
Title: The Molecular Tactics of a Crystal
Author: Baron William Thomson Kelvin
Release date: June 24, 2017 [eBook #54976]
Most recently updated: October 23, 2024
Language: English
Other information and formats: www.gutenberg.org/ebooks/54976
Credits: Produced by Turgut Dincer and The Online Distributed
Proofreading Team at http://www.pgdp.net (This file was
produced from images generously made available by The
Internet Archive/American Libraries.)
THE MOLECULAR TACTICS OF
A CRYSTAL
LORD KELVIN
London
HENRY FROWDE
Oxford University Press Warehouse
Amen Corner, E.C.

New York
MACMILLAN & CO., 66 FIFTH AVENUE
BY
LORD KELVIN, P.R.S.
PROFESSOR OF NATURAL PHILOSOPHY IN THE UNIVERSITY OF GLASGOW
AND FELLOW OF PETERHOUSE, CAMBRIDGE
Being the Second Robert Boyle Lecture, delivered before
the Oxford University Junior Scientific Club
on Tuesday, May 16, 1893/
WITH TWENTY ILLUSTRATIONS
Oxford
AT THE CLARENDON PRESS
1894
Oxford
PRINTED AT THE CLARENDON PRESS
BY HORACE HART, PRINTER TO THE UNIVERSITY
§ 1. My subject this evening is not the physical properties of crystals, not even their dynamics; it is merely the geometry of the structure—the arrangement of the molecules in the constitution of a crystal. Every crystal is a homogeneous assemblage of small bodies or molecules. The converse proposition is scarcely true, unless in a very extended sense of the term crystal (§ 20 below). I can best explain a homogeneous assemblage of molecules by asking you to think of a homogeneous assemblage of people. To be homogeneous every person of the assemblage must be equal and similar to every other: they must be seated in rows or standing in rows in a perfectly similar manner. Each person, except those on the borders of the assemblage, must have a neighbour on one side and an equi-distant neighbour on the other: a neighbour on the left front and an equi-distant neighbour behind on the right, a neighbour on the right front and an equi-distant neighbour behind on the left. His two neighbours in front and his two neighbours behind are members of two rows equal and similar to the rows6 consisting of himself and his right-hand and left-hand neighbours, and their neighbours’ neighbours indefinitely to right and left. In particular cases the nearest of the front and rear neighbours may be right in front and right in rear; but we must not confine our attention to the rectangularly grouped assemblages thus constituted. Now let there be equal and similar assemblages on floors above and below that which we have been considering, and let there be any indefinitely great number of floors at equal distances from one another above and below. Think of any one person on any intermediate floor and of his nearest neighbours on the floors above and below. These three persons must be exactly in one line; this, in virtue of the homogeneousness of the assemblages on the three floors, will secure that every person on the intermediate floor is exactly in line with his nearest neighbours above and below. The same condition of alignment must be fulfilled by every three consecutive floors, and we thus have a homogeneous assemblage of people in three dimensions of space. In particular cases every person’s nearest neighbour in the floor above may be vertically over him, but we must not confine our attention to assemblages thus rectangularly grouped in vertical lines.
§ 2. Consider now any particular person C (Fig. 1) on any intermediate floor, D and D′ his nearest neighbours, E and E′ his next nearest neighbours all on his own floor. His next next nearest neighbours on that floor will be in the positions F and F′ in the diagram. Thus we see that each person C is surrounded by six persons, DD′, EE′ and FF′, being his nearest, his next nearest, and his next next nearest neighbours on his7 own floor. Excluding for simplicity the special cases of rectangular grouping, we see that the angles of the six equal and similar triangles CDE, CEF, &c., are all acute: and because the six triangles are equal and similar we see that the three pairs of mutually remote sides of the hexagon DEFD′E′F′ are equal and parallel.
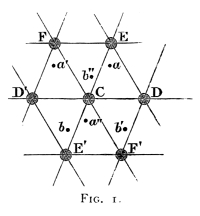
§ 3. Let now A, A′, A″, &c., denote places of persons of the homogeneous assemblage on the floor immediately above, and B, B′, B″, &c. on the floor immediately below, the floor of C. In the diagram let a, a′, a″ be points in which the floor of CDE is cut by perpendiculars to it through A, A′, A″ of the floor above, and b, b′, b″ by perpendiculars from B, B′, B″ of the floor below. Of all the perpendiculars from the floors immediately above and below, just two, one from each, cut the area of the parallelogram CDEF: and they cut it in points similarly situated in respect to the oppositely oriented triangles into which it is divided by either of its diagonals. Hence if a lies in the triangle CDE, the other five triangles of the hexagon must be cut in the corresponding points, as shown in the diagram. Thus, if we think only of the floor of C and of the floor immediately above it, we have points A, A′, A″ vertically above a, a′, a″. Imagine now a triangular pyramid, or tetrahedron, standing on the base CDE and having A for vertex: we see that each of its sides ACD, ADE, AEC, is an acute angled triangle, because, as we have already seen, CDE is an8 acute angled triangle, and because the shortest of the three distances, CA, DA, EA, is (§ 2) greater than CE (though it may be either greater than or less than DE). Hence the tetrahedron CDEA has all its angles acute; not only the angles of its triangular faces, but the six angles between the planes of its four faces. This important theorem regarding homogeneous assemblages was given by Bravais, to whom we owe the whole doctrine of homogeneous assemblages in its most perfect simplicity and complete generality. Similarly we see that we have equal and similar tetrahedrons on the bases D′CF, E′F′C; and three other tetrahedrons below the floor of C, having the oppositely oriented triangles CD′E′, &c. for their bases and B, B′, B″ for their vertices. These three tetrahedrons are equal and heterochirally1 similar to the first three. The consideration of these acute angled tetrahedrons, is of fundamental importance in respect to the engineering of an elastic solid, or crystal, according to Boscovich. So also is the consideration of the cluster of thirteen points C and the six neighbours DEFD′E′F′ in the plane of the diagram, and the three neighbours AA′A″ on the floor above, and BB′B″ on the floor below.
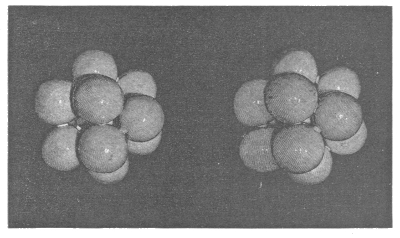
§ 4. The case in which each of the four faces of each of the tetrahedrons of § 3 is an equilateral triangle is particularly interesting. An assemblage fulfilling this condition may conveniently be called an ‘equilateral homogeneous assemblage,’ or, for brevity, an ‘equilateral assemblage.’ In an equilateral assemblage C’s twelve neighbours are all equi-distant from it. I hold in my hand a cluster of thirteen little black balls, made up by taking one of them and placing the twelve others 9 in contact with it (and therefore packed in the closest possible order), and fixing them all together by fish-glue. You see it looks, in size, colour, and shape, quite like a mulberry. The accompanying diagram shows a stereoscopic view of a similar cluster of balls painted white for the photograph.
§ 5. By adding ball after ball to such a cluster of thirteen, and always taking care to place each additional ball in some position in which it is properly in line with others, so as to make the whole assemblage homogeneous, we can exercise ourselves in a very interesting manner in the building up of any possible form of crystal of the class called ‘cubic’ by some writers and ‘octahedral’ by others. You see before you several examples. I advise any of you who wish to study crystallography to contract with a wood-turner, or a maker of beads for furniture tassels or for rosaries, for a thousand wooden balls of about half an inch10 diameter each. Holes through them will do no harm and may even be useful; but make sure that the balls are as nearly equal to one another, and each as nearly spherical, as possible.
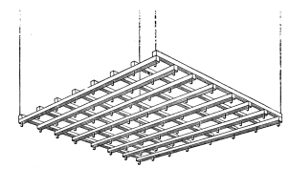
§ 6. You see here before you a large model which I have made to illustrate a homogeneous assemblage of points, on a plan first given, I believe, by Mr. William Barlow (Nature, December 20 and 27, 1883). The roof of the model is a lattice-frame (Fig. 3) consisting of two sets of eight parallel wooden bars crossing one another, and kept together by pins through the middles of the crossings. As you see, I can alter it to make parallelograms of all degrees of obliquity till the bars touch, and again you see I can make them all squares.
§ 7. The joint pivots are (for cheapness of construction) of copper wire, each bent to make a hook below the lattice frame. On these sixty-four hooks are hung sixty-four fine cords, firmly stretched by little lead weights. Each of these cords (Fig. 4) bears eight short perforated wooden cylinders, which may11 be slipped up and down to any desired position2. They are at present actually placed at distances consecutively each equal to the distance from joint to joint of the lattice frame.
