
Project Gutenberg's Paint Technology and Tests, by Henry A. Gardner This eBook is for the use of anyone anywhere at no cost and with almost no restrictions whatsoever. You may copy it, give it away or re-use it under the terms of the Project Gutenberg License included with this eBook or online at www.gutenberg.org Title: Paint Technology and Tests Author: Henry A. Gardner Release Date: September 13, 2011 [EBook #37420] Language: English Character set encoding: ISO-8859-1 *** START OF THIS PROJECT GUTENBERG EBOOK PAINT TECHNOLOGY AND TESTS *** Produced by Chris Curnow, Harry Lamé and the Online Distributed Proofreading Team at https://www.pgdp.net (This file was produced from images generously made available by The Internet Archive)
Please see Transcriber's Notes at the end of this document

BY
HENRY A. GARDNER
Assistant Director, The Institute of Industrial Research,
Washington, D. C.
Director, Scientific Section, Paint Manufacturers’ Association
of the United States, etc.
McGRAW-HILL BOOK COMPANY
239 WEST 39TH STREET, NEW YORK
6 BOUVERIE STREET, LONDON, E.C.
1911
Copyright, 1911, by the McGraw-Hill Book Company
THE·PLIMPTON·PRESS·NORWOOD·MASS·U·S·A
TO
MY MOTHER
A few years ago the producer and consumer of paints possessed comparatively little knowledge of the relative durability of various pigments and oils. There existed in some cases a prejudice for a few standard products, that often held the user in bondage, discouraging investigation and exciting suspicion whenever discoveries were made, that brought forth new materials. Such conditions indicated to the more progressive, the need of positive information regarding the value of various painting materials, and the advisability of having the questions at issue determined in a practical manner.
The desire that such work should be instituted, resulted in the creation of a Scientific Section, the scope of which was to make investigations to determine the relative merits of different types of paint, and to enlighten the industry on various technical problems. Paint exposure tests of an extensive nature were started in various sections of the country where climatic conditions vary. This field work was supplemented in the laboratory by a series of important researches into the properties of pigments, oils, and other raw products entering into the manufacture of protective coatings. The results of the work were published in bulletin form and given wide distribution. The demand for these bulletins early exhausted the original impress, and a general summary therefore forms a part of this volume.
The purpose of the book is primarily to serve as a reference work for grinders, painters, engineers, and students; matter of an important nature to each being presented. Without repetition of the matter found in other books, two chapters on raw products have been included, and they present in condensed form a summary of information that will prove of aid to one who desires to become conversant with painting materials with a view to continuing tests such as are outlined herein. In other chapters there has been compiled considerable matter from lectures and technical articles presented by the writer before various colleges, engineering societies, and painters’ associations.
[viii]The writer wishes to gratefully acknowledge the untiring efforts of the members of the Educational Bureau of the Paint Manufacturers’ Association, whose early endeavors made possible many of the tests described in this volume. Kind acknowledgment is also made to members of the International Association of Master House Painters and Decorators of the United States and Canada, who stood always ready to aid in investigations which promised to bring new light into their art and craft.
HENRY A. GARDNER.
Washington, October, 1911.
| CHAPTER | PAGE | |
| I | Paint Oils and Thinners | 1 |
| II | A Study of Driers and Their Effect | 21 |
| III | Paint Pigments and Their Properties | 42 |
| IV | Physical Laboratory Paint Tests | 70 |
| V | The Theory and Practice of Scientific Paint Making | 93 |
| VI | The Scope of Practical Paint Tests | 105 |
| VII | Conditions Noted at Inspection of Tests | 114 |
| VIII | Results of Atlantic City Tests | 124 |
| IX | Results of Pittsburg Tests | 135 |
| X | A Laboratory Study of Test Panels | 149 |
| XI | Additional Tests at Atlantic City and Pittsburg | 174 |
| XII | North Dakota Paint Tests | 182 |
| XIII | Tennessee Paint Tests | 201 |
| XIV | Washington Paint Tests | 207 |
| XV | Cement and Concrete Paint Tests | 214 |
| XVI | Structural Steel Paint Tests | 220 |
| XVII | The Sanitary Value of Wall Paints | 252 |
Constants and Characteristics of Oils and Their Effect upon Drying. An attempt has been made to give in this chapter a brief summary of the most important characteristics of those oils finding application in the paint and varnish industry. For methods of oil analysis, the reader is referred to standard works on this subject; the analytical constants herein being given only for comparative purposes.
It is well known that one of the most desirable features of a paint oil is the ability to set up in a short period to a hard surface that will not take dust. This drying property is dependent upon the chemical nature of the oil. If it is an unsaturated compound, like linseed oil, rapid absorption of oxygen will cause the film to dry rapidly and become hard. If the oil be of a fully satisfied nature, like mineral oil, oxygen cannot be taken up to any great extent and drying will not take place. The various animal and vegetable oils differ in their power of oxygen absorption to a lesser or greater extent. This difference is referred to by the chemist in terms of the iodine value. The iodine value of linseed oil is approximately 190, meaning that one gram of the oil will take up 190 centigrams of iodine. Oils with high iodine values have good drying powers, while those with low iodine values are, as a rule, very slow drying in nature.
For a description of the working and drying properties of various oils used in paints, see Chapter XIV. The oxygen absorption of various oils and mixtures is shown in Chapter II.
Linseed Oil. The seed of the flax plant which is extensively grown in North Dakota, Argentine Republic and Russia, contains approximately 36% of oil which may be obtained by grinding, heating, and expression. Ripe native seed generally produces[2] a pale oil of little odor; the oil from Argentine seed often having a greenish tint and an odor resembling sorghum. While filtering, pressing and ageing will remove considerable of the (“foots”) mucilaginous matter, phosphates, silica, etc., from the oil, the better grades which are intended for varnish making are often refined with sulphuric acid. A light colored oil which may be heated without “breaking” results from this treatment, but such oils are apt to contain considerable free fatty acid, unless they are washed with alkali subsequent to the sulphuric acid treatment. On account of its rapid drying properties and general adaptability for all classes of paints and varnishes, linseed oil has never been supplanted by any other oil. Chemically it consists of the glycerides of linoleic, oleic, and isolinoleic acid, its constitution being responsible for its very high iodine value.
Boiled linseed oil, a heavier and darker product, is made by heating the raw oil in open kettles to high temperatures, generally with the addition of metallic driers such as litharge, and black manganese. The resinates of lead and manganese are often added to oil heated at a lower temperature, to obtain a boiled oil of lighter color.

New type of Flax Harvester which pulls plant up by the roots, thus preventing infection of soil

Modern Concrete Elevators for storing Flaxseed

View of Linseed Oil Factory showing hydraulic press, tanks, etc.

Photographs courtesy of Spencer Kellogg Sons
Flaxseed Crushers

Filter Presses for removing extraneous matter from linseed oil

Linseed Cake from Oil Press

Glycine Hispida
Mammoth soya bean plants

Photographs courtesy of David Fairchild, Plant Explorer, U. S. Dept. of Agriculture
Glycine Hispida
Soya bean plants under cultivation at Arlington, Va.
By blowing air through linseed oil that has been heated to approximately 200 degrees Fahrenheit, either with or without drier, heavy bodied oils are obtained, which find special application in varnishes and technical paints. As the viscosity of these oils increase, the iodine values decrease, and a slight rise in [7]saponification value and specific gravity is observed. The following analyses of various types of linseed oil were recently made by the writer:
| Pure Raw Linseed Oil |
Boiled L. O. (Linoleate) |
Boiled L. O. (Resinate) |
Blown L. O. |
Litho. L. O. |
Old Treated Oil |
|
| Color | Amber | Dark | Reddish | Pale | Dark | Amber |
| Clear | Brown | Brown | Brown | Clear | ||
| Sp. Gr. at 15° C. | .933 Average |
.941 | .930 | .968 | .970 | .943 |
| Iodine No. | 180 | 172 | 176 | 133 | 102 | 172 |
| Saponification No. | 191 | 187 | 186 | 189 | 199 | 197 |
| Free Fatty Acid | 3.2 | 2.7 | 2.2 | 2.8 | 2.7 | 6.9 |
| Unsaponifiable | 1.4 | — | — | — | — | 1.8 |
| Maumene | 111 | — | — | — | — | 96 |
| Moisture | .2% | — | — | — | — | none |
Soya Bean Oil. The soya plant which is extensively cultivated in Asia produces a seed bearing up to 22% and over of a golden colored oil having a peculiar leguminous odor. The oil, which probably consists of the glycerides of oleic, linoleic, and palmitic[8] acids, is secured by crushing, steaming and pressing the seed. There are several varieties of the plant, and they are said to be the best annual legume for forage, the straw and fruit being rich in nitrogen and very fattening as a cattle food. Soya may be grown in nearly any country and is a great carrier of nitrogen to land deficient in this element. Although the oil has been used abroad for many years for soap-making purposes, its use as a drying oil is comparatively recent; being introduced into the paint industry of the United States during the year 1909, when linseed oil started on its phenomenal rise in price.
The oil has given fair service in some paints when mixed with upwards of 75% of pure linseed oil. It is of a semi-drying nature, but may be made to dry rapidly when mixed with manganese and lead linoleate driers. By compounding it under heat with tung oil and rosin, a substitute for linseed oil is produced, which some claim to be quite valuable.
Table I gives the constants of several samples of soya oil examined by the writer. Table II shows the iodine value of mixtures of soya and linseed oils. Table III shows the results of drying experiments on soya oils containing different percentages of lead and manganese driers.
| Sample No. | Specific gravity | Acid No. | Saponification No. |
Iodine No. | Per cent. of foots |
| 1 | 0.9233 | 1.87 | 188.4 | 127.8 | 3.81 |
| 2 | 0.9240 | 1.92 | 188.3 | 127.2 | — |
| 3 | 0.9231 | 1.90 | 187.8 | 131.7 | — |
| 4 | 0.9233 | 1.91 | 188.4 | 129.8 | — |
| 5 | — | — | — | 130.0 | — |
| 6 | — | — | — | 132.6 | — |
| 7 | — | — | — | 136.0 | — |
| Average | 0.9234 | 1.90 | 188.2 | 130.7 | — |
| Sample No. | Straight linseed |
Soya 25 per cent. Linseed 75 per cent. |
Soya 50 per cent. Linseed 50 per cent. |
Soya 75 per cent. Linseed 25 per cent. |
| 1 | 190.3 | 175.2 | 160.7 | 140.4 |
| 2 | 189.5 | 175.9 | 161.7 | 140.8 |
| 3 | 188.0 | 175.4 | 160.3 | 139.0 |
| Average | 189.3 | 175.5 | 160.9 | 140.4 |
| Per cent. PbO |
0.05 | 0.10 | 0.30 | 0.50 | 0.70 | 1.00 | 1.30 | 1.60 | |||
| ⎧ | 1 | day | — | 0.07 | 0.63 | 1.34 | 1.05 | 1.53 | 0.93 | 1.35 | |
| ⎪ | 3 | days | — | 0.07 | 3.52 | 4.31 | 2.75 | 4.86 | 4.82 | 4.12 | |
| ⎪ | 5 | days | — | 0.09 | 5.04 | 6.06 | 6.09 | 6.75 | 6.66 | 5.52 | |
| Per ct. gain | ⎨ | 12 | days | — | — | 6.88 | 7.54 | 7.43 | 7.76 | 7.32 | 6.47 |
| ⎪ | 15 | days | — | — | 8.84 | 8.93 | 8.59 | 8.81 | 8.44 | 7.46 | |
| ⎩ | 20 | days | 0.05 | 0.20 | 9.02 | 9.08 | 8.90 | 9.03 | 8.65 | 7.83 | |
| Per cent. MnO2 | 0.01 | 0.05 | 0.15 | 0.26 | 0.30 | |||
| Per ct. gain | ⎧ | 1 | day | — | — | 0.02 | 0.02 | 0.01 |
| ⎨ | 10 | days | — | 5.06 | 6.48 | 6.10 | 5.97 | |
| ⎩ | 20 | days | 0.05 | 9.07 | 8.80 | 6.78 | 6.51 | |
| Per cent. PbO | 0.20 | 0.30 | 0.50 | |||
| MnO2 | 0.05 | 0.15 | 0.25 | |||
| Per ct. gain | ⎧ | 1 | day | 3.04 | 3.77 | 3.74 |
| ⎨ | 8 | days | 5.96 | 6.43 | 6.47 | |
| ⎩ | 12 | days | 6.33 | 6.78 | 6.67 | |
Tung Oil. There are grown in China and Japan many varieties of the “aleurites cordata,” popularly known as the tung tree. This tree bears great quantities of large sized nuts containing as high as 40% of an oil which yields itself in a viscous yellow form upon heating and crushing of the fruit. The raw oil, which chemically consists of the glycerides of oleic, oleo-margaric, and probably isomeric acids, is distinguished by its rapid drying properties. When spread in a thin layer it produces a hard film with an opaque frosted surface, often showing a tendency to wrinkle. Treated tung oil will dry to a clear, water-shedding, elastic film. This oil is made by heating the raw tung oil at a comparatively low temperature with other oils and a metallic drier such as litharge.

Photographs courtesy of David Fairchild
Aleurites Cordata (Chinese Wood Oil) Barrel Factory at Cooperage Shop

Photographs courtesy of David Fairchild
Aleurites Fordii (Chinese Wood Oil)
Fruit from trees at the end of fourth year
The affinity of tung oil for rosin has resulted in the production of a series of moderate-priced varnishes most suitable for use in floor and deck paints or wherever great hardness is required. These varnishes are also finding application in the manufacture of concrete, steel, and flat wall paints; being especially suitable for the above purposes when compounded with kauri gum japan.

Aleurites Fordii
Wood Oil tree, thirty feet high and three feet in diameter, on
banks of Yangtse River, Western Szechuan, China. Opium Poppy in the foreground

Aleurites Cordata
Flowering specimen of the Chinese Wood Oil tree at Riverside, California,
planted in 1907. Photograph taken in 1910, when tree had borne fifty fruits
During the boiling of raw tung oil the temperature must not exceed much over 400 degrees Fahrenheit. Otherwise a peculiar “hamming” will take place, the whole mass becoming solid and of no further value as a varnish or paint vehicle. Some peculiar internal disturbance or rearrangement of the molecules is evidently effected by heat, and although the reaction is not clearly understood, it has been ascribed to auto-polymerization. Scott has stated that the phenomenon of gelatinization is due to the exposure of the surface of the oil to the air, and that boiling in vacuo obviates such results. The lusterless surface produced when tung oil varnishes are dried in vitiated air would tend to confirm the conclusion that the oil is very subject to atmospheric influences.
[12]Lumbang Oil, which is obtained from a tropical species of Tung, is very similar in appearance and properties to Linseed Oil.
| Sp. Gr. | Iodine No. | Saponifi- cation No. |
Acid No. | |
| No. 1 | .944 | 166 | 188 | 3.6 |
| No. 2 | .940 | 164 | 184 | 1.8 |

Photographs courtesy Alpin I. Dunn
Menhaden Net drying in the Sun

A big catch of Menhaden made off Narragansett Bay
Menhaden Oil. Of all the marine-animal oils, such as seal, herring, sardine, whale, and menhaden, the latter is the most valuable. It is produced by steam digestion and pressure of the menhaden or “piogey” fish, which are caught in great quantities off the Atlantic Coast. Prompt cooking and treatment of the fish results in a light-colored oil having very little odor, the residue left in the presses being of great value as a fertilizer. Although several grades of oil termed crude, brown, light, etc., are produced, the most satisfactory for use in paint is that grade termed “light winter pressed.” This oil is of a pale straw color and has a high iodine number which is responsible for its rapid drying value. It contains less of the stearates that precipitate from crude oil, but sufficient to render its film water-shedding and elastic. The presence of too great a quantity of stearates is apt to result in a[14] very soft film, and the use of hard driers, such as the metallic tungates, is therefore advisable with menhaden oil. When mixed with linseed oil paints the odor of menhaden oil is sometimes noticeable, but it disappears entirely after such paints are applied. Its use with linseed oil in technical paints exposed to the salty air of the Coast has given good results, often preventing “checking” and “chalking.”
The following constants were determined on samples of menhaden oil received in the writer’s laboratory:
| Sp. Gr. | Iodine Value |
Saponification Number |
Acid Number |
|
| Light | .927 | 175.8 | 187.9 | 7.55 |
| Medium | .925 | 178.7 | 187.6 | 6.19 |
| Dark | .927 | 178.0 | 187.3 | 7.19 |
Whale Oil. While ordinary whale oil is too dark and odorous to ever come into extensive use as a paint oil, it is probable that the refined oil will be utilized in the manufacture of certain technical paints. Whale oil is boiled from chopped whale blubber, the first trying being the lightest in color, while the later tryings, as well as the product made from bones, are of darker color and of very bad odor. Oil of mirbane is often used to mask this odor. The oil contains large quantities of stearin and palmitin, as well as wax-like constituents which are apt to be thrown out of solution in very cold weather, or when the oil is mixed with other oils. The refined oil, when ground with lead and zinc pigments and mixed with equal parts of linseed oil and treated tung oil, dries to an elastic and soft film. Experiments are being made to utilize whale oil in the linoleum industry.
The analyses of samples of whale oil tested by the writer are as follows:
| Sp. Gr. | Iodine Value |
Saponification Number |
Free Fatty Acid |
||
| Light Refined | .924 | 148 | 190 | .2 | 1.2 |
| Dark Yellow | .920 | 142 | 187 | 7.0 | |
| Dark Brown | .910 | 140 | 184 | 18.0 | |
Sunflower Oil. Sunflower oil is produced largely in Russia and Hungary, finding favor in those countries as an edible oil.[15] The ripe seeds of the sunflower plant contain over 30% of oil which is very pale in color and of a pleasant smell. It has been found that sunflowers may be grown to advantage in dry parts of the United States, and if suitable yields are obtained from a few experimental acres now being cultivated, the industry may receive encouragement in this country. The oil should be well suited for varnish making, and although the iodine number is not very high, it dries quite rapidly.
| Sp. Gr. | Iodine No. | Saponifica- tion No. |
Acid No. |
| .929 | 128 | 188 | 4 |
Cottonseed Oil. This oil is expressed from the seed of the cotton plant, varying in color according to the time of its pressing and degree of refinement. Being edible as well as highly suited for soap making, very little of it comes into the market as a paint oil. It contains large quantities of stearin and has a low iodine value, making it a slow drying oil. Some samples are extremely light in color and contain less mucilaginous matter and foots than is present in ordinary varieties.
| Sp. Gr. | Iodine No. | Saponifica- tion No. |
Acid No. |
| .922 | 106 | 190 | 2.4 |
[16]Corn Oil. As a by-product in the manufacture of starch and alcoholic liquids, this material comes into the market having a golden yellow color, and an odor resembling fermented grain. It has a lower drying value than cottonseed oil, and its use in the paint industry will probably be limited to color grinding, where an oil with a semi-drying value is often desired. Like cottonseed oil, it belongs more properly to the soap oil class. It contains glycerides of linoleic and especially palmitic acid.
| Sp. Gr. | Iodine No. | Saponification No. | Acid No. |
| .925 | 118 | 191 | 9.5 |
Rosin Oil. By the dry distillation of rosin, there is yielded a series of heavy dark oils consisting principally of hydrocarbons, resinous bodies, and free acid. These oils vary in their saponification number from 10 to 60, while their unsaponifiable value averages about 80. Of the grades termed first, second, third, and fourth run, the latter two are superior for use in paints, as a rule containing less free acid than the preliminary runs. Treatment with steam and alkali serve to neutralize the acid nature of the oils and to remove impurities. Refined oils are lighter in color and are often blown and bodied to fairly rapid drying products, especially when treated with manganese driers. Rosin oils are seldom used with lead pigments, on account of the presence of sulphur in the oils, which would result in darkening. Rosin oil paints work very smoothly, even when they are curdled, producing glossy surfaces. The rapid checking of rosin oil paints on wooden surfaces bars the use of this oil for such purposes.
| Sp. Gr. | Iodine Value |
Saponifica- tion No. |
Acid No. | ||
| A | .96 | 6 | 41 | 27 | 16.7 |
| B | .99 | 48 | 38 | 10.0 | |
Hydrocarbon Oils. Several grades of neutral or mineral oils, varying somewhat in gravity, color, and quality, are produced[17] as the last distillate in the refining of petroleum. These oils when mixed with drying oils and strong driers find application in the manufacture of some freight-car, barn, and other paints which sell at a low price. A small percentage of mineral oil is said to be valuable in structural steel paints, acting as a preventative of hard drying and thus keeping the film soft and elastic. Streaking and sweating is apt to ensue if any great quantity is used. Mineral oils have a characteristic bloom, showing a greenish fluorescence when examined by transmitted light. This bloom is due to the presence of some strongly fluorescent material which is shown up with intensity when mineral oils are exposed to ultraviolet rays such as emanate from an enclosed arc light. Outerbridge[1] first proposed this test for mineral oils, and he has worked out a “fluorescent scale,” by which very small percentages of hydrocarbon oils may be detected in other oils. Several types of so-called debloomed oil have been placed upon the market, and although such oils appear under ordinary light conditions to be free from bloom, they fluoresce quite strongly when given the Outerbridge test.
[1] Alexander E. Outerbridge, Jr.: “A Novel Method of Detecting Mineral Oil and Resin Oil in Other Oils.” Proc. 14th Annual Meet., Amer. Soc. for Testing Mater., Atlantic City, N.J., June 28, 1911.

