This eBook is for the use of anyone anywhere at no cost and with almost no restrictions whatsoever. You may copy it, give it away or re-use it under the terms of the Project Gutenberg License included with this eBook or online at www.gutenberg.org
Title: The Dyeing of Woollen Fabrics
Author: Franklin Beech
Release Date: December 1, 2006 [eBook #19985]
Last Updated: March 14, 2015
Language: English
Character set encoding: ISO-8859-1
***START OF THE PROJECT GUTENBERG EBOOK THE DYEING OF WOOLLEN FABRICS***
|
Transcriber's note: Obvious printer's errors have been corrected, and the original spelling has been retained. Additional notes are at the end of the text. |
In this little book the author has endeavoured to supply the dyer of woollen fabrics with a conveniently arranged handbook dealing with the various branches of the wool dyeing industry, and trusts that it will be found to meet the want which undoubtedly exists for such a book.
The text on which the book is based is expressed in the title "The
Dyeing of Woollen Fabrics," and in enlarging upon it the author has
endeavoured to describe clearly and in some detail the various
processes and operations generally, pointing out the principles
involved and illustrating these by numerous recipes, showing the
applications of a great variety of dyes in the production of the one
thousand and one tints and shades the wool dyer is called upon to
produce on the fabrics with which he is working. In pursuance of this
plan nothing is said of the composition and properties of the various
dyes, mordants, chemicals, etc., which are used. This is information
every wool dyer should possess, but the author believes it is better
dealt with in books devoted to Chemistry proper.
May, 1902.
CHAPTER III.
Dyeing Machinery and Dyeing Manipulations--
CHAPTER IV.
The Principles and Practice of Wool Dyeing--
CHAPTER VI.
CHAPTER VIII.
CHAPTER IX.
Fig.
1. Microscopical Sketch of Wool Fibre.
3. Sectional View of Wool Fibre.
4. Wool Fibres Showing Action of Alkalies.
5. Wool Fibres Showing Action of Acids.
7. Wool Cloth Washing Machine.
8. Woollen Cloth Washing Machine.
12. Delahunty's Dyeing Machine.
14. Holliday's Yarn Dyeing Machine.
15. Klauder-Weldon Yarn Dyeing Machine.
20. Plush Fabric Dyeing Machine.
22. Hawking Machine.
26. Cloth Washing Machine. (p. viii)
28. Soaping and Washing Machine.
29. Hydro-extractor.
30. Hydro-extractor.
33. Experimental Dye Apparatus.
Wool is one of the most important textile fibres used in the manufacture of woven fabrics of all kinds. It belongs to the group of animal fibres of which three kinds are met with in nature, and used in the manufacture of textile fibres; two of these are derived from quadruped animals, such as the sheep, goat, etc., while the third class comprises the products of certain insects, e.g., silk.
The skin of all animals is covered with more or less of a fibrous coat, which serves as a sort of protecting coat from the weather to the skin underneath. Two different kinds of fibres are found on animals; one is a stiff kind of fibre varying in length very much and called hairy fibres, these sometimes grow to a great length. The other class of animal fibres are the woolly fibres, short, elastic and soft; they are the most esteemed for the manufacture of textile fabrics, it is only when the hairy fibres are long that they are serviceable for this particular purpose. There is a slight difference in the structure of the two kinds of fibre, woolly fibres having a more scaly structure than hairy fibres; the latter also differ in being more cylindrical in form.
Wool.--By far the most important of the animal fibres is wool, the fibre of the domestic sheep. Other animals, the llama or alpaca, the Angora and Cashmere goats also yield fibres of a similar character, which are imported under the name (p. 002) of wools. There are many varieties of wools Which are yielded by the various breeds of sheep, but they may be roughly divided into two kinds, according to the length of "staple," as it is called. In the long-stapled wools the fibres average from 7-1/2 to 9-1/2 inches in length, while the short-stapled wools vary from 1 to 2 inches long. The diameter varies very considerably from 0.00033 to 0.0018 of an inch.

Two varieties of thread are spun from wool, one is known as "worsted," the other as "woollen" yarns; from these yarns two kinds of cloths are woven, distinguished as worsted and woollen cloths; the former are in general not subjected to any milling or felting process, while the latter invariably are.
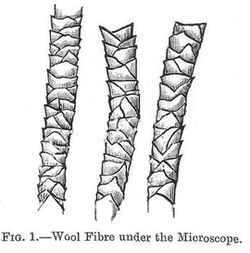
Physical Properties.--When seen under the microscope the wool fibres show a rod-like structure covered with broad scales, the edges of which project from the body of the fibre, and all point in one direction.
Fig. 1 shows typical wool fibres as viewed under the microscope; the sketch shows very well the scales.
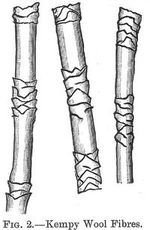
The shape of the scales varies in different breeds of wool. The (p.003) outer scales enclose inner medullary cells, which often contain pigment matter. A transversed section of the wool fibre shows the presence of a large number of cells. Sometimes wool fibres are occasionally met with which have a peculiar white horny appearance; these do not felt or dye well. They are known as "kempy" fibres. See figure 2. The microscope shows that they are largely devoid of structure, and are formed of very horny, impenetrable tissue, which is difficult to treat in the milling or dyeing process.
The curly or twisted character of the fibre is caused by the unequal contraction of the outer scales, and depends in a great measure upon the hygroscopic nature of the wool. It may be entirely removed for the time by wetting the wool in hot water, then drying it in a stretched condition, or the curl may be artificially induced by unequal drying, a fact which is turned to practical account in the curling of feathers and of hair.
The amount of curl in different varieties of wool is very variable, being as a rule greatest in the finer qualities, and diminishing as the fibre becomes coarser. The diameter of the (p. 004) wool fibre varies from 1/2000 to 1/5000 of an inch, and the number of curls from about 30 per cent. In fine wool as little as 1 or 2 per cent. in the thicker fibres.
Elasticity and strength are properties which, in common with silk, wool possesses in a greater degree than the vegetable fibres. When submitted to strain the wool fibre exhibits a remarkable strength, and when the breaking point is reached the fracture always takes place at the juncture of two rings of the outer scales, the embedded edges of the lower layer being pulled out of their seat. The scales themselves are never broken.
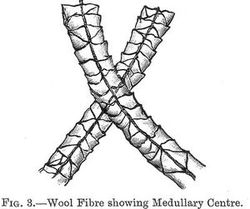
When first formed the cells are more or less of a spherical shape, and contain a nucleus surrounded by the ultimate photoplasmic substance. Those cells which constitute the core or central portion of the fibre retain to some extent this original globular form and pulpy condition. Surrounding this central portion or medulla, as it has been called (see fig. 3), and forming the main bulk of the fibre, there is a comparatively thick layer of partially flattened cells, which are also elongated in the direction of the length of the fibre, and outside this again there is a thinner stratum which may be compared to the bark of a tree. This outer covering differs materially (p. 005) from the rest of the fibre in its physical structure, but is, probably, nearly identical with it, though possibly not entirely so, in chemical composition. It consists of a series of flattened horny scales, each being probably an aggregation of many cells. The scales, which have been compared to the scales of a fish or to slates on a housetop, overlap each other, the free edges protruding more or less from the fibre, while the lower or covered edges are embedded and held in the inner layer of cells. The free edges always point away from the root of the fibre, just as do the bracts of a fir cone.
When viewing a section of a wool fibre there is, of course, no sharp line of division between the three portions above described, but the change from the central spherical cells to the elongated cellular portion, and from these again to the flattened horny scales, is quite gradual, so that the separation into zones, though well marked, is very indefinite in respect of boundaries.
The scaly structure of wool is of great importance in regard to what is known as felting property. When woollen fabrics are worked in boiling water, especially in the presence of soap, they shrink in length and breadth, but become thicker in substance, while there is a greater amalgamation of the fibres of the fabric together to form a more compact and dense cloth; this is due to the scaly structure of the wool fibres enabling them to become entangled and closely united together. In the manufacture of felt hats this is a property of very great value.
Variations in Physical Structure.--Wool fibres vary somewhat amongst themselves; fibres from different breeds of sheep, or even from different parts of the same animal, vary greatly, not only in thickness, length, etc., but also in actual structure. A typical wool fibre, such as may be obtained a good merino or Southdown fleece, will possess the typical structure described above, but frequently the type is departed (p. 006) from to such an extent that the central core of globular cells is entirely absent. Also the serrated character of the outermost layer of cells reaches a much higher state of development in some samples of wool than in others.
Wool is a much more hygroscopic fibre than cotton or any of the other vegetable fibres, usually it contains about 18 per cent. of water, but much depends upon the atmospheric conditions that prevail. This water is contained in the wool in two forms: (1) as water of hydration amounting to about 81 per cent., and (2) as hygroscopic water.
Experiments have shown that when a piece of dried wool is exposed to an atmosphere saturated with water vapour it will absorb 50 per cent. of its weight; cotton under the same conditions will take up 23 per cent.; flax, 27·5 per cent.; jute, 28·5 per cent., and silk, 36·5 per cent.
Heated to about 100° C. it parts with nearly the whole of its water and becomes hard, horny and brittle, exposed to the air, the dry wool again absorbs water and is restored to its former condition. When heated to 100° C. wool becomes somewhat plastic, so that whatever form is then imparted to it it will retain when it becomes cold, this property is very useful in certain processes of finishing wool fabrics, making hats, etc.
Chemical Composition.--In the natural or raw state each wool fibre is surrounded by a considerable amount of foreign matter, so that in treating of its chemical constitution it is necessary to distinguish between pure wool and the raw fibre. The incrusting substance is technically known as "Yolk," or "Suint," and is principally composed of a kind of natural soap, consisting of the potash salts of certain fatty acids, together with some fats which are incapable of saponification.
The amount of yolk present upon different samples of wool varies greatly, the finer varieties containing, as a rule, a larger proportion than the coarser, and less valuable sorts.
The (p. 007) variation in the relative amount of pure fibres and yolk is well shown in the following analyses which, however, do not by any means represent extreme cases.
ANALYSES OF RAW MERINO WOOL. DRIED AT 100° C.
| No. 1. | No. 2. | |||
| Moisture | 6·26 | 10·4 | ||
| Yolk | 47·30 | 27·0 | ||
| Pure Wool | 30·31 | 59·5 | ||
| Dirt | 11·13 | 3·1 | ||
| ------- | ------- | |||
| 100·00 | 100·00 |
Yolk consists very largely of two complex substances which have been termed wool perspiration and wool fat. The former is composed of the potash salts of fatty acids, principally oleic and stearic acids; the latter of the neutral carbohydrate, cholesterine, with other similar bodies. The wool perspiration may be removed by a simple washing with water, and on the Continent forms a valuable source of potash salts, since the ash after ignition contains 70 to 90 per cent. of potassium carbonate. The wool fat is insoluble in water, but dissolves readily in ether, benzene, carbon disulphide, etc.
It is also removed from the wool by a treatment with alkali, and it is not easy to explain the action in the case, since the wool fat is not a glyceride, and will not form a soap, but is probably emulsified by the wool perspiration.
Chemical Composition of the Pure Fibre.--The following analyses of purified and dried wool fibre indicate its percentage composition:--
Carbon
50·5
per cent.
50·8
per cent.
Hydrogen
6·8
"
7·2
"
Nitrogen
16·8
"
18·5
"
Oxygen
20·5
"
21·2
"
Sulphur
5·4
"
2·3
"
-------
-------
100·00
100·00
It (p. 008) is sometimes stated that wool fibre consists of a definite substance, keratine, but this view cannot now be admitted, since wool appears to be composed of a mixture or combination of several very complex substances. It is possible and even probable that the outer epidermal scales have a somewhat different composition to the bulk of the fibre, but whether that is the case or not is not known with any degree of certainty, this much can be asserted, that wool is not a simple definite chemical compound.
Sulphur is by far the most variable constituent of wool, sometimes as little as 1·5 and occasionally as much as 5 per cent. being found. It appears to be always present in two different forms, one portion being in very feeble combination and easily removed by alkalies, the remainder, which, according to Knecht, amounts to about 30 per cent. of the total sulphur, cannot be removed without complete disintegration of the fibre. This latter portion does not give a black coloration with plumbite of soda.
The amount of ash left on incinerating dry wool varies from 1 to 2 per cent., and some have considered this inorganic matter as an essential constituent. It consists principally of salts of potassium, calcium and aluminum, with, of course, sulphur.
The chemical composition of the wool fibre is evidently of a most complicated nature; judging from its behaviour in dyeing it is evident that it may contain two bodies, one of a basic character which enables it to combine with the azo and acid series of dyes, the other possessing acid characters enabling it to combine with the basic dyes of the magenta and auramine type. Dr. Knecht has isolated from the wool fibre by extraction with alkalies and precipitation with acids a substance to which the name of lanuginic acid has been given. It is soluble in hot water, precipitates both acid and basic colouring matters in the form of coloured lakes. (p. 009) It yields precipitates with alum, stannous chloride, chrome alum, silver nitrate, iron salts, copper sulphate. It appears to be an albuminoid body. From its behaviour with the dyes, and with tannic acid and metallic salts, it would appear that lanuginic acid contains both acidic and basic groups. It contains all the elements, carbon, hydrogen, oxygen, nitrogen and sulphur, found in wool.
If wool is dyed in a dilute solution of Magenta (hydrochloride of rosaniline), the whole of the base (rosaniline) is taken up, and the whole of the acid (HCl) left in the bath, not, however, in the free state, but probably as NH4Cl, the ammonia being derived from the wool itself. A further proof of the acid nature of lanuginic acid is that wool may be dyed a fine magenta colour in a colourless solution of rosaniline base; for since rosaniline base is colourless, and it only forms a colour when combined with acids, the fibre has evidently acted the part of an acid in the combination.
Chemical Properties. Action of Alkalies.--Alkalies have a powerful action on wool, varying, of course, with the nature of the alkali, strength of solution and temperature at which the action takes place.
An ammoniacal solution of copper hydroxide (Schweizer's reagent), has comparatively little action in the cold, but when hot it dissolves wool fairly readily.
The caustic alkalies; sodium hydroxide, NaOH, or potassium hydroxide KOH, have a most deleterious action on wool. Even when very dilute and used in the cold they act destructively, and leave the fibre with a harsh feel and very tender, they cannot therefore be used for scouring or cleansing wool. Hot solutions, even if weak, have a solvent action on the wool fibre, producing a liquid of a soapy character from which the wool is precipitated out on adding acids.
This action of alkalies has an important bearing on the scouring of wool, for if this operation be not carried out with due (p. 010) care there is in consequence great liability to impair the lustre and strength of this fibre. From microscopical examination this effect of alkalies is seen to be due to the fact that they tend to disintegrate the fibre, loosen and open the scales, this is shown by contrasting the two fibres A and B shown in figure 4, A being a normal wool fibre, B one strongly treated with an alkali.
The alkaline carbonates have but little action on wool, none if used dilute and at temperatures below 120° F.
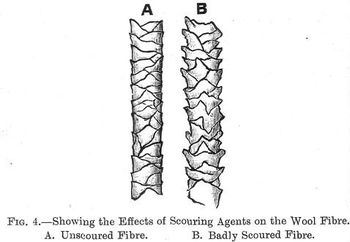
Soap has practically no action on wool, and is therefore an excellent scouring material for wool. The carbonate of ammonia is the best and has the least action of the alkaline carbonates, those of potash and soda if used too strong or too hot have a tendency to turn the wool yellow, the carbonate of potash leaves the wool softer and more lustrous than the carbonate of soda.
The influence of scouring agents on wool will be discussed in the chapter on cleansing wool fabrics in more detail.
Caustic or quick-lime has a similar injurious action on the wool fibre as the caustic alkalies.
Action (p. 011) of Acids.--Acids when dilute have but little influence on the wool fibre, their tendency is to cause a separation of the scales (see fig. 5) of the wool and so make it feel harsher. Strong acids have a disintegrating action on the wool fibre. There is a very considerable difference between the action of acids on wool and on cotton, and this difference of action is taken advantage of in the woollen industry to separate cotton from wool by the process commonly known as "carbonising," which consists in treating the fabric with a weak solution of hydrochloric acid or some other acid, then drying it; the cotton is disintegrated and falls away in the form of a powder, while the wool is not affected, sulphuric acid is used very largely in dyeing wool with the acid- and azo-colouring matters.
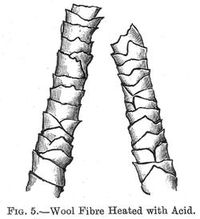
Nitric acid affects wool in a very similar manner to the acids named above when used in a dilute form; if strong it gives a deep yellow colour and acts somewhat destructively on the fibre.
Sulphurous acid (sulphur dioxide) has no effect on the actual fibre, but exercises a bleaching action on the yellow colouring matter which the wool contains, it is therefore largely (p. 012) used for bleaching wool, being applied either in the form of gas or in solution in water; the method will be found described in another chapter. Wool absorbs sulphur dioxide in large amount, and if present is liable to retard any subsequent dyeing processes.
Action of Other Substances.--Chlorine and the hypochlorites have an energetic action on wool, and although they exert a bleaching action they cannot well be used for bleaching wool. Hot solutions bring about a slight oxidation of the fibre, which causes it to have a greater affinity for colouring matters; advantage is taken of this fact in the printing of delaines and woollen fabrics, while the woollen dyer would occasionally find the treatment of service. A paper by Mr. E. Lodge, in the Journal of the Society of Dyers and Colourists, 1892 (p. 41), may be consulted with advantage on this subject. Wool treated with chlorine loses its felting property, and hence becomes unshrinkable, a fact of which advantage is taken in preparing unshrinkable woollen fabrics.
When wool is boiled with solutions of metallic salts, such as the sulphate of iron, chrome, aluminium and copper, the chlorides of tin, copper and iron, the acetates of the same metals, as well as with some other salts, decomposition of the salt occurs and a deposit of the metallic oxide on the wool is obtained with the production of an acid salt which remains in solution. In some cases this action is favourably influenced by the presence of some organic acid or organic salt, as, for examples, oxalic acid and cream of tartar (potassium tartrate), along with the metallic salt.
On this fact depends the process of mordanting wool with potassium bichromate, alum, alumina sulphate, ferrous sulphate, copper sulphate, etc. The exact nature of the action which occurs is not properly understood, but there is reason for thinking that the wool fibre has the capacity of assimilating both the acid and the basic constituents of the salt employed.
Excessive (p. 013) treatment with many metallic salts tends to make the wool harsh to the feel, partly owing to the scales being opened out and partly owing to the feel naturally imparted by the absorbed metallic salt.
The normal salts of the alkaline metals, such as sodium chloride, potassium sulphate, sodium sulphate, etc., have no action whatever on the wool fibre.
Wool has a strong affinity for many colouring matters. For some of the natural colours, turmeric, saffron, anotta, etc., and for the neutral and basic coal-tar colours it has a direct affinity, and will combine with them from their aqueous solutions. Wool is of a very permeable character, so that it is readily penetrated by dye liquors; in the case of wool fabrics much depends, however, upon the amount of felting to which the fabric has been subjected.
If wool be boiled in water for a considerable time it will be observed that it loses much of its beautiful lustre, feels harsher to the touch, and also becomes felted and matted together. This has to be carefully guarded against in all dyeing operations, where the handling or moving of the yarns is apt to produce this unfortunate effect.
After prolonged boiling the fibre shows signs of slight decomposition, from the traces of sulphuretted hydrogen and ammonia gases which it evolves.
When wool is dried at 212° F. it assumes a husky, harsh feel, and its strength is perceptibly impaired. According to Dr. Bowman, the wool fibre really undergoes a slight chemical change at this temperature, which becomes more obvious at 230° F., while at about 260° F. the fibre begins to disintegrate. According to the researches of Persoz, however, temperatures ranging from 260° F. to 380° F. can be employed without any harm to the wool, if it has previously been soaked in a 10 per cent. solution of glycerine.
When wool is heated to 212° F. (100° Cent.) it becomes quite (p. 014) pliant and plastic and may be moulded into almost any shape, which it still retains when cold. This fact is of much interest in the processes of finishing various goods, of embossing velvet where designs are stamped on the woven fabric while hot, and in the crabbing and steaming of woollen goods, making hats, etc.
Wool scouring takes place at two stages in the process of manufacture into cloth. First, in the raw state, to free the wool from the large amount of grease and dirt it naturally contains; second, after being manufactured into cloth, it is again scoured to free it from the oil that has been added to the scoured raw wool to enable it to spin easily. This oiling is generally known as wool batching, and before the spun yarns or woven fabrics can be dyed it is necessary to remove it.
Raw wool is a very impure substance, containing comparatively little wool fibre, rarely more than 50 to 60 per cent. in the cleanest fleeces, while it may be as low as 25 per cent. in the dirtiest.
First there is a small quantity of dirt; there is what is called the suint, a kind of soapy matter, which can be removed by washing in hot water. This soap has for its base potash, while its acids are numerous and complex. The wool contains a fatty-like substance of the nature of wax, called cholesterine, and this imparts to the fatty matter, which be extracted from the wool fibre, very peculiar properties. Besides these there are several other bodies of minor importance, all of which have to be removed from the wool before it can be manufactured into cloth.
Marker (p. 016) and Schulz give the following analysis of a good sample of raw wool:--
Moisture
23·48
per cent.
Wool fat
7·17
"
Wool soap (suint), soluble in water
21·13
"
Soluble in alcohol
0·35
"
Soluble in ether
0·29
"
Soluble in dilute hydrochloric acid
1·45
"
Wool fibre
43·20
"
Dirt
2·93
"
-------
100·00
Two principles underlie the methods which are in use for this purpose. The first principle and the one on which the oldest method is based is the abstraction of the whole of the grease, etc., from the wool by means of an alkaline or soapy liquor at one operation. This cannot nowadays be considered a scientific method. Although it extracts the grease, etc., from the wool, and leaves the latter in a good condition for after processes, yet with it one might almost say that the whole of the soap or alkali used, as well as the wool grease itself, is lost as a waste product; whereas any good process should aim at obtaining the wool grease for use in some form or another. The second principle which underlies all the most recent methods for extracting the grease from the wool, consists in treating the fibre with some solvent like benzol, carbon bisulphide, petroleum spirit, carbon tetrachloride, etc., which dissolves out the cholesterine and any other free fatty matter which is in the wool fibre, leaving the latter in such a condition that by washing with water the rest of the impurities in the wool can be extracted. By distilling off and recondensing the solvent can be recovered for future use, while the wool fat can also be obtained in a condition to use for various purposes. This is rather a more scientific method than the old one, but it has not as yet come into extensive use.
Wool (p. 017) Scouring. Old Methods.--In the early days of wool scouring this operation was done in a very primitive fashion, generally in a few tubs, which could be heated by steam or otherwise, and in which wool was worked by means of hand forks. These primitive processes are still in use in some small works, especially where the wool is dyed in the loose condition, but in all the large works machinery has been adopted, which machinery has been brought to a high state of perfection, and does its work very well, and without much attendant manual labour.
The alkaline substances used in this process of scouring demand some notice. These comprise soda ash, soda crystals, caustic soda, silicate of soda, potash, caustic potash, soaps of various kinds, stale urine, ammoniacal compounds. Which of these may be used in any particular case depends upon a variety of reasons. Potash is the best alkaline agent to use. It agrees better with the fibre than any other, leaving it soft and elastic. Ammonia is the next best, but it does not take out the grease as well as the potash. Soda does not suit as well as potash, as it has a tendency to leave the fibre harsh in feel and somewhat brittle, yet on account of its being so much cheaper it is the most largely used. The use of silicate of soda cannot be recommended, as it has a great tendency to leave the fibre hard, which may be ascribed to the deposition of silica on the fibre.
The caustic alkalies cannot be used as they have too solvent an action on the fibre. The carbonates, therefore, in the form of soda ash or potash, or pearl ash, are used, or better still, soap is used as it has a greater solvent action on the fatty matter of the wool than have the alkalies, and in this respect a potash soap is better than a soda soap.
The character of the wool determines the alkali to be used; fine, long-stapled wools, which are usually very free from grease in excess, should always be treated with potash, or (p. 018) a potash soap, which will remove the whole of the grease from the wool, leaving the latter in a fine, soft, silky condition.
Short-stapled wools can be treated with soap and a little soda ash, but too much of the latter is to be avoided. Coarse and greasy wools may be scoured with soap and soda ash, or other alkali which is almost necessary to remove the large amount of grease these wools contain.
Practically the only alkaline products now in use are the various hard and soft soaps, and the carbonates of soda and potash in their various forms of soda ash, soda crystals, potashes, pearl ash, etc. Ammonia and its compounds are rarely used, while stale urine, which acts in virtue of the ammonia it contains has practically gone out of use.
Hand-Scouring.--Wool scouring by hand is easily done and requires few appliances, simple tubs or vats of sufficient capacity in which steam pipes are placed, so that the scouring liquors can be heated up. The best temperatures to use are about 130° to 140° F., and it is not advisable to exceed the latter, as there is then some risk that the alkali may act on the fibre too strongly.
The strength of the scouring liquor necessarily varies with the kind of wool being treated, and with the kind of alkaline product used; if soft, fine wools are being treated, then the liquor may be made with 1 to 2 lb. of soap to 10 gallons of water (if a mixture of soap and alkali is used, then it may contain from 1/4 to 1/2 lb. soda ash, and 1/2 lb. to 1 lb. of soap). For coarse, greasy wools these quantities may be increased by about one-half. The best plan of scouring by hand is to treat the wool in a tub with a scouring liquor for about half an hour, then to squeeze out the surplus liquor and to treat again in a new liquor for half an hour; this liquor may be used for a new batch of wool. The wool is often put into nets, and these are lifted up and down in the liquor so as to cause it to penetrate to every part of the wool.
It (p. 019) is not advisable to work the wool about too much, otherwise felting might ensue and this must be avoided. The felting of the wool is one of the troubles of the wool-scourer and is often difficult to avoid, it is mostly brought about by excessive working of the wool during the process, and by the use of too high a temperature in the scouring bath. The remedies are obvious to the practical man, as little handling of the wool as possible, and at as low a temperature as possible. Still it is necessary to see that the scouring liquor penetrates to every part of the wool which is being treated.
To ensure this, care must be taken not to scour too much at one time, so that the wool is loosely placed in the scouring tub, if placed loose in the latter, the workmen can by means of forks work it to and fro while in process of treatment. After the wool has been through these scouring liquors it is thrown on a scray to drain, and is next placed in cisterns which have perforated false bottoms. In these cisterns it is washed with cold water two or three times, the water being run off from the wool between each washing; it is then spread out in a room to dry. As a rule, a man can wash from 500 lb. to 600 lb. of wool in a day by this method. Another plan which is sometimes adopted so as to avoid handling the wool as much as possible, and thus prevent felting, is to place the wool in cages having perforated sides which will hold about 1 cwt. of wool. They are lowered by means of cranes into the washing liquors, and the wool in them is then worked for a quarter of an hour, when the cages and their contents are lifted out and the surplus liquor allowed to drain off. They are then lowered into the next bath, treated or worked in this, again lifted out and dropped into the wash waters.
There is by this plan a saving of handling, and more wool can be got through in the same time, but it requires two men to work it. These hand processes are only in use in small (p. 020) works, having been replaced in all large works by mechanical methods described below.
Machine Scouring.--Wool-scouring machinery has been brought to a high state of perfection by the successive efforts of many inventors, and by their means wool washing has been much simplified and improved. Wool-washing machinery is made by several firms, among whom may be mentioned Messrs. J. & W. McNaught, and John Petrie, Junior, Limited, both of Rochdale.
(p. 021)Fig. 6 shows one form of wool-washing machine. It consists of a long trough which contains the scouring liquor. In this machine the wool enters at the left-hand end, and is seized by a fork or rake and carried forward by it a short distance, then another rake seizes it and carries it further forward to another rake, and this to the last rake of the machine, which draws it out of the machine to a pair of squeezing rollers which press out the surplus liquor, and from these rollers the scoured wool passes to a travelling band for delivery from the machine. Sometimes the wool is not entered into the trough direct, but is put on a travelling apron which opens it and delivers it in a more open form into the trough. The movement of the forks causes some degree of agitation in the scouring liquor which facilitates the penetration of the liquor through the wool, and thus brings about a better scouring.
After the wool has passed through the machine it is taken and run once more through the machine. Some scourers use the same liquor, but it is better to use fresh liquors, after which it is washed in the same machine with water two or three times. With a single machine there is some time and labour lost in transferring the wool from one end to the other between the separate treatments, and in large works where a great deal of wool is scoured it is usual to place three or four of these machines end to end.
The (p. 022) first is filled with strong scouring liquor, the second with a weaker liquor, while the third and fourth contains wash waters, and the wool is gradually passed by the action of the machine through the series without requiring any manual aid. Between each machine it is passed through squeezing rollers as before, and finally emerges thoroughly scoured. A good plan of working in connection with such a series of machines is to have four as above, two washing machines and two soaping machines, the soap liquor is run through these in a continuous stream, entering in at the delivery end of the second soaper and passing out at the entering end of the first soaper. The wool as it first enters the machine comes into contact with rather dirty soap liquor, but this suffices to rid it of a good deal of loose dirt; as it passes along the machine it comes in contact with cleaner and fresher soap liquor, which gradually takes all grease and dirt out of it, and, finally, when it passes out it comes in contact with fresh liquor, which removes out the last traces of dirt and grease. In the same way it passes through the washers, being treated at the last with clean water. By this plan the scouring is better done, while there is some saving of soap liquor and wash water, for of these rather less is required than by the usual system. These are matters of consideration for wool scourers. The wool-washing liquors after using should be stored in tanks to be treated for recovery of the grease which they contain.
The temperature of the scouring liquors should be about 100° F., certainly not more than 120° F., high temperatures are very liable to bring about felting, while tending to increase the harshness of the wool, particularly when soda is the agent used. By this method all the wool fat, suint, etc., of the wool find their way into the soap liquors. These were formerly thrown away, but they are generally treated with acid and the fat of the soap and wool recovered, under the name (p. 023) of wool grease or Yorkshire grease. (Vide G. H. Hurst, "Yorkshire Grease," Jour. Soc. Chem. Ind., February, 1889.)
The wool fat consists largely of a peculiar fat-like body known as cholesterine. This, however, is unsaponifiable, and cannot be made into soap; at the same time when it gets into, as it does, the recovered wool grease it spoils the latter for soap-making purposes.
Cholesterine has some properties which make it valuable for other purposes; it is a stable body not prone to decomposition, it is capable of absorbing a large quantity of water, and it is on these accounts useful for medicinal purposes in the production of ointments, and it might be useful in candle-making. When it gets into recovered grease it cannot be extracted from it in an economical manner. The wool suint consists largely of the potash soaps of oleic and stearic acids. These two fatty acids find their way into the recovered wool grease but the potash salts are lost, while they would be valuable for various purposes if they could be recovered.
Another form of wool-washing machine has a frame carrying a number of forks arranged transversely to the machine. The forks are by suitable gearing given a motion which consists of the following cycle of movements. The forks are driven forwards in the trough of the machine, carrying the wool along with them, they are then lifted out, carried back, and again allowed to drop into the machine, when they are ready to go forward again. Thus the forks continually push the wool from one end of the machine to the other.
It is a common plan to have three machines placed end to end, so that the wool passes from one to the other; in a set of this kind the first machine should have a capacity of 1,500 gallons or thereabouts, the second 1,000 gallons, and the third 750 gallons.
Wool Scouring by Solvents.--Of late years processes have (p. 024) been invented for the scouring of wool, either raw or spun by means of solvents, like carbon bisulphide, benzol, petroleum spirit, etc. Such processes are in a sense rather more scientific than the alkali processes, for whereas in the latter the grease, etc., of the wool and the oil used in batching it are practically lost for further use, and therefore wasted, being thrown away very often, although they may be partially recovered from the used scouring liquors, in the solvent processes the grease and oil may be recovered for future use for some purpose or other.
The great objection to these processes is the danger that attends their use, owing to the inflammable character of the solvents. Several other objections may be raised, some of which are mechanical, and due to the want of proper machinery for carrying out the processes. There are many ways in which solvents may be applied, some are the subject of patents. It is not possible to describe the details of all these, but two of the most recent will be mentioned.
In Singer's process, which was described in detail by Mr. Watson Smith some time ago before the Society of Dyers and Colourists, carbon bisulphide is used. The raw wool is placed between two endless bands of wire, and it is carried through a series of troughs containing bisulphide of carbon; during its passage through the troughs the solvent takes out the grease, and loosens the other constituents of the wool. After going through the bisulphide the wool is dried and passed through water which completes the process. The carbon bisulphide that has been used is placed in steam-heated stills, distilled off from the grease, condensed in suitable condensers, and used over again. In this process, with care, there is very little loss of solvent. The grease which is recovered can be used for various purposes, one of which is the manufacture of ointments, pomades, etc. The disadvantages of bisulphide are: (1) It tends after some time to (p. 025) cause the wool to acquire a yellow cast, due to the free sulphur which it contains, and which being left in the wool gradually causes it to turn yellow. By using redistilled bisulphide this defect may be avoided. (2) Another defect is the evil odour of the solvent. This, however, is less with redistilled bisulphide than with the ordinary quality, and with suitable apparatus is not insuperable. (3) Another defect is the volatility and inflammability of carbon bisulphide. On the other hand, bisulphide possesses the very great advantage of being at once heavier than, and insoluble in, water, and it can be, therefore, stored under water very much more safely than can any of the other solvents which are used.
Burnell's machine has two troughs filled with benzoline. In these are arranged a large central roller round which are some smaller rollers. The wool passes round the large roller and is subjected to a number of squeezings in passing the smaller rollers. A current of the benzoline is continually passing through the machine. The whole is enclosed in a hood to avoid loss of solvent as far as possible. After passing through the benzoline trough the wool passes through a similar trough filled with water. Benzoline is better than carbon bisulphide in that there is no tendency on the part of the wool to turn yellow after its use, on the other hand it is more inflammable, and when it does take fire is more dangerous, and being lighter than water is not so readily and safely stored. Another feature is that it is not so completely volatile at steam temperatures, so that a little may be left in the grease and thus tend to deteriorate it. Coal-tar benzol, the quality known as 90's, would be better to use.
The solvent processes are well worth the attention of wool scourers, all that is required for their proper development being the production and use of suitable machinery.
After the raw wool has been scoured it is batched, i.e., it is (p. 026) mixed with a quantity of oil for the purpose of lubricating the wool to enable it more easily to stand the friction to which it is subjected in the subsequent processes of spinning and weaving by giving it greater pliability.
For this purpose various kinds of oil are used. Olive oil is the principal favourite, the variety mostly used being Gallipoli oil. Ground-nut oil is also extensively employed, and is cheaper than olive. Oleic acid a by-product of the candle industry, is extensively used under the name of cloth oil, there is also used oleine, or wool oil, obtained by the distillation of Yorkshire grease.
So far as merely oiling the wool is concerned there is not much to choose between these different oils, olive perhaps works the best and agrees best with the wool. Mineral oils have been and can be used either alone or mixed with the oils above mentioned, and so far as lubricating the wool is concerned do very well and are much cheaper than the fatty oils named above.
The following are some analyses of various oils used as cloth oils which the author has had occasion to analyse.
| 1. | 2. | 3. | 4. | |
| Specific gravity at 60° F. | 0·9031 | 0·9091 | 0·6909 | 0·8904 |
| Free fatty acid | 55·02 | 64·42 | 51·52 | 68·05 |
| Unsaponifiable oil | 34·56 | 9·95 | 32·80 | 9·52 |
| Saponifiable oil | 10·32 | 25·32 | 15·68 | 12·43 |
| ------ | ------ | ------ | ------ | |
| 100·00 | 100·00 | 100·00 | 100·00 |
Nos. 1 and 2 are prepared from Yorkshire grease. The unsaponifiable matter in these is purely natural, it will be seen it varies within wide limits. Nos. 3 and 4 are made from the oleic acid of the candle factories, and the unsaponifiable matter is due to their containing mineral oil which has been added to them.
So far as regards the object for which the wool is oiled, the mineral oils will answer almost as well as the fatty oils and (p. 027) with most satisfactory results from an economical point of view, for they are much cheaper. But this is not the only point to be considered. The oil has to be got out of the wool before the latter can be dyed. Now while the fatty oils can be easily removed, by treatment with soap, and they can be recovered along with the fat of the soap, mineral oils cannot be entirely removed from the wool, what remains in will interfere very much with the satisfactory dyeing of the wool, and what is got out finding its way into the covered wool grease, spoils this for soap making and other uses, so that on the whole what is gained in lessened cost of oiling is lost by the increased liability to defects in dyeing and consequently depreciation in value of the wool, and to decrease in value of the recovered grease.
The amount of oil used varies from 7 per cent. with the best wools to 15 per cent. with shoddy wools. The scouring agents generally used are the same as those used in loose wool scouring, namely, carbonate of soda for coarse woollen yarns, soap and soda for medium yarns, and soap and ammonia for fine yarns. Prior to treating the yarns it is best to allow them to steep in hot water at about 170° F. for twenty minutes, then to allow them to cool. The actual scouring is often done in large wooden tubs, across which rods can be put on which to hang the hanks of yarn, and in which are placed steam pipes for heating up the bath. The best temperature to treat the yarn at is about 150° F.; too high a temperature must be avoided, as with increased heat the tendency to felt is materially augmented, and felting must be avoided. The hanks are treated for about twenty minutes in the liquor, and are then wrung out, drained, and again treated in new scouring liquor for the same length of time. After rinsing in cold water they are dried and finished.
When the oiled wool has been spun into yarns, whether worsted (p. 028) or woollen, and passes into the hands of the dyer, it is necessary to remove from it all the oil before any dyeing operations can be satisfactorily carried out. This oil is removed by the use of soap and weak alkaline liquors, using these at about 110° to 120° F. The most common way is to have the liquor in a rectangular wooden tank, and hang the hank of yarn in by sticks resting on the edges of the tank; from time to time the hanks are turned over until all the oil has been washed out, then they are wrung out and passed into a tank of clean water to wash out the soap, after which the yarn is ready for dyeing.
When the yarn is of such a character that it is liable to curl up, shrink and become entangled, it is necessary that it be stretched while it is being treated with the soap liquor; this is effected by a stretching apparatus consisting of two sets of rollers connected together by a screw attachment, so that the distance between the two sets of rollers can be varied. The hanks are hung between each pair of rollers, and can be stretched tightly as may be required.
For pressing out the surplus liquor from the hanks of yarn a pair of squeezing rollers is used.
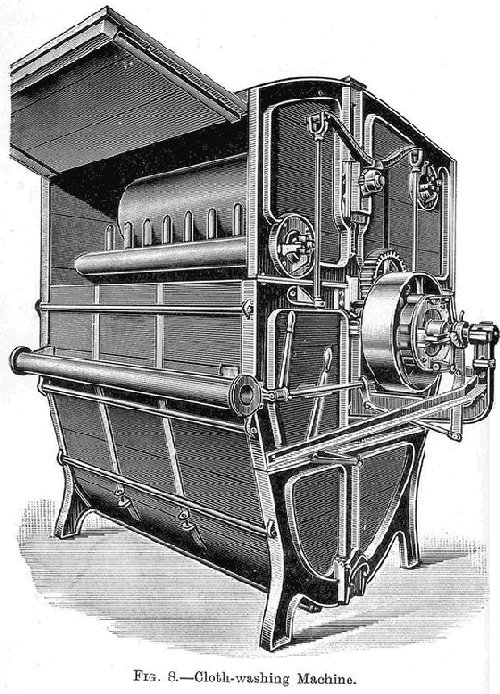
Scouring Woollen Piece Goods.--Very often before weaving the yarns are not scoured to remove the oil they contain, as the weaving is more conveniently done with oily yarn than with a scoured yarn. Before dyeing the oil must be taken out of the pieces, and this can be conveniently done by scouring in a washing machine such as is shown in figures 7 and 8, using soap and soda liquors as before, and following up with a good rinse with water.

The soap liquors used in scouring yarns and pieces become charged with oil, and they should be kept, and the oil recovered from them together with the fatty matter of the soap, by treatment with sulphuric acid. By subjecting the grease or fatty matter so obtained to a boil with caustic soda (p. 029) soap is obtained which may be again used in scouring wool.
Bleaching Wool.--The wool fibre has to be treated very differently from cotton fibre. It will not stand the action of as powerful bleaching agents, and, consequently, weaker ones must be used. This is a decided disadvantage, for whereas with cotton the colouring matter is effectually destroyed, so that the bleached cotton never regains its original colour, the same is not the case with wool, especially with sulphur-bleached wool, here the colouring matter of the fibre is, as it were, only hidden, and will under certain circumstances return. The two materials chiefly used for bleaching wool are sulphur and peroxide of hydrogen.
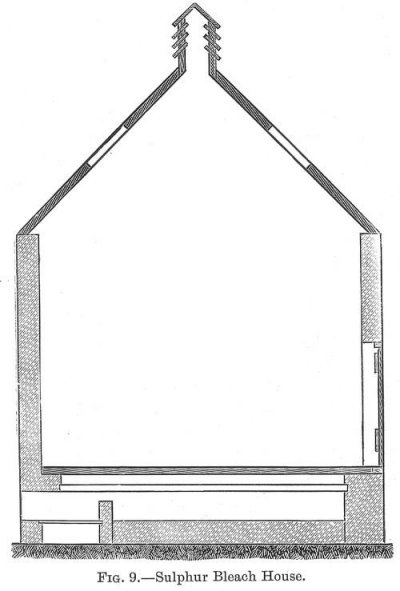
Sulphur (p. 030) Bleaching.--Bleaching wool by sulphur is a comparatively simple process. A sulphur house is built, the usual size being 12 feet high by 12 feet broad, and about 17 feet long. Brick is the most suitable material. The house should have well-fitting windows on two sides, and (p. 031) good tight doors at the ends (see fig. 9). Some houses have a small furnace at each corner for burning the sulphur, two of these furnaces are fitted with hoods, so that the sulphur gases can be conveyed to the upper part of the chamber, but a better plan, and one mostly adopted where the chamber is used for bleaching pieces, is to construct a false perforated bottom above the real bottom of the chamber, the sulphur being burnt in the space between the two floors. If yarn is being bleached the hanks are hung on wooden rods (p. 032) or poles in the chamber, while with pieces an arrangement is constructed so that the pieces which are stitched together are passed in a continuous manner through the chamber.
When all is ready the chamber doors are closed, and the furnaces are heated, some sulphur thrown upon them, which burning evolves sulphur dioxide gas, sulphurous acid, and this acting upon the wool bleaches it. The great thing is to cause a thorough circulation of the gas through every part of the chamber, so that the yarn or pieces are entirely exposed in every part to the bleaching action of the gas. This is effected by causing the gas to pass into the chamber at several points, and, seeing that it passes upwards, to the ventilator in the roof of the chamber. Generally speaking, a certain quantity of sulphur depending upon the quantity of goods being treated is placed in the chamber and allowed to burn itself out; the quantity used being about 6 to 8 per cent. of the weight of the goods. After the sulphuring the goods are simply rinsed in water and dried.
Sulphur bleaching is not an effective process, the colouring matter is not actually destroyed, having only entered into a chemical combination with the sulphur dioxide to form a colourless compound, and it only requires that the wool be treated with some material which will destroy this combination to bring the colour back again in all its original strength; washing in weak alkalies or in soap and water will do this. Another defect of the process lies in sulphur being volatilised in the free form, and settling upon the wool causes it to turn yellow, and this yellow colour cannot be got rid of.
The goods must be thoroughly rinsed with water after the bleaching, the object being to rid the wool of traces of sulphuric acid, which it often contains, and which if left in would in time cause the disintegration of the wool.
Sometimes (p. 033) the wool is washed in a little weak ammonia or soda liquor, but this is not advisable, as there is too much tendency for the colour of the wool to come back again, owing to the neutralising of the sulphur dioxide by the alkali.
Instead of using the gas, the sulphur dioxide may be applied in the form of a solution in water. The goods are then simply steeped for some hours in a solution of the gas in water until they are bleached, then they are rinsed in water and dried. In this method it is important that the solution of the gas be freshly made, otherwise it is liable to contain but little sulphurous acid, and plenty of sulphuric acid which has no bleaching properties, but, on the other hand, is liable to lead to damage of the goods if it be not washed out afterwards.
A better method of utilising the bleaching action of sulphur in a liquid form is to prepare a bath of bisulphite of soda, and acidify it with hydrochloric acid, then to enter the wool, stirring well for some time, and allowing it to steep for some hours, next to expose to the air for a while, and rinse as before.
It is better to allow the wool to steep for about an hour in a simple bath of bisulphite, then enter into a weak hydrochloric acid bath for a few hours. The acid liberates sulphur dioxide in a nascent condition, which then exerts a more powerful bleaching action than if it were already free.
Even with liquid bleaching the bleach is not any more perfect than it is with the gas bleaching; the colour is liable to come back again on being washed with soap or alkali, although there is a freedom from the defect of yellow stains being produced.
Goods properly bleached will stand exposure to air for some considerable time, but those imperfectly bleached exhibit a tendency to regain their yellow colour on exposure to air. One fault which is sometimes met with in sulphur bleaching (p. 034) is a want of softness in the wool, the process seeming to render the fibre harsh.
Washing in a little weak soft soap or in weak soda will remedy this and restore the suppleness of the wool; at the same time care must be taken that the alkaline treatment is not too strong, or otherwise the bleaching effect of the sulphur will be neutralised as pointed out above.
Bleaching Wool by Peroxide of Hydrogen.--During recent years there has come into use for bleaching the animal fibres peroxide of hydrogen, or, as the French call it, oxygenated water. This body is a near relation to water, being composed of the same two elements, oxygen and hydrogen; in different proportions in water these elements are combined in the proportion of 1 part of hydrogen to 8 parts of oxygen, while in the peroxide the proportions are 1 of hydrogen to 16 of oxygen. These proportions are by weight, and are expressed by the chemical formulæ for water H2O, and for hydrogen peroxide H2O2. Water, as is well known, is a very stable body, and although it can be decomposed, yet it requires some considerable power to effect it. Now the extra quantity of oxygen which may be considered to have been introduced into water to convert it into peroxide has also introduced an element of instability, the extra quantity of oxygen being ever ready to combine with some other body for which it has a greater affinity than for the water. This property can be utilised in the bleaching industry with great advantage, true bleaching being essentially a process of oxidation. The colouring matter of the fibre, which has to be destroyed so that the fibre shall appear white, is best destroyed by oxidation, but the process must not be carried out too strongly, otherwise the oxidation will not be confined to the colouring matter, but will extend to the fibre itself and disintegrate it, with the result that the fibre will become tendered and be rendered useless.
Peroxide (p. 035) of hydrogen is a weak oxidiser, and therefore, although strong enough to destroy the colouring matter of the fibre is not strong enough to decompose the fibre itself. Hydrogen peroxide is sold as a water-white liquid, without any odour or taste. Its strength is measured by the quantity of oxygen which is evolved when one volume of the liquid is treated with potassium permanganate; the most common strength is 10 volume peroxide, but 30 and 40 volume peroxide is made. On keeping it loses its oxygen, so that it is always advisable to use a supply up as quickly as possible.
Articles of all kinds can be bleached by simply placing them in a weak solution of the peroxide, leaving them there for a short time, then taking out and exposing to the air for some time. The best plan of applying peroxide of hydrogen is the following: Prepare the bleaching bath by mixing 1 part of peroxide with 4 parts of water. The strength can be varied; for those goods that only require a very slight bleach the proportions may be 1 to 12, while for dark goods the proportions first given may be used. This bath must be used in either a wooden or earthenware vessel. Metals of all kinds must be avoided, as they lead to a decomposition of the peroxide, and therefore a loss of material. To the bath so prepared just enough ammonia should be added to make it alkaline, a condition that may be ascertained by using a red litmus paper, which must just turn blue. Into the bath so prepared the well-scoured goods are entered and worked well, so that they become thoroughly saturated. They are then lightly wrung and exposed to the air for some hours, but must not be allowed to get dry, because only so long as they are moist is the bleaching going on; if they get dry the goods should be re-entered into the bath and again exposed to the air.
If one treatment is not sufficient the process should be repeated. The peroxide bath is not exhausted, and only requires (p. 036) new material to be added to it in sufficient quantity to enable the goods to be readily and easily worked in the liquor. Any degree of whiteness may be obtained with a sufficient number of workings. No further treatment is necessary. It is found in practice that an alkaline bath gives the best results.
Another plan of preparing the bleaching bath is to prepare a bath with peroxide and water as before, then add to a sufficient quantity of a solution of silicate of soda 4 parts of water to 1 of silicate of soda at 100° Tw., to make the bath alkaline. Into this bath the goods are entered and are then exposed to the air as before, after which they may be passed through a weak bath of sulphurous acid, being next well washed in water and dried.
The advantage of bleaching with peroxide is that, as it leaves only water in the goods as the result of action, there is no danger of their becoming tendered by an after development of acid due to defective washing, as is the case with the sulphur bleach. The goods never alter in colour afterwards, because there is nothing left in that will change colour. Some bleachers add a little magnesia to the bath, but this is not at all necessary.
Bleaching with Peroxide of Soda.--Peroxide of soda has come to the front of late for bleaching wool. With it a stronger bleaching bath can be made, while the product itself is more stable than peroxide of hydrogen, only it is needful to keep it in tightly closed metal vessels, free from any possibility of coming in contact with water or organic matter of any kind, or accidents may happen. In order to bleach 100 lb. of wool, a bath of water is prepared, and to this is added 6 lb. of sulphuric acid and then slowly 4 lb. of peroxide of sodium in small quantities at a time. Make the bath slightly alkaline by adding ammonia. Heat the bath to 150° F., enter the wool and allow to remain five to six hours, then (p. 037) rinse well and dry. If the colour does not come out sufficiently white repeat the process.
The Chlorination of Wool.
The employment of chlorine in wool dyeing and wool printing has of late years received an impetus in directions previously little thought of. The addition of a little chlorine to the decoction of logwood has been recommended as increasing the dyeing power of the wool. Treating the wool with chlorine has a material influence in increasing its capacity for taking dye-stuffs, and although but little attention has been paid to this circumstance by wool dyers, yet among wool printers it has come largely into use, and enables them to produce fuller and faster shades than would otherwise be possible.
The method involves the treatment of the wool first with an acid, then with a solution of a hypochlorite. The staple becomes soft and supple and assumes a silky character; in dyeing it shows a greater affinity for the dyes than it did previously. Although not deteriorated in strength, it almost entirely loses its felting properties. On account of this feature the process cannot be adopted for wool which has to be fulled, but it is of service where felting of the goods is to be avoided, for worsteds, underwear, woollen and half woollen hosiery, etc., in which the felting property that occurs on washing is rather objectionable.
By the chloring of the wool the intensity of the shade dyed is increased to such a degree that when dyeing with Acid black, Naphthol black, Naphthol green, Nigrosine, Fast blue, Water blue, and some others dyed in an acid bath, but little more than half the dye used on unchlored wool is required, while with Induline, more even and intense shades are obtained than is otherwise possible.
The operation of chlorination can be done either in one or (p. 038) two baths. The choice depends upon circumstances and the judgment of the dyer. The process by the two-bath method, with subsequent dyeing in the second or separate bath is (for 100 lb. of wool), as follows. The first bath contains, for light cloths, yarn, etc., from 3 to 4 lb. sulphuric acid, 168° Tw., and for heavier cloths and felt, where the penetration and equalisation of the colour is difficult, from 8 lb. to 10 lb. of acid. Generally speaking, a temperature of 170° to 175° F. is sufficient, although for heavy wool and for wool with poor dyeing qualities it is well to use the bath at the boil. The treatment lasts for half an hour, in which time the acid is almost completely absorbed.
The second bath contains a clear solution of 10 lb. bleaching powder, which solution is prepared as follows. Dry bleaching powder of the best quality is stirred in a wooden vat with 70 gallons of water, the mass is allowed to stand, the clear, supernatant liquor is run into the vat and the sediment stirred up and again allowed to settle, the clear liquor being run off as before, and 5 gallons more water is run in. The clear liquors of these three treatments are then mixed together to form the chloring bath. Special care should be taken that no undissolved particles of the bleaching powder should be left in, for if these settle on the wool they result in too great a development of chlorine, which injures the wool.
The goods after being in the acid bath are entered in this chlorine bath at a temperature of 70° F., which is then raised to the boil. If the acid bath has been strong, or been used at the boil, it is perhaps best to rinse the goods before entering into the chlorine bath. The hypochlorous acid disappears so completely from this bath that it may at once be used as the dye-bath, for which purpose it is only necessary to lift the goods, add the required amount of dye-stuff, re-enter the goods and work until the bath is exhausted, which generally happens (p. 039) when acid dyes are used. If a separate dye-bath be preferred, this is made and used as is ordinarily done.
To perform all the operations in one bath the acid bath is made with from 3 to 4 lb. sulphuric acid, and the wool is treated therein for thirty minutes at 170° F., until all the acid has been absorbed. Then the bath is allowed to cool down to 70° or 80° F., the clear bleaching powder solution is added, the goods are re-entered, and the bath is heated to the boil. When all the chlorine has disappeared add the dye-stuff, and dye as directed above.
In printing on wool the chlorination of the wool is a most important preliminary operation. For this purpose the cloth is passed for fifteen minutes at 170° F. through a bath containing 3/4 oz. sulphuric acid per gallon of water. Then it is passed through a cold bath of 3/4 oz. bleaching powder per gallon of water, after which the cloth is rinsed and dried and is then ready for printing.
Another method of chloring the wool is to pass the goods through a bath made with 100 gallons of water, 2 gallons hydrochloric acid and 2 gallons bleaching powder solution of 16° Tw. As some chlorine is given off it is best to use this in a well-ventilated place.
Wool is dyed in a variety of forms, raw, loose wool; partly manufactured fibre in the form of slubbing or sliver; spun fibres or yarns, in hanks or skeins and in warps, and lastly in the form of woven pieces. These different forms necessitate the employment of different forms of machinery and different modes of handling, it is evident to the least unobservant that it would be quite impossible to subject slubbing or sliver to the same treatment as yarn or cloth, otherwise the slubbing would be destroyed and rendered valueless.
In the early days all dyeing was done by hand in the simplest possible contrivances, but during the last quarter of a century there has been a great development in the quantity of dyeing that has been done, and this has really necessitated the application of machinery, for hand work could not possibly cope with the amount of dyeing now done. Consequently there has been devised during the past two decades a great variety of machines for dyeing every description of textile fabrics, some have not been found a practical success for a variety of reasons and have gone out of use, others have been successful and are in use in dye-works.
Hand Dyeing.--Dyeing by hand is carried on in the simplest possible appliances, much depends upon whether the work can be done at the ordinary temperature or at the boil. Figure 10 shows round and oval tubs and a rectangular vat (p. 041) much in use in dye-houses. These are made of wood, but copper dye-vats are also made, these may be used for all kinds of material--loose fibre, yarns or cloth. In the case of loose fibre this is stirred about either with poles or with rakes, care being taken to turn every part over and over and open out the masses of fibre as much as possible in order to avoid matting or clotting together. In the case of yarns or skeins, these are hung on sticks resting on the edges of the tub or vat. These sticks are best made of hickory, but ash or beech or any hard wood that can be worked smooth and which does not swell much when treated with water may be used. The usual method of working is to hang the skein on the stick, spreading it out as much as possible, then immerse the yarn in the liquor, lift it up and down two or three times to fully wet out the yarn, then turn the yarn over on the stick and repeat the dipping processes, then allow to steep in the dye-liquor. This is done with all the batch of yarn that is to be dyed at a time. When all the yarn has been entered into the dye-bath, the first stickful is lifted out, the yarn turned over and re-entered in the dye-liquor; this operation is carried out with all the sticks of yarn until the wool has become dyed of the required depth. In the case of long rectangular vats it is customary for two men, one on each side of the vat, to turn the yarns, each man taking charge of the yarn which is nearest to him.
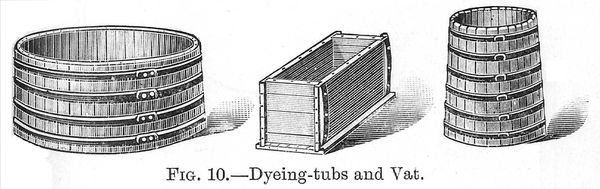
Woven goods may be dyed in the tub or vat, the pieces being drawn in and out by poles, but the results are not altogether (p. 042) satisfactory, and it is preferable to use machines for dyeing piece goods.
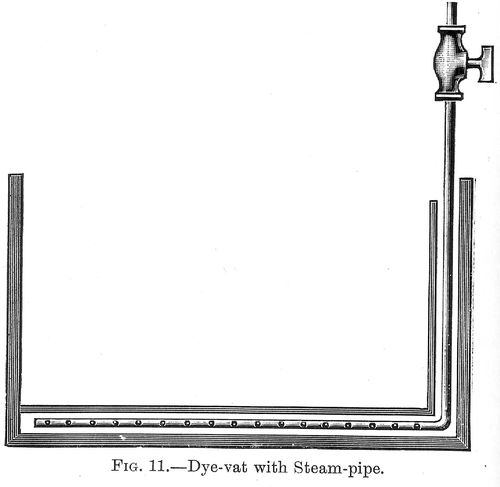
Plain tubs or vats, such as those shown in figure 10, are used for dyeing and otherwise treating goods in the cold, or at a lukewarm heat, when the supply of hot water can be drawn from a separate boiler. When, however, it is necessary to work at the boil, then the vat must be fitted with a steam coil. This is best laid along the bottom in a serpentine form. Above the pipe should be an open lattice-work bottom, which, while it permits the free circulation of boiling water in the vat, prevents the material being dyed from coming in contact with the steam pipe. This is important if uniform shades are to be dyed, for any excessive heating of any portion of the bath leads to stains being produced on the material in that part of the bath. Figure 11 shows a vat fitted with a steam pipe. That portion of (p. 043) the steam pipe which passes down at the end of the vat is in a small compartment boxed off from the main body of the vat, so that no part of the material which is being dyed can come in contact with it. A closed steam coil will, on the whole, give the best results, as then no weakening of the dye-liquor can take place through dilution by the condensation of the steam. Many dye-vats are, however, fitted with perforated, or as they are called, open steam coils, in which case there is, perhaps, better circulation of the liquor in the dye-vat, but as some of the steam must condense there is a little dilution of it.
Dyeing Machines.
Dye-tubs and vats, such as those described above, have been largely superseded by machines in which the handling or working of the materials being dyed is effected by mechanical means. There have been a large number of dyeing machines invented, some of these have not been found to be very practical, and so they have gone out of use. Space will not admit of a detailed account of every kind of machine, but only of those which are in constant use in dye-works.
Dyeing Loose or Raw Wool and Cotton.--Few machines have been designed for this purpose, and about the only successful one is
Delahunty's Dyeing Machine.--This is illustrated in figure 12. It consists of a drum made of lattice work which can revolve inside an outer wooden casing. The interior of the revolving drum is fitted with hooks or fingers, whose action is to keep the material open. One segment of the drum is made to open so that the loose cotton or wool to be dyed can be inserted. By suitable gearing the drum can be revolved, and the dye-liquor, which is in the lower half of the wooden casing, penetrates through the lattice work of the (p. 044) drum, and dyes the material contained in it. The construction of the machine is well shown in the drawing, while the mode of working is obvious from it and the description just given. The machine is very successful, and well adapted for dyeing loose or raw wool and cotton. The material may be scoured, bleached, dyed or otherwise treated in this machine.

The Obermaier Machine, presently to be described, may also be used for dyeing loose cotton or wool.
Dyeing Slubbing, Sliver or Carded Wool.--It is found in practice that the dyeing of loose wool is not altogether satisfactory, the impurities they naturally contain interfere with (p. 045) the purity of the shade they will take. Then again the dyes and mordants used in dyeing them are found to have some action on the wire of the carding engine through which they are passed; at any rate, a card does not last as long when working dyed wools as when used on undyed cotton or wool fibres. Yet for the production of certain fancy yarns for weaving some special classes of fabrics it is desirable to dye the wool before it is spun into thread. The best plan is undoubtedly to dye the fibre after it has been carded and partly spun into what is known as slubbing, or sliver. All the impurities have been removed, the wool fibres are laid straight, and so it becomes much easier to dye. On the other hand, as it is necessary to keep the sliver or slubbing straight and level, no working about in the dye-liquors can be allowed to take place, and so such must be dyed in specially constructed machines, and one of the best of these is the
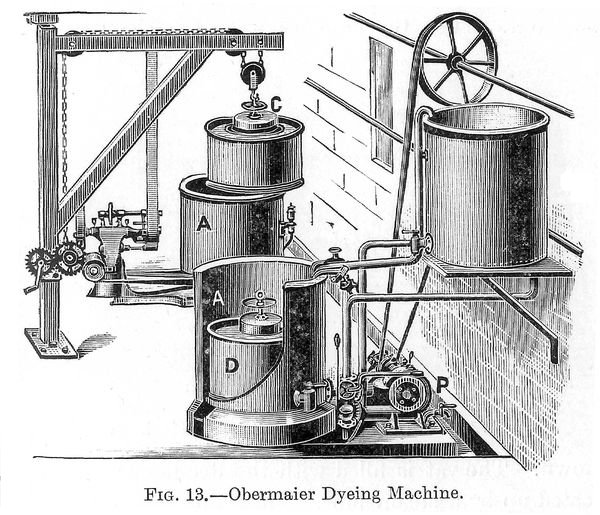
Obermaier (p. 046) Dyeing Machine, which is illustrated in figure 13.--In the Obermaier apparatus dye-vat, A, is placed a cage consisting of an inner perforated metal cylinder, C, and an outer perforated metal cylinder, D; between these two is placed the material to be dyed. C is in contact with the suction end of a centrifugal pump, P, the delivery end of which discharges into the dye-vat A. The working of the machine is as follows: the slubbing or sliver is placed in the space between C and D rather tightly, so that it will not move about. Then the inner cage is placed in the dye-vat as shown. The vat is filled with the dye-liquor, which can be heated up by a steam pipe. The pump is set in motion, the dye-liquor is drawn from A to C, and in so doing passes through the material packed in B and dyes it. The circulation of the liquor is carried on as long as experience shows to be necessary. The dye-liquor is run off, hot water is run in to wash the dyed material, and the pump is kept running for some time to ensure thorough rinsing, then the water is run off, and by keeping the pump running and air going through a certain amount of drying can be effected. This machine works very well, and with a little experience constant results can (p. 047) be obtained. The slubbing or sliver may be scoured, bleached, rinsed, dyed, washed, soaped, or otherwise treated without removing it from the machine, which is a most decided advantage.

Yarn Dyeing Machines.--In figure 14 is given an illustration of a machine for dyeing yarn in the hank form, made by Messrs. Read Holliday & Sons, of Huddersfield. The illustration gives a very good idea of the machine. It consists of a wooden dye-vat, which can be heated by steam pipes in the usual way. Extending over the vat are a number of reels or bobbins, these are best made of wood or enamelled iron. These reels are in connection with suitable gearing, so that they can be revolved. There is also an arrangement by means of which the reels can be lifted bodily in and out of the dye-vat for the purpose of taking on and off the hanks of yarn. A reel will hold about 2 lb. of yarn. The working of the machine is simple. The vat is filled with the requisite dye-liquor. The reels which are lifted out of the vat are then charged with the yarn, which has been previously wetted out. They are then set in revolution and dropped into the dye-vat, and kept there until it is seen that the yarn has acquired the desired shade. The reels are lifted out and the hanks removed when the machine is ready for another lot of yarn.
There are several makers of hank-dyeing machines of this type, and as a rule they work very well. The only source of trouble is a slight tendency for the yarn on one reel if hung loosely of becoming entangled with the yarn on other reels. This is to some extent obviated by hanging in the bottom of the hank a roller, which acts as a weight and keeps the yarn stretched and so prevents it flying about.
To some makes of these machines a hank wringer is attached.

Klauder-Weldon Hank-dyeing Machine.--This is illustrated in (p. 048) figure 15, which shows the latest form. It consists of a half-cylindrical dye-vat built of wood. On a central axis is built two discs or rod carriers, which can revolve in the dye-vat, the revolution being given by suitable gearing which is shown at the side of the machine. On the outer edge of the discs are clips for carrying rods on which one end of the hanks of yarn is hung, while the other end is placed on a similar rod carrier near the axle. The revolution of the discs carries the yarn through the dye-liquor contained in the lower semi-cylindrical part of the machine previously (p. 049) alluded to. At a certain point in every revolution of the discs the rods carrying the yarns are turned a little; this causes the yarn to move on the rods, and this motion helps to bring about greater evenness of dyeing. The most modern form of this machine is provided with an arrangement by means of which the whole batch of yarn can be lifted out of the dye-liquor. Arrangements are made by which from time to time fresh quantities of dyes can be added if required to bring up the dyed yarn to any desired shade. This machine works well and gives good results. Beyond the necessary labour in charging and discharging, and a little attention from time to time as the operation proceeds, to see if the dyeing is coming up to shade, the machine requires little attention.
Many other forms of hank-dyeing machine have been devised. There is Corron's, in which an ordinary rectangular dye-vat is used. Round this is a framework which carries a lifting and falling arrangement that travels to and fro along the vat. The hanks of yarn are hung on rods of a special construction designed to open them out in a manner as nearly approaching hand work as is possible. The machine works in this way. The lifting arrangement is at one end of the vat, the hanks are hung on the rods and placed in the vat. Then the lifter is set in motion and moves along the vat; as it does so it lifts up each rod full of yarn, turns it over, opening out the yarn in so doing, then it drops it again in the vat. When it has travelled to the end of the vat it returns, packing up the rods of yarn in so doing, and this motion is kept up until the dyeing is completed. This machine is very ingenious.
A type of machine which has been made by several makers consists of an ordinary rectangular dye-vat surrounded with a framework carrying a number of sets of endless chains, the links of which carry fingers. The hanks of yarn are hung (p. 050) on rods at one end of which is a tooth wheel that when in position fits into a rack on the side of the vat. The action of the machine is this, the hanks are hung on the rods and placed at the entrance end of the vat, by the moving of the chains it is carried along the vat and at the same time revolves, thus turning over the yarn, which hangs in the dye-liquor; when it reaches the opposite end of the vat, the rod full of yarn is lifted out, carried upwards and then towards the other end of the vat when it is again dropped into the dye-vat to go through the same cycle of movements which is continued until the yarn is properly dyed.
Piece Dyeing Machines.--Wherever it is possible it is far more preferable to dye textile fabrics in the form of woven pieces rather than in the yarn from which they are woven. During the process of weaving it is quite impossible to avoid the material getting dirty and somewhat greasy, and the operations of scouring necessary to remove this dirt and grease has an impairing action on the colour if dyed yarns have been used in weaving it. This is avoided when the pieces are woven first and dyed afterwards, and this can always be done when the cloths are dyed in one colour only. Of course when the goods are fancy goods containing several colours they have to be woven from dyed yarns.
The most common form of machine in which pieces are dyed is the jigger, commonly called the jig, this is shown in figure 16. It consists of a dye-vessel made long, sufficiently so to take the piece full width, wide at the top, narrow at the bottom. At the top on each side is placed a large winding roller on which the cloth is wound. At the bottom of the jig is placed a guide roller round which passes the cloth. In some makes of jigs there are two guide rollers at the bottom and one at the top as shown in the illustration, so that the cloth passes several times through the dye-liquor. In working the cloth is first wound on one of the rollers then (p. 051) threaded through the guide rollers and attached to the other winding roller. When this is done dye-liquor is run into the jig, and the gearing set in motion, and the cloth wound from the full on to the empty roller. With the object of keeping the piece tight a heavy press roller is arranged to bear on the cloth on the full roller. When all the cloth has passed from one roller to the other it is said to have been given "one end". The direction of motion is now changed and the cloth sent in the opposite direction through the jig and the piece has now received another "end". This alternation from one roller to the other is continued as long as is deemed necessary, much depending on the depth of colour which is being dyed, some pale shades may only take two or three ends, deeper shades may take more. When dyeing wool with acid colours which are all absorbed from the dye-liquor, or the bath is exhausted, it is a good plan to run the pieces several ends so as to ensure thorough fixation of the dye on the cloth.