§ 8. The roof of the model is hung by four cords, nearly vertical, of independently variable lengths, passing over hooks from fixed points above, and kept stretched by weights, each equal to one quarter of the weight of roof and pendants. You see now by altering the angles of the lattice work and placing it horizontal or in any inclined plane, as I am allowed to do readily by the manner in which it is hung, I have three independent variables, by varying which I can show you all varieties of homogeneous assemblages, in which three of the neighbours of every point are at equal distances from it. You see here, for example, we have the equilateral assemblage. I have adjusted the lattice roof to the proper angle, and its plane to the proper inclination to the vertical, to make a wholly equilateral assemblage of the little cylinders of wood on the vertical cords, a case, as we have seen, of special importance. If I vary also the distances between the little pieces of wood on the cords; and the distances between the joints of the lattice 12work (variations easily understood, though not conveniently producible in one model without more of mechanical construction than would be worth making), I have three other independent variables. By properly varying these six independent variables, three angles and three lengths, we may give any assigned value to each edge of one of the fundamental tetrahedrons of § 3.
§ 9. Our assemblage of people would not be homogeneous unless its members were all equal and similar and in precisely similar attitudes, and were all looking the same way. You understand what a number of people seated or standing on a floor or plain and looking the same way means. But the expression ‘looking’ is not conveniently applicable to things that have no eyes, and we want a more comprehensive mode of expression. We have it in the words ‘orientation,’ ‘oriented,’ and (verb) ‘to orient,’ suggested by an extension of the idea involved in the word ‘orientation,’ first used to signify positions relatively to east and west of ancient Greek and Egyptian temples and Christian churches. But for the orientation of a house or temple we have only one angle, and that angle is called ‘azimuth’ (the name given to an angle in a horizontal plane). For orientation in three dimensions of space we must extend our ideas and consider position with reference to east and west and up and down. A man lying on his side with his head to the north and looking east, would not be similarly oriented to a man standing upright and looking east. To provide for the complete specification of how a body is oriented in space we must have in the body a plane of reference, and a line of reference in this plane, belonging to the body and moving with it. We must also have a fixed plane and a fixed line of reference13 in it, relatively to which the orientation of the moveable body is to be specified; as, for example, a horizontal plane and the east and west horizontal line in it. The position of a body is completely specified when the angle between the plane of reference belonging to it, and the fixed plane is given; and when the angles between the line of intersection of the two planes and the lines of reference in them are also given. Thus we see that three angles are necessary and sufficient to specify the orientation of a moveable body, and we see how the specification is conveniently given in terms of three angles.
§ 10. To illustrate this take a book lying on the table before you with its side next the title-page up, and its back to the north. I now lift the east edge (the top of the book), keeping the bottom edge north and south on the table till the book is inclined, let us say, 20° to the table. Next, without altering this angle of 20°, between the side of the book and the table, I turn the book round a vertical axis, through 45° till the bottom edge lies north-east and south-west. Lastly, keeping the book in the plane to which it has been thus brought, I turn it round in this plane through 35°. These three angles of 20°, 45°, and 35°, specify, with reference to the horizontal plane of the table and the east and west line in it, the orientation of the book in the position to which you have seen me bring it, and in which I hold it before you.
§ 11. In Figs. 5 and 6 you see two assemblages, each of twelve equal and similar molecules in a plane. Fig. 5, in which the molecules are all same-ways oriented, is one homogeneous assemblage of twenty-four molecules. Fig. 6, in which in one set of rows the14 molecules are alternately oriented two different ways, may either be regarded as two homogeneous assemblages, each of twelve single molecules; or one homogeneous assemblage of twelve pairs of those single molecules.
§ 12. I must now call your attention to a purely geometrical question3 of vital interest with respect to homogeneous assemblages in general, and particularly the homogeneous assemblage of molecules constituting a crystal:—what can we take as ‘the’ boundary or ‘a’ boundary enclosing each molecule with whatever portion of space around it we are at liberty to choose for 15it, and separating it from neighbours and their portions of space given to them in homogeneous fairness?
§ 13. If we had only mathematical points to consider we should be at liberty to choose the simple obvious partitioning by three sets of parallel planes. Even this may be done in an infinite number of ways, thus:—Beginning with any point P of the assemblage, choose any other three points A, B, C, far or near, provided only that they are not in one plane with P, and that there is no other point of the assemblage in the lines PA, PB, PC, or within the volume of the parallelepiped of which these lines are conterminous edges, or within the areas of any of the faces of this parallelepiped. There will be points of the assemblage at each of the16 corners of this parallelepiped and at all the corners of the parallelepipeds equal and similar to it which we find by drawing sets of equi-distant planes parallel to its three pairs of faces. (A diagram is unnecessary.) Every point of the assemblage is thus at the intersection of three planes, which is also the point of meeting of eight neighbouring parallelepipeds. Shift now any one of the points of the assemblage to a position within the volume of any one of the eight parallelepipeds, and give equal parallel motions to all the other points of the assemblage. Thus we have every point in a parallelepipedal cell of its own, and all the points of the assemblage are similarly placed in their cells, which are themselves equal and similar.
§ 14. But now if, instead of a single point for each member of the assemblage, we have a group of points, or a globe or cube or other geometrical figure, or an individual of a homogeneous assemblage of equal, similar, similarly dressed, and similarly oriented ladies, sitting in rows, or a homogeneous assemblage of trees closely planted in regular geometrical order on a plane with equal and similar distributions of molecules, and parallel planes above and below, we may find that the best conditioned plane-faced parallelepipedal partitioning which we can choose would cut off portions properly belonging to one molecule of the assemblage and give them to the cells of neighbours. To find a cell enclosing all that belongs to each individual, for example, every part of each lady’s dress, however complexly it may be folded among portions of the equal and similar dresses of neighbours; or, every twig, leaf, and rootlet of each one of the homogeneous assemblage of trees; we must alter the boundary by17 give-and-take across the plane faces of the primitive parallelepipedal cells, so that each cell shall enclose all that belongs to one molecule, and therefore (because of the homogeneousness of the partitioning) nothing belonging to any other molecule. The geometrical problem thus presented, wonderfully complex as it may be in cases such as some of those which I have suggested, is easily performed for any possible case if we begin with any particular parallelepipedal partitioning determined for corresponding points of the assemblage as explained in § 13, for any homogeneous assemblage of single points. We may prescribe to ourselves that the corners are to remain unchanged, but if so they must to begin with either in interfaces of contact between the individual molecules, or in vacant space among the molecules. If this condition is fulfilled for one corner it is fulfilled for all, as the corners are essentially corresponding points relatively to the assemblage.
§ 15. Begin now with any one of the twelve straight lines between corners which constitute the twelve edges of the parallelepiped, and alter it arbitrarily to any curved or crooked line between the same pair of corners, subject only to the conditions (1) that it does not penetrate the substance of any member of the assemblage, and (2) that it is not cut by equal and similar parallel curves4 between other pairs of corners.
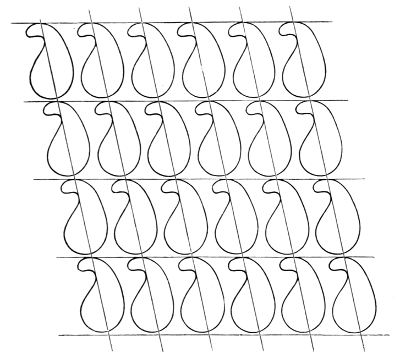
Considering now the three fours of parallel edges of the parallelepiped, let the straight lines of one set of four be altered to equal and similar parallel curves in the manner which I have described; and proceed by 18the same rule for the other two sets of four edges. We thus have three fours of parallel curved edges instead of the three fours of parallel straight edges of our primitive parallelepiped with corners (each a point of intersection of three edges) unchanged. Take now the quadrilateral of four curves substituted for the four straight edges of one face of the parallelepiped. We may call this quadrilateral a curvilineal parallelogram, because it is a circuit composed of two pairs of equal parallel curves. Draw now a curved surface (an infinitely thin sheet of perfectly extensible india-rubber if you please to think of it so) bordered by the four edges of our curvilineal parallelogram, and so shaped as not to cut any of the substance of any molecule of the assemblage. Do the same thing with an exactly similar and parallel sheet relatively to the opposite face of the parallelepiped; and again the same19 for each of the two other pairs of parallel faces. We thus have a curved-faced parallelepiped enclosing the whole of one molecule and no part of any other; and by similar procedure we find a similar boundary for every other molecule of the assemblage. Each wall of each of these cells is common to two neighbouring molecules, and there is no vacant space anywhere between them or at corners. Fig. 7 illustrates this kind of partitioning by showing a plane section parallel to one pair of plane faces of the primitive parallelepiped, for an ideal case. The plane diagram is in fact a realization of the two-dimensional problem of partitioning the pine pattern of a Persian carpet by parallelograms about as nearly rectilinear as we can make them. In the diagram faint straight lines are drawn to show the primitive parallelogrammatic partitioning. It will be seen that of all the crossings (marked with dots in the diagram) every one is similarly situated to every other in respect to the homogeneously repeated pattern figures: A, B, C, D are four of them at the corners of one cell.
