
After the original watercolour in the Mitchell Library, Sydney.
Title: The platypus
its discovery, zoological position, form and characteristics, habits, life history, etc.
Author: Harry Burrell
Release date: April 7, 2026 [eBook #78386]
Language: English
Original publication: Sydney: Angus & Robertson Limited, 1927
Other information and formats: www.gutenberg.org/ebooks/78386
Credits: deaurider and the Online Distributed Proofreading Team at https://www.pgdp.net (This file was produced from images generously made available by The Internet Archive)

v
ITS DISCOVERY, ZOOLOGICAL POSITION,
FORM AND CHARACTERISTICS, HABITS,
LIFE HISTORY, ETC.
BY
HARRY BURRELL, C.M.Z.S.
Co-author of
The Wild Animals of Australasia
With 35 Plates
AUSTRALIA:
ANGUS & ROBERTSON LIMITED
89 CASTLEREAGH STREET, SYDNEY
1927
vi
Wholly set up and printed in Australia by
Eagle Press Ltd., Allen Street, Waterloo
1927
Registered by the Postmaster-General for
transmission through the post as a book
vii
This book is the result of nearly twenty years’ personal observation of the platypus in its haunts, carried on while I was collecting specimens for the University of Sydney and the Commonwealth National Museum; but the limitations under which the work has had to be carried on, and the impossibility of obtaining official sanction to work as a private collector, have prevented me from exploring certain branches of the subject and have brought my field work practically to a standstill. Earlier observers, working without restraint, had opportunities of discovering details which I have been debarred from studying; nevertheless, I have not hesitated to criticize their observations where they differ from my own, and I have ventured to suggest several directions in which scientific investigators may proceed.
All my descriptions have been written from living specimens or from material freshly collected; museum specimens and records have been deliberately ignored. The illustrations also show living platypus, with the exception of a few photographs taken from fresh carcasses; and none of the photographs reproduced have been touched up in any way.
As the book is intended mainly for general readers, measurements have been given in inches where possible; but for scientific purposes it has sometimes been necessary to give them in millimetres. Roughly, 25 mm. equal 1 inch; therefore 3 mm. equal ⅛in., 12 mm. equal ½in., etc.
| PREFACE | vii | |
| I. | INTRODUCTORY | 1 |
| II. | DISCOVERY AND EARLY DESCRIPTIONS | 16 |
| III. | CONTROVERSY ON THE ZOOLOGICAL POSITION | 26 |
| IV. | CONTROVERSY ON THE LAYING OF EGGS | 33 |
| V. | GENERAL CHARACTERISTICS | 46 |
| VI. | NERVOUS ORGANIZATION AND SENSORY PERCEPTIONS | 63 |
| VII. | THE SPUR AND CRURAL GLAND | 76 |
| VIII. | THE NESTING-BURROW | 105 |
| IX. | DISTRIBUTION AND HAUNTS | 139 |
| X. | HABITS | 147 |
| XI. | BREEDING HABITS AND LIFE HISTORY | 167 |
| XII. | PRESERVATION AND ECONOMICS | 193 |
| XIII. | THE PLATYPUS IN CAPTIVITY | 202 |
| BIBLIOGRAPHY | 219 | |
| INDEX | 223 |
| THE PLATYPUS | Frontispiece |
| THE AUTHOR | 6 |
| MRS. HARRY BURRELL FONDLING TWIN NESTLINGS | 6 |
| UPPER AND LOWER JAWS FORCED WIDE OPEN | 10 |
| SKELETON OF PLATYPUS | 11 |
| JAWBONES, SHOWING DIVIDED EXTREMITIES | 14 |
| “AN AMPHIBIOUS ANIMAL OF THE MOLE KIND” | 18 |
| MALE PLATYPUS FLOATING | 18 |
| MALE PLATYPUS WATCHING INTENTLY | 30 |
| FEMALE PLATYPUS LISTENING INTENTLY | 30 |
| VENTRAL VIEW OF MALE PLATYPUS | 34 |
| HEAD OF FEMALE PLATYPUS, SHOWING DIRECT DORSAL VISION | 46 |
| VENTRAL VIEW OF LIPS | 46 |
| PLATYPUS PAWS, EXPANDED FOR SWIMMING, AND CONTRACTED FOR WALKING | 50 |
| FEMALE PLATYPUS SUCKLING YOUNG | 54 |
| FEMALE PLATYPUS | 54 |
| TOP OF LEFT HIND FOOT, WITH SWIMMING MEMBRANE FULLY EXTENDED | 58 |
| SOLE OF RIGHT HIND FOOT | 58 |
| HEAD OF INFURIATED MALE PLATYPUS | 62 |
| NESTLING PLATYPUS, ABOUT FIVE WEEKS OLD | 62 |
| EXPERIMENTS ON SENSORY PERCEPTIONS OF PLATYPUS BENEATH WATER | 66 |
| POISON GLAND CONNECTED BY DUCT TO WELL AT BASE OF SPUR | 94 |
| GENERAL VIEW OF MALE PLATYPUS | 94 |
| SOLE OF RIGHT HIND FOOT, SHOWING SPUR NORMALLY SHEATHED | 98 |
| SOLE OF LEFT HIND FOOT, SHOWING SPUR IN ACTION |
xiv 98 |
| UNEARTHING PLATYPUS, RETREAT STATION, MACDONALD RIVER, N.S.W. | 110 |
| A NEWLY-OPENED NESTING-CAVITY | 114 |
| NEST COMPOSED OF GUM-LEAVES AND GRASS STEMS | 118 |
| BROODING FEMALE PLATYPUS | 118 |
| MODELS OF NESTING-BURROW | 122, 124 |
| NESTING-BURROW WITH TWO OPENINGS | 126 |
| VARIETY IN NESTS | 126 |
| MALE PLATYPUS (1) | 130 |
| MALE PLATYPUS (2) | 130 |
| BURROW WITH FOUR NESTS | 134 |
| PLATYPUS EGGS AND YOUNG | 134 |
| MACDONALD RIVER, N.S.W. | 138 |
| “BLUE HOLE,” NAMOI RIVER, N.S.W. | 138 |
| PLATYPUS MANOEUVRING ALONG A SANDY BOTTOM | 142 |
| MALE PLATYPUS RUNNING AT TOP SPEED (1) | 146 |
| MALE PLATYPUS RUNNING AT TOP SPEED (2) | 146 |
| VENTRAL VIEW OF MALE PLATYPUS | 158 |
| FEMALE PLATYPUS TAKEN DIRECTLY FROM NEST AND PERSUADED TO ADOPT SLEEPING POSITION | 162 |
| TWIN EGGS IN NEST COMPOSED ENTIRELY OF REED ROOTS | 174 |
| TRIPLET NESTLINGS ABOUT FOUR DAYS OLD. NEST COMPOSED OF GUM-LEAVES AND GRASS | 174 |
| FEMALE PLATYPUS IN NEST OF GUM-LEAVES AND GRASS | 178 |
| MILK GLAND FROM LEFT SIDE | 182 |
| BROODING FEMALE STEALTHILY GROVELLING HER WAY FROM HER NEST WHICH HAS BEEN EXPOSED | 186 |
| TWIN NESTLINGS ABOUT FOUR WEEKS OLD, DIRECT FROM BURROW | 206 |
| NESTLING PLATYPUS, ABOUT THREE WEEKS OLD, SHOWING “MILK-LIPS” | 206 |
| EXHIBITING A PLATYPUS IN AN EXPERIMENTAL “PLATYPUSARY,” CONSTRUCTED IN 1910 | 210 |
| PORTABLE “PLATYPUSARY,” DESIGNED FOR TRAVELLING | 210 |
| INTERIOR OF PORTABLE “PLATYPUSARY” | 214 |


1
The first printed description of the platypus appeared in the tenth volume of the Naturalist’s Miscellany (1799) under the name Platypus anatinus and was written by Dr. George Shaw, of the British Museum, where the original skin, received in 1798, is still preserved. In 1800 the German anatomist Blumenbach received a skin from Sir Joseph Banks (one of two sent by Governor Hunter), and described it as Ornithorhynchus paradoxus. Shaw’s generic name Platypus was not permissible, it having been used in 1793 by Herbst for a genus of beetles. The correct name of the animal therefore became Ornithorhynchus anatinus Shaw; but ‘platypus’ has survived in the vernacular, to the exclusion of ‘duck-bill’ and ‘water-mole,’ which were in general use among the earlier colonists.
No animal has given rise to so much controversy among both laymen and professed zoologists. The first specimens which reached England were regarded with suspicion. Shaw’s account, quoted on page 17, shows clearly enough his doubts and fears, which were dissolved by the arrival of further specimens; Home’s anatomical research in 1802 put the genuineness of these beyond all question. But the general feeling is summed up by Knox (1823, p. 27), who writes:—
“It is well known that the specimens of this very extraordinary animal first brought to Europe were considered by many as impositions. They reached England by vessels which had navigated the Indian seas, a circumstance in itself sufficient to rouse the suspicions of the scientific naturalist, aware 2of the monstrous impostures which the artful Chinese had so frequently practised on European adventurers; in short, the scientific felt inclined to class this rare production of nature with eastern mermaids and other works of art; but these conjectures were immediately dispelled by an appeal to anatomy.”
The ‘eastern mermaid’ so mentioned is still to be seen occasionally in curiosity shops, and consists of the skin of the fore-part of a monkey skilfully sewn to the tail-part of a fish.
The singular nature of the platypus was not, however, confined to its external form, and the proof that its duck-like bill really belonged to it by no means ended the troubles of contemporary zoologists. A furred animal should obviously be a mammal—that is to say, it should have a uterus to contain its foetal young, and milk glands with external nipples for the suckling of the young when born.
But Home (1802) found no uterus, no mammary glands, and no nipples. Was the platypus a mammal? Home discovered that the oviducts of the female, instead of uniting to form a uterus as in the majority of mammals, opened separately into a cloaca, as in birds and reptiles.
Here was material for two further controversies. Should Ornithorhynchus be included in the mammalian class, or must a new vertebrate class be added for its inclusion? We know now that we may fairly include the platypus and its only near relative, the echidna, in a sub-class of the Mammalia (the Ornithodelphia), which, though so insignificant numerically, is the equivalent, zoologically, of the larger marsupial sub-class, Didelphia, and of the infinitely greater sub-class Monodelphia, which comprises all other mammals. But it was long before this point was settled. The varying opinions will be detailed in due course.
The second controversy concerned the method of reproduction. Persistent reports came from New South Wales that the platypus laid eggs, but definite evidence was lacking. Sir John Jamison, writing from Sydney on March 18, 1817 (Jamison, 1818, p. 585) states categorically:—“The female 3is oviparous, and lives in burrows in the ground.” But he offers no proof. Lesson (1839, p. 302) writes:—
“It is in New South Wales that one meets those singular and fantastic creatures to which naturalists have not been able to assign a position: the ornithorhynchuses or paradoxals with a duck’s beak, which live in the waters of the rivers, and which lay eggs, creatures set across the path of the scientific method to show its worthlessness.”
These statements were not, however, acceptable to European zoologists, with one or two exceptions, such as Etienne Geoffroy St.-Hilaire. At first two alternatives only were considered. Ornithorhynchus was either oviparous (egg-laying) or ovi-viviparous, that is, producing eggs which hatch within the parent body. But with the discovery in 1824 by Meckel, the great German anatomist, of the mammary glands, a third possibility was manifested. Did the platypus produce its young alive? Blainville and Meckel thought so. Home, and after him the great Richard Owen, stood out for ovi-viviparity. The St.-Hilaires in France, more especially Etienne Geoffroy, fought determinedly for oviparity. The controversy raged for eighty years, significant evidence being rejected from time to time because it did not fit in with the preconceived notions of the person to whose notice it came; and the question was not settled until 1884, when Caldwell reported his finding of the eggs.
Yet another controversy, which can scarcely be said to have been settled even yet, has been concerned with the spur which is found upon the hind limb of the male. This was first described by Shaw (1799), who includes it as a sixth digit, but mentions that it resembles a strong, sharp spur. Home, who also described it, suggested that it was used for holding the female in the sexual embrace. Jamison (1818) first mentioned the serious results of wounds inflicted by it. A number of independent observers in the early twenties—Hill, Traill, Blainville, Knox and others—described the spur and its duct and gland with varying degrees of completeness. Martin 4and Tidswell made a fairly exact investigation of the nature of the fluid secreted by the gland, and of its physiological effects. Numerous writers have quoted cases of severe wounds, while others, including Dr. George Bennett, who handled large numbers of male platypus, never knew the animal to attempt the use of its spurs for offensive purposes.
The mammary glands differ from those of mammals in general, and may be considered as modified sweat-glands. They open by a number of pores upon a small area of skin, there being no teats. After the end of the breeding-season they become so small as to be easily overlooked, and will apparently yield no milk at the time when the young are just hatched from the egg. In these respects they differ very markedly from the milk glands of ordinary mammals, and have afforded ground for further lively arguments. The fiercest opponent of their mammary function was Etienne Geoffroy; he was prepared to take any view other than the obvious one, which seemed fatal to his oviparity hypothesis.
The nesting habits of Ornithorhynchus have also taken a long time to elucidate. The early accounts of the nests are derived from information supplied at secondhand, and their fantastic nature may be due to misunderstanding on the part of the recorder. Hill (1822, p. 622) writes:—
“... a female Ornithorhynchus had been brought in alive, having been found on its nest in a lagoon near Campbell’s River, by Mr. Rawley, who says that he was obliged to tear the nest to pieces before he could get the animal out, the nest being formed of reeds and rushes, with a long tube or entrance into it, out of which the bill of the animal only was visible.”
It is easy to imagine that what Mr. Rawley really said was that he had to dig out the long tube, or burrow, and that, when he came to the nesting-chamber itself, the nest was made of reeds and rushes, out of which only the bill of the animal was visible. Maule (1832) gave the first account of the burrow, which was more fully described by Dr. George Bennett a little later. G. F. Bennett (1877) and Kershaw (1912) have increased our knowledge of the nesting habits.
5
Although at least the natural habits of the platypus have been ascertained, its life history and development are incompletely known. Development in the egg prior to the laying has been dealt with by Wilson and Hill (1908). Similar material obtained by Caldwell in 1883 was never described. A small number of ‘mammary foetus’ stages from the burrow have been dealt with by Owen, Parker, Poulton, Wilson, and others.
Finally, the habits of the platypus have been variously reported by different observers, some of whom would seem to have drawn upon a lively imagination for many of their ‘facts;’ moreover, the original accounts have become garbled in repetition. Most of the tales might have been disproved once for all by one or two published statements from colonists who were familiar with the animal. Curiously enough, however, there are no local accounts—at any rate, I have not been able to find any—during the first half of the nineteenth century. We owe all our early knowledge to English officials and visiting French naturalists. While scientific Europe thirsted for enlightenment, the colonist went blandly on with his pioneering. Fletcher (1901) has pointed out the main reasons for the apparent neglect of zoology in the early days of the colony. Sir Joseph Banks, who organized the early collecting, was more interested in botany; also, that study seemed more likely to have an economic value. So, while the flora was assiduously collected, catalogued, and described, the fauna was practically neglected. Nevertheless, such was the interest in Ornithorhynchus that in 1840 Broderip was able to write:—
“The form is now as familiar to us as most of our European animals, and with regard to its organization and natural history, it is perhaps better known than many.”
Bennett (1860, p. 94) writes:—
“Of all the Australian mammalia, none has excited so much attention as the Platypus or Water-Mole (Ornithorhynchus paradoxus Blum.), both from its peculiar form, and the great desire evinced to ascertain the habits and economy of so singular a creature.”
Lesson’s 6opinion has been quoted above. Shaw (1800, p. 229) says:—“Of all the Mammalia yet known it seems the most extraordinary in its conformation”; and again (1809, p. 80):—
“This most extraordinary and dubious quadruped is a native of Australasia.... If there be no mistake in the anatomical disquisitions hitherto made on the Duckbill, its internal structure is not less extraordinary than its external....”
Verreaux (1848, p. 128) commences his account:—
“The Ornithorhynchus is an animal bizarre of structure, and offers numerous analogies with a host of different species and even classes. In its external form it resembles in some degree the mole as to its body, the beaver as to its tail, and the duck as to its beak. Its internal structure, more astonishing still, resembles that of certain reptiles, and appears to form a link between the Mammals and Lizards.”
Every writer upon the platypus begins with an expression of wonder. Never was there such a disconcerting animal! This wonder finds a very curious expression in the determined efforts made to retain Blumenbach’s name paradoxus, against all the rules of zoological nomenclature. Even Everard Home, the English anatomist, writing less than two years after the publication of Shaw’s original description, uses the name given by the German author.
An amusing and barefaced attempt to justify the use of Blumenbach’s name is that made by Chenu (1879). On p. 349, after mentioning that Shaw in 1792 had described the echidna under the name of Myrmecophaga aculeata, he continues:—
“... four years later, Blumenbach, in 1796, having observed a skin of a curious animal which Banks had sent him, was struck by the resemblance of the curious kind of beak which terminates it to that of a duck, and made it the type of a new genus, which he called, because of this, Ornithorhynchus ...; he applied also appropriately to the species the name paradoxus.... Shaw, not knowing Blumenbach’s work, made the same animal the type of his Platypus anatinus.”
Shaw could scarcely have been expected to know Blumenbach’s 7work, since it did not appear until the year after his own description. In 1796, the year to which Chenu attributes Blumenbach’s observations, the platypus had not been discovered in Australia. But Chenu has not finished, and on p. 352 he writes:—
“As we have said in our general account of the Monotremes, the genus Ornithorhynchus was created, in 1800, by Blumenbach; this name, most happily chosen since it recalls one of the best characters of the included species, has nevertheless been changed by some zoologists. About the same time as Blumenbach, Shaw has named it Platypus, and Wiedemann, Dermipus; but the name of Ornithorhynchus has generally prevailed.”
Despite this brave effort to establish Blumenbach’s priority, Shaw’s specific name is now in general scientific use, though popular works on natural history frequently use the paradoxus of Blumenbach.
Before considering the platypus in all its aspects, a few general considerations may help to a better understanding of this ‘mystery mammal.’ To begin with, it must be borne in mind that existing monotremes are the survivors of a group which once formed the aristocracy of the animal kingdom. The Tertiary is commonly referred to as the Age of Mammals; but in late Mesozoic times there existed mammalian forms which must have been the first wearers of hair and drinkers of milk, and which were, in their day and according to their manner, the lords of creation. They were mammals of a relatively simple type and of a low grade of organization; but there was nothing above them. In great part, however, they almost literally rose on stepping-stones of their dead selves. The higher types to which they gave origin easily defeated them in the competition for existence—ate them, starved them out, thrust them into corners—and themselves went forward to still higher destinies.
The family trees of man and the platypus probably have a common origin in this group of primitive Mesozoic mammals. At the time, however, there was not even the faintest foreshadowing 8of man—not so much as an insignificant tree-shrew, which is supposed by many to be the kind of stock from which he sprang. But the anatomical features which the platypus and the echidna possess in common, and by virtue of which they differ from all other mammals, were, it is fairly certain, characteristic of the whole primitive group of Mesozoic mammals; in regard to these structural features the monotremes have not evolved at all.
Professor W. K. Parker, in his Mammalian Descent (1885), writes:—
“At present, I have only partially worked out the young of one of these kinds—the Ornithorhynchus; but although tolerably familiar with the structure of the Vertebrata generally, I am at a loss, even in this early stage of research, to see the meaning of many things in that type.
“Here is a beast—a primary kind of beast, a Prototherian—whose general structure puts it somewhere on the same level as low reptiles, and old sorts of birds; but in which there are characters much more archaic than anything seen in Serpents, Lizards, Tortoises, Crocodiles, or in Emeus. Therefore the existing reptiles and birds must stand aside as having nothing to do with the family tree of the Monotremes, although in some things they are like these beasts, and many of their organs are formed on a similar pattern; they are all equally below the morphological level of the nobler Mammalia.”
Apart from its primitive simplicity, however, the platypus is a very highly specialized animal—a statement not so self-contradictory as it may seem at first sight. Man himself owes his dominant position to the simplicity of his hand and the specialization of his brain. Kipling’s jungle folk, wanting a name for the human infant which has strayed into their midst, cannot find one among the mammals, and are forced back to the Amphibia—to Mowgli the frog. Man has retained a simple body, which has escaped the need for specialization because his evolution has been projected outside himself into an evolution of tools and weapons. Other animals in need of tools and 9weapons must evolve them from their own bodily parts; we therefore frequently find a specialized adaptation to environmental needs grafted on to primitive simplicity of structure.
Without troubling about its internal anatomy, which presents many archaic features, let us shortly balance the primitive external features against others which show specialization to the peculiar environment and mode of life of Ornithorhynchus. The platypus is an aquatic mammal, but its bodily form shows no very marked adaptation to aquatic conditions. During countless ages of life in the water it has not developed the ‘stream-lines’ of porpoises and seals. Its body is squat, clumsy, and reptilian, as are its short, thick limbs. The adaptations to aquatic life are mainly two: the enormous webbing of the fore-foot or paw, which is the actual swimming-organ, and the flattened tail, which is used as a rudder and helps in diving. Its habits in the water can be judged accurately enough from its form. It is not a rapid swimmer, nor an especially graceful one. It has quiet and leisurely ways, the quick dive when alarmed constituting its one speedy movement.
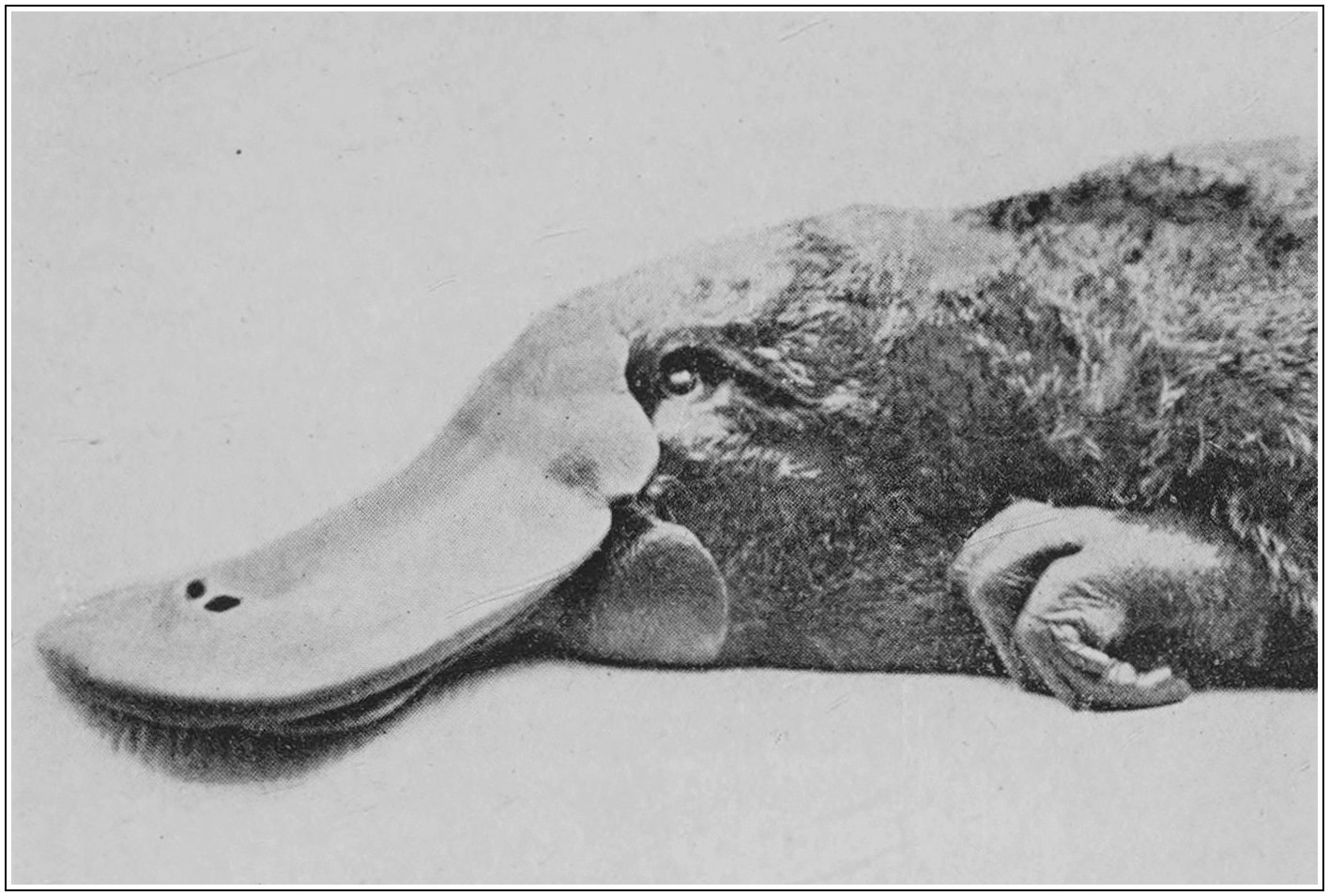
The arrangement of the hair is primitive. It lies straight along the back and belly and the upper and under surfaces of the limbs; but falls away gradually on the sides, following the slope of the surface. There are none of the special sensory hairs (vibrissae) which are almost universal in mammals,—the ‘whiskers’ of the cat will serve as an example. Against this simplicity we must set the extraordinary specialization of the muzzle,[1] with its innumerable special nerve-endings and its remarkable shape. It is easy and usual to compare this muzzle to the beak of a duck. Superficially, the two are much alike; in underlying structure, however, they are totally unlike, and it is with the muzzle of the lower mammals that comparison must be made. The integument is not horny, like that of a bird’s beak, but soft, moist, flexible, and 10extremely sensitive. The under-water life of the platypus is that of a blind animal. Keeping its eyes and ears tightly shut, it feels its way by means of the touch-corpuscles arranged all over the surface of its muzzle; by touch, combined probably with its “sixth sense,” it also finds its food. Altogether, the muzzle of the platypus is possibly the most remarkable organ for sensory perception found in the Mammalia. The resemblance to the beak of a duck has resulted from the similarity of the feeding habits of the two animals. The common use of terms such as ‘horny’ or ‘leathery’ is due to the fact that in stuffed specimens the soft, moist integument dries, hardens, and becomes thinner; but this circumstance is very misleading, as an examination of the living animal will show.
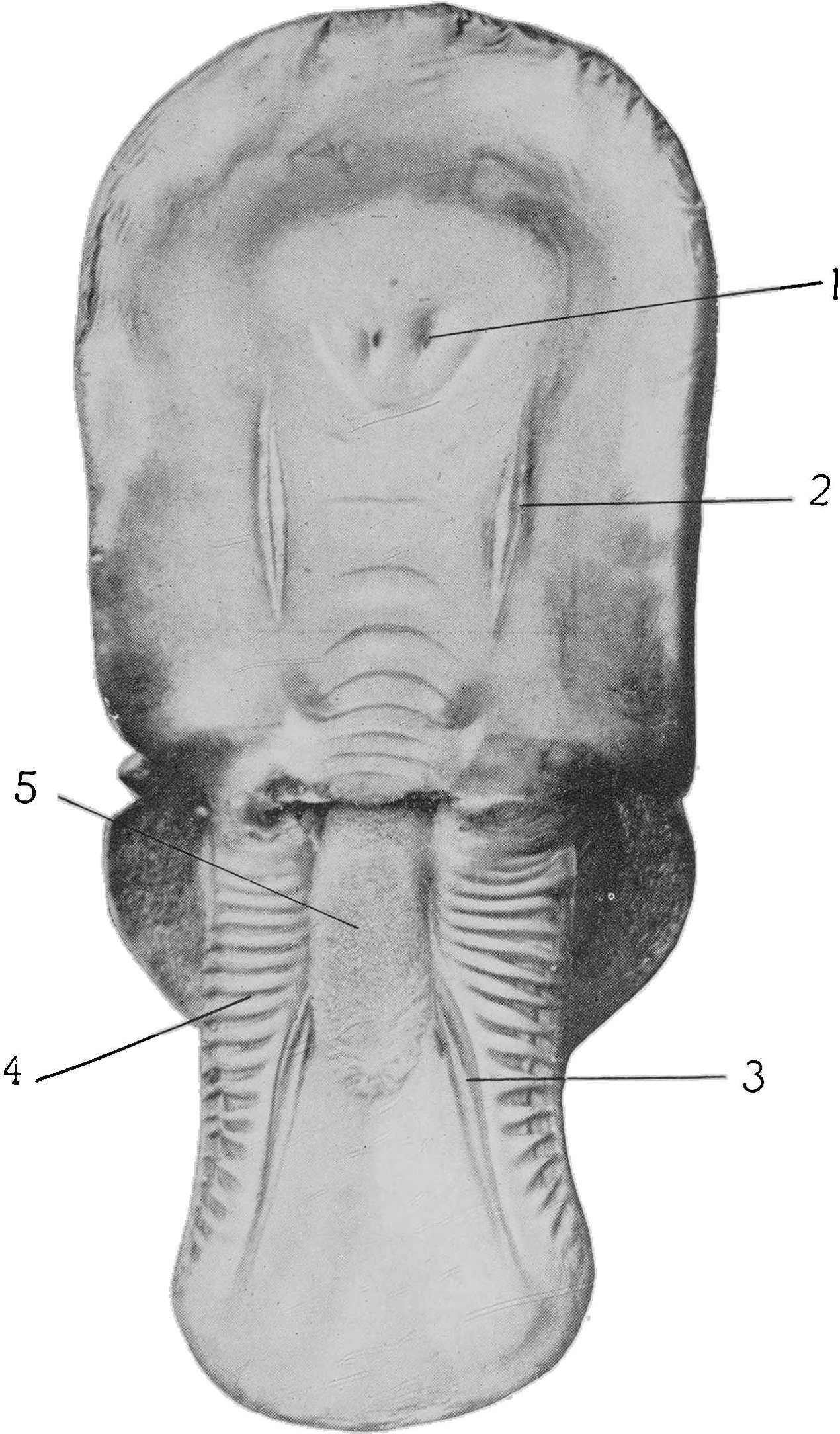

It will be seen in Plates 3 and 4 that both the upper and lower jawbones of Ornithorhynchus are divided at their anterior ends. They are also pliable and permit of the lips being puckered to the extent of forming thereby a small central suction tube. It seems probable that, by pressing the sides of the lower lip tightly against the upper jawbones, the maximum of food-matter is sucked in with the minimum of so-called mud. Most observers agree that the animal feeds to some extent on minute matter, such as the eggs and larvae of aquatic fauna found on the bed of the river. A suction system such as that described would be by far the surest method of obtaining such delicacies, as less sediment and foodstuffs would be disturbed by the gentle methodical probing of closed jaws than by abrupt champing or puddling.
The small, bright eyes are remarkable only for their position high up on the head. Since they are not used for vision under water, they have come to be placed where they will be of most service to the animal as it floats at the surface, munching its catch. The absence of an external ear would seem to be a primitive character; in its place there is a curious modification of the orifice of the auditory meatus. This orifice lies at the posterior end of a facial furrow, the eye lying at the anterior end, while the furrow is incompletely divided into 12two by an oblique fold of skin. The edges of this furrow act as a long pair of lids, by means of which both eye and ear may be tightly closed at the will of the animal. The aural aperture can also be dilated and contracted while the eyes are open, and can be “cocked” to catch sound (see Plate 6, fig. 2). The arrangement of the lids serves also to keep out water while the animal is submerged, and earth while it is engaged in burrowing.
The limbs are primitive and reptilian in general form, and progression on land is clumsy, shuffling, and sinuous, like that of a heavy-bodied lizard. The platypus can, however, raise its body off the ground when moving fast, though it does so only occasionally. The fore-paws serve both as the chief organ of locomotion in the water and as the burrowing tool. The extensive web is used only for swimming, and is folded away under the palms during running and digging operations. Being used for digging, the claws of the fore-paws are straighter, broader, and stouter than those of the hind feet. Both paw and foot have preserved the full primitive number of five digits. The feet are also very useful and versatile. Apart from their ordinary functions, when burrowing they can be reversed for backward progression; in self-defence they are used to grip the foe before applying the spurs; and during copulation they are indispensable. They are also used for toilet purposes.
The mouth exhibits a high degree of specialization. The true teeth (which are, in any case, reduced in number to a few molars and premolars) are lost early, and their place is taken by horny plates. Anteriorly occur two pairs of horny ridges, which are not preceded by teeth, and which serve to hold and kill the slippery prey; this is then passed back and kept segregated by the large bulb which forms the hinder part of the tongue. Two peculiar tooth-like projections, directed forward from the anterior border of this swollen portion, are of service in guiding the food to one side or the other as may be necessary, and possibly for manoeuvring the grit, etc., contained in the pouches.
13
My observations on the cheek-pouches of the platypus have led me to think that what has been written about their use for storing foodstuffs must be wrong, and in order to test my theory I secured (by concussion) three specimens of feeding platypus immediately on their appearance at the surface of the river. I found grit, mud, sediment, and the broken remains (including legs, wings, and hard coverings) of small aquatic fauna, not only cramming the pouches, but covering, in quantity, each set of grinders also. In their entrails, however, mud was the only one of these traceable.
Never yet have I found in the pouches sufficient remains of any one creature to determine its specific kind. I confess myself no expert in that branch of research; nevertheless, one would certainly expect to find at least something recognizable if the pouching of foodstuff beneath water was usual. Because of their bulk, adult shrimps and other large-sized prey cannot possibly be pouched whole, nor can they be passed through to the grinders before being dissected by the secateuring ridges of the bill; this I have frequently observed while feeding platypus in captivity. The necessary act of dissection, prior to mastication, takes place while the animal is floating with its bill skimming the water; therefore large prey must be carried up to the surface in the bill-like section of the capacious jaws.
These observations suggest to me that the purpose of the cheek-pouches is to act as containers of grit rather than as receptacles for food. The pouches open out at each side directly on to the grinders, which project very little above the level of the jaws, and my opinion is that the coarse grit aids considerably in their functioning as teeth, and, further, tends to toughen the gums and thus convert them into grinders, maintaining these essential conditions after the true teeth of the nestling stage have disappeared.
The fact that only the hard remains of foodstuff are found mixed with the grit may mean that this mixture is drawn upon and repouched, as occasion demands, over and 14over again; but whether the grit is deliberately pouched before meals, or collected haphazard, or even accidentally, with the food, I cannot say. It probably depends on the class of food offering at any particular time and on the frequently changing conditions of river beds—from mud to sand and shingle or otherwise—according to varying seasonal changes by which platypus and its many forms of living prey are naturally ruled.
It should be understood that the platypus, from choice or necessity, consumes various kinds of food, that some kinds are normally available only at certain times of the year, and that in abnormal seasons the variety, as well as the quantity, would be affected. At such times one finds platypus gorging themselves with inferior muddy matter, which would not, by preference, be included in their bill of fare. The various statements made on this subject by observers may, therefore, all be correct, though each is sceptical of the accuracy of the others.
Finally, mention must be made of the curious integumentary flap which projects backwards from the base of the naked muzzle, both above and below. This is not altogether, as has sometimes been suggested, a shield for the protection of the head during feeding and burrowing. It is an extension of the area for sensory perception, and from its position would appear to serve much the same function as the “whiskers” about the mouths of other mammals.
To this mixture of old and new physical characteristics there must correspond a somewhat similar mixture of psychological characteristics. Much of the behaviour of the platypus cannot be adequately explained by its present conditions of life, and there is no obvious reason why it adopted its present extraordinary precautions for the safety both of itself and of its young. The Murray cod and the monitor lizards may have eaten the platypus out of certain rivers, since there seems to be nothing that they will not eat; but of this there is no reliable evidence. The instinct for caution must be a heritage from some olden time, when it had to contend with conditions 15much more strenuous. During that period, which our imagination cannot reconstruct, the ancestral platypus may have used its cerebral cortex to the full for the winning of a livelihood in open competition, and for keeping safe, not only itself, but its young. It has a large brain for its size, proportionately much larger than those of many of the lower Didelphia and Monodelphia. Its brain does not show any of the furrowed convolutions which are looked upon as indicating a high degree of mental development, but the smooth condition may have been acquired. The brain of echidna, which is in general a much more stupid creature, is convoluted, and indicates that this animal still has enough worries to produce wrinkles on its cortical surface.

16
In the second volume of Collins’s Account of the English Colony in New South Wales (1802, p. 62), under date ‘November, 1797,’ there occurs the following account of what was apparently the first platypus captured by a European:—
“The Kangaroo, the Dog, the Opossum, the Flying Squirrel, the Kangaroo Rat, a spotted Rat, the common Rat, and the large Fox-bat (if entitled to a place in this society), made up the whole catalogue of animals that were known at this time, with the exception which must now be made of an amphibious animal, of the mole species, one of which had been lately found on the banks of a lake near the Hawkesbury. In size it was considerably larger than the land mole. The eyes were very small. The fore-legs, which were shorter than the hind, were observed, at the feet, to be provided with four claws, and a membrane, or web, that spread considerably beyond them, while the feet of the hind legs were furnished, not only with this membrane or web, but with four long and sharp claws, that projected as much beyond the web, as the web projected beyond the claws of the fore-feet. The tail of this animal was thick, short, and very fat; but the most extraordinary circumstance observed in its structure was, its having, instead of the mouth of an animal, the upper and lower mandibles of a duck. By these it was enabled to supply itself with food, like that bird, in muddy places, or on the banks of the lakes, in which its webbed feet enabled it to swim; while on shore its long and sharp claws were employed in burrowing; nature thus providing for it in its double or amphibious character. These little animals had been frequently noticed rising to the surface of 17the water, and blowing like the turtle.”
Collins also included an engraving of the animal “from a drawing made on the spot by Governor Hunter”, which appears on p. 63, with the following legend:—
“An Amphibious Animal of the Mole Kind which Inhabits the Banks of the fresh water Lagoons in New South Wales—its fore-feet are evidently their principal assistance in Swimming and their hind feet having the Claws extending beyond the Web’d part are useful in burrowing.”
A reproduction of this engraving is given on Plate 5.

The inference to be drawn from Collins’ description, read in conjunction
with the reference (under the engraving) to the hind feet being useful
in burrowing, is that the burrowing is done with the hind feet. If that
meaning was intended, Collins was evidently misled by anatomical probabilities
in assuming the function of the projecting hind-claws for that
purpose and ignoring the possibility of the projecting web of the fore-paws
being “palmed” to admit of the claws being used for burrowing.
The actual method of burrowing is described on pages 112 and 120.
It has not been possible to trace with certainty the subsequent history of this individual platypus. In the following year (1798), however, a specimen came into the hands of an English naturalist named Dobson, and—as already mentioned on page 1—was described by George Shaw. Shaw’s description was illustrated by two plates—Pl. 385, giving a view of the whole animal, and Pl. 386, on which are figured three aspects of the “bill”, and the fore-paw and hind foot. These are the first published figures. The work in which Shaw’s original description occurs is not easily accessible to the general reader, which fact is some justification for quoting the full text. But the surprise of the zoologist at the novel nature of the animal before him, and his uneasy doubt whether he is not being deceived by a clever taxidermist’s wiles, render the description too delightful to be omitted:—
“The animal exhibited on the present plate constitutes a new and singular genus, which, in the Linnaean arrangement of Quadrupeds, should be placed in the order Bruta, and should stand next to the genus Myrmecophaga.
“Of all the Mammalia yet known it seems the most extraordinary in its conformation, exhibiting the perfect resemblance of the beak of a Duck engrafted on the head of a quadruped. So accurate is the similitude, that, at first view, it naturally excites the idea of some deceptive preparation by artificial means: the very epidermis, proportion, serratures, manner of opening, and other particulars of the beak of a shoveler, or other broad-billed species of duck, presenting themselves18 to the view: nor is it without the most minute and rigid examination that we can persuade ourselves of its being the real beak or snout of a quadruped.
“The body is depressed, and has some resemblance to that of an Otter in miniature: it is covered with a very thick, soft, and beaver-like fur, and is of a moderately dark brown above, and of a subferruginous white beneath. The head is flattish, and rather small than large: the mouth or snout, as before observed, so exactly resembles that of some broad-billed species of duck that it might be mistaken for such: round the base is a flat, circular membrane, somewhat deeper or wider below than above; viz. below near the fifth of an inch, and above about an eighth. The tail is flat, furry like the body, rather short, and obtuse, with an almost bifid termination: it is broader at the base, and gradually lessens to the tip, and is about three inches in length: its colour is similar to that of the body. The length of the whole animal from the tip of the beak to that of the tail is thirteen inches: of the beak an inch and a half. The legs are very short, terminating in a broad web, which on the fore-feet extends to a considerable distance beyond the claws; but on the hind feet reaches no farther than the roots of the claws. On the fore-feet are five claws, straight, strong, and sharp-pointed: the two exterior ones somewhat shorter than the three middle ones. On the hind feet are six claws, longer and more inclining to a curved form than those on the fore-feet: the exterior toe and claw are considerably shorter than the four middle ones: the interior or sixth is seated much higher up than the rest, and resembles a strong, sharp spur. All the legs are hairy above: the fore-feet are naked both above and below; but the hind feet are hairy above, and naked below. The internal edges of the under mandible, (which is narrower than the upper) are serrated or channelled with numerous striae, as in a duck’s bill. The nostrils are small and round, and are situated about a quarter of an inch from the tip of the bill, and are about an eighth of an inch distant from each other. There is no appearance of teeth: the palate is removed, but seems to have resembled that of a duck: the 19tongue also is wanting in the specimen. The ears or auditory foramina are placed about half an inch beyond the eyes; they appear like a pair of oval holes of the eighth of an inch in diameter, there being no external ear. On the upper part of the head, on each side, a little beyond the beak, are situated two smallish, oval, white spots; in the lower part of each of which are imbedded the eyes, or at least the parts allotted to the animal for some kind of vision; for from the thickness of the fur and the smallness of the organs they seem to have been but obscurely calculated for distinct vision, and are probably like those of Moles, and some other animals of that tribe; or perhaps even subcutaneous; the whole apparent diameter of the cavity in which they were placed not exceeding the tenth of an inch.