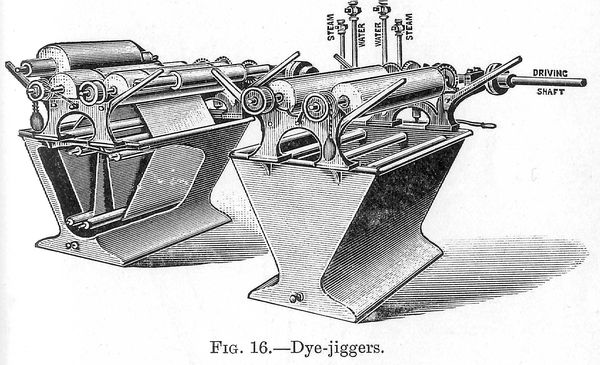
It is not advisable in working these jigs to add the whole of the dye to the liquor at the commencement, but only a part of it, then when one end is given another portion of the dye may (p. 052) be added, such portions being always in the form of solution. Adding dyes in powder form inevitably leads to the production of colour specks on the finished goods. The reason for thus adding the dye-stuff in portions is that with some dyes the affinity for the fibre is so great that if all were added at once it would be absorbed before the cloth had been given one end, and, further, the cloth would be very deep at the front end while it would shade off to no colour at the other end. By adding the dye in portions this difficulty is overcome and more level shades are obtained, but it is met with in all cases of jigger dyeing. It is most common in dyeing (p. 053) wool with basic dyes like Magenta, Auramine, Methyl Violet or Brilliant Green, and with acid dyes like Acid Green, Formyl Violets, Azo Scarlet or Acid Yellow.
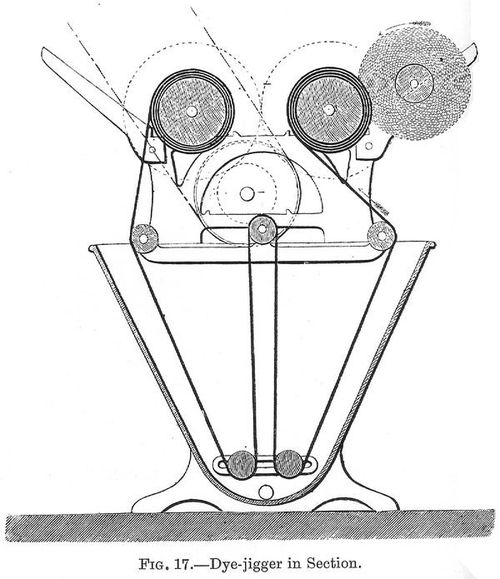
Some attempts have been made to make jiggers automatic in their reversing action, but they have not been successful owing to the greatly varying conditions of length of pieces, their thickness, etc., which have to be dyed, and it is next to impossible to make all allowances for such varying conditions.
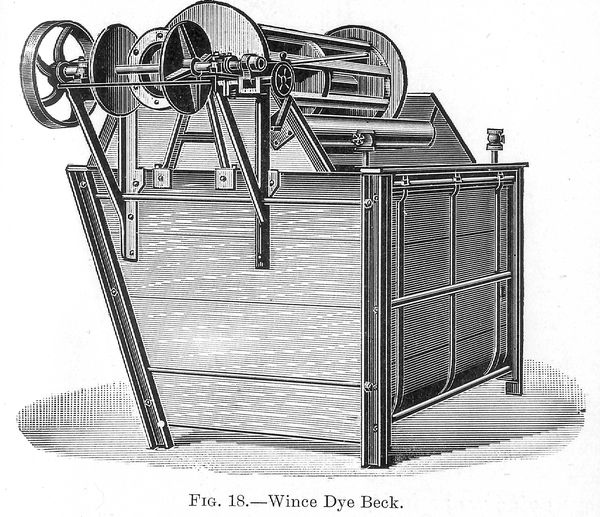
In figure 17 is shown the jig in section, when the working of the machine can be more easily traced.
The Jig Wince or Wince Dye Beck.--This dyeing machine is very largely used, particularly in the dyeing of woollen cloths. It is made by many makers, and varies somewhat in form accordingly. Figures 18 to 21 show three forms by different makers. In any make the jig wince or wince dye beck (p. 054) consists of a large rectangular, or in some cases semi-cylindrical, dye-vat. Probably the best shape would be to have a vat with one straight side at the front, and one curved side at the back.
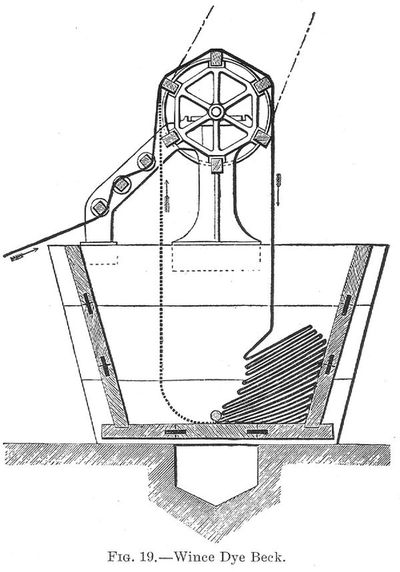
In some a small guide roller is fitted at the bottom, under which the pieces to be dyed pass. Steam pipes are provided for heating the dye-liquors. The beck should be fitted with a false bottom, made of wood, perforated with holes, or of wooden lattice work, and under which the steam pipes are placed. The object being to prevent the pieces from (p. 055) coming in contact with the steam pipes, and so preventing the production of stains. Above the dye-vat and towards the back is the wince, a revolving skeleton wheel, which draws the pieces out of the dye-vat at the front, and delivers them into it again at the back. The construction of this wince is well shown in the drawings. The wince will take the pieces full breadth, but often they are somewhat folded, and so several pieces, four, five or six, can be dealt with at one time. In this case a guide rail is provided in the front part of the machine. In this rail are pegs which serve to keep the pieces of cloth separate, and so prevent entanglements. The pieces are stitched end to end so as to form an endless band. When running through the vat they fall down in folds at the back part of the beck, and are drawn out from the bottom and up in the front. Each part thus remains for some time in the dye-liquor, during which it necessarily takes up the dye.
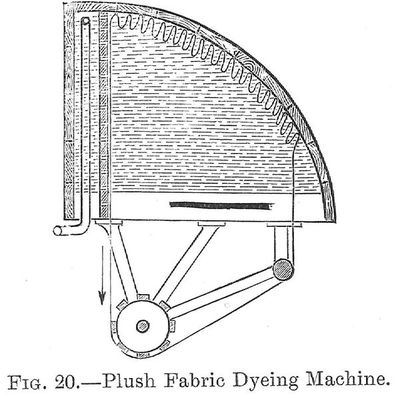
Figures 18 and 19 show forms of these wince dyeing machines, constructed of wood, and very largely used in the dyeing of woollen cloths. They are serviceable forms, and give very good results, being suitable for all dyes.
Figure 20 is a form of machine better adapted than the preceding (p. 056) for the dyeing of plush fabrics. In this kind of cloth it is important that the pile should not be interfered with in any way, and experience has shown that the winces of the form shown in figures 18 and 19 are rather apt to spoil the pile; further, of course, plush fabrics are dyed full breadth or open. In the wince now shown all troubles are (p. 057) avoided, and plush fabrics can be satisfactorily dyed in them.
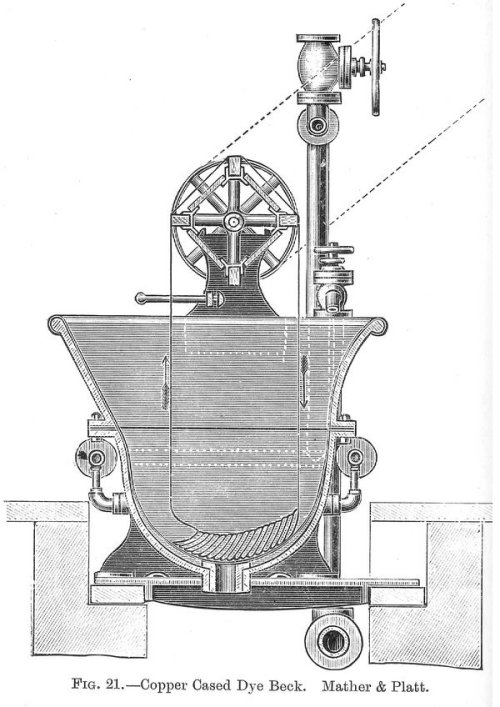
Figure 21 shows a dye-bath built of iron, cased with copper, suitable for dyeing most colours on woollen cloths.

In the jig and wince dyeing machines the pieces necessarily are for a part of the time, longer in the case of the jigger than in that of the wince, out of the dye-liquor and exposed to the air. In the case of some dyes, indigo especially, this is not desirable, and yet it is advisable to run the cloth open for some time in the liquor so as to get thoroughly impregnated with the dye-liquor.
The so-called hawking machine, figure 22, is an illustration of Read Holliday's hawking machine, made by Messrs. Read Holliday & Sons, of Huddersfield. There is the dye-vat as usual; in this is suspended the drawing mechanism, whose construction is well shown in the drawing. This is a pair of rollers driven by suitable gearing, between which the cloth passes, and by which it is drawn through the machine. A small roller ensures the cloth properly leaving the large rollers, (p. 058) then there is a lattice-work arrangement over the pieces are drawn. In actual work the whole of this arrangement is below the surface of the dye-liquor in the vat. The piece to be dyed is threaded through the machine the ends stitched together, then the arrangement is lowered into the dye-vat and set in motion, whereby the cloth is drawn continuously in the open form through the dye-liquor, this being done as long as experience shows to be necessary. This hawking machine will be found useful in dyeing indigo on wool, in mordanting and dyeing wool with the Alizarine series of dyes.
The various methods which are used in dyeing wool have, of course, underlying them certain principles on which they are based, and on the observance of which much of the success of the process depends. Sometimes these principles are overlooked by dyers, with the result that they do not get good results from their work. It must be obvious to any person with any technical knowledge that all processes of dyeing either wool or silk, or cotton or any other fibre, must take into consideration the properties of the fibre on the one hand, and that of the dye-stuff on the other. Wool must be treated differently from cotton, a process of dyeing which gives good results with the latter fibre would lead to nothing but disastrous effects with wool or silk; on the other hand, processes are used in the dyeing of wool which could not be possibly used for cotton on account of the very different properties of the fibre.
A few words as to the properties of wool as far as they relate to the methods of dyeing may be of use. Wool has the property of resisting the action of acids in a great degree, so that it may be treated with even strong acids with impunity. On the other hand, alkalies and alkaline solutions have strong action on it; the caustic alkalies rapidly dissolve wool, and their use must be avoided in all cases of dyeing this fibre. The carbonates of the alkalies have not so strong an action, and therefore may be used in moderation; nevertheless, too (p. 060) strong solutions of these should not be used. Soap has no disintegrating action on wool, and soap solutions may be used whenever necessary for cleansing or dyeing wool. Ammonia has no action on wool, and it may be used in place of soap if desired. There is one feature of wool that must be alluded to here, and that is its felting property. When wool is boiled with water and is handled a good deal, the fibres clot or felt together into a firm coherent mass. This should be avoided as much as possible, and when wool is cleansed and dyed in the loose condition it is absolutely necessary that every care be taken to avoid felting. This condition is much influenced by the temperature and the condition of the bath in which the wool is being treated, too high a temperature or too prolonged a treatment tends to increase the felting, therefore in dyeing wool prolonged treatment at the boil must be avoided.
Further, the condition of the bath has some influence on this point; it is found that an alkaline bath tends to considerably increase the felting properties of the wool, and on this account dyers invariably avoid the use of both the caustic and carbonated alkalies. Strong soap liquors have also some influence in the direction of increasing the felting, therefore soap should not be used if it can possibly be done without. Ammonia has not so strong a felting action as the other alkalies. Acids, on the other hand, exert a retarding action on the felting of the wool, and this is a matter of some interest and importance in the dyeing of wool, as an acid condition of the bath is necessary for dyeing by far the great majority of colouring matters on this fibre. Alkaline salts, such as Glauber's salt and common salt, exert little or no influence on this felting property, and can be added to dye-baths with impunity, and in many cases with good effect, so far as the quality of dyeing is concerned.
So far as the properties of the wool are concerned, it is seen (p. 061) that an acid condition of the dye-bath will work better than an alkaline condition, and wherever it is possible to use acids such should be added.
What has been said in regard to wool is equally true of all fibres derived from animals in the same way as wool is, such as horse-hair, fur of rabbits, hares and other animals, although, of course, there are some minor differences between different furs in their resistance to the action of acids and alkalies.
The next feature that influences the methods of dyeing wool is the varying properties of the dye-stuffs, or colouring matters. It is obvious that those which, like Magenta or Saffranine, have a strong affinity for the wool fibre must be dyed differently from those which, like Alizarine and Gambine, have no direct affinity for the wool fibre, and, further, which require the aid of mordants before they can be dyed, and on the character of which mordants the colour that is fixed on the fibre depends.
The dye-stuffs, independently of the question whether they be derived from natural sources or be of artificial origin, may be roughly divided into five groups, some of which may also be subdivided again as will be shown later on. These groups may be named the (1) Neutral, (2) Basic, (3) Acid, (4) Mordant, and (5) Indigo dye-stuffs. The first two classes are practically dyed in the same way; but as there is a great difference in the chemical composition of the colouring matters comprised in them, it will be best to consider them separately.
First Method.--This method is used in applying the now large and increasing group of azo dye-stuffs, which are characterised by being able to dye unmordanted cotton from a simple boiling bath. The dye-stuffs that are applied by the method now to be described include such as Benzopurpurine, Chrysamine, Chrysophenine, Titan red, Titan yellow, Benzo brown, Diamine (p. 062) red, Diamine brown, Diamine blue, Congo blue, Congo red, etc. The dyeing is done in a bath at the boil. If the bath contained only the dye-stuffs there would be a liability for the dyeing to be uneven, to prevent which a saline compound, such as salt, is added. Taking it all round, salt is the best body to add as it suits all colours very well indeed. Then come Glauber's salts; borax and phosphate of soda can also be used, but, owing to their slight alkaline properties, they are not so good as the neutral salts, like the two first named. When these colouring matters are dyed on cotton some of them dye best in a bath containing potash or soda, but these bodies, for reasons previously pointed out, are not available in wool dyeing, and should never be used. Wool dyes best in a slightly acid bath, and this may be taken advantage of in dyeing the yellows and blues of this group by adding a small quantity of acetic acid. The reds, as a rule, are affected by acids, and, therefore, it is not possible to use an acid bath with Benzopurpurine, Congo red, with the possible exception of the Titan reds and scarlets, Diamine scarlet, Benzo fast scarlet, Purpuramine, which are faster to acetic acid than the other reds of this class of dye-stuffs.
Probably the best plan of dyeing these colours is to first heat the bath to about 160° F., then enter the goods, and turn over two or three times to ensure that they are thoroughly impregnated with dye-liquor. The bath is now raised to the boil, and, steam being turned off, the goods are handled without further steam until the desired shade is obtained. Another plan is to enter the goods when the bath is at about 150° F., and, after raising to the boil, to work for half to one hour at that heat; but the plan first described gives rather better results, and is far preferable. The dye-baths, as a rule, are not completely exhausted, except when very pale shades are being dyed; in no case is it necessary to throw the dye-bath away, but simply to add the required amount of dye-stuff for a (p. 063) new batch; with those colouring matters which are not entirely exhausted from the bath a smaller amount, generally about three-fourths only, is required to be added, with about one-third the quantity of salt which was added to the first bath. Of course it is not advisable to keep the same bath or liquor in work always, but after about twenty or thirty batches of goods are dyed to throw it away and start a fresh liquor.
As a rule it will be found that these dye-stuffs are more thoroughly taken up from the bath than is the case in dyeing cotton; thus often with the same amount of dye-stuff in proportion to the material used the wool will dye rather a deeper shade than will cotton. In some cases, especially with the blues and violets, the shade is greatly different on wool from what it is on cotton, being generally redder and much stronger. (See the chapter on Union Dyeing.) While the shades are somewhat faster to light on wool than they are on cotton, they are no faster to soaping and in some cases not so fast. What may be the function of the salt, or other such added substance, is not very clear, probably it plays the same part as to similar bodies in dyeing the basic dye-stuffs. The dye-stuffs which are referred to above are all derived from coal-tar, and in the recipes which follow many examples of their use will be found.
There are but few natural dye-stuffs that have any direct affinity for wool. Turmeric, saffron, anotta, are about the only representatives, and these are not of much importance in wool dyeing by themselves, although they are sometimes used in conjunction with other natural dye-stuffs, when they are applied by a process which is adapted more especially for the other dye-stuff which is used.
Second Method.--The method of wool dyeing now being dealt with does not differ essentially from that described above, but as it is applied to quite a different class of dye-stuffs it is thought better to consider it as a second method. The (p. 064) dye-stuffs made use of in this method are what are called the basic coal-tar colours, and it may be remarked in passing that there are no natural colouring matters having the same properties. These dye-stuffs are derived from a number of so-called colour bases, such as Rosaniline, Pararosaniline, Methylrosaniline, Phenyl-rosaniline, and Auramine base. Many of these are colourless bodies containing the Amidogen group NH2, which imparts to them basic properties enabling them to combine with solids to form salts, and these salts have a strong colouring power. They form the commercial dye-stuffs Magenta, Saffranine, Thioflavine T, Auramine, Benzoflavine, Brilliant green, Methyl violet, etc., and these are salts (usually the hydrochloride) of colour bases. All these basic dye-stuffs have strong affinity for the wool fibre, and will immediately combine with it, dyeing it in colours which resist washing, etc., to a considerable extent, although there are great differences between the various members of the group in this respect. It has been shown that what takes place in dying wool with these colouring matters is that the colour base combines with the fibre the acid of the dye-stuff remaining in the dye-liquor.
Although it is possible to dye wool with the basic dyes from a plain bath containing water only, yet the results are not satisfactory, especially when working on a large scale; and for dyeing pale shades especially, the affinity of the dye-stuff for the fibre is so great that the first portions of the goods which are entered into the dye-bath have a great tendency to absorb all the dye-stuff, or the larger proportion of it, so that uneven dyeing is the result, one end of the piece of cloth being darker than the other end. This defect is particularly accentuated when pale tints are being dyed, the colouring matter being completely absorbed before all the goods are entered into the bath, but it may be remedied by adding the dye-stuff to the bath in small quantities at intervals during the (p. 065) process of dyeing. The best and most satisfactory method, however, is to add to the bath 10 per cent. of the weight of the wool of Glauber's salt, or some other neutral alkaline salt, which addition almost entirely prevents any defect of uneven dyeing. How these assistant mordants act is somewhat uncertain, the explanation generally given is that they exert a slightly solvent action on the dye-stuff, and so prevent it from going upon the fibre too readily. This is scarcely an adequate explanation, but in want of a better it will have to stand.
The affinity of the basic dyes for wool increases with increase of temperature. This is a property that has an important bearing on the method of dyeing, and to any person who pays some attention to theory in its practical applications it indicates the most rational method of working, which is to enter the goods into the bath cold, or, at the most, at a hand heat, then, after working a short time to get the goods thoroughly impregnated with the dye-stuff, to gradually raise the temperature to the boil and work for from half an hour to an hour longer, even if before this time the dye-bath be exhausted. The reason for giving a fair length of time in the bath is to get the colour properly fixed on the fibre. The combination of the dye-stuff and the fibre is a chemical one, and, as stated above, the dye-stuff has to be decomposed so that the base may combine with the essential constituent of the wool fibre, while it is obvious that this decomposition and then the union of the colour base with the wool must take time, and as it is effected more easily and completely at the boiling point, it is advisable to work the goods in the bath so as to fully insure that they are given the necessary time for the chemical change to take place.
The dye-bath is generally completely exhausted of colour, but if fairly clean it need not be thrown away, but used for another batch of wool by simply adding more Glauber's salt and (p. 066) dye-stuff. After a time the bath gets too dirty to used, when it may be thrown away, and a new dye-liquor made up.
In dyeing for pale shades it is best to add the dye-stuff in small quantities at intervals during the process of dyeing, and to run the goods quickly through the bath, so as not to give the dye-stuff too much opportunity to become absorbed by a portion of the goods only.
Working according to the hints given above, the dyeing of wool with the basic coal-tar colours may be carried out in a very satisfactory manner.
Third Method.--This method consists in dyeing the wool in a bath containing the dye-stuff, a little acid (usually sulphuric) with the addition of Glauber's salt, or some other alkaline salt, the essential feature or principle being that the bath is an acid one. This method is applicable to the large group of azo dye-stuffs derived from coal tar, and also to the acid dyes prepared from the basic coal-tar colours by the process of sulphonation.
It is also used to apply indigo carmine to wool, probably the only good example of a natural dye-stuff applied by this process. Most of the natural colouring matters, such as logwood and fustic, belong to another group of dye-stuffs.
The simple azo dyes are combinations of two or more organic bases, united together by a peculiar and characteristic group of nitrogen atoms. Such azo colours are, however, insoluble in water, and therefore they cannot be used in dyeing and textile colouring, although the firm of Messrs. Read Holliday & Sons years ago patented a process whereby these insoluble azo colours could be developed on the cotton fibre direct, and thus fabrics made from that fibre could be dyed in fast colours. When these insoluble azo colours are treated with sulphuric acid they are converted into sulpho acids, undergoing what is called sulphonation, an operation of the greatest (p. 067) importance and value in the preparation of dye-stuffs. The preparation of indigo extract or indigo carmine from indigo is also a case of sulphonation. The sulpho-acids of the azo colours, of the basic dyes, and of indigo are usually insoluble in water, although there are great differences in their properties in this respect. They will combine with bases such as soda, calcium and potash to form salts which are soluble in water, and it is usually in the form of sodium salts that these azo and acid dye-stuffs are sold to the dyer and calico printer. It is this power of combination with bases that makes them of value in wool dyeing. As Knecht and other authorities have pointed out, the wool fibre contains a basic principle capable of combining with acid bodies, and in wool dyeing with the colouring matters under discussion, this combination occurs between the sulpho-acid of the dye-stuff and the basic principle of the wool fibre.
This points to the fact that the dye-stuffs of this class do not combine with the wool in the form in which they are supplied to the dyer as sodium salts, which is shown by a property that many if not all of them possess, of not dyeing the wool fibre in a neutral bath. If a piece of wool be immersed in a solution of, say, a scarlet or indigo extract, which is neutral it is not dyed. The dye-liquor may penetrate thoroughly throughout the fabric, but if the piece of wool be lifted out, and allowed to drain, nearly all the liquor will drain away, and leave the wool nearly if not quite white, showing that the dye-stuff in the form in which it is sold has no affinity for the wool fibre. If now a few drops of sulphuric acid be added to the dye-liquor the wool will become dyed. The sulphuric acid liberates the free sulpho-acid of the dye-stuff, and this is now in a form to combine with the wool fibre, which it does. This is the fundamental principle underlying the acid method for dyeing wool with the acid group of colouring matters.
The (p. 068) practical application of the principle laid down above is a matter of simplicity compared with the other methods of dyeing. The composition of the bath is given above. It is best to enter the wool at from 150° to 160° F. and then to raise the temperature slowly to the boil. This method of proceeding gives time for the free colour acid of the dye to be liberated from the dye-stuff on the one hand, and for its combination with the wool fibre on the other. In dyeing pale tints with acid dye-stuffs it is a good plan not to add the acid until after the goods have been entered into the bath and worked for a short time to enable them to become impregnated with the dye-liquor; the acid may be then added, and the dyeing may be finished as usual.
By this plan of working more even dyeings can be obtained than by simply entering the goods direct into an acidified dye-liquor.
Any kind of acid may be employed, but generally sulphuric acid is used, partly because it is cheap, and partly because it is the commonest acid known. Acetic acid is also used in many cases.
Fourth Method.--We now come to the fourth method of dyeing wool. Strictly, perhaps, it is not a single method, but a group of methods, which are used to supply a certain class of dye-stuffs to the wool fibre; but as the governing principle depends upon the peculiar property of the dye-stuffs now to be noticed, which underlies all the variations of the process of dyeing, it has been thought better to speak of the fourth method rather than to subdivide further, in which case the fundamental principle might be lost sight of.
The class of dye-stuffs included in the fourth group was named by Bancroft the "adjective" group, because they require the aid of a second body, named the mordant, to properly develop and to fix the colour of the dye-stuff on the wool. It is sometimes known as the "mordant dye-stuff" class, (p. 069) and this is perhaps its best name. This group of colouring matters comprises dye-stuffs of both natural and artificial origin, the latter of which are getting very numerous and valuable, and bid fair to displace the natural members of the group. With but few exceptions the adjective dye-stuffs are not colouring matters of themselves, i.e., they will not dye wool or other fibres by themselves. Some are coloured bodies, such as fustic, logwood, Persian berries, Anthracene yellow, etc., but many are not so, and some possess but little colour, which, moreover, gives no clue to the colours that can be developed therefrom.
All the colouring matters of this class possess either a distinctively acid character, or belong to the class of phenols, which, while not being true acids, still possess weak acid functions that enable them to combine with bases like acids. These bodies have the property of combining with bases and metallic oxides, such as soda, potash, iron, alumina, chrome, tin, nickel, cobalt, etc., forming a series of salts. Those of soda and potash are usually soluble in water, while those of the other metals are insoluble, and are usually of strong colour. It is on this property of forming these insoluble coloured bodies, colour lakes, as they are called, that the value of the adjective dye-stuffs in dyeing depends.
The group of adjective colouring matters may be subdivided into two divisions, not depending upon any differences in the mode of application, but upon certain differences in the results they give. Perhaps the best example of an adjective dye-stuff is Alizarine. This body has a faint red colour, but of itself possesses absolutely no colouring power. When, however, it is brought into combination with such metallic oxide as alumina, iron and chrome, then it forms coloured bodies, the colour of which varies with the metal with which it is in union, thus with alumina, it is a bright red; with iron, a dark violet, almost black; with chrome, a (p. 070) deep red; with tin, a scarlet; and so on. This is a representative of the true adjective dyes, which comprise most of the so-called Alizarine dye-stuffs, and logwood, fustic, and most of the natural dye-stuffs. Another division of the group includes a few colouring matters of recent introduction, like Azo green, Alizarine yellow, Galloflavine, Anthracene yellow, Flavazol, etc., which, while forming insoluble colour lakes with metallic oxides, do not give different colours with different metals.
This class of dye-stuffs, owing to their forming these insoluble colours, gives really fast colours, capable of resisting lengthened exposure to light and air, and resisting washing, acids and alkalies. Of course there are differences between the various members of the group in this respect, and even the resisting power of an individual member depends a good deal on the metal with which it is combined, and the care with which the process of dyeing has been carried out.
In the dyeing of these adjective dye-stuffs, upon the various fibres, and on wool in any particular, the object is to bring about in any convenient way the formation on the fibre of the metallic combination of the colouring principle and the mordant, and it is obvious that if a satisfactory result is to be obtained, then this must be done in a very thorough manner. There are three ways in which this combination of colouring principle and mordant may be brought about in dyeing wool with these bodies, we may either mordant the wool first, and then apply the dye-stuff, or we may impregnate the wool with the dye-stuff first, and then fix or develop the colour afterwards, or, lastly, we may carry on both operations in one process. Each of these methods will now be discussed, and their relative advantages pointed out.
The mordanting method is one of the most generally useful. It consists in first causing a combination of the metal (p. 071) with the wool fibre. This is carried out by boiling the wool in a solution of the metal, such as bichromate of potash, chrome alum or chrome fluoride when chrome is to be used as a mordant, with alum or sulphate of alumina when alumina is required to be deposited on the fibre, and with copperas when iron is to be the mordant. It is best to add a little oxalic acid, cream of tartar, or tartaric acid to the mordanting bath, which addition helps in the decomposition of the metallic salt by the wool fibre, and the deposition of the metallic oxide on the wool. With bichromate of potash, sulphuric acid is often used, much depending upon the character of the mordant required. Some dye-stuffs, such as logwood for blacks, work best when the wool is mordanted with chromic acid, which is effected when sulphuric acid is the assistant mordant. Other dye-stuffs, such as fustic, Persian berries and Alizarine yellow, are best dyed on a basic chrome mordant, which is effected when tartar or oxalic acid is the assistant mordant used, or when some other form of chrome compound than bichrome is employed.
The actual mordanting is done by boiling the wool in a bath of the mordant, the quantity of which should be varied according to the particular mordant that is being employed and to the quantity of dye-stuffs which is to be used. It is obvious that for a fixing deep shade of, say, Alizarine on the wool, a larger quantity of mordant will be required than to fix a pale shade; sometimes this point is overlooked and the same amount of mordant employed for pale or deep shades. The best plan of carrying out the mordanting is to enter the wool in the cold bath or at a hand heat, and then raise to the boil and continue the boiling for one hour; of course the goods should be kept turned over during the process to facilitate the even mordanting of the wool. A great deal of the success of dyeing with the dye-stuffs now under consideration depends (p. 072) upon the efficiency with which the mordanting has been carried out. If this is at all unevenly done then no amount of care in the succeeding dyeing process will lead to the development of an even dyeing. After the mordanting is finished the goods should be rinsed with water, but it is not necessary to dry them.
The next stage in the process is the actual dyeing operations, which is done by immersing the mordanted wool in a bath of the dye-stuff or mixture of dye-stuffs.
The fundamental principle is to bring about the combination between the colouring principle of the dye-stuff and the metallic oxide which has been deposited on the wool in the previous mordanting process. As neither of these bodies, however, is very energetic it follows that the action must be a slow one, and, therefore, time is a highly important factor in the dyeing of wool by the mordanting process. The combination between the dye-stuff and the mordant is influenced also by temperature, and is most active at the boiling point of water. It is, therefore, needful to conduct this operation at that temperature, but it would be a wrong way to introduce the mordanted material into a boiling bath of the dye-stuff; nothing would conduce to uneven dyeing so much as that course. The best method of working, which, moreover, is most particularly applicable to the series of Alizarine dye-stuffs, is to enter the goods in a cold bath of the dye-stuff, and to work them for a short time to get them thoroughly impregnated, a condition which is essential if even dyeing is the goal aimed at, then to raise the temperature of the bath gradually to the boil, the goods being in the meantime well worked. The dyeing is continued for from one to one and a half hours at the boil.
It is important in dyeing by this process, especially when using Alizarine, to keep the temperature of the bath as uniform as possible, and the goods well worked. Alizarine, and some (p. 073) other members of this class, are rather sensitive to heat, and if a dye-vat be hot at the bottom and cold at the top uneven dyeing is sure to be the result; this is due to the greater affinity of the Alizarine for the mordant at the high than at the low temperature, and thus more is fixed on to the wool. The remedy for this is to so construct the heating arrangements of the vat that the temperature shall be as uniform as possible, while the goods should be kept continually turned over, and every portion of them brought into intimate contact with the dye-liquor. The continuance of the dyeing operations for one and a half to two hours after the vat has reached the boil is necessary to properly develop and fix the colour on the fibre; a short boil leaves the goods of a poor shade, without any solidity about it, and the colour is loose, while a longer boil brings up a solid shade and a fast colour.
Although it is not absolutely necessary to add any acid to the dye-bath during the dyeing operations, yet as the Alizarines and most of this class of dye-stuffs dye better in a slightly acid bath it is advisable to add a small quantity of acetic acid, say about one pint to every 100 lb. of goods; this serves to correct any alkalinity of the water, which may be due to its containing any lime. Dye-stuffs of the acid class, such as indigo extract, Cloth red, Acid magenta, etc., may be used along with the Alizarine dye-stuffs, in which case the addition of acid to the dye-bath becomes necessary, but too great an excess of acid should be avoided, as it interferes somewhat with the dyeing of the mordant dyes.
This is by far the best and most generally used method of applying these mordant dyes. It is not a costly process, being indeed economical, as it only requires just the right amounts of drugs and dye-stuffs, and there is the minimum loss of material in the mordanting and dye-baths. Shades can be brought up with the greatest ease, although it is well in (p. 074) the dyeing to add rather less dye-stuff than is actually required, and to add more when it is seen how the shade is coming up. The labour is the most important item in the mordanting and dyeing method.
The proportions of material used to the weight of the wool are: Of bichromate of potash, 3 per cent. for full shades, and 1 per cent. for pale shades; of fluoride of chrome, the same quantities; of acetate of chrome, according to the strength of the solution used; of alum, 10 to 20 per cent.; of sulphate of alumina, 5 to 10 per cent.; of copperas, 5 to 10 per cent.; of tartar, 1-1/2 to 2-1/2 per cent.; of oxalic acid, 1 to 1-1/2 per cent.; of sulphuric acid, 1 per cent.; of argol, 2-1/2 to 5 per cent.; of tartaric acid, 1 to 1-1/2 per cent.; but of course in an article like this it is impossible to give definite quantities.
Second Method. Stuffing and Saddening.--This method consists in first treating the wool with a solution of the dye-stuff, and then with a solution of the mordant required to develop and fix the colour. This method is more particularly applicable to such dye-stuffs as camwood, cutch, logwood, madder, fustic, etc., the colouring principles of which have some affinity for the wool fibre and will directly combine with it. It is not suitable for the application of the Alizarine colours. The saddening may be and is commonly done in the same bath, that is, after the wool has been stuffed it is lifted, the mordant--copperas, bluestone, bichrome, or alum--is added, and the wool is re-entered into the bath. This cannot be considered a good method of working; the shades obtained are full and deep and fairly fast, but there is usually a considerable loss of colouring matter, as the wool in no case abstracts the whole of the dye-stuff from the bath; what excess is left combines with the mordant when the latter is added, forming an insoluble colour lake, which falls down to the bottom of the dye-vat and is wasted, or it may go upon the (p. 075) wool in a loose, unfixed form, and cause it to rub badly and come off in milling. Then it is rather difficult to dye to shade, much of the result depending on conditions over which the dyer has little control. Working as he does with dye-stuffs of unknown colouring power, which may vary from time to time with every fresh batch of material, it is evident that, although the same quantities may be used at all times, at one time a deeper shade may be obtained than at another, and as it is impossible to see what is going to be the result, and if by mischance the shade does not come deep enough it cannot well be rectified by adding a quantity of dye-wood to the bath, because the mordant in the latter will prevent the colouring matter from being properly extracted, and only a part of that which is extracted is fixed on the wool, the rest being thrown away in the dye-bath, and partly on the particles of wood themselves, when logwood, camwood, etc., are used in the form of chips or powder. Dyers being well aware of this, are in the habit when mistakes occur of bringing up to shade with soluble dye-stuffs--archil, indigo extract, and such like.
This method, as stated above, is very wasteful, not only of dye-stuffs, but of mordants. In no case does the wool absorb the whole of the colouring matter from the bath, the unabsorbed portion goes down to the bottom of the bath when the mordant is added, so that when the dyeing is finished, the dye-bath is charged with a large quantity of colouring matter in an unusable form which has to be thrown away, thus at once adding to the pollution of the river into which it is run, and to the cost of the process of dyeing. As attention is being directed more and more to the question of the prevention of pollution of rivers, and as the waste liquors from dye-works add to the apparent pollution to a very considerable extent, dyers will have to develop other modes of dyeing than that of stuffing and saddening in one bath.
The (p. 076) principle of dyeing by stuffing and saddening may be carried out by the use of two separate baths; in fact, it is done in the case of dyeing a cutch brown from cutch and bichromate of potash. The goods are first treated in a bath of the dye-wood for a short time, then rinsed, and the colour is developed by padding into a saddening bath of the mordant. By this method the baths, which are never quite exhausted, can be retained for future use, only requiring about 1/2 to 3/4 of the original quantities to be added for each succeeding batch of the goods, in fact, in some cases, as in cutch, old baths work better than new ones.
The advantage attached to this method of working is that arising from economy of dye-stuff and mordant, and the reduction of the pollution of the stream on which the works are situated. The disadvantages are that the cost of labour is increased by there being two baths instead of one, and that the shades obtained are not always so full as with the one-bath method. This, of course, can be remedied by running the goods through the baths again, which, however, adds to the cost of the process, but there is this much to be said, the shade can be better brought up than by the one-bath process. In some cases the methods of mordanting, dyeing and saddening are combined together in the dyeing of wool, thus, for instance, a brown can be dyed by first mordanting with bichrome, then dyeing with camwood and saddening in the same bath with copperas. The shades obtained are fairly fast and will stand milling. The disadvantages of this process are the same as those attached to the dyeing and saddening in one bath.
Now we come to the last method of dyeing wool with mordant and colours, that in which the operation is carried out in one bath. This can only be done in those cases where the colour lake that is formed is somewhat soluble in dye-liquors, which usually have slightly acid properties; or (p. 077) where the affinity between the two bodies (colouring matter and mordant) is too great. This method can be carried out in, for instance, dyeing a cochineal scarlet with tin crystals, a yellow from fustic and alum, a black from logwood and copperas and bluestone, a red from madder and bichrome, and the dyeing of the Alizarine colours by the use of chrome fluoride, etc.
The shades obtained are usually not so deep as those got by the mordanting and dyeing process, but are frequently nearly so. In some cases, as in dyeing with fustic or logwood, it gives rather brighter colours, due to the fact that the tanning matters present in the dye-stuffs is not fixed on the wool, as is the case with the mordanting method, but is retained in the dye-bath. For dyeing with logwood and copperas or bluestone the process is not a good one, as it does not give as full shades as by the ordinary process. For dyeing with the Alizarine colours, using chrome fluoride as the mordant, it can be applied with fair success. There are advantages in the saving of time and labour and in the amount of steam required, all of which are important items in dyeing.
It is rather troublesome to match off by this process, but it can be done. For light shades the process will be found very useful, as these cost less than by any other process. The dye-baths may be retained for future use, although in process of time they become too dirty for use, when they must be thrown away.
Level Dyeing.--The first condition for successful dyeing is that the fibres to be treated are absolutely clean. A careful washing is not enough for this purpose. Cleanliness is undoubtedly the condition which the fibre must possess to enable the dye to hold on and not to come off the fibre, this latter causes a loss of dye-stuff, soils the whites, and gives rise to trouble between the dyer and finisher; it is also (p. 078) the condition for making the dye go on the wool evenly. The washing must be done at the boil, so that the fibre is well wetted out and all the air bubbles adhering to it are driven out. But this is not enough; it must be accompanied by a scouring operation, not only in the case of fibres of which the dyer does not know whether they have been scoured, but also when they have already been scoured and bleached. The kind of scouring that the fibres receive in this case need only be of a comparatively light character, but it must never be omitted, even for dark shades, as the traces of grease which the fibre contains are the causes of nearly irremediable stains in the dyeing operations. Even in dyeing black wool it is of the greatest importance to have the fibre suitably scoured.
The fatty matters which the fibre contains may belong to the components of the fibre itself and be natural matters, but in the case of wool yarns and cloths they are mostly dressing oils, from which the dyer cannot be too anxious to free the wool before dyeing. Some practical methods of preparatory treatment of the fibres before dyeing may therefore be described here with advantage.
Cotton is boiled off at actual boiling heat for two hours, with 8 per cent. of its weight of carbonate of soda and a little soft soap, which treatment is sufficient for dark colours.
For light colours it is necessary that the cotton be bleached. Wool is scoured with soda and soap in the proportion of 10 lb. soda and 2 lb. Marseilles soap for 100 lb. wool. Silk is scoured by boiling for one and a half hours in a boiling bath with 30 per cent. of its weight of soap. For light colours a second boiling should be given, with 15 per cent.
The careful cleaning of wool previous to dyeing is of exceptional importance. Raw wool is cleaned with carbonate of soda and ammonia. For 50 lb. wool to be cleaned 6 lb. carbonate (p. 079) of soda and 1-1/2 lb. ammonia are added to a bath of 150 gallons water. The wool is laid down in it for twenty minutes at 35° C., taken up, squeezed, treated for fifteen minutes in another bath, with 5 lb. carbonate of soda and then rinsed. The first bath must be renewed as often as possible, because it contains all the impurities. In the case of woollen yarn 30 lb. require two tubs of 40 gallons capacity. The first tub is to contain 35 gallons water and 2 lb. ammonia at 10° Be. After working the skeins for three minutes in it they are left to stand for fifteen minutes, then wrung out, and the operation is repeated in the second tub. Finally, the yarn is rinsed several times in soft water.
Woollen piece goods are treated in a large wooden tub at 40° C. with 4 lb. carbonate of soda and 2 lb. carbonate of ammonia for 80 lb. material. The pieces are moved about for twenty minutes, laid down in the bath overnight, again turned for ten minutes and hydro-extracted. They may also be handled for forty minutes in a bath of 2 oz. ammonia for 100 lb. wool at 60° C., and then for twenty minutes in clear water at 60° C.
After wetting or preparatory treatment, it will be best to proceed immediately to dyeing; if the fibres be left in a heap for too long a time, there is danger that they may become heated, or at least that the moisture may be irregularly distributed by the occurrence of partial drying, causing an uneven fixation of the colour in the first stages of dyeing. The first two conditions of successful dyeing are, therefore, a suitable wetting out and scouring. The dyer, however, must not be less careful to see that the dye-bath is what it ought to be.
Whenever possible the dye-stuff must be dissolved separately, or at least the bath not entered before the dye-stuff is well dissolved. Artificial dye-stuffs require particular attention to this point, because the presence of undissolved particles (p. 080) is the cause of irregularities, such as streaks, or, at least, specks. The solution is mostly made hot as follows: After pouring water at 180° F. upon the dye-stuff, stir gently, strain through flannel or through a very fine sieve, and pour more water upon the residue until nothing more is dissolved. As is well known, the artificial dye-stuffs often contain insoluble matter, resins, etc. It is therefore advisable to use only soft water for this operation.
The solutions of artificial dye-stuffs are ordinarily made at the rate of 1 to 5 lb. per 10 gallons of water, 2 lb. being the proportion mostly employed. This depends more or less on the solubility of the dye-stuff. Old solutions sometimes contain crystals of the dye-stuff which have separated out. These should be redissolved by heating before the solution is used. But it is best to make only such a quantity of solution as will suffice for immediate requirements.
With paste colours care should be taken to keep them in closed vessels in such a manner that they will not become hard by evaporation, and they should not be kept in any place where they are likely to freeze in winter time. In such an event it is not an uncommon circumstance for the casks or other vessels containing them to burst, with a consequent loss of dye-stuff. Before any of the paste is withdrawn from the cask, it is advisable to stir well up with a wooden stirrer.
In adding dye-stuff during the actual dyeing operation, it is advisable to add the dye-stuff to the bath in two or three portions, always taking out the goods before adding each lot of dye-stuff, and stirring up the contents of the bath before re-entering the goods. Another important condition of obtaining a level dyeing is to proceed slowly, beginning with a weak bath at a moderate temperature, and rising gradually to a boil. If necessary to retard the dyeing from the commencement, then an assistant mordant is added to the dye-bath, in the shape of soda crystals or phosphate of soda for the (p. 081) benzidine colours on cotton; bisulphate of soda or Glauber's salt in dyeing with azo colours or acid colours on wool; or tartar may be used in most cases with good effect, causing the wool to have a softer feel. Finally, the evenness of the dyeing is much increased by the frequent turning over of the material in the dye-bath, so managing this in the case of wool as to avoid felting.
When dyeing with a mordant, the dyer should see that the mordanting operation is thoroughly well done, for as much care is required for the mordanting as for the actual dyeing; in fact, if anything, the mordanting should be done with rather more care, as if it be at all defective no amount of care in the following dyeing operations will ensure a level dyeing. Chrome mordanted wool should be dyed without delay, as it is rather sensitive to light, especially the yellow sort, which gradually changes into the green sort of chromed wool.
One peculiarity of dyed wool is that it will continue to take up colour after it is removed from the dye-bath, especially if it contains any of the hot dye-liquor, therefore it is very desirable to wash the wool as soon as possible after its removal from the dye-bath. It is best, however, not to take the wool out of the hot bath, but to leave it in until the bath becomes cool, and then to take it out, by this means the colour becomes deeper and more solid looking, and is faster on the wool.
One cause of irregular dyeing may be mentioned, as it is occasionally met with, namely, the presence of foreign fibres in the goods, cotton in wool fabrics, and even of different varieties of the same fibre. All dyers know that dead or immature cotton will not dye up properly, a fact or defect more especially met with in indigo dyeing than probably in any other colour. Then wools from different breeds of sheep vary considerably in their dyeing power. Fine wools take up (p. 082) more colour than coarse, and, consequently, even from the same bath, will come out a deeper shade; if a fabric, therefore, contains the two kinds of cotton, or the two kinds of wool, they will not dye up evenly.
In the preceding sections brief notes have been given about the principal methods of dyeing wool, with some indications of the dyes which can be used under each method. In the succeeding sections will be given a number of recipes showing how, and with what dye-stuffs, various colours, shades and tints can be dyed upon wool. It will be understood that these recipes are applicable to all kinds of woollen fabrics, loose wool, slubbing, yarns in any form, woven worsted or woollen cloths, felts of any kind, etc., all these different forms require handling in a different way; it would not do, for instance, to treat a quantity of slubbing in the same way as a piece of worsted cloth, while hanks of yarn require a different mode of handling to a quantity of hat bodies. The different kinds of woollen fabrics require to be dealt with in different kinds of machines, and this has already been dealt with in the chapter on Dyeing Machinery and Dyeing Manipulations.
To describe and illustrate the application of all the various woollen dye-stuffs, whether of natural or artificial origin, and to show the great variety of shades, etc., which can be obtained with them, either all one or in combination, would require not one, but many volumes of the size that this present work is intended to be. Therefore, it becomes necessary to make a selection from the best-known and most used of the various dyes, and illustrate their application by a number of recipes, all of which, unless otherwise stated, are intended to be for 100 lb. weight of woollen material of any kind. It may also be pointed out that, as a rule, the recipes may be applied to the dyeing of fabrics made with other animal fibres than the wool of the sheep, as, for (p. 083) alpaca, cashmere, camel-hair, hare or rabbit fur, etc., inasmuch, as, with the exception of silk, all animal fibres practically possess the same dyeing properties.
It will be convenient to point out here that a very large proportion of the shades dyed on wool and other fabrics are obtained, not by the use of a single dye-stuff, although this should always be done, whenever possible, but by the combination of two or more dye-stuffs together in various proportions. It is truly astonishing what a great range of shades can thus be dyed by using two or three dyes suitably mixed together, and one of the things which go to making a successful dyer and colourist is the grasping of this fact by careful observation, and working accordingly. Dyers will find much assistance in acquiring a knowledge of colour and colour mixing from the two little books on Colour, by Mr. George H. Hurst, and the Science of Colour Mixing, by Mr. David Paterson, both issued by Messrs. Scott, Greenwood & Co., the publishers of the present work.
Black on Wool.--Until within a comparatively recent time black was dyed on wool solely by the use of logwood, combined with a few other natural dye-stuffs, such as fustic, indigo, etc., but of late the researches of colour chemists have resulted in the production of a large number of black dyes obtained from various coal-tar products. These have come largely into use, but still, so far they have not been able to entirely displace logwood, chiefly on the score of greater cost, the use of the natural dye still remaining the cheapest way of producing a black on wool; although the blacks yielded by some of the coal-tar black dyes are superior to it in point of intensity of colour and fastness to scouring, acids and light, as well as being easier to dye.
Blacks may be obtained from logwood by several methods, either by previous mordanting of the wool or by the stuffing and saddening methods, or by the one-bath process. The following (p. 084) recipes will show how these various methods are carried out in practice:--
Chrome Logwood Black.--The wool is first mordanted by boiling for one and a half hours with 3 lb. bichromate of potash and 1 lb. of sulphuric acid, working well the whole of the time. It is not advisable to exceed the amounts of either the bichromate or the acid here given, these quantities will result in a full bloomy black being obtained, but any excess gives rise to greyish dull blacks, which are undesirable. After mordanting rinse well with water, when the goods will be quite ready for the dye-bath.
The dyeing is done in a bath made from a decoction of 40 lb. of good logwood. It is perhaps preferable to start cold or only lukewarm, raise to the boil and work for one hour, then lift, rinse well, and pass into a boiling bath made from 1 lb. of bichromate of potash and 1/4 lb. of sulphuric acid for half an hour. This extra chrome bath fixes any colouring matter which may have been absorbed by the wool but not properly fixed by the mordant already on, it leads to fuller shades which are faster to rubbing and milling.
The mordanting bath may be kept standing and used again for fresh lots of wool, in which case it is only necessary to add 2-1/2 lb. of bichromate of potash and 1 lb. sulphuric acid to the bath for each additional lot of wool that is being dealt with. Old mordant baths work rather better than new ones, but the use cannot be prolonged indefinitely, there comes a time when the bath gets too dirty to use and then it must be thrown away.
During the operation the bichromate of potash becomes more or less decomposed and there is formed on the wool fibre a deposit of chromic acid and chromic oxide, this deposit forms the mordant that in the subsequent dye-bath combines with and fixes the colouring matter, the hæmatoxylin of the logwood, and develops the black on the wool.
In (p. 085) place of sulphuric acid, hydrochloric acid can be used with some advantage as regards the proportion of bichromate decomposed, and therefore an increase in the amount of chromium oxide deposited on the wool.
This gives a deep blue black, somewhat wanting in bloom. The following recipe gives a much bloomier black, but is rather more expensive to dye.
Chrome Logwood Black.--Mordant by boiling in a bath containing 3 lb. bichromate of potash and 7 lb. tartar. Dye and otherwise treat as in the last recipe; 4 lb. of tartaric acid used in place of the tartar, gives rather brighter and bloomier shades. The use of so-called tartar substitutes is not to be recommended, they give no better results than does sulphuric acid and are much dearer to use.
A somewhat greener shade of black than is yielded by either of the above two recipes is the following:--
Chrome Logwood Black.--Mordant the wool in a bath containing 4 lb. oxalic acid and 3 lb. bichromate of potash, afterwards dyeing as in the first recipe.
All the above recipes give blacks of a bluish tone, which on the whole have a good bloomy and solid appearance. Often what is called a jet black is wanted, this can be obtained by following the recipe given below.
Chrome Logwood Jet Black.--Mordant the wool by any of the methods given above. The dyeing is done in a bath made from 40 lb. logwood and 5 lb. fustic, working as described in the first recipe. Using these properties a good jet black is obtained, which is quite satisfactory on the score of solidity and fastness. It is not advisable to exceed the quantity of fustic here given, or otherwise the black will have a tendency to assume a greenish tone that is not at all desirable. This greening becomes more marked when from 7-1/2 to 10 lb. of fustic is used, or if alum be added to the mordant along with the bichromate of potash.
Chrome (p. 086) blacks are the best blacks which can be obtained from logwood. They have, however, a tendency to turn green on exposure to the weather, which tendency seems to be most prevalent in those blacks in which sulphuric acid has been used as the acid constituent of the mordanting bath. The greening may be reduced to a minimum by adding to the dye-bath about 1 to 2 lb. of Alizarine. Another plan which has been followed is to give the wool a bottom with 5 to 6 lb. of camwood or peachwood, then mordanting and dyeing us usual.
Logwood Black on Wool.--Boil first for one hour with a decoction of 8 lb. camwood, then lay down for fifty minutes in a boiling bath of 3 lb. bichromate of potash, 1 lb. alum, 1 lb. tartar. It is a good plan to allow the goods to hang overnight.
The dye-bath is prepared with 45 lb. logwood, 8 lb. fustic, 4 lb. sumac. Dye one hour at the boil, wash and dry.
Indigo Black.--This is sometimes called woaded black, and has an excellent reputation as a fast black. It is dyed by first giving the wool a medium blue bottom in the indigo vat by the method of vat dyeing, which will be described later on, and then dyeing by either the second or third recipe given above. The use of sulphuric acid is rather to be avoided in dyeing an indigo vat with chrome and logwood, as the chromic acid set free during the process is likely to attack and by destroying the indigo to materially reduce the intensity of the blue bottom. Or, after blueing in the vat, the black may be dyed or topped on by the process with copperas, which will be described below.
Iron Logwood Black.--Mordant the wool by boiling one and a half to two hours in a bath made with 5 lb. copperas, 2 lb. bluestone, 2 lb. alum, and 10 lb. argol. The dyeing is done in a bath of 50 lb. logwood.
It is not advisable to use more argol than is here given, for (p. 087) although a little excess will not materially affect the beauty or brilliancy of the resulting shade, yet such excess is wasteful, and makes the dyeing cost more than it otherwise would. On the other hand, too little will cause the shade to become greyish in tone and wanting in solidity. The copper sulphate (bluestone) added increases the fastness of the finished black to light, the best proportions to add are from 2 lb. to 4 lb. for 100 lb. of wool. The shade obtained in the above recipe is of a bluish-violet hue, if a jet black be wanted, add 5 lb. of fustic to the dye-bath. Another and very common method of working is the "stuffing and saddening" process, given in the next recipe.
Iron Logwood Black.--Make a bath of 50 lb. logwood, 6 lb. fustic, and 1 lb. sumac. Work the wool in this for one hour at the boil, lift, allow the bath to become cool, then add 6 lb. of copperas (ferrous sulphate) and 2 lb. bluestone; re-enter the wool, raise the temperature to the boil, and work half an hour, then lift, wash and dry. On the whole the first method is the most economical and yields the best blacks, fastest to rubbing.
The iron-copper-logwood blacks are not so fast to acids as the chrome-logwood blacks, but they are rather faster to light and air, and equally so to scouring and milling.
One-bath methods of dyeing blacks are sometimes preferred by wool dyers. Of these the following is an example.
Logwood Black.--Make a dye-bath with 50 lb. logwood, 5 lb. fustic, 6 lb. copperas, 2 lb. copper sulphate, and 4 lb. oxalic acid. Enter the goods and work at the boil to shade. The oxalic acid is added for the purpose of retaining the logwood-iron-copper black lake, which is formed on mixing the various ingredients together in solution. On boiling the wool in the liquor the fibre gradually extracts out the dye matter and becomes dyed. The use of some of the so-called "direct (p. 088) blacks" (noir reduit, Bonsor's black) is based on the same principle.
These dyes are mixtures of logwood, fustic or other dye-stuff with copperas, bluestone and oxalic acid, and only require adding to water to make the dye-bath. This method of working enables logwood to be used in conjunction with dihydroxynaphthalene and some other coal-tar derivatives to obtain blacks of good solidity and much faster to light, air, acids and scouring than the ordinary logwood blacks.
Another recipe for a one-bath logwood black, using the extracts in place of the dye-wood itself, is the following:--
Logwood Black.--Prepare a dye-bath with 12 lb. logwood extract, 2 lb. fustic extract, 6 lb. copperas, 4 lb. bluestone, 3 lb. oxalic acid, 2 lb. tartar. Boil the goods in this for one hour.
Some dyers use the dye-woods and prepare from them a decoction by boiling in water; in some respects this is the most economical plan, only the dyer has to get rid of the spent dye-wood from which the colouring matter has been extracted, and this is not always an easy matter. Some dyeing machines (Smithson's) have been devised which contain as one of their features a dye-wood extractor, in which the extraction of the colouring matter of the wood proceeds at the same time as the dyeing. Good results are got with such machines, although they leave something to be desired.
Many dyers use the dye-wood extracts which are now made on a large scale. These are for the dyer much more convenient to use, although naturally rather more costly. They are approximately five times the strength of the dye-wood, but they vary very greatly in this respect.
Logwood blacks can be readily distinguished from nearly all other blacks, in that by treatment with moderately strong hydrochloric acid they turn a bright red.
No other natural dye-stuff is used in the dyeing of black than these here given.
Of (p. 089) late years many black dyes derived from coal tar have been placed on the market. Among these may be enumerated the Acid Blacks of Messrs. Bead Holliday & Sons; the Naphthol and Naphthylamine Blacks of Leopold Cassella & Co.; the Victoria Blacks of the Farbenfabriken vorm, Fr. Bayer & Co.; the Wool Blacks of the Actiengesellschaft für Anilin Fabrikation; the Azo Blacks of the Farbwerke vorm, Meister, Lucius & Bruning; and one or two other blacks. These blacks are dyed very simply, as will be seen from the recipes given below, showing their application in the production of blacks of a great variety of tone. None of them dye a true jet black, but generally a bluish black or a violet black, but the tone may be readily changed to a jet or dead black by the addition of a little orange, yellow or green dye-stuff.
They give blacks of a very solid appearance and very bright in tone, and have the advantage over the logwood blacks of leaving the wool more supple and less liable to be felted. Moreover, as a rule they are faster to acids, alkalies and milling than are the logwood blacks, and as regards fastness to light they excel that dye-stuff. Unfortunately they are more costly to use, which tells against their entirely displacing logwood in dyeing blacks on wool.
Still, year by year their use is increasing, and as their price becomes less their employment will yet further extend. They may be combined with logwood, as they will dye with equal facility on mordanted and unmordanted wool.
Violet Black on Wool.--Make the dye-bath with 4 lb. Acid Black B, or Acid Black B B, 3 lb. sulphuric acid, and 10 lb. Glauber's salt. Work at the boil for one hour. The B brand of these blacks gives shades slightly redder in tone than the B B. The blacks are quite fast to light and acids, but not to soaping.
Blue Black on Wool.--Dye as in the last recipe, but use Acid (p. 090) Black S. This dye-stuff produces bluer shades of black than either B or B B, and they are faster to soaping.
Jet Black on Wool.--Make the dye-bath with 4-1/2 lb. Acid Black S, 1/2 lb. Fast Yellow F Y, 3 lb. sulphuric acid, and 10 lb. Glauber's salt. This shows how, by the addition of a little yellow dye-stuff, the blue shade may be changed to a full jet black.
Blue Black on Wool.--The dye-bath is made with 4-1/2 lb. Naphthol Black B (or 6 lb. Naphthol Black 3 B), 4 lb. sulphuric acid, and 10 lb. Glauber's salt. Work at the boil for one hour, then lift, wash and dry. The Naphthol Blacks have long been used in wool dyeing, and give excellent results, the 3 B brand dyeing much bluer shades than the B brand. There is also a 4 R brand giving violet blacks. These blacks are quite fast to acids and alkalies, are fast to light, and resist washing very well, the B brand being the fastest. The following recipe shows how a full jet shade can be obtained for these blacks:--
Jet Black on Wool.--Prepare the dye-bath with 4-1/2 lb, Naphthol Black B, 1 lb. Naphthol Green B, 1/4 lb. Indian Yellow, 4 lb. sulphuric acid, and 10 lb. Glauber's salt.
Blue Black on Wool.--Make the dye-bath with 5 lb. Anthracite Black B, 10 lb. Glauber's salt, and 5 lb. bisulphate of soda, working at the boil for one hour. Anthracite Black does not require a bath so acid as do some other coal-tar blacks. The shade obtained is a full blue black, which is fast to acids; alkalies turn it a little bluer, and soaping causes some loss of colour.
Violet Black on Wool.--Make the dye-bath with 5 lb. Anthracite Black R, and 10 lb. bisulphate of soda. The black thus obtained is a good one, fairly fast to acids, alkalies and soaping.
Dead Black on Wool.--Make the dye-bath with 6 lb. Anthracite Black R, 1 lb. Anthracene Yellow C, and 10 lb. bisulphate (p. 091) of soda. Work at the boil for one hour, then lift, add 3 lb. fluoride of chrome and work again at the boil for twenty minutes. This black is a very fine one, and is very fast.
Violet Black on Wool.--Make the dye-bath with 4 lb. Naphthylamine Black D, 10 lb. Glauber's salt, and 5 lb. acetic acid. This black is pretty fast to acids, alkalies and light, but is somewhat loose to soaping, and, therefore, cannot be used for black goods that have to be strongly milled. Naphthylamine Black 4 B dyes somewhat bluer shades than the B brand.
Blue Black on Wool.--Prepare the dye-bath with 6 lb. Victoria Blue Black, 20 lb. Glauber's salt, and 1-1/2 lb. acetic acid, working at the boil for one hour. A fine blue black, is obtained which is quite fast to acids, washing and light.
Greenish Black on Wool.--The dye-bath is made with 3 lb. Victoria Black Blue, 2 lb. Fast Yellow F Y, 20 lb. Glauber's, salt, and 1/1-2 lb. acetic acid. The dyeing is done at the boil and takes about an hour. This shade has a good full tone, and is fast.
Jet Black on Wool.--Make the dye-bath with 4 lb. Victoria Black B, 1/2 lb. Fast Yellow F Y, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil for one hour. A very fine shade is thus obtained, which is fast to acids, alkalies and soaping. By omitting the Fast Yellow a blue black is obtained, while by using Acid Green instead a greener tone is given to the black. In place of the Victoria Black B the two other brands, 5 G, and G, of these blacks may be used. These give equally fast blacks of a deeper and more jet black.
Black on Wool.--Prepare a bath with 5 lb. acetic acid, 9° Tw.; enter the wool for one hour, then lift and add 5lb. Naphthol Black 3 B, and 1/4 lb. Indian Yellow. Re-enter the goods and boil for one hour, wash and dry.
Many of the black dyes--Naphthol Black, Naphthylamine Blacks, (p. 092) Naphthyl Blue Black N, Acid Black B, etc.--are capable of slowly dyeing wool from neutral baths, that is, containing only Glauber's salt, or rather more quickly if a little acetic acid be present. Such dyes are very useful for dyeing heavily milled or felted fabrics, such as hat bodies for instance, as then the dye possesses greater penetrative properties and passes more into the substance of the fabric, which is, therefore, better dyed through. Also they are suitable for dyeing half-wool fabrics as will be seen on referring to the chapter dealing with the dyeing of union or cotton-wool fabrics.
It is quite possible to dye a black on wool by using a combination of acid and azo dye-stuffs, and below is given a recipe illustrating this method; it is one, however, rarely adopted.
Blue-Black on Wool.--Prepare the dye-bath with 10 lb. Glauber's salt, 2 lb. Patent Blue, 6 oz. Brilliant Orange, 4 oz. Amaranth, 4 oz. Acid Violet N, 4 lb. sulphuric acid. Enter the goods at about 150° F., raise to the boil and work to shade; lift, wash and dry. It may be of interest to note that by using a mixture of Azo Rubine and Acid green good blacks can be got.
There is a range of Acid and Azo dyes which are capable of dyeing from the usual acid baths on to wool, and yet can be developed and fixed on the fibre to good, full blacks. Types of such dyes are Anthracene Chrome Black F F, Diamond Black F, Chrome Patent Black D G and D G G, Fast Chrome Black, etc. Generally the blacks dyed on wool with these dyes are very fine, have a full, bloomy appearance, and are very fast. They are much used in dyeing hat bodies and fine cloths which have to be very fast to the weather.
The method of application will be gleaned from the recipes given below.
Black.--Prepare (p. 093) a dye-bath with 5 lb. Chromotrop S, 1/4 lb. Azo Yellow, 50 lb. Glauber's salt. Work for one and a half hours at the boil, then add 4 lb. sulphuric acid. Work at the boil for another half hour, then lift. Add to the same dye-bath 3 lb. bichromate of potash. Re-enter the goods and work at the boil for half an hour, then lift, rinse and dry.
Jet Black.--Mordant the wool by boiling for one hour in a bath made from 4 lb. bichromate of potash and 3 lb. of tartar. Then rinse, and dye in a bath containing 3-1/2 lb. Diamond Black, 1-1/4 lb. Alizarine Cyanine R R R double, and 1 lb. Gambine Yellow, working at the boil for from one to one and a half hours.
Diamond Black on Wool.--Mordant by boiling for one hour with 3 lb. bichromate of potash, 1 lb. oxalic acid. Wash and dry in a bath made with 2 lb. Diamond Black, 2 lb. acetic acid. Work at 120° F. for one hour, then heat to boil, and work until the dye is fully fixed. Lift, wash and dry.
A more common method of using the Diamond Black is given in the following recipe.
Diamond Black.--Prepare a dye-bath with 10 lb. Glauber's salt, 2-1/2 lb. Diamond Black, 1/2 lb. Diamond Green. Boil for an hour, then pass through a fresh bath of 2 lb. bichromate of potash for three-quarters of an hour at the boil; wash and dry.
This gives a fine jet shade of black, quite fast to a strong milling, and to light, alkalies and acids. Diamond Black by itself gives bluish shades. This dye is much used in the hat-dyeing trade.
Violet Black.--Mordant the wool by boiling for one and a half hours in a bath made with 3 lb. fluoride of chrome and 1 lb. oxalic acid, then rinse and dye in a bath containing 25 lb. Alizarine Cyanine Black G, 5 lb. acetate of ammonia, and 1 lb. acetic acid, working at the boil for one and a half hours. A (p. 094) fine full shade is obtained which is quite fast to acids, milling and light.
Brown Black.--Mordant the wool as in the last recipe, then dye in a new bath 25 lb. Alizarine Cyanine Black G, 3 lb. Anthracene Brown, 5 lb. acetate of ammonia, and 1 lb. acetic acid, working at the boil for one to one and a half hours.
Jet Black.--Mordant as in either of the above recipes, then dye in a bath containing 20 lb. Alizarine Black S W, and 2 lb. acetic acid. This black possesses a great degree of resistance to acid, alkali, milling and light, and is one of the best blacks at the disposal of the dyer.
Reddish Black on Wool.--Prepare the dye-bath containing 5 lb. Chromotrop 2 B, 10 lb. Glauber's salt, and 4 lb. sulphuric acid, work at the boil for one hour, then lift. Add to the same bath 3 lb. bichromate of potash and 1 lb. sulphuric acid, and work half an hour longer.
Blue Black.--Make the dye-bath with 6 lb. Chromotrop 10 B and 4 lb. sulphuric acid; dye, and develop the black by adding to the same bath 3 lb. bichromate of potash and 1 lb. sulphuric acid.
Jet Black.--Prepare the dye-bath with 5-1/2 lb. Chromotrop S, 1/4 lb. Alizarine Yellow G G W, 10 lb. Glauber's salt, and 4 lb. sulphuric acid. Slowly raise to the boil and work for one hour, then add to the same dye-bath 3 lb. bichromate of potash, and 1 lb. sulphuric acid, working at the boil for one hour.
These are but a few examples of how the Chromotrops (one of the most interesting series of dye-stuffs at the service of the dyer) may be used to dye blacks. They of themselves dye brilliant reds, from bright scarlet (2 R), crimson (6 B), and purple (8 B and 10 B), to maroon and clarets (S and S B). These being turned black on being chromed, give various shades--blue blacks, violet blacks, and jet blacks, which have the merit of being fast to acids, strong milling, and light in a great (p. 095) degree. The blue and violet blacks may be converted to jet shades by adding to the dye-bath some yellow dye-stuff, such as Azo Yellow, Alizarine Yellow, or Gambine Yellow, which will resist the action of the bichrome in the developing bath.
Chromotrop blacks while so very fast have the disadvantage of being expensive, but by combining them with logwood it is possible to obtain blacks that have a great degree of resistance to light, acids and milling. They are in this respect much superior to pure logwood blacks, while the cost is not prohibitive.
The following recipe will serve as an example of how these two dye-stuffs may be combined:--
Jet Black.--Make a bath with 2 lb. Chromotrop S, 15 lb. Glauber's salt, and 5 lb. hydrochloric acid. Work in this bath for one hour, then add 2-1/2 lb. bichromate of potash, and work again for half an hour, at the boil. Lift, rinse and dye in a new bath containing 25 lb. logwood, 1 lb. fustic extract and 1/4 lb. sulphuric acid, working at the boil for an hour.
Violet Black on Wool.--Dye the wool in the Chromotrop bath, and develop as in the last recipe. The final dye-bath is made with 6 lb. logwood, 8 oz. Patent Blue B, and 4 lb. sulphuric acid. By using logwood alone blue blacks can be dyed, by increasing the proportion of fustic a greener tone can be obtained, while by the use of a larger proportion of Chromotrop a redder tone of black is the result.
Jet Black.--Make the dye-bath with 20 lb. Glauber's salt, and 6 lb. Nyanza Black; when obtained is a good one and of solid appearance. Alkalies turn it red, but it is fast to dilute acid and soaping.
Black.--Prepare the dye-bath with 10 lb. Glauber's salt, 5 lb. oxalate of ammonia, 5 lb. acetic acid and 6 lb. Anthracene Chrome Black F. Work at the boil for three-quarters of (p. 096) an hour, or until the bath is exhausted of dye-stuff, then add 1-1/2 lb. bichromate of potash and 2 lb. hydrochloric acid to the same bath and work for half an hour longer.
