Compound.”
Kaiser states that the Waterbury Chemical Company
received four Grand Prix and four Gold Medals.
Briefly the proposition submitted by Max Kaiser is this:
For $400 he will make all arrangements for a manufacturers’
exhibit at one of the numerous “International Exhibitions.”
Further, he practically guarantees that this exhibit will
receive either a “grand prize” or a “gold medal”; in fact, the
manufacturer need not complete the payment of Kaiser’s
charges until the prize or medal has been awarded!
The value of “awards” obtained in this way is, of course,
evident. As the public becomes better informed on the subject
of international exhibitions, the grand prix, gold medals,
and other “awards” made at such exhibitions will be appraised
at their true value.—(From The Journal A. M. A., March
14, 1914.)
THE HYPOPHOSPHITE FALLACY
An Example of the Perpetuation of a False Theory
by Advertising
A false therapeutic notion born of speculation soon dies a
natural death if exposed unsupported to the cold world of
facts, but when nursed by commercial interests it may be
kept alive for generations. Interesting examples of this, to
name but two or three, are the misconceptions perpetuated
during the past half century concerning “lithia,” the “natural”
salicylates and the hypophosphites.
Take, for instance, the lithia delusion. The supposed solvent
powers of lithium compounds for uric acid were soon
disproved to the satisfaction of scientists, but proprietors of
lithia waters and nostrums for gout and rheumatism still
harp on the old string and utilize long-exploded theories.
Take, again, the alleged superiority of “natural” to “synthetic”
salicylates. In spite of experimental proof to the contrary,
proprietary interests have been able for twenty years
to persuade a large part of the medical profession that the
effects of pure salicylic acid made artificially differ from the
effects of the same substance obtained from natural sources.
The altogether undeserved continued popularity of hypophosphites
affords a striking example of the influence of
advertising in perpetuating therapeutic error, for hypophosphites
are given on a theory long since disproved. It may
be interesting to trace the origin and history of the theory
on which the practice of prescribing the hypophosphites is
founded. The early part of the last century was prolific in
chemical discoveries, and, as a corollary, in chemical theories
of disease. Many of the theories arose from the hasty application
of the chemical properties of new elements and compounds
to the explanation of the processes in the living body,
without due consideration of the conditions prevailing in the
animal organism.
THE ELEMENT PHOSPHORUS
The element phosphorus is eager for oxygen and readily
oxidizable. When taken into the system it acts as a violent
poison, and, in view of this, it was at first supposed—although
the supposition was based on no scientific data—that it
would prove to be a powerful therapeutic agent when given
in minute doses. In its elementary form, phosphorus is difficult
to handle, and therefore not convenient for use. Hence
it was natural that a compound should be sought which could
be used as a substitute for the element.
Broadly speaking, phosphorus forms three classes of salts
varying in the degree of oxidation: the phosphates, containing
the most oxygen, the phosphites, containing less, and the
hypophosphites, least of the three. The phosphates, being
saturated with oxygen, undergo little change in the body,
and because of this were thought to be of little value in
therapeutics. The phosphites contain less oxygen, are unstable
and are not used in medicine. The hypophosphites, containing
still less oxygen, stand nearest to elementary phosphorus
and are easily decomposed and readily oxidized to
phosphates. Hence the theory that the hypophosphites would
furnish an admirable source from which to obtain the action
of the element phosphorus.
CHURCHILL’S THEORY
The hypophosphites were introduced into medicine about
1855, as a substitute for elementary phosphorus by a Dr.
Churchill of Paris, and later of London, who advocated their
use as a specific remedy for consumption. Churchill conceived
the theory that phthisis is caused by a lack of oxygen
in the tissues; he therefore sought an agent capable of increasing
oxidation. He was led to the use of hypophosphites for
this purpose on the supposition that phosphorus exists in the
organism as a biologic element in a lower degree of oxidation
than the phosphate. He supposed that this form of phosphorus
acts by its chemical affinity as an initiatory agent in
attracting and utilizing the inspired oxygen. He believed that
when this form of phosphorus, which he called the “phosphide
element,” is deficient in quantity (because it had been oxidized
into phosphate, or because the supply from natural sources
was deficient), the degree of oxidation of the tissues is less
than normal. Therefore he advocated the use of hypophosphites
to supply the lacking oxidizing constituent. He
believed this “phosphide element” not only to be essential
for the oxidation of the tissues, but also to be the source
of energy of the nervous system.
THE FACTS
The theory was a pretty one; the facts, however, did not
support it. Subsequent investigations indicate that instead
of consumption being due to a lack of oxygen, there is in
that disease really an increased oxidation; in other words,
the respiratory exchanges in this disease are exaggerated.
The existence in the system of a form of phosphorus less
highly oxidized than the phosphates is unproved. No evidence
has been produced to show that phosphorus acts as an energizer
of oxidation. There is no proof that the hypophosphites
enter into general metabolism or affect disease processes
in any way. Not only is there no scientific evidence for
the utility of the hypophosphites, but science has long since
demonstrated their worthlessness.
In 1895 Boddaert153 published researches showing that hypophosphites
are rapidly eliminated through the kidneys
unchanged. Similar results have been reached by Paquelin
and Joly, who attributed to the hypophosphites only the action
of diuretics. In 1901 Massol and Gamel154 found by animal
experimentation that the hypophosphites did not act as diuretics,
but that the hypophosphorous acid was completely
eliminated in the form of sodium hypophosphite. The urea
was not increased and the relation of urea to total nitrogen
remained the same. Their results indicated no increase of
oxidizing actions within the system. Finally, Massol and
Gamel examined the urine of patients taking hypophosphites
and found the same conditions: the results were the same as
in the experiments on animals.
PROPRIETARY THERAPEUTICS
In spite of these facts the hypophosphites continue to be
employed by many practitioners. Why? Because the theory,
being plausible at the time when such chemical theories
were popular, gained a certain recognition and was accepted
without scientific investigation. Thus the hypophosphites
came into use. It was not long before they were taken up
by certain manufacturers, and the theory on which their
use was based became a commercial asset. As a result the
theory, which uncommercialized would have died of inanition,
was kept alive by continued advertisement.
The manufacturer of proprietaries having settled on a
plausible theory on which to sell his products has no further
need for science. Thus, while these theories are no longer
to be found in accredited text-books, they are still preached
by the proprietary interests. An elaborate pamphlet on
“Iodine and Phosphorus,” containing statements which are
known to be false, is one firm’s text-book supplied to physicians
to-day, and contains long quotations from Dr. Churchill’s
writings of sixty years ago. This book contains no intimation
that these theories have been overthrown. It is poor
economy to waste money in changing literature when the old
theories and the old plausible reasoning will sell goods just as
well. Consequently the old errors are drummed into those
physicians who are willing to read their physiology from the
neat monographs of proprietary literature and to sit at the
feet of glib salesmen who expound to them the proprietary
theory of therapeutics.—(From The Journal A. M. A., April
25, 1914.)
BUFFALO LITHIA WATER
Contains One-Fifth as Much Lithium as Potomac
River Water
Some years ago, Alexander Haig evolved the theory that
most diseases are due to uric acid. The data on which he
founded his theory were not corroborated by scientific men,
and investigation showed that his methods were unreliable.
In spite of the fact that Haig’s theories are utterly
discredited, and have been for years, the uric acid fallacy
still persists, although it is now largely confined to the public.
Shrewd business men, especially those who are more
intent on making money than they are concerned with
the manner in which that money is made, owe much to
Haig’s theory. As a business proposition, uric acid has been
one of the best-paying fallacies on the market—and possibly
still is. It is only necessary to refer to The Journal’s
recent article155 on the Turnock mail-order medical fraud to
emphasize this fact.

Showing how “Buffalo Lithia Water” in the course of time became
“Buffalo Lithia Springs Water.” The government has shown that, to
obtain a therapeutic dose of lithium from Buffalo Lithia Springs Water,
it would be necessary to drink 200,000 gallons of the water. The
government also declared that Potomac River water contained five times
as much lithium as does Buffalo Lithia Springs Water.
Contemporary with, and to a certain extent a corollary of,
the uric acid fallacy was another, viz., that lithium would
eliminate uric acid. This at once gave a good working
principle for the proprietary men. Uric acid, we were told,
causes disease; lithium, we were also told, would eliminate
uric acid; therefore, lithium is the new elixir of life! Could
anything be simpler?
Accepting this theory, it was inevitable that mineral waters
containing lithium salts should become highly popular.
Many exploiters of mineral waters began to place most
emphasis on the lithium salts in their waters even in those
cases in which lithium was present in such infinitesimal
amounts as to render its detection impossible by any but
spectroscopic methods.
One of the best known, because most widely advertised,
of the so-called lithia waters is Buffalo Lithia Water—or
what used to be called Buffalo Lithia Water. After the
Federal Food and Drugs Act came into effect, by which
falsification on the label was penalized, the name of Buffalo
Lithia Water was changed to Buffalo Lithia Springs Water.
The reason for this change was that when Buffalo Lithia
Water was subjected to examination by the government
chemists it was found to contain so little lithium that the
amount present was unweighable—it could be demonstrated
only by the spectroscope. It was evidently, therefore, not
a lithia water in that it did not contain—at least in quantities
that could be consumed—an amount of lithium that would
give the therapeutic effects of lithium: Possibly the company
imagined that by changing the name from “Buffalo Lithia
Water” to “Buffalo Lithia Springs Water” it had cleverly
evaded the federal law. Their argument was to this effect:
The springs from which this water is taken are known as
Buffalo Lithia Springs; therefore, it is not a misstatement of
facts to call this Buffalo Lithia Springs Water.
WHAT IS A LITHIA WATER?
The Supreme Court of the District of Columbia, holding
a district court, has recently given an opinion on the Buffalo
Lithia Springs Water case. The findings of the court are
refreshingly simple, and characterized by that broad commonsense
view that is becoming increasingly more common among
modern jurists. Read Judge Gould’s opinion as to what
constitutes a lithia water:
“Speaking generally, and as an individual of average
intelligence and information, it would seem that if one were
offered a water which the vendor told him was a ‘lithia’
water, one would have the right to expect enough lithium in
the water to justify its characterization as such, thus differentiating
it from ordinary potable water; and this amount
would reasonably be expected to have some effect on the
consumer of the water by reason of the presence of the
lithium.”
Certainly a reasonable attitude, and one which the man in
the street not only can understand but will agree with. Then
came the question as to the actual lithium content of Buffalo
Lithia Springs Water, and the court said:
“For a person to obtain a therapeutic dose of lithium by
drinking Buffalo Lithia Water he would have to drink from
one hundred and fifty thousand to two hundred and twenty-five
thousand gallons of water per day. It was further
testified, without contradiction, that Potomac River water
contains five times as much lithium per gallon as the water
in controversy.”
SOME TESTIMONIALS
Here, then, is a water that has for years been advertised
first, in medical journals, and later, in lay publications, as a
“lithia water” yet, actually, it contains less lithium, five to
one, than is to be found in ordinary river water. This is a
point for physicians to ponder well over. Turn to the back
volumes of medical journals and read, both in the advertising
and reading pages, the elaborate testimonials, given by
men high in the medical profession, on the marvelous effects
obtained by the use of Buffalo Lithia Water. Read the
following in light of the fact that the water from the Potomac
River contains five times as much lithium as Buffalo
Lithia Water:
“In the class of cases in which lithia, soda and potash are regarded
as most specially indicated, I have obtained far better results from the
Buffalo Lithia Waters than from any of the preparations of the lithium
salts of the Pharmacopeia.” (Statement by a member of the Faculty
of Medicine of Paris, France, etc.)
“Its [Buffalo Lithia Water] therapeutic effects, in my practice, have
been vastly superior to those obtained from Lithia Tablets or other
Lithia preparations.” (Statement by an ex-president of the University
College of Medicine, Richmond, Va., etc.)
“It [Buffalo Lithia Water] is strikingly superior to emergency solutions
of lithia tablets and pure water, even where the said solution is
an exceedingly strong one.” (Statement by a former Professor of Clinical
Medicine of the College of Physicians and Surgeons, New York,
and vice-president of the American Medical Association, etc.)
“When Lithia is indicated, I prescribe Buffalo Lithia Water in
preference to the Salts of Lithia, because it is therapeutically superior
to laboratory preparations of Lithia.” (Statement by a former professor
in the Medical College of Virginia and ex-president of the Medical
Society of Virginia, etc.)
“Buffalo Lithia Water ... by its richness of composition of
Lithia, is of marvelous efficacy, in cases of gout, of chronic, articular,
and muscular rheumatism ...” etc. (Statement by former Physician
in Ordinary to the Pope; Member of Academy of Rome, etc.)
“I have tried carbonate of lithia dissolved in water in various proportions;
but it certainly does not have the same effect as Buffalo Lithia
Water.” (Statement by a former Surgeon-General of the U. S. Army,
etc.)
These are but a few of many testimonials from physicians
that might be quoted. They are interesting from many
points of view. They show the worthlessness of testimony
of this sort—no matter from what source—and the fallacy of
that based on so-called clinical evidence.
To go back to the court’s findings: In the case of the
government against Buffalo Lithia Springs Water, one other
judicial opinion is worthy of attention, that referring to the
attempt on the part of the exploiters of the water to circumvent,
on a technicality, the evident intent of the Food and
Drugs Act. Said Judge Gould:
“The argument seems to be that if Buffalo Lithia Springs
are falsely named, being called ‘Lithia’ Springs, when they
do not flow water containing lithium, therefore the proprietors
have the right to sell the product as being Buffalo Lithia
Springs Water, thus perpetuating on the public the misnomer
connected with the origin of the water. It is not apparent
how the deceit practiced on the public by the label is mitigated
by carrying it back to the designation of the spring
from which the water comes.”
For years no one, apparently, ever criticized the claims
made for this product. Finally, we got the Food and Drugs
Act and the federal officials, acting under the authority
vested in them by that law, in December, 1910, declared
Buffalo Lithia Water misbranded. Thus this old established
vested interest was attacked. The company, of course,
fought. It first demurred to the charge brought, and in
April, 1912, the demurrer was sustained. At the same time
an amended libel was filed by the government, which the
company again demurred to. This demurrer was overruled
in June, 1912, whereon the company in December, 1912, filed
an answer denying that the water was misbranded. The
question has now (1914) been finally decided by the court
sitting as a jury, the matter having been submitted by agreement
to the court.
Buffalo Lithia Water has been sold since 1878. During
this period undoubtedly many physicians have prescribed
enormous quantities of this water, and many more laymen
have taken the water on their own initiative, based on the
advertised claims made for it. Practically all who purchased
the water, whether directly or on the advice of physicians, did
so in the belief that they were getting lithium. Had they
known that, to get a therapeutic dose of lithium they would
have had to drink 200,000 gallons of Buffalo Lithia Water,
they would have felt, and rightly so, that they were the
victims of an expensive hoax.—(From The Journal A. M. A.,
June 13, 1914.)