“When we consider the general form of this animal, and particularly its bill and webbed feet, we shall readily perceive that it must be a resident in watery situations; that it has the habits of digging or burrowing in the banks of rivers, or under ground; and that its food consists of aquatic plants and animals. This is all that can at present be reasonably guessed at: future observations, made in its native regions, will, it is hoped, afford us more ample information, and will make us fully acquainted with the natural history of an animal which differs so widely from all other quadrupeds, and which verifies in so striking a manner the observation of Buffon; viz. that whatever was possible for Nature to produce has actually been produced.
“On a subject so extraordinary as the present, a degree of scepticism is not only pardonable, but laudable; and I ought perhaps to acknowledge that I almost doubt the testimony of my own eyes with respect to the structure of this animal’s beak; yet must confess that I can perceive no appearance of any deceptive preparation; and the edges of the rictus, the insertion, &c., when tried by the test of maceration in water, so as to render every part completely moveable seem perfectly natural; nor can the most accurate examination of expert anatomists discover any deception in this particular.
“The Platypus is a native of Australasia or New Holland, 20and is at present in the possession of Mr. Dobson, so much distinguished by his exquisite manner of preparing specimens of vegetable anatomy.”
Two years later Shaw repeated this description in his General Zoology (1800, Vol. 1, pp. 228-232), with the following remarks as preface:—
“Of this most extraordinary genus the first description appeared in the Naturalist’s Miscellany; but as the individual there described was the only one which had been seen, it was impossible not to entertain some distant doubts as to the genuine nature of the animal, and to surmise, that, though in appearance perfectly natural, there might still have been practised some arts of deception in its structure. I, therefore, hesitated as to admitting it into the present History of Quadrupeds. Two more specimens, however, having been very lately sent over from New Holland, by Governor Hunter, to Sir Joseph Banks, the suspicions before mentioned are now completely dissipated.”
It is clear from this that up to 1800 only three specimens had come under Shaw’s notice. Whether from the words “as the individual there described was the only one which has been seen” we are justified in concluding that this individual was identical with that mentioned by Collins, is another matter; but it seems at least probable. Little is known of the subsequent history of the skin which served as the type for Shaw’s description, but it was ultimately presented by Latham to the British Museum, where it is now preserved. Mr. Oldfield Thomas writes:—
“I have lying in front of me the original type skin of Shaw’s Platypus anatinus, but I am afraid I can give you practically no details of its history more than is printed in my Catalogue of Mammalia.
“It is an immature male with the spurs cut off (No. 110c of Gray’s List of Mammalia in the British Museum, 1843) 343 millimetres in extreme length. There were no registers in those days.”
21
Mr. Tom Iredale has directed attention to the fact that Hunter, besides the specimens which he had forwarded to London, also sent to Newcastle a platypus which reached the Literary and Philosophical Society of that city in 1798, together with a wombat, probably the first to reach England. These two animals were described by Bewick in an addendum to the fourth and all subsequent editions of his General History of Quadrupeds. I have not seen the fourth edition, but quote from the seventh (1820, p. 524):—
“We are favoured by the Literary and Philosophical Society of Newcastle upon Tyne, with the figures and descriptions of two very rare animals, sent to them from New South Wales, by James Hunter, Esq. governor of that settlement.... The other seems to be an animal sui generis; it appears to possess a threefold nature, that of a fish, a bird, and a quadruped, and is related to nothing that we have hitherto seen: we shall not attempt to arrange it in any of the usual modes of classification, but content ourselves with giving the description of both these curious animals as they have been transmitted to us.”
On p. 528 appears a woodcut of the platypus with a legend—“An Amphibious Animal”—and the following description:—
“is found in freshwater lakes, and is about the size of a small Cat; it chiefly frequents the banks of the lakes; its bill is very similar to that of a Duck, and it probably feeds in muddy places in the same way; its eyes are very small; it has four short legs; the fore-legs are shorter than those of the hind, and their webs spread considerably beyond the claws, which enables it to swim with great ease; the hind legs are also webbed, and the claws are long and sharp. They are frequently seen on the surface of the water, where they blow like a turtle: their tail is thick, short, and very fat.
“The natives say they sometimes see them of a very large size.”
Bewick made no attempt to name the animal, and it appears in his index simply as “An Amphibious Animal of New South Wales.”
22
The first four specimens sent to England were in the form of dried skins. Home (1802, p. 69), writing after he had received specimens (in spirit) of the integumentary flap which surrounds the base of the muzzle, uses the following words:—
“In dried specimens that have been brought to England, the flap has been contracted in drying, and stands up perpendicularly. This, however, is now ascertained not to be its natural situation.”
As Home received from Banks the material which he described, it is reasonably certain that he refers to the two individuals sent by Hunter. One of these was sent to Blumenbach at Göttingen, who records (1800, p. 206) that he has received a stuffed specimen from “the Hon. Baronet Banks,” the first that had been sent from England to the Continent. This individual he described as Ornithorhynchus paradoxus. Weidemann, who also described the platypus in 1800 (as Dermipus anatinus), apparently had no specimen, and merely gave a German version of Shaw’s description with an indifferent copy of his figure.
Blumenbach (1801, p. 724) published an extract from a letter from Banks in which he stated that he now had a couple of spirit specimens, and gave a short description of them. These specimens formed the basis of the first anatomical description by Home (1802). The official correspondence in the Historical Records of New South Wales shows that one of these was sent by Governor King, who succeeded Hunter. King writes to Banks on 28 September, 1800:—“I send you by the hands of Capt. Kent, who is nephew to Governor Hunter, and commands the Buffalo, which he takes home with him, a cask in which is a water-mole ... (Historical Records of N.S.W., IV., p. 205), which I hope will all get safe, particularly the mole, which you desired.” In due course Kent writes from on board the Buffalo in Portsmouth harbour, on 1 July, 1801:—“The keg containing the water-mole and other articles in spirits ... I have still on board” (loc. cit., p. 427). We can find no record of the other spirit specimen sent about this time, but it is probable 23that this also was sent by King. A pencil note in Banks’s handwriting records that two animals were received.
A further spirit specimen is that of which Home (1802, p. 356) writes:—“... had I not been favoured by Sir Joseph Banks with a specimen of the paradoxus, brought from New South Wales by Mr. Belmain.” Balmain came out as assistant surgeon with the First Fleet, succeeded John White as head surgeon in 1796, and returned to England on leave by the Albion, arriving in March, 1802. A box of specimens was placed by King in his personal charge for delivery to Banks (Historical Records of N.S.W., IV, pp. 514-5).
It is of interest to note in passing that King uses the name ‘water-mole’ in the letter quoted above, but in a later letter to Banks (dated 5th June, 1802) writes:—“I do not recollect what things I sent by the Buffalo besides the duckbill....” (loc. cit., p. 784). These are the first mentions noted of the two names which were in vernacular use for many decades.
The first account of the habits of the platypus is that given to Home by Hunter. Home writes (1802, p. 67):—
“Governor Hunter, who has lately returned from New South Wales, where he had opportunities of seeing them alive, has favoured me with the following particulars respecting them.
“The Ornithorhynchus is only found in freshwater lakes, of which there are many in the interior parts of the country, some three-quarters of a mile long, and several hundred yards broad. This animal does not swim upon the surface of the water, but comes up occasionally to breathe, which it does in the same manner as the turtle. The natives sit upon the banks, with small wooden spears, and watch them every time they come to the surface, till they get a proper opportunity of striking them. This they do with much dexterity; and frequently succeed in catching them in this way.
“Governor Hunter saw a native watch one for above an hour before he attempted to spear it, which he did through the neck and fore leg; when on shore, it used its claws with so 24much force, that they were obliged to confine it between two pieces of board, while they were cutting off the barbs of the spear, to disengage it. When let loose, it ran upon the ground with as much activity as a land tortoise; which is faster than the structure of its fore-feet would have led us to believe. It inhabits the banks of the lakes, and is supposed to feed in the muddy places which surround them; but the particular kind of food on which it subsists, is not known.”
As the second volume of Collins’s New South Wales was not published until 1802—by which time Home’s paper had appeared, and a copy had been sent by him to Collins—the author was able to include a further account of the platypus (1802, pp. 321-8), abstracted from Home.
Hunter’s account forms the basis of most statements concerning the economy of the animal which appear in European works on natural history during the next three decades. Since the first discovery was made ‘on the banks of a lake near the Hawkesbury,’ the platypus is invariably described as a dweller on the banks of lakes, and its more customary habitat in rivers is not mentioned. Mr. Henry Selkirk, who has a long-standing familiarity with the Hawkesbury district, informs me that he thinks it probable that the lake mentioned as the site of the first discovery is one of several lagoons close to the town of Richmond. He himself, as a boy, made his first acquaintance with the platypus in those waters.
These meagre particulars are all that I have been able to gather concerning the discovery of the platypus. In view of the interest which it excited in Europe, and of the keen demand for specimens that must have followed immediately upon the first description, it is rather remarkable that contemporary journals and official documents should be so barren of reference to it. White and Tench, who would surely have told us more had they been in the colony at the time, had unfortunately returned to England before the discovery. Hunter, to whom we owe the first drawing of the animal, as well as the first description of its habits, appears to have taken his recall very 25much to heart. Little is known of his subsequent history, and he made no effort to continue his journal over the period of years which included the first finding of the platypus.
It is certain, however, that there was a brisk traffic in specimens. When the great controversy as to the zoological position of the platypus, and its method of reproduction, began in the second decade of the nineteenth century, most European museums were in possession of material. Much of this was, doubtless, brought back by the various French scientific expeditions of which mention is made hereafter; but a great deal must have passed through English hands, and records of this may be presumed to exist somewhere. They have not, however, been revealed, and the only other contemporary account which I can find is in Barrington’s History of New South Wales (1804), which is substantially reproduced, without acknowledgment, from Home.
26
Vertebrate animals are now divided into five classes—Fishes, Amphibia, Reptiles, Birds, and Mammals—distinguished from one another by such obvious characters that no zoological knowledge is required to classify any normal member of one of these great groups. When, however, the position of Ornithorhynchus first came under discussion, Amphibia were usually included with Reptilia, making four classes only.
To Shaw, the first zoologist to handle a platypus, the animal was obviously a mammal, since it had a furry covering; he placed it in the lowest Linnean Order, Bruta, which included what we know now as the Edentata, or toothless mammals (though not all are toothless), anteaters, sloths, and the like. Shaw had only a stuffed skin, and could know nothing of the anatomy. Yet he was fully seized of its remarkable character, as may be judged by his reference to Buffon’s aphorism in the passage quoted in the preceding chapter.
Blumenbach (1800) made anatomical observations on the bones contained within the skin sent him by Banks, but his paper is not available in Australia. The first account of the anatomy of specimens preserved in spirit is that by Home (1802), who received a male and female from Sir Joseph Banks, both probably sent by Governor King.
Home was an anatomist, not a systematist; in his first paper he does not concern himself with the systematic position of the animal whose anatomy he was describing. He notes the remarkable character of the organs of reproduction both in male and female, and remarks (1802, p. 81):—
“This structure27 of the female organs is unlike anything hitherto met with in quadrupeds; since, in all of them that I have examined, there is the body of the uterus, from which the horns go off as appendages. The opossum differs from all other animals in the structure of these parts, but has a perfectly formed uterus; nor can I suppose it wanting in any of the class Mammalia.”
Finding that these organs were not comparable with those of mammals, Home was led to examine the corresponding parts in birds and reptiles, and found most resemblance to the organs of ovi-viviparous lizards. He concludes (p. 82)
“There is therefore every reason to believe that this animal also is ovi-viviparous in its mode of generation.”
Later in the same year Home examined the anatomy of the echidna (Spiny Anteater), which Shaw has described in 1792 as Myrmecophaga aculeata. He at once recognized its close affinity to the platypus, and included it in the same genus as Ornithorhynchus hystrix (1802, p. 356). He sums up on p. 360 as follows:—
“The peculiar characters of the Ornithorhynchus, as a genus, or more properly a tribe of animals, are:
“The male having a spur upon the two hind legs, close to the heel.
“The female having no nipples.
“The beak being smooth, while the rest of the animal is covered with hair.
“The tongue having horny processes, answering the purposes of teeth.
“The penis of the male being appropriated to the passage of semen; and its external orifice being subdivided into several openings, so as to scatter the semen over an extent of surface, while the urine passes by a separate canal into the rectum.
“The female having no common uterus; and the tubes, which correspond to the horns of the uterus in other quadrupeds, receiving the semen immediately from the penis of the male.
“These characters distinguish the Ornithorhynchus, in a 28very remarkable manner, from all other quadrupeds, giving this new tribe a resemblance in some respects to birds, in others to the Amphibia; so that it may be considered as an intermediate link between the classes Mammalia, Aves, and Amphibia....
“Between it and the bird, no link of importance seems to be wanting.”
In a later paper (1819, p. 237) he returns to this linkage with birds:—
“The mode of formation of the ova in the ornithorhynchi, constitutes the intermediate link between that of the American opossum and the bird.”
This talk of “links” between classes, long before the advent of Charles Darwin, is interesting. Home may not have used the word in an evolutionary sense, but it seems likely that he did. In reading these early discussions on the platypus, one is naturally struck by the implied acceptance of the evolutionary idea which permeates them.
Home’s opinion was, then, that the genus Ornithorhynchus constituted a new tribe of animals, to which, however, he gave no designation. This opinion was based chiefly upon the structure of the organs of reproduction in the female, and a brief explanation of the importance of these organs may prove helpful to general readers. In birds and reptiles the female organs consist of a pair of ovaries placed on the dorsal wall of the body cavity, one on each side of the middle line. In close relation to the ovaries are the wide funnel-shaped mouths of a pair of tubes, the oviducts, which receive the ova shed from the ovaries, invest them with additional coats, albumen, shell-membranes, and shell, and pass them backwards to the cloaca—the common terminal chamber, into which the alimentary canal and urinary organs also open. In birds, the gradual development of a large-yolked egg, and the danger to the mother involved in the production of such large eggs in both oviducts simultaneously, has led to the reduction of the right ovary and oviduct to functionless vestiges, those of the left side alone remaining functional.
In the higher mammals the organs are essentially the same, 29but with certain marked differences. There is no longer a cloaca, the terminal aperture of the food-canal being quite separate from that of the urinogenital organs. Both ovaries and both oviducts are functional, since the mammalian ovum is very small. But, except in a few lower forms, the oviducts are no longer separate tubes; a varying amount of fusion has taken place, the fused portion being the uterus, or womb. This may be a single cavity, its double origin indicated only by the horns of which Home speaks and into the apices of which open the upper portions of the oviducts; or it may be more or less divided by a septum and thus afford more obvious evidence of its double nature. Within this uterus the young develop and are nourished until birth, being attached to the maternal wall by a placenta. Where no placenta exists, as in most marsupials, the young are born in a very immature condition; they become attached to an external teat, and are nourished by milk, which they cannot suck but which is impelled into them by muscular reaction on the mammary glands of the mother.
Turning again to Ornithorhynchus, we find that the oviducts open separately into a cloaca, that the left ovary and oviduct alone are functional, and, as will be shown later, that a shelled egg is laid, which is hatched outside the body of the mother. This last fact was not, of course, known to the naturalists who first discussed the position of the platypus in zoological systems. In all these features the platypus agrees with birds and reptiles, rather than with mammals. It must also be held in mind that its mammary glands, the outstanding characteristic of the mammalian class, had not yet been discovered; it was not until 1824 that Meckel announced their discovery—which was not, even then, accepted by all zoologists.
It is hardly to be wondered at that zoologists were in doubt as to the systematic position of this weird animal. Etienne Geoffroy (1803, p. 125) included it with the echidna in a new order, Monotremata, characterized by:—“Digits clawed; no true teeth; a common cloaca opening to the exterior by a single 30orifice;” but he did not give any precise indication as to where this order should be placed. Tiedemann (1808) evaded the difficulty by placing it in an appendix. Lamarck (1809) created a new class, Prototheria, for platypus and echidna, deciding that they were not mammals, for they had no mammary glands and were probably oviparous; they were not birds, for their lungs differed, and they had no wings; and they were not reptiles, for they possessed a four-chambered heart. Illiger (1811) placed them in a division Reptantia, intermediate between reptiles and mammals. Blainville (1812), on the other hand, was convinced that they were mammals, though belonging to a separate order, Ornithodelphia. He was the first to indicate their many close points of agreement with the marsupials, and gave a long list of mammalian resemblances.
Etienne Geoffroy was convinced from the outset that the platypus was oviparous, and was not a mammal. He entered into a controversy with Blainville which lasted for many years. Van der Hoeven and Latreille were on his side, while Cuvier and Meckel supported Blainville. Isidore Geoffroy, in his article on Ornithorhynchus in the Dictionnaire Classique d’Histoire Naturelle (1827) includes the animal in Mammalia, with a query, and gives a good summary of the position at that date, just after Meckel’s discovery of the mammary glands. He supports his father’s views, and argues that, even if the glands discovered by Meckel are true mammary glands (which he doubts), this does not necessarily make Ornithorhynchus a mammal. He cites (p. 395) his father, who claims (1826) that the so-called mammary glands are homologous with the flank glands of shrews. They differ remarkably in structure from any known milk-producing gland, and particularly from those of marsupials, to which one would expect to find most resemblance. Besides, how can one conceive of an animal with a horny beak sucking, especially when the supposed milk gland has no teat? It may be a scent-gland; or perhaps its secretion serves to coat the fur and render it less permeable 31by water. The crux of the whole matter is—Does Ornithorhynchus lay eggs, or does it not?
Meckel reported the discovery of the mammary glands in 1824, but it was not until 1826 that his full description of them was published. The glands lie between the main abdominal muscles and the muscular sheet (panniculus carnosus) lining the skin. They are two in number, each consisting of a considerable number of tubules which open by minute pores upon a small area of skin; there is no teat. They are large during the suckling period, but become very much reduced after it is over, and this accounts for their being so long overlooked. We now know that they are of the nature of modified sweat-glands, and so differ from the mammary glands of mammals in general; there was thus some justification for Geoffroy’s doubts. He returns to the charge again in 1829, and first traverses the conclusions he had reached and published in 1827. Monotremes, he maintained, were not mammals, as was thought, and as was apparently indicated by the identity of several important organs, e.g. limbs, hair, lungs, blood-vessels, heart with two ventricles, closure of the chest by a diaphragm, etc. They must be removed from the Mammalia, because their reproductive organs and several other parts placed them amongst the oviparous classes. They could not be included in the birds, because they had neither wings nor feathers. Nor could they be placed amongst the reptiles, for their blood was warm, and their lungs were enclosed in pleurae, and shut off from the abdomen by a diaphragm. Still less were they fish, which breathe by means of gills. They were therefore mammals without the mammalian character, without the special organization of viviparous animals, without all the consequences and functions of an apparatus capable of producing a placental foetus. Rejected on all sides, they did not belong to any recognized class of vertebrates; they must have a special class to themselves.
In spite of all this, Meckel presumes to discover mammary glands, and announces the fact in 1824. For two years scientific 32men were kept in suspense; then in 1826 appeared his wonderful description in which Blainville found confirmation of the notions which he had published in 1808. But Meckel writes (1826, pp. 159-160):—
“I thought, and I said, that the new elements introduced into this controversy were not what they had been announced to be. It is true that a richly glandular apparatus had been discovered on each side of the abdomen of an Ornithorhynchus; but it was a little too hasty to decide and declare that it was a mammary gland. I examined the facts again in a specimen preserved in spirit which formed part of Baron Cuvier’s collections; I saw a glandular structure, but simple, and merely composed of a considerable number of ampullae with long necks. There were none of the characters which distinguish a milk gland.”
Geoffroy proceeds to announce the discovery of eggs, the sad story of which will be told in another place, and concludes (p. 164):—
“We may to-day regard it as certain that the first zoological division, comprising vertebrate animals, should henceforth be divided into the five following types: Mammals, Monotremes, Birds, Reptiles, and Fishes.”
Despite these strenuous efforts, however, the monotremes were generally considered to be mammals, although the actual method of reproduction was not definitely known until 1884, and although there are still some doubtful points about the lactatation.
In this connection Etienne Geoffroy’s final desperate effort to disprove the mammary nature of the glands may be mentioned. He compared them to the so-called mammary glands of whales and porpoises, which secrete not milk but mucus. This coagulates on being ejected into the water, and is then devoured by the young. Unfortunately, just as he had elaborated this hypothesis, he had the ill-fortune to secure a nursing porpoise, and found that the glands secreted milk. Nevertheless, this endeavour may account for a statement by Verreaux which we shall have to consider later.
The production of milk by Ornithorhynchus was definitely recorded by Maule (1832) and by Bennett (1833).

33
Home (1802), the first anatomist to examine the platypus, suggested that, from the nature of the reproductive organs in the female, it might be ovi-viviparous. In 1819 (p. 238) he makes a categorical statement, without giving any evidence in support, as follows:—
“In the ornithorhynchi the yelk-bags are formed in the ovaria; received into the oviducts, in which they acquire the albumen, and are impregnated afterwards; the foetus is aerated by the vagina, and hatched in the oviduct, after which the young provides for itself, the mother not giving suck.”
Had this been put forward as merely the probable course of events, there would perhaps have been some justification for it. But it is given as a statement of fact, and it is hard to imagine how a worker of Home’s standing could have committed himself to so dogmatic a statement—which time has proved to be entirely erroneous, and for which not one jot of actual evidence existed at the time it was written.
Blumenbach (1801), after a study of the skull, suggested that the platypus might be oviparous; but although this long shot ultimately received confirmation, it hardly seems necessary to credit the Göttingen anatomist with any very remarkable acumen.
Those zoologists who considered the platypus to be definitely a mammal, of course believed that it brought forth its young alive. Among these we may include Blainville, Cuvier, and Oken. The last-named suggested (1817) that the mammary glands might occupy some unusual situation, and so might have escaped notice; later (1823), commenting on the ovarian 34ovum described by Dr. Patrick Hill (see below), he points out its resemblance to the mammalian Graafian follicle, and concludes by saying that the tales of oviparity and ovi-viviparity have all arisen from failure to observe the mammary glands. Oken’s assurance of the existence of mammary glands was justified, as we have seen, by the discovery of these glands by Meckel, announced in 1824.

It seemed likely, then, that despite the opposition of Etienne Geoffroy and the extraordinary form of the animal itself and of its reproductive apparatus, Ornithorhynchus would turn out to be quite an ordinary mammal, which produced its young alive. But persistent statements came from the colonists of New South Wales that it laid eggs. Sir John Jamison (1818, p. 585) wrote, without comment, and as if citing an established fact:—“The female is oviparous, and lives in burrows in the ground.” This would seem to indicate that nesting-burrows and eggs had been definitely observed. But no actual evidence was forthcoming to calm the agitation of European zoologists.
Dr. Patrick Hill (1822, p. 623) examined what he believed to be an impregnated female, in the left ovary of which was a large Graafian follicle. This specimen was taken to England by a Mr. Scott, and, as is recorded in a footnote, was presented to the University of Oxford. Hill’s error as to the nature of the ovum gave rise to a considerable misunderstanding. Fleming (1822, p. 215) stated that the eggs of Ornithorhynchus had been transmitted to London; although he was aware that these ‘eggs’ were merely ovarian ova, this statement was repeated in several French works. Hill concluded his letter with the following words:—“Cookoogong a native, chief of the Boorah-Boorah tribe, says, that it is a fact well known to them, that this animal lays two eggs, about the size, shape, and colour of those of a hen; ...” As the egg of the platypus measures only three-quarters of an inch in length, Cookoogong’s estimate of the size is rather wide of the mark; but this may have been due to misunderstanding on the part of Hill.
35
In 1824 an anonymous article, “Quelques observations nouvelles sur l’Ornithorhynque,” appeared in the Annales des Sciences Naturelles, (Vol. II). Quite possibly it was written by Geoffroy St.-Hilaire. On p. 75 the following words appear:—“On a vu à la Nouvelle-Hollande des oeufs d’Ornithorhynque, et c’est M. Jameson, l’un des principaux propriétaires et habitans de Botany-Bay, qui guarantit ce renseignement.” Jamison’s statement that the platypus lays eggs has here been converted into a definite assertion that the eggs have been seen.
In Vol. X of the same journal (1827) a second unsigned article appears:—“Sur les Habitudes de l’Ornithorhynque”—the account of the habits being quoted from the Antologia di Firenze, XXIV, p. 305, where also it appears anonymously. On p. 193 we read—“Ornithorhynchus inhabits the marshes of New Holland. It makes among tufts of reeds bordering the water a nest composed of down and interlaced roots, in which it deposits two white eggs, smaller than those of ordinary fowls: it broods on them for a long time, hatches them like a bird, and only abandons them when threatened by a formidable enemy.” The author of the article comments (pp. 194-5):—“We regret that the Antologia usually omits to make known the source whence it borrows the curious facts which it communicates to its readers. What we have just read bears a tolerably faithful resemblance to an account by Patrick Hill, ... but it differs in being more precise; which leads us to believe that it may be borrowed from another and a newer source.” It is quite obvious that the account was drawn from Hill, the greater precision being due to the journalistic sense of the writer.
However, these statements about eggs stirred Etienne Geoffroy to make enquiries, and the article closes with a quotation from a letter written by Southwood Smith to Milne Edwards in reply to Geoffroy:—“There are no eggs of Ornithorhynchus with the Linnean Society, nor in any other collection in London. One preparation had been seen, and was 36said to be furnished with an egg; Sir Everard Home had examined it, and was convinced that this was a mistake. This man of science added that no one had taken more trouble than himself to resolve all doubts on the subject of Ornithorhynchus, and that he had hopes of doing so, having interested the Secretary of State for the Colonies in this research.” This is a fairly literal translation of the French, as it appears in the Annales. The letter may have been written in English, in which case the re-translation of a translation would be certain to differ considerably from the original.
Lesson, writing in the Annales, Vol. VI (1825), says on p. 249:—
“Mr. Murdoch, superintendent of the farm at Emu Plains, assured me that he had found the eggs of Ornithorhynchus, and that they are of the size of those of a domestic fowl.”
In Vol. XVIII (1829) Etienne Geoffroy, flushed with triumph, produced a description and figure of veritable eggs. He writes on pp. 160-164:—
“But this fact, the more decisive as it is a fortunate confirmation of old stories, has just been sent to me by the learned Professor of Comparative Anatomy and Zoology in the free university lately founded in London, Mr. Robert E. Grant: this is, that Ornithorhynchus certainly lays eggs. I publish the full text of the letter which this savant has sent me; it is written from London, from the headquarters of the new university, under the date 14th September, 1829.
“Mr. Grant’s Letter.
“Sir,—
“My return to London has been delayed, as I stopped at Calais, Boulogne, Dover, and other places in England for the purpose of making zoological investigations on the coasts; but having now arrived here, I experience very great pleasure in communicating to you the information which I have just received, and which I have obtained from Mr. Leadbeater, concerning the eggs which he possesses, and which are said to be those of Ornithorhynchus.
37
“Mr. Holmes, well known to most naturalists in London as a gatherer of natural history collections, has lived for some years in New Holland. One day while hunting on the banks of the Hawkesbury, a river in the interior of the country, he saw very distinctly, some feet away, an Ornithorhynchus leave a sandbank and escape in the river. On examining the place where the animal had rested, Mr. Holmes saw a hollow in the sand about nine inches in diameter, and in this open cavity were some small twigs and the eggs in question.
“He found there four eggs:—all were brought to England, together with a collection of birds of the country. Mr. Holmes has since returned to New Holland, with his wife and family. Two of these eggs are in the Manchester Museum; the other two were given to Mr. Leadbeater, who will not part with them, neither for cash nor for any other consideration: he has refused to sell them to Sir Everard Home, to various other naturalists, and to myself.
“At the same time, despite the remarkable agreement of these facts with the results of your profound studies, you will only accord to this evidence such confidence as is due.
“These eggs seem to me to differ, in form and texture, from those of birds; they are remarkable for their regular spheroidal-oblong shape, of an equal width at both ends; they measure 1⅜ inches in length, by a breadth of ¾-inch; the shell is thin, fragile, semi-transparent, and of a uniform flat white colour; its external surface, under a lens, shows a wonderfully woven network; the calcareous matter forms the white walls of its innumerable and very minute cavities, which do not prevent the surface from remaining somewhat polished. One of the eggs was broken, and I examined its inner surface, which seemed also to be formed by the deposition of very small grains of calcareous matter.
“The size and shape of these eggs recall the eggs of lizards and snakes, reptiles which have not, however, a tenth of the bulk of an Ornithorhynchus.
“My friend Mr. Yarrell, who has also examined the eggs, 38thinks that they differ as much from those of birds as from those of reptiles. Other travellers have told me that the Hawkesbury River, on the banks of which these eggs were found, is reputed in that country to harbour great numbers of Ornithorhynchus.
“Mr. Holmes has been back in New Holland for nearly a year: and he will again be directing his attention to this matter.
“I regret being reduced to so few facts for communication to you on this subject of so great interest to science.
“I place at the bottom of my letter a drawing, made with the greatest care, of the entire egg which I saw at Mr. Leadbeater’s; it is shown natural size, and with the precise detail of its shape.
“Robert E. Grant.”
But, alas for the hopes of M. Geoffroy, this drawing, which he reproduces in Plate 3, is so faithfully done that an Australian zoologist has no difficulty in recognizing it as the egg of the common long-necked tortoise (Chelodina longicollis). Geoffroy was, of course, unaware of this. When, however, he came to consider the size of the egg, he realized that it could not possibly pass through the pelvic ring of the female platypus, the diameter of which was but five-twelfths of an inch, while that of the egg was nine-twelfths. Nothing daunted, Geoffroy first supposed that the egg passed to the cloaca in a not fully developed condition, and that it underwent further development there, but so rapidly as not to cause serious obstruction. Later, however, in 1833, he discarded this view, and believed that it remained in the oviduct until hatching took place, since it could not pass through the small pelvis. He would seem to have conveniently forgotten that the eggs were found laid in a nest in the sand.
Though, in support of oviparity, Geoffroy fell so often into error, he ultimately proved right. The great Richard Owen, on the other hand, through too firm and convinced a belief in ovi-viviparity, rejected evidence which, in the light of our later knowledge, was convincing enough; in the upshot he was proved wrong. The first piece of positive evidence was given by 39Lieutenant the Hon. Lauderdale Maule, of the 39th regiment, in a letter to Dr. Weatherhead, extracts from which were communicated to the Zoological Society of London and printed on pp. 145-6 of the volume for 1832. Maule writes:—
“During the spring of 1831, being detached in the interior of New South Wales, I was at some pains to discover the truths of the generally accepted belief, namely, that the female Platypus lays eggs and suckles its young.”
He goes on to say that he found and opened up a number of nesting-burrows, of which he gives a summary description. He obtained young which were forwarded to Dr. Weatherhead, and described by Owen; but “no eggs were found in a perfect state, but pieces of a substance resembling egg-shell were picked out of the debris of the nest” (p. 155). There is no reason to doubt that these were actually egg-shells, but Owen ignores this statement altogether, and Broderip (1840) writes in the Penny Cyclopaedia:—“The supposed portions of egg-shell found by Lieutenant Maule in the nest were probably portions of excrement, coated, as in birds, with the salts of the urine, that secretion and the faeces being expelled by the same orifice.”
It is probable that Owen based his views on the evidence of Dr. George Bennett, who spent many years in endeavouring to penetrate the secrets of the furtive and mysterious platypus. Bennett obtained impregnated females, and forwarded uteri containing ova to Owen, who described them in 1834. He also found young, but did not succeed in getting eggs, and became convinced that the young were brought forth alive. He tried to get precise information from the aborigines, but, again probably through lack of understanding, failed. We read on pp. 122-3 of his Gatherings of a Naturalist:—
“The various contradictory accounts that have been given, on the authority of the aborigines (who might be supposed, from their so often seeking these animals for food, to be able to state their habits correctly), as to the animal laying eggs and hatching them, induced me to take some pains to find out the cause of such an error, and being now perfectly satisfied as to the contents of the uteri, I could the more readily determine40 the accuracy or inaccuracy of the accounts which I might receive from the natives. I determined, however, not to ask any person who had been repeatedly questioned before on the same subject; but some time after, when I visited the out-stations in the Tumat country, where such questions had never been previously asked, I made inquiries among the most intelligent. The Yas natives at first asserted that the animals lay eggs, but very shortly afterwards contradicted themselves. To ascertain what dependence could be placed on them, I made a drawing of an oval egg, which was recognized to be like that of the Mallangong. I then made a drawing of a round egg, and that also was declared to be ‘cabango’ (egg) of the Mallangong. It was also declared that ‘old woman have eggs there in so many days’ (the number of which they did not know), that the young ones ‘tumble down,’ and that two eggs are laid in one day. An account subsequently obtained from a native, who appeared anxious to explain the fact, would lead to the belief that the animal is ovi-viviparous; but yet, from the difficulty they find in expressing themselves in our language, we often misunderstand them. He asserted the animal to be oviparous; but when desired to procure the egg, he replied, ‘Bel cabango (no egg) tumble down; pickaninny tumble down.’
“In the Tumat country the answers were readily and satisfactorily given; and afterwards, more minute questions being put to them through my interpreter, the result was the same. ‘Tambreet no make egg (corbuccor) tumble down; pickaninny make tumble down,’ was their reply. This accorded with my observations; for it was at the season when this inquiry was made that the young platypi were found in the burrow, as if just brought forth.”
Had Dr. Bennett relied more on individual effort instead of on aboriginal information, this vexed question could have been settled there and then.
The next account is that given by M. Jules Verreaux, of the Paris Museum, who spent some time in Tasmania; he wrote an article “Observations sur l’Ornithorhynque” in the Revue Zoologique for 1848. This article contains a curious admixture 41of fancy with some of which we shall have to deal later. The only point that interests us here is that on p. 130 Verreaux declares himself quite sure that the animal is ovi-viviparous, without giving any justification for his assertion. Owen (1848) reviews this article, and on p. 318 comments on Verreaux’s statement, which, apparently, he is quite prepared to accept, suggesting that it is based upon the same kind of evidence that has led to his own conclusions. He deplores the lack of absolute proof, but sets out in a footnote the reason for his belief: “It is to the absence of this proof that Dr. Carpenter appears to refer, where he remarks, in his excellent Principles of Human Physiology, 1842, p. 40, ‘No positive evidence has yet been obtained that its young are born alive.’ The minute size of the ovarian ovum and consequently of the vitellus; the presence of small ova with a delicate chorion and without chalazae or shell, in the uterine portion of the oviduct; the absence of any shell-forming portion of the oviduct—all are elements of a body of positive evidence in favour of the ovi-viviparity of the Ornithorhynchus, which needs only the discovery of the foetus in utero for decisive confirmation.”
In 1865 (1865, pp. 683-4) Owen published, with comment, two interesting letters from Australia, which are worth quoting in full:—
“Wood’s Point, September 21st, 1864.
“To Professor R. Owen.
“Sir,—I have great pleasure in being able to inform you of a very interesting discovery in the economy of the Ornithorhynchus paradoxus, and one which I have no doubt you will hail with delight. About ten months ago, a female Platypus was captured in the River Goulburn by some workman who gave it to the Gold-Receiver of this district. He, to prevent its escape, tied a cord to its leg and put it into a gin-case, where it remained during the night. The next morning, when he came to look at it, he found that it had laid two eggs. They were about the size of a crow’s egg, and were white, soft, and compressible, being without shell or anything approaching to a calcareous covering.
42
“I had an opportunity of examining them externally, and I found no evidence of their having had any recent vascular connexion with the maternal organs; but I am sorry to say that I never had a chance of examining their contents, as, on inquiring for them a day or two afterwards, I found they had been thrown away, much to my chagrin and disappointment.
“The animal itself was afterwards killed (next day), and I was told that numerous ova (in the words of my informant ‘eggs’) were found in it, in various stages of development, which in the aggregate somewhat resembled a bunch of grapes; but this I cannot personally vouch for.
“It may appear to you a matter of surprise that I did not examine more minutely this interesting animal; but I am sorry to say that the same spirit that dictated the throwing away of the eggs, prevented me making a more detailed investigation.
“I am in hopes that I shall be able to get another pregnant specimen; if so, I shall have much pleasure in sending it to you for your inspection.
“I have the honour to be, Sir,
“Your obedient Servant,
“Jno. Nicholson, M.D., etc.”
“Wood’s Point, Victoria, Australia.”
By a later mail Owen received from Dr. Mueller a letter from the “Gold-Receiver” referred to by Dr. Nicholson. It had been written in reply to enquiries, which vague reports of the occurrence had induced Dr. Mueller to make.
“Woods’ Point, September 25, 1864.
“Dear Sir,—In reply to your enquiries relative to the Ornithorhynchus paradoxus, I must in the first place correct an erroneous impression which the newspaper paragraph has conveyed.
“The Platypus is not now in my possession, and the eggs were laid the day after its capture. The animal was captured in the Goulburn and given to me. It was then fastened by a cord in a gin-case, and on examining it the next morning the 43two eggs were found in the bottom of the box, both of them having undoubtedly been laid during the night. In the course of the day the creature was killed by a would be scientific friend of mine, with the intention of preserving its skin; and on opening the body the ovaries were found to be clustered with ova in different stages of growth; but none of them so large as the eggs which were laid. These eggs were white, soft, and without shell, easily compressible, and about the size of a crow’s egg.
“Not being sufficiently versed in the subject I am not prepared to say whether these eggs might not have been abortions caused by fear, but there was no appearance on the surface of their ever having been vascularly connected with the maternal uterus, and reviewing all the facts observed I should undoubtedly say that the animal was oviparous.
“I am, dear Sir,
“Yours faithfully,
(Signed) “Geo. J. Rumby.”
Owen comments as follows upon these letters:—
“Assuming the fact of the oviposition in the month of December 1863 (Dr. Nicholson writes of the occurrence as having happened ‘about ten months’ before the date of his letter, September 21, 1864) by a female Ornithorhynchus, of two ova, about the size of a crow’s egg, ‘white, soft, compressible, without shell or anything approaching to a calcareous covering,’ the question is—What did they contain? Had the unvascular chorion been cut or torn open, an embryo or a yelk might have been seen. Better still would it have been if both ova had been at once immersed in a bottle of whatever colourless alcoholic liquor might be at hand. Probably no medical man had ever an opportunity or a chance of settling a point in comparative physiology of more interest, and with less trouble, than the gentleman who was privileged to be the first person to see and handle the new-laid eggs of the Ornithorhynchus paradoxus.”
44
I can sympathize with Owen in his disappointment. It would indeed be interesting to know what was inside those eggs. Probably young birds! However, without that knowledge it is not possible to decide whether they were the first eggs known to have been laid by a platypus. If the size mentioned by both Nicholson and Rumby is correct, they could not have been the eggs of Ornithorhynchus, which have but a third of the length and a tenth of the bulk of any Australian bird’s egg that could justifiably be referred to as that of a crow. The words “soft” and “compressible” in their descriptions are perplexing; if correct, then the size was wrong, and, if incorrect, the probability is that a practical joker introduced birds’ eggs.
At the outset Owen appears to have accepted this account at proving that Ornithorhynchus was oviparous; but later he inclined to the belief that the eggs might have been “abortions due to fear,” and was still enquiring for definite evidence.
In the Australian Journal of Education (vol. ii, 1869) there is a series of articles on the fauna of Australia, most of which are unsigned. That on the platypus appears on pages 104-5, under the pseudonym “Five Dock;” it contains nothing of any particular interest except a statement that the egg-laying idea is “exploded.”
On p. 16 of the Proceedings of the Royal Society of Tasmania (1878) we read:—
“Some members having expressed their opinion that the question as to the animal being oviparous had long been settled in the negative, Mr. E. D. Swan remarked that Professor McCoy, who would be recognized by all as an authority of the greatest weight, had recently written to the effect that he had received evidence of a most reliable character that the Ornithorhynchus was oviparous.”
But I cannot find that McCoy ever published this evidence.
In the same journal for 1879 Dr. Crowther makes some interesting observations on the platypus, and remarks on p. 96:—“when I tell you that the foetus has been found in utero, 45and in a hairless condition in the burrow ... (The former of these startling facts I hope to demonstrate to this society soon.)” It is hardly necessary to point out that this hope was never fulfilled.
The matter was not finally settled until 1884. In that year W. H. Caldwell, a Cambridge zoologist who came to Australia specially to investigate the reproduction of monotremes and marsupials, obtained eggs of both platypus and echidna in the Burnett River district of Queensland. He writes (1887, p. 464):—“In the second week of August I had similar stages in Ornithorhynchus,[2] but it was not until the third week that I got the laid eggs from the pouch of Echidna. In the following week (August 24) I shot an Ornithorhynchus whose first egg had been laid; her second egg was in a partially dilated os uteri. This egg, of similar appearance to, though slightly larger than, that of Echidna, was at a stage equal to a 36-hour chick. On the 29th I sent in the telegram ‘Monotremes oviparous, ovum meroblastic’ to a neighbouring station, where it would meet the passing mail-man, addressed to my friend Professor Liversidge, of the Sydney University, asking him to forward it to the British Association at Montreal.”
Caldwell’s cable was read before the Montreal meeting of the British Association for the Advancement of Science in September, 1884, and the scientific world at last had the solution of a question which had troubled it for more than eighty years. Monotremes laid eggs. By a curious coincidence, W. Haacke found on 25 August, 1884, an egg-shell in the pouch of an echidna, and exhibited it before the Royal Society of South Australia on 2 September, the very day on which Caldwell’s cable was read in Montreal. Haacke has made the claim that he first discovered the oviparous nature of the monotremes, but there can be no question that Caldwell has priority.
46
The general form of the platypus is by now familiar to all. It appears stuffed and mounted in all natural history museums, and illustrations of it adorn all ordinary natural history books. Yet, strange as it may seem, I have never seen a correctly mounted specimen in a museum, nor a correct figure of the whole animal in a book.
The most conspicuous feature about the bodily form of the platypus is its prevailing flatness. The naked muzzle is broad and flat; the head is so much depressed that the eyes have become more dorsal than lateral in position; the trunk is broad and flat, though in profile its outline rises from the shoulders to a point a little behind the middle of its length, and then falls towards the tail; a cross-section has the form of a low arch, a good deal more broad than high; finally, the tail is broad and flat, with a broadly rounded tip.
The skin, kimono-like, is over-large for the size of the body, and, except at all the animal’s extremities, it appears to be entirely dissociated from the primary nervous system. That is to say, a platypus will readily respond to the slightest touch on either feet, paws, bill, lips, or tail, but will not shrink from being soothingly stroked, from forehead to rump, along the fur. This characteristic is most apparent while the animal is submerged in water, where it is unable to see, hear, or smell the experimentalist.

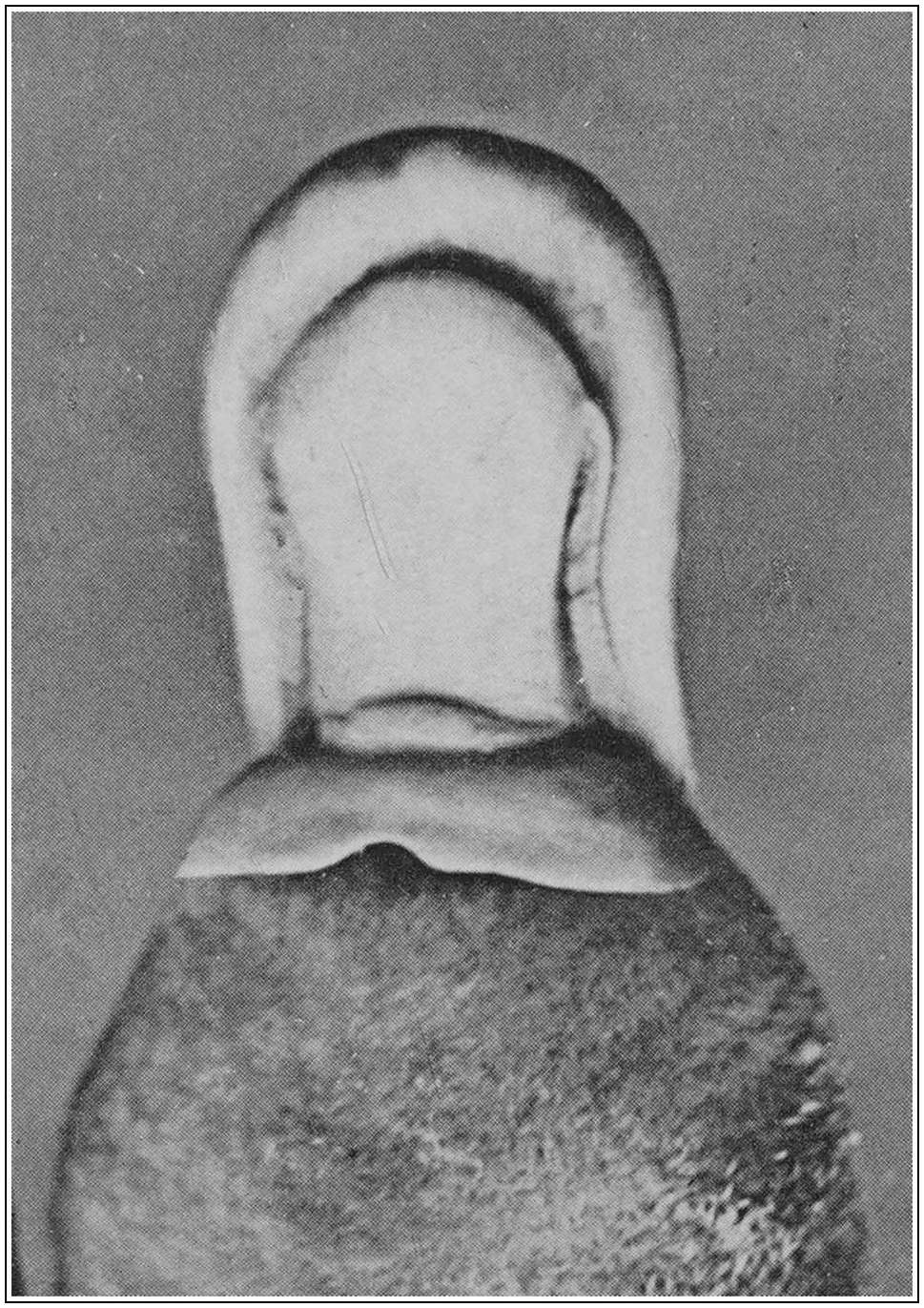
The muzzle with its backward-projecting flaps is naked, as also are the upper surfaces of the digits of paws and feet, and the palms and soles. All the rest of the body is covered with hair, which is of two main kinds. Closely investing all the 47body except the tail and extremities of the limbs is a fine dense under-fur, beyond which project the ends of a much longer and harsher kind of hair, with highly polished tips. The under-fur is woolly, grey at the base, inclining to brownish at the outer ends. The longer hairs are remarkable in that their projecting portions are flattened, giving them a lance-like appearance, and are generally curved so that the ends turn in to the fur. On the animal’s back and sides these hairs are usually dark brown in colour, but the tips are frequently silvery white, or, in reddish individuals, golden yellow. The prevailing paler colour of the under surface is due to the absence of dark pigment from the ends of these hairs, which have grey basal portions and tips ranging from silver-white through yellow to chestnut and (in some cases) deep ferruginous brown.
This longer fur determines the animal’s colour, which is variable within fairly narrow limits. The usual coloration is deep umber-brown on the back and sides of head and body and upon the exposed surfaces of the limbs; but reddish individuals are not uncommon, and some examples are almost black. The ends of the long hairs are frequently curly, and the prevailing ground-colour becomes powdered, as it were, with silver or gold, in consequence of the different colour of the tips.
The hair on the dorsal surface of the tail is coarse, and without flattened tips. Owing to the constant dragging of the under-side over the ground, this surface is usually denuded of hair, but in young individuals a delicate silky fur is present, usually of a golden colour, but ranging to warm brown.
The ventral surface of the body is described by Thomas (1888, p. 389) as ‘dirty greyish white’, which hardly does justice to the beauty of the fur on this part. It ranges from silver through yellow to warm chestnut, being lightest on the chest and throat, and darkening posteriorly. Old males sometimes have the whole ventral surface a warm brown in colour, with darker markings about the chin, limbs, and vent. The colour is, in all instances, due to the highly polished tips of the longer hairs, which have a brilliant sheen comparable with that of the 48breast-feathers of the tippet grebe (see Plate 7). An albino is recorded by Bennett, and one is exhibited in the Mammal Gallery of the Australian Museum, Sydney. Sometimes, too, the black skin of the extremities is mottled with white.
The coat of the platypus is devoid of stripes, bars, spots, or pattern of any description, save for a small yellowish-white to yellowish-brown spot placed just beneath the eye. Bennett (1860, p. 98) correctly described this spot, and noted that it occurred, with a single exception, in all specimens of either sex which had come under his observation. Thomas (1888, p. 389) dismisses it briefly as ‘a spot round each eye white or yellowish.’ Wood Jones (1923, p. 46) remarks—“There is said to be a ‘white or yellowish’ area around each eye; but this area, though almost always emphasized in text-book accounts of the animal, is not invariably present.”
To describe this spot as ‘around the eye’ is misleading. In some individuals the very slightly differentiated upper eyelashes certainly are light in colour, but it is only to this extent that the spot may be said to surround the eye. It is, as Bennett describes it, immediately below the inner angle. It is, moreover, present in all the platypus I have seen, including specimens from Queensland and Tasmania in the Australian Museum collections. That it is not invariably present is indicated by Bennett’s observation of an individual without it. Home (1802, p. 70) also records its absence in a male which he examined; he judged from its presence in the female that it was a character for the distinction of the sexes. This is not so, since it is commonly present in males. In dried skins, if the head be not fully filled out, it sometimes becomes involved in the facial furrow, and thus escapes notice; in spirit specimens, as well as in those wet with water, it is often curiously inconspicuous. Thus, out of ten photographs of the one animal illustrating the articles of Hornaday and Joseph (1922) which show the eye, only two (on pages 108 and 109) show the spot clearly. In one or two of the others it can just be made out, but in most it is not visible. Yet it is ordinarily a conspicuous 49mark, whether on dry land or under water, as may be judged from both subjects shown on Plate 6.
The hair of the platypus, as has been mentioned in the first chapter, has a general fore-and-aft direction, and, apart from the different kinds of hair described above, the only differentiation is to be found in the region of the facial furrow. Here the hair surrounding the eye is slightly modified to form eyelashes of a rudimentary nature, and the aural orifice is lined with short fine hairs, which help to prevent water and particles of soil from gaining admittance. There are no specialized sensory hairs in any part.
The muzzle, which shows some resemblance to a duck’s bill in the dry condition, is very different in the living animal. The naked skin is thick, but soft, moist, and flexible, very unlike the horny beak of a bird. On the upper surface it is slate-coloured; on the under, of a yellowish flesh-colour, often broadly mottled with greenish slate. From the base of each mandible a cuticular flap projects backwards over the fur of forehead and throat. The dorsal flap is longest in the middle line of the head, varying from 17 to 20 mm. in length, and is shorter at each side. The ventral flap is wider at its base than the dorsal, and has a shallow emargination in the middle of its posterior border. Although described separately as dorsal and ventral flaps, these really form round the base of the muzzle one continuous collar, which is only partially divided into dorsal and ventral portions by deep dorso-lateral emarginations. The nostrils open upon the upper mandible at about a third of its length from the anterior end. They are oval apertures with a slight rim-like border, set close together at each side of the middle line. Their anterior borders lie very close to each other, but they diverge obliquely behind. The dorsal portion of the muzzle measures, in an individual of average size, 60 mm. long by 50 mm. wide; it considerably overlaps the ventral portion, which in the same individual measures 42 mm. by 38 mm. (see Plate 8).
The muzzle, with its flaps, is a highly specialized sense-organ. 50The whole of its exposed surface, both above and below, is pitted with minute pores, which extend on to the cuticular flaps and mark the sites of the highly specialized touch-corpuscles. When under water, the animal depends principally on its delicate sense of touch for finding its way about, and it is fairly certain that the same condition obtains in the burrow. Even when in the open air, it probably depends largely upon the muzzle, since its eyes are so placed that it cannot easily see objects straight in front of it on the ground. The anterior border of the upper mandible is, from its position, the part most brought into use for testing objects, and would seem to be especially sensitive, for the platypus strongly resents its being touched, notwithstanding that it is used for shovelling away loose earth and for upturning river shingle. But the general distribution of touch-corpuscles over the whole of the naked area indicates that all parts are sensitive; and their continuation on to the flaps carries a sure implication that these, too, subserve a sensory function. Many dangers threaten an animal nosing about blindly under water; and it seems probable that the extension of the tactile area in this way is a provision against entering crevices and crannies in which it might be caught. It is well known that it can squeeze through very narrow spaces, and it is possible that the flaps are used as a gauge by means of which it can tell whether it is safe to go on, or wiser to withdraw. Whether this be so or not, the flaps are assuredly a part of the great tactile organ, and not a mechanical shield. In some of the earlier figures, drawn from dried skins, these flaps are shown standing up at right angles from the surface of the head; in the living animal they are always laid back upon the fur, nor does it seem able to raise and depress them to any appreciable extent.

The eye and ear are peculiarly arranged. In the half-grown young, running across the front of the head, just under the border of the dorsal flap, there is a groove which on each side turns backwards over the area occupied by the eye and ear (see 51Plate 12). In the adult the transverse portion of this groove has disappeared, but the lateral portions remain as the facial furrows, in which the eye and ear are situated. The eye, which has suffered most at the hands of artists and taxidermists, is placed at the anterior end of the groove, very close to the dorsal flap, by which it would be partly hidden were not the flap emarginated a little at each side. It is rather small, but exceedingly bright and beady, and has a dark brown iris almost indistinguishable from the pupil. Because of its position it can see only upwards and outwards, and its sole function would appear to be that of detecting moving objects when the beast is at the surface of the water or on the bank. In illustrations the eye is usually placed in the middle portion of the furrow, that is to say, between its true position and that of the ear. Even Gould’s plate exhibits this error. Moreover, the eye is generally depicted too large, and often with a light iris. The plates of Peron and Lesuer (1807) and of Lesson (1839) show a yellow iris, and Waterhouse (1846) in his black-and-white figure shows the iris white. A light iris has been given to many of the specimens mounted in museums, and in these, too, the eye is usually wrongly placed. In life the upper and lower eyelids are not well marked off from the borders of the furrow, but may be distinguished by a slight differentiation of the hair on their margins to form eyelashes. There is a nictitating membrane capable of covering the whole cornea, but I have never seen this employed by the living animal.
The portion of the furrow which contains the eye is partially separated from the auricular part by a fold of skin running obliquely upwards and backwards from the lower border. There is no auricle (external ear). The aural opening is in the form of a deep pit, occupying most of the posterior half of the furrow. The margin is lined inside with short, soft hairs, which prevent the entry of foreign bodies. The opening can be dilated and contracted at the will of the animal, and is in a constant state of motion when it is alarmed or excited. Under water, or while burrowing, the whole furrow is closed 52by the apposition of its upper and lower margins, and remains so until the head once more emerges. The dorsal position of the eyes, in conjunction with the bulging of the cheeks (due partly to the grit in the pouches) give the sides of the head a heavy-jowled appearance.
The head is joined to the trunk by means of a short and not very well-defined neck. The shape of the trunk varies according to the degree to which the animal is extended. When it is fully stretched in walking, the body appears oblong, and twice as long as it is wide. When squatting, the trunk appears both broader and higher in the posterior region.
The limbs are approximately equal in length, and are highly specialized. The metacarpal region of the fore-paw is covered with hair dorsally; the digits are naked, and are united by a black web, which is extended beyond the tips of the claws for a distance of 15 mm. (in spirit specimens), the extension being supported upon leathery thickenings which take origin from the under surface of the ungual phalanges. Each of the three middle digits has two of these thickenings, arranged symmetrically, but the two outer digits have only one each, placed towards the lateral margin of the paws. The claws are short, straight, and compressed. The palm is black and naked, finely wrinkled, but without any pads such as usually occur on mammalian feet. The fore-paw is the chief swimming-organ, its web being capable of considerable extension in the living creature. On land, that portion which extends beyond the finger-tips is folded under the palm, so that the stout claws may be used for walking and burrowing. The adaptation to two such totally diverse functions is very extraordinary. According to Wood Jones (1923, p. 48) the digital formula is 4 3 2 5 1; but the three middle digits are practically equal in length. In mounted specimens, and in illustrations, the web is always shown in the extended condition, but the animal when on land always has the web tucked under its palms. It is, moreover, never flat, as usually shown, but is always arched, with a dorsal convexity, even when extended.
53
A somewhat remarkable transformation of the fore-claws is essential before a young platypus can fend for itself. In the nestling stage, the slender claws curve downwards and are grooved beneath. The reason for this curvature is, apparently, to give the nestlings a firm grip of the mother’s abdominal fur when vigorously nuzzling to induce a free flow of milk (see Plate 10). As the nestling grows, the claws gradually become straighter and broader, and instead of remaining grooved, the nails become slightly convex below. By this time the paw is adequately webbed for swimming, and consequently the young one is free to forage alone (see Plate 9).
With each vigorous sweep of the paw when swimming, there is necessarily a severe pressure of the claws against the delicate webbing, and if the claws were not broad and regularly rounded off beneath they would undoubtedly puncture the swimming membrane. The same thing might happen during the toilet process. On examining living adult specimens I have found that, not only is the under surface of the claws convex, but the tips are slightly tilted and there is a considerable amount of resiliency in the nails. Yet, notwithstanding all this, distinct impressions of the tips may be clearly seen in the webbing (see Plate 9). It will thus be seen that although the platypus is a burrowing mammal, the nails are not designed entirely for that particular purpose. This supports my opinion that burrowing is normally attempted only in soil where the sensitivity-lipped bill can also be used.
The hind foot is less remarkable. On the upper side it is covered with hair almost to the ends of the digits, which are webbed. The webbing does not extend beyond the bases of the claws, except for a peculiar narrow prolongation on the inner margin, which lies just within the first digit, and looks like the shadow of its claw. The claws are laterally compressed, and strongly curved outwards and backwards. The digital formula, according to Wood Jones, is 4 5 3 2 1. This foot is scarcely used at all in swimming, except to help tread water, and to aid the tail in steering. On land, the main 54thrust is due to the feet, but the paws pull with a fair degree of strength. The sole, like the palm, is black, wrinkled, and naked, except for a fringe of hair along its inner border (see Plate 11). In the male a horn-coloured spur is set upon the heel, with a fleshy collar about its base; this spur is movable, and is directed backwards and inwards. In Bennett’s figure (1860, Pl. III), as well as in a number of others contained in authoritative zoological works, the spur is shown on the outside of the foot. This may have been due to a laudable desire to get the spur in at all costs, but it is apt to disconcert the seeker after precise knowledge. In the female, a depressed socket lined with wrinkled skin occupies a position corresponding to that of the spur in the male. The spur is developed in the young of both sexes; in the male it grows normally, but in the female it gradually disappears.
The tail is broad and flat, with the under surface usually a little concave. It is about a third longer than it is wide, with sides slightly converging as it passes backwards, and with broadly rounded end. The hair of its upper surface is exceedingly coarse and harsh; that of the under surface, when not entirely worn off, is softer and lighter in colour. It is a powerful and muscular organ, and an efficient aid in diving, as well as a somewhat less efficient rudder. In nursing females the hair is usually found to be worn off the upper surface. This is caused principally by pushing the pugs back into position in the burrow. Against this view, Wood Jones (1923, p. 48) writes:—
“On general lines, the form of the tail resembles that seen in the Beaver; and this form is, in both animals, an adaptation to surface swimming and rapid diving. It has nothing whatever to do (either in the Platypus or the Beaver) with any supposed habit of puddling clay or flattening down the floor of its burrow or nesting-chamber. When Beavers are alarmed they suddenly smack the water with their flat tails and dive beneath the surface. The loud sound made by the smack of the diving Beaver acts as a warning note to other Beavers in the neighbourhood, and it is probable that the same warning 55note is produced by the Platypus. The animal has several times been described as making a sound as it dives.”

