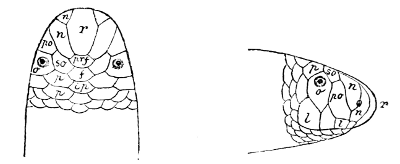
Fig. 1—Head of Typhlops braminus. (From “Fauna of British India”)
f, Frontal; ip, interparietal; l, labial; n, nasal; o, ocular; p, parietal; po, preocular; prf, prefrontal; r, rostral; so, supraocular.
Title: The Snakes of Europe
Author: George Albert Boulenger
Release date: August 15, 2014 [eBook #46590]
Most recently updated: October 24, 2024
Language: English
Other information and formats: www.gutenberg.org/ebooks/46590
Credits: Produced by Chris Curnow, Sonya Schermann and the Online
Distributed Proofreading Team at http://www.pgdp.net (This
file was produced from images generously made available
by The Internet Archive)
Transcriber's Notes:
This text uses the symbols for male (♂) and female (♀). These symbols may not be correctly displayed by some e-reader devices. An upside-down Y has been represented as [Y].
Some presumed printer's errors have been corrected. In particular, punctuation has been normalized and entries in the Index were altered to match the main text.
THE LIFE OF CRUSTACEA
BRITISH FRESHWATER FISHES
THE OX AND ITS KINDRED
THE LIFE OF THE MOLLUSCA
There is no work in the English language dealing with the Reptiles of Europe. I have therefore endeavoured to supply this desideratum, so far as the Snakes are concerned, by drawing up in a concise form an account of what is known of their characters, their distribution, and their life-histories. Professor Sordelli, of Milan, having kindly acceded to my request to reproduce some of the beautiful figures drawn by him for the work published in collaboration with the late Professor Jan under the title of “Iconographie Générale des Ophidiens,” I have been able to supplement my descriptions with illustrations which leave nothing to be desired from the point of view of accuracy. A few drawings have been made specially for this book by Mr. J. Green. I have further to acknowledge the permission given by the Trustees of the British Museum, the India Office, and the Zoological Society, to reproduce a few figures from previous publications of which I am the author.
In order to render this little book more useful, the account of the Snakes of Europe has been preceded by an Introduction summarizing what is known of Snakes generally.
I have purposely avoided overburdening a work of this kind, which aims at concision, with bibliographical references and synonymic lists. I am sure my readers will be thankful for being spared this display of erudition. Whenever I have had to compile, and to trespass on ground that is not my own, I have been careful to draw only from the writings of the most trustworthy authorities. The descriptions of the species are based on the collection in the British Museum, which has been considerably increased since the publication of the Catalogue of Snakes (1893-1896). I have also had access to Monsieur F. Lataste’s rich private collection, now under my care, and Dr. R. Gestro has kindly entrusted to me for study the collection of Italian Snakes in the Genoa Museum. I am indebted to Dr. L. W. Sambon for the chapter on Parasites, which he has written at my request.
To all who have helped me I beg to tender my hearty thanks.
| INTRODUCTION | ||
| CHAPTER | PAGE | |
| I. | Definition and Classification | 1 |
| II. | External Characters—Integument | 8 |
| III. | Coloration | 29 |
| IV. | Skeleton | 40 |
| V. | Dentition | 53 |
| VI. | Poison Apparatus—Different Kinds of Poisons | 62 |
| VII. | Nervous System-Sense Organs | 73 |
| VIII. | Viscera | 77 |
| IX. | Organs of Reproduction; Pairing; Oviposition; Development | 82 |
| X. | Habits | 91 |
| XI. | Parasites | 107 |
| XII. | Distribution | 118 |
| XIII. | Snakes in Relation to Man | 133 |
| SYSTEMATIC ACCOUNT OF THE SNAKES | ||
| OF EUROPE | ||
| First Family: TYPHLOPIDÆ | ||
| Genus TYPHLOPS, Schneider | 144 | |
| 1. | Typhlops vermicularis, Merrem—The Greek Blind-Snake | 144 |
| Second Family: BOIDÆ | ||
| Genus ERYX, Daudin | 147 | |
| 2. | Eryx jaculus, Linnæus—The Javelin Sand-Boa | 147 |
| Third Family: COLUBRIDÆ | ||
| Genus TROPIDONOTUS, Kuhl | 152 | |
| 3. | Tropidonotus natrix, Linnæus—The Grass-Snake, | |
| or Ring-Snake | 152 | |
| 4. | Tropidonotus tessellatus, Laurenti—The Tessellated | |
| Water-Snake | 160 | |
| 5. | Tropidonotus viperinus, Latreille—The Viperine | |
| Water-Snake | 165 | |
| Genus ZAMENIS, Wagler | 170 | |
| 6. | Zamenis gemonensis, Laurenti—The European Whip-Snake | 170 |
| 7. | Zamenis dahlii, Fitzinger—Dahl’s Whip-Snake | 177 |
| 8. | Zamenis hippocrepis, Linnæus—The Horseshoe Whip-Snake | 179 |
| Genus COLUBER, Linnæus | 181 | |
| 9. | Coluber quatuorlineatus, Lacepède—Aldrovandi’s Snake | 182 |
| 10. | Coluber dione, Pallas—The Dione Snake | 185 |
| 11. | Coluber longissimus, Laurenti—The Æsculapian Snake | 187 |
| 12. | Coluber leopardinus, Bonaparte—The Leopard Snake | 191 |
| 13. | Coluber scalaris, Schinz—The Ladder Snake | 194 |
| Genus CORONELLA, Laurenti | 196 | |
| 14. | Coronella austriaca, Laurenti—The Smooth Snake | 197 |
| 15. | Coronella girondica, Daudin—The Southern Smooth Snake | 202 |
| Genus CONTIA, Baird and Girard | 205 | |
| 16. | Contia modesta, Martin—The Dwarf Snake | 205 |
| Genus CŒLOPELTIS, Wagler | 207 | |
| 17. | Cœlopeltis monspessulana, Hermann—The Montpellier Snake | 208 |
| Genus MACROPROTODON, Guichenot | 212 | |
| 18. | Macroprotodon cucullatus, I. Geoffroy—The False | |
| Smooth Snake | 213 | |
| Genus TARBOPHIS, Fleischmann | 216 | |
| 19. | Tarbophis fallax, Fleischmann—The Cat-Snake | 217 |
| 20. | Tarbophis iberus, Eichwald—The Caucasian Cat-Snake | 219 |
| Fourth Family: VIPERIDÆ | ||
| Genus VIPERA, Laurenti | 221 | |
| 21. | Vipera ursinii, Bonaparte—Orsini’s Viper | 221 |
| 22. | Vipera renardi, Christoph—Renard’s Viper | 227 |
| 23. | Vipera berus, Linnæus—The Northern Viper, or Adder | 230 |
| 24. | Vipera aspis, Linnæus—The Asp Viper | 239 |
| 25. | Vipera latastii, Bosca—Lataste’s Viper | 247 |
| 26. | Vipera ammodytes, Linnæus—The Sand-Viper, or | |
| Long-Nosed Viper | 249 | |
| 27. | Vipera lebetina, Linnæus—The Blunt-Nosed Viper, or Kufi | 257 |
| Genus ANCISTRODON, Palisot de Beauvois | 261 | |
| 28. | Ancistrodon halys, Pallas—Pallas’s Pit-Viper | 262 |
| Index | 265 | |
| PLATE | FACING PAGE | |
| I. | Typhlops vermicularis, Eryx jaculus | 144 |
| II. | Tropidonotus natrix and Vars. cettii and persa | 152 |
| III. | Tropidonotus tessellatus, T. viperinus and | |
| var. aurolineatus | 160 | |
| IV. | Zamenis gemonensis and vars. persica and | |
| viridiflavus | 170 | |
| V. | Zamenis gemonensis, var. caspius, Z. dahlii, | |
| Z. hippocrepis | 176 | |
| VI. | Coluber quatuorlineatus and var. sauromates, | |
| C. dione | 182 | |
| VII. | Coluber longissimus, C. leopardinus and | |
| var. quadrilineatus | 188 | |
| VIII. | Coluber scalaris | 194 |
| IX. | Coronella austriaca | 196 |
| X. | Coronella girondica, Contia modesta | 202 |
| XI. | Cœlopeltis monspessulana, Macroprotodon | |
| cucullatus, Tarbophis iberus, T. fallax | 208 | |
| XII. | Vipera ursinii, V. renardi, V. berus | 220 |
| XIII. | Vipera aspis, V. latastii | 240 |
| XIV. | Vipera lebetina, V. ammodytes, Ancistrodon halys | 250 |
Snakes, Ophidia—regarded by some authorities as an order of the class Reptilia, by the author as a sub-order of the order Squamata, which includes besides the Lizards, Lacertilia, the Chameleons, Rhiptoglossa, and the extinct Dolichosauria and Mosasauria—may be defined as greatly elongate scaly Reptiles without limbs, or with mere vestiges of the hind pair, without movable eyelids, without ear-opening, with elongate, deeply forked tongue retractile into a basal sheath, with transverse vent and paired copulatory organs, and with the two halves of the lower jaw independently movable, connected at the symphysis by an elastic ligament.
The latter character alone distinguishes them from all Lizards, but no single Lizard possesses all the others in combination.
In their most highly developed form these Reptiles are adapted for rapid reptation and for swallowing 2 prey much exceeding their own calibre; hence the bones of the skull, on which a prehensile function devolves, are loosely attached to the cranium by ligamentous elastic tissue, or articulated in such a manner as to permit a wide buccal expansion; whilst the absence of a sternum and the mobile attachment of the ribs allow a corresponding dilatation of the body as the prey descends into the digestive canal.
The fatal venom which many of these Reptiles possess has so impressed the mind of men, even the scientific, that for a long time snakes were primarily divided into poisonous and non-poisonous, a classification in which the more important characters, derived from the general structure, and especially from the skull, were subordinated to the physiological. Such a system was far from reflecting natural relationships. Besides, as our knowledge progressed, drawing a distinction between poisonous and harmless snakes became more and more difficult, so many snakes previously regarded as harmless proving to be poisonous in various degrees—at least enough to paralyze the small prey on which they subsist, if not to be of serious danger to man.
In the division into families, as followed in this work, the presence or absence of a poison organ is left out of consideration. Further, in this as in many other groups of the animal kingdom, external characters do not furnish trustworthy indications for higher divisions, and the definitions of the 3 families are therefore based exclusively on osteological characters. For those who wish to name snakes with facility, the key which concludes the chapter on External Characters will, however, remedy this defect, and suffice for the identification of all the European species without any reference to their anatomy. Many attempts have been made to furnish an easy criterion for the distinction of harmless from poisonous snakes, but the characters hitherto suggested with this object can only be applied successfully to the small number of representatives in a limited area. Thus, in Southern Australia it might be stated that all snakes showing the regular nine large shields on the upper surface of the head are dangerous to man, whilst those with small shields or scales are harmless; but in most parts of Europe this criterion would have to be reversed. In some countries the shape of the pupil might be used for the purpose, in others the size of the ventral shields, or the presence or absence of a loreal shield, between the nasal and the preocular, and so on. But when we have to deal with the snakes of the whole world, about 2,000 species, of which nearly one-third are poisonous to a greater or less degree, every attempt at a definition of the two categories without regard to the dentition breaks down. Only those who have made a study of the snakes of the world can make a guess from the general appearance as to an unknown form being poisonous or not, and 4 even they may sometimes feel embarrassed, unless the dentition be examined; the mistakes which have occasionally been made by some experienced herpetologists are proof sufficient of the fallacy of external characters for this purpose.
The Ophidia are divided into nine families, the first, third, seventh, and ninth of which have representatives in Europe:
I. No transverse (ectopterygoid) bone; pterygoid not extending to quadrate or mandible; no supratemporal; nasals in contact with prefrontals; coronoid present; vestiges of pelvis.
Maxillary loosely attached to lower surface of cranium, toothed; lower jaw edentulous; a single pelvic bone
Maxillary bordering mouth, forming a suture with premaxillary, prefrontal, and frontal, toothless; pubis and ischium present, latter forming a symphysis
II. Transverse bone present; both jaws toothed.
A. Coronoid present; nasals in contact with prefrontals.
1. Vestiges of pelvis; supratemporal present.
Supratemporal large, suspending quadrate
(Subfamilies: Pythoninæ, Boinæ.)
Supratemporal small, intercalated in the cranial wall
5 2. No vestiges of pelvis; supratemporal absent
B. Coronoid absent; supratemporal present.
1. Maxillary horizontal; pterygoids reaching quadrate or mandible.
Nasals in contact with prefrontals
Nasals not in contact with prefrontals
Three series: A. Aglypha (subfamilies: Acrochordinæ, Colubrinæ, Dasypeltinæ); B. Opisthoglypha (Homalopsinæ, Dipsadomorphinæ, Elachistodontinæ); C. Proteroglypha (Hydrophiinæ, Elapinæ).
2. Maxillary horizontal, converging posteriorly towards palatine; pterygoid not reaching quadrate or mandible
3. Maxillary vertically erectile perpendicularly to transverse bone; pterygoid reaching quadrate or mandible
(Subfamilies: Viperinæ, Crotalinæ.)
The technical terms employed in the above synopsis will be found explained and illustrated by figures in the chapter on the Skeleton.
No serial arrangement can express the affinities of the various groups as conceived by the classificator; a diagram therefore follows to show the author’s views as to their interrelationships, and possibly their phylogeny. Leaving aside the Typhlopidæ and Glauconiidæ, which should be regarded 6 as burrowing types independently derived from some Ophidian form less specialized than any with which we are at present acquainted, and probably without direct relationship to the Lizards, the family Boidæ, and more especially the Pythons, claim the position of ancestral group, from which all other snakes may have been derived.
| Viperidæ | Amblycephalidæ | |||||||
| Colubridæ opisthoglyphæ | Colubridæ proteroglyphæ | |||||||
| Uropeltidæ | ||||||||
| Ilysiidæ | Xenopeltidæ | Colubridæ aglyphæ | ||||||
| Boidæ | ||||||||
Further remarks on this subject in the chapter on Dentition.
It is to be regretted that paleontology cannot help us at present as concerns the lines of evolution, the comparatively few fossil Ophidians known, from the Lower Eocene upwards, the remains of which can be identified with some measure of certainty, being either non-poisonous types (Boidæ, Ilysiidæ, Palæophiidæ, Colubridæ) or Viperidæ (Viperines from the Miocene of France and Germany, Crotalines from the Miocene of North America). The vertebræ from the Puerco Eocene of America, on the limit between the Cretaceous and Eocene periods, described as the 7 oldest snake remains, Helagras, Cope, are stated to approach the Lacertilian type.
Whether the vertebræ named Symoliophis, Sauvage, from the chalk of France, and Coniophis, Marsh, from the Laramie Cretaceous of North America, are Ophidian, as claimed by their describers, or Dolichosaurian, cannot be decided without further material.
The form varies enormously, worm-like in some, comparatively short and heavy, elongate and more or less slender, or extremely gracile and almost filiform, in others. In this respect our common Grass-snake occupies a central position, and for this reason is termed a moderately slender form, anything above or below this standard being described as comparatively short or elongate. Our shortest and stoutest European Snakes are the Vipers, especially Vipera ursinii; our longest and slenderest, the Coluber and Zamenis, especially Zamenis dahlii. These extremes in both directions are, however, far surpassed by many exotic snakes, as we find on comparing, for instance, one of the African Puff-adders (Bitis), with certain Oxybelis and Leptognathus from Tropical America. The body may be somewhat rigid, as in some burrowing and ground snakes, not unlike in appearance to our Slow-worm and other limbless Lizards; or extremely flexible, as in many Pythons and Boas and in the Tree-snakes generally. This flexibility may be accompanied by a vertical compression of the body in relation with an arboreal existence, whilst sluggish snakes, such as most of the 9 Viperidæ, may be remarkable for the flattening of the body, which they may further increase when basking in the sun or in order to assume a more formidable appearance on the approach of an enemy. This power of flattening out the whole or the anterior part of the body is possessed by many snakes, poisonous as well as harmless, and reaches its highest degree in the Cobras of India and Africa, the expanded anterior part being known as the “hood,” from the Portuguese name “Cobra di capello.”
Thoroughly aquatic snakes are often short and heavy, but some of the marine forms, or Hydrophids, may be extremely slender, with the posterior part of the body compressed. In some of these Sea-snakes the gracility of the anterior part, or “neck,” as it has been called, contrasts very strikingly with the great girth of the body towards the tail, and suggests a limbless Plesiosaur.
The tail, the part of the body behind the transversely cleft vent, is most frequently about one-fourth or one-fifth of the total length; but it may be much shorter, even reduced to a mere stump, as in the Typhlops, or, at the opposite extreme, enter for one half in the length of the snake, as in the African Xenurophis. This organ may taper gradually to a fine point; or end abruptly, as if mutilated; or terminate in a horny spine, such as we see in some of the Typhlops or in the Australian Death-adder, 10 Acanthophis, or in a series of horny segments which are vibrated like a rattle, as in the well-known Crotalus of America, to which we shall refer again at the end of this chapter. In some of the burrowing Uropeltidæ, the very short tail is obliquely truncated, with indurated shields above, and acts as a trowel. And, finally, the marine snakes of the subfamily Hydrophiinæ are distinguished by a strongly compressed, oar-shaped tail, with rounded vertical outline. In a few forms, arboreal or aquatic, the tail is more or less prehensile.
Males generally have a longer tail than females, and the genital organs, which are lodged in its base, cause a swelling of that region which contrasts with the more gradually tapering extremity of the female, thus affording a means of distinguishing the sexes externally in the majority of snakes.
The rudimentary hind limbs of Boid snakes, to be mentioned further on in the description of the skeleton, terminate in a claw-like horny spur, which appears on each side of the vent in the male, and sometimes also, though less distinctly, in the female. These spurs are probably of use in facilitating the pairing, an explanation which appears the more plausible from the fact that the snakes provided with them have the copulatory intromittent organs destitute of the erectile spines which are present in most others.
The head varies in shape as much as the body. 11 Although never actually compressed, except in the rostral region, it may be very narrow and elongate, whilst in the opposite extreme it may be strongly depressed, and so broad behind as to be abruptly defined from the anterior part of the body, or “neck.” This feature is very marked in some of the Viperidæ, and this has given rise to the incorrect generalization that poisonous snakes are distinguished from the harmless by a broad and flat head, notwithstanding the fact that some of the most dangerous, such as the Mambas, Cobras, and Kraits, have a comparatively narrow or small head, not or but slightly defined behind, whilst, on the other hand, the very opposite condition obtains in not a few of the harmless Colubrids.
Leaving the Typhlopidæ and Glauconiidæ aside for the present, snakes have a wide gape, cleft far beyond the vertical of the eyes, with, when closed, one or two notches in front for the passage of the protrusible, bifid tongue. In most snakes this chink is in the lower border of the rostral shield, capping the tip of the snout, and allows free passage to the whole tongue; in the Hydrophids, or Sea-snakes, there are two notches in the lower border of the rostral shield, through which only the bifid end of the tongue can be protruded. The eyes, varying from minute to enormous, are usually free from the surrounding shields, and may move under a transparent cap like a watch-glass, which appears to 12 represent the lower eyelid of Lizards. The view as to this homology is derived from our knowledge of various conditions in certain series of Lizards of the families Lacertidæ and Scincidæ, where we find a transparent disc appearing like a small window in the movable lower eyelid, gradually increasing in size so as to occupy the whole of the lower eyelid, which finally becomes fused with the rudimentary upper lid and loses its mobility. In Ilysia and in most of the Uropeltidæ, the transparent disc over the eye is confluent with a thick horny shield of which it occupies the middle.
The pupil is usually circular or vertical, rarely horizontal. In some forms it is difficult to decide whether it is round or vertically elliptic; in others, like the Boas and Vipers, for instance, it is decidedly vertical, and contracts to the same extent as a cat’s. In some Water-snakes, and in Sea-snakes generally, the round pupil may contract to a mere dot. The contraction of the pupil is independent on the two sides.
The snout, or the part of the head anterior to the eyes, may be short or long, rounded or pointed, depressed or compressed, sometimes projecting strongly beyond the mouth, turned up at the end, or terminating in one (Langaha) or two (Herpeton) long scaly dermal appendages. In some burrowing forms it is provided with a more or less trenchant horizontal or vertical edge. When the sides of the snout (loreal region) form an angle with the upper surface, 13 the angle is termed the “canthus rostralis,” which may be intensified by the loreal region being concave.
The deep pits which are sometimes present on the lips or between the nostril and the eye (loreal pit) will be alluded to further on under Sensory Organs.
The nostrils are either lateral, or, in the aquatic forms, directed upwards, sometimes entirely on the upper surface of the snout.
Most snakes have a longitudinal groove on the chin (mental groove) to allow for the distension caused by the lateral movements of the rami of the lower jaw.
In the Typhlopidæ, the head passes gradually into the vermiform body, and the small mouth is situated on the under surface of the projecting snout; the head so resembles the extremely short tail, and the mouth is so similar in shape and position to the vent, which is close to the posterior extremity of the snake, that such creatures are often believed by non-critical observers to have a head at each end. The eyes are very small, and covered over by the semi-transparent head-shields, or they may be completely concealed. There is no mental groove. It is much the same with the Glauconiidæ, which have, however, a somewhat less abbreviated tail. In both, the nostrils often open on the lower side of the snout, which may be excavated so as to appear hooked in profile, or may be provided with a sharp cutting horizontal edge.
14 Snakes are covered with epidermal folds in the form of scales and shields, the shape and arrangement of which affords important characters for their classification. Dermal ossifications are absent.
The scales on the body are usually elliptic or lanceolate and imbricate, forming straight longitudinal and oblique transverse series, and they are replaced on the belly and under the tail by transverse shields mostly corresponding in number with the series of scales, and also with the vertebræ. The body of the Typhlopidæ and Glauconiidæ is uniformly covered with polished, closely adherent, rounded, overlapping, sub-equal scales, without even an indication of ventral shields. In some of the Acrochordinæ, aberrant aquatic Colubrids, the scaling consists, above and beneath, of small juxtaposed, sometimes spinose granules, the skin being suggestive of the shagreen of sharks. In the marine snakes of the subfamily Hydrophiinæ, the ventral shields are often absent or merely indicated, and the scales are mostly juxtaposed or feebly imbricate, sometimes tetragonal or hexagonal, and occasionally studded with spinose tubercles. In the more typical Ophidia the imbricate scales may be long and narrow or short and broad, with every intermediate step between the two extremes; smooth or furnished with a longitudinal ridge or keel, or even several keels; nearly equal in size or with the median or outer series more or less enlarged, the longitudinal series in odd, rarely in even number; instead of running 15 in longitudinal series parallel with the axis of the body, as is the rule, they are sometimes disposed obliquely, and among those in which we meet with this peculiarity several genera are further remarkable in having some of the oblique lateral scales furnished with a serrated keel, to which we shall again allude in the chapter on Habits, when dealing with the rustling sounds produced by certain snakes. The number of longitudinal series of scales on the body varies from 10 (Herpetodryas) to nearly 100 (Python, Boa); in the European species from 17 (Contia modesta) to 50 (Eryx jaculus). The scales are sometimes furnished near the end with one or two shallow impressions, termed “apical pits,” which afford indications for the distinction of genera and species; unless of a lighter or darker colour, as is often the case, these pits are not always easy to see, except in a strong light and with the aid of a powerful magnifying glass.
The ventral shields, also called “gastrosteges,” usually occupy the whole width of the belly; but they may be much narrower—in Eryx, for instance. They are sometimes bent at an angle on the sides, and this angle may even form a sharp keel, accompanied by a notch in the posterior border, corresponding to the keel, as in several of the more arboreal genera of Colubrids. The shields under the tail, termed subcaudals or “urosteges,” are sometimes similar to the ventrals, but more often disposed in pairs; in certain 16 species or individuals some of the subcaudals are single, and the others paired. When the number of subcaudals is given in the descriptions, each pair is reckoned as one, and the conical or spine-like shield which caps the end of the tail is not included. These numbers afford important characters for the definition of species, and sometimes also for the distinction of sexes. The subcaudals are nearly always much fewer than the ventrals, but the difference is often not so great in the males as in the females, the tail of which is usually shorter in proportion to the body. It is noteworthy that in many species, if the number of subcaudals (C.) be added to that of the ventrals (V.), the total is nearly the same in the male as in the female, however much the respective numbers may differ when taken separately. The following figures may be given by way of example, taken from British specimens:
| Coronella austriaca: | ♂ | V. 154; | C. 58 = 212 |
| Coronella austriaca: | ♀ | V. 165; | C. 48 = 213 |
| Vipera berus: | ♂ | V. 138; | C. 35 = 173 |
| Vipera berus: | ♀ | V. 144; | C. 29 = 173 |
Although this rule is by no means universal, and does not apply at all to some species, it will be found to hold good in many cases, and is of interest in showing that the changes that have taken place in the vertebral column (the vertebræ corresponding in number to the shields), according to the sexes, have 17 been by a modification of the character of the segments about the anal region, a conversion of trunk vertebræ into caudals, or vice versa. In dealing with certain species—of Vipers, for instance—it is important, for systematic purposes, to keep the counts of shields distinct for the two sexes.
The shield which covers the vent, the anal shield, is either single or divided into two.
Some snakes have the head covered with scales or small tubercles similar to those on the body, but in the great majority the lepidosis is in the form of large symmetrical juxtaposed shields, the shape, proportions, and number of which furnish some of the most important characters for the distinction of genera and species. These head-shields belong to two primarily different types, from each of which all further modifications may be regarded as derived by alteration in shape or by disintegration. The first type is that shown by the Typhlopidæ and Glauconiidæ, which is explained by the figure on the next page.
The rostral, which is usually the largest of the head-shields, extends to the upper surface of the head, of which it may occupy the greater part. In the Glauconiidæ, the ocular usually borders the mouth.
As may be seen by a comparison of the first figure with the second, the arrangement of the head-shields is essentially different from that which prevails in the Colubrids and the majority of other snakes.
18 The second type is exemplified by the head of a member of the genus Zamenis.
In the descriptions, temporals 2 + 3 means two superposed temporals in the first row, three in the second. The internasals and the temporals, and the loreal and the preocular, are sometimes absent, and the prefrontal or the internasal may be single. One or two large shields are in rare cases present behind the parietals, and are called occipital.