§ 16. Confining our attention for a short time to the homogeneous division of a plane, remark that the division into parallelograms by two sets of crossing parallels is singular in this respect—each cell is contiguous with three neighbours at every corner. Any shifting, large or small, of the parallelograms by relative sliding in one direction or another violates this condition, brings us to a configuration like that of the faces of regularly hewn stones in ordinary bonded masonry, and gives a partitioning which fulfils the condition that at each corner each cell has only two neighbours. Each cell is now virtually a hexagon, as will be seen by the letters A, B, C, D, E, F in the diagram20 Fig. 8. A and D are to be reckoned as corners, each with an interior angle of 180°. In this diagram the continuous heavy lines and the continuous faint lines crossing them show a primitive parallelogrammatic partition by two sets of continuous parallel intersecting lines. The interrupted crossing lines (heavy) show, for the same homogeneous distribution of single points or molecules, the virtually hexagonal partitioning which we get by shifting the boundary from each portion of one of the light lines to the heavy line next it between the same continuous parallels.
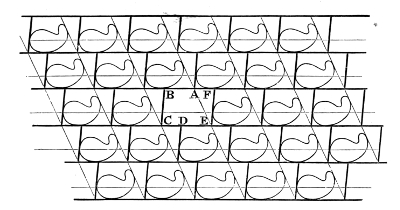
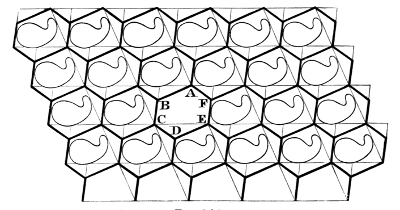
Fig. 8 bis represents a further modification of the boundary by which the 180° angles A, D, become angles of less than 180°. The continuous parallel lines (light) and the short light portions of the crossing lines show the configuration according to Fig. 8, from which this diagram is derived.
§ 17. In these diagrams (Figs. 8 and 8 bis) the object enclosed is small enough to be enclosable by a primitive parallelogrammatic partitioning of two sets of continuous crossing parallel straight lines, and by the partitioning21 of ‘bonded’ parallelograms both represented in Fig. 8, and by the derived hexagonal partitioning represented in Fig. 8 bis, with faint lines showing the primitive and the secondary parallelograms. In Fig. 7 the objects enclosed were too large to be enclosable by any rectilinear parallelogrammatic or hexagonal partitioning. The two sets of parallel faint lines in Fig. 7 show a primitive parallelogrammatic partitioning and the corresponding pairs of parallel curves intersecting at the corners of these parallelograms, of which A,B,C,D is a specimen, show a corresponding partitioning by curvilineal parallelograms. Fig. 9 shows for the same homogeneous distribution of objects a better conditioned partitioning, by hexagons in each of which one pair of parallel edges is curved. The sets of intersecting parallel straight lines in Fig. 9 show the same primitive parallelogrammatic partitioning as in Fig. 7, and the same slightly shifted to suit points chosen for well-conditionedness of hexagonal partitioning.

§ 18. For the division of continuous three-dimensional space5 into equal, similar, and similarly oriented cells, 22quite a corresponding transformation from partitioning by three sets of continuous mutually intersecting parallel planes to any possible mode of homogeneous partitioning, may be investigated by working out the three-dimensional analogue of §§ 16-17. Thus we find that the most general possible homogeneous partitioning of space with plane interfaces between the cells gives us fourteen walls to each cell, of which six are three pairs of equal and parallel parallelograms, and the other eight are four pairs of equal and parallel hexagons, each hexagon being bounded by three pairs of equal and parallel straight lines. This figure, being bounded by fourteen plane faces, is called a tetrakaidekahedron. It has thirty-six edges of intersection between faces; and twenty-four corners, in each of which three faces intersect. A particular case of it, which I call an orthic tetrakaidekahedron, being that in which the six parallelograms are23 equal squares, the eight hexagonal faces are equal equilateral and equiangular hexagons, and the lines joining corresponding points in the seven pairs of parallel faces are perpendicular to the planes of the faces, is represented by a stereoscopic picture in Fig. 10. The thirty-six edges and the twenty-four corners, which are easily counted in this diagram, occur in the same relative order in the most general possible partitioning, whether by plane-faced tetrakaidekahedrons or by the generalized tetrakaidekahedron described in § 19.
§ 19. The most general homogeneous division of space is not limited to plane-faced cells; but it still consists essentially of tetrakaidekahedronal cells, each bounded by three pairs of equal and parallel quadrilateral faces, and four pairs of equal and parallel hexagonal faces, neither the quadrilaterals nor the hexagons being necessarily plane. Each of the thirty-six edges may be straight or crooked or curved; the pairs of opposite edges, whether of the quadrilaterals or hexagons, need not be equal and parallel; neither the four corners of each quadrilateral nor the six corners of each hexagon need be in one plane. But every pair of corresponding edges of every pair of parallel corresponding faces, whether quadrilateral or hexagonal, must be equal and parallel. I have described an interesting case of partitioning by tetrakaidekahedrons of curved faces with curved edges in a paper6 published about seven years ago. In this case each of the quadrilateral faces is plane. Each hexagonal face is a slightly curved surface having three rectilineal diagonals through its centre in one plane.
The six sectors of the face between these diagonals lie alternately on opposite sides of their plane, and are bordered by six arcs of plane curves lying on three pairs of parallel planes. This tetrakaidekahedronal partitioning fulfils the condition that the angles between three planes meeting in an edge are everywhere each 120°; a condition that cannot be fulfilled in any plane-faced tetrakaidekahedron. Each hexagonal wall is an anticlastic surface of equal opposite curvatures at every point, being the surfaces of minimum area bordered by six curved edges. It is shown easily and beautifully, and with a fair approach to accuracy, by choosing six little circular arcs of wire, and soldering them together by their ends in proper planes for the six edges of the hexagon; and dipping it in soap solution and taking it out.
§ 20. Returning now to the tactics of a homogeneous assemblage, remark that the qualities of the assemblage as a whole depend both upon the character and orientation of each molecule, and on the character of the homogeneous assemblage formed by corresponding points of the molecules. After learning the simple mathematics of crystallography, with its indicial system7 for defining the faces and edges of a crystal according to the Bravais rows and nets and tetrahedrons of molecules in which we think only of a homogeneous assemblage of points, we are apt to forget that the true crystalline molecule, whatever its nature may be, has sides, and that generally two opposite sides of each molecule may be expected to be very different in quality, and we are almost surprised when mineralogists 26tell us that two parallel faces on two sides of a crystal have very different qualities in many natural crystals. We might almost as well be surprised to find that an army in battle array, which is a kind of large-grained crystal, presents very different appearance to any one looking at it from outside, according as every man in the ranks with his rifle and bayonet faces to the front or to the rear or to one flank or to the other.
§ 21. Consider, for example, the ideal case of a crystal consisting of hard equal and similar tetrahedronal solids all sameways oriented. A thin plate of crystal cut parallel to any one set of the faces of the constituent tetrahedrons would have very different properties on its two sides; as the constituent molecules would all present points outwards on one side and flat surfaces on the other. We might expect that the two sides of such a plate of crystal would become oppositely electrified when rubbed by one and the same rubber; and, remembering that a piece of glass with part of its surface finely ground but not polished and other parts polished becomes, when rubbed with white silk, positively electrified over the polished parts and negatively electrified over the non-polished parts, we might almost expect that the side of our supposed crystalline plate towards which flat faces of the constituent molecules are turned would become positively electrified, and the opposite side, showing free molecular corners, would become negatively electrified, when both are rubbed by a rubber of intermediate electric quality. We might also from elementary knowledge of the fact of piezo-electricity, that is to say, the development of opposite electricities on the two sides of a crystal by pressure, expect that our supposed crystalline plate, if27 pressed perpendicularly on its two sides, would become positively electrified on one of them and negatively on the other.
§ 22. Intimately connected with the subject of enclosing cells for molecules of given shape, assembled homogeneously, is the homogeneous packing together of equal and similar molecules of any given shape. In every possible case of any infinitely great number of similar bodies the solution is a homogeneous assemblage. But it may be a homogeneous assemblage of single solids all oriented the same way, or it may be a homogeneous assemblage of clusters of two or more of them placed together in different orientations. For example, let the given bodies be halves (oblique or not oblique) of any parallelepiped on the two sides of a dividing plane through a pair of parallel edges. The two halves are homochirally8 similar; and, being equal, we may make a homogeneous assemblage of them by orienting them all the same way and placing them properly in rows. But the closest packing of this assemblage would necessarily leave vacant spaces between the bodies: and we get in reality the closest possible packing of the given bodies by taking them in pairs oppositely oriented and placed together to form parallelepipeds. These clusters may be packed together so as to leave no unoccupied space.