The Anthracene Chrome Blacks, of which there are three brands, F, 5 B and F E, are excellent dyes, producing very fine blacks, and owing to the slowness of dyeing and great penetrative properties are very suitable for dyeing hat felts and other closely woven fabrics. The 5 B dyes more bluish shades than the F, while the F E brand gives full black. By combining these with Anthracene Yellow B N, Anthracene Acid Brown G, or other similar dyes, jet blacks can be got as per the following recipe:--
Jet Black.--Make the dye-bath with 6 lb. Anthracene Chrome Black F E, 5 oz. Anthracene Yellow B N, 10 lb. Glauber's salt, 2 lb. oxalate of ammonia and 5 lb. acetic acid, after dyeing, and the dye-bath, is exhausted of colour, add 1-1/2 lb. bichromate of potash and 3 lb. hydrochloric acid, and boil again for half an hour. Finish in the usual way.
One of the reasons for adding the oxalate of ammonia, is to precipitate out any lime which may be in the water in such a form that it will not react with the dye-stuff.
Fast Black.--Mordant the yarn with copperas (sulphate of iron). Dye in a bath with 5 lb. Gambine Y, 2 lb. Acid Mauve, 2 lb. bisulphate of soda. Proceed as described for full green.
Blue Black--3-1/2 lb. Naphthylamine Black S, 10 lb. Glauber's salt, and 5 lb. acetic acid; to fully exhaust the dye-bath add 8 lb. bisulphate of soda.
Jet Black.--5 lb. Naphthylamine Black S, 1/4 lb. Fast Acid Green B N, 10 lb. Glauber's salt, and 5 lb. acetic acid, adding 8 lb. bisulphate of soda to exhaust the bath.
Blue Black.--Give a deep blue bottom in the indigo vat and dye with 2 lb. Anthracite Black B, 10 lb. Glauber's salt and 2 lb. acetic acid.
Greys on Wool.--The dyeing of greys follows very naturally after (p. 097) the dyeing of blacks, for from a broad point of view greys are simply light blacks, and any dye-stuffs that will dye black will if used in smaller proportions give greys. There is a great variety of tone among greys: reddish greys, bluish greys, greenish greys, and so on. They may be dyed in a considerable variety of ways from a large number of dye-stuffs, both natural and artificial. Of these two classes the latter gives the best result as far as regards brightness of tone, and as regards other properties the greys obtained from the artificial coal-tar colours are fully equal to those from natural dyes.
A large number of recipes are in use by dyers for the production of greys, so many that it becomes almost an impossibility to do more than give a mere fraction of them here. However, a number of representative recipes will be given, covering all classes of dye-stuffs capable of being used for the purpose, and thus forming a guide to the methods of dyeing and the proportions of dye-stuffs to be used.
Light Grey.--Dye at the boil for three-quarters of an hour, in a bath containing 1 lb. perchloride of tin, 3 lb. alum, 3 oz. indigo extract, and 2 oz. cochineal.
Slate Grey.--Mordant by boiling with 4 lb. alum and 1 lb. argol, then dye with 6 lb. logwood, 6 oz. cudbear and 3 oz. indigo extract.
Slate Grey.--Another method is to boil the wool with 10 lb. logwood, 2 lb. Glauber's salt and 1 lb. sulphuric acid for three-quarters of an hour, then lift, add 1 lb. copperas, and re-enter the wool, working at the boil for three-quarters of an hour, then lift, wash and dry.
Reddish Grey.--Boil for an hour with 10 lb. fustic, 11 lb. cutch, 1/2 lb. bichromate of potash and 1-1/2 lb. copperas.
Pearl Grey.--Give a light blue ground in the indigo vat, then dye in a new bath with 2 lb. muriate of tin and 3/4 lb. cochineal, working at the boil to shade.
Silver (p. 098) Grey.--Prepare a bath with 3/4 lb. tannic acid; work for an hour in a warm bath, then sadden with 3 lb. nitrate of iron to shade, then lift, wash and dry.
Pearl Grey.--Prepare a bath with 3 lb. fluoride of chrome and 4 lb. Alizarine Bordeaux B. Enter into the bath when cold, then heat to the boil and work for one and a half hours, then lift, wash and dry.
Silver Grey.--The dye-bath is made with 3 lb. fluoride of chrome and 6-1/2 oz. Alizarine Cyanine G G, the dyeing being done as in the last recipe.
Greenish Grey.--A good shade is dyed with 3 lb. fluoride of chrome, 4 oz. Alizarine Bordeaux B, and 4 oz. Diamond Flavine G, working as given in the above recipe.
Grey.--Give a pale blue bottom with an indigo vat, then dye in a bath containing 1 lb. fluoride of chrome, 1/2 oz. Diamine Fast Red F, and 3/4 oz. Anthracene Yellow C; work at the boil for one hour, lift, wash, and dry.
Dark Grey.--A very fine dark grey, almost approaching a black is obtained by the following plan: bottom the wool with a medium blue by means of the indigo vat, dye in a bath containing 1 lb. fluoride of chrome, 3 oz. Diamine Fast Red F, and 3 oz. Anthracene Yellow C.
Slate Grey.--A good slate grey of a slightly greenish tone can be dyed in a bath of 5 lb. acetate of ammonia, 3/4 lb. Acid Blue 4 S, and 1/4 lb. Titan Brown R, working at the boil to shade.
Pale Slate Grey.--The dyeing is done in a bath made with 5 lb. acetate of ammonia, 5 oz. Acid Blue 4 S, and 1-1/2 oz. Titan Brown R, working at the boil for one hour.
Silver Grey.--A very nice shade is dyed with 3 oz. Acid Blue 4 S, 1/4 oz. Titan Red, and 5 oz. acetate of ammonia.
Silver Grey.--A shade similar to the last is dyed in a bath containing 10 lb. Glauber's salt, 5 lb. bisulphate of soda, and 3/4 oz. Anthracite Black R. By adding a little Thiocarmine R (p. 099) the shade can be turned bluer in tone, while the addition of a little Milling Yellow O, or Titan Yellow, turns it to the green side.
Pearl Grey.--Make the dye-bath with 10 lb. Glauber's salt, 5 lb. acetic acid, and 3/4 lb. Naphthylamine Black D. This gives fine shades of pearl grey.
Bluish Grey.--Mordant the wool by boiling in a bath made with 2 lb. bichromate of potash, 1 lb. tartar, and 1 lb. sulphuric acid. Dye in a bath containing 2 oz. Diamine Black (or 3/4 oz. Diamond Black and 1-1/2 oz. Alizarine Cyanine R), working at the boil for an hour and a half.
Grey.--This can be dyed with 3 oz. Nyanza Black B, and 10 lb. Glauber's salt, working at the boil.
Reddish Grey.--A good full shade is dyed with 1-1/2 oz. Cyanole extra, 1/4 oz. Orange extra, 3/4 oz. Archil Substitute N, 10 lb. Glauber's salt and 3 lb. sulphuric acid.
Slate Grey.--The dye-bath is made with 3 oz. Cyanole extra, 1/2 oz. Archil Substitute N, 3/4 oz. Orange extra, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Bright Pearl Grey.--Prepare a dye-bath with 3/4 oz. Patent Blue, 1/2 oz. Acid Violet N, 3/4 oz. Orange G, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Stone Grey on Wool.--The dye-bath is made with 1/2 oz. Chromotrop 2 R, 3/4 oz. Cyanole extra, 1-1/2 oz. Fast Acid Blue R, 3/4 oz. Acid Yellow, 20 lb. Glauber's salt, 3 lb. acetic acid. Enter at 80° F., then warm slowly and work to shade, lift, wash and dry.
These recipes will probably be sufficient to show the lines on which greys may be obtained in wool dyeing. It may be added that from the Acid Blacks B, B B, and S, good greys of a violet tone may be obtained, using from 1/2 to 3/4 oz. dye-stuff. The Naphthol Blacks will also be found useful in the same way, while the greys from Anthracene Chrome Blacks and the Alizarine blacks are very good and fast.
Red (p. 100) Shades on Wool.--The number of red shades that may be dyed on wool is infinite. They range over every variety of tint of red, from the palest blush-rose to the deepest crimson, and from the most brilliant pink to the dullest grenat shade.
It is quite impossible here to describe the dyeing of every imaginable shade of red, while the great variety of red dye-stuffs, both natural and artificial, adds to the difficulty of dealing in the space at command with all the various methods and dyes that may be used in the dyeing of reds on wool.
The methods that may be adopted for dyeing red shades on wool are many and various, depending not only on the particular dye-stuff used, but often on the particular shade that is being dyed. One method, which will yield a pale and useful tint with a particular dye-stuff, would fail if a full shade were necessary.
The greater number of red shades are now dyed by means of the artificial dye-stuffs, as these are much easier to dye than are the natural dyes, and they give, on the whole, more even and brilliant shades, while as regards fastness to milling, acids, and light they are fully equal, and in most cases superior, to the natural dyes.
The Direct Red Dyes.--Of this group of red dye-stuffs, Benzopurpurine, Titan Scarlet, Diamine Fast Red F, and Benzo Fast Red are types; many of them have been found to be very serviceable in wool dyeing. They may be dyed either from plain baths containing common salt or Glauber's salt, or from baths containing common salt or Glauber's salt and a little acetic acid.
Alkaline or soap baths do not work well as a rule, and must be avoided in wool dyeing. Generally the dye-bath is exhausted of colour, and full shades are easily obtained, while these reds are in general remarkable for the evenness and (p. 101) uniformity of tint which can be produced. The reds so dyed are, on the whole, fairly fast to soaping, and can be used for dyeing goods that have to be milled, while their resistance to light and air is fairly good. Benzopurpurine and Diamine Red are more or less affected by acids, but the Titan Red and some of the more modern reds, Diamine Brilliant Scarlet, Benzo Fast Scarlets, are all fast to acids. The fastness to washing and light of some of them, Benzo Fast Red, Diamine Fast Red F, Titan Red, is much increased by adding, after the wool has been dyed, 3 per cent. of fluoride of chromium to the dye-bath, and working a little longer.
The dyeing with these colours is done at the boil, and the goods may be entered direct into the boiling bath without fear of uneven shades being produced. This bath may be kept as a standing one, simply adding as each lot is dyed the necessary quantity of dye-stuff, a little fresh water to bring the bath up to its original volume, and a corresponding quantity of the salt originally added. The wool can then be entered and dyed.
In place of using salt or Glauber's salt, acetate of ammonia is an excellent assistant for this class of dyes.
The following are some recipes for dyeing various shades of red on wool with this class of dyes.
Scarlet.--The dye-bath is made with 3 lb. Titan Scarlet C B, and 10 lb. acetate of ammonia. This gives a good bright shade of scarlet, which is fast to acids and soaping, although not fast to light.
Scarlet.--Dye in a bath made with 3 lb. Diamine Scarlet B and 10 lb. Glauber's salt. This yields a light shade, not so fast to acids as the last, but equally fast to soaping and light.
Scarlet.--Make the dye-bath with 3 lb. Benzopurpurine 4 B, and 10 lb. Glauber's salt. This also gives a good shade of Scarlet fast to soaping. It is turned dark blue by acids, and is (p. 102) not fast to light. It is very largely used on underwear goods, but is not so satisfactory for this as the Titan Scarlet C B, or Benzo Fast Scarlet B S.
Scarlet.--The dye-bath may be made with 3 lb. Brilliant Congo G, 10 lb. Glauber's salt and 2 lb. acetate of ammonia. This gives a satisfactory shade of scarlet.
Bright Scarlet.--The dye-bath prepared with 2 lb. Geranine G, 5 lb. sulphate of soda, 5 lb. acetate of ammonia. Work at the boil for one hour, then wash and dry.
Dark Crimson.--Prepare a dye-bath with 1-1/2 lb. Chrysophenine, 1-1/2 lb. Hessian Violet, 25 lb. salt. Heat to 150° F., enter the goods, heat to boil and dye boiling for one hour, take out, rinse and wash.
Scarlet.--A brilliant shade of scarlet can be dyed in a bath of 3 lb. Benzo Fast Red, 1 lb. Chrysophenine, 10 lb. Glauber's salt and 2 lb. acetic acid.
Fast Red.--Dye the wool in a bath boiling, containing 1 lb. Diamine Fast Red F, 10 lb. Glauber's salt, and 2 lb. acetic acid, until the bath is exhausted, then add 3 lb. fluoride of Chrome and work half an hour longer at the boil.
Bordeaux.--Dye with 3 lb. Diamine Bordeaux, and 10 lb. Glauber's salt.
Pink.--Dye with 2 lb. Diamine Rose B D, 10 lb. Glauber's salt and 1 lb. acetic acid.
The basic red dyes are not very numerous, and comprise Magenta, Saffranine, Acridine Reds, Acridine Scarlets, Rhoduline Reds, Rhodamine and Neutral Beds. For successful dyeing they require a perfectly neutral bath. This bath should contain 10 per cent. of Glauber's salt, and is started cold and not too strong; when all the material has been entered the steam may be turned on and the temperature slowly raised, the material being turned over and over. The operation is continued only until the bath has been exhausted of colour, when it is stopped, and the wool taken out, and washed (p. 103) and dried. The liquor in the dye-baths may be allowed to cool down, and then it may be used for making the dye-bath for a second lot of goods, or it may be run away. It is best not to add the dye to the bath all at once, but in several portions as the work proceeds. The affinity of the wool for the basic dyes is usually so strong that if all were added to the dye-bath at the start, then the first portion of the goods entered might take up all, or nearly all, the colour, leaving but little for the last portion; the consequence being that the goods are dyed of an uneven colour, deeper in some parts than others. This defect is remedied by adding the dye in portions, entering the goods rather quickly, working cold, or by adding a little acetic acid and plenty of Glauber's salt. Notwithstanding all these precautions it is quite possible for the shades to come up somewhat uneven. These remarks are applicable not only to the basic reds but to the whole range of basic dyes, hence this class of dye-stuffs is but little used in the dyeing of wool.
Crimson.--Make the dye-bath with 2 lb. Magenta, and 15 lb. Glauber's salt, working as described above. This gives a fine crimson shade which, however, is not fast to soaping or to light. The quantity of dye-stuff given above should not be exceeded or the shades may come up bronzy, this may be avoided if a trace of acetic acid is added to the dye-bath.
Crimson.--Dye with 2-1/2 lb. of Saffranine and 15 lb. Glauber's salt. This dyes a fine Crimson shade.
Deep Red.--Use 3 lb. Rhoduline Red and 10 lb. Glauber's salt.
Scarlet.--The dye-bath is made with 1 lb. Saffranine Prima, 1 lb. Auramine, and 10 lb. Glauber's salt. The goods are entered into the dye-bath at about 120° F., and well worked about, then the temperature is raised slowly. When the dye-bath is exhausted the goods are lifted, washed and dried. There are no pure basic scarlets, and the above and similar combinations (p. 104) of a basic red and a basic yellow are the only ways in which a scarlet can be dyed on wool with basic coal-tar colours.
The basic colours are, in general, the hydrochlorides of some colour base, and in the process of dyeing the acid constituent of the wool fibre unites with the colour base, while the hydrochloric acid which is liberated passes into the dye-bath.
The acid reds are a very large group of red dyes, of somewhat varied chemical composition, which all have the property of dyeing from baths containing Glauber's salt and sulphuric acid or acetic acid, the usual proportions being 10 per cent. of the former, and 2 to 5 per cent. of the acid. Some are best dyed from a bath containing bisulphate of soda. The dyeing should be started cold, or at a lukewarm heat, then steam should be turned on and the temperature raised to the boil, at which it is maintained for an hour; this boiling serving to more intimately fix the dye-stuff on the woollen fibre.
The Eosine reds, of which Eosine in its various brands, Rose Bengale, Phloxine, Saffrosine and Erythrosine, are examples, are best dyed upon wool from a bath containing Glauber's salt and a little acetic acid. They do not require a very acid bath, hence the reason of using acetic acid. The method of dyeing is that given above as for basic reds, namely, enter into cold, or at most lukewarm bath, and raise the heat slowly, continuing the work until the shade required has been obtained. It is a good plan to start work in a neutral bath, and then when the material has become thoroughly impregnated with the dye-liquor to add the acetic acid. The shades obtained from these Eosine reds are remarkable for their brilliance, but unfortunately their fastness to light, washing, etc., is but slight, although it may be increased by treating the dyed wool in a bath of alum or acetate of lead.
Some (p. 105) of the acid reds, e.g., Acid Magenta, Acid Violet, belong to the group of sulphonated basic dyes. The vast majority belong to the group of azo dyes, which can be employed to dye from palest pinks to the deepest crimson reds. Some dye very brilliant shades, others only yield dull reds. Some dye shades remarkable for their fastness to all agencies, soap, acids, alkalies, light and air; others dye shades which may be fast to soap, but loose to acids and light. Generally even shades are readily obtained on any kind of woollen fabric. It is practically impossible to name all the acid reds that are known and that may be used, but a fairly representative series of recipes will be given.
Ponceau.--Wet out, then prepare a bath with 2 lb. Ponceau R, 10 lb. Glauber's salt, 2 lb. sulphuric acid. Enter the wool in the cold, bring to a boil and work to shade, wash and dry.
Crushed Strawberry.--Prepare a bath containing 10 lb. Glauber's salt, 4 oz. Scarlet R S, 9 oz. Indigo extract, 2 oz. Orange Y, 4 oz. sulphuric acid. Enter wool at 160° F., give four turns, raise temperature slowly to a boil, and turn to shade, lift and wash.
Scarlet.--Prepare a dye-bath with 2 lb. Azo cochineal, 10 lb. Glauber's salt, 4 lb. sulphuric acid. Work at the boil until the full shade is obtained, then lift, wash and dry.
Terra Cotta Red.--The dye-bath is made from 2-1/2 lb. Fast Acid Magenta B, 2-1/2 lb. Fast Yellow F Y, 10 lb. Glauber's salt, 2 lb. sulphuric acid. Work at the boil to shade.
Fast Scarlet.--Prepare a dye-bath with 3 lb. Glauber's salt, 1-1/4 lb. sulphuric acid, 2-1/2 lb. Brilliant Scarlet 4 R. Work at the boil for one and a half hours.
Scarlet.--Make the dye-bath with 2 lb. Scarlet 2 R J, 10 lb. Glauber's salt and 2 lb. sulphuric acid. The goods may be entered at about 150° F., and the temperature raised at the boil and maintained at that heat for one hour, then the goods are lifted, rinsed and dried.
The (p. 106) method given in the above recipes is that usually followed with the acid colours. When closely woven or thick goods are being dyed, where it is desired that the colour should penetrate well into the substance of the goods, the following modification of working may be adopted:--
The dye-bath is made up with the dye-stuff and Glauber's salt only, and the goods are worked in this at the boil until they are thoroughly impregnated with the dye-stuff liquor, then the acid is added in small quantities at a time, and the dyeing is continued for one hour to fix the colouring matter on the wool fibre. The goods may then be lifted out, washed and dried.
Scarlet.--Make the dye-bath with 2 lb. Scarlet F R, 10 lb. Glauber's salt and 2 lb. sulphuric acid. In place of scarlet F R, the F 2 R or F 3 R brands may be used, the latter giving the reddest shades.
Scarlet.--Make the dye-bath with 2 lb. Scarlet O O, 10 lb. Glauber's salt and 2 lb. sulphuric acid. Scarlet O dyes a yellower shade of scarlet, while scarlets O O and O O O dye slightly redder shades.
Scarlet.--The dye-bath is made with 3 lb. Brilliant Ponceau 2 R, 10 lb. Glauber's salt and 10 lb. bisulphate of soda. This gives a brilliant shade of scarlet. Brilliant Ponceau G, used in the same way, gives a much yellower tone of scarlet, the R gives a slightly yellower tone, while the 3 and 4 R brands dye redder shades.
Bluish Red.--The dye-bath is made with 2 lb. Brilliant Croceine B, 10 lb. Glauber's salt, and 10 lb. bisulphate of soda. Brilliant croceine B B and the brand M dye redder shades of scarlet.
Red.--Make the dye-bath with 3 lb. Milling Red R, 20 lb. Glauber's salt, and 5 lb. acetic acid. This is a good bright shade, and is quite fast to soaping and milling.
Deep Scarlet.--Dye with 3 lb. Chromotrop R, 10 lb. Glauber's (p. 107) salt, and 2 lb. sulphuric acid. This scarlet is very fast to milling, acid and light.
Red.--Make the dye-bath with 2 lb. Victoria Scarlet R, 1 lb. Victoria Rubine O, 10 lb. Glauber's salt, and 4 lb. sulphuric acid. A fine deep scarlet red is obtained.
Scarlet.--Dye with 2 lb. Brilliant Orseille C, 10 lb. Glauber's salt, and 3 lb. sulphuric acid. This gives a bright bluish shade of scarlet.
Red.--Dye with 1 lb. Emin Red and 5 lb. bisulphate of soda.
Scarlet.--Make the dye-bath with 3 lb. Croceine Scarlet 3 R, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Fawn Red.--Make the dye-bath with 1-1/2 oz. Cyanole, 1-1/2 oz. Orange extra, 2-1/2 oz. Archil Substitute N, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. This gives a nice light tint of fawn red, of a somewhat bluish tone.
Deep Fawn Red.--A very deep shade of fawn red is dyed with 4-1/2 oz. Cyanole, 2-1/4 lb. Orange extra, 1-1/4 lb. Archil Substitute N, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. The same dye-stuffs are used as in the last, but the result is a deeper shade, of a yellow tone.
Crushed Strawberry Red.--Use 4 oz. Chromotrop 2 R, 2 oz. Cyanine B, 1 oz. Azo yellow, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Pale Lilac Rose.--Dye with 1 oz. Chromotrop 2 R, 1/2 oz. Cyanine B, 1/2 oz. Azo yellow, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Deep Fawn.--Dye with 3-1/4 oz. Chromotrop 2 R, 1-1/2 oz. Orange G, 2 oz. Cyanine B, 4 oz. Fast Acid Blue R, 10 lb. acetic acid, and 20 lb. Glauber's salt.
Crimson.--Make the dye-bath with 3 lb. Titan Red 6 B, 20 lb. salt, with a little acetic acid, and work at the boil. This gives a fine shade of crimson, fast to acids and capable of standing milling very well.
Deep (p. 108) Crimson.--A bright and deep crimson is dyed with 4 lb. Fast Acid Magenta B, 10 lb. Glauber's salt, and 3 lb. sulphuric acid, working at the boil.
Pale Crimson.--Make the dye-bath with 2 lb. Fast Acid Magenta B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil. Level shades are readily obtained, and the dye is fast to washing.
Deep Crimson.--Make the dye-bath with 4 lb. Azo Fuchsine G, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. This gives a very deep shade of crimson, of a bluish tone.
Bluish Crimson.--Use in the dye-bath 2 lb. Azo Fuchsine G, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Pale Bluish Crimson.--Use in the dye-bath 1 lb. Azo Fuchsine G, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. This gives a very bright shade of pale crimson. The B brand of the Azo Fuchsines gives slightly bluer shades than the above.
Deep Crimson.--A very solid crimson is dyed in a bath containing 3 lb. Azo Red A, 2 oz. Orange extra, 2 oz. Cyanole extra, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. Work at the boil.
Bright Crimson.--A fine bluish crimson can be dyed on wool with 4 lb. Azo Red A, 10 lb. Glauber's salt, and 10 lb. bisulphate of soda. Work at the boil.
Deep Crimson.--A good shade can be dyed with 6 lb. Amaranth, 10 lb. Glauber's salt, and 10 lb. bisulphate of soda, working at the boil.
Brilliant Pale Bluish Crimson.--A really brilliant shade, bordering on a violet red, is dyed in a bath containing 1-1/2 lb. Fast Acid Violet R, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Bluish Crimson.--Make the dye-bath with 3 lb. Croceine Scarlet, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. This gives a good full shade of a bluish tone and very bright.
Bluish Crimson.--Dye with 3 lb. Chromotrop 6 B, 10 lb. Glauber's (p. 109) salt, and 3 lb. sulphuric acid. This gives a fine tint, very fast to acids, milling and light.
Purple.--Make the dye-bath with 3 lb. Chromotrop 10 B, 10 lb. Glauber's salt, and 3 lb. sulphuric acid.
The Chromotrops are remarkable for the fulness of the shades they dye, the brightness of their tint, and their fastness to acids, washing and light.
Purple.--Use 4 lb. Azo Fuchsine B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Bluish Purple.--A very dark shade of purple is dyed with 4 lb. Azo Acid Violet 4 R, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Bordeaux Reds.--These are shades that lie intermediately between the scarlets and the crimsons. They are in general duller than the scarlets, and have a more solid and fuller look; while they are less blue in tone than the crimson. They can be obtained from a large variety of dye-stuffs, and the recipes given below may be regarded as typical examples.
Bright Bordeaux Red.--Make the dye-bath with 1 lb. Azo Bordeaux, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil to shade. This is a very bright shade, of a somewhat bluish tone.
Cherry Red.--Make the dye-bath with 2-1/2 lb. Fast Acid Magenta B, 2-1/2 lb. Fast Yellow, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. This gives a fine deep shade.
Bright Cherry Red.--A very yellow shade of red, fast to milling, is dyed by making a dye-bath with 1-1/2 lb. Anthracene Yellow C, 3 lb. Diamine Fast Red F, 10 lb. Glauber's salt, 5 lb. acetate of soda, and 2 lb. bisulphate of soda. Work at the boil for one hour, then lift, add 3 lb. fluoride of chrome, re-enter the wool and work half an hour longer; wash and dry.
Deep Bordeaux Red.--The dye-bath is made with 4 lb. Diamine Fast Red F, 5 lb. acetate of soda, and 3 lb. bisulphate of (p. 110) soda. Work at the boil for one hour, then lift, add to the bath 3 lb. fluoride of chrome, re-enter the goods and work again for half an hour; lift, wash and dry.
Bright Cherry Red.--Make a dye-bath with 4 lb. Benzo Fast Red, 10 lb. Glauber's salt, and 2 lb. acetic acid. Work at the boil for one hour, then lift, add 3 lb. fluoride of chrome, re-enter the goods and work for half an hour longer; wash and dry.
Cherry Red.--Make the dye-bath with 2 lb. Azo Fuchsine G, 1-1/2 lb. Fast Yellow, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. Work at the boil for one hour.
Bluish Bordeaux Red.--For a very fast shade use 8 oz. Fast Acid Violet R, 8 oz. Orange G, 3/4 oz. Patent Blue B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. Work at the boil for one hour.
Bright Bordeaux Red.--A good bright and fast shade of red is dyed with 3-1/2 lb. Emin Red and 7 lb. bisulphate of soda. Work at the boil for one hour, then lift, add 3 lb. fluoride of chrome, work for three-quarters of an hour, then lift, wash and dry.
Bordeaux Red.--Use 3 lb. Titan Scarlet D, 1/4 lb. Titan Brown O, and 20 lb. salt. Work at the boil for one hour, then lift, wash and dry.
Claret Reds.--Claret reds are very useful shades and are great favourites of the dress-loving public. They are dark reds of a yellow tone, and can be dyed upon wool in a variety of ways, of which the following recipes just indicate a few.
Claret.--Make the dye-bath with 4 lb. Milling red R, 10 lb. Glauber's salt, and 4 lb. sulphuric acid.
Claret.--Use 4 lb. Archil Substitute N, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Claret.--Make the dye-bath with 2 lb. Bordeaux B L, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Claret.--A deep shade is dyed with 2-1/2 lb. Victoria Scarlet R, (p. 111) 2 lb. Victoria Rubine O, 1 oz. Cyanine Scarlet R, 2 lb. Victoria Rubine O, 1 oz. Cyanine B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Claret.--A fine deep shade is dyed with 2 lb. Azo Red A, 1/4 lb. Orange extra, 1/4 lb. Cyanole, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Maroon Reds.--From clarets to maroons is not a wide interval, they are both dark shade reds, the former tending to a yellow tone, the latter to a more bluish shade of red. A few recipes will be given to show some of the best methods of dyeing maroons.
Maroon.--Use 6 lb. Amaranth B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. This gives a fine bright shade.
Deep Maroon.--Make the dye-bath with 4-1/2 lb. Fast Acid Violet 10 B, 80 lb. Glauber's salt, and 3 lb. sulphuric acid. This gives a fine blue shade of maroon of great depth.
Maroon.--The dye-bath is made with 3 lb. Azo acid violet 4 R, 1 lb. Fast Yellow S, 1-1/2 oz. Fast Green Bluish, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Deep Maroon.--Make the dye-bath with 2 lb. Acid Magenta, 1/2 lb. Orange O, 1/2 lb. Patent Blue V, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Deep Maroon.--Make a dye-bath with 3 lb. Azo Acid Rubine, 1-1/2 oz. Acid Black B B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Maroon.--The dye-bath is made with 3 lb. Milling Red B, 1-1/2 oz. Naphthol Black 4 R, 10 lb. Glauber's salt, and 3 lb. sulphuric acid.
Deep Maroon.--Make the dye-bath with 1-1/2 lb. Victoria Scarlet R, 13 oz. Victoria Rubine O, 1/2 lb. Victoria Yellow, 2 lb. Keton Blue G, 10 lb. Glauber's salt, and 3 lb. sulphuric acid.
Bright Red.--A good shade is dyed with 4 lb. Lanafuchsine S G, and 10 lb. bisulphate of soda. Lanafuchsine S B dyes somewhat bluer shades.
Fast (p. 112) Red.--Dye with 4 lb. Milling Red B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Bright Scarlet.--Dye with 3 lb. Brilliant Cochineal 2 R, 10 lb. Glauber's salt, and 3 lb. sulphuric acid.
Deep Scarlet.--Dye with 3 lb. Brilliant Ponceau 4 R, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Pinks.--Pink.--Use 1-1/2 oz. Erythesine D, and 5 lb. acetic acid. These two pinks are very much alike and are very bright.
Bluish Pink.--Use 1-1/2 oz. Rose Bengale and 5 lb. acetic acid.
Pink.--Make the dye-bath with 3 oz. Azo Cochineal, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Bluish Pink.--Make the dye-bath with 3/4 to 1 oz. Fast Acid Violet R and a little Orange G, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Pink.--By using 1-1/2 oz. Fast Acid Violet R, 3/4 oz. Orange G, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, a good full pink is obtained.
Bluish Pink.--Use 2 oz. Fast Acid Violet R, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Many of the other acid reds may be used for dyeing pinks if from 2 to 4 oz. of dye-stuff be used.
Pink.--Use in the dye-bath 1-1/2 oz. Diamine Fast Red F, 5 lb. acetate of soda, and 3 lb. bisulphate of soda.
Coral Red.--Make the dye-bath with 1/2 lb. Diamine Scarlet B, 10 lb. Glauber's salt, and 1 lb. acetic acid.
Dark Cherry Red.--The dye-bath is made with 2-1/2 lb. Orange G G, 1 lb. Brilliant Orseille C, 3/4 oz. Cyanole extra, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Deep Crimson.--Use in the dye-bath 4 lb. Brilliant Orseille C, 1-1/2 oz. Cyanole extra, 3 oz. Orange G G, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Scarlet.--Make the dye-bath with 4 lb. Lanafuchsine S G, and 10 lb. bisulphate of soda. Work at the boil to shade.
Crimson.--Make (p. 113) the dye-bath with 4 lb. Lanafuchsine S B, and 10 lb. bisulphate of soda. Work at the boil to shade.
The Lanafuchsines, of which there are three brands, S G, S B, and 6 B, dye very good level shades of red from scarlet to crimson, which are of good fastness to milling, acids and light.
Salmon.--Use 1/2 lb. Rhodamine B, 1/4 oz. Naphthol Yellow S, 10 lb. Glauber's salt, and 2 lb. acetic acid.
Rose Red.--Use 1/4 lb. Lanafuchsine S B, 3 oz. Lanafuchsine S G, 10 lb. Glauber's salt, and 1 lb. sulphuric acid.
Salmon Red.--Use 1-1/2 oz. Lanafuchsine S G, 1/4 oz. Fast Yellow S, 10 lb. Glauber's salt, and 1/2 lb. sulphuric acid.
Deep Crimson.--The dye-bath is made with 2 lb. Naphthol Red C, 9 oz. Acid Magenta, 3/4 oz. Cyanole extra, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Purple Red.--Dye with 2-1/2 lb. Naphthol Red C, 3/4 lb. Acid Magenta, 1 oz. Cyanole extra, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Bordeaux Red.--Dye with 4 lb. Lanafuchsine S B, 1 oz. Orange extra, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Fawn Red.--Dye with 1/4 lb. Orange G G, 3 oz. Lanafuchsine S B, 1/2 oz. Cyanole extra, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Salmon.--Prepare the dye-bath with 1/4 oz. Fast Acid Violet R, 1/2 oz. Orange G, 10 lb. Glauber's salt, 1 lb. sulphuric acid. Work at the boil to shade.
The mordant reds are fairly numerous, and include both natural and artificial red dye-stuffs. The principle or property on which the application of this group of dye-stuffs to the dyeing of textile fabrics depends is that they are of an acid character and combine with metallic oxides, like those of iron, aluminium, or chromium to form insoluble coloured bodies, or "colour lakes" as they are called. The shade or tint (p. 114) of these colour lakes depends, firstly, upon the dye-stuff, and, secondly, upon the metallic oxide. Thus Alizarine with alumina gives a scarlet, with chrome a dark red, and with iron a dull violet. Alumina and chrome are the metallic mordants most commonly used in the dyeing of reds; sometimes tin is used, but never iron.
The coal-tar colour makers have placed at the service of dyers a great variety of mordant dyes, which may be classified somewhat roughly into groups, according to their chemical composition. The first group is called phenolic colours. These contain the group, or radical OH, hydroxyl, once or oftener. It is to the presence of this group that they owe their acid character and the property of combination with metallic oxides. To this group of dye-stuffs belong such dyes as Alizarine, Alizarine Cyanine, Anthragallol, Gambine, Coerulein, and some others. The natural red dye-stuffs, Cochineal, Brazil-wood, madder, etc., probably belong to this class.
None of these are essentially dyes of themselves, and used alone will not dye any fibre, it is only when they are brought into combination with the mordant that they will dye the wool fibre.
The next group may be called hydroxy-azo dyes, and are quite of modern introduction. They are azo dyes, one of whose constituents is a body like salicylic acid, amido-benzoic acid, dihydroxy-naphthalene-sulpho acid, which contain the group OH, hydroxyl with carboxyl COOH. The first group imparts phenolic characters, while the second gives true acid properties, and both of these acting together cause the dyes to be able to form colour lakes with metallic oxides. There is one point of difference between the two groups of dyes, the phenolic dyes are as a rule not dyes of themselves, some of them are practically free from colour, and it is only when brought into combination with the metallic oxide or (p. 115) mordant that they form a colour and dye a fibre. On the other hand the azo mordants are in general colouring matters, and can be used to dye wool without the aid of a mordant, the latter only serving to make the colour faster to light, acids, milling, etc., and it often has no material effect on the shade or tone of colour being dyed. Alizarine Yellow G G, Gambine Yellow, Anthracene Yellow, Chrome Violet, are examples of such dyes.
There are, however, some dyes (such as the Chromotrops, Azofuchsine, Anthracene Acid Browns, etc.) on which the mordant has a marked effect.
The methods adopted in practice for the application of this class of dyes are many and varied. The mordants used are alum, alumina sulphate, acetate of chrome, chrome alum, fluoride of chrome, ferrous sulphate and tin chloride, while, in addition, along with these true mordanting materials, assistant mordants are used, such as argol, tartar, tartaric acid, lactic acid, lignorosine, oxalic acid and sulphuric acid.
The mordanting may be done either before or after the dyeing, the first plan being that commonly adopted with the phenolic colours, while the second method may be used and is the best to use with azo-mordant dyes. Sometimes the mordanting and dyeing may be done in one bath, but this method is one which leads to a loss of colouring matter and often to the production of colours which are loose to rubbing, and cannot, therefore, be recommended.
Mordanting.--This operation is carried out in the same way in all cases. The goods are entered into the bath at a temperature of about 150° F. The heat is raised to the boil, and is then maintained for one and a half hours, after which the mordanted wool is lifted and well rinsed, when it is ready for the dye-bath. As mordanting materials bichromate of potash and fluoride of chrome are chiefly used when chrome mordants are required, sometimes chrome alum. (p. 116) With these are used sulphuric acid, oxalic acid, cream of tartar or argol, lactic acid, etc.
Which of these are used depends entirely on the results which are to be got and the dye-stuff to be used, more particularly is this the case when bichromate of potash is the mordanting material. When sulphuric acid is used as the assistant along with the bichrome, then there is formed on the wool fibre a deposit of chromic acid and chromium oxide, and this exerts an oxidising effect on the colouring matter or dye-stuff, which in some cases, as the Alizarine Blue, Alizarine Yellow, etc., leads to a destructive effect, and, therefore, the production of weak shades, so that it is not possible always to use an oxidising mordant. When tartar, argol, oxalic acid, lactic acids and other assistants of an organic nature are used, then a different effect is obtained, the bichromate is completely decomposed, and a deposit of chromium oxide formed on the wool. This does not exert any action on the colouring matter, and hence this mordant is known as the non-oxidising mordant. It may be pointed out that when wool is mordanted with potassium or sodium bichromate and sulphuric acid (oxidising mordant) it has a deep yellow colour, while when mordanted with bichromate or other chrome salt, and the organic assistants enumerated above (non-oxidising mordant), it has a green colour, and one sign of a well-mordanted wool is when it has a good bright tone free from yellowness.
Of the organic assistants tartar is undoubtedly the best in general use, and, although slow in its action, leaves a good deposit of oxide of chrome on the wool in a suitable condition to develop the best results on dyeing. Argols are only an impure tartar. They can only be used when dark shades are to be dyed. Oxalic acid does not work as well as tartar, and there is not so much chrome oxide deposited on the wool, while there is a slight tendency for a small proportion of this to (p. 117) be in the form of chromic acid. Of late years lactic acid and lignorosine have been added to the list of assistant mordants; both these give excellent results, they lead to a more complete and more uniform decomposition of the bichromate, and therefore the mordanting baths are more completely exhausted, so that rather less bichromate is required; the shades which are obtained are in general fuller and brighter. Examples of the use of these assistants will be found among the recipes given below.
With fluoride of chrome either oxalic acid or tartar is used, and a deposit of chromium oxide is formed on the wool, the general effect being the same as when bichromate of potash is used with oxalic acid or tartar.
Alumina is applied either in the form of alum or of sulphate of alumina, argol or tartar being used as the assistant, oxide of alumina being deposited on the fibre.
When ferrous sulphate (copperas) is used then tartar is almost invariably used as the assistant mordant, oxalic acid only rarely.
The dyeing with mordant dyes must be done in a special way and with great care, if uniform, level shades and fast colours are to be obtained.
The dye-bath must be started cold, and the wool be entered and worked for twenty to thirty minutes, the object being to cause the dye-stuffs to penetrate well into the substance of the fibre, then the temperature is slowly raised to the boil, not less than three-quarters of an hour being taken in doing so; the temperature is maintained at the boil for fully one and a half hours longer. During the boiling operation the mordant and dye-stuff combine together, and form the characteristic colour lake, and the boiling fixes this firmly on to the wool.
The water used plays a very important part. If too hard in character, the lime it contains shows a tendency to combine with (p. 118) the dye-stuff and form a colour lake, which is deposited in a loose form on the wool or in the bath, tending to make the shades dull and loose to rubbing. This defect can be remedied by adding a little acetic acid to the dye-bath, say about 3 lb. to 100 gallons of the water. It combines with and neutralises the influence of the lime, in so far as the formation of a loose colour lake is concerned; still the lime does unite with the dye-stuff, but the combination is formed more slowly, and in or on the wool fibre so that it is fast.
By working in the manner laid down above very fast shades can be dyed on wool with mordant dyes, and the following recipes will give the other details as to tints, shades, quantities, etc., not noted above.
Claret.--Mordant, 2 lb. bichromate of potash and 2 lb. tartar; dye, 8 lb. Alizarine Claret R.
Fawn.--Mordant, 3 lb. bichromate of potash and 1-1/2 lb. tartar; dye, 3 lb. Alizarine Orange N.
Maroon.--Mordant, 3 lb. bichromate of potash and 2-1/2 lb. tartar; dye, 15 lb. Alizarine Orange N.
Deep Crimson.--Mordant, 3 lb. bichromate of potash and 2-1/2 lb. tartar; dye, 8 lb. Alizarine Red 1 W S.
Lilac Rose.--Mordant, 1-1/2 lb. bichromate of potash and 1-1/2 lb. tartar; dye, 1 lb. Alizarine Red 1 W S.
Crushed Strawberry Tint.--Mordant, 2 lb. bichromate of potash and 1-1/2 lb. tartar; dye, 3 lb. Alizarine Red 2 W S.
Deep Claret.--Mordant, 3 lb. bichromate of potash and 2-1/2 lb. tartar; dye, 5 lb. Alizarine Red 1 W S.
Bright Fawn Red.--Mordant, 2 lb. bichromate of potash and 1-1/2 lb. tartar; dye, 1 lb. Alizarine Red 5 W S.
Scarlet.--Mordant, 10 lb. alum and 6 lb. tartar; dye, 4 lb. Alizarine Red 5 W S.
Rose.--Mordant, 6 lb. alum and 4 lb. tartar; dye, 1 lb. Alizarine Red 1 W S.
Deep (p. 119) Scarlet.--Mordant, 10 lb. alum and 6 lb. tartar; dye, 4 lb. Alizarine Red 1 W S.
Deep Maroon.--Mordant, 3 lb. bichromate of potash and 1 lb. sulphuric acid; dye, 5 lb. Alizarine Red 3 W S.
Bright Maroon.--Mordant, 3 lb. bichromate of potash and 2 lb. tartar; dye, 5 lb. Alizarine Red S W, 10 lb. Mordant Yellow.
Deep Fawn Red.--Mordant, 3 lb. bichromate of potash and 2-1/2 lb. tartar; dye, 10 lb. Alizarine Orange W and 1 lb. Mordant Yellow.
These typical recipes are here given to show what tints may be obtained from the alizarine and the quantity of dye-stuffs required. By using other proportions of dye-stuffs than those given a variety of other tints may be dyed.
The method of working described above is applicable to other mordant dyeing colours besides the alizarine reds, such as Alizarine Orange, Alizarine Blue, Anthracene Brown, Alizarine Cyanine, Galloflavine, Gambine, Chrome Violet, etc. It will therefore not be required to repeat this description of the process when the use of mordant colours for producing other colours, such as blues, navies, drabs, browns, etc., is dealt with.
Although the shades dyed with the alizarines and allied colouring matters are lacking in the brilliance characteristic of the azo scarlets, yet they have the very great advantage of being quite fast to washing, acids and light.
There is another method of using those alizarine reds that are sold in the form of powder, and which are distinguished by the letter S. They are of some value in dyeing heavy woollen cloths, and the method is indicated in the two recipes which follow:--
Brilliant Scarlet.--Prepare a dye-bath with 20 lb. Glauber's salt and 4 lb. Alizarine Red 1 W S, boil the wool in this for three-quarters of an hour; then lift, add to the same bath 4 (p. 120) lb. sulphuric acid, again work at the boil for three-quarters of an hour; then lift, add 10 lb. alum, re-enter the goods, and work three-quarters of an hour longer; then lift, wash and dry.
Claret.--Prepare a bath with 20 lb. Glauber's salt and 4 lb. Alizarine Red 1 W S, boil for three-quarters of an hour; then lift, add 4 lb. sulphuric acid, re-enter the wool, boil for three-quarters of an hour; then lift, add 3 lb. bichromate of potash, re-enter the wool, and boil for three-quarters of an hour longer; then lift, wash and dry.
Bluish Red.--Mordant, 2 lb. bichromate of potash and 2 lb. lactic acid; dye, 2 lb. Alizarine Red S. In this recipe there is used lactic acid as the assistant, and a very fine shade results.
Red.--Mordant, 3 lb. lignorosine, 2 lb. bichromate of soda and 1 lb. sulphuric acid; dye with 12 lb. Alizarine Orange 2 G.
Dark Bordeaux Red.--Mordant, 3 lb. lignorosine, 3 lb. bichromate of soda and 1-1/2 lb. sulphuric acid; dye, 12 lb. Alizarine S X.
Dark Red.--Mordant, 3 lb. lignorosine, 2-1/2 lb. bichromate of soda and 1-1/4 lb. sulphuric acid; dye, 6 lb. Alizarine Orange 2 G and 4 lb. Alizarine S X.
Lignorosine used as the assistant mordant in the above recipes works very well, and gives bright shades.
Fast Bordeaux.--Prepare a bath with 4 lb. Chromogene I, 1-1/2 lb. Alizarine Red 1 W S, 1 lb. Alizarine Red 5 W S, 1/2 lb. Fast Acid Violet R, 10 lb. Glauber's salt and 3 lb. sulphuric acid. Work at the boil for one hour, then lift; add to the same bath 3 lb. bichromate of potash and 1-1/2 lb. sulphuric acid. Re-enter the goods and work to shade, then lift, wash and dry.
Terra Cotta.--Make a dye-bath of 30 lb. Fustic, 8 lb. Turmeric, 30 lb. Sanders and 10 lb. Sumac. Boil the goods in (p. 121) this for one hour, then add 3 lb. sulphate of copper, previously dissolved in water, boil for one hour; cool, sadden with Copperas, using about 3-1/2 lb. or less if required; then rinse and dry.
Another method is to mordant the goods at a boil for one and a half hours in 2 lb. bichromate of potash and 2 lb. tartar. Drain and wash. Dye in a fresh bath with 8 lb. sanders and 10 lb. fustic; afterwards sadden with 1/4 lb. copperas; allow to stand one hour; wash and dry.
Orange Shades on Wool.
With Direct Dyes. Make a dye-bath with 2 lb. Titan Orange, 20 lb. Glauber's salt, and 1/2 lb. acetic acid. Work at the boil for one and a half hours, then lift, wash and dry.
Bright Orange.--Dye with 1-1/2 lb. Benzo Orange R, 10 lb. salt, and 1 lb. acetic acid, working at the boil for one hour.
Orange.--Dye with 2 lb. Chloramine Orange, 20 lb. salt, and a little acetic acid, working at the boil for one hour.
Orange.--Dye with 2 lb. Diamine Orange G C, and 20 lb. Glauber's salt.
Pale Orange.--Dye with 3 lb. Diamine Gold, 10 lb. Glauber's salt, and 5 lb. ammonium acetate.
Reddish Orange.--Dye with 3 lb. Diamine Orange D C and 20 lb. Glauber's salt.
Orange.--Dye with 2 lb. Diamine Scarlet B, 1 lb. Thioflavine S, and 20 lb. Glauber's salt.
Dark Orange.--Dye with 1 lb. Diamine Red 5 B, 1 lb. Thioflavine S, and 20 lb. Glauber's salt.
With Acid Colours. Orange.--Dye with 2 lb. Ponceau 3 G, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Bright Orange.--Dye with 2 lb. Mandarine G, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Orange.--Dye with 2 lb. Croceine Orange, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Bright (p. 122) Orange.--Use 3 lb. Orange G G, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, boiling for one hour.
Orange.--Use 3 lb. Orange R, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. Work at the boil. Orange Extra will give a slightly less red tone of orange, Croceine orange gives a good bright shade of a yellowish tone.
There are several brands of Orange dyes which can be used; they differ but little in shade from one another. In general they give fast colours. The Tropæolines also dye orange shades, but they are not so fast as the other dyes which have been named.
Gold Orange.--Make a dye-bath with 1/2 lb. Diamine scarlet B, 2 lb. Anthracene Yellow C, 50 lb. Glauber's salt, 5 lb. acetate of ammonia. Enter the wool, work for half an hour, then add 3 lb. bisulphate of soda. Boil again for half an hour, then lift. Add 3 lb. fluoride of chrome, re-enter the wool, boil again for half an hour, then lift, wash and dry. This gives a very fast orange.
With Mordant Dyes. Old Gold.--Mordant with 3 lb. bichromate of potash and 1 lb. sulphuric acid; dye with 6 lb. Alizarine Yellow R W.
Pale Orange.--Mordant with 6 lb. alum and 4 lb. tartar; dye with 1 lb. Alizarine Orange G G.
Deep Orange.--Mordant with 10 lb. alum and 6 lb. tartar; dye with 10 lb. Alizarine Orange N. This last dye-stuff gives a slightly redder shade of Orange than does the Alizarine Orange G.
Deep Orange.--Dye in a bath with 1-3/4 lb. Azo Alizarine Orange R R, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, and fix in the same bath with 1 lb. bichromate of potash.
Orange.--Dye in a bath with 1 lb. Alizarine Red 1 W S, 2 lb. Mordant Yellow O, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, then fix with 1-1/2 lb. bichromate of potash.
Orange.--Dye in a bath with 1 lb. Anthracene Red, 2 lb. Alizarine (p. 123) Yellow, 10 lb. Glauber's salt and 2 lb. sulphuric acid. After dyeing fix with 2 lb. fluoride of chrome.
Gold Orange. Mordant with 3 lb. bichromate of potash, and 2 lb. tartar, for one and a half hours at the boil; rinse. Then dye in a new bath with 1 lb. Alizarine Orange, 17 lb. Fustic extract. Work at 100° F. for half an hour, then heat gradually to the boil and dye for one and a half hours at that temperature; lift, rinse and wash.
Olive Yellow on Worsted Yarn.--Mordant the yarn by boiling for one hour or one and a half hours in a bath of 3 lb. bichromate of potash; then dye in a bath of 1-1/2 lb. Gambine Yellow and 10 lb. of fustic chips.
Red and orange form a kind of group of colours which shade off one into the other almost imperceptibly by using a range of dyes such as Croceine A Z, Brilliant Croceine 9 B, Brilliant Croceine 7 B, Brilliant Croceine 5 B, Brilliant Croceine 3 B, Brilliant Croceine M O O, Crystal Scarlet 6 R, Brilliant Cochineal 4 R, Brilliant Croceine B, Brilliant Cochineal 2 R, Orange E N Z, and Croceine Orange E N. It is possible to dye shades from a scarlet crimson to a bright orange.
Yellow Shades on Wool.
The number of yellow dye-stuffs is very great, and the variety of tints infinite. Yellow may be dyed with both natural and artificial dye-stuffs, and the recipes given will include examples showing the use of both kinds. Speaking generally, yellow dye-stuffs include amongst them some of the fastest colours known, and there is a larger proportion of fast yellow colouring matters than of any other class of dye-stuffs.
With Acid Yellows. Bright Yellow.--Make the dye-bath with 1 lb. Fast Yellow F Y, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil to shade.
Olive (p. 124) Yellow.--Prepare the dye-bath with 1 lb. Azo Carmine, 1-1/2 oz. indigo carmine, 1/2 lb. Fast Yellow, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil to shade.
Maize Yellow.--Prepare a dye-bath with 5 lb. acetate of ammonia, 3 oz. Anthracene Yellow C, 1/4 oz. Diamine Fast Red F. Work for twenty minutes at the boil, then add 3 lb. bisulphate of soda; work half an hour longer, and then wash and dry.
Bright Canary.--Prepare a dye-bath with 4 lb. bisulphate of soda, 1/2 lb. Nitrazine Yellow. Heat the bath to about 120° F., enter the goods and heat up to the boil, and work till the bath is exhausted, then lift; add to the dye-bath 3 lb. alum, 3 lb. tin spirits; re-enter the goods, and boil for twenty minutes longer; lift, wash and dry.
Bright Straw.--Dye with 3 oz. Phenoflavine and 20 lb. bisulphate of soda.
Straw.--Make the dye-bath with 1-1/4 oz. Azo Yellow, 1 dr. Cyanine B, 1 dr. Chromotrop 2 R, 10 lb. Glauber's salt, and 1 lb. sulphuric acid.
Greenish Straw.--Dye with 1/4 oz. Cyanine B, 1 oz. Victoria Yellow, 1/4 oz. Chromotrop 2 B, 10 lb. Glauber's salt, and 1 lb. sulphuric acid.
Olive Yellow.--Mordant with 3 lb. bichromate of potash and 1 lb. sulphuric acid; dye with 3 lb. Milling yellow O and 1 lb. acetic acid.
Bright Yellow.--A good shade is dyed in a bath of 2 lb. Milling yellow O, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil.
Olive Yellow.--Dye with 1-1/2 lb. Titan Yellow R, 10 lb. common salt, and 1 lb. acetic acid; after the colour has fully gone on to the wool, add to the bath 1-1/2 lb. fluoride of chrome and maintain at the boil for half an hour; then lift, wash and dry.
Deep Yellow.--The dye-bath is made with 1-1/2 lb. Titan Yellow R, (p. 125) 10 lb. common salt, and 1 lb. acetic acid, working at the boil to shade.
Yellow.--A good shade is dyed with 1-1/2 lb. Titan Yellow Y, 10 lb. common salt, and 1/2 lb. acetic acid, working at the boil to shade.
Golden Yellow.--Mordant with 3 lb. bichromate of potash and 2 lb. tartar; dye with 1 lb. Anthracene Yellow C.
Deep Golden Yellow.--Make the dye-bath with 3 lb. Anthracene Yellow C, and 3 lb. bisulphate of soda. Work at the boil for half an hour, then lift; add 3 lb. fluoride of chrome, re-enter the wool and work at the boil for another half-hour, then wash and dry.
Deep Olive Yellow.--Mordant with 3 lb. bichromate of potash and 2 lb. tartar; dye with 20 lb. fustic extract. This gives a very deep shade of olive Yellow.
Bright Lemon Yellow.--Make the dye-bath with 10 lb. Gambine Yellow, 7 lb. alum, and 2 lb. oxalic acid. Enter cold, then slowly heat to the boil and work to shade; then lift, wash and dry.
Leaf Yellow.--Mordant with 3 lb. bichromate of potash and 1/2 lb. sulphuric acid; then dye with 2 lb. Gambine Y and 1 lb. Yellow N.
Deep Leaf Yellow.--A somewhat deeper shade than the last is dyed by first mordanting with 2 lb. bichromate of potash and 1/2 lb. sulphuric acid, then dyeing with 2 lb. Gambine R and 1 lb. Yellow N.
Lemon Yellow.--Prepare a bath with 40 lb. fustic, 6 lb. alum, 6 lb. tartar, and 3/4 lb. tin crystals; enter the wool and work at the boil for one and a half hours, then lift, wash and dry.
Olive Yellow.--Mordant, 3 lb. bichromate of potash and 2 lb. tartar; dye, 3 lb. extract of fustic.
Deep Lemon.--Mordant, 3 lb. bichromate of potash and 2 lb. tartar; dye, 1 lb. Alizarine Yellow G G W.
Golden (p. 126) Yellow.--Mordant, 3 lb. bichromate of potash and 1 lb. sulphuric acid; dye, 10 lb. Alizarine Yellow G G W.
Light Straw.--Make the dye-bath with 3 oz. Anthracene Yellow B N, 5 lb. acetate of ammonia, and 3 lb. bisulphate of soda; work at the boil to shade, then lift, wash and dry.
Old Gold.--A very fine shade of old gold is obtained by dyeing in a bath of 3 lb. Anthracene Yellow C, 5 lb. acetate of ammonia, and 3 lb. bisulphate of soda. Work at the boil for three-quarters of an hour, then lift; add to the dye-bath 3 lb. fluoride of chrome, re-enter the wool, and work for one and a half hours longer at the boil; lift, wash and dry.
Deep Yellow.--Mordant, 3 lb. bichromate of potash and 2-1/2 lb. tartar; dye, 2 lb. Mordant Yellow D.
Pale Olive Yellow.--Dye with 3 lb. Anthracene Yellow G G, 10 lb. Glauber's salt, and 2 lb. acetic acid; after the dye-bath is exhausted of colour add 3 lb. fluoride of chrome, and work at the boil half an hour longer.
Gold Yellow.--Dye with 3 lb. Anthracene Yellow B N, 10 lb. Glauber's salt, and 3 lb. acetic acid; after half an hour's boil, add 1-1/2 lb. bichromate of potash, work for half an hour longer.
Gold Yellow.--Dye with 2 lb. Indian Yellow R, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Green Shades on Wool.
Of green shades there is an infinite variety, and these can be dyed in several ways. Either a simple green dye-stuff may be used or mixtures of blue and yellow dye-stuffs may be employed, this latter method being extremely common. It is somewhat interesting to notice that, notwithstanding the great prevalence of green in Nature, the dyer has at his command no natural green dye-stuff, but must, if he prefers to adopt natural dye-stuffs, use a mixture of blue and yellow dye-stuffs to produce green shades. There are but few green colouring (p. 127) matters derived from coal tar: Gambine, Dinitroso-resorcine, Alizarine Green, Brilliant Green, Malachite Green, Azo Green, Fast Green, Naphthol Green, Acid Green, Diamine Green, Benzo Green almost exhaust the list. Compared with the numerous red and blue dyes which are obtained from coal-tar products, green dyes are conspicuous by their fewness. On the other hand, the dyer has in the blue and yellow dyes from coal tar a means of producing any tint or shade of green he may require.
Members of all the classes of basic, direct, acid, azo and mordant dyes, can be found among the dye-stuffs which can be used in dyeing green, and the methods and principles of their application have been fully described in previous pages. The following recipes contain all the practical information that is needed:--
With Direct Dyes. Dark Green.--The dye-bath is made with 1 lb. Titan Blue 3 B, 1 lb. Titan Yellow Y, 2 lb. salt, and 1/2 lb. acetic acid.
Bright Green.--Prepare a dye-bath with 1 lb. Titan Yellow G, 1 lb. Titan Blue 3 B, 20 lb. salt, and 1/2 lb. acetic acid, working at the boil for one hour.
Dark Green.--Make a dye-bath with 4 lb. Acid Blue 4 S, 2 lb. Titan Yellow Y, and 5 lb. acetate of ammonia, working at the boil to shade.
Blue Green.--Make the dye-bath with 6 lb. Acid Blue 4 S, 2-1/2 lb. Titan Yellow Y, and 5 lb. acetate of ammonia, working at the boil to shade.
Bottle Green.--The dye-bath is made with 5 lb. Acid Blue 4 S, 2-1/2 lb. Titan Yellow Y, and 5 lb. acetate of ammonia, working at the boil to shade. The greens shown in the last three recipes are of a very satisfactory character, and show how, by the use of acetate of ammonia in the dye-bath, the direct dyeing Titan colours can be combined with acid colours.
Green.--Make the dye-bath with 5 lb. Glauber's salt, 5 lb. acetate (p. 128) of ammonia, 2 lb. Sulphon Cyanine, and 1-1/2 lb. Chrysophenine.<7p>
Dark Green.--The dye-bath is made with 2 lb. Sulphon Cyanine, 3/4 lb. Chrysophenine, 5 lb. Glauber's salt, and 5 lb. acetate of ammonia.
Pale Russian Green.--Make the dye-bath with 1/2 lb. Sulphon Cyanine, 2-1/2 oz. Chrysophenine, and 10 lb. Glauber's salt.
The last three shades have the merit of being fast to milling, and fairly so to light.
Olive.--Make a dye-bath with 1 lb. Nyanza Black B, 1 lb. Chrysamine, and 20 lb. Glauber's salt. Work at the boil to shade, lift, wash and dry.
With Acid Dyes. Blue Green.--Make a dye-bath with 10 lb. Glauber's salt, 2 lb. sulphuric acid, 2 lb. Patent Blue N, and 1 lb. Azo Yellow, working at the boil.
Sage Green.--The dye-bath is made with 10 lb. Glauber's salt, 2 lb. sulphuric acid, 2 lb. Azo Yellow, and 1 lb. Patent Blue N, working at the boil.
Olive Green.--Make the dye-bath with 3 lb. Naphthol Green B, 10 lb. Glauber's salt, 15 lb. bisulphate of soda, and 1 lb. copperas, working at the boil to shade.
Bright Green.--Make the dye-bath with 10 lb. Glauber's salt, 5 lb. bisulphate of soda, and 1-1/2 lb. Acid Green B, working at the boil to shade.
Emerald Green.--The dye-bath is made with 1/2 lb. Acid Green B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. The wool might also be previously mordanted with 15 lb. hyposulphite of soda, and 5 lb. sulphuric acid at the boil for one and a half hours, when it will give a bright shade of emerald green.
Grass Green.--Dye a medium indigo bottom on the wool from the vat, then dye in a bath with 1 lb. Milling Yellow O, 5 lb. Glauber's salt, and 5 lb. bisulphate of soda; lift, wash and dry.
The (p. 129) last recipe shows the use of the indigo vat in giving the blue constituent in dyeing greens and other compound colours on wool. This, while being a very effective method of dyeing, yet necessitates two operations which add very materially to the cost of dyeing such shades, hence it is not used for dyeing low class woollen fabrics, but for better class goods it is frequently adopted, fast colours being thus obtained.
In thus using the indigo vat as a bottom dye regard to the properties of indigo must be paid in carrying out any subsequent dyeing operation, so that the indigo on the fibre be not destroyed. As a rule, the indigo will resist any ordinary baths made with Glauber's salt, acetate of ammonia, sulphuric or acetic acids, but it will not resist mordanting operations with bichromate of potash, for the latter salt destroys the indigo. Fluoride of chrome, chrome acetate, or alum, may be used as mordants if necessary.
Pale Sea Green.--The dye-bath contains 1 oz. Cyanine B, 1 oz. Azo Yellow, 5 lb. Glauber's salt, and 1 lb. sulphuric acid.
Moss Green.--The dye-bath is made with 1/2 oz. Chromotrop 2 R, 2 oz. Cyanine B, 4 oz. Fast Acid Blue R, 3-1/4 oz. Azo Yellow, 5 lb. acetic acid, and 10 lb. Glauber's salt.
Deep Moss Green.--Prepare the dye-bath with 4-1/2 oz. Cyanine B, 9 oz. Fast Acid Blue R, 4-1/2 oz. Azo yellow, 1/2 oz. Chromotrop 2 R, 5 lb. acetic acid, and 10 lb. Glauber's salt.
Blue Green.--A very fine shade of blue green is dyed with 9-1/2 oz. Cyanine B, 1-1/4 lb. Fast Acid Blue R, 4 oz. Azo Yellow, 5 lb. acetic acid, and 10 lb. Glauber's salt.
Emerald Green.--A pale, but brilliant shade of green is dyed with 1-1/4 oz. Patent Blue V, 4-1/4 oz. Azo Yellow, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Bright Leaf Green.--Dye in a bath with 13 oz. Victoria Yellow, (p. 130) 1/2 lb. Patent Blue V, 1/2 oz. Chromotrop 2 R, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Deep Leaf Green.--The dye-bath is made with 22 oz. Cyanine B, 1 lb. Azo Yellow, 2-1/2 oz. Chromotrop 2 R, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Bright Peacock Green.--The dye-bath is made with 5 oz. Chromotrop 6 B, 4 oz. Patent Blue V, 7 oz. Azo Yellow, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Dark Beige Green.--Make the dye-bath with 1/2 lb. Fast Green Bluish, 6 oz. Fast Yellow F Y, 4-1/2 oz. Azo Fuchsine G, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Invisible Green.--Make the dye-bath with 1-1/2 lb. Fast Green Bluish, 1-1/4 lb. Fast Yellow F Y, 1 lb. Azo Fuchsine G, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Pale Sage Green.--Make the dye-bath with 1 lb. Azo Acid Brown, 1/2 lb. Fast Acid Violet 10 B, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Bright Grass Green.--Make a dye-bath with 10 lb. Glauber's salt, 2 lb. sulphuric acid, 3/4 lb. Phenoflavine, 3/4 lb. Azo Carmine B, and 5-3/4 lb. extract of indigo.
Moss Green.--Prepare a dye-bath with 1 lb. Azo Acid Brown, 1/4 lb. Fast Acid Violet 10 B, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Dark Sage Green.--Make the dye-bath with 3 lb. Azo Acid Brown, 1/2 lb. Fast Acid Violet 10 B, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Emerald Green.--A fine shade of emerald green can be dyed in a bath which is made from 1/2 lb. Fast Green Bluish, 1 lb. Fast Yellow F Y, 1 lb. Acid Violet 6 B, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Bottle Green.--Make a dye-bath with 1-1/2 lb. Victoria Violet 8 B S, 3/4 lb. Victoria Yellow, 2 oz. Naphthol Yellow S, 1 oz. Fast Acid Violet R, 1/2 oz. Cyanine B, 10 lb. Glauber's salt and 2 lb. sulphuric acid. Work for one hour at the boil, then (p. 131) lift; add 3 lb. fluoride of chrome, re-enter the wool, and work for half an hour at the boil.
Pale Pea Green.--A fine bright shade is dyed in a bath containing 1-1/2 oz. Cyanole, 3/4 oz. Naphthol Yellow and 10 lb. bisulphate of soda. By increasing the quantity of dye-stuff in proportion to the material, fine deep shades of green can be dyed.
Deep Electric Green.--Make the dye-bath with 2 lb. Cyanole, 1 lb. Indian Yellow G and 10 lb. bisulphate of soda, working at the boil for one hour; then lift, wash and dry.
With Mordant Dyes. Green.--Mordant with 10 lb. alum, 1 lb. bichromate of potash and 16 lb. tartar. Dye with 10 lb. indigo extract, 2 lb. fustic extract and 3 lb. alum, working at the boil; lift, wash and dry.
Dark Green.--Mordant with 3 lb. bichromate of potash, 8 lb. alum and 3 lb. tartar. Dye with 10 lb. extract of indigo, 2 lb. extract of fustic and 3 lb. alum, working at the boil.
Sea Green.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar at the boil for one and a half hours. Dye with 1-1/4 lb. Alizarine Blue D N W, 3-3/4 lb. Alizarine Yellow and 5 oz. Alizarine Brown, at the boil for two hours.
Bronze Green.--Make a dye-bath with 2 lb. Cyanole extra, 2 lb. Tropeoline O, 1 lb. Archil Substitute N and 10 lb. bisulphate of soda, working at the boil to shade.
Green.--A very fine shade of green is dyed as follows: Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar. Dye with 4 lb. Alizarine Blue D N W, 1-1/2 lb. Patent Blue A and 2-3/4 lb. Alizarine Yellow.
Blue Green.--Mordant as in the last recipe. Dye with 6 lb. Alizarine Blue D N W, 1-1/2 lb. Patent Blue A, and 1-1/4 lb. Alizarine Yellow G G W.
Bright Pale Sage Green.--Mordant with 3 lb. bichromate of potash and 2 lb. sulphuric acid. Dye with 5 lb. Alizarine Yellow (p. 132) G G W, 3/4 lb. Alizarine Brown and 1-1/4 lb. Alizarine Blue D N W.
Deep Sage Green.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar. Dye with 4 lb. Alizarine Yellow G G W, 3-1/4 lb. Anthracene Brown and 2-1/4 lb. Alizarine Blue D N W.
Pale Sea Green.--Mordant with 2 lb. bichromate of potash and 1-1/2 lb. tartar. Dye with 1 lb. Coeruleine B.
Bottle Green.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar. Dye with 20 lb. Coeruleine S W.
Slate Green.--Mordant with 2 lb. bichromate of potash and 1-1/2 lb. tartar. Dye with 3 lb. Alizarine Green S.
Invisible Green.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar. Dye with 17-1/2 lb. Alizarine Green S.
Peacock Green.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar. Dye with 8 lb. Alizarine Green S.
Dark Bottle Green.--Mordant with 4 lb. bichromate of potash and 3 lb. tartar. Dye with 15 lb. Anthracene Blue W G, and 1-1/2 lb. Mordant Yellow.
Invisible Green.--Mordant with 3-1/2 lb. bichromate of potash and 2-1/2 lb. tartar, working at the boil for one and a half hours. Dye with 20 lb. Alizarine Green S W, and 1 lb. acetic acid.
Sage Green.--Give a medium indigo ground to the wool in a vat, then dye for one hour at the boil in a vat containing 1/2 lb. Anthracite Black B, 2 lb. Anthracene Yellow C, 2 oz. Diamine Fast Red F, and 5 lb. acetate of ammonia; then lift, add 3 lb. fluoride of chrome, re-enter into the dye-bath and work half an hour longer at the boil; lift, wash and dry.
Peacock Green.--Give a medium indigo bottom on the vat, then dye for one hour at the boil in a dye-bath made with 1/2 lb. Anthracene Yellow C, 2 oz. Diamine Fast Red F, and 5 lb. acetic acid; then lift, add 3 lb. fluoride of chrome, work for half an hour longer at the boil, then lift, wash and dry.
Bottle Green.--Mordant by boiling in a bath of 3 lb. copperas (p. 133) and 1 lb. oxalic acid. Dye in a bath with 15 lb. Gambine R.
Light Green.--Mordant with 3 lb. copperas and 1 lb. oxalic acid. Dye with 2-1/2 lb. Gambine Y.
Medium Green.--Mordant as in the last dye with 10 lb. Gambine Y.