Without supporting evidence, the categorical statement that the tail of the platypus has nothing whatever to do with any supposed habit of puddling clay is not convincing, and I prefer to rely on my own field observations. I must disagree also as to the warning signals. These are common enough among gregarious animals, and the beaver is a gregarious animal. The platypus is not gregarious, but almost solitary, and therefore has no one to warn. Wood Jones writes, it is true, of ‘a considerable colony,’ but I have had no experience of colonies of platypus. The rare occasions when I have found two in company have always caused me surprise. I see no justification, therefore, for arguing by analogy from the herd habits of the beaver to the semi-solitary behaviour of the platypus. When surprised, and beating a hasty retreat, the animal certainly makes a noise in diving; but this has no significance, as I shall explain later on.
The only sure external mark of distinction between the sexes is the spur. Full-grown males are considerably larger than full-grown females; but the immature or young male may be smaller than the female. Ten males taken by me during the spring over a period of several years measured (mm.):—600, 529, 535, 538, 481, 533, 485, 475, 540, 510, averaging 522 mm. Nine females measured (mm.):—406, 402, 414, 460, 475, 433, 451, 421, 407, averaging 430 mm.
Measurements of spirit specimens and skins in the Australian Museum are:—
| Male. | Female. | |
| Spirit Specimens. |
445 mm. 405 mm. 455 mm. |
398 mm. 392 mm. — |
| Skins |
561 mm. 560 mm. 510 mm. 537 mm. |
483 mm. 478 mm. — — |
| Average of 7 = 496 mm. | Average of 4 = 413 mm. |
56
Bennett (1860, p. 102) gives the average measurements of freshly-killed males as from 19 to 20 inches (483 mm. to 508 mm.); and of females as from 18 to 19 inches (457 mm. to 483 mm.).
We may next consider the nature and heat of the blood. In amphibia, reptiles, and birds, the red blood-corpuscles are oval, nucleated discs. In mammals they are non-nucleated, and in all save the camels are circular. The platypus has typical mammalian corpuscles, of about the same size as those of man. Cold-blooded animals (amphibia and reptiles) have no heat-regulating apparatus, and their temperature tends to rise and fall in accordance with that of the surrounding medium, whether it be earth, air, or water. In mammals and birds, on the other hand, the heat-regulating mechanism tends to keep the temperature constant within a degree or two, regardless of the external medium. This difference in blood-heat causes striking differences in habit. The cold-blooded animal is forced to hibernate during the winter, at least in places where there is a considerable difference between the mean winter and summer temperatures; the depression of temperature renders it inactive. The warm-blooded animal, on the other hand, eats more food, and moves about actively in order to produce the heat necessary to keep up its body temperature.
It has been claimed that the monotremes exhibit an intermediate condition, and are not completely warm-blooded. On this point, however, I am in full agreement with Wood Jones (1923, pp. 38-39), and quote his discussion of the matter:—
“In the same way we must admit that the body temperature of the Monotreme is unusually low; but that is not to agree that they are akin to the Reptiles in failing to maintain their body temperatures within certain fairly definite limits.
“The temperatures of seven females of the Platypus were taken by Mr. Burrell. These females were removed from their nesting chambers, and the observations were made in September. The temperature of these animals ranged between57 30°C. and 33°C., and the average of all the observations was 32.2°C.
“Of Echidna there are many temperature records, since it is an animal which is easily kept and examined in captivity. Miklouho-Maklay, in 1879, examined a series of ‘porcupines,’ and concluded that their temperature ranged between such low figures as from 26.95°C. and 28.3°C. In 1886, R. von Lendenfeld, as the result of a series of observations, concluded that the body temperature was higher, but more variable, and they give the extremes as 28°C. and 35°C. Semon, in 1894, gave 26.5°C. to 34°C. as the range for Echidna. Sutherland, in 1897, found it to be as wide as from 22°C. to 36°C. Dr. C. J. Martin re-investigated the question in 1902, and during the last few years Dr. Wardlaw has made innumerable observations, and, thanks to his work, we may say that the peculiarities of the body temperature of the Monotremes are now well known.
“The temperature of the higher Mammals varies within fairly wide limits, and in the different species that have been examined it ranges from about 36°C. upwards for a few degrees. It is therefore obvious that both Ornithorhynchus, with its average of 32.2°C., and Echidna, with its average of 31.1°C., are creatures of relatively low temperature; but the difference between the Monotreme temperature and that of other Mammals is not nearly so great as it is often asserted to be. It must also be remembered that our knowledge of the temperatures of some of the lower Monodelphia is scanty, and it is possible that Echidna and Ornithorhynchus do not stand very far apart from some of the more sluggish and least advanced of the higher Mammals in the matter of their bodily heat. Again, the fact that the temperature of the Monotremes varies at different times of the day does not confer on them the distinction that some writers have imagined. Dr. Wardlaw determined that the morning temperature was always lower than the afternoon temperature; but the same is true of the higher Mammals and of man. Semon noted this variation 58in the temperature, but confessed that ‘a relation between these changes and the changing temperature of the outer air could, however, not be proved.’ That is to say, although the temperature of Echidna is variable, its rise and fall does not correspond with the rise and fall of the temperature of its surroundings, as is the case with the lower Vertebrates. There is, nevertheless, a time when the temperature of Echidna does tend to follow the temperature of the outside air, and when the Monotreme can be said to behave somewhat like a ‘cold-blooded’ or poikilothermic animal. This is during that period of the year when the animal undergoes a partial hibernation or aestivation. In the summer Dr. Wardlaw’s animals averaged 30°C. in the morning and 32.6°C. in the afternoon; but in the winter the morning temperature was 29.7°C. and the afternoon was 32.3°C.; but this fall in winter temperature is found in all hibernating or partially hibernating animals; and it can only be said that the Monotremes are animals having a rather low body temperature, but, nevertheless, being truly homoeothermic (or ‘warm-blooded’) within certain limits of temperature, and in the non-hibernating period. They have a typically good mammalian heat-regulating mechanism, effective between 27.6°C. and 32.6°C.; and it is only when the temperature is raised or lowered beyond these limits that the regulation tends to break down, and they behave as poikilothermic animals.”


Following is a table of cloacal and atmospheric temperatures (Fahr.) observed by me during five years of field work.
59
| Male Platypus. | ||
|
Cloacal temp. |
Atmospheric shade temp. |
|
|
8/10/23 18/9/25 19/9/25 |
84° 82° 87° |
53° 63° 79° |
| Female Platypus. | ||
|
20/9/20 23/9/21 5/10/21 8/10/23 15/9/24 7/10/24 8/10/24 14/3/25* 24/8/25 24/8/25 28/8/25 3/9/25 10/9/25 18/9/25 26/9/25 |
88° 87° 88° 84° 84° 82° 90° 90° 86° 92° 94° 82° 89° 82° 86° |
70° 82° 68° 53° — — 46° — 64° 64° — 70° 74° 63° 66° |
| March may be reckoned as midway between rutting periods. | ||
| * With this exception, all the females were brooders taken from nests. | ||
Two temperature readings of wild echidna specimens, made directly after collection in mid-September 1924 and 1925, were found by me to be respectively:—
1. Cloaca, 90° Fahr. Pouch, containing young, 84° Fahr.
2. Cloaca, 79° Fahr. Pouch, containing young, 82° Fahr.
I did not record the atmospheric temperature when the 1924 specimen was taken; but I noted that it was an exceptionally cold day. When the second was taken, it was 70°.
By attaching an adult platypus to a spring scale I found that it could pull steadily (on level ground) six pounds, and up to twelve by jerking. The bottom jaw, when open, can pull seven pounds in the gradual closing.
Characteristics common to the platypus, to reptiles, and to birds are the single aperture or vent into which the intestinal, 60urinary, and reproductive systems open, and the laying of eggs. The “scientific” name (Ornithorhynchus) together with its commonly-accepted early vernacular name of “duckbill,” its webbed feet, and the discovery of its eggs have contributed to the existing impression, except among zoologists, that the platypus is essentially bird-like; but, as a matter of fact, while it is clearly a mammal, its remote origin has been proved to be reptilian. Apart from this scientific conclusion I would like to outline for general readers some external characteristics of the platypus which I consider to be possessed also by certain reptiles.
The study of an animal in its adult stage is apt to lead one to look for its relations in more modern forms, whereas in its embryo and nestling stage it is more likely to exhibit the characteristics of its forbears. By looking backward we may, therefore, occasionally detect a trait connecting the platypus with much earlier forms in the evolutionary cycle—a trait which in the mature animal would easily escape notice, even if it had not entirely disappeared.
To begin with the egg—quite apart from the leathery texture of the capsule, the fact that the foetus has developed to a considerable extent before deposition is positively reptilian in character.
The tortoise-like overturning of a platypus, though inconspicuous in the adult, is, nevertheless, deliberately and systematically accomplished by the unfurred nestling, and this at a stage when their limbs are of little assistance. Like a tortoise, a young platypus, when floundering on its back, pivots its snout firmly in the ground, then arches its neck, throat uppermost, until, by such strenuous leverage, the shoulders are sufficiently raised to allow the wriggler to over-balance on to its abdomen.
Another reptilian trait peculiar to the young of platypus is their power of endurance when wholly submerged in water, and, strange though it may appear, the younger the subject the greater that power. This is described more fully in the chapter on the breeding-burrow.
61
The male platypus and echidna are the only known mammals with poison glands, and this forms another link with the reptiles.
From a dorsal aspect, the fur-tracts of Ornithorhynchus trend uniformly towards the tail, in a manner similar to that of the scale pattern conspicuous in snakes, etc. It is true that slight semi-whorls or waves of fur occur ventrally, and about the cheek-pouches, but even this is not entirely wanting in the scale pattern of reptiles.
Spurs, or “hooks” as I prefer to call them here, are to be found on the fore-flippers of the male Green Turtle; but whether they are used as weapons of defence I cannot say. My opinion is that, like those of a platypus, defence is one of their purposes. From personal observation of both creatures, however, I know that their hooks are used for gripping during copulation. Incidentally it may be remarked that both of these creatures perform this function in water. It is of prolonged duration in both cases; therefore an untiring grip is essential.
The casual gait of a platypus is somewhat akin to that of a Stump-tailed Lizard, while the chubby tails which both animals possess are sufficiently near in design not to be overlooked in this matter.
The small and beady bright eyes of Ornithorhynchus, also their position and the direction of their vision, are similar to those of certain reptiles, as is also the dorsal position of the rigid nostrils.
Although represented in swimming birds, the webbing of the feet of the platypus is found also in reptilian quadrupeds.
The contorting ability of an adult platypus is quite remarkable. It can extend its length six inches without raising its abdomen from the ground. When it is in a crouching attitude, with the back conspicuously humped, it is impossible to see daylight below, and the contour of the whole body resembles that of a squatting echidna. When lying fully extended on its back, the platypus can, by placing the lower portion of its bill on its breast, and without raising its head to 62any appreciable extent, double itself ventrally until its head passes its tail, and that pliable member is itself doubled until the creature becomes normally righted on all fours, dragging its tail behind it. This act it can accomplish in a tunnel equal in circumference to the performer so doubled. When balled-up, as in the sleeping position, the tip of its tail will reach over the head to the scapular region. In the reversed position the tail-tip reaches to the gullet, above the back. In turning sideways, with abdomen on the ground the while, tail and bill just make contact. The head can be turned sideways at a right angle to the body, and the tail can be moved either to form a right angle with the rump or to lie comfortably along the contour of the back, except at the base, where one can just insert the tip of an index finger. These few items are additional to the animal’s capacity for muscular expansion and oscillating limb-functions, and they give some notion of what the platypus is capable of when hard pressed or in a tight corner.
Finally, the platypus, like the reptiles, has no external ears and is flealess, though ticks are common to both. One is known by the company one keeps.


63
A primitive kind of mammal, which is in some respects intermediate between the higher mammals and the reptiles, might be expected to show a low grade of nervous organization and limited powers of sensory perception, as well as something of the characteristic reptilian cold-bloodedness. Strangely enough, these characters are not found in the platypus.
The brain is surprisingly large—much larger in proportion to the body-weight than that of any reptile. It is, moreover, definitely mammalian in its structure, except that it lacks the definite band of fibres, connecting one hemisphere of the brain with the other, which is known as the corpus callosum. This deficiency is not, however, confined to monotremes, but also characterizes marsupials. Wood Jones (1923, p. 32) writes:—
“The corpus callosum is a cerebral commissure, or a nerve fibre bundle, which keeps the higher centres of the right and left central hemispheres in communication across the middle line of the brain. It cannot be said that the living monotremes are deficient in the extent of their cerebral hemispheres—they are indeed mysteriously well endowed with cerebral cortex. But it may be asserted—borrowing an expression from the electrician—that their brains are underwired.”
In the size and structure of its brain, then, the platypus proves to be an animal with a considerable degree of intelligence, with a cerebrum better organized than that of the lower marsupials and even of some of the lower Monodelphia. A well-organized brain and a large surface of cerebral cortex 64indicate a degree of intelligence far removed from that of the reptiles.
The psychology and sensory perceptions of animals other than ourselves present a very difficult field for investigation. Philosophers have never tired of telling us that the world about us is, though not exactly a figment of our imaginations, yet an illusion due to the limitations of our sensory perceptions. The physicist further confuses our simple minds by supporting the philosopher with a statement that the trees and grass and bricks and mortar which we imagine we see are mere buzzings of electrons. We have, it is true, a proper scorn for these attempts to befog us; yet, when we come to inquire into the mental attributes or sense-reactions of any animal other than ourselves, we are brought up sharply by an uneasy suspicion that the philosopher may be right. Fabre, describing the behaviour of a wasp, may record all its visible actions with scrupulous accuracy; when he comes to interpretation of them, he can interpret only in terms of Henri Fabre. We cannot project our minds into the mind of any other animal; nor can we be sure that any other animal sees, smells, tastes, hears, or feels just what we see, smell, taste, hear, and feel. In fact, we know that there are differences, that our eyesight is much inferior to that of the vultures, our sense of smell to that of predatory carnivorous mammals in general, and so on. There is also plenty of evidence that other animals possess sensory perceptions denied to us. This being a matter which seriously affects human prestige, we hasten to reassure ourselves by means of a dogmatic statement that we reason, while the rest of the animal kingdom, unable to indulge in this majestic process, is at the mercy of tropisms and instincts.
This digression is merely a warning that what we conclude about the mentality and sensory processes of the platypus must be derived chiefly from what we know of these processes in ourselves. We say ‘chiefly,’ because we have, after all, an evolutionary history of the development of brains and sense-organs, and a surety (since our own have been derived through 65those of the lower Mammalia) that the two must have a great deal in common. Bearing in mind the limitations of the method, we may proceed to an examination, first of the sensory perceptions, and later of the intelligence, of the platypus, so far as we know anything about them.
The eye of the platypus is small; on this account it has generally been assumed that the animal’s vision is poor. The earlier observers, obsessed by the small size of the organ and the burrowing habits of its proprietor, were led to compare its powers of vision with those of the mole. As a matter of fact, though small, the eye is extraordinarily bright, and is during life not deep sunk (as is commonly reported from examinations of spirit specimens) but fairly prominent. The eyes are certainly placed high towards the dorsal surface of the head; but, since they are used only for sub-aerial vision, this position would seem to be a direct adaptation to the particular purpose. Wood Jones has certainly written, and that quite recently (1923, p. 52):—“Although the eye of the living animal is said to be brilliant, it is small and deep-set, and so situated that its range of vision must be very limited. In any case, one would not expect the sense of vision to be remarkably acute in an animal which spends so much of its time in a dark burrow, or at the bottom of the water.” I know that the animal does not use its eyes under water—an observation which was not available to Wood Jones—and I believe, though I have no proof of this, that they are not kept open in the burrow. Their whole purpose is that of sub-aerial vision, and they are so situated as to have the widest possible range of vision when the animal is floating at the surface with just the top of the flat head exposed.
Bennett (1860, p. 135) observes of his captive animals:—“When running, they were exceedingly animated, their little eyes glistened, and the orifices of their ears contracted and dilated with rapidity.... Their eyes being placed so high on the head, they do not see objects well in a straight line, and consequently run against everything in the room during their 66perambulations, spreading confusion among all the light and easily overturnable articles. I have occasionally seen them elevate the head, as if to regard objects above or around them.”
When the animal is swimming in the water, it often raises its head, so that the short neck is vertical to, and the plane of the head horizontal with, the surface. Anyone who has ever attempted to shoot a platypus is soon made to realize the sharpness of its vision. Bennett, Semon, and many others have shown how useless it is to move towards the animal when it is at the surface. The slightest movement is enough to send it down; and, when it dives in alarm, it rarely reappears during the same feeding period. It will also dive at the flash of a gun.
My own observations go to show that, while the vision is acute in diffused daylight (e.g., at the usual crepuscular feeding-times), bright sunlight seems to affect it very considerably. When the sun is shining, I find that there is no need to take precautions against abrupt movements, as these do not seem to be observed. A gun may be raised and pointed at the animal without the slightest notice being taken. I have on many occasions released, during the daytime, platypus collected for purposes of photography, and noted that, while the animal dashes hurriedly for the water, when once there it displays no further alarm. Waving the arms does not frighten it. The softest clap of the hands, however, is sufficient to cause it to dive instantly. I therefore believe that the auditory sense is much more acute than the visual, at least during bright sunlight. This observation may account for the apparent tameness of the animals observed by Geoffrey Smith.

I cannot say what kind of image is formed upon its retina; but we are not entitled to assume that its power of sight is limited to the detection of objects in movement, though that would appear to be the chief purpose subserved. My own experience tells me that moving objects are more easily perceived than stationary ones; and anyone who has had successful experience of approaching or taming wild animals well knows that the thing to be avoided at all costs is abrupt movement. 67The capacity for detecting this kind of movement is as fully developed in the platypus as in any other mammal, at least over the short range which is necessary for the animal’s safety and well-being. Apart from this, we have no criteria for comparing its powers of sight with those of other animals, and certainly no justification, apart from the small size of the eye, for assuming that these are limited.
The auditory sense is undoubtedly acute. The creature dives on hearing an unaccustomed noise just as rapidly as on seeing an unaccustomed movement. Bennett, in the quotation given above, remarks upon the dilatation and contraction of the aural orifice, which, in spite of the absence of a definite auricle, remind one irresistibly of the nervous twitchings and twistings of the ear made by a spirited and fidgety horse. No observer of the living animal could have any doubt that it depends very largely upon auditory sensations. Semon (1894, p. 11) writes:—“Every doubtful noise causes it to disappear. I saw one dive immediately on the discharge of a gun a mile away. It appeared again rather soon, which decidedly it would not have done had it been alarmed by a sound at close quarters.” In this instance, however, I think the diving and distant report were merely a coincidence. As regards Bennett’s statement concerning the twitching of the facial furrow at the region of the ear-hole, I find that the platypus is actually capable at will, of “cocking” it to act as a temporary auricle to pick up sounds (see Plate 6, fig. 2).
The olfactory organs are more definitely reptilian in character than the rest of the sense-organs. To determine the measure of this sense, a careful experimental enquiry would be necessary, and the platypus has not shown any inclination to become a tractable laboratory animal. The large size of the olfactory nerves, however, and the development of scent-glands, are sufficient proof that the platypus has the sense of smell.
The optic nerves are small; the olfactory relatively large, though not so enormously developed as in the echidna. Those branches of the trigeminal nerve which are distributed to the 68muzzle are, however, relatively enormous, a mass of nerve-fibres passing out through the infra-orbital foramen above, and the inferior dental and mental foramina below, and dividing up to go to every part of the sensitive naked skin. These nerves end in special tactile corpuscles, the sites of which are marked by innumerable pores (or pits) thickly scattered over the naked integument. The whole of this area forms, as has been indicated before, a remarkably sensitive tactile apparatus without parallel among the Mammalia.
The most sensitive portion of this remarkable muzzle is undoubtedly the anterior border of the upper lip. As Bennett has remarked, the animal exhibits signs of acute discomfort when this lip is touched or pressed, and struggles violently to withdraw. At the date of this statement Bennett was evidently unaware of the fact that the sensitive lip of a platypus is used vigorously as a shovel when burrowing. Of course, this portion of the muzzle would be the first to come into contact with those animals which serve as food. Verreaux (1848, p. 129) states that in burrowing the beak is first used to dig the soil.
The extension of the naked integument into dorsal and ventral flaps is very remarkable, and there has been much surmise as to the function of these. Bennett first suggested (1860, p. 100):—
“In the base of both the lower and upper mandibles is a transverse loose fold or flap of the integument, always similar in colour to the skin covering the mandibles, that is to say, of a dull dirty greyish-black in the upper, and white or mottled in the lower. In the upper mandible this is continued very nearly to the eyes, and may perhaps afford some protection to those organs when the animal is engaged in burrowing, or seeking its food in the mud. The upper fold or flap is continuous with another portion arising from the lower mandible, also at its base. Some consider the use of these folds to be to prevent the beak from being pushed into the soft mud beyond this part, which is so broad as completely to stop its further progress. From careful observation of the actions of living specimens, I can assign no other use to this part than that which I have just mentioned.”
69
Oldfield Thomas (1888, p. 388) states:—
“Beak smooth, evenly rounded, its junction with the head marked, both above and below, by a projecting leathery flap, evidently developed to save the face from injury when the head is plunged in mud or gravel.”
Since the platypus has been observed to bury itself up to half its body-length in mud and gravel, it becomes obvious that the purpose of these flaps cannot be that of preventing it from burying more than its ‘beak.’ If the function of the flaps be to protect the eyes, it is a remarkable fact that the dorsal flap is emarginated at each side just in front of the eye, where the protection would be most needed. Moreover, the ventral flap could take no part in such protection, and the eyes and ear are already quite sufficiently protected by the apposable lids of the facial furrow. I am thus forced to the conclusion that the flaps subserve a function wholly tactile, a conclusion supported by the fact that their anterior faces are studded with touch-corpuscles. In the platypus, therefore, these flaps may be said to take the place, and serve the purpose, of the facial vibrissae of higher mammals (see Plate 12, fig. 1).
Passing to the sense of taste, we again find ourselves without means for determining the precise degree of gustatory perception possessed by the platypus. Poulton (1883) has investigated the structure of the tongue. The anterior portion, which is free from the floor of the mouth for only a third of its length, and is therefore capable of only very limited movement, is covered with rough papillae directed backwards, between the bases of which lie many mucous glands. This part of the tongue contains only tactile terminal organs, somewhat like the Pacinian corpuscles of ducks, and is concerned with the passing of food back to the grinders. The taste-areas occur upon the posterior, swollen portion of the tongue, and are four in number. The anterior pair are situated in deep antero-lateral grooves, the posterior in much shallower postero-lateral depressions. All four grooves are crammed with taste-buds, approximately of the same type as those occurring in mammals in 70general. The platypus is a dainty feeder, and it would seem that its taste-sense is an important feature in its general economy. It prefers its food alive, although in captivity hunger will induce it to accept dead food. This must, however, be fresh.
In addition to the five commonly-accepted senses, which are provided for by special sense-organs, I have referred elsewhere to the remarkable development of a sense of direction which the platypus displays in locating the nearest water, in detecting obstacles beneath the earth (thus inducing it to alter the course of its burrow), and in procuring its food. The first of these powers is the common possession of many animals, but is not, so far as I am aware, understood or accounted for in any of them. The second and third are quite as remarkable. Even in man there are still traces of a sense of perception of something solid in the immediate neighbourhood. This so-called “sixth sense” is probably due to the intense development of some extremely sensitive organ not yet accurately defined. The ability to “sense” the proximity of certain objects may be exemplified in the bat, which flits unharmed about the darkest caverns, aided probably by highly-sensitized skin-folds situated on its snout, and in certain fishes, in which the well-known lateral-line organs help to serve a similar purpose.
My observations of the platypus under water support the view that, of the five senses ordinarily possessed by animals, the only one operative while the platypus is gathering its food at the bottom of a river or water-hole is that of touch. My opinion is that this animal must have developed some extraordinary means of finding its prey, apart from the sense of touch, and that the sensory apparatus through which this acts is connected in some way with the fleshy nature of the bill. If this “sixth sense” is not responsible, then we must fall back upon that makeshift word “instinct,” which is referred to by Hornaday (1922, p. 9) when he says “Instinct often functions as a sixth sense.”
In a recent controversy concerning its use of the senses of 71smell and touch while searching for food at the bottom of a river, the question arose as to whether it relied on either of these exclusively, or on both. No attempt had previously been made to solve the problem, so I there and then decided to investigate systematically, and had a glass tank constructed, with an observation chamber (see Plate 13). I was well aware at the outset that the intended captive would be nervous and excited, but concluded that such unavoidable conditions might be to some extent counterbalanced by liberating it suddenly into fifty gallons of cool, fresh water. At least, I imagined that a sudden stimulant of that nature would somewhat, if only temporarily, lessen the shock, since my presence would be unsuspected.
When all was in readiness for the try-out, I obtained an uninjured adult female from a burrow near by, and, so as to give her ample experience of the smell of human hands (to say nothing of the sight and feel of them), I handled her repeatedly and deliberately, occasionally rubbing my hands, back and front, over her sniffing nostrils, besides frequently fingering her sensitive lips. I reasoned that, if her sense of smell was only half as efficient below water as on top, she might be expected to shy clear of the submerged hands that had just previously overhauled her. But such was not the case; in fact, quite the reverse occurred, and to the marked astonishment of several onlookers she instantly sounded, and, after probing her way along the sandy bottom of the test-tank, reclined leisurely beneath the unscrupulous hands which, only a few minutes earlier, were beyond doubt the dread of her precious life. Several times she swam to the surface for a timely blow and repeated the first performance, notwithstanding that I had deliberately interfered with her several times below, with the specific object of forcing her into action (see Plate 13, fig. 3).
While she was practically stationary on the sand beneath my hands, I placed my finger directly above, and within an eighth of an inch of her nostrils. But obviously she was quite unaware of my presence or behaviour until my finger came in contact with the tip of her bill. “Cat-like,” then, she arched 72her back, raised her tail, turned a kind of twisting somersault, and swam away (see Plate 13, fig. 1). Again she returned and casually nuzzled my hands as though nothing unusual had happened. Possibly the tap on the snout did not seem to her to be any more than the sudden flip of a startled shrimp. Nevertheless, I am quite convinced that she did not return solely to satisfy her curiosity, or purposely for shelter, as she had already taken advantage of that elsewhere in the tank.
Judging from all that occurred, I ultimately came to the conclusion that, if the creature had any sense of smell at all beneath the water, she certainly did not display it during my investigations. The supposed abnormal sense of touch was also not nearly so keen as I had surmised, at any rate in the detection of immediate danger (at least of an unfamiliar nature), while she was wholly submerged. Still, the bill may be extremely sensitive and quite indispensable while sifting mud and selecting suitable foodstuffs below. In that case (which I think may be assumed) it is probably safe to assert that an adult platypus has no enemies of consequence to fear beneath the water. Otherwise the docile Ornithorhynchus would surely not have fared so well during the many evolutionary phases since prehistoric times.
What is it, then, that is responsible for the prolonged existence of this primeval creature? Is it a sixth sense, or simply because such creatures fare well—as they appear to do—on so-called mud? As regards the five ordinary senses of a platypus when on land, or afloat, I have nothing to add except direct confirmation, if necessary, of their existence; but I suggest that, beneath the water, where mud is possibly consumed regardless of taste, the sense of taste is without function, and that it remains so until the animal swims to the surface and leisurely masticates the food taken while below, at least in the case of large prey.
Of the senses of hearing and seeing, so far as I could observe, both were rendered inoperative, while the creature remained submerged, principally by means of a water-tight 73facial furrow which simultaneously envelops the orifice of both ear and eye directly the animal decides to sound. I endeavoured to attract its attention by shouting at the top of my voice, while it groped about my submerged hands and around the glass helmet enveloping my head, without demonstrating the slightest symptoms of agitation. Testing the sense of sight proved rather perplexing. However, I came to the conclusion that, strictly speaking, the animal could not then see, although, whenever it came to a part of the tank where the light was at all strong, it immediately turned and sought the most shaded parts.
As a guide to those interested in this subject, let me state that it is practically impossible, owing to its timid nature, to test accurately the sense of smell of a freshly-captured platypus beneath water by introducing natural foods. Hence the omission of such an attempt in this instance. Of course, a test could be made, and no doubt successfully, with a contented and undisturbed captive, but I have intentionally left that opening to other enthusiasts perchance intent on checking my slender theoretical views.
In a previous test case[3] I proved beyond doubt that a platypus in captivity will devour in assorted foodstuffs (not including mud) half its own weight nightly. Now, if similar quantities are consumed when the creature is at large, then, as a platypus providore, it is beyond my imagination to comprehend how it obtains such an enormous supply, unless mud be included in the bill of fare as a kind of “fill-up” necessity, if not as a staple food.
Of the many platypus that I have had occasion to dissect, the intestines and stomachs of most, especially the robust and healthy-looking specimens, contained much mud, and apparently little else, while the cheek-pouches of all bulged, crammed with grit, mud, and crunched aquatic creatures, seemingly of minute forms.
74
On one occasion, in the presence of the late Charles M. Hoy, who was collecting in Australia for the Smithsonian Institution, I extracted a small shrimp (entire) from the stomach of a foraging female platypus, captured at noon. Possibly, the crustacean slipped down accidentally. Nevertheless, that query does not upset my theory that platypus are indiscriminate mud-suckers, but it tends rather to strengthen my contention that the sense of taste is apt to cease functioning beneath water.
Live shrimps collected directly from the feeding-grounds of platypus are readily devoured by Ornithorhynchus in captivity. In fact most specimens prefer them to indigenous river-bank worms or other varieties of food supplied to them. From personal observation, I can vouch for one male platypus in captivity that lived entirely on dead raw prawns (salt-water variety) for eight months, and was then, apparently, in splendid condition. Furthermore, it is said to have lived four months longer, when unfortunately for the experimenters, it escaped from its neglected enclosure. I have no hesitation in saying that platypus are occasional mud-suckers, and that at least some part of their living food is both discovered and secured by that sucking process.
While under water, the sense of touch is apparently the only one of the five ordinary senses functioning fully, and, in the procuring of food, even that sense appears to be less effective than the animal’s mysterious ability to track and secure living prey.
Again, as mud is seemingly a necessity, if only to appease an enormous appetite, why has the platypus such an abnormal nervous system of touch? Surely the presence of mud could be readily determined by a puddling platypus, without such special equipment as super-sensitized abnormally-formed lips, etc.
If a platypus must actually touch before being aware of the presence of shrimps or other active prey that it so relishes and thrives upon in captivity, then, to my mind, such wary creatures would be rarely taken alive when at large. On the other hand, if a sixth sense functions to assure direction, then it would be 75quite possible for a platypus to collect half of its own weight in live animal foodstuffs nightly, but not otherwise.
The ease with which the platypus can be killed indicates that there is something delicate in its organization. The ordinary method in shooting is to fire a heavy rifle-bullet beneath the animal as it floats at the surface of the water, and the concussion is usually sufficient to cause its death.
What I have written is scanty enough; but I think it shows, despite our lack of more precise knowledge, that the platypus is an animal possessed of acute sensory perceptions, a delicate nervous organization, an active metabolism, and a degree of cunning which must be based upon considerable intelligence and which is far from justifying Professor W. K. Parker’s epithet of ‘frog-witted duckbill.’
A final point of distinction is that the platypus possesses a voice. Vocal powers are somewhat curiously distributed among vertebrate animals. Frogs are notorious for vocal ability, and the Australian species can hold their own with those of other parts of the world both in the variety and in the volume of their choruses. Reptiles, however, are practically voiceless. Snakes and some lizards hiss, and some geckoes can scream when disturbed or handled, but the vast majority are silent under all circumstances. The songs of birds have called forth many tributes. The lower orders of mammals are not famously vocal. The echidna, so far as I am aware, has no voice at all, the only sound it makes being a sniff through its nostrils. Marsupials are in general silent animals, the cough of the wallaby and staccato snort of the bandicoot affording examples of the sounds they do make. Phalangers can scream, but do so only exceptionally. The lower Monodelphia, too, have remarkably little in the way of voice. That the platypus should possess a voice was unexpected, and certainly few have recorded it. The furred young, when disturbed, keep up a continuous growling noise, which it is usual to compare to that of a growling puppy. The adults make a noise which can best be imitated by a tremulous snoring.
76
In both platypus and echidna there occurs on the inner side of each hind limb of the male a movable spur. This is found in the young female also; but in the full-grown female it has disappeared, leaving a depression to mark the spot where it had been. This condition suggests that the spur and depression are of the nature of secondary sexual characters, and have something to do with reproduction. Such was the general belief for twenty years after the first discovery of the platypus. Then it was reported from Australia that the spur of the male was capable of causing serious wounds, followed by dangerous symptoms due to a venom expressed through the spur. Thus began a lengthy controversy which has not yet been settled to the satisfaction of all.
Shaw (1799) described the spur as a sixth digit, but pointed out that it resembled a strong, sharp spur. Home (1801, p. 72) made the first surmise as to its function, and described it in the following terms:—
“In the male, just at the setting on of the heel, there is a strong crooked spur, half an inch long, with a sharp point, which has a joint between it and the foot, and is capable of motion in two directions. When the point of it is brought close to the leg, the spur is almost completely concealed among the hair; when directed outwards, it projects considerably, and is very conspicuous. It is probably by means of these spurs, or hooks, that the female is kept from withdrawing herself in the act of copulation, since they are very conveniently placed for laying hold of her body on that particular occasion. The female has no spur of this kind.”[4]
77
In March 1817 extracts from a letter from Sir John Jamison, dated at Regentville, New South Wales, September 10, 1816, were read before the Linnean Society of London and published in the Transactions for 1818 (pp. 584-5). Jamison writes:—
“I cannot avoid relating to you an extraordinary peculiarity which I have lately discovered in the Ornithorhynchus paradoxus. The male of this wonderful animal is provided with spurs on the hind feet or legs, like a cock. The spur is situated over a cyst of venomous fluid, and has a tube or cannula up its centre, through which the animal can, like a serpent, force the poison when it inflicts its wound. I wounded one with small shot; and on my overseer’s taking it out of the water, it stuck its spurs into the palm and back of his right hand with such force, and retained them in with such strength, that they could not be withdrawn until it was killed.[5] The hand instantly swelled to a prodigious bulk; and the inflammation having rapidly extended to his shoulder, he was in a few minutes threatened with locked-jaw, and exhibited all the symptoms of a person bitten by a venomous snake. The pain from the first was insupportable, and cold sweats and sickness of the stomach took place so alarmingly, that I found it necessary, besides the external application of oil and vinegar, to administer large quantities of the volatile alkali with opium, which I really think preserved his life. He was obliged to keep his bed for several days, and did not recover the perfect use of his hand for nine weeks. This unexpected and extraordinary occurrence induced me to examine the spur of the animal; and on pressing it down on the leg the fluid squirted through the tube: but for what purpose Nature has so armed these animals is as yet unknown to me.”
Blainville, also in 1817, described the canal in the spur and the cyst at the base; as the specimen he examined was a skin only, he did not find the gland. In the Edinburgh Philosophical Journal (vol. vi, 1822, p. 184) it is mentioned that Dr. Traill confirmed the existence of a canal through the spur, so 78small that he could not pass a horse-hair through it, though a human hair passed.
In the same year an extract of a letter from Mr. Patrick Hill, Surgeon in the Royal Navy, dated Sydney, 3rd January, 1821, and addressed to the Secretary of the Linnean Society of London, was read before the Society and printed in the Transactions for the year, pp. 622-4. Hill writes:—
“You will be gratified to learn, that I have been completely successful in establishing our friend Sir John Jamison’s account of the spur of the Ornithorhynchus paradoxus. I subjoin an extract from my notes:
“Sunday, Oct. 1, 1820—On the banks of Campbell’s River. In the morning shot a male Ornithorhynchus. On examination, soon after it was killed, I observed near the extremity of the convex side of the spur, a minute spot, like the orifice of a tube; and on endeavouring to pass a bristle from this spot, three successive drops of a limpid clear fluid issued from it. I then examined the other spur with the same result. On dissecting the foot of the animal, I found at the inner side of the root of the spur, immediately over the articulation, a small cyst, which I cut into; it did not at that time contain any fluid; but from it I, with great ease, passed a horse-hair through the spur.[6] This preparation I have sent to you, together with the dried cyst.”
Farther on he quotes Cookoogong, chief of the Boorah-Boorah tribe, as being “perfectly aware that a wound from the spur of the male is followed by swelling and great pain; but although he has seen many cases of it, he has never known it fatal.”
Knox (1823, p. 38) describes and gives the first published figure of the gland, duct, cyst, and spur in situ in a dissection of the leg. He opposes Home’s views as to the copulatory function, and is of opinion that the spur must be a weapon of offence. In the same year Home records the presence of the gland, and describes how Clift passed mercury through the 79duct—an experiment which Knox also performed. The specimen examined by Knox has a certain historic interest, it having been sent to the Royal Museum of the University of Edinburgh by the then Governor of New South Wales, Sir Thomas Brisbane.
In an anonymous article in the Annales des Sciences Naturelles (ii, 1824, pp. 75-6) from which I have previously quoted, the statement is made that Jamison first announced the perforation of the spur, that Van der Hoeven could not find it, but that Meckel confirmed its existence, as well as that of the gland, in a letter to Brechet, the editor of the Annales.
In 1826 Knox recorded the presence of a rudimentary spur in the female echidna, and claimed that this discovery disposed of Home’s suggestion of a copulatory function.
Isidore Geoffroy’s article on Ornithorhynchus in the Dictionnaire Classique d’Histoire Naturelle, published in 1827, refers to a publication by Dr. Parmeter in Sydney:—“Cet auteur établit que les mâles emploient leur ergots pour tenir les femelles immobiles dans l’acte de la copulation, et il a publié sur ce sujet un petit Mémoire imprimé dans la Gazette de Sidney.”
Dr. Parmeter was in practice on the Hawkesbury River, and must have had opportunities of observing the platypus, which was then so plentiful there. A weary search through the pages of the Sydney Gazette brought to light only two occurrences of his name, apart from casual attendances at the inquests which formed so prominent a feature of the early days. One of these is at the foot of an advertisement deploring the fact that his patients were not paying their bills, and threatening drastic action unless they mended their ways; the second was the advertisement of a forthcoming publication, The History of Australia by T. Parmeter, M.D., with a list of a few advance subscribers—but it would seem that the work was not published. In the Gazette of December 4, 1823, there does, however, appear a letter, signed “Observator,” which is almost certainly the memoir referred to.
Like most early statements about the platypus, it contains 80more conjecture than observation. It is a comment on an excerpt concerning Knox’s dissection of the spur and gland which had appeared in a previous issue. Parmeter says:—
“I take leave to remark, that I have dissected this animal particularly, to ascertain this much controverted point, and have not been able to trace, either in the living or dead animal, the virus supposed to be contained in the sac; and I am not solitary in my opinion, that there is no poison; nor is it, properly speaking, a gland, which the spur is conjoined to.... That the plectrum answers the particular purpose of a prehensile, is more rational to conjecture, than that nature should have furnished the male with a weapon (offensive or defensive), and not similarly provided the female.”
This can scarcely be said to justify Isidore Geoffroy’s statement that Parmeter has established the fact that the males employ their spurs to hold the females. Further on in Parmeter’s letter the following lines occur:—
“I therefore invite any Member of this infant Association[7] to correct me, from an authenticated fact, of any individual having been injured from the wound of this animal’s spur.”
This, from a medical practitioner who lived for many years among the farming community on the banks of the Hawkesbury at a time when the platypus was notoriously abundant in that river, and yet who had never known of a case of injury from its spurs, points to the great rarity of such an occurrence. The reason is that fully ninety per cent would be drowned in the fish-nets, and the remainder killed before being handled.
In 1829 there appeared in the Edinburgh New Philosophical Journal a letter from Thomas Axford, Esq., dated from Thorpe, Tasmania, 30th June, 1828. He writes (p. 400):—
“It is my firm conviction that the animal has not the power of instilling poison by its spur; and I believe this appendage is used principally for securing the female in the season of love, though it may also be useful in enabling the animal to climb the steep banks of rivers. I have taken several large 81females, and I suppose old ones, with the hair worn off, and only the fine fur left on their rump; and although I have killed very large males, never found one in that state.[8] The moment I saw the first old female thus denuded, it struck me that the denudation must arise from the action of the spur of the male in holding the female.”
Verreaux (1848, pp. 130, 133) stated that the spurs were used to hold the female in copulation, being applied to her posterior part; and that, though he had handled many male platypus, none had ever attempted to use its spurs as weapons of offence against him. Dr. Bennett records the same experience. He endeavoured (1860, p. 107) to incite the animal to use its spurs, placing it in a variety of positions, and teasing it to make it angry, but without success. He was convinced that the spur was not used for offensive purposes. Krefft (1871) writes:—
“It is possible that the spur has some poisonous properties during certain seasons of the year, because we remember being cautioned to handle a male Water-mole, by a gentleman who said that he once had been wounded in the arm by one, and had suffered severely in consequence.”
Spicer (1876, pp. 162-3) records the case of Mr. Augustus Simson, of Gould’s country in Tasmania, in a communication read before the Royal Society of Tasmania on 13 November, 1876:—
“About three weeks ago, he and Mr. Stephens, the School Inspector, were walking by the side of a lagoon, when their attention was attracted by a Platypus, which had swum across from the other side, and was on the point of making its way under the bank. Mr. Simson, an active, energetic man, at once rushed down the bank, and secured the animal. Now, I fancy, this fact alone is worthy of record; for of all the shy wary animals in existence, Platypus is amongst the shiest and most wary. Under ordinary circumstances, it is no easy matter to catch even a passing sight of one; but here the creature was caught in open daylight....’ The platypus was put safely in 82a bag, but escaped. Mr. Stephens now takes up the story:—
“After an exciting chase, Platypus was recaptured;[9] but this time he revenged himself by giving my friend a severe wound on the hand, one spur slightly tearing the palm, and the other the back of the hand, making a deep puncture between the knuckles of (I think) the first and second fingers. The pain from this was intense, and almost paralysing. But for the administration of small doses of brandy, he would have fainted on the spot. As it was, it was half an hour before he could stand without support. By that time the arm was swollen to the shoulder, and quite useless, and the pain in the hand very severe. No ammonia was to be had; no medical assistance was available; and the only treatment that could be adopted, was to keep the whole arm for a night and a day in wet bandages, which seemed to alleviate the pain a little, and to reduce the inflammation.
“A week later I was informed by letter that the swelling had subsided, the hand being still very tender, with a sensation as of a severe bruise. From this time there was a slow but gradual improvement.”
A letter from the victim substantiates this statement, and he adds (p. 163):—“Some natives tell me they would rather lay hold of a snake than a Platypus.” On the same page Stephens describes the mode of attack, which was precisely the same in this case as in that described by Jamison:—
“The mode of attack is not by scratching, but by a powerful lateral and inward movement of the hind legs, the spurs being thus brought together like the points of a pair of callipers.”
The late Professor Sir Anderson Stuart, of the University of Sydney, records further cases of poisoning in his Presidential Address to the Royal Society of New South Wales, published in the Society’s Journal for 1894. His account, which occurs on pp. 5-9, is as follows:—
“I have no doubt whatever that it is, at least at certain 83seasons, a powerful poison. I have from time to time made enquiry, and have also advertised for information, and I have two good accounts from very intelligent hunters of the animal, in widely separated parts of the Colony, which coincide perfectly, so that I have no doubt myself that they accurately represent the main features of the action of the poison in dogs and as observable by laymen.
“One account shows that the males fight very fiercely while in the water during the pairing season, mostly applying themselves to each other belly to belly. The scratches are mostly on the under surface of the tail. The females are very seldom found scratched. One of the hunters tells of a dog he had which was ‘stung’ on three different occasions—each time both spurs were employed, and the wounds were always on the dog’s cheeks. The wounds are always described as on the head or face somewhere, because they are inflicted while the dog is retrieving the wounded animal. The effects followed very quickly, like the sting of a bee. Within a couple of minutes the head began to swell, and on the first occasion had reached a ‘tremendous’ size within a quarter of an hour. This swelling gradually disappeared, and was gone in thirty-six, ten, and three hours on the first, second, and third occasions respectively. The swelling and all the other symptoms were less marked the second than they were the first time, and the third than the second time. The swollen head was tender to touch, for the dog ‘sang out’ when it was touched there. The eyes were at first closed up by the swelling, and when again visible were ‘wild-looking.’ The dog became sleepy, as if under the influence of a strong narcotic, so that he had to be carried to the camp, and he moaned from time to time. The dog would neither eat nor drink, but there was no salivation, vomiting, diarrhoea, tremor, convulsions, nor staggering. Breathing was difficult, but not very. This dog quite recovered.
“My other account is from one of two brothers who were both great hunters of platypus, and he confesses to having been wicked enough to have shot many thousands during his thirty-two years of work. He had four valuable water-dogs 84that died from the ‘stings.’ On one occasion he actually saw the platypus strike, heard the dog whine, saw the wound, and the train of symptoms ending in death. These were comparatively large dogs. He knew that after he himself gave up hunting, his brother, who went on with it, lost dogs too. The drowsiness was so intense that he has had to carry the dogs on horseback with him for as long as three hours.”
Here follow details of the accounts given by Jamison and Spicer, which are quoted above. Anderson Stuart sums up (p. 9):—
“Now if we review these four accounts, we note that—
“1. They are all absolutely independent, not one writer knowing anything of the other three, three hailing from different parts of the colony of New South Wales, and one from Tasmania, and all from different periods of time.
“2. Two were in the human subject and the rest in dogs.
“3. The train of symptoms, mutatis mutandis, agree most perfectly.
“4. In all cases the poison was allowed to follow its natural course, nothing but external applications, if anything at all, being ever employed by way of treatment.
“5. The symptoms were specific, and differed entirely from the ordinary surgical effects of lacerated wounds.”
He concludes that the gland secretes a powerful poison at certain seasons, probably during pairing-time.
Martin and Tidswell (1894) made a chemical examination of the fluid secreted by the crural gland, and an inquiry into its physiological effect when injected into rabbits. Their paper gives an excellent summary of the history of the controversy, and mentions cases of poisoning other than those given above. Thus on p. 476:—
“Under the heading: ‘The Poison of the Platypus’, the following extract from the Maitland Mercury is quoted in the Australian Journal of Education (1869):—‘On Tuesday, the 9th instant, when Mr. E. was fishing in the river near his residence, he found that a Platypus had got entangled in the net, and upon catching the animal it immediately struck the two 85spurs attached to its floats or arms into the forefinger of Mr. E.’s left hand, with such force that they penetrated through the skin and into the muscles of the finger, and it was with great difficulty that Mr. E. at last succeeded in ridding himself of his unwelcome intruder and eventually killed it. Mr. E. all the time suffered intense pain, and presently the wounded finger, then the hand, and ultimately the whole arm up to the shoulder swelled to a serious extent. The symptoms usually following snake-bite also set in, and after a day or two Mr. E’s state became so serious as to alarm his friends for his safety, and Dr. G. having been sent for, he applied ammonia and the usual remedies against snake poison, and we are glad to learn that Mr. E. has now entirely recovered.’”
On p. 479 Martin and Tidswell quote A. Nicols (Zoological Notes, London, 1882, p. 116):—
“A. Nicols records having wounded and captured a Platypus which was lively enough to scratch him with its sharp claws, but made no attempt to use its spurs when handled. The native who accompanied him, however, expressed fear of the spur. Nicols thought that the spur and its gland might be ‘a remnant of conditions of life very different from those under which the animal now exists.’ He considered that, although it might possibly be used in contests with its own kind, ‘there is no reason for attributing a poisonous character to this weapon.’”
On p. 480 the authors mention four more cases (two in men and two in dogs) recorded by Dr. Lalor in a communication abstracted in the British Medical Journal for June, 1894, p. 1332. There is thus a considerable body of evidence for the poisonous nature of the secretion.
Martin and Tidswell proceed (pp. 482-487) to an examination of the anatomy and histology of the gland and duct, which they figure on their Plate XXVIII. The glands are more or less kidney-shaped bodies, with the concave border outwards, symmetrically disposed on either side of the vertebral column above the acetabulum and femur. Each gland lies in a special compartment of the deep fascia, covered by the panniculus carnosus 86and skin, between the muscles of the leg on the outer side, and the gluteus maximus on the inner. An average gland measures 3 cm. in length, 2 cm. in breadth at its widest part, and about 1.5 cm. in thickness. The surface is lobulated.
The duct emerges from the posterior half of the outer border, and passes downwards, with the nerves and blood-vessels, on the posterior aspect of the leg. It measures about 5 cm. in length, and, in an undilated state, about 2 mm. in external diameter. After leaving the gland it passes down under the biceps muscle, crosses the tendon of the gastrocnemius, and reaches the spur. Here it becomes dilated into a sac, which is so deeply embedded in the ligamentous tissue at the back of the tarsus that its isolation is a matter of difficulty. From this dilation a prolongation extends into the canal in the spur, which is attached to a supernumerary tarsal ossicle, articulated to the astragalus and tibia.
The gland is enclosed in a capsule consisting of an outer layer of unstriped muscle, three or four cells deep, and an inner coat of white fibrous tissue, which is continued into the mass of the gland as a stroma separating the alveoli. It is of the compound racemose type. The duct, traced into the gland, divides repeatedly into smaller and smaller branches; the final branches open into the alveoli, which are dilated, and lined by a single layer of epithelial cells situated upon a basement membrane.
Sections across the duct show that it possesses a single lumen close to the spur, which becomes multiple as it is traced backwards towards the gland. The wall is composed of fibrous tissue, without any muscular elements whatever, and is lined by an epithelium composed of four layers of cells. The saccular dilatation at the base of the spur has the same structure.
The fluid secretion, which is described as ‘limpid and opalescent’ by Martin and Tidswell, was chemically examined, with the following result (p. 490):—
“Our conclusions as to the composition of the secretion drawn from the above experiments are:—
(1) It is a solution of proteids.
87
(2) That the greater portion is composed of a proteid belonging to the class of albumins, and that in addition a small quantity of proteose is present.
(3) Nucleo-albumins are absent.”
The authors remark that the toxicity of snake venom is known to be due to its proteose content; but in their experiments with the platypus fluid, because of the smallness of the quantity of material available, it was not possible for them to separate the proteose from the albumin. Consequently the limited number of experiments which were possible had to be made with the mixture.
Experiment I: A rabbit had .05 gramme of the ‘poison,’ dissolved in 5 cc. of .75-salt solution, injected under the skin of the abdomen. Upon the following day a swelling the size of a duck’s egg had appeared near the seat of the injection, which was tender to the touch. The animal was sick, with dull eye, eating sparingly, and its temperature had risen by one degree Fahrenheit. A day later, the swelling and temperature were reduced, the animal was much livelier and taking food. In a couple of days more it had completely recovered.
Experiment II: 6 cc. of a solution of the dried poison in .75-salt solution, of such strength that the dose actually contained .06 gramme of the actual gland secretion, was injected through a cannula into the jugular vein of an etherized rabbit. The experiment was arranged so that a simultaneous record of blood-pressure, respiratory movement, and time-rate was recorded upon a revolving drum covered with smoked paper. Within three seconds from the commencement of the injection the blood-pressure fell by 40 mm. of mercury, the heart-beats becoming less frequent. At the same time the respiration became hurried and exaggerated, and speedily terminated in a series of expiratory convulsions, in the course of which the blood-pressure rose again, but speedily fell. In a minute and a half the animal was dead. An immediate post-mortem disclosed that the right side of the heart and the whole of the venous system were full of clotted blood; the left chambers of the heart and the pulmonary veins contained fluid blood, 88and there was an extensive endocardial haemorrhage. Martin notes that this condition is precisely similar to that induced in rabbits by the intra-venous injection of snake venoms.
Two further experiments were made similar to the latter of the two above, but with smaller doses of poison. The dose for Experiment III on p. 494 of the paper is given as 0.4 gramme, but this obviously should read 0.04 gramme. The former quantity was equal to the total amount of poison held by the investigators (see p. 496). Experiment III gave the same ultimate result as Experiment II, but at a slower rate; in Experiment IV the blood was not clotted, but on being drawn off coagulated at a rate much slower than the normal.
The authors claim that these experiments prove the secretion of the gland to contain some body capable of producing very considerable toxic action when introduced into the bodies of rabbits. They admit that this toxic agent may be of other than proteid nature, but do not think it likely, since the results resemble those produced by proteid poisons. The whole of the experiments, including the apparently contradictory result obtained in Experiment IV, show a close agreement with results obtained by use of snake venoms. The venom of snake is, however, 5000 times as virulent as the preparation of platypus poison used. The latter appears to be much more powerful in the production of local oedema.
Their final conclusions follow (pp. 498-9):—
“At the conclusion of our survey of the literature of this subject, we pronounced the opinion that as far as the evidence adduced went, it presented a very strong case in favour of the contention that these glands, at any rate at some seasons, produce a poisonous secretion.
“We venture to think that the results of our experiments have established the fact that the secretion is poisonous at some time of the year. Whether the animal is capable of discharging a secretion possessed of poisonous properties at all seasons of the year is not at present determined. Creighton states definitely that the gland is subject to seasonal variations in size just as is the case with the mammary gland and testes 89(Bennett). We have been unable to find on what evidence Creighton makes this statement; but the differences in minute structure observed by us lend support to this view.[10]
“The idea naturally occurs to one that this apparatus, which is confined to the male sex, owes its peculiar development to the operation of sexual selection. That it is a weapon used by the males on one another when conflicting for the possession of the females, is an idea which would become extremely probable if it could be established that the gland is specially developed at or about the pairing season. This is a point which could be settled without difficulty, provided specimens could be obtained in sufficient number at suitable periods of the year, say August and February.
“Bennett found developing ova in the uterus as early as September, so that in all probability the animals pair during the latter part of August and earlier part of September.
“That the secretion obtained by us from the glands of an animal killed in June proved actively poisonous, whilst that from an animal killed in April was innocuous, is interesting in this respect, although June would indicate a somewhat early preparation for pairing. We cannot, however, place much stress on this isolated observation, as it is quite possible that the difference in development was due to quite other causes. In the meantime the biological significance of these extraordinary organs must remain an open question.”
The following interesting measurements and dates were recorded by me after dissecting three robust adult male platypus during the rut of 1923.
| Testes | Crural | ||
| Macdonald River, N. England, | 8-10-’23 | 7 × 10 mm. | 9 × 20 mm. |
| Namoi River, Manilla, | 10-10-’23 | 22 × 30 mm. | 15 × 44 mm. |
| Namoi River, Manilla, | 23-10-’23 | 5 × 10 mm. | 8 × 20 mm. |
The last two males were taken in the same locality.
90
Although I omitted to measure the scent-glands, I observed that their size was in proportion to that of the testes and crural glands. This, then, suggests to me sexual affinity, so to speak, between all three sets of glands in or out of season, and furthermore, that such conditions prevail until all are gradually exhausted. This may occur late or early during the rut, according to circumstances.
An aboriginal once informed me that he had seen the male platypus carrying nesting-material by means of its spurs. I do not, however, put much trust in this story, for, so far as I have observed, the female builds the nest unaided. As regards bachelor quarters (even during the cold months), all the males that I have unearthed to date were in cavities of bare earth.
We have, then, a mass of evidence, contradictory in nature, yet adduced by competent observers. It will be of interest to summarize the suggestions which have been put forward as to the function of the spur, and to see whether the apparent contradictions cannot be reconciled. The suggestions made are five in number:
(1). That put forward by several early naturalists (e.g. Axford), that it aided the animal in climbing banks. This may be dismissed without further comment, for it is quite obvious that the female has greater need of bank-climbing apparatus—if such be necessary at all—than the male.
(2). The suggestion by Baden-Powell, quoted by Spicer, that the secretion is simply a dressing for the fur, a function which, it will be remembered, Geoffroy Saint-Hilaire once suggested for the mammary gland. Here, again, if an oil for the fur be needed, the need of the female is as great as that of the male. Moreover, although the hair is combed principally by means of the claws of the hind foot, the spur is not suitably placed for assisting in the operation.
(3). The suggestion of Nicols, quoted by Martin and Tidswell, that the spurs are a remnant of conditions of life very different from those under which the animal now exists. Martin and Tidswell remark (p. 480) that, while it is very difficult 91to show that the suggestion is false, it does not appear to be reasonable. A complicated arrangement, in a condition of functional perfection, would hardly be retained if it were of no service to the individual or the species. With this I agree.
(4). The position taken by Jamison, Knox, Spicer, Martin and Tidswell, and others, that the spur and gland are weapons for the infliction of poisonous wounds, either upon other males when fighting for possession of the female, or upon enemies in general. The evidence for seasonal variation in the functional activity of the gland is definite enough, but is insufficient to rule out the notion that the spur is a weapon of offence against enemies in general. The demand for protection from enemies is not seasonal; it is an affair of all the year round, and the spur is dangerous at all times.
As one of the uses of the spur is almost certainly to inflict poisonous wounds, it is probably directed against other males of its own species as well as against natural enemies. One other observer besides myself (Verreaux) has claimed to have seen the act of mating. Here at least I have confirmatory evidence that this act takes place in the water. As to whether Verreaux’s description of the event is in every particular correct or otherwise, I am not concerned. There is, of course, no doubt that the secretion, when introduced into the blood-stream of a mammal, has a powerful toxic effect, and Martin and Tidswell claim no more than this. When two males fight, I judge from the position which they take up at first that the introduction of the venom is designed by both combatants. Combats among mammals for possession of the female are rarely fought to the death. They are, admittedly, at times severe, but are tests of physical strength and endurance, in which the weaker goes to the wall with nothing more than a few wounds. I have known platypus males placed together in confinement disagree with, and sometimes kill, one another, though I have no evidence that death was caused by poison from the spurs. But I have always thought that such might be the case.
Before solving the problem of keeping platypus in captivity 92for exhibition purposes, I handled many freshly-trapped uninjured males, first while collecting, and later with specimens imprisoned in a contraption which I termed, at that time, a “Platypusary.” Realizing that the less handling the animals received, the better for the test, I did not ruffle their tempers more than was necessary. Not once during manœuvrings did a male make any attempt to use his spurs on me. I naturally came to the hasty conclusion (as others have done) that male platypus are ridiculously docile, good-tempered creatures.
Since then my opinion of them in this respect has changed, and for this reason. I placed two freshly-caught adult males together (in the breeding-season) in a large tub half-filled with water. After several endeavours to crawl up the sides of the tub—which I prevented them from doing—they became enraged, not only with me, but, apparently, with themselves and one another. Presently a quarrel began in earnest; they were floating side by side at the time, each with its head to its opponent’s tail. As they became more and more tightly pressed together for their full length, they circled around as one centrally-pivoted concern. This was caused by each combatant, with gaping jaws, strenuously endeavouring to catch hold of the other’s tail at the tip. Each in turn eventually succeeded and hung on tenaciously as though all depended on this remarkable grip. Both seemed so concerned that now my presence appeared to be unheeded. Wriggling and paddling began with vim, presumably with the object of one turning the other over without being overturned itself. Had this fight been waged in deep water, it is difficult to imagine what advantage either would obtain over the other. Ultimately there was a great splash, and the antagonists were in firm holds, heads to tails, for their full length, beneath the water; but on bumping the foreign bottom they instantly released their holds and came to the surface for a blow. As I could not induce them to battle again, I released them into the Namoi River.
It is worth noting here that, while under water in the locked position as described, the vital external jugular veins 93of each (mentioned by Martin and Tidswell) would be exposed, and nothing less than a timely parry with the versatile fore-paws would suffice to prevent a death-dealing puncture, if not actually in the jugular vein, then in close proximity to it. In any case, if this did occur it could hardly be called accidental. As in the case of all fauna in a fight to a finish the combatants would probably receive many wounds in different parts before one was totally disabled.
This, then, brings me to another point. If the “dope” (as I term it) is not of a deadly nature during the mating session, why is it always ejected by the animal when it deliberately spurs at an enemy? Hoy’s encounter with an infuriated male in the height of the breeding-season[11] convinces me that the secretion is used for offensive rather than for amatory purposes.
Before placing the males together in the tub I experimented with the larger of the two (weighing exactly four pounds) purposely to explode my initial theory that males will not intentionally use their spurs when being handled. Of course, I knew the truth even then, but I required sufficient proof to satisfy the most sceptical. This is what occurred: I first placed a special rubber gauntlet over my right forearm, then gloved that hand adequately, leaving my left free for minor purposes. After placing the robust male platypus on a table, abdomen uppermost and tail towards me, with my bare hand I held the wriggling creature by the head. Then, to exasperate him, I placed my right arm on his tail and deliberately tantalized him by tickling him from back legs to brisket with my fingers. Several times he raised his spurs simultaneously as if sparring for a grip, but each time lowered them again. Tiring of such monotonous behaviour, I removed my arm to rearrange his position and to reverse my hand-holds temporarily. But, while I was bringing my bare hand back to grasp his tail, the platypus, as though aware that I was off my guard, like a flash struck with his spur and ripped the edge of my left hand for a distance of over an inch towards the little finger. This action was so quick and unexpected that it certainly 94gave me a shock. After placing the platypus back in the box, I repeatedly squeezed the wound and made it bleed very freely.
My wife then took a hand, and flooded the wound with iodine twice within half an hour; each time she did so, the pain was acute in the extreme—far more so than when first inflicted. The rip, which was jagged by my tearing away from the temporarily rigid spur, was 1⅛ inch in length, and ⅛ inch in depth. Now, whether the pulling away of my hand simultaneously with the striking of the spur saved me from receiving a full dose of dope, or any at all, is questionable. But two or three days later, when I had almost forgotten the event, I felt a slight pain under the arm, and noticed a redness leading thereto from the wound. Nothing more serious developed; but it occurred to me that, as the iodine entered the wound, so the secretion administered with the prod of a spur, during conflict, although not a deadly poison, might at any rate cause sufficient pain, when injected into a wound, to cause the rival at once to desist. If so, nothing more is necessary when duelling for possession of the gentler sex.
While on this subject, I will explain why and how the platypus is capable of inflicting very severe wounds with its spurs. When the animal is not anxious or prepared to grip, the spur may be lifted back with one’s thumb and forefinger. This, I know, has led most observers to suggest that the power of the grip would be insufficient to penetrate the pelt of an adult platypus. If the spur were rigid like that of a cock, and used in a like manner, all would readily agree to such a possibility. Well, I can assure my readers that when the platypus feels inclined, and is prepared to strike, the position of the spur can be styled as rigid. That is to say, it is propped back to the extreme point of erection, where, when striking, it will tear all before it, or become dislocated in the attempt. But the driving power then brought to bear is not alone in controlling the precise movements of the spur. All the leg muscles concerned in the sudden twisting of an ankle combine to force the weapon 95home. I am speaking from personal experience with wild specimens in the field—not from pickled carcasses.