Whatever the number of pieces in a cluster in the closest possible packing of solids may be for any particular shape, we may consider each cluster as itself a given single body, and thus reduce the problem to the packing closely together of assemblages of individuals all sameways oriented; and to this problem therefore it is convenient that we should now confine our attention.
§ 23. To avoid complexities such as those which we find in the familiar problem of homogeneous packing of forks or spoons or tea-cups or bowls, of any ordinary shape, we shall suppose the given body to be of such shape that no two of them similarly oriented can touch one another in more than one point. Wholly convex bodies essentially fulfil this condition; but it may also be fulfilled by bodies not wholly convex, as is illustrated in Fig. 11.
§ 24. To find close and closest packing of any number of our solids S1, S2, S3 ... of shape fulfilling the condition of § 23 proceed thus:—
(1) Bring S2 to touch S1 at any chosen point p of its surface (Fig. 12).
(2) Bring S3 to touch S1 and S2, at r and q respectively.
(3) Bring S4 (not shown in the diagram) to touch S1, S2, and S3.
(4) Place, any number of the bodies together in three rows continuing the lines of S1S2, S1S3, S1S4, and in three sets of equi-distant rows parallel to these. This makes a homogeneous assemblage. In the assemblage29 so formed the molecules are necessarily found to be in three sets of rows parallel respectively to the three pairs S2S3, S3S4, S4S2. The whole space occupied by an assemblage of n of our solids thus arranged has clearly 6n times the volume of a tetrahedron of corresponding points of S1, S2, S3, S4. Hence the closest of the close packings obtained by the operations (1) ... (4) is found if we perform the operations (1), (2), and (3) as to make the volume of this tetrahedron least possible.
§ 25. It is to be remarked that operations (1) and (2) leave for (3) no liberty of choice for the place of S4, except between two determinate positions on opposite sides of the group S1, S2, S3. The volume of the tetrahedron will generally be different for these two positions of S4, and, even if the volume chance to be equal in any case, we have differently shaped assemblages according as we choose one or other of the two places for S4.
This will be understood by looking at Fig. 12, showing S1 and neighbours on each side of it in the rows of S1S2, S1S3, and in a row parallel to that of S2S3. The plane of the diagram is parallel to the planes of corresponding points of these seven bodies, and the30 diagram is a projection of these bodies by lines parallel to the intersections of the tangent planes through p and r. If the three tangent planes through p, q, and r, intersected in parallel lines, q would be seen like p and r as a point of contact between the outlines of two of the bodies; but this is only a particular case, and in general q must, as indicated in the diagram, be concealed by one or other of the two bodies of which it is the point of contact. Now imagining, to fix our ideas and facilitate brevity of expression, that the planes of corresponding points of the seven bodies are horizontal, we see clearly that S4 may be brought into proper position to touch S1, S2, and S3 either from above or from below; and that there is one determinate place for it if we bring it into position from above, and another determinate place for it if we bring it from below.
§ 26. If we look from above at the solids of which Fig. 12 shows the outline, we see essentially a hollow leading down to a perforation between S1, S2, S3, and if we look from below we see a hollow leading upwards to the same perforation: this for brevity we shall call the perforation pqr. The diagram shows around S1 six hollows leading down to perforations, of which two are similar to pqr, and the other three, of which p′q′r′ indicates one, are similar one to another but are dissimilar to pqr. If we bring S4 from above into position to touch S1, S2, and S3, its place thus found is in the hollow pqr, and the places of all the solids in the layer above that of the diagram are necessarily in the hollows similar to pqr. In this case the solids in the layer below that of the diagram must lie in the hollows below the perforations dissimilar to pqr, in order to31 make a single homogeneous assemblage. In the other case, S4 brought up from below finds its place on the under side of the hollow pqr, and all solids of the lower layer find similar places: while solids in the layer above that of the diagram find their places in the hollows similar to p′q′r′. In the first case there are no bodies of the upper layer in the hollows above the perforations similar to p′q′r′, and no bodies of the lower layer in the hollows below the perforations similar to pqr. In the second case there are no bodies of the upper layer in the hollows above the perforations similar to pqr, and none of the under layer in the hollows below the perforations similar to p′q′r′.
§ 27. Going back now to operation (1) of § 23, remark that when the point of contact p is arbitrarily chosen on one of the two bodies S1, the point of contact on the other will be the point on it corresponding to the point or one of the points of S1, where its tangent plane is parallel to the tangent plane at p. If S1 is wholly convex it has only two points at which the tangent planes are parallel to a given plane, and therefore the operation (1) is determinate and unambiguous. But if there is any concavity there will be four or some greater even number of tangent planes parallel to any one of some planes, while there will be other planes to each of which only one pair of tangent planes is parallel. Hence, operation (1), though still determinate, will have a multiplicity of solutions, or only a single solution, according to the choice made of the position of p.
Henceforth however, to avoid needless complications of ideas, we shall suppose our solids to be wholly convex; and of some such unsymmetrical shape as those32 indicated in Fig. 12 of § 25, and shown by stereoscopic photograph in Fig. 13 of § 36. With or without this convenient limitation, operation (1) has two freedoms, as p may be chosen freely on the surface of S1; and operation (2) has clearly just one freedom after operation (1) has been performed. Thus, for a solid of any given shape, we have three disposables, or, as commonly called in mathematics, three ‘independent variables,’ all free for making a homogeneous assemblage according to the rule of § 22.
§ 28. In the homogeneous assemblage defined in § 24, each solid, S1, is touched at twelve points, being the three points of contact with S2, S3, S4, and the three 3’s of points on S1 corresponding to the points on S2, S3, S4, at which these bodies are touched by the others of the quartet. This statement is somewhat difficult to follow, and we see more clearly the twelve points of contact by not confining our attention to the quartet S1, S2, S3, S4 (convenient as this is for some purposes), but completing the assemblage and considering six neighbours around S1 in one plane layer of the solids as shown in Fig. 12, with their six points prq″p′r′q″′ of contact with S1; and the three neighbours of the two adjacent parallel layers which touch it above and below. This cluster of thirteen, S1 and twelve neighbours, is shown for the case of spherical bodies in the stereoscopic photograph of § 4 above. We might of course, if we pleased, have begun with the plane layer of which S1, S2, S4 are members, or with that of which S1, S3, S4 are members, or with the plane layer parallel to the fourth side S2 S3 S4 of the tetrahedron: and thus we have four different ways of grouping the twelve points of33 contact on S1 into one set of six and two sets of three.
§ 29. In this assemblage we have what I call ‘close order’ or ‘close packing.’ For closest of close packings the volume of the tetrahedron (§ 24) of corresponding points of S1, S2, S3, and S4 must be a minimum, and the least of minimums if, as generally will be the case, there are two more different configurations for each of which the volume is a minimum. There will in general also be configurations of minimax volume and of maximum volume, subject to the condition that each body is touched by twelve similarly oriented neighbours.
§ 30. Pause for a moment to consider the interesting kinematical and dynamical problems presented by a close homogeneous assemblage of smooth solid bodies of given convex shape, whether perfectly frictionless or exerting resistance against mutual sliding according to the ordinarily stated law of friction between dry hard solid bodies. First imagine that they are all similarly oriented and each in contact with twelve neighbours, except outlying individuals (which there must be at the boundary if the assemblage is finite, and each of which is touched by some number of neighbours less than twelve). The coherent assemblage thus defined constitutes a kinematic frame or skeleton for an elastic solid of very peculiar properties. Instead of the six freedoms, or disposables, of strain presented by a natural solid it has only three. Change of shape of the whole can only take place in virtue of rotation of the constituent parts relatively to any one chosen row of them, and the plane through it and another chosen row.
§ 31. Suppose first the solids to be not only perfectly smooth but perfectly frictionless. Let the assemblage be subjected to equal positive or negative pressure inwards all around its boundary. Every position of minimum, minimax, or maximum volume will be a position of equilibrium. If the pressure is positive the equilibrium will be stable if, and unstable unless, the volume is a minimum. If the pressure is negative the equilibrium will be stable if, and unstable unless, the volume is a maximum. Configurations of minimax volume will be essentially unstable.