Deep Grass Green.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar. Dye with 9 lb. Coerulein and 1-3/4 lb. Galloflavine.
Bright Grass Green.--Mordant with 4 lb. copperas and 1 lb. oxalic acid. Dye with 5 lb. Gambine Y, 1/2 lb. Yellow N, and 2 lb. bisulphate of soda.
Shades dyed with Gambine are very fast to milling and light.
Pale Sage Green.--Mordant with 3 lb. bichromate of potash and 1 lb. tartar. Dye with 1/2 lb. Milling Yellow O, 2 lb. Alizarine Black S W, and 2 lb. acetic acid.
Medium Green.--Mordant with 2-1/2 lb. bichromate of potash and 1-1/2 lb. oxalic acid. Dye with 1-1/2 lb. Diamond Yellow B, 3-1/2 lb. Brilliant Alizarine Blue G, and 1 lb. acetic acid.
Invisible Bronze Green.--Give a medium bottom on the indigo vat and then mordant with 3 lb. fluoride of chrome and 2 lb. tartar. Finally dye with 3 lb. Alizarine Bordeaux S, and 4 lb. Diamond Flavine, working at the boil for two hours.
Pale Slate Green.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar, and then dye with 1 lb. Alizarine Blue D N W, Alizarine Yellow and 5 oz. Alizarine Brown.
Light Green.--Mordant in the usual way with 2-1/2 lb. bichromate of potash and 2 lb. tartar. Dye with 1 lb. Methylene Blue and 1 lb. fustic extract, working at the boil.
Fast Green.--Mordant with 8 lb. alum, 2 lb. bichromate of potash, 2 lb. sulphuric acid and 3/4 lb. tin salt. Dye with 20 (p. 134) lb. indigo extract and 10 oz. fustic extract, working at the boil for one and a half hours.
Bottle Green.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar. Dye with 4 lb. extract of fustic, 1 lb. extract of logwood, and 2 oz. Anthracene Red. Work for one and a half hours, then add 3/4 lb. copperas, and work for half an hour longer.
Dark Green.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar. Dye with 1-1/2 lb. Methylene Blue, 1-1/2 lb. extract of logwood, and 4 lb. extract of fustic, working at the boil for two hours.
Olive.--Prepare a dye-bath with 1-1/2 lb. Yellow N, 1/4 lb. Archil Substitute, 4 lb. extract of indigo, 10 lb. Glauber's salt, 2 lb. sulphuric acid, and 2 lb. alum, working at the boil to shade.
Bright Green.--Prepare a dye-bath containing 8 oz. Acid Green Extra and 10 per cent. bisulphate of soda. Enter at 130° F., raise to the boil, boil for three-quarters of an hour, and rinse.
Bluish Green.--Prepare a dye-bath containing 8 oz. Fast Acid Green B N and 10 lb. bisulphate of soda. Enter at 130° F., raise to the boil, boil for three-quarters of an hour, and rinse.
Bluish Green.--Prepare a dye-bath containing 8 oz. Cyanole Green 6 G and 10 lb. bisulphate of soda. Enter at 130° F., raise to the boil, boil for three-quarters of an hour, and rinse.
Turquoise Green.--Prepare a dye-bath containing 8 oz. Cyanole Green B and 10 lb. bisulphate of soda. Enter at 130° F., raise to the boil, boil for three-quarters of an hour, and rinse.
Slate (p. 135) Green.--Mordant the wool by boiling for one and a half hours in a bath containing 3 lb. bichromate of potash, 1-1/4 lb. Copper sulphate and 2-1/4 lb. tartar; then rinse well, and dye in a bath containing 2-1/2 lb. Logwood Extract (dry), 1-1/4 lb. Fustic Extract (dry), and 3 lb. Sumac. Enter the goods in a warm bath, work for half an hour, then raise to the boil and work for three-quarters of an hour; lift, and sadden by adding 6 oz. Copperas. After re-entering the goods, work to shade.
Olive.--Boil two hours in a bath consisting of 1-1/2 lb. tin salt, 2-1/2 lb. bichromate of potash, 10 lb. alum and 2-1/2 lb. sulphuric acid. Then enter in a boiling dye-bath containing 1-1/2 lb. alum, 4 lb. fustic extract and 3-1/2 lb. indigo extract.
Fulling Fast Olive.--For one hour upon a bath containing 50 lb. Fustic, 5 lb. Bluestone, 2 lb. Tartar, 4 lb. Sumac, 1 lb. Copperas; lift and wash.
Fast Bright Olive.--Boil for one hour upon a bath of 50 lb. Fustic, 3 lb. Bluestone, 2 lb. tartar, 1 lb. copperas, 2 oz. indigo extract.
Yellow Olive.--Prepare a bath containing 10 lb. Glauber's salt, 1-1/2 lb. Anthracene Yellow B N, 2 lb. extract of indigo, 3 oz. Orange E N Z, 4 lb. sulphuric acid. Enter yarn at 160° F., give three turns, raise the temperature slowly to the boil, turn to shade; lift, and wash.
Olive Green.--Mordant with 2 lb. potash bichromate, 1-1/2 lb. sulphate of copper, 1/2 lb. sulphuric acid. Boil for an hour and a half. Dye in a bath with 8 lb. Fustic extract, 5 lb. Sumac, 5 lb. Logwood, at the boil for an hour and a half.
Olive Bronze.--Make the dye-bath with 10 oz. Fast Yellow S, 5 lb. Indigo extract, 5 oz. Orange E N Z, 4 lb. sulphuric acid, 10 lb. Glauber's salt. Enter yarn at 140° F., work for a few minutes, then bring slowly to the boil and work to shade.
Emerald Green.--Prepare the dye-bath with 1 lb. Acid Green (p. 136) B N, 2 oz. Naphthol Yellow S, 10 lb. Glauber's salt, 2 lb. sulphuric acid. Enter cold, then raise to the boil and work for a quarter of an hour; wash and dry.
Invisible Green.--First mordant the wool in a bath containing 3 lb. bichromate of potash, 1-1/2 lb. copper sulphate, 1 lb. sulphuric acid. Work at the boil for one and a half hours, then dye in a fresh bath containing 2 lb. Milling Yellow O, 2 lb. Logwood extract, 20 lb. Glauber's salt. Work at the boil for one and a half hours, then lift, wash and dry.
Sea Green.--Prepare a dye-bath with 5 lb. Glauber's salt, 2 lb. sulphuric acid, 2 lb. indigo extract, 1/2 per cent. Acid Green blue shade. Dye as usual.
Cyprus Green B, and Cyprus Blue B, belong to a new group of dyes that owe their value in wool dyeing to the fact that the dyeings after being treated with copper sulphate become very fast to light and washing. Three per cent. of each gives very full shades of bluish green or dark blue. The dyeing is done with Glauber's salt and acetic acid when reddish shades are got; these in a bath of copper sulphate turn green or blue.
Blue Shades on Wool.
There are a very large number of blue artificial dyes of every class, but only a few natural ones, indigo and logwood, and with these every imaginable tint and shade of blue from the palest sky tints to the darkest navy blue or blue black can be produced.
While some of the blue colouring matters possess no great powers of resistance to light, air, washing, etc., the great majority are remarkable for their fastness to those destructive agencies.
There are but two natural dye-stuffs, indigo and logwood, from which blue tints can be dyed. With the former, a great variety of shades can be dyed of a satisfactory character as regards (p. 137) fastness; with the latter, only dark blues can be dyed, these are fairly fast to milling, but only moderately so to light.
The artificial blues derived from coal tar are very numerous, and representatives of all classes, direct, basic, acid and mordant of dye-stuffs may be found among them. The direct blue dyes do not work very well on wool. They are apt to dye very red, and somewhat dull shades, which are, however, fairly fast to washing and light. The basic blue dyes are fairly numerous, and may be used to dye from pale sky to deep navy tints. They are apt to work somewhat unevenly on to wool, owing to their great affinity for the fibre. They give shades possessing some degree of resistance to light, but which are not very fast to washing and milling, although, in this respect, there are very great differences among them. The acid dyeing blues are fairly numerous, but they dye a great variety of tints, usually fairly fast to washing, milling and light. The mordant blues are pretty numerous and of great value for dyeing wool, as they give shades which are remarkable for their fastness to light, acids and milling, hence they are most extensively used, especially for dyeing fabrics that are subject to very hard wear.
Indigo Dyeing.--It will be most convenient to begin the description of the methods of dyeing blues by showing how, and in what manner, indigo is applied in wool dyeing.
The dyeing of indigo on wool is effected in two ways, either in the usual way with acid baths, as with acid scarlets, when the so-called indigo extract is used, or in vats, when indigo itself forms the dye-stuff.
Indigo is, as all dyers know, or should know, a natural dye-stuff, prepared from the leaves and twigs of the indigo plant by a species of fermentation which produces the indigo in a soluble form from the indigo substance in the plant, followed by oxidation which results in the separation of the indigo from this solution.
It (p. 138) comes into this country in the form of lumps, which have a dark blue to bronze blue colour. The dye-stuff is insoluble in water, cold alcohol, alkalies or weak acids. When heated with strong and fuming sulphuric acid it dissolves, forming a blue liquor from which the colouring matter may be obtained on addition of soda in the form of a paste, which is used in wool and silk dyeing under the name of indigo extract. But dissolving in sulphuric acid materially affects the properties of indigo as a dye-stuff, as will be seen later on.
By the action of reducing agents the insoluble blue indigo is converted into a soluble white indigo. This body is rather unstable, and on exposure to the air it rapidly becomes oxidised and converted back again into the blue indigo. Upon this principle is based the application of indigo in dyeing by means of the vat.
Various methods may be adopted to cause the indigo to become dissolved. These may be divided into two groups: (1) Fermentation vats, in which the action of reducing agents is brought about through the influences of the fermentation of organic bodies, such as woad, bran, treacle, etc; (2) Chemical vats in which the reducing effect is brought about by the reaction of various agents on one another.
Of such vats the copperas and lime and the hydrosulphite vats are examples. The fermentation vats, when in good order, work well and give good results, but they are most difficult to prepare or set. The chemical vats are the easiest to work, and (especially the hydrosulphite vats) are coming to the fore, and are gradually driving out the fermentation vats.
The actual method of dyeing with the indigo vat is the same with all methods of preparation. The material to be dyed is well wetted or wrung out in water. It is then dipped into the vat, handled a few minutes to ensure its thorough impregnation, then lifted out, the surplus liquor wrung out, and (p. 139) the material exposed to the air, when the indigo white on it soon absorbs oxygen and turns into blue indigo.
With these few preliminary remarks the methods of setting the various indigo vats will now be described in detail.
Woad Indigo Vats.--This is one of the most difficult of the various methods of setting vats. There are so many opportunities for it to go wrong, and to be able to set a woad vat successfully will go far to make a man a successful indigo dyer. No two woad vat dyers use exactly the same recipe in setting a woad vat, and each considers he has a secret art by means of which he ensures the successful working of this vat, and this he jealously guards. All these differences in the manner of setting the vat are brought about not by any radical differences in the materials used, but by some unnoticed differences in other surroundings; differences in the mean temperature of the water used, in the general conditions of the atmosphere of the indigo shed and in other similar circumstances, all of which have a material influence on the development of the vat, but which are, in the majority of cases, overlooked by the indigo dyer, the result being that a method of working which is successful in one place would not be so in another. The fermentation processes depend upon the reducing action brought about by certain organisms of the nature of the yeast plant which grow and develop in such vats.
To ensure the proper growth and development of these organisms every condition must be perfect, correct temperature, proper proportions of food for them to live on, and a certain degree of alkalinity or acidity of the vat, and these points are most difficult to regulate as they will vary very much from time to time.
A successful vat maker is one who closely observes his vats, and the way in which they are working, and who, as the (p. 140) result of such observations, is able to tell in what way his vats are deficient, so that he may know how to supply that deficiency.
The following method of setting a woad vat may be adopted. It is calculated for 100 gallons of liquor. The vat is filled with hot water, and 80 lb. of woad are allowed to steep overnight in it, having first been well stirred into the water, so as to ensure that every part is wetted out. The next morning there is added 8 lb. madder, 12 lb. bran, 5 lb. quick-lime (previously slaked with water), and 2-1/2 lb. soda. These are thoroughly stirred together, then from 5 to 7-1/2 lb. indigo is stirred in. The indigo should have been previously ground into a fine paste with water. The temperature of the vat should now be maintained at from 115° to 125° F. for two to three days, at the end of which time it ought to be in a state of quiet working. Should it be found that the fermentation is going on too rapidly, a little lime may be thrown in, which will retard it. On the other hand, if it should not be going on with sufficient energy, this may be remedied by adding a little bran, or better, a little treacle.
When in perfect condition the vat should have a slight smell of ammonia. If this is not noticed it indicates that the vat is deficient in alkalinity, and a little more lime should be added. Soda may be used in the place of lime, but it is so much more energetic in character that any additions of it have to be made with great care, or the vat will become too alkaline in character, and the fermentation will go on too rapidly, the ammoniacal odour is lost, and a peculiar putrid smell takes its place. As soon as this is noticed, lime ought to be added to retard the fermentation and to develop the ammoniacal smell. The colour of a good well-set vat is olive brown.
When all the indigo is dissolved and the colour of the vat (p. 141) is a clear olive yellow to brown the vat is then ready for dyeing, and may be used for a long time, until, in fact, the deposit gets too large and the wool becomes dirtied. But it must not be continually worked, or it will give bad shades and loose colours. When in a bad condition it will usually turn of a dark brown colour, and give dull greenish shades. To remedy this there should be added some bran, treacle, and a little madder, as well as indigo, and the vat should be left for a day, at a temperature of 130° F., to get up to full strength again. Every night when in work indigo ought to be added to the vat in proportion to that consumed during the day, with bran and lime, the latter in not too great amount, just sufficient to keep it of the necessary alkalinity.
Hydrosulphite Vat.--This is one of the best vats to use in dyeing with indigo on wool, or, indeed, on any textile fabric. It is easy to prepare and cleanly to work. While depending solely on chemical action for its preparation and use, it is freer from those peculiar defects to which organic vats, like the woad vats, are liable.
There is a further advantage about this vat, it is not necessary to prepare each individual vat separately, but a strong mother liquor or concentrated indigo solution may be prepared, and this only requires letting down with water to produce a vat of any required strength.
In the preparation of this vat, which was devised by Schutzenberger and Lalande, bisulphite of soda and zinc dust are used with either quick-lime or caustic soda. The bisulphite of soda is allowed to act on the zinc as will be detailed when an acid solution of sodium hydrosulphite NaHSO2, more strictly hydrogen sodium hydrosulphite, is obtained. The acid solution of hydrosulphite has the property of rapidly reducing and dissolving indigo, and this solution may be used in dyeing. To prepare the hydrosulphite a vessel (p. 142) which is fitted with an agitator and can be closed is filled with zinc, either in the form of dust, foils, or granules. Then bisulphite of soda of 50° to 60° Tw. strength is poured over the zinc in sufficient quantity to cover it. All access of air should be avoided as much as possible, as it leads to oxidation. In the case of using zinc powder the action is often so rapid as to lead to heating, which also should be avoided. The operation takes from an hour to two hours, when the liquor may be drawn off. It must be used immediately to dissolve the indigo; or otherwise, as it is a very unstable body, it is liable to decompose and become oxidised, when it loses its solvent properties. If more hydrosulphite is required, fresh bisulphite may be poured over the zinc which is left unused in the vessel; if no more is wanted the zinc which is left should be well rinsed in water and the vessel filled with water, so as to prevent any oxidation of the zinc, and so keep it ready for use when required. The liquor thus made will usually have a specific gravity of 62° Tw. The zinc which is used up in the preparation of the liquor is replaced by fresh zinc from time to time.
The liquor so obtained is, as stated above, rather unstable, and contains acid sodium hydrosulphite. By mixing with milk of lime, the acidity is neutralised, zinc oxide and calcium sulphite are thrown down, and a solution of neutral sodium hydrosulphite is obtained which is more stable and can be kept longer without decomposition. To prepare this, take 10 gallons of the acid liquor, as prepared in the manner described above, and mix it with 48 lb. of milk of lime, which is made from 2 lb. good quick-lime. Stir well together, allow all sediment to settle, or better, filter-press the mass. A liquor of 36° Tw. strength will usually be obtained. Do not let it stand too long before use, make it alkaline by adding a little lime.
To make the mother or stock indigo, the following method of (p. 143) procedure may be adopted. Indigo, say 10 lb., is ground into as fine a paste as possible with 13 lb. milk of lime, of such a strength that 1 gallon shall contain 30 oz. quick-lime. To this is then added so much of either the acid or the neutral sodium hydrosulphite as can be made from 90 lb. of bisulphite of soda, the mixture being kept at 150° F., until a comparatively clear, greenish yellow solution is obtained, this will contain about 1 lb. of indigo per gallon.
This mother liquor may be used in setting the vat as follows. The vat is filled with water which is heated to 120° F., about 200 gallons being used. To this is then added 1 gallon of either hydrosulphite or bisulphite of soda to destroy the free oxygen it contains, and prevent it from oxidising the indigo solution, which is next added. The quantity of the latter is solely regulated by the depth of shade it is desired to dye, and as soon as the requisite quantity has been added the dyeing may be proceeded with at once, and the first portion of goods put through will soon show the dyer whether too much or too little of the mother indigo has been added.
Continued use and the consequent agitation of the vat thereby generated causes it to become oxidised, and the vat acquires a greenish colour, and does not give fast colours. When this is noticed the use of the vat is stopped; it is heated to about 160° F., and a little lime and hydrosulphite added, when all the oxidised indigo in the vat will speedily be reduced, and the vat put into a workable condition again. By use this vat tends to become alkaline, and consequently will spoil the wool, making it harsh and brittle. This is remedied by adding a little hydrochloric acid.
Holliday's Patent Indigo Vat.--Messrs. Read Holliday & Sons have patented an improved method of making an indigo solution and the method of using it. They supply the indigo in the form of solution in two strengths, ordinary and concentrated. Both are used in the same way, only of the latter less, (p. 144) about one-fourth to one-third, is required than of the former. For those who would wish to buy their indigo ready prepared for use these are very convenient forms.
The best way of working the vat for wool is the following: 40 gallons of water heated to about 50° C., add 1/4 lb. of a mixture of 1-1/4 gallons bisulphite of soda, 52° Tw., and 1 lb. zinc dust, and, say, 1/2 gallon to 2 gallons, of the patent indigo solution, according to the depth of shade required. The boiled out wool is worked below the surface of the liquor for about three minutes, then taken out, and the excess of liquor squeezed back into the vat, the whole operation is repeated until the shade is arrived at. After dyeing, rinse in an acid bath of 1° to 2° Tw.
The advantages of this new vat are that brighter shades are obtained and the darker shades with fewer dips, while the goods are dyed cleaner and the shades are more quickly obtained, and, we think, somewhat faster than by the other process.
There is also the advantage that no lime or other alkali is used with this new indigo vat. The wool should be boiled out before dipping, if the best results and even shades are desired.
Potash-Indigo Vat.--This is also a fermentation vat, and is set in the following manner: 5 lb. of madder and 4 lb. of bran are mixed with 50 gallons of water and heated for from three to four hours, until a temperature of from 180° to 212° F. is attained. Then 15 lb. of carbonate of potash are added and the liquor is allowed to cool down to about 120° F. Next 10 lb., more or less according to shade required, of finely ground indigo is added, and the whole is left for from forty-eight to sixty hours to ferment, being stirred up at intervals of twelve hours. This vat ferments in much the same way as the woad vat, and presents the same general appearances. It is not so liable to get out of order as the woad (p. 145) vat, and in consequence is much more easily managed. It does not, however, give such bright shades as either of the vats previously described, but it dyes a little quicker, and deeper shades can be produced. It is the best vat to use where indigo dyeing is carried on at irregular intervals, also for dyeing dark shades of navy blue and for giving an indigo bottom for dark blues, browns and greens. Such shades stand milling and alkalies very well.
Soda-Indigo Vat.--The soda-indigo vat is set in the following manner: 100 lb. bran is boiled with 200 gallons of water for three hours, then the liquor is allowed to cool from 100° to 120° F. Then 20 lb. of soda crystals, 5 lb. slaked lime, and 10 to 15 lb. ground indigo are added, the mixture being left for two or three days to ferment, and stirred up at intervals.
Sometimes a little more soda or a little lime is added, as may be judged from the appearance of the vat, these appearances being practically the same as those met with in the woad vat, which have already been described in detail.
The soda vat closely resembles the potash vat, but is cheaper to produce. It keeps its dyeing power longer, but is somewhat more liable to get out of order. It is like the potash vat, easier to manage than the woad vat, as with all the woad vats it is necessary after working them for a day to replenish them with a little indigo, soda, or potash, as the case may be, and a little bran.
Cleaner vats are obtained if treacle be substituted for the bran, but the latter ferments better, and gives better results in working.
Urine-Indigo Vat.--This vat has almost, if not quite, gone out of use, being a rather unpleasant vat to work with, with few advantages over other vats. One advantage it possesses over the woad and potash vats is that it is the best for working on a small scale, but the modern zinc reduction vats run it (p. 146) very close in this respect. The vat is made as follows: To 50 gallons of stale urine 4 lb. of common salt are added, and the mixture heated to from 120° F. to 140° F. Then 1 lb. madder and 1 lb. ground indigo are added, and the mass is well stirred. Then the mixture is allowed to stand until the indigo is completely reduced, when the vat is ready for dyeing.
Indigo-Indophenol Vat.--Messrs. Durand, Huguenin & Co. have introduced the use of Indophenol along with indigo in wool dyeing. Indophenol can be reduced in the same way as indigo, and fibres dipped in this reduced product on exposure to air turn blue in the same way as if dipped in an indigo vat.
By itself indophenol has not met with much favour from dyers for a variety of reasons, but it has been found that, mixed with indigo, it can be used in dyeing with some advantage on the score of cheapness. The newly mixed vat is made in the following manner:--
In a convenient vessel 26 gallons of water, 15 lb. zinc dust, ground into a paste with 6 gallons of water, and 7 gallons bisulphite of soda of 55° Tw. strong are mixed. Then 8 pints caustic soda lye of 72° Tw., and 16 pints liquor ammonia are added, and the whole mass is well stirred up; 22 lb. good indigo of about 70 per cent. indigotine and 7-1/4 lb. Indophenol are thoroughly ground into a paste with 7 gallons of water and 2 pints caustic soda lye of 72° Tw. The paste is added to the previous mixture, and, after being well stirred in, sufficient water is added to make the total volume of liquor up to 100 gallons. The mass is stirred up from time to time during a period of from thirty-six to forty-eight hours, by which time, as a rule, the indigo and Indophenol will have been completely reduced, and the vat have acquired a canary-yellow colour; if it has not, add a little more zinc dust and bisulphite of soda. It is ready for use when it has a good yellow colour.
This (p. 147) forms what may be called a "mother," or stock vat, from which the dyeing vat is made in the following manner: Take a sufficient quantity of water to make the dyeing vat, add some hydrosulphite of soda (see below) to destroy any oxidising action the vat liquor may have, then add sufficient of the stock vat to give the required shade, this point is one which must be determined by experience. The vat is now quite ready for use, and the wool is entered and treated in the usual manner.
After dyeing each lot of wool it is advisable to add some of the stock vat to replace the indigo abstracted by the goods. When a number of dyeings have been done, it is possible that the vat may become charged with oxidised indigo and lose its clean, yellow colour. It may be restored to its former conditions by adding some hydrosulphite of soda. Of course, after considerable use this, like all other indigo vats, becomes too highly charged with sediment, etc., to give excellent results, in which case the only thing that can be done is to throw the old vat away and start a new one.
The hydrosulphite of soda referred to above is made in the following way: 4-1/2 lb. zinc dust are ground into a paste with 5-1/2 gallons of water and then mixed with 4 gallons bisulphite of soda at 55° Tw., stirring well so as to keep the temperature down. Then add 3 pints caustic soda lye of 72° Tw., and 3-1/2 pints liquor ammonia. Finally, add sufficient water to make 13 gallons. After standing for two or three days the preparation is ready for use. It should be alkaline in property; if not, add a little ammonia to make it so. This vat gives very good bright shades, from sky blue to dark navy, which are equally as fast as pure indigo shades.
Sometimes woollen goods dyed with indigo rub badly. The causes of this defect vary from time to time, and in many instances are often obscure in their origin. All goods intended for indigo dyeing, and more especially when shades fast (p. 148) to rubbing are desired, should be thoroughly cleansed, and before passing into the indigo vat should be thoroughly freed from any soap which may have been used in the boiling out. Then, after dyeing, they ought to be well rinsed in water and passed through a sour made with sulphuric acid (2 lb. in 10 gallons), and then washed again. Vats highly charged with sedimentary matter, or with zinc or lime, are often the cause of loose shades. The remedy is obvious, viz., the discarding of such vats and the preparation of new ones, in fact old vats are perhaps more fruitful sources of loose shades than any other cause. Soft water suits indigo dyeing better than hard water, and is to be preferred.
It is not advisable to attempt to get full or deep shades of indigo at one dip, for such would necessitate the use of strong baths. Dyeings produced in this way are liable to rub badly, because the indigo lies mostly on the surface, to which it is more or less mechanically attached. Light shades of indigo are fast to rubbing, and by repeated dippings in a light vat or a medium shade vat deep shades of fair fastness to rubbing can be got.
As repeatedly stated, no indigo vat can be worked continuously with good results; the continual agitation induced by the passage of the yarns or cloths into the liquor brings the liquor into contact with the air, and oxidation sets in, resulting in the indigo being thrown out of the liquor in its original form. When this happens the vat loses its original clear yellow or yellowish-brown colour and becomes greenish, a sure sign that the vat is getting out of condition to give good results. The remedy has been pointed out in dealing with each kind of vat, and consists essentially in adding to the vat more of the active reducing agent and allowing the vat to rest a while.
The dye-vats may be either round tubs or square wooden tanks; for yarn in hanks, when cloths or warps are being dyed, (p. 149) these may be fitted with winces and guide rollers so as to draw materials through the liquor.
The hawking machine shown in figure 22 is also very good for indigo cloth dyeing, and is largely used for this purpose.
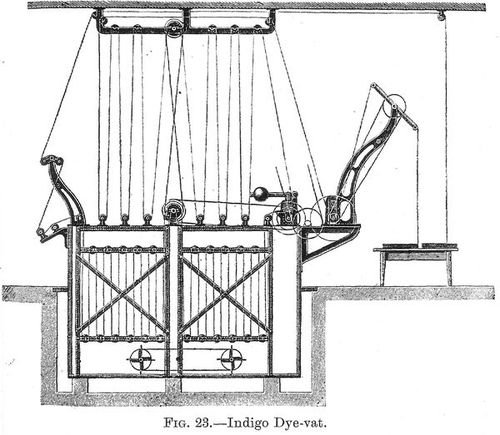
Figure 23 also shows an excellent machine for indigo dyeing on cloth. In this the vat has a frame carrying guide rollers, round which the cloth passes, so that it travels several times through the vat liquor in its passage from one end of the vat to the other, the amount of liquor in the vat being so arranged that the cloth is entirely immersed the whole time. After going through the liquor the cloth passes between a pair of squeezing rollers, in order to have any surplus liquor taken out, then it traverses the space between sets of guide rollers arranged over the vat, during which time the indigo becomes (p. 150) oxidised and the blue develops, while finally it is plaited down on a table. The illustration clearly shows the working of the machine.
Dyeing Wool with Indigo Extract.--Sulphonated indigo, prepared by dissolving indigo in sulphuric acid, is sold under the name of "indigo extract," or "indigo carmine," in two forms--paste (containing, perhaps, 25 to 30 per cent. actual colour) and powder. Both forms are freely soluble in water, although some makes are more so than others. This quality of solubility is dependent upon the proportion of sulphuric acid which may have been used in the preparation of the extract. When this is small, what is termed indigo monosulphonic acid only is formed, which is but slightly soluble in water, and gives red shades. If a larger proportion of acid be used, then the indigo disulphonic acid is formed, which is fairly easily soluble in water, and gives bluer shades than the former.
As all forms of indigo extract are regular articles of commerce, details for their preparation will not be given here. It will suffice to say that indigo is heated with strong sulphuric acid until test samples show that the indigo has been completely dissolved, and it is then diluted with water and filtered. Sometimes it is sold in this condition under the term "chemic," but if this be used in dyeing wool it gives rather unsatisfactory results. When "sour extract" is required, the liquor filtered out is next treated with salt until all the colour has been precipitated out, when it is filtered off, drained, pressed and sold. Should "neutral" or "sweet" extract be required, then the acid liquor is neutralised with soda, and the product is salted out as before, drained and pressed to a suitable consistence. It is then sold as "indigo extract," or dried, at 150° F., to a powder, which is known as "indigo carmine".
All forms of indigo extract are dyed on wool from baths of (p. 151) Glauber's salt and sulphuric acid, and therefore they can be classed with the acid-dyeing coal-tar colours. Indigo extract is notable for its level dyeing and penetrative properties, but it is not fast to light or milling.
Messrs. Read Holliday & Sons have a powder form of indigo extract which will be found very useful and to give better shades than the usual run of paste extract, while it only takes about one-fifth the quantity to give a similar shade. Working at the boil should be avoided with indigo extract, as tending to make the shades greenish in tone; from 170° to 180° F. will usually be found hot enough to dye good shades.
Indigo extract is not much used by itself in dyeing blues on wool, but it is extensively employed along with other dye-stuffs to produce an immense variety of shades--drabs, greens, fawns, greys, lilacs, etc., of which some examples will be given later on.
Indigo Blue.--Prepare a bath with 10 lb. indigo extract, 5 lb. sulphuric acid, and 10 lb. Glauber's salt. Work just under the boil to shade.
Sky Blue.--The dye-bath contains 1 lb. indigo extract, 2 lb. sulphuric acid, and 10 lb. Glauber's salt. Work at about 160° F. to shade.
Dyeing Wool Blue with Logwood.--This method of dyeing blue on wool has lost much of its importance since the introduction of the artificial dyes, but it is still employed when a blue fast to milling is wanted. Logwood gives dark navy blue shades. The process is as follows: The wool is first mordanted by boiling for one and a half hours in a bath of 3 lb. bichromate of potash and 2-1/2 lb. of tartar. The operation must be so carried out that the non-oxidising green chrome mordant is developed on the fibre, and therefore the boiling must be thorough. In place of tartar, argols and oxalic acid are frequently used, while lactic acid or lignorosine might (p. 152) be employed. The dyeing is done in a bath of 20 to 25 lb. logwood, or 5 to 8 lb. logwood extract; the bath is started cold, heated slowly to the boil, and kept at that heat for one to one and a half hours. Between the mordanting and dyeing the wool should be well rinsed.
Dyeing Blue with Coal-tar Dyes.
The blue dyes derived from coal tar are very numerous, direct, basic, acid and mordant blues being known. The direct and basic dyes are very little used, but the acid and mordant dyes are extensively employed, as is indicated in the following recipes.
Dyeing with Direct Dyes. Pale Blue.--Prepare a dye-bath with 1/2 lb. Sulphon Cyanine and 10 lb. Glauber's salt. Enter the goods, and work at the boil for one hour, then lift, wash and dry.
Black Blue.--Prepare a dye-bath with 3 lb. Sulphon Cyanine, 5 lb. Glauber's salt, and 5 lb. acetate of ammonia; work at the boil for one hour. Sulphon cyanine works well with other dye-stuffs, and gives shades which are fast to milling.
Dyeing with Acid Dyes. Bright Blue.--Prepare a bath with 2 lb. borax and 1 lb. Alkali Blue B. Enter the wool at about 170° F., then heat to the boil, and work for half an hour; then lift, rinse lightly, and pass into a weak sour bath, with sulphuric acid to raise to the colour.
Soda may be used in place of borax, but the latter salt maintains the softness of the wool fibre better.
By using various brands of Alkali Blue (3 R to 7 B), various shades of blue from a reddish with the 3 R to a pure blue with the 6 B and 7 B brands may be dyed. The Alkali Blues are fairly fast to light.
Dark Blue.--Prepare a dye-bath with 2 lb. Serge Blue, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil (p. 153) for one hour. This is a very common way of dyeing blues on serges, cashmeres and worsted goods. In place of serge blue, what are known as Blackley blues, or Dewsbury blues, may be employed. These have a similar composition, but vary a little in the tint of blue they give.
Navy Blue.--Prepare a dye-bath with 2 lb. Induline A, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil for one hour.
The Indulines are very useful colouring matters for dyeing navy or dark blues on wool. They have the defect of being liable to give uneven shades. This may be remedied by omitting the acid when first making up the bath, entering the wool, working for half an hour to thoroughly impregnate the material with the dye-liquor, then adding the acid, and continuing the working for another half-hour. Or the wool may be treated to a weak chlorine bath before it is dyed, by first passing it through a weak hydrochloric acid bath and then through a bath of bleaching powder. By using acetic acid in place of sulphuric acid more even shades are obtained.
Blue.--Prepare a dye-bath with 1 lb. Acid Blue 1 V, 9 oz. Acid Violet 1 V, 10 lb. Glauber's salt and 2 lb. sulphuric acid, working at the boil for one hour.
Blue Black.--For this the dye-bath is made with 8 lb. Acid Blue 1 V, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil for one hour.
Deep Navy Blue.--A very good shade is dyed with 5 lb. Acid Blue 1 V, 3 lb. Acid violet 1 V, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil for one hour.
Deep Navy.--Prepare a dye-bath with 1 lb. Fast acid Magenta B, 3 lb. Wool Blue B X, 4-3/4 oz. Orange I I, 5 lb. sulphuric acid, and 10 lb. Glauber's salt, working at the boil for one hour.
The Patent Blues work exceedingly well on wool, giving good bright shades of a fair degree of fastness. The following recipes (p. 154) will give some idea of the nature of the shades which may be obtained from them, while later on their use in combination with other dyes for the production of compound shades will be shown.
Bright Blue.--Prepare a dye-bath with 2 lb. Patent Blue N, or Patent Blue superior, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil for one hour.
Bright Greenish Blue.--Use 2 lb. Patent Blue V, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Royal Blue.--Use 2 lb. Patent Blue B, or 2 lb. Patent Blue J (No. 3), 10 lb. Glauber's salt, and 2 lb. sulphuric acid. Patent Blue J (No. 3) gives slightly more violet shades than Patent Blue N, but there is not much difference between them.
Saxony Blue.--Use 2 lb. Patent Blue J (No. 00), 2 lb. sulphuric acid, and 10 lb. Glauber's salt. Patent Blue J (No. 00) dyes shades very closely resembling those dyed with indigo extract, and where the latter is used in the dyeing of compound shades the former might be substituted.
Brilliant Royal Blue.--Prepare a bath with 1-1/2 lb. New Victoria Blue B, and 10 lb. Glauber's salt. Enter at about 100° F., then raise to the boil and work for one hour. This gives a very brilliant shade of blue of a violet tone.
Sky Blue.--Prepare a dye-bath with 1-1/2 oz. New Victoria Blue B and 2 lb. Glauber's salt, working in the manner described in the last recipe.
Dark Blue.--Prepare a dye-bath with 1-1/2 oz. Acid Violet 5 B, and 1-1/2 lb. Fast Green Bluish, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil to shade; then lift, wash and dry.
Deep Blue.--Make a dye-bath with 4 lb. Chromotrop 6 B, 10 lb. Glauber's salt, and 4 lb. acetic acid. Work for one hour at the boil; then lift, add 2 lb. bichromate of potash and 3 lb. acetic acid, re-enter the goods and work for one hour longer; lift, wash and dry.
The (p. 155) blues produced from the Chromotrops according to the last recipe are full, solid-looking shades, and have a great degree of fastness to milling and light. Some other examples showing the production of blue shades from the Chromotrops will be given later on.
Violet Blue.--Prepare a dye-bath with 2 lb. Victoria Violet 8 B S, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil to shade; then lift, wash and dry.
Deep Blue.--A fine deep blue is dyed on wool from a bath containing 6 lb. Victoria Violet 8 B S, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil to shade.
Deep Sky Blue.--A fine shade is dyed in a bath containing 4 oz. Cyanole extra, 10 lb. Glauber's salt and 2 lb. acetic acid.
Electric Blue.--Make the dye-bath with 4 oz. Cyanole extra, 1 oz. Acid Green extra, and 10 lb. bisulphate of soda.
Bright Blue.--A very fine shade of blue can be dyed in a bath containing 3 lb. Cyanole extra and 10 lb. bisulphate of soda.
Dark Navy Blue.--Prepare the dye-bath with 4 lb. Cyanole extra, 9 oz. Archil Substitute N, and 10 lb. bisulphate of soda.
Dark Navy.--Prepare the dye-bath with 5 lb. Black Blue O, 1-3/4 oz. Formyl Violet S 4 B, 4 oz. Patent Blue V, 25 lb. Glauber's salt, and 4 lb. bisulphate of soda, adding 1 lb. sulphuric acid when the dyeing is about half done.
The navy blues given in the last few recipes possess the merit of considerable resistance to light, air and milling.
Pale Blue.--Make the dye-bath with 1/2 oz. Chromotrop 2 R, 4 oz. Cyanine B, 7-1/2 oz. Fast Acid Blue R, 1/2 oz. Azo Yellow, 10 lb. acetic acid, and 15 lb. Glauber's salt.
Peacock Blue.--A fine shade is dyed with 14 oz. Cyanine B, 1-1/2 lb. Fast Acid Blue R, 2 oz. Azo Yellow, 10 lb. acetic acid, and 15 lb. Glauber's salt.
Dark (p. 156) Invisible Blue.--Make the dye-bath with 2 lb. Victoria Black Blue, 10 lb. Glauber's salt, and 3 lb. sulphuric acid.
Bright Blue.--A very fine shade of blue, not, however, fast to light, is dyed from a bath containing 1/2 lb. Victoria Blue B, and 10 lb. Glauber's salt.
Bright Electric Blue.--Prepare a dye-bath with 3/4 lb. Glacier Blue, 10 lb. Glauber's salt and 3 lb. sulphuric acid, working at the boil. This gives a very bright green shade of blue.
Dark Peacock Blue.--Make the dye-bath with 1 lb. Naphthol Blue Black, 10 lb. Glauber's salt, and 3 lb. sulphuric acid.
Peri Wool Blues B & G dye wool in very fast dark blue shades from baths of Glauber's salt and acetic acid. They are dye-stuffs which form with copper blue colour lakes of some fastness. The copper is amalgamated with the dye-stuffs as put on the market.
Pale Navy Blue.--Mordant, 4 lb. bichromate of potash and 1-1/2 lb. oxalic acid. Dye, 2-1/2 lb. Alizarine Bordeaux B.
Navy Blue.--Mordant, 4 lb. bichromate of potash and 2 lb. oxalic acid. Dye, 7 lb. Alizarine Bordeaux G.
Bright Violet Blue.--Mordant, 3 lb. fluoride of chrome and 2 lb. oxalic acid. Dye, 3/4 lb. Celestine Blue B.
Navy Blue.--A reddish shade of navy blue is dyed by mordanting with 3 lb. fluoride of chrome and 2 lb. oxalic acid, and dyeing with 3 lb. Celestine Blue B and 3/4 lb. Diamond Black.
The Alizarine Cyanines are excellent dye-stuffs for giving dark blue and navy blue shades on wool. They dye fairly easily, and uniform shades are readily obtained, while they possess some considerable penetrative power, so that they are well adapted for dyeing heavy piece goods. The following recipes show their use and indicate the character of the shades (p. 157) the various brands yield. It may be added that the shades are fast to light and milling.
Red Navy Blue.--Mordant, 4 lb. bichromate of potash, 2 lb. tartar, and 1-1/2 oz. sulphuric acid. Dye, 6 lb. Alizarine Cyanine R R R double. By using a mordant of 4 lb. fluoride of chrome and 2 lb. oxalic acid the shade is made brighter and not so red in tone.
Dark Blue.--A red shade of blue almost approaching a navy is obtained by mordanting with bichromate of potash, as in the last recipe, and dyeing with 12 lb. Alizarine Cyanine R R, or with 13 lb. Alizarine Cyanine R. The shade with the latter dye-stuff is scarcely so red as with the former.
Dark Blue.--Mordant with 4 lb. fluoride of chrome and 2 lb. oxalic acid and dye with 13 lb. Alizarine Cyanine R.
Dark Blue.--A somewhat brighter and less red shade than is obtained by working as in the last recipe is given by mordanting with 3 lb. bichromate of potash, 2 lb. tartar, and 2-1/2 oz. sulphuric acid, and then dyeing with 17 lb. Alizarine Cyanine G extra.
Dark Blue.--Mordant with 3-1/2 lb. bichromate of potash, 2 lb. tartar, and 3 oz. sulphuric acid. Dye with 18 lb. Alizarine Cyanine G G.
Peacock Blue.--Mordant with 4 lb. fluoride of chrome and 2 lb. oxalic acid. Dye with 18 lb. Alizarine Cyanine G G.
The addition of from 2 lb. to 5 lb. acetate of ammonia in working with the Alizarine Cyanines is a considerable advantage, by causing the dye-stuff to penetrate the fibre better and to give more uniform shades.
Medium Blue.--Mordant with 3 lb. bichromate of potash and 2 lb. oxalic acid. Dye with 5 lb. Brilliant Alizarine Blue G, and 2 lb. acetic acid.
Black Blue.--Mordant as in the last. Dye with 20 lb. Brilliant Alizarine Blue G and 2 lb. acetic acid.
Dark Navy.--Mordant as in the last recipe and dye with 5 (p. 158) lb. Alizarine Cyanine 3 R double, 5 lb. Alizarine Blue G W, 2 lb. Brilliant Alizarine Blue G, and 2 lb. acetic acid.
Medium Blue.--Mordant as in the last. Dye with 5 lb. Alizarine Blue G W, 2-1/2 lb. Brilliant Alizarine Blue G, and 2 lb. acetic acid.
Lavender Blue.--Mordant with 3 lb. bichromate of potash and 2-1/4 lb. tartar. Dye with 2 lb. Alizarine Blue A.
Navy.--Mordant as in the last recipe, and dye with 20 lb. Alizarine Blue A.
Deep Sky Blue.--Mordant with 3 lb. bichromate of potash and 1 lb. oxalic acid, then dye with 2-1/2 lb. Chrome Blue.
Bright Blue.--A very fine bright shade is obtained by mordanting as in the last, and then dyeing with 10 lb. Chrome Blue.
Lilac Blue.--Mordant with 2 lb. bichromate of potash and 1-1/2 lb. tartar. Dye with 4 lb. Alizarine Blue D N W. Alizarine Blue R gives somewhat bluer shades than the D N W brand.
Slate Blue.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar. Dye with 2-1/2 lb. Alizarine Blue D N W, 4 oz. Alizarine Brown, and 1-2/3 oz. Alizarine Yellow.
Peacock Blue.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar. Dye with 6 lb. Alizarine Blue D N W, 3 lb. Alizarine Yellow, and 1-1/2 lb. Patent Blue A, adding a little acetic acid to the dye-bath.
Paris Blue.--Mordant as in the last recipe. Dye with 3 lb. Galleine, 1 lb. Alizarine Blue D N W, and 1 lb. Patent Blue A, adding a little acetic acid.
Grey Blue.--Mordant as above and dye with 4-1/2 lb. Alizarine Blue D N W, and 1 lb. Alizarine Brown.
Blue.--Mordant with 10 lb. alum, 3 lb. tartar, and 2 lb. oxalic acid. Dye with 15 lb. Anthracene Blue W G, 3 lb. acetate of lime, and 1 lb. tannic acid.
Red Navy.--Mordant as in the last recipe and dye with 15 (p. 159) lb. Anthracene Blue B W, 3 lb. acetate of lime, and 3/4 lb. tannic acid.
Dark Blue.--Mordant with 1 lb. bichromate of potash and 2 lb. tartar. Then dye with 20 lb. Anthracene Blue W B. Anthracene Blue W G gives slightly greener shades than the W B brand, while the W R blue gives redder shades.
Grounding wool with various tints of indigo is a favourite method of producing many useful shades on wool. In general it is a good plan, as the bottom so given is a fast and permanent one, and is not in any way affected (so far as the stability of the colour is concerned) by the subsequent dyeing operations, care of course being taken that these are the usual acid or mordanting baths. The only drawback against bottoming with indigo is the increased cost of dyeing necessitated by the extra labour, and materials required to dye the bottom. As to the methods and materials required, they are just those usually employed in indigo dyeing, and these have been described. The hydrosulphite vat, or Messrs. Holliday's patent indigo, is, perhaps, the most convenient method to adopt.
Dark Slate.--Give a medium indigo bottom, then mordant with 3 lb. fluoride of chrome and 1 lb. oxalic acid, and dye with 1-1/2 lb. Anthracene Brown W, 1/2 lb. Alizarine Bordeaux G, and 1 oz. Diamond Flavine.
Dark Navy.--Give a medium indigo bottom in the vat, then mordant with 3 lb. fluoride of chrome and 1-1/2 lb. tartar, finally dyeing with 6-1/2 lb. Alizarine Cyanine G, and 1-1/2 lb. Alizarine Bordeaux G.
Dark Blue.--Give a medium indigo bottom, then mordant with 6 lb. fluoride of chrome and 2 lb. oxalic acid, finally dyeing with 14 lb. Alizarine Cyanine Black.
Blue Black.--Give a deep indigo bottom in the vat, then mordant with 3 lb. bichromate of potash and 2 lb. tartar, finally (p. 160) dyeing with 6 lb. Alizarine Cyanine Black and 1-1/2 lb. Alizarine Cyanine 3 R double.
Violet Shades on Wool.
Violet shades can only be obtained from the coal-tar colours, and of these there are not many. The recipes which are given below will serve to show what dye-stuffs are available, and will give some idea of the tints they dye.
With Direct Dyes. Pale Violet.--Prepare the dye-bath with 1/2 lb. Sulphon Cyanine, 1/4 lb. Geranine B, 5 lb. Glauber's salt, and 5 lb. acetate of ammonia, working at the boil for one hour.
With Basic Dyes. Violet.--The dye-bath is made with 1 lb. Methyl Violet 3 B, and 10 lb. Glauber's salt. A fine pure shade of violet is obtained. Methyl Violet is made in many brands, distinguished as B, B B, 2 B, 4 B, etc. By using either one or the other of these, a variety of tints of violet, from a red shade with Methyl Violet R through violet (B) to a violet blue with Methyl Violet 7 B, can be dyed.
Puce.--A very bright shade of puce is dyed by using Methyl Violet R, and 10 lb. Glauber's salt.
With Acid Dyes. Violet.--Make the dye-bath with 2 lb. Acid Violet 4 B S, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. This gives a pure violet shade. If Acid Violet 6 B S be used a bluer shade is obtained.
Reddish Puce.--A very bright red tint of puce is obtained by using 2 lb. Acid Violet 4 R S, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Bluish Violet.--Make the dye-bath with 3 lb. Acid Violet 5 B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil for one hour.
Lavender.--Use 4 oz. Acid Violet 5 B, 1 oz. Azo Fuchsine G, 1/16 oz. Fast Green bluish, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Deep Violet.--A fine deep shade is obtained by using 2-3/4 lb. Chromotrop (p. 161) 6 R, 2-1/2 lb. Cyanine B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil for one hour.
Mauve.--Use 2 lb. Acid Mauve B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Bright Violet.--Use 2 lb. Formyl Violet S 4 B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Bright Violet.--Use 2 lb. Acid Violet 6 B N, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Violet.--Use 2 lb. Acid Violet N, 2 lb. sulphuric acid, and 10 lb. Glauber's salt.
With Mordant Dyes. Violet.--Mordant the wool with 3 lb. bichromate of potash and 2 lb. tartar, and dye with 10 lb. Chrome Violet.
Dark Violet.--Mordant as in the last recipe. Then dye with 3 lb. Chrome Bordeaux 6 B double and 2 lb. Brilliant Alizarine blue G.
Brown Shades on Wool.
Brown is a very important colour, of which there is an infinite variety of shades and it can be dyed in a great variety of ways and from a variety of dye-stuffs, as will be seen on looking through the recipes which follow, although these do not by any means exhaust the methods by which browns may be dyed on woollen goods, but they may be taken as representative and will serve to show by what combinations of dyes various tints of browns may be obtained.
With Direct Dyes. Brown.--Make the dye-bath with 1 lb. Nyanza Black B, 2 lb. Congo Brown R, and 20 lb. Glauber's salt, working at the boil for one hour; then lift, wash and dry.
With Acid Dyes. Yellow Brown.--Make the dye-bath with 1 lb. Azo Carmine, 1 lb. Fast Yellow, 1 lb. Indigo Carmine D, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. A good shade is thus obtained.
Olive (p. 162) Brown.--Use 3/4 lb. Azo Acid Violet 4 R, 2 lb. Fast Yellow, 3 oz. Fast Green bluish, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil for one hour; then lift, wash and dry.
Dark Chestnut.--Dye in a bath containing 6-1/2 oz. Patent Blue V, 3-1/4 oz. Acid Violet V, 1 lb. Azo Yellow, 2 lb. Orange No. 2, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil for one hour; then lift, wash and dry.
Mouse.--Make the dye-bath with 4 oz. Patent Blue V, 1-2/3 oz. Acid Violet N, 13 oz. Orange G, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Deep Seal.--Dye in a bath containing 1 lb. Orange G G, 1/2 lb. Patent Blue J 3, 1/2 lb. Azo Yellow, 3-1/4 oz. Acid Violet N, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Deep Brown.--Make the dye-bath with 1-3/4 lb. Chromotrop 2 R, 1-1/4 lb. Victoria Yellow, 4 lb. Keton Blue G, 2-1/2 oz. Acid Violet 5 B E, 25 lb. Glauber's salt, and 4 lb. sulphuric acid, working at the boil for one hour.
Walnut.--A fine shade can be dyed with 1-3/4 lb. Azo Acid Magenta G, 14-1/2 oz. Patent Blue V, 3/4 lb. Victoria Yellow, 15 lb. Glauber's salt and 2 lb. sulphuric acid.
Olive Brown.--Make a dye-bath with 2 lb. sulphuric acid, 10 lb. Glauber's salt, 1 lb. Azo Fuchsine G, 1/2 lb. Fast Yellow, and 1/2 lb. Fast Green extra bluish.
Dark Olive Brown.--A very fine shade can be dyed with 1 lb. Fast Acid Violet 10 B, 1-1/2 lb. Orange 11, 1/2 lb. Fast Green bluish, 7 oz. Fast Yellow, 20 lb. Glauber's salt, and 3 lb. sulphuric acid.
Walnut.--Use 1 lb. Cyanole, 1 lb. Orange extra, 1/2 lb. Archil Substitute N, 10 lb. Glauber's salt and 2 lb. sulphuric acid, working at the boil for one hour.
Dark Seal.--Use 1 lb. Cyanole, 1 lb. Orange extra, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Golden Brown.--A fine shade is dyed with 1-1/4 lb. Victoria Yellow, (p. 163) 9-1/2 oz. Chromotrop 2 R, 3-1/2 oz. Patent Blue V, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
With Mordant Dyes. Golden Brown.--Make the dye-bath with 1 lb. Diamine Fast Red F, 1-1/2 lb. Anthracene Yellow C, and 5 lb. acetate of ammonia. Work for half an hour; then add 5 lb. bisulphate of soda and work for half an hour longer, then add 3 lb. fluoride of chrome, and work for half an hour at the boil.
Bright Golden Brown.--Use 3/4 lb. Diamine Fast Red F, 1-1/2 lb. Anthracene Yellow C, 5 lb. bisulphate of soda, as indicated in the last recipe. The shades so obtained are very fine, and have the merit of being fast to washing and light.
Chestnut.--Give a medium indigo bottom in the vat, then dye in a bath containing 1-3/4 lb. Anthracene Yellow C, 1 lb. Diamine Fast Red F, and 5 lb. bisulphate of soda. Work again for half an hour, then add 3 lb. fluoride of chrome, and work again for another half hour; lift, wash and dry.
Dark Brown.--Use a dye-bath containing 1-1/4 lb. Diamine Fast Red F, 3/4 lb. Anthracene Yellow C, 1-1/2 lb. Anthracite Black B, and 5 lb. acetate of ammonia. After half an hour's boiling, add 5 lb. bisulphate of soda, work half an hour longer, add 3 lb. fluoride of chrome, and work together another half hour; then lift, wash and dry.
Brown.--A very fine shade can be dyed in the following way: First give a medium indigo bottom in the vat, then mordant in a bath containing 3 lb. bichromate of potash and 2-1/2 lb. tartar, and finally dye in a bath made from 1-1/2 lb. Alizarine Orange R, 4 lb. Diamond Flavine, and 2 lb. acetic acid.
Dark Seal.--Give a medium indigo bottom in the vat, and Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar, and finally dye in a bath containing 3-1/2 lb. Alizarine Orange R, 1 lb. Anthracene Brown R, 2 lb. Diamond Flavine, and 2 lb. acetic acid.
Brown.--A (p. 164) full shade is dyed by first mordanting with 3 lb. bichromate of potash and 2 lb. tartar, and then dyeing with 10 lb. Anthracene Brown W, and 1 lb. Mordant Yellow.
Buff.--Mordant as in the last, and dye with 5 lb. Anthracene Brown W, and 1/4 lb. Mordant Yellow O.
Nut.--Mordant with 3 lb. bichromate of potash and 1 lb. oxalic acid, and dye with 20 lb. Diamond Brown.
Pale Old Gold Brown.--Mordant as in the last, and dye with 5 lb. Diamond Brown.
Dark Violet Brown.--Mordant as in the last recipes, and dye with 30 lb. Chrome Brown R.
Bright Chestnut.--Mordant with 3 lb. bichromate of potash and 1 lb. sulphuric acid, and dye with 30 lb. Gambine R.
Pale Chestnut.--Mordant as in the last recipes, and dye with 20 lb. Gambine Y.
Olive Brown.--Mordant as in the last recipes, and dye with 10 lb. Gambine B. The browns dyed with Gambine have the merit of being fast to milling and light.
Dark Brown.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar; then dye with 15 lb. Alizarine Brown.
Bright Buff.--Mordant as in the last recipe; then dye with 4-3/4 lb. Alizarine Brown, 4 lb. Alizarine Yellow, 1-3/4 oz. Alizarine Blue D N W, and 2 lb. acetic acid.
Dark Violet Brown.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar. Then dye with 18 lb. Alizarine Brown, 6 lb. Alizarine Orange H, and 2 lb. acetic acid.
Dark Walnut.--Mordant with 3 lb. bichromate of potash and 1 lb. sulphuric acid; then dye with 8 lb. Alizarine Brown, 2 lb. Alizarine Red 3 W S, and 2 lb. Alizarine Yellow G G W.
Mode Colours on Wool.
Under the general designation of "mode colours" are included a great variety of tints or shades unusually described more (p. 165) specifically as drabs, buffs, greys, fawns, slates, etc. It is impossible here to do more than give a few recipes for their production.
With Direct Dyes. Drab.--Make a dye-bath with 3 oz. Nyanza Black B, 1-1/2 oz. Chrysamine G, 2 oz. Congo orange R, and 20 lb. Glauber's salt, working at the boil for one hour; then lift, wash and dry.
With Acid Dyes. Bright Buff.--Dye in a bath containing 3/4 oz. each Cyanole, Orange extra, and Indian Yellow R, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Slate.--Use a dye-bath containing 3 oz. Cyanole, 1/4 oz. Archil Substitute N, 1/2 oz. Orange extra, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Silver Grey.--Use 1-1/4 oz. Orange extra, 3/4 oz. Archil Substitute N, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Pale Drab.--Make the dye-bath with 1/2 oz. Cyanine B, 3/4 oz. Azo Yellow, 1/4 oz. Chromotrop 2 R, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Grey.--Make the dye-bath with 1 oz. Chromotrop 2 R, 1-1/4 oz. Cyanine B, 2-1/2 oz. Fast Acid Blue R, 2 oz. Azo Yellow, 10 lb. Glauber's salt and 5 lb. acetic acid.
Bright Fawn.--The dye-bath is made with 2 oz. Chromotrop 2 R, 8 oz. Orange G, 2-1/4 oz. Fast Acid Blue R, 1-1/4 oz. Cyanine B, 10 lb. Glauber's salt and 5 lb. acetic acid.
Dark Buff.--Use 2 oz. Cyanine B, 5 oz. Azo Yellow, 2-1/2 oz. Chromotrop 2 R, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Lilac Grey.--Use 3 oz. each Fast Acid Violet 10 B, Fast Green bluish, and Fast Yellow, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Pale Fawn Drab.--Use 1 oz. Patent Blue V, 1 oz. Rhodamine, 1-3/4 oz. Orange G, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Dark (p. 166) Grey.--Use 1 lb. Wool Grey R, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Stone.--Use 1 oz. Patent Blue J B, 1-3/4 oz. Orange G, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Pale Fawn Brown.--Use 4 oz. Fast Acid Violet R, 2 oz. Patent Blue J O O, 3 oz. Orange G, 10 lb. Glauber's salt and 3 lb. sulphuric acid.
Drab.--Use 3 oz. Azo Carmine, 1-1/2 oz. Fast Yellow, 1-1/4 oz. Indigo Carmine D, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Lilac.--Use 1/2 lb. Azo carmine, 1/2 lb. Indigo Carmine D, 1-1/2 oz. Fast Yellow, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
With Mordant Dyes. Pale Drab.--Mordant with 2 lb. bichromate of potash and 1-1/2 lb. tartar. Dye with 1 lb. Alizarine Brown paste.
Violet Grey.--Mordant as in the last recipe, and dye with 1 lb. Alizarine Grey B.
Pale Fawn.--Mordant with 3 lb. bichromate of potash and 2-1/2 lb. tartar, and dye with 4-1/2 lb. Alizarine Yellow, 13 oz. Alizarine Brown, 11-1/2 oz. Alizarine Orange N, and 2 lb. acetic acid.
Pale Stone.--Mordant with 2 lb. bichromate of potash and 1-1/2 lb. tartar. Dye with 13 oz. Alizarine Yellow and 1-1/4 lb. Alizarine Brown.
Dark Slate.--Mordant with 3 lb. bichromate of potash and 2 lb. tartar. Dye with 2-1/2 lb. Alizarine Blue D N W, and 10 oz. Alizarine Yellow.
Lavender Grey.--Mordant with 2 lb. bichromate of potash and 1-1/2 lb. tartar. Dye with 13 oz. Alizarine Blue D N W, and 2 oz. Galleine.
Drab.--Mordant as in the last recipe; then dye with 4 oz. Alizarine Blue, 1-1/2 lb. Alizarine Yellow and 14 oz. Alizarine Brown.
Drab.--Mordant (p. 167) with 3 lb. bichromate of potash and 1 lb. sulphuric acid, and dye with 1 lb. Gambine R.
Dark Grey.--Give a light indigo bottom in the vat, and then dye in a bath containing 3/4 oz. Diamine Fast Red F, 3/4 oz. Anthracene Yellow C, and 5 lb. acetate of ammonia. Work at the boil for half an hour, then add 5 lb. bisulphate of soda, work half an hour longer, then add 1 lb. fluoride of chrome, and work for another half hour at the boil; then lift, wash and dry.
There is now produced a great variety of textile fabrics of every conceivable texture by combining the two fibres, cotton and wool, in a number of ways. The variety of these fabrics has of late years considerably increased, which increase may be largely ascribed to the introduction of the direct dyeing colouring matters--the Diamine dyes, the Benzo dyes, the Congo and the Zambesi dyes; for in the dyeing of wool-cotton fabrics they have made a revolution. The dyer of union fabrics, that is fabrics composed of wool and cotton, was formerly put to great straits to obtain uniform shades on the fabrics supplied to him owing to the difference in the affinity of the fibres for the dye-stuffs then known. Now the direct dyes afford him a means of easily dyeing a piece of cotton-wool cloth in any colour of a uniform shade, while the production of two-coloured effects is much more under his control, and has led to the increased production of figured dress fabrics with the ground in one fibre (wool) and colour, and the design in another fibre (cotton) and colour. The number of direct dyes issued by the various colour manufacturers is so great that it would take a fairly considerable space to discuss them all.
To obtain good results it is needful that the dyer of union fabrics should be a man of keen observation and have a thorough knowledge of the dyes he is using, for each dye makes a rule to itself as regards its power of dyeing wool and (p. 169) cotton; some go better on to the cotton than on to the wool, and vice versa. Some dye wool best at the boil, others equally well below that heat; some go on the cotton at a moderate temperature, others require the dye-bath to be boiling; some will go to the cotton only and appear to ignore the wool.
The presence or absence in the dye-bath of such bodies as carbonate of soda, Glauber's salt, etc., has a material influence on the degree of the affinity of the dye-stuff for the two fibres, as will perhaps be noted hereafter. Again, while some of the dyes produce equal colours on both fibres, there are others where the tone is different. With all these peculiarities of the Diamine and other direct dyes the union dyer must make himself familiar. These dyes are used in neutral baths, that is, along with the dye-stuff. It is often convenient to use along with the direct dyes some azo or acid dyes which have the property of dyeing the wool from neutral baths; many examples of such will be found in the practical recipes given below. The dyes now under consideration may be conveniently classed into five groups.
(1) Those dyes which dye the cotton and wool from the same bath to the same shade, or nearly so.--Among such are Thioflavine S, Diamine Fast Yellow B, Diamine Orange B, Diamine Rose B D, Diamine Reds 4 B, 5 B, 6 B and 10 B, Diamine Fast Red F, Diamine Bordeaux B, Diamine Brown N, Diamine Brown 3 G, B and G W, Diamine Blue R W, B X, Diamine Blue G, Diamine Greens G and B, Diamine Black H W, Diamine Dark Blue B, Union Black B and S, Oxydiamine Blacks B, M, D and A, Diamine Catechine G, Union Blue B B, Oxyphenine, Chloramine Yellow, Thioflavine S, Alkali Yellow R, Chromine G, Titan Scarlet S, Mimosa, Primuline, Auroline, Congo Corinth B, Thiazol Yellow, Columbia Yellow, Oxydiamine Yellow G G, Oxydiamine Oranges G (p. 170) and R, Diamine Orange O, Oxydiamine Red S.
(2) Dyes which dye the cotton a deeper shade than the wool.--The following belong to this group. Diamine Fast Yellow A, Diamine Orange G and D, Diamine Catechine G, Diamine Catechine B, Diamine sky Blue, Diamine Blues 2 B, Diamine Blue 3 B, Diamine Blue B G, Diamine Brilliant Blue G, Diamine New Blue R, Diamine Steel Blue L, Diamine Black R O, Diamine Black B O, Diamine Black B H, and Oxydiamine Black S O O O, Diamine Nitrazol Brown G, Diamine Catechine B, Diamine Sky Blue F F, Diamine Dark Blue B, Diamine Bordeaux B, Diamine Violet N, Oxydiamine Violet B, Columbia Black B and F B, Zambesi Black B, Congo Brown G, Direct Yellow G, Direct Orange R, Clayton Yellow, Cotton Yellow, Orange T A, Benzopurpurine B, Brilliant Congo R, Chicago Blues B, 4 B and 6 B.
(3) Dyes which dye wool a deeper shade than the cotton.--The dyes in this group are not numerous. They are Diamine Gold, Diamine Scarlet B, Diamine Scarlet 3 B, Diamine Bordeaux S, Diamine Blue R W, and Diamine Green G, Diamine Red N O and B, Chicago Blue G and R R W, Brilliant Purpurine R, Diamine Scarlet B, Deltapurpurine 5 B, Chrysamine, Titan Blue, Titan Pink, Congo Oranges G and R, Erie Blue 2 G, Congo R, Brilliant Congo R, Erika B N, Benzopurpurine 4 B and 10 B, Chrysophenine, Titan Yellow, Titan Brown Y, R and O, Congo Brown G, Sulphon Azurine B, Zambesi Black D.
(4) Dyes which produce different shades on the two fibres.--Diamine Brown G and Diamine Blue 3 R, Diamine Brown V, Diamine Brown S, Diamine Nitrazol Brown B, Diamine Blue B X and 3 R, Diamine Blue Black E, Benzo Blue Black G, Benzopurpurine 10 B, Benzo Azurine R G and 3 G, Columbia Red S B, Brilliant Azurine 5 G, Titan Marine (p. 171) Blue, Congo Corinths G and B, Azo Blue, Hessian Violet, Titan Blue, Azo Mauve, Congo Brown, Diamine Bronze G, Zambesi Browns G and 2 G, Zambesi Black F.
(5) Azo acid dyes which dye wool from neutral baths, and are therefore suitable for shading up the wool to the cotton in union fabric dyeing.--Among the dyes thus available may be enumerated Naphthol Blue G and E, Naphthol Blue Black, Formyl Violet 10 B, Lanacyl Blue B B, Lanacyl Blue R, Alkaline Blue, Formyl Violet S 4 B and 6 B, Rocceleine, Azo Red A, Croceine A Z, Brilliant Scarlet, Orange extra, Orange E N Z, Indian Yellow G, Indian Yellow R, Tropæoline O O, Naphthylamine Black 4 B, and Naphthol Blue Black, Brilliant Scarlet G, Lanacyl Violet B, Brilliant Milling Green B, Thiocarmine R, Formyl Blue B, Naphthylamine Blacks D, 4 B and 6 B, Azo Acid Yellow, Curcumine Extra, Mandarine G, Ponceau 3 R B, Acid Violet 6 B, Guinea Violet 4 B, Guinea Green B, Wool Black 6 B.
Regarding the best methods of dyeing, that in neutral baths yields the most satisfactory results in practical working. It is done in a boiling hot or in a slightly boiling bath with the addition of 6-1/4 oz. crystallised Glauber's salt per gallon water for the first bath, and when the baths are kept standing 20 per cent. crystallised Glauber's salt reckoned upon the weight of the goods for each succeeding lot.
In dyeing unions, the dye-baths must be as concentrated as possible and must not contain more than from 25 to 30 as much water as the goods weigh. In this respect it serve as a guide that concentrated baths are best used dyeing dark shades while light shades can be dyed in more dilute baths. The most important factor for producing uniform dyeings is the appropriate regulation of the temperature of the dye-bath. Concerning this the dyer must bear in mind that the direct colours possess a greater affinity for cotton if dyed below the boiling-point, and only go on the (p. 172) wool when the bath is boiling, especially so the longer and more intensely the goods are boiled.
The following method of dyeing is perhaps the best one. Charge the dye-bath with the requisite dye-stuff and Glauber's salt, boil up, shut off the steam, enter the goods and let run for half an hour, without steam, then sample. If the shade of both cotton and wool is too light, add some more of the dye-stuffs used for both fibres, boil up once more, and boil for a quarter to half an hour. If the wool only is too light, or its shade different from that of the cotton, add some more of the dye-stuff used for shading the wool and bring them again to the boil. If, however, the cotton turns out too light or does not correspond in shade to the wool, add some more of the dye-stuffs used for dyeing the cotton, without, however, raising the temperature. Prolonged boiling is necessary only very rarely, and generally only if the goods to be dyed are difficult to penetrate or contain qualities of wool which only with difficulty take up the dye-stuff. In such cases, in making up the bath, dye-stuffs are to be selected some of which go only on the wool and others which go only on the cotton (those belonging to the second group).
The goods can then be boiled for some time, and perfect penetration and level shades will result. If the wool takes up the dye-stuff easily (as is frequently the case with goods manufactured from shoddy) and are therefore dyed too dark a shade, then dye-stuffs have to be used which principally dye the cotton, and a too high temperature is to be avoided. In such cases it is advisable to diminish the affinity of the wool by the addition of one-fifth of the original quantity of Glauber's salt (about 3/8 oz. per gallon of water), and from three-quarters to four-fifths of the dye-stuff used for the first lot. Care has to be taken that not much of the dye-liquor is lost when taking out the dyed goods, otherwise the quantities of Glauber's salt and dye-stuff will have to be increased proportionately. Wooden (p. 173) vats such as are generally used for piece dyeing have proved the most suitable, they are heated with direct or still better with indirect steam. The method which has proved most advantageous is to let the steam run into a space separated from the vat by a perforated wall into which space the required dye-stuffs and salt are placed.
The mode of working is influenced by the character of the goods, and the following notes will be found useful by the union dyer.