Fig. 1—Head of Typhlops braminus. (From “Fauna of British India”)
f, Frontal; ip, interparietal; l, labial; n, nasal; o, ocular; p, parietal; po, preocular; prf, prefrontal; r, rostral; so, supraocular.
A breaking up into smaller shields takes place in many snakes. In the Pythons, for instance, the frontal may be divided into two by a longitudinal cleft, and separated from the prefrontals by small shields. In some Vipers, such as V. berus and V. ursinii, in which the frontal and parietals, though reduced in size, usually preserve their primitive condition, the former is normally separated from the supraocular by a series of small shields, and the internasals and prefrontals are broken up; in these 19 snakes the small shield or shields behind the rostral are termed “apical,” and those on the upper edge of the snout are termed “canthals.” The shield which, in Vipers, separates the rostral from the nasal is called “naso-rostral.” Allusion has been made above to the scaly dermal appendages which terminate the snout in certain genera. Some Viperidæ are furnished 20 with horn-like erect spines above the eyes or at the end of the snout, which add greatly to their sinistral appearance.
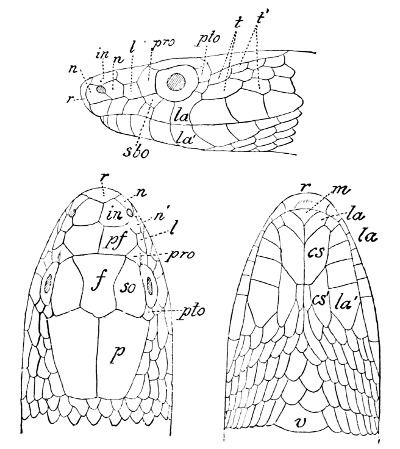
Fig. 2—Head of Zamenis ventrimaculatus. (From “Fauna of British India”)
cs, Chin-shields (anterior); cs´, chin-shields (posterior); f, frontal; in, internasal; l, loreal; la, labial (upper); la´, labial (lower); m, mental; n, nasal; p, parietal; pf, prefrontal; pro, preocular; pto, postocular; r, rostral; sbo, subocular; so, supraocular; t, temporals (first row); t´, temporals (second row); v, first ventral.
The periodical shedding of the outer layer of the epidermis in a single piece, including even the covering of the eye, is one of the most striking peculiarities of snakes, although paralleled in the Lizards of the family Anguidæ, to which our British Slow-worm belongs. The skin becomes detached at the lips, and is turned inside out from head to tail, without any sort of laceration when the snake is in good health. These exuviæ are transparent, but often carry a certain amount of pigment, especially those of the Vipers, in which the characteristic dark markings are perfectly visible; they usually exceed the length of the reptile, owing to stretching. In Sea-snakes the epidermis is cast piecemeal, and sloughing is a longer operation than in ordinary snakes.
In Rattlesnakes each piece of the rattle, or “crotalon,” in which the tail terminates, represents a retained portion of the sloughed epidermis. This remarkable appendage looks like a number of horny rings, but it consists in reality of hollow, bell-like pieces, similar to the terminal one, or “button,” each with a circular constriction, in which the incurved free edge of the following piece fits, thus keeping the pieces together without impairing the mobility necessary to produce the rattling sound for which 21 the apparatus is intended. At each exuviation one bell-shaped horny piece is added. The number of segments in the rattle is, therefore, not an index to age, as formerly believed; nor is it to the number of exuviations, for whilst segments are being added at the base of the apparatus the terminal ones break off and are lost. A Crotalus sixteen months old may have six pieces to the rattle if there have been six exuviations and no loss. No rattle appears ever to comprise more than about twenty pieces, even in old specimens. The size of the terminal button shows whether it was formed at birth or at any later period, no growth taking place in the horny tissue.
So far as trustworthy records are concerned, the largest snakes known, the Malay Python reticulatus and the South American Anaconda, Eunectes murinus, reach a length of 25 to 30 feet. Measurements of skins must be accepted with caution, as a skin may easily be stretched to once and a half its real length; in estimating the exact length from such a stretched skin, it is necessary to deduct the interstitial spaces showing between the scales, and about one-fourth of the scale to allow for the overlap. The smallest snake known is 4 inches long (Glauconia dissimilis). The largest European snake (Coluber quatuorlineatus) is reported to reach a length of 8 feet; the smallest (Typhlops vermicularis) does not exceed 14 inches.
I. Eyes minute, under the head-shields; mouth small, inferior; body vermiform, covered with uniform scales above and beneath; vent close to the end of the body, the extremely short tail ending in a small spine
II. Eyes very small, with vertical pupil; upper surface of head covered with small scales; ventral shields much narrower than the body; tail short, ending obtusely; subcaudals single, or mostly single; scales smooth or feebly keeled, in 40 to 50 rows
III. Eyes small, moderate, or large; ventral shields at least nearly as broad as the body; tail tapering to a point; subcaudals paired.
A. Pupil round; upper surface of head with nine large shields; no upper labial in contact with the parietal; anal shield usually divided.
1. Dorsal scales strongly keeled, with paired apical pits; a single anterior temporal.
a. Nostrils lateral; internasals broadly truncate in front.
Scales in 19 rows; normally 1 pre- and 3 postoculars; usually 7 upper labials, third and fourth entering the eye; ventrals 157-181; subcaudals 50-88
23 b. Nostrils directed upwards; internasals much narrowed in front.
Scales in 19 rows; normally 2 pre- and 3 or 4 postoculars; suboculars sometimes present; usually 8 upper labials, fourth or fourth and fifth entering the eye; ventrals 160-187; subcaudals 48-79
Scales in 21 (rarely 19 or 23) rows; normally 1 or 2 pre- and 2 postoculars; usually 7 upper labials, third and fourth entering the eye; ventrals 147-164; subcaudals 46-72
2. Dorsal scales smooth or feebly keeled; normally a single loreal.
a. Two or three superposed anterior temporals (very rarely one); nostril usually between two nasals.
α. A subocular below the preocular.
* Scales smooth, in 17 or 19 rows.
Two upper labials entering the eye; preocular not in contact with the frontal; scales with two apical pits; ventrals more or less distinctly angulate laterally, 160-230; subcaudals 87-131
Two upper labials entering the eye; preocular usually in contact with the frontal; scales with a single apical pit; ventrals very distinctly angulate laterally, 205-218; subcaudals 98-132
** Scales in 23 to 29 rows (usually 25 or 27), with two apical pits.
24 Upper labials usually separated from the eye by a series of suboculars; preocular in contact with the frontal; scales smooth; ventrals very distinctly angulate laterally, 222-258; subcaudals 77-107
Two upper labials entering the eye; preocular not in contact with the frontal; scales feebly but distinctly keeled; ventrals not angulate laterally, 195-234; subcaudals 56-90
Two upper labials entering the eye; preocular not in contact with the frontal; scales smooth or faintly keeled; ventrals not or but very obtusely angulate laterally, 172-214; subcaudals 50-80
β. No subocular; scales smooth, or faintly keeled on the posterior part of the body.
* Ventrals more than 200; scales with two apical pits.
Snout obtuse; rostral broader than deep; scales in 21 or 23 rows; ventrals distinctly angulate laterally, 212-248; subcaudals 60-91
Snout obtuse; rostral broader than deep; scales in 25 or 27 rows; ventrals not angulate laterally, 222-260; subcaudals 68-90
Snout pointed, strongly projecting; rostral deeper than broad, wedged in between the internasals; scales in 25 to 29 rows; ventrals not angulate laterally, 201-220; subcaudals 48-68
25 ** Ventrals not more than 200; scales mostly with a single apical pit.
Rostral at least as deep as broad, often wedged in between the internasals; usually 7 upper labials, third and fourth entering the eye; scales in 19 (rarely 21) rows; ventrals 153-199; subcaudals 41-70
Rostral broader than deep; usually 8 upper labials, fourth and fifth entering the eye; scales in 21 (rarely 19 or 23) rows; ventrals 170-200; subcaudals 49-72
b. A single anterior temporal; nostril in a single nasal; scales smooth, with single apical pits, in 17 rows; ventrals 150-191; subcaudals 53-78
3. Scales longitudinally grooved in the adult, in 17 or 19 rows; two loreals; canthus rostralis strongly marked; frontal very narrow, in contact with the preocular; ventrals 160-189; subcaudals 68-102
B. Pupil vertical or vertically subelliptic (sometimes appearing round in Macroprotodon).
1. Scales smooth, mostly with single apical pits; upper surface of head with nine large shields.
Frontal 11⁄2 to 2 times as long as broad; loreal separated from the eye by the preocular; one upper labial usually in contact with the parietal; scales in 19 to 23 (rarely 25) rows; ventrals 153-192; anal divided; subcaudals 40-54
26 Frontal 11⁄4 to 11⁄2 times as long as broad, much shorter than the parietals; loreal entering the eye; scales oblique, in 19 or 21 rows; ventrals 186-222; anal divided; subcaudals 48-73
Frontal 11⁄4 to 11⁄2 times as long as broad, nearly as long as the parietals; loreal entering the eye; scales oblique, in 19 or 21 rows; ventrals 203-235; anal entire; subcaudals 54-70
2. Scales keeled, with two apical pits; anal shield entire.
a. No pit between the nostril and the eye; upper head-shields small, if present; nasal separated from the rostral by a naso-rostral; eye separated from the upper labials by suboculars.
α. Snout not turned up at the end; supraocular usually extending posteriorly beyond the vertical of the posterior border of the eye; frontal and parietal shields usually well developed; usually a single series of scales between the eye and the upper labials.
Snout obtusely pointed, flat above, or with the canthus slightly raised; rostral usually in contact with a single apical shield, rarely with two; 6 to 9 upper labials, usually 7 or 8; scales in 19 rows, rarely 21; ventrals: ♂ 120-135, ♀ 125-142
27 Snout pointed, with raised canthus; rostral in contact with a single apical shield; 8 or 9 upper labials; scales in 21 rows, rarely 19; ventrals: ♂ 130-148, ♀ 130-150
Snout truncate or broadly rounded, flat above or with slightly raised canthus; rostral in contact with two apical shields, rarely with one; 8 or 9 upper labials; scales in 21 rows, rarely 19 or 23; ventrals: ♂ 132-150, ♀ 132-158
β. Snout usually more or less turned up at the end or produced into a scaly dermal appendage; supraocular not extending posteriorly beyond the vertical of the posterior border of the eye; frontal and parietals often absent or very small; 2 or 3 series of scales between the eye and the upper labials; 9 to 13 upper labials; scales in 21 or 23 rows, rarely 19 or 25.
Snout simply turned up, the raised portion bearing 2 or 3 scales; rostral not more than once and a half as deep as broad; ventrals: ♂ 134-158, ♀ 141-169
Snout simply turned up or produced into a small appendage, the raised portion with 5 or 6 (rarely 3) scales; rostral 11⁄2 to 2 times as deep as broad; ventrals: ♂ 125-146, ♀ 135-147
Snout produced into an appendage covered with 10 to 20 scales; rostral not reaching the summit of the rostral appendage; ventrals: ♂ 133-161, ♀ 135-163
28 γ. Snout not turned up at the end; supraocular narrow or broken up into several small shields; upper surface of head with small, usually keeled scales; two or three series of scales between the eye and the upper labials; scales in 23 to 27 rows, usually 25; ventrals: ♂ 151-177, ♀ 153-180
b. A pit between the nostril and the eye; upper surface of head with 9 large shields; nasal in contact with the rostral; third upper labial entering the eye; scales in 23 rows; ventrals 149-174; subcaudals 31-44
In dealing with the coloration, we have first to distinguish between the colour and the markings. The former is very often highly variable among snakes of the same species, to say nothing of the changes which may take place with age or with the condition of the individuals, whether before or after exuviation; it is not unusual to find among specimens from the same locality a great range of variation, from greyish-white to brown, or red, or black, as, for instance, in our Common Viper. The latter afford more important characters, and often furnish valuable indications for the distinction of species; but even the disposition of the markings is subject to great individual variations, more likely to mislead than to help the inexperienced student in the discrimination of species. It is therefore always advisable to resort in the first instance to structural characters for the purpose of specific identification, and to fall back on coloration only as a means of confirmation. If we were to be guided by colour and markings alone, how could we believe that an adult four-lined Coluber quatuorlineatus is of the 30 same species as the handsomely spotted Coluber sauromates; and yet, if we compare the young of these two snakes we find them to be absolutely identical in their markings, and, in the absence of any structural differences, we are forced to conclude that they only represent two forms of the same species, of which the latter is the more primitive.
It is nevertheless a fact that, with a few exceptions, the markings, however variable they may be, are reducible to certain fundamental patterns to which the innumerable variations may be traced back, and their derivation followed and scientifically explained. Let us consider, for instance, another species of Coluber, highly variable in its markings: C. leopardinus, of which the typical form, so called from having been the first described and named, is not by any means to be regarded as the most primitive.
First, we must take for granted that the markings of all such snakes, whether consisting of spots, stripes, or bars, start from a regular arrangement, which may be theoretically represented by four paired longitudinal series on the head and body: (1) Dorsal series (D); (2) Dorso-lateral (DL); (3) Lateral (L); (4) Ventro-lateral (VL). The first starts from the middle line of the head, and is continued along the spine; the second occupies the space between the first and third, which originates at the tip of the snout, passes through the eye, and 31 is continued on the temple and along the side of the body; the fourth follows the lower lip, and extends along each side of the belly. Bearing this in mind, we find that the variety of C. leopardinus named schwoederi, with a vertebral series of paired spots, is to be regarded as the most primitive, from which we can derive, on the one hand, the true leopardinus by imagining a transverse fusion of the spots of series D into a single row, some of the spots often actually revealing, in their biscuit shape, their dual origin; whilst, on the other hand, confluence of the paired spots of the same series into two longitudinal stripes produces the variety named quadrilineatus (see Plate VII.). In this particular instance, the paired series D has fused into a single streak on the head, and the series L appears to have departed from its primitive course to extend on the upper surface of the head, both in front of and behind the eye.
Many snakes show an interocular band extending from lip to lip, through the eyes, across the interorbital region. In others the lateral stripe L may bifurcate in front of the eye, an upper branch extending across the snout, through transverse fusion of series D and DL, and it may also bifurcate in like manner on the temporal region, fusing with the corresponding marking on the other side to form a W-shaped figure. The pattern of markings on the upper surface of the head is, however, often very complicated, and hence difficult of explanation.
32 As a second example of the derivation of patterns, we may mention Vipera aspis, which varies enormously as to its mid-dorsal markings, forming, in different individuals or even on different parts of the body, single or paired spots, a zigzag band, or transverse bars; all these are derived from the paired spots of series D. Each pair of spots may fuse and form transversely oval or elliptical spots or bars, or the spots may assume an alternate disposition from which, through confluence, the zigzag or sinuous band results. Thus, spotted and striped patterns may be traced to a common origin, however fundamental the difference between them appears at first sight. If the elements of the four series, D, DL, L, and VL, unite transversely with each other, and also with the spots on the ventral surface, we obtain ringed forms such as the Coral-snakes. That the black nuchal collar of our common Grass-snake is actually formed by the fusion of the spots of three originally distinct series has been proved by tracing the development of the markings in the embryo.
In various species a pair of light streaks extends along the back, bordering the D area, without interfering with the other markings, as we see, among European snakes, in some specimens of Tropidonotus natrix and viperinus, and Vipera berus.
Although it sometimes happens that a definite system of markings prevails throughout a genus, 33 such as the annulate form in the South American Elaps, this is far from being universally the case; many closely allied species, or individuals of the same species, may be distinguished by very different patterns. Even on the same individual we may find two opposite types of markings without any transition, as in two Central American species of widely different genera, Polyodontophis annulatus and Zamenis mexicanus, in which the anterior part of the body is annulate or barred, and the rest longitudinally striped.
It is also a remarkable fact that very often the two sides of the body are not alike in their markings, appearing as if formed of the union, on the median line, of the right and left halves of two individuals. Thus it may happen, in annulate forms, that some of the annuli are broken exactly in the mid-dorsal and mid-ventral lines, and that the halves do not correspond in number on the two sides. In the handsome South American Lachesis alternatus, which derives its specific name from the two series of large C-shaped, dark, light-edged markings which adorn its back, these markings are not always alternating, as is the rule; but some may lie opposite to each other and back to back, this being due to the fact that the numbers of the markings do not correspond on the two sides. In one specimen I count twenty-four of these markings on the left side, and twenty-seven on the right. This shows that 34 great importance cannot be attached to the number of the markings, for systematic purposes. In fact, in some Coral-snakes, Elaps fulvius for instance, the number of annuli may vary from twelve to fifty-two, with every gradation between the extremes. The bilateral asymmetry to which we have alluded produces the chess-board arrangement of the ventral spots in many snakes.
Among the markings which call for investigation as to their meaning, we must allude to the presence, in some Colubrids, of a small, light, dark-edged spot, or of a pair of light dots close together, in the middle of the parietal shields or on each side of the suture between these shields, which correspond in their position to the parietal organ of many Lizards. May not this marking be in some way correlated with sensory organs, like the apical pits on the scales of the body? And what is the explanation of such bizarre signs as the spectacle or the eye-spot on the hood of the Indian Cobra? At present it is as inexplicable as the lugubrious emblem on the thorax of the Death’s-head Moth. It cannot be suggested that it is a warning mark intended to terrify intruders, for when the Cobra is at rest the hood is folded, and the characteristic marking is not displayed; whilst as soon as it is aroused, and the hood expanded, it faces its enemy in such a way that the spectacle, or ocellus, is not to be seen.
First among the most brilliantly coloured snakes, 35 of which there are many, stand the Coral-snakes, Elaps, of America, mostly annulate with red, yellow or white, and black. This striking coloration obtains also in diverse harmless snakes inhabiting the same part of the world, and this coincidence has been adduced in favour of the theory of mimicry, correlated with that of natural selection, which accounts for the resemblance as being of advantage to a harmless species, which is thus mistaken for one notorious for its deadly poison, and advertised as such by its brilliant colours (warning coloration). But other poisonous and much more dangerous snakes are not, as a rule, endowed with brilliant colours. It is true that these also may have their mimics: the Krait, Bungarus cæruleus, and Lycodon aulicus, in India, the Pit-viper, Ancistrodon himalayanus, and Psammodynastes pulverulentus, in the Himalayas and Assam, are good examples of such cases. On the other hand, there are equally striking instances of what one would regard as mimics if they only occurred together; thus, there is no better case of general resemblance between a poisonous and a harmless snake than we find in the Indian Cobra and the Coluber corais of tropical America, where Cobras are absent, or between a Viper and the Boid Enygrus asper, from New Guinea, where no Vipers exist.
Without attempting to offer any suggestion to account for the similarity of markings which prevails 36 in certain parts of the world, attention may be drawn to the predominance of longitudinal dark and light stripes in the Indo-Malayan representatives of the American Elaps, shared by many innocuous snakes of similar form inhabiting the same region, and to the striped tails common to various Colubrids of Madagascar, as if the snakes of a district had agreed to conform to certain fashions in dress.
It is further noteworthy, in relation to the theory of warning coloration, that many Uropeltids, innocent burrowing creatures living underground or concealed under stones or rotting tree-trunks in the forests of Southern India and Ceylon, hardly ever showing themselves in daylight, are among the most striking for their bright yellow or red and black markings. We may point out at the same time the very marked resemblance in form and coloration between the Uropeltid Melanophidium bilineatum, and the Apodal Batrachian Ichthyophis glutinosus, both occurring together in Southern India.
The colour of snakes often harmonizes with their surroundings. Thus, many Tree-snakes, Boid, Colubrid, or Viperid, are of a bright green, like the foliage in which they are concealed. On the other hand, other Tree-snakes are not green, or only some specimens are green, as in the genera Dendraspis and Dispholidus. Desert-snakes are of the yellowish or reddish colour of the sand or rock on which they live, and in species whose range extends over different 37 districts the desert individuals are paler, without or with less distinct markings, as compared to their fellows among other surroundings. In addition to their markings, some snakes are adorned with a metallic iridescent gloss, due to a fine striation of the scales.
The iris is often metallic, gold, bronze, or copper-red, and the black streaks of the head sometimes extend over it.
Although, unlike many lizards, snakes are unable to rapidly alter their colours, some produce a semblance of this phenomenon when inflating their neck or body; this is due to the presence of dark and light markings or of a bright pigment in the interstitial skin, which is not seen when the scales overlap. Thus, in the Indian Tree-snake Dryophis mycterizans the skin between the green or brown scales in the anterior part of the body is black and white, producing a striped pattern when the neck is inflated; the skin of the same region is bright vermilion in the Malay Tropidonotus subminiatus; many more examples could be quoted. The spectacle marking on the hood of the Indian Cobra involves the scales as well as the interstitial skin.
As a rule there are no sexual differences in colour. Yet these are so marked in our Common Adder that the sex of a specimen can nearly always be recognized by the coloration. This is, however, the 38 exception, even in the genus to which the Adder belongs. A nuptial dress is unknown in snakes.
A special livery for the young is rather exceptional, but very often the new-born is more vividly coloured than its parents, and in many black varieties the young is similar to the typical form. Some green Tree-Boids (Chondropython and Corallus caninus) are not green, but yellowish, cream-colour, or pinkish, when young, the green appearing around the white spots, which are the remains of the ground colour, and gradually spreading over the whole body. Conversely, the young of a variety of the Pit-viper Lachesis wagleri, common in the Malay Peninsula, is green, and the adult black and yellow. In the young of Grayia ornata, a West African Water-snake, the markings of the young are to those of the adult like positive and negative in photography, the white bars, forked on the sides, which extend across the black back of the former being gradually transformed into black bars on a light ground in the latter; in such a case it is impossible to decide whether the dark or the light parts are to be considered as the ground colour.
That the skin of many snakes contains soluble colouring matter of a special kind is well known, green snakes, such as Dryophis prasinus and Lachesis gramineus staining the spirit in which they are preserved. Chemists have not yet paid attention to this question, which requires investigation.
39 Melanism is frequent in snakes, and sometimes affects all individuals in the same locality. It seems undesirable to bestow varietal names on such aberrations, as is so frequently done by systematists, any more than we should in the case of albinos. Melanism may be produced in two ways: by an extension of the black markings, which invade the whole surface, as in the males of Vipera berus; or by a general darkening of the ground colour and of the markings, as in the females of the same species. In the latter case, the markings reappear under certain lights or after a prolonged sojourn in spirits. Sometimes, as in Zamenis gemonensis, the uniform black colour appears only as the snake approaches the adult condition, the young having the normal livery.
Partial albinism is rare; perfect albinism, characterized by absence of black pigment in the eye, rarer still. Cases have been observed, among European species, in Tropidonotus natrix and tessellatus, in Coluber longissimus, and in Coronella austriaca.
The typical Ophidian skull is characterized by a solidly ossified brain-case, with the distinct frontals and the united parietals extending downwards to the basisphenoid, which is large and produced forward into a rostrum extending to the ethmoidal region. The nasal region is less completely ossified, and the paired nasals are often attached only at their base. The occipital condyle is either trilobate and formed by the basioccipital and the exoccipitals, or a simple knob formed by the basioccipital; the supraoccipital is excluded from the foramen magnum. The basioccipital may bear a strong, curved ventral process or hypapophysis (in the Vipers).
The prefrontal is situated, on each side, between the frontal and the maxillary, and may or may not be in contact with the nasal; the postfrontal, usually present, borders the orbit behind, rarely also above, and in the Pythons a supraorbital is intercalated between it and the prefrontal.
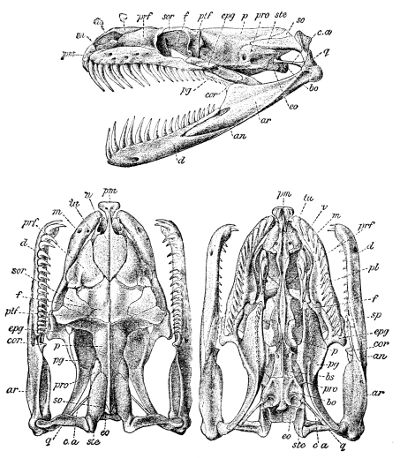
Fig. 3—Skull of Python amethystinus. (From British Museum Catalogue of Snakes)
an, Angular; ar, articular; bo, basioccipital; bs, basisphenoid; cor, coronoid; c.a, columella auris (stapes); d, dentary; eo, exoccipital; epg, ectopterygoid (transverse); f, frontal; m, maxillary; n, nasal; p, parietal; pl, palatine; pm, premaxillary; prf, prefrontal; pro, proötic; pg, pterygoid; ptf, postfrontal; q, quadrate; so, supraoccipital; sor, supraorbital; sp, splenial; ste, supratemporal; tu, turbinal; v, vomer.
The premaxillary is single and small, and as a rule connected with the maxillary only by ligament. The paired vomer is narrow. The palatine and 42 pterygoid are elongate and parallel to the axis of the skull, the latter diverging behind and extending to the quadrate or to the articular extremity of the mandible; the pterygoid is connected with the maxillary by the ectopterygoid or transverse bone, which may be very elongate, and the maxillary often emits a process towards the palatine, the latter bone being usually produced inwards and upwards towards the anterior extremity of the basisphenoid. The quadrate is usually large and elongate, and attached to the cranium through the supratemporal (often regarded as the squamosal). In rare cases (Miodon, Polemon) the transverse bone is forked, and articulates with two branches of the maxilla. The quadrate and the maxillary and palatopterygoid arches are more or less movable to allow for the distension required by the passage of prey, often much exceeding the calibre of the mouth. For the same reason, the rami of the lower jaw, which consist of dentary, splenial, angular, and articular elements, with the addition of a coronoid in the Boidæ and a few other small families, are connected at the symphysis by a very extensible elastic ligament.
The hyoid apparatus is reduced to a pair of cartilaginous filaments situated below the trachea, and united in front.
There are various modifications according to the genera. A large vacuity may be present between the frontal bones and the basisphenoid (Psammophis, 43 Cœlopeltis); the maxillary may be much abbreviated and movable vertically, as in the Viperidæ; the pterygoids may taper and converge posteriorly, without any connexion with the quadrate, as in the Amblycephalidæ; the supratemporal may be much reduced, and wedged in between the adjacent bones of the cranium; the quadrate may be short or extremely large; the prefrontals may join in a median suture in front of the frontals; the dentary may be freely movable, and detached from the articular posteriorly.
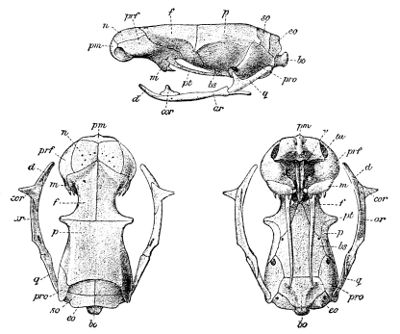
Fig. 4—Skull of Typhlops lumbricalis. (From British Museum Catalogue of Snakes)
Lettering of the bones as in Fig. 3
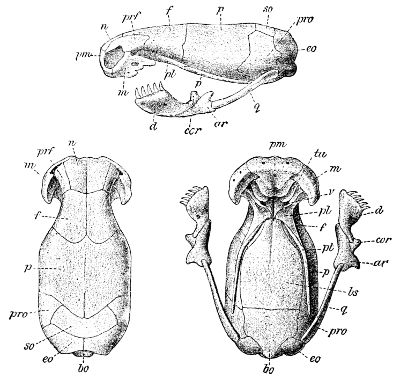
Fig. 5—Skull of Glauconia macrolepis. (From British Museum Catalogue of Snakes)
Lettering of the bones as in Fig. 3
The deviation from the normal type is much greater still when we consider the degraded, worm-like members of the families Typhlopidæ (Fig. 4, p. 43) and Glauconiidæ (Fig. 5), in which the skull is very compact and the maxillary much reduced. In the former this bone is loosely attached to the lower aspect of the cranium; in the latter it borders the mouth, and is suturally joined to the premaxillary and the prefrontal. In both the tranverse bone and the supratemporal are absent, but the coronoid element is present in the mandible.
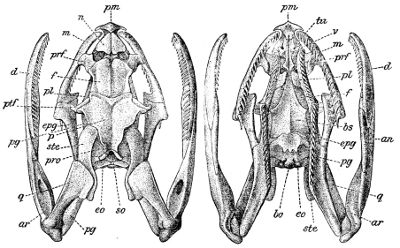
Fig. 6—Skull of Tropidonotus natrix. (From British Museum Catalogue of Snakes)
Lettering of the bones as in Fig. 3
The principal modifications of the skull in the European genera may be contrasted as in the following synopsis:
I. Quadrate articulating with the cranium, supratemporal absent; mandible much shorter than the skull, with coronoid bone; maxillary small, on lower aspect of cranium; pterygoids not extending to quadrate; nasals forming long sutures with the premaxillary, prefrontals, and frontal
II. Quadrate suspended from the supratemporal; mandible at least as long as the skull; pterygoids extending to quadrate or mandible.
46 A. Mandible with coronoid bone; nasals in sutural contact with frontals and prefrontals; transverse bone short, not projecting much beyond cranium; maxillary not half as long as mandible, which is not longer than skull (to occiput)
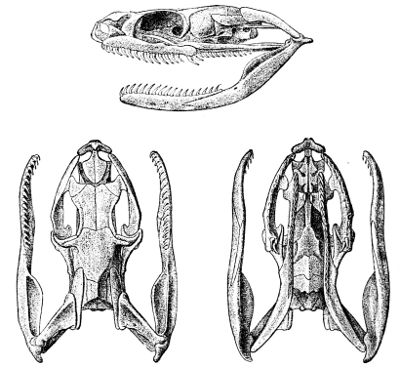
Fig. 7—Skull of Zamenis gemonensis. (From British Museum Catalogue of Snakes)
B. No coronoid bone; nasals isolated.
1. Maxillary elongate, not movable vertically.
a. Maxillary half as long as mandible.
Supratemporal half as long as skull, projecting far 47 beyond cranium; mandible much longer than skull
Supratemporal not half as long as skull, projecting far beyond cranium; mandible much longer than skull
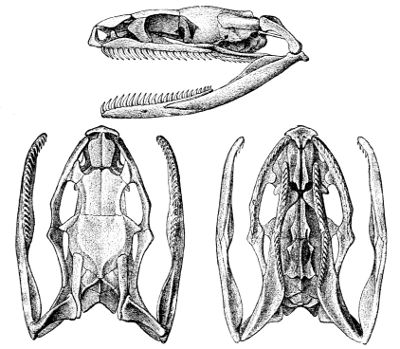
Fig. 8—Skull of Coluber longissimus. (From British Museum Catalogue of Snakes)
Supratemporal not half as long as skull, projecting but slightly beyond cranium; mandible much longer than skull
Supratemporal not half as long as skull, not projecting beyond cranium; mandible not longer than skull
48 b. Maxillary not half as long as mandible, which is longer than skull; supratemporal not half as long as skull, projecting beyond cranium.
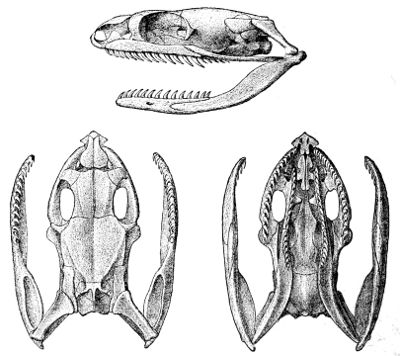
Fig. 9—Skull of Coronella austriaca. (From British Museum Catalogue of Snakes)
Quadrate longer than supratemporal; maxillary much longer than quadrate, nearly straight in front of prefrontal; a large vacuity between the frontal bones and the basisphenoid
Quadrate not longer than supratemporal; maxillary little longer than quadrate, strongly curved in front of prefrontal
49 Quadrate longer than supratemporal; maxillary little longer than quadrate, nearly straight in front of prefrontal
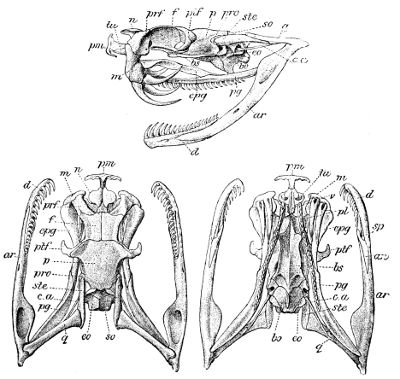
Fig. 10—Skull of Vipera lebetina. (From British Museum Catalogue of Snakes)
Lettering of the bones as in Fig. 3
2. Maxillary much abbreviated and erectile; supratemporal not half as long as skull; mandible much longer than skull; basioccipital with a strong process.
50 Maxillary bone solid
Maxillary bone hollowed out
The vertebræ number 130 to 500—in the European forms 147 (Vipera ursinii) to 330 (Coluber leopardinus).
The vertebral column consists of an atlas (composed of two vertebræ) without ribs; numerous precaudal vertebræ, all of which, except the first or first three, bear long, movable, curved ribs with a small posterior tubercle at the base, the last of these ribs sometimes forked; two to ten so-called “lumbar vertebræ” without ribs, but with bifurcate transverse processes (lymphapophyses) enclosing the lymphatic vessels; and a number of ribless caudal vertebræ with simple transverse processes. When bifid, the ribs or transverse processes have the branches regularly superposed.
The centra have the usual cup-and-ball articulation, with the nearly hemispherical or transversely elliptic condyle at the back (procœlous vertebræ), whilst the neural arch is provided with additional articular surfaces in the form of pre- and post-zygapophyses, broad, flattened, and overlapping, and of a pair of anterior wedge-shaped processes called zygosphene, fitting into a pair of corresponding concavities, zygantrum, just below the base of the neural spine. Thus the vertebræ of snakes articulate with each other by eight joints in addition to the cup-and-ball on the centrum, and interlock by parts reciprocally receiving and entering one another, like the joints called “tenon-and-mortice” 51 in carpentry. The precaudal vertebræ have a more or less high neural spine which, as a rare exception (Xenopholis), may be expanded and plate-like above, and short or moderately long transverse processes to which the ribs are attached by a single facet. The centra of the anterior vertebræ emit more or less developed descending processes, or hæmapophyses, which are sometimes continued throughout (Fig. 11, A), as in Tropidonotus, Vipera, and Ancistrodon, among European genera.
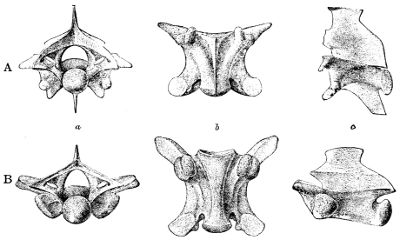
Fig. 11—Posterior Precaudal Vertebræ of Lioheterodon (A) AND Heterodon (B). (From British Museum Catalogue of Snakes)
a, Back view; b, lower view; c, side view.
In the caudal region, elongate transverse processes take the place of ribs, and the hæmapophyses are paired, one on each side of the hæmal canal. In the Rattlesnakes the seven or eight last vertebræ are enlarged and fused into one.
52 No snake shows any rudiments of the pectoral arch, but remains of the pelvic are found in the Typhlopidæ, the Glauconiidæ, the Boidæ, and the Ilysiidæ. In the first these vestiges are reduced to a single bone (ilium?) on each side; in the second they consist of ilium, pubis, and ischium, the latter forming a ventral symphysis, and a rudimentary femur; whilst in the third there is a long ilium, attached to the lower branch of the first bifurcate transverse process of the lumbar vertebræ, bearing three short bones, the longest of which, regarded as the femur, terminates in a claw-like spur which, in males at least, usually appears externally on each side of the vent.
In the most generalized snakes—those which show the nearest approach to lizards—teeth are present not only on the rami of both jaws, but also on the premaxillary bone, on the palatines, and on the pterygoids. A reduction of the dentition takes place in various genera, in which the teeth of either the upper or the lower jaw, and of the palatines or pterygoids, or both, may be absent, and the premaxillary is devoid of teeth in the great majority, including all European representatives, of the Ophidia.
In the egg-eating snakes of the genera Dasypeltis and Elachistodon the dentition is very much reduced, in accordance with the peculiar régime, and this deficiency is compensated by the development on some of the anterior thoracic vertebræ of long, tooth-like processes (hypapophyses) directed forwards, and capped with a remarkably dense, vitreous tissue simulating enamel, the function of these tooth-like processes being to break the shell of the egg within the gullet, where none of its contents are lost, the shell being afterwards rejected through the mouth in the form of a pellet.
54 With the exception of the worm-like Typhlopidæ, which are provided with a few teeth in the upper jaw only, European snakes have teeth on the maxillary, palatine, pterygoid, and dentary bones. Unless the maxillary be strongly abbreviated and modified in connexion with the poison apparatus, as in the Viperidæ, the teeth in the jaws as well as on the palate form single longitudinal series; they are elongate, conical, with or without a sharp posterior edge, more or less recurved, acutely pointed, sometimes needle-like, and directed backwards, as behoves their function, which, in addition to attack and defence, is to prevent the retrogression of the prey in the act of prehension and deglutition. A notable exception occurs in the genus Iguanognathus, from Sumatra, all the teeth having spatulate crowns ribbed along the outer side. Unfortunately, nothing is known as to the food of this remarkable snake. The teeth are coated with a thin layer of enamel. It was held, for a time, that the glossy outer coating was only due to a denser structure of the dentine. As in all living Reptiles with the exception of the Crocodiles, the teeth are not implanted in true sockets, but simply ankylosed to the bone on which, when detached, their slightly enlarged base, or rather the bony tissue on which it rests, leaves a shallow impression, or pseudo-socket. In the process of biting or feeding, some of the teeth are frequently lost, and are readily replaced by others lying in reserve in the 55 gum at the inner side, and becoming fixed to the bone soon after a vacancy occurs. Such replacement teeth, of different grades of development, form several series, so that in a snake like our common Tropidonotus the mouth may contain four times as many teeth as are functional, without reckoning different earlier stages of tooth germs which escape ordinary observation, being placed vertically one above the other.
Three types of teeth, connected by every intermediate step, are distinguished: the solid, the grooved, and the canaliculated or tubular, so-called “perforated”; the third, as we shall explain, being only a further modification of the second. In the grooved tooth, a sulcus runs along the anterior or outer surface, its object being to convey into the wound the secretion of a poison gland. It varies in depth according to the species, and may be so slight as to escape detection without a very strong magnifying glass. In some the sulcus may be very deep and wide, forming a canal round which the tooth folds to the extent of its borders nearly meeting; from this condition the so-called “perforated” fang is derived through the complete fusion of the borders of the tooth, and the obliteration of the line of union except at each extremity. The structure of such a fang may be best understood by imagining a tooth, lined all round with the same layer of dentine and enamel, being flattened out in a vertical plane and then folded over, the outer edges coalescing on the front 56 median line in such a way that the inner wall of the tooth is in reality the anterior surface, and the outer wall the posterior surface, of the ordinary tooth.
Grooved teeth, with open canal, are situated either at the anterior extremity (Proteroglyphs) or at the posterior extremity (Opisthoglyphs) of the maxillary bone, usually followed or preceded by a series of solid teeth, which in some cases may likewise show a more or less distinct groove. Such may also be present on the teeth of the lower jaw, as in the European Cœlopeltis, in some specimens of which a faint groove is visible on the outer side with the aid of a strong lens.
The tubular fangs of the Viperidæ are inserted on the posterior extremity of the much abbreviated and erectile maxillary bone, which bears no other teeth. The Proteroglyphs (Cobras, Coral-snakes, Sea-snakes) and the Solenoglyphs (Vipers, Pit-vipers, Rattlesnakes) may be regarded as the diverging extremes in the development of the poison apparatus, both culminating in forms with tubular fangs, the former as derived directly from the Aglyphs (harmless snakes), the latter from the Opisthoglyphs, likewise evolved out of the Aglyphs. That the insertion of the poison fangs of the Viperidæ is really on the posterior extremity of the maxillary bone is evident from the condition of the bone in its recumbent position, especially in the African Viper, Causus, which in 57 several respects departs less markedly from the Colubrid type than our European Vipers.
The poison fangs of the Viperidæ appear to be movable, folding in the mouth when at rest, and erected, or even thrust forward, when ready to act. This, however, is simply due to the mobility of the maxillary bone, to which they are ankylosed as in all other snakes. There are normally two equally-developed fangs, close together and side by side, to each maxillary, followed by several replacement fangs loosely attached behind them, usually in two series of four. When the two fangs are in situ, they of course both function in the act of biting, although only one is in relation with the single poison duct; often, however, there is only one fang in position, either the right or the left, the place of the other being indicated by a shallow socket which will soon be filled by one of the posterior reserve fangs moving forward and becoming ankylosed to the bone. Snake-charmers who extract the poison fangs of the snakes they use for their performances have therefore to renew the operation frequently, unless they amputate the bone on which the fangs are inserted, an injury which the creature does not long survive.
The dentition of the snakes in which the maxillary bone is not movable vertically falls under three divisions: the Aglyphs, in which the teeth are all solid; the Opisthoglyphs, in which one or more (usually two) of the hindermost teeth are provided with a 58 groove; and the Proteroglyphs, in which grooved or canaliculated teeth are situated in front, followed or not by solid teeth. Beyond these three principal divisions, the dentition furnishes important characters for the classification, although that importance has sometimes been over-estimated. The maxillary teeth may be equal in length (Isodonts), or the anterior the longer (Lycodonts), or the posterior the longer, increasing gradually in size (Coryphodonts) or abruptly, without (Syncranterians) or with a diastema, or break, in front of them (Diacranterians). These categories are, however, so completely connected as to preclude their use in taxonomy beyond helping to define genera. The number of maxillary teeth and the relative proportions and disposition of the mandibular teeth also afford useful generic characters.
The European genera may be arranged as follows, according to the dentition:
I. Teeth few, disposed in a transverse series in the upper jaw only
II. Teeth in both jaws and on the palatines and pterygoids.
A. A series of solid teeth along the maxillary; no grooved teeth.
1. Anterior maxillary and mandibular teeth longest; 9 or 10 maxillary teeth
2. Maxillary teeth equal, or increasing in size posteriorly.
59 a. Mandibular teeth 17 to 30; maxillary teeth 15 to 22.
Posterior maxillary teeth longest; mandibular teeth subequal, more than 20
Posterior maxillary teeth longest; mandibular teeth not more than 20, posterior smallest
Maxillary teeth subequal; mandibular teeth 20 to 25, posterior smallest
b. Mandibular teeth 14 or 15, subequal; maxillary teeth 12 to 15.
Maxillary teeth increasing in size
Maxillary teeth subequal
B. One or two enlarged grooved fangs behind the series of solid maxillary teeth.
14 to 17 subequal solid maxillary teeth, forming a continuous series; 21 to 23 mandibular teeth, anterior strongly enlarged
9 to 11 solid maxillary teeth, fourth and fifth or fifth and sixth enlarged, followed by an interspace; sixth mandibular tooth fang-like, followed by an interspace
9 or 10 solid maxillary teeth, forming a continuous series, decreasing in length posteriorly; anterior mandibular teeth strongly enlarged
C. Maxillary with only two large canaliculated fangs side by side, one of which may be missing; anterior mandibular teeth longest
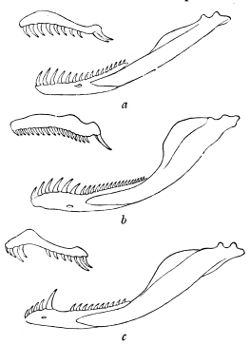
Fig. 12—Maxillary and Mandible of—(a) Tarbophis fallax; (b) Cœlopeltis monspessulana; (c) Macroprotodon cucullatus. (From British Museum Catalogue of Snakes)
In counting the teeth for the purpose of using this key, care must be taken to ascertain the full number, as it frequently happens that one or more are missing; but their place is indicated by the shallow pits in which their base was implanted, the overlooking of which might convey the impression of a hiatus such as is characteristic of certain genera—Macroprotodon, for instance. Needless to say, the loose teeth which are in reserve on the inner side of the jaws or behind the tubular fangs are not taken into consideration, the numbers given being those of 61 functional teeth only. Although as a rule the teeth can be counted easily, on a specimen preserved in spirit, by simply pushing aside the lips and gums with the finger, it is sometimes necessary to remove and clean the bones of the jaws, an operation which does not require much skill.
The gland which secretes the poison is a modification of the parotid salivary gland of other Vertebrates, and is usually situated on each side of the head below and behind the eye, invested in a muscular sheath. It is provided with large alveoli in which the venom is stored before being conveyed by a duct to the base of the channelled or tubular fang through which it is ejected.
In the Vipers, which furnish examples of the most highly developed poison apparatus, although inferior to some in its toxic effects, the poison gland is very large and in intimate relation with the masseter or temporal muscle, consisting of two bands, the superior arising from behind the eye, the inferior extending from the gland to the mandible. When the snake bites, the jaws close up, causing the gland to be powerfully wrung, and the poison pressed out into the duct. From the anterior extremity of the gland the duct passes, below the eye and above the maxillary bone, where it makes a bend, to the basal orifice of the poison fang, described above (p. 55), 63 which is ensheathed in a thick fold of mucous membrane, the vagina dentis. By means of the movable maxillary bone (supra, p. 49) hinged to the prefrontal, and connected with the tranverse bone which is pushed forward by muscles set in action by the opening of the mouth, the tubular fang is erected and the poison discharged through the distal orifice in which it terminates.
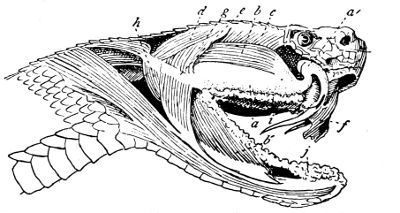
Fig. 13—Poison Apparatus of Rattlesnake: Venom Gland and Muscles (Lateral View). (After Duvernoy)
a, Venom gland; a´, venom duct; b, anterior temporal muscle; b´, mandibular portion of same; c, posterior temporal muscle; d, digastricus muscle; e, posterior ligament of gland; f, sheath of fang; g, middle temporal muscle; h, external pterygoid muscle; i, maxillary salivary gland; j, mandibulary salivary gland.
In some of the Proteroglyphous Colubrids, as we have seen, the poison fangs are not tubular, but only channelled and open along the anterior surface; and as the maxillary bone in these snakes is more or less elongate, and not or but slightly movable vertically, 64 the poison duct runs above the latter, making a bend only at its anterior extremity, and the tranverse bone has not the same action on the erection of the fangs. Otherwise the mechanism is the same.