§ 32. Consider now the assemblage of § 31 in a position of stable equilibrium under the influence of a given constant uniform pressure inwards all round its boundary. It will have rigidity in simple proportion to the amount of this pressure. If now by the superposition of non-uniform pressure at the boundary, for example equal and opposite pressures on two sides of the assemblage, a finite change of shape is produced: the whole assemblage essentially swells in bulk. This is the ‘dilatancy’ which Osborne Reynolds has described9 in an exceedingly interesting manner with reference to a sack of wheat or sand, or an india-rubber bag tightly filled with sand or even small shot. Consider, for example, a sack of wheat filled quite full and standing up open. It is limp and flexible. Now shake it down well, fill it quite full, shake again, so as to get as much into it as possible, and tie the mouth very tightly close. The sack becomes almost as stiff as a log of wood of the same shape. Open the mouth partially, and it becomes again limp, especially 35in the upper parts of the bag. In Reynolds’ observations on india-rubber bags of small shot his ‘dilatancy’ depends, essentially and wholly, on breaches of some of the contacts which exist between the molecules in their configuration of minimum volume: and it is possible that in all his cases the dilatations which he observed are chiefly, if not wholly, due to such breaches of contact.
But it is possible, it almost seems probable, that in bags or boxes of sand or powder, of some kinds of smooth rounded bodies of any shape, not spherical or ellipsoidal, subjected persistently to unequal pressures in different directions, and well shaken, stable positions of equilibrium are found with almost all the particles each touched by twelve others.
Here is a curious subject of Natural History through all ages till 1885, when Reynolds brought it into the province of Natural Philosophy by the following highly interesting statement:—‘A well-marked phenomenon receives its explanation at once from the existence of dilatancy in sand. When the falling tide leaves the sand firm, as the foot falls on it the sand whitens and appears momentarily to dry round the foot. When this happens the sand is full of water, the surface of which is kept up to that of the sand by capillary attractions; the pressure of the foot causing dilatation of the sand more water is required, which has to be obtained either by depressing the level of the surface against the capillary attractions, or by drawing water through the interstices of the surrounding sand. This latter requires time to accomplish, so that for the moment the capillary forces are overcome; the surface of the water is lowered below36 that of the sand, leaving the latter white or drier until a sufficient supply has been obtained from below, when the surface rises and wets the sand again. On raising the foot it is generally seen that the sand under the foot and around becomes momentarily wet; this is because, on the distorting forces being removed, the sand again contracts, and the excess of water finds momentary relief at the surface.’
This proves that the sand under the foot, as well as the surface around it, must be dry for a short time after the foot is pressed upon it, though we cannot see it whitened, as the foot is not transparent. That it is so has been verified by Mr. Alex. Galt, Experimental Instructor in the Physical Laboratory of Glasgow University, by laying a small square of plate-glass on wet sand on the sea-shore of Helensburgh, and suddenly pressing on it by a stout stick with nearly all his weight. He found the sand, both under the glass and around it in contact with the air, all became white at the same moment. Of all the two hundred thousand million men, women, and children who, from the beginning of the world, have ever walked on wet sand, how many, prior to the British Association Meeting at Aberdeen in 1885, if asked, ‘Is the sand compressed under your foot?’ would have answered otherwise than ‘Yes!’?
(Contrast with this the case of walking over a bed of wet sea-weed!)
§ 33. In the case of globes packed together in closest order (and therefore also in the case of ellipsoids, if all similarly oriented), our condition of coherent contact between each molecule and twelve neighbours implies absolute rigidity of form and constancy of bulk.37 Hence our convex solid must be neither ellipsoidal nor spherical in order that there may be the changes of form and changes of bulk which we have been considering as dependent on three independent variables specifying the orientation of each solid relatively to rows of the assemblage. An interesting dynamical problem is presented by supposing any mutual forces, such as might be produced by springs, to act between the solid molecules, and investigating configurations of equilibrium on the supposition of frictionless contacts. The solution of it of course is that the potential energy of the springs must be a minimum or a minimax or a maximum for equilibrium, and a minimum for stable equilibrium. The solution will be a configuration of minimum or minimax, or maximum, volume, only in the case of pressure equal in all directions.
§ 34. A purely geometrical question, of no importance in respect to the molecular tactics of a crystal but of considerable interest in pure mathematics, is forced on our attention by our having seen (§ 27) that a homogeneous assemblage of solids of given shape, each touched by twelve neighbours, has three freedoms which may be conveniently taken as the three angles specifying the orientation of each molecule relatively to rows of the assemblage as explained in § 30.
Consider a solid S1 and the twelve neighbours which touch it, and try if it is possible to cause it to touch more than twelve of the bodies. Attach ends of three thick flexible wires to any places on the surface of S1; carry the wires through interstices of the assemblage, and attach their other ends at any three places of A, B, C, respectively, these being any three of the bodies outside38 the cluster of S1 and its twelve neighbours. Cut the wires across at any chosen positions in them; and round off the cut ends, just leaving contact between the rounded ends, which we shall call f′f, g′g, h′h. Do homogeneously for every other solid of the assemblage what we have done for S1. Now bend the wires slightly so as to separate the pairs of points of contact, taking care to keep them from touching any other bodies which they pass near on their courses between S1 and A, B, C respectively. After having done this, thoroughly rigidify all the wires thus altered. We may now, having three independent variables at our disposal, so change the orientation of the molecules, relatively to rows of the assemblage, as to bring f′f, g′g, and h′h again into contact. We have thus six fresh points of S1; of which three are f′, g′, h′; and the other three are on the three extensions of S1 corresponding to the single extensions of A, B, C respectively, which we have been making. Thus we have a real solution of the interesting geometrical problem:—It is required so to form a homogeneous assemblage of solids of any arbitrarily given shape that each solid shall be touched by eighteen others. This problem is determinate, because the making of the three contacts f′f, g′g, h′h, uses up the three independent variables left at our disposal after we have first formed a homogeneous assemblage with twelve points of contact on each solid. But our manner of finding a shape for each solid which can allow the solution of the problem to be real, proves that the solution is essentially imaginary for every wholly convex shape.
§ 35. Pausing for a moment longer to consider afresh the geometrical problem of putting arbitrarily given39 equal and similar solids together to make a homogeneous assemblage of which each member is touched by eighteen others, we see immediately that it is determinate (whether it has any real solution or not), because when the shape of each body is given we have nine disposables for fixing the assemblage: six for the character of the assemblage of the corresponding points, and three for the orientation of each molecule relatively to rows of the assemblage of corresponding points. These nine disposables are determined by the condition that each body has nine pairs of contacts with others.
Suppose now a homogeneous assemblage of the given bodies, in open order with no contacts, to be arbitrarily made according to any nine arbitrarily chosen values for the six distances between a point of S1 and the corresponding points of its six pairs of nearest and next nearest neighbours (§ 1 above), and the three angles (§ 9 above) specifying the orientation of each body relatively to rows of the assemblage. We may choose in any nine rows through S1 any nine pairs of bodies at equal distances on the two sides of S1 far or near, for the eighteen bodies which are to be in contact with S1. Hence there is an infinite number of solutions of the problem of which only a finite number can be real. Every solution of the problem of eighteen contacts is imaginary when the shape is wholly convex.
§ 36. Without for a moment imagining the molecules of matter to be hard solids of convex shape, we may derive valuable lessons in the tactics of real crystals by studying the assemblage described in §§ 24 and 25 and represented in Figs. 12 and 13. I must for the present40 forego the very attractive subject of the tactics presented by faces not parallel to one or other of the four faces of the primitive tetrahedrons which we found in § 24, and ask you only to think of the two sides of a plate of crystal parallel to any one of them, that is to say, an assemblage of such layers as those represented geometrically in Fig. 12 and shown in stereoscopic view in Fig. 13. If, as is the case with the solids10 photographed in Fig. 13, the under side of each solid is nearly plane but slightly convex, and the top is somewhat sharply curved, we have the kind of difference between the upper and under of the two parallel sides of the crystal which I have already described to you in § 21 above. In this case the assemblage is formed by letting the solids fall down from above and settle in the hollows to which they come most readily, or which give them the stablest position. It would, we may suppose, be the hollows 41p′ q′ r′, not p q r, (Fig. 12) that would be chosen; and thus, of the two formations described in § 25, we should have that in which the hollows above p′ q′ r′ are occupied by the comparatively flat under sides of the molecules of the layer above, and the hollows below the apertures p q r by the comparatively sharp tops of the molecules of the layers below.
§ 37. For many cases of natural crystals of the wholly asymmetric character, the true forces between the crystalline molecules will determine precisely the same tactics of crystallization as would be determined by the influence of gravity and fluid viscosity in the settlement from water, of sand composed of uniform molecules of the wholly unsymmetrical convex shape represented in Figs. 12 and 13. Thus we can readily believe that a real crystal which is growing by additions to the face seen in Fig. 12, would give layer after layer regularly as I have just described. But if by some change of circumstances the plate, already grown to a thickness of many layers in this way, should come to have the side facing from us in the diagram exposed to the mother-liquor, or mother-gas, and begin to grow from that face, the tactics might probably be that each molecule would find its resting-place with its most nearly plane side in the wider hollows under p′ q′ r′, instead of with its sharpest corner in the narrower and steeper hollows under p q r, as are the molecules in the layer below that shown in the diagram in the first formation. The result would be a compound crystal consisting of two parts, of different crystalline quality, cohering perfectly together on the two sides of an interfacial plane. It seems probable that this double structure may be found in nature, presented by crystals42 of the wholly unsymmetric class, though it may not hitherto have been observed or described in crystallographic treatises.