MEAT EXTRACTS AND MEAT JUICES[AU]
Their Composition and Relative Values
The Bureau of Chemistry of the Department of Agriculture
has recently given in Bulletin No. 114 much new and
valuable data regarding the commercial meat products. The
work contained in this bulletin is practically an elaboration
or continuation of that published in The Journal of May 11,
1907, p. 1612. It was taken up to determine the condition
and quality of meat preparations in general and from the
results obtained to prepare tentative standards for the preparation
and composition of such meat preparations. The
results as well as the methods of analysis of many meat
products are given, showing the composition and relative value
of the various preparations. The comments of many investigators
regarding the food value of such products is also a
valuable contribution to the knowledge of meat extracts, and
will help in deciding the real value of the preparations.
The preparations taken up are divided into three general
classes (1) Solid and Fluid Meat Extracts; (2) Meat Juices;
(3) Miscellaneous Preparations. For each of these the tentative
standards submitted by the Committee on Food Standards
of the Association of Official Agricultural Chemists are
given along with the tabulated results of the chemical analysis.
The preparations examined showed, for the most part,
that they conformed to the standards, and only those which
are at variance in one or more particulars will be mentioned
in this review.
SOLID MEAT EXTRACTS
For solid meat extracts the following are the requirements:
“Meat extract is the product obtained by extracting meat
with boiling water and concentrating the liquid portion by
evaporation after removal of fat, and contains not less than
75 per cent. total solids of which not over 27 per cent. is ash
and not over 12 per cent. is sodium chlorid (calculated from
the total chlorin present), not over 0.6 per cent. is fat and
not less than 7 per cent. is nitrogen. The nitrogenous compounds
contain not less than 40 per cent. of meat bases and
not less than 10 per cent. of kreatin.”
With the above as the standard, several of the solid meat
extract preparations examined were not up to grade on one
or more points, though in some cases it is true they were
very slightly below the standard set. The following products
were found wanting in some respects and the requirements
which they failed to meet are given:
“Rex” Brand Beef Extract (Cudahy Packing Co., Omaha)
contained 26.50 per cent. water instead of the standard 25
per cent.
Extract of Beef Premier (Libby, McNeil & Libby, Chicago)
contained 30.92 per cent. of ash instead of the standard
27 per cent.; 18.32 per cent. of sodium chlorid (standard, 12
per cent.); 6.02 nitrogen (standard, 7 per cent.).
Beef Extract (Swift & Co., Chicago) contained 13.51 per
cent. sodium chlorid (standard, 12 per cent.); 6.60 per cent.
nitrogen (standard, 7 per cent.).
Beef Extract, Coin Special (G. H. Hammond Co., Hammond,
Ind.) contains 13.25 per cent. of sodium chlorid (standard,
12 per cent.); and 6.86 per cent. nitrogen (standard, 7
per cent.).
With these few exceptions, the solid meat extracts were
found to comply with the standards given.
FLUID MEAT EXTRACTS
For fluid meat extract the following standards have been
suggested:
“Fluid meat extract is identical with meat extract except
that it is concentrated to a lower degree and contains not
more than 75 per cent. and not less than 50 per cent. of total
solids.”
According to this standard all excepting one of the fluid
meat extracts examined were found to be below grade in one
respect, that of solids. The following are preparations examined
and the percentage of solids found:
| Per cent. |
Concentrated Fluid Beef Extract (Armour & Co., Chicago) | 42. | 25 |
Meat Juice (Valentine’s Meat Juice Co., Richmond, Va.) | 42. | 36 |
Beef Juice (John Wyeth & Bro., Philadelphia) | 41. | 16 |
Vigoral (Armour & Co., Chicago) | 50. | 06 |
“Rex” Fluid Beef Extract (Cudahy Packing Co., Omaha) | 44. | 01 |
Fluid Extract of Beef (Cibilis Co., New York) | 35. | 37 |
Fluid Beef Jelly (Mosquera-Julia Food Co., Detroit) | 31. | 03 |
Special notice is directed to the price of some of these preparations,
which in spite of their large water content, are
higher priced than some of the solid meat extracts.
MEAT JUICES
The following is given as the standard for preparations of
meat juice:
“Meat juice ... is the fluid portion of muscle fiber
obtained by pressure or otherwise, and may be concentrated
by evaporation at a temperature below the coagulating point
of the soluble proteids. The solids contain not more than 15
per cent. of ash, not more than 2.5 per cent. of sodium chlorid
(calculated from the total chlorin present), not more than 4
per cent. nor less than 2 per cent. of phosphoric acid (P2O5),
and not less than 12 per cent. of nitrogen. The nitrogenous
bodies contain not less than 35 per cent. of coagulable proteids
and not more than 40 per cent. of meat bases.”
It is especially noticeable among the meat juices, so called,
that none shows any appreciable amount of coagulable proteids.
Valentine’s Meat Juice and Wyeth’s Beef Juice, besides
being below the standard in total solids as fluid extracts,
are misbranded when called meat or beef juices, as can readily
be seen by comparing the results of the analyses and the
standard.
Wyeth’s Beef Juice is advertised as containing “all the
albuminous principles of beef in an active and soluble form”
and “in an unaltered form”—two statements that are on the
face of them untrue and misleading. To say that all the
albuminous principles of meat are present is to say that not
only the juice of the meat but all the fiber is present, which
evidently is not true. Then again, to say that it is present
in an unaltered form is far from the facts, for, as is stated
on page 18 of the Bulletin: “It appears impracticable to prepare
a true meat juice for market, as the temperature necessary
for the preservation of food products in hermetically
sealed packages coagulates the proteids and changes the
nature of the product.” On page 55: “When prepared under
the best possible conditions a commercial meat extract is, of
necessity, in order that it may not spoil, deprived of the
greater part of the coagulable proteids, which constitute the
chief nutritious elements of the juice.”
On examining the tables of analysis, it is seen that Wyeth’s
Beef Juice contains but 23 per cent. of its total proteids in a
coagulable form, while the standard calls for 35 per cent.,
thus showing it to be no more valuable as a food product
than any other so-called meat juice, the statements of the
manufacturers to the contrary notwithstanding.
In the case of Valentine’s Meat Juice we note a large discrepancy
between the standard requirements and the results
of the government analysis, for instead of the proteid matter
containing 35 per cent. in the coagulable form, it contains
but 1.6 per cent. These figures show, then, that Valentine’s
preparation contains practically no coagulable proteids, and
since the quantity of these measures the food value of such
preparations, the conclusion must be drawn that Valentine’s
Meat Juice has practically no value as a food and should
certainly not be classed as a meat juice.
Bovinine, another widely advertised meat preparation,
which, according to statements on “The Bovinine Co.’s” letter
head, is “a concentrated beef juice” and “the only perfect food
in the world” was analyzed and found below the standard
set for meat juices, since it contains only 3.38 per cent. of
coagulable proteids. Yet in spite of this discrepancy, the
manufacturers of Bovinine persist in exploiting it as a food,
stating it to be “... a concentrated easily assimilable,
nitrogenous food,” and in another place it is stated that
Bovinine “is an ideal food.” As it is deficient in coagulable
proteids and thus below the requirements as a food, it is
misbranded when called a food of any sort, for to quote
again the Bulletin, page 55: “... meat extracts ... must
not be looked on as representing in any notable degree
the food value of the beef or other meat from which they are
derived”; and, again: “They are not, however, concentrated
foods, having, on the contrary, but comparatively little
nutritive value.”
Taken individually or as a class, meat extracts are not to
be considered foods, and should, therefore, not be advertised
as such, a conclusion which the government officials
have come to and voiced in the conclusion of the Bulletin as
follows:
VALUE AND LIMITATIONS
“It seems to be the consensus of opinion among scientific
investigators who have studied this question that the food
value of these meat extracts is rather limited, and although
they are a source of energy to the body they must not be
looked on as representing in any notable degree the food value
of the beef or other meat from which they are derived. When
prepared under the best possible conditions a commercial
meat extract is of necessity, in order that it may not spoil,
deprived of the greater part of the coagulable proteids, which
constitute the chief nutritious elements of the juice.”—(From
The Journal A. M. A., Jan. 23, 1908.)
PHARMACEUTICAL MANUFACTURERS AND THE
GREAT AMERICAN FRAUD
At various times we have given more or less complete
accounts of the prosecutions the United States Government
has brought against nostrum exploiters under the Food and
Drugs Act. One of the more recent of these, while of comparatively
little interest per se, is of importance to the medical
profession, because of certain elements connected with it.
The case is known technically as “Notice of Judgment No.
284” and deals with the “Alleged Misbranding of Danderine.”
The gist of the case is as follows: Casks of Danderine—a
widely advertised “hair tonic”—were shipped in carload lots
from Michigan to West Virginia, where the product was
bottled, labeled and put in condition to be retailed. Danderine
contains a percentage of alcohol which, while given on
the labels of the bottles in which it is sold, was not stated on
the casks in which the preparation was shipped in bulk. The
government sought to confiscate, under the Food and Drugs
Act, sixty-five casks thus shipped because the quantity or
proportion of alcohol in the casks was not stated. The Knowlton
Danderine Company resisted the confiscation and the
court upheld the company’s claim.
The point in this case which is—or should be—of interest to
the medical profession is to be found in the “statement of
facts” presented by the Knowlton Danderine Company in its
own defense. Here it is said that: “Parke, Davis & Co.,
who are mentioned in the said libel as shippers ... are
under contract with the said Knowlton Danderine Company ... to
compound the said formula ...” Elsewhere it
is stated: “Parke, Davis & Co. were ... the manufacturing
agents, under contract, of the owner, the Danderine
Company ...”
This evidently means that Parke, Davis & Co., who are
generally supposed to manufacture only “ethical” preparations—proprietary
or otherwise—and as such to desire the
respect and good wishes of the medical profession, are in the
business of furnishing the supplies for nostrum venders.
What Danderine is, it is hardly necessary to specify. The
widely distributed advertisements of this “hair tonic” nostrum
with the slogan: “Danderine Grows Hair and We Can Prove
It” are sufficiently well-known to all who read to make a
lengthy disquisition on the product unnecessary.
It is interesting in this connection to note that according
to newspaper dispatches the Danderine Company has absorbed
the Sterling Remedy Company, which exploits “Cascarets.”
Three years ago a physician, who is also a pharmacist, wrote
to the Medical World regarding the manufacture of Cascarets:
“... I have positive evidence, which I will gladly submit,
that P., D. & Co., make all of them [Cascarets], and
that they have a contract with the Cascaret people not to
make anything similar for any one else.”
In the circular which comes in the Danderine packages two
other “specialties” are advertised: “Neuralgine” for “sick,
weak, tired nerves” and “Drake’s Palmetto Compound” for
“weak stomachs, sluggish livers, disordered kidneys,” and
various other derangements of the system. The question naturally
arises, are these, too, shipped in casks from Parke,
Davis & Co., and merely bottled and labelled in West
Virginia?
Not that the Danderine case is the first one in which Parke,
Davis & Co. have been exposed as manufacturers of nostrum
supplies. “Vitaopathy” a method of “treatment” practiced by
the notorious New York Institute of Physicians and Surgeons
in the person of “Prof.” Adkin and apparently consisting
of “absent treatment” and pills, was finally put out of
business by a fraud-order from the post office department.
The concern used to advertise:
“In Professor Adkin’s laboratory, his chemists are daily engaged in
extracting the life-and-health-giving principle from rare vegetables, fruits
and plants.”
“Prof.” Adkin had no laboratory; his chemists, according
to the government report, were Parke, Davis & Co., from
whom he purchased the tablets which formed part of his
stock-in-trade of quackery.
The Nutriola Company of Chicago was declared fraudulent
by the postal authorities and a full account of the methods
of this fake medical concern appeared in The Journal, April
28, 1906. Nutriola was advertised as:
“The greatest Chemical-Medical Preparation ever prepared by the
skill of man.”
“Nutriola and Nature are the only invincible conquerors of diseases
ever known.”
The promoter of this scheme was one Edward F. Hanson,
who was questioned by the government inspectors regarding
the manufacture of the Nutriola nostrum. Quoting from the
government report:
“Q. Please name the chemists who now manufacture the remedies
of the Nutriola Company.”
“A. Parke, Davis & Company, Detroit; E. L. Patch Manufacturing
Company, Stoneham, Mass.; Seabury & Johnson, New York.”
Not that the course pursued by Parke, Davis & Co. is by
any means an exceptional one in the pharmaceutical world.
It may be recalled that The Journal has previously referred
to the fact that Sharp & Dohme are reported to make or to
have made the “Getwell Tablets” for the “patent medicine”
concern which exploits the nostrum; and that Frederick
Stearns & Co. make or did make the widely advertised “cures”
Shac and Zymole Trokeys also has been mentioned. That
Seabury & Johnson made preparations for a fake medicine
concern was brought to light by Mr. Adams in the “Great
American Fraud” series. And unquestionably there are many
others. The attitude taken by such houses seems to be that
they are willing to furnish anything in the pharmaceutical
line that anyone is willing to pay for, whether it is for legitimate
use of the physician or pharmacist or for furthering
the business by which the ignorant or gullible sick are humbugged
and defrauded.—(From The Journal A. M. A., July
2, 1910.)
DOWD’S PHOSPHATOMETER
Dowd’s Phosphatometer, according to its inventor, is a
device “for taking the phosphatic index or pulse of the nervous
system.” Its originator, J. Henry Dowd, M.D., Buffalo,
N. Y., writes enthusiastically of his instrument:
“Physicians who use the Phosphatometer are sending 50 per cent.
less patients away for consultation, getting 75 per cent. better results
at home, because the Phosphatometer tells the cause and what to
do, and the Comp. Phosphorus Tonic gives results in 80 per cent.
of all conditions of illness.”
The “Comp. Phosphorus Tonic” referred to in the above
quotation is a sideline of Dr. Dowd’s, put out by the
Richardson Company, of Buffalo. The stationery of the
Richardson Company gives its address as 334 Franklin Street,
but directs that all communications be addressed to 40 North
Pearl Street, which is the private address of J. Henry Dowd.
According to the Buffalo directories, 334 Franklin Street is
the drug store of Arthur E. Reimann.
To those who read the Dowd “literature,” the Phosphatometer
will appear to be either one of the wonders of the age
or an unscientific absurdity. To the thinking man it will be
the latter. It pretends to determine the amount of phosphates
in the system. This is accomplished—alleged—by
taking the second urine passed in the morning and mixing a
portion of it in the instrument with a solution which is the
well-known magnesia mixture. The height to which the crystals
settle in ten minutes determines, according to Dowd, the
amount of phosphates! Was ever a test devised that violated
more of the first principles of quantitative chemical analysis?
If so, we never heard of it. Dowd’s system does not require
any determination of the amount of urine passed in twenty-four
hours or even of the amount passed at the second micturition
in the morning.
If a patient whose urine was being “tested” by the Dowd
method, should drink two cups of coffee for breakfast instead
of one, his urine might be so dilute that the phosphates
would fall below the “normal” mark. Dowd says that his
Phosphatometer “takes the pulse of the nervous system.”
What about the patient who eats several eggs or consumes
a sweetbread or other nuclein-containing articles of diet?
The increased amount of phosphates in such a diet might
easily lead to an apparent excess in the urine. Every physician,
nay, every sophomore medical student, knows that the
amount and kind of food ingested governs almost entirely the
amount of phosphates excreted in the urine.
