 Fig. 1.
Fig. 1.
The Project Gutenberg EBook of Outlines of Dairy Bacteriology, 8th edition, by
H. L. Russell
This eBook is for the use of anyone anywhere at no cost and with
almost no restrictions whatsoever. You may copy it, give it away or
re-use it under the terms of the Project Gutenberg License included
with this eBook or online at www.gutenberg.org
Title: Outlines of Dairy Bacteriology, 8th edition
A Concise Manual for the Use of Students in Dairying
Author: H. L. Russell
Release Date: January 11, 2009 [EBook #27778]
Language: English
Character set encoding: ISO-8859-1
*** START OF THIS PROJECT GUTENBERG EBOOK DAIRY BACTERIOLOGY, 8TH EDITION ***
Produced by Mark C. Orton, Linda McKeown, Josephine Paolucci
and the Online Distributed Proofreading Team at
https://www.pgdp.net.
EIGHTH EDITION
Thoroughly Revised
MADISON, WISCONSIN
H. L. RUSSELL
1907
Copyrighted 1905
BY
H. L. RUSSELL
STATE JOURNAL PRINTING COMPANY,
Printers And Stereotypers,
Madison, Wis.
Transcriber's note: Minor typos have been corrected.
Knowledge in dairying, like all other technical industries, has grown mainly out of experience. Many facts have been learned by observation, but the why of each is frequently shrouded in mystery.
Modern dairying is attempting to build its more accurate knowledge upon a broader and surer foundation, and in doing this is seeking to ascertain the cause of well-established processes. In this, bacteriology is playing an important rôle. Indeed, it may be safely predicted that future progress in dairying will, to a large extent, depend upon bacteriological research. As Fleischmann, the eminent German dairy scientist, says: "The gradual abolition of uncertainty surrounding dairy manufacture is the present important duty which lies before us, and its solution can only be effected by bacteriology."
It is therefore natural that the subject of Dairy Bacteriology has come to occupy an important place in the curriculum of almost every Dairy School. An exposition of its principles is now recognized as an integral part of dairy science, for modern dairy practice is rapidly adopting the methods that have been developed as the result of bacteriological study. The rapid development of the subject has necessitated a frequent revision of this work, and it is gratifying to the writer that the attempt which has been made to keep these Outlines abreast of bacteriological advance has been appreciated by students of dairying.
While the text is prepared more especially for the practical[Pg iv] dairy operator who wishes to understand the principles and reasons underlying his art, numerous references to original investigations have been added to aid the dairy investigator who wishes to work up the subject more thoroughly.
My acknowledgments are due to the following for the loan of illustrations: Wisconsin Agricultural Experiment Station; Creamery Package Mfg. Co., Chicago, Ill.; and A. H. Reid, Philadelphia, Pa.
Chapter I. Structure of the bacteria and conditions governing their development and distribution 1
Chapter II. Methods of studying bacteria 13
Chapter III. Contamination of milk 19
Chapter IV. Fermentations in milk and their treatment 62
Chapter V. Relation of disease-bacteria to milk 82
Diseases transmissible from animal to man through diseased milk 84
Diseases transmissible to man through infection of milk after withdrawal 94
Chapter VI. Preservation of milk for commercial purposes 102
Chapter VII. Bacteria and butter making 134
Bacterial defects in butter 156
Chapter VIII. Bacteria in cheese 160
Influence Of bacteria in normal cheese processes 160
Influence of bacteria in abnormal cheese processes 182
Before one can gain any intelligent conception of the manner in which bacteria affect dairying, it is first necessary to know something of the life history of these organisms in general, how they live, move and react toward their environment.
Nature of Bacteria. Toadstools, smuts, rusts and mildews are known to even the casual observer, because they are of evident size. Their plant-like nature can be more readily understood from their general structure and habits of life. The bacteria, however, are so small, that under ordinary conditions, they only become evident to our unaided senses by the by-products of their activity.
When Leeuwenhoek (pronounced Lave-en-hake) in 1675 first discovered these tiny, rapidly-moving organisms he thought they were animals. Indeed, under a microscope, many of them bear a close resemblance to those minute worms found in vinegar that are known as "vinegar-eels." The idea that they belonged to the animal kingdom continued to hold ground until after the middle of the nineteenth century; but with the improvement in microscopes, a more thorough study of these tiny structures was made possible, and their vegetable nature demonstrated. The bacteria as a class are separated from the fungi mainly by their method of growth; from the lower algae by the absence of chlorophyll, the green coloring matter of vegetable organisms.[Pg 2]
Structure of bacteria. So far as structure is concerned the bacteria stand on the lowest plane of vegetable life. The single individual is composed of but a single cell, the structure of which does not differ essentially from that of many of the higher types of plant life. It is composed of a protoplasmic body which is surrounded by a thin membrane that separates it from neighboring cells that are alike in form and size.
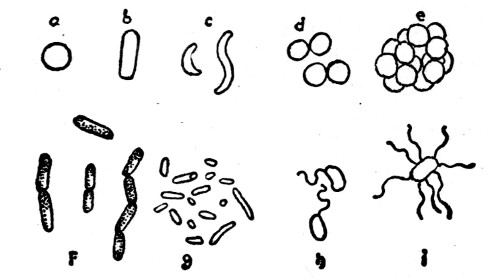
Form and size. When a plant is composed of a single cell but little difference in form is to be expected. While there are intermediate stages that grade insensibly into each other, the bacteria may be grouped into three main types, so far as form is concerned. These are spherical, elongated, and spiral, and to these different types are given the names, respectively, coccus, bacillus and spirillum (plural, cocci, bacilli, spirilla) (fig. 1). A ball, a short rod, and a corkscrew serve as convenient models to illustrate these different forms.
 Fig. 1.
Fig. 1.
Different forms of bacteria. a, b, c, represent different types as to form: a, coccus, b, bacillus, c, spirillum; d, diplococcus or twin coccus; e, staphylococcus or cluster coccus; f and g, different forms of bacilli, g shows internal endospores within cell; h and i, bacilli with motile organs (cilia).
In size, the bacteria are the smallest organisms that are known to exist. Relatively there is considerable difference in[Pg 3] size between the different species, yet in absolute amount this is so slight as to require the highest powers of the microscope to detect it. As an average diameter, one thirty-thousandth of an inch may be taken. It is difficult to comprehend such minute measurements, but if a hundred individual germs could be placed side by side, their total thickness would not equal that of a single sheet of paper upon which this page is printed.
Manner of Growth. As the cell increases in size as a result of growth, it elongates in one direction, and finally a new cell wall is formed, dividing the so-called mother-cell into two, equal-sized daughter-cells. This process of cell division, known as fission, is continued until growth ceases and is especially characteristic of bacteria.
Cell Arrangement. If fission goes on in the same plane continually, it results in the formation of a cell-row. A coccus forming such a chain of cells is called strepto-coccus (chain-coccus). If only two cells cohere, it is called a diplo-coccus (twin-coccus). If the second cell division plane is formed at right angles to the first, a cell surface or tetrad is formed. If growth takes place in three dimensions of space, a cell mass or sarcina is produced. Frequently, these cell aggregates cohere so tenaciously that this arrangement is of value in distinguishing different species.
Spores. Some bacteria possess the property of forming spores within the mother cell (called endospores, fig. 1g) that are analogous in function to the seeds of higher plants and spores of fungi. By means of these structures which are endowed with greater powers of resistance than the vegetating cell, the organism is able to protect itself from the effect of an unfavorable environment. Many of the bacilli form endospores but the cocci do not. It is these[Pg 4] spore forms that make it so difficult to thoroughly sterilize milk.
Movement. Many bacteria are unable to move from place to place. They have, however, a vibrating movement known as the Brownian motion that is purely physical. Many other kinds are endowed with powers of locomotion. Motion is produced by means of fine thread-like processes of protoplasm known as cilia (sing. cilium) that are developed on the outer surface of the cell. By means of the rapid vibration of these organs, the cell is propelled through the medium. Nearly all cocci are immotile, while the bacilli may or may not be. These cilia are so delicate that it requires special treatment to demonstrate their presence.
Classification. In classifying or arranging the different members of any group of living objects, certain similarities and dissimilarities must be considered. These are usually those that pertain to the structure and form, as such are regarded as most constant. With the bacteria these differences are so slight that they alone do not suffice to distinguish distinctly one species from another. As far as these characters can be used, they are taken, but in addition, many characteristics of a physiological nature are added. The way that the organism grows in different kinds of cultures, the by-products produced in different media, and effect on the animal body when injected into the same are also used as data in distinguishing one species from another.
Conditions favoring bacterial growth. The bacteria, in common with all other living organisms are affected by external conditions, either favorably or unfavorably. Certain conditions must prevail before development can occur. Thus, the organism must be supplied with an adequate[Pg 5] and suitable food supply and with moisture. The temperature must also range between certain limits, and finally, the oxygen requirements of the organism must be considered.
Food supply. Most bacteria are capable of living on dead, inert, organic matter, such as meats, milk and vegetable material, in which case, they are known as saprophytes. In contradistinction to this class is a smaller group known as parasites, which derive their nourishment from the living tissues of animals or plants. The first group comprise by far the larger number of known organisms which are concerned for the most part in the decomposition of organic matter. The parasitic group includes those which are the cause of various communicable diseases. Between these two groups there is no sharp line of division, and in some cases, certain species possess the faculty of growing either as parasites or saprophytes, in which case they are known as facultative parasites or saprophytes.
The great majority of bacteria of interest in dairying belong to the saprophytic class; only those species capable of infecting milk through the development of disease in the animal are parasites in the strict sense of the term. Most disease-producing species, as diphtheria or typhoid fever, while parasitic in man lead a saprophytic method of life so far as their relation to milk is concerned.
Bacteria require for their growth, nitrogen, hydrogen, carbon, oxygen, together with a limited amount of mineral matter. The nitrogen and carbon are most available in the form of organic compounds, such as albuminous material. Carbon in the form of carbohydrates, as sugar or starch, is most readily attacked by bacteria.
Inasmuch as the bacteria are plant-cells, they must imbibe[Pg 6] their food from material in solution. They are capable of living on solid substances, but in such cases, the food elements must be rendered soluble, before they can be appropriated. If nutritive liquids are too highly concentrated, as in the case of syrups and condensed milk, bacteria cannot grow therein, although all the necessary ingredients may be present. Generally, bacteria prefer a neutral or slightly alkaline medium, rather than one of acid reaction; but there are numerous exceptions to this general rule, especially among the bacteria found in milk.
Temperature. Growth of bacteria can only occur within certain temperature limits, the extremes of which are designated as the minimum and maximum. Below and above these respective limits, life may be retained in the cell for a time, but actual cell-multiplication is stopped. Somewhere between these two cardinal temperature points, and generally nearer the maximum limit is the most favorable temperature for growth, known as the optimum. The temperature zone of most dairy bacteria in which growth occurs ranges from 40°-45° F. to somewhat above blood-heat, 105°-110° F., the optimum being from 80°-95° F. Many parasitic species, because of their adaptation to the bodies of warm-blooded animals, generally have a narrower range, and a higher optimum, usually approximating the blood heat (98°-99° F). The broader growth limits of bacteria in comparison with other kinds of life explain why these organisms are so widely distributed in nature.
Air supply. Most bacteria require as do the green plants and animal life, the free oxygen of the air for their respiration. These are called aerobic. Some species, however, and some yeasts as well possess the peculiar property of taking the oxygen which they need from organic compounds[Pg 7] such as sugar, etc., and are therefore able to live and grow under conditions where the atmospheric air is excluded. These are known as anaerobic. While some species grow strictly under one condition or the other, and hence are obligate aerobes or anaerobes, others possess the ability of growing under either condition and are known as facultative or optional forms. The great majority of milk bacteria are either obligate or facultative aerobes.
Rate of growth. The rate of bacterial development is naturally very much affected by external conditions, food supply and temperature exerting the most influence. In the neighborhood of the freezing point but little growth occurs. The rate increases with a rise in temperature until at the optimum point, which is generally near the blood heat or slightly below (90°-98° F.), a single cell will form two cells in 20 to 30 minutes. If temperature rises much above blood heat rate of growth is lessened and finally ceases. Under ideal conditions, rapidity of growth is astounding, but this initially rapid rate of development cannot be maintained indefinitely, for growth is soon limited by the accumulation of by-products of cell activity. Thus, milk sours rapidly at ordinary temperatures until the accumulation of acid checks its development.
Detrimental effect of external conditions. Environmental influences of a detrimental character are constantly at work on bacteria, tending to repress their development or destroy them. These act much more readily on the vegetating cell than on the more resistant spore. A thorough knowledge of the effect of these antagonistic forces is essential, for it is often by their means that undesirable bacteria may be killed out.[Pg 8]
Effect of cold. While it is true that chilling largely prevents fermentative action, and actual freezing stops all growth processes, still it does not follow that exposure to low temperatures will effectually destroy the vitality of bacteria, even in the vegetative condition. Numerous non-spore-bearing species remain alive in ice for a prolonged period, and recent experiments with liquid air show that even a temperature of -310° F. for hours does not effectually kill all exposed cells.
Effect of heat. High temperatures, on the other hand, will destroy any form of life, whether in the vegetative or latent stage. The temperature at which the vitality of the cell is lost is known as the thermal death point. This limit is not only dependent upon the nature of the organism, but varies with the time of exposure and the condition in which the heat is applied. In a moist atmosphere the penetrating power of heat is great; consequently cell-death occurs at a lower temperature than in a dry atmosphere. An increase in time of exposure lowers the temperature point at which death occurs.
For vegetating forms the thermal death point of most bacteria ranges from 130°-140° F. where the exposure is made for ten minutes which is the standard arbitrarily selected. In the spore stage resistance is greatly increased, some forms being able to withstand steam at 210°-212° F. from one to three hours. If dry heat is employed, 260°-300° F. for an hour is necessary to kill spores. Where steam is confined under pressure, a temperature of 230°-240° F. for 15-20 minutes suffices to kill all spores.
Drying. Spore-bearing bacteria like anthrax withstand drying with impunity; even tuberculous material, although not possessing spores retains its infectious properties for[Pg 9] many months. Most of the dairy bacteria do not produce spores, and yet in a dry condition, they retain their vitality unimpaired for considerable periods, if they are not subjected to other detrimental influences.
Light. Bright sunlight exerts on many species a powerful disinfecting action, a few hours being sufficient to destroy all cells that are reached by the sun's rays. Even diffused light has a similar effect, although naturally less marked. The active rays in this disinfecting action are those of the chemical or violet end of the spectrum, and not the heat or red rays.
Influence of chemical substances. A great many chemical substances exert a more or less powerful toxic action of various kinds of life. Many of these are of great service in destroying or holding bacterial growth in check. Those that are toxic and result in the death of the cell are known as disinfectants; those that merely inhibit, or retard growth are known as antiseptics. All disinfectants must of necessity be antiseptic in their action, but not all antiseptics are disinfectants even when used in strong doses. Disinfectants have no place in dairy work, except to destroy disease bacteria, or preserve milk for analytical purposes. Corrosive sublimate or potassium bichromate are most frequently used for these purposes. The so-called chemical preservatives used to "keep" milk depend for their effect on the inhibition of bacterial growth. With a substance so violently toxic as formaldehyde (known as formalin, freezene) antiseptic doses are likely to be exceeded. In this country most states prohibit the use of these substances in milk. Their only function in the dairy should be to check fermentative or putrefactive processes outside of milk and so keep the air free from taints.[Pg 10]
Products of growth. All bacteria in their development form certain more or less characteristic by-products. With most dairy bacteria, these products are formed from the decomposition of the medium in which the bacteria may happen to live. Such changes are known, collectively, as fermentations, and are characterised by the production of a large amount of by-products, as a result of the development of a relatively small amount of cell-life. The souring of milk, the formation of butyric acid, the making of vinegar from cider, are all examples of fermentative changes.
With many bacteria, especially those that affect proteid matter, foul-smelling gases are formed. These are known as putrefactive changes. All organic matter, under the action of various organisms, sooner or later undergoes decay, and in different stages of these processes, acids, alkalies, gases and numerous other products are formed. Many of these changes in organic matter occur only when such material is brought in direct contact with the living bacterial cell.
In other instances, soluble, non-vital ferments known as enzyms are produced by the living cell, which are able to act on organic matter, in a medium free from live cells, or under conditions where the activity of the cell is wholly suspended. These enzyms are not confined to bacteria but are found throughout the animal and plant world, especially in those processes that are concerned in digestion. Among the better known of these non-vital ferments are rennet, the milk-curdling enzym; diastase or ptyalin of the saliva, the starch-converting enzym; pepsin and trypsin, the digestive ferments of the animal body.
Enzyms of these types are frequently found among the bacteria and yeasts and it is by virtue of this characteristic[Pg 11] that these organisms are able to break down such enormous quantities of organic matter. Most of these enzyms react toward heat, cold and chemical poisons in a manner quite similar to the living cells. In one respect they are readily differentiated, and that is, that practically all of them are capable of producing their characteristic chemical transformations under anaesthetic conditions, as in a saturated ether or chloroform atmosphere.
Distribution of bacteria. As bacteria possess greater powers of resistance than most other forms of life, they are to be found more widely distributed than any other type. At the surface of the earth, where conditions permit of their growth, they are found everywhere, except in the healthy tissues of animals and plants. In the superficial soil layers, they exist in myriads, as here they have abundance of nourishment. At the depth of several feet however, they diminish rapidly in numbers, and in the deeper soil layers, from six to ten feet or more, they are not present, because of the unsuitable growth conditions.
The bacteria are found in the air because of their development in the soil below. They are unable to grow even in a moist atmosphere, but are so readily dislodged by wind currents that over land areas the lower strata of the air always contain them. They are more numerous in summer than in winter; city air contains larger numbers than country air. Wherever dried fecal matter is present, as in barns, the air contains many forms.
Water contains generally enough organic matter in solution, so that certain types of bacterial life find favorable growth conditions. Water in contact with the soil surface takes up many impurities, and is of necessity rich in microbes. As the rain water percolates into the soil, it loses[Pg 12] its germ content, so that the normal ground water, like the deeper soil layers, contains practically no bacterial life. Springs therefore are relatively deficient in germ life, except as they become infected with soil organisms, as the water issues from the soil. Water may serve to disseminate certain infectious diseases as typhoid fever and cholera among human beings, and a number of animal maladies.
While the inner tissues of healthy animals are free from bacteria, the natural passages as the respiratory and digestive tracts, being in more direct contact with the exterior, become more readily infected. This is particularly true with reference to the intestinal tract, for in the undigested residue, bacterial activity is at a maximum. The result is that fecal matter contains enormous numbers of organisms so that the possibility of pollution of any food medium such as milk with such material is sure to introduce elements that seriously affect the quality of the product.
Necessity of bacterial masses for study. The bacteria are so extremely small that it is impossible to study individual germs separately without the aid of first-class microscopes. For this reason, but little advance was made in the knowledge of these lower forms of plant life, until the introduction of culture methods, whereby a single organism could be cultivated and the progeny of this cell increased to such an extent in a short course of time, that they would be visible to the unaided eye.
This is done by growing the bacteria in masses on various kinds of food media that are prepared for the purpose, but inasmuch as bacteria are so universally distributed, it becomes an impossibility to cultivate any special form, unless the medium in which they are grown is first freed from all pre-existing forms of germ life. To accomplish this, it is necessary to subject the nutrient medium to some method of sterilization, such as heat or filtration, whereby all life is completely destroyed or eliminated. Such material after it has been rendered germ-free is kept in sterilized glass tubes and flasks, and is protected from infection by cotton stoppers.
Culture media. For culture media, many different substances are employed. In fact, bacteria will grow on almost any organic substance whether it is solid or fluid, provided the other essential conditions of growth are furnished. The food substances that are used for culture purposes are divided into two classes; solids and liquids.[Pg 14]
Solid media may be either permanently solid like potatoes, or they may retain their solid properties only at certain temperatures like gelatin or agar. The latter two are of utmost importance in bacteriological research, for their use, which was introduced by Koch, permits the separation of the different forms that may happen to be in any mixture. Gelatin is used advantageously because the majority of bacteria present wider differences due to growth upon this medium than upon any other. It remains solid at ordinary temperatures, becoming liquid at about 70° F. Agar, a gelatinous product derived from a Japanese sea-weed, has a much higher melting point, and can be successfully used, especially with those organisms whose optimum growth point is above the melting point of gelatin.
Besides these solid media, different liquid substances are extensively used, such as beef broth, milk, and infusions of various vegetable and animal tissues. Skim-milk is of especial value in studying the milk bacteria and may be used in its natural condition, or a few drops of litmus solution may be added in order to detect any change in its chemical reaction due to the bacteria.
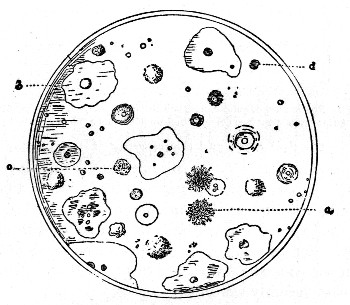 Fig. 2. A gelatin plate culture showing appearance of
different organisms in a sample of milk. Each mass represents a
bacterial growth (colony) derived from a single cell. Different forms
react differently toward the gelatin, some liquefying the same, others
growing in a restricted mass. a, represents a colony of the ordinary
bread mold; b, a liquefying bacterium; c, and d, solid forms.
Fig. 2. A gelatin plate culture showing appearance of
different organisms in a sample of milk. Each mass represents a
bacterial growth (colony) derived from a single cell. Different forms
react differently toward the gelatin, some liquefying the same, others
growing in a restricted mass. a, represents a colony of the ordinary
bread mold; b, a liquefying bacterium; c, and d, solid forms.
Methods of isolation. Suppose for instance one wishes to isolate the different varieties of bacteria found in milk. The method of procedure is as follows: Sterile gelatin in glass tubes is melted and cooled down so as to be barely warm. To this gelatin which is germ-free a drop of milk is added. The gelatin is then gently shaken so as to thoroughly distribute the milk particles, and poured out into a sterile flat glass dish and quickly covered. This is allowed to stand on a cool surface until the gelatin hardens. After the culture plate has been left for twenty-four to thirty-six[Pg 15] hours at the proper temperature, tiny spots will begin to appear on the surface, or in the depth of the culture medium. These patches are called colonies and are composed of an almost infinite number of individual germs, the result of the continued growth of a single organism that was in the drop of milk which was firmly held in place when the gelatin solidified. The number of these colonies represents approximately the number of germs that were present in the milk drop. If the plate is not too thickly sown with these germs, the colonies will continue to grow and increase in size, and as they do, minute differences will begin to appear. These differences may be in the color, the contour and the texture of the colony, or[Pg 16] the manner in which it acts toward gelatin. In order to make sure that the seeding in not too copious so as to interfere with continued study, an attenuation is usually made. This consists in taking a drop of the infected gelatin in the first tube, and transferring it to another tube of sterile media. Usually this operation is repeated again so that these culture plates are made with different amounts of seed with the expectation that in at least one plate the seeding will not be so thick as to prevent further study. For transferring the culture a loop made of platinum wire is used. By passing this through a gas flame, it can be sufficiently sterilized.
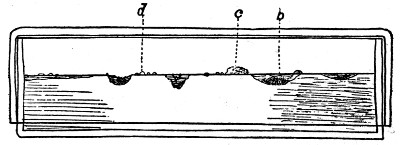 Fig. 3.
Fig. 3.
Profile view of gelatin plate culture; b, a liquefying form that dissolves the gelatin; c and d, surface colonies that do not liquefy the gelatin.
To further study the peculiarities of different germs, the separate colonies are transferred to other sterile tubes of culture material and thus pure cultures of the various germs are secured. These cultures then serve as a basis for continued study and must be planted and grown upon all the different kinds of media that are obtainable. In this way the slight variations in the growth of different forms are detected and the peculiar characteristics are determined, so that the student is able to recognize this form when he meets it again.
These culture methods are of essential importance in bacteriology, as it is the only way in which it is possible to secure a quantity of germs of the same kind.[Pg 17]
The microscope in bacterial investigation. In order to verify the purity of the cultures, the microscope is in constant demand throughout all the different stages of the isolating process. For this purpose, it is essential that the instrument used shall be one of strong magnifying powers (600-800 diameters), combined with sharp definition.
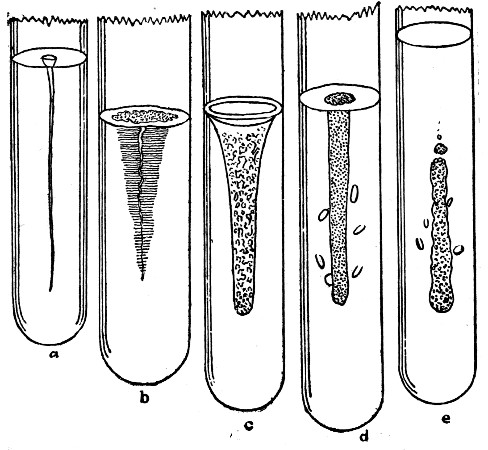 Fig. 4.
Fig. 4.
Pure cultures of different kinds of bacteria in gelatin tubes. a, growth slight in this medium; b, growth copious at and near surface. Fine parallel filaments growing out into medium liquefying at surface; c, a rapid liquefying form; d, a gas-producing form that grows equally well in lower part of tube as at surface (facultative anaerobe); e, an obligate anaerobe, that develops only in absence of air.
The microscopical examination of any germ is quite as[Pg 18] essential as the determination of culture characteristics; in fact, the two must go hand in hand. The examination reveals not only the form and size of the individual germs, but the manner in which they are united with each other, as well as any peculiarities of movement that they may possess.
In carrying out the microscopical part of the work, not only is the organism examined in a living condition, but preparations are made by using solutions of anilin dyes as staining agents. These are of great service in bringing out almost imperceptible differences. The art of staining has been carried to the highest degree of perfection in bacteriology, especially in the detection of germs that are found in diseased tissues in the animal or human body.
In studying the peculiarities of any special organism, not only is it necessary that these cultural and microscopical characters should be closely observed, but special experiments must be carried out along different lines, in order to determine any special properties that the germ may possess. Thus, the ability of any form to act as a fermentative organism can be tested by fermentation experiments; the property of causing disease, studied by the inoculation of pure cultures into animals. A great many different methods have been devised for the purpose of studying special characteristics of different bacteria, but a full description of these would necessarily be so lengthy that in a work of this character they must be omitted. For details of this nature consult standard reference books on bacteriological technique.
No more important lesson is to be learned than that which relates to the ways in which milk is contaminated with germ life of various kinds; for if these sources of infection are thoroughly recognized they can in large measure be prevented, and so the troubles which they engender overcome. Various organisms find in milk a congenial field for development. Yeasts and some fungi are capable of growth, but more particularly the bacteria.
Milk a suitable bacterial food. The readiness with which milk undergoes fermentative changes indicates that it is well adapted to nourish bacterial life. Not only does it contain all the necessary nutritive substances but they are diluted in proper proportions so as to render them available for bacterial as well as mammalian life.
Of the nitrogenous compounds, the albumen is in readily assimilable form. The casein, being insoluble, is not directly available, until it is acted upon by proteid-dissolving enzyms like trypsin which may be secreted by bacteria. The fat is relatively resistant to change, although a few forms are capable of decomposing it. Milk sugar, however, is an admirable food for many species, acids and sometimes gases being generally produced.
Condition when secreted. When examined under normal conditions milk always reveals bacterial life, yet in the secreting cells of the udder of a healthy cow germ life is not found. Only when the gland is diseased are bacteria[Pg 20] found in any abundance. In the passage of the milk from the secreting cells to the outside it receives its first infection, so that when drawn from the animal it generally contains a considerable number of organisms.
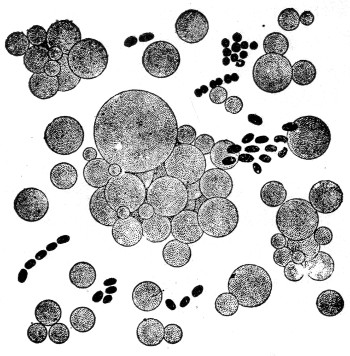 Fig. 5. Microscopic appearance of milk showing relative
size of fat globules and bacteria.
Fig. 5. Microscopic appearance of milk showing relative
size of fat globules and bacteria.
Contamination of milk. From this time until it is consumed in one form or another, it is continually subjected to contamination. The major part of this infection occurs while the milk is on the farm and the degree of care which is exercised while the product is in the hands of the milk producer is the determining factor in the course of bacterial changes involved. This of course does not exclude the possibility of contamination in the factory, but usually milk is so thoroughly seeded by the time it reaches the factory that the infection which occurs here plays a relatively minor rôle to that which happens earlier. The great majority of the organisms in milk are in no wise dangerous to health, but many species are capable of producing various fermentative changes that injure the quality of the product for butter or cheese. To be able to control abnormal changes of an undesirable character one must know the sources of infection which permit of the introduction of these unwelcome intruders.[Pg 21]
Sources of infection. The bacterial life that finds its way into milk while it is yet on the farm may be traced to several sources, which may be grouped under the following heads: unclean dairy utensils, fore milk, coat of animal, and general atmospheric surroundings. The relative importance of these various factors fluctuates in each individual instance.
Dairy utensils. Of first importance are the vessels that are used during milking, and also all storage cans and other dairy utensils that come in contact with the milk after it is drawn. By unclean utensils, actually visible dirt need not always be considered, although such material is often present in cracks and angles of pails and cans. Unless cleansed with especial care, these are apt to be filled with foul and decomposing material that suffices to seed thoroughly the milk. Tin utensils are best. Where made with joints, they should be well flushed with solder so as to be easily cleaned (see Fig. 6). In much of the cheaper tin ware on the market, the soldering of joints and seams is very imperfect, affording a place of refuge for bacteria and dirt.
Cans are often used when they are in a condition wholly unsuitable for sanitary handling of milk. When the tin coating becomes broken and the can is rusty, the quality of the milk is often profoundly affected. Olson[1] of the Wisconsin Station has shown that the action of rennet is greatly impaired where milk comes in contact with a rusty iron surface.
 Fig. 6.
Fig. 6.
With the introduction of the form or hand separator a new milk utensil has been added to those previously in use and one which is very frequently not well cleaned. Where[Pg 22] water is run through the machine to rinse out the milk particles, gross bacterial contamination occurs and the use of the machine much increases the germ content of the milk. Every time the separator is used it should be taken apart and thoroughly cleaned and dried before reassembling.[2]
Use of milk-cans for transporting factory by-products. The general custom of using the milk-cans to carry back to the farm the factory by-products (skim-milk or whey) has much in it that is to be deprecated. These by-products are generally rich in bacterial life, more especially where the closest scrutiny is not given to the daily cleaning of the vats and tanks. Too frequently the cans are not cleaned immediately upon arrival at the farm, so that the conditions are favorable for rapid fermentation. Many of the taints that bother factories are directly traceable to such a cause. A few dirty patrons will thus seriously infect the whole supply. The responsibility for this defect should, however, not be laid entirely upon the shoulders of the producer. The factory operator should see that the refuse material does not accumulate in the waste vats from day to day and is not transformed into a more or less putrid mass. A dirty whey tank is not an especially good object lesson to the patron to keep his cans clean.
It is possible that abnormal fermentations or even contagious diseases may thus be disseminated.
Suppose there appears in a dairy an infectious milk trouble, such as bitter milk. This milk is taken to the factory and passes unnoticed into the general milk-supply. The skim-milk from the separator is of course infected with the germ, and if conditions favor its growth, the whole lot soon becomes tainted. If this waste product is returned to the different patrons in the same cans that are used for the fresh milk, the probabilities are strongly in[Pg 23] favor of some of the cans being contaminated and thus infecting the milk supply of the patrons. If the organism is endowed with spores so that it can withstand unfavorable conditions, this taint may be spread from patron to patron simply through the infection of the vessels that are used in the transportation of the by-products. Connell has reported just such a case in a Canadian cheese factory where an outbreak of slimy milk was traced to infected whey vats. Typhoid fever among people, foot and mouth disease and tuberculosis among stock are not infrequently spread in this way. In Denmark, portions of Germany and some states in America, compulsory heating of factory by-products is practiced to eliminate this danger.[3]
Pollution of cans from whey tanks. The danger is greater in cheese factories than in creameries, for whey usually represents a more advanced stage of fermentation than skim-milk. The higher temperature at which it is drawn facilitates more rapid bacterial growth, and the conditions under which it is stored in many factories contribute to the ease with which fermentative changes can go on in it. Often this by-product is stored in wooden cisterns or tanks, situated below ground, where it becomes impossible to clean them out thoroughly. A custom that is almost universally followed in the Swiss cheese factories, here in this country, as in Switzerland, is fully as reprehensible as any[Pg 24] dairy custom could well be. In Fig. 7 the arrangement in vogue for the disposal of the whey is shown. The hot whey is run out through the trough from the factory into the large trough that is placed over the row of barrels, as seen in the foreground. Each patron thus has allotted to him in his individual barrel his portion of the whey, which he is supposed to remove day by day. No attempt is made to clean out these receptacles, and the inevitable result is that they become filled with a foul, malodorous liquid, especially in summer. When such material is taken home in the same set of cans that is used to bring the fresh milk (twice a day as is the usual custom in Swiss factories), it is no wonder that this industry is seriously handicapped by "gassy" fermentations that injure materially the quality of the product. Not only is the above danger a very[Pg 25] considerable one, but the quality of the factory by-product for feeding purposes, whether it is skim-milk or whey, is impaired through the development of fermentative changes.
 Fig. 7.
Fig. 7.
Swiss cheese factory (Wisconsin), showing careless way in which whey is handled. Each patron's share is placed in a barrel, from which it is removed by him. No attempt is made to cleanse these receptacles.
Improved methods of disposal of by-products. The difficulties which attend the distribution of these factory by-products have led to different methods of solution. One is to use another separate set of receptacles to carry back these products to the farm. This method has been tried, and while it is deemed impracticable by many to handle two sets of vessels, yet some of the most progressive factories report excellent results where this method is in use.
Large barrels could be used for this purpose to economize in wagon space.
Another method that has met with wider acceptance, especially in creameries, is the custom of pasteurizing or scalding the skim-milk immediately after it is separated, so that it is returned to the farmer in a hot condition. In factories where the whole milk is pasteurized, further treatment of the by-product is not necessary. In most factories steam, generally exhaust, is used directly in the milk, and experience has shown that such milk, without any cooling, will keep sweet for a considerable number of hours longer than the untreated product. It is noteworthy that the most advanced and progressive factories are the ones that appreciate the value of this work, and although it involves some time and expense, experience has shown the utility of the process in that a better grade of milk is furnished by the patrons of factories which follow this practice.[4] The exclusion of all danger of animal or human disease is also possible in this way.
Cleaning dairy utensils. The thorough cleaning of all dairy apparatus that in any way comes in contact with the milk is one of the most fundamental and important problems in dairying. All such apparatus should be so constructed as to permit of easy cleaning. Tinware, preferably of the pressed variety, gives the best surface for this purpose and is best suited for the handling of milk.
Milk vessels should never be allowed to become dry when dirty, for dried particles of milk residue are extremely difficult to remove. In cleaning dairy utensils they should first be rinsed in lukewarm instead of hot water, so as to remove organic matter without coagulating the milk. Then wash thoroughly in hot water, using a good washing powder. The best washing powders possess considerable disinfecting action.[5] Strong alkalies should not be used. After washing rinse thoroughly in clean hot water. If steam is available, as it always is in creameries, cans and pails should be turned over jet for a few moments. While a momentary exposure will not suffice to completely sterilize such a vessel, yet many bacteria are killed in even a short exposure, and the cans dry more thoroughly and quickly when heated by steam.
Not only should the greatest care be paid to the condition of the cans and milk-pails, but all dippers, strainers, and other utensils that come in contact with the milk must be kept equally clean. Cloth strainers, unless attended to, are objectionable, for the fine mesh of the cloth retains so much moisture that they become a veritable hot-bed of bacterial life, unless they are daily boiled or steamed.
The inability to thoroughly render vessels bacteria-free with the conveniences which are generally to be found on the farm has led in some cases to the custom of washing and sterilizing the milk cans at the factory.