In the Opisthoglyphous Colubrids, with grooved teeth situated at the posterior extremity of the maxilla, a small posterior portion of the upper labial or salivary gland is converted into a poison-secreting organ, distinguished by a light yellow colour, provided with a duct larger than any of those of the labial gland, and proceeding inward and downward to the base of the grooved fang; the duct is not in direct connexion with the groove, but the two communicate through the mediation of the cavity enclosed by the folds of mucous membrane surrounding the tooth, and united in front.
The reserve or successional teeth, which are always present just behind or on the side of the functional fang of all venomous snakes, are in no way connected with the duct until called upon to replace a fang that has been lost. It could not be otherwise, since the duct would require a new terminal portion for each new fang; and as the replacement takes place alternately from two parallel series, the new poison-conveying tooth does not occupy exactly the same position as its predecessor.
Two genera, Doliophis among the Elapine Colubrids, and Causus among the Viperids, are highly remarkable for having the poison gland and its duct 65 of a great length, extending along each side of the body and terminating in front of the heart. Instead of the muscles of the temporal region serving to press out the poison into the duct, this action is performed by those of the side of the body.
When biting, a Viperid snake merely strikes, discharging the venom the moment the fangs penetrate the skin, and then immediately leaves go. A Proteroglyph or Opisthoglyph, on the contrary, closes its jaws like a dog on the part bitten, often holding on firmly for a considerable time.
The poison, which is mostly a clear limpid fluid of a pale straw or amber colour, more rarely greenish, sometimes with a certain amount of suspended matter, is exhausted after several bites, and the glands have to recuperate.
It must be added that the poison can be ejected otherwise than by a bite, as in the so-called Spitting Snakes of the genera Naia and Sepedon. The fact that some of these deadly snakes when irritated are in the habit of shooting poison from the mouth, at a distance of 4 to 8 feet, even apparently aiming at a man’s face, has been too often witnessed in India and Malaya, and especially in Africa, from the days of the ancient Egyptians, for any doubt to subsist as to their being endowed with this faculty, but the mechanism by which this action is produced has not been satisfactorily explained. In all probability, the poison escapes from the sheath of 66 mucous membrane surrounding the base of the fangs, and is mixed with ordinary saliva, the membranes of the mouth perhaps acting as lips, in which case the term “spitting” would not be incorrect. The spitting, which may take place three or four times in succession, has been observed to be preceded by some chewing movements of the jaws. If reaching the eye, the poisonous fluid causes severe inflammation of the cornea and conjunctiva, but no more serious results if washed away at once.
Snake poisons is a subject which has always attracted much attention, and which has made great progress within the last quarter of a century, especially as regards the defensive reaction by which the blood may be rendered proof against their effect by processes similar to vaccination—antipoisonous serotherapy. The studies to which we allude have not only conduced to a method of treatment against snake-bites, but have thrown a new light on the great problem of immunity. They have shown that the antitoxic serums do not act as chemical antidotes in destroying the venom, but as physiological antidotes; that, in addition to the poison glands, snakes possess other glands supplying their blood with substances antagonistic to the poison, such as also exist in various animals refractory to snake poison, the hedgehog and the mungoose for instance. Unfortunately, the specificity of the different snake poisons is such that, even when the physiological 67 action appears identical, serum injections or graduated direct inoculations confer immunity towards one species or a few allied species only. Thus, a European in Australia who had become immune to the poison of the deadly Notechis scutatus, manipulating these snakes with impunity, and was under the impression that his immunity extended also to other species, when bitten by a Denisonia superba, an allied Elapine, died the following day. In India, the serum prepared with the venom of Naia tripudians has been found to be without effect on the poison of Naia bungarus, the two species of Bungarus, and the Vipers Vipera russelli, Echis carinatus, and Lachesis gramineus. Vipera russelli serum is without effect on Colubrine venoms, and on those of Echis and Lachesis. In Brazil, serum prepared with the venom of Lachesis lanceolatus has proved to be without action on Crotalus poison. These examples, and others which could be given, show that the hopes which were at first entertained as to the benefits to be conferred on mankind by the serum treatment were somewhat over-sanguine—at least as regards countries like India, where, different kinds of poisonous snakes occurring together, it is sometimes impossible to know by which the bite has been inflicted.
Chemistry teaches that snake venoms consist for the most part of solutions of modified proteids, and all attempts to separate the toxic principles from 68 such proteids have hitherto been unsuccessful. Accordingly, at the present time we must regard such toxic principles as residing in some special grouping of a portion of the atoms in the complex venom proteid molecule. The analysis of their physiological actions has proved them to be made up of a great many more constituents than would be imagined from their chemical composition.
The effect of the poison of Proteroglyphous Colubrids (Hydrophids, Cobras, Bungarus, Elaps, Pseudechis, Notechis, Acanthophis) is mainly on the nervous system, respiratory paralysis being quickly produced by bringing the poison into contact with the central nervous mechanism which controls respiration; the pain and local swelling which follow a bite are not usually severe.
Viper poison (Vipera, Echis, Lachesis, Crotalus) acts more on the vascular system, bringing about coagulation of the blood and clotting of the pulmonary arteries; its action on the nervous system is not great, no individual group of nerve cells appears to be picked out, and the effect upon respiration is not so direct; the influence upon the circulation explains the great depression which is a symptom of Viperine poisoning. The pain of the wound is severe, and is speedily followed by swelling and discoloration. The symptoms produced by the bite of the European Vipers are thus described by the best authorities on snake poison (Martin and Lamb): 69 The bite is immediately followed by local pain of a burning character; the limb soon swells and becomes discoloured, and within one to three hours great prostration, accompanied by vomiting, and often diarrhœa, sets in. Cold, clammy perspiration is usual. The pulse becomes extremely feeble, and slight dyspnœa and restlessness may be seen. In severe cases, which occur mostly in children, the pulse may become imperceptible and the extremities cold; the patient may pass into coma. In from twelve to twenty-four hours these severe constitutional symptoms usually pass off; but in the meantime the swelling and discoloration have spread enormously. The limb becomes phlegmonous, and occasionally suppurates. Within a few days recovery usually occurs somewhat suddenly, but death may result from the severe depression or from the secondary effects of suppuration. That cases of death, in adults as well as in children, are not infrequent in some parts of the Continent is mentioned in the last chapter of this Introduction.
The bite of all the Proteroglyphous Colubrids, even of the smallest and gentlest, such as the Elaps or Coral-snakes, is, so far as known, deadly to man. The Viperidæ differ much among themselves in the toxicity of their venom. Some, such as the Indian Vipera russelli and Echis carinatus, the American Ancistrodon, Crotalus, Lachesis mutus and lanceolatus, the African Causus, Bitis, and Cerastes, cause fatal 70 results unless a remedy be speedily applied. On the other hand, the Indian and Malay Lachesis seldom cause the death of man, their bite in some instances being no worse than the sting of a hornet. The bite of the larger European Vipers may be very dangerous, and followed by fatal results, especially in children, at least in the hotter parts of the Continent; whilst the small Vipera ursinii, which hardly ever bites unless roughly handled, does not seem to be possessed of a very virulent poison, and, although very common in some parts of Austria-Hungary, is not known to have ever caused a serious accident.
It is noteworthy that the size of the poison fangs is in no relation to the virulence of the venom. The comparatively innocent Indo-Malay Lachesis alluded to above have enormous fangs, whilst the smallest fangs are found in the most justly dreaded of all snakes, the Hydrophids.
Little is known of the physiology of the poison of the Opisthoglyphous Colubrids, except that in most cases it approximates to that of the Proteroglyphs. Experiments on Cœlopeltis, Psammophis, Trimerorhinus, Dipsadomorphus, Trimorphodon, Dryophis, Tarbophis, Hypsirhina, and Cerberus, have shown these snakes to be possessed of a specific poison, small mammals, lizards, or fish, being rapidly paralyzed and succumbing in a very short time, whilst others (Eteirodipsas, Ithycyphus) do not seem 71 to be appreciably venomous. Man, it is true, is not easily affected by the bite of these snakes, since, at least in most of those which have a long maxillary bone, the grooved fangs are placed too far back to inflict a wound under ordinary circumstances. There are, however, exceptions. A case was reported a few years ago of a man in South Africa nearly dying as a result of the bite of the Boomslang, Dispholidus typus, the symptoms, carefully recorded, being those characteristic of Viperine poisoning, an important fact to oppose to the conclusions, based on the physiological experiments on Cœlopeltis, which appeared to disprove the theory that the Viperidæ may have been derived from Opisthoglyphous Colubrids.
Experiments made with the secretion of the parotid gland of Tropidonotus and Zamenis have shown that even Aglyphous snakes are not entirely devoid of venom, and point to the conclusion that the physiological difference between so-called harmless and poisonous snakes is only one of degree, just as there are various steps in the transformation of an ordinary parotid gland into a poison gland or of a solid tooth into a tubular fang.
The question whether all snakes are immune to their own poison is not yet definitely settled. Most snakes certainly are, and it is a remarkable fact that certain harmless species, such as the North American Coronella getula and the Brazilian Rhachidelus brazili, 72 are proof against the poison of the Crotalines which frequent the same districts, and which they are able to overpower and feed upon. The Cribo, Spilotes variabilis, is the enemy of the Fer-de-lance in St. Lucia, and it is said that in their encounters the Cribo is invariably the victor. Repeated experiments have shown our Common Snake, Tropidonotus natrix, not to be affected by the bite of Vipera berus and V. aspis, this being due to the presence, in the blood of the harmless snake, of toxic principles secreted by the parotid and labial glands, and analogous to those of the venom of these Vipers.
The Hedgehog, the Mungoose, the Secretary Bird, and a few other birds feeding on snakes, are known to be immune to an ordinary dose of snake poison; whether the pig may be considered so is still uncertain, although it is well known that, owing to its subcutaneous layer of fat, it is often bitten with impunity. The Garden Dormouse (Myoxus quercinus) has recently been added to the list of animals refractory to Viper poison.
The brain is small and of very oblong shape. It consists of smooth cerebral hemispheres, small optic lobes, a still smaller cerebellum, and long olfactory lobes; the pineal body is not accompanied by a parietal organ. The spinal accessory cranial nerve is absent, and the sympathetic system is but feebly developed.
The eyes have been noticed above (p. 12). When normally developed they are susceptible of a slight movement under the transparent disc, quite independent from the cornea, which covers them, and from which they are separated by the so-called “lacrymal chamber.” There are two lacrymal glands, one in front and one behind; the lacrymal duct opens into the posterior nares. A sclerotic bony ring is absent.
The olfactory organ proper is little developed, but is accompanied by an accessory organ, Jacobson’s organ, consisting of a pair of pediculate, cup-shaped sacs, between the nasal sacs and the roof of the mouth, encapsuled by the vomers and the turbinal bones, lined by olfactory epithelium, and opening in the mouth just in front of the choanæ. As this 74 organ, richly provided with nerves, communicates with the inside of the mouth, its function may be to smell the prey as it passes through previous to deglutition. Snakes cannot be credited with a keen sense of smell, although undoubtedly guided by it during the nuptial period.
In the more thoroughly aquatic snakes, the nostril may be closed, when respiration is suspended, by a spongy tissue, which acts as a stopper, and such nostrils are called “valvular,” although a valve is not, in the strict sense, present; when the animal breathes, the nostril is opened by a compression, through special muscles, of the cavernous tissue. In some Sand-snakes the narial opening may be reduced to a crescentic slit.
The sense of hearing is not much developed. Tympanum, tympanic cavity, and Eustachian tubes are absent. In the typical snakes a long columellar rod (the stapes), with a fibrous or cartilaginous pad at the outer end, extends from the fenestra ovalis in the cranium to the quadrate, but in the degraded burrowing forms the stapes is a small bony plate closing the fenestra ovalis.
With one exception (Eryx jaculus, which is said by Schreiber to lap like a lizard), the tongue is not used for drinking or for the prehension or gustation of food, nor for hissing, but is a tactile organ protruded on any object the snake wishes to probe. It is slender and deeply bifid at the end, smooth, very protractile, 75 often quite to the length of the head, and furnished with many sensory corpuscules. It is darted and vibrated on the least excitement, and is usually looked upon by the ignorant as a “sting.” In most snakes it is much pigmented, dark brown or black; in a few it is flesh-coloured or bright red. The tongue is entirely retractile into a sheath below the glottis and opening in front of it; it is always withdrawn into the sheath when the snake bites or feeds.
Other organs, which, in the absence of a satisfactory explanation of their use, have been termed “organs of a sixth sense,” reside in the head-shields and scales of many snakes, and in the deep pits on the sides of the head which are characteristic of various Boidæ and a few Colubridæ.
Scales often show, near their posterior extremity, one or two small light spots or impressions, caused by a thinning of the epidermis, which have been called “apical pits”; they appear to coincide with the terminations of nerve fibres extending along the epidermal folds of the skin. Similar organs sometimes form series on the borders of some of the head-shields, this being particularly noticeable in the Typhlopidæ.
The large and deep pit situated between the nostril and the eye (loreal pit) in the Crotaline Viperidæ—whence the name Pit-vipers, or that of “cuatro naricas” which is bestowed on them by the Spaniards of Mexico—is divided into two chambers: an outer 76 with large external orifice, and an inner, rather more posterior in position and occupying an excavation on the outer face of the maxillary bone. The inner walls of these chambers are very thin and membranous, and form a partition separating the two, except for the presence of a minute opening; this partition is stretched across the hollow of the maxillary bone like the membrane of a drum, and is supplied with blood-vessels and nerves, the latter terminating in cells of variable form. The use of the organ, thus situated at the base of the poison fang, and therefore in close proximity to the sphincter of the poison duct, is still unknown.
Several of the Boidæ, such as Python and Corallus, have deep pits in some of the upper and lower labial shields, or also on each side of the rostral shield; these problematic organs are in all probability also sensory.
In most snakes there is a very marked asymmetry of the viscera and their blood-supply, the organs of the right side being anterior to, as well as larger than, those of the left.
The heart in most cases is situated between the anterior seventh and the anterior fourth of the body; it may be much farther back, beyond the anterior third, in Doliophis, Platurus, and some Viperidæ and Amblycephalidæ, in the middle in Chersydrus. It is of rather elongate form, enclosed in a pericardium in which it lies freely, and has a sinus venosus, two auricles, and a single ventricle divided by a septum. Three arteries leave the ventricle, the pulmonary and two systemic arches. The right systemic arch gives off the carotid artery, which in many snakes, the common Grass-snake for instance, may branch into two, or in others be double from its origin. The anterior abdominal vein is single in most snakes, double in some Boidæ, and conveys blood from the ventral body-wall to the liver. The caudal vein is continued as the renal portal. Veins which have been regarded as remains of the two posterior cardinal 78 of lower Vertebrates have been found in some of the Boidæ.
The bifurcate transverse processes of the vertebræ at the limit between the body and tail enclose the lymph-hearts, which are large and more or less elongate, metamerically divided into several chambers, the right often more developed than the left. The thymus gland lies on each side of the trachea, near the heart, and the thyroid gland is in the middle line, close to the base of the carotid artery.
The trachea is long, and the tracheal rings may be complete in front and incomplete behind, or incomplete throughout. The bronchus opens at once into the more or less elongate, usually single lung, with or without a rudiment of a second, which seems to be constantly the left; in some snakes the lung extends nearly to the cloacal region. In most of the Boidæ there are two well-developed lungs, the left shorter than the right. The lung has highly cellular walls in front, and becomes thin-walled, smooth, or but little vascular, behind, where it may receive its blood from the systemic and not from the pulmonary circulation. In the Typhlopidæ and Viperidæ, as well as in some of the Boidæ, Colubridæ, and Amblycephalidæ, the posterior end or the greater part of the trachea may have its wall enlarged and provided with air cells, resembling the normal lung, with which it is usually continuous; this has been called the “tracheal lung,” but, although serving as an accessory breathing organ, 79 it is not a prolongation of the true lung, nor does it represent the missing left lung, as has been believed by some authors.
The glottis has a longitudinal slit, and can be projected forwards when the pharynx is obstructed by a voluminous prey. An epiglottis is usually absent, or represented by a rudiment. It is, however, present in some large American species of Coluber (Pityophis), said to produce, when hissing, a loud and hoarse sound which has been compared to the bellow of the bull—hence the popular name of Bull-snakes by which they are known. It has also been found in a few allied species from Mexico, for which the genus Epiglottophis has been proposed. This epiglottis is a narrow, thin flap, erect in front of the glottis; it is not hinged, and therefore not capable of falling down to cover the opening of the windpipe during the process of swallowing, its function evidently being to increase the sound produced by the escape of the air from the windpipe.
The larynx is represented by two longitudinal bands of cartilage, united by transverse bands; it is extremely long in some snakes (Leptognathus).
The œsophagus, which may be extremely elongate, sometimes measuring almost one-third of the digestive canal, passes into the tubular or sac-like stomach, often with thickened walls, which itself gradually or abruptly merges into the narrower intestine. The windings of the small intestine are connected by ligamentous tissue, and enclosed in 80 a common sheath of peritoneum. In several of the Glyphodont Water-snakes (Homalopsinæ and Hydrophiinæ), the intestine is much convoluted; in Herpeton it is even longer than the body, although when coiled occupying only one-fourth of that length. The rectum is sometimes very short, sometimes rather long, and its anterior portion may have a short cæcum; it may be divided by transverse septa, with median or lateral perforation.
In snakes which swallow hard-shelled snails, the anterior part of the intestine has its inner wall furnished with zigzag muscular folds producing a reticulate appearance, followed farther down by transverse and then longitudinal folds. In these snakes the intestine is abruptly constricted behind the stomach, at which point the shells are broken or crushed after their contents have been digested; whilst in the egg-eating snakes, in which the eggshell has to be broken previous to its contents reaching the stomach, the oesophagus is narrowed in front of the latter, at the point where the tooth-like ventral processes of the vertebræ project and pierce the wall of the œsophagus in order to aid in this function, after which the broken shell is rejected through the mouth.
The more or less elongate, feebly-lobed kidneys are placed in the posterior part of the body, often extending nearly to the cloaca; the right is usually a little longer than the left, or extends a little farther forward, or even may commence where the other ends. 81 The suprarenal bodies are narrow and elongate, placed on the renal veins or on the vena cava inferior.
The ureters leave the hind ends of the kidneys, and open through the side-walls of the cloaca on a papilla which in the males contains also the opening of the vas deferens. There is no urinary bladder. The genital organs will be mentioned in the next chapter. The liver is usually long and narrow, measuring one-fifth to one-fourth the length of the body, on the right side of the alimentary canal, commencing just behind the heart or farther back. It is exceptionally short in Chersydrus. It is sometimes divided by transverse furrows. Its posterior extremity is bilobate, and the left lobe usually extends beyond the right, although the reverse has been observed in some snakes. The gall-bladder, which may be absent, is remarkable for its distance from the liver. The pancreas, elongate but comparatively small, is located near the spleen, on the left side of the alimentary canal, at a considerable distance from the liver.
The peritoneal part of the body-cavity is subdivided into a number of spaces or cœlomic compartments enclosed in serous capsules—viz., a posterior or intestino-genital, a gastric on the left side, and a pair round the liver, corresponding to its two lobes.
Fat-bodies are much developed, either in the form of small separate lobes, or as a continuous, much folded band, on each side of the body.
The genital glands are situated anterior to the kidneys, the right extending farther forward and often larger than the left. The testes are elongate. The vas deferens is closely folded proximally, and runs along the outer side of the kidney into the cloaca close to the ureter. The ovaries are elongate, and consist of two lamellæ, with a lymph-space between them. The oviduct extends from near the anterior extremity of the ovary to a common chamber, or vagina, which is above the rectum and opens into the cloaca; this vaginal chamber may be more or less completely divided into two.
The males are provided with a pair of intromittent organs, or hemipenes, each connected with one of the caudal vertebræ by a muscle (retractor penis) which often exceeds it in length. These organs are cylindrical or club-shaped and hollow, with the inner surface divided into numerous cavities and beset with papillæ, and usually also with hard spines, of which those towards the apex may be greatly developed, folded against the walls, and directed towards the extremity. Such spines are absent in 83 the snakes provided with claw-like rudiments of hind limbs. The cavities of the hemipenis are connected by a branch with the dorsal artery, and it is by a flow of blood into them that erection of the organ is accomplished. Each hemipenis is lodged in a cavity on each side of the base of the tail; when protruded it turns inside out, and the inner surface becomes the outer, the papillæ and erected spines serving to maintain a firm hold in the vagina, from which the organ cannot be withdrawn except by invagination. It has been observed that the presence of spines on the hemipenis is associated with much tougher vaginal walls. The organ is grooved along its entire length, the groove being the sulcus spermaticus, which, when the edges of the two hemipenes meet, forms with its fellow a canal to convey the semen into the oviduct; this sulcus may be bifurcate, as in the Viperids and some Colubrids.
Anal pockets, secretory organs on each side of the vent and lodged in the base of the tail, seem, in females, to be the homologues of the hemipenes; but this view cannot be held, since the same organs are present, though smaller, in males also, situated dorsally to the hemipenes. The glands with which they are provided produce the strong and offensive odour which appears to be a means of defence in our Grass-snake and other species, and which also serves to bring the sexes together, the glands being 84 more active during the breeding season. A Viper-catcher in France is said to obtain good results by rubbing his boots with these glands, as a means of attracting the snakes in the spring.
In European species pairing takes place in spring, sometimes again at the end of summer or in autumn. After hibernation the testes of the males are rather voluminous, and the sperm-ducts are often full of spermatozoa. The male gets alongside the female, sometimes seizing her round the neck with his jaws, and remains stretched out against her or twists the posterior part of his body in a few coils around hers. In the Vipers the bodies of the pairing individuals are completely entwined. The male then endeavours to bring the two anal orifices together, and when he has succeeded in getting the female to distend her cloacal opening, the intromittent organs are suddenly everted into the vagina. The union of the sexes sometimes lasts only a few minutes, but usually an hour or more; it has even been observed to last a whole day. Several copulations may take place at intervals of a few days. Many snakes are gregarious during the breeding season, and great numbers of males have been seen wriggling round the females, forming with their coils huge lumps or an entangled mass like a ball. The more or less prehensile tail with which thoroughly aquatic snakes, such as Hydrophis and Acrochordus, are provided, is no doubt of use in 85 facilitating the pairing, when it has to take place in the water. Our European Water-snakes pair on land.
During the rutting season a slight pressure on the base of the male’s tail may cause the protrusion of the hemipenes, and so may a violent blow on the spine of the reptile. Thus, recently killed specimens of our Adder, with the organs everted, have more than once been taken by the ignorant for snakes with hind limbs, a mistake which must be pardoned when we remember that male embryos of the slow-worm and of snakes, in which the hemipenes are normally everted, have been described by zoologists, who should have known better, as examples showing external vestiges of limbs.
The spermatozoa soon make their way up the oviducts, in which the ripe ova have previously descended, or which gradually descend shortly after, these ducts becoming dilated in consequence. There are usually more eggs in the right than in the left oviduct, although the reverse has occasionally been observed.
Some snakes lay eggs shortly after impregnation, or a few weeks later; in others the young undergo their development within the oviducts, each enveloped in a thin, transparent, membranous capsule, which is torn immediately before or immediately after parturition, such species being termed “ovoviviparous.” Just before oviposition the female curves 86 the base of the tail upwards, in order to extend the cloacal opening. The eggs are all produced together, usually at intervals of a few minutes, and generally adhere to one another by means of a sticky fluid secreted by the oviducts, thus forming a clump. In ovoviviparous snakes the young are born in succession, in the course of a few hours or of a few days. In many oviparous species it is the rule for freshly-laid eggs to contain more or less developed embryos, and Coronella punctata is said to produce thin-shelled eggs which hatch in less than half the time required for the eggs of its American congeners under the most favourable circumstances. There is thus almost every degree between oviparity and ovoviviparity.
These two modes of parturition bear no relation to the natural affinities of snakes. Thus, the European Coronella austriaca is ovoviviparous, and its North American congeners are oviparous; whilst, curiously, it is the inverse in the genus Tropidonotus. It was long believed to be an invariable rule for the Viperidæ to bring forth live young, the name Viper being derived from this well-known peculiarity, but it has now been ascertained that the South American Lachesis mutus, the Indo-Malay Lachesis monticola, and the African Causus and Atractaspis, lay eggs. All exclusively aquatic snakes, such as the Hydrophiinæ, are ovoviviparous, and thus dispensed from going on land for parturition.
87 The yolk entirely fills the eggshell; there is no albumen, or, if any exists, it is so much reduced as to easily escape observation. The eggshell in oviparous species contains a small amount of lime, and is not hard, but tough and parchment-like, white or yellowish; it is usually smooth, but in Pythons its surface is studded with minute pores, and in the American Zamenis constrictor it is rough, as if sprinkled over with loose grains of salt. The shape varies from a short oval to a long ellipse. It has been observed in some snakes that the eggs, on leaving the cloaca, are of an elongate shape, suggestive of a short cigar, and immediately after assume a more oval form. After they have been laid, the eggs absorb moisture and thus increase in size, especially in width; eggs which are at first twice as long as broad may be almost globular just before the birth of the young.
The number of eggs or young of one brood varies much according to the species, and also according to the age of the mother, large females usually producing a higher number and of a larger size than smaller specimens of the same species. Our European Zamenis, Coluber, and Coronella produce only 2 to 15; our Tropidonotus, 15 to 48; our Vipers, 3 to 22. Among exotics we may mention, as the most prolific, Bitis nasicornis, up to 47 young; Tropidonotus fasciatus, Abastor erythrogrammus, and Farancia abacura, 50; Lachesis lanceolatus, 60; Vipera russelli, 63; Boa constrictor, 88 64; Tropidonotus ordinatus, 78; Pseudaspis cana, 80; Python molurus, nearly 100 eggs.
The eggs are deposited in holes without any sort of nest, under moss or decomposing leaves, in accumulations of saw-dust, or in manure-heaps. In many cases it has been observed that the female remains for some time with her eggs or young, and in the large Pythons a sort of incubation takes place, the female remaining coiled in a spiral over the mass of eggs for six to eight weeks; an increase of several degrees in her temperature at that period has been ascertained by experiments conducted with every possible care, a remarkable fact in the case of a so-called “cold-blooded” animal.
The numerous reports of young snakes seeking refuge in their mother’s gullet have not been substantiated by satisfactory scientific evidence, and, although it is perhaps wise to say that the question remains an open one, it may be mentioned that, in Europe at least, trained observers who have devoted special attention to the habits of Vipers, in districts where these reptiles are exceedingly abundant, have never come across an instance of the form of maternal solicitude with which these snakes in particular have been credited. Not a single reported case of a female snake swallowing her young for protection rests on satisfactory evidence.
The embryo is closely coiled up in a spiral. Just before birth it is distinguished by a large, convex 89 head, with large, prominent eyes, and a comparatively short body, the scales and ventral shields being much shorter than later in life. The umbilicus is situated in the posterior part of the body, from six to ten times as far from the head as from the vent. Long after birth the umbilical slit remains visible, and affords a means of distinguishing very young snakes from older examples of smaller species. In oviparous species the embryo is provided with a very conspicuous egg-tooth, pointing forwards and projecting from the notch in the lower border of the rostral shield; this egg-tooth is much reduced, and sometimes very indistinct, in the ovoviviparous species. The function of the egg-tooth is to cut through the tough eggshell. This, after the young has left it, shows one or several slits in its anterior extremity, cut as clean as if with a sharp knife. The egg-tooth becomes loose soon after birth, and is shed within a few hours or a few days, sometimes even before birth in ovoviviparous species.
Frequent cases have been observed of dicephalous embryos or young, which may live for a short time; there are even records of a three-headed snake, stated to have been seen at Lake Ontario, and of snakes with two heads and two tails.
Unless prematurely born with a considerable mass of vitellus attached to the umbilicus, the young immediately after birth resent all interference, hissing, snapping, or puffing themselves up, after the 90 manner of their parents. The first shedding of the outer coating of the epidermis follows soon after birth; not before then does the young take to food.
No snake appears to be able to breed before it is four years old.
Well-authenticated instances of different species interbreeding are unknown, but specimens intermediate between Vipera berus and V. aspis, and between V. berus and V. ammodytes, have been assumed, with much probability, to be hybrids.
Snakes may be grouped, according to their mode of life, in five principal categories, gradually merging into each other, or two of them not infrequently found combined in one and the same species. These categories are:—Ground-snakes, Sand-snakes, Burrowing-snakes, Tree-snakes, and Water-snakes.
Ground-snakes may be defined as living above ground, and only occasionally climbing bushes or entering the water. Among European genera, Coronella and Vipera are perfect examples of this type, whilst Coluber and Zamenis approach the Tree-snakes in often ascending bushes, or even trees.
Sand-snakes are adapted for living on loose sand, in which they seek concealment. Such are Lytorhynchus and some Psammophis among the Colubridæ, Cerastes among the Viperidæ. Eryx connects this category with the next.
Burrowing-snakes live chiefly underground, and often have the visual organ atrophied in consequence, as in Typhlops; all the Typhlopidæ, Glauconiidæ, 92 and Uropeltidæ, belong to this category; the Viperid Atractaspis is also a burrowing type.
Tree-snakes spend the greater part of their life on bushes or trees. Corallus among the Boidæ, Dendrophis and Dendraspis among the Colubridæ, Atheris and various species of Lachesis among the Viperidæ, may be quoted as examples.
Of Water-snakes, some are exclusively aquatic, like the marine Hydrophiinæ and the typical Acrochordinæ (Acrochordus, Chersydrus) and Homalopsinæ (Hipistes, Herpeton). Chersydrus and Hipistes occur in the sea as well as in fresh water. Many species of Tropidonotus (T. tessellatus and T. viperinus in Europe), as well as the genera Helicops, Grayia, Boulengerina, etc., among the Colubridæ, Eunectes among the Boidæ, Ancistrodon piscivorus among the Viperidæ, are chiefly but not exclusively aquatic.
Our Tropidonotus natrix stands between the Ground-snakes and the Water-snakes; Boas and Pythons are as much Water-snakes as Tree-snakes. As shown by these and many other examples which might be given, a division into categories cannot always be applied with precision, nor does it convey an expression of the natural relationships of the species, as was believed by many systematists of the last century, who appealed to such adaptations for the definition of families.
A vertical pupil denotes more or less nocturnal habits. Nevertheless our European Vipers, which 93 are provided with such a contractile pupil, are far from exclusively nocturnal, delighting to bask in the sun, and pairing and feeding in the day-time. The Boidæ appear to be more nocturnal, but no snake is known to be absolutely so, and the two species of Coluber which have been found living in perfect darkness in limestone caves in the Malay Peninsula and China, where they feed chiefly on bats, occur also outside the caves, and probably never breed in them.
It is often stated in books that the organs of locomotion for the exceedingly elongate body of snakes are the ribs, and these creatures have even been compared to Centipedes. This statement is no doubt true to a certain extent for slow locomotion on uneven ground, when the ribs and the corresponding ventral shields afford a point of support; but it does not account for the rapid movements, as when a snake darts like an arrow in pursuit of its prey or to escape from an enemy. Besides, the winding motions are not different from those of a Slow-worm or Glass-snake, in which, encased as they are in a bony armour, the ribs cannot come into play at all. The action of the muscles alone is quite sufficient to account for the reptation of snakes, without the ribs having to play an essential part.
Not only the Cobras, but several harmless snakes, are able to raise the anterior third of the body vertically, when taking up a threatening attitude in 94 the presence of an enemy, at the same time widening or inflating the region behind the head.
Most snakes can climb, and in this case the ribs and ventral shields are of great assistance. The Tree-snakes, usually characterized by a very slender, sometimes compressed, body, or by a prehensile tail, are specially adapted for twining themselves round branches, and in several of them the presence of a keel on each side of the ventral and subcaudal shields, accompanied by a notch corresponding to the keel, affords an additional help for climbing on vertical uneven surfaces, such as the trunks of trees. This condition of the ventral shields has a bearing on the extraordinary mode of locomotion with which some Tree-snakes (Chrysopelea, and probably also Dendrophis) have long been credited by the Malays. We allude to the so-called Flying-snakes, remarkable for their habit of shooting down from trees and descending to the ground at an oblique angle, the body being kept rigid the whole time of the “flight.” It has been observed in Chrysopelea that the ventral surface between the lateral keels, which may be compared to hinges, can be drawn in and become deeply concave, whilst at the same time a slight dorso-ventral flattening of the body takes place. During this muscular contraction the snake is like a piece of bamboo bisected longitudinally, and is buoyed up in such a way as to explain its parachute-like descent.
All snakes are able to swim, and the more aquatic 95 kinds may spend a few hours under the water. A Python molurus is known to have remained alive in a basket sunk for thirty-six hours in a river. The best adapted for aquatic life are the Hydrophiinæ, or Sea-snakes, most of which never leave the water, and are quite helpless and soon die when brought on shore; their body is more or less compressed posteriorly, and the tail oar-shaped. Sea-weeds and barnacles sometimes settle on them. Algæ have also been observed growing on the fresh-water snake Herpeton tentaculatum.
As regards food, Burrowing-snakes, as well as a few small Ground-snakes, subsist mostly on worms, insects, and myriopods; Tree-snakes on lizards, frogs, birds and their eggs; Water-snakes on fishes and batrachians. Among the other types, some show a predilection for mammals, others for lizards or snakes, whilst not a few feed indiscriminately upon mammals, birds, reptiles and batrachians, even on slugs, insects, and worms, in addition. However surprising, it is a fact that spiny mammals are occasionally eaten, spines of the Madagascar Hedgehog (Ericulus) having been found in the excrements of a Boa madagascariensis. Even hard-shelled eggs and molluscs may constitute the principal or exclusive food of certain snakes.
Thus, Dasypeltis eats nothing but birds’ eggs, the shells of which are crushed in the gullet, by a special contrivance mentioned above (p. 80), and are soon 96 after rejected through the mouth as a pellet. Other snakes, such as Coluber and Lioheterodon show themselves partial to eggs in addition to live prey, but their alimentary canal does not depart from the normal, the eggs being broken in the stomach and the remains of the shells passed with the excrements.
The Amblycephalidæ subsist almost entirely on snails and slugs, the shells of the former being crushed in the anterior part of the intestine after their contents have been digested, and the débris are rejected through the vent. A small land tortoise has been found in the stomach of a Cobra (Naia haie) from Algeria.
Snakes which take large prey secure it according to three methods: By catching it simply with the jaws, and immediately proceeding to swallow it, as in Tropidonotus and in some of the Constrictors when dealing with small animals; by constriction, after having seized it with the jaws, crushing it in the coils of their body and thus killing it previous to feeding, as in the Boidæ and Coluber; or by poisoning, by a mere stroke with the fangs, the result being awaited before the meal is begun, as in most of the Viperidæ. Other poisonous snakes proceed according to the first method, the use of the venom being to reduce the struggles of the victim and to relax its muscles. Such snakes as are in the habit of previously killing their prey show little reluctance to accept dead food in confinement, a 97 thing which others usually refuse to do; they may, however, be deceived by the dead animal being agitated before them, and the system now adopted in our Zoological Gardens, of offering all snakes previously-killed animals, has been attended with comparative success.
Some species feed almost exclusively on other snakes, and often manage to swallow individuals as large as, or even a little larger than, themselves. Examples are known of harmless snakes showing a predilection for dangerous species, to whose poison they are immune (see p. 71).
As a rule snakes that eat fish will also eat batrachians, but nothing higher in the scale, although exceptions have been reported, such as the Anaconda feeding on mammals, birds, reptiles, and fish, and our Grass-snake having taken mice and birds. Some that feed chiefly on lizards and snakes will occasionally eat also mammals, and vice versa, but rarely frogs. On the other hand, European Vipers accommodate themselves to a more varied bill of fare, being known to feed on mammals, birds, reptiles, batrachians, insects, and slugs, and they have even been observed to eat voles showing signs of putrefaction.
The enormous prey which some snakes are able to swallow is quite astounding. Anacondas and Pythons, the largest snakes, have been known to swallow calves and good-sized antelopes with their horns, animals which, even after being somewhat 98 crushed by constriction, very much exceed the calibre of the snake. A Python molurus 17 feet long is reported on good evidence to have swallowed a gravid Axis deer. A Grass-snake half an inch in diameter can manage a frog or toad three times that width, and a Dasypeltis of the same size a hen’s egg. Such feats are rendered possible by the mobility of the jaws and palato-pterygoid arch on the cranium, and the elasticity of the ligaments by which they are attached (see above, p. 42), as well as by the mobility of the ribs and the absence of sternal apparatus, together with the great distensibility of the skin. When a snake proceeds to dispose of a large prey, which, if it be a mammal or bird, is usually seized head-first, it pulls itself forward by alternate movements of the jaws, the maxillary and the mandibular ramus of the one side, and then of the other, being extended anteriorly and laterally, the snake at the same time producing an abundant salivation which renders the prey very slimy. Several repeated alternate movements of the jaws bring the head of the prey to the gullet, where the muscles and ribs come into play, and the two sides of the jaws work no longer alternately, but together. When once in the œsophagus, the prey progresses with much greater facility, and usually reaches the stomach in a few minutes, whilst the previous process of deglutition may have lasted half an hour. While this laborious operation is going 99 on, the breathing of the snake is not impaired owing to a remarkable contrivance: the trachea can be protruded in such a manner as to bring its opening outside the mouth.
In cases where the victim is eaten alive, the snake has to contend with its struggles, but retrogression is rendered impossible by the backwardly-directed sharp teeth with which the jaws and palate are beset. A frog is usually caught by one of the hind limbs and swallowed back-first, the long hind limbs stretching forwards as they fold against the body; its struggles are often still apparent when it has reached the œsophagus. Snakes when caught immediately after a meal are in the habit of disgorging their food, and it sometimes happens that a frog or toad is thus vomited alive. An instance is known of a naturalist having captured a Grass-snake and put it in a linen bag. On opening it a short time after, great was his surprise to find the snake had escaped through a small hole in the bag, leaving instead a living toad too big to pass through the hole.
If not of too large a size, several animals will often be swallowed in rapid succession, after which the gorged snake will allow its digestive organs several days, or even weeks, of repose. A large Anaconda in the Paris Jardin des Plantes fed only thirty-six times in the course of seven years. Digestion is usually rapid in the small snakes, defecation taking place twenty-four to forty-eight hours after the feeding; it 100 lasts much longer in the large Boas and Pythons. Thus, in the above-mentioned Anaconda it has been observed to take from nine to thirty-eight days. Even the hardest bones of birds are decomposed by the gastric juices, but hairs, feathers, and horny productions, are passed with the excrements, sometimes forming regular balls. It is in most cases possible to tell, from an inspection of the dried fæces, what a snake has been feeding on, hairs, feathers, beaks, claws, epidermal horny shields, bits of tooth-enamel, being found mixed with the chalky matter which represents the decomposed bones. As a rule there is but one defecation after each meal, but there are in addition more frequent renal dejections, consisting chiefly of uric acid.
In captivity snakes show themselves capricious in the choice of food, one individual preferring mammals, whilst another, of the same species, will only take birds; and many, although to all appearances perfectly healthy, will persist in refusing all food, and allow themselves to die of starvation—a suicide which may require months, or even years, to accomplish. A Rattle-snake in the menagerie of the Jardin des Plantes in Paris has lived two years and two months without taking any food, a Python sebæ nearly two years and a half, a Boa madagascariensis four years and a month. A Viper aspis was kept for three years without food and without losing its vicious temper. Specimens thus fasting do not, as a rule, 101 renew their epidermis, or do so but very rarely. Our Common Adder can very seldom be induced to feed in captivity. Other snakes may rid themselves of all shyness to the extent of taking food from the hand, or show such appetite as to seize a prey immediately on being released from the small box or bag in which they have travelled for a considerable time.