From experiment and observations made in connection with the actual voluntary administration of poison by an enraged platypus, and also by personally lifting to full-cock the clamped weapon of another fresh carcass, I came to the conclusion that, in the erection of the spur to a given point, say, from half-cock to full, the poison dose is regulated accordingly. For instance, when the spur is fully cocked the fluid will flow until a dew-like drop appears at the orifice near the tip. Here, ready for immediate action, the weapon stands fully charged. Its mission at this stage is first to puncture, with solid tip, the skin of the opposing subject. By so doing, the tip readily tears a way, thus preventing clogging of the orifice until the poison makes contact with at least first blood. The desire of the animal is to strike at a vulnerable spot, and the flow of poison would probably be stemmed temporarily if the spur were embedded deeply; but whether the amount of poison is measured automatically at the cocking of the spur, or whether the platypus is able to control the flow after striking, has not been determined. But the fact must be noted that, when the animal attempts to insert the spur and misjudges the mark, the spur is carried on into the fur of its own leg, and the dose spilt thereon is apparently in greater quantity than that which the tube of the spur could possibly hold with the one filling. Seemingly, then, the well at the base of the heel is drained also with the complete clamping of the spur, having been shut off from the main supply at the initial filling. It seems reasonable to suppose that the spur must be re-cocked before a similar operation can take place, otherwise there would probably be a constant leakage while the spur is clamped normally out of action.
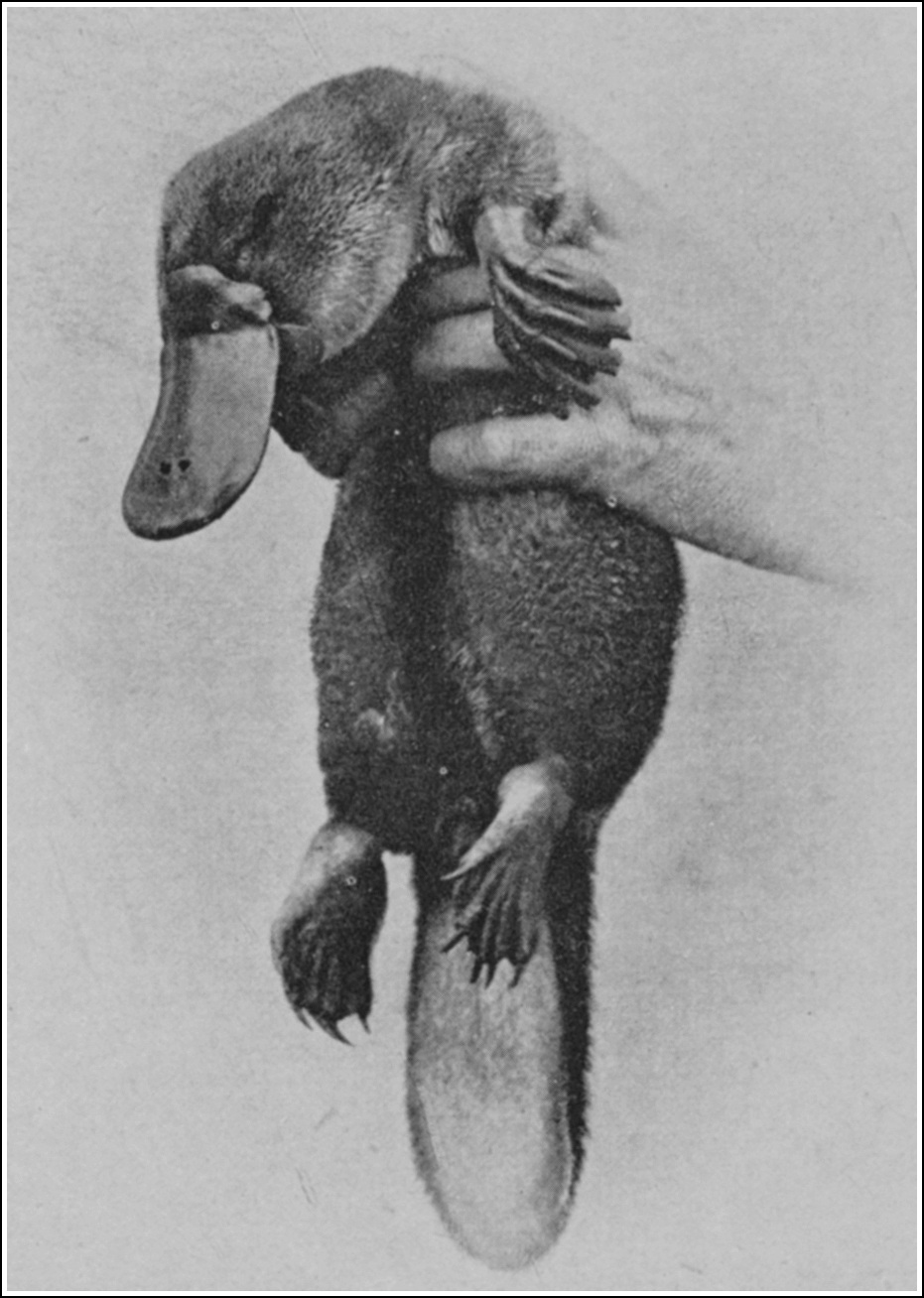
Apart from the controversy as to whether the poison is deadly or not, I strongly advise the uninitiated not to meddle recklessly with a male platypus, in or out of season, for I am quite convinced that when once the animal grips correctly, especially with both spurs, unless the platypus releases voluntarily 96(which he is not likely to do readily) the creature would surely have to be killed before the spurs could be extracted. Failing this, there is obviously only one other way out of it, and that is to lance or tear the tortured flesh of the victim.
The sudden twist of the ankles, referred to above, may best be illustrated thus. Clench both fists, place the hands side by side with the fingers uppermost, then open the palms to the extreme, suddenly, and note the rigid position of thumbs. While in that position, regard the palms as the soles of platypus feet, and the extended thumbs as “set-back” spurs. Of course, in the case of the platypus the incurved shape would incline towards the finger-tips, but to follow this curve with the thumbs the rigidity essential for striking purposes would be lost. Very well, let that be understood. At this point, then, imagine an enemy to be lodged in the palms, and held there firmly by clutching talons. This may be represented by clamping the extremities of the fingers tightly, without closing the palms. Then, with a jerk simultaneously twist both wrists inwards to the full, finishing with the backs of the hands uppermost, and thumbs clenched beneath. This will serve to convey some idea as to the awkward position of the victim so secured by an infuriated platypus.
Of an endeavour to test the effect of platypus poison on a freshly-trapped rabbit my report is as follows. Not being fully equipped with the poison collected directly from the spurs of a living specimen, because of its being swabbed up by the fur on the thigh of the animal each time the fluid was ejected through the then clamped spur, I was compelled to extract the secretion from the glands of a freshly-killed specimen. Using a hypodermic syringe, I injected into the thigh muscle (not a vital part) of the rabbit what I considered to be a double dose of the poison, that is to say, as much as the platypus would inject with both spurs simultaneously. I observed the rabbit closely for two hours after, but did not notice any change in its behaviour. My final examination, twenty-four hours later, found the subject very quiet, but otherwise apparently well. On killing the rabbit I found a discoloured swelling of the 97muscle surrounding the punctured portion, and between the skin and muscle a frothy cream-coloured corruption, quite foreign to the immediate conditions of those parts. This then is slight evidence that, at least, the rabbit suffered pain, but to what extent I could not estimate. Still, if the effect is sudden, as I believe, then it would serve the essential purpose of putting an opponent temporarily out of action at the desired moment, and for a sufficient length of time.
Where the method of striking has been observed and described, observers agree that the hind legs are brought together, causing a wound on both sides of the intervening object, which must be of comparatively small dimensions—a finger, a hand on edge, or the sharp muzzle of a dog. The animals concerned are wounded in several instances and badly frightened in others.
Taking warning from the mistakes of zoologists who have, in the past, argued plausibly about the platypus, only to be proved wrong in the end, one should refrain from argument unless armed with practical experience and observation in the field. My observations of the method of copulation were made without thought as to the function of the spurs, and I think they prove clearly that that function is definitely connected with copulation. This brings us to the fifth suggestion:—
(5). That the spurs are used for holding the female during the sexual embrace. This suggestion was first put forward by Home in 1802. He elaborated it in his Lectures on Comparative Anatomy (1823), in which he writes:—
“When I first saw the spur, I had no doubt from its situation but that one of its purposes was to prevent the escape of the female during the act of the coitus; in this I was confirmed when I found in the female, exactly in the same situation, a regular socket, lined with strong cuticle, adapted to the reception of the spur.... Having ascertained that a secretion is emitted through the spur of the male into this socket, and the parts being so minute as to require glasses of considerable power, I got Mr. Bauer to examine the socket in the female; and after overcoming considerable difficulties, the parts being very much corrugated, 98and yet retaining their elasticity, he made out the form of this socket, which corresponds exactly in shape to the spur itself: so that, when completely introduced, it must be so grasped that the male would be unable to withdraw it when coitus was over; in this respect resembling the effect of suction. The male, it would appear—at least this is the best conjecture I can make by reasoning from analogy, there being no facts to guide us—by throwing some of the secretion of the gland in the thigh into the socket, dilates it, and releases the spur; the liquor injected being acrimonious, will also irritate the female, and make her use efforts to escape.”[12]
Martin and Tidswell (loc. cit., p. 481) comment on the above as follows:—
“Home’s suggestion, which had the powerful support of Dr. Bennett, cannot be absolutely denied, as no one has, as far as we are aware, ever seen these animals copulating. Home’s main reason for such an hypothesis was that in the female there are situated in corresponding situations slight hairless depressions.
“It seems to us that for the male to apply its spurs to these depressions during copulation in the manner suggested by Home would involve an amount of gymnastic ability of which even an Ornithorhynchus is incapable.
“Moreover, Knox and Owen have shown that these depressions in the female are merely the rudiments of the male spur, and that the young female, indeed, actually possesses a spur which disappears prior to the dawn of sexual life.”
The statement contained in the last paragraph seems capable of an interpretation very different from that of Martin and Tidswell. The young of both sexes have a genital protuberance of similar size; in the male it develops into the intromittent organ and in the female it is found in a reduced condition as the comparatively insignificant clitoris, situated on the margin of an invagination. The history of the spur affords an exact analogy.
Until of late years I was under the impression that female 99platypus might shed their spurs on attaining maturity, if not before leaving the nest. On that account I was persistently on the look-out for cast-offs among the nesting-material, where furred young were found. Now, however, I have come to the conclusion that the spurs are retained by the females, but that, instead of hardening like those of the male, they soften and collapse within themselves to form the so-called socket or calloused area about the ankles, which corresponds in dimensions with the base of the male spur. This, then, would prevent the spur tip from pricking, accidentally, the ankle of the female.

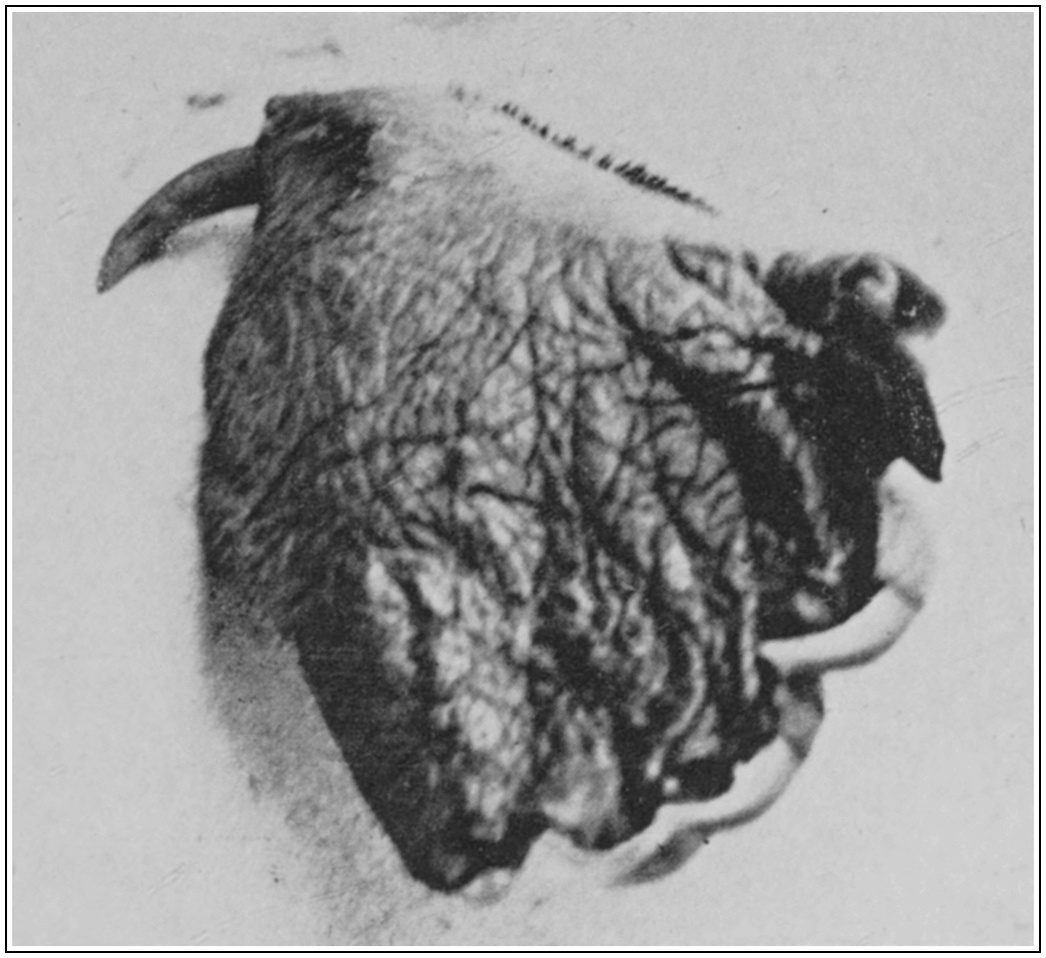
In my description of the method of copulation I think it is shown conclusively that the platypus has the gymnastic ability to grip the female with its spurs. Moreover, in the tail-to-tail position, which I shall describe later as occurring throughout the greater part of the coitus, the only possible grip the animals can have of one another is by means of the spur and foot. That this use is feasible has been shown by an experiment with recently killed animals, used while they were still in a pliable condition.
If we now turn to echidna, as we very well may—for an explanation which is adequate for the platypus must be adequate also for the echidna—what do we find? A gland is present, popliteal rather than femoral in position (which leads Owen to prefer the word “crural” as covering both), but much reduced in size, which is equal to that of a small pea, with a reduced duct running to the spur. I rely on Owen’s account in the Penny Cyclopaedia, confirmed by a recent reinvestigation by Mackenzie and Owen (1919, p. 38), in which the authors conclude:—“In our opinion this body shows evidence of retrogression similar to that seen in the vermiform appendage and other structures.”
No case of venomous wound caused by the spur has been recorded against the echidna. Semon (1894, p. 7) writes as follows:—
“There can be no doubt that the spur of the male, and the glandular apparatus connected with it, is to be looked upon as100 an organ for sexual excitation. Without at present bringing forward any theory as to the function of the organ, I can at least completely substantiate Bennett’s observation that the significance of the spur is not as a poison weapon, nor, indeed, any kind of weapon at all. Not one of the hundreds of echidnas which I have handled in a living state ever tried to use its spur as a weapon.”
It is generally believed that the echidna has evolved its spiny covering for protective purposes, but it is obvious that spines are useless as offensive weapons. Semon has apparently been influenced by Home’s theory as to the use of the spur for sexual excitation, and by Bennett’s opinion that the spur is not used as a poison weapon. His own experience in handling hundreds of echidnas is not conclusive evidence against the use of the spur as a weapon, for the reason that, when handled, they tuck their legs within the protective covering of their spines. The only way to prevent the echidna from rolling itself into a ball is to place it on its back on a board, with straps at the corners securing all its legs. In that position its spurs are out of action. The animal apparently has no need of an offensive weapon against enemies in general; but in all forms of animal life there is rivalry among the males, and whether the echidna ever used, or still uses, its spurs during copulation, or not, it would appear reasonable that the spur is the natural weapon for males to use on each other, especially as the only way in which they can possibly come together in combat is on their hind legs, using the out-turned claws as supports, and with the front of their bodies in contact for their full length. In this position their spurs could be used on the only vulnerable portion of each other, whereas, if the contest for possession of the female were to be carried on in any other position, the result must be a stalemate owing to the impregnable covering of spines.
Bill Lancaster, of Manilla, N.S.W. (who has been my assistant in field work), informed me that only once in his sixty years of bush experience did he see the echidna in the act of copulating. It occurred on the side of a mountain. 101He described the participators as forming one large ball of quills with a pair of muzzles just protruding at one end, the whole resembling a spiny melon with split stalk attached. After a minute or two Lancaster rolled them over with his boot and, as this had no effect, he deliberately kicked them down the mountain side, but even this failed to separate them and they remained together for some time after reaching the flat ground below.
I have since examined many echidna specimens (both sexes) and noted particularly the “cowlick” formation of their quills about the hinder parts. These they are obviously unable to close down completely even when walking in freedom in the bush, quite unaware of the presence of an observer. I therefore consider it impossible for these creatures to copulate in any other position than that described by Lancaster—abdomen to abdomen, and head to head. Of course, they may prefer to lie on their sides in the initial stages, but I consider it more feasible for them to stand up on their hind legs, partly supporting each other with their fore-paws until the desired position is attained; after that, any attitude may be assumed; but, for the sake of safety to both during the breakaway, the side-to-side movement should prove the simplest.
On August 11, 1919, I examined the spurs of an adult male echidna, and found them embedded in a swollen fleshy sheath on the external portion of its heels, or lower legs. On pressing the sheath down, the spurs appeared to shoot up suddenly, like a splinter from festered flesh. Here I discovered, around the base of both spurs, a creamy-coloured discharge, which I consider, if applied to an open wound, would not tend to heal the flesh. The condition appeared to be quite normal, but I do not know of any previous record of a similar observation, and cannot offer any opinion as to its purpose, or whether it was peculiar to the mating season.
If it is impossible for the male to make use of his spurs during copulation while in the position described by Lancaster, the grip is evidently obtained by the curiously-shaped long claws of both sexes.
102
Wood Jones (1923, p. 37) sums up the position in regard to the platypus in the following terms:—
“Thoroughly reliable observations, however, carry us this far. The male Ornithorhynchus can, and at times does, strike with its spurs when handled. The spurs are capable of inflicting a wound, and this wound is followed by definite symptoms. The symptoms are somewhat akin to those present in cases of non-fatal snake-bite, and men and dogs are equally affected when wounded by the spur. So far as I can ascertain, no recorded observations have ever given support to any of the various opinions that the apparatus has functions other than that of an offensive and defensive weapon. That the recorded observations show that the spur is not invariably made use of when the animal is handled, is of little moment.
“We may say, therefore, that the male Monotreme possesses a secreting gland, a duct, and a hollow spur, as an anatomical entity; that the gland undergoes seasonal changes in activity; that its secretion, when injected into animals, is decidedly poisonous, and that its composition, and its effects, are probably akin to those of snake venom; that the symptoms produced by the experimental injection of the secretion into animals are similar to those described in well-authenticated cases of wounding of men, or dogs, by the spur of the living male Platypus.
“Because we have limited ourselves to the statement of certain well-ascertained facts concerning its use as a weapon, it must not be imagined that it is considered impossible that the animal may put the spur to other uses. The Monotremes are not easy animals to observe, and possibly many of their habits remain quite unknown for lack of proper opportunities for observation. But there is little to be gained by mere speculation as to the function of structures found in living animals. Observation of the living creature is required.”
Despite this judicial warning, I must plead guilty to indulgence in speculation. It has, however, been based on observations which were not available to Wood Jones. One concerns the method of copulation; this observation is admittedly incomplete, 103and it will probably be necessary to take a pair in the act of copulation to settle the matter. The second may now be detailed.
While drying a male platypus upon my knee with a towel, I felt an increasing pressure of the soles of the animal’s feet upon my thigh, and hurriedly raised it, thinking that it might be trying to use its spurs. I then noticed that the spurs had penetrated through a fold in the cloth of my trousers, which was moist from the expressed secretion. On experimenting further with this animal, I noted that there was no attempt to strike with the spurs, as does a cock. The intervening object was first grasped with the hind feet, which were then pressed firmly against it, and the spurs were brought together with a deliberate probing motion, which may be likened to the process of sewing with a packing-needle.
Finally, there is the question of expression of the secretion. Several observers have commented that they were unable to force it out by pressure upon the gland and duct. Martin and Tidswell remark upon the absence of muscular elements in the duct, and wonder how the secretion is kept from wasting. The solution of the matter may be found in Jamison’s observation—“This unexpected and extraordinary occurrence induced me to examine the spur of the animal; and on pressing it down on the leg the fluid squirted through the tube” (1818, pp. 584-5). The animal is, of course, able to bring this about by muscular action, and is not dependent upon pressure against external objects for expression of secretion. Plate 15 shows a photograph of the foot and spur of the male referred to above. In preparation for taking the photograph, a piece of card, in which a slit shaped like a button-hole had been cut, was passed over the foot to serve as a background. In adjusting this card so that the slit should not appear in the photograph, it was revolved about a quarter turn, a process which caused continuous irritation to the muscles above the base of the spur while the point was resting on the card. The result was a trail of secretion which is clearly visible in the illustration.
104
After weighing all the evidence, and as the result of my own observations, I have therefore come to the conclusion that the uses of the spur of the male platypus are those set out in the fourth and fifth suggestions, namely, as weapons and for holding the female during copulation.

105
The earliest statement concerning the burrowing habits of Ornithorhynchus that I have been able to find is contained in Jamison’s brief sentence (1818, p. 585):—“The female is oviparous, and lives in burrows in the ground, so that it is seldom seen either on shore or in the water.” This would imply that the colonists were familiar with the burrowing habits of the animal, and it seems strange that no account of the nesting-burrow was published prior to that of Maule in 1832.
Hill (1822, p. 623) certainly gives a garbled account, of which we have made mention before:—“On returning, however, we were gratified in finding that a female Ornithorhynchus had been brought in alive, having been found on its nest in a lagoon near Campbell’s River, by Mr. Rawley, who says that he was obliged to tear the nest to pieces before he could get the animal out, the nest being formed of reeds and rushes, with a long tube or entrance into it, out of which the bill of the animal only was visible.” On the following page Hill gives us a second piece of information, obtained from his aboriginal informant, Cookoogong:—“... the female sits a considerable time on her eggs in a nest which is always found among the reeds on the surface of the water.” It seems probable that Hill misunderstood both his informants. Rawley’s description is quite incomprehensible as it stands; that of Cookoogong has no foundation at all in fact.
In the anonymous article in the Antologia di Firenze, quoted in the Annales des Sciences Naturelles for 1827, we find on pp. 193-4:—
“Ornithorhynchus inhabits the marshes of New Holland. It makes among the tufts of reeds bordering the water a nest106 composed of down and interlaced roots, in which it deposits two white eggs smaller than those of ordinary fowls: it broods on them for a long time, hatches them like a bird, and only abandons them when threatened by a formidable enemy. It seems that during the whole time it eats neither seed nor herb, but contents itself with mud, taken close at hand, which serves to nourish it; at least, that is the only substance found in its stomach.”
With the exception of the reference to the mud, this account seems to be a mixture of that given by Rawley and Cookoogong, with a dash of Lesson, and seasoned to taste by the anonymous author’s imagination. It is possible, however, that the writer had access to some account which has escaped observation, and even that he had had personal experience.
The first actual description of a platypus’ nesting-burrow is that given by Lieutenant the Hon. Lauderdale Maule of the 39th Regiment (P.Z.S., 1832, pp. 145-6):—
“By the care of a soldier of the 39th Regiment who was stationed at a post on the Fish River, a mountain stream abounding with Platypi, several nests of this shy and extraordinary animal were discovered.
“The Platypus burrows in the banks of rivers, choosing generally a spot where the water is deep and sluggish, and the bank precipitous and covered with reeds or overhung by trees. Considerably beneath the level of the stream’s surface is the main entrance to a narrow passage which leads directly into the bank, bearing away from the river (at a right angle to it) and gradually rising above its highest watermark. At a distance of some few yards from the river’s edge this passage branches into two others, which, describing each a circular course to the right and left, unite again in the nest itself, which is a roomy excavation, lined with leaves and moss, and situated seldom more than twelve yards from the water, or less than two feet beneath the surface of the earth. Several of their nests were, with considerable labour and difficulty, discovered.”
Dr. George Bennett (1835, p. 248) published the first figure 107of a burrow—a woodcut, of which the details were not very true to the facts as we now know them. He also described the burrow, believing himself to be the first European who had opened one. As he does not give the precise date, one cannot speak with certainty; but it is most probable that Maule was before him, as it is hardly likely that Bennett, had he made such important observations during, or prior to, 1831, would have withheld them from publication. Bennett’s description in Gatherings of a Naturalist (1860), is substantially the same as in the paper of 1835. Describing the first burrow discovered, he writes, commencing on page 114:—
“The entrance or vestibule of the burrow was large, particularly when compared with the width of the passage continued from it, measuring 1 foot 3 inches in depth and 1 foot 1 inch in breadth. Instead of laying the burrow entirely open from the entrance to the termination, which would have been a laborious undertaking, holes were opened at certain distances in the direction of its course, according to the method adopted by the natives. Daraga assisted us by digging with a sharp-pointed stick; and he was able to effect his object with much greater rapidity by it than we with our spades. The burrow became narrower as it receded from the entrance, its diameter being about the usual breadth of the animal. We traced it for the distance of 10 feet 4 inches; and having just delved down upon it again, so as to perceive it still continuing its course up the bank, the beak and head of a Water-Mole were seen protruding for an instant from the upper part, as if it had been disturbed from its repose and had come down to see what we were about. It only remained for an instant; for as soon as it beheld us,—imagining, no doubt, that we could not be making such a noise there for any benevolent purpose,—it immediately turned up to take refuge in that part of the burrow which yet remained unexplored. In turning round, however, it was seized by the hind leg and dragged out. The animal appeared very much alarmed and astonished when it was hauled out of its subterranean dwelling—that is, if so paradoxical a creature could look surprised at anything....
108
“It was a great curiosity to the European residents in the vicinity, who, though often seeing them dead, had never before had an opportunity of observing one alive. Although they were supposed to be burrowing animals, yet I believe this was the first burrow explored, and the first living Ornithorhynchus captured by a European....
“I found by measurement that the distance of the entrance of this burrow from the water’s edge was 5 feet; it was on a moderately steep bank, abounding with long wiry grass and shrubs, among which, and concealed by them, was the opening of the subterranean dwelling. From the judgment which I have been enabled to form from the examination of this, as well as of several other burrows, I do not imagine that the natives have ever seen, or that anyone could see (except in a state of confinement), the mother in the act of suckling her offspring; for in the tedious process of digging the old animal is disturbed, and either endeavours to escape, or succeeds in escaping long before the termination of the burrow is attained. I did not observe any heaps of earth in the vicinity of the holes I examined; nor can I form any opinion how, in the process of excavation, the animal disposes of the loose mould. May we not suppose that the animal carries away the earth collected during the excavation, in order that the heap, which would otherwise be formed, may not point out the situation of its retreat? The burrow we explored ran up the bank in a serpentine course, approaching nearer to the surface of the earth towards its termination, at which part the nest is situated.
“No nest had yet been made in the termination of this burrow; for that appears to be formed about the time of bringing forth the young, and consists merely of dried grass, weeds, etc., strewed over the floor of this part of the habitation. The whole extent of the burrow, from the entrance to the termination, I found by measurement to be 20 feet. The burrows of the Ornithorhynchi are situated above the usual river height, but do not appear to be out of the reach of the extensive floods of the river which frequently take place during the winter season.”
109
On p. 126 of the same work there is another reference to the burrow:—
“I had previously remarked, that the situations where burrows of these animals were known to exist, had been selected by their instinct where the ponds of the river contained water even during the dry summer season, and when other parts of the river were nearly dry, or formed at best a mere trickling rivulet. Of course, where the water remained the river-weeds flourished, and the flowers now produced by them probably attracted insects, which would furnish these animals with food, in addition to the minute shell-fish which might also be found among the plants. Can they, I asked myself, confine themselves to their holes during the period of gestation? To ascertain this, two burrows were dug up, about the entrance of which tracks had been seen: one was only half completed, the animal having very probably been killed before the habitation had been finished; the second was empty, the owner having probably met with the same fate. The long grass and shrubs were very luxuriant and dense at this, the summer season of the year, rendering the exploration or even discovery of the burrows more difficult than we had before experienced; and the thick grass afforded shelter for venomous reptiles, among which black and brown snakes were numerous, rendering the process not a little dangerous.”
On pp. 130-131 another burrow is described:—
“I left Yas on the 23rd of December, and arrived at Lansdown Park, Goulburn Plains, on the 24th. On the 28th of this month, with a small party of aborigines, we visited a very beautiful part of the Wollondilly River, which passes near this estate, and which has the native name of Koroa. It was a noble sheet of water, extending to some distance, and abounding in wild ducks of various species. We then proceeded to explore the burrow of an Ornithorhynchus which had been discovered. The aborigines used their hard-pointed sticks, called kiar by them (the same name is applied to our spade in their language); and although the ground was firm, they succeeded as quickly as we could have done with our spades. The method of laying open110 the burrow was by holes dug at about 4 or 5 feet apart, a stick being passed up to ascertain the direction of the excavation.
“As we proceeded in exploring, there were abundant good omens to encourage us; for, besides fresh tracks of the feet of the animal, pieces of grass, weeds, etc. (such as they strew at the bottom of the termination of the burrow to form a warm nest for their young), were seen. On every indication of the presence of the inmate, the older blacks quietly passed either the earth from the under surface of the burrow bearing recent impressions of its feet or tail, or the pieces of grass, reeds, etc., to one another, for the opinion of each, and if in favour of the presence of the occupant, the digging up of the burrow was continued, the indications so well known to them giving fresh hopes and renewed vigour to the diggers. The extent to which this burrow was continued up the bank in a serpentine form was very great; and after a most laborious task in exploring it, in consequence of the hardness of the ground, the termination was attained at a distance of 35 feet from the entrance. Extensive as this may appear, burrows have been found of even 50 feet in length.
“On arriving at the extremity of this very large burrow, a growling was distinctly heard: this I at first thought proceeded from the old one, which I now believed I should have an opportunity of viewing with her young; but, on reconsideration, thinking it more probable that the old one had forsaken them (as I noticed during the course of laying open the burrow that we had not seen her come down, in the usual manner, to ascertain why we destroyed her habitation), I could not account for it, more especially when, on the termination of the burrow being laid a little more open, the fur of the animal or animals was seen. What then surprised me was, that although there was abundance of growling, there was no movement of the animals to escape. On being taken out, they were found to be full-furred young ones, coiled up asleep, and they growled exceedingly at being exposed to the light of day. There were two of them, a male and a female, of the dimensions of 10 inches from the extremity of the beak to that of the tail. They had a most 111beautiful, sleek and delicate appearance, and seemed never to have left the burrow. The nest, if it may be so termed, consisted of dry river-weeds, the epidermis of reeds, and small dry fibrous roots, strewed over the floor of the cavity, which was of sufficient size to contain the mother and her young.”
The only further reference to the burrow which need concern us here occurs on p. 146:—
“I have no doubt the Water-Moles make their burrows high in the banks—at such a height as to be out of the reach of the floods which occasionally prevail: if they did not adopt some plan of the kind, they would be destroyed, or drowned in their burrows by the floods; for although very amphibious in their habits, they require to repose on the dry land, and also to respire atmospheric air at short intervals of time.”
George Bennett maintained his interest to the very end of his long life. But the pioneer discoverer rarely wins through to complete and adequate knowledge of his discovery; and Bennett, owing to the fact that he adopted the aboriginal method of opening up the burrows, and did not follow them along their full length, missed several important features in the structure of the burrow.
The next description of the burrow is that of Verreaux (1848, pp. 128-9), who observed the platypus in Tasmania. His account is passably accurate. He makes no specific claim to have opened up burrows himself, but mentions (p. 131) that a Dr. Casy found two nests, one with one young one, the other with two; so it may be that his description is founded on Casy’s observation rather than on his own. However that may be, the translation of the account is as follows:—
“The Ornithorhynchus dwells by preference in marshy places, but is nevertheless not so wholly aquatic as one might suppose from its structure. It digs deep burrows, that is to say, of considerable extent, which are at most fifteen to eighteen inches below the surface of the ground. These burrows have two or three outlets, and are usually divided into twelve or fifteen branches; usually one of these outlets is placed beneath, or at the level of, the water, to facilitate retreat in case of danger. Although112 these burrows, which are dug in clayey banks, have a great number of passages, they ordinarily include but a single nest, placed right at the end farthest from the water, and in an enlarged space; this space seems able to hold three or four of the animals. The nest is composed of débris of reeds and other aquatic plants, and forms a bed thick enough to protect the Ornithorhynchus from the damp produced by the constantly percolating water.... It is ... an excellent digger; and I have watched one, in very gravelly and very hard ground, successfully dig a hole more than two feet deep in less than ten minutes. For the purpose of this operation, the webs of the fore-feet, which are so highly developed for swimming, undergo a curious transformation, disappear, and leave visible only the powerful claws, which are equally useful for climbing when it is a question of surmounting an obstacle. In the attitude which it assumes when burrowing, one might take this animal for a mole rather than a swimmer. I have witnessed the quickness with which they can dig in the muddy places which they prefer. The beak is first used to dig the earth, then the claws are brought into play.
“One observation worthy of note, which shows a resemblance between Ornithorhynchus and the beaver, is that, as it digs, it uses its tail to beat and consolidate the earth. During this work the animal, twisted in the shape of an auger, turns upon itself. The tail, moved by powerful muscles, follows the movement. I have observed this fact with several live individuals, which I placed in a box filled with moist earth, and have been able to study at all times.”
The account of Ornithorhynchus in Gould’s beautiful folio Mammals of Australia (1863) is based very largely upon the observations of Bennett and Verreaux, supplemented by those of the author himself. He writes, on p. 1, of the burrow as—“a retreat to which it resorts during the day or on the approach of danger.” No distinction is made between resting- and nesting-burrows; nor does Gould add anything from personal observation to the sum of knowledge of the burrowing habits.
The first precise description, with measurements, of nesting-burrows 113is that of Mr. G. F. Bennett (1877, pp. 161-166), son of Dr. George Bennett, who examined three burrows on Lockyer Creek, a tributary of the Bremer River, near Helidon in southern Queensland. The first one opened was twenty feet in total length, with cross-section four inches by three. The entrance is shown as just under water; five feet from it a chamber measuring twelve by eight by six inches opened off the burrow on the right side. Five feet farther on, and also on the right side, a second chamber occurred; ten feet beyond that, the nest. The nesting-chamber measured eighteen inches by ten, with a height of eight inches; in the nest, which was composed of dried grass, reeds, and gum-leaves—all, from their blackened appearance, evidently collected under water—were two young ones, estimated to be a month old. This nest was opened on 27 October, 1876.
Almost a month later a second burrow was found, eleven feet in length, with two offset chambers, one on each side, and with a nesting-chamber excavated but containing no nest. A third contained three offset chambers, and two young in a nest, which was four feet underground—a very unusual depth—and twenty-two feet above the level of the entrance.
Caldwell has nothing to say about the burrows. His only reference (1887, p. 466) is as follows:—
“In September my friend Bloxsome superintended the transfer of the camp to the colder river Mole, further south, where we hoped to dig out the later stages of Ornithorhynchus from their nests. I employed some white navvies, who opened up a large number of burrows, but the renewed exposure in Queensland had brought on my fever again, and this seriously interfered with the completion of the Ornithorhynchus series.
“The later stages of Monotreme development have, therefore, to be worked out, mainly with Echidna material.”
The last sentence might imply that some later platypus material was obtained; but, if this were the case, it is remarkable that Caldwell has made no mention of it. His remarks are so non-committal as to justify a suspicion that he got neither eggs nor young from the burrow.
114
Semon (1899, p. 42) describes the burrows thus:—
“These burrows have one entrance above and one below the water, uniting in a tube of 20 to 50 feet, which leads slantingly upwards from the river-surface and ends in a little cave. I have never found burrows with a greater number of tubes labyrinthically entwined, and believe that their existence may be regarded as exceptional.”
Semon here repeats the popular belief that there are two entrances, one below, and one above the level of the water. This does happen occasionally, and the reason will be explained later; but it is the exception rather than the rule.
Lucas and Le Souef (1909, p. 145) allow the burrow one paragraph:—
“The home of the Platypus is a long narrow tunnel-like burrow, which begins with an entrance under the surface of the water, and then runs obliquely upwards in the bank for a distance of 20-50 feet. It eventually terminates in a rounded chamber or living apartment, lined with grass and leaves, and situated not very far below the surface of the ground. Very often the main burrow gives off at intervals lateral branches also terminating in chambers. Sometimes the burrow is provided with an additional opening above the level of the water, though this is certainly not always present.”
Despite its brevity, this paragraph contains some minor errors. The burrow does not begin with an entrance below water-level; the nesting-burrow is not a ‘home;’ and the lateral chambers (pug-pits) are excavated in the side walls of the main tunnel, and are not connected with it by branches.