Germ content of milk utensils. Naturally the number of bacteria found in different milk utensils after they have received their regular cleaning will be subject to great fluctuations; but, nevertheless, such determinations are of value as giving a scientific foundation for practical methods of improvement. The following studies may serve to indicate the relative importance of the utensils as a factor in milk contamination.
Two cans were taken, one of which had been cleaned in the ordinary way, while the other was sterilized by steaming. Before milking, the udder was thoroughly cleaned and special precautions taken to avoid raising of dust; the fore milk was rejected. Milk drawn into these two cans showed the following germ content:
| No. bacteria per cc. | Hours before souring. | |
| Steamed pail | 165 | 28-1/2 |
| Ordinary pail | 426 | 523 |
Harrison[6] has shown how great a variation is in the bacterial content in milk cans. The utensils were rinsed with 100 cc. of sterile water and numerical determinations of this rinsing water made. In poorly cleaned cans, the average germ content was 442,000; in cans washed in tepid water and then scalded—the best farm practice—54,000, and in cans carefully washed and then steamed for five minutes, 880.
Another method used by the writer is to wash the utensil with 100 cc. sterile wash water, using a sterile swab to remove dirt. Then repeat the process twice or more with fresh rinsing waters, making plate cultures from each. The following data were obtained from three such determinations:[Pg 28]
| No. bacteria in different washings. | |||
| I. | II. | III. | Total No. bacteria. |
| 7,800,000 | 1,450,000 | 49,000 | 9,299,000 |
| 283,000 | 43,400 | 35,000 | 361,400 |
| 1,685,000 | 105,000 | 61,400 | 1,851,400 |
Infection of milk in udder cavity. A frequently neglected but considerable factor of infection is that which is attributable to the bacteria which are present in the udder and which are removed in large numbers during the milking process. An examination of the fore milk, i. e., the first few streams from each teat, and that which is subsequently withdrawn, generally reveals a very much larger number of organisms in the fore milk.[7] Not infrequently will this part of the milk when drawn under as careful conditions as possible, contain several score thousand organisms per cc. If successive bacterial determinations are made at different periods of the milking, as shown in the following experiment, a marked diminution is to be noted after the first portion of the milk is removed:
| Fore milk. | 200th cc. | 2000th cc. | 4300th cc. | 6500th cc. | Strippings. | |
| Expt. 1 | 6,500 | 1,700 | 475 | 220 | 75 | 5 |
| Expt. 2 | 8,100 | 1,650 | 400 | 240 | 50 | 10 |
By some observers it has been claimed that it is possible to secure absolutely sterile milk in the strippings but this is rarely so. It is quite probable that such reported results are due to the fact that too small quantities of milk were used in the examinations and so erroneous conclusions were drawn. This marked diminution in numbers indicates that the larger proportion of the organisms found in[Pg 29] the fore milk are present in the lower portion of the udder and milk ducts. When consideration is given to the structure of the udder, it is readily apparent that infection will be greater here than above.
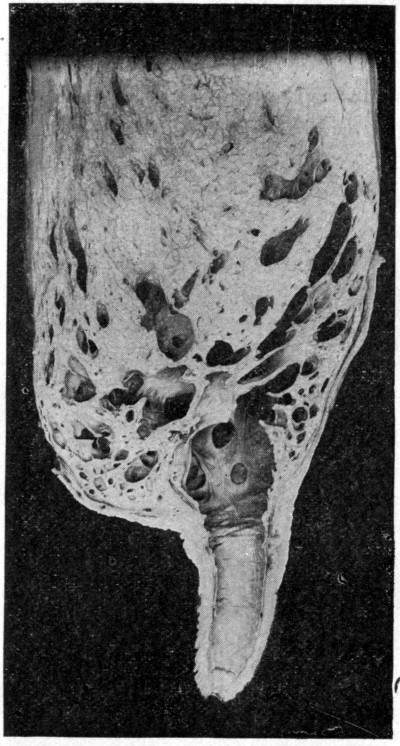 Fig. 8.
Fig. 8.
Sectional view of udder showing teat with milk duct connecting exterior with the milk cistern. Milk sinuses are mostly shown in cross section interspersed and below the secreting tissue (Moore and Ward).]
The udder is composed of secreting tissue (gland cells) held in place by fibrous connective tissue. Ramifying throughout this glandular structure are numerous channels (milk sinuses) that serve to convey the milk from the cells where it is produced into the milk cistern, a common receptacle just above the teats. This cavity is connected with the exterior through the milk duct in the teat, which is more or less tightly closed by the circular sphincter muscles, thus preventing the milk from flowing out. The mucous membranes of the milk duct and cistern are naturally moist. The habits of the animal render it impossible to prevent infection of the[Pg 30] external opening at the end of the teat and there is no mechanical reason why bacteria cannot readily find their way along the moist lining membrane for some distance. If organisms are adapted to this kind of an environment, ideal conditions exist for their multiplication, as moisture, warmth and suitable food supply are present. The question arises how far up into this organ is penetration possible? Within late years numerous observations have been made on the presence of organisms in the upper portion of the udder in contact with the secreting tissue.[8]
These investigations prove that bacteria are distributed throughout the whole of the udder, although numerically they are much less abundant in the region of the milk-secreting tissue than in the lower portion. Ward's conclusions are "that milk when secreted by the glands of a healthy udder is sterile. It may however, immediately become contaminated by the bacteria which are normally present in the smaller milk ducts of the udder."
Nature of bacteria in fore milk. Generally speaking the number of different species found in the fore milk is not large, and of those which do appear many occur at only occasional intervals. Moore[9] in the examination of 9 udders found 20 different forms, and of these only 3 species predominated, all of which proved to be micrococci. Streptococci have also been quite frequently reported. Freudenreich[10] found the most common types to be cocci, belonging to both the liquefying and non-liquefying class.
Peptonizing[11] and spore-bearing[12] species have also been reported as well as gas-producing[13] forms allied to the colon bacillus. Such findings are, however, due in all probability to accidental invasion. Most investigators report the absence of the distinctively lactic-acid group of organisms.[14]
Origin of bacteria in udder. There is no question but that many of the types of bacteria found in the udder gain access from the outside. Those belonging to the spore-bearing, digesting and intestinal types have such a favorable opportunity for introduction from outside and are so unlikely to have come directly from the body of the animal, that the external source of infection is much more probable. Whether this explanation answers the origin of the cocci that are so generally found in the upper portion of the udder is questionable. The statement is ordinarily made that the inner tissues of healthy organs are bacteria-free, but the studies of Ford[15] seem to indicate that 70 per cent. of such organs, removed under aseptic conditions from guinea pigs, rabbits, dogs and cats contained living organisms. Others have reported similar results in which cocci have been found[16] very similar to those occurring in the udder. These findings increase the probability that the origin of this type is from the blood. The persistence[Pg 32] of certain species in the udder for months as noted by Ward indicates possibility of growth of some forms at least. Stocking[17] has shown where cows are not milked clean that the germ content of succeeding milkings is greatly increased.
Artificial introduction of bacteria into udder. If bacteria are capable of actually developing in the udder proper, it ought to be possible to easily demonstrate this by the artificial introduction of cultures. In a number of cases[18] such experiments have been made with various saprophytic forms, such as B. prodigiosus, lactic acid bacilli and others. In no case has it appeared evident that actual growth has occurred, although the introduced organism has been demonstrated in diminishing numbers for 5-6 days. Even the common lactic acid germ and a yellow liquefying coccus isolated from the fore milk failed to persist for more than a few days when thus artificially introduced. This failure to colonize is indeed curious and needs explanation. Is it due to unsuitable environmental conditions or attributable to the germicidal influence of the milk?
Various body fluids are known to possess the property of destroying bacteria and it is claimed by Fokker[19] that this same property was found in freshly drawn milk. This peculiarity has also been investigated by Freudenreich,[20] and Hunziker[21] who find a similar property.
No material increase in germ content takes place in milk for several hours when chilled to 40°-70° F.; on the other[Pg 33] hand an actual, but usually not a marked decrease is observed for about 6 hours. This phenomenon varies with the milk of different cows. Nothing is known as to the cause of this apparent germicidal action. The question is yet by no means satisfactorily settled, although the facts on which the hypothesis is based are not in controversy. If such a peculiarity belongs to milk, it is not at all improbable that it may serve to keep down the germ content in the udder. Freudenreich[22] found that udders which were not examined for some time after death showed abundant growth, which fact he attributed to the loss of this germicidal property.
The infection of the whole milk can be materially reduced by rejecting the fore milk, but it is questionable whether such rejection is worth while, except in the case of "sanitary" dairies where milk is produced with as low a germ content as possible. The intrinsic loss in butter fat in the fore milk is inconsiderable as the first few streams contain only about one-fifth the normal fat content.
Infection of milk after withdrawal from animal. The germ content of the milk, when it is being drawn from the animal is immediately increased upon contact with the atmosphere. These organisms are derived from the surrounding air and the utensils in which the milk is received and stored. The number of organisms which find their way into the milk depends largely upon the character of the surroundings. Bacteria are so intimately associated with dirt, dust and filth of all kinds that wherever the latter are found, the former are sure to be present in abundance.
The most important factors in the infection of the milk after withdrawal are the pollution which is directly traceable[Pg 34] to the animal herself and the condition of the milk utensils. Fortunately both of these sources of contamination are capable of being greatly minimized by more careful methods of handling.
Infection directly from the cow. It is a popular belief that the organisms found in milk are derived from the feed and water which the cow consumes, the same passing directly from the intestinal tract to the milk by the way of the blood circulation. Such a view has no foundation in fact as bacteria absorbed into the circulation are practically all destroyed in the tissues by the action of the body fluids and cells.[23] While organisms cannot pass readily from the intestine to the udder, yet this must not be interpreted as indicating that no attention should be given to the bacterial character of the material consumed. The water supply given should be pure and wholesome and no decomposed or spoiled food should be used.
The infection traceable directly to the cow is modified materially by the conditions under which the animal is kept and the character of the feed consumed. The nature of the fecal matter is in part dependent upon the character of the food. The more nitrogenous rations with which animals are now fed leads to the production of softer fecal discharges which is more likely to soil the coat of the animal unless care is taken. The same is true with animals kept on pasture in comparison with those fed dry fodder.
Stall-fed animals, however, are more likely to have their flanks fouled, unless special attention is paid to the removal of the manure. All dairy stalls should be provided[Pg 35] with a manure drop which should be cleaned as frequently as circumstances will permit.
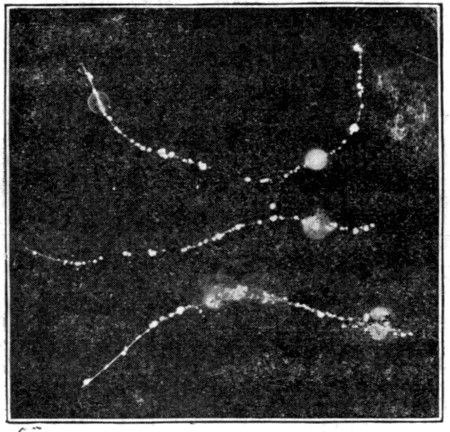 Fig. 9.
Fig. 9.
Showing the bacterial contamination arising from hair. These hairs were allowed to fall on a sterile gelatin surface. The adherent bacteria developed readily in this medium, and the number of bacteria thus introduced into the milk from these hairs can be estimated by the number of developing colonies.]
The animal herself contributes materially to the quota of germ life finding its way into the milk through the dislodgment of dust and filth particles adhering to its hairy coat. The nature of this coat is such as to favor the retention of these particles. Unless care is taken the flanks and udder become polluted with fecal matter, which upon drying is displaced with every movement of the animal. Every hair or dirt particle so dislodged and finding its way into the milk-pail adds its quota of organisms to the liquid. This can be readily demonstrated by placing cow's hairs collected with care on the moist surface of gelatin culture plates. Almost invariably, bacteria will be found in considerable numbers adhering to such hairs as is indicated in Fig. 9. Dirt particles are even richer in germ life. Not only is there the dislodgment of hairs, epithelial scales and masses of dirt and filth, but during[Pg 36] the milking process, as at all other times, every motion of the animal is accompanied by a shower of invisible particles more or less teeming with bacterial life.
The amount of actual impurities found in milk is often considerable and when it is remembered that about one-half of fresh manure dissolves in milk,[24] and thus does not appear as sediment, the presence of this undissolved residue bespeaks filthy conditions as to milking. From actual tests made, it is computed that the city of Berlin, Germany consumes about 300 pounds of such dirt and filth daily. Renk has laid down the following rule with reference to this insoluble dirt: If a sample of milk shows any evidence of impurity settling on a transparent bottom within two hours, it should be regarded as too dirty for use.
While the number of organisms here introduced is at all times large, the character of the species is of even greater import. Derived primarily from dirt and fecal matter, it is no wonder that such forms are able to produce very undesirable fermentative changes.
Influence of milker. The condition of the milker is not to be ignored in determining all possible factors of infection, for when clothed in the dust-laden garments that have been worn all day, a favorable opportunity for direct contamination is possible. The filthy practice of wetting the hands with milk just before milking is to be condemned. The milker's hands should be washed immediately before milking in clean water and dried. A pinch of vaseline on hands is sometimes used to obtain a firmer grasp and prevents the ready dislodgment of scales.[25] It must also be[Pg 37] borne in mind that the milker may spread disease through the milk. In typhoid fever and diphtheria, the germs often remain in the system for weeks and thus make infection possible. Stocking[26] has shown that the individual milker exerts a potent influence on the total germ content of milk, even where the procedure is quite the same. In sanitary dairies milkers are usually clad in white duck suits.
Milking by machinery. Several mechanical devices have been invented for milking, some of which have been tested bacteriologically as to their efficiency. Harrison[27] has examined the "Thistle" machine but found a much higher germ content than with hand-drawn milk. The recent introduction of the Burrel-Lawrence-Kennedy machine has led to numerous tests in which very satisfactory results have been obtained. If the rubber parts of the milker are thoroughly cleaned and kept in lime water solution, they remain nearly sterile. When milk is properly handled, the germ content may be greatly reduced.
Reduction in dirt and adherent bacteria. No factor of contamination is so susceptible of improvement as that which relates to the reduction in filth and dirt which gains access during and immediately subsequent to the milking. The care which is taken to keep the coat of the animal clean by grooming lessens very much the grosser portion of such contamination, but with a dry, hairy coat, fine scales and dust particles must of necessity be dislodged.[28] Ordinarily the patron thinks all evidence of such dirt is removed if the milk is strained, but this process only lessens the difficulty; it does not overcome it. Various methods are in use, the effectiveness of which is subject to[Pg 38] considerable variation. Some of these look to the elimination of the bacteria after they are once introduced into the milk; others to the prevention of infection in the first place.
1. Straining the milk. Most of the visible, solid particles of filth, such as hairs, dirt particles, etc., can be removed by simple straining, the time-honored process of purification. As ordinarily carried out, this process often contributes to instead of diminishing the germ life in milk. The strainer cloths unless washed and thoroughly sterilized by boiling harbor multitudes of organisms from day to day and may thus actually add to the organisms present. Various methods have been suggested for this simple process, but the most practical and efficient strainer is that made of fine wire gauze to which is added 3-4 layers of cheese cloth, the whole to set over the storage milk can.
2. Filtration. In Europe especially, the system of cleaning milk by filtration through sand, gravel and other substances has been quite extensively used. These filters are built in sections and the milk passes from below upward. The filtering substance is washed in hot water immediately after use and then steamed and finally baked. While it is possible to remove the solid impurities in this way, the germ content cannot be greatly reduced.[29] Cellulose filters have also been suggested[30] as an improvement over the sand filters. Methods of filtration of this character have not been used under commercial conditions here in this country.
3. Clarification in separator. Within recent years the custom has grown of clarifying milk or removing the visible[Pg 39] dirt by passing the milk through a centrifugal separator the cream and skim milk being remixed after separation. This process naturally removes the solid impurities as dirt, hairs, epithelial scales and cells, also some of the casein, making what is known as centrifuge slime. This conglomerate mass is incomparably rich in germ life and the natural inference would be that the bacterial content of the milk would be greatly reduced by this procedure. Eckles and Barnes[31] noted a reduction of 37 to 56 per cent. of the bacteria but others have failed to observe such reductions.[32] This condition is explained by the more thorough breaking up of the bacterial masses in the process, thus apparently not reducing them in numbers.
It is somewhat surprising that in spite of the elimination of much organic matter and bacteria, such clarified milk sours as rapidly as the untreated product.[33]
The mechanical shock of separation ruptures the clusters of fat globules and so delays creaming and also lessens the consistency of cream derived from such milk. This practical disadvantage together with the increased expense of the operation and the failure to materially enhance the keeping quality of the product outweigh the advantage which might come from removal of solid impurities which can be largely accomplished on the farm by efficient straining.
4. Washing the udder. If a surface is moist, bacteria adherent to it cannot be dislodged by ordinary movements. Thus the air over snow-covered mountains or oceans is[Pg 40] relatively devoid of germ life. The method of moistening the udder is applied with success to the hairy coat of the animal thus subserving the double purpose of cleaning the animal and preventing in large measure the continual dislodgment of dust particles. After these parts have been well carded to remove loose hairs and dirt particles, the skin should be thoroughly moistened with clean water and then wiped. It has been urged that this procedure lessens the yield of milk but Eckles[34] concludes from experiments that when the animal becomes accustomed to this treatment, no noticeable change in amount of milk or butter-fat is produced.
The effectiveness of this method in reducing the actual amount of dirt and filth introduced into the milk as well as the great diminution in germ life is shown by the instructive experiments of Fraser[35] who found that the actual amount of dirt dislodged from udders of apparently clean animals during the milking process was three and one-half times as much as when the cow's udders were washed. From udders visibly polluted one ounce of such filth was removed in 275 pounds of milk, while from cows whose udders had been washed, the same amount of dirt was distributed through 24,030 pounds.
Fraser observed as a result of 420 examinations that the average germ content of 4-inch culture dishes under clean but unwashed udders was 578, while under washed animals it was reduced to 192. From numerous tests made in the writer's laboratory, it is evident that the germ content of the milk in the pail is increased from 20,000-40,000 bacteria per minute during the milking period. By far the[Pg 41] larger part of this pollution can be easily prevented by cleaning and dampening the udder.
5. Diminishing exposed surface of pail. The entrance of organisms into the milk can be greatly reduced by lessening the area of the milk pail directly exposed to the dust shower. A number of so-called sanitary or hygienic milk pails have been devised for this purpose. In one case the pail is smaller at the top than bottom, but in most of them the common form is kept and the exposed area is lessened by means of a cover, the milk being received through a narrower opening. In some cases, strainers are also interposed so as to remove more effectually the coarse particles. It is necessary to have these covers and strainers constructed in such a way so they can be easily removed and cleaned.
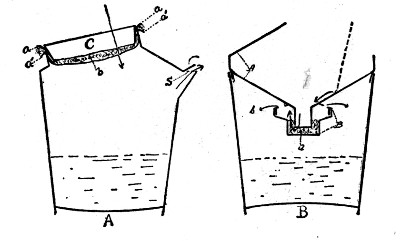 Fig. 10. Sanitary milk pails designed to diminish the
introduction of hairs, scales, dirt, etc., into milk.
Fig. 10. Sanitary milk pails designed to diminish the
introduction of hairs, scales, dirt, etc., into milk.
Stocking tested one of these pails (A, Fig. 10) and found that 63 per cent of the dirt and 29 per cent. of the bacteria were prevented from passing into the milk. Eckles examined one in which the germ content was found to be[Pg 42] 3200 per cc. as against 43200 per cc. in a common open pail. This milk did not sour until it was 64 hours old in the first case while in the latter it curdled in 43 hours.
Air in barn. The atmosphere of the barn where the milking is done may frequently contribute considerable infection. Germ life is incapable of development in the air, but in a dried condition, organisms may retain their vitality for long periods. Anything which contributes to the production of dust in the stable and aids in the stirring up of the same increases the number of organisms to be found in the air (Fig. 11). Thus, the feeding of dry fodder and the bedding of animals with straw adds greatly to the germ life floating in the air. Dust may vary much in its germ content depending upon its origin. Fraser found the[Pg 43] dust from corn meal to contain only about one-sixth to one-eighth as much germ life as that from hay or bran.[36] In time most of these dust particles settle to the floor, but where the herd is kept in the barn, the constant movement of the animals keeps these particles more or less in motion. Much can be done by forethought to lessen the germ content of stables. Feeding dry feed should not be done until after milking.[37] In some of the better sanitary dairies, it is customary to have a special milking room that is arranged with special reference to the elimination of all dust. In this way this source of infection may be quite obviated as the air of a clean, still room is relatively free from bacteria, especially if the floor is moistened. It has often been noted that the milk of stall-fed animals does not keep as well as that milked out of doors, a condition in part attributable to the lessened contamination.
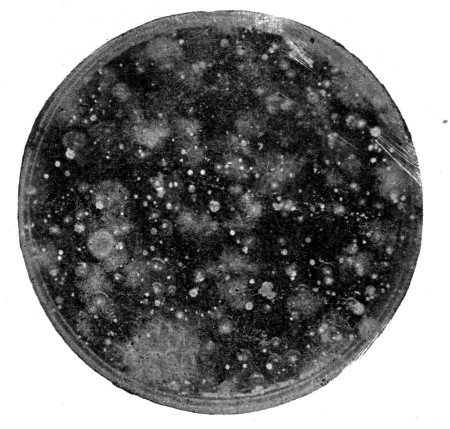 Fig. 11.
Fig. 11.
Effect of contaminated air. The number of spots indicate the colonies that have developed from the bacteria which fell in 30 seconds on the surface of the gelatin plate (3 inches in diameter). This exposure was made at time the cows were fed.
Relative importance of different sources of infection. It is impossible to measure accurately the influence of the different sources of infection as these are continually subject to modification in each and every case. As a general rule, however, where milk is drawn and handled without any special care, the utensils and the animal contribute the larger proportion of dirt and bacteria that find their way into milk. Where the manner of milking and handling is designed to exclude the largest number of organisms possible, the bacteria appearing in the fore milk make up the major portion remaining. By putting into practice the various suggestions that have been made with reference to diminishing the bacterial content of milk, it is possible to greatly reduce the number of organisms found therein, and at the same time materially improve the keeping[Pg 44] quality of the milk. Backhaus[38] estimates that the germ life in milk can be easily reduced to one-two thousandth of its original number by using care in milking. He reports a series of experiments covering two years in which milk was secured that averaged less than 10,000 bacteria per cc., while that secured under ordinary conditions averaged over 500,000.
 Fig. 12.
Fig. 12.
Bacterial content of milk handled in ordinary way. Each spot represents a colony growing on gelatin plate. Compare with Fig. 13, where same quantity of milk is used in making culture. Over 15,000 bacteria per cc. in this milk.
Fig. 13 gives an illustration as to what care in milking will do in the way of eliminating bacteria. Fig. 12 shows a gelatin plate seeded with the same quantity of milk that was used in making the culture indicated by Fig. 13. The first plate was inoculated with milk drawn under good conditions, the germ content of which was found to be[Pg 45] 15,500 bacteria per cc., while the sample secured under as nearly aseptic conditions as possible (Fig. 13) contained only 330 organisms in the same volume.
 Fig. 13.
Fig. 13.
Bacterial content of milk drawn with care. Diminished germ content is shown by smaller number of colonies (330 bacteria per cc.). Compare this culture with that shown in Fig. 12.
"Sanitary" or "certified" milk. Within recent years there has been more or less generally introduced into many cities, the custom of supplying high grade milk that has been handled in a way so as to diminish its germ content as much as possible. Milk of this character is frequently known as "sanitary," "hygienic" or "certified," the last term being used in connection with a certification from veterinary authorities or boards of health as to the freedom of animals from contagious disease. Frequently a numerical bacterial standard is exacted as a pre-requisite[Pg 46] to the recommendation of the board of examining physicians. Thus, the Pediatric Society of Philadelphia requires all children's milk that receives its recommendation to have not more than 10,000 bacteria per cc. Such a standard has its value in the scrupulous cleanliness that must prevail in order to secure these results. This in itself is practically a guarantee of the absence of those bacteria liable to produce trouble in children. The number of organisms found in such milks is surprisingly low when compared with ordinary milk. Naturally, there is considerable fluctuation from day to day, and occasionally the germ content is increased to a high figure without any apparent reason. The average results though, show a greatly reduced number of organisms. De Schweinitz[39] found in a Washington dairy in 113 examinations extending throughout a year, an average of 6,485 bacteria per cc. The daily analyses made of the Walker-Gordon supply sold in Philadelphia for an entire year, showed that the milk almost always contained less than 5,000 bacteria per cc. and on 120 days out of the year the germ content was 1,000 organisms per cc. or less.
From a practical point of view, the improvement in quality of sanitary milk, in comparison with the ordinary product is seen in the enhanced keeping quality. During the Paris Exposition in 1900, milk and cream from several such dairies in the United States were shipped to Paris, arriving in good condition after 15 to 18 days transit. When milk has been handled in such a way, it is evident that it is much better suited to serve as a food supply than where it has undergone the fermentative changes incident to the development of myriads of organisms.[Pg 47]
Application of foregoing precautions to all milk producers. Milk is so susceptible to bacterial changes that it is necessary to protect it from invasion, if its original purity is to be maintained, and yet, from a practical point of view, the use to which it is destined has much to do with the care necessary to take in handling. The effect of the bacterial contamination of milk depends largely upon the way in which the product is used. To the milk-man engaged in the distribution of milk for direct consumption, all bacterial life is more or less of a detriment, while to the butter-maker and cheese-maker some forms are a direct necessity. It is unnecessary and impracticable to require the same degree of care in handling milk destined to be worked up into factory products as is done, for instance, in sanitary milk supplies, but this fact should not be interpreted to mean that the care of milk for factories is a matter of small consequence. In fact no more important dairy problem exists, and the purer and better the quality of the raw material the better the product will be. Particularly is this true with reference to cheese-making.
Dairymen have learned many lessons in the severe school of experience, but it is earnestly to be hoped that future conditions will not be summed up in the words of the eminent German dairy scientist, Prof. Fleischmann, when he says that "all the results of scientific investigation which have found such great practical application in the treatment of disease, in disinfection, and in the preservation of various products, are almost entirely ignored in milking."
Growth of bacteria in milk. Milk is so well suited as a medium for the development of germ life that it might be[Pg 48] expected that all microörganisms would develop rapidly therein, and yet, as a matter of fact, growth does not begin at once, even though the milk may be richly seeded. At ordinary temperatures, such as 70° F., no appreciable increase is to be noted for a period of 6-9 hours; at lower temperatures (54° F.) this period is prolonged to 30-40 hours or even longer. After this period has elapsed, active growth begins and continues more or less rapidly until after curdling.
The cause of this suspended development is attributed to the germicidal properties inherent to the milk.[40]
Milk is of course seeded with a considerable variety of organisms at first. The liquefying and inert species are the most abundant, the distinctively lactic acid class occurring sparsely, if at all. As milk increases in age, germ growth begins to occur. More or less development of all types go on, but soon the lactic species gain the ascendency, owing to their being better suited to this environment; they soon outstrip all other species, with the result that normal curdling generally supervenes. The growth of this type is largely conditioned by the presence of the milk sugar. If the sugar is removed from milk by dialysis, the liquid undergoes putrefactive changes due to the fact that the putrefactive bacteria are able to grow if no acid is produced.
Relation of temperature to growth. When growth does once begin in milk, the temperature at which it is stored exerts the most profound effect on the rate of development. When milk is not artificially cooled, it retains its heat for some hours, and consequently the conditions become very[Pg 49] favorable for the rapid multiplication of the contained organisms, as is shown in following results obtained by Freudenreich[41]:
| 77° F. | 95° F. | ||||
| 5 | hrs. | after | milking | 10,000 | 30,000 |
| 8 | " | " | " | 25,000 | 12,000,000 |
| 12 | " | " | " | 46,000 | 35,280,000 |
| 26 | " | " | " | 5,700,000 | 50,000,000 |
 Fig. 14. Effect of cooling milk on the growth of
bacteria.
Fig. 14. Effect of cooling milk on the growth of
bacteria.
Conn[42] is inclined to regard temperature of more significance in determining the keeping quality than the original infection of the milk itself. Milk which curdled in 18 hours at 98° F., did not curdle in 48 hours at 70°, and often did not change in two weeks, if the temperature was kept at 50° F.
Where kept for a considerable period at this low temperature, the milk becomes filled with bacteria of the undesirable putrefactive type, the lactic group being unable to form acid in any appreciable amounts. Running well[Pg 50] water can be used for cooling, if it is possible to secure it at a temperature of 48°-50° F. The use of ice, of course, gives better results, and in summer is greatly to be desired. The influence of these lowered temperatures makes it possible to ship milk long distances[43] by rail for city supplies, if the temperature is kept low during transit.
Mixing night and morning milk. Not infrequently it happens when old milk is mixed with new, that the course of the fermentative changes is more rapid than would have been the case if the two milks had been kept apart. Thus, adding the cooled night milk to the warm morning milk sometimes produces more rapid changes in both. The explanation for this often imperfectly understood phenomenon is that germ growth may have gone on in the cooled milk, and when this material is added to the warmer, but bacteria-poor, fresh milk, the temperature of the whole mass is raised to a point suitable for the more rapid growth of all bacteria than would have occurred if the older milk had been kept chilled.
Number of bacteria in milk. The number of organisms found in milk depends upon (1) the original amount of contamination, (2) the age of the milk, and (3) the temperature at which it has been held. These factors all fluctuate greatly in different cases; consequently, the germ life is subject to exceedingly wide variations. Here in America, milk reaches the consumer with less bacteria than in Europe, although it may often be older. This is due largely to the more wide-spread use of ice for chilling the milk en route to market. Examinations have been made of various supplies with the following results: Sedgwick and Batchelder[Pg 51] found in 57 tests of Boston milk from 30,000-4,220,000 per cc. Jordan and Heineman found 30% of samples of Chicago milk to range from 100,000 to 1,000,000 while nearly one half were from 1-20,000,000 per cc. The germ content of city milks increase rapidly in the summer months. Park[44] found 250,000 organisms per cc. in winter, about 1,000,000 in cool weather and 5,000,000 per cc. in hot summer weather. Knox and Bassett in Baltimore report 1,500,000 in spring and nearly 4,500,000 in summer. Eckles[45] studied milk under factory conditions. He finds from 1,000,000 to 5,000,000 per cc. in winter, and in summer from 15-30 millions.
Bacterial standards for city supplies. It would be very desirable to have a hygienic standard for city milk supplies, as there is a butter fat and milk-solid test, but the wide spread variation in germ content and the impracticability of utilizing ordinary bacterial determinations (on account of time required) makes the selection of such a standard difficult. Some hold, as Park, that such a standard is feasible. The New York City Milk commission has set a standard of 30,000 bacteria per cc. for their certified milk and 100,000 per cc. for inspected milk. Rochester, N. Y. has attempted the enforcement of such a standard (limit, 100,000 per cc.) with good results it is claimed while Boston has placed the legal limit at 500,000 per cc. Quantitative standards would seem more applicable to "certified" or sanitary supplies than to general city supplies, where the wide range in conditions lead to such enormous variations that the bacterial standard seems too refined a method for practical routine inspection.[Pg 52]
Other tests. Any test to be of much service must be capable of being quickly applied. The writer believes for city milk inspectors that the acid test would serve a very useful purpose. This test measures the acidity of the milk. There is, of course, no close and direct relationship between the development of acidity and the growth of bacteria, yet in a general way one follows the other at normal temperatures. Where the temperature is kept rather low, bacterial growth might go on without much acid development, but in the great majority of cases a high degree of acidity means either old milk, in which there has been a long period of incubation, or high temperature, where rapid bacterial growth has been possible. Either of these conditions encourages germ growth and thus impairs the quality of the milk.
The rapid determination of acidity may be made in an approximate manner so as to serve as a test at the weigh-can or intake. The test is best made by the use of the well known alkaline tablet which is composed of a solid alkali, and the indicator, phenolphthalein. The tablets are dissolved in water, one to each ounce used. A number of white cups are filled with the proper quantity of the solution necessary to neutralize say, 0.2 per cent. lactic acid. Then, as the milk is delivered, the proper quantity is taken from each can to which is added the tablet solution. A retention of the pink color shows that there is not sufficient acid in the milk to neutralize the alkali used; a disappearance of color indicates an excess of acid. The standard selected is of course arbitrarily chosen. In our experience, 0.2 per cent. acidity (figured as lactic), has proven a satisfactory point. With carefully handled milk the acidity ought to be reduced to about 0.15 per cent.[Pg 53] The acidity of the milk may be abnormally reduced if milk is kept in rusty cans, owing to action of acid on the metal.
 Fig. 15.
Fig. 15.
Apparatus used in making rapid acid test. A definite quantity of the alkali solution and indicator is placed in the white tea cup. To this is added the quantity of milk by means of the cartridge measure which would just be neutralized if the acidity was 0.2 per cent. A retention of the pink color shows a low acid milk; its disappearance, a high acid milk.
Kinds of bacteria in milk. The number of bacteria in milk is not of so much consequence as the kinds present. With reference to the number of different species, the more dirt and foreign matter the milk contains, the larger the number of varieties found in the same. While milk may contain forms that are injurious to man, still the great majority of them have no apparent effect on human health.[Pg 54] In their effect on milk, the case is much different. Depending upon their action in milk, they may be grouped into three classes:
1. Inert group, those producing no visible change in the milk.
2. Sour milk forms, those breaking up the milk sugar with or without the formation of gas.
3. Digesting or peptonizing group, those capable of rendering the casein of milk soluble and more or less completely digested.
A surprisingly large number of bacteria that are found in milk belong to the first class. Undoubtedly they affect the chemical characteristics of the milk somewhat, but not to the extent that it becomes physically perceptible. Eckles[46] reports in a creamery supply from 20 to 55 per cent. of entire flora as included in this class.
By far the most important group is that embraced under the second head. It includes not only the true lactic acid types in which no gas is formed, but those species capable of producing gases and various kinds of acids. These organisms are the distinctively milk bacteria, although they do not predominate when the milk is first drawn. Their adaptation to this medium is normally shown, however, by this extremely rapid growth, in which they soon gain the ascendency over all other species present. It is to this lactic acid class that the favorable flavor-producing organisms belong which are concerned in butter-making. They are also indispensable in cheese-making.
The third class represents those capable of producing a liquefied or digested condition on gelatin or in milk. They[Pg 55] are usually the spore-bearing species which gain access from filth and dirt. Their high powers of resistance due to spores makes it difficult to eradicate this type, although they are materially held in subjection by the lactic bacteria. The number of different kinds that have been found in milk is quite considerable, something over 200 species having been described more or less thoroughly. In all probability, however, many of these forms will be found to be identical when they are subjected to a more critical study.
Direct absorption of taints. A tainted condition in milk may result from the development of bacteria, acting upon various constituents of the milk, and transforming these in such a way as to produce by-products that impair the flavor or appearance of the liquid; or it may be produced by the milk being brought in contact with any odoriferous or aromatic substance, under conditions that permit of the direct absorption of such odors.
This latter class of taints is entirely independent of bacterial action, and is largely attributable to the physical property which milk possesses of being able to absorb volatile odors, the fat in particular, having a great affinity for many of these substances. This direct absorption may occur before the milk is withdrawn from the animal, or afterwards if exposed to strong odors.
It is not uncommon for the milk of animals advanced in lactation to have a more or less strongly marked odor and taste; sometimes this is apt to be bitter, at other times salty to the taste. It is a defect that is peculiar to individual animals and is liable to recur at approximately the same period in lactation.[Pg 56]
The peculiar "cowy" or "animal odor" of fresh milk is an inherent peculiarity that is due to the direct absorption of volatile elements from the animal herself. This condition is very much exaggerated when the animal consumes strong-flavored substances as garlic, leeks, turnips and cabbage. The volatile substances that give to these vegetables their characteristic odor are quickly diffused through the system, and if such foods are consumed some few hours before milking, the odor in the milk will be most pronounced. The intensity of such taints is diminished greatly and often wholly disappears, if the milking is not done for some hours (8-12) after such foods are consumed.