Most snakes drink, and pretty often—not by lapping with the tongue, but by drawing in water from the mouth and immersing the anterior part of the head. Some are said to be fond of milk, but there is no foundation for the belief held by peasants, that they enter sheds with the object of sucking milk from the cows, which would be a material impossibility; their real purpose in visiting such places being a search for suitable dung-heaps in which to deposit their eggs.
Snakes cannot be credited with much intelligence or educability, nor do they display any very marked instincts. The least stupid and most easily tamed are the species of the genera Coluber and Coronella. There is, however, considerable difference in this respect between individuals of the same species. Most snakes, when freshly caught, defend themselves by biting, and some individuals retain their savage temper after months of captivity; others hardly ever bite, even if molested. The Common Grass-snake, for instance, hisses loudly and takes up a very threatening attitude, or even pretends to snap with 102 open mouth, but very seldom bites; its principal defensive action when caught consists in voiding a most repulsive secretion from its anal glands, which it evidently controls, as it ceases doing so when accustomed to being handled. The same snake also produces, during the spring, an oily exudation from the skin which has the same repulsive smell. Mr. H. N. Ridley has observed a Malay snake allied to Tropidonotus, Macropisthodon rhodomelas, to exude drops of a white viscid liquid from the skin of its neck, which is flattened out like that of a Cobra when in an attitude of defence, and he noticed that his dog, seizing the snake to worry it, foamed at the mouth as if he had been biting a toad.
The hissing is produced by the rapid expulsion of air from the lungs through the trachea and the notch at the end of the mouth, which is kept shut at the time. Snakes provided with an epiglottis (see p. 79) produce a much louder hissing. Other sounds are produced by some snakes. Thus, the Indian and African Vipers of the genera Echis and Cerastes make a curious, prolonged, rustling noise, by rubbing the folds of the sides of the body against one another. This sound is produced by friction between the serrated keels of the lateral scales, which are disposed obliquely with their tips directed downwards and backwards; the noise can even be repeated after the death of the animal, by twisting the body and thus rubbing or rasping these little saws against 103 one another. The same thing probably takes place in the African genus Dasypeltis, in which we find a similar arrangement of the scales, though to a less degree.
The best known sounding apparatus is that of the Rattlesnakes, described on p. 20. When alarmed, these snakes gather the body in a few coils or roll themselves up in a spiral, with the tail erect in the centre, and vibrating with great rapidity, whilst the head is ready for attack. Other snakes, such as the Ancistrodon and some species of Coluber and Zamenis, when excited, vibrate the tail in the same manner; but, being deprived of the sound-producing apparatus, this expression of their anger does not attract the same attention. It is from such a habit, however, that the rattle must have been evolved and perfected, not necessarily in a Lamarckian sense, but through the different steps by which evolution or creation has proceeded; Natura non fecit saltus, as Linnæus well said. Many suggestions have been made as to the use of the rattle. One of them is that the rattling resembles the sound made by locusts, and serves to decoy insect-eating birds; another, that it serves to call the sexes together. Probably it is useful to the snake as a warning to keep off disturbers which cannot serve as food, and thus prevents useless expenditure of venom, or even the breaking of the fangs. At any rate, it gives expression to the snake’s excitement, as does the voice 104 in the case of many other animals, and it seems reasonable to suppose that it may be applied to different purposes. With the advent of man, this means of attracting attention must tend to the more rapid extermination of the snakes which possess it.
Another curious behaviour is that of feigning death, as observed in a harmless but vicious-looking snake, Heterodon, often called Puff-adder in America. It looks more like a Viper than a harmless snake, and when disturbed hisses loudly and flattens out the anterior part of the body, much as does a Cobra, and pretends to strike, although it is one of the few snakes that never bite man. If, however, this display proves of no avail in frightening away the intruder, the snake rolls on its back and opens its mouth, and then lies for a time, which may exceed a quarter of an hour, absolutely motionless, as if dead. As soon as it thinks the danger over, it awakens from its spasm and rapidly moves off. It is the opinion of those who have most experience of this snake that this extraordinary behaviour is not to be explained as a convulsion or faint due to fright, but constitutes a deliberate trick to save its life. Individuals of the South African Ringhals (Sepedon hæmachates) and of the Common Grass-snake have also been observed to feign death.
The notion that snakes fascinate their prey, attracting it or reducing it to immobility by a mysterious power in their glittering eyes, is pure 105 fable. Animals placed in a cage with a snake evince no particular fright, and fly away when pursued, if not actually turning round to defend themselves. It is even dangerous to offer a good-sized snake a wild rat for food, as all keepers of menageries know.
In cold and temperate climates snakes hibernate, lying more or less torpid in holes or hollow trees, sometimes assembled in numbers and coiled together in a mass. The first thing they do in awakening in the spring is to cast the outer coating of the epidermis, as described above (p. 20). Several exuviations take place during the period of activity, sometimes pretty regularly every month, sometimes at very irregular intervals. A few days previous to this operation the snake is languid and abstains from feeding; its skin is dull and the sight impaired by the opaque condition of the lid; a day or two before moulting, the outer stratum of the epidermis becomes again transparent and the eye clear, through this stratum becoming detached from the subjacent tissue, until it is pulled off in one piece, by the snake rubbing itself against stones or bushes. The first exuviation takes place very shortly after birth.
Snakes are long-lived, although the limit of duration of life is not known in any of them. They grow slowly, and do not appear ever to reach sexual maturity until the fourth year, when they continue increasing in size for a long period. A Python reticulatus 106 and an Ancistrodon piscivorus are reported to have lived twenty-one years in captivity in Paris. The young of many snakes are very secretive, and are not often found in the open, those that are met with being as a rule either new-born or approaching sexual maturity.
Snakes are tenacious of life, and remarkable for the reflex movements which take place after they have been cut to pieces, the severed parts of the body and tail wriggling for a considerable time, and the head endeavouring to bite. Accounts of decapitated Rattlesnakes turning round and striking with their bloody stumps are probably not snake stories.
Like all other animals, snakes are infested with a multitude of vegetable and animal parasites, both external and internal. About 300 species of Ophidian parasites have been recorded; yet our knowledge of them is very imperfect. Although some 2,000 species of snakes are known, parasites have not been recorded for more than 168 species, and in the great majority of these (102) only a single parasite: a tick, a hæmogregarine, or some intestinal worm. Owing to the more frequent opportunity of dissecting them, the common menagerie snakes have yielded better records, notwithstanding the fact that they usually lose most of their parasites through constant handling, prolonged fasting, and artificial surroundings. Thus, we have a list of thirteen species for the Indian Python molurus, and one of twenty-two species for the Boa constrictor. But no systematic search appears to have been attempted, save, perhaps, in the case of a few European species.
It is interesting to notice that it was the finding of an Ophidian parasite which prompted Francesco Redi to write his famous “Observations on the Living 108 Animals which are found within Living Animals.” This work, a veritable treatise of comparative parasitology, published in 1684, caused the great naturalist, physician, and poet to be regarded as the father of that science. He tells us that in dissecting a curious dicephalous Vipera aspis, caught at Pisa, he found within the intestines a number of roundworms (Ascaris cephaloptera), and on the surface of one of the two lobes of the liver five cysts enclosing a small worm, which he rightly ascribed to the same species.
The parasites of snakes are here enumerated by Dr. L. W. Sambon, in systematic order.
Arthropoda.—Two families of the class Arachnida, the Ixodidæ and the Linguatulidæ, furnish numerous species parasitic on snakes.
Of the Ticks (Ixodidæ) we find, as a rule, species of the genera Amblyomma and Aponomma, the latter genus being almost entirely confined to Reptiles. A single species of the genus Hæmaphysalis (H. punctata, Can. and Franz, 1877) has been reported once from Vipera aspis. A few larval forms found on various snakes have been reported under the generic name Ixodes, but they probably belong either to Amblyomma or Aponomma.
The Ophidian Tick-parasites, like those of mammals, birds, lizards, and tortoises, appear to be in many cases the means of transmission of protozoal infections from snake to snake.
The Tongue-worms (Linguatulidæ) are, without 109 doubt, of the greatest possible interest. Their systematic position has ever been a puzzle to zoologists, and even now is a matter of controversy. They have been looked upon as Hirudinea by Winsberg (1765), Cestoda by Chabert (1787), Acanthocephala by Humboldt (1808), Trematoda by Rudolphi (1809), and Nematoda by Nordmann (1832). It was Van Beneden (1848) who first recognized their Arthropod nature, but he placed them amongst the Crustacea. Schubärt (1853) suggested that their proper position is amongst the Mites (Acarina), and Leuckart (1860) adduced important anatomical and embryological evidence in support of this view, which was confirmed by Railliet in 1883 and by Sambon in 1910.
No less than three out of the four genera of Linguatulids so far established are represented by species parasitic on snakes. They are the genera Porocephalus, Reighardia, and Raillietiella.
The genus Porocephalus is of special interest, because some of its species, such as Porocephalus armillatus, a parasite of African Pythons (Python regius, P. sebæ) and Puff-adders (Bitis arietans, B. nasicornis, B. gabonica), and Porocephalus moniliformis, a parasite of Oriental Pythons (Python molurus, P. reticulatus), are, in their nymphal stage, deadly parasites of mammals, including man.
The genus Reighardia was established by Professor H. B. Ward, in 1899, for a Linguatulid of gulls and 110 terns, first described, in 1861, by De Filippi. In 1910 Sambon included in this genus other similarly structured Linguatulids from crocodiles, monitors, and snakes.
The genus Raillietiella was established by Sambon in 1910 for a Linguatulid (Raillietiella boulengeri) of the African Puff-adders (Bitis arietans, B. gabonica). Amongst the characters of this genus is one of great structural and phylogenetic importance—viz., the position of the female sexual orifice at the anterior end of the abdomen, whilst in the other known genera it is at the posterior extremity.
According to Prowazek, Sambon, and Laveran, the Ophidian Linguatulids, which live as blood-suckers in the air-passages of their hosts, are able to foster and transmit the hæmogregarines of these hosts.
Acanthocephala.—The early encysted stages of several species of Thorn-headed worms (Acanthocephala), belonging to the family Echinorhynchidæ, have been reported from snakes belonging to very different genera, such as Boa, Tropidonotus, Zamenis, Drymobius, Xenodon, Dipsadomorphus, Oxyrhopus, Erythrolamprus, Diemenia, Naja, Elaps, Vipera, Lachesis. Their further development probably occurs in ophiophagous birds. Thus, Echinorhynchus oligacanthoides, Rud., the immature stages of which occur encapsuled within the body cavity of Lachesis lanceolatus and other neotropical snakes, when adult 111 is found attached to the intestinal mucosa of Milvus bidentatus.
Nematoda.—The roundworms (Nematoda) so far described from snakes belong to the families Ascaridæ, Strongylidæ, Trichotrachelidæ, and Filariidæ. Some of the genera belonging to these families, such as Cucullanus, Nematoxys, Oxysoma, are as yet represented by a single species in a single host; others, such as Ascaris, Polydelphis, Heterakis, Strongylus, Diaphanocephalus, Physaloptera, Trichosoma, number already several species more or less widely distributed.
Eelworm infection (ascariasis) is very common in snakes, and not infrequently the infection is a heavy one; Sambon twice found over fifty specimens of Polydelphis in Puff-adders (Bitis arietans). This investigator has shown that the snake eelworms undergo an encysted stage of development within the body cavity of their hosts before migrating into the intestinal lumen for the purpose of fertilization and oviposition. Thus, Redi was quite right in considering the immature, encysted forms found in one of the livers of his double-headed Asp as belonging to the same species of eelworm (Ascaris cephaloptera) as that which the snake harboured in its intestine.
Professor A. Railliet, whilst examining specimens of Polydelphis which had been preserved for nearly two months in a 3 per cent. solution of formalin, 112 found that the ova within their uterine tubes had undergone development, and still contained living embryos; indeed, some of these hatched under the microscope, and moved very actively in the preserving fluid. This is in no way surprising, because even after several years of preservation in formalin solution the embryos of other species of eelworms (Ascaris equorum, A. marginata) have been found in a living condition.
Trematoda.—The Flukes (Trematoda) of snakes, so far described, belong to the following genera: Agamodistomum, Astiotrema, Brachylaimus, Cotylotretus, Dicrocœlium, Diplodiscus, Distoma, Halipegus, Lecithodendrium, Metorchis, Opisthogonimus, Opisthorchis, Plagiorchis, Saphedera, Telorchis, Tetracotyle, Zeugorchis.
Cestoda.—Save a few larval forms (Cysticercoides, Piestocystis, Sparganum), the known tapeworms (Cestoda) of the Ophidia belong to the genera Bothridium and Proteocephalus.
Protozoa.—Numerous species of Hæmogregarines have been described from snakes. As a rule the forms seen in the peripheral blood are sporonts, the schizogonic cycle occurring in the lungs. The sporonts do not greatly alter their host cells; they are invariably doubled up within a more or less thick capsule. Some species show a marked sexual differentiation, others not. Trypanosomes, Spiroechaudinniæ, and Plasmodidæ have also been described from the blood of various snakes.
113 Within the alimentary tube have been found species of Trichomonas and Caryospora.
Bacteria.—Acid-fast bacilli have been described in tubercular lesions found in snakes by Sibley, Gibbs and Shurley, Shattock, Hausemann, and Sambon.
The so-called “canker,” which so frequently develops in the oral cavity of captive snakes, is also a bacterial disease, due to a specific bacterium of thick, rod-shaped form.
Acanthocephala.
Echinorhynchus inæqualis, Rudolphi.
Echinorhynchus polyacanthus, Creplin.
Nematoda.
Strongylus auricularis, Zeder.
Strongylus catanensis, Rizzo.
Trichosoma mingazzini, Rizzo.
Oxysoma brevicaudatum, Zeder.
Nematoxys commutatus, Rudolphi.
Ascaris cephaloptera, Rudolphi.
Trematoda.
Opisthorchis caudatum, Polonio.
Dicrocœlium assula, Dujardin.
Diplodiscus conicum, Polonio.
Tetracotyle colubri, v. Linstow.
114
Distoma acervocalciferum, Gastaldi.
Distoma allostomum, Diesing.
Distoma nematoides, Mühling.
Saphedera naja, Rudolphi.
Brachylaimus signatum, Dujardin.
Telorchis ercolanii, Monticelli.
Lecithodendrium nigrovenosum, Bellingham.
Plagiorchis mentulatus, Rudolphi.
Cestoda.
Ligula panceri, Polonio.
Nematoda.
Strongylus denudatus, Rudolphi.
Physaloptera abbreviata, Rudolphi.
Physaloptera striata, v. Linstow.
Trematoda.
Plagiorchis mentulatus, Rudolphi.
Acanthocephala.
Echinorhynchus lobianchii, Monticelli.
Trematoda.
Distoma allostomum, Diesing.
Opisthorchis caudatum, Polonio.
Telorchis ercolanii, Monticelli.
Astiotrema monticellii, Stossich.
Cestoda.
Ligula pancerii, Polonio.
115 Protozoa.
Hæmogregarina viperina, Billet.
Acanthocephala.
Echinorhynchus cinctus, Rudolphi.
Echinorhynchus polyacanthus, Creplin.
Echinorhynchus heterorhynchus, Parona.
Nematoda.
Strongylus catanensis, Rizzo.
Filaria parvomucronata, Rizzo.
Trichosoma sonsinoi, Parona.
Trematoda.
Distoma subflavum, Sonsino.
Brachylaimus baraldii, Sonsino.
Saphedera naja, Rudolphi.
Cestoda.
Cysticercus acanthotetra, Parona.
Cysticercoides rostratus, Mingazzini.
Acanthocephala.
Echinorhynchus oligacanthus, Rudolphi.
Nematoda.
Ascaris cephaloptera, Rudolphi.
Trematoda.
Plagiorchis sauromates, Poirier.
Protozoa.
Hæmogregarina, sp.
Protozoa.
Hæmogregarina colubri, Börner.
Nematoda.
Tricheilonema megalochilum, Diesing.
Physaloptera colubri, Rudolphi.
Cestoda.
Piestocystis dithyridium, Diesing.
Protozoa.
Monocercomonas colubrorum, Hammersch.
Protozoa.
Hæmogregarina coronellæ, França.
Nematoda.
Physaloptera dentata, v. Linstow.
Trematoda.
Agamodistomum viperæ, v. Linstow.
Tetracotyle colubri, v. Linstow.
Arthropoda.
Hæmaphysalis punctata, Can. & Franz.
Acanthocephala.
Echinorhynchus cinctus, Rudolphi.
117 Nematoda.
Ascaris cephaloptera, Rudolphi.
Diaphanocephalus viperæ, Rudolphi.
Protozoa.
Caryospora simplex, Léger.
Hæmogregarina samboni, Giordano.
Nematoda.
Ascaris ammodytis, Rudolphi.
Ascaris cephaloptera, Rudolphi.
Representatives of the order Ophidia are found over the whole world, with the exception of Iceland, Ireland, and New Zealand, between the Northern limit of 67° in Europe (Vipera berus), 60° in Asia (Vipera berus), and 52° in America (Tropidonotus ordinatus), and the Southern limit of 44° (Philodryas schotti). The highest altitudes reached by them are 14,000 feet in the Himalayas (Tropidonotus baileyi), 9,700 feet in the Alps (Vipera aspis), and 9,000 feet in the Andes (Liophis albiventris). They are most numerous between the tropics, and the number of species gradually diminishes to the North and South.
For the purpose of showing the distribution of the principal groups, we will follow the divisions into families and subfamilies enumerated above (p. 4).
Typhlopidæ.—S.E. Europe, S. Asia, Africa, Australia (exclusive of Tasmania), C. and S. America, and W. Indies.
Glauconiidæ.—S. Asia (as far E. as Sind), Africa (exclusive of Madagascar), C. America (extending into the S. parts of N. America), S. America.
Pythoninæ.—S. Asia, Africa (exclusive of Madagascar), Australia (exclusive of Tasmania), C. America.
119 Boinæ.—S.E. Europe, C. and S. Asia, N. Africa, Madagascar, Mauritius, W. Polynesia, S.W. of N. America, C. and S. America, and W. Indies.
Ilysiidæ.—S.E. Asia, S. America.
Uropeltidæ.—India and Ceylon.
Xenopeltidæ.—S.E. Asia.
Acrochordinæ.—S.E. Asia, C. America.
Colubrinæ.—The whole range of Ophidia, except Tasmania.
Dasypeltinæ.—Africa (exclusive of Madagascar).
Homalopsinæ.—S.E. Asia, N. Australia.
Dipsadomorphinæ.—S. Europe, C. and S. Asia, Africa, Australia (exclusive of Tasmania), C. America (extending into the S. parts of N. America), S. America.
Elachistodontinæ.—India.
Hydrophiinæ.—Indian and Pacific Oceans.
Elapinæ.—S. Asia, Africa (exclusive of Madagascar), Australia and Tasmania, Fiji Islands, C. America (extending into the S. parts of N. America), S. America.
Amblycephalidæ.—S.E. Asia, C. and S. America.
Viperinæ.—Europe, Asia, Africa (exclusive of Madagascar).
Crotalinæ.—S.E. Europe, Asia, America.
The Zoogeographical Regions into which the world is usually divided (Palæarctic or Europo-Asiatic, Oriental or Indian, Ethiopian or African, Australian, Nearctic or North American, Neotropical or South 120 American) do not lend themselves any better than the ordinary divisions of physical geography to the study of the distribution of Snakes. Contrary to what we find in dealing with the Tortoises, Australia does not show any special affinity to South America, and, as in the case of the Lizards, it must be regarded as an impoverished extension of the Indo-Malay fauna; as with the Lizards, also, Europe and Africa hang together, whilst Madagascar stands apart, distinguished by many negative features and some points of agreement with South America (Boidæ). There is a greater difference between the Snakes of Europe and those of Eastern Asia than there is between the latter and those of North America, whilst in Lizards a primary distinction must be made between the Old World and the New. Southern Asia east of Persia (the Oriental Region) is the great Ophidian centre, all the groups mentioned above, with the exception of the Dasypeltinæ, having representatives within its limits, and a large and very distinct family, the Uropeltidæ, being confined to it. The Pythoninæ occur along with the Boinæ, the Viperinæ with the Crotalinæ, and the Elapinæ are represented by varied forms, as they are also in Africa and still more in Australia, where they form the overwhelming majority, and in some parts, as well as in Tasmania, the exclusive Ophidian population. The coasts of India and Malaya are also the home of the great majority of the Hydrophiinæ. Large genera 121 like Tropidonotus, Zamenis, and Coluber, extend over the Europo-Asiatic and North American regions, but they are equally well represented in the Oriental. The great difference between Madagascar and Africa is, as we have said, very striking. Madagascar possesses Boidæ generically identical with those of South America, but otherwise only Typhlopidæ, Colubrinæ, and Dipsadomorphinæ; whilst in the greater part of Africa the Boinæ are replaced by the Pythoninæ, and the Glauconiidæ, Elapinæ, and Viperinæ are generally distributed. North America agrees with Asia and South America in its Crotalinæ, otherwise its Ophidian fauna is not very different from that of Europe, although much richer, and South America shares the Glauconiidæ with Africa and the Ilysiidæ with Southern Asia. South America is rich in Colubrinæ and Dipsadomorphinæ, nearly all generically different from those of other parts of the world, and the Elapinæ are represented by the single genus Elaps, with many species, two of which extend to the southern parts of North America.
This rapid sketch of the principal facts of Ophidian distribution suffices to show how difficult it would be to frame geographical regions that would give expression to these facts. Such regions would necessarily be very different from those adopted in dealing with the distribution of the other divisions of the class Reptilia. This is a task which need not be attempted on the present occasion.
122 A few words as to the salient characters of the European fauna, which is a poor one as compared with other parts of the world. The single species of the genera Typhlops and Eryx must be regarded as outposts from South-Western Asia; the single species of Ancistrodon, which extends from Central Asia into a very small territory to the south-east, is also an Asiatic type. The genera Tropidonotus, Zamenis, Coluber, Coronella, and Contia, are characteristic of the Northern Hemisphere, and the first three are, besides, equally well represented in the Oriental region; a few species of Tropidonotus are also found in Africa and Madagascar. Cœlopeltis, Macroprotodon, and Tarbophis are the northern outposts of an Afro-Indian group, although, with the exception of the third, exclusively confined to the circum-Mediterranean district. The genus Vipera is also represented in East Africa and in Southern Asia, but the species V. berus is essentially a northern type, extending to the highest latitude reached by any snake, and ranging all over Northern Asia to the Amur and Sachalien. The same species reaches the greatest altitude at which any snake has been observed on the northern side of the Alps—viz., 9,000 feet.
Of the twenty-eight species inhabiting Europe, only two are generally distributed: Tropidonotus natrix and Coronella austriaca. One is to be regarded as a northern form, although occurring locally in the south: Vipera berus. It is the reverse with Coluber longissimus. 123 The others may be described as southern forms, two only as ranging from west to east: Zamenis gemonensis and Cœlopeltis monspessulana; one of more central habitat: Vipera ursinii. The remainder may be divided into two groups—those of more western, and those of more eastern distribution. To the first group belong Tropidonotus viperinus, Zamenis hippocrepis, Coluber scalaris, Coronella girondica, Macroprotodon cucullatus, Vipera aspis and Vipera latastii; to the second, Typhlops vermicularis, Eryx jaculus, Tropidonotus tessellatus, Zamenis dahlii, Coluber quatuorlineatus, dione, leopardinus, Contia modesta, Tarbophis fallax and iberus, Vipera renardi, ammodytes, lebetina, and Ancistrodon halys.
A remarkable fact in the distribution of European Snakes is the altitudinal range of Vipera berus, V. aspis, and V. ursinii. The first being the northernmost snake, generally distributed in Northern Europe and more locally in the south, should, one would expect, be a mountain form in the south. This is so in Switzerland, where it occurs chiefly between 2,500 and 9,000 feet, on the northern aspect of the Alps, whilst V. aspis lives at altitudes below 5,000 feet; but on the southern aspect of the same chain things are reversed, and V. berus is replaced by V. aspis, which reaches an altitude of 9,700 feet, whilst the former shows a tendency to abandon the mountains, and has established itself in a few localities in the plain of North Italy. Again, in France 124 V. berus is the northern and V. aspis the southern species, yet the latter is the only one found on the French side of the Pyrenees (up to 7,250 feet), whilst the former reappears in North-Western Spain and Portugal at very low altitudes, even at sea-level. V. ursinii is a mountain form in Italy (Abruzzi), in France (Basses-Alpes), and in the Balkan Peninsula (up to 6,800 feet); but it is restricted to the plain in Lower Austria and Hungary, where V. berus occurs only in the mountains.
Only three species are entirely confined to Europe:
Coluber scalaris, Vipera ursinii, and V. aspis.
Of the species which range outside Europe, the following occur both in Western Asia and in North Africa:
Eryx jaculus, Tropidonotus natrix, Cœlopeltis monspessulana, Vipera lebetina.
In Western Asia and the North-East of Egypt:
Tropidonotus tessellatus, Zamenis dahlii.
In Western Asia:
Typhlops vermicularis, Zamenis gemonensis, Coluber quatuorlineatus, C. dione, C. longissimus, C. leopardinus, Coronella austriaca, Contia modesta, Tarbophis fallax, T. iberus, Vipera renardi, V. berus, V. ammodytes, Ancistrodon halys.
In North Africa:
Macroprotodon cucullatus.
125 In North-West Africa:
Tropidonotus viperinus, Zamenis hippocrepis, Coronella girondica, Vipera latastii.
The following lists will help to elucidate the distribution of the snakes in the different parts of Europe:
1. Tropidonotus natrix (as far north as 65°).
2. Coronella austriaca (as far north as 63°).
3. Vipera berus (as far north as 67°).
1. Tropidonotus natrix (England and Wales, extreme south-east of Scotland).
2. Coronella austriaca (Surrey, Berkshire, Hampshire, and Dorsetshire).
3. Vipera berus.
1. Tropidonotus natrix.
2. Coronella austriaca.
3. Vipera berus.
1. Tropidonotus natrix.
2. Tropidonotus tessellatus (Middle Rhine and Moselle, Saxony).
3. Coluber longissimus (Denmark, Schlangenbad, Treves).
126 4. Coronella austriaca.
5. Vipera berus.
6. Vipera aspis (Black Forest, Lorraine).
1. Tropidonotus natrix.
2. Tropidonotus viperinus (as far north as South Brittany and Fontainebleau).
3. Zamenis gemonensis (south, locally as far north as the Sarthe and Aube).
4. Coluber longissimus (locally as far north as South Brittany, South Normandy, and Fontainebleau).
5. Coluber scalaris (Mediterranean Littoral).
6. Coronella austriaca.
7. Coronella girondica (south and west as far north as the Charente-Inférieure).
8. Cœlopeltis monspessulana (Mediterranean Littoral).
9. Vipera ursinii (Basses-Alpes).
10. Vipera berus (as far south as the Loire basin, the Central Plateau, and the Alps).
11. Vipera aspis (as far north as the Loire basin, Fontainebleau, and Lorraine).
1. Tropidonotus natrix.
2. Tropidonotus viperinus.
127 3. Zamenis gemonensis (Catalonia).
4. Zamenis hippocrepis (absent from the north).
5. Coluber longissimus (Andalucia).
6. Coluber scalaris.
7. Coronella austriaca (north and north-west).
8. Coronella girondica.
9. Cœlopeltis monspessulana.
10. Macroprotodon cucullatus (centre and south, Baleares).
11. Vipera berus (north-west).
12. Vipera aspis (Pyrenees).
13. Vipera latastii (absent from the north).
1. Tropidonotus natrix.
2. Tropidonotus tessellatus (as far south as Naples; absent from the islands).
3. Tropidonotus viperinus (Liguria, Piedmont, Ticino, Corsica, Sardinia, Sicily).
4. Zamenis gemonensis.
5. Zamenis hippocrepis (Sardinia).
6. Coluber quatuorlineatus (south and Sicily).
7. Coluber longissimus (absent from Corsica).
8. Coluber leopardinus (south and Sicily).
9. Coronella austriaca (absent from Corsica and Sardinia).
10. Coronella girondica (absent from Corsica and Sardinia).
11. Cœlopeltis monspessulana (Western Liguria, Sicily).
128 12. Vipera ursinii (Abruzzi).
13. Vipera berus (the Continental part only).
14. Vipera aspis (absent from Corsica and Sardinia).
15. Vipera ammodytes (Northern Venetia).
1. Tropidonotus natrix.
2. Tropidonotus tessellatus.
3. Zamenis gemonensis (South Tyrol, Littoral, South Hungary).
4. Coluber quatuorlineatus (Istria).
5. Coluber longissimus.
6. Coluber leopardinus (Istria).
7. Coronella austriaca.
8. Coronella girondica (South Tyrol).
9. Cœlopeltis monspessulana (Istria).
10. Tarbophis fallax (Istria).
11. Vipera ursinii (Lower Austria, Littoral, Hungary).
12. Vipera berus.
13. Vipera aspis (South Tyrol, Littoral).
14. Vipera ammodytes (South Tyrol, Styria, Carinthia, Carniola, Littoral, South Hungary).
1. Typhlops vermicularis (Greece, Turkey, Bulgaria).
129 2. Eryx jaculus (Greece, Turkey, Roumania).
3. Tropidonotus natrix.
4. Tropidonotus tessellatus.
5. Zamenis gemonensis.
6. Zamenis dahlii (coast of the Adriatic, Greece).
7. Coluber quatuorlineatus.
8. Coluber longissimus.
9. Coluber leopardinus.
10. Coronella austriaca.
11. Cœlopeltis monspessulana (West Coast, Greece, and islands).
12. Tarbophis fallax (West Coast, Greece and islands, Constantinople).
13. Vipera ursinii (Bulgaria, Bosnia, Herzegovina, Montenegro).
14. Vipera berus (Bosnia, Herzegovina, Roumania).
15. Vipera aspis (Bosnia).
16. Vipera ammodytes.
17. Vipera lebetina (Cyclades).
1. Tropidonotus natrix (as far north as 60°).
2. Tropidonotus tessellatus (south).
3. Zamenis gemonensis (south).
4. Zamenis dahlii (Caucasus).
5. Coluber quatuorlineatus (south).
6. Coluber dione (south, between Volga and Ural).
7. Coluber longissimus (south, Poland).
130 8. Coluber leopardinus (Crimea).
9. Coronella austriaca (as far north as 57°).
10. Contia modesta (Caucasus).
11. Tarbophis iberus (Caucasus).
12. Vipera renardi (south).
13. Vipera berus (as far north as 64°).
14. Ancistrodon halys (south, between Volga and Ural).
Without attempting anything like a complete bibliography, we have compiled a list of faunistic works and papers dealing with the snakes of Europe:
Schreiber, E.: Herpetologia Europæa. Zweite Auflage, Jena, 1912, 8vo.
Steinheil, F.: Die Europaeischen Schlangen. Kupferdrucktafeln nach Photographien der lebenden Tiere. Jena, 1913, 4to. (in progress).
Bell, T.: A History of British Reptiles. 2nd edit., London, 1849, 8vo.
Cook, M. C.: Our Reptiles. London, 1865, 8vo.
Leighton, G.: The Life-History of British Serpents. Edinburgh and London, 1901, 8vo.
Gadeau de Kerville, H.: Faune de la Normandie. IV. Reptiles. Paris, 1897, 8vo.
131 Lataste, F.: Essai d’une Faune Herpétologique de la Gironde. Bordeaux, 1876, 8vo.
Martin, R., et Rollinat, R.: Vertébrés sauvages du Département de l’Indre. Paris, 1894, 8vo.
Fatio, V.: Faune des Vertébrés de la Suisse. III. Reptiles et Batraciens. Geneva and Basle, 1872, 8vo.
Boscá, E.: Catalogue des Reptiles et Amphibies de la Péninsule Ibérique et des Îles Baléares (Bull. Soc. Zool. France, 1880).
Bonaparte, C. L.: Iconografia della Fauna Italica. II. Anfibi. Rome, 1832-1841, fol.
Camerano, L.: Monografia degli Ofidi Italiani (Mem. Acc. Torin., [2] xxxix., 1888, and xli., 1891).
Dürigen, B.: Deutschlands Amphibien und Reptilien. Magdeburg, 1890-1897, 8vo.
Leydig, F.: Ueber die Einheimischen Schlangen (Abh. Senck. Ges., xiii., 1883).
Werner, F.: Die Reptilien und Amphibien Oesterreich-Ungarns und der Occupationsländer. Vienna, 1897, 8vo.
Bedriaga, J. de: Die Amphibien und Reptilien Griechenlands (Bull. Soc. Nat. Mosc., 1881).
Bory de St. Vincent, J. B.: Expédition Scientifique de Morée. Reptiles. Paris, 1832-1835, 4to., fol. atlas.
Kowatcheff, W. T.: Herpetological Fauna of Bulgaria. Philippopolis, 1912, 8vo. [Bulgarian text.]
Kiritzescu, C.: Contribution à la Faune Herpétologique de Roumanie. Sauriens et Ophidiens (Bull. Soc. Rom. Bucarest, x., 1901).
Nikolsky, A.: Herpetologia Rossica. St. Petersburg, 1905, 4to. [Russian text.]
Strauch, A.: Die Schlangen des Russischen Reichs. St. Petersburg, 1873, 4to.
Under this head, the question of poisonous snakes naturally occupies the first place. In addition to what has been said above in Chapter VI., dealing with the anatomical and physiological aspects of the subject, we have to allude to the accidents caused by these dangerous reptiles, and the measures taken to combat them.
The enormous mortality for which snake-bite is responsible in India is well known. Statistics establish the fact that an average of 20,000 human lives are thus lost annually: 24,264 is the official return for 1911. In Australia, where highly poisonous snakes of various genera and species abound, the fatal cases are likewise very numerous, though less in proportion than in South America, and no doubt also in Africa. In the small island of Martinique, the Fer-de-Lance, Lachesis lanceolatus, causes every year the death of about 100 human creatures. Though numerous in species, the poisonous snakes of Ceylon cause a comparatively small mortality—200 per annum.
Modern research has resulted in the discovery of 134 the only effective antidote for snake-venom intoxication: the serotherapic treatment. An animal that has been treated over a length of time with the venom of a poisonous snake, such as a Cobra, yields a serum which is antitoxic towards that venom; but the great difficulty resides in the specificity of the different poisons, which often renders the use of the serum ineffective in countries like India and Australia, where several kinds of poisonous snakes occur in the same district (see above, p. 67). In India, where a special laboratory has been established for the supply of antivenine, at the Central Institute of Kasauli, it has been found impossible to obtain any venoms but those of the Cobra and Russell’s Viper in sufficient quantity to immunize animals, and thus produce the serum necessary for dealing with the bite of the King Cobra, the Krait, and the Echis Viper.
In Pondicherry the French Government places annually a sum of 200 rupees at the disposal of the director of the hospital for obtaining Cobra poison, the snakes, to be brought alive, being paid for to the natives at the rate of half a rupee to one rupee each, according to size and condition. Six hundred and fifty-three specimens were thus purchased in less than two years (1901-1903). The poison is utilized for the preparation of Calmette’s antivenine, which, as we have said above, is only effective against cobra poison, and, unfortunately, useless for the cure of bites from other species.
135 In Brazil, where the number of accidents is estimated at 19,200 per annum, and that of fatal cases at 4,800, over 2,000 snakes (Lachesis and Crotalus) are brought annually to the Serotherapic Institute of Batantan, in the province of S. Paolo, for the preparation of the antitoxic serum, which is given in exchange for the snakes. According to the latest report of the Institute (1911), two serums are distributed: the anti-crotaline (for Rattlesnake bite) and the anti-bothropine (for Lachesis bite); the third, the anti-elapine (for Coral-snake bite), is in course of preparation.
In many countries a premium has for years been paid for the heads of poisonous snakes, and has led to the destruction of enormous numbers of them, without, however, resulting in a very appreciable diminution of the dangerous reptiles. More than £12,000 has been spent for this purpose in India alone; the numbers destroyed in 1885 and 1886 throughout British India amount to 420,044 and 417,596 respectively. About forty years ago the Governor of St. Lucia offered a reward of 4d. for every Fer-de-Lance’s head. But the negroes caught them alive and bred families of snakes for the sake of the reward, and thereby made what was for them a little fortune, these snakes bringing forth up to sixty young at a birth. The reward had to be abolished very soon.
Now about the Vipers of Europe, the only really dangerous snakes of this part of the world.
136 Although the Adder, Vipera berus, is quite common in many parts of England and Scotland, accidents caused by its bite are rarely heard of, and cases of death are few and far between. It is not so, however, on the Continent, where the same species, and especially its close ally, the more southern V. aspis, are responsible for many fatalities, due no doubt to the more virulent action of the venom in a warmer climate.
In the French Departments Loire-Inférieure and Vendée, where these snakes are very plentiful, three or four cases of death are reported annually. From 1860 to 1868, 370 serious accidents to man have been carefully recorded, 53 ending in death, not only in the case of children, but also of adults of all ages, in 10 cases within one to twenty-four hours. In the Puy-de-Dôme cases of death are of frequent occurrence. In Germany and in Switzerland, 12 or 13 per cent. of the cases on record have ended fatally. Instances of death from the bite of the south-eastern V. ammodytes are also not infrequent. On the other hand, the bite of V. ursinii, which is but seldom inflicted, is not known to have ever resulted in death.
It must be borne in mind that accidents are much more frequent in districts where the poorer classes are in the habit of going about barefoot.
Anyhow, it is certain that Vipers are a serious danger in many parts of Europe, not only to man, but also to horses, cattle, and dogs. And it is not surprising 137 that efforts have been made to reduce their numbers. The most efficacious means, besides the protection of certain animals and birds which feed on Vipers, appeared to be the institution of premiums to be paid for the heads of the dangerous snakes. By offering 21⁄2d. per head, 500,000 Vipers (V. aspis) were destroyed from 1864 to 1890 in three French departments, Haute-Saône, Doubs, and Jura, and in one district (Chaumont) of the Haute-Marne 57,045 were killed from 1856 to 1861; this gives an idea of the extraordinary abundance of these snakes in some parts of France. In the Puy-de-Dôme the premium was fixed for a time at 5d., and one man managed to destroy in the course of seven years 9,175 Vipers (V. berus and V. aspis). A woman in the Deux-Sèvres has made a living for many years by catching Vipers, the heads of which were paid to her at the rate of 5d. each. The average number of her captures amounted to 2,062 per annum (mostly V. aspis). Around Oesnitz in Saxony, 2,140 V. berus were killed in 1889, and 3,335 in 1890. In a single district in Southern Styria the heads of 4,197 V. berus and 7,381 V. ammodytes were sent in for the reward in the course of two years (1892, 1893).
In spite of all this effort, the institution of the bounty has not answered expectations, and, with the exception of a few districts, Vipers remain as plentiful as ever, showing what little man can do in altering the equilibrium of Nature, except by interfering 138 with the natural conditions under which animals live. Cultivation of the ground or destruction by fire of the vegetation of the wilderness seems to be the only efficacious means of getting rid of so abundant and prolific a creature as the Viper.
A word may be said, however, in defence of Vipers: they do a great deal of good to agriculture by the destruction of small rodents, on which they feed chiefly, and whose multiplication they serve to keep in check. It must be pointed out that, with the exception of the species of Coluber and Zamenis, other European snakes are to be regarded as indirectly injurious to agriculture, feeding as they do mainly on lizards or frogs and toads, which, as insectivores, deserve to be protected.
Snakes are not of much economic value to man. Tanned skins of Boas and Pythons are utilized for making shoes and fancy articles, such as purses, pocket-books, blotters, etc., and the Siamese make the drum-heads of native drums out of the skins of Pythons and Acrochordus. To say nothing of savages, who seem to be partial to the flesh of large snakes, the peasantry in some parts of France do not disdain snakes as an article of food, the Grass-snake being occasionally served in village inns under the name of Anguilles de haies, or hedge-eels.
Viper fat has for a long time been in request as an ointment in the case of various affections, and much used by quack doctors in the preparation of their 139 remedies. Some forty years ago a chemist in Challans (Vendée) collected Vipers (V. aspis) for medicinal purposes, and was able to send several thousands to Paris in the course of a few years, thus realizing a considerable sum of money, but the demand has gradually fallen off since.
Very frequent in the past, snake-worship is still prevalent in many parts of India, where the Cobra is held in great veneration, and is never willingly killed by the Hindoo. In pre-Buddhist days the gods were represented with a canopy of five or seven Cobras over them. The North African Cobra was sacred to the ancient Egyptians, and is profusely represented on the monuments and tombs; it was also an emblem of the physical sun, and, as a sign of royal power, along with the sun’s disc, formed part of the headdress of all solar deities. The Greeks and Romans also worshipped snakes, and the god of medicine is represented holding a snake, which is supposed to be Coluber longissimus, the so-called “Æsculapian snake”; the occurrence at the present day of certain common Italian species (Zamenis gemonensis, Coluber longissimus, Tropidonotus tessellatus) in isolated localities of Central Europe, formerly Roman settlements, has been attributed to their importation for use in the temples.
Snake-charmers have existed from the remotest antiquity, and are still to be found among all races of men, from the accomplished Indian juggler down 140 to the more commonplace European snake-catcher, who boasts of his immunity, and of his art of attracting snakes by devices of which he has the secret. The Libyan Psillii of the ancient Romans have handed down their art to the present day, and their performances are to be witnessed in most of the towns of Egypt and Tunisia. But India above all lands is reputed for its snake-charmers, and the favourite species used by them is the Cobra, which, by the way in which it raises the anterior part of the body and expands the region behind the head, lends itself better than any other to the display. Constantly facing the man before him, and swaying the raised anterior part of the body, it seems to dance to the music performed by the snake-man, people believing it to be charmed by the sounds of the instrument. However, anyone sitting on the ground in front of a Cobra, and swaying the body from side to side as does the man, can obtain the same result without the aid of any sort of music.
The most puzzling thing about these performances is how the man can thus play with impunity with so deadly a snake. It is a mistake to think that the snake is rendered harmless through the poison fangs having been extracted, although this subterfuge is frequently resorted to by the less accomplished jugglers. The immunity of the snake-charmer is to be explained by the fact that the man has submitted himself to a series of successive and graduated inoculations 141 of the venom, a process similar to vaccination, which renders his blood proof against the venom of the particular species of snake, and that one only, used for his performances.
Another deadly snake shown by the snake-charmers in North Africa is the Horned Viper, Cerastes cornutus. The presence of an erect spike above the eye is, however, not a constant character in this snake, and hornless specimens are made to look more formidable by spines of the hedgehog being inserted in the proper place; the illusion is such that even naturalists have been deceived by this trick.
Indian snake-charmers profess to have a belief in the efficacy of snake-stones, or bezoar stones, as a remedy to be applied on the part bitten by a poisonous snake, a belief shared by the natives of many tropical countries. These stones, extracted from various reptiles, birds, and mammals, are calcareous concretions from the stomach or bladder, sometimes composed of superphosphate of lime, sometimes of phosphate of ammonia or magnesia. The value of a bezoar stone being supposed to increase with its size, the larger are sold in India at very high prices.
In many places a popular belief prevails that such stones are found in the heads of snakes. Mr. J. A. Bucknill, now Attorney-General at Hong-Kong, who spent five years in Cyprus, has informed the author 142 that the Viper of the latter island, Vipera lebetina, is commonly believed to contain a stone which, when applied to the bite of a poisonous snake, quickly nullifies the effect; it is also believed that, when this stone is allowed to stand in a glass of water and the water is drunk, it endows the drinker with surprising virility. Indeed, there was an action tried by the English judge at Larnaka in which the plaintiff claimed the return, or damages for the non-return, of one of these “Viper-stones” which he had lent for a monetary consideration to the defendant for the promotion of his manly vigour, and Mr. Bucknill’s recollection is that the plaintiff recovered £10 for the loss.
Skull compact, with short, toothless lower jaw, without transverse bone; palatine and pterygoid reduced and toothless; maxillary small, loosely attached to lower surface of cranium and bearing a few small teeth; no supratemporal, the quadrate articulated to the proötic; a coronoid element in the lower jaw. Rudiments of a pelvic arch, reduced to a single bone. Body vermiform, covered with uniform cycloid scales; head small, not distinct from the body; mouth small, crescentic, inferior; eyes under the more or less transparent head-shields, sometimes entirely hidden. Worm-like, smooth, shiny snakes, of small or very small size, the largest measuring little over 2 feet, of subterranean habits, or found in rotten trees, under stones, or in the saw-dust of sawmills; rarely appearing on the surface except when the ground is soaked by heavy rains.
Inhabit the intertropical parts of the whole world, as well as South Africa, Southern Asia, and Southern 144 Australia. One species occurs in South-Eastern Europe. About 120 species are known.
Head with large shields; nostril in a single or divided nasal. Tail extremely short.
Fig. 14 (after Sordelli)
Form.—Slender, worm-like, the greatest diameter of the body 40 to 52 times in the total length. Tail about as long as broad, ending in a short spine. Snout depressed, rounded, strongly projecting. Eyes distinguishable, appearing as a small black spot surrounded by an unpigmented circle; nostrils lateral.
Head-Shields.—Rostral about one-third, or a little less than one-third, the width of the head, extending on its upper surface nearly to the level of the eyes. Nasal incompletely divided, the cleft proceeding from the second labial. Preocular present, about as broad as the ocular, in contact with the second and third labials. Upper head-scales feebly enlarged and subequal. Four upper labials.