To Kershaw (1912, pp. 102-105) must be given the credit of the first adequate account of the nesting-burrow, several examples of which he carefully examined in the banks of the Hopkins River near Mortlake in Victoria. He writes:—
“The river, at the spot visited, is a fairly broad, quiet stream, winding about through an extensive grassy flat, margined and shaded with large old red-gum trees. The banks in places are steep; in others they slope rapidly to the water’s edge. At the time of my visit the river was low, but when in flood it rises fully ten or twelve feet.
115
“After a careful search along the banks several burrows were located, places where the soil is easily worked being usually selected. The entrances to the burrows, when once seen, may always be easily recognised. In every instance these were situated some feet above the level of the water, varying from four to fully twelve feet above the present level. In no case did the burrows open out below the water, though during heavy floods several of those seen would certainly be covered. In nearly every instance there was an opening to the surface higher up the bank, and in one case a double opening was found in the same burrow—one immediately beyond the other—the narrow ridge of earth separating the two holes being worn smooth, as though the animal, when passing up, came out of one and immediately entered the other. These surface-holes are probably for use during flood times, when the entrance is under water.
“Along the steep, muddy bank, close to the water’s edge, the tracks made by the animals when passing to and from the water through the grass tussocks were easily recognised, being worn flat and smooth by the animal’s wet bodies. Claw marks could also be seen here and there. Such places where the projecting roots of the gum-trees run into the water are, apparently, most favoured for landing.
“The entrances to the burrows varied from four to five inches wide and three inches high. They are always arched above and quite flat below, following the shape of the animal’s body. This form is followed throughout the whole length of the burrow, though the latter may be enlarged here and there. The entrance is occasionally blocked up with earth.[13]
“The burrows do not follow a direct line, but wind about, at times very considerably. They always follow up the slope of the bank, parallel to and rarely more than a foot below the surface. In no instance was one found deeper than fifteen inches, while here and there they approach within a few inches of the surface. A short branch, from one to three feet long, is frequently 116made on either side of the main burrow; in one instance four were seen—two on either side. These end abruptly, without any apparent cause. Although in the same class of soil, the length of the burrows varies, the shortest examined being ten feet, and the longest, carefully measured, thirty-five feet. While usually terminating in the nest-chamber, the burrow is occasionally continued beyond the nest for a foot or two.
“A peculiarity which, I believe, has not been noted before is that when the burrow is occupied it is completely blocked with earth for about a foot. This occurred in three places in one burrow, and is apt to lead one to conclude that the end has been reached. From this it is also assumed that the female does not leave the nest for some time after the eggs are laid or the helpless young are hatched, and during the time the latter are attached to the mother. The object in so securely blocking the burrow is, probably, for protection against flood water in the event of a sudden rise of the river, or from possible enemies. Unless the block is removed from time to time, however, it is difficult to conceive how the animal obtains sufficient air during her lengthened seclusion.
“The nest-chamber is rounded in form, the bottom always being much lower than the floor of the burrow, which enters about its centre. All those examined were of about the same dimensions, the two carefully measured being each twelve inches deep, thirteen inches wide, and eleven inches from the entrance.
“The nest itself is composed of fine grass and gum-leaves, and completely fills the cavity. The bottom of the chamber is first covered with a thin layer of grass, followed by a thick layer of gum-leaves about four inches deep, which is continued up the sides, and—at least in one case—completely encircles the cavity, forming a compact circular nest, with the entrance in the side opposite the tunnel. It was noticeable that many of the leaves were green, and appeared to be quite fresh. The bottom of the chamber in every instance was wet, owing to surface soakage, and it is apparently with the object of avoiding this that the bottom of the chamber is excavated below the level of the burrow and so thickly lined with leaves.
117
“The first burrow examined had been opened up by Mr. Hood on the 17th October (nine days prior to my visit), and from the nest two fresh eggs were taken. These, when found, were attached to one another, and had been kept in a small box; but in two or three days they collapsed. When I received them on the 26th October they were flattened and dry. Several other burrows close by, though having the appearance of recent occupation, were untenanted, but each contained a nest, more or less complete. From one of these the shrivelled remains of two eggs were obtained.
“Later in the day a burrow was discovered which had every appearance of being occupied. It was situated on the sloping side of the bank, fully twenty-five feet from the water, and was the only one I saw with recently scratched earth at the entrance. The burrow followed up the slope of the bank for about twenty-five feet, terminating in a nest-chamber, some ten or twelve feet above the level of the river. This one was blocked up in three separate places, the last within three feet of the nest. When about a foot from the nest a peculiar grating growl was heard, and the next shovelful of earth exposed the side of the very compact structure, through which the old one hurriedly forced her way. She was permitted to traverse the excavation for a couple of feet to a depression, where she was covered with clods of earth, while a careful examination of the nest was made. This proved to be the most complete of all those examined, the whole cavity being completely filled with leaves. No opening into the interior of the nest could be seen, the entrance being evidently closed by the animal during occupation. As was the case in all those examined, it was composed of gum-leaves, with a small layer of grass at the bottom of the chamber. The lower portion was tightly compressed from the weight of the animal, while those forming the sides and top were loosely interwoven. The interior measured six inches across and four and a half inches deep. The shrunken remains of one egg were found in the nest.”
With the exception of the blocking of entrances, and two 118or three other minor matters, I can confirm Kershaw’s statements after a long term of practical experience in the field. What this has involved in labour alone may best be judged from the remarks of Professors Wilson and Hill (1908), who, like myself, spent a number of years getting together material for embryological study. They write:—
“The records of Australian biology bear sufficient witness to the fact that it is extremely difficult, even for residents in Australia, to procure the material necessary for an investigation into the development of the eggs of Ornithorhynchus. It is only through the organisation of special expeditions, with ample resources both of time and money, that any large measure of success in this direction can be hoped for.
“The animal itself, though pretty widely distributed, and probably still far from becoming extinct, is to be found, in any one locality, only in comparatively small numbers. It is now much less plentiful than formerly, owing to the demand for its fur—a demand which is still satisfied in spite of the measure of legal protection which the animal has obtained in the various Australian states. The depredations of the fur-hunter are not easily repaired, since the animal breeds only once in the year and produces but two eggs at a time.
“The eggs, when laid, are deposited in a burrow which it is far from easy to locate, and whose opening up involves a considerable amount of labour, since, apart from its great length, the river-bank in which it is situated is commonly enough permeated by tree-roots. And when at length the actual dwelling chamber or nest is successfully opened up, no reward at all may be forthcoming, or the material which is obtained may be unsuitable for the immediate purpose in view....
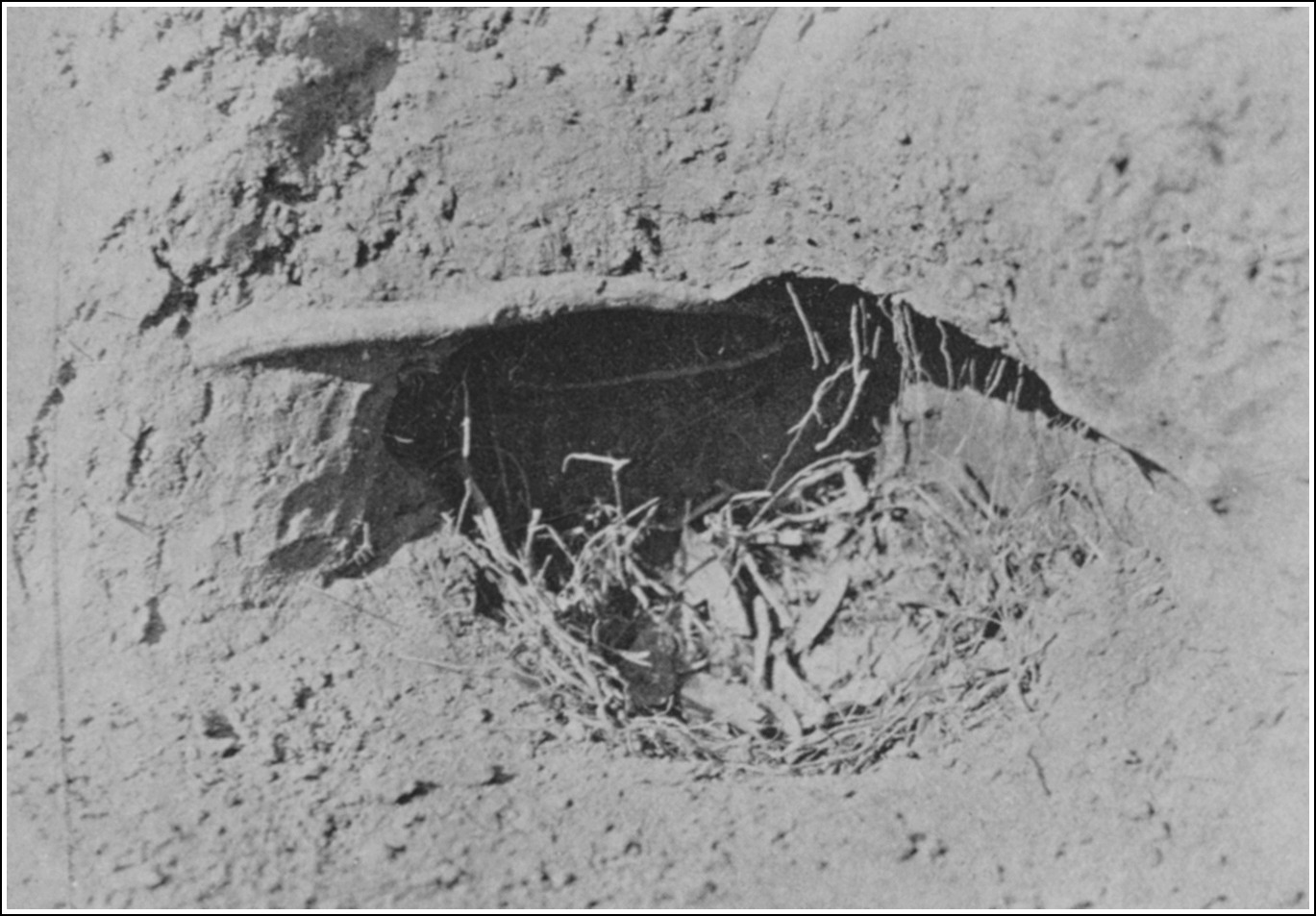
“We have only to a very limited extent been able to superintend personally the work of collection, and have found it necessary to rely largely upon the efforts of the scientifically untrained collector in the accumulation of the material for our investigations. Unfortunately, the material result of the work of such a collector during an entire season is so meagre that he requires considerable inducement to carry on the work at all; 119whilst the product to the investigator may be practically nil. The genuineness of the difficulty in procuring this kind of material is sufficiently indicated by the fact that whilst Semon, in his expedition, was successful in obtaining a very considerable number of eggs and foetal specimens of Echidna, his collection of Ornithorhynchus and embryos would appear to have been limited to a small number of early intra-uterine eggs.
“Our own collection is very far indeed from being complete, though the work of collection has been going on for quite a number of years.”

Semon (1894, p. 13) tells a similar story:—
“Concerning the development of the eggs after being laid and concerning the young after hatching I can unfortunately make no statements. I had not the good fortune even to find eggs or young in the numerous burrows which I and my white companions opened. My blacks showed the greatest dislike for this work, and were averse to troubling about Ornithorhynchus at all: we ourselves were too absorbed with the several other works in hand ... to be able to give our whole time and zeal to the wearisome and for the most part disappointing work of digging out Ornithorhynchus nests.”
The two great expeditions of Caldwell and Semon failed to secure eggs and young of the platypus from the nest. Wilson and Hill, after many years of endeavour, obtained only a very limited number, most of which have not yet been described. Since the eggs and young, which cannot escape from the nest, are so difficult to get, it may well be imagined that the doings of the elusive female, hidden in the darkness of her burrow, are infinitely more difficult to observe.
The interest attaching to precise observations of the actual burrowing method is great, the chief point being to determine the relative extent to which the highly sensitive muzzle and the powerful claws of the fore-paws are used in the process. I therefore experimented with a single female platypus which I had in captivity at my home.
The creature was placed in a specially contrived enclosure, of which the essential features were a plate-glass front and a 120wooden back, four inches apart at the bottom, widening to six inches at the top. This enclosure was filled with sifted soil, which was put in a bucketful at a time, each bucket of soil being followed by a bucket of water, until the frame was full, when the whole was tamped and thoroughly flooded with water. The object of the taper was to cause the soil to wedge, and so prevent it from collapsing on the burrowing animal. Water was used to consolidate the soil to something like the consistency of the banks in which the platypus burrows naturally in the wild state.
The enclosure thus prepared was allowed to stand for twenty-four hours, and the animal was then introduced at one side, where a portion of the earth was removed to make room for it, at 2 p.m. No attempt was made to burrow until about 5.30 p.m.; but I am not certain whether this time bears any relation to what normally happens. The observations which follow were made during a period of about an hour, by means of the plate-glass front, and during this time I kept myself concealed as far as possible from the animal.
The platypus, in order to obtain the greatest purchase before beginning to burrow, tucked the tapering end of its pliable tail between its hind legs and simultaneously hooked its out-turned hind-claws into the earth at each side. While in this crouching attitude, with stiffened top lip and splayed fore-claws it proceeded smartly to break away the earth. After burrowing for several inches it rested awhile; then it energetically contorted its neck and body so as to tamp the freshly-loosened earth tightly into the hollow surrounding it.
While stationary, the creature occasionally beat the walls with its trowel-like tail; but whether this action was deliberately intended as part of the tamping process, or was due to muscular reaction as the result of the strenuous exertion, or both, I do not profess to know. Nevertheless, it certainly did not distort or tend to break away the true design of the burrow by its spasmodic action. In fact, the structure and shape of the tail suggest that it is designed as an actual modelling-tool.
121
After a few minutes’ rest, during which the breathing was laboured and was accompanied by an apparently involuntary gulping, the animal again deliberately shovelled the earth over its head with the end of its snout. In this way it created a crude cavity in which to loll its head to one side, while, with neck shortened, it reached to the utmost with one web-palmed paw, and scratched a hemispherical hollow to one side of the tunnel, contorting its shoulders the while. The energetic digger then placed its head in the recess so made, and, without any hesitation, performed a similar operation on the opposite side with the other splayed paw. Then, to dislodge the partition separating the two recesses, it probed its muzzle vigorously into it, and then proceeded as far as the out-stretched neck permitted, to form another “loll-hole” to one side with its muzzle, in readiness for its head in burrowing a further section. Meanwhile, the well-worked earth trickled round the wriggling creature’s body, principally about the powerful shoulders, whence a portion eventually filtered down as far as the hips.
While the fore-paws and rooting muzzle were working at high tension, the hind legs were alternately keeping the specialized fore-parts well up to their work by clinging tenaciously to the solid earth. In their struggle to do this, the hind-claws actually aided the process of excavation by cutting the lateral angles which complete the arched design of the burrow.
A remarkable feature of the burrow is that no soil is ejected, but the whole of it is reduced to a consistency suitable for packing into the sides of the burrow, and even the soil dislodged when commencing a burrow is consolidated into the river-bank. When the platypus has burrowed about six inches, it packs the loose soil by contorting its body and pressing against the sides, at the same time beating the floor with its tail. The width of the excavation is about double that of the finished burrow, that is to say, the earth excavated is tamped into about half its original bulk. The whole of the earth taken from each section is not always beaten in before a new section is commenced; but the animal appears to work very systematically, 122and, according to the consistency of the soil, it moves backward from time to time and adds the finishing touches. In the experiment the captive repeated the burrowing and tamping alternately, until it eventually broke through the surface.
The powerful claws of the fore-paws do practically all the burrowing, the only help they receive being a probing or lateral shearing movement of the muzzle (when the earth is not too hard) and the pressure exerted by the hind feet. Although apparently preferring to burrow in the normal position, the platypus will readily lie on its back, or on either side, and at times will work in a spiral fashion, either while excavating or while tamping the tunnel, even in the absence of any serious obstacle; but eventually it reverts to the normal position for the purpose of finally shaping the burrow. The position in which it works does not affect the rate of its progress in the least. When confronted with temporary obstacles, such as impoverished or caked earth between the roots of trees, etc., the platypus will, miner-like, in order to avoid retracing its steps or branching off in another direction, throw itself into the required position and follow, not the line of least resistance, but of favourable soil.
The thickness of the pug or pugs tamped into position with the tail by the female prior to retiring to the nest for the purpose of laying and incubating the eggs, is about six inches, and it is packed so closely as to be indistinguishable from the surrounding soil.

During the process of dragging back and packing into the sides of the burrow the soil scratched out by the fore-paws and muzzle, the platypus exhibits its remarkable ability to reverse the action of its hind legs, and actually “advances” backwards. Students of anatomy will have observed, no doubt, that the hind feet of a platypus have a tendency to turn outwards from the flanks, and that the grooved, curved claws continue in that trend towards the tail. This is as it should be for the purpose of gripping and scratching back surplus soil while tunnelling ahead. It is also as it should be when 123reversing. Especially is this so when burdened with a packing of adhesive pug, and when, incidentally, the fore-paws are practically out of action. The muscular contortions of the body at that time certainly afford great assistance. Nevertheless the hind quarters and cumbersome tail would during one of its essential functions become useless, if not a burden, to the platypus, but for the natural provision of a “two-way” action of the hind limbs. Preparatory to pulling backwards, the trend of the hind feet continues until the sets of claws are facing one another beneath the tail. This contortion enables the animal to take a firm grip of the flattened earth-floor; it then pulls the body backwards step by step, while the fore-paws, when freed, do the shoving. The alternate action of the fore-legs in walking forward is imitated exactly by the hind legs in the reverse direction, and the platypus can thus, at will, reverse the direction of its movements. It is rather amusing to witness this act, for, at the outset, the fore-parts are usually obliterated with earth, and the tail, which in contour and elevation somewhat resembles the head, sometimes puts one at a loss to guess whether the creature is really coming or going.
In addition to the uses of the hind-claws and to the reversible nature of the entire limb, the hind foot of the platypus, especially of the male, is just as versatile in its actions as that of a chimpanzee, even to the power of deliberately gripping. During a backward march it resembles, in action and general appearance, the fore-limb of a grizzly bear, or, more nearly still, that of a long-clawed sloth. Is it possible that this versatility is reptilian in origin? What other mammal in the world is able thus to interchange the functions of its hind- and fore-legs to such advantage? Such ability may be characteristic of moles, etc., but it is certainly a remarkable adaptation.
I have already mentioned the stiffening of the top lip, and, when one realizes its extreme limpness when not employed in burrowing, it will be obvious that stiffening is necessary in order that a groove may be made in earth by lateral oscillation (“lipped-in,” so to speak). While this groove is being formed, 124and until it is sufficiently deep for the bony prong of the upper jaw to be used for shovelling the earth away, the direction of the stiffened lip is maintained by the earth above and below it.
Given favourable conditions, a platypus can excavate a cavity of approximately 6 × 5 × 5 inches in five minutes. It can tamp loosened earth completely into a six-inch section of a tunnel in fifteen minutes. This makes twenty minutes in all to a shift. Therefore, if the tunneller continued unceasingly in this manner, in sixteen hours it would have completed a tunnel twenty-four feet in length. This is the average length of a breeding-burrow.
Quite possibly the spasmodic gulping observed in the throat of a burrowing platypus is due to involuntary exhalations of spent air, corresponding to the periodical bubbling when the creature blows beneath water. Under open conditions, normal breathing may be reasonably governed at will, but this exhaust spasm appears to be quite beyond control. This is probably so that it may collect its food unhampered beneath water, and that it may also delve freely while tunnelling, without having to relinquish such immediate business for the purpose of replenishing spent air. The breathing observed while the platypus was tunnelling was at the rate of thirty respirations per minute, and the gulps in the throat approximately at intervals of twenty-five seconds. The normal breathing of a sleeping platypus I have found to be fifteen respirations per minute.
During the experiment the animal apparently kept the facial furrows, embracing the orifices of the eyes and ears, tightly closed, as when swimming under water.
Accounts of the bionomics of the platypus in current works on natural history invariably assume that the nesting-burrow is the home of the animal, in the same sense in which a rabbit-warren is the home of the colony that it contains. As has already been stated, this is not so, each burrow being inhabited by a single female, and that only during the nursing period. The usual statement that the burrow has two entrances, one under water and one above, is also erroneous. It is true that 125such a condition often exists; but this merely indicates that a rise in the river has covered the original entrance, which is always put in above water-level, and that a new entrance has been broken out, or that the secondary under-water “getaway” has been formed later by a washaway at the root of an adjacent tree.

The female excavates her burrow unaided. She selects a suitable site, preferably in a high sloping bank, well held together by the roots of trees; it is all the better if a fringe of reeds adorns its edge, so that she may come and go as safe from observation as possible. The entrance is made above water, at a height varying from nearly water-level to as much as twelve feet. When fresh, it has a definite shape—low-arched above and flat below—and measures from four to six inches in width, and from three to four inches in height, corresponding pretty accurately to the cross-section of the occupant. On account of this definite shape, it is easily distinguishable from the burrows of water-rats (Hydromys) and rabbits found in similar localities. But after a time this entrance may become adventitiously enlarged and altered in shape, so that the distinction is not always completely maintained. Bennett suggests that the entrance is usually concealed among the vegetation of the bank; but, though this may happen where banks are covered with a lush vegetation, my observations agree with those of Kershaw that there is no attempt actually to ‘camouflage’ the entrance, even though ample vegetation may be found growing close by. The entrance is usually quite obvious, and, in any case, attention directed to it by the runs of the animal, worn smooth and hard by the dragging of its wet body over them. Upon these runs the impress of the tail is usually to be seen, and the pricked tracks made by the claws of the hind feet are unmistakable.

The burrows exhibit an infinite variety of form and length, but all show the arched roof and flat floor adapted to the size of the female which has constructed them. This size may therefore be gauged with reasonable accuracy during the process of digging the creature out. Here and there, however, 126there may be, in the calibre of the tunnel, some irregularity apparently due to the adoption of an unusual position during digging in order to deal with some particular obstruction. The tunnel usually follows a sinuous course, but invariably keeps at about the same distance (from 12 to 18 inches) below the surface. This power of the platypus to judge how far it is below the surface is as remarkable as that sense which warns it when it is approaching another burrow, whether of its own kind, or of some other animal. In constructing its tunnel, it sometimes circles round, so that, if it continued on its course, it would break into an earlier section of its own burrow. This, however, never happens. In one case it was observed that the platypus stopped a foot from this earlier section, and forthwith constructed its nesting-chamber. In a number of others, it avoided breaking into its own burrow by passing under it at a distance of a foot, coming up on the other side to the usual distance below the ground-level (see page 127 and Plate 20). In the same way it avoids the burrows of water-rats and rabbits by tunnelling clear of them. I have known it go down to the very unusual depth of four feet to avoid a rabbit-burrow. In some cases, instead of going under an obstructing burrow, it turns aside, and continues its tunnel in a different direction. One more example of this uncanny sense may be quoted. Plate 19 illustrates a model of a burrow made to scale. In this the nesting-cavity is shown at a vertical depth of about three feet. The animal burrowed in at the 127usual level below the bank where it sloped, until it came to a place where the bank suddenly rose steeply. Here two alternatives were open to it. Either it must go straight upwards through rocky soil for more than two feet, or it could construct its nest where it found itself, three feet below the level of the upper bank, but still the normal distance from the lower face. It chose the latter alternative.

A great amount of discrimination is necessarily displayed in selecting a suitable soil in which to burrow. Burrows commenced in soil that ultimately proves unsuitable because of its friable nature, are—as often—promptly deserted. Although a preference for soft soil is indicated, I noticed that in one burrow which I investigated the animal had removed three inches of compacted river-gravel from the face of the bank before reaching the ideal earth behind, which she evidently knew was to be found by perseverance. Sandy banks, unless reinforced with lengthy lucerne or similar roots, are left severely alone, since the burrows tend so easily to collapse.
No general statement can be made as to the length and direction of the nesting-burrows, since both are infinitely variable. The length varies from five feet to upwards of sixty in one season’s burrowing; but all the short lengths recorded are probably abnormal, and due to exigencies brought about by flood-conditions. From fifteen to twenty feet would appear to be a fairly adequate length, though that is under the average, which is raised by a number of extraordinary length.
What it is that impels the animal sometimes to go on burrowing up to the amazing distance of 100 feet, I will here explain. These abnormalities occur only in ground that has been occupied undisturbed by platypus for many years. Indirectly, floods are responsible for the trouble that compels platypus to enlarge and renovate. These shy and timid creatures naturally do not wish to make their ideal breeding-bank more conspicuous to their enemies than is necessary; they therefore prefer to use the same entrance (when convenient) year after year, rather than perforate the face of the bank with tell-tale porches for which there is no necessity. Leave well alone, 128is their motto. Now suppose that a platypus puts in a burrow in any one season, and that it is, at some later time, covered by flood waters. Being in soft soil, it soon becomes silted up—at least, here and there. The following year, the same or even another female enters the porch and traverses the tunnel, as far as it is habitable, to investigate. If she approves of it up to a certain distance, she will cut a pit in the side wall there and then, and plug up the section which is not to her liking; then she will tunnel anew in another direction to the average distance, as if commencing from the original porch. This may go on year after year, with flood after flood, until the enormous length of 100 feet is recorded by the collector as the longest breeding-burrow discovered. Peter Yates, my able assistant, can vouch for that fact.
The nesting-chamber is generally placed at the extreme end of the burrow, though sometimes a blind branch is carried on beyond it. It varies somewhat in size, but is usually more wide than high, and measures on an average about twelve inches by eleven. In this chamber a nest is constructed, a variety of materials being used, the nature of which depends upon the locality. In the New England district the commonest type of nest is composed almost entirely of eucalyptus leaves and the rhizomes of couch grass, but I have also found willow “swishes,” roots, branchlets, and leaves; roots and stems of the reed Arundo phragmites; and the leafless branches of she-oaks (Casuarina spp.). These materials are arranged as a lining of the cavity, except for the opening where the burrow enters, and are of even thickness, except on the floor, where the lining is distinctly thicker. I have been informed by an aboriginal that he has seen a male carrying nesting-material by means of his spurs, but am not disposed to believe it, although I am quite sure he could do so. All my own observations suggest that the female alone digs the burrow and makes the nest. They also show that the amount of nesting-material never more than half fills the cavity until the babies begin to grow and toss the litter about. For instance, where 129triplets are concerned one is apt to find the nest-cavity crammed to the dome with such material.
In all the years during which I have been making observations I have never once actually seen the female in the act of carrying in nesting-material. It seems likely, therefore, that nest-building is done at night. The materials used are those closest to hand. As the foundation of one nest I found a willow “swish” more than five feet in length. It would surely be awkward to drag such a thing to the end of a long burrow. It seems likely that the animal collects a great deal of material that is lying loose upon the ground, or even in the water. But there is no doubt that it is also able to cut off reeds and grass rhizomes by means of the elongate horny ridges in the anterior part of the mouth. These ridges are also used to shred the stems of reeds, reducing them to a mass of soft fibres (see Plate 3).
As soon as the nest is completed, and before the eggs are laid, the most remarkable habit of all comes into evidence—that of plugging the burrow. G. F. Bennett (1877) was the first to observe the lateral pits from which the earth to form these plugs is drawn; but he did not observe the plugs, and therefore missed the import of the pits. Kershaw (1912, p. 103) saw the plugs, but did not connect them with the lateral pits. He writes:—
“A peculiarity which, I believe, has not been noted before is that when the burrow is occupied it is completely blocked with earth for about a foot. This occurred in three places in one burrow, and is apt to lead one to conclude that the end has been reached. From this it is also assumed that the female does not leave the nest for some time after the eggs are laid or the helpless young are hatched, and during the time the latter are attached to the mother. The object in so securely blocking the burrow is, probably, for protection against flood water in the event of a sudden rise of the river, or from possible enemies. Unless the block is removed from time to time, however, it is difficult to conceive how the animal obtains sufficient air during her lengthened seclusion.”
The mining term “pug” seems appropriate to the earth 130composing these plugs, and “pug-pits” to the cavities from which it is derived. I therefore propose to use these terms. What apparently happens is this. When the female has completed the nest to her satisfaction, she goes to the entrance of the burrow, and, turning, re-enters it. At a varying distance from the entrance she excavates a chamber opening off the side of the tunnel and compacts the earth so obtained into a pug, which is always placed close to the first pug-pit, and between it and the entrance. Proceeding towards the nest, she excavates a second pug-pit in the same way, compacting the pug behind her, and so on, without having to turn once from beginning to end. I have found from two to nine of these pugs in different burrows, the usual number being three. The pits would seem to occur indifferently on either side of the burrow, but I have noticed that the latter usually changes direction immediately after a pug. This device has nothing to do with preventing the entry of ordinary flood waters. The nest is almost always placed at a sufficient height above water-level to escape these, and the platypus can easily break out a new entrance anywhere it pleases. It is obviously a cunning device, partly for protection from enemies, but principally for securing ideal brooding conditions. It has doubtless baffled many investigators, human and otherwise. With all my experience, it has frequently taken me some time, after having arrived at a pug, to pick up the further course of a burrow. Consequently it is easy to imagine that a less experienced observer would be entirely misled, believing he had come to the end of an untenanted burrow, and thus retiring in disgust. This probably accounts for the long time it has taken to ascertain fully the animal’s nesting habits. A final pug is always placed near the entrance to the nesting-chamber.


Now to explain how the labyrinths mentioned by Verreaux are made. Very much the same thing happens as that described above, except that more breeders make use of the bank at the same time, and, in their endeavours not to clash with one another, they take most erratic courses to avoid neighbouring 131burrows. Admittedly they do not see these obstacles, any more than Verreaux did. Nevertheless they are quite aware of their proximity, and act accordingly. Hence the maze as seen and wondered at by man.
Another point is that, where an objectionable section of a burrow has been pugged off in the past, the pug, through soakage during floods, becomes solidified like the surrounding earth. When the burrow is followed by man later to that particular partition, this is generally looked upon as a blind or dead end; yet possibly, a foot or two farther on (not necessarily in a direct line), a so-called “beginningless” subway may be found further to bewilder the weary mattock-wielder. Again, every time a flood occurs, fresh strata of silt and débris are left lying on the surface of the bank after the water recedes, and thereon grass and herbage readily take root, thus increasing the depth of earth above the ancient tunnels. These, when hit upon, appear to the student as extraordinarily deep for platypus to work, in comparison to the depths previously described by the more fortunate collector who accidentally happened upon a positively new burrow, which, throughout its entire length, did not lie more than fifteen inches below the surface.
My experience goes to show that the female does not leave the nest between the times when the eggs are laid and the young are able to suck, and that she digs through and replaces the pugs whenever she leaves the nest or returns to it. The constant removal and working of the soil of which the pugs are built makes it so soft and fine that it sometimes becomes quite velvety to the touch, and clings like flour if squeezed in the hand. It is always slightly damp after being worked by the platypus; but on one occasion when I took a nest containing large triplets without their mother, the pug near the nest had dried and shrunk away from the sides of the tunnel so much that I was able to remove it in a mass, though it fell to powder in my hands when slightly squeezed. Possibly, that absent female had met her death in some way, for I am positive that it would require some days for the pug to dry and shrink to 132such an extent. However, the young, which I presented later to the Australian Museum, appeared none the worse for their term of imprisonment without food.
How does the animal make and replace the pug? Wood Jones states (p. 48):—“... the form of the tail resembles that seen in the Beaver ... It has nothing whatever to do (either in the Platypus or the Beaver) with any supposed habit of puddling clay....” After much experience of observing the habits of platypus in their wild state, I succeeded in 1925 in proving the accuracy of my own notions on this subject. While collecting on the Namoi River for the proposed National Museum which is to be erected at Canberra, I unearthed a female platypus on her nest, which contained twin young. While I was registering the temperature of the nesting-cavity, the mother made off from the nest along an offset tunnel. Knowing that I could secure her later, I leisurely finished my duties with the young and carefully extracted the nest (whole). About fifteen minutes later my assistant started in pursuit of the mother. To our surprise, the offset, which usually measures only a few feet in length, on this occasion measured about seven. After following along for about three feet we discovered a pug sealing the runway. I carefully examined the hurriedly-constructed barrier, and then followed closely along the workings as my assistant tactfully broke the earth away. Presently another pile of pug met my gaze, and beyond it the platypus could be clearly seen up against solid earth scratching for her life, and at the same time deliberately shoving the refined earth behind her with the dorsal end of her tail to pug herself in. Of course, this tunnel was there prior to her leaving the nest, but was evidently pugged off at the first section from the nest before she retired to perform her maternal duties. This pug, then, she had burrowed through and built again behind her, and she had almost succeeded in excavating sufficient solid earth at the end of the runway to pug herself in with the idea of outwitting us.
During an exceptionally wet season the earth, in certain situations, becomes sodden, like clay. At such times the pugging in 133the tunnels is found to be a conglomerate mass of marble-sized mud balls, more closely compacted than shot in a gun-shell. This confirms my statement that the nesting-cavity is sealed up before incubation begins, and that in building the pug, the tail is used for rolling the earth along the runway into position. Hence the baldness of that trowel-like member. It proves also that, wet or dry, pugging is worked methodically into position a portion at a time.
The hair on the dorsal surface of the tail of the female becomes increasingly abraded during the nursing period, so that by the time the young are in fur she usually exhibits a bald patch of some size. So much is this the case that I have several times accurately guessed the size of the young from the degree of baldness of the mother’s tail. Now this particular hair is exceedingly coarse and harsh, and could only be worn away by some such drastic use as in the tamping of the pugs, which must be removed and replaced so many times during the nursing period. Wood Jones’ suggestion that the flat tail is used to warn neighbours by continuous smacking of the surface of the water is certainly not supported by my observations of the living animal.
The thickness of the pugs varies from three inches to a foot; ordinarily it is from six to eight inches. I have found pug-material beneath the nest itself to a depth of several inches; the animal must therefore dig up and work the earth on the floor of the cavity before she begins to build the actual nest. This is probably a provision for the more rapid carrying away of any water that may reach the nest by seepage; but, on the other hand, it may serve to hold moisture, and thus maintain the damp atmosphere which seems to be necessary for successful incubation.

The platypus would appear to dig a new burrow for each breeding-season, and it certainly does so after being disturbed. Occasionally an old burrow may be refurnished and remodelled, and one main entrance used for several seasons. On two occasions only have I found more than one nest in a burrow; and a diagram of one of these burrows is given here. It contained, 134in different positions, no fewer than four nests, three of which were in various stages of dilapidation; the fourth, though scanty in material, contained twins. Mr. Hoy examined these four nests with me, and agreed that they bore the appearance of having been built at different times, probably at yearly intervals. The bank in which this burrow was placed was unusually faulty for the purpose, having a sandy subsoil. It was on a beautiful stretch of water about three miles below the New England waterfalls, where the river is in places actually bridged by rocks, so that when it is at a low level, as in periods of drought, the river becomes a chain of ponds, and the platypus is prevented from travelling far. Possibly, therefore, the animal or animals which inhabited the four nests successively had become river-locked, so to speak, and compelled to make shift in the only available quarters. We dug out three other unoccupied burrows in the same bank, each of which had evidently been abandoned on account of the surface sand collapsing while the nesting-chamber was being excavated. The platypus was probably compelled to shift along the bank until, in the only part which could be used for its purpose, it found the old burrow, and so contented itself with burrowing on past the vacant nests.
A second example was opened up on the Macdonald River on September 28, 1920. After a rainy spring the river was eighteen inches above its normal level. The original entrance to the burrow was then twelve inches below water, but a new entrance had been broken out seven inches above. The main tunnel wound gradually up the sloping bank for 36 feet, and then divided into two, one arm, ten feet long, going to the 135first nest—which contained a female and twin eggs—the second branching off to the left to another nest, which, at fifteen feet from the junction, contained a female and a single egg. The two nests were eleven feet apart in a direct line. The season of 1920 was abnormal in rainfall; but, even so, it was remarkable to find two females occupying self-contained flats in one and the same burrow. The accident that the females and their eggs were duly preserved in the interests of science probably saved them from considerable complications, which might have ensued if they had been allowed to bring out their young.
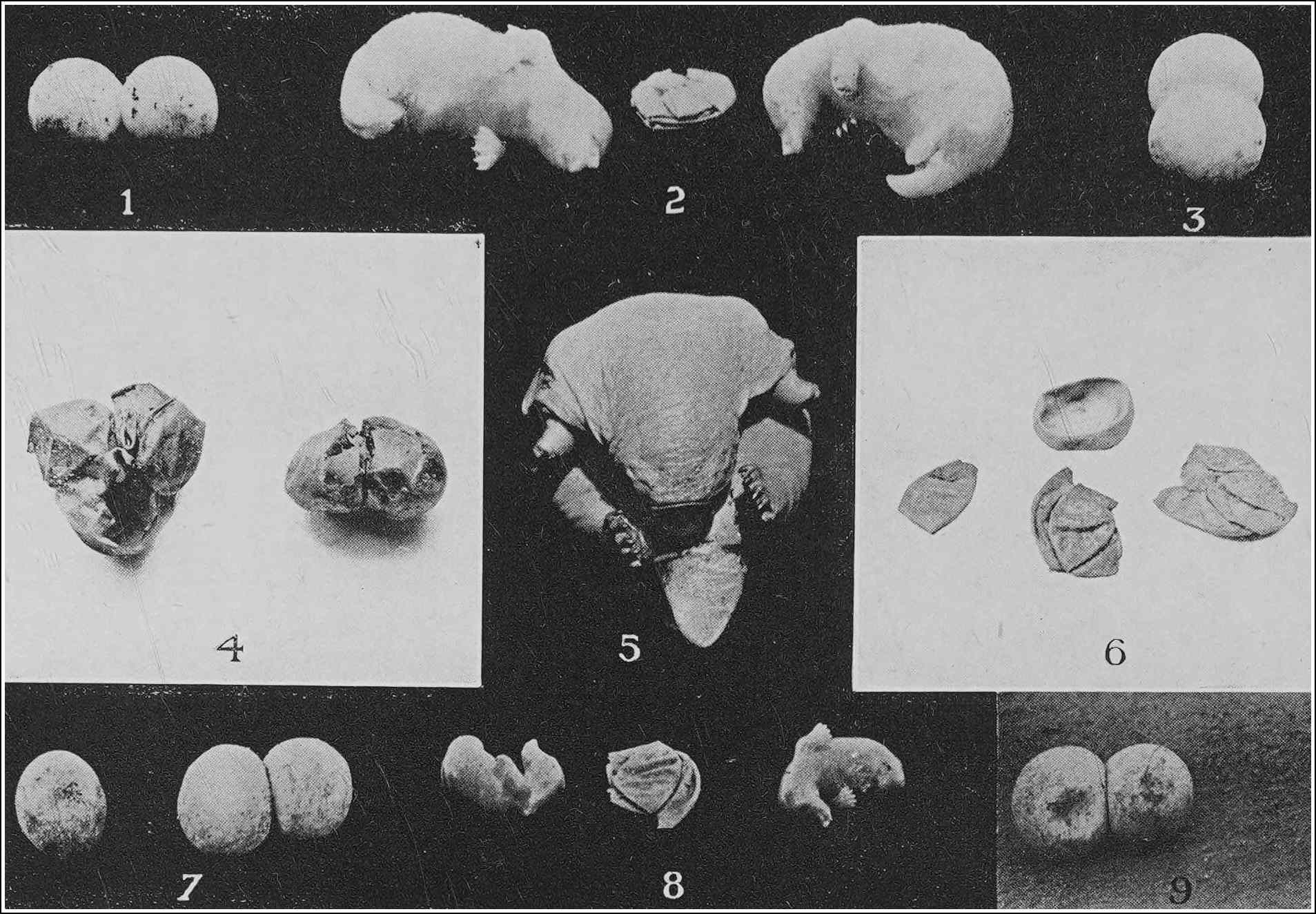
1 and 3. Eggs, new-laid; 2. Week-old young and egg capsules; 4. Relaxed egg capsules (triplets and twins), showing adhesion, also rents made by young in hatching; 5. Young, about fourteen days old; 6. (above) Dried-up egg, (below) single, twin, and triplet collapsed capsules; 7 and 9. Eggs, partly incubated; 8. Day-old young, and capsules from which hatched.
(About two-thirds natural size)It is of some interest to note in passing that on the previous day I had suffered a defeat only fifty feet away from this burrow. Opening up a burrow, I came to a pug. When this was opened it broke into a rabbit-burrow, and was discarded as a ‘duffer.’ The following morning, on examining the excavation again, I found a freshly-dug platypus-shaped hole in the side of the trench, and, following up this clue, came upon a finished but empty nest, its tenant having deserted it during the night. Such a congestion of tenanted burrows is, however, quite unusual, and in this case was due primarily to the fact that the ordinary conditions of the season had been considerably upset by freshes in the river. I have since found several platypus breeding in the same bank side by side, but not from choice nor because of a neighbourly disposition. Scarcity of suitable soil is the main reason for overcrowding, but rabbits and even the European rat are invading these ideal water frontages and are ousting Ornithorhynchus from his birthright. Otherwise, as I have explained before, breeding platypus prefer “solitary confinement.”
To describe a number of burrows in detail would not serve any good purpose, for they are too variable to be sorted into types, and each seems to be constructed according to the vagaries of its builder. One burrow which I examined at Manilla on 22 September 1920 had an entrance four feet above water-level in an almost perpendicular bank. It was opened up and followed for a distance of twenty feet. 136and came to an end just as it was tending back towards the river. The nesting-chamber was found five feet from the end, but was placed in a very unusual situation, twelve inches above, and just to one side of, the tunnel, so that it was approached by a short lead from below. As this passage was securely pugged, the nest was extremely difficult to locate and might easily have been overlooked. However, since then. I have found a similar burrow in New England, thus proving that the first find was not unique.
The platypus invariably begins its burrow above water-level, and the presence of other entrances, whether above or below water, is due to variations of that level. I at first thought that some of these additional entrances might be of the nature of air-shafts, but found that the pugging of the burrow would prevent their being of any use for such a purpose. Kershaw, too, wonders how the animal and its young obtain sufficient air-supply, plugged in as they are in an almost air-tight chamber. The resting-burrows are often provided with two or more adventitious entrances, which may perhaps serve as apertures for ventilation; but additional entrances to the breeding-burrow could not serve this purpose.
Occasionally the entrance to a breeding-burrow is at a considerable distance from the water; I have found one as much as forty feet from the river, and fifteen feet above its surface—the river being at the time two feet above normal level.
I have also found burrows coming to the surface on the river side of a sheep-track, and disappearing into the ground on the other side of it. Kershaw (1912, p. 103) noted one such, in which the narrow ridge of ground separating the two holes was worn smooth, as if the animal passed up, came out of one, and immediately entered the other. I think the reason is that stock using an accustomed track may annoy the platypus by bringing down her roof, until in exasperation, she abandons the section of burrow underneath.
My opinion is that the vagaries observed in breeding-burrows are due to the nature of the ground. Obstacles 137such as roots, stones, and rabbit-burrows—or, more important still, patches of friable soil which tends to cave in—force the female to continue her efforts until she finds a suitable position for her nesting-cavity. As I have said before, the faulty nature of the bank often leads to the abandonment of a burrow.
As to the question of a sufficient air-supply, I find that in the case of the very young little air is required up to the age when they begin to take milk. Seeing that the mother is, up to that time and until disturbed, always found in the pugged-up cavity with her eggs or her young clamped to her abdomen,[14] it is evident that the only air obtained is that which percolates through the several pugs arranged here and there throughout the tunnel. From tests made with very young platypus thus taken direct from the mother, I find that, the younger the nestling, the longer it can remain under water.
At Manilla, on 3 September, 1925, I unearthed a female platypus nursing twins. The temperature of the outer air was 70 deg.; of the nesting-cavity (containing young) 68 deg.; the river water 62 deg.; of the mother (cloacal) 82 deg. The young were apparently about three days old and measured 28 mm. from tip to tip. The mother’s cloacal temperature would, I think, correspond approximately with her body heat when coiled up around the young. Immediately after being taken from the nest, the twins were put into a bottle containing river water; yet one of them lived for three hours; the other survived for three and a half hours before drowning; and both would probably have survived much longer but for the shock caused by the sudden change of temperature from 82 to 62 deg. In other cases, when drowning young platypus taken for scientific purposes, I have always tried to be sure that they were dead before preparing them for pickling; but on several occasions, when they have been placed in wadding for the removal of moisture and while I have been engaged on other work, they have been found crawling about and have had to be “redrowned.” This is on all fours with the 138fact that flies, apparently drowned and then placed in the sunshine, recover their vitality.
On an earlier occasion (3.35 p.m. on 2 October 1920) in bitterly cold weather I took twin young ones measuring 40 mm. from a nest at the Macdonald River and put them into a bottle of about 6 2½ inches, into the bottom half of which I pressed a handkerchief tightly, so that they might not crawl within its folds. The bottle was then securely corked and carried to camp in a billy-can—four miles over a rough bush track on the floor of a stiffly-sprung van. On arrival I was surprised to find them alive, but they were apparently asleep, snugly coiled. On my flicking the bottle with my finger, both uncoiled and pawed the air, only to subside again. This performance was repeated half-hourly for 5½ hours, until, being tired after a hard day in the field, I decided to chance results, wrapped the bottle in a chaff-bag, and retired to my canvas sleeping-bag until morning. At 6.40 a.m. I found them asleep, and when the bottle was uncorked they were as lively as when collected. Three hours later they were “snap-shotted” with the shells of their eggs (see Plate 23, fig. 8), and, as I had another busy day ahead, I concluded the observation by dropping them into a bottle of fixative. During the day I unearthed a set of triplets of the same age as the twins. These, with a portion of the nest, were placed in a loosely-woven calico collecting-bag. After being several times exposed to light and air, they were found dead at the end of six hours.
Tests made with young at various ages prove that, from the time when they begin to consume milk, the time required for drowning is gradually lessened. I have observed that, during the period when the mother has to replenish her food and milk supply, the pugs, through being continually removed, are less compactly re-arranged. Fresh air is, therefore, introduced into the nesting-cavity in graduated supplies, and this would indicate that the young require more air in proportion to their growth.