§ 38. This asymmetric double crystal becomes simply the well-known symmetrical ‘twin-crystal’11 in the particular case in which each of the constituent molecules is symmetrical on the two sides of a plane through it parallel to the plane of our diagrams, and also on the two sides of some plane perpendicular to this plane. We see, in fact, that in this case if we cut in two the double crystal by the plane of Fig. 14, and turn one part ideally through 180° round the intersection of these two planes, we bring it into perfect coincidence with the other part.
This we readily understand by looking at Fig. 14, in which the solid shown in outline may be either an egg-shaped figure of revolution, or may be such a figure flattened by compression perpendicular to the plane of the diagram. The most readily chosen and the most stable resting-places for the constituents of each successive layer might be the wider hollows p′ q′ r′: and therefore if, from a single layer to begin with, the assemblage were to grow by layer after layer added to it on each side, it might probably grow as a twin-crystal. But it might also be that the presence of a molecule in the wider hollow p′ q′ r′ on one side, might render the occupation of the corresponding hollow on the other side by another molecule less probable, or even impossible. Hence, according to the configuration and the molecular forces of the particular crystalline molecule in natural crystallization, there may be necessarily, or almost necessarily, the twin, when growth proceeds simultaneously on the two sides: or the twin growth may be impossible, because the first occupation of the wider hollows on one side, may compel the continuity of the crystalline quality throughout, by leaving only the narrower hollows p q r free for occupation by molecules attaching themselves on the other side.
§ 39. Or the character of the crystalline molecule may be such that when the assemblage grows by the addition of layer after layer on one side only, with a not very strongly decided preference to the wider hollows p′ q′ r′, some change of circumstances may cause the molecules of one layer to place themselves in a hollow p q r. The molecules in the next layer after this would find the hollows p′ q′ r′44 occupied on the far side, and would thus have a bias in favour of the hollows p q r. Thus layer after layer might be added, constituting a twinned portion of the growth, growing, however, with less strong security for continued homogeneousness than when the crystal was growing, as at first, by occupation of the wider hollows p′ q′ r′. A slight disturbance might again occur, causing the molecules of a fresh layer to settle, not in the narrow hollows p q r, but in the wider hollows p′ q′ r′, notwithstanding the nearness of molecules already occupying the wider hollows on the other side. Disturbances such as these occurring irregularly during the growth of a crystal, might produce a large number of successive twinnings at parallel planes with irregular intervals between them, or a large number of twinnings in planes at equal intervals might be produced by some regular periodic disturbance occurring for a certain number of periods, and then ceasing. Whether regular and periodic, or irregular, the tendency would be that the number of twinnings should be even, and that after the disturbances cease the crystal should go on growing in the first manner, because of the permanent bias in favour of the wider hollows p′ q′ r′. These changes of molecular tactics, which we have been necessarily led to by the consideration of the fortuitous concourse of molecules, are no doubt exemplified in a large variety of twinnings and counter-twinnings found in natural minerals. In the artificial crystallization of chlorate of potash they are of frequent occurrence, as is proved, not only by the twinnings and counter-twinnings readily seen in the crystalline forms, but also by the brilliant iridescence observed in many of45 the crystals found among a large multitude, which was investigated scientifically by Sir George Stokes ten years ago, and described in a communication to the Royal Society ‘On a remarkable phenomenon of crystalline reflection’ (Proc. R.S., vol. xxxviii, 1885, p. 174).
§ 40. A very interesting phenomenon, presented by what was originally a clear homogeneous crystal of chlorate of potash, and was altered by heating to about 245°-248° Cent., which I am able to show you through the kindness of Lord Rayleigh, and of its discoverer, Mr. Madan, presents another very wonderful case of changing molecular tactics, most instructive in respect of the molecular constitution of elastic solids. When I hold this plate before you with the perpendicular to its plane inclined at 10° or more to your line of vision, you see a tinsel-like appearance, almost as bright as if it were a plate of polished silver, on this little area, which is a thin plate of chlorate of potash cemented for preservation between two pieces of glass; and, when I hold a light behind, you see that the little plate is almost perfectly opaque like metal foil. But now when I hold it nearly perpendicular to your line of vision the tinsel-like appearance is lost. You can see clearly through the plate, and you also see that very little light is reflected from it. As a result both of Mr. Madan’s own investigations, and further observations by himself, Lord Rayleigh came to the conclusion that the almost total reflection of white light which you see is due to the reflection of light at many interfacial planes between successive layers of twinned and counter-twinned crystal of small irregular thicknesses, and not to any splits or cavities46 or any other deviation from homogeneousness than that presented by homogeneous portions of oppositely twinned-crystals in thorough molecular contact at the interfaces.
§ 41. When the primitive clear crystal was first heated very gradually by Madan to near its melting-point (359° according to Carnelly), it remained clear, and only acquired the tinsel appearance after it had cooled to about 245° or 248°12. Rayleigh found that if a crystal thus altered was again and again heated it always lost the tinsel appearance, and became perfectly clear at some temperature considerably below the melting-point, and regained it at about the same temperature in cooling. It seems, therefore, certain that at temperatures above 248°, and below the melting-point, the molecules had so much of thermal motions as to keep them hovering 47about the positions of p q r, p′ q′ r′, of our diagrams, but not enough to do away with the rigidity of the solid; and that when cooled below 248° the molecules were allowed to settle in one or other of the two configurations, but with little of bias for one in preference to the other. It is certainly a very remarkable fact in Natural History, discovered by these observations, that, when the molecules come together to form a crystal out of the watery solution, there should be so much more decided a bias in favour of continued homogeneousness of the assemblage than when, by cooling, they are allowed to settle from their agitations in a rigid, but nearly melting, solid.
§ 42. But even in crystallization from watery solution of chlorate of potash the bias in favour of thorough homogeneousness is not in every contingency decisive. In the first place, beginning, as the formation seems to begin, from a single molecular plane layer such as that ideally shown in Fig. 14, it goes on, not to make a homogeneous crystal on the two sides of this layer, but probably always so as to form a twin-crystal on its two sides, exactly as described in § 38, and, if so, certainly for the reason there stated. This is what Madan calls the ‘inveterate tendency to produce twins (such as would assuredly drive a Malthus to despair)13’; and it is to this that he alludes as ‘the inevitable twin-plate’ in the passage from his paper given in the foot-note to § 41 above.
§ 43. In the second place, I must tell you that many of the crystals produced from the watery solution by the ordinary process of slow evaporation and crystallization, show twinnings and counter-twinnings at 48irregular intervals in the otherwise homogeneous crystal on either one or both sides of the main central twin-plane, which henceforth, for brevity, I shall call (adopting the hypothesis already explained, which seems to me undoubtedly true) the ‘initial plane.’ Each twinning is followed, I believe, by a counter-twinning at a very short distance from it; at all events Lord Rayleigh’s observations14 prove that the whole number of twinnings and counter-twinnings in a thin disturbed stratum of the crystal on one side of the main central twin-plane is generally, perhaps always, even; so that, except through some comparatively very small part or parts of the whole thickness, the crystal on either side of the middle or initial plane is homogeneous. This is exactly the generally regular growth which I have described to you (§ 39) as interrupted occasionally or accidentally by some unexplained disturbing cause, but with an essential bias to the homogeneous continuance of the more easy or natural one of the two configurations.
§ 44. I have now great pleasure in showing you a most interesting collection of the iridescent crystals of chlorate of potash, each carefully mounted for preservation between two glass plates, which have been kindly lent to us for this evening by Mr. Madan. In March, 1854, Dr. W. Bird Herapath sent to Prof. Stokes some crystals of chlorate of potash showing the brilliant and beautiful colours you now see, and, thirty years later, Prof. E. J. Mills recalled his attention to the subject by sending him ‘a fine collection of splendidly coloured crystals of chlorate of potash of considerable size, several of the plates having an area 49of a square inch or more, and all of them being thick enough to handle without difficulty.’ The consequence was that Stokes made a searching examination into the character of the phenomenon, and gave the short, but splendidly interesting, communication to the Royal Society of which I have already told you. The existence of these beautifully coloured crystals had been well known to chemical manufacturers for a long time, but it does not appear that any mention of them was to be found in any scientific journal or treatise prior to Stokes’ paper of 1885. He found that the colour was due to twinnings and counter-twinnings in a very thin disturbed stratum of the crystal showing itself by a very fine line, dark or glistening, according to the direction of the incident light when a transverse section of the plate of crystal was examined in a microscope. By comparison with a spore of lycopodium he estimated that the breadth of this line, and therefore the thickness of the disturbed stratum of the crystal, ranged somewhere about the one-thousandth of an inch. He found that the stratum was visibly thicker in those crystals which showed red colour than in those which showed blue. He concluded that ‘the seat of the coloration is certainly a thin twinned stratum’ (that is to say, a homogeneous portion of crystal between a twinning and a counter-twinning), and found that ‘a single twin-plane does not show anything of the kind.’