TYPHLOPS VERMICULARIS
After Sordelli

ERYX JACULUS
After Sordelli
145 Scales.—Equal, 22 or 24 round the body.
Coloration.—Brown or yellowish-brown above, yellowish beneath.
Total Length.—10 inches. A specimen from Cyprus is reported to measure 14 inches.
Distribution.—This species has long been known from Greece, the Ionian Islands, and the Grecian Archipelago. It is on record from the Eli-Deren Pass, in Bulgaria. A specimen stated to come from Constantinople is preserved in the British Museum. The range further extends over a considerable part of South-Western Asia, viz., Asia Minor, Syria, Cyprus, Transcaucasia, Persia, Turkestan, and Afghanistan.
Habits.—Pretty alert in its movements, this little snake has considerable constricting powers, and coils itself fast round the fingers when handled. It lives much after the manner of earth-worms, and if dug out of loam or sand a specimen must be instantly grasped, as it draws back with extraordinary quickness. Its food probably consists mainly of earthworms and small insects. Some exotic species of the genus are known to feed on termites, and are often dug out of their nests.
Reproduction.—No observations have been made 146 that I am aware of, but, as some of the exotic species of which we know something more lay large, elongate eggs, it is probable that this species also is oviparous.
Maxillary, palatine, and pterygoid bones movable; transverse bone present; pterygoid extending to quadrate or mandible; supratemporal present, attached scale-like to cranium, suspending quadrate; prefrontal in contact with nasal; a coronoid element in the lower jaw. Teeth in both jaws. Vestiges of pelvis and hind limbs, usually terminating, at least in males, in a claw-like horny spur on each side of the vent.
This family contains, besides the gigantic Boas and Pythons, several small more or less burrowing forms, among which the genus Eryx, its only European representative, belonging to the subfamily Boinæ, characterized by the absence of a supratemporal bone and of premaxillary teeth. This subfamily, the largest members of which inhabit tropical America, is distributed over the hotter parts of America, Asia west of the Bay of Bengal, Madagascar, the Mascarene Islands, Africa north of the equator, Papuasia, and some islands of the South Pacific. The habitat of the European species is confined to the eastern and southern countries of the Mediterranean district.
This very varied family, including terrestrial, 147 arboreal, aquatic, and burrowing forms, is a comparatively small one as regards the number of species, viz., about sixty, of which one-third pertain to the Pythoninæ, which inhabit tropical and South Africa, Southern Asia, Papuasia, Australia, and Mexico.
Anterior maxillary and mandibular teeth longer than the posterior. Head small, not distinct from neck, covered with small scales; a large rostral shield. Eye very small, with vertical pupil. Body cylindrical; scales small; ventral shields narrow. Tail very short; subcaudal shields mostly single.
The range of this genus, embracing eight species, extends from South-Eastern Europe and Africa north of the equator to Central Asia and India.
Form.—Stout. Head small, not distinct from neck; snout projecting beyond the mouth; eye directed upwards and outwards; a feeble mental groove. Tail, ending very obtusely, one-tenth to one-sixteenth of the total length. Anal spurs more or less developed, often absent in the female.
Head-Shields.—Rostral very large and broad, with angular horizontal edge, followed by a pair of internasals and a second row of two or three small shields, 148 the rest of the upper surface of the head covered with scale-like shields, 5 to 8 from eye to eye across the vertex, 7 to 11 round the eye; 9 to 12 upper labials, second or third deepest, separated from the eye by one or two series of scales. Nostril between the internasal and two nasals, the anterior of which sometimes fuses with the former. Two or three series of scales between the nasals and the eye.
Fig. 15 (after Sordelli)
Scales.—Smooth, feebly keeled on the posterior part of the body and on the tail, in 40 to 51 rows. Ventrals narrow, occupying about one-third of the ventral surface, 163 to 200; anal small, entire; subcaudals all or greater part single, 15 to 29.
Coloration.—Pale greyish, reddish, or yellowish-brown above, with brown, purplish-brown, or blackish markings, which may be very irregular or form a single or alternating series of large blotches or cross-bands on the back; the sides with smaller spots; these markings may be confluent and so large as to 149 reduce the ground colour to small yellowish spots; one, two, or three short, dark stripes often present on the nape; a dark streak from the eye to the angle of the mouth; sometimes a dark curved band from eye to eye across the upper surface of the snout. Lower parts yellowish-white, uniform or with small blackish spots.
Size.—21⁄4 feet is the greatest length which this snake is known to attain.
Distribution.—Originally described from Lower Egypt, and extending westwards to Algeria, this Eryx has been found in Greece, in Corfu, in the Cyclades, in Turkey, and in Roumania. It occurs also in Asia Minor, in Transcaucasia, in Transcaspia, in Northern Persia, and in Syria. It has been found at an altitude of 5,000 feet in Persia, to the west of Lake Urmia. A closely allied form (E. miliaris, Pallas), which has been confounded with this species, extends from Transcaspia to Turkestan and Afghanistan.
The reported occurrence of this snake in Bulgaria is based on a specimen labelled “Bulgaria (?)” in the Sofia University Museum. The species is omitted from Kovatscheff’s latest list of Bulgarian Reptiles.
Habits.—This diminutive Boid is a burrower in arid, sandy districts, appearing only early in the morning or towards dusk; it is as a rule more crepuscular than nocturnal. Notwithstanding its rather heavy form, it is capable of very quick movements, 150 darting like an arrow upon its prey, which consists chiefly of small mammals and lizards. A constrictor, like all the members of the family to which it pertains, it crushes its prey before swallowing it. If given several mice at a time, it will catch and kill them all in succession before proceeding to feed. Specimens recently discovered in the Danube Valley in Roumania were found to live in the sand at the bottom of small limestone caves, going about at night and feeding principally on slugs. Unlike other snakes, it is said to lap dewdrops with its tongue. It is a gentle snake, seldom attempting to bite.
Egyptian jugglers are in the habit of implanting the claw of a bird or small mammal in the skin of the head of this snake, above each eye, in order to give it a more formidable appearance.
Reproduction.—Like the other species of Eryx, this snake is ovoviviparous, but, beyond this fact, nothing appears to have been observed concerning the breeding habits, although many examples have been kept in captivity.
Maxillary, palatine, and pterygoid bones movable; transverse bone present; pterygoid extending to quadrate or mandible; supratemporal present, attached scale-like to cranium, suspending quadrate; prefrontal not in contact with nasal; maxillary horizontal, not movable perpendicularly to the transverse 151 bone; no coronoid bone. Teeth in both jaws. No vestiges of pelvic arch.
An enormous group, comprising the great majority of snakes. Divided into three parallel series:
A. Aglypha, with all the teeth solid.
B. Opisthoglypha, with one or more of the posterior maxillary teeth grooved.
C. Proteroglypha, with the anterior maxillary teeth grooved or canaliculated.
The third, which is not represented in Europe, includes some of the most deadly snakes, such as the Cobras, Kraits, Death-adders, etc.
The European genera are thus distributed in the two other series:
Aglypha (Colubrinæ): Tropidonotus, Zamenis, Coluber, Coronella, Contia.
Opisthoglypha (Dipsadomorphinæ): Cœlopeltis, Macroprotodon, Tarbophis.
These genera give but a feeble idea of the variety of forms included in this family, which comprises adaptations to every mode of life for which snakes are fitted.
The distribution of the family coincides with that of the order, extending over the whole world with the exception of the Arctic and Antarctic regions, and Ireland and New Zealand, as well as most of the smaller islands of the Pacific Ocean.
Maxillary teeth increasing in size posteriorly. Head more or less distinct from neck; eye moderate or rather small, with round pupil. Body more or less elongate; scales keeled, with apical pits. Tail moderate.
This large genus, comprising about ninety species, and of almost cosmopolitan distribution, with the exception of South America and the greater part of Australia, may be divided into several subgenera, two of which are represented in Europe—Tropidonotus proper, with the common T. natrix, and Nerodia, Baird and Girard, with two closely related species of more thoroughly aquatic habits, T. tessellatus and T. viperinus.
Form.—Moderately slender; snout short, obtuse, not prominent; eyes and nostrils lateral, the former moderately large. Tail four to six and a half times in the total length.
Head-Shields.—Rostral broader than deep, visible from above. Nasal divided, very rarely semidivided. Internasals at least as broad as long, trapezoid, shorter than the prefrontals. Frontal broader than 153 the supraocular, once and one-third to once and a half as long as broad, as long as or a little shorter than its distance from the end of the snout, shorter than the parietals, not in contact with the preocular. Loreal deeper than long. One (rarely two) pre- and three (rarely two or four) postoculars. Temporals 1 + 2. Upper labials seven (rarely six or eight), third and fourth (or fourth and fifth) entering the eye. Four or five lower labials in contact with the anterior chin-shields, which are shorter than the posterior.
Scales with two apical pits, in nineteen rows, strongly keeled on the body, of outer row smooth or faintly keeled. Ventral shields 157 to 181; anal divided; subcaudals 50 to 88.
Fig. 16 (after Sordelli)
Coloration.—Very variable. We shall first describe the typical form, and then allude to the principal varieties and individual variations with which we are acquainted.
Grey, bluish-grey, olive, or brown, above, usually with black spots or narrow bars on the back, and vertical bars on the sides; upper lip whitish or 154 yellowish, with the sutures between the shields black; the preocular, and sometimes the postoculars, yellow in the young; a white, yellow, or orange collar on the nape, sometimes uninterrupted, more often divided in the middle, bordered behind by two black subtriangular or crescentic blotches, which usually meet on the median line; the bright collar often becomes faint, or even entirely disappears, in large females (Plate II., first figure); belly usually checkered black and grey or white, more rarely grey with small black spots, or entirely black. Iris dark brown or reddish-brown, with a golden circle round the pupil. This is the form found in England and Central Europe and in some parts of Southern Europe.
In Jersey, in the Spanish Peninsula, and in Cyprus, the white or yellow collar, which is always present in the very young, soon disappears, and so does usually the black collar, which is either much reduced or entirely absent (var. astreptophorus, Seoane). Some large specimens from the Spanish Peninsula are uniform olive, without any markings.
Another variation (Plate II., third figure), rare in France, but common in Italy, South-Eastern Europe, and Asia Minor (var. persa, Pallas; bilineatus, Bibr.; murorum, Bonap.) has the collar well marked, though widely interrupted in the middle, and a white, yellow, or orange streak extends along each side of the back, which may bear the usual black markings in addition.
In some specimens from Austria and Corfu (var. 155 subfasciatus, Werner) the belly is white, with black bars occupying the free edge of each ventral shield.
A very remarkable variety (var. cettii, Gené) from Corsica and Sardinia (Plate II., second figure) is grey or olive above, with the black markings confluent into more or less regular annuli, which are nearly as wide as the spaces between them; these annuli are often broken up on the middle line of the back, and alternating; the collar is absent, or is transformed into the first annulus, and the upper surface of the head is more or less spotted or blotched with black. This pattern is most distinct in young and half-grown specimens; in large examples the annuli may break up into spots, disposed with great symmetry in transverse series. The belly is black, spotted with white.
A specimen 20 inches long, from Bona, Algeria (Lataste collection), has the posterior half of the head, from between the eyes and behind the postocular shields, of an intense black, followed by the usual yellow and black collar; two light dots close together on the parietal shields.
Some specimens are entirely or nearly entirely black. In the var. picturatus, Jan, from the Caucasus, the upper parts are sprinkled all over with light dots, and the yellow collar is present; the belly is grey, dotted with black, and with white spots on the sides. In others the body is black above, and checkered black and white beneath (var. scutatus, 156 Pall.), or entirely black (var. ater, Eichw.). This melanism never appears until the second or third year of life, the young being marked like the typical form.
Albinos have occasionally been met with, yellowish flesh-colour with reddish markings, and a white or yellow collar, the eye and the tongue red. Such an albino, from Horsted Keynes, Sussex, is preserved in the British Museum. A remarkable aberration, to be regarded as an imperfect albino, has been found in Dorsetshire, and described as uniform whitish, with a well-defined broad longitudinal central dorsal pale yellow-brown band.
Size.—May reach a length of 6 feet 8 inches. Such giants, females, known from Sardinia, Sicily, and Istria, are, however, very exceptional, individuals of this species seldom exceeding a length of 4 feet. The largest British specimen on record, from Wales, is stated to measure 5 feet 10 inches. Males rarely exceed 3 feet.
Monstrosity.—A dicephalous young, with the two well-formed heads side by side, is preserved in the British Museum, and several others have been described, one being reported to have lived for about a month.

TROPIDONOTUS NATRIX
After Sordelli

T. NATRIX, VAR. CETTII
After Sordelli

T. NATRIX, VAR. PERSA
Distribution.—Tropidonotus natrix occurs all over Europe, with, of course, the exception of Ireland, as far north as the extreme south-east of Scotland, and the sixty-fifth degree in Scandinavia and Finland, 157 and as high up as 7,450 feet in the Italian Alps. With the exception of a few districts in England and in Central Europe, as well as in the extreme north, it is common everywhere, in the north as well as in the south. On the Mediterranean islands it is absent from the Baleares and Malta. In North Africa it is known from Algeria and Tunisia, north of the Atlas, where it does not seem, however, to be at all common. It has a wide range in Asia, extending eastwards to Lake Baikal, and southwards to Cyprus, Asia Minor, and Northern Persia. In the south-east of its range, the bilineated variety predominates over the typical form. The melanistic so-called varieties are not geographically restricted, but occur all over the habitat of the species, though not recorded from England.
Habits.—Although fond of water, and often seen swimming in ponds or streams or creeping by the water’s edge, this snake is far less aquatic than its two congeners described hereafter; it often occurs on dry chalk hills or in woods far from any water. It is moderately agile in its movements, and easily caught, on which occasions it hisses loudly and emits a nauseous smell from its anal glands, together with the renal dejections, but makes no attempt to bite; exceptionally an individual may go so far as to strike with open mouth, but cases of this snake really biting are extremely rare. However, Gené says of the male of his Natrix cettii, “iracundum et 158 mordacissimum animal.” Dr. Gadow relates his experience with aggressive specimens which inhabited a swamp with a little stream to the north of Oporto, close to the coast. To his utter surprise, some of them actually made for him, swimming along rapidly with the head erect, about 6 inches above the water, and darting forwards with widely opened jaws; but they did not bite. According to Professor Kathariner, this snake when caught has been observed to sham death, lying rigid and motionless, with open gape. Some specimens do well in captivity, and are known to have lived for many years; others refuse all food and die of starvation. After a time they become tolerably tame, and cease to produce the offensive odour when handled.
The food consists of frogs and toads—the latter being preferred notwithstanding their poisonous secretion, which protects them from the attacks of most animals—occasionally of newts, seldom of fish; these snakes are reported to have a predilection for tree-frogs, and to feed occasionally on mice and birds, but most observers agree that they will not take anything higher in the zoological scale than frogs. The prey is swallowed alive, and, if not very large, four or five frogs or toads are often taken in succession; a case is known of a snake having swallowed twenty very small frogs at one meal. The young feed on worms and batrachian larvæ, in addition to very small frogs and toads.
159 The Grass-snake gets on very well with the Adder, to whose venom it is immune.
It has more than once been met with swimming in the sea, and a case is reported of one having been captured in the open sea twenty-five miles from the nearest land, no doubt carried away by the current, but still perfectly lively.
The hibernating season is spent in holes in walls or at the root of trees, often under manure-heaps, and the awakening occurs in March or April, soon to be followed by the first exuviation and the pairing.
Reproduction.—Pairing takes place in April or in May, according to the climate, and the eggs are laid between June and August, the young emerging six to ten weeks later. It is probable that a second pairing occasionally takes place in the autumn, as eggs have sometimes been found in manure-heaps at the end of winter. Females do not breed until about 2 feet long, males a little sooner. The eggs number 11 to 48, according to the size of the female, and, after being produced in a string, stick together in a mass, without any regularity.
The eggs measure 1 to 11⁄2 inches in length, and when newly laid are about once and a half as long as broad. They often contain at the time they are produced a more or less developed embryo. They are sometimes laid in recesses in walls, in heaps of sawdust near sawmills, under dead leaves, but preferably in manure, for which purpose females 160 often approach farms during the period of oviposition. Holes near baking ovens at the back of village houses are sometimes selected as breeding resorts. The female rolls herself up, and by violent contortions makes a sort of chamber in the manure, in which she may remain for some days after the eggs have been produced. It is not very unusual for several females to congregate for the purpose of laying, and as many as 1,200 eggs have been found in the same hole. The young on emerging has lost the umbilical cord, and measures 6 to 81⁄2 inches. It often remains for a considerable time, sometimes until the following spring, in the hole or manure-heap in which it was born, feeding principally on worms. Very young specimens are never found in the water.
Form.—Rather slender; head rather long and narrow; snout obtuse, not prominent; eyes and nostrils directed upwards and outwards, the former rather small, the latter somewhat valvular. Tail four to six times in the total length.

TROPIDONOTUS TESSELLATUS

TROPIDONOTUS VIPERINUS
After Sordelli

T. VIPERINUS, VAR. AUROLINEATUS
After Sordelli
Head-Shields.—Rostral broader than deep, visible from above. Nasal often semidivided. Internasals usually as long as broad or longer, subtriangular, truncate in front, as long or nearly as long as the 161 prefrontals. Frontal a little broader than the supraocular, once and a half to twice as long as broad, as long as or a little shorter than its distance from the end of the snout, shorter than the parietals, not in contact with the preocular. Loreal as deep as or deeper than long. Two (rarely one or three) preoculars, with or without a small subocular below; three postoculars, often with one or two suboculars below. Temporals 1 + 2. Upper labials eight (rarely seven, nine, or ten), fourth or fourth and fifth (rarely third or fifth) entering the eye. Five (rarely four) lower labials in contact with the anterior chin-shields, which are shorter than the posterior.
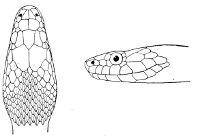
Fig. 17
Scales with two apical pits, in nineteen rows, strongly keeled, of outer row smooth or feebly keeled. Ventrals 160 to 187; anal divided; subcaudals 48 to 79.
Coloration.—Olive, olive-grey, or brown above, with dark spots usually arranged quincuncially or forming narrow bars on the back (Plate III.); sides often with lighter vertical bars; a more or less distinct Λ-shaped dark band on the nape, sometimes produced as a 162 median streak to the frontal shield; upper lip yellowish, with dark bars on the sutures between the shields. Lower parts whitish, yellow, orange, or red, marbled or checkered with black, or nearly entirely black. Iris golden, bronzy, or coppery red.
Some specimens depart very strikingly from the coloration thus briefly defined. We will now mention the principal variations which have been described: Sides of body checkered with black and yellow or black and red (var. rubro-maculosus, Dürigen). With four dark stripes along the anterior part of the back (var. lineaticollis, Werner). Above with four light streaks in addition to the dark markings. Uniform grey or light brown above (var. concolor, Jan, hagenbecki, Werner). Uniform black or blackish (var. nigrescens, De Betta). The most remarkable variety is the var. vosseleri, Werner, from Asia Minor: above with small black and yellowish spots, beneath yellowish with three blackish stripes beginning at some distance from the head, the median much weaker than the outer; the scales are less strongly keeled than in the typical form. There are also specimens with two very regular black stripes along the belly.
A case of chlorochroism, in a specimen from Dalmatia, has been observed by Peracca. The snake was sulphur yellow with black markings; a black band along the belly; iris golden.
An imperfect albino, which has been met with 163 several times in Dalmatia, has been described as var. flavescens, Werner. Yellowish-white or brownish-yellow above, with small blackish spots; belly whitish in the middle, with a series of black spots, bright yellow on the sides; eye and tongue red.
Size.—This snake occasionally reaches a length of 4 feet, but specimens over 3 feet are rare. The largest specimen in the British Museum measures 3 feet 10 inches.
Distribution.—The Tessellated Snake has a wide range in Europe and Asia. It is found south of the Alps, from Liguria to Naples, and eastwards, extending northwards over the greater part of Austria-Hungary, and even as far as Saxony, and again reappears to the west in various localities of the Middle Rhine district (from Bingen to Coblenz and Kreuznach, from Nassau to Lahnstein) and of the Moselle. From Southern Russia it extends into Siberia as far as the Altai, the extreme west of China, and the extreme north-west of India; it is also found in Asia Minor, Transcaucasia, Persia, Mesopotamia, Syria, and the neighbouring parts of Egypt. Italy and the Rhine constitute the western limit of its range in Europe. It does not ascend to any considerable altitude in the mountains of Europe, but it is on record from 6,000 feet elevation in Chitral.
Habits.—This is a far more aquatic species than the preceding, being seldom found in summer away from the water, in which it swims and dives to 164 perfection; which does not prevent it from being equally agile on land. In accordance with these thoroughly aquatic habits, it feeds mostly on fish, although occasionally taking frogs and toads and their tadpoles. Small fish are swallowed in the water, but large ones are landed. This snake does not object to salt water, and it has been observed on the seashore near Odessa, chasing small fish, mostly gobies, in shallow water. Hibernation and pairing take place on land, and it is not until the latter function is accomplished that the snakes of this species resort to the water, which the females leave again for oviposition. Like the Grass-snake, the Tessellated Snake seldom bites.
Reproduction.—Pairing takes place in spring, when large numbers have been observed to congregate for the purpose. As in the Viperine Snake, a second pairing may occur in the autumn, Dr. Werner having found a pair in copula on September 14, at Trebinje, Herzegovina, the female laying her eggs in the following July, which with the beginning of August is the time for oviposition. The eggs measure a little over an inch in length and two-thirds of an inch in width, and number 5 to 25; they are deposited under stones, in the fissures of walls and rocks, or under the refuse of tanneries.
Form.—Moderately slender; head shorter than in the preceding species; snout obtuse, not prominent; eyes and nostrils directed upwards and outwards, the former rather small, the latter somewhat valvular; tail four to six times in the total length.
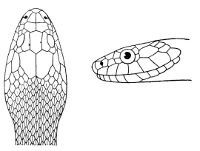
Fig. 18 (after Sordelli)
Head-Shields.—Rostral broader than deep, visible from above. Nasal usually semi-divided. Internasals as long as broad or longer, subtriangular, truncate in front, as long as the prefrontals. Frontal usually broader than the supraocular, once and a half to twice as long as broad, as long as or slightly longer than its distance from the end of the snout, shorter than the parietals, not in contact with the preocular. Loreal as deep as or a little deeper than long. One or two preoculars and two (rarely three) postoculars. Temporals 1 + 2 or 1 + 3. Upper labials seven (rarely eight), third and fourth (or third, fourth, or fourth and fifth) entering the eye. Four (rarely five) lower 166 labials in contact with the anterior chin-shields, which are usually shorter than the posterior.
Scales with two apical pits, in twenty-one (rarely nineteen or twenty-three) rows, strongly keeled, of outer row smooth or feebly keeled. Ventrals 147 to 164; anal divided; subcaudals 46 to 72.
Coloration.—Grey, brown, or reddish above with two alternating series of dark brown or black spots on the back, or with a black zigzag dorsal band (Plate III., second figure), rarely with a single series of black vertebral spots; a lateral series of black spots, usually ocellar, with yellow centres; upper surface of head with dark symmetrical markings; a more or less distinct dark band on the temple, and another on each side of the nape, often edged with yellow in front; upper lip yellow, with dark bars on the sutures between the shields, or dark with a yellow spot on each shield. Lower parts yellow or red, checkered with black, or entirely black; the black of the belly may be connected with the ocellar lateral spots by black vertical bars. Iris golden, often mixed with brown.
A specimen from Ponte Carrega, near Genoa, preserved in the Genoa Museum, is remarkable as being of a dark olive-grey, with three series of black and yellow ocellar spots. It is further exceptional in having the scales in nineteen rows. A second specimen, from the same locality, with the normal number of scales, has some of the vertebral spots 167 ocellar. Specimens with ocellar vertebral spots are found also in Sardinia and in Spain.
As in T. natrix, there occur, in the South of France, in Sardinia, in the Spanish Peninsula, and in North Africa, specimens with two light yellow or reddish lines along the back (Plate III., third figure), in addition to the usual markings (C. aurolineatus, Gervais, T. chersoides, Duméril and Bibron).
Melanism is rare in this species, only one specimen being known, from Nantes in Southern Brittany; uniform black, with the exception of a few white spots on the belly. A remarkable variety (var. incertus, Fatio), connecting this species with the preceding, occurs in Switzerland near Geneva. Not only is its coloration sometimes very similar to that of T. tessellatus, but it agrees with it in the scales being often disposed in nineteen rows instead of twenty-one, and in the presence of eight upper labials, fourth or third and fourth entering the eye; however, the frequent presence of ocellar spots on the sides, and the low number of ventral shields (147 to 151), show that it should be referred to T. viperinus.
Size.—Rarely reaches a length of 3 feet in Europe, the largest specimens being from Sardinia. An Algerian specimen 3 feet 3 inches long is on record.
Distribution.—France as far north as Southern Brittany, the Forest of Fontainebleau, and the Department Aube, the whole of the Spanish Peninsula 168 and the Balearic Islands, Southern Switzerland, north and south of the Alps, Liguria, Piedmont, Corsica, Sardinia, and Sicily. In Africa in Morocco, Algeria, and Tunisia, penetrating into the northern parts of the Sahara.
In Liguria, Piedmont, and Ticino, T. viperinus occurs alongside with T. tessellatus. It reaches an altitude of nearly 4,000 feet in the Alps.
Habits.—Very much the same as in the preceding species, although slightly less thoroughly aquatic, large individuals being sometimes met with at some distance from water. Ponds and marshes are the favourite abode of the Viperine Snake, huge numbers being often found on the borders, diving into the water when disturbed. Frogs and toads, tadpoles, newts, fishes, and large earthworms, are its principal food when adult, the young feeding chiefly on batrachian larvæ, young fishes, and earthworms. A case is known of this snake having eaten a water-shrew (Crossopus fodiens). When a fish has been caught, it is usually eaten on land; in captivity dead fish are rather readily accepted, provided they be quite fresh. Some specimens bite when handled; others are as gentle as the Grass-snake.
For hibernation, hollow trees, fissures in rocks, holes in the ground or in railway embankments, are selected, and numerous individuals sometimes congregate in the same retreat. In the mild winters of the South of Europe they remain quiet, without 169 being torpid, and resume activity very early in the spring.
In the Alemtejo, according to Gadow, when during the rainless and hot summer the small rivers have nearly dried up, these snakes collect in great quantities in the remaining stagnant and muddy pools, and, as the stock of suitable fish gets exhausted, are often reduced to a deplorably emaciated condition. By the month of August they have become so thoroughly aquatic that they cannot be kept alive in dry surroundings for twenty-four hours, apparently dying from some kind of cutaneous suffocation. The same observer once caught a Viperine Snake in a ditch whilst it was swallowing an eel of nearly its own length.
Some specimens show so great a superficial resemblance to the Common Adder, Vipera berus, which, however, being a more northern reptile, very seldom occurs in the same localities—that this snake well deserves its name Viperinus. A celebrated herpetologist, Constant Duméril, was once himself deceived by this resemblance and bitten by a Vipera berus which he had picked up in the Forest of Senart, near Paris, believing it to be a Tropidonotus viperinus; whilst, conversely, a specimen of the harmless snake was killed in mistake for a Viper by no less an expert than Dr. Viaud-Grandmarais.
Breeding.—This snake pairs in March and April, and sometimes again in the autumn; but the eggs 170 are only laid at one season, in June or July, and hatch in August, September, or October. The eggs, numbering four to twenty, are deposited in holes not far from water, often in abandoned galleries of voles or moles. The young at birth measure 4 to 61⁄2 inches, and soon resort to the water, where, unlike those of the Grass-snake, they are frequently met with.
Maxillary teeth increasing in size posteriorly, the two last often separated from the others by a narrow interspace. Head elongate, distinct from neck; eye rather large, with round pupil. One or more subocular shields. Body much elongate; scales smooth, with apical pits. Tail long.
The species of this genus, about thirty in number, are distributed over Europe, North Africa, Asia, and North and Central America. Three inhabit Europe.
Form.—Slender; snout rounded, with distinct canthus, moderately prominent, concave on each side in front of the eye. Tail three and one-third to four and one-third times in the total length.
ZAMENIS GEMONENSIS
Young, after Sordelli

Z. GEMONENSIS, VAR. PERSICUS
After Sordelli

Z. GEMONENSIS, VAR. VIRIDIFLAVUS
Head-Shields.—Rostral a little broader than deep, the portion visible from above measuring one-fourth to two-fifths its distance from the frontal. Frontal 171 more or less bell-shaped, not or but little broader than the supraocular, once and two-thirds to twice as long as broad, as long as or a little longer than its distance from the end of the snout, a little shorter than the parietals. Loreal as long as deep or longer. One preocular (rarely two), extending to the upper surface of the head, but never in contact with the frontal; a small subocular below the preocular; two postoculars (rarely three). Temporals 2 + 2 or 2 + 3 (rarely 1 + 2). Upper labials eight, fourth and fifth entering the eye, fifth and seventh deepest. Five lower labials (rarely four) in contact with the anterior chin-shields, which are usually shorter than the posterior.
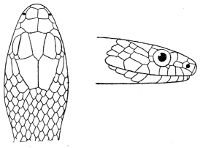
Fig. 19 (after Sordelli)
Scales with two apical pits, in nineteen (rarely seventeen or twenty-one) rows. Ventral shields more or less distinctly angulate laterally, 160 to 230 (usually under 200 in the typical form and the var. caspius, 190 or more in the vars. viridiflavus and asianus); anal divided; subcaudals 87 to 131.
Coloration.—In the typical Z. gemonensis the upper parts are yellowish-brown or pale olive, anteriorly 172 with blackish cross-bars or numerous small black spots, the black scales with a yellowish shaft, the lower parts yellowish-white or pale yellow, rarely more orange; the sides of the head are yellow, the shields edged with blackish. A female, 31⁄4 feet long, from Levico, Trentino, preserved in the Genoa Museum, is uniform reddish-brown above, with mere traces of darker markings on the head and nape. There is every gradation between this form and the var. viridiflavus or atrovirens (Plate IV., third figure), which is dark green or black above, with yellow spots forming transverse series or bars on the anterior part of the body, and longitudinal streaks, following the series of scales, on the posterior part and on the tail; the yellow sometimes predominates over the black, or may appear as a shaft along each dark scale; the preocular and postocular shields are yellow, the labials likewise yellow, with black spots or bars. The lower parts are yellow or greenish-white, with or without black dots, and usually with a series of large black spots on each side.
Some specimens of both the typical form and the var. viridiflavus are entirely black or nearly black. (Z. carbonarius, Bonaparte; Z. sardus, Suckow). In some localities and islands only black specimens occur.
In the var. caspius, Iwan (trabalis, Pallas, Plate V., first figure; persicus, Jan, Plate IV., second figure), from Hungary, Bosnia, Herzegovina, Corfu, Bulgaria, 173 Roumania, Greece, Turkey, Southern Russia, Northern Asia Minor, and North-West Persia, the upper parts are pale olive or reddish-brown, with or without brown or black spots, and each scale bears a yellowish or pale brown longitudinal streak; there is often a dark longitudinal streak on the nape; the belly is uniform orange or red.
Another variety, var. asianus, Boettger, from Asia Minor, Rhodes, Cyprus, and Syria, has the upper parts brown or olive, each scale with a longitudinal light streak, and there are usually large black spots relieved by yellowish shafts; the belly is red, spotted or dotted with black. Melanism is frequent in this form, such specimens being entirely black except on the chin and throat, which are yellow variegated with red.
The very young of the typical form, as well as that of the var. viridiflavus, has a striking livery (Plate IV., first figure), the head and nape black with yellow markings, or olive with black-edged yellow markings, contrasting sharply with the pale olive-grey of the body; the most conspicuous and constant of the yellow markings consist of a bar between the eyes, interrupted on the frontal shield, but sometimes continuous with the yellow of the postoculars, five or six small round spots on the parietal shields, and a V- or W-shaped line just behind the parietals, followed by one or two others separating the dark cross-bars which may be present on the nape, and 174 occasionally even continue some way down the anterior part of the body. This livery persists in some half-grown specimens.
In young individuals from Syria (var. asianus) the head is not differently coloured from the olive-brown body, and the markings described above appear as mere traces; on the other hand, the whole body has black and yellow spots or cross-bars above, and the belly is profusely marked with round black spots.
In the new-born of the var. caspius, of which I have examined only one specimen, 11 inches long, from the Crimea, the head is olive-brown like the body, which bears dark brown spots and narrow cross-bars; and there is a dark brown streak along the middle of the nape, as is sometimes the case in the typical form. The belly is unspotted. A young from Malta is intermediate in its markings between the typical form and this variety.
The young of the so-called black variety are not black at birth, but similar to the normal young of the races to which they belong.
The four principal forms—viridiflavus, gemonensis, caspius, and asianus—are so completely connected that I cannot regard them as more than geographical races or varieties.
Size.—This handsome snake grows to a length of 6 feet, the var. caspius even to 8 feet. I have seen a specimen of this variety, from Salonica, which measures 72⁄3 feet.
175 Distribution.—From the Atlantic coast of Europe to South-Western Asia. The typical form, in its narrowest sense, inhabits the Southern Tyrol, the north-eastern corner of Italy, and the countries to the east of the Adriatic, as far as Greece and Crete. The specimens from France, Switzerland, Italy, Giglio, Montecristo, Elba, Corsica, Sardinia, Sicily, and Malta, are mostly referable to the form known as Z. viridiflavus. Farther to the east the species is represented by the vars. caspius and asianus, of which the distribution has been mentioned above. From Spain, this snake is only on record from Catalonia, not far from the French frontier.
Rare or local in the north of its range (Maine-et-Loire, Vienne, Indre, Sarthe, Haute-Saône, Yonne, Aube, in France, Switzerland north of the Alps), it is one of the commonest snakes in Italy and on the borders and islands of the Adriatic, as well as on practically all the islands of the Mediterranean east of the Baleares. The highest altitudes at which it has been met with are 3,900 feet in the Alps, 4,500 feet in the Balkan Peninsula.
Habits.—The name “Whip-snake,” under which an American representative of this genus (Z. flagelliformis) is known, like that of “Fouet” and “Loucinglant,” which have been bestowed on it in some parts of France, expresses the quick movements with which, when captured, this snake lashes its long, slender tail, at the same time furiously biting the 176 hand that has seized it. The generic term Zamenis, of Greek derivation, alludes to its viciousness, which also accounts for its German name, “Zornnatter.” This snake, occurring in Malta, may well have been the “Viper” which fastened on the hand of St. Paul. Some specimens have been kept for months in captivity without losing their savage temper, hissing and flying with open mouth at anyone approaching the glass walls of their prison; others, on the other hand, become quite tame in a very short time, such as one which I kept for nearly two years. Except when sunning itself on a cold early morning in the spring, this snake is always on the alert, and difficult to capture, uncoiling itself and darting away like an arrow at the least disturbance. It lives in preference among shrubs or on the edges of woods, avoiding damp localities, and females at least appear to have sedentary tastes. Lataste tells us of one, near Bordeaux, which he repeatedly met for over two years within 20 yards of the same spot, a bush between a wood and a meadow, without ever being able to capture it.