139
The platypus is confined to that part of Australia which lies east of 138° E. long., and to Tasmania. It has not so far been recorded from the Cape York Peninsula north of 15° S. lat.; but this country is little known, and it is not improbable that the animal will be found there when the rivers are examined. The most northerly rivers from which we have definite records are the Mitchell and its tributaries on the western side, and the Barron on the east. A number of rivers farther to the north—such as the Coleman, Archer, and Batavia, running into the Gulf of Carpentaria, and the Normanby and Kennedy rivers entering Princess Charlotte Bay—are probably suitable as habitats, and may in time be found to contain Ornithorhynchus.
The first record of the platypus in the Gulf rivers is given by Armit (1878, p. 413). He observed one swimming in a large water-hole 150 miles west of Georgetown on the road to Normanton. It is not clear whether this water-hole was part of a river bed; but it could not, in any case, have been far away from the Gilbert River, along which the road runs. He also stated that platypus occurred in the Leichhardt River, which is the most westerly record. Waite (1896) published records from the Norman River at Normanton on the Gulf side, and from the Barron, Herbert, and Burdekin rivers on the east coast. Through the kindness of Mr. H. A. Longman, of the Queensland Museum, Mr. A. H. Chisholm, and a number of other correspondents, I have gathered a considerable series of records for Queensland waters. The platypus is exceedingly plentiful in the upper waters of the Mitchell and its tributaries, in the Barron, and the Herbert, especially on the tableland. It is also common enough in the small 140creeks draining into the Herbert below the range. It would seem to be almost equally plentiful in all the chief coastal river systems to the south—Burdekin, Fitzroy, Burnett, and Mary—although it is not so frequently seen in the more closely settled portions of the river valleys. No platypus has ever been seen (so far as I can ascertain) in the Diamantina and Cooper’s Creek, which drain into the interior; nor can I find any records for the Paroo and Warrego. They are found, however, in the Condamine, Macintyre, and Dumaresq rivers, and a tributary of the last is named the Mole River from the occurrence of the duck-mole in it. It was, indeed, on this stream that Caldwell collected some of his material. Mr. Longman mentions that his Museum has records of platypus from the Brisbane district in the early days, but none of recent date.
In New South Wales Ornithorhynchus is found in the upper waters of all the rivers draining to the east coast, and even in small creeks such as Ourimbah Creek, north of Gosford. It is especially numerous in the trout streams of the southern Alps, both those draining into the Murray system, and those running southwards into the Snowy. Inland, the records cover the upper waters of the Gwydir, Namoi, Castlereagh, and Macquarie in the north, but not the Bogan. In the south, the upper waters of the Lachlan, Murrumbidgee, and Murray still hold a goodly store. It will be remembered that the first platypus was discovered in a lagoon off the Hawkesbury. Two correspondents of the Sydney Daily Telegraph mention the occurrence of the animal in swamps and ponds not directly connected with streams. Mr. C. R. Stranger, writing from Curlewis, states that, when the great drought broke in June and July of 1920, the Curlewis swamp, which had been dry for several years, filled. Soon afterwards a platypus was seen in the swamp, and was there for a considerable time, always about the one spot. The swamp was four or five miles from the nearest stream, the Mooki River, though the two waters may have been connected by a backwater during the flood. Mr. George Boyd has often seen platypus in swamp lagoons of a permanent nature, situated in the neighbourhood 141of Wyong Creek but not connected with it except in times of flood. A third correspondent describes the finding of a platypus, after a thunderstorm, in a small water-hole in the rocks near the top of Black Mountain, a few miles from Dundee, and three or four miles from the nearest permanent water.
Krefft in his Catalogue of Mammals in the Australian Museum (1864) records three females in spirits from the Botanic Gardens; but these, if taken from the ponds, must have been previously released in them. He also records an adult male from North Shore, Sydney, and a female from George’s River, from both of which places they have long since disappeared.[15] The upper reaches of the Nepean and its tributaries are, however, still tenanted, so that the platypus still occurs fairly close to Sydney.
The Victorian records include most of the southern tributaries of the Murray system, such as the Campaspe, Loddon, and Avoca rivers. The Snowy River, to the east, has already been mentioned. Kershaw made his observations upon the Hopkins in the west, and there are records from the Glenelg, close to the South Australian border. Mr. Noel Learmonth, writing to the Australasian (30 June, 1923), comments on the fact that the platypus is absent from the Eumeralla, Darlots, and Fitzroy rivers, though inhabiting the Wannon on one side and the Hopkins on the other. Mr. E. R. Waite, Director of the Adelaide Museum, has kindly supplied the known South Australian records—the Murray River at Murray Bridge and Tailem Bend, the Onkaparinga River, and the Glenelg, which just crosses into South Australia close to its mouth.
For information as to the distribution in Tasmania, I am indebted to Mr. Clive Lord, Director of the Tasmanian Museum and Art Gallery. He writes:—
“This species is generally distributed throughout Tasmania. From personal observation I can state that it can be found in most of the inland lakes that are not close to the settled areas. 142In certain of the National Park lakes (Mt. Field—20 miles NW. of Hobart) the platypus is numerous, and, as the area is an absolute sanctuary for our native fauna, it has a chance of living there in peace; but, despite the fact that the platypus is totally protected by law, they are very largely taken in other parts. At the Great Lake large numbers were captured for their skins, but we could not get a conviction. During the recent Christmas holidays I saw numbers in the lakes in the Cradle Mountain and Barn Bluffs district (NW. Tasmania). Along some of the rivers of the NW. coast the platypus is still to be found; but, generally speaking, the advance of settlement is reducing the number in the rivers, and the mountain lakes will be its stronghold in the future. There are numbers in Lake St. Clair, and, as previously noted, they frequent even the highest alpine lakes, over 3000 ft. above sea level.”
This sketch of its distribution shows that Ornithorhynchus is still widely spread throughout the eastern portion of the mainland and Tasmania, and is in no immediate danger of becoming extinct provided present laws are enforced. It is very probable that closer observation would discover its presence in many streams where its existence is wholly unsuspected, and it is remarkable that it has not been recorded from main rivers such as the Darling and Murray and the main channels of their larger tributaries, such as the Lachlan and Murrumbidgee. Although the creature seems partial to high places, it cannot be influenced altogether by altitude, for it occurs commonly throughout the low creeks and rivers of the coastal plain. Nor is rapidly running water essential, since the Tasmanian lakes are so well stocked. Certainly the platypus prefers clean, clear water, but it manages to survive quite comfortably in rivers like the Mary and the Burnett, which are far from being limpid streams. This, however, may be a matter of necessity, not of preference. It is probable that it finds a greater abundance of the food that most delights it in the more rapid waters of the upper stream. It may be, too, that the occurrence of large Murray cod (Oligorus macquariensis) 143in the larger rivers of the plains has something to do with the matter. The platypus, foraging blindly under water, or floating idly at the surface, would have no protection against these voracious creatures.

The hind legs are kept stationary to balance the buoyant body (or, to use a nautical phrase, to maintain an even keel). The one on the right is touching the sand with its muzzle, its right paw is fully extended, and the left is being brought forward for the next stroke. The finished stroke is shown by the position of the fore-paw (close to the body) of the one swimming on the left.
Plate 25The antiquity of the platypus, and the changes in river systems during the ages, render it probable that the animal inhabited at one time or another all the waters of Tasmania and of eastern Australia. Closer settlement is very likely responsible for its disappearance from certain rivers of the (Australian) coastal plain, and, as that area is not now subject to general flooding, it is hardly to be expected that the denuded rivers will ever be replenished with platypus by natural means.
That the platypus does travel over land is quite satisfactorily determined. But how far it can travel is not known. During floods it has to move about in search of food, and usually works towards the top of billabongs and backwaters, where supplies are more easily obtainable. As the waters recede, it is often left isolated in some deep water-hole, where it may continue to live happily for a time. Ultimately, however, it makes back to the main water, either by following the channel, or, if the river be nearer by another way, by cutting directly across country. I have observed this happening on many occasions, and have no doubt that the platypus possesses a highly-developed sense of direction, by means of which it makes straight for water. Animals released close to, but out of sight of, water, never hesitate; they make straight off towards the water closest at hand. In corroboration I quote the following paragraph from the Northern Daily Leader (Tamworth, N.S.W.) of 6 April, 1922:—
“The duck-billed platypus at times shows a remarkable knowledge of locality. I carried one in a bag from its native river, where it was caught on a night-line set for eels, to a shallow lake about a mile away. It was then gently slid into the water, but promptly swam ashore and started on the long crawl, straight for the home river. The duck-bill was recaptured and returned to the lake, which, it may be remarked, had 144no banks suitable for a platypus burrow. But again the animal swam out and made tracks for the old address. Eventually the beast was put far out in the lake and left to its own devices. A few days afterwards it was found dead in a patch of scrub about half-a-mile from the river in a direct line with the pool from which it was taken.”
The observations of correspondents indicate that the animal can cover a land distance of four or five miles, and may cross intervening high land. Since it has considerable powers of endurance, and, despite its voracious appetite, can survive without food for comparatively long periods, it is possible that it has managed to contrive its own distribution by direct means.
Platypus have often been observed working in a general direction either up or down stream. These movements are almost certainly influenced by food conditions. A migration of platypus was observed by the late Mr. William Hill in 1859, when he was manager of the Pallamallawa cattle station (now a township) on the Gwydir River. About fifty aborigines, under ‘King’ Binamoore, were camped on the river-bank not far from the homestead. One evening Mr. and Mrs. Hill strolled down to the camp for a yarn with Binamoore, who was an old friend, but the chat was interrupted by a sound from up river, similar to that made by a mob of cattle fording a stream. Always on the alert for movements of his herd, Mr. Hill, accompanied by his wife, walked down to the water’s edge to investigate. The river at the time was low, but still flowing, with occasional deep holes, connected by rapidly running shallows. Presently the noise was heard again, this time closer at hand, in fact so close that Binamoore and his tribe, and the tribal dogs, took fright, and bolted off to the next station, deserting their gunyahs. Mr. and Mrs. Hill then clearly observed a mob of platypus all swimming together at top speed with the current, and estimated that there were at least a hundred of them. There could be no doubt about the identification, as the animals were frequently obliged to expose themselves in climbing over obstructions in the shallows. On 145the following morning only an occasional platypus was to be seen, floating at the surface or swimming about in a leisurely way. I have every confidence in my informants, each of whom has repeated the story to me several times. I know of no other record of any mass movement of platypus, and my explanation is that the mob had gradually congregated from streams in which food supplies had been affected by flood, and were following the main channel in search of further supplies.
To anyone knowing the rivers and lakes mentioned in the records of distribution it will be obvious that the platypus inhabits a varied range of waters, from clear, icy, rapid alpine streams of the southern Alps to warm, turbid waters of the Queensland coastal plain, and from large lakes to small waterholes. Despite this apparent catholicity of taste there are certain conditions which determine the parts of these various waters chosen by the animal. The chief is that of abundant food supply.
Semon (1894, p. 10), who made careful observations on the Burnett, writes:—
“Ornithorhynchus anatinus inhabits the banks of running water within the area of distribution. It follows these from their source of origin down to the plains. The main condition for its occurrence is the scooping out of comparatively expansive depressions in the bed of the river, in which the water flows slowly, so that fine silt collects on the bottom and a vegetation of aquatic plants can develop, which serves as a resort for innumerable aquatic creatures, worms, crustaceans, larvae of insects, and molluscs. Here the Duckbill seeks its food, and hides from its hunter by diving. Here it can remain in periods of extended drought, which dry up the rest of the river bed, in a small amount of water. At such times those Duckbills which live in the smaller waterholes, so soon as the level of the water sinks too low, wander out into larger holes which are less likely to dry up. Then one notices an increase in the latter, and a coming together of the platypus folk.”
My own observations in the Manilla and New England 146districts confirm this statement. There the streams are partially blocked at frequent intervals by granite boulders, and a succession of quiet pools is formed between stretches of rapidly running water. In these pools the platypus finds its food. When freshes come down the river, however, coarse sand and gravel are deposited on the silt bottoms, and for a time the feeding-grounds are spoiled. At such times the animal is forced to seek for food where it may be found, and generally works its way upstream. After prolonged floods the animals are often found in considerable numbers dead upon the banks. This would seem to indicate that they had starved to death, but it must also be borne in mind that they dislike turbid water, and a continuance of disturbed conditions may have some physical effect upon them.
Heat and cold appear to have little effect, since the platypus seems equally at home in the warm streams of tropical northern Queensland and at a height of 6000 feet on the Kosciusko tableland—which, though not continuously snow-clad, nevertheless experiences falls of snow during the summer.


147
The early records contain little in the way of precise information as to the general habits of the platypus. Bennett (1860, p. 95) complains that, as long ago as 1829, “notwithstanding all the inquiries I made of persons long resident in the colony, I could get no correct information; I found then, as I have found during an extended residence in the colony, that the majority preferred forming theories of their own, and arguing upon their plausibility, to devoting their time to the collection of facts.” It is the same to-day.
Bennett went out to seek his first platypus at sunset, “knowing, as I did, the crepuscular nature of the animals,” and it was not long before his companion directed his attention to one on the surface of the water, not far from the bank on which they were standing. He writes (loc. cit., p. 104):—
“In such circumstances they may be readily recognized by their dark bodies just seen level with the surface, above which the head is slightly raised, and by the circles made in the water around them by their paddling action. On seeing them, the spectator must remain perfectly stationary, as the slightest noise or movement will cause the timid creature instantly to disappear, so acute are they in sight or hearing, or perhaps in both; and they seldom reappear when once frightened. By remaining perfectly quiet, however, when the animal is paddling about, it is possible to obtain an excellent view of its movements on the water; it seldom remains longer than one or two minutes playing on the surface, but dives, and reappears a short distance above or below the place at which it was observed to descend. Although the animal may ‘come up’ close to the place where the sportsman is standing, it would 148be useless to attempt to level the gun, for that action alone would cause its instantaneous disappearance; but, after waiting patiently until the animal dives, and watching the direction in which it sinks, preparation must be made to receive it with the discharge of the piece instantly on its reappearance at the surface, which (when it descends unfrightened) is almost certain to take place in a short time.”
And again on p. 108:—
“These creatures are seen in the Australian rivers at all seasons of the year, but are most abundant during the spring and summer months, and I think a question may arise whether they do not hibernate. The best time for seeing them is early in the morning, or late in the evening. During floods and freshes, they are frequently perceived travelling up and down the rivers: when going down, they appear to allow themselves to be carried by the force of the stream, without making any exertion; but when swimming against the current, their muscular power is exerted to the utmost to stem its force, and generally with success. I recollect, however, seeing two make repeated and ineffectual attempts to pass a small waterfall in a rapid part of the river, and, after many persevering efforts, they were unable to attain their object. The opinion that so generally prevails that these animals must be shot dead instantly, or otherwise they would sink and not reappear, I did not find from my own observations to be correct.”
These observations are of special interest, since they were made as long ago as 1829 in a district which had but recently been settled, so that the platypus could not have had time to alter its ways on coming into contact with Europeans. More than sixty years later the distinguished German zoologist Richard Semon spent some time on the Burnett River, where he reports that the platypus was very plentiful and was not molested by the white colonists or hunted by the aborigines. His account of its habits agrees in all essential particulars with that of Bennett. In the English translation of his popular account of his journey (Semon, 1899) will be found a summary of his observations; I prefer to give a translation from 149his paper in the Forschungsreise, which is somewhat fuller. He writes (p. 10):—
“On the Upper and Middle Burnett, where alone I observed the animal, it was plentiful everywhere in suitable localities along the river. In Ornithorhynchus a considerable preponderance of males over females was observed, so that for each female captured there were two or three males. As in Echidna, the fully grown male is larger than the female. Like Echidna, too, Ornithorhynchus does not lead an exclusively nocturnal life. More than once I saw the animals swimming in the river in daylight, searching for food; yet this could be termed exceptional. It is during the twilight of morning and evening that the animal leaves its home and passes to the water. Generally they turn to the river shortly before sunrise, and remain there till the sun has completely risen. In the evening it is the same—from a short time before sunset to a little while afterwards, a period of twenty to thirty minutes. This holds good for the winter months, from June to the beginning of September, when the nights are cold and the thermometer in the mountainous regions of the Burnett is almost at freezing point. One is then sure to meet with plenty of Ornithorhynchus at suitable places both morning and evening; and now and again to see them in broad daylight. This is changed, however, when the Australian summer begins. The days become hot, and the nights warm. In the middle of September, during 1891 and 1892, I very seldom found an Ornithorhynchus in the river during the morning and evening hours, and still less frequently by day. Doubtless they then use the nights for these excursions, for they must enter the water to obtain their food. I took great pains actually to observe the animal in the river by night. I did not succeed, however, nor even in bright moonlight. Since, however, Ornithorhynchus lies flat like a plank in the water, scarcely projects beyond the surface, and is absolutely noiseless in diving and reappearing, one would need an exceptionally favourable occasion to perceive and observe the animal by night.
150
“In the cool part of the year, on the other hand, I could observe the behaviour of the animal in the river during dawn and during daylight as long as the sun was low. As has been already mentioned, Ornithorhynchus lies quite flat in the water, with only the flat head projecting, and that very inconspicuously. Usually the animal lies motionless at the surface. After a few minutes it dives suddenly and noiselessly, stays under water two or three minutes, and comes up again just as suddenly, and just as noiselessly. During its dive it has collected by means of its flat bill, after the manner of a duck, all sorts of aquatic life, and richly filled its cheek-pouches. The reappearance takes place in order that it may get air, and chew and swallow the contents of these pouches....
“If an animal is frightened while in the water, it dives immediately, and does not reappear. Although it is a good diver, it can only spend a certain amount of time under water. Those which become accidentally caught in fishing-nets at night, and are held fast for a long time under water, are always found drowned next morning.
“The pursuit of the animal is not difficult if one knows its habits. Small as is its eye, and deeply buried in the fur as is the opening of the ear, yet sight and hearing are distinctly sharp; it is therefore useless to begin matters by trying to creep up while the animal is at the surface. The position of the eyes enables it to observe exactly what is going on on the rising banks above it. It does not recognise danger, however, unless the observer moves. But the mere lifting of a gun is sufficient to scare the animal away. Every dubious noise causes it to disappear.... Once scared off, the animals nearly always seek their homes, and do not reappear during that same morning or evening. And yet, it is easily taken if one approaches while it is under water, and at once stops motionless when it appears.”
These accounts of two very competent observers, both made in districts where the animal had not yet been disturbed or harassed, show that the platypus has not developed its alertness and timidity since the advent of man. The same remark applies to its predominantly crepuscular habits. It has sometimes 151been erroneously claimed that these have been adopted to escape from man and his gun. It must also be remarked that the months during which Semon noted occasional animals in the water by day are those of the breeding-season. At that season, when opening up burrows containing suckers, I have frequently found that the mothers were absent. It seems certain that they are forced to feed at irregular times during the nursing period, and that this is the principal reason for their being found in the river in broad daylight.
As to the preponderance of males noted by Semon, here again we must consider the time at which the observations were made. Wilson and Hill (1908, pp. 33-4) state:—“During the breeding-season, however, the pregnant female appears to keep much more closely to the burrow, so that one may then commonly enough shoot five or six males to one female.” As far as my observations go, the sexes seem to be numerically about equal.
A few other earlier observations are of interest. It is worth remembering that Charles Darwin (1845) saw the platypus with his own eyes. On January 19, 1836, while staying a day at “a farm called Walerawang,” he had the following experience.
“In the dusk of the evening I took a stroll along a chain of ponds, which in this dry country represented the course of a river, and had the good fortune to see several of the famous Ornithorhynchus paradoxus. They were diving and playing about the surface of the water, but showed so little of their bodies that they might easily have been mistaken for water-rats. Mr. Brown shot one: certainly it is a most extraordinary animal; a stuffed specimen does not at all give a good idea of the appearance of the head and beak when fresh, the latter becoming hard and contracted.”
Crowther (1879, p. 97) gives the only detailed description of its swimming motions known to me:—
“When swimming, the fore-paws are widely expanded, being convex forwards, propelling themselves by an antero-lateral movement. The hind paws and tail take but little share; the former have a slight 152lateral horizontal movement, the tail lazily moving with the body. When diving, the head is rapidly thrown beneath the body, front paws quickly moving until the bottom is arrived at, when they are seen turning rapidly over with their bills the sand, and even large stones.... A train of small bubbles proceeding from the nose marks the exact course the animal is taking.”
Crowther also states that the tail is the first part to reach the surface of the water.
Allport (1878, pp. 30-31) also watched ‘a large specimen, and one of that variety which has very red fur on the sides’ feeding over a gravelled bottom in the clear waters of the Mount River (a tributary of the Huon) in Tasmania; “his method of proceeding was to burrow his head and more than half the body amongst the stones, causing a cloud of sediment to rise at each dive.... He never remained under water longer than a trifle over half a minute, and stayed on the surface between the dives rather less than half a minute.” Allport also records that a large number of small bubbles were continuously emitted, and that the animal fed for three-quarters of an hour, and then went off to his burrow. I also have observed platypus feeding in clear water, and have seen them tear stones aside with their claws, or lever them up with their powerful bills and shoulders, to get at food underneath; but bubbles are certainly not emitted continuously while the animal is under water, as all platypus-hunters know. When it dives in alarm, the breath is apparently held, and no tell-tale bubbles issue. The first bubbles that come to the surface, after the animal dives, are not emitted by the platypus at all, but are formed by the imprisonment of air when the bill is suddenly submerged, and, as the body turns downwards, the bubbles traverse the contour of the back and break away at the rump to ascend rapidly to the air. This was formerly believed to be escaping breath; but as the animal, before sounding, undoubtedly fills its lungs to the utmost capacity so that it may remain under water, if need be, for several minutes in search of food, it is 153not likely to exhale until compelled to do so, and then only in brief spasmodic puffs.
From the sum of these observations, and without drawing further upon my own, we may safely conclude that the platypus is normally crepuscular, and was so before the advent of European man; that it resorts to the water twice daily to feed, and remains there for a period of from twenty minutes to three-quarters of an hour, perhaps longer when food is scarce; that the sum total of its aquatic life amounts to less than two hours out of each twenty-four; that it is so wary as to disappear for the day at the first sight of some unaccustomed movement on the river-bank, or the hearing of some unaccustomed noise close at hand; that while feeding undisturbed it remains at the surface for a period variously estimated at from rather less than half a minute to one minute, for the purpose of chewing and swallowing its catch, and then dives to collect more food, remaining under water for a similarly varied time; that in feeding it burrows with its sensitive, tactile muzzle among mud and stones, sometimes burying more than half its body, and thrusting fairly large stones aside with its powerful shoulders; that the greatly webbed fore-paws are the chief swimming-organs; and that, when floating at the surface, it lies flat, with the body just submerged except for the upper part of the flattened head and the middle of its back and tail, though occasionally it will be seen as in Plate 5, fig. 2.
Most of those which have occasionally been observed during the daytime (especially during the breeding-season) are undoubtedly females; but in well-shaded pools it is not uncommon to see individuals of both sexes abroad during the day. The time spent in feeding is bound to vary according to the supply of the invertebrate fauna and other commodities which constitute its food at that particular period of the year. Where food is plentiful, an adequate meal can be collected quickly, and the time required may be no more than Semon’s minimum of twenty minutes. The gravel bank referred to by Allport would not be so productive as a silt-bottomed water-hole, full of aquatic plants, in the Burnett, and 154it might easily take three-quarters of an hour to obtain a full meal. The nature of the food itself has been variously reported. For the Burnett, Semon (1894, p. 11) reports that the main food consisted of molluscs, and that the cheek-pouches were usually found to be full of immature individuals of a bivalve, Corbicula nepeanensis, 10 to 15 mm. in length. He mentions in addition, however, worms and insect larvae. Bennett (1860, p. 113) writes:—
“On examining the cheek-pouches or the stomachs of these animals, I always observed the food to consist of débris of insects of the family Nauceridae, very small shell-fish, etc., which were constantly found comminuted and mingled with mud or gravel. This latter might be required to aid digestion, as I never observed the food unmingled with it. The natives say that they also feed on river-weeds; but as I have never seen that kind of food in their pouches, I cannot confirm the correctness of the statement.”
Allport (loc. cit.) states that the food found in the cheek-pouches consisted of larvae of caddis-flies; Crowther found freshwater shrimps, water-fleas, and hard black beetles.
Wood Jones (1923, p. 55) notes that Bennett recorded the feeding of captive animals on freshwater mussels; but the mussels were broken up before they were given. From the nature of the masticatory apparatus he argues against shell-fish as a diet, and he concludes that common river-shrimps may be regarded as the staple article of diet. Since such thoroughly competent observers as Semon and Bennett have both actually found molluscs in the cheek-pouches, these cannot be put out of consideration as food. There is, moreover, a great deal of difference between the ‘very small shell-fish’ which Bennett mentions, and the large swan-mussels which were probably (though it is not specifically stated) the kind given in broken form to the captives. All observers agree that the platypus finds its food by nuzzling in the mud or gravel of the river-bottom after the manner of a duck. When it is remembered that its eyes are kept closed while beneath the water, it becomes extremely unlikely that the 155animal should catch such active creatures as freshwater shrimps except very occasionally. Crowther certainly mentions shrimps; but he probably refers to crustacea like the curious and archaic Anaspides, which is found in Tasmania among stones on the river-bottom, and which runs rather than swims.
The food of the platypus must necessarily vary in different places and at different times of the year, limited as it is to the invertebrate fauna present in the water at any given spot. It consists of immature molluscs, aquatic worms, the aquatic larvae of many insects such as dragon-flies, caddis-flies, may-flies, and the like, the larvae and perfect insects of groups such as the water-beetles and water-fleas—which are wholly aquatic—bottom-feeding water-bugs, and such crustacea as inhabit the bottoms of streams. This food is taken in with a certain quantity of mud or sand, which, as Bennett has noted, appears to be absolutely essential for the purpose of mastication. The living prey is killed while held in the long horny ridges situated in the anterior part of the mouth-cavity, and is passed rapidly backwards, being prevented from reaching the gullet by the bulbous hinder end of the tongue, and directed to one side or other by the tooth-like projections at the base of the bulb.
I cannot agree with Crowther’s observation that the tail is the first part of the animal to reach the surface. Apart from the inherent improbability that so cautious an animal should expose its blind end first, my observations, both in the open and in a tank in captivity, show that the tip of the muzzle is always the first part to appear, followed by the top of the head. The eyes immediately open, and, if there be no suspicious sight or sound, the rest of the body is allowed to float towards the surface, beneath which, however, it usually remains almost submerged. The animal floats in a spread-eagled fashion, with its webbed hands moving slowly, palms upward, to maintain its position, and causing the rings of ripples to which Bennett refers. Semon’s comparison to a plank is not very happy, nor do I concur in his statement that the animal usually lies motionless at the surface. Bennett (1860, p. 106) puts it thus:—
“When the fur of the Ornithorhynchus is wet, it has a sordid156 and far from attractive appearance, resembling rather a lump of dirty weeds than any production of the animal kingdom; indeed, were it not for their paddling motion on the water, these creatures would often escape observation; for their suppleness and colour, when wet, would cause them to be regarded only as masses of weeds, which are so often seen floating about the rivers.”
At the surface the animal chews the results of its catch in a leisurely manner, its jaws working laterally like those of a cow when chewing its cud; having swallowed the contents, it dives in search of more. In diving, the head is first immersed by bending the anterior half of the body downwards and almost simultaneously humping the back. The head is then flexed violently against the chest, giving a very powerful downward thrust comparable with that produced by a crayfish through the sudden flexure of its abdomen. This carries the body below the surface, a process which is aided by an upward thrust of the muscular tail. The webbed fore-paws are used alternately, the web being arched in the manner described by Crowther; the feet and tail trail behind. The animal swims in a determined fashion, at a steady but not fast pace, to the bottom, which is first touched by the tip of the muzzle. The fore-limbs are now laid back beside the trunk, and the webs are kept beating upwards, to hold the animal at the bottom. The body is strongly arched, the sides of the tail curve inwards so as to produce a concavity of its lower surface, and the end of this concave surface is applied to the bottom as if helping to keep the platypus down. In this somewhat constrained position it ‘bobs’ along the bottom of the river, testing every part with its tactile muzzle. It has every appearance of having to work hard against a natural tendency of the body to float to the surface. After two or three minutes it raises its head, relaxes the tense attitude of its body, immediately begins to float upwards, and hauls itself to the surface hand over hand by means of the webs of the fore-paws. If suspicious or frightened, it may merely show enough of the tip of the muzzle to expose the nostrils, take in a new 157supply of air, and dive again immediately. When the animal is undisturbed, the dive is practically noiseless; but when it is alarmed, there is often a distinct double splash made by the almost simultaneous strokes of head and tail, one downward, the other upward.
The swimming motions described above are those employed during feeding-time. When the platypus is definitely on the move, it swims fairly rapidly at the surface, and frequently raises its head out of and parallel with the water, so that it looks rather like a duck. Both myself and the late Charles M. Hoy have on several occasions noticed another kind of progression in the water. The naturalist’s way of collecting platypus is not to shoot it, but to place a bullet from a heavy rifle beneath it when the animal is floating. The concussion from the impact is usually sufficient to kill it out-right; but sometimes it is only temporarily stunned, and, on coming to, makes for the bank, where it can be easily secured. When a platypus has been temporarily stunned by concussion, and then recovers, it can almost be declared that when forging for the bank, it actually crawls along the surface of the water, for the fore-paws can sometimes be seen clearly above the surface, which the tail frequently lashes. On such occasions, two of which I have witnessed, the pace is decidedly fast and noisy, while the water is beaten and churned considerably by the action. Hoy, writing to me from Ravenshoe in northern Queensland on September 3, 1920, describes an uninjured male doing much the same thing:—“Saw male and female evidently copulating, and secured female—left ovary very much enlarged. Although usually very shy, the male came to the surface and swam up to within three feet of me, while I was taking the female from the river. He elevated his head from the surface of the water, splashing vigorously with his limbs and tail and taking on a very pugnacious attitude.” It is worth noting here that this particular male persisted in his charge until he was within three feet of Hoy, although the latter tried hard to frighten him back by throwing up his hands. Hoy admitted to me later that 158he was afraid of being spurred by the “hissing” ferocious creature. On the strength of this authentic observation, made in broad daylight, and of others that I have made at dusk, I am quite convinced that this method of swimming is also indulged in during nocturnal frolics. Apart from the actions of the infuriated male encountered by Hoy, platypus cautiously submerge themselves to the extreme during the daytime; but this is not the case when darkness sets in, principally because their vision then becomes acute. The late Geoffrey Smith (1909, p. 124) has observed this mode of swimming, and writes:—“I was surprised at the extraordinary tameness of the Platypus, several of which swam quite near the boat with the whole of their body exposed above the water and shining with a curious grey appearance in the sunlight.” This occurred at Mount Read, in north-western Tasmania.
Semon has observed that a platypus which has been shot and killed invariably floats high out of water. I can substantiate his statement, and have observed also that the submerged animal has to work continuously to keep itself down. Just how it manages to float with the body submerged in the way described above I am not certain, but the slight motions of the hind limbs and tail probably aid in this act. The swimming stroke of the fore-paw is outwards, backwards, and upwards, and ends with the hands palm-upwards against the sides of the body. When performed slowly and gently, this stroke would serve to pull the fore-part of the body down in the water. As the limb is thrust forward again, the resistance of the water keeps the web folded on the palm, until it is voluntarily extended to begin a new stroke (see Plate 25).
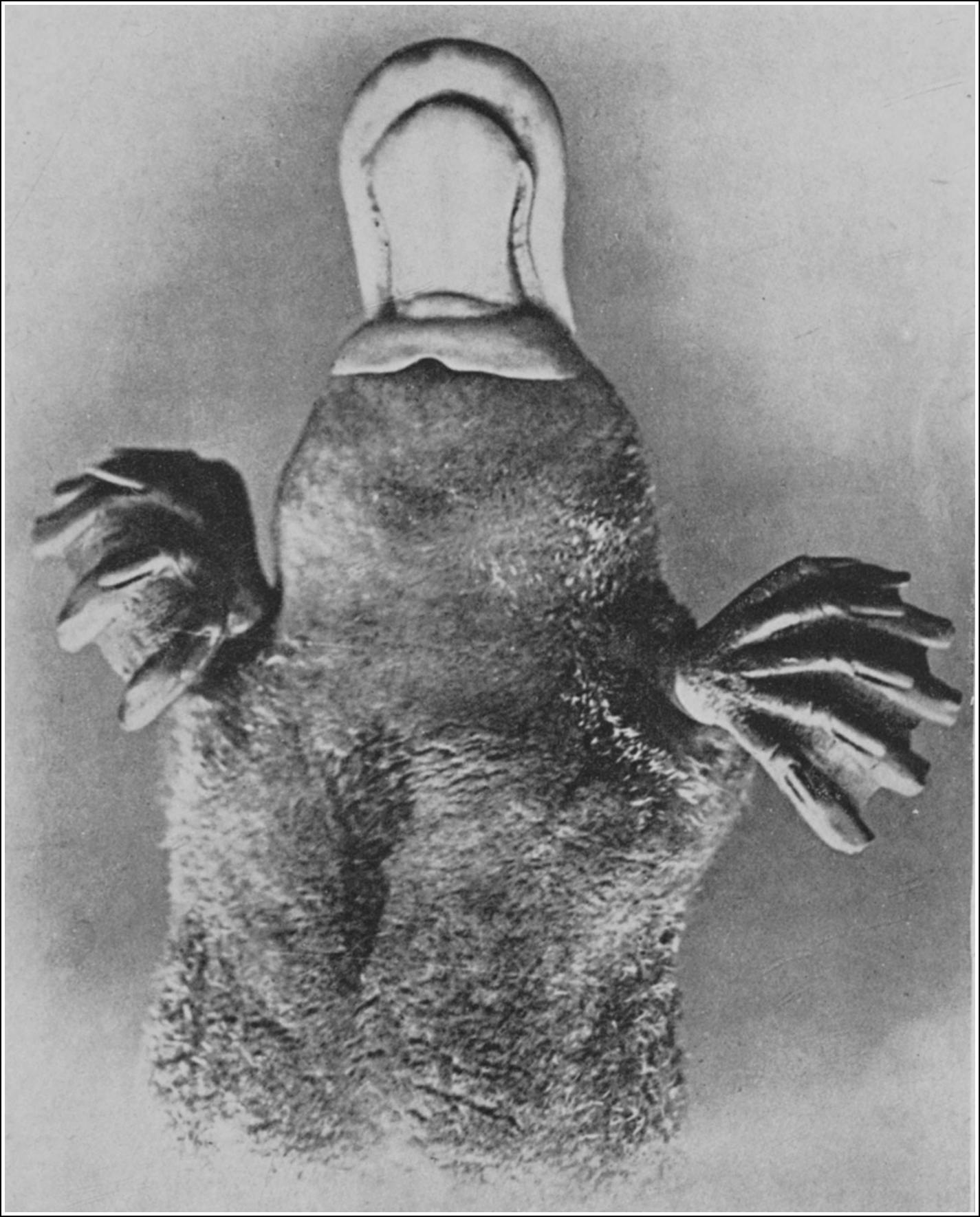
On land the progress of the platypus is not nearly so happy. The web of the fore-paw is folded under the palm, and the two limbs of one side are rotated outwards and forwards together. The nails of the fore-paw take a grip, and the creature pulls itself forward, going over on its knuckles as it does so. Meantime the backwardly directed claws of the foot have taken purchase, and the body is levered forward on the right limb. 159Then the limbs of the other side come into play. The result is a shuffling, sinuous motion, resembling, as I have said before, that of a heavy-bodied lizard, such as the Blue-tongue (Tiliqua scincoides). Both the trunk and tail drag upon the ground. Considering how clumsy its method of locomotion is, it is surprising how fast the platypus can move when it has need of haste. When going at top speed, it can raise its body almost completely from the ground, as the photograph reproduced in Plate 26 shows, and can travel on level ground at the rate of 200 feet per minute (by stop-watch).
The platypus cannot remain for any great length of time in water without some means of resting, otherwise it will become exhausted and drown, for it must be remembered that the platypus is a mammal and resorts to water mainly for its food, after which it must dry its coat and retire to its burrow for warmth. A freshly-trapped robust male was placed in an open tank while I leaned over so that it could clearly see me. The animal immediately dived and, by maintaining upward pressure with its fore-paws reversed, remained at the bottom for several periods of six minutes each, with intervals of one minute at the surface, during which it exposed the tip of its bill as far as the nostrils. While collecting two specimens of brooding females for the University of Sydney, and as they had to be killed in the process, I took the opportunity to ascertain that they survived 5½ and 6 minutes respectively before drowning in small wire cages. These observations do not prove that the male can stay under water longer than the female, as, during a test made midway between breeding seasons, a female survived for 10 minutes. This is by far the longest period which I have observed.
The aquatic life of the platypus occupies but a small portion of its time. Besides feeding and tunnelling, its only other duty would seem to be that of making its toilet—the rest of its time being spent in sleep. It performs its toilet, as a rule, while resting on the bank, or at all events rids itself of most of the moisture from its fur before going off to continue toilet operations in its resting-burrow. Bennett 160(1860, p. 119) gives an account of the operations performed by a captive which was tethered by means of a cord attached to one leg:—
“After it had wandered some distance up the chain of ponds, feeding about the shallow water and mud near the margin, it crawled up the bank, and, lying down on the grass, enjoyed the luxury of scratching itself and rolling about. In this process of cleaning its skin, the hind-claws were brought into use—first the claws of one hind leg, then those of the other; but finding that it could not use the one to which the string was attached so well as the other, which was disengaged, after repeated trials it gave up the attempt. The body, being so capable of contraction, was readily brought within reach of the hind feet, the head also coming in for its share of the process. The animal remained for more than an hour cleaning itself, after which it had a more sleek and glossy appearance than before. Placing my hand on a part which it was scratching at the time, the claws passed over it instead of the animal’s body, when I found that it performed the combing in a remarkably gentle manner. On my attempting to scratch the place gently, it started away, but not far, and soon resumed the method of cleaning itself, which I had interrupted.”
From personal experience with platypus in captivity, I am convinced that Bennett’s tethered specimen was actually dying of shock and starvation the while he imagined it was enjoying the pleasure of scratching itself and rolling about.
Bennett also describes the animal as preening its fur with its muzzle, like a duck. Semon (1894, p. 11) records that on two occasions he saw duckbills lying in the grass on the river-bank, stretching themselves and cleaning their fur.
My observations show that the hind-claws play the principal part in toilet operations, but that the fore-paws are used as well. The animal squats on its haunches, and by its gymnastic ability is able to bring almost every part of the body within reach of one or other of the hind feet. It aids this process in a rather ludicrous way by pulling the loose skin round into the desired position with the paws, the webs being extended but 161not expanded in the action. It also cleans itself with bill and fore-paws, just as a seal rubs itself with the hairy palm of its flippers while lolling in calm water. I have seen a wet platypus sit up and pull its exceptionally loose skin about with its fore-paws, as if actually massaging itself. The pressure of the broadened claws against the intervening thin webbing is evidently quite sufficient for that particular phase of its toilet. In that case the webbing serves for a rub-down in the same way as a moistened chamois pelt, if nothing more. Seeing, however, that the fore-claws are used principally for scratching into solid soil, and that the versatile and oscillating hands are adapted for grasping, the platypus may readily transform them into excellent combs by merely palming the webbing, as when burrowing, walking, running, or climbing, and may use them for combing the parts which cannot be reached with the hind-claws.
In respect of thorough cleanliness the platypus easily holds its own among mammals. In person and in dwelling-quarters, even when brooding, it always exhibits conditions exceptionally tidy. How different is this monotreme from its only known living relative, the echidna. To my mind, as regards toilet at least, the contrast is extreme. Every time I have examined a freshly-caught platypus—wet or dry, young or old—it was particularly clean in skin, pelage, and external orifices, such as facial furrows, nostrils, etc. Of course, this may be partly due to its semi-aquatic nature. Its water-dreading cousin, meanwhile, is about the dirtiest wanderer afoot. Quite apart from its perpetually begrimed muzzle and snuffling nostrils, the eyelids of an adult echidna of either sex are frequently found to be caked with a mixture of earth and secretions from the eyes, while the skin of the abdomen and the hair thereon are seldom found to be clean. Again, the temporary pouch of the female echidna, prior to weaning the occupant, is filthy, and this condition is certainly not entirely due to milk stains. Obviously, then, it is a blessing for both that the pouch is not a permanent feature, otherwise the mother 162would have to evolve something better than awkwardly-turned toe-nails and, possibly, a tacky tongue to complete her so-called toilet. Incidentally, it is fortunate for the young one that it is transferred, in a lethargic state, to sweeter quarters before attaining sufficient sense to observe its surroundings.
Though I have often disturbed platypus on river-banks, I have never seen them engaged in performing their toilet in the open. They resort, as a rule, to flat ledges under projecting roots, or to similar cover; these places, judging from the tracks leading to them, and from the way in which they are flattened down as if through long usage, would seem to serve as toilet tables of a comparatively permanent nature—that is to say, regular resorts of a given animal while it is in any particular locality. In such a place the platypus, secure from observation, and able to slip quietly into the water at the slightest alarm, removes the water from its fur, and reduces the latter to that condition of sleekness which is so essential to its well-being. As with most wild animals, the first sign of ill-health is the neglect of toilet, and this neglect in the case of Ornithorhynchus is rapidly followed by death. I have frequently noticed that sickly and enfeebled animals will attempt to perform the toilet motions, but are so little able to correlate their activities that the claws pass helplessly up and down without touching the body at all. In this condition they take no heed of man’s presence, being practically unconscious. It should also be noted that the feet are used for scratching the body while the platypus is floating at the surface; but this is probably due to the irritation caused by parasites. Ornithorhynchus, although flealess, shares with a very few animals the rather doubtful distinction of having a tick, Ixodes ornithorhynchi, all to itself.