What actually does “Dowd’s Phosphatometer,” when used
according to instructions, show? It shows the presence of
phosphates in the urine; it permits a guess—with not the
slightest claim to accuracy—as to the amount in the specimen
tested; it gives no possible clue to the normal or abnormal
relation of the phosphates in the urine or as to whether the
source of the crystals precipitated is the nerve tissue or the
food. Yet here are some of the claims made for it:
“The Phosphatometer shows nervous metabolism the same as the
ureometer shows muscular; the former errs in about 3 per cent.; the
latter in 40 per cent.”
“The Phosphatometer shows the amount of nerve food being used
and present in the nerve cells.”
“Over 50 per cent. of pain and human suffering is due to the nerves
crying for food; the Phosphatometer will show the true cause in ten
minutes.”
“The Index not only tells the present condition, but foretells the
future, thus preventing serious complications which might arise.”
“The Phosphatometer measures the amount of phosphorus in the
system.”
“The Dowd Phosphatometer not only takes blood-pressure, it tells
how to regulate it.”
“The Phosphatometer measures the amount of phosphorus in the
nerve cells; it is as positive in nerve troubles as the x-ray in fractures.”
These claims are essentially false. As a matter of fact, a
simple examination of the urine for phosphates cannot tell us
the condition of the nervous system. This must be evident
from the fact that only a portion of the phosphates is excreted
in the urine, a very considerable part passing out with the
feces. Further, the bulk of the phosphorus excreted comes
from the food and only a small portion from the waste of
the nervous system. The amount excreted by the urine which
comes from torn-down nerve tissue is so small that it is practically
impossible to estimate variations in it even by the
most careful analytic methods.
In brief, Dowd’s “scientific method” is nothing more than
unscientific humbug.—(From The Journal A. M. A., Dec.
20, 1913.)
AMORPHOUS PHOSPHORUS[AV]
A Practically Inert Substance Introduced as a Valuable
Therapeutic Agent
Amorphous phosphorus is a chemical anomaly contrasting
markedly with ordinary phosphorus in its physical, chemical
and pharmacologic properties. Ordinary phosphorus is soluble
in certain solvents, such as oil; amorphous phosphorus is
insoluble. Ordinary phosphorus is poisonous; amorphous
phosphorus is not poisonous. Ordinary phosphorus has been
regarded as of some therapeutic value; amorphous phosphorus,
because of its insolubility and other physical properties, has
never been so regarded. Pharmacologists, therefore, have
paid very little attention to it. Some of them do not even
mention it, though there are a few accounts of experimental
work.
Noé,156 in experiments on the action of phosphorus with
yeast, found that yeast acted on ordinary phosphorus, producing
PH3 (hydrogen phosphid), but on amorphous phosphorus
it had no action. His experiments show that
amorphous phosphorus was not toxic to animals.
Thornton157 quotes Reese as publishing a report of a case in
which 30 grains of amorphous phosphorus were taken by a
young woman with suicidal intent, but no toxic symptoms
were manifested. Thornton found it non-toxic to animals.
Witthaus and Becker (Medical Jurisprudence, Forensic
Medicine and Toxicology, iv, 635) say: “The form of phosphorus
is practically non-poisonous, probably by reason of its
insolubility. It has been administered to dogs to the extent
of 200 gm. (nearly half a pound) in twelve days without
causing poisoning.”
C. D. F. Phillips (Materia Medica, Pharmacology and Therapeutics,
Inorganic Substances, Ed. 3, p. 46) makes the following
statement: “Amorphous phosphorus has been, by some
observers, credited with physiologic activity. Thus, Bednar
used it for a long period in small doses, and observed symptoms
of excitation, trembling and clonic convulsions; but as
much as 1 ounce has been given to dogs without perceptible
effect. Thompson, in twelve carefully observed cases, found
its action nil, and plausibly attributes its supposed powers to
a slight admixture of ordinary phosphorus (Pharm. Jour.,
1875). I believe it is practically inert.”
HOW INTRODUCED
The foregoing represents our scientific knowledge as to the
action of amorphous phosphorus. Now, however, comes Dr.
I. L. Nascher and introduces amorphous phosphorus as a
remedy of remarkable value for the arteriosclerosis of
old age. The method of introduction is somewhat peculiar.
The treatment seems first to have been brought to notice
through a printed slip sent to medical journals generally. This
slip consisted of an extract from Nascher’s book on old age,
which at the time had not been published! Nascher also published
an article on this subject in an obscure journal, the
American Practitioner, for December, 1913. Neither the matter
copied from his book nor the article referred to contain
a single scientific fact that would warrant the claims made
for it as a therapeutic agent. No record is given of animal
experiments, and the clinical evidence presented certainly
cannot be regarded as scientific.
As already stated, this form of phosphorus has not been
previously used and has been regarded as without effect on
the human system because of its insolubility in any of the
liquids of the body. Nascher himself has not been able to find
any new way to dissolve it. He says: “I made a number of
experiments to find a solvent. The only substance which
appears to dissolve it is serum, but I am still uncertain
whether it is a solution or a very fine suspension. The
phosphorus is precipitated in a few days, but the serum
remains tinged.” The fact that it separates from the serum
on standing is quite conclusive evidence that it is insoluble
in that liquid. Since no way of making it soluble has been
discovered, there is no reason for expecting it to have any
effect on the system. An insoluble and non-absorbable substance
can produce no general systemic effect; if, when
ingested, it produces any effect whatever, this effect must be
local and will be shown by symptoms of gastro-intestinal
disturbance. Nascher, however, took 15 grains, and no symptoms
of gastro-intestinal disturbance followed. Hence, we
must conclude that it is without effect on the gastro-intestinal
mucous membrane. While Nascher records no experiments
on animals which is much to be regretted, he did experiment
on himself and says:
“Ten grains produced a frontal headache, restlessness, excessive
mental stimulation, ideas arising with such vividness as to appear as
actual occurrences. There was a sense of weight or oppression in the
stomach and priapism, the latter probably psychic, as I was looking
for such a result. These symptoms passed away in a few hours.”
Without doubt his explanations of the priapism can be
applied to the whole experience; whatever symptoms there
were, they were unquestionably psychic. The consideration
of these subjective symptoms may be dismissed, since it is
reasonable to assume that an insoluble, unabsorbable substance
which produces no disturbance of the gastro-intestinal tract
will have no effect on the rest of the organism.
Amorphous phosphorus did not produce such symptoms as
Nascher relates in experiments similar to his made by us.
The drug was taken in 10-grain doses by six different individuals.
In no case did the symptoms described by Nascher follow;
in fact, there were no symptoms whatever.
NASCHER’S THEORY
Nascher, after relating his subjective experiences and those
of his patients, proceeds to build a theory to account for the
unproved action of amorphous phosphorus in disease, especially
in arteriosclerosis. It would have been more appropriate
if before advancing the theory, he had made some experiments
to prove that the substance has some action. But we
give his theory as found in the quotation from his book, sent
to medical journals, as already referred to. Here it is:
“Amorphous Phosphorus in Senile Arteriosclerosis: The author
has used the red amorphous phosphorus in senile arteriosclerosis for
several years. Given originally as a substitute for ordinary phosphorus
in senile debility, it was found that it was eliminated as amorphous
phosphate of lime and that the lime elimination was thereby increased.
Weil’s experiments showed that the lime elimination in arteriosclerosis
was diminished. Phosphorus has the property of combining with lime
and increasing the lime assimilation. In the small doses which can be
given when the ordinary phosphorus is employed, the phosphorus will
combine with the lime of the food and increase the amount of lime
salts in the body. When given as amorphous phosphorus, the dose
is 2 grains or more several times a day, and with a lime-free diet the
lime required for the combination necessary to secure the elimination of
the phosphorus excess is drawn from the abnormal lime deposits. This
appears to be the rationale of the treatment and explains the good
results obtained from its use. From ‘Diseases of Old Age,’ by I. L.
Nascher, M.D., to be published shortly.”
Thus, according to Nascher, the phosphorus, after being
oxidized to phosphoric acid, catches the calcium and drags
it out of the system! What are the facts? The human
body contains a large store of phosphates which are excreted
in the urine in combination with sodium and potassium—and
yet these do not draw the calcium from the blood, brain
and bones! To be blunt, Nascher’s theory is absurd. The
calcium in its various deposits in the body is already combined
with phosphoric acid. Why should the phosphorus
introduced take calcium from the phosphate radical with
which it is already in combination? Nascher asserts that the
phosphorus which is introduced as amorphous phosphorus is
excreted as amorphous phosphate of lime within twenty-four
hours. How does he know it is? It is, of course, very appropriate
that amorphous phosphorus should form the amorphous
phosphate of lime, but, unfortunately, phosphates made from
the ordinary phosphorus also are precipitated in the amorphous
condition. By what private mark does Dr. Nascher
identify the amorphous phosphate produced by his amorphous
phosphorus? Is it not a fact that he found the urine alkaline
and detected a precipitate of amorphous calcium phosphate—always
present in alkaline urine—and concluded that this must
be his particular amorphous phosphorus in combination with
calcium?
Dr. Nascher makes no record of examinations of the feces,
although a great part—sometimes the greater part—of
ingested phosphorus is found in the feces in experimental
work on phosphorus metabolism. If he had examined the
feces he would doubtless have found the total quantity of
amorphous phosphorus unchanged.
Nascher refers to several cases in which he has used this
remedy and states that he had the most gratifying results.
So far as we can learn, the benefit was entirely in the subjective
symptoms of the patient. It seems evident, therefore,
that his claims for the value of this remedy rest on no better
foundation than an unproved theory without experimental
basis.
ITS COMMERCIALIZATION
Thus far we have considered only the scientific aspects of
amorphous phosphorus therapy. It is unfortunate that we
cannot stop here. Some of our readers will have seen in
recent medical journals half-page advertisements of amorphous
phosphorus reading:
The striking physical and chemical properties possessed by
common phosphorus, together with the fact that phosphorus
is one of the constituents of nerve-tissue, are probably
responsible for the reputation which this element acquired
generations ago as a remedy for sexual impotence and mental
decay. Among scientific men this reputation was a fleeting
one, for, when put to the test, the product failed. Like so
many products with a similar history, the unearned reputation
it obtained from medical men survived in the minds of
the laity, and, as is always the case, the survival has been
taken advantage of by quacks. Among charlatans and nostrum
makers phosphorus is still in vogue. “Weak men’s specialists”
and venders of “lost manhood” and alleged aphrodisiac
drugs “play up” the phosphorus fallacy for all it is
worth.
It is worth noting that the present exploitation of amorphous
phosphorus is following along somewhat similar lines.
The asserted actions of amorphous phosphorus are such as
may be calculated to appeal to the sexual neurasthenic.
There is no doubt that the Sharp & Dohme advertisements
will bring about an extensive use of this remedy, especially
by the uncritical. The psychic element—which plays so large
a part in the sexual neurasthenic—will result in favorable
reports being given on the drug. Articles may be expected
to appear in a certain class of medical journals, telling of the
marvelous results that Dr. John Doe has had in the use
of “Pill Phosphorus Amorphous S. & D.” A luxuriant crop
of testimonials may be expected to follow, and the tout
ensemble will go far to sustain the Sharp & Dohme propaganda.

Reproduction—reduced—of half-page advertisement appearing in medical
journals.
We are prompted to believe that Messrs. Sharp & Dohme
do not fully realize the potentialities for harm that lie in
their present exploitation of amorphous phosphorus. It
hardly seems possible that a firm of standing would knowingly
put on the market and advertise a worthless drug
with an appeal to susceptible, infirm old men. The function
of introducing new remedies to the medical profession
is a responsible one, and a firm that assumes it should
have among its officers some one sufficiently conversant with
pharmacologic science to prevent such unscientific absurdities
as that exhibited in the marketing of amorphous phosphorus,
especially under such claims as those contained in the advertisements.—(From
The Journal A. M. A., March 7, 1914.)
Dr. Nascher’s Reply to The Journal’s Article—Comments
To the Editor:—Regarding the article on Amorphous Phosphorus
in the March 7 issue of The Journal of the American
Medical Association, I want first of all to clear myself of
the implied charge of commercialism in connection with the
marketing of the Pill Phosphorus Amorphous by Sharpe &
Dohme. I have never had anything to do with the manufacture
or sale of those pills, never had any business dealings
with Sharpe & Dohme and I have no commercial interest
whatsoever in either this or any other drug or drug house.
I knew nothing about the advertisement which you reproduced
until I saw it in the medical journals. I immediately
protested against this unwarranted use of my name and was
assured that the statement “Made under the direction of
Doctor I. L. Nascher, New York,” was not made for the
purpose of misleading and that the ad. would be immediately
withdrawn. I gave my approval to the pills made by this
firm as I would give my approval to the pills made by any
other reliable house for I claim the right to endorse any
drug or preparation which I believe to be of value whether it
is approved by the Council of [on] Pharmacy and Chemistry
or not.
In your general charge of commercialism you make it
appear that the exploitation of amorphous phosphorus had
the ulterior purpose of appealing to the sexual neurasthenic
along the lines of the “lost manhood” ads. So far as this
relates to Sharpe & Dohme, I have no interest, but you have
included me in your general denunciation. The only reference
I ever made to aphrodisiac effects of Amorphous Phosphorus,
in all my writings, is contained in these words in the four-page
article in the American Practitioner, “In a few cases
aphrodisiac effects were noticed.” I have never recommended
amorphous phosphorus as an aphrodisiac and in the chapters
on “Impotence and Neurasthenia” in my book on “Diseases
of Old Age,” I have not mentioned amorphous phosphorus
at all.
You say “the treatment seems first to have been brought
to notice through a printed slip sent to medical journals
generally.” This slip containing an extract from my book
which was then in press was sent out about four months ago
while I have referred to amorphous phosphorus repeatedly
in medical articles during the past three years. In my paper
on Senile Debility, Medical Record, Jan. 21, 1911, I said
amorphous phosphorus had no effect, as I was then looking
for the usual effects of the ordinary phosphorus. In a paper
on Senile Mentality, International Clinics, Vol. 4, 21st series,
I said I was using amorphous phosphorus but had not yet
determined its value. I recommended it in several of my
papers, articles and lectures in 1912 and 1913 after I had
found that under its use in some cases of senile arteriosclerosis,
symptoms were relieved. I sent these slips to the medical
journals as a general reply to many inquiries I received about
amorphous phosphorus and I stated this in the letters I sent
with the slips to some editors. Further inquiries for more
information led me to write the paper which appeared in the
December issue of that “obscure journal” the American Practitioner.
I felt that a medical journal which carried articles
by Sir James Barr, ex-president of the British Medical Association,
Sir R. W. Philip, R. Murray Leslie, Halliday Sutherland,
and such American authorities as Adami, Hare,
Brooks, Hirschberg, Knopf, Starkey, Ely, Bissell, Wilcox,
Col. Maus, U. S. A., etc., was a representative high class
journal and I was pleased to have my paper in it.