This same principle applies in lesser degree to many green fodders that are more suitable as feed for animals, as silage, green rye, rape, etc. Not infrequently, such fodders as these produce so strong a taint in milk as to render it useless for human use. Troubles from such sources could be entirely obviated by feeding limited quantities of such material immediately after milking. Under such conditions the taint produced is usually eliminated before the next milking. The milk of swill-fed cows is said to possess a peculiar taste, and the urine of animals fed on this food is said to be abnormally acid. Brewers' grains and distillery slops when fed in excess also induce a similar condition in the milk.
Milk may also acquire other than volatile substances directly from the animal, as in cases where drugs, as belladonna, castor oil, sulfur, turpentine, jalap, croton oil, and many others have been used as medicine. Such mineral poisons as arsenic have been known to appear eight hours after ingestion, and persist for a period of three weeks before being eliminated.[Pg 57]
Absorption of odors after milking. If milk is brought in contact with strong odors after being drawn from the animal, it will absorb them readily, as in the barn, where frequently it is exposed to the odor of manure and other fermenting organic matter.
It has long been a popular belief that milk evolves odors and cannot absorb them so long as it is warmer than the surrounding air, but from experimental evidence, the writer[47] has definitely shown that the direct absorption of odors takes place much more rapidly when the milk is warm than when cold, although under either condition, it absorbs volatile substances with considerable avidity. In this test fresh milk was exposed to an atmosphere impregnated with odors of various essential oils and other odor-bearing substances. Under these conditions, the cooler milk was tainted very much less than the milk at body temperature even where the period of exposure was brief. It is therefore evident that an exposure in the cow barn where the volatile emanations from the animals themselves and their excreta taint the air will often result in the absorption of these odors by the milk to such an extent as to seriously affect the flavor.
The custom of straining the milk in the barn has long been deprecated as inconsistent with proper dairy practice, and in the light of the above experiments, an additional reason is evident why this should not be done.
Even after milk is thoroughly cooled, it may absorb odors as seen where the same is stored in a refrigerator with certain fruits, meats, fish, etc.
Distinguishing bacterial from non-bacterial taints. In perfectly fresh milk, it is relatively easy to distinguish between[Pg 58] taints caused by the growth of bacteria and those attributable to direct absorption.
If the taint is evident at time of milking, it is in all probability due to character of feed consumed, or possibly to medicines. If, however, the intensity of the taint grows more pronounced as the milk becomes older, then it is probably due to living organisms, which require a certain period of incubation before their fermentative properties are most evident.
Moreover, if the difficulty is of bacterial origin, it can be frequently transferred to another lot of milk (heated or sterilized is preferable) by inoculating same with some of the original milk. Not all abnormal fermentations are able though to compete with the lactic acid bacteria, and hence outbreaks of this sort soon die out by the re-establishment of more normal conditions.
Treatment of directly absorbed taints. Much can be done to overcome taints of this nature by exercising greater care in regard to the feed of animals, and especially as to the time of feeding and milking. But with milk already tainted, it is often possible to materially improve its condition. Thorough aeration has been frequently recommended, but most satisfactory results have been obtained where a combined process of aeration and pasteurization was resorted to. Where the milk is used in making butter, the difficulty has been successfully met by washing the cream with twice its volume of hot water in which a little saltpeter has been dissolved (one teaspoonful per gallon), and then separating it again.[48]
The treatment of abnormal conditions due to bacteria has been given already under the respective sources of infection,[Pg 59] and is also still further amplified in following chapter.
Aeration. It is a common belief that aeration is of great aid in improving the quality of milk, yet, when closely studied, no material improvement can be determined, either where the milk is made into butter or sold as milk. Dean in Canada and Storch in Denmark have both experimented on the influence of aeration in butter making, but with negative results. Marshall and Doane failed to observe any material improvement in keeping quality, but it is true that odors are eliminated from the milk during aeration. The infection of the milk during aeration often more than counterbalances the reputed advantage. Especially is this so if the aeration is carried out in an atmosphere that is not perfectly clean and pure.
In practice aeration differs greatly. In some cases, air is forced into the milk; in others, the milk is allowed to distribute itself in a thin sheet over a broad surface and fall some distance so that it is brought intimately in contact with the air. This latter process is certainly much more effective if carried out under conditions which preclude infection. It must be remembered that aeration is frequently combined with cooling, in which case the reputed advantages may not be entirely attributable to the process of aeration.
Infection of milk in the factory. The problem of proper handling of milk is not entirely solved when the milk is delivered to the factory or creamery, although it might be said that the danger of infection is much greater while the milk is on the farm.
In the factory, infection can be minimized because effective measures of cleanliness can be more easily applied.[Pg 60] Steam is available in most cases, so that all vats, cans, churns and pails can be thoroughly scalded. Special emphasis should be given to the matter of cleaning pumps and pipes. The difficulty of keeping these utensils clean often leads to neglect and subsequent infection. In Swiss cheese factories, the custom of using home-made rennet solutions is responsible for considerable factory infection. Natural rennets are soaked in whey which is kept warm in order to extract the rennet ferment. This solution when used for curdling the milk often adds undesirable yeasts and other gas-generating organisms, which are later the cause of abnormal ferment action in the cheese (See page 186).
The influence of the air on the germ content of the milk is, as a rule, overestimated. If the air is quiet, and free from dust, the amount of germ life in the same is not relatively large. In a creamery or factory, infection from this source ought to be much reduced, for the reason that the floors and wall are, as a rule, quite damp, and hence germ life cannot easily be dislodged. The majority of organisms found under such conditions come from the person of the operators and attendants. Any infection can easily be prevented by having the ripening cream-vats covered with a canvas cloth. The clothing of the operator should be different from the ordinary wearing-apparel. If made of white duck, the presence of dirt is more quickly recognized, and greater care will therefore be taken than if ordinary clothes are worn.
The surroundings of the factory have much to do with the danger of germ infection. Many factories are poorly constructed and the drainage is poor, so that filth and slime collect about and especially under the factory. The emanations from these give the peculiar "factory odor" that indicates fermenting matter. Not only are these odors[Pg 61] absorbed directly, but germ life from the same is apt to find its way into the milk. Connell[49] has recently reported a serious defect in cheese that was traced to germ infection from defective factory drains.
The water supply of a factory is also a question of prime importance. When taken from a shallow well, especially if surface drainage from the factory is possible, the water may be contaminated to such an extent as to introduce undesirable bacteria in such numbers that the normal course of fermentation may be changed. The quality of the water, aside from flavor, can be best determined by making a curd test (p. 76) which is done by adding some of the water to boiled milk and incubating the same. If "gassy" fermentations occur, it signifies an abnormal condition. In deep wells, pumped as thoroughly as is generally the case with factory wells, the germ content should be very low, ranging from a few score to a few hundred bacteria per cc. at most.
Harrison[50] has recently traced an off-flavor in cheese in a Canadian factory to an infection arising from the water-supply. He found the same germ in both water and cheese and by inoculating a culture into pasteurized milk succeeded in producing the undesirable flavor. The danger from ice is much less, for the reason that good dairy practice does not sanction using ice directly in contact with milk or cream. Then, too, ice is largely purified in the process of freezing, although if secured from a polluted source, reliance should not be placed in the method of purification; for even freezing does not destroy all vegetating bacteria.
[1] Olson. 24 Rept. Wis. Expt. Stat., 1907.
[2] Erf and Melick Bull. 131, Kan. Expt. Stat., Apr. 1905.
[3] Storch (40 Rept. Danish Expt. Stat., Copenhagen, 1898) has devised a test whereby it can be determined whether this treatment has been carried out or not: Milk contains a soluble enzym known as galactase which has the property of decomposing hydrogen peroxid. If milk is heated to 176° F. (80° C.) or above, this enzym is destroyed so that the above reaction no longer takes place. If potassium iodid and starch are added to unheated milk and the same treated with hydrogen peroxid, the decomposition of the latter agent releases oxygen which acts on the potassium salt, which in turn gives off free iodine that turns the starch blue.
[4] McKay, N. Y. Prod. Rev., Mch. 22, 1899.
[5] Doane, Bull. 79, Md. Expt. Stat., Jan. 1902.
[6] Harrison, 22 Rept. Ont. Agr'l Coll., 1896, p. 113.
[7] Moore and Ward, Bull. 158, Cornell Expt. Stat., Jan. 1899; Ward, Bull. 178, Cornell Expt. Stat., Jan. 1900.
[8] Harrison, 22 Rept. Ont. Agr. Coll., 1896, p. 108; Moore, 12 Rept. Bur. Animal Ind., U. S. Dept. Ag., 1895-6, p. 261.
[9] Moore, Bacteria in Milk, N. Y. Dept. Ag., 1902.
[10] Freudenreich, Cent. f. Bakt., II Abt., 10: 418, 1903.
[11] Harrison, 22 Rept. Ont. Agr. Coll., 1896, p. 108.
[12] Marshall, Bull. 147, Mich. Expt. Stat., p. 42.
[13] Moore and Ward, Bull. 158, Cornell Expt. Stat., Jan. 1899.
[14] Burr, R. H. Cent. f. Bakt., II Abt., 8: 236, 1902. Freudenreich, l. c. p. 418. Ward, Bull. 178, Cornell Expt. Stat., p. 277. Bolley (Cent. f. Bakt., II Abt., 1: 795, 1895), in 30 experiments found 12 out of 16 species to belong to lactic class. Harrison (Trans. Can. Inst., 7: 474, 1902-3) records the lactic type as most commonly present.
[15] Ford, Journ. of Hyg., 1901, 1: 277.
[16] Freudenreich, l. c. p. 421.
[17] Stocking, Bull. 42, Storrs Expt. Stat., June, 1906.
[18] Dinwiddie, Bull, 45 Ark. Expt. Stat., p. 57. Ward, Journ. Appld. Mic. 1: 205, 1898. Appel, Milch Zeit., No. 17, 1900. Harrison and Cumming, Journ. Appld. Mic. 5: 2087. Russell and Hastings, 21 Rept. Wis. Expt. Stat., 158, 1904.
[19] Fokker, Zeit. f. Hyg., 9: 41, 1890.
[20] Freudenreich, Ann. de Microg., 3: 118, 1891.
[21] Hunziker, Bull. 197, Cornell Expt. Stat., Dec. 1901.
[22] Freudenreich, Cent. f. Bakt., II Abt., 10: 417, 1903.
[23] This general statement is in the main correct, although Ford (Journ. of Hyg., 1: 277, 1901) claims to have found organisms sparingly present in healthy tissues.
[24] Backhaus, Milch Zeit., 26: 357, 1897.
[25] Freudenreich, Die Bakteriologie, p. 30.
[26] Stocking, Bull. 42, Storrs Expt. Stat., June 1906.
[27] Harrison, Cent. f. Bakt., II Abt., 5: 183, 1899.
[28] Drysdale, Trans. High. and Agr. Soc. Scotland. 5 Series, 10: 166, 1898.
[29] Schuppan, (Cent. f. Bakt., 13: 155, 1893) claims to have found a reduction of 48 per cent. in the Copenhagen filters while in the more extended work of Dunbar and Kister (Milch Zeit., pp. 753, 787, 1899) the bacterial content was higher in the filtered milk in 17 cases out of 22.
[30] Backhaus and Cronheim, Journ. f. Landw., 45: 222, 1897.
[31] Eckles and Barnes, Bull. 159 Iowa Expt. Stat., Aug. 1901.
[32] Dunbar and Kister, Milch Zeit., p. 753, 1899. Harrison and Streit, Trans. Can. Inst., 7: 488, 1902-3.
[33] Doane, Bull. 88 Md. Expt. Stat., May 1903.
[34] Eckles, Hoard's Dairyman, July 8, 1898.
[35] Fraser, Bull. 91, Ill. Expt. Stat.
[36] Fraser, Bull. 91, Ill. Expt. Stat., Dec. 1903.
[37] Stocking, Bull. 42, Storrs Expt. Stat., June, 1906.
[38] Backhaus. Ber. Landw. Inst. Univ. Königsberg 2: 12, 1897.
[39] De Schweinitz, Nat. Med. Rev., April, 1899.
[40] Conn, Proc. Soc. Amer. Bacteriologists, 1902.
[41] Freudenreich, Ann. de Microg., 2:115, 1890.
[42] Conn, Bull. 26, Storrs Expt. Stat.
[43] New York City is supplied with milk that is shipped 350 miles.
[44] Park, N. Y. Univ. Bull., 1: 85, 1901.
[45] Eckles, Bull. 59, Iowa Expt. Stat., Aug. 1901.
[46] Eckles, Bull. 59, Iowa Expt. Stat., Aug. 1901.
[47] Russell, 15 Rept. Wis. Expt. Stat. 1898, p. 104.
[48] Alvord, Circ. No. 9, U. S. Dept. Agric. (Div. of Bot.).
[49] Connell, Rept. of Commissioner of Agr., Canada, 1897, part XVI, p. 15.
[50] Harrison, Hoard's Dairyman, March 4, 1898.
Under the conditions in which milk is drawn, it is practically impossible to secure the same without bacterial contamination. The result of the introduction of these organisms often changes its character materially as most bacteria cause the production of more or less pronounced fermentative processes. Under normal conditions, milk sours, i. e., develops lactic acid, but at times this more common fermentation may be replaced by other changes which are marked by the production of some other more or less undesirable flavor, odor or change in appearance.
In referring to these changes, it is usually customary to designate them after the most prominent by-product formed, but it must be kept in mind that generally some other decomposition products are usually produced. Whether the organisms producing this or that series of changes prevail or not depends upon the initial seeding, and the conditions under which the milk is kept. Ordinarily, the lactic acid organisms grow so luxuriantly in the milk that they overpower all competitors and so determine the nature of the fermentation; but occasionally the milk becomes infected with other types of bacteria in relatively large numbers and the conditions may be especially suitable to the development of these forms, thereby modifying the course of the normal changes that occur.[Pg 63]
The kinds of bacteria that find it possible to develop in milk may be included under two heads:
1. Those which cause no appreciable change in the milk, either in taste, odor or appearance. While these are frequently designated as the inert bacteria, it must not be supposed that they have absolutely no effect on milk. It is probably true in most cases that slight changes of a chemical nature are produced, but the nature of the changes do not permit of ready recognition.
2. This class embraces all those organisms which, as a result of their growth, are capable of producing evident changes. These transformations may be such as to affect the taste, as in the sour milk or in the bitter fermentations, or the odor, as in some of the fetid changes, or the appearance of the milk, as in the slimy and color changes later described.
Souring of milk. Ordinarily if milk is allowed to stand for several days at ordinary temperatures it turns sour. This is due to the formation of lactic acid, which is produced by the decomposition of the milk-sugar. While this change is well nigh universal, it does not occur without a pre-existing cause, and that is the presence of certain living bacterial forms. These organisms develop in milk with great rapidity, and the decomposition changes that are noted in souring are due to the by-products of their development.
The milk-sugar undergoes fermentation, the chief product being lactic acid, although various other by-products, as other organic acids (acetic, formic and succinic), different alcohols and gaseous products, as CO2, H, N and methane (CH4) are produced in small amounts.
In this fermentation, the acidity begins to be evident to the taste when it reaches about 0.3 per cent., calculated as lactic acid. As the formation of acid goes on, the casein[Pg 64] is precipitated and incipient curdling or lobbering of the milk occurs. This begins to be apparent when the acidity is about 0.4 per cent., but the curd becomes more solid with increasing acidity. The rapidity of curdling is also dependent upon the temperature of the milk. Thus milk which at ordinary temperatures might remain fluid often curdles when heated. The growth of the bacteria is continued until about 0.8 to 1.0 per cent. acid is formed, although the maximum amount fluctuates considerably with different lactic acid species. Further formation then ceases even though all of the milk-sugar is not used up, because of the inability of the lactic bacteria to continue their growth in such acid solutions.
As this acidity is really in the milk serum, cream never develops so much acid as milk, because a larger proportion of its volume is made up of butter-fat globules. This fact must be considered in the ripening of cream in butter-making where the per cent. of fat is subject to wide fluctuations.
The formation of lactic acid is a characteristic that is possessed by a large number of bacteria, micrococci as well as bacilli being numerously represented. Still the preponderance of evidence is in favor of the view that a few types are responsible for most of these changes. The most common type found in spontaneously soured milk changes the milk-sugar into lactic acid without the production of any gas. This type has been described by various workers on European as well as American milks, and is designated by Conn as the Bact. lactis acidi type.[51] It is subject to considerable variation under different conditions.
Curiously enough if milk which has been drawn with special care is examined immediately after milking, the lactic organisms are not usually found. They are incapable of development in the udder itself, as shown by injections into the milk cistern. They abound, however, on hay, in dust, in the barn air, on the hairy coat of the animal, and from these sources easily gain access to the milk. In this medium they find an exceptionally favorable environment and soon begin a very rapid growth, so that by the time milk is consumed, either in the form of milk or milk products, they make up numerically the larger portion of the bacteria present.
Another widely disseminated, although numerically less prevalent, type is B. lactis aerogenes. This type forms gas in milk so that the soured milk is torn by the presence of gas bubbles. It also grows more luxuriantly in contact with the air.
Other types occur more or less sporadically, some of which are capable of liquefying the casein of milk while at the same time they also develop lactic acid. Conn and Aikman refer to the fact that over one hundred species capable of producing variable quantities of lactic acid are already known. It is fair to presume, however, that a careful comparative study of these would show that simply racial differences exist in many cases, and therefore, that they are not distinct species.
As a group these bacteria are characterized by their inability to liquefy gelatin or develop spores. On account of this latter characteristic they are easily destroyed when milk is pasteurized. They live under aerobic or anaerobic conditions, many of them being able to grow in either environment, although, according to McDonnell,[52] they are more virulent when air is not excluded.
While growth of these lactic forms may go on in milk throughout a relatively wide range in temperature, appreciable quantities of acid are not produced except very slowly at temperatures below 50° F.[53]
From the standpoint of frequency the most common abnormal changes that occur in milk are those in which gases of varying character are developed in connection with acids, from the milk sugar. Other volatile products imparting bad flavors usually accompany gas production. These fermentations are of most serious import in the cheese industry, as they are especially prone to develop in the manufacture of milk into certain types of cheese. Not often is their development so rapid that they appear in the milk while it is yet in the hands of the milk producer, but almost invariably the introduction of the causal organisms takes place while the milk is on the farm. Numerous varieties of bacteria possess this property of producing gas (H and CO2 are most common although N and methane (CH4) are sometimes produced). The more common forms are those represented by B. lactis aerogenes and the common fecal type, B. coli commune. The ordinary habitat of this type is dirt and intestinal filth. Hence careless methods of milk handling invite this type of abnormal change in milk.
It is a wide-spread belief that thunder storms cause milk to sour prematurely, but this idea has no scientific foundation. Experiments[54] with the electric spark, ozone and loud detonations show no effect on acid development, but the atmospheric conditions usually incident to a thunder storm are such as permit of a more rapid growth of organisms. There is no reason to believe but that the phenomenon[Pg 67] of souring is wholly related to the development of bacteria. Sterile milks are never affected by the action of electric storms.
"Gassy" milks. Where these gas bacteria abound, the amount of lactic acid is generally reduced, due to the splitting up of some of the sugar into gaseous products. This type of germ life does not seem to be able to develop well in the presence of the typical lactic acid non gas-forming bacteria.
 Fig. 16. Cheese made from "gassy" milk.
Fig. 16. Cheese made from "gassy" milk.
"Sweet curdling" and digesting fermentations. Not infrequently milk, instead of undergoing spontaneous souring, curdles in a weakly acid or neutral condition, in which state it is said to have undergone "sweet curdling." The coagulation of the milk is caused by the action of enzyms of a rennet type that are formed by the growth of various species of bacteria. Later the whey separates more or less perfectly from the curd, producing a "wheyed off" condition. Generally the coagulum in these cases is soft and[Pg 68] somewhat slimy. The curd usually diminishes in bulk, due to the gradual digestion or peptonization of the casein by proteid-dissolving enzyms (tryptic type) that are also produced by the bacteria causing the change.
A large number of bacteria possess the property of affecting milk in this way. So far as known they are able to liquefy gelatin (also a peptonizing process) and form spores. The Tyrothrix type of bacteria (so named by Duclaux on account of the supposed relation to cheese ripening) belongs to this class. The hay and potato forms are also digesters. Organisms of this type are generally associated with filth and manure, and find their way into the milk from the accumulations on the coat of the animal.
Conn[55] has separated the rennet enzym from bacterial cultures in a relatively pure condition, while Fermi[56] has isolated the digestive ferment from several species.
Duclaux[57] has given to this digesting enzym the name casease or cheese ferment. These isolated ferments when added to fresh milk possess the power of causing the characteristic curdling and subsequent digestion quite independent of cell development. The quantity of ferment produced by different species differs materially in some cases. In these digestive fermentations, the chemical transformations are profound, the complex proteid molecule being broken down into albumoses, peptones, amido-acids (tyrosin and leucin) and ammonia as well as fatty acids.
Not infrequently these fermentations gain the ascendency over the normal souring change, but under ordinary conditions they are held in abeyance, although this type of bacteria is always present to some extent in milk. When[Pg 69] the lactic acid bacteria are destroyed, as in boiled, sterilized or pasteurized milk, these rennet-producing, digesting species develop.
Butyric acid fermentations. The formation of butyric acid in milk which may be recognized by the "rancid butter" odor is not infrequently seen in old, sour milk, and for a long time was thought to be a continuation of the lactic fermentation, but it is now believed that these organisms find more favorable conditions for growth, not so much on account of the lactic acid formed as in the absence of dissolved oxygen in the milk which is consumed by the sour-milk organisms.
Most of the butyric class of bacteria are spore-bearing, and hence they are frequently present in boiled or sterilized milk. The by-products formed in this series of changes are quite numerous. In most cases, butyric acid is prominent, but in addition to this, other organic acids, as lactic, succinic, and acetic, are produced, likewise different alcohols. Concerning the chemical origin of butyric acid there is yet some doubt. Duclaux[58] affirms that the fat, sugar and casein are all decomposed by various forms. In some cases, the reaction of the milk is alkaline, with other species it may be neutral or acid. This type of fermentation has not received the study it deserves.
In milk these organisms are not of great importance, as this fermentation does not readily gain the ascendency over the lactic bacteria.
Ropy or slimy milk. The viscosity of milk is often markedly increased over that which it normally possesses. The intensity of this abnormal condition may vary much; in some cases the milk becoming viscous or slimy; in others[Pg 70] stringing out into long threads, several feet in length, as in Fig. 17. Two sets of conditions are responsible for these ropy or slimy milks. The most common is where the milk is clotted or stringy when drawn, as in some forms of garget. This is generally due to the presence of viscid pus, and is often accompanied by a bloody discharge, such a condition representing an inflamed state of the udder. Ropiness of this character is not usually communicable from one lot of milk to another.
 Fig. 17. Ropy milk.
Fig. 17. Ropy milk.
The communicable form of ropy milk only appears after the milk has been drawn from the udder for a day or so, and is caused by the development of various species of bacteria which find their way into the milk after it is drawn. These defects are liable to occur at any season of the year. Their presence in a dairy is a source of much trouble, as the unsightly appearance of the milk precludes its use as food, although there is no evidence that these ropy fermentations are dangerous to health.
There are undoubtedly a number of different species of bacteria that are capable of producing these viscid changes,[59] but it is quite probable that they are not of equal importance in infecting milk under natural conditions.
In the majority of cases studied in this country,[60] the[Pg 71] causal organism seems to be B. lactis viscosus, a form first found by Adametz in surface waters.[61] This organism possesses the property of developing at low temperatures (45°-50° F.), and consequently it is often able in winter to supplant the lactic-acid forms. Ward has found this germ repeatedly in water tanks where milk cans are cooled; and under these conditions it is easy to see how infection of the milk might occur. Marshall[62] reports an outbreak which he traced to an external infection of the udder; in another case, the slime-forming organism was abundant in the barn dust. A defect of this character is often perpetuated in a dairy for some time, and may therefore become exceedingly troublesome. In one instance in the writer's experience, a milk dealer lost over $150 a month for several months from ropy cream. Failure to properly sterilize cans, and particularly strainer cloths, is frequently responsible for a continuance of trouble of this sort.
The slimy substance formed in milk comes from various constituents of the milk, and the chemical character of the slime produced also varies with different germs. In some cases the slimy material is merely the swollen outer cell membrane of the bacteria themselves as in the case of B. lactis viscosus; in others it is due to the decomposition of the proteids, but often the chief decomposition product appears to come from a viscous fermentation of the milk-sugar.
An interesting case of a fermentation of this class being utilized in dairying is seen in the use of "lange wei" (long or stringy whey) which is employed as a starter in Holland to control the gassy fermentations in Edam cheese.[Pg 72] This slimy change is due to the growth of Streptococcus Hollandicus.[63]
Alcoholic fermentations. Although glucose or cane-sugar solutions are extremely prone to undergo alcoholic fermentation, milk sugar does not readily undergo this change. Where such changes are produced it is due to yeasts. Several outbreaks attributable to such a cause have been reported.[64] Russell and Hastings[65] have found these milk-sugar splitting yeasts particularly abundant in regions where Swiss cheese is made, a condition made possible by the use of whey-soaked rennets in making such cheese.
Kephir and Koumiss are liquors much used in the Orient which are made from milk that has undergone alcoholic fermentation. Koumiss was originally made from mare's milk but is now often made from cows' milk by adding cane sugar and yeast. In addition to the CO2 developed, alcohol, lactic acid, and casein-dissolving ferments are formed. Kephir is made by adding to milk Kephir grains, which are a mass of yeast and bacterial cells. The yeasts produce alcohol and CO2 while the bacteria change the casein of milk, rendering it more digestible. These beverages are frequently recommended to persons who seem to be unable to digest raw milk readily. The exact nature of the changes produced are not yet well understood.[66]
Bitter milk. The presence of bitter substances in milk may be ascribed to a variety of causes. A number of plants, such as lupines, ragweed and chicory, possess the property of affecting milk when the same are consumed by animals.[Pg 73] At certain stages in lactation, a bitter salty taste is occasionally to be noted that is peculiar to individual animals.
A considerable number of cases of bitter milk have, however, been traced to bacterial origin. For a number of years the bitter fermentation of milk was thought to be associated with the butyric fermentation, but Weigmann[67] showed that the two conditions were not dependent upon each other. He found that the organism which produced the bitter taste acted upon the casein.
Conn[68] observed a coccus form in bitter cream that was able to impart a bitter flavor to milk. Sometimes a bitter condition does not develop in the milk, but may appear later in the milk products, as in the case of a micrococcus which Freudenreich[69] found in cheese.
Harrison[70] has traced a common bitter condition in Canadian milk to a milk-sugar splitting yeast, Torula amara which not only grows rapidly in milk but produces an undesirable bitterness in cheddar cheese.
Cream ripened at low temperatures not infrequently develops a bitter flavor, showing that the optimum temperature for this type of fermentation is below the typical lactic acid change.
Milk that has been heated often develops a bitter condition. The explanation of this is that the bacteria producing the bitter substances usually possess endospores, and that while the boiling or sterilizing of milk easily kills the lactic acid germs, these forms on account of their greater resisting powers are not destroyed by the heat.
Soapy milk: A soapy flavor in milk was traced by Weigmann and Zirn[71] to a specific bacillus, B. lactis saponacei, that they found gained access to the milk in one case from the bedding and in another instance from hay. A similar outbreak has been reported in this country,[72] due to a germ acting on the casein and albumen.
Red milk. The most common trouble of this nature in milk is due to presence of blood, which is most frequently caused by some wound in the udder. The ingestion of certain plants as sedges and scouring rushes is also said to cause a bloody condition; madders impart a reddish tinge due to coloring matter absorbed. Defects of this class can be readily distinguished from those due to germ growth because they are apparent at time of milking. Where blood is actually present, the corpuscles settle out in a short time if left undisturbed.
There are a number of chromogenic or color-producing bacteria that are able to grow in milk, but their action is so slow that generally they are not of much consequence. Moreover their development is usually confined to the surface of the milk as it stands in a vessel. The most important is the well-known B. prodigiosus. Another form found at times in milk possessing low acidity[73] is B. lactis erythrogenes. This species only develops the red color in the dark. In the light, it forms a yellow pigment. Various other organisms have been reported at different times.[74]
Blue milk. Blue milk has been known for many years, its communicable nature being established as long ago as 1838. It appears on the surface of milk first as isolated[Pg 75] particles of bluish or grey color, which later become confluent, the blue color increasing in intensity as the acidity increases. The causal organism, B. cyanogenes, is very resistant toward drying,[75] thus accounting for its persistence. In Mecklenberg an outbreak of this sort once continued for several years. It has frequently been observed in Europe in the past, but is not now so often reported. Occasional outbreaks have been reported in this country.
Other kinds of colored milk. Two or three chromogenic forms producing still other colors have occasionally been found in milk. Adametz[76] discovered in a sample of cooked milk a peculiar form (Bacillus synxanthus) that produced a citron-yellow appearance which precipitated and finally rendered soluble the casein. Adametz, Conn, and List have described other species that confer tints of yellow on milk. Some of these are bright lemon, others orange, and some amber in color.
Still other color-producing bacteria, such as those that produce violet or green changes in the milk, have been observed. In fact, almost any of the chromogenic bacteria are able to produce their color changes in milk as it is such an excellent food medium. Under ordinary conditions, these do not gain access to milk in sufficient numbers so that they modify the appearance of it except in occasional instances.
Treatment of abnormal fermentations. If the taint is recognized as of bacterial origin (see p. 57) and is found in the mixed milk of the herd, it is necessary to ascertain, first, whether it is a general trouble, or restricted to one or more animals. This can sometimes be done by separating[Pg 76] the milk of the different cows and noting whether any abnormal condition develops in the respective samples.
Fermentation tests. The most satisfactory way to detect the presence of the taints more often present is to make a fermentation test of one kind or another. These tests are most frequently used at the factory, to enable the maker to detect the presence of milk that is likely to prove unfit for use, especially in cheese making. They are based upon the principle that if milk is held at a moderately high temperature, the bacteria will develop rapidly. A number of different methods have been devised for this purpose. In Walther's lacto-fermentator samples of milk are simply allowed to stand in bottles or glass jars until they sour. They are examined at intervals of several hours. If the curdled milk is homogeneous and has a pure acid smell, the milk is regarded as all right. If it floats in a turbid serum, is full of gas or ragged holes, it is abnormal. As generally carried out, no attempt is made to have these vessels sterile. Gerber's test is a similar test that has been extensively employed in Switzerland. Sometimes a few drops of rennet are added to the milk so as to curdle the same, and thus permit of the more ready detection of the gas that is evolved.
Wisconsin curd test. The method of testing milk described below was devised at the Wisconsin Experiment Station in 1895 by Babcock, Russell and Decker.[77] It was used first in connection with experimental work on the influence of gas-generating bacteria in cheese making, but its applicability to the detection of all taints in milk produced by bacteria makes it a valuable test for abnormal fermentations in general.
In the curd test a small pat of curd is made in a glass[Pg 77] jar from each sample of milk. These tests may be made in any receptacle that has been cleaned in boiling water, and to keep the temperature more nearly uniform these jars should be immersed in warm water, as in a wash tub or some other receptacle. When the milk is about 95° F., about ten drops of rennet extract are added to each sample and mixed thoroughly with the milk. The jars should then remain undisturbed until the milk is completely curdled; then the curd is cut into small pieces with a case knife and stirred to expel the whey. The whey should be poured off at frequent intervals until the curd mats. If the sample be kept at blood heat (98° F.) for six to eight hours, it will be ready to examine.
 Fig. 18.
Fig. 18.
Improved bottles for making curd test. A, test bottle complete; B, bottle showing construction of cover; S, sieve to hold back the curd when bottle is inverted; C, outer cover with (D H) drain holes to permit of removal of whey.]
More convenient types of this test than the improvised apparatus just alluded to have been devised by different dairy manufacturers. Generally, they consist of a special bottle having a full-sized top, thus permitting the easy removal of the curd. The one shown in Fig. 18 is provided[Pg 78] with a sieve of such construction that the bottles will drain thoroughly if inclined in an inverted position.
Interpretation of results of test. The curd from a good milk has a firm, solid texture, and should contain at most only a few small pin holes. It may have some large, irregular, "mechanical" holes where the curd particles have failed to cement, as is seen in Fig. 19. If gas-producing bacteria are very prevalent in the milk, the conditions under which the test is made cause such a rapid growth of the same that the evidence of the abnormal fermentation may be readily seen in the spongy texture of the curd (Fig. 20). If the undesirable organisms are not very abundant and the conditions not especially suited to their growth, the "pin holes" will be less frequent.
 Fig. 19. Curd from a good milk. The large irregular holes
are mechanical.
Fig. 19. Curd from a good milk. The large irregular holes
are mechanical.
Sometimes the curds show no evidence of gas, but their abnormal condition can be recognized by the "mushy" texture and the presence of "off" flavors that are rendered more apparent by keeping them in closed bottles. This condition is abnormal and is apt to produce quite as serious results as if gas was formed.[Pg 79]
Overcoming taints by use of starters. Another method of combatting abnormal fermentations that is often fruitful, is that which rests upon the inability of one kind of bacteria to grow in the same medium in competition with certain other species.
Some of the undesirable taints in factories can be controlled in large part by the introduction of starters made from certain organisms that are able to obtain the ascendency over the taint-producing germ. Such a method is commonly followed when a lactic ferment, either a commercial pure culture, or a home-made starter, is added to milk to overcome the effect of gas-generating bacteria.
 Fig. 20. Curd from a badly tainted milk. Large ragged
holes are mechanical; numerous small holes due to gas. This curd was a
"floater."
Fig. 20. Curd from a badly tainted milk. Large ragged
holes are mechanical; numerous small holes due to gas. This curd was a
"floater."
A similar illustration is seen in the case of the "lange wei" (slimy whey), that is used in the manufacture of Edam cheese to control the character of the fermentation of the milk.
This same method is sometimes applied in dealing with certain abnormal fermentations that are apt to occur on the farm. It is particularly useful with those tainted milks known as "sweet curdling." The ferment organisms concerned[Pg 80] in this change are unable to develop in the presence of lactic acid bacteria, so the addition of a clean sour milk as a starter restores the normal conditions by giving the ordinary milk bacteria the ascendency.
Chemical disinfection. In exceptional instances it may be necessary to employ chemical disinfectants to restore the normal conditions. Of course with such diseases as tuberculosis, very stringent measures are required, as they are such a direct menace to human life, but with these abnormal or taint-producing fermentations, care and cleanliness, well directed, will usually overcome the trouble.
If it becomes necessary to employ chemical substances as disinfecting agents, their use should always be preceded by a thorough cleansing with hot water so that the germicide may come in direct contact with the surface to be disinfected.
It must be borne in mind that many chemicals act as deodorants, i.e., destroy the offensive odor, without destroying the cause of the trouble.
Sulfur is often recommended as a disinfecting agent, but its use should be carefully controlled, otherwise the vapors have but little germicidal power. The common practice of burning a small quantity in a room or any closed space for a few moments has little or no effect upon germ life. The effect of sulfur vapor (SO2) alone upon germ life is relatively slight, but if this gas is produced in the presence of moisture, sulfurous acid (H2SO3) is formed, which is much more efficient. To use this agent effectively, it must be burned in large quantities in a moist atmosphere (three lbs. to every 1,000 cubic feet of space), for at least twelve hours. After this operation, the space should be thoroughly aired.
Formalin, a watery solution of a gas known as formaldehyde, is a new disinfectant that recent experience has[Pg 81] demonstrated to be very useful. It may be used as a gas where rooms are to be disinfected, or applied as a liquid where desired. It is much more powerful in its action than sulfur, and it has a great advantage over mercury and other strong disinfectants, as it is not so poisonous to man as it is to the lower forms of life.
Bleaching powder or chloride of lime is often recommended where a chemical can be advantageously used. This substance is a good disinfectant as well as a deodorant, and if applied as a wash, in the proportion of four to six ounces of the powder to one gallon of water, it will destroy most forms of life. In many cases this agent is inapplicable on account of its odor.
Corrosive sublimate (HgCl2) for most purposes is a good disinfectant, but it is such an intense poison that its use is dangerous in places that are at all accessible to stock.
For the disinfection of walls in stables and barns, common thin white wash Ca(OH)2 is admirably adapted if made from freshly-burned quick lime. It possesses strong germicidal powers, increases the amount of light in the barn, is a good absorbent of odors, and is exceedingly cheap.