View of Stills Where Petroleum Paint Thinners are Manufactured (Waverly)
| Sp. Gr. | Iodine No. | Saponification No. | Acid No. |
| .92 | 12 | 4 | 0 |
[2] Oil of mirbane present, probably as a deblooming agent, or to mask the odor.
Pine Oil. This oil is produced by the redistillation of the heavy, high boiling point fractions resulting from the steam distillation of wood turpentine. It is a heavy straw-colored oil, and should be of some use in the paint and varnish industry, where a high boiling point solvent with an oxidizing principle is desired. It will probably find application in the manufacture of Baking Japans, Asphalt Paints and Enamels. Its oxidizing and solvent values are very high. It has a distinctive sweet pine smell, which makes it popular in the manufacture of turpentine substitutes from petroleum spirits.
The writer has examined samples of this material, and the following appear to be of the best grade:
| No. 1 | No. 2 | |
| Color | Straw Color | Light Yellow |
| Specific Gravity at 15° C. | .934 | .936 |
| Boiling Point | 192° C. | 202° C. |
| Distillation | 95% distils between 192-270° C. |
95% distils between 202-280° C. |
| Residue on Evaporation | 14.34% | 14.60% |
| Polymerization Test | 32⁄3% unpolymerized at end of 1⁄2 hour |
21⁄2% unpolymerized at end of 1⁄2 hour |
| Flash-Point | 72° C. | 76° C. |
| Spot Test | Leaves no grease spot but only evaporates completely in 24 hours |
Same as Pine Oil No. 1 |
Turpentine. By direct fire or steam distillation of the sap drippings collected in pockets cut into pine trees, there is obtained the turpentine of commerce. It consists largely of pinene and isomeric terpenes, and has the property of attracting oxygen, with the formation of peroxides which stimulate the drying of oils. It is a high-grade solvent for various gums, and is therefore[19] used in the manufacture of many lacquers as well as for thinning down oil-gum varnishes.
| Color | Water White |
| Specific Gravity at 15° C. | .862-.875 |
| Boiling Point | About 156° C. |
| Distillation | 95% should distil between 153 and 165° C. |
| Residue on Evaporation | Not over 2% |
| Polymerization | Not over 5% should remain unpolymerized at end of half hour |
| Flash-Point | Over 40.5° C. |
| Spot Test | No grease spot should remain when dropped on paper and allowed to evaporate |
| Water | None |
Wood Turpentine. High-grade wood turpentine is now produced by the steam distillation of finely cut fat pine wood. The lower-grade qualities are often produced from the destructive distillation of sawdust, stumpage, etc., and these products, on account of their content of formaldehyde, are objectionable in odor. In the steam distillation process, however, a high quality product is obtained by cutting out the heavy fractions and redistilling the lower and purer fractions. It has a high oxidizing value, causing the rapid drying of paints and varnishes to which it has been added. Its solvent value is often greater than that of gum turpentine. When properly refined it has a sweet smell and is to be highly recommended.
Analyses of samples of pure wood turpentine which have come to the writer for examination follow:
| No. 1 | No. 2 | |
| Sp. Gr. at 15° C. | .862 | .862 |
| Boiling Point | 158° C. | 162° C. |
| Distillation: 95% distils between | 158 and 185° C. | 162 and 177° C. |
| Residue on Evaporation | 1.03% | 3.06% |
| Polymerization Test | 4.1% remains unpoly- merized at end of 1⁄2 hour |
0.1 cc. out of 6 cc. unpolymerized = 1.66% |
| Spot Test | No grease spot on evap- oration |
No grease spot on evap- oration |
| Odor | Excellent | Not objectionable |
| Color | Water White | Water White |
| Flash Point | 47.6° C. |
Petroleum Spirits. There are produced from Texas crude oil which has an asphaltum base, and Pennsylvania crude oil which has a paraffin base, high boiling-point petroleum spirits which have come into wide use as paint and varnish thinners. When such materials have the proper evaporating value, high flash-point and freedom from sulphur, they are to be highly recommended as paint thinners. The following shows the analyses of a few of these materials examined in the writer’s laboratory:
| Texas Base | California Base | Penna. Base | |
| Color | Water White | White | Water White |
| Specific Gravity | .811 | .79 | .81 |
| Boiling Point | 156° C. | 138° C. | 146° C. |
| Flash-Point | 44° C. | 40.5° C. | 43° C. |
| Residue on Evaporation | .2 | .15 | .12 |
Benzol. “Solvent naphtha” or 160-degree benzol is a product obtained from the distillation of coal tar, differing from benzine, a product obtained from the distillation of petroleum. It is a valuable thinner to use in the reduction of paints for the priming of resinous lumber and refractory woods such as cypress and yellow pitch pine. The penetrating and solvent values of benzol are high, and it often furnishes a unison between paint and wood, that is a prime foundation to subsequent coatings, preventing the usual scaling and sap exudations which often appear on a painted surface. Because of the great solvent action of benzol, it should never be used in second and third coatings. The writer has successfully painted inferior grades of cypress with a paint containing benzol in the priming coat.
Benzine. Benzine is seldom used in paints on account of its rapid evaporation, which is apt to cause pinholing of films and other surface defects. In paints of the dipping type where rapid evaporation is essential, benzine finds its widest application.
The proper drying of oils and their behavior with various siccatives in varying quantity is an interesting problem, and obviously of considerable importance from a practical standpoint. Unfortunately there is a decided scarcity of reliable literature dealing with the subject for the guidance of those concerned in the manufacture or application of siccative products. Furthermore, when the problem is investigated, it is not difficult to see why this is so.
Uniform Conditions. At a glance it is evident that a decided obstacle in experimentation on the drying properties of oils is the difficulty in obtaining identical conditions for comparative purposes. Inasmuch as a multitude of factors, such as uniformity and homogeneity of the driers and the oils themselves, intensity and source of light, temperature, uniformity of application, and many others, play a decisive part in the siccative tendencies of oils, the resources and ingenuity of the chemist engaged in the research are severely taxed.
Oxygen Absorption. It is a well-known fact that linseed oil, when applied to a clean surface, such as a glass plate, will undergo oxidation and take up oxygen to the extent of about 16%, forming a hard, elastic, non-sticky product which has been called linoxyn. This material, unlike the oil from which it has been formed, is insoluble in most solvents. Other oils, such as cottonseed, hemp, rape, olive, etc., are more fully satisfied in nature and have not the power to absorb the amount of oxygen taken up by linseed oil.
In carrying out the following tests, on the drying of oils, a quantity of pure linseed oil of the following analysis was secured:
| Specific gravity at 15° C. | 0.934 | |
| Acid number | 5 | |
| Saponification number | 191 | 1⁄2 |
| Iodine number | 188 |
This oil was distributed into a number of 8-oz. oil sample bottles, and to a series of these bottles was added varying quantities of a very concentrated drier made by boiling oil to 400 degrees Fahrenheit in an open kettle, with the subsequent addition of lead oxide. The amount of drier added to each bottle varied according to the percentage desired; being calculated on the lead content of the drier, which was very accurately determined by analysis.
There was secured in this manner a series of oils containing varying amounts of lead oxide, and from this lot was selected a certain number of samples which would be representative and typical of paint vehicles now found in the market.
Another series of tests were made by combining with a large number of samples of pure linseed oil as used above, various percentages of a manganese drier made by boiling oil at 400° F. and incorporating therewith manganese dioxide.
Still another series of tests were made upon a number of oils into which were incorporated various small quantities of lead oxide and manganese oxide together, using the standard driers made in the above manner, all of which were carefully analyzed to determine their contents.
In view of the errors in manipulation that could occur where so many tests were made, it was not deemed advisable, in carrying out the tests, to use glass plates on which only a minute quantity of oil could be maintained. A much better solution of the difficulty presented itself in using a series of small, round, crimped-edge tin plates, about three inches in diameter, such as are used for lids of friction-top cans.
With paints it is impossible to secure films as thin as those presented by layers of oil on glass, nor would it be desirable to secure films of this same relative thickness. For this reason an endeavor was made to conduct the following tests with films of the same relative thickness as that possessed by the average coating of paint. The drying of the films did not take place in the same short period, nor in the same ratio, as with the thin layer that is secured by flowing oil upon glass. The results, however, are more practical, and of greater value to the manufacturer.
The cans were carefully numbered in consecutive order, corresponding to the numbers on the various samples of oil.[23] A very small quantity of oil was placed in each of the can covers, which were previously weighed, and allowed to distribute itself over the bottom surface thereof. Reweighing of the covers gave the amount of oil which was taken for each test. The test samples in the covers were all placed in a large box with glass sides, having a series of perforated shelves. In the side of this box is an opening through which a tube was passed, carrying a continual current of air washed and dried in sulphuric acid. Oxidation of the oil films commenced at once, and the amount of oxygen absorbed was determined at suitable periods by weighing, the increase in weight giving this factor. This test was kept up for a period of twenty days.
A test was also made in the same manner with a current of damp air passing into the box, to observe the relative oxidation under such conditions. A chart of the results obtained has been made (Table VI), to show the effect of the various driers.
Results of Tests. The following outline will present to the mind of the reader the most salient points which have been gleaned from these experiments, and which should give the manufacturer definite knowledge as to the best percentage of oxides to use either in boiled oil, paints or varnishes.
In the case of lead oxide, an increase in the percentage of lead oxide in the oil causes a relative increase in the oxygen absorption, but when a very large percentage of lead has been added, the film of oil dries to a leathery skin.
In the case of manganese oxide, the increase in oxygen absorption on the first day is much more pronounced than is the case with lead oxides. Furthermore, the oxidation of manganese oils seems to be relative to the increase in manganese up to a certain period, when the reverse of this law seems to take place, and beyond a certain definite percentage of manganese, added percentages seem to be of no value. It was furthermore observed that the films dry to a more brittle and harder skin than is the case when lead oxide is used. The oxygen absorption with oils high in manganese has been noticed to be excessive, and the film of oil becomes surface-coated, drying beneath in a very slow manner; a condition that often leads to checking. The critical percentage where the amount of manganese appears to give the greatest efficiency seems to be 0.02%. This critical percentage, as it may be termed, should not be exceeded, and any added amount of manganese has the effect of making the film much more brittle and causes the so-called “burning up” of the paint. The loading of paint with drier and the bad result therefrom may be explained to some extent from the above results.
| Per cent. MnO2 | 0.02 | 0.05 | 0.15 | 0.25 | 0.35 | 0.45 | 0.55 | 0.70 | 1.00 | |||
| Per cent. gain | ⎧ | 1 | day | 0.08 | 0.11 | 0.16 | — | 3.21 | 3.46 | 3.27 | 3.01 | 2.76 |
| ⎪ | 2 | days | 0.16 | 5.88 | 4.48 | — | 3.63 | 4.01 | 3.70 | 3.51 | 3.18 | |
| ⎪ | 3 | days | 0.21 | 6.79 | 4.61 | — | 3.83 | 4.31 | — | 3.91 | — | |
| ⎪ | 4 | days | — | — | 4.64 | — | — | — | — | — | — | |
| ⎪ | 5 | days | 3.01 | 6.84 | — | — | 4.13 | 4.68 | 4.19 | 3.91 | 3.99 | |
| ⎪ | 6 | days | 8.00 | — | 4.88 | — | 4.37 | — | 4.51 | 4.32 | 4.13 | |
| ⎪ | 7 | days | 8.58 | 6.92 | 4.90 | — | 4.48 | — | 4.61 | 4.52 | 4.23 | |
| ⎨ | 8 | days | 9.06 | — | 5.03 | — | 4.55 | 5.23 | 4.77 | 4.62 | 4.44 | |
| ⎪ | 9 | days | — | — | 5.12 | — | 4.63 | 5.40 | 4.94 | 4.79 | 4.51 | |
| ⎪ | 10 | days | 9.07 | 6.89 | 5.18 | — | 4.81 | 5.47 | — | 4.98 | 4.73 | |
| ⎪ | 11 | days | 9.15 | 7.03 | — | — | — | — | — | — | — | |
| ⎪ | 12 | days | — | — | — | — | 4.98 | — | 5.45 | 5.33 | 5.22 | |
| ⎪ | 13 | days | 9.22 | 7.17 | — | — | 5.25 | 6.00 | 5.60 | 5.42 | 5.33 | |
| ⎪ | 14 | days | 9.25 | 7.18 | 5.55 | — | — | — | — | — | — | |
| ⎩ | 20 | days | — | 7.21 | 5.81 | — | 5.84 | 6.70 | 5.94 | 5.84 | 5.77 | |
| Per cent. MnO2 | 0.02 | 0.05 | 0.15 | 0.25 | 0.35 | 0.45 | 0.55 | 0.70 | 1.00 | |||
| ⎧ | 1 | day | — | 3.12 | 4.42 | 3.86 | — | 3.19 | 2.98 | 3.27 | 2.56 | |
| ⎪ | 2 | days | — | 6.15 | 4.73 | — | — | 3.51 | 3.28 | 3.70 | 2.96 | |
| ⎪ | 3 | days | 0.28 | 6.29 | — | 4.12 | 3.72 | — | 3.39 | 3.71 | 3.15 | |
| ⎪ | 4 | days | 3.83 | 6.32 | 4.75 | 4.21 | 3.87 | 3.61 | 3.58 | 4.05 | 3.43 | |
| ⎪ | 5 | days | 6.64 | — | 4.84 | 4.23 | 3.94 | 3.73 | 3.65 | 4.21 | 3.56 | |
| Per cent. gain | ⎨ | 6 | days | 8.61 | — | 4.87 | — | 4.08 | 3.81 | 3.78 | 4.35 | 3.73 |
| ⎪ | 7 | days | 9.07 | 6.35 | 5.00 | 4.41 | 4.18 | 3.91 | 3.85 | 4.54 | 3.87 | |
| ⎪ | 9 | days | 9.25 | 6.39 | 5.16 | — | 4.44 | 4.11 | 4.21 | 4.63 | 4.26 | |
| ⎪ | 11 | days | — | — | — | 4.63 | 4.59 | 4.36 | 4.31 | 5.07 | 4.46 | |
| ⎩ | 16 | days | — | 6.43 | 5.30 | 4.91 | 4.83 | 4.72 | 4.71 | 5.40 | 4.87 | |
| Per cent. PbO | 0.00 | 0.05 | 0.10 | 0.30 | 0.50 | 0.70 | 1.00 | 1.30 | 1.60 | 1.30 | 1.60 | ||||||||||||
| ⎧ | 1 | day | 0.042 | 0.04 | 9 | 0.09 | 2 | 0.05 | 8 | 0.06 | 6 | 0.06 | 2 | 0.06 | 2 | 0.07 | 9 | 0.03 | 9 | 0.14 | 0.72 | ||
| ⎪ | 2 | days | 0.098 | 0.10 | 4 | 0.15 | 3 | 0.11 | 6 | 0.15 | 8 | — | 0.19 | 4 | 4.83 | 4.79 | 5.27 | 6.11 | |||||
| ⎪ | 3 | days | 0.128 | 0.15 | 9 | 0.17 | 0 | 0.13 | 7 | 0.27 | 9 | 0.18 | 5 | 7.11 | 8.60 | 5.35 | 7.89 | 8.28 | |||||
| ⎪ | 4 | days | 0.164 | 0.21 | 4 | 0.20 | 6 | 0.17 | 8 | — | 4.07 | 7.39 | 9.55 | 8.53 | 7.93 | 8.68 | |||||||
| ⎪ | 5 | days | 0.176 | — | 0.30 | 6 | — | 0.34 | 0 | 7.60 | 7.47 | 9.87 | 8.78 | 8.18 | — | ||||||||
| ⎪ | 6 | days | 0.188 | 0.23 | 1 | — | 0.24 | 3 | 0.47 | 2 | 9.36 | 7.64 | 10.01 | 9.00 | 8.24 | 9.09 | |||||||
| Per cent. gain | ⎨ | 7 | days | 0.206 | 0.25 | 1 | — | 0.25 | 3 | 1.08 | 0 | 10.06 | — | 10.14 | — | — | — | ||||||
| ⎪ | 8 | days | 0.212 | 0.25 | 3 | — | 0.28 | 0 | 4.80 | 10.38 | 7.70 | 10.22 | 9.05 | — | — | ||||||||
| ⎪ | 9 | days | 0.226 | 0.29 | 1 | 0.30 | 6 | 0.33 | 1 | 7.36 | 10.41 | 7.73 | 10.23 | 9.07 | — | — | |||||||
| ⎪ | 13 | days | 0.327 | 0.42 | 8 | 0.51 | 0 | 0.67 | 4 | 11.01 | 10.67 | 7.91 | 10.48 | 9.29 | 8.62 | — | |||||||
| ⎪ | 15 | days | 0.466 | 0.45 | 5 | 0.65 | 0 | 2.41 | 11.05 | — | 7.92 | 10.50 | 9.30 | — | — | ||||||||
| ⎩ | 20 | days | 0.521 | 1.08 | 1.78 | 8.76 | 11.25 | 10.67 | 7.98 | 10.52 | 9.36 | — | — | ||||||||||
| Per cent. PbO | 0.1 | 0.3 | 0.5 | 0.7 | 0.9 | 1.1 | 1.4 | |||||||
| Per cent. MnO2 | .005 | .015 | 0.025 | 0.35 | 0.45 | 0.55 | 0.7 | |||||||
| Per cent. gain | ⎧ | 1 | day | 0.02 | 6 | 0.06 | 1 | 0.05 | 5 | 0.02 | 2 | 0.16 | 0.11 | 3.06 |
| ⎪ | 2 | days | 0.09 | 4 | 0.08 | 7 | 0.14 | 3 | 0.16 | 5.21 | 6.28 | 3.37 | ||
| ⎪ | 3 | days | 0.11 | 8 | — | 0.17 | 4.23 | 7.63 | 8.31 | 3.74 | ||||
| ⎪ | 4 | days | — | 0.11 | 0.23 | 7.36 | 8.87 | 9.20 | 4.02 | |||||
| ⎪ | 5 | days | 0.12 | 0 | 0.12 | 0.29 | 9.04 | 9.13 | 9.37 | 4.17 | ||||
| ⎨ | 6 | days | 0.17 | 0.13 | 1.44 | 9.88 | 9.26 | 9.51 | 4.34 | |||||
| ⎪ | 7 | days | 0.21 | 0.18 | 4.65 | 10.11 | 9.28 | — | 4.45 | |||||
| ⎪ | 11 | days | 0.30 | 0.26 | 10.03 | 10.35 | 9.61 | 9.85 | 5.11 | |||||
| ⎪ | 12 | days | — | — | — | 10.45 | 9.66 | — | — | |||||
| ⎪ | 13 | days | 0.35 | 0.54 | 10.37 | 10.51 | 9.67 | 10.03 | 5.33 | |||||
| ⎩ | 18 | days | 0.49 | 3.43 | 10.38 | 10.62 | 9.68 | — | 5.73 | |||||
[26]In the same way with lead driers, excessive amounts of lead oxide seem to have no beneficial effects on the drying of an oil, and when the percentage which seems to be the most beneficial, namely 0.5% lead oxide, is exceeded, the film is apt to become brittle.
Oils containing lead oxide driers are less influenced in their drying tendencies by conditions of moisture in the atmosphere than oils containing manganese, but frequently, however, the former dry much better in a dry atmosphere. As a general rule, varnishes rich in manganese dry more quickly in a dry atmosphere, while those containing small quantities dry more quickly in a damp atmosphere.
Volatile Products Formed. It was furthermore noticed in these tests that sulphuric acid, placed in dishes on the bottom of the large box in which the samples of oil were drying, was discolored and turned brown after several days, showing that the acid had taken up some material of a volatile nature that was a product of the oxidation.
Another curious feature of these tests was the development of a peculiar aromatic odor which was given off by the oils upon drying in dry air. When the oils were dried in moist air, a rank odor resembling propionic acid was observed, and this led the observer to believe that a reaction was effected by the absorbed oxygen, that caused the glycerin combined with the linoleic acid as linolein to split up into evil-smelling compounds. It has been suggested that the oxygen first attacks the glycerin, transforming it into carbonic acid, water, and other volatile compounds, which are eliminated before the oil is dried to linoxyn. Toch,[3] however, has shown that the drying of linseed oil gives off only very small percentages of carbon dioxide. Mulder has observed that in the process of linseed oil being oxidized, glycerin is set free, which becomes oxidized to formic,[27] acetic, and other acids, while the acid radicals are converted by oxygen into the anhydrides, from which they pass by further oxidation into linoxyn.
[3] Toch: The Chem. and Tech. of Mixed Paints, p. 89. D. Van Vostand Co., N. Y.
Auto-Oxidation of Oil. The theory of auto-oxidation of linseed oil has been very ably treated by Blackler, whose experiments indicated that during the drying process the slow absorption of oxygen was, at a critical period, followed by a rapid absorption, which he attributes to the presence of peroxides. The materials produced by this peroxide formation may act as catalyzers and accelerate the formation of more peroxide. Lead and manganese oxides may also be oxidized to peroxides by the action of oxygen, and in this event might act as very active catalyzing agents or carriers of oxygen. Blackler’s statement, that the presence of driers do not increase, but have a tendency to decrease the initial velocity of oxygen absorption, has been confirmed by these experiments, but it has been noticed throughout the tests that the driers have an accelerative action at a later period.
Effect of Metals on Drying of Oils. Some most interesting results were secured by dipping extremely fine copper gauze into linseed oil, and then suspending the gauze in the air. The adhesion of the oil to the copper caused the formation of films between the network, and remarkable drying action was observed. The copper or any superficial coating of copper oxide which may have been present on the metal, undoubtedly affected the result to some extent. It has been found that metallic lead is even more efficient than copper in this respect, but this may be due to the action of free acid in the linseed oil, forming lead linoleates, products that greatly accelerate drying. Another interesting experiment was made by immersing pieces of gauze cloth in linseed oil. After the excess oil had been removed, by pressing, the cloth was again weighed to determine the amount of oil used for the experiment. The increase in oxygen absorption in this case was very rapid, and the result obtained confirmed the results in the other experiments.
In order to secure a more evenly distributed state of the oil, tests were conducted by saturating pieces of stiff blotting papers, and, after exposure, weighing as usual.
Influence of Light. The influence of light on the drying of oils is unquestionably a potent one. The practical painter knows[28] that a certain varnish will dry quicker when exposed to the light than when in the dark.
Chevreul was one of the first pioneers in this field of research to observe the effects of colored lights on drying, and he claimed that oil exposed under white glass dried more rapidly than when exposed under red glass, which eliminates all light of short wave lengths.
Genthe obtained interesting results in the drying of oil submitted to the effect of the mercury lamp. Oxidation without driers was effected probably through the formation of peroxides. In commenting on this subject, Blackler[4] gives a description of the use of the Uveol Lamp, which is similar to the mercury lamp, but has, instead of a glass casing which cuts off the valuable rays, a fused-quartz casing which allows their passage.
[4] M. B. Blackler: “The Use and Abuse of Driers,” P. and V. Society, London, Sept. 9, 1909.
Driers in Boiled Oil. In the boiling of linseed oil, by certain processes the oil is heated to 250° F. and manganese resinate is incorporated therein. It goes into solution quite rapidly. In other processes the oil is heated to 400° F. or over, and manganese as an oxide is boiled into the oil. Although it is unsafe to say that a small percentage of rosin, such as would be introduced by the use of resinate driers, is not harmful, yet it appears that this process should give a good oil, inasmuch as it has been found that no matter whether the manganese is added to the oil, as a resinate, borate or oxide, practically the same drying effect is noticed in every case where the percentage of manganese is the same. It is the opinion of some, however, that the resinate driers are not as well suited for durability as oxide driers. However, if a boiled oil is found to contain on analysis a small percentage of rosin less than 0.5% or a percentage only sufficient to combine with the metal present, it should not be suspected of adulteration. Practical tests should be made with such oil along with an oil made with an oxide drier, before pronouncing on their relative values. Inasmuch as the addition of certain driers to linseed oil lessens the durability of the film, it is more practical to use the smallest amount of drier that will serve the purpose desired, that is, set the oil up to a hard condition which will not take dust and which will stand abrasion.
[29]The results of this investigation would indicate that when lead or manganese linoleates are used, the most efficient drying is shown with 0.5% lead or with 0.02% manganese, or with a combination of 0.5% lead and 0.02% manganese.
Until more definite results have been obtained with the tungates, which will probably prove of exceptional interest as driers, the above driers will probably be used to the greatest extent.
Co-operative Drying Tests. A series of important drying tests made by members of a special committee[5] appointed by the American Society for Testing Materials, of which the writer was chairman, is herewith shown:
[5] Sub-Committee C of Committee D-1, on Testing Paint Vehicles. Proc. Amer. Soc. for Test. Mater., 1911.
“At the January meeting of Committee D-1, a sub-committee consisting of the following members was appointed to investigate paint vehicles:
| G. B. Heckel, |
| Glenn H. Pickard, |
| Allen Rogers, |
| A. H. Sabin, |
| H. A. Gardner, Chairman. |
“At a subsequent meeting of the sub-committee it was determined to start the investigations with a series of tests on certain drying, semi-drying, and non-drying oils, determining their drying values, rate of oxygen absorption, etc., when spread out in thin films. A quantity of the following oils was selected for the tests and subsequently secured from sources known to be reliable:
| Lead and manganese linoleate drier.[6] | Cottonseed oil. |
| Lithographic linseed oil. | Sunflower oil. |
| Boiled linseed oil (resinate type). | Menhaden oil. |
| Boiled linseed oil (linoleate type). | Chinese wood oil, raw. |
| Blown linseed oil (containing drier while being blown). | Chinese wood oil, treated. |
| Heavy mineral oil. | Perilla oil.[7] |
| Rosin oil. | Lumbang oil.[7] |
| Soya bean oil. | Dry rosin 20%, boiled in 80% linseed oil. |
| Corn oil. |
[6] The drier used, upon analysis, showed the presence of 4.36% PbO and 2.51% MnO2.
[7] The lumbang and perilla oils were imported and arrived subsequent to the starting of the tests. They were therefore not included in the tests.
[30]“Four-ounce sample bottles of each oil were sent to the Committee members, with the request to proceed with the tests along the lines agreed upon at the Committee meeting. The instructions for making these tests are outlined as follows:
(a) A series of small glass plates, approximately 5 by 7 ins., are to be prepared by each member of the Committee. These plates are to be thoroughly cleaned and carefully numbered and weighed upon a chemical balance. The oils to be used for the tests are to be numbered corresponding to the plates. A test of each oil is to be made by painting it upon the surface of a glass plate with a camel’s-hair brush, subsequently weighing the plate and the oil. These tests are to be exposed under constant conditions of temperature, if possible, for three weeks’ time, making weighings of each plate every day for six days and then every other day for twelve days.
(b) Another series of tests shall be made, in which 80% of raw linseed oil is to be combined with each of the above oils named. Previous to making any of the tests, there should be added to each oil, or to each combination, 5% of a drier containing lead and manganese. The drier to be used is of the standard grade submitted, together with the oil samples. The results of the tests are to be charted and submitted at the end of the tests, so that they may be compared with the results obtained by each member of the Committee.
(c) If possible, the oils and mixture of oils used in the above tests are to be ground with pure silica and painted out upon sized paper, three-coat work, the films to be stripped and tested for strength upon a paint filmometer, at two periods two months apart.”
The drying of oils to a firm surface when spread in a thin layer is accompanied by an increase in weight, due to the absorption of oxygen. The percentage of oxygen absorbed often affords a criterion of the drying of the oil under examination, and this factor, together with data regarding the appearance of the oil film, should be taken into consideration when judging the value of an oil or oil mixture. Conditions of light, air, temperature, etc., often cause great variations in the drying of oils and the percentage of oxygen absorbed, as shown by the results obtained in the following tests. Although it was impossible in these tests to have the conditions under which each experimenter worked[31] parallel in nature, the tests afford nevertheless considerable information for guiding future work of a similar nature.
An examination of the results obtained showed generally that the greatest increase in weight occurred during the period in which the oil dried up to a firm film. This occurred in most cases within 48 hours. After this period a slight increase in weight was often noticed, and then a more or less steady decline, varying with the oil examined. Had the oil tests been continued for a greater length of time, a much greater loss might have been observed.
It was impossible to include in the tests the oil-silica film work, on account of lack of time. It is believed, however, that these tests should be conducted, as they would throw much light on the elasticity and strength given to paint films by various oils.
| Table I.—(a) Boiled Linseed Oil (Resinate Type) 100 Per Cent. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.1997 | 11.9 | 12.5 | 12.7 | 13.1 | 12.8 | 12.7 | — | 12.7 | — | 12.6 | — | 12.8 | — | 12.8 | — | 12.7 | — | 12.9 | — | { | Dried to firm, smooth film in 2 days. |
||||||||||||||||||||||||||
| Sabin | 0.6242 | 14.4 | 2 | 13.3 | 7 | 12.5 | 3 | 11.7 | 11.0 | 3 | — | 10.1 | 7 | 10.3 | 4 | 10.1 | 2 | 10.0 | 0 | — | 9.6 | 9 | — | — | 9.0 | 4 | — | 8.6 | 8 | — | 8.1 | 3 | ||||||||||||||||
| Pickard | 0.5027 | 10.2 | 1 | 10.0 | 0 | 9.5 | 7 | 9.6 | 5 | 8.9 | 9 | — | 8.5 | 7 | — | 8.9 | 3 | — | 8.8 | 1 | — | 9.3 | 1 | — | 9.4 | 3 | — | — | 9.1 | 1 | — | |||||||||||||||||
| Rogers North |
} | 0.6024 | 13.6 | 9 | 13.0 | 1 | 12.5 | 0 | 12.2 | 9 | 12.0 | 0 | 12.2 | 5 | — | 11.6 | 4 | — | 10.7 | 3 | — | 10.6 | 8 | — | 11.1 | 8 | — | 10.6 | 8 | — | — | — | { | Tacky at end of 1st day. Nearly dry, end of 2d day. Perfectly dry, end of 10th day. |
||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.1933 | 13.6 | 14.7 | 14.9 | 14.9 | 14.8 | 14.8 | — | 14.8 | — | 14.8 | — | 14.7 | — | 14.5 | — | 14.7 | — | 14.7 | — | { | Dried to firm, smooth film in 2 days. |
||||||||||||||||||||||||||
| Sabin | 0.3660 | 0.5 | 7 | 1.6 | 6 | 10.5 | 0 | 13.3 | 0 | — | — | 12.5 | 1 | — | 11.4 | 0 | — | — | 10.2 | 0 | — | — | 9.8 | 4 | — | — | — | — | ||||||||||||||||||||
| Pickard | 0.4640 | 12.4 | 8 | 11.9 | 2 | 11.4 | 9 | 11.1 | 0 | 10.8 | 4 | — | 9.4 | 8 | — | 7.4 | 1 | — | 7.5 | 6 | — | 8.3 | 6 | — | 8.5 | 4 | — | — | 8.5 | 1 | — | |||||||||||||||||
| Rogers North |
} | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Table II.—(a) Boiled Linseed Oil (Linoleate Type) 100 Per Cent. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.1226 | 10.9 | 12.2 | 12.7 | 12.5 | 12.8 | 12.2 | — | 12.2 | — | 12.4 | — | 12.1 | — | 12. | — | 12.1 | — | 12.1 | — | { | Dried firmly with smooth, even film in 2 days. |
||||||||||||||||||||||||||
| Sabin | 0.5384 | 14.3 | 4 | 13.2 | 6 | 12.1 | 8 | 11.2 | 9 | 10.7 | 5 | — | 9.8 | 8 | 10.2 | 5 | 10.0 | 1 | 9.9 | 1 | — | 9.6 | 0 | — | — | 9.1 | 2 | — | 8.3 | 7 | — | 8.3 | 0 | |||||||||||||||
| Pickard | 0.5696 | 10.2 | 5 | 10.4 | 1 | 10.2 | 2 | 10.1 | 6 | 9.9 | 0 | — | 9.6 | 0 | — | 9.7 | 2 | — | 9.4 | 8 | — | 9.9 | 7 | — | 10.3 | 6 | — | — | 9.5 | 9 | — | |||||||||||||||||
| Rogers North |
} | 0.3306 | 12.0 | 9 | 11.3 | 3 | 10.9 | 4 | 11.1 | 0 | 10.8 | 6 | 11.2 | 5 | — | 10.8 | 7 | — | 9.7 | 2 | — | 10.0 | 2 | — | 10.6 | 2 | — | 10.4 | 6 | — | — | — | { | Tacky at end of 1st day. Slightly tacky, end 2d day. Dry, but curled, end of 10th day. |
||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.1843 | 11.8 | 13.9 | 15.1 | 15.2 | 15.0 | 14.6 | — | 14.6 | — | 14.5 | — | 14.4 | — | 14.4 | — | 14.6 | — | 14.7 | — | { | Dried with smooth film in 2 days. |
||||||||||||||||||||||||||
| Sabin | 0.5790 | 10.1 | 4 | 15.7 | 1 | 13.2 | 9 | 12.1 | 2 | 11.4 | 3 | — | 10.0 | 5 | 10.2 | 6 | 9.5 | 5 | 9.3 | 2 | — | 8.8 | 4 | — | — | 8.4 | 6 | — | 7.6 | 8 | — | 7.5 | 5 | |||||||||||||||
| Pickard | 0.4653 | 12.4 | 0 | 11.9 | 0 | 11.5 | 0 | 11.1 | 1 | 10.9 | 0 | — | 9.3 | 7 | — | 8.5 | 3 | — | 7.4 | 8 | — | 8.4 | 3 | — | 8.0 | 2 | — | — | 7.2 | 7 | — | |||||||||||||||||
| Rogers North |
} | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Table III.—(a) Lithographic Linseed Oil 100 Per Cent.[33] | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.4011 | 6.9 | 8.5 | 8.9 | 8.9 | 8.7 | 8.6 | — | 8.6 | — | 8.6 | — | 8.6 | — | 8.4 | — | 8.4 | — | 8.3 | — | { | Dried to glossy, firm film, slightly crinkled in 2 days. Oil made very thick film on account of heavy body. |
||||||||||||||||||||||||||
| Sabin | 0.8733 | 0.8 | 7 | 3.8 | 5 | 5.1 | 4 | 6.0 | 7 | 6.4 | 0 | — | 6.8 | 4 | 7.2 | 2 | 7.3 | 6 | 7.5 | 7 | — | 7.7 | 5 | — | — | 7.9 | 8 | — | 7.8 | 3 | — | 7.8 | 0 | |||||||||||||||
| Pickard | 0.8812 | 3.6 | 0 | 5.1 | 9 | 5.9 | 9 | 6.7 | 8 | 6.9 | 7 | — | 7.3 | 8 | — | 7.4 | 2 | — | 7.4 | 4 | — | 8.0 | 1 | — | 8.0 | 3 | — | — | 7.9 | 9 | — | |||||||||||||||||
| Rogers North |
} | 2.7318 | .0 | 51 | .0 | 51 | .0 | 51 | .0 | 41 | .0 | 81 | .1 | 69 | — | .1 | 9 | — | .7 | 52 | — | 1.1 | 84 | — | 1.6 | 41 | — | 2.0 | 0 | — | — | — | { | Remained sticky to 10 days, and even at end of 38 days was slightly tacky. |
||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.1300 | 10.2 | 11.3 | 11.9 | 12.0 | 11.8 | 11.8 | — | 11.8 | — | 11.8 | — | 11.6 | — | 11.8 | — | 11.9 | — | 11.9 | — | { | Dried to firm, glossy film in 2 days. |
||||||||||||||||||||||||||
| Sabin | 0.7750 | 11.3 | 5 | 11.4 | 8 | 10.9 | 3 | 10.7 | 7 | 10.2 | 5 | — | 9.5 | 1 | 9.9 | 3 | 9.8 | 0 | 9.6 | 8 | — | 9.6 | 5 | — | — | 9.5 | 1 | — | 9.0 | 7 | — | 8.6 | 7 | |||||||||||||||
| Pickard | 0.6538 | 9.9 | 4 | 10.4 | 1 | 10.3 | 9 | 10.3 | 5 | 9.9 | 3 | — | 9.5 | 4 | — | 9.3 | 6 | — | 8.9 | 9 | — | 9.6 | 1 | — | 9.7 | 0 | — | — | 9.1 | 3 | — | |||||||||||||||||
| Table IV.—(a) Blown Linseed Oil 100 Per Cent. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.2105 | 8.5 | 10.2 | 10.2 | 10.2 | 10.0 | 9.9 | — | 9.8 | — | 9.8 | — | 9.7 | — | 9.8 | — | 9.8 | — | 9.9 | — | { | Ropiness of oil made very thick film, but dried in less than 2 days to smooth film. Films exhibited ridges. |
||||||||||||||||||||||||||
| Sabin | 0.8394 | 9.3 | 0 | 8.9 | 7 | 5.3 | 0 | 9.3 | 0 | 8.9 | 9 | — | 8.4 | 9 | 8.8 | 9 | 8.7 | 3 | 8.8 | 9 | — | 8.7 | 3 | — | — | 8.5 | 2 | — | 8.0 | 7 | — | 7.7 | 4 | |||||||||||||||
| Pickard | 0.8457 | 5.0 | 7 | 6.1 | 6 | 6.4 | 8 | 6.9 | 4 | 6.7 | 3 | — | 6.9 | 9 | — | 6.8 | 9 | — | 7.1 | 1 | — | 7.6 | 0 | — | 7.9 | 5 | — | — | 7.8 | 6 | — | |||||||||||||||||
| Rogers North |
} | 1.0398 | 4.4 | 1 | 4.9 | 1 | 5.2 | 2 | 5.6 | 2 | 5.7 | 3 | 6.0 | 6 | — | 6.4 | 3 | — | 6.1 | 8 | — | 6.5 | 1 | — | 6.9 | 5 | — | 7.0 | 0 | — | — | — | { | Formed skin, end 1st day. Slightly tacky, end 2d; dry, but curled, end of 10th day. |
||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.0774 | 10.4 | 12.8 | 13.1 | 12.9 | 12.1 | 11.9 | — | 12.0 | — | 11.8 | — | 11.7 | — | 11.6 | — | 11.6 | — | 11.8 | — | { | Dried up to very glossy film in 2 days. |
||||||||||||||||||||||||||
| Sabin | 0.5329 | 11.8 | 2 | 12.7 | 6 | 10.9 | 8 | 10.3 | 9 | 9.8 | 1 | — | 8.6 | 9 | 9.1 | 5 | 8.9 | 1 | 8.9 | 7 | — | 8.6 | 7 | — | — | 8.2 | 2 | — | 7.6 | 3 | — | 7.3 | 2 | |||||||||||||||
| Pickard | 0.6218 | 10.7 | 1 | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | { | Glass broke. | |||||||||||||||||||||||||
| Table V.—(a) Mineral Oil 100 Per Cent.[34] | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.1632 | [8]12.5 | [8]14.2 | [8]16.7 | [8]19.4 | [8]19.4 | [8]19.5 | — | [8]19.5 | — | [8]19.5 | — | [8]19.3 | — | [8]19.4 | — | [8]19.5 | — | [8]19.5 | — | { | Oil lost in weight throughout test on account of presence of volatiles. No drying action observed. Film wet at end of test. |
||||||||||||||||||||||||||
| Sabin | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | { | Broken before weighings were made. |
||||||||||||||||||||||||||
| Pickard | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||
| Rogers North |
} | 0.1975 | [8]8.1 | 2 | [8]16.2 | 2 | [8]21.2 | 3 | [8]25.5 | 8 | [8]28.4 | 1 | [8]28.9 | 2 | — | [8]35.2 | 5 | — | [8]35.7 | 6 | — | [8]43.8 | 6 | — | [8]45.2 | 8 | — | [8]48.0 | 8 | — | — | — | { | Remained oily during entire test. |
||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.1884 | 6.4 | 6.8 | 7.2 | 7.8 | 8.1 | 7.9 | — | 7.9 | — | 8.1 | — | 7.8 | — | 7.8 | — | 7.8 | — | 7.8 | — | { | Fair drying observed end of 2d day. Film tacky until end 8th day; after that, fairly firm film shown. |
||||||||||||||||||||||||||
| Sabin | 0.5663 | 11.5 | 1 | 8.2 | 1 | 6.5 | 1 | 5.1 | 9 | 4.3 | 6 | — | 2.7 | 2 | 3.1 | 2 | 2.8 | 2 | 2.5 | 9 | — | 2.3 | 5 | — | — | 1.3 | 6 | — | 0.5 | 3 | — | [8]0.1 | 4 | |||||||||||||||
| Pickard | 0.405 | [9]9.6 | 6 | [9]8.9 | 2 | [9]6.8 | 2 | [9]6.0 | 3 | [9]4.6 | 8 | — | [9]2.6 | 4 | — | [8]0.3 | 0 | — | [8]0.5 | 6 | — | [8]0.0 | 4 | — | [8]0.1 | 4 | — | — | [8]0.8 | 6 | — | |||||||||||||||||
| Rogers North |
} | 0.2598 | [9]6.6 | 9 | [9]5.0 | 6 | [9]2.8 | 8 | [9]1.5 | 2 | [9]1.2 | 9 | [9]1.6 | 8 | — | [9]2.0 | 7 | — | [8]0.0 | 8 | — | [8]0.9 | 3 | — | [8]0.5 | 4 | — | — | — | — | — | { | Sticky, end of 1st day; tacky, end of 2d day and end of 38 days. |
|||||||||||||||
| Table VI.—(a) Soya Bean Oil 100 Per Cent.[35] | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.1377 | 7.5 | 8.4 | 9.5 | 12.8 | 12.9 | 12.7 | — | 12.6 | — | 12.5 | — | 12.4 | — | 12.3 | — | 12.3 | — | 12.3 | — | { | Film tacky until 3d day. Clear and fairly firm after 4th day. |
||||||||||||||||||||||||||
| Sabin | 0.3972 | 9.7 | 9 | 9.6 | 9 | 8.5 | 6 | 7.6 | 0 | 7.0 | 9 | — | 6.0 | 0 | 6.2 | 2 | 6.0 | 0 | 5.5 | 4 | — | 5.3 | 6 | — | — | 4.7 | 3 | — | 4.2 | 3 | — | 3.7 | 0 | |||||||||||||||
| Pickard | 0.4366 | 9.8 | 7 | 9.8 | 7 | 9.3 | 5 | 8.6 | 6 | 8.1 | 3 | — | 6.4 | 4 | — | 4.8 | 8 | — | 4.2 | 6 | — | 4.9 | 9 | — | 4.9 | 4 | — | — | 4.9 | 4 | — | |||||||||||||||||
| Rogers North |
} | 0.3564 | 8.2 | 5 | 7.5 | 8 | 7.0 | 2 | 6.7 | 4 | 6.4 | 6 | 6.7 | 4 | — | 6.4 | 6 | — | 5.4 | 0 | — | 5.5 | 9 | — | 5.8 | 0 | — | 5.6 | 7 | — | — | — | { | Sticky, end of 1st day; tacky, end of 2d day; slightly tacky, end of 10th and 38th days. |
||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.2218 | 11.5 | 11.8 | 12.5 | 13.9 | 14.0 | 14.0 | — | 14.1 | — | 14.1 | — | 13.8 | — | 13.6 | — | 13.6 | — | 13.6 | — | { | Clear, firm film observed at end of 2d day. |
||||||||||||||||||||||||||
| Sabin | 0.2877 | 12.7 | 8 | 12.7 | 8 | 11.7 | 4 | 12.2 | 3 | 10.6 | 0 | — | 9.3 | 5 | 10.0 | 8 | 9.7 | 6 | 9.5 | 9 | — | 9.5 | 9 | — | — | 9.0 | 0 | — | 8.0 | 9 | — | 8.0 | 0 | |||||||||||||||
| Pickard | 0.4581 | 13.1 | 6 | 12.6 | 4 | 11.8 | 4 | 11.5 | 0 | 11.0 | 1 | — | 9.1 | 5 | — | 7.2 | 9 | — | 6.6 | 1 | — | 7.4 | 3 | — | 6.9 | 6 | — | — | 6.6 | 6 | — | |||||||||||||||||
| Rogers North |
} | 0.2249 | 11.7 | 4 | 12.2 | 7 | 10.3 | 8 | 9.4 | 3 | 9.6 | 6 | 9.7 | 5 | — | 10.2 | 9 | — | 9.0 | 8 | — | 8.1 | 8 | — | 8.9 | 5 | — | — | — | — | — | { | Tacky at end of 1st and 2d days. Dry, end 10th day. |
|||||||||||||||
| Table VII.—(a) Rosin Oil 100 Per Cent. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.2590 | 1.5 | 1.5 | 1.8 | 3.0 | 5.2 | 4.9 | — | 4.8 | — | 4.8 | — | 4.8 | — | 4.8 | — | 4.8 | — | 4.8 | — | { | Tacky throughout test. | ||||||||||||||||||||||||||
| Sabin | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||
| Pickard | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | { | Too much on. Showed constantly increasing loss owing to the fact that it did not dry and ran off glass. |
||||||||||||||||||||||||||
| Rogers North |
} | 0.4822 | 2.2 | 4 | 2.5 | 3 | 2.3 | 2 | 1.2 | 7 | 1.0 | 6 | 0.6 | 6 | — | 0.2 | 4 | — | 0.7 | 8 | — | 0.6 | 8 | — | 0.4 | 1 | — | 0.3 | 9 | — | — | — | { | Oily on 1st and 2d days. Tacky, end of 10 and 38 days. |
||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.1636 | 7.4 | 7.8 | 8.5 | 8.5 | 8.4 | 8.1 | — | 8.0 | — | 8.0 | — | 8.0 | — | 7.9 | — | 7.9 | — | 8.2 | — | { | Film dried up nicely during 3d day, but remained slightly soft. |
||||||||||||||||||||||||||
| Sabin | 0.7105 | 6.6 | 4 | 6.4 | 0 | 6.0 | 5 | 5.6 | 3 | 5.2 | 3 | — | 4.4 | 2 | 4.9 | 2 | 4.8 | 3 | 4.5 | 7 | — | 4.6 | 8 | — | — | 4.1 | 3 | — | 3.8 | 1 | — | 3.4 | 3 | |||||||||||||||
| Pickard | 0.4016 | 12.2 | 1 | 11.4 | 5 | 11.1 | 3 | 10.5 | 3 | 10.1 | 3 | — | 8.8 | — | 8.1 | 2 | — | 7.4 | 5 | — | 8.2 | 7 | — | 8.5 | 2 | — | — | 8.6 | 2 | — | ||||||||||||||||||
| Rogers North |
} | 0.3263 | 11.4 | 8 | 12.0 | 2 | 10.6 | 0 | 10.2 | 6 | 10.4 | 2 | 10.4 | 2 | — | 10.9 | 5 | — | 9.9 | 6 | — | 9.5 | 3 | — | 9.9 | 6 | — | — | — | — | — | { | Oily at end of 1st and 2d days. Slightly tacky, end of 10th day. |
|||||||||||||||
| Table VIII.—(a) Corn Oil 100 Per Cent. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.0574 | 1.9 | 4.2 | 4.6 | 4.8 | 7.5 | 7.1 | — | 7.1 | — | 7.1 | — | 7.2 | — | 7.1 | — | 7.0 | — | 6.9 | — | { | Film soft and sticky throughout test. Very soapy in appearance. |
||||||||||||||||||||||||||
| Sabin | 0.5858 | [8]0.2 | 2 | 7.0 | 3 | 8.7 | 9 | 7.4 | 3 | 7.1 | 7 | — | 5.8 | 5 | 6.0 | 2 | 5.8 | 4 | 5.5 | 8 | — | 5.3 | 8 | — | — | 4.7 | 8 | — | 4.1 | 5 | — | 3.6 | 3 | |||||||||||||||
| Pickard | 0.4981 | 1.2 | 2 | 5.8 | 6 | 7.2 | 7 | [10]11.3 | 5 | 11.3 | 5 | — | 11.3 | 7 | — | 6.2 | 6 | — | 4.9 | 7 | — | 5.6 | 2 | — | 5.3 | 4 | — | — | 5.3 | 4 | — | |||||||||||||||||
| Rogers North |
} | 0.3300 | 4.6 | 3 | 7.2 | 7 | 7.1 | 4 | 6.9 | 9 | 6.6 | 9 | 6.9 | 3 | — | 6.8 | 4 | — | 5.1 | 1 | — | 5.1 | 7 | — | 5.3 | 8 | — | 5.1 | 7 | — | — | — | ||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.1664 | 7.5 | 8.4 | 8.6 | 10.2 | 10.4 | 10.6 | — | 10.5 | — | 10.3 | — | 10.3 | — | 10.3 | — | 10.2 | — | 10.0 | — | { | Film tacky at end of test. | ||||||||||||||||||||||||||
| Sabin | 0.5469 | 13.0 | 1 | 12.4 | 1 | — | 11.1 | 3 | 11.5 | 2 | — | 11.2 | 2 | 10.9 | 8 | 10.3 | 8 | 9.6 | 4 | — | 9.0 | 7 | — | — | 8.3 | 8 | — | 8.7 | 7 | — | — | |||||||||||||||||
| Pickard | 0.3716 | 13.8 | 1 | 12.9 | 2 | 12.1 | 6 | 11.7 | 1 | 11.1 | 1 | — | 9.2 | 3 | — | 8.2 | 9 | — | 7.2 | 4 | — | 8.4 | 2 | — | 8.2 | 6 | — | — | 7.9 | 4 | — | |||||||||||||||||
| Rogers North |
} | 0.1711 | 11.8 | 7 | 11.6 | 9 | 9.7 | 8 | 8.3 | 3 | 8.5 | 0 | 8.6 | 2 | — | 9.6 | 1 | — | 8.1 | 6 | — | 7.0 | 0 | — | 8.2 | 8 | — | — | — | — | — | { | Tacky, end of 1st and 2d days. Dry, end 10th day. |
|||||||||||||||
| [37]Table IX.—(a) Cotton Seed Oil 100 Per Cent. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.2026 | 4.5 | 4.8 | 4.8 | 5.1 | 8.6 | 8.7 | — | 8.1 | — | 7.9 | — | 8.0 | — | 8.0 | — | 8.1 | — | 8.0 | — | { | Film showed very little hardening and remained soft and tacky. |
||||||||||||||||||||||||||
| Sabin | 0.7247 | 8.0 | 3 | 7.4 | 8 | 6.6 | 8 | 6.0 | 0 | 5.6 | 5 | — | 4.8 | 5 | 5.0 | 9 | 4.9 | 5 | 4.8 | 0 | — | — | — | — | — | — | — | — | — | |||||||||||||||||||
| Pickard | 0.4135 | 7.0 | 4 | 7.1 | 6 | 6.6 | 2 | 6.2 | 4 | 5.7 | 8 | — | 3.7 | 2 | — | 2.0 | 8 | — | 1.7 | 2 | — | 2.5 | 2 | — | 2.3 | 5 | — | — | 2.3 | 2 | — | |||||||||||||||||
| Rogers North |
} | 0.3583 | 6.6 | 7 | 5.6 | 1 | 4.8 | 5 | 4.6 | 5 | 4.3 | 7 | 4.7 | 1 | — | 4.5 | 7 | — | 2.9 | 7 | — | 3.1 | 1 | — | 3.3 | 9 | — | 3.3 | 9 | — | — | — | { | Tacky, end 2d day. Slightly tacky, end 10th and 38th days. |
||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.1516 | 8.5 | 8.7 | 9.1 | 10.8 | 11.9 | 11.8 | — | 11.9 | — | 11.9 | — | 11.8 | — | 11.8 | — | 11.8 | — | 10.7 | — | { | Fair drying observed at end of 4th day. Film slightly tacky at end of test. |
||||||||||||||||||||||||||
| Sabin | 0.9498 | 11.0 | 0 | 11.1 | 5 | 10.5 | 8 | 10.1 | 7 | 9.8 | 2 | — | 9.0 | 2 | 9.4 | 2 | 9.3 | 5 | 9.2 | 7 | — | 9.3 | 2 | — | — | 8.8 | 1 | — | 8.2 | 4 | — | 7.9 | 2 | |||||||||||||||
| Pickard | 0.6160 | 10.9 | 4 | 10.8 | 1 | 10.5 | 1 | 10.3 | 7 | 9.8 | 7 | — | 8.9 | 3 | — | 8.9 | 0 | — | 8.7 | 0 | — | 9.2 | 9 | — | 9.6 | 3 | — | — | 8.4 | 7 | — | |||||||||||||||||
| Rogers North |
} | 0.2553 | 11.8 | 3 | 11.8 | 3 | 10.1 | 5 | 9.2 | 9 | 9.2 | 9 | 9.4 | 5 | — | 10.0 | 0 | — | 8.9 | 5 | — | 8.0 | 6 | — | 8.6 | 1 | — | — | — | — | — | { | Tacky on 1st and 2d days. Dry on 10th day. |
|||||||||||||||
| Table X.—(a) Sun Flower Oil 100 Per Cent. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.1414 | 6.3 | 8.2 | 11.5 | 11.6 | 11.5 | 11.5 | — | 11.3 | — | 11.3 | — | 11.3 | — | 11.3 | — | 11.2 | — | 11.0 | — | { | Film fairly firm, end of 3d day. |
||||||||||||||||||||||||||
| Sabin | 0.6292 | 9.6 | 9 | 9.4 | 2 | 7.9 | 9 | 7.4 | 3 | 7.0 | 4 | — | 6.1 | 2 | 6.4 | 5 | 6.1 | 2 | 5.9 | 2 | — | 5.6 | 9 | — | — | 5.2 | 4 | — | 4.5 | 7 | — | 4.2 | 6 | |||||||||||||||
| Pickard | 0.5837 | 7.8 | 5 | 7.7 | 3 | 7.4 | 5 | 7.0 | 2 | 6.3 | 6 | — | 5.1 | 6 | — | 4.5 | 7 | — | 4.2 | 0 | — | 4.5 | 4 | — | 4.6 | 1 | — | — | 4.3 | 0 | — | |||||||||||||||||
| Rogers North |
} | 0.2540 | 8.3 | 9 | 6.9 | 4 | 6.2 | 1 | 6.1 | 3 | 5.8 | 1 | 6.0 | 1 | — | 6.0 | 9 | — | 4.8 | 1 | — | 4.7 | 3 | — | 4.8 | 1 | — | 5.0 | 1 | — | — | — | { | Sticky, end 1st day; tacky, end 2d day; slightly tacky, end 10th day. |
||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.1600 | 9.5 | 11.0 | 11.1 | 11.3 | 11.4 | 10.9 | — | 10.8 | — | 10.8 | — | 10.8 | — | 10.6 | — | 10.6 | — | 10.9 | — | { | Good firm, glossy film shown at end of 2d day. |
||||||||||||||||||||||||||
| Sabin | 0.5030 | 14.2 | 1 | 14.2 | 1 | 12.6 | 6 | 14.0 | 1 | 11.5 | 9 | — | 10.2 | 4 | 10.6 | 3 | 10.3 | 4 | 10.3 | 4 | — | 10.2 | 7 | — | — | 11.3 | 3 | — | 10.7 | 3 | — | 10.3 | 0 | |||||||||||||||
| Pickard | 0.4470 | 12.6 | 2 | 12.0 | 2 | 11.4 | 8 | 11.6 | 5 | 10.2 | 5 | — | 8.1 | 4 | — | 6.2 | 6 | — | 5.5 | 4 | — | 6.2 | 2 | — | 5.8 | 2 | — | — | 5.3 | 5 | — | |||||||||||||||||
| Rogers North |
} | 0.2261 | 11.5 | 4 | 11.8 | 5 | 9.9 | 2 | 9.1 | 3 | 8.9 | 5 | 9.0 | 4 | — | 9.5 | 2 | — | 8.5 | 5 | — | 7.6 | 7 | — | 8.2 | 0 | — | — | — | — | — | { | Dry on 1st, 2d and 10th days. | |||||||||||||||
| Table XI.—(a) Menhaden Oil 100 Per Cent. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.1944 | 7.7 | 8.1 | 8.9 | 10.1 | 9.8 | 9.8 | — | 9.8 | — | 9.8 | — | 9.8 | — | 9.6 | — | 9.6 | — | 9.6 | — | { | Good drying during 2d day. Fairly firm film. |
||||||||||||||||||||||||||
| Sabin | 0.5282 | 12.4 | 7 | 12.1 | 7 | 11.7 | 0 | 11.4 | 7 | 11.1 | 3 | — | 10.2 | 8 | 11.2 | 0 | 11.1 | 5 | 11.0 | 2 | — | 11.3 | 7 | — | — | 10.8 | 5 | — | 10.3 | 4 | — | 9.9 | 0 | |||||||||||||||
| Pickard | 0.7005 | 10.7 | 9 | 10.9 | 8 | 10.8 | 5 | 10.9 | 0 | 10.5 | 7 | — | 9.2 | 7 | — | 8.4 | 8 | — | 8.2 | 7 | — | 8.9 | 1 | — | 8.7 | 5 | — | — | 9.2 | 1 | — | |||||||||||||||||
| Rogers North |
} | 0.3150 | 11.2 | 7 | 10.1 | 6 | 9.7 | 2 | 9.9 | 7 | 9.9 | 4 | 10.2 | 7 | — | 10.3 | 6 | — | 8.8 | 0 | — | 9.2 | 2 | — | 9.4 | 0 | — | 9.3 | 1 | — | — | — | { | Sticky, end 1st day. Slightly sticky, end 2d and 10th days. |
||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.2448 | 8.5 | 10.4 | 12.2 | 12.9 | 12.9 | 12.9 | — | 12.9 | — | 12.9 | — | 12.9 | — | 12.8 | — | 12.7 | — | 12.9 | — | { | Good firm, elastic film shown after 2d day. |
||||||||||||||||||||||||||
| Sabin | 0.4959 | 14.1 | 1 | 13.4 | 7 | 12.6 | 8 | 12.0 | 4 | 11.5 | 9 | — | 10.4 | 4 | 11.0 | 9 | 11.0 | 4 | 10.7 | 4 | — | 10.9 | 0 | — | — | 10.1 | 8 | — | 9.4 | 8 | — | 8.9 | 3 | |||||||||||||||
| Pickard | 0.4201 | 13.1 | 9 | 12.8 | 8 | 12.2 | 3 | 11.8 | 1 | 11.1 | 7 | — | 9.5 | 0 | — | 8.4 | 8 | — | 7.7 | 7 | — | 8.3 | 3 | — | 8.2 | 4 | — | — | 8.1 | 2 | — | |||||||||||||||||
| Rogers North |
} | 0.2456 | 10.9 | 9 | 11.2 | 8 | 9.5 | 6 | 8.9 | 0 | 8.7 | 2 | 8.7 | 2 | — | 9.3 | 4 | — | 8.4 | 0 | — | 7.3 | 7 | — | 8.1 | 1 | — | — | — | — | — | { | Nearly dry on 1st and 2d days. | |||||||||||||||
| Table XII.—(a) Raw Chinese Wood Oil 100 Per Cent.[39] | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.2266 | 4.1 | 11.2 | 14.9 | 14.4 | 14.4 | 14.2 | — | 14.2 | — | 14.2 | — | 14.2 | — | 14.2 | — | 14.2 | — | 14.5 | — | { | Film crystallized and remained soft until 3d day. Hard but opaque film shown after 4th day. |
||||||||||||||||||||||||||
| Sabin | 0.5545 | — | — | 11.0 | 2 | 11.5 | 3 | 11.0 | 3 | — | 10.5 | 3 | 10.7 | 4 | 10.4 | 7 | 10.2 | 7 | — | 10.2 | 2 | — | — | 9.8 | 0 | — | 9.2 | 5 | — | 8.8 | 6 | |||||||||||||||||
| Pickard | 0.4933 | 0.5 | 9 | 2.0 | 9 | 5.1 | 3 | 7.5 | 6 | 8.6 | 8 | — | 10.1 | 1 | — | 9.6 | 5 | — | 9.4 | 3 | — | 9.7 | 7 | — | 9.7 | 3 | — | — | 9.3 | 3 | — | |||||||||||||||||
| Rogers North |
} | 0.4036 | 0.5 | 4 | 2.8 | 0 | 5.1 | 0 | 6.0 | 0 | 6.2 | 7 | 7.0 | 9 | — | 8.3 | 9 | — | 8.0 | 1 | — | 8.5 | 5 | — | 9.1 | 3 | — | 9.2 | 7 | — | — | — | { | Sticky, end of 1st and 2d days; dry but drawn, end of 10th day. |
||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.2087 | 9.0 | 12.1 | 12.9 | 12.8 | 12.8 | 12.8 | — | 12.7 | — | 12.6 | — | 12.6 | — | 12.5 | — | 12.5 | — | 12.7 | — | { | Clear and firm film shown after 3d day. |
||||||||||||||||||||||||||
| Sabin | 0.2967 | 14.4 | 6 | 13.1 | 1 | 11.7 | 2 | 10.6 | 8 | 9.7 | 7 | — | 8.6 | 6 | 8.8 | 6 | 8.8 | 0 | 8.4 | 9 | — | 8.1 | 5 | — | — | 8.0 | 5 | — | 7.4 | 1 | — | 7.0 | 4 | |||||||||||||||
| Pickard | 0.3683 | 14.3 | 7 | 13.6 | 6 | 13.1 | 1 | 12.4 | 1 | 11.7 | 8 | — | 10.5 | 1 | — | 8.7 | 2 | — | 7.0 | — | 8.8 | 2 | — | 8.3 | 9 | — | — | 7.9 | 8 | — | ||||||||||||||||||
| Rogers North |
} | 0.2285 | 11.9 | 9 | 11.9 | 0 | 10.1 | 4 | 9.3 | 0 | 9.0 | 8 | 9.3 | 0 | — | 9.7 | 0 | — | 8.9 | 0 | — | 7.3 | 4 | — | 7.7 | 8 | — | — | — | — | — | Dry at end of 1st day. | ||||||||||||||||
| Table XIII.—(a) Chinese Wood Oil (Treated) 100 Per Cent. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.1678 | [8]38.0 | [8]30.0 | [8]28.0 | [8]28.0 | [8]28.0 | [8]28.0 | — | [8]28.0 | — | 27.5 | — | [8]26.0 | — | [8]26.0 | — | [8]26.0 | — | [8]26.2 | — | { | Loss observed due to presence of volatiles. Firm, clear film shown at end of 1st day. |
||||||||||||||||||||||||||
| Sabin | 0.4159 | [8]19.0 | 6 | [8]20.1 | 6 | [8]20.4 | 7 | [8]20.4 | 7 | [8]20.8 | 0 | — | [8]21.0 | 9 | [8]20.8 | 7 | [8]20.9 | 8 | [8]20.7 | 8 | — | [8]20.7 | 0 | — | — | [8]20.9 | 7 | — | [8]21.2 | 2 | — | [8]21.1 | 1 | |||||||||||||||
| Pickard | 0.2934 | [8]0.9 | 2 | [8]0.4 | 1 | 0.7 | 2 | 0.7 | 9 | 0.1 | 3 | — | 0.2 | 2 | — | 0.4 | 6 | — | 0.4 | 4 | — | 0.4 | 3 | — | 0.4 | 2 | — | — | 0.4 | 3 | — | |||||||||||||||||
| Rogers North |
} | 0.3937 | 3.5 | 3 | 3.5 | 8 | 3.2 | 5 | 3.2 | 5 | 3.3 | 3 | 2.9 | 3 | — | 2.5 | 5 | — | 3.4 | 0 | — | 3.2 | 3 | — | 2.6 | 1 | — | 2.4 | 8 | — | — | — | Dry at end of 1st day. | |||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.1638 | 8.4 | 9.4 | 9.8 | 9.7 | 9.9 | 9.9 | — | 10.0 | — | 9.6 | — | 9.5 | — | 9.5 | — | 9.5 | — | 9.6 | — | { | Clear and hard film shown during 2d day. |
||||||||||||||||||||||||||
| Sabin | 0.6572 | 9.2 | 5 | 8.0 | 7 | 7.3 | 6 | 6.7 | 5 | 6.2 | 5 | — | 5.4 | 9 | 5.8 | 7 | 5.7 | 0 | 5.6 | 7 | — | 4.3 | 7 | — | — | 5.1 | 5 | — | 4.6 | 9 | — | 4.1 | 7 | |||||||||||||||
| Pickard | 0.4892 | 8.9 | 3 | 8.7 | 1 | 8.4 | 4 | 8.1 | 6 | 7.9 | 5 | — | 6.7 | 5 | — | 5.9 | 9 | — | 5.5 | 0 | — | 6.4 | 0 | — | 6.0 | 1 | — | — | 5.8 | 7 | — | |||||||||||||||||
| Rogers North |
} | 0.2644 | 3.2 | 1 | 3.4 | 8 | 2.1 | 5 | 1.5 | 8 | 1.5 | 6 | 1.7 | 7 | — | 2.3 | 0 | — | 1.6 | 2 | — | 0.8 | 6 | — | 1.5 | 0 | — | — | — | — | — | { | Dry at end of 1st day. | |||||||||||||||
| Table XIV.—(a) 20 Per Cent. Dry Rosin in 80 Per Cent. Linseed Oil 100 Per Cent. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Gardner | 0.2030 | 12.0 | 14.1 | 14.8 | 14.2 | 14.5 | 14.0 | — | 14.1 | — | 14.1 | — | 14.0 | — | 14.0 | — | 14.0 | — | 14.1 | — | { | Rapid drying observed. Hard film shown during 2d day. |
||||||||||||||||||||||||||
| Sabin | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||
| Pickard | 0.5185 | 3.7 | 6 | 8.7 | 6 | 9.2 | 0 | 9.2 | 0 | 8.4 | 9 | — | 9.0 | 7 | — | 9.0 | 1 | — | 9.0 | 9 | — | 10.5 | 0 | — | 10.1 | 6 | — | — | 10.1 | 8 | — | |||||||||||||||||
| Rogers North |
} | 0.2554 | 1.8 | 0 | 11.7 | 8 | 12.1 | 7 | 12.2 | 9 | 12.0 | 2 | 12.4 | 9 | — | 13.1 | 5 | — | 11.8 | 5 | — | 11.7 | 8 | — | 12.6 | 9 | — | 12.8 | 3 | — | — | — | { | Oily, end 1st and 2d days; slightly tacky, end 10th day. |
||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Gardner | 0.1500 | 10.9 | 13.5 | 13.6 | 13.0 | 13.0 | 13.0 | — | 13.1 | — | 13.1 | — | 13.0 | — | 12.9 | — | 13.0 | — | 13.2 | — | { | Clear, hard film after 2d day. | ||||||||||||||||||||||||||
| Sabin | 0.7105 | 14.1 | 9 | 13.1 | 7 | 11.8 | 4 | 11.4 | 6 | 10.8 | 7 | — | 9.8 | 0 | 10.3 | 3 | 10.4 | 0 | 10.0 | 4 | — | 10.3 | 5 | — | — | 9.6 | 4 | — | 8.9 | 8 | — | 8.6 | 2 | |||||||||||||||
| Pickard | 0.4568 | 12.8 | 6 | 12.7 | 3 | 12.1 | 3 | 12.0 | 2 | 11.3 | 0 | — | 10.9 | 5 | — | 11.2 | 1 | — | 10.5 | 3 | — | 11.2 | 1 | — | 10.8 | 8 | — | — | 11.4 | 3 | — | |||||||||||||||||
| Rogers North |
} | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Table XV.—(a) Raw Linseed Oil 100 Per Cent.[11][41] | ||||||||||||||||||||||||||||||||||||||||||||||||
| Observer. | Wt. of Oil for Test, grams. |
Percentage Increase in Weight, in Days. | Remarks. | |||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | ||||||||||||||||||||||||||||||
| Sabin | 0.5274 | 0.2 | 6 | 0.5 | 1 | 0.1 | 1 | 2.3 | 5 | 9.1 | 4 | — | 14.4 | 8 | 14.4 | 8 | 14.1 | 8 | 13.8 | 6 | — | 13.0 | 0 | — | — | 12.2 | 3 | — | 11.6 | 6 | — | 11.0 | 7 | |||||||||||||||
| Pickard | 0.5326 | 12.4 | 2 | 12.3 | 9 | 11.8 | 8 | 11.8 | 3 | 11.0 | 8 | — | 10.2 | 9 | — | 9.5 | 6 | — | 9.8 | 5 | — | 10.3 | 0 | — | 10.1 | 2 | — | — | 10.7 | 8 | — | |||||||||||||||||
| (b) Drier 100 Per Cent. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Rogers North |
} | 0.3445 | 48.9 | 5 | 48.5 | 3 | 48.6 | 8 | 48.6 | 8 | 48.4 | 8 | 48.2 | 6 | — | 48.4 | 3 | — | 48.8 | 9 | — | 48.2 | 2 | — | 48.2 | 2 | — | — | — | — | — | { | Dry at end of 1st day. | |||||||||||||||
[10] Moth got in.
[11] The test of this oil was made without the addition of 5 per cent. of drier, the quantity used in all the other tests.
For the student of paint technology, who is not already acquainted with the chemistry and physics of the various raw pigments which are largely used in the manufacture of paints, the writer advises a careful reading of this chapter, in which the matter has been condensed as much as possible. In order to more thoroughly acquaint the reader with the physical constitution of the pigments under consideration, there has been included photomicrographs, which show to advantage the structure of each.[12]
[12] The author gratefully acknowledges the assistance of Dr. J. A. Schaeffer in the preparation of the photomicrographs shown in this chapter.
 |
 |
| By Polarized Light | By Transmitted Light |
| Basic Carbonate-White Lead | |
Basic Carbonate-White Lead. This pigment is made by stacking clay pots containing dilute acetic acid and lead buckles, in tiers, and covering them with tan bark. Fermentation of the tan bark, with subsequent formation of carbon dioxide acting on the acetate of lead formed within the pots, produces basic carbonate of lead. After complete corrosion, the white lead is ground, floated, and dried. Corroded white lead has a specific gravity of 6.8 and contains about 85% lead oxide and[43] 15% of carbon dioxide and water. Its opaque nature and excellent body renders it extremely valuable as a constituent of paints. Checking and chalking progress rapidly when the pigment is used alone. The various sized particles, both large and small, resulting from the corrosion process, are prominently shown in the photomicrograph.
On account of its alkaline nature, this pigment acts upon the saponifiable oil in which it is ground, forming lead soaps which[44] accelerate chalking of white lead—the greatest evil attending its use. Solubility in carbonic acid of the atmosphere and decay in the presence of sodium chloride may be active causes of the rapid chalking of this pigment at the seashore. Checking in some climates appears to proceed rapidly on white lead paints, in a deep hexagonal form, leaving a series of rough crests and cracks. This checking is secondary to the chalking which takes place.
[45]White Lead (Quick Process). By acting on atomized metallic lead, contained within large revolving wooden cylinders, with dilute acetic acid and carbon dioxide, the quick-process white lead is produced. Its value is equal to the Dutch-process white lead, and it is considered by some as possessing greater spreading value.

Sheet iron box luted at bottom with water. Atomized lead, blown into box with steam, falls to bottom and becomes hydrated (Mild Process)
White Lead (Mild Process). The Mild Process of manufacturing white lead consists of first melting the pig lead and converting it into the finest kind of lead powder, then mixing thoroughly with air and water. The lead takes up water and oxygen and forms a basic hydroxide of lead. Carbon dioxide gas is next pumped slowly through the cylinders which contain the basic hydroxide of lead. The result is basic carbonate of lead—the dry white lead of commerce. The process is called “Mild” because it is the mildest process possible for the manufacture of white lead. It is the only method in practical operation which does not require the use of acids, alkalis or other chemicals, every trace of which should be removed from the finished product by expensive purifying processes. The failure of such washing and purifying means a product of inferior quality, which necessarily reduces the durability of any paint in which it is used.

Steam Jected Pans for Drying White Lead
Basic Sulphate-White Lead (Sublimed White Lead). By the action of the oxygen of the air on the fume produced by the roasting and subsequent volatilization of galena, this fine, white, amorphous pigment is made. On analysis, its composition shows approximately 75% of lead sulphate, 20% of lead oxide, and 5% of zinc oxide. It has a specific gravity of 6.2.[47] Possessed of extreme stability, it finds wide use as a constituent of paints and as a base for tinting colors. The photomicrograph of this pigment shows its extremely fine, amorphous nature with complete absence of crystals. In fineness it closely approaches zinc oxide. On account of its non-poisonous properties it is replacing corroded lead in many places. Unified paints containing sublimed white lead are of great value, showing upon long exposure very little decay.

View of Goosenecks Used for Collecting Sublimed White Lead Fume

Bag Room Where Sublimed White Lead is Deposited
Photographs courtesy of Picher Lead Co.

View of largest Zinc Oxide Works in America, at Hazards, Pa.
Sublimed Blue Lead. Sublimed blue lead is made by burning coarsely broken lumps of galena, admixed with bituminous coal, in a special form of furnace. The fumes which are volatilized from this mixture are very complex in their chemical make-up, and in color are white, blue, and black. After being drawn through the cooling pipes by the suction of huge fans, whereby the fumes are cooled, the pigment is deposited in bags. This pigment is bluish black in color, and has been highly recommended for use on iron and steel. Its composition runs approximately as follows:
| Lead sulphate | 50% |
| Lead oxide | 35% |
| Lead sulphide | 5% |
| Lead sulphite | 5% |
| Zinc oxide | 2% |
| Carbon | 3% |

View of Zinc Oxide Furnaces

Photographs courtesy Geo. B. Heckel and N. J. Zinc Co.
View of Zinc Oxide Fume Pipes with electrically driven Suction Fans
The color of the pigment is largely due to the carbon and the lead sulphide. Its specific gravity is 6.4, and it grinds in 10% of oil to a stiff paste, 100 lbs. of which may be thinned with about 26 lbs. of oil to working consistency. Paint manufacturers use it in mixture with iron oxide and other pigments for the production of paints for metal surfaces. Wood and others have found it of great value for this purpose. It has a[51] tendency to chalk, but this may be overcome by admixture with other pigments such as zinc oxide and iron oxide. Lane has found it to be very durable when admixed with lampblack.