To take up the scientific criticism of amorphous phosphorus,
permit me to say at the outset that I am a general practitioner,
specially interested in geriatrics, and more concerned
about obtaining favorable clinical results in my cases than
in solving laboratory problems. Nevertheless I have tried
for years to obtain the cooperation of expert laboratory
workers to help me determine the properties, chemical, physical
and physiological, of amorphous phosphorus. In 1909 or
1910 the Rockefeller Institute, in reply to my request for
permission to experiment there with amorphous phosphorus,
said it did not accept volunteer workers. Four heads of
college laboratories could not spare the time. I asked the
Council on Pharmacy and Chemistry last November to take
up its investigation and was informed that it could not do
so at present. I have been perforce compelled to depend
mainly on empirical methods with such little experimentation
as the facilities of the physician’s office permitted and such
little literature as I could find.
You reject empirical methods as being unscientific notwithstanding
the fact that most of our therapeutic knowledge
is based on empiricism. (I use the terms empirical and
empiricism here in the sense of knowledge obtained from
experience and observation, not in the bad sense in which
they might be construed.) It would therefore be folly on my
part to argue with you that I have obtained beneficial results
from amorphous phosphorus in many cases of senile arteriosclerosis.
I did not obtain such results from a single dose,
but gave it in some cases for many weeks or months. It is
unfair to judge of the value of a drug from a single dose or
several doses unless it is a drug which is expected to show
immediate effects. It would be greater folly on my part to
pit my knowledge of pharmacy and chemistry against the
knowledge of your staff of experts. I can but repeat what I
have said on many occasions that under the persistent use
of amorphous phosphorus in cases of senile arteriosclerosis
symptoms were frequently relieved. I never claimed that
amorphous phosphorus will cure arteriosclerosis. In the
chapter on Arteriosclerosis in my book I say: “Senile arteriosclerosis
being a natural, normal condition, is incurable in
the sense that it can be neither prevented nor removed. The
best that we can hope for is to retard its progress and relieve
disagreeable symptoms, etc.”
You say in reference to the elimination of the amorphous
phosphorus as amorphous phosphate of calcium, “Is it not
a fact that he (I) found the urine alkaline and detected a
precipitate of amorphous calcium phosphate—always present
in alkaline urine—and concluded that this must be his
particular amorphous phosphorus in combination with calcium?”
No. The specimens of urine were examined in
reliable laboratories and I have reports showing acid and
neutral urine as well as alkaline urine having the amorphous
phosphate precipitate. Nor is the amorphous phosphate
“always present in alkaline urine.”
As for the theory I advanced, it is simply a theory based
on reasoning without facts to prove it. If I had facts to
prove it, it would no longer be a theory or open for discussion.
Being a theory, it is the province of the wise man
to ridicule it and call it absurd. I will confess that your
criticism of it is not clear to me and I still do not see its
absurdity. I don’t see what relation your argument, that
the phosphates of sodium and potassium do not draw the
calcium from the blood, brains and bones, has to the theory
I advanced. It is true that I have no private mark by which
I can identify the amorphous phosphate produced by amorphous
phosphorus, but such argument is puerile. When medical
science has so far progressed that the physician will be
able to put his tag on the molecule of drug substance and
follow it through the various metabolic processes to its final
elimination we will not need any Council on Pharmacy and
Chemistry to decry what it cannot understand. Let me say
here that scientific criticism does not stoop to ridicule for
ridicule is usually based on animus or bias.
The conclusive proof of the value of a drug is not its
action on the healthy dog, frog or guinea-pig but its action
on the individual patient, and no amount of animal experimentation
can dispose of the personal factor which is so
marked in senile cases. This is no criticism of animal experimentation
as a whole but of the insistence on animal experimentation
to determine the value of a drug in a class of
cases for which the healthy animal can furnish no comparison.
You say amorphous phosphorus is practically inert and
quote Noé, Witthaus and Becker, Thornton and Phillips.
The quotations of the first three are little more than statements
that amorphous phosphorus is non-toxic. Phillips
makes two references, one of which is to Badner who obtained
decided effects from its prolonged use. Thornton, whom you
quote in your contention that amorphous phosphorus is inert,
says that on prolonged use in doses of 3⁄10 grains every two
hours it produced headache, vertigo, mental excitement,
priapism, etc. (See footnote under Phosphorus, U. S. Dispensatory).
Shoemaker’s Materia Medica and Therapeutics
says it is toxic and is called the servant-girl’s poison. Phillips
suggested that Badner probably used an impure drug. I suggested
that Thornton probably used an impure drug. On the
other hand, Badner and Thornton obtained positive results
from prolonged use, not from the single dose.
You say it has not been used on account of its insolubility
in any of the liquids of the body. Roscoe and Schorlemmer,
quoting Neuman, said if injected into the blood the usual
symptoms of phosphorus poisoning appear. In a letter from
Dr. Hatcher he says Nassé injected 0.2 gm. of the purest
amorphous phosphorus into a rabbit’s vein and the animal
presented the usual symptoms of phosphorus poisoning. There
are also references to amorphous phosphorus action in
Kobert’s Lehrbuch der Intoxicationen, in Blythe’s Poisons, etc.
You say of your four quotations, “the foregoing represents
our scientific knowledge as to the action of amorphous phosphorus.”
Did you not know of these other authorities, or
are their statements unscientific, or were they omitted because
they disprove your contention that amorphous phosphorus is
practically inert?
Your denunciation of ordinary phosphorus has no bearing
on the subject as I do not recommend the amorphous phosphorus
as a substitute for the other.
I have worked for eight years to arouse medical and public
interest in the aged and their ailments and I cannot afford
charges of commercialism, foisting worthless drugs as aphrodisiacs
or other unethical conduct to stand against me. As
for the charge of unscientific work, I can only point to my
work on Diseases of Old Age, and my medical papers, and
express the hope that others better equipped for laboratory
research will take up the laboratory investigation of amorphous
phosphorus. I have faith in its therapeutic value and
believe competent clinical observers will have favorable
results from it in suitable cases.
I. L. Nascher, M.D., New York.
Comment.—Accompanying the preceding letter was a note
from Dr. Nascher in which he says: “I want this published in
full without elision or change. If you do not intend to publish
it as written, I want it returned and enclose postage.”
The letter therefore is given in full in spite of the fact that
much of it is irrelevant to the question discussed.
Dr. Nascher’s protest to Sharpe and Dohme against the
“unwarranted use” of his name in connection with “Pill
Phosphorus Amorphous, S & D” seems to have resulted in
various modifications of the phrases connecting his name
with the exploitation of this pill. What was apparently the
original advertisement, contained the phrase:
“Made under the direction of Dr. I. L. Nascher, New York.”
Later advertisements, while identical in all other respects
with the first, had this phrase modified to read:
“Made at the suggestion of Dr. I. L. Nascher, New York.”
Still other advertisements, also identical with the first in
other respects, are modified to read:
“Made with the approval of Dr. I. L. Nascher, New York.”
That Dr. Nascher was directly or indirectly connected with
the commercializing of this product, The Journal has never
suggested, inferentially or otherwise. That the exploitation
of amorphous phosphorus by Sharpe and Dohme is one that
appeals to the sexual neurasthenic, no one who has read the
advertisements can deny. As a matter of fact, it would be
difficult to sell phosphorus in any form as a medicament,
without appealing to the sexual neurasthenic. The word
“phosphorus” has become, in the minds of both laymen and
physicians, more or less synonymous with the treatment of
so-called sexual weakness and it is a practical impossibility
to divorce the word from the idea suggested. How true this
is, Dr. Nascher himself unwittingly admits when he tells
that the result of his first experiment on himself with amorphous
phosphorus was a priapism that he acknowledges was
“probably psychic, as I was looking for such a result.” But
the Sharpe and Dohme advertisements plainly state that the
amorphous phosphorus pill they are marketing is a “new and
successful method of treatment for ... functional and
senile impotence....”
Dr. Nascher’s explanation of how he came to send out the
slip regarding amorphous phosphorus to medical journals
leaves him the victim of an unfortunate coincidence. It is at
least unusual for authors to send out advance extracts from
books that are about to be published, especially when such
extracts deal wholly with a drug that is coincidently being
introduced as a new proprietary product by some enterprising
pharmaceutical house.
Dr. Nascher takes exception to our statement that the
treatment seems first to have been brought to notice through
the printed slip sent to medical journals, and states that he
has “referred to amorphous phosphorus repeatedly in medical
articles appearing during the last three years.” His articles
for 1912 and 1913 have been examined for the purpose of
learning when the treatment as now presented to the profession
was first announced. In his article “Errors in the Treatment
of Senile Cases,” New York Medical Journal, Oct. 12,
1912, he speaks of the iodids in senile arteriosclerosis, but
says nothing about amorphous phosphorus. It may be
assumed, therefore, that the treatment had not been brought
to general notice at that time. The new treatment is very
briefly described in the New York Medical Journal, July 13,
1913, in an article whose title, “Longevity and Rejuvenescence”
gave no indication that it dealt with amorphous
phosphorus. Under the circumstances, it is not strange that
its therapeutic value was not learned of until Dr. Nascher’s
printed slips were sent out.
Dr. Nascher admits that his theory is based on empirical
methods. Most of the serious errors in therapeutics have had
their origin in this very method. It was on just such methods
that physicians reported wonderful results in the use of
alleged “lithia waters” that actually contained less lithium
than ordinary river water! So unscientific is the empirical
method that it is hardly worth taking the space to demonstrate
its imperfections.
Neither is it worth while to discuss the question of a constant
occurrence of a sediment of amorphous calcium phosphate
in alkaline urine. If there are exceptions to this rule,
they must be rare indeed.
In The Journal’s article authors were quoted to show that
amorphous phosphorus is regarded as inert. It was not suggested
that the authorities referred to were all that could be
found. Dr. Nascher refers to Thornton, Shoemaker, Neuman,
Blythe and Kobert, and asks whether the various statements
on the subject, made by these men, are unscientific or
were “omitted because they disproved” the contention that
amorphous phosphorus is practically inert. Thornton’s article
was omitted because it is unscientific in that he does not
report experiments made by himself, but refers to an unpublished
paper by one Kelly. Who Kelly is, or was, he does
not tell us. Kelly’s report, therefore, should be and was disregarded,
since it is the work of an unknown author and
there is nothing in the article to indicate that Thornton was
in any position to vouch for Kelly’s work. Incidentally, it
may be said that Kelly’s report merely recorded subjective
symptoms; Dr. Nascher himself indicates his distrust of
Kelly’s alleged results by suggesting that an impure preparation
was used!
Shoemaker’s report was not given, for a similar reason.
Shoemaker says:
“Amorphous phosphorus is almost completely destitute of taste or
odor, has no immediate caustic effect, and is claimed to be less toxic
than white phosphorus; but in the form of matches [Italics ours.—Ed.]
has caused many deaths and is known as the ‘servant girls’ poison.’”
It is well known that commercial amorphous phosphorus
is usually impure, and it is more than probable that if toxic
effects were produced by the ingestion of match-heads, these
matches were made either of white phosphorus or of very
impure red phosphorus. In any case, Shoemaker’s statement
has no bearing whatever on the pharmacologic action of
pure amorphous phosphorus.
The statement of Neuman quoted from Roscoe and Schorlemmer,
as well as that of Nassé, referred to by Hatcher,
had no bearing on the question at issue, as these men injected
the material into the blood-stream. If, when the amorphous
phosphorus is injected into the blood, it produces the ordinary
symptoms of phosphorus poisoning, one would naturally
expect the same symptoms when the substance is given by
mouth—if amorphous phosphorus were soluble or absorbable.
The fact that such symptoms are not produced when amorphous
phosphorus is taken into the alimentary canal, sustains
the views held by chemists, pharmacologists and physicians,
that the drug is practically insoluble and unabsorbable—in
other words, inert.
Dr. Nascher declares that he “never claimed that amorphous
phosphorus will cure arteriosclerosis.” Yet he insists
that amorphous phosphorus removes lime from the “abnormal
lime deposits” that occur in arteriosclerosis. What is
this but claiming curative action?
Summed up, Dr. Nascher’s own admissions amply confirm
the main contentions of The Journal’s article. He admits
that he has no experimental basis for the use of this remedy;
he admits that his theory “is simply a theory without facts
to prove it.” The only conclusions that can be reached
from his reply coincide closely with the very statement made
by The Journal, and which we here reiterate:
“It seems evident, therefore, that his claims for the value
of this remedy rest on no better foundation than an unproved
theory without experimental basis.”—(From The Journal
A. M. A., March 28, 1914.)
INDEX
(Including References to Articles Not Contained
in This Book)
The following is an index (1) to topics discussed in this
volume, and (2) to products discussed in reports and other
articles not included here:
1. The references to topics discussed or mentioned in this
volume are printed in CAPITALS.
2. The references to articles published elsewhere are
printed in small letters. These references include papers
published in The Journal of the American Medical Association,
papers published in the “Annual Reports of the Council
on Pharmacy and Chemistry” and a few published in the
“Annual Reports of the Chemical Laboratory of the American
Medical Association.” A number of these papers have
appeared both in The Journal and in the Council Reports.
In such cases, for the benefit of those who may not have
access to files of both The Journal and the Council Reports,
references are given to both sources. Some of this matter is
also issued in reprint form.