Carbolic acid, creosote, and such products, while excellent disinfectants, cannot well be used on account of their odor, especially in factories.
For gutters, drains, and waste pipes in factories, vitriol salts (sulfates of copper, iron and zinc) are sometimes used. These are deodorants as well as disinfectants, and are not so objectionable to use on account of their odor.
These suggestions as to the use of chemicals, however, only apply to extreme cases and should not be brought into requisition until a thorough application of hot water, soap, a little soda, and the scrubbing brush have failed to do their work.
[51] Günther and Thierfelder, Arch. f. Hyg., 25:164, 1895; Leichmann, Cent. f. Bakt., 2:281, 1896; Esten, 9 Rept. Storrs Expt. Stat., p. 44, 1896; Dinwiddie, Bull. 45, Ark. Expt. Stat., May, 1897; Kozai, Zeit. f. Hyg., 38:386, 1901; Weigmann, Hyg. Milk Congress, Hamburg, 1903, p. 375.
[52] McDonnell, Inaug. Diss., Kiel. 1899, p. 39.
[53] Kayser, Cent. f. Bakt. II. Abt. 1:436.
[54] Treadwell, Science, 1894, 17:178.
[55] Conn, 5 Rept. Storrs Expt. Stat., 1892, p. 396.
[56] Fermi, Arch. f. Hyg., 1892, 14:1.
[57] Duclaux, Le Lait, p. 121.
[58] Duclaux, Principes de Laiterie, p. 67.
[59] Guillebeau (Milch Zeit., 1892, p. 808) has studied over a dozen different forms that possess this property.
[60] Ward, Bull. 165, Cornell Expt. Stat., Mch., 1899; also Bull. 195, Ibid., Nov., 1901.
[61] Adametz, Landw. Jahr., 1891, p. 185.
[62] Marshall, Mich. Expt. Stat., Bull. 140.
[63] Milch Zeit., 1899, p. 982.
[64] Duclaux, Principes de Laiterie, p. 60. Heinze and Cohn, Zeit. f. Hyg., 46: 286, 1904.
[65] Bull. 128, Wis. Expt. Stat., Sept. 1905.
[66] Freudenreich, Landw. Jahr. d. Schweiz, 1896, 10; 1.
[67] Weigmann, Milch Zeit., 1890, p. 881.
[68] Conn, 3 Rept. Storrs Expt. Stat., 1890, p. 158.
[69] Freudenreich, Fühl. Landw. Ztg. 43: 361.
[70] Harrison, Bull. 120 Ont. Agr'l. Coll., May, 1902.
[71] Milch Zeit. 22:569.
[72] Marshall, Bull. 146, Mich. Expt. Stat., p. 16.
[73] Grotenfelt, Milch Zeit., 1889, p. 263.
[74] Menge, Cent. f. Bakt., 6:596; Keferstein, Cent. f. Bakt., 21:177.
[75] Heim, Arb. a. d. Kais. Gesundheitsamte, 5:578.
[76] Adametz, Milch Zeit., 1890, p. 225.
[77] 12 Rept. Wis. Expt. Stat., 1895, p. 148; also Bull. 67, Ibid., June, 1898.
Practical experience with epidemic disease has abundantly demonstrated the fact that milk not infrequently serves as a vehicle for the dissemination of contagion. Attention has been prominently called to this relation by Ernest Hart,[78] who in 1880 compiled statistical evidence showing the numerous outbreaks of various contagious diseases that had been associated with milk infection up to that time. Since then, further compilations have been made by Freeman,[79] and also by Busey and Kober,[80] who have collected the data with reference to outbreaks from 1880 to 1899.
These statistics indicate the relative importance of milk as a factor in the dissemination of disease.
The danger from this source is much intensified for the reason that milk, generally speaking, is consumed in a raw state; and also because a considerable number of disease-producing bacteria are able, not merely to exist, but actually thrive and grow in milk, even though the normal milk bacteria are also present. Moreover the recognition of the presence of such pathogenic forms is complicated by the fact that often they do not alter the appearance of[Pg 83] the milk sufficiently so that their presence can be detected by a physical examination. These facts which have been experimentally determined, coupled with the numerous clinical cases on record, make a strong case against milk serving as an agent in the dissemination of disease.
Origin of pathogenic bacteria in milk. Disease-producing bacteria may be grouped with reference to their relation toward milk into two classes, depending upon the manner in which infection occurs:
Class I. Disease-producing bacteria capable of being transmitted directly from a diseased animal to man through the medium of infected milk.
Class II. Bacteria pathogenic for man but not for cattle which are capable of thriving in milk after it is drawn from the animal.
In the first group the disease produced by the specific organism must be common to both cattle and man. The organism must live a parasitic life in the animal, developing in the udder, and so infect the milk supply. It may, of course, happen that diseases toward which domestic animals alone are susceptible may be spread from one animal to another in this way without affecting human beings.
In the second group, the bacterial species lives a saprophytic existence, growing in milk, if it happens to find its way therein. In such cases milk indirectly serves as an agent in the dissemination of disease, by giving conditions favorable to the growth of the disease germ.
By far the most important of diseases that may be transmitted directly from animal to man through a diseased milk supply is tuberculosis, but in addition to this, foot and mouth disease (aphthous fever in children), anthrax and acute enteric troubles have also been traced to a similar source of infection.[Pg 84]
The most important specific diseases that have been disseminated through subsequent pollution of the milk are typhoid fever, diphtheria, scarlet fever and cholera, but, of course, the possibility exists that any disease germ capable of living and thriving in milk may be spread in this way. In addition to these diseases that are caused by the introduction of specific organisms (the causal organism of scarlet fever has not yet been definitely determined), there are a large number of more or less illy-defined troubles of an intestinal character that occur especially in infants and young children that are undoubtedly attributable to the activity of microörganisms that gain access to milk during and subsequent to the milking, and which produce changes in milk before or after its ingestion that result in the formation of toxic products.
Tuberculosis. In view of the wide-spread distribution of this disease in both the human and the bovine race, the relation of the same to milk supplies is a question of great importance. It is now generally admitted that the different types of tubercular disease found in different kinds of animals and man are attributable to the development of the same organism, Bacillus tuberculosis, although there are varieties of this organism found in different species of animals that are sufficiently distinct to permit of recognition.
The question of prime importance is, whether the bovine type is transmissible to the human or not. Artificial inoculation of cattle with tuberculous human sputum as well as pure cultures of this variety show that the human type[Pg 85] is able to make but slight headway in cattle. This would indicate that the danger of cattle acquiring the infection from man would in all probability be very slight, but these experiments offer no answer as to the possibility of transmission from the bovine to the human. Manifestly it is impossible to solve this problem by direct experiment upon man except by artificial inoculation, but comparative experiments upon animals throw some light on the question.
Theo. Smith[81] and others[82] have made parallel experiments with animals such as guinea pigs, rabbits and pigeons, inoculated with both bovine and human cultures of this organism. The results obtained in the case of all animals tested show that the virulence of the two types was much different, but that the bovine cultures were much more severe. While of course this does not prove that transmission from bovine to human is possible, still the importance of the fact must not be overlooked.
In a number of cases record of accidental infection from cattle to man has been noted.[83] These have occurred with persons engaged in making post-mortem examinations on tuberculous animals, and the tubercular nature of the wound was proven in some cases by excision and inoculation.
In addition to data of this sort that is practically experimental in character, there are also strong clinical reasons for considering that infection of human beings may occur through the medium of milk. Naturally such infection should produce intestinal tuberculosis, and it is noteworthy that this phase of the disease is quite common in children[Pg 86] especially between the ages of two and five.[84] It is difficult to determine, though, whether primary infection occurred through the intestine, for, usually, other organs also become involved. In a considerable number of cases in which tubercular infection by the most common channel, inhalation, seems to be excluded, the evidence is strong that the disease was contracted through the medium of the milk, but it is always very difficult to exclude the possibility of pulmonary infection.
Tuberculosis as a bovine disease has increased rapidly during recent decades throughout many portions of the world. This has been most marked in dairy regions. Its extremely insidious nature does not permit of an early recognition by physical means, and it was not until the introduction of the tuberculin test[85] in 1892, as a diagnostic aid that accurate knowledge of its distribution was possible. The quite general introduction of this test in many regions has revealed an alarmingly large percentage of animals as affected. In Denmark in 1894 over forty per cent were diagnosed as tubercular. In some parts of Germany almost as bad a condition has been revealed. Slaughter-house statistics also show that the disease has increased rapidly since 1890. In this country the disease on the average is much less than in Europe and is also very irregularly distributed. In herds where it gained a foothold some years ago, often the majority of animals are frequently infected; many[Pg 87] herds, in fact the great majority, are wholly free from all taint. The disease has undoubtedly been most frequently introduced through the purchase of apparently healthy but incipiently affected animals. Consequently in the older dairy regions where stock has been improved the most by breeding, more of the disease exists than among the western and southern cattle.
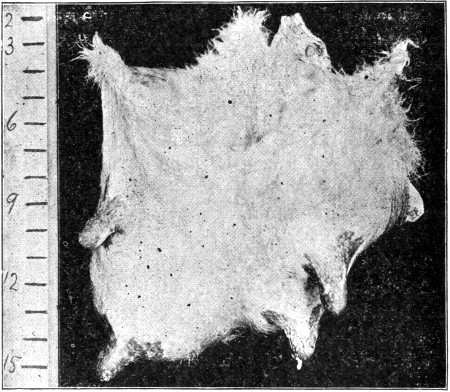 Fig. 21: Front view of a tuberculous udder, showing extent
of swelling in single quarter.
Fig. 21: Front view of a tuberculous udder, showing extent
of swelling in single quarter.
Infectiousness of milk of reacting animals. Where the disease appears in the udder the milk almost invariably contains the tubercle organism. Under such conditions the appearance of the milk is not materially altered at first, but as the disease progresses the percentage of fat generally diminishes, and at times in the more advanced stages where the physical condition of the udder is changed (Fig. 21),[Pg 88] the milk may become "watery"; but the percentage of animals showing such udder lesions is not large, usually not more than a few per cent. (4 per cent. according to Ostertag.)
On the other hand, in the earlier phases of the disease, where its presence has been recognized solely by the aid of the tuberculin test, before there are any recognizable physical symptoms in any part of the animal, the milk is generally unaffected. Between these extremes, however, is found a large proportion of cases, concerning which so definite data are not available. The results of investigators on this point are conflicting and further information is much desired. Some have asserted so long as the udder itself shows no lesions that no tubercle bacilli would be present,[86] but the findings of a considerable number of investigators[87] indicate that even when the udder is apparently not diseased the milk may contain the specific organism as revealed by inoculation experiments upon animals. In some cases, however, it has been demonstrated by post-mortem examination that discoverable udder lesions existed that were not recognizable before autopsy was made. In the experimental evidence collected, a varying percentage of reacting animals were found that gave positive results; and this number was generally sufficient to indicate that the danger of using milk from reacting animals was considerable, even though apparently no disease could be found in the udder.
The infectiousness of milk can also be proven by the frequent contraction of the disease in other animals, such as calves and pigs which may be fed on the skim milk. The very rapid increase of the disease among the swine of Germany[Pg 89] and Denmark,[88] and the frequently reported cases of intestinal infection of young stock also attest the presence of the organism in milk.
The tubercle bacillus is so markedly parasitic in its habits, that, under ordinary conditions, it is incapable of growing at normal air temperatures. There is, therefore, no danger of the germ developing in milk after it is drawn from the animal, unless the same is kept at practically blood heat.
Even though the milk of some reacting animals may not contain the dangerous organism at the time of making the test, it is quite impossible to foretell how long it will remain free. As the disease becomes more generalized, or if tuberculous lesions should develop in the udder, the milk may pass from a healthy to an infectious state.
This fact makes it advisable to exclude from milk supplies intended for human use, all milk of animals that respond to the tuberculin test; or at least to treat it in a manner so as to render it safe. Whether it is necessary to do this or not if the milk is made into butter or cheese is a somewhat different question. Exclusion or treatment is rendered more imperative in milk supplies, because the danger is greater with children with whom milk is often a prominent constituent of their diet, and also for the reason that the child is more susceptible to intestinal infection than the adult.
The danger of infection is much lessened in butter or cheese, because the processes of manufacture tend to diminish the number of organisms originally present in the milk, and inasmuch as no growth can ordinarily take place in these products the danger is minimized. Moreover, the fact that[Pg 90] these foods are consumed by the individual in smaller amounts than is generally the case where milk is used, and also to a greater extent by adults, lessens still further the danger of infection.
Notwithstanding this, numerous observers[89] especially in Germany have succeeded in finding the tubercle bacillus in market butter, but this fact is not so surprising when it is remembered that a very large fraction of their cattle show the presence of the disease as indicated by the tuberculin test, a condition that does not obtain in any large section in this country.
The observations on the presence of the tubercle bacillus in butter have been questioned somewhat of late[2] by the determination of the fact that butter may contain an organism that possesses the property of being stained in the same way as the tubercle organism. Differentiation between the two forms is rendered more difficult by the fact that this tubercle-like organism is also capable of producing in animals lesions that stimulate those of tuberculosis, although a careful examination reveals definite differences. Petri[90] has recently determined that both the true tubercle and the acid-resisting butter organism may be readily found in market butter.
In the various milk products it has been experimentally determined that the true tubercle bacillus is able to retain its vitality in butter for a number of months and in cheese for nearly a year.
Treatment of milk from tuberculosis cows. While it has been shown that it is practically impossible to foretell whether the milk of any reacting animal actually contains[Pg 91] tubercle bacilli or not, still the interests of public health demand that no milk from such stock be used for human food until it has been rendered safe by some satisfactory treatment.
1. Heating. By far the best treatment that can be given such milk is to heat it. The temperature at which this should be done depends upon the thermal death point of the tubercle bacillus, a question concerning which there has been considerable difference of opinion until very recently. According to the work of some of the earlier investigators, the tubercle bacillus in its vegetative stage is endowed with powers of resistance greater than those possessed by any other pathogenic organism. This work has not been substantiated by the most recent investigations on this subject. In determining the thermal death point of this organism, as of any other, not only must the temperature be considered, but the period of exposure as well, and where that exposure is made in milk, another factor must be considered, viz., the presence of conditions permitting of the formation of a "scalded layer," for as Smith[91] first pointed out, the resistance of the tubercle organism toward heat is greatly increased under these conditions. If tuberculous milk is heated in a closed receptacle where this scalded membrane cannot be produced, the tubercle bacillus is killed at 140° F. in 15 to 20 minutes. These results which were first determined by Smith, under laboratory conditions, and confirmed by Russell and Hastings,[92] where tuberculous milk was heated in commercial pasteurizers, have also been verified by Hesse.[93] A great practical advantage which accrues from the treatment of milk at[Pg 92] 140° F. is that the natural creaming is practically unaffected. Of course, where a higher temperature is employed, the period of exposure may be materially lessened. If milk is momentarily heated to 176° F., it is certainly sufficient to destroy the tubercle bacillus. This is the plan practiced in Denmark where all skim milk and whey must be heated to this temperature before it can be taken back to the farm, a plan which is designed to prevent the dissemination of tuberculosis and foot and mouth disease by means of the mixed creamery by-products. This course renders it possible to utilize with perfect safety, for milk supplies, the milk of herds reacting to the tuberculin test, and as butter of the best quality can be made from cream or milk heated to even high temperatures,[94] it thus becomes possible to prevent with slight expense what would otherwise entail a large loss.
2. Dilution. Another method that has been suggested for the treatment of this suspected milk is dilution with a relatively large volume of perfectly healthy milk. It is a well known fact that to produce infection, it requires the simultaneous introduction of a number of organisms, and in the case of tuberculosis, especially that produced by ingestion, this number is thought to be considerable. Gebhardt[95] found that the milk of tuberculous cows, which was virulent when injected by itself into animals, was innocuous when diluted with 40 to 100 times its volume of healthy milk. This fact is hardly to be relied upon in practice, unless the proportion of reacting to healthy cows is positively known.
It has also been claimed in the centrifugal separation of[Pg 93] cream from milk[96] that by far the larger number of tubercle bacilli were thrown out with the separator slime. Moore[97] has shown that the tubercle bacilli in an artificially infected milk might be reduced in this way, so as to be no longer microscopically demonstrable, yet the purification was not complete enough to prevent the infection of animals inoculated with the milk.
Another way to exclude all possibility of tubercular infection in milk supplies is to reject all milk from reacting animals. This method is often followed where pasteurization or sterilization is not desired. In dairies where the keeping quality is dependent upon the exclusion of bacteria by stringent conditions as to milking and handling ("sanitary" or "hygienic" milk), the tuberculin test is frequently used as a basis to insure healthy milk.
Foot and mouth disease. The wide-spread extension of this disease throughout Europe in recent years has given abundant opportunity to show that while it is distinctively an animal malady, it is also transmissible to man, although the disease is rarely fatal. The causal organism has not been determined with certainty, but it has been shown that the milk of affected animals possesses infectious properties[98] although appearing unchanged in earlier phases of the disease.
Hertwig showed the direct transmissibility of the disease to man by experiments made on himself and others. By ingesting milk from an affected animal, he was able to produce the symptoms of the disease, the mucous membrane of the mouth being covered with the small vesicles that characterize the malady. It has also been shown that[Pg 94] the virus of the disease may be conveyed in butter.[99] This disease is practically unknown in this country, although widely spread in Europe.
There are a number of other bovine diseases such as anthrax,[100] lockjaw,[101] and hydrophobia[102] in which it has been shown that the virus of the disease is at times to be found in the milk supply, but often the milk becomes visibly affected, so that the danger of using the same is greatly minimized.
There are also a number of inflammatory udder troubles known as garget or mammitis. In most of these, the physical appearance of the milk is so changed, and often pus is present to such a degree as to give a very disagreeable appearance to the milk. Pus-forming bacteria (staphylococci and streptococci) are to be found associated with such troubles. A number of cases of gastric and intestinal catarrh have been reported as caused by such milks.[103]
Milk is so well adapted to the development of bacteria in general, that it is not surprising to find it a suitable medium for the growth of many pathogenic species even at ordinary temperatures. Not infrequently, disease-producing bacteria are able to grow in raw milk in competition with the normal milk bacteria, so that even a slight contamination may suffice to produce infection.
The diseases that are most frequently disseminated in[Pg 95] this way are typhoid fever, diphtheria, scarlet fever and cholera, together with the various illy-defined intestinal troubles of a toxic character that occur in children, especially under the name of cholera infantum, summer complaint, etc.
Diseases of this class are not derived directly from animals because cattle are not susceptible to the same.
Modes of infection. In a variety of ways, however, the milk may be subject to contaminating influences after it is drawn from the animal, and so give opportunity for the development of disease-producing bacteria. The more important methods of infection are as follows:
1. Infection directly from a pre-existing case of disease on premises. Quite frequently a person in the early stage of a diseased condition may continue at his usual vocation as helper in the barn or dairy, and so give opportunity for direct infection to occur. In the so-called cases of "walking typhoid," this danger is emphasized. It is noteworthy in typhoid fever that the bacilli frequently persist in the urine and in diphtheria they often remain in the throat until after convalescence. In some cases infection has been traced to storage of the milk in rooms in the house where it became polluted directly by the emanations of the patient.[104] Among the dwellings of the lower classes where a single room has to be used in common this source of infection has been most frequently observed.
2. Infection through the medium of another person. Not infrequently another individual may serve in the capacity of nurse or attendant to a sick person, and also assist in the handling of the milk, either in milking the animals or[Pg 96] caring for the milk after it has been drawn. Busey and Kober report twenty-one outbreaks of typhoid fever in which dairy employees also acted in the capacity of nurses.
3. Pollution of milk utensils. The most frequent method of infection of cans, pails, etc., is in cleaning them with water that may be polluted with disease organisms. Often wells may be contaminated with diseased matter of intestinal origin, as in typhoid fever, and the use of water at normal temperatures, or even in a lukewarm condition, give conditions permitting of infection. Intentional adulteration of milk with water inadvertently taken from polluted sources has caused quite a number of typhoid outbreaks.[105] Sedgwick and Chapin[106] found in the Springfield, Mass., epidemic of typhoid that the milk cans were placed in a well to cool the milk, and it was subsequently shown that the well was polluted with typhoid fecal matter.
4. Pollution of udder of animal by wading in infected water, or by washing same with contaminated water. This method of infection would only be likely to occur in case of typhoid. An outbreak at the University of Virginia in 1893[107] was ascribed to the latter cause.
5. Pollution of creamery by-products, skim-milk, etc. Where the milk supply of one patron becomes infected with pathogenic bacteria, it is possible that disease may be disseminated through the medium of the creamery, the infective agent remaining in the skim milk after separation and so polluting the mixed supply. This condition is more likely to prevail with typhoid because of the greater tolerance of this organism for acids such as would be found in raw[Pg 97] milk. The outbreaks at Brandon,[108] England, in 1893, Castle Island,[109] Ireland, and Marlboro,[110] Mass., in 1894, were traced to such an origin.
While most outbreaks of disease associated with a polluted milk supply originate in the use of the milk itself, yet infected milk may serve to cause disease even when used in other ways. Several outbreaks of typhoid fever have been traced to the use of ice cream where there were strong reasons for believing that the milk used in the manufacture of the product was polluted.[111] Hankin[112] details a case of an Indian confection made largely from milk that caused a typhoid outbreak in a British regiment.
Although the evidence that milk may not infrequently serve as an agent in spreading disease is conclusive enough to satisfactorily prove the proposition, yet it should be borne in mind that the organism of any specific disease in question has rarely ever been found. The reasons for this are quite the same as those that govern the situation in the case of polluted waters, except that the difficulties of the problem are much greater in the case of milk than with water. The inability to readily separate the typhoid germ, for instance, from the colon bacillus, an organism frequently found in milk, presents technical difficulties not easily overcome. The most potent reason of failure to find disease bacteria is the fact that infection in any case must occur sometime previous to the appearance of the outbreak. Not only is there the usual period of incubation, but it rarely happens that an outbreak is investigated until a number of[Pg 98] cases have occurred. In this interim the original cause of infection may have ceased to be operative.
Typhoid fever. With reference to the diseases likely to to be disseminated through the medium of milk, infected after being drawn from the animal, typhoid fever is the most important. The reason for this is due (1) to the wide spread distribution of the disease; (2) to the fact that the typhoid bacillus is one that is capable of withstanding considerable amounts of acid, and consequently finds even in raw milk containing the normal lactic acid bacteria conditions favorable for its growth.[113] Ability to grow under these conditions can be shown not only experimentally, but there is abundant clinical evidence that even a slight infection often causes extensive outbreaks, as in the Stamford, Conn., outbreak in 1895 where 386 cases developed in a few weeks, 97 per cent. of which occurred on the route of one milk-man. In this case the milk cans were thoroughly and properly cleaned, but were rinsed out with cold water from a shallow well that was found to be polluted.
The most common mode of pollution of milk with typhoid organisms is where the milk utensils are infected in one way or another.[114] Second in importance is the carrying of infection by persons serving in the dual capacity of nurse and dairy attendant.
Cholera. This germ does not find milk so favorable a nutrient medium as the typhoid organism, because it is much more sensitive toward the action of acids. Kitasato[Pg 99][115] found, however, that it could live in raw milk from one to four days, depending upon the amount of acid present. In boiled or sterilized milk it grows more freely, as the acid-producing forms are thereby eliminated. In butter it dies out in a few days (4 to 5).
On account of the above relation not a large number of cholera outbreaks have been traced to milk, but Simpson[116] records a very striking case in India where a number of sailors, upon reaching port, secured a quantity of milk. Of the crew which consumed this, every one was taken ill, and four out of ten died, while those who did not partake escaped without any disease. It was later shown that the milk was adulterated with water taken from an open pool in a cholera infected district.
Diphtheria. Milk occasionally, though not often, serves as a medium for the dissemination of diphtheria. Swithinbank and Newman[117] cites four cases in which the causal organism has been isolated from milk. It has been observed that growth occurs more rapidly in raw than in sterilized milk.[118]
Infection in this disease is more frequently attributable to direct infection from patient on account of the long persistence of this germ in the throat, or indirectly through the medium of an attendant.
Scarlet fever. Although it is more difficult to study the relation of this disease to contaminated milk supplies, because the causal germ of scarlet fever is not yet known, yet the origin of a considerable number of epidemics has[Pg 100] been traced to polluted milk supplies. Milk doubtless is infected most frequently from persons in the earlier stages of the disease when the infectivity of the disease is greater.
Diarrhoeal diseases. Milk not infrequently acquires the property of producing diseases of the digestive tract by reason of the development of various bacteria that form more or less poisonous by-products. These troubles occur most frequently during the summer months, especially with infants and children, as in cholera infantum and summer complaint. The higher mortality of bottle-fed infants[119] in comparison with those that are nursed directly is explicable on the theory that cows' milk is the carrier of the infection, because in many cases it is not consumed until there has been ample time for the development of organisms in it. Where milk is pasteurized or boiled it is found that the mortality among children is greatly reduced. As a cause of sickness and death these diseases exceed in importance all other specific diseases previously referred to. These troubles have generally been explained as produced by bacteria of the putrefactive class which find their way into the milk through the introduction of filth and dirt at time of milking.[120] Flügge[121] has demonstrated that certain peptonizing species possess toxic properties for animals. Recent experimental inquiry[122] has demonstrated that the dysentery bacillus (Shiga) probably bears a causal relation to some of these summer complaints.
Ptomaine poisoning. Many cases of poisoning from food products are also reported with adults. These are due to the formation of various toxic products, generally ptomaines, that are produced as a result of infection of foods by different bacteria. One of these substances, tyrotoxicon, was isolated by Vaughan[123] from cheese and various other products of milk, and found to possess the property of producing symptoms of poisoning similar to those that are noted in such cases. He attributes the production of this toxic effect to the decomposition of the elements in the milk induced by putrefactive forms of bacteria that develop where milk is improperly kept.[124] Often outbreaks of this character[125] assume the proportions of an epidemic, where a large number of persons use the tainted food.
[78] Hart, Trans. Int. Med. Cong., London, 1881, 4:491-544.
[79] Freeman, Med. Rec., March 28, 1896.
[80] Busey and Kober, Rept. Health Off. of Dist. of Col., Washington, D. C., 1895, p. 299. These authors present in this report an elaborate article on morbific and infectious milk, giving a very complete bibliography of 180 numbers. They append to Hart's list (which is published in full) additional outbreaks which have occurred since, together with full data as to extent of epidemic, circumstances governing the outbreak, as well as name of original reporter and reference.
[81] Smith, Theo., Journ. of Expt. Med., 1898, 3:451.
[82] Dinwiddie, Bull. 57, Ark. Expt. Stat., June, 1899; Ravenel, Univ. of Penn. Med. Bull., Sept. 1901.
[83] Ravenel, Journ. of Comp. Med. & Vet. Arch., Dec. 1897; Hartzell, Journ. Amer. Med. Ass'n, April 16, 1898.
[84] Stille, Brit. Med. Journ., Aug. 19, 1899.
[85] This test is made by injecting into the animal a small quantity of tuberculin, which is a sterilized glycerin extract of cultures of the tubercle bacillus. In a tuberculous animal, even in the very earliest phases of the disease, tuberculin causes a temporary fever that lasts for a few hours. By taking the temperature a number of times before and after injection it is possible to readily recognize any febrile condition. A positive diagnosis is made where the temperature after inoculation is at least 2.0° F. above the average normal, and where the reaction fever is continued for a period of some hours.
[86] Martin, Brit. Med. Journ. 1895, 1:937; Nocard, Les Tuberculoses animales, 1895.
[87] C. O. Jensen, Milch Kunde und Milch hygiene, p. 69.
[88] Ostertag, Milch Zeit., 22:672.
[89] Obermüller, Hyg. Rund., 1897, p. 712; Petri, Arb. a. d. Kais. Ges. Amte, 1898, 14: 1; Hormann und Morgenroth, Hyg. Rund., 1898, p. 217.
[90] Rabinowitsch, Zeit. f. Hyg., 1897, 26: 90.
[91] Th. Smith. Journ. of Expt. Med., 1899, 4:217.
[92] Russell and Hastings, 18 Rept. Wis. Expt. Stat., 1901.
[93] Hesse, Zeit. f. Hyg., 1900, 34:346.
[94] Practically all of the finest butter made in Denmark is made from cream that has been pasteurized at temperatures varying from 160°-185° F.
[95] Gebhardt, Virch. Arch., 1890, 119:12.
[96] Scheurlen, Arb. a. d. k. Ges. Amte, 1891, 7:269; Bang, Milch Zeit., 1893, p. 672.
[97] Moore, Year Book of U. S. Dept. Agr., 1895, p. 432.
[98] Weigel and Noack, Jahres. d. Ges. Med., 1890, p. 642; Weissenberg, Allg. med. Cent. Zeit., 1890, p. 1; Baum, Arch. f. Thierheilkunde, 1892, 18:16.
[99] Schneider, Münch, med. Wochenschr., 1893, No. 27; Fröhner, Zeit f. Fleisch u. Milchhygiene, 1891, p. 55.
[100] Feser, Deutsche Zeit. f. Thiermed., 1880, 6:166.
[101] Nocard, Bull. Gén., 1885, p. 54.
[102] Deutsche Viertelsjahr. f. offentl. Gesundheitspflege, 1890, 20:444.
[103] Zeit. f. Fleisch und Milch hygiene, 11:114.
[104] E. Roth, Deutsche Vierteljahresschr. f. offentl. Gesundheitspfl., 1890, 22:238
[105] S. W. North, London Practitioner, 1889, 43:393.
[106] Sedgwick and Chapin, Boston Med. & Surg. Journ., 1893, 129:485.
[107] Dabney, Phila. Med. News, 1893, 63:630.
[108] Welphy, London Lancet, 1894, 2:1085.
[109] Brit. Med. Journ., 1894, 1:815.
[110] Mass. Bd. Health Rept., 1894, p. 765.
[111] Turner, London Practitioner, 1892, 49:141; Munro, Brit. Med. Journ., 1894, 2:829.
[112] Hankin, Brit. Med. Journ., 1894, 2:613.
[113] Heim (Arb. a. d. Kais. Gesundheitsamte, 1889, 5:303) finds it capable of living from 20-30 days in milk.
[114] Schüder (Zeit. f. Hyg., 1902, 38:34) examined the statistics of 638 typhoid epidemics. He found 71 per cent. due to infected drinking water, 17 per cent. to infected milk, and 3.5 per cent. caused by other forms of food.
[115] Kitasato. Arb. a. d. Kais. Gesundheitsamte, 1:470.
[116] Simpson, London Practitioner, 1887, 39:144.
[117] Swithinbank and Newman, Bacteriology of Milk, p. 341.
[118] Schottelius and Ellerhorst. Milch Zeit., 1897, pp. 40 and 73.
[119] Baginsky, Hyg. Rund., 1895, p. 176.
[120] Gaffky, Deutsch. med. Wochen., 18:14.
[121] Flügge. Zeit., f. Hyg., 17:272, 1894.
[122] Duval and Bassett, Studies from the Rockefeller Inst. for Med. Research, 2:7, 1904.
[123] Zeit. f. physiol. Chemie, 10:146; 9 Intern. Hyg. Cong. (London), 1891, p. 118.
[124] Vaughan and Perkins, Arch. f. Hyg., 27:308.
[125] Newton and Wallace (Phila. Med. News, 1887, 50:570) report three outbreaks at Long Branch, N. J., two of which occurred in summer hotels.
To the milk dealer or distributor, bacteria are more or less of a detriment. None of the organisms that find their way into milk, nor the by-products formed by their growth, improve the quality of milk supplies. It is therefore especially desirable from the milk-dealer's point of view that these changes should be held in abeyance as much as possible. Then too, the possibility that milk may serve as a medium for the dissemination of disease-breeding bacteria makes it advisable to protect this food supply from all possible infection from suspicious sources.
In considering, therefore, the relation of bacteria to general milk supplies, the economic and the hygienic standpoints must be taken into consideration. Ordinarily much more emphasis is laid upon the first requirement. If the supply presents no abnormal feature as to taste, odor and appearance, unfortunately but little attention is paid to the possibility of infection by disease germs. The methods of control which are applicable to general milk supplies are based on the following foundations: (1) the exclusion of all bacterial life, as far as practicable, at the time the milk is drawn, and the subsequent storage of the same at temperatures unfavorable for the growth of the organisms that do gain access; (2) the removal of the bacteria, wholly or in part, after they have once gained access.[Pg 103]
Until within comparatively recent years, practically no attention was given to the character of milk supplies, except possibly as to the percentage of butter fat, and sometimes the milk solids which it contained. So long as the product could be placed in the hands of the consumer in such shape as not to be rejected by him as unfit for food, no further attention was likely to be given to its character. At present, however, much more emphasis is being given to the quality of milk, especially as to its germ content; and the milk dealer is beginning to recognize the necessity of a greater degree of control. This control must not merely concern the handling of the product after it reaches him, but should go back to the milk producer on the farm. Here especially, it is necessary to inculcate those methods of cleanliness which will prevent in large measure the wholesale infection that ordinarily occurs.
The two watch words which are of the utmost importance to the milk dealer are cleanliness and cold. If the milk is properly drawn from the animal in a clean manner and is immediately and thoroughly chilled, the dealer has little to fear as to his product. Whenever serious difficulties do arise, attributable to bacterial changes, it is because negligence has been permitted in one or both directions. The influence of cleanliness in diminishing the bacterial life in milk and that of low temperatures in repressing the growth of those forms which inevitably gain access has been fully dealt with in preceding chapters. It is of course not practicable to take all of these precautions to which reference has been made in the securing of large supplies of market milk for city use, but great improvement over existing conditions could be secured if the public would demand a better supervision of this important food article. Boards of health in[Pg 104] our larger cities are awakening to the importance of this question and are becoming increasingly active in the matter of better regulations and the enforcement of the same.
New York City Board of Health has taken an advanced position in requiring that all milk sold in the city shall be chilled down to 45° F. immediately after milking and shall be transported to the city in refrigerator cars.
Reference has already been made to the application of the acid test (page 52) in the inspection of city milk supplies, and it is the opinion of the writer that the curd test (see page 76) could also be used with advantage in determining the sanitary character of milk. This test reveals the presence of bacteria usually associated with dirt and permits of the recognition of milks that have been carelessly handled. From personal knowledge of examinations made of the milk supplies in a number of Wisconsin cities it appears that this test could be utilized with evident advantage.
"Sanitary" or "certified" milk supplies. In a number of the larger cities, the attempt has been made to improve the quality of the milk supplies by the installation of dairies in which is produced an especially high grade of milk. Frequently the inspection of the dairy as well as the examination of the milk at stated intervals is under the control of milk commissions or medical societies and as it is customary to distribute the certificate of the examining board with the product, such milks are frequently known as "certified." In such dairies the tuberculin test is used at regular intervals, and the herd inspected frequently by competent veterinarians. The methods of control inaugurated as to clean milking and subsequent handling are such as to insure the diminution of the bacteria to the lowest possible point. The bacterial limit set by the Pediatric[Pg 105] Society of Philadelphia is 10,000 organisms per cc. Often it is possible to improve very materially on this standard and not infrequently is the supply produced where it contains only a few thousand organisms per cc. Where such a degree of care is exercised, naturally a considerably higher price must be paid for the product,[126] and it should be remembered that the development of such a system is only possible in relatively large centers where the dealer can cater to a selected high-class trade. Moreover, it should also be borne in mind that such a method of control is only feasible in dairies that are under individual control. The impossibility of exercising adequate control with reference to the milking process and the care which should be given the milk immediately thereafter, when the same is produced on different farms under various auspices is evident.
While much can be done to improve the quality of milk supplies by excluding a large proportion of the bacteria which normally gain access to the milk, and preventing the rapid growth of those that do find their way therein, yet for general municipal purposes, any practical method of preservation[127] that is applicable on a commercial scale must rest largely upon the destruction of bacteria that are present in the milk.