View of Bag Room receiving Zinc Oxide
Zinc Oxide. This extremely white and fine pigment is prepared by the roasting and sublimation of franklinite, zincite, and other zinc-bearing ores largely found in New Jersey. Its purity approaches in most instances 99.5 or more. It has a specific gravity of 5.2. On account of its stability, whiteness, and opacity, it is invaluable as a pigment when a constituent in a combination formula. Its extreme hardness renders it less resistant to temperature changes, when used alone. Under the microscope the fineness and structure of the particles are clearly evident. The French-process zinc oxide produced in America by the sublimation and oxidation of spelter is the purest made, and superior to imported grades which often contain ultramarine blue as a whitening agent.
 |
 |
| Zinc Oxide | Zinc Lead White |
 |
|
| Zinc Lead. By transmitted light (The Pigment shows black) |
|
 |
 |
| Lithopone | Magnesium Silicate (Asbestine) |
Zinc Lead White. This extremely fine pigment, consisting of about equal parts of zinc oxide and lead sulphate,[53] results from the reduction, volatilization and subsequent oxidation of sulphur-bearing lead and zinc ores. It has a specific gravity of 4.4. Its slightly yellowish tint bars it from being used alone very extensively, but when mixed with white lead, zinc oxide and inert pigments, or used as a base for colored paints, it is of considerable value. The magnification of the particles shows the peculiar way in which the pigment agglomerates, and the characteristics of a fine, uniform pigment.

Asbestine Mine at Easton, Pa.

American Barytes. Transmitted light
(The Pigment shows black)
German Barytes. Mag. 250 Diam.
(The Pigment shows white)
Lithopone. Lithopone, probably the whitest of pigments, results from the double decomposition of zinc sulphate and barium sulphide, thereby forming a molecular combination of zinc sulphide and barium sulphate. The peculiar property which it possesses, of darkening under the actinic rays of the sun, makes it essential that it be combined with other, more stable pigments to prolong its life when exposed to weather. Lithopone contains approximately 70% barium sulphate, 25 to 28% zinc sulphide, and as high as 5% of zinc oxide. Its specific gravity is about 4.25. It is excellently suited[55] for interior use in the manufacture of enamels and wall finishes. When properly mixed with other pigments, such as zinc oxide and calcium carbonate, fair results are obtained as a pigment for outside work. Lead pigments are never used with lithopone, as lead sulphide results, giving a black appearance. Its characteristic flocculent, non-crystalline nature is plainly evident when examined under the microscope.
 |
 |
| By Polarized Light | By Transmitted Light |
| Barium Sulphate (Barytes) | |
Magnesium Silicate (Asbestine and Talcose). This pigment comes in two forms: as asbestine and as talcose (talc, etc.). The former is very fibrous in nature and is a very stable pigment to use in the manufacture of paint, on account of its inert nature and tendency to hold up heavier pigments, and prevent settling. It also has the property of strengthening a paint coat in which it is used. The talcose variety is very tabular in form. Both varieties are transparent in oil, and very inert. They have a gravity of about 2.7 and grind in about 32% of oil.

Barium Carbonate. Mag. 250 Diam.
(The Pigment shows white)
 |
 |
| Barium Sulphate (Blanc Fixe) | Calcium Carbonate (Whiting) |
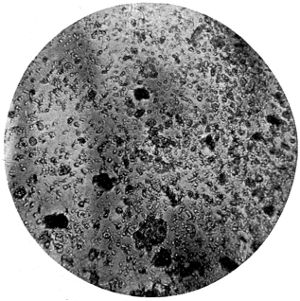
Calcium Carbonate. By transmitted light
(The Pigment shows black)

Calcium Sulphate. By transmitted light
(The Pigment shows black)
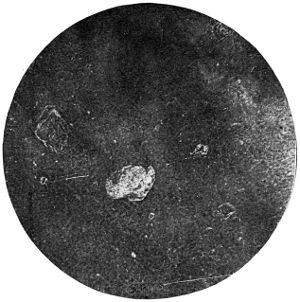
Calcium Sulfate
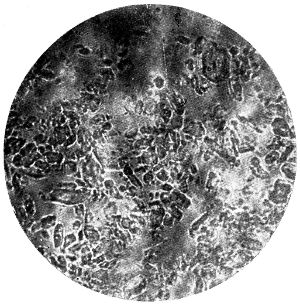 |
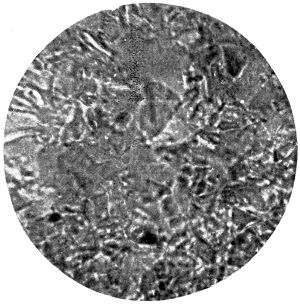 |
| Calcium Sulphate (Gypsum) | Silica (Silex) |
Silex. Mag. 250 Diam.
(The Pigment shows white)
Barium Sulphate (Barytes). By grinding the crude ore, treating with acid to remove the iron, and finally washing, floating, and drying, there is produced the commercial form of this valuable pigment. It is used in large quantity as a base upon which to precipitate colors, and also together with other white pigments in the manufacture of ready-mixed paints. It renders the paint coating more resistant to abrasion, and gives to the paint certain very important brushing qualities. It is a very stable pigment, not being materially affected by either acid or alkali, and can be used with the most delicate colors. In oil it is transparent and must be mixed with opaque pigments [60]when used in ready-mixed paints. It is generally used with lighter pigments, such as asbestine, in order to prevent settling. Under the microscope, both by polarized and transmitted light, the sharp angles of the particles appear distinctly, with no tendency to mass into a compact form. Although transparent in oil, it is valuable in moderate percentage in a ready-mixed paint.
Barium Sulphate (Blanc Fixe). Blanc fixe is the precipitated form of barium sulphate, resulting from the action of soluble barium salts on soluble sulphates. The specific gravity (4.2) of this compound is lower than that of barytes. Possessing greater opacity in oil, it is of more value as a paint pigment for some purposes. It comes in for its greatest use as a base on which to precipitate lake colors. The very fine particles show a slight tendency to agglomerate.
Calcium Carbonate (Whiting). The natural form of calcium carbonate, prepared from chalk, has a much higher specific gravity (2.74) than that of the artificial form (2.5) prepared by the precipitation of calcium carbonate. The latter, however, possesses greater hiding properties. Both grades find a wide use in distemper work and in the manufacture of putty. It is often used in small percentage in many ready-mixed paints. The photomicrograph of the pigment shows the presence of many large particles.
Calcium Sulphate (Gypsum). The mineral gypsum, consisting of calcium sulphate and about 21% of water of combination, is sometimes used as a paint pigment after grinding and dehydration. Being slightly soluble in water it has a tendency to pass into solution when exposed to atmospheric agencies. It lacks hiding power in oil. Its specific gravity is 2.3. As in the case of all pigments prepared directly from mineral substances, the many-sized and shaped particles appear clearly when enlarged. Partially and wholly dehydrated forms of gypsum are also used in paint.
Silica (Silex.) This white pigment possesses great tooth and spreading properties. It is of use as a wood filler and as a constituent in combination paints. It wears especially well when used in combination with zinc oxide and white lead. Its purity often approaches 97%. The particles when enlarged are seen to have sharp angles and are not uniform in size, which accounts for its marked tooth and properties.
 |
 |
| Aluminum Silicate (China Clay) | Ochre |
 |
 |
| Raw | Burnt |
| Sienna | |
 |
 |
| Raw | Burnt |
| Umber | |
[62]Aluminum Silicate (China Clay). China clay, or aluminum silicate, is a permanent and valuable white pigment showing very little hiding power in oil. It is found widely distributed in granitic formations. It is very stable, with a gravity of 2.6. Particles are found in many shapes and sizes, showing sharp and definite angles.
Ochre. Ochre is a hydrated ferric oxide permeating a clay base, largely used as a tinting material. It has a specific gravity of about 3.5, and a decidedly golden yellow color. A good quality should contain 20% or over of iron oxide. The particles of this pigment are flocculent and very uniform in appearance.
Sienna. Sienna, like umber, is essentially a silicate of iron and alumina, containing manganic oxide. It contains, however, a lower percentage of the latter than in the case of umbers. The photomicrograph of the burnt variety shows clearly the fine condition of the pigment, while large particles are shown in the raw variety.
Umber. Umber, another naturally occurring pigment, consists of iron and aluminum silicates, containing varying proportions of manganic oxide, its color and tone varying according to the percentage of the latter. The raw variety is drab in color, which in burning changes to reddish brown. A marked percentage of large-sized particles exist in this pigment.
Indian Red. Indian red is the term applied to natural hematite ore pigments and to those produced by the roasting of copperas (iron sulphate). They generally contain 95% or more of iron oxide, with varying percentages of silica. The pigment is heavier (specific gravity 5.2) than that of Metallic Brown. The crystalline, mineral-like structure of the particles differ greatly from the amorphous particles of Metallic Brown.
Metallic Brown. The natural hydrated iron oxide or carbonate as mined largely in Pennsylvania, yields, when roasted, a sesquioxide of iron known as Metallic Brown. It contains a high percentage of alumina and silica, and has a characteristic brown color with a gravity of 3.1. It finds wide application as a pigment for protective purposes. The particles when enlarged show the usual appearance of a natural compound which has been roasted and ground.
| No. | Name | Iron Oxide | Calc. Sulph. (CaSO4) |
Alumina (Al2O3) |
Insoluble (Silica and Silicates) |
Color | |
| FeO | Fe2O3 | ||||||
| % | % | % | % | % | |||
| 0 | Bright Red | 0.71 | 96.52 | — | — | .30 | Bright Scarlet |
| 1 | Bright Red | .71 | 95.92 | — | — | .30 | Scarlet Tone |
| 2 | Indian Red | .57 | 96.00 | .78 | 1.40 | .90 | Indian Red, Medium Shade |
| 3 | Indian Red | 0.29 | 97.82 | .85 | — | .52 | Indian Red, Dark Shade |
| 4 | Indian Red | 0.28 | 95.72 | 1.21 | 1.26 | .58 | Indian Red, Light Shade |
| 5 | Persian Gulf Mix | 4.53 | 62.25 | 1.75 | — | 27.64 | Rich, Medium Red |
| 7 | Native Red Oxide | 0.85 | 89.00 | — | 0.91 | 6.09 | Medium Red, Brownish Tone |
| 8 | Special Red | 0.57 | 43.87 | 50.88 | 2.03 | 1.30 | Scarlet Tone |
| 10 | Red Oxide | 1.44 | 60.25 | .78 | 5.41 | 15.78 | Brownish-Red |
| 11 | Venetian Red | .30 | 34.08 | 52.60 | 2.20 | 3.39 | Bright Red-Brown |
| 12 | B. Oxide | 0.58 | 67.68 | — | 2.48 | 1.97 | Dark Red Brown |
| 13 | Venetian Red | 0.29 | 25.92 | 58.62 | 2.16 | 1.42 | Medium Red Tone |
| 14 | Venetian Red | 0.57 | 35.36 | .99 | 12.06 | 47.97 | Brown |
| 15 | Metallic Brown | 2.59 | 64.00 | .63 | 5.82 | 23.42 | Rich Brown |
| 16 | Crimson Oxide | 0.57 | 66.24 | 1.77 | 3.60 | 25.63 | Rich Dark Red |
| 17 | Red Oxide | 2.30 | 80.39 | .37 | .03 | 9.63 | Medium Brown |
| 18 | Red Oxide | 0.57 | 61.28 | .97 | 2.68 | 15.94 | Light Chocolate Brown |
| 20 | Red Oxide | 7.78 | 46.72 | 1.70 | 7.64 | 20.38 | Dark Reddish Brown |
| 23 | Special French Oxide | 0.58 | 72.48 | — | 8.80 | 4.48 | Deep Chocolate Brown |
| 24 | Micaceous Black Oxide | 2.02 | 86.27 | — | 2.04 | 9.50 | Dark Gray Tone |
| 25 | Black Oxide | 33.12 | 57.12 | — | 1.44 | — | Jet Black |
| 26 | Red Oxide | 0.57 | 84.16 | 5.00 | 2.00 | .63 | Deep Red |
| 27 | Special Red | 0.57 | 38.40 | 55.62 | 2.12 | 1.53 | Medium Red |
| 28 | Oxide C | — | 30.40 | .94 | 13.60 | 42.30 | Brown |
Analysis of Iron Oxide Pigments. Because of the great consideration now being given to iron oxide paints, the writer secured a series of oxides widely used in this country, and has determined the most important constituents of each.
Basic Lead Chromate (American Vermilion). By boiling white lead with chromate of soda and subsequently treating with small quantities of sulphuric acid, American vermilion, or basic lead chromate, is prepared. It contains 98% of lead compounds, frequently free chromates, and has a gravity of 6.8. The particles appear granular and large, frequently assuming a square structure.
Red Lead. By the continued oxidation of litharge in reverberatory furnaces, red lead is produced as a brilliant red pigment with a specific gravity of 8.7. The pigment particles appear to be of many sizes, showing a slight tendency to form a compact mass.
Paranitraniline Red. Paranitraniline red, a very bright red material largely used in tinting paints, is prepared by diazotizing paranitraniline in hydrochloric acid by means of sodium nitrite in the cold. This compound is rendered insoluble when precipitated directly on barytes, by acting on it with an alkaline solution of beta naphthol. It is the most stable and permanent bright red organic pigment which the paint manufacturer uses. The particles of this pigment appear in various sizes, due, no doubt, to a massing of the particles in the precipitation process.
Chrome Yellow. The neutral chromate of lead, made from either the nitrate or acetate of lead and chromate of soda, finds wide use as a tinting pigment. When precipitated on a white pigment base, various trade names are given to it. The microscope shows clearly the physical character of this pigment.
Zinc Chromate. This pigment is made either from zinc salts and bichromate of potash or zinc oxide heated with chrome salts, frequently in the presence of acid. Like the rest of the chromate pigments, it is a very slow-drying material, often requiring over a week to set up, unless considerable drier is added. In spite of the impurities which it carries, it has shown itself to be one of the most inhibitive pigments known and has demonstrated its value in even small percentages in paints for iron and steel. It dries to a hard adherent film that tends to protect metal from corrosion.
 |
 |
| Indian Red | Metallic Brown |
 |
 |
| Basic Lead Chromate (American Vermilion) | Red Lead |
 |
 |
| Paranitraniline | Chrome Yellow |
[66]Prussian Blue. On oxidizing the precipitate resulting from the interaction of solutions of prussiate of potash and copperas (iron sulphate), Prussian blue as used in the paint trade is prepared. It has a specific gravity of 1.9. The pigment shows an amorphous structure, the particles varying greatly in size.
Ultramarine Blue. This bright blue pigment is prepared by burning silica, china clay, soda ash and sulphur in pots or furnaces. It has a specific gravity of 2.4. It is of little value as a paint pigment on account of its sulphur content, which causes darkening when mixed with lead pigments, and corrosion when applied to iron or steel. The darkness of the photograph is due to the massing of the pigment particles.
Chrome Green. Chrome green is prepared as a paint pigment from nitrate of lead, Chinese blue, and bichromate of soda. It has a gravity of 4 and is liable to contain slight traces of lead salts. The particles when magnified appear very fine and flocculent. This color is often precipitated on pigments, such as barytes, which do not reduce its tone.
Bone Black. By grinding the carbonaceous matter resulting from the charring of bones, in iron retorts, the pigment bone black is prepared. It contains about 15% of carbon and 85% of calcium phosphate. It has a gravity of 2.7. Comparatively large particles of charred bone can be seen scattered throughout the mass, resulting from the difficulty of grinding to a uniform size.
Carbon Black. This form of very pure carbon results from the combustion of gas. Its gravity, 1.09, is lower than that of lampblack, which shows a gravity of 1.8. It is used in much the same way and for the same purposes as lampblack. In physical appearance it shows great similarity to the particles of lampblack.
Lampblack. This pigment, made from the combustion of oils, consists very often of more than 99% carbon. It has wonderful tinting value. The particles show a fine, fibrous structure with a tendency toward agglomeration. They differ greatly in physical appearance from those of either graphite or bone black, being exceedingly more uniform than the latter.
 |
 |
| Zinc Chromate | Prussian Blue |
 |
 |
| Ultramarine Blue | Chrome Green |
 |
 |
| Bone Black | Carbon Black |
[68]Graphite. Graphite, both in the natural and artificial form, contains impurities such as silica, iron oxide and alumina, but the natural form has a much greater percentage of these foreign materials, in some cases as high as 40%. Graphite is usually mixed with other pigments, such as red lead and sublimed blue lead, thus serving better as a paint coating. The difference in physical appearance of the various carbon pigments is interesting, as each pigment has characteristics of its own. In graphite we find a great tendency toward agglomeration or massing of particles.
Mineral Black. Mineral black is a pigment made by grinding a black form of slate. It contains a comparatively low percentage of carbon and consequently has low tinting value. It finds use as an inert pigment in compounded paints, especially for machine fillers. The pigment has a flocculent appearance, the particles showing a strong tendency to mass.
Photomicrographs of two combination paint pigments are here given, to show the various pigments as they appear under the microscope, when in combination.
| White lead (corroded) | 9% | Chrome green, 25%, color extra dark | 17% |
| White lead (sublimed) | 10% | Graphite (pure) | 40% |
| Zinc lead (American) | 12% | Indian red, (98%) | 20% |
| French process zinc oxide | 17% | Ochre, yellow, American | 26% |
| American process zinc oxide | 16% | Ochre, yellow, French | 28% |
| Blanc fixe | 30% | Ochre, golden | 28% |
| Barytes (natural) | 9% | Red, Venetian | 23% |
| Paris white (whiting) | 20% | Red, Oxide | 25% |
| Terra alba (gypsum) | 22% | Red, Tuscan | 27% |
| Floated silica or Silex | 26% | Red, Turkey | 28% |
| Kaolin (China clay) | 28% | Red, lead | 12% |
| Asbestine | 32% | Red, lake | 55% |
| Blue, ultramarine | 27% | Sienna, Italian, raw | 52% |
| Blue, Chinese or Prussian | 50% | Sienna, Italian, burnt | 45% |
| Black, gas carbon | 82% | Sienna, American, burnt | 38% |
| Black, lamp | 72% | Sienna, American, raw | 40% |
| Black, drop | 60% | Ultramarine green | 28% |
| Black, bone | 50% | Umber, Turkey, raw | 48% |
| Brown, mineral | 24% | Umber, Turkey, burnt | 47% |
| Brown, vandyke | 50% | Umber, American, burnt | 36% |
| Chrome yellow, lemon | 23% | Umber, American, raw | 38% |
| Chrome yellow, medium | 30% | Verona green (terra verte or green earth) | 32% |
| Chrome yellow, orange | 20% | Vermilion, English (quicksilver) | 14% |
| Chrome yellow, dark orange | 15% | Vermilion, American (chrome red) | 16% |
| Chrome green, Chem. pure light | 21% | Paris green, American | 23% |
| Chrome green, Chem. pure extra dark | 25% | Zinc chromate (permanent yellow) | 15% |
| Chrome green, 25%, color light | 13% |
 |
 |
| Lampblack | Graphite |
 |
|
| Mineral Black | |
 |
 |
| Asbestine and Whiting | Silica and Asbestine |
For the paint chemist who desires to familiarize himself with the more recent analytical methods worked out in American laboratories, reference may be had to treatises on the analysis of paints, by Gardner and Schaeffer,[13] and Holley and Ladd.[14] Analytical methods are not included in this chapter, the writer’s desire being to treat the subject from the standpoint of the physical properties of painting materials. The work outlined herein is of a nature that affords a wide field of research, and a brief study will doubtless suggest similar work to the student of paint.
[13] The Analysis of Paints and Painting Materials. McGraw-Hill Book Co., New York, 1910.
[14] Mixed Paints, Color Pigments and Varnishes. John Wiley & Sons, New York, 1908.
Preparation of Paint Films. The study of paint films is one that has become of vital importance, and is receiving at the present time great attention. Among the many methods which have been suggested and attempted for securing paint films, a few already well known may be mentioned.
By painting upon zinc and eating away the zinc with acid: The objection to this method is very evident, namely, the action of the acid upon the paint coating, which is likely to be very severe. Another method has been to spread paraffin on a glass plate, and painting upon this surface. When the paint is dried, the paraffin is melted off and thus the film is obtained. This method is open to objections, in that the paraffin surface is not a comparable one upon which to paint, and also that the complete removal of the paraffin is not assured.
Another method consists in covering a piece of glass with tin foil, painting out the film upon the foil, and after drying properly, to remove the sheet of foil with its coating of paint and immerse in a bath of mercury which, by amalgamation of the tin, leaves the paint film.
[71]We now come to a method worked out in our laboratories, which can be recommended as being not only simple but efficient and practical. It has been found that a size from noodle glue, when painted upon ordinary fair-quality paper, makes a surface from which the paint may be subsequently stripped. The paint is applied in the ordinary way to the paper, which is held during the operation by thumb tacks, and allowed to dry. The paint may be separated by immersion in water kept at about 50 degrees Centigrade. By this method large films may be obtained, but it has been found very unhandy to work with films exceeding an area of eight inches square. When the film of paint has been detached from the sized paper through the dissolving of the noodle glue, the paint film is then immersed in a fresh solution of water, in order to remove whatever excess of noodle glue there may be remaining. A glass rod is then introduced into the bath, in which the paint film is floated upon the glass rod, which is then hung up to dry in a suitable container to prevent the accumulation of dust, etc.

Bottles Showing Relative Permeability of Films by Amount of Whiting Formed Within
The Permeability of Paint Films. A series of tests were made to determine the water-excluding values of various combinations of painting pigments ground in pure linseed oil. White pine boards, six inches long, four inches wide, and one inch thick, were[72] carefully prepared and numbered and given three coats of a white paint formula of the corresponding number. After drying, the boards were carefully weighed and immersed in a tub of water for three weeks. After removal, the surfaces of the boards were dried with blotting paper and the boards weighed. The gain in weight, corresponding to the amount of water penetrating through the pores of the wood, was observed. The boards were again immersed and at the end of two months the following results were obtained:
| Formula No. |
Grammes of water absorbed through paint |
||||
| 1. | Soya bean oil | 120 | |||
| 2. | Linseed oil | 102 | |||
| 3. | Calcium sulphate | 93 | |||
| 4. | Barytes | 88 | |||
| 5. | Asbestine | 74 | |||
| 6. | Corroded white lead | 59 | |||
| 7. | ⎧ | Basic carb.—White lead | 25% | ⎫ | 58 |
| ⎪ | Basic sulph.—White lead | 20% | ⎪ | ||
| ⎨ | Zinc oxide | 25% | ⎬ | ||
| ⎪ | Calcium sulphate | 25% | ⎪ | ||
| ⎩ | Calcium carbonate | 5% | ⎭ | ||
| 8. | Sublimed white lead | 56 | |||
| 9. | Zinc oxide | 56 | |||
| ⎧ | Zinc lead white | 30% | ⎫ | ||
| ⎪ | Zinc oxide | 40% | ⎪ | ||
| 10. | ⎨ | Basic carb.—White lead | 20% | ⎬ | 42 |
| ⎩ | Calcium carbonate | 10% | ⎭ | ||
| 11. | { | Basic carb.—White lead | 50% | } | 42 |
| Zinc oxide | 50% | ||||
| 12. | ⎧ | Basic carb.—White lead | 38% | ⎫ | 38 |
| ⎨ | Zinc oxide | 48% | ⎬ | ||
| ⎩ | Silica | 14% | ⎭ | ||
The test boards were then exposed, with their content of water, to the action of the sun’s rays. Blistering of the painted surfaces took place in many cases, caused by the rapid withdrawal of the water and its consequent action on the paint film. The tests seem to indicate that a mixture of white lead and zinc oxide, with or without a small percentage of the inert pigments, is not as subject to the action of the water as the single pigment paints. In order, however, to corroborate these tests, it occurred to the writer to develop a more visible means of demonstrating the passage of moisture through paint films.

Bell Jar Apparatus for Testing Permeability of Paint Films
Paint films sealed over mouths of Bottles containing Lime Water. Carbonic Acid Gas generated under Bell Jar passes through Plate Films and precipitates Calcium Carbonate
[74]Another series of white pine boards were therefore soaked in a solution of iron sulphate for several hours. After removal, the surface of each board was dried and coated with one coat of the paints previously tested. After thorough drying for forty-eight hours, there was placed on the surface of each board a few drops of a solution of potassium ferrocyanide. This solution has the effect of producing a blue coloration with iron sulphate, and in every case when it was placed on a paint of considerable porosity, the solution penetrated through and formed a blue coloration beneath the paint. The results corroborated the original tests referred to above.
A series of sheets or films of paints were then prepared according to the method referred to on page 71. These films were placed over glass dialyzing cups, allowing the inner surfaces to sag so as to hold a small amount of dilute ammonium chloride solution. Distilled water was placed on the reverse side of the dialyzing apparatus and the tests started. At the end of six days the distilled water in each test was examined and the following results obtained:
Test No. 1 (corroded white lead and asbestine film) allowed
the passage of 0.002 gm. ammonium chloride.
Test No. 2 (corroded white lead and zinc oxide film) allowed
the passage of 0.0003 gm. ammonium chloride.
Tests were also made with dilute solutions of other salts such as ferric chloride, having a dilute solution of potassium sulpho-cyanide on the reverse side of the apparatus. In the latter case the formation of a pink color, characteristic upon the mingling of these solutions, was obtained in a few hours.
Film-Testing Machine. A film-testing apparatus, termed a “filmometer” by its originator, Mr. R. S. Perry, was constructed, with the following features: A graduated upright tube is fixed by means of sealing wax to two metallic plates which carry an evenly bored hole, exactly under the hole in the upright tube. This hole measures exactly one square centimeter in area, and is circular. The upright tube is graduated into lineal centimeters and is called the pressure tube.
Attached to the lower end of this pressure tube, close to the metallic plates which serve as carriers for the paint film to be tested, is a side-neck, which is inclined at an angle of 45 degrees to the pressure tube, and serves the purpose of introducing the mercury, as will be described later. Immediately under the openings in the metallic plates which carry the film are arranged two pieces of iron inclined at a 90-degree angle, so arranged that when the pressure of mercury is applied and causes rupture of the film, the falling mercury shall be caught between these two insulated plates and cause contact. These two plates are connected up by wire with a pair of magnets, thence to an electric bell, and from there to storage batteries which supply the current.
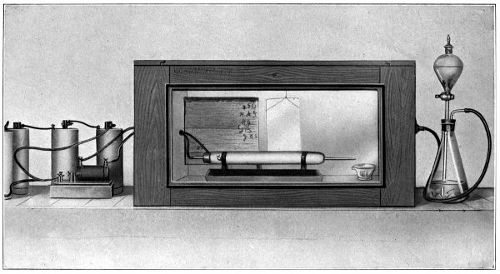
Gardner Accelerated Test Box
[77]A film of paint is tested in the following manner: A piece of film one inch square is cut out and placed between the two metallic plates which hold the film immediately under the pressure tube. Mercury is run in from a burette through the side-neck and applies pressure upon the film by gravity. As the mercury is run in it rises of course in the tubes until this pressure becomes so great as to finally break the film. At this point the mercury will run out, and, falling upon the two insulated iron plates immediately below, will cause contact and close the circuit which rings an electric bell, which is a signal for the operator to shut off the inflow of mercury through the side-neck from the burette.
The pressure tube is also supplied with a piston which is made of a piece of thin iron wire having a disc attached to its lower end. As the mercury rises in the pressure tube this iron wire is pushed up, being very delicately counterpoised over a wheel. Upon the breaking of the film the mercury runs out, but upon falling upon the two iron plates underneath causes contact to be made, which causes the current to run through the pair of magnets before mentioned, which, becoming electrified, attract the piston in the pressure tube, giving a reading for the maximum height of the column of mercury.
The supply of mercury being shut off, the operator is now in a position to determine the total sum of both the elasticity and ductility of the paint film, and also the pressure at which the film broke. The breaking pressure of course is read directly upon the pressure column, which is divided into centimeters as has been described above, the piston indicating the maximum height of the mercury column. What may be termed the elasticity of the film can now be calculated. As is perfectly evident, the film in stretching does so by distending from a flat surface to a curved or cup-like surface. If the pressure tube is calibrated in cubic centimeters reckoned from a flat surface where the film was introduced, the stretch of the paint film in distending from a flat surface to a curved surface may be[79] determined. The cubic contents of the pressure tube and side-arm become increased, owing to the cup-like shape the paint film takes on. By subtracting the amount of mercury indicated by the piston in the pressure tube from the amount of mercury delivered from the burette, the amount contained in the distended paint film is obtained, which serves as a measure of elasticity. The temperature is a most important point to consider in running daily tests upon the filmometer. The tests made by the writer were conducted at 70 degrees Fahrenheit throughout.
Gardner-de Horvath Filmometer. Another type of filmometer which gives very concordant results was recently devised by the writer and de Horvath. This apparatus is shown above.
[80]It consists of a three-necked Wolff bottle having provision at one of its necks for exhausting the air from the bottle. The reverse neck is provided with a gauged glass tube dipping into a porcelain crucible containing mercury, thus acting as a manometer. The middle neck is fitted to accommodate two ground glass plates. Both these plates are provided with a central orifice one millimeter in diameter. Between the plates is placed a small section of paint film. The plates may be pressed together or clamped together and placed over the middle neck of the bottle, a close contact being made with Canada balsam. As the air is exhausted from the bottle, the mercury in the tube will rise and continue in its ascent until the film, which is exposed to atmospheric pressure, has offered it maximum resistance, which is shown by the breaking point. This point is observed on the manometer and the result expressed in centimeters of mercury.
Table of Film Testing Results. By means of the Perry film-testing apparatus, described in the above, interesting results have been obtained, which are embodied in the following table:
| No. Coats | Pressure | Thickness | Stretch | ||
| 1. | Zinc oxide | 3 | 33.2 | 0028 | .30 |
| 2. | Zinc lead | 3 | 32.7 | 0034 | .35 |
| 3. | Asbestine | 3 | 28.0 | 0045 | .15 |
| 4. | Sublimed white lead | 3 | 17.9 | 0024 | .38 |
| 5. | Barytes | 3 | 13.3 | 0042 | .33 |
| 6. | Lithopone | 3 | 13.1 | 0024 | .49 |
| 7. | Whiting | 3 | 13.0 | 0033 | .32 |
| 8. | Quick process white lead | 3 | 11.3 | 0025 | .38 |
| 9. | Gypsum | 3 | 10.8 | 0039 | .29 |
| 10. | China clay | 3 | 10.8 | 0035 | .16 |
| 11. | Silex | 3 | 9.6 | 0032 | .32 |
| 12. | Blanc fixe | 3 | 8.5 | 0030 | .28 |
| 13. | Corroded white lead | 3 | 7.3 | 0020 | .33 |
| 14. | Barium carbonate | 3 | 7.2 | 0028 | .16 |
By means of this machine it is possible to obtain very valuable information concerning the effect of age upon a paint as influencing its strength and elasticity. These are two vital qualities in a paint, as it is through its strength that a paint resists abrasion, cracking, peeling, and blistering. That elasticity is a vital qualification[81] of a paint may easily be seen through the checking of oil paintings, which, as Ostwalt has pointed out, is due to the unequal coefficients of expansion between the ground and the paint. This is particularly noticeable in the alligatoring of many enamels which contain large percentages of zinc.
Curves have been prepared having pressure as an abscissa and elasticity as ordinate. These curves show remarkable differences in different pigments. For instance, in the case of white lead, the curve takes a steep upward trend when it apparently reaches a maximum, the curve then flattening out and finally taking another steep upward trend just before breaking. This may be construed as follows: That under low pressures the white lead film is perfectly elastic, when a maximum is obtained, beyond which elasticity does not extend. This point is the maximum point of the upward trend. From here on pressure may be applied without any increase in stretch, this being represented by the flat part of the curve, while the steep upward trend just before breaking shows where the paint begins to tear, finally culminating in breaking. In the case of asbestine, however, the curve is more of a straight line up to the breaking point, which would go to prove that elasticity is proportionate to pressure in the case of this pigment.
Moisture Absorption. The structure of certain pigments is such that when they are ground in linseed oil and painted out, films are produced which are very water-resistant. This action is possibly due to the filling of the voids in the oil, thus making a compact and water-resistant film. Pigments which are coarse and which present an angular crystalline structure, often produce films which contain a relatively large number of voids and are less waterproof. Certain pigments are chemically active and tend to produce, when ground in oil, metallic soaps which act for a time more or less as varnish gums, in keeping out moisture. Later on, however, such films are apt to break down and admit moisture in quantity. The tests herein described were designed by the author to determine the water-excluding value of a number of typical pigments when ground in linseed oil and painted out into films. Unfortunately, no method has been devised by which films of the same gauge could be prepared. The variations in the thickness of the films used in these experiments, however, are not very great.
[83]A series of small glass bottles with wide mouths, holding about two ounces, were half filled with concentrated sulphuric acid, and paint films were tightly sealed over the mouths of the bottles with Canada balsam. The bottles were then carefully labeled, numbered, and accurately weighed upon chemical balances. Later they were exposed under a large glass bell jar containing air saturated with moisture and kept at a constant temperature. The bottles were removed from the receptacle every week and reweighed. The increase in weight, due to the amount of moisture which had penetrated through the films, and absorbed by the sulphuric acid, owing to its hygroscopic nature, was thus determined. In another series of bottles, lumps of calcium chloride were substituted for the sulphuric acid. The results obtained from these tests correspond to those of the former tests, and led to the conclusion that the porosity of linseed oil films varied when different pigments were used in the oil.
Figures Given Express Percentage Gain in Weight,
e.g., Water Absorbed
| 7 days | 21 days | 49 days | ||||
| White lead and zinc oxide | 0.043 | % | 0.115 | % | 0.266 | % |
| Zinc lead white | 0.049 | 0.130 | 0.284 | |||
| Red lead | 0.049 | 0.130 | 0.295 | |||
| Sublimed white lead | 0.049 | 0.128 | 0.292 | |||
| Zinc chromate | 0.064 | 0.176 | 0.417 | |||
| Zinc oxide | 0.065 | 0.172 | 0.391 | |||
| Barytes | 0.074 | 0.202 | 0.466 | |||
| Willow charcoal | 0.077 | 0.236 | 0.694 | |||
| Lithopone | 0.083 | 0.228 | 0.550 | |||
| Chinese blue | 0.092 | 0.276 | 0.671 | |||
| Natural graphite | 0.104 | 0.350 | 0.951 | |||
| Ultramarine | 0.119 | 0.336 | 0.814 | |||
Another series of tests was started, in which were used films prepared from various oils and varnishes made especially for the test from different gums. The results of this series are very interesting, as they indicate that certain gums are more powerful than others in making oils resistant to moisture. The reader should study with care the data on treated Chinese wood oil, most excellent results having been obtained when it was used in the proper percentage.
| 6 days | 18 days | 24 days | ||||
| Linseed oil, 100% | .233 | .68 | 6 | .89 | 5 | |
| Soya bean oil, 100% | .340 | 1.06 | 1.39 | |||
| Linseed oil, 80% | } | .250 | .75 | 5 | .98 | 7 |
| Soya bean oil, 20% | ||||||
| Linseed oil, 60% | } | .289 | .85 | 7 | 1.12 | 5 |
| Soya bean oil, 40% | ||||||
| Linseed oil, 40% | } | .355 | 1.05 | 1.39 | ||
| Soya bean oil, 60% | ||||||
| Linseed oil, 20% | } | .260 | .78 | 9 | 1.03 | |
| Soya bean oil, 80% | ||||||
| China wood oil treated, 100% | .130 | .29 | 7 | .37 | 5 | |
| Linseed oil, 80% | } | .182 | .55 | 9 | .72 | 8 |
| China wood oil treated, 20% | ||||||
| Linseed oil, 60% | } | .173 | .54 | 0 | .70 | 8 |
| China wood oil treated, 40% | ||||||
| Linseed oil, 40% | } | .119 | .34 | 8 | .45 | 0 |
| China wood oil treated, 60% | ||||||
| Linseed oil, 20% | } | .127 | .37 | 5 | .49 | 4 |
| China wood oil treated, 80% | ||||||
| Kauri gum, 33% | ⎫ | .061 | .19 | 1 | .30 | 2 |
| Linseed oil, 33% | ⎬ | |||||
| Turpentine, 33% | ⎭ | |||||
| Kauri gum, 25% | ⎫ | .096 | .26 | 6 | .34 | 6 |
| Linseed oil, 50% | ⎬ | |||||
| Turpentine, 25% | ⎭ | |||||
| Kauri gum, 20% | ⎫ | .122 | .36 | 7 | .44 | 9 |
| Linseed oil, 60% | ⎬ | |||||
| Turpentine, 20% | ⎭ | |||||
| Kauri gum, 15% | ⎫ | .187 | .42 | 1 | .60 | 1 |
| Linseed oil, 70% | ⎬ | |||||
| Turpentine, 15% | ⎭ | |||||
| Congo copal gum, 20% | ⎫ | .228 | — | — | ||
| Linseed oil, 50% | ⎬ | |||||
| Turpentine, 30% | ⎭ | |||||
| Sierra Leone copal, 20% | ⎫ | .099 | — | — | ||
| Linseed oil, 50% | ⎬ | |||||
| Turpentine, 30% | ⎭ | |||||
| Zanzibar gum, 20% | ⎫ | .082 | — | — | ||
| Linseed oil, 50% | ⎬ | |||||
| Turpentine, 30% | ⎭ | |||||
| Amimi gum, 20% | ⎫ | .080 | — | — | ||
| Linseed oil, 50% | ⎬ | |||||
| Turpentine, 30% | ⎭ | |||||
| Boiled linseed oil (linoleate type) | .210 | — | — | |||
| Collodion solution (6 oz.), 80% | } | .201 | — | — | ||
| Boiled linseed oil, 20% | ||||||
 |
 |
| Microscopic view of section of cedar | Microscopic view of section of maple |
 |
|
| Microscopic view of section of white pine | |
Gardner photomicroscope in position against painted surface
[86]Use of the Microscope. 4. The microscope is a necessary adjunct of every well-ordered paint laboratory, as has been recognized throughout the whole paint industry. The writer has attempted to collect certain data which may materially assist those manufacturers who employ this instrument to judge of the quality of their raw and finished products. The fineness of grinding considerably affects the quality of the paint, and this can be easily controlled through the intelligent use of the microscope. This instrument may also be used to detect certain adulterations. Appended is a table giving the fineness of grinding of the various pigments, together with their characteristics under the microscope. In this table measurements are given both in millimeters and in inches, in order to accommodate itself to the use of those chemists employing a millimeter stage micrometer, or those employing the English or inch system. Although it is not yet certain that any and all combinations of pigments may be detected under the microscope the writer is working toward a method which will allow a manipulator to judge of the composition of the paint under observation.

Inside White on White Pine
In order to properly prepare a paint for microscopic examination, the following method is recommended: A microscopic turn-table[87] is a convenient accessory of the microscope, and its use is to be recommended. A glass slide being placed in position upon the turn-table, a very small amount of either the pigment rubbed up in oil, or the paint, is applied to the slide; a small drop of Canada balsam is then applied by means of a glass rod dipped in a solution of balsam in xylol, and dropped upon the slide. The rod is then used to thoroughly incorporate the pigment with the balsam, and a cleaned cover glass is dropped over the whole and pressed down tightly, so that a small amount of balsam will exude from under the edges and thus firmly seal the glass. In order to make permanent slides it has been found advisable to rim the cover glass with balsam and even follow this up with some suitable black varnish, the slide being then carefully labeled and placed in the collection. Following is a table of the characteristics of the fourteen chief pigments:
| No. | Name | Diameter in Millimeters | Diameter in Inches | ||||||||||
| Small | Aver. | Large | Small | Aver. | Large | ||||||||
| 1 | Asbestine | .002 | — | .12 | .00015 | — | .049 | ||||||
| 2 | China clay | .003 | — | .06 | 5 | .00009 | — | .025 | |||||
| 3 | Barium carbonate | .000 | 76 | .005 | 5 | .01 | 72 | .00003 | .00024 | .001 | 1 | ||
| 4 | Blanc fixe | .000 | 73 | .003 | 7 | .00 | 73 | .00003 | .00014 | .000 | 3 | ||
| 5 | Silex | .003 | 7 | .009 | 2 | .03 | .00014 | .00036 | .001 | 2 | |||
| 6 | Gypsum | .003 | 7 | .011 | .05 | .00014 | .00044 | .002 | 2 | ||||
| 7 | Amer.-Paris white | .001 | 5 | .005 | 0 | .04 | .00006 | .00022 | .001 | 8 | |||
| 8 | Barytes | .001 | 5 | .009 | 2 | .05 | .00006 | .00036 | .002 | 1 | |||
| 9 | Zinc lead | .000 | 37 | .001 | 8 | .00 | 37 | .00001 | 4 | .00007 | .000 | 14 | |
| 10 | Sublimed white lead | .000 | 37 | .001 | 8 | .00 | 37 | .00001 | 4 | .00007 | .000 | 14 | |
| 11 | Lithopone | .000 | 76 | .001 | 8 | — | .00003 | .00007 | — | ||||
| 12 | Zinc oxide | .000 | 46 | .001 | 8 | .00 | 037 | .00002 | .00007 | .000 | 14 | ||
| 13 | Quick Pro. lead | .000 | 61 | .003 | 0 | .00 | 48 | .00002 | .00012 | .000 | 18 | ||
| 14 | Dutch Pro. lead | .000 | 61 | .001 | 8 | .00 | 66 | .00002 | .00007 | .000 | 26 | ||
Film Sectioning and Deductions to be Drawn Therefrom. 5. Investigations were undertaken into the innermost structure of paint films as revealed under the microscope. Up to the present time, work has been done upon barytes, asbestine, blanc fixe, and white lead, painted upon wood, and a combination paint upon wood. The films, the preparation of which has been described in the foregoing, were sectioned and prepared for microscopic examination in the following manner:
[88]A solidifying dish was partly filled with low melting-point paraffin which was allowed to harden, when a small piece of paint was thrown upon it and then more paraffin poured over it. After hardening, sections were obtained of the paint film by means of a microtome.
Section Barytes Film
A view of these sections of paint films under the microscope gave to the operator a better idea of the structure of a paint than had ever been afforded heretofore. It was easy to perceive the relative position of the pigment particles and the three coats. The penetration of one coat into another was easily discernible, and measurements were made upon the sections in order to determine the average thickness of coat and its general appearance.
Sections were also made of Inside and Outside White upon wood. These sections revealed under the microscope the thickness of the coats and also the penetration of the priming coat into the wood. Appended is a table giving microscopic measurements.
| Millimeters | Inches | |||
| Barytes | 3 coats (sum) | .1068 | .0042 | 1 |
| Single coat | .0356 | .0014 | 0 | |
| Inside. White on wood | 3 coats (sum) | .1624 | .0063 | 9 |
| Outside coat | .0230 | .0009 | 1 | |
| Next coat | .0443 | .0017 | 5 | |
| Field in photographs | Next coat | .0620 | .0024 | 5 |
| Penetration | .0294 | .0011 | 6 | |
| White lead | Inside | .0215 | .0008 | 5 |
| Middle | .0405 | .0015 | 9 | |
| Outside | .0184 | .0007 | 3 | |
| 3 coats (sum) | .0811 | .0031 | 9 | |
| Asbestine | 3 coats (sum) | .1840 | .0072 | 5 |
| Blanc fixe | 3 coats (sum) | .1068 | .0042 | |
| Single coat | .0356 | .0001 | 4 | |
| Outside. White on wood | Outside coat | .1329 | .0052 | 3 |
| Inside | .1845 | .0072 | 7 | |
| Penetration | .0737 | .0029 | 0 | |
Polar Micro-Examinations and Photomicrographs. By Polar Micro-Examination is meant the examination of pigments under polarized light. A polarizing apparatus, though not an essential in the hands of the paint chemist, is nevertheless much to be desired, for by its help deductions may be drawn as to the contents of a paint, which by other means might not be possible. The polarizing apparatus as marketed by most manufacturers of the microscope is attached in the following manner:
The diaphragm immediately under the sub-stage container is swung out and opened to its widest limit, allowing the insertion of the polarizer. This polarizer carries one of the pair of Nicols prisms and is countersunk to allow of the introduction of gypsum or selenite plates. The analyzer fits over the eyepiece on the tube.
The use of polarized light upon paint is valuable on account of its action upon crystalline substances. The re-enforcing pigments, such as Asbestine, China Clay, Gypsum, Silex, Barytes, etc., are crystalline and consequently act upon the polarized light. In most cases these pigments are used in ready-mixed paints in small amounts, varying between 5 and 25%. When a slide containing a small amount—for example, less than[90] 3%—of these crystalline pigments is examined under the microscope by ordinary transmitted light, they will often escape observation, owing to the small amount in which they are present. However, in the case of polarized light, this could hardly happen.
Microscopic View of Barytes under Polarized Light
A slide of paint containing these re-enforcing pigments is prepared in the usual manner. On examining this under the microscope and using the polarizing apparatus, the crystalline pigments are at once detected by revolving the analyzer. At one position of the analyzer, one sees an ordinary field, as with transmitted light, but if one revolves the analyzer, the field gradually becomes darker until total darkness is obtained throughout, except in such places where crystalline substances are present, when the crystal is shown up with beautiful distinctness. Photomicrographs of various single pigments and pigment combinations are shown under Chapter III.
Effect of Pigments on Oil. Certain pigments have the property of acting upon the linseed oil in which they are ground, forming metallic linoleates which accelerate the drying of oil. This is especially true of lead and zinc pigments. The inert crystalline pigments, when ground in linseed oil and painted out, distribute the oil so as to allow a great surface to be exposed to the air. Thus by physical action, and possibly catalytic or contact action, these inert pigments stimulate the drying of oil[91] paints in which they are ground. Lead and zinc paints, of course, have the greatest drying values on account of the added effect of the linoleates formed, as outlined above. The writer has made a series of tests in which the action of various pigments upon linseed oil is shown. The tests were made in the following manner:
Five grams of each of a series of commonly used paint pigments, including those of inert crystalline nature as well as the more valuable amorphous pigments which are considered more or less chemically active, were ground separately in an agate mortar, with 5 grams of raw linseed oil. The ground paste in each case was placed in a marked glass beaker, and allowed to stand in a dustless section of the laboratory for one month. The oil-pigment paste from each beaker was then separately extracted with benzine to remove the linseed oil from the pigment. The benzine solutions of oil were then heated to remove the benzine and the residue of oil burned to ash in crucibles. The ash from each test was weighed, and if it ran above the percentage of ash determined on a blank sample of linseed oil (namely, .003%), the ash was analyzed qualitatively for metallic constituents. The following table of results shows the percentage increase in ash, as well as the constituents of ash on the various samples tested:
| Pigment in Oil | Per cent. of Ash in Oil Extracted from Oil-Pigment Paste |
Analysis of Ash | |
| Raw linseed oil without pigment | 0.003 | — | |
| Barytes | 0.003 | — | |
| Blanc fixe | 0.003 | — | |
| Silica | 0.003 | — | |
| Asbestine | 0.005 | — | |
| China clay | 0.007 | — | |
| Whiting | 0.008 | — | |
| Chrome yellow | 0.025 | Lead oxide (PbO) | |
| Lithopone | 0.031 | Zinc oxide (ZnO) | |
| Prussian blue | 0.032 | Iron oxide (Fe2O3) | |
| Sublimed white lead | 0.033 | Lead oxide (PbO) | |
| Zinc oxide | 0.105 | Zinc oxide (ZnO) | |
| Corroded white lead | 0.116 | Lead oxide (PbO) | |
| Red lead | 0.211 | 2 | Lead oxide (PbO) |
[92]Observation of these results shows that pigments such as Barytes, Blanc Fixe, and Silica have no chemical action on the linseed oil. The results on Asbestine and China Clay also are negative, the extremely slight increase in amount of ash from these samples probably being due to traces carried over mechanically into the oil mixture; the last named pigments being more fluffy and difficult to separate from oil. Slight action seemed to be apparent in the case of whiting, a pigment somewhat alkaline in nature. A longer test might have shown this pigment to have possessed still greater action. Corroded white lead showed considerable action, resulting in the formation of lead linoleate or some other organic compound. Zinc oxide and lithopone, the latter pigment containing 30% of zinc sulphide, both indicated action on the oil. Chrome yellow (chromate of lead) showed some action, as did also Prussian blue, the ash from the last named pigment showing a heavy percentage of iron oxide.
Red Lead showed a most astounding gain in these tests, chemical action of the pigment on the oil being apparent soon after the tests were started, as shown by the formation of a hard cake with the linseed oil.
The Raw Linseed Oil which was used in these tests had an acid value of 1.84%, which is very low. The neutralization of this free fatty acid by some of the alkaline pigments used, may account for part of the increased percentage of ash, but in most cases the pigments, and more especially the basic pigments, had a direct saponifying action upon the glycerides of the oil.
Laws of Paint Making. To secure a proper comprehension of the composition of paints, and to be able to interpret the functions of their various constituents, requires an understanding of the general physical principles involved. The modern grinder has accepted the Law of Minimum Voids, and upon this law he bases the design of paint formulæ, aiming toward the production of what have been properly termed Scientifically Prepared Paints. Perry’s formulation of the Law of Minimum Voids in a paint coating, and the analogy which he has drawn between a scientifically prepared paint and a well-proportioned concrete, was the result of genuine scientific thought following observation and experimentation. It must be admitted that analogies are not always safe to draw conclusions from, but it surely is no fallacy in reasoning to draw analogies between these two materials, when they resemble each other in so many ways. To carry out processes of reasoning, and to formulate laws from such close analogies, is certainly a step in the right direction.
A graphic summary of the analogies between a properly proportioned concrete and a paint, are shown on next page.
Although this table graphically summarizes the principles involved, the matter is presented with greater clearness in the following:
Law No. 1—The law of minimum voids to be observed in constructing a paint formula—this law having already been accepted as mathematically correct and technically proved in the technology of concrete and cement.
Corollary—The requisite thickness of a paint film together with the utmost attainable strength and impermeability can best be obtained by a properly proportioned blend of pigments of three or more determinate sizes.
| 1 | Concrete aggregate = solids + vehicle | Paint aggregate = solids + vehicle | |||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 |
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 |
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||
| 4 |
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||
| 5 | Vehicle + reaction = hydrosilicates, etc. | Vehicle + reaction = linoxyn | |||||||||||||||||||||||||||||||||||||||||||||||||||
| (setting) | (drying) | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 |
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||
| 7 | Final product = concrete | Final product = paint coating | |||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
| **** | **** | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| If we assume for both paint and concrete | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| proper lubrication | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| proper proportion of vehicle and solids | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Then the essential difference between a thin film of | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concrete | and | Paint | |||||||||||||||||||||||||||||||||||||||||||||||||||
| is | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cement Binder | Linoxyn Binder | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Disadvantages | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-elastic and hence an impracticable binder for a film to protect non-similar structural surfaces. | Slowly perishable from oxidation by the air. | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Advantages | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Durable and with the qualities of a natural mineral. | Semi-elastic and therefore a practicable binder for a film to protect structural surfaces. | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Postulate (def. Webster's Dictionary—A self-evident problem) | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Postulate No. 1—The organic linoxyn or semi-elastic binder of the paint vehicle (unlike the cement binder) is perishable and its purity, strength and protection from attack means life to the paint coating, and hence the life of the oil is the life of the paint. | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Postulate No. 2—The inorganic or powdered mineral solids of a paint coating will crumble unless held together by the binder, but the imperishable pigments must be so ground and blended in the binder that they will protect the binder and present the greatest possible solid front to the atmospheric attack. | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| **** | **** | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| A paint, to secure the greatest protection and life for the linoxyn, together with the durable qualities of cement, | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Therefore | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Should expose to air decay | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| within limits of physical strength | within limits required for elasticity, | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| The greatest amount of pigm't material | etc. The least amount of exposed linoxyn | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| (which is) | or | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Durable and with the inert qualities of natural mineral | Considering the linoxyn present between pigment particles as the void or point of attack, | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Then | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| the minimum exposure of linoxyn | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| or minimum voids obtainable by proportioned pigments of different particle sizes. | |||||||||||||||||||||||||||||||||||||||||||||||||||||
[95]Law No. 2—The law of the flat arch in paint coatings—i.e., the fact that in studying the fundamental physical principles governing the strength and durability of a paint coating it is necessary to regard the coating as consisting of a series of flat arches, in which the pigment particles of largest characteristic size serve as the piers or supports for the flat arches of which the continuous film is composed.
Corollary A—The strength and durability of a paint coating is determined by the strength and durability of the piers or supports (which consist of the characteristic pigment particles of the largest size).
Corollary B—Owing to their inherent strength and durability the pigment particles of largest characteristic size which serve as supports for the paint coating should consist, in part at least, of chemically inert pigments, such as natural crystalline barium sulphate, calcium carbonate, magnesium silicate, etc.
Corollary C—It follows directly that the thickness of a paint coating is determined by the particles of pigments having the largest characteristic size, even if that pigment be present only in moderate percentage. Upon this principle depends the comparatively great thickness of film and moderate spreading rate of paints composed of such pigments as basic carbonate—white lead, red lead, barytes, etc., and the strongly contrasted thinness of film and high spreading rate of paints composed of the sublimated pigments such as lamp black, zinc oxide, basic sulphate—white lead, zinc-lead white, leaded zinc, etc.
In commenting upon the announced laws set forth above, Heckel says: “The recognition of these laws was an exercise of pure deduction. Paint manufacturers before Mr. Perry’s announcement were producing paints containing three or more pigments with particles of varying characteristic sizes; but their procedure was based largely on empirical knowledge, the result of accumulated experience, due to a conscientious endeavor to produce the highest type of paints for economic service. In the absence of any law to govern or to limit the use of the reinforcing pigments, inexperienced manufacturers had brought upon the market paints which were badly proportioned as to the several pigments, or burdened beyond the limits of effectiveness with reinforcing pigments. To all paint manufacturers Perry rendered a substantial service in deducing for them the laws set forth in his address. In the results following a recognition of these laws there was nothing new or startling, but Perry[96] was the first to give the principles from which it can be determined in advance whether a paint formula will prove to be physically good or bad in practice.