ZAMENIS GEMONENSIS, VAR. CASPIUS
After Sordelli
ZAMENIS DAHLII
After Sordelli
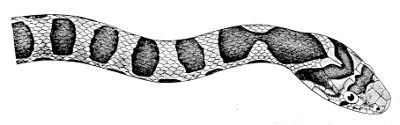
ZAMENIS HIPPOCREPIS
After Sordelli
The food of this snake is very varied, consisting of voles and mice, young birds which it takes from the nests, being a good climber on bushes and low trees, occasionally of frogs, but above all of other reptiles: lizards, slow-worms, and snakes, which it does not attempt to crush before deglutition. It has even been observed in Istria to eat locusts (Acridium ægyptium) and sphyngid moths.
177 Reproduction.—Eggs, laid at the end of June or beginning of July in a well-sheltered hole, are a little over twice as long as broad, and measure 1·2 to 1·4 inches in length. The number of eggs is eight to fifteen according to Fatio, about a dozen according to Tomasini, five according to Werner. The pairing was observed by Schreiber at the end of May, the male and female seizing each other reciprocally by the neck with their jaws; this mode of pairing must not, however, be regarded as the rule in this species, for in other cases observed by Schreiber and by Honnorat the pairs were simply entwined by their coils.
Form.—Very slender; head narrow, snout moderately prominent, obtuse. Tail about one-third of the total length.
Head-Shields.—Rostral a little broader than deep, just visible from above. Frontal not or but little broader than the supraocular, once and two-thirds to once and three-fourths as long as broad, as long as or longer than its distance from the end of the snout, shorter than the parietals. Loreal longer than deep. One preocular, usually in contact with the frontal, with a subocular below it; two postoculars. Temporals 2 + 2 or 2 + 3 (rarely 1 + 2). Upper labials eight or nine, fourth and fifth or fifth and sixth 178 entering the eye. Four or five lower labials in contact with the anterior chin-shields, which are shorter than the posterior.
Scales with a single apical pit, very narrow, in nineteen rows. Ventral shields very distinctly angulate laterally, 205 to 218; anal divided; subcaudals 98 to 132.
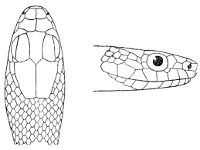
Fig. 20 (after Sordelli)
Coloration.—Olive in front, with a few large black, white- or yellow-edged spots on each side, the anterior of which is sometimes confluent with its fellow and forms a nuchal collar, as in the specimen figured on Plate V.; the greater part of the body and tail uniform pale olive, yellowish, or reddish above, yellowish-white beneath. Head uniform olive-brown above, the labial, preocular, and postocular shields yellowish-white.
Total Length.—3 feet, rarely nearly 4 feet.
Distribution.—Southern Europe east of the Adriatic, as far north as Dalmatia, Asia Minor, Cis- and Trans-Caucasia, North-Western Persia, Cyprus, and Syria. Has also been recorded from Lower Egypt.
Habits.—This snake is even more lively than Z. gemonensis, and does not stand captivity long. It 179 seeks dry, bushy localities, and feeds on small lizards, occasionally on locusts. It does not seem to be very common anywhere in Europe, except perhaps in Dalmatia, whence most of the specimens sold by dealers are imported.
Reproduction.—The pairing has been observed at the end of May. According to Werner, the eggs number usually three only, measuring 11⁄2 inches by 1⁄2 inch.
Form.—Slender; snout obtuse, feebly prominent. Tail one-fifth to one-fourth of the total length.
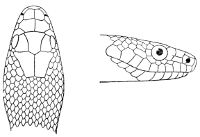
Fig. 21 (after Sordelli)
Head-Shields.—Rostral once and one-third to once and a half as broad as deep, the portion visible from above measuring about one-fourth to one-third its distance from the frontal. Frontal bell-shaped, considerably broader in front than the supraocular, once and one-fourth to once and a half as long as broad, as long as or a little longer than its distance from the end of the snout, shorter than the 180 parietals. Loreal longer than deep, sometimes divided into two. One preocular (sometimes divided into two), in contact with the frontal; two postoculars; a series of three or four suboculars, usually completely separating the eye from the labials. Temporals 2 + 3 or 3 + 3. Eight or nine (rarely ten) upper labials, fifth or sixth very rarely entering the eye. Four lower labials in contact with the anterior chin-shields, which are shorter than the posterior.
Scales with two apical pits, in twenty-five to twenty-nine rows, usually twenty-seven. Ventral shields very distinctly angulate laterally, 222 to 258; anal divided (rarely entire); subcaudals 77 to 107.
Coloration.—Brown, pale olive, reddish, yellow, or orange above, with a dorsal series of large dark brown, black-edged rhomboidal spots, often bordered with yellow, on each side of which is a series of smaller, alternating spots (Plate V.); these spots may become entirely black in the adult, and so large as to reduce the ground colour to a mere network or series of X-shaped pale lines. A dark cross-band between the eyes, and a Λ- or horseshoe-shaped band on the back of the head, which may be confluent with an elongate spot on the nape; a light circle often present in the middle between the parietal shields. The spots often more or less confluent into three longitudinal streaks on the tail. Yellow, orange, or red beneath, with or without black dots, but constantly with a lateral series of black spots, which 181 may be very large or unite with the spots higher up on the sides to form vertical bars.
Size.—Examples 5 feet long are on record; the largest examined by me measures 4 feet 3 inches.
Distribution.—Spain and Portugal, Sardinia, Pantellaria, Morocco, Algeria, Tunisia. Does not reach the North of Spain nor penetrate into the Sahara.
Habits.—This very handsome snake is as a rule as irascible as its European congeners. In Spain as well as in Algeria it is often found about the dwellings of man, occasionally entering houses in search of mice, on which it principally feeds; it is also fond of birds, and, climbing with great facility, plunders the nests of sparrows in towns and villages. It must be regarded as a useful commensal of man, and deserving of protection.
Reproduction.—F. Doumergue found in a hole in a rock near Oran, in September, the recently-laid eggs, five in number and as large as pigeons’.
Maxillary teeth equal or nearly equal in length. Head elongate, distinct from neck; eye moderately large, with round pupil. Body more or less elongate; scales smooth or feebly keeled, with apical pits. Tail moderate or long.
This large genus, embracing close upon fifty species, is represented in Europe, Asia, and North and tropical America. Five species in Europe. Very 182 nearly allied to Zamenis, but distinguished principally by the posterior teeth of the upper jaw not being at all enlarged, and, further, in being, like Coronella, constrictors.
Form.—Moderately slender. Snout obtuse, scarcely prominent. Tail one-sixth to one-fourth of the total length.
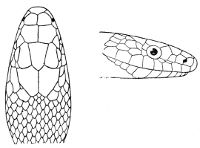
Fig. 22 (after Sordelli)
Head-Shields.—Rostral broader than deep, just visible from above. Frontal once and one-fourth to once and a half as long as broad, as long as its distance from the rostral, shorter than the parietals. Loreal nearly as long as deep, with one or two small shields below it. One preocular, rarely divided, with a subocular below it; two or three postoculars. Temporals 2 + 3 or 3 + 4. Upper labials eight (exceptionally nine), fourth and fifth (or fifth and sixth) entering the eye. Four or five (rarely three) 183 lower labials in contact with the anterior chin-shields, which are longer than the posterior.

COLUBER QUATUORLINEATUS
Young, after Sordelli
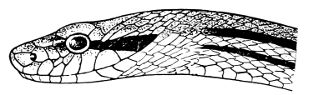
COLUBER QUATUORLINEATUS
After Werner

COLUBER QUATUORLINEATUS, VAR. SAUROMATES
After Sordelli

COLUBER DIONE
After Sordelli
Scales feebly but distinctly keeled, except on the outer rows, with two apical pits, in twenty-five (rarely twenty-three or twenty-seven) rows. Ventral shields not or but very obtusely angulate laterally, 195 to 234; anal divided; subcaudals 56 to 90.
Coloration.—Young (Plate VI., top) with three or five alternating longitudinal series of dark brown, black-edged spots on a yellowish, grey, or pale brown ground, the spots of the median series largest, transversely elliptical or rhomboidal; a dark streak across the forehead, black bars on the labial shields, and a black oblique streak from the eye to the angle of the mouth. In specimens from Italy and the countries bordering the Adriatic (the typical C. quatuorlineatus) the markings very gradually disappear with age, with the exception of the temporal streak, whilst a pair of black streaks appear along each side of the body, at a short distance from the head, the lower corresponding to the postocular streak, the adult being brown without spots, but four-lined (Plate VI., second figure). In more eastern specimens (C. sauromates, Pallas), which may be regarded as representing the original form, the markings of the young persist throughout life, or, if they disappear, they are not replaced by dark streaks (Plate VI., third figure). Lower parts pale yellow, closely spotted or marbled with brown, these markings 184 usually disappearing in the adult, except on the tail. Iris dark brown.
Size.—The largest European snake, stated to reach a length of 8 feet. The largest specimen examined by me measures, however, only 41⁄2 feet.
Distribution.—Aldrovandi’s Snake inhabits Southern Italy and Sicily, Istria, Croatia, Dalmatia, Herzegovina, Greece, and eastwards to Southern Russia, Transcaucasia, Asia Minor, and Persia. It has been observed at an altitude of 2,600 feet in Herzegovina.
All the specimens from Roumania, Bulgaria, Turkey, and eastwards, belong to the var. sauromates, which is regarded by some authors as worthy of specific rank. The reported occurrence of C. quatuorlineatus in various parts of France is certainly due to confusion with C. scalaris and C. longissimus.
Habits.—Dry as well as marshy localities are the abode of this large and handsome snake, which often approaches the dwellings of man, attracted by the poultry. Comparatively slow in its movements, it is more easily captured than any of the other large Colubrids of Europe, and does well in captivity, where it should be provided with a tank, in which it will remain for hours under water. It is as good at swimming as at climbing. Biting readily when captured, it becomes of gentle disposition after a short period of captivity. In consequence of its slow, phlegmatic temperament, it often allows itself to be picked up when surprised in liberty, but as soon as it feels the 185 grasp it turns round and defends itself. It appears to feed exclusively on mammals and birds, up to the size of a rat or dove, and will readily take dead food. It has a predilection for eggs, and has often been observed to swallow hens’ eggs.
Reproduction.—In Herzegovina pairing takes place from the middle of June to the middle of July, and the eggs are laid soon after, to hatch in September or beginning of October. The eggs number six to sixteen, and measure 2 inches by 11⁄3 inches. The young measure 8 to 14 inches at birth.
Form.—Similar to the preceding. Head more convex, a little narrower; snout obtuse, scarcely prominent. Tail about one-fifth of the total length.
Head-Shields.—Rostral broader than deep, just visible from above. Frontal once and one-fourth to once and a half as long as broad, as long as its distance from the end of the snout, shorter than the parietals. Loreal as long as deep, or a little longer than deep. A large preocular, with a subocular below it, the latter very exceptionally absent; two or three postoculars. Temporals 2 + 3 or 3 + 3. Upper labials eight or nine (very rarely seven), fourth and fifth or fifth and sixth entering the eye. Four or five lower labials in contact with the 186 anterior chin-shields, which are nearly as long as the posterior.
Scales smooth or faintly keeled, with two apical pits, in twenty-five or twenty-seven (rarely twenty-three) rows. Ventral shields not or but very obtusely angulate laterally, 172 to 214; anal divided; subcaudals 50 to 80.
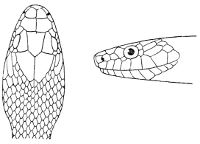
Fig. 23 (after Sordelli)
Coloration.—Pale brown or greyish-olive above, with blackish cross-lines or dark brown or reddish, black-edged spots, and usually two or three more or less distinct pale longitudinal bands; two dark longitudinal stripes on the nape, usually united on the head and terminating on the frontal shield; a curved dark cross-band from eye to eye, and another, oblique, from the eye to the angle of the mouth. Lower parts yellowish, usually dotted or spotted with blackish.
Size.—Seldom exceeds a length of 3 feet. The largest specimen examined by me measures 37 inches.
Distribution.—Across temperate Asia from Asia Minor, Transcaucasia, and the southern border of the Caspian Sea, to the Amur, Corea, and China. 187 In Europe the habitat of this snake is restricted to the steppes of Southern Russia, between the Caucasus and the Lower Ural. The specimen figured on Plate VI. is from Sarepta, on the Volga.
Habits.—This snake frequents arid, sandy localities, and is only exceptionally found in small woods. Nothing more is known of its habits.
Form.—Slender. Snout obtuse, scarcely prominent; head narrow. Tail about one-fifth to one-fourth of the total length.
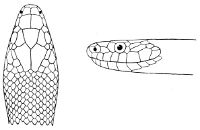
Fig. 24
Head-Shields.—Rostral broader than deep, just visible from above. Frontal once and one-fourth to once and one-third as long as broad, as long as its distance from the rostral or the end of the snout, shorter than the parietals. Loreal as long as deep or longer than deep. One pre- and two postoculars. Temporals 2 + 3. Upper labials 188 eight or nine, fourth and fifth or fifth and sixth entering the eye. Four or five lower labials in contact with the anterior chin-shields, which are as long as or a little longer than the posterior.
Scales smooth or feebly keeled on the posterior part of the body, with two apical pits, in twenty-three (rarely twenty-one) rows. Ventral shields distinctly angulate laterally, 212 to 248; anal divided; subcaudals 60 to 91.
Coloration.—Yellowish-grey to dark olive-brown above, some of the scales with whitish lines on the margins occasionally forming a network; sometimes with a yellowish vertebral stripe or with four darker stripes along the body (var. romanus, Suckow); upper lip, and often also a triangular patch on each side behind the temple, pale yellow; a more or less distinct dark band on the temple, and a vertical dark bar below the eye (Plate VII., first figure). Lower parts uniform pale yellow. Young (second figure) with dark brown dorsal spots, forming four to seven longitudinal series, a Λ-shaped black marking on the nape behind the yellow nuchal blotches, which are brighter than in the adult, a dark brown bar across the forehead, and a black vertical line below the eye; belly greyish or yellowish-olive. Iris dark grey or brown. Tongue pinkish-brown.

COLUBER LONGISSIMUS

COLUBER LONGISSIMUS
Young, after Sordelli

COLUBER LEOPARDINUS
After Sordelli
C. LEOPARDINUS, VAR. QUADRILINEATUS
After Sordelli
Melanism is rare in this snake. Such specimens are entirely black above and beneath (var. niger, 189 Nikolsky), or blackish-grey to black above, dark grey beneath (var. subgriseus, Werner), the angular line on each side of the belly often remaining light. An albino found near Vienna has been described as pale orange-yellow above, with small white spots; pupil and tongue red.
Size.—Grows to 6 feet. Specimens over 41⁄2 feet are, however, very rarely met with.
Distribution.—Generally distributed over the greater part of Austria, Italy, with Sardinia and Sicily, and the whole of South-Eastern Europe, this snake has a very broken range in France, Switzerland, Germany, and is found, quite isolated, as far north as Denmark and Poland. According to Segerus, quoted by Lacepède, it used to be quite common near Copenhagen at the end of the eighteenth century, but it is now much rarer. Its northern limit in France is in Southern Brittany, the Department Orne, and the Forest of Fontainebleau; in Germany, Schlangenbad, near Wiesbaden, perhaps also Baden-Baden and Treves. It is on record from Southern Spain. Its discontinuous distribution in Central Europe, and its presence in various localities near former Roman thermal stations, has been ascribed to its introduction from Italy as an inmate of the temples erected to Æsculapius; but I am more inclined to look upon its sporadic occurrence in the North as the indication of a once more widely distributed species 190 now in process of extinction over part of its range.
In Asia the Æsculapian Snake is only found in Transcaucasia. It occurs in the mountains as well as in the plain, being recorded from 5,200 feet altitude in the Tyrol, 3,200 feet in the Apennines.
Habits.—The Æsculapian Snake lives in woods; among shrubby vegetation; in meadows, where it is often found under haystacks; occasionally about old walls. It climbs well, and often ascends trees. Although a good swimmer, it seldom enters the water of its own accord. It feeds chiefly on small mammals, occasionally on birds and their eggs, and lizards. Specimens which I kept in confinement fed on mice only, refusing sparrows and lizards. Very savage when fresh caught, most individuals soon become tame, and like being handled by people to whom they are accustomed, although still resenting the intrusion of strangers. However, this snake never becomes so thoroughly domesticated as the Smooth Snake, and cannot be trained to take food from the hand, according to R. Rollinat, who has devoted many years to experiments on the taming of reptiles. This observer had no difficulty in feeding his Æsculapian Snakes on mice and voles placed dead in their cage.
This snake is particularly sensitive to cold, and does not emerge until late in the spring from the vole galleries and hollow trees which constitute its 191 winter-quarters. It also avoids excessive heat, never showing itself in the daytime during the hotter months in the South of Europe.
Reproduction.—Pairing takes place between the middle of May and the middle of June. The eggs are laid towards the end of June or in July, in holes in walls or hollow trees, under moss, sometimes even in the dung-heaps of farms, and hatch in September. According to trustworthy observers, the eggs, which measure 11⁄2 to 2 inches in length, and less than 1 inch in width, number only five or six, rarely up to eight.
The young on emerging are highly suggestive of young Grass-snakes in colour and markings, as well as in their much less slender shape as compared with the adult. They measure about 5 inches, and are at once most ready to bite.
Form.—Slender. Snout obtuse, scarcely prominent. Tail about one-fifth of the total length.
Head-Shields.—Rostral broader than deep, just visible from above. Frontal once and one-third to once and a half as long as broad, as long as its distance from the end of the snout, shorter than the parietals. Loreal longer than deep. One pre- and two postoculars. Temporals 1 + 2 or 2 + 3. Upper labials 192 eight (rarely seven), fourth and fifth (rarely third and fourth) entering the eye. Four or five lower labials in contact with the anterior chin-shields, which are longer than the posterior.
Scales smooth, with two apical pits, in twenty-five or twenty-seven rows. Ventral shields rounded, not angulate laterally, 222 to 260; anal divided; subcaudals 68-90.
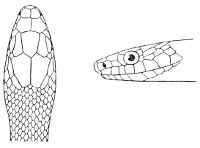
Fig. 25 (after Sordelli)
Coloration.—Typical form (Plate VII., third figure) greyish or pale brown above, with one dorsal series of dark brown, reddish-brown, or bright red, black-edged transverse spots and a lateral alternating series of smaller black spots with or without lighter centres; usually a [Y]-shaped dark marking on the occiput and nape; a crescentic black band from eye to eye across the prefrontal shields, an oblique black band from behind the eye to the angle of the mouth, and a black spot or vertical bar below the eye. Lower parts white, checkered with black, or nearly entirely black. Iris reddish-golden.
In some specimens (var. quadrilineatus) the dorsal spots are replaced by two brown or red, black-edged stripes bordering a pale greyish or yellowish vertebral 193 stripe (Plate VII., fourth figure); such specimens are so coloured from birth. This colour variety, which is so strikingly different from the typical form, is connected with the latter by the var. schwoederi, Werner, in which the spots form two vertebral series, and the var. elsneri, Werner, in which the light vertebral band is broken up by dark transverse bars, producing a ladder-like pattern.
Size.—Rarely exceeding a length of 3 feet.
Distribution.—Southern Italy, Sicily, Malta, Istria, Dalmatia, and other parts of the Balkan Peninsula, Grecian islands, Crimea, Asia Minor. The altitudinal range does not extend beyond 1,600 feet.
Habits.—This is not only the prettiest European snake as regards its markings, whether in the form of spots or of stripes, but also the most graceful in its movements. Unless compelled to fly for safety, there is something slow and deliberate in its behaviour which is more suggestive of Coronella than of most other species of Coluber. It is fond of climbing, and if the terrarium in which it is kept be provided with a bush or small tree, it will spend most of the time gracefully coiled round the branches. Usually very savage when fresh caught, some specimens become quite tame in captivity. In Dalmatia, where it is not uncommon, this snake is found principally among prickly shrubs, in hedges, or on old walls. It awakens from its winter slumber later than other South European snakes. Although occasionally 194 taking lizards, its principal food consists of mammals and birds, which are killed before being devoured, the Leopard Snake being, like the other members of the genus Coluber, a constrictor.
Reproduction.—According to Werner, the eggs, two to five in number, are deposited in midsummer; they are remarkably elongate: 21⁄2 inches long, 2⁄3 inch broad.
Form.—Moderately slender. Snout pointed, strongly projecting beyond the mouth. Tail one-sixth to one-fifth of the total length.
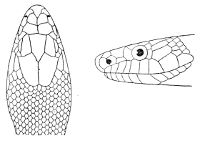
Fig. 26 (after Sordelli)
Head-Shields.—Rostral deeper than broad, forming an acute angle above, wedged in between the internasals, the portion visible from above nearly as long as its distance from the frontal. Frontal about once and one-third to once and a half as long as broad, as long as or shorter than its distance from the end of the snout, nearly as long as the parietals. Loreal longer than deep. One pre- and two or three 195 post-oculars. Temporals 2 + 3 or 2 + 4. Upper labials seven or eight (rarely nine), fourth or fourth and fifth (or fifth and sixth) entering the eye. Four or five lower labials in contact with the anterior chin-shields, which may be either longer or shorter than the posterior.
COLUBER SCALARIS
After Sordelli
Scales smooth, with two apical pits, in twenty-seven (rarely twenty-five or twenty-nine) rows. Ventral shields not angulate laterally, 201 to 220; anal divided, rarely entire; subcaudals 48 to 68.
Coloration.—Young yellowish-grey, or pale brown, above, with a series of regular H-shaped black or blackish-brown markings along the back, forming a ladder-like pattern—whence the name scalaris—and small black spots on the sides; a V-shaped black marking on the snout, a black oblique streak from the eye to the angle of the mouth, and a black spot below the eye; belly yellow, spotted or checkered with black or nearly entirely black. These dorsal markings disappear in the adult, and are replaced by a pair of brown stripes running along the back (Plate VIII.); the belly loses the black markings, and becomes uniform yellow. Iris dark brown.
Size.—Grows to a length of 31⁄2 feet, exceptionally 41⁄2 feet.
Distribution.—The Mediterranean coast of France, Spain and Portugal, and Minorca. Its occurrence in Algeria is very doubtful.
Habits.—Not uncommon near the coast in France, 196 in hedges and vineyards, often climbing on shrubs. In the Spanish Peninsula, according to Boscá, it is common in forests and on the sheltered side of valleys, under stones or in holes in the ground. A specimen I kept alive for a short time showed a more furious temper than I have ever witnessed in any snake, repeatedly flying with open mouth against the glass of its cage whenever I entered the room in which it was kept. Other specimens are reported to have become quite tame after a certain time. It is one of the quickest of European snakes, one of the most difficult to catch; it is a good climber. The food consists of mice, birds, and lizards; the young are said to occasionally eat grasshoppers.
Reproduction.—According to J. von Fischer, the eggs, nine in number, are deposited twenty-five days after the pairing, which takes place in May or June, and measure about 2 inches by 2⁄3 inch.
Maxillary teeth increasing in size posteriorly. Head not or but slightly distinct from neck; eye rather small, with round pupil. No subocular shields. Body moderately elongate; scales smooth, with apical pits. Tail moderate.
CORONELLA AUSTRIACA
After Sordelli
This genus, embracing about twenty species, is represented in the different parts of the Northern Hemisphere, extending a little beyond the Equator in East Africa. Two species are European.
Form.—Moderately slender; snout more or less prominent, sometimes decidedly pointed; tail one-fourth (males) to one-sixth (females) of the total length. The considerable differences to be observed in the shape of the snout are merely individual, specimens with more prominent snout and a corresponding development of the rostral shield (C. italica, Fitz., fitzingeri, Bonap.) occurring over the greater part of the range of the species.
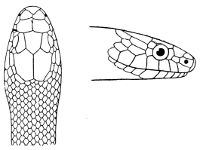
Fig. 27 (after Sordelli)
Head-Shields.—Rostral at least as deep as broad, more or less produced posteriorly between the internasals, the portion visible from above at least half as long (in some specimens quite as long) as its distance from the frontal, rarely separating the internasals. Frontal once and one-fourth to once and a half as long as broad, much broader than the supraocular, as long as or longer than its distance from the end of the snout, shorter than the parietals, widely separated 198 from the preocular. Nasal rarely undivided; loreal longer than deep. One (very rarely two) pre- and two postoculars. Temporals 2 + 2 or 2 + 3 (very rarely 1 + 2). Upper labials seven (rarely eight), third and fourth (or fourth and fifth) entering the eye. Four lower labials (rarely three) in contact with the anterior chin-shields, which are as long as or longer than the anterior.
Scales with one or two apical pits, the pit usually single on the back and paired on the sides, in nineteen (rarely twenty-one) rows.[2] Ventral shields 153 to 199; anal divided (rarely entire); subcaudals 41 to 70.
Coloration.—Grey, brown, or reddish above, with small blackish, dark brown, or brick-red spots usually disposed in pairs, sometimes forming cross-bars; sometimes with one or three lighter stripes; one or two black dots precede on each scale the single or paired apical pit; frequently two blackish, dark brown, or brick-red stripes on the nape, usually confluent with a large dark blotch on the occiput; the top of the head occasionally nearly entirely blackish, especially in the young; a dark streak on each side of the head, from the nostril to the angle of the mouth, passing through the eye, sometimes extending along the side of the neck or even of the whole body. Lower parts red, orange, brown, grey, or black, uniform or speckled or closely spotted with black and white, the sides often lighter (Plate IX.).
199 A colour variety, of which I have examined a single specimen from near Vienna, is pale brown above, with four black lines along the anterior part of the body, and two small, yellowish, dark-edged spots close together on the back of the head, separated by the suture between the parietal shields.
Werner has described another variety, also from near Vienna, which resembles Coluber leopardinus, having two series of large, brown, dark-edged spots along the back, some of the spots alternating, others uniting across the back. Apparently very similar to the last variety, and also said to be suggestive of Coluber leopardinus, is the var. scalaris, Sternfeld, from Lüneburg in Hanover, reddish-brown above, with two rows of bright red, black-edged spots, partly confluent and connected across the spine by transverse bars producing a ladder-like pattern. Specimens of a uniform greyish-brown, without any markings, are very rare. The var. veithi, Schreiber, established on a single specimen from Carinthia, represents a case of melanism: bluish-black, with the normal markings of an intense black. Two specimens of a “black variety” are said to have been found in this country, near Poole.
Size.—Seldom exceeds a length of 2 feet, and in many districts, in England for instance, does not appear to often reach that size. The largest specimen, from Austria, examined by me, measures 25 inches; one from Hampshire measures 24 inches.
200 Distribution.—The range of the Smooth Snake extends over nearly the whole of Europe, as far north as 63° in Norway; it becomes rare and more local in the south, being absent from part of Spain and the islands of the Mediterranean, with the exception of Sardinia. It is common in the hilly parts of Belgium, Northern and Central France, Germany, and Austria. In Sweden it appears to be restricted to the oak region. In Great Britain it has been found in four counties in the South of England: Surrey, Hampshire, Dorsetshire, and Berkshire, in some parts of which it is less uncommon than usually supposed. Its reported occurrence in Dumfriesshire is the result of an error; the snake figured as Coluber dumfriesiensis represents an American species. In a very interesting article written for Science Gossip in 1888, Mr. A. L. Beldy says that about 1868, when Bournemouth was but a very small village, surrounded by large expanses of moorland, Coronella austriaca was extraordinarily abundant, and during a hot summer examples were to be seen literally in scores and great numbers were killed. Since then, however, their numbers have gradually decreased. About 1880 the snake was occasionally found near Wellington College, Berks, and as many as five were captured by one person in the course of one year; it is believed to be now extinct in that neighbourhood. From South-Eastern Europe the range of this species extends to South-Western Asia. 201 The ascertained altitudinal range is 4,000 feet in the Alps, 6,000 feet in Bosnia, and 6,500 feet in the Caucasus.
Habits.—The Smooth Snake lives on heathland, stony wastes, and wooded hills, showing a preference for dry localities. Although not infrequent on the Dorsetshire and Hampshire heaths, where it was first discovered in 1853, it was not recorded as a British reptile until 1859; it was discovered much later on the sandy heaths between Haslemere and Farnham, where it occurs in small numbers, and in Berkshire. These localities are likewise inhabited by the rarer British lizard, the Sand Lizard. Notwithstanding its gentle, timid appearance, this snake when fresh caught is usually very ready to bite; either it snaps angrily, or, without hissing or other warning, it suddenly fastens its jaws into the finger of its captor, even if it be gently handled. The food consists mostly of lizards, occasionally of slow-worms or small snakes, more rarely of voles or mice, even shrews, which are seized, constrictor-like, and crushed by the coils of the body. O. von Tomasini has observed one swallowing a Coluber longissimus as large as itself.
In Central Europe this snake becomes active towards the end of March or beginning of April, and retires in September or October. It does well in captivity, and becomes very tame. It is one of the most intelligent of snakes, second to none in educability; 202 it can be trained to feed in the hand of its master.
Reproduction.—The Smooth Snake pairs in early spring, and is ovoviviparous. The young, two to fifteen in number, are born late in August or in September, enveloped by a thin membrane which they tear immediately; they measure 5 to 6 inches. Embryos 31⁄2 or 4 inches long have the scaling and the characteristic markings fully developed, but the scales and shields much abbreviated, the former broader than long. A dicephalous young is preserved in the Bosnian Museum at Sarajev, and another was caught near Karlsruhe, in Germany, in 1881, and kept alive for some time. According to Rollinat, a second autumnal pairing sometimes takes place in France.
Distinguished from the preceding by a somewhat more slender form, a more obtuse, scarcely prominent snout, a much lower rostral shield, which is considerably broader than deep and just visible from above, not penetrating between the internasals, constantly eight upper labials, fourth and fifth entering the eye, and the scales in twenty-one (rarely nineteen or twenty-three) rows. Ventrals 170 to 200; anal divided; subcaudals 49 to 72.
CONTIA MODESTA
After Sordelli
CORONELLA GIRONDICA
After Sordelli
Coloration.—Brown, greyish, yellowish, or reddish above, with dark brown or black spots or transverse bars, sometimes with four dark stripes in addition; dark dots in front of the apical pits as in the preceding species; a pair of elongate dark spots or a U-shaped marking on the nape; a dark streak from the eye to the angle of the mouth, and a dark cross-bar from eye to eye, across the prefrontal shields; a dark line below the eye. Lower parts yellow, orange, or coral red, with large, mostly quadrangular black spots, often arranged in chess-board fashion, or with two series of black spots (Plate X.), which may be confluent into two longitudinal bands.
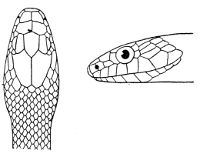
Fig. 28 (after Sordelli)
Total Length.—26 inches.
Distribution.—South of France (as far north as the Charente-Inférieure to the west, the Dauphiné to the east), the whole of Spain and Portugal, Southern Tyrol, Italy, and Sicily. It has not been recorded from higher than 2,500 feet in the Alps. Rare in Northern Morocco and Algeria. In many localities in Europe it occurs alongside with C. austriaca.
204 Habits.—All that is known to me of the habits of this close ally of the preceding species is derived from the works of Bonaparte, Gené, and Schreiber, and from a note by Gachet, who observed it near Bordeaux and described it under the name of Coluber rubens. According to these authors, it frequents dry and rocky localities as well as old walls, in which it finds a refuge and a good supply of the lizards on which it feeds. A large specimen from Albano, near Rome, preserved in the Genoa Museum, had swallowed a full-grown Chalcides tridactylus. This Coronella is crepuscular, rarely showing itself in the daytime, leaving its retreat only after sunset, and has been observed to crawl about by moonlight. Its movements are slow, which accounts for crushed specimens being often met with on paths or roads. Contrary to the rule in C. austriaca, it is extremely gentle, seldom attempting to bite.
Reproduction.—Whether this species is ovoviviparous, like its European congener, has not, I think, been ascertained. All we know on this matter is that a female found dead on a road near Bordeaux by M. Lataste at the end of June contained eggs which showed no trace of embryos. This does not, however, settle the question, as the young would not be born until at least two months later. According to Gené, pairing takes place in May, when specimens have been observed to congregate in considerable numbers.
Maxillary teeth subequal. Head not or but slightly distinct from neck; eye moderate or rather small, with round pupil. Nasal single; no subocular shields. Body moderately elongate; scales smooth, with apical pits. Tail moderate.
This genus, with certain modifications in the above definition, is made to embrace about twenty-five species from South-Western Asia and Sind and North and Central America. One of the species inhabiting Asia extends into a very small part of Europe.
Form.—Moderately slender. Head small, quite flat above; snout obtuse, feebly prominent. Length of tail four to five times in the total length.
Head-Shields.—Rostral a little broader than deep, visible from above. Suture between the internasals as long as or a little shorter than that between the prefrontals. Frontal once and a half to once and two-thirds as long as broad, as long as or longer than its distance from the end of the snout, shorter than the parietals, as broad as or a little broader than the supraocular, widely separated from the preocular. Nostril in the middle or upper part of the nasal. Loreal square or longer than deep. One 206 (rarely two) pre- and two (rarely one) postoculars. Temporals 1 + 2; parietal sometimes nearly touching the fifth upper labial. Upper labials seven, third and fourth entering the eye. Four (rarely five) lower labials in contact with the anterior chin-shield; posterior chin-shields smaller than the anterior, and separated from each other by one or two rows of scales.
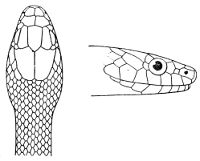
Fig. 29 (after Sordelli)
Scales with a single apical pit, in seventeen rows. Ventral shields 150 to 191; anal divided; subcaudals 53 to 78.
Coloration.—Not unlike that of a young Zamenis gemonensis. Greyish-olive above, uniform or each scale lighter in the centre. The greater part of the upper surface of the head behind the snout, together with the nape, black in the young, with a yellow cross-bar or a pair of yellow spots between the eyes, the bar sometimes confluent with the yellow postoculars, and a horseshoe-shaped band of the same colour on the temples and across the occiput (Plate X.); the black of the nape again edged with yellow behind. More or less distinct traces of these markings are preserved in adult specimens. Upper lip yellowish, with black spots or bars on 207 the sutures between the shields. Lower parts uniform white or yellowish.
In the var. semimaculata, Boettger, from Chios, small dark spots are scattered over the upper parts of the anterior half of the body.
Size.—This snake rarely reaches a length of 19 inches. It is the smallest Colubrid of Europe.
Distribution.—The Caucasus up to about 5,000 feet, Asia Minor, Chios, Cyprus, Syria, Mesopotamia, and North-Western Persia. The northern slope of the Caucasus appears to be the only part of Europe included in its habitat. The British Museum possesses two specimens labelled as from Constantinople, but the presence of this species in European Turkey requires confirmation.
A closely allied species, which has been confounded with C. modesta, C. collaris (Ménétriès), and which also inhabits the Caucasus without having been recorded from the northern slope, is distinguished by having the scales in fifteen rows (very rarely seventeen), and the posterior chin-shields in contact with each other.
Habits.—Nothing is known as regards this species, but the North American members of the genus Contia are chiefly insectivorous and oviparous.
Maxillary teeth small and subequal, followed after a short interspace by one or two very large grooved 208 fangs situated below the posterior border of the eye; anterior mandibular teeth strongly enlarged. Head not very distinct from neck, with angular canthus rostralis and projecting supraocular; eye large, with round pupil; nostril a crescentic slit in a single or divided nasal. Body elongate; scales smooth, more or less distinctly grooved longitudinally in the adult, with apical pits. Tail moderately long.
The range of this genus, which comprises only two species, extends over Southern Europe, South-Western Asia, and North Africa.
Form.—Slender; head elongate, narrow, concave above on the snout and between the eyes; snout projecting, rounded, with raised canthus and concave loreal region. Tail about one-fifth to one-fourth of the total length.