alpha-table
| APAGE |
|
|
Acetanilid, Harmful Effects of305 |
|
Acetphenetidin and Phenacetin—Their Relative Purity414 |
Acetphenetidin, Harmful Effects of305 |
|
|
|
Adrenalin, Name, versus the Name Epinephrin454 |
|
Agar-Lac and Agar-Lac, Inc.10 |
| Agmel (Maguey Products Co.), The Journal, Oct. 12, 1912, p. 1392. |
| Agurin Tablets (Bayer Co.), Reports Council Pharm. & Chem., 1915, p. 162. |
|
|
| Alborum (Whitehouse Chemical Co., Inc.), The Journal, Dec. 12, 1914, p. 2148; Reports Council Pharm. & Chem., 1914, p. 129. |
| Aletrin, The Journal, Nov. 13, 1909, p. 1655; Reports Council Pharm. & Chem., 1909, p. 135. |
| Aletris Compound, Elixir (Parke, Davis and Co., Ray Chemical Co.), Reports Council Pharm. & Chem., 1912, p. 46. |
|
|
| Alfatone (Norwich Pharmacal Company), The Journal, Aug. 7, 1915, p. 548; Reports Council Pharm. & Chem., 1915, p. 62. |
Alienist and Neurologist, Advertising in31 |
| Alkaline Digestine (Parke, Davis and Co.), Reports Council Pharm. & Chem., 1912, p. 44. |
| Alkalol (Alkalol Company), The Journal, Nov. 6, 1915, p. 1665. |
|
Almond Preparations, Various Diabetic450 |
American Journal of Clinical Medicine, Advertising in303, 304, 342 |
American Journal of Obstetrics, Advertising in342 |
|
American Malted Food Company319 |
|
|
|
| Amolin Deodorant Powder (Amolin Chemical Co.), The Journal, Feb. 22, 1908, p. 626; Reports Chem. Lab., 1909, p. 63. |
|
Ammonol Chemical Company338 |
|
|
Analgesic Balm, Bengué’s267 |
Analgesic Cream, Stearns’267 |
|
| Analutos and Analutos Tablets (Royal Pharmaceutical Works, Meppel, Holland), The Journal, Feb. 20, 1915, p. 684; Reports Council Pharm. & Chem., 1915, p. 135. |
|
Anasarcin Chemical Company11, 18 |
|
|
Anedemin Chemical Company12, 16, 18 |
Angier Chemical Company170 |
Angier’s Petroleum Emulsion169 |
Angier’s Throat Tablets173 |
Anglo-American Pharmaceutical Company, Ltd.58 |
Animal Therapy Company317 |
| Anistamina (M. Olivetti), Reports Council Pharm. & Chem., 1915, p. 162. |
Annals of Surgery, Advertising in35, 421 |
Anodyne Balm, P-M Company267 |
|
|
|
| Antikamnia and Quinin (Antikamnia Chemical Co.), The Journal, July 1, 1905, p. 55. |
|
| Antimeristem-Schmidt (Laboratorium W. Schmidt), The Journal, March 8, 1913, p. 766. |
Anti-Neuralgic Ointment267 |
| Antiphlogistine (Denver Chemical Manufacturing Company), The Journal, June 1, 1907, p. 1875. |
Antipyrin, Harmful Effects of305 |
Antiseptic Powder, Maignen19 |
|
| Antiseptic Tablets, Clover (Sharp & Dohme), The Journal, Aug. 26, 1911, p. 755. |
Antiseptic W-A, Intestinal103 |
|
Anusol Hemorrhoidal Suppositories227, 280, 281 |
|
| Arbor Vitae, Reports Council Pharm. & Chem., 1912, p. 38. |
Archives of Pediatrics, Advertising in31 |
|
Army and Navy Magazine434 |
|
Arnold’s Zymotoid and Arnold’s Zymotoid Company412 |
Aromatic Digestive Tablets229 |
|
|
|
Atlanta Journal-Record of Medicine, Advertising in31, 35, 296 |
|
|
Aubergier’s Syrup of Lactucarium399 |
| Autolysin (Autolysin Laboratory), The Journal, July 24, 1915, p. 336; Nov. 6, 1915, pp. 1647, 1662. |
| Avenin Compound Tablets (Parke, Davis & Co.), Reports Council Pharm. & Chem., 1912, p. 44. |
| B |
| Baby Taeniafuge Grape (Grape Capsule Co.), Reports Council Pharm. & Chem., 1915, p. 174. |
| Bacillicide (Prophytol Products Co.), The Journal, Nov. 14, 1914, p. 1778; Reports Council Pharm. & Chem., 1914, p. 125. |
|
Baking Powder, Jireh Diabetic450 |
| Bakurol (Sharp & Dohme), The Journal, July 10, 1915, p. 175. |
|
Ballard-Snow Liniment Company43 |
Ballard’s Snow Liniment205 |
| Baneberry, Reports Council Pharm. & Chem., 1912, p. 38. |
| Baptisin, The Journal, Nov. 13, 1909, p. 1655; Reports Council Pharm. & Chem., 1909, p. 135. |
Bannerman, Wm., & Co., and Bannerman’s Intravenous Solution105 |
|
|
|
|
|
Bauer Chemical Company366 |
Baume Analgésique Bengué267 |
| Bee, Honey, Reports Council Pharm. & Chem., 1912, p. 38. |
|
|
Bell-Ans (Pa-Pay-Ans, Bell)151, 282 |
| Benetol (Northern Chemical Assn.), The Journal, April 15, 1911, p. 1128; Reports Chem. Lab., 1911, p. 82. |
|
Beyer, Charles and Frank299 |
|
Bischof’s Diabetic Gluten Bread450 |
Bischof’s Gluten Flour449 |
|
Biscuit, Various Diabetic449, 450 |
| Bismuth Iodo-Resorcin Sulphonate, The Journal, Feb. 11, 1911, p. 441; Reports Chem. Lab., 1911, p. 14. |
| Bismuth, Opium and Phenol Tablets (Hance Bros. & White, Wm. S. Merrell Chemical Co., Parke, Davis & Co., Sharp & Dohme, F. Stearns & Co., Truax, Greene & Co., H. K. Wampole & Co., Wm. R. Warner & Co.), The Journal, July 25, 1908, p. 330; Dec. 17, 1910, p. 2169; May 6, 1911, p. 1344; Reports Chem. Lab., 1909, p. 28; 1910, p. 85; 1911, p. 22. |
| Bitter Bark, Reports Council Pharm. & Chem., 1912, p. 39. |
| Bladder Wrack, Reports Council Pharm. & Chem., 1912, p. 39. |
|
| Blandine Laxative, Mulford (H. K. Mulford Co.), Reports Council Pharm. & Chem., 1914, p. 136. |
| Blaud Capsules and Blaud Arsenic and Strychnine Capsules, Frosst’s (C. E. Frosst & Co.), Reports Council Pharm. & Chem., 1915, p. 164. |
Bliss, Alonzo O., Company463 |
| Blood Tonic, Alterative (Parke, Davis & Co.), Reports Council Pharm. & Chem., 1912, p. 47. |
| Blue Cohosh, The Journal, Sept. 11, 1915, p. 972. |
|
| Boneset, Tall, Reports Council Pharm. & Chem., 1912, p. 45. |
|
| Borolyptol (Palisade Mfg. Co.), The Journal, Nov. 15, 1913, p. 1812. |
Bouma, Dr., Sugar-Free Fat-Milk449 |
|
|
Bradbury’s Eczema Lotion245 |
Brant, J. W., Company, Ltd411 |
Breads, Various Diabetic450 |
Breakfast Food, Gum Gluten450 |
Breitenbach, M. J., Company159 |
Bristol-Myers Company87, 179 |
| Brobor (Gaynor-Bagstad Co.), Reports Council Pharm. & Chem., 1915, p. 164. |
| Bromides with Cypripedium Compound (Truax, Greene & Co.), Reports Council Pharm. & Chem., 1912, p. 43. |
|
|
Bromin-Iodin Chemical Company and Bromin-Iodin Compound285 |
| Bromo-Mangan (Reinschild Chemical Company), Reports Council Pharm. & Chem., 1915, p. 165. |
| Broom Corn, Reports Council Pharm. & Chem., 1912, p. 39. |
|
Brown’s Iron Bitters Company43 |
| Bruschettini Curative Vaccine (A. Bruschettini), Reports Council Pharm. & Chem., 1915, p. 176. |
Brusson Chocolate with Added Gluten450 |
Buchu and Hyoscyamus Comp., Tyree’s Elixir407 |
| Buchu and Hyoscyamus Compound, Tyree’s Elixir of (J. S. Tyree), Reports Council Pharm. & Chem., 1915, p. 167. |
| Buchu and Pareira Compound Elixir (Parke, Davis & Co.), Reports Council Pharm. & Chem., 1912, p. 46. |
| Buchu, Juniper and Acetate Potassium, Elixir of (Pitman-Moore Co.), Reports Council Pharm. & Chem., 1915, p. 166. |
| Budwell’s Emulsion of Cod-Liver Oil, Nos. 1 and 2 (Budwell Pharmacal Company), The Journal, Feb. 20, 1915, p. 684; Reports Council Pharm. & Chem., 1915, p. 135. |
|
Buffalo Medical Journal, Advertising in31, 35, 300 |
Burnham’s Soluble Iodine110, 233 |
Burnham’s Soluble Iodine Company110 |
Butters, Various Diabetic450 |
| C |
|
| Cactin (The Abbott Laboratories) and Cactina (Sultan Drug Company), The Journal, Sept. 21, 1907, p. 1021; March 21, 1908, p. 956; April 4, 1908, p. 1140; Aug. 6, 1910, p. 455. |
|
|
|
| Cactus Grandiflorus, The Journal, Sept. 21, 1907, p. 1021; Jan. 7, 1911, p. 26. |
| Calcidin Abbott and Calcidin Tablets (The Abbott Laboratories), The Journal, Sept. 7, 1907, p. 865; Reports Chem. Lab., 1909, p. 7. |
|
Callard’s Preparations for Diabetics450 |
|
|
|
Campho-Phenique Company40, 42 |
|
Canada Lancet, Advertising in279, 300 |
Canadian Medical Association Journal, Advertising in300 |
Canadian Practitioner and Review, Advertising in300 |
Cancer Hospital (Kellam)426 |
| Cannabis Compound, Syrup of (Pitman-Moore Co.), Reports Council Pharm. & Chem., 1915, p. 168. |
| Captol (Mülhens & Kropff), The Journal, Sept. 10, 1910, p. 959; Reports Chem. Lab., 1910, p. 70. |
|
|
Carnrick, G. W., Company185, 403 |
| Caroid and Essence of Caroid (Mead, Johnson & Co.), Reports Council Pharm. & Chem., 1914, p. 109. |
|
| Casca-Aletris (Pullen-Richardson Chemical Co.), Reports Council Pharm. & Chem., 1912, p. 46. |
|
|
Casoid Diabetic Preparations449, 450 |
| Caviblen (A. Grimme), Reports Council Pharm. & Chem., 1915, p. 176. |
| Cedron Seed, Reports Council Pharm. & Chem., 1912, p. 40. |
|
|
| Celery and various Elixirs of Celery (Hance Bros. & White, Nelson, Baker Co., Parke, Davis & Co., Ray Chemical Co., Smith, Kline & French Co., F. Stearns & Co.), Reports Council Pharm. & Chem., 1912, p. 40. |
| Cellasin (Mead Johnson & Co.), The Journal, Sept. 12, 1908, p. 931; Oct. 30, 1909, p. 1496; Reports Council Pharm. & Chem., 1905–8, p. 198; 1909, p. 118. |
Cereo Soy Bean Gruel Flour450 |
|
|
Chambers, Arthur and Leslie T.147 |
Chambers, J. H. and M. E.146 |
|
Charlotte Medical Journal, Advertising in31, 35, 422 |
Chemische Fabrik Falkenberg85 |
Chesebrough, Robert A.161 |
Chicago Medical Recorder, Advertising in31 |
|
|
| Chiodrastis (H. K. Wampole & Co.), Chionacea (Nelson, Baker & Co., Inc.), Elixir Chionanthus Compound (Ray Chemical Co.), and Elixir Chionanthus (Special) (Parke, Davis & Co.), Reports Council Pharm. & Chem., 1912, p. 42. |
|
|
Chocolate, proprietary diabetic450 |
|
| Chologestin (F. H. Strong Co.), The Journal, Dec. 11, 1915, p. 2108. |
| Chromiac Tablets (Maltbie Chemical Co.), Reports Council Pharm. & Chem., 1912, p. 44. |
|
|
| Citarin (The Bayer Company, Inc.), The Journal, Feb. 20, 1915, p. 685; Reports Council Pharm. & Chem., 1914, p. 135. |
|
|
| Clover Compound, Syrup Red (Nelson, Baker & Co.), Reports Council Pharm. & Chem., 1912, p. 40. |
Cocillana Compound, Syrup of396 |
| Cod Liver Ext., Stearns’ Wine of, with Peptonate of Iron (Frederick Stearns & Co.). Reports Council Pharm. & Chem., 1915, p. 177. |
Cod liver oil and cod liver oil cordials, comparative nutrient value of442 |
| Cod-Liver Oil, Budwell’s Emulsion of, Nos. 1 and 2 (Budwell Pharmacal Co.), The Journal, Feb. 20, 1915, p. 684; Reports Council Pharm. & Chem., 1915, p. 135. |
|
| Cohosh, Blue, and Fluidextract Blue Cohosh Compound (Parke, Davis & Co.), Reports Council Pharm. & Chem., 1912, p. 40. |
| Colchi-Methyl Capsules (H. K. Wampole & Co.), Reports Council Pharm. & Chem., 1915, p. 169. |
|
|
Columbus Pharmacal Company344 |
|
| Compound, Waterbury’s (Waterbury Chemical Co.), The Journal, March 20, 1915, p. 1016; Reports Council Pharm. & Chem., 1915, p. 138. |
| Condurango, Reports Council Pharm. & Chem., 1911, p. 54. |
|
Consolidated Color and Chemical Works328 |
|
| Corydalis Compound, Elixir (Parke, Davis & Co.), Reports Council Pharm. & Chem., 1912, p. 47. |
|
| Coto and Cotoin, Reports Council Pharm. & Chem., 1913, p. 39. |
|
|
Cramp Bark Compound, Fluid Extract of410 |
|
|
| Cuprase (in “Chemotherapy and Tumors”), The Journal, April 17, 1915, p. 1283; Reports Council Pharm. & Chem., 1915, p. 28. |
Cudahy Packing Company471, 472 |
| Curare and Curarin, The Journal, Jan. 15, 1910, p. 219; Reports Council Pharm. & Chem., 1910, p. 7. |
|
| Cystitis Tablet (Parke, Davis & Co., Smith, Kline & French Co.), Reports Council Pharm. & Chem., 1912, p. 45. |
Cystogen, Cystogen Aperient, Cystogen-Lithia and Cystogen Chemical Company60 |
|
| D |
|
| Damiana, Allan’s Compound Extract of (Allan-Pfeiffer Chemical Co.), The Journal, July 19, 1913, p. 211. |
|
|
Daniel’s Concentrated Tincture of Passiflora156, 332 |
|
|
|
|
Denver Medical Times and Utah Medical Journal, advertising in31, 35, 49 |
Detroit Medical Journal, advertising in300 |
| Diabetic Biscuit and Flour and other Foods, Jireh (Jireh Diabetic Food Company), The Journal, March 22, 1913, p. 922, and Dec. 14, 1912, p. 2174. |
Diabetic foods offered for sale in the United States446 |
| Dianol I, Dianol II and Dianol III (Kalle & Co.), Reports Council Pharm. & Chem., 1913, p. 34. |
| Diastos, Liquor (H. K. Mulford Co.), The Journal, Feb. 9, 1907, p. 533. |
|
|