Series of Paint Chasers, Mixers, and Grinders

Overhead Churn Mixer

Battery of Paint Mixers and Grinders of Modern Underdriven Type

Photographs courtesy of Ernest Heath
View showing Shrinkage in Bulk of Paint Pigment after being ground in Oil. Filled Barrel on Right with the Oil forms one-third Barrel Paste as shown in Barrel on Left

View showing careful Dressing of Bull Stone Mill from Grinder
“As has been before stated, he was not the first to recognize the law governing minimum voids, but by that scientific use of the imagination which Tyndall so highly commends, he recognized, as by inspiration, the fundamental similarity existing between a film composed of solid particles cemented together by a semi-solid homogeneous menstruum and a layer of concrete composed of solid particles cemented together by a solid homogeneous medium. His application of the law permits the paint manufacturers to design a paint formula with full knowledge of the controlling conditions, so that it shall produce a coating neither too thick, and therefore uneconomical and subject to excessive internal strains, nor too thin, and thus weak and inefficient for protection. That Mr. Perry’s contention was well-founded, other paint technologists have since demonstrated; notably Mr. Wirt Tassin, in his microscopic studies of paint films in situ, and Prof. G. W. Thompson who, in his address to the Penna. Association of Master Painters at Reading, said:—“I want to agree with Mr. Perry * * * where he says that a pigment should be made up of particles of different sizes. Mr. Perry also draws a further parallel between paint and concrete where he refers to the form of the reinforcing pigment particles and suggests that in paint coatings as in concrete a field can be[99] found for the chemically inert pigments with rod-like or hair-like structure, to strengthen the film, just as the steel rods and iron mesh are used to reinforce concrete in structural work—a suggestion which, since the first publication of the address, has been widely accepted as a practical aid in the manufacture of good paints.”
Use of Inert Pigments. There seems to be no reasonable doubt as to the efficiency of a small amount of inert pigments in paint, and the writer has often compared the manufacture of paint of the above type to the making of various alloys wherein zinc, copper, and other metals are added to gold in order to make a product possessed of greater durability, etc.

Batteries of Color Grinding Mills
There has been considerable inquiry as to just what is meant by the statement that “a moderate percentage of inert pigments, combined with properly adjusted mixtures of white lead and zinc oxide, have given wonderful service in all the tests.” The writer has been asked to define what “moderate” means. A “moderate percentage of inert pigments” should be defined as that amount of natural crystalline pigments that will, when mixed with white lead and zinc oxide, not materially detract from the hiding power of white lead and zinc oxide. It is possible to mix a certain percentage of these crystalline pigments with white lead and zinc oxide, and, by thorough grinding, incorporate them in such a manner that the mixture will show nearly as good a hiding power as the straight white lead and zinc oxide.[100] When certain limits have been reached, however, and these limits must be determined by the manufacturer and painter in making practical tests, the further addition of inert pigments lowers the hiding power of the paint and therefore lowers the value of the paint. These remarks do not apply to artificial crystalline pigments, such as precipitated whiting, which possess greater hiding values than the natural pigments.
Perry’s Principles of Paint Making. Parts of the original paper[15] in which Perry so clearly set forth the principles from which the preceding laws were formed, follow:
[15] Physical Characteristics of a Paint Coating. R. S. Perry. Michigan Chapter, Amer. Institute of Architects, 1907.
Sealing Quality or Imperviousness of the Coating. “It has been emphasized that for durability and protection, the strength and imperviousness of a paint coating are vital factors. The protective value of the paint coating of course ceases with its chalking or disintegration, but, while it is true that the protecting or final life of the coating ceases with this disintegration, it is also true that a paint coating has always during its true life more or less porosity from the nature of the linoxin or oxidized linseed oil. Therefore during its protecting life the degree of its imperviousness influences its resistance to attack upon its own life and its protection of the underlying materials. The more impervious the paint coating without loss of strength, the slower the oxidation or disintegration of the paint coating itself and the greater protection to the underlying material.
“A coating of linseed oil alone is not only weak, but the simplest and crudest experiments will show its porosity and this porosity increases rapidly with progressive oxidation, the porosity of course definitely hastening the over-oxidation or chalking. In proportion, therefore, to our success in filling the voids in the linseed oil film with proper pigment materials, we will in that degree succeed in excluding agencies of decay, not only from the mass of the paint coating itself, but also from the surface to be protected. These conditions are exactly parallel in the requirements and performance of the best-made concrete, and Taylor & Thompson in their work on concrete have clearly stated that to obtain imperviousness there must be freedom from voids, and that to obtain these conditions, the materials used must have at least three determining sizes.

Equal Volume (One Cubic Centimetre) of Each Size of Shot Taken. Note that the Smaller Shot Cover more than Half as much again as the Larger Shot and the Voids are Smaller.
Diagram Illustrating Two Determining Sizes of Solid Particles in Concrete
Diagram Illustrating Three Determining Sizes of Solid Particles in Concrete
“‘It is a fact that with particles of different sizes as against uniform size the densest mixture can be obtained. This is so evident as to require no proof.’ It follows that the least density and hence the largest percentage of voids occur when the grains are all of the same size, and it is shown that the most voids occur in a mass of large particles. The least voids occur when the voids between the large particles are filled with smaller particles and when these smaller voids between the smaller particles are in turn filled with still finer particles. In other words—particles with three determining sizes will fill up a given space more completely than particles of two determining sizes and very much more completely than particles of one size.
Elasticity and Strength. “The paint coating here again is governed by many of the laws which govern the similar material, i.e., concrete. We find, by again referring to Taylor & Thompson, on Concrete, page 275, that tests at the Watertown Arsenal on concrete convinced the investigators that the ultimate strength of a concrete is identical with the shearing strength of particles of stone making up the aggregate.
“This means that in its ultimate form the good concrete will crack or shear through the broken rock contained therein, and resistance to shearing is directly proportionate to the strength of the broken rock chosen for the mixture. The film of semi-liquid linseed oil when fresh is extremely weak, but as it hardens, its characteristics and physical properties will obviously be those qualities which are a composite of the qualities of the solid particles and of the semi-solid linolein incorporated together in the paint coating. These physical properties of the suspended and incorporated pigments profoundly modify the film in this respect.
“The dried vehicle, linoxin, is notable for its elasticity, and it is weak in crushing and tensile strength, and in hardness or resistance to surface wear. The fact that it is a semi-solid furnishes an opportunity to modify and improve those characteristics of a solid in which it is deficient. The semi-solid, rubber-like linoxin between the coarser particles of the pigment obviously uses these coarser particles as supporting points. The medium sized particles of the second group of alteration products serve the same purpose as the broken rock in concrete. The coarser particles absolutely do not, and can not, serve the purpose of stiffening or of reinforcing or modifying the consistency and[103] qualities of the semi-solid linoxin, for a number of reasons, one of which may be mentioned, namely, that particles of the first, or coarse, class have a determining size which is a large fraction—a heavy percentage—of the total thickness of coating, and are in some instances thicker in diameter than the thickness of an oil coating not reinforced with the fine or fire group.
“We must think of the coarser particles as piers. The mixture of linoxin with the other two groups of particles in the spaces between these coarser particles, or piers, is the true paint body and consists of flat reinforced arches which have the extra support of falsework, in the shape of the structural material on which the coating rests. Asbestine pulp, a natural product and one of our most important natural reinforcing pigments, serves not only in the coarse group as supporting particles for the linoxin arch, but also because of its peculiar properties serves the more important purposes of reinforcement. It retains, no matter how finely ground, its peculiar needle-like, or rod-like, form of particles, and obviously serves the purpose of reinforcing the flat arch of linoxin, exactly as iron bars or iron netting serve in reinforced concrete arches. The medium sized particles of the second group of pigments produced by chemical alteration or precipitation, serve the purpose of the broken rock in concrete, and together with the coarser supporting particles and the finest reinforcing particles, give minimum voids and a maximum imperviousness to agencies of internal decay.
“It goes without saying that the pigments of any one group contain particles of dimensions which fall into the other two groups, but no one pigment supplies the correct proportion of each of the three required dimensions, and each pigment has so large a percentage of approximate dimensions as to bar it from exclusive use in the other two groups. Given similar homogeneous coatings under identical conditions, we recognize the law that elasticity will vary directly with thickness. Direct deduction from this law teaches us that of two paint coatings equal in wear, in strength, opaqueness, and in all other qualities except thickness, we should choose the thinner coating. Therefore if we have two paint coatings fulfilling every requirement, the first compounded with pigments giving a thicker coating and the second with pigments yielding a thinner coating, we must choose the second formula and obtain the thinner coating.
[104]Adhesive Power. “The adhesion of the linoxin to the coarse group of particles and to the underlying material is vital to the life of the paint coating. If the coating parts from the surface beneath, we have scaling or peeling. It is universally admitted that this will result from use of zinc oxide as the sole pigment. We have only to conceive of our flat arch of reinforced linoxin and leave out our points of support, to realize that this is the inevitable result if the coating be subject to extreme exposure, although good results may be obtained from zinc oxide used alone, as, for instance, in interior house painting where extreme changes of temperature and exposure are avoided.
“Three major lines of force hold our linoxin in place—adhesion toward the underneath surface, adhesion to the coarse particles, and cohesion within the linoxin itself. These lines must be represented by a flat arch of linoxin with a downward pointing magnet therefrom, to represent adhesion to the surface. Magnets on each side of the arch pointing toward the supporting coarse particles, and two magnets within the arch and pointing toward each other, or to the centre of the arch, these latter to represent the force of cohesion.”
The Pigment Contention. During the year 1906 officials of the North Dakota Agricultural Experiment Station examined a number of paints on sale in the northwestern States. The presence of large quantities of inert pigments as well as water, in some of these paints, prompted agitation for State laws requiring the formula-labeling of paints. Certain paints made of white opaque pigments such as white lead and zinc oxide were exempted from the statute. The white opaque pigments used in these paints were believed by certain manufacturers as well as by many prominent paint authorities of high standing to be benefited in their wearing value by the addition of small percentages of inert crystalline pigments, such as barytes, silica, China clay, etc. Laboratory experiments had already determined that these inert crystalline pigments had a certain definite action in increasing the life of paints, but it had become evident that they should be used with discretion, in moderation, and with a proper understanding of their limitations, if the best results were to be obtained. The addition of very large quantities of such pigments was not indulged in by discriminating manufacturers, but the exact percentage to use was a matter of great doubt, even to the most experienced. In order to determine just what percentage of crystalline pigments, admixed with white opaque paint pigments, would give the best service and results, it seemed imperative that practical paint tests should be made. A series of paint tests on commercial brands of paint had already been started at the Fargo Agricultural College, and, at the suggestion of the Paint Manufacturers’ Association of the United States, another series of practical paint tests were instituted, and carried out under the supervision of Dr. E. F. Ladd, Director of the North Dakota Experiment Station.
Test Fences to Solve the Problem. It was apparent that the pigment question could be solved only through field tests[106] made on a comprehensive basis and placed under the control of scientific and technical societies of renown, so that they might be fair and unbiased from every standpoint. In order to secure a comparison of the wearing of different paint formulas in various sections of the country and under differing climatic conditions, another series of tests was started in the East soon after the North Dakota tests had been started. Simultaneously fences were erected at Atlantic City, N. J., and Pittsburg, Pa. The site of the Atlantic City fence is a strip of land running due north from Atlantic and Savannah Avenues and within a short distance from the Atlantic Ocean, the exposure being a severe one. The site of the Pittsburg fence is back of the athletic field of the Carnegie Technical Schools, the fence running east and west and being exposed to the heavily charged sooty atmosphere coming from the many industrial plants near by.
Construction of Framework of Fences. At these two locations framework fences were built, upon which were placed a series of painted panels. Heavy yellow pine posts six inches square were set in the ground about six feet apart and to the depth of about four feet, upon a concrete base. The posts were solidly tamped and then braced at the top with supplementary studding braces two inches thick. Connecting the posts was a line of studding six inches by two inches, forming a solid framework, the bottom of which was approximately fifteen inches from the ground. The bottoms and tops of the fences were protected by heavy boards two inches thick, so that the moisture and rain might be prevented from working itself up into the wood. The whole fence was sheathed with twelve-inch planed white pine, thus forming a solid background for the test panels.
Lumber for Panels. The lumber for the test panels was most carefully selected, being of three grades—white pine, yellow pine, and cypress. A large amount of each grade of lumber was secured, and after the best portion had been made up into panels, the panels were inspected by an expert lumber classer; nearly 40% being rejected on account of the presence of knots or sappy places which appeared upon the surface. Each of the panels finally passed upon as suitable for the test was branded with a hot iron with consecutive numbers running from 1 to 186. The grade of wood used for each panel was indicated by an abbreviated mark—W for white pine, C for[107] cypress, and Y for yellow pine. In order that a record of each panel might be kept on file, previous to the application of paint to the panels, a complete series of photographs was taken of the panels in sets of four. This work seemed advisable so that the future failure of paint on any one panel, which might be thought due to faulty wood, could be either verified or refuted by a reference to the series of photographs made of the bare panels.

View of Atlantic City Test Fence
Construction of Panels. The panels were constructed of Dutch weather boarding, tongued and grooved together in strips of three pieces and capped at the top with a weather strip, forming a finished surface three feet long and fifteen and a half inches high. They were firmly braced together at their backs and nailed in such a manner that no portion of the nails would appear on the surface of the panel, thus preventing the staining of the panel from rust. The construction of the framework of the fences at Atlantic City and Pittsburg was of such a nature that they would each accommodate 560 panels of this type.
Starting of Tests. On account of the lateness of the season, it was found necessary to do the painting of the tests within a building, so that each formula might be subjected to fair and equal conditions of application, thus excluding the blowing of dust or rain upon the painted surfaces, which would have taken place had the panels been painted upon the fence. The painting of the panels began in January, 1908, the temperature within the buildings in which the work was done averaging 50 degrees Fahrenheit throughout the work.
It was decided to test each formula in three colors, in duplicate, and on each grade of wood, exposing the duplicates on either[108] side of the fence. Thus for one paint formula there were required 18 panels, or 6 painted in each color and on 3 grades of wood.
Paints for Tests. The mixed paints received for the tests were in quart cans, having been especially prepared from the formulas submitted to manufacturers by the technical committee in charge of the work. They were properly labeled with their number and color, in each case. The formulas decided upon for the test are described later. The various white leads and other single pigment paints which were used were received in kegs weighing 121⁄2 pounds each, having been bought in the open market and then given a formula number. The formulas of the paints designed for both the Atlantic City and Pittsburg tests, as well as the numbers of the panels upon which the paints were applied, are shown on pages 131-133-145. The analysis of one of the combination paints applied is herewith given, to show the correct method of stating the composition of a paint.
| Percentage Composition | |||
| Pigment | Vehicle | Total | |
| Corroded white lead | 67.01 | — | 42.84 |
| Zinc oxide | 19.89 | — | 12.71 |
| Asbestine | 3.86 | — | 2.47 |
| Calcium carbonate | 9.24 | — | 5.91 |
| Raw oil | — | 94.30 | 34.02 |
| Japan drier | — | 3.89 | 1.40 |
| Turpentine | — | 1.81 | 0.65 |
| 100.00 | 100.00 | 100.00 | |
Brushes. Heavy 7-O round bristle brushes were used for the priming coat so that the paint might be well worked into the wood, while for the second and third coats three-inch chisel edge brushes were used. These brushes were, of course, washed several times with turpentine after painting each panel, so that pigments from one paint could not be carried over into a paint containing other pigments.

Cypress Panels
Shellacking Panels. The shellacking of any bad places of minor nature which may have been present on the surfaces of some of the panels, was done with the highest grade orange shellac. It was thought advisable to determine whether shellacking over the priming coat of paint or on the bare wood[110] previous to the application of the priming coat, was the better method. Panels Nos. 1 to 8 in each test were therefore shellacked over the priming coat, while on all other panels the shellacking was done directly on the bare wood previous to the application of the priming coat of paint.
Application of Paints. In order to determine just how much paint was applied to each panel and to reckon the spreading rate therefrom, careful weighings were made during the application of every paint. This was carried out by placing a quart can of paint as received, upon a laboratory balance, the gross weight being taken and recorded. The can was shaken and its contents transferred to a quart-size enameled cup where with the aid of a paddle it was broken up into a mixture of even consistency. A portion of this paint was then transferred to two small sample cans carefully numbered with the formula number, for future reference and analysis. The reduction of the paint was then made. The brush used on the priming coat was placed with the pot and the paint on the balance and the weight taken by the official weigher. The pot was then given to the painter who applied the priming coat to one panel. The brush, pot, and paint were then handed back to the official weigher and the difference in weight recorded. From these data could be reckoned the spreading rate of the formula applied.
The drying of the panels was noted every few hours and observations made to determine whether the paints were penetrating properly into the surface of the wood. A period of eight days was allowed between each coat in order that thoroughly hard setting might take place.
During the application of the second coat of paint to the panels, fresh cans of paint were used in every case so that fresh reductions could be made of the proper consistency. Full data were also recorded on the ease of application, working, and nature of drying shown, as well as appearance presented by each paint after each coat had been applied. New packages of paint were also used for the third coat, and, as a rule, the paint was applied without reduction or with full oil reduction, turpentine being eliminated in nearly every case for the third coat work.
Reductions. The single pigment paints, such as white leads, were reduced by the so-called ounce system, each ounce of oil added to 121⁄2 ounces of paste pigment representing one gallon[111] of vehicle to one hundred pounds of lead. A complete report of the reductions, spreading rates, etc., used in the tests would take up three or four hundred pages of printed matter. The reductions shown on the following formulas are, however, fairly representative of the reductions used on the combination and single pigment paints.
White and Yellow
1st Coat
Condition when opened—good.
Consistency when broken up—heavy.
Reduction recommended by manufacturer—none.
Reduction used—3 pints raw oil, 1 pint turps, 1 gallon paint.
Consistency after reducing—good, stiff.
Working—fair.
Drying—fair on pines; cypress—poor.
Penetration, pines—good; cypress—poor.
2nd Coat
Consistency when broken up—heavy.
Reduction used—11⁄2 pints turpentine, 1 pint boiled oil.
Consistency after reducing—good.
Working—good.
Hiding—medium.
Drying on pines—good; cypress—poor. One-half pint japan added to gallon of paint.
Penetration—fair.
3rd Coat
Reduction used—11⁄2 pints oil, 1⁄2 pint turpentine.
Reductions for Lead Pastes
Calculated on 100 lb. keg.
Formulas Nos. 37-38. (Corroded White Lead.)
1st Coat
61⁄2 gallons oil, 1⁄2 gallon turpentine, 1 pint turpentine japan.
2nd Coat
31⁄2 gallons oil, 1 gallon turpentine, 1 pint japan.
3rd Coat
3 gallons oil, 1 pint turpentine, 1⁄2 pint japan.
Hiding Power of Paints. When the priming coat had thoroughly dried on each panel, the painter carefully stencilled a black Geneva cross over the priming coat with lampblack in oil. The object of this black cross was to make a determination of[112] the comparative opacity or hiding power of the different paints applied. It is well known that various pigments when ground in oil differ in their hiding power in direct proportion to the difference in the refractive indices of the pigments and oils used, those containing high percentages of pigments such as white lead and zinc oxide being superior in hiding power. After the second and third coat of paint had been applied to each panel, there was evident a remarkable difference in the hiding power, as the black cross showed through in some cases quite clearly, while in other cases it was almost completely hidden. The hiding power of a paint is one of the properties which the master painter looks upon as most essential, but it should, of course, be accompanied in a satisfactory paint by good spreading power and longevity.
Actinic Light Tests. After the drying of all the paints, it was decided that it would be of extreme interest to conduct a test on the resistance of certain paints to actinic light. It is well known that the ultraviolet or chemical rays of the sun are most energetic in causing chemical reactions that result in the early decay of certain types of paint. It was thought that the disintegrating effect of these rays, as well as their effect in the bleaching out of colors, might be prevented by placing upon certain panels small orange colored glass slides which would prevent the passing of these rays to the painted surface. The slides used were five inches long and three inches wide and were placed upon the middle board of certain panels, with picture framing, putty, and galvanized iron tacks. The preservation of the underlying surface from the sun’s rays would, it was thought, prevent the deterioration of the paint, and at the same time preserve its original color so that it might be compared to the color of the exposed portion at the time of inspection.
Supervision of Tests. The Atlantic City tests were under the constant supervision of Committee E of the American Society for Testing Materials, this committee having accepted the inspection of the fence. A representative was constantly present throughout the work in order to see that each formula received fair treatment. The actual painting work was under the supervision of the writer, together with a master painter representing George Butler who was chosen by the Master Painters’ Association of Philadelphia as the official painter of[113] the Atlantic City test fence. Mr. J. B. Campbell of Chicago also acted as an official of the Paint Manufacturers’ Association in the application of the formulas to both the Atlantic City and Pittsburg fences.
At Pittsburg the fence was placed directly under the supervision and control of the Carnegie Technical Schools, who chose for the fence work a committee of their technical force. Drs. James and Schaeffer of this institution were present throughout most of the work and were constantly represented during the test. The Pittsburg Master Painters’ Association appointed a committee consisting of Messrs. Dewar, Rapp, and Cluley, for the actual painting work, and they were represented with the writer throughout the tests.
Great interest was exhibited in the work by the committees in charge, and the skill of the practical painters, combined with the care of the inspectors, made the treatment of each formula fair and satisfactory.
Inspection of Atlantic City Tests. During the month of March, just one year after the placing of the painted panels on the Atlantic City fence, an inspection was made jointly by a committee representing the Master Painters’ Association of Pennsylvania, the Scientific Section of the Paint Manufacturers’ Association of the United States, and certain members of sub-Committee E of the American Society for Testing Materials.
Methods Used at Inspection. One of the most important tests made when inspecting paint is the determination of the chalking taking place.[16] There was developed during the inspection of the Atlantic City panels a new method for determining the comparative chalking of the various paints. It was thought desirable to secure a method, if possible, that would show results which might be photographed and even tabulated in percentage form, if desired. The apparatus for the new test consisted of a small strip of black felt three inches wide by five inches long, placed across a small block of wood which would fit in the palm of the inspector’s hand. This outfit resembled a blackboard eraser and was used in a similar way. By holding the apparatus firmly against the panel and drawing it half-way across the panel in a straight line toward the operator, there was obtained on the black cloth a white mark proportional in intensity to the amount of chalking which had taken place on the given area. When a series of these cloths were made, they were assembled and photographed for comparison. It should be noted that the above chalking test is useful only where the painted panels under examination have been exposed over a period of one to two years, during which period the chalking of paints has been shown to be greatest and the chalked surface of a fairly adherent nature. Where longer exposures have been made and where rains have removed from the painted panels a considerable amount of the chalked pigment which has formed, such a test would not be fairly representative of the amount of chalking which had taken place.
[16] Mr. Macgregor of the Picher Lead Co. has just developed a new test to determine the relative imperviousness of paints which have begun to chalk. He draws a mark about two inches long upon the painted surface with a fountain pen. The ink mark will spread rapidly to a wide area if the chalking is of a bad order. If the chalking is slight and the film in good condition, the ink mark will not spread.

Series of Black Felt Cloths used in making the Chalk Tests on the Various Formulas. Numbers over Cloths represent Panels

CHALKING.—Type of Decay Exhibited by Improperly Made Paint (magnified view)

CHECKING.—Type of Decay Exhibited by Improperly Made Paint (magnified view)
BLISTERING.—Type of Decay Exhibited by Improperly Made Paint (magnified view)
CRACKING.—Type of Decay Exhibited by Improperly Made Paint (magnified view)

GENERAL DISINTEGRATION.—Type of Decay Exhibited by Improperly Made Paint (magnified view)

SCALING.—Type of Decay Exhibited by Improperly Made Paint (magnified view)
Gloss. The gloss of the various panels was a condition which was also reported upon, the middle board of each panel being washed with a wet sponge one day before the inspection so that any surface dirt might be removed. By looking at a panel from the side, a day after the washing, the inspector was enabled to get a fair idea of the degree of gloss exhibited by each formula.
Hiding Power. The hiding power of each paint was determined, as before described, by observing the degree to which the stencilled lampblack cross on the priming coat was visible through the second and third coats. Single pigment paints such as white lead possessed very great hiding power and obscured the black cross almost completely, while the cross was quite visible through paints containing high percentages of crystalline pigments.
Checking. The checking of each panel was determined by examining with a small high-power hand glass magnifying fifteen diameters. It is well known that examinations with such a hand glass will not determine whether so-called fine matt checking is taking place, but it will determine whether checking has appeared to any marked extent. Fine matt checking is the first sign of the decomposition of a paint, and is preliminary to the visible checking seen by the naked eye, which is often followed by alligatoring. Examination of some formulas disclosed this so-called alligatoring and even the exposed wood between the fissured surface which had developed from what were at first fine hair checks. It is, in the opinion of the writer, possible to predict with a fair degree of accuracy by examination of a painted surface, one year after exposure, how the paint will wear in the future and what its appearance will be at the end of another year.
Hardness. The hardness of each panel could not be determined with any degree of accuracy, but the inspectors were able to roughly determine this condition by very close inspection.[123] From practical experience of the wearing of white lead and zinc oxide, and the comparative hardness of these two pigments, zinc oxide was selected as the maximum for hardness and termed number 10, while white lead was selected as the minimum and termed number 1. The varying degrees of hardness exhibited by the formulas were recorded in terms from one to ten. This comparison of course was only an approximate one.
General Condition. The so-called general conditions of the panels was, as a rule, the consensus of the judgment held by the various inspectors, with due regard to such properties as chalking, checking, gloss, hiding power, color maintenance, condition of surface, etc.
Results on Various Woods. On the Atlantic City Fence all the tests made on yellow pine and cypress were found to be in an unsatisfactory condition for a report, for in every case the sap and small knots contained in such wood had a very bad effect upon the paint, causing peeling and scaling. The white pine panels were in very much better condition, and it was therefore decided to make the inspection entirely from the white pine panels and in the future to remove the yellow pine and the cypress panels from the fence and from the test. The Committee advised that all future tests be made on white pine, as it is obviously unfair to use anything but the highest grade wood for a paint test in which the desire is to determine the comparative wearing value of pigments.
Note.—Recent tests have shown that Cypress may be successfully painted when the priming coat of paint is thinned with Benzol (Solvent Naphtha).

Panels on Atlantic City Fence
Two Lower Sets of Panels are painted with Lithopone Paints. Rapid Failure shown

Panels on Atlantic City Fence

Panels on Atlantic City Fence
Two Lower Sets of Panels are Painted with Combination Pigment Paints. Excellent Results shown
[128]Paints Containing Lithopone. One of the most striking exhibitions of paint disintegration in the whole test was the failure of nearly all the lithopone formulas tested. At the time these formulas were suggested for the test, various European technical journals had advocated the use of lithopone in large percentage for paints to be used on exterior surfaces. Good results had been obtained in the northwestern section of Europe, with this pigment in certain mixtures, and the object of these lithopone tests at Atlantic City and Pittsburg was to determine whether satisfactory paints could be made of this pigment for exposure in this country. Failure of the tests, however, in nearly every case except where zinc oxide and whiting were mixed with the lithopone, indicated that pigments such as zinc and whiting are necessary in order to prevent the decomposition of lithopone pigment paints. The decay of lithopone paints after they are applied seems to start with rapid oxidation of the linseed oil, and this oxidation seems to continue in a progressive and even accelerated way; after six months’ exposure the surface of the paint being chalked to a great extent and showing rapid decomposition of the binder or vehicle. Inasmuch as lithopone is really an inert pigment, this rapid decomposition of its vehicle cannot be explained in the same way as the decomposition of the vehicle of pure white lead paints, where the alkaline nature of the lead is probably responsible for the formation of easily destroyed compounds. As complete failure had taken place in nearly every case where lithopone had been used, it was decided to condemn the lithopone panels on the fence, consisting of formulas 21 to 27, including panels 151 to 164 in white, panels 131 to 144 in yellow, and 109 to 122 in gray. These lithopone tests were later on replaced by new tests in 1909, which will be reported upon later in this book.
General Results. From these tests, the inspectors reached the unanimous conclusion that a paint made from any mixture of more than one white opaque pigment, either when used alone or in combination with small percentages of inert pigments, is far superior to any one single pigment paint. It was found that the straight white lead paints failed in every case, and this failure was so marked as to make it a conclusive demonstration of the unfitness of white lead along the Atlantic coast, when used without other pigments. Paints made with large percentages of white lead, however, gave excellent results.
Gypsum was found unsafe to use in any large proportion in a paint, because of its solubility and liability to percolate through the coating of linoxyn or dried film, thus destroying the surface of the paint. Whiting, or calcium carbonate, demonstrated that it could be used in moderate percentage with some efficiency, but it was evident that any great excess of this pigment must also be avoided on account of its tendency towards rapid chalking. Magnesium silicate, aluminum silicate, and silica are three inert pigments which proved to be of great value in strengthening and reinforcing paints, especially when they were used in small percentage. In the same way, black fixe and barytes, or barium sulphate, also appeared to be useful in strengthening a paint. As these two last named pigments are chemically the same but physically different, the use of both in a paint formula is considered advantageous, because of the differences in size and form of their particles.
Color Tests. It was the unanimous conclusion of all the[129] inspectors that panels of all formulas which were tinted either gray or yellow were showing far superior wear and less chalking and checking than those which were painted in plain white. The reinforcing action of the tinting materials must be credited for this lengthening of the wear of such paints. Formulas 5, 6, 9, and 16, for instance, in the gray, were in most excellent condition, and in these formulas were used ochre, umber, bone-black, carbon-black, Venetian red and other inert bases. On the yellow panels, formulas 5, 6, 9, and 16 were also in very superior condition, and in these formulas chrome yellow and inert pigments were also used.
Some of the color tests included the priming of boards with white lead, zinc oxide, sublimed white lead, lithopone, and other single pigment paints. Over these priming coats was placed a high grade brilliant paranitraniline red. Fairly good results were obtained in every case, but especially when lithopone or zinc oxide was used as a priming base. These pigments seemed to have no effect upon the constitution of the para red.
Prussian blue, a colored pigment largely used, but one liable to react with certain paint pigments, was admixed with various paints applied to certain panels. This color was found in some cases to have faded materially, especially when mixed with alkaline pigments such as white lead. Sublimed white lead and zinc oxide, which are more inert in nature, did not have such action on Prussian blue, and the tinted bases of these pigments stood up in a remarkable manner. The greens which were tested were all in very good condition, with absence of fading, and showing only slight mildew.
Condensed Results of Inspection. The results of inspection as obtained by the fence committee[17] having in charge the inspection of the test, have been condensed into table form, and are presented on pages 130-131.
[17] R. S. Perry, Director Scientific Section, Paint Manufacturers’ Association of the U. S.; George Butler, Official Painter, representing Master House Painters’ & Decorators’ Association, H. A. Gardner, Asst. Director.
Second Annual Inspection of the Atlantic City Test Fence. After the original paints which had been applied to the Atlantic City Fence had been exposed for over two years, another inspection was made by a committee representing the Master Painters’ Association of Philadelphia and the Scientific Section of the[130] Paint Manufacturers’ Association of the United States. A digest of the report of this committee[18] follows:
[18] George Butler, Official Painter Atlantic City Test Fence, representing Philadelphia Master Painters’ Association; Charles Macnichol, Master Painter; Henry A. Gardner, Director Scientific Section, Paint Manufacturers’ Association of the U. S.
“The painted panels were all carefully inspected by the inspectors in the usual manner. With the aid of high-power magnifying glasses, checking was determined. The degree of chalking exhibited by the various paints was ascertained by rubbing a piece of black cloth across the surface of each paint. Close observance was made to determine scaling, peeling, cracking, gloss, color, and the other factors to be considered when examining a painted surface. From these observations it was possible for the inspectors to state whether a panel exhibited general good condition, general fair condition, or general poor condition.
| For- mula No. |
Car- bon- ate Lead |
Zinc Ox- ide |
Sub- limed White Lead |
Zinc Lead White |
INERT PIGMENTS | First Coat |
Sec- ond Coat |
Third Coat |
Aver- age |
Pa- nel No. |
Condition | Hiding Power |
Color | Hard- ness |
Checking | Chalking | Gloss | Remarks | |||||||||||
| Calci- um Car- bon- ate |
Calci- um Sul- phate |
Mag- ne- sium Sili- cate |
Bari- um Sul- phate |
Silica | Blanc Fixe |
||||||||||||||||||||||||
| % | % | % | % | % | % | % | % | % | % | ||||||||||||||||||||
| 1 | 30.0 | 70.0 | 610 | 987 | 664 | 754 | 1 | Good | Good | Excellent | 8 | Very Slight | High | Like rubbed varnish work. | |||||||||||||||
| 2 | 50.0 | 50.0 | 913 | 1066 | 948 | 976 | 3 | Good | Good | Good | 5 | Hard Matt | Moderate | Med. High | |||||||||||||||
| 3 | 20.0 | 50.0 | 20.0 | 10.0 | 912 | 914 | 786 | 871 | 5 | Good | Fair | Good | 4 | Medium | Slight | ||||||||||||||
| 4 | 48.5 | 48.5 | 3.0 | 759 | 939 | 1047 | 915 | 7 | Good | Good | Good | 5 | Very Slight | Med. High | |||||||||||||||
| 5 | 22.0 | 50.0 | 2.0 | 26.0 | 714 | 1000 | 709 | 808 | 9 | Good | Weak | Good | 8 | 1⁄2 | Slight | High | Hard surface. | ||||||||||||
| 6 | 64.0 | 36.0 | 928 | 1189 | 863 | 993 | 11 | Fairly Good | Weak | Good | 8 | Matt | Good | Surface rough. | |||||||||||||||
| 7 | 37.0 | 63.0 | 763 | 972 | 891 | 875 | 13 | Good | Good | Off Color | 7 | Slight | High | ||||||||||||||||
| 8 | 38.0 | 48.0 | 14.0 | 786 | 910 | 767 | 821 | 15 | Good | Good | Good | 8 | 1⁄2 | Slight | High | ||||||||||||||
| 9 | 73.0 | 2.0 | 25.0 | 716 | 1081 | 812 | 870 | 17 | Fair | Poor | Good | 9 | Heavy Matt | Medium | High | Peeling started. | |||||||||||||
| 10 | 44.0 | 46.0 | 5.0 | 5.0 | 861 | 1014 | 862 | 912 | 19 | Good | Fair | Good | 5 | Some | Med. High | ||||||||||||||
| 11 | 50.0 | 50.0 | 822 | 959 | 918 | 900 | 21 | Good | Good | Excellent | 7 | 1⁄2 | Med. Matt | Some | Med. High | Some washing and discoloration. | |||||||||||||
| 12 | 60.0 | 34.0 | 6% Inert Pigments | 862 | 965 | 734 | 854 | 23 | Good | Medium | Good | 4 | Heavy Matt | Bad | Medium | ||||||||||||||
| 13 | 27.0 | 60.0 | 3.0 | 10.0 | 916 | 1031 | 1121 | 1073 | 25 | Good | Good | Good | 4 | Medium | Fair | ||||||||||||||
| 14 | 25.0 | 25.0 | 20.0 | 5.0 | 25.0 | 564 | 806 | 785 | 718 | 27 | Bad | Medium | Good | 5 | Evident | Some | Medium | Dead, spongy, surface. White incrustations. | |||||||||||
| 15 | 20.0 | 40.0 | 30.0 | 10.0 | 935 | 1044 | 1359 | 1113 | 29 | Good | Medium | Good | 8 | 1⁄2 | Coarse Matt | Slight | High | ||||||||||||
| 16 | 33.0 | 33.0 | 34.0 | 799 | 903 | 994 | 899 | 31 | Fair | Fair | Good | 7 | 1⁄2 | Bad | Slight | Good | White incrustations. | ||||||||||||
| 17 | 40.0 | 40.0 | 3.0 | 13.0 | 4.0 | 806 | 1016 | 884 | 902 | 33 | Good | Fair | Good | 4 | Some | Fair | |||||||||||||
| 18 | 75.0 | 25.0 | 788 | 1257 | 973 | 1006 | 145 | Good | Good | Excellent | 3 | Hard Matt | Moderate | Medium | |||||||||||||||
| 19 | 25.0 | 75.0 | 700 | 1183 | 1400 | 1094 | 147 | Good | Good | Excellent | 2 | Hard Matt | Slight | Very Little | |||||||||||||||
| 20 | 67.0 | 19.5 | 10.0 | 3.5 | 776 | 1063 | 877 | 905 | 149 | Good | Good | Good | 5 | Very Little | Medium | ||||||||||||||
| 33 | 15.0 | 30.0 | 25.0 | 30.0 | 512 | 836 | 689 | 679 | 176 | Fair | Good | Rough surface. | |||||||||||||||||
| 34 | 38.9 | 5 | 33.5 | 8 | 4.8 | 1 | 19.4 | 8 | 3.1 | 8 | 523 | 800 | 810 | 711 | 175 | Good | Medium | Good | 4 | Evident | Slight | Egg Shell | |||||||
| 35 | 37.5 | 1 | 25.8 | 7 | 7.8 | 4 | 20.3 | 6 | 8.4 | 2 | 450 | 893 | 724 | 689 | 180 | Good | Good | Good | 4 | Matt | Egg Shell | ||||||||
| 36 | 100.0 | 408 | 711 | 861 | 660 | 181 | Bad | Good | Good | 1 | Very Apparent | Bad | Egg Shell | Same as 177, but checking not so bad. | |||||||||||||||
| 37 | 100.0 | 524 | 1065 | 828 | 806 | 182 | Bad | Good | Good | 1 | Very Apparent | Bad | Egg Shell | Same as 177 but wood shows more plainly. | |||||||||||||||
| 38 | 100.0 | 555 | 888 | 794 | 746 | 177 | Bad | Good | Good | 1 | Bad | Bad | Egg Shell | Cracking and perishing evident. | |||||||||||||||
| 39 | 100.0 | 550 | 941 | 916 | 802 | 178 | Good | Fair | Good | 6 | Some | Good | |||||||||||||||||
| 40 | 100.0 | 643 | 810 | 998 | 817 | 168 | Good | Good | Good | 2 | Considerable | Egg Shell | |||||||||||||||||
| 45 | 100.0 | 850 | 170 | Fair | Fair | Good | 9 | Very Evident | High | ||||||||||||||||||||
| 46 | 61.0 | 39.0 | 783 | 169 | Fair | Good | Good | 9 | Some | Good | |||||||||||||||||||
| 47 | 100.0 | 730 | 172 | Good | Good | 10 | Apparent | Good | Indication of scaling. | ||||||||||||||||||||
[131]“An inspection of the white lead paints on the fence indicated in every instance a rough, chalked, and disintegrated surface that seemed to be well worn, in some cases nearly to the wood. The strongly oxidizing air of the seacoast is probably responsible for the early decay of this pigment.
“It was observed that the combination type of paint showed better hiding power than white lead, over the black crosses placed on the priming coat of each panel, as a hiding power test.