§ 45. A year or two later Lord Rayleigh entered on the subject with an exhaustive mathematical investigation of the reflection of light at a twin-plane of a crystal (Philosophical Magazine, September, 1888), by the application of which, in a second paper ‘On the remarkable50 phenomenon of Crystalline Reflection described by Prof. Stokes,’ published in the same number of the Philosophical Magazine, he gave what seems certainly the true explanation of the results of Sir George Stokes’ experimental analysis of these beautiful phenomena. He came very decidedly to the conclusion that the selective quality of the iridescent portion of the crystal, in virtue of which it reflects almost totally light nearly of one particular wave-length for one particular direction of incidence (on which the brilliance of the coloration depends), cannot be due to merely a single twin-stratum, but that it essentially is due to a considerable number of parallel twin-strata at nearly equal distances. The light reflected by this complex stratum is, for any particular direction of incident and reflected ray, chiefly that of which the wave-length is equal to twice the length of the period of the twinning and counter-twinning, on a line drawn through the stratum in the direction of either the incident or the reflected ray.
§ 46. It seems to me probable that each twinning is essentially followed closely by a counter-twinning. Probably three or four of these twin-strata might suffice to give colour; but in any of the brilliant specimens as many as twenty or thirty, or more, might probably be necessary to give so nearly monochromatic light as was proved by Stokes’ prismatic analysis of the colours observed in many of his specimens. The disturbed stratum of about a one-thousandth of an inch thickness, seen by him in the microscope, amply suffices for the 5, 10, or 100 half wave-lengths required by Rayleigh’s theory to account for perceptible or brilliant coloration. But what can be the cause of any approach to regular51 periodicity in the structure sufficiently good to give the colours actually observed? Periodical motion of the mother-liquor relatively to the growing crystal might possibly account for it. But Lord Rayleigh tells us that he tried rocking the pan containing the solution without result. Influence of light has been suggested, and I believe tried, also without result, by several enquirers. We know, by the beautiful discovery of Edmond Becquerel, of the prismatic colours photographed on a prepared silver plate by the solar spectrum, that ‘standing waves’ (that is to say, vibrations with stationary nodes and stationary places of maximum vibration), due to co-existence of incident and reflected waves, do produce such a periodic structure as that which Rayleigh’s theory shows capable of giving a corresponding tint when illuminated by white light. It is difficult, therefore, not to think that light may be effective in producing the periodic structure in the crystallization of chlorate of potash, to which the iridescence is due. Still, experimental evidence seems against this tempting theory, and we must perforce be content with the question unanswered:—What can be the cause of 5, or 10, or 100 pairs of twinning and counter-twinning following one another in the crystallization with sufficient regularity to give the colour: and why, if there are twinnings and counter-twinnings, are they not at irregular intervals, as those produced by Madan’s process, and giving the observed white tinsel-like appearance with no coloration?
§ 47. And now I have sadly taxed your patience: and I fear I have exhausted it and not exhausted my subject! I feel I have not got halfway through what I hoped I might be able to put before you this evening52 regarding the molecular structure of crystals. I particularly desired to speak to you of quartz crystal with its ternary symmetry and its chirality15; and to have told you of the etching16 by hydrofluoric acid which, as it were, commences to unbuild the crystal by taking away molecule after molecule, but not in the reverse order of the primary up-building; and which thus reveals differences of tactics in the alternate faces of the six-sided pyramid which terminates at either end, sometimes at both ends, the six-sided prism constituting generally the main bulk of the crystal. I must confine myself to giving you a geometrical symbol for the ternary symmetry of the prism and its terminal pyramid.
§ 48. Make an equilateral equiangular hexagonal
prism, with its diagonal from edge to edge ninety-five
hundredths17 of its length. Place a number of these
close together, so as to make up a hexagonal plane layer
with its sides perpendicular to the sides of the constituent
hexagonal prisms: see Fig. 15 and imagine the
semicircles replaced by their diameters. You see in
each side of the hexagonal assemblage, edges of the
constituent prisms, and you see at each corner of
the assemblage a face (not an edge) of one of the
constituent prisms. Build up a hexagonal prismatic
assemblage by placing layer after layer over it with
the constituent prisms of each layer vertically over
those in the layer below; and finish the assemblage
53with a six-sided pyramid by building upon the upper
end of the prism, layer after layer of diminishing
hexagonal groups, each less by one circumferential
row than the layer below it. You thus have a crystal
of precisely the shape of a symmetrical specimen of
rock crystal, with the faces of its terminal pyramid
inclined at 38° 13′ to the faces of the prism from which
they spring. But the assemblage thus constituted has
‘senary’ (or six-rayed symmetry). To reduce this to
ternary symmetry, cut a groove through the middle
of each alternate face of the prismatic molecule, making
this groove in the first place parallel to the edges: and
add a corresponding projection, or fillet, to the middles
of the other three faces, so that two of the cylinders
similarly oriented would fit together, with the projecting
fillet on one side of one of them entering the
groove in the anti-corresponding side of the other.
The prismatic portion of the assemblage thus formed
shows (see Fig. 15), on its alternate edges, faces of54
molecules with projections and faces of molecules
with grooves; and shows only orientational differences
between alternate faces, whether of the pyramid or
of the prism. Having gone only so far from ‘senary’
symmetry, we have exactly the triple, or three-pair,
anti-symmetry required for the piezo-electricity of
quartz investigated so admirably by the brothers
Curie18, who found that a thin plate of quartz crystal
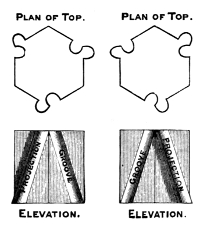 Fig. 16. Fig. 17.
cut from any position perpendicular to a pair of faces
of a symmetrical crystal, becomes positively electrified
on one side and negatively on the other when pulled
in a direction perpendicular to those faces. But this
assemblage has not the chiral piezo-electric quality
discovered theoretically by
Voigt19, and experimentally
in quartz and in tourmaline
by himself and Riecke20, nor
the well-known optic chirality
of quartz.
Fig. 16. Fig. 17.
cut from any position perpendicular to a pair of faces
of a symmetrical crystal, becomes positively electrified
on one side and negatively on the other when pulled
in a direction perpendicular to those faces. But this
assemblage has not the chiral piezo-electric quality
discovered theoretically by
Voigt19, and experimentally
in quartz and in tourmaline
by himself and Riecke20, nor
the well-known optic chirality
of quartz.
§ 49. Change now the directions of the grooves and fillets to either of the oblique configurations shown in Fig. 16, which I call right-handed, because the directions of the projections are tangential to the threads of a three-thread right-handed screw, and Fig. 17 (left-handed). The prisms with their grooves and fillets will still all fit together if they are all right-handed, or all left-handed.
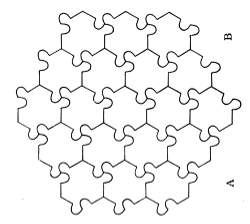
Fig. 18 shows the upper side of a hexagonal layer of an assemblage thus composed of the right-handed molecule of Fig. 16. Fig. 15 unchanged, still represents a horizontal section through the centres of the molecules. A prism built up of such layers, and finished at each end with a pyramid according to the rule of § 48, has all the qualities of ternary chiral symmetry required for the piezo-electricity of quartz; for the orientational differences of the alternate pairs of prismatic faces; for the absolute difference between the alternate pairs of faces of each pyramid which are shown in the etching by hydrofluoric acid; for the merely orientational difference between the parallel faces of the two pyramids; and for the well-known chiro-optic21 property of quartz. Look at two contiguous faces A, B of our geometrical model quartz crystal now before you, with its axis vertical. You will see a difference between them: turn it upside 56down; B will be undistinguishable from what A was, and A will be undistinguishable from what B was. Look at the two terminal pyramids, and you will find that the face above A and the face below B are identical in quality, and that they differ from the face above B and below A. This model is composed of the right-handed constituent molecules shown in Fig. 16. It is so placed before you that the edge of the prismatic part of the assemblage nearest to you shows you filleted faces of the prismatic molecules. You see two pyramidal faces; the one to your right hand, over B, presents complicated projections and hollows at the corners of the constituent molecules; and the pyramidal face next your left hand, over A, presents their unmodified corners. But it will be the face next your left hand which will present the complex bristling corners, and the face next your right hand that will present the simple corners, if, for the model before you, you substitute a model composed of left-handed molecules such as those shown in Fig. 17.