The two possible methods by which bacteria can be destroyed after they have once gained access is (1) by the use of chemical preservatives; (2) by the aid of physical methods.[Pg 106]
Chemical preservatives. Numerous attempts have been made to find some chemical substance that could be added to milk which would preserve it without interfering with its nutritive properties, but as a general rule a substance that is toxic enough to destroy or inhibit the growth of bacterial life exerts a prejudicial effect on the tissues of the body. The use of chemicals, such as carbolic acid, mercury salts and mineral acids, that are able to entirely destroy all life, is of course excluded, except when milk is preserved for analytical purposes; but a number of milder substances are more or less extensively employed, although the statutes of practically all states forbid their use.
The substances so used may be grouped in two classes:
1. Those that unite chemically with certain by-products of bacterial growth to form inert substances. Thus bicarbonate of soda neutralizes the acid in souring milk, although it does not destroy the lactic acid bacteria.
2. Those that act directly upon the bacteria in milk, restraining or inhibiting their development. The substances most frequently utilized are salicylic acid, formaldehyde and boracic acid. These are nearly always sold to the milk handler, under some proprietary name, at prices greatly in excess of what the crude chemicals could be bought for in the open market. Formaldehyde has been widely advertised of late, but its use is fraught with the greatest danger, for it practically renders insoluble all albuminous matter and its toxic effect is greatly increased in larger doses.
These substances are generally used by milk handlers who know nothing of their poisonous action, and although it may be possible for adults to withstand their use in dilute form, without serious results, yet their addition to[Pg 107] general milk supplies that may be used by children is little short of criminal. The sale of these preparations for use in milk finds its only outlet with those dairymen who are anxious to escape the exactions that must be met by all who attempt to handle milk in the best possible manner. Farrington has suggested a simple means for the detection of preservalin (boracic acid).[128] When this substance is added to fresh milk, it increases the acidity of milk without affecting its taste. As normal milk tastes sour when it contains about 0.3 per cent lactic acid, a milk that tests as much or more than this without tasting sour has been probably treated with this antiseptic agent.
Physical methods of preservation. Methods based upon the application of physical forces are less likely to injure the nutritive value of milk, and are consequently more effective, if of any value whatever. A number of methods have been tried more or less thoroughly in an experimental way that have not yet been reduced to a practical basis, as electricity, use of a vacuum, and increased pressure.[129] Condensation has long been used with great success, but in this process the nature of the milk is materially changed. The keeping quality in condensed milk often depends upon the action of another principle, viz., the inhibition of bacterial growth by reason of the concentration of the medium. This condition is reached either by adding sugar and so increasing the soluble solids, or by driving off the water by evaporation, preferably in a vacuum pan. Temperature changes are, however, of the most value in preserving milk, for by a variation in temperature all bacterial growth can be brought to a standstill, and under proper conditions thoroughly destroyed.
Use of low temperatures. The effect of chilling or rapid cooling on the keeping quality of milk is well known. When the temperature of milk is lowered to the neighborhood of 45° F., the development of bacterial life is so slow as to materially increase the period that milk remains sweet. Within recent years, attempts have been made to preserve milk so that it could be shipped long distances by freezing the product, which in the form of milk-ice could be held for an indefinite period without change.[130] A modification of this process known as Casse's system has been in use more or less extensively in Copenhagen and in several places in Germany. This consists of adding a small block of milk-ice (frozen milk) to large cans of milk (one part to about fifty of milk) which may or may not be pasteurized.[131] This reduces the temperature so that the milk remains sweet considerably longer. Such a process might permit of the shipment of milk for long distances with safety but as a matter of fact, the system has not met with especial favor.
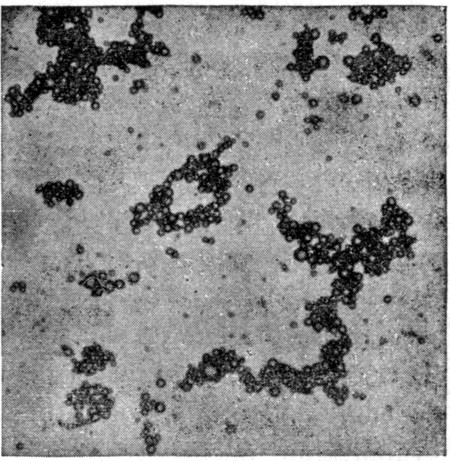 Fig. 22. Microscopic appearance of normal milk showing
the fat-globules aggregated in clusters.
Fig. 22. Microscopic appearance of normal milk showing
the fat-globules aggregated in clusters.
Use of high temperatures. Heat has long been used as a preserving agent. Milk has been scalded or cooked to keep it from time immemorial. Heat may be used at different[Pg 109] temperatures, and when so applied exerts a varying effect, depending upon temperature employed. All methods of preservation by heat rest, however, upon the application of the heat under the following conditions:
1. A temperature above the maximum growing-point (105°-115° F.) and below the thermal death-point (130°-140° F.) will prevent further growth, and consequently fermentative action.
2. A temperature above the thermal death-point destroys bacteria, and thereby stops all changes. This temperature varies, however, with the condition of the bacteria, and for spores is much higher than for vegetative forms.
Attempts have been made to employ the first principle in shipping milk by rail, viz., prolonged heating above growing temperature, but when milk is so heated, its physical appearance is changed.[132] The methods of heating most satisfactorily used are known as sterilization and pasteurization, in which a degree of temperature is used approximating the boiling and scalding points respectively.
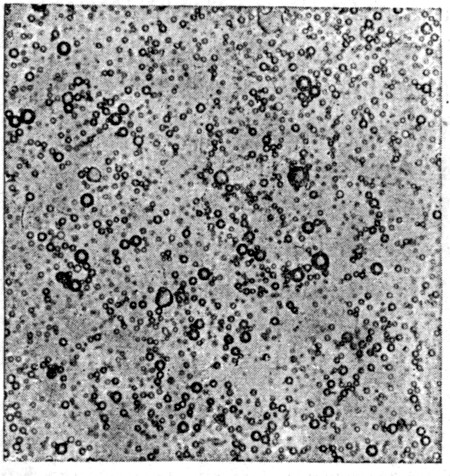 Fig. 23.
Fig. 23.
Microscopic appearance of milk heated above 140° F., showing the homogeneous distribution of fat-globules. The physical change noted in comparison with Fig. 22 causes the diminished consistency of pasteurized cream.]
Effect of heat on milk. When milk is subjected to the action of heat, a number of changes in its physical and chemical properties are to be noted.[Pg 110]
1. Diminished "body." When milk, but more especially cream, is heated to 140° F. or above, it becomes thinner in consistency or "body," a condition which is due to a change in the grouping of the fat globules. In normal milk, the butter fat for the most part is massed in microscopic clots as (Fig. 22). When exposed to 140° F. or above for ten minutes these fat-globule clots break down, and the globules become homogeneously distributed (Fig. 23). A momentary exposure to heat as high as 158°-160° may be made without serious effect on the cream lime; but above this the cream rises so poorly and slowly that it gives the impression of thinner milk.
2. Cooked Taste. If milk is heated for some minutes to 160° F., it acquires a cooked taste that becomes more pronounced as the temperature is further raised. Milk so heated develops on its surface a pellicle or "skin." The cause of this change in taste is not well known. Usually it has been explained as being produced by changes in the nitrogenous elements in the milk, particularly in the albumen. Thoerner[133] has pointed out the coincidence that exists between the appearance of a cooked taste and the loss of certain gases that are expelled by heating. He finds that the milk heated in closed vessels from which the gas cannot escape has a much less pronounced cooked flavor than if heated in an open vessel. The so-called "skin" on the surface of heated milk is not formed when the milk is heated in a tightly-closed receptacle. By some[134] it is asserted that this layer is composed of albumen, but there is[Pg 111] evidence to show that it is modified casein due to the rapid evaporation of the milk serum at the surface of the milk.
3. Digestibility. Considerable difference of opinion has existed in the minds of medical men as to the relative digestibility of raw and heated milks. A considerable amount of experimental work has been done by making artificial digestion experiments with enzyms, also digestion experiments with animals, and in a few cases with children. The results obtained by different investigators are quite contradictory, although the preponderance of evidence seems to be in favor of the view that heating does impair the digestibility of milk, especially if the temperature attains the sterilizing point.[135] It has been observed that there is a noteworthy increase in amount of rickets,[136] scurvy and marasmus in children where highly-heated milks are employed. These objections do not obtain with reference to milk heated to moderate temperatures, as in pasteurization, although even this lower temperature lessens slightly its digestibility. The successful use of pasteurized milks in children's hospitals is evidence of its usefulness.
4. Fermentative changes. The normal souring change in milk is due to the predominance of the lactic acid bacteria, but as these organisms as a class do not possess spores, they are readily killed when heated above the thermal death-point of the developing cell. The destruction of the lactic forms leaves the spore-bearing types possessors of the field, and consequently the fermentative changes in heated milk[Pg 112] are not those that usually occur, but are characterized by the curdling of the milk from the action of rennet enzyms.
5. Action of rennet. Heating milk causes the soluble lime salts to be precipitated, and as the curdling of milk by rennet (in cheese-making) is dependent upon the presence of these salts, their absence in heated milks greatly retards the action of rennet. This renders it difficult to utilize heated milks in cheese-making unless the soluble lime salts are restored, which can be done by adding solutions of calcium chlorid.
Sterilization. As ordinarily used in dairying, sterilization means the application of heat at temperatures approximating, if not exceeding, 212° F. It does not necessarily imply that milk so treated is sterile, i. e., germ-free; for, on account of the resistance of spores, it is practically impossible to destroy entirely all these hardy forms. If milk is heated at temperatures above the boiling point, as is done where steam pressure is utilized, it can be rendered practically germ-free. Such methods are employed where it is designed to keep milk sweet for a long period of time. The treatment of milk by sterilization has not met with any general favor in this country, although it has been more widely introduced abroad. In most cases the process is carried out after the milk is bottled; and considerable ingenuity has been exercised in the construction of devices which will permit of the closure of the bottles after the sterilizing process has been completed. Milks heated to so high a temperature have a more or less pronounced boiled or cooked taste, a condition that does not meet with general favor in this country. The apparatus suitable for this purpose must, of necessity, be so constructed as to withstand steam pressure, and consequently is considerably[Pg 113] more expensive than that required for the simpler pasteurizing process.
Pasteurization. In this method the degree of heat used ranges from 140° to 185° F. and the application is made for only a limited length of time. The process was first extensively used by Pasteur (from whom it derives its name) in combating various maladies of beer and wine. Its importance as a means of increasing the keeping quality of milk was not generally recognized until a few years ago; but the method is now growing rapidly in favor as a means of preserving milk for commercial purposes. The method does not destroy all germ-life in milk; it affects only those organisms that are in a growing, vegetative condition; but if the milk is quickly cooled, it enhances the keeping quality very materially. It is unfortunate that this same term is used in connection with the heating of cream as a preparatory step to the use of pure cultures in cream-ripening in butter-making. The objects to be accomplished vary materially and the details of the two processes are also quite different.
While pasteurizing can be performed on a small scale by the individual, the process can also be adapted to the commercial treatment of large quantities of milk. The apparatus necessary for this purpose is not nearly so expensive as that used in sterilizing, a factor of importance when other advantages are considered. In this country pasteurization has made considerable headway, not only in supplying a milk that is designed to serve as children's food, but even for general purposes.
Requirements essential in pasteurization. While considerable latitude with reference to pasteurizing limits is[Pg 114] permitted, yet there are certain conditions which should be observed, and these, in a sense, fix the limits that should be employed. These may be designated as (1) the physical, and (2) the biological requirements.
Physical requirements. 1. Avoidance of scalded or cooked taste. The English and American people are so averse to a scalded or cooked flavor in milk that it is practically impossible for a highly heated product to be sold in competition with ordinary raw milk. In pasteurization then, care must be taken not to exceed the temperature at which a permanently cooked flavor is developed. As previously observed, this point varies with the period of exposure. A momentary exposure to a temperature of about 170° F. may be made without any material alteration, but if the heat is maintained for a few minutes (ten minutes or over), a temperature of 158° to 160° F. is about the maximum that can be employed with safety.
2. Normal creaming of the milk. It is especially desirable that a sharp and definite cream line be evident on the milk soon after pasteurization. If this fails to appear, the natural inference of the consumer is that the milk is skimmed. If the milk be heated to a temperature sufficiently high to cause the fat-globule clusters to disintegrate (see Figs. 22 and 23), the globules do not rise to the surface as readily as before and the cream line remains indistinct. Where the exposure is made for a considerable period of time (10 minutes or more), the maximum temperature which can be used without producing this change is about 140° F.; if the exposure is made for a very brief time, a minute or less, the milk may be heated to 158°-160 F.° without injuring the creaming property.[Pg 115]
3. No diminution in cream "body." Coincident with this change which takes place in the creaming of the milk is the change in body or consistency which is noted where cream is pasteurized at too high a temperature. For the same reason as given under (2) cream heated above these temperatures is reduced in apparent thickness and appears to contain less butter-fat. Of course the pasteurizing process does not change the fat content, but its "body" is apparently so affected. Thus a 25 per cent. cream may seem to be no thicker or heavier than an 18 per cent. raw cream. This real reduction in consistency naturally affects the readiness with which the cream can be whipped.
Biological requirements. 1. Enhanced keeping quality. In commercial practice the essential biological requirement is expressed in the enhanced keeping quality of the pasteurized milk. This expresses in a practical way the reduction in germ life accomplished by the pasteurizing process. The improvement in keeping quality depends upon the temperature and time of exposure, but fully as much also on the way in which the pasteurized product is handled after heating. The lowest temperature which can be used with success to kill the active, vegetative bacteria is about 140° F., at which point it requires about ten minutes exposure. If this period is curtailed the temperature must be raised accordingly. An exposure to a temperature of 175° F. for a minute has approximately the same effect as the lower degree of heat for the longer time.
The following bacteriological studies as to the effect which a variation in temperature exerts on bacterial life in milk are of importance as indicating the foundation for the selection of the proper limits. In the following[Pg 116] table the exposures were made for a uniform period (20 minutes):
| 45° C. | 50° C. | 55° C. | 60° C. | 65° C. | 70° C. | ||
| Unheated | 113° F. | 122° F. | 131° F. | 140° F. | 149° F. | 158° F. | |
| Series I. | 2,895,000 | —— | 1,260,000 | 798,000 | 32,000 | 5,770 | 3,900 |
| Series II. | 750,000 | 665,000 | 262,400 | 201,000 | 950 | 700 | 705 |
| Series III. | 1,350,000 | 1,100,000 | 260,000 | 215,000 | 575 | 610 | 650 |
| Series IV. | 1,750,000 | —— | 87,360 | —— | 4,000 | 3,500 | 3,600 |
It appears from these results that the most marked decrease in temperature occurs at 140° F. (60° C.). It should also be observed that an increase in heat above this temperature did not materially diminish the number of organisms present, indicating that those forms remaining were in a spore or resistant condition. It was noted, however, that the developing colonies grew more slowly in the plates made from the highly heated milk, showing that their vitality was injured to a greater extent even though not killed.
2. Destruction of disease bacteria. While milk should be pasteurized so as to destroy all active, multiplying bacteria, it is particularly important to destroy any organisms of a disease nature that might find their way into the same. Fortunately most of the bacteria capable of thriving in milk before or after it is drawn from the animal are not able to form spores and hence succumb to proper pasteurization. Such is the case with the diphtheria, cholera and typhoid organisms.
The organism that is invested with most interest in this connection is the tubercle bacillus. On account of its more or less frequent occurrence in milk and its reputed high[Pg 117] powers of resistance, it may well be taken as a standard in pasteurizing.
Thermal death limits of tubercle bacillus. Concerning the exact temperature at which this germ is destroyed there is considerable difference of opinion. Part of this arises from the inherent difficulty in determining exactly when the organism is killed (due to its failure to grow readily on artificial media), and part from the lack of uniform conditions of exposure. The standards that previously have been most generally accepted are those of De Man,[137] who found that thirty minutes exposure at 149° F., fifteen minutes at 155° F., or ten minutes at 167° F., sufficed to destroy this germ.
More recently it has been demonstrated,[138] and these results confirmed,[139] that if tuberculous milk is heated in closed receptacles where the surface pellicle does not form, the vitality of this disease germ is destroyed at 140° F. in 10-15 minutes, while an exposure at 160° F. requires only about one minute.[140] If the conditions of heating are such that the surface of the milk is exposed to the air, the resistance of bacteria is greatly increased. When heated in open vessels Smith found that the tubercle organism was not killed in some cases where the exposure was made for at least an hour. Russell and Hastings[141] have shown an instance where the thermal death-point of a micrococcus isolated from pasteurized milk was increased 12.5° F., by heating it under conditions that permitted of the formation of the scalded layer. It is therefore apparent that apparatus used for[Pg 118] pasteurization should be constructed so as to avoid this defect.
Methods of treatment. Two different systems of pasteurization have grown up in the treatment of milk. One of these has been developed from the hygienic or sanitary aspect of the problem and is used more particularly in the treatment of cream and relatively small milk supplies. The other system has been developed primarily from the commercial point of view where a large amount of milk must be treated in the minimum time. In the first method the milk is heated for a longer period of time, about fifteen minutes at a relatively low temperature from 140°-155° F.; in the other, the milk is exposed to the source of heat only while it is passing rapidly through the apparatus. Naturally, the exposure under such conditions must be made at a considerably higher temperature, usually in the neighborhood of 160° F.
The types of apparatus used in these respective processes naturally varies. Where the heating is prolonged, the apparatus employed is built on the principle of a tank or reservoir in which a given volume of milk may be held at any given temperature for any given period of time.
When the heat is applied for a much shorter period of time, the milk is passed in a continuous stream through the machine. Naturally the capacity of a continuous-flow apparatus is much greater than a machine that operates on the intermittent principle; hence, for large supplies, as in city distribution, this system has a great advantage. The question as to relative efficiency is however one which should be given most careful consideration.[Pg 119]
Pasteurizing apparatus. The problems to be solved in the pasteurization of milk and cream designed for direct consumption are so materially different from where the process is used in butter-making that the type of machinery for each purpose is quite different. The equipment necessary for the first purpose may be divided into two general classes:
1. Apparatus of limited capacity designed for family use.
2. Apparatus of sufficient capacity to pasteurize on a commercial scale.
Domestic pasteurizers. In pasteurizing milk for individual use, it is not desirable to treat at one time more than will be consumed in one day; hence an apparatus holding a few bottles will suffice. In this case the treatment can best be performed in the bottle itself, thereby lessening the danger of infection. Several different types of pasteurizers are on the market; but special apparatus is by no means necessary for the purpose. The process can be efficiently performed by any one with the addition of an ordinary dairy thermometer to the common utensils found in the kitchen. Fig. 24 indicates a simple contrivance that can be readily arranged for this purpose.
The following suggestions indicate the different steps of the process:
1. Use only fresh milk.
2. Place milk in clean bottles or fruit cans, filling to a uniform level, closing bottles tightly with a cork or cover. If pint and quart cans are used at the same time, an inverted bowl will equalize the level. Set these in a flat-bottomed tin pail and fill with warm water to same level as milk. An inverted pie tin punched with holes will serve as a stand on which to place the bottles during the heating process.[Pg 120]
3. Heat water in pail until the temperature of same reaches 155° to 160° F.; then remove from source of direct heat, cover with a cloth or tin cover, and allow the whole to stand for half an hour. In the preparation of milk for children, it is not advisable to use the low-temperature treatment (140° F.) that is recommended for commercial city delivery.
 Fig. 24. A home-made pasteurizer.
Fig. 24. A home-made pasteurizer.
4. Remove bottles of milk and cool them as rapidly as possible without danger to bottles and store in a refrigerator.
Commercial pasteurizers. The two methods of pasteurization practiced commercially for the preservation of milk and cream have been developed because of the two types of machinery now in use. Apparatus constructed on the reservoir or tank principle permits of the retention of the milk for any desired period of time. Therefore, a lower temperature can be employed in the treatment. In[Pg 121] those machines where the milk flows through the heater in a more or less continuous stream, the period of exposure is necessarily curtailed, thereby necessitating a higher temperature.
Reservoir pasteurizers. The simplest type of apparatus suitable for pasteurizing on this principle is where the milk is placed in shotgun cans and immersed in water heated by steam. Ordinary tanks surrounded with water spaces can also be used successfully. The Boyd cream ripening vat has also been tried. In this the milk is heated by a swinging coil immersed in the vat through which hot water circulates.
In 1894 the writer[142] constructed a tank pasteurizer which consisted of a long, narrow vat surrounded by a steam-heated water chamber. Both the milk and the water chambers were provided with mechanical agitators having a to-and-fro movement.
 Fig. 25. Pott's pasteurizer.
Fig. 25. Pott's pasteurizer.
Another machine which has been quite generally introduced is the Potts' rotating pasteurizer. This apparatus has a central milk chamber that is surrounded with an outer shell containing hot water. The whole machine revolves on a horizontal axis, and the cream or milk is thus thoroughly agitated during the heating process.
Continuous-flow pasteurizers. The demand for greater capacity than can be secured in the reservoir machines has led to the perfection of several kinds of apparatus where the milk is heated momentarily as it flows through the apparatus. Most of these were primarily introduced for the treatment of cream for butter-making purposes, but they are frequently employed for the treatment of milk on a large scale in city milk trade. Many of them are of European origin although of late years several have been devised in this country.
The general principle of construction is much the same in most of them. The milk is spread out in a thin sheet, and is treated by passing it over a surface, heated either with steam directly or preferably with hot water.
Where steam is used directly, it is impossible to prevent the "scalding on" of the milk proteids to the heated surface.
In some of these machines (Thiel, Kuehne, Lawrence, De Laval, and Hochmuth), a ribbed surface is employed over which the milk flows, while the opposite surface is heated with hot water or steam. Monrad, Lefeldt and Lentsch employ a centrifugal apparatus in which a thin layer of milk is heated in a revolving drum.
In some types of apparatus, as in the Miller machine, an American pasteurizer, the milk is forced in a thin sheet between two heated surfaces, thereby facilitating the heating process. In the Farrington machine heated discs rotate in a reservoir through which the milk flows in a continuous stream.
One of the most economical types of apparatus is the regenerator type (a German machine), in which the milk passes over the heating surface in a thin stream and then[Pg 123] is carried back over the incoming cold milk so that the heated liquid is partially cooled by the inflowing fresh milk. In machines of this class it requires very much less steam to heat up the milk than in those in which the cold milk is heated wholly by the hot water.
A number of machines have been constructed on the principle of a reservoir which is fed by a constantly flowing stream. In some kinds of apparatus of this type no attempt is made to prevent the mixing of the recently introduced milk with that which has been partially heated. The pattern for this reservoir type is Fjord's heater, in which the milk is stirred by a stirrer. This apparatus was originally designed as a heater for milk before separation, but it has since been materially modified so that it is better adapted to the purposes of pasteurization. Reid was the first to introduce this type of machine into America.
Objections to continuous flow pasteurizers. In all continuous flow pasteurizers certain defects are more or less evident. While they fulfill the important requirement of large capacity, an absolute essential where large volumes of milk are being handled, it does not of necessity follow that they conform to all the hygienic and physical requirements that should be kept in mind. The greatest difficulty is the shortened period of exposure. The period which the milk is actually heated is often not more than a minute or so. Another serious defect is the inability to heat all of the milk for a uniform period of time. At best, the milk is exposed for an extremely short time, but even then portions pass through the machine much more quickly than do the remainder. Those portions in contact with the walls of the apparatus are retarded by friction and are materially delayed in their passage, while the particles in[Pg 124] the center of the stream, however thin, flow through in the least possible time.
The following simple method enables the factory operator to test the period of exposure in the machine: Start the machine full of water, and after the same has become heated to the proper temperature, change the inflow to full-cream milk, continuing at the same rate. Note the exact time of change and also when first evidence of milkiness begins to appear at outflow. If samples are taken from first appearance of milky condition and thereafter at different intervals for several minutes, it is possible, by determining the amount of butter-fat in the same, to calculate with exactness how long it takes for the milk to entirely replace the water.
Tests made by the writer[143] on the Miller pasteurizer showed, when fed at the rate of 1,700 pounds per hour, the minimum period of exposure to be 15 seconds, and the maximum about 60-70 seconds, while about two-thirds of the milk passed the machine in 40-50 seconds. This manifest variation in the rate of flow of the milk through the machine is undoubtedly the reason why the results of this type of treatment are subject to so much variation. Naturally, even a fatal temperature to bacterial life can be reduced to a point where actual destruction of even vegetating cells does not occur.
Bacterial efficiency of reservoir pasteurizers. The bacterial content of pasteurized milk and cream will depend somewhat on the number of organisms originally present in the same. Naturally, if mixed milk brought to a creamery is pasteurized, the number of organisms remaining after treatment would be greater than if the raw material was fresh and produced on a single farm.
An examination of milk and cream pasteurized on a commercial[Pg 125] scale in the Russell vat at the Wisconsin Dairy school showed that over 99.8 per cent of the bacterial life in raw milk or cream was destroyed by the heat employed, i. e., 155° F. for twenty minutes duration.[144] In nearly one-half of the samples of milk, the germ content in the pasteurized sample fell below 1,000 bacteria per cc., and the average of twenty-five samples contained 6,140 bacteria per cc. In cream the germ content was higher, averaging about 25,000 bacteria per cc. This milk was taken from the general creamery supply, which was high in organisms, containing on an average 3,675,000 bacteria per cc. De Schweinitz[145] has reported the germ content of a supply furnished in Washington which was treated at 158° to 160° F. for fifteen minutes. This supply came from a single source. Figures reported were from 48-hour-old agar plates. Undoubtedly these would have been higher if a longer period of incubation had been maintained. The average of 82 samples, taken for the period of one year, showed 325 bacteria per cc.
 Fig. 26. Effect of pasteurizing on germ content of milk.
Black square represents bacteria of raw milk; small white square, those
remaining after pasteurization.
Fig. 26. Effect of pasteurizing on germ content of milk.
Black square represents bacteria of raw milk; small white square, those
remaining after pasteurization.
Bacterial efficiency of continuous-flow pasteurizers. A quantitative determination of the bacteria found in milk and cream when treated in machinery of this class almost always shows a degree of variation in results that is not to be noted in the discontinuous apparatus.
 Fig. 27. Reid's Continuous Pasteurizer.
Fig. 27. Reid's Continuous Pasteurizer.
Harding and Rogers[146] have tested the efficiency of one[Pg 127] of the Danish type of continuous pasteurizers. These experiments were made at 158°, 176° and 185° F. They found the efficiency of the machine not wholly satisfactory at the lower temperatures. At 158° F. the average of fourteen tests gave 15,300 bacteria per cc., with a maximum to minimum range from 62,790 to 120. Twenty-five examinations at 176° F. showed an average of only 117, with a range from 300 to 20. The results at 185° F. showed practically the same results as noted at 176° F. Considerable trouble was experienced with the "scalding on" of the milk to the walls of the machine when milk of high acidity was used.
Jensen[147] details the results of 139 tests in 1899, made by the Copenhagen Health Commission. In 66 samples from one hundred thousand to one million organisms per cc. were found, and in 22 cases from one to five millions. Nineteen tests showed less than 10,000 per cc.
In a series of tests conducted by the writer[148] on a Miller pasteurizer in commercial operation, an average of 21 tests showed 12,350 bacteria remaining in the milk when the milk was pasteurized from 156°-164° F. The raw milk in these tests ran from 115,000 to about one million organisms per cc.
A recently devised machine of this type (Pasteur) has been tested by Lehmann, who found that it was necessary to heat the milk as high as 176° to 185° F., in order to secure satisfactory results on the bacterial content of the cream.
The writer tested Reid's pasteurizer at 155° to 165° F. with the following results: in some cases as many as 40 per cent. of the bacteria survived, which number in some cases exceeded 2,000,000 bacteria per cc.[Pg 128]
Pasteurizing details. While the pasteurizing process is exceedingly simple, yet, in order to secure the best results, certain conditions must be rigidly observed in the treatment before and after the heating process.
It is important to select the best possible milk for pasteurizing, for if the milk has not been milked under clean conditions, it is likely to be rich in the spore-bearing bacteria. Old milk, or milk that has not been kept at a low temperature, is much richer in germ-life than perfectly fresh or thoroughly chilled milk.
The true standard for selecting milk for pasteurization should be to determine the actual number of bacterial spores that are able to resist the heating process, but this method is impracticable under commercial conditions.
The following method, while only approximate in its results, will be found helpful: Assuming that the age or treatment of the milk bears a certain relation to the presence of spores, and that the acid increases in a general way with an increase in age or temperature, the amount of acid present may be taken as an approximate index of the suitability of the milk for pasteurizing purposes. Biological tests were carried out in the author's laboratory[149] on milks having a high and low acid content, and it was shown that the milk with the least acid was, as a rule, the freest from spore-bearing bacteria.
This acid determination can be made at the weigh-can by employing the Farrington alkaline tablet which is used in cream-ripening. Where milk is pasteurized under general creamery conditions, none should be used containing more than 0.2 per cent acidity. If only perfectly fresh milk is used, the amount of acid will generally be about 0.15 per cent with phenolphthalein as indicator.[Pg 129]
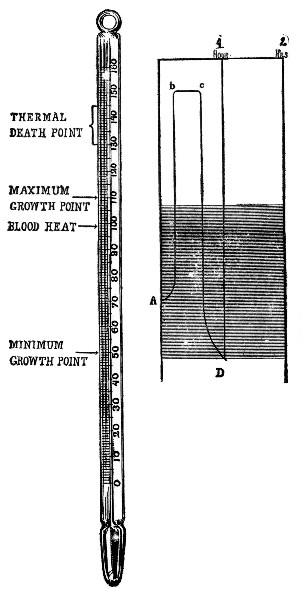 Fig. 28.
Fig. 28.
Diagram showing temperature changes in pasteurizing, and the relation of same to bacterial growth.
Shaded zone represents limits of bacterial growth, 50°-109° F. (10°-43° C.), the intensity of shading indicating rapidity of development. The solid black line shows temperature of milk during the process. The necessity for rapid cooling is evident as the milk falls in temperature to that of growing zone.[Pg 130]]
Emphasis has already been laid on the selection of a proper limit of pasteurizing (p. 114). It should be kept constantly in mind that the thermal death-point of any organism depends not alone on the temperature used, but on the period of exposure. With the lower limits given, 140° F., it is necessary to expose the milk for not less than fifteen minutes. If a higher heat is employed (and the cooked flavor disregarded) the period of exposure may be curtailed.
Chilling the milk. It is very essential in pasteurizing that the heated milk be immediately chilled in order to prevent the germination of the resistant spores, for if germination once occurs, growth can go on at relatively low temperatures.
The following experiments by Marshall[150] are of interest as showing the influence of refrigeration on germination of spores:
Cultures of organisms that had been isolated from pasteurized milk were inoculated into bouillon. One set was left to grow at room temperature, another was pasteurized and allowed to stand at same temperature, while another heated set was kept in a refrigerator. The unheated cultures at room temperature showed evidence of growth in thirty trials in an average of 26 hours; 29 heated cultures at room temperature all developed in an average of 50 hours, while the heated cultures kept in refrigerator showed no growth in 45 days with but four exceptions.
Practically all of the rapid-process machines are provided with especially constructed cooling devices. In some of them, as in the Miller and Farrington, the cooling is effected by passing the milk through two separate coolers that are constructed in the same general way as the heater. With the first cooler, cold running water is employed, the temperature often being lowered in this way to 58° or 60° F. Further lessening of the temperature is secured by an additional ice water or brine cooler which brings the temperature down to 40°-50° F.
In the economical use of ice the ice itself should be applied as closely as possibly to the milk to be cooled, for the larger part of the chilling value of ice comes from the melting of the same. To convert a pound of ice at 32° F. into a pound of water at the same temperature, if we disregard radiation, would require as much heat as would suffice to raise 142 pounds of water one degree F., or one pound of water 142° F. The absorptive capacity of milk for heat (specific heat) is not quite the same as it is with water, being .847 for milk in comparison with 1.0 for water.[151] Hot milk would therefore require somewhat less ice to cool it than would be required by any equal volume of water at the same temperature.
Bottling the product. If the milk has been properly pasteurized, it should, of course, be dispensed in sterilized bottles. Glass bottles with plain pulp caps are best, and these should be thoroughly sterilized in steam before using. The bottling can best be done in a commercial bottling machine. Care must be taken to thoroughly clean this apparatus after use each day. Rubber valves in these machines suffer deterioration rapidly.
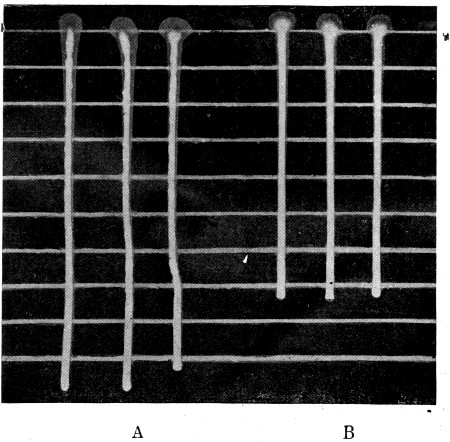 Fig. 29. Relative consistency of pasteurized cream before
(A) and after (B) treatment with viscogen as shown by rate of flow down
inclined glass plate.
Fig. 29. Relative consistency of pasteurized cream before
(A) and after (B) treatment with viscogen as shown by rate of flow down
inclined glass plate.
Restoration of "body" of pasteurized cream. The action of heat causes the tiny groupings of fat globules in normal milk (Fig. 22) to break up, and with this change, which occurs in the neighborhood of 140° F., where the milk is heated for about 15 minutes and at about 160-165° F. where rapidly heated in a continuous stream, the consistency of the liquid is diminished, notwithstanding the fact that the fat-content remains unchanged. Babcock and the writer[152] devised the following "cure" for this apparent defect. If a strong solution of cane sugar is added to[Pg 133] freshly slacked lime and the mixture allowed to stand, a clear fluid can be decanted off. The addition of this alkaline liquid, which is called "viscogen," to pasteurized cream in proportions of about one part of sugar-lime solution to 100 to 150 of cream, restores the consistency of the cream, as it causes the fat globules to cluster together in small groups.
The relative viscosity of creams can easily be determined by the following method (Fig. 29):
Take a perfectly clean piece of glass (plate or picture glass is preferable, as it is less liable to be wavy). Drop on one edge two or three drops of cream at intervals of an inch or so. Then incline piece of glass at such an angle as to cause the cream to flow down surface of glass. The cream, having the heavier body or viscosity, will move more slowly. If several samples of each cream are taken, then the aggregate lengths of the different cream paths may be taken, thereby eliminating slight differences due to condition of glass.
[126] From 10 to 16 cents per quart is usually paid for such milks.
[127] Much improvement in quality could be made by more careful control of milk during shipment, especially as to refrigeration; also as to the care taken on the farms. The use of the ordinary milking machine (see page 37), would go far to reduce the germ content of milk.
[128] Farrington, Journ. Amer. Chem. Soc., Sept., 1896.
[129] Hite, Bull. 58, West Va. Expt. Stat., 1899.
[130] Milch Zeit., 1895, No. 9.
[131] Ibid., 1897, No. 33.
[132] Bernstein, Milch Zeit., 1894, pp. 184, 200.
[133] Thoerner, Chem. Zeit., 18:845.
[134] Snyder, Chemistry of Dairying, p. 59.
[135] Doane and Price (Bull. 77, Md. Expt. Stat., Aug. 1901) give quite a full resumé of the work on this subject in connection with rather extensive experiments made by them on feeding animals with raw, pasteurized and sterilized milks.
[136] Rickets is a disease in which the bones lack sufficient mineral matter to give them proper firmness. Marasmus is a condition in which the ingested food seems to fail to nourish the body and gradual wasting away occurs.
[137] De Man, Arch. f. Hyg., 1893, 18:133.
[138] Th. Smith, Journ. of Expt. Med., 1899, 4:217.
[139] Russell and Hastings, 17 Rept. Wis. Expt. Stat., 1900, p. 147.
[140] Russell and Hastings, 21 Rept. Ibid., 1904.
[141] Russell and Hastings, 18 Rept. Ibid., 1901.
[142] Russell, Bull. 44, Wis. Expt. Stat.
[143] Russell, 22 Wis. Expt. Stat. Rept., 1905, p. 232.
[144] Russell, 12 Wis. Expt. Stat. Rept., 1895, p. 160.