Front of Fence showing Present Rearrangement of Panels
| FORMULAS | REPORT OF INSPECTION | Pan- el Num- ber |
||||||||||||||||||||
| For- mu- la Num- ber |
Basic Car- bon- ate White Lead |
Zinc Ox- ide |
Basic Sul- phate White Lead |
Zin Lead White |
inert pigments | |||||||||||||||||
| Calci- um Car- bon- ate |
Calci- um Sul- phate |
Mag- ne- sium Sili- cate |
Bari- um Sul- phate |
Silica | Blanc Fixe |
|||||||||||||||||
| CHALKING | CHECKING | GENERAL CONDITION |
REMARKS | |||||||||||||||||||
| % | % | % | % | % | % | % | % | % | % | |||||||||||||
| 1 | 30 | 70 | — | — | — | — | — | — | — | — | Very slight | Very slight | Good | 1 | ||||||||
| 2 | 50 | 50 | — | — | — | — | — | — | — | — | Medium | Slight | Very good | 3 | ||||||||
| 3 | 20 | 50 | 20 | — | 10 | — | — | — | — | — | Medium | Slight | Good | 5 | ||||||||
| 4 | 48 | .5 | 48 | .5 | — | — | 3 | .0 | — | — | — | — | — | Very slight | Slight | Good | 7 | |||||
| 5 | 22 | 50 | — | — | 2 | — | 26 | — | — | — | Slight | Slight | Good | 9 | ||||||||
| 6 | — | 64 | — | — | — | — | — | 36 | — | — | Very slight | Slight | Good | 11 | ||||||||
| 7 | 37 | 63 | — | — | — | — | — | — | — | — | Medium | Slight | Good | 13 | ||||||||
| 8 | 38 | 48 | — | — | — | — | — | — | 14 | — | Slight | Very slight | Good | 15 | ||||||||
| 9 | — | 73 | — | — | 2 | — | — | — | 25 | — | Very bad | Deep, with scaling | Poor | 17 | ||||||||
| 10 | 44 | 46 | — | — | 5 | — | 5 | — | — | — | Heavy | Deep | Medium | 19 | ||||||||
| 11 | 50 | 50 | — | — | — | — | — | — | — | — | Medium | Medium | Fair | 21 | ||||||||
| 12 | 60 | 34 | — | — | — | 6% Inert Pigment | — | Medium | Deep | Fair | 23 | |||||||||||
| 13 | — | 27 | 60 | — | 3 | — | 10 | — | — | — | Medium | Slight | Very good | 25 | ||||||||
| 14 | 25 | 25 | 20 | — | 5 | 25 | — | — | — | — | Medium | Lateral | Fair | 27 | ||||||||
| 15 | 20 | 40 | — | 30 | 10 | — | — | — | — | — | Slight | Visible with naked eye | Poor | 29 | ||||||||
| 16 | 33 | 33 | — | — | — | — | — | 34 | — | — | Slight | Slight | Good | 31 | ||||||||
| 17 | 40 | 40 | — | — | — | — | 3 | 13 | — | 4 | Medium | Slight | Good | 33 | ||||||||
| 18 | 75 | 25 | — | — | — | — | — | — | — | — | Medium | Slight | Very good | 145 | ||||||||
| 19 | — | 25 | 75 | — | — | — | — | — | — | — | Considerable | Deep | Good | 147 | ||||||||
| 20 | 67 | .0 | 19 | .5 | — | — | 10 | .0 | — | 3 | .5 | — | — | — | Medium | Slight | Good | 149 | ||||
| 33 | 15 | 30 | 25 | — | — | — | — | — | 30 | — | Medium | Slight | Very good | 176 | ||||||||
| 34 | 38 | .95 | 33 | .58 | 4 | .81 | — | 19 | .48 | — | — | 1 | .59 | 1 | .59 | — | Slight | Slight lateral | Good | 175 | ||
| 35 | 37 | .51 | 25 | .87 | 7 | .84 | — | 20 | .36 | — | — | 4 | .21 | 4 | .21 | — | Slight | Lateral | Good | 180 | ||
| 36 | 100 | — | — | — | — | — | — | — | — | — | Considerable | Heavy | Fair | Rough surface | 181 | |||||||
| 37 | 100 | — | — | — | — | — | — | — | — | — | Considerable | Heavy and deep | Poor | Rough surface | 182 | |||||||
| 38 | 100 | — | — | — | — | — | — | — | — | — | More than Panel no. 182 | Very deep | Poor | 177 | ||||||||
| 39 | — | — | — | 100 | — | — | — | — | — | — | Considerable | Very slight | Good | 178 | ||||||||
| 40 | — | — | 100 | — | — | — | — | — | — | — | Heavy | Slight | Good | 168 | ||||||||
| 45 | — | 90 | — | — | 10 | — | — | — | — | — | Slight | Slight | Good | 170 | ||||||||
| 46 | — | 61 | — | — | — | — | — | 39 | — | — | Slight | Medium | Fair | 169 | ||||||||
| 47 | — | 100 | — | — | — | — | — | — | — | — | None | Very deep | Poor | 172 | ||||||||
[134]“There are no pigments possessing greater hiding properties when first used than white leads, but the lack of hiding power on the white lead panels after two years’ exposure was caused by the chalking away of the lead. The superior hiding power of the composite paints was due to the action of the other pigments in these combination paints in preventing the lead from chalking away.
“The Committee finds that the addition of a reasonable percentage of zinc oxide to white lead increases its durability and retards its chalking, renders it whiter, and forms a surface that presents a much better repainting condition. The combinations of white lead and zinc oxide on the Atlantic City Test Fence were in general good condition throughout.
“Corroded white lead, sublimed white lead, zinc oxide, and zinc lead are the standard white opaque pigments. They were all tested on the Atlantic City Fence and it was found that to use any one alone results in inferior protection to the wood. Barium sulphate, silica, asbestine, china clay, and calcium carbonate are the standard crystalline pigments. In the past, the overloading of paints with these crystalline or inert pigments has been the cause of the prejudice that painters have had against their use. It has been established beyond controversy, however, that the use of these pigments, in moderate percentage, combined with any of the standard opaque white pigments, such as white leads, zinc oxide, etc., undoubtedly results in better service from every standpoint and forms the most satisfactory white paint for general outside use. Some of the most perfect painted surfaces on the fence were those made on the above basis as reference to the charted report will show.”
The First Annual Inspection of the Pittsburg Test Fence took place during May, 1909, a little over one year after the painted panels had been placed in position. The inspectors found that in Pittsburg a heavy deposit of soot had formed on the panels, and they considered it therefore inadvisable to make a detailed report of the inspection until the second year of the exposure. The general results of the Pittsburg inspection as reported by the three committees[19] having supervision over the work, is, however, given herewith.
[19]
J. H. James, Chairman Test Fence Committee, Carnegie Technical
Schools.
A. C. Rapp, Chairman Fence Committee, Pittsburg Branch Pennsylvania
State Association of Master Painters.
R. S. Perry, Director Scientific Section, Paint Manufacturers’ Association
of the U. S.; H. A. Gardner, Asst. Director.

Pittsburg Test Fence
During the inspection of the Pittsburg tests it was decided to condemn the lithopone panels on the fence, which consisted of formulas 21 to 27, including panels 151 to 164 in white, 131 to 144 in yellow, 109 to 122 in gray. Almost complete failure had taken place in every case where lithopone had been used. These lithopone tests were later on replaced by new tests which are described later in this book.
“Wood Most Valuable for Test. As on the Atlantic City Fence, the white pine panels afforded the best results and gives the best indication of the comparative wearing of the paints and affords no unfair condition, such as other woods might offer, to interfere with the test.
[136]“Condition of Cypress. Cypress showed inferior conditions, except that it was more pronounced and more discoloration of the panels was noticed on this grade of wood, which seems to be extremely greasy in nature and difficult to properly prime, even when the paint used upon this wood contains a large percentage of volatile diluent.
“Removal of Lithopone Panels. The Joint Committees confirmed the previous recommendation to remove all the lithopone formulas, and they decided to remove the cypress and the yellow pine panels in every formula except in the white paints.
“It was decided to reassemble all the white pine panels and group them together for purposes of comparison, and in place of the panels condemned and removed, to substitute a series of new formulas, to further widen the scope of the tests.
“Ultimate Value of Mixed Paints. The results of the inspection conclusively show that a mixture of more than one prime white pigment, whether this mixture be alone or in combination with a small percentage of inert pigment, produces a paint far superior to a paint manufactured from one pigment alone.
“As a general statement of the comparative wearing of the paints, it might be said that the composite formulas are less advanced toward destruction than the paints made from single pigments such as lithopones, white leads and zinc oxides. It is not to be understood from this statement that it is the opinion of the committee that all of the composite formulas are of equal value or that all of them are to be recommended, but it is meant that the higher types, as evidenced by the appearance of the panels, are in the above relation to the single pigment paints.

Panels on Pittsburg Test Fence
[138]“Lithopone Destroyed Rapidly at Pittsburg. It was evident some time ago that the formulas containing large percentages of lithopone were rapidly failing, and their appearance was very much the same as those formulas of a similar type at Atlantic City. There seems, however, to be some difference in the way these formulas broke down; those on the Pittsburg Fence having shown the quicker destruction, possibly due to the action of the acid gases in the air upon the paint coating. This further confirms the statement that paint compositions containing such heavy percentages of lithopone and intended for outside use must be designed with relation to the particular uses of the product and to the climate in which they are to be used. It will also be necessary to consider more carefully the vehicle of the paints which are to be made of this pigment.
“Possible Value of Excluding Vehicle for Lithopone. It was the belief of the committee that much better paints containing lithopone could be designed by varying the percentages of the materials contained in the formulas, and it was suggested that a less penetrable vehicle, made more on the line of a varnish, and not as easily affected as straight linoxyn, should be experimented with in connection with these lithopone formulas.
“The success of certain European countries in using lithopone as a pigment, even in a very high percentage, may be due to the use of a special vehicle, and, if it is found in future tests that this material, which has been reported as well suited in Northern European climates, may be benefited and made of service by the addition of special oils and special vehicles, then this test would be of great value to the whole paint trade at large.
“Preliminary inspections were made on October 6th and later on December 12th, 1908, and a marked difference was observed at the two inspections in the wearing of the various formulas.
“The lapse of the two months between these inspections gave opportunity during which cold weather caused contraction of the paint film which had been previously subjected to the hot summer sun, and caused marked chalking of the white lead formulas. On October 6th this chalking was just commencing, while in the December inspection it was well advanced, and at the annual inspection, had proceeded to such an extent that the pigment had been washed from the panels representing those paints which had started early chalking.
“Panel 177, representing Zinc Lead, was found to be extremely dark in color throughout the coating and was more on the order of a grayish tint. It resisted all attempts to wash it down to a white surface. The panel, however, in other respects, was in fairly good condition.
“Condition of Corroded White Lead Panels. Panel 174, representing Type B Pure Basic Carbonate-White Lead, was very badly perished and discolored, and an examination of the surface showed very bad checking. Long continued washing[139] with a sponge removed a discolored surface and showed but a rather thin coating. Panel 175, representing Type C Pure Basic Carbonate-White Lead, showed most marked checking and was in very much the same condition as 174 and 176. Panel 176, representing Type A Pure Basic Carbonate-White Lead, was in the same condition as the Type B and C Basic Carbonate-White Leads.
“Condition of Sublimed White Lead. Panel 178, representing Sublimed White Lead (Basic Sulphate-White Lead,) was chalking, and the paint coat was somewhat disintegrated. The chalking present on this formula, however, showed that the disintegration of the paint coat had not taken place for several months after the Basic Carbonate-White Leads. This panel maintained good color, not being acted upon by sulphur gases.
“Blackening of Corroded White Lead. The black and gray formation on all the Basic Carbonate-White Lead panels was probably due to the action of sulphur gases which are present in the district immediate to Pittsburg, and which may cause the formation of black sulphide of lead.
“Possibly a general conclusion from all these panels might be described as a perishing of the paint coating, with the formation of sulphide of lead which to a certain extent protects the coating beneath it, but the perishing has proceeded to such an extent that the unaltered paint coating left is but a slight protection to the wood, being extremely thin.
“The committee resolved that the detailed observations of the panels could not be made and that they would not be justified in making detailed comparisons between the various formulas, giving the gloss, hardness, general condition, checking, etc. Precision in this work at such a time was impossible, and it was decided that a further period would have to elapse before such a detailed comparison could be made between the various blended or composite formulas on the fence.
“Report on Colors. It was resolved that at the next inspection of the Pittsburg Fence, portions of the original samples of the original paints used for the yellows and grays should be on hand, previously painted out on small panels for comparison for the deterioration of the colors on these same panels on the fence.
[140]“An examination of the combination formula grays by the committee led to the general conclusion that those grays which did not contain a very large percentage of white lead were superior in their maintenance of tone and tint and general condition to any of the other grays upon the fence. However, the presence of umber, ochre, and red oxide in some of the grays which showed to the best advantage may account for their permanence of tone. Some of these grays were the so-called warm grays and were much darker in tone and tint than the ordinary drab which is generally applied.
“The straight pure Basic Carbonate-White Lead paints were not painted out in grays or yellow, the test upon this material being only in white.
“On Panels 120 and 126, which represent formulas 6 and 9 respectively, the grays are in most excellent condition, and it will be found, by reference to formulas 6 and 9, that there is an absence of white lead in their composition. These formulas, however, contained a small percentage of umber and ochre. Formulas 5 and 16 contained over 20% White Lead and the gray of these formulas maintained their blue tone very well. These formulas were tinted solely with lampblack.
“An inspection of Panel 138, which represents Formula 15, showed good maintenance of color in the gray, and was in much better condition as regards permanence of color than the other grays containing white lead.
“A study of the yellow panels on the fence led to the unanimous conclusion that a liberal amount of Basic Carbonate-White Lead seemed to have a beneficial result in preserving the bright tone of the chrome yellow in tints so strong as those used on the fence. It was noted that Panel 108, which represents Formula 28, and in which zinc yellow was used, showed great permanence of tone and tint. Unfortunately this zinc chromate was added to a formula containing a large percentage of lithopone, and the destruction of the lithopone to a great extent affected the value of this test.
 |
|
| Whiteness of Sublimed White Lead | Darkness of Corroded White Lead |
| On Pittsburg Test Fence | |
[142]“Maintenance of Para Reds. A study of the paranitraniline or azo reds painted over the various pigments as priming coats demonstrated that the reds on this fence are in better condition than the reds at Atlantic City. As is well known, para red is manufactured by precipitation in an acid solution and is best maintained under acid conditions. The acidity of the Pittsburg atmosphere, caused by the large amount of acid gases which are being poured into the air, day in and day out, and which are constantly condensing on the surface of structures, may account for the better preservation of these reds.
“It was noted that the para reds which were applied to panels prime coated with white lead seemed to be brightening in color and seemed to be gradually working over toward a lightening which may in the future show a pinkish tint.
“Report on Greens. The bronze green is in most excellent condition and shows an absence of the mildew appearance which was observed at Atlantic City.
“The chrome green is standing up exceedingly well, there being practically no change whatsoever in the color since it was exposed.
“Best Base for Blues. An inspection of the blues showed that those which gave the greatest permanence and the least amount of fading were applied in combination with either Sublimed White Lead (Basic Sulphate-White Lead), or zinc oxide, while those blues which were applied in combination with Basic Carbonate-White Lead showed marked failure and were completely bleached out, due, of course, to the alkaline nature of the corroded white lead; Prussian blues being transformed by alkalies to a white compound.
“Superior Value of Composite Formulas. Some of the mixed leads, or so-called graded leads, which are combinations of white leads with other high-grade pigments and containing some inert pigments, were not deteriorated so far as the white lead formulas, and the general conclusion was that they were upward of six months behind the deterioration of the straight white leads, and this was confirmed by the presence of moderate chalking, showing an excellent repainting surface and a better thickness and condition of the paint coating.
“The same conclusions which were reached at Atlantic City, as to the best method of shellacking, obtained also on the Pittsburg Fence, namely, that application of the shellac to the wood previous to the first coat is the better method.
“Analysis of Paints. At the time of the painting of the fence a sample of each paint was placed in small friction top cans, carefully labeled, and sent to the Carnegie Technical Schools’[143] laboratory for analysis. The analyses of these paints were made by members of the Test Fence Committee, representing the schools, and appear in this bulletin. The results obtained conform very closely to the formulas which were applied to the fence, a variance of only one or two per cent. being shown in the amount of the different pigments.”
Second Annual Inspection of Pittsburg Test Fence. The second annual inspection of the Pittsburg Test Fence was made on Thursday, May 7th, 1910. The panels in Pittsburg after having weathered for over two years presented an appearance which allowed the making of a detailed inspection, this having been found impossible during the first annual inspection. The inspection party[20] included those master painters who represented the Pittsburg Master Painters’ Association, who were in charge of the application of the paints in 1907, 1908, and 1909, together with the test fence committee from the faculty of the Carnegie Technical Schools, and representatives of the Scientific Section. A summary of the report issued by this committee follows:
[20] A. C. Rapp, Chairman, Test Fence Committee, Pittsburg Branch, Master Painters’ Association; John Dewar, member Fence Committee, Pittsburg Branch, Pennsylvania State Association of Master Painters; J. H. James, Chairman, Carnegie Technical Schools’ Test Fence Committee; John A. Schaeffer, member Test Fence Committee, Carnegie Technical Schools; Henry A. Gardner, Director Scientific Section, Paint Manufacturers’ Association of the U. S.
“Two of the members of the inspection party have been impressed with the lumber lottery existing in some field tests, which have been conducted, and feel that when the object of a test is to determine the relative value of paints, such tests should be conducted on a standard grade of wood, such as white pine. The use of cypress, pitch pine, and other faulty woods, is often the cause of the failure of a paint, which on good wood would show up well. For this reason, only the white pine panels painted with white paints were considered in the inspection, the yellow pine panels and cypress panels having been thrown out of the test at last year’s inspection.
“Checking, cracking, and alligatoring on the painted surfaces were determined by using a magnifying glass. The degree of chalking existing was decided upon by using small pieces of black[144] felt cloth, rubbing them against the surface of the panel; the degree of whiteness removed upon the cloth being indicative of the amount of chalking taking place. General condition was decided upon after carefully weighing the opinion of each member of the inspection party, as regards the general characteristics shown by each paint, such as checking, chalking, scaling, condition for repainting, hiding power, etc. The results have been charted and presented in this manner:[21]
[21] An endeavor was made to use uniform terms in reporting on each formula. In some cases it was necessary to bring out more forcibly the condition by the insertion of qualifying remarks.

Panel on Left Painted with Single Pigment Paint; Panel on Right Painted with Combination Pigment Paint. Photograph taken after Two Years’ Exposure on Pittsburg Test Fence
“Conclusions Reached from the Test. The primary object of the test made at Pittsburg was to determine whether a combination paint, made of two or more pigments, would be equal or superior to single pigment paints. After one year’s exposure, the combination type of paint proved more durable than the single pigment paints.
“It was early apparent that the combination type of paints, that is, those paints made of more than one pigment, indicated in most cases very excellent wear, with a minimum of blackness and a general good condition of surface.
| FORMULAS | REPORT OF INSPECTION | Pan- el Num- ber |
|||||||||||||||||||||||
| For- mu- la Num- ber |
Basic Car- bon- ate Wh. L’d |
Zinc Ox- ide |
Basic Sul- phate Wh. L’d |
Zinc Lead White |
inert pigments | ||||||||||||||||||||
| Calci- um Car- bon- ate |
Calci- um Sul- phate |
Mag- ne- sium Sili- cate |
Bari- um Sul- phate |
Silica | Blanc Fixe |
||||||||||||||||||||
| CHALKING | CHECKING | GENERAL CONDITION |
REMARKS | ||||||||||||||||||||||
| % | % | % | % | % | % | % | % | % | % | ||||||||||||||||
| 1 | 30 | 70 | — | — | — | — | — | — | — | — | Slight | None | Good | Slight scaling; fairly white surface | 2 | ||||||||||
| 2 | 50 | 50 | — | — | — | — | — | — | — | — | Medium | Very slight | Fair | Panels quite dark and some scaling | 4 | ||||||||||
| 3 | 20 | 50 | 20 | — | 10 | — | — | — | — | — | Considerable | None | Good | Fairly white | 6 | ||||||||||
| 4 | 48 | .5 | 48 | .5 | — | — | 3 | .0 | — | — | — | — | — | Considerable | Lateral and irregular | Fair | White surface | 8 | |||||||
| 5 | 22 | 50 | — | — | 2 | — | 26 | — | — | — | Medium | Very slight | Very good | Extremely white surface | 10 | ||||||||||
| 6 | — | 64 | — | — | — | — | — | 36 | — | — | Very slight | Very bad; rough surface | Poor | Black surface | 12 | ||||||||||
| 7 | 37 | 63 | — | — | — | — | — | — | — | — | Slight | Slight | Good | Medium white surface | 14 | ||||||||||
| 8 | 38 | 48 | — | — | — | — | — | — | 14 | — | Slight | Slight | Good | White surface; slight scaling | 16 | ||||||||||
| 9 | — | 73 | — | — | 2 | — | — | — | 25 | — | None | Deep; peeling in places | Very poor | Film brittle and surface dark | 18 | ||||||||||
| 10 | 44 | 46 | — | — | 5 | — | 5 | — | — | — | Medium | Slight lateral in places | Good | Surface very white | 20 | ||||||||||
| 11 | 50 | 50 | — | — | — | — | — | — | — | — | Considerable | Deep matt checking | Fair | Considerable scaling; formation of black coating shattered off |
22 | ||||||||||
| 12 | 60 | 34 | — | — | — | 6% Inert Pigment | Medium | Slight | Fairly good | Surface white | 24 | ||||||||||||||
| 13 | — | 27 | 60 | — | 3 | — | 10 | — | — | — | Medium | None | Excellent | Very white | 26 | ||||||||||
| 14 | 25 | 25 | 20 | — | 5 | 25 | — | — | — | — | Considerable | Medium | Fair | Panel fairly white | 28 | ||||||||||
| 15 | 20 | 40 | — | 30 | 10 | — | — | — | — | — | Slight | Medium | Good | Surface quite dark | 30 | ||||||||||
| 16 | 33 | 33 | — | — | — | — | — | 34 | — | — | Medium | Very slight | Good | Quite white | 32 | ||||||||||
| 17 | 40 | 40 | — | — | — | — | 3 | 13 | — | 4 | Considerable | Slight, along lateral lines | Fair | Surface fairly white | 34 | ||||||||||
| 18 | 75 | 25 | — | — | — | — | — | — | — | — | Medium | Slight, with some scaling | Good | Surface has become quite dark | 36 | ||||||||||
| 19 | — | 25 | 75 | — | — | — | — | — | — | — | Considerable | None | Excellent | No black coating; surface very white, due to inertness of pigment or progressive chalking |
38 | ||||||||||
| 20 | 67 | .0 | 19 | .5 | — | — | 10 | .0 | — | 3 | .5 | — | — | — | Medium | Medium | Good | 40 | |||||||
| 33 | 15 | 30 | 25 | — | — | — | — | — | 30 | — | Heavy | None | Fair | White surface | 168 | ||||||||||
| 34 | 38 | .95 | 33 | .58 | 4 | .81 | — | 19 | .48 | — | — | 1 | .59 | 1 | .59 | — | Considerable | Very slight | Good | Surface is very white; progressive chalking may have prevented formation of black coating |
172 | ||||
| 35 | 37 | .51 | 25 | .87 | 7 | .84 | — | 20 | .36 | — | — | 4 | .21 | 4 | .21 | — | Bad | None | Good | Very white; no black coating evi- dent |
173 | ||||
| 36 | 100 | — | — | — | — | — | — | — | — | — | Bad | Bad | Fair | Surface is dead black; shattered in places |
174 | ||||||||||
| 37 | 100 | — | — | — | — | — | — | — | — | — | Extremely | Medium | Fair | Very black surface and mottled in places |
175 | ||||||||||
| 38 | 100 | — | — | — | — | — | — | — | — | — | Very bad and quite dusty | Very bad, with scaling | Poor | Black surface is loose and shattered | 176 | ||||||||||
| 39 | — | — | — | 100 | — | — | — | — | — | — | Considerable | Slight | Good | Panel surface quite white | 177 | ||||||||||
| 40 | — | — | 100 | — | — | — | — | — | — | — | Very bad | Slight | Good | Surface very white, possibly due to progressive chalking or inert- ness of pigment |
178 | ||||||||||
| 45 | — | 90 | — | — | 10 | — | — | — | — | — | Slight | Considerable | Fair | White surface | 169 | ||||||||||
| 46 | — | 61 | — | — | — | — | — | — | 39 | — | Slight | Slight | Fair | Considerable scaling present; sur- face fairly white |
170 | ||||||||||
| 47 | — | 100 | — | — | — | — | — | — | — | — | Bad | Bad | Bad | Bad condition throughout | 171 | ||||||||||
 |
|
| Middle white panel is painted with a combination pigment formula | Middle white panel is painted with pure Corroded White Lead |
| Notice Difference in Color after Two Years’ Wear | |
[147]“Recommendation. On account of the peculiar conditions which obtain in and around Pittsburg, as exemplified by these tests, the committee finds, as a result thereof, that the best white paint for general exterior use is made of white lead combined with zinc oxide and a moderate percentage of inert pigments, such as silica, asbestine, or barytes.
“Some Peculiar Conditions Affecting the Tests. The inspectors were most impressed during the inspection by the blackness exhibited to such a high degree by certain panels, and the fair degree of whiteness by others. It is well known that in Pittsburg nearly all paints become darkened by the deposition on their surface of carbon particles emanating from the combustion of soft coal. Certain of the paints, however, presented fairly white surfaces, and it would thus appear that the extreme darkness shown by other paints was due to their composition. Corroded white lead when used alone was uniformly covered by black particles, and the higher the percentage of corroded white lead in a paint the darker was the surface. It was at first thought that this darkness was due to the softness of the white lead pigment or to its roughened surface, in causing adherence of soot particles. Sublimed white lead, however, which is also a soft pigment, chalked even more progressively than corroded white lead, but its surface was not rough, and presented a very white appearance. Scrapings from the different panels are being taken, and after a careful analysis the findings from the investigations will be reported by a member of the Inspection Committee.”
| A. C. Rapp. | Chairman Test Fence Committee, Pittsburg Branch, Master Painters’ Association |
| John Dewar. | Member Fence Committee, Pittsburg Branch, Penna. State Association of Master Painters |
| J. H. James. | Chairman Carnegie Technical Schools’ Fence Committee |
| J. A. Schaeffer. | Instructor in Chemical Practice, Carnegie Technical Schools Pittsburg, Pa. |
| H. A. Gardner. | Director Scientific Section, Paint Mfrs. Asso. of U. S. |
May 31, 1910
| Formula Number |
First Coat (sq. ft.) |
Second Coat (sq. feet) |
Third Coat (sq. ft.) |
Average Spreading Rate (sq. feet) |
Spreading Rate 3-Coat Work (sq. feet) |
| 1 | 759 | 1020 | 768 | 849 | 283 |
| 2 | 694 | 975 | 1229 | 966 | 322 |
| 3 | 743 | 873 | 770 | 795 | 265 |
| 4 | 537 | 987 | 1019 | 848 | 283 |
| 5 | 509 | 896 | 886 | 764 | 255 |
| 6 | 765 | 1045 | 994 | 935 | 312 |
| 7 | 734 | 922 | 996 | 884 | 295 |
| 8 | 565 | 862 | 854 | 760 | 253 |
| 9 | 622 | 926 | 1160 | 903 | 301 |
| 10 | 610 | 1013 | 1070 | 900 | 300 |
| 11 | 651 | 933 | 1010 | 865 | 288 |
| 12 | 675 | 1027 | 623 | 775 | 258 |
| 13 | 663 | 892 | 981 | 845 | 282 |
| 14 | 498 | 785 | 807 | 697 | 232 |
| 15 | 688 | 1000 | 984 | 891 | 297 |
| 16 | 669 | 880 | 860 | 803 | 268 |
| 17 | 635 | 982 | 1077 | 900 | 300 |
| 18 | 636 | 959 | 1031 | 875 | 292 |
| 19 | 626 | 1076 | 1037 | 913 | 304 |
| 20 | 591 | 1015 | 929 | 845 | 282 |
| 21 | 595 | 948 | 910 | 818 | 273 |
| 22 | 617 | 868 | 810 | 765 | 255 |
| 23 | 549 | 1002 | 986 | 846 | 282 |
| 24 | 539 | 918 | 783 | 747 | 249 |
| 25 | 530 | 929 | 850 | 770 | 257 |
| 26 | 532 | 916 | 1011 | 820 | 273 |
| 27 | 520 | 850 | 656 | 675 | 225 |
| 33 | 600 | 1340 | 810 | 917 | 306 |
| 34 | 471 | 743 | 690 | 635 | 212 |
| 35 | 402 | 598 | 645 | 548 | 183 |
| 36 | 398 | 668 | 838 | 635 | 212 |
| 37 | 579 | 653 | 838 | 690 | 230 |
| 38 | 463 | 615 | 704 | 594 | 198 |
| 39 | 474 | 954 | 849 | 759 | 253 |
| 40 | 446 | 815 | 871 | 711 | 237 |
| 45 | 527 | 841 | 916 | 761 | 254 |
| 46 | 605 | 740 | 818 | 721 | 240 |
| 47 | 735 | 961 | 993 | 896 | 299 |
Panel Sections for Laboratory Test. In order to make a laboratory study of the painted panels on the Atlantic City and Pittsburg fences, it was thought advisable to remove small sections from representative areas and transfer them to the laboratory for such work. The fences were visited by the official inspection committees soon after the first annual inspection, and the panels were carefully looked over. Upon each was marked out a representative portion, care being exercised to select areas where previous inspections had not disturbed the surface of the film in any manner. The inspectors then placed the number of the panel upon the areas which had been marked off, as well as their initials. The marked sections were sawed out, wrapped in tissue paper, and then transferred to the laboratory where they were placed upon models of the respective fences from which they had been removed. The illustration shows the model test fences set up together. It is very apparent that the Pittsburg panels are much the darker in color, due to the soot, and in some cases lead sulphide formed upon their surfaces. This difference was undoubtedly due to the atmospheric conditions prevailing where the tests were made. One would be led to suppose that a paint film exposed to an atmosphere such as is found in Pittsburg would show deterioration more rapidly than one exposed in Atlantic City. In all the tests and experiments, however, the Atlantic City panels appeared broken down to a much greater extent; though it is true that the Pittsburg panels had darkened considerably and presented a rather mottled appearance. The deposit of soot on the Pittsburg panel seemed to act as a preservative coating for the film beneath, and prevented marked disintegration.

Sections of Atlantic City and Pittsburg Fences Arranged for Laboratory Examination

Sections of Atlantic City and Pittsburg Fences

Upper set of tests made on Panels from Atlantic City Fence
Lower set of tests made on Panels from Pittsburg Fence
Figures at left indicate Formula Number
Figures at right indicate Degree of Chalking

Color Standard used in Comparison of Panel Section
Chalking Test. Small strips of black felt, about one inch square, were firmly attached to a block of wood, and by a clamp having the same pressure in each case, the wood with its surface of black felt was fixed to the panel. This apparatus, which resembles a blackboard eraser, is firmly drawn across the panel in one direction for a certain definite distance, during which time it gathers all the chalked surface presented by the painted wood. Upon detaching the apparatus from the panel it is observed that the black cloth becomes whitened to an extent proportionate to the chalking that has taken place on the given area.
After each one of the panels had been treated in the same manner by the same operator, the black cloths were assembled on one large board and photographed. A definite standard of chalking was made up, and the operator was enabled to put down opposite the report on each panel the degree of chalking which had taken place, No. 1 representing the least amount and No. 10 the greatest amount of chalking.
Degree of Whiteness Shown by Panels. It was a very simple matter to gauge the whiteness of the various panels, by comparing them with a series of standard boards painted with three coats of white paint. Florence Brand, New Jersey zinc oxide, was used as the standard for whiteness and termed “No. 1.” In making “No. 2” standard, to the zinc oxide was added .01% of lampblack. By adding .02% of lampblack to the zinc, standard “No. 3” was obtained, and so on, increasing the amount of lampblack in each case by .01%. These standards were run up to “No. 30,” and the various panels on the different fences compared with them. The degrees of whiteness are recorded in progressive numbers, No. 1 being the standard for whiteness and No. 30 the darkest. The Atlantic City panels ranged from 3 to 8 in the scale of whiteness, while the Pittsburg panels required the use of the entire range of standards.
Resistance to Abrasion. The apparatus used for determining the abrasion resistance of a paint was made of a glass tube about six feet long, having an internal bore of 7⁄8 inch. This was supported in an upright position over a dish which held the panel under test at an angle of 45 degrees. The abrasive material consisted of No. 00 emery, which was dropped into the tube through a funnel having a bore of 5 mm. When the emery reached the bottom of the long tube it scattered itself so as to strike a surface on the panel about an inch in diameter. The emery was constantly poured in until the paint coating had worn away, showing the bare wood. The weight in pounds of emery powder required to show the disruption of the coating is recorded and reported as the measure of the “abrasion resist.” The panel requiring the greatest weight of emery to cause abrasion is evidently the most resistant to abrasion. Paint is often subjected to serious abrasion, through the blowing of sand, especially at the seashore, and to withstand such action should contain a proportion of pigments especially resistant to abrasion, such as silica, zinc oxide, asbestine, and barytes.

Apparatus for Determining the Abrasion Resistance of Paints
 |
Formula No. 1, A. C. | |
| Formula No. 2, A. C. |  |
|
 |
Formula No. 3, A. C. | |
| Formula No. 4, A. C.[156] |  |
|
 |
Formula No. 5, A. C. | |
| Formula No. 6, A. C. |  |
|
| Note: The author wishes to acknowledge the assistance of Dr. J. A. Schaeffer in the preparation of the photomicrographs herewith shown. | ||
| [157] | ||
 |
Formula No. 7, A. C. | |
| Formula No. 8, A. C. |  |
|
 |
Formula No. 9, A. C. | |
| [158]Formula No. 10, A. C. |  |
|
 |
Formula No. 11, A. C. | |
| Formula No. 12, A. C. |  |
|
[159] |
Formula No. 13, A. C. | |
| Formula No. 14, A. C. |  |
|
 |
Formula No. 15, A. C. | |
| [160]Formula No. 16, A. C. |  |
|
 |
Formula No. 17, A. C. | |
| Formula No. 18, A. C. |  |
|
[161] |
Formula No. 19, A. C. | |
| Formula No. 20, A. C. |  |
|
 |
Formula No. 33, A. C. | |
| [162]Formula No. 34, A. C. |  |
|
 |
Formula No. 35, A. C. | |
| Formula No. 36, A. C. |  |
|
[163] |
Formula No. 37, A. C. | |
| Formula No. 38, A. C. |  |
|
 |
Formula No. 39, A. C. | |
| [164]Formula No. 40, A. C. |  |
|
 |
Formula No. 45, A. C. | |
| Formula No. 46, A. C. |  |
|
[165] |
Formula No. 47, A. C. | |
Making Photomicrographs. The photomicrographs which are herewith shown were made in the following manner: A part of a panel was placed upon the stage of the microscope and held firmly in place with clips. By varying the adjustment and carefully running over the field the condition of the surface was readily given, using the same eye-piece and objective throughout the tests, and obtaining a magnification of thirty-three. Great care was exercised to secure an average field showing the general and typical appearance of every panel. Little difficulty was experienced in so doing, as the laboratory panels gave very representative surfaces of the large panels on the fence. The instrument was then inclined horizontally and the eye-piece was fitted into the camera nose. In the back of the bellows of the camera was placed the ground glass for focusing. To secure[166] illumination the light from an electric arc lamp was reflected from a mirror directly upon the painted surface of the panel, which in turn was reflected through the camera on to the ground glass. The plate-holder was then put in position and six-second exposures were made, afterward developing and printing.
Checking and Cracking. What was termed “fine matt checking” at the First Annual Inspection was not visible at the time to certain members of the Inspection Committee, but it is an established fact that the checking was an existing condition, as the photomicrographs have shown. This checking has a very peculiar characteristic in that the lines are very narrow and hair-like, being somewhat interlaced and peculiarly forked. That this hair matt checking is a preliminary condition which afterwards develops into matt checking and into marked or heavy checking seems to be indicated.
It appears from an examination of the photomicrographs of the paint films that a paint coating closely resembles the surface of the earth, and is subject to the same basic laws that have caused the various geodetic changes in the earth’s crust. Observation of a dried pond or lake bed will disclose types of fissuring and cracking similar to those shown by dried paint coatings in which the oil has been fully oxidized, and especially in the case of paints containing pigments which act upon the oil to produce compounds brittle in nature.
At Atlantic City the panels were all clean and free from dirt, presenting continuous exposure of the films, and thus maintaining conditions for active checking. At Pittsburg, soon after the panels began to chalk, the large amount of dust and black soot in the atmosphere completely covered the panels with a very thick, resistant coating of carbon, which acted as a seal or protector, preventing disintegration to a great extent. This coating was extremely hard to remove, and photomicrographs, before and after removal of this coating by rubbing with a damp cloth, failed to reveal marked checking on any of the formulas except those made of strictly pure basic carbonate-white lead. The checking, even on these, was not as marked as at Atlantic City. It is presumed that after the chalking had taken place and the chalked pigment had been washed from the panels, the gradually increasing coat of carbon and lead sulphide had protected the panels from checking, or possibly the atmosphere of Pittsburg, which in other respects had deteriorated the panels to a greater extent than at Atlantic City, did not have the extreme action in causing checking that the Atlantic City atmosphere seemed to have effected.
 |
 |
| Combination Formula No. 1, Pittsburg | |
| Before Washing | After Washing |
| Mottled surface due to external coating of impurities. | |
| [168] | |
 |
 |
| Formula No. 4, Pittsburg | |
| Before Washing | After Washing |
[169] |
 |
| Formula No. 38, Pittsburg Basic Carbonate—White Lead Panels on Fence |
|
| Before Washing | After Washing |
| Checking evident even through the outer covering of foreign matter. | |
[170] |
 |
| Formula No. 36, Pittsburg Basic Carbonate—White Lead Panels on Fence |
|
| Before Washing | After Washing |
| Peculiar network-like checking appearing through outer coat of impurities. | |
[171] |
 |
| Formula No. 40, Pittsburg | Formula No. 45, Pittsburg |
[172]Results on Combination Pigment Paints. It will be noticed that the checking on most of the combination pigment paints made of lead, zinc, and inert pigments, was moderate, and in many cases of a fine order. It has been observed that the percentage of zinc oxide in a paint is not always a criterion upon which future checking may be judged. Nor could it be said that the checking is dependent upon the percentage of basic carbonate-white lead added to the paint. However, it appears that scientific blending of the various pigments, with regard to their physical properties in oil, such as their strength and elastic limit, develops the greatest resistance to both cracking and checking. Elasticity is vital, but strength must be combined therewith in order to prevent disruptions of the paint coating. Paint films made of certain inert pigments, when tested on the filmometer, were relatively high in strength, but relatively low in elasticity. Such pigments, when used in large percentage, form coatings which are hard and apt to crack. The use, however, of these pigments in moderate percentages seems very beneficial in overcoming the effect of using an excessive percentage of white lead, or of zinc oxide.
Results on White Lead Paints. The maximum checking was observed on the basic carbonate-white lead panels, the size of the checks in some cases being several times larger than those on the other panels.
On some of the basic carbonate-white leads the checking was of a very peculiar nature, consisting of very broad fissures in the paint coating, disclosing the wood surfaces beneath. The type of checking existing was also distinct in its structure, being hexagonal in shape. One of the most marked features shown by the basic carbonate-white lead films was the extreme roughness of their surfaces. This roughness is most likely due to the excessive chalking which had taken place.
Results on Silica and Barytes Paints. The checking of paints very high in silica resolved itself into fine hair-like lines which are generally lateral to each other, and indicate a cracked appearance. The checking of paints containing very high percentages[173] of barytes was also of a distinct nature, being generally forked in appearance and of no definite striation.
Surface Condition of Fume Pigment Paints. The panels painted with basic sulphate-white lead (sublimed white lead) showed complete absence of checking. This was also true of the panels painted with zinc lead. These are both fume products and are extremely fine in their physical size, which may account for this condition. Although zinc oxide is made in a similar manner, it gives a much harder paint coating than either of the afore-mentioned pigments, and presents a surface which develops considerable checking, generally of a medium order. The past theories regarding zinc oxide, in which it has been maintained that zinc oxide gives the maximum checking, are evidently incorrect, as the checking found on the zinc oxide panels was not as marked or deep as the checking on the basic carbonate-white lead panels; in fact, the checking might be more in the line of a cracking, possibly due to the brittle nature of the coating composed of straight zinc. This is especially true of zinc paints containing insufficient oil.
The Importance of the Physical Nature of Pigments. It appears that very fine grinding of materials, chosen for their characteristic fineness, with the absence of any unfavorable physical condition or chemical sensitiveness, are important factors in the making of a paint to resist cracking or checking. The purity of the essential materials, as well as the scientific compounding of these materials, with due regard to the law of minimum voids, are great factors which enhance the qualities of paints, greater, perhaps, than the variation of percentages of the various pigments which go to make up a paint.
A series of new test panels to take the place of those panels which were condemned and subsequently removed from the Atlantic City and Pittsburg fences, were painted and exposed during June, 1909. These new test panels are of white pine, this wood having been selected by the joint inspection committee as offering the best condition for future tests. The method used in painting these panels was the same as in the previous tests, together with the adoption of certain refinements in the reductions, application, etc. Thirty-six formulas were selected with careful regard to the percentage of components, including several paints containing lithopone combined with whiting and zinc oxide,[22] two pigments which gave promise of supporting the lithopone for outside use. Some of these lithopone paints contained special vehicles which it was thought would prevent the destructive action which lithopone seems to have upon linseed oil. In order to obtain a criterion of the value of the new formulas applied, as against the wearing of straight white leads, the original white leads used in the previous tests were included, and other brands were added. Each formula was painted out in white, yellow, and gray, upon panels of white pine wood arranged in sequence upon the fence, and properly identified. The customary opacity test, in the form of a small black square, was stencilled over the priming coat of each panel, as in the former tests. The composition of the vehicle in all the new tests was standard, using pure linseed oil with a small percentage of turpentine drier. The tints used in each formula were secured at the time of application by the use of standard colors, lampblack, and medium chrome yellow, using an approximate amount for each formula.
[22] A brief study of the theory of solutions (See Cushman and Gardner on “Corrosion and Preservation of Iron and Steel”), involving the modes of iron formation, will be invaluable to the student who is inquiring into the cause of the peculiar fogging of lithopone, with the idea in view of correcting this evil by physical or chemical treatment. Inasmuch as our observations thus far have led us to believe that the fogging of lithopone takes place in the presence of moisture, with the contributory and necessary action of chemically active rays from the sun or other source, it is fair to assume that under these conditions the insoluble molecule of zinc sulphide and barium sulphate reverts by intricate molecular disturbance and ionization back to the soluble barium sulphide and zinc sulphate from which the lithopone is formed by metathesis. If this be true, then the acid nature of these soluble salts is no doubt combated and overcome at the moment of formation by the basic nature of zinc oxide and calcium carbonate, which tend to ionize to an alkaline reaction. The value of zinc oxide and calcium carbonate in lithopone paints as detergents of blackness, has been demonstrated at both Atlantic City and Pittsburg.” H. A. G.