Very little difficulty will be met with in dyeing such light fabrics as Italians, cashmeres, serges and similar thin textiles lightly woven from cotton warp and woollen weft. When deep shades (blacks, dark blues, browns and greens) are being dyed it is not advisable to make up the dye-bath with the whole of the dyes at once. It is much better to add these in quantities of about one-fourth at a time at intervals during the dyeing of the piece. It is found that the affinity of the wool for the dyes at the boil is so much greater than is that of the cotton that it would, if the whole of the dye were used, take up too much of the colour and then would come up too deep in shade. Never give a strong boil with such fabrics, but keep the bath just under the boil which results in the wool dyeing much more nearly like to cotton.
On Union Flannels.--In this class of goods it is important that the soft open feel of the goods be retained as much as possible, and for this purpose no class of dyes offers so many advantages as the direct colours. Only one bath being required, there is not the same amount of manipulation needed in the dyeing operation, hence there is less risk that the soft feel and woolly structure will be affected. As no mordants are needed there is nothing to impart a harsh feel to the fabrics.
On Dress Goods, Suitings and Coatings.--A large quantity of fabrics for gentlemen's suits, coats and cloths in general are (p. 174) now made from wool and cotton. Formerly the dyeing of these offered many difficulties before the application of the direct dyes was properly understood. Now, however the ease with which such dyes may be applied has given considerable impetus to this class of goods, and the trade has grown by leaps and bounds during recent years, and has been one cause of the great cheapening of clothes which has occurred in the same period. The dyeing of the goods with the direct colours offers very little difficulty, and only requires that a little attention be paid, particularly to goods in which the cotton either appears on the surface forming a design, or is spun or twisted together with the wool.
A good deal of shoddy is used in making the cheaper class of these goods, and it is quite natural that such "artificial wool" behaves differently from pure wool, not only with regard to its shade resulting from mixing and working together differently dyed waste wools, but also on account of its possessing a greater affinity for all kinds of dye-stuff than raw wool; this in consequence of the carbonisation and washing processes it has undergone, and also of the mordants which the material may retain from previous processes. Therefore (and especially in dyeing light shades on goods manufactured of shoddy) only a small quantity of soda or borax is to be added to the dye-bath and severe boiling is to be avoided. Wherever it is possible goods which are to be dyed in light shades should be made from the palest materials, and the dark qualities only used for goods which are to be dyed in dark shades.
This rule can, of course, not always be adhered to. Quite often a light and bright shade is to be dyed on comparatively dark material. This cannot be achieved by simply dyeing it, the goods must be stripped or bleached before dyeing. For this purpose either energetically reacting, oxidising reducing agents are applied. Of the former, bichromate of potassium (p. 175) is principally used. Boil the goods for half to three-quarters of an hour with 3 to 5 per cent. bichromate of potassium, 2 to 4 per cent. oxalic acid, and 3 to 5 per cent. sulphuric acid, wash in a fresh warm bath charged with soda in order to entirely neutralise the acid which has remained in the goods, or else the wool would be dyed too deep a shade. In some cases hydrosulphite has proved a useful reducing agent; it can be easily prepared from ordinary bisulphite of soda in the following manner. Add 10 oz. ammonia (0·9 specific gravity) to a gallon of bisulphite of soda, 32° Tw.; then add slowly under a brisk stirring 10 oz. zinc-dust, and let the entire mixture settle well, using only the clear solution. Treat the goods from fifteen to twenty minutes in a bath of 140° F., to which first add at the boil 3/4 oz. acetic acid, 10° Tw., per gallon water, and then 4 to 6 gallons clear hydrosulphite solution per 100 gallons liquor. Then rinse very well and dye in the usual manner; avoiding, however, too high a temperature. As on this class of goods dark shades are mostly dyed, the goods need only very rarely be stripped.
Bright Yellow.--Use 2 lb. Thioflavine S in a bath which contains 4 lb. Glauber's salt per 10 gallons of dye-liquor.
Good Yellow.--A very fine deep shade is dyed with 2-1/2 lb. Diamine Gold, and 24 lb. Diamine Fast Yellow A in the same way as the last. Here advantage is taken of the fact that while the Diamine Gold dyes the wool better than the cotton the Diamine Yellow dyes the cotton the deepest shade, and between the two a uniform shade of yellow is got.
Pale Gold Yellow.--Use a dye-liquor containing 4 lb. Glauber's salt in every 10 gallons, 2-1/2 lb. Diamine Fast Yellow A, 2 oz. Indian Yellow G, and 3-1/2 oz. Indian Yellow R. In this recipe we use in the two last dyes purely wool yellows, which dye the wool the same tint as the Fast Yellow A dyes the cotton.
Bright (p. 176) Yellow.--Use in the same way as the last 2-1/2 lb Diamine Fast Yellow B and 3 oz. Indian Yellow G.
Gold Orange.--Use as above 2 lb. Diamine orange G, 3-1/2 oz. Indian Yellow R, and 1-1/2 oz. Orange E N Z.
Deep Orange.--Use 2-1/2 lb. Diamine Orange D C, 6-1/2 oz. Orange E N Z, and 3-1/4 oz. Indian Yellow R.
Black.--Use 4-1/2 lb. Union Black S, 2 oz. Diamine Fast Yellow A, 5 oz. Naphthol Blue Black, 3-1/4 oz. Formyl Violet S 4 B, and 4 lb. Glauber's salt in 10 gallons dye-liquor.
The goods are treated at the boil in this bath for one hour, Italian cloths have frequently if not always to pass through a finishing process to give them lustre. This treatment, especially with blues and blacks, has a tendency to affect the shades, reddening them. With some dye the colour comes back on the goods becoming cold again, but with others this is not the case. If desired the goods may be subjected after dyeing to a treatment with alum or, better, bichromate of potash. The goods after being dyed are rinsed and then passed into a bath at a temperature of 140° F., containing 3 lb. bichromate of potash and 1-1/2 to 2 oz. sulphuric acid. After being chromed in this for about half an hour they are well washed. This chroming thoroughly fixes the colour on the cotton and it will not change while being finished, either by crabbing, steaming or hot pressing.
Gold Brown.--Use 1-1/2 lb. Diamine Cutch, 6-1/2 oz. Diamine Fast Yellow B, 1 oz. each Union Black, Naphthol Blue Black and Azo Red A.
Walnut Brown.--A fine shade is got with 1-1/4 lb. Union Black S, 1-1/4 lb. Diamine Brown M, 3-1/4 oz. Diamine Fast Yellow B, 13 oz. Indian Yellow G, and 1 oz. Naphthol Blue Black. After dyeing the goods should be chromed with 3 lb. bichromate of potash and 2 oz. sulphuric acid.
Dark Blue.--A good full shade is got with 2-1/4 lb. Union Black S, 9-1/2 oz. Diamine Brilliant Blue G, 6-1/2 oz. Alkaline Violet (p. 177) C A, and 1/4 lb. Alkaline Blue F. Treatment in a bath of 1/2 lb. alum and 1/2 oz. soda at 130° F. will fix the colour against finishing.
Silver Grey.--A fine grey can be got from 1-3/4 oz. Diamine Black B H, 1/2 oz. Diamine Orange B, 1/2 oz. Naphthol Blue Black, and 1/2 oz. Formyl Violet.
Navy Blue.--Use 1-1/4 lb. Union Black S, 3 lb. Diamine Black B H, 1/2 oz. Naphthol Blue Black, 1/2 lb. Formyl Violet S 4 B, and 2-1/2 oz. Alkaline Blue B.
Red Plum.--Use a dye-bath containing 2-1/2 lb. Oxydiamine Violet B and 3-1/4 oz. Formyl Violet S 4 B.
Dark Green.--A fine shade can be dyed in a bath containing 3 lb. Diamine Green B and 1-1/2 lb. Diamine Black H W.
Dark Slate.--Use 4 lb. Diamine Black H W, 2 oz. Naphthol Blue Black, and 3 oz. Azo Red A.
Sage.--Use a dye-bath containing 4 lb. Diamine Bronze G and 1-1/4 oz. Naphthol Blue Black.
Dark Brown.--A fine dark shade is got from 2-1/2 lb. Diamine Brown V, and 2 oz. Naphthol Blue Black.
Peacock Green.--Use 3-3/4 lb. Diamine Steel Blue L, 13 oz. Diamine Fast Yellow B, 14-1/2 oz. Thiocarmine R, and 2-1/4 oz. Indian Yellow G in a bath of 4 lb. Glauber's salt per gallon of dye-liquor.
Dark Sea Green.--Use 9 oz. Diamine Steel Blue L, 3-3/4 oz. Diamine Fast Yellow B, 1/2 oz. Diamine Orange G, 1-1/4 oz. Naphthol Blue Black, and 3/4 oz. Indian Yellow G.
Dark Brown.--Use 1 lb. Diamine Orange B, 1 lb. Diamine Fast Yellow B, 13-3/4 oz. Union Black S, 1 lb. Diamine Brown M, and 1/2 lb. Indian Yellow G. Fix in an alum bath after dyeing.
Dark Stone.--Use 1/2 lb. Diamine Orange B, 3-3/4 oz. Union Black, 1/4 oz. Diamine Bordeaux B, 1-1/2 oz. Azo Red A, and 3/4 oz. Naphthol Blue Black.
Black.--A very fine black can be got from 3-1/2 lb. Oxydiamine Black (p. 178) R M, 2 lb. Union Black S, 9-1/2 oz. Naphthol Blue Black and 4 oz. Formyl Violet S 4 B, chroming after dyeing as described above.
Dark Grey.--A fine bluish, shade of grey is got from 7 oz. Diamine Black B H, 2-1/4 oz. Diamine Orange G, 2-1/2 oz. Naphthol Blue Black, and 1 oz. Orange E N Z.
Dark Blue.--A fine shade is got by using 2 lb. Diamine Black B H, 1/2 lb. Diamine Black H W and 3-1/2 oz. Alkaline Blue 6 B.
Drab.--Use 3-1/2 oz. Diamine Orange B, 3/4 oz. Union Black, 1/8 oz. Diamine Bordeaux B, 3/4 oz. Azo Red A, and 1/4 oz. Naphthol Blue Black.
Plum.--Use 2-1/2 lb. Diamine Violet N, 9-1/2 oz. Union Black, and 1 lb. Formyl Violet S 4 B.
Bright Yellow.--Use a dye-bath containing 4 lb. Thioflavine S, 2 lb. Naphthol Yellow S, 10 lb. Glauber's salt, and 2 lb, acetic acid.
Pink.--Use 1/6 oz. Diamine Rose B D, 1/4 oz. Diamine Scarlet B, 1/2 oz. Rhodamine B and 20 lb. Glauber's salt.
Scarlet.--A fine shade is got from 1-1/2 lb. Diamine Scarlet B, 1/2 oz. Diamine Red 5 B and 20 lb. Glauber's salt.
Orange.--Use a dye-bath containing 3-1/2 lb. Diamine Orange G, 14-1/2 oz. Tropæoline O O, and 2-3/4 oz. Orange extra.
Sky Blue.--Use 1-1/2 oz. Diamine Sky Blue and 1-1/4 oz. Alkaline Blue B.
Bright Blue.--A fine shade similar to that formerly known as Royal Blue is got by using 1-1/2 lb. Diamine Brilliant Blue G, and 9-1/4 oz. Alkaline Blue 6 B.
Maroon.--Use 3 lb. Diamine Bordeaux B, 2 lb. Diamine Violet N, and 3-1/4 oz. Formyl Violet S 4 B.
Green.--A fine green similar in shade to that used for billiard-table cloth is got from 2 lb. Diamine Fast Yellow B, 2 lb. Diamine Steel Blue L, 14-1/2 oz. Thiocarmine R and 7-1/4 oz. Indian Yellow G.
Gold (p. 179) Brown.--A fine brown is got from 3 lb. Diamine Orange B, 1/2 lb. Union Black, 2-1/2 oz. Diamine Brown, 3/4 oz. Naphthol Blue Black, and 1/2 lb. Indian Yellow G.
Navy Blue.--Use 3-1/4 lb. Diamine Black B H, 1-1/2 lb. Diamine Brilliant Blue G, and 1/2 lb. Alkaline Blue.
Fawn Drab.--A fine shade is got by dyeing in a bath containing 6-3/4 oz. Diamine Orange B, 1-3/4 lb. Union Black, 1/4 oz. Naphthol Blue Black, 1/4 oz. Diamine Bordeaux B, and 1 oz. Azo Red A.
In all these colours the dye-baths contain Glauber's salt at the rate of 4 lb. per 10 gallons.
Dark Brown.--2-1/2 lb. Diamine Orange B, 13 oz. Diamine Bordeaux B, 1-1/2 lb. Diamine Fast Yellow B, 1-3/4 lb. Union Black, and 3-1/2 oz. Naphthol Black.
Drab.--1-3/4 lb. Diamine Fast Yellow R, 3-1/4 oz. Diamine Bordeaux B, 2-1/2 oz. Union Black, 1/2 oz. Naphthol Blue Black, and 1-1/4 oz. Indian Yellow G.
Dark Blue.--Use in the dye-bath 4-1/4 lb. Diamine Dark Blue B, 1-1/2 lb. Diamine Brilliant Blue G, 3/4 lb. Formyl Violet S 4 B, and 5 oz. Naphthol Blue Black.
Blue Black.--Use 3-1/4 lb. Union Black S, 1-1/2 lb. Oxydiamine Black B M, 6-1/2 oz. Naphthol Blue Black, and 1/4 lb. Formyl violet S 4 B.
Dark Walnut.--2-3/4 lb. Diamine Brown M, 1-1/2 lb. Union Black S, and 11-1/4 oz. Indian Yellow G.
Peacock Green.--Use in the dye-bath 3-1/2 lb. Diamine Black H W, 5-1/6 oz. Diamine Fast Yellow B, 1-1/2 lb. Thiocarmine R, and 1-1/6 oz. Indian Yellow G.
Slate Blue.--Use in the dye-bath 6-1/2 oz. Diamine Catechine B, 4-3/4 oz. Diamine Orange B, 2-1/2 oz. Union Black, 2-3/4 oz. Orange E N Z, and 1-3/4 oz. Naphthol Blue Black.
Dark Sage.--A good shade is dyed with 1 lb. Diamine Orange B, 6-1/2 oz. Union Black, 1-3/4 oz. Diamine Brown M, 3-1/4 oz. Azo Red A, and 2-1/4 oz. Naphthol Blue Black.
Navy (p. 180) Blue.--Use 2 lb. Diamine Dark Blue B, 1-1/4 lb. Lanacyl Violet B, and 7 oz. Naphthol Blue Black.
Bronze Green.--A good shade is dyed with 2 lb. Diamine Orange B, 5 oz. Diamine Brown N, 3/4 lb. Union Black S, 1 lb. Indian Yellow G, and 2 oz. Naphthol Blue Black.
Black.--Use 2-1/2 lb. Oxydiamine Black B M and 1-1/2 lb. Naphthylamine Black 6 B. Another recipe, 2-1/4 lb. Oxydiamine Black B M, 1 lb. Diamine Brown M, 1 lb. Orange E N Z, and 2 oz. Naphthol Blue Black.
Dark Brown.--Use 1-1/2 lb. Oxydiamine Black B M, 15-1/2 oz. Diamine Brown M, 1-3/4 lb. Indian Yellow G, and 2-3/4 oz. Naphthol Blue Black. Another combination, 1-1/2 lb. Oxydiamine Black B M, 1-1/2 lb. Orange E N Z, 1 lb. Indian Yellow G, and 5 oz. Naphthol Blue Black.
Scarlet.--3 lb. Benzopurpurine 4 B, 3/4 oz. Ponceau 3 R B, and 1/2 lb. Curcumine S.
Crimson.--1/2 lb. Congo Corinth G, 2 lb. Benzopurpurine 10 B, and 1/2 lb. Curcumine S.
Bright Blue.--2 lb. Chicago Blue 6 B, 3 oz. Alkali Blue 6 B, 1-1/2 oz. Zambesi Blue R X. After dyeing, rinse and develop in a bath of 8 oz. sulphuric acid in 10 gallons water, then rinse well.
Dark Blue.--2-1/2 lb. Columbia Fast Blue 2 G, 3 oz. Sulphon Azurine D, 3 oz. Alkali Blue 6 B. After dyeing, rinse and develop in a bath of 8 oz. sulphuric acid in 20 gallons of water.
Orange.--9 oz. Congo Brown G, 1-1/2 lb. Mikado Orange 4 R O, and 1-1/2 oz. Mandarine G.
Dark Green.--2 lb. Columbia Green, 1/2 lb. Sulphon Azurine D, 1/2 lb. Zambesi Blue B X, 1-1/2 oz. Curcumine S.
Black.--4 lb. Columbia Black F B, and 2 lb. Wool Black 6 B.
Pale Sage Green.--5 oz. Zambesi Black D, 3/4 lb. Chrysophenine G, and 1-1/2 lb. Curcumine S.
Slate.--1/2 (p. 181) lb. Zambesi Black D, 3/4 oz. Zambesi Blue R X, 1/2 oz. Mikado Orange 4 R O, and 1-1/2 oz. Acid Violet 6 B.
Dark Grey.--1 lb. Columbia Black F B, 3 oz. Zambesi Black B, and 3/4 oz. Sulphon Azurine D.
Drab.--1-1/2 oz. Zambesi Black D, 3/4 oz. Mandarine G extra, 1/4 oz. Curcumine extra, and 3 oz. Mikado Orange 4 R O.
Brown.--5 oz. Zambesi Black D, 3/4 oz. Mandarine G extra, 1-1/2 oz. Orange T A, and 2 oz. Mikado Orange 4 R O.
Nut Brown.--3/4 lb. Congo Brown G, 1/4 lb. Chicago Blue R W, and 3/4 lb. Mikado Orange 4 R O.
Dark Brown.--1 lb. Congo Brown G, 1-1/2 lb. Benzopurpurine 4 B, 1-1/2 lb. Zambesi Black F, and 1/2 lb. Wool Black 6 B.
Stone.--1 oz. Zambesi Black D, 1/4 oz. Mandarine G, 1/4 oz. Curcumine extra, and 1-1/4 oz. Mikado Orange 4 R O.
Slate Green.--3 oz. Zambesi Black D, 1-1/2 oz. Guinea Green B.
Sage Brown.--1/2 lb. Zambesi Black D, 1-1/2 oz. Mandarine G extra, 3 oz. Curcumine extra, 3 oz. Acid Violet 6 B, 6 oz. Mikado Orange 4 R O, and 4-1/2 oz. Curcumine S.
Cornflower Blue.--3 oz. Chicago Blue 4 R, 1/4 lb. Zambesi Blue R X, 1/4 lb. Acid Violet 6 B, and 3/4 oz. Zambesi Brown G.
Dark Brown.--1-1/2 lb. Brilliant Orange G, 1/2 lb. Orange T A, 1 lb. Columbia Black F B, and 1/4 lb. Wool Black 6 B.
Dark Blue.--2 lb. Chicago Blue R W, 1 lb. Zambesi Blue R X, 1/2 lb. Columbia Black F B, 10 oz. Guinea Green B, and 1/2 lb. Guinea Violet 4 B.
The Janus dyes may be used for the dyeing of half wool union fabrics. The best plan of working is to prepare a bath with 5 lb. of sulphate of zinc. In this the goods are worked at the boil for five minutes, then there is added the dyes (previously dissolved in water), and the working continued for a quarter of an hour; then there is added 20 lb. Glauber's salt and the working at the boil continued for one (p. 182) hour, at the end of which time the dye-bath will be fairly well exhausted of colour. The goods are now taken out and put into a fixing bath of sumac or tannin, in which they are treated for fifteen minutes. To this same bath there is next added tartar emetic and 1 lb. sulphuric acid, and the working continued for a quarter of an hour; then the bath is heated to 160° F., when the goods are lifted, rinsed and dried. In the recipes the quantities of dyes, sumac or tannin, and tartar emetic only are given, the other ingredients and processes are the same in all.
Dark Blue.--2-1/4 lb. Janus Dark Blue B, and 1/2 lb. Janus Green B, in the dye-bath; 16 lb. sumac extract and 2 lb. tartar emetic in the fixing bath.
Blue Black.--3-1/2 lb Janus Black I and 1/3 lb. Janus Black I I in the dye-bath, and 16 lb. sumac extract and 2 lb. tartar emetic in the fixing bath.
Dark Brown.--2-1/2 lb. Janus Brown B, 1 lb. Janus Black I, 3-1/2 oz. Janus Yellow G, and 5 oz. Janus Red B in the dye-bath, with 16 lb. sumac extract and 2 lb. tartar emetic in the fixing bath.
Drab.--1-1/2 oz. Janus Yellow R, 1/4 oz. Janus Red B, 1 oz. Janus Blue R, and 1/4 oz. Janus Grey B B, in the dye-bath, and 4 lb. sumac extract and 1 lb. tartar emetic in the fixing-bath.
Grey.--5 oz. Janus Blue R, 3-1/4 oz. Janus Grey B, 1-1/2 oz. Janus Yellow R, and 1/4 oz. Janus Red B in the dye-bath, with 4 lb. sumac extract and 1 lb. tartar emetic in the fixing-bath.
Nut Brown.--1 lb. Janus Brown R, 8 oz. Janus Yellow R, and 1-1/2 oz. Janus Blue B in the dye-bath, and 8 lb. sumac extract and 1 lb. tartar emetic in the fixing-bath.
Walnut Brown.--3 lb. Janus Brown B, 1 lb. Janus Red B, 1 lb. Janus Yellow R, and 1-1/4 oz. Janus Green B in the dye-bath, with 8 lb. sumac extract and 1 lb. tartar emetic in the fixing-bath.
Crimson.--2-1/2 (p. 183) lb. Janus Red B, and 8 oz. Janus Claret Red B in the dye-bath, with 8 lb. sumac extract and 1 lb. tartar emetic in the fixing-bath.
Dark Green.--1-1/2 lb. Janus Green B, 1 lb. Janus Yellow R, and 8 oz. Janus Grey B in the dye-bath, with 4 lb. sumac extract and 1-1/4 lb. tartar emetic in the fixing-bath.
Chestnut Brown.--1 lb. Janus Brown R and 1 lb. Janus Yellow R in the dye-bath, and 8 lb. sumac extract and 1 lb. tartar emetic in the fixing-bath.
Before the introduction of the direct dyes the method usually followed, and indeed is now to a great extent, is that known as Cross-dyeing. The goods were woven with dyed cotton threads of the required shade and undyed woollen threads; after weaving and cleansing the woollen part of the fabric was dyed with acid dyes such as Acid Magenta, Scarlet R, Acid Yellow, etc. In such methods care has to be taken that the dyes used for dyeing the cotton are such as stand acids, a by no means easy condition to fulfil at one time. Many of the direct dyes are fast to acids and therefore lend themselves more or less readily to cross-dyeing. For details of the dyes for cotton reference may be made to the sections on dyeing with the direct colours in the companion volume to this book on Dyeing of Cotton Fabrics.
Shot Effects.--A pleasing kind of textile fabric which is now made and is a great favourite for ladies' dress goods is where the cotton of a mixed fabric is thrown up to form a figured design. It is possible to dye the two fibres in different colours and so produce a variety of shot effects. These latter are so endless that it is impossible here to enumerate all that may be produced. It will have to suffice to lay down the lines which may be followed to the best advantage, and then give some recipes to illustrate the remarks that have been made. The best plan for the production of shot effects upon union fabrics is to take advantage of the property of certain (p. 184) acid dyes which dye only the wool in an acid bath and of many of the direct colours which will only dye the cotton in an alkaline bath. The process, working on these lines, becomes as follows: The wool is first dyed in an acid bath with the addition of Glauber's salt and bisulphate of soda or sulphuric acid, the goods are then washed with water containing a little ammonia to free them from the acid and afterwards dyed with the direct colour in an alkaline bath.
Fancy or the mode shades are obtained by combining suitable dye-stuffs.
If the cotton is to be dyed in light shades it is advantageous to dye on the liquor at 65° to 80° F., with the addition of 3-1/4 oz. Glauber's salt, and from 20 to 40 grains borax per gallon water. The addition of an alkali is advisable in order to neutralise slight quantities of acid which may have remained in the wool, and to prevent the dye-stuff from dyeing the cotton too deep a shade.
Very light shades can also be done on the padding machine. The dye-stuffs of Group (2), which have been previously enumerated, do not stain the wool at all or only very slightly and are therefore the most suitable. Less bright effects can be produced by simply dyeing the goods in one bath. The wool is first dyed at the boil with the wool dye-stuff in a neutral bath, the steam is then shut off and the cotton dyed by adding the cotton dye-stuff to the bath and dyeing without again heating. By passing the goods through cold water to which some sulphuric or acetic acid is added the brightness of most effects is greatly increased.
Gold and Green.--First bath, 1 lb. Cyanole extra, 7-1/4 oz. Acid Green, 1-1/2 oz. Orange G G, and 10 lb. bisulphate of soda; work at the boil for one hour, then lift and rinse well. Second bath, 4 lb. Diamine Orange G and 15 lb. Glauber's salt; work in the cold or at a lukewarm heat. Third bath at 120° F., 4 oz. Chrysoidine and 1/4 oz. Safranine.
Black (p. 185) and Blue.--First bath, 3-1/2 lb. Naphthol Black 3 B and 10 lb. bisulphate of soda. Second bath, 2 lb. Diamine Sky Blue and 13 lb. Glauber's salt. Third bath, 6-1/2 oz. New Methylene Blue N; work as in the last recipe.
Green and Claret.--First bath, 3-1/2 lb. Naphthol Red C and 10 lb. bisulphate of soda. Second bath, 2 lb. Diamine Sky Blue F F, 1-1/4 lb. Thioflavine S, and 15 lb. Glauber's salt.
Gold Brown and Blue.--First bath, 2-1/2 oz. Orange E N Z, 1-1/2 oz. Orange G G, 1/4 oz. Cyanole extra, and 10 lb. bisulphate of soda. Second bath, 14 oz. Diamine Sky Blue F F and 15 lb. Glauber's salt.
Dark Brown and Blue.--First bath, 1/2 lb. Orange G G, 1-1/2 oz. Orange E N Z, 1-1/2 oz. Cyanole extra and 10 lb. bisulphate of soda. Second bath, 12 oz. Diamine Sky Blue F F and 15 lb. Glauber's salt.
Black and Green Blue.--First bath, 3 lb. Orange G G, 1 lb. Brilliant cochineal 4 R, 1 lb. Fast Acid Green B N, and 10 lb. Glauber's salt. Second bath, 1-3/4 lb. Diamine Sky Blue F F, 3-1/4 lb. Thioflavine S, and 15 lb. Glauber's salt.
We may here note that in all the above recipes the second bath (for dyeing the cotton) should be used cold or at a lukewarm heat, and as strong as possible. It is not completely exhausted of colour, only about one-half going on the fibre. If kept as a standing bath this feature should be borne in mind and less dye-stuff used in the dyeing of the second and following lots of goods.
Blue and Gold Yellow.--3 lb. Diamine Orange G, 13 oz. Naphthol Blue G, 14-1/2 oz. Formyl Violet S 4 B, and 15 lb. Glauber's salt; work at just under the boil.
Brown and Blue.---1 lb. Diamine Steel Blue L, 9-1/2 oz. Diamine Sky Blue, 1 lb. Orange E N Z, 1 lb. Indian Yellow G, 1-3/4 oz. Naphthol Blue Black and 15 lb. Glauber's salt. Work at 170° to 180° F.
In these two last recipes only one bath is used, all the dyes (p. 186) being added at once. This is possible if care be taken that dye-stuffs are used which will dye wool and not cotton from neutral baths and dyes which dye cotton better than wool. The temperature should also be kept below the boil and carefully regulated as the operation proceeds and the results begin to show themselves.
Grey and Orange.--First bath, 3 oz. Orange extra, 1-1/4 lb. Cyanole extra, 11 lb. Azo Red A, and 10 lb. bisulphate of soda. Second bath, 5 oz. Diamine Orange D C and 3 oz. Diamine Fast Yellow B.
Green and Red.--First bath, 2 lb. Croceine A Z and 10 lb. Glauber's salt. Second bath, 1 lb. Diamine Sky Blue F F, 1/2 lb. Thioflavine S, and 15 lb. Glauber's salt.
Brown and Violet.--First bath, 3/4 lb. Orange extra, 3/4 lb. Cyanole extra, and 10 lb. bisulphate of soda. Second bath, 5 oz. Diamine Brilliant Blue G and 15 lb. Glauber's salt.
Black and Yellow.--First bath, 7 lb. Naphthol Black B, 1/2 lb. Fast Yellow S, and 10 lb. bisulphate of soda. Second bath, 3 lb. Diamine Fast Yellow A and 15 lb. Glauber's salt.
Black and Pink.--Black as above. Pink with Diamine Rose B D (see above).
Green and Buff.--First bath, 1/4 lb. Orange extra, 3/4 oz. Fast Yellow S and 10 lb. bisulphate of soda. Second bath, 3/4 lb. Diamine Sky Blue F F, 1/2 lb. Thioflavine S, and 15 lb. Glauber's salt.
Orange and Violet.--First bath, 9 oz. Orange extra and 10 lb. bisulphate of soda. Second bath, 3/4 lb. Diamine Violet N and 10 lb. Glauber's salt.
Black and Blue.--First bath, Naphthol Black, as given above. Second bath, Diamine Sky Blue, as given above.
Black and Yellow.--Add first 1 lb. Wool Black 6 B and 10 lb. Glauber's salt, then when the wool has been dyed add 2 lb. Curcumine S to dye the cotton in the same bath.
Green and Red.--Dye the wool by using 3 lb. Guinea Green B, (p. 187) 1/4 lb. Curcumine extra, and 10 lb. Glauber's salt, then add to the bath 3/4 lb. Erika B N and 3/4 lb. Congo Corinth G.
Orange and Blue.--Dye the wool first with 1-1/4 lb. Mandarine G, 2 oz. Wool Black 6 B, and 10 lb. Glauber's salt; then the cotton with 2 lb. Columbia Blue G.
Blue and Orange.--Dye the wool first with 3/4 lb. Guinea Violet B, 3/4 lb. Guinea Green B, and 10 lb. Glauber's salt; then dye the cotton with 2 lb. Mikado Orange 4 R O.
Green and Orange.--Dye the wool with 3 lb. Guinea Green B, 1/4 lb. Curcumine extra and 10 lb. Glauber's salt, then dye the cotton in the same bath with 1-1/2 lb. Mikado Orange 4 R O.
Gloria is a material which during the last few years has become of considerable importance as furnishing a fine lustrous fabric at a comparatively low price. The perfection to which the art of dyeing has attained and the facilities now available to the dyer, enable this to be produced more beautiful than ever, and naturally an increased demand for it as a dress fabric has developed.
Gloria is woven from the two fibres, wool and silk, of a fine texture to enable it to be used in the place of a silk fabric. Formerly it was usually woven with the wool and silk yarns already dyed, especially when a "shot" effect was to be produced, this being done by a twill weave of the fabric and by the use of yarns of two very different colours in the case of "shot" fabrics. By the introduction of dye-stuffs derived from coal tar the cloth is now dyed after being woven, care being taken to choose those which will dye the two fibres equally well when self-shades are wanted, or those which will dye one fibre better than the other, and thus allow a woven piece of gloria to be dyed of two different colours. As most dyers know, the most brilliant effects are obtained when the finished woven piece can be dyed. Then all the grease and dirt which has become attached to it during the operations of spinning the yarns and weaving the pieces can be removed before dyeing, thus leaving the fabric in a perfectly clean condition. Thus no after cleansing is required, (p. 189) whereas when the fibres are dyed in the yarn the goods must be cleansed after weaving to free them from dirt, and such cleaning has a somewhat deleterious effect upon the brilliancy of the colour of the finished fabric, more especially in the case of light colours.
Gloria may be in one colour only, a self-colour as it is called; this case is comparatively simple, the only care that is required being to select dyes which have an equal affinity for the two fibres or which give but slightly different shades. Still, some good effects are obtained when dyes are used which dye the silk and wool different colours but give the combined effect of a self-colour. Or the fibre may be purposely dyed in two different colours in some cases to give the "shot" effect. This is much more troublesome, but with a little care can be carried out with good results. The dyes available for dyeing gloria may be classified, according to their behaviour in regard to their dyeing of the two fibres, into three groups as follows:--
Group A.--Those which will dye the two fibres of equal shade.
Group B.--Those which will dye the wool at boiling heat more readily than the silk.
Group C.--Those which will dye the silk only in a cold bath.
Group A consists of those dyes which can be used in dyeing self-colours on gloria from acid baths. It includes Alkali Blue, Naphthylamine Blacks, Naphthol Green B, Indian Yellow, Croceine A Z, Croceine Orange, Orange R, Brilliant Croceine M, Rose Bengale, Thiocarmine R, Soluble Blue, Formyl Violet S 4 B, Acid Green, Croceine Orange G, Carmoisin, Acid Violet 5 B, Fast Acid Violet 10 B, Fast Green Bluish, Rhodamine, Silk Blue, Victoria Black, Archil, Turmeric, Safranine, Auramine, Quinoline Yellow, Azoflavine, Victoria Blue and Bismarck Brown.
Group (p. 190) B comprises those dye-stuffs which in a boiling acid bath dye the wool deeper than the silks, in other words have more affinity for the wool than the silk, Tropæoline O, Acid Magenta, Indigo Extract, Phloxine, Naphthol Yellow, Orange G G, Scarlet S, Azo Red A, Eosines, Thiocarmine R, Naphthol Black B B, New Victoria Black Blue, Erythrosine, and Roccelline.
The silk becomes tinted to a more or less extent when in such a bath, but often the colour is readily removed either by subsequent passage through boiling water or through hot soap liquor. A very good clearing can be effected by the use of a bath of acetate of ammonia. Naphthol Yellow, for instance, only imparts a very faint shade of yellow when thus dyed, and this is easily removed by boiling-water treatment.
Group C.--Those dye-stuffs which will dye the silk more readily in a cold bath than the wool. These comprise most of the basic dyes, such as Thioflavine T, Safranine, Brilliant Green, Methyl Violet, Magenta, New Methylene Blue, Bismarck Brown, Rose Bengale, Phloxine, Acid Greens, Formyl Violet S 4 B, Rhodamine, Solid Blue, etc.
Gloria may be dyed either by a one-bath or two-bath process, and either one or two colours, as may be required. In both cases advantage may be taken of the different affinities of the two fibres for the dye-stuffs used, as, for instance, the silk may be dyed brown, the wool olive by using a mixture of Acid Yellow, Indigo extract and Orange G. Indigo extract, Cochineal, Acid Magenta, Picric acid, Naphthol Yellow, and Tartrazine dye the wool only at the boil.
The following recipes will serve to illustrate the foregoing remarks and show how this important fabric may be dyed:--
Deep Gold.--The dye-bath is made from 2 lb. Indian Yellow, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, dyed at the boil. In this and following recipes the quantities are for 100 lb.
Orange.--The (p. 191) dye-bath is made with 2 lb. Indian Yellow, 19 lb. Glauber's salt, and 2 lb. sulphuric acid.
Scarlet.--Make the dye-bath with 2 lb. Scarlet 3 R, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. Another scarlet is got from 2 lb. sulphuric acid. Another scarlet is got from 2 lb. Croceine Scarlet 3 B, 2 lb. sulphuric acid, and 10 lb. Glauber's salt; by using the 5 B Scarlet a bluer shade can be dyed. Azo Cochineal also dyes a fine scarlet on gloria.
Crimson.--Make the dye-bath with 1 lb. Carmoisin B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. The 7 B Croceine Scarlet also dyes a fine crimson of a more fiery tone than the last, while 2-1/2 lb. Azo Fuchsine G dyes a bluer shade of crimson.
Rose.--A fine rose is obtained with 2 lb. Rhodamine B, 10 lb. Glauber's salt, and a little acetic acid. 1 lb. Phloxine dyes a fine deep rose; the silk comes out a paler colour than the wool, but the general effect is good.
Deep Maroon.--Make the dye-bath from 1-1/2 lb. Croceine A Z, 1/2 lb. Indian Yellow, 1/4 lb. Formyl Violet S 4 B, 10 lb. bisulphate of soda. Enter the goods, work at the boil for an hour, then cool down to 120° F., enter an equal quantity of dye-stuff and work for an hour longer.
Pale Maroon.--Make the dye-bath with 3 lb. Azo Bordeaux, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Black.--Prepare the dye-bath with 5 lb. Naphthylamine Black D, 1 lb. Acid Green B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid; work at the boil for twenty minutes, then allow to cool to 120° or 130° F., then work an hour longer. Another black can be dyed in a similar way from 5 lb. Victoria Black B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Violet.--Use 2 lb. Acid Violet 5 B, or 2 lb. Formyl Violet S 4 B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. Fast Acid Violet 10 B gives a bluer shade than the above.
Green.--Make (p. 192) the dye-bath with 2 lb. Acid Green G G, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil. This gives a bright yellow shade of green; a bluer shade can be got from Acid Green 6 B or Acid Green B, while Fast Green Bluish gives very blue greens.
Coeruleum Blue.--Dye with 3/4 lb. Silk Blue B E S, 10 lb. Glauber's salt, and 2 lb. sulphuric acid; this gives a very fine bright blue.
Deep Indigo Blue.--Dye with 4-1/2 lb. Solid Blue R, 2 lb. Thiocarmine R paste, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Deep Violet Brown.--Dye with 3 lb. Croceine A Z, 1-1/4 lb. Indian Yellow, 1-3/4 lb. Formyl Violet S 4 B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid for an hour at the boil, and for an hour at 120° F.
Blue Black.--Make the dye-bath with 5 lb. New Victoria Blue Black, 10 lb. Glauber's salt, and 2 lb. sulphuric acid, working at the boil. Another plan is to use 5 lb. Naphthylamine Black 4 B and 10 lb. bisulphate of soda.
Dark Grey.--Prepare a dye-bath with 3 lb. Naphthol Black 3 B, 4 lb. Naphthol Green B, 1 lb. Amaranth, 10 lb. Glauber's salt, 8 lb. copperas, and 3 lb. sulphuric acid, working at the boil for an hour and then rinsing in water to which a little acetate of ammonia has been added. The silk is dyed grey and the wool a black.
Brown.--A fine yellow brown shot with lilac is obtained by first dyeing in a bath of 5 lb. Naphthol Yellow, 10 lb. Glauber's salt and 2 lb. sulphuric acid. Wash in hot water, then dye with 2-1/2 lb. Solid Blue P G, 1-1/2 oz. Methyl Violet B O, and 5 lb. acetic acid in the cold.
Wool, Orange; Silk, Pale Green.--Dye the wool with 1-1/2 lb. Orange G G, 6 oz. Naphthol Green B, 2-1/2 oz. Naphthol Red C, 10 lb. bisulphate of soda, and 2 lb. sulphuric acid; and the silk with 1/2 lb. Milling Yellow and 1/2 lb. Acid Green.
Wool, (p. 193) Black; Silk, Light Grey.--Dye in a bath with 5 lb. Anthracene Acid Black S T, 4-1/2 oz. Fast Yellow S, 10 lb. bisulphate of soda, and 2 lb. sulphuric acid. The silk is cleaned by boiling for ten minutes in a soap bath.
Wool, Bright Red; Silk, Blush Rose.--The gloria silk is dyed in a bath of 3 lb. Naphthol Red O, 10 lb. bisulphate of soda, and 2 lb. sulphuric acid. After dyeing, soap for ten minutes.
Wool, Black; Silk, Green.--Dye the wool in a bath containing 5 lb. Anthracene Acid Black S T, 5 oz. Fast Yellow S, 2 lb. oxalic acid, 10 lb. Glauber's salt, and 15 lb. acetic acid. Work the goods in this at the boil for an hour, then lift, add 3/4 lb. bichromate of potash, and boil for twenty minutes longer. Clean the silk by boiling in a bath of soap for twenty minutes, then dye in a cold bath containing 1 lb. Thioflavine T and 1 lb. Brilliant Green.
Wool, Dark Maroon; Silk, Pale Blue.--After the manner described in the first recipe, dye the wool with 1 lb. Orange G G, 3 lb. Naphthol Green B, 2 lb. Brilliant Cochineal 2 R, 10 lb. bisulphate of soda, and 2 lb. sulphuric acid. Dye the silk with 1-1/2 lb. Pure Blue O T.
Wool, Violet; Silk, Green.--Make the dye-bath with 1 lb. Acid Violet 4 B, 9 oz. Indigotine extra, 10 lb. bisulphate of soda, and 2 lb. sulphuric acid. The dyeing is carried on at the boil until the bath is exhausted of colour, whereupon the goods are well rinsed in water. They are next soaped at 160° F. for ten minutes in a liquor containing 1/2 oz. soap per gallon, then rinsed. Next a dye-bath is made with 1 lb. Acid Green, 8 oz. Milling Yellow O, and 1 lb. acetic acid, the goods being treated in this in the cold until the desired shade is obtained, then lifted, rinsed and dried.
Violet and Pink.--A fine effect of violet shot with pink is obtained by dyeing in a bath of 1-1/2 lb. Indigo extract, 1/2 lb. Rhodamine B, 10 lb. Glauber's salt, and 2 lb. sulphuric acid.
Brown Olive and Green is dyed in a bath made with 1 lb. Quinoline (p. 194) Yellow, 1 lb. Azo Fuchsine G, 1/4 lb. Fast Green Bluish, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. By using about half the above quantities of dye-stuffs a drab effect shot with green can be obtained.
Crimson and Green.--The first bath is made from 4 lb. Azo Red A and 10 lb. bisulphate of soda, worked for an hour at the boil; then treat in a weak bath of acetate of ammonia; and dye the silk in a cold bath of 2 oz. Solid Green Crystals, 1/4 lb. Thioflavine T, and 5 lb. acetic acid.
Violet and Pink.--Dye in a bath of 1-1/4 lb. Indigo extract, 10 lb. Glauber's salt and 2 lb. sulphuric acid.
Brown and Pink.--This is dyed in a bath made from 1-1/4 lb. Fast Yellow, 5 oz. Rhodamine B, 1/4 lb. indigo extract, 10 lb. Glauber's salt, and 2 lb. sulphuric acid. The silk dyes a pale pink while the general effect is that of a fine fawn brown with a reddish shot effect.
Dark Green and Pale Crimson.--This is done in two baths, the first is made with 8 lb. Naphthol Green B, 10 lb. Glauber's salt, 3 lb. sulphuric acid, and 7 lb. copperas, working at the boil; then treat with hot water and dye in a fresh bath with 6 oz. Safranine Prima and 5 lb. acetic acid in the cold. The combined effect of the two is that of a brown shot with green.
Orange and Green.--This gives a splendid shot effect and is dyed as follows. Work for an hour at the boil, for thirty minutes in a bath of boiling water, then enter into a cold bath of 5 oz. Thioflavine T, 3 oz. Brilliant Green, and 3 lb. acetic acid; work for thirty minutes, or until shade is obtained.
Orange and Blue.--Use first dye-bath as in the last, then, after washing in hot water, dye in a bath of 2 oz. New Methylene Blue N, and 3 lb. acetic acid.
Silk, Sky Blue; Wool, Drab.--Make a dye-bath with 20 lb. acetic acid, 3/4 oz. Indigotine, 3 oz. Fast Yellow extra and 2 (p. 195) oz. Azo Fuchsine G. Work at the boil for one hour at 100° F., then pass into a bath of 3/4 oz. Turquoise Blue B B, and 2 lb. acetic acid, working for half an hour at 80°.
Silk, Pink; Wool, Pale Blue.--Make a dye-bath with 15 lb. acetic acid and 4-1/2 oz. Indigotine. Work at the boil for an hour, then pass into a bran bath as before; next enter into a dye-bath at 80° to 90° of 3/4 oz. Brilliant Rhoduline R B, 1-1/2 oz. Auramine I I, and 2 lb. acetic acid.
Silk, Green; Wool, Dark Crimson.--The first bath is made from 3 lb. Azo Fuchsine G, 1 lb. Indian Yellow G and 20 lb. acetic acid; then follows the bran and the final dye-bath, which is made from 1-1/2 oz. Imperial Green G I, and 2 lb. acetic acid.
Silk, Orange; Wool, Black.--A dye-bath is made from 2 lb. Indigotine, 2 lb. Indian Yellow G, 1/2 lb. Rhodamine G, and 20 lb. acetic acid. Work at the boil for one hour; then lift, wash and dry.
Silk, Light Green; Wool, Dark Blue.--Make a dye-bath from 1/2 lb. Azo Fuchsine G, 2 lb. Fast Light Green, and 20 lb. acetic acid. Work at the boil to shade; then lift, wash and dry.
Silk, Yellow; Wool, Terra Cotta.--A dye-bath is made from 1-1/2 oz. Indigotine, 3/4 lb. Azo Fuchsine G, 9 oz. Indian Yellow R, and 20 lb. acetic acid. Work at the boil for one hour; then lift, wash and dry.
Silk, Light Sea Green; Wool, Pale Sage.--Make the dye-bath with 1/2 lb. Fast Yellow extra, 3 oz. Azo Fuchsine G, 1-1/2 oz. Fast Green bluish, and 20 lb. acetic acid. Work as in the last recipe.
Silk, Light Green; Wool, Brown.--Make the dye-bath with 1 lb. Azo Fuchsine G, 2-1/2 lb. Fast Yellow extra, 1/2 lb. Fast Green bluish, and 20 lb. acetic acid. Work at the boil for one hour.
Silk, Pale Blue; Wool, Crimson.--Make a dye-bath with 2 lb. (p. 196) Azo Crimson L and 20 lb. acetic acid. Work at the boil for one hour, then pass into a bran bath for half an hour at 90° F., and into another bath containing 1/2 lb. Turquoise Blue G, and 2 lb. acetic acid, at 90° F., for half an hour; then wash and dry.
Silk, Light Drab; Wool, Lavender.--Make the first dye-bath from 3 oz. Indigotine, 2 oz. Azo Fuchsine G, and 20 lb. acetic acid. After working an hour at the boil, pass into a bran bath for half an hour, afterwards topping with 1-1/2 oz. Bismarck Brown R and 2 lb. acetic acid.
After loose wool, or woollen yarns or piece goods of every description have been dyed, before they can be sent out for sale they have to pass through various operations of a purifying character. There are some operations through which cloths pass that have as their object the imparting of a certain appearance and texture to them, these are generally known as finishing processes, of these it is not intended here to speak, but only of those which precede them but follow on the dyeing operations.
These processes are usually of a very simple character, and common to most colours which are dyed, and here will be noticed the appliances and manipulations necessary in the carrying out of these operations.
Squeezing or Wringing.--It is advisable when the goods are taken out of the dye-bath to squeeze or wring them according to circumstances in order to express out all surplus dye-liquor, which can be returned to the dye-bath if needful to be used again. This is an economical proceeding in many cases, especially in working with many of the old tannin materials, like sumac, divi-divi, myrobalans, and the modern direct dyes, which during the dyeing operations are not completely extracted out of the bath, or in other words the dye-bath is not exhausted of colouring matter, and therefore it can be used again for another lot of goods simply by adding fresh material to make up for that absorbed by the first lot.
Loose (p. 198) wool and loose cotton are somewhat difficult to deal with by squeezing or wringing, but the material may be passed through a pair of squeezing rollers such as are shown in figure 24, which will be more fully dealt with later on.
Yarns in Hanks.--In the hand-dyeing process of hank-dyeing the hanks are wrung by placing one end of the hank on a wringing-horse placed over the dye-tub, and a dye-stick in the other end of the hank, giving two or three sharp pulls to straighten out the yarn and then twisting the stick round; the twisting of the yarns puts some pressure on the fibres thoroughly and uniformly squeezing out the surplus liquor from the yarn.
Hank-Wringing Machines.--Several forms of hank-wringing machines have been devised. One machine consists of a pair of discs fitted on an axle, these discs carry strong hooks on which the hanks are placed. The operator places a hank on a pair of the hooks. The discs revolve and carry round the hank, during the revolution the hank is twisted and the surplus liquor wrung out, when the revolution of the discs carries the hank to the spot where it entered the machine, the hooks fly back to their original position, the hank unwinds, it is then removed and a new hank put in its place, and so the machine works on, hanks being put on and taken off as required. The capacity of such a machine is great and the efficiency of its working good.
Mr. S. Spencer, of Whitefield, makes a hank-wringing machine which consists of a pair of hooks placed over a vat. One of the hooks is fixed, the other is made to rotate. A hank hung between the hooks is naturally twisted and all the surplus liquor wrung out, the liquor falling into the vat.
Roller Squeezing Machines for Yarn.--Hanks may be passed through a pair of indiarubber squeezing rollers which may be so arranged that they can be fixed as required on the dye-bath. (p. 199) Such a pair of rollers is a familiar article and quite of common and general use in dye-houses.
Piece Goods.--These are generally passed open through a pair of squeezing rollers, which are often attached to the dye-vat in which the pieces are dyed.

Read Holiday's Squeezing Machine.--In figure 24 is shown a squeezing machine very largely employed for squeezing all kinds of piece goods after dyeing or washing. It consists of a pair of heavy rollers on which, by means of the screws shown at the top, a very considerable pressure can be brought to bear. The piece is run through the eye shown on the left, by which it is made into a rope form, then over the (p. 200) guiding rollers and between the squeezing rollers and into waggons for conveyance to other machines. This machine is effective.
Another plan on which roller, or rather in this case disc, squeezing machines are made is to make the bottom roller with a square groove in the centre, into this fits a disc, the cloth passing between them. The top disc can, by suitable screws, be made to press upon the cloth in the groove and thus squeeze the water out of it.
Washing.
One of the most important operations following that of dyeing is the washing with water to free the goods, whether cotton or woollen, from all traces of loose dye, acids, mordanting materials, etc., which it is not desirable should be left in, as they might interfere with the subsequent finishing operations. For this purpose a plentiful supply of good clean water is required, this should be as soft as possible, free from any suspended matter which might settle upon the dyed goods and stain or speck them.
Washing may be done by hand, as it frequently was in olden days, by simply immersing the dyed fabrics in a tub of water, shaking, then wringing out, again placing in fresh water to finish off. Or if the dye-works were on the banks of a running stream of clean water the dyed goods were simply hung in the stream to be washed in a very effectual manner.
In these days it is best to resort to washing machines adapted to deal with the various kinds of fibrous materials and fabrics, in which they can be subjected to a current of water.
Loose Wool.--If this has been dyed by hand then the washing may also be done in the same way by hand in a plain vat. If the dyeing has been done on a machine then the washing can be done on the same machine.
(p. 201)Yarn (p. 202) in Hanks.--A very common form of washing machine is shown in figure 25. As will be seen it consists of a wooden vat, over which are arranged a series of revolving reels on which the hanks are hung, the hanks are kept in motion through the water and so every part of the yarn is thoroughly washed. Guides keep the hanks of yarn separate and prevent any entanglement one with another. A pipe delivers constantly a current of clean water, while another pipe carries away the used water. Motion is given to the reels in this case by a donkey engine attached to the machine, but it may also be driven by a belt from the main driving shaft of the works. This machine is very effective.

Piece Goods.--Piece goods are mostly washed in machines, of which two broad types may be recognised. First those where the pieces are dealt with in the form of ropes or in a twisted form, and second those where the pieces are washed while opened out full width. There are some machines in which the cloths may be treated either in the open or rope form as may be thought most desirable.
Figure 26 represents a fairly well-known machine in which the (p. 203) pieces are treated in a rope-like form. It consists of a trough in which a constant current of water is maintained; at one end of this trough is a square beating roller, at the other a wood lattice roller, above the square beater and out of the trough are a pair of rollers whose purpose is to draw the cloth through the machine and also partly to act as squeezing rollers. As will be seen the cloth is threaded in rope form spirally round the rollers, passing in at one end and out at the other, pegs in a guide rail serving to keep the various portions separate. The square beater in its revolutions has (p. 204) a beating action on the cloth, tending to more effectual washing. The lattice roller is simply a guide roller.
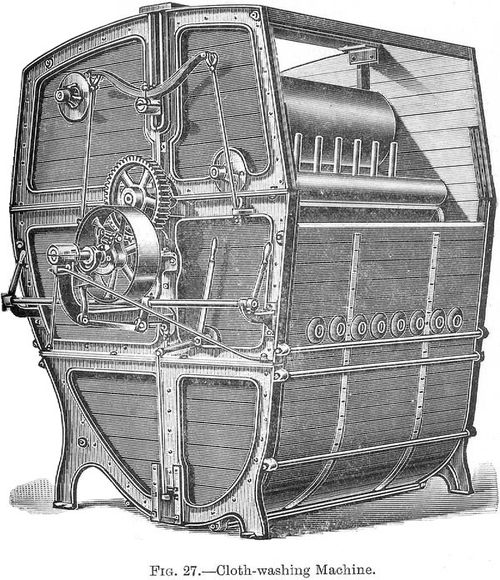
Figure 27 shows a washing machine very largely used in the wool-dyeing trade. The principal portion of this machine is of wood.
The internal parts consist of a large wooden bowl, or oftener, as in the machine under notice, of a pair of wooden bowls which are pressed together by springs with some small degree of force. Between these bowls the cloth is placed, more or less loosely twisted up in a rope form, and the machines are made to take four, six or eight pieces or lengths at one time, the ends of the pieces being stitched together so as to make a continuous band. A pipe running along the front of the machine conveys a constant current of clean water, which is caused to impinge in the form of jets on the pieces of cloth as they run through the machine, while an overflow carries away the used water. The goods are run in this machine as long as is considered necessary for a sufficient wash, which may take half to one and a half hours.
In figure 30 is shown a machine designed to wash pieces in the broad or open state. The machine contains a large number of guide rollers built more or less open, round which the pieces are guided, the ends of the pieces being stitched together, pipes carrying water are so arranged that jets of clean water impinge on and thoroughly wash cloth as it passes through, the construction of the guide rollers facilitating the efficient washing of the goods.
Soaping.
Sometimes yarns or cloths have to be passed through a soap-bath after being dyed in order to brighten up the colours or develop them in some way. In the case of yarns (p. 205) this can be done on the reel washing machines such as are shown in figure 25. In the case of piece goods a continuous machine in which the washing, soaping, etc., can be carried on simultaneously is often employed. Such a machine is shown in figure 28. It consists of a number of compartments fitted with guide rollers, so that the cloth passes up and down several times through the liquors in the compartment; between one compartment and another is placed a pair of squeezing rollers. The cloth is threaded in a continuous manner, well shown in the drawing, through the machine; in one compartment it is treated with water, in another with soap liquor, and another with water, and so on, and these machines may be made with two, three or more compartments, as may be necessary for the particular work in hand. As seen in the drawing the cloth passes in at one end, and out at the other finished. It is usually arranged that a continuous current of the various liquors used flows through the various compartments, thus ensuring the most perfect treatment of the cloths.
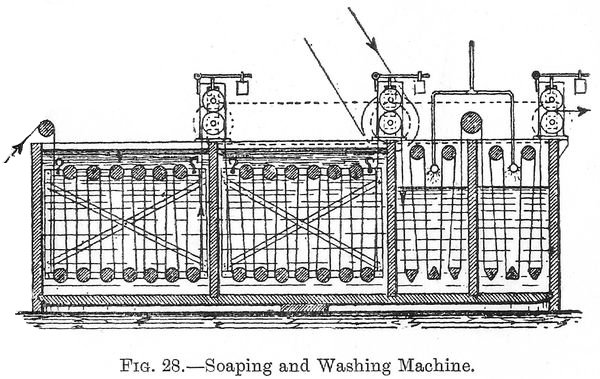
Following on the washing comes the final operation of the dyeing process, that of drying the dyed and washed goods. Now textile fabrics of all kinds after they have passed through dye-baths, washing machines, etc., contain a large amount of water, often exceeding in amount that of the fabric itself, and to take the goods direct from the preceding operations to the drying plant means that a considerable amount of fuel must be expended to drive off this large amount of water. It is therefore very desirable that the goods be freed from as much of this water as possible before they are sent into any drying chambers, and this may be done in three ways, by wringing, squeezing and hydro-extracting. The first two methods have already been described (pp. 198, 199) and need not again be alluded to, the last needs some account.

Hydro-extractors (p. 207) are a most efficient means for extracting water out of textile fabrics. They are made in a variety of forms by several makers. Essentially they consist of a cylindrical vessel with perforated sides, so constructed that it can be revolved at a high speed. This vessel is enclosed in an outer cage. The goods are placed in the basket, as it is termed, and then this is caused to revolve; at the high speed at which it revolves centrifugal action comes into play and the water contained in the goods finds its way to the outside of the basket through the perforations and so away from the goods. Hydro-extractors are made in a variety of sizes and forms, in some the driving gear is above, in others below the basket, in some the driving is done by belt gearing, in others a steam engine is directly connected with the basket. Figures 29 and 30 show two forms which are much in use in the textile industry. They are very efficient and extract water from textile goods more completely than any other means, as will be obvious from a study of the table below.
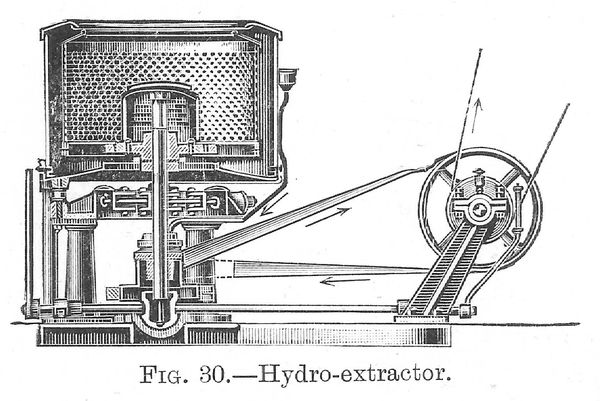
The relative efficiency of the three systems of extracting the moisture out of textile fabrics has been investigated by Grothe, who gives in his Appretur der Gewebe, published in 1882, the following table showing the percentage amount of water removed in fifteen minutes:--
(p. 208)| Yarns. | Wool. | Silk. | Cotton. | Linen. |
| Wringing | 44·5 | 45·4 | 45·3 | 50·3 |
| Squeezing | 60·0 | 71·4 | 60·0 | 73·6 |
| Hydro-extracting | 83·5 | 77 | 81·2 | 82·8 |
| Pieces. | ||||
| Wringing | 33·4 | 44·5 | 44·5 | 54·6 |
| Squeezing | 64·0 | 69·7 | 72·2 | 83·0 |
| Hydro-extracting | 77·8 | 75·5 | 82·3 | 86·0 |
In the practical working of hydro-extractors it is of the utmost importance that the goods be carefully and regularly laid in the basket, not too much in one part and too little in another. Any unevenness in this respect at the speed at which they are driven leaves such a strain on the bearings as to seriously endanger the safety of the machine.
After being wrung, squeezed or hydro-extracted the goods are ready to be dried. In the case of yarns this may be done in rooms heated by steam pipes placed on the floor, the hanks being hung on rods suspended from racks arranged for the purpose.
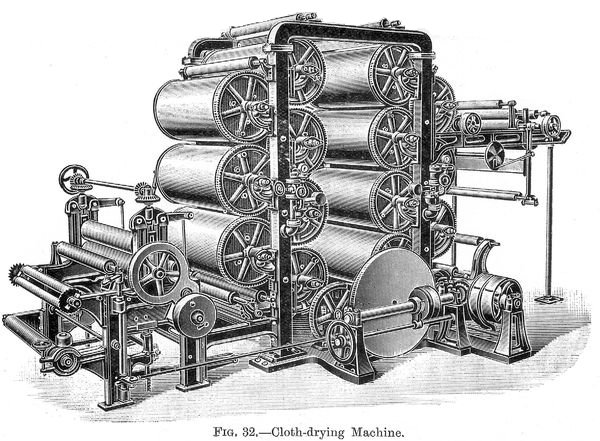
Where large quantities of yarn have to be dried it is most economical to employ a yarn or drying machine, and one form of such is shown in figure 31. The appearance of the machine is that of one long room from the outside, internally it is divided into compartments, each of which is heated up by suitably arranged steam pipes, but the degree of heating in (p. 209) each compartment varies, at the entrance end it is high, at the exit end low. The yarn is fed in at one end, being hung on rods, and by suitable gearing it is carried directly through the various chambers or sections, and in its passage the heat to which it is subject drives off the water it contains. The yarn requires no attention from the time it passes (p. 210) in wet at the one end of the machine and comes out dry at the other end. The amount of labour required is slight, only that represented by filling the sticks with wet yarn and emptying the dried yarn. The machine works regularly and well.
The drying is accomplished by circulating heated air through the yarns, this heating being effected by steam coils; fresh air continually enters the chambers while water-saturated air is as continually being taken out at the top of the chamber. One of the great secrets in all drying operations is to have a constant current of fresh hot air playing on the goods to be dried, this absorbs the moisture they contain, and the water-charged air thus produced must be taken away as quickly as possible.
Piece Goods.--The most convenient manner of drying piece goods is to employ the steam cylinder drying machine such as is shown in figure 32. This consists of a number of hollow tin or copper cylinders which can be heated by steam passing in through the axles of the cylinders, which are made hollow on purpose. The cloth to be dried passes round these cylinders, which revolve while the cloth passes. They work very effectually.
Every dyer ought to be able to make experiments in the mordanting and dyeing of textile fibres for the purpose of ascertaining the best methods of applying mordants or dye-stuffs, the best methods of obtaining any desired shade, and for the purpose of making comparative tests of dyes or mordanting materials with the object of determining their strength and value. This is not by any means difficult, nor does it involve the use of any expensive apparatus, so that a dyer need not hesitate to set up a small dyeing laboratory for fear of the expense which it might entail.
In order to carry out the work indicated above there will be required several pieces of apparatus. First a small chemical balance; one that will carry 50 grammes in each pan is quite large enough, and such a one, quite accurate enough for this work, can be bought for 25s. to 30s., while if the dyer be too poor even for this a cheap pair of apothecaries' scales might be used. It is advisable to procure a set of gramme weights and to get accustomed to them, which is not by any means difficult.
In using the balance always put the substance to be weighed on the left-hand pan and the weights on the right-hand pan. Never put chemicals of any kind direct on the pan, but weigh them in a watch-glass, small porcelain basin, or glass beaker (which has first been weighed), according to the nature of the material which is being weighed. The sets of (p. 212) weights are always fitted into a block or box, and every time they are used they should be put back into their proper place.
The experimenter will find it convenient to provide himself with a few small porcelain basins, glass beakers, cubic centimetre measures, two or three 200 c.c. flasks with a mark on the neck, a few pipettes of various sizes, 10 c.c., 20 c.c., 25 c.c.
The most important feature is the dyeing apparatus. Where only a single dye test is to be made a small copper or enamelled iron saucepan, such as can be bought at any ironmongers may be used; this may conveniently be heated by a gas-boiling burner, such as can also be bought at an ironmongers or plumbers for 2s.
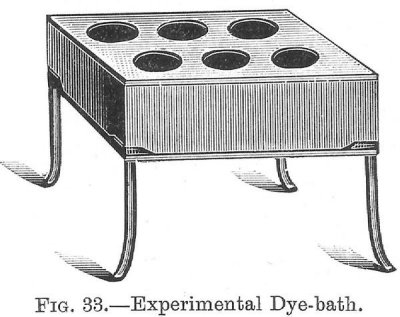
It is, however, advisable to have means whereby several dyeing experiments can be made at one time and under precisely the same conditions, and this cannot be done by using the simple means noted above.
To be able to make perfectly comparative dyeing experiments it is best to use porcelain dye-pots (these may be bought from most dealers in chemical apparatus), and to heat these in a water-bath arrangement.
The simplest arrangement is sketched in figure 33; it consists of a copper bath measuring 15 inches long by 10-1/2 inches broad (p. 213) and 6-1/2 inches deep; this is covered by a lid on which are six apertures to take the porcelain dye-baths. The bath is heated by two round gas-boiling burners of the type already referred to.
The copper bath is filled with water which, on being heated to the boil by the gas burners, heat up the dye-liquors in the dye-pots. The temperature in the dye-pots under such conditions can never reach the boiling point; where it is desirable, as in some cases of wool mordanting and dyeing that it should be so high, then there should be added to the water in the copper bath a quantity of calcium chloride, which forms a solution that has a much higher boiling point than that of water, and so the dye-liquors in the dye-pots may be heated up to the boil.
An objection might be raised that with such an apparatus the temperature in every part of the bath may not be uniform, and so the temperature of the dye-liquors in the pots might vary also, and differences of temperature often have a considerable influence on the shade of the colour which is being dyed. This is a minor objection, which is more academic in its origin than of practical importance. To obviate it Mr. William Marshall, of the Rochdale Technical School, has devised a circular form of dye-bath, in which the temperature in every part can be kept quite uniform.
The dyeing laboratories of Technical Schools and Colleges are generally provided with a more elaborate set of dyeing appliances. These in the latest constructed consist of a copper bath supported on a hollow pair of trunnions, so that it can be turned over if needed. Into the bath are firmly fixed three earthenware or porcelain dye-pots; steam for heating can be sent through the trunnions. After the dyeing tests have been made the apparatus can be turned over and the contents of the dye-pots emptied into a sink which is provided for the purpose.
Many (p. 214) other pieces of apparatus have been devised and made for the purpose of carrying on dyeing experiments on the small scale, but it will not be needful to describe these in detail. After all no more efficient apparatus can be desired than that described above.
Dyeing experiments can be made with either yarns or pieces of cloth, swatches as they are commonly called; a very convenient size is a small skein of yarn or a piece of cloth weighing 5 grammes. These test skeins or pieces ought to be well washed in hot water before use, so that they are clean and free from any size or grease. A little soda or soap will facilitate the cleansing process.
In carrying out a dyeing test the dye-pot should be filled with the water required, using as little as is consistent with the dye-swatch being handled comfortably therein, then there is added the required mordants, chemicals, dyes, etc., according to the character of the work which is being done.
Of such chemicals as soda, caustic soda, sodium sulphate (Glauber's salt), tartar, bichromate of potash, it will be found convenient to prepare stock solutions of known strength, say 50 grammes per litre, and then by means of a pipette any required quantity can be conveniently added. The same might be followed in the case of dyes which are constantly in use, in this case 5 grammes per litre will be found strong enough.
Supposing it is desired to make a test of a sample of Acid Red, using the following proportions, 2 per cent. dye-stuff, 3 per cent. sulphuric acid and 15 per cent. Glauber's salt, and the weight of the swatch which is being used is 5 grammes, the following calculations are to be made to give the quantities of the ingredients required:--
For the dye-stuff, 5 (weight of swatch) multiplied by 2 (per cent. of dye) and divided by 100 equals (5 x 2) / 100 = 0·1 gramme of dye.
For (p. 215) the acid we have similarly (5 x 3) / 100 = 0·15 gramme of acid.
For the Glauber's salt (5 x 15) / 100 = 0·75 gramme of Glauber's salt.
These quantities may be weighed out and added to the dye-bath, or if solutions are kept a calculation can be made as to the number of cubic centimetres which contain the above quantities, and these measured out and added to the dye-bath.
When all is ready the bath is heated up, the swatch put in and the work of the test entered upon.
Students are recommended to make experiments on such points as:--
The shades obtained by using various proportions of dye-stuffs.
The influence of various assistants: common salt, soda, Glauber's salt, borax, phosphate of soda in the bath.
The influence of varying proportions of mordants on the shade of dyeing.
The value of various assistants, tartar, oxalic acid, lactic acid, sulphuric acid, on the fixation of mordants.
The relative value of tannin matters, etc.
Each dyer should make himself a pattern book into which he should enter his tests, with full particulars as to how they have been produced at the side.
It is important that a dyer should be able to make comparative dye-tests to ascertain the relative strength of any two or more samples of dyes which may be sent to him.
This is not difficult but requires considerable care in carrying out the various operations involved.
0·5 gramme of each of the samples of dyes should be weighed out and dissolved in 100 c.c. of water, care being taken (p. 216) that every portion of the dye is dissolved before any of the solution is used in making up the dye-vats. Care should be taken that the skeins of yarn or swatches of cloth are exactly equal in weight, that the same volume of water is placed in each of the dye-pots, that the same amounts of sulphate of soda or other dye assistants are added, that the quantities of dye-stuffs and solutions used are equal, in fact that in all respects the conditions of dyeing are exactly the same, such in fact being the vital conditions in making comparative dye-tests of the actual dyeing strength of several samples of dyes.
After the swatches have been dyed they are rinsed and then dried, when the depths of shade dyed on them may be compared one with another. To prevent any mistakes it is well to mark the swatches with one, two, three or more cuts as may be required.
It is easier to ascertain if two dyes are different in strength of colour than to ascertain the relative difference between them. There are two plans available for this purpose; one is a dyeing test, the other is a colorimetric test made with the solutions of the dyes.