[145] De Schweinitz, Nat. Med. Rev., 1899, No. 11.
[146] Harding and Rogers. Bull. 182, N. Y. (Geneva) Expt. Stat., Dec., 1899.
[147] Jensen, Milchkunde und Milch Hygiene, p. 132.
[148] 22 Wis. Expt. Stat. Rept., 1905, p. 236.
[149] Shockley, Thesis, Univ. of Wis., 1896.
[150] Marshall, Mich. Expt. Stat., Bull. 147, p. 47.
[151] Fleischmann, Landw. Versuchts Stat., 17:251.
[152] Babcock and Russell, Bull. 54, Wis. Expt. Stat., Aug. 1896.
In making butter from the butter fat in milk, it is necessary to concentrate the fat globules into cream, preliminary to the churning process. The cream may be raised by the gravity process or separated from the milk by centrifugal action. In either case the bacteria that are normally present in the milk differentiate themselves in varying numbers in the cream and the skim-milk. The cream always contains per cc. a great many more than the skim-milk, the reason for this being that the bacteria are caught and held in the masses of fat globules, which, on account of their lighter specific gravity, move toward the surface of the milk or toward the interior of the separator bowl. This filtering action of the fat globules is similar to what happens in muddy water upon standing. As the suspended particles fall to the bottom they carry with them a large number of the organisms that are in the liquid.
Various creaming methods. The creaming method has an important bearing on the kind as well as the number of the bacteria that are to be found in the cream. The difference in species is largely determined by the difference in ripening temperature, while the varying number is governed more by the age of the milk.
1. Primitive gravity methods. In the old shallow-pan process, the temperature of the milk is relatively high, as the milk is allowed to cool naturally. This comparatively high temperature favors especially the development of those forms whose optimum growing-point is near the air[Pg 135] temperature. By this method the cream layer is exposed to the air for a longer time than with any other, and consequently the contamination from this source is greater. Usually cream obtained by the shallow-pan process will contain a larger number of species and also have a higher acid content.
2. Modern gravity methods. In the Cooley process, or any of the modern gravity methods where cold water or ice is used to lower the temperature, the conditions do not favor the growth of a large variety of species. The number of bacteria in the cream will depend largely upon the manner in which the milk is handled previous to setting. If care is used in milking, and the milk is kept so as to exclude outside contamination, the cream will be freer from bacteria than if carelessness prevails in handling the milk. Only those forms will develop in abundance that are able to grow at the low temperature at which the milk is set. Cream raised by this method is less frequently infected with undesirable forms than that which is creamed at a higher temperature.
3. Centrifugal method. Separator cream should contain less germ-life than that which is secured in the old way. It should contain only those forms that have found their way into the milk during and subsequent to the milking, for the cream is ordinarily separated so soon that there is but little opportunity of infection, if care is taken in the handling. As a consequence, the number of species found therein is smaller.
Where milk is separated, it is always prudent to cool the cream so as to check growth, as the milk is generally heated before separating in order to skim efficiently.
Although cream is numerically much richer in bacteria[Pg 136] than milk, yet the changes due to bacterial action are slower; hence milk sours more rapidly than cream. For this same reason, cream will sour sooner when it remains on the milk than it will if it is separated as soon as possible. This fact indicates the necessity of early creaming, so as to increase the keeping quality of the product, and is another argument in favor of the separator process.
Ripening of cream. If cream is allowed to remain at ordinary temperatures, it undergoes a series of fermentation changes that are exceedingly complex in character, the result of which is to produce in butter made from the same the characteristic flavor and aroma that are so well known in this article. We are so accustomed to the development of these flavors in butter that they are not generally recognized as being intimately associated with bacterial activity unless compared with butter made from perfectly fresh cream. Sweet-cream butter lacks the aromatic principle that is prominent in the ripened product, and while the flavor is delicate, it is relatively unpronounced.
In the primitive method of butter-making, where the butter was made on the farm, the ripening of cream became a necessity in order that sufficient material might be accumulated to make a churning. The ripening change occurred spontaneously without the exercise of any especial control. With the development of the creamery system came the necessity of exercising a control of this process, and therefore the modern butter-maker must understand the principles which are involved in this series of complex changes that largely give to his product its commercial value.
In these ripening changes three different factors are to be taken into consideration: the development of acid, flavor and aroma. Much confusion in the past has arisen from a[Pg 137] failure to discriminate between these qualities. While all three are produced simultaneously in ordinary ripening, it does not necessarily follow that they are produced by the same cause. If the ripening changes are allowed to go too far, undesirable rather than beneficial decomposition products are produced. These greatly impair the value of butter, so that it becomes necessary to know just to what extent this process should be carried.
In cream ripening there is a very marked bacterial growth, the extent of which is determined mainly by the temperature of the cream. Conn and Esten[153] find that the number of organisms may vary widely in unripened cream, but that the germ content of the ripened product is more uniform. When cream is ready for the churn, it often contains 500,000,000 organisms per cc., and frequently even a higher number. This represents a germ content that has no parallel in any natural material.
The larger proportion of bacteria in cream as it is found in the creamery belong to the acid-producing class, but in the process of ripening, these forms seem to thrive still better, so that when it is ready for churning the germ content of the cream is practically made up of this type.
Effect on churning. In fresh cream the fat globules which are suspended in the milk serum are surrounded by a film of albuminous material which prevents them from coalescing readily. During the ripening changes, this enveloping substance is modified, probably by partial solution, so that the globules cohere when agitated, as in churning. The result is that ripened cream churns more easily, and as it is possible to cause a larger number of the smaller fat-globules to cohere to the butter granules, the yield is[Pg 138] slightly larger—a point of considerable economic importance where large quantities of butter are made.
Development of acid. The result of this enormous bacterial multiplication is that acid is produced in cream, lactic being the principal acid so formed.
Other organic acids are undoubtedly formed as well as certain aromatic products. While the production of acid as a result of fermentative activity is usually accompanied with a development of flavor, the flavor is not directly produced by the formation of acid. If cream is treated in proper proportions with a commercial acid, as hydrochloric,[154] it assumes the same churning properties as found in normally ripened cream, but is devoid of the desired aromatic qualities. Lactic acid[155] has also been used in a similar way but with no better results.
The amount of acidity that should be developed under natural conditions so as to secure the optimum quality as to flavor and aroma is the most important question in cream ripening. Concerning this there have been two somewhat divergent views as to what is best in practice, some holding that better results were obtained with cream ripened to a high degree of acidity than where a less amount was developed.[156] The present tendency seems to be to develop somewhat more than formerly, as it is thought that this secures more of the "high, quick" flavor wanted in the market. On the average, cream is ripened to about 0.5 to 0.65 per cent. acidity, a higher percentage than this giving a strong-flavored butter. In the determination of acidity, the most convenient method is to employ the Farrington[Pg 139] alkaline tablet, which permits of an accurate and rapid estimation of the acidity in the ripening cream. The amount of acidity to be produced must of necessity be governed by the amount of butter-fat present, for the formation of acid is confined to the serum of the cream; consequently, a rich cream would show less acid by titration than a thinner cream, and still contain really as much acid as the other. The importance of this factor is evident in gathered-cream factories.
The rate of ripening is dependent upon the conditions that affect the rate of growth of bacterial life, such as time and temperature, number of organisms in cream and also the per cent of butter fat in the cream. Some years ago it was customary to ripen cream at about 50° to 60° F., but more recently better results have been obtained, it is claimed, where the ripening temperature is increased and the period of ripening lessened. As high a temperature as 70° to 75° F. has been recommended. It should be said that this variation in practice may have a valid scientific foundation, for the temperature of the ripening cream is undoubtedly the most potent factor in determining what kind of bacteria will develop most luxuriantly. It is well known that those forms that are capable of producing bitter flavors are able to thrive better at a lower temperature than some of the desirable ripening species.
The importance of this factor would be lessened where a pure culture was used in pasteurized cream, because here practically the selected organism alone controls the field.
It is frequently asserted that better results are obtained by stirring the cream and so exposing it to the air as much as possible. Experiments made at the Ontario Agricultural College, however, show practically no difference in[Pg 140] the quality of the butter made by these two methods. The great majority of the bacteria in the cream belong to the facultative class, and are able to grow under conditions where they are not in direct contact with the air.
Flavor and aroma. The basis for the peculiar flavor or taste which ripened cream-butter possesses is due, in large part, to the formation of certain decomposition products formed by various bacteria. Aroma is a quality often confounded with flavor, but this is produced by volatile products only, which appeal to the sense of smell rather than taste. Generally a good flavor is accompanied by a desirable aroma, but the origin of the two qualities is not necessarily dependent on the same organisms. The quality of flavor and aroma in butter is, of course, also affected by other conditions, as, for instance, the presence or absence of salt, as well as the inherent qualities of the milk, that are controlled, to some extent at least, by the character of the feed which is consumed by the animal. The exact source of these desirable but evanescent qualities in butter is not yet satisfactorily determined. According to Storch,[157] flavors are produced by the decomposition of the milk sugar and the absorption of the volatile flavors by the butter fat. Conn[158] holds that the nitrogenous elements in cream serve as food for bacteria, and in the decomposition of which the desired aromatic substance is produced. The change is unquestionably a complex one, and cannot be explained as a single fermentation.
There is no longer much doubt but that both acid-forming and casein-digesting species can take part in the production of proper flavors as well as desirable aromas. The researches[Pg 141] of Conn,[159] who has studied this question most exhaustively, indicate that both of these types of decomposition participate in the production of flavor and aroma. He has shown that both flavor and aroma production are independent of acid; that many good flavor-producing forms belong to that class which renders milk alkaline, or do not change the reaction at all. Some of these species liquefied gelatin and would therefore belong to the casein-dissolving class. Those species that produced bad flavors are also included in both fermentative types. Conn has found a number of organisms that are favorable flavor-producers; in fact they were much more numerous than desirable aroma-yielding species. None of the favorable aroma forms according to his investigations were lactic-acid species,—a view which is also shared by Weigmann.[160]
McDonnell[161] has found that the production of aroma in certain cases varies at different temperatures, the most pronounced being evolved near the optimum growing temperature, which, as a general rule, is too high for cream ripening.
The majority of bacteria in ripening cream do not seem to exert any marked influence in butter. A considerable number of species are positively beneficial, inasmuch as they produce a good flavor or aroma. A more limited number are concerned in the production of undesirable ripening changes. This condition being true, it may seem strange that butter is as good as it is, because so frequently the requisite care is not given to the development of proper ripening. In all probability the chief reason why this is so is that those bacteria that find milk and cream pre-eminently[Pg 142] suited to their development, e. g. the lactic-acid class, are either neutral or beneficial in their effect on butter.
Use of starters. Experience has amply demonstrated that it is possible to control the nature of the fermentative changes that occur in ripening cream to such an extent as to materially improve the quality of the butter. This is frequently done by the addition of a "starter." While starters have been employed for many years for the purpose mentioned, it is only recently that their nature has been understood. A starter may be selected from widely divergent sources, but in all cases it is sure to contain a large number of bacteria, and the presumption is that they are of such a nature as to produce desirable fermentative changes in the cream.
In the selection of these so-called natural starters, it follows that they must be chosen under such conditions as experience has shown to give favorable results. For this purpose, whole milk from a single animal is often used where the same is observed to sour with the production of no gas or other undesirable taint. A skim-milk starter from a mixed supply is recommended by many. Butter milk is frequently employed, but in the opinion of butter experts is not as suitable as the others mentioned.
It not infrequently happens that the practical operator may be misled in selecting a starter that is not desirable, or by continuing its use after it has become contaminated.
In 1890[162] a new system of cream ripening was introduced in Denmark by Storch that possesses the merit of being a truly scientific and at the same time practical method. This consisted in the use of pure cultures of[Pg 143] specific organisms that were selected on account of their ability to produce a desirable ripening change in cream. The introduction of these so-called culture starters has become universal in Denmark, and in parts of Germany. Their use is also rapidly extending in this country, Australia and New Zealand.
Principles of pure-culture cream-ripening. In the proper use of pure cultures for ripening cream, it is necessary first to eliminate as far as possible the bacteria already present in cream before the culture starter is added. This result is accomplished by heating the cream to a temperature sufficiently high to destroy the vegetating organisms. The addition of a properly selected starter will then give the chosen organism such an impetus as will generally enable it to gain the ascendency over any other bacteria and so control the character of the ripening. The principle employed is quite like that practiced in raising grain. The farmer prepares his soil by plowing, in this way killing the weeds. Then he sows his selected grain, which is merely a pure culture, and by the rapid growth of this, other forms are held in check.
The attempt has been made to use these culture starters in raw sweet cream, but it can scarcely be expected that the most beneficial results will be attained in this way. This method has been justified on the basis of the following experiments. Where cream is pasteurized and no starter is added, the spore-bearing forms frequently produce undesirable flavors. These can almost always be controlled if a culture starter is added, the obnoxious form being repressed by the presence of the added starter. This condition is interpreted as indicating that the addition of a starter to cream which already contains developing bacteria will[Pg 144] prevent those originally present in the cream from growing.[163] This repressive action of one species on another is a well-known bacteriological fact, but it must be remembered that such an explanation is only applicable in those cases where the culture organism is better able to develop than those forms that already exist in the cream.
If the culture organism is added to raw milk or cream which already contains a flora that is well suited to develop in this medium, it is quite doubtful whether it would gain the supremacy in the ripening cream. The above method of adding a culture to raw cream renders cream-ripening details less burdensome, but at the same time Danish experience, which is entitled to most credence on this question, is opposed to this method.
Reputed advantages of culture starters. 1. Flavor and aroma. Naturally the flavor produced by pure-culture ferments depends upon the character of the organism used. Those which are most extensively used are able to produce a perfectly clean but mild flavor, and a delicate but not pronounced aroma. The "high, quick" flavor and aroma that is so much desired in the American market is not readily obtained by the use of cultures. It is quite problematical whether the use of any single species will give any more marked aroma than normally occurs in natural ripening.
2. Uniformity of product. Culture starters produce a more uniform product because the type of fermentation is under more complete control, and herein is the greatest advantage to be derived from their use. Even the best butter-maker at times will fail to secure uniform results if his starter is not perfectly satisfactory.
3. Keeping quality of product. Butter made from pasteurized cream to which a pure-culture starter has been added will keep much better than the ordinary product, because the diversity of the bacterial flora is less and the milk is therefore not so likely to contain those organisms that produce an "off" condition.
4. Elimination of taints. Many defective conditions in butter are attributable to the growth of undesirable bacteria in the cream that result in the formation of "off" flavors and taints. If cream is pasteurized, thereby destroying these organisms, then ripened with pure ferments, it is generally possible to eliminate the abnormal conditions.[164] Taints may also be present in cream due to direct absorption from the cow or through exposure to foul odors.[165] Troubles of this sort may thus be carried over to the butter. This is particularly true in regions where leeks and wild onions abound, as in some of the Atlantic States. The heating of the cream tends to expel these volatile taints, so that a fairly good article of butter can be made from what would otherwise be a relatively worthless product.
Characteristics desired in culture starters. Certain conditions as the following are desirable in starters made from pure cultures:
1. Vigorous growth in milk at ordinary ripening temperatures.
2. Ability to form acid so as to facilitate churning and increase the yield of butter.
3. Able to produce a clean flavor and desirable aroma.
4. Impart a good keeping quality to butter.
5. Not easily modified in its flavor-producing qualities by artificial cultivation.
These different conditions are difficult to attain, for the reason that some of them seem to be in part incompatible. Weigmann[166] found that a good aroma was generally an evanescent property, and therefore opposed to good keeping quality. Conn has shown that the functions of acid-formation, flavor and aroma production are not necessarily related, and therefore the chances of finding a single organism that possesses all the desirable attributes are not very good.
In all probability no one germ possesses all of these desirable qualities, but natural ripening is the resultant of the action of several forms.[167] This idea has led to the attempt at mixing selected organisms that have been chosen on account of certain favorable characteristics which they might possess. The difficulty of maintaining such a composite culture in its correct proportions when it is propagated in the creamery is seemingly well nigh insuperable, as one organism is very apt to develop more or less rapidly than the other.
A very satisfactory way in which these cultures are marketed is to mix the bacterial growth with some sterile, inert, dry substance. This is the method used in most of the Danish cultures. In this country, some of the more prominent cultures employed are marketed in a liquid form.
Culture vs. home-made starters. One great advantage which has accrued from the use of culture or commercial starters has been that in emphasizing the need of closer control of the ripening process, greater attention has been[Pg 147] paid to the carrying out of the details. In the hands of the better operators, the differences in flavor of butter made with a culture or a natural starter are not marked,[168] but in the hands of those who fail to make a good product under ordinary conditions, an improvement is often secured where a commercial culture is used.
Pasteurization as applied to butter-making. This process, as applied to butter making, is often confounded with the treatment of milk and cream for direct consumption. It is unfortunate that the same term is used in connection with the two methods, for they have but little in common except in the use of heat to destroy the germ life of the milk. In pasteurizing cream for butter-making, it is not necessary to observe the stringent precautions that are to be noted in the preservation of milk; for the addition of a rapidly developing starter controls at once the fermentative changes that subsequently occur. Then again, the physical requirement as to the production of a cooked taste is not so stringent in butter-making. While a cooked taste is imparted to milk or even cream at about 158° F., it is possible to make butter that shows no permanent cooked taste from cream that has been raised as high as 185° or even 195° F. This is due to the fact that the fat does not readily take up those substances that give to scalded milk its peculiar flavor.
Unless care is taken in the manipulation of the heated cream, the grain or body of the butter may be injured. This tendency can be overcome if the ripened cream is[Pg 148] chilled to 48° F. for about two hours before churning. It is also essential that the heated cream should be quickly and thoroughly chilled after being pasteurized.
The Danes, who were the first to employ pasteurization in butter-making, used, in the beginning, a temperature ranging from 158° to 167° F., but owing to the prevalence of such diseases as tuberculosis and foot-and-mouth disease, it became necessary to treat all of the skim milk that was returned from the creameries. For this purpose the skim milk is heated to a temperature of 176° F., it having been more recently determined that this degree of heat is sufficient to destroy the seeds of disease. With the use of this higher temperature the capacity of the pasteurizing apparatus is considerably reduced, but the higher temperature is rendered necessary by the prevailing conditions as to disease.
When the system was first introduced in Denmark, two methods of procedure were followed: the whole milk was heated to a sufficiently high temperature to thoroughly pasteurize it before it was separated, or it was separated first, and the cream pasteurized afterwards. In the latter case, it is necessary to heat the skim milk after separation to destroy the disease organisms, but this can be quickly done by the use of steam directly. Much more care must be used in heating the cream in order to prevent injury to the grain of the butter. In spite of the extra trouble of heating the cream and skim milk separately, this method has practically supplanted the single heating. With the continual spread of tuberculosis in America the heating of skim milk separately is beginning to be introduced.[169]
Use of starters in pasteurized and unpasteurized cream. In order to secure the beneficial results presumably attributable to the use of a starter, natural as well as a pure culture, it should be employed in cream in which the bacteria have first been killed out by pasteurization. This is certainly the most logical and scientific method and is the way in which the process has been developed in Denmark.
Here in this country, the use of pure cultures has been quite rapidly extended, but the system of heating the cream has been used in only a slight measure. The increased labor and expense incurred in pasteurizing the cream has naturally militated somewhat against the wide-spread use of the process, but doubtless the main factor has been the inability to secure as high a flavor where the cream was heated as in the unheated product. As the demands of the market change from a high, quick flavor to one that is somewhat milder but of better keeping quality, doubtless pasteurization of the cream will become more and more popular. That such a change is gradually occurring is already evident, although as yet only a small proportion of butter made in this country is now made in this way. Where the cream is unheated, a considerable number of species will be found, and even the addition of a pure culture, if that culture is of the lactic acid-producing species, will to some extent control the type of fermentation that occurs. Such would not be the case with a culture composed of the casein-digesting type of bacteria. Only those forms could thus be used which are especially well suited to development in raw cream. For this reason the pure culture ferments that are generally employed in creamery practice are organisms of the lactic acid type, able to grow rapidly in cream and produce a pure cream flavor in the butter.[Pg 150]
Purity of commercial starters. Naturally the butter maker is forced to rely on the laboratory for his commercial starter, and the question will often arise as to the purity and vigor of the various ferments employed. As there is no way for the factory operator to ascertain the actual condition of the starter, except by using the same, the greatest care should be taken by the manufacturer to insure the absolute purity of the seed used.
A bacteriological examination of the various cultures which have been placed on the market not infrequently reveals an impure condition. In several cases the writer has found a not inconsiderable number of liquefying bacteria mixed with the selected organism. Molds not infrequently are found in cultures put up in the dry form. Doubtless the effect of these accidental contaminations is considerably less in the case of a starter composed of a distinctively lactic acid-producing organism than with a form which is less capable of thriving vigorously in milk, and it should be said that these impurities can frequently be eliminated by continued propagation.
The virility and vigor of the starter is also a fluctuating factor, dependent in part at least, upon the conditions under which the organism is grown. In some cases the germ is cultivated in solutions in which acid cannot be formed in abundance. Where the conditions permit of the formation of acid, as would be the case if sugar was present with a lactic acid-producing species, the vitality of the culture is often impaired by the action of the gradually accumulating acid. Some manufacturers attempt to minimize this deleterious condition by adding carbonate of lime which unites with the acid that is formed.[Pg 151]
Propagation of starters for cream-ripening. The preparation and propagation of a starter for cream-ripening is a process involving considerable bacteriological knowledge, whether the starter is of domestic origin or prepared from a pure-culture ferment. In any event, it is necessary that the starter should be handled in a way so as to prevent the introduction of foreign bacteria as far as possible. It should be remembered at all times that the starter is a live thing and must be handled throughout its entire history in a way so as to retain its vitality and vigor unimpaired. The following points should be taken into consideration in growing the starter and transferring it from day to day:
1. If a commercial starter is used, see that it is fresh and that the seal has not been broken. If the culture is too old, the larger part of the organisms may have died out before it is transferred, in which case the effect of its addition to the sterilized milk would be of little value.
When the commercial ferment is received, it should be stored in the refrigerator pending its use so as to retard as much as possible the changes that naturally go on in the culture liquid. Be careful that the bottle is not exposed to the influence of direct sunlight for in a transparent medium the organisms may be readily killed by the disinfecting action of the sun's rays.
2. If a home-made starter is employed, use the greatest possible care in selecting the milk that is to be used as a basis for the starter.
3. For the propagation and perpetuation of the starter from day to day, it is necessary that the same should be grown in milk that is as germ-free as it is possible to secure it. For this purpose sterilize some fresh skim-milk in a[Pg 152] covered can that has previously been well steamed. This can be done easily by setting cans containing skim-milk in a vat filled with water and heating the same to 180° F. or above for one-half hour or more. Steam should not be introduced directly. This process destroys all but a few of the most resistant spore-bearing organisms. This will give a cooked flavor to the milk, but will not affect the cream to which the starter is added. Dairy supply houses are now introducing the use of starter cans that are specially made for this purpose.
4. After the heated milk is cooled down to about 70° or 80° F., it can be inoculated with the desired culture. Sometimes it is desirable to "build up" the starter by propagating it first in a smaller volume of milk, and then after this has developed, adding it to a larger amount.
This method is of particular value where a large amount of starter is needed for the cream-ripening.
5. After the milk has been inoculated, it should be kept at a temperature that is suitable for the rapid development of the contained bacteria, 65°-75° F., which temperature should be kept as uniform as possible.
This can best be done by setting the covered can in a vat filled with warm water. The starter cans are often arranged so that temperature can be controlled by circulating water.
6. The starter should not be too thoroughly curdled when it is needed for use, but should be well soured and only partially curdled for it is difficult to break up thoroughly the curd particles if the starter is completely curdled. If[Pg 153] these curd masses are added to ripening cream, white specks may appear in the butter.
7. The vigor of the starter is in all probability stronger when the milk is on the point of curdling than it is after the curd has been formed some time. The continued formation of lactic acid kills many of the bacteria and thus weakens the fermentative action. It is therefore highly important that the acidity of the starter should be closely watched.
8. Do not refrigerate the starter when it has reached the proper stage of development, as this retards the bacterial growth in the same manner as cold weather checks the growth of grain. It is preferable to dilute the starter, if it cannot be used when ready, with sufficient freshly sterilized sweet milk to hold the acidity at the proper point and thus keep the bacteria in the starter in a condition which will favor vigorous growth.
9. The starter should be propagated from day to day by adding a small quantity to a new lot of freshly prepared milk. For this purpose two propagating cans should be provided so that one starter may be in use while the other is being prepared.
How long should a starter be propagated? No hard-and-fast rule can be given for this, for it depends largely upon how carefully the starter is handled during its propagation. If the starter is grown in sterilized milk kept in steamed vessels and is handled with sterile dippers, it is possible to maintain it in a state of relative purity for a considerable period of time; if, however, no especial care is given, it will soon become infected by the air, and the[Pg 154] retention of its purity will depend more upon the ability of the contained organism to choke out foreign growths than upon any other factor. Experience seems to indicate that pure-culture starters "run out" sooner than domestic starters. While it is possible, by bacteriological methods, to determine with accuracy the actual condition of a starter as to its germ content, still such methods are inapplicable in creamery practice. Here the maker must rely largely upon the general appearance of the starter as determined by taste and smell. The supply houses that deal in cultures of this class generally expect to supply a new culture at least every month.
Bacteria in butter. As ripened cream is necessarily rich in bacteria, it follows that butter will also contain germ life in varying amounts, but as butter-fat is not well adapted for bacterial food, the number of germs in butter is usually less than in ripened cream.
Sweet-cream butter is naturally poorer in germ life than that made from ripened cream. Grotenfelt reports in sweet-cream butter, the so-called "Paris butter," only a few bacteria while in acid cream butter the germ content runs from scores to hundreds of thousands.
Effect of bacteria in wash water. An important factor in contamination may be the wash water that is used. Much carelessness often prevails regarding the location and drainage of the creamery well, and if same becomes polluted with organic matter, bacterial growth goes on apace. Melick[170] has made some interesting studies on using pasteurized and sterilized well waters for washing. He found a direct relation to exist between the bacterial content of the wash water and the keeping quality of the butter. Some creameries have tried filtered water but under ordinary conditions a filter, unless it is tended to with great regularity, becomes a source of infection rather than otherwise.
Changes in germ content. The bacteria that are incorporated with the butter as it first "comes" undergo a slight increase for the first few days. The duration of this period of increase is dependent largely upon the condition of the butter. If the buttermilk is well worked out of the butter, the increase is slight and lasts for a few days only, while the presence of so nutritious a medium as buttermilk affords conditions much more favorable for the continued growth of the organisms.
While there may be many varieties in butter when it is fresh, they are very soon reduced in kind as well as number. The lactic acid group of organisms disappear quite rapidly; the spore-bearing species remaining for a somewhat longer time. Butter examined after it is several months old is often found to be almost free from germs.
In the manufacture of butter there is much that is dependent upon the mechanical processes of churning, washing, salting and working the product. These processes do not involve any bacteriological principles other than those that are incident to cleanliness. The cream, if ripened properly, will contain such enormous numbers of favorable forms that the access of the few organisms that are derived from the churn, the air, or the water in washing will have little effect, unless the conditions are abnormal.
Rancid change in butter. Fresh butter has a peculiar aroma that is very desirable and one that enhances the market price, if it can be retained; but this delicate flavor is more or less evanescent, soon disappearing, even in the best makes. While a good butter loses with age some of the peculiar aroma that it possesses when first made, yet a[Pg 156] gilt-edged product should retain its good keeping qualities for some length of time. All butters, however, sooner or later undergo a change that renders them worthless for table use. This change is usually a rancidity that is observed in all stale products of this class. The cause of this rancid condition in butter was at first attributed to the formation of butyric acid, but it is now recognized that other changes also enter in.[171] Light and especially air also exert a marked effect on the flavor of butter. Where butter is kept in small packages it is much more prone to develop off flavors than when packed in large tubs. From the carefully executed experiments of Jensen it appears that some of the molds as well as certain species of bacteria are able to incite these changes. These organisms are common in the air and water and it therefore readily follows that inoculation occurs.
Practically, rancidity is held in check by storing butter at low temperatures where germ growth is quite suspended.
Lack of flavor. Often this may be due to improper handling of the cream in not allowing it to ripen far enough, but sometimes it is impossible to produce a high flavor. The lack of flavor in this case is due to the absence of the proper flavor-producing organisms. This condition can usually be overcome by the addition of a proper starter.
Putrid butter. This specific butter trouble has been observed in Denmark, where it has been studied by Jensen.[172] Butter affected by it rapidly acquires a peculiar putrid odor that ruins it for table use. Sometimes, this flavor may be developed in the cream previous to churning.[Pg 157]
Jensen found the trouble to be due to several different putrefactive bacteria. One form which he called Bacillus f[oe]tidus lactis, a close ally of the common feces bacillus, produced this rotten odor and taste in milk in a very short time. Fortunately, this organism was easily killed by a comparatively low heat, so that pasteurization of the cream and use of a culture starter quickly eliminated the trouble, where it was tried.
Turnip-flavored butter. Butter sometimes acquires a peculiar flavor recalling the order of turnips, rutabagas, and other root crops. Often this trouble is due to feeding, there being in several of these crops, aromatic substances that pass directly into the milk, but in some instances the trouble arises from bacteria that are able to produce decomposition products,[173] the odor and taste of which strongly recalls these vegetables.
"Cowy" butter. Frequently there is to be noted in milk a peculiar odor that resembles that of the cow stable. Usually this defect in milk has been ascribed to the absorption of impure gases by the milk as it cools, although the gases and odors naturally present in fresh milk have this peculiar property that is demonstrable by certain methods of aeration. Occasionally it is transmitted to butter, and recently Pammel[174] has isolated from butter a bacillus that produced in milk the same peculiar odor so commonly present in stables.
Lardy and tallowy butter. The presence of this unpleasant taste in butter may be due to a variety of causes. In some instances, improper food seems to be the source of the trouble; then again, butter exposed to direct sunlight[Pg 158] bleaches in color and develops a lardy flavor.[175] In addition to these, cases have been found in which the defect has been traced to the action of bacteria. Storch[176] has described a lactic-acid form in a sample of tallowy butter that was able to produce this disagreeable odor.
Oily butter. Jensen has isolated one of the causes of the dreaded oily butter that is reported quite frequently in Denmark. The specific organism that he found belongs to the sour-milk bacteria. In twenty-four hours it curdles milk, the curd being solid like that of ordinary sour milk. There is produced, however, in addition to this, an unpleasant odor and taste resembling that of machine oil, a peculiarity that is transmitted directly to butter made from affected cream.
Bitter butter. Now and then butter develops a bitter taste that may be due to a variety of different bacterial forms. In most cases, the bitter flavor in the butter is derived primarily from the bacteria present in the cream or milk. Several of the fermentations of this character in milk are also to be found in butter. In addition to these defects produced by a biological cause, bitter flavors in butter are sometimes produced by the milk being impregnated with volatile, bitter substances derived from weeds.
Moldy butter. This defect is perhaps the most serious because most common. It is produced by the development of a number of different varieties of molds. The trouble appears most frequently in packed butter on the outside of the mass of butter in contact with the tub. Mold spores are so widely disseminated that if proper conditions are[Pg 159] given for their germination, they are almost sure to develop. In some cases the mold is due to the growth of the ordinary bread mold, Penicillium glaucum; in other cases a black mold develops, due often to Cladosporium butyri. Not infrequently trouble of this character is associated with the use of parchment wrappers. The difficulty can easily be held in check by soaking the parchment linings and the tubs in a strong brine, or paraffining the inside of the tub.
Fishy butter. Considerable trouble has been experienced in Australian butter exported to Europe in which a fishy flavor developed. It was noted that the production of this defect seemed to be dependent upon the storage temperature at which the butter was kept. When the butter was refrigerated at 15° F. no further difficulty was experienced. It is claimed that the cause of this condition is due to the formation of trimethylamine (herring brine odor) due to the growth of the mold fungus Oidium lactis, developing in combination with the lactic-acid bacteria.
A fishy taste is sometimes noted in canned butter. Rogers[177] has determined that this flavor is caused by yeasts (Torula) which produce fat-splitting enzyms capable of producing this undesirable change.
[153] Conn and Esten, Cent. f. Bakt., II Abt., 1901, 7:746.
[154] Tiemann, Milch Zeit., 23:701.
[155] Milch Zeit., 1889, p. 7; 1894, p. 624; 1895, p. 383.
[156] Dean, Ont. Agr. Coll., 1897, p. 66.
[157] Storch, Nogle, Unders. over Floed. Syrning, 1890.
[158] Conn, 6 Storrs Expt. Stat., 1893, p. 66.
[159] Conn, 9 Storrs Expt. Stat., 1896, p. 17.
[160] Weigmann, Milch Zeit., 1891, p. 793
[161] McDonnell, ü. Milchsäure Bakterien (Diss. Kiel, 1899), p. 43.
[162] Storch, Milch Zeit., 1890, p. 304.
[163] Conn, 9 Storrs Expt. Stat., 1896, p. 25.
[164] Milch Zeit., 1891, p. 122; 1894, p. 284; 1895, p. 56; 1896, p. 163.
[165] McKay, Bull. 32, Iowa Expt. Stat., p. 47
[166] Weigmann, Landw. Woch. f. Schl. Hol., No. 2, 1890.
[167] Weigmann, Cent. f. Bakt., II Abt., 3:497, 1897.
[168] At the National Creamery Buttermakers' Association for 1901, 193 out of 240 exhibitors used starters. Of those that employed starters, nearly one-half used commercial cultures. There was practically no difference in the average score of the two classes of starters, but those using starters ranked nearly two points higher in flavor than those that did not.
[169] Russell, Bull. 143, Wis. Expt. Stat., Feb. 1907.
[170] Melick, Bull. 138, Kansas Expt. Stat., June 1906.
[171] Reinmann, Cent. f. Bakt., 1900, 6:131; Jensen, Landw. Jahr. d. Schweiz, 1901.
[172] Jensen, Cent. f. Bakt., 1891, 11:409.
[173] Jensen, Milch Zeit., 1892, 6, Nos. 5 and 6.
[174] Pammel, Bull. 21, Iowa Expt. Stat., p. 803.
[175] Fischer, Hyg. Rund., 5:573.
[176] Storch, 18 Rept. Danish Agric. Expt. Stat., 1890.
[177] Rogers Bull. 57, B. A. I. U. S. Dept Agric., 1904.
The art of cheese-making, like all other phases of dairying, has been developed mainly as a result of empirical methods. Within the last decade or so, the subject has received more attention from the scientific point of view and the underlying causes determined to some extent. Since the subject has been investigated from the bacteriological point of view, much light has been thrown on the cause of many changes that were heretofore inexplicable. Our knowledge, as yet, is quite meager, but enough has already been determined to indicate that the whole industry is largely based on the phenomena of ferment action, and that the application of bacteriological principles and ideas is sure to yield more than ordinary results, in explaining, in a rational way, the reasons underlying many of the processes to be observed in this industry.
The problem of good milk is a vital one in any phase of dairy activity, but it is pre-eminently so in cheese-making, for the ability to make a first-class product depends to a large extent on the quality of the raw material. Cheese contains so large a proportion of nitrogenous constituents that it is admirably suited, as a food medium, to the development of bacteria; much better, in fact, than butter.
In the manufacture of cheddar cheese bacteria exert a marked influence in the initial stages of the process. To produce the proper texture that characterizes cheddar cheese, it is necessary to develop a certain amount of acid[Pg 161] which acts upon the casein. This acidity is measured by the development of the lactic-acid bacteria that normally abound in the milk; or, as the cheese-maker expresses it, the milk is "ripened" to the proper point. The action of the rennet, which is added to precipitate the casein of the milk, is markedly affected by the amount of acid present, as well as the temperature. Hence it is desirable to have a standard amount of acidity as well as a standard temperature for coagulation, so as to unify conditions. It frequently happens that the milk is abnormal with reference to its bacterial content, on account of the absence of the proper lactic bacteria, or the presence of forms capable of producing fermentative changes of an undesirable character. In such cases the maker attempts to overcome the effect of the unwelcome bacteria by adding a "starter;" or he must vary his method of manufacture to some extent to meet these new conditions.