CŒLOPELTIS MONSPESSULANA
After Sordelli

MACROPROTODON CUCULLATUS
TARBOPHIS IBERUS
After Sordelli

TARBOPHIS FALLAX
Head-Shields.—Rostral nearly as deep as broad, just visible from above. Internasals much shorter than the prefrontals. Frontal very narrow, twice to twice and a half as long as broad, its width in the middle not more than half that of the supraocular, widening in front and extending beyond the supraoculars to join the preoculars, longer than its distance from the end of snout, as long as or a little longer 209 than the parietals. Two loreals. One preocular, the upper portion of which is much enlarged, and encroaches upon the area occupied in other snakes by the prefrontal and the supraocular; two (rarely three) postoculars. Temporals 2 + 3 or 4. Upper labials eight (rarely nine), fourth and fifth (or fifth and sixth) entering the eye. Four or five lower labials in contact with the anterior chin-shields, which are as long as or shorter than the posterior.
Scales with single apical pits, in seventeen or nineteen rows, longitudinally grooved in the adult, less distinctly in the young. Ventral shields 160 to 189; anal divided; subcaudals 68 to 102.
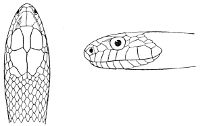
Fig. 30
Coloration.—The young is elegantly marked with dark brown and yellowish-white on a pale brown ground. On the head, the principal dark markings usually are an oblique band on the posterior half of the supraocular shield, and another, or a large spot, on the parietal, sometimes produced backwards, and forming with its fellow a Λ-shaped band, separated from a large occipital blotch by a yellowish space; 210 anterior half of the frontal shield and shields on the snout edged with dark brown; a dark streak, sometimes broken up into small spots, on the temporal region; yellow spots on the pre- and post-oculars; lips brown, with large, yellow, black-edged spots, or yellow with brown spots; chin with three brown longitudinal streaks. Back with a vertebral series of large roundish dark spots or narrow cross-bars; small spots on the sides, these sometimes forming longitudinal series or accompanied by yellowish streaks or dots; these markings often confluent into three longitudinal streaks on the tail. Belly pale brownish, greyish, or reddish, with numerous pale spots, sometimes with a dark brown line on each side. The adult is greyish, reddish-brown, or olive above. Some specimens preserve more or less the markings of the young, and the dark dorsal markings (Plate XI.) may be edged with yellowish and ocellar in appearance (var. insignitus, Geoffroy); the belly is yellowish, with small dark spots which usually form longitudinal series, and may be confluent into streaks. A variety common in Dalmatia (neumayeri, Fitzinger) is brown or olive above, without spots, sides with a bluish-grey lateral band, the scales on which are edged with black, the belly uniform yellow. Other specimens are brown or reddish, with light edges to the scales on the sides, or with yellowish lateral lines, or dark olive or dark brown above and black on the sides, each scale with a yellowish central spot; in the last-mentioned 211 the second third of the back may be almost entirely black, and the belly dark olive-grey in the middle and yellowish on the sides. Iris brown, with a golden or coppery circle round the pupil.
Size.—This handsome snake grows to a length of 61⁄2 feet. Specimens 5 to 6 feet long are not uncommon.
Distribution.—Mediterranean coast of France and Western Liguria, Spain and Portugal, Sicily, Lampedusa, eastern coast of the Adriatic, Greece and eastern islands of the Mediterranean, Mediterranean coast of Asia and Sinaitic Peninsula, eastwards to the Caucasus and Persia, North Africa from Egypt to Rio de Oro. It is not known to occur above 2,300 feet altitude in Europe.
Habits.—A lively, swift snake, living on land and on low bushes, often found near human habitations. Some specimens are very vicious, whilst others show a gentle disposition after a short period of captivity. A specimen nearly 6 feet long, which I kept for some time, never attempted to bite when handled, and some have become so tame as to take food from the hand. The sense of sight appears to be better developed than in any other European snake. The food consists chiefly of mammals, even large rats and young rabbits, birds such as chickens, partridges, and quails, lizards, and other snakes, which, if of considerable size, are not swallowed until paralyzed or killed by the effect of the poison. In Eastern 212 Europe, Vipera ammodytes is said to be the principal enemy of Cœlopeltis, and the two snakes are consequently seldom found together in the same locality.
Many experiments have been made on the action of the poison of this Opisthoglyph. Peracca and Deregibus, as well as, later, Phisalix, found a striking similarity with the symptoms of Cobra poison in their experiments on small animals, the suspension of the respiration occurring in a few minutes, the blood being otherwise unaffected. It has been stated by some authors that Cœlopeltis poison has little or no action on man, but a French zoologist, E. Taton-Baulmont, having been bitten in the index-finger by a four-foot-long specimen at Algiers, the swelling extended within thirty hours up to the shoulder, and was accompanied by fever and nervous troubles. As a rule, however, the bite of this snake has no poisonous effect on man, from the fact that the fangs conveying the venom are situated so far back in the mouth as not to come into action.
Reproduction.—According to Werner, the eggs, four to twelve in number, are laid in July, and measure 2 inches in length and 1⁄2 inch in width.
Maxillary teeth few and very unequal in size, fourth and fifth or fifth and sixth enlarged and followed by an interspace, the two last teeth fang-like and grooved, situated just behind the eye; sixth mandibular tooth 213 fang-like, and separated from the remainder by an interspace. Head slightly distinct from neck; eye rather small, the pupil vertically elliptic or subelliptic when contracted. Body moderately elongate; scales smooth, with apical pits. Tail moderate or rather short.
A single species.
Form.—Very similar to the Smooth Snakes, with which it has been confounded, but snout broader and very strongly depressed. Tail five and a half to six and a half times in the total length.
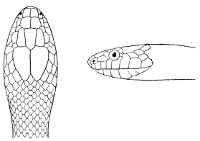
Fig. 31
Head-Shields.—Rostral at least twice as broad as deep, not or but scarcely visible from above. Internasals as long as or a little shorter than the prefrontals. Frontal not much broader than the supraocular in the adult, once and a half to twice as long as broad, as long as or longer than its distance from the end of the snout, shorter than the parietals. Nasal usually semidivided. Loreal once 214 and a half to twice as long as deep. One preocular, extending to the upper surface of the head, but not reaching the frontal; two (rarely one or three) postoculars. Temporals 1 + 2. Upper labials eight, fourth and fifth entering the eye, sixth usually in contact with the parietal. Four or five lower labials in contact with the anterior chin-shields, which are as long as or a little shorter than the posterior.
Scales with mostly single apical pits, the pits sometimes paired on the sides of the body, in twenty-one or twenty-three rows (nineteen to twenty-five in North African specimens). Ventral shields 153 to 192; anal divided; subcaudals 40 to 54.
Coloration.—Pale brown or greyish above, with small dark brown or blackish spots or with more or less distinct darker and lighter longitudinal streaks. Upper surface of head with dark brown vermiculations; a dark brown or black, often light-edged occipito-nuchal band, extending downwards to the gular region and produced forwards into a point to between the parietal shields; a dark brown or black streak on each side of the head from the end of the snout, through the eye, to the last lower labial shield, traversing the four last upper labials, which are yellowish above and below the streak (Plate XI.). Lower parts yellow or coral red, with black spots, which may form a tessellated pattern, two longitudinal series, or be so crowded as to fuse into a band along the middle of the belly and tail.
215 The above description is taken from Spanish specimens (Badajos, Algeciras, Andalucia), but the variations are very great when we take North Africa into consideration. The nuchal band may be narrow or broken up into spots, the median of which sometimes forms a longitudinal streak, or so much enlarged as to fuse with the dark markings on the upper surface of the head; in some specimens (from Morocco and Algeria) the upper surface of the head and the nape may be entirely ink black, or the whole head black above and beneath with the exception of a whitish streak bordering the upper lip. The dark streak from the eye to the angle of the mouth may be absent, or reduced to a short oblique streak below the eye. Irrespective of the variations in the markings of the upper parts, the lower parts may be more or less spotted with black, or immaculate.
Some specimens of this small snake bear a general resemblance to Coronella girondica, with which Macroprotodon has sometimes been confounded. But a careful examination of its whole structure shows it to be more affine to Cœlopeltis and Tarbophis, the other European representatives of the Opisthoglyphous Colubrids.
Size.—The largest European specimen examined measures 171⁄2 inches. Specimens up to 22 inches long occur in Algeria and Tunisia.
Distribution.—In Europe this snake is only known 216 from Spain (Estremadura, New Castille, Andalucia), Portugal (Alemtejo), the Balearic Islands (Majorca and Minorca), and the island of Lampedusa. In North Africa it is generally distributed from the north coast of Egypt to the Rio de Oro; in Algeria it penetrates into the northern parts of the Sahara. The specimen figured on Plate XI. is from Algeciras.
Habits.—Appear to be similar to those of Coronella girondica. Crepuscular in its habits, it is usually found under stones or in burrows in the ground. Unless pursued, when it darts off with great rapidity, its movements are slow. It is very ready to bite, but no experiments have been made on the effects of its poison. The food consists chiefly of small lizards.
Reproduction.—All that is known on this head is that, according to Doumergue, eggs are laid in July in Algeria.
Maxillary teeth few, anterior longest, gradually decreasing in size posteriorly, and followed, after an interspace, by a pair of enlarged, grooved fangs, situated below the posterior border of the eye; anterior mandibular teeth strongly enlarged. Head distinct from neck; eye moderate or rather small, with vertically elliptic pupil. Body moderately elongate; scales smooth, oblique, with apical pits. Tail moderate or rather short.
The eight species of this genus inhabit South-Eastern 217 Europe, South-Western Asia, and Africa. Two are dealt with here.
Form.—Moderately slender. Head much depressed. Tail five and a half to seven times in the total length.
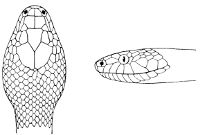
Fig. 32
Head-Shields.—Rostral broader than deep, just visible from above. Internasals shorter than the prefrontals. Frontal much broader than the supraocular, once and one-fourth to once and a half as long as broad, as long as its distance from the end of the snout, shorter than the parietals. Nasal divided or semidivided. Loreal twice and a half to thrice as long as deep, entering the eye below the preocular, which is in contact with the frontal. Two (rarely three) postoculars. Temporals small, scale-like, 2 or 3 + 3 or 4. Upper labials eight (rarely seven or nine), third, fourth, and fifth (rarely fourth and fifth, or fourth, fifth, and 218 sixth) entering the eye. Three or four lower labials in contact with the anterior chin-shields; posterior chin-shields very small and widely separated from each other by scales.
Scales with single or paired apical pits, in nineteen or twenty-one rows, usually nineteen in European specimens. Ventral shields 186 to 222; anal divided; subcaudals 48 to 73.
Coloration.—Greyish above, with 40 to 57 brown or black spots or bars on the body; a lateral series of smaller spots or vertical bars, alternating with the dorsals; the first spot, on the nape, elongate, usually with one or three linear processes in front, extending on the head (Plate XI.); usually a dark streak on each side of the head, from the eye to the angle of the mouth. Lower parts whitish, speckled, spotted, or marbled with grey or brown. Iris brown, with a golden circle round the pupil.
Size.—This species grows to a length of 2 feet 10 inches.
Distribution.—From Istria and Dalmatia to Greece, the Archipelago, Constantinople, Asia Minor, Cyprus, and Northern Syria; 2,600 feet appears to be its altitudinal limit.
Habits.—Although to a certain extent crepuscular or nocturnal, the Cat-snake is often seen hunting in the daytime, its food consisting almost exclusively of lizards, rarely of small mammals. Its movements are rather slow. The names Katzenschlange and 219 Ailurophis, translated Cat-snake, probably originated from the way in which this snake stalks its prey, and suddenly pounces upon it. According to Eiffe, the poison causes the death of a Lacerta vivipara in one minute, and P. de Grijs observed the larger Lacerta agilis to die in two or three minutes. As a rule even fresh-caught specimens allow themselves to be handled without attempting to bite; some specimens, on the other hand, are very savage. Stony localities, old walls, and ruins, are the favourite abodes of this snake, which does well in captivity.
Reproduction.—Seven or eight eggs are laid in July; they measure about 11⁄4 inches in length and 1⁄2 inch in width.
Very closely allied to the preceding, and differing from it only in the following points: Parietals shorter, slightly longer than the frontal, and anal entire. Loreal twice to twice and a half as long as deep. Fourth and fifth, or third, fourth, and fifth, labials entering the eye. Scales in nineteen or twenty-one rows. Ventrals 203 to 235; subcaudals 54 to 70.
Grey above, with 35 to 40 blackish spots on the body, the anterior largest and darkest; a lateral series of smaller spots or vertical bars. Lower parts 220 blackish, with small whitish spots and dots. Reaches a length of 31⁄2 feet.
This species inhabits the Caucasus, and, being on record from the northern slope (Kuban River), has to be included in the European fauna. It occurs also in Mesopotamia, a specimen from Bagdad being preserved in the British Museum. The young specimen figured on Plate XI. is stated to be from Constantinople.
Nothing is known of its habits, which are probably the same as those of Tarbophis fallax.
Maxillary, palatine, and pterygoid bones movable, the first much abbreviated, erectile perpendicularly to the large transverse bone, and supporting a pair of large canaliculated poison fangs; mandible without coronoid bone. No vestiges of pelvic arch.
All more or less poisonous, some being among the most dangerous of snakes.
Divided into two subfamilies, each of which is represented by one genus in Europe:
Viperinæ.—No pit on the side of the snout; maxillary bone not hollowed out.
Crotalinæ.—A deep pit on each side of the snout, between the nostril and the eye; maxillary bone hollowed out above.
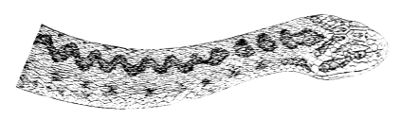
VIPERA URSINII
From Zoological Society’s Proceedings

VIPERA RENARDI
From Zoological Society’s Proceedings

VIPERA BERUS
After Sordelli
The Viperinæ inhabit nearly the whole of Europe, 221 Asia, and Africa; the Crotalinæ are Asiatic (one species extending its range into a small part of South-Eastern Europe) and American.
Head distinct from neck, covered with small shields or scales, with or without distinct frontal and parietal shields; eye moderate or small, with vertical pupil, separated from the labial shields by scales; nasal separated from the rostral by a naso-rostral. Body short. Scales keeled, with apical pits. Tail short.
Of the eleven species of this genus, six are found in Europe; two inhabit South-Western Asia, one the Indo-Malay region, and two Eastern Africa.
The distinction of the European species is one of considerable difficulty, owing to their close relationship and the presence of intermediate forms connecting them. Matters being so, it seems curious that the Common Adder should have been regarded by so many authors as generically distinct from the Asp Viper, under the name of Pelias berus. It is highly probable that hybrids are produced in those districts where two species coexist, as in some parts of France, North Italy, and Austria.
Form.—Short and stout. Snout obtusely pointed, flat above or with the canthus slightly raised. Eye 222 very small, usually smaller than the nasal shield, its horizontal diameter usually not exceeding its distance from the posterior border of the nostril, its vertical diameter often less than and rarely exceeding its distance from the mouth. Length of tail seven to eight times in total length in males, nine and a half to twelve times in females.
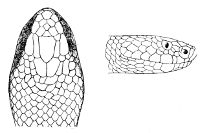
Fig. 33 (From Proceedings of the Zoological Society, 1893)
Head-Shields.—Rostral as deep as broad or slightly deeper than broad, visible from above, in contact with one apical shield (rarely with two). Distinct frontal and (usually) parietal shields, the former once and a half to once and two-thirds (rarely once and one-third) as long as broad, as long as its distance from the rostral or the end of the snout, and nearly always longer than the parietals; the latter always in contact with the former, rarely broken up into small shields. Four to seven small shields on the snout between the canthals, of which there are two on each side. Supraocular well developed, extending posteriorly beyond the vertical of the eye, separated 223 from the frontal by one to three shields, very rarely in contact with it. Six to ten scales round the eye, usually eight or nine, the upper preocular usually in contact with the nasal; a single series of scales between the eye and the labials. Nasal single. Temporal scales smooth. Upper labials six to nine, usually seven or eight, usually third or third and fourth below the eye. Three (rarely four) lower labials in contact with the chin-shields, of which there is but one pair.
Scales in nineteen (rarely twenty or twenty-one) rows, with two apical pits, strongly keeled on the back, less strongly on the sides, outer row smooth. Ventral shields 120 to 135 in males, 125 to 142 in females; anal entire; subcaudals 30 to 37 in males, 20 to 28 in females. By adding the subcaudals to the ventrals in a hundred specimens, the total numbers are 153 to 169 in males, 150 to 168 in females.
Coloration.—Unlike its ally V. berus, V. ursinii shows no sexual differences in the coloration. The ground colour of the back is usually yellowish or pale brown, sharply defined from the darker grey or brown colour of the sides; some specimens, however, are of an almost uniform brown ground colour. The light colour of the back is relieved by a series of more or less regular transversely oval, elliptic, or rhomboidal dark brown, black-edged spots, some or all of which may run together to form a wavy or zigzag band (Plate XII.). Two or three longitudinal series of dark 224 brown or black spots extend along the sides, the upper series, if present, occupying the space between the series of spots continued from the postocular band and the large dorsal spots or vertebral band, the lowermost following the outer row of scales. Small dark spots and one or two Λ-shaped markings are present on the upper surface of the head; an oblique dark band proceeds from the eye to the angle of the mouth, and is not infrequently confluent with the branches of the occipital Λ. The rostral and the labial shields are uniform yellowish-white, rarely with a few small, blackish spots or with brown borders. The chin and throat are yellowish-white, rarely with some blackish spots. The ventral and subcaudal shields are black, with transverse series of small white spots, or grey checkered with black and white, or whitish with small round black spots; the tail is but rarely (females) tipped with yellow.
The form recently described as V. macrops, Méhely, from Bosnia and Herzegovina, is distinguished by a usually larger eye, the vertical diameter of which equals or a little exceeds its distance from the mouth, and the parietals are often broken up into small shields. The postocular dark band is often reduced, originating at some distance from the eye, and is not prolonged beyond the mouth. In this geographical race melanic specimens occasionally occur, which are dark brown or blackish above, the lower parts not differing from those of the typical form.
225 Size.—20 inches appears to be the usual maximum size reached by this species, but, Dr. Werner informs me, a female 2 feet long has been found in Lower Austria.
Distribution.—First discovered in Italy in the Abruzzi, this species has since been found in the Basses-Alpes, near Digne, in various parts of Hungary, in Lower Austria, on the island of Veglia in Istria, and in Bosnia, Bulgaria, Herzegovina, and Montenegro. A very broken and curious distribution, the more so as V. ursinii is essentially a form of the plain in Lower Austria and Hungary, and an alpine form in Italy, in France, and in the Balkan Peninsula, where it only occurs between 3,000 and 6,800 feet. In no part of its habitat does it appear ever to be found in company with V. berus.
Habits.—Only a few specimens have hitherto been found in Italy and in France, but the species occurred up to a few years ago in extraordinary numbers in Lower Austria, in the immediate vicinity of Laxenburg. The intendant of the imperial castle pays a premium for the destruction of Vipers, and in the course of one year (1892) more than 1,000 specimens were brought to him. These snakes are found principally, though not exclusively, in the marshy meadows around the park, where they may be seen about in the daytime from May to September, feeding chiefly on lizards (Lacerta agilis), and also on small rodents. The lizards are swallowed as soon as seized, without 226 the effect of the poison being awaited as in other Viperid snakes. This Viper is as a rule of gentle disposition, allowing itself to be handled without attempting to bite, and village boys have been seen playing with them. Although occurring in such enormous numbers at Laxenburg, no accident from snake-bite has ever been heard of. The form from the Balkan Peninsula (V. macrops) is even more pacific still, and is believed never to make use of its poison apparatus, its food consisting of orthopterous insects. According to Captain Veith, who has collected a large number of specimens of this Southern form, the contents of the stomach as well as the excrements show this snake to feed exclusively on grasshoppers. On one occasion a big specimen showed such a swelling of the body as to lead to the conclusion that it had swallowed a mouse, but it soon after disgorged what proved to be a ball made up of the agglutinated remains of at least a hundred grasshoppers. When handled, this Viper hisses or even pretends to snap, but with closed mouth, never biting unless seriously hurt. The poison appears to have little effect on man.
Reproduction.—Nothing has been published on the breeding habits of this species, but in a letter to the author, dated January 14, 1913, Herr L. von Kirchroth, who has examined over 4,000 specimens since 1890, says the young are born in July or August, exceptionally as early as June. Young females bring forth from six to eight young, 227 older females from eight to eighteen; but a large female from Lower Austria is reported to have contained as many as twenty-two. The length of the new-born young is from 5 to 6 inches, and it grows rapidly within the first week, probably through stretching out, without taking any food.
According to Captain Veith, the form described as V. macrops brings forth only from three to five young.
Form.—Similar to the preceding species, but snout more pointed, the raised canthi rostrales meeting at an acute angle. Eye usually as large as in V. berus, nearly as large as the nasal shield; its horizontal diameter equal to its distance from the posterior or anterior border of the nostril, its vertical diameter equal to or a little less than its distance from the mouth. Length of tail seven and a half to nine times in total length in males, eight to ten times in females.
Head-Shields.—Rostral as deep as broad or a little deeper than broad, just visible from above, and in contact with a single apical shield. Distinct frontal and (usually) parietal shields, the former once and two-thirds to twice and one-third as long as broad, as long as or longer than its distance from the end of the snout, usually longer than the parietals; the latter 228 always in contact with the former, unless broken up into small shields. Two to six, usually three or four, small shields on the snout between the canthals, of which there are two on each side, the second broadly in contact with the supraocular. Supraocular well developed, extending posteriorly beyond the vertical of the eye, separated from the frontal by one to four shields. Nine to eleven, usually ten, scales round the eye, the upper preocular usually in contact with the nasal; either a single series of scales between the eye and the labials, or two series except under the centre of the eye, which is separated from the fourth labial by a single scale. Nasal single. Temporal scales all smooth, or the upper faintly keeled. Upper labials, eight or nine, fourth or fourth and fifth below the eye. Four (rarely five) lower labials in contact with the chin-shields, of which there is but one pair.
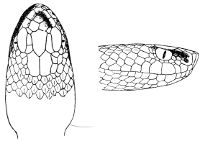
Fig. 34 (From Proceedings of the Zoological Society, 1893)
Scales in twenty-one (very rarely nineteen) rows, with two apical pits, strongly keeled, outer row smooth or feebly keeled. Ventral shields 130 to 148 in males, 229 130 to 150 in females; anal entire; subcaudals 31 to 37 in males, 24 to 30 in females.
Coloration.—As in V. ursinii, the sexes are alike in coloration. European specimens (Plate XII.) are very similar to V. ursinii, except that the labial shields are markedly dark-edged and speckled or spotted with brown or black. The dorsal band or series of spots is dark brown, edged with blackish; the ground colour of the middle of the back and of the scales of the two outer rows on each side is yellowish, of the sides (four rows of scales) greyish-brown with two or three series of dark brown spots; two dark Λ-shaped markings on the head; a dark postocular streak, extending or not to the side of the neck. The lower parts are whitish or pale greyish, with blackish dots, of which there is a series of larger ones along each side of the belly. The tip of the tail is never yellow.
Central Asian specimens are of a pale yellowish sand-colour, with a brown, dark-edged dorsal zigzag band or series of spots and two series of small spots on the sides. Belly whitish, dotted or spotted with black, or uniform blackish.
Size.—23 inches is the length of the largest specimen examined.
Distribution.—In Europe V. renardi, which has long been confounded with V. berus, is abundant in the district of Uralsk, in the steppe around Sarepta, in Crimea, and it is also found in Cis-Caucasia and 230 in Bessarabia. Its range extends far into Central Asia, being known from the Khirghiz steppes, the Emba steppes, the steppes near the Alatau, on the borders of the Urdshar, and in the Semipolatinsk district. Around Sarepta it is common in the bare steppe, and only exceptionally occurs in localities overgrown with willows and small shrubs.
Habits.—Nothing has been published concerning the habits of this snake, except that it is more sensitive to cold than V. berus and does not appear before the middle of April, retiring to its winter-quarters in the beginning of October. The food consists of small mammals and lizards.
Reproduction.—Pairs in May, and brings forth five to seven young in August, these young at birth measuring about 51⁄2 inches.
Form.—Short and stout. Snout flat above, rarely slightly concave, the upper contour broadly rounded or truncate in front, the canthus well marked, sometimes slightly raised, the loreal region nearly vertical. Eye as a rule smaller in females than in males, as large or nearly as large as the nasal shield; its vertical diameter equals or a little exceeds its distance from the mouth. Length of tail five and a half to nine times in total length in males, eight to ten and three-quarter times in females.
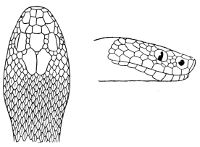
Fig. 35 (after Sordelli)
Head-Shields.—Rostral as deep as broad or slightly broader than deep, rarely once and one-third as deep as broad, not or but scarcely visible from above. In addition to the supraoculars, three large shields, the frontal and the parietals, are as a rule present on the top of the head. Frontal as long as broad or a little longer than broad, rarely much longer than broad, once and a half to twice and a half as broad as the supraocular, from which it is as a rule separated by one to four shields, as long as or a little shorter than its distance from the rostral, as long as or a little shorter than the parietals. Parietals usually in contact with the frontal and separated from the supraoculars by small shields, but sometimes in contact with both, or separated from the frontal. Exceptionally, in specimens from Great Britain, Germany, and Austria, the parietals, or the frontal and the parietals, are broken up into scales, and this is more frequently the case in specimens from North-Western Spain (var. seoanei). Upper surface of snout bordered by six (rarely by five or four) small shields, viz., two apicals (rarely one), and on each side two canthals, 232 the second of which is usually in contact with the supraocular; canthals very rarely united into one shield; the space between these shields covered by four to twenty flat or convex, juxtaposed scales, which very exceptionally are fused into a single large shield. Supraocular usually extending posteriorly beyond the vertical of the eye. Six to thirteen scales round the eye, usually eight to ten; two or three superposed scales, rarely two vertical series of scales, separate the preoculars from the nasal, which is single. As a rule a single series of scales intervenes between the eye and the labials; specimens with two series are of very exceptional occurrence (single specimens from Isle of Arran, Normandy, Southern Norway, and Carniola, in the British Museum), but there are occasionally two series except just below the centre of the eye. Upper labials six to ten, usually eight or nine; fourth or fourth and fifth (rarely third and fourth) below the eye. Temporal scales smooth, rarely feebly keeled. Three or four (rarely five) lower labials in contact with the single pair of chin-shields.
Scales in twenty-one (rarely nineteen or twenty-three) rows, with two apical pits, strongly keeled, those of the outer row smooth or feebly keeled. Ventral shields 132 to 150 (usually 137 to 147) in males, 132 to 158 (usually 140 to 150) in females; anal entire; subcaudals 32 to 46 (usually 35 to 40) in males, 24 to 38 (usually 28 to 33) in females.
233 Coloration.—It is characteristic of this species, contrary to the rule in snakes, to present such marked differences of colour, according to the sexes, that these can be distinguished in most cases from that character alone.
Whitish or pale grey specimens, with black belly and jet black dorsal markings (Plate XII.), are males. Brown and brick-red specimens, with the markings of a more or less dark brown or red, are females. There are also brown, reddish-brown or olive males with the markings of a deep black, and grey males with brown markings. A very pretty colour variety, which affects only females, is olive with brick-red band and spots. Some males, just before exuviation, have the lower surface of a pale greyish-blue (Coluber cæruleus, Sheppard), with the outer ends of the shields black. Specimens with yellowish-white chin and throat, which may be tinged with red, are females; males have the throat black, or whitish with the scales spotted or edged with black. Exceptional females occur (in Carniola) which in this respect resemble the males.
The markings vary considerably. Those on the back usually consist of a wavy or zigzag longitudinal band, flanked on each side with a series of spots corresponding to its sinuses; but this band may be partly or even entirely broken up into rhomboidal or transversely oval spots, or, losing its indentations, form a straight stripe edged on each side with a yellowish 234 streak (as in some specimens of the var. seoanei, from North-Western Spain). The markings may be absent altogether (var. concolor, Jan), or reduced to a narrow straight vertebral band (Pelias dorsalis, Gray). In the var. seoanei the zigzag band is often replaced by a dark brown vertebral band, three to five scales wide, bordered on each side by a series of subtriangular or crescentic black spots opposite to each other, as in the Pyrenean specimens of V. aspis. A pair of elongate dark markings are usually present on the back of the head, affecting the following shapes: |\,)(,)(,)(; By uniting together, this pair of markings may form a Λ or an X. An oblique dark streak extends on each side from the eye to the last labial shields, being sometimes prolonged a short way down the neck. The snout and vertex may be uniform or bear some symmetrical dark spots, or in some males be entirely black, the black involving the apex of the Λ-shaped marking. The labial shields are whitish or yellowish, those at least which are anterior to the eye being more or less broadly edged with brown or black.
The belly and the lower surface of the tail vary from grey or brown to bluish, blackish-grey, or black, the sides usually dotted or spotted with whitish; sometimes, especially in females, the belly is dark grey, each shield with a white posterior border which is broken up by a series of small roundish black spots. The end of the tail is often yellowish, bright yellow, 235 or pale orange below, rarely coral red, more commonly in females than in males.
The iris is usually coppery red, more rarely golden suffused with brown.
Black specimens occur, more or less frequently, all over the habitat of this species, and are often referred to as V. prester, Linnæus. A distinction has to be made between individuals which are black through darkening of the ground colour, and such as are thus coloured through expansion and confluence of the markings. The latter are males, and among them we may find intermediate stages showing how this melanism is brought about; in one case the black of the back is separated from the black of the sides by a narrow light brown wavy stripe, the remains of the ground colour. When, as in all females, and occasionally in males, the black is the result of a gradual darkening of the ground colour, the typical markings may still be detected under certain lights. Some specimens (from Schneeberg, Lower Austria) are black, with scattered golden dots, or of a dark mahogany brown speckled with yellowish. In nearly all the black specimens at least a few dots of whitish are visible on the lips, and of yellow under the end of the tail.
Most of the variations enumerated above occur irrespective of the geographical distribution. Two forms, however, deserve to be regarded as ill-defined local races: the var. seoanei, Lataste, from North-Western 236 Spain, in which, in addition to the peculiarities of coloration mentioned in the description, the canthus rostralis is frequently more distinctly raised, and the frontal and parietal shields are often disintegrated into scales; and the var. bosniensis, Boettger, from Bosnia, Carniola, and Carinthia, which is sometimes very suggestive of, and has been taken for, the typical form of V. aspis, having like it, though not at all constantly, two series of scales between the eye and the labials, and the zigzag band replaced by a series of dark bars across the back. The var. pseudaspis, Schreiber, from the plains of Sclavonia, described as straw yellow above, with narrow dark cross-bars, is hardly separable from the var. bosniensis.
Size.—Vipera berus is said to reach very exceptionally a length of 2 feet 11 inches. The largest specimen in the British Museum (from Belgium) measures 2 feet 31⁄2 inches: the largest British specimen 2 feet 3 inches. Both these specimens are females. The largest male measures 2 feet 2 inches.
Distribution.—Vipera berus ranges over the whole of Northern Europe, to the extreme north of Scotland, and the sixty-seventh degree in Scandinavia, and right across Northern Asia as far east as the island of Saghalien. It is generally distributed in Great Britain, occurring also on the Isles of Arran, Islay, Skye, Lewis, and Mull, rare or absent in some districts, common in others. Its distribution in Central and Southern Europe is irregular. In 237 Western France it does not extend much beyond the Loire to the south, only isolated captures being on record from the departments Vendée, Deux-Sèvres, Vienne, and Indre. Of rare occurrence south of Paris, in the departments Yonne and Allier and in the mountains of Auvergne, it is again abundant in some parts of the Central Plateau. To the east it is recorded from the departments Aude, Haute-Marne, and Vosges. In Belgium, it is known from Flanders, Limburg, the Meuse Valley, and the Ardennes; in Holland it is pretty generally distributed in the uncultivated parts. It is spread over nearly the whole of the German Empire with the exception of the vine districts, where it is absent or extremely rare; it is also very scarce in the mountains of the Black Forest; it is on record from only a few localities in Lorraine, and has never been found in Alsatia. In South Germany it is rarely found below 1,000 feet altitude. In Switzerland it is absent from the Jura, but occurs in the Alps chiefly between 2,500 and 9,000 feet. To the East it extends to Russia, as far north as 64°, Austria-Hungary, confined to the hills in the south, and Roumania. In the Balkan Peninsula it occurs in the mountains of Bosnia, of Herzegovina, and of Bulgaria, up to 7,000 feet. Absent from the South of France, it curiously reappears in the hills of the north coast of Spain, even at sea-level in Galicia, and in a few localities in North Portugal. On the southern side 238 of the Alps it is much rarer than V. aspis, but it has established itself in a few low-lying districts in Lombardy, Venetia, and the neighbouring part of Emilia.
Habits.—As we see from the above sketch of its distribution, the Adder generally avoids the hotter parts of Europe; when found in the plain in the South, as in Italy, it dwells in marshy localities, and Bonaparte called it Marasso palustre (Marsh Viper) in opposition to his Marasso alpino, Vipera ursinii. In the North, however, it usually selects in preference dry moors, sandy heaths, and hills well exposed to the sun, in which, although to a certain extent a nocturnal reptile, it delights to bask. Its food is very varied: weasels, mice, voles, shrews, moles, birds, lizards, slow-worms, frogs, salamanders, large slugs, have been found in the stomach, and the very young feed also on insects and worms. Of irascible temper as a rule, Adders are very ready to bite when fresh caught, but instances are known of their becoming quite tame in captivity, allowing themselves to be handled. As a rule they refuse food in captivity, but some have been known to live for as long as five years, being fed on lizards. Accidents from their bite, although seldom heard of in this country, are of frequent occurrence in France and in Germany, where many cases of fatal results on people have been recorded.
239 Reproduction.—Pairing takes place in April and May, and the young, five to twenty in number, are born in August or September, exceptionally as early as the end of July; the young, on releasing themselves from the thin, transparent membrane in which they are enclosed at birth, measure 6 to 8 inches. According to J. Geithe, a black female from Saxony gave birth to seventeen young, of which only one, a male, was black.
It is probable that exceptionally some individuals pair late in the summer or in the autumn. There is a trustworthy record, by Eiffe, of three pregnant females having been caught near Hamburg on March 12, 1882, one of them giving birth to young on the following day.
Dicephalous young have occasionally been observed. One 6 inches long was found crawling in a field near Hornburg in Germany in October, 1895, and, having been kept alive for some time, was observed to hiss and open the two mouths alternately when taking up a defensive attitude. Another similar monster, from Cornwall, is reported to have been sent alive to the London Zoological Gardens in 1854.
Form.—Rather more elongate than in the preceding. Snout flat above, more or less distinctly turned up at the end, with sharp, not or but very slightly 240 raised canthus, and vertical or nearly vertical loreal region. Vertical diameter of the eye equal to or a little less than its distance from the mouth. The raised upper border of the transversely truncate or obtusely pointed extremity of the snout, coupled with the downward slant of the supraocular region and canthus rostralis, gives the head, seen from the side, a peculiar expression; the eye is so oblique that a vertical line drawn from the posterior extremity of the supraocular shield to the lip usually passes through the eye or down its posterior border; but the extent to which the snout is turned up at the end varies considerably, some specimens approaching V. berus in this respect, others V. latastii. Length of tail five and a half to eight times in total length in males, seven to nine times in females.
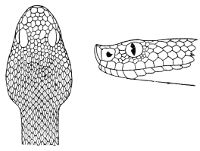
Fig. 36 (after Sordelli)
Head-Shields.—Rostral deeper than broad, its width two-thirds to seven-eighths its depth, extending to the upper edge of the snout. As a rule, with the exception of the large supraocular, the upper surface of the head is covered with small, subimbricate scales, which are smooth, very rarely feebly keeled, between the eyes 241 and on the snout; however, an enlarged frontal shield, or even a frontal and a pair of parietals, are sometimes present, though rarely so large as in a typical V. berus; when present, the frontal is separated from the supraocular by one or two series of scales; when the frontal is absent, four to seven series of scales separate the supraoculars. Upper surface of snout usually bordered by eight or nine small shields—viz., two or three apicals, in contact with the tip of the rostral and raised to form the turned-up nose, and, on each side, two canthals and the upper preocular, which separates the supraocular from the canthals, or three canthals; sometimes, however, the border is formed by six or seven small shields, the second canthal being in contact with the supraocular, as in V. berus. Supraocular usually with very convex outer border, not extending posteriorly beyond the vertical of the eye. Eight to thirteen scales round the eye, usually ten to twelve; one or two vertical series of scales separate the preoculars from the nasal, which is single or divided, and often rather deeply hollowed out. As a rule two series of scales (very rarely three) separate the eye from the labials; sometimes, however, there is but one scale between the eye and the fourth labial, the second series being incomplete. Upper labials nine to thirteen, usually nine to eleven, fourth and fifth, rarely fourth to sixth or fifth and sixth, below the eye. Temporal scales smooth or feebly keeled. 242 Four (rarely five) lower labials in contact with the single pair of chin-shields.
VIPERA ASPIS
After Calmette