Section of Fence Showing New Panels Recently Placed

Appearance of 1909 Tests
An inspection of these new tests was made during June, 1910, and the results of the inspection are shown on pages 178 to 181. The results of the inspection prove that it is unsafe to use lithopone in a paint containing white lead of any type, early darkening and failure being shown in every case where such a combination existed. The formulas in the new test, which were properly balanced and which had a low percentage of lithopone combined with zinc oxide and whiting, presented in some cases very good surfaces. A rough, sandy surface, however, was shown where lithopone was used in any great quantity.
| FORMULAS | REPORT OF INSPECTION | Pa- nel Num- ber |
|||||||||||||||
| For- mu- la Num- ber |
Basic Car- bonate White Lead |
Zinc Oxide |
Basic Sul- phate White Lead |
Preci- pitated White Lead |
Zinc Lead |
Litho- pone |
INERT PIGMENTS | ||||||||||
| Cal- cium Car- bonate |
Silica | Asbes- tine |
China Clay |
Bary- tes |
Blanc Fixe |
||||||||||||
| CHALKING | CHECKING | GENERAL CONDITION |
REMARKS | ||||||||||||||
| % | % | % | % | % | % | % | % | % | % | % | % | ||||||
| 1 | — | — | 45 | — | — | 40 | 15 | — | — | — | — | — | None | None | Rough surface, but fair for repainting |
1 | |
| 2 | — | — | 45 | — | — | 40 | — | 15 | — | — | — | — | None | None | Fair; rough surface and slightly dark |
2 | |
| 3 | — | 45 | — | — | — | 45 | 10 | — | — | — | — | — | Very slight | Very slight | Good; very white surface | 3 | |
| 4 | — | — | 45 | — | — | 45 | 10 | — | — | — | — | — | None | None | Rough surface and slightly dark |
4 | |
| 5 | — | 40 | — | — | — | 40 | 20 | — | — | — | — | — | Very slight | Very slight | Good; very white surface | 5 | |
| 6 | — | — | 45 | — | — | 35 | — | — | 20 | — | — | — | None | None | Rough surface; dark | 6 | |
| 7 | 50 | — | — | — | 36 | — | — | — | 2 | 8 | 4 | — | None | Very slight lateral checking | Good | 7 | |
| 8 | — | — | 50 | — | — | 36 | — | — | 2 | 8 | 4 | — | Heavy | Slight | Excellent; very white | 8 | |
| 9 | — | — | 50 | — | — | 36 | — | — | 2 | — | 12 | — | Heavy | Some | Excellent; very white | 9 | |
| 10 | — | 36 | 50 | — | — | — | — | — | 2 | 8 | 4 | — | None | Slight | Good | 10 | |
| 11 | 28 | 55 | — | — | — | — | — | — | 3 | — | 7 | 7 | None | Slight | Good; slightly dark | 11 | |
| 12 | — | 55 | 28 | — | — | — | — | — | 3 | — | 7 | 7 | None | Slight lateral | Good | 12 | |
| 13 | — | 60 | — | — | — | 30 | 10 | — | — | — | — | — | Very slight | Considerable lateral running along grain of wood |
Fair | 13 | |
| 14 | — | 30 | 30 | — | — | 30 | 10 | — | — | — | — | — | Very slight | Considerable lateral running along grain of wood |
Fair | 14 | |
| 15 | — | — | 60 | — | — | 30 | — | — | 10 | — | — | — | Heavy | Slight lateral checking | Fair | 15 | |
| 16 | — | — | — | — | — | 100 | — | — | — | — | — | — | Heavy | Considerable | Dark color; rough surface | 16 | |
| 17 | — | — | — | — | — | 100 | — | — | — | — | — | — | Considerable | Medium | Better than No. 16; not as rough or dark |
17 | |
| 18 | 33 | 33 | — | — | — | — | — | 17 | — | 17 | — | — | Very slight | None | Good | 18 | |
| 19 | 34 | 33 | — | — | — | — | — | 33 | — | — | — | — | Very slight | Slight | Good | 19 | |
| 20 | 34 | 33 | — | — | — | — | — | — | — | 33 | — | — | Very slight | None | Good | 20 | |
| 21 | [23]100 | — | — | — | — | — | — | — | — | — | — | — | Slight | Slight | Fair; rough surface | 21 | |
| 22 | 100 | — | — | — | — | — | — | — | — | — | — | — | Very slight | Lateral cracking | Fairly good | 22 | |
| 23 | 100 | — | — | — | — | — | — | — | — | — | — | — | Medium | Lateral cracking | Fair | 23 | |
| 24 | — | — | 100 | — | — | — | — | — | — | — | — | — | Slight | Slight cracking | Good for repainting | 24 | |
| 25 | — | — | — | — | 100 | — | — | — | — | — | — | — | Medium | None | Good surface | 25 | |
| 26 | — | — | — | 100 | — | — | — | — | — | — | — | — | Heavy | Slight cracking | Fair; surface rough & dark | 26 | |
| 27 | 100 | — | — | — | — | — | — | — | — | — | — | — | Heavy | Lateral cracking | Fair | 27 | |
| 28 | 100 | — | — | — | — | — | — | — | — | — | — | — | Medium | Considerable | Poor; very rough, dark surface |
28 | |
| 29 | 24 | 45 | 13 | — | — | — | — | — | 18 | — | — | — | Slight | None | Good | 29 | |
| 30 | 45 | — | — | — | — | 40 | 15 | — | — | — | — | — | Heavy | Heavy checking and alligatoring |
Poor | 30 | |
| 31 | 45 | — | — | — | — | 40 | — | 15 | — | — | — | — | None | Alligatoring | Rough surface; dark | 31 | |
| 32 | 45 | — | — | — | — | 35 | — | — | 20 | — | — | — | Slight | Medium | Dark and rough surface | 32 | |
| 33 | 50 | — | — | — | — | 36 | — | — | 2 | — | 12 | — | Considerable | Slight | Poor; dark surface | 33 | |
| 34 | 75 | — | 25 | — | — | — | — | — | — | — | — | — | None | None | Fair; dark surface | 34 | |
| 35 | 50 | — | 50 | — | — | — | — | — | — | — | — | — | None | Slight | Fair; rough surface | 35 | |
| 36 | — | — | — | — | — | — | — | 100 | — | — | — | — | Extremely bad | Medium | Fair | Vehicle disintegrated; spotted in places |
36 |
[23] This pigment on analysis proved to be zinc lead.
| FORMULAS | REPORT OF INSPECTION | Pa- nel Num- ber |
|||||||||||||||
| For- mu- la Num- ber |
Basic Car- bonate White Lead |
Zinc Oxide |
Basic Sul- phate White Lead |
Preci- pitated White Lead |
Zinc Lead |
Litho- pone |
INERT PIGMENT | ||||||||||
| Cal- cium Car- bonate |
Silica | Asbes- tine |
China Clay |
Bary- tes |
Blanc Fixe |
||||||||||||
| CHALKING | CHECKING | GENERAL CONDITION |
REMARKS | ||||||||||||||
| % | % | % | % | % | % | % | % | % | % | % | % | ||||||
| 1 | — | — | 45 | — | — | 40 | 15 | — | — | — | — | — | Considerable | Slight | Fair | Dark in places. Diffused | 1 |
| 2 | — | — | 45 | — | — | 40 | — | 15 | — | — | — | — | Slight | Bad | Fair | Dark in places | 2 |
| 3 | — | 45 | — | — | — | 45 | 10 | — | — | — | — | — | Medium | None | Good | Darkening shown in places | 3 |
| 4 | — | — | 45 | — | — | 45 | 10 | — | — | — | — | — | Considerable | None | Good | Medium dark | 4 |
| 5 | — | 40 | — | — | — | 40 | 20 | — | — | — | — | — | Slight | None | Good | No excessive darkness | 5 |
| 6 | — | — | 45 | — | — | 35 | — | — | 20 | — | — | — | Medium | Slight | Good | Surface fairly white | 6 |
| 7 | 50 | — | — | — | 36 | — | — | — | 2 | 8 | 4 | — | Medium | None | Excellent | Whitest surface of new tests | 7 |
| 8 | — | — | 50 | — | — | 36 | — | — | 2 | 8 | 4 | — | Extremely bad | Slight | Fair | Surface darkening | 8 |
| 9 | — | — | 50 | — | — | 36 | — | — | 2 | — | 12 | — | Extremely bad | Slight | Fair | Not as bad as No. 8 | 9 |
| 10 | — | 36 | 50 | — | — | — | — | — | 2 | 8 | 4 | — | Slight | None | Good | Excellent surface; very white | 10 |
| 11 | 28 | 55 | — | — | — | — | — | — | 3 | — | 7 | 7 | Slight | None | Excellent | Surface fairly white; thin soot | 11 |
| 12 | — | 55 | 28 | — | — | — | — | — | 3 | — | 7 | 7 | Medium | None | Good | Surface white | 12 |
| 13 | — | 60 | — | — | — | 30 | 10 | — | — | — | — | — | Medium | Very bad in spots | Fair | Slight darkening | 13 |
| 14 | — | 30 | 30 | — | — | 30 | 10 | — | — | — | — | — | Heavy | Considerable | Fair | Slight darkening | 14 |
| 15 | — | — | 60 | — | — | 30 | — | — | 10 | — | — | — | Extremely bad | Slight | Fair | Fairly white | 15 |
| 16 | — | — | — | — | — | 100 | — | — | — | — | — | — | Extremely bad | Advanced and deep | Bad | Surface rough with considerable disintegration and much darkness |
16 |
| 17 | — | — | — | — | — | 100 | — | — | — | — | — | — | Not as bad as No. 16 |
Less advanced than No. 16 |
Fair | Not as dark as No. 16; slightly mottled in places; buff color |
17 |
| 18 | 33 | 33 | — | — | — | — | — | 17 | — | 17 | — | — | Very slight | Practically none | Fair | Surface white | 18 |
| 19 | 34 | 33 | — | — | — | — | — | 33 | — | — | — | — | Very slight | None | Good | Surface fairly white | 19 |
| 20 | 34 | 33 | — | — | — | — | — | — | — | 33 | — | — | None | None | Good | Surface fairly white | 20 |
| 21 | 100 | — | — | — | — | — | — | — | — | — | — | — | Slight | Slight | Fair | Surface very rough and dark | 21 |
| 22 | [24]100 | — | — | — | — | — | — | — | — | — | — | — | Medium | Slight | Fair | Surface fairly white | 22 |
| 23 | 100 | — | — | — | — | — | — | — | — | — | — | — | Slight | Bad | Fair | Surface rough and darkest on fence | 23 |
| 24 | — | — | 100 | — | — | — | — | — | — | — | — | — | Bad | None | Good | Surface white | 24 |
| 25 | — | — | — | — | 100 | — | — | — | — | — | — | — | Slight | None | Good | Fairly white surface | 25 |
| 26 | — | — | — | 100 | — | — | — | — | — | — | — | — | Medium | Slight | Fair | Rough and very dark; chalking is disrupting black coating |
26 |
| 27 | 100 | — | — | — | — | — | — | — | — | — | — | — | Medium | Slight | Good | Surface fairly white | 27 |
| 28 | 100 | — | — | — | — | — | — | — | — | — | — | — | Medium | Deep; evident without glass |
Poor | Surface rough and very dark | 28 |
| 29 | 24 | 45 | 13 | — | — | — | — | — | 18 | — | — | — | Slight | Slight | Good | Very white surface | 29 |
| 30 | 45 | — | — | — | — | 40 | 15 | — | — | — | — | — | None | Slight | Fair | Color dark | 30 |
| 31 | 45 | — | — | — | — | 40 | — | 15 | — | — | — | — | Very slight | Advanced | Fair | Color very dark | 31 |
| 32 | 45 | — | — | — | — | 35 | — | — | 20 | — | — | — | Extremely slight | Considerable | Fair | Color very dark; rough surface | 32 |
| 33 | 50 | — | — | — | — | 36 | — | — | 2 | — | 12 | — | Extremely slight | Slight | Fair | Surface dark and rough | 33 |
| 34 | 75 | — | 25 | — | — | — | — | — | — | — | — | — | Slight | Deep | Fair | Surface medium dark | 34 |
| 35 | 50 | — | 50 | — | — | — | — | — | — | — | — | — | Considerable | Slight | Fair | Surface medium dark | 35 |
| 36 | — | — | — | — | — | — | — | 100 | — | — | — | — | Extremely bad | None | Fair | Vehicle disintegrated, leaving very white, chalked surface of pigment |
36 |
[24] This pigment on analysis proved to be zinc lead.
An inspection of the original test fence, erected and painted by the North Dakota Agricultural College, on the grounds of the agricultural Experiment Station at Fargo, was made by the inspection committee[25] representing the Paint Manufacturers’ Association of the United States, on the 19th and 20th of November, 1909. The fence was erected in 1906 and painted with commercial paints, procured in the open market. The east side of the fence was built of soft pine and cedar weather-boarding, such as is almost universally used on houses in that locality, presenting a very good surface for test purposes, while the west side was built largely of flat trimmed boards of hard pitch pine which, unfortunately, contained knots, pitch pockets, and uneven surfaces, causing to a greater or lesser extent cracking, scaling, and bad general results on all paints applied thereto.
[25] Henry A. Gardner, Director Scientific Section, Educational Bureau, Paint Manufacturers’ Association of U. S.; George Butler, Master Painter; Charles Macnichol, Master Painter.
The fences built in 1907 and 1908 at the suggestion of the Paint Manufacturers’ Association, were inspected on the 20th, 21st, and 22nd of November, 1909, and the detailed results of the inspection of all these fences follow in this report. The same general conclusions as to the woods represented in the 1906 fence also apply to the 1907 and 1908 fences, and because of the general bad quality of wood used on the western exposure of all fences, the detailed reports were made only from an examination of the eastern side of the fences, both on cedar and soft pine.
The following general summary of the inspection and its results applies to all the test fences on the grounds of the college and is the unanimous conclusion drawn by the inspectors from this work:
“Non-absorbent woods, difficult to penetrate, such as those on the west side of the fences, would undoubtedly have given much better results had they been painted with paints properly reduced to suit the nature of the wood. This treatment seems to have been overlooked in the North Dakota tests, and the painting of the hard pine boards was done with the same consistency of mixtures and the same reductions as upon soft pine. Scaling of course resulted. One of the chief purposes of the fences, however, was to study the different types of wood, and compliance with this desire resulted in the bad conditions herein noted. It has been shown in many other field tests that adherence of paints to hard wood surfaces can be obtained only by causing the priming coat to become amalgamated with the woody fibre, by the use of a large percentage of volatile diluent turpentine, benzole, asphaltum spirits, etc., to secure penetration. If such treatment is omitted, failure soon results, as was evidenced by the uniformly bad conditions presented by the paints on the hard pine panels.


North Dakota Test Fences
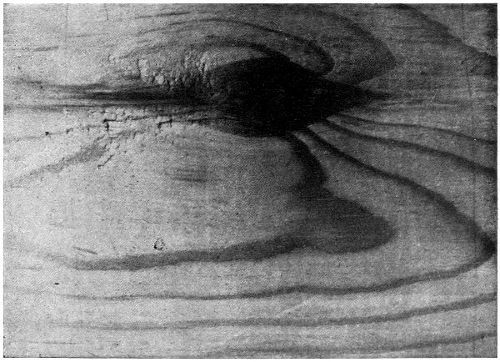
Typical Sample of Hard Pine Trim Board Showing Knot and Sappy Grain

Test No. 13—1906 Fence
Complete Disintegration and Failure of Cheap Paint
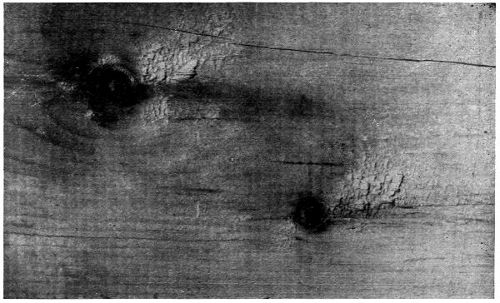
Pine Weatherboarding Showing Knots and Grain

Condition of Lumber Affecting Paint, West Side 1906 Fence

Hail-stone Abrasions on House Repainting Tests

Hail-stone Effect, West Side of 1907 Test Fence
[189]“During July, 1908, a violent hailstorm occurred in Fargo, and left its impression on nearly every wooden structure; in many cases deep dents being made into the wood. The west side of the test fences, which received the most injury from this storm, was covered with these dents over almost its entire surface, causing cracks in the form of concentric rings to appear on the abraded paint coatings. The bad condition of the wood, improper method of applying priming coat, combined with the hailstorm effect on the painted surfaces on the west side of the fences, were undoubtedly responsible for the universal failure of the paints thereon, and, for these reasons, the west side was eliminated from the detailed inspection, only general observations of these tests being made. These general observations, however, showed that paints Nos. 6 and 8 on the 1906 fence, and paints Nos. 8, 10, and 13 on the 1907 fence, proved the most satisfactory on the western exposure.[26]
[26] These formulas were the same as those respectively numbered on the Atlantic City and Pittsburg fences.

Peculiar Crystallization Effect on Section 41. New Special Fence Paint Applied During Cold Weather
“Ochre was tried out as a priming coat on several formulas, but it was found to be most unsatisfactory, affecting the subsequent coats of paint and causing early failure, as evidenced by broad checking, discoloration, and general bad condition. These[190] conditions also apply to those panels on the 1908 fence coated with shellac as a primer.
“The colored formulas in every case showed a great superiority over the same paints in white untinted, and demonstrated that a percentage of color has a wonderful influence on the preservation of the paint coating, reducing chalking, checking, and general disintegration. This condition is probably due to the reinforcing value of the color pigments used.
“It is safe to state that the combination formulas tinted yellow were of better appearance than the corroded white leads tinted yellow, the latter appearing quite dark in many cases.
“The wearing of the paints made solely from white lead and zinc oxide seemed to indicate that a percentage of a third pigment, of an inert nature, would have been beneficial.
“The high-type mixtures of pigments containing lead and zinc, with moderate percentages of inert pigments, on good wood, were in most excellent general condition; in fact, much superior to the single pigment paints. Their surface exhibited only minor checking and moderate chalking with good maintenance of color, and presenting surfaces well adapted to repainting.
“The sublimed white lead was in fair condition, with very little checking, and offering a fair repainting surface. The corroded white lead was somewhat whiter than the sublimed white lead, but a careful observation of the surface of the corroded lead revealed deep checking.
“It was clearly demonstrated, however, that in climates of the North Dakota type, white lead alone is not entirely satisfactory. The addition of zinc oxide to white lead forms paint that has proved much superior to the white lead alone.
“It was conclusively demonstrated that mixtures of white lead and zinc oxide, properly blended with moderate percentages of reinforcing pigments, such as asbestine, barytes, silica and calcium carbonate, are most satisfactory from every standpoint, and are superior to mixtures of prime white pigments not reinforced with inert pigments.
“The white leads painted out on the 1908 fence exhibited different degrees of checking, the mild-process lead and sublimed white lead which presented the best surfaces, being free from checking, while the old-process leads seemed to show very deep and marked checking, even after one year’s wear.
 |
|
| Corroded White Lead | Sublimed White Lead |
| Condition of Two White Leads on Two Grades of Wood | |


Photomicrographic Apparatus and Method of Use
Fargo, N. D., Nov. 19-23, 1909
No gloss shown by any of the paints. Formulas in white on white pine only included here, on east side of fence
| Test No. |
FORMULAS | REPORT OF CONDITION | |||||||||||||||||||||||||||||||||
| Pigment | Vehicle | ||||||||||||||||||||||||||||||||||
| Cor- roded White Lead |
Sub- limed White Lead |
Zinc Oxide |
Cal- cium Car- bonate |
Silica and Sili- cates |
Barium Sul- phate |
Magne- sium Sili- cate |
Clay and Silica |
Bary- tes and Sili- cate |
Lin- seed Oil |
Turp. and Drier |
Japan Drier |
Water | Ben- zine Drier |
Vola- tile Oil |
Chalking | checking | hiding power |
color | condition for repainting |
||||||||||||||||
| % | % | % | % | % | % | % | % | % | % | % | % | % | % | % | |||||||||||||||||||||
| 1 | 100 | — | — | — | — | — | — | — | — | — | — | — | — | — | — | Very bad | Extremely deep | Good | Good | Only fair | |||||||||||||||
| 2 | — | 100 | — | — | — | — | — | — | — | — | — | — | — | — | — | Bad | Very slight | Good | Light yellowish tint |
Fair | |||||||||||||||
| 3 | 50 | — | 50 | — | — | — | — | — | — | 90 | 10 | — | — | — | — | Medium | Fine matt—deep in places |
Good | Fair | Fair to good | |||||||||||||||
| 4 | — | 60 | 40 | — | — | — | — | — | — | 90 | — | 10 | — | — | — | Medium | Surface checking, very slight |
Good | Good | Fair | |||||||||||||||
| 5 | 28 | .7 | — | 71 | .3 | — | — | — | — | — | — | 93 | 7 | — | — | — | — | Slight | Quite deep | Medium | Good | Poor. Coating wrin- kled and hard |
|||||||||||||
| 6 | 40 | .2 | — | 50 | .3 | 4 | .1 | 5 | .4 | — | — | — | — | 90 | .7 | 9 | .3 | — | — | — | — | Medium | Slight surface checking | Good | Good | Good | |||||||||
| 7 | 21 | .9 | 21 | .9 | 45 | .8 | 10 | .4 | — | — | — | — | — | 89 | .6 | 9 | .7 | — | 0 | .7 | — | — | Medium | Surface checking with slight cracking |
Fair | Good | Slight shelling from wood |
||||||||
| 8 | 44 | .1 | — | 46 | .0 | 4 | .6 | — | — | 5 | .3 | — | — | 86 | .0 | 12 | .6 | — | 1 | .4 | — | — | Medium | Very slight | Good | Good | Good | ||||||||
| 9 | In gray only No report. | ||||||||||||||||||||||||||||||||||
| 10 | 13 | .9 | — | 34 | .9 | 26 | .8 | — | — | — | — | 24 | .4 | 72 | .2 | — | — | 24 | .0 | 3 | .8 | — | Slight | Very bad | Bad condition throughout. | ||||||||||
| 11 | 55 | .0 | — | 15 | .2 | — | — | — | — | — | 29 | .8 | Test not finished | ||||||||||||||||||||||
| 12 | — | 5 | .1 | 25 | .0 | — | — | — | — | — | 69 | .9 | — | — | — | — | — | — | Medium | Medium | Deficient | Good | Shelling from wood | ||||||||||||
| 13 | — | — | 31 | .3 | 45 | .4 | — | 22 | .8 | — | 0 | .5 | — | 57 | .2 | — | — | 16 | .1 | 26 | .7 | — | Worst looking surface in North Dakota tests. | ||||||||||||
| 14 | 34 | .8 | 5 | .4 | 59 | .2 | — | — | — | — | — | — | 86 | .0 | 13 | .7 | — | 0 | .3 | — | — | Medium | Slight surface checking and peeling |
Fair | Good | Good | |||||||||
| 15 | — | — | 64 | — | — | 36 | — | — | — | 98 | — | — | — | — | 2 | Slight | Lateral cracking quite deep |
Good | Good | Hard film | |||||||||||||||
Fargo, North Dakota, Nov. 19-23, 1909
| Test No. |
FORMULAS | REPORT OF CONDITION | |||||||||||||||||||||||||||||||||||||||||
| pigment | vehicle | ||||||||||||||||||||||||||||||||||||||||||
| Cor- roded White Lead |
Sub- limed White Lead |
Zinc Oxide |
Cal- cium Car- bonate |
Alu- minum and Magne- sium Sili cate |
Barytes | Silica | Inert | Magne- sium Sili- cate |
Cal- cium Sul- phate |
Zinc Lead |
Lin- seed Oil |
Tur- pen- tine Drier |
Tur- pen- tine and Japan |
Water | Turpen- tine and Ben- zine Japan Drier |
Drier | Vola- tile Oil |
Ben- zine |
Chalking | Checking | Hiding Power |
Color | Condition for Repainting |
||||||||||||||||||||
| 1 | 30 | — | 70 | — | — | — | — | — | — | — | — | 93 | 7 | — | — | — | — | — | — | Medium | Considerable with lateral cracking |
Fair | Fair | Poor surface; too hard | |||||||||||||||||||
| 2 | 50 | — | 50 | — | — | — | — | — | — | — | — | 86 | — | 10 | 4 | — | — | — | — | Medium | Considerable with lateral cracking |
Good | Fair | Rather poor | |||||||||||||||||||
| 3 | 20 | 20 | 50 | 10 | — | — | — | — | — | — | — | 90 | — | — | — | 10 | — | — | — | Bad | Medium—scaling some | Good | Good | Fair | |||||||||||||||||||
| 4 | 48 | .5 | — | 48 | .5 | 3 | — | — | — | — | — | — | — | 83 | — | — | — | 17 | — | — | — | Medium | Considerable with lateral cracking |
Good | Good | Medium | |||||||||||||||||
| 5 | 22 | — | 50 | 2 | 26 | — | — | — | — | — | — | 90 | — | — | — | — | 10 | — | — | Slight | Slight | Good | Good | Good | |||||||||||||||||||
| 6 | — | — | 64 | — | — | 36 | — | — | — | — | — | 98 | — | — | — | — | — | 2 | — | Medium | Considerable | Medium | Medium | Fair | |||||||||||||||||||
| 7 | 37 | — | 63 | — | — | — | — | — | — | — | — | 85 | 13 | — | 2 | — | — | — | — | Considerable | Present; long cracks | Fair | Fair | Poor | |||||||||||||||||||
| 8 | 38 | — | 48 | — | — | — | 14 | — | — | — | — | 91 | 9 | — | — | — | — | — | — | Slight | Surface checking | Good | Good | Fair | |||||||||||||||||||
| 9 | — | — | 73 | 2 | — | — | 25 | — | — | — | — | 66 | — | — | 12 | 22 | — | — | — | Not evident | Considerable with lateral cracking |
Medium | Good | Medium | |||||||||||||||||||
| 10 | 44 | — | 46 | 5 | — | — | — | — | — | — | — | 86 | .0 | 12 | .5 | — | 1 | .5 | — | — | — | — | Medium | Very slight | Good | Good | Good | ||||||||||||||||
| 11 | 50 | — | 50 | — | — | — | — | — | 5 | — | — | 78 | 22 | — | — | — | — | — | — | Slight | Lateral cracking | Fair | Fair | Fair | |||||||||||||||||||
| 12 | 60 | — | 34 | — | — | — | — | 6 | — | — | — | 91 | 7 | — | 2 | — | — | — | — | Considerable | Present with slight cracking and scaling |
Fair | Fair | Not very good | |||||||||||||||||||
| 13 | — | 60 | 27 | 3 | — | — | — | — | 10 | — | — | 90 | — | — | — | — | 10 | — | — | Medium | Surface checking only | Good | Good | Good | |||||||||||||||||||
| 14 | 25 | 20 | 25 | 5 | — | — | — | — | — | 25 | — | 90 | — | 6 | — | — | — | — | 4 | Considerable | Considerable with lateral cracking |
Medium | Fair | Medium; some washing shown |
|||||||||||||||||||
| 15 | — | 20 | 40 | 10 | — | — | — | — | — | — | 30 | 90 | — | 8 | 2 | — | — | — | — | Medium | Medium | Good | Good | Medium | |||||||||||||||||||
| 16 | 33 | — | 33 | — | — | 34 | — | — | — | — | — | 90 | — | 10 | — | — | — | — | — | Medium | Slight; some shelling | Fair | Good | Medium | |||||||||||||||||||
| 17 | 100 | (Type | A) | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | Bad | Alligatoring; deep checking | Good | Fair | Poor | |||||||||||||||||||
| 18 | 100 | ( „ | B) | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | Bad | Alligatoring; deep checking | Fair | Fair | Poor | |||||||||||||||||||
| 19 | 100 | ( „ | C) | — | — | — | — | — | — | — | — | 10 gal. oil reduction | — | — | — | — | Bad | Deep | Good | Fair | Poor | ||||||||||||||||||||||
| 20 | — | 100 | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | Considerable | Slight | Good | Fair | Fair | |||||||||||||||||||
| 21 | — | — | 100 | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | — | Not evident | Considerable; slight cracking; scaling |
Fair | Good | Poor | |||||||||||||||||||
| 22 | — | — | — | — | — | — | — | — | — | — | 100 | — | — | — | — | — | — | — | — | Medium | Lateral cracking; split | Good | Good | Fair | |||||||||||||||||||
| 23 | 100 | (Type | C) | — | — | — | — | — | — | — | — | 51⁄2 gal. oil reduction for priming | Bad | Medium deep | Good | Good | Fair | ||||||||||||||||||||||||||
| 24 | 37 | .51 | 7 | .84 | 25 | .87 | 20 | .36 | — | — | 8 | .42 | (Michigan Seal White Lead) | — | — | — | — | — | — | — | Considerable | Slight; lateral cracking | Fair | Good | Good | ||||||||||||||||||
| 25 | 38 | .95 | 4 | .81 | 33 | .58 | 19 | .48 | — | — | 3 | .18 | (Railway White Lead) | — | — | — | — | — | — | — | — | Considerable | Some; lateral cracking | Fair | Good | Excellent | |||||||||||||||||
| 200 | 15 | .625 | — | — | 1 | .875 | — | — | — | — | 1 | .250 | — | 43 | .750 | 32 | .250 | 4 | .000 | — | 1 | .250 | — | — | — | — | Medium | Bad cracking | Good | Good | Fair | ||||||||||||
[196]“As before stated, the committee believes that a serious mistake was made on the test fence in painting out the leads and other formulas on the various woods without any special attention to reduction to suit the nature of the wood, thus accounting largely for the difference of the wearing of the paints on the different woods.
“The reduction of the white leads especially was to be criticised in these tests, in many cases too much oil and not sufficient turpentine being present to cause penetration.
“The application of paint to cedar was satisfactory in most all cases, and this wood showed much better results than the other woods upon the fences. The exudation of resinous pitch on the hard pine was extremely serious, in some cases coming through the paint in large streaks, causing bad results.
“It is to be regretted that the house repainting tests which were conducted are of no special value, inasmuch as no information is on file as to the composition of the old paints originally on the houses before the application of the test paints. Imperfections in the old coating, such as excessive chalking, deep checking, scaling, rosin exudations, etc., affected the subsequent coats in such a manner as to prevent any knowledge of where the new and old paint troubles began. The committee, therefore, omitted a detailed inspection of such tests.
“Examination of the three houses which were painted over new wood showed results which correspond with the results obtained from the fence tests. That is, they showed the ultimate value of high type mixtures of several pigments over one pigment alone. These tests seem to indicate that very good results can be secured from most of the paints sold in North Dakota. If the consumer or householder would exercise more care in the selection of wood and preparation of surfaces, with due regard to the proper reduction for various coats, more satisfactory results would be obtained.
“From an examination of certain paints on the 1908 fence containing petroleum spirits, it would appear that this paint thinner is of value, and in the face of conditions such as are presented by the present scarcity of turpentine, the use of petroleum spirits in moderate quantity would be justified.”
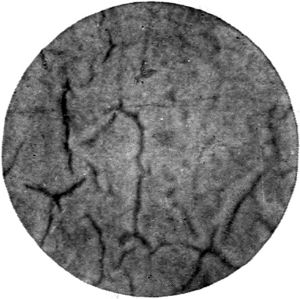 |
 |
| 1. Formula No. 21, Section 31, on 1907 Fence | 2. Section 80, on 1908 Fence |
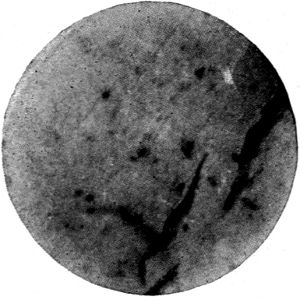 |
 |
| 3. Formula No. 6, Section 9, on 1907 Fence | 4. Formula No. 2, Section 3, on 1907 Fence |
 |
 |
| 5. Formula No. 1, Section 1, on 1907 Fence | 6. Formula No. 14, Section 21, on 1907 Fence |
[198] |
 |
| 7. Formula No. 13, Panel 19, on 1907 Fence | 8. Formula No. 19, Panel 28. Broad, Deep Checking on Corroded White Lead on 1907 Fence |
 |
 |
| 9. Formula No. 24, Panel 36, on 1907 Fence. Good Condition. Surface Checking Only | 10. Formula No. 25, Section 37, on 1907 Fence. Good Condition. Surface Checking Only |
 |
 |
| 11. Formula No. 8, Panel 12, on 1907 Fence | 12. Formula No. 10, Panel 15, on 1907 Fence |
[199] |
 |
| 13. Panel No. 34, Formula 23, on 1907 Fence. Deep Checking on Corroded White Lead | 14. Test No. 13 on 1906 Fence. White Spots Show Paint Left on Wood. Balance of Paint Split and Disintegrated from Surface |
 |
 |
| 15. Test No. 6 on 1906 Fence. Surface Checking Only | 16. Test No. 2, 1906 Fence. Sublimed White Lead |
 |
 |
| 17. Cracks in Test No. 15 on 1906 Fence | 18. Effect of Cracking on Hard Pine, Causing Splitting of Painting Coating |
[200] |
 |
| 19. Formula No. 22, Section 23, 1907 Fence. Cracks in Paint Coating, Caused by Cracks in Wood; Coating Otherwise in Good Condition | 20. Test No. 8, on 1906 Fence. Surface Checking Only |
 |
|
| 21. Combination Cracking and Checking on Section 69, on 1908 Fence |
|
 |
 |
| 22. Cracks in Paint Coating, Caused by Cracking of Hard Pine Wood | 23. Section 65 on 1908 Fence. Showing Early Breakdown of Corroded White Lead |
Location and Object of Tests. On September 15, 1910, the erection of a wooden test fence was completed on the State Fair Grounds at Nashville, Tenn. Upon this fence were exposed forty-two samples of white paint, the object of the test being to determine whether the combination type of formula is superior to the single pigment type in the southern plateau, of which Nashville is the centre.
Construction of Tests. The construction and outline of these tests differ somewhat from those conducted at Atlantic City and elsewhere by the Scientific Section. The fence frame is 150 feet long, being made of 6-inch bevelled girders supported three feet from the ground by 4-inch posts set six feet apart. Upon this girder were placed a series of forty-two test panels supported at top and bottom with weather strips and braces. The test panels used were 40 inches high, 30 inches wide, and one inch thick, being made of the highest grade white pine, tongued and grooved together, and protected on the edges by weather strips projecting from the surface of the panels. Each panel was painted on both sides with the same paint, thus giving an eastern and western exposure, the fence running north and south. The formulas used in the test vary in their percentage composition, being made up in some cases of single pigments, and again with combinations of the opaque white pigments, with and without certain percentages of the crystalline or inert pigments. The paints were applied under the supervision of prominent master painters and a committee representing the Scientific Section and other technical organizations.
Other field tests have shown that the sap and knots in hard-grained woods, such as yellow pine, cypress, etc., have been the cause of the failure of even the best paints, and that all tests should be conducted upon soft woods, such as white pine and poplar, if definite results are to be obtained. Paints tinted with ochre,[202] chrome yellow, lampblack, iron oxide, etc., have shown on the other field tests which have been conducted at Atlantic City, Pittsburg, and Fargo the value of these pigments in giving to the paints increased wearing properties. On the Southern Test Fence, therefore, all the formulas were ground in white only and placed upon white pine so as to make the test primarily one to determine the value of the various white pigments upon good wood.

Tennessee Test Fences
Oil and Thinner Tests. Upon one series of panels on the fence was placed one of the formulas which had given universal satisfaction on the various test fences in the past, and this formula was made up with various oils other than linseed oil, in order to determine the value of these oils as painting materials. For instance, the vehicle part of the one formula referred to is made up of 50% linseed oil and 50% soya bean oil, and again 50% linseed oil and 50% rosin oil, etc., an effort being made to test out a few of the available semi-drying oils.
The same formula referred to was ground in pure linseed oil and subjected to a series of tests where it has been thinned for application as priming and second coats with a series of wood turpentines obtained from the United States Forest Products Laboratory at Madison, Wis. These turpentines were made from southern pine stumps and sawdust, and they vary greatly in their properties. Some were objectionable in odor, while others were of excellent quality, having an odor almost equal to that of pure gum spirits.



Views of Fence
[204]One product under test on the Southern Test Fence is pine oil, a high boiling point product obtained from the manufacture of wood turpentine from sawdust. This oil has a boiling point of over 210 degrees Centigrade as against the 150 degrees of ordinary gum spirits. It is almost water white and has the same penetrating qualities as the pure gum spirits; when mixed with 50% linseed oil forming a paint oil of extremely light color, that produces a semi-flat paint of great whiteness.
Reductions and Application. Formulas No. 1 to No. 37 were all ground in pure refined linseed oil. They were made in the form of semi-paste and then thinned down with sufficient refined linseed oil so that each would have a relative viscosity. To each formula was then added a sufficient amount of pure lead and manganese linoleate drier to give proper drying qualities. On thinning for the priming coat, one pint of turpentine was added to each gallon of paint. For the second coat, one-half pint turpentine and one-half pint refined linseed oil were added to each gallon. For the third coat work, reduction was made with one pint of refined linseed oil.
In the case of formulas 31 to 37, reductions were the same, except that a series of specially prepared wood turpentines were used in place of the pure gum spirits used in formulas 1 to 31.
Formulas 38 to 41, as will be shown, were ground in equal parts of the oils tested. These formulas, however, were all thinned for application with pure gum spirits of turpentine, and the respective vehicle in which they were ground.
No inspection of the Tennessee Test Fence has yet been made. The formulas tested are as follows:
Vehicle: Bleached Linseed Oil with Lead and Manganese Linoleate Drier.
| For- mula No. |
||
| 1 | [27]Corroded white lead | 100% |
| 2 | [27]Sublimed white lead | 100% |
| 3 | Zinc oxide XX | 100% |
| 4 | Zinc lead white | 100% |
| 5 | Leaded zinc 65%, corroded white lead | 35% |
| 6 | [27]Corroded white lead | 100% |
| 7 | [27]Corroded white lead | 100% |
[27] Corroded White Lead is the Basic Carbonate of Lead. Sublimed White Lead is the Basic Sulphate of Lead.
| No. 8 | |
| Corroded white lead | 85% |
| Zinc oxide | 15% |
| 100% | |
| No. 9 | |
| Corroded white lead | 65% |
| Zinc oxide | 35% |
| 100% | |
| No. 10 | |
| Corroded white lead | 50% |
| Zinc oxide | 50% |
| 100% | |
| No. 11 | |
| Corroded white lead | 40% |
| Zinc oxide | 60% |
| 100% | |
| No. 12 | |
| Corroded white lead | 30% |
| Zinc oxide | 70% |
| 100% | |
| No. 13 | |
| Corroded white lead | 45% |
| Zinc oxide | 45% |
| Silica | 10% |
| 100% | |
| No. 14 | |
| Corroded white lead | 45% |
| Zinc oxide | 45% |
| Asbestine | 10% |
| 100% | |
| No. 15 | |
| Corroded white lead | 45% |
| Zinc oxide | 45% |
| China clay | 10% |
| 100% | |
| No. 16 | |
| Corroded white lead | 45% |
| Zinc oxide | 45% |
| Barytes | 10% |
| 100% | |
| No. 17 | |
| Corroded white lead | 45% |
| Zinc oxide | 40% |
| Silica | 15% |
| 100% | |
| No. 18 | |
| Corroded white lead | 45% |
| Zinc oxide | 40% |
| Asbestine | 15% |
| 100% | |
| No. 19 | |
| Corroded white lead | 45% |
| Zinc oxide | 40% |
| Barytes | 15% |
| 100% | |
| No. 20 | |
| Sublimed white lead | 45% |
| Zinc oxide | 40% |
| Silica | 15% |
| 100% | |
| No. 21 | |
| Sublimed white lead | 45% |
| Zinc oxide | 40% |
| Asbestine | 15% |
| 100% | |
| No. 22 | |
| Sublimed white lead | 45% |
| Zinc oxide | 40% |
| Barytes | 15% |
| 100% | |
| No. 23 | |
| Zinc oxide | 90% |
| Calcium carbonate | 10% |
| 100% | |
| No. 24 | |
| Sublimed white lead | 40% |
| Zinc oxide | 45% |
| Calcium carbonate | 15% |
| 100% | |
| No. 25 | |
| Corroded white lead | 35% |
| Zinc oxide | 50% |
| Silica | 15% |
| 100% | |
| No. 26 | |
| Corroded white lead | 20% |
| Sublimed white lead | 30% |
| Zinc oxide | 40% |
| Asbestine | 10% |
| 100% | |
| No. 27[206] | |
| Corroded white lead | 20% |
| Sublimed white lead | 20% |
| Zinc oxide | 40% |
| Barytes | 10% |
| Asbestine | 10% |
| 100% | |
| No. 28 | |
| Corroded white lead | 20% |
| Sublimed white lead | 20% |
| Zinc oxide | 40% |
| Calcium carbonate | 10% |
| Silica | 10% |
| 100% | |
| No. 29 | |
| Sublimed white lead | 20% |
| Corroded white lead | 20% |
| Zinc oxide | 30% |
| Barytes | 10% |
| Asbestine | 10% |
| Calcium carbonate | 10% |
| 100% | |
| No. 30 | |
| Corroded white lead | 33% |
| Zinc oxide | 33% |
| Barytes | 33% |
| 99% | |
| No. 31 | |
| Corroded white lead | 45% |
| Zinc oxide | 45% |
| Asbestine | 5% |
| Calcium carbonate | 5% |
| 100% | |
| For- mula No. |
|
| 32. | Same as No. 31 but thinned with wood turpentine No. 1. |
| 33. | Same as No. 31 but thinned with wood turpentine No. 2. |
| 34. | Same as No. 31 but thinned with wood turpentine No. 3. |
| 35. | Same as No. 31 but thinned with wood turpentine No. 4. |
| 36. | Same as No. 31 but thinned with wood turpentine No. 5. |
| 37. | Same as No. 31 but thinned with high-boiling-point petroleum spirits (turpentine substitute). |
| 38. | Same as No. 31 but ground in 50% raw linseed oil, 50% soya bean oil. |
| 39. | Same as No. 31 but ground in 50% raw linseed oil, 50% corn oil. |
| 40. | Same as No. 31 but ground in 50% raw linseed oil, 50% cotton seed oil. |
| 41. | Same as No. 31 but ground in 50% raw linseed oil, 50% rosin oil. |
| 42. | Same as No. 31 but ground in 50% raw linseed oil, 50% pine oil. |
The new vehicle test fence at Washington is fully described in the writer’s paper[28] as presented before the American Society for Testing Materials, as follows:
[28] The Practical Testing of Drying and Semi-Drying Paint Oils, by Henry A. Gardner. Paper presented at Fourteenth Annual Meeting, Amer. Soc. for Test. Mater., Atlantic City, N.J., June, 1911.
“The high price attained by linseed oil during the past two years of over a dollar a gallon, together with the unusual scarcity of this valuable oil, has led many investigators into the field of research, with a view of discovering some mixture of other oils to partly replace linseed oil. Many valuable contributions to oil technology have resulted, but the makers and users of paints have wisely demanded specific and authoritative information as to the practical value of proposed mixtures before adopting them. The Institute of Industrial Research, at the request of the Paint Manufacturers’ Association of the United States, has recently started a series of practical paint vehicle tests designed to decide the question at issue.
“Forty-eight white-pine panels have been placed upon a test frame on the grounds of the new laboratory building of the Institute, at Washington, D. C. They are painted with a standard white pigment formula reduced with a different oil formula for every panel. White-pine panels were selected for the test on account of the good painting surface which this type of lumber presents; the grade selected was free from knots or pitch pockets—defects which often ruin a paint test. Each panel was constructed of four tongued-and-grooved planed boards, 22 inches long, 1 inch thick, and 9 inches wide. The boards were leaded together and capped at the sides with weather strips, making the finished panels about 2 feet wide and 3 feet high. The fence upon which the panels were placed was constructed of 4-inch squared yellow pine with open framework, allowing the panels[208] a resting place upon which they were finally secured with sherardized screws.
“Before erecting the panels, they were carefully painted in a paint laboratory especially fitted out for the tests. The work was done during the months of April and May, the temperature averaging from 60 degrees to 90 degrees Fahrenheit. This precaution was taken in order that the paint in each case might become thoroughly dry and hard before exposure, so that there would be no accumulation of dust or effect from exposure during the drying period. The actual painting of each panel was done personally by Mr. Charles Macnichol, master painter, of Washington, D. C., who has had a wide experience in the practical application and testing of paints.

View of Panels on Washington Test Fence
“The viscous nature of several of the oils tested precluded the possibility of grinding each oil formula with the white pigment base selected; great heating of the paint mills and a paste of insufficient fineness was the result of an early attempt at this method. It was decided, therefore, to grind the standard pigment formula to a thick paste in the minimum amount of raw linseed oil. Subsequently a weighed amount of the white pigment base was thinned with the oil formula to be tested, to a standard viscosity, judged by the experienced master painter in charge of the practical application of the formulas as sufficiently heavy for third-coat work. When making the reductions with oil mixtures, an allowance was made for the amount of linseed oil already contained in the ground white pigment base.
“During the application of the first coat an equal amount of turpentine was added to each formula, in the proportion of one-half[209] pint to a gallon of paint; in the application of the second coat there was added to each formula a like amount of an equal mixture of turpentine and the oil formula under test. The third coat was applied without the addition of thinners of any kind.
“It is well known that the time of drying and the condition of the dried film of any oil or mixture of drying or semi-drying oils will vary widely. It is for the purpose of causing oils to set up to a hard film in a short time that metallic driers in the form of salts of manganese and lead, soluble in oil, are added to a paint. Some oils require a large amount of drier, while others require only a very small amount. Those which require a large amount are apt, upon exposure, to be burned up by the drier, resulting in the formation of a powdered and disintegrated film. To add various types of drier or even differing amounts of a drier to the oils under test, seemed very unfair from every standpoint, and it was therefore decided to eliminate the drier question entirely, so as not to vitiate the results by bringing in a factor of this nature. The plan of omitting driers proved successful in the Atlantic City steel-panel paint tests, erected three years ago by the writer under the supervision of Committee A-5 of this Society.
“The systematic methods which are necessary when making paint tests were carefully followed. A standard weighed amount of white pigment paste was placed in a clean paint cup and thinned to the proper consistency with a weighed amount of the oil under test. Proper reductions were made, as before stated. Weighings of the paint, cup, and brush were made before and after application to the panel, in order to determine the quantity of paint used and the spreading power. A period of fifteen days was allowed between the application of successive coats, in order to give each formula sufficient time to dry thoroughly. Although several of the formulas remained tacky for over a week, all dried thoroughly in the time allotted. (Oils which when used alone have slow drying properties, have been found to yield good firm films when used with drying pigments such as lead and zinc.) The backs and edges of each panel were painted with two coats of the paint used on the face of the panel, so as to prevent the admission of moisture. After erection, the panels were numbered with aluminum figures pressed into the surface. Frequent[210] inspections will be made, and at the proper time reports will be issued giving the results of the tests.
“During the painting of the panels considerable interesting data were collected, of which the following is a brief résumé:
“The hiding power of a paint is one of its most important requisites. It was found in the tests that some oils had the effect of lessening, while others had the effect of increasing the hiding power of the standard pigment formula. This may be due in part to the varying refractive indices of the oils used, as well as to the difference in the quantity of oil required in each test. Some oils were very viscous, while others were very light.
“The stiff working of heavy-bodied, blown, or heat-oxidized oils, produced films which in some cases gave a very glossy surface, even on the priming coat. Some of these resembled varnished work when finished. It will be of importance to watch these tests carefully for any signs of early breakdown, which might come from too thick a film. The treated Chinese wood oil paints worked rather stiff but produced very smooth films. The rosin oil paints became slightly lumpy on standing, but worked out to a smooth finish somewhat yellowish in color. The marine animal oils, especially the menhaden oil mixtures, dried to a film slightly flatter than straight linseed oil. Any odor which was present in the paints made from the animal oils seemed to disappear a few hours after application. The cotton seed and corn oil mixtures made the slowest drying paints, but at the end of the second week of the drying period they set up rapidly to firm films. Soya bean and perilla oils behaved like straight linseed oil, the former being a little slower and the latter slightly more rapid in drying properties. The perilla oil was made from one of the first importations into this country, and was dark in appearance. It made, however, a very easy-working and hard-drying paint.
“The oils used in the tests were obtained from reliable sources. After they were received, they were carefully analyzed. The results of the analyses appear in Table 1.
| Specific Gravity |
Saponifi- cation Number |
Iodine Number |
Acid Number |
|||||
| Raw linseed oil | 0.93 | 1 | 188 | 186 | 2.0 | |||
| Boiled linseed oil (linoleate type) | 0.94 | 1 | 187 | 172 | 2.7 | |||
| Boiled linseed oil (resinate type) | 0.93 | 0 | 186 | 176 | 2.2 | |||
| Blown linseed oil | 0.96 | 8 | 189 | 133 | 2.8 | |||
| Lithographic linseed oil | 0.97 | 0 | 199 | 102 | 2.7 | |||
| Soya bean oil | 0.92 | 4 | 189 | 129 | 2.3 | |||
| Menhaden oil | 0.93 | 2 | 187 | 158 | 3.9 | |||
| Perilla oil | 0.94 | 188 | 180 | 2.0 | ||||
| Chinese wood oil (raw) | 0.94 | 4 | 183 | 166 | 3.8 | |||
| Chinese wood oil (treated) [29] | 0.89 | 8[29] | 128 | [29] | 104 | [29] | 6.8 | [29] |
| Corn oil | 0.92 | 5 | 191 | 118 | 9.5 | |||
| Cottonseed oil | 0.92 | 1 | 193 | 105 | 3.6 | |||
| Rosin oil | 0.96 | 6 | 27 | 41 | 16.7 | |||
| Whale oil | 0.92 | 4 | 191 | 148 | — | |||
| Neutral petroleum oil[30] | 0.91 | 6 | 6 | 12 | — | |||
[29] Low constants due to presence of over 40% of volatile matter, largely petroleum spirits.
[30] This oil contained over 20% of petroleum spirits.
“The pigment formula selected for the tests had the following composition:
| Basic carbonate-white lead | 20% | ||
| Zinc oxide | 35% | ||
| Magnesium silicate | 10% | ||
| Barytes | 5% | ||
| 100 lbs. of pigment base ground to a stiff paste in 16 lbs. of linseed oil. | |||
“While this pigment formula was not selected as being superior to certain other formulas, it is of a type that has given very fair service in paint tests throughout the country, and will no doubt serve admirably for the purpose designed in these tests.
“The vehicle formulas in the finished paints are as follows:
| No. 1 | |||
| Raw linseed oil | 100% | ||
| No. 2[31] | |||
| Soya bean oil | 100% | ||
| No. 3[32] | |||
| Menhaden oil | 100% | ||
| No. 4 | |||
| Raw linseed oil | 25% | ||
| Boiled linseed oil (resinate) | 75% | ||
| No. 5 | |||
| Raw linseed oil | 25% | ||
| Boiled linseed oil (linoleate) | 75% | ||
| No. 6 | |||
| Raw linseed oil | 50% | ||
| Boiled linseed oil (resinate) | 50% | ||
| No. 7 | |||
| Raw linseed oil | 50% | ||
| Boiled linseed oil (linoleate) | 50% | ||
| No. 8[212] | |||
| Raw linseed oil | 50% | ||
| Blown linseed oil | 50% | ||
| No. 9 | |||
| Raw linseed oil | 50% | ||
| Litho. linseed oil | 50% | ||
| No. 10 | |||
| Raw linseed oil | 50% | ||
| Soya bean oil | 50% | ||
| No. 11 | |||
| Raw linseed oil | 50% | ||
| Menhaden oil | 50% | ||
| No. 12 | |||
| Raw linseed oil | 50% | ||
| Perilla oil | 50% | ||
| No. 13 | |||
| Raw linseed oil | 50% | ||
| Treated wood oil | 50% | ||
| No. 14 | |||
| Raw linseed oil | 50% | ||
| Corn oil | 50% | ||
| No. 15 | |||
| Raw linseed oil | 50% | ||
| Cottonseed oil | 50% | ||
| No. 16 | |||
| Raw linseed oil | 50% | ||
| Rosin oil | 50% | ||
| No. 17 | |||
| Raw linseed oil | 50% | ||
| Whale oil | 50% | ||
| No. 18 | |||
| Raw linseed oil | 75% | ||
| Soya bean oil | 25% | ||
| No. 19 | |||
| Raw linseed oil | 75% | ||
| Menhaden oil | 25% | ||
| No. 20 | |||
| Raw linseed oil | 75% | ||
| Perilla oil | 25% | ||
| No. 21 | |||
| Raw linseed oil | 75% | ||
| Treated wood oil | 25% | ||
| No. 22 | |||
| Raw linseed oil | 75% | ||
| Corn oil | 25% | ||
| No. 23 | |||
| Raw linseed oil | 75% | ||
| Cottonseed oil | 25% | ||
| No. 24 | |||
| Raw linseed oil | 75% | ||
| Rosin oil | 25% | ||
| No. 25 | |||
| Raw linseed oil | 50% | ||
| Soya bean oil | 25% | ||
| Menhaden oil | 25% | ||
| No. 26 | |||
| Raw linseed oil | 50% | ||
| Soya bean oil | 25% | ||
| Treated wood oil | 25% | ||
| No. 27 | |||
| Blown linseed oil | 50% | ||
| Soya bean oil | 50% | ||
| No. 28 | |||
| Raw linseed oil | 25% | ||
| Soya bean oil | 25% | ||
| Menhaden oil | 25% | ||
| Treated wood oil | 25% | ||
| No. 29 | |||
| Raw linseed oil | 25% | ||
| Soya bean oil | 25% | ||
| Menhaden oil | 25% | ||
| Corn oil | 25% | ||
| No. 30 | |||
| Raw linseed oil | 25% | ||
| Soya bean oil | 25% | ||
| Menhaden oil | 25% | ||
| Cottonseed oil | 25% | ||
| No. 31 | |||
| Raw linseed oil | 25% | ||
| Soya bean oil | 25% | ||
| Menhaden oil | 25% | ||
| Rosin oil | 25% | ||
| No. 32 | |||
| Raw linseed oil | 25% | ||
| Soya bean oil | 25% | ||
| Treated wood oil | 25% | ||
| Rosin oil | 25% | ||
| No. 33 | |||
| Raw linseed oil | 20% | ||
| Soya bean oil | 20% | ||
| Treated wood oil | 20% | ||
| Menhaden oil | 20% | ||
| Cottonseed oil | 20% | ||
| No. 34[213] | |||
| Raw linseed oil | 20% | ||
| Soya bean oil | 20% | ||
| Treated wood oil | 20% | ||
| Menhaden oil | 20% | ||
| Rosin oil | 20% | ||
| No. 35 | |||
| Raw linseed oil | 40% | ||
| Soya bean oil | 20% | ||
| Corn oil | 20% | ||
| Cottonseed oil | 20% | ||
| No. 36 | |||
| Whale oil | 33% | ||
| Treated wood oil | 33% | ||
| Raw linseed oil | 33% | ||
| No. 37 | |||
| Raw linseed oil | 25% | ||
| L. O.[33] | 75% | ||
| No. 38 | |||
| Raw linseed oil | 50% | ||
| Raw Chinese wood oil | 50% | ||
| No. 39 | |||
| Raw linseed oil | 75% | ||
| Reducing oil[34] | 25% | ||
| No. 40 | |||
| Raw linseed oil | 50% | ||
| Soya bean oil | 35% | ||
| Neutral petroleum oil | 15% | ||
| No. 41 | |||
| Raw linseed oil | 50% | ||
| Soya bean oil | 25% | ||
| Neutral petroleum oil | 15% | ||
| Tungate drier | 10% | ||
| No. 42 | |||
| Linseed oil | 25% | ||
| Soya bean oil | 37% | ||
| Neutral petroleum oil | 23% | ||
| Tungate drier | 15% | ||
| No. 43 | |||
| Raw linseed oil | 25% | ||
| Soya bean oil | 37% | ||
| Whale oil | 19% | ||
| Tungate drier | 19% | ||
| No. 44 | |||
| Special test on white base of the following composition, in pure linseed oil: | |||
| Asbestine | 10% | ||
| Corroded white lead | 20% | ||
| Sublimed white lead | 30% | ||
| Zinc oxide | 40% | ||
| Upper board of panel reduced with straight turpentine on priming coat. | |||
| Second board of panel reduced with wood turpentine on priming coat. | |||
| Third board of panel reduced with pine oil on priming coat. | |||
| Bottom board of panel reduced with petroleum spirits on priming coat. | |||
| No. 45 | |||
| Same pigment formula as No. 44, reduced with: | |||
| Pine oil | 50% | ||
| Linseed oil | 50% | ||
| No. 46 | |||
| Special test of white base of the following composition, in pure linseed oil: | |||
| Corroded white lead | 20% | ||
| Sublimed white lead | 30% | ||
| Zinc oxide | 35% | ||
| Asbestine | 15% | ||
| No. 47 | |||
| Cypress panel unpainted. | |||
| No. 48 | |||
| Cypress panel painted with formula No. 1, thinned with benzol on the priming coat. | |||
[31] Dry pigment formula in soya bean oil.
[32] Dry pigment formula in menhaden oil.
[33] Mixture of boiled tung and soya bean oil, thinned with petroleum and turpentine.
Damp-proofing and Waterproofing. The decoration and preservation of cement and concrete is a subject which is being given the careful consideration of many technologists on account of the wide usage of cement for structural purposes, and the necessity of properly guarding it against the destructive effects of moisture.
To obtain with various paints decorative effects, and, at the same time, provide a high degree of damp-proofing, is a process quite distinct from that of water-proofing cement and concrete superstructures. The use, in small percentage, of stearic acid solutions, aluminum stearate, marine animal soaps, and other lime-reacting materials, as a component of concrete while it is being mixed, has been in practice for some time, the resulting mixture being used largely upon base-work subjected to water under high pressure. Although some of the materials used for such purposes actually do give to the concrete a high power of water resistance, the degree of waterproofing to be obtained through the use of many such compounds varies to a wide extent, often interfering with the lime-silica reactions, and ultimately affecting the strength of the finished concrete.
Decorative and Preservative Coatings. The necessity of obtaining suitable paint coatings for cement and concrete surfaces suggested to the writer a series of tests on paints designed to prevent the destructive action of the lime which, by seepage and other physical action, is brought to the surface, causing saponification of some oil coatings, as well as destruction of color. The tests referred to were carried out during 1908, and although great advances have been made since that time in the preparation of concrete paints, the tests have, nevertheless, afforded information of a valuable nature as indicating the proper methods to follow in the painting of cement, as well as suitable materials to use in the manufacture of cement paints. The tests, moreover, show the comparative durability of a number of paints typical of those prominent in the market at the time the tests were started.