Dietetic and Hygienic Gazette, advertising in342 |
Digestive tablets, aromatic229 |
| Digestive Tonic (Truax, Greene & Co.), Reports Council Pharm. & Chem., 1912, p. 44. |
| Digitalis preparations, proprietary, The Journal, Dec. 4, 1915, p. 2024; Reports Council Pharm. & Chem., 1915, p. 89. |
| Digitalone (Parke, Davis & Co.), The Journal, June 12, 1909, p. 1938; Dec. 7, 1912, p. 2074; Jan. 11, 1913, p. 143. |
| Digitalysatum (E. Bischoff & Co.), The Journal, Feb. 15, 1913, p. 499; Jan. 8, 1916, p. 135; Reports Council Pharm. & Chem., 1915, p. 93. |
|
|
|
| Dioscorea, various elixirs of (H. K. Mulford Co., Parke, Davis & Co., Ray Chemical Co., F. Stearns & Co.), Reports Council Pharm. & Chem., 1912, pp. 41, 46. |
|
|
|
| Diurol (H. K. Mulford Co.), Reports Council Pharm. & Chem., 1912, p. 45. |
|
| Dogwood, Flowering, Reports Council Pharm. & Chem., 1912, p. 41. |
Dominion Medical Monthly, advertising in300 |
Dowd, J. Henry, and Dowd’s Phosphatometer476 |
Drake’s Palmetto Compound475 |
Dublin Journal Medical Science, advertising in279 |
| Duodenin-Armour (Armour & Co.), The Journal, Aug. 4, 1915, p. 639; Jan. 15, 1916, p. 178; Reports Council Pharm. & Chem., 1915, pp. 99, 151. |
| Dyspepsia Compound, Elixir (H. K. Mulford Co.), Reports Council Pharm. & Chem., 1912, p. 44. |
| Dyspepsia, Elixir Atonic, Phenolated (Wm. S. Merrell Chemical Company), The Journal, Feb. 9, 1907, p. 533. |
| E |
|
|
|
|
Eclectic Medical Journal, advertising in31, 35, 49, 342 |
|
Eczema Lotion, Dr. Bradbury’s245 |
| Edema Tablet (Parke, Davis & Co., Smith, Kline & French Co.), Reports Council Pharm. & Chem., 1912, pp. 41, 45. |
|
|
|
| Elder, Reports Council Pharm. & Chem., 1912, p. 41. |
| Electro-Selenium (in “Chemotherapy and Tumors”), The Journal, April 17, 1915, p. 1283; Reports Council Pharm. & Chem., 1915, p. 28. |
Ellingwood’s Therapeutist, advertising in31, 35 |
| Emulsio Minerolein and Emulsio Phen-Oleum (T. R. D. Barse Co.), Reports Council Pharm. & Chem., 1915, p. 169. |
| Endotin (Morgenstern & Co.), Reports Council Pharm. & Chem., 1914, p. 136. |
| Enesol (Fougera & Co.), The Journal, July 26, 1913, p. 293. |
Enteronol and Enteronol Company294 |
| Eosin-Selenium (in “Chemotherapy and Tumors”), The Journal, April 17, 1915, p. 1283; Reports Council Pharm. & Chem., 1915, p. 28. |
Epinephrin, name, versus the name adrenalin454 |
| Episan (Gaynor-Bagstad Co.), Reports Council Pharm. & Chem., 1915, p. 164. |
|
| Ergone (Parke, Davis & Co.), The Journal, Oct. 7, 1911, p. 1211; Oct. 14, 1911, p. 1302. |
| Ergotole (Sharp & Dohme), The Journal, Oct. 7, 1911, p. 1211; Oct. 14, 1911, p. 1302. |
|
| Eryngo, Water, Reports Council Pharm. & Chem., 1912, p. 47. |
|
|
| Eunatrol (C. Bischoff & Co.), The Journal, Feb. 22, 1908, p. 627. |
|
| Eusoma (Eusoma Pharmaceutical Co.), Reports Council Pharm. & Chem., 1912, p. 38. |
Eusoma Pharmaceutical Company81 |
Expurgo Anti-Diabetes and Expurgo Manufacturing Company299 |
|
| Expurgo Lapis (Expurgo Manufacturing Co.), The Journal, Nov. 8, 1913, p. 1733. |
|
| F |
|
Farbenfabriken of Elberfeld Company311, 414 |
| Febrisol (Tilden Co.), The Journal, June 29, 1912, p. 2043. |
| Febri-Tone (Arthur Veter & Co.), The Journal, Feb. 1, 1908, p. 379. |
Fellows’ Syrup of Hypophosphites436 |
|
| Fermenlactyl (Anglo-American Pharmacal Co., Ltd.), The Journal, Jan. 30, 1909, pp. 372, 397. |
| Ferric Arsenite, Soluble, Reports Council Pharm. & Chem., 1912, p. 30. |
| Figwort, Reports Council Pharm. & Chem., 1912, p. 42. |
| Filudine (Geo. J. Wallau, Inc.), The Journal, Sept. 18, 1915, p. 1045; Reports Council Pharm. & Chem., 1915, p. 156. |
Flours, various diabetic449, 450 |
Foods, diabetic, offered for sale in the United States446 |
|
|
| Formamint (A. Wulfing Co.), The Journal, Aug. 28. 1915, p. 816; Reports Council Pharm. & Chem., 1915, p. 64. |
| Formidin (Parke, Davis & Co.), The Journal, Sept. 5, 1908, p. 818; Reports Council Pharm. & Chem., 1905–8, p. 192. |
|
|
|
|
Franco-American Ferment Company120 |
|
Fraud, Great American, and pharmaceutical manufacturers474 |
Frederick, Purdue, Company100 |
| Friedmann’s Vaccine (Standard Distributing Co.), Reports Council Pharm. & Chem., 1914, p. 136. |
| Fringe Tree, Reports Council Pharm. & Chem., 1912, p. 42. |
Fromm’s diabetic breads and chocolate450 |
| Frosst’s Blaud Capsules and Frosst’s Blaud, Arsenic and Strychnine Capsules (C. E. Frosst & Co.), Reports Council Pharm. & Chem., 1915, p. 164. |
| G |
| G. G. Phenoleum Disinfectant (G. G. Chemical Co., Inc.), The Journal, Jan. 30, 1915, p. 456; Reports Council Pharm. & Chem., 1915, p. 131. |
| Galactagogue (Eli Lilly & Co.), Reports Council Pharm. & Chem., 1912, p. 43. |
|
|
Gardner-Barada Chemical Company256 |
Gardner’s Syrup of Hydriodic Acid97 |
|
| Gelsemine Hydrochlorid and Gelseminine, Reports Council Pharm. & Chem., 1911, p. 57. |
|
| Genitone (Wm. S. Merrell Chemical Co.), Reports Council Pharm. & Chem., 1912, p. 44. |
|
German Council on Pharmacy and Chemistry459 |
German Propaganda for Reform458 |
|
|
|
| Ginseng, Reports Council Pharm. & Chem., 1912, p. 42; The Journal, Oct. 24, 1914, p. 1486. |
| Ginseng Compound, Elixir (H. K. Mulford Co.), and Ginseng Compound (Special), Elixir (Parke, Davis & Co.), Reports Council Pharm. & Chem., 1912, p. 42. |
Glasgow Medical Journal, advertising in279 |
|
| Glidine (Menley & James), The Journal, June 28, 1913, p. 2037. |
| Globeol (Geo. J. Wallau, Inc.), The Journal, Sept. 18, 1915, p. 1046; Reports Council Pharm. & Chem., 1915, p. 157. |
Gluten products, various, for diabetic use450 |
| Glutol-Schleich (Schering & Glatz), Reports Council Pharm. & Chem., 1915, p. 170. |
|
| Glycerine Tonic Comp., Gray’s (Purdue Frederick Co.), The Journal, July 10, 1915, p. 189; Reports Council Pharm. & Chem., 1915, p. 56. |
| Glycero-Lecithin, Pill (Westerfield Pharmacal Co.), Reports Council Pharm. & Chem., 1915, p. 170. |
| Glycerole of Lecithin (Fairchild Bros. & Foster), Reports Council Pharm. & Chem., 1915, p. 123. |
|
|
|
|
|
|
| Goat’s Rue, Reports Council Pharm. & Chem., 1912, p. 42. |
|
| Gonococcide (Cox Chem. Co.), The Journal, Aug. 24, 1907, p. 708. |
|
|
Grand prix and gold medals for sale462 |
| Gray’s Glycerine Tonic Comp. (Purdue Frederick Co.), The Journal, July 10, 1915, p. 189; Reports Council Pharm. & Chem., 1915, p. 56. |
Grisard, A. F., B. A., Dr. John P. and Dr. John W.18 |
| Guaialin (Organic Chemical Mfg. Co.), The Journal, Sept. 5, 1908, p. 818; May 8, 1909, p. 1511; Reports Council Pharm. & Chem., 1905–8, p. 166; Reports Council Pharm. & Chem., 1909, p. 76. |
| Guarana and Celery, various Elixirs of (Hance Bros. & White, Parke, Davis & Co., and Ray Chemical Co.), Reports Council Pharm. & Chem., 1912, p. 40. |
|
|
Gum Gluten Breakfast Food450 |
| H |
| H-M-C; see Hyoscin-Morphin Cactin. |
Hagee’s Cordial of Cod-Liver-Oil51, 289, 443 |
| Hair Cap Moss, Reports Council Pharm. & Chem., 1912, p. 43. |
Hammond, G. H., Company472 |
|
|
Hayden’s Viburnum Compound218, 409 |
|
|
Headache Powders, Koehler’s10 |
“Health Food” diabetic preparation450 |
|
|
| Helonias, elixirs and fluidextracts of, various (Hance Bros. & White, H. K. Mulford Co., Parke, Davis & Co., Ray Chemical Co. and Smith, Kline & French Co.), Reports Council Pharm. & Chem., 1912, pp. 41, 46. |
| Helonin Comp., Mist. (Schlotterbeck & Foss), The Journal, Dec. 18, 1915, p. 2186. |
|
Hemorrhoidal Suppositories, Anusol227, 280 |
Henry Pharmacal Company198 |
|
|
| Hexa-co-sal-in (Hexa-Co-Sal-In Company), The Journal, Oct. 2, 1915, p. 1203; Reports Council Pharm. & Chem., 1915, p. 159. |
| Hex-a-lith (Smith-Dorsey Company), The Journal, Feb. 14, 1914, p. 555. |
|
Hoffmann-La Roche Chemical Works68, 252 |
|
|
| Horse Nettle, Reports Council Pharm. & Chem., 1912, p. 43. |
Hospital Bulletin of the University of Maryland, advertising in35 |
Howell, H. B. & Co., Ltd.326 |
Huntley and Palmer’s Akoll Biscuit449 |
|
| Hydragogin (C. Bischoff & Co.), The Journal, Jan. 27, 1906, p. 288; The Journal, Sept. 4, 1915, p. 894; Reports Council Pharm. & Chem., 1915, p. 154. |
| Hydrangea and Lithia, Elixir (Hance Bros. & White), Reports Council Pharm. & Chem., 1912, p. 45. |
| Hydrangea, Lithiated (Lambert Pharmacal Co.), Reports Council Pharm. & Chem., 1912, p. 42. |
| Hydrastis and Cramp Bark Compound, Hydrastis and Viburnum Compound, |
|
Hydriodic Acid, Gardner’s Syrup of97 |
Hydrocyanate of Iron, Tilden235 |
| Hydroleine (Charles N. Crittenton Company), Reports Council Pharm. & Chem., 1915, p. 171. |
| Hydron (Wm. S. Merrell Chemical Co.), Reports Council Pharm. & Chem., 1912, p. 44. |
|
| Hydropsin (Ernst Bischoff Co.), The Journal, Jan. 8, 1916, p. 135; Reports Council Pharm. & Chem., 1915, p. 94. |
|
|
| Hyoscin-Morphin Cactin (now Hyoscin-Morphin-Cactoid) (Abbott Laboratories), The Journal, Dec. 21, 1907, p. 2103. |
|
|
Hypophosphites, Fellows’ Syrup of436 |
|
| I |
| Ichthynate (Mallinckrodt Chemical Works), Reports Chem. Lab., 1912, p. 110. |
Indianapolis Medical Journal, advertising in31, 35, 300 |
|
|
|
| Interol (Van Horn & Sawtell), The Journal, July 10, 1915, p. 175. |
International Journal of Surgery, advertising in31, 49, 342, 429 |
Interstate Medical Journal, advertising in49 |
Intestinal Antiseptic W-A.103 |
| Intravenin P-H (Intravenin Products Co.), Reports Council Pharm. & Chem., 1915, p. 120. |
Intravenous Products Company212, 214 |
Intravenous Solution, Bannerman’s105 |
|
|
|
| Iodin Petrogen (John Wyeth & Bro.), The Journal, Nov. 30, 1912, p. 1992. |
Iodine, Burnham’s Soluble and Burnham’s Soluble Iodine Tablets110, 233 |
| Iodival (Knoll & Co.), The Journal, March 4, 1911, p. 685. |
| Iodo-Bromide of Calcium Comp. “Without Mercury” and “With Mercury,” Elixir of (Tilden Co.), The Journal, Nov. 6, 1915, p. 1662; Reports Council Pharm. & Chem., 1915, p. 160. |
| Iodomuth (Organic Chemical Mfg. Co.), The Journal, Sept. 5, 1908, p. 818; May 8, 1909, p. 1511; Reports Council Pharm. & Chem., 1905–8, p. 166; Reports Council Pharm. & Chem., 1909, p. 75. |
|
|
|
| Iodum-Miller and Iod-Izd-Oil, Miller’s (Iodum-Miller Co.), The Journal, Oct. 2, 1915, p. 1202; Reports Council Pharm. & Chem., 1915, p. 76. |
|
|
Iowa Medical Journal, advertising in35, 300 |
|
Iron, Hydrocyanate of, Tilden235 |
| Iron Solution for Intravenous Therapy—Perkins and Ross (Perkins and Ross), The Journal, Nov. 14, 1914, p. 1778; Reports Council Pharm. & Chem., 1914, p. 125. |
|
| Isopral (Farbenfabriken of Elberfeld Co.), The Journal, Aug. 8, 1908, p. 487; Reports Council Pharm. & Chem., 1905–8, p. 119. |
| J |
| Jaroma (Jaroma Company), The Journal, Sept. 2, 1911, p. 835; Reports Chem. Lab., 1913, p. 103. |
|
| Jireh Diabetic (Diatetic) Biscuit, Flour and other foods (Jireh Diabetic Food Company), The Journal, March 22, 1913, p. 922; Dec. 14, 1912, p. 2174. |
Jireh Diabetic (Diatetic) food products449, 450 |
Jireh Diabetic Food Company451 |
Johns, L. D., Company and Dr. John’s Tablets154 |
|
Journal of Nervous and Mental Diseases, advertising in35 |
Journal Tropical Medicine and Hygiene, advertising in279 |
| Jubol (Geo. J. Wallau, Inc.), The Journal, Aug. 14, 1915, p. 639; Reports Council Pharm. & Chem., 1915, p. 152. |
| Juglandin, The Journal, Nov. 13, 1909, p. 1655; Reports Council Pharm. & Chem., 1909, p. 135. |
| K |
|
|
|
Kansas City Medical Record, advertising in296 |
Katharmon Chemical Company51 |
|
| Kefilac (Kefilac Co.), The Journal, Jan. 30, 1909, pp. 372, 397. |
Kellam [cancer] Hospital426 |
Kellogg’s diabetic products449, 450 |
Kennedy’s Pinus Canadensis, Light and Dark47 |
|
| Keratin, Reports Council Pharm. & Chem., 1911, p. 58. |
| Kinazyme (G. W. Carnrick Co.), The Journal, Nov. 1, 1913, p. 1649. |
|
|
Knowlton Danderine Company474 |
|
Koehler’s Headache Powders10 |