Dyeing Test.--This method of ascertaining the relative value of two dyes as regards strength of colour is carried out as follows. A preliminary test will show which sample is stronger than the other; then there is prepared a series of dye-vats, one contains a swatch with the deepest of the two dyes, which is taken as the standard, the others with the other dye but containing 2, 5 and 10 per cent. more dye-stuff, and all these are dyed together, and after drying a comparison can be made between these and the standard swatch, and a judgment formed as to the relative strength of the two dyes; a little experience will soon enable the dyer to form a correct judgment of the difference in strength between two samples of dye-stuff.
The (p. 217) colorimetric test is based on the principle that the colour of a solution of dye-stuff is proportionate to its strength. Two white glass tubes, equal in diameter, are taken; solutions of the dye-stuffs, 0·5 gramme in 100 c.c. of water, are prepared, care being taken that the solution is complete. 5 c.c. of one of these solutions is taken and placed in one of the glass tubes, and 5 c.c. of the other solution is placed in the other glass tube, 25 c.c. of water is now added to each tube and then the colour of the diluted liquids is compared by looking through in a good light. That sample which gives the deepest solution is the strongest in colouring power. By diluting the strongest solution with water until it is of the same depth of colour as the weakest, it may be assumed that the length of the columns of liquid in the two tubes is in proportion to the relative strength of the two samples. Thus if in one tube there are 30 centimetres of liquid and in the other 25 centimetres, then the relative strength is as 30 to 25, and if the first is taken as the standard at 100 a proportion sum may be worked out as follows:--
30: 25 :: 100 : 83·3;
that is, the weakest sample has only 83·3 per cent. of the strength of the strongest sample.
It is frequently desirable that dyers should be able to ascertain with some degree of accuracy what dyes have been used to dye any particular sample of dyed cloth that has been offered to them to match. In these days of the thousand-and-one different dyes that are known it is by no means an easy thing to do, and when, as is most often the case, two or three dye-stuffs have been used in the production of a shade, the difficulty is materially increased.
The only available method is to try the effect of various acid and alkaline reagents on the sample, noting whether any change of colour occurs, and judging accordingly. It would be a good thing for dyers to accustom themselves to test the dyeings they do and so accumulate a fund of practical experience which will stand them in good stead whenever they have occasion to examine a dyed pattern of unknown origin.
The limits of this book do not permit of there being given a series of elaborate tables showing the action of various chemical reagents on fabrics dyed with various colours, and such indeed serve very little purpose, for it is most difficult to describe the minor differences which often serve to distinguish one colour from another. Instead of doing so we will point out in some detail the methods of carrying out the various tests, and advise all dyers to carry these out for themselves on samples dyed with known colours, and when they have an unknown colour to test to make (p. 219) tests comparatively with known colours that they think are likely to have been used in the production of the dyed fabric they are testing.
One very common method is to spot the fabric, that is to put a drop of the reagent on it, usually with the aid of the stopper of the reagent bottle, and to observe the colour changes, if any, which ensue.
This is a very useful test and should not be omitted; and it is often employed in the testing of indigo dyed goods with nitric acid, those of logwood with hydrochloric acid, alizarine with caustic soda, and many others. It is simple and easy to carry out, and only takes a few minutes.
To make a complete series of tests of dyed fabrics there should be provided the following reagents:--
1. Strong sulphuric acid as bought.
2. Dilute sulphuric acid, being the strong acid diluted
with 20 times its volume of water.
3. Concentrated hydrochloric acid as bought.
4. Dilute hydrochloric acid, 1 acid to 20 water.
5. Concentrated nitric acid as bought.
6. Dilute nitric acid, 1 acid to 20 water.
7. Acetic acid.
8. Caustic soda solution, 5 grammes in 100 c.c. water.
9. Ammonia (strong).
10. Dilute ammonia, 1 strong ammonia to 10 water.
11. Carbonate of soda solution, 5 grammes in 100 c.c.
water.
12. Bleaching powder solution, 2° Tw.
13. Bisulphite of soda, 72° Tw.
14. Stannous chloride, 10 grammes crystals in 100 c.c.
water, with a little hydrochloric acid.
15. Methylated spirit.
Small swatches of the dyed goods are put in clean porcelain basins, and some of these solutions poured over them. Any (p. 220) change of colour of the fabric is noted as well as whether any colour is imparted to the solutions. After making observations of the effects in the cold, the liquids may be warmed, and the results again noted. After being treated with the acids the swatches should be well washed with water, when the original colour may be wholly or partially restored.
To give tables showing the effects of these reagents on the numerous dyes now known would take up too much room and not serve a very useful purpose, as such tables if too much relied on leave the operator somewhat uncertain as to what he has before him. The reader will find in Hurst's Dictionary of Coal-Tar Colours some useful notes as to the action of acids and alkalies on the various colours that may be of service to him.
Alizarine and the series of dye-stuffs to which it has given its name, fustic, cochineal, logwood and other dyes of a similar class, require the fabric to be mordanted, and the presence of such mordant is occasionally an indirect proof of the presence of these dyes.
To detect these mordants a piece of the swatch should be burnt in a porcelain or platinum crucible over a bunsen burner, care being taken that all carbonaceous matter be burnt off. A white ash will indicate the presence of alumina mordants, red ash that of iron mordants, and a greenish ash chrome mordants.
To confirm these the following chemical tests may be applied. Boil the ash left in the crucible with a little strong hydrochloric acid and dilute with water. Pass a current of sulphuretted hydrogen gas through the solution, if there be any tin present a brown precipitate of tin sulphide will be obtained. This can be filtered off. The filtrate is boiled for a short time with nitric acid, and ammonia is added to the solution when alumina is thrown down as a white, gelatinous precipitate, (p. 221) iron is thrown down as a brown red, bulky precipitate, while chrome is thrown down as a greyish-looking, gelatinous precipitate. The precipitate obtained with the ammonia is filtered off and a drop of ammonium sulphide added, when any zinc present will be thrown down as white precipitate of zinc sulphide; to the filtrate from this ammonium oxalate may be added, when if lime is present a white precipitate of calcium oxalate is obtained.
A test for iron is to dissolve some of the ash in a little hydrochloric acid and add a few drops of potassium ferrocyanide solution, when if any iron be present a blue precipitate will be obtained.
To make more certain of the presence of chrome, heat a little of the ash of the cloth with caustic soda and chlorate of soda in a porcelain crucible until well fused, then dissolve in water, acidify with acetic acid and add lead acetate, a yellow precipitate indicates the presence of chrome.
A book on qualitative chemical analysis should be referred to for further details and tests for metallic mordants.
The fastness of colours to light, air, rubbing, washing, soaping, acids and alkalies is a feature of some considerable importance, there are indeed few colours that will resist all these influences, and such are fully entitled to be called fast. The degree of fastness varies very considerably, some colours will resist acids and alkalies well, but are not fast to light and air; some will resist washing and soaping, but are not fast to acids; some may be fast to light, but are not so to washing. The following notes will show how to test these features.
Fastness to Light and Air.--This is simply tested by hanging a piece of the dyed cloth in the air, keeping a piece in a drawer to refer to, so that the influence on the original colour can be noted from time to time. If the piece is left out in the open one gets not only the effect of light but also that (p. 222) of climate on the colour, and there is no doubt rain, hail and snow have some influence on the fading of the colour. If the piece is exposed under glass the climatic influences do not come into play, and one gets the effect of light alone.
In making tests of fastness the dyer will and does pay due regard to the character of the influences that the material will be subjected to in actual use, and these vary very considerably; thus the colour of underclothing need not be fast to light, for it is rarely subjected to that agent of destruction; on the other hand, it must be fast to washing, for that is an operation to which underclothing is subjected week by week.
Window curtains are much exposed to light and air, and, therefore, the colours in which they are dyed should be fast to light and air. On the other hand, these curtains are rarely washed, and so the colour need not be quite fast to washing. And so with other kinds of fabrics; there are scarcely two kinds which are subjected to the same influences and require the colours to have the same degree of fastness.
The fastness to rubbing is generally tested by rubbing the dyed cloth with a piece of white paper.
Fastness to Washing.--This is generally tested by boiling a swatch of the cloth in a solution of soap containing 4 grammes of a good neutral curd soap per litre for ten minutes, and noting the effect whether the soap solution becomes coloured and to what degree, or whether it remains colourless, and also whether the colour of the swatch has changed at all.
One very important point in connection with the soaping tests is whether a colour will run into a white fabric that may be soaped along with it. This is tested by twisting strands of the dyed yarn or cloth with white yarn or cloth and boiling them in the soap liquor for ten minutes and then noting the (p. 223) effect, particularly observing whether the white pieces have taken up any colour.
Fastness to acids and fastness to alkalies is observed while carrying out the various acid and alkali tests given above.
A.
Acetate of ammonia,
93,
94,
101,
102,
127,
128,
129,
132,
167,
192,
194.
------- of chrome,
115.
------- of lime,
158,
159.
Acetic acid,
127.
Acid black,
37,
89.
---- ----- B,
92,
99.
---- ----- B B,
99,
111,
112.
---- ----- S,
90,
99.
---- blue 4 S,
98,
127.
---- ---- 1 V,
153.
---- dyes for blue,
152.
---- ---- for brown,
161.
---- ---- for green,
128.
---- ---- for mode colours,
165.
---- ---- for violet,
160.
---- dye-stuffs,
61.
---- green,
53,
91,
92,
127,
184,
189,
190,
192,
193.
---- ----- B,
128,
191.
---- ----- blue shade,
136.
---- ----- B N,
136.
---- ----- extra,
155.
---- ----- G G,
192.
---- magenta,
73,
105,
111,
113,
183,
190.
---- mauve,
96.
---- ----- B,
161.
---- red, test for,
214.
---- violet,
105.
---- ------ 4 B,
193.
---- ------ 5 B,
154,
160,
189,
191.
---- ------ 5 B E,
162.
---- ------ 6 B,
130,
171,
181.
---- ------ 10 B,
191.
---- ------ N,
92,
99,
161,
162.
---- ------ 6 R N,
161.
---- ------ 4 R S,
160.
---- ------ V,
162.
---- ---- 1 V,
153.
---- yellow,
53,
99,
123,
183,
190.
Acids, action on wool,
11.
Acridine red,
102.
-------- scarlet,
102.
Adjective group of dye-stuffs,
68.
Alizarine,
61,
69,
72,
73,
86,
114,
220.
--------- black,
99.
--------- ----- S W,
94,
113.
--------- blue,
116,
119,
166.
--------- ---- A,
158.
--------- ---- D N W,
131,
132,
133,
158,
164,
166.
--------- ---- S W,
108.
--------- Bordeaux,
133.
--------- -------- B,
98,
155.
--------- -------- G,
155,
159.
--------- brown,
131,
132,
133,
158,
164,
166.
--------- claret R,
118.
--------- colours,
77.
--------- cyanine,
111,
119,
156.
--------- ------- black,
93,
94,
159,
160.
--------- ------- G,
159.
--------- ------- G G,
98,
157.
--------- ------- G extra,
157.
--------- ------- R,
99,
157.
--------- ------- R R,
157.
--------- ------- R R R,
93,
157.
--------- ------- 3 R double,
157,
160.
--------- G,
122.
--------- green,
127.
--------- ----- S,
132.
--------- ----- S W,
132.
--------- grey B,
166.
--------- orange,
119,
123.
--------- ------ 2 G,
120.
--------- ------ H,
164.
--------- ------ N,
118,
122,
166.
--------- ------ W,
119.
--------- ------ R,
163.
--------- ------ R R,
122.
--------- red 1 W S,
118,
119,
120,
122.
--------- --- 2 W S,
118.
--------- --- 3 W S,
119,
164.
--------- --- 5 W S,
118,
120.
--------- S X,
120.
--------- yellow,
70,
71,
115,
116,
123,
131,
133,
156,
164,
166.
--------- ------ G G,
115,
122.
--------- ------ G G W,
94,
125,
126,
131,
132,
164.
--------- ------ R W,
122.
Alkali blue,
152,
189.
------ ---- B,
152.
------ ---- 6 B,
180.
------ yellow R,
169.
Alkalies, action on wool,
9.
Alkaline blue 6 B,
178.
-------- ----
171,
177.
Alpaca,
1,
83.
Alum,
74,
77,
85,
86,
97,
115,
117,
129,
131.
Alumina,
114.
------- sulphate,
115,
117.
Aluminium salts,
8.
Amaranth,
92,
108,
111,
192.
Amido-benzoic acid,
114.
Ammonia,
17,
27,
33,
78.
------- action on wool,
60.
Angora goat,
1.
Annotta,
13,
63.
Anthracene acid black S T,
193.
---------- ---- browns,
115.
---------- blue W B,
159.
---------- ---- W G,
132,
158,
159.
---------- brown,
94,
119,
132.
---------- ----- R,
163.
---------- ----- W,
159,
164.
---------- chrome black,
96,
99.
---------- ------ ----- F,
95.
---------- ------ ----- F F,
92,
96.
---------- red,
122,
134.
---------- yellow,
69,
70,
115.
---------- ------ B N,
96,
126,
135.
---------- ------ C,
90,
98,
109,
122,
124,
125,
126,
132,
163,
167.
---------- ------ G G,
126.
Anthracite black B,
90,
96,
132,
163.
---------- ----- R,
90,
98.
Anthragallol,
114.
Archil,
75,
189.
------ substitute N,
99,
107,
110,
131,
155,
162,
165.
Argol,
86,
97,
115,
116,
117,
151.
----- lactic acid,
116.
Artificial wool,
174.
Auramine,
53,
64,
103,
189.
-------- base,
64.
-------- I I,
195.
Auroline,
169.
Azo acid brown,
130.
--- ---- magenta G,
162.
--- ---- rubine,
111.
--- ---- violet 4 R,
109,
111,
161.
--- ---- yellow,
171.
--- black,
89.
--- blue,
171.
--- Bordeaux,
109,
191.
--- carmine,
124,
161,
166.
--- ------- B,
130.
--- cochineal,
105,
112,
191.
--- crimson L,
196.
--- dye-stuffs,
61,
66.
--- flavine,
189.
--- ------- S,
210.
--- fuchsine,
109,
115.
--- -------- G,
108,
130,
160,
162,
191,
194,
195,
196.
--- green,
70,
127.
--- mauve,
171.
--- red A,
108,
111,
171,
176,
177,
179,
190,
194.
--- rubine,
92.
--- scarlet,
53.
--- yellow,
93,
124,
128,
129,
130,
155,
162,
165.
B.
Basic dyes for violet,
160.
----- dye-stuffs,
61.
Batching of wool,
15,
25.
Benzo azurine 3 G,
170.
----- ------- R G,
170.
----- blue black G,
170.
----- brown,
61.
----- dyes,
168.
----- fast red,
100,
102,
110.
----- ---- scarlet,
62.
----- ---- ------- B S,
102.
----- flavine,
64.
----- green,
127.
----- orange R,
121.
Benzol,
16,
24.
Benzoline,
25.
Benzopurpurine,
61,
100.
-------------- B,
170.
-------------- 4 B,
111,
170,
180,
181.
-------------- 10 B,
170,
180.
Bichromate of potash,
16,
115,
131,
132,
133,
134,
135,
166,
167,
193.
---------- of potassium,
175.
Bisulphate of soda,
33,
131,
141,
146,
167,
184,
192,
193,
194.
Bismarck brown,
189,
190.
-------- ----- R,
196.
Black,
93,
95,
176,
177,
180,
191,
193,
195.
----- and blue,
185,
186.
----- and green blue,
185.
----- and pink,
186.
----- and yellow,
186.
----- blue,
152,
157.
----- ---- O,
155.
----- on wool,
83,
91.
Bleaching wool,
29.
Blue,
153,
158.
---- and gold yellow,
185.
---- and orange,
187.
---- black,
96,
153,
159,
179,
182,
192.
---- ----- on wool,
89,
90,
91,
92,
94.
---- green,
127,
128,
129,
130.
---- shades on wool,
136.
Bluestone,
74,
86,
87,
88,
135.
Bluish Bordeaux red,
110.
------ crimson,
108.
------ green,
134.
------ red,
106,
120.
------ pink,
111,
112.
------ purple,
109.
------ violet,
160.
------ rose,
193.
Borax,
215.
Bordeaux,
102.
-------- B L,
110.
-------- red,
109,
110,
113.
Bottle green,
127,
130,
132,
134.
Bran,
138,
144,
145.
Brazil wood,
114.
Bright blue,
152,
153,
155,
156,
158,
180.
------ Bordeaux red,
109,
110.
------ buff,
164,
165.
------ canary,
124.
------ cherry red,
109,
110.
------ chestnut,
164.
------ crimson,
108.
------ fawn,
165.
------ ---- red,
118.
------ electric blue.
156.
------ golden brown,
163.
------ grass green,
130,
133.
------ green,
127,
128,
134.
------ greenish blue,
154.
------ leaf green,
129.
------ lemon yellow,
125.
------ maroon,
119.
------ moss green,
129.
------ orange,
121,
122.
------ pale sage green,
131.
------ peacock green,
130.
------ red,
111,
193.
------ scarlet,
102,
112.
------ straw,
124.
------ violet,
161.
------ ------ blue,
156.
------ yellow,
123,
124,
175,
176,
178.
Brilliant alizarine blue G,
133,
157,
158,
161.
--------- azurine 5 G,
170.
--------- cochineal 2 R,
112,
123,
193.
--------- --------- 4 R,
123,
185.
--------- Congo G,
102.
--------- ----- R,
170.
--------- croceine B,
106,
123.
--------- -------- 3 B,
123.
--------- -------- 5 B,
123.
--------- -------- 7 B,
123.
--------- -------- 9 B,
123.
--------- -------- B B,
106.
--------- -------- M,
106.
--------- -------- M O O,
123.
--------- -------- N,
189.
--------- green,
53,
64,
127,
190,
193,
194.
--------- milling green B,
171.
--------- orange,
92.
--------- orseille C,
107,
112.
--------- pale bluish crimson,
108.
--------- ponceau G,
106.
--------- ------- 2 R,
106.
--------- ------- 4 R,
112.
--------- purpurine R,
170.
--------- rhoduline R B,
195.
--------- royal blue,
154.
--------- scarlet,
119,
171.
--------- ------- G,
171.
--------- ------- 4 R,
105.
Bronze green,
131,
180.
Brown,
161,
163,
164,
181,
192,
195.
----- and violet,
186.
----- and pink,
194.
----- and blue,
185.
----- black,
94.
----- olive and green,
193.
----- shades on wool,
161.
Buff,
164.
C.
Calcium salts,
8.
Camel-hair,
83.
Camwood,
76,
86.
Carbohydrate,
7.
Carbonate of soda,
27,
78,
169.
Carbon disulphide,
16,
24.
Carbonising of wool,
11.
Carded wool, dyeing of,
44.
Carmoisin,
189.
--------- B,
191.
Cashmere,
83,
173.
-------- goat,
1.
Caustic soda,
141.
------- lye,
147.
Celestine blue B,
155.
Chemical vats,
138.
Chemic extract,
150.
Cherry red,
109,
110.
Chestnut,
163.
-------- brown,
184.
Chicago blue B,
170.
------- ---- 4 B,
170.
------- ---- 6 B,
170,
180.
------- ---- G,
170.
------- ---- R W,
181.
------- ---- R R W,
170.
Chloramine orange,
121.
---------- yellow,
169.
Chlorination of wool,
37.
Chlorine, action on wool,
12.
Cholesterine,
7,
23.
Chrome,
114.
------ acetate,
129.
------ alum,
115.
------ blue,
158.
------ Bordeaux 6 B,
161.
------ brown R,
164.
------ fluoride,
77.
------ logwood black,
84,
85.
------ ------ jet black,
85.
------ mordant,
151.
------ patent black D G,
92.
------ violet,
115,
119.
Chromine G,
169.
Chromogene I,
120.
Chromotrop,
115.
---------- 2 B,
125.
---------- 6 B,
108,
154.
---------- 10 B,
94,
109.
---------- R,
106.
---------- 2 R,
99,
107,
129,
130,
155,
162,
165.
---------- S,
93,
94.
Chrysamine,
61,
128,
170.
---------- G,
165.
Chrysoidine,
184.
Chrysophenine,
61,
102,
128,
170.
------------- G,
180.
Claret,
110,
111,
118,
120.
------ red,
110.
Clayton yellow,
170.
Cloth-drying machine,
209.
----- red,
73.
----- washing machine,
29,
30,
202,
203.
Coal tar,
137.
---- --- colours,
114.
---- --- dyes,
63.
---- --- ---- for dyeing blue,
152.
Coatings,
173.
Cochineal,
97,
114,
190,
220.
--------- scarlet,
77.
Coerulein,
114,
133.
--------- B,
132.
--------- S W,
132.
--------- blue,
192.
Colour lakes,
113.
------ strength, test for,
216.
------ testing,
218.
Columbia black B,
170.
-------- ---- F B,
170,
181.
-------- red 8 B,
170.
-------- yellow,
169.
Congo blue,
62.
----- brown G,
170,
180.
----- ----- R,
161,
171.
----- Corinth G,
171,
180,
187.
----- ------- B,
169,
171.
----- dyes,
168.
----- orange G,
170.
----- ------ R,
165,
170.
----- R,
170.
----- red,
62.
Copperas,
74,
86,
87,
88,
97,
133,
134,
135.
-------- vats,
138.
Copper-cased dye beck,
56.
Coral red,
112.
Cornflower blue,
181.
Corron's hank-dyeing machine,
49.
Cotton yellow,
170.
Cream of tartar,
116.
Crimson,
103,
108,
113,
180,
183,
191,
194.
Croceine A Z,
123,
171,
189,
191,
192.
-------- orange,
121,
122,
189.
-------- ------ E N,
123.
-------- scarlet,
108.
-------- ------- 3 B,
191.
-------- ------- 3 R,
167.
Cross dyeing,
183.
Crushed strawberry,
105.
------- ---------- red,
107,
118.
Crystal scarlet 6 R,
123.
Cudbear,
97.
Curcumine extra,
171,
181,
187.
--------- S,
180,
186.
Cutch,
76,
97.
----- brown,
76.
Cyanine B,
107,
111,
124,
129,
130,
155,
165.
------- scarlet E,
111.
Cyanole,
107,
111,
131,
165.
------- extra,
99,
108,
112,
113,
131,
155,
184,
185,
186.
------- green B,
134.
------- ----- 6 G,
134.
Cyprus green B,
136.
------ ----- R,
136.
D.
Dark beige green,
130.
---- blue,
152,
154,
157,
159,
176,
178,
179,
180,
181,
182,
195.
---- Bordeaux red,
120.
---- bottle green,
131,
132.
---- brown and blue,
185.
---- -----
163,
164,
177,
179,
180,
181,
182.
---- buff,
165.
---- chestnut,
162.
---- cherry red,
112.
---- crimson,
102,
195.
---- green,
127,
128,
131,
177,
180,
183.
---- ----- and pale crimson,
194.
---- grey,
98,
166,
167,
178,
181,
192.
---- invisible blue,
156.
---- maroon,
193.
---- navy,
157,
159.
---- ---- blue,
155.
---- olive brown,
162.
---- orange,
121.
---- peacock blue,
156.
---- red,
120.
---- sage,
179.
---- ---- green,
130.
---- sea green,
171.
---- seal,
162,
163.
---- slate,
159,
166,
177.
---- stone,
177.
---- violet,
161.
---- ------ brown,
164.
---- walnut,
164,
179.
Dead black on wool,
90.
Deep blue,
154,
155.
---- Bordeaux red,
109.
---- brown,
162.
---- crimson,
108,
112,
113,
118.
---- electric green,
131.
---- fawn,
107.
---- ---- red,
107,
119.
---- golden yellow,
125.
---- indigo blue,
192.
---- leaf green,
130.
---- ---- yellow,
125.
---- lemon,
125.
---- maroon,
111,
119,
191.
---- navy,
153.
---- ---- blue,
153.
---- olive yellow,
125.
---- orange,
122,
176.
---- red,
103.
---- sage green,
131,
132.
---- scarlet,
106,
112,
119.
---- seal,
162.
---- sky blue,
155.
---- violet,
160.
---- ------ brown,
192.
---- yellow,
124,
126.
Delahunty's dyeing machine,
43,
44.
Deltapurpurine 5 B,
130.
Diamine black,
99,
155.
------- ----- B H,
170,
177,
178.
------- ----- B O,
170.
------- ----- H W,
169,
177,
178.
------- ----- R O,
170.
------- blue,
62.
------- ---- 2 B,
170.
------- ---- 3 B,
170.
------- ---- B G,
170.
------- ---- B X,
170.
------- ---- G,
169.
------- ---- R W, 169,
170.
------- ---- 3 R,
170.
------- ---- black E,
170.
------- Bordeaux,
102.
------- -------- B,
169,
170,
177,
179.
------- -------- S,
170.
------- brilliant blue G,
170,
176,
178,
179.
------- bronze G,
171,
177.
------- brown,
62,
179.
------- ----- B,
169.
------- ----- G,
170.
------- ----- 3 G,
169.
------- ----- G W,
169.
------- ----- N,
169,
177.
------- ----- S,
170.
------- ----- V,
170,
177.
------- catechine B,
170,
179.
------- --------- G,
169,
170.
------- cutch,
176.
------- dark blue B,
169,
170,
180.
------- dyes,
168.
------- fast yellow A,
170,
175,
186.
------- ---- ------ B,
169,
176,
177,
179.
------- ---- red F,
98,
100,
102,
109,
112,
124,
132,
163,
167,
169.
------- gold,
121,
170,
175.
------- green,
127.
------- ----- B,
169.
------- ----- G,
169,
170.
------- new blue R,
170.
------- --- nitrazol brown B,
170.
------- --- G,
170.
------- orange B,
169,
177,
178,
179.
------- ------ D,
170.
------- ------ D C,
121.
------- ------ G,
170,
175,
178,
184,
185.
------- ------ G C,
121.
------- ------ O,
170.
------- red,
62,
169.
------- --- B,
170.
------- --- 5 B,
178.
------- --- N O,
170.
------- rose B D,
102,
169,
178,
186.
------- scarlet B,
112,
121,
122,
170,
178.
------- ------- 3 B,
170.
------- sky blue,
170,
185.
------- --- F F,
170,
185,
186.
------- steel blue L,
170,
177,
185.
------- violet N,
170,
178,
186.
Diamond black,
93,
99.
------- ----- F,
92.
------- ----- on wool,
93.
------- brown,
164.
------- flavine,
133,
163.
------- ------- G,
98.
------- green,
93.
------- yellow B,
133.
Dihydroxynaphthalene,
88.
----------- sulpho acid,
116.
Dinitroso-resorcine,
127.
Direct black,
88.
------ dyes,
197.
------ ---- for blue,
152.
------ ---- for brown,
161.
------ ---- for green,
127.
------ ---- for mode colours,
165.
------ ---- for orange,
121.
------ ---- for violet,
160.
------ orange R,
170.
------ red dyes,
100.
------ yellow G,
170.
Divi-divi,
197.
Drab,
165,
166,
167,
178,
179,
181,
182,
194.
Dress goods,
173.
Drying of goods,
205.
Dyeing machinery,
40,
43.
------ test,
216.
------ tubs,
41.
Dye-jiggers,
51,
52.
---- tests,
211.
---- vat with steam pipe,
42.
E.
Electric blue,
155.
Emerald green,
128,
129,
130,
135.
Emin red,
107,
110.
Eosine,
190.
------ red,
104.
Erie blue, 2 G,
170.
Erika B N,
170,
187.
Erythesine D,
112.
Erythrosine,
104,
190.
Experimental dye-bath,
212.
------------ dyeing,
211.
F.
Fast acid violet 10 B,
111,
130,
162,
165,
189.
---- ---- ------ R,
108,
110,
111,
112,
113,
120,
130,
166.
---- ---- blue R,
99,
107,
129,
155,
165.
---- ---- green B N,
96,
134,
185.
---- ---- magenta B,
105,
108,
109,
153.
---- black,
96.
---- blue,
37.
---- bright olive,
135.
---- chrome black,
92.
---- green,
127,
133.
---- green bluish,
111,
130,
154,
160,
162,
165,
189,
192,
194,
195.
---- ----- extra bluish,
162.
---- light green,
195.
---- red,
102,
111.
---- scarlet,
105.
---- yellow,
109,
124,
161,
162,
165,
166,
194.
---- ------ F Y,
90,
91,
105,
123,
130.
---- ------ S,
111,
113,
135,
186,
193.
---- ------ extra,
194,
195.
Fastness to acid, test for,
223.
-------- to alkalies, test for,
223.
-------- to light and air, test for,
221.
-------- to washing, test for,
222.
Fawn,
118.
---- drab,
179.
---- red,
107,
113.
Ferrous sulphate,
115,
117.
Fermentation vats,
138.
Flavazol,
70.
Fluoride of chrome,
91,
98,
102,
110,
115,
117,
129,
132,
133,
167.
Formyl blue B,
171.
------ violet,
53.
------ ------ 6 B,
171.
------ ------ 10 B,
171.
------ ------ S 4 B,
155,
161,
171,
175,
176,
178,
179,
180,
185,
189,
190,
191.
Fulling fast olive,
135.
Fustic,
66,
69,
70,
77,
83,
85,
86,
87,
97,
120,
220.
------ extract,
88,
123,
131,
133,
134,
135.
G.
Galleine,
166.
Gallipoli oil,
26.
Galloflavine,
70,
119,
133.
Gambine,
61,
114,
119,
127.
------- B,
164.
------- R, 133,
164,
167.
------- V,
96,
125,
133,
164.
------- yellow,
93,
115,
125.
Geranine B,
160.
-------- G,
102.
Glacier blue,
155.
Glauber's salt,
81,
89,
91,
99,
127,
128,
129,
130,
135,
150,
151,
169,
171,
172,
184,
215.
Gloria, dyeing of,
188.
Gold and green,
184.
---- brown,
176,
179,
185.
---- orange,
122,
123,
176.
---- yellow,
126.
Golden brown,
162,
163.
------ yellow,
125,
126.
Good yellow,
175.
Grass green,
128.
Green,
127,
131,
178,
192,
193,
195.
----- and buff,
186.
----- and claret,
185.
----- and red,
186.
----- and orange,
187.
Greenish,
98.
-------- black on wool,
91.
-------- straw,
124.
Grey,
98,
165,
182.
---- and orange,
186.
---- blue,
158.
---- on wool,
96.
Guinea green B,
171,
181,
187.
------ violet 4 B,
171,
181.
H.
Hæmatoxylin of logwood,
84.
Hand dyeing,
40.
---- scouring of wool,
18.
Hank-washing machine,
201.
---- wringing machines,
198.
Hare fur,
83.
Hessian violet,
102,
171.
Holliday's patent indigo vat,
143.
Hydrochloric acid,
88.
Hydrochloride of rosaniline,
9.
Hypochlorites, action on wool,
12.
Hydro-extractor,
206,
207.
Hydrosulphite of soda,
147.
------------- vats,
138,
141.
Hydroxy-azo dyes,
114.
I.
Imperial green G 1,
195.
Indian yellow,
90,
91,
189,
191,
192.
------ ------ G,
131,
171,
175,
176,
177,
179,
180,
185,
195.
------ ------ R,
126,
165,
171,
175,
176,
195.
Indigo,
83,
85,
136,
141.
------ black,
86.
------ blue,
151.
------ carmine,
66,
150.
------ ------- D,
161,
166.
------ dyeing,
137.
------ dye-stuffs,
61.
------ dye-vat,
149.
------ extract,
73,
75,
97,
105,
131,
133,
134,
135,
151,
190,
194.
------ ------- for dyeing wool,
150.
------ indophenol vat,
146.
Indigotine,
194.
---------- extra,
193,
195,
196.
Indophenol,
146.
Induline,
37.
-------- A,
153.
Invisible bronze green,
133.
--------- green,
130,
132,
136.
Iron logwood black,
86,
87.
Italian cloths,
176.
J.
Janus black I,
182.
----- ----- I I,
182.
----- blue R,
182.
----- brown B,
182.
----- ----- R,
182,
183.
----- claret red B,
183.
----- dark blue B,
182.
----- dyes,
181.
----- green B,
182,
183.
----- grey B,
182,
183.
----- ---- B B,
182.
----- red B,
182,
183.
----- yellow G,
182.
----- ------ R,
182,
183.
Jet black,
93,
94,
95,
96.
--- ----- on wool,
90,
91,
93.
Jig wince,
53.
K.
"Kempy" fibres,
3.
Keratine,
8.
Keton blue G,
111,
162.
Klauder-Weldon hank-dyeing machine,
47,
48.
L.
Lactic acid,
115,
116,
117,
151,
215.
Lanafuchsine 6 B,
113.
------------ S B,
111,
113.
------------ S G,
111,
112,
113.
Lanacyl blue B B,
171.
------- ---- R,
171.
------- violet B,
171,
180.
Lavender,
160,
196.
-------- blue,
158.
-------- grey,
166.
Leaf yellow,
125.
Lemon yellow,
125.
Level dyeing,
77.
Light drab,
196.
----- green,
133,
195.
----- grey,
97,
193.
----- sea green,
195.
----- straw,
126.
Lignorosine,
115,
117,
151.
Lilac,
166.
----- blue,
158.
----- grey,
165.
Lime,
117,
140.
---- vats,
138.
Liquor ammonia,
147.
Llama,
1.
Logwood,
66,
69,
70,
83,
85,
86,
87,
97,
136,
151,
220.
------- black,
87,
88.
------- ----- on wool,
86.
------- extract,
88,
133,
135,
136.
Loose wool, dyeing of,
43.
----- ---- washing of,
200.
M.
Machine-scouring of wool,
20.
Madder,
77,
144.
Magenta,
53,
61,
64,
102,
103,
190.
Maize yellow,
124.
Malachite green,
127.
Mandarine G,
121,
171,
180,
181,
187.
Maroon,
111,
118,
178.
------ red,
111.
Marseilles soap,
78.
Mauve,
161.
McNaught's wool-washing machine,
20.
Metallic salts, action on wool,
12.
Methylene blue,
133,
134.
Methylrosaniline,
64.
Methyl violet,
53,
64,
190.
------ ------ 3 B,
160.
------ ------ B O,
192.
------ ------ R,
160.
Medulla,
4.
Medium blue,
157,
158.
------ green,
133.
Merino wool,
5.
Mikado orange 4 R O,
180,
181,
187.
Milling red B,
111.
------- --- R,
106,
110.
------- yellow,
192.
------- ------ O,
99,
125,
193.
Mimosa,
169.
Mode colours on wool,
164.
Mordant dyes for brown,
163.
------- ---- for orange,
122.
------- dye-stuffs,
61,
68.
------- dyes for violet,
161.
Mordanting,
115.
---------- of wool,
12.
Mordant yellow,
119,
122,
132.
------- ------ D,
126.
------- ------ O,
164.
Moss green,
129,
130.
Mother vat,
147.
Mouse,
162.
Muriate of tin,
97.
Myrobalan,
197.
N.
Naphthol black,
37,
89,
99,
186.
-------- ----- B,
90.
-------- ----- B B,
196.
-------- ----- 3 B,
90,
91,
185,
192.
-------- ----- 4 R,
111.
-------- blue G,
171,
185.
-------- ---- R,
171.
-------- ---- black,
155,
171,
175,
177,
178,
179,
180,
185.
-------- green B,
37,
90,
127,
128,
189,
192,
193,
194.
-------- red C,
113,
185,
192.
-------- --- O,
193.
-------- yellow,
131,
136,
190.
-------- ------ S,
113,
130,
178.
Naphthyl blue black N,
92.
Naphthylamine black,
89,
92,
189.
------------- ----- 4 B,
91,
171,
192.
------------- ----- 6 B,
171,
180.
------------- ----- D,
91,
99,
171,
191.
------------- ----- S,
96.
Navy,
158.
---- blue,
153,
136,
177,
179,
180.
Neutral dye-stuffs,
61.
------- extract,
150.
------- red,
162.
New methylene blue,
190.
--- --------- ---- N,
185,
194.
--- Victoria black blue,
190.
--- -------- blue B,
154.
--- -------- ---- black,
192.
Nigrosine,
37.
Nitrate of iron,
98.
Nitrazine yellow,
124.
Nut,
164.
--- brown,
181,
182.
Nyanza black,
95.
------ ----- B,
99,
128,
161,
165.
O.
Obermaier dyeing machine,
44,
45,
46.
Old gold,
122,
126.
Oleic acid,
7,
26.
Oleine,
26.
Olive,
128,
134,
135.
----- brown,
162,
164.
----- bronze,
135.
----- green,
128,
135.
----- oil,
26.
----- yellow,
124,
125.
Orange,
121,
122,
178,
180,
191,
192,
195.
------ No. 2,
162.
------ blue,
187,
194.
------ green,
194.
------ violet,
186.
------ croceine G,
189.
------ E N Z,
123,
135,
171,
176,
178,
179,
180,
185.
------ extra,
99,
107,
108,
111,
113,
122,
162,
163,
171,
178.
------ G,
99,
107,
110,
113,
162,
165,
166,
190.
------ G G,
112,
113,
122,
162,
184,
185,
190,
191,
193.
------ I I,
153,
162.
------ O,
111.
------ R,
122,
189.
------ shades on wool,
121.
------ T A,
170,
181.
Oxalate of ammonia,
95.
Oxalic acid,
85,
87,
88,
93,
115,
116,
133,
151,
215.
Oxydiamine black A,
169.
---------- ----- B,
169.
---------- ----- B M,
180.
---------- ----- D,
169.
---------- ----- M,
169.
---------- ----- S O O O,
170.
---------- Orange G,
170,
178.
---------- ------ R,
170.
---------- red S,
170.
---------- violet B,
170.
---------- yellow G G,
170.
Oxyphenine,
169.
P.
Pale blue,
152,
155,
193,
195.
---- bluish crimson,
108.
---- chestnut,
164.
---- crimson,
108.
---- drab,
165,
166.
---- fawn,
166.
---- ---- drab,
165.
---- ---- brown,
166.
---- gold yellow,
175.
---- green,
192.
---- lilac rose,
107.
---- maroon,
191.
---- navy blue,
156.
---- old gold brown,
164.
---- olive yellow,
126.
---- orange,
121,
122.
---- pea-green,
131.
---- Russian green,
128.
---- sage,
195.
---- ---- green,
130,
133,
180.
---- sea green,
129,
132.
---- slate green,
133.
---- ---- grey,
98.
---- stone,
166.
---- violet,
160.
Pararosaniline,
64.
Paris blue,
158.
Patent blue,
92,
99.
------ ---- A,
131,
158.
------ ---- B,
95,
110,
154.
------ ---- J,
154,
162.
------ ---- J B,
166.
------ ---- J O O,
166.
------ ---- N,
128,
154.
------ ---- V,
111,
129,
130,
154,
155,
162,
168.
------ ---- superior,
154.
Peach wood,
86.
Peacock blue,
155,
157,
158.
------- green,
131,
132,
177,
179.
Pearl ash,
17.
----- grey,
97,
98.
Perchloride of tin,
97.
Peri wool blue,
155.
Peroxide of hydrogen for bleaching wool,
29,
34.
-------- of soda for bleaching wool,
36.
Persian berries,
69,
71.
Petrie's wool-washing machine,
20.
Petroleum spirit,
16,
24.
Phenoflavine,
124,
130.
Phenolic colours,
114.
Phenyl rosaniline,
64.
Phloxine,
104,
190,
191.
Phosphate of soda,
218.
Picric acid,
190.
Piece-dyeing machines,
50.
----- goods, drying of,
210.
----- ---- washing of,
202.
----- ---- wringing of,
199.
Pink,
102,
111,
112,
178,
195.
Plum,
178.
Plush fabric dyeing machine,
55.
Ponceau,
105.
------- 3 G,
121.
------- R,
65.
------- 3 R B,
171,
180.
Potash,
17.
------ indigo vat,
144.
------ salts,
7.
Potassium salts,
8.
Primuline,
169.
Puce,
160.
Pure blue O T,
193.
Purple,
109.
------ red,
113.
Purpuramine,
62.
Q.
Quick lime,
141.
Quinoline yellow,
189,
194.
R.
Rabbit fur,
83.
Raw merino wool, analysis of,
7.
Read Holliday's hawking machine,
57.
---- ---------- indigo extract,
151.
---- ---------- squeezing machine,
199.
---- ---------- yarn dyeing machine,
46,
47.
Red,
106,
107,
120.
--- navy,
158.
--- ---- blue,
157.
--- plum,
177.
--- shades on wool,
100.
Reddish black,
94.
------- grey,
97.
------- orange,
121.
------- puce,
160.
Rhodamine,
165,
189,
190.
--------- B,
113,
191,
193,
197.
--------- G,
195.
--------- red,
102.
Rhoduline red,
102,
103.
Rocceleine,
171,
190.
Roller-squeezing machine,
198.
Rose,
118.
Rosaniline,
64.
Rose bengale,
104,
112,
189,
190.
---- red,
113.
Royal blue,
154.
S.
Saddening of wool,
74.
Saffranine,
61,
64,
102,
103,
184,
189,
190.
---------- prima,
103,
194.
Saffron,
13,
63.
Saffrosine,
104.
Sage,
177.
---- brown,
181.
---- green,
128.
Salicylic acid,
114.
Salmon,
113.
------ red,
113.
Salt,
215.
Sanders,
120,
121.
Saxony blue,
154.
Scarlet,
101,
102,
103,
105,
106,
107,
112,
118,
178,
180,
191.
------- F R,
106.
------- O O,
106.
------- R,
183.
------- 3 R,
191.
------- 2 R J,
105.
------- R S,
105.
------- S,
190.
Schutzenberger and Lalande's vat,
141.
Schweizer's reagent,
9.
Scouring of wool,
15,
17.
-------- of woollen piece goods,
28.
Sea green,
131,
136.
Serge,
173.
Silicate of soda,
17.
Silk blue,
189.
---- ---- B E S,
192.
Silver grey,
98,
165,
177.
Sheep,
1.
Short-stapled wools, scouring of,
18.
Shot effects,
183.
Sky blue,
151,
154,
178,
194.
Slaked lime,
145.
Slate,
165,
181.
----- blue,
158,
179.
----- green,
131,
132,
181.
----- grey,
97,
98.
Sliver, dyeing of,
44.
Slubbing, dyeing of,
44.
Smithson's dyeing machine,
88.
Soap,
27.
---- action on wool,
10,
66.
Soaping and washing machine,
205.
------- of goods,
204.
Soda,
17,
215.
---- ash,
17.
---- crystals,
145.
---- indigo vat,
145.
Sodium hydrosulphite,
143.
Solid blue,
190.
----- ---- R,
192.
----- ---- P G,
192.
----- green crystals,
194.
Soluble blue,
189.
Sour extract,
150.
Southdown wool,
5.
Spencer's hank-wringing machine,
198.
Squeezing of goods,
197.
Stale urine,
17,
18.
Stearic acid,
7.
Stone,
166,
181.
Straw,
124.
Stuffing of wool,
74.
Suint,
15.
Suitings,
173.
Sulphon azurine B,
170.
------- ------- D,
180.
------- cyanine,
128,
152,
160.
Sulphur,
8.
------- bleach house,
31.
------- dioxide,
33.
------- bleaching,
29,
30.
Sulphuric acid,
99,
115,
116,
215.
Sumac,
86,
120,
121,
135,
197.
----- extract,
182,
183.
Sweet extract,
150.
T.
Tannic acid,
98.
Tannin materials,
197,
215.
Tartar,
85,
86,
88,
93,
115,
116,
117,
131,
132,
133,
134,
135,
151,
166,
167,
215.
------ emetic,
182,
183.
Tartaric acid,
85,
115.
Tartrazine,
190.
Terra-cotta,
195.
----- ----- red,
105,
120.
Tin chloride,
115.
--- crystals,
77.
--- salt,
133.
Thiazol yellow,
169.
Thiocarmine R,
98,
171,
177,
179,
189,
190.
Thioflavine S,
121,
169,
175,
178,
185,
186.
----------- T,
64,
190,
193,
194.
Titan blue,
170,
171.
----- ---- 3 B,
127.
----- brown O,
110,
170.
----- ----- R, 98,
170.
----- ----- T,
170.
----- marine B,
171.
----- pink,
170.
----- red,
61,
98,
107.
----- scarlet,
100.
----- ------- C B,
101,
102.
----- ------- D,
110.
----- ------- S,
169.
----- yellow,
61,
99,
170.
----- ------ G,
127.
----- ------ R,
125.
----- ------ Y,
125,
127.
Treacle,
138.
Tropæoline,
122,
131.
---------- O,
190.
---------- O O,
171,
178.
Turmeric,
13,
63,
120,
189.
Turquoise blue B B,
195.
--------- ---- G,
196.
--------- green,
134.
U.
Union black B,
169.
----- ----- S,
169,
176,
178,
179,
180.
----- blue B B,
169.
----- fabrics, dyeing of,
168.
----- flannels,
173.
Urine indigo vat,
145.
V.
Velvet, embossing of,
14.
Victoria black,
89,
189.
-------- ----- B,
91,
191.
-------- ----- blue,
155.
-------- blue,
189.
-------- ---- B,
155.
-------- ---- black,
91.
-------- scarlet R,
107,
110,
111.
-------- rubine O,
107,
111.
-------- violet 8 B S,
130,
155.
-------- yellow,
111,
124,
130,
162.
Violet,
160,
192,
193.
------ and pink,
193,
194.
------ black on wool,
89,
90,
91,
93,
95.
------ blue,
155.
------ grey,
166.
------ shades on wool,
160.
W.
Walnut,
162.
------ brown,
176,
182.
Washing of goods,
200.
Water blue,
37.
White indigo,
138.
Wince dye beck,
53,
54.
Woad,
138.
---- indigo vats,
139.
---- vat,
145.
Woaded black,
86.
Wool,
1.
---- action of acid on,
11.
---- alkalies, action of on,
9.
---- batching,
15.
---- black,
89.
---- ----- 6 B,
171,
180,
181,
186.
---- bleaching,
29.
---- --------- peroxide of hydrogen,
34.
---- --------- -------- of soda,
36.
---- --------- with sulphur,
30.
---- chemical composition of,
6.
---- chlorination of,
37.
---- blue B X,
153.
---- ---- dyeing with logwood,
161.
---- fibre under microscope,
2.
---- ----- unscoured,
10.
---- ----- chemical composition of,
7.
---- ----- scoured badly,
10.
---- ----- showing medullary centre,
4.
--- ----- heated with acid,
11.
---- grey R,
166.
Woollen piece goods, scouring of,
28.
------- yarn,
2.
Wool oil,
26.
Wool, physical properties of,
2.
---- -------- structure, variations in,
5.
Wool scouring,
15,
17.
---- -------- by solvents,
23.
Wool-washing machine,
20,
21.
Worsted yarn,
2.
Wringing of goods,
197.
Y.
Yarn-drying machine,
208.
Yarn, washing of, in hanks,
202.
Yarn wringing,
198.
Yellow,
125,
195.
------ brown,
161.
------ N,
125,
133,
134.
------ olive,
135.
------ shades on wool,
123.
Yolk,
7.
Yorkshire grease,
26.
Z.
Zambesi black B,
170,
181.
------- ----- D,
170,
180,
181.
------- ----- F,
171.
------- blue R A,
180,
181.
------- brown G,
171,
181.
------- ----- 2 G,
171.
------- dyes,
168.
Zinc dust,
141.
Agricultural Chemistry, 10.
Air, Industrial Use of, 12.
Alum and its Sulphates, 9.
Ammonia, 9.
Aniline Colours, 3.
Animal Fats, 6.
Anti-corrosive Paints, 4.
Architecture, Terms in, 30.
Architectural Pottery, 15.
Artificial Perfumes, 7.
Balsams, 10.
Bibliography, 32.
Bleaching, 23.
Bone Products, 8.
Bookbinding, 31.
Brick-making, 15,
16.
Burnishing Brass, 28.
Carpet Yarn Printing, 21.
Ceramic Books, 14,
15.
Charcoal, 8.
Chemical Essays, 9.
Chemistry of Pottery, 16.
Chemistry of Dye-stuffs, 23.
Clay Analysis, 16.
Coal-dust Firing, 26.
Colour Matching, 22.
Colliery Recovery Work, 25.
Colour-mixing for Dyers, 22.
Colour Theory, 22.
Combing Machines, 24.
Compounding Oils, 6.
Condensing Apparatus, 26.
Cosmetics, 8.
Cotton Dyeing, 23.
Cotton Spinning, 24.
Damask Weaving, 20.
Dampness in Buildings, 30.
Decorators' Books, 28.
Decorative Textiles, 20.
Dental Metallurgy, 25.
Dictionary of Paint Materials, 2.
Drying Oils, 5.
Drying with Air, 12.
Dyeing Marble, 31.
Dyeing Woollen Fabrics, 23.
Dyers' Materials, 22.
Dye-stuffs, 23.
Enamelling Metal, 18.
Enamels, 18.
Engraving, 31.
Essential Oils, 7.
Evaporating Apparatus, 26.
External Plumbing, 27.
Fats, 5, 6.
Faults in Woollen Goods, 21.
Gas Firing, 26.
Glass-making Recipes, 16.
Glass Painting, 17.
Glue Making and Testing, 8.
Greases, 5.
Hat Manufacturing, 20.
History of Staffs Potteries, 16.
Hops, 28.
Hot-water Supply, 28.
How to make a Woollen Mill Pay, 21.
India-rubber, 13.
Industrial Alcohol, 10.
Inks, 3, 11.
Iron-corrosion, 4.
Iron, Science of, 26.
Japanning, 28.
Lace-Making, 20.
Lacquering, 28.
Lake Pigments, 2.
Lead and its Compounds, 11.
Leather Industry, 13.
Leather-working Materials, 14.
Lithography, 31.
Lubricants, 5, 6.
Manures, 8, 10.
Mineral Pigments, 3.
Mine Ventilation, 25.
Mine Haulage, 25.
Oil and Colour Recipes, 3.
Oil Boiling, 5.
Oil Merchants' Manual, 7.
Oils, 5.
Ozone, Industrial Use of, 12.
Paint Manufacture, 2.
Paint Materials, 3.
Paint-material Testing, 4.
Paper-pulp Dyeing, 17.
Petroleum, 6.
Pigments, Chemistry of, 2.
Plumbers' Work, 27.
Porcelain Painting, 17.
Pottery Clays, 16.
Pottery Manufacture, 14.
Power-loom Weaving, 19.
Preserved Foods, 30.
Printers' Ready Reckoner, 31.
Printing Inks, 3.
Recipes for Oilmen, etc., 3.
Resins, 10.
Risks of Occupations, 11.
Riveting China, etc., 16.
Sanitary Plumbing, 27.
Sealing Waxes, 11.
Silk Dyeing, 22.
Silk Throwing, 18.
Smoke Prevention, 26.
Soaps, 7.
Spinning, 21.
Staining Marble, and Bone, 31.
Steam Drying, 12.
Sugar Refining, 32.
Steel Hardening, 26.
Sweetmeats, 30.
Terra-cotta, 15.
Testing Paint Materials, 4.
Testing Yarns, 20.
Textile Fabrics, 20.
Textile Materials, 19, 20.
Timber, 29.
Varnishes, 5.
Vegetable Fats, 7.
Waste Utilisation, 10.
Water, Industrial Use, 12.
Waterproofing Fabrics, 21.
Weaving Calculations, 21.
Wood Waste Utilisation, 29.
Wood Dyeing, 31.
Wool Dyeing, 22, 23.
Writing Inks, 11.
X-Ray Work, 13.
Yarn Testing, 20.
THE CHEMISTRY OF PIGMENTS. By Ernest J. Parry, B.Sc. (Lond.), F.I.C., F.C.S., and J. H. Coste, F.I.C., F.C.S. Demy 8vo. Five Illustrations. 285 pp. Price 10s. 6d. net. (Post free, 10s. 10d. home; 11s. 3d. abroad.)
Contents.
Introductory. Light -- White Light -- The Spectrum -- The Invisible Spectrum -- Normal Spectrum -- Simple Nature of Pure Spectral Colour -- The Recomposition of White Light -- Primary and Complementary Colours -- Coloured Bodies -- Absorption Spectra -- The Application of Pigments. Uses of Pigments: Artistic, Decorative, Protective -- Methods of Application of Pigments: Pastels and Crayons, Water Colour, Tempera Painting, Fresco, Encaustic Painting, Oil-colour Painting, Keramic Art, Enamel, Stained and Painted Glass, Mosaic -- Inorganic Pigments. White Lead -- Zinc White -- Enamel White -- Whitening -- Red Lead -- Litharge -- Vermilion -- Royal Scarle t-- The Chromium Greens -- Chromates of Lead, Zinc, Silver and Mercury -- Brunswick Green -- The Ochres -- Indian Red -- Venetian Red -- Siennas and Umbers -- Light Red -- Cappagh Brown -- Red Oxides -- Mars Colours -- Terre Verte -- Prussian Brown -- Cobalt Colours -- Coeruleum -- Smalt -- Copper Pigments -- Malachite -- Bremen Green -- Scheele's Green -- Emerald Green -- Verdigris -- Brunswick Green -- Non-arsenical Greens -- Copper Blues -- Ultramarine -- Carbon Pigments -- Ivory Black -- Lamp Black -- Bistre -- Naples Yellow -- Arsenic Sulphides: Orpiment, Realgar -- Cadmium Yellow -- Vandyck Brown -- Organic Pigments. Prussian Blue -- Natural Lakes -- Cochineal -- Carmine -- Crimson -- Lac Dye -- Scarlet -- Madder -- Alizarin -- Campeachy -- Quercitron -- Rhamnus -- Brazil Wood -- Alkanet -- Santal Wood -- Archil -- Coal-tar Lakes -- Red Lakes -- Alizarin Compounds -- Orange and Yellow Lakes -- Green and Blue Lakes -- Indigo -- Dragon's Blood -- Gamboge -- Sepia -- Indian Yellow, Puree -- Bitumen, Asphaltum, Mummy -- Index.
THE MANUFACTURE OF PAINT. A Practical Handbook for Paint Manufacturers, Merchants and Painters. By J. Cruickshank Smith, B.Sc. Demy 8vo. 200 pp. Sixty Illustrations and One Large Diagram. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Preparation of Raw Material -- Storing of Raw Material -- Testing and Valuation of Raw Material -- Paint Plant and Machinery -- The Grinding of White Lead -- Grinding of White Zinc -- Grinding of other White Pigments -- Grinding of Oxide Paints -- Grinding of Staining Colours -- Grinding of Black Paints -- Grinding of Chemical Colours -- Yellows -- Grinding of Chemical Colours -- Blues -- Grinding Greens -- Grinding Reds -- Grinding Lakes -- Grinding Colours in Water -- Grinding Colours in Turpentine -- The Uses of Paint -- Testing and Matching Paints -- Economic Considerations -- Index.
DICTIONARY OF CHEMICALS AND RAW PRODUCTS USED IN THE MANUFACTURE OF PAINTS, COLOURS, VARNISHES AND ALLIED PREPARATIONS. By George H. Hurst, F.C.S. Demy 8vo. 380 pp. Price 7s. 6d. net. (Post free, 8s. home; 8s. 6d. abroad.)
THE MANUFACTURE OF LAKE PIGMENTS FROM ARTIFICIAL COLOURS. By Francis H. Jennison, F.I.C., F.C.S. Sixteen Coloured Plates, showing Specimens of Eighty-nine Colours, specially prepared from the Recipes given in the Book. 136 pp. Demy 8vo. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
The Groups of the Artificial Colouring Matters -- The Nature and Manipulation of Artificial Colours -- Lake-forming Bodies for Acid Colours -- Lake-forming Bodies' Basic Colours -- Lake Bases -- The Principles of Lake Formation -- Red Lakes -- Orange, Yellow, Green, Blue, Violet and Black Lakes -- The Production of Insoluble Azo Colours in the Form of Pigments -- The General Properties of Lakes Produced from Artificial Colours -- Washing, Filtering and Finishing -- Matching and Testing Lake Pigments -- Index.
THE MANUFACTURE OF MINERAL AND LAKE (p. c03) PIGMENTS. Containing Directions for the Manufacture of all Artificial, Artists and Painters' Colours, Enamel, Soot and Metallic Pigments. A Text-book for Manufacturers, Merchants, Artists and Painters. By Dr. Josef Bersch. Translated by A. C. Wright, M.A. (Oxon.), B.Sc. (Lond.). Forty-three Illustrations. 476 pp., demy 8vo. Price 12s. 6d. net. (Post free, 13s. home; 13s. 6d. abroad.)
Contents.
Introduction -- Physico-chemical Behaviour of Pigments -- Raw Materials Employed in the Manufacture of Pigments -- Assistant Materials -- Metallic Compounds -- The Manufacture of Mineral Pigments -- The Manufacture of White Lead -- Enamel White -- Washing Apparatus -- Zinc White -- Yellow Mineral Pigments -- Chrome Yellow -- Lead Oxide Pigments -- Other Yellow Pigments -- Mosaic Gold -- Red Mineral Pigments -- The Manufacture of Vermilion -- Antimony Vermilion -- Ferric Oxide Pigments -- Other Red Mineral Pigments -- Purple of Cassius -- Blue Mineral Pigments -- Ultramarine -- Manufacture of Ultramarine -- Blue Copper Pigments -- Blue Cobalt Pigments -- Smalts -- Green Mineral Pigments -- Emerald Green -- Verdigris -- Chromium Oxide -- Other Green Chromium Pigments -- Green Cobalt Pigments -- Green Manganese Pigments -- Compounded Green Pigments -- Violet Mineral Pigments -- Brown Mineral Pigments -- Brown Decomposition Products -- Black Pigments -- Manufacture of Soot Pigments -- Manufacture of Lamp Black -- The Manufacture of Soot Black without Chambers -- Indian Ink -- Enamel Colours -- Metallic Pigments -- Bronze Pigments -- Vegetable Bronze Pigments.
Pigments of Organic Origin -- Lakes -- Yellow Lakes -- Red Lakes -- Manufacture of Carmine -- The Colouring Matter of Lac -- Safflower or Carthamine Red -- Madder and its Colouring Matters -- Madder Lakes -- Manjit (Indian Madder) -- Lichen Colouring Matters -- Red Wood Lakes -- The Colouring Matters of Sandal Wood and Other Dye Woods -- Blue Lakes -- Indigo Carmine -- The Colouring Matter of Log Wood -- Green Lakes -- Brown Organic Pigments -- Sap Colours -- Water Colours -- Crayons -- Confectionery Colours -- The Preparation of Pigments for Painting -- The Examination of Pigments -- Examination of Lakes -- The Testing of Dye-Woods -- The Design of a Colour Works -- Commercial Names of Pigments -- Appendix: Conversion of Metric to English Weights and Measures -- Centigrade and Fahrenheit Thermometer Scales -- Index.
RECIPES FOR THE COLOUR, PAINT, VARNISH, OIL, SOAP AND DRYSALTERY TRADES. Compiled by An Analytical Chemist. 350 pp. Demy 8vo. Price 7s. 6d. net. (Post free, 8s. home; 8s. 3d. abroad.)
Contents.
Pigments or Colours for Paints, Lithographic and Letterpress Printing Inks, etc. -- Mixed Paints and Preparations for Paint-making, Painting, Lime-washing, Paperhanging, etc. -- Varnishes for Coach-builders, Cabinetmakers, Wood-workers, Metal-workers, Photographers, etc. -- Soaps for Toilet, Cleansing, Polishing, etc. -- Perfumes -- Lubricating Greases, Oils, etc. -- Cements, Pastes, Glues and Other Adhesive Preparations -- Writing, Marking, Endorsing and Other Inks -- Sealing-wax and Office Requisites -- Preparations for the Laundry, Kitchen, Stable and General Household Uses -- Disinfectant Preparations -- Miscellaneous Preparations -- Index.
OIL COLOURS AND PRINTERS' INKS. By Louis Edgar Andés. Translated from the German. 215 pp. Crown 8vo. 56 Illustrations. Price 5s. net. (Post free, 5s. 4d. home; 5s. 6d. abroad.)
Contents.
Linseed Oil -- Poppy Oil -- Mechanical Purification of Linseed Oil -- Chemical Purification of Linseed Oil -- Bleaching Linseed Oil -- Oxidizing Agents for Boiling Linseed Oil -- Theory of Oil Boiling -- Manufacture of Boiled Oil -- Adulterations of Boiled Oil -- Chinese Drying Oil and Other Specialities -- Pigments for House and Artistic Painting and Inks -- Pigment for Printers' Black Inks -- Substitutes for Lampblack -- Machinery for Colour Grinding and Rubbing -- Machines for mixing Pigments with the Vehicle -- Paint Mills -- Manufacture of House Oil Paints -- Ship Paints -- Luminous Paint -- Artists' Colours -- Printers' Inks: -- VEHICLES -- Printers' Inks: -- PIGMENTS and MANUFACTURE -- Index.
(See also Writing Inks.)
THREE HUNDRED SHADES FOR DECORATORS AND HOW TO MIX THEM.
(See page 28.)
CASEIN. (p. c04) By Robert Scherer. Translated from the German by Chas. Salter. Demy 8vo. Illustrated. 160 pp. Price 7s. 6d. net, (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Casein: its Origin, Preparation and Properties. Various Methods of Preparing Casein. Composition and Properties of Casein. Casein Paints. -- "Marble-Lime" Colour for Outside Work -- Casein Enamel Paint -- Casein Façade Paint -- Cold-Water Paint in Powder Form -- Kistory's Recipe for Casein Paint and Varnish -- Pure Casein Paints for Walls, etc. -- Casein Paints for Woodwork and Iron -- Casein-Silicate Paints -- Milk Paints -- Casein-Silicate Paint Recipes -- Trojel's Boiled Oil Substitute -- Calsomine Wash -- Quick-Drying Casein Paint -- Boiled Oil Substitute -- Ring's Cold-Water Paint -- Formo-lactin -- Waterproof Paint for Playing Cards -- Casein Colour Lake -- Casein-Cement Paint. The Technics of Casein Painting. Casein Adhesives and Putties. -- Casein Glue in Plates or Flakes -- Jeromin's Casein Adhesive -- Hall's Casein Glue -- Waterproof Glue -- Liquid Casein Glue -- Casein and Borax Glue -- Solid Casein Adhesive -- Casein Solution -- Glue Powder -- Casein Putties -- Washable Cement for Deal Boards -- Wenk's Casein Cement -- Casein and Lime Cement "Pitch Barm" -- Casein Stopping -- Casein Cement for Stone. The Preparation of Plastic Masses from Casein. -- Imitation Ivory -- Anti-Radiation and Anti-Corrosive Composition -- Dickmann's Covering for Floors and Walls -- Imitation Linoleum -- Imitation Leather -- Imitation Bone -- Plastic Mass of Keratin and Casein -- Insulating Mass -- Plastic Casein Masses -- Horny Casein Mass -- Plastic Mass from Celluloid -- Casein Cellulose Composition -- Fire-proof Cellulose Substitute -- Nitrocellulose and Casein Composition -- Franquet's Celluloid Substitute -- Galalith. Uses of Casein in the Textile Industry, for Finishing Colour Printing, etc. -- Caseogum -- "Glutin" -- Casein Dressing for Linen and Cotton Fabrics -- Printing Colour with Metallic Lustre -- Process for Softening, Sizing and Loading -- Fixing Casein and Other Albuminoids on the Fibre -- Fixing Insoluble Colouring Matters -- Waterproofing and Softening Dressing -- Casein for Mercerising Crèpe -- Fixing Zinc White on Cotton with Formaldehyde -- Casein-Magnesia -- Casein Medium for Calico Printing -- Loading Silk. Casein Foodstuffs. -- Casein Food -- Synthetic Milk -- Milk Food -- Emulsifiable Casein -- Casein Phosphate for Baking -- Making Bread, Low in Carbohydrates, from Flour and Curd -- Preparing Soluble Casein Compounds with Citrates -- Casein Food. Sundry Applications of Casein. -- Uses of Casein in the Paper Industry -- Metachromotype Paper -- Sizing Paper with Casein -- Waterproofing Paper -- Casein Solution for Coating Paper -- Horn's Clear Solution of Casein -- Water- and Fire-proof Asbestos Paper and Board -- Paper Flasks, etc., for Oils and Fats -- Washable Drawing and Writing Paper--Paper Wrappering for Food, Clothing, etc. -- Paint Remover -- Casein Photographic Plates -- Wood-Cement Roofing Pulp -- Cask Glaze of Casein and Formaldehyde -- Artists' Canvas -- Solidifying Mineral Oils -- Uses of Casein in Photography -- Casein Ointment -- Clarifying Glue with Casein -- Casein in Soap-making -- Casein-Albumose Soap -- Casein in Sheets, Blocks, etc. -- Waterproof Casein.
SIMPLE METHODS FOR TESTING PAINTERS' MATERIALS. By A. C. Wright, M.A. (Oxon.), B.Sc. (Lond.). Crown 8vo. 160 pp. Price 5s. net. (Post free, 5s. 3d. home; 5s. 6d. abroad.)
Contents. Necessity for Testing -- Standards -- Arrangement -- The Apparatus -- The Reagents -- Practical Tests -- Dry Colours -- Stiff Paints -- Liquid and Enamel Paints -- Oil Varnishes -- Spirit Varnishes -- Driers -- Putty -- Linseed Oil -- Turpentine -- Water Stains -- The Chemical Examination -- Dry Colours and Paints -- White Pigments and Paints -- Yellow Pigments and Paints -- Blue Pigments and Paints -- Green Pigments and Paints -- Red Pigments and Paints -- Brown Pigments and Paints -- Black Pigments and Paints -- Oil Varnishes -- Linseed Oil -- Turpentine.
IRON-CORROSION, ANTI-FOULING AND ANTI-CORROSIVE PAINTS. Translated from the German of Louis Edgar Andés. Sixty-two Illustrations. 275 pp. Demy 8vo. Price 10s. 6d. net. (Post free, 10s. 10d. home; 11s. 3d. abroad.)
Contents.
Iron-rust and its Formation -- Protection from Rusting by Paint -- Grounding the Iron with Linseed Oil, etc. -- Testing Paints -- Use of Tar for Painting on Iron -- Anti-corrosive Paints -- Linseed Varnish -- Chinese Wood Oil -- Lead Pigments -- Iron Pigments -- Artificial Iron Oxides -- Carbon -- Preparation of Anti-corrosive Paints -- Results of Examination of Several Anti-corrosive Paints -- Paints for Ship's Bottoms -- Anti-fouling Compositions -- Various Anti-corrosive and Ship's Paints -- Official Standard Specifications for Ironwork Paints -- Index.
THE TESTING AND VALUATION OF RAW MATERIALS USED IN PAINT AND COLOUR MANUFACTURE. By M. W. Jones, F.C.S. A Book for the Laboratories of Colour Works. 88 pp. Crown 8vo. Price 5s. net. (Post free, 5s. 3d. home and abroad.)
Aluminium Compounds -- China Clay -- Iron Compounds -- Potassium Compounds -- Sodium Compounds -- Ammonium Hydrate -- Acids -- Chromium Compounds -- Tin Compounds -- Copper Compounds -- Lead Compounds -- Zinc Compounds -- Manganese Compounds -- Arsenic Compounds -- Antimony Compounds -- Calcium Compounds -- Barium Compounds -- Cadmium Compounds -- Mercury Compounds -- Ultramarine -- Cobalt and Carbon Compounds -- Oils -- Index.
STUDENTS' HANDBOOK OF PAINTS, COLOURS, OILS AND VARNISHES. By John Furnell. Crown 8vo. 12 Illustrations. 96 pp. Price 2s. 6d. net. (Post free, 2s. 9d. home and abroad.)
Contents.
Plant -- Chromes -- Blues -- Greens -- Earth Colours -- Blacks -- Reds -- Lakes -- Whites -- Painters' Oils -- Turpentine -- Oil Varnishes -- Spirit Varnishes -- Liquid Paints -- Enamel Paints.
OIL CRUSHING, REFINING AND BOILING, THE MANUFACTURE OF LINOLEUM, PRINTING AND LITHOGRAPHIC INKS, AND INDIA-RUBBER SUBSTITUTES. By John Geddes McIntosh. Being Volume I. of the Second, greatly enlarged, English Edition, in three Volumes, of "The Manufacture of Varnishes and Kindred Industries," based on and including the work of Ach. Livache. Demy 8vo. 150 pp. 29 Illustrations. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Oil Crushing and Refining; Oil Boiling -- Theoretical and Practical; Linoleum Manufacture; Printing Ink Manufacture; Rubber Substitutes; The Manufacture of Driers; The Detection of Adulteration in Linseed and other Drying Oils by Chemical, Physical and Organoleptic Methods.