View of Concrete Paint Test Panels
[215]Acid Reacting Compounds. A series of acid reacting washes were included in the tests, having been designed as prime coaters for use previous to the application of oil paints. The application of many of these washes has the effect of neutralizing the lime within cement and concrete surfaces, and often precipitate insoluble lime compounds which aid in filling up the outer voids, thus presenting a surface more suitable to receive oil coatings. To the writer who has since made a careful study of the painting of concrete, it would seem advisable for painters to avoid, when possible, the use of these lime neutralizing washes, as some of them have more or less disintegrating and weakening influences upon concrete. Recent laboratory experiments, however, have indicated that zinc sulphate, an acid reacting material used for many years as a wash for concrete surfaces by Macnichol, actually has a strengthening effect upon cement and concrete surfaces. The more successful coatings of to-day, however, are those which may be placed directly upon the cement and concrete surfaces without the aid of such washes. Several fairly successful paints of this type have recently appeared in the market; some of them being made of acid rosins compounded with vegetable oils. Probably one of the first mixtures of this sort was the so-called suction varnish which the master painter has for years used as a prime coating on plastered walls previous to painting. These suction varnishes generally contain a high percentage of rosin,[216] a material having an exceptionally high acid value and thus lending itself successfully to the neutralization of free lime. It has been claimed, however, by certain practical painters that the lime-rosin compounds formed when such paints are applied to the exterior of buildings, are of a brittle nature and subject to early failure. If this is true, it would seem advisable to use in a concrete paint an oil of a relatively unsaponifiable nature, which would withstand successfully the action of the lime, and, at the same time, prevent disruption of the coating and failure of the color used in the paint.
Outline of Tests. The tests referred to as carried out by the writer were made on a brick wall forty feet long, surface-coated with a four-inch coating of Portland cement mortar made of one part of Portland cement and three parts of sharp, clean sand. After the cement had hardened for three days, the solutions under test were applied.
In many of the tests outlined above, one-coat, as well as two-coat work, was used on different sections of the test surfaces. It was shown that the two-coat work gave far better results than with the one-coat work, and the writer would recommend for the painting of concrete at least two-coat work. Whenever paints containing Prussian blue or chrome green are applied to concrete surfaces, immediate whitening in the case of the blue, and yellowing in the case of the green, will take place, if any degree of action has been exerted by the lime within the concrete. For this reason, green is an especially delicate color to test and should be utilized for this purpose.
The materials used, and the results shown at an inspection made after two years’ exposure, are given herewith.
Test No. 1. Concrete primed with a 25% solution of zinc sulphate crystals dissolved in water. A wide brush was used for the application, and the spreading rate was approximately 200 square feet per gallon. Second and third coated on the second day with No. 119 blue paint of the following composition:
No. 119 Blue Paint
| Sublimed white lead | 50% |
| Zinc oxide | 35% |
| Silica and barytes | 12% |
| Prussian blue | 3% |
| Ground in linseed oil, turpentine and drier. | |
[217]This panel, after three years’ exposure, is in good condition. Slight checking observed.
Test No. 2. Concrete primed with a 20% solution of (alum) (aluminum sulphate). Second and third coated with No. 119 blue.
In similar condition to Test No. 1.
Test No. 3. Concrete primed with zinc sulphate followed by two coats of para red.
Para Red Formula
| Blanc fixe | 60% |
| Whiting | 25% |
| Zinc oxide | 3% |
| Paranitraniline lake | 12% |
| Ground in linseed oil, turpentine and drier. | |
Panel in fair condition with exception of slight crazing. Characteristic dullness of color after exposure shown. Bright red color restored upon washing.
Test No. 4. Concrete primed with an 8% solution of stearic acid and rosin dissolved in benzine. Second and third coated with No. 119 blue.
This panel is not in as good condition as Tests Nos. 1 and 2, and would indicate the inferiority of the priming liquid used. Color failing in spots and checking observed.
Test No. 5. Concrete primed with mixture used in Test No. 4, and then given two coats of para red.
Test is in about the same condition as No. 4.
Test No. 6. Concrete primed with a 10% mixture of acid calcium phosphate, followed with two coats of No. 119 blue.
The acid phosphate solution evidently had a neutralizing effect upon the lime in the concrete, as the paint is in fair condition.
Test No. 7. Concrete primed with one coat of a soap emulsion of the following composition, then painted with two coats of No. 119 blue.
| Water | 85% |
| Linseed oil | 12% |
| Alkali | 3% |
Very poor results obtained. Destruction of color and peeling resulted.
[218]Test No. 8. Concrete primed with one coat of white paint of the following composition:
Primer
| Zinc oxide | 25% |
| Silica | 35% |
| Corroded white lead | 20% |
| Gypsum | 15% |
| Whiting, etc. | 5% |
| Ground in a vehicle of linseed oil and containing 35% of volatile hydrocarbon spirits and drier. |
|
This coat was followed by one of the following composition, tinted blue:
| Zinc oxide | 60% |
| Gypsum | 20% |
| Silica | 20% |
| Ground in linseed oil with 12% of turpentine and drier. |
|
Fair results shown during first year, but a breakdown occurred during the second year, and cracking and scaling resulted.
Test No. 9. This test was a duplicate of No. 8 with the addition of 5% of zinc sulphate solution emulsified into the primer.
Slightly superior to Test No. 8.
Test No. 10. Primed with a white paste paint thinned with turpentine. Second coated with same paint tinted blue.
Formula of Paste
| Zinc oxide | 40% |
| Whiting | 30% |
| Silica | 20% |
| Alumina and gypsum | 10% |
| Ground in 16% of linseed oil vehicle. |
|
Scaling and peeling due to lack of binder and use of saponifiable oil resulted during the first six months’ exposure. Entire destruction of coating at end of two years.
Test No. 11. Primed with a white mixture, and second coated with the same mixture tinted blue.
Formula of Mixture
| Whiting | 30% |
| Silica | 30% |
| Zinc oxide | 40% |
| Stirred into a 5% solution of glue in water, until a fairly thick paste was obtained. |
|
[219]Much chalking was shown, and a bleaching of color. It is evident that this mixture would not serve to keep moisture out.
Test No. 12 A. Primed with a 5% solution of soluble nitrated cotton and paraffin dissolved in equal parts of amyl acetate and benzine. Second coated with No. 119 blue.
Not very good results were obtained, chalking and slight scaling resulting.
Test No. 12 B. Primed with a heavy varnish containing Chinese wood oil and kauri gum. Second coated with No. 119 blue.
Fair results obtained.
Tests Nos. 13, 14, 15, and 16. Primed with a solution made by dissolving 10 parts of sodium oxalate in 100 parts of water. Second and third coated with linseed oil paints in red, brown, blue, and green.
Very good results shown at end of test.
Test No. 20, Special. Primed and second coated with a green paint containing zinc oxide and barytes, ground in an oil having a low saponification value. Very slow drying was shown. Excellent results. No failure of color. Extremely glossy, waterproof surface presented.
The Necessity of Protective Coatings. Most painters have in the past considered of minor importance the painting of iron and steel; any paint that would properly hide the surface of the metal being accepted without much question. The demand, however, for structural steel for office buildings, factories, steel cars, railroad equipment, etc., has doubled the output of structural paints, and created a demand for painters having a knowledge of the proper materials to use in the painting of steel, so that its life may be preserved, and its strength maintained. Such knowledge is as important to the painter as a knowledge of how to properly select materials for the painting of wood, and how to temper these materials to suit the various conditions met with.
The Cause of Rust. Everyone is familiar with the appearance of rust, but few actually understand what causes rust. No attempt will be made here to present even an outline of the many theories advanced to explain the phenomenon of the rusting of iron, for the subject is as diverse as it is interesting. A brief résumé, however, will be given of the now generally accepted theory that explains the subject. This theory is called the electrolytic theory. “Auto-electrolysis” is the term used to define the peculiar tendency of iron to be transformed from a metal possessing a hard lustrous surface, high tensile strength, and other useful properties, to a crumbling oxide that falls to the ground and again becomes part of the earth from which it was originally taken by man.

A Side View of Steel Test Fences
[222]This “going back to nature” is more readily accomplished by most of the steel produced to-day than by the old hand-made irons produced many years ago. It seems to be a curious fact that the more quickly a product or an article is fashioned by man, the more quickly it tends to return again to its original oxidized condition. Some manufacturers of steel, however, through an understanding of the causes of rust, have progressed in the manufacture of slow rusting materials, either by the elimination, or by the proper distribution of impurities.
When iron is brought into contact with moisture, currents of electricity flow over the surface of the iron between points that are relatively pure and points that contain impurities. These currents stimulate the natural tendency of the iron to go into solution, and the solution proceeds with vigor at the positive points. The air which the water contains oxidizes the iron which has gone into solution, and precipitates the familiar brown iron rust. Thus water, which acts as an acid, and air, which acts as an oxidizer, have combined together to accomplish the downfall of the metal.
Three Photomicrographs of Corroding Steel
Inhibition and Stimulation of Rust. It is obvious that if means could be devised to stop the solution pressure of iron and make it resistant to the flow of surface electric currents, rust could be prevented. Such methods have been devised, and to better illustrate how they operate, an analogy may be drawn between iron in water and shellac in alcohol.
It is common knowledge that when shellac is placed in alcohol, the shellac will force itself into solution in the alcohol, and form a clear, transparent lacquer. If, however, there should be mixed[223] with the alcohol a quantity of water, it would be found that the shellac could no longer go into solution, and it would remain in its original condition. In the same way, if there be placed in water a small quantity of material, such as soluble chromates, or an alkaline substance like caustic soda or lime, it will be found that iron will no longer have a tendency to go into solution in this treated water, but will stay bright and clean. These materials which prevent the rusting of iron have been called by Cushman, who first advanced these explanations, “rust inhibitors,” or materials which inhibit rusting. The paint maker, realizing the importance of these rust inhibitors, is incorporating them into paints designed for the protection of iron and steel, and the success which paints of this type have met with from a practical standpoint is a justification of what was first called the “electrolytic theory,” which suggested their use.
By placing small, brightly polished steel plates into a mush of paint pigment and water, a determination may be made of the pigment’s effect upon the metal. Some pigments, under such conditions, cause rapid corrosion of the steel plates. Such pigments are stimulators of corrosion, on account of acid impurities which they contain, or because of their effect in stimulating galvanic currents. Many carbonaceous pigments are of this type. Other pigments have the effect of keeping bright the steel plates and preventing rust. Such pigments are of the inhibitive type, and their action is to check or retard the solution pressure of the iron.
The Effects of Moisture. It might occur to the reader that although paint pigments, when mixed up with water and brought into contact with the surface of steel, might show either an inhibitive or stimulative action, that it is by no means certain that the same tendency will be exhibited by pigments when they are properly mixed with linseed oil and laid out as a film upon the surface of steel. In answer to this, it may be well to state that almost no material used by mankind is absolutely dry. Linseed oil, as it is pressed from the seed, comes from the cells, carrying with it a certain small definite percentage of water, and it is quite certain that even the best linseed oil that goes into use is not theoretically dry. Everyone knows, of course, that oil and water do not readily mix and are, in fact, more or less repellent to each other. It is, however, true that, in spite of this, oils can[224] carry quite a percentage of water, without the admixture being apparent to the eye. In addition to this, careful experiments have proved very conclusively that linseed oil films, even after they have oxidized and hardened, have the power to a certain extent of absorbing water from the atmosphere. It is, therefore, safe to say that no linseed oil film in a paint coating is dry all the time. As a matter of fact, there is abundant evidence to show that in rainy weather, and, in fact, when the humidity in the air is high, paint films have absorbed water. As the sun comes out and warms the paint coating, and the humidity content of the atmosphere falls, this water to a large extent evaporates out of the film, only to be taken up again when the weather conditions change. This action may be likened to a breathing of the paint film, that is to say, an indrawing of water under humid conditions, followed by an exhaling of water under dry conditions. With these facts in mind, it must be apparent that pigments laid out in intimate contact with the surface of steel are subjected at all times either more or less to the reactions produced by water contact. Furthermore, as it is a property of water to become saturated with the gases of the atmosphere, such as oxygen, carbonic and sulphurous acids, and other impurities, there is present in a protective paint film at all times the elements necessary to carry on the corrosive process and reactions.
An outline of Cushman’s original research work, upon which has been based the classification of pigments as inhibitors, stimulators, and inerts, is clearly presented in his report[35] as Chairman of Committee U of the American Society for Testing Materials, of which the following is an excerpt:
[35] Page 73, 1910 Proceedings of the American Society for Testing Materials.
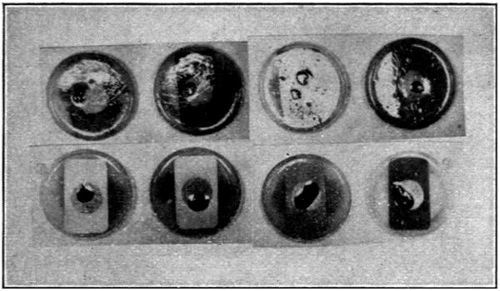
Ferroxyl Tests on Painted Steel Surfaces. Upper Row Painted with Stimulative Paints—Lower Row with Inhibitive Paints.
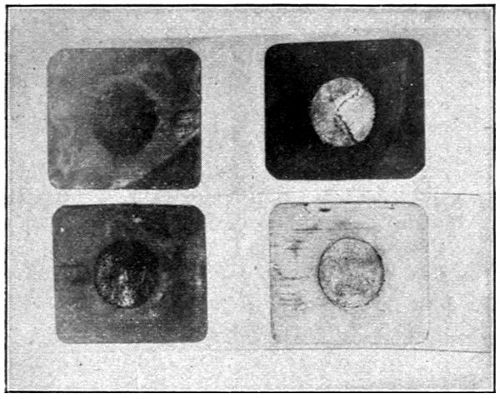
Water Test on Plates Painted—Except in Center Spot. Left Hand Plates Painted with Stimulative Paints, Right Hand Plates Painted with Inhibitive Paints.
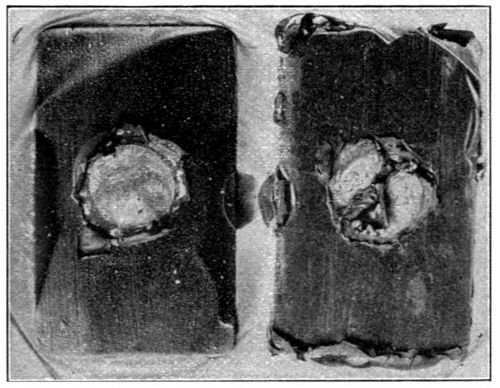
View of Steel Plates Painted with Stimulative Paints, after Immersion in Ferroxyl Jelly.
[226]“Three years ago the suggestion was made in a paper presented before the Tenth Annual Meeting of this Society that the various types of substances used as pigments in protective coatings might exert a stimulative or an inhibitive action on the rate and tendency to corrosion of the underlying metal. It was further suggested on a theoretical ground that slightly soluble chromates should exert a protective action when employed as pigments by maintaining the surface of the iron in a passive condition in case water and oxygen penetrated the paint film. In view also of the well-known fact that alkalies inhibit while acids stimulate the corrosion of iron, it was suggested that the action of more or less pure pigments on iron in the presence of water should be thoroughly investigated. Two years ago this Committee invited the co-operation of Committee D-1 (then known as Committee E) in the investigation, and a special sub-committee representing the two main committees was appointed.
“The methods and results of the water-pigment tests have previously been reported and published, and need not be given in detail. Briefly, the method consisted in immersing samples of steel in water suspensions of the various pigments and blowing air through the containers for definite periods of time, the corrosion being measured by the loss in weight sustained by the test pieces. About fifty pigments which are in more or less common use for painting steel were purchased in the open market and distributed among a number of the members of the Committee, who agreed to carry out the work. Each investigator worked independently of the others, except that the same general method was followed; the time of exposure to the corroding action, however, varied in the different experiments. When the results were compared and analyzed by the sub-committee, it was felt that the general agreement of the results obtained by the several investigators was striking and merited further and more systematic work. As a result of these tests the sub-committee tentatively divided the pigments into inhibitors, stimulators, and indeterminates. The word ‘indeterminate’ was selected after considerable discussion, because the words ‘neutral’ or ‘inert’ already possess a special meaning as applied to paint technology. The Committee takes this occasion to emphatically state that in adopting this tentative classification, the words ‘inhibitive’ and ’stimulative’ as used by them up to the present time apply only to the results obtained in the water tests, and the inference that the results obtained have decided which class the pigment will fall into when made into a paint with the usual vehicles and used as a protective coating on iron and steel, is not justified. In order to make this point quite clear, it has been agreed by the Committee to qualify the classification so as to speak of the various materials tested as ‘water stimulative’ or ‘water inhibitive.’”
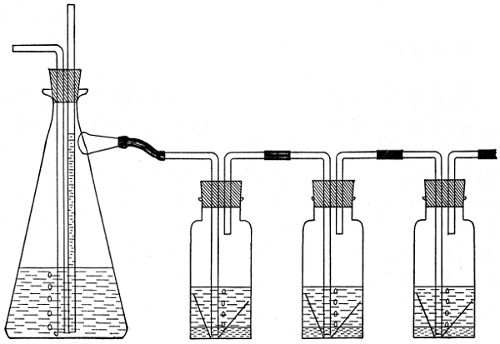
Apparatus for Testing the Inhibitive Value of Pigments
[228]Importance of Field Tests. Although the laboratory accelerated tests for the determination of the relative value of structural steel paints afford information of some import, there seems to be a general opinion that the best method to follow, if information of a reliable character is to be obtained, is to make actual field exposure tests upon large surfaces. The results of the above described water-pigment tests suggested the erection of a series of steel panels on which to test out the same pigments under practical service conditions. The Paint Manufacturers’ Association of the United States erected and painted the panels, the work being under the constant supervision of the writer, and the inspection of the work under Committee U of the American Society for Testing Materials. A brief résumé of the work[36] is herewith presented.
[36] Page 181, “Corrosion and Preservation of Iron and Steel”—Cushman and Gardner—McGraw-Hill Book Co., New York City.
Pickling and Preparation of Plates. The three types of metal[37] selected for the test were rolled to billets, the middle of which were selected, and worked up into plates 24 inches wide, 36 inches high, and 1⁄8 inch in diameter—approximately 11 gauge. A number of plates of each of the metals selected, in all 450, were pickled in 10% sulphuric acid, kept at 180 to 200 degrees Fahrenheit, in order to remove the mill-scale. The plates were then washed in water, and later in 10% solution of caustic soda. Finally the plates were again washed in water and wiped dry. They were then packed in boxes containing dry lime, in order to prevent superficial corrosion. By this method the plates were secured in perfect condition, the surfaces being smooth and free from scale. Upon these pickled plates paints were applied with a definite spreading rate of 900 square feet per gallon. The unpickled plates, coated with mill-scale, were painted with the same paints, but without adopting any special spreading rate, thus following more closely the ordinary method of painting structural steel. A few extra plates of special Bessemer steel and Swedish charcoal iron were also included in the test, some of which were painted, while others were exposed without any protective coating. Plates of the three types of metal already mentioned were also exposed unpainted, both in the black and pickled condition.
[37] Bessemer Steel, Open Hearth Steel, and Pure Iron.

Front View of Steel Test Fences
Fence Erection and Preparation for Work. The fences which were erected for the holding of the plates were constructed of[230] yellow pine, the posts being set deeply in the ground and properly braced. The framework of the fence was open, with a ledge upon the lateral girders, upon which the plates might rest, and to which the plates were secured by the use of steel buttons. After the framework had been erected, painted, and made ready for the placement of the panels, a small shed was built upon the ground, and the materials for the field test placed therein. The steel plates were unpacked from the boxes in which they were shipped, brushed off, and stacked up ready for painting. Small benches were erected, and the accessories of the work, such as cans, brushes, pots, balances, etc., were placed in position.
Methods Followed in Painting Plates. A frame resting upon the workbench served to hold the plates in a lateral position while being painted, room being allowed beneath the plate for the operator to place his hands in order to lift the plates from the under surface after the painting had been finished.
A pickled plate having been placed upon the framework everything was in readiness for the work. The specific gravity and weight per gallon of the paint to be applied was determined, and the amount, in grams, to be applied to each individual panel was calculated according to the following formula:
| Spreading rate | Sq. ft. in plate | Grams paint in gal. | ||||
| 900 sq. ft. | : | 6 | :: | 5400 | : | x |
The reciprocal of x being the number of grams of paint to be applied to the panels.
An enamel cup was then filled with the paint and a brush well stirred within. The cup, paint, and brush were placed upon the balances and accurately weighed in grams. After most of the paint had been applied to the panel, cross-brushing of the panel was continued until the pot with brush and paint exactly counterbalanced the deducted weight. The painted panel was then set in a rack, in a horizontal position to dry.
A period of eight days elapsed between the drying of each coat. The greatest care was taken in the painting of the edges of the plates, and the racks for containing the plates after they were painted were so constructed that the paint would not be abraded while sliding the plates back and forth. The working properties of each paint, and the appearance of the surface of each plate after painting, were carefully noted and included in[231] the report. No reductions were made to any of the paints applied except in three cases, where the viscosity was so great that it was necessary to add a small amount of pure spirits of turpentine. The amount of paint was proportionately increased in such cases, so that the evaporation of the turpentine would leave upon the plate the amount of paint originally intended.
The appearance of the completed series of test panels is shown on page 221.
Vehicles Used and Reasons for Avoidance of Japan Driers. The pigments used were selected with the view to securing as nearly as possible purity and strength, and as already noted, were out of the same lots used in making the preliminary laboratory tests on inhibitives. They were ground in a vehicle composed of two parts of raw linseed oil and one part of pure boiled oil. Paint is generally caused to dry rapidly by the use of japan or driers. These materials contain a large amount of metallic oxides which might have some effect in either exciting or retarding corrosion. To prevent the introduction of such a factor, these materials were not used in the test. The boiled oil, with its small percentages of metallic oxides, was sufficient, however, to cause the paints to dry in a short time after they were spread.
Testing Effect of Various Prime Coats. Some of the special tests made included a series of plates prime-coated with different inhibitive pigments, and these tests were designed to determine which pigments offer the best results for such work. These plates were all second-coated with the same paint. It is the opinion of the authors that any good excluding paint may be used whether it be inhibitive in action or not, provided the contact coat is inhibitive. If, however, both coats can be designed so as to have the maximum possible value from both these points of view, the best results would, of course, accrue. The only way such data can be obtained is by careful observation of the results of exposure tests.
Combination Formulas Tested. By selecting a series of pigments which in the water tests showed inhibitive tendencies, and properly combining these pigments into a paint, it was thought possible that a more or less inhibitive paint would be produced. If this proved to be the case, it would follow that the selection and introduction into a paint of the stimulative pigments would inevitably produce a paint unfit for use on iron or steel.
[232]Data on Application of Paints. The recorded data on the application of the paint to the panels is voluminous. There is presented herewith, however, the data on two of the paints.
| No. 2, Quick Process White Lead: | |||||||
| Sp. Gr. of pigment | 6.78 | ||||||
| Lbs. to gallon oil | 20.34 | ||||||
| Sp. Gr. of paint as received | 2.47 | ||||||
| Wt. of paint per gallon | 20.56 | ||||||
| Grams to panel | 62 | ||||||
| Condition of paint | Good | ||||||
| Working properties | Works easy | ||||||
| Drying | 24 hrs. all coats | ||||||
| 1 coat | Oct. | 26 | T 60 | B 29.94 | W. fair | ||
| 2 coat | Nov. | 3 | T 54 | B 30.23 | W. clear | ||
| 3 coat | Nov. | 7 | T 52 | B 29.66 | W. cloudy | ||
| No. 9, Orange Mineral (American): | |||||||
| Sp. Gr. of pigment | 8.97 | ||||||
| Lbs. to gallon oil | 26.91 | ||||||
| Sp. Gr. of paint as received | 2.97 | ||||||
| Wt. of paint per gallon | 24.74 | ||||||
| Grams to panel | 74.7 | ||||||
| Condition of paint | Good | ||||||
| Working properties | Smooth—no brush marks | ||||||
| Drying | Good | ||||||
| 1 coat | Oct. | 28 | T 58 | B 30.01 | W. cloudy | ||
| 2 coat | Nov. | 4 | T 65 | B 29.61 | W. cloudy | ||
| 3 coat | Nov. | 9 | T 58 | B 29.91 | W. clear | ||
Composition of Paints. The following table gives data regarding the composition, etc., of paints applied to the steel panels.
Results of Inspection. The results of an inspection of the steel test plates, made by Sub-committee D representing Committee D-1 of the American Society for Testing Materials, is herewith presented:
“On Wednesday, June 28, 1911, the second inspection of the Atlantic City Steel Test Panels, erected in October, 1908, was made by Sub-committee D of Committee D-1, this Committee having agreed to report upon the condition of the painted surfaces, leaving any report on the comparative corrosion of the various types of metal used in the test to Committee A-5 on the corrosion of iron.
| Pig- ment No. |
Name | Sp. Gr. of Pig- ment |
Wt. of Pigment to Gal. of oil Lbs. |
Sp. Gr. of Paint Rec’d |
Wt. of Paint per Gal. Lbs. |
Grams Paint to Panel at 900 Sq. ft. spread- ing rate |
|||||||||
| 1 | Dutch process white lead | 6 | .83 | 20 | .49 | 2 | .45 | 20 | .49 | 61 | .0 | ||||
| 2 | Quick process white lead | 6 | .78 | 20 | .34 | 2 | .47 | 20 | .34 | 62 | .0 | ||||
| 3 | Zinc oxide | 5 | .56 | 16 | .68 | 2 | .12 | 16 | .68 | 59 | .0 | ||||
| 4 | Sublimed white lead | 6 | .45 | 19 | .17 | 2 | .36 | 19 | .17 | 59 | .0 | ||||
| 5 | Sublimed blue lead | 6 | .39 | 19 | .17 | 2 | .42 | 19 | .17 | 61 | .0 | ||||
| 6 | Lithopone | 4 | .26 | 12 | .78 | 1 | .80 | 12 | .78 | 45 | .3 | ||||
| 7 | Zinc lead white | 4 | .42 | 13 | .26 | 1 | .96 | 13 | .26 | 49 | .4 | ||||
| 9 | American orange mineral | 8 | .97 | 26 | .91 | 2 | .97 | 26 | .91 | 74 | .7 | ||||
| 10 | Red lead | 8 | .70 | 26 | .10 | 2 | .93 | 26 | .10 | 73 | .6 | ||||
| 12 | Bright red oxide | 5 | .26 | 15 | .78 | 2 | .05 | 15 | .78 | 60 | .0 | ||||
| 14 | Venetian red | 3 | .1 | 9 | .30 | 1 | .52 | 9 | .30 | 38 | .0 | ||||
| 15 | Prince’s metallic brown | 3 | .17 | 9 | .51 | 1 | .50 | 9 | .51 | 37 | .7 | ||||
| 16 | Natural graphite | 2 | .60 | 7 | .80 | 1 | .37 | 7 | .80 | 34 | .4 | ||||
| 17 | Acheson graphite | 2 | .21 | 6 | .63 | 1 | .22 | 6 | .63 | 30 | .8 | ||||
| 19 | { | Lampblack | 1 | .82 | 1 | .82 | } | 1 | .60 | 1 | .82 | 40 | .2 | ||
| Barytes | 8 | .92 | 8 | .92 | |||||||||||
| 20 | Willow charcoal | 1 | .49 | 4 | .47 | 1 | .08 | 4 | .47 | 27 | .0 | ||||
| 21 | { | Gas carbon black | 1 | .85 | 1 | .39 | } | 1 | .67 | 1 | .39 | 50 | .7 | ||
| Natural barytes | 10 | .03 | 10 | .03 | |||||||||||
| 24 | French yellow ochre | 2 | .94 | 8 | .82 | 1 | .46 | 8 | .82 | 37 | .0 | ||||
| 27 | Natural barytes | 4 | .46 | 13 | .38 | 1 | .83 | 13 | .38 | 46 | .0 | ||||
| 28 | Precipitated barytes (blanc fixe) | 4 | .23 | 12 | .69 | 1 | .84 | 12 | .69 | 46 | .0 | ||||
| 29 | Calcium carbonate (whiting) | 5 | .48 | 8 | .22 | 1 | .37 | 8 | .22 | 34 | .5 | ||||
| 30 | Calcium carbonate precipitated | 2 | .56 | 7 | .68 | 1 | .35 | 7 | .68 | 34 | .0 | ||||
| 31 | Calcium sulphate (gypsum) | 2 | .33 | 6 | .99 | 1 | .25 | 6 | .99 | 31 | .4 | ||||
| 32 | China clay (kaolin) | 2 | .67 | 8 | .01 | 1 | .34 | 8 | .01 | 34 | .0 | ||||
| 33 | Asbestine (silicate of magnesium) | 2 | .75 | 8 | .25 | 1 | .38 | 8 | .25 | 34 | .7 | ||||
| 34 | American vermilion (chrome scarlet) | 6 | .83 | 20 | .49 | 20 | .49 | 64 | .5 | ||||||
| 36 | Medium chrome yellow | 5 | .88 | 17 | .64 | 17 | .64 | 67 | .1 | ||||||
| 39 | Zinc chromate | 3 | .57 | 10 | .71 | 1 | .57 | 10 | .71 | 39 | .2 | ||||
| 40 | Zinc and barium chromate | 3 | .45 | 10 | .35 | 1 | .58 | 10 | .35 | 40 | .0 | ||||
| 41 | Chrome green (blue tone) | 4 | .44 | 13 | .32 | 1 | .94 | 13 | .32 | 49 | .0 | ||||
| 44 | Prussian blue | 1 | .96 | 5 | .88 | 5 | .88 | 30 | .0 | ||||||
| 45 | Prussian blue | 1 | .93 | 5 | .79 | 5 | .79 | 34 | .5 | ||||||
| 48 | Ultramarine blue | 2 | .40 | 7 | .20 | 1 | .29 | 7 | .20 | 32 | .5 | ||||
| 49 | Zinc and lead chromate | 4 | .76 | 14 | .28 | 1 | .92 | 14 | .28 | 48 | .3 | ||||
| 51 | Magnetic black oxide | 15 | .00 | 1 | .92 | 15 | 48 | .3 | |||||||
| Composite Paints | |||||||||||||||
| 111 | Brown | ⎫ | Made from pigments that were inhibitive in the water test |
10 | .82 | 1 | .30 | 10 | .82 | 32 | .7 | ||||
| 222 | Black | ⎬ | 10 | .86 | 1 | .30 | 10 | .86 | 32 | .8 | |||||
| 333 | White | ⎪ | 14 | .52 | 1 | .74 | 14 | .52 | 43 | .8 | |||||
| 444 | Green | ⎭ | 12 | .77 | 1 | .53 | 12 | .77 | 38 | .6 | |||||
| 555 | Black | ⎫ | Made from pigments that were stimulative in the water test |
9 | .37 | 1 | .125 | 9 | .37 | 28 | . | ||||
| 666 | Brown | ⎬ | 11 | .74 | 1 | .41 | 11 | .74 | 35 | .5 | |||||
| 777 | White | ⎪ | 14 | .55 | 1 | .75 | 14 | .55 | 44 | . | |||||
| 888 | Green | ⎭ | 14 | .57 | 1 | .75 | 14 | .57 | 14 | .57 | |||||
[234]“According to the amount of rust apparent on the painted surfaces of the panels, as well as the degree of checking, chalking, scaling, cracking, peeling, loss of color, and other signs of paint failure shown, ratings were given each panel. The system of rating which took into consideration all the above conditions, was similar to the system used at the first inspection during 1910, when 0 (zero) recorded the worst results and 10 (ten) the best results.
“In Table No. 1 there is shown the rating accorded by each inspector to each panel, as well as an average for each panel.
| Panel No. |
Pigment | W. H. Walker |
P. H. Walker |
H. A. Gardner Chair- man |
C. Chap- man |
Aver- age |
||||||
| 1 | Dutch process white lead | 2 | 3 | 3 | 5 | 3.7 | ||||||
| 2 | Quick process white lead | 4 | 4 | 3 | 6 | 4.2 | ||||||
| 3 | Zinc oxide (XX) | 1 | 1 | ½ | 1 | 2 | ½ | 1.5 | ||||
| 4 | Sublimed white lead | 9 | 9 | ½ | 9 | 8 | ½ | 9.0 | ||||
| 5 | Sublimed blue lead | 9 | 9 | ½ | 9 | ½ | 7 | ½ | 8.8 | |||
| 6 | Lithopone | 2 | 1 | ½ | 2 | 3 | ½ | 2.2 | ||||
| 7 | Zinc lead white | 3 | 4 | 5 | 7 | 4.7 | ||||||
| 9 | Orange mineral | 9 | 9 | 9 | 6 | ½ | 8.3 | |||||
| 10 | Red lead | 9 | 9 | 9 | 6 | ½ | 8.3 | |||||
| 12 | Bright red oxide | 8 | ½ | 9 | 8 | 7 | 8.1 | |||||
| 14 | Venetian red | 7 | 9 | 7 | 9 | 8.0 | ||||||
| 15 | Prince’s metallic brown | 5 | 7 | ½ | 6 | 8 | 6.3 | |||||
| 16 | Natural graphite | 6 | 8 | 4 | 9 | ½ | 6.8 | |||||
| 17 | Artificial graphite | 5 | 7 | ½ | 4 | 7 | 5.9 | |||||
| 19 | Lampblack | 5 | 7 | ½ | 5 | 8 | 6.3 | |||||
| 20 | Willow charcoal | 9 | 8 | ½ | 9 | 9 | 8.8 | |||||
| 21 | Carbon black | 7 | 8 | ½ | 5 | 8 | ½ | 7.2 | ||||
| 24 | Yellow ochre (French) | 5 | 7 | 2 | 8 | 5.5 | ||||||
| 27 | Barytes (natural) | 1 | 1 | 1 | 0 | 0.7 | ||||||
| 28 | Barytes (precipitated) | 2 | 1 | ½ | 2 | 2 | 1.8 | |||||
| 29 | Calcium carbonate (whiting) | 0 | 0 | 0 | 0 | 0.0 | ||||||
| 30 | Calcium carbonate (precipitated) | 0 | 0 | 0 | 0 | 0.0 | ||||||
| 31 | Calcium sulphate (gypsum) | 1 | 1 | 1 | 3 | 1.7 | ||||||
| 32 | China clay (kaolin) | 6 | 6 | 7 | 6 | ½ | 6.3 | |||||
| 33 | Asbestine (magnes. silicate) | 5 | 4 | ½ | 6 | 5 | 5.1 | |||||
| 34 | American vermilion | 10 | 10 | 10 | 10 | 10.0 | ||||||
| 36 | Lead chromate | 7 | 7 | ½ | 8 | ½ | 8 | 7.7 | ||||
| 39 | Zinc chromate | 9 | 9 | 10 | 9 | ½ | 9.5 | |||||
| 40 | Zinc and barium chromate | 9 | 9 | ½ | 10 | 9 | ½ | 9.5 | ||||
| 41 | Chrome green (blue tone) | 10 | 10 | 10 | 9 | ½ | 9.8 | |||||
| 44 | Prussian blue, W. S | 9 | 9 | ½ | 9 | ½ | 9 | 9.0 | ||||
| 45 | Prussian blue, W. I | 8 | 9 | ½ | 8 | ½ | 8 | ½ | 8.5 | |||
| 48 | Ultramarine blue | 0 | 0 | 0 | 0 | 0.0 | ||||||
| 49 | Zinc and lead chromate | 10 | 9 | ½ | 10 | 9 | ½ | 9.7 | ||||
| 51 | Magnetic black oxide | 9 | 9 | ½ | 10 | 9 | ½ | 9.5 | ||||
| 111 | Brown composite paint | 7 | 9 | 9 | 9 | 8.5 | ||||||
| 222 | Black composite paint | 9 | 9 | 9 | 8 | ½ | 8.8 | |||||
| 3333 | White composite paint | 4 | 4 | 7 | 3 | 4.5 | ||||||
| 444 | Green composite paint | 5 | 7 | 7 | 8 | 6.7 | ||||||
| 555 | Black composite paint | 9 | 9 | 6 | 9 | 8.2 | ||||||
| 666 | Brown composite paint | 8 | 8 | 6 | 9 | 7.7 | ||||||
| 777 | White composite paint | 7 | 10 | 5 | 7 | 7.2 | ||||||
| 888 | Green composite paint | 7 | 8 | 8 | 9 | 8.0 | ||||||
| 2000 | 1 coat zinc chromate | } | 8 | 8 | ½ | 8 | 8 | 8.1 | ||||
| 1 coat iron oxide excluder | ||||||||||||
| 3000 | 1 coat lead chromate | 7 | 8 | 7 | 7 | ½ | 7.3 | |||||
| 4000 | 1 coat red lead | } | 7 | 8 | ½ | 8 | 7 | ½ | 7.7 | |||
| 1 coat iron oxide excluder | ||||||||||||
| 100 | Straight carbon black paint with turps and drier | 5 | 8 | ½ | 4 | 8 | ½ | 6.5 | ||||
| 90 | Straight lampblack paint with turps and drier | 5 | 7 | 3 | 8 | 5.7 | ||||||
| 5555 | Coal tar paint over red lead | 4 | 8 | 2 | 7 | 5.2 | ||||||
| 1000 | Chrome resinate in oil (1 coat) | 1 | 0 | 0 | 2 | 0.7 | ||||||
| 1 plate | 3 coats boiled linseed oil | 1 | 0 | 1 | 4 | 1.5 | ||||||
“In Table No. 2 there is shown the rating obtained by those panels which were considered by the committee as meriting from 8 to 10, and having given the best all-round service.
| Plate | Pigment | Average |
| 34 | American vermilion (basic chromate of lead) | 10.0 |
| 41 | Chrome green | 9.8 |
| 49 | Lead and zinc chromate | 9.7 |
| 39 | Zinc chromate | 9.5 |
| 40 | Zinc and barium chromate | 9.5 |
| 51 | Black oxide of iron | 9.5 |
| 4 | Sublimed white lead | 9.0 |
| 44 | Prussian blue | 9.0 |
| 5 | Sublimed blue lead | 8.8 |
| 20 | Willow charcoal | 8.8 |
| 222 | Composite paint | 8.8 |
| 45 | Prussian blue | 8.5 |
| 111 | Composite formula | 8.5 |
| 9 | Orange mineral | 8.3 |
| 10 | Red lead | 8.3 |
| 555 | Composite paint | 8.2 |
| 12 | Bright red oxide of iron | 8.1 |
| 2000 | 1 coat zinc chromate; 1 coat iron oxide | 8.1 |
| 14 | Venetian red | 8.0 |
| 888 | Composite paint | 8.0 |
Comparison of Results. It is of interest to compare with Table 2 of the above report, Table 2 of the 1910 report of Committee U of the American Society for Testing Materials. Both charts show the highly inhibitive pigments to be in the lead.
(Only resistance to corrosion was considered, and only
pigments which were common
to both tests are included)
| No. | Pigment | Average |
| 34 | American vermilion (chrome scarlet) | 9.8 |
| 41 | Chrome green (blue tone) | 9.7 |
| 40 | Zinc and barium chromate | 9.7 |
| 5 | Sublimed blue lead | 9.6 |
| 4 | Sublimed white lead | 9.5 |
| 49 | Zinc and lead chromate | 9.5 |
| 39 | Zinc chromate | 9.4 |
| 12 | Bright red oxide | 9.3 |
| 44 | Prussian blue (water stimulative) | 9.2 |
| 16 | Natural graphite | 9.1 |
| 9 | Orange mineral (American) | 9.0 |
| 36 | Medium chrome yellow | 9.0 |
| 2 | White lead (quick process) | 8.9 |
| 20 | Willow charcoal | 8.8 |
| 45 | Prussian blue (water inhibitive) | 8.8 |
| 1 | White lead (Dutch process) | 8.7 |
| 10 | Red lead | 8.7 |
| 7 | Zinc lead white | 8.0 |
The writer has recently made a careful inspection of the panels painted with single pigment paints, and has made the following brief summary of the characteristic appearance of each.
Panel No. 1—Dutch Process White Lead. The excessive chalking which took place began to disappear at the end of a year, being washed away by the rains and carried away by the winds, so that there was left upon the surface but a thin coating of pigment, insufficient to give good protection. Slight corrosion was apparent beneath the film.
Panel No. 2—Quick Process White Lead. In the same condition as Panel No. 1.
Panel No. 3—Zinc Oxide. Panel covered with thin lateral streaks of rust, due to the admittance of moisture in cracks caused by brittleness of film. Result doubtless due to insufficient amount of oil used with pigment. Removal of film shows steel very bright except where cracks have formed.
Panel No. 4—Sublimed White Lead. Although sublimed white lead chalked very heavily, the chalked pigment seemed to[237] be tenacious and adhered to the plate, presenting an excellent surface with absence of rust. Film of good color and quite elastic.
Panel No. 5—Sublimed Blue Lead. In same condition as Panel No. 4, but color has slightly faded.
Panel No. 6—Lithopones. Lithopone was early destroyed, as is usual with this pigment when used alone on exterior surfaces. It became rough and discolored, presenting a very blotchy appearance and disclosed the formation of rust working through the film.
Panel No. 7—Zinc Lead White. In general good condition with the exception of the color, which is slightly dark. Medium chalking was apparent but only very slight corrosion appeared.
Panel No. 9—Orange Mineral. In excellent condition, showing a good firm surface with no checking or corrosion apparent. Shortly after exposure the film became covered with a white coating of carbonate of lead, which indicates action of the red lead with the carbonic acid of the atmosphere. Removal of this white coating with water discloses the brilliant color of the unaffected portion of the red lead.
Panel No. 10—Red Lead. In same condition as Panel No. 9.
Panel No. 12—Bright Red Iron Oxide. In general good condition. Film intact and unfading in color.
Panel No. 14—Venetian Red. Similar to Panel No. 12, but slight corrosion apparent beneath, in localized spots, and film showing slight wart-like formations.
Panel No. 15—Prince’s Metallic Brown. Similar to Panel No. 14.
Panel No. 16—Natural Graphite. Deeply pitted in spots, showing bulbous eruptions, indicating the stimulative nature of this pigment.
Panel No. 17—Artificial Graphite. In same condition as Panel No. 16.
Panel No. 19—Lampblack and Barytes. Although the film seems to be intact, there are apparent abrasions of the surface showing stimulative corrosion effects of a pronounced nature.
Panel No. 21—Carbon Black and Barytes. In same condition as Panel No. 19.[238]

Corrosion Pits on Graphite Panel

Rust on Stripped Graphite Film

Section of Wire Painted with a Stimulative Carbonaceous Paint

Corroded and Pitted Surface of Plate Painted with Stimulative Paint
The longevity of lampblack and carbon black paint films when applied to wood has been attributed to the slow drying nature of these pigments when mixed with oil. It is assumed that they have the property of keeping the oil in a semi-drying condition, which will not disintegrate as early as when the oil is thoroughly dried to linoxyn. If this is true, it would seem advisable to use with hard-drying pigments, a proportion of some oil that is semi-drying in nature or one which will leave a film not too hard. Soya bean oil, wood oil, and fish oil present themselves as candidates for such use. How they will work in practice, however, is a question not yet determined. On the other hand, it is well known that these pigments require enormous quantities of oil in order to grind to a working consistency, and it is possible that[242] the life of such coatings is due rather to the property of these pigments, of taking up large quantities of oil, than to their effect upon the slow drying of oil. Excessive oil carrying, however, should be avoided, as shown by the early failure and pitting of those carbon black and lampblack paints ground with very large quantities of oil, as is the usual practice. When these carbon and lampblack pigments were ground with barytes (which is a heavy pigment and requires only about 9 pounds of oil to 100 pounds of pigment, as against 175 pounds of oil to 100 pounds of lampblack), it was found that the lampblack and carbon black paints were reinforced and made more suitable for actual practice. The stimulative nature of these black pigments, however, asserted itself in both cases, and large pittings and eruptions were evident at the end of a year. Carbon black, lampblack, graphite, or any other good conductor of electricity should never be placed next to the surface of iron. They are good as top-coatings, but not as prime-coaters. Some pigments are stimulators of corrosion, because they contain water-soluble impurities that hasten the rusting, while others, like graphite, hasten it simply because, being good conductors, they stimulate surface electrolysis.
Panel No. 20—Willow Charcoal. In excellent condition throughout. Presence of small quantities of potash may be responsible for the inhibitive nature of this black pigment.
Panel No. 24—Ochre. While the film seems intact, it has a very mottled appearance and examination shows eruptions of rust through the film, in several places.
Panel No. 27—Natural Barytes. Within a year the film became pin-holed, and corrosion was apparent. At the end of three years very little of the pigment was left upon the plate, having chalked and scaled off. Barytes has proved its usefulness as a constituent of a combination type of paint, but it should not be used alone.
Panel No. 28—Blanc Fixe. In the same condition as Panel No. 27, but slightly more chalking and disintegration was shown.