| Kola Compound, Elixirs (H. K. Mulford Co., Parke, Davis & Co. and Ray Chemical Co.), Reports Council Pharm. & Chem., 1912, p. 40. |
| Kolynos (Kolynos Co.), The Journal, Nov. 15, 1913, p. 1812. |
|
| Koyol (Koyol Co.), Reports Council Pharm. & Chem., 1915, p. 172. |
|
Kutnow Brothers, Ltd., and S. Kutnow316 |
|
| Kutnow’s Powder (Kutnow Bros.), The Journal, Nov. 9, 1907, p. 1619. |
| L |
|
Labordine Pharmacal Company116, 119 |
|
| Lactone (Parke, Davis & Co.), The Journal, Jan. 30, 1909, pp. 372, 397. |
|
| Lactopeptine and Elixir Lactopeptine (New York Pharmacal Association), The Journal, Oct. 23, 1915, p. 1477; Reports Council Pharm. & Chem., 1915, p. 79. |
Lactucarium, Aubergier’s Syrup of399 |
Laine Chemical Company196 |
Lancet, advertising in279 |
Lancet-Clinic, advertising in31, 342 |
Laryngoscope, advertising in35 |
|
|
|
| Lecibrin (Fairchild Bros. & Foster), Reports Council Pharm. & Chem., 1915, p. 123. |
| Lecithin, Reports Council Pharm. & Chem., 1915, p. 122. |
| Lecithin Solution (Fairchild Bros. & Foster), Reports Council Pharm. & Chem., 1915, p. 123. |
| Lecithol (Armour & Co.), Reports Council Pharm. & Chem., 1915, p. 123. |
|
| Lettuce Calmative (Nelson, Baker & Co.), Reports Council Pharm. & Chem., 1912, p. 43. |
| Lettuce, Wild, Reports Council Pharm. & Chem., 1912, p. 43. |
|
|
|
|
Lippincott’s Magazine, advertising in419 |
|
|
|
|
| Lithia and Broom Corn Compound, Elixir (Parke, Davis & Co.), Reports Council Pharm. & Chem., 1912, p. 39. |
| Lithia and Hydrangea, Elixirs (Parke, Davis & Co., Ray Chemical Co. and Smith, Kline & French Co.), Reports Council Pharm. & Chem., 1912, p. 45. |
| Lithiated Sorghum Compound (Sharp & Dohme), Reports Council Pharm. & Chem., 1912, p. 39. |
|
|
| Liver Leaf, Reports Council Pharm. & Chem., 1912, p. 43. |
Loeb’s Imported Gluten Flour450 |
Louisville Monthly Journal of Medicine and Surgery, advertising in31, 35, 300 |
Louisville Pharmacal Works410 |
Luyties’ Homeopathic Pharmacy Company50, 240, 323 |
|
|
| Lysoform and Lysoform, Crude (Lysoform Gesellschaft), The Journal, Nov. 21, 1914, p. 1870; Reports Council Pharm. & Chem., 1914, p. 126. |
|
| M |
Magistral Chemical Company328 |
| Maignen Antiseptic Powder and Maignen Institute for the Study of |
|
|
| Maizavena (William S. Merrell Chemical Co.), Reports Council Pharm. & Chem., 1912, p. 44. |
|
|
|
| Malt Extract with Pepsin and Pancreatin (Wm. S. Merrell Chemical Co., Parke, Davis & Co.), The Journal, Feb. 9, 1907, p. 533. |
| Malt, Williams’ Syrup of (American Malt Extract Co.), The Journal, Sept. 4, 1915, p. 895; Reports Council Pharm. & Chem., 1915, p. 155. |
Maltbie Chemical Company40 |
|
|
| Manaca and various manaca preparations (Hance Bros. & White, William S. Merrell Chemical Co., H. K. Mulford Co., Nelson, Baker & Co., Parke, Davis & Co., Pullen-Richardson Chemical Co., Ray Chemical Co., Sharp & Dohme, Smith, Kline & French Co., F. Stearns & Co. and Truax, Greene & Co.), Reports Council Pharm. & Chem., 1912, pp. 43, 44. |
|
|
|
|
Mariani & Co. and Mariani’s Wine221 |
| Marienbad Tablets (J. Sieke), The Journal, July 18, 1908, p. 237. |
Marmalade, Casoid Sugarless449 |
|
Maryland Medical Journal, advertising in31, 35 |
Massachusetts Medical Journal, advertising in35, 49, 300 |
| Matico Compound, Elixir (Parke, Davis & Co.), Reports Council Pharm. & Chem., 1912, p. 45. |
Mayfield Medicine Mfg. Company43 |
|
Meat extracts and meat juices123, 470 |
|
Medical Century, advertising in49, 342 |
Medical Council, advertising in49 |
Medical Fortnightly, advertising in31, 300 |
Medical Herald, advertising in31, 35, 300 |
Medical Journals and the Great American Fraud426 |
Medical Journal Advertising422 |
Medical Press and Circular, advertising in279 |
|
Medical Review, advertising in35, 300 |
Medical Review of Reviews, advertising in31, 35, 49, 300, 304 |
Medical Sentinel, advertising in31, 35, 49 |
|
Medical Summary, advertising in31, 300 |
|
Medical World, advertising in31, 35, 131 |
|
| Med-O-Lin (Waverly Oil Works Co.), Reports Council Pharm. & Chem., 1915, p. 172. |
Meister, Lucius & Bruning135 |
Memphis Medical Monthly, advertising in35 |
Menley and James, Ltd.107 |
|
Merck’s Archives, advertising in35 |
|
| Mercury Sozoiodolate and Mercury Sozoiodolate Solution, The Journal, Feb. 13, 1909, p. 573. |
|
Metcalf’s diabetic products450 |
Methyl Salicylate Ointment267 |
| Methyl-Santal (H. K. Mulford Co.), Reports Council Pharm. & Chem., 1915, p. 173. |
Metropolitan Pharmacal Co.188 |
|
Meyer, Dr. H. F. C., and Meyer’s Blood Purifier80 |
|
Micajah’s Medicated Uterine Wafers240 |
Microbe Killer, Radam’s413 |
|
|
|
| Miller’s Iod-Izd-Oil (Iodum Miller Co.), The Journal, Oct. 2, 1915, p. 1202; Reports Council Pharm. & Chem., 1915, p. 77. |
Milwaukee Medical Journal, advertising in296 |
Mineral oil, mineral glycerine161 |
| Mist. Helonin Comp. (Schlotterbeck & Foss), The Journal, Dec. 18, 1915, p. 2186. |
| Mitchella Compound (Dr. J. H. Dye Medical Institute), Reports Council Pharm. & Chem., 1912, pp. 41, 46. |
Mizer, Blake V., and Mizer Sanatorium457 |
Monthly Cyclopedia and Medical Bulletin, advertising in35 |
|
Mosquera-Julia Food Co.472 |
|
| Mother’s Cordial (Eli Lilly & Co., Ray Chemical Co.), Reports Council Pharm. & Chem., 1912, p. 46. |
| Motherwort, Reports Council Pharm. & Chem., 1912, p. 44. |
Mu-col and Mu-col Co. (Inc.)329 |
|
Muller’s tomatoes für diabetiker450 |
| Muthol (Demuth’s Laboratories), The Journal, July 10, 1915, p. 175. |
| N |
| Narcophin (Merck and Co.), The Journal, Nov. 21, 1914, p. 1872. |
|
|
Nashville Journal of Medicine and Surgery, advertising in31, 35 |
Nashville Sanitarium diabetic food products449, 450 |
|
| Nephritin (Reed & Carnrick Co.), The Journal, Oct. 5, 1907, p. 1198; Reports Council Pharm. & Chem., 1905–8, p. 79. |
| Nephroson (William S. Merrell Chemical Co.), Reports Council Pharm. & Chem., 1912, pp. 39, 45. |
Nerve Vitalizer, Wheeler’s411 |
|
|
| Neuro-Lecithin-Abbott (Abbott Laboratories), Reports Council Pharm. & Chem., 1915, p. 123. |
| Neurocaine (Schieffelin & Co.), Reports Council Pharm. & Chem., 1915, p. 173. |
|
|
New Orleans Medical and Surgical Journal, advertising in31, 35 |
New York Health Journal433 |
New York Institute of Physicians and Surgeons475 |
New York Medical Journal, advertising in31, 303, 304, 342 |
New York Pharmaceutical Association121 |
|
|
| Nose-Ions (Nose-Ions Company), The Journal, Dec. 4, 1915, p. 2026. |
|
| Nujol (Standard Oil Co. of New Jersey), The Journal, July 10, 1915, p. 175. |
|
Nutrient Wine of Beef Peptone133 |
|
Nutritive Liquid Peptone133 |
Nut products, various diabetic449, 450 |
| O |
|
| Oats, Reports Council Pharm. & Chem., 1912, p. 44. |
|
Old Dominion Journal of Medicine and Surgery, advertising in131 |
|
|
|
Orchitic Fluid Tablets317 |
| Orsudan (Burroughs, Wellcome & Co.), The Journal, April 16, 1910, p. 1323. |
Oxychlorine and Oxychlorine Chemical Co.147, 261 |
| Oxydendron Compound, Fluidextract (Nelson, Baker & Co.), Reports Council Pharm. & Chem., 1912, pp. 43, 45. |
|
| Oxyntin (Fairchild Brothers & Foster), Reports Council Pharm. & Chem., 1915, p. 174. |
|
| P |
Pacific Medical Journal, advertising in31, 279, 342 |
| Palmetto Compound (William S. Merrell Chem. Co.), Reports Council Pharm. & Chem., 1912, p. 44. |
|
|
|
Pancreatin and pepsin mixtures157, 229 |
| Pancreatin and pepsin preparations, various (Eli Lilly & Co., H. K. Mulford Co., Parke, Davis & Co., Sharp & Dohme, Smith, Kline & French Co., F. Stearns & Co. and William R. Warner & Co.), The Journal, Feb. 9, 1907, p. 533. |
| Papain, The Journal, Feb. 9, 1907, p. 522. |
|
|
| Para Coto and Paracotoin, Reports Council Pharm. & Chem., 1913, p. 39. |
Paraffin, liquid, paraffin oil161 |
Parke, Davis & Co.62, 133, 231, 267, 311, 344, 396, 410, 474, 476 |
|
|
Pas-Avena and Pas-Avena Co.333 |
|
Patch, E. L., Manufacturing Co.476 |
| Pautauberge’s Solution (Geo. J. Wallau), The Journal, March 7, 1910, p. 1560. |
|
Peacock Chemical Co. and Peacock’s Bromides28 |
|
|
Pepsin and Pancreatin Mixtures157, 229 |
| Pepsin and pepsin and pancreatin preparations, various (Columbus Pharmacal Co., Eli Lilly & Co., H. K. Mulford Co., Parke, Davis & Co., Arthur Peters & Co., Sharp & Dohme, Smith, Kline & French Co., F. Stearns & Co. and William R. Warner & Co.), The Journal, Feb. 9, 1907, p. 533. |
| Peptenzyme, Elixir and Powder (Reed & Carnrick Co.), The Journal, Feb. 9, 1907, p. 533; Oct. 5, 1907, p. 1198; Reports Council Pharm. & Chem., 1905–8, p. 79. |
|
| Peptone, Reports Council Pharm. & Chem., 1913, p. 41. |
Peptones, Liquid, and Peptonic Elixir133 |
| Perfection Liquid Food (Perfection Liquid Food Co.), Reports Council Pharm. & Chem., 1913., p. 44. |
|
Petralol, Petro, Names for Liquid Petrolatum161 |
Petrolatum, liquid, clinical experiences with167 |
Petrolatum Liquidum, Grave, and Leve, Description of166 |
Petrolax, Petrolia, Petronol, Petrosio, names for liquid petrolatum161 |
Petroleum Emulsion, Angier’s169 |
Pharmaceutical Manufacturers and the Great American Fraud474 |
Phecolates, Phecolax, Phecotones and Phecozymes174 |
Phenacetin and Acetphenetidin, their relative purity414 |
|
|
Pheno-Bromate and Pheno-Bromate Co.343 |
|
|
|
Phenolphthalein Laxative344 |
|
Phosphatometer, Dowd’s476 |
|
Phosphorus Amorphous, Pill, S. & D.482 |
Phosphorus Tonic, Comp.476 |
|
|
|
Pierce’s Favorite Prescription410 |
|
|
Pinus Canadensis, Kennedy’s47 |
| Piperazine Water (Lehn & Fink), The Journal, Feb. 29, 1908, p. 704. |
Piso’s Consumption Cure242 |
|
Pix Cresol and Pix Cresol Chemical Co.247 |
| Plasmon (Plasmon Milk Products Co.), Reports Chem. Lab., 1914, p. 88. |
Platinum Co. of America312 |
|
| Pluto Spring Water, Concentrated (French Lick Springs Hotel Co.), The Journal, March 29, 1913, p. 1013. |
|
| Ponca Compound (Mellier Drug Co.), The Journal, July 17, 1915, p. 269; Reports Council Pharm. & Chem., 1915, p. 60. |
| Poslam (Emergency Laboratories), The Journal, May 22, 1909, p. 1678; Reports Chem., Lab., 1909, p. 25. |
| Potassium Iodo-Resorcin Sulphonate, The Journal, Feb. 11, 1911, p. 441; Reports Chem. Lab., 1911, p. 21. |
Powers-Weightman-Rosengarten Co.252, 415 |
|
Practitioner, advertising in279 |
|
Proctologist, advertising in296 |
Proto Puffs, Health Food Nos. 1 and 2450 |
|
| Protonuclein and Protonuclein Beta (Reed & Carnrick), The Journal, Jan. 1, 1916, p. 48; Reports Council Pharm. & Chem., 1915, p. 90. |
|
|
Protosoy Diabetic Flour and Wafers450 |
|
|
| Pulsatilla, Reports Council Pharm. & Chem., 1912, p. 44. |
|
|
|
Pyramidon in patent medicines327 |
| Q |
| Queen of the Meadow, Reports Council Pharm. & Chem., 1912, p. 45. |
| Quina LaRoche (E. Fougera & Co.), The Journal, March 21, 1908, p. 978. |
| Quinin Arsenate, The Journal, July 16, 1910, p. 235; Reports Council Pharm. & Chem., 1910, p. 73. |
| Quinine Glycerophosphate, Reports Chem. Lab., 1912, p. 107. |
| R |
Radam’s Microbe Killer413 |
| Radelium and the Radelium Generator (Radio-Active Water Company), Reports Pharm. & Chem., 1915, p. 128. |
Rademann’s diabetic preparations449, 450 |
| Radium Solution for Intravenous Use, Standard (Radium Chemical Co.), The Journal, June 26, 1915, p. 2156; Reports Council Pharm. & Chem., 1915, p. 147. |
| Rattlesnake-Venom, The Journal, March 15, 1913, p. 850; March 29, 1913, p. 1001; June 7, 1913, p. 1811. |
|
|
|
Renne’s Pain Killing Oil205 |
Renova Distributing Co.434 |
|
|
| Respirazone (Tilden Company), The Journal, June 14, 1913, p. 1899. |
Rex Brand Beef Extract471, 472 |
| Rheumalgine (Eli Lilly & Co.), The Journal, June 26, 1915, p. 2156; Reports Council Pharm. & Chem., 1915, p. 148. |
| Rheumatic Bacterin (Mixed) No. 47, Swan’s (Swan-Myers Co.), The Journal, Nov. 6, 1915, p. 1662; Reports Council Pharm. & Chem., 1915, p. 160. |
Richards, John Morgan, and Sons121 |