Use of starters. A starter may be employed to hasten the ripening of milk that is extremely sweet, so as to curtail the time necessary to get the cheese to press; or it may be used to overcome the effect of abnormal conditions.
The starter that is employed is generally one of domestic origin, and is usually taken from skim milk that has been allowed to ferment and sour under carefully controlled conditions. Of course much depends upon the quality of the starter, and in a natural starter there is always the possibility that it may not be perfectly pure.
Within recent years the attempt has been made to control the effect of the starter more thoroughly by using pure cultures of some desirable lactic-acid form.[178] This has rendered[Pg 162] the making of cheese not only more uniform, but has aided in repressing abnormal fermentations particularly those that are characterized by the production of gas.
Recently, pure cultures of Adametz's B. nobilis, a digesting organism that is claimed to be the cause of the breaking down of the casein and also of the peculiar aroma of Emmenthaler cheese, has been placed on the market under the name Tyrogen. It is claimed that the use of this starter, which is added directly to the milk and also rubbed on the surface of the cheese, results in the improvement of the curds, assists in the development of the proper holes, imparts a favorable aroma and hastens ripening.[179]
Campbell[180] states that the discoloration of cheese in England, which is due to the formation of white spots that are produced by the bleaching of the coloring matter in the cheese, may be overcome by the use of lactic-acid starters.
The use of stringy or slimy whey has been advocated in Holland for some years as a means of overcoming the tendency toward gas formation in Edam cheese which is made from practically sweet milk. This fermentation, the essential feature of which is produced by a culture of Streptococcus Hollandicus,[181] develops acid in a marked degree, thereby inhibiting the production of gas.
The use of masses of moldy bread in directing the fermentation of Roquefort cheese is another illustration of the empirical development of starters, although in this instance it is added after the curds have been prepared for the press.
Pasteurizing milk for cheese-making. If it were possible to use properly pasteurized milk in cheese-making,[Pg 163] then practically all abnormal conditions could be controlled by the use of properly selected starters. Numerous attempts have been made to perfect this system with reference to cheddar cheese, but so far they have been attended with imperfect success. The reason for this is that in pasteurizing milk, the soluble lime salts are precipitated by the action of heat, and under these conditions rennet extract does not curdle the casein in a normal manner. This condition can be restored, in part at least, by the addition of soluble lime salts, such as calcium chlorid; but in our experience, desirable results were not obtained where heated milks to which this calcium solution had been added were made into cheddar cheese. Considerable experience has been gained in the use of heated milks in the manufacture of certain types of foreign cheese. Klein[182] finds that Brick cheese can be successfully made even where the milk is heated as high as 185° F. An increased weight is secured by the addition of the coagulated albumin and also increased moisture.
Bacteria in rennet. In the use of natural rennets, such as are frequently employed in the making of Swiss cheese, considerable numbers of bacteria are added to the milk. Although these rennets are preserved in salt, alcohol or boric acid, they are never free from bacteria. Adametz[183] found ten different species and from 640,000 to 900,000 bacteria per cc. in natural rennets. Freudenreich has shown that rennet extract solutions can be used in Swiss cheese-making quite as well as natural rennets; but to secure the best results, a small quantity of pure lactic ferment must be added to simulate the conditions that prevail when natural rennets are soaked in whey, which, it must be remembered, is a fluid rich in bacterial life.
Where rennet extract or tablets are used, as is generally the case in cheddar making, the number of bacteria added is so infinitesimal as to be negligible.
Development of acid. In the manufacture of cheddar cheese, the development of acid exerts an important influence on the character of the product. This is brought about by holding the curds at temperatures favorable to the growth of the bacteria in the same. Under these conditions the lactic-acid organisms, which usually predominate, develop very rapidly, producing thereby considerable quantities of acid which change materially the texture of the curds. The lactic acid acts upon the casein in solutions containing salt, causing it to dissolve to some extent, thus forming the initial compounds of digestion.[184] This solution of the casein is expressed physically by the "stringing" of the curds on a hot iron. This causes the curds to mat, producing a close, solid body, free from mechanical holes. Still further, the development of this acid is necessary for the digestive activity of the pepsin in the rennet extract.
In some varieties of cheese, as the Swiss, acid is not developed and the character of the cheese is much different from that of cheddar. In all such varieties, a great deal more trouble is experienced from the production of "gassy" curds, because the development of the gas-producing bacteria is held in check by the rapid growth of the lactic acid-producing species.
Bacteria in green cheese. The conditions under which cheese is made permit of the development of bacteria throughout the entire process. The cooking or heating of curds to expel the excessive moisture is never so high as to be fatal to germ life; on the contrary, the acidity of the[Pg 165] curd and whey is continually increased by the development of bacteria in the same.
The body of green cheese fresh from the press is, to a considerable extent, dependent upon the acid produced in the curds. If the curds are put to press in a relatively sweet condition the texture is open and porous. The curd particles do not mat closely together and "mechanical holes," rough and irregular in outline, occur. Very often, at relatively high temperatures, such cheese begin to "huff," soon after being taken from the press, a condition due to the development of gas, produced by gas-generating bacteria acting on the sugar in the curd. This gas finds its way readily into these ragged holes, greatly distending them, as in Fig. 30.
 Fig. 30.
Fig. 30.
L, a sweet curd cheese direct from the press. "Mechanical" holes due to lack of acid development; P, same cheese four days later, mechanical holes distended by development of gas.
Physical changes in ripening cheese. When a green cheese is taken from the press, the curd is tough, firm, but elastic. It has no value as a food product for immediate use,[Pg 166] because it lacks a desirable flavor and is not readily digestible. It is nothing but precipitated casein and fat. In a short time, a deep-seated change occurs. Physically this change is demonstrated in the modification that the curd undergoes. Gradually it breaks down and becomes plastic, the elastic, tough curd being changed into a softened mass. This change in texture of the cheese is also accompanied by a marked change in flavor. The green cheese has no distinctively cheese flavor, but in course of time, with the gradual change of texture, the peculiar flavor incident to ripe cheese is developed.
The characteristic texture and flavor are susceptible of considerable modification that is induced not only by variation in methods of manufacture, but by the conditions under which the cheese are cured. The amount of moisture incorporated with the curd materially affects the physical appearance of the cheese, and the rate of change in the same. The ripening temperature, likewise the moisture content of the surrounding air, also exerts a marked influence on the physical properties of the cheese. To some extent the action of these forces is purely physical, as in the gradual loss by drying, but in other respects they are associated with chemical transformations.
Chemical changes in ripening cheese. Coincident with the physical breaking down of the curd comes a change in the chemical nature of the casein. The hitherto insoluble casein is gradually transformed into soluble nitrogenous substances (caseone of Duclaux, or caseogluten of Weigmann). This chemical phenomenon is a breaking-down process that is analogous to the peptonization of proteids, although in addition to the peptones and albumoses characteristic of peptic digestion, amido-acids and ammonia are[Pg 167] to be found. The quantity of these lower products increases with the age of the cheese.
The chemical reaction of cheese is normally acid to phenolphthalein, although there is generally no free acid, as shown by Congo red, the lactic acid being converted into salts as fast as formed. In very old cheese, undergoing putrefactive changes, especially on the outside, an alkaline reaction may be present, due to the formation of free ammonia.
The changes that occur in a ripening cheese are for the most part confined to the proteids. According to most investigators the fat remains practically unchanged, although the researches of Weigmann and Backe[185] show that fatty acids are formed from the fat. In the green cheese considerable milk-sugar is present, but, as a result of the fermentation that occurs, this is rapidly converted into acid products.
Bacterial flora of cheese. It might naturally be expected that the green cheese, fresh from the press, would contain practically the same kind of bacteria that are in the milk, but a study of cheese shows a peculiar change in the character of the flora. In the first place, fresh cottage cheese, made by the coagulation of the casein through the action of acid, has a more diversified flora than cheese made with rennet, for the reason, as given by Lafar,[186] that the fermentative process is farther advanced.
When different varieties of cheese are made from milk in the same locality, the germ content of even the ripened product has a marked similarity, as is illustrated by Adametz's work[187] on Emmenthaler or Swiss hard cheese,[Pg 168] and Schweitzer Hauskäse, a soft variety. Of the nine species of bacilli and cocci found in mature Emmenthaler, eight of them were also present in ripened Hauskäse.
Different investigators have studied the bacterial flora of various kinds of cheese, but as yet little comparative systematic work has been done. Freudenreich[188] has determined the character and number of bacteria in Emmenthaler cheese, and Russell[189] the same for cheddar cheese. The same general law has also been noted in Canadian[190] and English[191] cheese. At first a marked decrease in numbers is usually noted, lasting for a day or two. This is followed by an enormous increase, caused by the rapid growth of the lactic-acid type. The development may reach scores of millions and often over a hundred million organisms per gram. Synchronous with this increase, the peptonizing and gas-producing bacteria gradually disappear. This rapid development, which lasts only for a few weeks, is followed by a general decline.
In the ripening of cheese a question arises as to whether the process goes on throughout the entire mass of cheese, or whether it is more active at or near the surface. In the case of many of the soft cheese, such as Brie and limburger, bacterial and mold development is exceedingly active on the exterior, and the enzyms secreted by these organisms diffuse toward the interior. That such a condition occurs in the hard type of cheese made with rennet is extremely improbable. Most observers agree that in this type of cheese the ripening progresses throughout the entire mass, although Adametz opposes this view and considers that in[Pg 169] Emmenthaler cheese the development of the specific aroma-producing organism occurs in the superficial layers. Jensen has shown, however, that the greatest amount of soluble nitrogenous products are to be found in the innermost part of the cheese, a condition that is not reconcilable with the view that the most active ripening is on the exterior.[192]
The course of development of bacteria in cheddar cheese is materially influenced by the ripening temperature. In cheese ripened at relatively low temperatures (50°-55° F.),[193] a high germ content is maintained for a much longer period of time than at higher temperatures. Under these conditions the lactic-acid type continues in the ascendancy as usual. In cheese cured at high temperatures (80°-86° F.) the number of organisms is greatly diminished, and they fail to persist in appreciable numbers for as long a time as in cheese cured at temperatures more frequently employed.
Influence of temperature on curing. Temperature exerts a most potent influence on the quality of the cheese, as determined not only by the rate of ripening but the nature of the process itself. Much of the poor quality of cheese is attributable to the effect of improper curing conditions. Probably in the initial stage of this industry cheese were allowed to ripen without any sort of control, with the inevitable result that during the summer months the temperature generally fluctuated so much as to impair seriously the quality. The effect of high temperatures (70° F. and above) is to produce a rapid curing, and, therefore, a short lived cheese; also a sharp, strong flavor, and generally a[Pg 170] more or less open texture. Unless the cheese is made from the best quality of milk, it is very apt to undergo abnormal fermentations, more especially those of a gassy character.
 Fig. 31. Influence of curing temperature on texture of
cheese. Upper row ripened eight months at 60° F.; lower row at 40° F.
Fig. 31. Influence of curing temperature on texture of
cheese. Upper row ripened eight months at 60° F.; lower row at 40° F.
Where cheese is ripened at low temperatures, ranging from 50° F. down to nearly the freezing temperatures, it is found that the quality is greatly improved.[194] Such cheese are thoroughly broken down from a physical point of view even though they may not show such a high per cent of soluble nitrogenous products. They have an excellent texture, generally solid and firm, free from all tendency to openness; and, moreover, their flavor is clean and entirely devoid of the sharp, undesirable tang that so frequently appears in old cheese. The keeping quality of such cheese is much superior to the ordinary product. The introduction[Pg 171] of this new system of cheese-curing promises much from a practical point of view, and undoubtedly a more complete study of the subject from a scientific point of view will aid materially in unraveling some of the problems as to flavor production.
Theories of cheese curing. Within the last few years considerable study has been given the subject of cheese curing or ripening, in order to explain how this physical and chemical transformation is brought about.
Much of the misconception that has arisen relative to the cause of cheese ripening comes from a confusion of terms. In the ordinary use of the word, ripening or curing of cheese is intended to signify the sum total of all the changes that result in converting the green product as it comes from the press into the edible substance that is known as cured cheese. As previously shown, the most marked chemical transformation that occurs is that which has to do with the peptonization or breaking down of the casein. It is true that under ordinary conditions this decomposition process is also accompanied with the formation of certain flavor-producing substances, more or less aromatic in character; but it by no means follows that these two processes are necessarily due to the same cause. The majority of investigators have failed to consider these two questions of casein decomposition and flavor as independent, or at least as not necessarily related. They are undoubtedly closely bound together, but it will be shown later that the problems are quite different and possibly susceptible of more thorough understanding when considered separately.
In the earlier theories of cheese ripening it was thought to be purely a chemical change, but, with the growth of[Pg 172] bacteriological science, evidence was forthcoming that seemed to indicate that the activity of organisms entered into the problem. Schaffer[195] showed that if milk was boiled and made into cheese, the casein failed to break down. Adametz[196] added to green cheese various disinfectants, as creolin and thymol, and found that this practically stopped the curing process. From these experiments he drew the conclusion that bacteria must be the cause of the change, because these organisms were killed; but when it is considered that such treatment would also destroy the activity of enzyms as well as vital ferments, it is evident that these experiments were quite indecisive.
A determination of the nature of the by-products found in maturing cheese indicates that the general character of the ripening change is a peptonization or digestion of the casein.
Until recently the most widely accepted views relating to the cause of this change have been those which ascribed the transformation to the activity of micro-organisms, although concerning the nature of these organisms there has been no unanimity of opinion. The overwhelming development of bacteria in all cheeses naturally gave support to this view; and such experiments as detailed above strengthened the idea that the casein transformation could not occur where these ferment organisms were destroyed.
The very nature of the changes produced in the casein signified that to take part in this process any organism must possess the property of dissolving the proteid molecule, casein, and forming therefrom by-products that are most generally found in other digestive or peptonizing changes of this class.
Digestive bacterial theory. The first theory propounded was that of Duclaux,[197] who in 1887 advanced the idea that this change was due to that type of bacteria which is able to liquefy gelatin, peptonize milk, and cause a hydrolytic change in proteids. To this widely-spread group that he found in cheese, he gave the generic name Tyrothrix (cheese hairs). According to him, these organisms do not function directly as ripening agents, but they secrete an enzym or unorganized ferment to which he applies the name casease. This ferment acts upon the casein of milk, converting it into a soluble product known as caseone. These organisms are found in normal milk, and if they function as casein transformers, one would naturally expect them to be present, at least frequently, if not predominating in the ripening cheese; but such is not the case. In typical cheddar or Swiss cheese, they rapidly disappear (p. 168), although in the moister, softer varieties, they persist for considerable periods of time. According to Freudenreich, even where these organisms are added in large numbers to the curd, they soon perish, an observation that is not regarded as correct by the later adherents to the digestive bacterial theory, as Adametz and Winkler.
Duclaux's experiments were made with liquid media for isolation purposes, and his work, therefore, cannot be regarded as satisfactory as that carried out with more modern technical methods. Recently this theory has been revived by Adametz,[198] who claims to have found in Emmenthaler cheese a digesting species, one of the Tyrothrix type, which is capable of peptonizing the casein and at the same time producing the characteristic flavor of this class of cheese.[Pg 174] This organism, called by him Bacillus nobilis, the Edelpilz of Emmenthaler cheese, has been subjected to comparative experiments, and in the cheese made with pure cultures of this germ better results are claimed to have been secured. Sufficient experiments have not as yet been reported by other investigators to warrant the acceptance of the claims made relative to the effect of this organism.
Lactic-acid bacterial theory. It has already been shown that the lactic-acid bacteria seems to find in the green cheese the optimum conditions of development; that they increase enormously in numbers for a short period, and then finally decline. This marked development, coincident with the breaking down of the casein, has led to the view which has been so ably expounded by Freudenreich[199] that this type of bacterial action is concerned in the ripening of cheese. This group of bacteria is, under ordinary conditions, unable to liquefy gelatin, or digest milk, or, in fact, to exert, under ordinary conditions, any proteolytic or peptonizing properties. This has been the stumbling-block to the acceptance of this hypothesis, as an explanation of the breaking down of the casein. Freudenreich has recently carried on experiments which he believes solve the problem. By growing cultures of these organisms in milk, to which sterile, freshly precipitated chalk had been added, he was able to prolong the development of bacteria for a considerable period of time, and as a result finds that an appreciable part of the casein is digested; but this action is so slow compared with what normally occurs in a cheese, that exception may well be taken to this type of experiment alone. Weigmann[200] inclines to the view that the[Pg 175] lactic-acid bacteria are not the true cause of the peptonizing process, but that their development prepares the soil, as it were, for those forms that are more directly concerned in the peptonizing process. This they do by developing an acid substratum that renders possible the more luxuriant growth of the aroma-producing species. According to Gorini,[201] certain of the Tyrothrix forms function at high temperatures as lactic acid producing bacteria, while at lower temperatures they act as peptonizers. On this basis he seeks to reconcile the discrepancies that appear in the experiments of other investigators.
Digestive milk enzym theory. In 1897 Babcock and the writer[202] showed that milk underwent digestive changes spontaneously when bacterial activity was suspended by the addition of such anaesthetics as ether, chloroform and benzol. The chemical nature of the by-products produced by this auto-digestion of milk resembles quite closely those found in ripened cheese, except that ammonia is not produced as is the case in old cheese. The cause of the decomposition of the casein, they found to be due to the action of a milk enzym which is inherent to the milk itself. This digestive ferment may be separated from fresh milk by concentrating centrifuge slime extracts by the usual physiological reagents. This ferment, called by them galactase, on account of its origin in milk, is a proteolytic enzym of the tryptic type. Its activity is destroyed by strong chemicals such as formaldehyde, corrosive sublimate, also when heated to 175° F. or above. When such extracts are added to boiled milk, the digestive process is started anew, and the by-products produced are very similar to those noted in a normal cheese.
Jensen[203] has also shown that the addition of pancreatic extracts to cheese accelerated the formation of soluble nitrogenous products.
The action of galactase in milk and cheese has been confirmed by Freudenreich[204] and Jensen,[205] as well as by American investigators, and this enzym is now generally accepted as one of the factors concerned in the decomposition of the casein. Freudenreich believes it is able to change casein into albumose and peptones, but that the lactic-acid bacteria are chiefly responsible for the further decomposition of the nitrogen to amid form.
Failure before to recognize the presence of galactase in milk is attributable to the fact that all attempts to secure sterile milk had been made by heating the same, in which case galactase was necessarily destroyed. A brief exposure at 176° F. is sufficient to destroy its activity, and even an exposure at lower temperatures weakens its action considerably, especially if the reaction of the medium is acid. This undoubtedly explains the contradictory results obtained in the ripening of cheese from pasteurized milk, such cheese occasionally breaking down in an abnormal manner.
The results mentioned on page 172, in which cheese failed to ripen when treated with disinfectants,—experiments which were supposed at that time to be the foundation of the bacterial theory of casein digestion—are now explicable on an entirely different basis. In these cases the casein was not peptonized, because these strong disinfectants destroyed the activity of the enzyms as well as the bacteria.
Another important factor in the breaking down of the casein is the pepsin in the rennet extract. The digestive[Pg 177] influence of this agent was first demonstrated for cheddar cheese by Babcock, Russell and Vivian,[206] and simultaneously, although independently, by Jensen[207] in Emmenthaler cheese. In this digestive action, only albumoses and higher peptones are produced. The activity of pepsin does not become manifest until there is about 0.3 per cent. acid which is approximately the amount developed in the cheddar process. These two factors undoubtedly account for by far the larger proportion of the changes in the casein; and yet, the formation of ammonia in well ripened cheese is not accounted for by these factors. This by-product is the main end product of proteid digestion by the liquefying bacteria but their apparent infrequency in cheese makes it difficult to understand how they can function prominently in the change, unless the small quantity of digestive enzyms excreted by them in their growth in milk is capable of continuing its action until a cumulative effect is obtained. Although much light has been thrown on this question by the researches of the last few years, the matter is far from being satisfactorily settled at the present time and the subject needs much more critical work. If liquefying bacteria abound in the milk, doubtless they exert some action, but the rôle of bacteria is doubtless much greater in the production of flavor than in the decomposition of the curd.
Conditions determining quality. In determining the quality of cheese, several factors are to be taken into consideration. First and foremost is the flavor, which determines more than anything else the value of the product. This should be mild and pleasant, although with age the intensity of the same generally increases but at no time should it have any bitter, sour, or otherwise undesirable taste or[Pg 178] aroma. Texture registers more accurately the physical nature of the ripening. The cheese should not be curdy and harsh, but should yield quite readily to pressure under the thumb, becoming on manipulation waxy and plastic instead of crumbly or mealy. Body refers to the openness or closeness of the curd particles, a close, compact mass being most desirable. The color of cheese should be even, not wavy, streaked or bleached.
For a cheese to possess all of these characteristics in an optimum degree is to be perfect in every respect—a condition that is rarely reached.
So many factors influence this condition that the problem of making a perfect cheese becomes exceedingly difficult. Not only must the quality of the milk—the raw material to be used in the manufacture—be perfectly satisfactory, but the factory management while the curds are in the vat demands great skill and careful attention; and finally, the long period of curing in which variation in temperature or moisture conditions may seriously affect the quality,—all of these stages, more or less critical, must be successfully gone through, before the product reaches its highest state of development.
It is of course true that many phases of this complex series of processes have no direct relation to bacteria, yet it frequently happens that the result attained is influenced at some preceding stage by the action of bacteria in one way or another. Thus the influence of the acidity developed in the curds is felt throughout the whole life of the cheese, an over-development of lactic-acid bacteria producing a sour condition that leaves its impress not only on flavor but texture. An insufficient development of acid fails to soften the curd-particles so as to permit of[Pg 179] close matting, the consequence being that the body of the cheese remains loose and open, a condition favorable to the development of gas-generating organisms.
Production of flavor. The importance of flavor as determining the quality of cheese makes it imperative that the nature of the substances that confer on cheese its peculiar aromatic qualities and taste be thoroughly understood. It is to be regretted that the results obtained so far are not more satisfactory, for improvement in technique is hardly to be expected until the reason for the process is thoroughly understood.
The view that is most generally accepted is that this most important phase of cheese curing is dependent upon bacterial activity, but the organisms that are concerned in this process have not as yet been satisfactorily determined. In a number of cases, different species of bacteria have been separated from milk and cheese that have the power of producing aromatic compounds that resemble, in some cases, the peculiar flavors and odors that characterize some of the foreign kinds of cheese; but an introduction of these into curd has not resulted in the production of the peculiar variety, even though the methods of manufacture and curing were closely followed. The similarity in germ content in different varieties of cheese made in the same locality has perhaps a bearing on this question of flavor as related to bacteria. Of the nine different species of bacteria found in Emmenthaler cheese by Adametz, eight of them were also present in ripened Hauskäse. If specific flavors are solely the result of specific bacterial action, it might naturally be expected that the character of the flora would differ.
Some suggestive experiments were made by Babcock and Russell on the question of flavor as related to bacterial[Pg 180] growth, by changing the nature of the environment in cheese by washing the curds on the racks with warm water. In this way the sugar and most of the ash were removed. Under such conditions the character of the bacterial flora was materially modified. While the liquefying type of bacteria was very sparse in normal cheddar, they developed luxuriantly in the washed cheese. The flavor at the same time was markedly affected. The control cheddar was of good quality, while that made from the washed curds was decidedly off, and in the course of ripening became vile. It may be these two results are simply coincidences, but other data[208] bear out the view that the flavor was to some extent related to the nature of the bacteria developing in the cheese. This was strengthened materially by adding different sugars to washed curds, in which case it was found that the flavor was much improved, while the more normal lactic-acid type of bacteria again became predominant.
Ripening of moldy cheese. In a number of foreign cheeses, the peculiar flavor obtained is in part due to the action of various fungi which grow in the cheese, and there produce certain by-products that flavor the cheese. Among the most important of these are the Roquefort cheese of France, Stilton of England, and Gorgonzola of Italy.
Roquefort cheese is made from goat's or cow's milk, and in order to introduce the desired mold, which is the ordinary bread-mold, Penicillium glaucum, carefully-prepared moldy bread-crumbs are added to the curd.
At ordinary temperatures this organism develops too rapidly, so that the cheese to ripen properly must be kept at a low temperature. The town of Roquefort is situated[Pg 181] in a limestone country, in a region full of caves, and it is in these natural caves that most of the ripening is done. These caverns are always very moist and have a temperature ranging from 35° to 44° F., so that the growth of the fungus is retarded considerably. The spread of the mold throughout the ripening mass is also assisted in a mechanical way. The partially-matured cheese are run through a machine that pricks them full of small holes. These slender canals allow the mold organism to penetrate the whole mass more thoroughly, the moldy straw matting upon which the ripening cheese are placed helping to furnish an abundant seeding of the desired germ.
When new factories are constructed it is of advantage to introduce this necessary germ in quantities, and the practice is sometimes followed of rubbing the walls and cellars of the new location with material taken from the old established factory. In this custom, developed in purely an empirical manner, is to be seen a striking illustration of a bacteriological process crudely carried out.
In the Stilton cheese, one of the highly prized moldy cheeses of England, the desired mold fungus is introduced into the green cheese by exchanging plugs taken with a cheese trier from a ripe Stilton.
Ripening of soft cheese. The type of ripening which takes place in the soft cheeses is materially different from that which occurs in the hard type. The peptonizing action does not go on uniformly throughout the cheese, but is hastened by the development of molds and bacteria on the outside that exert a solvent action on the casein. For this reason, soft cheeses are usually made up in small sizes, so that this action may be hastened. The organisms that take part in this process are those that are[Pg 182] able to form enzyms (similar in their action to trypsin, galactase, etc.), and these soluble ferments gradually diffuse from the outside through the cheese.
Most of these peptonizing bacteria are hindered in their growth by the presence of lactic acid, so that in many cases the appearance of the digesting organisms on the surface is delayed until the acidity of the mass is reduced to the proper point by the development of other organisms, principally molds, which prefer an acid substratum for their growth.
In Brie cheese a blue coating of mold develops on the surface. In the course of a few weeks, a white felting appears which later changes to red. This slimy coat below the mold layer is made up of diverse species of bacteria and fungi that are able to grow after the acid is reduced by the blue mold. The organisms in the red slimy coat act upon the casein, producing an alkaline reaction that is unfavorable to the growth of the blue mold. Two sets of organisms are, therefore essential in the ripening process, one preparing the soil for the ferment that later produces the requisite ripening changes. As ordinarily carried on, the process is an empirical one, and if the red coat does not develop as expected, the maker resorts to all kinds of devices to bring out the desired ferment. The appearance of the right form is dependent, however, upon the proper reaction of the cheese, and if this is not suitable, the wished-for growth will not appear.
The reason why cheese is more subject to abnormal fermentation than butter is because its high nitrogen content favors the continued development of bacteria for some time[Pg 183] after it is made. It must be borne in mind, in considering the more important of these changes, that not all defective conditions in cheese are attributable to the influence of living organisms. Troubles frequently arise from errors in manufacturing details, as too prolonged cooking of curds, too high heating, or the development of insufficient or too much acid. Then again, the production of undesirable flavors or impairment in texture may arise from imperfect curing conditions.
Our knowledge regarding the exact nature of these indefinite faults is as yet too inadequate to enable many of these undesirable conditions to be traced to their proper source; but in many cases the taints observed in a factory are due to the abnormal development of certain bacteria, capable of evolving unpleasant or even putrid odors. Most of them are seeded in the milk before it comes to the factory and are due to careless manipulation of the milk while it is still on the farm. Others gain access to the milk in the factory, owing to unclean conditions of one sort or another. Sometimes the cheese-maker is able to overcome these taints by vigorous treatment, but often they pass on into the cheese, only to detract from the market value of the product. Most frequently these "off" flavors appear in cheese that are cured at too high temperatures, say above 65° F.
"Gassy" fermentations in cheese. One of the worst and at the same time most common troubles in cheese-making is where the cheese undergoes a fermentation marked by the evolution of gas. The presence of gas is recognized by the appearance either of spherical or lens-shaped holes of various sizes in the green cheese; often they appear in the curd before it is put to press. Usually in this condition[Pg 184] the curds look as if they had been punctured with a pin, and are known as "pin holey" curds. Where the gas holes are larger, they are known as "Swiss holes" from their resemblance to the normal holes in the Swiss product. If the development of gas is abundant, these holes are restricted in size. Often the formation of gas may be so intense as to cause the curds to float on the surface of the whey before they are removed. Such curds are known as "floaters" or "bloaters."
If "gassy" curds are put to press, the abnormal fermentation may continue. The further production of gas causes the green cheese to "huff" or swell, until it may be considerably distorted as in Fig. 33. In such cases the texture of the cheese is greatly injured, and the flavor is generally impaired.
 Fig. 33. Cheese made from gassy milk.
Fig. 33. Cheese made from gassy milk.
Such abnormal changes may occur at any season of the year, but the trouble is most common in summer, especially in the latter part.
This defect is less likely to occur in cheese that is well[Pg 185] cheddared than in sweet curd cheese. When acidity is produced, these gassy fermentations are checked, and in good cheddar the body is so close and firm as not readily to permit of gaseous changes.
In Swiss cheese, which is essentially a sweet curd cheese, these fermentations are very troublesome. Where large holes are formed in abundance (blähen), the trouble reaches its maximum. If the gas holes are very numerous and therefore small it is called a "nissler." Sometimes the normal "eyes" are even wanting when it is said to be "blind" or a "gläsler."
 Fig. 34. Block Swiss cheese showing "gassy"
fermentation.
Fig. 34. Block Swiss cheese showing "gassy"
fermentation.
One method of procedure which is likely to cause trouble in Swiss factories is often produced by the use of sour, fermented whey in which to soak the natural rennets. Freudenreich and Steinegger[209] have shown that a much more uniform quality of cheese can be made with rennet extract if it is prepared with a starter made from a pure lactic ferment.
The cause of the difficulty has long been charged to various[Pg 186] sources, such as a lack of aeration, improper feeding, retention of animal gases, etc., but in all these cases it was nothing more than a surmise. Very often the milk does not betray any visible symptom of fermentation when received, and the trouble is not to be recognized until the process of cheese-making is well advanced.
Studies from a biological standpoint have, however, thrown much light on this troublesome problem; and it is now known that the formation of gas, either in the curd or after it has been put to press, is due entirely to the breaking down of certain elements, such as the sugar of milk, due to the influence of various living germs. This trouble is, then, a type fermentation, and is, therefore, much more widely distributed than it would be if it was caused by a single specific organism. These gas-producing organisms are to be found, sparingly at least, in almost all milks, but are normally held in check by the ordinary lactic species. Among them are a large number of the bacteria, although yeasts and allied germs are often present and are likewise able to set up fermentative changes of this sort. In these cases the milk-sugar is decomposed in such a way as to give off CO2 and H, and in some cases, alcohol. Russell and Hastings[210] found a lactose-splitting yeast in a severe outbreak of gassy cheese in a Swiss factory. In this case the gas did not develop until the cheese were a few weeks old. In severe cases the cheese actually cracked to pieces.
According to Guillebeau, a close relation exists between those germs that are able to produce an infectious inflammation (mastitis) in the udder of the cow and some forms capable of gas evolution.
If pure cultures of these gas-producing bacteria are added[Pg 187] to perfectly sweet milk, it is possible to artificially produce the conditions in cheese that so frequently appear in practice.
Treatment of "pin-holey" curds. When this type of fermentation appears during the manufacture of the cheese, the maker can control it in part within certain limits. These methods of treatment are, as a rule, purely mechanical, as when the curds are piled and turned, and subsequently ground in a curd mill. After the gas has been forced out, the curds are then put to press and the whole mats into a compact mass.
Another method of treatment based upon bacteriological principles is the addition of a starter to induce the formation of acid. Where acid is developed as a result of the growth of the lactic-acid bacteria, the gas-producing species do not readily thrive. Another reason why acid aids in repressing the development of gas is that the curd particles are partially softened or digested by the action of the acid. This causes them to mat together more closely, and there is not left in the cheese the irregular mechanical openings in which the developing gas may find lodgment.
Another method that is also useful with these curds is to employ salt. This represses gaseous fermentations, and the use of more salt than usual in making the cheese will very often restrain the production of gas. Tendency to form gas in Edam cheese is controlled by the addition of a starter prepared from slimy whey (lange wei) which is caused by the development of an acid-forming organism.
Some have recommended the custom of washing the curds to remove the whey and the gas-producing bacteria contained therein. Care must be taken not to carry this[Pg 188] too far, for the removal of the sugar permits taint-producing organisms to thrive.[211]
The temperature at which the cheese is cured also materially affects the development of gas. At high curing temperatures, gas-producing organisms develop rapidly; therefore more trouble is experienced in summer than at other seasons.
If milks which are prone to undergo "gassy" development are excluded from the general supply, it would be possible to eliminate the source of the entire trouble. To aid in the early recognition of such milks that are not apparently affected when brought to the factory, fermentation or curd tests (p. 76) are of great value. The use of this test in the hands of the factory operator often enables him to detect the exact source of the trouble, which may frequently be confined to the milk delivered by a single patron.
"Fruity" or "sweet" flavor. Not infrequently the product of a factory may acquire during the process of ripening what is known as a "sweet" or "fruity" flavor. This flavor resembles the odor of fermented fruit or the bouquet of certain kinds of wine. It has been noted in widely different sections of the country and its presence bears no relation to the other qualities of the cheese. The cause of this trouble has recently been traced[212] to the presence of various kinds of yeasts. Ordinarily yeasts are rarely present in good cheese, but in cheese affected with this trouble they abound. The addition of starters made from yeast cultures resulted in the production of the undesirable condition.
Mottled cheese. The color of cheese is sometimes cut to that extent that the cheese presents a wavy or mottled appearance. This condition is apt to appear if the ripening temperature is somewhat high, or larger quantities of rennet used than usual. The cause of the defect is obscure, but it has been demonstrated that the same is communicable if a starter is made by grating some of this mottled cheese into milk. The bacteriology of the trouble has not yet been worked out, but the defect is undoubtedly due to an organism that is able to grow in the ripening cheese. It has been claimed that the use of a pure lactic ferment as a starter enables one to overcome this defect.
Bitter cheese. Bitter flavors are sometimes developed in cheese especially where the ripening process is carried on at a low temperature in the presence of an excess of moisture for a considerable length of time.
Guillebeau[213] isolated several forms from Emmenthaler cheese which he connected with udder inflammation that were able to produce a bitter substance in cheese.
Von Freudenreich[214] has described a new form Micrococcus casei amari (micrococcus of bitter cheese) that was found in a sample of bitter cheese. This germ is closely related to Conn's micrococcus of bitter milk. It develops lactic acid rapidly, coagulating the milk and producing an intensely bitter taste in the course of one to three days. When milk infected with this organism is made into cheese, there is formed in a few days a decomposition product that imparts a marked bitter flavor to the cheese.
Harrison[215] has recently found a yeast that grows in the milk and also in the cheese which produces an undesirable bitter change.
It is peculiar that some of the organisms that are able to produce bitter products in milk do not retain this property when the milk is worked up into cheese.
Putrid or rotten cheese. Sometimes cheese undergoes a putrefactive decomposition in which the texture is profoundly modified and various foul smelling gases are evolved. These often begin on the exterior as small circumscribed spots that slowly extend into the cheese, changing the casein into a soft slimy mass. Then, again, the interior of the cheese undergoes this slimy decomposition. The soft varieties are more prone toward this fermentation than the hard, although the firm cheeses are by no means exempt from the trouble. The "Verlaufen" or "running" of limburger cheese is a fermentation allied to this. It is where the inside of the cheese breaks down into a soft semi-fluid mass. In severe cases, the rind may even be ruptured, in which case the whole interior of the cheese flows out as a thick slimy mass, having sometimes a putrid odor. The conditions favoring this putrid decomposition are usually associated with an excess of moisture, and an abnormally low ripening temperature.
Rusty spot. This name is applied to the development of small yellowish-red or orange spots that are formed sometimes throughout the whole mass of cheddar cheese. A close inspection shows the colored points to be located along the edges of the curd particles. According to Harding,[216] this trouble is most common in spring and fall. The cause of the difficulty has been traced by Connell[217] to the development of a chromogenic bacterium, Bacillus rudensis. The organism can be most readily isolated on a potato[Pg 191] surface rather than with the usual isolating media, agar or gelatin.