Panel Painted with Blanc Fixe. Right Side Stripped of Paint to Show Corrosion

Scaled Whiting Films
Chemically Active Pigments and Their Effect After Eighteen Months’ Wear

Plate Showing Effect of Chemically Active Pigments on Oil after One Year’s Wear
Panel No. 29—Whiting. Plates coated with calcium carbonate or whiting in oil presented a very fair appearance at the start of the test, but they soon began to chalk and disintegrate. It is well known that whiting, being alkaline, has the property of acting on oil and causing its early disintegration by saponification. As a matter of fact, six months after the whiting plates were exposed, crumbling of the surface appeared, and twelve months was sufficient for the total destruction of the paint. At this time the rusted surface of the plates which had been painted with calcium carbonate, seemed not to rust as fast as those plates which were exposed without paint coatings, and the rust which had formed appeared to be of an even, fine texture. On the lower left-hand corner of these plates had been lettered the figures “29” and “30,” using lampblack in oil. One of the most remarkable things which appears on the fence to-day is the perfect condition of these lampblack letters over their priming coat of calcium carbonate, standing out in clear relief against the rusted metal. This test would suggest, therefore, that if the surface of metal is properly protected with a pigment which[246] is slightly alkaline or inhibitive in nature, and then topped with a good weather-resisting material, such as lampblack, graphite or carbon black, good results would be obtained. Further tests will be made to determine the value of this suggestion.
Panel No. 30—Precipitated Calcium Carbonate. Showed more rapid destruction than Panel No. 29.

Corrosion Adhering to Film Stripped from Panel Painted with Gypsum (Calcium Sulphate)
Panel No. 31—Calcium Sulphate. Under the paint film of gypsum, rust soon appeared, showing that the film was not a good excluder of moisture. Although the film remained intact, rusting progressed throughout the test and considerably darkened the color of the paint.
Panel No. 32—China Clay. This pigment gave excellent service for eighteen months. Afterwards indications of corrosion were shown, and apparent breakdown of the film was indicated.
 |
||
| China Clay |
Asbestine | Gypsum |
Panel No. 33—Asbestine. In the same condition as Panel No. 32.

Excellent Surface shown by American Vermilion after nearly Four Years’ Exposure
Panel No. 34—American Vermilion. This pigment has given perfect protection to the plates. The film is strong and elastic, and upon removal reveals the bright steel. No chalking, checking, discoloration, or other signs of paint failure are shown. It would appear that the inhibitive characteristics of this pigment are pronounced, and it promises to give efficient service for several years more.
Panel No. 36—Lead Chromate. This panel is in generally fair condition, but slight checking is shown.

Perfect Condition of Plate Painted with Zinc Chromate; One Half Stripped. (Negative cracked)
Panel No. 39—Zinc Chromate. This panel is in condition similar to Panel No. 34, presenting a perfect appearance, with decided maintenance of color, elasticity of film, and freedom from any bad characteristics. It has proved to be one of the highest type rust inhibitive pigments.
Panel No. 40—Zinc-and-Barium-Chromate. Although the color of this pigment is not very pleasing, it has proved itself to be the equal of zinc chromate in its protective value.
Panel No. 41—Chrome Green. In excellent condition. Presents an appearance similar to Panels Nos. 34 and 39. Its surface is perfect and will doubtless give service for many years.
Panel No. 44—Prussian Blue. This panel stands forth as the most wonderful moisture-excluder in the whole test, its surface presenting an appearance similar to a varnished plate, even after three years’ exposure. Action between the pigment and the oil, resulting in the formation of iron linoleate, may account for this property.
Panel No. 45—Prussian Blue. In same condition as Panel No. 44.
Panel No. 48—Ultramarine Blue. Soon after this test was exposed, early vehicle decay and excessive chalking were observed. The admittance of moisture may have caused the formation of acid with the sulphur content of the pigment, which would account for the rapid corrosion which followed. It is of a pronounced stimulative type. The effect of stimulative under-coatings is well shown on some special plates on the fence, which when received were not pickled before painting, but had upon their surfaces the ordinary coating of mill scale. Over this had been stencilled in a triangular form the trade mark of the manufacturer. The stencilling material was made of ultramarine blue. When these plates were painted with some of the special paints, and exposed, the stimulative nature of the ultramarine blue began to assert itself, and within a short time, wherever the stencil marks were located, signs of rust began to appear through the coatings of top paint which had been applied. Corrosion under these stencil marks became so great that the trade mark was plainly outlined in letters of rust. This would seem to be final proof that pigments of a stimulative nature should never be used for the priming of iron and steel.
Panel No. 49—Zinc-Lead Chromate. In excellent condition throughout, with a smooth surface and showing no corrosion. Stands in the same class as Panels Nos. 34 and 39.

Effect of Stimulative Paint. Manufacturer’s Trade Mark Stencilled on Bare Metal in Triangular Form, showing Through Subsequent Paint Coating
Panel No. 51—Black Magnetic Oxide of Iron. In excellent condition.
Decoration and Sanitation. The proper decoration of the interior of dwellings and public buildings has become of even greater importance than the protection and decoration of exteriors. There is, moreover, an increasing demand for harmonious effects and the production of more sanitary conditions than have prevailed in the past. Up until a few years ago a great variety of wall papers of more or less pleasing appearance were almost exclusively used for the decoration of walls in the interior of buildings, and their application was commonly considered the most effective means of wall decoration. There seems to be no question, however, that the use of wall paper is steadily decreasing, and that the art of interior decoration is undergoing a transition to the almost universal use of paint.
Modern progress demands the maintenance of sanitary conditions for the benefit of the public welfare, and there is no doubt that from the standpoint of sanitation and hygiene, properly painted wall surfaces are far superior to papered walls. There is an abundance of evidence which shows that dust germs may easily be harbored, and thus disease transmitted from wall paper. In the tenement houses, which are common to the larger cities, and to a lesser extent in the dwellings found in smaller communities, where tenants are more or less transient, the continued maintenance of sanitary conditions presents a difficult problem. Infectious and epidemic illnesses generally leave behind bacilli of different types, which may find a culture medium in the fibrous and porous surfaces presented by wall paper, backed up as they invariably must be by starch, casein, or other organic pastes. Occasionally the restrictions of local boards of health provide in such events for proper fumigation, but too often no precautions are taken to destroy the disease germs which are caught in the dust which collects on wall paper. As a rule, both tenant and landlord are oblivious to all conditions which cannot be readily seen or detected. Burning sulphur, one of the most effective means of fumigation, will generally cause bleaching and consequent fading of the delicate colors used in printing the designs upon wall paper. Washing of the paper with antiseptic solutions will destroy its adhesiveness to the plaster and often cause bulging and general destruction.
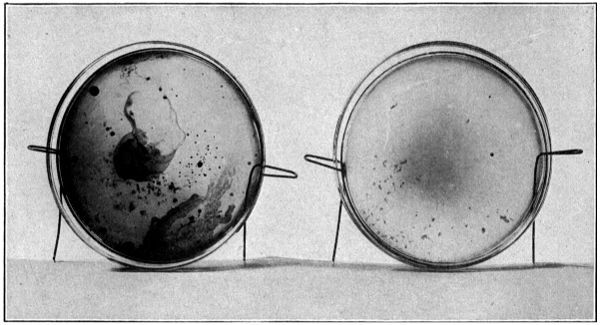 |
|
| Heavy Colonies of Bacteria Developing in Agar Jelly Treated with Washings from Wall Paper | Practically no Development of Bacterial Colonies in Agar Jelly Treated with Washings from Sanitary Wall Paint |
Hospital Practice. In hospitals, where it is necessary to maintain sanitary conditions, the walls are invariably painted, and requirements should demand the use of paints which can be washed frequently, so that there will be no possibility of uncleanliness. Inquiry made of a prominent surgeon[38] connected with one of the large metropolitan hospitals substantiated the writer’s findings regarding the greater sanitary value of wall paints, and brought forth the information that in hospitals under construction provision had been made for the finishing of walls so that a hard, non-absorbent, and washable surface might be obtained. The same authority stated that the common practice, in apartments and tenements, of covering the old wall paper over with a layer of new each time a tenant moved in, should be condemned, and that from a hygienic standpoint the use of sanitary wall paints should be advocated in all dwellings as well as public buildings.
[38] Dr. F. F. Gwyer, Cornell Uni. Med. Col., New York City.
If such conditions are maintained in hospitals, where special attention is paid to sanitation, it would appear that similar precautions should be equally as necessary in public buildings and in dwellings—wherever, in fact, people congregate or live.
Sanitary Wall Paints. There have recently appeared in trade a number of wall paints composed of non-poisonous pigments ground in paint vehicles having valuable waterproofing and binding properties, and of a nature to produce the flat or semi-flat finish that has become so popular. Such paints produce a sanitary, waterproof surface, which permits of frequent washing. By their use it is possible to secure a more permanent and a wider range of tints than can be obtained with wall paper, as they are produced in a myriad of shades, tints and solid colors, from which any desired combination may be selected.[255] On the border or on the body of walls decorated with such paints, attractive stencil designs, which bring out in relief the color combinations, may be applied.
For the decoration of chambers and living rooms, delicate French grays, light buffs, cream tints and ivory whites may be used, while in the library and other rooms richer and more solid colors, such as greens, reds, and blues, may be harmoniously combined.
Defects of Wall Paper. It recently occurred to the writer to investigate the conditions which obtain in many apartment houses in the larger cities. Inspection of a number of such places, in which wall paper had been exclusively used on the walls, showed generally bad conditions; bulging of the surfaces, caused by dampness in the walls, which had loosened up the binder, as well as peeling and dropping of the paper from the ceilings, were frequently observed. In many cases a shabby appearance was shown, accompanied by an odor which suggested decomposition of the paste binder used on the paper. The writer was impressed with the fact that such conditions could easily be avoided by the very simple expedient of using properly manufactured wall paints, which are so easily made dustproof and waterproof.
Samples of wall paper, which had been applied to plastered walls for a year or more, were obtained, and examination under the microscope showed a most uncleanly surface. Cultures were made of these samples, and bacilli of different types were developed in the culture medium in a short time.
Experimental Evidence. That the above conditions could not have existed, had proper wall paints been used, seemed doubtless, and suggested a carefully conducted experiment to prove the relative sanitary values of wall paper and wall paints. A large sheet of fibre board, such as is occasionally used to replace plastered walls, was painted on one side with a high-grade wall paint, three-coat work. A similar sheet was papered on one side with a clean, new wall paper. These test panels were placed where unsanitary conditions, such as dampness, foul odors, and a scarcity of air were present. After a short period of exposure, the panels were taken to the bacteriological laboratory and a small section of the painted surface, about two inches square, as well as a small section of the papered surface of similar size, were[256] removed and used for making cultures. In each case the surface of the section under test was washed with 100 c.c. of distilled, sterilized water. The washings which dripped from the surface were collected in a graduated flask. One c.c. of the washings was used in each case, admixed with bouillon and again with agar-agar. The enormous development of bacteria in the bouillon, treated with the washings from the wall-papered surface, was sufficient evidence to convince one of the greater sanitary value of the wall paint, the washings from which gave a culture practically free from bacteria. The colonies of bacteria shown in the petri-dish test made of the washings from wall paper further supports these findings. It will be noticed that the tests made from the washings of the wall paint show practical absence of bacteria, and was clear, as was the bouillon-solution test of the paint. The washings from the wall paper showed active development of bacteria, both in the bouillon and agar tests.
 |
|
| Development of Bacteria in Bouillon Solutions | |
| Note Practical Freedom of Bacteria in Clear Bouillon Solution Treated with Washings from Sanitary Wall Paint | Note Milky Appearance of Solution Due to Heavy Development of Bacteria in Bouillon Treated with Washings from Wall Paper |
[257]From the Conservation Standpoint: It would be of interest to sum up in figures the acreage and cordage of wood that annually is transformed into pulp for the manufacture of wall paper. Unfortunately, there are no available statistics on this subject. It is clear, however, that from the standpoint of conservation the use of wall paints should take precedence over the use of wall paper.
| PAGE | ||
| Abrasion, apparatus for determining resistance to, | 153 | |
| Acid reacting compounds, | 215 | |
| Actinic light tests, | 112 | |
| Adhesive power of Paint Coating, | 104 | |
| Aluminum Silicate, | 62 | |
| American Vermilion, | 64 | |
| Analogies of Paint and Concrete manufacture, | 94 | |
| Analyses of Averages in Atlantic City steel paint test, | 235, 236 | |
| Corn Oil, | 16 | |
| Cottonseed Oil, | 15 | |
| Debloomed Mineral Paint Oil, | 18 | |
| Iron Oxide Pigments, table, | 63 | |
| Linseed Oil, | 7 | |
| Menhaden Oil, | 14 | |
| Oils used in Washington tests, | 211 | |
| Petroleum Spirits, | 20 | |
| Rosin Oil, | 16 | |
| Soya Bean Oil, | 8 | |
| Sunflower Oil, | 15 | |
| Tung Oil, | 12 | |
| Whale Oil, | 14 | |
| Wood Turpentine, | 19 | |
| Asbestine, | 55 | |
| Atlantic City fence tests, | 107 | |
| steel paint tests, | 228-235 | |
| Checking, | 122 | |
| Gloss, | 122 | |
| Hiding power, | 122 | |
| inspection of, | 114 | |
| Methods used, | 114 | |
| Results, | 124 | |
| Auto-electrolysis, | 220 | |
| Bacteria in wall paper, | 256 | |
| Barium Sulphate, | 55 | |
| Barytes, | 55 | |
| and Silica Paints in Pittsburg tests, | 172 | |
| Basic Carbonate-White Lead, | 42 | |
| Benzine, | 20 | |
| Benzol, | 20 | |
| [260]Blanc Fixe, | 60 | |
| Blue Lead, Sublimed, | 47 | |
| Blue Paint for concrete wall, formula, | 215 | |
| Blue paints in Pittsburg tests, | 142 | |
| Boiled Linseed Oil, | 2 | |
| Driers in, | 28 | |
| Bone Black, | 66 | |
| Calcium Carbonate, | 60 | |
| Calcium Sulphate, | 60 | |
| Carbon Black, | 66 | |
| Cause of rust in steel work, | 220 | |
| Chalking test for laboratory, | 149 | |
| Checking and cracking in Pittsburg tests, | 166 | |
| Checking, in Atlantic City tests, | 122 | |
| China Clay, | 62 | |
| Chrome Green, | 66 | |
| Chrome Yellow, | 64 | |
| Coatings for cement and concrete, | 214 | |
| Colored formulas in North Dakota tests, | 190 | |
| Colors, report of, in Pittsburg tests, | 139 | |
| Combination formulas in inhibitive paints, | 231 | |
| Composite formulas in North Dakota tests, | 190 | |
| in Pittsburg tests, | 142 | |
| Composition of paints, in steel test, | 232 | |
| Conclusion from Pittsburg tests, | 144 | |
| Concrete primer formula, | 218 | |
| Constants of Pine Oil, | 18 | |
| Pure Gum Turpentine, | 19 | |
| Co-operative tests of Driers, | 29-41 | |
| Corn Oil, | 16 | |
| Cottonseed Oil, | 15 | |
| Damp-proofing and Waterproofing, | 214 | |
| Decay of Lithopone paints, | 124 | |
| Decomposition of Paint, | 122 | |
| Driers, Co-operative tests of, | 29-41 | |
| in Boiled Oil, | 28 | |
| Tests of Manganese, Lead and Combination, tables, | 24-25 | |
| Drying Properties of Oil, | 1, 26, 27 | |
| Elasticity and Strength of Paint Coating, | 102 | |
| Fence tests of paints, | 105 | |
| Supervision of, | 112 | |
| Film sectioning, | 87 | |
| Film testing results, table, | 80 | |
| Filmometers, | 74-79 | |
| [261]Formula for Blue Paint for concrete wall, | 215 | |
| Concrete primer, | 218 | |
| Para Red Paint for concrete wall, | 217 | |
| Formulas of Atlantic City fence test, | 108 | |
| Tennessee tests, | 202, 204 | |
| Washington tests, | 208, 211 | |
| Fume Pigments Paints in Pittsburg tests, | 173 | |
| General results of Atlantic City tests, | 128 | |
| Gloss, in Atlantic City tests, | 122 | |
| Graphite, | 66 | |
| Green paints in Pittsburg tests, | 142 | |
| Grinding Pigments, | 87 | |
| Gums as moisture resisters, | 84 | |
| Gypsum, | 60 | |
| Hailstorm, effects of in North Dakota tests, | 185 | |
| Hospital, painting practice, | 254 | |
| House paint tests in North Dakota, | 196 | |
| Hydrocarbon Oils, | 16 | |
| Imperviousness of paint coating, | 100 | |
| Indian Red, | 62 | |
| Inert Pigments, use of, | 99 | |
| Inhibition of rust, | 222 | |
| Iodine Values of Linseed and Mixed Oils, table, | 8 | |
| Iron Oxide Paints, | 64 | |
| Japan driers in tests on steel, | 231 | |
| Laboratory tests, panels for, | 149 | |
| Lampblack, | 66 | |
| Laws of Paint Making, | 93 | |
| Lime action on paint, | 214 | |
| Linoxyn, | 21 | |
| Linseed Oil, boiled, | 2 | |
| Chemical action of pigments upon, | 91 | |
| Table of Analyses of Various Types of, | 7 | |
| tests of Driers with, | 24, 25 | |
| Lithopone, | 53 | |
| paint in Pittsburg tests, | 136 | |
| tests at Atlantic City, | 124 | |
| Lumbang Oil, | 12 | |
| Magnesium Silicate, | 55 | |
| Manufacturing Barytes, | 55 | |
| Blue Lead, | 47 | |
| [262]Manufacturing Bone Black, | 66 | |
| Paint Pigments, | 42-68 | |
| White Lead, | 42 | |
| Menhaden Oil, | 12 | |
| Constants of, table, | 14 | |
| Metallic Brown, | 62 | |
| Microscope, use of in paint laboratory, | 86 | |
| Microscopic examination of paint, preparation for, | 86 | |
| measurements of paint sections, | 89 | |
| Mineral Black, | 68 | |
| Oils, | 17 | |
| Moisture Absorption, tests in, | 84 | |
| experiments with various Pigments, | 83 | |
| North Dakota Paints tests, | 182 | |
| test fence, | 105 | |
| report of, table, | 193-195 | |
| Ochre, | 62 | |
| Oil and Thinner tests, | 202 | |
| Oil, Corn, | 16 | |
| Cottonseed, | 15 | |
| Effects of Pigments on, | 90 | |
| Linseed, | 1 | |
| Linseed, Analyses of Various Types of, table, | 7 | |
| Linseed, Iodine Values of, table, | 8 | |
| Linseed, Tests of Driers with, | 24, 25 | |
| Lumbang, | 12 | |
| Menhaden, | 12 | |
| Menhaden, Constants of, table, | 14 | |
| Perilla, | 21 | |
| Pine, | 18 | |
| Rosin, | 16 | |
| Soya Bean, and Driers, table, | 9 | |
| Soya Bean, | 7 | |
| Chemical Characteristics of, table, | 8 | |
| Sunflower, | 14 | |
| Tung, | 9 | |
| Whale, | 14 | |
| Oils, Constants and Characteristics of, | 1 | |
| Drying properties of, | 1, 26 | |
| Hydrocarbon, | 16 | |
| In Washington paint tests, | 210 | |
| Iodine Value of Linseed and Mixed, table, | 8 | |
| Mineral, | 17 | |
| Moisture resistance of, | 84 | |
| Oxygen Absorbing qualities, | 21 | |
| Oils, Outline of tests of paints on concrete walls, | 216 | |
| Oxygen Absorption in Oils, | 21 | |
| Paint Coating, Adhesive power of, | 104 | |
| Elasticity and Strength of, | 102 | |
| imperviousness of, | 100 | |
| decomposition of, | 122 | |
| films, action of water upon, | 223 | |
| permeability of, | 71 | |
| Testing machine, | 74 | |
| preparation of, | 70 | |
| in Hospitals, | 254 | |
| making, Laws of, | 93 | |
| Perry’s Principles of, | 100 | |
| pigments, | 42-69 | |
| pigments, properties of, | 42 | |
| preparation for microscopic examination of, | 86 | |
| tests at North Dakota Experiment Station, | 105 | |
| at Washington, | 207-213 | |
| supervisors of, | 113 | |
| woods used on, | 124, 135 | |
| Painting steel plates for tests, | 230 | |
| Paints for cement and concrete surfaces, | 214 | |
| composition of in steel test, | 233 | |
| hiding power of, | 111 | |
| sanitary value of, | 252 | |
| Panels for laboratory tests, | 149 | |
| Para Red formula for concrete wall, | 217 | |
| Paranitraniline paints in Pittsburg tests, | 140 | |
| Paranitraniline Red, | 64 | |
| Perilla Oil, | 21 | |
| Perry’s analogies of paint and concrete manufacture, | 99 | |
| principles of Paint Making, | 100 | |
| Petroleum Spirits, | 20 | |
| Photomicrographs, | 89, 165 | |
| Pigment contention, the, | 105 | |
| grinding, | 87 | |
| Pigments, | 42-69 | |
| as stimulators of rust, | 223 | |
| Chemical action of upon Linseed Oil, table, | 91 | |
| Effects of on Oil, | 90 | |
| inert, use of, | 99 | |
| moisture experiments with, table, | 83 | |
| percentages of Oil required for grinding, | 68 | |
| re-enforcing, | 89 | |
| report of results of steel paint tests, | 236-251 | |
| Water resistance of, | 81 | |
| [264]Pine Oil, | 18 | |
| Pittsburg fence tests, | 107 | |
| Polar Micro-Examinations and Photomicrographs, | 89 | |
| Primer for concrete, | 218 | |
| Properties of Paint Pigments, | 42 | |
| Prussian Blue, | 66 | |
| Red Lead, | 64 | |
| Reductions used in fence tests, | 111 | |
| Re-enforcing Pigments, | 89 | |
| Results of new test at Atlantic City test fence in 1910, table, | 178-181 | |
| Pittsburg tests, | 135 | |
| steel test plates, | 232 | |
| Rosin Oil, | 16 | |
| Rust, cause of in steel work, | 220 | |
| inhibition of, | 222 | |
| stimulation of, | 223 | |
| Sanitary value of paints, | 252 | |
| wall paints, | 254 | |
| Sienna, | 62 | |
| Silex, | 60 | |
| Silica, | 60 | |
| Silica and Barytes Paints in Pittsburg tests, | 172 | |
| Solvent Naphtha, | 20 | |
| Soya Bean Oil and Driers, table, | 9 | |
| Chemical Characteristics of, table, | 8 | |
| Steel Paint test, rating report, | 234 | |
| reports on pigments used, | 236-251 | |
| Steel paint, result of tests at Atlantic City, | 234, 235 | |
| Steel, preparation of for paint tests, | 228 | |
| water contact and paint, | 224 | |
| Structural steel paint tests, | 220 | |
| Sublimed Blue Lead, | 47 | |
| Sublimated White Lead, | 46 | |
| Suction varnish, | 215 | |
| Sunflower Oil, | 14 | |
| Constants of, table, | 15 | |
| Supervisors of paint tests, | 113 | |
| Table Analysis of Averages in Atlantic City Steel Paint test, | 235, 236 | |
| Analyses of Corn Oil, | 16 | |
| Analyses of Debloomed Mineral Paint Oil, | 18 | |
| Analysis of Iron Oxide Pigments, | 63 | |
| Analyses of Oils used in Washington tests, | 211 | |
| Analyses of Petroleum Spirits, | 20 | |
| Analyses of Rosin Oil, | 16 | |
| [265]Table Analyses of various types of Linseed Oil, | 7 | |
| Analyses of wood Turpentine, | 19 | |
| Atlantic City test fence formula, | 108 | |
| Chemical Characteristics of Soya Bean Oil, | 8 | |
| Comparative spreading rates of White Paint in Pittsburg tests, | 148 | |
| Composition of Blue Lead, | 49 | |
| Composition of paints in Atlantic City Steel test, | 233 | |
| Constants of Cottonseed Oil, table, | 15 | |
| Constants of Menhaden Oil, | 14 | |
| Constants of Pine Oil, | 18 | |
| Constants of Sunflower Oil, | 15 | |
| Constants of Whale Oil, | 14 | |
| Co-operative drying tests, | 32-41 | |
| Excluding tests for moisture absorbed, | 84 | |
| Fineness for grinding pigments, | 87 | |
| Formulas in Tennessee tests, | 204 | |
| Iodine Value of Linseed Oil and Mixed Oils, | 84 | |
| Moisture experiments with various pigments, | 83 | |
| Paint section measurements under microscope, | 89 | |
| Percentages of Oil required for grinding various dry pigments, | 68 | |
| Permeability of Paints, | 72 | |
| Ratings of Atlantic City Steel Paint test, | 234 | |
| Report of North Dakota test fence, | 193-195 | |
| Results of Atlantic City test fence, | 130, 131 | |
| Results of new tests at Atlantic City test fence in 1910, | 178-181 | |
| Results of second annual inspection Atlantic City test fence, | 133 | |
| Results of second annual inspection in Pittsburg tests, | 145 | |
| Showing action of various pigments upon Linseed Oil, | 91 | |
| Soya Bean Oil and Driers, | 9 | |
| Tests of Linseed Oil and Manganese, Lead and Combination Driers, | 24, 25 | |
| Talcose, | 55 | |
| Tennessee Paint tests, | 201-206 | |
| Test Fences in Paint Experiments, | 105 | |
| at Atlantic City, | 114-134 | |
| at Pittsburg, | 135-148 | |
| at Washington, | 207-213 | |
| Cement and concrete, | 214 | |
| in Tennessee, | 201-206 | |
| laboratory, chalking, | 149 | |
| North Dakota, | 182 | |
| of Oil and Thinners, | 202 | |
| of various pigments in steel paint, | 236-251 | |
| panel sections for, | 149 | |
| Structural steel paints, | 220 | |
| Water pigment, | 226 | |
| Thinner, Wood Turpentine as a, | 202 | |
| Tung Oil, | 9 | |
| [266]Tung Varnishes, | 11 | |
| Turpentine, | 18 | |
| Ultramarine Blue, | 66 | |
| Umber, | 62 | |
| Varnishes from Tung Oil, | 11 | |
| Vermilion, American, | 64 | |
| Wall paints, | 252 | |
| Wall paper, defects of, | 255 | |
| Washington Paint tests, | 207-213 | |
| Water, action of upon paint films, | 223 | |
| contact with steel and paint, | 224 | |
| resistance of Pigments, | 81 | |
| tests, | 226 | |
| Water-pigment tests, | 226 | |
| Waterproofing and damp-proofing, | 214 | |
| Whale Oil, | 14 | |
| White Lead, Basic Carbonate, | 42 | |
| Basic Sulphate, | 46 | |
| Mild Process, | 46 | |
| Quick Process, | 45 | |
| in Pittsburg tests, | 139 | |
| in North Dakota tests, | 190 | |
| Paints, checking in Pittsburg tests, | 172 | |
| processes of manufacture of, | 43-46 | |
| Whiting, | 60 | |
| Wood Turpentine, | 19 | |
| experiments with as a thinner, | 202 | |
| Woods used in paint tests, | 124, 135 | |
| Zinc Chromate, | 64 | |
| Zinc Lead White, | 51 | |
| Zinc Oxide, | 51 | |
End of Project Gutenberg's Paint Technology and Tests, by Henry A. Gardner
*** END OF THIS PROJECT GUTENBERG EBOOK PAINT TECHNOLOGY AND TESTS ***
***** This file should be named 37420-h.htm or 37420-h.zip *****
This and all associated files of various formats will be found in:
https://www.gutenberg.org/3/7/4/2/37420/
Produced by Chris Curnow, Harry Lamé and the Online
Distributed Proofreading Team at https://www.pgdp.net (This
file was produced from images generously made available
by The Internet Archive)
Updated editions will replace the previous one--the old editions
will be renamed.
Creating the works from public domain print editions means that no
one owns a United States copyright in these works, so the Foundation
(and you!) can copy and distribute it in the United States without
permission and without paying copyright royalties. Special rules,
set forth in the General Terms of Use part of this license, apply to
copying and distributing Project Gutenberg-tm electronic works to
protect the PROJECT GUTENBERG-tm concept and trademark. Project
Gutenberg is a registered trademark, and may not be used if you
charge for the eBooks, unless you receive specific permission. If you
do not charge anything for copies of this eBook, complying with the
rules is very easy. You may use this eBook for nearly any purpose
such as creation of derivative works, reports, performances and
research. They may be modified and printed and given away--you may do
practically ANYTHING with public domain eBooks. Redistribution is
subject to the trademark license, especially commercial
redistribution.
*** START: FULL LICENSE ***
THE FULL PROJECT GUTENBERG LICENSE
PLEASE READ THIS BEFORE YOU DISTRIBUTE OR USE THIS WORK
To protect the Project Gutenberg-tm mission of promoting the free
distribution of electronic works, by using or distributing this work
(or any other work associated in any way with the phrase "Project
Gutenberg"), you agree to comply with all the terms of the Full Project
Gutenberg-tm License (available with this file or online at
https://gutenberg.org/license).
Section 1. General Terms of Use and Redistributing Project Gutenberg-tm
electronic works
1.A. By reading or using any part of this Project Gutenberg-tm
electronic work, you indicate that you have read, understand, agree to
and accept all the terms of this license and intellectual property
(trademark/copyright) agreement. If you do not agree to abide by all
the terms of this agreement, you must cease using and return or destroy
all copies of Project Gutenberg-tm electronic works in your possession.
If you paid a fee for obtaining a copy of or access to a Project
Gutenberg-tm electronic work and you do not agree to be bound by the
terms of this agreement, you may obtain a refund from the person or
entity to whom you paid the fee as set forth in paragraph 1.E.8.
1.B. "Project Gutenberg" is a registered trademark. It may only be
used on or associated in any way with an electronic work by people who
agree to be bound by the terms of this agreement. There are a few
things that you can do with most Project Gutenberg-tm electronic works
even without complying with the full terms of this agreement. See
paragraph 1.C below. There are a lot of things you can do with Project
Gutenberg-tm electronic works if you follow the terms of this agreement
and help preserve free future access to Project Gutenberg-tm electronic
works. See paragraph 1.E below.
1.C. The Project Gutenberg Literary Archive Foundation ("the Foundation"
or PGLAF), owns a compilation copyright in the collection of Project
Gutenberg-tm electronic works. Nearly all the individual works in the
collection are in the public domain in the United States. If an
individual work is in the public domain in the United States and you are
located in the United States, we do not claim a right to prevent you from
copying, distributing, performing, displaying or creating derivative
works based on the work as long as all references to Project Gutenberg
are removed. Of course, we hope that you will support the Project
Gutenberg-tm mission of promoting free access to electronic works by
freely sharing Project Gutenberg-tm works in compliance with the terms of
this agreement for keeping the Project Gutenberg-tm name associated with
the work. You can easily comply with the terms of this agreement by
keeping this work in the same format with its attached full Project
Gutenberg-tm License when you share it without charge with others.
1.D. The copyright laws of the place where you are located also govern
what you can do with this work. Copyright laws in most countries are in
a constant state of change. If you are outside the United States, check
the laws of your country in addition to the terms of this agreement
before downloading, copying, displaying, performing, distributing or
creating derivative works based on this work or any other Project
Gutenberg-tm work. The Foundation makes no representations concerning
the copyright status of any work in any country outside the United
States.
1.E. Unless you have removed all references to Project Gutenberg:
1.E.1. The following sentence, with active links to, or other immediate
access to, the full Project Gutenberg-tm License must appear prominently
whenever any copy of a Project Gutenberg-tm work (any work on which the
phrase "Project Gutenberg" appears, or with which the phrase "Project
Gutenberg" is associated) is accessed, displayed, performed, viewed,
copied or distributed:
This eBook is for the use of anyone anywhere at no cost and with
almost no restrictions whatsoever. You may copy it, give it away or
re-use it under the terms of the Project Gutenberg License included
with this eBook or online at www.gutenberg.org
1.E.2. If an individual Project Gutenberg-tm electronic work is derived
from the public domain (does not contain a notice indicating that it is
posted with permission of the copyright holder), the work can be copied
and distributed to anyone in the United States without paying any fees
or charges. If you are redistributing or providing access to a work
with the phrase "Project Gutenberg" associated with or appearing on the
work, you must comply either with the requirements of paragraphs 1.E.1
through 1.E.7 or obtain permission for the use of the work and the
Project Gutenberg-tm trademark as set forth in paragraphs 1.E.8 or
1.E.9.
1.E.3. If an individual Project Gutenberg-tm electronic work is posted
with the permission of the copyright holder, your use and distribution
must comply with both paragraphs 1.E.1 through 1.E.7 and any additional
terms imposed by the copyright holder. Additional terms will be linked
to the Project Gutenberg-tm License for all works posted with the
permission of the copyright holder found at the beginning of this work.
1.E.4. Do not unlink or detach or remove the full Project Gutenberg-tm
License terms from this work, or any files containing a part of this
work or any other work associated with Project Gutenberg-tm.
1.E.5. Do not copy, display, perform, distribute or redistribute this
electronic work, or any part of this electronic work, without
prominently displaying the sentence set forth in paragraph 1.E.1 with
active links or immediate access to the full terms of the Project
Gutenberg-tm License.
1.E.6. You may convert to and distribute this work in any binary,
compressed, marked up, nonproprietary or proprietary form, including any
word processing or hypertext form. However, if you provide access to or
distribute copies of a Project Gutenberg-tm work in a format other than
"Plain Vanilla ASCII" or other format used in the official version
posted on the official Project Gutenberg-tm web site (www.gutenberg.org),
you must, at no additional cost, fee or expense to the user, provide a
copy, a means of exporting a copy, or a means of obtaining a copy upon
request, of the work in its original "Plain Vanilla ASCII" or other
form. Any alternate format must include the full Project Gutenberg-tm
License as specified in paragraph 1.E.1.
1.E.7. Do not charge a fee for access to, viewing, displaying,
performing, copying or distributing any Project Gutenberg-tm works
unless you comply with paragraph 1.E.8 or 1.E.9.
1.E.8. You may charge a reasonable fee for copies of or providing
access to or distributing Project Gutenberg-tm electronic works provided
that
- You pay a royalty fee of 20% of the gross profits you derive from
the use of Project Gutenberg-tm works calculated using the method
you already use to calculate your applicable taxes. The fee is
owed to the owner of the Project Gutenberg-tm trademark, but he
has agreed to donate royalties under this paragraph to the
Project Gutenberg Literary Archive Foundation. Royalty payments
must be paid within 60 days following each date on which you
prepare (or are legally required to prepare) your periodic tax
returns. Royalty payments should be clearly marked as such and
sent to the Project Gutenberg Literary Archive Foundation at the
address specified in Section 4, "Information about donations to
the Project Gutenberg Literary Archive Foundation."
- You provide a full refund of any money paid by a user who notifies
you in writing (or by e-mail) within 30 days of receipt that s/he
does not agree to the terms of the full Project Gutenberg-tm
License. You must require such a user to return or
destroy all copies of the works possessed in a physical medium
and discontinue all use of and all access to other copies of
Project Gutenberg-tm works.
- You provide, in accordance with paragraph 1.F.3, a full refund of any
money paid for a work or a replacement copy, if a defect in the
electronic work is discovered and reported to you within 90 days
of receipt of the work.
- You comply with all other terms of this agreement for free
distribution of Project Gutenberg-tm works.
1.E.9. If you wish to charge a fee or distribute a Project Gutenberg-tm
electronic work or group of works on different terms than are set
forth in this agreement, you must obtain permission in writing from
both the Project Gutenberg Literary Archive Foundation and Michael
Hart, the owner of the Project Gutenberg-tm trademark. Contact the
Foundation as set forth in Section 3 below.
1.F.
1.F.1. Project Gutenberg volunteers and employees expend considerable
effort to identify, do copyright research on, transcribe and proofread
public domain works in creating the Project Gutenberg-tm
collection. Despite these efforts, Project Gutenberg-tm electronic
works, and the medium on which they may be stored, may contain
"Defects," such as, but not limited to, incomplete, inaccurate or
corrupt data, transcription errors, a copyright or other intellectual
property infringement, a defective or damaged disk or other medium, a
computer virus, or computer codes that damage or cannot be read by
your equipment.
1.F.2. LIMITED WARRANTY, DISCLAIMER OF DAMAGES - Except for the "Right
of Replacement or Refund" described in paragraph 1.F.3, the Project
Gutenberg Literary Archive Foundation, the owner of the Project
Gutenberg-tm trademark, and any other party distributing a Project
Gutenberg-tm electronic work under this agreement, disclaim all
liability to you for damages, costs and expenses, including legal
fees. YOU AGREE THAT YOU HAVE NO REMEDIES FOR NEGLIGENCE, STRICT
LIABILITY, BREACH OF WARRANTY OR BREACH OF CONTRACT EXCEPT THOSE
PROVIDED IN PARAGRAPH 1.F.3. YOU AGREE THAT THE FOUNDATION, THE
TRADEMARK OWNER, AND ANY DISTRIBUTOR UNDER THIS AGREEMENT WILL NOT BE
LIABLE TO YOU FOR ACTUAL, DIRECT, INDIRECT, CONSEQUENTIAL, PUNITIVE OR
INCIDENTAL DAMAGES EVEN IF YOU GIVE NOTICE OF THE POSSIBILITY OF SUCH
DAMAGE.
1.F.3. LIMITED RIGHT OF REPLACEMENT OR REFUND - If you discover a
defect in this electronic work within 90 days of receiving it, you can
receive a refund of the money (if any) you paid for it by sending a
written explanation to the person you received the work from. If you
received the work on a physical medium, you must return the medium with
your written explanation. The person or entity that provided you with
the defective work may elect to provide a replacement copy in lieu of a
refund. If you received the work electronically, the person or entity
providing it to you may choose to give you a second opportunity to
receive the work electronically in lieu of a refund. If the second copy
is also defective, you may demand a refund in writing without further
opportunities to fix the problem.
1.F.4. Except for the limited right of replacement or refund set forth
in paragraph 1.F.3, this work is provided to you 'AS-IS' WITH NO OTHER
WARRANTIES OF ANY KIND, EXPRESS OR IMPLIED, INCLUDING BUT NOT LIMITED TO
WARRANTIES OF MERCHANTIBILITY OR FITNESS FOR ANY PURPOSE.
1.F.5. Some states do not allow disclaimers of certain implied
warranties or the exclusion or limitation of certain types of damages.
If any disclaimer or limitation set forth in this agreement violates the
law of the state applicable to this agreement, the agreement shall be
interpreted to make the maximum disclaimer or limitation permitted by
the applicable state law. The invalidity or unenforceability of any
provision of this agreement shall not void the remaining provisions.
1.F.6. INDEMNITY - You agree to indemnify and hold the Foundation, the
trademark owner, any agent or employee of the Foundation, anyone
providing copies of Project Gutenberg-tm electronic works in accordance
with this agreement, and any volunteers associated with the production,
promotion and distribution of Project Gutenberg-tm electronic works,
harmless from all liability, costs and expenses, including legal fees,
that arise directly or indirectly from any of the following which you do
or cause to occur: (a) distribution of this or any Project Gutenberg-tm
work, (b) alteration, modification, or additions or deletions to any
Project Gutenberg-tm work, and (c) any Defect you cause.
Section 2. Information about the Mission of Project Gutenberg-tm
Project Gutenberg-tm is synonymous with the free distribution of
electronic works in formats readable by the widest variety of computers
including obsolete, old, middle-aged and new computers. It exists
because of the efforts of hundreds of volunteers and donations from
people in all walks of life.
Volunteers and financial support to provide volunteers with the
assistance they need are critical to reaching Project Gutenberg-tm's
goals and ensuring that the Project Gutenberg-tm collection will
remain freely available for generations to come. In 2001, the Project
Gutenberg Literary Archive Foundation was created to provide a secure
and permanent future for Project Gutenberg-tm and future generations.
To learn more about the Project Gutenberg Literary Archive Foundation
and how your efforts and donations can help, see Sections 3 and 4
and the Foundation web page at https://www.pglaf.org.
Section 3. Information about the Project Gutenberg Literary Archive
Foundation
The Project Gutenberg Literary Archive Foundation is a non profit
501(c)(3) educational corporation organized under the laws of the
state of Mississippi and granted tax exempt status by the Internal
Revenue Service. The Foundation's EIN or federal tax identification
number is 64-6221541. Its 501(c)(3) letter is posted at
https://pglaf.org/fundraising. Contributions to the Project Gutenberg
Literary Archive Foundation are tax deductible to the full extent
permitted by U.S. federal laws and your state's laws.
The Foundation's principal office is located at 4557 Melan Dr. S.
Fairbanks, AK, 99712., but its volunteers and employees are scattered
throughout numerous locations. Its business office is located at
809 North 1500 West, Salt Lake City, UT 84116, (801) 596-1887, email
business@pglaf.org. Email contact links and up to date contact
information can be found at the Foundation's web site and official
page at https://pglaf.org
For additional contact information:
Dr. Gregory B. Newby
Chief Executive and Director
gbnewby@pglaf.org
Section 4. Information about Donations to the Project Gutenberg
Literary Archive Foundation
Project Gutenberg-tm depends upon and cannot survive without wide
spread public support and donations to carry out its mission of
increasing the number of public domain and licensed works that can be
freely distributed in machine readable form accessible by the widest
array of equipment including outdated equipment. Many small donations
($1 to $5,000) are particularly important to maintaining tax exempt
status with the IRS.
The Foundation is committed to complying with the laws regulating
charities and charitable donations in all 50 states of the United
States. Compliance requirements are not uniform and it takes a
considerable effort, much paperwork and many fees to meet and keep up
with these requirements. We do not solicit donations in locations
where we have not received written confirmation of compliance. To
SEND DONATIONS or determine the status of compliance for any
particular state visit https://pglaf.org
While we cannot and do not solicit contributions from states where we
have not met the solicitation requirements, we know of no prohibition
against accepting unsolicited donations from donors in such states who
approach us with offers to donate.
International donations are gratefully accepted, but we cannot make
any statements concerning tax treatment of donations received from
outside the United States. U.S. laws alone swamp our small staff.
Please check the Project Gutenberg Web pages for current donation
methods and addresses. Donations are accepted in a number of other
ways including including checks, online payments and credit card
donations. To donate, please visit: https://pglaf.org/donate
Section 5. General Information About Project Gutenberg-tm electronic
works.
Professor Michael S. Hart was the originator of the Project Gutenberg-tm
concept of a library of electronic works that could be freely shared
with anyone. For thirty years, he produced and distributed Project
Gutenberg-tm eBooks with only a loose network of volunteer support.
Project Gutenberg-tm eBooks are often created from several printed
editions, all of which are confirmed as Public Domain in the U.S.
unless a copyright notice is included. Thus, we do not necessarily
keep eBooks in compliance with any particular paper edition.
Most people start at our Web site which has the main PG search facility:
https://www.gutenberg.org
This Web site includes information about Project Gutenberg-tm,
including how to make donations to the Project Gutenberg Literary
Archive Foundation, how to help produce our new eBooks, and how to
subscribe to our email newsletter to hear about new eBooks.