|
Richardson’s Life-Preserving Bitters43 |
| Ricinol-Grape Tape-Worm Remedy and Baby Taeniafuge-Grape (Grape Capsule Co.), Reports Council Pharm. & Chem., 1915, p. 174. |
|
Rio Chemical Company43, 49 |
|
|
|
Royal Neighbor, clean advertising in418 |
|
Rusks, Callard’s Casoid450 |
Russian Mineral oil, Russian paraffin oil161 |
| S |
St. Paul Medical Journal, advertising in35 |
|
|
|
| Salesthyl and Sal-Hyl (New York Salesthyl Corporation), The Journal, Feb. 20, 1915, p. 684; Reports Council Pharm. & Chem., 1915, p. 134. |
Saliodin and Saliodin Chemical Co.249 |
| Salit (Heyden Chemical Works), The Journal, June 5, 1909, p. 1852; Reports Council Pharm. & Chem., 1909, p. 106. |
|
Sanatogen and Medical Journals431 |
| Sanguinarine Nitrate, Reports Council Pharm. & Chem., 1911, p. 59. |
|
Sanol Anti-Diabetes and Sanol Manufacturing Co., Ltd.299 |
|
Saxol, Liquid Saxoline161 |
|
| Scopolamin-Morphin Mixtures, The Journal, Feb. 5, 1910, p. 446; Feb. 12, 1910, p. 516; June 7, 1913, p. 1814; Reports Council Pharm. & Chem., 1910, p. 11. |
|
| Scullcap Compound, Fluidextract (Parke, Davis & Co.), Reports Council Pharm. & Chem., 1912, p. 43. |
|
Secretogen Elixir and Tablets185 |
| Secretogen and Elixir Secretogen (G. W. Carnrick Co.), The Journal, Jan. 15, 1916, p. 178; Reports Council Pharm. & Chem., p. 99. |
| Sedobrol “Roche” (Hoffman-LaRoche Chemical Works), The Journal, Jan. 2, 1915, p. 71; Reports Council Pharm. & Chem., 1914. |
| Selenium compounds (in “Chemotherapy and Tumors”), The Journal, April 17, 1915, p. 1283; Reports Council Pharm. & Chem., 1915, p. 28. |
| Semprolin (W. Browning & Co.), The Journal, July 10, 1915, p. 175. |
|
| Seng (Sultan Drug Co.), Reports Council Pharm. & Chem., 1915, p. 129. |
|
| Serum Vaccine, Bruschettini (R. G. Berlingieri), The Journal, Nov. 21, 1914, p. 1870; Reports Council Pharm. & Chem., 1914, p. 127. |
| Seven-Bark, Reports Council Pharm. & Chem., 1912, p. 45. |
|
|
|
|
| Sirolin (Sirolin Co.), The Journal, June 21, 1913, p. 1974. |
Smith Bile Beans Co. and Smith’s Bile Beans43 |
|
Smith, Kline and French Co.410 |
Smith, Martin H., Co.82, 88 |
Society of Chemical Industry178 |
|
|
|
| Sourwood and Sourwood Elixirs (Eli Lilly & Co. and Parke, Davis & Co.), Reports Council Pharm. & Chem., 1912, p. 45. |
South Texas Medical Record, creditable advertising in440 |
Southern Practitioner, advertising in31, 35, 279 |
Southwest Journal of Medicine and Surgery, advertising in300 |
|
Spencer’s Almond Paste450 |
|
| Squaw Root, Reports Council Pharm. & Chem., 1912, p. 40. |
| Squaw-vine and squaw-vine preparations (Eli Lilly & Co., Parke, Davis & Co.), Reports Council Pharm. & Chem., 1912, p. 45. |
|
| Standard Radium Solution for Intravenous Use (Radium Chemical Co.), The Journal, June 26, 1915, p. 2156; Reports Council Pharm. & Chem., 1915, p. 147. |
|
| Stearns’ Wine (Frederick Stearns & Co.), Reports Council Pharm. & Chem., 1915, p. 177. |
|
Stevenson & Jester Co.133 |
| Stillingia preparations, various (Hance Bros. & White, H. K. Mulford Co., Parke, Davis & Co., Ray Chemical Co. and Smith, Kline & French Co.), Reports Council Pharm. & Chem., 1912, pp. 42, 47. |
| Stone Root, Reports Council Pharm. & Chem., 1912, p. 46. |
|
| Strychnin Arsenate, The Journal, Sept. 24, 1910, p. 1128; Reports Council Pharm. & Chem., 1910, p. 74. |
|
Succus Cineraria Maritima (Walker)50 |
| Sugar Cane, Chinese, Reports Council Pharm. & Chem., 1912, p. 39. |
|
|
|
|
Swain’s All-Healing Ointment and Swain’s Laboratory43 |
| Swan’s Rheumatic Bacterin (Mixed) No. 47 (Swan-Myers Co.), The Journal, Nov. 6, 1915, p. 1662; Reports Council Pharm. & Chem., 1915, p. 160. |
|
|
Syrup of Cocillana Compound396 |
Syrup of Hydriodic Acid, Gardner’s97 |
Syrup of Lactucarium, Aubergier’s399 |
| Syrup of Malt, Williams’ (American Malt Extract Co.), The Journal, Sept. 4, 1915, p. 895. |
|
| T |
| Tablogestin (F. H. Strong Co.), The Journal, Dec. 11, 1915, p. 2108. |
Taka-Diastase and Liquid Taka-Diastase62 |
| Tannyl (Charles Goslar), Reports Chem. Lab., 1912, p. 108. |
Tartarlithine and Tartarlithine Co.401 |
Taurocol Tablets and Compound Tablets198 |
|
Terralbolia, Terraline, names for liquid petrolatum161 |
Texas Medical Journal, advertising in31, 35, 49 |
Texas Medical News, advertising in31, 35 |
|
Theobromin Sodium Salicylate versus Diuretin251 |
Therapeutic Gazette, advertising in31, 35 |
Therapeutic Record, advertising in279, 300 |
|
| Thiocol and Syrup Thiocol-Roche (Hoffmann-La Roche Chemical Works), The Journal, June 21, 1913, p. 1974; Reports Council Pharm. & Chem., 1913, p. 16. |
| Thioform (Otto Hann & Bro.), Reports Chem., Lab., 1909, p. 79. |
Thompson’s Malted Food Co.319 |
|
Three Chlorides (Henry)198, 201 |
|
Toledo Medical and Surgical Reporter, advertising in296 |
| Tonga and various tonga preparations (Hance Bros. & White, Eli Lilly & Co., William S. Merrell Chemical Co., Nelson, Baker & Co., Parke, Davis & Co., Ray Chemical Co., Sharp & Dohme, F. Stearns & Co., William R. Warner & Co. and H. K. Wampole & Co.), The Journal, May 10, 1913, p. 1478; Reports Council Pharm. & Chem., 1912, p. 46. |
| Tongaline and Tongaline Tablets (Mellier Drug Co.), The Journal, July 17, 1915, p. 269; Reports Council Pharm. & Chem., 1915, p. 58. |
|
| Toxinol (Hughes Chemical Co.), Reports Council Pharm. & Chem., 1912, p. 39. |
|
| Trifolium preparations, various (Hance Bros. & White, Eli Lilly & Co., William S. Merrell Chemical Co., H. K. Mulford Co., Parke, Davis & Co., Ray Chemical Co. and F. Stearns & Co.), Reports Council Pharm. & Chem., 1912, pp. 39, 40. |
| Trilene Tablets, Reports Council Pharm. & Chem., 1912, p. 39. |
| Trophonine (Reed & Carnrick Co.), The Journal, Oct. 5, 1907, p. 1198; Reports Council Pharm. & Chem., 1905–8, p. 79. |
|
|
| Tubercle Vaccine, Non-Virulent, Sherman’s (G. H. Sherman), The Journal, Nov. 21, 1914, p. 1870; Reports Council Pharm. & Chem., 1914, p. 128. |
| Tuberculin Test Plate, Keller’s (A. H. Keller), The Journal, Dec. 19, 1914, p. 2250. |
| Tuberculoids (Columbus Pharmacal Company), The Journal, Feb. 22, 1908, p. 704. |
| Tubo-Arg (Tubo Pharmacal Co.), Reports Council Pharm. & Chem., 1915, p. 175. |
| Turkey Corn, Reports Council Pharm. & Chem., 1912, p. 47. |
Turner, F. M., and The Dr. Turner Co.211 |
|
Tyree’s Antiseptic Powder21, 404 |
Tyree’s Elixir Buchu and Hyoscyamus Comp.407 |
| Tyree’s Elixir of Buchu and Hyoscyamus Compound (J. S. Tyree), Reports Council Pharm. & Chem., 1915, p. 167. |
| U |
| Ulax Salt (F. H. Strong Co.), Reports Council Pharm. & Chem., 1915, p. 175. |
|
Unguentum Selenio Vanadic (v. Roemer)207 |
|
United States Health Reports433 |
|
| Uranoblen (A. Grimme), Reports Council Pharm. & Chem., 1915, p. 176. |
| Urasol (Organic Chemical Mfg. Co.), The Journal, Sept. 5, 1908, p. 818; May 8, 1909, p. 1511; Reports Council Pharm. & Chem., 1905–8, p. 166; 1909, p. 64. |
|
|
| Uricsol (Uricsol Chemical Co.), The Journal, Aug. 14, 1915, p. 638; Reports Council Pharm. & Chem., 1915, p. 149. |
|
| Urodonal (Geo. J. Wallau, Inc.), The Journal, Aug. 14, 1915, p. 639; Reports Council Pharm. & Chem., 1915, p. 153. |
| Uron (Uron Chemical Company), The Journal, Nov. 3, 1906, p. 1500; Reports Council Pharm. & Chem., 1905–8, p. 26. |
|
Uterine Sedative Elixir410 |
| Uterine Sedative, Elixir (Eli Lilly & Co.), The Journal, Aug. 31, 1912, p. 735; Reports Council Pharm. & Chem., 1912, p. 44. |
| Uterine tonics, various (Abbott Alkaloidal Co., Girard Chemical Co., Maltbie Chemical Co., H. K. Mulford Co., Nelson, Baker & Co., Parke, Davis & Co. and F. Stearns & Co.), Reports Council Pharm. & Chem., 1912, pp. 41, 46. |
Uterine Wafers, Micajah’s Medicated240 |
| V |
| Vaccine, Curative, Bruschettini (A. Bruschettini), Reports Council Pharm. & Chem., 1915, p. 176. |
| Vaccine, Friedmann’s (Standard Distributing Co.), Reports Council Pharm. & Chem., 1914, p. 136. |
|
| Vaccine, Non-Virulent Tubercle Bacillus (G. H. Sherman), The Journal, Sept. 20, 1913, p. 979. |
|
|
Valentine’s Meat Juice Co.472 |
| Vanadic Acid, The Journal, May 9, 1908, p. 1548; July 24, 1909, p. 309. |
| Vanadiol, Vanadioseptol and Vanadium Solution for Intravenous |
|
| Vanadium, The Journal, June 3, 1911, p. 1648. |
|
|
|
|
|
|
|
|
|
Vermont Medical Monthly, advertising in35 |
| Veroform Germicide (Veroform Hygienic Co.), The Journal, Nov. 22, 1913, p. 1920. |
Vial, New York Laboratories of59 |
| Viburn-Ovaro (Ray Chemical Co.), Reports Council Pharm. & Chem., 1912, p. 46. |
Viburnum Compound, Elixir of410 |
Viburnum Compound, Hayden’s218, 409 |
| Viburnum preparations, various (Fraser Tablet Co., Parke, Davis & Co. and Smith, Kline & French Co.), Reports Council Pharm. & Chem., 1912, p. 46. |
|
|
|
|
|
| Vinum Ext. Morrhuae, Stearns (Frederick Stearns & Co.), Reports Council Pharm. & Chem., 1915, p. 177. |
Virginia Medical Semi-Monthly, advertising in131 |
|
|
| W |
|
|
Wallau, George J., Inc.106 |
Wampole, Henry K., & Co., Inc.26, 52 |
Wampole’s Perfected and Tasteless Preparation of an Extract of Cod Liver52, 443 |
|
| Water Eryngo, Reports Council Pharm. & Chem., 1912, p. 47. |
|
|
Western Canada Medical Journal, advertising in300 |
Wheeler Chemical Works245 |
Wheeler’s Nerve Vitalizer411 |
| Whiteruss (Lubric Oil Co.), The Journal, July 10, 1915, p. 175. |
| White Sulphur Salts (White Sulphur Springs, Inc.), The Journal, Nov. 21, 1914, p. 1870; Reports Council Pharm. & Chem., 1914. |
Whiting’s Sugar-Free Milk449 |
|
|
|
| Williams’ Syrup of Malt (American Malt Extract Co.), The Journal, Sept. 4, 1915, p. 895; Reports Council Pharm. & Chem., 1915, p. 155. |
|
| Wine of Cod Liver Ext. with Peptonate of Iron, Stearns’ (Frederick Stearns & Co.), Reports Council Pharm. & Chem., 1915, p. 177. |
Wisconsin Medical Recorder, advertising in31, 35 |
Woman’s Medical Journal, advertising in31, 49 |
|
|
| X |
| Xanol (Wm. S. Merrell Chemical Co.), Reports Council Pharm. & Chem., 1911, p. 64. |
| Y |
|
| Yogurt (Yogurt Co.), The Journal, Jan. 30, 1909, pp. 372, 397. |
| Z |
|
|
|
|
|
Return to transcriber’s notes
Spelling inconsistencies:
quinin/quinine
soluble/solubile/soluable
glucosid/glucoside
peroxid/peroxide
oxid/oxide
Hydrastin/Hydrastine
small-pox/smallpox
protochlorid/protochloride
narcotin/narcotine
anilpyrine/anilipyrine
vender/vendor
chemic/chemical
strychnin/strychnine
pseudo-scientific/pseudoscientific
semi-secret/semisecret
beta-naphthol/betanaphthol
to-day/today
acetyl-salicylic/acetysalicylic
indorse/endorse
indorsement/endorsement
gm./Gm.
Spelling corrections:
folowing —> following
wafer —> water
Amedemin —> Anedemin
Prepration —> Preparation
he —> the
decription —> description
administation —> administration
oven —> even
test —> tests
constitutent —> constituent
iutensive —> intensive
accurary —> accuracy
Helionas —> Helonias
Phamacopeia —> Pharmacopeia
cancerns —> concerns
repiration —> respiration
commitee —> committee
ampoules —> ampules
fradulent —> fraudulent
Thp —> The
Diastate —> Diastase
diminshed —> diminished
acetphenentidin —> acetphenetidin
1,169 —> 1,669 (in table)
effciency —> efficiency
that —> than
thought —> bought
Furthemore —> Furthermore
bareley —> barley
acetphentidin –> acetphenetidin
prramidon —> pyramidon
valuless —> valueless
containg —> containing
American —> America
article —> articles
nessary —> necessary
out —> our
strotium —> strontium
sercretin —> secretin
radicles —> radicals
Speciment —> Specimen
atained —> attained
wo —> woe
wounders —> wonders
lives —> livers
matiére —> matière
Geseundhitslehrer —> Geseundheitslehrer
readibly —> readily
alkoloids —> alkaloids
is —> it
methol —> menthol
amyolytic —> amylolytic
Return to transcriber’s notes
*** END OF THE PROJECT GUTENBERG EBOOK 47766 ***





















