§ 50. To give all the qualities of symmetry and anti-symmetry of the pyro-electric and piezo-electric properties of tourmaline investigated theoretically by Voigt22, and experimentally by himself and Friecke23, make a hollow in one terminal face of each of our constituent prisms, and a corresponding projection in its other terminal face.
§ 51. Coming back to quartz, we can now understand
perfectly the two kinds of macling which are well
known to mineralogists as being found in many natural
specimens of the crystal, and which I call respectively
57the orientational macling, and the chiral macling. In
the orientational macling all the crystalline molecules
are right-handed, or all left-handed; but through all
of some part of the crystal, each of our component
hexagonal prisms is turned round its axis through 60°
from the position it would have if the structure were
homogeneous throughout. In each of the two parts
the structure is homogeneous, and possesses all the
electric and optic properties which any homogeneous
portion of quartz crystal presents, and the facial properties
of natural uncut crystal, shown in the etching
by hydrofluoric acid; but there is a discontinuity at
the interface, not generally plane, between the two
parts, which in our geometrical model would be shown
by non-fittings between the molecules on the two sides
of the interface, while all the contiguous molecules in
one part, and all the contiguous molecules in the other
part, fit into one another perfectly. In chiral macling,
which is continually found in amethystine quartz, and
sometimes in ordinary clear quartz crystals, some parts
are composed of right-handed molecules, and others of
left-handed molecules. It is not known whether, in
this chiral macling, there is or there is not also the
orientational macling on the two sides of each interface;
but we may say probably not; because we know that
the orientational macling occurs in nature without any
chiral macling, and because there does not seem reason
to expect that chiral macling would imply orientational
macling on the two sides of the same interface. I would
like to have spoken to you more of this most interesting
subject; and to have pointed out to you that some
of the simplest and most natural suppositions we can
make as to the chemical forces (or electrical forces,58
which probably means the same thing) concerned in
a single chemical molecule of quartz, SiO2, and acting
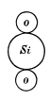 Fig. 19.
between it and similar neighbouring molecules,
would lead essentially to these molecules coming
together in triplets, each necessarily either right-handed
or left-handed, but with as much probability
of one configuration as of the other: and
to have shown you that these triplets of silica
3(SiO2) can form a crystalline molecule with
all the properties of ternary chiral symmetry, typified
by our grooved hexagonal prisms, and can build
up a quartz crystal by the fortuitous concourse of
atoms. I should like also to have suggested and explained
the possibility that a right-handed crystalline
molecule thus formed may, in natural circumstances
of high temperature, or even of great pressure, become
changed into a left-handed crystal, or vice-versa. My
watch, however, warns me that I must not enter on
this subject.
Fig. 19.
between it and similar neighbouring molecules,
would lead essentially to these molecules coming
together in triplets, each necessarily either right-handed
or left-handed, but with as much probability
of one configuration as of the other: and
to have shown you that these triplets of silica
3(SiO2) can form a crystalline molecule with
all the properties of ternary chiral symmetry, typified
by our grooved hexagonal prisms, and can build
up a quartz crystal by the fortuitous concourse of
atoms. I should like also to have suggested and explained
the possibility that a right-handed crystalline
molecule thus formed may, in natural circumstances
of high temperature, or even of great pressure, become
changed into a left-handed crystal, or vice-versa. My
watch, however, warns me that I must not enter on
this subject.
§ 52. Coming back to mere molecular tactics of crystals, remark that our assemblage of rounded, thoroughly scalene, tetrahedrons, shown in the stereoscopic picture (§ 36, Fig. 13 above), essentially has chirality because each constituent tetrahedron, if wholly scalene, has chirality24. I should like to have explained to you how a single or double homogeneous assemblage of points has essentially no chirality, and how three assemblages of single points, or a single assemblage of triplets of points, can have chirality, though a single triplet of points cannot have chirality. I should like indeed to have brought somewhat thoroughly before you the geometrical theory of chirality; and in 59illustration to have explained the conditions under which four points, or two lines, or a line and two points, or a combination of point, line and plane, can have chirality: and how a homogeneous assemblage of non-chiral objects can have chirality; but in pity I forbear, and I thank you for the extreme patience with which you have listened to me.
2 The holes in the cylinders are bored obliquely, as shown in Fig. 4, which causes them to remain at any desired position on the cord and allows them to be freed to move up and down by slackening the cord for a moment.
3 ‘On the Homogeneous Division of Space,’ by Lord Kelvin, Royal Society Proceedings, vol. lv, Jan. 18, 1894.
4 Similar curves are said to be parallel when the tangents to them at corresponding points are parallel.
6 ‘On the Division of Space with Minimum Partitional Area,’ Philosophical Magazine, vol. xxiv, 1887, p. 502, and Acta Mathematica of the same year.
7 A. Levy, Edinburgh Philosophical Journal, April, 1822; Whewell, Phil. Trans. Royal Society, 1825; Miller, Treatise on Crystallography.
8 I call any geometrical figure, or group of points, chiral, and say that it has chirality, if its image in a plane mirror, ideally realized, cannot be brought to coincide with itself. Two equal and similar right hands are homochirally similar. Equal and similar right and left hands are heterochirally similar or ‘allochirally’ similar (but heterochirally is better). These are also called ‘enantiomorphs,’ after a usage introduced, I believe, by German writers. Any chiral object and its image in a plane mirror are heterochirally similar.
9 Philosophical Magazine, vol. xx, 1885, second half year, p. 469, and British Association Report, 1885, Aberdeen, p. 896.
10 The solids of the photograph are castings in fine plaster of Paris from a scalene tetrahedron of paraffin wax, with its corners and edges rounded, used as a pattern.
11 ‘A twin-crystal is composed of two crystals joined together in such a manner that one would come into the position of the other by revolving through two right angles round an axis which is perpendicular to a plane which either is, or may be, a face of either crystal. The axis will be called the twin-axis, and the plane to which it is perpendicular the twin-plane.’ Miller’s Treatise on Crystallography, p. 103. In the text the word ‘twin-plane,’ quoted from the writings of Stokes and Rayleigh, is used to signify the plane common to the two crystals in each of the cases referred to: and not the plane perpendicular to this plane, in which one part of the crystal must be rotated to bring it into coincidence with the other, and which is the twin-plane as defined by Miller.
12 ‘A clear transparent crystal of potassium chlorate, from which the inevitable twin-plate had been ground away so as to reduce it to a single crystal film about 1 mm. in thickness, was placed between pieces of mica and laid on a thick iron plate. About 3 cm. from it was laid a small bit of potassium chlorate, and the heat of a Bunsen burner was applied below this latter, so as to obtain an indication when the temperature of the plate was approaching the fusing-point of the substance (359° C according to Prof. Carnelly). The crystal plate was carefully watched during the heating, but no depreciation took place, and no visible alteration was observed, up to the point at which the small sentinel crystal immediately over the burner began to fuse. The lamp was now withdrawn, and when the temperature had sunk a few degrees a remarkable change spread quickly and quietly over the crystal plate, causing it to reflect light almost as brilliantly as if a film of silver had been deposited upon it. No further alteration occurred during the cooling; and the plate, after being ground and polished on both sides, was mounted with Canada balsam between glass plates for examination. Many crystals have been similarly treated with precisely similar results; and the temperature at which the change takes place, has been determined to lie between 245° and 248°, by heating the plates upon a bath of melted tin in which a thermometer was immersed. With single crystal plates no decrepitation has ever been observed, while with the ordinary twinned-plates it always occurs more or less violently, each fragment showing the brilliant reflective power above noticed.’—Nature, May 20, 1886.
13 Nature, May 20, 1886.
14 Philosophical Magazine, 1888, second half year, p. 260.
16 Widmanstätten, 1807. Leydolt (1855, Wien. Akad. Ber. 15, 59, T. 9, 10. Baumhauer, Pogg. Ann. 138, 563 (1869); 140, 271; 142, 324; 145, 460; 150, 619.) For an account of these investigations, see Mallard, Traité de Crystallographie (Paris, 1884), Tome II, chapitre xvi.
17 More exactly .9525, being 3/4 × cot 38° 13′; see p. 53.
18 J. and P. Curie and C. Friedel, Comptes Rendus, 1882, 1883, 1886, 1892.
19 Allgemeine Theorie der piëzo- und pyroelectrischen Erscheinungen an Krystallen. W. Voigt, Königl. Gesellschaft der Wissenschaften zu Göttingen, August 2, 1890.
20 Wiedemann, Annalen, 1892, xlv, p. 923.
21 Generally miscalled ‘rotational.’
22 See foot-note (2) to p. 54 above.
23 See foot-note (3) to p. 54 above.
THE END
Oxford
PRINTED AT THE CLARENDON PRESS
BY HORACE HART, PRINTER TO THE UNIVERSITY