Other pigment changes. Occasionally, with the hard type of cheese, but more frequently with the softer foreign varieties, various abnormal conditions arise that are marked by the production of different pigments in or on the cheese. More frequently these are merely superficial and affect only the outer layers of the cheese. Generally they are attributable to the development of certain chromogenic organisms (bacteria, molds and yeasts), although occasionally due to other causes, as in the case of a blue discoloration sometimes noted in foreign cheese made in copper kettles.[218]
De Vries[219] has described a blue condition that is found in Edam cheese. It appears first as a small blue spot on the inside, increasing rapidly in size until the whole mass is affected. This defect he was able to show was produced by a pigment-forming organism, B. cyaneo-fuscus. By the use of slimy whey (lange wei) this abnormal change was controlled.
Moldy cheese. With many varieties of cheese, especially some of the foreign types, the presence of mold on the exterior is not regarded as detrimental; in fact a limited development is much desired. In hard rennet cheese as cheddar or Swiss, the market demands a product free from mold, although it should be said that this condition is imposed by the desire to secure a good-looking cheese rather than any injury in flavor that the mold causes. Mold spores are so widely distributed that, if proper temperature and moisture conditions prevail, these spores will always develop. At temperatures in the neighborhood of 40° F.[Pg 192] and below, mold growth is exceedingly slow, and often fructification does not occur, the only evidence of the mold being the white, felt-like covering that is made up of the vegetating filaments. The use of paraffin has been suggested as a means of overcoming this growth, the cheese being dipped at an early stage into melted paraffin. Recent experiments have shown that "off" flavors sometimes develop where cheese are paraffined directly from the press. If paraffin is too hard, it has a tendency to crack and separate from the rind, thus allowing molds to develop beneath the paraffin coat, where the conditions are ideal as to moisture, for evaporation is excluded and the air consequently saturated. The use of formalin (2% solution) has been suggested as a wash for the outside of the cheese. This substance or sulfur is also applied in a gaseous form. Double bandaging is also resorted to as a means of making the cheese more presentable through the removal of the outer bandage.
The nature of these molds has not been thoroughly studied as yet. The ordinary blue-green bread mold, Penicillium glaucum, is most frequently found, but there are numerous other forms that appear, especially at low temperatures.
Poisonous cheese. Cases of acute poisoning arising from the ingestion of cheese are reported from time to time. Vaughan has succeeded in showing that this condition is due to the formation of a highly poisonous alkaloid which he has isolated, and which he calls tyrotoxicon.[220] This poisonous ptomaine has also been demonstrated in milk and other milk products, and is undoubtedly due to the development of various putrefactive bacteria that find their way into the milk. It seems quite probable that the development[Pg 193] of these toxic organisms can also go on in the cheese after it is taken from the press.
Prevention or cheese defects. The defective conditions previously referred to can rarely be overcome in cheese so as to improve the affected product, for they only become manifest in most cases during the later stages of the curing process. The only remedy against future loss is to recognize the conditions that are apt to prevail during the occurrence of an outbreak and see that the cheese are handled in such a way as to prevent a recurrence of the difficulty.
Many abnormal and undesirable results are incident to the manufacture of the product, such as "sour" or "mealy" cheese, conditions due to the development of too much acid in the milk or too high a "cook." These are under the direct control of the maker and for them he alone is responsible. The development of taints due to the growth of unwelcome bacteria that have gained access to the milk while it is yet on the farm are generally beyond the control of the cheese maker, unless they are so pronounced as to appear during the handling of the curds. If this does occur he is sometimes able, through the intervention of a starter or by varying some detail in making, to handle the milk in such a way as to minimize the trouble, but rarely is he able to eliminate it entirely.
One of the most strenuous duties which the maker must perform at all times is to point out to his patrons the absolute necessity of their handling the milk in such a way as to prevent the introduction of organisms of a baleful type.
[178] Russell, 13 Rept. Wis. Expt. Stat., 1896, p. 112; Campbell, Trans. High. & Agr. Soc. Scotland, 5 ser., 1898, 10:181.
[179] Winkler, Milch Zeit. (Hildesheim), Nov. 24, 1900.
[180] Campbell, No. Brit., Agric., May 12, 1897.
[181] Weigmann, Milch Zeit., No. 50, 1889.
[182] Klein, Milch Zeit. (Hildesheim), No. 17, 1900.
[183] Adametz, Landw. Jahr., 18:256.
[184] Van Slyke and Hart, Bull. 214, N. Y. Expt. Stat., July 1902.
[185] Milch Zeit., 1898, No. 49.
[186] Lafar, Technical Mycology, p. 216.
[187] Adametz, Landw. Jahr., 18:228.
[188] Freudenreich, Landw. Jahr. d. Schweiz, 4:17; 5:16.
[189] Russell, 13 Rept. Wis. Expt. Stat., 1896, p. 95.
[190] Harrison and Connell, Rev. gen. du Lait, Nos. 4, 5, 6, 7 and 8, 1903-04.
[191] Lloyd, Bath and West of Eng. Soc. Rept., 1892, 2:180.
[192] Freudenreich, Landw. Jahr. d. Schweiz, 1900; Adametz, Oest. Molk. Zeit., 1899, No. 7.
[193] Russell, 14 Wis. Expt. Stat., 1897, p. 203. Harrison and Connell, Rev. gen. du Lait Nos. 4, etc., 1903-04.
[194] Babcock and Russell, 18 Rept. Wis. Expt. Stat., 1901. Dean, Harrison and Harcourt, Bull. 121, Ont. Agr'l. Coll., June 1902.
[195] Schaffer, Milch Zeit., 1889, p. 146.
[196] Adametz, Landw. Jahr., 18:261.
[197] Duclaux, Le Lait, p. 213.
[198] Adametz, Oest. Molk. Zeit., 1900, Nos. 16-18.
[199] Freudenreich, Landw. Jahr. d. Schweiz, 1897, p. 85.
[200] Weigmann, Cent. f. Bakt., II Abt., 1898, 4:593; also 1899, 5:630.
[201] Gorini, Abs. in Expt. Stat. Rec., 11:388.
[202] Babcock and Russell, 14 Rept. Wis. Expt. Stat., 1897, p. 161.
[203] Jensen, Cent. f. Bakt., II Abt., 3:752.
[204] Freudenreich, Cent. f. Bakt., II Abt., 1900, 6:332.
[205] Jensen, Ibid., 1900, 6:734.
[206] 17 Rept. Wis. Expt. Stat., 1900, p. 102.
[207] Jensen, Landw. Jahr. d. Schweiz, 1900.
[208] Babcock and Russell, 18 Rept. Wis. Expt. Stat., 1901.
[209] Cent. f. Bakt. 1899, p. 14.
[210] Bull. 128, Wis. Expt. Stat., Sept. 1905.
[211] Babcock and Russell, 18 Rept. Wis. Expt. Stat., 1901.
[212] Harding, Rogers and Smith, Bull. 183, N. Y. (Geneva) Expt. Stat., Dec., 1900.
[213] Guillebeau, Landw. Jahr., 1890, p. 27.
[214] Freudenreich, Füehl. Landw. Ztg., 43:361.
[215] Harrison, Bull. 123 Ont. Agr'l. Coll., May, 1902.
[216] Bull. 183, N. Y. (Geneva) Expt. Stat., Dec. 1900.
[217] Connell, Bull. Canadian Dept. of Agr., 1897.
[218] Schmöger, Milch Zeit., 1883, p. 483.
[219] De Vries, Milch Zeit., 1888, pp. 861, 885.
[220] Zeit. f. physiol. Chemie, 10:146.
Acid, effect of, on churning, 137;
in butter-making, 138.
Acid test, 52.
Aeration of milk, 59.
Aerobic bacteria, 7.
Alcoholic fermentation in milk, 72.
Anaerobic bacteria, 7.
Animal, influence of, on milk infection, 34.
Animal odor, 56.
Anthrax, 94.
Antiseptics, 9, 88.
Aroma, of butter, 140.
Bacillus: definition of, 2.
acidi lactici, 64;
cyaneo-fuscus, 188;
cyanogenus, 74;
foetidus lactis, 157;
lactis aerogenes, 65;
lactis erythrogenes, 74;
lactis saponacei, 67;
lactis viscosus, 71;
nobilis, 162, 174;
prodigiosus, 74;
rudensis, 188;
synxanthus, 75;
tuberculosis, 84.
Bacteria:
on hairs, 35;
kinds in milk, 63;
in barn air, 42;
in milk pails, 27;
in butter, 154;
classification of, 4;
in cheese, 160;
culture of, 17;
in cream, 128;
discovery of, 1;
external conditions affecting, 8;
form of, 2;
in butter, 142;
in butter-making, 127;
in centrifuge slime, 39;
In fore milk, 28;
in rennet, 163;
In separator slime, 39;
manure, 37;
number of, in milk, 50.
Distribution of:
milk of American cities, 50;
European cities, 50;
in relation to cheese, 168.
Of disease:
anthrax, 94;
cholera, 98;
diphtheria, 99;
lockjaw, 94;
toxic, 100;
tuberculosis, 84;
typhoid fever, 98.
Methods of study of:
culture, 15;
culture media, 13;
isolation, 14.
Bitter butter, 158;
cheese, 189;
milk, 72.
Bloody milk, 74.
Blue cheese, 191;
milk, 74.
Bovine tuberculosis, 84.
Brie cheese, 182.
Butter:
bacteria in, 154;
bitter, 158;
"cowy,"157;
fishy, 159;
lardy, 157;
moldy, 158;
mottled, 156;
oily, 158;
putrid, 156;
rancid, 155;
tallowy, 157;
turnip flavor in, 157.
Making:
aroma, 140;
flavor in, 140;
pure culture, 143;
in ripening of cream, 136.
Butyric acid fermentation, 69.
By-products of factory, methods of preserving, 25.
Casease, 68.
Caseone, 68.
Centrifugal force, cleaning milk by, 38.
Cheese:
bacterial flora of, 168;
bitter, 189;
blue, 187;
Brie, 182;
Edam, 72, 162;
Emmenthaler, 185;
flavor of, 179;
gassy fermentations in, 183;
Gorgonzola, 180;
molds on, 191;
mottled, 189;
"nissler,"185;
poisonous, 192;
putrid, 190;
ripening of moldy, 180;
ripening of soft, 181;
Roquefort, 180;
rusty spot in, 188;
Stilton, 180;
Swiss, 185.
Making and curing:
chemical changes in curing, 166;
influence of temperature on curing, 169;
[Pg 196]influence of rennet, 177;
physical changes in curing, 165;
prevention of defects, 193;
starters in, 161;
temperature in relation to bacterial influence, 169.
Theories of curing:
digestive, 173;
galactase, 175, 177;
lactic acid, 174.
Chemical changes in cheese-ripening, 166.
Chemical disinfectants in milk:
bleaching powder, 81;
corrosive sublimate, 81;
formalin, 80;
sulfur, 80;
whitewash, 81;
vitriol, 81.
Chemical preservatives, 80.
Children, milk for, 45.
Cholera in milk, 98.
Classification by separator, 38.
Coccus, definition of, 2.
Cold, influence on bacteria, 8, 48.
Contamination of milk through disease germs, 95, 191.
Covered milk pails, 41.
Cream, bacterial changes in, 135;
mechanical causes for bacteria in, 135;
pasteurized, 113;
restoration of consistency of pasteurized, 132.
Ripening of, 136;
advantage of pure cultures in, 144;
by natural starters, 142;
characteristics of pure cultures in, 145;
objections to pure cultures in, 146;
principles of pure cultures in, 143;
propagation of pure cultures, 151;
purity of commercial starters, 150;
home-made starters in, 146.
Creaming methods, 134.
Curd test, 76.
Dairy utensils a source of contamination, 21.
Diarrhoeal diseases, 100.
Digesting bacteria, 67.
Digestibility of heated milk, 111.
Diphtheria, 99.
Dirt in milk, 34.
Dirt, exclusion of, 36.
Disease germs in milk, 95;
effect of heat on, 91;
origin of, 83.
Disinfectants, 9:
carbolic acid, 81;
chloride of lime, 81;
corrosive sublimate, 81;
formalin, 80;
sulfur, 80;
vitriol salts, 81;
whitewash, 79.
Disinfectants in milk:
alkaline salts, 106;
boracic acid, 106;
formalin, 106;
preservaline, 107;
salicylic acid, 106.
Domestic pasteurizing apparatus, 119.
Drugs, taints in milk due to, 56.
Drying, effect of, 8.
Edam cheese, 72, 162.
Emmenthaler cheese, 185.
Endospores, 3.
Enzyms, 10.
Factory by-products, 22;
treatment of, 25.
Farrington alkaline tablet, 52.
Fecal bacteria, effect of, on butter, 35.
Fermentation:
In cheese: gassy, 183.
In milk:
alcoholic, 72;
bitter, 72;
blue, 74;
butyric, 69;
digesting, 67;
gassy, 66;
kephir, 72;
koumiss, 72;
lactic acid, 63;
lange-wei, 72;
red, 74;
ropy, 69;
slimy, 69;
soapy, 73;
souring, 63;
sweet curdling, 67;
treatment of, 75.
Tests, 76;
Gerber's, 76;
Walther's, 76;
Wisconsin curd, 76.
Filtration of milk, 38.
Fishy butter, 159.
Flavor:
of butter, 140;
of cheese, 179.
Foot and mouth disease, 93.
Fore milk, 28.
Formaldehyde, 80.
Formalin, 80.
[Pg 197]Fruity flavor in cheese, 188.
Galactase in cheese, 175.
Gassy fermentations:
in cheese, 183;
in milk, 67;
in Swiss cheese, 167.
Gläsler, 185.
Gorgonzola cheese, 180.
Growth of bacteria, essential conditions for, 4;
in milk, 46.
Hair, bacteria on, 35.
Heat, influence on bacterial growth, 8.
Heated milk:
characteristics of, 109;
action toward rennet, 112;
body, 110;
digestibility, 111;
fermentative changes, 111;
flavor, 110;
hydrogen peroxid test in, 23;
Storch's test, 23.
Hygienic milk, bacteria in, 45.
Infection of milk:
animal, 34;
dairy utensils, 21;
fore milk, 28;
milker, 36.
Isolation of bacteria, methods of, 14.
Kephir, 72.
Koumiss, 72.
Lactic acid:
fermentation in milk, 63;
theory in cheese-curing, 174.
Lange-wei, 72.
Lardy butter, 157.
Light, action on bacteria, 9.
Manure, bacteria in, 33.
Methods:
of isolation, 14;
culture, 15.
Micrococcus casei amari, 189.
Microscope, use of, 17.
Milk:
a bacterial food medium, 19;
bacteria in, 48.
Disease organisms in:
anthrax, 94;
cholera, 98;
diphtheria, 99;
foot and mouth disease, 93;
poisonous, 101;
ptomaines, 101;
scarlet fever, 99;
tuberculosis, 84;
typhoid fever, 98.
Contamination, 20:
from air, 42;
from animal odors, 55;
dirt, 34;
distinction between bacterial and non-bacterial, 57;
fore milk, 28;
infection in factory, 59;
milker, 36;
relative importance of various kinds, 43;
utensils, 21.
Milk fermentations:
alcoholic, 72;
bitter, 72;
bloody, 74;
blue, 74;
butyric acid, 69;
gassy, 66, 167;
kephir, 72;
koumiss, 72;
lactic acid, 63;
red, 72;
ropy, 69;
slimy, 69;
soapy, 74;
souring, 63;
sweet curdling, 67;
tests for, 76;
treatment of, 75;
yellow, 75.
Milk, heated:
action towards rennet, 112;
digestibility, 111;
flavor of, 110;
fermentative changes in, 111;
hydrogen peroxid test, 110.
Milking machines, influence of, on germ content, 37.
Milk preservation:
chemical agents in, 106;
condensation, 107;
freezing, 108;
heat, 108;
pasteurization, 113;
sterilization, 112.
Milk-sugar as bacterial food, 19.
Mold, in butter, 158;
in cheese, 191.
Mottled cheese, 189.
"Nissler" cheese, 185.
Odors, direct absorption of, in milk, 55.
Oidium lactis, 159.
Oily butter, 158.
Pasteurization of milk;
acid test in, 128;
bacteriological study of, 124, 126, 149;
for butter, 147;
for cheese, 162;
for direct use, 113;
of skim milk, 25;
details of, 128;
temperature and time limit in, 118.
Pasteurizing apparatus:
continuous flow, 122;
coolers, 131;
Danish, 123;
domestic, 119;
Farrington, 122;
intermittant flow, 121;
Miller, 122;
Potts, 121;
regenerator, 122;
Reid, 126;
Russell, 121;
[Pg 198]testing rate of flow, 124.
Penicillium glaucum, 159, 180, 190.
Pepsin, 10.
Physical changes in cheese-ripening, 165.
Poisonous bacteria:
in cheese, 192;
in milk, 100, 101.
Preservaline, 167.
Preservation of milk:
by exclusion, 103;
chemical agents, 106;
condensing, 107;
filtration, 38;
freezing, 108;
pasteurization, 112;
physical agents, 107;
sterilization, 112.
Ptomaine poisoning, 101.
Pure cultures, 15.
Pure culture starters:
advantages of, 144;
characteristics of, 145;
home-made cultures compared with, 146;
propagation of, 151.
Putrid cheese, 190;
butter, 156.
Rancidity in butter, 155.
Red milk, 74.
Rennet:
action in heated milk, 112;
bacteria in, 163;
influence of, on cheese-ripening, 177.
Restoration of consistency in pasteurized cream, 132.
Ripening of cheese:
moldy cheese, 180;
soft cheese, 181.
Of cream, 136;
artificial starters, 143;
natural starters, 142;
principles of pure culture starters in, 143.
Ropy milk, 69.
Roquefort cheese, 180.
Rusty spot in cheese, 190.
Rusty cans: effect of, on acidity, 53.
Sanitary milk, 45, 104.
Sanitary pails, 41.
Scarlet fever in milk, 99.
Separator slime:
bacteria in, 39;
tubercle bacillus in, 93.
Scalded layer, resistance of bacteria in, 91.
Skim-milk, a distributor of disease, 96.
Slimy milk, 69.
Soapy milk, 74.
Soft cheese, ripening of, 186.
Sources of contamination in milk:
barn air, 42;
dairy utensils, 21;
dirt from animals, 34;
factory cans, 25;
fore-milk, 28;
milker, 36.
Souring of milk, 63.
Spirillum, definition of, 2.
Spores, 3.
Starters:
in cheese-making, 161;
in butter-making, 142;
propagation of, 151;
pure cultures in cream-ripening, 143.
Sterilization of milk, 112.
Streptococcus Hollandicus, 72, 162.
Stilton cheese, 181.
Storch's test, 23.
Sulfur as a disinfectant, 81.
Sweet curdling milk, 68.
Sweet flavor in cheese, 188.
Swiss cheese, 177;
gassy fermentations in, 24, 185.
Taints, absorption of, 55.
Taints, bacterial vs. physical, 58.
Taints in milk, absorption of, 55.
Taints, use of starters in overcoming, 79.
Taints in butter:
putrid, 156;
rancidity, 155;
turnip flavor, 157.
Tallowy butter, 157.
Temperature:
effect on bacterial development, 6, 48;
effect of low, 108;
effect of high, 108;
and time limit in milk pasteurization, 113.
Tests for milk:
fermentation, 76;
Storch's, 23;
acid, 52.
Theories in cheese-curing:
digestive, 171;
galactase, 175, 177;
lactic acid, 174.
[Pg 199]Trypsin, 10.
Tubercle bacillus:
in milk, 88;
in separator slime, 93;
thermal death limits, 117.
Tuberculin test, 86.
Tuberculosis, bovine, 84.
Turnip flavor in butter, 157.
Typhoid fever, 98.
Tyrogen, 162.
Tyrotoxicon, 101, 190.
Udder:
artificial introduction of bacteria into, 32;
milk germ-free in, 19;
infection of, 28;
washing, 89;
tuberculosis in, 87.
Viscogen, 132.
Water: as a source of infection, 61.
Whey, pollution of vats, 23;
method of preserving, 25;
treatment of, in vats, 25.
Whitewash, 81.
Wisconsin curd test, 76.
Yeasts:
alcoholic ferments in milk, 73;
fruity flavor in cheese, 186;
gassy due to yeasts, 186;
in bitter cheese, 189;
in canned butter, 159;
kephir, 72.
End of the Project Gutenberg EBook of Outlines of Dairy Bacteriology, 8th
edition, by H. L. Russell
*** END OF THIS PROJECT GUTENBERG EBOOK DAIRY BACTERIOLOGY, 8TH EDITION ***
***** This file should be named 27778-h.htm or 27778-h.zip *****
This and all associated files of various formats will be found in:
https://www.gutenberg.org/2/7/7/7/27778/
Produced by Mark C. Orton, Linda McKeown, Josephine Paolucci
and the Online Distributed Proofreading Team at
https://www.pgdp.net.
Updated editions will replace the previous one--the old editions
will be renamed.
Creating the works from public domain print editions means that no
one owns a United States copyright in these works, so the Foundation
(and you!) can copy and distribute it in the United States without
permission and without paying copyright royalties. Special rules,
set forth in the General Terms of Use part of this license, apply to
copying and distributing Project Gutenberg-tm electronic works to
protect the PROJECT GUTENBERG-tm concept and trademark. Project
Gutenberg is a registered trademark, and may not be used if you
charge for the eBooks, unless you receive specific permission. If you
do not charge anything for copies of this eBook, complying with the
rules is very easy. You may use this eBook for nearly any purpose
such as creation of derivative works, reports, performances and
research. They may be modified and printed and given away--you may do
practically ANYTHING with public domain eBooks. Redistribution is
subject to the trademark license, especially commercial
redistribution.
*** START: FULL LICENSE ***
THE FULL PROJECT GUTENBERG LICENSE
PLEASE READ THIS BEFORE YOU DISTRIBUTE OR USE THIS WORK
To protect the Project Gutenberg-tm mission of promoting the free
distribution of electronic works, by using or distributing this work
(or any other work associated in any way with the phrase "Project
Gutenberg"), you agree to comply with all the terms of the Full Project
Gutenberg-tm License (available with this file or online at
https://gutenberg.org/license).
Section 1. General Terms of Use and Redistributing Project Gutenberg-tm
electronic works
1.A. By reading or using any part of this Project Gutenberg-tm
electronic work, you indicate that you have read, understand, agree to
and accept all the terms of this license and intellectual property
(trademark/copyright) agreement. If you do not agree to abide by all
the terms of this agreement, you must cease using and return or destroy
all copies of Project Gutenberg-tm electronic works in your possession.
If you paid a fee for obtaining a copy of or access to a Project
Gutenberg-tm electronic work and you do not agree to be bound by the
terms of this agreement, you may obtain a refund from the person or
entity to whom you paid the fee as set forth in paragraph 1.E.8.
1.B. "Project Gutenberg" is a registered trademark. It may only be
used on or associated in any way with an electronic work by people who
agree to be bound by the terms of this agreement. There are a few
things that you can do with most Project Gutenberg-tm electronic works
even without complying with the full terms of this agreement. See
paragraph 1.C below. There are a lot of things you can do with Project
Gutenberg-tm electronic works if you follow the terms of this agreement
and help preserve free future access to Project Gutenberg-tm electronic
works. See paragraph 1.E below.
1.C. The Project Gutenberg Literary Archive Foundation ("the Foundation"
or PGLAF), owns a compilation copyright in the collection of Project
Gutenberg-tm electronic works. Nearly all the individual works in the
collection are in the public domain in the United States. If an
individual work is in the public domain in the United States and you are
located in the United States, we do not claim a right to prevent you from
copying, distributing, performing, displaying or creating derivative
works based on the work as long as all references to Project Gutenberg
are removed. Of course, we hope that you will support the Project
Gutenberg-tm mission of promoting free access to electronic works by
freely sharing Project Gutenberg-tm works in compliance with the terms of
this agreement for keeping the Project Gutenberg-tm name associated with
the work. You can easily comply with the terms of this agreement by
keeping this work in the same format with its attached full Project
Gutenberg-tm License when you share it without charge with others.
1.D. The copyright laws of the place where you are located also govern
what you can do with this work. Copyright laws in most countries are in
a constant state of change. If you are outside the United States, check
the laws of your country in addition to the terms of this agreement
before downloading, copying, displaying, performing, distributing or
creating derivative works based on this work or any other Project
Gutenberg-tm work. The Foundation makes no representations concerning
the copyright status of any work in any country outside the United
States.
1.E. Unless you have removed all references to Project Gutenberg:
1.E.1. The following sentence, with active links to, or other immediate
access to, the full Project Gutenberg-tm License must appear prominently
whenever any copy of a Project Gutenberg-tm work (any work on which the
phrase "Project Gutenberg" appears, or with which the phrase "Project
Gutenberg" is associated) is accessed, displayed, performed, viewed,
copied or distributed:
This eBook is for the use of anyone anywhere at no cost and with
almost no restrictions whatsoever. You may copy it, give it away or
re-use it under the terms of the Project Gutenberg License included
with this eBook or online at www.gutenberg.org
1.E.2. If an individual Project Gutenberg-tm electronic work is derived
from the public domain (does not contain a notice indicating that it is
posted with permission of the copyright holder), the work can be copied
and distributed to anyone in the United States without paying any fees
or charges. If you are redistributing or providing access to a work
with the phrase "Project Gutenberg" associated with or appearing on the
work, you must comply either with the requirements of paragraphs 1.E.1
through 1.E.7 or obtain permission for the use of the work and the
Project Gutenberg-tm trademark as set forth in paragraphs 1.E.8 or
1.E.9.
1.E.3. If an individual Project Gutenberg-tm electronic work is posted
with the permission of the copyright holder, your use and distribution
must comply with both paragraphs 1.E.1 through 1.E.7 and any additional
terms imposed by the copyright holder. Additional terms will be linked
to the Project Gutenberg-tm License for all works posted with the
permission of the copyright holder found at the beginning of this work.
1.E.4. Do not unlink or detach or remove the full Project Gutenberg-tm
License terms from this work, or any files containing a part of this
work or any other work associated with Project Gutenberg-tm.
1.E.5. Do not copy, display, perform, distribute or redistribute this
electronic work, or any part of this electronic work, without
prominently displaying the sentence set forth in paragraph 1.E.1 with
active links or immediate access to the full terms of the Project
Gutenberg-tm License.
1.E.6. You may convert to and distribute this work in any binary,
compressed, marked up, nonproprietary or proprietary form, including any
word processing or hypertext form. However, if you provide access to or
distribute copies of a Project Gutenberg-tm work in a format other than
"Plain Vanilla ASCII" or other format used in the official version
posted on the official Project Gutenberg-tm web site (www.gutenberg.org),
you must, at no additional cost, fee or expense to the user, provide a
copy, a means of exporting a copy, or a means of obtaining a copy upon
request, of the work in its original "Plain Vanilla ASCII" or other
form. Any alternate format must include the full Project Gutenberg-tm
License as specified in paragraph 1.E.1.
1.E.7. Do not charge a fee for access to, viewing, displaying,
performing, copying or distributing any Project Gutenberg-tm works
unless you comply with paragraph 1.E.8 or 1.E.9.
1.E.8. You may charge a reasonable fee for copies of or providing
access to or distributing Project Gutenberg-tm electronic works provided
that
- You pay a royalty fee of 20% of the gross profits you derive from
the use of Project Gutenberg-tm works calculated using the method
you already use to calculate your applicable taxes. The fee is
owed to the owner of the Project Gutenberg-tm trademark, but he
has agreed to donate royalties under this paragraph to the
Project Gutenberg Literary Archive Foundation. Royalty payments
must be paid within 60 days following each date on which you
prepare (or are legally required to prepare) your periodic tax
returns. Royalty payments should be clearly marked as such and
sent to the Project Gutenberg Literary Archive Foundation at the
address specified in Section 4, "Information about donations to
the Project Gutenberg Literary Archive Foundation."
- You provide a full refund of any money paid by a user who notifies
you in writing (or by e-mail) within 30 days of receipt that s/he
does not agree to the terms of the full Project Gutenberg-tm
License. You must require such a user to return or
destroy all copies of the works possessed in a physical medium
and discontinue all use of and all access to other copies of
Project Gutenberg-tm works.
- You provide, in accordance with paragraph 1.F.3, a full refund of any
money paid for a work or a replacement copy, if a defect in the
electronic work is discovered and reported to you within 90 days
of receipt of the work.
- You comply with all other terms of this agreement for free
distribution of Project Gutenberg-tm works.
1.E.9. If you wish to charge a fee or distribute a Project Gutenberg-tm
electronic work or group of works on different terms than are set
forth in this agreement, you must obtain permission in writing from
both the Project Gutenberg Literary Archive Foundation and Michael
Hart, the owner of the Project Gutenberg-tm trademark. Contact the
Foundation as set forth in Section 3 below.
1.F.
1.F.1. Project Gutenberg volunteers and employees expend considerable
effort to identify, do copyright research on, transcribe and proofread
public domain works in creating the Project Gutenberg-tm
collection. Despite these efforts, Project Gutenberg-tm electronic
works, and the medium on which they may be stored, may contain
"Defects," such as, but not limited to, incomplete, inaccurate or
corrupt data, transcription errors, a copyright or other intellectual
property infringement, a defective or damaged disk or other medium, a
computer virus, or computer codes that damage or cannot be read by
your equipment.
1.F.2. LIMITED WARRANTY, DISCLAIMER OF DAMAGES - Except for the "Right
of Replacement or Refund" described in paragraph 1.F.3, the Project
Gutenberg Literary Archive Foundation, the owner of the Project
Gutenberg-tm trademark, and any other party distributing a Project
Gutenberg-tm electronic work under this agreement, disclaim all
liability to you for damages, costs and expenses, including legal
fees. YOU AGREE THAT YOU HAVE NO REMEDIES FOR NEGLIGENCE, STRICT
LIABILITY, BREACH OF WARRANTY OR BREACH OF CONTRACT EXCEPT THOSE
PROVIDED IN PARAGRAPH F3. YOU AGREE THAT THE FOUNDATION, THE
TRADEMARK OWNER, AND ANY DISTRIBUTOR UNDER THIS AGREEMENT WILL NOT BE
LIABLE TO YOU FOR ACTUAL, DIRECT, INDIRECT, CONSEQUENTIAL, PUNITIVE OR
INCIDENTAL DAMAGES EVEN IF YOU GIVE NOTICE OF THE POSSIBILITY OF SUCH
DAMAGE.
1.F.3. LIMITED RIGHT OF REPLACEMENT OR REFUND - If you discover a
defect in this electronic work within 90 days of receiving it, you can
receive a refund of the money (if any) you paid for it by sending a
written explanation to the person you received the work from. If you
received the work on a physical medium, you must return the medium with
your written explanation. The person or entity that provided you with
the defective work may elect to provide a replacement copy in lieu of a
refund. If you received the work electronically, the person or entity
providing it to you may choose to give you a second opportunity to
receive the work electronically in lieu of a refund. If the second copy
is also defective, you may demand a refund in writing without further
opportunities to fix the problem.
1.F.4. Except for the limited right of replacement or refund set forth
in paragraph 1.F.3, this work is provided to you 'AS-IS' WITH NO OTHER
WARRANTIES OF ANY KIND, EXPRESS OR IMPLIED, INCLUDING BUT NOT LIMITED TO
WARRANTIES OF MERCHANTIBILITY OR FITNESS FOR ANY PURPOSE.
1.F.5. Some states do not allow disclaimers of certain implied
warranties or the exclusion or limitation of certain types of damages.
If any disclaimer or limitation set forth in this agreement violates the
law of the state applicable to this agreement, the agreement shall be
interpreted to make the maximum disclaimer or limitation permitted by
the applicable state law. The invalidity or unenforceability of any
provision of this agreement shall not void the remaining provisions.
1.F.6. INDEMNITY - You agree to indemnify and hold the Foundation, the
trademark owner, any agent or employee of the Foundation, anyone
providing copies of Project Gutenberg-tm electronic works in accordance
with this agreement, and any volunteers associated with the production,
promotion and distribution of Project Gutenberg-tm electronic works,
harmless from all liability, costs and expenses, including legal fees,
that arise directly or indirectly from any of the following which you do
or cause to occur: (a) distribution of this or any Project Gutenberg-tm
work, (b) alteration, modification, or additions or deletions to any
Project Gutenberg-tm work, and (c) any Defect you cause.
Section 2. Information about the Mission of Project Gutenberg-tm
Project Gutenberg-tm is synonymous with the free distribution of
electronic works in formats readable by the widest variety of computers
including obsolete, old, middle-aged and new computers. It exists
because of the efforts of hundreds of volunteers and donations from
people in all walks of life.
Volunteers and financial support to provide volunteers with the
assistance they need are critical to reaching Project Gutenberg-tm's
goals and ensuring that the Project Gutenberg-tm collection will
remain freely available for generations to come. In 2001, the Project
Gutenberg Literary Archive Foundation was created to provide a secure
and permanent future for Project Gutenberg-tm and future generations.
To learn more about the Project Gutenberg Literary Archive Foundation
and how your efforts and donations can help, see Sections 3 and 4
and the Foundation web page at https://www.pglaf.org.
Section 3. Information about the Project Gutenberg Literary Archive
Foundation
The Project Gutenberg Literary Archive Foundation is a non profit
501(c)(3) educational corporation organized under the laws of the
state of Mississippi and granted tax exempt status by the Internal
Revenue Service. The Foundation's EIN or federal tax identification
number is 64-6221541. Its 501(c)(3) letter is posted at
https://pglaf.org/fundraising. Contributions to the Project Gutenberg
Literary Archive Foundation are tax deductible to the full extent
permitted by U.S. federal laws and your state's laws.
The Foundation's principal office is located at 4557 Melan Dr. S.
Fairbanks, AK, 99712., but its volunteers and employees are scattered
throughout numerous locations. Its business office is located at
809 North 1500 West, Salt Lake City, UT 84116, (801) 596-1887, email
business@pglaf.org. Email contact links and up to date contact
information can be found at the Foundation's web site and official
page at https://pglaf.org
For additional contact information:
Dr. Gregory B. Newby
Chief Executive and Director
gbnewby@pglaf.org
Section 4. Information about Donations to the Project Gutenberg
Literary Archive Foundation
Project Gutenberg-tm depends upon and cannot survive without wide
spread public support and donations to carry out its mission of
increasing the number of public domain and licensed works that can be
freely distributed in machine readable form accessible by the widest
array of equipment including outdated equipment. Many small donations
($1 to $5,000) are particularly important to maintaining tax exempt
status with the IRS.
The Foundation is committed to complying with the laws regulating
charities and charitable donations in all 50 states of the United
States. Compliance requirements are not uniform and it takes a
considerable effort, much paperwork and many fees to meet and keep up
with these requirements. We do not solicit donations in locations
where we have not received written confirmation of compliance. To
SEND DONATIONS or determine the status of compliance for any
particular state visit https://pglaf.org
While we cannot and do not solicit contributions from states where we
have not met the solicitation requirements, we know of no prohibition
against accepting unsolicited donations from donors in such states who
approach us with offers to donate.
International donations are gratefully accepted, but we cannot make
any statements concerning tax treatment of donations received from
outside the United States. U.S. laws alone swamp our small staff.
Please check the Project Gutenberg Web pages for current donation
methods and addresses. Donations are accepted in a number of other
ways including including checks, online payments and credit card
donations. To donate, please visit: https://pglaf.org/donate
Section 5. General Information About Project Gutenberg-tm electronic
works.
Professor Michael S. Hart was the originator of the Project Gutenberg-tm
concept of a library of electronic works that could be freely shared
with anyone. For thirty years, he produced and distributed Project
Gutenberg-tm eBooks with only a loose network of volunteer support.
Project Gutenberg-tm eBooks are often created from several printed
editions, all of which are confirmed as Public Domain in the U.S.
unless a copyright notice is included. Thus, we do not necessarily
keep eBooks in compliance with any particular paper edition.
Most people start at our Web site which has the main PG search facility:
https://www.gutenberg.org
This Web site includes information about Project Gutenberg-tm,
including how to make donations to the Project Gutenberg Literary
Archive Foundation, how to help produce our new eBooks, and how to
subscribe to our email newsletter to hear about new eBooks.