Its toilet completed, the animal retires to its resting-burrow to sleep. Between this burrow and the nesting-burrow of the breeding-season a sharp distinction must be drawn. Existing accounts of the habits of the animal assume that the nesting-burrow of the breeding-season is the home of the platypus all the year round. Nothing could be farther from the truth. 163The complicated nesting-burrow is constructed by a single female for the purpose of laying her eggs and rearing her young, and is not resorted to by any other individual. Resting-burrows are, however, made by both males and females at all times of year. They are usually semicircular excavations under the roots of large red-gum trees and the entrance at each end is concealed by overhanging ledges, generally in the neighbourhood of the ‘toilet tables.’ The diameter of these tunnels is irregular, but always much greater than that of the nesting-burrows; the sleeping-chambers, from one to three in number, are simply enlargements of the burrow, containing no sign whatever of nesting-material or bedding. These burrows appear to be either purely male quarters or purely female quarters, the sexes not being found together in them. At most I have found two males occupying a single burrow.
When the animals are on satisfactory feeding-grounds, such burrows appear to be used regularly by the same individuals. But when they are forced by flood or freshes to move elsewhere, it becomes necessary to find new quarters. During these times platypus are sometimes captured in hollow logs or, in districts where alluvial mining is carried on, in sluice-boxes. Under stress the animal appears to make use of any kind of cover available. The males evidently make their own camping-burrows, as they are frequently found with the bald patch near the end of the tail. This is caused, as in the case of the female, by plugging off undesirable subways, etc. Burrows used by males may also be distinguished from breeding-burrows by a foxy odour which is compared by Mackenzie and Owen (1919, p. 12) to that of an onion. The odour is caused by the secretion of the scent-glands, which are situated at the base of the neck, just in front of the shoulder (see Plate 27).
Although, except during the breeding-season, platypus may be considered solitary animals, they are occasionally found in company for short periods, when forced by seasonal conditions to migrate. The greatest number that I have observed at the same time was four, and they did not appear to be consorting 164with one another, but simply feeding about quite independently. On one occasion I unearthed two adult males which were camping contentedly together in the one burrow. On another occasion I secured two males in a “turret trap” which had been set in the early morning for only an hour and a half. On a third occasion three males were trapped in one night, either while leaving or entering a camping-bank beneath a giant red-gum—a “buck barracks,” as I have since named the haunt.
Although Bennett has made the suggestion that Ornithorhynchus hibernates, my investigations do not bear him out. In the New England district of New South Wales the pairing season commences in July, which is the midwinter month on those cold highlands. The platypus lives an active life the whole year through, and eats prodigious quantities of food.
The thing one first notices in a living platypus is its extraordinarily restless activity. It is far more difficult to hold than any other animal of its size with which I am familiar, not so much on account of the actual strength and violence of its struggles, but because of its persistence in them. Bennett frequently remarks on the way in which the “looseness of its integuments” renders it difficult to hold. He refers to the skin and its underlying muscle-layer (the panniculus carnosus), which, in most mammals a very thin sheet, is in the platypus a thick muscular jacket, with slits through which the limbs and tail project and with numerous strong slips attaching it to various parts (see Plate 10). The animal seems to be able to slip its body through this loose overlying sheet in an uncanny invertebrate fashion, struggling forward for a time, and then, when it finds its progress prevented, suddenly changing its direction and slipping backwards out of one’s hands. At the same time all four powerful limbs are brought into play, and the head, flexed downwards by strong depressor muscles, also helps in the struggle. The only way in which the animal can be held safely is by grasping the scruff of the neck with the left hand and the tail with the right. While most animals after a time resign themselves to the inevitable, the platypus continues to 165struggle until exhausted, unless one is fully acquainted with its ways and coaxes it (see Plate 28).
Many observers have borne witness to the nervous temperament and restless activity of the animal in captivity. Three individuals which were forwarded by me at various times to the Department of Zoology at the University of Sydney, in order that some observations might be made, were all very impatient of the excitement and fuss of constant inspection and handling, and none lived longer than seventy-two hours. As the animals are easily capable of standing fasts for much longer periods, death would appear to have been due to nervous exhaustion, and certainly not to inanition—although, it is true, they took very little of the food that was provided for them.
It does not seem that the platypus displays anything of that affection for its mate which is so characteristic of the behaviour of birds and higher mammals. The period during which the mated pair are together is short, and I have no evidence that the male takes any further interest in the female and its young when once impregnation has been accomplished. The solitary observation of Hoy, quoted earlier—which might be taken to mean that the male charged in defence of his mate—is equally open to another, and, it seems to me, a more natural interpretation. Anger at the actual deprivation may have played a greater part than affection, for the actions were those of an angry beast. Moreover, since the occurrence took place in broad daylight, it is possible that the animal had only a very incomplete conception of what was going on. Nevertheless it remains to the credit of this particular male, that he did not dive and disappear after the report of the rifle, and the impact of the bullet close to him in the water.
Nor would the female platypus seem to show any actual affection for her offspring. She uses great cunning to keep them safe, and sees to their physical well-being with meticulous care, reducing herself to a state of emaciation and exhaustion in the process. But she makes no attempt to stay by and 166defend them when a burrow is opened up, her chief anxiety being to escape. I have never known a female in the burrow to show fight, or attempt resistance of any kind. Yet, even so, she shows signs of agitation, which is more than some marsupials do. When one tears the young from the back of a koala (Phascolaretos) for instance, the mother, after one vacant look in the direction of her puling infant, goes on calmly chewing gum-leaves.
The male platypus sometimes proves both pugnacious and aggressive. One which I was using for photographic purposes showed a great dislike of the process and deliberately charged and bit me on the arm and fingers. I compare the bite—which was made with the anterior pairs of horny ridges—with that of a gander, and it was sufficiently severe to cause me to take precautions against further attacks. Since then I have experienced bites from both male and female platypus which were deliberately roused for the purpose.
How far it is permissible to attribute to intelligence habits of wariness and cunning which have become what we are pleased to call instinctive is a moot point. The wary ways of the platypus have been described more than once, and there is no need to repeat them here. We may, however, assume that these ways, though they are proving useful against the onslaughts of European man, were used before his arrival.
167
Since the platypus is such a shy and secretive creature, rarely seen at all except by those who take the trouble of investigation, it is not surprising to find that, in the past, very little was gathered as to its mating and nest-building habits. One platypus-hunter quoted by Anderson Stuart makes the statement that the males fight furiously for possession of the females. Although I have never seen such combat between platypus at large, from what I have seen in this direction with specimens in captivity I feel justified in agreeing fully with that statement.
Early in the breeding-season there is in the male a correlated rapid growth and enlargement of three sets of glands—the testes, the crural glands, and the scent-glands. The enlarging of the testes (which from quite a small size become as big as pigeon’s eggs) needs no explanation. The scent-glands are more prominent in the male than in the female, and the fact that they become so remarkably enlarged at mating-time implies that the female seeks the male. This is rendered more probable by the fact that, when once impregnation has been effected, the female (so far as I have been able to ascertain) carries out the remaining duties of reproduction entirely unaided—quite a mammalian trait.
At breeding-time platypus usually leave their accustomed feeding-grounds and select a stretch of bank suitable for nesting. Under normal conditions nesting-burrows are not found close together. For the upper Namoi, the Macdonald, and the Manilla rivers, upon which most of my observations have been made, the average is one tenanted burrow to six miles of 168bank—that is, to three miles of river. Under abnormal conditions (in times of flood, etc.,) a different state of affairs often occurs, as will be evident from what follows, but normally the female does not seem to care about neighbours at this season. She requires a stretch of undisturbed water to herself while hatching her eggs and nursing her young, so that she may be assured of a near and easily accessible food supply.
Verreaux claims to have witnessed the actual act of copulation. According to his own account he spent whole days and nights hidden in a specially constructed hut, and was completely successful. I give his account for what it is worth (1848, p. 130):—
“The male, after chasing the female for nearly an hour, ended always by taking her into the middle of the rushes. There, clinging tightly to her with the help of his bill, he clutched the skin of her neck, while his hind-spurs grabbed her hinder end. The female, struggling violently against him, swam along uttering plaintive cries that were rather like the squeaks of a young porker and grew louder as they went on. The act took five or six minutes, and afterwards the two animals played together for more than an hour.”
If Verreaux meant that the male grasped the skin on the female’s neck, as a drake does, he manifestly did not understand that the extent to which the upper mandible overhangs the lower would render that impossible, quite apart from the pliable nature of the lips and the fact that both jawbones are divided at their extremities and are pliable as far back as the secateuring ridges. With regard to the squeak, I have never heard any sound from a platypus which might be so described, and I am convinced that either the whole account is imaginary or Verreaux was misled by a sound from some other source, possibly overhead.
Two observations which I was fortunate enough to make were widely separated. The first was made at 7.30 a.m. on August 27, 1909, in the Namoi River at Manilla. A platypus appeared on the surface, and was immediately followed by a second. As neither exhibited any surprise or alarm, it is probable that they had been together for some time. After 169a short interval one animal began to swim in a circle round the other, which, however, soon followed, so that the two were swimming in a circle one behind the other. They were about equal in size—probably an old female and an adult, but young, male—and it was on this account difficult to distinguish which had been the one to start the proceedings. After about a minute of this circling, one of the animals (which proved to be the female) submerged its body and tail, and floated perfectly still with its head alone above water. The male then came slowly up, and mounted in a leisurely fashion. The whole process offered a very close resemblance to the early stages in copulation of a drake and duck, with the exception that the male platypus did not take a grip with his ‘bill.’ The male then threw himself back into a sitting posture, partly out of water, but at this moment there was a great splash, and both animals disappeared.
The second observation was made twelve years later, on September 23, 1921, in the same river at a spot three miles from the first, and at 7 o’clock in the morning. There I saw what looked at first like a large male platypus floundering on the surface of the river as if sick or hurt; this, after a moment or so, dived. Immediately another object followed in its wake, partly-submerged and upside down. In a couple of minutes a second platypus (this time apparently of smaller size) rose to the surface; more floundering, or wallowing, took place at the surface, and I could now perceive quite distinctly that what I had seen was a pair of platypus coupled in an extraordinary position. The tail of each was laid flat along the belly of the other, completely hiding the bright gold and silver of the ventral fur. The precise position of the hind limbs could not be made out, as no movement thereabouts was discernible; but it must have been the grip of these that kept the animals together. So closely were they apposed that they appeared at times like a single giant platypus. The processes of diving, blowing, and floundering at the surface were repeated several times at short intervals. On each occasion the first to rise for a blow would be the only one visible above water, the 170second being clearly visible only as it trailed upside down in the wake of the first on diving. During the floundering and rolling which took place at the surface the under animal could be seen dimly. The animals rose alternately, apparently for breathing purposes, and on each occasion the second animal seemed to follow, helpless and rudder-like, upon the movements of the first. The whole of the manoeuvring was carried out in a calm, slow, deliberate manner, and almost noiselessly. How long they had been coupled together before I observed them I do not know; but my observation lasted fully three minutes; then the couple separated beneath water, and simultaneously appeared again on the surface, finally diving and disappearing.
I made an attempt to follow the actual process of copulation, making use of a recently killed male and female which, though dead, were still limp and pliable. I found that, when the male rises into the sitting position which I have already described, the tips of his spurs may easily be inserted into the sockets which occupy the corresponding position on the hind legs of the female. If the male then throws himself right back, and at the same time the female draws her pliable tail between his legs, the position observed in the final stages is reached, and in this position the cloacal apertures may easily be opposed. This use of the spur would supply the reason for its being movable, a matter for discussion later.
To insert the spur into the socket to its full extent, as stated by Home, would certainly involve gymnastic ability. My firm belief as to what really happens, is this: When in the sitting position, the male straddles the hips, keeping his feet at right angles with his legs. In this attitude the spur can be erected to its fullest extent (in a manner similar to the action of one’s thumb when the clenched hand is opened suddenly). In an adult male the space between the foot and the tip of the spur, at that time, measures over an inch. This, then, is more than sufficient to embrace the female ankle comfortably. Presumably the female does not resist the application of the grip, and, when it is securely taken, she flicks her pliable tail 171into position. It is with such simplicity then that the grip is secured and steadfastly retained to the end, though, possibly, aided with two pairs of clutching feet.
Now if we take into consideration the shape and length of the curved spur, which follow befittingly the depth and ovalness of the female ankle, it will not be difficult to ascertain the reason why it is impossible for the male to insert more than the tip of the spur into the socket, even were he desirous of doing so.
As regards Home’s statement that the male ejects the secretion of the spur into the female socket to bring about release, to me this also appears wide of the mark. On examining the spur, which averages ¾ in. long and is shaped somewhat like that of a cock, it will be found that the aperture is not at the tip, but back from it, and, furthermore, is situated on the convex surface farthest away from the socket during the embracing position. I therefore fail to see how a fluid ejected beneath water could enter the socket. As a matter of fact, the poison cannot be ejected unless the spur is pressed against the male’s own leg. How, considering the shape of the weapon, can this possibly be done while the thickness of the female’s ankle, or lower leg, intervenes? The purpose of the aperture’s being set back from the ripping tip of the spur is obviously to prevent clogging while fleshing prior to administering the poison. This strengthens my belief that the secretion is intended to irritate a wound.
The breeding-season of the platypus in northern New South Wales may be estimated with fair accuracy from my long series of observations and records. I have observed copulation, as I have already stated, on two dates only, 27 August 1909, and 23 September 1921; but I had a report from a competent observer, Mr. J. Maclean, that platypus were “courting” in the Macdonald River in the middle of July, 1920. The earliest date on which I have actually taken eggs from the nest is 24 August (1925). On the same day I unearthed twin young ones which I estimated 172to be three days old. This would carry back the date of laying to, perhaps, the first week of August. I have no guide as to the time which elapses between the laying of the egg and the hatching of the young, but I think it must be less than that necessary for the incubation of birds’ eggs, owing to the partial development of the foetus before the egg is laid.
The latest date upon which I have taken eggs is 22 October (in 1918); Kershaw (1912) has recorded the taking of eggs upon the same day of October in 1912 in Victoria. Consequently the period during which oviposition has actually been observed ranges from about the middle of August to a little after the middle of October. There are certain conditions, however, which affect the breeding-season, and I do not believe that normally it has so great a length. In the first place there is, as one would expect, a general tendency towards earlier breeding in the north; as one proceeds south littering becomes progressively later. In central Queensland July and August are the most active months; in New South Wales, August and September; in Victoria, September and October. I have no information as to the breeding-season in Tasmania. But the normality of the breeding-season may be seriously interfered with by conditions of weather, and particularly rainfall. Heavy rains occurring in August and September flood the burrows, which are consequently deserted. This flooding is brought about chiefly by seepage through the soil, as the upper parts of the burrow are almost always above ordinary flood-level of the river. It is thus clear that the pugging of the tunnel is not a precaution against the entrance of water from the river, as has sometimes been suggested, but is for the purpose of providing conditions necessary for incubation. The effect of floods upon the breeding-season in the New England district was observed during September and early October of 1920. In July, the rivers were in flood. Platypus were reported plentiful after the waters had receded in the early part of August. A second flush occurred late in August, when the waters rose to six feet above their normal level; and sporadic rain continued 173until September 18. Under these conditions a number of burrows which I opened up were found to be deserted and the contained nests sodden with water. More remarkable still, several tenanted burrows were found in close proximity to one another, which is unusual; and in one case two females were found occupying nests in burrows which had a common entrance, an occurrence without parallel in my observations. Furthermore, several tenanted burrows showed signs of extremely hasty construction, being only a few feet in length, and with fewer pug-pits than usual. Again, in 1923 an eight-foot rise in the river had washed out all the breeding females, and no tenanted burrows at all were found on the plateau. A female killed on October 9, when examined, was found to have in her left uterus eggs in the early stages of segmentation. In a normal season eggs are laid during the later part of August and early September, and from late September on to the end of October large young are found in the nests.
The ovum of Ornithorhynchus (that portion of the egg which corresponds to the yolk of a fowl’s egg) is, at the time of fertilization, a small yellow sphere about 3 millimetres in diameter. After fertilization it becomes surrounded by an exceedingly thin layer of albumen, and, outside this, by a thin, transparent, horny shell. In birds there is a thick layer of albumen (the white of the egg) deposited in several distinct layers, the innermost having spirally twisted prolongations which serve to hold the yolk suspended in the middle of the egg. Outside this thick albumen are two delicate membranes, closely adherent everywhere except at the larger end of the egg, where they separate to enclose an air-chamber; these membranes are closely applied to the rigid, calcareous shell. The most remarkable difference between the egg of a bird and that of a monotreme is that in the former the shell is deposited round the fully-formed egg, and there is no increase in its size during the growth of the embryo, whereas in the monotreme the eggshell 174increases in size and alters in structure during the intra-uterine development of the embryo. The thin shell, which is at first only 4 millimetres in diameter and spherical in shape, stretches and thickens until a diameter of 10 millimetres has been attained. At this stage the egg is still practically spherical, but the shell has become thicker, and has been rendered opaque by the deposition of lime-salts. As it goes on growing, it becomes ellipsoidal in shape, the longer axis corresponding to that of the contained embryo (Wilson and Hill, 1908), and finally attains its maximum size (16 to 18 millimetres long by 14 to 15 wide) just prior to being laid. There can be no doubt that this curious growth of the egg, which has no parallel in nature outside the monotremes, is due to the imbibition of fluid from the uterus. There is not sufficient nutrient material in the yolk-mass of the fertilized ovum to produce the young platypus which hatches from the egg; consequently the intra-oval foetus is nourished in a considerable measure by secretions from the maternal uterine wall. Since secretions can pass into the egg through the mesh-work of horny fibres of which the shell is at first composed, it is also possible that foetal secretions may pass out; there is at present, however, no evidence that such a process takes place. The point is of interest, for, in the higher mammals, a secretion from the foetus—of the nature of a hormone, or chemical messenger—which passes into the maternal blood-stream, is conveyed to the mammary glands, and stimulates them to the active secretion of milk; but, in the platypus, neither at the time of oviposition, nor later, when the young actually hatch out, are the milk glands of the mother in active condition. The stimulus to lactation would seem to be derived in some external and mechanical way from the presence of the young. This question will be further considered below.
It is thus obvious that, though the monotreme egg is usually looked upon as similar to those of birds and reptiles, it is in reality very remarkably different. The nourishment of the embryo within the egg by secretions from the uterine wall is distinctly a mammalian feature. Such a process does not occur 175among birds and reptiles;[16] in these, when once the egg-shell is formed, the embryo has to depend altogether upon the nutrient material packed within the egg. It must be admitted, however, that what occurs in the monotremes appears to afford very little clue as to the evolution of the placental habit in the higher mammals.


The platypus lays one, two, or three eggs, two being the usual number. There is no record of four eggs, or four young, ever having been discovered, and the statements of Bennett and by Lucas and Le Souef that the number of eggs is one to four are probably based on Geoffroy St. Hilaire’s unfortunate experience (see p. 36). When two eggs are laid, these are invariably found joined together side by side. So closely do the apposed walls adhere that considerable force, comparable to that required to pull apart two fairly dry ‘tangle-foot’ fly-papers, must be exerted to separate them. The only records of triplet eggs are of two sets taken after hatching and one intra-uterine set. Each of the former sets of shells was in the form of a cluster, with the length of all three eggs lying practically parallel, but the ends free for emergence of the young. In one of the sets the three were all joined to each other; in the second set there was a small space separating two of the shells, but both of these were joined to the third. This difference is evidently unimportant, and occurs when the eggs are being clamped together immediately after they are laid.
There is no longer any mystery as to the way in which the eggs are laid and afterwards fastened together. When the eggs are ready to be laid, they are well apart from one another in the uterus, with their longer axes in the direction of the length of that organ. The capsule of each separate egg is at that time completely covered with a sticky fluid. When about to lay, the female squats on her rump in the nest, with her tail between her legs, and her fore-paws in readiness 176at each side of the aperture of the protruding cloaca. Presumably, the eggs are deposited singly into her soft rubber-like hands and held there firmly until each egg capsule, where in immediate contact with its fellow, becomes affixed thereto; then, with the help of the under surface of her fat, pliable tail, they are clamped to her warm abdomen. Meanwhile the solution still covering the remaining portions of the capsules soon loses its stickiness through either evaporation or absorption. It is improbable that the eggs are held for long in the fore-paws, or that their adhesion and the drying of the exterior portion of the capsules is a lengthy process.
It may interest my readers to know how I have come to these conclusions, seeing that the eggs are laid in a pugged cavity beneath the solid earth. While collecting embryological material for the Canberra National Museum on September 2, 1925, at Manilla, I took a female platypus with mature triplet eggs in her left uterus, and on extracting these I found that their capsules were conspicuously glossed with a thick coating of sticky liquid. I immersed them separately in a powerful drying fixative called “Bles;” but notwithstanding the bleaching and hardening properties of this mixture of formalin, spirit, and acetic acid, the eggs not only stuck firmly together on coming into contact with each other, but also adhered to the glass and to a paper label attached to another specimen in the bottle. This, I think, proves that the eggs adhere to each other only after being laid, and not even in the cloaca, as has been hitherto supposed. Drying off after adhesion and exposure to the atmosphere would appear to be quite natural; this could not occur prior to deposition. But one may ask why, if the eggs will stick to paper and glass, they do not stick to the hands of the mother during the act of manipulation. The probable explanation is that the rubber-like membrane of the fore-paws of a platypus is always cold and moist, however dry and warm the rest of the animal’s body may be. This is as it should be, because of the various functions that the versatile fore-paws are called upon to perform at a moment’s notice, such as walking and swimming, involving rapid action similar 177to the alternate opening and closing of an umbrella. It will be obvious that the very existence of the platypus depends on the versatile manipulation of this membrane. Nature would not permit any temporary clogging of this intricate member. In 1924, while Mr. Ramsay and I were making a moving picture of the natural habits of monotremes, I tried with strong fish glue to fix the staff of an Australian flag in the closed palm of a living platypus, but found that it was impossible.
My observations of intra-uterine eggs apply to two pairs in the early stages of segmentation. Each pair was found in the gravid left uterus, which alone is functional; in each case the eggs, one behind the other, were separated by an appreciable interval. Wilson and Hill obtained a number of intra-uterine eggs, some in a fairly advanced state of development; but they give no information as to how the eggs were situated in the uterus. They do, however, figure (1908, Pl. 4, fig. 6) a section of a uterus containing a fairly advanced single egg, which shows that the egg lies in the position one would expect, namely, with its longer axis in the direction of the length of the uterus. Caldwell (1887, p. 464) states that he shot a platypus which had laid her first egg, while the second lay in the mouth of the uterus. He does not, however, state that he actually found the first egg, and it may be that he went on the assumption that the animal invariably laid two eggs, which we know not to be the case. Gold-receiver Rumby’s platypus is reported to have laid two separate eggs in a gin-case (see pp. 41-43); but there is so much that is dubious about this particular story that it cannot be accepted as definite proof. If the evidence could be relied upon, it would show that the eggs are laid separately, and the fact that they were not found cemented together might be attributed to the unnatural circumstances in which the mother found herself. As, however, the two eggs follow each other down the uterus lengthwise, it is obvious that they cannot in the process become joined together side by side. Moreover, when there are three eggs it would not be possible for them to be laid if they became cemented together within the body of the mother. It is therefore 178certain that this cementing takes place after they are laid.
My observations, together with those of Wilson and Hill, show that any pair of eggs contains embryos in an identical state of development, even in the earliest stages of segmentation. This would imply uniform conditions for both eggs, and from this it may be presumed that the ova are extruded from the ovary and are fertilized simultaneously, that they pass down the oviduct (including its uterine portion) close to one another, but end to end, and that they are laid practically at the same moment. At the moment of extrusion they are coated with a sticky secretion from the oviduct walls, and it is a simple enough matter for them to become adherent. But the fact that their long axes are always parallel after laying would seem to necessitate definite action by the platypus. There might be one other suggestion—that the eggs, having passed through the pelvis separately, are retained in the cloaca long enough to become attached to one another before their final extrusion; but this I consider impossible. A further curious feature is that the embryo lies along the long axis of the egg, parallel to that portion of the shell which is cemented to its fellow, and, after the egg is bleached, may be seen through the shell with the naked eye.
My observations of the right ovary and oviduct go to show that, though not functioning in the true sense, they appear to be affected in a certain proportion to the activities of those on the left. For instance, while the left is maturing triplet eggs, the right becomes enlarged to about the size that the organs on the left would have when carrying a single egg. In cases of twin and single eggs, the enlargement is proportionately less.
Of seventy tenanted nests examined by me, eleven contained either one egg or one young one; fifty-four contained two; only five contained three. Of six nests recorded by Kershaw, one contained a single egg and five either twin eggs or twin young. Hill’s record of stages of eggs taken from nests comprises one single egg and four twins. Wilson and Hill’s paper (1907) on the intra-uterine stages does not state clearly 179what was the exact proportion of singles to twins in the material examined. There are a number of older records by Maule, the Bennetts (father and son), and others, but the figures given above are sufficient to indicate the approximate frequency of each egg-number. Triplets are rare, the only record apart from my five (four times of young in nest, and once of intra-uterine eggs) being a single record of young by Dr. George Bennett. Ignoring triplets altogether, singles are in a proportion of roughly twenty per cent, and twins of eighty per cent.

Kershaw (1912, p. 106) gives the measurements of a pair of eggs as 18 × 15 mm. and 16 × 14 mm., and of a single egg as 18 × 15 mm. My observations give the following measurements (in millimetres):—Shrivelled egg (unhatched), 16 × 10; single egg, 18 × 14; twin intra-uterine eggs, 3 × 3; twin eggs, 16 × 12; twin eggs, 17 × 14, 16 × 14; single egg, 17 × 13; twin eggs, 15 × 26 and 17 × 26; twin eggs, 15 × 25 and 16 × 25; twin intra-uterine eggs, 5 × 5; twin eggs, 18 × 14, 15 × 14; twin eggs, 18 × 25 and 16 × 25; single egg, 15 × 13; twin intra-uterine eggs, 6 × 6; triplet intra-uterine eggs 17 × 15, 18 × 15, third smashed (abortion). No triplet eggs have yet been taken from a nest. It would appear from the measurements given above that the average of the single egg is usually larger than that of either of twin eggs, the measurements being 17.25 × 14 mm. against 17.5 × 13.8 mm. It will also be noticed that in only one of five pairs of twin eggs are the eggs of equal size. In the others one is usually distinctly smaller than the other.
Of the intra-uterine specimens listed above, the 1920 pair measured 3 mm. in diameter; they were spherical in shape, yellow in colour, and were situated in the oviduct close to the left ovary. The 1923 intra-uterine pair were 5 mm. in diameter, spherical in shape, yellow in colour, and were situated in left uterus. The 1925 pair measured 6 mm. in diameter, were spherical in shape, “orange” in colour, and surrounded by albumen.
180
The 1925 triplets measured (1) 17 × 15, (2) 18 × 15, the third being smashed through abortion while in the uterus but uniform in size with the others. They were ellipsoidal in shape, glossy white in colour, and were situated in an abnormally enlarged left uterus, two adjoining nearest to the oviduct, the other in a separate compartment close to the cloaca. All three were lying lengthwise with the trend of the uterus, while the ovary connected therewith was partly covered with a crystal jelly encased in a thin transparent film. This jelly, or “hydatid-like” sac, was almost as large as the swollen ovary, and, except where it was attached, somewhat similar in shape to its host ovary. Whether it was for the purpose of supplying the compound which envelops the eggs when capsuled in the uterus, I cannot say; but it was preserved intact for later investigation.
When first deposited, the eggs have a full outline, but as incubation proceeds they frequently become dented, as well as discoloured. Kershaw (1912, p. 106) notes that mere exposure to dry air produces denting in a few minutes. I think, however, that the denting is due to the age of the egg, because on two occasions I have exposed apparently new-laid eggs for thirty minutes in the open while photographing them, and they retained their full outline perfectly throughout, although handled considerably. It may be that the necessity for a moist atmosphere is part of the reason for the pugging of the burrow.
The shell is thin, and easily compressible. It is composed of a mesh-work of keratin fibres, impregnated with calcareous salts; except in shape, it resembles that of a lizard. The shape is broadly ellipsoidal, the ends being shortly and evenly rounded. The colour of the newly laid egg is a glossy white, which, when the egg is dry, turns to a flat chalky white, and later becomes stained brownish. The shells from which the young have been hatched have a more polished and translucent brownish-white appearance, which may best be compared with that of the celluloid balls used in “ping-pong,” when they have become crushed (see Plate 23).
181
Incubation is carried out by the female alone. During many years of observation no male has been found in a breeding-burrow, and Joseph’s account of platypus in captivity (p. 205) indicates that females resent the intrusion of the male even into their resting-places. On one occasion I was fortunate enough to catch a mother-platypus napping, and she did not wake until an attempt was made to insert a thermometer into the middle of her curled-up form. Her tail was turned up over her abdomen, holding against it two 65 mm. young. I am convinced that the eggs are held during incubation in a somewhat similar manner, and that their cohesion supports this view (see Plate 30). Kershaw (1912, p. 105) records the finding of newly hatched young, one of which was attached firmly to the skin of the mother. At this stage the young have not begun to suck, but they are very helpless and might become entangled in the fur of the mother’s abdomen, where they invariably nestle. Kershaw has also expressed this view, and he mentions, incidentally, that the other young one fell off as the mother left the nesting-chamber. Of two observations by Kershaw (loc. cit.), and six by myself, of nests in which eggs were found, in no instance was the mother absent from the burrow. This would surely indicate that the female incubates her eggs in the way described.
The point is of importance, because there is a considerable difference between the temperature of the nest and the body temperature of the mother. I have found that the average temperature of the nesting-chamber (taken by thrusting in a thermometer the moment it was opened up, and blocking the hole with a sack) was 64.5° Fahrenheit (19.2° centigrade), whereas the average cloacal temperature of seven brooding females was 90° Fahrenheit (32.2° centigrade). Such a difference in temperature would have a considerable influence upon the duration of incubation. The length of the incubation period is unlikely to be ascertained by observation in the field, but I think it should be possible to discover it artificially. At present it can only be conjectured from comparison with other 182oviparous animals. Passerine birds which lay eggs of the same size as those of the platypus have an incubation period of from twelve to fourteen days; they also have a higher blood-temperature. But, as against this, Caldwell has made the observation (1887, p. 464) that the egg of Ornithorhynchus, when ready to be laid, contains an embryo already in approximately the same stage of development as a thirty-six-hour chick. It may not be very wide of the mark to suggest that the incubation period of the platypus is about fourteen days, though possibly less.
I am convinced that, during the three weeks or more which elapse between the laying of the eggs and the onset of lactation, the female not only does not leave the nest, but also passes into a condition of partial aestivation. I think the eggs are laid in the fore-paws and placed, in the manner described above, between the upturned tail and the abdomen, and that they remain there throughout the incubation period and possibly beyond, until mammary secretion is excited by the mechanical stimulus of the movements of the young. These convictions pass beyond the observed facts, but are, nevertheless, consistent with them.
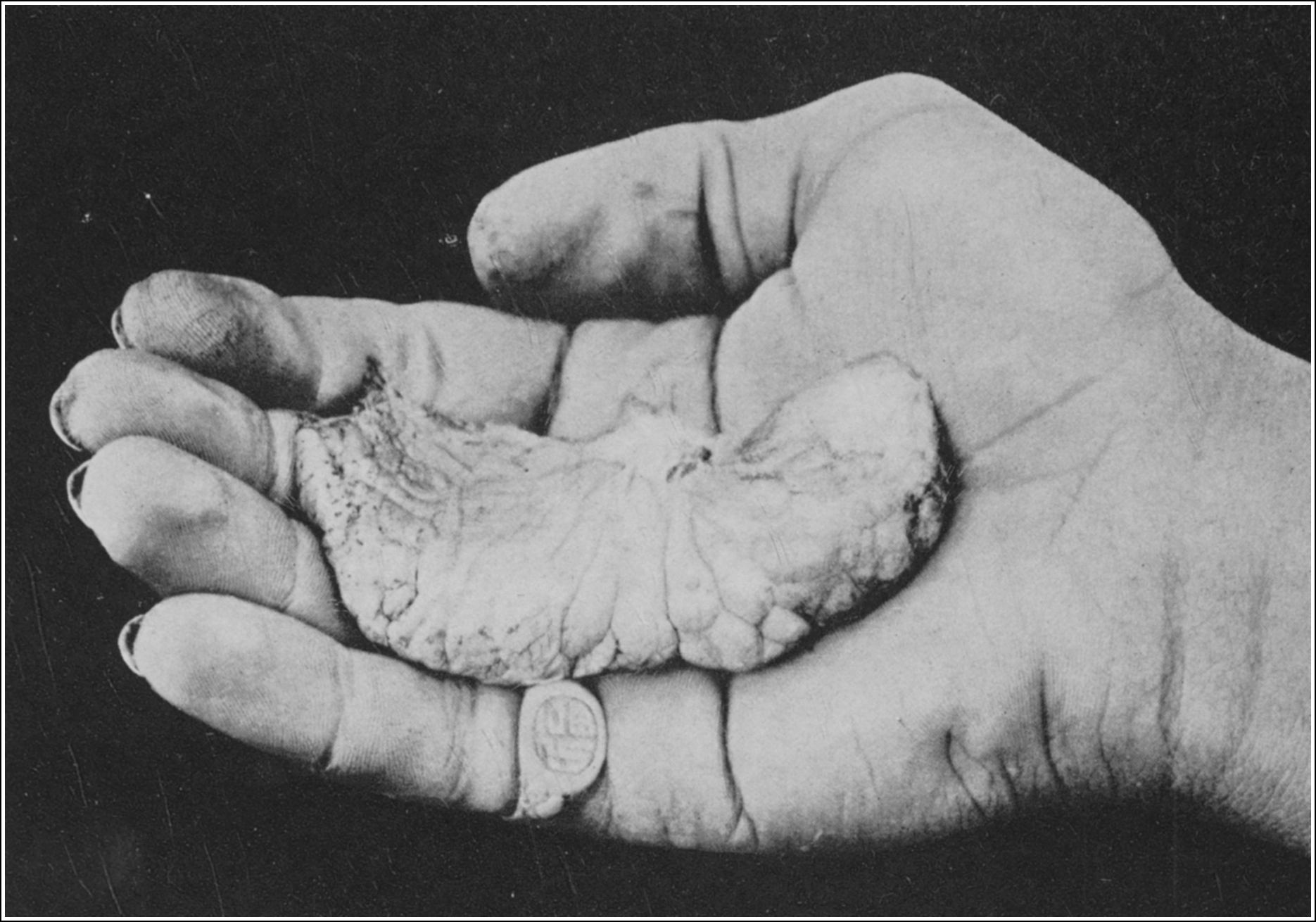
The hatching process has not been observed completely. The shells from which the young have hatched are always found in the nest in a flattened condition. On relaxing them in warm water, months later, I have found that they can be made to resume their original shape. It is worth noting here that the warm water has no effect upon the substance which caused the eggs to stick together when laid. The young are seen to have emerged through ragged rents, and seldom is any portion of the shell missing. The twin eggs examined all show that both young emerged at adjoining ends. In the only two sets of three shells recorded, a somewhat different state of affairs was found (see Plate 23, fig. 4). In two of the eggs the rents were at opposite ends; in the 183third the rent opened at right angles to the plane of the other two; the result being three openings as far removed from one another as the surfaces of the eggs would allow. It is impossible to credit the mother with an instinct enabling her to arrange the eggs with regard to the planes of embryos which are only very little formed at the time of laying. It is likewise difficult to conceive that the embryos themselves can possess any instinct which leads them to break their way out in a given direction in which their passage is assured. The probability is that the eggs are invariably attached to each other by their sides, and that the young always break out at the ends, and so no complication can arise.
When young from the nest were first described by Owen, it was thought that the caruncle on the muzzle, which is hard and sharp a couple of weeks after hatching, corresponded to the ‘egg-tooth’ found in many birds and some reptiles, and was the means by which the young one cut its way out of the egg. The discovery of earlier mammary foetus stages has shown, however, that at the time of hatching the caruncle is merely a fleshy knob, which could not possibly serve as a cutting instrument. Professor J. T. Wilson states that in the newly hatched young there is a small egg-tooth anterior to the caruncle; but this must be a very evanescent structure, for no sign of it can be seen in young ones 18 mm. long, the earliest mammary foetus (a term to be explained later) collected by me. These, from the soft, moist condition of the shells, and the umbilical protrusions exhibited by the young, I considered to be newly hatched, but Professor Wilson’s specimens are smaller. Whether helped by an egg-tooth or not, it is probably an easy matter for a restless, muscular creature like the platypus embryo to break out through the thin shell, using the caruncle as a point of resistance against the pull of the fore-paws, with an action similar to that adopted by the young when pummelling the mother’s abdomen and nuzzling for milk. I have observed a similar action on the part of overturned naked young regaining their feet.
184
The young, when first hatched, show few of the characteristic features of the adult platypus. There is no sign of the curious muzzle, which Geoffroy St.-Hilaire protested would be such a stumbling-block to the imbibing of milk from mammary glands. Indeed, except for the flattening of the rudimentary tail, and the shortness of the limbs, with their even fingers and toes, the young platypus has little to distinguish it upon casual examination from any early mammalian foetus. It grows into characteristic platypus form at a later stage.
The most remarkable and mysterious feature about the baby platypus is that it is not suckled at all by the mother for some days after hatching, for the very good reason that the maternal mammary glands are not yet actively functional. Investigations of this extraordinary phenomenon have advanced far enough to place the matter beyond doubt. On October 9, 1923, I took three female platypus, two with the mammary glands almost dry, the third with them quite so. I noted the inflamed condition of the uterus of the third individual, and it proved on examination to contain two eggs in the early stages of segmentation. On September 27, 1921, I took a female with a young one 45 mm. long. Here the mammary glands had begun to enlarge, but had not reached half the maximum size, and would seem not to have been actively functional, though four or five days must have elapsed since the time when the young one was hatched. Against this it must be recorded that Kershaw (1912, p. 105) took a 30-mm. young one, “so securely attached to the skin as to require a little force to detach it.” Kershaw does not state the method of attachment; but, as the limbs are very feebly developed at this stage, it is certain that the little creature must have been sticking to the fur accidentally. In all my experience I have never found the young ‘attached’ to the fur. Whether the individual which Kershaw observed was really obtaining nourishment cannot now be determined. Had he dissected the mother, he would have discovered that at that stage the milk glands were dry. I have made a careful examination of the 185glands of a mother with 20-mm. young, and could not induce milk to flow by external pressure—a process which can easily be carried out when the young are older—nor, on dissection, could I find any trace of milk, the glands appearing quite dry. An examination of a considerable series of mammary glands from nursing mothers which have been collected with their young has convinced me that during the first week, at least, after hatching there cannot be more than a very slight milk-secretion, if any at all, and I think this characteristic applies to the echidna also.
I have made the experiment of placing young apparently two weeks old upon the abdominal fur of the mother, who was laid upon her back and held in that position. The young ones crawled aimlessly about, and passed and repassed over the mammary area without appearing to be aware of its presence. They made no attempt whatever to cling to the fur with either mouth or feet. Older ones, however, clung tightly with their fore-paws, using their hind limbs as additional supports when the mother was held up vertically (see Plate 10). While observing these latter young, I noticed a peculiar action which appears to lend some support to a rather speculative conclusion previously reached as to the function of the caruncle. The young were observed to bend their heads under their bodies, and to withdraw them in such a way that the dorsal surface of the muzzle, towards the end of which the caruncle is borne, was dragged forward over the mammary area so that the sharp point of the caruncle scored its surface. It had occurred to me previously that, since the caruncle could not, for reasons given above, have the function of an egg-tooth, it might well serve the purpose of exciting mammary secretion. It is at least a significant coincidence that the onset of obvious lactation occurs simultaneously with the completed growth of the caruncle. It may be that the stimulus afforded by this ‘milk-spur’ is necessary to induce the very primitive milk glands of Ornithorhynchus to act; and the delayed lactation which I have observed may be due to the incomplete development of the necessary stimulus in the early 186young. How the young platypus is nourished in the meantime I do not know.
On one occasion only have I found the mother to be absent from a nest containing young less than two weeks old. It must be remembered, however, that burrows are always opened up by daylight, and, as the female is certain to be more than usually wary during the nursing period, it is safe to assume that she would generally feed under cover of darkness. When the young are older, they are frequently found by themselves, presumably because the mother has to feed long and often in order to maintain her milk supply. When the female leaves the burrow, all the pugs are closed behind her, which means that she digs her way through each of them, passing the earth under her body and compacting it with her tail as she goes. Since the same process has to be repeated on her return, and since as many as nine pugs have been observed in a burrow, feeding excursions must be a laborious business and are not likely to be indulged in more often than is necessary.
During the early nursing period, then, the mother spends most of her time curled up about her babies, holding them snugly against her abdomen by means of her tail. The young at this stage are generally referred to by the term ‘mammary foetus,’ which is the name given to the pouch-young of marsupials during the time they remain on the teat. There is, however, no close correspondence between the two. The marsupial, coming to active life for a brief space after birth—long enough, it is said, to reach the pouch of the mother by its own voluntary effort—relapses into a foetal condition in which it is apparently incapable of voluntary movement. It cannot even suck, the milk being pumped into it by the mother. This act is obviously impossible for the teatless monotremes, hence their fasting. The young platypus, although its bodily form undergoes a fair degree of modification during the first fortnight, and though it is not hatched entirely after the image of its parents, is continuously capable of voluntary movement from the time it leaves the egg. In the newly hatched young the movements are comparatively feeble; but after the first week 187the young are in a state of perpetual motion, at least when exposed to daylight, which they appear to dislike. In the darkness of the nest, however, they probably spend most of their time in sleep.
In the echidna the mammary areas open into the pouch, which is formed as a temporary structure during the breeding-season, and the young one laps its nourishment with its slender projecting tongue. The platypus has no pouch, and the milk oozes out through numerous fine apertures upon two mammary areas of the abdomen, each about half an inch in diameter. These areas are covered with fur, and are not in any way distinguishable from their surroundings, so that they are difficult to locate by external examination. Pressure upon the mammary glands, however, causes the milk to flow gently out, and their precise position can then be seen. The hair covering the areas serves, apparently, in place of a teat, and the young pluck at this and suck the milk from it, much as a little aboriginal eats his honey by sucking it from a piece of shredded bark. The “lips” of the young, owing to the shortness and undeveloped form of the bill at this early stage, are adapted for sucking in conjunction with the tongue (see plate 33).
An unsuccessful attempt was made to keep alive two young ones (about a fortnight old), which had been sent to the Department of Zoology at the University of Sydney, by inducing them to suck cow’s milk from pads of cotton wool. Even when the milk was squeezed out upon their tiny muzzles, they did not appear to take the slightest interest in it. I had much the same experience in the field with a pair of well-grown young, which were in fur and had their eyes open. I placed drops of milk upon the hairy portion of my forearm, but they merely brushed it aside. After crawling inside my sleeve (evidently to avoid daylight), they did, however, pluck at the skin of my arm with their jaws, with a sucking action sufficiently strong to bring a flush of blood to the spot. The opening of the jaws was accompanied by a feeble kiss-like explosive sound, resembling that made by a man releasing smoke from the side of his mouth when lighting a pipe.
188
The naked young range from bright red to rose-pink in colour when alive, and their delicate skin has a silken sheen, and is minutely wrinkled. In preserved specimens these wrinkles become much exaggerated, owing to the contraction of the underlying muscle-layer. When the hair-follicles begin to develop, the skin of the back assumes a bluish appearance, like that of a shaven chin, but the under surface of the body still keeps its pink colour (see Plate 33). Finally, with the growth of the hair, which is at first fine and silky, the adult appearance is reached.
A feature of interest in connection with the nursing habits is that the nest never shows any sign of having been fouled with excreta. Young which I have had in temporary confinement defaecate freely; the faeces take the form of an odourless, greenish, oily slime, which may be compared with the contents of the gall-bladder of an ox. (Adults, in captivity or at large, always defaecate in water.) As the nest is always clean, this inoffensive matter would readily filter through the nesting-material without causing more discomfort than that endured by nestling Owlet Nightjars—in fact, not so much, since it would not form hard masses like the bird droppings. But it is reasonable to suppose that very little faecal matter passes from either the mother or the young during the period of incubation and the nursing of little ones. While suckling, the mother leaves the nest occasionally to feed, and, presumably, to defaecate also. The nest is then probably relined from time to time, as is the custom of the Owlet Nightjar.
It is for these reasons that I maintain that platypus, when brooding, fall into a lethargic state and, thus requiring little food, do not evacuate in the nest unless abruptly disturbed.