DRYING OILS, BOILED OIL AND SOLID AND LIQUID DRIERS. By L. E. Andés. Expressly Written for this Series of Special Technical Books, and the Publishers hold the Copyright for English and Foreign Editions. Forty-two Illustrations. 342 pp. Demy 8vo. Price 12s. 6d. net. (Post free, 13s. home; 13s. 3d. abroad.)
Contents.
Properties of the Drying Oils; Cause of the Drying Property; Absorption of Oxygen; Behaviour towards Metallic Oxides, etc. -- The Properties of and Methods for obtaining the Drying Oils -- Production of the Drying Oils by Expression and Extraction; Refining and Bleaching; Oil Cakes and Meal; The Refining and Bleaching of the Drying Oils; The Bleaching of Linseed Oil -- The Manufacture of Boiled Oil; The Preparation of Drying Oils for Use in the Grinding of Paints and Artists' Colours and in the Manufacture of Varnishes by Heating over a Fire or by Steam, by the Cold Process, by the Action of Air, and by Means of the Electric Current; The Driers used in Boiling Linseed Oil; The Manufacture of Boiled Oil and the Apparatus therefor; Livache's Process for Preparing a Good Drying Oil and its Practical Application -- The Preparation of Varnishes for Letterpress, Lithographic and Copperplate Printing, for Oilcloth and Waterproof Fabrics; The Manufacture of Thickened Linseed Oil, Burnt Oil, Stand Oil by Fire Heat, Superheated Steam, and by a Current of Air -- Behaviour of the Drying Oils and Boiled Oils towards Atmospheric Influences, Water, Acids and Alkalies -- Boiled Oil Substitutes -- The Manufacture of Solid and Liquid Driers from Linseed Oil and Rosin; Linolic Acid Compounds of the Driers -- The Adulteration and Examination of the Drying Oils and Boiled Oil.
LUBRICATING OILS, FATS AND GREASES: Their Origin, Preparation, Properties, Uses and Analyses. A Handbook for Oil Manufacturers, Refiners and Merchants, and the Oil and Fat Industry in General. By George H. Hurst, F.C.S. Second Revised and Enlarged Edition. Sixty-five Illustrations. 317 pp. Demy 8vo. Price 10s. 6d. net. (Post free, 11s. home; 11s. 3d. abroad.)
Contents.
Introductory -- Hydrocarbon Oils -- Scotch Shale Oils -- Petroleum -- Vegetable and Animal Oils -- Testing and Adulteration of Oils -- Lubricating Greases -- Lubrication -- Appendices -- Index.
TECHNOLOGY (p. c06) OF PETROLEUM: Oil Fields of the World -- Their History, Geography and Geology -- Annual Production and Development -- Oil-well Drilling -- Transport. By Henry Neuberger and Henry Noalhat. Translated from the French by J. G. McIntosh. 550 pp. 153 Illustrations. 26 Plates. Super Royal 8vo. Price 21s. net. (Post free, 21s. 9d. home; 23s. 6d. abroad.)
Contents.
Study of the Petroliferous Strata.
Excavations--Hand Excavation or Hand Digging of Oil Wells.
Methods of Boring.
Accidents -- Boring Accidents -- Methods of preventing them -- Methods of remedying them -- Explosives and the use of the "Torpedo" Levigation -- Storing and Transport of Petroleum -- General Advice -- Prospecting, Management and carrying on of Petroleum Boring Operations.
General Data -- Customary Formulæ -- Memento. Practical Part. General Data bearing on Petroleum -- Glossary of Technical Terms used in the Petroleum Industry -- Copious Index.
THE PRACTICAL COMPOUNDING OF OILS, TALLOW AND GREASE FOR LUBRICATION, ETC. By An Expert Oil Refiner. 100 pp. Demy 8vo. Price 7s. 6d. net. (Post free. 7s. 10d. home; 8s. abroad.)
Contents.
Introductory Remarks on the General Nomenclature of Oils, Tallow and Greases suitable for Lubrication -- Hydrocarbon Oils -- Animal and Fish Oils -- Compound Oils -- Vegetable Oils -- Lamp Oils -- Engine Tallow, Solidified Oils and Petroleum Jelly -- Machinery Greases: Loco and Anti-friction -- Clarifying and Utilisation of Waste Fats, Oils, Tank Bottoms, Drainings of Barrels and Drums, Pickings Up, Dregs, etc. -- The Fixing and Cleaning of Oil Tanks, etc. -- Appendix and General Information.
ANIMAL FATS AND OILS: Their Practical Production, Purification and Uses for a great Variety of Purposes. Their Properties, Falsification and Examination. Translated from the German of Louis Edgar Andés. Sixty-two Illustrations. 240 pp. Second Edition, Revised and Enlarged. Demy 8vo. Price 10s. 6d. net. (Post free, 10s. 10d. home; 11s. 3d. abroad.)
Contents.
Introduction -- Occurrence, Origin, Properties and Chemical Constitution of Animal Fats -- Preparation of Animal Fats and Oils -- Machinery -- Tallow-melting Plant -- Extraction Plant -- Presses -- Filtering Apparatus -- Butter: Raw Material and Preparation, Properties, Adulterations, Beef Lard or Remelted Butter, Testing -- Candle-fish Oil -- Mutton-Tallow -- Hare Fat -- Goose Fat -- Neatsfoot Oil -- Bone Fat: Bone Boiling, Steaming Bones, Extraction, Refining -- Bone Oil -- Artificial Butter: Oleomargarine, Margarine Manufacture in France, Grasso's Process, "Kaiser's Butter," Jahr & Münzberg's Method, Filbert's Process, Winter's Method -- Human Fat -- Horse Fat -- Beef Marrow -- Turtle Oil -- Hog's Lard: Raw Material -- Preparation, Properties, Adulterations, Examination -- Lard Oil -- Fish Oils -- Liver Oils -- Artificial Train Oil -- Wool Fat: Properties, Purified Wool Fat -- Spermaceti: Examination of Fats and Oils in General.
THE MANUFACTURE OF LUBRICANTS, SHOE POLISHES AND LEATHER DRESSINGS. By Richard Brunner. Translated from the Sixth German Edition by Chas. Salter. 10 Illustrations. Crown 8vo. 170 pp. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
The Manufacture of Lubricants and Greases -- Properties of the Bodies used as Lubricants -- Raw Materials for Lubricants -- Solid Lubricants -- Tallow Lubricants -- Palm Oil Greases -- Lead Soap Lubricants -- True Soap Greases -- Caoutchouc Lubricants -- Other Solid Lubricants -- Liquid Lubricants -- Lubricating Oils in General -- Refining Oils for Lubricating Purposes -- Cohesion Oils -- Resin Oils -- Lubricants of Fat and Resin Oil -- Neatsfoot Oil -- Bone Fat -- Lubricants for Special Purposes -- Mineral Lubricating Oils -- Clockmakers' and Sewing Machine Oils -- The Application of Lubricants to Machinery -- Removing Thickened Grease and Oil -- Cleaning Oil Rags and Cotton Waste -- The Use of Lubricants -- Shoe Polishes and Leather Softening Preparations -- The Manufacture of Shoe Polishes and Preparations for Varnishing and Softening Leather -- The Preparation of Bone Black -- Blacking and Shoe Polishes -- Leather Varnishes -- Leather Softening Preparations -- The Manufacture of Dégras.
THE (p. c07) OIL MERCHANTS' MANUAL AND OIL TRADE READY RECKONER. Compiled by Frank P. Sherriff. Second Edition Revised and Enlarged. Demy 8vo. 214 pp. 1904. With Two Sheets of Tables. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. 3d. abroad.)
Contents.
Trade Terms and Customs -- Tables to Ascertain Value of Oil sold per cwt. or ton -- Specific Gravity Tables -- Percentage Tare Tables -- Petroleum Tables -- Paraffine and Benzoline Calculations -- Customary Drafts -- Tables for Calculating Allowance for Dirt, Water, etc. -- Capacity of Circular Tanks Tables, etc., etc.
VEGETABLE FATS AND OILS: Their Practical Preparation. Purification and Employment for Various Purposes, their Properties, Adulteration and Examination. Translated from the German of Louis Edgar Andés. Ninety-four Illustrations. 340 pp. Second Edition. Demy 8vo. Price 10s. 6d. net. (Post free, 11s. home; 11s. 6d. abroad.)
Contents.
General Properties -- Estimation of the Amount of Oil in Seeds -- The Preparation of Vegetable Fats and Oils -- Apparatus for Grinding Oil Seeds and Fruits -- Installation of Oil and Fat Works -- Extraction Method of Obtaining Oils and Fats -- Oil Extraction Installations -- Press Moulds -- Non-drying Vegetable Oils -- Vegetable drying Oils -- Solid Vegetable Fats -- Fruits Yielding Oils and Fats -- Wool-softening Oils -- Soluble Oils -- Treatment of the Oil after Leaving the Press -- Improved Methods of Refining -- Bleaching Fats and Oils -- Practical Experiments on the Treatment of Oils with regard to Refining and Bleaching -- Testing Oils and Fats.
THE CHEMISTRY OF ESSENTIAL OILS AND ARTIFICIAL PERFUMES. By Ernest J. Parry, B.Sc. (Lond.), F.I.C., F.C.S. 411 pp. 20 Illustrations. Demy 8vo. Price 12s. 6d. net. (Post free, 13s. home; 13s. 6d. abroad.)
Contents.
The General Properties of Essential Oils -- Compounds occurring in Essential Oils -- The Preparation of Essential Oils -- The Analysis of Essential Oils -- Systematic Study of the Essential Oils -- Terpeneless Oils -- The Chemistry of Artificial Perfumes -- Appendix: Table of Constants -- Index.
SOAPS. A Practical Manual of the Manufacture of Domestic, Toilet and other Soaps. By George H. Hurst, F.C.S. 390 pp. 66 Illustrations. Price 12s. 6d. net. (Post free, 13s. home; 13s. 6d. abroad.)
Contents.
Introductory -- Soap-maker's Alkalies -- Soap Fats and Oils -- Perfumes -- Water as a Soap Material -- Soap Machinery -- Technology of Soap-making -- Glycerine in Soap Lyes -- Laying out a Soap Factory -- Soap Analysis -- Appendices.
TEXTILE SOAPS AND OILS. Handbook on the Preparation, Properties and Analysis of the Soaps and Oils used in Textile Manufacturing, Dyeing and Printing. By George H. Hurst, F.C.S. Crown 8vo. 195 pp. 1904. Price 5s. net. (Post free, 5s. 4d. home; 5s. 6d. abroad.)
Contents.
Methods of Making Soaps -- Hard Soap -- Soft Soap. Special Textile Soaps -- Wool Soaps -- Calico Printers' Soaps -- Dyers' Soaps. Relation of Soap to Water for Industrial Purposes -- Treating Waste Soap Liquors -- Boiled Off Liquor -- Calico Printers and Dyers' Soap Liquors -- Soap Analysis -- Fat in Soap.
ANIMAL AND VEGETABLE OILS AND FATS -- Tallow -- Lard -- Bone Grease-Tallow Oil. Vegetable Soap, Oils and Fats -- Palm Oil -- Coco-nut Oil -- Olive Oil -- Cottonseed Oil -- Linseed Oil -- Castor Oil -- Corn Oil -- Whale Oil or Train Oil -- Repe Oil.
GLYCERINE.
TEXTILE OILS -- Oleic Acid -- Blended Wool Oils -- Oils for Cotton Dyeing, Printing and Finishing -- Turkey Red Oil -- Alizarine Oil -- Oleine -- Oxy Turkey Red Oils -- Soluble Oil-Analysis of Turkey Red Oil -- Finisher's Soluble Oil -- Finisher's Soap Softening -- Testing and Adulteration of Oils -- Index.
COSMETICS: MANUFACTURE, EMPLOYMENT AND TESTING OF ALL COSMETIC MATERIALS AND COSMETIC SPECIALITIES. Translated from the German of Dr. Theodor Koller. Crown 8vo. 262 pp. Price 5s. net. (Post free, 5s. 4d. home; 5s. 6d. abroad.)
Contents.
Purposes and Uses of, and Ingredients used in the Preparation of Cosmetics -- Preparation of Perfumes by Pressure, Distillation, Maceration, Absorption or Enfleurage, and Extraction Methods -- Chemical and Animal Products used in the Preparation of Cosmetics -- Oils and Fats used in the Preparation of Cosmetics -- General Cosmetic Preparations -- Mouth Washes and Tooth Pastes -- Hair Dyes, Hair Restorers and Depilatories -- Cosmetic Adjuncts and Specialities -- Colouring Cosmetic Preparations -- Antiseptic Washes and Soaps -- Toilet and Hygienic Soaps -- Secret Preparations for Skin, Complexion, Teeth, Mouth, etc. -- Testing and Examining the Materials Employed in the Manufacture of Cosmetics -- Index.
GLUE AND GLUE TESTING. By Samuel Rideal, D.Sc. (Lond.), F.I.C. Fourteen Engravings. 144 pp. Demy 8vo. Price 10s. 6d. net. (Post free, 10s. 10d. home; 11s. abroad.)
Contents.
Constitution and Properties: Definitions and Sources, Gelatine, Chondrin and Allied Bodies, Physical and Chemical Properties, Classification, Grades and Commercial Varieties -- Raw Materials and Manufacture: Glue Stock, Lining, Extraction, Washing and Clarifying, Filter Presses, Water Supply, Use of Alkalies, Action of Bacteria and of Antiseptics, Various Processes, Cleansing, Forming, Drying, Crushing, etc., Secondary Products -- Uses of Glue: Selection and Preparation for Use, Carpentry, Veneering, Paper-Making, Bookbinding, Printing Rollers, Hectographs, Match Manufacture, Sandpaper, etc., Substitutes for other Materials, Artificial Leather and Caoutchouc -- Gelatine: General Characters, Liquid Gelatine, Photographic Uses, Size, Tanno-, Chrome and Formo-Gelatine, Artificial Silk, Cements, Pneumatic Tyres, Culinary, Meat Extracts, Isinglass, Medicinal and other Uses, Bacteriology -- Glue Testing: Review of Processes, Chemical Examination, Adulteration, Physical Tests, Valuation of Raw Materials -- Commercial Aspects.
BONE PRODUCTS AND MANURES: An Account of the most recent Improvements in the Manufacture of Fat, Glue, Animal Charcoal, Size, Gelatine and Manures. By Thomas Lambert, Technical and Consulting Chemist. Illustrated by Twenty-one Plans and Diagrams. 162 pp. Demy 8vo. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Chemical Composition of Bones -- Arrangement of Factory -- Properties of Glue -- Glutin and Chondrin -- Skin Glue -- Liming of Skins -- Washing -- Boiling of Skins -- Clarification of Glue Liquors -- Glue-Boiling and Clarifying-House -- Specification of a Glue -- Size -- Uses and Preparation and Composition of Size -- Concentrated Size -- Properties of Gelatine -- Preparation of Skin Gelatine -- Drying -- Bone Gelatine -- Selecting Bones -- Crushing -- Dissolving -- Bleaching -- Boiling -- Properties of Glutin and Chondrin -- Testing of Glues and Gelatines -- The Uses of Glue, Gelatine and Size in Various Trades -- Soluble and Liquid Glues -- Steam and Waterproof Glues -- Manures -- Importation of Food Stuffs -- Soils -- Germination -- Plant Life -- Natural Manures -- Water and Nitrogen in Farmyard Manure -- Full Analysis of Farmyard Manure -- Action on Crops -- Water-Closet System -- Sewage Manure -- Green Manures -- Artificial Manures -- Mineral Manures -- Nitrogenous Matters -- Shoddy -- Hoofs and Horns -- Leather Waste -- Dried Meat -- Dried Blood -- Superphosphates -- Composition -- Manufacture -- Common Raw Bones -- Degreased Bones -- Crude Fat -- Refined Fat -- Degelatinised Bones -- Animal Charcoal -- Bone Superphosphates -- Guanos -- Dried Animal Products -- Potash Compounds -- Sulphate of Ammonia -- Extraction in Vacuo -- French and British Gelatines compared -- Index.
REISSUE OF CHEMICAL ESSAYS OF C. W. SCHEELE. First Published in English in 1786. Translated from the Academy of Sciences at Stockholm, with Additions. 300 pp. Demy 8vo, Price 5s. net. (Post free, 5s. 6d. home; 5s. 9d. abroad.)
Contents.
Memoir: C. W. Scheele and his work (written for this edition by J. G. McIntosh) -- On Fluor Mineral and its Acid -- On Fluor Mineral -- Chemical Investigation of Fluor Acid, with a View to the Earth which it Yields, by Mr. Wiegler -- Additional Information Concerning Fluor Minerals -- On Manganese, Magnesium, or Magnesia Vitrariorum -- On Arsenic and its Acid -- Remarks upon Salts of Benzoin--On Silex, Clay and Alum -- Analysis of the Calculus Vesical -- Method of Preparing Mercurius Dulcis Via Humida -- Cheaper and more Convenient Method of Preparing Pulvis Algarothi -- Experiments upon Molybdæna -- Experiments on Plumbago -- Method of Preparing a New Green Colour -- Of the Decomposition of Neutral Salts by Unslaked Lime and Iron -- On the Quantity of Pure Air which is Daily Present in our Atmosphere -- On Milk and its Acid -- On the Acid of Saccharum Lactis -- On the Constituent Parts of Lapis Ponderosus or Tungsten -- Experiments and Observations on Ether -- Index.
THE MANUFACTURE OF ALUM AND THE SULPHATES AND OTHER SALTS OF ALUMINA AND IRON. Their Uses and Applications as Mordants in Dyeing and Calico Printing, and their other Applications in the Arts, Manufactures, Sanitary Engineering, Agriculture and Horticulture. Translated from the French of Lucien Geschwind. 195 Illustrations. 400 pp. Royal 8vo. Price 12s. 6d. net. (Post free, 13s. home; 13s. 6d. abroad.)
Contents.
Theoretical Study of Aluminium, Iron, and Compounds of these Metals -- Aluminium and its Compounds -- Iron and Iron Compounds.
Manufacture of Aluminium Sulphates and Sulphates of Iron -- Manufacture of Aluminium Sulphate and the Alums -- Manufacture of Sulphates of Iron.
Uses of the Sulphates of Aluminium and Iron -- Uses of Aluminium Sulphate and Alums -- Application to Wool and Silk -- Preparing and using Aluminium Acetates -- Employment of Aluminium Sulphate in Carbonising Wool -- The Manufacture of Lake Pigments -- Manufacture of Prussian Blue -- Hide and Leather Industry -- Paper Making -- Hardening Plaster -- Lime Washes -- Preparation of Non-inflammable Wood, etc. -- Purification of Waste Waters. -- Uses and Applications of Ferrous Sulphate and Ferric Sulphates -- Dyeing -- Manufacture of Pigments -- Writing Inks -- Purification of Lighting Gas -- Agriculture -- Cotton Dyeing -- Disinfectant -- Purifying Waste Liquors -- Manufacture of Nordhausen Sulphuric Acid -- Fertilising.
Chemical Characteristics of Iron and Aluminium -- Analysis of Various Aluminous or Ferruginous Products -- Aluminium -- Analysing Aluminium Products --Alunite Alumina -- Sodium Aluminate -- Aluminium Sulphate -- Iron -- Analytical Characteristics of Iron Salts -- Analysis of Pyritic Lignite -- Ferrous and Ferric Sulphates -- Rouil Mordant -- Index.
AMMONIA AND ITS COMPOUNDS: Their Manufacture and Uses. By Camille Vincent, Professor at the Central School of Arts and Manufactures, Paris. Translated from the French by M. J. Salter. Royal 8vo. 114 pp. Thirty-two Illustrations. Price 5s. net. (Post free, 5s. 4d. home; 5s. 6d. abroad.)
Contents.
General Considerations: Various Sources of Ammoniacal Products; Human Urine as a Source of Ammonia -- Extraction of Ammoniacal Products from Sewage -- Extraction of Ammonia from Gas Liquor -- Manufacture of Ammoniacal Compounds from Bones, Nitrogenous Waste, Beetroot Wash and Peat -- Manufacture of Caustic Ammonia, and Ammonium Chloride, Phosphate and Carbonate -- Recovery of Ammonia from the Ammonia-Soda Mother Liquors -- Index.
INDUSTRIAL (p. c10) ALCOHOL. A Practical Manual on the Production and Use of Alcohol for Industrial Purposes and for Use as a Heating Agent, as an Illuminant and as a Source of Motive Power. By J. G. M'Intosh, Lecturer on Manufacture and Applications of Industrial Alcohol at The Polytechnic, Regent Street, London. Demy 8vo. 1907. 250 pp. With 75 Illustrations and 25 Tables. Price 7s. 6d. net. (Post free, 7s. 9d. home; 8s. abroad.)
Contents.
Alcohol and its Properties. -- Ethylic Alcohol -- Absolute Alcohol -- Adulterations -- Properties of Alcohol -- Fractional Distillation -- Destructive Distillation -- Products of Combustion -- Alcoholometry -- Proof Spirit -- Analysis of Alcohol -- Table showing Correspondence between the Specific Gravity and Per Cents. of Alcohol over and under Proof -- Other Alcohol Tables. Continuous Aseptic and Antiseptic Fermentation and Sterilisation in Industrial Alcohol Manufacture. The Manufacture of Industrial Alcohol from Beets. -- Beet Slicing Machines -- Extraction of Beet Juice by Maceration, by Diffusion -- Fermentation in Beet Distilleries -- Plans of Modern Beet Distillery, The Manufacture of Industrial Alcohol from Grain. -- Plan of Modern Grain Distillery. The Manufacture of Industrial Alcohol from Potatoes. The Manufacture of Industrial Alcohol from Surplus Stocks of Wine, Spoilt Wine, Wine Marcs, and from Fruit in General. The Manufacture of Alcohol from the Sugar Cane and Sugar Cane Molasses -- Plans. Plant, etc., for the Distillation and Rectification of Industrial Alcohol. -- The Caffey and other "Patent" Stills -- Intermittent versus Continuous Rectification -- Continuous Distillation -- Rectification of Spent Wash. The Manufacture and Uses of Various Alcohol Derivatives, Ether, Haloid Ethers, Compound Ethers, Chloroform -- Methyl and Amyl Alcohols and their Ethereal Salts, Acetone -- Barbet's Ether, Methyl Alcohol and Acetone Rectifying Stills. The Uses of Alcohol in Manufactures, etc. -- List of Industries in which Alcohol is used, with Key to Function of Alcohol in each Industry. The Uses of Alcohol for Lighting, Heating, and Motive Power.
ANALYSIS OF RESINS AND BALSAMS. Translated from the German of Dr. Karl Dieterich. Demy 8vo. 340 pp. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. 3d. abroad.)
MANUAL OF AGRICULTURAL CHEMISTRY. By Herbert Ingle, F.I.C., Lecturer on Agricultural Chemistry, the Yorkshire College; Lecturer in the Victoria University. 388 pp. 11 Illustrations. Demy 8vo. Price 7s. 6d. net. (Post free, 8s. home; 8s. 6d. abroad.)
Contents.
Introduction -- The Atmosphere -- The Soil -- The Reactions occurring in Soils -- The Analysis of Soils -- Manures, Natural -- Manures (continued) -- The Analysis of Manures -- The Constituents of Plants -- The Plant -- Crops -- The Animal -- Foods and Feeding -- Milk and Milk Products -- The Analysis of Milk and Milk Products -- Miscellaneous Products used in Agriculture -- Appendix -- Index.
THE UTILISATION OF WASTE PRODUCTS. A Treatise on the Rational Utilisation, Recovery and Treatment of Waste Products of all kinds. By Dr. Theodor Koller. Translated from the Second Revised German Edition. Twenty-two Illustrations. Demy 8vo. 280 pp. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. 3d. abroad.)
Contents.
The Waste of Towns -- Ammonia and Sal-Ammoniac -- Rational Processes for Obtaining these Substances by Treating Residues and Waste -- Residues in the Manufacture of Aniline Dyes -- Amber Waste -- Brewers' Waste -- Blood and Slaughter-House Refuse -- Manufactured Fuels -- Waste Paper and Bookbinders' Waste -- Iron Slags -- Excrement -- Colouring Matters from Waste -- Dyers' Waste Waters -- Fat from Waste -- Fish Waste -- Calamine Sludge -- Tannery Waste -- Gold and Silver Waste -- India-rubber and Caoutchouc Waste -- Residues in the Manufacture of Rosin Oil -- Wood Waste -- Horn Waste -- Infusorial Earth -- Iridium from Goldsmiths' Sweepings -- Jute Waste -- Cork Waste -- Leather Waste -- Glue Makers' Waste -- Illuminating Gas from Waste and the By-Products of the Manufacture of Coal Gas -- Meerschum -- Molasses--Metal Waste -- By-Products in the Manufacture of Mineral Waters -- Fruit -- The By-Products of Paper and Paper Pulp Works -- By-Products in the Treatment of Coal Tar Oils -- Fur Waste -- The Waste Matter in the Manufacture of Parchment Paper -- Mother of Pearl Waste -- Petroleum Residues -- Platinum Residues -- Broken Porcelain, Earthenware and Glass -- Salt Waste -- Slate Waste -- Sulphur -- Burnt Pyrites -- Silk Waste -- Soap Makers' Waste -- Alkali Waste and the Recovery of Soda--Waste Produced in Grinding Mirrors -- Waste Products in the Manufacture of Starch -- Stearic Acid -- Vegetable Ivory Waste -- Turf -- Waste Waters of Cloth Factories -- Wine Residues -- Tinplate Waste -- Wool Waste -- Wool Sweat -- The Waste Liquids from Sugar Works -- Index.
INK MANUFACTURE: Including Writing, Copying, Lithographic, Marking, Stamping, and Laundry Inks. By Sigmund Lehner. Three Illustrations. Crown 8vo. 162 pp. Translated from the German of the Fifth Edition. Price 5s. net. (Post free, 5s. 3d. home; 5s. 6d. abroad.)
Contents.
Varieties of Ink -- Writing Inks -- Raw Materials of Tannin Inks -- The Chemical Constitution of the Tannin Inks -- Recipes for Tannin Inks -- Logwood Tannin Inks -- Ferric Inks -- Alizarine Inks--Extract Inks -- Logwood Inks -- Copying Inks -- Hektographs -- Hektograph Inks -- Safety Inks -- Ink Extracts and Powders -- Preserving Inks -- Changes in Ink and the Restoration of Faded Writing -- Coloured Inks -- Red Inks -- Blue Inks -- Violet Inks -- Yellow Inks -- Green Inks -- Metallic Inks -- Indian Ink -- Lithographic Inks and Pencils -- Ink Pencils -- Marking Inks -- Ink Specialities -- Sympathetic Inks -- Stamping Inks -- Laundry or Washing Blue -- Index.
SEALING-WAXES, WAFERS AND OTHER ADHESIVES FOR THE HOUSEHOLD, OFFICE, WORKSHOP AND FACTORY. By H. C. Standage. Crown 8vo. 96 pp. Price 5s. net. (Post free, 5s. 3d. home; 5s. 6d. abroad.)
Contents.
Materials Used for Making Sealing=Waxes -- The Manufacture of Sealing-Waxes -- Wafers -- Notes on the Nature of the Materials Used in Making Adhesive Compounds -- Cements for Use in the Household -- Office Gums, Pastes and Mucilages -- Adhesive Compounds for Factory and Workshop Use.
LEAD AND ITS COMPOUNDS. By Thos. Lambert, Technical and Consulting Chemist. Demy 8vo. 226 pp. Forty Illustrations. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. 3d. abroad.)
Contents.
History -- Ores of Lead -- Geographical Distribution of the Lead Industry -- Chemical and Physical Properties of Lead -- Alloys of Lead -- Compounds of Lead -- Dressing of Lead Ores -- Smelting of Lead Ores -- Smelting in the Scotch or American Ore-hearth -- Smelting in the Shaft or Blast Furnace -- Condensation of Lead Fume -- Desilverisation, or the Separation of Silver from Argentiferous Lead -- Cupellation -- The Manufacture of Lead Pipes and Sheets -- Protoxide of Lead -- Litharge and Massicot -- Red Lead or Minium -- Lead Poisoning -- Lead Substitutes -- Zinc and its Compounds -- Pumice Stone -- Drying Oils and Siccatives -- Oil of Turpentine Resin -- Classification of Mineral Pigments -- Analysis of Raw and Finished Products -- Tables -- Index.
NOTES ON LEAD ORES: Their Distribution and Properties. By Jas. Fairie, F.G.S. Crown 8vo. 64 pages. Price 2s. 6d. net. (Post free, 2s. 9d. home; 3s. abroad.)
THE RISKS AND DANGERS TO HEALTH OF VARIOUS OCCUPATIONS AND THEIR PREVENTION. By Leonard A. Parry, M.D., B.Sc. (Lond.). 196 pp. Demy 8vo. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Occupations which are Accompanied by the Generation and Scattering of Abnormal Quantities of Dust -- Trades in which there is Danger of Metallic Poisoning -- Certain Chemical Trades -- Some Miscellaneous Occupations --Trades in which Various Poisonous Vapours are Inhaled --General Hygienic Considerations -- Index.
DRYING BY MEANS OF AIR AND STEAM. Explanations, Formulæ, and Tables for Use in Practice. Translated from the German of E. Hausrrand. Two folding Diagrams and Thirteen Tables. Crown 8vo. 72 pp. Price 5s. net. (Post free, 5s. 3d. home; 5s. 6d. abroad.)
Contents.
British and Metric Systems Compared -- Centigrade and Fahr. Thermometers -- Estimation of the Maximum Weight of Saturated Aqueous Vapour which can be contained in 1 kilo. of Air at Different Pressure and Temperatures -- Calculation of the Necessary Weight and Volume of Air, and of the Least Expenditure of Heat, per Drying Apparatus with Heated Air, at the Atmospheric Pressure: A, With the Assumption that the Air is Completely Saturated with Vapour both before Entry and after Exit from the Apparatus -- B, When the Atmospheric Air is Completely Saturated before entry, but at its exit is only 3/4, 1/2 or 1/4 Saturated -- C, When the Atmospheric Air is not Saturated with Moisture before Entering the Drying Apparatus -- Drying Apparatus, in which, in the Drying Chamber, a Pressure is Artificially Created, Higher or Lower than that of the Atmosphere -- Drying by Means of Superheated Steam, without Air --Heating Surface, Velocity of the Air Current, Dimensions of the Drying Room, Surface of the Drying Material, Losses of Heat -- Index.
(See also "Evaporating, Condensing and Cooling Apparatus.")
PURE AIR, OZONE AND WATER. A Practical Treatise of their Utilisation and Value in Oil, Grease, Soap, Paint, Glue and other Industries, By W. B. Cowell. Twelve Illustrations. Crown 8vo. 85 pp. Price 5s. net. (Post free, 5s. 3d. home; 5s. 6d. abroad.)
Contents.
Atmospheric Air; Lifting of Liquids; Suction Process; Preparing Blown Oils; Preparing Siccative Drying Oils -- Compressed Air; Whitewash -- Liquid Air; Retrocession -- Purification of Water; Water Hardness -- Fleshings and Bones -- Ozonised Air in the Bleaching and Deodorising of Fats, Glues, etc.; Bleaching Textile Fibres -- Appendix: Air and Gases; Pressure of Air at Various Temperatures; Fuel; Table of Combustibles; Saving of Fuel by Heating Feed Water; Table of Solubilities of Scale Making Minerals; British Thermal Units Tables; Volume of the Flow of Steam into the Atmosphere; Temperature of Steam -- Index.
THE INDUSTRIAL USES OF WATER. COMPOSITION--EFFECTS--TROUBLES--REMEDIES--RESIDUARY WATERS--PURIFICATION--ANALYSIS. By H. de la Coux. Royal 8vo. Translated from the French and Revised by Arthur Morris. 364 pp. 135 Illustrations. Price 10s. 6d. net. (Post free, 11s. home; 11s. 6d. abroad.)
Contents.
Chemical Action of Water in Nature and in Industrial Use -- Composition of Waters -- Solubility of Certain Salts in Water Considered from the Industrial Point of View -- Effects on the Boiling of Water -- Effects of Water in the Industries -- Difficulties with Water -- Feed Water for Boilers -- Water in Dye works, Print Works, and Bleach Works -- Water in the Textile Industries and in Conditioning -- Water in Soap Works -- Water in Laundries and Washhouses -- Water in Tanning -- Water in Preparing Tannin and Dyewood Extracts -- Water in Papermaking -- Water in Photography -- Water in Sugar Refining -- Water in Making Ices and Beverages -- Water in Cider Making -- Water in Brewing -- Water in Distilling -- Preliminary Treatment and Apparatus -- Substances Used for Preliminary Chemical Purification -- Commercial Specialities and their Employment -- Precipitation of Matters in Suspension in Water -- Apparatus for the Preliminary Chemical Purification of Water -- Industrial Filters -- Industrial Sterilisation of Water -- Residuary Waters and their Purification -- Soil Filtration -- Purification by Chemical Processes -- Analyses -- Index.
(See Books on Smoke Prevention, Engineering and Metallurgy etc.)
PRACTICAL X RAY WORK. By Frank T. Addyman, B.Sc. (Lond.), F.I.C., Member of the Roentgen Society of London; Radiographer to St. George's Hospital; Demonstrator of Physics and Chemistry, and Teacher of Radiography in St. George's Hospital Medical School. Demy 8vo. Twelve Plates from Photographs of X Ray Work. Fifty-two Illustrations. 200 pp. Price 10s. 6d. net. (Post free, 10s. 10d. home; 11s. 3d. abroad.)
Contents.
Historical -- Work leading up to the Discovery of the X Rays -- The Discovery--Apparatus and its Management -- Electrical Terms -- Sources of Electricity -- Induction Coils -- Electrostatic Machines -- Tubes -- Air Pumps -- Tube Holders and Stereoscopic Apparatus -- Fluorescent Screens -- Practical X Ray Work -- Installations -- Radioscopy -- Radiography -- X Rays in Dentistry -- X Rays in Chemistry -- X Rays in War -- Index.
List of Plates.
Frontispiece -- Congenital Dislocation of Hip-Joint. -- I., Needle in Finger. -- II., Needle in Foot. -- III., Revolver Bullet in Calf and Leg. -- IV., A Method of Localisation. -- V., Stellate Fracture of Patella showing shadow of "Strapping". -- VI., Sarcoma. -- VII., Six-weeks-old Injury to Elbow showing new Growth of Bone. -- VIII., Old Fracture of Tibia and Fibula badly set. -- IX., Heart Shadow. -- X., Fractured Femur showing Grain of Splint. -- XI., Barrell's Method of Localisation.
INDIA-RUBBER AND GUTTA-PERCHA. Translated from the French of T. Seeligmann, G. Lamy Torvilhon and H. Falconnet by John Geddes McIntosh. Royal 8vo.
[Out of print. Second Edition in preparation.]
Contents.
India-Rubber -- Botanical Origin -- Climatology -- Soil -- Rational Culture and Acclimation of the Different Species of India-Rubber Plants -- Methods of Obtaining the Latex -- Methods of Preparing Raw or Crude India-Rubber -- Classification of the Commercial Species of Raw Rubber -- Physical and Chemical Properties of the Latex and of India-Rubber -- Mechanical Transformation of Natural Caoutchouc into Washed or Normal Caoutchouc (Purification) and Normal Rubber into Masticated Rubber -- Softening, Cutting, Washing, Drying -- Preliminary Observations -- Vulcanisation of Normal Rubber -- Chemical and Physical Properties of Vulcanised Rubber -- General Considerations -- Hardened Rubber or Ebonite -- Considerations on Mineralisation and other Mixtures -- Coloration and Dyeing -- Analysis of Natural or Normal Rubber and Vulcanised Rubber -- Rubber Substitutes -- Imitation Rubber.
Gutta Percha -- Botanical Origin -- Climatology -- Soil -- Rational Culture -- Methods of Collection -- Classification of the Different Species of Commercial Gutta Percha -- Physical and Chemical Properties -- Mechanical Transformation -- Methods of Analysing -- Gutta Percha Substitutes -- Index.
PRACTICAL TREATISE ON THE LEATHER INDUSTRY. By A. M. Villon. Translated by Frank T. Addyman, B.Sc. (Lond.), F.I.C., F.C.S.; and Corrected by an Eminent Member of the Trade. 500 pp., royal 8vo. 123 Illustrations. Price 21s. net. (Post free, 21s. 6d. home; 22s. 6d. abroad.)
Contents.
Preface--Translator's Preface--List of Illustrations.
Part I., Materials used in Tanning -- Skins: Skin and its Structure; Skins used in Tanning; Various Skins and their Uses -- Tannin and Tanning Substances: Tannin; Barks (Oak); Barks other than Oak; Tanning Woods; Tannin-bearing Leaves; Excrescences; Tan-bearing Fruits; Tan-bearing Roots and Bulbs; Tanning Juices; Tanning Substances used in Various Countries; Tannin Extracts; Estimation of Tannin and Tannin Principles.
Part II., Tanning -- The Installation of a Tannery: Tan Furnaces; Chimneys, Boilers, etc.; Steam Engines -- Grinding and Trituration of Tanning Substances: Cutting up Bark; Grinding Bark; The Grinding of Tan Woods; Powdering Fruit, Galls and Grains; Notes on the Grinding of Bark -- Manufacture of Sole Leather: Soaking; Sweating and Unhairing; Plumping and Colouring; Handling; Tanning; Tanning Elephants' Hides; Drying; Striking or Pinning -- Manufacture of Dressing Leather: Soaking; Depilation; New Processes for the Depilation of Skins; Tanning; Cow Hides; Horse Hides; Goat Skins; Manufacture of (p. c14) Split Hides -- On Various Methods of Tanning: Mechanical Methods; Physical Methods; Chemical Methods; Tanning with Extracts -- Quantity and Quality; Quantity; Net Cost; Quality of Leather -- Various Manipulations of Tanned Leather: Second Tanning; Grease Stains; Bleaching Leather; Waterproofing Leather; Weighting Tanned Leather; Preservation of Leather -- Tanning Various Skins.
Part III., Currying -- Waxed Calf: Preparation; Shaving; Stretching or Slicking; Oiling the Grain; Oiling the Flesh Side; Whitening and Graining; Waxing; Finishing; Dry Finishing; Finishing in Colour; Cost -- White Calf: Finishing in White -- Cow Hide for Upper Leathers: Black Cow Hide; White Cow Hide; Coloured Cow Hide -- Smooth Cow Hide -- Black Leather -- Miscellaneous Hides: Horse; Goat; Waxed Goat Skin; Matt Goat Skin -- Russia Leather: Russia Leather; Artificial Russia Leather.
Part IV., Enamelled, Hungary and Chamoy Leather, Morocco, Parchment, Furs and Artificial Leather -- Enamelled Leather: Varnish Manufacture; Application of the Enamel; Enamelling in Colour -- Hungary Leather: Preliminary; Wet Work or Preparation; Aluming; Dressing or Loft Work; Tallowing; Hungary Leather from Various Hides -- Tawing: Preparatory Operations; Dressing; Dyeing Tawed Skins; Rugs -- Chamoy Leather -- Morocco: Preliminary Operations; Morocco Tanning: Mordants used in Morocco Manufacture; Natural Colours used in Morocco Dyeing; Artificial Colours; Different Methods of Dyeing; Dyeing with Natural Colours; Dyeing with Aniline Colours; Dyeing with Metallic Salts; Leather Printing; Finishing Morocco; Shagreen; Bronzed Leather -- Gilding and Silvering: Gilding; Silvering; Nickel and Cobalt -- Parchment -- Furs and Furriery: Preliminary Remarks; Indigenous Furs; Foreign Furs from Hot Countries; Foreign Furs from Cold Countries; Furs from Birds' Skins; Preparation of Furs; Dressing; Colouring; Preparation of Birds' Skins; Preservation of Furs -- Artificial Leather: Leather made from Scraps; Compressed Leather; American Cloth; Papier Mâché; Linoleum; Artificial Leather.
Part V., Leather Testing and the Theory of Tanning -- Testing and Analysis of Leather: Physical Testing of Tanned Leather; Chemical Analysis -- The Theory of Tanning and the other Operations of the Leather and Skin Industry: Theory of Soaking; Theory of Unhairing; Theory of Swelling; Theory of Handling; Theory of Tanning; Theory of the Action of Tannin on the Skin; Theory of Hungary Leather Making; Theory of Tawing; Theory of Chamoy Leather Making; Theory of Mineral Tanning.
Part VI., Uses of Leather -- Machine Belts: Manufacture of Belting; Leather Chain Belts; Various Belts; Use of Belts -- Boot and Shoe-making: Boots and Shoes; Laces -- Saddlery: Composition of a Saddle; Construction of a Saddle -- Harness: The Pack Saddle; Harness -- Military Equipment -- Glove Making -- Carriage Building -- Mechanical Uses.
Appendix, The World's Commerce in Leather -- Europe; America; Asia; Africa; Australasia -- Index.
THE LEATHER WORKER'S MANUAL. Being a Compendium of Practical Recipes and Working Formulæ for Curriers, Bootmakers, Leather Dressers, Blacking Manufacturers, Saddlers, Fancy Leather Workers. By H. C. Standage. Demy 8vo. 165 pp. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Blackings, Polishes, Glosses, Dressings, Renovators, etc., for Boot and Shoe Leather -- Harness Blackings, Dressings, Greases, Compositions, Soaps, and Boot-top Powders and Liquids, etc., etc. -- Leather Grinders' Sundries -- Currier's Seasonings, Blacking Compounds, Dressings, Finishes, Glosses, etc. -- Dyes and Stains for Leather --Miscellaneous Information -- Chrome Tannage -- Index.
THE MANUAL OF PRACTICAL POTTING. Compiled by Experts, and Edited by Chas. F. Binns. Revised Third Edition and Enlarged. 200 pp. Price 17s. 6d. net. (Post free, 17s. 10d. home; 18s. 3d. abroad.)
Contents.
Introduction. The Rise and Progress of the Potter's Art -- Bodies. China and Porcelain Bodies, Parian Bodies, Semi-porcelain and Vitreous Bodies, Mortar Bodies, Earthenwares Granite and C.C. Bodies, Miscellaneous Bodies, Sagger and Crucible Clays, Coloured Bodies, Jasper Bodies, Coloured Bodies for Mosaic Painting, Encaustic Tile Bodies, Body Stains, Coloured Dips -- Glazes. China Glazes, Ironstone Glazes, Earthenware Glazes, Glazes without Lead, Miscellaneous Glazes, Coloured Glazes, Majolica Colours -- Gold and Gold Colours. Gold, Purple of Cassius, Marone and Ruby, Enamel Coloured Bases, Enamel Colour Fluxes, Enamel Colours, Mixed Enamel Colours, Antique and Vellum Enamel Colours, Underglaze Colours, Underglaze Colour Fluxes, Mixed Underglaze Colours, Flow Powders, Oils and Varnishes -- Means and Methods. Reclamation of Waste Gold, The Use of Cobalt, Notes on Enamel Colours, Liquid or Bright Gold -- Classification and Analysis. Classification of Clay Ware, Lord Playfair's Analysis of Clays, The Markets of the (p. c15) World, Time and Scale of Firing, Weights of Potter's Material, Decorated Goods Count -- Comparative Loss of Weight of Clays -- Ground Felspar Calculations -- The Conversion of Slop Body Recipes into Dry Weight -- The Cost of Prepared Earthenware Clay -- Forms and Tables. Articles of Apprenticeship, Manufacturer's Guide to Stocktaking, Table of Relative Values of Potter's Materials, Hourly Wages Table, Workman's Settling Table, Comparative Guide for Earthenware and China Manufacturers in the use of Slop Flint and Slop Stone, Foreign Terms applied to Earthenware and China Goods, Table for the Conversion of Metrical Weights and Measures on the Continent and South America -- Index.
CERAMIC TECHNOLOGY: Being some Aspects of Technical Science as Applied to Pottery Manufacture. Edited by Charles F. Binns. 100 pp. Demy 8vo. Price 12s. 6d. net. (Post free, 12s. 10d. home; 13s. abroad.)
Contents.
Preface -- The Chemistry of Pottery -- Analysis and Synthesis -- Clays and their Components--The Biscuit Oven -- Pyrometry -- Glazes and their Composition -- Colours and Colour-making -- Index.
A TREATISE ON THE CERAMIC INDUSTRIES. A Complete Manual for Pottery, Tile and Brick Works. By Emile Bourry. Translated from the French by Wilton P. Rix, Examiner in Pottery and Porcelain to the City and Guilds of London Technical Institute, Pottery Instructor to the Hanley School Board. Royal 8vo. 760 pp. 323 Illustrations. Price 21s. net. (Post free, 22s. home; 24s. abroad.)
Contents.
Part I., General Pottery Methods. Definition and History. Definitions and Classification of Ceramic Products -- Historic Summary of the Ceramic Art -- Raw Materials of Bodies. Clays: Pure Clay and Natural Clays -- Various Raw Materials: Analogous to Clay -- Agglomerative and Agglutinative -- Opening -- Fusible -- Refractory -- Trials of Raw Materials -- Plastic Bodies. Properties and Composition -- Preparation of Raw Materials: Disaggregation -- Purification -- Preparation of Bodies: By Plastic Method -- By Dry Method -- By Liquid Method -- Formation, Processes of Formation: Throwing -- Expression -- Moulding by Hand, on the Jolley, by Compression, by Slip Casting -- Slapping -- Slipping -- Drying. Drying of Bodies -- Processes of Drying; By Evaporation -- By Aeration -- By Heating -- By Ventilation -- By Absorption -- Glazes. Composition and Properties -- Raw Materials -- Manufacture and Application -- Firing. Properties of the Bodies and Glazes during Firing -- Description of the Kilns -- Working of the Kilns -- Decoration. Colouring Materials -- Processes of Decoration.
Part II., Special Pottery Methods. Terra Cottas. Classification: Plain Ordinary, Hollow, Ornamental, Vitrified, and Light Bricks -- Ordinary and Black Tiles -- Paving Tiles -- Pipes -- Architectural Terra Cottas -- Vases, Statues and Decorative Objects -- Common Pottery -- Pottery for Water and Filters -- Tobacco Pipes -- Lustre Ware -- Properties and Tests for Terra Cottas--Fireclay Goods. Classification: Argillaceous, Aluminous, Carboniferous, Silicious and Basic Fireclay Goods -- Fireclay Mortar (Pug) -- Tests for Fireclay Goods -- Faiences. Varnished Faiences -- Enamelled Faiences -- Silicious Faiences -- Pipeclay Faiences -- Pebble Work -- Feldspathic Faiences -- Composition, Processes of Manufacture and General Arrangements of Faience Potteries -- Stoneware. Stoneware Properly So-called: Paving Tiles -- Pipes -- Sanitary Ware -- Stoneware for Food Purposes and Chemical Productions -- Architectural Stoneware -- Vases, Statues and other Decorative Objects -- Fine Stoneware -- Porcelain. Hard Porcelain for Table Ware and Decoration, for the Fire, for Electrical Conduits, for Mechanical Purposes; Architectural Porcelain, and Dull or Biscuit Porcelain -- Soft Phosphated or English Porcelain -- Soft Vitreous Porcelain, French and New Sèvres -- Argillaceous Soft or Seger's Porcelain -- Dull Soft or Parian Porcelain -- Dull Feldspathic Soft Porcelain -- Index.
POTTERY DECORATING, By R. Hainbach. Translated from the German. Crown 8vo. 22 Illustrations. Deals with Glazes, Colours, etc. [In the Press.]
ARCHITECTURAL POTTERY. Bricks, Tiles, Pipes, Enamelled Terra-cottas, Ordinary and Incrusted Quarries, Stoneware Mosaics, Faïences and Architectural Stoneware. By Leon Lefêvre. With Five Plates. 950 Illustrations in the Text, and numerous estimates. 500 pp., royal 8vo. Translated from the French by K. H. Bird, M.A., and W. Moore Binns. Price 15s. net. (Post free, 15s. 6d. home; 16s. 6d. abroad.)
Contents.
Part I. Plain Undecorated Pottery. -- Clays, Bricks, Tiles, Pipes, Chimney Flues, Terra-cotta.
Part II. Made-up or Decorated Pottery.
THE (p. c16) ART OF RIVETING GLASS, CHINA AND EARTHENWARE. By J. Howarth. Second Edition. Paper Cover. Price 1s. net; by post, home or abroad, 1s. 1d.
NOTES ON POTTERY CLAYS. Their Distribution, Properties, Uses and Analyses of Ball Clays, China Clays and China Stone. By Jas. Fairie, F.G.S. 132 pp. Crown 8vo. Price 3s. 6d. net. (Post free, 3s. 9d. home; 3s. 10d. abroad.)
A Reissue of
THE HISTORY OF THE STAFFORDSHIRE POTTERIES; AND THE RISE AND PROGRESS OF THE MANUFACTURE OF POTTERY AND PORCELAIN. With References to Genuine Specimens, and Notices of Eminent Potters. By Simeon Shaw. (Originally Published in 1829.) 265 pp. Demy 8vo. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. 3d. abroad.)
Contents.
Introductory Chapter showing the position of the Pottery Trade at the present time (1899) -- Preliminary Remarks -- The Potteries, comprising Tunstall, Brownhills, Greenfield and New Field, Golden Hill, Latebrook, Green Lane, Burslem, Longport and Dale Hall, Hot Lane and Cobridge, Hanley and Shelton, Etruria, Stoke, Penkhull, Fenton, Lane Delph, Foley, Lane End -- On the Origin of the Art, and its Practice among the early Nations -- Manufacture of Pottery, prior to 1700 -- The Introduction of Red Porcelain by Messrs. Elers, of Bradwell, 1690 -- Progress of the Manufacture from 1700 to Mr. Wedgwood's commencement in 1760 -- Introduction of Fluid Glaze -- Extension of the Manufacture of Cream Colour -- Mr. Wedgwood's Queen's Ware -- Jasper, and Appointment of Potter to Her Majesty -- Black Printing -- Introduction of Porcelain. Mr. W. Littler's Porcelain -- Mr. Cookworthy's Discovery of Kaolin and Petuntse, and Patent -- Sold to Mr. Champion -- resold to the New Hall Com. -- Extension of Term -- Blue Printed Pottery. Mr. Turner, Mr. Spode (1), Mr. Baddeley, Mr. Spode (2), Messrs. Turner, Mr. Wood, Mr. Wilson, Mr. Minton -- Great Change in Patterns of Blue Printed -- Introduction of Lustre Pottery. Improvements in Pottery and Porcelain subsequent to 1800.
A Reissue of
THE CHEMISTRY OF THE SEVERAL NATURAL AND ARTIFICIAL HETEROGENEOUS COMPOUNDS USED IN MANUFACTURING PORCELAIN, GLASS AND POTTERY. By Simeon Shaw. (Originally published in 1837.) 750 pp. Royal 8vo. Price 14s. net. (Post free, 15s. home; 17s. abroad.)
RECIPES FOR FLINT GLASS MAKING. By a British Glass Master and Mixer. Sixty Recipes. Being Leaves from the Mixing Book of several experts in the Flint Glass Trade, containing up-to-date recipes and valuable information as to Crystal, Demi-crystal and Coloured Glass in its many varieties. It contains the recipes for cheap metal suited to pressing, blowing, etc., as well as the most costly crystal and ruby. Crown 8vo. Price 10s. 6d. net. (Post free, 10s. 9d. home; 10s. 10d. abroad.)
Contents.
Ruby -- Ruby from Copper -- Flint for using with the Ruby for Coating -- A German Metal -- Cornelian, or Alabaster -- Sapphire Blue -- Crysophis -- Opal -- Turquoise Blue -- Gold Colour -- Dark Green -- Green (common) -- Green for Malachite -- Blue for Malachite -- Black for Malachite -- Black -- Common Canary Batch -- Canary -- White Opaque Glass -- Sealing-wax Red -- Flint -- Flint Glass (Crystal and Demi) -- Achromatic Glass -- Paste Glass -- White Enamel -- Firestone--Dead White (for moons) -- White Agate -- Canary -- Canary Enamel -- Index.
A (p. c17) TREATISE ON THE ART OF GLASS PAINTING. Prefaced with a Review of Ancient Glass. By Ernest R. Suffling. With One Coloured Plate and Thirty-seven Illustrations. Demy 8vo. 140 pp. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
A Short History of Stained Glass -- Designing Scale Drawings --Cartoons and the Cut Line -- Various Kinds of Glass Cutting for Windows -- The Colours and Brushes used in Glass Painting -- Painting on Glass, Dispersed Patterns -- Diapered Patterns -- Aciding -- Firing -- Fret Lead Glazing -- Index.
PAINTING ON GLASS AND PORCELAIN AND ENAMEL PAINTING. A Complete Introduction to the Preparation of all the Colours and Fluxes used for Painting on Porcelain, Enamel, Faïence and Stoneware, the Coloured Pastes and Coloured Glasses, together with a Minute Description of the Firing of Colours and Enamels. By Felix Hermann, Technical Chemist. With Eighteen Illustrations. 300 pp. Translated from the German second and enlarged Edition. Price 10s. 6d. net. (Post free, 10s. 10d. home; 11s. abroad.)
THE DYEING OF PAPER PULP. A Practical Treatise for the use of Papermakers, Paperstainers, Students and others. By Julius Erfurt, Manager of a Paper Mill. Translated into English and Edited with Additions by Julius Hübner, F.C.S., Lecturer on Papermaking at the Manchester Municipal Technical School. With Illustrations and 157 patterns of paper dyed in the pulp. Royal 8vo, 180 pp. Price 15s. net. (Post free, 15s. 6d. home; 16s. 6d. abroad.)
Contents.
Behaviour of the Paper Fibres during the Process of Dyeing, Theory of the Mordant -- Colour Fixing Mediums (Mordants) -- Influence of the Quality of the Water Used -- Inorganic Colours -- Organic Colours -- Practical Application of the Coal Tar Colours according to their Properties and their Behaviour towards the Different Paper Fibres -- Dyed Patterns on Various Pulp Mixtures -- Dyeing to Shade -- Index.
THE PAPER MILL CHEMIST. By Henry P. Stevens, M.A., Ph.D., F.I.C. Royal 12mo. 60 Illustrations. [In the press.]
Contents.
Introduction. -- Dealing with the Apparatus required in Chemical Work and General Chemical Manipulation, introducing the subject of Qualitative and Quantitative Analysis. Fuels. -- Analysis of Coal, Coke and other Fuels -- Sampling and Testing for Moisture, Ash, Calorific Value, etc. -- Comparative Heating Value of different Fuels and Relative Efficiency. Water. -- Analysis for Steam Raising and for Paper Making Purposes generally -- Water Softening and Purification -- A List of the more important Water Softening Plant, giving Power required, Weight, Space Occupied, Out-put and Approximate Cost. Raw Materials and Detection of Adulterants. -- Analysis and Valuation of the more important Chemicals used in Paper Making, including Lime, Caustic Soda, Sodium Carbonate, Mineral Acids, Bleach Antichlor, Alum, Rosin and Rosin Size, Glue Gelatin and Casein, Starch, China Clay, Blanc Fixe, Satin White and other Loading Materials, Mineral Colours and Aniline Dyes. Manufacturing Operations. -- Rags and the Chemical Control of Rag Boiling -- Esparto Boiling -- Wood Boiling -- Testing Spent Liquors and Recovered Ash -- Experimental Tests with Raw Fibrous Materials -- Boiling in Autoclaves -- Bleaching and making up Hand Sheets -- Examination of Sulphite Liquors -- Estimation of Moisture in Pulp and Half-stuff -- Recommendations of the British Wood Pulp Association. Finished Products. -- Paper Testing, including Physical, Chemical and Microscopical Tests, Area, Weight, Thickness, Apparent Specific Gravity, Bulk or Air Space. Determination of Machine Direction, Thickness, Strength, Stretch, Resistance to Crumpling and Friction, Transparency, Absorbency and other qualities of Blotting Papers -- Determination of the Permeability of Filtering Papers -- Detection and Estimation of Animal and Vegetable Size in Paper -- Sizing Qualities of Paper -- Fibrous Constituents -- Microscopical Examination of Fibres -- The Effect of Beating on Fibres -- Staining Fibres -- Mineral Matter -- Ash -- Qualitative and Quantitative Examination of Mineral Matter -- Examination of Coated Papers and Colouring Matters in Paper.
CONTENTS (p. c18) OF "THE TESTING OF PAPER"--continued.
Tables. -- English and Metrical Weights and Measures with Equivalents -- Conversion of Grams to Grains and vice versa -- Equivalent Costs per lb., cwt., and ton -- Decimal Equivalents of lbs., qrs., and cwts. -- Thermometric and Barometric Scales -- Atomic Weights and Molecular Weights -- Factors for Calculating the Percentage of Substance Sought from the Weight of Substance Found -- Table of Solubilities of Substances Treated of in Paper Making -- Specific Gravity Tables of such substances as are used in Paper Making, including Sulphuric Acid Hydrochloric Acid, Bleach, Milk of Lime, Caustic Soda, Carbonate of Soda, etc., giving Percentage Strength with Specific Gravity and Degrees Tw. -- Hardness Table for Soap Tests -- Dew Point -- Wet and Dry Bulb Tables -- Properties of Saturated Steam, giving Temperature, Pressure and Volume -- List of Different Machines used in the Paper Making Industry, giving Size, Weight, Space Occupied, Power to Drive, Out-put and Approximate Cost -- Calculation of Moisture in Pulp -- Rag-Boiling Tables, giving Percentages of Lime Soda and Time required -- Loss in Weight in Rags and other Raw Materials during Boiling and Bleaching -- Conditions of Buying and Selling as laid down by the Paper Makers' Association -- Table of Names and Sizes of Papers -- Table for ascertaining the Weight per Ream from the Weight per Sheet -- Calculations of Areas and Volumes -- Logarithms -- Blank pages for Notes.
THE TREATMENT OF PAPER FOR SPECIAL PURPOSES. By L. E. Andés. Translated from the German. Crown 8vo. 48 Illustrations. 250 pp. [In the Press.]
Contents.
I., Parchment Paper, Vegetable Parchment. -- The Parchment Paper Machine -- Opaque Supple Parchment Paper -- Thick Parchment -- Krugler's Parchment Paper and Parchment Slates -- Double and Triple Osmotic Parchment -- Utilising Waste Parchment Paper -- Parchmented Linen and Cotton -- Parchment Millboard -- Imitation Horn and Ivory from Parchment Paper -- Imitation Parchment Paper -- Artificial Parchment -- Testing the Sulphuric Acid. II., Papers for Transfer Pictures. III., Papers for Preservative and Packing Purposes. -- Butter Paper -- Wax Paper -- Paraffin Paper -- Wrapping Paper for Silverware -- Waterproof Paper -- Anti-corrosive Paper. IV., Grained Transfer Papers. V., Fire-proof and Antifalsification Papers, VI., Paper Articles. -- Vulcanised Paper Maché -- Paper Bottles -- Plastic Articles of Paper -- Waterproof Coverings for Walls and Ceilings -- Paper Wheels, Roofing and Boats -- Paper Barrels -- Paper Boxes -- Paper Horseshoes. VII., Gummed Paper. VIII., Hectograph Papers. IX., Insecticide Papers. -- Fly Papers -- Moth Papers. X., Chalk and Leather Papers. -- Glacé Chalk Paper -- Leather Paper -- Imitation Leather. XI., Luminous Papers -- Blue-Print Papers -- Blotting Papers. XII., Metal Papers -- Medicated Papers. XIII., Marbled Papers. XIV., Tracing and Copying Papers -- Iridescent or Mother of Pearl Papers. XV., Photographic Papers -- Shellac Paper -- Fumigating Papers -- Test Papers. XVI., Papers for Cleaning and Polishing Purposes -- Glass Paper -- Pumice Paper -- Emery Paper. XVII., Lithographic Transfer Papers. XIX., Sundry Special Papers -- Satin Paper -- Enamel Paper -- Cork Paper -- Split Paper -- Electric Paper -- Paper Matches -- Magic Pictures -- Laundry Blue Papers -- Blue Paper for Bleachers. XX., Waterproof Papers -- Washable Drawing Papers -- Washable Card -- Washable Coloured Paper--Waterproof Millboard -- Sugar Paper. XXI., The Characteristics of Paper -- Paper Testing.
ENAMELS AND ENAMELLING. For Enamel Makers, Workers in Gold and Silver, and Manufacturers of Objects of Art. By Paul Randau. Translated from the German. With Sixteen Illustrations. Demy 8vo. 180 pp. Price 10s. 6d. net. (Post free, 10s. 10d. home; 11s. abroad.)
THE ART OF ENAMELLING ON METAL. By W. Norman Brown. Twenty-eight Illustrations. Crown 8vo. 60 pp. Price 2s. 6d. net. (Post free, 2s. 9d. home and abroad.)
SILK THROWING AND WASTE SILK SPINNING. By Hollins Rayner. Demy 8vo. 170 pp. 117 Illus. Price 5s. net. (Post free, 5s. 4d. home; 5s. 6d. abroad.)
Contents.
The Silkworm -- Cocoon Reeling and Qualities of Silk -- Silk Throwing -- Silk Wastes -- The Preparation of Silk Waste for Degumming -- Silk Waste Degumming, Schapping and Discharging -- The Opening and Dressing of Wastes -- Silk Waste "Drawing" or "Preparing" Machinery -- Long Spinning -- Short Spinning -- Spinning and Finishing Processes -- Utilisation of Waste Products -- Noil Spinning -- Exhaust Noil Spinning.
THE CHEMICAL TECHNOLOGY OF TEXTILE FIBRES: Their Origin, Structure, Preparation, Washing, Bleaching, Dyeing, Printing and Dressing. By Dr. Georg von Georgievics. Translated from the German by Charles Salter. 320 pp. Forty-seven Illustrations. Royal 8vo. Price 10s. 6d. net. (Post free, 11s. home; 11s. 3d. abroad.)
Contents.
The Textile Fibres -- Washing, Bleaching, Carbonising -- Mordants and Mordanting -- Dyeing -- Printing -- Dressing and Finishing.
POWER-LOOM WEAVING AND YARN NUMBERING. According to Various Systems, with Conversion Tables. Translated from the German of Anthon Gruner. With Twenty-Six Diagrams in Colours. 150 pp. Crown 8vo. Price 7s. 6d. net. (Post free, 7s. 9d. home; 8s. abroad.)
Contents.
Power-Loom Weaving in General. Various Systems of Looms -- Mounting and Starting the Power-Loom. English Looms -- Tappet or Treadle Looms -- Dobbies -- General Remarks on the Numbering, Reeling and Packing of Yarn -- Appendix -- Useful Hints. Calculating Warps -- Weft Calculations -- Calculations of Cost Price in Hanks.
TEXTILE RAW MATERIALS AND THEIR CONVERSION INTO YARNS. (The Study of the Raw Materials and the Technology of the Spinning Process.) By Julius Zipser. Translated from German by Charles Salter. 302 Illustrations. 500 pp. Demy 8vo. Price 10s. 6d. net. (Post free, 11s. home; 11s. 6d. abroad.)
Contents.
PART 1. -- The Raw Materials Used in the Textile Industry. Mineral Raw Materials. Vegetable Raw Materials. Animal Raw Materials.
PART II. -- The Technology of Spinning or the Conversion of Textile Raw Materials into Yarn.
Spinning Vegetable Raw Materials. Cotton Spinning -- Installation of a Cotton Mill -- Spinning Waste Cotton and Waste Cotton Yarns -- Flax Spinning -- Fine Spinning -- Tow Spinning -- Hemp Spinning -- Spinning Hemp Tow String -- Jute Spinning -- Spinning Jute Line Yarn -- Utilising Jute Waste.
PART III. -- Spinning Animal Raw Materials.
Spinning Carded Woollen Yarn -- Finishing Yarn -- Worsted Spinning -- Finishing Worsted Yarn -- Artificial Wool or Shoddy Spinning -- Shoddy and Mungo Manufacture -- Spinning Shoddy and other Wool Substitutes -- Spinning Waste Silk -- Chappe Silk -- Fine Spinning -- Index.
GRAMMAR OF TEXTILE DESIGN. By H. Nisbet, Weaving and Designing Master, Bolton Municipal Technical School. Demy 8vo. 280 pp. 490 Illustrations and Diagrams. Price 6s. net. (Post free, 6s. 10d. home; 7s. abroad.)
Contents.
Chapter I., Introduction. -- General Principle of Fabric Structure and the use of Design Paper.
Chapter II., The Plain Weave and its Modifications. -- The Plain, Calico, or Tabby Weave. -- Firmness of Texture -- Variety of Texture -- Variety of Form: Ribbed Fabrics -- Corded Fabrics -- Matt Weaves.
Chapter III., Twill and Kindred Weaves. -- Classification of Twill Weaves. -- 1. Continuous Twills -- (a) Warp-face Twills -- (b) Weft-face Twills -- (c) Warp and Weft-face Twills -- The Angle of Twill -- Influences affecting the Prominence of Twills and Kindred Weaves (a) Character of Weave, (b) Character of Yarn, (c) Number of Threads per Inch, (d) Direction of Twill in Relation to the Direction of Twist in Yarn -- 2. Zigzag or Wavy Twills -- 3. Rearranged Twills: Satin Weaves -- Table of Intervals of Selection for the Construction of Satin Weaves -- Corkscrew Twills -- Rearrangement of Twill Weaves on Satin and other Bases -- 4. Combined Twills -- 5. Broken Twills -- 6. Figured or Ornamented Twills.
Chapter IV., Diamond and Kindred Weaves, -- Diamond Weaves. -- Honeycomb and Kindred Weaves -- Brighton Weaves -- Sponge Weaves -- Huck-a-Back and Kindred Weaves -- Grecian Weaves -- Linear Zigzag Weaves.
Chapter (p. c20) V., Bedford Cords. -- Plain Calico-ribbed Bedford Cords -- Plain Twill-ribbed Bedford Cords -- Figured Bedford Cords -- Tabulated Data of Particulars relating to the Manufacture of Seventeen Varieties of Bedford Cord Fabrics described in this Chapter.
Chapter VI., Backed Fabrics. -- Weft-backed Fabrics -- Warp-backed Fabrics -- Reversible or Double-faced Fabrics.
Chapter VII., Fustians. -- Varieties of Fustians. -- Imperials or Swansdowns -- Cantoons or Diagonals -- Moleskins -- Beaverteens -- Velveteens and Velveteen Cutting -- Ribbed or Corded Velveteen -- Figured Velveteen -- Corduroy -- Figured Corduroy -- Corduroy Cutting Machines.
Chapter VIII., Terry Pile Fabrics. -- Methods of producing Terry Pile on Textile Fabrics -- Terry-forming Devices -- Varieties of Terry Fabrics -- Action of the Reed in Relation to Shedding -- Figured Terry Weaving -- Practical Details of Terry Weaving.
Chapter IX., Gauze and Leno Fabrics. -- Gauze, Net Leno, and Leno Brocade Varieties of Cross-Weaving. -- Plain Gauze, and a Heald Gauze or Leno Harness -- Net Leno Fabrics -- Gauze and Net Leno Figuring by means of several Back Standard Healds to each Doup Heald -- Leno Specialities produced by a System of Crossing Warp Ends in front of the Reed -- A Device for the Production of Special Leno Effects -- Full Cross Leno Fabrics -- Relative Merits of a Top and a Bottom Doup Harness -- Relative Merits of Different Types of Dobbies for Gauze and Leno Fabrics -- Shaking Devices for Leno Weaving -- Practical Details of Leno Weaving -- Tempered Steel-wire Doup Harnesses for Cross-weaving -- Mock or Imitation Leno Fabrics.
Chapter X., Tissue, Lappet, and Swivel Figuring; also Ondulé Effects, and Looped Fabrics. -- Tissue Figuring -- Madras Muslin Curtains -- Lappet Figuring -- Spot Lappet Figuring -- Swivel Figuring -- Woven Ondulé Effects -- Loom for Weaving Ondulé Effects -- Weft Ondulé Effects -- Looped Fabrics. -- Index.
NEEDLEWORK AND DESIGN. By Miss M. E. Wilkinson. Quarto. 24 Plates and Text. 52 pp. [In the Press.]
HOME LACE-MAKING. A Handbook for Teachers and Pupils. By M. E. W. Milroy. Crown 8vo. 64 pp. With 3 Plates and 9 Diagrams. Price 1s. net. (Post free, 1s. 3d. home; 1s. 4d. abroad.)