VIPERA LATASTII
Scales in twenty-one or twenty-three (rarely nineteen or twenty-five) rows, with two apical pits, strongly keeled, those of the outer row more or less distinctly keeled, rarely perfectly smooth. Ventral shields 134 to 158 (usually 143 to 153) in males, 141 to 169 (usually 145 to 157) in females; anal entire; subcaudals 32 to 49 (usually 37 to 45) in males, 30 to 43 (usually 32 to 38) in females; the terminal caudal shield is sometimes shorter and less spine-like than in V. berus, quite obtuse in some specimens.
Coloration.—Grey, greyish-brown, brown, reddish-brown, coppery red, or orange, is the ground colour in individuals from the same district; in this respect sexual differences are less marked than in the preceding species, red or copper-coloured specimens being found in both sexes, and silvery white specimens do not seem ever to occur. In rare cases markings are entirely absent. In specimens from the greater part of France, Italy, and the Southern Tyrol (see Plate XIII.), the dark brown or black markings on the body are mostly in the form of narrow cross-bars, continuous across the back or broken on the vertebral line and often alternating with each other and with similar bars on the sides, thus producing a pattern not unlike that frequently found in Tropidonotus natrix; a narrow dark line 243 running straight or zigzag along the spine may connect these cross-bars, and in rare cases it is so broad as to produce a zigzag band similar to that of V. berus. In specimens from South-Western France and the Pyrenees, rarely in some from other parts of France and Italy, there is a broad dark grey or brown vertebral band between two series of black or blackish-brown spots, opposite to each other or alternating; this band may be straight or wavy, sometimes forming a regular zigzag; there is another series of blackish spots or short bars lower down on the side, alternating with those of the dorsal series.
The upper surface of the head may be devoid of any markings, or bear merely the two oblique dark streaks forming the branches of a Λ; or a dark cross-bar may be present on the snout, followed or not by smaller spots or a pair of oblique streaks on the occiput; the Λ on the back of the head may be united with the first cross-bar on the nape, and enclose a cordiform figure of the lighter ground colour. A light line sometimes borders the upper edge of the snout and the outer edge of the supraocular shield. A blackish band or a mere line extends obliquely from the eye to the first lateral spot; below this the upper lip is whitish, yellowish, or pinkish, with or without dark vertical bars on the sutures between the labial shields. The iris is golden or coppery red.
The lower parts vary as much as the upper: 244 sometimes black or steel blue, with or without whitish or reddish dots or spots, sometimes yellowish or pale reddish, with brown dots or marblings; in some young, white with greyish dots. The throat is yellowish white or pale reddish, uniform or speckled with blackish, sometimes (males) nearly entirely black. The end of the tail is usually bright yellow or reddish, or at least with a few bright spots. And finally we must mention black specimens—some nearly black by darkening of the ground colour, others intensely black by enlargement of the markings. A specimen from Piedmont, in the Turin Museum, shows the ground colour reduced to mere narrow light bars disposed in pairs. In most of these black specimens the chin and throat remain entirely or partially yellowish or reddish, and a few spots of the same colour are to be seen under the end of the tail.
A remarkable form of V. aspis, which some herpetologists would perhaps regard as entitled to rank as a species, is the var. hugyi, Schinz, from Calabria and Sicily. It is in some respects intermediate between V. aspis and V. latastii. The snout is rather more pointed than usual in the typical form, often, though not constantly, more strongly turned up at the end, and the canthus rostralis may be distinctly raised. Constantly two canthal shields, the second in contact with the supraocular. Ventral shields 134 to 148; subcaudals 30 to 43. Pale 245 greyish, yellowish, brownish, or reddish, above, with a broad wavy dark brown vertebral band, edged with darker; this band sometimes broken up into transversely oval spots; a lateral series of blackish-brown spots, each corresponding to the sinus of the dorsal band.
Specimens so completely intermediate between Vipera aspis and V. berus as to render their naming arbitrary are known from parts of France and Italy where the two species coexist, and are probably to be regarded as hybrids.
Size.—The largest specimen examined (St. Sever, Landes, in the Lataste Collection) measures 2 feet 21⁄2 inches. It is a male. The largest female in the British Museum is 2 inches shorter.
Distribution.—Vipera aspis is found over the whole of France south of a line connecting the departments Loire-Inférieure, Orne, Seine-et-Marne, and Meurthe-et-Moselle, and ascends the Pyrenees to the altitude of 7,250 feet. In Germany it is known from Lorraine and the Black Forest, in Switzerland from the western and southern parts, up to 5,000 feet on the northern side of the Alps. It occurs also in Austria, in the Southern Tyrol and in the Karst, and is distributed over the whole of Italy and Sicily, reaching an altitude of 9,700 feet in the Alps. Most of the specimens from the western parts of the Balkan Peninsula which have been referred to this species belong, apparently, to V. berus, var. 246 bosniensis, but one from Jahorin in Bosnia, altitude 5,650 feet, preserved in the Bosnian Museum, is pronounced by Werner to be an unquestionable V. aspis.
Habits.—This Viper shows a predilection for hot and dry localities. It is both diurnal and nocturnal, and does not seem to wander far from its hole in a rock or in the earth. It is slow in its movements, but very irascible, and innumerable accidents, in some cases fatal to man, are caused yearly in many parts of France, where it is extremely abundant. Its food consists principally of small mammals, young birds, and lizards, but the very young eat insects and worms. In France it retires into its winter-quarters at the end of October or in November, and numerous specimens often congregate in the same hole; it resumes its activity towards the end of March or the beginning of April, sometimes as early as the end of February. In rare cases it will even leave its retreat in the middle of winter, to bask in the sun. In captivity it long retains its savage temper, and usually refuses all food.
Reproduction.—Vipera aspis pairs in April and May; the pair are entwined in each other’s coils. The young, four to eighteen in number, but rarely more than ten, are born in August or September, and measure 7 or 8 inches.
Several cases of dicephaly in young specimens have been described.
Form.—Heavier than in the preceding. Head similar, but snout more pointed and loreal region slanting towards the lip, well visible when the head is viewed from above. The extent to which the snout is turned up at the end varies considerably, sometimes similar to certain specimens of V. aspis, sometimes forming an appendage which is only a little less developed than in V. ammodytes. Length of tail six and a half to seven and a half times in total length in males, seven and a half to nine times in females.
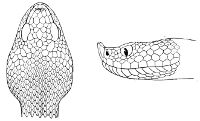
Fig. 37
Head-Shields.—Rostral once and a half to twice as deep as broad, nearly reaching the tip of the rostral wart. Upper surface of head covered with small, smooth or feebly keeled, subimbricate scales, among which a slightly enlarged frontal, or a frontal and a 248 pair of parietals, may sometimes be distinguished; four to seven longitudinal series of scales between the supraoculars, which are large, and do not as a rule extend posteriorly beyond the vertical of the eye. Five or six (rarely three) scales on the posterior aspect of the raised part of the snout; two canthal scales on each side, the second in contact with the supraocular, or separated from it by the uppermost preocular. Eight to thirteen (usually nine to twelve) scales round the eye; two or three series of scales between the eye and the labials. Nasal hollowed out, entire, separated from the preoculars by one or two vertical series of scales; naso-rostral sometimes divided into two in North African specimens. Temporal scales smooth or feebly keeled. Upper labials nine to eleven (rarely eight), fourth and fifth (rarely third and fourth) below the eye. Four or five lower labials in contact with the single pair of chin-shields.
Scales in twenty-one rows, with two apical pits, strongly keeled, outer row smooth or feebly keeled. Ventral shields 125 to 146 in males, 135 to 147 in females; anal entire; subcaudals 35 to 45 in males, 32 to 38 in females.
Coloration.—Grey or brown above, the back often paler than the sides, with a broad darker, usually black-edged, wavy or zigzag band along the spine, and a lateral series of spots (Plate XIII.); the band sometimes replaced by large rhombic or transversely oval 249 spots. Head with or without dark markings above, sometimes with two oblique dark streaks on the occiput; a dark streak from behind the eye to the first lateral spot, sometimes originating at a considerable distance from the eye; upper lip white or pale brown, more or less speckled or spotted with black. Lower parts grey, spotted with black and white, or blackish speckled with white, the end of the tail usually yellow or with yellow spots.
Size.—This Viper is not known to exceed a length of 2 feet.
Distribution.—Locally distributed over the greater part of Spain and Portugal, as far north as Burgos and Barcelona. Also found in Morocco near Tangier, and in Algeria near Bona and Guyotville.
Habits.—Lataste’s Viper lives in stony and arid districts, and also in forests. The food consists chiefly of small mammals, but remains of a scorpion have been found in the stomach of an adult, and of a centipede in that of a young. According to Graells, this Viper easily climbs low trees in search of young birds, five of which have been found in the stomach of one specimen. The bite is believed to be less dangerous than that of V. aspis, and rarely causes the death of man and domestic animals.
This species may be divided into several geographical forms. The typical form will be described first.
250 Form.—Short and heavy. Snout pointed, produced into an erect, horn-like dermal appendage covered with scales; canthus rostralis strong, sometimes slightly raised, loreal region slanting more or less towards the lip. Vertical diameter of the eye less than its distance from the mouth in the adult. Length of tail six to nine and a half times in the total length in males, eight to eleven times in females.
Fig. 38 (after Sordelli)
Head-Shields.—Rostral usually broader than deep. Naso-rostral (rarely divided into two) usually reaching the canthus rostralis, and extending considerably higher up than the upper border of the rostral. Rostral appendage covered with ten to seventeen scales, arranged in three (rarely two or four) transverse series between the rostral shield and the apex. Upper surface of head covered with small smooth or faintly keeled, subimbricate scales, among which a feebly enlarged frontal shield or a frontal and a pair of parietals are rarely distinguishable; when present, the frontal is separated from the supraocular by two series of scales; on the vertex five to eight series of scales separate the supraoculars. Two (rarely three) 251 canthal scales, the second separated from the supraocular by the upper preocular. Supraocular with very convex outer border, usually not extending posteriorly beyond the vertical of the eye. Ten to thirteen scales round the eye; one or two vertical series of scales separate the preoculars from the nasal, which is single or rarely divided, and hollowed out. Two series of scales between the eye and the labials. Upper labials eight to twelve, usually nine or ten, usually fourth and fifth below the eye. Temporal scales smooth or feebly keeled. Four or five lower labials in contact with the single pair of chin-shields.
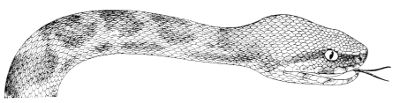
VIPERA LEBETINA
After Sordelli

VIPERA AMMODYTES
After Sordelli

ANCISTRODON HALYS
After Sordelli
Scales in twenty-one or twenty-three (rarely twenty-five) rows, with two apical pits, strongly keeled, those of the outer row smooth or feebly keeled. Ventral shields 143 to 161 in males, 147 to 160 in females; anal entire; subcaudals 27 to 40 in males, 24 to 37 in females.
Coloration.—Grey, pinkish-grey, brown, yellowish-brown, or brick red above, with a more or less distinct wavy or zigzag black or brown, usually black-edged, band along the back, or a series of large rhombs connected on the median line, with or without a lateral series of dark spots; specimens with the markings of an intense black are males (Plate XIV.). Head with or without dark markings, very variable in disposition, sometimes forming a Λ on the occiput, or a lyre-shaped figure confluent with the dorsal 252 band; a dark streak or broad band from the eye to the angle of the mouth, sometimes continued along the neck; dark vertical bars often present on the sides of the snout and under the eye; on the lower lip, the dark shade, if present, broken up by light bars separated by two to four labial shields. Belly greyish or pink, powdered with brown or black, or dark bluish-grey, with or without black and white spots; lower surface of end of tail orange or coral red, rarely yellow. Iris golden. Specimens with a straight brown vertebral band flanked with triangular black spots pointing outwards (var. steindachneri, Werner) are known from Hungary, and resemble certain colour varieties of V. berus and V. aspis. Black specimens are very rare.
The following varieties are important geographical forms occurring in Europe:
Var. montandoni, Boulenger: Naso-rostral shield never reaching the canthus rostralis nor the summit of the rostral shield, which is deeper than broad (once and one-seventh to once and a half); rostral appendage clad with ten to fourteen scales, in three (rarely two or four) transverse series between the rostral shield and the apex. Scales in twenty-one rows. Ventral shields 149 to 158; subcaudals 30 to 38. A more or less distinct dark blotch on the lower lip, involving five to seven labial shields without complete interruption. Lower surface of end of tail yellow.
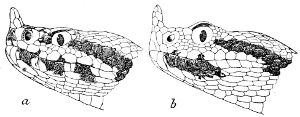
Fig. 39—Side Views of Heads of V. ammodytes typica (a) and Var. meridionalis (b)
Fig. 40—Front Views of End of Snout, showing the Lepidosis.
(From Proceedings of the Zoological Society, 1903)
a, Form typica; b, var. meridionalis; c, var. montandoni
Var. meridionalis, Boulenger: Naso-rostral shield never reaching the canthus rostralis, and but rarely extending higher up than the upper border of the rostral, which is often as deep as broad or a little deeper than broad; rostral appendage clad with fourteen to twenty scales, in four or five (rarely three) transverse series between the rostral shield and the apex. Supraciliary edge usually more prominent than in the typical form, sometimes slightly angular. Scales in twenty-one rows (very rarely twenty-three). Ventral shields 133 to 147; subcaudals 24 to 35. 254 A more or less distinct dark blotch on the lower lip, involving five or six labial shields without interruption. Lower surface of end of tail yellow.
Size.—This Viper exceptionally attains a length of 3 feet. The largest male in the British Museum measures 2 feet 6 inches, the largest female 2 feet 4 inches. In V. berus females grow to a larger size than males; in this species, as in V. aspis, the reverse appears to be the rule.
Hybrid.—A female specimen, presumed to be a hybrid between V. berus and V. ammodytes, was obtained by Captain Veith in 1902 in Carinthia, in a locality where both these species occur together. The shape of the head is exactly that of a typical V. aspis, the snout distinctly turned up at the end, but without wart or scaly appendage, the raised portion being covered by the apex of the rostral shield and three apical shields. The rostral shield, which is a little deeper than broad (5 : 4), extends above the level of the slightly raised canthus rostralis, which bears two shields, the second in contact with the supraocular. The naso-rostral extends to the canthus rostralis, where it joins the first canthal and the lateral apical shield; one series of scales between the nasal shield and the preoculars. On the upper surface of the head the snout is covered with fifteen subimbricate smooth scales, in addition to the canthals and apicals. A frontal shield and a pair of parietals are well developed, 255 although smaller than in an average V. berus; the parietals are in contact with the frontal, between which and the supraoculars two series of scales intervene. The supraocular, as in V. berus, extends backwards considerably beyond the vertical of the eye. Eleven scales round the eye; two series of scales between the eye and the labials. Temporal scales smooth. Upper labials nine, fourth and fifth below the eye. Scales in twenty-one rows, outer row smooth. Ventrals 159; subcaudals 31.
Grey-brown above, with a reddish-brown or mahogany-coloured zigzag vertebral band and a lateral series of paler reddish-brown spots; temporal band ill-defined in front; no markings on the upper surface of the head; lips pinkish, with a few reddish-brown spots. Ventral shields pale brownish, finely speckled or powdered with blackish, and with small whitish spots on the free edge and reddish brown spots on the sides. Tail orange red below. Iris fire red.
Total length 2 feet 21⁄3 inches; tail 22⁄3 inches.
Distribution.—The typical form is known from Northern Venetia, Austria-Hungary (Styria, Carinthia, Southern Tyrol, Carniola, Illyria, Istria, Croatia, Slavonia, and eastward through Southern Hungary to Transylvania), Dalmatia, Bosnia, Herzegovina, Montenegro, Albania, and Servia. In the Alps up to 1,300 feet, in the Balkan Peninsula up to 7,500 feet. The var. montandoni inhabits Roumania and Bulgaria. 256 The var. meridionalis inhabits Greece, with the Archipelago, Asia Minor, and Syria.
The specimens from Transcaucasia constitute a further variety (var. transcaucasiana, Boulenger), agreeing with the var. montandoni in the rostral scutellation and in the number of ventral shields (150 to 156), but differing in the markings on the back; these consist of dark bars or alternating paired dark spots, as in the typical form of V. aspis. The dark and light markings on the lower lip are as in the typical V. ammodytes, and the lower surface of the tail is pale yellow or greenish towards the end.
Habits.—Notwithstanding its name ammodytes, this Viper is by no means restricted to sandy localities; on the contrary, it shows a predilection for dry stony hills with low vegetation, and has often been found climbing bushes. It avoids thick forest, but occurs on the edges of woods and in clearings, as well as on the borders of roads through woods. In the cooler regions of the mountains, which it ascends to a considerable altitude, it is essentially diurnal, leaving its retreat only when the sun shines; but in warm localities it is stated to be principally nocturnal, appearing in numbers by moonlight. The length of its period of hibernation depends entirely on the climate, but when the winter is mild it may be seen about in midwinter whenever the sun shines. The poison of 257 this Viper is stated to be more active than that of V. berus and V. aspis, and, as the snake is very common in some parts of Austria and the Balkan Peninsula, fatal accidents to man are frequent.
The food consists of small mammals and birds, and also of lizards. V. ammodytes does much better in captivity than its European congeners, and takes food more readily.
The hissing is louder than in V. berus and V. aspis, and it is often produced, on the approach of man, by specimens lying in such perfect concealment that their presence would not otherwise be suspected; this habit, like the rattling of the Crotalus, is evidently detrimental to the species in its relation to man.
This species is extremely abundant in some parts of Austria, and over 7,000 specimens were killed in a district of Southern Styria in the course of two years (1892, 1893). According to Werner, it is the commonest of all snakes in Bosnia and Herzegovina.
Reproduction.—Pairing takes place in the spring, sooner or later according to altitudes, and the young, five to fourteen in number, are born in August.
Form.—Short and heavy. Snout rounded, obtuse, usually with well-marked canthus, and the loreal region slanting towards the mouth. Eye small, its 258 vertical diameter less than its distance from the mouth in the adult. Nostril large and directed backwards. Length of tail six (males) to ten times (females) in the total length.
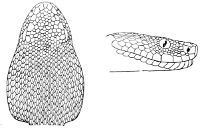
Fig. 41 (after Sordelli)
Head-Shields.—Rostral as deep as broad, a little broader than deep, or slightly deeper than broad, reaching or nearly reaching the upper surface of the snout, and in contact with two or three apical shields. Upper surface of head covered with small subimbricate scales, which are all more or less distinctly keeled, or, rarely, smooth on the snout and forehead; seven to twelve longitudinal series of scales between the eyes (supraoculars included); two to four canthal shields, of which the anterior is the largest, and may be regarded as a supranasal. Supraocular narrow, usually broken up into two or more small shields. Twelve to eighteen scales round the eye; two or three series of scales between the eye and the labials; two or three vertical series of scales separate the preoculars from the nasal, which is single and often strongly hollowed out, 259 and usually partially fused with the naso-rostral. Upper labials nine to twelve, usually fourth and fifth below the eye. Temporal scales keeled. Four or five lower labials in contact with the single pair of chin-shields.
Scales in twenty-three to twenty-seven rows, usually twenty-five, with two apical pits, strongly keeled, those of the outer row smooth or feebly keeled. Ventral shields 151 to 177 in males, 153 to 180 in females; anal entire; subcaudals 42 to 51 in males, 38 to 49 in females.
Coloration.—Very variable. The typical form, which alone is represented in Europe, and was originally described from Cyprus, is grey, greyish-buff, or pale brown, above, with two dorsal series of darker spots, which may stand in pairs, alternate, or unite to form cross-bars, and a lateral series of large dark spots or bars. A more or less distinct dark band on each side of the head, passing through the eye and often extending to the neck; a dark bar or triangular spot below the eye, and usually another below the nostril. Lower parts pinkish-white, powdered with grey-brown, with or without dark brown spots; end of tail yellow. The ground colour of the young is pink or flesh-colour. In specimens from desert sandy regions in Asia and North Africa the markings may be very indistinct, the snake being of a nearly uniform pale buff.
In the var. mauritanica, Guichenot, from Morocco 260 and Algeria, the back has three series of very large dark brown or reddish-brown spots, separated by a network of the yellowish ground colour, or the middle series may be transformed into a wavy or zigzag band. The scales in this variety are usually in twenty-seven rows, instead of twenty-five (rarely twenty-three) as in the typical form.
Size.—This species, the largest of European Vipers, grows to a length of 41⁄2 feet.
Distribution.—The European habitat of V. lebetina is restricted to the Cyclades, where it is not uncommon on the island of Tinos, and appears to be found also on Kimoli. It is common on Cyprus, where it is called Kufi, or Deaf Snake, and extends from Syria and Asia Minor through Transcaucasia, Mesopotamia, Persia, Northern Baluchistan, to Afghanistan and Cashmere. It is further found on the Atlas of Morocco and Algeria, near Oran and Bona, and in Tunisia. Its reported occurrence in Egypt has not been confirmed by recent investigations.
Habits.—According to M. Doumergue—who has had ample opportunities of observing this Viper near Oran, where it is common—it is a nocturnal reptile, rarely moving about in the daytime. It inhabits rocky localities, where there is brushwood, and vineyards. During the day it remains sluggish under large stones. It is most frequently met with in April and May.
261 On Milos, Dr. de Bedriaga observed this much-dreaded snake, the bite of which is probably as bad as that of its Indian ally, the Daboia, V. russelli, to occur frequently in gardens, and to crawl about near houses in villages after sunset. The same observer has noted a sort of valvular closing of the nostril through raising of the posterior part of the nasal shield when the snake prepares to strike.
The food consists principally of mammals up to the size of a rabbit.
Reproduction.—According to Doumergue’s observations in Algeria, the young, up to thirteen in number, are born in May and June.
Head distinct from neck, its upper surface covered with large shields, as in the normal Colubridæ; a loreal pit; eye moderate or small, with vertical pupil. Body moderately elongate or short. Scales keeled (or smooth), with apical pits. Tail moderate or short.
This genus is distributed over nearly the whole of Asia, the eastern parts of the United States of America, and Mexico and Central America. One of the Asiatic species just penetrates into South-Eastern Europe, and is the sole representative of the Crotalinæ in this part of the world.
Form.—Moderately elongate. Head subtriangular, flat or slightly concave above, swollen in the temporal region, very distinct from neck; snout rounded or obtusely pointed, slightly turned up at the end, with obtuse canthus and vertical or slightly oblique loreal region; eye rather small. Tail seven to eight and a half times in the total length.
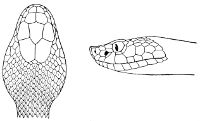
Fig. 42
Head-Shields.—Rostral as deep as broad or slightly broader than deep, just visible from above. A pair of internasals and a pair of larger prefrontals. Frontal as broad as the supraocular, as long as or a little longer than its distance from the end of the snout, as long as or a little shorter than the rather short parietals. Supraocular extending beyond the vertical of the posterior border of the eye. Loreal pit between three shields, separated from the labials. Nostril between two nasals, the posterior of which 263 is separated from the upper preocular by a loreal. Two preoculars, one or two postoculars, and a subocular. Three large lower temporals, anterior largest. Upper labials seven or eight, third and fourth largest, third entering the eye. A pair of small chin-shields.
Scales sharply keeled, with two apical pits, in twenty-three rows. Ventral shields 149 to 174; anal entire; subcaudals 31 to 44 pairs.
Coloration.—Pale yellowish-grey, greyish-brown, or reddish, sometimes greenish in young specimens, with transverse series of darker spots or with more or less regular dark bars with serrated edges across the back (Plate XIV.); these bars may be narrower than the interspaces between them, or so large as to cause the back to appear brown with light cross-bars; the bars sometimes broken up on the vertebral line, and the two halves alternating. The sides usually paler and bearing two alternating longitudinal series of small spots, the lower of which are usually darker, and sometimes extend on the outer ends of the ventral shields. Head pale above, with a dark spot on the middle of the snout, a cross-bar or a pair of spots between the eyes, a spot or short band on each side of the parietal region, and a horseshoe-shaped band on the occiput, the branches of which are more or less produced on the nape; all these markings sometimes confluent. A broad, dark, light-edged band on the temple. Lips whitish, speckled 264 with brown. Lower parts whitish, more or less profusely speckled with grey or brown. The horny shield terminating the tail usually dark brown or black at the end.
Size.—This species rarely reaches a length of 29 inches. The largest specimen in the British Museum measures only 19 inches.
Distribution.—From the north and east coasts of the Caspian Sea, across Central Asia to the Upper Yenissei, as far north as 51°. In Europe it is only known from two arid tracts between the Volga and the Ural, near the Caspian Sea, viz., the Saltan-Murat desert and the Induski hills.
Habits.—Nothing has been published on the habits of this snake, but they are probably similar to those of its near and more eastern relative, A. blomhoffi, Schlegel, which inhabits China and Japan. A. blomhoffi is said to be more or less nocturnal, although showing a predilection for localities well exposed to the sun. It is ovoviviparous. The symptoms of its bite, which is rarely fatal to man, are the same as in the Vipers.
All the species of Ancistrodon, so far as they have been observed, are in the habit of raising and vibrating the tail, like the Rattlesnakes, when coiling themselves up in a defensive attitude.
| A | B | C | D | E | F | G | H | I | J | K | L | M |
| N | O | P | Q | R | S | T | U | V | W | X | Z |
Acanthocephala, 110
Acrochordinæ, 5
Adder, 230
Æsculapian Snake, 187
æsculapii (Coluber), 187
Ailurophis vivax, 217
Albinism, 39
Aldrovandi’s Snake, 182
Amblycephalidæ, 5
ammodytes (Vipera), 249
Anal pockets, 83
Anal shield, 17
Ancistrodon, 261
Ancistrodon halys, 262
Antitoxic serums, 66
Arteries, 77
Ascariasis, 111
asianus (Zamenis gemonensis, var.), 173
Asp Viper, 239
aspis (Vipera), 239
astreptophorus (Tropidonotus natrix, var.), 154
ater (Tropidonotus natrix, var.), 156
atrovirens (Coluber), 170
aurolineatus (Coluber), 167
austriaca (Coronella), 197
Bacteria, 113
berus (Pelias), 221
berus (Vipera), 230
Bezoar stones, 141
bilineatus (Tropidonotus natrix, var.), 154
Blind Snake, 144
Blood-supply, 77
Blunt-nosed Viper, 257
Body, 8
bosniensis (Vipera berus, var.), 236
Brain, 73
Burrowing Snakes, 91
cæruleus (Coluber), 233
Canker, 113
Canthus rostralis, 13
Captivity, 100
carbonarius (Zamenis), 172
caspius (Zamenis gemonensis, var.), 172
Cat-Snake, 217
Caucasian Cat-Snake, 219
cervone (Elaphis), 182
Cestoda, 112
cettii (Tropidonotus natrix, var.), 155
chersoides (Tropidonotus), 167
Classification, 2
Cobra di capello, 9
Cœlopeltis, 207
Cœlopeltis monspessulana, 208
collaris (Contia), 207
Coloration, 29
Colour, 29
Coluber, 181
Coluber æsculapii, 187
Coluber atrovirens, 170
Coluber aurolineatus, 167
Coluber cæruleus, 233
Coluber dione, 185
Coluber dumfriesiensis, 200
Coluber hydrus, 160
Coluber insignitus, 208
Coluber leopardinus, 191
Coluber longissimus, 187
Coluber quadrilineatus, 191
Coluber quatuorradiatus, 182
Coluber quatuorlineatus, 182
Coluber riccioli, 202
Coluber rubens, 204
Coluber sauromates, 183
Coluber scalaris, 194
Coluber torquatus, 152
Coluber viridiflavus, 170
concolor (Tropidonotus tessellatus, var.), 162
concolor (Vipera berus, var.), 234
Coniophis, 7
Constriction, 96
Contia, 205
Contia modesta, 205
Coronella, 196
Coronella austriaca, 197
Coronella girondica, 202
Coronella italica, 197
Coryphodonts, 58
Crotalon, 20
Crotalus, 21
cucullatus (Macroprotodon), 213
Dahl’s Whip-Snake, 177
dahlii (Zamenis), 177
Dasypeltinæ, 5
Defecation, 99
Definition, 1
Deglutition, 98
Dentition, 53
Desert Snakes, 36
Development, 88
Diacranterians, 58
Dicephalous young, 89
Digestion, 99
dione (Coluber), 185
Dione Snake, 185
Disgorging, 99
Distribution, 118
Dolichosauria, 1
dorsalis (Pelias), 234
Drinking, 101
dumfriesiensis (Coluber), 200
Dwarf Snake, 205
Economic value, 138
Eggs, 85
Egg-tooth, 89
Elachistodontinæ, 5
Elaphis cervone, 182
Elapinæ, 5
Elaps, 35
elsneri (Coluber leopardinus, var.), 193
Embryo, 88
Epiglottis, 79
Eryx, 147
Eryx jaculus, 147
European Whip-Snake, 170
External characters, 8
Exuviæ, 20
Exuviation, 105
False Smooth Snake, 213
fallax (Tarbophis), 217
Fangs, 55
Fascination, 104
Fat-bodies, 81
Faunistic works, 130
Feigning death, 104
fitzingeri (Coronella), 197
flavescens (Coluber), 187
flavescens (Tropidonotus tessellatus, var.), 163
Flukes, 112
Flying Snakes, 94
Food, 95
Form, 8
Fossils, 6
Gape, 11
Gastrosteges, 15
gemonensis (Zamenis), 170
Genital organs, 82
girondica (Coronella), 202
Glottis, 79
Grass Snake, 152
Greek Blind Snake, 144
Ground Snakes, 91
Habits, 91
Hæmogregarines, 112
hagenbecki (Tropidonotus tessellatus, var.), 162
halys (Ancistrodon), 261
Head, 10
Head-shields, 17
Hearing, 74
Heart, 77
Helagras, 7
Hemipenes, 82
Hibernation, 105
Hind limbs, 10
hippocrepis (Zamenis), 179
Hissing, 102
Homalopsinæ, 5
Hood, 9
Horseshoe Whip-Snake, 179
hugyi (Vipera aspis, var.), 244
Hybrids, 90
Hydrophiinæ, 5
hydrus (Coluber), 160
Hyoid apparatus, 42
iberus (Tarbophis), 219
Iguanognathus, 54
Ilysiidæ, 4
incertus (Tropidonotus viperinus, var.), 167
Incubation, 88
Intelligence, 101
insignitus (Coluber), 208
Integument, 8
Intestines, 79
Iris, 37
Isodonts, 58
italica (Coronella), 197
Jacobson’s organ, 73
jaculus (Eryx), 147
Javelin Sand-Boa, 147
Key to the identification, 22
Kidneys, 80
Kufi, 257
Labial pits, 76
Lacertilia, 1
lacertina (Natrix), 208
Ladder Snake, 194
Largest Snakes, 21
Larynx, 79
Lataste’s Viper, 247
latastii (Vipera), 247
lebetina (Vipera), 257
Leopard Snake, 191
leopardinus (Coluber), 191
lineaticollis (Tropidonotus tessellatus, var.), 162
Liver, 81
Livery of the young, 38
Locomotion, 93
Long-nosed Viper, 249
longissimus (Coluber), 187
Loreal pit, 75
Loreal region, 12
Lungs, 78
Lycodonts, 58
Lymph-hearts, 78
Macroprotodon, 212
Macroprotodon cucullatus, 213
macrops (Vipera), 224
Markings, 29
mauritanica (Vipera lebetina, var.), 259
Melanism, 39
Mental groove, 13
meridionalis (Vipera ammodytes, var.), 253
Mimicry, 35
modesta (Contia), 205
monspessulana (Cœlopeltis), 208
montandoni (Vipera ammodytes, var.), 252
Montpellier Snake, 208
Mortality from Snake-bite, 133
Mosasauria, 1
murorum (Tropidonotus natrix, var.), 154
Natrix lacertina, 208
Natrix vulgaris, 152
natrix (Tropidonotus), 152
Neck, 9
Nematoda, 111
Nerodia, 152
Nervous system, 73
neumayeri (Cœlopeltis monspessulana, var.), 210
niger (Coluber longissimus, var.), 188
nigrescens (Tropidonotus tessellatus, var.), 162
Nocturnal Snakes, 93
Northern Viper, 230
Œsophagus, 79
Olfactory organ, 73
Ophidia, 1
Organs of a sixth sense, 75
Orsini’s Viper, 221
Oviparous Snakes, 86
Oviposition, 85
Ovoviviparous Snakes, 85
Pairing, 84
Pallas’s Pit-Viper, 262
Parasites, 107
Pelias berus, 221
Pelias dorsalis, 234
Pelvic arch, 52
persa (Tropidonotus natrix, var.), 154
persicus (Zamenis gemonensis, var.), 172
picturatus (Tropidonotus natrix, var.), 155
Poison apparatus, 62
Poison glands, 62
Poisons, 66
prester (Vipera), 235
Protozoa, 112
pseudaspis (Vipera berus, var.), 236
Pupil, 12
Pythoninæ, 4
quadrilineatus (Coluber), 191
quatuorradiatus (Coluber), 182
quatuorlineatus (Coluber), 182
Rattle, 20
Rattling, 103
Reflex movements, 106
Relation to man, 133
Renard’s Viper, 227
renardi (Vipera), 227
Replacement teeth, 55
Reproduction, 82
Reptation, 93
Reptilia, 1
Rhiptoglossa, 1
riccioli (Coluber), 202
Ring-Snake, 152
romanus (Coluber longissimus, var.), 188
rubens (Coluber), 204
rubro-maculosus (Tropidonotus tessellatus, var.), 162
Rustling, 102
Sand-Boa, 147
Sand-Snakes, 91
Sand-Viper, 249
sardus (Zamenis), 172
sauromates (Coluber), 183
scalaris (Coluber), 194
scalaris (Coronella austriaca, var.), 199
Scales, 14
schwoederi (Coluber leopardinus, var.), 193
scutatus (Tropidonotus natrix, var.), 155
semimaculata (Contia modesta, var.), 207
Sense organs, 73
seoanei (Vipera berus, var.), 235
Serum treatment, 67
Shedding of the epidermis, 20
Shields, 14
Skeleton, 40
Skull, 40
Smallest Snakes, 21
Smooth Snake, 197
Snake-charmers, 139
Snake-stones, 141
Snake swallowing her young, 88
Snake-worship, 139
Snout, 12
Solenoglyphs, 56
Southern Smooth Snake, 202
Squamata, 1
steindachneri (Vipera ammodytes, var.), 252
Stomach, 79
Subcaudal shields, 15
subfasciatus (Tropidonotus natrix, var.), 155
subgriseus (Coluber longissimus, var.), 189
Symoliophis, 7
Syncranterians, 58
Tail, 9
Tarbophis, 216
Tarbophis fallax, 217
Tarbophis iberus, 219
Teeth, 53
Tessellated Water-Snake, 160
tessellatus (Tropidonotus), 160
Thymus gland, 78
Ticks, 108
Tongue, 74
Tongue-worms, 108
torquatus (Coluber), 152
trabalis (Zamenis gemonensis, var.), 172
Trachea, 78
transcaucasiana (Vipera ammodytes, var.), 256
Trematoda, 112
Tropidonotus, 152
Tropidonotus chersoides, 167
Tropidonotus natrix, 152
Tropidonotus tessellatus, 160
Tropidonotus viperinus, 165
Typhlops, 144
Typhlops vermicularis, 144
Ureters, 81
Uropeltidæ, 5
Urosteges, 15
ursinii (Vipera), 221
Veins, 77
veithi (Coronella austriaca, var.), 199
Ventral shields, 15
vermicularis (Typhlops), 144
Vertebræ, 50
Vertebral column, 50
Vipera, 221
Vipera ammodytes, 249
Vipera aspis, 239
Vipera berus, 230
Vipera latastii, 247
Vipera lebetina, 257
Vipera macrops, 224
Vipera prester, 235
Vipera renardi, 227
Vipera ursinii, 221
Viperine Water-Snake, 165
viperinus (Tropidonotus), 165
viridiflavus (Coluber), 170
Viscera, 77
vivax (Ailurophis), 217
vosseleri (Tropidonotus tessellatus, var.), 162
vulgaris (Natrix), 152
Whip-Snake, 170
Worms, 110
Xenopeltidæ, 5
Zamenis, 170
Zamenis carbonarius, 172
Zamenis dahlii, 177
Zamenis gemonensis, 170
Zamenis hippocrepis, 179
Zamenis sardus, 172
Zoogeographical Regions, 119
|
For a key to the identification of the species, see above, p. 22. |
|
The only specimen with twenty-one rows I have examined is a male from Albano, near Rome (Genoa Museum). |