About six weeks after hatching, the young will have reached a length of twelve inches. By this time their eyes are open, their fur is a quarter of an inch in length, and they are able to crawl freely about the burrow. (See Plate 12, fig. 2). This 189may seem a rapid rate of growth, but such rapidity is explained by the fact that when once the young commence to suck, their appetites increase rapidly. The quantities of food found in their stomachs on dissection are surprising. (Because of this, the stomachs must be tapped and the contents drained before fixing in “Bles,” otherwise the milk putrefies and is apt to spoil the entrails.)
Just prior to the appearance of pelage on a large nestling, the intestines and stomach are found actually bloated with milk. When fully extended, the skin of the abdominal area becomes remarkably tight-fitting; but elsewhere the trunk does not nearly fill out the skin. By holding such a specimen, back down, on one’s palm, and slightly tilting the hand from side to side, the entire contents of this out-size skin slop about as a quantity of mercury would if placed in a pliable bladder.
The largest young that I have found in the nesting-burrow measured thirteen inches in length. The smallest known by me to have been found at large were fourteen inches long, captured in shallow water among weeds. My own youngest capture at large measured fifteen inches. It would thus appear that the young are generally from thirteen to fourteen inches long when they leave the burrow. For some time prior to this, however, they would seem to run about the burrow, and, possibly, to enter the water in company with their mother. When once their eyes are open, the young are able to swim. The mother then no longer replaces the pugs in her tunnel, and one which contains large furred young may be recognized during excavation by the fact that nesting-material is strewn along its course. This is mentioned casually by Bennett (1860, p. 130), and it seems probable that this material is scattered about by the young in running to and fro. At this stage the breeding-burrow is about to be deserted.
Bennett (l.c., p. 131) records the capturing of twin young, ten inches in length, which had a most beautiful, sleek, and delicate appearance, and seemed never to have left the burrow. These were kept alive for some little time, and enjoyed playing in a dish of shallow water, in which, however, they never remained 190longer than fifteen minutes at a time. It would thus seem likely that platypus of that age have already begun to make short excursions into the water. Bennett captured a female which he assumed to be the mother of these, and found her to be in a very poor condition, with the mammary glands practically dry. Two healthy young, ten inches in length, would certainly need more nourishment than could be supplied by glands in this condition; but possibly they had just previously sucked the mother dry. But we may assume that the young are by this time learning to eat the food of the adults, and that, as this is always eaten in the water (unless taken from the mother’s mouth, pigeon-fashion), they go to the water to be fed. Verreaux’s observations are certainly interesting, but I cannot entirely agree with him. He records (1848. p. 131, Owen’s translation):—
“I redoubled my attention and care, and by dint of perseverance, having at my disposal (always on the banks of the New Norfolk) a pretty considerable number of adults and young, I saw the latter accompany their mothers, with which they played, especially when they were too far from the bank to take their nourishment. I distinguished very well that when they wished to procure it they profited by the moment when the mother was among the aquatic plants, near the land, and where there was no current. The female having her back exposed, one can easily conceive that on the exercise of a strong pressure, the milk would float to a little distance, and that the young might suck it up with facility; this it does, turning about so as to lose as little as possible. The manoeuvre is the more easy to be distinguished, since one can see the beak move with rapidity. I cannot better compare the greasy liquid of the female than to the iridescent colours produced by the solar rays upon stagnant water. I have witnessed the same fact repeated daily and nightly. I have also remarked that the young, when it was fatigued, climbed upon the mother’s back, who brought it to land, where it caressed her.”
It is improbable that the female discharges milk into the 191water. Most likely she catches and crunches food for her young, and releases it into the water where they can pick it up; this would be good schooling for both swimming and diving. It is possible that Verreaux witnessed some such process, though he mistook its nature.
I have observed a young captive platypus feeding upon an aquatic plant, stripping the leaves one after another from the stem with its mouth, and using its fore-paws to assist in the process. The adult may disdain a vegetable diet; but, if any significance can be attached to this single observation, it may be that the young devour a certain amount of vegetable food, for which they can forage for themselves in the shallows. Bennett’s aborigines, who were not altogether reliable concerning what took place in the burrows, informed him that the animals ate water-weeds, and that the mother fed the young first with milk, and then with comminuted insects and molluscs (1860, p. 131).
The furred young are more vocal than the adults, and, when disturbed or hungry, indulge in a puppy-like growling, accompanied by a kiss-like popping sound. Bennett records a squeaking call, which was answered; this I have never heard. In a passage quoted on p. 160 he describes the careful toilet which they make. They habitually sleep in a curious position, sitting up upon their hind quarters, with the muzzle laid flat against the chest, and the tail drawn up over it (see Plate 28).
Little is known of the age at which the platypus reaches sexual maturity. Judging from the size which it attains in a couple of months, one would imagine that it could breed by the following season. Against this, however, is Semon’s opinion that only those two years old, or older, take part in breeding. Semon’s observations are based upon the examination of a long series of individuals; but I collected a female in “full milk,” on October 27, 1922, measuring 407 mm. (16 inches), only four inches longer than a 12-inch nestling of my collecting. To me this is rather perplexing, seeing that the nestling would 192grow another inch or two more before deserting the nest. And who can say that that was her first breeding-season? Therefore I cannot agree altogether with Semon’s statement.
All that can be said at present is that the female begins to breed when sixteen inches in length, and finally reaches a maximum size of eighteen inches; but there is no precise information as to the length of time involved.
The length of life of the platypus is not known. It is my intention to ring-mark some fully-furred young as opportunity offers, and it may be that we shall gain some information on this point at a later date, if these marked individuals are captured.
193
So long ago as 1819, Barron Field published the despondent couplet—
Since his day Cassandras have not been wanting. Spicer (1876, p. 166) writes from Tasmania:—
“This is all I have been able to bring together anent Platypus and his spur, and little enough it is. I dare say when Australia is more settled, and Ornithorhynchus has been improved off the face of the earth, biologists will have leisure and thought to bestir themselves to enquire into the matter. Just as now we are searching for Dodo’s bones, and writing books about them, and doing work which ought to have been done two centuries ago.”
Wilson and Hill (1908, p. 33) express their views as follows:—
“The animal itself, though pretty widely distributed, and probably still far from becoming extinct, is to be found, in any one locality, only in comparatively small numbers. It is now much less plentiful than formerly, owing to the demand for its fur—a demand which is still satisfied in spite of the measure of legal protection which the animal has obtained in the various Australian States. The depredations of the fur-hunter are not easily repaired, since the animal breeds only once in the year and produces but two eggs at a time.”
Lucas and Le Souef (1909, p. 144) comment on the protection extended to the animal by the States of New South 194Wales and Victoria, and add—“so we may hope that his tenure of existence may at least be considerably prolonged.”
The platypus is probably in no present danger of extinction; given a reasonable measure of effective protection, it is likely to last as long as the waters in which it lives. Quite apart from the measure of safety resulting from its secretive ways, it dwells in mountain streams running through many hundreds of miles of almost entirely uninhabited ranges, much of which never will be inhabited owing to the inhospitable nature of the country. Its aquatic habit saves it from the action of what has been the chief means of bringing about a diminution of the marsupial fauna—the opening up to pastoral and agricultural settlement of a great part of the country. Australia is not guiltless as concerns the destruction of marsupials, but it is not generally recognized how much of this destruction was inevitable. For that which was not inevitable—the wholesale destruction of grass-eating marsupials simply because they ate grass intended for sheep and cattle—the pastoral industry is already being called to account. The dingo, deprived of its natural marsupial food, is attacking flocks in an unprecedented manner, and greed and stupidity have defeated their own ends. The destruction of forests has brought with it the extermination of their tree-living inhabitants, as well as of the smaller terrestrial forms which sheltered beneath the trees. But the platypus has not been affected by these things.
One thing, and one thing only, can bring about the extinction of Ornithorhynchus, and that is the permitting of open traffic in its skin. The skin is small; but, in the present world-shortage of fur-skins, the beauty of the fur is sufficient to create a keen demand for it, if it once be made available. The Australian bushman is well able to follow the animal to its ultimate fastnesses if it be worth his while; but he is patriotic enough to refrain from slaughtering Australian fauna when his reason is appealed to.
At the present time the platypus is, and for some years has been, completely protected in all the Australian States in which it occurs. The laws of the different States vary in 195detail; but, after years of effort on the part of individuals and societies interested in animal preservation, they have been brought closely into line and all except certain scheduled native animals are protected—the eaters of grass and similar heinous offenders. Even these may be granted the protection of a close season, or of a period of absolute protection, in a district in which they are becoming depleted, at the discretion of the minister administering the Act. On paper, then, the future of the Australian fauna appears to be a rosy one.
Legislative enactment does not, however, entirely prevent traffic in platypus skins, and there is no doubt that large numbers are smuggled out of Australia under wrong descriptions, mixed in with parcels of rabbit and other small skins. The extent of this traffic cannot be ascertained, owing to the perfectly natural reticence of the people who engage in it.
Fortunately it is only in comparatively recent times that the skin of the platypus has become commercially desirable, and the growth of a national sentiment, together with the increasing difficulties placed in the path of the poacher, have combined to keep destruction down to a minimum. A certain amount of thoughtless and wanton destruction is almost inevitable, since youths in the country districts of eastern Australia grow up in an atmosphere of callous indifference to the lives of individual animals. This indifference is simply the natural outcome of large-scale pastoral operations in a country subject to sudden meteorological vicissitudes. Men who have grown accustomed to seeing hundreds of animals dying, which they are powerless to aid, cannot be expected to become sentimental about a platypus. In 1863 Gould (p. 1) wrote:—
“the diminution in its members is solely due to the wholesale destruction dealt out to it by the settlers, which, if not restrained, will ere long lead to the utter extirpation of this harmless and inoffensive animal, a circumstance which would be much to be regretted; it is in fact often killed from mere wantonness, or at most for no more useful purpose than to make slippers of its skin.”
According to Semon (1894, p. 12):—
“The fur is beautiful196 and thick, yet its qualities are not so good as to warrant the wearisome and deluding pursuit. Consequently the Duckbill is little pursued either by whites or blacks, and the early extinction of this paradoxical creature need not be feared.”
At present there is little danger of an export trade being developed; the Federal minister for customs can prevent the export of animals and animal products by the simple process of issuing an order-in-council. There is no doubt that the measure of protection enforced by the States during the last decade has had a valuable effect in increasing the numbers of platypus. A healthy popular sentiment in favour of Australian birds and mammals has also had a most gratifying growth during the same period. Books on Australian natural history are numerous, and additions are constantly being made to their number, whereas twenty years ago there were none of a popular character. The next generation of Australians will look upon its fauna with different eyes, and it is not likely that the platypus will be deprived of the protection it now enjoys.
In one respect, however, enthusiasm for animal protection is beginning to prove embarrassing, since accredited representatives of scientific institutions are finding it increasingly difficult to obtain permits to collect specimens for scientific purposes. The platypus is, from a scientific point of view, perhaps the most important mammal that exists, and a great deal of anatomical investigation remains to be done. The number of individuals required for scientific investigation is negligible when compared with the ordinary annual wastage. Moreover, zoologists are more interested than any other class of men in the continued existence of the animal, and are therefore least likely to threaten that existence. The rights of science should be safeguarded in any scheme of animal protection; but the enthusiastic people who raise their voices loudest for the preservation of native fauna have not, as a rule, a sufficient knowledge of science and its aims to appreciate those rights. They desire that no wild innocuous animal whatever shall be killed—which is in some respects a very laudable desire; but such an attitude betrays the want of a sense of proportion, and of knowledge 197of the balance of nature. It has been alleged quite seriously that collectors for overseas museums, though operating under jealously restricted permits, were bringing rare species within danger of extinction; and there was some outcry against the expedition on behalf of the British Museum being allowed to collect at all. Such allegations are ludicrously far from the truth, but they seem, when made loud and often, to carry a certain amount of weight with authorities who do not ask for responsible advice.
Natural enemies of the platypus appear to be few in number. The carpet-snake (Python variegatus) occasionally catches one, as has been found on opening up a gorged snake; but the toll it collects must be quite negligible, as a constricting snake could not take its prey in the burrows, and must depend upon their approaching its lurking-place on the open bank. It is probable that the omnivorous monitor lizard (Varanus varius) takes platypus on land, and possibly the Whistling Eagle and the Murray cod, in the water; but of this I have no definite evidence.
Its principal enemy at the present day, apart from man, is without doubt the introduced rabbit, which by a process of peaceful penetration has riddled banks where once the platypus enjoyed a quiet seclusion. I hold the opinion that the presence of rabbits tends in time to drive platypus from their accustomed haunts, and without suitable soil for brooding burrows they cannot multiply. I attribute their disappearance from settled regions almost entirely to this cause. The water-rat (Hydromys chrysogaster), which also burrows in the banks of streams, is in a less degree a disturbing factor.
The rabbit is followed by the trapper, and rabbit-traps set on river-banks and elsewhere are responsible for destruction of both the platypus and the echidna. The latter, being useless from a commercial point of view, is usually released, and frequently heals its wounds with reptilian indifference. The trapped platypus is, however, knocked on the head and relieved of its skin—for which proceeding there is at least this justification, that it would not be likely to survive its injuries, and, 198worse still, even the skin is destroyed when the whole carcass is thrown into the river to rot.
The greatest menace to the animal is, however, the fish-trap. Despite the fact that in most waters the use of traps is illegal, most homesteads on a river-frontage make use of traps to catch food for home consumption. These are heavy wire cages. They have a conical entrance, through which fish pass in, but are unable to escape. Platypus, in their blind nosings about the bottom, enter these traps simply by chance; as they cannot reach the surface to breathe, they are drowned. Naturally, since the animals are dead when the trap is examined in the morning, their skins are taken. It would be a simple enough matter to save the lives of the animals thus accidentally caught, if the users of fish-traps would take the trouble. An elongated funnel-shaped outlet of wire-netting, fixed vertically to the upper part of the trap, with the top portion open above water, would allow platypus, water-rats, and tortoise to escape by climbing up the spout, and still retain the fish. The device is quite simple to construct, and is also inexpensive.
Closer settlement will bring about the disappearance of snakes, lizards, rabbits, and rabbit-trappers. It may bring about an adequate regulation of fish-traps. The more important question is—Will it bring about the disappearance of the platypus? I do not see any reason why it should, if settlers will but refrain from the grosser methods of disturbing the creature. If shot at and hunted, either in wantonness or for their fur, the survivors will undoubtedly desert the waters in which they have been harried. But if a favourable public sentiment can be aroused, and some interest taken in the preservation of an interesting animal in the midst of closely settled communities, there is little doubt that the platypus will contrive to flourish, despite settlement. At Belltrees on the Hunter River in New South Wales, Mr. H. L. White—to whom Australian natural history owes a great debt—has made the homestead area a sanctuary, and the animal flourishes in those pleasant reaches of the river which meander through the lucerne flats of the home farm. Belltrees is quite a village, but the mere presence 199of many men, engaged in activities about the river-banks, has not frightened the platypus away.
Economically there is little to be said either for or against the platypus. It does no positive good; neither does it do any positive harm. It has a valuable fur, but neither exists in sufficient numbers, nor breeds sufficiently rapidly, nor lends itself to any scheme of cultivation in such a way as to hold out hope of its becoming an important national asset. It is not good to eat, and apparently serves no purpose useful to man.
The value of its fur is difficult to arrive at, since there is no open trade in it. During the Pan-Pacific Conference in 1923 a Sydney naturalist’s shop had a window-display of skins, priced ‘from thirty shillings each.’ Platypus rugs—the staple form of use of the fur in Australia in the past—rarely appear in auction-rooms now, but good ones, containing from fifty to sixty skins, are usually sold at about a guinea a skin. Should platypus fur ever be allowed to come into the open market, there is little doubt that prices much higher than those quoted would be obtained—and no doubt that the animal would be in danger of early extinction.
Mr. Clifford Coles has kindly supplied me with the following notes on the skin from a furrier’s point of view:—
“The fur of the platypus is without exception the best-wearing of all Australian furs. It is short, dense, glossy, and hard to the touch; in colour, dark brown down the back, graduating to a silvery whiteness on the flanks. Being somewhat even in length over the whole of the body, the utmost use can be made of the skin. The surface-hairs are very much coarser than the under-fur, which is dense, soft, and practically impervious to water. The skin is not in much request, primarily because of the thickness of its pelt rendering the finished article somewhat stiff. Modern methods have made this difficulty easy to overcome, but the reputation which the skin of the platypus obtained in the years when it was an article of commerce still holds with it, and the later generation, who only 200know the platypus of old, always think of it as a heavy-pelted fur and consequently do not seek it.
“One of the principal drawbacks to its use as a fur is the difficulty of manipulating the skin without leaving the furrier’s ‘cut’ marks in ‘dropping’ conspicuous to the view. The art of the furrier is to-day a great feature in fashionable furs. The skins, instead of being split and divided into sections end on end, are now, by a series of ‘V’ cuts, called ‘drops,’ altered to any shape desired. This is a matter of great difficulty in a fur having a short texture, as the knife-marks are conspicuous; and it is this fact which renders the platypus so unattractive to the furrier.
“The average woman of to-day, if shown a fur made of rabbit-skin properly dressed and dyed to a fashionable colour, and another of similar size made of platypus, would choose the rabbit-skin—not because she would be protecting one of the rarest animals in the world by the choice, but because the rabbit looks better and is very much softer to the touch.
“Platypus skins can be plucked so that the outer coarse hairs are entirely removed. When this is done, a soft and most delightful fur is obtained, closely resembling otter. In this form it is most durable. The writer has known platypus skins used in this way which have not been worn out after twenty years of service.
“Because of the restrictions which have been placed upon the capture of the animals, generally speaking, the skins are not on the market. One can go for a whole season through all the wholesale stores of Sydney and not see nor have offered him a single platypus pelt.
“Tasmania produces the finest quality of these skins. The fur there is longer, and the skins very much larger, than those from the mainland. Reports from fur-trappers all along the eastern watershed of Australia and Tasmania indicate that the animals are increasing[17] in numbers.”
The flesh of the platypus must be extremely distasteful, 201since neither aborigines nor dogs will eat it. Bennett certainly states that the eyes of his aborigines glistened at the sight of fat young ones in fur, taken from the burrow, and that they said the animals were very good to eat; but he does not record any definite instance of their eating platypus flesh, and it may be that the sight of plump young animals called forth the remarks, and not any previous gastronomic experience. Semon (1894, p. 12) writes:—“The blacks on the Burnett do not hunt the animal, since they scorn its flesh entirely; as a matter of fact it gives forth an objectionable smell when skinned.”[18] A correspondent of the Sydney Daily Telegraph (10 February, 1923) records that some miners ate one, and found it a somewhat oily dish, with a taste between those of red herring and wild duck. The objectionable smell is due to scent-glands situated at the base of the neck; but it is unlikely that, even after these have been removed, the platypus will ever be a popular item on a bill of fare.
On the negative side, the only accusation brought against the animal is that by Jamison (1818), that he found ova and fry of fish in the platypus he examined. No one has repeated that observation. From the discussion of the animal’s feeding habits given earlier, it will be obvious that it is not likely to catch much in the way of actively-swimming fry; and its habits in captivity show that it does not care about fish as food. It may eat a certain amount of demersal spawn (though there is no satisfactory evidence that it does), but such a diet would be more or less accidental. Consequently its presence in streams which have been stocked with introduced trout cannot be said to be a danger to the fish. Large numbers of both platypus and trout are said to co-exist in the Tasmanian lakes.
202
Australian mammals have so far proved disappointing. The dingo will certainly parade his enclosure by day, but he is just a dog, after all. The kangaroos and wallabies show well enough, but one is very much like another. The long series of interesting and beautiful marsupials, which one would much like to see in our Zoological Gardens—pouched mice, flying ‘squirrels,’ ‘opossums,’ bandicoots, and others—are not only nocturnal, but so cryptozoic that they are rarely seen, even in the bush. In the ‘Zoo’ they are represented merely by labels on apparently untenanted cages, though occasionally one may catch a glimpse of something furry rolled up in a ball in the heavy shadow of the sleeping-hutch. Perhaps some day we shall have a special Marsupial House, lit by powerful electric lamps all night, to simulate daylight and drive our fauna to slumber, and darkened down by day to the intensity of bright moonlight, in which we may come to know the grace and beauty of the smaller arboreal forms.
In September 1921 I sent two living adult and three nestling platypus to the Zoology Department at the University of Sydney. The limited number of people who on that occasion were privileged to see the platypus swimming and diving in a large glass-fronted tank were all agreed as to the interest and beauty of the exhibition. The loveliness of the fur on the ventral surface, with its gold and silver lights as seen under water, was certainly something to remember. This same fur, by the way, is described in the British Museum Catalogue of Mammalia as ‘dirty-white’—an indication of the difference between the stuffed and the living animal.
The platypus can be kept alive in captivity; and it is very 203probable that, with sufficient pains and interest, it can be satisfactorily exhibited. As it is probably the most interesting animal in the whole world, it would certainly be worth while to make the attempt.
Until quite recently, although the animal has been kept alive for fairly long periods in Australia, no living platypus had ever been exported from the country. Chenu (1879, p. 351) certainly writes:—“Ces animaux longtemps rares dans nos collections européennes y sont actuellement plus fréquemment apportés, et l’on en a même de vivants, surtout en Angleterre;” but there is no truth in the statement. It is probably founded on lying reports of rascally dealers. Bennett relates one such case, in which it was reported that a loud quacking noise, like that of a duck, was heard coming from an East London pond; that a gentleman struck with his stick at the spot from which the noise was proceeding; and that some hours later a fine male Ornithorhynchus was found dying on the bank. It was now stuffed, and might be seen in Mr. So-and-so’s shop.
Maule (P.Z.S., 1832, p. 146) says:—
“in one of the nests he was fortunate enough to secure an old female and two young. The female lived for about two weeks on worms and bread and milk, being abundantly supplied with water, and supported her young, as it was supposed, by similar means. She was killed by accident on the fourteenth day after her capture.”
Verreaux (1848, p. 133) had, of course, no difficulty whatever in keeping them in captivity. During all the time that he was making his important and painstaking series of observations, he had platypus of all sorts and sizes in his possession. He fed them on broken rice mixed with yolk of egg—which, after a time, they preferred to their natural food. They fed at night, and growled and got angry if no food was provided. Seeing how easy it all was, and how glad the French zoologists would have been to welcome them, it is remarkable that Verreaux did not take his pets back to France, or even send them by a reliable hand. But there is no record of his making the attempt.
Dr. George Bennett was very anxious to send the living 204animal to Europe and made an ill-planned effort to accustom the platypus to captivity; but, as he evidently had not studied its feeding habits, he was not successful. He gives an extended account of his attempts in his Gatherings of a Naturalist, from which these few paragraphs are quoted:—
“I arrived with my little family of Ornithorhynchi safe at Sydney, and as they survived for some time, an opportunity was afforded me of observing their habits. The little animals appeared often to dream of swimming, for I have frequently seen their fore-paws in movement as if in the act. If I placed them on the ground during the day, they ran about, seeking some dark corner for repose; and when put in a dark place, or in a box, they huddled themselves up as soon as they became a little reconciled to the locality, and went to sleep. I found that they would sleep on a table, sofa, or indeed anywhere; but, if permitted, would always resort to that spot in which they had previously been accustomed to repose. Although for days together they would sleep in the bed made up for them, yet on a sudden, from some unaccountable caprice, they would shift their resting-place and seek repose behind a box, or in some dark retirement, in preference to their former habitation. They usually reposed side by side, looking like a pair of furred balls, and surly little growls issued from them when disturbed; nevertheless, when very sound asleep, they might be handled and examined without evincing any signs of annoyance. One evening both the little pets came out about dusk, went as usual and ate food from the saucer, and then commenced playing, like two puppies, attacking each other with their mandibles, raising their fore-paws, and tumbling one over the other.... In the struggle one would get thrust down; and at the moment when the spectator would expect it to rise again and renew the combat, it would commence scratching itself, its antagonist looking on and waiting for the sport to be renewed. When running, they were exceedingly animated, their little eyes glistened, and the orifices of their ears contracted and dilated with rapidity; if taken into the hands at this time for examination, they struggled violently to escape, and their loose integuments 205rendered it difficult to retain them. Their eyes being placed so high on the head, they do not see objects well in a straight line, and consequently run against every thing in the room during their perambulations, spreading confusion among all the light and easily overturnable articles ... besides combing their fur to clean it when wet, I have seen them preen it with their beak (if the term may be allowed) as a duck would clean its feathers. It is, indeed, interesting to watch them engaged in the operations of the toilet, by which their coats acquire an increased bright and glossy appearance. When I placed them in a pan of deep water, they were eager to get out after being there only a short time; but when the water was shallow, with a turf of grass placed in one corner, they enjoyed it exceedingly. They would sport together, attacking one another with their mandibles, and rolling over in the water in the midst of their gambols; and afterwards, when tired, get on to the turf, where they would lie combing themselves, until the fur was quite smooth and shining. It was most ludicrous to observe these uncouth-looking little creatures, running about, overturning and seizing one another with their mandibles, and then, in the midst of their fun and frolic, coolly inclining to one side and scratching themselves in the gentlest manner imaginable. After the cleaning operation was concluded, they would perambulate the room for a short time, and then seek repose. They seldom remained longer than ten or fifteen minutes in the water at a time. As they were not confined during the night, I sometimes heard them growling; they seemed as if they were fighting or playing, and as if the saucer containing their food had been upset in the scuffle; but, on the following morning, they were quietly rolled up, fast asleep, side by side, in the temporary nest I had formed for them.... One evening, when both were running about, the female uttered a squeaking noise, as if calling to her companion, which was in some part of the room behind the furniture, and was invisible; he immediately answered her in a similar note; and marking the direction from which the answer to her signal came, she ran at once to the place where he had secreted himself.
206
“It was very ludicrous to see the uncouth little creatures open their mandible-like lips and yawn, stretching out the fore-paws and extending the webs of the fore-feet to their utmost expansion. Although this was natural, yet, not being in the habit of seeing a duck yawn, it had the semblance of being perfectly ridiculous. It often surprised me how they contrived to reach the summit of a book-case, or any other elevated piece of furniture. This was at last discovered to be effected by the animal supporting its back against the wall, placing its feet against the book-case, and thus, by means of the strong cutaneous muscles of the back and the claws of the feet, contriving to reach the top very expeditiously. They often performed this mode of climbing, so that I had frequent opportunities of witnessing the manner in which it was done. The food I gave them was bread soaked in water, chopped egg, and meat, minced very small. Although at first I presented them with milk, they did not seem to prefer it to water.
“Some time after my arrival at Sydney, to my great regret, the little creatures became meagre, and their coats lost the sleek and beautiful appearance which had before called forth so much admiration; they ate little; yet they ran about the room as before, and appeared lively. But these external symptoms argued strongly against their being in a state of health. When wet, their fur became matted, never appearing to dry so readily as before; and the mandibles, and indeed every part of the animal, indicated anything but a satisfactory condition. How different was their appearance now, from the time when I removed them from the burrow! then their plump and sleek appearance roused even the apathetic blacks; now the poor creatures could only excite commiseration. The young female died on the 29th of January, and the male on the 2nd of February, having been kept alive only during the space of nearly five weeks; and thus my expectations of conveying them to Europe in a living state were frustrated, and the ladies of England lost an opportunity of beholding these really ‘darling little ducks’ of quadrupeds.”

From my experience with platypus in captivity, I think 207the so-called engaging antics of Bennett’s captives were really the desperate struggles of slowly starving nestlings, while the food supplied and general treatment were quite the reverse of their requirements.
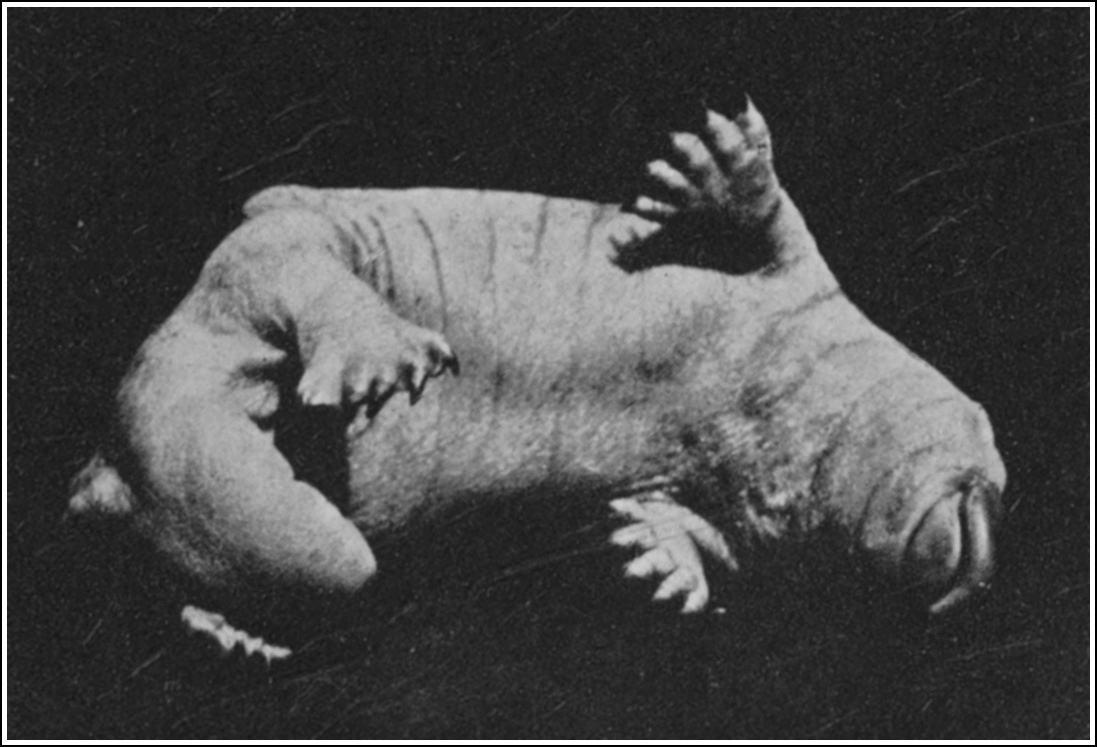
I settled at Manilla, on the Namoi River, in northern New South Wales, in 1901, and soon established a small zoological garden, in which I kept a variety of marsupials and birds. While engaged in getting water-weeds for my wild ducks, I met my first platypus, and confess to having fallen in love at first sight. From this first meeting, the idea of establishing a platypus amongst my pets never left my mind. I wrote to the zoological authorities in Sydney, asking for advice how to proceed with my plan, but received the discouraging reply that the animal would not live in confinement, and that if I felt disposed to experiment for myself, I should give a milk diet. Far from being discouraged, I became the more determined to attempt this apparent impossibility. Local people could give me no precise information as to the food on which the platypus normally subsisted, until one day I learned that one of the creatures had been caught upon a fishing-line baited with a worm. Here was a ray of light on the food-question, and I at once set about devising a suitable vivarium, in which to keep a supply of worms. A kerosene-tin with small perforations in the bottom, filled with alternate layers of grass-roots and earth, and topped off with a layer of grass (which was renewed daily), served admirably. This was hung in a shady place, and kept moist, and the worms throve in it.
The first enclosure used (Plate 34, top figure) consisted of a brick structure, roofed over, and floored with a thick layer of river-earth—to represent the burrow—which was connected by a submerged tunnel with a cemented pond, also fitted with a cover, the top being made of wire-netting. The experiment was begun on 14 January, 1910, with a female which had been trapped in the river. Within a short space of time two more females and two males were added. Here I recorded the interesting fact that, when the males (which were caught 208together in the same trap) were placed in the tank, they circled round a few times, then entered the submerged tunnel and made their way to the ‘burrow,’ where they were received with growls and muffled snorts by the females, who evidently resented the intrusion. This was the first occasion upon which I had heard an adult platypus utter a vocal sound. To find a sufficient supply of food for five animals—two more specimens were added later—was a difficult problem. I worked six hours daily, with mattock and shrimping-net, and served my captives with a mixed diet of earth-worms, freshwater shrimps, larvae of scarab beetles, and pond-snails, supplemented by a certain amount of water-weed, and fresh water daily. But my utmost endeavours succeeded in producing only about two pounds of animal food a day; and this, apparently, was not enough. One by one the animals died, until there was left only the original female, ‘Biddy,’ with whom the experiment had begun. This powerful animal escaped by tearing her way through the wire-netting covering the tank, after having been in captivity for nine weeks and five days.
So ended the first attempt, but it pointed the way to success. The most important discovery arising out of it was made during the period of Biddy’s sole survivorship, when it was found that she could eat quite comfortably the amount of food that had been thought sufficient for herself and her four companions. It had not previously been suspected that a platypus could eat half its own body-weight of food in the course of a single night. The natural food, and the order of its preference, had also been ascertained—shrimps, earth-worms, and insect larvae, in that order; tadpoles in default of something better; molluscs and water-weeds only very sparingly, even when the creature was hungry. River fish she would not touch, even though shrimp-size and served alive.
Consideration was now given to the question of improving the enclosure. In the first primitive structure the water was contained in a cemented tank. Food had to be given alive in this tank; as the animal invariably defaecates in the water, it rapidly became foul, and had to be changed each morning. 209Running water being out of the question where the sole supply consisted of rain-water caught in tanks, some better system of pond had to be devised, which would allow of easier emptying and renewal.
The sleeping-compartment had not been opened during the time of the first experiment. It was, as has been said, built with brick walls and a cemented floor, and was filled with river-earth. When the cover was removed, the interior was found to be in a foul and dank condition, the water brought in by the animals on their bodies having converted it into a bog, with a few mounds of damp soil projecting above the general level. Obviously such a retreat could not have been healthy for the captives. The next fault lay in the use of brick and cement, which caused sores upon their knuckles. Finally it was noted that, as long as the animals were able to see anything of their terrestrial surroundings, they spent all their time trying to escape; and it was resolved that wire-netting should be used only as top cover, in such a way that clear sky alone could be seen through it.
Coupled with a wish to improve the enclosure in these different directions, I next considered the question of portability. The outcome of my deliberations was the designing and construction of a portable artificial habitat, which I called a Platypusary, and which Dr. W. T. Hornaday, Director of the New York Zoological Park, subsequently described as “the most amazing contraption that we have ever seen in use in animal transportation.”
The nature of the invention may be gathered from Plates 34 and 35. It was planned, as in the first attempt, in two distinct parts—a tank to represent a river, and a labyrinth to simulate the burrow in the bank, the two being connected by a sheet-metal tunnel. The general arrangement is best seen from above (the view shown in Plate 35). The tank was to the left, the labyrinth to the right. Taking the latter first, it will be seen that an animal coming from the tank through the tunnel would enter the small chamber at the upper left-hand corner. Here it would rid itself of some of the water from its body, 210and possibly rest awhile and chew some of the food in peace and comfort. It would then pass on through a series of holes cut in the partitions, first passing through an aperture in the short longitudinal partition, then through one at the right-hand end of the upper transverse partition, and so on, the holes in the remaining partitions being alternately left and right, until it reached the sleeping-chamber shown at the lower right-hand corner. It will be seen that, altogether, it must pass through six of these holes, each of which was guarded by a soft rubber gasket with an aperture smaller than the body of the animal, so that the wet fur was effectively ‘squeezed’ and the animal arrived at its sleeping-quarters with fur almost dry. Not only was this essential to its comfort, but it was necessary also to prevent, to some extent, the observation glass above the burrows from becoming so moist as to obscure the view when exhibiting the sleeping occupants.
A second feature of the labyrinth was that the apertures through the transverse partitions were placed progressively higher up, and the floors of the runways sloped up accordingly, so that the platypus had to climb a kind of zig-zag ramp on its way to bed. This was a simulation of the natural rising course of its burrow in a river-bank; and, though some critics suggested that it was over-elaborate, my experience of the platypus in captivity is that no precaution can be too elaborate (especially during a test case) for the successful treatment of a creature so delicately organized.

The labyrinth was further improved by using curved mouldings in all the corners, so that no right angles might remain, and by fastening sheet rubber on the floors of the runways, to minimize injury to the animal’s knuckles and feet. It was covered with a wooden lid, beneath which was a framed sheet of plate-glass, permitting inspection of the burrow.
The tank portion was of much larger size, and was constructed of galvanized iron, set in a wooden crate, the sides of which were carried up to such a height that animals in the water could see nothing but the sky above them. To the right is the exercising and feeding-tank. This, at the bottom, contained 211sand and shell-grit. Of course, mud would have been preferable, but it would have tended to discolour the water, and, without a continuous supply of fresh water, the animal would have died. The metal drum in the middle, the flat top of which projects three inches above the water, forms an island upon which the captive may rest, but its special purpose is to give the animal an endless swim. To the left is a smaller tank in which aquatic plants are grown in mud. The space at each side of it was filled with clean sand to form two banks, on which the animal could exercise by digging, or take a rest, at will. A continuous stream of water could be run from the main tank into the mud tank, which was kept at a constant though lower level (to prevent pollution of the feeding-tank) by means of an overflow pipe. The platypus is intolerant of water which is not clean—hence the necessity for clean sand and flowing water, where these can be provided. The doors covering the tank were made with wooden frames, upon which wire-netting was stretched, so that the water was open to the sky, day and night, in all weathers.

By means of this enclosure I succeeded during 1910 in achieving my aim of placing the platypus on exhibition in the Sydney Zoological Gardens, then at Moore Park. One animal lived for three months in good condition; but on the approach of winter it became difficult to secure for it a supply of natural food, and it was released in one of the ponds of the Centennial Park. Discouraged by what I considered to be a lack of interest in my endeavours, I took no further measures for some time, and my contraption was scrapped.
In 1913, however, I interviewed Ellis Stanley Joseph, a well-known animal-dealer, who became keenly interested in a scheme for taking a live platypus to America. I at once recovered my discarded artificial burrows, etc., from the scrap-heap, and set them up again for Joseph at his Moore Park vivarium. Here I instructed him in the art of feeding and caring generally for the appetites and other requirements of captive platypus. Owing to his occasional absence, however, matters proceeded but slowly. Later, at Granville, two more of these enclosures 212were built for Joseph under my supervision. Subsequently a number of animals were obtained and kept for various periods. Joseph, in the New York Zoological Society Bulletin for September 1922, gives the following account of his experiences:—
“My first attempt to bring one to New York was in 1916, and I must say that it was not a fair trial, either to the animal or to myself. Mr. Burrell had secured one for me from the Namoi River, and it arrived in Sydney one day before I sailed on the S.S. Niagara, one of the Canadian-Australian liners. I kept it for exactly one week, and then unfortunately it died.
“On my return to Australia from the United States in February, 1917, I was unable to give the matter much attention, as I was too busy getting a collection together, and so deferred action until I had made another trip to America. When I returned to Sydney on October 27, 1917, I put all my energy into the keeping of the platypus alive in captivity. I secured two, and kept one for 96 days and the other for 125 days. Then I got several others from time to time and kept them for varying periods, from one week to over one year. On August 27, 1918, I secured one and kept him until April 26, 1919, on which date the animal died. I honestly believe that the animal would have lived longer had it been possible for me to give it my personal attention, because for three weeks before its death I saw very little of it. I was ill with the pneumonic influenza—raging at the time all over the world—and had to depend on others to see to his keeping.
“As soon as I was up, I secured several more specimens, and on one occasion I had three females and one male quartered together. To my sorrow I found that the male fought them and just worried them, and in one instance there were several scratches on the bill of the female which I am sure could not have been made except by the claws, or possibly by the spur, of the male. The females ultimately were afraid to go in their sleeping-box; for I put them in myself on several occasions in the course of a day, and they would struggle hard to get out. It seems to me that it was actual fear of the male.
“On August 22, 1919, I secured a fine male measuring 23½ 213inches, and during a period of nine months he was responsible for the death of no less than two females and one male. In August, 1920, I approached the authorities in Sydney for a permit to take this specimen to the New York Zoological Society, but was refused. Keenly disappointed, I left Sydney on September 3, 1920, on the S.S. Bellbuckle, bound for New York, without my platypus. On October 15, 1920, while I was still on the sea, the animal escaped from his tank because the lid had not been properly secured. I certainly would have liked to have kept him till he died, as it would have been very interesting to learn how long he could have lived in captivity.
“When I decided to go to the United States again, I felt that I should bring a platypus along, as I knew the great desire of the Zoological Park authorities in New York to obtain a specimen. At first things looked black for securing a permit from the Federal Government for the exportation.”
Here follows an account of his difficulties, which were finally solved through the good offices of the Federal member for Parramatta, Mr. E. K. Bowden, and Sir Baldwin Spencer. Joseph continues:—
“Having secured the Federal permits, I made arrangements to get the animals. I secured six, five of which were males. Unfortunately the female died sixteen days after I received it.
“I left Sydney with five male platypus on May 12, 1922, on the U.S.S. West Henshaw (Shipping Board Steamer), bound for San Francisco via Newcastle, N.S.W., and Honolulu. On May 19, while we were still in Newcastle, one of the medium-sized ones died. The remaining four were absolutely in the best of health as far as I could make out, because they were eating well; that being about the only sign by which I could determine their condition. We left Newcastle on May 20, and for six days it blew a regular gale. The seas were mountain-high, and unfortunately the ship was more under water than over it, and certainly that did the animals no good. On the night of May 25 a huge sea came aboard, and rushing along the deck smashed into one of the platypus-tanks, bent the metal neck and jammed it so effectively that the animals were imprisoned214 in the sleeping-quarters for that night. From then on that pair ate less and less, but they lived till the 5th and 6th of June respectively, on which dates they died in a comparatively poor condition.
“Of the remaining two, one was fine and healthy but the second was just about medium, and I am sorry to state that on the 14th of June while the ship was at Honolulu, it also died. My feelings can readily be imagined. I would rather have lost all of my shipment of a very valuable cargo of birds, animals, and reptiles. This was not because the platypus was worth more (far from it), but because it was my ambition to bring one alive to America. I am glad to say that good fortune eventually favoured me, since on June 30, 1922, I landed in San Francisco with the first living platypus ever brought to America.
“I had to stay five days in San Francisco in order to procure a new supply of fresh worms, and this I can assure the reader was not an easy matter. From Honolulu I had sent two wireless messages to different parties to try and get some for me, but to my great disappointment I found on my arrival in San Francisco that neither of them had been able to get any. After a lot of work, worry, and expense I managed to secure sufficient for the trip across the continent to New York. That trip was the hardest part of the long journey, for the shaking and the jolting of the train was a very great strain on the animal. When two days out from San Francisco, the platypus began to get hungry, for he used to get out whenever the train stopped and look for food, and so I was up day and night, and whenever the train stopped for any length of time I put water in the tank and also food, and that gave the animal a chance to eat a little.
“I was glad when we arrived in Chicago, where I rested for two nights, and that gave the animal a fresh start. I started on the last lap on Tuesday night and on Thursday, July 14, arrived in New York, both man and animal completely tired out.”
What Dr. Hornaday thought about the matter finds expression 215in his article in the same number of the Society’s Bulletin, some paragraphs from which are here quoted:—
“The spell of ten thousand years has been broken.
“The most wonderful of all living mammals has been carried alive from the insular confines of its far-too-distant native land, and introduced abroad. Through a combination of favoring circumstances it has been the good fortune of New York to give hospitality and appreciation to the first platypus that ever left Australia and landed alive on a foreign shore....
“No matter what evil fate may hereafter overtake the platypus species, nothing can rob us of the fact that New York has looked upon a living Ornithorhynchus paradoxus, and found it mighty interesting. It cost us $1400, but it was worth it.
“When our first platypus arrived at the Zoological Park on July 14, 1922, after a journey (of 10,000 miles) that had consumed the lives of four companions, we felt reasonably certain that when fairly out from under the excitement of foreign travel the queer little beast would die in a very few days. We hoped that it might live for one week, but we resigned ourselves to the impending loss. It lived at the Park forty-nine days, and was on exhibition daily for one hour.
“At the outset we observed that the platypus is an animal of nervous temperament, and easily excited by too many observing eyes. It was evident that one hour of daily exhibition was all that the little animal could endure, and subsequent observations proved the correctness of this estimate. We are sure that a longer exhibition period would speedily have proven fatal to the distinguished stranger....
“The exhibition of the platypus was accomplished by removing the wire-netting tops of the contraption, admitting visitors in a line, in single file, and permitting them to pass entirely around the man-made habitat of the animal. As the stream of visitors marched and countermarched, the platypus briskly swam and emerged, scrambled, and climbed up the wire-netting walls of its main salon in efforts to get out....
“The two features of a living platypus that make the daily 216life of its keeper a nightmare and a burden are its food habits and its water habits. In this vale of tears there seem to be just about five kinds of food that it will consider and consume. Named in the order of their acceptability these are: angle worms, very small shrimps, wood-grubs (of the kind most difficult to find!), oysters, and water insects. Of any one of the three leading kinds the animal soon tires, and requires a change. The cost of digging angleworms, even if it is possible to preserve them, is quite serious; and wood-grubs cost us ten cents each, with few purveyors.
“While it lived, our little platypus—about half grown—cost us between $4 and $5 per day to feed. Even in summer the food problem kept us moving, but we looked forward with apprehension to the horrors of winter.
“One trouble lies in the voracious appetite of a healthy platypus.... One day’s ration of our specimen was as follows:
“August 6; ½ lb. earthworms, 40 shrimps, and 40 grubs.
“One week’s food supply for the platypus consisted of the following:
“Each day, between three and four o’clock, the animal was given one-quarter of a pound of earthworms. On Tuesday nights and Friday nights one-quarter of a pound of shrimps and one-half pound of grubs were given. On the evenings of Monday, Wednesday, Thursday, Saturday and Sunday he received one-half pound of shrimps each day....
“Really, it seems incredible that an animal so small could chamber a food supply so large. I know of nothing equal to it among other mammals.”
During an experiment which I made in September 1910 to test the appetite of an adult platypus, I found that in the space of 72 hours it ate 70 ten-inch red worms (resembling an ordinary lead pencil in both length and girth), 10 ground-grubs, and 600 salt-water prawns (of the size usually bought in packets as bait). The record for one night was:—red worms, 30; prawns, 300.
Hornaday remarks on the fact that the creature showed itself, in captivity, to be of nervous and active temperament, a 217fact that must strike any observer who has had to do with it. That is why I would not allow my captives to observe any surroundings other than those within their enclosure. Because of this, I claim to have practically solved the problem of keeping platypus in captivity. By comparison the echidna is a dull animal, unless taken young and trained systematically for exhibition purposes. It does not pretend to be a gourmet, and thrives quite well on raw egg-and-milk, a diet utterly different from that to which it is accustomed when at large. True, it uses its very remarkable bodily strength to explore all possible ways of escape; but, once having found the task hopeless, it becomes resigned. It does seem to show an intermediate mentality, as much reptilian as mammalian, despite the fact that its brain has a convoluted surface, while that of the platypus is smooth. The natural food of the platypus resembles that of a bird, rather than that of a mammal. It demands a varied diet, and will starve to death in the presence of food which no longer pleases it. It must have clean, clear water, and sweet, dry sleeping-quarters. It is impatient of observation, and resents being handled. It is easily killed by too much excitement. These things bear witness to an organization of a higher grade than that of many of the lower Didelphia and Monodelphia, despite its Ornithodelphian plan.
This being so, is it possible to exhibit the animal satisfactorily in a Zoological Garden? I believe so, provided that the lessons to be learned from experience up to the present are fully realized. Young animals (not necessarily nestlings) which have never tasted the true delights of wild life, will pretty certainly take more kindly to confinement than adults. The questions of food and transport may be looked upon as solved. The method of exhibition, however, could be greatly improved. It is remarkable that the New York animal should have lived for so long as seven weeks, when for an hour each day it was surrounded by a continuous stream of curious visitors, and underwent constant handling by the keeper. Think of it!
What I suggest, apart from improved burrows, is a raised tank, with a plate-glass front. The eyes of the platypus are 218kept closed under water, and it would never see people standing below the water-level of its tank. It would thus be saved the greater part of the fret and excitement of the exhibition period. The animal would soon become accustomed to a regular feeding-time in the afternoon, and would not need the intervention of a keeper to turn it out into the exhibition tank. Given these conditions, there seems to be no reason whatever why the platypus should not live in Zoological Gardens at least so long on the average as other animals which are commonly kept in such places. Unfortunately, many of these do not survive so long as one might expect.

219
Allport, Morton. Notes on the Platypus (Ornithorhynchus anatinus): Report of the Royal Society of Tasmania, 1878, pp. 30-31.
Annales des Sciences Naturelles, ii, 1824, pp. 74-77—Quelques Observations nouvelles sur l’Ornithorhynque; x, 1827, pp. 193-195—Sur les Habitudes de l’Ornithorhynque. See also references to articles by Lesson, 1825, and Etienne Geoffroy, 1826 and 1829.
Antologia di Firenze, xxiv, p. 305, quoted in Annales des Sciences Naturelles, x, 1827, p. 194.
Armit, Wm. E. Notes on the Presence of Tachyglossus and Ornithorhynchus in Northern and North-eastern Queensland: Journal of the Linnean Society of London (Zoology), xiv, 1878, pp. 411-413.
Axford, T. Notice regarding the Ornithorhynchus: Edinburgh New Philosophical Journal, vi, 1829, pp. 399-400; id. Oken, Isis, viii, 1832, col. 806.
Baden-Powell, G. S. New Homes for the Old Country, 1872, pp. 296-314, 3 figures.
Bennett, George. Gatherings of a Naturalist in Australasia, 1860, pp. 94-146, 1 plate; Notes on the Natural History and Habits of the Ornithorhynchus paradoxus Blum.: Transactions of the Zoological Society of London, i, 1835, pp. 229-258, plate 34.
Bennett, George Frederick. Notes on Ornithorhynchus paradoxus: Proceedings of the Zoological Society of London, 1877, pp. 161-166, 2 figures.
Bewick, Thomas. General History of Quadrupeds, 7th edition, 1820, p. 528, 1 figure.
Blainville, Henri D. de. Dissertation sur la place que la famille des Ornithorhynques et des Echidnés doit occuper dans les séries naturelles, Paris, 1812; Sur l’organe appelé Ergot dans l’Ornithorhynque: Bulletin, Société Philomatique, Paris, 1817, pp. 82-84: Mémoire sur la nature du produit femelle de la génération dans l’Ornithorhynque: Nouvelles Annales du Muséum d’Histoire Naturelle, ii, 1833, pp. 369-416, plate xii.
Blumenbach, Johann F. Sur un nouveau genre de quadrupède édenté, nommé Ornithorhynchus paradoxus: Bulletin, Société Philomatique, Paris, ii, 1800, p. 113; De Ornithorhynchi paradoxi fabrica observationes quâedam anatomicæ: Voigt’s Magazin, ii, 1800, pp. 284-291; also article in iii, 1801.
Broderip, W. J. Ornithorhynchus: Penny Cyclopaedia, xvii, 1840, pp. 28-36, 16 figures.
Caldwell, W. H. The Embryology of Monotremata and Marsupialia, Part I: Philosophical Transactions of the Royal Society, Series B, vol. 179, 1887, pp. 463-486, 3 plates.
220
Chenu, Jean C. Monotremes: Encyclopédie d’Histoire Naturelle—Pachydermes, etc., 1879, pp. 349-358, 1 plate, 2 figures.
Collins, David. An Account of the English Colony in New South Wales, ii, 1802, pp. 321-328, or, second edition, 1804, pp. 425-428, 1 figure.
Crowther, A. B. On some Points of Interest connected with the Platypus: Papers and Proceedings of the Royal Society of Tasmania, 1879, pp. 96-99.
Cuvier, G. Recherches sur les Ossemens Fossiles, v, 3rd edition, Paris, 1825, pp. 143-158.
Darwin, Charles. A Naturalist’s Voyage ... Round the World in H.M.S. Beagle, 1884, pp. 441-2, or later editions.
Fleming, John. The Philosophy of Zoology, ii, 1822, pp. 213-215.
Fletcher, J. J. On the Rise and Early Progress of Our Knowledge of the Australian Fauna: Report of the 8th Meeting of the Australasian Association for the Advancement of Science, Melbourne, 1900, pp. 69-104.
Geoffroy Saint-Hilaire, Etienne. Sur l’Identité des deux espèces nominales d’Ornithorhynque: Annales des Sciences Naturelles, ix, 1826, pp. 451-460; Considérations sur les œufs d’Ornithorhynque, formant de nouveaux documens pour la question de la classification des Monotrêmes: ibid, xviii, 1829, pp. 157-164.
Geoffroy Saint-Hilaire, Isidore. Monotrêmes: Dictionnaire Classique d’Histoire Naturelle, xi, 1827, pp. 102-107; Ornithorhynque: ibid, xii, 1827, pp. 393-411.
Gould, John. The Mammals of Australia, i, 1863, pp. 1-4, plate i.
Hill, Patrick. [Observations on Ornithorhynchus]: Transactions of the Linnean Society of London, xiii, 1822, pp. 621-624.
Home, Sir E. A Description of the Anatomy of the Ornithorhynchus paradoxus: Philosophical Transactions of the Royal Society of London, 1802, pp. 67-84, 3 plates.
Hornaday, W. T. New York’s Duck-Billed Platypus: New York Zoological Society Bulletin, xxv, 1922, pp. 99-104, 15 illustrations; The Minds and Manners of Wild Animals, 1922, p. 9.
Illiger, C. Prodromus Systematis Mammalium et Avium, Berlin, 1811, pp. 114-5.
Jamison, Sir John. [Observations on Ornithorhynchus]: Transactions of the Linnean Society of London, xii, 1818, pp. 584-5.
Jones, F. Wood. The Mammals of South Australia, Part I, containing the Monotrêmes, 1923.
Joseph, Ellis S. My Experience with the Platypus in Captivity: New York Zoological Society Bulletin, xxv, 1922, pp. 105-111.
Kershaw, J. A. Notes on the Breeding Habits and Young of the Platypus, Ornithorhynchus anatinus, Shaw: Victorian Naturalist, xxix, 1912, pp. 102-106, 2 plates.
Knox, Robert. Observations on the Anatomy of the Duckbilled Animal of New South Wales, the Ornithorhynchus paradoxus of Naturalists: Memoirs of the Wernerian Natural History Society, v, 1823-4, pp. 26-41, plate i.
Krefft, G. The Mammals of Australia, plate xv and explanation, 1871; Catalogue of Mammalia in the Collection of the Australian Museum, 1864, p. 56.
Learmonth, Noel. [Natural history notes]. Australasian, Melbourne, 30 June, 1923, p. 1317.
221
Lesson, R. P. Observations générales d’Histoire Naturelle, faites pendant un voyage dans les Montagnes-Bleues de la Nouvelles-Galles du Sud: Annales des Sciences Naturelles (i), vi, 1825, pp. 241-266 [Ornythorhynchus, p. 249]; Voyage autour du Monde Enterpris par Ordre du Gouvernement sur la Corvette la Coquille, ii, Paris, 139, p. 302 and plate.
Lucas, A. H. S., and W. H. Dudley Le Souef. The Animals of Australia, 1909, pp. 143-146, 4 figures.
Mackenzie, W. C., and W. J. Owen. The Glandular System in Monotremes and Marsupials, 1919, pp. 11-35, 12 figures.
Martin, C. J., and F. Tidswell. Observations on the Femoral Gland of Ornithorhynchus and its Secretion; together with an experimental enquiry concerning its supposed toxic action: Proceedings of the Linnean Society of New South Wales, (2), ix, 1894, pp. 471-500, 4 plates.
Maule, Lauderdale. [Habits and Economy of the Ornithorhynchus]: Proceedings of the Zoological Society of London, 1832, pp. 145-6; id., Philosophical Transactions of the Royal Society, 1832, pp. 533-4.
Meckel, J. F. Ueber den Stachel und das Giftorgan des Ornithorhynchus: Deutsches Archiv für die Physiologie, viii, 1823, pp. 592-595; Ornithorhynchi paradoxi descriptio anatomica, Lipsiae, 1826.
Owen, Richard. On the Mammary Glands of the Ornithorhynchus paradoxus: Philosophical Transactions of the Royal Society of London, 1832, pp. 517-534; On the Ova of Ornithorhynchus paradoxus: Philosophical Transactions of the Royal Society of London, 1834, pp. 555-566, 1 plate; On the Young of the Ornithorhynchus paradoxus Blum.: Proceedings of the Zoological Society of London, 1834, pp. 43-44; id., Transactions of the Zoological Society of London, i, 1835, pp. 221-228, 2 plates; Monotremata: Todd’s Cyclopaedia of Anatomy and Physiology, iii, 1839-47, pp. 366-407, 6 figures; Remarks on the ‘Observations sur l’Ornithorhynque’ par M. Jules Verreaux: Annals and Magazine of Natural History. (2), ii, 1848, pp 317-322; On the Marsupial Pouches. Mammary Glands, and Mammary Foetus in the Echidna hystrix: Philosophical Transactions of the Royal Society of London, clv, 1865, pp. 671-686, 3 plates and 1 figure.
Parker, W. K. On Mammalian Descent: the Hunterian Lectures for 1884.
Peron, F., and L. Freycinet. Voyage de Découvertes aux Terres Australes, Atlas, Paris, 1807, plate xxxiv.
Poulton, Edward B. The Tongue of Ornithorhynchus paradoxus: the Origin of Taste Bulbs and the parts upon which they occur. Quarterly Journal of Microscopical Science, new series, xxiii, 1883, pp. 453-472, 1 plate.
Semon, R. Zoologische Forschungsreisen in Australien und dem Malayischen Archipel, ii-iii, 1894-1908; In the Australian Bush, 1899, p. 164.
Shaw, George. The Duck-Billed Platypus: The Naturalists’ Miscellany, x, 1799, plate 385; General Zoology, i, 1800, pp. 228-232.
Smith, Geoffrey. A Naturalist in Tasmania, 1909, pp. 124-126.
Spicer, W. W. On the Effects of Wounds on the Human Subject inflicted by the Spurs of the Platypus (Ornithorhynchus anatinus): Papers and Proceedings and Report of the Royal Society of Tasmania, 1876, pp. 162-167.
222
Stuart, T. P. Anderson. Poison of the Platypus: Journal of the Royal Society of New South Wales, xxviii, 1894, pp. 5-9.
Sutherland, Alexander. Temperatures of Reptiles, Monotremes and Marsupials: Proceedings of the Royal Society of Victoria, new series, ix, 1897, pp. 57-67, 1 plate.
Sydney Gazette, December 4, 1823.
Thomas, Oldfield. Catalogue of the Marsupialia and Monotremata in the Collection of the British Museum (Natural History), 1888, pp. 373-391.
Tiedemann, F., Zoologie, i, Landshut und Heidelberg, 1808, p. 589.
Traill, T. S. On the Spurs of the Ornithorhynchus: Edinburgh Philosophical Journal, vi, 1821-2, p. 184.
Verreaux, Jules. Observations sur l’Ornithorhynque: Revue Zoologique, xi, 1848, pp. 127-134.
Waite, Edgar R. The Range of the Platypus: Proceedings of the Linnean Society of New South Wales, xxi, 1896, pp. 500-502.
Waterhouse, G. R. A Natural History of the Mammalia, i, 1846, pp. 24-39, 1 plate and 4 figures.
Wiedemann, C. R. W. Nachricht von einem äusserst sonderbaren, neuentdeckten Säugethiere, Platypus anatinus: Wiedemann, Archiv für Zoologie und Zootomie, i, 1800, pp. 175-180.
Wilson, J. T., and J. P. Hill. Observations on the Development of Ornithorhynchus: Philosophical Transactions of the Royal Society of London, Series B, vol. 199, 1908, pp. 31-168, 17 plates, 15 text-figures.
[1] In this book the words muzzle, snout, bill, and beak are used synonymously, when the upper and lower mandibles combined are referred to. The pliable tissue extending beyond the jawbones is always referred to as the lips.
[2] That is, segmenting eggs from the uterus.
[3] For details, see page 208.
[4] The spurs are not for laying hold of the body, but, if at all, of the hind legs, ankles, or feet, of the female.
[5] With this statement I agree. See page 96.
[6] I have never succeeded in passing a fine horse-hair through, even after extracting the inner tube (see Plate 14).
[7] The Australian Philosophical Society.
[8] I have found both males and females in this state. The denudation is the result of moulting, and has nothing to do with the use of the spur.
[9] Platypus are frequently seen during daylight, and can easily be taken by hand beneath water, even though it may be necessary to jump into the water to do so.
[10] I find that the scent-gland enlarges and diminishes in unison with the other glands described here. This strengthens the statement that the poison gland is subject to seasonal variations.
[12] With the theory that the discharge is for the purpose of dilating the socket I entirely disagree.
[13] I have never known an entrance to be blocked up, and I have examined over 150 breeding-burrows, all of which were glaringly open to view.
[14] See description of breeding habits in chapter xi.
[15] Since this was written Mr. H. R. Carne reports the capture of a platypus in George’s River at Glenfield.
[16] In a few species of viviparous lizards, e.g., Tiliqua, a primitive placental apparatus is found.
[17] Trappers are not likely to report otherwise.
[18] During the breeding-season, to which my experience is limited, the smell is only occasionally noticeable on living specimens; but it is always found in the process of skinning.
223
228
Eagle Press Ltd., Allen St., Waterloo
Transcriber’s Notes.
Evident typographical and punctuation errors have been corrected silently. Inconsistent spelling/hyphenation has been normalised.
Half-titles and reiterations of chapter titles have been discarded.
End of page footnotes have been sequentially numbered and relocated to the end of the text.
Chapter numbers have been added to the Table of Contents.
On page 179, a reference to “Mawle” has been changed to “Maule”
On page 220, the entry for “Learmouth” has been change to “Learmonth”
The text in the list of illustrations for page 186 has been changed from “Female Platypus” to “Brooding Female” to match the caption.
Illustrations have been moved between paragraphs/chapters to improve text flow.
The List of Illustrations has been expanded where necessary to allow links to images with the same page number.
Page and plate numbers in the text may no longer be relevant, but will link to the image location.
New original cover art included with this eBook is granted to the public domain.