THE CHEMISTRY OF HAT MANUFACTURING. Lectures delivered before the Hat Manufacturers' Association. By Watson Smith, F.C.S., F.I.C. Revised and Edited by Albert Shonk, Crown 8vo. 132 pp. 16 Illustrations. Price 7s. 6d. net. (Post free, 7s. 9d. home; 7s. 10d. abroad.)
Contents.
Textile Fibres, principally Wool, Fur, and Hair -- Water: its Chemistry and Properties; Impurities and their Action; Tests of Purity -- Acids and Alkalis -- Boric Acid, Borax, Soap -- Shellac, Wood Spirit, and the Stiffening and Proofing Process -- Mordants: their Nature and Use -- Dye-stuffs and Colours -- Dyeing of Wool and Fur; and Optical Properties of Colours-Index.
THE TECHNICAL TESTING OF YARNS AND TEXTILE FABRICS. With Reference to Official Specifications. Translated from the German of Dr. J. Herzfeld. Second Edition. Sixty-nine Illustrations. 200 pp. Demy 8vo. Price 10s. 6d. net. (Post free, 10s. 10d. home; 11s. abroad.)
Contents.
Yarn Testing. Determining the Yarn Number -- Testing the Length of Yarns -- Examination of the External Appearance of Yarn -- Determining the Twist of Yarn and Twist -- Determination of Tensile Strength and Elasticity -- Estimating the Percentage of Fat in Yarn -- Determination of Moisture (Conditioning) -- Appendix.
DECORATIVE AND FANCY TEXTILE FABRICS. By R. T. Lord. Manufacturers and Designers of Carpets, Damask, Dress and all Textile Fabrics. 200 pp. Demy 8vo. 132 Designs and Illustrations. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
THEORY AND PRACTICE OF DAMASK WEAVING. By H. Kinzer and K. Walter. Royal 8vo. Eighteen Folding Plates Six Illustrations. Translated from the German. 110 pp. Price 8s. 6d. net. (Post free, 9s. home; 9s. 6d. abroad.)
The Various Sorts of Damask Fabrics -- Drill (Ticking, Handloom-made) -- Whole Damask for Tablecloths -- Damask with Ground- and Connecting-warp Threads -- Furniture Damask -- Lampas or Hangings -- Church Damasks -- The Manufacture of Whole Damask -- Damask Arrangement with and without Cross-Shedding -- The Altered Cone-arrangement -- The Principle of the Corner Lifting Cord -- The Roller Principle -- The Combination of the Jacquard with the so-called Damask Machine -- The Special Damask Machine -- The Combination of Two Tyings.
FAULTS IN THE MANUFACTURE OF WOOLLEN GOODS AND THEIR PREVENTION. By Nicolas Reiser. Translated from the Second German Edition. Crown 8vo. Sixty-three Illustrations. 170 pp. Price 5s. net. (Post free, 5s. 4d. home; 5s. 6d. abroad.)
Contents.
Improperly Chosen Raw Material or Improper Mixtures -- Wrong Treatment of the Material in Washing, Carbonisation, Drying, Dyeing and Spinning -- Improper Spacing of the Goods in the Loom -- Wrong Placing of Colours -- Wrong Weight or Width of the Goods -- Breaking of Warp and Weft Threads -- Presence of Doubles, Singles, Thick, Loose, and too Hard Twisted Threads as well as Tangles, Thick Knots and the Like -- Errors in Cross-weaving--Inequalities, i.e., Bands and Stripes -- Dirty Borders -- Defective Selvedges -- Holes and Buttons -- Rubbed Places -- Creases -- Spots -- Loose and Bad Colours -- Badly Dyed Selvedges -- Hard Goods -- Brittle Goods -- Uneven Goods -- Removal of Bands, Stripes, Creases and Spots.
SPINNING AND WEAVING CALCULATIONS, especially relating to Woollens. From the German of N. Reiser. Thirty-four Illustrations. Tables. 160 pp. Demy 8vo. 1904. Price 10s. 6d. net. (Post free, 10s. 10d. home; 11s. abroad.)
Contents.
Calculating the Raw Material -- Proportion of Different Grades of Wool to Furnish a Mixture at a Given Price -- Quantity to Produce a Given Length -- Yarn Calculations -- Yarn Number -- Working Calculations -- Calculating the Reed Count -- Cost of Weaving, etc.
WATERPROOFING OF FABRICS. By Dr. S. Mierzinski. Crown 8vo, 104 pp. 29 Illus. Price 5s. net. (Post free, 5s. 3d. home; 5s. 4d. abroad.)
Contents.
Introduction -- Preliminary Treatment of the Fabric -- Waterproofing with Acetate of Alumina -- Impregnation of the Fabric -- Drying -- Waterproofing with Paraffin -- Waterproofing with Ammonium Cuprate -- Waterproofing with Metallic Oxides -- Coloured Waterproof Fabrics -- Waterproofing with Gelatine, Tannin, Caseinate of Lime and other Bodies -- Manufacture of Tarpaulin -- British Waterproofing Patents -- Index.
HOW TO MAKE A WOOLLEN MILL PAY. By John Mackie. Crown 8vo. 76 pp. Price 3s. 6d. net. (Post free, 3s. 9d. home; 3s. 10d. abroad.)
Contents.
Blends, Piles, or Mixtures of Clean Scoured Wools -- Dyed Wool Book -- The Order Book -- Pattern Duplicate Books -- Management and Oversight -- Constant Inspection of Hill Departments -- Importance of Delivering Goods to Time, Shade, Strength, etc. -- Plums.
(For "Textile Soaps and Oils" see p. 7.)
THE COLOUR PRINTING OF CARPET YARNS. Manual for Colour Chemists and Textile Printers. By David Paterson, F.C.S. Seventeen Illustrations. 136 pp. Demy 8vo. Price 7s. 6d. net. (Post free, 7s. 10d, home; 8s. abroad.)
Contents.
Structure and Constitution of Wool Fibre -- Yarn Scouring -- Scouring Materials -- Water for Scouring -- Bleaching Carpet Yarns -- Colour Making for Yarn Printing -- Colour Printing Pastes -- Colour Recipes for Yarn Printing -- Science of Colour Mixing -- Matching of Colours -- "Hank" Printing -- Printing Tapestry Carpet Yarns -- Yarn Printing -- Steaming Printed Yarns -- Washing of Steamed Yarns -- Aniline Colours Suitable for Yarn Printing -- Glossary of Dyes and Dye-wares used in Wood Yarn Printing -- Appendix.
THE (p. c22) SCIENCE OF COLOUR MIXING. A Manual intended for the use of Dyers, Calico Printers and Colour Chemists. By David Paterson, F.C.S. Forty-one Illustrations, Five Coloured Plates, and Four Plates showing Eleven Dyed Specimens of Fabrics. 132 pp. Demy 8vo. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Colour a Sensation; Colours of Illuminated Bodies; Colours of Opaque and Transparent Bodies; Surface Colour -- Analysis of Light; Spectrum; Homogeneous Colours; Ready Method of Obtaining a Spectrum -- Examination of Solar Spectrum; The Spectroscope and Its Construction; Colourists' Use of the Spectroscope -- Colour by Absorption: Solutions and Dyed Fabrics; Dichroic Coloured Fabrics in Gaslight -- Colour Primaries of the Scientist versus the Dyer and Artist; Colour Mixing by Rotation and Lye Dyeing; Hue, Purity, Brightness; Tints; Shades, Scales, Tones, Sad and Sombre Colours -- Colour Mixing; Pure and Impure Greens, Orange and Violets; Large Variety of Shades from few Colours; Consideration of the Practical Primaries: Red, Yellow and Blue -- Secondary Colours; Nomenclature of Violet and Purple Group; Tints and Shades of Violet; Changes in Artificial Light -- Tertiary Shades; Broken Hues; Absorption Spectra of Tertiary Shades -- Appendix: Four Plates with Dyed Specimens Illustrating Text -- Index.
DYERS' MATERIALS: An Introduction to the Examination, Evaluation and Application of the most important Substances used in Dyeing, Printing, Bleaching and Finishing. By Paul Heerman, Ph.D. Translated from the German by A. C. Wright, M.A. (Oxon.), B.Sc. (Lond.). Twenty-four Illustrations. Crown 8vo. 150 pp. Price 5s. net. (Post free, 5s. 4d. home; 5s. 6d. abroad.)
COLOUR MATCHING ON TEXTILES. A Manual intended for the use of Students of Colour Chemistry, Dyeing and Textile Printing. By David Paterson, F.C.S. Coloured Frontispiece. Twenty-nine Illustrations and Fourteen Specimens Of Dyed Fabrics. Demy 8vo. 132 pp. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Colour Vision and Structure of the Eye -- Perception of Colour -- Primary and Complementary Colour Sensations -- Daylight for Colour Matching -- Selection of a Good Pure Light -- Diffused Daylight, Direct Sunlight, Blue Skylight, Variability of Daylight, etc., etc. -- Matching of Hues -- Purity and Luminosity of Colours -- Matching Bright Hues -- Aid of Tinted Films -- Matching Difficulties Arising from Contrast -- Examination of Colours by Reflected and Transmitted Lights -- Effect of Lustre and Transparency of Fibres in Colour Matching -- Matching of Colours on Velvet Pile -- Optical Properties of Dye-stuffs, Dichroism, Fluorescence -- Use of Tinted Mediums -- Orange Film -- Defects of the Eye -- Yellowing of the Lens -- Colour Blindness, etc. -- Matching of Dyed Silk Trimmings and Linings and Bindings -- Its Difficulties -- Behaviour of Shades in Artificial Light -- Colour Matching of Old Fabrics, etc. -- Examination of Dyed Colours under the Artificial Lights -- Electric Arc, Magnesium and Dufton, Gardner Lights, Welsbach, Acetylene, etc. -- Testing Qualities of an Illuminant -- Influence of the Absorption Spectrum in Changes of Hue under the Artificial Lights -- Study of the Causes of Abnormal Modifications of Hue, etc.
COLOUR: A HANDBOOK OF THE THEORY OF COLOUR. By George H. Hurst, F.C.S. With Ten Coloured Plates and Seventy-two Illustrations. 160 pp. Demy 8vo. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Colour and Its Production -- Cause of Colour in Coloured Bodies -- Colour Phenomena and Theories -- The Physiology of Light -- Contrast -- Colour in Decoration and Design -- Measurement of Colour.
Reissue of
THE ART OF DYEING WOOL, SILK AND COTTON. Translated from the French of M. Hellot, M. Macquer and M. le Pileur D'Apligny. First Published in English in 1789. Six Plates. Demy 8vo. 446 pp. Price 5s. net. (Post free, 5s. 6d. home; 6s. abroad.)
Contents.
Part I., The Art of Dyeing Wool and Woollen Cloth, Stuffs, Yarn, Worsted, etc. Part II., The Art of Dyeing Silk. Part III., The Art of Dyeing Cotton and Linen Thread, together with the Method of Stamping Silks, Cottons, etc.
THE (p. c23) CHEMISTRY OF DYE-STUFFS. By Dr. Georg Von Georgievics. Translated from the Second German Edition. 412 pp. Demy 8vo. Price 10s. 6d. net. (Post free, 11s. home; 11s. 6d. abroad.)
Contents.
Introduction -- Coal Tar -- Intermediate Products in the Manufacture of Dye-stuffs--The Artificial Dye-stuffs (Coal-tar Dyes) -- Nitroso Dye-stuffs -- Nitro Dye-stuffs -- Azo Dye-stuffs -- Substantive Cotton Dye-stuffs -- Azoxystilbene Dye-stuffs -- Hydrazones -- Ketoneimides -- Triphenylmethane Dye-stuffs -- Rosolic Acid Dye-stuffs -- Xanthene Dye-stuffs -- Xanthone Dye-stuffs -- Flavones -- Oxyketone Dye-stuffs -- Quinoline and Acridine Dye-stuffs -- Quinonimide or Diphenylamine Dye-stuffs -- The Azine Group: Eurhodines, Safranines and Indulines -- Eurhodines -- Safranines -- Quinoxalines -- Indigo -- Dye-stuffs of Unknown Constitution -- Sulphur or Sulphine Dye stuffs -- Development of the Artificial Dye-stuff Industry -- The Natural Dye-stuffs -- Mineral Colours -- Index.
THE DYEING OF COTTON FABRICS: A Practical Handbook for the Dyer and Student. By Franklin Beech, Practical Colourist and Chemist. 272 pp. Forty-four Illustrations of Bleaching and Dyeing Machinery. Demy 8vo. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Structure and Chemistry of the Cotton Fibre -- Scouring and Bleaching of Cotton --Dyeing Machinery and Dyeing Manipulations -- Principles and Practice of Cotton Dyeing -- Direct Dyeing; Direct Dyeing followed by Fixation with Metallic Salts; Direct Dyeing followed by Fixation with Developers; Direct Dyeing followed by Fixation with Couplers; Dyeing on Tannic Mordant; Dyeing on Metallic Mordant; Production of Colour Direct upon Cotton Fibres; Dyeing Cotton by Impregnation with Dye-stuff Solution -- Dyeing Union (Mixed Cotton and Wool) Fabrics -- Dyeing Half Silk (Cotton-Silk, Satin) Fabrics -- Operations following Dyeing -- Washing, Soaping, Drying -- Testing of the Colour of Dyed Fabrics -- Experimental Dyeing and Comparative Dye Testing -- Index.
The book contains numerous recipes for the production on Cotton Fabrics of all kinds of a great range of colours.
THE DYEING OF WOOLLEN FABRICS. By Franklin Beech, Practical Colourist and Chemist. Thirty-three Illustrations. Demy 8vo. 228 pp. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
The Wool Fibre -- Structure, Composition and Properties -- Processes Preparatory to Dyeing -- Scouring and Bleaching of Wool -- Dyeing Machinery and Dyeing Manipulations -- Loose Wool Dyeing, Yarn Dyeing and Piece Dyeing Machinery -- The Principles and Practice of Wool Dyeing -- Properties of Wool Dyeing -- Methods of Wool Dyeing -- Groups of Dyes -- Dyeing with the Direct Dyes -- Dyeing with Basic Dyes -- Dyeing with Acid Dyes -- Dyeing with Mordant Dyes -- Level Dyeing -- Blacks on Wool -- Reds on Wool -- Mordanting of Wool -- Orange Shades on Wool -- Yellow Shades on Wool -- Green Shades on Wool -- Blue Shades on Wool -- Violet Shades on Wool -- Brown Shades on Wool -- Mode Colours on Wool -- Dyeing Union (Mixed Cotton Wool) Fabrics -- Dyeing of Gloria -- Operations following Dyeing -- Washing, Soaping, Drying -- Experimental Dyeing and Comparative Dye Testing -- Testing of the Colour of Dyed Fabrics -- Index.
A PRACTICAL TREATISE ON THE BLEACHING OF LINEN AND COTTON YARN AND FABRICS. By L. Tailfer, Chemical and Mechanical Engineer. Translated from the French by John Geddes McIntosh. Demy 8vo. 303 pp. Twenty Illus. Price 12s. 6d. net. (Post free, 13s. home; 13s. 6d. abroad.)
COTTON SPINNING (First Year). By Thomas Thornley, Spinning Master, Bolton Technical School. 160 pp. Eighty-four Illustrations. Crown 8vo. Second Impression. Price 3s. net. (Post free, 3s. 4d. home; 3s. 6d. abroad.)
Contents.
Syllabus and Examination Papers of the City and Guilds of London Institute -- Cultivation, Classification, Ginning, Baling and Mixing of the Raw Cotton -- Bale-Breakers, Mixing Lattices and Hopper Feeders -- Opening and Scutching -- Carding -- Indexes.
COTTON (p. c24) SPINNING (Intermediate, or Second Year). By Thomas Thornley. 180 pp. Seventy Illustrations. Crown 8vo. Price 5s. net. (Post free, 5s. 4d. home; 5s. 6d. abroad.)
Contents.
Syllabuses and Examination Papers of the City and Guilds of London Institute -- The Combing Process -- The Drawing Frame -- Bobbin and Fly Frames -- Mule Spinning -- Ring Spinning -- General Indexes.
COTTON SPINNING (Honours, or Third Year). By Thomas Thornley. 216 pp. Seventy-four Illustrations. Crown 8vo. Second Edition. Price 5s. net. (Post free, 5s. 4d. home; 5s. 6d. abroad.)
Contents.
Syllabuses and Examination Papers of the City and Guilds of London Institute -- Cotton--The Practical Manipulation of Cotton Spinning Machinery -- Doubling and Winding -- Reeling -- Warping -- Production and Costs -- Main Driving -- Arrangement of Machinery and Mill Planning -- Waste and Waste Spinning -- Indexes.
COTTON COMBING MACHINES. By Thos. Thornley, Spinning Master, Technical School, Bolton. Demy 8vo. 117 Illustrations. 300 pp. Price 7s. 6d. net. (Post free, 8s. home; 8s. 6d. abroad.)
Contents.
The Sliver Lap Machine and the Ribbon Cap Machine -- General Description of the Heilmann Comber -- The Cam Shaft -- On the Detaching and Attaching Mechanism of the Comber -- Resetting of Combers -- The Erection of a Heilmann Comber -- Stop Motions: Various Calculations -- Various Notes and Discussions -- Cotton Combing Machines of Continental Make -- Index.
MODERN FLAX, HEMP AND JUTE SPINNING AND TWISTING. A Practical Handbook for the use of Flax, Hemp and Jute Spinners, Thread, Twine and Rope Makers. By Herbert R. Carter, Mill Manager, Textile Expert and Engineer, Examiner in Flax Spinning to the City and Guilds of London Institute. Demy 8vo. 1907. With 92 Illustrations. 200 pp. Price 7s. 6d. net. (Post free, 7s. 9d. home; 8s. abroad.)
Contents.
Raw Fibre. -- Origin of Flax -- Hemp and Jute Fibre -- Description of the Plants -- Mode of Cultivation -- Suitable Climate and Soil -- Sowing -- Harvesting -- Rippling Flax and Hemp -- Water Retting -- Dew Retting -- Extraction of the Fibre -- Marketing the Fibre -- Bracquing -- Flax, Hemp and Jute Marks -- Comparative Prices -- Ports of Export -- Trade Centres -- Fibre Selling Conditions -- Duty on Fibre -- Fibre Exports. Hackling. -- Sorting and Storing the Raw Fibre -- Softening Hemp and Jute -- Jute Batching -- Cutting -- Piecing Out -- Roughing -- Hackling by Hand and Machine -- Tippling -- Sorting -- Ventilation of Hackling Rooms. Sliver Formation. -- Spreading Line -- Heavy Spreading System -- Good's Combined Hackle and Spreader -- Jute Breaking and Carding -- Flax and Hemp Tow Carding -- Bell Calculation -- Clock System -- Theory of Spreading. Line and Tow Preparing. -- Drawing and Doubling -- Draft Calculation -- Set Calculation -- Tow Combing -- Compound Systems -- Automatic Stop Motions and Independent Heads -- Details of Preparing Machinery -- Ventilation -- Humidification. Gill Spinning. -- Gill Spinning for Shoe Threads, Rope Yarns, Binder and Trawl Twines -- The Automatic Gill Spinner -- Rope and Reaper Yarn Numbering. The Flax, Hemp and Jute Roving Frame. -- Bobbin Winding -- Differential Motion -- Twist Calculation -- Practical Changing -- Rove Stock. Dry and Half-Dry Spinning. -- Flyer and Ring Frames -- Draft and Twist Calculation -- Bobbin Dragging -- Reaches -- Set of Breast Beam and Tin-rod. Wet Spinning of Flax, Hemp and Tow -- Hot and Cold Water Spinning -- Improvements in the Water Trough -- Turn off and Speed of Spindles -- Reaches -- Belting -- Band Tying -- Tape Driving -- Oiling -- Black Threads -- Cuts per Spindle -- Ventilation of the Wet Spinning Room. Yarn Department. -- Reeling -- Cop Winding -- Cheese and Spool Winding -- Balling Shoe Thread, Reaper Yarn, etc. -- Yarn Drying and Conditioning -- Yarn Bundling -- Yarn Baling -- Weight of Yarn -- Yarn Tables -- Duty on Yarn Imports. Manufacture of Threads, Twines and Cords. -- Hank Winding -- Wet and Dry Twisting -- Cabling -- Fancy Yarns -- Twine Laying -- Sizing and Polishing Threads and Twines -- Softening Threads -- Skeining Threads -- Balling Twines -- Leeson's Universal Winder -- Randing Twines -- Spooling Sewing Threads -- Comparative Prices of Flax and Hemp Cords, Lines and Threads. Rope Making. -- Construction of Hawsers and Cables -- Stranding -- Laying and Closing -- Compound Rope Machines -- Rules for Rope Makers -- Weight of Ropes -- Balling and Coiling Ropes. Mechanical Department. -- Boilers, Engines and Turbines -- Power Transmission by Belts and Ropes -- Electric Light and Power Transmission -- Fans -- Oils and Oiling -- Repairs -- Fluting. Mill Construction. -- Flax, Hemp and Jute Spinning Mills and Rope works -- Heating -- Roofs -- Chimneys, etc.
RECOVERY WORK AFTER PIT FIRES. By Robert Lamprecht, Mining Engineer and Manager. Translated from the German. Illustrated by Six large Plates, containing Seventy-six Illustrations. 175 pp., demy 8vo. Price 10s. 6d. net. (Post free, 10s. 10d. home; 11s. abroad.)
Contents.
Causes of Pit Fires -- Preventive Regulations: (1) The Outbreak and Rapid Extension of a Shaft Fire can be most reliably prevented by Employing little or no Combustible Material in the Construction of the Shaft; (2) Precautions for Rapidly Localising an Outbreak of Fire in the Shaft; (3) Precautions to be Adopted in case those under 1 and 2 Fail or Prove Inefficient. Precautions against Spontaneous Ignition of Coal. Precautions for Preventing Explosions of Fire-damp and Coal Dust. Employment of Electricity in Mining, particularly in Fiery Pits. Experiments on the Ignition of Fire-damp Mixtures and Clouds of Coal Dust by Electricity -- Indications of an Existing or Incipient Fire -- Appliances for Working in Irrespirable Gases: Respiratory Apparatus; Apparatus with Air Supply Pipes; Reservoir Apparatus; Oxygen Apparatus -- Extinguishing Pit Fires: (a) Chemical Means; (b) Extinction with Water. Dragging down the Burning Masses and Packing with Clay; (c) Insulating the Seat of the Fire by Dams. Dam Building. Analyses of Fire Gases. Isolating the Seat of a Fire with Dams: Working in Irrespirable Gases ("Gas-diving"): Air-Lock Work. Complete Isolation of the Pit. Flooding a Burning Section isolated by means of Dams. Wooden Dams: Masonry Dams. Examples of Cylindrical and Dome-shaped Dams. Dam Doors: Flooding the Whole Pit -- Rescue Stations: (a) Stations above Ground; (b) Underground Rescue Stations -- Spontaneous Ignition of Coal in Bulk -- Index.
VENTILATION IN MINES. By Robert Wabner, Mining Engineer. Translated from the German. Royal 8vo. Thirty Plates and Twenty-two Illustrations. 240 pp. Price 10s. 6d. net. (Post free, 11s. home; 11s. 3d. abroad.)
Contents.
The Causes of the Contamination of Pit Air -- The Means of Preventing the Dangers resulting from the Contamination of Pit Air -- Calculating the Volume of Ventilating Current necessary to free Pit Air from Contamination -- Determination of the Resistance Opposed to the Passage of Air through the Pit -- Laws of Resistance and Formulæ therefor -- Fluctuations in the Temperament or Specific Resistance of a Pit -- Means for Providing a Ventilating Current in the Pit -- Mechanical Ventilation -- Ventilators and Fans -- Determining the Theoretical, Initial, and True (Effective) Depression of the Centrifugal Fan -- New Types of Centrifugal Fan of Small Diameter and High Working Speed -- Utilising the Ventilating Current to the utmost Advantage and distributing the same through the Workings -- Artificially retarding the Ventilating Current -- Ventilating Preliminary Workings -- Blind Headings -- Separate Ventilation -- Supervision of Ventilation -- Index.
HAULAGE AND WINDING APPLIANCES USED IN MINES. By Carl Volk. Translated from the German. Royal 8vo. With Six Plates and 148 Illustrations. 150 pp. Price 8s. 6d. net. (Post free, 9s. home; 9s. 3d. abroad.)
Contents.
Haulage Appliances -- Ropes -- Haulage Tubs and Tracks -- Cages and Winding Appliances -- Winding Engines for Vertical Shafts -- Winding without Ropes -- Haulage in Levels and Inclines -- The Working of Underground Engines -- Machinery for Downhill Haulage.
DENTAL METALLURGY: MANUAL FOR STUDENTS AND DENTISTS. By A. B. Griffiths, Ph.D. Demy 8vo. Thirty-six Illustrations. 200 pp. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Introduction -- Physical Properties of the Metals -- Action of Certain Agents on Metals -- Alloys -- Action of Oral Bacteria on Alloys -- Theory and Varieties of Blowpipes -- Fluxes -- Furnaces and Appliances -- Heat and Temperature -- Gold -- Mercury -- Silver -- Iron -- Copper -- Zinc -- Magnesium -- Cadmium -- Tin -- Lead -- Aluminium -- Antimony -- Bismuth -- Palladium -- Platinum -- Iridium -- Nickel -- Practical Work -- Weights and Measures.
THE PREVENTION OF SMOKE. Combined with the Economical Combustion of Fuel. By W. C. Popplewell, M.Sc., A.M. Inst., C.E., Consulting Engineer. Forty-six Illustrations. 190 pp. Demy 8vo. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. 3d. abroad.)
Contents.
Fuel and Combustion -- Hand Firing in Boiler Furnaces -- Stoking by Mechanical Means -- Powdered Fuel -- Gaseous Fuel -- Efficiency and Smoke Tests of Boilers -- Some Standard Smoke Trials -- The Legal Aspect of the Smoke Question -- The Best Means to be adopted for the Prevention of Smoke -- Index.
GAS AND COAL DUST FIRING. A Critical Review of the Various Appliances Patented in Germany for this purpose since 1885. By Albert Pütsch. 130 pp. Demy 8vo. Translated from the German. With 103 Illustrations. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Generators -- Generators Employing Steam -- Stirring and Feed Regulating Appliances -- Direct Generators -- Burners -- Regenerators and Recuperators -- Glass Smelting Furnaces -- Metallurgical Furnaces -- Pottery Furnace -- Coal Dust Firing -- Index.
THE HARDENING AND TEMPERING OF STEEL IN THEORY AND PRACTICE. By Fridolin Reiser. Translated from the German of the Third Edition. Crown 8vo. 120 pp. Price 5s. net. (Post free, 5s. 3d. home; 5s. 4d. abroad.)
Contents.
Steel -- Chemical and Physical Properties of Steel, and their Casual Connection -- Classification of Steel according to Use -- Testing the Quality of Steel -- Steel-Hardening -- Investigation of the Causes of Failure in Hardening -- Regeneration of Steel Spoilt in the Furnace -- Welding Steel -- Index.
SIDEROLOGY: THE SCIENCE OF IRON (The Constitution of Iron Alloys and Slags). Translated from German of Hanns Freiherr v. Jüptner. 350 pp. Demy 8vo. Eleven Plates and Ten Illustrations. Price 10s. 6d. net. (Post free, 11s. home; 11s. 6d. abroad.)
Contents.
The Theory of Solution. -- Solutions -- Molten Alloys -- Varieties of Solutions -- Osmotic Pressure -- Relation between Osmotic Pressure and other Properties of Solutions -- Osmotic Pressure and Molecular Weight of the Dissolved Substance -- Solutions of Gases -- Solid Solutions -- Solubility -- Diffusion -- Electrical Conductivity -- Constitution of Electrolytes and Metals -- Thermal Expansion. Micrography. -- Microstructure -- The Micrographic Constituents of Iron -- Relation between Micrographical Composition, Carbon-Content, and Thermal Treatment of Iron Alloys -- The Microstructure of Slags. Chemical Composition of the Alloys of Iron. -- Constituents of Iron Alloys -- Carbon -- Constituents of the Iron Alloys, Carbon -- Opinions and Researches on Combined Carbon -- Applying the Curves of Solution deduced from the Curves of Recalescence to the Determination of the Chemical Composition of the Carbon present in Iron Alloys -- The Constituents of Iron -- Iron -- The Constituents of Iron Alloys -- Manganese -- Remaining Constituents of Iron Alloys -- A Silicon -- Gases. The Chemical Composition of Slag. -- Silicate Slags -- Calculating the Composition of Silicate Slags -- Phosphate Slags -- Oxide Slags -- Appendix -- Index.
EVAPORATING, CONDENSING AND COOLING APPARATUS. Explanations, Formulæ and Tables for Use in Practice. By E. Hausbrand, Engineer. Translated by A. C. Wright, M.A. (Oxon.), B.Sc. (Lond.). With Twenty-one Illustrations and Seventy-six Tables. 400 pp. Demy 8vo. Price 10s. 6d. net. (Post free, 11s. home; 11s. 6d. abroad.)
Contents (p. c27) of "Evaporating, Condensing: and Cooling-Apparatus".<7p>
ReCoefficient of Transmission of Heat, k/, and the Mean Temperature Difference, [**Greek: th θ]/m -- Parallel and Opposite Currents -- Apparatus for Heating with Direct Fire -- The Injection of Saturated Steam -- Superheated Steam -- Evaporation by Means of Hot Liquids -- The Transference of Heat in General, and Transference by means of Saturated Steam in Particular -- The Transference of Heat from Saturated Steam in Pipes (Coils) and Double Bottoms -- Evaporation in a Vacuum -- The Multiple-effect Evaporator -- Multiple-effect Evaporators from which Extra Steam is Taken -- The Weight of Water which must be Evaporated from 100 Kilos, of Liquor in order its Original Percentage of Dry Materials from 1-25 per cent. up to 20-70 per cent. -- The Relative Proportion of the Heating Surfaces in the Elements of the Multiple Evaporator and their Actual Dimensions -- The Pressure Exerted by Currents of Steam and Gas upon Floating Drops of Water -- The Motion of Floating Drops of Water upon which Press Currents of Steam -- The Splashing of Evaporating Liquids -- The Diameter of Pipes for Steam, Alcohol, Vapour and Air -- The Diameter of Water Pipes -- The Loss of Heat, from Apparatus and Pipes to the Surrounding Air, and Means for Preventing the Loss -- Condensers -- Heating Liquids by Means of Steam -- The Cooling of Liquids -- The Volumes to be Exhausted from Condensers by the Air-pumps -- A Few Remarks on Air-pumps and the Vacua they Produce -- The Volumetric Efficiency of Air-pumps -- The Volumes of Air which must be Exhausted from a Vessel in order to Reduce its Original Pressure to a Certain Lower Pressure -- Index.
EXTERNAL PLUMBING WORK. A Treatise on Lead Work for Roofs. By John W. Hart, R.P.C. 180 Illustrations. 272 pp. Demy 8vo. Second Edition Revised. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Cast Sheet Lead -- Milled Sheet Lead -- Roof Cesspools -- Socket Pipes -- Drips -- Gutters -- Gutters (continued) -- Breaks -- Circular Breaks -- Flats -- Flats (continued) -- Rolls on Flats -- Roll Ends -- Roll Intersections -- Seam Rolls -- Seam Rolls (continued) -- Tack Fixings -- Step Flashings -- Step Flashings (continued) -- Secret Gutters -- Soakers -- Hip and Valley Soakers -- Dormer Windows -- Dormer Windows (continued) -- Dormer Tops -- Internal Dormers -- Skylights -- Hips and Ridging -- Hips and Ridging (continued) -- Fixings for Hips and Ridging -- Ornamental Ridging -- Ornamental Curb Rolls -- Curb Rolls -- Cornices -- Towers and Finials -- Towers and Finials (continued) -- Towers and Finials (continued) -- Domes -- Domes (continued) -- Ornamental Lead Work -- Rain Water Heads -- Rain Water Heads (continued) -- Rain Water Heads (continued).
HINTS TO PLUMBERS ON JOINT WIPING, PIPE BENDING AND LEAD BURNING. Third Edition, Revised and Corrected. By John W. Hart, R.P.C. 184 Illustrations. 313 pp. Demy 8vo. Price 7s. 6d. net. (Post free, 8s. home; 8s. 6d. abroad.)
Contents.
Pipe Bending -- Pipe Bending (continued) -- Pipe Bending (continued) -- Square Pipe Bendings-- Half-circular Elbows -- Curved Bends on Square Pipe -- Bossed Bends -- Curved Plinth Bends -- Rain-water Shoes on Square Pipe -- Curved and Angle Bends -- Square Pipe Fixings -- Joint-wiping -- Substitutes for Wiped Joints -- Preparing Wiped Joints -- Joint Fixings -- Plumbing Irons -- Joint Fixings -- Use of "Touch" in Soldering -- Underhand Joints -- Blown and Copper Bit Joints -- Branch Joints -- Branch Joints (continued) -- Block Joints -- Block Joints (continued) -- Block Fixings -- Astragal Joints -- Pipe Fixings -- Large Branch Joints -- Large Underhand Joints -- Solders -- Autogenous Soldering or Lead Burning -- Index.
SANITARY PLUMBING AND DRAINAGE. By John W. Hart. Demy 8vo. With 208 Illustrations. 250 pp. 1904, Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)
Contents.
Sanitary Surveys -- Drain Testing -- Drain Testing with Smoke -- Testing Drains with Water -- Drain Plugs for Testing -- Sanitary Defects -- Closets -- Baths and Lavatories -- House Drains--Manholes -- Iron Soil Pipes -- Lead Soil Pipes -- Ventilating Pipes -- Water-closets -- Flushing Cisterns -- Baths -- Bath Fittings -- Lavatories -- Lavatory Fittings -- Sinks -- Waste Pipes -- Water Supply -- Ball Valves -- Town House Sanitary Arrangements -- Drainage -- Jointing Pipes -- Accessible Drains -- Iron Drains -- Iron Junctions -- Index.
THE (p. c28) PRINCIPLES AND PRACTICE OF DIPPING, BURNISHING, LACQUERING AND BRONZING BRASS WARE. By W. Norman Brown. 35 pp. Crown 8vo. Price 2s. net. (Post free, 2s. 3d. home and abroad.)
A HANDBOOK ON JAPANNING AND ENAMELLING FOR CYCLES, BEDSTEADS, TINWARE, ETC. By William Norman Brown. 52 pp. and Illustrations. Crown 8vo. Price 2s. net. (Post free, 2s. 3d. home and abroad.)
THE PRINCIPLES OF HOT WATER SUPPLY. By John W. Hart, R.P.C. With 129 Illustrations. 177 pp., demy 8vo. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. abroad.)<7p>
Contents.
Water Circulation -- The Tank System -- Pipes and Joints -- The Cylinder System -- Boilers for the Cylinder System -- The Cylinder System -- The Combined Tank and Cylinder System -- Combined Independent and Kitchen Boiler -- Combined Cylinder and Tank System with Duplicate Boilers -- Indirect Heating and Boiler Explosions -- Pipe Boilers -- Safety Valves -- Safety Valves -- The American System -- Heating Water by Steam -- Steam Kettles and Jets -- Heating Power of Steam -- Covering for Hot Water Pipes -- Index.
THREE HUNDRED SHADES FOR DECORATORS AND HOW TO MIX THEM. By A. Desaint. Quarto. The book will consist of a wide range of shades and tints suitable for decorators carefully numbered and mounted for easy reference, with full particulars as to the composition of each shade.
[In the press.]
HOUSE DECORATING AND PAINTING. By W. Norman Brown. Eighty-eight Illustrations. 150 pp. Crown 8vo. Price 3s. 6d. net. (Post free, 3s. 9d, home and abroad.)
A HISTORY OF DECORATIVE ART. By W. Norman Brown. Thirty-nine Illustrations. 96 pp. Crown 8vo. Price 2s. 6d. net. (Post free, 2s. 9d. home and abroad.)
WORKSHOP WRINKLES for Decorators, Painters, Paper-hangers and Others. By W. N. Brown. Crown 8vo. 128 pp. Second Edition. Price 2s. 6d. net. (Post free, 2s. 9d. home; 2s. 10d. abroad.)
HOPS IN THEIR BOTANICAL, AGRICULTURAL AND TECHNICAL ASPECT, AND AS AN ARTICLE OF COMMERCE. By Emmanuel Gross, Professor at the Higher Agricultural College, Tetschen-Liebwerd. Translated from the German. Seventy-eight Illustrations. 340 pp. Demy 8vo. Price 12s. 6d. net. (Post free, 13s. home; 13s. 6d. abroad.)
Contents.
HISTORY OF THE HOP -- THE HOP PLANT -- Introductory -- The Roots -- The Stem -- and Leaves -- Inflorescence and Flower: Inflorescence and Flower of the Male Hop; Inflorescence and Flower of the Female Hop -- The Fruit and its Glandular Structure: The Fruit (p. c29) and Seed -- Propagation and Selection of the Hop -- Varieties of the Hop: (a) Red Hops; (b) Green Hops; (c) Pale Green Hops -- Classification according to the Period of Ripening: Early August Hops; Medium Early Hops; Late Hops -- Injuries to Growth -- Leaves Turning Yellow, Summer or Sunbrand, Cones Dropping Off, Honey Dew, Damage from Wind, Hail and Rain; Vegetable Enemies of the Hop: Animal Enemies of the Hop -- Beneficial Insects on Hops -- CULTIVATION -- The Requirements of the Hop in Respect of Climate, Soil and Situation: Climate; Soil; Situation -- Selection of Variety and Cuttings -- Planting a Hop Garden: Drainage; Preparing the Ground; Marking-out for Planting; Planting; Cultivation and Cropping of the Hop Garden in the First Year -- Work to be Performed Annually in the Hop Garden: Working the Ground; Cutting; The Non-cutting System; The Proper Performance of the Operation of Cutting: Method of Cutting: Close Cutting, Ordinary Cutting, The Long Cut, The Topping Cut; Proper Season for Cutting: Autumn Cutting, Spring Cutting; Manuring; Training the Hop Plant: Poled Gardens, Frame Training; Principal Types of Frames; Pruning, Cropping, Topping, and Leaf Stripping the Hop Plant; Picking, Drying and Bagging -- Principal and Subsidiary Utilisation of Hops and Hop Gardens -- Life of a Hop Garden; Subsequent Cropping -- Cost of Production, Yield and Selling Prices.
Preservation and Storage -- Physical and Chemical Structure of the Hop Cone -- Judging the Value of Hops.
Statistics of Production -- The Hop Trade -- Index.
TIMBER: A Comprehensive Study of Wood in all its Aspects (Commercial and Botanical), showing the Different Applications and Uses of Timber in Various Trades, etc. Translated from the French of Paul Charpentier. Royal 8vo. 437 pp. 178 Illustrations. Price 12s. 6d. net. (Post free, 13s. home; 14s. abroad.)
Contents.
Physical and Chemical Properties of Timber -- Composition of the Vegetable Bodies -- Chief Elements -- M. Fremy's Researches -- Elementary Organs of Plants and especially of Forests -- Different Parts of Wood Anatomically and Chemically Considered -- General Properties of Wood -- Description of the Different Kinds of Wood -- Principal Essences with Caducous Leaves -- Coniferous Resinous Trees -- Division of the Useful Varieties of Timber in the Different Countries of the Globe -- European Timber -- African Timber -- Asiatic Timber -- American Timber -- Timber of Oceania -- Forests -- General Notes as to Forests; their Influence -- Opinions as to Sylviculture -- Improvement of Forests -- Unwooding and Rewooding -- Preservation of Forests -- Exploitation of Forests -- Damage caused to Forests -- Different Alterations -- The Preservation of Timber -- Generalities -- Causes and Progress of Deterioration -- History of Different Proposed Processes -- Dessication -- Superficial Carbonisation of Timber -- Processes by Immersion -- Generalities as to Antiseptics Employed -- Injection Processes in Closed Vessels -- The Boucherie System, Based upon the Displacement of the Sap -- Processes for Making Timber Uninflammable -- Applications of Timber -- Generalities -- Working Timber -- Paving -- Timber for Mines -- Railway Traverses -- Accessory Products -- Gums -- Works of M. Fremy -- Resins -- Barks -- Tan -- Application of Cork -- The Application of Wood to Art and Dyeing -- Different Applications of Wood -- Hard Wood -- Distillation of Wood -- Pyroligneous Acid -- Oil of Wood -- Distillation of Resins -- Index.
THE UTILISATION OF WOOD WASTE. Translated from the German of Ernst Hubbard. Crown 8vo. 192 pp. Fifty Illustrations. Price 5s. net. (Post free, 5s. 4d. home; 5s. 6d. abroad.)
Contents.
General Remarks on the Utilisation of Sawdust -- Employment of Sawdust as Fuel, with and without Simultaneous Recovery of Charcoal and the Products of Distillation -- Manufacture of Oxalic Acid from Sawdust -- Process with Soda Lye; Thorn's Process; Bohlig's Process -- Manufacture of Spirit (Ethyl Alcohol) from Wood Waste -- Patent Dyes (Organic Sulphides, Sulphur Dyes, or Mercapto Dyes) -- Artificial Wood and Plastic Compositions from Sawdust -- Production of Artificial Wood Compositions for Moulded Decorations -- Employment of Sawdust for Blasting Powders and Gunpowders -- Employment of Sawdust for Briquettes -- Employment of Sawdust in the Ceramic Industry and as an Addition to Mortar -- Manufacture of Paper Pulp from Wood -- Casks -- Various Applications of Sawdust and Wood Refuse -- Calcium Carbide -- Manure -- Wood Mosaic Plaques -- Bottle Stoppers -- Parquetry -- Fire-lighters -- Carborundum -- The Production of Wood Wool -- Bark -- Index.
THE PREVENTION OF DAMPNESS IN BUILDINGS; with Remarks on the Causes, Nature and Effects of Saline, Efflorescences and Dry-rot, for Architects, Builders, Overseers, Plasterers Painters and House Owners. By Adolf Wilhelm Keim. Translated from the German of the second revised Edition by M. J. Salter, F.I.C. F.C.S. Eight Coloured Plates and Thirteen Illustrations. Crown 8vo. 115 pp. Price 5s. net. (Post free, 5s. 3d. home; 5s. 4d. abroad.)
Contents.
The Various Causes of Dampness and Decay of the Masonry of Buildings, and the Structural and Hygienic Evils of the Same -- Precautionary Measures during Building against Dampness and Efflorescence -- Methods of Remedying Dampness and Efflorescences in the Walls of Old Buildings -- The Artificial Drying of New Houses, as well as Old Damp Dwellings and the Theory of the Hardening of Mortar -- New, Certain and Permanently Efficient Methods for Drying Old Damp Walls and Dwellings -- The Cause and Origin of Dry-rot: its Injurious Effect on Health, its Destructive Action on Buildings, and its Successful Repression -- Methods of Preventing Dry-rot to be Adopted During Construction -- Old Methods of Preventing Dry-rot -- Recent and More Efficient Remedies for Dry-rot -- Index.
HANDBOOK OF TECHNICAL TERMS USED IN ARCHITECTURE AND BUILDING, AND THEIR ALLIED TRADES AND SUBJECTS. By Augustine C. Passmore. Demy 8vo. 380 pp. Price 7s. 6d. net. (Post free, 8s. home; 8s. 6d, abroad.)
THE MANUFACTURE OF PRESERVED FOODS AND SWEETMEATS. By A. Hausner. With Twenty-eight Illustrations. Translated from the German of the third enlarged Edition. Crown 8vo. 225 pp. Price 7s. 6d. net. (Post free, 7s. 9d. home; 7s. 10d. abroad.)
Contents.
The Manufacture of Conserves -- Introduction -- The Causes of the Putrefaction of Food -- The Chemical Composition of Foods -- The Products of Decomposition -- The Causes of Fermentation and Putrefaction -- Preservative Bodies -- The Various Methods of Preserving Food -- The Preservation of Animal Food -- Preserving Meat by Means of Ice -- The Preservation of Meat by Charcoal -- Preservation of Meat by Drying -- The Preservation of Meat by the Exclusion of Air -- The Appert Method -- Preserving Flesh by Smoking -- Quick Smoking -- Preserving Meat with Salt -- Quick Salting by Air Pressure -- Quick Salting by Liquid Pressure -- Gamgee's Method of Preserving Meat -- The Preservation of Eggs -- Preservation of White and Yolk of Egg -- Milk Preservation -- Condensed Milk -- The Preservation of Fat -- Manufacture of Soup Tablets -- Meat Biscuits -- Extract of Beef -- The Preservation of Vegetable Foods in General -- Compressing Vegetables -- Preservation of Vegetables by Appert's Method -- The Preservation of Fruit -- Preservation of Fruit by Storage -- The Preservation of Fruit by Drying -- Drying Fruit by Artificial Heat -- Roasting Fruit -- The Preservation of Fruit with Sugar -- Boiled Preserved Fruit -- The Preservation of Fruit in Spirit, Acetic Acid or Glycerine -- Preservation of Fruit without Boiling -- Jam Manufacture -- The Manufacture of Fruit Jellies -- The Making of Gelatine Jellies -- The Manufacture of "Sulzen" -- The Preservation of Fermented Beverages -- The Manufacture of Candies -- Introduction -- The Manufacture of Candied Fruit -- The Manufacture of Boiled Sugar and Caramel -- The Candying of Fruit -- Caramelised Fruit -- The Manufacture of Sugar Sticks, or Barley Sugar -- Bonbon Making -- Fruit Drops -- The Manufacture of Dragées -- The Machinery and Appliances used in Candy Manufacture -- Dyeing Candies and Bonbons -- Essential Oils used in Candy Making -- Fruit Essences -- The Manufacture of Filled Bonbons, Liqueur Bonbons and Stamped Lozenges -- Recipes for Jams and Jellies -- Recipes for Bonbon Making -- Dragées -- Appendix -- Index.
THE ART OF DYEING AND STAINING MARBLE, ARTIFICIAL STONE, BONE, HORN, IVORY AND WOOD, AND OF IMITATING ALL SORTS OF WOOD. A Practical Handbook for the Use of Joiners, Turners, Manufacturers of Fancy Goods, Stick and Umbrella Makers, Comb Makers, etc. Translated from the German of D. H. Soxhlet, Technical Chemist. Crown 8vo. 168 pp. Price 5s. net. (Post free, 5s. 3d. home; 5s. 4d. abroad.)
Contents.
Mordants and Stains -- Natural Dyes -- Artificial Pigments -- Coal Tar Dyes -- Staining Marble and Artificial Stone -- Dyeing, Bleaching and Imitation of Bone, Horn and Ivory -- Imitation of Tortoiseshell for Combs: Yellows, Dyeing Nuts -- Ivory -- Wood Dyeing -- Imitation of Mahogany: Dark Walnut, Oak, Birch-Bark, Elder-Marquetry, Walnut, Walnut-Marquetry, Mahogany, Spanish Mahogany, Palisander and Rose Wood, Tortoiseshell, Oak, Ebony, Pear Tree -- Black Dyeing Processes with Penetrating Colours -- Varnishes and Polishes: English Furniture Polish, Vienna Furniture Polish, Amber Varnish, Copal Varnish, Composition for Preserving Furniture -- Index.
CELLULOID. The Raw Material, Manufacture and Uses. By Dr. Fr. Böckmann. 49 Illus. Crown 8vo.
[In the Press.]
PRACTICAL LITHOGRAPHY. By Alfred Seymour. Demy 8vo. With Frontispiece and 33 Illus. 120 pp. Price 5s. net. (Post free, 5s. 4d. home; 5s. 6d. abroad.)
Contents.
Stones -- Transfer Inks -- Transfer Papers -- Transfer Printing -- Litho Press -- Press Work -- Machine Printing -- Colour Printing -- Substitutes for Lithographic Stones -- Tin Plate Printing and Decoration -- Photo-Lithography.
PRINTERS' AND STATIONERS' READY RECKONER AND COMPENDIUM. Compiled by Victor Graham. Crown 8vo. 112 pp. 1904. Price 3s. 6d. net. (Post free, 3s. 9d. home; 3s. 10d. abroad.)
Contents.
Price of Paper per Sheet, Quire, Ream and Lb. -- Cost of 100 to 1000 Sheets at various Sizes and Prices per Ream -- Cost of Cards -- Quantity Table -- Sizes and Weights of Paper, Cards, etc. -- Notes on Account Books -- Discount Tables -- Sizes of spaces -- Leads to a lb. -- Dictionary -- Measure for Bookwork -- Correcting Proofs, etc.
ENGRAVING FOR ILLUSTRATION. HISTORICAL AND PRACTICAL NOTES. By J. Kirkbride. 72 pp. Two Plates and 6 Illustrations. Crown 8vo. Price 2s. 6d. net. (Post free, 2s. 9d. home; 2s. 10d. abroad.)
Contents.
Its Inception -- Wood Engraving -- Metal Engraving -- Engraving in England -- Etching -- Mezzotint -- Photo-Process Engraving -- The Engraver's Task -- Appreciative Criticism -- Index.
PRACTICAL BOOKBINDING. By Paul Adam. Translated from the German. Crown 8vo. 180 pp. 127 Illustrations. Price 5s. net. (Post free, 5s. 4d. home; 5s. 6d. abroad.)
Contents.
Materials for Sewing and Pasting -- Materials for Covering the Book -- Materials for Decorating and Finishing -- Tools -- General Preparatory Work -- Sewing -- Forwarding, Cutting, (p. c32) Rounding and Backing -- Forwarding, Decoration of Edges and Headbanding -- Boarding -- Preparing the Cover -- Work with the Blocking Press -- Treatment of Sewn Books, Fastening in Covers, and Finishing Off -- Handtooling and Other Decoration -- Account Books -- School Books, Mounting Maps, Drawings, etc. -- Index.
THE TECHNOLOGY OF SUGAR: Practical Treatise on the Modern Methods of Manufacture of Sugar from the Sugar Cane and Sugar Beet. By John Geddes McIntosh. Second Revised and Enlarged Edition. Demy 8vo. Fully Illustrated. 436 pp. Seventy-six Tables. 1906. Price 10s. 6d. net. (Post free, 11s. home; 11s. 6d. abroad.)
Contents.
Chemistry of Sucrose, Lactose, Maltose, Glucose, Invert Sugar, etc. -- Purchase and Analysis of Beets -- Treatment of Beets -- Diffusion -- Filtration -- Concentration -- Evaporation -- Sugar Cane: Cultivation -- Milling -- Diffusion -- Sugar Refining -- Analysis of Raw Sugars -- Chemistry of Molasses, etc.
(See "Evaporating, Condensing, etc., Apparatus.")
CLASSIFIED GUIDE TO TECHNICAL AND COMMERCIAL BOOKS. Compiled by Edgar Greenwood. Demy 8vo. 224 pp. 1904. Being a Subject-list of the Principal British and American Books in print; giving Title, Author, Size, Date, Publisher and Price. Price 7s. 6d. net. (Post free, 7s. 10d. home; 8s. 3d. abroad.)
THE TECHNICAL BOOKS in this Catalogue can be obtained through all Booksellers, or post free direct from the Publishers by remitting the amount given in brackets.
Full Particulars of Contents of any of the above books will be sent on application.
Books In the Press.--The Publishers will send Contents and prices of books in the press as soon as ready to any one sending their address.
Technical Books upon all Subjects can be obtained through Scott, Greenwood & Son, if requirements are fully stated.
|
Transcriber's note: A less obvious printer's error has been corrected on page 16. - chlorestine to cholesterine. The following words could not be checked: - Caseogum (page c04), - Crysophis (page c16), - Dufton (page c22), - Bracquing (page c24). |
***END OF THE PROJECT GUTENBERG EBOOK THE DYEING OF WOOLLEN FABRICS***
******* This file should be named 19985-h.txt or 19985-h.zip *******
This and all associated files of various formats will be found in:
http://www.gutenberg.org/1/9/9/8/19985
Updated editions will replace the previous one--the old editions will be renamed.
Creating the works from public domain print editions means that no one owns a United States copyright in these works, so the Foundation (and you!) can copy and distribute it in the United States without permission and without paying copyright royalties. Special rules, set forth in the General Terms of Use part of this license, apply to copying and distributing Project Gutenberg-tm electronic works to protect the PROJECT GUTENBERG-tm concept and trademark. Project Gutenberg is a registered trademark, and may not be used if you charge for the eBooks, unless you receive specific permission. If you do not charge anything for copies of this eBook, complying with the rules is very easy. You may use this eBook for nearly any purpose such as creation of derivative works, reports, performances and research. They may be modified and printed and given away--you may do practically ANYTHING with public domain eBooks. Redistribution is subject to the trademark license, especially commercial redistribution.
*** START: FULL LICENSE ***
THE FULL PROJECT GUTENBERG LICENSE
PLEASE READ THIS BEFORE YOU DISTRIBUTE OR USE THIS WORK
To protect the Project Gutenberg-tm mission of promoting the free
distribution of electronic works, by using or distributing this work
(or any other work associated in any way with the phrase "Project
Gutenberg"), you agree to comply with all the terms of the Full Project
Gutenberg-tm License (available with this file or online at
http://www.gutenberg.org/license).
Section 1. General Terms of Use and Redistributing Project Gutenberg-tm
electronic works
1.A. By reading or using any part of this Project Gutenberg-tm
electronic work, you indicate that you have read, understand, agree to
and accept all the terms of this license and intellectual property
(trademark/copyright) agreement. If you do not agree to abide by all
the terms of this agreement, you must cease using and return or destroy
all copies of Project Gutenberg-tm electronic works in your possession.
If you paid a fee for obtaining a copy of or access to a Project
Gutenberg-tm electronic work and you do not agree to be bound by the
terms of this agreement, you may obtain a refund from the person or
entity to whom you paid the fee as set forth in paragraph 1.E.8.
1.B. "Project Gutenberg" is a registered trademark. It may only be
used on or associated in any way with an electronic work by people who
agree to be bound by the terms of this agreement. There are a few
things that you can do with most Project Gutenberg-tm electronic works
even without complying with the full terms of this agreement. See
paragraph 1.C below. There are a lot of things you can do with Project
Gutenberg-tm electronic works if you follow the terms of this agreement
and help preserve free future access to Project Gutenberg-tm electronic
works. See paragraph 1.E below.
1.C. The Project Gutenberg Literary Archive Foundation ("the Foundation"
or PGLAF), owns a compilation copyright in the collection of Project
Gutenberg-tm electronic works. Nearly all the individual works in the
collection are in the public domain in the United States. If an
individual work is in the public domain in the United States and you are
located in the United States, we do not claim a right to prevent you from
copying, distributing, performing, displaying or creating derivative
works based on the work as long as all references to Project Gutenberg
are removed. Of course, we hope that you will support the Project
Gutenberg-tm mission of promoting free access to electronic works by
freely sharing Project Gutenberg-tm works in compliance with the terms of
this agreement for keeping the Project Gutenberg-tm name associated with
the work. You can easily comply with the terms of this agreement by
keeping this work in the same format with its attached full Project
Gutenberg-tm License when you share it without charge with others.
1.D. The copyright laws of the place where you are located also govern
what you can do with this work. Copyright laws in most countries are in
a constant state of change. If you are outside the United States, check
the laws of your country in addition to the terms of this agreement
before downloading, copying, displaying, performing, distributing or
creating derivative works based on this work or any other Project
Gutenberg-tm work. The Foundation makes no representations concerning
the copyright status of any work in any country outside the United
States.
1.E. Unless you have removed all references to Project Gutenberg:
1.E.1. The following sentence, with active links to, or other immediate
access to, the full Project Gutenberg-tm License must appear prominently
whenever any copy of a Project Gutenberg-tm work (any work on which the
phrase "Project Gutenberg" appears, or with which the phrase "Project
Gutenberg" is associated) is accessed, displayed, performed, viewed,
copied or distributed:
This eBook is for the use of anyone anywhere at no cost and with
almost no restrictions whatsoever. You may copy it, give it away or
re-use it under the terms of the Project Gutenberg License included
with this eBook or online at www.gutenberg.org
1.E.2. If an individual Project Gutenberg-tm electronic work is derived
from the public domain (does not contain a notice indicating that it is
posted with permission of the copyright holder), the work can be copied
and distributed to anyone in the United States without paying any fees
or charges. If you are redistributing or providing access to a work
with the phrase "Project Gutenberg" associated with or appearing on the
work, you must comply either with the requirements of paragraphs 1.E.1
through 1.E.7 or obtain permission for the use of the work and the
Project Gutenberg-tm trademark as set forth in paragraphs 1.E.8 or
1.E.9.
1.E.3. If an individual Project Gutenberg-tm electronic work is posted
with the permission of the copyright holder, your use and distribution
must comply with both paragraphs 1.E.1 through 1.E.7 and any additional
terms imposed by the copyright holder. Additional terms will be linked
to the Project Gutenberg-tm License for all works posted with the
permission of the copyright holder found at the beginning of this work.
1.E.4. Do not unlink or detach or remove the full Project Gutenberg-tm
License terms from this work, or any files containing a part of this
work or any other work associated with Project Gutenberg-tm.
1.E.5. Do not copy, display, perform, distribute or redistribute this
electronic work, or any part of this electronic work, without
prominently displaying the sentence set forth in paragraph 1.E.1 with
active links or immediate access to the full terms of the Project
Gutenberg-tm License.
1.E.6. You may convert to and distribute this work in any binary,
compressed, marked up, nonproprietary or proprietary form, including any
word processing or hypertext form. However, if you provide access to or
distribute copies of a Project Gutenberg-tm work in a format other than
"Plain Vanilla ASCII" or other format used in the official version
posted on the official Project Gutenberg-tm web site (www.gutenberg.org),
you must, at no additional cost, fee or expense to the user, provide a
copy, a means of exporting a copy, or a means of obtaining a copy upon
request, of the work in its original "Plain Vanilla ASCII" or other
form. Any alternate format must include the full Project Gutenberg-tm
License as specified in paragraph 1.E.1.
1.E.7. Do not charge a fee for access to, viewing, displaying,
performing, copying or distributing any Project Gutenberg-tm works
unless you comply with paragraph 1.E.8 or 1.E.9.
1.E.8. You may charge a reasonable fee for copies of or providing
access to or distributing Project Gutenberg-tm electronic works provided
that
- You pay a royalty fee of 20% of the gross profits you derive from
the use of Project Gutenberg-tm works calculated using the method
you already use to calculate your applicable taxes. The fee is
owed to the owner of the Project Gutenberg-tm trademark, but he
has agreed to donate royalties under this paragraph to the
Project Gutenberg Literary Archive Foundation. Royalty payments
must be paid within 60 days following each date on which you
prepare (or are legally required to prepare) your periodic tax
returns. Royalty payments should be clearly marked as such and
sent to the Project Gutenberg Literary Archive Foundation at the
address specified in Section 4, "Information about donations to
the Project Gutenberg Literary Archive Foundation."
- You provide a full refund of any money paid by a user who notifies
you in writing (or by e-mail) within 30 days of receipt that s/he
does not agree to the terms of the full Project Gutenberg-tm
License. You must require such a user to return or
destroy all copies of the works possessed in a physical medium
and discontinue all use of and all access to other copies of
Project Gutenberg-tm works.
- You provide, in accordance with paragraph 1.F.3, a full refund of any
money paid for a work or a replacement copy, if a defect in the
electronic work is discovered and reported to you within 90 days
of receipt of the work.
- You comply with all other terms of this agreement for free
distribution of Project Gutenberg-tm works.
1.E.9. If you wish to charge a fee or distribute a Project Gutenberg-tm
electronic work or group of works on different terms than are set
forth in this agreement, you must obtain permission in writing from
both the Project Gutenberg Literary Archive Foundation and Michael
Hart, the owner of the Project Gutenberg-tm trademark. Contact the
Foundation as set forth in Section 3 below.
1.F.
1.F.1. Project Gutenberg volunteers and employees expend considerable
effort to identify, do copyright research on, transcribe and proofread
public domain works in creating the Project Gutenberg-tm
collection. Despite these efforts, Project Gutenberg-tm electronic
works, and the medium on which they may be stored, may contain
"Defects," such as, but not limited to, incomplete, inaccurate or
corrupt data, transcription errors, a copyright or other intellectual
property infringement, a defective or damaged disk or other medium, a
computer virus, or computer codes that damage or cannot be read by
your equipment.
1.F.2. LIMITED WARRANTY, DISCLAIMER OF DAMAGES - Except for the "Right
of Replacement or Refund" described in paragraph 1.F.3, the Project
Gutenberg Literary Archive Foundation, the owner of the Project
Gutenberg-tm trademark, and any other party distributing a Project
Gutenberg-tm electronic work under this agreement, disclaim all
liability to you for damages, costs and expenses, including legal
fees. YOU AGREE THAT YOU HAVE NO REMEDIES FOR NEGLIGENCE, STRICT
LIABILITY, BREACH OF WARRANTY OR BREACH OF CONTRACT EXCEPT THOSE
PROVIDED IN PARAGRAPH F3. YOU AGREE THAT THE FOUNDATION, THE
TRADEMARK OWNER, AND ANY DISTRIBUTOR UNDER THIS AGREEMENT WILL NOT BE
LIABLE TO YOU FOR ACTUAL, DIRECT, INDIRECT, CONSEQUENTIAL, PUNITIVE OR
INCIDENTAL DAMAGES EVEN IF YOU GIVE NOTICE OF THE POSSIBILITY OF SUCH
DAMAGE.
1.F.3. LIMITED RIGHT OF REPLACEMENT OR REFUND - If you discover a
defect in this electronic work within 90 days of receiving it, you can
receive a refund of the money (if any) you paid for it by sending a
written explanation to the person you received the work from. If you
received the work on a physical medium, you must return the medium with
your written explanation. The person or entity that provided you with
the defective work may elect to provide a replacement copy in lieu of a
refund. If you received the work electronically, the person or entity
providing it to you may choose to give you a second opportunity to
receive the work electronically in lieu of a refund. If the second copy
is also defective, you may demand a refund in writing without further
opportunities to fix the problem.
1.F.4. Except for the limited right of replacement or refund set forth
in paragraph 1.F.3, this work is provided to you 'AS-IS,' WITH NO OTHER
WARRANTIES OF ANY KIND, EXPRESS OR IMPLIED, INCLUDING BUT NOT LIMITED TO
WARRANTIES OF MERCHANTIBILITY OR FITNESS FOR ANY PURPOSE.
1.F.5. Some states do not allow disclaimers of certain implied
warranties or the exclusion or limitation of certain types of damages.
If any disclaimer or limitation set forth in this agreement violates the
law of the state applicable to this agreement, the agreement shall be
interpreted to make the maximum disclaimer or limitation permitted by
the applicable state law. The invalidity or unenforceability of any
provision of this agreement shall not void the remaining provisions.
1.F.6. INDEMNITY - You agree to indemnify and hold the Foundation, the
trademark owner, any agent or employee of the Foundation, anyone
providing copies of Project Gutenberg-tm electronic works in accordance
with this agreement, and any volunteers associated with the production,
promotion and distribution of Project Gutenberg-tm electronic works,
harmless from all liability, costs and expenses, including legal fees,
that arise directly or indirectly from any of the following which you do
or cause to occur: (a) distribution of this or any Project Gutenberg-tm
work, (b) alteration, modification, or additions or deletions to any
Project Gutenberg-tm work, and (c) any Defect you cause.
Section 2. Information about the Mission of Project Gutenberg-tm
Project Gutenberg-tm is synonymous with the free distribution of
electronic works in formats readable by the widest variety of computers
including obsolete, old, middle-aged and new computers. It exists
because of the efforts of hundreds of volunteers and donations from
people in all walks of life.
Volunteers and financial support to provide volunteers with the
assistance they need, is critical to reaching Project Gutenberg-tm's
goals and ensuring that the Project Gutenberg-tm collection will
remain freely available for generations to come. In 2001, the Project
Gutenberg Literary Archive Foundation was created to provide a secure
and permanent future for Project Gutenberg-tm and future generations.
To learn more about the Project Gutenberg Literary Archive Foundation
and how your efforts and donations can help, see Sections 3 and 4
and the Foundation web page at http://www.gutenberg.org/fundraising/pglaf.
Section 3. Information about the Project Gutenberg Literary Archive
Foundation
The Project Gutenberg Literary Archive Foundation is a non profit
501(c)(3) educational corporation organized under the laws of the
state of Mississippi and granted tax exempt status by the Internal
Revenue Service. The Foundation's EIN or federal tax identification
number is 64-6221541. Contributions to the Project Gutenberg
Literary Archive Foundation are tax deductible to the full extent
permitted by U.S. federal laws and your state's laws.
The Foundation's principal office is located at 4557 Melan Dr. S.
Fairbanks, AK, 99712., but its volunteers and employees are scattered
throughout numerous locations. Its business office is located at
809 North 1500 West, Salt Lake City, UT 84116, (801) 596-1887, email
business@pglaf.org. Email contact links and up to date contact
information can be found at the Foundation's web site and official
page at http://www.gutenberg.org/about/contact
For additional contact information:
Dr. Gregory B. Newby
Chief Executive and Director
gbnewby@pglaf.org
Section 4. Information about Donations to the Project Gutenberg
Literary Archive Foundation
Project Gutenberg-tm depends upon and cannot survive without wide
spread public support and donations to carry out its mission of
increasing the number of public domain and licensed works that can be
freely distributed in machine readable form accessible by the widest
array of equipment including outdated equipment. Many small donations
($1 to $5,000) are particularly important to maintaining tax exempt
status with the IRS.
The Foundation is committed to complying with the laws regulating
charities and charitable donations in all 50 states of the United
States. Compliance requirements are not uniform and it takes a
considerable effort, much paperwork and many fees to meet and keep up
with these requirements. We do not solicit donations in locations
where we have not received written confirmation of compliance. To
SEND DONATIONS or determine the status of compliance for any
particular state visit http://www.gutenberg.org/fundraising/pglaf
While we cannot and do not solicit contributions from states where we
have not met the solicitation requirements, we know of no prohibition
against accepting unsolicited donations from donors in such states who
approach us with offers to donate.
International donations are gratefully accepted, but we cannot make
any statements concerning tax treatment of donations received from
outside the United States. U.S. laws alone swamp our small staff.
Please check the Project Gutenberg Web pages for current donation
methods and addresses. Donations are accepted in a number of other
ways including checks, online payments and credit card donations.
To donate, please visit: http://www.gutenberg.org/fundraising/donate
Section 5. General Information About Project Gutenberg-tm electronic
works.
Professor Michael S. Hart is the originator of the Project Gutenberg-tm
concept of a library of electronic works that could be freely shared
with anyone. For thirty years, he produced and distributed Project
Gutenberg-tm eBooks with only a loose network of volunteer support.
Project Gutenberg-tm eBooks are often created from several printed
editions, all of which are confirmed as Public Domain in the U.S.
unless a copyright notice is included. Thus, we do not necessarily
keep eBooks in compliance with any particular paper edition.
Each eBook is in a subdirectory of the same number as the eBook's
eBook number, often in several formats including plain vanilla ASCII,
compressed (zipped), HTML and others.
Corrected EDITIONS of our eBooks replace the old file and take over
the old filename and etext number. The replaced older file is renamed.
VERSIONS based on separate sources are treated as new eBooks receiving
new filenames and etext numbers.
Most people start at our Web site which has the main PG search facility:
http://www.gutenberg.org
This Web site includes information about Project Gutenberg-tm,
including how to make donations to the Project Gutenberg Literary
Archive Foundation, how to help produce our new eBooks, and how to
subscribe to our email newsletter to hear about new eBooks.
EBooks posted prior to November 2003, with eBook numbers BELOW #10000,
are filed in directories based on their release date. If you want to
download any of these eBooks directly, rather than using the regular
search system you may utilize the following addresses and just
download by the etext year.
http://www.gutenberg.org/dirs/etext06/
(Or /etext 05, 04, 03, 02, 01, 00, 99,
98, 97, 96, 95, 94, 93, 92, 92, 91 or 90)
EBooks posted since November 2003, with etext numbers OVER #10000, are
filed in a different way. The year of a release date is no longer part
of the directory path. The path is based on the etext number (which is
identical to the filename). The path to the file is made up of single
digits corresponding to all but the last digit in the filename. For
example an eBook of filename 10234 would be found at:
http://www.gutenberg.org/dirs/1/0/2/3/10234
or filename 24689 would be found at:
http://www.gutenberg.org/dirs/2/4/6/8/24689
An alternative method of locating eBooks:
http://www.gutenberg.org/dirs/GUTINDEX.ALL
*** END: FULL LICENSE ***